User login
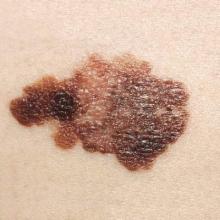
Dual immune checkpoint blockade found durable in melanoma
CHICAGO – Immune checkpoint blockade, especially with a combination of agents having complementary mechanisms of action, has durable efficacy when used as initial therapy for advanced melanoma, according to an update of the CheckMate 067 trial.
The trial randomized 945 treatment-naive patients with unresectable stage III or IV melanoma evenly to double-blind treatment with nivolumab, an antibody to the cell surface receptor programmed death 1 (PD-1); ipilimumab, an antibody to the T-cell receptor cytotoxic T-lymphocyte–associated antigen 4 (CTLA4); or the combination.
Initial results, after a median follow-up of about 12.4 months, showed that the risk of progression-free survival events was 58% lower with the combination and 43% lower with nivolumab alone as compared with ipilimumab alone (N Engl J Med. 2015;373[1]:23-34).
The update, now with a median follow-up of 20.7 months, showed that these results held up, with respective 58% and 45% reductions in the risk of events, researchers reported at the annual meeting of the American Society of Clinical Oncology. The combination was also superior to nivolumab alone, netting a 24% lower risk of events. Additionally, no cumulative or new toxicities were seen.
“Based on available evidence, the combination of nivolumab and ipilimumab represents a means to improve outcomes versus nivolumab alone,” said first author Jedd D. Wolchok, MD, PhD, chief of the Melanoma & Immunotherapeutics Service at the Memorial Sloan Kettering Cancer Center in New York. “Additional insights will be gained with the emergence of overall survival data.”
Neither tumor expression of PD-L1, a ligand of PD-1, nor presence of a BRAF mutation was very helpful in identifying patients who would benefit to a greater extent from these therapies.
The findings add to evidence establishing the efficacy of combination immunotherapy in melanoma, according to invited discussant Marc S. Ernstoff, MD, professor and chair of the department of medicine at the Roswell Park Cancer Institute in Buffalo, N.Y. At the same time, the trial left unanswered questions such as what strategy should be used after progression on either or both agents, and what are the appropriate doses and durations of therapy. Also unclear is which type of therapy to use first line in patients with BRAF mutations, he added. “Whether you start with immunotherapy or targeted therapy in BRAF-mutated patients is still in equipoise, and I would encourage everyone here to participate in the ECOG 6134 trial looking at the randomization of immune checkpoint therapy versus targeted therapy in BRAF-mutated patients,” he said. “The biomarker studies are still provocative, and we still need a lot more data to be able to preselect patients who might benefit from either of these therapies.”
“One has to recognize that these agents are costly,” Dr. Ernstoff maintained, with the acquisition cost of the checkpoint inhibitors ranging from roughly $140,000 to $290,000 per year depending on the agent(s) used. This issue will also have to be addressed going forward.
“The future is very bright. There are now 76 trials listed in PDQ [Physician Data Query] of combination PD-1 therapies in melanoma alone,” he concluded. “Immunotherapy continues to capture our imagination.”
The updated intent-to-treat analyses of CheckMate 067 – conducted after all patients had at least 18 months of follow-up – showed that median progression-free survival, one of the trial’s primary endpoints, was now 11.5 months with the combination of nivolumab (Opdivo) and ipilimumab (Yervoy), 6.9 months with nivolumab alone, and 2.9 months with ipilimumab alone, Dr. Wolchok reported at the meeting.
The differences translated to significantly better outcomes with the combination (hazard ratio, 0.42) and with nivolumab (HR, 0.55) as compared with ipilimumab. Moreover, the combination was superior to nivolumab (HR, 0.76).
The overall response rate, the trial’s other primary endpoint, was 57.6% with the combination and 43.7% with nivolumab alone, both of which were superior to the 19.0% with ipilimumab alone.
“While the response rates have not changed, some partial responses have evolved into complete responses over time,” Dr. Wolchok noted.
Findings were similar when patients were stratified by BRAF mutational status. And in exploratory analyses, outcomes were numerically better with the combination than with nivolumab alone regardless of whether tumors had high or low PD-L1 expression.
Safety results were much the same as previously reported. The rate of grade 3 or 4 treatment-related adverse events was 56.5% with the combination, 19.8% with nivolumab monotherapy, and 27.0% with ipilimumab monotherapy. There were no treatment-related deaths with the combination and one with each of the monotherapies.
“There is no common signature adverse event with this combination,” Dr. Wolchok pointed out. “The majority of grade 3 or 4 adverse events resolved in all of the groups with the use of established algorithms. However, as observed in prior studies, most of the endocrine events did not resolve and required hormone replacement.”
About 40% of the combination therapy group stopped treatment because of adverse events. “Interestingly, 68% of these patients who discontinued due to treatment-related adverse events developed a response, and 50% of these responses occurred after treatment had ended,” he reported. “This is very important information for us as we talk to patients and their families about the difficulties of stopping treatment.”
Dr. Wolchok disclosed that he is a consultant for Bristol-Myers Squibb, Genentech, Jounce Therapeutics, Medimmune, Merck, Polaris, Polynoma, Potenza, Tizona, Ziopharm, F-Star, Beigene, Lilly, Advaxis, and Sellas, and that he receives grant/research support from Bristol-Myers Squibb. The trial was sponsored by Bristol-Myers Squibb. Dako collaborated on development of the automated anti–PD-L1 immunohistochemistry assay.
CHICAGO – Immune checkpoint blockade, especially with a combination of agents having complementary mechanisms of action, has durable efficacy when used as initial therapy for advanced melanoma, according to an update of the CheckMate 067 trial.
The trial randomized 945 treatment-naive patients with unresectable stage III or IV melanoma evenly to double-blind treatment with nivolumab, an antibody to the cell surface receptor programmed death 1 (PD-1); ipilimumab, an antibody to the T-cell receptor cytotoxic T-lymphocyte–associated antigen 4 (CTLA4); or the combination.
Initial results, after a median follow-up of about 12.4 months, showed that the risk of progression-free survival events was 58% lower with the combination and 43% lower with nivolumab alone as compared with ipilimumab alone (N Engl J Med. 2015;373[1]:23-34).
The update, now with a median follow-up of 20.7 months, showed that these results held up, with respective 58% and 45% reductions in the risk of events, researchers reported at the annual meeting of the American Society of Clinical Oncology. The combination was also superior to nivolumab alone, netting a 24% lower risk of events. Additionally, no cumulative or new toxicities were seen.
“Based on available evidence, the combination of nivolumab and ipilimumab represents a means to improve outcomes versus nivolumab alone,” said first author Jedd D. Wolchok, MD, PhD, chief of the Melanoma & Immunotherapeutics Service at the Memorial Sloan Kettering Cancer Center in New York. “Additional insights will be gained with the emergence of overall survival data.”
Neither tumor expression of PD-L1, a ligand of PD-1, nor presence of a BRAF mutation was very helpful in identifying patients who would benefit to a greater extent from these therapies.
The findings add to evidence establishing the efficacy of combination immunotherapy in melanoma, according to invited discussant Marc S. Ernstoff, MD, professor and chair of the department of medicine at the Roswell Park Cancer Institute in Buffalo, N.Y. At the same time, the trial left unanswered questions such as what strategy should be used after progression on either or both agents, and what are the appropriate doses and durations of therapy. Also unclear is which type of therapy to use first line in patients with BRAF mutations, he added. “Whether you start with immunotherapy or targeted therapy in BRAF-mutated patients is still in equipoise, and I would encourage everyone here to participate in the ECOG 6134 trial looking at the randomization of immune checkpoint therapy versus targeted therapy in BRAF-mutated patients,” he said. “The biomarker studies are still provocative, and we still need a lot more data to be able to preselect patients who might benefit from either of these therapies.”
“One has to recognize that these agents are costly,” Dr. Ernstoff maintained, with the acquisition cost of the checkpoint inhibitors ranging from roughly $140,000 to $290,000 per year depending on the agent(s) used. This issue will also have to be addressed going forward.
“The future is very bright. There are now 76 trials listed in PDQ [Physician Data Query] of combination PD-1 therapies in melanoma alone,” he concluded. “Immunotherapy continues to capture our imagination.”
The updated intent-to-treat analyses of CheckMate 067 – conducted after all patients had at least 18 months of follow-up – showed that median progression-free survival, one of the trial’s primary endpoints, was now 11.5 months with the combination of nivolumab (Opdivo) and ipilimumab (Yervoy), 6.9 months with nivolumab alone, and 2.9 months with ipilimumab alone, Dr. Wolchok reported at the meeting.
The differences translated to significantly better outcomes with the combination (hazard ratio, 0.42) and with nivolumab (HR, 0.55) as compared with ipilimumab. Moreover, the combination was superior to nivolumab (HR, 0.76).
The overall response rate, the trial’s other primary endpoint, was 57.6% with the combination and 43.7% with nivolumab alone, both of which were superior to the 19.0% with ipilimumab alone.
“While the response rates have not changed, some partial responses have evolved into complete responses over time,” Dr. Wolchok noted.
Findings were similar when patients were stratified by BRAF mutational status. And in exploratory analyses, outcomes were numerically better with the combination than with nivolumab alone regardless of whether tumors had high or low PD-L1 expression.
Safety results were much the same as previously reported. The rate of grade 3 or 4 treatment-related adverse events was 56.5% with the combination, 19.8% with nivolumab monotherapy, and 27.0% with ipilimumab monotherapy. There were no treatment-related deaths with the combination and one with each of the monotherapies.
“There is no common signature adverse event with this combination,” Dr. Wolchok pointed out. “The majority of grade 3 or 4 adverse events resolved in all of the groups with the use of established algorithms. However, as observed in prior studies, most of the endocrine events did not resolve and required hormone replacement.”
About 40% of the combination therapy group stopped treatment because of adverse events. “Interestingly, 68% of these patients who discontinued due to treatment-related adverse events developed a response, and 50% of these responses occurred after treatment had ended,” he reported. “This is very important information for us as we talk to patients and their families about the difficulties of stopping treatment.”
Dr. Wolchok disclosed that he is a consultant for Bristol-Myers Squibb, Genentech, Jounce Therapeutics, Medimmune, Merck, Polaris, Polynoma, Potenza, Tizona, Ziopharm, F-Star, Beigene, Lilly, Advaxis, and Sellas, and that he receives grant/research support from Bristol-Myers Squibb. The trial was sponsored by Bristol-Myers Squibb. Dako collaborated on development of the automated anti–PD-L1 immunohistochemistry assay.
CHICAGO – Immune checkpoint blockade, especially with a combination of agents having complementary mechanisms of action, has durable efficacy when used as initial therapy for advanced melanoma, according to an update of the CheckMate 067 trial.
The trial randomized 945 treatment-naive patients with unresectable stage III or IV melanoma evenly to double-blind treatment with nivolumab, an antibody to the cell surface receptor programmed death 1 (PD-1); ipilimumab, an antibody to the T-cell receptor cytotoxic T-lymphocyte–associated antigen 4 (CTLA4); or the combination.
Initial results, after a median follow-up of about 12.4 months, showed that the risk of progression-free survival events was 58% lower with the combination and 43% lower with nivolumab alone as compared with ipilimumab alone (N Engl J Med. 2015;373[1]:23-34).
The update, now with a median follow-up of 20.7 months, showed that these results held up, with respective 58% and 45% reductions in the risk of events, researchers reported at the annual meeting of the American Society of Clinical Oncology. The combination was also superior to nivolumab alone, netting a 24% lower risk of events. Additionally, no cumulative or new toxicities were seen.
“Based on available evidence, the combination of nivolumab and ipilimumab represents a means to improve outcomes versus nivolumab alone,” said first author Jedd D. Wolchok, MD, PhD, chief of the Melanoma & Immunotherapeutics Service at the Memorial Sloan Kettering Cancer Center in New York. “Additional insights will be gained with the emergence of overall survival data.”
Neither tumor expression of PD-L1, a ligand of PD-1, nor presence of a BRAF mutation was very helpful in identifying patients who would benefit to a greater extent from these therapies.
The findings add to evidence establishing the efficacy of combination immunotherapy in melanoma, according to invited discussant Marc S. Ernstoff, MD, professor and chair of the department of medicine at the Roswell Park Cancer Institute in Buffalo, N.Y. At the same time, the trial left unanswered questions such as what strategy should be used after progression on either or both agents, and what are the appropriate doses and durations of therapy. Also unclear is which type of therapy to use first line in patients with BRAF mutations, he added. “Whether you start with immunotherapy or targeted therapy in BRAF-mutated patients is still in equipoise, and I would encourage everyone here to participate in the ECOG 6134 trial looking at the randomization of immune checkpoint therapy versus targeted therapy in BRAF-mutated patients,” he said. “The biomarker studies are still provocative, and we still need a lot more data to be able to preselect patients who might benefit from either of these therapies.”
“One has to recognize that these agents are costly,” Dr. Ernstoff maintained, with the acquisition cost of the checkpoint inhibitors ranging from roughly $140,000 to $290,000 per year depending on the agent(s) used. This issue will also have to be addressed going forward.
“The future is very bright. There are now 76 trials listed in PDQ [Physician Data Query] of combination PD-1 therapies in melanoma alone,” he concluded. “Immunotherapy continues to capture our imagination.”
The updated intent-to-treat analyses of CheckMate 067 – conducted after all patients had at least 18 months of follow-up – showed that median progression-free survival, one of the trial’s primary endpoints, was now 11.5 months with the combination of nivolumab (Opdivo) and ipilimumab (Yervoy), 6.9 months with nivolumab alone, and 2.9 months with ipilimumab alone, Dr. Wolchok reported at the meeting.
The differences translated to significantly better outcomes with the combination (hazard ratio, 0.42) and with nivolumab (HR, 0.55) as compared with ipilimumab. Moreover, the combination was superior to nivolumab (HR, 0.76).
The overall response rate, the trial’s other primary endpoint, was 57.6% with the combination and 43.7% with nivolumab alone, both of which were superior to the 19.0% with ipilimumab alone.
“While the response rates have not changed, some partial responses have evolved into complete responses over time,” Dr. Wolchok noted.
Findings were similar when patients were stratified by BRAF mutational status. And in exploratory analyses, outcomes were numerically better with the combination than with nivolumab alone regardless of whether tumors had high or low PD-L1 expression.
Safety results were much the same as previously reported. The rate of grade 3 or 4 treatment-related adverse events was 56.5% with the combination, 19.8% with nivolumab monotherapy, and 27.0% with ipilimumab monotherapy. There were no treatment-related deaths with the combination and one with each of the monotherapies.
“There is no common signature adverse event with this combination,” Dr. Wolchok pointed out. “The majority of grade 3 or 4 adverse events resolved in all of the groups with the use of established algorithms. However, as observed in prior studies, most of the endocrine events did not resolve and required hormone replacement.”
About 40% of the combination therapy group stopped treatment because of adverse events. “Interestingly, 68% of these patients who discontinued due to treatment-related adverse events developed a response, and 50% of these responses occurred after treatment had ended,” he reported. “This is very important information for us as we talk to patients and their families about the difficulties of stopping treatment.”
Dr. Wolchok disclosed that he is a consultant for Bristol-Myers Squibb, Genentech, Jounce Therapeutics, Medimmune, Merck, Polaris, Polynoma, Potenza, Tizona, Ziopharm, F-Star, Beigene, Lilly, Advaxis, and Sellas, and that he receives grant/research support from Bristol-Myers Squibb. The trial was sponsored by Bristol-Myers Squibb. Dako collaborated on development of the automated anti–PD-L1 immunohistochemistry assay.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Nivolumab-ipilimumab combination therapy and nivolumab monotherapy are more efficacious than ipilimumab monotherapy when used in the first line for advanced melanoma.
Major finding: The risk of progression-free survival events was lower with nivolumab plus ipilimumab (HR, 0.42) and with nivolumab alone (HR, 0.55) as compared with ipilimumab alone.
Data source: A phase III randomized trial among 945 treatment-naive patients with advanced melanoma (CheckMate 067).
Disclosures: Dr. Wolchok disclosed that he is a consultant for Bristol-Myers Squibb, Genentech, Jounce Therapeutics, Medimmune, Merck, Polaris, Polynoma, Potenza, Tizona, Ziopharm, F-Star, Beigene, Lilly, Advaxis, and Sellas, and that he receives grant/research support from Bristol-Myers Squibb. The trial was sponsored by Bristol-Myers Squibb. Dako collaborated on development of the automated anti-PD-L1 immunohistochemistry assay.
Including quality-of-life scores may aid decision making for patients with advanced ovarian cancer
CHICAGO – Physical function, role function, global health status and abdominal/gastrointestinal symptoms (AGIS) each predicted overall survival and were significantly associated with the early cessation of chemotherapy among women with platinum-resistant/refractory recurrent ovarian cancer in the Gynecologic Cancer InterGroup (GCIG) Symptom Benefit Study.
The findings from the international prospective cohort study suggest that baseline assessment of quality of life could help identify patients with platinum-resistant/refractory recurrent ovarian cancer (PRR-ROC) who are unlikely to benefit from palliative chemotherapy, Dr. Felicia Roncolato reported at the annual meeting of the American Society of Clinical Oncology.
In 570 women with PRR-ROC enrolled in the Symptom Benefit Study, median overall survival was 11.1 months and median progression-free survival was 3.6 months.
Factors shown on multivariable analysis to predict overall survival included hemoglobin (hazard ratio, 0.94 per 10 g/L increase), ascites (HR, 1.60), AGIS (HR, 1.24), platelets (HR, 1.10 per 100 x 109 unit increase), Log CA125 (HR, 1.18 per unit increase), and neutrophil:lymphocyte ratio (HR, 1.79 for 5 or more). These were all statistically significant predictors of overall survival, said Dr. Roncolato of St. George Hospital, Sydney.
As for baseline quality of life data as a predictor of overall survival, the hazard ratios were 1.60 for low physical function, 1.54 for low role function, 1.55 for global health status, 2.37 for worst vs. least AGIS, and 1.75 for intermediate vs. least AGIS. After adjusting for all of these clinical factors, the multivariable analysis showed that low physical function, role function, and global health status, and worst AGIS remained statistically significant predictors of overall survival (HR, 1,45, 1.37, 1.34, 1.49, and 1.49, respectively). Median overall survival was 7 vs. 12 months in those with lower vs. higher physical function, role function, and global health status, 9 months vs. 14 months for those with lower vs. higher role function scores, and 8, 11, and 18 months in those with worst, intermediate, and least AGIS.
A sensitivity analysis supported the validity of the cut-points used for each of these scores, Dr. Roncolato noted.
As for early cessation of chemotherapy, 110 of the 570 women (19%) stopped chemotherapy within 8 weeks. Most (46%) stopped due to disease progression; other reasons for early cessation included death (18%), patient preference (12%), “other” (12%), adverse event (7%), and clinician preference (6%).
In these women, median progression-free survival and median overall survival were 1.3 months and 2.9 months, respectively, Dr. Roncolato said.
On univariable analysis, the same four quality of life domains (physical function, role function, global health status, and AGIS) each were significantly associated with overall survival (odds ratios were 2.45 for low physical function, 2.71 for low role function, 2.38 for global health status, 2.31 for worst vs. least AGIS, and 1.17 for intermediate vs. least AGIS).
Most patients with ovarian cancer have advanced stage disease at diagnosis and develop recurrent disease despite initial response, and most ultimately develop platinum resistant/refractory disease, Dr. Roncolato said.
The goals of treatment are to improve length and quality of life, but response rates are low; median progression-free survival is 3 months, and median overall survival is less than 12 months, she noted.
“To date there is no evidence that chemotherapy actually increases overall survival in the resistant/refractory setting, and one of our biggest challenges is identifying the patients who are most and least likely to benefit,” she said, adding that over the last decade, little has changed in terms of chemotherapy outcomes remaining poor in patients with PRR-ROC (median overall survival of about 45% at 12 months).
A substantial number of patients stop treatment early.
The Symptom Benefit Study was designed based on a recommendation of the 3rd GCIG Ovarian Cancer Consensus meeting, which called for more robust and reliable methods to quantify symptom improvement in patients with platinum-resistant/refractory ovarian cancer. The primary aim of the study was to develop criteria for quantifying symptom benefit for clinical trials in such patients. The initial portion of the study was known as MOST (Measure of Ovarian Cancer Symptoms and Treatment Concerns). The aim of the current portion of the study was to identify baseline characteristics associated with early cessation of chemotherapy and with poor overall survival.
Patients included in the study were women with PRR-ROC and patients receiving a third or subsequent line of treatment. All had a life expectancy of more than 3 months, and had an Eastern Cooperative Oncology Group (ECOG) performance status score of 0-3.
Quality of life measures, including EORTC QLQ-C30, QLQ-OV28, and others were performed at baseline and before each cycle of chemotherapy.
“The health-related quality of life scores identified a subset of women with resistant/refractory disease who have a very poor prognosis. It’s more informative than a clinician-assigned ECOG performance status, and including baseline health-related quality of life together with clinical prognostic factors improved the prediction of survival in women with PRR-ROC,” Dr. Roncolato said, adding that having this additional prognostic information could improve stratification in clinical trials, patient-doctor communication about prognosis, and clinical decision-making.
This study was funded by the Australian National Health and Medical Research Council. Dr. Roncolato reported having no disclosures.
CHICAGO – Physical function, role function, global health status and abdominal/gastrointestinal symptoms (AGIS) each predicted overall survival and were significantly associated with the early cessation of chemotherapy among women with platinum-resistant/refractory recurrent ovarian cancer in the Gynecologic Cancer InterGroup (GCIG) Symptom Benefit Study.
The findings from the international prospective cohort study suggest that baseline assessment of quality of life could help identify patients with platinum-resistant/refractory recurrent ovarian cancer (PRR-ROC) who are unlikely to benefit from palliative chemotherapy, Dr. Felicia Roncolato reported at the annual meeting of the American Society of Clinical Oncology.
In 570 women with PRR-ROC enrolled in the Symptom Benefit Study, median overall survival was 11.1 months and median progression-free survival was 3.6 months.
Factors shown on multivariable analysis to predict overall survival included hemoglobin (hazard ratio, 0.94 per 10 g/L increase), ascites (HR, 1.60), AGIS (HR, 1.24), platelets (HR, 1.10 per 100 x 109 unit increase), Log CA125 (HR, 1.18 per unit increase), and neutrophil:lymphocyte ratio (HR, 1.79 for 5 or more). These were all statistically significant predictors of overall survival, said Dr. Roncolato of St. George Hospital, Sydney.
As for baseline quality of life data as a predictor of overall survival, the hazard ratios were 1.60 for low physical function, 1.54 for low role function, 1.55 for global health status, 2.37 for worst vs. least AGIS, and 1.75 for intermediate vs. least AGIS. After adjusting for all of these clinical factors, the multivariable analysis showed that low physical function, role function, and global health status, and worst AGIS remained statistically significant predictors of overall survival (HR, 1,45, 1.37, 1.34, 1.49, and 1.49, respectively). Median overall survival was 7 vs. 12 months in those with lower vs. higher physical function, role function, and global health status, 9 months vs. 14 months for those with lower vs. higher role function scores, and 8, 11, and 18 months in those with worst, intermediate, and least AGIS.
A sensitivity analysis supported the validity of the cut-points used for each of these scores, Dr. Roncolato noted.
As for early cessation of chemotherapy, 110 of the 570 women (19%) stopped chemotherapy within 8 weeks. Most (46%) stopped due to disease progression; other reasons for early cessation included death (18%), patient preference (12%), “other” (12%), adverse event (7%), and clinician preference (6%).
In these women, median progression-free survival and median overall survival were 1.3 months and 2.9 months, respectively, Dr. Roncolato said.
On univariable analysis, the same four quality of life domains (physical function, role function, global health status, and AGIS) each were significantly associated with overall survival (odds ratios were 2.45 for low physical function, 2.71 for low role function, 2.38 for global health status, 2.31 for worst vs. least AGIS, and 1.17 for intermediate vs. least AGIS).
Most patients with ovarian cancer have advanced stage disease at diagnosis and develop recurrent disease despite initial response, and most ultimately develop platinum resistant/refractory disease, Dr. Roncolato said.
The goals of treatment are to improve length and quality of life, but response rates are low; median progression-free survival is 3 months, and median overall survival is less than 12 months, she noted.
“To date there is no evidence that chemotherapy actually increases overall survival in the resistant/refractory setting, and one of our biggest challenges is identifying the patients who are most and least likely to benefit,” she said, adding that over the last decade, little has changed in terms of chemotherapy outcomes remaining poor in patients with PRR-ROC (median overall survival of about 45% at 12 months).
A substantial number of patients stop treatment early.
The Symptom Benefit Study was designed based on a recommendation of the 3rd GCIG Ovarian Cancer Consensus meeting, which called for more robust and reliable methods to quantify symptom improvement in patients with platinum-resistant/refractory ovarian cancer. The primary aim of the study was to develop criteria for quantifying symptom benefit for clinical trials in such patients. The initial portion of the study was known as MOST (Measure of Ovarian Cancer Symptoms and Treatment Concerns). The aim of the current portion of the study was to identify baseline characteristics associated with early cessation of chemotherapy and with poor overall survival.
Patients included in the study were women with PRR-ROC and patients receiving a third or subsequent line of treatment. All had a life expectancy of more than 3 months, and had an Eastern Cooperative Oncology Group (ECOG) performance status score of 0-3.
Quality of life measures, including EORTC QLQ-C30, QLQ-OV28, and others were performed at baseline and before each cycle of chemotherapy.
“The health-related quality of life scores identified a subset of women with resistant/refractory disease who have a very poor prognosis. It’s more informative than a clinician-assigned ECOG performance status, and including baseline health-related quality of life together with clinical prognostic factors improved the prediction of survival in women with PRR-ROC,” Dr. Roncolato said, adding that having this additional prognostic information could improve stratification in clinical trials, patient-doctor communication about prognosis, and clinical decision-making.
This study was funded by the Australian National Health and Medical Research Council. Dr. Roncolato reported having no disclosures.
CHICAGO – Physical function, role function, global health status and abdominal/gastrointestinal symptoms (AGIS) each predicted overall survival and were significantly associated with the early cessation of chemotherapy among women with platinum-resistant/refractory recurrent ovarian cancer in the Gynecologic Cancer InterGroup (GCIG) Symptom Benefit Study.
The findings from the international prospective cohort study suggest that baseline assessment of quality of life could help identify patients with platinum-resistant/refractory recurrent ovarian cancer (PRR-ROC) who are unlikely to benefit from palliative chemotherapy, Dr. Felicia Roncolato reported at the annual meeting of the American Society of Clinical Oncology.
In 570 women with PRR-ROC enrolled in the Symptom Benefit Study, median overall survival was 11.1 months and median progression-free survival was 3.6 months.
Factors shown on multivariable analysis to predict overall survival included hemoglobin (hazard ratio, 0.94 per 10 g/L increase), ascites (HR, 1.60), AGIS (HR, 1.24), platelets (HR, 1.10 per 100 x 109 unit increase), Log CA125 (HR, 1.18 per unit increase), and neutrophil:lymphocyte ratio (HR, 1.79 for 5 or more). These were all statistically significant predictors of overall survival, said Dr. Roncolato of St. George Hospital, Sydney.
As for baseline quality of life data as a predictor of overall survival, the hazard ratios were 1.60 for low physical function, 1.54 for low role function, 1.55 for global health status, 2.37 for worst vs. least AGIS, and 1.75 for intermediate vs. least AGIS. After adjusting for all of these clinical factors, the multivariable analysis showed that low physical function, role function, and global health status, and worst AGIS remained statistically significant predictors of overall survival (HR, 1,45, 1.37, 1.34, 1.49, and 1.49, respectively). Median overall survival was 7 vs. 12 months in those with lower vs. higher physical function, role function, and global health status, 9 months vs. 14 months for those with lower vs. higher role function scores, and 8, 11, and 18 months in those with worst, intermediate, and least AGIS.
A sensitivity analysis supported the validity of the cut-points used for each of these scores, Dr. Roncolato noted.
As for early cessation of chemotherapy, 110 of the 570 women (19%) stopped chemotherapy within 8 weeks. Most (46%) stopped due to disease progression; other reasons for early cessation included death (18%), patient preference (12%), “other” (12%), adverse event (7%), and clinician preference (6%).
In these women, median progression-free survival and median overall survival were 1.3 months and 2.9 months, respectively, Dr. Roncolato said.
On univariable analysis, the same four quality of life domains (physical function, role function, global health status, and AGIS) each were significantly associated with overall survival (odds ratios were 2.45 for low physical function, 2.71 for low role function, 2.38 for global health status, 2.31 for worst vs. least AGIS, and 1.17 for intermediate vs. least AGIS).
Most patients with ovarian cancer have advanced stage disease at diagnosis and develop recurrent disease despite initial response, and most ultimately develop platinum resistant/refractory disease, Dr. Roncolato said.
The goals of treatment are to improve length and quality of life, but response rates are low; median progression-free survival is 3 months, and median overall survival is less than 12 months, she noted.
“To date there is no evidence that chemotherapy actually increases overall survival in the resistant/refractory setting, and one of our biggest challenges is identifying the patients who are most and least likely to benefit,” she said, adding that over the last decade, little has changed in terms of chemotherapy outcomes remaining poor in patients with PRR-ROC (median overall survival of about 45% at 12 months).
A substantial number of patients stop treatment early.
The Symptom Benefit Study was designed based on a recommendation of the 3rd GCIG Ovarian Cancer Consensus meeting, which called for more robust and reliable methods to quantify symptom improvement in patients with platinum-resistant/refractory ovarian cancer. The primary aim of the study was to develop criteria for quantifying symptom benefit for clinical trials in such patients. The initial portion of the study was known as MOST (Measure of Ovarian Cancer Symptoms and Treatment Concerns). The aim of the current portion of the study was to identify baseline characteristics associated with early cessation of chemotherapy and with poor overall survival.
Patients included in the study were women with PRR-ROC and patients receiving a third or subsequent line of treatment. All had a life expectancy of more than 3 months, and had an Eastern Cooperative Oncology Group (ECOG) performance status score of 0-3.
Quality of life measures, including EORTC QLQ-C30, QLQ-OV28, and others were performed at baseline and before each cycle of chemotherapy.
“The health-related quality of life scores identified a subset of women with resistant/refractory disease who have a very poor prognosis. It’s more informative than a clinician-assigned ECOG performance status, and including baseline health-related quality of life together with clinical prognostic factors improved the prediction of survival in women with PRR-ROC,” Dr. Roncolato said, adding that having this additional prognostic information could improve stratification in clinical trials, patient-doctor communication about prognosis, and clinical decision-making.
This study was funded by the Australian National Health and Medical Research Council. Dr. Roncolato reported having no disclosures.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Physical function, role function, global health status, and abdominal/gastrointestinal symptoms (AGIS) appear to predict overall survival and early cessation of chemotherapy among women with platinum-resistant/refractory recurrent ovarian cancer.
Major finding: Multivariable analysis showed that low physical function, role function, and global health status, and worse AGIS were statistically significant predictors of overall survival (hazard ratios, 1,45, 1.37, 1.34, 1.49, and 1.49, respectively).
Data source: 570 patients from the international prospective GCIG Symptom Benefit Study.
Disclosures: This study was funded by the Australian National Health and Medical Research Council. Dr. Roncolato reported having no disclosures.
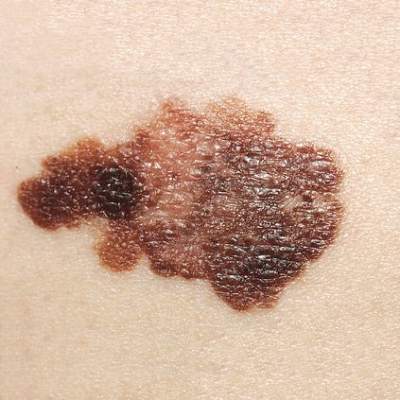
Checkpoint-blocking antibodies appear safe, beneficial in elderly melanoma patients
CHICAGO – Programmed cell death protein 1 (PD-1) inhibitors and combination ipilimumab and nivolumab prolonged survival, compared with ipilimumab alone in patients aged 80 and older with advanced melanoma, and the patients experienced no more adverse events than did patients of all age groups in prior published phase III studies, according to a retrospective case review.
Of 106 patients included in the current review, 74 received at least 1 dose of the anti–cytotoxic T-lymphocyte antigen 4 agent ipilimumab, 13 received at least 1 dose of an anti–PD-1 agent (pembrolizumab or nivolumab), and 8 received at least 1 dose of combination ipilimumab and nivolumab. Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively, Dr. Claire Frances Friedman reported at the annual meeting of the American Society of Clinical Oncology.
The differences between the groups did not reach statistical significance, said Dr. Friedman of Memorial Sloan Kettering Cancer Center, New York
Of note, a graphic representation of overall survival by treatment type shows a plateau after 24 months of treatment with ipilimumab (Ipi).
“About 20% of patients treated with Ipi have had a durable survival lasting many years,” she said, noting that this finding is similar to that based on pooled data from a previous study showing a survival rate of 22% with Ipi.
The findings are important because the highest incidence of advanced melanoma is among patients aged 80-84 years, and those over age 75 have a much higher mortality from melanoma, compared with younger patients, she said, adding that “the treatment of advanced melanoma has benefited from great strides in the area of immunotherapy, specifically the use of checkpoint-blocking antibodies.”
These antibodies work by blocking negative regulators of T-cell function, she explained.
The three used in this study are currently approved for the treatment of advanced melanoma. The combination of ipilimumab and nivolumab was approved when it was shown to improve progression-free survival vs. ipilimumab alone.
“Of note, responses to this class of drugs tend to be durable,” Dr. Friedman said.
In a prior study of nivolumab, the 5-year survival rate was 34%, she said.
However, the immune-related adverse events (irAEs) – “a unique set of side effects” associated with checkpoint-blocking antibodies – are concerning. The incidence of high-grade irAEs can vary with the type of checkpoint-blocking antibody – ranging from 16.3% with single-agent anti–PD-1 treatment to 55% with combination treatment in previous reports. The type of irAEs experienced tend to be similar across treatment types, and in a study published in 2015, were associated with a relatively high rate of treatment discontinuation (36.4% for combination treatment, 14.8% with ipilimumab, and 7.7% with nivolumab).
Given the increased mortality and morbidity in older patients, Dr. Friedman and her colleagues were interested in examining the toxicity and efficacy of checkpoint-blocking antibodies in patients aged 80 years and older. They performed the current review involving all patients aged 80 years and older at Memorial Sloan Kettering Cancer Center who were started on checkpoint blockade for stage 3 or 4 unresected melanoma between January 2008 and December 2015.
All patients had at least one infusion visit and one follow-up visit, and 12 of those in the Ipi group received subsequent anti–PD-1 therapy and were included in the toxicity analyses for both agents.
An irAE occurred in 88% of the 74 patients in the Ipi group, and 30% experienced a grade 3 or 4 irAE. The most common high-grade irAEs in this group were diarrhea, transaminitis, and rash.
Of the 25 patients receiving pembrolizumab or nivolumab, 84% experienced any irAE, and 16% experienced a grade 3 event. The most common adverse events in this group were pruritus, rash, fatigue, and musculoskeletal complaints, and one case each of grade 3 lipase, diarrhea, anemia, and nausea occurred.
Of the eight patients who received combination ipilimumab/nivolumab, 88% experienced any irAE and 63% experienced a high-grade 3 or 4 irAE. The most common high-grade irAEs in this group were elevated lipase, diarrhea, and transaminitis, occurring in 38%, 25%, and 25% of patients, respectively.
No deaths occurred in any of the groups, Dr. Friedman said.
The use of systemic steroids to manage irAEs was similar in the Ipi and anti–PD-1 monotherapy groups (28% in both groups), but 75% of the combination therapy patients were treated with systemic steroids. Infliximab was required for diarrhea in 7% of the Ipi patients and 38% of the combination therapy patients. None of the patients in any of the three groups required mycophenolate mofetil for steroid-refractory transaminitis, Dr. Friedman noted.
The findings suggest that when it comes to selecting the appropriate treatment for older patients, there are several things to consider, she said.
For example, in the elderly population, diarrhea and colitis are associated with an increased risk of dehydration and subsequent renal failure. Also, administering prednisone could lead to delirium or altered mental status, and can be of concern due to the increased incidence of comorbid conditions such as high blood pressure and diabetes, which may be worsened by steroid therapy.
Another concern is the fact that data from the rheumatology literature suggest that infliximab in the elderly may lead to increased risk of infections; a caveat is that this was in cases involving repeated doses, Dr. Friedman noted.
Treatment discontinuation was high across the groups, with 41%, 72%, and 63% of those in the Ipi, anti–PD-1, and combination therapy groups stopping treatment early. Toxicity was the reason for discontinuation in 31%, 20%, and 50%, respectively. Progression of disease or other unrelated issues were the reasons for discontinuation in the remaining patients.
“Patients aged 80 and older treated with ipilimumab had durable survival at a rate similar to that of the general population,” Dr. Friedman said.
Further, median overall survival is prolonged in older patients treated with anti–PD-1 drugs and with combination nivolumab/ipilimumab, but not significantly.
Importantly, irAEs occur more frequently with ipilimumab-based regimens in this population, she said, noting that discontinuation rates for toxicity are also higher in those aged 80 and older.
“So given these data, as clinicians we need to start thinking about individualizing treatment for our patients – using more than just the age. Physicians should consider incorporating assessments of functional status, such as the Mini-Cog or the Get Up and Go Test when deciding which of the checkpoint antibodies is appropriate for any individual patients,” she said, adding that as the population ages, more data in the elderly population will be needed.
“Clinicians should encourage older folks to participate in immuno-oncology clinical trials, and I hope that these data will encourage physicians to consider the use of checkpoint-blocking antibodies in the treatment of other malignancy types such as non–small cell lung cancer and bladder cancer, which also have a large proportion of adults over the age of 75,” she concluded.
Dr. Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
CHICAGO – Programmed cell death protein 1 (PD-1) inhibitors and combination ipilimumab and nivolumab prolonged survival, compared with ipilimumab alone in patients aged 80 and older with advanced melanoma, and the patients experienced no more adverse events than did patients of all age groups in prior published phase III studies, according to a retrospective case review.
Of 106 patients included in the current review, 74 received at least 1 dose of the anti–cytotoxic T-lymphocyte antigen 4 agent ipilimumab, 13 received at least 1 dose of an anti–PD-1 agent (pembrolizumab or nivolumab), and 8 received at least 1 dose of combination ipilimumab and nivolumab. Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively, Dr. Claire Frances Friedman reported at the annual meeting of the American Society of Clinical Oncology.
The differences between the groups did not reach statistical significance, said Dr. Friedman of Memorial Sloan Kettering Cancer Center, New York
Of note, a graphic representation of overall survival by treatment type shows a plateau after 24 months of treatment with ipilimumab (Ipi).
“About 20% of patients treated with Ipi have had a durable survival lasting many years,” she said, noting that this finding is similar to that based on pooled data from a previous study showing a survival rate of 22% with Ipi.
The findings are important because the highest incidence of advanced melanoma is among patients aged 80-84 years, and those over age 75 have a much higher mortality from melanoma, compared with younger patients, she said, adding that “the treatment of advanced melanoma has benefited from great strides in the area of immunotherapy, specifically the use of checkpoint-blocking antibodies.”
These antibodies work by blocking negative regulators of T-cell function, she explained.
The three used in this study are currently approved for the treatment of advanced melanoma. The combination of ipilimumab and nivolumab was approved when it was shown to improve progression-free survival vs. ipilimumab alone.
“Of note, responses to this class of drugs tend to be durable,” Dr. Friedman said.
In a prior study of nivolumab, the 5-year survival rate was 34%, she said.
However, the immune-related adverse events (irAEs) – “a unique set of side effects” associated with checkpoint-blocking antibodies – are concerning. The incidence of high-grade irAEs can vary with the type of checkpoint-blocking antibody – ranging from 16.3% with single-agent anti–PD-1 treatment to 55% with combination treatment in previous reports. The type of irAEs experienced tend to be similar across treatment types, and in a study published in 2015, were associated with a relatively high rate of treatment discontinuation (36.4% for combination treatment, 14.8% with ipilimumab, and 7.7% with nivolumab).
Given the increased mortality and morbidity in older patients, Dr. Friedman and her colleagues were interested in examining the toxicity and efficacy of checkpoint-blocking antibodies in patients aged 80 years and older. They performed the current review involving all patients aged 80 years and older at Memorial Sloan Kettering Cancer Center who were started on checkpoint blockade for stage 3 or 4 unresected melanoma between January 2008 and December 2015.
All patients had at least one infusion visit and one follow-up visit, and 12 of those in the Ipi group received subsequent anti–PD-1 therapy and were included in the toxicity analyses for both agents.
An irAE occurred in 88% of the 74 patients in the Ipi group, and 30% experienced a grade 3 or 4 irAE. The most common high-grade irAEs in this group were diarrhea, transaminitis, and rash.
Of the 25 patients receiving pembrolizumab or nivolumab, 84% experienced any irAE, and 16% experienced a grade 3 event. The most common adverse events in this group were pruritus, rash, fatigue, and musculoskeletal complaints, and one case each of grade 3 lipase, diarrhea, anemia, and nausea occurred.
Of the eight patients who received combination ipilimumab/nivolumab, 88% experienced any irAE and 63% experienced a high-grade 3 or 4 irAE. The most common high-grade irAEs in this group were elevated lipase, diarrhea, and transaminitis, occurring in 38%, 25%, and 25% of patients, respectively.
No deaths occurred in any of the groups, Dr. Friedman said.
The use of systemic steroids to manage irAEs was similar in the Ipi and anti–PD-1 monotherapy groups (28% in both groups), but 75% of the combination therapy patients were treated with systemic steroids. Infliximab was required for diarrhea in 7% of the Ipi patients and 38% of the combination therapy patients. None of the patients in any of the three groups required mycophenolate mofetil for steroid-refractory transaminitis, Dr. Friedman noted.
The findings suggest that when it comes to selecting the appropriate treatment for older patients, there are several things to consider, she said.
For example, in the elderly population, diarrhea and colitis are associated with an increased risk of dehydration and subsequent renal failure. Also, administering prednisone could lead to delirium or altered mental status, and can be of concern due to the increased incidence of comorbid conditions such as high blood pressure and diabetes, which may be worsened by steroid therapy.
Another concern is the fact that data from the rheumatology literature suggest that infliximab in the elderly may lead to increased risk of infections; a caveat is that this was in cases involving repeated doses, Dr. Friedman noted.
Treatment discontinuation was high across the groups, with 41%, 72%, and 63% of those in the Ipi, anti–PD-1, and combination therapy groups stopping treatment early. Toxicity was the reason for discontinuation in 31%, 20%, and 50%, respectively. Progression of disease or other unrelated issues were the reasons for discontinuation in the remaining patients.
“Patients aged 80 and older treated with ipilimumab had durable survival at a rate similar to that of the general population,” Dr. Friedman said.
Further, median overall survival is prolonged in older patients treated with anti–PD-1 drugs and with combination nivolumab/ipilimumab, but not significantly.
Importantly, irAEs occur more frequently with ipilimumab-based regimens in this population, she said, noting that discontinuation rates for toxicity are also higher in those aged 80 and older.
“So given these data, as clinicians we need to start thinking about individualizing treatment for our patients – using more than just the age. Physicians should consider incorporating assessments of functional status, such as the Mini-Cog or the Get Up and Go Test when deciding which of the checkpoint antibodies is appropriate for any individual patients,” she said, adding that as the population ages, more data in the elderly population will be needed.
“Clinicians should encourage older folks to participate in immuno-oncology clinical trials, and I hope that these data will encourage physicians to consider the use of checkpoint-blocking antibodies in the treatment of other malignancy types such as non–small cell lung cancer and bladder cancer, which also have a large proportion of adults over the age of 75,” she concluded.
Dr. Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
CHICAGO – Programmed cell death protein 1 (PD-1) inhibitors and combination ipilimumab and nivolumab prolonged survival, compared with ipilimumab alone in patients aged 80 and older with advanced melanoma, and the patients experienced no more adverse events than did patients of all age groups in prior published phase III studies, according to a retrospective case review.
Of 106 patients included in the current review, 74 received at least 1 dose of the anti–cytotoxic T-lymphocyte antigen 4 agent ipilimumab, 13 received at least 1 dose of an anti–PD-1 agent (pembrolizumab or nivolumab), and 8 received at least 1 dose of combination ipilimumab and nivolumab. Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively, Dr. Claire Frances Friedman reported at the annual meeting of the American Society of Clinical Oncology.
The differences between the groups did not reach statistical significance, said Dr. Friedman of Memorial Sloan Kettering Cancer Center, New York
Of note, a graphic representation of overall survival by treatment type shows a plateau after 24 months of treatment with ipilimumab (Ipi).
“About 20% of patients treated with Ipi have had a durable survival lasting many years,” she said, noting that this finding is similar to that based on pooled data from a previous study showing a survival rate of 22% with Ipi.
The findings are important because the highest incidence of advanced melanoma is among patients aged 80-84 years, and those over age 75 have a much higher mortality from melanoma, compared with younger patients, she said, adding that “the treatment of advanced melanoma has benefited from great strides in the area of immunotherapy, specifically the use of checkpoint-blocking antibodies.”
These antibodies work by blocking negative regulators of T-cell function, she explained.
The three used in this study are currently approved for the treatment of advanced melanoma. The combination of ipilimumab and nivolumab was approved when it was shown to improve progression-free survival vs. ipilimumab alone.
“Of note, responses to this class of drugs tend to be durable,” Dr. Friedman said.
In a prior study of nivolumab, the 5-year survival rate was 34%, she said.
However, the immune-related adverse events (irAEs) – “a unique set of side effects” associated with checkpoint-blocking antibodies – are concerning. The incidence of high-grade irAEs can vary with the type of checkpoint-blocking antibody – ranging from 16.3% with single-agent anti–PD-1 treatment to 55% with combination treatment in previous reports. The type of irAEs experienced tend to be similar across treatment types, and in a study published in 2015, were associated with a relatively high rate of treatment discontinuation (36.4% for combination treatment, 14.8% with ipilimumab, and 7.7% with nivolumab).
Given the increased mortality and morbidity in older patients, Dr. Friedman and her colleagues were interested in examining the toxicity and efficacy of checkpoint-blocking antibodies in patients aged 80 years and older. They performed the current review involving all patients aged 80 years and older at Memorial Sloan Kettering Cancer Center who were started on checkpoint blockade for stage 3 or 4 unresected melanoma between January 2008 and December 2015.
All patients had at least one infusion visit and one follow-up visit, and 12 of those in the Ipi group received subsequent anti–PD-1 therapy and were included in the toxicity analyses for both agents.
An irAE occurred in 88% of the 74 patients in the Ipi group, and 30% experienced a grade 3 or 4 irAE. The most common high-grade irAEs in this group were diarrhea, transaminitis, and rash.
Of the 25 patients receiving pembrolizumab or nivolumab, 84% experienced any irAE, and 16% experienced a grade 3 event. The most common adverse events in this group were pruritus, rash, fatigue, and musculoskeletal complaints, and one case each of grade 3 lipase, diarrhea, anemia, and nausea occurred.
Of the eight patients who received combination ipilimumab/nivolumab, 88% experienced any irAE and 63% experienced a high-grade 3 or 4 irAE. The most common high-grade irAEs in this group were elevated lipase, diarrhea, and transaminitis, occurring in 38%, 25%, and 25% of patients, respectively.
No deaths occurred in any of the groups, Dr. Friedman said.
The use of systemic steroids to manage irAEs was similar in the Ipi and anti–PD-1 monotherapy groups (28% in both groups), but 75% of the combination therapy patients were treated with systemic steroids. Infliximab was required for diarrhea in 7% of the Ipi patients and 38% of the combination therapy patients. None of the patients in any of the three groups required mycophenolate mofetil for steroid-refractory transaminitis, Dr. Friedman noted.
The findings suggest that when it comes to selecting the appropriate treatment for older patients, there are several things to consider, she said.
For example, in the elderly population, diarrhea and colitis are associated with an increased risk of dehydration and subsequent renal failure. Also, administering prednisone could lead to delirium or altered mental status, and can be of concern due to the increased incidence of comorbid conditions such as high blood pressure and diabetes, which may be worsened by steroid therapy.
Another concern is the fact that data from the rheumatology literature suggest that infliximab in the elderly may lead to increased risk of infections; a caveat is that this was in cases involving repeated doses, Dr. Friedman noted.
Treatment discontinuation was high across the groups, with 41%, 72%, and 63% of those in the Ipi, anti–PD-1, and combination therapy groups stopping treatment early. Toxicity was the reason for discontinuation in 31%, 20%, and 50%, respectively. Progression of disease or other unrelated issues were the reasons for discontinuation in the remaining patients.
“Patients aged 80 and older treated with ipilimumab had durable survival at a rate similar to that of the general population,” Dr. Friedman said.
Further, median overall survival is prolonged in older patients treated with anti–PD-1 drugs and with combination nivolumab/ipilimumab, but not significantly.
Importantly, irAEs occur more frequently with ipilimumab-based regimens in this population, she said, noting that discontinuation rates for toxicity are also higher in those aged 80 and older.
“So given these data, as clinicians we need to start thinking about individualizing treatment for our patients – using more than just the age. Physicians should consider incorporating assessments of functional status, such as the Mini-Cog or the Get Up and Go Test when deciding which of the checkpoint antibodies is appropriate for any individual patients,” she said, adding that as the population ages, more data in the elderly population will be needed.
“Clinicians should encourage older folks to participate in immuno-oncology clinical trials, and I hope that these data will encourage physicians to consider the use of checkpoint-blocking antibodies in the treatment of other malignancy types such as non–small cell lung cancer and bladder cancer, which also have a large proportion of adults over the age of 75,” she concluded.
Dr. Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: PD-1 inhibitors and combination ipilimumab/nivolumab prolonged survival vs. ipilimumab alone in patients aged 80 and older with advanced melanoma, and had an adverse event profile similar to that in all age groups.
Major finding: Median survival was 7.5 months, 14.2 months, and 23.5 months for ipilimumab, anti–PD-1 agents, and combination therapy groups, respectively. The survival rates at 1 year were 40%, 56%, and 63%, respectively,
Data source: A retrospective review of 106 cases.
Disclosures: Dr Friedman reported receiving funds for travel, accommodations, and expenses from Bristol-Myers Squibb.
Acalabrutinib shows efficacy as monotherapy in untreated CLL
CHICAGO – Acalabrutinib, which has shown efficacy in relapsed chronic lymphocytic leukemia, has now shown efficacy as a monotherapy for patients with previously untreated CLL, based on results from an ongoing phase I-II study presented as a poster at the annual meeting of the American Society of Clinical Oncology.
In a 74-patient study, best overall response rate was 96%, and median time to response was 2 months. CLL has not progressed in any of the patients, and none have experienced Richter’s transformation, Dr. John C. Byrd, the D. Warren Brown Chair of Leukemia Research at The Ohio State University, Columbus, and his colleagues reported. Based on its favorable safety profile and durable response rates, a phase III trialof acalabrutinib therapy has been initiated (NCT02475681).
Oral acalabrutinib was given at doses of 100 mg twice daily to 37 patients or 200 mg daily to 37 other patients. About half of the patients had bulky lymph nodes of at least 5 cm and 38 of 67 patients had an unmutated IGHV gene. Median time on the study was 11 months.
All patients had rapid declines in lymphadenopathy. Both dose schedules were associated with clinical activity, with Bruton’s tyrosine kinase occupancy highest at 98% with twice-daily dosing and 93% with once-daily dosing. Treatment-related lymphocytosis occurred in 39 of 74 patients and resolved in 38 of the 39. In general, lymphocytosis peaked at a median of 1 week and resolved by a median of 7 weeks.
Acalabrutinib was well tolerated with 72 of 74 patients continuing on the drug. Most adverse events were grade 2 or less, and included headache (42%), diarrhea (35%), arthralgia (22%), contusion (18%), nausea (18%) and increased weight (18%). Grade 3-4 adverse events that occurred in at least two patients included syncope (two patients) and hypertension (two patients). There was one grade 3 upper GI bleed from a gastric ulcer and aspirin use, and one grade 5 case of pneumonia. No atrial fibrillation was reported.
Dr. Byrd receives research funding from Acerta Pharma, the maker of acalabrutinib, as well as from Genentech and Pharmacyclics.
On Twitter @maryjodales
CHICAGO – Acalabrutinib, which has shown efficacy in relapsed chronic lymphocytic leukemia, has now shown efficacy as a monotherapy for patients with previously untreated CLL, based on results from an ongoing phase I-II study presented as a poster at the annual meeting of the American Society of Clinical Oncology.
In a 74-patient study, best overall response rate was 96%, and median time to response was 2 months. CLL has not progressed in any of the patients, and none have experienced Richter’s transformation, Dr. John C. Byrd, the D. Warren Brown Chair of Leukemia Research at The Ohio State University, Columbus, and his colleagues reported. Based on its favorable safety profile and durable response rates, a phase III trialof acalabrutinib therapy has been initiated (NCT02475681).
Oral acalabrutinib was given at doses of 100 mg twice daily to 37 patients or 200 mg daily to 37 other patients. About half of the patients had bulky lymph nodes of at least 5 cm and 38 of 67 patients had an unmutated IGHV gene. Median time on the study was 11 months.
All patients had rapid declines in lymphadenopathy. Both dose schedules were associated with clinical activity, with Bruton’s tyrosine kinase occupancy highest at 98% with twice-daily dosing and 93% with once-daily dosing. Treatment-related lymphocytosis occurred in 39 of 74 patients and resolved in 38 of the 39. In general, lymphocytosis peaked at a median of 1 week and resolved by a median of 7 weeks.
Acalabrutinib was well tolerated with 72 of 74 patients continuing on the drug. Most adverse events were grade 2 or less, and included headache (42%), diarrhea (35%), arthralgia (22%), contusion (18%), nausea (18%) and increased weight (18%). Grade 3-4 adverse events that occurred in at least two patients included syncope (two patients) and hypertension (two patients). There was one grade 3 upper GI bleed from a gastric ulcer and aspirin use, and one grade 5 case of pneumonia. No atrial fibrillation was reported.
Dr. Byrd receives research funding from Acerta Pharma, the maker of acalabrutinib, as well as from Genentech and Pharmacyclics.
On Twitter @maryjodales
CHICAGO – Acalabrutinib, which has shown efficacy in relapsed chronic lymphocytic leukemia, has now shown efficacy as a monotherapy for patients with previously untreated CLL, based on results from an ongoing phase I-II study presented as a poster at the annual meeting of the American Society of Clinical Oncology.
In a 74-patient study, best overall response rate was 96%, and median time to response was 2 months. CLL has not progressed in any of the patients, and none have experienced Richter’s transformation, Dr. John C. Byrd, the D. Warren Brown Chair of Leukemia Research at The Ohio State University, Columbus, and his colleagues reported. Based on its favorable safety profile and durable response rates, a phase III trialof acalabrutinib therapy has been initiated (NCT02475681).
Oral acalabrutinib was given at doses of 100 mg twice daily to 37 patients or 200 mg daily to 37 other patients. About half of the patients had bulky lymph nodes of at least 5 cm and 38 of 67 patients had an unmutated IGHV gene. Median time on the study was 11 months.
All patients had rapid declines in lymphadenopathy. Both dose schedules were associated with clinical activity, with Bruton’s tyrosine kinase occupancy highest at 98% with twice-daily dosing and 93% with once-daily dosing. Treatment-related lymphocytosis occurred in 39 of 74 patients and resolved in 38 of the 39. In general, lymphocytosis peaked at a median of 1 week and resolved by a median of 7 weeks.
Acalabrutinib was well tolerated with 72 of 74 patients continuing on the drug. Most adverse events were grade 2 or less, and included headache (42%), diarrhea (35%), arthralgia (22%), contusion (18%), nausea (18%) and increased weight (18%). Grade 3-4 adverse events that occurred in at least two patients included syncope (two patients) and hypertension (two patients). There was one grade 3 upper GI bleed from a gastric ulcer and aspirin use, and one grade 5 case of pneumonia. No atrial fibrillation was reported.
Dr. Byrd receives research funding from Acerta Pharma, the maker of acalabrutinib, as well as from Genentech and Pharmacyclics.
On Twitter @maryjodales
FROM 2016 ASCO ANNUAL MEETING
Key clinical point: Acalabrutinib, which has shown efficacy in relapsed CLL, has now shown efficacy as a monotherapy for patients with previously untreated CLL.
Major finding: In a 74-patient study, best overall response rate was 96%, and median time to response was 2 months.
Data source: An ongoing phase I-II study.
Disclosures: Dr. Byrd receives research funding from Acerta Pharma, the maker of acalabrutinib, as well as from Genentech and Pharmacyclics.
Early results promising for pembrolizumab in combination regimens for NSCLC
CHICAGO – Overall response rates ranged from 48% to 71% when pembrolizumab was added to chemotherapy combinations in patients with advanced non–small cell lung cancer (NSCLC), and close to 30% when added to an R2 inhibitor in patients with advanced NSCLC and other tumor types, according to two early phase clinical trials presented at the annual meeting of the American Society of Clinical Oncology.
Safety and efficacy of pembrolizumab in combination with either carboplatin and paclitaxel; carboplatin, paclitaxel, and bevacizumab; or carboplatin and pemetrexed were evaluated in the KEYNOTE-021 trial. Patients were split between two doses of pembrolizumab: 2 or 10 mg/kg every 3 weeks. The overall response rate for the entire cohort of 74 patients with advanced NSCLC was 57% (95% confidence interval, 45-68), reported Dr. Shirish Gadgeel of Karmanos Cancer Institute, Detroit.
For the 25 patients who received pembrolizumab, carboplatin, and paclitaxel (cohort A) the overall response was 52% (95% CI, 31-72). For the 25 patients who also received bevacizumab in addition to pembrolizumab, carboplatin, and paclitaxel (cohort B), the overall response rate was 48% (95% CI, 28-69).
Among 24 patients who received pemetrexed with pembrolizumab and carboplatin (cohort C), the overall response was the highest at 71% (95% CI, 49-87).
Grade 3 or 4 adverse events occurred in 36%, 46%, and 42% of patients in cohort A, B, and C, respectively. The most common adverse events were anemia and neutropenia including febrile neutropenia.
“Pembro in combination with standard chemotherapy regimens is feasible and yields substantial clinical efficacy regardless of pembro dose or PD-L1 status in treatment-naïve advanced NSCLC,” Dr Gadgeel said.
A randomized phase III study evaluating pemetrexed/platinum with or without pembrolizumab is currently recruiting, he said.
These combinations, while perhaps better than chemotherapy alone, may be no better than just immunotherapy alone, session moderator Dr. Scott Antonia of the Moffitt Cancer Center, Tampa, Fla., commented. “And the truth is that concurrent therapy is clearly more toxic.”
In another study presented at the meeting, led by Dr. Roy Herbst of Yale Cancer Center, New Haven, Conn., pembrolizumab was combined with ramucirumab, a recently approved vascular endothelial growth factor receptor–2 inhibitor. “Hallmarks of tumor growth include angiogenesis and immunosuppression,” Dr. Herbst and his coauthors wrote in their abstract, noting that this was the first study to combine these agents to target both processes simultaneously.
“The idea is the R2 inhibitor might have an effect on the microimmune environment and drive T cells into the tumor,” Dr. Herbst said.
The study cohort included patients with advanced gastric or gastroesophageal junction adenocarcinoma, NSCLC, and urothelial carcinoma.
Overall, the response rate was close to 30% in the first 10 patients, Dr. Herbst reported. Furthermore, the preliminary data did not reveal any unexpected safety signals.
Although the data was from a small, ongoing phase I trial, Dr. Herbst noted the data is promising. Researchers are continuing to collect data on the safety profile of this drug combination, and are planning to enroll patients with additional tumor types.
The KEYNOTE-021 trial was funded by Merck Sharp and Dohme. Dr. Gadgeel reported having a consulting or advisory role and receiving research funding from multiple companies. The trial headed by Dr. Herbst was funded by Eli Lilly. Dr. Herbst reported having a consulting or advisory role and receiving honoraria and research funding from multiple companies including Eli Lilly.
On Twitter @jessnicolecraig
CHICAGO – Overall response rates ranged from 48% to 71% when pembrolizumab was added to chemotherapy combinations in patients with advanced non–small cell lung cancer (NSCLC), and close to 30% when added to an R2 inhibitor in patients with advanced NSCLC and other tumor types, according to two early phase clinical trials presented at the annual meeting of the American Society of Clinical Oncology.
Safety and efficacy of pembrolizumab in combination with either carboplatin and paclitaxel; carboplatin, paclitaxel, and bevacizumab; or carboplatin and pemetrexed were evaluated in the KEYNOTE-021 trial. Patients were split between two doses of pembrolizumab: 2 or 10 mg/kg every 3 weeks. The overall response rate for the entire cohort of 74 patients with advanced NSCLC was 57% (95% confidence interval, 45-68), reported Dr. Shirish Gadgeel of Karmanos Cancer Institute, Detroit.
For the 25 patients who received pembrolizumab, carboplatin, and paclitaxel (cohort A) the overall response was 52% (95% CI, 31-72). For the 25 patients who also received bevacizumab in addition to pembrolizumab, carboplatin, and paclitaxel (cohort B), the overall response rate was 48% (95% CI, 28-69).
Among 24 patients who received pemetrexed with pembrolizumab and carboplatin (cohort C), the overall response was the highest at 71% (95% CI, 49-87).
Grade 3 or 4 adverse events occurred in 36%, 46%, and 42% of patients in cohort A, B, and C, respectively. The most common adverse events were anemia and neutropenia including febrile neutropenia.
“Pembro in combination with standard chemotherapy regimens is feasible and yields substantial clinical efficacy regardless of pembro dose or PD-L1 status in treatment-naïve advanced NSCLC,” Dr Gadgeel said.
A randomized phase III study evaluating pemetrexed/platinum with or without pembrolizumab is currently recruiting, he said.
These combinations, while perhaps better than chemotherapy alone, may be no better than just immunotherapy alone, session moderator Dr. Scott Antonia of the Moffitt Cancer Center, Tampa, Fla., commented. “And the truth is that concurrent therapy is clearly more toxic.”
In another study presented at the meeting, led by Dr. Roy Herbst of Yale Cancer Center, New Haven, Conn., pembrolizumab was combined with ramucirumab, a recently approved vascular endothelial growth factor receptor–2 inhibitor. “Hallmarks of tumor growth include angiogenesis and immunosuppression,” Dr. Herbst and his coauthors wrote in their abstract, noting that this was the first study to combine these agents to target both processes simultaneously.
“The idea is the R2 inhibitor might have an effect on the microimmune environment and drive T cells into the tumor,” Dr. Herbst said.
The study cohort included patients with advanced gastric or gastroesophageal junction adenocarcinoma, NSCLC, and urothelial carcinoma.
Overall, the response rate was close to 30% in the first 10 patients, Dr. Herbst reported. Furthermore, the preliminary data did not reveal any unexpected safety signals.
Although the data was from a small, ongoing phase I trial, Dr. Herbst noted the data is promising. Researchers are continuing to collect data on the safety profile of this drug combination, and are planning to enroll patients with additional tumor types.
The KEYNOTE-021 trial was funded by Merck Sharp and Dohme. Dr. Gadgeel reported having a consulting or advisory role and receiving research funding from multiple companies. The trial headed by Dr. Herbst was funded by Eli Lilly. Dr. Herbst reported having a consulting or advisory role and receiving honoraria and research funding from multiple companies including Eli Lilly.
On Twitter @jessnicolecraig
CHICAGO – Overall response rates ranged from 48% to 71% when pembrolizumab was added to chemotherapy combinations in patients with advanced non–small cell lung cancer (NSCLC), and close to 30% when added to an R2 inhibitor in patients with advanced NSCLC and other tumor types, according to two early phase clinical trials presented at the annual meeting of the American Society of Clinical Oncology.
Safety and efficacy of pembrolizumab in combination with either carboplatin and paclitaxel; carboplatin, paclitaxel, and bevacizumab; or carboplatin and pemetrexed were evaluated in the KEYNOTE-021 trial. Patients were split between two doses of pembrolizumab: 2 or 10 mg/kg every 3 weeks. The overall response rate for the entire cohort of 74 patients with advanced NSCLC was 57% (95% confidence interval, 45-68), reported Dr. Shirish Gadgeel of Karmanos Cancer Institute, Detroit.
For the 25 patients who received pembrolizumab, carboplatin, and paclitaxel (cohort A) the overall response was 52% (95% CI, 31-72). For the 25 patients who also received bevacizumab in addition to pembrolizumab, carboplatin, and paclitaxel (cohort B), the overall response rate was 48% (95% CI, 28-69).
Among 24 patients who received pemetrexed with pembrolizumab and carboplatin (cohort C), the overall response was the highest at 71% (95% CI, 49-87).
Grade 3 or 4 adverse events occurred in 36%, 46%, and 42% of patients in cohort A, B, and C, respectively. The most common adverse events were anemia and neutropenia including febrile neutropenia.
“Pembro in combination with standard chemotherapy regimens is feasible and yields substantial clinical efficacy regardless of pembro dose or PD-L1 status in treatment-naïve advanced NSCLC,” Dr Gadgeel said.
A randomized phase III study evaluating pemetrexed/platinum with or without pembrolizumab is currently recruiting, he said.
These combinations, while perhaps better than chemotherapy alone, may be no better than just immunotherapy alone, session moderator Dr. Scott Antonia of the Moffitt Cancer Center, Tampa, Fla., commented. “And the truth is that concurrent therapy is clearly more toxic.”
In another study presented at the meeting, led by Dr. Roy Herbst of Yale Cancer Center, New Haven, Conn., pembrolizumab was combined with ramucirumab, a recently approved vascular endothelial growth factor receptor–2 inhibitor. “Hallmarks of tumor growth include angiogenesis and immunosuppression,” Dr. Herbst and his coauthors wrote in their abstract, noting that this was the first study to combine these agents to target both processes simultaneously.
“The idea is the R2 inhibitor might have an effect on the microimmune environment and drive T cells into the tumor,” Dr. Herbst said.
The study cohort included patients with advanced gastric or gastroesophageal junction adenocarcinoma, NSCLC, and urothelial carcinoma.
Overall, the response rate was close to 30% in the first 10 patients, Dr. Herbst reported. Furthermore, the preliminary data did not reveal any unexpected safety signals.
Although the data was from a small, ongoing phase I trial, Dr. Herbst noted the data is promising. Researchers are continuing to collect data on the safety profile of this drug combination, and are planning to enroll patients with additional tumor types.
The KEYNOTE-021 trial was funded by Merck Sharp and Dohme. Dr. Gadgeel reported having a consulting or advisory role and receiving research funding from multiple companies. The trial headed by Dr. Herbst was funded by Eli Lilly. Dr. Herbst reported having a consulting or advisory role and receiving honoraria and research funding from multiple companies including Eli Lilly.
On Twitter @jessnicolecraig
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Overall response rates ranged from 30% to 71% when pembrolizumab was added to chemotherapy combinations or paired with an R2 inhibitor in patients with advanced NSCLC and other tumor types.
Major finding: Overall response rates ranged from 48% to 71% for NSCLC patients receiving pembrolizumab in combination with standard chemotherapy regimens, one with bevacizumab. In combinations with an R2-inhibitor, the overall response rate was nearly 30% for patients with advanced NSCLC or gastric or urothelial tumors.
Data source: The phase I/II KEYNOTE-021 trial of 74 patients with advanced NSCLC, and the phase I trial that included patients with advanced gastric or gastroesophageal junction adenocarcinoma, NSCLC, and urothelial carcinoma.
Disclosures: The KEYNOTE-021 trial was funded by Merck Sharp and Dohme. Dr. Gadgeel reported having a consulting or advisory role and receiving research funding from multiple companies. The trial headed by Dr. Herbst was funded by Eli Lilly. Dr. Herbst reported having a consulting or advisory role and receiving honoraria and research funding from multiple companies including Eli Lilly.
Metformin increases efficacy of everolimus-letrozole combo for EEC
CHICAGO – The novel combination of everolimus, letrozole, and metformin was associated with improved clinical benefit and objective response rates, compared with everolimus and letrozole without metformin in patients with advanced or recurrent, previously treated endometrioid endometrial cancer (EEC) in an ongoing open-label, single-arm, prospective phase II study.
In 49 patients with an evaluable response to the combination therapy, 29 (60%) derived clinical benefit – defined as complete response, partial response, or stable disease confirmed at 8 weeks by RECIST 1.1, Dr. Pamela T. Soliman reported at the annual meeting of the American Society of Clinical Oncology.
The best overall response to date was 29% partial response and 31% stable disease (60% clinical benefit rate) with a median of six cycles, said Dr. Soliman of the University of Texas MD Anderson Cancer Center, Houston.
So far, 58 patients have been enrolled in the study, and 194 cycles have been completed. Eight patients have received more than 14 cycles, and of those, 6 have achieved a partial response and 2 have stable disease.
Eight remain on active treatment.
“Of note, at the time of first assessment at 8 weeks, the objective response rate was only 14%, so over half of the responses were seen after at least 4 cycles of therapy were administered,” she said.
Endometrial cancer is the most common gynecologic malignancy, with an estimated 52,000 cases to be diagnosed this year. Most cases are diagnosed early, and the overall prognosis is good; the 5-year survival rate is nearly 90%.
“Unfortunately, for those women who recur, treatment options are limited,” Dr. Soliman said, noting that the best response rate seen in studies of various single and combination chemotherapy agents has been about 25% – occurring with paclitaxel and with bevacizumab-temsirolimus.
The novel combination therapy used in the current study was developed based on prior studies, including one conducted by Dr. Soliman and her colleagues, showing promising results with combination everolimus and letrozole for recurrent EEC. The investigators found that in one study of 35 patients on the everolimus and letrozole regimen, the overall response rate was 23%, the clinical benefit rate was 38%, and progression-free survival was 1.9 months, but they noticed that in a subgroup of patients who had received metformin during the course of the study, the corresponding rates were 56%, 78%, and 14 months. Preclinical data suggested that the benefit of adding metformin to the combination was particularly pronounced in patients with KRAS mutation. The current study was designed to evaluate this approach.
Study subjects had advanced/recurrent EEC and up to two prior chemotherapy regimens for recurrence. Molecular testing was performed to determine KRAS mutation status.
A pretreatment biopsy was performed for histological confirmation and molecular analysis, followed by a 1 week metformin lead in and then treatment with 10 mg oral everolimus daily, 2.5 mg or oral letrozole daily, and 500 mg of oral metformin twice daily. A single dose reduction of everolimus was permitted, and patients were treated until their disease progressed or until they experienced toxicity. Response was evaluated by imaging at 8 weeks or after 2 cycles of therapy, and confirmation of response was performed at 16 weeks, or after 4 cycles of therapy.
In this preliminary evaluation, no significant difference was seen in the clinical benefit rate based on KRAS mutation status (67% in 15 patients with KRAS mutation, 77% in 26 patients without the mutation), but further correlation between molecular findings and response are currently being evaluated.
The most common side effects were diarrhea, anemia, fatigue, nausea, mucositis, hypertriglyceridemia, and elevated aspartate aminotransferase, Dr. Soliman said, noting that 43% of patients experienced a grade 3/4 toxicity, 4 required a dose reduction, and 2 withdrew from the study.
Additionally, one patient with a partial response after 1 cycle was removed from treatment due to increased liver enzymes.
A comparison with findings from the prior everolimus and letrozole study showed that metformin did not add significant toxicity to the regimen, she noted.
“The addition of metformin to the everolimus and letrozole backbone, increased the clinical benefit rate from 40% to 60%. Overall the toxicity of all three drugs was manageable and few patients came off study due to toxicity ... Based on these findings and other studies, the use of targeted therapy in women with recurrent endometrial endometrioid cancer appears to benefit patients over standard cytotoxic chemotherapy,” Dr. Soliman concluded, adding that further development of this combination for use in patients with EEC is warranted.
She noted that an ongoing randomized clinical trial – GOG 3007 – is comparing everolimus and letrozole with tamoxifen alternating with megestrol acetate.
“This trial has recently completed accrual and results are pending. This will allow us to better identify how this treatment strategy compares to hormonal therapy alone in women with recurrent endometrial cancer. In addition, at our institution we are investigating other agents to be added to the everolimus and letrozole backbone to enhance response to patients with endometrioid endometrial cancer,” she said.
Dr. Soliman reported receiving research funding (to her institution) from Novartis.
CHICAGO – The novel combination of everolimus, letrozole, and metformin was associated with improved clinical benefit and objective response rates, compared with everolimus and letrozole without metformin in patients with advanced or recurrent, previously treated endometrioid endometrial cancer (EEC) in an ongoing open-label, single-arm, prospective phase II study.
In 49 patients with an evaluable response to the combination therapy, 29 (60%) derived clinical benefit – defined as complete response, partial response, or stable disease confirmed at 8 weeks by RECIST 1.1, Dr. Pamela T. Soliman reported at the annual meeting of the American Society of Clinical Oncology.
The best overall response to date was 29% partial response and 31% stable disease (60% clinical benefit rate) with a median of six cycles, said Dr. Soliman of the University of Texas MD Anderson Cancer Center, Houston.
So far, 58 patients have been enrolled in the study, and 194 cycles have been completed. Eight patients have received more than 14 cycles, and of those, 6 have achieved a partial response and 2 have stable disease.
Eight remain on active treatment.
“Of note, at the time of first assessment at 8 weeks, the objective response rate was only 14%, so over half of the responses were seen after at least 4 cycles of therapy were administered,” she said.
Endometrial cancer is the most common gynecologic malignancy, with an estimated 52,000 cases to be diagnosed this year. Most cases are diagnosed early, and the overall prognosis is good; the 5-year survival rate is nearly 90%.
“Unfortunately, for those women who recur, treatment options are limited,” Dr. Soliman said, noting that the best response rate seen in studies of various single and combination chemotherapy agents has been about 25% – occurring with paclitaxel and with bevacizumab-temsirolimus.
The novel combination therapy used in the current study was developed based on prior studies, including one conducted by Dr. Soliman and her colleagues, showing promising results with combination everolimus and letrozole for recurrent EEC. The investigators found that in one study of 35 patients on the everolimus and letrozole regimen, the overall response rate was 23%, the clinical benefit rate was 38%, and progression-free survival was 1.9 months, but they noticed that in a subgroup of patients who had received metformin during the course of the study, the corresponding rates were 56%, 78%, and 14 months. Preclinical data suggested that the benefit of adding metformin to the combination was particularly pronounced in patients with KRAS mutation. The current study was designed to evaluate this approach.
Study subjects had advanced/recurrent EEC and up to two prior chemotherapy regimens for recurrence. Molecular testing was performed to determine KRAS mutation status.
A pretreatment biopsy was performed for histological confirmation and molecular analysis, followed by a 1 week metformin lead in and then treatment with 10 mg oral everolimus daily, 2.5 mg or oral letrozole daily, and 500 mg of oral metformin twice daily. A single dose reduction of everolimus was permitted, and patients were treated until their disease progressed or until they experienced toxicity. Response was evaluated by imaging at 8 weeks or after 2 cycles of therapy, and confirmation of response was performed at 16 weeks, or after 4 cycles of therapy.
In this preliminary evaluation, no significant difference was seen in the clinical benefit rate based on KRAS mutation status (67% in 15 patients with KRAS mutation, 77% in 26 patients without the mutation), but further correlation between molecular findings and response are currently being evaluated.
The most common side effects were diarrhea, anemia, fatigue, nausea, mucositis, hypertriglyceridemia, and elevated aspartate aminotransferase, Dr. Soliman said, noting that 43% of patients experienced a grade 3/4 toxicity, 4 required a dose reduction, and 2 withdrew from the study.
Additionally, one patient with a partial response after 1 cycle was removed from treatment due to increased liver enzymes.
A comparison with findings from the prior everolimus and letrozole study showed that metformin did not add significant toxicity to the regimen, she noted.
“The addition of metformin to the everolimus and letrozole backbone, increased the clinical benefit rate from 40% to 60%. Overall the toxicity of all three drugs was manageable and few patients came off study due to toxicity ... Based on these findings and other studies, the use of targeted therapy in women with recurrent endometrial endometrioid cancer appears to benefit patients over standard cytotoxic chemotherapy,” Dr. Soliman concluded, adding that further development of this combination for use in patients with EEC is warranted.
She noted that an ongoing randomized clinical trial – GOG 3007 – is comparing everolimus and letrozole with tamoxifen alternating with megestrol acetate.
“This trial has recently completed accrual and results are pending. This will allow us to better identify how this treatment strategy compares to hormonal therapy alone in women with recurrent endometrial cancer. In addition, at our institution we are investigating other agents to be added to the everolimus and letrozole backbone to enhance response to patients with endometrioid endometrial cancer,” she said.
Dr. Soliman reported receiving research funding (to her institution) from Novartis.
CHICAGO – The novel combination of everolimus, letrozole, and metformin was associated with improved clinical benefit and objective response rates, compared with everolimus and letrozole without metformin in patients with advanced or recurrent, previously treated endometrioid endometrial cancer (EEC) in an ongoing open-label, single-arm, prospective phase II study.
In 49 patients with an evaluable response to the combination therapy, 29 (60%) derived clinical benefit – defined as complete response, partial response, or stable disease confirmed at 8 weeks by RECIST 1.1, Dr. Pamela T. Soliman reported at the annual meeting of the American Society of Clinical Oncology.
The best overall response to date was 29% partial response and 31% stable disease (60% clinical benefit rate) with a median of six cycles, said Dr. Soliman of the University of Texas MD Anderson Cancer Center, Houston.
So far, 58 patients have been enrolled in the study, and 194 cycles have been completed. Eight patients have received more than 14 cycles, and of those, 6 have achieved a partial response and 2 have stable disease.
Eight remain on active treatment.
“Of note, at the time of first assessment at 8 weeks, the objective response rate was only 14%, so over half of the responses were seen after at least 4 cycles of therapy were administered,” she said.
Endometrial cancer is the most common gynecologic malignancy, with an estimated 52,000 cases to be diagnosed this year. Most cases are diagnosed early, and the overall prognosis is good; the 5-year survival rate is nearly 90%.
“Unfortunately, for those women who recur, treatment options are limited,” Dr. Soliman said, noting that the best response rate seen in studies of various single and combination chemotherapy agents has been about 25% – occurring with paclitaxel and with bevacizumab-temsirolimus.
The novel combination therapy used in the current study was developed based on prior studies, including one conducted by Dr. Soliman and her colleagues, showing promising results with combination everolimus and letrozole for recurrent EEC. The investigators found that in one study of 35 patients on the everolimus and letrozole regimen, the overall response rate was 23%, the clinical benefit rate was 38%, and progression-free survival was 1.9 months, but they noticed that in a subgroup of patients who had received metformin during the course of the study, the corresponding rates were 56%, 78%, and 14 months. Preclinical data suggested that the benefit of adding metformin to the combination was particularly pronounced in patients with KRAS mutation. The current study was designed to evaluate this approach.
Study subjects had advanced/recurrent EEC and up to two prior chemotherapy regimens for recurrence. Molecular testing was performed to determine KRAS mutation status.
A pretreatment biopsy was performed for histological confirmation and molecular analysis, followed by a 1 week metformin lead in and then treatment with 10 mg oral everolimus daily, 2.5 mg or oral letrozole daily, and 500 mg of oral metformin twice daily. A single dose reduction of everolimus was permitted, and patients were treated until their disease progressed or until they experienced toxicity. Response was evaluated by imaging at 8 weeks or after 2 cycles of therapy, and confirmation of response was performed at 16 weeks, or after 4 cycles of therapy.
In this preliminary evaluation, no significant difference was seen in the clinical benefit rate based on KRAS mutation status (67% in 15 patients with KRAS mutation, 77% in 26 patients without the mutation), but further correlation between molecular findings and response are currently being evaluated.
The most common side effects were diarrhea, anemia, fatigue, nausea, mucositis, hypertriglyceridemia, and elevated aspartate aminotransferase, Dr. Soliman said, noting that 43% of patients experienced a grade 3/4 toxicity, 4 required a dose reduction, and 2 withdrew from the study.
Additionally, one patient with a partial response after 1 cycle was removed from treatment due to increased liver enzymes.
A comparison with findings from the prior everolimus and letrozole study showed that metformin did not add significant toxicity to the regimen, she noted.
“The addition of metformin to the everolimus and letrozole backbone, increased the clinical benefit rate from 40% to 60%. Overall the toxicity of all three drugs was manageable and few patients came off study due to toxicity ... Based on these findings and other studies, the use of targeted therapy in women with recurrent endometrial endometrioid cancer appears to benefit patients over standard cytotoxic chemotherapy,” Dr. Soliman concluded, adding that further development of this combination for use in patients with EEC is warranted.
She noted that an ongoing randomized clinical trial – GOG 3007 – is comparing everolimus and letrozole with tamoxifen alternating with megestrol acetate.
“This trial has recently completed accrual and results are pending. This will allow us to better identify how this treatment strategy compares to hormonal therapy alone in women with recurrent endometrial cancer. In addition, at our institution we are investigating other agents to be added to the everolimus and letrozole backbone to enhance response to patients with endometrioid endometrial cancer,” she said.
Dr. Soliman reported receiving research funding (to her institution) from Novartis.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Adding metformin to the everolimus and letrozole backbone increases the clinical benefit rate in patients with advanced or recurrent, previously treated endometrioid endometrial cancer.
Major finding: The combination was associated with a confirmed clinical benefit rate of 60% and an objective response rate of 29%.
Data source: An open-label, single-arm, prospective phase II study of 49 patients (with evaluable response thus far).
Disclosures: Dr. Soliman reported receiving research funding (to her institution) from Novartis.
Better ways to drive CAR T-cell therapy

© ASCO/Brian Powers
CHICAGO—Treatment dose and schedule, as well as a patient’s tumor burden, influence the outcome of therapy with chimeric antigen receptor (CAR) T cells, according to research presented at the 2016 ASCO Annual Meeting.
One presentation suggested the dose and schedule of CTL019 can impact both complete response (CR) rates and the incidence of cytokine release syndrome (CRS).
Another presentation indicated that disease burden may determine the risk of toxicity and correlate with the efficacy of JCAR015.
CTL019
Noelle Frey, MD, of the University of Pennsylvania in Philadelphia, presented results observed with CTL019 in 30 adults with relapsed/refractory B-cell acute lymphoblastic leukemia (ALL) treated on 2 trials (NCT02030847 and NCT01029366) as abstract 7002.*
Dr Frey noted that CAR T-cell therapies, including CTL019, have led to “unprecedented remission rates of between 70% and 90%.” However, immune activation that leads to high response rates also confers significant treatment-related toxicity—namely, CRS.
She reported that, in the 2 trials of adult ALL patients, a high dose of CTL019 led to a 100% response rate and a 100% CRS rate. Splitting the dose over 3 days led to an 86% response rate and a 66% CRS rate. A single low dose reduced efficacy to 33% and CRS to 66%.
Three of 6 patients who received a single high dose developed CRS and died within weeks. All of these patients had infections or sepsis that likely led to their deaths, Dr Frey said. She suggested clinicians aggressively monitor infections and treat with antimicrobials prior to CAR T-cell therapy.
“The infusion dose and schedule of CTL019 correlate with toxicity and response,” she observed. “A fractionated dosing scheme allows for real-time intra-patient dose modification in response to toxicity and the maintenance of high response rates. Concurrent sepsis and CRS confers a poor outcome.”
Dr Frey said future studies will determine the optimal approach to minimize toxicity while maintaining high efficacy. A fractionated dosing scheme is only one way to mitigate CRS. Other attractive approaches include inverse dosing based on disease burden and varying the timing of anti-cytokine-directed therapy.
JCAR015
Jae Park, MD, of Memorial Sloan Kettering Cancer Center in New York, reported data from a phase 1 clinical trial (NCT01044069) of JCAR015 in 51 adults with relapsed/refractory ALL as abstract 7003.*
Treatment with JCAR015 led to high CR rates and minimal residual disease (MRD)-negativity, regardless of the amount of disease at baseline. However, outcomes were superior among patients with minimal disease at baseline.
For patients with morphologic disease, 77% achieved a CR by 20 days after treatment, and 90% were MRD-negative.
For those with minimal disease, 90% achieved a CR by 25 days after treatment, and 78% were MRD-negative. These patients experienced significantly less CRS and neurotoxicity than patients with morphologic disease.
“High CR and MRD-negativity rates were observed regardless of pre-T-cell burden,” Dr Park noted. “We observed durable responses and survivals in a subset of patients with no subsequent allotransplant in both morphological and minimal disease cohorts.” ![]()
*Data in the abstracts differ from the presentations.

© ASCO/Brian Powers
CHICAGO—Treatment dose and schedule, as well as a patient’s tumor burden, influence the outcome of therapy with chimeric antigen receptor (CAR) T cells, according to research presented at the 2016 ASCO Annual Meeting.
One presentation suggested the dose and schedule of CTL019 can impact both complete response (CR) rates and the incidence of cytokine release syndrome (CRS).
Another presentation indicated that disease burden may determine the risk of toxicity and correlate with the efficacy of JCAR015.
CTL019
Noelle Frey, MD, of the University of Pennsylvania in Philadelphia, presented results observed with CTL019 in 30 adults with relapsed/refractory B-cell acute lymphoblastic leukemia (ALL) treated on 2 trials (NCT02030847 and NCT01029366) as abstract 7002.*
Dr Frey noted that CAR T-cell therapies, including CTL019, have led to “unprecedented remission rates of between 70% and 90%.” However, immune activation that leads to high response rates also confers significant treatment-related toxicity—namely, CRS.
She reported that, in the 2 trials of adult ALL patients, a high dose of CTL019 led to a 100% response rate and a 100% CRS rate. Splitting the dose over 3 days led to an 86% response rate and a 66% CRS rate. A single low dose reduced efficacy to 33% and CRS to 66%.
Three of 6 patients who received a single high dose developed CRS and died within weeks. All of these patients had infections or sepsis that likely led to their deaths, Dr Frey said. She suggested clinicians aggressively monitor infections and treat with antimicrobials prior to CAR T-cell therapy.
“The infusion dose and schedule of CTL019 correlate with toxicity and response,” she observed. “A fractionated dosing scheme allows for real-time intra-patient dose modification in response to toxicity and the maintenance of high response rates. Concurrent sepsis and CRS confers a poor outcome.”
Dr Frey said future studies will determine the optimal approach to minimize toxicity while maintaining high efficacy. A fractionated dosing scheme is only one way to mitigate CRS. Other attractive approaches include inverse dosing based on disease burden and varying the timing of anti-cytokine-directed therapy.
JCAR015
Jae Park, MD, of Memorial Sloan Kettering Cancer Center in New York, reported data from a phase 1 clinical trial (NCT01044069) of JCAR015 in 51 adults with relapsed/refractory ALL as abstract 7003.*
Treatment with JCAR015 led to high CR rates and minimal residual disease (MRD)-negativity, regardless of the amount of disease at baseline. However, outcomes were superior among patients with minimal disease at baseline.
For patients with morphologic disease, 77% achieved a CR by 20 days after treatment, and 90% were MRD-negative.
For those with minimal disease, 90% achieved a CR by 25 days after treatment, and 78% were MRD-negative. These patients experienced significantly less CRS and neurotoxicity than patients with morphologic disease.
“High CR and MRD-negativity rates were observed regardless of pre-T-cell burden,” Dr Park noted. “We observed durable responses and survivals in a subset of patients with no subsequent allotransplant in both morphological and minimal disease cohorts.” ![]()
*Data in the abstracts differ from the presentations.

© ASCO/Brian Powers
CHICAGO—Treatment dose and schedule, as well as a patient’s tumor burden, influence the outcome of therapy with chimeric antigen receptor (CAR) T cells, according to research presented at the 2016 ASCO Annual Meeting.
One presentation suggested the dose and schedule of CTL019 can impact both complete response (CR) rates and the incidence of cytokine release syndrome (CRS).
Another presentation indicated that disease burden may determine the risk of toxicity and correlate with the efficacy of JCAR015.
CTL019
Noelle Frey, MD, of the University of Pennsylvania in Philadelphia, presented results observed with CTL019 in 30 adults with relapsed/refractory B-cell acute lymphoblastic leukemia (ALL) treated on 2 trials (NCT02030847 and NCT01029366) as abstract 7002.*
Dr Frey noted that CAR T-cell therapies, including CTL019, have led to “unprecedented remission rates of between 70% and 90%.” However, immune activation that leads to high response rates also confers significant treatment-related toxicity—namely, CRS.
She reported that, in the 2 trials of adult ALL patients, a high dose of CTL019 led to a 100% response rate and a 100% CRS rate. Splitting the dose over 3 days led to an 86% response rate and a 66% CRS rate. A single low dose reduced efficacy to 33% and CRS to 66%.
Three of 6 patients who received a single high dose developed CRS and died within weeks. All of these patients had infections or sepsis that likely led to their deaths, Dr Frey said. She suggested clinicians aggressively monitor infections and treat with antimicrobials prior to CAR T-cell therapy.
“The infusion dose and schedule of CTL019 correlate with toxicity and response,” she observed. “A fractionated dosing scheme allows for real-time intra-patient dose modification in response to toxicity and the maintenance of high response rates. Concurrent sepsis and CRS confers a poor outcome.”
Dr Frey said future studies will determine the optimal approach to minimize toxicity while maintaining high efficacy. A fractionated dosing scheme is only one way to mitigate CRS. Other attractive approaches include inverse dosing based on disease burden and varying the timing of anti-cytokine-directed therapy.
JCAR015
Jae Park, MD, of Memorial Sloan Kettering Cancer Center in New York, reported data from a phase 1 clinical trial (NCT01044069) of JCAR015 in 51 adults with relapsed/refractory ALL as abstract 7003.*
Treatment with JCAR015 led to high CR rates and minimal residual disease (MRD)-negativity, regardless of the amount of disease at baseline. However, outcomes were superior among patients with minimal disease at baseline.
For patients with morphologic disease, 77% achieved a CR by 20 days after treatment, and 90% were MRD-negative.
For those with minimal disease, 90% achieved a CR by 25 days after treatment, and 78% were MRD-negative. These patients experienced significantly less CRS and neurotoxicity than patients with morphologic disease.
“High CR and MRD-negativity rates were observed regardless of pre-T-cell burden,” Dr Park noted. “We observed durable responses and survivals in a subset of patients with no subsequent allotransplant in both morphological and minimal disease cohorts.” ![]()
*Data in the abstracts differ from the presentations.
Lenalidomide maintenance after transplant improves OS in MM

the ASCO Annual Meeting
© ASCO/Todd Buchanan
CHICAGO—Lenalidomide maintenance after high-dose melphalan and autologous stem cell transplant (ASCT) should be considered the standard of care in
newly diagnosed multiple myeloma (MM) patients, according to a meta-analysis presented at the 2016 ASCO Annual Meeting.
Lenalidomide maintenance increased overall survival (OS), with a 26% reduction in the risk of death, representing an estimated 2.5-year increase in median survival.
Several studies have demonstrated that lenalidomide maintenance post-ASCT reduces the risk of disease progression or death in patients with MM by about 50%.
However, these studies were not powered for OS, said Philip McCarthy, MD, of Roswell Park Cancer Institute in Buffalo, New York.
With this in mind, Dr McCarthy and his colleagues conducted a meta-analysis to assess the effect of post-ASCT lenalidomide maintenance on OS using a pooled analysis of primary source patient data. A search revealed 17 randomized, controlled trials using lenalidomide post-ASCT.
Three trials met pre-specified inclusion criteria and had sufficient OS events to test a treatment effect. The studies intended for lenalidomide maintenance to be given until progression.
In these trials, 1209 MM patients, with a median age of 58, were randomized from 2005 to 2009 to receive lenalidomide (605 patients) at 10 mg/day on days 1-21/28 or days 1-28/28. The remaining 604 patients served as controls. Baseline characteristics were generally balanced between the 2 groups.
With a median follow-up of 6.6 years, 491 patients (41%) had died.
After induction and single (82%) or tandem (18%) ASCT, 55% of patients achieved a complete response (CR) or very good partial response.
The median OS has not been reached in the lenalidomide arm but was 86 months for the control arm.
“There is a 26% reduction in the risk of death, representing an estimated 2.5-year increase in median survival,” Dr McCarthy said.
The OS benefit favoring lenalidomide was generally consistent across the majority of subgroup analyses, including age, sex, ISS stage, response after ASCT, and prior induction therapy.
All studies contributed to the positive results of the meta-analysis. Heterogeneity tests showed significant differences across trials, mainly because of a difference in the magnitude of treatment effect, Dr McCarthy said.
The mean treatment duration of maintenance was 25 to 30 months in the lenalidomide group and 13 to 20 months in controls. About one-third to more than half of the patients received therapy for 3 or more years, Dr McCarthy said.
Lenalidomide led to an increased risk in the cumulative incidence of hematologic and solid tumor second primary malignancies. However, Dr McCarthy said the OS benefit of lenalidomide maintenance outweighs the risk of developing second primary malignancy.
“This large meta-analysis demonstrates that lenalidomide maintenance significantly prolonged OS post-ASCT, including in patients who achieved CR, demonstrating benefit in patients in all response categories,” Dr McCarthy said.
“Lenalidomide maintenance after ASCT can be considered a standard of care for newly diagnosed multiple myeloma patients. However, we have more to learn. Understanding the role of minimal residual disease detection and immune reconstitution after transplant should allow us to further improve OS. Critically, developing early endpoints as surrogates for long-term outcome and OS is important for the future. Otherwise, trials may continue for 10 years or longer.”
Dr McCarthy presented these findings as abstract 8001. ![]()

the ASCO Annual Meeting
© ASCO/Todd Buchanan
CHICAGO—Lenalidomide maintenance after high-dose melphalan and autologous stem cell transplant (ASCT) should be considered the standard of care in
newly diagnosed multiple myeloma (MM) patients, according to a meta-analysis presented at the 2016 ASCO Annual Meeting.
Lenalidomide maintenance increased overall survival (OS), with a 26% reduction in the risk of death, representing an estimated 2.5-year increase in median survival.
Several studies have demonstrated that lenalidomide maintenance post-ASCT reduces the risk of disease progression or death in patients with MM by about 50%.
However, these studies were not powered for OS, said Philip McCarthy, MD, of Roswell Park Cancer Institute in Buffalo, New York.
With this in mind, Dr McCarthy and his colleagues conducted a meta-analysis to assess the effect of post-ASCT lenalidomide maintenance on OS using a pooled analysis of primary source patient data. A search revealed 17 randomized, controlled trials using lenalidomide post-ASCT.
Three trials met pre-specified inclusion criteria and had sufficient OS events to test a treatment effect. The studies intended for lenalidomide maintenance to be given until progression.
In these trials, 1209 MM patients, with a median age of 58, were randomized from 2005 to 2009 to receive lenalidomide (605 patients) at 10 mg/day on days 1-21/28 or days 1-28/28. The remaining 604 patients served as controls. Baseline characteristics were generally balanced between the 2 groups.
With a median follow-up of 6.6 years, 491 patients (41%) had died.
After induction and single (82%) or tandem (18%) ASCT, 55% of patients achieved a complete response (CR) or very good partial response.
The median OS has not been reached in the lenalidomide arm but was 86 months for the control arm.
“There is a 26% reduction in the risk of death, representing an estimated 2.5-year increase in median survival,” Dr McCarthy said.
The OS benefit favoring lenalidomide was generally consistent across the majority of subgroup analyses, including age, sex, ISS stage, response after ASCT, and prior induction therapy.
All studies contributed to the positive results of the meta-analysis. Heterogeneity tests showed significant differences across trials, mainly because of a difference in the magnitude of treatment effect, Dr McCarthy said.
The mean treatment duration of maintenance was 25 to 30 months in the lenalidomide group and 13 to 20 months in controls. About one-third to more than half of the patients received therapy for 3 or more years, Dr McCarthy said.
Lenalidomide led to an increased risk in the cumulative incidence of hematologic and solid tumor second primary malignancies. However, Dr McCarthy said the OS benefit of lenalidomide maintenance outweighs the risk of developing second primary malignancy.
“This large meta-analysis demonstrates that lenalidomide maintenance significantly prolonged OS post-ASCT, including in patients who achieved CR, demonstrating benefit in patients in all response categories,” Dr McCarthy said.
“Lenalidomide maintenance after ASCT can be considered a standard of care for newly diagnosed multiple myeloma patients. However, we have more to learn. Understanding the role of minimal residual disease detection and immune reconstitution after transplant should allow us to further improve OS. Critically, developing early endpoints as surrogates for long-term outcome and OS is important for the future. Otherwise, trials may continue for 10 years or longer.”
Dr McCarthy presented these findings as abstract 8001. ![]()

the ASCO Annual Meeting
© ASCO/Todd Buchanan
CHICAGO—Lenalidomide maintenance after high-dose melphalan and autologous stem cell transplant (ASCT) should be considered the standard of care in
newly diagnosed multiple myeloma (MM) patients, according to a meta-analysis presented at the 2016 ASCO Annual Meeting.
Lenalidomide maintenance increased overall survival (OS), with a 26% reduction in the risk of death, representing an estimated 2.5-year increase in median survival.
Several studies have demonstrated that lenalidomide maintenance post-ASCT reduces the risk of disease progression or death in patients with MM by about 50%.
However, these studies were not powered for OS, said Philip McCarthy, MD, of Roswell Park Cancer Institute in Buffalo, New York.
With this in mind, Dr McCarthy and his colleagues conducted a meta-analysis to assess the effect of post-ASCT lenalidomide maintenance on OS using a pooled analysis of primary source patient data. A search revealed 17 randomized, controlled trials using lenalidomide post-ASCT.
Three trials met pre-specified inclusion criteria and had sufficient OS events to test a treatment effect. The studies intended for lenalidomide maintenance to be given until progression.
In these trials, 1209 MM patients, with a median age of 58, were randomized from 2005 to 2009 to receive lenalidomide (605 patients) at 10 mg/day on days 1-21/28 or days 1-28/28. The remaining 604 patients served as controls. Baseline characteristics were generally balanced between the 2 groups.
With a median follow-up of 6.6 years, 491 patients (41%) had died.
After induction and single (82%) or tandem (18%) ASCT, 55% of patients achieved a complete response (CR) or very good partial response.
The median OS has not been reached in the lenalidomide arm but was 86 months for the control arm.
“There is a 26% reduction in the risk of death, representing an estimated 2.5-year increase in median survival,” Dr McCarthy said.
The OS benefit favoring lenalidomide was generally consistent across the majority of subgroup analyses, including age, sex, ISS stage, response after ASCT, and prior induction therapy.
All studies contributed to the positive results of the meta-analysis. Heterogeneity tests showed significant differences across trials, mainly because of a difference in the magnitude of treatment effect, Dr McCarthy said.
The mean treatment duration of maintenance was 25 to 30 months in the lenalidomide group and 13 to 20 months in controls. About one-third to more than half of the patients received therapy for 3 or more years, Dr McCarthy said.
Lenalidomide led to an increased risk in the cumulative incidence of hematologic and solid tumor second primary malignancies. However, Dr McCarthy said the OS benefit of lenalidomide maintenance outweighs the risk of developing second primary malignancy.
“This large meta-analysis demonstrates that lenalidomide maintenance significantly prolonged OS post-ASCT, including in patients who achieved CR, demonstrating benefit in patients in all response categories,” Dr McCarthy said.
“Lenalidomide maintenance after ASCT can be considered a standard of care for newly diagnosed multiple myeloma patients. However, we have more to learn. Understanding the role of minimal residual disease detection and immune reconstitution after transplant should allow us to further improve OS. Critically, developing early endpoints as surrogates for long-term outcome and OS is important for the future. Otherwise, trials may continue for 10 years or longer.”
Dr McCarthy presented these findings as abstract 8001. ![]()
Better pembrolizumab outcomes with increasing PD-L1 in NSCLC
CHICAGO – The PD-1 inhibitor, pembrolizumab, is superior to docetaxel in prolonging overall and progression-free survival in patients with non–small cell lung cancer (NSCLC), according to the results of the phase III KEYNOTE-010 trial.
Furthermore, increasing PD-L1 expression was associated with more favorable outcomes with pembrolizumab while level of PD-L1 expression had no effect on outcomes in docetaxel.
Results of the KEYNOTE-010 study were published earlier this year in the Lancet, but the results reported at the annual meeting of the American Society of Clinical Oncology further categorized study participants by tumor proportion score, a measure of viable tumor cells showing partial or complete membrane staining for PD-L1.
This finding verifies PD-L1 as a “predictive biomarker for ‘pembro’ in NSCLC,” lead study author Dr. Paul Baas of the Netherlands Cancer Institute, Amsterdam, said.
Out of 690 patients receiving pembrolizumab, 184 had a tumor proportion score (TPS) of 75% or more. For those 184 patients, median overall survival was 16.6 months and median progression-free survival was 6.2 months. The overall response rate was 33.7% for patients receiving pembrolizumab with TPS greater than 75%.
These survival and response rates were significantly higher, compared with the 8.2 month median overall survival, 4.0 month progression-free survival, and 7% overall response rate observed in 100 patients with the same TPS score who instead received docetaxel (overall survival: hazard ratio, 0.51; 95% confidence interval, 0.36-0.73; P less than .0001; progression-free survival: HR, 0.52; 95% CI, 0.38-0.69; P less than .0001; overall response rate: P less than .0001).
For the 106 patients receiving pembrolizumab who had a TPS between 50% and 74%, median OS was 15.8 months, PFS was 4.3 months, and the ORR was 22.6%. Median overall survival was significantly higher for pembrolizumab than docetaxel which was 8.2 months (HR, 0.58; 95% CI, 0.36-0.95; P = .01). ORR was also significantly higher for pembrolizumab than for docetaxel, which was 9.6% (P = .01).
The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and receiving honoraria and financial compensation from multiple companies including Merck Sharp & Dohme.
CHICAGO – The PD-1 inhibitor, pembrolizumab, is superior to docetaxel in prolonging overall and progression-free survival in patients with non–small cell lung cancer (NSCLC), according to the results of the phase III KEYNOTE-010 trial.
Furthermore, increasing PD-L1 expression was associated with more favorable outcomes with pembrolizumab while level of PD-L1 expression had no effect on outcomes in docetaxel.
Results of the KEYNOTE-010 study were published earlier this year in the Lancet, but the results reported at the annual meeting of the American Society of Clinical Oncology further categorized study participants by tumor proportion score, a measure of viable tumor cells showing partial or complete membrane staining for PD-L1.
This finding verifies PD-L1 as a “predictive biomarker for ‘pembro’ in NSCLC,” lead study author Dr. Paul Baas of the Netherlands Cancer Institute, Amsterdam, said.
Out of 690 patients receiving pembrolizumab, 184 had a tumor proportion score (TPS) of 75% or more. For those 184 patients, median overall survival was 16.6 months and median progression-free survival was 6.2 months. The overall response rate was 33.7% for patients receiving pembrolizumab with TPS greater than 75%.
These survival and response rates were significantly higher, compared with the 8.2 month median overall survival, 4.0 month progression-free survival, and 7% overall response rate observed in 100 patients with the same TPS score who instead received docetaxel (overall survival: hazard ratio, 0.51; 95% confidence interval, 0.36-0.73; P less than .0001; progression-free survival: HR, 0.52; 95% CI, 0.38-0.69; P less than .0001; overall response rate: P less than .0001).
For the 106 patients receiving pembrolizumab who had a TPS between 50% and 74%, median OS was 15.8 months, PFS was 4.3 months, and the ORR was 22.6%. Median overall survival was significantly higher for pembrolizumab than docetaxel which was 8.2 months (HR, 0.58; 95% CI, 0.36-0.95; P = .01). ORR was also significantly higher for pembrolizumab than for docetaxel, which was 9.6% (P = .01).
The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and receiving honoraria and financial compensation from multiple companies including Merck Sharp & Dohme.
CHICAGO – The PD-1 inhibitor, pembrolizumab, is superior to docetaxel in prolonging overall and progression-free survival in patients with non–small cell lung cancer (NSCLC), according to the results of the phase III KEYNOTE-010 trial.
Furthermore, increasing PD-L1 expression was associated with more favorable outcomes with pembrolizumab while level of PD-L1 expression had no effect on outcomes in docetaxel.
Results of the KEYNOTE-010 study were published earlier this year in the Lancet, but the results reported at the annual meeting of the American Society of Clinical Oncology further categorized study participants by tumor proportion score, a measure of viable tumor cells showing partial or complete membrane staining for PD-L1.
This finding verifies PD-L1 as a “predictive biomarker for ‘pembro’ in NSCLC,” lead study author Dr. Paul Baas of the Netherlands Cancer Institute, Amsterdam, said.
Out of 690 patients receiving pembrolizumab, 184 had a tumor proportion score (TPS) of 75% or more. For those 184 patients, median overall survival was 16.6 months and median progression-free survival was 6.2 months. The overall response rate was 33.7% for patients receiving pembrolizumab with TPS greater than 75%.
These survival and response rates were significantly higher, compared with the 8.2 month median overall survival, 4.0 month progression-free survival, and 7% overall response rate observed in 100 patients with the same TPS score who instead received docetaxel (overall survival: hazard ratio, 0.51; 95% confidence interval, 0.36-0.73; P less than .0001; progression-free survival: HR, 0.52; 95% CI, 0.38-0.69; P less than .0001; overall response rate: P less than .0001).
For the 106 patients receiving pembrolizumab who had a TPS between 50% and 74%, median OS was 15.8 months, PFS was 4.3 months, and the ORR was 22.6%. Median overall survival was significantly higher for pembrolizumab than docetaxel which was 8.2 months (HR, 0.58; 95% CI, 0.36-0.95; P = .01). ORR was also significantly higher for pembrolizumab than for docetaxel, which was 9.6% (P = .01).
The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and receiving honoraria and financial compensation from multiple companies including Merck Sharp & Dohme.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Pembrolizumab is superior to docetaxel in patients with non–small cell lung cancer especially at higher TPS. PD-L1 may be a predictive biomarker for pembrolizumab.
Major finding: For patients with tumor proportion scores of 75% or higher, median overall survival for pembrolizumab as single agent therapy was 16.6 months and only 8.2 months for single agent docetaxel (hazard ratio, .51; 95% confidence interval, .36-.73; P less than .0001).
Data source: The phase III KEYNOTE-010 trial of 1,034 patients with NSCLC.
Disclosures: The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and having received honoraria and financial compensation from multiple companies, including Merck Sharp & Dohme.
MITO8: Re-treat with platinum-based chemo for recurrent OC
CHICAGO – Artificially prolonging the platinum-free interval by introducing a non–platinum-based chemotherapy before re-treatment with platinum-based chemotherapy in patients with partially platinum-sensitive recurrent ovarian cancer did not improve efficacy, and actually worsened the outcomes in patients in the randomized, open-label, phase III Multicentre Italian Trials in Ovarian Cancer 8 (MITO8) trial.
Based on the findings, immediate re-treatment with platinum-based chemotherapy remains the standard treatment strategy in such patients, Dr. Sandro Pignata reported at the annual meeting of the American Society of Clinical Oncology.
The idea that a non-platinum therapy might artificially extend the platinum-free interval, thereby improving outcomes in ovarian cancer patients who experience recurrence between 6 and 12 months after responding to platinum-based therapy, was first proposed more than 20 years ago. Despite a lack of prospective data with respect to this non–platinum-therapy approach, the idea has been used successfully to market non-platinum chemotherapy agents such as paclitaxel, topotecan, pegylated liposomal doxorubicin (PLD), and more recently, PLD-trabectedin for use before reinitiating platinum chemotherapy, explained Dr. Pignata of S.C. Oncologia Medica Ginecologica, Istituto Nazionale Tumori, Napoli.
In fact, a 2007 retrospective analysis (SOCRATES) showed that 35% of patients with recurrence at 6-12 months after platinum-based therapy were treated with a non-platinum single agent before re-treatment with a platinum-based therapy, and that the approach has been used as the control arm in multiple phase III trials that included patients with a platinum-free interval of 6-12 months. The findings indicate that the unproven hypothesis has been adapted into clinical practice, but the investigators were unable to demonstrate an advantage with the approach and concluded that further investigation was warranted.
MITO8, a multi-organization collaborative effort, was designed to test whether artificial prolongation of the platinum-free interval with a single-agent non-platinum treatment would improve overall survival in recurrent partially platinum-sensitive ovarian cancer patients.
In 107 such patients randomized to the experimental arm – receiving a non–platinum-based chemotherapy first to prolong the platinum-free interval, followed by platinum-based chemotherapy after progression – the median time from previous platinum to randomization was 8 months, and from randomization to platinum, 7.8 months. Median overall survival, the primary endpoint of the study, was 21.8 months.
In 108 patients randomized to immediate standard therapy with platinum-based chemotherapy followed by non-platinum therapy, median time from previous platinum to randomization was 8 months, and overall survival was 24.5 months (hazard ratio favoring platinum chemotherapy, 1.38), Dr. Pignata said, noting that the difference in overall survival approached but did not reach statistical significance.
A secondary endpoint of progression-free survival after the second treatment was 12.8 months in the experimental arm, vs. 16.4 months in the platinum-based chemotherapy arm (adjusted HR, 1.41). This difference was statistically significant.
No differences with respect to safety or toxicity were seen between the groups.
MITO8 subjects were women with ovarian cancer recurring between 6 and 12 months after previous platinum-based chemotherapy. The patients had received no more than two prior lines of chemotherapy, had good performance status, and had normal organ function.
Non–platinum-based therapy used in the study included pegylated liposomal doxorubicin, except during a period of the study when the agent was in short supply and other single agents were used, including gemcitabine and topotecan. Most patients, however, received PLD.
Platinum-based therapy was carboplatin plus paclitaxel or, in patients with neurotoxicity at baseline, carboplatin plus gemcitabine.
The shortage of PLD was just one of numerous challenges encountered during the course of the beleaguered study. Enrollment began in 2009, but was temporarily suspended in 2011 because of the PLD shortage. In 2012, the amendment to allow other non–platinum-based chemotherapies to be used was approved at most sites and enrollment resumed. However, accrual slowed and enrollment was closed in 2015. The study was stopped early, and the final analysis was conducted in March 2016 when events plateaued. The investigators, in conjunction with the independent data monitoring committee, determined that the results “still had to be presented early because they still may affect clinical practice.”
Dr. Pignata said that the hypothesis “was based on the fact that there were more responses in the patients treated with a platinum after a non-platinum, but this was not the case ... Responses were more frequent in the patients who received platinum first and then non-platinum later,” he said. The response rates for the experimental vs. standard approaches, respectively, were 43% vs. 56% among RECIST responders, and 70% vs. 75% among CA125 responders.
The findings of MITO8, one of the first clinical trials in ovarian cancer to evaluate two different sequences of chemotherapy, send “a strong message that even in the presence of a very nice hypothesis, we have to await the results of prospective clinical trials before changing clinical practice,” he said.
Dr. Maurie Markman of Cancer Treatment Centers of America in Boca Raton, Fla., the invited discussant, praised the work of Dr. Pignata and his colleagues, calling their study “elegant,” and “an extraordinary effort.”
“In this unbelievably important analysis, the hypothesis was flat-out wrong,” he said.
The study is an example of “a simple, provocative, and potentially highly clinically relevant hypothesis that should be able to be quickly tested in either a cooperative group setting, in multiple institutions interested in gynecologic malignancies ... when or very shortly after it is initially proposed,” he said.
Waiting 20 years is unacceptable, he said, adding: “This is tragic, this length of time to get an answer. And I must ask ... how many ovarian cancer patients over this many-year period have received second-line chemotherapy based on this conceptual approach, and may have actually been harmed?”
To prevent such outcomes in the future, Dr. Markman proposed heavier reliance on “nonrandomized population-based studies including real-world patients managed by a variety of approaches with securely privacy-protected data included within a public database,” to test noninvestigational approaches to disease management.
“And for this database, I would propose ASCO’s CancerLinq,” he said.
Dr. Pignata reported receiving honoraria from, and serving as a consultant or adviser for AstraZeneca, PharmaMar, and Roche, and receiving research funding from Roche. Dr. Markman is a consultant or advisor for Amgen, Celgene, CritiTech, and Pfizer; is on the speakers bureau for AstraZeneca and Genentech/Roche, and has provided expert testimony for several entities
CHICAGO – Artificially prolonging the platinum-free interval by introducing a non–platinum-based chemotherapy before re-treatment with platinum-based chemotherapy in patients with partially platinum-sensitive recurrent ovarian cancer did not improve efficacy, and actually worsened the outcomes in patients in the randomized, open-label, phase III Multicentre Italian Trials in Ovarian Cancer 8 (MITO8) trial.
Based on the findings, immediate re-treatment with platinum-based chemotherapy remains the standard treatment strategy in such patients, Dr. Sandro Pignata reported at the annual meeting of the American Society of Clinical Oncology.
The idea that a non-platinum therapy might artificially extend the platinum-free interval, thereby improving outcomes in ovarian cancer patients who experience recurrence between 6 and 12 months after responding to platinum-based therapy, was first proposed more than 20 years ago. Despite a lack of prospective data with respect to this non–platinum-therapy approach, the idea has been used successfully to market non-platinum chemotherapy agents such as paclitaxel, topotecan, pegylated liposomal doxorubicin (PLD), and more recently, PLD-trabectedin for use before reinitiating platinum chemotherapy, explained Dr. Pignata of S.C. Oncologia Medica Ginecologica, Istituto Nazionale Tumori, Napoli.
In fact, a 2007 retrospective analysis (SOCRATES) showed that 35% of patients with recurrence at 6-12 months after platinum-based therapy were treated with a non-platinum single agent before re-treatment with a platinum-based therapy, and that the approach has been used as the control arm in multiple phase III trials that included patients with a platinum-free interval of 6-12 months. The findings indicate that the unproven hypothesis has been adapted into clinical practice, but the investigators were unable to demonstrate an advantage with the approach and concluded that further investigation was warranted.
MITO8, a multi-organization collaborative effort, was designed to test whether artificial prolongation of the platinum-free interval with a single-agent non-platinum treatment would improve overall survival in recurrent partially platinum-sensitive ovarian cancer patients.
In 107 such patients randomized to the experimental arm – receiving a non–platinum-based chemotherapy first to prolong the platinum-free interval, followed by platinum-based chemotherapy after progression – the median time from previous platinum to randomization was 8 months, and from randomization to platinum, 7.8 months. Median overall survival, the primary endpoint of the study, was 21.8 months.
In 108 patients randomized to immediate standard therapy with platinum-based chemotherapy followed by non-platinum therapy, median time from previous platinum to randomization was 8 months, and overall survival was 24.5 months (hazard ratio favoring platinum chemotherapy, 1.38), Dr. Pignata said, noting that the difference in overall survival approached but did not reach statistical significance.
A secondary endpoint of progression-free survival after the second treatment was 12.8 months in the experimental arm, vs. 16.4 months in the platinum-based chemotherapy arm (adjusted HR, 1.41). This difference was statistically significant.
No differences with respect to safety or toxicity were seen between the groups.
MITO8 subjects were women with ovarian cancer recurring between 6 and 12 months after previous platinum-based chemotherapy. The patients had received no more than two prior lines of chemotherapy, had good performance status, and had normal organ function.
Non–platinum-based therapy used in the study included pegylated liposomal doxorubicin, except during a period of the study when the agent was in short supply and other single agents were used, including gemcitabine and topotecan. Most patients, however, received PLD.
Platinum-based therapy was carboplatin plus paclitaxel or, in patients with neurotoxicity at baseline, carboplatin plus gemcitabine.
The shortage of PLD was just one of numerous challenges encountered during the course of the beleaguered study. Enrollment began in 2009, but was temporarily suspended in 2011 because of the PLD shortage. In 2012, the amendment to allow other non–platinum-based chemotherapies to be used was approved at most sites and enrollment resumed. However, accrual slowed and enrollment was closed in 2015. The study was stopped early, and the final analysis was conducted in March 2016 when events plateaued. The investigators, in conjunction with the independent data monitoring committee, determined that the results “still had to be presented early because they still may affect clinical practice.”
Dr. Pignata said that the hypothesis “was based on the fact that there were more responses in the patients treated with a platinum after a non-platinum, but this was not the case ... Responses were more frequent in the patients who received platinum first and then non-platinum later,” he said. The response rates for the experimental vs. standard approaches, respectively, were 43% vs. 56% among RECIST responders, and 70% vs. 75% among CA125 responders.
The findings of MITO8, one of the first clinical trials in ovarian cancer to evaluate two different sequences of chemotherapy, send “a strong message that even in the presence of a very nice hypothesis, we have to await the results of prospective clinical trials before changing clinical practice,” he said.
Dr. Maurie Markman of Cancer Treatment Centers of America in Boca Raton, Fla., the invited discussant, praised the work of Dr. Pignata and his colleagues, calling their study “elegant,” and “an extraordinary effort.”
“In this unbelievably important analysis, the hypothesis was flat-out wrong,” he said.
The study is an example of “a simple, provocative, and potentially highly clinically relevant hypothesis that should be able to be quickly tested in either a cooperative group setting, in multiple institutions interested in gynecologic malignancies ... when or very shortly after it is initially proposed,” he said.
Waiting 20 years is unacceptable, he said, adding: “This is tragic, this length of time to get an answer. And I must ask ... how many ovarian cancer patients over this many-year period have received second-line chemotherapy based on this conceptual approach, and may have actually been harmed?”
To prevent such outcomes in the future, Dr. Markman proposed heavier reliance on “nonrandomized population-based studies including real-world patients managed by a variety of approaches with securely privacy-protected data included within a public database,” to test noninvestigational approaches to disease management.
“And for this database, I would propose ASCO’s CancerLinq,” he said.
Dr. Pignata reported receiving honoraria from, and serving as a consultant or adviser for AstraZeneca, PharmaMar, and Roche, and receiving research funding from Roche. Dr. Markman is a consultant or advisor for Amgen, Celgene, CritiTech, and Pfizer; is on the speakers bureau for AstraZeneca and Genentech/Roche, and has provided expert testimony for several entities
CHICAGO – Artificially prolonging the platinum-free interval by introducing a non–platinum-based chemotherapy before re-treatment with platinum-based chemotherapy in patients with partially platinum-sensitive recurrent ovarian cancer did not improve efficacy, and actually worsened the outcomes in patients in the randomized, open-label, phase III Multicentre Italian Trials in Ovarian Cancer 8 (MITO8) trial.
Based on the findings, immediate re-treatment with platinum-based chemotherapy remains the standard treatment strategy in such patients, Dr. Sandro Pignata reported at the annual meeting of the American Society of Clinical Oncology.
The idea that a non-platinum therapy might artificially extend the platinum-free interval, thereby improving outcomes in ovarian cancer patients who experience recurrence between 6 and 12 months after responding to platinum-based therapy, was first proposed more than 20 years ago. Despite a lack of prospective data with respect to this non–platinum-therapy approach, the idea has been used successfully to market non-platinum chemotherapy agents such as paclitaxel, topotecan, pegylated liposomal doxorubicin (PLD), and more recently, PLD-trabectedin for use before reinitiating platinum chemotherapy, explained Dr. Pignata of S.C. Oncologia Medica Ginecologica, Istituto Nazionale Tumori, Napoli.
In fact, a 2007 retrospective analysis (SOCRATES) showed that 35% of patients with recurrence at 6-12 months after platinum-based therapy were treated with a non-platinum single agent before re-treatment with a platinum-based therapy, and that the approach has been used as the control arm in multiple phase III trials that included patients with a platinum-free interval of 6-12 months. The findings indicate that the unproven hypothesis has been adapted into clinical practice, but the investigators were unable to demonstrate an advantage with the approach and concluded that further investigation was warranted.
MITO8, a multi-organization collaborative effort, was designed to test whether artificial prolongation of the platinum-free interval with a single-agent non-platinum treatment would improve overall survival in recurrent partially platinum-sensitive ovarian cancer patients.
In 107 such patients randomized to the experimental arm – receiving a non–platinum-based chemotherapy first to prolong the platinum-free interval, followed by platinum-based chemotherapy after progression – the median time from previous platinum to randomization was 8 months, and from randomization to platinum, 7.8 months. Median overall survival, the primary endpoint of the study, was 21.8 months.
In 108 patients randomized to immediate standard therapy with platinum-based chemotherapy followed by non-platinum therapy, median time from previous platinum to randomization was 8 months, and overall survival was 24.5 months (hazard ratio favoring platinum chemotherapy, 1.38), Dr. Pignata said, noting that the difference in overall survival approached but did not reach statistical significance.
A secondary endpoint of progression-free survival after the second treatment was 12.8 months in the experimental arm, vs. 16.4 months in the platinum-based chemotherapy arm (adjusted HR, 1.41). This difference was statistically significant.
No differences with respect to safety or toxicity were seen between the groups.
MITO8 subjects were women with ovarian cancer recurring between 6 and 12 months after previous platinum-based chemotherapy. The patients had received no more than two prior lines of chemotherapy, had good performance status, and had normal organ function.
Non–platinum-based therapy used in the study included pegylated liposomal doxorubicin, except during a period of the study when the agent was in short supply and other single agents were used, including gemcitabine and topotecan. Most patients, however, received PLD.
Platinum-based therapy was carboplatin plus paclitaxel or, in patients with neurotoxicity at baseline, carboplatin plus gemcitabine.
The shortage of PLD was just one of numerous challenges encountered during the course of the beleaguered study. Enrollment began in 2009, but was temporarily suspended in 2011 because of the PLD shortage. In 2012, the amendment to allow other non–platinum-based chemotherapies to be used was approved at most sites and enrollment resumed. However, accrual slowed and enrollment was closed in 2015. The study was stopped early, and the final analysis was conducted in March 2016 when events plateaued. The investigators, in conjunction with the independent data monitoring committee, determined that the results “still had to be presented early because they still may affect clinical practice.”
Dr. Pignata said that the hypothesis “was based on the fact that there were more responses in the patients treated with a platinum after a non-platinum, but this was not the case ... Responses were more frequent in the patients who received platinum first and then non-platinum later,” he said. The response rates for the experimental vs. standard approaches, respectively, were 43% vs. 56% among RECIST responders, and 70% vs. 75% among CA125 responders.
The findings of MITO8, one of the first clinical trials in ovarian cancer to evaluate two different sequences of chemotherapy, send “a strong message that even in the presence of a very nice hypothesis, we have to await the results of prospective clinical trials before changing clinical practice,” he said.
Dr. Maurie Markman of Cancer Treatment Centers of America in Boca Raton, Fla., the invited discussant, praised the work of Dr. Pignata and his colleagues, calling their study “elegant,” and “an extraordinary effort.”
“In this unbelievably important analysis, the hypothesis was flat-out wrong,” he said.
The study is an example of “a simple, provocative, and potentially highly clinically relevant hypothesis that should be able to be quickly tested in either a cooperative group setting, in multiple institutions interested in gynecologic malignancies ... when or very shortly after it is initially proposed,” he said.
Waiting 20 years is unacceptable, he said, adding: “This is tragic, this length of time to get an answer. And I must ask ... how many ovarian cancer patients over this many-year period have received second-line chemotherapy based on this conceptual approach, and may have actually been harmed?”
To prevent such outcomes in the future, Dr. Markman proposed heavier reliance on “nonrandomized population-based studies including real-world patients managed by a variety of approaches with securely privacy-protected data included within a public database,” to test noninvestigational approaches to disease management.
“And for this database, I would propose ASCO’s CancerLinq,” he said.
Dr. Pignata reported receiving honoraria from, and serving as a consultant or adviser for AstraZeneca, PharmaMar, and Roche, and receiving research funding from Roche. Dr. Markman is a consultant or advisor for Amgen, Celgene, CritiTech, and Pfizer; is on the speakers bureau for AstraZeneca and Genentech/Roche, and has provided expert testimony for several entities
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Prolonging the platinum-free interval by introducing a non–platinum-based chemotherapy before re-treatment with platinum-based chemotherapy for recurrent ovarian cancer actually worsened the outcomes in the MITO8 trial.
Major finding: The median overall survival was 21.8 vs. 24.5 months in the experimental and standard therapy groups, respectively.
Data source: The randomized, open-label, phase III MITO8 study of 215 patients.
Disclosures: Dr. Pignata reported receiving honoraria from, and serving as a consultant or adviser for AstraZeneca, PharmaMar, and Roche, and receiving research funding from Roche. Dr. Markman is a consultant or advisor for Amgen, Celgene, CritiTech, and Pfizer; is on the speakers bureau for AstraZeneca and Genentech/Roche, and has provided expert testimony for several entities.