User login
Skin of Color in Preclinical Medical Education: A Cross-Institutional Comparison and A Call to Action
A ccording to the US Census Bureau, more than half of all Americans are projected to belong to a minority group, defined as any group other than non-Hispanic White alone, by 2044. 1 Consequently, the United States rapidly is becoming a country in which the majority of citizens will have skin of color. Individuals with skin of color are of diverse ethnic backgrounds and include people of African, Latin American, Native American, Pacific Islander, and Asian descent, as well as interethnic backgrounds. 2 Throughout the country, dermatologists along with primary care practitioners may be confronted with certain cutaneous conditions that have varying disease presentations or processes in patients with skin of color. It also is important to note that racial categories are socially rather than biologically constructed, and the term skin of color includes a wide variety of diverse skin types. Nevertheless, the current literature thoroughly supports unique pathophysiologic differences in skin of color as well as variations in disease manifestation compared to White patients. 3-5 For example, the increased lability of melanosomes in skin of color patients, which increases their risk for postinflammatory hyperpigmentation, has been well documented. 5-7 There are various dermatologic conditions that also occur with higher frequency and manifest uniquely in people with darker, more pigmented skin, 7-9 and dermatologists, along with primary care physicians, should feel prepared to recognize and address them.
Extensive evidence also indicates that there are unique aspects to consider while managing certain skin diseases in patients with skin of color.8,10,11 Consequently, as noted on the Skin of Color Society (SOCS) website, “[a]n increase in the body of dermatological literature concerning skin of color as well as the advancement of both basic science and clinical investigational research is necessary to meet the needs of the expanding skin of color population.”2 In the meantime, current knowledge regarding cutaneous conditions that diversely or disproportionately affect skin of color should be actively disseminated to physicians in training. Although patients with skin of color should always have access to comprehensive care and knowledgeable practitioners, the current changes in national and regional demographics further underscore the need for a more thorough understanding of skin of color with regard to disease pathogenesis, diagnosis, and treatment.
Several studies have found that medical students in the United States are minimally exposed to dermatology in general compared to other clinical specialties,12-14 which can easily lead to the underrecognition of disorders that may uniquely or disproportionately affect individuals with pigmented skin. Recent data showed that medical schools typically required fewer than 10 hours of dermatology instruction,12 and on average, dermatologic training made up less than 1% of a medical student’s undergraduate medical education.13,15,16 Consequently, less than 40% of primary care residents felt that their medical school curriculum adequately prepared them to manage common skin conditions.14 Although not all physicians should be expected to fully grasp the complexities of skin of color and its diagnostic and therapeutic implications, both practicing and training dermatologists have acknowledged a lack of exposure to skin of color. In one study, approximately 47% of dermatologists and dermatology residents reported that their medical training (medical school and/or residency) was inadequate in training them on skin conditions in Black patients. Furthermore, many who felt their training was lacking in skin of color identified the need for greater exposure to Black patients and training materials.15 The absence of comprehensive medical education regarding skin of color ultimately can be a disadvantage for both practitioners and patients, resulting in poorer outcomes. Furthermore, underrepresentation of skin of color may persist beyond undergraduate and graduate medical education. There also is evidence to suggest that noninclusion of skin of color pervades foundational dermatologic educational resources, including commonly used textbooks as well as continuing medical education disseminated at national conferences and meetings.17 Taken together, these findings highlight the need for more diverse and representative exposure to skin of color throughout medical training, which begins with a diverse inclusive undergraduate medical education in dermatology.
The objective of this study was to determine if the preclinical dermatology curriculum at 3 US medical schools provided adequate representation of skin of color patients in their didactic presentation slides.
Methods
Participants—Three US medical schools, a blend of private and public medical schools located across different geographic boundaries, agreed to participate in the study. All 3 institutions were current members of the American Medical Association (AMA) Accelerating Change in Medical Education consortium, whose primary goal is to create the medical school of the future and transform physician training.18 All 32 member institutions of the AMA consortium were contacted to request their participation in the study. As part of the consortium, these institutions have vowed to collectively work to develop and share the best models for educational advancement to improve care for patients, populations, and communities18 and would expectedly provide a more racially and ethnically inclusive curriculum than an institution not accountable to a group dedicated to identifying the best ways to deliver care for increasingly diverse communities.
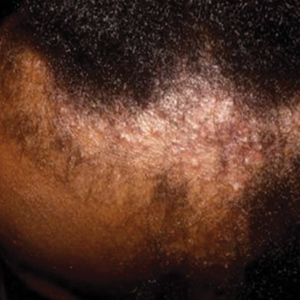
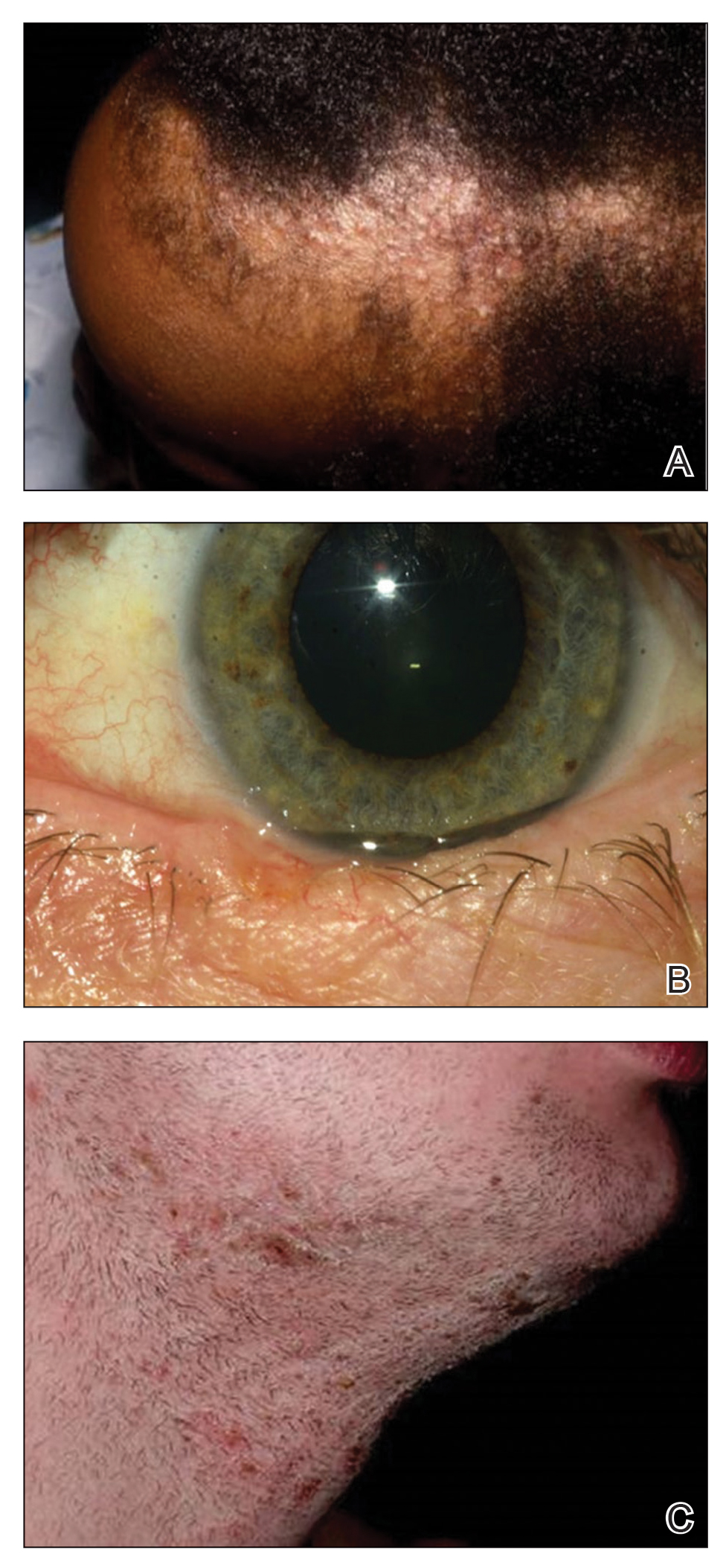
Data Collection—Lectures were included if they were presented during dermatology preclinical courses in the 2015 to 2016 academic year. An uninvolved third party removed the names and identities of instructors to preserve anonymity. Two independent coders from different institutions extracted the data—lecture title, total number of clinical and histologic images, and number of skin of color images—from each of the anonymized lectures using a standardized coding form. We documented differences in skin of color noted in lectures and the disease context for the discussed differences, such as variations in clinical presentation, disease process, epidemiology/risk, and treatment between different skin phenotypes or ethnic groups. Photographs in which the coders were unable to differentiate whether the patient had skin of color were designated as indeterminate or unclear. Photographs appearing to represent Fitzpatrick skin types IV, V, and VI19 were categorically designated as skin of color, and those appearing to represent Fitzpatrick skin types I and II were described as not skin of color; however, images appearing to represent Fitzpatrick skin type III often were classified as not skin of color or indeterminate and occasionally skin of color. The Figure shows examples of images classified as skin of color, indeterminate, and not skin of color. Photographs often were classified as indeterminate due to poor lighting, close-up view photographs, or highlighted pathology obscuring the surrounding skin. We excluded duplicate photographs and histologic images from the analyses.
We also reviewed 19 conditions previously highlighted by the SOCS as areas of importance to skin of color patients.20 The coders tracked how many of these conditions were noted in each lecture. Duplicate discussion of these conditions was not included in the analyses. Any discrepancies between coders were resolved through additional slide review and discussion. The final coded data with the agreed upon changes were used for statistical analyses. Recent national demographic data from the US Census Bureau in 2019 describe approximately 39.9% of the population as belonging to racial/ethnic groups other than non-Hispanic/Latinx White.21 Consequently, the standard for adequate representation for skin of color photographs was set at 35% for the purpose of this study.
Results
Across all 3 institutions included in the study, the proportion of the total number of clinical photographs showing skin of color was 16% (290/1812). Eight percent of the total photographs (145/1812) were noted to be indeterminate (Table). For institution 1, 23.6% of photographs (155/658) showed skin of color, and 12.6% (83/658) were indeterminate. For institution 2, 13.1% (76/578) showed skin of color and 7.8% (45/578) were indeterminate. For institution 3, 10.2% (59/576) showed skin of color and 3% (17/576) were indeterminate.
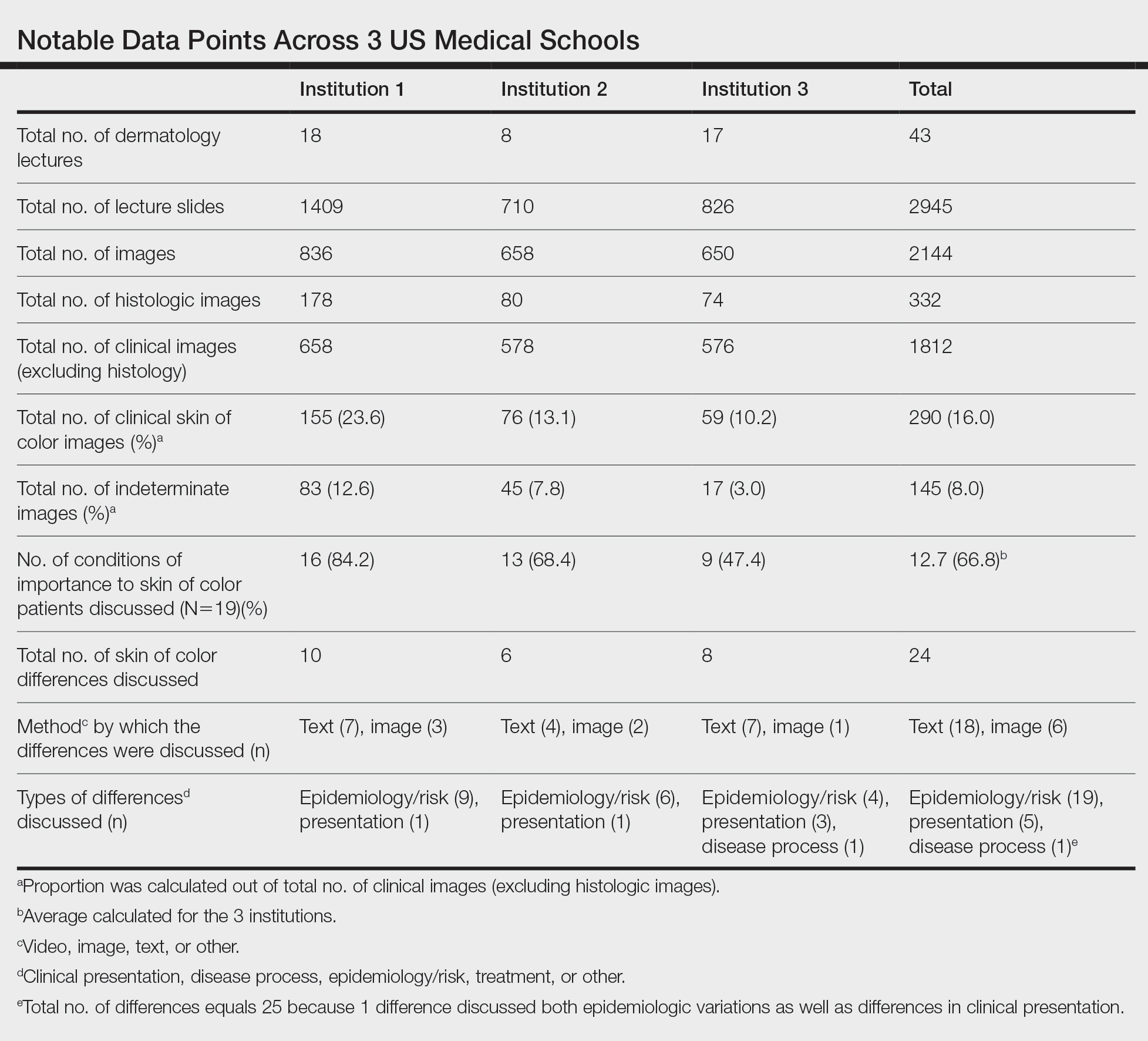
Institutions 1, 2, and 3 had 18, 8, and 17 total dermatology lectures, respectively. Of the 19 conditions designated as areas of importance to skin of color patients by the SOCS, 16 (84.2%) were discussed by institution 1, 11 (57.9%) by institution 2, and 9 (47.4%) by institution 3 (eTable 1). Institution 3 did not include photographs of skin of color patients in its acne, psoriasis, or cutaneous malignancy lectures. Institution 1 also did not include any skin of color patients in its malignancy lecture. Lectures that focused on pigmentary disorders, atopic dermatitis, infectious conditions, and benign cutaneous neoplasms were more likely to display photographs of skin of color patients; for example, lectures that discussed infectious conditions, such as superficial mycoses, herpes viruses, human papillomavirus, syphilis, and atypical mycobacterial infections, were consistently among those with higher proportions of photographs of skin of color patients.

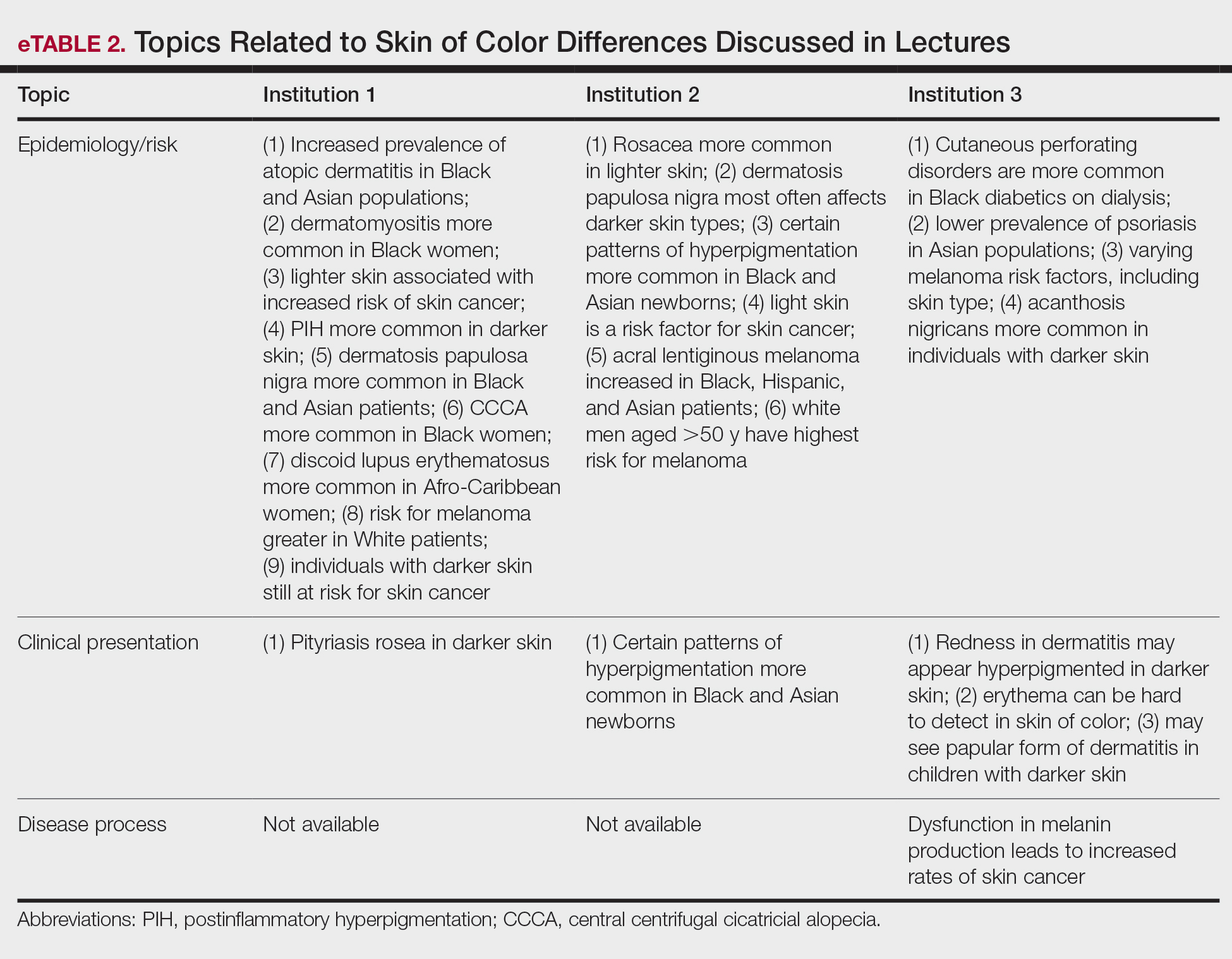
Throughout the entire preclinical dermatology course at all 3 institutions, of 2945 lecture slides, only 24 (0.8%) unique differences were noted between skin color and non–skin of color patients, with 10 total differences noted by institution 1, 6 by institution 2, and 8 by institution 3 (Table). The majority of these differences (19/24) were related to epidemiologic differences in prevalence among varying racial/ethnic groups, with only 5 instances highlighting differences in clinical presentation. There was only a single instance that elaborated on the underlying pathophysiologic mechanisms of the discussed difference. Of all 24 unique differences discussed, 8 were related to skin cancer, 3 were related to dermatitis, and 2 were related to the difference in manifestation of erythema in patients with darker skin (eTable 2).
Comment
The results of this study demonstrated that skin of color is underrepresented in the preclinical dermatology curriculum at these 3 institutions. Although only 16% of all included clinical photographs were of skin of color, individuals with skin of color will soon represent more than half of the total US population within the next 2 decades.1 To increase representation of skin of color patients, teaching faculty should consciously and deliberately include more photographs of skin of color patients for a wider variety of common conditions, including atopic dermatitis and psoriasis, in addition to those that tend to disparately affect skin of color patients, such as pseudofolliculitis barbae or melasma. Furthermore, they also can incorporate more detailed discussions about important differences seen in skin of color patients.
More Skin of Color Photographs in Psoriasis Lectures—At institution 3, there were no skin of color patients included in the psoriasis lecture, even though there is considerable data in the literature indicating notable differences in the clinical presentation, quality-of-life impact, and treatment of psoriasis in skin of color patients.11,22 There are multiple nuances in psoriasis manifestation in patients with skin of color, including less-conspicuous erythema in darker skin, higher degrees of dyspigmentation, and greater body surface area involvement. For Black patients with scalp psoriasis, the impact of hair texture, styling practices, and washing frequency are additional considerations that may impact disease severity and selection of topical therapy.11 The lack of inclusion of any skin of color patients in the psoriasis lecture at one institution further underscores the pressing need to prioritize communities of color in medical education.
More Skin of Color Photographs in Cutaneous Malignancy Lectures—Similarly, while a lecturer at institution 2 noted that acral lentiginous melanoma accounts for a considerable proportion of melanoma among skin of color patients,23 there was no mention of how melanoma generally is substantially more deadly in this population, potentially due to decreased awareness and inconsistent screening.24 Furthermore, at institutions 1 and 3, there were no photographs or discussion of skin of color patients during the cutaneous malignancy lectures. Evidence shows that more emphasis is needed for melanoma screening and awareness in skin of color populations to improve survival outcomes,24 and this begins with educating not only future dermatologists but all future physicians as well. The failure to include photographs of skin of color patients in discussions or lectures regarding cutaneous malignancies may serve to further perpetuate the harmful misperception that individuals with skin of color are unaffected by skin cancer.25,26
Analysis of Skin of Color Photographs in Infectious Disease Lectures—In addition, lectures discussing infectious etiologies were among those with the highest proportion of skin of color photographs. This relatively disproportionate representation of skin of color compared to the other lectures may contribute to the development of harmful stereotypes or the stigmatization of skin of color patients. Although skin of color should continue to be represented in similar lectures, teaching faculty should remain mindful of the potential unintended impact from lectures including relatively disproportionate amounts of skin of color, particularly when other lectures may have sparse to absent representation of skin of color.
More Photographs Available for Education—Overall, our findings may help to inform changes to preclinical dermatology medical education at other institutions to create more inclusive and representative curricula for skin of color patients. The ability of instructors to provide visual representation of various dermatologic conditions may be limited by the photographs available in certain textbooks with few examples of patients with skin of color; however, concerns regarding the lack of skin of color representation in dermatology training is not a novel discussion.17 Although it is the responsibility of all dermatologists to advocate for the inclusion of skin of color, many dermatologists of color have been leading the way in this movement for decades, publishing several textbooks to document various skin conditions in those with darker skin types and discuss unique considerations for patients with skin of color.27-29 Images from these textbooks can be utilized by programs to increase representation of skin of color in dermatology training. There also are multiple expanding online dermatologic databases, such as VisualDx, with an increasing focus on skin of color patients, some of which allow users to filter images by degree of skin pigmentation.30 Moreover, instructors also can work to diversify their curricula by highlighting more of the SOCS conditions of importance to skin of color patients, which have since been renamed and highlighted on the Patient Dermatology Education section of the SOCS website.20 These conditions, while not completely comprehensive, provide a useful starting point for medical educators to reevaluate for potential areas of improvement and inclusion.
There are several potential strategies that can be used to better represent skin of color in dermatologic preclinical medical education, including increasing awareness, especially among dermatology teaching faculty, of existing disparities in the representation of skin of color in the preclinical curricula. Additionally, all dermatology teaching materials could be reviewed at the department level prior to being disseminated to medical students to assess for instances in which skin of color could be prioritized for discussion or varying disease presentations in skin of color could be demonstrated. Finally, teaching faculty may consider photographing more clinical images of their skin of color patients to further develop a catalog of diverse images that can be used to teach students.
Study Limitations—Our study was unable to account for verbal discussion of skin of color not otherwise denoted or captured in lecture slides. Additional limitations include the utilization of Fitzpatrick skin types to describe and differentiate varying skin tones, as the Fitzpatrick scale originally was developed as a method to describe an individual’s response to UV exposure.19 The inability to further delineate the representation of darker skin types, such as those that may be classified as Fitzpatrick skin types V or VI,19 compared to those with lighter skin of color also was a limiting factor. This study was unable to assess for discussion of other common conditions affecting skin of color patients that were not listed as one of the priority conditions by SOCS. Photographs that were designated as indeterminate were difficult to elucidate as skin of color; however, it is possible that instructors may have verbally described these images as skin of color during lectures. Nonetheless, it may be beneficial for learners if teaching faculty were to clearly label instances where skin of color patients are shown or when notable differences are present.
Conclusion
Future studies would benefit from the inclusion of audio data from lectures, syllabi, and small group teaching materials from preclinical courses to more accurately assess representation of skin of color in dermatology training. Additionally, future studies also may expand to include images from lectures of overlapping clinical specialties, particularly infectious disease and rheumatology, to provide a broader assessment of skin of color exposure. Furthermore, repeat assessment may be beneficial to assess the longitudinal effectiveness of curricular changes at the institutions included in this study, comparing older lectures to more recent, updated lectures. This study also may be replicated at other medical schools to allow for wider comparison of curricula.
Acknowledgment—The authors wish to thank the institutions that offered and agreed to participate in this study with the hopes of improving medical education.
- Colby SL, Ortman JM. Projections of the size and composition of the US population: 2014 to 2060. United States Census Bureau website. Published March 2015. Accessed September 14, 2021. https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Learn more about SOCS. Skin of Color Society website. Accessed September 14, 2021. http://skinofcolorsociety.org/about-socs/
- Taylor SC. Skin of color: biology, structure, function, and implications for dermatologic disease. J Am Acad Dermatol. 2002;46(suppl 2):S41-S62.
- Berardesca E, Maibach H. Ethnic skin: overview of structure and function. J Am Acad Dermatol. 2003;48(suppl 6):S139-S142.
- Callender VD, Surin-Lord SS, Davis EC, et al. Postinflammatory hyperpigmentation. Am J Clin Dermatol. 2011;12:87-99.
- Davis EC, Callender VD. Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol. 2010;3:20-31.
- Grimes PE, Stockton T. Pigmentary disorders in blacks. Dermatol Clin. 1988;6:271-281.
- Halder RM, Nootheti PK. Ethnic skin disorders overview. J Am Acad Dermatol. 2003;48(suppl 6):S143-S148.
- Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
- Callender VD. Acne in ethnic skin: special considerations for therapy. Dermatol Ther. 2004;17:184-195.
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Ramsay DL, Mayer F. National survey of undergraduate dermatologic medical education. Arch Dermatol.1985;121:1529-1530.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59, viii.
- Knable A, Hood AF, Pearson TG. Undergraduate medical education in dermatology: report from the AAD Interdisciplinary Education Committee, Subcommittee on Undergraduate Medical Education. J Am Acad Dermatol. 1997;36:467-470.
- Ebede T, Papier A. Disparities in dermatology educational resources. J Am Acad Dermatol. 2006;55:687-690.
- Skochelak SE, Stack SJ. Creating the medical schools of the future. Acad Med. 2017;92:16-19.
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol. 1988;124:869-871.
- Skin of Color Society. Patient dermatology education. Accessed September 22, 2021. https://skinofcolorsociety.org/patient-dermatology-education
- QuickFacts: United States. US Census Bureau website. Updated July 1, 2019. Accessed September 14, 2021. https://www.census.gov/quickfacts/fact/table/US#
- Kaufman BP, Alexis AF. Psoriasis in skin of color: insights into the epidemiology, clinical presentation, genetics, quality-of-life impact, and treatment of psoriasis in non-white racial/ethnic groups. Am J Clin Dermatol. 2018;19:405-423.
- Bradford PT, Goldstein AM, McMaster ML, et al. Acral lentiginous melanoma: incidence and survival patterns in the United States, 1986-2005. Arch Dermatol. 2009;145:427-434.
- Dawes SM, Tsai S, Gittleman H, et al. Racial disparities in melanoma survival. J Am Acad Dermatol. 2016;75:983-991.
- Pipitone M, Robinson JK, Camara C, et al. Skin cancer awareness in suburban employees: a Hispanic perspective. J Am Acad Dermatol. 2002;47:118-123.
- Imahiyerobo-Ip J, Ip I, Jamal S, et al. Skin cancer awareness in communities of color. J Am Acad Dermatol. 2011;64:198-200.
- Taylor SSC, Serrano AMA, Kelly AP, et al, eds. Taylor and Kelly’s Dermatology for Skin of Color. 2nd ed. McGraw-Hill Education; 2016.
- Dadzie OE, Petit A, Alexis AF, eds. Ethnic Dermatology: Principles and Practice. Wiley-Blackwell; 2013.
- Jackson-Richards D, Pandya AG, eds. Dermatology Atlas for Skin of Color. Springer; 2014.
- VisualDx. New VisualDx feature: skin of color sort. Published October 14, 2020. Accessed September 22, 2021. https://www.visualdx.com/blog/new-visualdx-feature-skin-of-color-sort/
A ccording to the US Census Bureau, more than half of all Americans are projected to belong to a minority group, defined as any group other than non-Hispanic White alone, by 2044. 1 Consequently, the United States rapidly is becoming a country in which the majority of citizens will have skin of color. Individuals with skin of color are of diverse ethnic backgrounds and include people of African, Latin American, Native American, Pacific Islander, and Asian descent, as well as interethnic backgrounds. 2 Throughout the country, dermatologists along with primary care practitioners may be confronted with certain cutaneous conditions that have varying disease presentations or processes in patients with skin of color. It also is important to note that racial categories are socially rather than biologically constructed, and the term skin of color includes a wide variety of diverse skin types. Nevertheless, the current literature thoroughly supports unique pathophysiologic differences in skin of color as well as variations in disease manifestation compared to White patients. 3-5 For example, the increased lability of melanosomes in skin of color patients, which increases their risk for postinflammatory hyperpigmentation, has been well documented. 5-7 There are various dermatologic conditions that also occur with higher frequency and manifest uniquely in people with darker, more pigmented skin, 7-9 and dermatologists, along with primary care physicians, should feel prepared to recognize and address them.
Extensive evidence also indicates that there are unique aspects to consider while managing certain skin diseases in patients with skin of color.8,10,11 Consequently, as noted on the Skin of Color Society (SOCS) website, “[a]n increase in the body of dermatological literature concerning skin of color as well as the advancement of both basic science and clinical investigational research is necessary to meet the needs of the expanding skin of color population.”2 In the meantime, current knowledge regarding cutaneous conditions that diversely or disproportionately affect skin of color should be actively disseminated to physicians in training. Although patients with skin of color should always have access to comprehensive care and knowledgeable practitioners, the current changes in national and regional demographics further underscore the need for a more thorough understanding of skin of color with regard to disease pathogenesis, diagnosis, and treatment.
Several studies have found that medical students in the United States are minimally exposed to dermatology in general compared to other clinical specialties,12-14 which can easily lead to the underrecognition of disorders that may uniquely or disproportionately affect individuals with pigmented skin. Recent data showed that medical schools typically required fewer than 10 hours of dermatology instruction,12 and on average, dermatologic training made up less than 1% of a medical student’s undergraduate medical education.13,15,16 Consequently, less than 40% of primary care residents felt that their medical school curriculum adequately prepared them to manage common skin conditions.14 Although not all physicians should be expected to fully grasp the complexities of skin of color and its diagnostic and therapeutic implications, both practicing and training dermatologists have acknowledged a lack of exposure to skin of color. In one study, approximately 47% of dermatologists and dermatology residents reported that their medical training (medical school and/or residency) was inadequate in training them on skin conditions in Black patients. Furthermore, many who felt their training was lacking in skin of color identified the need for greater exposure to Black patients and training materials.15 The absence of comprehensive medical education regarding skin of color ultimately can be a disadvantage for both practitioners and patients, resulting in poorer outcomes. Furthermore, underrepresentation of skin of color may persist beyond undergraduate and graduate medical education. There also is evidence to suggest that noninclusion of skin of color pervades foundational dermatologic educational resources, including commonly used textbooks as well as continuing medical education disseminated at national conferences and meetings.17 Taken together, these findings highlight the need for more diverse and representative exposure to skin of color throughout medical training, which begins with a diverse inclusive undergraduate medical education in dermatology.
The objective of this study was to determine if the preclinical dermatology curriculum at 3 US medical schools provided adequate representation of skin of color patients in their didactic presentation slides.
Methods
Participants—Three US medical schools, a blend of private and public medical schools located across different geographic boundaries, agreed to participate in the study. All 3 institutions were current members of the American Medical Association (AMA) Accelerating Change in Medical Education consortium, whose primary goal is to create the medical school of the future and transform physician training.18 All 32 member institutions of the AMA consortium were contacted to request their participation in the study. As part of the consortium, these institutions have vowed to collectively work to develop and share the best models for educational advancement to improve care for patients, populations, and communities18 and would expectedly provide a more racially and ethnically inclusive curriculum than an institution not accountable to a group dedicated to identifying the best ways to deliver care for increasingly diverse communities.
Data Collection—Lectures were included if they were presented during dermatology preclinical courses in the 2015 to 2016 academic year. An uninvolved third party removed the names and identities of instructors to preserve anonymity. Two independent coders from different institutions extracted the data—lecture title, total number of clinical and histologic images, and number of skin of color images—from each of the anonymized lectures using a standardized coding form. We documented differences in skin of color noted in lectures and the disease context for the discussed differences, such as variations in clinical presentation, disease process, epidemiology/risk, and treatment between different skin phenotypes or ethnic groups. Photographs in which the coders were unable to differentiate whether the patient had skin of color were designated as indeterminate or unclear. Photographs appearing to represent Fitzpatrick skin types IV, V, and VI19 were categorically designated as skin of color, and those appearing to represent Fitzpatrick skin types I and II were described as not skin of color; however, images appearing to represent Fitzpatrick skin type III often were classified as not skin of color or indeterminate and occasionally skin of color. The Figure shows examples of images classified as skin of color, indeterminate, and not skin of color. Photographs often were classified as indeterminate due to poor lighting, close-up view photographs, or highlighted pathology obscuring the surrounding skin. We excluded duplicate photographs and histologic images from the analyses.

We also reviewed 19 conditions previously highlighted by the SOCS as areas of importance to skin of color patients.20 The coders tracked how many of these conditions were noted in each lecture. Duplicate discussion of these conditions was not included in the analyses. Any discrepancies between coders were resolved through additional slide review and discussion. The final coded data with the agreed upon changes were used for statistical analyses. Recent national demographic data from the US Census Bureau in 2019 describe approximately 39.9% of the population as belonging to racial/ethnic groups other than non-Hispanic/Latinx White.21 Consequently, the standard for adequate representation for skin of color photographs was set at 35% for the purpose of this study.
Results
Across all 3 institutions included in the study, the proportion of the total number of clinical photographs showing skin of color was 16% (290/1812). Eight percent of the total photographs (145/1812) were noted to be indeterminate (Table). For institution 1, 23.6% of photographs (155/658) showed skin of color, and 12.6% (83/658) were indeterminate. For institution 2, 13.1% (76/578) showed skin of color and 7.8% (45/578) were indeterminate. For institution 3, 10.2% (59/576) showed skin of color and 3% (17/576) were indeterminate.

Institutions 1, 2, and 3 had 18, 8, and 17 total dermatology lectures, respectively. Of the 19 conditions designated as areas of importance to skin of color patients by the SOCS, 16 (84.2%) were discussed by institution 1, 11 (57.9%) by institution 2, and 9 (47.4%) by institution 3 (eTable 1). Institution 3 did not include photographs of skin of color patients in its acne, psoriasis, or cutaneous malignancy lectures. Institution 1 also did not include any skin of color patients in its malignancy lecture. Lectures that focused on pigmentary disorders, atopic dermatitis, infectious conditions, and benign cutaneous neoplasms were more likely to display photographs of skin of color patients; for example, lectures that discussed infectious conditions, such as superficial mycoses, herpes viruses, human papillomavirus, syphilis, and atypical mycobacterial infections, were consistently among those with higher proportions of photographs of skin of color patients.

Throughout the entire preclinical dermatology course at all 3 institutions, of 2945 lecture slides, only 24 (0.8%) unique differences were noted between skin color and non–skin of color patients, with 10 total differences noted by institution 1, 6 by institution 2, and 8 by institution 3 (Table). The majority of these differences (19/24) were related to epidemiologic differences in prevalence among varying racial/ethnic groups, with only 5 instances highlighting differences in clinical presentation. There was only a single instance that elaborated on the underlying pathophysiologic mechanisms of the discussed difference. Of all 24 unique differences discussed, 8 were related to skin cancer, 3 were related to dermatitis, and 2 were related to the difference in manifestation of erythema in patients with darker skin (eTable 2).

Comment
The results of this study demonstrated that skin of color is underrepresented in the preclinical dermatology curriculum at these 3 institutions. Although only 16% of all included clinical photographs were of skin of color, individuals with skin of color will soon represent more than half of the total US population within the next 2 decades.1 To increase representation of skin of color patients, teaching faculty should consciously and deliberately include more photographs of skin of color patients for a wider variety of common conditions, including atopic dermatitis and psoriasis, in addition to those that tend to disparately affect skin of color patients, such as pseudofolliculitis barbae or melasma. Furthermore, they also can incorporate more detailed discussions about important differences seen in skin of color patients.
More Skin of Color Photographs in Psoriasis Lectures—At institution 3, there were no skin of color patients included in the psoriasis lecture, even though there is considerable data in the literature indicating notable differences in the clinical presentation, quality-of-life impact, and treatment of psoriasis in skin of color patients.11,22 There are multiple nuances in psoriasis manifestation in patients with skin of color, including less-conspicuous erythema in darker skin, higher degrees of dyspigmentation, and greater body surface area involvement. For Black patients with scalp psoriasis, the impact of hair texture, styling practices, and washing frequency are additional considerations that may impact disease severity and selection of topical therapy.11 The lack of inclusion of any skin of color patients in the psoriasis lecture at one institution further underscores the pressing need to prioritize communities of color in medical education.
More Skin of Color Photographs in Cutaneous Malignancy Lectures—Similarly, while a lecturer at institution 2 noted that acral lentiginous melanoma accounts for a considerable proportion of melanoma among skin of color patients,23 there was no mention of how melanoma generally is substantially more deadly in this population, potentially due to decreased awareness and inconsistent screening.24 Furthermore, at institutions 1 and 3, there were no photographs or discussion of skin of color patients during the cutaneous malignancy lectures. Evidence shows that more emphasis is needed for melanoma screening and awareness in skin of color populations to improve survival outcomes,24 and this begins with educating not only future dermatologists but all future physicians as well. The failure to include photographs of skin of color patients in discussions or lectures regarding cutaneous malignancies may serve to further perpetuate the harmful misperception that individuals with skin of color are unaffected by skin cancer.25,26
Analysis of Skin of Color Photographs in Infectious Disease Lectures—In addition, lectures discussing infectious etiologies were among those with the highest proportion of skin of color photographs. This relatively disproportionate representation of skin of color compared to the other lectures may contribute to the development of harmful stereotypes or the stigmatization of skin of color patients. Although skin of color should continue to be represented in similar lectures, teaching faculty should remain mindful of the potential unintended impact from lectures including relatively disproportionate amounts of skin of color, particularly when other lectures may have sparse to absent representation of skin of color.
More Photographs Available for Education—Overall, our findings may help to inform changes to preclinical dermatology medical education at other institutions to create more inclusive and representative curricula for skin of color patients. The ability of instructors to provide visual representation of various dermatologic conditions may be limited by the photographs available in certain textbooks with few examples of patients with skin of color; however, concerns regarding the lack of skin of color representation in dermatology training is not a novel discussion.17 Although it is the responsibility of all dermatologists to advocate for the inclusion of skin of color, many dermatologists of color have been leading the way in this movement for decades, publishing several textbooks to document various skin conditions in those with darker skin types and discuss unique considerations for patients with skin of color.27-29 Images from these textbooks can be utilized by programs to increase representation of skin of color in dermatology training. There also are multiple expanding online dermatologic databases, such as VisualDx, with an increasing focus on skin of color patients, some of which allow users to filter images by degree of skin pigmentation.30 Moreover, instructors also can work to diversify their curricula by highlighting more of the SOCS conditions of importance to skin of color patients, which have since been renamed and highlighted on the Patient Dermatology Education section of the SOCS website.20 These conditions, while not completely comprehensive, provide a useful starting point for medical educators to reevaluate for potential areas of improvement and inclusion.
There are several potential strategies that can be used to better represent skin of color in dermatologic preclinical medical education, including increasing awareness, especially among dermatology teaching faculty, of existing disparities in the representation of skin of color in the preclinical curricula. Additionally, all dermatology teaching materials could be reviewed at the department level prior to being disseminated to medical students to assess for instances in which skin of color could be prioritized for discussion or varying disease presentations in skin of color could be demonstrated. Finally, teaching faculty may consider photographing more clinical images of their skin of color patients to further develop a catalog of diverse images that can be used to teach students.
Study Limitations—Our study was unable to account for verbal discussion of skin of color not otherwise denoted or captured in lecture slides. Additional limitations include the utilization of Fitzpatrick skin types to describe and differentiate varying skin tones, as the Fitzpatrick scale originally was developed as a method to describe an individual’s response to UV exposure.19 The inability to further delineate the representation of darker skin types, such as those that may be classified as Fitzpatrick skin types V or VI,19 compared to those with lighter skin of color also was a limiting factor. This study was unable to assess for discussion of other common conditions affecting skin of color patients that were not listed as one of the priority conditions by SOCS. Photographs that were designated as indeterminate were difficult to elucidate as skin of color; however, it is possible that instructors may have verbally described these images as skin of color during lectures. Nonetheless, it may be beneficial for learners if teaching faculty were to clearly label instances where skin of color patients are shown or when notable differences are present.
Conclusion
Future studies would benefit from the inclusion of audio data from lectures, syllabi, and small group teaching materials from preclinical courses to more accurately assess representation of skin of color in dermatology training. Additionally, future studies also may expand to include images from lectures of overlapping clinical specialties, particularly infectious disease and rheumatology, to provide a broader assessment of skin of color exposure. Furthermore, repeat assessment may be beneficial to assess the longitudinal effectiveness of curricular changes at the institutions included in this study, comparing older lectures to more recent, updated lectures. This study also may be replicated at other medical schools to allow for wider comparison of curricula.
Acknowledgment—The authors wish to thank the institutions that offered and agreed to participate in this study with the hopes of improving medical education.
A ccording to the US Census Bureau, more than half of all Americans are projected to belong to a minority group, defined as any group other than non-Hispanic White alone, by 2044. 1 Consequently, the United States rapidly is becoming a country in which the majority of citizens will have skin of color. Individuals with skin of color are of diverse ethnic backgrounds and include people of African, Latin American, Native American, Pacific Islander, and Asian descent, as well as interethnic backgrounds. 2 Throughout the country, dermatologists along with primary care practitioners may be confronted with certain cutaneous conditions that have varying disease presentations or processes in patients with skin of color. It also is important to note that racial categories are socially rather than biologically constructed, and the term skin of color includes a wide variety of diverse skin types. Nevertheless, the current literature thoroughly supports unique pathophysiologic differences in skin of color as well as variations in disease manifestation compared to White patients. 3-5 For example, the increased lability of melanosomes in skin of color patients, which increases their risk for postinflammatory hyperpigmentation, has been well documented. 5-7 There are various dermatologic conditions that also occur with higher frequency and manifest uniquely in people with darker, more pigmented skin, 7-9 and dermatologists, along with primary care physicians, should feel prepared to recognize and address them.
Extensive evidence also indicates that there are unique aspects to consider while managing certain skin diseases in patients with skin of color.8,10,11 Consequently, as noted on the Skin of Color Society (SOCS) website, “[a]n increase in the body of dermatological literature concerning skin of color as well as the advancement of both basic science and clinical investigational research is necessary to meet the needs of the expanding skin of color population.”2 In the meantime, current knowledge regarding cutaneous conditions that diversely or disproportionately affect skin of color should be actively disseminated to physicians in training. Although patients with skin of color should always have access to comprehensive care and knowledgeable practitioners, the current changes in national and regional demographics further underscore the need for a more thorough understanding of skin of color with regard to disease pathogenesis, diagnosis, and treatment.
Several studies have found that medical students in the United States are minimally exposed to dermatology in general compared to other clinical specialties,12-14 which can easily lead to the underrecognition of disorders that may uniquely or disproportionately affect individuals with pigmented skin. Recent data showed that medical schools typically required fewer than 10 hours of dermatology instruction,12 and on average, dermatologic training made up less than 1% of a medical student’s undergraduate medical education.13,15,16 Consequently, less than 40% of primary care residents felt that their medical school curriculum adequately prepared them to manage common skin conditions.14 Although not all physicians should be expected to fully grasp the complexities of skin of color and its diagnostic and therapeutic implications, both practicing and training dermatologists have acknowledged a lack of exposure to skin of color. In one study, approximately 47% of dermatologists and dermatology residents reported that their medical training (medical school and/or residency) was inadequate in training them on skin conditions in Black patients. Furthermore, many who felt their training was lacking in skin of color identified the need for greater exposure to Black patients and training materials.15 The absence of comprehensive medical education regarding skin of color ultimately can be a disadvantage for both practitioners and patients, resulting in poorer outcomes. Furthermore, underrepresentation of skin of color may persist beyond undergraduate and graduate medical education. There also is evidence to suggest that noninclusion of skin of color pervades foundational dermatologic educational resources, including commonly used textbooks as well as continuing medical education disseminated at national conferences and meetings.17 Taken together, these findings highlight the need for more diverse and representative exposure to skin of color throughout medical training, which begins with a diverse inclusive undergraduate medical education in dermatology.
The objective of this study was to determine if the preclinical dermatology curriculum at 3 US medical schools provided adequate representation of skin of color patients in their didactic presentation slides.
Methods
Participants—Three US medical schools, a blend of private and public medical schools located across different geographic boundaries, agreed to participate in the study. All 3 institutions were current members of the American Medical Association (AMA) Accelerating Change in Medical Education consortium, whose primary goal is to create the medical school of the future and transform physician training.18 All 32 member institutions of the AMA consortium were contacted to request their participation in the study. As part of the consortium, these institutions have vowed to collectively work to develop and share the best models for educational advancement to improve care for patients, populations, and communities18 and would expectedly provide a more racially and ethnically inclusive curriculum than an institution not accountable to a group dedicated to identifying the best ways to deliver care for increasingly diverse communities.
Data Collection—Lectures were included if they were presented during dermatology preclinical courses in the 2015 to 2016 academic year. An uninvolved third party removed the names and identities of instructors to preserve anonymity. Two independent coders from different institutions extracted the data—lecture title, total number of clinical and histologic images, and number of skin of color images—from each of the anonymized lectures using a standardized coding form. We documented differences in skin of color noted in lectures and the disease context for the discussed differences, such as variations in clinical presentation, disease process, epidemiology/risk, and treatment between different skin phenotypes or ethnic groups. Photographs in which the coders were unable to differentiate whether the patient had skin of color were designated as indeterminate or unclear. Photographs appearing to represent Fitzpatrick skin types IV, V, and VI19 were categorically designated as skin of color, and those appearing to represent Fitzpatrick skin types I and II were described as not skin of color; however, images appearing to represent Fitzpatrick skin type III often were classified as not skin of color or indeterminate and occasionally skin of color. The Figure shows examples of images classified as skin of color, indeterminate, and not skin of color. Photographs often were classified as indeterminate due to poor lighting, close-up view photographs, or highlighted pathology obscuring the surrounding skin. We excluded duplicate photographs and histologic images from the analyses.

We also reviewed 19 conditions previously highlighted by the SOCS as areas of importance to skin of color patients.20 The coders tracked how many of these conditions were noted in each lecture. Duplicate discussion of these conditions was not included in the analyses. Any discrepancies between coders were resolved through additional slide review and discussion. The final coded data with the agreed upon changes were used for statistical analyses. Recent national demographic data from the US Census Bureau in 2019 describe approximately 39.9% of the population as belonging to racial/ethnic groups other than non-Hispanic/Latinx White.21 Consequently, the standard for adequate representation for skin of color photographs was set at 35% for the purpose of this study.
Results
Across all 3 institutions included in the study, the proportion of the total number of clinical photographs showing skin of color was 16% (290/1812). Eight percent of the total photographs (145/1812) were noted to be indeterminate (Table). For institution 1, 23.6% of photographs (155/658) showed skin of color, and 12.6% (83/658) were indeterminate. For institution 2, 13.1% (76/578) showed skin of color and 7.8% (45/578) were indeterminate. For institution 3, 10.2% (59/576) showed skin of color and 3% (17/576) were indeterminate.

Institutions 1, 2, and 3 had 18, 8, and 17 total dermatology lectures, respectively. Of the 19 conditions designated as areas of importance to skin of color patients by the SOCS, 16 (84.2%) were discussed by institution 1, 11 (57.9%) by institution 2, and 9 (47.4%) by institution 3 (eTable 1). Institution 3 did not include photographs of skin of color patients in its acne, psoriasis, or cutaneous malignancy lectures. Institution 1 also did not include any skin of color patients in its malignancy lecture. Lectures that focused on pigmentary disorders, atopic dermatitis, infectious conditions, and benign cutaneous neoplasms were more likely to display photographs of skin of color patients; for example, lectures that discussed infectious conditions, such as superficial mycoses, herpes viruses, human papillomavirus, syphilis, and atypical mycobacterial infections, were consistently among those with higher proportions of photographs of skin of color patients.

Throughout the entire preclinical dermatology course at all 3 institutions, of 2945 lecture slides, only 24 (0.8%) unique differences were noted between skin color and non–skin of color patients, with 10 total differences noted by institution 1, 6 by institution 2, and 8 by institution 3 (Table). The majority of these differences (19/24) were related to epidemiologic differences in prevalence among varying racial/ethnic groups, with only 5 instances highlighting differences in clinical presentation. There was only a single instance that elaborated on the underlying pathophysiologic mechanisms of the discussed difference. Of all 24 unique differences discussed, 8 were related to skin cancer, 3 were related to dermatitis, and 2 were related to the difference in manifestation of erythema in patients with darker skin (eTable 2).

Comment
The results of this study demonstrated that skin of color is underrepresented in the preclinical dermatology curriculum at these 3 institutions. Although only 16% of all included clinical photographs were of skin of color, individuals with skin of color will soon represent more than half of the total US population within the next 2 decades.1 To increase representation of skin of color patients, teaching faculty should consciously and deliberately include more photographs of skin of color patients for a wider variety of common conditions, including atopic dermatitis and psoriasis, in addition to those that tend to disparately affect skin of color patients, such as pseudofolliculitis barbae or melasma. Furthermore, they also can incorporate more detailed discussions about important differences seen in skin of color patients.
More Skin of Color Photographs in Psoriasis Lectures—At institution 3, there were no skin of color patients included in the psoriasis lecture, even though there is considerable data in the literature indicating notable differences in the clinical presentation, quality-of-life impact, and treatment of psoriasis in skin of color patients.11,22 There are multiple nuances in psoriasis manifestation in patients with skin of color, including less-conspicuous erythema in darker skin, higher degrees of dyspigmentation, and greater body surface area involvement. For Black patients with scalp psoriasis, the impact of hair texture, styling practices, and washing frequency are additional considerations that may impact disease severity and selection of topical therapy.11 The lack of inclusion of any skin of color patients in the psoriasis lecture at one institution further underscores the pressing need to prioritize communities of color in medical education.
More Skin of Color Photographs in Cutaneous Malignancy Lectures—Similarly, while a lecturer at institution 2 noted that acral lentiginous melanoma accounts for a considerable proportion of melanoma among skin of color patients,23 there was no mention of how melanoma generally is substantially more deadly in this population, potentially due to decreased awareness and inconsistent screening.24 Furthermore, at institutions 1 and 3, there were no photographs or discussion of skin of color patients during the cutaneous malignancy lectures. Evidence shows that more emphasis is needed for melanoma screening and awareness in skin of color populations to improve survival outcomes,24 and this begins with educating not only future dermatologists but all future physicians as well. The failure to include photographs of skin of color patients in discussions or lectures regarding cutaneous malignancies may serve to further perpetuate the harmful misperception that individuals with skin of color are unaffected by skin cancer.25,26
Analysis of Skin of Color Photographs in Infectious Disease Lectures—In addition, lectures discussing infectious etiologies were among those with the highest proportion of skin of color photographs. This relatively disproportionate representation of skin of color compared to the other lectures may contribute to the development of harmful stereotypes or the stigmatization of skin of color patients. Although skin of color should continue to be represented in similar lectures, teaching faculty should remain mindful of the potential unintended impact from lectures including relatively disproportionate amounts of skin of color, particularly when other lectures may have sparse to absent representation of skin of color.
More Photographs Available for Education—Overall, our findings may help to inform changes to preclinical dermatology medical education at other institutions to create more inclusive and representative curricula for skin of color patients. The ability of instructors to provide visual representation of various dermatologic conditions may be limited by the photographs available in certain textbooks with few examples of patients with skin of color; however, concerns regarding the lack of skin of color representation in dermatology training is not a novel discussion.17 Although it is the responsibility of all dermatologists to advocate for the inclusion of skin of color, many dermatologists of color have been leading the way in this movement for decades, publishing several textbooks to document various skin conditions in those with darker skin types and discuss unique considerations for patients with skin of color.27-29 Images from these textbooks can be utilized by programs to increase representation of skin of color in dermatology training. There also are multiple expanding online dermatologic databases, such as VisualDx, with an increasing focus on skin of color patients, some of which allow users to filter images by degree of skin pigmentation.30 Moreover, instructors also can work to diversify their curricula by highlighting more of the SOCS conditions of importance to skin of color patients, which have since been renamed and highlighted on the Patient Dermatology Education section of the SOCS website.20 These conditions, while not completely comprehensive, provide a useful starting point for medical educators to reevaluate for potential areas of improvement and inclusion.
There are several potential strategies that can be used to better represent skin of color in dermatologic preclinical medical education, including increasing awareness, especially among dermatology teaching faculty, of existing disparities in the representation of skin of color in the preclinical curricula. Additionally, all dermatology teaching materials could be reviewed at the department level prior to being disseminated to medical students to assess for instances in which skin of color could be prioritized for discussion or varying disease presentations in skin of color could be demonstrated. Finally, teaching faculty may consider photographing more clinical images of their skin of color patients to further develop a catalog of diverse images that can be used to teach students.
Study Limitations—Our study was unable to account for verbal discussion of skin of color not otherwise denoted or captured in lecture slides. Additional limitations include the utilization of Fitzpatrick skin types to describe and differentiate varying skin tones, as the Fitzpatrick scale originally was developed as a method to describe an individual’s response to UV exposure.19 The inability to further delineate the representation of darker skin types, such as those that may be classified as Fitzpatrick skin types V or VI,19 compared to those with lighter skin of color also was a limiting factor. This study was unable to assess for discussion of other common conditions affecting skin of color patients that were not listed as one of the priority conditions by SOCS. Photographs that were designated as indeterminate were difficult to elucidate as skin of color; however, it is possible that instructors may have verbally described these images as skin of color during lectures. Nonetheless, it may be beneficial for learners if teaching faculty were to clearly label instances where skin of color patients are shown or when notable differences are present.
Conclusion
Future studies would benefit from the inclusion of audio data from lectures, syllabi, and small group teaching materials from preclinical courses to more accurately assess representation of skin of color in dermatology training. Additionally, future studies also may expand to include images from lectures of overlapping clinical specialties, particularly infectious disease and rheumatology, to provide a broader assessment of skin of color exposure. Furthermore, repeat assessment may be beneficial to assess the longitudinal effectiveness of curricular changes at the institutions included in this study, comparing older lectures to more recent, updated lectures. This study also may be replicated at other medical schools to allow for wider comparison of curricula.
Acknowledgment—The authors wish to thank the institutions that offered and agreed to participate in this study with the hopes of improving medical education.
- Colby SL, Ortman JM. Projections of the size and composition of the US population: 2014 to 2060. United States Census Bureau website. Published March 2015. Accessed September 14, 2021. https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Learn more about SOCS. Skin of Color Society website. Accessed September 14, 2021. http://skinofcolorsociety.org/about-socs/
- Taylor SC. Skin of color: biology, structure, function, and implications for dermatologic disease. J Am Acad Dermatol. 2002;46(suppl 2):S41-S62.
- Berardesca E, Maibach H. Ethnic skin: overview of structure and function. J Am Acad Dermatol. 2003;48(suppl 6):S139-S142.
- Callender VD, Surin-Lord SS, Davis EC, et al. Postinflammatory hyperpigmentation. Am J Clin Dermatol. 2011;12:87-99.
- Davis EC, Callender VD. Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol. 2010;3:20-31.
- Grimes PE, Stockton T. Pigmentary disorders in blacks. Dermatol Clin. 1988;6:271-281.
- Halder RM, Nootheti PK. Ethnic skin disorders overview. J Am Acad Dermatol. 2003;48(suppl 6):S143-S148.
- Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
- Callender VD. Acne in ethnic skin: special considerations for therapy. Dermatol Ther. 2004;17:184-195.
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Ramsay DL, Mayer F. National survey of undergraduate dermatologic medical education. Arch Dermatol.1985;121:1529-1530.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59, viii.
- Knable A, Hood AF, Pearson TG. Undergraduate medical education in dermatology: report from the AAD Interdisciplinary Education Committee, Subcommittee on Undergraduate Medical Education. J Am Acad Dermatol. 1997;36:467-470.
- Ebede T, Papier A. Disparities in dermatology educational resources. J Am Acad Dermatol. 2006;55:687-690.
- Skochelak SE, Stack SJ. Creating the medical schools of the future. Acad Med. 2017;92:16-19.
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol. 1988;124:869-871.
- Skin of Color Society. Patient dermatology education. Accessed September 22, 2021. https://skinofcolorsociety.org/patient-dermatology-education
- QuickFacts: United States. US Census Bureau website. Updated July 1, 2019. Accessed September 14, 2021. https://www.census.gov/quickfacts/fact/table/US#
- Kaufman BP, Alexis AF. Psoriasis in skin of color: insights into the epidemiology, clinical presentation, genetics, quality-of-life impact, and treatment of psoriasis in non-white racial/ethnic groups. Am J Clin Dermatol. 2018;19:405-423.
- Bradford PT, Goldstein AM, McMaster ML, et al. Acral lentiginous melanoma: incidence and survival patterns in the United States, 1986-2005. Arch Dermatol. 2009;145:427-434.
- Dawes SM, Tsai S, Gittleman H, et al. Racial disparities in melanoma survival. J Am Acad Dermatol. 2016;75:983-991.
- Pipitone M, Robinson JK, Camara C, et al. Skin cancer awareness in suburban employees: a Hispanic perspective. J Am Acad Dermatol. 2002;47:118-123.
- Imahiyerobo-Ip J, Ip I, Jamal S, et al. Skin cancer awareness in communities of color. J Am Acad Dermatol. 2011;64:198-200.
- Taylor SSC, Serrano AMA, Kelly AP, et al, eds. Taylor and Kelly’s Dermatology for Skin of Color. 2nd ed. McGraw-Hill Education; 2016.
- Dadzie OE, Petit A, Alexis AF, eds. Ethnic Dermatology: Principles and Practice. Wiley-Blackwell; 2013.
- Jackson-Richards D, Pandya AG, eds. Dermatology Atlas for Skin of Color. Springer; 2014.
- VisualDx. New VisualDx feature: skin of color sort. Published October 14, 2020. Accessed September 22, 2021. https://www.visualdx.com/blog/new-visualdx-feature-skin-of-color-sort/
- Colby SL, Ortman JM. Projections of the size and composition of the US population: 2014 to 2060. United States Census Bureau website. Published March 2015. Accessed September 14, 2021. https://www.census.gov/content/dam/Census/library/publications/2015/demo/p25-1143.pdf
- Learn more about SOCS. Skin of Color Society website. Accessed September 14, 2021. http://skinofcolorsociety.org/about-socs/
- Taylor SC. Skin of color: biology, structure, function, and implications for dermatologic disease. J Am Acad Dermatol. 2002;46(suppl 2):S41-S62.
- Berardesca E, Maibach H. Ethnic skin: overview of structure and function. J Am Acad Dermatol. 2003;48(suppl 6):S139-S142.
- Callender VD, Surin-Lord SS, Davis EC, et al. Postinflammatory hyperpigmentation. Am J Clin Dermatol. 2011;12:87-99.
- Davis EC, Callender VD. Postinflammatory hyperpigmentation: a review of the epidemiology, clinical features, and treatment options in skin of color. J Clin Aesthet Dermatol. 2010;3:20-31.
- Grimes PE, Stockton T. Pigmentary disorders in blacks. Dermatol Clin. 1988;6:271-281.
- Halder RM, Nootheti PK. Ethnic skin disorders overview. J Am Acad Dermatol. 2003;48(suppl 6):S143-S148.
- Alexis AF, Sergay AB, Taylor SC. Common dermatologic disorders in skin of color: a comparative practice survey. Cutis. 2007;80:387-394.
- Callender VD. Acne in ethnic skin: special considerations for therapy. Dermatol Ther. 2004;17:184-195.
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- McCleskey PE, Gilson RT, DeVillez RL. Medical student core curriculum in dermatology survey. J Am Acad Dermatol. 2009;61:30-35.
- Ramsay DL, Mayer F. National survey of undergraduate dermatologic medical education. Arch Dermatol.1985;121:1529-1530.
- Hansra NK, O’Sullivan P, Chen CL, et al. Medical school dermatology curriculum: are we adequately preparing primary care physicians? J Am Acad Dermatol. 2009;61:23-29.
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59, viii.
- Knable A, Hood AF, Pearson TG. Undergraduate medical education in dermatology: report from the AAD Interdisciplinary Education Committee, Subcommittee on Undergraduate Medical Education. J Am Acad Dermatol. 1997;36:467-470.
- Ebede T, Papier A. Disparities in dermatology educational resources. J Am Acad Dermatol. 2006;55:687-690.
- Skochelak SE, Stack SJ. Creating the medical schools of the future. Acad Med. 2017;92:16-19.
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol. 1988;124:869-871.
- Skin of Color Society. Patient dermatology education. Accessed September 22, 2021. https://skinofcolorsociety.org/patient-dermatology-education
- QuickFacts: United States. US Census Bureau website. Updated July 1, 2019. Accessed September 14, 2021. https://www.census.gov/quickfacts/fact/table/US#
- Kaufman BP, Alexis AF. Psoriasis in skin of color: insights into the epidemiology, clinical presentation, genetics, quality-of-life impact, and treatment of psoriasis in non-white racial/ethnic groups. Am J Clin Dermatol. 2018;19:405-423.
- Bradford PT, Goldstein AM, McMaster ML, et al. Acral lentiginous melanoma: incidence and survival patterns in the United States, 1986-2005. Arch Dermatol. 2009;145:427-434.
- Dawes SM, Tsai S, Gittleman H, et al. Racial disparities in melanoma survival. J Am Acad Dermatol. 2016;75:983-991.
- Pipitone M, Robinson JK, Camara C, et al. Skin cancer awareness in suburban employees: a Hispanic perspective. J Am Acad Dermatol. 2002;47:118-123.
- Imahiyerobo-Ip J, Ip I, Jamal S, et al. Skin cancer awareness in communities of color. J Am Acad Dermatol. 2011;64:198-200.
- Taylor SSC, Serrano AMA, Kelly AP, et al, eds. Taylor and Kelly’s Dermatology for Skin of Color. 2nd ed. McGraw-Hill Education; 2016.
- Dadzie OE, Petit A, Alexis AF, eds. Ethnic Dermatology: Principles and Practice. Wiley-Blackwell; 2013.
- Jackson-Richards D, Pandya AG, eds. Dermatology Atlas for Skin of Color. Springer; 2014.
- VisualDx. New VisualDx feature: skin of color sort. Published October 14, 2020. Accessed September 22, 2021. https://www.visualdx.com/blog/new-visualdx-feature-skin-of-color-sort/
Practice Points
- The United States rapidly is becoming a country in which the majority of citizens will have skin of color.
- Our study results strongly suggest that skin of color may be seriously underrepresented in medical education and can guide modifications to preclinical dermatology medical education to develop a more comprehensive and inclusive curriculum.
- Efforts should be made to increase images and discussion of skin of color in preclinical didactics.
The Role of Inpatient Dermatology Consultations
Dermatology is an often-underutilized resource in the hospital setting. As the health care landscape has evolved, so has the role of the inpatient dermatologist.1-3 Structural changes in the health system and advances in therapies have shifted dermatology from an admitting service to an almost exclusively outpatient practice. Improved treatment modalities led to decreases in the number of patients requiring admission for chronic dermatoses, and outpatient clinics began offering therapies once limited to hospitals.1,4 Inpatient dermatology consultations emerged and continue to have profound effects on hospitalized patients regardless of their reason for admission.1-11
Inpatient dermatologists supply knowledge in areas primary medical teams lack, and there is evidence that dermatology consultations improve the quality of care while decreasing cost.2,5-7 Establishing correct diagnoses, preventing exposure to unnecessary medications, and reducing hospitalization duration and readmission rates are a few ways dermatology consultations positively impact hospitalized patients.2,5-7,9,10 This study highlights the role of the dermatologist in the care of hospitalized patients at a large academic medical center in an urban setting and reveals how consultation supports the efficiency and efficacy of other services.
Materials and Methods
Study Design—This single-institution, cross-sectional retrospective study included all hospitalized patients at the Thomas Jefferson University Hospital (Philadelphia, Pennsylvania), who received an inpatient dermatology consultation completed by physicians of Jefferson Dermatology Associates between January 1, 2019, and December 31, 2019. The institutional review board at Thomas Jefferson University approved this study.
Data Collection—A list of all inpatient dermatology consultations in 2019 was provided by Jefferson Dermatology Associates. Through a retrospective chart review, data regarding the consultations were collected from the electronic medical record (Epic Systems) and recorded into the Research Electronic Data Capture system. Data on patient demographics, the primary medical team, the dermatology evaluation, and the hospital course of the patient were collected.
Results
Patient Characteristics—Dermatology received 253 inpatient consultation requests during this time period; 53% of patients were female and 47% were male, with a mean age of 55 years. Most patients were White (57%), while 34% were Black. Five percent and 4% of patients were Asian and Hispanic or Latino, respectively (Table 1). The mean duration of hospitalization for all patients was 15 days, and the average number of days to discharge following the first encounter with dermatology was 10 days.
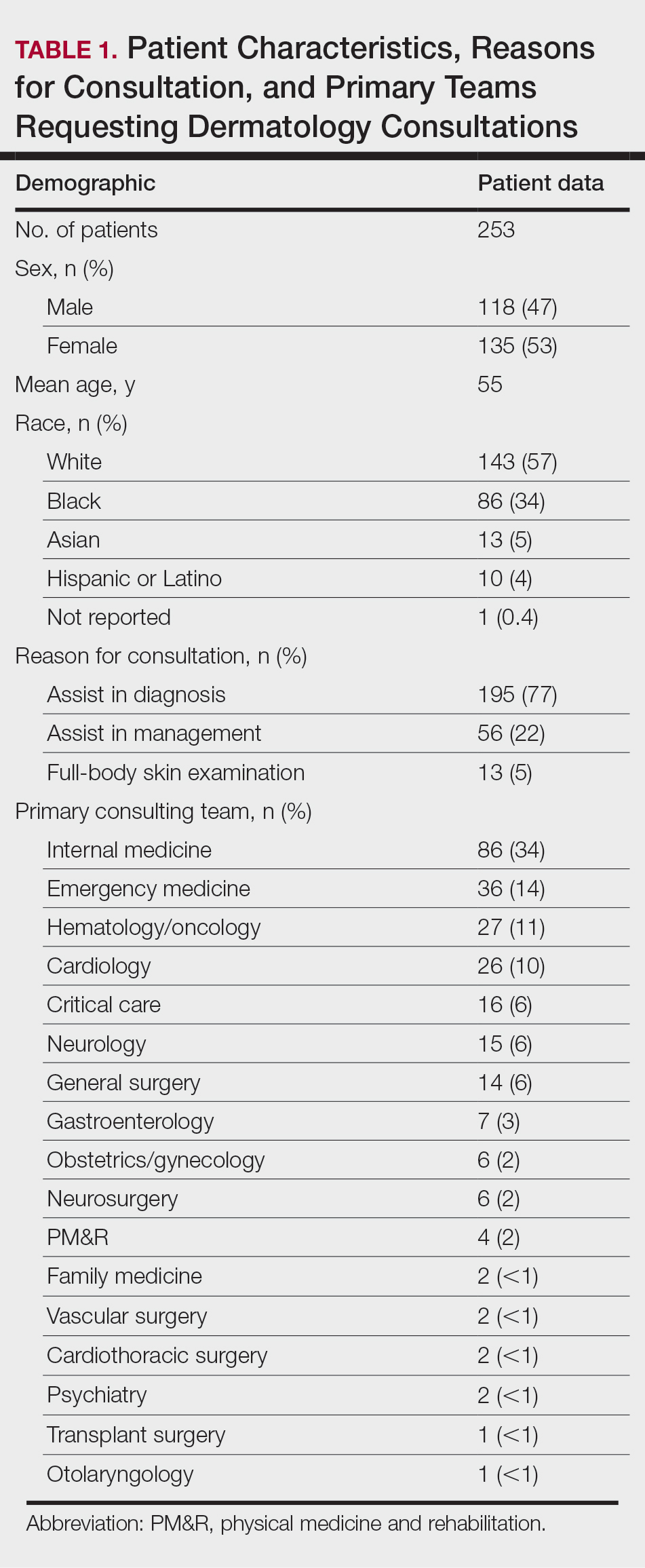
Requesting Team and Reason for Consultation—Internal medicine consulted dermatology most frequently (34% of all consultations), followed by emergency medicine (14%) and a variety of other services (Table 1). Most dermatology consultations were placed to assist in achieving a diagnosis of a cutaneous condition (77%), while a minority were to assist in the management of a previously diagnosed disease (22%). A small fraction of consultations (5%) were to complete full-body skin examinations (FBSEs) to rule out infection or malignancy in candidates for organ transplantation, left ventricular assist devices, or certain chemotherapies. One FBSE was conducted to search for a primary tumor in a patient diagnosed with metastatic melanoma.
Most Common Final Diagnoses and Consultation Impact—Table 2 lists the most common final diagnosis categories, as well as the effects of the consultation on diagnosis, management, biopsies, hospitalization, and clinical improvement as documented by the primary medical provider. The most common final diagnoses were inflammatory and autoimmune (39%), such as contact dermatitis and seborrheic dermatitis; infectious (23%), such as varicella (primary or zoster) and bacterial furunculosis; drug reactions (20%), such as morbilliform drug eruptions; vascular (8%), such as vasculitis and calciphylaxis; neoplastic (7%), such as keratinocyte carcinomas and leukemia cutis; and other (15%), such as xerosis, keratosis pilaris, and miliaria rubra.
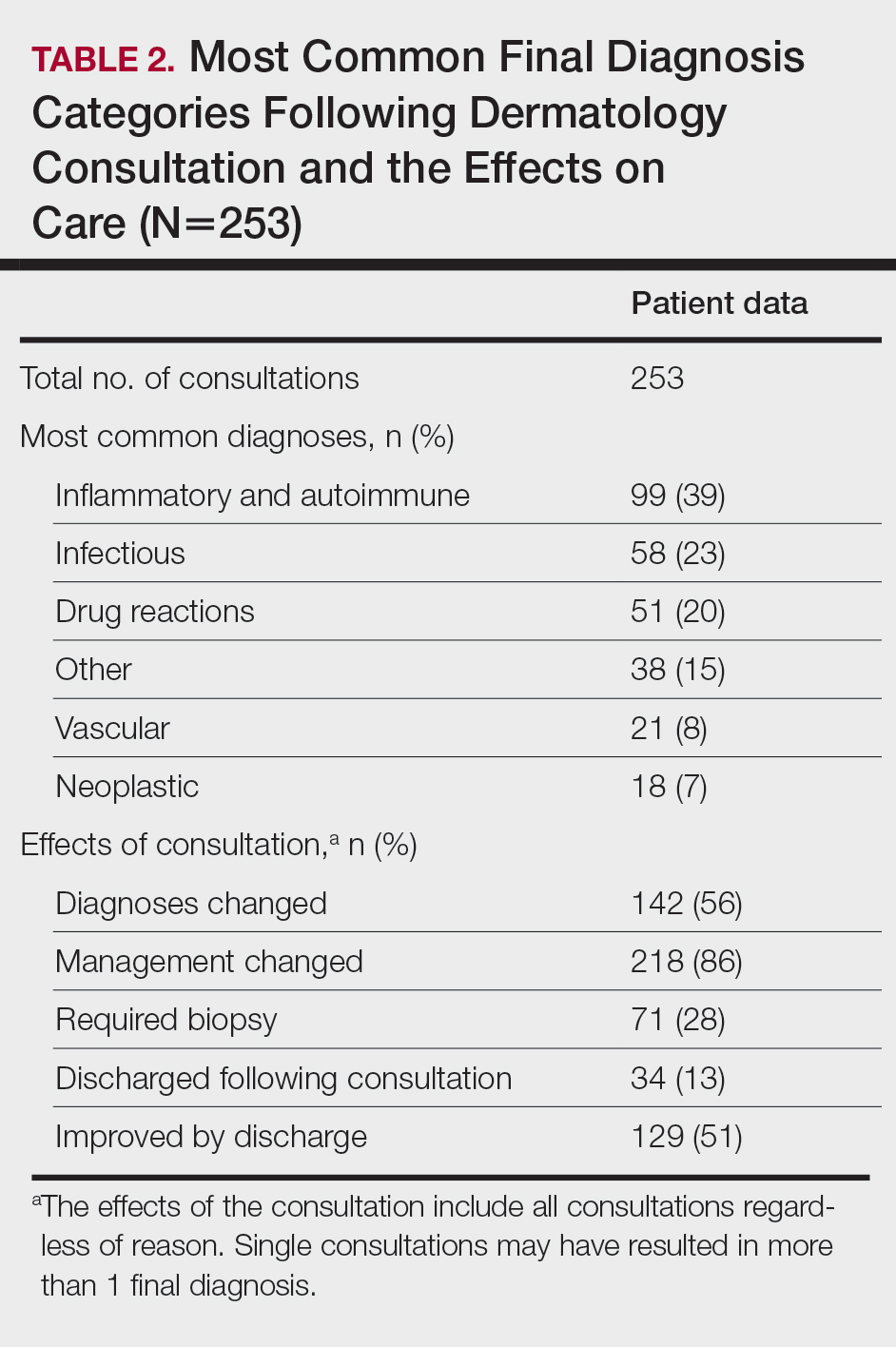
Impact on Diagnosis—Fifty-six percent of all consultations resulted in a change in diagnosis. When dermatology was consulted specifically to assist in the diagnosis of a patient (195 consultations), the working diagnosis of the primary team was changed 69% of the time. Thirty-five of these consultation requests had no preliminary diagnosis, and the primary team listed the working diagnosis as either rash or a morphologic description of the lesion(s). Sixty-three percent of suspected drug eruptions ended with a diagnosis of a form of drug eruption, while 20% of consultations for suspected cellulitis or bacterial infections were confirmed to be cellulitis or soft tissue infections.
Impact on Management—Regardless of the reason for the consultation, most consultations (86%) resulted in a change in management. The remaining 14% consisted of FBSEs with benign findings; cases of cutaneous metastases and leukemia cutis managed by oncology; as well as select cases of purpura fulminans, postfebrile desquamation, and postinflammatory hyperpigmentation.
Changes in management included alterations in medications, requests for additional laboratory work or imaging, additional consultation requests, biopsies, or specific wound care instructions. Seventy-five percent of all consultations were given specific medication recommendations by dermatology. Most (61%) were recommended to be given a topical steroid, antibiotic, or both. However, 45% of all consultations were recommended to initiate a systemic medication, most commonly antihistamines, antibiotics, steroids, antivirals, or immunomodulators. Dermatology recommended discontinuing specific medications in 16% of all consultations, with antibiotics being the most frequent culprit (17 antibiotics discontinued), owing to drug eruptions or misdiagnosed infections. Vancomycin, piperacillin-tazobactam, and trimethoprim-sulfamethoxazole were the most frequently discontinued antibiotics.
Dermatology was consulted for assistance in management of previously diagnosed cutaneous conditions 56 times (22% of all consultations), often regarding complicated cases of hidradenitis suppurativa (9 cases), pyoderma gangrenosum (5 cases), bullous pemphigoid (4 cases), or erythroderma (4 cases). Most of these cases required a single dermatology encounter to provide recommendations (71%), and 21% required 1 additional follow-up. Sixty-three percent of patients consulted for management assistance were noted to have improvement in their cutaneous condition by time of discharge, as documented by the primary provider in the medical record.
Twenty-eight percent of all consultations required at least 1 biopsy. Seventy-two percent of all biopsies were consistent with the dermatologist’s working diagnosis or highest-ranked differential diagnosis, and 16% of biopsy results were consistent with the second- or third-ranked diagnosis. The primary teams requested a biopsy 38 times to assist in diagnosis, as documented in the progress note or consultation request. Only 21 of these consultations (55% of requests) received at least 1 biopsy, as the remaining consultations did not require a biopsy to establish a diagnosis. The most common final diagnoses of consultations receiving biopsies included drug eruptions (5), leukemia cutis (4), vasculopathies (4), vasculitis (4), and calciphylaxis (3).
Impact on Hospitalization and Efficacy—Dermatology performed 217 consultations regarding patients already admitted to the hospital, and 92% remained hospitalized either due to comorbidities or complicated cutaneous conditions following the consultation. The remaining 8% were cleared for discharge. Dermatology received 36 consultation requests from emergency medicine physicians. Fifty-three percent of these patients were admitted, while the remaining 47% were discharged from the emergency department or its observation unit following evaluation.
Fifty-one percent of all consultations were noted to have improvement in their cutaneous condition by the time of discharge, as noted in the physical examination, progress note, or discharge summary of the primary team. Thirty percent of cases remained stable, where improvement was not noted in in the medical record. Most of these cases involved keratinocyte carcinomas scheduled for outpatient excision, benign melanocytic nevi found on FBSE, and benign etiologies that led to immediate discharge following consultation. Three percent of all consultations were noted to have worsened following consultation, including cases of calciphylaxis, vasculopathies, and purpura fulminans, as well as patients who elected for palliative care and hospice. The cutaneous condition by the time of discharge could not be determined from the medical record in 16% of all consultations.
Eighty-five percent of all consultations required a single encounter with dermatology. An additional 10% required a single follow-up with dermatology, while only 5% of patients required 3 or more encounters. Notably, these cases included patients with 1 or more severe cutaneous diseases, such as Sweet syndrome, calciphylaxis, Stevens-Johnson syndrome/toxic epidermal necrolysis, and hidradenitis suppurativa.
Comment
Although dermatology often is viewed as an outpatient specialty, this study provides a glimpse into the ways inpatient dermatology consultations optimize the care of hospitalized patients. Most consultations involved assistance in diagnosing an unknown condition, but several regarded pre-existing skin disorders requiring management aid. As a variety of medical specialties requested consultations, dermatology was able to provide care to a diverse group of patients with conditions varying in complexity and severity. Several specialties benefited from niche dermatologic expertise: hematology and oncology frequently requested dermatology to assist in diagnosis and management of the toxic effects of chemotherapy, cutaneous metastasis, or suspected cutaneous infections in immunocompromised patients. Cardiology patients were frequently evaluated for potential malignancy or infection prior to heart transplantation and initiation of antirejection immunosuppressants. Dermatology was consulted to differentiate cutaneous manifestations of critical illness from underlying systemic disease in the intensive care unit, and patients presenting to the emergency department often were examined to determine if hospital admission was necessary, with 47% of these consultations resulting in a discharge following evaluation by a dermatologist.
Our results were consistent with prior studies1,5,6 that have reported frequent changes in final diagnosis following dermatology consultation, with 69% of working diagnoses changed in this study when consultation was requested for diagnostic assistance. When dermatology was consulted for diagnostic assistance, several of these cases lacked a preliminary differential diagnosis. Although the absence of a documented differential diagnosis may not necessarily reflect a lack of suspicion for a particular etiology, 86% of all consultations included a ranked differential or working diagnosis either in the consultation request or progress note prior to consultation. The final diagnoses of consultations without a preliminary diagnosis varied from the mild and localized to systemic and severe, further suggesting these cases reflected knowledge gaps of the primary medical team.
Integration of dermatology into the care of hospitalized patients could provide an opportunity for education of primary medical teams. With frequent consultation, primary medical teams may become more comfortable diagnosing and managing common cutaneous conditions specific to their specialty or extended hospitalizations.
Several consultations were requested to aid in management of cases of hidradenitis suppurativa, pyoderma gangrenosum, or bullous pemphigoid that either failed outpatient therapy or were complicated by superinfections. Despite the ranges in complexity, the majority of all consultations required a single encounter and led to improvement by the time of discharge, demonstrating the efficacy and efficiency of inpatient dermatologists.
Dermatology consultations often led to changes in management involving medications and additional workup. Changes in management also extended to specific wound care instructions provided by dermatology, as expected for cases of Stevens-Johnson syndrome/toxic epidermal necrolysis, Sweet syndrome, hidradenitis suppurativa, and pyoderma gangrenosum. However, patients with the sequelae of extended hospitalizations, such as chronic wounds, pressure ulcers, and edema bullae, also benefited from this expertise.
When patients required a biopsy, the final diagnoses were consistent with the dermatologist’s number one differential diagnosis or top 3 differential diagnoses 72% and 88% of the time, respectively. Only 55% of cases where the primary team requested a biopsy ultimately required a biopsy, as many involved clinical diagnoses such as urticaria. Not only was dermatology accurate in their preliminary diagnoses, but they decreased cost and morbidity by avoiding unnecessary procedures.
This study provided additional evidence to support the integration of dermatology into the hospital setting for the benefit of patients, primary medical teams, and hospital systems. Dermatology offers high-value care through the efficient diagnosis and management of hospitalized patients, which contributes to decreased cost and improved outcomes.2,5-7,9,10 This study highlighted lesser-known areas of impact, such as the various specialty-specific services dermatology provides as well as the high rates of reported improvement following consultation. Future studies should continue to explore the field’s unique impact on hospitalized medicine as well as other avenues of care delivery, such as telemedicine, that may encourage dermatologists to participate in consultations and increase the volume of patients who may benefit from their care.
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Noe MH, Rosenbach M. Inpatient dermatologists—crucial for the management of skin diseases in hospitalized patients [editorial]. JAMA Dermatol. 2018;154:524-525. doi:10.1001/jamadermatol.2017.6195
- Strowd LC. Inpatient dermatology: a paradigm shift in the management of skin disease in the hospital. Br J Dermatol. 2019;180:966-967. doi:10.1111/bjd.17778
- Kirsner RS, Yang DG, Kerdel FA. The changing status of inpatient dermatology at American academic dermatology programs. J Am Acad Dermatol. 1999;40:755-757. doi:10.1016/s0190-9622(99)70158-1
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480. doi:10.1001/jamadermatol.2015.5098
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis. JAMA Dermatol. 2018;154:529-533. doi:10.1001/jamadermatol.2017.6196
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543. doi:10.1001/jamadermatol.2017.6197
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Imadojemu S, Rosenbach M. Dermatologists must take an active role in the diagnosis of cellulitis. JAMA Dermatol. 2017;153:134-135. doi:10.1001/jamadermatol.2016.4230
- Hughey LC. The impact dermatologists can have on misdiagnosis of cellulitis and overuse of antibiotics: closing the gap. JAMA Dermatol. 2014;150:1061-1062. doi:10.1001/jamadermatol.2014.1164
- Ko LN, Kroshinsky D. Dermatology hospitalists: a multicenter survey study characterizing the infrastructure of consultative dermatology in select American hospitals. Int J Dermatol. 2018;57:553-558. doi:10.1111/ijd.13939
Dermatology is an often-underutilized resource in the hospital setting. As the health care landscape has evolved, so has the role of the inpatient dermatologist.1-3 Structural changes in the health system and advances in therapies have shifted dermatology from an admitting service to an almost exclusively outpatient practice. Improved treatment modalities led to decreases in the number of patients requiring admission for chronic dermatoses, and outpatient clinics began offering therapies once limited to hospitals.1,4 Inpatient dermatology consultations emerged and continue to have profound effects on hospitalized patients regardless of their reason for admission.1-11
Inpatient dermatologists supply knowledge in areas primary medical teams lack, and there is evidence that dermatology consultations improve the quality of care while decreasing cost.2,5-7 Establishing correct diagnoses, preventing exposure to unnecessary medications, and reducing hospitalization duration and readmission rates are a few ways dermatology consultations positively impact hospitalized patients.2,5-7,9,10 This study highlights the role of the dermatologist in the care of hospitalized patients at a large academic medical center in an urban setting and reveals how consultation supports the efficiency and efficacy of other services.
Materials and Methods
Study Design—This single-institution, cross-sectional retrospective study included all hospitalized patients at the Thomas Jefferson University Hospital (Philadelphia, Pennsylvania), who received an inpatient dermatology consultation completed by physicians of Jefferson Dermatology Associates between January 1, 2019, and December 31, 2019. The institutional review board at Thomas Jefferson University approved this study.
Data Collection—A list of all inpatient dermatology consultations in 2019 was provided by Jefferson Dermatology Associates. Through a retrospective chart review, data regarding the consultations were collected from the electronic medical record (Epic Systems) and recorded into the Research Electronic Data Capture system. Data on patient demographics, the primary medical team, the dermatology evaluation, and the hospital course of the patient were collected.
Results
Patient Characteristics—Dermatology received 253 inpatient consultation requests during this time period; 53% of patients were female and 47% were male, with a mean age of 55 years. Most patients were White (57%), while 34% were Black. Five percent and 4% of patients were Asian and Hispanic or Latino, respectively (Table 1). The mean duration of hospitalization for all patients was 15 days, and the average number of days to discharge following the first encounter with dermatology was 10 days.

Requesting Team and Reason for Consultation—Internal medicine consulted dermatology most frequently (34% of all consultations), followed by emergency medicine (14%) and a variety of other services (Table 1). Most dermatology consultations were placed to assist in achieving a diagnosis of a cutaneous condition (77%), while a minority were to assist in the management of a previously diagnosed disease (22%). A small fraction of consultations (5%) were to complete full-body skin examinations (FBSEs) to rule out infection or malignancy in candidates for organ transplantation, left ventricular assist devices, or certain chemotherapies. One FBSE was conducted to search for a primary tumor in a patient diagnosed with metastatic melanoma.
Most Common Final Diagnoses and Consultation Impact—Table 2 lists the most common final diagnosis categories, as well as the effects of the consultation on diagnosis, management, biopsies, hospitalization, and clinical improvement as documented by the primary medical provider. The most common final diagnoses were inflammatory and autoimmune (39%), such as contact dermatitis and seborrheic dermatitis; infectious (23%), such as varicella (primary or zoster) and bacterial furunculosis; drug reactions (20%), such as morbilliform drug eruptions; vascular (8%), such as vasculitis and calciphylaxis; neoplastic (7%), such as keratinocyte carcinomas and leukemia cutis; and other (15%), such as xerosis, keratosis pilaris, and miliaria rubra.

Impact on Diagnosis—Fifty-six percent of all consultations resulted in a change in diagnosis. When dermatology was consulted specifically to assist in the diagnosis of a patient (195 consultations), the working diagnosis of the primary team was changed 69% of the time. Thirty-five of these consultation requests had no preliminary diagnosis, and the primary team listed the working diagnosis as either rash or a morphologic description of the lesion(s). Sixty-three percent of suspected drug eruptions ended with a diagnosis of a form of drug eruption, while 20% of consultations for suspected cellulitis or bacterial infections were confirmed to be cellulitis or soft tissue infections.
Impact on Management—Regardless of the reason for the consultation, most consultations (86%) resulted in a change in management. The remaining 14% consisted of FBSEs with benign findings; cases of cutaneous metastases and leukemia cutis managed by oncology; as well as select cases of purpura fulminans, postfebrile desquamation, and postinflammatory hyperpigmentation.
Changes in management included alterations in medications, requests for additional laboratory work or imaging, additional consultation requests, biopsies, or specific wound care instructions. Seventy-five percent of all consultations were given specific medication recommendations by dermatology. Most (61%) were recommended to be given a topical steroid, antibiotic, or both. However, 45% of all consultations were recommended to initiate a systemic medication, most commonly antihistamines, antibiotics, steroids, antivirals, or immunomodulators. Dermatology recommended discontinuing specific medications in 16% of all consultations, with antibiotics being the most frequent culprit (17 antibiotics discontinued), owing to drug eruptions or misdiagnosed infections. Vancomycin, piperacillin-tazobactam, and trimethoprim-sulfamethoxazole were the most frequently discontinued antibiotics.
Dermatology was consulted for assistance in management of previously diagnosed cutaneous conditions 56 times (22% of all consultations), often regarding complicated cases of hidradenitis suppurativa (9 cases), pyoderma gangrenosum (5 cases), bullous pemphigoid (4 cases), or erythroderma (4 cases). Most of these cases required a single dermatology encounter to provide recommendations (71%), and 21% required 1 additional follow-up. Sixty-three percent of patients consulted for management assistance were noted to have improvement in their cutaneous condition by time of discharge, as documented by the primary provider in the medical record.
Twenty-eight percent of all consultations required at least 1 biopsy. Seventy-two percent of all biopsies were consistent with the dermatologist’s working diagnosis or highest-ranked differential diagnosis, and 16% of biopsy results were consistent with the second- or third-ranked diagnosis. The primary teams requested a biopsy 38 times to assist in diagnosis, as documented in the progress note or consultation request. Only 21 of these consultations (55% of requests) received at least 1 biopsy, as the remaining consultations did not require a biopsy to establish a diagnosis. The most common final diagnoses of consultations receiving biopsies included drug eruptions (5), leukemia cutis (4), vasculopathies (4), vasculitis (4), and calciphylaxis (3).
Impact on Hospitalization and Efficacy—Dermatology performed 217 consultations regarding patients already admitted to the hospital, and 92% remained hospitalized either due to comorbidities or complicated cutaneous conditions following the consultation. The remaining 8% were cleared for discharge. Dermatology received 36 consultation requests from emergency medicine physicians. Fifty-three percent of these patients were admitted, while the remaining 47% were discharged from the emergency department or its observation unit following evaluation.
Fifty-one percent of all consultations were noted to have improvement in their cutaneous condition by the time of discharge, as noted in the physical examination, progress note, or discharge summary of the primary team. Thirty percent of cases remained stable, where improvement was not noted in in the medical record. Most of these cases involved keratinocyte carcinomas scheduled for outpatient excision, benign melanocytic nevi found on FBSE, and benign etiologies that led to immediate discharge following consultation. Three percent of all consultations were noted to have worsened following consultation, including cases of calciphylaxis, vasculopathies, and purpura fulminans, as well as patients who elected for palliative care and hospice. The cutaneous condition by the time of discharge could not be determined from the medical record in 16% of all consultations.
Eighty-five percent of all consultations required a single encounter with dermatology. An additional 10% required a single follow-up with dermatology, while only 5% of patients required 3 or more encounters. Notably, these cases included patients with 1 or more severe cutaneous diseases, such as Sweet syndrome, calciphylaxis, Stevens-Johnson syndrome/toxic epidermal necrolysis, and hidradenitis suppurativa.
Comment
Although dermatology often is viewed as an outpatient specialty, this study provides a glimpse into the ways inpatient dermatology consultations optimize the care of hospitalized patients. Most consultations involved assistance in diagnosing an unknown condition, but several regarded pre-existing skin disorders requiring management aid. As a variety of medical specialties requested consultations, dermatology was able to provide care to a diverse group of patients with conditions varying in complexity and severity. Several specialties benefited from niche dermatologic expertise: hematology and oncology frequently requested dermatology to assist in diagnosis and management of the toxic effects of chemotherapy, cutaneous metastasis, or suspected cutaneous infections in immunocompromised patients. Cardiology patients were frequently evaluated for potential malignancy or infection prior to heart transplantation and initiation of antirejection immunosuppressants. Dermatology was consulted to differentiate cutaneous manifestations of critical illness from underlying systemic disease in the intensive care unit, and patients presenting to the emergency department often were examined to determine if hospital admission was necessary, with 47% of these consultations resulting in a discharge following evaluation by a dermatologist.
Our results were consistent with prior studies1,5,6 that have reported frequent changes in final diagnosis following dermatology consultation, with 69% of working diagnoses changed in this study when consultation was requested for diagnostic assistance. When dermatology was consulted for diagnostic assistance, several of these cases lacked a preliminary differential diagnosis. Although the absence of a documented differential diagnosis may not necessarily reflect a lack of suspicion for a particular etiology, 86% of all consultations included a ranked differential or working diagnosis either in the consultation request or progress note prior to consultation. The final diagnoses of consultations without a preliminary diagnosis varied from the mild and localized to systemic and severe, further suggesting these cases reflected knowledge gaps of the primary medical team.
Integration of dermatology into the care of hospitalized patients could provide an opportunity for education of primary medical teams. With frequent consultation, primary medical teams may become more comfortable diagnosing and managing common cutaneous conditions specific to their specialty or extended hospitalizations.
Several consultations were requested to aid in management of cases of hidradenitis suppurativa, pyoderma gangrenosum, or bullous pemphigoid that either failed outpatient therapy or were complicated by superinfections. Despite the ranges in complexity, the majority of all consultations required a single encounter and led to improvement by the time of discharge, demonstrating the efficacy and efficiency of inpatient dermatologists.
Dermatology consultations often led to changes in management involving medications and additional workup. Changes in management also extended to specific wound care instructions provided by dermatology, as expected for cases of Stevens-Johnson syndrome/toxic epidermal necrolysis, Sweet syndrome, hidradenitis suppurativa, and pyoderma gangrenosum. However, patients with the sequelae of extended hospitalizations, such as chronic wounds, pressure ulcers, and edema bullae, also benefited from this expertise.
When patients required a biopsy, the final diagnoses were consistent with the dermatologist’s number one differential diagnosis or top 3 differential diagnoses 72% and 88% of the time, respectively. Only 55% of cases where the primary team requested a biopsy ultimately required a biopsy, as many involved clinical diagnoses such as urticaria. Not only was dermatology accurate in their preliminary diagnoses, but they decreased cost and morbidity by avoiding unnecessary procedures.
This study provided additional evidence to support the integration of dermatology into the hospital setting for the benefit of patients, primary medical teams, and hospital systems. Dermatology offers high-value care through the efficient diagnosis and management of hospitalized patients, which contributes to decreased cost and improved outcomes.2,5-7,9,10 This study highlighted lesser-known areas of impact, such as the various specialty-specific services dermatology provides as well as the high rates of reported improvement following consultation. Future studies should continue to explore the field’s unique impact on hospitalized medicine as well as other avenues of care delivery, such as telemedicine, that may encourage dermatologists to participate in consultations and increase the volume of patients who may benefit from their care.
Dermatology is an often-underutilized resource in the hospital setting. As the health care landscape has evolved, so has the role of the inpatient dermatologist.1-3 Structural changes in the health system and advances in therapies have shifted dermatology from an admitting service to an almost exclusively outpatient practice. Improved treatment modalities led to decreases in the number of patients requiring admission for chronic dermatoses, and outpatient clinics began offering therapies once limited to hospitals.1,4 Inpatient dermatology consultations emerged and continue to have profound effects on hospitalized patients regardless of their reason for admission.1-11
Inpatient dermatologists supply knowledge in areas primary medical teams lack, and there is evidence that dermatology consultations improve the quality of care while decreasing cost.2,5-7 Establishing correct diagnoses, preventing exposure to unnecessary medications, and reducing hospitalization duration and readmission rates are a few ways dermatology consultations positively impact hospitalized patients.2,5-7,9,10 This study highlights the role of the dermatologist in the care of hospitalized patients at a large academic medical center in an urban setting and reveals how consultation supports the efficiency and efficacy of other services.
Materials and Methods
Study Design—This single-institution, cross-sectional retrospective study included all hospitalized patients at the Thomas Jefferson University Hospital (Philadelphia, Pennsylvania), who received an inpatient dermatology consultation completed by physicians of Jefferson Dermatology Associates between January 1, 2019, and December 31, 2019. The institutional review board at Thomas Jefferson University approved this study.
Data Collection—A list of all inpatient dermatology consultations in 2019 was provided by Jefferson Dermatology Associates. Through a retrospective chart review, data regarding the consultations were collected from the electronic medical record (Epic Systems) and recorded into the Research Electronic Data Capture system. Data on patient demographics, the primary medical team, the dermatology evaluation, and the hospital course of the patient were collected.
Results
Patient Characteristics—Dermatology received 253 inpatient consultation requests during this time period; 53% of patients were female and 47% were male, with a mean age of 55 years. Most patients were White (57%), while 34% were Black. Five percent and 4% of patients were Asian and Hispanic or Latino, respectively (Table 1). The mean duration of hospitalization for all patients was 15 days, and the average number of days to discharge following the first encounter with dermatology was 10 days.

Requesting Team and Reason for Consultation—Internal medicine consulted dermatology most frequently (34% of all consultations), followed by emergency medicine (14%) and a variety of other services (Table 1). Most dermatology consultations were placed to assist in achieving a diagnosis of a cutaneous condition (77%), while a minority were to assist in the management of a previously diagnosed disease (22%). A small fraction of consultations (5%) were to complete full-body skin examinations (FBSEs) to rule out infection or malignancy in candidates for organ transplantation, left ventricular assist devices, or certain chemotherapies. One FBSE was conducted to search for a primary tumor in a patient diagnosed with metastatic melanoma.
Most Common Final Diagnoses and Consultation Impact—Table 2 lists the most common final diagnosis categories, as well as the effects of the consultation on diagnosis, management, biopsies, hospitalization, and clinical improvement as documented by the primary medical provider. The most common final diagnoses were inflammatory and autoimmune (39%), such as contact dermatitis and seborrheic dermatitis; infectious (23%), such as varicella (primary or zoster) and bacterial furunculosis; drug reactions (20%), such as morbilliform drug eruptions; vascular (8%), such as vasculitis and calciphylaxis; neoplastic (7%), such as keratinocyte carcinomas and leukemia cutis; and other (15%), such as xerosis, keratosis pilaris, and miliaria rubra.

Impact on Diagnosis—Fifty-six percent of all consultations resulted in a change in diagnosis. When dermatology was consulted specifically to assist in the diagnosis of a patient (195 consultations), the working diagnosis of the primary team was changed 69% of the time. Thirty-five of these consultation requests had no preliminary diagnosis, and the primary team listed the working diagnosis as either rash or a morphologic description of the lesion(s). Sixty-three percent of suspected drug eruptions ended with a diagnosis of a form of drug eruption, while 20% of consultations for suspected cellulitis or bacterial infections were confirmed to be cellulitis or soft tissue infections.
Impact on Management—Regardless of the reason for the consultation, most consultations (86%) resulted in a change in management. The remaining 14% consisted of FBSEs with benign findings; cases of cutaneous metastases and leukemia cutis managed by oncology; as well as select cases of purpura fulminans, postfebrile desquamation, and postinflammatory hyperpigmentation.
Changes in management included alterations in medications, requests for additional laboratory work or imaging, additional consultation requests, biopsies, or specific wound care instructions. Seventy-five percent of all consultations were given specific medication recommendations by dermatology. Most (61%) were recommended to be given a topical steroid, antibiotic, or both. However, 45% of all consultations were recommended to initiate a systemic medication, most commonly antihistamines, antibiotics, steroids, antivirals, or immunomodulators. Dermatology recommended discontinuing specific medications in 16% of all consultations, with antibiotics being the most frequent culprit (17 antibiotics discontinued), owing to drug eruptions or misdiagnosed infections. Vancomycin, piperacillin-tazobactam, and trimethoprim-sulfamethoxazole were the most frequently discontinued antibiotics.
Dermatology was consulted for assistance in management of previously diagnosed cutaneous conditions 56 times (22% of all consultations), often regarding complicated cases of hidradenitis suppurativa (9 cases), pyoderma gangrenosum (5 cases), bullous pemphigoid (4 cases), or erythroderma (4 cases). Most of these cases required a single dermatology encounter to provide recommendations (71%), and 21% required 1 additional follow-up. Sixty-three percent of patients consulted for management assistance were noted to have improvement in their cutaneous condition by time of discharge, as documented by the primary provider in the medical record.
Twenty-eight percent of all consultations required at least 1 biopsy. Seventy-two percent of all biopsies were consistent with the dermatologist’s working diagnosis or highest-ranked differential diagnosis, and 16% of biopsy results were consistent with the second- or third-ranked diagnosis. The primary teams requested a biopsy 38 times to assist in diagnosis, as documented in the progress note or consultation request. Only 21 of these consultations (55% of requests) received at least 1 biopsy, as the remaining consultations did not require a biopsy to establish a diagnosis. The most common final diagnoses of consultations receiving biopsies included drug eruptions (5), leukemia cutis (4), vasculopathies (4), vasculitis (4), and calciphylaxis (3).
Impact on Hospitalization and Efficacy—Dermatology performed 217 consultations regarding patients already admitted to the hospital, and 92% remained hospitalized either due to comorbidities or complicated cutaneous conditions following the consultation. The remaining 8% were cleared for discharge. Dermatology received 36 consultation requests from emergency medicine physicians. Fifty-three percent of these patients were admitted, while the remaining 47% were discharged from the emergency department or its observation unit following evaluation.
Fifty-one percent of all consultations were noted to have improvement in their cutaneous condition by the time of discharge, as noted in the physical examination, progress note, or discharge summary of the primary team. Thirty percent of cases remained stable, where improvement was not noted in in the medical record. Most of these cases involved keratinocyte carcinomas scheduled for outpatient excision, benign melanocytic nevi found on FBSE, and benign etiologies that led to immediate discharge following consultation. Three percent of all consultations were noted to have worsened following consultation, including cases of calciphylaxis, vasculopathies, and purpura fulminans, as well as patients who elected for palliative care and hospice. The cutaneous condition by the time of discharge could not be determined from the medical record in 16% of all consultations.
Eighty-five percent of all consultations required a single encounter with dermatology. An additional 10% required a single follow-up with dermatology, while only 5% of patients required 3 or more encounters. Notably, these cases included patients with 1 or more severe cutaneous diseases, such as Sweet syndrome, calciphylaxis, Stevens-Johnson syndrome/toxic epidermal necrolysis, and hidradenitis suppurativa.
Comment
Although dermatology often is viewed as an outpatient specialty, this study provides a glimpse into the ways inpatient dermatology consultations optimize the care of hospitalized patients. Most consultations involved assistance in diagnosing an unknown condition, but several regarded pre-existing skin disorders requiring management aid. As a variety of medical specialties requested consultations, dermatology was able to provide care to a diverse group of patients with conditions varying in complexity and severity. Several specialties benefited from niche dermatologic expertise: hematology and oncology frequently requested dermatology to assist in diagnosis and management of the toxic effects of chemotherapy, cutaneous metastasis, or suspected cutaneous infections in immunocompromised patients. Cardiology patients were frequently evaluated for potential malignancy or infection prior to heart transplantation and initiation of antirejection immunosuppressants. Dermatology was consulted to differentiate cutaneous manifestations of critical illness from underlying systemic disease in the intensive care unit, and patients presenting to the emergency department often were examined to determine if hospital admission was necessary, with 47% of these consultations resulting in a discharge following evaluation by a dermatologist.
Our results were consistent with prior studies1,5,6 that have reported frequent changes in final diagnosis following dermatology consultation, with 69% of working diagnoses changed in this study when consultation was requested for diagnostic assistance. When dermatology was consulted for diagnostic assistance, several of these cases lacked a preliminary differential diagnosis. Although the absence of a documented differential diagnosis may not necessarily reflect a lack of suspicion for a particular etiology, 86% of all consultations included a ranked differential or working diagnosis either in the consultation request or progress note prior to consultation. The final diagnoses of consultations without a preliminary diagnosis varied from the mild and localized to systemic and severe, further suggesting these cases reflected knowledge gaps of the primary medical team.
Integration of dermatology into the care of hospitalized patients could provide an opportunity for education of primary medical teams. With frequent consultation, primary medical teams may become more comfortable diagnosing and managing common cutaneous conditions specific to their specialty or extended hospitalizations.
Several consultations were requested to aid in management of cases of hidradenitis suppurativa, pyoderma gangrenosum, or bullous pemphigoid that either failed outpatient therapy or were complicated by superinfections. Despite the ranges in complexity, the majority of all consultations required a single encounter and led to improvement by the time of discharge, demonstrating the efficacy and efficiency of inpatient dermatologists.
Dermatology consultations often led to changes in management involving medications and additional workup. Changes in management also extended to specific wound care instructions provided by dermatology, as expected for cases of Stevens-Johnson syndrome/toxic epidermal necrolysis, Sweet syndrome, hidradenitis suppurativa, and pyoderma gangrenosum. However, patients with the sequelae of extended hospitalizations, such as chronic wounds, pressure ulcers, and edema bullae, also benefited from this expertise.
When patients required a biopsy, the final diagnoses were consistent with the dermatologist’s number one differential diagnosis or top 3 differential diagnoses 72% and 88% of the time, respectively. Only 55% of cases where the primary team requested a biopsy ultimately required a biopsy, as many involved clinical diagnoses such as urticaria. Not only was dermatology accurate in their preliminary diagnoses, but they decreased cost and morbidity by avoiding unnecessary procedures.
This study provided additional evidence to support the integration of dermatology into the hospital setting for the benefit of patients, primary medical teams, and hospital systems. Dermatology offers high-value care through the efficient diagnosis and management of hospitalized patients, which contributes to decreased cost and improved outcomes.2,5-7,9,10 This study highlighted lesser-known areas of impact, such as the various specialty-specific services dermatology provides as well as the high rates of reported improvement following consultation. Future studies should continue to explore the field’s unique impact on hospitalized medicine as well as other avenues of care delivery, such as telemedicine, that may encourage dermatologists to participate in consultations and increase the volume of patients who may benefit from their care.
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Noe MH, Rosenbach M. Inpatient dermatologists—crucial for the management of skin diseases in hospitalized patients [editorial]. JAMA Dermatol. 2018;154:524-525. doi:10.1001/jamadermatol.2017.6195
- Strowd LC. Inpatient dermatology: a paradigm shift in the management of skin disease in the hospital. Br J Dermatol. 2019;180:966-967. doi:10.1111/bjd.17778
- Kirsner RS, Yang DG, Kerdel FA. The changing status of inpatient dermatology at American academic dermatology programs. J Am Acad Dermatol. 1999;40:755-757. doi:10.1016/s0190-9622(99)70158-1
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480. doi:10.1001/jamadermatol.2015.5098
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis. JAMA Dermatol. 2018;154:529-533. doi:10.1001/jamadermatol.2017.6196
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543. doi:10.1001/jamadermatol.2017.6197
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Imadojemu S, Rosenbach M. Dermatologists must take an active role in the diagnosis of cellulitis. JAMA Dermatol. 2017;153:134-135. doi:10.1001/jamadermatol.2016.4230
- Hughey LC. The impact dermatologists can have on misdiagnosis of cellulitis and overuse of antibiotics: closing the gap. JAMA Dermatol. 2014;150:1061-1062. doi:10.1001/jamadermatol.2014.1164
- Ko LN, Kroshinsky D. Dermatology hospitalists: a multicenter survey study characterizing the infrastructure of consultative dermatology in select American hospitals. Int J Dermatol. 2018;57:553-558. doi:10.1111/ijd.13939
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Noe MH, Rosenbach M. Inpatient dermatologists—crucial for the management of skin diseases in hospitalized patients [editorial]. JAMA Dermatol. 2018;154:524-525. doi:10.1001/jamadermatol.2017.6195
- Strowd LC. Inpatient dermatology: a paradigm shift in the management of skin disease in the hospital. Br J Dermatol. 2019;180:966-967. doi:10.1111/bjd.17778
- Kirsner RS, Yang DG, Kerdel FA. The changing status of inpatient dermatology at American academic dermatology programs. J Am Acad Dermatol. 1999;40:755-757. doi:10.1016/s0190-9622(99)70158-1
- Kroshinsky D, Cotliar J, Hughey LC, et al. Association of dermatology consultation with accuracy of cutaneous disorder diagnoses in hospitalized patients: a multicenter analysis. JAMA Dermatol. 2016;152:477-480. doi:10.1001/jamadermatol.2015.5098
- Ko LN, Garza-Mayers AC, St John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis. JAMA Dermatol. 2018;154:529-533. doi:10.1001/jamadermatol.2017.6196
- Li DG, Xia FD, Khosravi H, et al. Outcomes of early dermatology consultation for inpatients diagnosed with cellulitis. JAMA Dermatol. 2018;154:537-543. doi:10.1001/jamadermatol.2017.6197
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Imadojemu S, Rosenbach M. Dermatologists must take an active role in the diagnosis of cellulitis. JAMA Dermatol. 2017;153:134-135. doi:10.1001/jamadermatol.2016.4230
- Hughey LC. The impact dermatologists can have on misdiagnosis of cellulitis and overuse of antibiotics: closing the gap. JAMA Dermatol. 2014;150:1061-1062. doi:10.1001/jamadermatol.2014.1164
- Ko LN, Kroshinsky D. Dermatology hospitalists: a multicenter survey study characterizing the infrastructure of consultative dermatology in select American hospitals. Int J Dermatol. 2018;57:553-558. doi:10.1111/ijd.13939
Practice Points
- Inpatient dermatologists fill knowledge gaps that often alter the diagnosis, management, and hospital course of hospitalized patients.
- Several medical specialties benefit from niche expertise of inpatient dermatologists specific to their patient population.
- Integration of inpatient dermatology consultations can prevent unnecessary hospital admissions and medication administration.
Treatment Stacking: Optimizing Therapeutic Regimens for Hidradenitis Suppurativa
Hidradenitis suppurativa (HS) is a debilitating chronic condition that often is recalcitrant to first-line treatments, and mechanisms underlying its pathology remain unclear. Existing data suggest a multifactorial etiology with different pathophysiologic contributors, including genetic, hormonal, and immune dysregulation factors. At this time, only one medication (adalimumab) is US Food and Drug Administration approved for HS, but multiple medical and procedural therapies are available.1 Herein, we discuss the concept of treatment stacking, or the combination of unique therapeutic modalities—an approach we believe is key to optimizing management of HS patients.
Stacking Treatments for HS
Unlike psoriasis, in which a single biologic agent may provide 100% clearance (psoriasis area and severity index 100 [PASI 100]) without adjuvant treatment,2,3 the field of HS currently lacks medications that are efficacious to that degree of success as monotherapy. In HS, the benchmark for a positive treatment outcome is Hidradenitis Suppurativa Clinical Response 50 (HiSCR50),4 a 50% reduction in inflammatory lesion count—a far less stringent marker for disease improvement. Thus, providers should design HS treatment regimens with a model of combining therapies and shift away from monotherapy. Targeting different pathophysiologic pathways by stacking multiple treatments may provide synergistic benefits for HS patients. Treatment stacking is a familiar concept in acne; for instance, patients who benefit tremendously from isotretinoin may still require a hormone-modulating treatment (eg, spironolactone) to attain optimal results.
Adherence to a rigid treatment algorithm based on disease severity limits the potential to create comprehensive regimens that account for unique patient characteristics and clinical manifestations. When evaluating an HS patient, providers should systematically consider each pathophysiologic factor and target the ones that appear to be most involved in that particular patient. The North American HS guidelines illustrate this point by supporting use of several treatments across different Hurley stages, such as recommending hormonal treatment in patients with Hurley stages 1, 2, or 3.1 Of note, treatment stacking also includes procedural therapies. Surgeons typically prefer a patient’s disease management to be optimized prior to surgery, including reduced drainage and inflammation. In addition, even after surgery, patients often still require medical management to prevent continued disease worsening.
Treatment Pathways for HS
A multimodal approach with treatment stacking (Figure) can be useful to all HS patients, from those with the mildest to the most severe disease. Modifiable pathophysiologic factors and examples of their targeted treatments include (1) follicular occlusion (eg, oral retinoids), (2) metabolic dysfunction (eg, metformin), (3) hormones (eg, oral contraceptive pills, spironolactone, finasteride), (4) dysbiosis (eg, antibiotics such as clindamycin and rifampin combination therapy), (5) immune dysregulation (eg, biologic agents), and (6) friction/irritation (eg, weight loss, clothing recommendations).
Combining treatments from different pathways enables potentiation of individual treatment efficacies. A female patient with only a few HS nodules that flare with menses may be well controlled with spironolactone as her only systemic agent; however, she still may benefit from use of an antiseptic wash, topical clindamycin, and lifestyle changes such as weight loss and reduction of mechanical irritation. A patient with severe recalcitrant HS could notably benefit from concomitant biologic, systemic antibiotic, and hormonal/metabolic treatments. If disease control is still inadequate, agents within the same class can be switched (eg, choosing a different biologic) or other disease-modifying agents such as colchicine also can be added. The goal is to create an effective treatment toolbox with therapies targeting different pathophysiologic arms of HS and working together in synergy. Each tool can be refined by modifying dosing frequency and duration of use to strive for optimal response. At this time, the literature on HS combination therapy is sparse. A retrospective study of 31 patients reported promising combinations, including isotretinoin with spironolactone for mild disease, isotretinoin or doxycycline with adalimumab for moderate disease, and cyclosporine with adalimumab for severe disease.5 Larger prospective studies on clinical response to different combination regimens are warranted.
Optimizing Therapy for HS and Its Comorbidities
Additional considerations may further optimize treatment plans. Some therapies benefit all patients; for example, providers should counsel all HS patients on healthy weight management, optimized clothing choices,6 and friction reduction in the intertriginous folds. Providers also may consider adding therapies with faster onset of efficacy as a bridge to long-term, slower-onset therapies. For instance, female HS patients with menstrual flares who are prescribed spironolactone also may benefit from a course of systemic antibiotics, which typically provides more prompt relief. Treatment regimens also can concomitantly treat HS and its comorbidities.7 For example, metformin serves a dual purpose in HS patients with diabetes mellitus, and adalimumab in patients with both HS and inflammatory bowel disease.
Final Thoughts
The last decade has seen tremendous growth in HS research8 coupled with a remarkable expansion in the therapeutic pipeline.9 However, currently no single therapy for HS can guarantee satisfactory disease remission or durability of remission. The contrast between clinical trials and real-world practice should be acknowledged; the former often is restrictive in design with monotherapy and allowance of very limited concomitant treatments, such as topical or oral antibiotics. This limits our ability to draw conclusions regarding the additive synergistic potential of different therapeutics in combination. In clinical practice, we are not restricted by monotherapy trial protocols. As we await new tools, treatment stacking allows for creating a framework to best utilize the tools that are available to us.
Although HS has continued to affect the lives of many patients, improved understanding of underlying pathophysiology and a well-placed sense of urgency from all stakeholders (ie, patients, clinicians, researchers, industry partners) has pushed this field forward. Until our therapeutic armamentarium has expanded to include highly efficacious monotherapy options, providers should consider treatment stacking for every HS patient.
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi:10.1016/j.jaad.2019.02.068
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMoa2102383
- Imafuku S, Nakagawa H, Igarashi A, et al. Long-term efficacy and safety of tildrakizumab in Japanese patients with moderate to severe plaque psoriasis: results from a 5-year extension of a phase 3 study (reSURFACE 1). J Dermatol. 2021;48:844-852. doi:10.1111/1346-8138.15763
- Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434. doi:10.1056/NEJMoa1504370
- McPhie ML, Bridgman AC, Kirchhof MG. Combination therapies for hidradenitis suppurativa: a retrospective chart review of 31 patients. J Cutan Med Surg. 2019;23:270-276. doi:10.1177/1203475418823529
- Loh TY, Hendricks AJ, Hsiao JL, et al. Undergarment and fabric selection in the management of hidradenitis suppurativa. Dermatol Basel Switz. 2021;237:119-124. doi:10.1159/000501611
- Garg A, Malviya N, Strunk A, et al. Comorbidity screening in hidradenitis suppurativa: evidence-based recommendations from the US and Canadian Hidradenitis Suppurativa Foundations [published online January 23, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.01.059
- Savage KT, Brant EG, Flood KS, et al. Publication trends in hidradenitis suppurativa from 2008 to 2018. J Eur Acad Dermatol Venereol. 2020;34:1885-1889. doi:10.1111/jdv.16213
- van Straalen KR, Schneider-Burrus S, Prens EP. Current and future treatment of hidradenitis suppurativa. Br J Dermatol. 2020;183:E178-E187. doi:10.1111/bjd.16768
Hidradenitis suppurativa (HS) is a debilitating chronic condition that often is recalcitrant to first-line treatments, and mechanisms underlying its pathology remain unclear. Existing data suggest a multifactorial etiology with different pathophysiologic contributors, including genetic, hormonal, and immune dysregulation factors. At this time, only one medication (adalimumab) is US Food and Drug Administration approved for HS, but multiple medical and procedural therapies are available.1 Herein, we discuss the concept of treatment stacking, or the combination of unique therapeutic modalities—an approach we believe is key to optimizing management of HS patients.
Stacking Treatments for HS
Unlike psoriasis, in which a single biologic agent may provide 100% clearance (psoriasis area and severity index 100 [PASI 100]) without adjuvant treatment,2,3 the field of HS currently lacks medications that are efficacious to that degree of success as monotherapy. In HS, the benchmark for a positive treatment outcome is Hidradenitis Suppurativa Clinical Response 50 (HiSCR50),4 a 50% reduction in inflammatory lesion count—a far less stringent marker for disease improvement. Thus, providers should design HS treatment regimens with a model of combining therapies and shift away from monotherapy. Targeting different pathophysiologic pathways by stacking multiple treatments may provide synergistic benefits for HS patients. Treatment stacking is a familiar concept in acne; for instance, patients who benefit tremendously from isotretinoin may still require a hormone-modulating treatment (eg, spironolactone) to attain optimal results.
Adherence to a rigid treatment algorithm based on disease severity limits the potential to create comprehensive regimens that account for unique patient characteristics and clinical manifestations. When evaluating an HS patient, providers should systematically consider each pathophysiologic factor and target the ones that appear to be most involved in that particular patient. The North American HS guidelines illustrate this point by supporting use of several treatments across different Hurley stages, such as recommending hormonal treatment in patients with Hurley stages 1, 2, or 3.1 Of note, treatment stacking also includes procedural therapies. Surgeons typically prefer a patient’s disease management to be optimized prior to surgery, including reduced drainage and inflammation. In addition, even after surgery, patients often still require medical management to prevent continued disease worsening.
Treatment Pathways for HS
A multimodal approach with treatment stacking (Figure) can be useful to all HS patients, from those with the mildest to the most severe disease. Modifiable pathophysiologic factors and examples of their targeted treatments include (1) follicular occlusion (eg, oral retinoids), (2) metabolic dysfunction (eg, metformin), (3) hormones (eg, oral contraceptive pills, spironolactone, finasteride), (4) dysbiosis (eg, antibiotics such as clindamycin and rifampin combination therapy), (5) immune dysregulation (eg, biologic agents), and (6) friction/irritation (eg, weight loss, clothing recommendations).

Combining treatments from different pathways enables potentiation of individual treatment efficacies. A female patient with only a few HS nodules that flare with menses may be well controlled with spironolactone as her only systemic agent; however, she still may benefit from use of an antiseptic wash, topical clindamycin, and lifestyle changes such as weight loss and reduction of mechanical irritation. A patient with severe recalcitrant HS could notably benefit from concomitant biologic, systemic antibiotic, and hormonal/metabolic treatments. If disease control is still inadequate, agents within the same class can be switched (eg, choosing a different biologic) or other disease-modifying agents such as colchicine also can be added. The goal is to create an effective treatment toolbox with therapies targeting different pathophysiologic arms of HS and working together in synergy. Each tool can be refined by modifying dosing frequency and duration of use to strive for optimal response. At this time, the literature on HS combination therapy is sparse. A retrospective study of 31 patients reported promising combinations, including isotretinoin with spironolactone for mild disease, isotretinoin or doxycycline with adalimumab for moderate disease, and cyclosporine with adalimumab for severe disease.5 Larger prospective studies on clinical response to different combination regimens are warranted.
Optimizing Therapy for HS and Its Comorbidities
Additional considerations may further optimize treatment plans. Some therapies benefit all patients; for example, providers should counsel all HS patients on healthy weight management, optimized clothing choices,6 and friction reduction in the intertriginous folds. Providers also may consider adding therapies with faster onset of efficacy as a bridge to long-term, slower-onset therapies. For instance, female HS patients with menstrual flares who are prescribed spironolactone also may benefit from a course of systemic antibiotics, which typically provides more prompt relief. Treatment regimens also can concomitantly treat HS and its comorbidities.7 For example, metformin serves a dual purpose in HS patients with diabetes mellitus, and adalimumab in patients with both HS and inflammatory bowel disease.
Final Thoughts
The last decade has seen tremendous growth in HS research8 coupled with a remarkable expansion in the therapeutic pipeline.9 However, currently no single therapy for HS can guarantee satisfactory disease remission or durability of remission. The contrast between clinical trials and real-world practice should be acknowledged; the former often is restrictive in design with monotherapy and allowance of very limited concomitant treatments, such as topical or oral antibiotics. This limits our ability to draw conclusions regarding the additive synergistic potential of different therapeutics in combination. In clinical practice, we are not restricted by monotherapy trial protocols. As we await new tools, treatment stacking allows for creating a framework to best utilize the tools that are available to us.
Although HS has continued to affect the lives of many patients, improved understanding of underlying pathophysiology and a well-placed sense of urgency from all stakeholders (ie, patients, clinicians, researchers, industry partners) has pushed this field forward. Until our therapeutic armamentarium has expanded to include highly efficacious monotherapy options, providers should consider treatment stacking for every HS patient.
Hidradenitis suppurativa (HS) is a debilitating chronic condition that often is recalcitrant to first-line treatments, and mechanisms underlying its pathology remain unclear. Existing data suggest a multifactorial etiology with different pathophysiologic contributors, including genetic, hormonal, and immune dysregulation factors. At this time, only one medication (adalimumab) is US Food and Drug Administration approved for HS, but multiple medical and procedural therapies are available.1 Herein, we discuss the concept of treatment stacking, or the combination of unique therapeutic modalities—an approach we believe is key to optimizing management of HS patients.
Stacking Treatments for HS
Unlike psoriasis, in which a single biologic agent may provide 100% clearance (psoriasis area and severity index 100 [PASI 100]) without adjuvant treatment,2,3 the field of HS currently lacks medications that are efficacious to that degree of success as monotherapy. In HS, the benchmark for a positive treatment outcome is Hidradenitis Suppurativa Clinical Response 50 (HiSCR50),4 a 50% reduction in inflammatory lesion count—a far less stringent marker for disease improvement. Thus, providers should design HS treatment regimens with a model of combining therapies and shift away from monotherapy. Targeting different pathophysiologic pathways by stacking multiple treatments may provide synergistic benefits for HS patients. Treatment stacking is a familiar concept in acne; for instance, patients who benefit tremendously from isotretinoin may still require a hormone-modulating treatment (eg, spironolactone) to attain optimal results.
Adherence to a rigid treatment algorithm based on disease severity limits the potential to create comprehensive regimens that account for unique patient characteristics and clinical manifestations. When evaluating an HS patient, providers should systematically consider each pathophysiologic factor and target the ones that appear to be most involved in that particular patient. The North American HS guidelines illustrate this point by supporting use of several treatments across different Hurley stages, such as recommending hormonal treatment in patients with Hurley stages 1, 2, or 3.1 Of note, treatment stacking also includes procedural therapies. Surgeons typically prefer a patient’s disease management to be optimized prior to surgery, including reduced drainage and inflammation. In addition, even after surgery, patients often still require medical management to prevent continued disease worsening.
Treatment Pathways for HS
A multimodal approach with treatment stacking (Figure) can be useful to all HS patients, from those with the mildest to the most severe disease. Modifiable pathophysiologic factors and examples of their targeted treatments include (1) follicular occlusion (eg, oral retinoids), (2) metabolic dysfunction (eg, metformin), (3) hormones (eg, oral contraceptive pills, spironolactone, finasteride), (4) dysbiosis (eg, antibiotics such as clindamycin and rifampin combination therapy), (5) immune dysregulation (eg, biologic agents), and (6) friction/irritation (eg, weight loss, clothing recommendations).

Combining treatments from different pathways enables potentiation of individual treatment efficacies. A female patient with only a few HS nodules that flare with menses may be well controlled with spironolactone as her only systemic agent; however, she still may benefit from use of an antiseptic wash, topical clindamycin, and lifestyle changes such as weight loss and reduction of mechanical irritation. A patient with severe recalcitrant HS could notably benefit from concomitant biologic, systemic antibiotic, and hormonal/metabolic treatments. If disease control is still inadequate, agents within the same class can be switched (eg, choosing a different biologic) or other disease-modifying agents such as colchicine also can be added. The goal is to create an effective treatment toolbox with therapies targeting different pathophysiologic arms of HS and working together in synergy. Each tool can be refined by modifying dosing frequency and duration of use to strive for optimal response. At this time, the literature on HS combination therapy is sparse. A retrospective study of 31 patients reported promising combinations, including isotretinoin with spironolactone for mild disease, isotretinoin or doxycycline with adalimumab for moderate disease, and cyclosporine with adalimumab for severe disease.5 Larger prospective studies on clinical response to different combination regimens are warranted.
Optimizing Therapy for HS and Its Comorbidities
Additional considerations may further optimize treatment plans. Some therapies benefit all patients; for example, providers should counsel all HS patients on healthy weight management, optimized clothing choices,6 and friction reduction in the intertriginous folds. Providers also may consider adding therapies with faster onset of efficacy as a bridge to long-term, slower-onset therapies. For instance, female HS patients with menstrual flares who are prescribed spironolactone also may benefit from a course of systemic antibiotics, which typically provides more prompt relief. Treatment regimens also can concomitantly treat HS and its comorbidities.7 For example, metformin serves a dual purpose in HS patients with diabetes mellitus, and adalimumab in patients with both HS and inflammatory bowel disease.
Final Thoughts
The last decade has seen tremendous growth in HS research8 coupled with a remarkable expansion in the therapeutic pipeline.9 However, currently no single therapy for HS can guarantee satisfactory disease remission or durability of remission. The contrast between clinical trials and real-world practice should be acknowledged; the former often is restrictive in design with monotherapy and allowance of very limited concomitant treatments, such as topical or oral antibiotics. This limits our ability to draw conclusions regarding the additive synergistic potential of different therapeutics in combination. In clinical practice, we are not restricted by monotherapy trial protocols. As we await new tools, treatment stacking allows for creating a framework to best utilize the tools that are available to us.
Although HS has continued to affect the lives of many patients, improved understanding of underlying pathophysiology and a well-placed sense of urgency from all stakeholders (ie, patients, clinicians, researchers, industry partners) has pushed this field forward. Until our therapeutic armamentarium has expanded to include highly efficacious monotherapy options, providers should consider treatment stacking for every HS patient.
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi:10.1016/j.jaad.2019.02.068
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMoa2102383
- Imafuku S, Nakagawa H, Igarashi A, et al. Long-term efficacy and safety of tildrakizumab in Japanese patients with moderate to severe plaque psoriasis: results from a 5-year extension of a phase 3 study (reSURFACE 1). J Dermatol. 2021;48:844-852. doi:10.1111/1346-8138.15763
- Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434. doi:10.1056/NEJMoa1504370
- McPhie ML, Bridgman AC, Kirchhof MG. Combination therapies for hidradenitis suppurativa: a retrospective chart review of 31 patients. J Cutan Med Surg. 2019;23:270-276. doi:10.1177/1203475418823529
- Loh TY, Hendricks AJ, Hsiao JL, et al. Undergarment and fabric selection in the management of hidradenitis suppurativa. Dermatol Basel Switz. 2021;237:119-124. doi:10.1159/000501611
- Garg A, Malviya N, Strunk A, et al. Comorbidity screening in hidradenitis suppurativa: evidence-based recommendations from the US and Canadian Hidradenitis Suppurativa Foundations [published online January 23, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.01.059
- Savage KT, Brant EG, Flood KS, et al. Publication trends in hidradenitis suppurativa from 2008 to 2018. J Eur Acad Dermatol Venereol. 2020;34:1885-1889. doi:10.1111/jdv.16213
- van Straalen KR, Schneider-Burrus S, Prens EP. Current and future treatment of hidradenitis suppurativa. Br J Dermatol. 2020;183:E178-E187. doi:10.1111/bjd.16768
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi:10.1016/j.jaad.2019.02.068
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMoa2102383
- Imafuku S, Nakagawa H, Igarashi A, et al. Long-term efficacy and safety of tildrakizumab in Japanese patients with moderate to severe plaque psoriasis: results from a 5-year extension of a phase 3 study (reSURFACE 1). J Dermatol. 2021;48:844-852. doi:10.1111/1346-8138.15763
- Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434. doi:10.1056/NEJMoa1504370
- McPhie ML, Bridgman AC, Kirchhof MG. Combination therapies for hidradenitis suppurativa: a retrospective chart review of 31 patients. J Cutan Med Surg. 2019;23:270-276. doi:10.1177/1203475418823529
- Loh TY, Hendricks AJ, Hsiao JL, et al. Undergarment and fabric selection in the management of hidradenitis suppurativa. Dermatol Basel Switz. 2021;237:119-124. doi:10.1159/000501611
- Garg A, Malviya N, Strunk A, et al. Comorbidity screening in hidradenitis suppurativa: evidence-based recommendations from the US and Canadian Hidradenitis Suppurativa Foundations [published online January 23, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.01.059
- Savage KT, Brant EG, Flood KS, et al. Publication trends in hidradenitis suppurativa from 2008 to 2018. J Eur Acad Dermatol Venereol. 2020;34:1885-1889. doi:10.1111/jdv.16213
- van Straalen KR, Schneider-Burrus S, Prens EP. Current and future treatment of hidradenitis suppurativa. Br J Dermatol. 2020;183:E178-E187. doi:10.1111/bjd.16768
Psoriasis and Psoriatic Arthritis: A Supplement to Dermatology News
Psoriasis and Psoriatic Arthritis: A Supplement to Dermatology News 2021
- Psoriasis severity redefined by expert group
- Mitigating PSA risk with biologic therapy
- Lack of diversity seen in psoriasis trials
- PSA Comorbidities effect on treatment responses
With Commentaries by Joel M. Gelfand, MD, MSCE and Alan Menter, MD
And more…
Psoriasis and Psoriatic Arthritis: A Supplement to Dermatology News 2021
- Psoriasis severity redefined by expert group
- Mitigating PSA risk with biologic therapy
- Lack of diversity seen in psoriasis trials
- PSA Comorbidities effect on treatment responses
With Commentaries by Joel M. Gelfand, MD, MSCE and Alan Menter, MD
And more…
Psoriasis and Psoriatic Arthritis: A Supplement to Dermatology News 2021
- Psoriasis severity redefined by expert group
- Mitigating PSA risk with biologic therapy
- Lack of diversity seen in psoriasis trials
- PSA Comorbidities effect on treatment responses
With Commentaries by Joel M. Gelfand, MD, MSCE and Alan Menter, MD
And more…
Nonsteroidal topical found effective for psoriasis in 52-week study
Treatment with presented as a late-breaker at the virtual annual congress of the European Academy of Dermatology and Venereology.
The drug has several unique features with meaningful clinical differences from other topical psoriasis therapies, according to Linda Stein Gold, MD, director of dermatology clinical research, Henry Ford Health System, Detroit.
“The currently available nonsteroidal topical therapies are typically associated with significant irritation. We did not see that with tapinarof,” said Dr. Stein Gold. This is one of several reasons she believes this drug will be a valuable addition if it receives regulatory approval.
Tapinarof is a small-molecule aryl hydrocarbon receptor (AhR) modulating agent. AhR is widely expressed in immune cells, including macrophages, mast cells, and antigen-presenting cells. It is believed that modulation of AhR signaling by tapinarof reverses immune dysregulation that is involved in the formation of psoriatic lesions.
The newly presented PSOARING 3 data with tapinarof 1% build on the data from the 12-week PSOARING 1 and PSOARING 2 trials, which were released in August 2020 but have yet to be published.
The primary endpoint in both of the 12-week trials, each of which enrolled about 500 patients with plaque psoriasis, was a Physician Global Assessment (PGA) score of 0 (clear) or 1 (almost clear). Relative to a placebo response rate of about 6% in both trials, the proportion of patients who achieved scores of 0/1 with tapinarof 1% was 35.4% and 40.2% in the PSOARING 1 and PSOARING 2 trials, respectively (P < .0001 vs. placebo in both studies).
For the key secondary endpoint of at least 75% improvement in the Psoriasis Area and Severity Index (PASI 75), the relative advantage for tapinarof over placebo was similar. The results were highly statistically significant (P < .0001) in both of the 12-week trials.
More than 90% of the patients who participated in PSOARING 1 and PSOARING 2 and were eligible for the open-label PSOARING 3 extension trial, according to Dr. Stein Gold.
For the 79 patients with a score of 0 at the time of enrollment, tapinarof 1% was reapplied only if the PGA score reached at least 2 during the course of the study. For the 680 patients who entered with a PGA score of at least 1, once-daily applications of tapinarof 1% cream were maintained until a PGA score of 0 was achieved.
In the outcome analysis, response was defined as the proportion of patients with an initial PGA score of at least2 who achieved PGA 0. A remittive effect was defined as duration of a PGA score of 0 or 1 while off therapy after achieving a PGA score of 0. Durability of response was defined as the proportion of patients who achieved a PGA sore of 0 or 1 at least once during the study while on therapy. This last outcome provided a test of tachyphylaxis.
“Overall, 40.9% of patients achieved complete disease clearance at least once during the trial, and 58.2% who entered the study with a PGA score of 2 or higher achieved a PGA score of 0 or 1,” Dr. Stein Gold reported.
For the 79 patients who entered PSOARING 3 with a PGA score of 0 and were off treatment, the median duration of a remittive effect was 115 days. For the patients who entered the trial with a higher PGA score but who achieved a score of 0 during the study (312 patients), the mean remittive effect after discontinuing therapy was 130 days.
There was no evidence of tachyphylaxis. Rather, “there was no loss of effect despite intermittent therapy observed over the course of the trial,” Dr. Stein Gold reported.
The most common treatment-emergent adverse events in PSOARING 3, as in the previous PSOARING studies, were folliculitis, which was observed in 24.0% of patients; contact dermatitis, which occurred in 5.9% of patients; and headache, which was reported in 2%. Rates of study drug discontinuations for folliculitis and contact dermatitis were 1.2% and 1.4%, respectively. Headache did not lead to any study discontinuations.
Calling tapinarof a “first-in-class nonsteroidal,” Dr. Stein Gold suggested that this is likely to be a useful adjunctive therapy for psoriasis control. It avoids the adverse events associated with long-term topical steroid use, and its tolerability might be particularly attractive for use in sensitive areas.
“This is likely to be very useful in patients who are looking for a topical therapy for skin folds or the face, where there is a need for well-tolerated topical treatments,” Dr. Stein Gold said.
There are a lot of reasons to be positive about a new, well-tolerated topical agent for psoriasis, particularly as an alternative to topical steroids, agreed Adam Friedman, MD, director of translational research and professor and chair of the department of dermatology at George Washington University, Washington. He considers the data with tapinarof promising in general, but he also likes any new, effective topical psoriasis therapy.
“Patients and physicians are always hungry for new options, especially psoriasis patients, given many have ‘been there and done that’ with topical steroids,” Dr. Friedman said.
“Topical steroids are not irritating, but long-term use beyond recommended dosing can lead to skin thinning, lightening, tachyphylaxis, and, if really abused, HPA [hypothalamic-pituitary-adrenal] axis suppression and adrenal insufficiency,” he observed.
A topical therapy with a durable effect is particularly intriguing.
“The other issue with topical steroids is that psoriatic plaques return rather easily after stopping. The data I have seen with tapinarof show more sustainability after cessation, owing to its mechanism of action,” Dr. Friedman said. Rather than its potential for application to sensitive areas, such as the face, the durability “to me is more interesting.”
He suspects that, owing to “the incurable steroid phobia that haunts many of our patients,” an effective nonsteroidal topical option is also likely to lead to better compliance with topical treatment over time.
“A well-tolerated nonsteroidal topical drug will probably find an important place in the future management of chronic inflammatory diseases,” Marius-Anton Ionescu, MD, PhD, a dermatologist at the Hôpital Saint Louis, Paris, said in an interview. He referred to the positive effects of treatment with tapinarof in clinical trials in adults with atopic dermatitis, in addition to psoriasis.
Tapinarof 1% is also being investigated in a phase 3 study involving patients with moderate to severe atopic dermatitis. In that study, patients are as young as age 2 years. The drug is under review at the Food and Drug Administration for the plaque psoriasis indication in adults.
Dr. Stein Gold has financial relationships with Arcutis, Amgen, Bristol-Myers Squibb, Eli Lilly, Leo Pharma Ortho Dermatologic, UCB, and Dermavant Sciences, which is developing tapinarof and is provided funding for the PSOARING 3 trial. Dr. Friedman reported financial relationships with Amgen, Biogen, Encore, Galderma, GlaxoSmithKline, IntraDerm, Johnson & Johnson, Nerium, Novartis, Oculus, Onset, Pfizer, Sanova, and Valeant Pharmaceuticals. Dr. Ionescu has been a speaker or investigator (honoraria) for Celgene, Novartis, Lilly, and Uriage Cosmetics.
A version of this article first appeared on Medscape.com.
Treatment with presented as a late-breaker at the virtual annual congress of the European Academy of Dermatology and Venereology.
The drug has several unique features with meaningful clinical differences from other topical psoriasis therapies, according to Linda Stein Gold, MD, director of dermatology clinical research, Henry Ford Health System, Detroit.
“The currently available nonsteroidal topical therapies are typically associated with significant irritation. We did not see that with tapinarof,” said Dr. Stein Gold. This is one of several reasons she believes this drug will be a valuable addition if it receives regulatory approval.
Tapinarof is a small-molecule aryl hydrocarbon receptor (AhR) modulating agent. AhR is widely expressed in immune cells, including macrophages, mast cells, and antigen-presenting cells. It is believed that modulation of AhR signaling by tapinarof reverses immune dysregulation that is involved in the formation of psoriatic lesions.
The newly presented PSOARING 3 data with tapinarof 1% build on the data from the 12-week PSOARING 1 and PSOARING 2 trials, which were released in August 2020 but have yet to be published.
The primary endpoint in both of the 12-week trials, each of which enrolled about 500 patients with plaque psoriasis, was a Physician Global Assessment (PGA) score of 0 (clear) or 1 (almost clear). Relative to a placebo response rate of about 6% in both trials, the proportion of patients who achieved scores of 0/1 with tapinarof 1% was 35.4% and 40.2% in the PSOARING 1 and PSOARING 2 trials, respectively (P < .0001 vs. placebo in both studies).
For the key secondary endpoint of at least 75% improvement in the Psoriasis Area and Severity Index (PASI 75), the relative advantage for tapinarof over placebo was similar. The results were highly statistically significant (P < .0001) in both of the 12-week trials.
More than 90% of the patients who participated in PSOARING 1 and PSOARING 2 and were eligible for the open-label PSOARING 3 extension trial, according to Dr. Stein Gold.
For the 79 patients with a score of 0 at the time of enrollment, tapinarof 1% was reapplied only if the PGA score reached at least 2 during the course of the study. For the 680 patients who entered with a PGA score of at least 1, once-daily applications of tapinarof 1% cream were maintained until a PGA score of 0 was achieved.
In the outcome analysis, response was defined as the proportion of patients with an initial PGA score of at least2 who achieved PGA 0. A remittive effect was defined as duration of a PGA score of 0 or 1 while off therapy after achieving a PGA score of 0. Durability of response was defined as the proportion of patients who achieved a PGA sore of 0 or 1 at least once during the study while on therapy. This last outcome provided a test of tachyphylaxis.
“Overall, 40.9% of patients achieved complete disease clearance at least once during the trial, and 58.2% who entered the study with a PGA score of 2 or higher achieved a PGA score of 0 or 1,” Dr. Stein Gold reported.
For the 79 patients who entered PSOARING 3 with a PGA score of 0 and were off treatment, the median duration of a remittive effect was 115 days. For the patients who entered the trial with a higher PGA score but who achieved a score of 0 during the study (312 patients), the mean remittive effect after discontinuing therapy was 130 days.
There was no evidence of tachyphylaxis. Rather, “there was no loss of effect despite intermittent therapy observed over the course of the trial,” Dr. Stein Gold reported.
The most common treatment-emergent adverse events in PSOARING 3, as in the previous PSOARING studies, were folliculitis, which was observed in 24.0% of patients; contact dermatitis, which occurred in 5.9% of patients; and headache, which was reported in 2%. Rates of study drug discontinuations for folliculitis and contact dermatitis were 1.2% and 1.4%, respectively. Headache did not lead to any study discontinuations.
Calling tapinarof a “first-in-class nonsteroidal,” Dr. Stein Gold suggested that this is likely to be a useful adjunctive therapy for psoriasis control. It avoids the adverse events associated with long-term topical steroid use, and its tolerability might be particularly attractive for use in sensitive areas.
“This is likely to be very useful in patients who are looking for a topical therapy for skin folds or the face, where there is a need for well-tolerated topical treatments,” Dr. Stein Gold said.
There are a lot of reasons to be positive about a new, well-tolerated topical agent for psoriasis, particularly as an alternative to topical steroids, agreed Adam Friedman, MD, director of translational research and professor and chair of the department of dermatology at George Washington University, Washington. He considers the data with tapinarof promising in general, but he also likes any new, effective topical psoriasis therapy.
“Patients and physicians are always hungry for new options, especially psoriasis patients, given many have ‘been there and done that’ with topical steroids,” Dr. Friedman said.
“Topical steroids are not irritating, but long-term use beyond recommended dosing can lead to skin thinning, lightening, tachyphylaxis, and, if really abused, HPA [hypothalamic-pituitary-adrenal] axis suppression and adrenal insufficiency,” he observed.
A topical therapy with a durable effect is particularly intriguing.
“The other issue with topical steroids is that psoriatic plaques return rather easily after stopping. The data I have seen with tapinarof show more sustainability after cessation, owing to its mechanism of action,” Dr. Friedman said. Rather than its potential for application to sensitive areas, such as the face, the durability “to me is more interesting.”
He suspects that, owing to “the incurable steroid phobia that haunts many of our patients,” an effective nonsteroidal topical option is also likely to lead to better compliance with topical treatment over time.
“A well-tolerated nonsteroidal topical drug will probably find an important place in the future management of chronic inflammatory diseases,” Marius-Anton Ionescu, MD, PhD, a dermatologist at the Hôpital Saint Louis, Paris, said in an interview. He referred to the positive effects of treatment with tapinarof in clinical trials in adults with atopic dermatitis, in addition to psoriasis.
Tapinarof 1% is also being investigated in a phase 3 study involving patients with moderate to severe atopic dermatitis. In that study, patients are as young as age 2 years. The drug is under review at the Food and Drug Administration for the plaque psoriasis indication in adults.
Dr. Stein Gold has financial relationships with Arcutis, Amgen, Bristol-Myers Squibb, Eli Lilly, Leo Pharma Ortho Dermatologic, UCB, and Dermavant Sciences, which is developing tapinarof and is provided funding for the PSOARING 3 trial. Dr. Friedman reported financial relationships with Amgen, Biogen, Encore, Galderma, GlaxoSmithKline, IntraDerm, Johnson & Johnson, Nerium, Novartis, Oculus, Onset, Pfizer, Sanova, and Valeant Pharmaceuticals. Dr. Ionescu has been a speaker or investigator (honoraria) for Celgene, Novartis, Lilly, and Uriage Cosmetics.
A version of this article first appeared on Medscape.com.
Treatment with presented as a late-breaker at the virtual annual congress of the European Academy of Dermatology and Venereology.
The drug has several unique features with meaningful clinical differences from other topical psoriasis therapies, according to Linda Stein Gold, MD, director of dermatology clinical research, Henry Ford Health System, Detroit.
“The currently available nonsteroidal topical therapies are typically associated with significant irritation. We did not see that with tapinarof,” said Dr. Stein Gold. This is one of several reasons she believes this drug will be a valuable addition if it receives regulatory approval.
Tapinarof is a small-molecule aryl hydrocarbon receptor (AhR) modulating agent. AhR is widely expressed in immune cells, including macrophages, mast cells, and antigen-presenting cells. It is believed that modulation of AhR signaling by tapinarof reverses immune dysregulation that is involved in the formation of psoriatic lesions.
The newly presented PSOARING 3 data with tapinarof 1% build on the data from the 12-week PSOARING 1 and PSOARING 2 trials, which were released in August 2020 but have yet to be published.
The primary endpoint in both of the 12-week trials, each of which enrolled about 500 patients with plaque psoriasis, was a Physician Global Assessment (PGA) score of 0 (clear) or 1 (almost clear). Relative to a placebo response rate of about 6% in both trials, the proportion of patients who achieved scores of 0/1 with tapinarof 1% was 35.4% and 40.2% in the PSOARING 1 and PSOARING 2 trials, respectively (P < .0001 vs. placebo in both studies).
For the key secondary endpoint of at least 75% improvement in the Psoriasis Area and Severity Index (PASI 75), the relative advantage for tapinarof over placebo was similar. The results were highly statistically significant (P < .0001) in both of the 12-week trials.
More than 90% of the patients who participated in PSOARING 1 and PSOARING 2 and were eligible for the open-label PSOARING 3 extension trial, according to Dr. Stein Gold.
For the 79 patients with a score of 0 at the time of enrollment, tapinarof 1% was reapplied only if the PGA score reached at least 2 during the course of the study. For the 680 patients who entered with a PGA score of at least 1, once-daily applications of tapinarof 1% cream were maintained until a PGA score of 0 was achieved.
In the outcome analysis, response was defined as the proportion of patients with an initial PGA score of at least2 who achieved PGA 0. A remittive effect was defined as duration of a PGA score of 0 or 1 while off therapy after achieving a PGA score of 0. Durability of response was defined as the proportion of patients who achieved a PGA sore of 0 or 1 at least once during the study while on therapy. This last outcome provided a test of tachyphylaxis.
“Overall, 40.9% of patients achieved complete disease clearance at least once during the trial, and 58.2% who entered the study with a PGA score of 2 or higher achieved a PGA score of 0 or 1,” Dr. Stein Gold reported.
For the 79 patients who entered PSOARING 3 with a PGA score of 0 and were off treatment, the median duration of a remittive effect was 115 days. For the patients who entered the trial with a higher PGA score but who achieved a score of 0 during the study (312 patients), the mean remittive effect after discontinuing therapy was 130 days.
There was no evidence of tachyphylaxis. Rather, “there was no loss of effect despite intermittent therapy observed over the course of the trial,” Dr. Stein Gold reported.
The most common treatment-emergent adverse events in PSOARING 3, as in the previous PSOARING studies, were folliculitis, which was observed in 24.0% of patients; contact dermatitis, which occurred in 5.9% of patients; and headache, which was reported in 2%. Rates of study drug discontinuations for folliculitis and contact dermatitis were 1.2% and 1.4%, respectively. Headache did not lead to any study discontinuations.
Calling tapinarof a “first-in-class nonsteroidal,” Dr. Stein Gold suggested that this is likely to be a useful adjunctive therapy for psoriasis control. It avoids the adverse events associated with long-term topical steroid use, and its tolerability might be particularly attractive for use in sensitive areas.
“This is likely to be very useful in patients who are looking for a topical therapy for skin folds or the face, where there is a need for well-tolerated topical treatments,” Dr. Stein Gold said.
There are a lot of reasons to be positive about a new, well-tolerated topical agent for psoriasis, particularly as an alternative to topical steroids, agreed Adam Friedman, MD, director of translational research and professor and chair of the department of dermatology at George Washington University, Washington. He considers the data with tapinarof promising in general, but he also likes any new, effective topical psoriasis therapy.
“Patients and physicians are always hungry for new options, especially psoriasis patients, given many have ‘been there and done that’ with topical steroids,” Dr. Friedman said.
“Topical steroids are not irritating, but long-term use beyond recommended dosing can lead to skin thinning, lightening, tachyphylaxis, and, if really abused, HPA [hypothalamic-pituitary-adrenal] axis suppression and adrenal insufficiency,” he observed.
A topical therapy with a durable effect is particularly intriguing.
“The other issue with topical steroids is that psoriatic plaques return rather easily after stopping. The data I have seen with tapinarof show more sustainability after cessation, owing to its mechanism of action,” Dr. Friedman said. Rather than its potential for application to sensitive areas, such as the face, the durability “to me is more interesting.”
He suspects that, owing to “the incurable steroid phobia that haunts many of our patients,” an effective nonsteroidal topical option is also likely to lead to better compliance with topical treatment over time.
“A well-tolerated nonsteroidal topical drug will probably find an important place in the future management of chronic inflammatory diseases,” Marius-Anton Ionescu, MD, PhD, a dermatologist at the Hôpital Saint Louis, Paris, said in an interview. He referred to the positive effects of treatment with tapinarof in clinical trials in adults with atopic dermatitis, in addition to psoriasis.
Tapinarof 1% is also being investigated in a phase 3 study involving patients with moderate to severe atopic dermatitis. In that study, patients are as young as age 2 years. The drug is under review at the Food and Drug Administration for the plaque psoriasis indication in adults.
Dr. Stein Gold has financial relationships with Arcutis, Amgen, Bristol-Myers Squibb, Eli Lilly, Leo Pharma Ortho Dermatologic, UCB, and Dermavant Sciences, which is developing tapinarof and is provided funding for the PSOARING 3 trial. Dr. Friedman reported financial relationships with Amgen, Biogen, Encore, Galderma, GlaxoSmithKline, IntraDerm, Johnson & Johnson, Nerium, Novartis, Oculus, Onset, Pfizer, Sanova, and Valeant Pharmaceuticals. Dr. Ionescu has been a speaker or investigator (honoraria) for Celgene, Novartis, Lilly, and Uriage Cosmetics.
A version of this article first appeared on Medscape.com.
Clinical Edge Journal Scan Commentary: Psoriasis October 2021
High-potency topical steroids such as clobetasol are commonly used as first-line treatment for psoriasis. A recent study (Sidgiddi S et al. Dermatol Ther (Heidelb). 2021 Aug 28) compared the efficacy and safety of clobetasol 0.05% vs. 0.025% cream. Patients were randomized to receive either clobetasol 0.05% cream or one of two different formulations of clobetasol 0.025% cream twice a day for two weeks. PGA success rates (clear or almost clear skin) were higher with the 0.025% formulations (38.9% and 36.8%) than with the 0.05% cream (30.8%). Safety also appeared to be better or comparable with the 0.025% formulation as measured by the proportion of patients with an abnormal ACTH stimulation test (20.7% and 17.2% in the 0.025% group compared with 30.0% in the 0.05% group). Due to the small study size (88 subjects) these differences did not reach statistical significance, although they suggest that high efficacy and perhaps better safety (reduced hypothalamic–pituitary–adrenal axis suppression) can be achieved with lower concentration formulations of clobetasol cream.
The oral phosphodiesterase 4 inhibitor apremilast is FDA-approved to treat psoriasis and psoriatic arthritis and recent studies have shown that it is more effective than placebo in treating patients with mild-moderate psoriasis and scalp psoriasis. A recent prospective cohort study followed 45 adult patients with plaque and nail psoriasis with a fingernail Nail Psoriasis Severity Index (NAPSI) score of 12 or more treated with apremilast 30 mg twice daily for 52 weeks (Muñoz-Santos C et al. J Dermatol. 2021 Aug 12). The primary endpoint, the percentage of patients with a Nail Assessment in Psoriasis and Psoriatic Arthritis-Patient Benefit Index of 2 or more at week 52, was achieved in 52% of patients. A median improvement of 53% in fingernail NAPSI score and a mean reduction in nail pain of 68% were observed at week 52. These findings show that apremilast can be useful in improving the quality-of-life impairment caused by nail psoriasis.
Traditional systemic therapies such as methotrexate and acitretin are cost-effective options for many psoriasis patients. The association of psoriasis with other comorbid conditions, particularly cardiovascular disease, has raised awareness of the importance of considering how therapies impact not just skin disease but also the risk posed by these psoriasis-associated comorbidities. A large Taiwanese retrospective cohort study compared patients treated with methotrexate (13,777) or acitretin (6,020) and found that in comparison to those treated with acitretin, patients treated with methotrexate were at lower risk of experiencing adverse cardiovascular outcomes, including ischemic heart disease and stroke, (adjusted hazard ratio [aHR], 0.84; 95% confidence interval [CI], 0.76-0.94) and had a lower risk of all-cause mortality (aHR, 0.75; 95% CI, 0.66-0.85).
Many disease and patient factors must be considered when choosing the right therapy for a patient. These studies provide valuable information to incorporate into this process and highlight the utility of topical and oral therapies for psoriasis.
High-potency topical steroids such as clobetasol are commonly used as first-line treatment for psoriasis. A recent study (Sidgiddi S et al. Dermatol Ther (Heidelb). 2021 Aug 28) compared the efficacy and safety of clobetasol 0.05% vs. 0.025% cream. Patients were randomized to receive either clobetasol 0.05% cream or one of two different formulations of clobetasol 0.025% cream twice a day for two weeks. PGA success rates (clear or almost clear skin) were higher with the 0.025% formulations (38.9% and 36.8%) than with the 0.05% cream (30.8%). Safety also appeared to be better or comparable with the 0.025% formulation as measured by the proportion of patients with an abnormal ACTH stimulation test (20.7% and 17.2% in the 0.025% group compared with 30.0% in the 0.05% group). Due to the small study size (88 subjects) these differences did not reach statistical significance, although they suggest that high efficacy and perhaps better safety (reduced hypothalamic–pituitary–adrenal axis suppression) can be achieved with lower concentration formulations of clobetasol cream.
The oral phosphodiesterase 4 inhibitor apremilast is FDA-approved to treat psoriasis and psoriatic arthritis and recent studies have shown that it is more effective than placebo in treating patients with mild-moderate psoriasis and scalp psoriasis. A recent prospective cohort study followed 45 adult patients with plaque and nail psoriasis with a fingernail Nail Psoriasis Severity Index (NAPSI) score of 12 or more treated with apremilast 30 mg twice daily for 52 weeks (Muñoz-Santos C et al. J Dermatol. 2021 Aug 12). The primary endpoint, the percentage of patients with a Nail Assessment in Psoriasis and Psoriatic Arthritis-Patient Benefit Index of 2 or more at week 52, was achieved in 52% of patients. A median improvement of 53% in fingernail NAPSI score and a mean reduction in nail pain of 68% were observed at week 52. These findings show that apremilast can be useful in improving the quality-of-life impairment caused by nail psoriasis.
Traditional systemic therapies such as methotrexate and acitretin are cost-effective options for many psoriasis patients. The association of psoriasis with other comorbid conditions, particularly cardiovascular disease, has raised awareness of the importance of considering how therapies impact not just skin disease but also the risk posed by these psoriasis-associated comorbidities. A large Taiwanese retrospective cohort study compared patients treated with methotrexate (13,777) or acitretin (6,020) and found that in comparison to those treated with acitretin, patients treated with methotrexate were at lower risk of experiencing adverse cardiovascular outcomes, including ischemic heart disease and stroke, (adjusted hazard ratio [aHR], 0.84; 95% confidence interval [CI], 0.76-0.94) and had a lower risk of all-cause mortality (aHR, 0.75; 95% CI, 0.66-0.85).
Many disease and patient factors must be considered when choosing the right therapy for a patient. These studies provide valuable information to incorporate into this process and highlight the utility of topical and oral therapies for psoriasis.
High-potency topical steroids such as clobetasol are commonly used as first-line treatment for psoriasis. A recent study (Sidgiddi S et al. Dermatol Ther (Heidelb). 2021 Aug 28) compared the efficacy and safety of clobetasol 0.05% vs. 0.025% cream. Patients were randomized to receive either clobetasol 0.05% cream or one of two different formulations of clobetasol 0.025% cream twice a day for two weeks. PGA success rates (clear or almost clear skin) were higher with the 0.025% formulations (38.9% and 36.8%) than with the 0.05% cream (30.8%). Safety also appeared to be better or comparable with the 0.025% formulation as measured by the proportion of patients with an abnormal ACTH stimulation test (20.7% and 17.2% in the 0.025% group compared with 30.0% in the 0.05% group). Due to the small study size (88 subjects) these differences did not reach statistical significance, although they suggest that high efficacy and perhaps better safety (reduced hypothalamic–pituitary–adrenal axis suppression) can be achieved with lower concentration formulations of clobetasol cream.
The oral phosphodiesterase 4 inhibitor apremilast is FDA-approved to treat psoriasis and psoriatic arthritis and recent studies have shown that it is more effective than placebo in treating patients with mild-moderate psoriasis and scalp psoriasis. A recent prospective cohort study followed 45 adult patients with plaque and nail psoriasis with a fingernail Nail Psoriasis Severity Index (NAPSI) score of 12 or more treated with apremilast 30 mg twice daily for 52 weeks (Muñoz-Santos C et al. J Dermatol. 2021 Aug 12). The primary endpoint, the percentage of patients with a Nail Assessment in Psoriasis and Psoriatic Arthritis-Patient Benefit Index of 2 or more at week 52, was achieved in 52% of patients. A median improvement of 53% in fingernail NAPSI score and a mean reduction in nail pain of 68% were observed at week 52. These findings show that apremilast can be useful in improving the quality-of-life impairment caused by nail psoriasis.
Traditional systemic therapies such as methotrexate and acitretin are cost-effective options for many psoriasis patients. The association of psoriasis with other comorbid conditions, particularly cardiovascular disease, has raised awareness of the importance of considering how therapies impact not just skin disease but also the risk posed by these psoriasis-associated comorbidities. A large Taiwanese retrospective cohort study compared patients treated with methotrexate (13,777) or acitretin (6,020) and found that in comparison to those treated with acitretin, patients treated with methotrexate were at lower risk of experiencing adverse cardiovascular outcomes, including ischemic heart disease and stroke, (adjusted hazard ratio [aHR], 0.84; 95% confidence interval [CI], 0.76-0.94) and had a lower risk of all-cause mortality (aHR, 0.75; 95% CI, 0.66-0.85).
Many disease and patient factors must be considered when choosing the right therapy for a patient. These studies provide valuable information to incorporate into this process and highlight the utility of topical and oral therapies for psoriasis.
First-in-class TYK inhibitor shows durable effect for psoriasis
of follow-up, according to late-breaking data from two pivotal trials presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
From benefit reported on the two coprimary endpoints previously reported at 16 weeks, longer follow-up showed further gains out to 24 weeks and then persistent efficacy out to 52 weeks across these and multiple secondary endpoints, reported Richard Warren, MBChB, PhD, professor of dermatology and therapeutics, University of Manchester (England).
“This could be a unique oral therapy and an important treatment option for moderate to severe psoriasis,” Dr. Warren contended.
The multinational double-blind trials, called POETYK PSO-1 and PSO-2, enrolled 666 and 1,020 patients, respectively. The designs were similar. Patients with moderate to severe plaque psoriasis were randomly assigned in a 2:1:1 ratio to deucravacitinib (6 mg once daily), placebo, or apremilast (Otezla; 30 mg twice daily). At 16 weeks, those on placebo were switched to deucravacitinib.
For the coprimary endpoint of PASI 75 (75% clearance on the Psoriasis and Severity Index), the similar rate of response for deucravacitinib in the two studies (58.7%/53.6%) at week 16 was superior to the rates observed on both apremilast (35.1%/40.2%) and placebo (12.7%/9.4%).
By week 24, the proportion of deucravacitinib patients with a PASI 75 response had reached 69.3% and 58.7% in the POETYK PSO-1 and PSO-2 trials, respectively. The proportion of patients on apremilast with PASI 75 at this time point did not increase appreciably in one study and fell modestly in the other.
By week 52, the response rates achieved with deucravacitinib at week 24 were generally unchanged and nearly double those observed on apremilast.
The pattern of relative benefit on the other coprimary endpoint, which was a score of 0 or 1, signifying clear or almost clear skin on the static Physicians Global Assessment (sPGA), followed the same pattern. At week 16, 53.6% of patients had achieved sPGA 0/1. This was significantly higher than that observed on either apremilast or placebo, and this level of response was sustained through week 52.
When patients on placebo were switched to deucravacitinib at week 16, the PASI 75 response climbed quickly. There was complete catch-up by 32 weeks. In both groups, a PASI 75 response rate of about 65% or higher was maintained for the remainder of the study.
On a prespecified analysis, prior treatment exposure was not associated with any impact on the degree of response with deucravacitinib. This included a comparison between patients exposed to no prior biologic, one prior biologic, or two or more biologics, Dr. Warren reported.
Unlike patients in POETYK PSO-1, those with a PASI 75 response at 16 weeks in the POETYK PSO-2 trial were rerandomized to remain on deucravacitinib or switch to placebo. Designed to evaluate response durability, this analysis showed a relatively gradual decline in disease control.
“The median time to a loss of response was 12 weeks,” Dr. Warren said. He was referring in this case to the PASI 75 response, but the slope of decline was similar for sPGA score 0/1. At the end of 52 weeks, 31.3% of patients who had been rerandomized to placebo still maintained a PASI 75 while 80.4% of those who stayed on deucravacitinib still had PASI 75 clearance.
In the 52-week data from these two trials, several secondary endpoints have already been examined, and Dr. Warren said more analyses are coming. So far, the pattern of response has been similar for all endpoints.
Reporting on one as an example, Dr. Warren said that sPGA 0/1 for scalp psoriasis was achieved at week 16 by 70.3% of those randomly assigned to deucravacitinib versus 17.4% of those in the placebo arm. Among those switched from placebo to deucravacitinib at 16 weeks, the scalp response had caught up to that observed in those initiated on deucravacitinib by week 28. The response was sustained out to 52 weeks in both groups.
In the long-term trials, there have been no new safety concerns, according to Dr. Warren. He described this drug as “well tolerated,” adding that no significant laboratory abnormalities have been observed on long-term treatment. Although there has been a trend for increased risk of viral infections, such as herpes zoster, relative to apremilast, cases have so far been mild.
The Janus kinase inhibitor tofacitinib (Xeljanz, Xeljanz XR) has been approved for psoriatic arthritis, and numerous other JAK inhibitors are now in clinical trials for plaque psoriasis. These agents vary for their relative selectivity for JAK1, 2, and 3 kinases, but deucravacitinib is the first JAK inhibitor to reach clinical trials that target TYK2, which inhibits interleukin-23 and other cytokines implicated in the pathogenesis of plaque psoriasis.
“Deucravacitinib is very distinct from the other JAK inhibitors, and I think we are seeing this in the clinical studies,” Dr. Warren said. As a result of responses in the POETYK PRO trials that rival those achieved with monoclonal antibodies, he expects this drug, if approved, to be an important option for those with moderate to severe disease who prefer oral therapies.
Mark G. Lebwohl, MD, professor of dermatology and dean for clinical therapeutics, Icahn School of Medicine at Mount Sinai, New York, shares this opinion. In an interview, he emphasized the unique mechanism of deucravacitinib and its clinical potential.
“Unlike other less specific JAK inhibitors, deucravacitinib has a unique binding site on TYK2, the regulatory domain of the molecule. This makes deucravacitinib more targeted and therefore safer than other JAK inhibitors,” said Dr. Lebwohl.
“After cyclosporine, which has many side effects, deucravacitinib is the most effective oral therapy we have for psoriasis and one of the safest,” he added.
The POETYK PSO-1 and PSO-2 trials received funding from Bristol-Myers Squibb. Dr. Warren has financial relationships with AbbVie, Almirall, Boehringer Ingelheim, Celgene, Eli Lilly, Janssen, Leo Pharma, Novartis, Pfizer, Sanofi, UCB, and Xenoport. Dr. Lebwohl has financial relationships with more than 20 pharmaceutical companies, including Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
of follow-up, according to late-breaking data from two pivotal trials presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
From benefit reported on the two coprimary endpoints previously reported at 16 weeks, longer follow-up showed further gains out to 24 weeks and then persistent efficacy out to 52 weeks across these and multiple secondary endpoints, reported Richard Warren, MBChB, PhD, professor of dermatology and therapeutics, University of Manchester (England).
“This could be a unique oral therapy and an important treatment option for moderate to severe psoriasis,” Dr. Warren contended.
The multinational double-blind trials, called POETYK PSO-1 and PSO-2, enrolled 666 and 1,020 patients, respectively. The designs were similar. Patients with moderate to severe plaque psoriasis were randomly assigned in a 2:1:1 ratio to deucravacitinib (6 mg once daily), placebo, or apremilast (Otezla; 30 mg twice daily). At 16 weeks, those on placebo were switched to deucravacitinib.
For the coprimary endpoint of PASI 75 (75% clearance on the Psoriasis and Severity Index), the similar rate of response for deucravacitinib in the two studies (58.7%/53.6%) at week 16 was superior to the rates observed on both apremilast (35.1%/40.2%) and placebo (12.7%/9.4%).
By week 24, the proportion of deucravacitinib patients with a PASI 75 response had reached 69.3% and 58.7% in the POETYK PSO-1 and PSO-2 trials, respectively. The proportion of patients on apremilast with PASI 75 at this time point did not increase appreciably in one study and fell modestly in the other.
By week 52, the response rates achieved with deucravacitinib at week 24 were generally unchanged and nearly double those observed on apremilast.
The pattern of relative benefit on the other coprimary endpoint, which was a score of 0 or 1, signifying clear or almost clear skin on the static Physicians Global Assessment (sPGA), followed the same pattern. At week 16, 53.6% of patients had achieved sPGA 0/1. This was significantly higher than that observed on either apremilast or placebo, and this level of response was sustained through week 52.
When patients on placebo were switched to deucravacitinib at week 16, the PASI 75 response climbed quickly. There was complete catch-up by 32 weeks. In both groups, a PASI 75 response rate of about 65% or higher was maintained for the remainder of the study.
On a prespecified analysis, prior treatment exposure was not associated with any impact on the degree of response with deucravacitinib. This included a comparison between patients exposed to no prior biologic, one prior biologic, or two or more biologics, Dr. Warren reported.
Unlike patients in POETYK PSO-1, those with a PASI 75 response at 16 weeks in the POETYK PSO-2 trial were rerandomized to remain on deucravacitinib or switch to placebo. Designed to evaluate response durability, this analysis showed a relatively gradual decline in disease control.
“The median time to a loss of response was 12 weeks,” Dr. Warren said. He was referring in this case to the PASI 75 response, but the slope of decline was similar for sPGA score 0/1. At the end of 52 weeks, 31.3% of patients who had been rerandomized to placebo still maintained a PASI 75 while 80.4% of those who stayed on deucravacitinib still had PASI 75 clearance.
In the 52-week data from these two trials, several secondary endpoints have already been examined, and Dr. Warren said more analyses are coming. So far, the pattern of response has been similar for all endpoints.
Reporting on one as an example, Dr. Warren said that sPGA 0/1 for scalp psoriasis was achieved at week 16 by 70.3% of those randomly assigned to deucravacitinib versus 17.4% of those in the placebo arm. Among those switched from placebo to deucravacitinib at 16 weeks, the scalp response had caught up to that observed in those initiated on deucravacitinib by week 28. The response was sustained out to 52 weeks in both groups.
In the long-term trials, there have been no new safety concerns, according to Dr. Warren. He described this drug as “well tolerated,” adding that no significant laboratory abnormalities have been observed on long-term treatment. Although there has been a trend for increased risk of viral infections, such as herpes zoster, relative to apremilast, cases have so far been mild.
The Janus kinase inhibitor tofacitinib (Xeljanz, Xeljanz XR) has been approved for psoriatic arthritis, and numerous other JAK inhibitors are now in clinical trials for plaque psoriasis. These agents vary for their relative selectivity for JAK1, 2, and 3 kinases, but deucravacitinib is the first JAK inhibitor to reach clinical trials that target TYK2, which inhibits interleukin-23 and other cytokines implicated in the pathogenesis of plaque psoriasis.
“Deucravacitinib is very distinct from the other JAK inhibitors, and I think we are seeing this in the clinical studies,” Dr. Warren said. As a result of responses in the POETYK PRO trials that rival those achieved with monoclonal antibodies, he expects this drug, if approved, to be an important option for those with moderate to severe disease who prefer oral therapies.
Mark G. Lebwohl, MD, professor of dermatology and dean for clinical therapeutics, Icahn School of Medicine at Mount Sinai, New York, shares this opinion. In an interview, he emphasized the unique mechanism of deucravacitinib and its clinical potential.
“Unlike other less specific JAK inhibitors, deucravacitinib has a unique binding site on TYK2, the regulatory domain of the molecule. This makes deucravacitinib more targeted and therefore safer than other JAK inhibitors,” said Dr. Lebwohl.
“After cyclosporine, which has many side effects, deucravacitinib is the most effective oral therapy we have for psoriasis and one of the safest,” he added.
The POETYK PSO-1 and PSO-2 trials received funding from Bristol-Myers Squibb. Dr. Warren has financial relationships with AbbVie, Almirall, Boehringer Ingelheim, Celgene, Eli Lilly, Janssen, Leo Pharma, Novartis, Pfizer, Sanofi, UCB, and Xenoport. Dr. Lebwohl has financial relationships with more than 20 pharmaceutical companies, including Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
of follow-up, according to late-breaking data from two pivotal trials presented at the virtual annual congress of the European Academy of Dermatology and Venereology.
From benefit reported on the two coprimary endpoints previously reported at 16 weeks, longer follow-up showed further gains out to 24 weeks and then persistent efficacy out to 52 weeks across these and multiple secondary endpoints, reported Richard Warren, MBChB, PhD, professor of dermatology and therapeutics, University of Manchester (England).
“This could be a unique oral therapy and an important treatment option for moderate to severe psoriasis,” Dr. Warren contended.
The multinational double-blind trials, called POETYK PSO-1 and PSO-2, enrolled 666 and 1,020 patients, respectively. The designs were similar. Patients with moderate to severe plaque psoriasis were randomly assigned in a 2:1:1 ratio to deucravacitinib (6 mg once daily), placebo, or apremilast (Otezla; 30 mg twice daily). At 16 weeks, those on placebo were switched to deucravacitinib.
For the coprimary endpoint of PASI 75 (75% clearance on the Psoriasis and Severity Index), the similar rate of response for deucravacitinib in the two studies (58.7%/53.6%) at week 16 was superior to the rates observed on both apremilast (35.1%/40.2%) and placebo (12.7%/9.4%).
By week 24, the proportion of deucravacitinib patients with a PASI 75 response had reached 69.3% and 58.7% in the POETYK PSO-1 and PSO-2 trials, respectively. The proportion of patients on apremilast with PASI 75 at this time point did not increase appreciably in one study and fell modestly in the other.
By week 52, the response rates achieved with deucravacitinib at week 24 were generally unchanged and nearly double those observed on apremilast.
The pattern of relative benefit on the other coprimary endpoint, which was a score of 0 or 1, signifying clear or almost clear skin on the static Physicians Global Assessment (sPGA), followed the same pattern. At week 16, 53.6% of patients had achieved sPGA 0/1. This was significantly higher than that observed on either apremilast or placebo, and this level of response was sustained through week 52.
When patients on placebo were switched to deucravacitinib at week 16, the PASI 75 response climbed quickly. There was complete catch-up by 32 weeks. In both groups, a PASI 75 response rate of about 65% or higher was maintained for the remainder of the study.
On a prespecified analysis, prior treatment exposure was not associated with any impact on the degree of response with deucravacitinib. This included a comparison between patients exposed to no prior biologic, one prior biologic, or two or more biologics, Dr. Warren reported.
Unlike patients in POETYK PSO-1, those with a PASI 75 response at 16 weeks in the POETYK PSO-2 trial were rerandomized to remain on deucravacitinib or switch to placebo. Designed to evaluate response durability, this analysis showed a relatively gradual decline in disease control.
“The median time to a loss of response was 12 weeks,” Dr. Warren said. He was referring in this case to the PASI 75 response, but the slope of decline was similar for sPGA score 0/1. At the end of 52 weeks, 31.3% of patients who had been rerandomized to placebo still maintained a PASI 75 while 80.4% of those who stayed on deucravacitinib still had PASI 75 clearance.
In the 52-week data from these two trials, several secondary endpoints have already been examined, and Dr. Warren said more analyses are coming. So far, the pattern of response has been similar for all endpoints.
Reporting on one as an example, Dr. Warren said that sPGA 0/1 for scalp psoriasis was achieved at week 16 by 70.3% of those randomly assigned to deucravacitinib versus 17.4% of those in the placebo arm. Among those switched from placebo to deucravacitinib at 16 weeks, the scalp response had caught up to that observed in those initiated on deucravacitinib by week 28. The response was sustained out to 52 weeks in both groups.
In the long-term trials, there have been no new safety concerns, according to Dr. Warren. He described this drug as “well tolerated,” adding that no significant laboratory abnormalities have been observed on long-term treatment. Although there has been a trend for increased risk of viral infections, such as herpes zoster, relative to apremilast, cases have so far been mild.
The Janus kinase inhibitor tofacitinib (Xeljanz, Xeljanz XR) has been approved for psoriatic arthritis, and numerous other JAK inhibitors are now in clinical trials for plaque psoriasis. These agents vary for their relative selectivity for JAK1, 2, and 3 kinases, but deucravacitinib is the first JAK inhibitor to reach clinical trials that target TYK2, which inhibits interleukin-23 and other cytokines implicated in the pathogenesis of plaque psoriasis.
“Deucravacitinib is very distinct from the other JAK inhibitors, and I think we are seeing this in the clinical studies,” Dr. Warren said. As a result of responses in the POETYK PRO trials that rival those achieved with monoclonal antibodies, he expects this drug, if approved, to be an important option for those with moderate to severe disease who prefer oral therapies.
Mark G. Lebwohl, MD, professor of dermatology and dean for clinical therapeutics, Icahn School of Medicine at Mount Sinai, New York, shares this opinion. In an interview, he emphasized the unique mechanism of deucravacitinib and its clinical potential.
“Unlike other less specific JAK inhibitors, deucravacitinib has a unique binding site on TYK2, the regulatory domain of the molecule. This makes deucravacitinib more targeted and therefore safer than other JAK inhibitors,” said Dr. Lebwohl.
“After cyclosporine, which has many side effects, deucravacitinib is the most effective oral therapy we have for psoriasis and one of the safest,” he added.
The POETYK PSO-1 and PSO-2 trials received funding from Bristol-Myers Squibb. Dr. Warren has financial relationships with AbbVie, Almirall, Boehringer Ingelheim, Celgene, Eli Lilly, Janssen, Leo Pharma, Novartis, Pfizer, Sanofi, UCB, and Xenoport. Dr. Lebwohl has financial relationships with more than 20 pharmaceutical companies, including Bristol-Myers Squibb.
A version of this article first appeared on Medscape.com.
NIAMS director reflects on her mentors, spotlights research projects underway
After many years at the University of California, San Francisco, Lindsey A. Criswell, MD, MPH, DSc, began a new chapter in February 2021 as the director of the National Institute of Arthritis and Musculoskeletal and Skin Disease, part of the National Institutes of Health. NIH Director Francis S. Collins, MD, PhD, selected her for the post.
“Dr. Criswell has rich experience as a clinician, researcher, and administrator,” Dr. Collins said in a prepared statement. “Her ability to oversee the research program of one of the country’s top research-intensive medical schools, and her expertise in autoimmune diseases, including rheumatoid arthritis and lupus, make her well positioned to direct NIAMS.” Dr. Criswell, a rheumatologist, was named a full professor of medicine at UCSF in 2007 and had served as vice chancellor of research at the university since 2017. She has authored more than 250 peer-reviewed scientific papers, and her efforts have contributed to the identification of more than 30 genes linked to autoimmune disorders. In her first media interview, Dr. Criswell opens up about her mentors, operational challenges posed by the COVID-19 pandemic, and highlights many NIAMS research projects underway.
Who inspired you most early in your career as a physician scientist? I have had great opportunities to work with fabulous mentors. Wallace (Wally) Epstein, MD, was my mentor when I was a rheumatology fellow and junior faculty member at UCSF. He was broadly admired for the breadth of his experience as a clinician and a researcher, and he was noteworthy at that time for his strong support for women and students of color. One of the many things I appreciated about him was his diverse range of interests outside of work, which included cello playing and woodworking.
Another mentor was Ephraim (Eph) Engleman, MD, the first academic rheumatologist in California. Eph continued to see patients beyond the age of 100. Perhaps his most important contributions were his efforts towards advocacy for funding for research and education in rheumatology. A prodigy violinist, he too had a broad range of personal interests.
What research into the genetics and epidemiology of human autoimmune disease that you have been a part of has most surprised you, in term of its ultimate clinical impact? Some of my most rewarding and impactful work has focused on the shared genetic basis of autoimmune diseases. We’ve identified dozens of genes that contribute to the risk and outcome of rheumatoid arthritis, lupus, and other autoimmune disorders. These discoveries regarding shared genes and pathways among such a diverse set of conditions have helped to inform optimal therapeutic target and treatment strategies across multiple diseases. For example, exploration of RA genes and pathways has revealed that approved agents for other conditions, such as cancer, may be appropriately repurposed for the treatment of RA. These are critical observations that have the potential to dramatically accelerate progress in developing new therapies for autoimmune diseases, such as RA.
Did you have much interaction with Stephen I. Katz, MD, PhD, your longtime predecessor who passed away unexpectedly in 2018? If so, what do you remember most about him? I regret that I had very little interaction with Steve, but I am well aware of the impact he had on NIAMS, NIH, and the research enterprise overall. He inspired so many people in a personal way, and I am energized by the legacy that he left behind.
What are your goals for the early part of your tenure as the new director of NIAMS? An important goal is getting to know the NIAMS community and expanding my knowledge of the Institute’s musculoskeletal and skin portfolios. I am also conducting outreach to Institute/Center directors and other NIH leadership to increase opportunities for input and advice. In doing this, I am identifying shared research interests, best practices, and potential partners for possible future collaborations. Another important goal is to increase NIAMS’ visibility within and beyond NIH. Ultimately, I want to contribute to the great work of the Institute and improve the lives of people with rheumatic, musculoskeletal, and skin diseases.
How would you characterize your management style? I like to lead with a flat hierarchy and work collectively to address opportunities and challenges. I value team building and tend to tap a variety of perspectives and expertise at all levels to achieve consensus, where possible.
The Accelerating Medicines Partnership (AMP) program was launched in 2014, with projects in three disease areas including the autoimmune disorders RA and lupus. What are some recent highlights from this program with respect to RA and lupus? AMP RA/SLE was dedicated to identifying promising therapeutic targets for RA and systemic lupus erythematosus. AMP-funded researchers have applied cutting-edge technologies to study cells from the synovial tissues of the joints of people with RA, and from the kidneys of people with lupus nephritis. In 2014, studying tissues in patients where the disease is active was a novel approach, since most research was conducted in mouse models or human blood samples.
The AMP RA/SLE Network developed a rich dataset that is available to the research community. Investigators are now using the data to facilitate RA and lupus research. For example, using AMP data, NIAMS-supported researchers identified potential biomarkers that could help predict an imminent RA flare. Work from another NIAMS-supported group suggests that targeting the regulatory transcription factor HIF-1, which drives inflammation and tissue damage, might be an effective approach for treating renal injury in lupus.
The data generated are accessible to the scientific community through two NIH websites: the database of Genotypes and Phenotypes (dbGaP) and the Immunology Database and Analysis Portal (IMMPORT).
Given the success of AMP RA/SLE, NIH plans to launch an “AMP 2.0” later in 2021. The AMP Autoimmune and Immune-Mediated Diseases (AMP AIM) program will provide an opportunity to leverage the accomplishments of AMP RA/SLE to new conditions, including psoriatic spectrum diseases and Sjögren’s syndrome.
What are some recent highlights from NIAMS-supported research in skin diseases? NIAMS-supported investigators continue to make significant strides in our understanding of skin biology and disease. For example, researchers recently demonstrated that imiquimod, a drug used to treat precancerous skin lesions, can help mouse ear wounds heal without scarring.
Another team addressed the safety and potential benefit of Staphylococcus hominis A9, a bacterium isolated from healthy human skin, as a topical therapy for atopic dermatitis.
Moving forward, AMP AIM will refine and extend the single-cell analysis of tissues to additional diseases, including psoriasis, setting the stage for the discovery of new therapeutic targets for the disease.
How has the COVID-19 pandemic changed the landscape of research, at least for the short term? This is a once-in-a-century pandemic that none of us were fully prepared for. We understand that it has been particularly challenging for women scientists, scientists with young children, and trainees and junior faculty who are at critically important and vulnerable stages of their careers. There isn’t a lab or clinical setting that hasn’t been negatively impacted in some way.
During the pandemic, the NIH instituted administrative flexibilities to support the grantee community, including extensions in time. In addition, the agency has issued several funding opportunities specific to COVID-19, some of which involve NIAMS participation.
What is NIAMS doing to help early/young investigators as well as female investigators and those from minority groups? Structural racism in biomedical research is a heightened concern. Earlier this year, Dr. Collins established the UNITE initiative to address structural racism and promote racial equity and inclusion at the NIH and within the larger biomedical community that we support. NIAMS is fully committed to this effort. One example is the Diversity Supplement Program, which is designed to attract and encourage eligible individuals from underrepresented populations to research careers.
Early-stage investigators are another top priority. In a tribute to the beloved former NIAMS director, NIH recently established the Stephen I. Katz Early Stage Investigator Research Grant Program. The R01 award provides support for a project unrelated to an early investigator’s area of postdoctoral study. (No preliminary data are allowed.) This award mechanism is a unique opportunity for early-stage investigators to take their research in a completely new direction.
Managing work and family life is an important concern, particularly for female investigators. Many NIH grant awards allow for reimbursement of actual, allowable costs incurred for childcare and parental leave. The NIH is exploring initiatives to promote research continuity and retention of eligible investigators facing major life events, such as pregnancy, childbirth, and adoption, at vulnerable career stages.
Who inspires you most in your work today? I am inspired by the ongoing struggles of our patients, junior investigators, and by the committed staff members on my team.
After many years at the University of California, San Francisco, Lindsey A. Criswell, MD, MPH, DSc, began a new chapter in February 2021 as the director of the National Institute of Arthritis and Musculoskeletal and Skin Disease, part of the National Institutes of Health. NIH Director Francis S. Collins, MD, PhD, selected her for the post.
“Dr. Criswell has rich experience as a clinician, researcher, and administrator,” Dr. Collins said in a prepared statement. “Her ability to oversee the research program of one of the country’s top research-intensive medical schools, and her expertise in autoimmune diseases, including rheumatoid arthritis and lupus, make her well positioned to direct NIAMS.” Dr. Criswell, a rheumatologist, was named a full professor of medicine at UCSF in 2007 and had served as vice chancellor of research at the university since 2017. She has authored more than 250 peer-reviewed scientific papers, and her efforts have contributed to the identification of more than 30 genes linked to autoimmune disorders. In her first media interview, Dr. Criswell opens up about her mentors, operational challenges posed by the COVID-19 pandemic, and highlights many NIAMS research projects underway.
Who inspired you most early in your career as a physician scientist? I have had great opportunities to work with fabulous mentors. Wallace (Wally) Epstein, MD, was my mentor when I was a rheumatology fellow and junior faculty member at UCSF. He was broadly admired for the breadth of his experience as a clinician and a researcher, and he was noteworthy at that time for his strong support for women and students of color. One of the many things I appreciated about him was his diverse range of interests outside of work, which included cello playing and woodworking.
Another mentor was Ephraim (Eph) Engleman, MD, the first academic rheumatologist in California. Eph continued to see patients beyond the age of 100. Perhaps his most important contributions were his efforts towards advocacy for funding for research and education in rheumatology. A prodigy violinist, he too had a broad range of personal interests.
What research into the genetics and epidemiology of human autoimmune disease that you have been a part of has most surprised you, in term of its ultimate clinical impact? Some of my most rewarding and impactful work has focused on the shared genetic basis of autoimmune diseases. We’ve identified dozens of genes that contribute to the risk and outcome of rheumatoid arthritis, lupus, and other autoimmune disorders. These discoveries regarding shared genes and pathways among such a diverse set of conditions have helped to inform optimal therapeutic target and treatment strategies across multiple diseases. For example, exploration of RA genes and pathways has revealed that approved agents for other conditions, such as cancer, may be appropriately repurposed for the treatment of RA. These are critical observations that have the potential to dramatically accelerate progress in developing new therapies for autoimmune diseases, such as RA.
Did you have much interaction with Stephen I. Katz, MD, PhD, your longtime predecessor who passed away unexpectedly in 2018? If so, what do you remember most about him? I regret that I had very little interaction with Steve, but I am well aware of the impact he had on NIAMS, NIH, and the research enterprise overall. He inspired so many people in a personal way, and I am energized by the legacy that he left behind.
What are your goals for the early part of your tenure as the new director of NIAMS? An important goal is getting to know the NIAMS community and expanding my knowledge of the Institute’s musculoskeletal and skin portfolios. I am also conducting outreach to Institute/Center directors and other NIH leadership to increase opportunities for input and advice. In doing this, I am identifying shared research interests, best practices, and potential partners for possible future collaborations. Another important goal is to increase NIAMS’ visibility within and beyond NIH. Ultimately, I want to contribute to the great work of the Institute and improve the lives of people with rheumatic, musculoskeletal, and skin diseases.
How would you characterize your management style? I like to lead with a flat hierarchy and work collectively to address opportunities and challenges. I value team building and tend to tap a variety of perspectives and expertise at all levels to achieve consensus, where possible.
The Accelerating Medicines Partnership (AMP) program was launched in 2014, with projects in three disease areas including the autoimmune disorders RA and lupus. What are some recent highlights from this program with respect to RA and lupus? AMP RA/SLE was dedicated to identifying promising therapeutic targets for RA and systemic lupus erythematosus. AMP-funded researchers have applied cutting-edge technologies to study cells from the synovial tissues of the joints of people with RA, and from the kidneys of people with lupus nephritis. In 2014, studying tissues in patients where the disease is active was a novel approach, since most research was conducted in mouse models or human blood samples.
The AMP RA/SLE Network developed a rich dataset that is available to the research community. Investigators are now using the data to facilitate RA and lupus research. For example, using AMP data, NIAMS-supported researchers identified potential biomarkers that could help predict an imminent RA flare. Work from another NIAMS-supported group suggests that targeting the regulatory transcription factor HIF-1, which drives inflammation and tissue damage, might be an effective approach for treating renal injury in lupus.
The data generated are accessible to the scientific community through two NIH websites: the database of Genotypes and Phenotypes (dbGaP) and the Immunology Database and Analysis Portal (IMMPORT).
Given the success of AMP RA/SLE, NIH plans to launch an “AMP 2.0” later in 2021. The AMP Autoimmune and Immune-Mediated Diseases (AMP AIM) program will provide an opportunity to leverage the accomplishments of AMP RA/SLE to new conditions, including psoriatic spectrum diseases and Sjögren’s syndrome.
What are some recent highlights from NIAMS-supported research in skin diseases? NIAMS-supported investigators continue to make significant strides in our understanding of skin biology and disease. For example, researchers recently demonstrated that imiquimod, a drug used to treat precancerous skin lesions, can help mouse ear wounds heal without scarring.
Another team addressed the safety and potential benefit of Staphylococcus hominis A9, a bacterium isolated from healthy human skin, as a topical therapy for atopic dermatitis.
Moving forward, AMP AIM will refine and extend the single-cell analysis of tissues to additional diseases, including psoriasis, setting the stage for the discovery of new therapeutic targets for the disease.
How has the COVID-19 pandemic changed the landscape of research, at least for the short term? This is a once-in-a-century pandemic that none of us were fully prepared for. We understand that it has been particularly challenging for women scientists, scientists with young children, and trainees and junior faculty who are at critically important and vulnerable stages of their careers. There isn’t a lab or clinical setting that hasn’t been negatively impacted in some way.
During the pandemic, the NIH instituted administrative flexibilities to support the grantee community, including extensions in time. In addition, the agency has issued several funding opportunities specific to COVID-19, some of which involve NIAMS participation.
What is NIAMS doing to help early/young investigators as well as female investigators and those from minority groups? Structural racism in biomedical research is a heightened concern. Earlier this year, Dr. Collins established the UNITE initiative to address structural racism and promote racial equity and inclusion at the NIH and within the larger biomedical community that we support. NIAMS is fully committed to this effort. One example is the Diversity Supplement Program, which is designed to attract and encourage eligible individuals from underrepresented populations to research careers.
Early-stage investigators are another top priority. In a tribute to the beloved former NIAMS director, NIH recently established the Stephen I. Katz Early Stage Investigator Research Grant Program. The R01 award provides support for a project unrelated to an early investigator’s area of postdoctoral study. (No preliminary data are allowed.) This award mechanism is a unique opportunity for early-stage investigators to take their research in a completely new direction.
Managing work and family life is an important concern, particularly for female investigators. Many NIH grant awards allow for reimbursement of actual, allowable costs incurred for childcare and parental leave. The NIH is exploring initiatives to promote research continuity and retention of eligible investigators facing major life events, such as pregnancy, childbirth, and adoption, at vulnerable career stages.
Who inspires you most in your work today? I am inspired by the ongoing struggles of our patients, junior investigators, and by the committed staff members on my team.
After many years at the University of California, San Francisco, Lindsey A. Criswell, MD, MPH, DSc, began a new chapter in February 2021 as the director of the National Institute of Arthritis and Musculoskeletal and Skin Disease, part of the National Institutes of Health. NIH Director Francis S. Collins, MD, PhD, selected her for the post.
“Dr. Criswell has rich experience as a clinician, researcher, and administrator,” Dr. Collins said in a prepared statement. “Her ability to oversee the research program of one of the country’s top research-intensive medical schools, and her expertise in autoimmune diseases, including rheumatoid arthritis and lupus, make her well positioned to direct NIAMS.” Dr. Criswell, a rheumatologist, was named a full professor of medicine at UCSF in 2007 and had served as vice chancellor of research at the university since 2017. She has authored more than 250 peer-reviewed scientific papers, and her efforts have contributed to the identification of more than 30 genes linked to autoimmune disorders. In her first media interview, Dr. Criswell opens up about her mentors, operational challenges posed by the COVID-19 pandemic, and highlights many NIAMS research projects underway.
Who inspired you most early in your career as a physician scientist? I have had great opportunities to work with fabulous mentors. Wallace (Wally) Epstein, MD, was my mentor when I was a rheumatology fellow and junior faculty member at UCSF. He was broadly admired for the breadth of his experience as a clinician and a researcher, and he was noteworthy at that time for his strong support for women and students of color. One of the many things I appreciated about him was his diverse range of interests outside of work, which included cello playing and woodworking.
Another mentor was Ephraim (Eph) Engleman, MD, the first academic rheumatologist in California. Eph continued to see patients beyond the age of 100. Perhaps his most important contributions were his efforts towards advocacy for funding for research and education in rheumatology. A prodigy violinist, he too had a broad range of personal interests.
What research into the genetics and epidemiology of human autoimmune disease that you have been a part of has most surprised you, in term of its ultimate clinical impact? Some of my most rewarding and impactful work has focused on the shared genetic basis of autoimmune diseases. We’ve identified dozens of genes that contribute to the risk and outcome of rheumatoid arthritis, lupus, and other autoimmune disorders. These discoveries regarding shared genes and pathways among such a diverse set of conditions have helped to inform optimal therapeutic target and treatment strategies across multiple diseases. For example, exploration of RA genes and pathways has revealed that approved agents for other conditions, such as cancer, may be appropriately repurposed for the treatment of RA. These are critical observations that have the potential to dramatically accelerate progress in developing new therapies for autoimmune diseases, such as RA.
Did you have much interaction with Stephen I. Katz, MD, PhD, your longtime predecessor who passed away unexpectedly in 2018? If so, what do you remember most about him? I regret that I had very little interaction with Steve, but I am well aware of the impact he had on NIAMS, NIH, and the research enterprise overall. He inspired so many people in a personal way, and I am energized by the legacy that he left behind.
What are your goals for the early part of your tenure as the new director of NIAMS? An important goal is getting to know the NIAMS community and expanding my knowledge of the Institute’s musculoskeletal and skin portfolios. I am also conducting outreach to Institute/Center directors and other NIH leadership to increase opportunities for input and advice. In doing this, I am identifying shared research interests, best practices, and potential partners for possible future collaborations. Another important goal is to increase NIAMS’ visibility within and beyond NIH. Ultimately, I want to contribute to the great work of the Institute and improve the lives of people with rheumatic, musculoskeletal, and skin diseases.
How would you characterize your management style? I like to lead with a flat hierarchy and work collectively to address opportunities and challenges. I value team building and tend to tap a variety of perspectives and expertise at all levels to achieve consensus, where possible.
The Accelerating Medicines Partnership (AMP) program was launched in 2014, with projects in three disease areas including the autoimmune disorders RA and lupus. What are some recent highlights from this program with respect to RA and lupus? AMP RA/SLE was dedicated to identifying promising therapeutic targets for RA and systemic lupus erythematosus. AMP-funded researchers have applied cutting-edge technologies to study cells from the synovial tissues of the joints of people with RA, and from the kidneys of people with lupus nephritis. In 2014, studying tissues in patients where the disease is active was a novel approach, since most research was conducted in mouse models or human blood samples.
The AMP RA/SLE Network developed a rich dataset that is available to the research community. Investigators are now using the data to facilitate RA and lupus research. For example, using AMP data, NIAMS-supported researchers identified potential biomarkers that could help predict an imminent RA flare. Work from another NIAMS-supported group suggests that targeting the regulatory transcription factor HIF-1, which drives inflammation and tissue damage, might be an effective approach for treating renal injury in lupus.
The data generated are accessible to the scientific community through two NIH websites: the database of Genotypes and Phenotypes (dbGaP) and the Immunology Database and Analysis Portal (IMMPORT).
Given the success of AMP RA/SLE, NIH plans to launch an “AMP 2.0” later in 2021. The AMP Autoimmune and Immune-Mediated Diseases (AMP AIM) program will provide an opportunity to leverage the accomplishments of AMP RA/SLE to new conditions, including psoriatic spectrum diseases and Sjögren’s syndrome.
What are some recent highlights from NIAMS-supported research in skin diseases? NIAMS-supported investigators continue to make significant strides in our understanding of skin biology and disease. For example, researchers recently demonstrated that imiquimod, a drug used to treat precancerous skin lesions, can help mouse ear wounds heal without scarring.
Another team addressed the safety and potential benefit of Staphylococcus hominis A9, a bacterium isolated from healthy human skin, as a topical therapy for atopic dermatitis.
Moving forward, AMP AIM will refine and extend the single-cell analysis of tissues to additional diseases, including psoriasis, setting the stage for the discovery of new therapeutic targets for the disease.
How has the COVID-19 pandemic changed the landscape of research, at least for the short term? This is a once-in-a-century pandemic that none of us were fully prepared for. We understand that it has been particularly challenging for women scientists, scientists with young children, and trainees and junior faculty who are at critically important and vulnerable stages of their careers. There isn’t a lab or clinical setting that hasn’t been negatively impacted in some way.
During the pandemic, the NIH instituted administrative flexibilities to support the grantee community, including extensions in time. In addition, the agency has issued several funding opportunities specific to COVID-19, some of which involve NIAMS participation.
What is NIAMS doing to help early/young investigators as well as female investigators and those from minority groups? Structural racism in biomedical research is a heightened concern. Earlier this year, Dr. Collins established the UNITE initiative to address structural racism and promote racial equity and inclusion at the NIH and within the larger biomedical community that we support. NIAMS is fully committed to this effort. One example is the Diversity Supplement Program, which is designed to attract and encourage eligible individuals from underrepresented populations to research careers.
Early-stage investigators are another top priority. In a tribute to the beloved former NIAMS director, NIH recently established the Stephen I. Katz Early Stage Investigator Research Grant Program. The R01 award provides support for a project unrelated to an early investigator’s area of postdoctoral study. (No preliminary data are allowed.) This award mechanism is a unique opportunity for early-stage investigators to take their research in a completely new direction.
Managing work and family life is an important concern, particularly for female investigators. Many NIH grant awards allow for reimbursement of actual, allowable costs incurred for childcare and parental leave. The NIH is exploring initiatives to promote research continuity and retention of eligible investigators facing major life events, such as pregnancy, childbirth, and adoption, at vulnerable career stages.
Who inspires you most in your work today? I am inspired by the ongoing struggles of our patients, junior investigators, and by the committed staff members on my team.
Acceptance of biosimilars grows but greater use may hinge on switching, interchangeability studies
It took years for Elle Moxley to get a diagnosis that explained her crippling gastrointestinal pain, digestion problems, fatigue, and hot, red rashes. And after learning in 2016 that she had Crohn’s disease, a chronic inflammation of the digestive tract, she spent more than 4 years trying medications before getting her disease under control with a biologic drug called Remicade.
So Ms. Moxley, 33, was dismayed to receive a notice from her insurer in January that Remicade would no longer be covered as a preferred drug on her plan. Another drug, Inflectra, which the Food and Drug Administration says has no meaningful clinical differences from Remicade, is now preferred. It is a “biosimilar” drug.
“I felt very powerless,” said Ms. Moxley, who recently started a job as a public relations coordinator for Kansas City (Mo.) Public Schools. “I have this decision being made for me and my doctor that’s not in my best interest, and it might knock me out of remission.”
After Ms. Moxley’s first Inflectra infusion in July, she developed a painful rash. It went away after a few days, but she said she continues to feel extremely fatigued and experiences gastrointestinal pain, constipation, diarrhea and nausea.
Many medical professionals look to biosimilar drugs as a way to increase competition and give consumers cheaper options, much as generic drugs do, and they point to the more robust use of these products in Europe to cut costs.
Yet the United States has been slower to adopt biosimilar drugs since the first such medicine was approved in 2015. That’s partly because of concerns raised by patients like Moxley and their doctors, but also because brand-name biologics have kept biosimilars from entering the market. The companies behind the brand-name drugs have used legal actions to extend the life of their patents and incentives that make offering the brand biologic more attractive than offering a biosimilar on a formulary, listing which drugs are covered on an insurance plan.
“It distorts the market and makes it so that patients can’t get access,” said Jinoos Yazdany, MD, MPH, a professor of medicine and chief of the rheumatology division at Zuckerberg San Francisco General Hospital.
The FDA has approved 31 biosimilar medications since 2015, but only about 60% have made it to market, according to an analysis by NORC, a research organization at the University of Chicago.
Remicade’s manufacturer, Johnson & Johnson, and Pfizer, which makes the Remicade biosimilar Inflectra, have been embroiled in a long-running lawsuit over Pfizer’s claims that Johnson & Johnson tried to choke off competition through exclusionary contracts with insurers and other anticompetitive actions. In July, the companies settled the case on undisclosed terms.
In a statement, Pfizer said it would continue to sell Inflectra in the United States but noted ongoing challenges: “Pfizer has begun to see progress in the overall biosimilars marketplace in the U.S. However, changes in policy at a government level and acceptance of biosimilars among key stakeholders are critical to deliver more meaningful uptake so patients and the health care system at large can benefit from the cost savings these medicines may deliver.”
Johnson & Johnson said it is committed to making Remicade available to patients who choose it, which “compels us to compete responsibly on both price and value.”
Biologic medicines, which are generally grown from living organisms such as animal cells or bacteria, are more complex and expensive to manufacture than drugs made from chemicals. In recent years, biologic drugs have become a mainstay of treatment for autoimmune conditions like Crohn’s disease and rheumatoid arthritis, as well as certain cancers and diabetes, among other conditions.
Other drugmakers can’t exactly reproduce these biologic drugs by following chemical recipes as they do for generic versions of conventional drugs.
Instead, biosimilar versions of biologic drugs are generally made from the same types of materials as the original biologics and must be “highly similar” to them to be approved by the FDA. They must have no clinically meaningful differences from the biologic drug, and be just as safe, pure and potent. More than a decade after Congress created an approval pathway for biosimilars, they are widely accepted as safe and effective alternatives to brand biologics.
Medical experts hope that as biosimilars become more widely used they will increasingly provide a brake on drug spending.
From 2015 to 2019, drug spending overall grew 6.1%, while spending on biologics grew more than twice as much – 14.6% – according to a report by IQVIA, a health care analytics company. In 2019, biologics accounted for 43% of drug spending in the United States
Biosimilars provide a roughly 30% discount over brand biologics in the United States but have the potential to reduce spending by more than $100 billion in the next 5 years, the IQVIA analysis found.
In a survey of 602 physicians who prescribe biologic medications, more than three-quarters said they believed biosimilars are just as safe and effective as their biologic counterparts, according to NORC.
But they were less comfortable with switching patients from a brand biologic to a biosimilar. While about half said they were very likely to prescribe a biosimilar to a patient just starting biologic therapy, only 31% said they were very likely to prescribe a biosimilar to a patient already doing well on a brand biologic.
It can be challenging to find a treatment regimen that works for patients with complicated chronic conditions, and physicians and patients often don’t want to rock the boat once that is achieved.
In Ms. Moxley’s case, for example, before her condition stabilized on Remicade, she tried a conventional pill called Lialda, the biologic drug Humira and a lower dose of Remicade.
Some doctors and patients raise concerns that switching between these drugs might cause patients to develop antibodies that cause the drugs to lose effectiveness. They want to see more research about the effects of such switches.
“We haven’t seen enough studies about patients going from the biologic to the biosimilar and bouncing back and forth,” said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “We don’t want our patients to be guinea pigs.”
Manufacturers of biologic and biosimilar drugs have participated in advertising, exhibit or sponsorship opportunities with the American College of Rheumatology, according to ACR spokesperson Jocelyn Givens.
But studies show a one-time switch from Remicade to a biosimilar like Inflectra does not cause side effects or the development of antibodies, said Ross Maltz, MD, a pediatric gastroenterologist at Nationwide Children’s Hospital in Columbus, Ohio, and former member of the Crohn’s & Colitis Foundation’s National Scientific Advisory Committee. Studies may be conducted by researchers with extensive ties to the industry and funded by drugmakers.
Situations like Ms. Moxley’s are unusual, said Kristine Grow, senior vice president of communications at AHIP, an insurer trade group.
“For patients who have been taking a brand-name biologic for some time, health insurance providers do not typically encourage them to switch to a biosimilar because of a formulary change, and most plans exclude these patients from any changes in cost sharing due to formulary changes,” she said.
Drugmakers can seek approval from the FDA of their biosimilar as interchangeable with a biologic drug, allowing pharmacists, subject to state law, to switch a physician’s prescription from the brand drug, as they often do with generic drugs.
However, the FDA has approved only one biosimilar (Semglee, a form of insulin) as interchangeable with a biologic (Lantus).
Like Ms. Moxley, many other patients using biologics get copay assistance from drug companies, but the money often isn’t enough to cover the full cost. In her old job as a radio reporter, Ms. Moxley said, she hit the $7,000 maximum annual out-of-pocket spending limit for her plan by May.
In her new job, Ms. Moxley has an individual plan with a $4,000 maximum out-of-pocket limit, which she expects to blow past once again within months.
But she received good news recently: Her new plan will cover Remicade.
“I’m still concerned that I will have developed antibodies since my last dose,” she said. “But it feels like a step in the direction of good health again.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
It took years for Elle Moxley to get a diagnosis that explained her crippling gastrointestinal pain, digestion problems, fatigue, and hot, red rashes. And after learning in 2016 that she had Crohn’s disease, a chronic inflammation of the digestive tract, she spent more than 4 years trying medications before getting her disease under control with a biologic drug called Remicade.
So Ms. Moxley, 33, was dismayed to receive a notice from her insurer in January that Remicade would no longer be covered as a preferred drug on her plan. Another drug, Inflectra, which the Food and Drug Administration says has no meaningful clinical differences from Remicade, is now preferred. It is a “biosimilar” drug.
“I felt very powerless,” said Ms. Moxley, who recently started a job as a public relations coordinator for Kansas City (Mo.) Public Schools. “I have this decision being made for me and my doctor that’s not in my best interest, and it might knock me out of remission.”
After Ms. Moxley’s first Inflectra infusion in July, she developed a painful rash. It went away after a few days, but she said she continues to feel extremely fatigued and experiences gastrointestinal pain, constipation, diarrhea and nausea.
Many medical professionals look to biosimilar drugs as a way to increase competition and give consumers cheaper options, much as generic drugs do, and they point to the more robust use of these products in Europe to cut costs.
Yet the United States has been slower to adopt biosimilar drugs since the first such medicine was approved in 2015. That’s partly because of concerns raised by patients like Moxley and their doctors, but also because brand-name biologics have kept biosimilars from entering the market. The companies behind the brand-name drugs have used legal actions to extend the life of their patents and incentives that make offering the brand biologic more attractive than offering a biosimilar on a formulary, listing which drugs are covered on an insurance plan.
“It distorts the market and makes it so that patients can’t get access,” said Jinoos Yazdany, MD, MPH, a professor of medicine and chief of the rheumatology division at Zuckerberg San Francisco General Hospital.
The FDA has approved 31 biosimilar medications since 2015, but only about 60% have made it to market, according to an analysis by NORC, a research organization at the University of Chicago.
Remicade’s manufacturer, Johnson & Johnson, and Pfizer, which makes the Remicade biosimilar Inflectra, have been embroiled in a long-running lawsuit over Pfizer’s claims that Johnson & Johnson tried to choke off competition through exclusionary contracts with insurers and other anticompetitive actions. In July, the companies settled the case on undisclosed terms.
In a statement, Pfizer said it would continue to sell Inflectra in the United States but noted ongoing challenges: “Pfizer has begun to see progress in the overall biosimilars marketplace in the U.S. However, changes in policy at a government level and acceptance of biosimilars among key stakeholders are critical to deliver more meaningful uptake so patients and the health care system at large can benefit from the cost savings these medicines may deliver.”
Johnson & Johnson said it is committed to making Remicade available to patients who choose it, which “compels us to compete responsibly on both price and value.”
Biologic medicines, which are generally grown from living organisms such as animal cells or bacteria, are more complex and expensive to manufacture than drugs made from chemicals. In recent years, biologic drugs have become a mainstay of treatment for autoimmune conditions like Crohn’s disease and rheumatoid arthritis, as well as certain cancers and diabetes, among other conditions.
Other drugmakers can’t exactly reproduce these biologic drugs by following chemical recipes as they do for generic versions of conventional drugs.
Instead, biosimilar versions of biologic drugs are generally made from the same types of materials as the original biologics and must be “highly similar” to them to be approved by the FDA. They must have no clinically meaningful differences from the biologic drug, and be just as safe, pure and potent. More than a decade after Congress created an approval pathway for biosimilars, they are widely accepted as safe and effective alternatives to brand biologics.
Medical experts hope that as biosimilars become more widely used they will increasingly provide a brake on drug spending.
From 2015 to 2019, drug spending overall grew 6.1%, while spending on biologics grew more than twice as much – 14.6% – according to a report by IQVIA, a health care analytics company. In 2019, biologics accounted for 43% of drug spending in the United States
Biosimilars provide a roughly 30% discount over brand biologics in the United States but have the potential to reduce spending by more than $100 billion in the next 5 years, the IQVIA analysis found.
In a survey of 602 physicians who prescribe biologic medications, more than three-quarters said they believed biosimilars are just as safe and effective as their biologic counterparts, according to NORC.
But they were less comfortable with switching patients from a brand biologic to a biosimilar. While about half said they were very likely to prescribe a biosimilar to a patient just starting biologic therapy, only 31% said they were very likely to prescribe a biosimilar to a patient already doing well on a brand biologic.
It can be challenging to find a treatment regimen that works for patients with complicated chronic conditions, and physicians and patients often don’t want to rock the boat once that is achieved.
In Ms. Moxley’s case, for example, before her condition stabilized on Remicade, she tried a conventional pill called Lialda, the biologic drug Humira and a lower dose of Remicade.
Some doctors and patients raise concerns that switching between these drugs might cause patients to develop antibodies that cause the drugs to lose effectiveness. They want to see more research about the effects of such switches.
“We haven’t seen enough studies about patients going from the biologic to the biosimilar and bouncing back and forth,” said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “We don’t want our patients to be guinea pigs.”
Manufacturers of biologic and biosimilar drugs have participated in advertising, exhibit or sponsorship opportunities with the American College of Rheumatology, according to ACR spokesperson Jocelyn Givens.
But studies show a one-time switch from Remicade to a biosimilar like Inflectra does not cause side effects or the development of antibodies, said Ross Maltz, MD, a pediatric gastroenterologist at Nationwide Children’s Hospital in Columbus, Ohio, and former member of the Crohn’s & Colitis Foundation’s National Scientific Advisory Committee. Studies may be conducted by researchers with extensive ties to the industry and funded by drugmakers.
Situations like Ms. Moxley’s are unusual, said Kristine Grow, senior vice president of communications at AHIP, an insurer trade group.
“For patients who have been taking a brand-name biologic for some time, health insurance providers do not typically encourage them to switch to a biosimilar because of a formulary change, and most plans exclude these patients from any changes in cost sharing due to formulary changes,” she said.
Drugmakers can seek approval from the FDA of their biosimilar as interchangeable with a biologic drug, allowing pharmacists, subject to state law, to switch a physician’s prescription from the brand drug, as they often do with generic drugs.
However, the FDA has approved only one biosimilar (Semglee, a form of insulin) as interchangeable with a biologic (Lantus).
Like Ms. Moxley, many other patients using biologics get copay assistance from drug companies, but the money often isn’t enough to cover the full cost. In her old job as a radio reporter, Ms. Moxley said, she hit the $7,000 maximum annual out-of-pocket spending limit for her plan by May.
In her new job, Ms. Moxley has an individual plan with a $4,000 maximum out-of-pocket limit, which she expects to blow past once again within months.
But she received good news recently: Her new plan will cover Remicade.
“I’m still concerned that I will have developed antibodies since my last dose,” she said. “But it feels like a step in the direction of good health again.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
It took years for Elle Moxley to get a diagnosis that explained her crippling gastrointestinal pain, digestion problems, fatigue, and hot, red rashes. And after learning in 2016 that she had Crohn’s disease, a chronic inflammation of the digestive tract, she spent more than 4 years trying medications before getting her disease under control with a biologic drug called Remicade.
So Ms. Moxley, 33, was dismayed to receive a notice from her insurer in January that Remicade would no longer be covered as a preferred drug on her plan. Another drug, Inflectra, which the Food and Drug Administration says has no meaningful clinical differences from Remicade, is now preferred. It is a “biosimilar” drug.
“I felt very powerless,” said Ms. Moxley, who recently started a job as a public relations coordinator for Kansas City (Mo.) Public Schools. “I have this decision being made for me and my doctor that’s not in my best interest, and it might knock me out of remission.”
After Ms. Moxley’s first Inflectra infusion in July, she developed a painful rash. It went away after a few days, but she said she continues to feel extremely fatigued and experiences gastrointestinal pain, constipation, diarrhea and nausea.
Many medical professionals look to biosimilar drugs as a way to increase competition and give consumers cheaper options, much as generic drugs do, and they point to the more robust use of these products in Europe to cut costs.
Yet the United States has been slower to adopt biosimilar drugs since the first such medicine was approved in 2015. That’s partly because of concerns raised by patients like Moxley and their doctors, but also because brand-name biologics have kept biosimilars from entering the market. The companies behind the brand-name drugs have used legal actions to extend the life of their patents and incentives that make offering the brand biologic more attractive than offering a biosimilar on a formulary, listing which drugs are covered on an insurance plan.
“It distorts the market and makes it so that patients can’t get access,” said Jinoos Yazdany, MD, MPH, a professor of medicine and chief of the rheumatology division at Zuckerberg San Francisco General Hospital.
The FDA has approved 31 biosimilar medications since 2015, but only about 60% have made it to market, according to an analysis by NORC, a research organization at the University of Chicago.
Remicade’s manufacturer, Johnson & Johnson, and Pfizer, which makes the Remicade biosimilar Inflectra, have been embroiled in a long-running lawsuit over Pfizer’s claims that Johnson & Johnson tried to choke off competition through exclusionary contracts with insurers and other anticompetitive actions. In July, the companies settled the case on undisclosed terms.
In a statement, Pfizer said it would continue to sell Inflectra in the United States but noted ongoing challenges: “Pfizer has begun to see progress in the overall biosimilars marketplace in the U.S. However, changes in policy at a government level and acceptance of biosimilars among key stakeholders are critical to deliver more meaningful uptake so patients and the health care system at large can benefit from the cost savings these medicines may deliver.”
Johnson & Johnson said it is committed to making Remicade available to patients who choose it, which “compels us to compete responsibly on both price and value.”
Biologic medicines, which are generally grown from living organisms such as animal cells or bacteria, are more complex and expensive to manufacture than drugs made from chemicals. In recent years, biologic drugs have become a mainstay of treatment for autoimmune conditions like Crohn’s disease and rheumatoid arthritis, as well as certain cancers and diabetes, among other conditions.
Other drugmakers can’t exactly reproduce these biologic drugs by following chemical recipes as they do for generic versions of conventional drugs.
Instead, biosimilar versions of biologic drugs are generally made from the same types of materials as the original biologics and must be “highly similar” to them to be approved by the FDA. They must have no clinically meaningful differences from the biologic drug, and be just as safe, pure and potent. More than a decade after Congress created an approval pathway for biosimilars, they are widely accepted as safe and effective alternatives to brand biologics.
Medical experts hope that as biosimilars become more widely used they will increasingly provide a brake on drug spending.
From 2015 to 2019, drug spending overall grew 6.1%, while spending on biologics grew more than twice as much – 14.6% – according to a report by IQVIA, a health care analytics company. In 2019, biologics accounted for 43% of drug spending in the United States
Biosimilars provide a roughly 30% discount over brand biologics in the United States but have the potential to reduce spending by more than $100 billion in the next 5 years, the IQVIA analysis found.
In a survey of 602 physicians who prescribe biologic medications, more than three-quarters said they believed biosimilars are just as safe and effective as their biologic counterparts, according to NORC.
But they were less comfortable with switching patients from a brand biologic to a biosimilar. While about half said they were very likely to prescribe a biosimilar to a patient just starting biologic therapy, only 31% said they were very likely to prescribe a biosimilar to a patient already doing well on a brand biologic.
It can be challenging to find a treatment regimen that works for patients with complicated chronic conditions, and physicians and patients often don’t want to rock the boat once that is achieved.
In Ms. Moxley’s case, for example, before her condition stabilized on Remicade, she tried a conventional pill called Lialda, the biologic drug Humira and a lower dose of Remicade.
Some doctors and patients raise concerns that switching between these drugs might cause patients to develop antibodies that cause the drugs to lose effectiveness. They want to see more research about the effects of such switches.
“We haven’t seen enough studies about patients going from the biologic to the biosimilar and bouncing back and forth,” said Marcus Snow, MD, chair of the American College of Rheumatology’s Committee on Rheumatologic Care. “We don’t want our patients to be guinea pigs.”
Manufacturers of biologic and biosimilar drugs have participated in advertising, exhibit or sponsorship opportunities with the American College of Rheumatology, according to ACR spokesperson Jocelyn Givens.
But studies show a one-time switch from Remicade to a biosimilar like Inflectra does not cause side effects or the development of antibodies, said Ross Maltz, MD, a pediatric gastroenterologist at Nationwide Children’s Hospital in Columbus, Ohio, and former member of the Crohn’s & Colitis Foundation’s National Scientific Advisory Committee. Studies may be conducted by researchers with extensive ties to the industry and funded by drugmakers.
Situations like Ms. Moxley’s are unusual, said Kristine Grow, senior vice president of communications at AHIP, an insurer trade group.
“For patients who have been taking a brand-name biologic for some time, health insurance providers do not typically encourage them to switch to a biosimilar because of a formulary change, and most plans exclude these patients from any changes in cost sharing due to formulary changes,” she said.
Drugmakers can seek approval from the FDA of their biosimilar as interchangeable with a biologic drug, allowing pharmacists, subject to state law, to switch a physician’s prescription from the brand drug, as they often do with generic drugs.
However, the FDA has approved only one biosimilar (Semglee, a form of insulin) as interchangeable with a biologic (Lantus).
Like Ms. Moxley, many other patients using biologics get copay assistance from drug companies, but the money often isn’t enough to cover the full cost. In her old job as a radio reporter, Ms. Moxley said, she hit the $7,000 maximum annual out-of-pocket spending limit for her plan by May.
In her new job, Ms. Moxley has an individual plan with a $4,000 maximum out-of-pocket limit, which she expects to blow past once again within months.
But she received good news recently: Her new plan will cover Remicade.
“I’m still concerned that I will have developed antibodies since my last dose,” she said. “But it feels like a step in the direction of good health again.”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
More severe psoriasis linked to an increased risk of PsA
Factors that predict the development of psoriasis in patients with psoriasis include nail, inverse, and scalp psoriasis; family history of PsA; as well as severity of skin disease. And like psoriasis, “PsA is associated with a multitude of comorbidities, including cardiovascular disease, metabolic syndrome, Crohn’s disease, obesity, diabetes, uveitis, anxiety, and depression, with correspondingly higher healthcare utilization and direct healthcare costs,” wrote corresponding author Joseph F. Merola, MD, MMSc, and colleagues. The study was published online in the Journal of the American Academy of Dermatology. “Timely and accurate diagnosis of PsA is important for improved patient outcomes and appropriate disease management and may prevent prolonged inflammation that leads to structural joint damage and worsening physical function,” they added.
The mean time of onset of PsA among patients with psoriasis who develop PsA is 10 years after the first signs of psoriasis appear. An estimated 20%-30% of patients with psoriasis have a concurrent diagnosis of PsA, and the annual incidence of PsA has been reported to be 2.7 cases per 100 patients with psoriasis. While previous studies have suggested that a higher incidence of PsA is associated with greater disease severity, there are limited data in the United States on the topic.
For the study, Dr. Merola, a dermatologist and rheumatologist who directs the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, and his colleagues drew from the Optum EHR database to identify adult patients newly diagnosed with psoriasis between Jan. 1, 2009, and March 31, 2019. Patients diagnosed with psoriasis or PsA prior to the index date were excluded from the analysis for evaluation of incidence but included for evaluation of prevalence. The patients were followed from the index date until the earliest PsA event, death, or end of study or follow-up, whichever came first. The researchers calculated the incidence of PsA among adults with psoriasis as the number of incident PsA events divided by the number of patient-years (PY) at risk, which was reported as the raw incidence per 100 psoriasis PY. They calculated the prevalence of PsA among adults with psoriasis as “the number of prevalent PsA events divided by the number of eligible patients with [psoriasis] and reported by years in the follow-up period,” which was a median of 3.7 years.
A total of 114,868 patients were included in the analysis. At baseline, their mean age was 54 years, 53% were female, 89% were White, and 39% were obese. Most patients (102,553) were on nonsystemic agents during the year after their psoriasis diagnosis, while 6,345 were on nonbiologic systemic therapies (NBSTs) and 5,970 were on biologics. The researchers classified patients as having mild psoriasis if they were taking nonsystemic agents, moderate disease if they were taking NBSTs, or severe disease if they were taking biologics.
The overall incidence rate of PsA was 2.9 events per 100 PY and increased by severity of disease. When calculated by severity, the incidence was 2.1 events per 100 PY for patients with mild psoriasis, 9.9 events per 100 PY for those with moderate psoriasis, and 17.6 events per 100 PY for those with severe psoriasis.
When the researchers excluded patients diagnosed with PsA up to 1 year after being diagnosed with psoriasis, the overall incidence was lower (1.7 events per 100 PY), with similar trends for categories of treatment severity. Specifically, the incidence was 1.5, 3.1, and 4.7 events per 100 PY among those with mild, moderate, and severe psoriasis, based on their treatment groups, respectively.
Among the 120,523 patients with psoriasis who were eligible for the assessment of prevalence of PsA, the overall 5-year prevalence of PsA was 14.2% and rose with severity of disease: 9.9% in patients with mild psoriasis, 35% in patients with moderate psoriasis, and 54.9% in patients with severe psoriasis.
Other predictors of PsA onset for both index-date cohorts included weight of 90 kg or greater, female gender, age group 25-65 years (compared with the age group over 65 years), and rheumatic risk factors such as wrist pain and unspecified rheumatism.
“To ensure timely diagnosis and treatment for management and prevention of PsA, patients with [psoriasis] should be routinely screened, especially those with more severe disease and other PsA risk factors,” the authors advised.
Dr. Merola and colleagues acknowledged certain limitations of their analysis, including the potential for selection bias and its reliance on EHR data which “lacked clinical measures of disease severity such as the PASI, and data on BSA were not available for all study participants; therefore, treatment groups were used as a surrogate for disease severity,” they wrote. “As a result, some patients may have been miscategorized, especially patients with severe disease who were untreated.”
The study was sponsored by Novartis. Dr. Merola disclosed that he is a consultant and/or investigator for Merck, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Celgene, Sanofi, Regeneron, Arena, Sun Pharmaceuticals, Biogen, Pfizer, EMD Serono, Avotres, and LEO Pharma. Four authors are Novartis employees, or employees of a consulting company that provides services to Novartis; and another author disclosed serving as an investigator or consultant for several pharmaceutical companies, including Novartis.
Factors that predict the development of psoriasis in patients with psoriasis include nail, inverse, and scalp psoriasis; family history of PsA; as well as severity of skin disease. And like psoriasis, “PsA is associated with a multitude of comorbidities, including cardiovascular disease, metabolic syndrome, Crohn’s disease, obesity, diabetes, uveitis, anxiety, and depression, with correspondingly higher healthcare utilization and direct healthcare costs,” wrote corresponding author Joseph F. Merola, MD, MMSc, and colleagues. The study was published online in the Journal of the American Academy of Dermatology. “Timely and accurate diagnosis of PsA is important for improved patient outcomes and appropriate disease management and may prevent prolonged inflammation that leads to structural joint damage and worsening physical function,” they added.
The mean time of onset of PsA among patients with psoriasis who develop PsA is 10 years after the first signs of psoriasis appear. An estimated 20%-30% of patients with psoriasis have a concurrent diagnosis of PsA, and the annual incidence of PsA has been reported to be 2.7 cases per 100 patients with psoriasis. While previous studies have suggested that a higher incidence of PsA is associated with greater disease severity, there are limited data in the United States on the topic.
For the study, Dr. Merola, a dermatologist and rheumatologist who directs the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, and his colleagues drew from the Optum EHR database to identify adult patients newly diagnosed with psoriasis between Jan. 1, 2009, and March 31, 2019. Patients diagnosed with psoriasis or PsA prior to the index date were excluded from the analysis for evaluation of incidence but included for evaluation of prevalence. The patients were followed from the index date until the earliest PsA event, death, or end of study or follow-up, whichever came first. The researchers calculated the incidence of PsA among adults with psoriasis as the number of incident PsA events divided by the number of patient-years (PY) at risk, which was reported as the raw incidence per 100 psoriasis PY. They calculated the prevalence of PsA among adults with psoriasis as “the number of prevalent PsA events divided by the number of eligible patients with [psoriasis] and reported by years in the follow-up period,” which was a median of 3.7 years.
A total of 114,868 patients were included in the analysis. At baseline, their mean age was 54 years, 53% were female, 89% were White, and 39% were obese. Most patients (102,553) were on nonsystemic agents during the year after their psoriasis diagnosis, while 6,345 were on nonbiologic systemic therapies (NBSTs) and 5,970 were on biologics. The researchers classified patients as having mild psoriasis if they were taking nonsystemic agents, moderate disease if they were taking NBSTs, or severe disease if they were taking biologics.
The overall incidence rate of PsA was 2.9 events per 100 PY and increased by severity of disease. When calculated by severity, the incidence was 2.1 events per 100 PY for patients with mild psoriasis, 9.9 events per 100 PY for those with moderate psoriasis, and 17.6 events per 100 PY for those with severe psoriasis.
When the researchers excluded patients diagnosed with PsA up to 1 year after being diagnosed with psoriasis, the overall incidence was lower (1.7 events per 100 PY), with similar trends for categories of treatment severity. Specifically, the incidence was 1.5, 3.1, and 4.7 events per 100 PY among those with mild, moderate, and severe psoriasis, based on their treatment groups, respectively.
Among the 120,523 patients with psoriasis who were eligible for the assessment of prevalence of PsA, the overall 5-year prevalence of PsA was 14.2% and rose with severity of disease: 9.9% in patients with mild psoriasis, 35% in patients with moderate psoriasis, and 54.9% in patients with severe psoriasis.
Other predictors of PsA onset for both index-date cohorts included weight of 90 kg or greater, female gender, age group 25-65 years (compared with the age group over 65 years), and rheumatic risk factors such as wrist pain and unspecified rheumatism.
“To ensure timely diagnosis and treatment for management and prevention of PsA, patients with [psoriasis] should be routinely screened, especially those with more severe disease and other PsA risk factors,” the authors advised.
Dr. Merola and colleagues acknowledged certain limitations of their analysis, including the potential for selection bias and its reliance on EHR data which “lacked clinical measures of disease severity such as the PASI, and data on BSA were not available for all study participants; therefore, treatment groups were used as a surrogate for disease severity,” they wrote. “As a result, some patients may have been miscategorized, especially patients with severe disease who were untreated.”
The study was sponsored by Novartis. Dr. Merola disclosed that he is a consultant and/or investigator for Merck, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Celgene, Sanofi, Regeneron, Arena, Sun Pharmaceuticals, Biogen, Pfizer, EMD Serono, Avotres, and LEO Pharma. Four authors are Novartis employees, or employees of a consulting company that provides services to Novartis; and another author disclosed serving as an investigator or consultant for several pharmaceutical companies, including Novartis.
Factors that predict the development of psoriasis in patients with psoriasis include nail, inverse, and scalp psoriasis; family history of PsA; as well as severity of skin disease. And like psoriasis, “PsA is associated with a multitude of comorbidities, including cardiovascular disease, metabolic syndrome, Crohn’s disease, obesity, diabetes, uveitis, anxiety, and depression, with correspondingly higher healthcare utilization and direct healthcare costs,” wrote corresponding author Joseph F. Merola, MD, MMSc, and colleagues. The study was published online in the Journal of the American Academy of Dermatology. “Timely and accurate diagnosis of PsA is important for improved patient outcomes and appropriate disease management and may prevent prolonged inflammation that leads to structural joint damage and worsening physical function,” they added.
The mean time of onset of PsA among patients with psoriasis who develop PsA is 10 years after the first signs of psoriasis appear. An estimated 20%-30% of patients with psoriasis have a concurrent diagnosis of PsA, and the annual incidence of PsA has been reported to be 2.7 cases per 100 patients with psoriasis. While previous studies have suggested that a higher incidence of PsA is associated with greater disease severity, there are limited data in the United States on the topic.
For the study, Dr. Merola, a dermatologist and rheumatologist who directs the Center for Skin and Related Musculoskeletal Diseases at Brigham and Women’s Hospital, Boston, and his colleagues drew from the Optum EHR database to identify adult patients newly diagnosed with psoriasis between Jan. 1, 2009, and March 31, 2019. Patients diagnosed with psoriasis or PsA prior to the index date were excluded from the analysis for evaluation of incidence but included for evaluation of prevalence. The patients were followed from the index date until the earliest PsA event, death, or end of study or follow-up, whichever came first. The researchers calculated the incidence of PsA among adults with psoriasis as the number of incident PsA events divided by the number of patient-years (PY) at risk, which was reported as the raw incidence per 100 psoriasis PY. They calculated the prevalence of PsA among adults with psoriasis as “the number of prevalent PsA events divided by the number of eligible patients with [psoriasis] and reported by years in the follow-up period,” which was a median of 3.7 years.
A total of 114,868 patients were included in the analysis. At baseline, their mean age was 54 years, 53% were female, 89% were White, and 39% were obese. Most patients (102,553) were on nonsystemic agents during the year after their psoriasis diagnosis, while 6,345 were on nonbiologic systemic therapies (NBSTs) and 5,970 were on biologics. The researchers classified patients as having mild psoriasis if they were taking nonsystemic agents, moderate disease if they were taking NBSTs, or severe disease if they were taking biologics.
The overall incidence rate of PsA was 2.9 events per 100 PY and increased by severity of disease. When calculated by severity, the incidence was 2.1 events per 100 PY for patients with mild psoriasis, 9.9 events per 100 PY for those with moderate psoriasis, and 17.6 events per 100 PY for those with severe psoriasis.
When the researchers excluded patients diagnosed with PsA up to 1 year after being diagnosed with psoriasis, the overall incidence was lower (1.7 events per 100 PY), with similar trends for categories of treatment severity. Specifically, the incidence was 1.5, 3.1, and 4.7 events per 100 PY among those with mild, moderate, and severe psoriasis, based on their treatment groups, respectively.
Among the 120,523 patients with psoriasis who were eligible for the assessment of prevalence of PsA, the overall 5-year prevalence of PsA was 14.2% and rose with severity of disease: 9.9% in patients with mild psoriasis, 35% in patients with moderate psoriasis, and 54.9% in patients with severe psoriasis.
Other predictors of PsA onset for both index-date cohorts included weight of 90 kg or greater, female gender, age group 25-65 years (compared with the age group over 65 years), and rheumatic risk factors such as wrist pain and unspecified rheumatism.
“To ensure timely diagnosis and treatment for management and prevention of PsA, patients with [psoriasis] should be routinely screened, especially those with more severe disease and other PsA risk factors,” the authors advised.
Dr. Merola and colleagues acknowledged certain limitations of their analysis, including the potential for selection bias and its reliance on EHR data which “lacked clinical measures of disease severity such as the PASI, and data on BSA were not available for all study participants; therefore, treatment groups were used as a surrogate for disease severity,” they wrote. “As a result, some patients may have been miscategorized, especially patients with severe disease who were untreated.”
The study was sponsored by Novartis. Dr. Merola disclosed that he is a consultant and/or investigator for Merck, AbbVie, Dermavant, Eli Lilly, Novartis, Janssen, UCB, Celgene, Sanofi, Regeneron, Arena, Sun Pharmaceuticals, Biogen, Pfizer, EMD Serono, Avotres, and LEO Pharma. Four authors are Novartis employees, or employees of a consulting company that provides services to Novartis; and another author disclosed serving as an investigator or consultant for several pharmaceutical companies, including Novartis.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY