User login
Large study reaffirms rare risk of TNF inhibitor–induced psoriasis in patients with RA, IBD
according to a new study published in JAMA Dermatology.
Despite this finding, the authors of the large Danish nationwide cohort study noted that TNFi-induced psoriasis is still a rare adverse event. “Practitioners and patients should be aware and observant of the potential for TNFi-associated psoriasis during TNFi treatment but keep in mind that the absolute risk appears to be low,” David Thein, MB, of the department of dermatology at Bispebjerg Hospital, University of Copenhagen, and colleagues wrote in the study.
They analyzed 109,085 patients with RA and IBD enrolled in Danish national registries between 1995 and 2018 without a previous diagnosis of psoriasis, who received either TNFi (20,910 patients) or conventional treatments (108,024 patients) and were followed for 5 years. They were a mean of 50 years old when they started treatment, 62% were women, with 87.8% of patients in the TNFi group receiving prior conventional therapy and 1% of patients in the conventional therapy group receiving prior TNFi treatment.
The investigators assessed the risk of developing any psoriasis, nonpustular psoriasis, and pustular psoriasis in the two groups using ICD-10 codes as well as a record of two consecutive prescriptions for topical vitamin D analogs.
Overall, 1,471 patients (1.4%) developed psoriasis of any type; 1,332 had non-pustular psoriasis, 127 had palmoplantar pustulosis, and 12 had generalized pustulosis.
The incidence rate of developing any psoriasis was 3.0 per 1,000 patient-years (95% confidence interval, 2.9-3.2) for patients receiving conventional therapy and 7.8 per 1,000 patient-years (95% CI, 7.5-8.9) for patients receiving TNFi treatment. Compared with conventional treatment, the risk of developing nonpustular psoriasis was twofold higher among patients receiving TNFi treatment (hazard ratio, 2.12; 95% CI, 1.87-2.40; P < .001). The risk of developing pustular psoriasis was more than sixfold higher among those on a TNFi (HR, 6.50; 95% CI, 4.60-9.23; P < .001).
Dr. Thein and colleagues estimated that the exposure needed to harm 1 additional patient was 241 patient-years for any psoriasis type, 342 patient-years for nonpustular psoriasis, and 909 patient-years for pustular psoriasis, with an estimated absolute risk difference of 5 per 1,000 patient-years.
Best evidence to date on risk
Asked to comment on the study findings, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, said that he applauded the researchers for performing this well-designed study to determine the risk of TNF inhibitor–induced psoriasis in patients with RA and IBD.
The strengths of the study include excluding patients with a history of psoriasis to rule out disease recurrence and having a large comparator group of patients with IBD and RA who were taking medications other than TNF inhibitors, while one limitation was the potential accuracy of the ICD-10 codes used as the basis for diagnosing psoriasis. “It’s probably closer to the truth of what the true risk is compared to studies done in the past,” he said in an interview.
Dr. Fernandez noted that the results aren’t likely to change how dermatologists, rheumatologists, or gastroenterologists practice, but the message to stay the course in initially treating TNFi-induced psoriasis also holds value. “We don’t need to change anything in our clinical practice when it comes to TNF-alpha inhibitors.”
For patients with RA or IBD who develop TNFi-induced psoriasis with disease that is well controlled with TNFi treatment, keeping them on that treatment is a priority, Dr. Fernandez explained. “The first and foremost goal is, if the TNF inhibitor is working very well to control the disease that it was prescribed for, then you exhaust your efforts to try to control the psoriasis and allow those patients to stay on the TNF inhibitor.”
In his experience, most patients with RA and IBD who develop TNFi-induced psoriasis are controlled with topical medications. Switching to another TNFi is not recommended, he noted, as patients are “likely to have that reaction with any TNF inhibitor.”
However, Dr. Fernandez said that won’t be an option for all patients with RA and IBD. “In some patients you do simply have to stop the TNF inhibitor” and try an alternative treatment with a different mechanism of action.
The cause of TNFi-induced psoriasis is still not well understood. “There certainly is evidence to support that interferon alpha production by plasmacytoid dendritic cells is playing some role in this phenomenon,” but there is “more to the story” and unanswered questions remain, Dr. Fernandez said.
What’s most interesting about this phenomenon, he added, is that “patients can develop it at any time when exposed to a TNF inhibitor.” For instance, most patients develop drug reactions within 2-3 weeks of starting a treatment, but TNFi-induced psoriasis can appear after a single dose or several years after initiating treatment.
“Why so few patients, and why is there such variability in terms of how long they’re on the TNF inhibitor before the reaction occurs?” he asked. “That really points to ... some other trigger besides exposure to the TNF inhibitor needed for the initiation of this reaction.”
He noted that it would be valuable to identify triggers – or the most likely triggers – which would be challenging, but could “potentially impact clinical practice.”
The authors reported personal and institutional relationships in the form of personal and institutional research grants, honoraria, personal fees, investigator fees paid to university, consultancies, and speaker’s bureau positions for a variety of pharmaceutical companies, data companies, hospitals, and foundations. Dr. Fernandez reported he has nonbranded speaking, consulting, and research relationships with AbbVie and Novartis; and is a consultant for UCB, Bristol-Myers Squibb, and Boehringer Ingelheim on related products.
according to a new study published in JAMA Dermatology.
Despite this finding, the authors of the large Danish nationwide cohort study noted that TNFi-induced psoriasis is still a rare adverse event. “Practitioners and patients should be aware and observant of the potential for TNFi-associated psoriasis during TNFi treatment but keep in mind that the absolute risk appears to be low,” David Thein, MB, of the department of dermatology at Bispebjerg Hospital, University of Copenhagen, and colleagues wrote in the study.
They analyzed 109,085 patients with RA and IBD enrolled in Danish national registries between 1995 and 2018 without a previous diagnosis of psoriasis, who received either TNFi (20,910 patients) or conventional treatments (108,024 patients) and were followed for 5 years. They were a mean of 50 years old when they started treatment, 62% were women, with 87.8% of patients in the TNFi group receiving prior conventional therapy and 1% of patients in the conventional therapy group receiving prior TNFi treatment.
The investigators assessed the risk of developing any psoriasis, nonpustular psoriasis, and pustular psoriasis in the two groups using ICD-10 codes as well as a record of two consecutive prescriptions for topical vitamin D analogs.
Overall, 1,471 patients (1.4%) developed psoriasis of any type; 1,332 had non-pustular psoriasis, 127 had palmoplantar pustulosis, and 12 had generalized pustulosis.
The incidence rate of developing any psoriasis was 3.0 per 1,000 patient-years (95% confidence interval, 2.9-3.2) for patients receiving conventional therapy and 7.8 per 1,000 patient-years (95% CI, 7.5-8.9) for patients receiving TNFi treatment. Compared with conventional treatment, the risk of developing nonpustular psoriasis was twofold higher among patients receiving TNFi treatment (hazard ratio, 2.12; 95% CI, 1.87-2.40; P < .001). The risk of developing pustular psoriasis was more than sixfold higher among those on a TNFi (HR, 6.50; 95% CI, 4.60-9.23; P < .001).
Dr. Thein and colleagues estimated that the exposure needed to harm 1 additional patient was 241 patient-years for any psoriasis type, 342 patient-years for nonpustular psoriasis, and 909 patient-years for pustular psoriasis, with an estimated absolute risk difference of 5 per 1,000 patient-years.
Best evidence to date on risk
Asked to comment on the study findings, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, said that he applauded the researchers for performing this well-designed study to determine the risk of TNF inhibitor–induced psoriasis in patients with RA and IBD.
The strengths of the study include excluding patients with a history of psoriasis to rule out disease recurrence and having a large comparator group of patients with IBD and RA who were taking medications other than TNF inhibitors, while one limitation was the potential accuracy of the ICD-10 codes used as the basis for diagnosing psoriasis. “It’s probably closer to the truth of what the true risk is compared to studies done in the past,” he said in an interview.
Dr. Fernandez noted that the results aren’t likely to change how dermatologists, rheumatologists, or gastroenterologists practice, but the message to stay the course in initially treating TNFi-induced psoriasis also holds value. “We don’t need to change anything in our clinical practice when it comes to TNF-alpha inhibitors.”
For patients with RA or IBD who develop TNFi-induced psoriasis with disease that is well controlled with TNFi treatment, keeping them on that treatment is a priority, Dr. Fernandez explained. “The first and foremost goal is, if the TNF inhibitor is working very well to control the disease that it was prescribed for, then you exhaust your efforts to try to control the psoriasis and allow those patients to stay on the TNF inhibitor.”
In his experience, most patients with RA and IBD who develop TNFi-induced psoriasis are controlled with topical medications. Switching to another TNFi is not recommended, he noted, as patients are “likely to have that reaction with any TNF inhibitor.”
However, Dr. Fernandez said that won’t be an option for all patients with RA and IBD. “In some patients you do simply have to stop the TNF inhibitor” and try an alternative treatment with a different mechanism of action.
The cause of TNFi-induced psoriasis is still not well understood. “There certainly is evidence to support that interferon alpha production by plasmacytoid dendritic cells is playing some role in this phenomenon,” but there is “more to the story” and unanswered questions remain, Dr. Fernandez said.
What’s most interesting about this phenomenon, he added, is that “patients can develop it at any time when exposed to a TNF inhibitor.” For instance, most patients develop drug reactions within 2-3 weeks of starting a treatment, but TNFi-induced psoriasis can appear after a single dose or several years after initiating treatment.
“Why so few patients, and why is there such variability in terms of how long they’re on the TNF inhibitor before the reaction occurs?” he asked. “That really points to ... some other trigger besides exposure to the TNF inhibitor needed for the initiation of this reaction.”
He noted that it would be valuable to identify triggers – or the most likely triggers – which would be challenging, but could “potentially impact clinical practice.”
The authors reported personal and institutional relationships in the form of personal and institutional research grants, honoraria, personal fees, investigator fees paid to university, consultancies, and speaker’s bureau positions for a variety of pharmaceutical companies, data companies, hospitals, and foundations. Dr. Fernandez reported he has nonbranded speaking, consulting, and research relationships with AbbVie and Novartis; and is a consultant for UCB, Bristol-Myers Squibb, and Boehringer Ingelheim on related products.
according to a new study published in JAMA Dermatology.
Despite this finding, the authors of the large Danish nationwide cohort study noted that TNFi-induced psoriasis is still a rare adverse event. “Practitioners and patients should be aware and observant of the potential for TNFi-associated psoriasis during TNFi treatment but keep in mind that the absolute risk appears to be low,” David Thein, MB, of the department of dermatology at Bispebjerg Hospital, University of Copenhagen, and colleagues wrote in the study.
They analyzed 109,085 patients with RA and IBD enrolled in Danish national registries between 1995 and 2018 without a previous diagnosis of psoriasis, who received either TNFi (20,910 patients) or conventional treatments (108,024 patients) and were followed for 5 years. They were a mean of 50 years old when they started treatment, 62% were women, with 87.8% of patients in the TNFi group receiving prior conventional therapy and 1% of patients in the conventional therapy group receiving prior TNFi treatment.
The investigators assessed the risk of developing any psoriasis, nonpustular psoriasis, and pustular psoriasis in the two groups using ICD-10 codes as well as a record of two consecutive prescriptions for topical vitamin D analogs.
Overall, 1,471 patients (1.4%) developed psoriasis of any type; 1,332 had non-pustular psoriasis, 127 had palmoplantar pustulosis, and 12 had generalized pustulosis.
The incidence rate of developing any psoriasis was 3.0 per 1,000 patient-years (95% confidence interval, 2.9-3.2) for patients receiving conventional therapy and 7.8 per 1,000 patient-years (95% CI, 7.5-8.9) for patients receiving TNFi treatment. Compared with conventional treatment, the risk of developing nonpustular psoriasis was twofold higher among patients receiving TNFi treatment (hazard ratio, 2.12; 95% CI, 1.87-2.40; P < .001). The risk of developing pustular psoriasis was more than sixfold higher among those on a TNFi (HR, 6.50; 95% CI, 4.60-9.23; P < .001).
Dr. Thein and colleagues estimated that the exposure needed to harm 1 additional patient was 241 patient-years for any psoriasis type, 342 patient-years for nonpustular psoriasis, and 909 patient-years for pustular psoriasis, with an estimated absolute risk difference of 5 per 1,000 patient-years.
Best evidence to date on risk
Asked to comment on the study findings, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, said that he applauded the researchers for performing this well-designed study to determine the risk of TNF inhibitor–induced psoriasis in patients with RA and IBD.
The strengths of the study include excluding patients with a history of psoriasis to rule out disease recurrence and having a large comparator group of patients with IBD and RA who were taking medications other than TNF inhibitors, while one limitation was the potential accuracy of the ICD-10 codes used as the basis for diagnosing psoriasis. “It’s probably closer to the truth of what the true risk is compared to studies done in the past,” he said in an interview.
Dr. Fernandez noted that the results aren’t likely to change how dermatologists, rheumatologists, or gastroenterologists practice, but the message to stay the course in initially treating TNFi-induced psoriasis also holds value. “We don’t need to change anything in our clinical practice when it comes to TNF-alpha inhibitors.”
For patients with RA or IBD who develop TNFi-induced psoriasis with disease that is well controlled with TNFi treatment, keeping them on that treatment is a priority, Dr. Fernandez explained. “The first and foremost goal is, if the TNF inhibitor is working very well to control the disease that it was prescribed for, then you exhaust your efforts to try to control the psoriasis and allow those patients to stay on the TNF inhibitor.”
In his experience, most patients with RA and IBD who develop TNFi-induced psoriasis are controlled with topical medications. Switching to another TNFi is not recommended, he noted, as patients are “likely to have that reaction with any TNF inhibitor.”
However, Dr. Fernandez said that won’t be an option for all patients with RA and IBD. “In some patients you do simply have to stop the TNF inhibitor” and try an alternative treatment with a different mechanism of action.
The cause of TNFi-induced psoriasis is still not well understood. “There certainly is evidence to support that interferon alpha production by plasmacytoid dendritic cells is playing some role in this phenomenon,” but there is “more to the story” and unanswered questions remain, Dr. Fernandez said.
What’s most interesting about this phenomenon, he added, is that “patients can develop it at any time when exposed to a TNF inhibitor.” For instance, most patients develop drug reactions within 2-3 weeks of starting a treatment, but TNFi-induced psoriasis can appear after a single dose or several years after initiating treatment.
“Why so few patients, and why is there such variability in terms of how long they’re on the TNF inhibitor before the reaction occurs?” he asked. “That really points to ... some other trigger besides exposure to the TNF inhibitor needed for the initiation of this reaction.”
He noted that it would be valuable to identify triggers – or the most likely triggers – which would be challenging, but could “potentially impact clinical practice.”
The authors reported personal and institutional relationships in the form of personal and institutional research grants, honoraria, personal fees, investigator fees paid to university, consultancies, and speaker’s bureau positions for a variety of pharmaceutical companies, data companies, hospitals, and foundations. Dr. Fernandez reported he has nonbranded speaking, consulting, and research relationships with AbbVie and Novartis; and is a consultant for UCB, Bristol-Myers Squibb, and Boehringer Ingelheim on related products.
FROM JAMA DERMATOLOGY
Autoimmune disease linked to better late-stage breast cancer survival
CHICAGO – Comorbid autoimmune disease is associated with a greater chance of survival among women with stage IV breast cancer, according to a retrospective study presented at the annual meeting of the American Society of Clinical Oncology.
“It’s counterintuitive that, if you have two diseases instead of one, that you live longer, so then we had to scratch our heads a little bit and think about why these people are living longer,” said lead author Demitrios Dedousis, MD, University Hospitals, Case Medical Center, Cleveland.
Dr. Dedousis and colleagues conducted a retrospective analysis of patients from Surveillance, Epidemiology, and End Results–Medicare databases between 2007 and 2014 with breast cancer. The study included data from 137,324 patients diagnosed between 2007 and 2012, before the widespread use of immunotherapy. 27% of patients had an autoimmune disease, most commonly rheumatoid arthritis (23%), psoriasis (2.4%), and systemic lupus erythematosus (1.1%).
When all patients were included in the analysis, those with autoimmune disorders had slightly longer survival times, but these weren’t clinically significant. A subanalysis found a greater difference in survival.
The association appears more pronounced in metastatic cancer. Patients with stage 4 breast cancer and autoimmune disease had a longer mean overall survival (36 months vs. 30 months; hazard ratio, 1.46; P < .0001. Cancer-specific survival: HR, 1.39; P < .0001). Patients with autoimmune disease and stage 1-3 breast cancer had lower overall survival (P < .0001, P < 0.0001, and P = 0.026 respectively), compared with patients without autoimmune disease.
“What we thought was happening is that the lack of increased survival in stages 1 through 3 was hiding the increase in survival among the stage IV patients when looking at the overall cohort,” Dr. Dedousis said.
The retrospective nature of the study makes it impossible to draw any firm conclusions about causation. It could be that patients who have already been diagnosed with an autoimmune disease are more vigilant about going to health checkups. “There are other possible explanations, but the one that’s most interesting to us is that their immune system is involved in fighting the cancer. Our study certainly didn’t prove that, but it’s suggesting that’s a possibility,” Dr. Dedousis said.
He and his coauthors anticipate conducting similar studies in other cancers to see if there are similar relationships. Some preliminary work has already suggested something similar in lung cancer. “I think demonstrating this in a few kinds of cancer goes part of the way towards showing that this is a real biological phenomenon,” he said.
Another research avenue is to examine the immune systems and pathology specimens in patients with both an autoimmune disease and cancer to see if there is a greater immune response within the tumor. If so, that could suggest new immunotherapy strategies.
Another possibility is to look at the specific immune pathways within “protective” autoimmune conditions. “For the sake of argument, if we find a particular autoimmune condition is improving survival across multiple kinds of cancers, we could look at those pathways that are specifically involved in that autoimmune condition. It might help us identify a target for drug development,” Dr. Dedousis said.
Asked why a potential benefit might be more apparent in late-stage disease, he suggested that, in early-stage breast cancer, surgery and other treatments may be so effective that the immune system’s role only rarely makes a difference. It could play a larger role in late-stage disease when there are less effective therapies. It could also be that the immune system doesn’t recognize the cancer until it has spread beyond the regional lymph nodes on its way to metastasizing.
According to the National Cancer Institute, 10%-30% of people with cancer also have an autoimmune disease.
Dr. Dedousis has no relevant financial disclosures.
CHICAGO – Comorbid autoimmune disease is associated with a greater chance of survival among women with stage IV breast cancer, according to a retrospective study presented at the annual meeting of the American Society of Clinical Oncology.
“It’s counterintuitive that, if you have two diseases instead of one, that you live longer, so then we had to scratch our heads a little bit and think about why these people are living longer,” said lead author Demitrios Dedousis, MD, University Hospitals, Case Medical Center, Cleveland.
Dr. Dedousis and colleagues conducted a retrospective analysis of patients from Surveillance, Epidemiology, and End Results–Medicare databases between 2007 and 2014 with breast cancer. The study included data from 137,324 patients diagnosed between 2007 and 2012, before the widespread use of immunotherapy. 27% of patients had an autoimmune disease, most commonly rheumatoid arthritis (23%), psoriasis (2.4%), and systemic lupus erythematosus (1.1%).
When all patients were included in the analysis, those with autoimmune disorders had slightly longer survival times, but these weren’t clinically significant. A subanalysis found a greater difference in survival.
The association appears more pronounced in metastatic cancer. Patients with stage 4 breast cancer and autoimmune disease had a longer mean overall survival (36 months vs. 30 months; hazard ratio, 1.46; P < .0001. Cancer-specific survival: HR, 1.39; P < .0001). Patients with autoimmune disease and stage 1-3 breast cancer had lower overall survival (P < .0001, P < 0.0001, and P = 0.026 respectively), compared with patients without autoimmune disease.
“What we thought was happening is that the lack of increased survival in stages 1 through 3 was hiding the increase in survival among the stage IV patients when looking at the overall cohort,” Dr. Dedousis said.
The retrospective nature of the study makes it impossible to draw any firm conclusions about causation. It could be that patients who have already been diagnosed with an autoimmune disease are more vigilant about going to health checkups. “There are other possible explanations, but the one that’s most interesting to us is that their immune system is involved in fighting the cancer. Our study certainly didn’t prove that, but it’s suggesting that’s a possibility,” Dr. Dedousis said.
He and his coauthors anticipate conducting similar studies in other cancers to see if there are similar relationships. Some preliminary work has already suggested something similar in lung cancer. “I think demonstrating this in a few kinds of cancer goes part of the way towards showing that this is a real biological phenomenon,” he said.
Another research avenue is to examine the immune systems and pathology specimens in patients with both an autoimmune disease and cancer to see if there is a greater immune response within the tumor. If so, that could suggest new immunotherapy strategies.
Another possibility is to look at the specific immune pathways within “protective” autoimmune conditions. “For the sake of argument, if we find a particular autoimmune condition is improving survival across multiple kinds of cancers, we could look at those pathways that are specifically involved in that autoimmune condition. It might help us identify a target for drug development,” Dr. Dedousis said.
Asked why a potential benefit might be more apparent in late-stage disease, he suggested that, in early-stage breast cancer, surgery and other treatments may be so effective that the immune system’s role only rarely makes a difference. It could play a larger role in late-stage disease when there are less effective therapies. It could also be that the immune system doesn’t recognize the cancer until it has spread beyond the regional lymph nodes on its way to metastasizing.
According to the National Cancer Institute, 10%-30% of people with cancer also have an autoimmune disease.
Dr. Dedousis has no relevant financial disclosures.
CHICAGO – Comorbid autoimmune disease is associated with a greater chance of survival among women with stage IV breast cancer, according to a retrospective study presented at the annual meeting of the American Society of Clinical Oncology.
“It’s counterintuitive that, if you have two diseases instead of one, that you live longer, so then we had to scratch our heads a little bit and think about why these people are living longer,” said lead author Demitrios Dedousis, MD, University Hospitals, Case Medical Center, Cleveland.
Dr. Dedousis and colleagues conducted a retrospective analysis of patients from Surveillance, Epidemiology, and End Results–Medicare databases between 2007 and 2014 with breast cancer. The study included data from 137,324 patients diagnosed between 2007 and 2012, before the widespread use of immunotherapy. 27% of patients had an autoimmune disease, most commonly rheumatoid arthritis (23%), psoriasis (2.4%), and systemic lupus erythematosus (1.1%).
When all patients were included in the analysis, those with autoimmune disorders had slightly longer survival times, but these weren’t clinically significant. A subanalysis found a greater difference in survival.
The association appears more pronounced in metastatic cancer. Patients with stage 4 breast cancer and autoimmune disease had a longer mean overall survival (36 months vs. 30 months; hazard ratio, 1.46; P < .0001. Cancer-specific survival: HR, 1.39; P < .0001). Patients with autoimmune disease and stage 1-3 breast cancer had lower overall survival (P < .0001, P < 0.0001, and P = 0.026 respectively), compared with patients without autoimmune disease.
“What we thought was happening is that the lack of increased survival in stages 1 through 3 was hiding the increase in survival among the stage IV patients when looking at the overall cohort,” Dr. Dedousis said.
The retrospective nature of the study makes it impossible to draw any firm conclusions about causation. It could be that patients who have already been diagnosed with an autoimmune disease are more vigilant about going to health checkups. “There are other possible explanations, but the one that’s most interesting to us is that their immune system is involved in fighting the cancer. Our study certainly didn’t prove that, but it’s suggesting that’s a possibility,” Dr. Dedousis said.
He and his coauthors anticipate conducting similar studies in other cancers to see if there are similar relationships. Some preliminary work has already suggested something similar in lung cancer. “I think demonstrating this in a few kinds of cancer goes part of the way towards showing that this is a real biological phenomenon,” he said.
Another research avenue is to examine the immune systems and pathology specimens in patients with both an autoimmune disease and cancer to see if there is a greater immune response within the tumor. If so, that could suggest new immunotherapy strategies.
Another possibility is to look at the specific immune pathways within “protective” autoimmune conditions. “For the sake of argument, if we find a particular autoimmune condition is improving survival across multiple kinds of cancers, we could look at those pathways that are specifically involved in that autoimmune condition. It might help us identify a target for drug development,” Dr. Dedousis said.
Asked why a potential benefit might be more apparent in late-stage disease, he suggested that, in early-stage breast cancer, surgery and other treatments may be so effective that the immune system’s role only rarely makes a difference. It could play a larger role in late-stage disease when there are less effective therapies. It could also be that the immune system doesn’t recognize the cancer until it has spread beyond the regional lymph nodes on its way to metastasizing.
According to the National Cancer Institute, 10%-30% of people with cancer also have an autoimmune disease.
Dr. Dedousis has no relevant financial disclosures.
AT ASCO 2022
Cutaneous Body Image: How the Mental Health Benefits of Treating Dermatologic Disease Support Military Readiness in Service Members
According to the US Department of Defense, the term readiness refers to the ability to recruit, train, deploy, and sustain military forces that will be ready to “fight tonight” and succeed in combat. Readiness is a top priority for military medicine, which functions to diagnose, treat, and rehabilitate service members so that they can return to the fight. This central concept drives programs across the military—from operational training events to the establishment of medical and dental standards. Readiness is tracked and scrutinized constantly, and although it is a shared responsibility, efforts to increase and sustain readiness often fall on support staff and military medical providers.
In recent years, there has been a greater awareness of the negative effects of mental illness, low morale, and suicidality on military readiness. In 2013, suicide accounted for 28.1% of all deaths that occurred in the US Armed Forces.1 Put frankly, suicide was one of the leading causes of death among military members.
The most recent Marine Corps Order regarding the Marine Corps Suicide Prevention Program stated that “suicidal behaviors are a barrier to readiness that have lasting effects on Marines and Service Members attached to Marine Commands. . .Families, and the Marine Corps.” It goes on to say that “[e]ffective suicide prevention requires coordinated efforts within a prevention framework dedicated to promoting mental, physical, spiritual, and social fitness. . .[and] mitigating stressors that interfere with mission readiness.”2 This statement supports the notion that preventing suicide is not just about treating mental illness; it also involves maximizing physical, spiritual, and social fitness. Although it is well established that various mental health disorders are associated with an increased risk for suicide, it is worth noting that, in one study, only half of individuals who died by suicide had a mental health disorder diagnosed prior to their death.3 These statistics translate to the military. The 2015 Department of Defense Suicide Event Report noted that only 28% of service members who died by suicide and 22% of members with attempted suicide had been documented as having sought mental health care and disclosed their potential for self-harm prior to the event.1,4 In 2018, a study published by Ursano et al5 showed that 36.3% of US soldiers with a documented suicide attempt (N=9650) had no prior mental health diagnoses.
Expanding the scope to include mental health issues in general, only 29% of service members who reported experiencing a mental health problem actually sought mental health care in that same period. Overall, approximately 40% of service members with a reported perceived need for mental health care actually sought care over their entire course of service time,1 which raises concern for a large population of undiagnosed and undertreated mental illnesses across the military. In response to these statistics, Reger et al3 posited that it is “essential that suicide prevention efforts move outside the silo of mental health.” The authors went on to challenge health care providers across all specialties and civilians alike to take responsibility in understanding, recognizing, and mitigating risk factors for suicide in the general population.3 Although treating a service member’s acne or offering to stand duty for a service member who has been under a great deal of stress in their personal life may appear to be indirect ways of reducing suicide in the US military, they actually may be the most critical means of prevention in a culture that emphasizes resilience and self-reliance, where seeking help for mental health struggles could be perceived as weakness.1
In this review article, we discuss the concept of cutaneous body image (CBI) and its associated outcomes on health, satisfaction, and quality of life in military service members. We then examine the intersections between common dermatologic conditions, CBI, and mental health and explore the ability and role of the military dermatologist to serve as a positive influence on military readiness.
What is cutaneous body image?
Cutaneous body image is “the individual’s mental perception of his or her skin and its appendages (ie, hair, nails).”6 It is measured objectively using the Cutaneous Body Image Scale, a questionnaire that includes 7 items related to the overall satisfaction with the appearance of skin, color of skin, skin of the face, complexion of the face, hair, fingernails, and toenails. Each question is rated using a 10-point Likert scale (0=not at all; 10=very markedly).6
Some degree of CBI dissatisfaction is expected and has been shown in the general population at large; for example, more than 56% of women older than 30 years report some degree of dissatisfaction with their skin. Similarly, data from the American Society of Plastic Surgeons showed that while 10.9 million cosmetic procedures were performed in 2006, 9.1 million of them involved minimally invasive procedures such as botulinum toxin type A injections with the purpose of skin rejuvenation and improvement of facial appearance.7 However, lower than average CBI can contribute to considerable psychosocial morbidity. Dissatisfaction with CBI is associated with self-consciousness, feelings of inferiority, and social exclusion. These symptoms can be grouped into a construct called interpersonal sensitivity (IS). A 2013 study by Gupta and Gupta6 investigated the relationship between CBI, IS, and suicidal ideation among 312 consenting nonclinical participants in Canada. The study found that greater dissatisfaction with an individual’s CBI correlated to increased IS and increased rates of suicidal ideation and intentional self-injury.6
Cutaneous body image is particularly relevant to dermatologists, as many common dermatoses can cause cosmetically disfiguring skin conditions; for example, acne and rosacea have the propensity to cause notable disfigurement to the facial unit. Other common conditions such as atopic dermatitis or psoriasis can flare with stress and thereby throw patients into a vicious cycle of physical and psychosocial stress caused by social stigma, cosmetic disfigurement, and reduced CBI, in turn leading to worsening of the disease at hand. Dermatologists need to be aware that common dermatoses can impact a patient’s mental health via poor CBI.8 Similarly, dermatologists may be empowered by the awareness that treating common dermatoses, especially those associated with poor cosmesis, have 2-fold benefits—on the skin condition itself and on the patient’s mental health.
How are common dermatoses associated with mental health?
Acne—Acne is one of the most common skin diseases, so much so that in many cases acne has become an accepted and expected part of adolescence and young adulthood. Studies estimate that 85% of the US population aged 12 to 25 years have acne.9 For some adults, acne persists even longer, with 1% to 5% of adults reporting to have active lesions at 40 years of age.10 Acne is a multifactorial skin disease of the pilosebaceous unit that results in the development of inflammatory papules, pustules, and cysts. These lesions are most common on the face but can extend to other areas of the body, such as the chest and back.11 Although the active lesions can be painful and disfiguring, if left untreated, acne may lead to permanent disfigurement and scarring, which can have long-lasting psychosocial impacts.
Individuals with acne have an increased likelihood of self-consciousness, social isolation, depression, and suicidal ideation. This relationship has been well established for decades. In the 1990s, a small study reported that 7 of 16 (43.8%) cases of completed suicide in dermatology patients were in patients with acne.12 In a recent meta-analysis including 2,276,798 participants across 5 separate studies, researchers found that suicide was positively associated with acne, carrying an odds ratio of 1.50 (95% CI, 1.09-2.06).13
Rosacea—Rosacea is a common chronic inflammatory skin disease characterized by facial erythema, telangiectasia, phymatous changes, papules, pustules, and ocular irritation. The estimated worldwide prevalence is 5.5%.14 In addition to discomfort and irritation of the skin and eyes, rosacea often carries a higher risk of psychological and psychosocial distress due to its potentially disfiguring nature. Rosacea patients are at greater risk for having anxiety disorders and depression,15 and a 2018 study by Alinia et al16 showed that there is a direct relationship between rosacea severity and the actual level of depression.Although disease improvement certainly leads to improvements in quality of life and psychosocial status, Alinia et al16 noted that depression often is associated with poor treatment adherence due to poor motivation and hopelessness. It is critical that dermatologists are aware of these associations and maintain close follow-up with patients, even when the condition is not life-threatening, such as rosacea.
Hidradenitis Suppurativa—Hidradenitis suppurativa (HS) is a chronic inflammatory disease of the pilosebaceous unit that is characterized by the development of painful, malodorous, draining abscesses, fistulas, sinus tracts, and scars in sensitive areas such as the axillae, breasts, groin, and perineum.17 In severe cases, surgery may be required to excise affected areas. Compared to other cutaneous disease, HS is considered one of the most life-impacting disorders.18 The physical symptoms themselves often are debilitating, and patients often report considerable psychosocial and psychological impairment with decreased quality of life. Major depression frequently is noted, with 1 in 4 adults with HS also being depressed. In a large cross-sectional analysis of 38,140 adults and 1162 pediatric patients with HS, Wright et al17 reported the prevalence of depression among adults with HS as 30.0% compared to 16.9% in healthy controls. In children, the prevalence of depression was 11.7% compared to 4.1% in the general population.17 Similarly, 1 out of every 5 patients with HS experiences anxiety.18
In the military population, HS often can be duty limiting. The disease requires constant attention to wound care and frequent medical visits. For many service members operating in field training or combat environments, opportunities for and access to showers and basic hygiene is limited. Uniforms and additional necessary combat gear often are thick and occlusive. Taken as a whole, these factors may contribute to worsening of the disease and in severe cases are simply not conducive to the successful management of the condition. However, given the most commonly involved body areas and the nature of the disease, many service members with HS may feel embarrassed to disclose their condition. In uniform, the disease is not easily visible, and for unaware persons, the frequency of medical visits and limited duty status may seem unnecessary. This perception of a service member’s lack of productivity due to an unseen disease may further add to the psychosocial stress they experience.
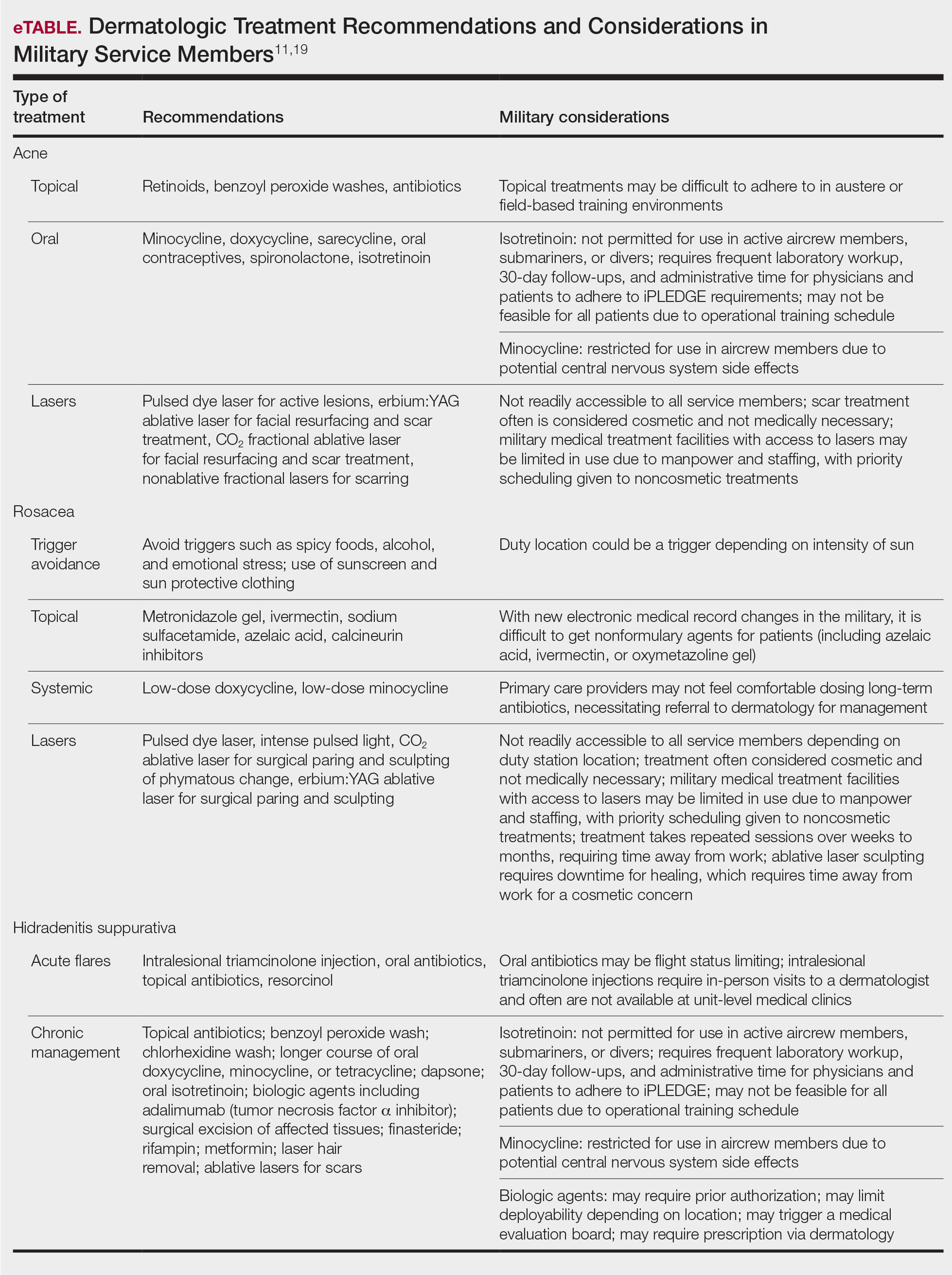
The treatments for acne, rosacea, and HS are outlined in the eTable.11,19 Also noted are specific considerations when managing an active-duty service member due to various operational duty restrictions and constraints.
Final Thoughts
Maintaining readiness in the military is essential to the ability to not only “fight tonight” but also to win tonight in whatever operational or combat mission a service member may be. Although many factors impact readiness, the rates of suicide within the armed forces cannot be ignored. Suicide not only eliminates the readiness of the deceased service member but has lasting ripple effects on the overall readiness of their unit and command at large. Most suicides in the military occur in personnel with no prior documented mental health diagnoses or treatment. Therefore, it is the responsibility of all service members to recognize and mitigate stressors and risk factors that may lead to mental health distress and suicidality. In the medical corps, this translates to a responsibility of all medical specialists to recognize and understand unique risk factors for suicidality and to do as much as they can to reduce these risks. For military dermatologists and for civilian physicians treating military service members, it is imperative to predict and understand the relationship between common dermatoses; reduced satisfaction with CBI; and increased risk for mental health illness, self-harm, and suicide. Military dermatologists, as well as other specialists, may be limited in the care they are able to provide due to manpower, staffing, demand, and institutional guidelines; however, to better serve those who serve in a holistic manner, consideration must be given to rethink what is “medically essential” and “cosmetic” and leverage the available skills, techniques, and equipment to increase the readiness of the force.

- Ghahramanlou-Holloway M, LaCroix JM, Koss K, et al. Outpatient mental health treatment utilization and military career impact in the United States Marine Corps. Int J Environ Res Public Health. 2018;15:828. doi:10.3390/ijerph15040828
- Ottignon DA. Marine Corps Suicide Prevention System (MCSPS). Marine Corps Order 1720.2A. 2021. Headquarters United States Marine Corps. Published August 2, 2021. Accessed May 25, 2022. https://www.marines.mil/Portals/1/Publications/MCO%201720.2A.pdf?ver=QPxZ_qMS-X-d037B65N9Tg%3d%3d
- Reger MA, Smolenski DJ, Carter SP. Suicide prevention in the US Army: a mission for more than mental health clinicians. JAMA Psychiatry. 2018;75:991-992. doi:10.1001/jamapsychiatry.2018.2042
- Pruitt LD, Smolenski DJ, Bush NE, et al. Department of Defense Suicide Event Report Calendar Year 2015 Annual Report. National Center for Telehealth & Technology (T2); 2016. Accessed May 20, 2022. https://health.mil/Military-Health-Topics/Centers-of-Excellence/Psychological-Health-Center-of-Excellence/Department-of-Defense-Suicide-Event-Report
- Ursano RJ, Kessler RC, Naifeh JA, et al. Risk factors associated with attempted suicide among US Army soldiers without a history of mental health diagnosis. JAMA Psychiatry. 2018;75:1022-1032. doi:10.1001/jamapsychiatry.2018.2069
- Gupta MA, Gupta AK. Cutaneous body image dissatisfaction and suicidal ideation: mediation by interpersonal sensitivity. J Psychosom Res. 2013;75:55-59. doi:10.1016/j.jpsychores.2013.01.015
- Gupta MA, Gupta AK. Evaluation of cutaneous body image dissatisfaction in the dermatology patient. Clin Dermatol. 2013;31:72-79. doi:10.1016/j.clindermatol.2011.11.010
- Hinkley SB, Holub SC, Menter A. The validity of cutaneous body image as a construct and as a mediator of the relationship between cutaneous disease and mental health. Dermatol Ther (Heidelb). 2020;10:203-211. doi:10.1007/s13555-020-00351-5
- Stamu-O’Brien C, Jafferany M, Carniciu S, et al. Psychodermatology of acne: psychological aspects and effects of acne vulgaris. J Cosmet Dermatol. 2021;20:1080-1083. doi:10.1111/jocd.13765
- Sood S, Jafferany M, Vinaya Kumar S. Depression, psychiatric comorbidities, and psychosocial implications associated with acne vulgaris. J Cosmet Dermatol. 2020;19:3177-3182. doi:10.1111/jocd.13753
- Brahe C, Peters K. Fighting acne for the fighting forces. Cutis. 2020;106:18-20, 22. doi:10.12788/cutis.0057
- Cotterill JA, Cunliffe WJ. Suicide in dermatological patients. Br J Dermatol. 1997;137:246-250.
- Xu S, Zhu Y, Hu H, et al. The analysis of acne increasing suicide risk. Medicine (Baltimore). 2021;100:E26035. doi:10.1097/MD.0000000000026035
- Chen M, Deng Z, Huang Y, et al. Prevalence and risk factors of anxiety and depression in rosacea patients: a cross-sectional study in China [published online June 16, 2021]. Front Psychiatry. doi:10.3389/fpsyt.2021.659171
- Incel Uysal P, Akdogan N, Hayran Y, et al. Rosacea associated with increased risk of generalized anxiety disorder: a case-control study of prevalence and risk of anxiety in patients with rosacea. An Bras Dermatol. 2019;94:704-709. doi:10.1016/j.abd.2019.03.002
- Alinia H, Cardwell LA, Tuchayi SM, et al. Screening for depression in rosacea patients. Cutis. 2018;102:36-38.
- Wright S, Strunk A, Garg A. Prevalence of depression among children, adolescents, and adults with hidradenitis suppurativa [published online June 16, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.06.843
- Misitzis A, Goldust M, Jafferany M, et al. Psychiatric comorbidities in patients with hidradenitis suppurativa. Dermatol Ther. 2020;33:E13541. doi:10.1111/dth.13541
- Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017.
According to the US Department of Defense, the term readiness refers to the ability to recruit, train, deploy, and sustain military forces that will be ready to “fight tonight” and succeed in combat. Readiness is a top priority for military medicine, which functions to diagnose, treat, and rehabilitate service members so that they can return to the fight. This central concept drives programs across the military—from operational training events to the establishment of medical and dental standards. Readiness is tracked and scrutinized constantly, and although it is a shared responsibility, efforts to increase and sustain readiness often fall on support staff and military medical providers.
In recent years, there has been a greater awareness of the negative effects of mental illness, low morale, and suicidality on military readiness. In 2013, suicide accounted for 28.1% of all deaths that occurred in the US Armed Forces.1 Put frankly, suicide was one of the leading causes of death among military members.
The most recent Marine Corps Order regarding the Marine Corps Suicide Prevention Program stated that “suicidal behaviors are a barrier to readiness that have lasting effects on Marines and Service Members attached to Marine Commands. . .Families, and the Marine Corps.” It goes on to say that “[e]ffective suicide prevention requires coordinated efforts within a prevention framework dedicated to promoting mental, physical, spiritual, and social fitness. . .[and] mitigating stressors that interfere with mission readiness.”2 This statement supports the notion that preventing suicide is not just about treating mental illness; it also involves maximizing physical, spiritual, and social fitness. Although it is well established that various mental health disorders are associated with an increased risk for suicide, it is worth noting that, in one study, only half of individuals who died by suicide had a mental health disorder diagnosed prior to their death.3 These statistics translate to the military. The 2015 Department of Defense Suicide Event Report noted that only 28% of service members who died by suicide and 22% of members with attempted suicide had been documented as having sought mental health care and disclosed their potential for self-harm prior to the event.1,4 In 2018, a study published by Ursano et al5 showed that 36.3% of US soldiers with a documented suicide attempt (N=9650) had no prior mental health diagnoses.
Expanding the scope to include mental health issues in general, only 29% of service members who reported experiencing a mental health problem actually sought mental health care in that same period. Overall, approximately 40% of service members with a reported perceived need for mental health care actually sought care over their entire course of service time,1 which raises concern for a large population of undiagnosed and undertreated mental illnesses across the military. In response to these statistics, Reger et al3 posited that it is “essential that suicide prevention efforts move outside the silo of mental health.” The authors went on to challenge health care providers across all specialties and civilians alike to take responsibility in understanding, recognizing, and mitigating risk factors for suicide in the general population.3 Although treating a service member’s acne or offering to stand duty for a service member who has been under a great deal of stress in their personal life may appear to be indirect ways of reducing suicide in the US military, they actually may be the most critical means of prevention in a culture that emphasizes resilience and self-reliance, where seeking help for mental health struggles could be perceived as weakness.1
In this review article, we discuss the concept of cutaneous body image (CBI) and its associated outcomes on health, satisfaction, and quality of life in military service members. We then examine the intersections between common dermatologic conditions, CBI, and mental health and explore the ability and role of the military dermatologist to serve as a positive influence on military readiness.
What is cutaneous body image?
Cutaneous body image is “the individual’s mental perception of his or her skin and its appendages (ie, hair, nails).”6 It is measured objectively using the Cutaneous Body Image Scale, a questionnaire that includes 7 items related to the overall satisfaction with the appearance of skin, color of skin, skin of the face, complexion of the face, hair, fingernails, and toenails. Each question is rated using a 10-point Likert scale (0=not at all; 10=very markedly).6
Some degree of CBI dissatisfaction is expected and has been shown in the general population at large; for example, more than 56% of women older than 30 years report some degree of dissatisfaction with their skin. Similarly, data from the American Society of Plastic Surgeons showed that while 10.9 million cosmetic procedures were performed in 2006, 9.1 million of them involved minimally invasive procedures such as botulinum toxin type A injections with the purpose of skin rejuvenation and improvement of facial appearance.7 However, lower than average CBI can contribute to considerable psychosocial morbidity. Dissatisfaction with CBI is associated with self-consciousness, feelings of inferiority, and social exclusion. These symptoms can be grouped into a construct called interpersonal sensitivity (IS). A 2013 study by Gupta and Gupta6 investigated the relationship between CBI, IS, and suicidal ideation among 312 consenting nonclinical participants in Canada. The study found that greater dissatisfaction with an individual’s CBI correlated to increased IS and increased rates of suicidal ideation and intentional self-injury.6
Cutaneous body image is particularly relevant to dermatologists, as many common dermatoses can cause cosmetically disfiguring skin conditions; for example, acne and rosacea have the propensity to cause notable disfigurement to the facial unit. Other common conditions such as atopic dermatitis or psoriasis can flare with stress and thereby throw patients into a vicious cycle of physical and psychosocial stress caused by social stigma, cosmetic disfigurement, and reduced CBI, in turn leading to worsening of the disease at hand. Dermatologists need to be aware that common dermatoses can impact a patient’s mental health via poor CBI.8 Similarly, dermatologists may be empowered by the awareness that treating common dermatoses, especially those associated with poor cosmesis, have 2-fold benefits—on the skin condition itself and on the patient’s mental health.
How are common dermatoses associated with mental health?
Acne—Acne is one of the most common skin diseases, so much so that in many cases acne has become an accepted and expected part of adolescence and young adulthood. Studies estimate that 85% of the US population aged 12 to 25 years have acne.9 For some adults, acne persists even longer, with 1% to 5% of adults reporting to have active lesions at 40 years of age.10 Acne is a multifactorial skin disease of the pilosebaceous unit that results in the development of inflammatory papules, pustules, and cysts. These lesions are most common on the face but can extend to other areas of the body, such as the chest and back.11 Although the active lesions can be painful and disfiguring, if left untreated, acne may lead to permanent disfigurement and scarring, which can have long-lasting psychosocial impacts.
Individuals with acne have an increased likelihood of self-consciousness, social isolation, depression, and suicidal ideation. This relationship has been well established for decades. In the 1990s, a small study reported that 7 of 16 (43.8%) cases of completed suicide in dermatology patients were in patients with acne.12 In a recent meta-analysis including 2,276,798 participants across 5 separate studies, researchers found that suicide was positively associated with acne, carrying an odds ratio of 1.50 (95% CI, 1.09-2.06).13
Rosacea—Rosacea is a common chronic inflammatory skin disease characterized by facial erythema, telangiectasia, phymatous changes, papules, pustules, and ocular irritation. The estimated worldwide prevalence is 5.5%.14 In addition to discomfort and irritation of the skin and eyes, rosacea often carries a higher risk of psychological and psychosocial distress due to its potentially disfiguring nature. Rosacea patients are at greater risk for having anxiety disorders and depression,15 and a 2018 study by Alinia et al16 showed that there is a direct relationship between rosacea severity and the actual level of depression.Although disease improvement certainly leads to improvements in quality of life and psychosocial status, Alinia et al16 noted that depression often is associated with poor treatment adherence due to poor motivation and hopelessness. It is critical that dermatologists are aware of these associations and maintain close follow-up with patients, even when the condition is not life-threatening, such as rosacea.
Hidradenitis Suppurativa—Hidradenitis suppurativa (HS) is a chronic inflammatory disease of the pilosebaceous unit that is characterized by the development of painful, malodorous, draining abscesses, fistulas, sinus tracts, and scars in sensitive areas such as the axillae, breasts, groin, and perineum.17 In severe cases, surgery may be required to excise affected areas. Compared to other cutaneous disease, HS is considered one of the most life-impacting disorders.18 The physical symptoms themselves often are debilitating, and patients often report considerable psychosocial and psychological impairment with decreased quality of life. Major depression frequently is noted, with 1 in 4 adults with HS also being depressed. In a large cross-sectional analysis of 38,140 adults and 1162 pediatric patients with HS, Wright et al17 reported the prevalence of depression among adults with HS as 30.0% compared to 16.9% in healthy controls. In children, the prevalence of depression was 11.7% compared to 4.1% in the general population.17 Similarly, 1 out of every 5 patients with HS experiences anxiety.18
In the military population, HS often can be duty limiting. The disease requires constant attention to wound care and frequent medical visits. For many service members operating in field training or combat environments, opportunities for and access to showers and basic hygiene is limited. Uniforms and additional necessary combat gear often are thick and occlusive. Taken as a whole, these factors may contribute to worsening of the disease and in severe cases are simply not conducive to the successful management of the condition. However, given the most commonly involved body areas and the nature of the disease, many service members with HS may feel embarrassed to disclose their condition. In uniform, the disease is not easily visible, and for unaware persons, the frequency of medical visits and limited duty status may seem unnecessary. This perception of a service member’s lack of productivity due to an unseen disease may further add to the psychosocial stress they experience.
The treatments for acne, rosacea, and HS are outlined in the eTable.11,19 Also noted are specific considerations when managing an active-duty service member due to various operational duty restrictions and constraints.

Final Thoughts
Maintaining readiness in the military is essential to the ability to not only “fight tonight” but also to win tonight in whatever operational or combat mission a service member may be. Although many factors impact readiness, the rates of suicide within the armed forces cannot be ignored. Suicide not only eliminates the readiness of the deceased service member but has lasting ripple effects on the overall readiness of their unit and command at large. Most suicides in the military occur in personnel with no prior documented mental health diagnoses or treatment. Therefore, it is the responsibility of all service members to recognize and mitigate stressors and risk factors that may lead to mental health distress and suicidality. In the medical corps, this translates to a responsibility of all medical specialists to recognize and understand unique risk factors for suicidality and to do as much as they can to reduce these risks. For military dermatologists and for civilian physicians treating military service members, it is imperative to predict and understand the relationship between common dermatoses; reduced satisfaction with CBI; and increased risk for mental health illness, self-harm, and suicide. Military dermatologists, as well as other specialists, may be limited in the care they are able to provide due to manpower, staffing, demand, and institutional guidelines; however, to better serve those who serve in a holistic manner, consideration must be given to rethink what is “medically essential” and “cosmetic” and leverage the available skills, techniques, and equipment to increase the readiness of the force.

According to the US Department of Defense, the term readiness refers to the ability to recruit, train, deploy, and sustain military forces that will be ready to “fight tonight” and succeed in combat. Readiness is a top priority for military medicine, which functions to diagnose, treat, and rehabilitate service members so that they can return to the fight. This central concept drives programs across the military—from operational training events to the establishment of medical and dental standards. Readiness is tracked and scrutinized constantly, and although it is a shared responsibility, efforts to increase and sustain readiness often fall on support staff and military medical providers.
In recent years, there has been a greater awareness of the negative effects of mental illness, low morale, and suicidality on military readiness. In 2013, suicide accounted for 28.1% of all deaths that occurred in the US Armed Forces.1 Put frankly, suicide was one of the leading causes of death among military members.
The most recent Marine Corps Order regarding the Marine Corps Suicide Prevention Program stated that “suicidal behaviors are a barrier to readiness that have lasting effects on Marines and Service Members attached to Marine Commands. . .Families, and the Marine Corps.” It goes on to say that “[e]ffective suicide prevention requires coordinated efforts within a prevention framework dedicated to promoting mental, physical, spiritual, and social fitness. . .[and] mitigating stressors that interfere with mission readiness.”2 This statement supports the notion that preventing suicide is not just about treating mental illness; it also involves maximizing physical, spiritual, and social fitness. Although it is well established that various mental health disorders are associated with an increased risk for suicide, it is worth noting that, in one study, only half of individuals who died by suicide had a mental health disorder diagnosed prior to their death.3 These statistics translate to the military. The 2015 Department of Defense Suicide Event Report noted that only 28% of service members who died by suicide and 22% of members with attempted suicide had been documented as having sought mental health care and disclosed their potential for self-harm prior to the event.1,4 In 2018, a study published by Ursano et al5 showed that 36.3% of US soldiers with a documented suicide attempt (N=9650) had no prior mental health diagnoses.
Expanding the scope to include mental health issues in general, only 29% of service members who reported experiencing a mental health problem actually sought mental health care in that same period. Overall, approximately 40% of service members with a reported perceived need for mental health care actually sought care over their entire course of service time,1 which raises concern for a large population of undiagnosed and undertreated mental illnesses across the military. In response to these statistics, Reger et al3 posited that it is “essential that suicide prevention efforts move outside the silo of mental health.” The authors went on to challenge health care providers across all specialties and civilians alike to take responsibility in understanding, recognizing, and mitigating risk factors for suicide in the general population.3 Although treating a service member’s acne or offering to stand duty for a service member who has been under a great deal of stress in their personal life may appear to be indirect ways of reducing suicide in the US military, they actually may be the most critical means of prevention in a culture that emphasizes resilience and self-reliance, where seeking help for mental health struggles could be perceived as weakness.1
In this review article, we discuss the concept of cutaneous body image (CBI) and its associated outcomes on health, satisfaction, and quality of life in military service members. We then examine the intersections between common dermatologic conditions, CBI, and mental health and explore the ability and role of the military dermatologist to serve as a positive influence on military readiness.
What is cutaneous body image?
Cutaneous body image is “the individual’s mental perception of his or her skin and its appendages (ie, hair, nails).”6 It is measured objectively using the Cutaneous Body Image Scale, a questionnaire that includes 7 items related to the overall satisfaction with the appearance of skin, color of skin, skin of the face, complexion of the face, hair, fingernails, and toenails. Each question is rated using a 10-point Likert scale (0=not at all; 10=very markedly).6
Some degree of CBI dissatisfaction is expected and has been shown in the general population at large; for example, more than 56% of women older than 30 years report some degree of dissatisfaction with their skin. Similarly, data from the American Society of Plastic Surgeons showed that while 10.9 million cosmetic procedures were performed in 2006, 9.1 million of them involved minimally invasive procedures such as botulinum toxin type A injections with the purpose of skin rejuvenation and improvement of facial appearance.7 However, lower than average CBI can contribute to considerable psychosocial morbidity. Dissatisfaction with CBI is associated with self-consciousness, feelings of inferiority, and social exclusion. These symptoms can be grouped into a construct called interpersonal sensitivity (IS). A 2013 study by Gupta and Gupta6 investigated the relationship between CBI, IS, and suicidal ideation among 312 consenting nonclinical participants in Canada. The study found that greater dissatisfaction with an individual’s CBI correlated to increased IS and increased rates of suicidal ideation and intentional self-injury.6
Cutaneous body image is particularly relevant to dermatologists, as many common dermatoses can cause cosmetically disfiguring skin conditions; for example, acne and rosacea have the propensity to cause notable disfigurement to the facial unit. Other common conditions such as atopic dermatitis or psoriasis can flare with stress and thereby throw patients into a vicious cycle of physical and psychosocial stress caused by social stigma, cosmetic disfigurement, and reduced CBI, in turn leading to worsening of the disease at hand. Dermatologists need to be aware that common dermatoses can impact a patient’s mental health via poor CBI.8 Similarly, dermatologists may be empowered by the awareness that treating common dermatoses, especially those associated with poor cosmesis, have 2-fold benefits—on the skin condition itself and on the patient’s mental health.
How are common dermatoses associated with mental health?
Acne—Acne is one of the most common skin diseases, so much so that in many cases acne has become an accepted and expected part of adolescence and young adulthood. Studies estimate that 85% of the US population aged 12 to 25 years have acne.9 For some adults, acne persists even longer, with 1% to 5% of adults reporting to have active lesions at 40 years of age.10 Acne is a multifactorial skin disease of the pilosebaceous unit that results in the development of inflammatory papules, pustules, and cysts. These lesions are most common on the face but can extend to other areas of the body, such as the chest and back.11 Although the active lesions can be painful and disfiguring, if left untreated, acne may lead to permanent disfigurement and scarring, which can have long-lasting psychosocial impacts.
Individuals with acne have an increased likelihood of self-consciousness, social isolation, depression, and suicidal ideation. This relationship has been well established for decades. In the 1990s, a small study reported that 7 of 16 (43.8%) cases of completed suicide in dermatology patients were in patients with acne.12 In a recent meta-analysis including 2,276,798 participants across 5 separate studies, researchers found that suicide was positively associated with acne, carrying an odds ratio of 1.50 (95% CI, 1.09-2.06).13
Rosacea—Rosacea is a common chronic inflammatory skin disease characterized by facial erythema, telangiectasia, phymatous changes, papules, pustules, and ocular irritation. The estimated worldwide prevalence is 5.5%.14 In addition to discomfort and irritation of the skin and eyes, rosacea often carries a higher risk of psychological and psychosocial distress due to its potentially disfiguring nature. Rosacea patients are at greater risk for having anxiety disorders and depression,15 and a 2018 study by Alinia et al16 showed that there is a direct relationship between rosacea severity and the actual level of depression.Although disease improvement certainly leads to improvements in quality of life and psychosocial status, Alinia et al16 noted that depression often is associated with poor treatment adherence due to poor motivation and hopelessness. It is critical that dermatologists are aware of these associations and maintain close follow-up with patients, even when the condition is not life-threatening, such as rosacea.
Hidradenitis Suppurativa—Hidradenitis suppurativa (HS) is a chronic inflammatory disease of the pilosebaceous unit that is characterized by the development of painful, malodorous, draining abscesses, fistulas, sinus tracts, and scars in sensitive areas such as the axillae, breasts, groin, and perineum.17 In severe cases, surgery may be required to excise affected areas. Compared to other cutaneous disease, HS is considered one of the most life-impacting disorders.18 The physical symptoms themselves often are debilitating, and patients often report considerable psychosocial and psychological impairment with decreased quality of life. Major depression frequently is noted, with 1 in 4 adults with HS also being depressed. In a large cross-sectional analysis of 38,140 adults and 1162 pediatric patients with HS, Wright et al17 reported the prevalence of depression among adults with HS as 30.0% compared to 16.9% in healthy controls. In children, the prevalence of depression was 11.7% compared to 4.1% in the general population.17 Similarly, 1 out of every 5 patients with HS experiences anxiety.18
In the military population, HS often can be duty limiting. The disease requires constant attention to wound care and frequent medical visits. For many service members operating in field training or combat environments, opportunities for and access to showers and basic hygiene is limited. Uniforms and additional necessary combat gear often are thick and occlusive. Taken as a whole, these factors may contribute to worsening of the disease and in severe cases are simply not conducive to the successful management of the condition. However, given the most commonly involved body areas and the nature of the disease, many service members with HS may feel embarrassed to disclose their condition. In uniform, the disease is not easily visible, and for unaware persons, the frequency of medical visits and limited duty status may seem unnecessary. This perception of a service member’s lack of productivity due to an unseen disease may further add to the psychosocial stress they experience.
The treatments for acne, rosacea, and HS are outlined in the eTable.11,19 Also noted are specific considerations when managing an active-duty service member due to various operational duty restrictions and constraints.

Final Thoughts
Maintaining readiness in the military is essential to the ability to not only “fight tonight” but also to win tonight in whatever operational or combat mission a service member may be. Although many factors impact readiness, the rates of suicide within the armed forces cannot be ignored. Suicide not only eliminates the readiness of the deceased service member but has lasting ripple effects on the overall readiness of their unit and command at large. Most suicides in the military occur in personnel with no prior documented mental health diagnoses or treatment. Therefore, it is the responsibility of all service members to recognize and mitigate stressors and risk factors that may lead to mental health distress and suicidality. In the medical corps, this translates to a responsibility of all medical specialists to recognize and understand unique risk factors for suicidality and to do as much as they can to reduce these risks. For military dermatologists and for civilian physicians treating military service members, it is imperative to predict and understand the relationship between common dermatoses; reduced satisfaction with CBI; and increased risk for mental health illness, self-harm, and suicide. Military dermatologists, as well as other specialists, may be limited in the care they are able to provide due to manpower, staffing, demand, and institutional guidelines; however, to better serve those who serve in a holistic manner, consideration must be given to rethink what is “medically essential” and “cosmetic” and leverage the available skills, techniques, and equipment to increase the readiness of the force.

- Ghahramanlou-Holloway M, LaCroix JM, Koss K, et al. Outpatient mental health treatment utilization and military career impact in the United States Marine Corps. Int J Environ Res Public Health. 2018;15:828. doi:10.3390/ijerph15040828
- Ottignon DA. Marine Corps Suicide Prevention System (MCSPS). Marine Corps Order 1720.2A. 2021. Headquarters United States Marine Corps. Published August 2, 2021. Accessed May 25, 2022. https://www.marines.mil/Portals/1/Publications/MCO%201720.2A.pdf?ver=QPxZ_qMS-X-d037B65N9Tg%3d%3d
- Reger MA, Smolenski DJ, Carter SP. Suicide prevention in the US Army: a mission for more than mental health clinicians. JAMA Psychiatry. 2018;75:991-992. doi:10.1001/jamapsychiatry.2018.2042
- Pruitt LD, Smolenski DJ, Bush NE, et al. Department of Defense Suicide Event Report Calendar Year 2015 Annual Report. National Center for Telehealth & Technology (T2); 2016. Accessed May 20, 2022. https://health.mil/Military-Health-Topics/Centers-of-Excellence/Psychological-Health-Center-of-Excellence/Department-of-Defense-Suicide-Event-Report
- Ursano RJ, Kessler RC, Naifeh JA, et al. Risk factors associated with attempted suicide among US Army soldiers without a history of mental health diagnosis. JAMA Psychiatry. 2018;75:1022-1032. doi:10.1001/jamapsychiatry.2018.2069
- Gupta MA, Gupta AK. Cutaneous body image dissatisfaction and suicidal ideation: mediation by interpersonal sensitivity. J Psychosom Res. 2013;75:55-59. doi:10.1016/j.jpsychores.2013.01.015
- Gupta MA, Gupta AK. Evaluation of cutaneous body image dissatisfaction in the dermatology patient. Clin Dermatol. 2013;31:72-79. doi:10.1016/j.clindermatol.2011.11.010
- Hinkley SB, Holub SC, Menter A. The validity of cutaneous body image as a construct and as a mediator of the relationship between cutaneous disease and mental health. Dermatol Ther (Heidelb). 2020;10:203-211. doi:10.1007/s13555-020-00351-5
- Stamu-O’Brien C, Jafferany M, Carniciu S, et al. Psychodermatology of acne: psychological aspects and effects of acne vulgaris. J Cosmet Dermatol. 2021;20:1080-1083. doi:10.1111/jocd.13765
- Sood S, Jafferany M, Vinaya Kumar S. Depression, psychiatric comorbidities, and psychosocial implications associated with acne vulgaris. J Cosmet Dermatol. 2020;19:3177-3182. doi:10.1111/jocd.13753
- Brahe C, Peters K. Fighting acne for the fighting forces. Cutis. 2020;106:18-20, 22. doi:10.12788/cutis.0057
- Cotterill JA, Cunliffe WJ. Suicide in dermatological patients. Br J Dermatol. 1997;137:246-250.
- Xu S, Zhu Y, Hu H, et al. The analysis of acne increasing suicide risk. Medicine (Baltimore). 2021;100:E26035. doi:10.1097/MD.0000000000026035
- Chen M, Deng Z, Huang Y, et al. Prevalence and risk factors of anxiety and depression in rosacea patients: a cross-sectional study in China [published online June 16, 2021]. Front Psychiatry. doi:10.3389/fpsyt.2021.659171
- Incel Uysal P, Akdogan N, Hayran Y, et al. Rosacea associated with increased risk of generalized anxiety disorder: a case-control study of prevalence and risk of anxiety in patients with rosacea. An Bras Dermatol. 2019;94:704-709. doi:10.1016/j.abd.2019.03.002
- Alinia H, Cardwell LA, Tuchayi SM, et al. Screening for depression in rosacea patients. Cutis. 2018;102:36-38.
- Wright S, Strunk A, Garg A. Prevalence of depression among children, adolescents, and adults with hidradenitis suppurativa [published online June 16, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.06.843
- Misitzis A, Goldust M, Jafferany M, et al. Psychiatric comorbidities in patients with hidradenitis suppurativa. Dermatol Ther. 2020;33:E13541. doi:10.1111/dth.13541
- Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017.
- Ghahramanlou-Holloway M, LaCroix JM, Koss K, et al. Outpatient mental health treatment utilization and military career impact in the United States Marine Corps. Int J Environ Res Public Health. 2018;15:828. doi:10.3390/ijerph15040828
- Ottignon DA. Marine Corps Suicide Prevention System (MCSPS). Marine Corps Order 1720.2A. 2021. Headquarters United States Marine Corps. Published August 2, 2021. Accessed May 25, 2022. https://www.marines.mil/Portals/1/Publications/MCO%201720.2A.pdf?ver=QPxZ_qMS-X-d037B65N9Tg%3d%3d
- Reger MA, Smolenski DJ, Carter SP. Suicide prevention in the US Army: a mission for more than mental health clinicians. JAMA Psychiatry. 2018;75:991-992. doi:10.1001/jamapsychiatry.2018.2042
- Pruitt LD, Smolenski DJ, Bush NE, et al. Department of Defense Suicide Event Report Calendar Year 2015 Annual Report. National Center for Telehealth & Technology (T2); 2016. Accessed May 20, 2022. https://health.mil/Military-Health-Topics/Centers-of-Excellence/Psychological-Health-Center-of-Excellence/Department-of-Defense-Suicide-Event-Report
- Ursano RJ, Kessler RC, Naifeh JA, et al. Risk factors associated with attempted suicide among US Army soldiers without a history of mental health diagnosis. JAMA Psychiatry. 2018;75:1022-1032. doi:10.1001/jamapsychiatry.2018.2069
- Gupta MA, Gupta AK. Cutaneous body image dissatisfaction and suicidal ideation: mediation by interpersonal sensitivity. J Psychosom Res. 2013;75:55-59. doi:10.1016/j.jpsychores.2013.01.015
- Gupta MA, Gupta AK. Evaluation of cutaneous body image dissatisfaction in the dermatology patient. Clin Dermatol. 2013;31:72-79. doi:10.1016/j.clindermatol.2011.11.010
- Hinkley SB, Holub SC, Menter A. The validity of cutaneous body image as a construct and as a mediator of the relationship between cutaneous disease and mental health. Dermatol Ther (Heidelb). 2020;10:203-211. doi:10.1007/s13555-020-00351-5
- Stamu-O’Brien C, Jafferany M, Carniciu S, et al. Psychodermatology of acne: psychological aspects and effects of acne vulgaris. J Cosmet Dermatol. 2021;20:1080-1083. doi:10.1111/jocd.13765
- Sood S, Jafferany M, Vinaya Kumar S. Depression, psychiatric comorbidities, and psychosocial implications associated with acne vulgaris. J Cosmet Dermatol. 2020;19:3177-3182. doi:10.1111/jocd.13753
- Brahe C, Peters K. Fighting acne for the fighting forces. Cutis. 2020;106:18-20, 22. doi:10.12788/cutis.0057
- Cotterill JA, Cunliffe WJ. Suicide in dermatological patients. Br J Dermatol. 1997;137:246-250.
- Xu S, Zhu Y, Hu H, et al. The analysis of acne increasing suicide risk. Medicine (Baltimore). 2021;100:E26035. doi:10.1097/MD.0000000000026035
- Chen M, Deng Z, Huang Y, et al. Prevalence and risk factors of anxiety and depression in rosacea patients: a cross-sectional study in China [published online June 16, 2021]. Front Psychiatry. doi:10.3389/fpsyt.2021.659171
- Incel Uysal P, Akdogan N, Hayran Y, et al. Rosacea associated with increased risk of generalized anxiety disorder: a case-control study of prevalence and risk of anxiety in patients with rosacea. An Bras Dermatol. 2019;94:704-709. doi:10.1016/j.abd.2019.03.002
- Alinia H, Cardwell LA, Tuchayi SM, et al. Screening for depression in rosacea patients. Cutis. 2018;102:36-38.
- Wright S, Strunk A, Garg A. Prevalence of depression among children, adolescents, and adults with hidradenitis suppurativa [published online June 16, 2021]. J Am Acad Dermatol. doi:10.1016/j.jaad.2021.06.843
- Misitzis A, Goldust M, Jafferany M, et al. Psychiatric comorbidities in patients with hidradenitis suppurativa. Dermatol Ther. 2020;33:E13541. doi:10.1111/dth.13541
- Bolognia J, Schaffer J, Cerroni L, eds. Dermatology. 4th ed. Elsevier; 2017.
Practice Points
- The term readiness refers to the ability to recruit, train, deploy, and sustain military forces that are ready to “fight tonight” and succeed in combat.
- Maintaining readiness requires a holistic approach, as it is directly affected by physical and mental health outcomes.
- Cutaneous body image (CBI) refers to an individual’s mental perception of the condition of their hair, nails, and skin. Positive CBI is related to increased quality of life, while negative CBI, which often is associated with dermatologic disease, is associated with poorer health outcomes and even self-injury.
- Treatment of dermatologic disease in the context of active-duty military members can positively influence CBI, which may in turn increase service members’ quality of life and overall military readiness.
Biologics, Women, and Pregnancy: What’s Known?
As the and the child’s development.
“I get asked a lot about fertility,” Vivian Shi, MD, associate professor of dermatology at the University of Arkansas, Little Rock, said at MedscapeLive’s Women’s and Pediatric Dermatology Seminar. Patients want to know, she said, if they go on a specific drug, whether it will affect their chances of conceiving and what else they need to know about safety.
She told the audience what she tells her patients: The answers are not complete but are evolving at a steady pace.
“Putting this talk together was kind of like a scavenger hunt,” said Dr. Shi, who gathered data from pregnancy exposure registries, published research, the Food and Drug Administration, and other sources on biologics. As more studies emerge each year, she said, recommendations will become stronger for considering treatment by certain biological drugs, taking into account effects on fertility, pregnancy, lactation, and the infant.
Among the biologics commonly used in dermatology are:
- Tumor necrosis factor (TNF) inhibitors (etanercept, adalimumab, infliximab, certolizumab).
- Interleukin (IL)–12 and -23 antagonist (ustekinumab).
- IL-17 antagonists (ixekizumab, secukinumab, brodalumab).
- IL-23 antagonists (risankizumab, tildrakizumab, guselkumab).
- IL-4, -13 antagonist (dupilumab) and IL-13 antagonist (tralokinumab).
- CD20-directed cytolytic antibody (rituximab).
To help with decision-making, Dr. Shi discussed the relatively new FDA labeling regulations as well as pregnancy exposure registries, research studies, and recommendations.
FDA pregnancy risk summaries
Under the previous system of classification of drugs in pregnancy, the FDA rated drugs as A, B, C, D, X. These categories ranged from showing no risks to the fetus to clear risk, but were oversimplistic and confusing, Dr. Shi said. Category C was especially confusing, as a drug with no animal or human data was put in the same category as a drug with adverse fetal effects on animals, she noted.
However, effective June 30, 2015, the FDA replaced pregnancy categories with risk summaries by medication. As of June, 2020, all prescription drugs were to remove pregnancy letter labeling. The risk summaries note human data when they are available and also note when no data are available. This information, Dr. Shi said, originates from many sources, including studies published in the medical literature, postmarketing studies conducted by companies, and pregnancy exposure registries, conducted by some companies and others. The FDA does not endorse any specific registries, but does post a list of such registries. Another helpful resource, she said, is Mother to Baby, a service of the nonprofit Organization of Teratology Information Specialists (OTIS).
Known, not known
Citing published literature, Dr. Shi said that TNF inhibitors have the most robust safety data from preconception to after birth. Less is known, she said, about the reproductive safety effects of other biologics used for dermatologic conditions, as they are newer than the anti-TNF medicines.
She reviewed a variety of research studies evaluating the safety of biologics during pregnancy and beyond. Highlights include results from a large registry, the Psoriasis Longitudinal Assessment and Registry (PSOLAR), of 298 pregnancies in about 220 women from 2007 to 2019, looking at 13 different biologics. The overall and live-birth outcomes in the women on biologics for psoriasis were similar to those for the general population and the rate of congenital anomalies was 0.8%, researchers reported in 2021, lower than the generally cited annual figure of U.S. births.
Studies evaluating biologics for nondermatologic conditions suggest safety. A prospective cohort study of women who took adalimumab in pregnancy (for rheumatoid arthritis or Crohn’s disease) found no increased risk for birth defects. In another study looking at women who were breastfeeding, researchers found no increased risk of infections or delay in developmental milestones in the children of women taking biologics for inflammatory bowel disease, compared with those not on the medications.
A report using data from the World Health Organization concludes that dupilumab appears to be safe during pregnancy, based on an evaluation of 36 pregnancy-related reports among more than 37,000 unique adverse event reports related to dupilumab in a global database.
Recommendations about biologic use from different organizations don’t always mesh, Dr. Shi said, noting that European guidelines tend to be stricter, as some reviews show.
If a mother is exposed to any biologic therapy other than certolizumab during the third trimester, after 27 weeks, Dr. Shi said, “you want to consider avoiding a live vaccine for the first 6 months of the baby’s life.” It turns out, she said, the only recommended live vaccine during that period is the rotavirus vaccine, and she suggests doctors recommend postponing that one until the babies are older if women have been on biologics other than certolizumab.
Her other take-home messages: TNF inhibitors have the most robust safety data from before conception through lactation. Under current guidelines, certolizumab is viewed as the safest to use throughout pregnancy. Dr. Shi’s message to her colleagues fielding the same questions she gets from patients: “There is more data coming out every year. Ultimately, we will have better information to inform our patients.”
At the conference, Lawrence F. Eichenfield, MD, a course director and professor of dermatology and pediatrics at the University of California, San Diego and Rady Children’s Hospital San Diego, encouraged Dr. Shi to write up her presentation as a resource for other dermatologists – which she said is in progress.
Medscape Live and this news organization are owned by the same parent company. Dr. Shi disclosed consulting and investigative and research funding from several pharmaceutical firms, but not directly related to the content of her presentation.
As the and the child’s development.
“I get asked a lot about fertility,” Vivian Shi, MD, associate professor of dermatology at the University of Arkansas, Little Rock, said at MedscapeLive’s Women’s and Pediatric Dermatology Seminar. Patients want to know, she said, if they go on a specific drug, whether it will affect their chances of conceiving and what else they need to know about safety.
She told the audience what she tells her patients: The answers are not complete but are evolving at a steady pace.
“Putting this talk together was kind of like a scavenger hunt,” said Dr. Shi, who gathered data from pregnancy exposure registries, published research, the Food and Drug Administration, and other sources on biologics. As more studies emerge each year, she said, recommendations will become stronger for considering treatment by certain biological drugs, taking into account effects on fertility, pregnancy, lactation, and the infant.
Among the biologics commonly used in dermatology are:
- Tumor necrosis factor (TNF) inhibitors (etanercept, adalimumab, infliximab, certolizumab).
- Interleukin (IL)–12 and -23 antagonist (ustekinumab).
- IL-17 antagonists (ixekizumab, secukinumab, brodalumab).
- IL-23 antagonists (risankizumab, tildrakizumab, guselkumab).
- IL-4, -13 antagonist (dupilumab) and IL-13 antagonist (tralokinumab).
- CD20-directed cytolytic antibody (rituximab).
To help with decision-making, Dr. Shi discussed the relatively new FDA labeling regulations as well as pregnancy exposure registries, research studies, and recommendations.
FDA pregnancy risk summaries
Under the previous system of classification of drugs in pregnancy, the FDA rated drugs as A, B, C, D, X. These categories ranged from showing no risks to the fetus to clear risk, but were oversimplistic and confusing, Dr. Shi said. Category C was especially confusing, as a drug with no animal or human data was put in the same category as a drug with adverse fetal effects on animals, she noted.
However, effective June 30, 2015, the FDA replaced pregnancy categories with risk summaries by medication. As of June, 2020, all prescription drugs were to remove pregnancy letter labeling. The risk summaries note human data when they are available and also note when no data are available. This information, Dr. Shi said, originates from many sources, including studies published in the medical literature, postmarketing studies conducted by companies, and pregnancy exposure registries, conducted by some companies and others. The FDA does not endorse any specific registries, but does post a list of such registries. Another helpful resource, she said, is Mother to Baby, a service of the nonprofit Organization of Teratology Information Specialists (OTIS).
Known, not known
Citing published literature, Dr. Shi said that TNF inhibitors have the most robust safety data from preconception to after birth. Less is known, she said, about the reproductive safety effects of other biologics used for dermatologic conditions, as they are newer than the anti-TNF medicines.
She reviewed a variety of research studies evaluating the safety of biologics during pregnancy and beyond. Highlights include results from a large registry, the Psoriasis Longitudinal Assessment and Registry (PSOLAR), of 298 pregnancies in about 220 women from 2007 to 2019, looking at 13 different biologics. The overall and live-birth outcomes in the women on biologics for psoriasis were similar to those for the general population and the rate of congenital anomalies was 0.8%, researchers reported in 2021, lower than the generally cited annual figure of U.S. births.
Studies evaluating biologics for nondermatologic conditions suggest safety. A prospective cohort study of women who took adalimumab in pregnancy (for rheumatoid arthritis or Crohn’s disease) found no increased risk for birth defects. In another study looking at women who were breastfeeding, researchers found no increased risk of infections or delay in developmental milestones in the children of women taking biologics for inflammatory bowel disease, compared with those not on the medications.
A report using data from the World Health Organization concludes that dupilumab appears to be safe during pregnancy, based on an evaluation of 36 pregnancy-related reports among more than 37,000 unique adverse event reports related to dupilumab in a global database.
Recommendations about biologic use from different organizations don’t always mesh, Dr. Shi said, noting that European guidelines tend to be stricter, as some reviews show.
If a mother is exposed to any biologic therapy other than certolizumab during the third trimester, after 27 weeks, Dr. Shi said, “you want to consider avoiding a live vaccine for the first 6 months of the baby’s life.” It turns out, she said, the only recommended live vaccine during that period is the rotavirus vaccine, and she suggests doctors recommend postponing that one until the babies are older if women have been on biologics other than certolizumab.
Her other take-home messages: TNF inhibitors have the most robust safety data from before conception through lactation. Under current guidelines, certolizumab is viewed as the safest to use throughout pregnancy. Dr. Shi’s message to her colleagues fielding the same questions she gets from patients: “There is more data coming out every year. Ultimately, we will have better information to inform our patients.”
At the conference, Lawrence F. Eichenfield, MD, a course director and professor of dermatology and pediatrics at the University of California, San Diego and Rady Children’s Hospital San Diego, encouraged Dr. Shi to write up her presentation as a resource for other dermatologists – which she said is in progress.
Medscape Live and this news organization are owned by the same parent company. Dr. Shi disclosed consulting and investigative and research funding from several pharmaceutical firms, but not directly related to the content of her presentation.
As the and the child’s development.
“I get asked a lot about fertility,” Vivian Shi, MD, associate professor of dermatology at the University of Arkansas, Little Rock, said at MedscapeLive’s Women’s and Pediatric Dermatology Seminar. Patients want to know, she said, if they go on a specific drug, whether it will affect their chances of conceiving and what else they need to know about safety.
She told the audience what she tells her patients: The answers are not complete but are evolving at a steady pace.
“Putting this talk together was kind of like a scavenger hunt,” said Dr. Shi, who gathered data from pregnancy exposure registries, published research, the Food and Drug Administration, and other sources on biologics. As more studies emerge each year, she said, recommendations will become stronger for considering treatment by certain biological drugs, taking into account effects on fertility, pregnancy, lactation, and the infant.
Among the biologics commonly used in dermatology are:
- Tumor necrosis factor (TNF) inhibitors (etanercept, adalimumab, infliximab, certolizumab).
- Interleukin (IL)–12 and -23 antagonist (ustekinumab).
- IL-17 antagonists (ixekizumab, secukinumab, brodalumab).
- IL-23 antagonists (risankizumab, tildrakizumab, guselkumab).
- IL-4, -13 antagonist (dupilumab) and IL-13 antagonist (tralokinumab).
- CD20-directed cytolytic antibody (rituximab).
To help with decision-making, Dr. Shi discussed the relatively new FDA labeling regulations as well as pregnancy exposure registries, research studies, and recommendations.
FDA pregnancy risk summaries
Under the previous system of classification of drugs in pregnancy, the FDA rated drugs as A, B, C, D, X. These categories ranged from showing no risks to the fetus to clear risk, but were oversimplistic and confusing, Dr. Shi said. Category C was especially confusing, as a drug with no animal or human data was put in the same category as a drug with adverse fetal effects on animals, she noted.
However, effective June 30, 2015, the FDA replaced pregnancy categories with risk summaries by medication. As of June, 2020, all prescription drugs were to remove pregnancy letter labeling. The risk summaries note human data when they are available and also note when no data are available. This information, Dr. Shi said, originates from many sources, including studies published in the medical literature, postmarketing studies conducted by companies, and pregnancy exposure registries, conducted by some companies and others. The FDA does not endorse any specific registries, but does post a list of such registries. Another helpful resource, she said, is Mother to Baby, a service of the nonprofit Organization of Teratology Information Specialists (OTIS).
Known, not known
Citing published literature, Dr. Shi said that TNF inhibitors have the most robust safety data from preconception to after birth. Less is known, she said, about the reproductive safety effects of other biologics used for dermatologic conditions, as they are newer than the anti-TNF medicines.
She reviewed a variety of research studies evaluating the safety of biologics during pregnancy and beyond. Highlights include results from a large registry, the Psoriasis Longitudinal Assessment and Registry (PSOLAR), of 298 pregnancies in about 220 women from 2007 to 2019, looking at 13 different biologics. The overall and live-birth outcomes in the women on biologics for psoriasis were similar to those for the general population and the rate of congenital anomalies was 0.8%, researchers reported in 2021, lower than the generally cited annual figure of U.S. births.
Studies evaluating biologics for nondermatologic conditions suggest safety. A prospective cohort study of women who took adalimumab in pregnancy (for rheumatoid arthritis or Crohn’s disease) found no increased risk for birth defects. In another study looking at women who were breastfeeding, researchers found no increased risk of infections or delay in developmental milestones in the children of women taking biologics for inflammatory bowel disease, compared with those not on the medications.
A report using data from the World Health Organization concludes that dupilumab appears to be safe during pregnancy, based on an evaluation of 36 pregnancy-related reports among more than 37,000 unique adverse event reports related to dupilumab in a global database.
Recommendations about biologic use from different organizations don’t always mesh, Dr. Shi said, noting that European guidelines tend to be stricter, as some reviews show.
If a mother is exposed to any biologic therapy other than certolizumab during the third trimester, after 27 weeks, Dr. Shi said, “you want to consider avoiding a live vaccine for the first 6 months of the baby’s life.” It turns out, she said, the only recommended live vaccine during that period is the rotavirus vaccine, and she suggests doctors recommend postponing that one until the babies are older if women have been on biologics other than certolizumab.
Her other take-home messages: TNF inhibitors have the most robust safety data from before conception through lactation. Under current guidelines, certolizumab is viewed as the safest to use throughout pregnancy. Dr. Shi’s message to her colleagues fielding the same questions she gets from patients: “There is more data coming out every year. Ultimately, we will have better information to inform our patients.”
At the conference, Lawrence F. Eichenfield, MD, a course director and professor of dermatology and pediatrics at the University of California, San Diego and Rady Children’s Hospital San Diego, encouraged Dr. Shi to write up her presentation as a resource for other dermatologists – which she said is in progress.
Medscape Live and this news organization are owned by the same parent company. Dr. Shi disclosed consulting and investigative and research funding from several pharmaceutical firms, but not directly related to the content of her presentation.
FROM MEDSCAPELIVE WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
Serum brodalumab levels linked with treatment outcomes in patients with psoriasis
In a study of patients with psoriasis who had previously failed treatment with interleukin-17 receptor A inhibitor therapy, “all patients with quantifiable levels of brodalumab after 12 weeks of therapy experienced PASI reductions” and subquantifiable brodalumab levels were associated with a lack of response after 12 weeks, they wrote in JAMA Dermatology.
Lead study author Christian Enevold, PhD, a researcher at the Institute for Inflammation Research at Copenhagen University Hospital, and colleagues monitored patients with plaque psoriasis who had not improved with previous IL-17A inhibitor therapy, to evaluate whether trough levels and antidrug antibodies were associated with clinical response in this group of patients.
The 20 consecutive adult patients were treated at two academic hospital dermatology clinics between 2018 and 2020 and ranged in age from 19 to 66 years; 13 were male. At baseline, their weight ranged from 59 to 182 kg (median, 103 kg), their body mass index (BMI) ranged from 20 to 50 (median, 32), and their Psoriasis Area and Severity Index (PASI) scores ranged from 7 to 26 (median, 13). All had failed treatment with at least one IL-17A inhibitor, and 90% had failed treatment with at least one tumor necrosis factor–alpha or IL-12/-23 inhibitor.
Patients stopped taking systemic psoriasis therapies for 4 weeks before entering the study, then received subcutaneous injections of 210 mg of the IL-17A inhibitor brodalumab (Siliq) at weeks 0, 1, 2, and every 2 weeks thereafter. Patients whose PASI scores did not improve at least 75% from baseline (PASI 75) after 12 weeks of brodalumab discontinued treatment and left the study, while those who maintained PASI 75 were monitored for up to 52 weeks.
The researchers used assays to compare decreases in PASI score with brodalumab levels as well as with antibrodalumab antibodies at 12 weeks, and determined the following:
- Participants with quantifiable brodalumab levels (≥ 0.05 mcg/mL) showed a greater drop in PASI scores (median, 93%; range, 61%-100%) than those without quantifiable brodalumab levels (median, −3; range, −49% to 94%) (P = .006).
- Four of 5 patients (80%) who did not achieve a PASI 75, compared with 3 of 14 PASI 75 responders (21%), had drug levels too low to be measured (< 0.05 mcg/mL).
- The eight patients who did not have obesity (BMI < 30) had PASI reductions of at least 77%, and seven of the eight patients (88%) had quantifiable brodalumab levels.
- Six of the 12 patients with obesity (BMI ≥ 30) had brodalumab levels too low to be measured. Of those, four had increased PASI after 12 weeks of treatment. For all patients with obesity with quantifiable brodalumab levels, PASI scores dropped by at least 61% after 12 weeks.
- Five of the 12 (42%) patients with obesity versus 7 of the 8 (88%) patients without obesity had quantifiable brodalumab levels.
- None of the seven patients (35%) with subquantifiable drug levels after 12 weeks remained PASI responders.
- No antibrodalumab antibodies were detected in any serum samples.
The authors acknowledged that there were limitations of the study, including its retrospective design and restriction to the few available participants with a history of treatment failure.
George Han, MD, PhD, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said in an interview that he found the study interesting. “The authors did an admirable job looking at many factors to try to understand response to treatment in a challenging population of patients who had failed at least one, and in many cases, numerous, biologics from different classes.”
“The most interesting finding is that patients with higher BMIs had much higher rates of low-to-undetectable drug concentration,” said Dr. Han, who was not involved in the study. “This very practical finding could help patient care immediately. While it’s impractical to start performing assays of drug concentration in clinical practice, this finding certainly would guide my conversations with my heavier-set patients who have had multiple failures on previous biologics.
“I’m looking forward to further studies that explore this issue and provide better evidence-based guidance for treating patients who have experienced multi-biologic failure,” he added.
Robert A. Dorschner, MD, assistant professor of dermatology at the UC San Diego Health System, also welcomed the study’s results.
“Current psoriasis treatment is based on trial-and-error application of various biologics targeting different pathways, with initial selection frequently based on insurance preference, not patient characteristics,” he said in an interview.
“Studies like this help clinicians make more informed decisions about whether a patient may benefit from a different dose or may require a different drug, and make those decisions earlier in therapy,” he said. “This can improve patient care and decrease costs associated with prolonged treatments with ineffective drugs.”
But Dr. Dorschner, who also was not involved in the study, cautions clinicians to not draw conclusions about dose adjustments from these results. “These findings need to be verified in a larger cohort,” he advised, “and they should drive future studies with larger cohorts and prospective designs.”
“The last couple of decades have seen an explosion in the availability of biologics targeting different cytokines, with significant benefits to patients,” Dr. Dorschner explained. “However, there is a dearth of information on how to choose the right biologic for a particular patient and how to assess the benefit of dose alteration versus changing the drug target. Medicine needs more studies like this one.”
Several authors of the study report financial relationships with LEO Pharma and other pharmaceutical companies. Most authors, including Dr. Enevold, reported no relevant financial relationships. Dr. Dorschner reported no relevant financial relationships. Dr. Han reported financial relationships with pharmaceutical companies not involved in the study. The study was funded by LEO Pharma and the Danish Biotechnology Program.
In a study of patients with psoriasis who had previously failed treatment with interleukin-17 receptor A inhibitor therapy, “all patients with quantifiable levels of brodalumab after 12 weeks of therapy experienced PASI reductions” and subquantifiable brodalumab levels were associated with a lack of response after 12 weeks, they wrote in JAMA Dermatology.
Lead study author Christian Enevold, PhD, a researcher at the Institute for Inflammation Research at Copenhagen University Hospital, and colleagues monitored patients with plaque psoriasis who had not improved with previous IL-17A inhibitor therapy, to evaluate whether trough levels and antidrug antibodies were associated with clinical response in this group of patients.
The 20 consecutive adult patients were treated at two academic hospital dermatology clinics between 2018 and 2020 and ranged in age from 19 to 66 years; 13 were male. At baseline, their weight ranged from 59 to 182 kg (median, 103 kg), their body mass index (BMI) ranged from 20 to 50 (median, 32), and their Psoriasis Area and Severity Index (PASI) scores ranged from 7 to 26 (median, 13). All had failed treatment with at least one IL-17A inhibitor, and 90% had failed treatment with at least one tumor necrosis factor–alpha or IL-12/-23 inhibitor.
Patients stopped taking systemic psoriasis therapies for 4 weeks before entering the study, then received subcutaneous injections of 210 mg of the IL-17A inhibitor brodalumab (Siliq) at weeks 0, 1, 2, and every 2 weeks thereafter. Patients whose PASI scores did not improve at least 75% from baseline (PASI 75) after 12 weeks of brodalumab discontinued treatment and left the study, while those who maintained PASI 75 were monitored for up to 52 weeks.
The researchers used assays to compare decreases in PASI score with brodalumab levels as well as with antibrodalumab antibodies at 12 weeks, and determined the following:
- Participants with quantifiable brodalumab levels (≥ 0.05 mcg/mL) showed a greater drop in PASI scores (median, 93%; range, 61%-100%) than those without quantifiable brodalumab levels (median, −3; range, −49% to 94%) (P = .006).
- Four of 5 patients (80%) who did not achieve a PASI 75, compared with 3 of 14 PASI 75 responders (21%), had drug levels too low to be measured (< 0.05 mcg/mL).
- The eight patients who did not have obesity (BMI < 30) had PASI reductions of at least 77%, and seven of the eight patients (88%) had quantifiable brodalumab levels.
- Six of the 12 patients with obesity (BMI ≥ 30) had brodalumab levels too low to be measured. Of those, four had increased PASI after 12 weeks of treatment. For all patients with obesity with quantifiable brodalumab levels, PASI scores dropped by at least 61% after 12 weeks.
- Five of the 12 (42%) patients with obesity versus 7 of the 8 (88%) patients without obesity had quantifiable brodalumab levels.
- None of the seven patients (35%) with subquantifiable drug levels after 12 weeks remained PASI responders.
- No antibrodalumab antibodies were detected in any serum samples.
The authors acknowledged that there were limitations of the study, including its retrospective design and restriction to the few available participants with a history of treatment failure.
George Han, MD, PhD, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said in an interview that he found the study interesting. “The authors did an admirable job looking at many factors to try to understand response to treatment in a challenging population of patients who had failed at least one, and in many cases, numerous, biologics from different classes.”
“The most interesting finding is that patients with higher BMIs had much higher rates of low-to-undetectable drug concentration,” said Dr. Han, who was not involved in the study. “This very practical finding could help patient care immediately. While it’s impractical to start performing assays of drug concentration in clinical practice, this finding certainly would guide my conversations with my heavier-set patients who have had multiple failures on previous biologics.
“I’m looking forward to further studies that explore this issue and provide better evidence-based guidance for treating patients who have experienced multi-biologic failure,” he added.
Robert A. Dorschner, MD, assistant professor of dermatology at the UC San Diego Health System, also welcomed the study’s results.
“Current psoriasis treatment is based on trial-and-error application of various biologics targeting different pathways, with initial selection frequently based on insurance preference, not patient characteristics,” he said in an interview.
“Studies like this help clinicians make more informed decisions about whether a patient may benefit from a different dose or may require a different drug, and make those decisions earlier in therapy,” he said. “This can improve patient care and decrease costs associated with prolonged treatments with ineffective drugs.”
But Dr. Dorschner, who also was not involved in the study, cautions clinicians to not draw conclusions about dose adjustments from these results. “These findings need to be verified in a larger cohort,” he advised, “and they should drive future studies with larger cohorts and prospective designs.”
“The last couple of decades have seen an explosion in the availability of biologics targeting different cytokines, with significant benefits to patients,” Dr. Dorschner explained. “However, there is a dearth of information on how to choose the right biologic for a particular patient and how to assess the benefit of dose alteration versus changing the drug target. Medicine needs more studies like this one.”
Several authors of the study report financial relationships with LEO Pharma and other pharmaceutical companies. Most authors, including Dr. Enevold, reported no relevant financial relationships. Dr. Dorschner reported no relevant financial relationships. Dr. Han reported financial relationships with pharmaceutical companies not involved in the study. The study was funded by LEO Pharma and the Danish Biotechnology Program.
In a study of patients with psoriasis who had previously failed treatment with interleukin-17 receptor A inhibitor therapy, “all patients with quantifiable levels of brodalumab after 12 weeks of therapy experienced PASI reductions” and subquantifiable brodalumab levels were associated with a lack of response after 12 weeks, they wrote in JAMA Dermatology.
Lead study author Christian Enevold, PhD, a researcher at the Institute for Inflammation Research at Copenhagen University Hospital, and colleagues monitored patients with plaque psoriasis who had not improved with previous IL-17A inhibitor therapy, to evaluate whether trough levels and antidrug antibodies were associated with clinical response in this group of patients.
The 20 consecutive adult patients were treated at two academic hospital dermatology clinics between 2018 and 2020 and ranged in age from 19 to 66 years; 13 were male. At baseline, their weight ranged from 59 to 182 kg (median, 103 kg), their body mass index (BMI) ranged from 20 to 50 (median, 32), and their Psoriasis Area and Severity Index (PASI) scores ranged from 7 to 26 (median, 13). All had failed treatment with at least one IL-17A inhibitor, and 90% had failed treatment with at least one tumor necrosis factor–alpha or IL-12/-23 inhibitor.
Patients stopped taking systemic psoriasis therapies for 4 weeks before entering the study, then received subcutaneous injections of 210 mg of the IL-17A inhibitor brodalumab (Siliq) at weeks 0, 1, 2, and every 2 weeks thereafter. Patients whose PASI scores did not improve at least 75% from baseline (PASI 75) after 12 weeks of brodalumab discontinued treatment and left the study, while those who maintained PASI 75 were monitored for up to 52 weeks.
The researchers used assays to compare decreases in PASI score with brodalumab levels as well as with antibrodalumab antibodies at 12 weeks, and determined the following:
- Participants with quantifiable brodalumab levels (≥ 0.05 mcg/mL) showed a greater drop in PASI scores (median, 93%; range, 61%-100%) than those without quantifiable brodalumab levels (median, −3; range, −49% to 94%) (P = .006).
- Four of 5 patients (80%) who did not achieve a PASI 75, compared with 3 of 14 PASI 75 responders (21%), had drug levels too low to be measured (< 0.05 mcg/mL).
- The eight patients who did not have obesity (BMI < 30) had PASI reductions of at least 77%, and seven of the eight patients (88%) had quantifiable brodalumab levels.
- Six of the 12 patients with obesity (BMI ≥ 30) had brodalumab levels too low to be measured. Of those, four had increased PASI after 12 weeks of treatment. For all patients with obesity with quantifiable brodalumab levels, PASI scores dropped by at least 61% after 12 weeks.
- Five of the 12 (42%) patients with obesity versus 7 of the 8 (88%) patients without obesity had quantifiable brodalumab levels.
- None of the seven patients (35%) with subquantifiable drug levels after 12 weeks remained PASI responders.
- No antibrodalumab antibodies were detected in any serum samples.
The authors acknowledged that there were limitations of the study, including its retrospective design and restriction to the few available participants with a history of treatment failure.
George Han, MD, PhD, associate professor of dermatology at Hofstra University, Hempstead, N.Y., said in an interview that he found the study interesting. “The authors did an admirable job looking at many factors to try to understand response to treatment in a challenging population of patients who had failed at least one, and in many cases, numerous, biologics from different classes.”
“The most interesting finding is that patients with higher BMIs had much higher rates of low-to-undetectable drug concentration,” said Dr. Han, who was not involved in the study. “This very practical finding could help patient care immediately. While it’s impractical to start performing assays of drug concentration in clinical practice, this finding certainly would guide my conversations with my heavier-set patients who have had multiple failures on previous biologics.
“I’m looking forward to further studies that explore this issue and provide better evidence-based guidance for treating patients who have experienced multi-biologic failure,” he added.
Robert A. Dorschner, MD, assistant professor of dermatology at the UC San Diego Health System, also welcomed the study’s results.
“Current psoriasis treatment is based on trial-and-error application of various biologics targeting different pathways, with initial selection frequently based on insurance preference, not patient characteristics,” he said in an interview.
“Studies like this help clinicians make more informed decisions about whether a patient may benefit from a different dose or may require a different drug, and make those decisions earlier in therapy,” he said. “This can improve patient care and decrease costs associated with prolonged treatments with ineffective drugs.”
But Dr. Dorschner, who also was not involved in the study, cautions clinicians to not draw conclusions about dose adjustments from these results. “These findings need to be verified in a larger cohort,” he advised, “and they should drive future studies with larger cohorts and prospective designs.”
“The last couple of decades have seen an explosion in the availability of biologics targeting different cytokines, with significant benefits to patients,” Dr. Dorschner explained. “However, there is a dearth of information on how to choose the right biologic for a particular patient and how to assess the benefit of dose alteration versus changing the drug target. Medicine needs more studies like this one.”
Several authors of the study report financial relationships with LEO Pharma and other pharmaceutical companies. Most authors, including Dr. Enevold, reported no relevant financial relationships. Dr. Dorschner reported no relevant financial relationships. Dr. Han reported financial relationships with pharmaceutical companies not involved in the study. The study was funded by LEO Pharma and the Danish Biotechnology Program.
FROM JAMA DERMATOLOGY
FDA approves topical tapinarof for plaque psoriasis
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
Severe infections often accompany severe psoriasis
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
FROM BRITISH JOURNAL OF DERMATOLOGY
Reduced-frequency methotrexate monitoring causes no harm
Reducing the frequency of routine blood monitoring for methotrexate in patients with rheumatoid arthritis during the COVID-19 pandemic was associated with no adverse outcomes for patients, British researchers have found.
Similar laboratory results were recorded in patients who were switched from testing once per month to once every 3 or 5 months, Natasha Wood, a general practice trainee at North Devon District Hospital in Barnstaple, England, reported at the annual meeting of the British Society for Rheumatology.
“Less frequent monitoring did not result in patient harm,” she said.
“There’s an increasing evidence base; we wonder whether now’s the time to reconsider our DMARD-monitoring strategy,” Ms. Wood said.
Changes in monitoring because of pandemic
Methotrexate monitoring is important to minimize the risk of harm to patients, and it is recommended that standard laboratory tests, such as a complete blood count, creatinine, and liver enzymes are measured regularly. Indeed, both the BSR and the American College of Rheumatology have specific recommendations on the monitoring of methotrexate and other conventional synthetic disease-modifying antirheumatic drugs (csDMARDS).
“The BSR used to advise for monthly blood tests in patients taking methotrexate,” Ms. Wood said, but the BSR moved to recommend testing patients on a stable dose every 3 months in 2017.
“Things of course changed again rapidly with COVID, with the BSR quickly updating their guidelines advising for less frequent monitoring in this patient group,” Ms. Wood said.
As a result, the North Devon Clinical Commissioning Group, which covers the hospital where Ms. Wood works, agreed to allow testing every 6 months for patients on a stable methotrexate dose. “This was across specialties, so not just rheumatology, but dermatology and gastroenterology as well,” she said.
“This provided us with a really exciting and unique opportunity to look at this patient group and see what happened,” Ms. Wood explained.
Effect of less frequent monitoring
At the meeting, Ms. Wood presented the results of an audit of 854 patients found via a search of hospital pathology records who were stable on methotrexate monotherapy for at least 12 months.
Two subanalyses were performed: One looked at patients who had changed from blood testing once every month to once every 3 months (n = 229) and the other looking at a group of 120 patients who had gone from testing once every 3 months to approximately every 5 months.
The mean age of patients was 67 for monthly testing, 69 for testing every 3 months, and 66 for testing about every 5 months, with around two-thirds of patients being of female sex.
A comparison of the number of blood tests performed to the end of April 2020 with the number performed to the end of April 2021 showed that there had mainly been a shift from testing once per month to once every 3 months, with some patients being tested in line with the revised BSR guidelines at around 5 months.
“Interestingly, a third of this group had no changed monitoring frequency despite the change in guidelines,” Ms. Wood said.
“Prepandemic, most patients [were] having monthly bloods despite BSR advice from 2017, and despite the pandemic with the updated shared care guidelines,” patients were still having blood drawn every 3 months, Ms. Wood noted. This perhaps needs further investigation and consideration to understand why recommended changes to the frequency of testing are not being adhered to.
The overall distribution of laboratory findings was similar among those who went from testing once per month to once every 3 months and from every 3 months to every 5 months. This included the distribution of neutrophils, whole blood counts, and alanine aminotransferase. There were some changes for platelets, mean cell volume, and the estimated glomerular filtration rate, but these were not clinically significant.
“Abnormal blood results aren’t common in stable methotrexate monotherapy patients,” Ms. Wood reported. “Where abnormalities did occur, it was in the context of patients being concurrently unwell and symptomatic.”
Time for patient-initiated testing?
There are several advantages of less frequent methotrexate monitoring, Ms. Wood said. One is the practicalities of getting to and from appointments, particularly in remote locations, such as where she works.
In addition to reducing workloads and pressure on already busy hospitals and primary care, this could have a huge environmental impact, she suggested.
Moreover, “moderate-quality evidence” supports the current monitoring frequency recommendation.
“We know that our numbers are small – we’re a small center – but our findings are consistent with much larger studies across the U.K.,” Ms. Wood said.
“We wonder whether there’s the possibility of moving towards annual monitoring with good safety netting and patient education for additional blood tests if they are unwell,” she said, adding that “now may be the time for patient-initiated methotrexate monitoring.”
Ms. Wood disclosed Janssen sponsorship for attending the BSR 2022 annual meeting.
Reducing the frequency of routine blood monitoring for methotrexate in patients with rheumatoid arthritis during the COVID-19 pandemic was associated with no adverse outcomes for patients, British researchers have found.
Similar laboratory results were recorded in patients who were switched from testing once per month to once every 3 or 5 months, Natasha Wood, a general practice trainee at North Devon District Hospital in Barnstaple, England, reported at the annual meeting of the British Society for Rheumatology.
“Less frequent monitoring did not result in patient harm,” she said.
“There’s an increasing evidence base; we wonder whether now’s the time to reconsider our DMARD-monitoring strategy,” Ms. Wood said.
Changes in monitoring because of pandemic
Methotrexate monitoring is important to minimize the risk of harm to patients, and it is recommended that standard laboratory tests, such as a complete blood count, creatinine, and liver enzymes are measured regularly. Indeed, both the BSR and the American College of Rheumatology have specific recommendations on the monitoring of methotrexate and other conventional synthetic disease-modifying antirheumatic drugs (csDMARDS).
“The BSR used to advise for monthly blood tests in patients taking methotrexate,” Ms. Wood said, but the BSR moved to recommend testing patients on a stable dose every 3 months in 2017.
“Things of course changed again rapidly with COVID, with the BSR quickly updating their guidelines advising for less frequent monitoring in this patient group,” Ms. Wood said.
As a result, the North Devon Clinical Commissioning Group, which covers the hospital where Ms. Wood works, agreed to allow testing every 6 months for patients on a stable methotrexate dose. “This was across specialties, so not just rheumatology, but dermatology and gastroenterology as well,” she said.
“This provided us with a really exciting and unique opportunity to look at this patient group and see what happened,” Ms. Wood explained.
Effect of less frequent monitoring
At the meeting, Ms. Wood presented the results of an audit of 854 patients found via a search of hospital pathology records who were stable on methotrexate monotherapy for at least 12 months.
Two subanalyses were performed: One looked at patients who had changed from blood testing once every month to once every 3 months (n = 229) and the other looking at a group of 120 patients who had gone from testing once every 3 months to approximately every 5 months.
The mean age of patients was 67 for monthly testing, 69 for testing every 3 months, and 66 for testing about every 5 months, with around two-thirds of patients being of female sex.
A comparison of the number of blood tests performed to the end of April 2020 with the number performed to the end of April 2021 showed that there had mainly been a shift from testing once per month to once every 3 months, with some patients being tested in line with the revised BSR guidelines at around 5 months.
“Interestingly, a third of this group had no changed monitoring frequency despite the change in guidelines,” Ms. Wood said.
“Prepandemic, most patients [were] having monthly bloods despite BSR advice from 2017, and despite the pandemic with the updated shared care guidelines,” patients were still having blood drawn every 3 months, Ms. Wood noted. This perhaps needs further investigation and consideration to understand why recommended changes to the frequency of testing are not being adhered to.
The overall distribution of laboratory findings was similar among those who went from testing once per month to once every 3 months and from every 3 months to every 5 months. This included the distribution of neutrophils, whole blood counts, and alanine aminotransferase. There were some changes for platelets, mean cell volume, and the estimated glomerular filtration rate, but these were not clinically significant.
“Abnormal blood results aren’t common in stable methotrexate monotherapy patients,” Ms. Wood reported. “Where abnormalities did occur, it was in the context of patients being concurrently unwell and symptomatic.”
Time for patient-initiated testing?
There are several advantages of less frequent methotrexate monitoring, Ms. Wood said. One is the practicalities of getting to and from appointments, particularly in remote locations, such as where she works.
In addition to reducing workloads and pressure on already busy hospitals and primary care, this could have a huge environmental impact, she suggested.
Moreover, “moderate-quality evidence” supports the current monitoring frequency recommendation.
“We know that our numbers are small – we’re a small center – but our findings are consistent with much larger studies across the U.K.,” Ms. Wood said.
“We wonder whether there’s the possibility of moving towards annual monitoring with good safety netting and patient education for additional blood tests if they are unwell,” she said, adding that “now may be the time for patient-initiated methotrexate monitoring.”
Ms. Wood disclosed Janssen sponsorship for attending the BSR 2022 annual meeting.
Reducing the frequency of routine blood monitoring for methotrexate in patients with rheumatoid arthritis during the COVID-19 pandemic was associated with no adverse outcomes for patients, British researchers have found.
Similar laboratory results were recorded in patients who were switched from testing once per month to once every 3 or 5 months, Natasha Wood, a general practice trainee at North Devon District Hospital in Barnstaple, England, reported at the annual meeting of the British Society for Rheumatology.
“Less frequent monitoring did not result in patient harm,” she said.
“There’s an increasing evidence base; we wonder whether now’s the time to reconsider our DMARD-monitoring strategy,” Ms. Wood said.
Changes in monitoring because of pandemic
Methotrexate monitoring is important to minimize the risk of harm to patients, and it is recommended that standard laboratory tests, such as a complete blood count, creatinine, and liver enzymes are measured regularly. Indeed, both the BSR and the American College of Rheumatology have specific recommendations on the monitoring of methotrexate and other conventional synthetic disease-modifying antirheumatic drugs (csDMARDS).
“The BSR used to advise for monthly blood tests in patients taking methotrexate,” Ms. Wood said, but the BSR moved to recommend testing patients on a stable dose every 3 months in 2017.
“Things of course changed again rapidly with COVID, with the BSR quickly updating their guidelines advising for less frequent monitoring in this patient group,” Ms. Wood said.
As a result, the North Devon Clinical Commissioning Group, which covers the hospital where Ms. Wood works, agreed to allow testing every 6 months for patients on a stable methotrexate dose. “This was across specialties, so not just rheumatology, but dermatology and gastroenterology as well,” she said.
“This provided us with a really exciting and unique opportunity to look at this patient group and see what happened,” Ms. Wood explained.
Effect of less frequent monitoring
At the meeting, Ms. Wood presented the results of an audit of 854 patients found via a search of hospital pathology records who were stable on methotrexate monotherapy for at least 12 months.
Two subanalyses were performed: One looked at patients who had changed from blood testing once every month to once every 3 months (n = 229) and the other looking at a group of 120 patients who had gone from testing once every 3 months to approximately every 5 months.
The mean age of patients was 67 for monthly testing, 69 for testing every 3 months, and 66 for testing about every 5 months, with around two-thirds of patients being of female sex.
A comparison of the number of blood tests performed to the end of April 2020 with the number performed to the end of April 2021 showed that there had mainly been a shift from testing once per month to once every 3 months, with some patients being tested in line with the revised BSR guidelines at around 5 months.
“Interestingly, a third of this group had no changed monitoring frequency despite the change in guidelines,” Ms. Wood said.
“Prepandemic, most patients [were] having monthly bloods despite BSR advice from 2017, and despite the pandemic with the updated shared care guidelines,” patients were still having blood drawn every 3 months, Ms. Wood noted. This perhaps needs further investigation and consideration to understand why recommended changes to the frequency of testing are not being adhered to.
The overall distribution of laboratory findings was similar among those who went from testing once per month to once every 3 months and from every 3 months to every 5 months. This included the distribution of neutrophils, whole blood counts, and alanine aminotransferase. There were some changes for platelets, mean cell volume, and the estimated glomerular filtration rate, but these were not clinically significant.
“Abnormal blood results aren’t common in stable methotrexate monotherapy patients,” Ms. Wood reported. “Where abnormalities did occur, it was in the context of patients being concurrently unwell and symptomatic.”
Time for patient-initiated testing?
There are several advantages of less frequent methotrexate monitoring, Ms. Wood said. One is the practicalities of getting to and from appointments, particularly in remote locations, such as where she works.
In addition to reducing workloads and pressure on already busy hospitals and primary care, this could have a huge environmental impact, she suggested.
Moreover, “moderate-quality evidence” supports the current monitoring frequency recommendation.
“We know that our numbers are small – we’re a small center – but our findings are consistent with much larger studies across the U.K.,” Ms. Wood said.
“We wonder whether there’s the possibility of moving towards annual monitoring with good safety netting and patient education for additional blood tests if they are unwell,” she said, adding that “now may be the time for patient-initiated methotrexate monitoring.”
Ms. Wood disclosed Janssen sponsorship for attending the BSR 2022 annual meeting.
FROM BSR 2022
Parents’ autoimmune diseases may affect children’s development
Results of a meta-analysis carried out by a French team indicate that there is a link between a father’s or mother’s autoimmune disease and their children’s risk of developing certain neurodevelopmental disorders (autism spectrum disorder [ASD] and attention-deficit/hyperactivity disorder). This meta-analysis is the first to separately explore the link between a father’s or mother’s autoimmune disease and the onset of neurodevelopmental disorders in their children.
According to its authors, these associations may result from exposure to environmental factors that contribute to autoimmune disorders, such as exposure to pollutants or cigarette smoke, and/or genetic predisposition, including genes relating to cytokines or to the HLA system.
Research is needed to determine the pathophysiologic links between these associations. This study suggests that there could be a shared mechanism between both parents, even though the maternal route seems to constitute an additional excess risk.
Why is this important?
Neurodevelopmental disorders are said to occur because of a close interrelationship between a person’s genes and environment. Immune-mediated adverse reactions may play an important role in triggering such disorders, as has been shown in associated epidemiologic studies and in animal studies. Autoimmune and autoinflammatory disorders are effectively characterized by the activation of the immune system, the circulation of autoantibodies, and the secretion of cytokines that are harmful to certain tissues.
Some relevant studies suggest a link between autoimmune disorders in the family or in the mother and the onset of neurodevelopmental disorders in their children. However, none of the studies have distinguished the influence of each of the parents so as to provide data that can be used to assess whether this association is more likely to be direct, and thus established during pregnancy, or rather genetic or environmental.
Main findings
Overall, the meta-analysis involved 14 studies that included 845,411 mothers and 601,148 fathers with an autoimmune disease, 4,984,965 control mothers and 4,992,854 control fathers. There were 182,927 children with neurodevelopmental disorders and 14,168,474 with no such diagnosis.
Globally, autoimmune diseases in mothers (adjusted odds ratio, 1.27 [1.03-1.57]; P = .02; I2 = 65%) and in fathers (AOR, 1.18 [1.07-1.30]; P = .01; I2 = 15.5%) are associated with a diagnosis of ASD in children. Similarly, they are associated with an increased risk of ADHD in children (AOR, 1.31 [1.11-1.55]; P = .001; I2 = 93% and AOR, 1.14 [1.10-1.17]; P < .0001; I2 = 0%, respectively, for mothers and fathers).
In mothers, type 1 diabetes (AOR, 1.60 [1.18-2.18]; P = .002; I2 = 0%), psoriasis (AOR, 1.45 [1.14-1.85]; P = .002; I2 = 0%), and rheumatoid arthritis (AOR, 1.38 [1.14-1.68]; P = .001; I2 = 0.8%) were associated with a risk of ASD in children. These three conditions also predisposed children to the risk of ADHD (AOR, 1.36 [1.24-1.52]; 1.41 [1.29-1.54]; and 1.32 [1.25-1.40], respectively, all P < .0001).
In fathers, type 1 diabetes considered in isolation was associated with a risk of ASD and ADHD in children (AOR, 1.42 [1.10-1.83] and 1.19 [1.08-1.31], respectively), while psoriasis (AOR, 1.18 [1.12-1.24]; P < .0001) is associated with a risk of ADHD in children.
A version of this article first appeared on Medscape.com.
Results of a meta-analysis carried out by a French team indicate that there is a link between a father’s or mother’s autoimmune disease and their children’s risk of developing certain neurodevelopmental disorders (autism spectrum disorder [ASD] and attention-deficit/hyperactivity disorder). This meta-analysis is the first to separately explore the link between a father’s or mother’s autoimmune disease and the onset of neurodevelopmental disorders in their children.
According to its authors, these associations may result from exposure to environmental factors that contribute to autoimmune disorders, such as exposure to pollutants or cigarette smoke, and/or genetic predisposition, including genes relating to cytokines or to the HLA system.
Research is needed to determine the pathophysiologic links between these associations. This study suggests that there could be a shared mechanism between both parents, even though the maternal route seems to constitute an additional excess risk.
Why is this important?
Neurodevelopmental disorders are said to occur because of a close interrelationship between a person’s genes and environment. Immune-mediated adverse reactions may play an important role in triggering such disorders, as has been shown in associated epidemiologic studies and in animal studies. Autoimmune and autoinflammatory disorders are effectively characterized by the activation of the immune system, the circulation of autoantibodies, and the secretion of cytokines that are harmful to certain tissues.
Some relevant studies suggest a link between autoimmune disorders in the family or in the mother and the onset of neurodevelopmental disorders in their children. However, none of the studies have distinguished the influence of each of the parents so as to provide data that can be used to assess whether this association is more likely to be direct, and thus established during pregnancy, or rather genetic or environmental.
Main findings
Overall, the meta-analysis involved 14 studies that included 845,411 mothers and 601,148 fathers with an autoimmune disease, 4,984,965 control mothers and 4,992,854 control fathers. There were 182,927 children with neurodevelopmental disorders and 14,168,474 with no such diagnosis.
Globally, autoimmune diseases in mothers (adjusted odds ratio, 1.27 [1.03-1.57]; P = .02; I2 = 65%) and in fathers (AOR, 1.18 [1.07-1.30]; P = .01; I2 = 15.5%) are associated with a diagnosis of ASD in children. Similarly, they are associated with an increased risk of ADHD in children (AOR, 1.31 [1.11-1.55]; P = .001; I2 = 93% and AOR, 1.14 [1.10-1.17]; P < .0001; I2 = 0%, respectively, for mothers and fathers).
In mothers, type 1 diabetes (AOR, 1.60 [1.18-2.18]; P = .002; I2 = 0%), psoriasis (AOR, 1.45 [1.14-1.85]; P = .002; I2 = 0%), and rheumatoid arthritis (AOR, 1.38 [1.14-1.68]; P = .001; I2 = 0.8%) were associated with a risk of ASD in children. These three conditions also predisposed children to the risk of ADHD (AOR, 1.36 [1.24-1.52]; 1.41 [1.29-1.54]; and 1.32 [1.25-1.40], respectively, all P < .0001).
In fathers, type 1 diabetes considered in isolation was associated with a risk of ASD and ADHD in children (AOR, 1.42 [1.10-1.83] and 1.19 [1.08-1.31], respectively), while psoriasis (AOR, 1.18 [1.12-1.24]; P < .0001) is associated with a risk of ADHD in children.
A version of this article first appeared on Medscape.com.
Results of a meta-analysis carried out by a French team indicate that there is a link between a father’s or mother’s autoimmune disease and their children’s risk of developing certain neurodevelopmental disorders (autism spectrum disorder [ASD] and attention-deficit/hyperactivity disorder). This meta-analysis is the first to separately explore the link between a father’s or mother’s autoimmune disease and the onset of neurodevelopmental disorders in their children.
According to its authors, these associations may result from exposure to environmental factors that contribute to autoimmune disorders, such as exposure to pollutants or cigarette smoke, and/or genetic predisposition, including genes relating to cytokines or to the HLA system.
Research is needed to determine the pathophysiologic links between these associations. This study suggests that there could be a shared mechanism between both parents, even though the maternal route seems to constitute an additional excess risk.
Why is this important?
Neurodevelopmental disorders are said to occur because of a close interrelationship between a person’s genes and environment. Immune-mediated adverse reactions may play an important role in triggering such disorders, as has been shown in associated epidemiologic studies and in animal studies. Autoimmune and autoinflammatory disorders are effectively characterized by the activation of the immune system, the circulation of autoantibodies, and the secretion of cytokines that are harmful to certain tissues.
Some relevant studies suggest a link between autoimmune disorders in the family or in the mother and the onset of neurodevelopmental disorders in their children. However, none of the studies have distinguished the influence of each of the parents so as to provide data that can be used to assess whether this association is more likely to be direct, and thus established during pregnancy, or rather genetic or environmental.
Main findings
Overall, the meta-analysis involved 14 studies that included 845,411 mothers and 601,148 fathers with an autoimmune disease, 4,984,965 control mothers and 4,992,854 control fathers. There were 182,927 children with neurodevelopmental disorders and 14,168,474 with no such diagnosis.
Globally, autoimmune diseases in mothers (adjusted odds ratio, 1.27 [1.03-1.57]; P = .02; I2 = 65%) and in fathers (AOR, 1.18 [1.07-1.30]; P = .01; I2 = 15.5%) are associated with a diagnosis of ASD in children. Similarly, they are associated with an increased risk of ADHD in children (AOR, 1.31 [1.11-1.55]; P = .001; I2 = 93% and AOR, 1.14 [1.10-1.17]; P < .0001; I2 = 0%, respectively, for mothers and fathers).
In mothers, type 1 diabetes (AOR, 1.60 [1.18-2.18]; P = .002; I2 = 0%), psoriasis (AOR, 1.45 [1.14-1.85]; P = .002; I2 = 0%), and rheumatoid arthritis (AOR, 1.38 [1.14-1.68]; P = .001; I2 = 0.8%) were associated with a risk of ASD in children. These three conditions also predisposed children to the risk of ADHD (AOR, 1.36 [1.24-1.52]; 1.41 [1.29-1.54]; and 1.32 [1.25-1.40], respectively, all P < .0001).
In fathers, type 1 diabetes considered in isolation was associated with a risk of ASD and ADHD in children (AOR, 1.42 [1.10-1.83] and 1.19 [1.08-1.31], respectively), while psoriasis (AOR, 1.18 [1.12-1.24]; P < .0001) is associated with a risk of ADHD in children.
A version of this article first appeared on Medscape.com.
FROM TRANSLATIONAL PSYCHIATRY
NB-UVB phototherapy plays a key role in psoriasis treatment, expert says
BOSTON – In 2012, about 50% of patients receiving phototherapy at Brigham and Women’s Hospital in Boston were being treated for psoriasis. A decade later, that proportion has dropped to 20%.
Several factors have contributed to this trend, namely, the development of biologics, the COVID-19 pandemic, “and the rise of home phototherapy options,” Elizabeth A. Buzney, MD, codirector of the phototherapy center at Brigham and Women’s department of dermatology, said at the annual meeting of the American Academy of Dermatology. In her clinical opinion, phototherapy plays an essential role in the treatment of psoriasis.
“It is medically and financially responsible to review the option of phototherapy with every psoriasis patient,” Dr. Buzney said. “Many patients are not medical or financial candidates for biologic/apremilast therapy, or just would prefer nonsystemic therapy.”
In one meta-analysis, the proportion of patients achieving Psoriasis Area and Severity Index (PASI) 75 with NB-UVB therapy was 70% after 20-40 sessions, just below the efficacy of newer biologics – but better than ustekinumab and adalimumab.
“Phototherapy is not so far out of range as you might think it is,” she said, noting that other studies of NB-UVB therapy show PASI 75 responses of 62% and PASI 90 responses of 40%.
Phototherapy can also be an appealing option because biologics aren’t the best option for all patients with psoriasis. They are expensive for the health care system and potentially for patients, require initial and potentially continued lab testing and monitoring, and require injections, “which some patients don’t like,” said Dr. Buzney, who is also vice-chair of clinical affairs at the Brigham and Women’s Hospital department of dermatology. “There’s an infrequent risk of serious infection and there is risk in patients with HIV, TB, and hepatitis that you have to address. There is also concern for the impact of biologics on patients with a recent cancer.”
On the other hand, few contraindications to NB-UVB exist. According to joint American Academy of Dermatology-National Psoriasis Foundation guidelines on the management and treatment of psoriasis with phototherapy, published in 2019, NB-UVB therapy is only contraindicated in patients with xeroderma pigmentosa and other photosensitive disorders. Concurrent use of cyclosporine and NB-UVB treatment is also contraindicated because of the calculated increase in risk of skin cancer, extrapolated from data on risk with cyclosporine and PUVA (psoralen and ultraviolet A therapy).
The guidelines state that NB-UVB can be used with caution in lupus patients with no history of photosensitivity and who are SS-A negative, as well as patients with a history of melanoma or multiple nonmelanoma skin cancers, a history of recurrent oral herpes simplex virus infection, a history of arsenic intake, prior exposure to ionizing radiation, and those taking photosensitizing medications (since NB-UVB lamps emit “negligible” UVA).
It’s also safe to use during pregnancy and in children. “It’s safe and effective for the right patient,” Dr. Buzney said, discussing how phototherapy can be modified to accommodate children. “You can consider a slower dose-increased regimen. Will children keep the eye protection on? That’s a tricky one. How are you going to manage their anxiety during treatment and involve their family?”
Subgroups of patients who demonstrate a better response to NB-UVB treatment include those with guttate psoriasis, compared with plaque psoriasis, nonsmokers, those with a lower BMI, those with a higher baseline PASI, and those who demonstrate a faster trajectory of clinical response over the first 2-3 weeks of treatment.
Why would one not use phototherapy for psoriasis? “Cost and convenience,” Dr. Buzney said. “There is lost time/revenue to commute to treatment, which may involve multiple times per week. Coming to a public space when COVID-19 is still lingering is another concern, as are the out-of-pocket costs for copays and parking.”
For these reasons, she considers home phototherapy as a transformative option for many patients. Home phototherapy booths provide a safe and effective way to use NB-UVB phototherapy while minimizing copays and commuting costs. The one-time price tag of home NB-UVB booths runs between $5,000 and $7,000, but that is “much less expensive than the biologics,” which can cost $40,000-$50,000 per year, she said.
A small cross-sectional study of office- versus home-based NB-UVB in patients with vitiligo found a cost savings for home-based NB-UVB after 3 months.
One of the challenges with home phototherapy is the lack of long-term studies on patient use. In a small study Dr. Buzney conducted of 30 patients who were prescribed home phototherapy in the last 5 years, 65% practiced (or had practiced) conservative dosing, 83% had continued care with a dermatologist, 19% reported sunburns (5 mild and 1 severe), and 50% had discontinued the therapy at the time of survey because of a perceived lack of efficacy and inconvenience. But 30% of those who had stopped had done so within one month of getting their home booth.
“This tells me that we have to educate our patients better about what expectations should be and make sure they understand how to use their booths,” she said. “Home phototherapy has changed my practice, but not everyone is a candidate for it. Some patients are not dependable. Others are unable to understand instructions.”
Cost to purchase a NB-UVB booth is also an issue, she noted. “Typically, a percentage of cost is covered by insurance, but it’s problematic to purchase a booth if patients don’t know it’s going to work for them or not. Then you have college students who don’t have the space in their apartment or dorm room for a booth.”
Dr. Buzney reported having no relevant financial conflicts.
BOSTON – In 2012, about 50% of patients receiving phototherapy at Brigham and Women’s Hospital in Boston were being treated for psoriasis. A decade later, that proportion has dropped to 20%.
Several factors have contributed to this trend, namely, the development of biologics, the COVID-19 pandemic, “and the rise of home phototherapy options,” Elizabeth A. Buzney, MD, codirector of the phototherapy center at Brigham and Women’s department of dermatology, said at the annual meeting of the American Academy of Dermatology. In her clinical opinion, phototherapy plays an essential role in the treatment of psoriasis.
“It is medically and financially responsible to review the option of phototherapy with every psoriasis patient,” Dr. Buzney said. “Many patients are not medical or financial candidates for biologic/apremilast therapy, or just would prefer nonsystemic therapy.”
In one meta-analysis, the proportion of patients achieving Psoriasis Area and Severity Index (PASI) 75 with NB-UVB therapy was 70% after 20-40 sessions, just below the efficacy of newer biologics – but better than ustekinumab and adalimumab.
“Phototherapy is not so far out of range as you might think it is,” she said, noting that other studies of NB-UVB therapy show PASI 75 responses of 62% and PASI 90 responses of 40%.
Phototherapy can also be an appealing option because biologics aren’t the best option for all patients with psoriasis. They are expensive for the health care system and potentially for patients, require initial and potentially continued lab testing and monitoring, and require injections, “which some patients don’t like,” said Dr. Buzney, who is also vice-chair of clinical affairs at the Brigham and Women’s Hospital department of dermatology. “There’s an infrequent risk of serious infection and there is risk in patients with HIV, TB, and hepatitis that you have to address. There is also concern for the impact of biologics on patients with a recent cancer.”
On the other hand, few contraindications to NB-UVB exist. According to joint American Academy of Dermatology-National Psoriasis Foundation guidelines on the management and treatment of psoriasis with phototherapy, published in 2019, NB-UVB therapy is only contraindicated in patients with xeroderma pigmentosa and other photosensitive disorders. Concurrent use of cyclosporine and NB-UVB treatment is also contraindicated because of the calculated increase in risk of skin cancer, extrapolated from data on risk with cyclosporine and PUVA (psoralen and ultraviolet A therapy).
The guidelines state that NB-UVB can be used with caution in lupus patients with no history of photosensitivity and who are SS-A negative, as well as patients with a history of melanoma or multiple nonmelanoma skin cancers, a history of recurrent oral herpes simplex virus infection, a history of arsenic intake, prior exposure to ionizing radiation, and those taking photosensitizing medications (since NB-UVB lamps emit “negligible” UVA).
It’s also safe to use during pregnancy and in children. “It’s safe and effective for the right patient,” Dr. Buzney said, discussing how phototherapy can be modified to accommodate children. “You can consider a slower dose-increased regimen. Will children keep the eye protection on? That’s a tricky one. How are you going to manage their anxiety during treatment and involve their family?”
Subgroups of patients who demonstrate a better response to NB-UVB treatment include those with guttate psoriasis, compared with plaque psoriasis, nonsmokers, those with a lower BMI, those with a higher baseline PASI, and those who demonstrate a faster trajectory of clinical response over the first 2-3 weeks of treatment.
Why would one not use phototherapy for psoriasis? “Cost and convenience,” Dr. Buzney said. “There is lost time/revenue to commute to treatment, which may involve multiple times per week. Coming to a public space when COVID-19 is still lingering is another concern, as are the out-of-pocket costs for copays and parking.”
For these reasons, she considers home phototherapy as a transformative option for many patients. Home phototherapy booths provide a safe and effective way to use NB-UVB phototherapy while minimizing copays and commuting costs. The one-time price tag of home NB-UVB booths runs between $5,000 and $7,000, but that is “much less expensive than the biologics,” which can cost $40,000-$50,000 per year, she said.
A small cross-sectional study of office- versus home-based NB-UVB in patients with vitiligo found a cost savings for home-based NB-UVB after 3 months.
One of the challenges with home phototherapy is the lack of long-term studies on patient use. In a small study Dr. Buzney conducted of 30 patients who were prescribed home phototherapy in the last 5 years, 65% practiced (or had practiced) conservative dosing, 83% had continued care with a dermatologist, 19% reported sunburns (5 mild and 1 severe), and 50% had discontinued the therapy at the time of survey because of a perceived lack of efficacy and inconvenience. But 30% of those who had stopped had done so within one month of getting their home booth.
“This tells me that we have to educate our patients better about what expectations should be and make sure they understand how to use their booths,” she said. “Home phototherapy has changed my practice, but not everyone is a candidate for it. Some patients are not dependable. Others are unable to understand instructions.”
Cost to purchase a NB-UVB booth is also an issue, she noted. “Typically, a percentage of cost is covered by insurance, but it’s problematic to purchase a booth if patients don’t know it’s going to work for them or not. Then you have college students who don’t have the space in their apartment or dorm room for a booth.”
Dr. Buzney reported having no relevant financial conflicts.
BOSTON – In 2012, about 50% of patients receiving phototherapy at Brigham and Women’s Hospital in Boston were being treated for psoriasis. A decade later, that proportion has dropped to 20%.
Several factors have contributed to this trend, namely, the development of biologics, the COVID-19 pandemic, “and the rise of home phototherapy options,” Elizabeth A. Buzney, MD, codirector of the phototherapy center at Brigham and Women’s department of dermatology, said at the annual meeting of the American Academy of Dermatology. In her clinical opinion, phototherapy plays an essential role in the treatment of psoriasis.
“It is medically and financially responsible to review the option of phototherapy with every psoriasis patient,” Dr. Buzney said. “Many patients are not medical or financial candidates for biologic/apremilast therapy, or just would prefer nonsystemic therapy.”
In one meta-analysis, the proportion of patients achieving Psoriasis Area and Severity Index (PASI) 75 with NB-UVB therapy was 70% after 20-40 sessions, just below the efficacy of newer biologics – but better than ustekinumab and adalimumab.
“Phototherapy is not so far out of range as you might think it is,” she said, noting that other studies of NB-UVB therapy show PASI 75 responses of 62% and PASI 90 responses of 40%.
Phototherapy can also be an appealing option because biologics aren’t the best option for all patients with psoriasis. They are expensive for the health care system and potentially for patients, require initial and potentially continued lab testing and monitoring, and require injections, “which some patients don’t like,” said Dr. Buzney, who is also vice-chair of clinical affairs at the Brigham and Women’s Hospital department of dermatology. “There’s an infrequent risk of serious infection and there is risk in patients with HIV, TB, and hepatitis that you have to address. There is also concern for the impact of biologics on patients with a recent cancer.”
On the other hand, few contraindications to NB-UVB exist. According to joint American Academy of Dermatology-National Psoriasis Foundation guidelines on the management and treatment of psoriasis with phototherapy, published in 2019, NB-UVB therapy is only contraindicated in patients with xeroderma pigmentosa and other photosensitive disorders. Concurrent use of cyclosporine and NB-UVB treatment is also contraindicated because of the calculated increase in risk of skin cancer, extrapolated from data on risk with cyclosporine and PUVA (psoralen and ultraviolet A therapy).
The guidelines state that NB-UVB can be used with caution in lupus patients with no history of photosensitivity and who are SS-A negative, as well as patients with a history of melanoma or multiple nonmelanoma skin cancers, a history of recurrent oral herpes simplex virus infection, a history of arsenic intake, prior exposure to ionizing radiation, and those taking photosensitizing medications (since NB-UVB lamps emit “negligible” UVA).
It’s also safe to use during pregnancy and in children. “It’s safe and effective for the right patient,” Dr. Buzney said, discussing how phototherapy can be modified to accommodate children. “You can consider a slower dose-increased regimen. Will children keep the eye protection on? That’s a tricky one. How are you going to manage their anxiety during treatment and involve their family?”
Subgroups of patients who demonstrate a better response to NB-UVB treatment include those with guttate psoriasis, compared with plaque psoriasis, nonsmokers, those with a lower BMI, those with a higher baseline PASI, and those who demonstrate a faster trajectory of clinical response over the first 2-3 weeks of treatment.
Why would one not use phototherapy for psoriasis? “Cost and convenience,” Dr. Buzney said. “There is lost time/revenue to commute to treatment, which may involve multiple times per week. Coming to a public space when COVID-19 is still lingering is another concern, as are the out-of-pocket costs for copays and parking.”
For these reasons, she considers home phototherapy as a transformative option for many patients. Home phototherapy booths provide a safe and effective way to use NB-UVB phototherapy while minimizing copays and commuting costs. The one-time price tag of home NB-UVB booths runs between $5,000 and $7,000, but that is “much less expensive than the biologics,” which can cost $40,000-$50,000 per year, she said.
A small cross-sectional study of office- versus home-based NB-UVB in patients with vitiligo found a cost savings for home-based NB-UVB after 3 months.
One of the challenges with home phototherapy is the lack of long-term studies on patient use. In a small study Dr. Buzney conducted of 30 patients who were prescribed home phototherapy in the last 5 years, 65% practiced (or had practiced) conservative dosing, 83% had continued care with a dermatologist, 19% reported sunburns (5 mild and 1 severe), and 50% had discontinued the therapy at the time of survey because of a perceived lack of efficacy and inconvenience. But 30% of those who had stopped had done so within one month of getting their home booth.
“This tells me that we have to educate our patients better about what expectations should be and make sure they understand how to use their booths,” she said. “Home phototherapy has changed my practice, but not everyone is a candidate for it. Some patients are not dependable. Others are unable to understand instructions.”
Cost to purchase a NB-UVB booth is also an issue, she noted. “Typically, a percentage of cost is covered by insurance, but it’s problematic to purchase a booth if patients don’t know it’s going to work for them or not. Then you have college students who don’t have the space in their apartment or dorm room for a booth.”
Dr. Buzney reported having no relevant financial conflicts.
AT AAD 22