User login
‘Tis the season … for fireworks injuries
according to the Consumer Product Safety Commission.
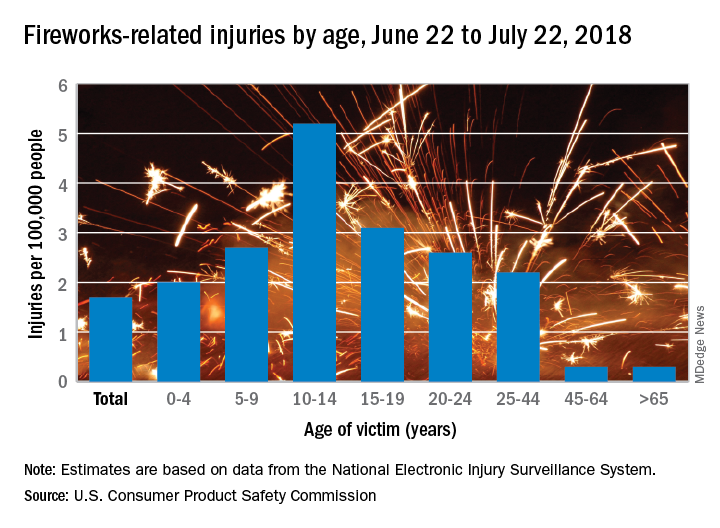
Of the estimated 9,100 fireworks-related injuries treated in emergency departments last year, 5,600 (62%) occurred between June 22 and July 22, 2018. That works out to a rate of 1.7 ED-treated injuries per 100,000 people for that 1 month and a rate of 2.8 per 100,000 for the entire year, the CPSC said in its 2018 Fireworks Annual Report.
Children had higher injury rates than adults in the Fourth of July window, and those aged 10-14 years had the highest rate of all, 5.2 injuries per 100,000 population. They were followed by teens aged 15-19 years (3.1 per 100,000) and children aged 5-9 (2.7 per 100,000), the CPSC investigators said based on data from the National Electronic Injury Surveillance System.
A deeper dive into the data pool shows that firecrackers caused more injuries – 19% of the total for the month – than any other type of firework device (reloadable shells were second at 12%). Burns were the most common type of injury, making up 44% of the total, and hands and fingers were the body parts most often injured (28% of the total), they reported.
There were five fireworks-related deaths last year – below the average of 7.6 per year since 2003 – but the total for 2018 may go up because reporting for the year is not yet complete. In one of the 2018 cases, an 18-year-old taped a tube to a football helmet and tried to launch a mortar shell while wearing the helmet. The first one worked, but the second shell got stuck and exploded in the tube, the CPSC said.
“CPSC works year-round to help prevent deaths and injuries from fireworks, by verifying fireworks meet safety regulations in our ports, marketplace, and on the road,” acting CPSC Chairman Ann Marie Buerkle said in a written statement. “Beyond CPSC’s efforts, we want to make sure everyone takes simple safety steps to celebrate safely with their family and friends.”
according to the Consumer Product Safety Commission.

Of the estimated 9,100 fireworks-related injuries treated in emergency departments last year, 5,600 (62%) occurred between June 22 and July 22, 2018. That works out to a rate of 1.7 ED-treated injuries per 100,000 people for that 1 month and a rate of 2.8 per 100,000 for the entire year, the CPSC said in its 2018 Fireworks Annual Report.
Children had higher injury rates than adults in the Fourth of July window, and those aged 10-14 years had the highest rate of all, 5.2 injuries per 100,000 population. They were followed by teens aged 15-19 years (3.1 per 100,000) and children aged 5-9 (2.7 per 100,000), the CPSC investigators said based on data from the National Electronic Injury Surveillance System.
A deeper dive into the data pool shows that firecrackers caused more injuries – 19% of the total for the month – than any other type of firework device (reloadable shells were second at 12%). Burns were the most common type of injury, making up 44% of the total, and hands and fingers were the body parts most often injured (28% of the total), they reported.
There were five fireworks-related deaths last year – below the average of 7.6 per year since 2003 – but the total for 2018 may go up because reporting for the year is not yet complete. In one of the 2018 cases, an 18-year-old taped a tube to a football helmet and tried to launch a mortar shell while wearing the helmet. The first one worked, but the second shell got stuck and exploded in the tube, the CPSC said.
“CPSC works year-round to help prevent deaths and injuries from fireworks, by verifying fireworks meet safety regulations in our ports, marketplace, and on the road,” acting CPSC Chairman Ann Marie Buerkle said in a written statement. “Beyond CPSC’s efforts, we want to make sure everyone takes simple safety steps to celebrate safely with their family and friends.”
according to the Consumer Product Safety Commission.

Of the estimated 9,100 fireworks-related injuries treated in emergency departments last year, 5,600 (62%) occurred between June 22 and July 22, 2018. That works out to a rate of 1.7 ED-treated injuries per 100,000 people for that 1 month and a rate of 2.8 per 100,000 for the entire year, the CPSC said in its 2018 Fireworks Annual Report.
Children had higher injury rates than adults in the Fourth of July window, and those aged 10-14 years had the highest rate of all, 5.2 injuries per 100,000 population. They were followed by teens aged 15-19 years (3.1 per 100,000) and children aged 5-9 (2.7 per 100,000), the CPSC investigators said based on data from the National Electronic Injury Surveillance System.
A deeper dive into the data pool shows that firecrackers caused more injuries – 19% of the total for the month – than any other type of firework device (reloadable shells were second at 12%). Burns were the most common type of injury, making up 44% of the total, and hands and fingers were the body parts most often injured (28% of the total), they reported.
There were five fireworks-related deaths last year – below the average of 7.6 per year since 2003 – but the total for 2018 may go up because reporting for the year is not yet complete. In one of the 2018 cases, an 18-year-old taped a tube to a football helmet and tried to launch a mortar shell while wearing the helmet. The first one worked, but the second shell got stuck and exploded in the tube, the CPSC said.
“CPSC works year-round to help prevent deaths and injuries from fireworks, by verifying fireworks meet safety regulations in our ports, marketplace, and on the road,” acting CPSC Chairman Ann Marie Buerkle said in a written statement. “Beyond CPSC’s efforts, we want to make sure everyone takes simple safety steps to celebrate safely with their family and friends.”
Is Melatonin a Biomarker in Episodic Migraine?
Urinary melatonin metabolites do not predict migraine attacks in children and adolescents, however, they may be predictive in those who experience premonitory phase symptoms as part of their migraine attacks. This according to a study that examined whether evening urinary melatonin metabolite levels could predict migraine the next day in children and adolescents with migraine. Among the details:
- Twenty-one children and adolescents with migraine were recruited to provide urine samples for 10 days and maintain a prospective headache diary during the same period.
- Mean aMT6s levels the night prior to a migraine attack were 56.2 ±39.0 vs 55.4 ±46.6 ng/mL.
- Mean melatonin metabolite levels the night following migraine were 55.5 ±46.9 vs 57.0 ±37.7 ng/mL.
- However, in post hoc exploratory analyses, aMT6s levels were lower the night before a migraine in those who experienced aura or premonitory symptoms.
Berger A, et al. Preliminary evidence that melatonin is not a biomarker in children and adolescents with episodic migraine. [Published online ahead of print May 3, 2019]. Headache. doi: 10.1111/head.13547.
Urinary melatonin metabolites do not predict migraine attacks in children and adolescents, however, they may be predictive in those who experience premonitory phase symptoms as part of their migraine attacks. This according to a study that examined whether evening urinary melatonin metabolite levels could predict migraine the next day in children and adolescents with migraine. Among the details:
- Twenty-one children and adolescents with migraine were recruited to provide urine samples for 10 days and maintain a prospective headache diary during the same period.
- Mean aMT6s levels the night prior to a migraine attack were 56.2 ±39.0 vs 55.4 ±46.6 ng/mL.
- Mean melatonin metabolite levels the night following migraine were 55.5 ±46.9 vs 57.0 ±37.7 ng/mL.
- However, in post hoc exploratory analyses, aMT6s levels were lower the night before a migraine in those who experienced aura or premonitory symptoms.
Berger A, et al. Preliminary evidence that melatonin is not a biomarker in children and adolescents with episodic migraine. [Published online ahead of print May 3, 2019]. Headache. doi: 10.1111/head.13547.
Urinary melatonin metabolites do not predict migraine attacks in children and adolescents, however, they may be predictive in those who experience premonitory phase symptoms as part of their migraine attacks. This according to a study that examined whether evening urinary melatonin metabolite levels could predict migraine the next day in children and adolescents with migraine. Among the details:
- Twenty-one children and adolescents with migraine were recruited to provide urine samples for 10 days and maintain a prospective headache diary during the same period.
- Mean aMT6s levels the night prior to a migraine attack were 56.2 ±39.0 vs 55.4 ±46.6 ng/mL.
- Mean melatonin metabolite levels the night following migraine were 55.5 ±46.9 vs 57.0 ±37.7 ng/mL.
- However, in post hoc exploratory analyses, aMT6s levels were lower the night before a migraine in those who experienced aura or premonitory symptoms.
Berger A, et al. Preliminary evidence that melatonin is not a biomarker in children and adolescents with episodic migraine. [Published online ahead of print May 3, 2019]. Headache. doi: 10.1111/head.13547.
Risk of cardiac events jumps after COPD exacerbation
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
particularly in older individuals, new research has found.
In Respirology, researchers report the outcomes of a nationwide, register-based study involving 118,807 patients with chronic obstructive pulmonary disease (COPD) who experienced a major adverse cardiac event after an exacerbation.
They found that the risk of any major cardiac adverse event increased 270% in the 4 weeks after the onset of an exacerbation (95% confidence interval, 3.60-3.80). The strongest association was seen for cardiovascular death, for which there was a 333% increase in risk, but there was also a 257% increase in the risk of acute MI and 178% increase in the risk of stroke.
The risk of major adverse cardiac events was even higher among individuals who were hospitalized because of their COPD exacerbation (odds ratio, 5.92), compared with a 150% increase in risk among those who weren’t hospitalized but were treated with oral corticosteroids and 108% increase among those treated with amoxicillin with enzyme inhibitors.
The risk of a major cardiac event after a COPD exacerbation also increased with age. Among individuals younger than 55 years, there was a 131% increase in risk, but among those aged 55-69 years there was a 234% increase, among those aged 70-79 years the risk increased 282%, and among those aged 80 years and older it increased 318%.
Mette Reilev, from the department of public health at the University of Southern Denmark, Odense, and coauthors suggested that acute exacerbations were associated with elevated levels of systemic inflammatory markers such as fibrinogen and interleukin-6, which were potently prothrombotic and could potentially trigger cardiovascular events.
“Additionally, exacerbations may trigger type II myocardial infarctions secondary to an imbalance in oxygen supply and demand,” they wrote.
The authors raised the question of whether cardiovascular prevention strategies should be part of treatment recommendations for people with COPD, and suggested that prevention of COPD exacerbations could be justified even on cardiovascular grounds alone.
“Studies investigating the effect of cardiovascular treatment on the course of disease among COPD exacerbators are extremely scarce,” they wrote. “Thus, it is currently unknown how to optimize treatment and mitigate the increased risk of [major adverse cardiovascular events] following the onset of exacerbations.”
However, they noted that prednisolone treatment for more severe exacerbations may have a confounding effect, as oral corticosteroids could induce dyslipidemia, hypertension, and hyperglycemia, and increase long-term cardiovascular risk.
Six authors declared funding from the pharmaceutical industry – three of which were institutional support – unrelated to the study.
SOURCE: Reilev M et al. Respirology. 2019 Jun 21. doi: 10.1111/resp.13620.
FROM RESPIROLOGY
Opioids: Overprescribing, alternatives, and clinical guidance
Vemurafenib has durable activity in NSCLC harboring BRAF V600 mutations
The oral BRAF(V600E) kinase inhibitor vemurafenib (Zelboraf) has durable activity in non–small cell lung cancer (NSCLC) harboring BRAF V600 mutations and possibly provides greater benefit for treatment-naive patients, found the phase 2, open-label, single-arm VE-BASKET trial.
“Targetable oncogenic drivers in NSCLC with robust clinical validation include EGFR mutations and ALK and ROS1 fusions, but identifying other targetable, clinically important subgroups of NSCLC is a high priority,” wrote the investigators, who were led by Vivek Subbiah, MD, of the University of Texas MD Anderson Cancer Center, Houston. Roughly 1%-4% of NSCLC patients have tumors harboring a BRAF V600 mutation, they noted.
Analyses were based on 62 patients with BRAF V600–mutant NSCLC enrolled in the trial’s NSCLC cohort or all-comers cohort. All received vemurafenib (960 mg twice daily) until disease progression or unacceptable toxicity.
Thirteen percent of the patients had not received any previous systemic therapy. Among those previously treated, the median number of systemic regimens received was two.
Results reported in JCO Precicion Oncology showed that the median treatment duration was 6.0 months for all patients (12.0 months for previously untreated patients and 5.7 months for previously treated patients).
The objective response rate, the trial’s primary endpoint, was 37.1% overall; it was similar in the previously untreated group and the previously treated group (37.5% and 37.0%, respectively). The clinical benefit rate was 48.4% overall, with a larger differential according to previous treatment at 62.5% and 46.3%.
Median progression-free survival was 6.5 months in all patients (12.9 months in those previously untreated and 6.1 months in those previously treated), and median overall survival was 15.4 months in all patients (not estimable in those previously untreated, but 15.4 months in those previously treated).
The most common adverse events of any grade were nausea (seen in 40% of patients), hyperkeratosis (34%), and decreased appetite (32%). The most common grade 3 or worse adverse event was anemia (10%). The safety profile generally resembled that previously observed among patients with melanoma, with no new signals.
“Vemurafenib showed promising activity in patients with NSCLC harboring BRAF V600 mutations,” Dr. Subbiah and colleagues concluded. “The prolonged [overall survival] … in the NSCLC population represents promising durability of effect with single-agent BRAF inhibition.”
“The apparent increase in median [progression-free survival] in previously untreated patients compared with previously treated patients warrants additional investigation of earlier treatment in this patient population,” they maintained.
Dr. Subbiah disclosed having a consulting or advisory role with or receiving research funding from numerous pharmaceutical companies. The study was sponsored by Hoffmann-La Roche.
SOURCE: Subbiah V et al. JCO Precis Oncol. 2019 June 27. doi: 10.1200/PO.18.00266.
The oral BRAF(V600E) kinase inhibitor vemurafenib (Zelboraf) has durable activity in non–small cell lung cancer (NSCLC) harboring BRAF V600 mutations and possibly provides greater benefit for treatment-naive patients, found the phase 2, open-label, single-arm VE-BASKET trial.
“Targetable oncogenic drivers in NSCLC with robust clinical validation include EGFR mutations and ALK and ROS1 fusions, but identifying other targetable, clinically important subgroups of NSCLC is a high priority,” wrote the investigators, who were led by Vivek Subbiah, MD, of the University of Texas MD Anderson Cancer Center, Houston. Roughly 1%-4% of NSCLC patients have tumors harboring a BRAF V600 mutation, they noted.
Analyses were based on 62 patients with BRAF V600–mutant NSCLC enrolled in the trial’s NSCLC cohort or all-comers cohort. All received vemurafenib (960 mg twice daily) until disease progression or unacceptable toxicity.
Thirteen percent of the patients had not received any previous systemic therapy. Among those previously treated, the median number of systemic regimens received was two.
Results reported in JCO Precicion Oncology showed that the median treatment duration was 6.0 months for all patients (12.0 months for previously untreated patients and 5.7 months for previously treated patients).
The objective response rate, the trial’s primary endpoint, was 37.1% overall; it was similar in the previously untreated group and the previously treated group (37.5% and 37.0%, respectively). The clinical benefit rate was 48.4% overall, with a larger differential according to previous treatment at 62.5% and 46.3%.
Median progression-free survival was 6.5 months in all patients (12.9 months in those previously untreated and 6.1 months in those previously treated), and median overall survival was 15.4 months in all patients (not estimable in those previously untreated, but 15.4 months in those previously treated).
The most common adverse events of any grade were nausea (seen in 40% of patients), hyperkeratosis (34%), and decreased appetite (32%). The most common grade 3 or worse adverse event was anemia (10%). The safety profile generally resembled that previously observed among patients with melanoma, with no new signals.
“Vemurafenib showed promising activity in patients with NSCLC harboring BRAF V600 mutations,” Dr. Subbiah and colleagues concluded. “The prolonged [overall survival] … in the NSCLC population represents promising durability of effect with single-agent BRAF inhibition.”
“The apparent increase in median [progression-free survival] in previously untreated patients compared with previously treated patients warrants additional investigation of earlier treatment in this patient population,” they maintained.
Dr. Subbiah disclosed having a consulting or advisory role with or receiving research funding from numerous pharmaceutical companies. The study was sponsored by Hoffmann-La Roche.
SOURCE: Subbiah V et al. JCO Precis Oncol. 2019 June 27. doi: 10.1200/PO.18.00266.
The oral BRAF(V600E) kinase inhibitor vemurafenib (Zelboraf) has durable activity in non–small cell lung cancer (NSCLC) harboring BRAF V600 mutations and possibly provides greater benefit for treatment-naive patients, found the phase 2, open-label, single-arm VE-BASKET trial.
“Targetable oncogenic drivers in NSCLC with robust clinical validation include EGFR mutations and ALK and ROS1 fusions, but identifying other targetable, clinically important subgroups of NSCLC is a high priority,” wrote the investigators, who were led by Vivek Subbiah, MD, of the University of Texas MD Anderson Cancer Center, Houston. Roughly 1%-4% of NSCLC patients have tumors harboring a BRAF V600 mutation, they noted.
Analyses were based on 62 patients with BRAF V600–mutant NSCLC enrolled in the trial’s NSCLC cohort or all-comers cohort. All received vemurafenib (960 mg twice daily) until disease progression or unacceptable toxicity.
Thirteen percent of the patients had not received any previous systemic therapy. Among those previously treated, the median number of systemic regimens received was two.
Results reported in JCO Precicion Oncology showed that the median treatment duration was 6.0 months for all patients (12.0 months for previously untreated patients and 5.7 months for previously treated patients).
The objective response rate, the trial’s primary endpoint, was 37.1% overall; it was similar in the previously untreated group and the previously treated group (37.5% and 37.0%, respectively). The clinical benefit rate was 48.4% overall, with a larger differential according to previous treatment at 62.5% and 46.3%.
Median progression-free survival was 6.5 months in all patients (12.9 months in those previously untreated and 6.1 months in those previously treated), and median overall survival was 15.4 months in all patients (not estimable in those previously untreated, but 15.4 months in those previously treated).
The most common adverse events of any grade were nausea (seen in 40% of patients), hyperkeratosis (34%), and decreased appetite (32%). The most common grade 3 or worse adverse event was anemia (10%). The safety profile generally resembled that previously observed among patients with melanoma, with no new signals.
“Vemurafenib showed promising activity in patients with NSCLC harboring BRAF V600 mutations,” Dr. Subbiah and colleagues concluded. “The prolonged [overall survival] … in the NSCLC population represents promising durability of effect with single-agent BRAF inhibition.”
“The apparent increase in median [progression-free survival] in previously untreated patients compared with previously treated patients warrants additional investigation of earlier treatment in this patient population,” they maintained.
Dr. Subbiah disclosed having a consulting or advisory role with or receiving research funding from numerous pharmaceutical companies. The study was sponsored by Hoffmann-La Roche.
SOURCE: Subbiah V et al. JCO Precis Oncol. 2019 June 27. doi: 10.1200/PO.18.00266.
FROM JCO PRECISION ONCOLOGY
Real-world experience with dupilumab in AD mirrors clinical trial efficacy
MILAN – A retrospective, multicenter Maria Fargnoli, MD, reported at the World Congress of Dermatology.
By the end of 4 weeks of treatment, participants’ mean Eczema Area and Severity Index (EASI) score had dropped from 33.3 to 15.3, a 54.2% reduction. At 16 weeks, the mean EASI score was 9.2, a reduction of 72.5% from baseline (P less than .001 for both time points, compared with baseline). At 16 weeks, 87.2% of patients achieved EASI 50, 60.6% achieved EASI 75, and 32.4% achieved EASI 90.
“In a real-life context, dupilumab significantly improved disease severity, pruritus, sleep loss, and quality of life in adult moderate to severe atopic dermatitis patients,” said Dr. Fargnoli, presenting results of the study during a late-breaking abstract session at the meeting. “All measures improved at 4 weeks, and a further decline was seen at 16 weeks. … These results confirm data from clinical trials, and from other real-life experiences with dupilumab.”
The study, conducted in Italy, tracked outcomes for 109 patients treated for moderate to severe atopic dermatitis at 39 centers from June 2018 to February 2019. Adult patients with EASI scores of at least 24 with contraindications, failure, or intolerance of corticosteroid therapy were included and followed for at least 16 weeks. Those who had concomitant systemic anti-inflammatory or immunomodulator use were excluded, as were those with missing data, said Dr. Fargnoli, chair of the department of dermatology at the University of L’Aquila (Italy).
Patients were given a loading dose of two 300-mg subcutaneous injections of dupilumab, followed by 300-mg injections at 2-week intervals.
Patients were assessed at baseline and after 4 and 16 weeks of treatment. In addition to EASI score, itch and sleep were measured via numeric rating scales; mean itch scores dropped from 8.4 at baseline to 4.1 after 4 weeks, and to 2.5 at 16 weeks (P less than .001 for both time points, compared with baseline).
Sleep scores also improved, from a mean 6.9 at baseline to 3.3 at four weeks, and 1.9 at 16 weeks (P less than .001 for both time points, compared with baseline).
Patients also completed the Dermatology Life Quality Index. At the 4-week mark, patients saw a reduction to 8.3 points from the baseline score of 17.6 points (out of a possible 30, with higher scores indicating worse quality of life); scores dropped to 5.4 by week 16 (P less than .001 for both time points, compared with baseline).
Dupilumab was generally well tolerated, with conjunctivitis – seen in 11% of patients – being the most commonly reported adverse event. This falls in line with other recently published real-world studies of dupilumab, Dr. Fargnoli noted.
Efficacy, as measured by EASI reduction and improvement in itch and sleep, were also comparable between the Italian cohort and clinical trial results, as well as other real-life studies in Denmark, France, the Netherlands, and Spain, she said.
Patients, about one-third female, had a mean body mass index of about 24 kg/m2. Mean age was about 38 years (range, 19-80 years). The mean age of disease onset was about 14 years (range, 0-77 years).
Atopic dermatitis was characterized by phenotype for each patient; groupings included classic adult type (73%), nummular dermatitis (7%), prurigo (8%), and erythrodermic dermatitis (12%). About three in four patients (76.1%) had facial involvement; 61.5% had hand involvement, and 22.9% had genital involvement.
Allergic comorbidities were reported by many patients; 44.9% had rhinitis, 38.5% had asthma, 33% had conjunctivitis, and 15.6% reported food allergies. Other notable comorbidities included psychiatric or psychological conditions, present in 11% of patients, and hypertension or other cardiovascular disorders, seen in 9.1% of patients.
Most patients had tried treatment with both cyclosporine A and corticosteroids (88.9% and 88.1%, respectively). Almost half (45.8%) had tried UV-light therapy, and about a quarter had tried methotrexate.
“The results give real-life data on patterns of treatment response according to heterogeneous atopic dermatitis phenotypes, and on long-term efficacy and safety,” said Dr. Fargnoli.
The study was not funded by any company, according to Dr. Fargnoli. She has served on the advisory board for and has received honoraria for lectures and research grants from Sanofi-Genzyme.
MILAN – A retrospective, multicenter Maria Fargnoli, MD, reported at the World Congress of Dermatology.
By the end of 4 weeks of treatment, participants’ mean Eczema Area and Severity Index (EASI) score had dropped from 33.3 to 15.3, a 54.2% reduction. At 16 weeks, the mean EASI score was 9.2, a reduction of 72.5% from baseline (P less than .001 for both time points, compared with baseline). At 16 weeks, 87.2% of patients achieved EASI 50, 60.6% achieved EASI 75, and 32.4% achieved EASI 90.
“In a real-life context, dupilumab significantly improved disease severity, pruritus, sleep loss, and quality of life in adult moderate to severe atopic dermatitis patients,” said Dr. Fargnoli, presenting results of the study during a late-breaking abstract session at the meeting. “All measures improved at 4 weeks, and a further decline was seen at 16 weeks. … These results confirm data from clinical trials, and from other real-life experiences with dupilumab.”
The study, conducted in Italy, tracked outcomes for 109 patients treated for moderate to severe atopic dermatitis at 39 centers from June 2018 to February 2019. Adult patients with EASI scores of at least 24 with contraindications, failure, or intolerance of corticosteroid therapy were included and followed for at least 16 weeks. Those who had concomitant systemic anti-inflammatory or immunomodulator use were excluded, as were those with missing data, said Dr. Fargnoli, chair of the department of dermatology at the University of L’Aquila (Italy).
Patients were given a loading dose of two 300-mg subcutaneous injections of dupilumab, followed by 300-mg injections at 2-week intervals.
Patients were assessed at baseline and after 4 and 16 weeks of treatment. In addition to EASI score, itch and sleep were measured via numeric rating scales; mean itch scores dropped from 8.4 at baseline to 4.1 after 4 weeks, and to 2.5 at 16 weeks (P less than .001 for both time points, compared with baseline).
Sleep scores also improved, from a mean 6.9 at baseline to 3.3 at four weeks, and 1.9 at 16 weeks (P less than .001 for both time points, compared with baseline).
Patients also completed the Dermatology Life Quality Index. At the 4-week mark, patients saw a reduction to 8.3 points from the baseline score of 17.6 points (out of a possible 30, with higher scores indicating worse quality of life); scores dropped to 5.4 by week 16 (P less than .001 for both time points, compared with baseline).
Dupilumab was generally well tolerated, with conjunctivitis – seen in 11% of patients – being the most commonly reported adverse event. This falls in line with other recently published real-world studies of dupilumab, Dr. Fargnoli noted.
Efficacy, as measured by EASI reduction and improvement in itch and sleep, were also comparable between the Italian cohort and clinical trial results, as well as other real-life studies in Denmark, France, the Netherlands, and Spain, she said.
Patients, about one-third female, had a mean body mass index of about 24 kg/m2. Mean age was about 38 years (range, 19-80 years). The mean age of disease onset was about 14 years (range, 0-77 years).
Atopic dermatitis was characterized by phenotype for each patient; groupings included classic adult type (73%), nummular dermatitis (7%), prurigo (8%), and erythrodermic dermatitis (12%). About three in four patients (76.1%) had facial involvement; 61.5% had hand involvement, and 22.9% had genital involvement.
Allergic comorbidities were reported by many patients; 44.9% had rhinitis, 38.5% had asthma, 33% had conjunctivitis, and 15.6% reported food allergies. Other notable comorbidities included psychiatric or psychological conditions, present in 11% of patients, and hypertension or other cardiovascular disorders, seen in 9.1% of patients.
Most patients had tried treatment with both cyclosporine A and corticosteroids (88.9% and 88.1%, respectively). Almost half (45.8%) had tried UV-light therapy, and about a quarter had tried methotrexate.
“The results give real-life data on patterns of treatment response according to heterogeneous atopic dermatitis phenotypes, and on long-term efficacy and safety,” said Dr. Fargnoli.
The study was not funded by any company, according to Dr. Fargnoli. She has served on the advisory board for and has received honoraria for lectures and research grants from Sanofi-Genzyme.
MILAN – A retrospective, multicenter Maria Fargnoli, MD, reported at the World Congress of Dermatology.
By the end of 4 weeks of treatment, participants’ mean Eczema Area and Severity Index (EASI) score had dropped from 33.3 to 15.3, a 54.2% reduction. At 16 weeks, the mean EASI score was 9.2, a reduction of 72.5% from baseline (P less than .001 for both time points, compared with baseline). At 16 weeks, 87.2% of patients achieved EASI 50, 60.6% achieved EASI 75, and 32.4% achieved EASI 90.
“In a real-life context, dupilumab significantly improved disease severity, pruritus, sleep loss, and quality of life in adult moderate to severe atopic dermatitis patients,” said Dr. Fargnoli, presenting results of the study during a late-breaking abstract session at the meeting. “All measures improved at 4 weeks, and a further decline was seen at 16 weeks. … These results confirm data from clinical trials, and from other real-life experiences with dupilumab.”
The study, conducted in Italy, tracked outcomes for 109 patients treated for moderate to severe atopic dermatitis at 39 centers from June 2018 to February 2019. Adult patients with EASI scores of at least 24 with contraindications, failure, or intolerance of corticosteroid therapy were included and followed for at least 16 weeks. Those who had concomitant systemic anti-inflammatory or immunomodulator use were excluded, as were those with missing data, said Dr. Fargnoli, chair of the department of dermatology at the University of L’Aquila (Italy).
Patients were given a loading dose of two 300-mg subcutaneous injections of dupilumab, followed by 300-mg injections at 2-week intervals.
Patients were assessed at baseline and after 4 and 16 weeks of treatment. In addition to EASI score, itch and sleep were measured via numeric rating scales; mean itch scores dropped from 8.4 at baseline to 4.1 after 4 weeks, and to 2.5 at 16 weeks (P less than .001 for both time points, compared with baseline).
Sleep scores also improved, from a mean 6.9 at baseline to 3.3 at four weeks, and 1.9 at 16 weeks (P less than .001 for both time points, compared with baseline).
Patients also completed the Dermatology Life Quality Index. At the 4-week mark, patients saw a reduction to 8.3 points from the baseline score of 17.6 points (out of a possible 30, with higher scores indicating worse quality of life); scores dropped to 5.4 by week 16 (P less than .001 for both time points, compared with baseline).
Dupilumab was generally well tolerated, with conjunctivitis – seen in 11% of patients – being the most commonly reported adverse event. This falls in line with other recently published real-world studies of dupilumab, Dr. Fargnoli noted.
Efficacy, as measured by EASI reduction and improvement in itch and sleep, were also comparable between the Italian cohort and clinical trial results, as well as other real-life studies in Denmark, France, the Netherlands, and Spain, she said.
Patients, about one-third female, had a mean body mass index of about 24 kg/m2. Mean age was about 38 years (range, 19-80 years). The mean age of disease onset was about 14 years (range, 0-77 years).
Atopic dermatitis was characterized by phenotype for each patient; groupings included classic adult type (73%), nummular dermatitis (7%), prurigo (8%), and erythrodermic dermatitis (12%). About three in four patients (76.1%) had facial involvement; 61.5% had hand involvement, and 22.9% had genital involvement.
Allergic comorbidities were reported by many patients; 44.9% had rhinitis, 38.5% had asthma, 33% had conjunctivitis, and 15.6% reported food allergies. Other notable comorbidities included psychiatric or psychological conditions, present in 11% of patients, and hypertension or other cardiovascular disorders, seen in 9.1% of patients.
Most patients had tried treatment with both cyclosporine A and corticosteroids (88.9% and 88.1%, respectively). Almost half (45.8%) had tried UV-light therapy, and about a quarter had tried methotrexate.
“The results give real-life data on patterns of treatment response according to heterogeneous atopic dermatitis phenotypes, and on long-term efficacy and safety,” said Dr. Fargnoli.
The study was not funded by any company, according to Dr. Fargnoli. She has served on the advisory board for and has received honoraria for lectures and research grants from Sanofi-Genzyme.
REPORTING FROM WCD2019
Opioids: Overprescribing, alternatives, and clinical guidance
Optimal management of pregnant women with opioid misuse
Optimal management of postpartum and postoperative pain
Responders to r-TMS may engage in more physical activity after treatment
Responders to repeated transcranial magnetic stimulation for treatment of depression are more likely to engage in light physical activity, compared with those who do not respond to treatment, recent research shows.
“It is remarkable that there is so little evidence on whether treatments for depression among adults have an impact on physical activity and whether changes in physical activity mediate the outcomes of these treatments,” Matthew James Fagan, a PhD student at the University of British Columbia, Vancouver, and colleagues wrote. “Further research is required in understanding the covariation of [physical activity] with depression treatment response.”
The researchers performed a secondary analysis of 30 individuals with major depressive disorder (MDD) who underwent either repeated transcranial magnetic stimulation or intermittent theta burst stimulation for 4-6 weeks. The participants’ 17-item Hamilton Rating Scale for Depression was measured along with their level of physical activity before and after treatment. Physical activity was classified as either light physical activity (LPA) – defined as any waking activity between 1.5 and 3.0 metabolic equivalents – or moderate to vigorous physical activity (MVPA), which was defined as waking behavior at 3.0 metabolic equivalents or higher.
A total of 16 participants responded to treatment (greater than or equal to 18 on the Hamilton Rating Scale for Depression) and 14 participants were deemed nonresponders. The researchers found no significant differences in LPA or MVPA between groups at baseline, but a significant treatment effect was seen among responders who increased LPA by 55 min/day, compared with nonresponders (P = .009). There was also a nonsignificant treatment effect that increased MVPA favoring responders, according to an analysis of covariance.
“Simply, our findings indicate that patients moved more after r-TMS treatment, and this may reinforce the treatment effect,” Mr. Fagan and colleagues reported.
“Future work should systematically examine the role of PA before, during, and after depression treatments as important synergistic mechanisms may be at play in the treatment of MDD,” they wrote.
Mr. Fagan reported no relevant financial disclosures. One or more authors reported support from several entities, including Brainsway, the Canadian Institutes of Health Research, the National Institutes of Health, and the Vancouver Coastal Health Research Institute, and reported relationships with ANT Neuro, BrainCheck, Brainsway, Lundbeck, Restorative Brain Clinics, and TMS Neuro Solutions.
SOURCE: Fagan MJ et al. Ment Health Phys Act. 2019 Apr 24. doi: 10.1016/j.mhpa.2019.03.003.
Responders to repeated transcranial magnetic stimulation for treatment of depression are more likely to engage in light physical activity, compared with those who do not respond to treatment, recent research shows.
“It is remarkable that there is so little evidence on whether treatments for depression among adults have an impact on physical activity and whether changes in physical activity mediate the outcomes of these treatments,” Matthew James Fagan, a PhD student at the University of British Columbia, Vancouver, and colleagues wrote. “Further research is required in understanding the covariation of [physical activity] with depression treatment response.”
The researchers performed a secondary analysis of 30 individuals with major depressive disorder (MDD) who underwent either repeated transcranial magnetic stimulation or intermittent theta burst stimulation for 4-6 weeks. The participants’ 17-item Hamilton Rating Scale for Depression was measured along with their level of physical activity before and after treatment. Physical activity was classified as either light physical activity (LPA) – defined as any waking activity between 1.5 and 3.0 metabolic equivalents – or moderate to vigorous physical activity (MVPA), which was defined as waking behavior at 3.0 metabolic equivalents or higher.
A total of 16 participants responded to treatment (greater than or equal to 18 on the Hamilton Rating Scale for Depression) and 14 participants were deemed nonresponders. The researchers found no significant differences in LPA or MVPA between groups at baseline, but a significant treatment effect was seen among responders who increased LPA by 55 min/day, compared with nonresponders (P = .009). There was also a nonsignificant treatment effect that increased MVPA favoring responders, according to an analysis of covariance.
“Simply, our findings indicate that patients moved more after r-TMS treatment, and this may reinforce the treatment effect,” Mr. Fagan and colleagues reported.
“Future work should systematically examine the role of PA before, during, and after depression treatments as important synergistic mechanisms may be at play in the treatment of MDD,” they wrote.
Mr. Fagan reported no relevant financial disclosures. One or more authors reported support from several entities, including Brainsway, the Canadian Institutes of Health Research, the National Institutes of Health, and the Vancouver Coastal Health Research Institute, and reported relationships with ANT Neuro, BrainCheck, Brainsway, Lundbeck, Restorative Brain Clinics, and TMS Neuro Solutions.
SOURCE: Fagan MJ et al. Ment Health Phys Act. 2019 Apr 24. doi: 10.1016/j.mhpa.2019.03.003.
Responders to repeated transcranial magnetic stimulation for treatment of depression are more likely to engage in light physical activity, compared with those who do not respond to treatment, recent research shows.
“It is remarkable that there is so little evidence on whether treatments for depression among adults have an impact on physical activity and whether changes in physical activity mediate the outcomes of these treatments,” Matthew James Fagan, a PhD student at the University of British Columbia, Vancouver, and colleagues wrote. “Further research is required in understanding the covariation of [physical activity] with depression treatment response.”
The researchers performed a secondary analysis of 30 individuals with major depressive disorder (MDD) who underwent either repeated transcranial magnetic stimulation or intermittent theta burst stimulation for 4-6 weeks. The participants’ 17-item Hamilton Rating Scale for Depression was measured along with their level of physical activity before and after treatment. Physical activity was classified as either light physical activity (LPA) – defined as any waking activity between 1.5 and 3.0 metabolic equivalents – or moderate to vigorous physical activity (MVPA), which was defined as waking behavior at 3.0 metabolic equivalents or higher.
A total of 16 participants responded to treatment (greater than or equal to 18 on the Hamilton Rating Scale for Depression) and 14 participants were deemed nonresponders. The researchers found no significant differences in LPA or MVPA between groups at baseline, but a significant treatment effect was seen among responders who increased LPA by 55 min/day, compared with nonresponders (P = .009). There was also a nonsignificant treatment effect that increased MVPA favoring responders, according to an analysis of covariance.
“Simply, our findings indicate that patients moved more after r-TMS treatment, and this may reinforce the treatment effect,” Mr. Fagan and colleagues reported.
“Future work should systematically examine the role of PA before, during, and after depression treatments as important synergistic mechanisms may be at play in the treatment of MDD,” they wrote.
Mr. Fagan reported no relevant financial disclosures. One or more authors reported support from several entities, including Brainsway, the Canadian Institutes of Health Research, the National Institutes of Health, and the Vancouver Coastal Health Research Institute, and reported relationships with ANT Neuro, BrainCheck, Brainsway, Lundbeck, Restorative Brain Clinics, and TMS Neuro Solutions.
SOURCE: Fagan MJ et al. Ment Health Phys Act. 2019 Apr 24. doi: 10.1016/j.mhpa.2019.03.003.
FROM MENTAL HEALTH AND PHYSICAL ACTIVITY


