User login
Dyspareunia: Keys to biopsychosocial evaluation and treatment planning
Dyspareunia is persistent or recurrent pain before, during, or after sexual contact and is not limited to cisgender individuals or vaginal intercourse.1-3 With a prevalence as high as 45% in the United States,2-5 it is one of the most common complaints in gynecologic practices.5,6
Causes and contributing factors
There are many possible causes of dyspareunia.2,4,6 While some patients have a single cause, most cases are complex, with multiple overlapping causes and maintaining factors.4,6 Identifying each contributing factor can help you appropriately address all components.
Physical conditions. The range of physical contributors to dyspareunia includes inflammatory processes, structural abnormalities, musculoskeletal dysfunctions, pelvic organ disorders, injuries, iatrogenic effects, infections, allergic reactions, sensitization, hormonal changes, medication effects, adhesions, autoimmune disorders, and other pain syndromes (TABLE 12-4,6-11).

Inadequate arousal. One of the primary causes of pain during vaginal penetration is inadequate arousal and lubrication.1,2,9-11 Arousal is the phase of the sexual response cycle that leads to genital tumescence and prepares the genitals for sexual contact through penile/clitoral erection, vaginal engorgement, and lubrication, which prevents pain and enhances pleasurable sensation.9-11
While some physical conditions can lead to an inability to lubricate, the most common causes of inadequate lubrication are psychosocial-behavioral, wherein patients have the same physical ability to lubricate as patients without genital pain but do not progress through the arousal phase.9-11 Behavioral factors such as inadequate or ineffective foreplay can fail to produce engorgement and lubrication, while psychosocial factors such as low attraction to partner, relationship stressors, anxiety, or low self-esteem can have an inhibitory effect on sexual arousal.1,2,9-11 Psychosocial and behavioral factors may also be maintaining factors or consequences of dyspareunia, and need to be assessed and treated.1,2,9-11
Psychological trauma. Exposure to psychological traumas and the development of posttraumatic stress disorder (PTSD) have been linked with the development of pain disorders in general and dyspareunia specifically. Most patients seeking treatment for chronic pain disorders have a history of physical or sexual abuse.12 Changes in physiologic processes (eg, neurochemical, endocrine) that occur with PTSD interfere with the sexual response cycle, and sexual traumas specifically have been linked with pelvic floor dysfunction.13,14 Additionally, when PTSD is caused by a sexual trauma, even consensual sexual encounters can trigger flashbacks, intrusive memories, hyperarousal, and muscle tension that interfere with the sexual response cycle and contribute to genital pain.13
Vaginismus is both a physiologic and psychological contributor to dyspareunia.1,2,4 Patients experiencing pain can develop anxiety about repeated pain and involuntarily contract their pelvic muscles, thereby creating more pain, increasing anxiety, decreasing lubrication, and causing pelvic floor dysfunction.1-4,6 Consequently, all patients with dyspareunia should be assessed and continually monitored for symptoms of vaginismus.
Continue to: Anxiety
Anxiety. As with other pain disorders, anxiety develops around pain triggers.10,15 When expecting sexual activity, patients can experience extreme worry and panic attacks.10,15,16 The distress of sexual encounters can interfere with physiologic arousal and sexual desire, impacting all phases of the sexual response cycle.1,2
Relationship issues. Difficulty engaging in or avoidance of sexual activity can interfere with romantic relationships.2,10,16 Severe pain or vaginismus contractions can prevent penetration, leading to unconsummated marriages and an inability to conceive through intercourse.10 The distress surrounding sexual encounters can precipitate erectile dysfunction in male partners, or partners may continue to demand sexual encounters despite the patient’s pain, further impacting the relationship and heightening sexual distress.10 These stressors have led to relationships ending, patients reluctantly agreeing to nonmonogamy to appease their partners, and patients avoiding relationships altogether.10,16
Devalued self-image. Difficulties with sexuality and relationships impact the self-image of patients with dyspareunia. Diminished self-image may include feeling “inadequate” as a woman and as a sexual partner, or feeling like a “failure.”16 Women with dyspareunia often have more distress related to their body image, physical appearance, and genital self-image than do women without genital pain.17 Feeling resentment toward their body, or feeling “ugly,” embarrassed, shamed, “broken,” and “useless” also contribute to increased depressive symptoms found in patients with dyspareunia.16,18
Making the diagnosis
Most patients do not report symptoms unless directly asked2,7; therefore, it is recommended that all patients be screened as a part of an initial intake and before any genital exam (TABLE 22-4,6,7,9,11,19,20).4,7,21 If this screen is positive, a separate appointment may be needed for a thorough evaluation and before any attempt is made at a genital exam.4,7
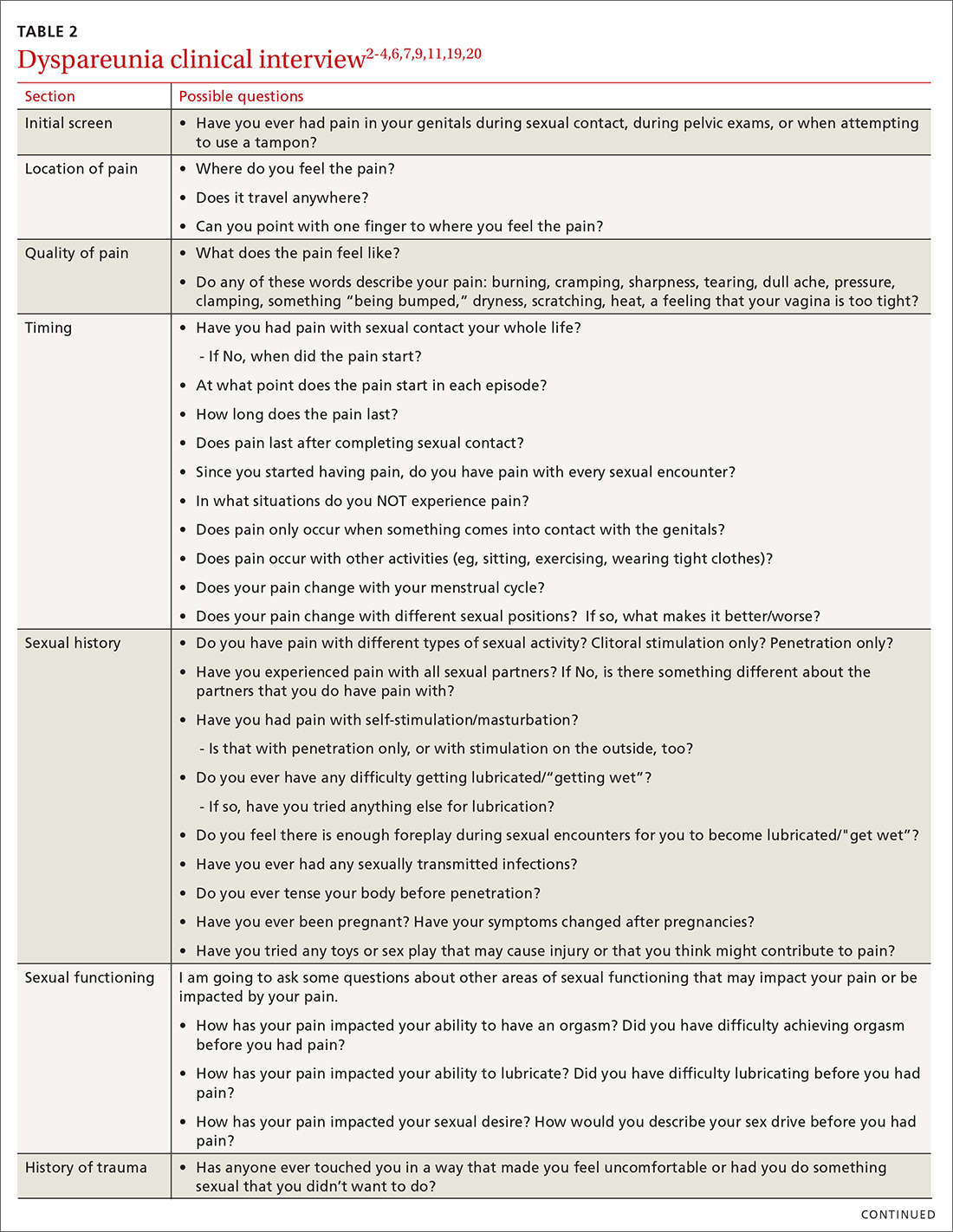
Items to include in the clinical interview
Given the range of possible causes of dyspareunia and its contributing factors and symptoms, a thorough clinical interview is essential. Begin with a review of the patient’s complete medical and surgical history to identify possible known contributors to genital pain.4 Pregnancy history is of particular importance as the prevalence of postpartum dyspareunia is 35%, with risk being greater for patients who experienced dyspareunia symptoms before pregnancy.22

Knowing the location and quality of pain is important for differentiating between possible diagnoses, as is specifying dyspareunia as lifelong or acquired, superficial or deep, and primary or secondary.1-4,6 Confirm the specific location(s) of pain—eg, at the introitus, in the vestibule, on the labia, in the perineum, or near the clitoris.2,4,6 A diagram or model may be needed to help patients to localize pain.4
To help narrow the differential, include the following elements in your assessment: pain quality, timing (eg, initial onset, episode onset, episode duration, situational triggers), alleviating factors, symptoms in surrounding structures (eg, bladder, bowel, muscles, bones), sexual history, other areas of sexual functioning, history of psychological trauma, relationship effects, and mental health (TABLE 22-4,6,7,9,11,19,20 and Table 323-28). Screening for a history of sexual trauma is particularly important, as a recent systematic review and meta-analysis found that women with a history of sexual assault had a 42% higher risk of gynecologic problems overall, a 74% higher risk of dyspareunia, and a 71% higher risk of vaginismus than women without a history of sexual assault.29 Using measures such as the Female Sexual Function Index or the McGill Pain Questionnaire can help patients more thoroughly describe their symptoms (TABLE 323-28).3
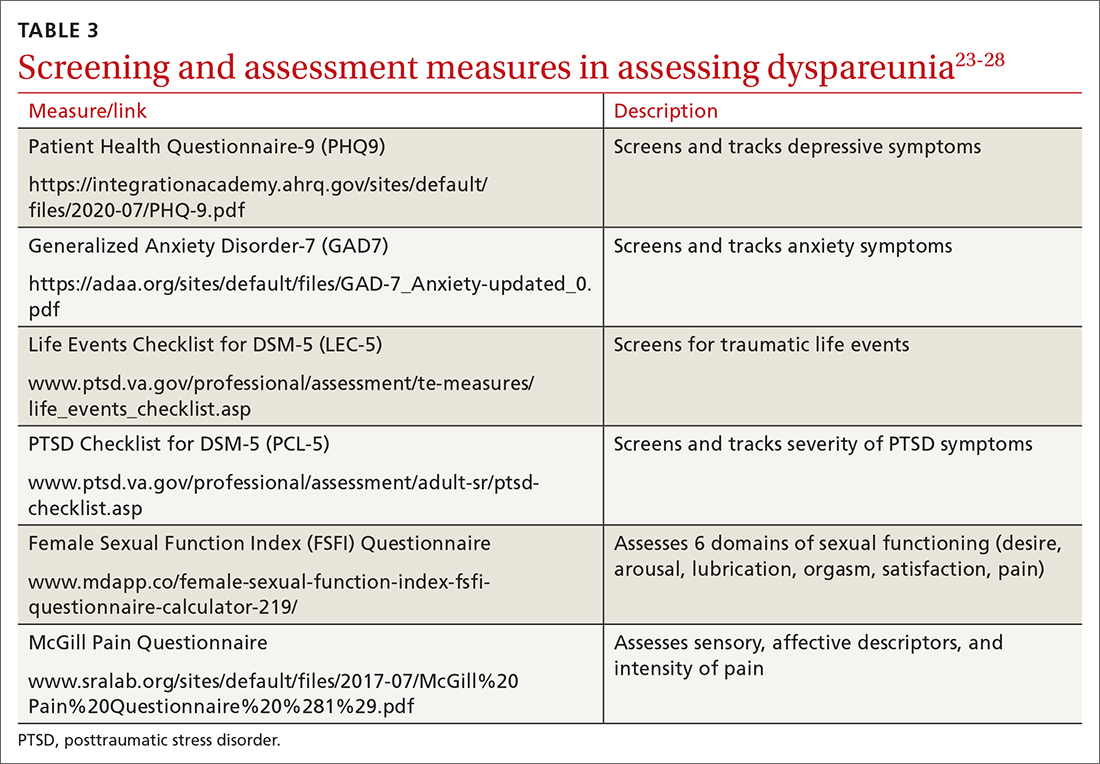
Continue to: Guidelines for the physical exam
Guidelines for the physical exam
Before the exam, ensure the patient has not used any topical genital treatment in the past 2 weeks that may interfere with sensitivity to the exam.4 To decrease patients’ anxiety about the exam, remind them that they can stop the exam at any time.7 Also consider offering the use of a mirror to better pinpoint the location of pain, and to possibly help the patient learn more about her anatomy.2,7
Begin the exam by palpating surrounding areas that may be involved in pain, including the abdomen and musculoskeletal features.3,6,19 Next visually inspect the external genitalia for lesions, abrasions, discoloration, erythema, or other abnormal findings.2,3,6 Ask the patient for permission before contacting the genitals. Because the labia may be a site of pain, apply gentle pressure in retracting it to fully examine the vestibule.6,7 Contraction of the pelvic floor muscles during approach or initial palpation could signal possible vaginismus.4
After visual inspection of external genitalia, use a cotton swab to map the vulva and vestibule in a clockwise fashion to precisely identify any painful locations.2-4,6 If the patient’s history of pain has been intermittent, it’s possible that the cotton swab will not elicit pain on the day of the initial exam, but it may on other days.4
Begin the internal exam by inserting a single finger into the first inch of the vagina and have the patient squeeze and release to assess tenderness, muscle tightness, and control.2,6 Advance the finger further into the vagina and palpate clockwise, examining the levator muscles, obturator muscles, rectum, urethra, and bladder for abnormal tightness or reproduction of pain.2,4,6 Complete a bimanual exam to evaluate the pelvic organs and adnexa.2,4 If indicated, a more thorough evaluation of pelvic floor musculature can be performed by a physical therapist or gynecologist who specializes in pelvic pain.2-4
If the patient consents to further evaluation, consider using a small speculum, advanced slowly, for further internal examination, noting any lesions, abrasions, discharge, ectropion, or tenderness.2-4,7 A rectal exam may also be needed in cases of deep dyspareunia.6 Initial work-up may include a potassium hydroxide wet prep, sexually transmitted infection testing, and pelvic ultrasound.2,4 In some cases, laparoscopy or biopsy may be needed.2,4
Treatments for common causes
Treatment often begins with education about anatomy, to help patients communicate about symptoms and engage more fully in their care.3 Additional education may be needed on genital functioning and the necessity of adequate stimulation and lubrication prior to penetration.1,2,9-11 A discussion of treatments for the wide range of possible causes of dyspareunia is outside the scope of this article. However, some basic behavioral changes may help patients address some of the more common contributing factors.
For example, if vaginal infection is suspected, advise patients to discontinue the use of harsh soaps, known vaginal irritants (eg, perfumed products, bath additives), and douches.3 Recommend using only preservative- and alcohol-free lubricants for sexual contact, and avoiding lubricants with added functions (eg, warming).3 It’s worth noting that avoidance of tight clothing and thong underwear due to possible risk for infections may not be necessary. A recent study found that women who frequently wore thong underwear (more than half of the time) were no more likely to develop urinary tract infections, yeast vaginitis, or bacterial vaginosis than those who avoid such items.30 However, noncotton underwear fabric, rather than tightness, was associated with yeast vaginitis30; therefore, patients may want to consider using only breathable underwear.3
Continue to: Medication
Medication. Medication may be used to treat the underlying contributing conditions or the symptom of pain directly. Some common options are particularly important for patients whose dyspareunia does not have an identifiable cause. These medications include anti-inflammatory agents, topical anesthetics, tricyclic antidepressants, and hormonal treatments.2-4 Since effectiveness varies based on subtypes of pain, select a medication according to the location, timing, and hypothesized mechanism of pain.3,31,32
Medication for deep pain. A meta-analysis and systematic review found that patients with some types of chronic pelvic pain with pain deep in the vagina or pelvis experienced greater than 50% reduction in pain using medroxyprogesterone acetate compared with placebo.33 Other treatments for deep pain depend on physical exam findings.
Medication for superficial pain. Many remedies have been tried, with at least 26 different treatments for vulvodynia pain alone.16 Only some of these treatments have supporting evidence. For patients with vulvar pain, an intent-to-treat RCT found that patients using a topical steroid experienced a 23% reduction in pain from pre-treatment to 6-month follow-up.32
Surgery is also effective for vulvar pain.34,35 For provoked vestibulodynia (in which pain is localized to the vestibule and triggered by contact with the vulva), or vulvar vestibulitis, RCTs have found that vestibulectomy has stronger effects on pain than other treatments,31,35 with a 53% reduction in pain during intercourse and a 70% reduction in vestibular pain overall.35 However, while vestibulectomy is effective for provoked vestibulodynia, it is not recommended for generalized vulvodynia, in which pain is diffuse across the vulva and occurs without vulvar contact.34
Unsupported treatments. A number of other treatments have not yet been found effective. Although lidocaine for vulvar pain is often used, RCTs have not found any significant reduction in symptoms, and a double-blind RCT found that lidocaine ointment actually performed worse than placebo.31,34 Similarly, oral tricyclics have not been found to decrease vulvar pain more than placebo in double-blind studies.31,34 Furthermore, a meta-analysis of RCTs comparing treatments with placebo for vestibular pain found no significant decrease in dyspareunia for topical conjugated estrogen, topical lidocaine, oral desipramine, oral desipramine with topical lidocaine, laser therapy, or transcranial direct current.32
Tx risks to consider. Risks and benefits of dyspareunia treatment options should be thoroughly weighed and discussed with the patient.2-4 Vestibulectomy, despite reducing pain for many patients, has led to increased pain for 9% of patients who underwent the procedure.35 Topical treatments may lead to allergic reactions, inflammation, and worsening of symptoms,4 and hormonal treatments have been found to increase the risk of weight gain and bloating and are not appropriate for patients trying to conceive.33
Coordinate care with other providers
While medications and surgery can reduce pain, they have not been shown to improve other aspects of sexual functioning such as sexual satisfaction, frequency of sexual intercourse, or overall sense of sexual functioning.35 Additionally, pain reduction does not address muscle tension, anxiety, self-esteem, and relationship problems. As a result, a multidisciplinary approach is generally needed.3,4,32,33
Continue to: Physical therapists
Physical therapists. Pelvic floor physical therapists are often members of the dyspareunia treatment team and can provide a thorough evaluation and treatment of pelvic floor disorders.2-4 An RCT with intent-to-treat analysis found that pain was reduced by 71% following pelvic floor physical therapy.36 Another RCT found that 90% of patients reported a clinically meaningful decrease in pain with pelvic floor physical therapy.37 In addition to addressing pain, pelvic floor physical therapy has also been found to improve sexual functioning, sexual satisfaction, distress, and patient perception of improvement.34,36,37
Behavioral health specialists. Psychotherapists, especially those trained in sex therapy, couples therapy, or cognitive behavioral therapy (CBT), are also typically on the treatment team. Multiple RCTs have found evidence of CBT’s effectiveness in the direct treatment of dyspareunia pain. Bergeron et al35 found a 37.5% reduction in vulvar vestibulitis pain intensity during intercourse after patients completed group CBT. Another intent-to-treat RCT found that patients receiving CBT experienced more pain reduction (~ 30%) than patients who were treated with a topical steroid.38
In addition to having a direct impact on pain, CBT has also been found to have a clinically and statistically significant positive impact on other aspects of sexual experience, such as overall sexuality, self-efficacy, overall sexual functioning, frequency of intercourse, and catastrophizing.34,38 A recent meta-analysis of RCTs found that about 80% of vaginismus patients were able to achieve penetrative intercourse after treatment with behavioral sex therapy or CBT.39 This success rate was not exceeded by physical or surgical treatments.39
When PTSD is thought to be a contributing factor, trauma therapy will likely be needed in addition to treatments for dyspareunia. First-line treatments for PTSD include cognitive processing therapy, prolonged exposure, trauma-focused CBT, and cognitive therapy.40
Psychotherapists can also help patients reduce anxiety, reintroduce sexual contact without triggering pain or anxiety, address emotional and self-esteem effects of dyspareunia, address relationship issues, and refocus sexual encounters on pleasure rather than pain avoidance.2-4 Despite patient reports of high treatment satisfaction following therapy,38 many patients may initially lack confidence in psychotherapy as a treatment for pain35 and may need to be educated on its effectiveness and multidimensional benefits.
Gynecologists. Often a gynecologist with specialization in pelvic pain is an essential member of the team for diagnostic clarification, recommendation of treatment options, and performance of more advanced treatments.2,3 If pain has become chronic, the patient may also benefit from a pain management team and support groups.2,3
Follow-up steps
Patients who screen negative for dyspareunia should be re-screened periodically. Continue to assess patients diagnosed with dyspareunia for vaginismus symptoms (if they are not initially present) to ensure that the treatment plan is appropriately adjusted. Once treatment has begun, ask about adverse effects and confidence in the treatment plan to minimize negative impacts on treatment adherence and to anticipate a need for a change in the treatment approach.31,35 In addition to tracking treatment effects on pain, continue to assess for patient-centered outcomes such as emotional functioning, self-esteem, and sexual and relationship satisfaction.34 The Female Sexual Function Index can be a useful tool to track symptoms.27,34
Finally, patients who do not experience sufficient improvement in symptoms and functioning with initial treatment may need continued support and encouragement. Given the broad range of contributing factors and the high number of potential treatments, patients may find hope in learning that multiple other treatment options may be available.
CORRESPONDENCE
Adrienne A. Williams, PhD, Department of Family and Community Medicine, University of Illinois at Chicago College of Medicine, 1919 W Taylor Street, MC 663, Chicago, IL 60612; awms@uic.edu
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th Ed. American Psychiatric Publishing; 2013.
2. Seehusen DA, Baird DC, Bode DV. Dyspareunia in women. Am Fam Phys. 2014;90:465-470.
3. Sorensen J, Bautista KE, Lamvu G, et al. Evaluation and treatment of female sexual pain: a clinical review. Cureus. 2018;10:e2379.
4. MacNeill C. Dyspareunia. Obstet Gynecol Clin North Am. 2006;33:565-77.
5. Latthe P, Latthe M, Say L, et al. WHO systematic review of prevalence of chronic pelvic pain: a neglected reproductive health morbidity. BMC Public Health. 2006;6:177.
6. Steege JF, Zolnoun DA. Evaluation and treatment of dyspareunia. Obstet Gynecol. 2009;113:1124-1136.
7. Williams AA, Williams M. A guide to performing pelvic speculum exams: a patient-centered approach to reducing iatrogenic effects. Teach Learn Med. 2013;25:383-391.
8. Ünlü Z, Yentur A, Çakil N. Pudendal nerve neuropathy: An unknown-rare cause of pelvic pain. Arch Rheumatol. 2016;31:102-103.
9. Dewitte M, Borg C, Lowenstein L. A psychosocial approach to female genital pain. Nat Rev Urol. 2018;15:25-41.
10. Masters WH, Johnson VE. Human Sexual Inadequacy. 1st ed. Little, Brown; 1970.
11. Rathus SA, Nevid JS, Fichner-Rathus L. Human Sexuality in a World of Diversity. 5th ed. Allyn and Bacon; 2002.
12. Bailey BE, Freedenfeld RN, Kiser RS, et al. Lifetime physical and sexual abuse in chronic pain patients: psychosocial correlates and treatment outcomes. Disabil Rehabil. 2003;25:331-342.
13. Yehuda R, Lehrner A, Rosenbaum TY. PTSD and sexual dysfunction in men and women. J Sex Med. 2015;12:1107-1119.
14. Postma R, Bicanic I, van der Vaart H, et al. Pelvic floor muscle problems mediate sexual problems in young adult rape victims. J Sex Med. 2013;10:1978-1987.
15. Binik YM, Bergeron S, Khalifé S. Dyspareunia and vaginismus: so-called sexual pain. In: Leiblum SR, ed. 4th ed. Principles and Practice of Sex Therapy. The Guilford Press; 2007:124-156.
16. Ayling K, Ussher JM. “If sex hurts, am I still a woman?” The subjective experience of vulvodynia in hetero-sexual women. Arch Sex Behav. 2008;37:294-304.
17. Pazmany E, Bergeron S, Van Oudenhove L, et al. Body image and genital self-image in pre-menopausal women with dyspareunia. Arch Sex Behav. 2013;42:999-1010.
18. Maillé DL, Bergeron S, Lambert B. Body image in women with primary and secondary provoked vestibulodynia: a controlled study. J Sex Med. 2015;12:505-515.
19. Ryan L, Hawton K. Female dyspareunia. BMJ. 2004;328:1357.
20. Waldura JF, Arora I, Randall AM, et al. Fifty shades of stigma: exploring the health care experiences of kink-oriented patients. J Sex Med. 2016;13:1918-1929.
21. Hinchliff S, Gott M. Seeking medical help for sexual concerns in mid- and later life: a review of the literature. J Sex Res. 2011;48:106-117.
22. Banaei M, Kariman N, Ozgoli G, et al. Prevalence of postpartum dyspareunia: a systematic review and meta-analysis. Int J Gynaecol Obstet. 2021;153:14-24.
23. Kroenke K, Spitzer RL. The PHQ-9: A new depression diagnostic and severity measure. Psychiatr Ann. 2002;32:509-515.
24. Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
25. U.S. Department of Veterans Affairs. PTSD: National Center for PTSD. Life events checklist for DSM-5 (LEC-5). Accessed February 3, 2022. www.ptsd.va.gov/professional/assessment/te-measures/life_events_checklist.asp
26. Weathers FW, Litz BT, Keane TM, et al. The PTSD checklist for DSM-5 (PCL-5). 2013. Accessed February 3, 2022. www.ptsd.va.gov/professional/assessment/adult-sr/ptsd-checklist.asp
27. Rosen R, Brown C, Heiman J, et al. The female sexual function index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26:191-208.
28. Melzack R. The short-form McGill Pain Questionnaire. Pain. 1987;30:191-197.
29. Hassam T, Kelso E, Chowdary P, et al. Sexual assault as a risk factor for gynaecological morbidity: an exploratory systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2020;255:222-230.
30. Hamlin AA, Sheeder J, Muffly TM. Brief versus thong hygiene in obstetrics and gynecology (B-THONG): a survey study. J Obstet Gynaecol Res. 2019;45:1190-1196.
31. Foster DC, Kotok MB, Huang LS, et al. Oral desipramine and topical lidocaine for vulvodynia: a randomized controlled trial. Obstet Gynecol. 2010;116:583-593.
32. Pérez-López FR, Bueno-Notivol J, Hernandez AV, et al. Systematic review and meta-analysis of the effects of treatment modalities for vestibulodynia in women. Eur J Contracept Reprod Health Care. 2019;24:337-346.
33. Cheong YC, Smotra G, Williams AC. Non-surgical interventions for the management of chronic pelvic pain. Cochrane Database Syst Rev. 2014;(3):CD008797.
34. Goldstein AT, Pukall CF, Brown C, et al. Vulvodynia: assessment and treatment. J Sex Med. 2016;13:572-590.
35. Bergeron S, Binik YM, Khalifé S, et al. A randomized comparison of group cognitive-behavioral therapy, surface electromyographic biofeedback, and vestibulectomy in the treatment of dyspareunia resulting from vulvar vestibulitis. Pain. 2001;91:297-306.
36. Schvartzman R, Schvartzman L, Ferreira CF, et al. Physical therapy intervention for women with dyspareunia: a randomized clinical trial. J Sex Marital Ther. 2019;45:378-394.
37. Morin M, Dumoulin C, Bergeron S, et al. Multimodal physical therapy versus topical lidocaine for provoked vestibulodynia: a multicenter, randomized trial. Am J Obstet Gynecol. 2021;224:189.e1-189.e12.
38. Bergeron S, Khalifé S, Dupuis M-J, et al. A randomized clinical trial comparing group cognitive-behavioral therapy and a topical steroid for women with dyspareunia. J Consult Clin Psychol. 2016;84:259-268.
39. Maseroli E, Scavello I, Rastrelli G, et al. Outcome of medical and psychosexual interventions for vaginismus: a systematic review and meta-analysis. J Sex Med. 2018;15:1752-1764.
40. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. 2017. Accessed February 3, 2022. www.apa.org/ptsd-guideline/ptsd.pdf
Dyspareunia is persistent or recurrent pain before, during, or after sexual contact and is not limited to cisgender individuals or vaginal intercourse.1-3 With a prevalence as high as 45% in the United States,2-5 it is one of the most common complaints in gynecologic practices.5,6
Causes and contributing factors
There are many possible causes of dyspareunia.2,4,6 While some patients have a single cause, most cases are complex, with multiple overlapping causes and maintaining factors.4,6 Identifying each contributing factor can help you appropriately address all components.
Physical conditions. The range of physical contributors to dyspareunia includes inflammatory processes, structural abnormalities, musculoskeletal dysfunctions, pelvic organ disorders, injuries, iatrogenic effects, infections, allergic reactions, sensitization, hormonal changes, medication effects, adhesions, autoimmune disorders, and other pain syndromes (TABLE 12-4,6-11).

Inadequate arousal. One of the primary causes of pain during vaginal penetration is inadequate arousal and lubrication.1,2,9-11 Arousal is the phase of the sexual response cycle that leads to genital tumescence and prepares the genitals for sexual contact through penile/clitoral erection, vaginal engorgement, and lubrication, which prevents pain and enhances pleasurable sensation.9-11
While some physical conditions can lead to an inability to lubricate, the most common causes of inadequate lubrication are psychosocial-behavioral, wherein patients have the same physical ability to lubricate as patients without genital pain but do not progress through the arousal phase.9-11 Behavioral factors such as inadequate or ineffective foreplay can fail to produce engorgement and lubrication, while psychosocial factors such as low attraction to partner, relationship stressors, anxiety, or low self-esteem can have an inhibitory effect on sexual arousal.1,2,9-11 Psychosocial and behavioral factors may also be maintaining factors or consequences of dyspareunia, and need to be assessed and treated.1,2,9-11
Psychological trauma. Exposure to psychological traumas and the development of posttraumatic stress disorder (PTSD) have been linked with the development of pain disorders in general and dyspareunia specifically. Most patients seeking treatment for chronic pain disorders have a history of physical or sexual abuse.12 Changes in physiologic processes (eg, neurochemical, endocrine) that occur with PTSD interfere with the sexual response cycle, and sexual traumas specifically have been linked with pelvic floor dysfunction.13,14 Additionally, when PTSD is caused by a sexual trauma, even consensual sexual encounters can trigger flashbacks, intrusive memories, hyperarousal, and muscle tension that interfere with the sexual response cycle and contribute to genital pain.13
Vaginismus is both a physiologic and psychological contributor to dyspareunia.1,2,4 Patients experiencing pain can develop anxiety about repeated pain and involuntarily contract their pelvic muscles, thereby creating more pain, increasing anxiety, decreasing lubrication, and causing pelvic floor dysfunction.1-4,6 Consequently, all patients with dyspareunia should be assessed and continually monitored for symptoms of vaginismus.
Continue to: Anxiety
Anxiety. As with other pain disorders, anxiety develops around pain triggers.10,15 When expecting sexual activity, patients can experience extreme worry and panic attacks.10,15,16 The distress of sexual encounters can interfere with physiologic arousal and sexual desire, impacting all phases of the sexual response cycle.1,2
Relationship issues. Difficulty engaging in or avoidance of sexual activity can interfere with romantic relationships.2,10,16 Severe pain or vaginismus contractions can prevent penetration, leading to unconsummated marriages and an inability to conceive through intercourse.10 The distress surrounding sexual encounters can precipitate erectile dysfunction in male partners, or partners may continue to demand sexual encounters despite the patient’s pain, further impacting the relationship and heightening sexual distress.10 These stressors have led to relationships ending, patients reluctantly agreeing to nonmonogamy to appease their partners, and patients avoiding relationships altogether.10,16
Devalued self-image. Difficulties with sexuality and relationships impact the self-image of patients with dyspareunia. Diminished self-image may include feeling “inadequate” as a woman and as a sexual partner, or feeling like a “failure.”16 Women with dyspareunia often have more distress related to their body image, physical appearance, and genital self-image than do women without genital pain.17 Feeling resentment toward their body, or feeling “ugly,” embarrassed, shamed, “broken,” and “useless” also contribute to increased depressive symptoms found in patients with dyspareunia.16,18
Making the diagnosis
Most patients do not report symptoms unless directly asked2,7; therefore, it is recommended that all patients be screened as a part of an initial intake and before any genital exam (TABLE 22-4,6,7,9,11,19,20).4,7,21 If this screen is positive, a separate appointment may be needed for a thorough evaluation and before any attempt is made at a genital exam.4,7

Items to include in the clinical interview
Given the range of possible causes of dyspareunia and its contributing factors and symptoms, a thorough clinical interview is essential. Begin with a review of the patient’s complete medical and surgical history to identify possible known contributors to genital pain.4 Pregnancy history is of particular importance as the prevalence of postpartum dyspareunia is 35%, with risk being greater for patients who experienced dyspareunia symptoms before pregnancy.22

Knowing the location and quality of pain is important for differentiating between possible diagnoses, as is specifying dyspareunia as lifelong or acquired, superficial or deep, and primary or secondary.1-4,6 Confirm the specific location(s) of pain—eg, at the introitus, in the vestibule, on the labia, in the perineum, or near the clitoris.2,4,6 A diagram or model may be needed to help patients to localize pain.4
To help narrow the differential, include the following elements in your assessment: pain quality, timing (eg, initial onset, episode onset, episode duration, situational triggers), alleviating factors, symptoms in surrounding structures (eg, bladder, bowel, muscles, bones), sexual history, other areas of sexual functioning, history of psychological trauma, relationship effects, and mental health (TABLE 22-4,6,7,9,11,19,20 and Table 323-28). Screening for a history of sexual trauma is particularly important, as a recent systematic review and meta-analysis found that women with a history of sexual assault had a 42% higher risk of gynecologic problems overall, a 74% higher risk of dyspareunia, and a 71% higher risk of vaginismus than women without a history of sexual assault.29 Using measures such as the Female Sexual Function Index or the McGill Pain Questionnaire can help patients more thoroughly describe their symptoms (TABLE 323-28).3

Continue to: Guidelines for the physical exam
Guidelines for the physical exam
Before the exam, ensure the patient has not used any topical genital treatment in the past 2 weeks that may interfere with sensitivity to the exam.4 To decrease patients’ anxiety about the exam, remind them that they can stop the exam at any time.7 Also consider offering the use of a mirror to better pinpoint the location of pain, and to possibly help the patient learn more about her anatomy.2,7
Begin the exam by palpating surrounding areas that may be involved in pain, including the abdomen and musculoskeletal features.3,6,19 Next visually inspect the external genitalia for lesions, abrasions, discoloration, erythema, or other abnormal findings.2,3,6 Ask the patient for permission before contacting the genitals. Because the labia may be a site of pain, apply gentle pressure in retracting it to fully examine the vestibule.6,7 Contraction of the pelvic floor muscles during approach or initial palpation could signal possible vaginismus.4
After visual inspection of external genitalia, use a cotton swab to map the vulva and vestibule in a clockwise fashion to precisely identify any painful locations.2-4,6 If the patient’s history of pain has been intermittent, it’s possible that the cotton swab will not elicit pain on the day of the initial exam, but it may on other days.4
Begin the internal exam by inserting a single finger into the first inch of the vagina and have the patient squeeze and release to assess tenderness, muscle tightness, and control.2,6 Advance the finger further into the vagina and palpate clockwise, examining the levator muscles, obturator muscles, rectum, urethra, and bladder for abnormal tightness or reproduction of pain.2,4,6 Complete a bimanual exam to evaluate the pelvic organs and adnexa.2,4 If indicated, a more thorough evaluation of pelvic floor musculature can be performed by a physical therapist or gynecologist who specializes in pelvic pain.2-4
If the patient consents to further evaluation, consider using a small speculum, advanced slowly, for further internal examination, noting any lesions, abrasions, discharge, ectropion, or tenderness.2-4,7 A rectal exam may also be needed in cases of deep dyspareunia.6 Initial work-up may include a potassium hydroxide wet prep, sexually transmitted infection testing, and pelvic ultrasound.2,4 In some cases, laparoscopy or biopsy may be needed.2,4
Treatments for common causes
Treatment often begins with education about anatomy, to help patients communicate about symptoms and engage more fully in their care.3 Additional education may be needed on genital functioning and the necessity of adequate stimulation and lubrication prior to penetration.1,2,9-11 A discussion of treatments for the wide range of possible causes of dyspareunia is outside the scope of this article. However, some basic behavioral changes may help patients address some of the more common contributing factors.
For example, if vaginal infection is suspected, advise patients to discontinue the use of harsh soaps, known vaginal irritants (eg, perfumed products, bath additives), and douches.3 Recommend using only preservative- and alcohol-free lubricants for sexual contact, and avoiding lubricants with added functions (eg, warming).3 It’s worth noting that avoidance of tight clothing and thong underwear due to possible risk for infections may not be necessary. A recent study found that women who frequently wore thong underwear (more than half of the time) were no more likely to develop urinary tract infections, yeast vaginitis, or bacterial vaginosis than those who avoid such items.30 However, noncotton underwear fabric, rather than tightness, was associated with yeast vaginitis30; therefore, patients may want to consider using only breathable underwear.3
Continue to: Medication
Medication. Medication may be used to treat the underlying contributing conditions or the symptom of pain directly. Some common options are particularly important for patients whose dyspareunia does not have an identifiable cause. These medications include anti-inflammatory agents, topical anesthetics, tricyclic antidepressants, and hormonal treatments.2-4 Since effectiveness varies based on subtypes of pain, select a medication according to the location, timing, and hypothesized mechanism of pain.3,31,32
Medication for deep pain. A meta-analysis and systematic review found that patients with some types of chronic pelvic pain with pain deep in the vagina or pelvis experienced greater than 50% reduction in pain using medroxyprogesterone acetate compared with placebo.33 Other treatments for deep pain depend on physical exam findings.
Medication for superficial pain. Many remedies have been tried, with at least 26 different treatments for vulvodynia pain alone.16 Only some of these treatments have supporting evidence. For patients with vulvar pain, an intent-to-treat RCT found that patients using a topical steroid experienced a 23% reduction in pain from pre-treatment to 6-month follow-up.32
Surgery is also effective for vulvar pain.34,35 For provoked vestibulodynia (in which pain is localized to the vestibule and triggered by contact with the vulva), or vulvar vestibulitis, RCTs have found that vestibulectomy has stronger effects on pain than other treatments,31,35 with a 53% reduction in pain during intercourse and a 70% reduction in vestibular pain overall.35 However, while vestibulectomy is effective for provoked vestibulodynia, it is not recommended for generalized vulvodynia, in which pain is diffuse across the vulva and occurs without vulvar contact.34
Unsupported treatments. A number of other treatments have not yet been found effective. Although lidocaine for vulvar pain is often used, RCTs have not found any significant reduction in symptoms, and a double-blind RCT found that lidocaine ointment actually performed worse than placebo.31,34 Similarly, oral tricyclics have not been found to decrease vulvar pain more than placebo in double-blind studies.31,34 Furthermore, a meta-analysis of RCTs comparing treatments with placebo for vestibular pain found no significant decrease in dyspareunia for topical conjugated estrogen, topical lidocaine, oral desipramine, oral desipramine with topical lidocaine, laser therapy, or transcranial direct current.32
Tx risks to consider. Risks and benefits of dyspareunia treatment options should be thoroughly weighed and discussed with the patient.2-4 Vestibulectomy, despite reducing pain for many patients, has led to increased pain for 9% of patients who underwent the procedure.35 Topical treatments may lead to allergic reactions, inflammation, and worsening of symptoms,4 and hormonal treatments have been found to increase the risk of weight gain and bloating and are not appropriate for patients trying to conceive.33
Coordinate care with other providers
While medications and surgery can reduce pain, they have not been shown to improve other aspects of sexual functioning such as sexual satisfaction, frequency of sexual intercourse, or overall sense of sexual functioning.35 Additionally, pain reduction does not address muscle tension, anxiety, self-esteem, and relationship problems. As a result, a multidisciplinary approach is generally needed.3,4,32,33
Continue to: Physical therapists
Physical therapists. Pelvic floor physical therapists are often members of the dyspareunia treatment team and can provide a thorough evaluation and treatment of pelvic floor disorders.2-4 An RCT with intent-to-treat analysis found that pain was reduced by 71% following pelvic floor physical therapy.36 Another RCT found that 90% of patients reported a clinically meaningful decrease in pain with pelvic floor physical therapy.37 In addition to addressing pain, pelvic floor physical therapy has also been found to improve sexual functioning, sexual satisfaction, distress, and patient perception of improvement.34,36,37
Behavioral health specialists. Psychotherapists, especially those trained in sex therapy, couples therapy, or cognitive behavioral therapy (CBT), are also typically on the treatment team. Multiple RCTs have found evidence of CBT’s effectiveness in the direct treatment of dyspareunia pain. Bergeron et al35 found a 37.5% reduction in vulvar vestibulitis pain intensity during intercourse after patients completed group CBT. Another intent-to-treat RCT found that patients receiving CBT experienced more pain reduction (~ 30%) than patients who were treated with a topical steroid.38
In addition to having a direct impact on pain, CBT has also been found to have a clinically and statistically significant positive impact on other aspects of sexual experience, such as overall sexuality, self-efficacy, overall sexual functioning, frequency of intercourse, and catastrophizing.34,38 A recent meta-analysis of RCTs found that about 80% of vaginismus patients were able to achieve penetrative intercourse after treatment with behavioral sex therapy or CBT.39 This success rate was not exceeded by physical or surgical treatments.39
When PTSD is thought to be a contributing factor, trauma therapy will likely be needed in addition to treatments for dyspareunia. First-line treatments for PTSD include cognitive processing therapy, prolonged exposure, trauma-focused CBT, and cognitive therapy.40
Psychotherapists can also help patients reduce anxiety, reintroduce sexual contact without triggering pain or anxiety, address emotional and self-esteem effects of dyspareunia, address relationship issues, and refocus sexual encounters on pleasure rather than pain avoidance.2-4 Despite patient reports of high treatment satisfaction following therapy,38 many patients may initially lack confidence in psychotherapy as a treatment for pain35 and may need to be educated on its effectiveness and multidimensional benefits.
Gynecologists. Often a gynecologist with specialization in pelvic pain is an essential member of the team for diagnostic clarification, recommendation of treatment options, and performance of more advanced treatments.2,3 If pain has become chronic, the patient may also benefit from a pain management team and support groups.2,3
Follow-up steps
Patients who screen negative for dyspareunia should be re-screened periodically. Continue to assess patients diagnosed with dyspareunia for vaginismus symptoms (if they are not initially present) to ensure that the treatment plan is appropriately adjusted. Once treatment has begun, ask about adverse effects and confidence in the treatment plan to minimize negative impacts on treatment adherence and to anticipate a need for a change in the treatment approach.31,35 In addition to tracking treatment effects on pain, continue to assess for patient-centered outcomes such as emotional functioning, self-esteem, and sexual and relationship satisfaction.34 The Female Sexual Function Index can be a useful tool to track symptoms.27,34
Finally, patients who do not experience sufficient improvement in symptoms and functioning with initial treatment may need continued support and encouragement. Given the broad range of contributing factors and the high number of potential treatments, patients may find hope in learning that multiple other treatment options may be available.
CORRESPONDENCE
Adrienne A. Williams, PhD, Department of Family and Community Medicine, University of Illinois at Chicago College of Medicine, 1919 W Taylor Street, MC 663, Chicago, IL 60612; awms@uic.edu
Dyspareunia is persistent or recurrent pain before, during, or after sexual contact and is not limited to cisgender individuals or vaginal intercourse.1-3 With a prevalence as high as 45% in the United States,2-5 it is one of the most common complaints in gynecologic practices.5,6
Causes and contributing factors
There are many possible causes of dyspareunia.2,4,6 While some patients have a single cause, most cases are complex, with multiple overlapping causes and maintaining factors.4,6 Identifying each contributing factor can help you appropriately address all components.
Physical conditions. The range of physical contributors to dyspareunia includes inflammatory processes, structural abnormalities, musculoskeletal dysfunctions, pelvic organ disorders, injuries, iatrogenic effects, infections, allergic reactions, sensitization, hormonal changes, medication effects, adhesions, autoimmune disorders, and other pain syndromes (TABLE 12-4,6-11).

Inadequate arousal. One of the primary causes of pain during vaginal penetration is inadequate arousal and lubrication.1,2,9-11 Arousal is the phase of the sexual response cycle that leads to genital tumescence and prepares the genitals for sexual contact through penile/clitoral erection, vaginal engorgement, and lubrication, which prevents pain and enhances pleasurable sensation.9-11
While some physical conditions can lead to an inability to lubricate, the most common causes of inadequate lubrication are psychosocial-behavioral, wherein patients have the same physical ability to lubricate as patients without genital pain but do not progress through the arousal phase.9-11 Behavioral factors such as inadequate or ineffective foreplay can fail to produce engorgement and lubrication, while psychosocial factors such as low attraction to partner, relationship stressors, anxiety, or low self-esteem can have an inhibitory effect on sexual arousal.1,2,9-11 Psychosocial and behavioral factors may also be maintaining factors or consequences of dyspareunia, and need to be assessed and treated.1,2,9-11
Psychological trauma. Exposure to psychological traumas and the development of posttraumatic stress disorder (PTSD) have been linked with the development of pain disorders in general and dyspareunia specifically. Most patients seeking treatment for chronic pain disorders have a history of physical or sexual abuse.12 Changes in physiologic processes (eg, neurochemical, endocrine) that occur with PTSD interfere with the sexual response cycle, and sexual traumas specifically have been linked with pelvic floor dysfunction.13,14 Additionally, when PTSD is caused by a sexual trauma, even consensual sexual encounters can trigger flashbacks, intrusive memories, hyperarousal, and muscle tension that interfere with the sexual response cycle and contribute to genital pain.13
Vaginismus is both a physiologic and psychological contributor to dyspareunia.1,2,4 Patients experiencing pain can develop anxiety about repeated pain and involuntarily contract their pelvic muscles, thereby creating more pain, increasing anxiety, decreasing lubrication, and causing pelvic floor dysfunction.1-4,6 Consequently, all patients with dyspareunia should be assessed and continually monitored for symptoms of vaginismus.
Continue to: Anxiety
Anxiety. As with other pain disorders, anxiety develops around pain triggers.10,15 When expecting sexual activity, patients can experience extreme worry and panic attacks.10,15,16 The distress of sexual encounters can interfere with physiologic arousal and sexual desire, impacting all phases of the sexual response cycle.1,2
Relationship issues. Difficulty engaging in or avoidance of sexual activity can interfere with romantic relationships.2,10,16 Severe pain or vaginismus contractions can prevent penetration, leading to unconsummated marriages and an inability to conceive through intercourse.10 The distress surrounding sexual encounters can precipitate erectile dysfunction in male partners, or partners may continue to demand sexual encounters despite the patient’s pain, further impacting the relationship and heightening sexual distress.10 These stressors have led to relationships ending, patients reluctantly agreeing to nonmonogamy to appease their partners, and patients avoiding relationships altogether.10,16
Devalued self-image. Difficulties with sexuality and relationships impact the self-image of patients with dyspareunia. Diminished self-image may include feeling “inadequate” as a woman and as a sexual partner, or feeling like a “failure.”16 Women with dyspareunia often have more distress related to their body image, physical appearance, and genital self-image than do women without genital pain.17 Feeling resentment toward their body, or feeling “ugly,” embarrassed, shamed, “broken,” and “useless” also contribute to increased depressive symptoms found in patients with dyspareunia.16,18
Making the diagnosis
Most patients do not report symptoms unless directly asked2,7; therefore, it is recommended that all patients be screened as a part of an initial intake and before any genital exam (TABLE 22-4,6,7,9,11,19,20).4,7,21 If this screen is positive, a separate appointment may be needed for a thorough evaluation and before any attempt is made at a genital exam.4,7

Items to include in the clinical interview
Given the range of possible causes of dyspareunia and its contributing factors and symptoms, a thorough clinical interview is essential. Begin with a review of the patient’s complete medical and surgical history to identify possible known contributors to genital pain.4 Pregnancy history is of particular importance as the prevalence of postpartum dyspareunia is 35%, with risk being greater for patients who experienced dyspareunia symptoms before pregnancy.22

Knowing the location and quality of pain is important for differentiating between possible diagnoses, as is specifying dyspareunia as lifelong or acquired, superficial or deep, and primary or secondary.1-4,6 Confirm the specific location(s) of pain—eg, at the introitus, in the vestibule, on the labia, in the perineum, or near the clitoris.2,4,6 A diagram or model may be needed to help patients to localize pain.4
To help narrow the differential, include the following elements in your assessment: pain quality, timing (eg, initial onset, episode onset, episode duration, situational triggers), alleviating factors, symptoms in surrounding structures (eg, bladder, bowel, muscles, bones), sexual history, other areas of sexual functioning, history of psychological trauma, relationship effects, and mental health (TABLE 22-4,6,7,9,11,19,20 and Table 323-28). Screening for a history of sexual trauma is particularly important, as a recent systematic review and meta-analysis found that women with a history of sexual assault had a 42% higher risk of gynecologic problems overall, a 74% higher risk of dyspareunia, and a 71% higher risk of vaginismus than women without a history of sexual assault.29 Using measures such as the Female Sexual Function Index or the McGill Pain Questionnaire can help patients more thoroughly describe their symptoms (TABLE 323-28).3

Continue to: Guidelines for the physical exam
Guidelines for the physical exam
Before the exam, ensure the patient has not used any topical genital treatment in the past 2 weeks that may interfere with sensitivity to the exam.4 To decrease patients’ anxiety about the exam, remind them that they can stop the exam at any time.7 Also consider offering the use of a mirror to better pinpoint the location of pain, and to possibly help the patient learn more about her anatomy.2,7
Begin the exam by palpating surrounding areas that may be involved in pain, including the abdomen and musculoskeletal features.3,6,19 Next visually inspect the external genitalia for lesions, abrasions, discoloration, erythema, or other abnormal findings.2,3,6 Ask the patient for permission before contacting the genitals. Because the labia may be a site of pain, apply gentle pressure in retracting it to fully examine the vestibule.6,7 Contraction of the pelvic floor muscles during approach or initial palpation could signal possible vaginismus.4
After visual inspection of external genitalia, use a cotton swab to map the vulva and vestibule in a clockwise fashion to precisely identify any painful locations.2-4,6 If the patient’s history of pain has been intermittent, it’s possible that the cotton swab will not elicit pain on the day of the initial exam, but it may on other days.4
Begin the internal exam by inserting a single finger into the first inch of the vagina and have the patient squeeze and release to assess tenderness, muscle tightness, and control.2,6 Advance the finger further into the vagina and palpate clockwise, examining the levator muscles, obturator muscles, rectum, urethra, and bladder for abnormal tightness or reproduction of pain.2,4,6 Complete a bimanual exam to evaluate the pelvic organs and adnexa.2,4 If indicated, a more thorough evaluation of pelvic floor musculature can be performed by a physical therapist or gynecologist who specializes in pelvic pain.2-4
If the patient consents to further evaluation, consider using a small speculum, advanced slowly, for further internal examination, noting any lesions, abrasions, discharge, ectropion, or tenderness.2-4,7 A rectal exam may also be needed in cases of deep dyspareunia.6 Initial work-up may include a potassium hydroxide wet prep, sexually transmitted infection testing, and pelvic ultrasound.2,4 In some cases, laparoscopy or biopsy may be needed.2,4
Treatments for common causes
Treatment often begins with education about anatomy, to help patients communicate about symptoms and engage more fully in their care.3 Additional education may be needed on genital functioning and the necessity of adequate stimulation and lubrication prior to penetration.1,2,9-11 A discussion of treatments for the wide range of possible causes of dyspareunia is outside the scope of this article. However, some basic behavioral changes may help patients address some of the more common contributing factors.
For example, if vaginal infection is suspected, advise patients to discontinue the use of harsh soaps, known vaginal irritants (eg, perfumed products, bath additives), and douches.3 Recommend using only preservative- and alcohol-free lubricants for sexual contact, and avoiding lubricants with added functions (eg, warming).3 It’s worth noting that avoidance of tight clothing and thong underwear due to possible risk for infections may not be necessary. A recent study found that women who frequently wore thong underwear (more than half of the time) were no more likely to develop urinary tract infections, yeast vaginitis, or bacterial vaginosis than those who avoid such items.30 However, noncotton underwear fabric, rather than tightness, was associated with yeast vaginitis30; therefore, patients may want to consider using only breathable underwear.3
Continue to: Medication
Medication. Medication may be used to treat the underlying contributing conditions or the symptom of pain directly. Some common options are particularly important for patients whose dyspareunia does not have an identifiable cause. These medications include anti-inflammatory agents, topical anesthetics, tricyclic antidepressants, and hormonal treatments.2-4 Since effectiveness varies based on subtypes of pain, select a medication according to the location, timing, and hypothesized mechanism of pain.3,31,32
Medication for deep pain. A meta-analysis and systematic review found that patients with some types of chronic pelvic pain with pain deep in the vagina or pelvis experienced greater than 50% reduction in pain using medroxyprogesterone acetate compared with placebo.33 Other treatments for deep pain depend on physical exam findings.
Medication for superficial pain. Many remedies have been tried, with at least 26 different treatments for vulvodynia pain alone.16 Only some of these treatments have supporting evidence. For patients with vulvar pain, an intent-to-treat RCT found that patients using a topical steroid experienced a 23% reduction in pain from pre-treatment to 6-month follow-up.32
Surgery is also effective for vulvar pain.34,35 For provoked vestibulodynia (in which pain is localized to the vestibule and triggered by contact with the vulva), or vulvar vestibulitis, RCTs have found that vestibulectomy has stronger effects on pain than other treatments,31,35 with a 53% reduction in pain during intercourse and a 70% reduction in vestibular pain overall.35 However, while vestibulectomy is effective for provoked vestibulodynia, it is not recommended for generalized vulvodynia, in which pain is diffuse across the vulva and occurs without vulvar contact.34
Unsupported treatments. A number of other treatments have not yet been found effective. Although lidocaine for vulvar pain is often used, RCTs have not found any significant reduction in symptoms, and a double-blind RCT found that lidocaine ointment actually performed worse than placebo.31,34 Similarly, oral tricyclics have not been found to decrease vulvar pain more than placebo in double-blind studies.31,34 Furthermore, a meta-analysis of RCTs comparing treatments with placebo for vestibular pain found no significant decrease in dyspareunia for topical conjugated estrogen, topical lidocaine, oral desipramine, oral desipramine with topical lidocaine, laser therapy, or transcranial direct current.32
Tx risks to consider. Risks and benefits of dyspareunia treatment options should be thoroughly weighed and discussed with the patient.2-4 Vestibulectomy, despite reducing pain for many patients, has led to increased pain for 9% of patients who underwent the procedure.35 Topical treatments may lead to allergic reactions, inflammation, and worsening of symptoms,4 and hormonal treatments have been found to increase the risk of weight gain and bloating and are not appropriate for patients trying to conceive.33
Coordinate care with other providers
While medications and surgery can reduce pain, they have not been shown to improve other aspects of sexual functioning such as sexual satisfaction, frequency of sexual intercourse, or overall sense of sexual functioning.35 Additionally, pain reduction does not address muscle tension, anxiety, self-esteem, and relationship problems. As a result, a multidisciplinary approach is generally needed.3,4,32,33
Continue to: Physical therapists
Physical therapists. Pelvic floor physical therapists are often members of the dyspareunia treatment team and can provide a thorough evaluation and treatment of pelvic floor disorders.2-4 An RCT with intent-to-treat analysis found that pain was reduced by 71% following pelvic floor physical therapy.36 Another RCT found that 90% of patients reported a clinically meaningful decrease in pain with pelvic floor physical therapy.37 In addition to addressing pain, pelvic floor physical therapy has also been found to improve sexual functioning, sexual satisfaction, distress, and patient perception of improvement.34,36,37
Behavioral health specialists. Psychotherapists, especially those trained in sex therapy, couples therapy, or cognitive behavioral therapy (CBT), are also typically on the treatment team. Multiple RCTs have found evidence of CBT’s effectiveness in the direct treatment of dyspareunia pain. Bergeron et al35 found a 37.5% reduction in vulvar vestibulitis pain intensity during intercourse after patients completed group CBT. Another intent-to-treat RCT found that patients receiving CBT experienced more pain reduction (~ 30%) than patients who were treated with a topical steroid.38
In addition to having a direct impact on pain, CBT has also been found to have a clinically and statistically significant positive impact on other aspects of sexual experience, such as overall sexuality, self-efficacy, overall sexual functioning, frequency of intercourse, and catastrophizing.34,38 A recent meta-analysis of RCTs found that about 80% of vaginismus patients were able to achieve penetrative intercourse after treatment with behavioral sex therapy or CBT.39 This success rate was not exceeded by physical or surgical treatments.39
When PTSD is thought to be a contributing factor, trauma therapy will likely be needed in addition to treatments for dyspareunia. First-line treatments for PTSD include cognitive processing therapy, prolonged exposure, trauma-focused CBT, and cognitive therapy.40
Psychotherapists can also help patients reduce anxiety, reintroduce sexual contact without triggering pain or anxiety, address emotional and self-esteem effects of dyspareunia, address relationship issues, and refocus sexual encounters on pleasure rather than pain avoidance.2-4 Despite patient reports of high treatment satisfaction following therapy,38 many patients may initially lack confidence in psychotherapy as a treatment for pain35 and may need to be educated on its effectiveness and multidimensional benefits.
Gynecologists. Often a gynecologist with specialization in pelvic pain is an essential member of the team for diagnostic clarification, recommendation of treatment options, and performance of more advanced treatments.2,3 If pain has become chronic, the patient may also benefit from a pain management team and support groups.2,3
Follow-up steps
Patients who screen negative for dyspareunia should be re-screened periodically. Continue to assess patients diagnosed with dyspareunia for vaginismus symptoms (if they are not initially present) to ensure that the treatment plan is appropriately adjusted. Once treatment has begun, ask about adverse effects and confidence in the treatment plan to minimize negative impacts on treatment adherence and to anticipate a need for a change in the treatment approach.31,35 In addition to tracking treatment effects on pain, continue to assess for patient-centered outcomes such as emotional functioning, self-esteem, and sexual and relationship satisfaction.34 The Female Sexual Function Index can be a useful tool to track symptoms.27,34
Finally, patients who do not experience sufficient improvement in symptoms and functioning with initial treatment may need continued support and encouragement. Given the broad range of contributing factors and the high number of potential treatments, patients may find hope in learning that multiple other treatment options may be available.
CORRESPONDENCE
Adrienne A. Williams, PhD, Department of Family and Community Medicine, University of Illinois at Chicago College of Medicine, 1919 W Taylor Street, MC 663, Chicago, IL 60612; awms@uic.edu
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th Ed. American Psychiatric Publishing; 2013.
2. Seehusen DA, Baird DC, Bode DV. Dyspareunia in women. Am Fam Phys. 2014;90:465-470.
3. Sorensen J, Bautista KE, Lamvu G, et al. Evaluation and treatment of female sexual pain: a clinical review. Cureus. 2018;10:e2379.
4. MacNeill C. Dyspareunia. Obstet Gynecol Clin North Am. 2006;33:565-77.
5. Latthe P, Latthe M, Say L, et al. WHO systematic review of prevalence of chronic pelvic pain: a neglected reproductive health morbidity. BMC Public Health. 2006;6:177.
6. Steege JF, Zolnoun DA. Evaluation and treatment of dyspareunia. Obstet Gynecol. 2009;113:1124-1136.
7. Williams AA, Williams M. A guide to performing pelvic speculum exams: a patient-centered approach to reducing iatrogenic effects. Teach Learn Med. 2013;25:383-391.
8. Ünlü Z, Yentur A, Çakil N. Pudendal nerve neuropathy: An unknown-rare cause of pelvic pain. Arch Rheumatol. 2016;31:102-103.
9. Dewitte M, Borg C, Lowenstein L. A psychosocial approach to female genital pain. Nat Rev Urol. 2018;15:25-41.
10. Masters WH, Johnson VE. Human Sexual Inadequacy. 1st ed. Little, Brown; 1970.
11. Rathus SA, Nevid JS, Fichner-Rathus L. Human Sexuality in a World of Diversity. 5th ed. Allyn and Bacon; 2002.
12. Bailey BE, Freedenfeld RN, Kiser RS, et al. Lifetime physical and sexual abuse in chronic pain patients: psychosocial correlates and treatment outcomes. Disabil Rehabil. 2003;25:331-342.
13. Yehuda R, Lehrner A, Rosenbaum TY. PTSD and sexual dysfunction in men and women. J Sex Med. 2015;12:1107-1119.
14. Postma R, Bicanic I, van der Vaart H, et al. Pelvic floor muscle problems mediate sexual problems in young adult rape victims. J Sex Med. 2013;10:1978-1987.
15. Binik YM, Bergeron S, Khalifé S. Dyspareunia and vaginismus: so-called sexual pain. In: Leiblum SR, ed. 4th ed. Principles and Practice of Sex Therapy. The Guilford Press; 2007:124-156.
16. Ayling K, Ussher JM. “If sex hurts, am I still a woman?” The subjective experience of vulvodynia in hetero-sexual women. Arch Sex Behav. 2008;37:294-304.
17. Pazmany E, Bergeron S, Van Oudenhove L, et al. Body image and genital self-image in pre-menopausal women with dyspareunia. Arch Sex Behav. 2013;42:999-1010.
18. Maillé DL, Bergeron S, Lambert B. Body image in women with primary and secondary provoked vestibulodynia: a controlled study. J Sex Med. 2015;12:505-515.
19. Ryan L, Hawton K. Female dyspareunia. BMJ. 2004;328:1357.
20. Waldura JF, Arora I, Randall AM, et al. Fifty shades of stigma: exploring the health care experiences of kink-oriented patients. J Sex Med. 2016;13:1918-1929.
21. Hinchliff S, Gott M. Seeking medical help for sexual concerns in mid- and later life: a review of the literature. J Sex Res. 2011;48:106-117.
22. Banaei M, Kariman N, Ozgoli G, et al. Prevalence of postpartum dyspareunia: a systematic review and meta-analysis. Int J Gynaecol Obstet. 2021;153:14-24.
23. Kroenke K, Spitzer RL. The PHQ-9: A new depression diagnostic and severity measure. Psychiatr Ann. 2002;32:509-515.
24. Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
25. U.S. Department of Veterans Affairs. PTSD: National Center for PTSD. Life events checklist for DSM-5 (LEC-5). Accessed February 3, 2022. www.ptsd.va.gov/professional/assessment/te-measures/life_events_checklist.asp
26. Weathers FW, Litz BT, Keane TM, et al. The PTSD checklist for DSM-5 (PCL-5). 2013. Accessed February 3, 2022. www.ptsd.va.gov/professional/assessment/adult-sr/ptsd-checklist.asp
27. Rosen R, Brown C, Heiman J, et al. The female sexual function index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26:191-208.
28. Melzack R. The short-form McGill Pain Questionnaire. Pain. 1987;30:191-197.
29. Hassam T, Kelso E, Chowdary P, et al. Sexual assault as a risk factor for gynaecological morbidity: an exploratory systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2020;255:222-230.
30. Hamlin AA, Sheeder J, Muffly TM. Brief versus thong hygiene in obstetrics and gynecology (B-THONG): a survey study. J Obstet Gynaecol Res. 2019;45:1190-1196.
31. Foster DC, Kotok MB, Huang LS, et al. Oral desipramine and topical lidocaine for vulvodynia: a randomized controlled trial. Obstet Gynecol. 2010;116:583-593.
32. Pérez-López FR, Bueno-Notivol J, Hernandez AV, et al. Systematic review and meta-analysis of the effects of treatment modalities for vestibulodynia in women. Eur J Contracept Reprod Health Care. 2019;24:337-346.
33. Cheong YC, Smotra G, Williams AC. Non-surgical interventions for the management of chronic pelvic pain. Cochrane Database Syst Rev. 2014;(3):CD008797.
34. Goldstein AT, Pukall CF, Brown C, et al. Vulvodynia: assessment and treatment. J Sex Med. 2016;13:572-590.
35. Bergeron S, Binik YM, Khalifé S, et al. A randomized comparison of group cognitive-behavioral therapy, surface electromyographic biofeedback, and vestibulectomy in the treatment of dyspareunia resulting from vulvar vestibulitis. Pain. 2001;91:297-306.
36. Schvartzman R, Schvartzman L, Ferreira CF, et al. Physical therapy intervention for women with dyspareunia: a randomized clinical trial. J Sex Marital Ther. 2019;45:378-394.
37. Morin M, Dumoulin C, Bergeron S, et al. Multimodal physical therapy versus topical lidocaine for provoked vestibulodynia: a multicenter, randomized trial. Am J Obstet Gynecol. 2021;224:189.e1-189.e12.
38. Bergeron S, Khalifé S, Dupuis M-J, et al. A randomized clinical trial comparing group cognitive-behavioral therapy and a topical steroid for women with dyspareunia. J Consult Clin Psychol. 2016;84:259-268.
39. Maseroli E, Scavello I, Rastrelli G, et al. Outcome of medical and psychosexual interventions for vaginismus: a systematic review and meta-analysis. J Sex Med. 2018;15:1752-1764.
40. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. 2017. Accessed February 3, 2022. www.apa.org/ptsd-guideline/ptsd.pdf
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th Ed. American Psychiatric Publishing; 2013.
2. Seehusen DA, Baird DC, Bode DV. Dyspareunia in women. Am Fam Phys. 2014;90:465-470.
3. Sorensen J, Bautista KE, Lamvu G, et al. Evaluation and treatment of female sexual pain: a clinical review. Cureus. 2018;10:e2379.
4. MacNeill C. Dyspareunia. Obstet Gynecol Clin North Am. 2006;33:565-77.
5. Latthe P, Latthe M, Say L, et al. WHO systematic review of prevalence of chronic pelvic pain: a neglected reproductive health morbidity. BMC Public Health. 2006;6:177.
6. Steege JF, Zolnoun DA. Evaluation and treatment of dyspareunia. Obstet Gynecol. 2009;113:1124-1136.
7. Williams AA, Williams M. A guide to performing pelvic speculum exams: a patient-centered approach to reducing iatrogenic effects. Teach Learn Med. 2013;25:383-391.
8. Ünlü Z, Yentur A, Çakil N. Pudendal nerve neuropathy: An unknown-rare cause of pelvic pain. Arch Rheumatol. 2016;31:102-103.
9. Dewitte M, Borg C, Lowenstein L. A psychosocial approach to female genital pain. Nat Rev Urol. 2018;15:25-41.
10. Masters WH, Johnson VE. Human Sexual Inadequacy. 1st ed. Little, Brown; 1970.
11. Rathus SA, Nevid JS, Fichner-Rathus L. Human Sexuality in a World of Diversity. 5th ed. Allyn and Bacon; 2002.
12. Bailey BE, Freedenfeld RN, Kiser RS, et al. Lifetime physical and sexual abuse in chronic pain patients: psychosocial correlates and treatment outcomes. Disabil Rehabil. 2003;25:331-342.
13. Yehuda R, Lehrner A, Rosenbaum TY. PTSD and sexual dysfunction in men and women. J Sex Med. 2015;12:1107-1119.
14. Postma R, Bicanic I, van der Vaart H, et al. Pelvic floor muscle problems mediate sexual problems in young adult rape victims. J Sex Med. 2013;10:1978-1987.
15. Binik YM, Bergeron S, Khalifé S. Dyspareunia and vaginismus: so-called sexual pain. In: Leiblum SR, ed. 4th ed. Principles and Practice of Sex Therapy. The Guilford Press; 2007:124-156.
16. Ayling K, Ussher JM. “If sex hurts, am I still a woman?” The subjective experience of vulvodynia in hetero-sexual women. Arch Sex Behav. 2008;37:294-304.
17. Pazmany E, Bergeron S, Van Oudenhove L, et al. Body image and genital self-image in pre-menopausal women with dyspareunia. Arch Sex Behav. 2013;42:999-1010.
18. Maillé DL, Bergeron S, Lambert B. Body image in women with primary and secondary provoked vestibulodynia: a controlled study. J Sex Med. 2015;12:505-515.
19. Ryan L, Hawton K. Female dyspareunia. BMJ. 2004;328:1357.
20. Waldura JF, Arora I, Randall AM, et al. Fifty shades of stigma: exploring the health care experiences of kink-oriented patients. J Sex Med. 2016;13:1918-1929.
21. Hinchliff S, Gott M. Seeking medical help for sexual concerns in mid- and later life: a review of the literature. J Sex Res. 2011;48:106-117.
22. Banaei M, Kariman N, Ozgoli G, et al. Prevalence of postpartum dyspareunia: a systematic review and meta-analysis. Int J Gynaecol Obstet. 2021;153:14-24.
23. Kroenke K, Spitzer RL. The PHQ-9: A new depression diagnostic and severity measure. Psychiatr Ann. 2002;32:509-515.
24. Spitzer RL, Kroenke K, Williams JB, et al. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092-1097.
25. U.S. Department of Veterans Affairs. PTSD: National Center for PTSD. Life events checklist for DSM-5 (LEC-5). Accessed February 3, 2022. www.ptsd.va.gov/professional/assessment/te-measures/life_events_checklist.asp
26. Weathers FW, Litz BT, Keane TM, et al. The PTSD checklist for DSM-5 (PCL-5). 2013. Accessed February 3, 2022. www.ptsd.va.gov/professional/assessment/adult-sr/ptsd-checklist.asp
27. Rosen R, Brown C, Heiman J, et al. The female sexual function index (FSFI): a multidimensional self-report instrument for the assessment of female sexual function. J Sex Marital Ther. 2000;26:191-208.
28. Melzack R. The short-form McGill Pain Questionnaire. Pain. 1987;30:191-197.
29. Hassam T, Kelso E, Chowdary P, et al. Sexual assault as a risk factor for gynaecological morbidity: an exploratory systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2020;255:222-230.
30. Hamlin AA, Sheeder J, Muffly TM. Brief versus thong hygiene in obstetrics and gynecology (B-THONG): a survey study. J Obstet Gynaecol Res. 2019;45:1190-1196.
31. Foster DC, Kotok MB, Huang LS, et al. Oral desipramine and topical lidocaine for vulvodynia: a randomized controlled trial. Obstet Gynecol. 2010;116:583-593.
32. Pérez-López FR, Bueno-Notivol J, Hernandez AV, et al. Systematic review and meta-analysis of the effects of treatment modalities for vestibulodynia in women. Eur J Contracept Reprod Health Care. 2019;24:337-346.
33. Cheong YC, Smotra G, Williams AC. Non-surgical interventions for the management of chronic pelvic pain. Cochrane Database Syst Rev. 2014;(3):CD008797.
34. Goldstein AT, Pukall CF, Brown C, et al. Vulvodynia: assessment and treatment. J Sex Med. 2016;13:572-590.
35. Bergeron S, Binik YM, Khalifé S, et al. A randomized comparison of group cognitive-behavioral therapy, surface electromyographic biofeedback, and vestibulectomy in the treatment of dyspareunia resulting from vulvar vestibulitis. Pain. 2001;91:297-306.
36. Schvartzman R, Schvartzman L, Ferreira CF, et al. Physical therapy intervention for women with dyspareunia: a randomized clinical trial. J Sex Marital Ther. 2019;45:378-394.
37. Morin M, Dumoulin C, Bergeron S, et al. Multimodal physical therapy versus topical lidocaine for provoked vestibulodynia: a multicenter, randomized trial. Am J Obstet Gynecol. 2021;224:189.e1-189.e12.
38. Bergeron S, Khalifé S, Dupuis M-J, et al. A randomized clinical trial comparing group cognitive-behavioral therapy and a topical steroid for women with dyspareunia. J Consult Clin Psychol. 2016;84:259-268.
39. Maseroli E, Scavello I, Rastrelli G, et al. Outcome of medical and psychosexual interventions for vaginismus: a systematic review and meta-analysis. J Sex Med. 2018;15:1752-1764.
40. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. 2017. Accessed February 3, 2022. www.apa.org/ptsd-guideline/ptsd.pdf
PRACTICE RECOMMENDATIONS
› Screen all patients for sexual dysfunctions, as patients often do not report symptoms on their own. B
› Refer patients with dyspareunia for psychotherapy to address both pain and psychosocial causes and sequela of dyspareunia. A
› Refer patients with dyspareunia for pelvic floor physical therapy to address pain and sexual functioning. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Former physician sentenced to 20 years in pill mill case
A former pain medicine physician received a sentence of 20 years in prison for selling opioids and writing prescriptions for patients who were abusing or diverting the medications.
Patrick Titus, 58, operated Lighthouse Internal Medicine in Milford, Delaware, from 2005-2014.
Federal prosecutors said Mr. Titus unlawfully distributed or dispensed opioids including fentanyl, morphine, methadone, OxyContin, and oxycodone outside the scope of practice and often prescribed them in combination with each other or in other dangerous combinations. Mr. Titus distributed over 1 million pills, said the government.
In a 2018 indictment, the government said that Mr. Titus would, “at the first and nearly every follow-up visit” prescribe opioids in high dosages, often without conducting an exam or reviewing any urine test results. He would also write prescriptions for opioids without getting patients’s prior medical records or reviewing test results and rarely referred patients to alternative pain treatments such as physical therapy, psychotherapy, or massage.
According to the indictment, he ignored “red flags,” including that patients would come from long distances, sometimes from out of state, and would pay cash, despite having Medicaid coverage.
“Today’s sentencing makes clear that medical professionals who recklessly prescribe opioids and endanger the safety and health of patients will be held accountable,” said Anne Milgram, a Drug Enforcement Administration administrator.
“This sentence is a reminder that the Department of Justice will hold accountable those doctors who are illegitimately prescribing opioids and fueling the country’s opioid crisis,” said Assistant Attorney General Kenneth A. Polite Jr., of the Justice Department’s Criminal Division, in the same statement. “Doctors who commit these unlawful acts exploit their roles as stewards of their patients’s care for their own profit,” he added.
The sentence follows Mr. Titus’s 2-week jury trial in 2021, when he was convicted of 13 counts of unlawful distribution and dispensing of controlled substances and one count of maintaining his practice primarily as a location to sell drugs. Mr. Titus faced a maximum of 20 years per count.
At the time of his conviction, Mr. Titus’s attorney said he planned to appeal, according to Delaware Online.
Delaware suspended Mr. Titus’s registration to prescribe controlled substances for 1 year in 2011. At the time, the state said it had determined that his continued prescribing “poses [an] imminent danger to the public health or safety.”
The state found that from January to November 2011, Mr. Titus issued 3,941 prescriptions for almost 750,000 pills for 17 different controlled substances, all sent to a single pharmacy.
The state also alleged that he wrote prescriptions for controlled substances to patients with felony convictions for drug trafficking and to at least one patient who his staff told him was selling the opioid that Mr. Titus had prescribed. It later determined that Mr. Titus continued prescribing even after it had suspended his DEA registration.
According to a 2014 consent agreement, the state subsequently ordered another 1-year suspension of his DEA registration, to be followed by a 3-year probation period.
Meanwhile, the same year, the state Board of Medical Licensure put Mr. Titus’s medical license on probation for 2 years and ordered him to complete 15 continuing medical education credits in medical recordkeeping, ethics, how to detect diversion and abuse, and in some other areas, and to pay a $7,500 fine.
In 2016, the medical board revoked Mr. Titus’s license, after finding that he continued to prescribe pain medications to patients he did not screen or monitor and for a multitude of other infractions.
A version of this article first appeared on Medscape.com.
A former pain medicine physician received a sentence of 20 years in prison for selling opioids and writing prescriptions for patients who were abusing or diverting the medications.
Patrick Titus, 58, operated Lighthouse Internal Medicine in Milford, Delaware, from 2005-2014.
Federal prosecutors said Mr. Titus unlawfully distributed or dispensed opioids including fentanyl, morphine, methadone, OxyContin, and oxycodone outside the scope of practice and often prescribed them in combination with each other or in other dangerous combinations. Mr. Titus distributed over 1 million pills, said the government.
In a 2018 indictment, the government said that Mr. Titus would, “at the first and nearly every follow-up visit” prescribe opioids in high dosages, often without conducting an exam or reviewing any urine test results. He would also write prescriptions for opioids without getting patients’s prior medical records or reviewing test results and rarely referred patients to alternative pain treatments such as physical therapy, psychotherapy, or massage.
According to the indictment, he ignored “red flags,” including that patients would come from long distances, sometimes from out of state, and would pay cash, despite having Medicaid coverage.
“Today’s sentencing makes clear that medical professionals who recklessly prescribe opioids and endanger the safety and health of patients will be held accountable,” said Anne Milgram, a Drug Enforcement Administration administrator.
“This sentence is a reminder that the Department of Justice will hold accountable those doctors who are illegitimately prescribing opioids and fueling the country’s opioid crisis,” said Assistant Attorney General Kenneth A. Polite Jr., of the Justice Department’s Criminal Division, in the same statement. “Doctors who commit these unlawful acts exploit their roles as stewards of their patients’s care for their own profit,” he added.
The sentence follows Mr. Titus’s 2-week jury trial in 2021, when he was convicted of 13 counts of unlawful distribution and dispensing of controlled substances and one count of maintaining his practice primarily as a location to sell drugs. Mr. Titus faced a maximum of 20 years per count.
At the time of his conviction, Mr. Titus’s attorney said he planned to appeal, according to Delaware Online.
Delaware suspended Mr. Titus’s registration to prescribe controlled substances for 1 year in 2011. At the time, the state said it had determined that his continued prescribing “poses [an] imminent danger to the public health or safety.”
The state found that from January to November 2011, Mr. Titus issued 3,941 prescriptions for almost 750,000 pills for 17 different controlled substances, all sent to a single pharmacy.
The state also alleged that he wrote prescriptions for controlled substances to patients with felony convictions for drug trafficking and to at least one patient who his staff told him was selling the opioid that Mr. Titus had prescribed. It later determined that Mr. Titus continued prescribing even after it had suspended his DEA registration.
According to a 2014 consent agreement, the state subsequently ordered another 1-year suspension of his DEA registration, to be followed by a 3-year probation period.
Meanwhile, the same year, the state Board of Medical Licensure put Mr. Titus’s medical license on probation for 2 years and ordered him to complete 15 continuing medical education credits in medical recordkeeping, ethics, how to detect diversion and abuse, and in some other areas, and to pay a $7,500 fine.
In 2016, the medical board revoked Mr. Titus’s license, after finding that he continued to prescribe pain medications to patients he did not screen or monitor and for a multitude of other infractions.
A version of this article first appeared on Medscape.com.
A former pain medicine physician received a sentence of 20 years in prison for selling opioids and writing prescriptions for patients who were abusing or diverting the medications.
Patrick Titus, 58, operated Lighthouse Internal Medicine in Milford, Delaware, from 2005-2014.
Federal prosecutors said Mr. Titus unlawfully distributed or dispensed opioids including fentanyl, morphine, methadone, OxyContin, and oxycodone outside the scope of practice and often prescribed them in combination with each other or in other dangerous combinations. Mr. Titus distributed over 1 million pills, said the government.
In a 2018 indictment, the government said that Mr. Titus would, “at the first and nearly every follow-up visit” prescribe opioids in high dosages, often without conducting an exam or reviewing any urine test results. He would also write prescriptions for opioids without getting patients’s prior medical records or reviewing test results and rarely referred patients to alternative pain treatments such as physical therapy, psychotherapy, or massage.
According to the indictment, he ignored “red flags,” including that patients would come from long distances, sometimes from out of state, and would pay cash, despite having Medicaid coverage.
“Today’s sentencing makes clear that medical professionals who recklessly prescribe opioids and endanger the safety and health of patients will be held accountable,” said Anne Milgram, a Drug Enforcement Administration administrator.
“This sentence is a reminder that the Department of Justice will hold accountable those doctors who are illegitimately prescribing opioids and fueling the country’s opioid crisis,” said Assistant Attorney General Kenneth A. Polite Jr., of the Justice Department’s Criminal Division, in the same statement. “Doctors who commit these unlawful acts exploit their roles as stewards of their patients’s care for their own profit,” he added.
The sentence follows Mr. Titus’s 2-week jury trial in 2021, when he was convicted of 13 counts of unlawful distribution and dispensing of controlled substances and one count of maintaining his practice primarily as a location to sell drugs. Mr. Titus faced a maximum of 20 years per count.
At the time of his conviction, Mr. Titus’s attorney said he planned to appeal, according to Delaware Online.
Delaware suspended Mr. Titus’s registration to prescribe controlled substances for 1 year in 2011. At the time, the state said it had determined that his continued prescribing “poses [an] imminent danger to the public health or safety.”
The state found that from January to November 2011, Mr. Titus issued 3,941 prescriptions for almost 750,000 pills for 17 different controlled substances, all sent to a single pharmacy.
The state also alleged that he wrote prescriptions for controlled substances to patients with felony convictions for drug trafficking and to at least one patient who his staff told him was selling the opioid that Mr. Titus had prescribed. It later determined that Mr. Titus continued prescribing even after it had suspended his DEA registration.
According to a 2014 consent agreement, the state subsequently ordered another 1-year suspension of his DEA registration, to be followed by a 3-year probation period.
Meanwhile, the same year, the state Board of Medical Licensure put Mr. Titus’s medical license on probation for 2 years and ordered him to complete 15 continuing medical education credits in medical recordkeeping, ethics, how to detect diversion and abuse, and in some other areas, and to pay a $7,500 fine.
In 2016, the medical board revoked Mr. Titus’s license, after finding that he continued to prescribe pain medications to patients he did not screen or monitor and for a multitude of other infractions.
A version of this article first appeared on Medscape.com.
Mindfulness intervention curbs opioid misuse, chronic pain
In a randomized clinical trial, 250 adults with both opioid misuse and chronic pain received either the intervention, called mindfulness-oriented recovery enhancement (MORE), or supportive psychotherapy.
Results showed the first group was twice as likely to reduce opioid misuse after 9 months than the latter group.
The intervention was developed by Eric Garland, PhD, director of the Center on Mindfulness and Integrative Health Intervention Development (C-MIIND), University of Utah, Salt Lake City. “As the largest and longest-term clinical trial of MORE ever conducted, this study definitively establishes the efficacy of MORE as a treatment for chronic pain and opioid misuse,” he told this news organization.
The findings were published online Feb. 28 in JAMA Internal Medicine.
Self-regulation
Study participants included 250 adults (64% women; mean age, 51.8 years) with co-occurring opioid misuse and chronic pain who were randomly allocated to receive MORE or supportive psychotherapy, which served as a control group.
Both interventions were delivered by trained clinical social workers in six primary care clinics in Utah to groups of 6-12 participants across 8 weekly 2-hour sessions.
The MORE intervention, detailed on Dr. Garland’s website, provides sequenced training in mindfulness, reappraisal, and savoring skills.
Mindfulness consisted of meditation on breathing and body sensations to strengthen self-regulation of compulsive opioid use and to mitigate pain and opioid craving by reinterpreting these experiences as innocuous sensory information.
Reappraisal consisted of reframing maladaptive thoughts to decrease negative emotions and engender meaning in life.
Savoring consisted of training in focusing awareness on pleasurable events and sensations to amplify positive emotions and reward.
Fewer depressive symptoms
Through 9 months of follow-up, the MORE group had about a twofold greater likelihood than the supportive psychotherapy group for reduction in opioid misuse (odds ratio [OR], 2.06; 95% confidence interval, 1.17-3.61; P = .01)
“MORE reduced opioid misuse by 45% 9 months after the end of treatment, more than doubling the effect of standard supportive psychotherapy and exceeding the effect size of other therapies for opioid misuse among people with chronic pain,” Dr. Garland said.
Members of the MORE group experienced greater reduction in pain severity and pain-related functional interference compared with members of the control group.
“MORE’s effect size on chronic pain symptoms was greater than that observed for CBT, the current gold standard psychological treatment for chronic pain,” Dr. Garland noted.
Compared with supportive psychotherapy, MORE decreased emotional distress, depressive symptoms, and real-time reports of opioid craving in daily life.
“Although nearly 70% of participants met criteria for depression at the beginning of the trial, on average, patients in MORE no longer exhibited symptoms consistent with major depressive disorder by the end of the study,” Dr. Garland said.
The current study builds on prior studies of MORE showing similar results, as reported previously by this news organization.
MORE can be successfully delivered in routine primary care, Dr. Garland noted. “In this trial, we delivered MORE in conference rooms, break rooms, and lunch rooms at community primary care clinics,” he added.
‘Powerful program’
To date, Dr. Garland has trained more than 450 physicians, nurses, social workers, and psychologists in health care systems across the country to implement MORE as an insurance-reimbursable group visit for patients in need.
One of them is Nancy Sudak, MD, chief well-being officer and director of integrative health, Essentia Health, Duluth, Minn.
“MORE is a very powerful program that teaches patients how to turn down the volume of their pain. I’ve been quite impressed by the power of MORE,” Dr. Sudak told this news organization
She noted that “buy-in” from patients is key – and the more a clinician knows a patient, the easier the buy-in.
“I recruited most of the patients in my groups from my own practice, so I already knew the patients quite well and there wasn’t really a need to sell it,” Dr. Sudak said.
“We have tried to operationalize it through our system and find that, as long as our recruitment techniques are robust enough, it’s not that hard to find patients to fill the groups, especially because chronic pain is just so common,” she added.
Dr. Sudak has found that patients who participate in MORE “bond and learn with each other and support each other. Patients love it, providers love it, and it’s a way to address isolation and loneliness” that can come with certain conditions.
“There are really only upsides to the group visit model and I think we’ll be seeing quite a bit more of it in the future,” she added.
Evidence-based data
Anna Parisi, PhD, is also delivering MORE to patients. She told this news organization, she was “really drawn” to the MORE program because oftentimes patients who require the most sophisticated therapies receive the ones with the least evidence.
This is often “what folks in the community are getting when they’re struggling with substance use,” added Dr. Parisi, a postdoctoral research associate working with Dr. Garland at the University of Utah. Dr. Parisi was not a coauthor on the current study.
“With MORE, all of the strategies and techniques are tied to mechanistic studies of their efficacy, so you know that what you’re delivering has a rationale behind it,” she said.
Like Dr. Sudak, Dr. Parisi said her patients, for the most part, have been receptive to the program. Although at first some were skeptical about mindfulness – with one patient using the term “tree-hugging” – they found immediate benefit even after the first session.
“That really helps them stay motivated to finish the program,” Dr. Parisi said.
This work was supported by a grant from the National Institute on Drug Abuse. Dr. Garland serves as director of the Center on Mindfulness and Integrative Health Intervention Development, which provides MORE, mindfulness-based therapy, and CBT in the context of research trials for no cost to research participants. He receives honoraria and payment for delivering seminars, lectures, and teaching engagements related to training clinicians in MORE and mindfulness and receives royalties from BehaVR and from the sales of books related to MORE outside the submitted work. Dr. Sudak and Dr. Parisi have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a randomized clinical trial, 250 adults with both opioid misuse and chronic pain received either the intervention, called mindfulness-oriented recovery enhancement (MORE), or supportive psychotherapy.
Results showed the first group was twice as likely to reduce opioid misuse after 9 months than the latter group.
The intervention was developed by Eric Garland, PhD, director of the Center on Mindfulness and Integrative Health Intervention Development (C-MIIND), University of Utah, Salt Lake City. “As the largest and longest-term clinical trial of MORE ever conducted, this study definitively establishes the efficacy of MORE as a treatment for chronic pain and opioid misuse,” he told this news organization.
The findings were published online Feb. 28 in JAMA Internal Medicine.
Self-regulation
Study participants included 250 adults (64% women; mean age, 51.8 years) with co-occurring opioid misuse and chronic pain who were randomly allocated to receive MORE or supportive psychotherapy, which served as a control group.
Both interventions were delivered by trained clinical social workers in six primary care clinics in Utah to groups of 6-12 participants across 8 weekly 2-hour sessions.
The MORE intervention, detailed on Dr. Garland’s website, provides sequenced training in mindfulness, reappraisal, and savoring skills.
Mindfulness consisted of meditation on breathing and body sensations to strengthen self-regulation of compulsive opioid use and to mitigate pain and opioid craving by reinterpreting these experiences as innocuous sensory information.
Reappraisal consisted of reframing maladaptive thoughts to decrease negative emotions and engender meaning in life.
Savoring consisted of training in focusing awareness on pleasurable events and sensations to amplify positive emotions and reward.
Fewer depressive symptoms
Through 9 months of follow-up, the MORE group had about a twofold greater likelihood than the supportive psychotherapy group for reduction in opioid misuse (odds ratio [OR], 2.06; 95% confidence interval, 1.17-3.61; P = .01)
“MORE reduced opioid misuse by 45% 9 months after the end of treatment, more than doubling the effect of standard supportive psychotherapy and exceeding the effect size of other therapies for opioid misuse among people with chronic pain,” Dr. Garland said.
Members of the MORE group experienced greater reduction in pain severity and pain-related functional interference compared with members of the control group.
“MORE’s effect size on chronic pain symptoms was greater than that observed for CBT, the current gold standard psychological treatment for chronic pain,” Dr. Garland noted.
Compared with supportive psychotherapy, MORE decreased emotional distress, depressive symptoms, and real-time reports of opioid craving in daily life.
“Although nearly 70% of participants met criteria for depression at the beginning of the trial, on average, patients in MORE no longer exhibited symptoms consistent with major depressive disorder by the end of the study,” Dr. Garland said.
The current study builds on prior studies of MORE showing similar results, as reported previously by this news organization.
MORE can be successfully delivered in routine primary care, Dr. Garland noted. “In this trial, we delivered MORE in conference rooms, break rooms, and lunch rooms at community primary care clinics,” he added.
‘Powerful program’
To date, Dr. Garland has trained more than 450 physicians, nurses, social workers, and psychologists in health care systems across the country to implement MORE as an insurance-reimbursable group visit for patients in need.
One of them is Nancy Sudak, MD, chief well-being officer and director of integrative health, Essentia Health, Duluth, Minn.
“MORE is a very powerful program that teaches patients how to turn down the volume of their pain. I’ve been quite impressed by the power of MORE,” Dr. Sudak told this news organization
She noted that “buy-in” from patients is key – and the more a clinician knows a patient, the easier the buy-in.
“I recruited most of the patients in my groups from my own practice, so I already knew the patients quite well and there wasn’t really a need to sell it,” Dr. Sudak said.
“We have tried to operationalize it through our system and find that, as long as our recruitment techniques are robust enough, it’s not that hard to find patients to fill the groups, especially because chronic pain is just so common,” she added.
Dr. Sudak has found that patients who participate in MORE “bond and learn with each other and support each other. Patients love it, providers love it, and it’s a way to address isolation and loneliness” that can come with certain conditions.
“There are really only upsides to the group visit model and I think we’ll be seeing quite a bit more of it in the future,” she added.
Evidence-based data
Anna Parisi, PhD, is also delivering MORE to patients. She told this news organization, she was “really drawn” to the MORE program because oftentimes patients who require the most sophisticated therapies receive the ones with the least evidence.
This is often “what folks in the community are getting when they’re struggling with substance use,” added Dr. Parisi, a postdoctoral research associate working with Dr. Garland at the University of Utah. Dr. Parisi was not a coauthor on the current study.
“With MORE, all of the strategies and techniques are tied to mechanistic studies of their efficacy, so you know that what you’re delivering has a rationale behind it,” she said.
Like Dr. Sudak, Dr. Parisi said her patients, for the most part, have been receptive to the program. Although at first some were skeptical about mindfulness – with one patient using the term “tree-hugging” – they found immediate benefit even after the first session.
“That really helps them stay motivated to finish the program,” Dr. Parisi said.
This work was supported by a grant from the National Institute on Drug Abuse. Dr. Garland serves as director of the Center on Mindfulness and Integrative Health Intervention Development, which provides MORE, mindfulness-based therapy, and CBT in the context of research trials for no cost to research participants. He receives honoraria and payment for delivering seminars, lectures, and teaching engagements related to training clinicians in MORE and mindfulness and receives royalties from BehaVR and from the sales of books related to MORE outside the submitted work. Dr. Sudak and Dr. Parisi have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In a randomized clinical trial, 250 adults with both opioid misuse and chronic pain received either the intervention, called mindfulness-oriented recovery enhancement (MORE), or supportive psychotherapy.
Results showed the first group was twice as likely to reduce opioid misuse after 9 months than the latter group.
The intervention was developed by Eric Garland, PhD, director of the Center on Mindfulness and Integrative Health Intervention Development (C-MIIND), University of Utah, Salt Lake City. “As the largest and longest-term clinical trial of MORE ever conducted, this study definitively establishes the efficacy of MORE as a treatment for chronic pain and opioid misuse,” he told this news organization.
The findings were published online Feb. 28 in JAMA Internal Medicine.
Self-regulation
Study participants included 250 adults (64% women; mean age, 51.8 years) with co-occurring opioid misuse and chronic pain who were randomly allocated to receive MORE or supportive psychotherapy, which served as a control group.
Both interventions were delivered by trained clinical social workers in six primary care clinics in Utah to groups of 6-12 participants across 8 weekly 2-hour sessions.
The MORE intervention, detailed on Dr. Garland’s website, provides sequenced training in mindfulness, reappraisal, and savoring skills.
Mindfulness consisted of meditation on breathing and body sensations to strengthen self-regulation of compulsive opioid use and to mitigate pain and opioid craving by reinterpreting these experiences as innocuous sensory information.
Reappraisal consisted of reframing maladaptive thoughts to decrease negative emotions and engender meaning in life.
Savoring consisted of training in focusing awareness on pleasurable events and sensations to amplify positive emotions and reward.
Fewer depressive symptoms
Through 9 months of follow-up, the MORE group had about a twofold greater likelihood than the supportive psychotherapy group for reduction in opioid misuse (odds ratio [OR], 2.06; 95% confidence interval, 1.17-3.61; P = .01)
“MORE reduced opioid misuse by 45% 9 months after the end of treatment, more than doubling the effect of standard supportive psychotherapy and exceeding the effect size of other therapies for opioid misuse among people with chronic pain,” Dr. Garland said.
Members of the MORE group experienced greater reduction in pain severity and pain-related functional interference compared with members of the control group.
“MORE’s effect size on chronic pain symptoms was greater than that observed for CBT, the current gold standard psychological treatment for chronic pain,” Dr. Garland noted.
Compared with supportive psychotherapy, MORE decreased emotional distress, depressive symptoms, and real-time reports of opioid craving in daily life.
“Although nearly 70% of participants met criteria for depression at the beginning of the trial, on average, patients in MORE no longer exhibited symptoms consistent with major depressive disorder by the end of the study,” Dr. Garland said.
The current study builds on prior studies of MORE showing similar results, as reported previously by this news organization.
MORE can be successfully delivered in routine primary care, Dr. Garland noted. “In this trial, we delivered MORE in conference rooms, break rooms, and lunch rooms at community primary care clinics,” he added.
‘Powerful program’
To date, Dr. Garland has trained more than 450 physicians, nurses, social workers, and psychologists in health care systems across the country to implement MORE as an insurance-reimbursable group visit for patients in need.
One of them is Nancy Sudak, MD, chief well-being officer and director of integrative health, Essentia Health, Duluth, Minn.
“MORE is a very powerful program that teaches patients how to turn down the volume of their pain. I’ve been quite impressed by the power of MORE,” Dr. Sudak told this news organization
She noted that “buy-in” from patients is key – and the more a clinician knows a patient, the easier the buy-in.
“I recruited most of the patients in my groups from my own practice, so I already knew the patients quite well and there wasn’t really a need to sell it,” Dr. Sudak said.
“We have tried to operationalize it through our system and find that, as long as our recruitment techniques are robust enough, it’s not that hard to find patients to fill the groups, especially because chronic pain is just so common,” she added.
Dr. Sudak has found that patients who participate in MORE “bond and learn with each other and support each other. Patients love it, providers love it, and it’s a way to address isolation and loneliness” that can come with certain conditions.
“There are really only upsides to the group visit model and I think we’ll be seeing quite a bit more of it in the future,” she added.
Evidence-based data
Anna Parisi, PhD, is also delivering MORE to patients. She told this news organization, she was “really drawn” to the MORE program because oftentimes patients who require the most sophisticated therapies receive the ones with the least evidence.
This is often “what folks in the community are getting when they’re struggling with substance use,” added Dr. Parisi, a postdoctoral research associate working with Dr. Garland at the University of Utah. Dr. Parisi was not a coauthor on the current study.
“With MORE, all of the strategies and techniques are tied to mechanistic studies of their efficacy, so you know that what you’re delivering has a rationale behind it,” she said.
Like Dr. Sudak, Dr. Parisi said her patients, for the most part, have been receptive to the program. Although at first some were skeptical about mindfulness – with one patient using the term “tree-hugging” – they found immediate benefit even after the first session.
“That really helps them stay motivated to finish the program,” Dr. Parisi said.
This work was supported by a grant from the National Institute on Drug Abuse. Dr. Garland serves as director of the Center on Mindfulness and Integrative Health Intervention Development, which provides MORE, mindfulness-based therapy, and CBT in the context of research trials for no cost to research participants. He receives honoraria and payment for delivering seminars, lectures, and teaching engagements related to training clinicians in MORE and mindfulness and receives royalties from BehaVR and from the sales of books related to MORE outside the submitted work. Dr. Sudak and Dr. Parisi have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Practice guidelines highlights from past year
Based on the most recent Infectious Diseases Society of America (IDSA) guidelines, what would be the preferred therapy?
A) Metronidazole
B) Fidaxomicin + bezlotoxumab
C) Vancomycin
D) Fecal microbiota transplant
The recommendations from the 2021 guidelines would be to treat with fidaxomicin and add bezlotoxumab.1 The guidelines highlight the following changes:
- In patients with an initial Clostridioides difficile infections (CDI) fidaxomicin is preferred over vancomycin.
- In patients with a recurrent CDI episode, fidaxomicin is favored over vancomycin. For patients with multiple recurrences, vancomycin in a tapered and pulsed regimen, vancomycin followed by rifaximin, and fecal microbiota transplantation are options in addition to fidaxomicin.
- Addition of bezlotoxumab to standard of care antibiotics is recommended for recurrence of CDI within the first 6 months over standard of care antibiotics alone
The feasibility of these recommendations is up for debate. The cost of a course of fidaxomicin is $2,800, and the cost of bezlotoxumab is about $4,500. Cost effectiveness studies that helped drive the recommendations show a savings by reducing future hospitalizations for C. diff.2 Unfortunately, this enthusiasm is not shared by many insurance companies for outpatient treatment.
Knee osteoarthritis
I will save you the excitement of the new acromegaly guidelines and focus on something we see all the time: knee osteoarthritis. The American Academy of Orthopedic Surgeons has released guidelines for this condition.3 The useful points I found were as follows:
- Topical application of nonsteroidal anti-inflammatory drugs (e.g., diclofenac) should be used to improve function and quality of life in patients with knee osteoarthritis.
- Exercise routines (i.e, supervised, unsupervised, and/or aquatic) are recommended versus no exercise for improving pain and function in patients with knee osteoarthritis.
- Not recommended is the use of oral narcotics (including tramadol), as they are not effective at improving pain or function, and their use results in a significant increased risk of adverse events.
- Not recommended for routine use in symptomatic knee osteoarthritis is intra-articular injection of hyaluronic acid.
I was happy to see topical NSAIDS recommended, as they are a much safer option in older patients than oral NSAIDS (which were also recommended). The recommendation against narcotics, including tramadol, is a shift from the recommendation of tramadol in the 2013 guidelines.4 Acetaminophen was enthusiastically recommended, and is still worth a try.
Sexually transmitted infections
- The dosing for the treatment of gonorrhea has increased to 500 mg of ceftriaxone (was 250 mg in 2015 guidelines), with a dose of 1 gram for patients who weigh more than 150 kg.
- Chlamydia infections should be treated with a 7-day course of doxycycline as the preferred antibiotic, except in pregnant women (where azithromycin is recommended).
- Herpes simplex virus 2 recurrences can be treated with twice-daily dosing of 800 mg of acyclovir for 5 days, or acyclovir 800 mg three times a day for 2 days. The shortest course for recurrence is famciclovir 1 gram twice a day for 1 day.
- The Centers for Disease Control and Prevention has removed the recommendation for avoidance of alcohol when taking metronidazole.
I hope these highlights of guidelines for common issues we see are helpful!
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Johnson S et al. Clinical practice guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused update guidelines on management of Clostridioides difficile Infection in adults. Clin Infect Dis. 2021 Sep 7;73(5):e1029-e1044.
2. Pabhu VS et al. Cost-effectiveness of bezlotoxumab compared with placebo for the prevention of recurrent Clostridium difficile infection. Clin Infect Dis. 2018 Feb 1;66(3):355-62.
3. American Academy of Orthopaedic Surgeons: Management of osteoarthritis of the knee (non-arthroplasty) – Evidence-based clinical practice guideline (2021 Aug 31. https://www.aaos.org/oak3cpg).
4. Jevsevar DS. Treatment of osteoarthritis of the knee: Evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013: Sep;21(9):571-6.
5. Sexually transmitted infections treatment guidelines, 2021 recommendations and reports. MMWR 2021 Jul 23;70(4):1-187.
Based on the most recent Infectious Diseases Society of America (IDSA) guidelines, what would be the preferred therapy?
A) Metronidazole
B) Fidaxomicin + bezlotoxumab
C) Vancomycin
D) Fecal microbiota transplant
The recommendations from the 2021 guidelines would be to treat with fidaxomicin and add bezlotoxumab.1 The guidelines highlight the following changes:
- In patients with an initial Clostridioides difficile infections (CDI) fidaxomicin is preferred over vancomycin.
- In patients with a recurrent CDI episode, fidaxomicin is favored over vancomycin. For patients with multiple recurrences, vancomycin in a tapered and pulsed regimen, vancomycin followed by rifaximin, and fecal microbiota transplantation are options in addition to fidaxomicin.
- Addition of bezlotoxumab to standard of care antibiotics is recommended for recurrence of CDI within the first 6 months over standard of care antibiotics alone
The feasibility of these recommendations is up for debate. The cost of a course of fidaxomicin is $2,800, and the cost of bezlotoxumab is about $4,500. Cost effectiveness studies that helped drive the recommendations show a savings by reducing future hospitalizations for C. diff.2 Unfortunately, this enthusiasm is not shared by many insurance companies for outpatient treatment.
Knee osteoarthritis
I will save you the excitement of the new acromegaly guidelines and focus on something we see all the time: knee osteoarthritis. The American Academy of Orthopedic Surgeons has released guidelines for this condition.3 The useful points I found were as follows:
- Topical application of nonsteroidal anti-inflammatory drugs (e.g., diclofenac) should be used to improve function and quality of life in patients with knee osteoarthritis.
- Exercise routines (i.e, supervised, unsupervised, and/or aquatic) are recommended versus no exercise for improving pain and function in patients with knee osteoarthritis.
- Not recommended is the use of oral narcotics (including tramadol), as they are not effective at improving pain or function, and their use results in a significant increased risk of adverse events.
- Not recommended for routine use in symptomatic knee osteoarthritis is intra-articular injection of hyaluronic acid.
I was happy to see topical NSAIDS recommended, as they are a much safer option in older patients than oral NSAIDS (which were also recommended). The recommendation against narcotics, including tramadol, is a shift from the recommendation of tramadol in the 2013 guidelines.4 Acetaminophen was enthusiastically recommended, and is still worth a try.
Sexually transmitted infections
- The dosing for the treatment of gonorrhea has increased to 500 mg of ceftriaxone (was 250 mg in 2015 guidelines), with a dose of 1 gram for patients who weigh more than 150 kg.
- Chlamydia infections should be treated with a 7-day course of doxycycline as the preferred antibiotic, except in pregnant women (where azithromycin is recommended).
- Herpes simplex virus 2 recurrences can be treated with twice-daily dosing of 800 mg of acyclovir for 5 days, or acyclovir 800 mg three times a day for 2 days. The shortest course for recurrence is famciclovir 1 gram twice a day for 1 day.
- The Centers for Disease Control and Prevention has removed the recommendation for avoidance of alcohol when taking metronidazole.
I hope these highlights of guidelines for common issues we see are helpful!
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Johnson S et al. Clinical practice guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused update guidelines on management of Clostridioides difficile Infection in adults. Clin Infect Dis. 2021 Sep 7;73(5):e1029-e1044.
2. Pabhu VS et al. Cost-effectiveness of bezlotoxumab compared with placebo for the prevention of recurrent Clostridium difficile infection. Clin Infect Dis. 2018 Feb 1;66(3):355-62.
3. American Academy of Orthopaedic Surgeons: Management of osteoarthritis of the knee (non-arthroplasty) – Evidence-based clinical practice guideline (2021 Aug 31. https://www.aaos.org/oak3cpg).
4. Jevsevar DS. Treatment of osteoarthritis of the knee: Evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013: Sep;21(9):571-6.
5. Sexually transmitted infections treatment guidelines, 2021 recommendations and reports. MMWR 2021 Jul 23;70(4):1-187.
Based on the most recent Infectious Diseases Society of America (IDSA) guidelines, what would be the preferred therapy?
A) Metronidazole
B) Fidaxomicin + bezlotoxumab
C) Vancomycin
D) Fecal microbiota transplant
The recommendations from the 2021 guidelines would be to treat with fidaxomicin and add bezlotoxumab.1 The guidelines highlight the following changes:
- In patients with an initial Clostridioides difficile infections (CDI) fidaxomicin is preferred over vancomycin.
- In patients with a recurrent CDI episode, fidaxomicin is favored over vancomycin. For patients with multiple recurrences, vancomycin in a tapered and pulsed regimen, vancomycin followed by rifaximin, and fecal microbiota transplantation are options in addition to fidaxomicin.
- Addition of bezlotoxumab to standard of care antibiotics is recommended for recurrence of CDI within the first 6 months over standard of care antibiotics alone
The feasibility of these recommendations is up for debate. The cost of a course of fidaxomicin is $2,800, and the cost of bezlotoxumab is about $4,500. Cost effectiveness studies that helped drive the recommendations show a savings by reducing future hospitalizations for C. diff.2 Unfortunately, this enthusiasm is not shared by many insurance companies for outpatient treatment.
Knee osteoarthritis
I will save you the excitement of the new acromegaly guidelines and focus on something we see all the time: knee osteoarthritis. The American Academy of Orthopedic Surgeons has released guidelines for this condition.3 The useful points I found were as follows:
- Topical application of nonsteroidal anti-inflammatory drugs (e.g., diclofenac) should be used to improve function and quality of life in patients with knee osteoarthritis.
- Exercise routines (i.e, supervised, unsupervised, and/or aquatic) are recommended versus no exercise for improving pain and function in patients with knee osteoarthritis.
- Not recommended is the use of oral narcotics (including tramadol), as they are not effective at improving pain or function, and their use results in a significant increased risk of adverse events.
- Not recommended for routine use in symptomatic knee osteoarthritis is intra-articular injection of hyaluronic acid.
I was happy to see topical NSAIDS recommended, as they are a much safer option in older patients than oral NSAIDS (which were also recommended). The recommendation against narcotics, including tramadol, is a shift from the recommendation of tramadol in the 2013 guidelines.4 Acetaminophen was enthusiastically recommended, and is still worth a try.
Sexually transmitted infections
- The dosing for the treatment of gonorrhea has increased to 500 mg of ceftriaxone (was 250 mg in 2015 guidelines), with a dose of 1 gram for patients who weigh more than 150 kg.
- Chlamydia infections should be treated with a 7-day course of doxycycline as the preferred antibiotic, except in pregnant women (where azithromycin is recommended).
- Herpes simplex virus 2 recurrences can be treated with twice-daily dosing of 800 mg of acyclovir for 5 days, or acyclovir 800 mg three times a day for 2 days. The shortest course for recurrence is famciclovir 1 gram twice a day for 1 day.
- The Centers for Disease Control and Prevention has removed the recommendation for avoidance of alcohol when taking metronidazole.
I hope these highlights of guidelines for common issues we see are helpful!
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Johnson S et al. Clinical practice guideline by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA): 2021 Focused update guidelines on management of Clostridioides difficile Infection in adults. Clin Infect Dis. 2021 Sep 7;73(5):e1029-e1044.
2. Pabhu VS et al. Cost-effectiveness of bezlotoxumab compared with placebo for the prevention of recurrent Clostridium difficile infection. Clin Infect Dis. 2018 Feb 1;66(3):355-62.
3. American Academy of Orthopaedic Surgeons: Management of osteoarthritis of the knee (non-arthroplasty) – Evidence-based clinical practice guideline (2021 Aug 31. https://www.aaos.org/oak3cpg).
4. Jevsevar DS. Treatment of osteoarthritis of the knee: Evidence-based guideline, 2nd edition. J Am Acad Orthop Surg. 2013: Sep;21(9):571-6.
5. Sexually transmitted infections treatment guidelines, 2021 recommendations and reports. MMWR 2021 Jul 23;70(4):1-187.
Epidural may lower odds of severe maternal birth complications
Use of neuraxial analgesia for vaginal delivery is associated with a 14% decreased risk for severe maternal morbidity, in part from a reduction in postpartum hemorrhage, new research shows.
The findings indicate that increasing the use of epidural or combined spinal-epidural analgesia may improve maternal health outcomes, especially for Hispanic, Black, and uninsured women who are less likely than White women to receive these interventions, according to the researchers, who published their findings online in JAMA Network Open.
About 80% of non-Hispanic White women receive neuraxial analgesia during labor in the United States, compared with 70% of non-Hispanic Black women and 65% of Hispanic women, according to birth certificate data. Among women without health insurance, half receive epidurals.
Programs that inform pregnant women about epidural use, expand Medicaid, and provide in-house obstetric anesthesia teams “may improve patient participation in clinical decision making and access to care,” study author Guohua Li, MD, DrPH, of Columbia University, New York, said in a statement about the research.
Earlier data from France showed that neuraxial analgesia is associated with reduced risk for severe postpartum hemorrhage. To examine the association between labor neuraxial analgesia and severe maternal morbidity in the United States, Dr. Li and colleagues analyzed more than 575,000 vaginal deliveries in New York hospitals between 2010 and 2017; about half (47.4%) of the women received epidurals during labor.
The researchers focused on severe maternal morbidity, including 16 complications, such as heart failure and sepsis, and five procedures, including hysterectomy and ventilation.
They also considered patient characteristics and comorbidities and hospital-related factors to identify patients who were at higher risk for injury or death.
Severe maternal morbidity occurred in 1.3% of the women. Of the 7,712 women with severe morbidity, more than one in three (35.6%) experienced postpartum hemorrhage.
The overall incidence of severe maternal morbidity was 1.3% among women who received an epidural injection and 1.4% among those who did not. In a weighted analysis, the adjusted odds ratio of severe maternal morbidity associated with epidurals was 0.86 (95% confidence interval, 0.82-0.90).
The study is limited by its observational design and does not prove causation, the authors acknowledged.
“Labor neuraxial analgesia may facilitate early evaluation and management of the third stage of labor to avoid escalation of postpartum hemorrhaging into grave complications and death,” study author Jean Guglielminotti, MD, PhD, an anesthesiologist at Columbia University, said in a statement.
Concerning trends
The Department of Health & Human Services has labeled severe maternal morbidity a public health priority. Recent data from the Centers for Disease Control and Prevention show an increase in maternal mortality rates and worsening disparities by race and ethnicity.
According to the CDC, 861 women died of maternal causes in 2020, up from 754 in 2019. The rate of maternal mortality increased from 20.1 to 23.8 deaths per 100,000 live births.
For Black women, however, the maternal mortality rate was far higher: 55.3 deaths per 100,000 live births – nearly triple the figure of 19.1 per 100,000 for White women. Between 2019 and 2020, the mortality rate increased significantly for Black and Hispanic women, but not White mothers.
Researchers affiliated with University of Toronto and the Hospital for Sick Children agreed in an accompanying editorial that more access to neuraxial labor analgesia for vaginal delivery might improve maternal health outcomes and “may be a strategy well worth pursuing in public health policy.”
The intervention is relatively safe and can “alleviate discomfort and distress,” they wrote.
Neuraxial anesthesia in surgical procedures has been shown to decrease the risk for complications like deep vein thrombosis, pulmonary embolus, transfusion requirements, and kidney failure, said editorialists Evelina Pankiv, MD; Alan Yang, MSc; and Kazuyoshi Aoyama, MD, PhD.
Benefits potentially could stem from improving blood flow, mitigating hypercoagulation, or reducing surgical stress response. But there are rare risks to consider as well, including hemorrhage, infection, and neurologic injury, they added.
Guglielminotti disclosed grants from the National Institute on Minority Health and Health Disparities. Dr. Aoyama reported receiving grants from the Perioperative Services Facilitator Grant Program and Hospital for Sick Children. The other authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of neuraxial analgesia for vaginal delivery is associated with a 14% decreased risk for severe maternal morbidity, in part from a reduction in postpartum hemorrhage, new research shows.
The findings indicate that increasing the use of epidural or combined spinal-epidural analgesia may improve maternal health outcomes, especially for Hispanic, Black, and uninsured women who are less likely than White women to receive these interventions, according to the researchers, who published their findings online in JAMA Network Open.
About 80% of non-Hispanic White women receive neuraxial analgesia during labor in the United States, compared with 70% of non-Hispanic Black women and 65% of Hispanic women, according to birth certificate data. Among women without health insurance, half receive epidurals.
Programs that inform pregnant women about epidural use, expand Medicaid, and provide in-house obstetric anesthesia teams “may improve patient participation in clinical decision making and access to care,” study author Guohua Li, MD, DrPH, of Columbia University, New York, said in a statement about the research.
Earlier data from France showed that neuraxial analgesia is associated with reduced risk for severe postpartum hemorrhage. To examine the association between labor neuraxial analgesia and severe maternal morbidity in the United States, Dr. Li and colleagues analyzed more than 575,000 vaginal deliveries in New York hospitals between 2010 and 2017; about half (47.4%) of the women received epidurals during labor.
The researchers focused on severe maternal morbidity, including 16 complications, such as heart failure and sepsis, and five procedures, including hysterectomy and ventilation.
They also considered patient characteristics and comorbidities and hospital-related factors to identify patients who were at higher risk for injury or death.
Severe maternal morbidity occurred in 1.3% of the women. Of the 7,712 women with severe morbidity, more than one in three (35.6%) experienced postpartum hemorrhage.
The overall incidence of severe maternal morbidity was 1.3% among women who received an epidural injection and 1.4% among those who did not. In a weighted analysis, the adjusted odds ratio of severe maternal morbidity associated with epidurals was 0.86 (95% confidence interval, 0.82-0.90).
The study is limited by its observational design and does not prove causation, the authors acknowledged.
“Labor neuraxial analgesia may facilitate early evaluation and management of the third stage of labor to avoid escalation of postpartum hemorrhaging into grave complications and death,” study author Jean Guglielminotti, MD, PhD, an anesthesiologist at Columbia University, said in a statement.
Concerning trends
The Department of Health & Human Services has labeled severe maternal morbidity a public health priority. Recent data from the Centers for Disease Control and Prevention show an increase in maternal mortality rates and worsening disparities by race and ethnicity.
According to the CDC, 861 women died of maternal causes in 2020, up from 754 in 2019. The rate of maternal mortality increased from 20.1 to 23.8 deaths per 100,000 live births.
For Black women, however, the maternal mortality rate was far higher: 55.3 deaths per 100,000 live births – nearly triple the figure of 19.1 per 100,000 for White women. Between 2019 and 2020, the mortality rate increased significantly for Black and Hispanic women, but not White mothers.
Researchers affiliated with University of Toronto and the Hospital for Sick Children agreed in an accompanying editorial that more access to neuraxial labor analgesia for vaginal delivery might improve maternal health outcomes and “may be a strategy well worth pursuing in public health policy.”
The intervention is relatively safe and can “alleviate discomfort and distress,” they wrote.
Neuraxial anesthesia in surgical procedures has been shown to decrease the risk for complications like deep vein thrombosis, pulmonary embolus, transfusion requirements, and kidney failure, said editorialists Evelina Pankiv, MD; Alan Yang, MSc; and Kazuyoshi Aoyama, MD, PhD.
Benefits potentially could stem from improving blood flow, mitigating hypercoagulation, or reducing surgical stress response. But there are rare risks to consider as well, including hemorrhage, infection, and neurologic injury, they added.
Guglielminotti disclosed grants from the National Institute on Minority Health and Health Disparities. Dr. Aoyama reported receiving grants from the Perioperative Services Facilitator Grant Program and Hospital for Sick Children. The other authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of neuraxial analgesia for vaginal delivery is associated with a 14% decreased risk for severe maternal morbidity, in part from a reduction in postpartum hemorrhage, new research shows.
The findings indicate that increasing the use of epidural or combined spinal-epidural analgesia may improve maternal health outcomes, especially for Hispanic, Black, and uninsured women who are less likely than White women to receive these interventions, according to the researchers, who published their findings online in JAMA Network Open.
About 80% of non-Hispanic White women receive neuraxial analgesia during labor in the United States, compared with 70% of non-Hispanic Black women and 65% of Hispanic women, according to birth certificate data. Among women without health insurance, half receive epidurals.
Programs that inform pregnant women about epidural use, expand Medicaid, and provide in-house obstetric anesthesia teams “may improve patient participation in clinical decision making and access to care,” study author Guohua Li, MD, DrPH, of Columbia University, New York, said in a statement about the research.
Earlier data from France showed that neuraxial analgesia is associated with reduced risk for severe postpartum hemorrhage. To examine the association between labor neuraxial analgesia and severe maternal morbidity in the United States, Dr. Li and colleagues analyzed more than 575,000 vaginal deliveries in New York hospitals between 2010 and 2017; about half (47.4%) of the women received epidurals during labor.
The researchers focused on severe maternal morbidity, including 16 complications, such as heart failure and sepsis, and five procedures, including hysterectomy and ventilation.
They also considered patient characteristics and comorbidities and hospital-related factors to identify patients who were at higher risk for injury or death.
Severe maternal morbidity occurred in 1.3% of the women. Of the 7,712 women with severe morbidity, more than one in three (35.6%) experienced postpartum hemorrhage.
The overall incidence of severe maternal morbidity was 1.3% among women who received an epidural injection and 1.4% among those who did not. In a weighted analysis, the adjusted odds ratio of severe maternal morbidity associated with epidurals was 0.86 (95% confidence interval, 0.82-0.90).
The study is limited by its observational design and does not prove causation, the authors acknowledged.
“Labor neuraxial analgesia may facilitate early evaluation and management of the third stage of labor to avoid escalation of postpartum hemorrhaging into grave complications and death,” study author Jean Guglielminotti, MD, PhD, an anesthesiologist at Columbia University, said in a statement.
Concerning trends
The Department of Health & Human Services has labeled severe maternal morbidity a public health priority. Recent data from the Centers for Disease Control and Prevention show an increase in maternal mortality rates and worsening disparities by race and ethnicity.
According to the CDC, 861 women died of maternal causes in 2020, up from 754 in 2019. The rate of maternal mortality increased from 20.1 to 23.8 deaths per 100,000 live births.
For Black women, however, the maternal mortality rate was far higher: 55.3 deaths per 100,000 live births – nearly triple the figure of 19.1 per 100,000 for White women. Between 2019 and 2020, the mortality rate increased significantly for Black and Hispanic women, but not White mothers.
Researchers affiliated with University of Toronto and the Hospital for Sick Children agreed in an accompanying editorial that more access to neuraxial labor analgesia for vaginal delivery might improve maternal health outcomes and “may be a strategy well worth pursuing in public health policy.”
The intervention is relatively safe and can “alleviate discomfort and distress,” they wrote.
Neuraxial anesthesia in surgical procedures has been shown to decrease the risk for complications like deep vein thrombosis, pulmonary embolus, transfusion requirements, and kidney failure, said editorialists Evelina Pankiv, MD; Alan Yang, MSc; and Kazuyoshi Aoyama, MD, PhD.
Benefits potentially could stem from improving blood flow, mitigating hypercoagulation, or reducing surgical stress response. But there are rare risks to consider as well, including hemorrhage, infection, and neurologic injury, they added.
Guglielminotti disclosed grants from the National Institute on Minority Health and Health Disparities. Dr. Aoyama reported receiving grants from the Perioperative Services Facilitator Grant Program and Hospital for Sick Children. The other authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Rheumatology patients seek guidance on CBD, cannabis products
Although there is a lack of evidence for use of cannabidiol (CBD) products and cannabis in rheumatology, many patients are using them anyway and want to discuss the use of these products with their rheumatologists, according to a speaker at the 2022 Rheumatology Winter Clinical Symposium.
While cannabis is still regulated as a Schedule I drug in the United States, CBD products are “all over the place,” Orrin Troum, MD, a rheumatologist at the University of Southern California, Los Angeles, said in his presentation. “You can get it at the pharmacy; you can get it at the dispensaries.”
Patients in rheumatology are also increasingly using cannabis across the United States, Dr. Troum said. In an abstract from the 2019 American College of Rheumatology annual meeting, researchers examined data from FORWARD, the National Databank for Rheumatic Diseases, and found 17.6% of 11,006 respondents reported using cannabis in 2017, an increase from 6.3% of respondents in 2014.
“Putting your personal biases aside, you have to be able to discuss this, and I try to do that openly with my patients,” he said.
According to a 2018 report from the World Health Organization, CBD is “generally well tolerated with a good safety profile.” While CBD itself is safe, CBD products offered over the counter as pills, lotions, foods, drinks, shampoos, cosmetics, oils, and other products carry the risk of being manufactured with “unverified contents” because they are not subject to regulatory oversight.
“There may be heavy metals, pesticides, microbes, [and] mycotoxins that are in these substances that you’re recommending to patients,” Dr. Troum said. There may also be tetrahydrocannabinol (THC) in certain CBD products, he added. Other concerns about CBD products include potential drug-drug interactions with medications used in rheumatology, and potential inhibition of drug metabolism through the CYP450 pathway.
Rheumatologists should be careful when recommending CBD products for this reason, Dr. Troum cautioned. In the absence of products approved by the Food and Drug Administration, “try to get at least products that have a good manufacturing practices certification.”
Dr. Troum highlighted the additional problem of dispensaries recommending specific products, and emphasized that treatment shouldn’t be managed by dispensary personnel without a medical background. “Our patients are being promoted this, either from the dispensaries or even in some clinicians’ offices, without the real true knowledge as to what we’re dealing with,” he said.
Evidence of health effects of CBD, cannabis
When it comes to actual evidence of clinical benefit, “I can tell you there’s lacking data for the majority of what we’re being asked every day in our practices,” Dr. Troum said.
The greatest evidence for the health benefits of cannabinoids appears to be for chronic pain, according to a 2017 report from the National Academies of Sciences, Engineering, and Medicine. Within rheumatology, a position statement released by the Canadian Rheumatology Association in 2019 found insufficient evidence to recommend cannabinoids for use in fibromyalgia, osteoarthritis, RA, or back pain, but acknowledged medical cannabis may relieve symptoms based on evidence from other conditions.
There is some preliminary evidence that cannabis can be used as a substitute for opioids when treating chronic pain, to improve symptoms of fibromyalgia and inflammatory bowel disease, and although a trial of patients with Crohn’s disease failed its primary outcome of disease remission, 10 of 11 patients who smoked cigarettes with THC saw significant improvements in clinical outcomes (P = .028).
In RA, “clinical research focusing on the cannabinoids’ disease-modifying qualities is still lacking,” Dr. Troum said, although an active randomized, controlled trial led by researchers at the University of California, Los Angeles, is testing patients for clinical response to CBD. A separate randomized, double-blind, placebo-controlled trial in Denmark is evaluating whether CBD, followed by open-label add-on of THC, improves chronic pain for patients with RA or ankylosing spondylitis.
The lack of data in this area largely has to do with how cannabis is regulated at the federal level and the differing regulations between U.S. states. “There’s a lot of hurdles you have to go through, and therefore, I think, really has decreased the availability of good studies,” he said.
Overarching principles for medical cannabis in rheumatology
For the rheumatologist counseling a patient who either is self-medicating or wants to start using medical cannabis, the Canadian Rheumatology Association created overarching principles as part of their position statement to guide decision-making for clinicians.
First, clinicians should know that cannabis shouldn’t be used as an alternative treatment for standard of care in rheumatology, and the CRA noted that patients aged under 25 years should not use cannabis. CRA also recommended that clinicians try currently available treatment strategies for common reasons patients seek to use medical cannabis, such as pain relief or a sleep aid, before attempting to use medical cannabis. The CRA noted long-term effects of medical cannabis are not known for patients with rheumatic diseases.
In an interview, Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego, and director of RWCS, said that CBD and cannabis “come up quite frequently” at his clinic. “Many patients have already tried CBD, especially the topical formulation, prior to discussing it with me. In general, I do not dissuade patients from trying CBD, especially topical.”
Typically, he said his practice situation gives him access to a counselor from the anesthesia department with “significant expertise” in dosing and formulations. “It would be great if there were proper controlled trials of specific formulations to allow us to have real scientific data that may help the patients make optimal choices.”
One issue that is brought up by patients is cost. “These preparations can be relatively expensive,” Dr. Kavanaugh said, but noted that this is also a consideration when patients decide to use any therapy.
Dr. Troum reported having financial relationships with eight pharmaceutical companies.
Although there is a lack of evidence for use of cannabidiol (CBD) products and cannabis in rheumatology, many patients are using them anyway and want to discuss the use of these products with their rheumatologists, according to a speaker at the 2022 Rheumatology Winter Clinical Symposium.
While cannabis is still regulated as a Schedule I drug in the United States, CBD products are “all over the place,” Orrin Troum, MD, a rheumatologist at the University of Southern California, Los Angeles, said in his presentation. “You can get it at the pharmacy; you can get it at the dispensaries.”
Patients in rheumatology are also increasingly using cannabis across the United States, Dr. Troum said. In an abstract from the 2019 American College of Rheumatology annual meeting, researchers examined data from FORWARD, the National Databank for Rheumatic Diseases, and found 17.6% of 11,006 respondents reported using cannabis in 2017, an increase from 6.3% of respondents in 2014.
“Putting your personal biases aside, you have to be able to discuss this, and I try to do that openly with my patients,” he said.
According to a 2018 report from the World Health Organization, CBD is “generally well tolerated with a good safety profile.” While CBD itself is safe, CBD products offered over the counter as pills, lotions, foods, drinks, shampoos, cosmetics, oils, and other products carry the risk of being manufactured with “unverified contents” because they are not subject to regulatory oversight.
“There may be heavy metals, pesticides, microbes, [and] mycotoxins that are in these substances that you’re recommending to patients,” Dr. Troum said. There may also be tetrahydrocannabinol (THC) in certain CBD products, he added. Other concerns about CBD products include potential drug-drug interactions with medications used in rheumatology, and potential inhibition of drug metabolism through the CYP450 pathway.
Rheumatologists should be careful when recommending CBD products for this reason, Dr. Troum cautioned. In the absence of products approved by the Food and Drug Administration, “try to get at least products that have a good manufacturing practices certification.”
Dr. Troum highlighted the additional problem of dispensaries recommending specific products, and emphasized that treatment shouldn’t be managed by dispensary personnel without a medical background. “Our patients are being promoted this, either from the dispensaries or even in some clinicians’ offices, without the real true knowledge as to what we’re dealing with,” he said.
Evidence of health effects of CBD, cannabis
When it comes to actual evidence of clinical benefit, “I can tell you there’s lacking data for the majority of what we’re being asked every day in our practices,” Dr. Troum said.
The greatest evidence for the health benefits of cannabinoids appears to be for chronic pain, according to a 2017 report from the National Academies of Sciences, Engineering, and Medicine. Within rheumatology, a position statement released by the Canadian Rheumatology Association in 2019 found insufficient evidence to recommend cannabinoids for use in fibromyalgia, osteoarthritis, RA, or back pain, but acknowledged medical cannabis may relieve symptoms based on evidence from other conditions.
There is some preliminary evidence that cannabis can be used as a substitute for opioids when treating chronic pain, to improve symptoms of fibromyalgia and inflammatory bowel disease, and although a trial of patients with Crohn’s disease failed its primary outcome of disease remission, 10 of 11 patients who smoked cigarettes with THC saw significant improvements in clinical outcomes (P = .028).
In RA, “clinical research focusing on the cannabinoids’ disease-modifying qualities is still lacking,” Dr. Troum said, although an active randomized, controlled trial led by researchers at the University of California, Los Angeles, is testing patients for clinical response to CBD. A separate randomized, double-blind, placebo-controlled trial in Denmark is evaluating whether CBD, followed by open-label add-on of THC, improves chronic pain for patients with RA or ankylosing spondylitis.
The lack of data in this area largely has to do with how cannabis is regulated at the federal level and the differing regulations between U.S. states. “There’s a lot of hurdles you have to go through, and therefore, I think, really has decreased the availability of good studies,” he said.
Overarching principles for medical cannabis in rheumatology
For the rheumatologist counseling a patient who either is self-medicating or wants to start using medical cannabis, the Canadian Rheumatology Association created overarching principles as part of their position statement to guide decision-making for clinicians.
First, clinicians should know that cannabis shouldn’t be used as an alternative treatment for standard of care in rheumatology, and the CRA noted that patients aged under 25 years should not use cannabis. CRA also recommended that clinicians try currently available treatment strategies for common reasons patients seek to use medical cannabis, such as pain relief or a sleep aid, before attempting to use medical cannabis. The CRA noted long-term effects of medical cannabis are not known for patients with rheumatic diseases.
In an interview, Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego, and director of RWCS, said that CBD and cannabis “come up quite frequently” at his clinic. “Many patients have already tried CBD, especially the topical formulation, prior to discussing it with me. In general, I do not dissuade patients from trying CBD, especially topical.”
Typically, he said his practice situation gives him access to a counselor from the anesthesia department with “significant expertise” in dosing and formulations. “It would be great if there were proper controlled trials of specific formulations to allow us to have real scientific data that may help the patients make optimal choices.”
One issue that is brought up by patients is cost. “These preparations can be relatively expensive,” Dr. Kavanaugh said, but noted that this is also a consideration when patients decide to use any therapy.
Dr. Troum reported having financial relationships with eight pharmaceutical companies.
Although there is a lack of evidence for use of cannabidiol (CBD) products and cannabis in rheumatology, many patients are using them anyway and want to discuss the use of these products with their rheumatologists, according to a speaker at the 2022 Rheumatology Winter Clinical Symposium.
While cannabis is still regulated as a Schedule I drug in the United States, CBD products are “all over the place,” Orrin Troum, MD, a rheumatologist at the University of Southern California, Los Angeles, said in his presentation. “You can get it at the pharmacy; you can get it at the dispensaries.”
Patients in rheumatology are also increasingly using cannabis across the United States, Dr. Troum said. In an abstract from the 2019 American College of Rheumatology annual meeting, researchers examined data from FORWARD, the National Databank for Rheumatic Diseases, and found 17.6% of 11,006 respondents reported using cannabis in 2017, an increase from 6.3% of respondents in 2014.
“Putting your personal biases aside, you have to be able to discuss this, and I try to do that openly with my patients,” he said.
According to a 2018 report from the World Health Organization, CBD is “generally well tolerated with a good safety profile.” While CBD itself is safe, CBD products offered over the counter as pills, lotions, foods, drinks, shampoos, cosmetics, oils, and other products carry the risk of being manufactured with “unverified contents” because they are not subject to regulatory oversight.
“There may be heavy metals, pesticides, microbes, [and] mycotoxins that are in these substances that you’re recommending to patients,” Dr. Troum said. There may also be tetrahydrocannabinol (THC) in certain CBD products, he added. Other concerns about CBD products include potential drug-drug interactions with medications used in rheumatology, and potential inhibition of drug metabolism through the CYP450 pathway.
Rheumatologists should be careful when recommending CBD products for this reason, Dr. Troum cautioned. In the absence of products approved by the Food and Drug Administration, “try to get at least products that have a good manufacturing practices certification.”
Dr. Troum highlighted the additional problem of dispensaries recommending specific products, and emphasized that treatment shouldn’t be managed by dispensary personnel without a medical background. “Our patients are being promoted this, either from the dispensaries or even in some clinicians’ offices, without the real true knowledge as to what we’re dealing with,” he said.
Evidence of health effects of CBD, cannabis
When it comes to actual evidence of clinical benefit, “I can tell you there’s lacking data for the majority of what we’re being asked every day in our practices,” Dr. Troum said.
The greatest evidence for the health benefits of cannabinoids appears to be for chronic pain, according to a 2017 report from the National Academies of Sciences, Engineering, and Medicine. Within rheumatology, a position statement released by the Canadian Rheumatology Association in 2019 found insufficient evidence to recommend cannabinoids for use in fibromyalgia, osteoarthritis, RA, or back pain, but acknowledged medical cannabis may relieve symptoms based on evidence from other conditions.
There is some preliminary evidence that cannabis can be used as a substitute for opioids when treating chronic pain, to improve symptoms of fibromyalgia and inflammatory bowel disease, and although a trial of patients with Crohn’s disease failed its primary outcome of disease remission, 10 of 11 patients who smoked cigarettes with THC saw significant improvements in clinical outcomes (P = .028).
In RA, “clinical research focusing on the cannabinoids’ disease-modifying qualities is still lacking,” Dr. Troum said, although an active randomized, controlled trial led by researchers at the University of California, Los Angeles, is testing patients for clinical response to CBD. A separate randomized, double-blind, placebo-controlled trial in Denmark is evaluating whether CBD, followed by open-label add-on of THC, improves chronic pain for patients with RA or ankylosing spondylitis.
The lack of data in this area largely has to do with how cannabis is regulated at the federal level and the differing regulations between U.S. states. “There’s a lot of hurdles you have to go through, and therefore, I think, really has decreased the availability of good studies,” he said.
Overarching principles for medical cannabis in rheumatology
For the rheumatologist counseling a patient who either is self-medicating or wants to start using medical cannabis, the Canadian Rheumatology Association created overarching principles as part of their position statement to guide decision-making for clinicians.
First, clinicians should know that cannabis shouldn’t be used as an alternative treatment for standard of care in rheumatology, and the CRA noted that patients aged under 25 years should not use cannabis. CRA also recommended that clinicians try currently available treatment strategies for common reasons patients seek to use medical cannabis, such as pain relief or a sleep aid, before attempting to use medical cannabis. The CRA noted long-term effects of medical cannabis are not known for patients with rheumatic diseases.
In an interview, Arthur Kavanaugh, MD, professor of medicine at the University of California, San Diego, and director of RWCS, said that CBD and cannabis “come up quite frequently” at his clinic. “Many patients have already tried CBD, especially the topical formulation, prior to discussing it with me. In general, I do not dissuade patients from trying CBD, especially topical.”
Typically, he said his practice situation gives him access to a counselor from the anesthesia department with “significant expertise” in dosing and formulations. “It would be great if there were proper controlled trials of specific formulations to allow us to have real scientific data that may help the patients make optimal choices.”
One issue that is brought up by patients is cost. “These preparations can be relatively expensive,” Dr. Kavanaugh said, but noted that this is also a consideration when patients decide to use any therapy.
Dr. Troum reported having financial relationships with eight pharmaceutical companies.
FROM RWCS 2022
CDC releases updated draft guidance on opioid prescribing
The Centers for Disease Controls and Prevention has released a draft update of its current Clinical Practice Guidelines for Prescribing Opioids for pain management and is asking for public comment before moving forward.
The last guidance on this topic was released in 2016 and, among other things, noted that clinicians should be cautious when considering increasing dosage of opioids to 50 or more morphine milligram equivalents (MME)/day and should avoid increasing to a dose of 90 or more MME/day. It also noted that 3 days or less “will often be sufficient” regarding the quantity of lowest effective dose of immediate-release opioids to be prescribed for acute pain – and that more than 7 days “will rarely be needed.”
In the new report from the CDC’s National Center for Injury Prevention and Control (NCIPC), those dose limits have been replaced with the suggestion that clinicians use their best judgement – albeit still urging conservative use and even the possibility of nonopioid treatments.
The updated recommendations are now open for public comment via the Federal Register’s website through April 11.
“This comment period provides another critical opportunity for diverse audiences to offer their perspective on the draft clinical practice guideline,” Christopher M. Jones, PharmD, DrPH, acting director for the NCIPC, said in a release.
“We want to hear many voices from the public, including people living with pain and health care providers who help their patients manage pain,” Dr. Jones added.
Outpatient recommendations
The CDC noted that the updated guidance provides “evidence-based recommendations” for treatment of adults with acute, subacute, or chronic pain. It does not include guidance for managing pain related to sickle cell disease, cancer, or palliative care.
It is aimed at primary care clinicians and others who manage pain in an outpatient setting, including in dental and postsurgical practices and for those discharging patients from emergency departments. It does not apply to inpatient care.
The draft guidance includes 12 recommendations focused on four key areas:
- Helping clinicians determine whether or not to initiate opioid treatment for pain
- Opioid selection and dosage
- Duration of use and follow-up
- Assessing risk and addressing potential harms from use
The overall aim “is to ensure people have access to safe, accessible, and effective pain management that improves their function and quality of life while illuminating and reducing risks associated with prescription opioids and ultimately reducing the consequences of prescription opioid misuse and overdose,” the CDC notes.
In addition, the guidance itself “is intended to be a clinical tool to improve communication between providers and patients and empower them to make informed, patient-centered decisions,” the agency said in a press release.
It added that the new recommendations “are not intended to be applied as inflexible standards of care.” Rather, it is intended as a guide to support health care providers in their clinical decisionmaking as they provide individualized patient care.
Patients, caregivers, and providers are invited to submit comments over the next 60 days through the Federal Register docket.
“It is vitally important to CDC that we receive, process, and understand public feedback during the guideline update process,” the agency noted.
“The ultimate goal of this clinical practice guideline is to help people set and achieve personal goals to reduce their pain and improve their function and quality of life. Getting feedback from the public is essential to achieving this goal,” Dr. Jones said.
A version of this article first appeared on Medscape.com.
The Centers for Disease Controls and Prevention has released a draft update of its current Clinical Practice Guidelines for Prescribing Opioids for pain management and is asking for public comment before moving forward.
The last guidance on this topic was released in 2016 and, among other things, noted that clinicians should be cautious when considering increasing dosage of opioids to 50 or more morphine milligram equivalents (MME)/day and should avoid increasing to a dose of 90 or more MME/day. It also noted that 3 days or less “will often be sufficient” regarding the quantity of lowest effective dose of immediate-release opioids to be prescribed for acute pain – and that more than 7 days “will rarely be needed.”
In the new report from the CDC’s National Center for Injury Prevention and Control (NCIPC), those dose limits have been replaced with the suggestion that clinicians use their best judgement – albeit still urging conservative use and even the possibility of nonopioid treatments.
The updated recommendations are now open for public comment via the Federal Register’s website through April 11.
“This comment period provides another critical opportunity for diverse audiences to offer their perspective on the draft clinical practice guideline,” Christopher M. Jones, PharmD, DrPH, acting director for the NCIPC, said in a release.
“We want to hear many voices from the public, including people living with pain and health care providers who help their patients manage pain,” Dr. Jones added.
Outpatient recommendations
The CDC noted that the updated guidance provides “evidence-based recommendations” for treatment of adults with acute, subacute, or chronic pain. It does not include guidance for managing pain related to sickle cell disease, cancer, or palliative care.
It is aimed at primary care clinicians and others who manage pain in an outpatient setting, including in dental and postsurgical practices and for those discharging patients from emergency departments. It does not apply to inpatient care.
The draft guidance includes 12 recommendations focused on four key areas:
- Helping clinicians determine whether or not to initiate opioid treatment for pain
- Opioid selection and dosage
- Duration of use and follow-up
- Assessing risk and addressing potential harms from use
The overall aim “is to ensure people have access to safe, accessible, and effective pain management that improves their function and quality of life while illuminating and reducing risks associated with prescription opioids and ultimately reducing the consequences of prescription opioid misuse and overdose,” the CDC notes.
In addition, the guidance itself “is intended to be a clinical tool to improve communication between providers and patients and empower them to make informed, patient-centered decisions,” the agency said in a press release.
It added that the new recommendations “are not intended to be applied as inflexible standards of care.” Rather, it is intended as a guide to support health care providers in their clinical decisionmaking as they provide individualized patient care.
Patients, caregivers, and providers are invited to submit comments over the next 60 days through the Federal Register docket.
“It is vitally important to CDC that we receive, process, and understand public feedback during the guideline update process,” the agency noted.
“The ultimate goal of this clinical practice guideline is to help people set and achieve personal goals to reduce their pain and improve their function and quality of life. Getting feedback from the public is essential to achieving this goal,” Dr. Jones said.
A version of this article first appeared on Medscape.com.
The Centers for Disease Controls and Prevention has released a draft update of its current Clinical Practice Guidelines for Prescribing Opioids for pain management and is asking for public comment before moving forward.
The last guidance on this topic was released in 2016 and, among other things, noted that clinicians should be cautious when considering increasing dosage of opioids to 50 or more morphine milligram equivalents (MME)/day and should avoid increasing to a dose of 90 or more MME/day. It also noted that 3 days or less “will often be sufficient” regarding the quantity of lowest effective dose of immediate-release opioids to be prescribed for acute pain – and that more than 7 days “will rarely be needed.”
In the new report from the CDC’s National Center for Injury Prevention and Control (NCIPC), those dose limits have been replaced with the suggestion that clinicians use their best judgement – albeit still urging conservative use and even the possibility of nonopioid treatments.
The updated recommendations are now open for public comment via the Federal Register’s website through April 11.
“This comment period provides another critical opportunity for diverse audiences to offer their perspective on the draft clinical practice guideline,” Christopher M. Jones, PharmD, DrPH, acting director for the NCIPC, said in a release.
“We want to hear many voices from the public, including people living with pain and health care providers who help their patients manage pain,” Dr. Jones added.
Outpatient recommendations
The CDC noted that the updated guidance provides “evidence-based recommendations” for treatment of adults with acute, subacute, or chronic pain. It does not include guidance for managing pain related to sickle cell disease, cancer, or palliative care.
It is aimed at primary care clinicians and others who manage pain in an outpatient setting, including in dental and postsurgical practices and for those discharging patients from emergency departments. It does not apply to inpatient care.
The draft guidance includes 12 recommendations focused on four key areas:
- Helping clinicians determine whether or not to initiate opioid treatment for pain
- Opioid selection and dosage
- Duration of use and follow-up
- Assessing risk and addressing potential harms from use
The overall aim “is to ensure people have access to safe, accessible, and effective pain management that improves their function and quality of life while illuminating and reducing risks associated with prescription opioids and ultimately reducing the consequences of prescription opioid misuse and overdose,” the CDC notes.
In addition, the guidance itself “is intended to be a clinical tool to improve communication between providers and patients and empower them to make informed, patient-centered decisions,” the agency said in a press release.
It added that the new recommendations “are not intended to be applied as inflexible standards of care.” Rather, it is intended as a guide to support health care providers in their clinical decisionmaking as they provide individualized patient care.
Patients, caregivers, and providers are invited to submit comments over the next 60 days through the Federal Register docket.
“It is vitally important to CDC that we receive, process, and understand public feedback during the guideline update process,” the agency noted.
“The ultimate goal of this clinical practice guideline is to help people set and achieve personal goals to reduce their pain and improve their function and quality of life. Getting feedback from the public is essential to achieving this goal,” Dr. Jones said.
A version of this article first appeared on Medscape.com.
‘Encouraging’ new national data on chronic pain management
Most adults in the United States who have chronic pain favor a combination of nondrug and nonopioid approaches to control their pain, which is “encouraging,” new research shows.
A national survey reveals 55% of adults with chronic pain used pain management techniques that did not involve any opioids at all during the prior 3-month period.
However, few participants took advantage of cognitive-behavioral therapy (CBT), which is effective for easing chronic pain, Cornelius Groenewald, MB ChB, department of anesthesiology and pain medicine, University of Seattle, and colleagues write.
The results were published online in a research letter Feb. 7 in JAMA Network Open.
First time for pain questions
An estimated 50.2 million U.S. adults experience chronic pain, according to the 2019 National Health Interview Survey.
The 2019 version of the survey included questions on pain management techniques for the first time. Adults with chronic pain were asked to report on their use of 11 pain management techniques during the previous 3 months.
Among the 31,916 survey respondents, 64% were women; 69% were non-Hispanic White, 13% were Hispanic, and 11% were non-Hispanic Black; 71% were between 18 and 64 years of age, and 29% were 65 and older.
Among the key findings, an estimated 55% of adults with chronic pain used only nonopioid pain management techniques, 11% used both opioids and nonopioid techniques, and 4% used only opioids for chronic pain management; 30% did not report any pain management techniques during the previous 3 months.
Complementary therapies were the most commonly used nonopioid pain management technique (by 35% of adults with chronic pain), followed by physical, occupational, or rehabilitative therapies (19%).
Only about 4% of adults with chronic pain used CBT.
Other techniques used included self-management programs (5%) and chronic pain peer support groups (2%). In addition, 39% of adults with chronic pain reported using other pain approaches not specifically captured in the data set.
Benchmark data
Participants using complementary and psychological or psychotherapeutic interventions were more likely to be younger women with more education, the investigators report.
Adults using physical, occupational, or rehabilitative therapy were more likely to be highly educated older women with medical insurance.
Prescription opioid use for chronic pain was more common among older adults aged 45-64 years vs. those aged 18-44 years (19% vs. 8%).
It was also more common in women than men (17% vs. 13%), in adults with vs. without health insurance (16% vs. 6%), and in those with a high school education or lower, compared with those had more than a high school education (17% vs. 14%).
Prescription opioid use was less common among adults making $100,000 or more annually than in those making less than $35,000 a year (9% vs. 20%).
“While effective for some, opioids prescribed for chronic pain management remain an important determinant of the national opioid crisis,” the investigators write.
The study “provides baseline information on opioid and nonopioid pain management techniques used for chronic pain and serves as a benchmark for evaluating the outcome of health care policies aimed at reducing prescription opioid use,” they add.
The study had no specific funding. The investigators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Most adults in the United States who have chronic pain favor a combination of nondrug and nonopioid approaches to control their pain, which is “encouraging,” new research shows.
A national survey reveals 55% of adults with chronic pain used pain management techniques that did not involve any opioids at all during the prior 3-month period.
However, few participants took advantage of cognitive-behavioral therapy (CBT), which is effective for easing chronic pain, Cornelius Groenewald, MB ChB, department of anesthesiology and pain medicine, University of Seattle, and colleagues write.
The results were published online in a research letter Feb. 7 in JAMA Network Open.
First time for pain questions
An estimated 50.2 million U.S. adults experience chronic pain, according to the 2019 National Health Interview Survey.
The 2019 version of the survey included questions on pain management techniques for the first time. Adults with chronic pain were asked to report on their use of 11 pain management techniques during the previous 3 months.
Among the 31,916 survey respondents, 64% were women; 69% were non-Hispanic White, 13% were Hispanic, and 11% were non-Hispanic Black; 71% were between 18 and 64 years of age, and 29% were 65 and older.
Among the key findings, an estimated 55% of adults with chronic pain used only nonopioid pain management techniques, 11% used both opioids and nonopioid techniques, and 4% used only opioids for chronic pain management; 30% did not report any pain management techniques during the previous 3 months.
Complementary therapies were the most commonly used nonopioid pain management technique (by 35% of adults with chronic pain), followed by physical, occupational, or rehabilitative therapies (19%).
Only about 4% of adults with chronic pain used CBT.
Other techniques used included self-management programs (5%) and chronic pain peer support groups (2%). In addition, 39% of adults with chronic pain reported using other pain approaches not specifically captured in the data set.
Benchmark data
Participants using complementary and psychological or psychotherapeutic interventions were more likely to be younger women with more education, the investigators report.
Adults using physical, occupational, or rehabilitative therapy were more likely to be highly educated older women with medical insurance.
Prescription opioid use for chronic pain was more common among older adults aged 45-64 years vs. those aged 18-44 years (19% vs. 8%).
It was also more common in women than men (17% vs. 13%), in adults with vs. without health insurance (16% vs. 6%), and in those with a high school education or lower, compared with those had more than a high school education (17% vs. 14%).
Prescription opioid use was less common among adults making $100,000 or more annually than in those making less than $35,000 a year (9% vs. 20%).
“While effective for some, opioids prescribed for chronic pain management remain an important determinant of the national opioid crisis,” the investigators write.
The study “provides baseline information on opioid and nonopioid pain management techniques used for chronic pain and serves as a benchmark for evaluating the outcome of health care policies aimed at reducing prescription opioid use,” they add.
The study had no specific funding. The investigators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Most adults in the United States who have chronic pain favor a combination of nondrug and nonopioid approaches to control their pain, which is “encouraging,” new research shows.
A national survey reveals 55% of adults with chronic pain used pain management techniques that did not involve any opioids at all during the prior 3-month period.
However, few participants took advantage of cognitive-behavioral therapy (CBT), which is effective for easing chronic pain, Cornelius Groenewald, MB ChB, department of anesthesiology and pain medicine, University of Seattle, and colleagues write.
The results were published online in a research letter Feb. 7 in JAMA Network Open.
First time for pain questions
An estimated 50.2 million U.S. adults experience chronic pain, according to the 2019 National Health Interview Survey.
The 2019 version of the survey included questions on pain management techniques for the first time. Adults with chronic pain were asked to report on their use of 11 pain management techniques during the previous 3 months.
Among the 31,916 survey respondents, 64% were women; 69% were non-Hispanic White, 13% were Hispanic, and 11% were non-Hispanic Black; 71% were between 18 and 64 years of age, and 29% were 65 and older.
Among the key findings, an estimated 55% of adults with chronic pain used only nonopioid pain management techniques, 11% used both opioids and nonopioid techniques, and 4% used only opioids for chronic pain management; 30% did not report any pain management techniques during the previous 3 months.
Complementary therapies were the most commonly used nonopioid pain management technique (by 35% of adults with chronic pain), followed by physical, occupational, or rehabilitative therapies (19%).
Only about 4% of adults with chronic pain used CBT.
Other techniques used included self-management programs (5%) and chronic pain peer support groups (2%). In addition, 39% of adults with chronic pain reported using other pain approaches not specifically captured in the data set.
Benchmark data
Participants using complementary and psychological or psychotherapeutic interventions were more likely to be younger women with more education, the investigators report.
Adults using physical, occupational, or rehabilitative therapy were more likely to be highly educated older women with medical insurance.
Prescription opioid use for chronic pain was more common among older adults aged 45-64 years vs. those aged 18-44 years (19% vs. 8%).
It was also more common in women than men (17% vs. 13%), in adults with vs. without health insurance (16% vs. 6%), and in those with a high school education or lower, compared with those had more than a high school education (17% vs. 14%).
Prescription opioid use was less common among adults making $100,000 or more annually than in those making less than $35,000 a year (9% vs. 20%).
“While effective for some, opioids prescribed for chronic pain management remain an important determinant of the national opioid crisis,” the investigators write.
The study “provides baseline information on opioid and nonopioid pain management techniques used for chronic pain and serves as a benchmark for evaluating the outcome of health care policies aimed at reducing prescription opioid use,” they add.
The study had no specific funding. The investigators have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Opioid deaths in North America predicted to soar
“Over the past quarter-century, the opioid epidemic has taken nearly 600,000 lives and triggered a cascade of public health catastrophes such as disability, family breakdown, unemployment, and child neglect in North America,” commission chair Keith Humphreys, PhD, said in a news release.
“If no action is taken, by the end of this decade, we are predicting the number of deaths to be twice as high as it has been over the last 20 years,” said Dr. Humphreys, professor of psychiatry and behavioral sciences at Stanford (Calif.) University.
The report was published online Feb. 2, 2022, in The Lancet.
Blame it on COVID-19?
The COVID-19 pandemic has both overshadowed and exacerbated the opioid crisis in North America, the commission pointed out in their report.
Their analysis suggests that 2020 was the worst year on record for overdose deaths in the United States and Canada in terms of both the total number of deaths and percentage annual increase.
In the United States, opioid overdose deaths increased by 37%, from 51,133 in 2019 to 70,168 in 2020, bringing the total number of deaths since 1999 to 583,000.
In Canada, opioid overdose deaths jumped by 72%, from 3,668 in 2019 to 6,306 in 2020, with a further 3,515 deaths reported in the first 6 months of 2021.
Although the 2020 spikes might be partly caused by the effects of the COVID-19 pandemic, a rising trajectory of deaths was evident in both the United States and Canada before the pandemic hit, the Stanford-Lancet Commission said.
Profit motives, lack of regulation
The commission blames the opioid epidemic on a lack of adequate regulation and oversight coupled with profit motives of the pharmaceutical and health care industry.
“To ensure safeguards are in place to curb the opioid addiction epidemic and prevent future ones involving other addictive drugs, we must end the pharmaceutical and health care industry’s undue influence on the government and its unregulated push for opioid use,” commission member Howard Koh, MD, MPH, said in the news release.
“This includes insulating the medical community from pharmaceutical company influence and closing the constantly revolving door between regulators and industry,” said Dr. Koh, with the Harvard School of Public Health, Boston.
In addition to regulation and policy reform, the commission said prevention efforts that focus on treating addiction as a chronic condition are key.
The United States in particular lacks accessible, high-quality, nonstigmatizing, and integrated health and social care services for people experiencing opioid use disorder, the Commission notes.
Addiction-related services must become a permanent feature of health and social care systems in the United States and Canada, in line with established chronic disease management models that are financed and organized as a core public health commitment, the commission said.
“Addiction is an enduring part of population health and should not be treated as a moral failing that needs punishment but as a chronic health condition that requires ongoing treatment and long-term support,” commission member Yasmin Hurd, PhD, director of the Addiction Institute at Icahn School of Medicine at Mount Sinai, New York, said in the release.
Investing in young people to reduce the risk of addiction will also be important going forward.
“Preventing drug addiction should be part of a comprehensive public health strategy that starts in childhood and lays the foundation for long-term declines in addiction,” said commission member Chelsea Shover, PhD, with the University of California, Los Angeles.
‘Audacious but achievable goal’
The commission calls for a nuanced approach to pain management that prioritizes innovation both in society’s response to drug addiction through policy reform and by supporting the development of new, nonaddictive pain management options.
“Opioids should not be viewed as good or bad, but instead as a class of medications essential to the management of pain. However, opioids also come with serious risks, some of which can be difficult to recognize,” commission member David Juurlink, MD, PhD, said in the release.
“Clinicians should begin learning about responsible pain management prescribing in medical school and continue to learn about it as part of their commitment to continued medical education throughout their careers,” said Dr. Juurlink, with Sunnybrook Health Sciences Centre in Toronto.
Humphreys said ending the opioid epidemic in North America and preventing its global spread is “an audacious but achievable goal” that will require a “dramatic shift in policy and culture where innovation, collaboration, and regulation are encouraged.
“We can save and improve lives by summoning the resources and political will necessary to eliminate the sources of addiction and boldly implement policies that will maximize efforts to treat it,” Dr. Humphreys added.
The study was funded by Stanford University.
A version of this article first appeared on Medscape.com.
“Over the past quarter-century, the opioid epidemic has taken nearly 600,000 lives and triggered a cascade of public health catastrophes such as disability, family breakdown, unemployment, and child neglect in North America,” commission chair Keith Humphreys, PhD, said in a news release.
“If no action is taken, by the end of this decade, we are predicting the number of deaths to be twice as high as it has been over the last 20 years,” said Dr. Humphreys, professor of psychiatry and behavioral sciences at Stanford (Calif.) University.
The report was published online Feb. 2, 2022, in The Lancet.
Blame it on COVID-19?
The COVID-19 pandemic has both overshadowed and exacerbated the opioid crisis in North America, the commission pointed out in their report.
Their analysis suggests that 2020 was the worst year on record for overdose deaths in the United States and Canada in terms of both the total number of deaths and percentage annual increase.
In the United States, opioid overdose deaths increased by 37%, from 51,133 in 2019 to 70,168 in 2020, bringing the total number of deaths since 1999 to 583,000.
In Canada, opioid overdose deaths jumped by 72%, from 3,668 in 2019 to 6,306 in 2020, with a further 3,515 deaths reported in the first 6 months of 2021.
Although the 2020 spikes might be partly caused by the effects of the COVID-19 pandemic, a rising trajectory of deaths was evident in both the United States and Canada before the pandemic hit, the Stanford-Lancet Commission said.
Profit motives, lack of regulation
The commission blames the opioid epidemic on a lack of adequate regulation and oversight coupled with profit motives of the pharmaceutical and health care industry.
“To ensure safeguards are in place to curb the opioid addiction epidemic and prevent future ones involving other addictive drugs, we must end the pharmaceutical and health care industry’s undue influence on the government and its unregulated push for opioid use,” commission member Howard Koh, MD, MPH, said in the news release.
“This includes insulating the medical community from pharmaceutical company influence and closing the constantly revolving door between regulators and industry,” said Dr. Koh, with the Harvard School of Public Health, Boston.
In addition to regulation and policy reform, the commission said prevention efforts that focus on treating addiction as a chronic condition are key.
The United States in particular lacks accessible, high-quality, nonstigmatizing, and integrated health and social care services for people experiencing opioid use disorder, the Commission notes.
Addiction-related services must become a permanent feature of health and social care systems in the United States and Canada, in line with established chronic disease management models that are financed and organized as a core public health commitment, the commission said.
“Addiction is an enduring part of population health and should not be treated as a moral failing that needs punishment but as a chronic health condition that requires ongoing treatment and long-term support,” commission member Yasmin Hurd, PhD, director of the Addiction Institute at Icahn School of Medicine at Mount Sinai, New York, said in the release.
Investing in young people to reduce the risk of addiction will also be important going forward.
“Preventing drug addiction should be part of a comprehensive public health strategy that starts in childhood and lays the foundation for long-term declines in addiction,” said commission member Chelsea Shover, PhD, with the University of California, Los Angeles.
‘Audacious but achievable goal’
The commission calls for a nuanced approach to pain management that prioritizes innovation both in society’s response to drug addiction through policy reform and by supporting the development of new, nonaddictive pain management options.
“Opioids should not be viewed as good or bad, but instead as a class of medications essential to the management of pain. However, opioids also come with serious risks, some of which can be difficult to recognize,” commission member David Juurlink, MD, PhD, said in the release.
“Clinicians should begin learning about responsible pain management prescribing in medical school and continue to learn about it as part of their commitment to continued medical education throughout their careers,” said Dr. Juurlink, with Sunnybrook Health Sciences Centre in Toronto.
Humphreys said ending the opioid epidemic in North America and preventing its global spread is “an audacious but achievable goal” that will require a “dramatic shift in policy and culture where innovation, collaboration, and regulation are encouraged.
“We can save and improve lives by summoning the resources and political will necessary to eliminate the sources of addiction and boldly implement policies that will maximize efforts to treat it,” Dr. Humphreys added.
The study was funded by Stanford University.
A version of this article first appeared on Medscape.com.
“Over the past quarter-century, the opioid epidemic has taken nearly 600,000 lives and triggered a cascade of public health catastrophes such as disability, family breakdown, unemployment, and child neglect in North America,” commission chair Keith Humphreys, PhD, said in a news release.
“If no action is taken, by the end of this decade, we are predicting the number of deaths to be twice as high as it has been over the last 20 years,” said Dr. Humphreys, professor of psychiatry and behavioral sciences at Stanford (Calif.) University.
The report was published online Feb. 2, 2022, in The Lancet.
Blame it on COVID-19?
The COVID-19 pandemic has both overshadowed and exacerbated the opioid crisis in North America, the commission pointed out in their report.
Their analysis suggests that 2020 was the worst year on record for overdose deaths in the United States and Canada in terms of both the total number of deaths and percentage annual increase.
In the United States, opioid overdose deaths increased by 37%, from 51,133 in 2019 to 70,168 in 2020, bringing the total number of deaths since 1999 to 583,000.
In Canada, opioid overdose deaths jumped by 72%, from 3,668 in 2019 to 6,306 in 2020, with a further 3,515 deaths reported in the first 6 months of 2021.
Although the 2020 spikes might be partly caused by the effects of the COVID-19 pandemic, a rising trajectory of deaths was evident in both the United States and Canada before the pandemic hit, the Stanford-Lancet Commission said.
Profit motives, lack of regulation
The commission blames the opioid epidemic on a lack of adequate regulation and oversight coupled with profit motives of the pharmaceutical and health care industry.
“To ensure safeguards are in place to curb the opioid addiction epidemic and prevent future ones involving other addictive drugs, we must end the pharmaceutical and health care industry’s undue influence on the government and its unregulated push for opioid use,” commission member Howard Koh, MD, MPH, said in the news release.
“This includes insulating the medical community from pharmaceutical company influence and closing the constantly revolving door between regulators and industry,” said Dr. Koh, with the Harvard School of Public Health, Boston.
In addition to regulation and policy reform, the commission said prevention efforts that focus on treating addiction as a chronic condition are key.
The United States in particular lacks accessible, high-quality, nonstigmatizing, and integrated health and social care services for people experiencing opioid use disorder, the Commission notes.
Addiction-related services must become a permanent feature of health and social care systems in the United States and Canada, in line with established chronic disease management models that are financed and organized as a core public health commitment, the commission said.
“Addiction is an enduring part of population health and should not be treated as a moral failing that needs punishment but as a chronic health condition that requires ongoing treatment and long-term support,” commission member Yasmin Hurd, PhD, director of the Addiction Institute at Icahn School of Medicine at Mount Sinai, New York, said in the release.
Investing in young people to reduce the risk of addiction will also be important going forward.
“Preventing drug addiction should be part of a comprehensive public health strategy that starts in childhood and lays the foundation for long-term declines in addiction,” said commission member Chelsea Shover, PhD, with the University of California, Los Angeles.
‘Audacious but achievable goal’
The commission calls for a nuanced approach to pain management that prioritizes innovation both in society’s response to drug addiction through policy reform and by supporting the development of new, nonaddictive pain management options.
“Opioids should not be viewed as good or bad, but instead as a class of medications essential to the management of pain. However, opioids also come with serious risks, some of which can be difficult to recognize,” commission member David Juurlink, MD, PhD, said in the release.
“Clinicians should begin learning about responsible pain management prescribing in medical school and continue to learn about it as part of their commitment to continued medical education throughout their careers,” said Dr. Juurlink, with Sunnybrook Health Sciences Centre in Toronto.
Humphreys said ending the opioid epidemic in North America and preventing its global spread is “an audacious but achievable goal” that will require a “dramatic shift in policy and culture where innovation, collaboration, and regulation are encouraged.
“We can save and improve lives by summoning the resources and political will necessary to eliminate the sources of addiction and boldly implement policies that will maximize efforts to treat it,” Dr. Humphreys added.
The study was funded by Stanford University.
A version of this article first appeared on Medscape.com.
FROM THE LANCET
Potential new neuromodulation treatment for migraines
Most people avoid smartphones when they have a migraine headache, but a noninvasive treatment for episodic migraines may change that.
that can help ease migraine pain.
Tina Montgomery, 58, has suffered from migraines since childhood and spent years looking for something to help manage them. Doctors consider her a “chronic” sufferer in that she has more than 14 migraines a month (fewer than 14 is considered “episodic”). Prescription antidepressants, anticonvulsants, and botulinum toxin shots as preventive treatments helped a little but not enough.
A few years ago, she found some relief using a new preventive injectable medication that targets a peptide known as CGRP, combined with an oral CGRP rescue medication, ubrogepant (Ubrelvy). However, by early 2021, Ms. Montgomery’s chronic migraines were back as she faced stress from the pandemic and her role as a caregiver for her aging parents.
“I was going through so much medication. I just didn’t feel good taking so much,” she said.
Looking for relief, she read about Nerivio, a wearable migraine treatment device that uses remote electrical neuromodulation (REN). She mentioned the device to her neurologist, and he agreed she might benefit from trying it out. Today, she uses the device whenever she feels a migraine may be imminent, she said.
“It really helps me stave off migraines I feel coming on and the milder ones where I would normally hesitate to use prescription medication because [insurance] limits the number of pills they give you in a month,” she said. “I follow through with the Nerivio treatment and usually find that my migraine doesn’t fully develop or is completely gone, and I don’t get a migraine at all.”
Taking it on the arm
The device works by stimulating nerves at the back of the arm right around the triceps. “Those nerve fibers relay information to the brain stem [so it can] work its magic and use the brain’s own natural mechanisms for reducing pain,” said Brian M. Grosberg, MD, director of the Hartford Healthcare Ayer Neuroscience Institute Headache Center, West Hartford, Conn.
These mechanisms are like a bait-and-switch for the brain, said Britany Klenofsky, MD, assistant professor of neurology, Icahn School of Medicine at Mount Sinai, New York. “You’re trying to stimulate pain somewhere else [on the body] to tell the brain to protect itself and release [the neurotransmitter] serotonin,” she said. “You do this by putting the device on your arm, an area that’s away from the head where the pain is actively occurring, turning the device on, and increasing the stimulation to a nearly painful stimulus.”
This pseudo pain prompts the brain to release serotonin, the feel-good hormone along with norepinephrine and noradrenaline. The device works best when it’s used as soon as a migraine starts, so patients should hook up Nerivio within the first 20-30 minutes of onset of pain, said Dr. Grosberg, who was an investigator on the double-blind treatment study that led to FDA clearance. If patients wait too long, the device may not work.
This is why as soon as Ms. Montgomery feels a migraine aura (there are six types of migraine auras, including visual changes and muscle weakness) that occurs right before a migraine strikes, she puts the device armband on her upper arm and launches its smartphone app. Then she turns on the device for a 45-minute treatment, which begins with what she characterizes as tingling and vibration sensations on her arm. She turns up the intensity of the sensations, which are mild electric currents, until they are well-felt but not painful.
Ms. Montgomery said she can use the device and multitask since there’s no need for her to lie down or sit in a darkened room. And since it is worn on the arm, she can wear it under a shirtsleeve while working or out in public without anyone noticing. She also uses the app’s migraine diary and guided meditation to help reduce the anxiety that often accompanies her migraines.
The device is approved for adolescents and adults and can be used for both episodic and chronic migraines. From an efficacy standpoint, the device provides relief about as well as a commonly used pharmaceutical class of drugs, triptans. About 37% of people with episodic migraine achieved complete freedom from pain 2 hours after their treatment. In addition, about two-thirds of people reported pain relief after 2 hours, which is better success than people find with many prescription and nonprescription drugs.
A separate study looked at acute treatment for chronic migraine sufferers and found nearly 60% of people using the device found relief and 21% said they were pain-free after 2 hours. Almost two-thirds of those who experienced pain relief were pain-free 24 hours after the treatment.
Finding the perfect patient
There are other FDA-cleared noninvasive devices to treat migraines. One device, CEFALY, is an external trigeminal nerve stimulation device that sits on the forehead. Another device, SpringTMS, uses transcranial magnetic stimulation on the back of the head. A third option, the gammaCore Sapphire, is placed on the neck to stimulate the vagus nerve. All three have been cleared by the FDA to work as preventive and acute treatments for migraine.
Theranica, the company that developed Nerivio, is trying to boost use of the device by allowing patients to get a prescription via telehealth visits with a physician.
The company, as well as the companies behind the other neuromodulation devices, are marketing their treatments to children ages 12 and up since nonpharmacologic options are often preferable for parents, said Thomas Berk, MD, a clinical associate professor in the division of headache at NYU Langone Health in New York.
Dr. Berk said the devices could be appealing for those people who don’t want or can’t take medication, such as pregnant women or those who don’t respond well to drugs. “[They] could also be used by somebody who needs something in addition to a medication,” he said.
For now, people like Ms. Montgomery say they are happy to have another tool in their migraine arsenal. “Overall, I’m taking less medication because I haven’t had to have my Ubrelvy refilled as often as I used to,” she said. “It’s really helped me manage changes and stresses in my life.”
A version of this article first appeared on Medscape.com.
Most people avoid smartphones when they have a migraine headache, but a noninvasive treatment for episodic migraines may change that.
that can help ease migraine pain.
Tina Montgomery, 58, has suffered from migraines since childhood and spent years looking for something to help manage them. Doctors consider her a “chronic” sufferer in that she has more than 14 migraines a month (fewer than 14 is considered “episodic”). Prescription antidepressants, anticonvulsants, and botulinum toxin shots as preventive treatments helped a little but not enough.
A few years ago, she found some relief using a new preventive injectable medication that targets a peptide known as CGRP, combined with an oral CGRP rescue medication, ubrogepant (Ubrelvy). However, by early 2021, Ms. Montgomery’s chronic migraines were back as she faced stress from the pandemic and her role as a caregiver for her aging parents.
“I was going through so much medication. I just didn’t feel good taking so much,” she said.
Looking for relief, she read about Nerivio, a wearable migraine treatment device that uses remote electrical neuromodulation (REN). She mentioned the device to her neurologist, and he agreed she might benefit from trying it out. Today, she uses the device whenever she feels a migraine may be imminent, she said.
“It really helps me stave off migraines I feel coming on and the milder ones where I would normally hesitate to use prescription medication because [insurance] limits the number of pills they give you in a month,” she said. “I follow through with the Nerivio treatment and usually find that my migraine doesn’t fully develop or is completely gone, and I don’t get a migraine at all.”
Taking it on the arm
The device works by stimulating nerves at the back of the arm right around the triceps. “Those nerve fibers relay information to the brain stem [so it can] work its magic and use the brain’s own natural mechanisms for reducing pain,” said Brian M. Grosberg, MD, director of the Hartford Healthcare Ayer Neuroscience Institute Headache Center, West Hartford, Conn.
These mechanisms are like a bait-and-switch for the brain, said Britany Klenofsky, MD, assistant professor of neurology, Icahn School of Medicine at Mount Sinai, New York. “You’re trying to stimulate pain somewhere else [on the body] to tell the brain to protect itself and release [the neurotransmitter] serotonin,” she said. “You do this by putting the device on your arm, an area that’s away from the head where the pain is actively occurring, turning the device on, and increasing the stimulation to a nearly painful stimulus.”
This pseudo pain prompts the brain to release serotonin, the feel-good hormone along with norepinephrine and noradrenaline. The device works best when it’s used as soon as a migraine starts, so patients should hook up Nerivio within the first 20-30 minutes of onset of pain, said Dr. Grosberg, who was an investigator on the double-blind treatment study that led to FDA clearance. If patients wait too long, the device may not work.
This is why as soon as Ms. Montgomery feels a migraine aura (there are six types of migraine auras, including visual changes and muscle weakness) that occurs right before a migraine strikes, she puts the device armband on her upper arm and launches its smartphone app. Then she turns on the device for a 45-minute treatment, which begins with what she characterizes as tingling and vibration sensations on her arm. She turns up the intensity of the sensations, which are mild electric currents, until they are well-felt but not painful.
Ms. Montgomery said she can use the device and multitask since there’s no need for her to lie down or sit in a darkened room. And since it is worn on the arm, she can wear it under a shirtsleeve while working or out in public without anyone noticing. She also uses the app’s migraine diary and guided meditation to help reduce the anxiety that often accompanies her migraines.
The device is approved for adolescents and adults and can be used for both episodic and chronic migraines. From an efficacy standpoint, the device provides relief about as well as a commonly used pharmaceutical class of drugs, triptans. About 37% of people with episodic migraine achieved complete freedom from pain 2 hours after their treatment. In addition, about two-thirds of people reported pain relief after 2 hours, which is better success than people find with many prescription and nonprescription drugs.
A separate study looked at acute treatment for chronic migraine sufferers and found nearly 60% of people using the device found relief and 21% said they were pain-free after 2 hours. Almost two-thirds of those who experienced pain relief were pain-free 24 hours after the treatment.
Finding the perfect patient
There are other FDA-cleared noninvasive devices to treat migraines. One device, CEFALY, is an external trigeminal nerve stimulation device that sits on the forehead. Another device, SpringTMS, uses transcranial magnetic stimulation on the back of the head. A third option, the gammaCore Sapphire, is placed on the neck to stimulate the vagus nerve. All three have been cleared by the FDA to work as preventive and acute treatments for migraine.
Theranica, the company that developed Nerivio, is trying to boost use of the device by allowing patients to get a prescription via telehealth visits with a physician.
The company, as well as the companies behind the other neuromodulation devices, are marketing their treatments to children ages 12 and up since nonpharmacologic options are often preferable for parents, said Thomas Berk, MD, a clinical associate professor in the division of headache at NYU Langone Health in New York.
Dr. Berk said the devices could be appealing for those people who don’t want or can’t take medication, such as pregnant women or those who don’t respond well to drugs. “[They] could also be used by somebody who needs something in addition to a medication,” he said.
For now, people like Ms. Montgomery say they are happy to have another tool in their migraine arsenal. “Overall, I’m taking less medication because I haven’t had to have my Ubrelvy refilled as often as I used to,” she said. “It’s really helped me manage changes and stresses in my life.”
A version of this article first appeared on Medscape.com.
Most people avoid smartphones when they have a migraine headache, but a noninvasive treatment for episodic migraines may change that.
that can help ease migraine pain.
Tina Montgomery, 58, has suffered from migraines since childhood and spent years looking for something to help manage them. Doctors consider her a “chronic” sufferer in that she has more than 14 migraines a month (fewer than 14 is considered “episodic”). Prescription antidepressants, anticonvulsants, and botulinum toxin shots as preventive treatments helped a little but not enough.
A few years ago, she found some relief using a new preventive injectable medication that targets a peptide known as CGRP, combined with an oral CGRP rescue medication, ubrogepant (Ubrelvy). However, by early 2021, Ms. Montgomery’s chronic migraines were back as she faced stress from the pandemic and her role as a caregiver for her aging parents.
“I was going through so much medication. I just didn’t feel good taking so much,” she said.
Looking for relief, she read about Nerivio, a wearable migraine treatment device that uses remote electrical neuromodulation (REN). She mentioned the device to her neurologist, and he agreed she might benefit from trying it out. Today, she uses the device whenever she feels a migraine may be imminent, she said.
“It really helps me stave off migraines I feel coming on and the milder ones where I would normally hesitate to use prescription medication because [insurance] limits the number of pills they give you in a month,” she said. “I follow through with the Nerivio treatment and usually find that my migraine doesn’t fully develop or is completely gone, and I don’t get a migraine at all.”
Taking it on the arm
The device works by stimulating nerves at the back of the arm right around the triceps. “Those nerve fibers relay information to the brain stem [so it can] work its magic and use the brain’s own natural mechanisms for reducing pain,” said Brian M. Grosberg, MD, director of the Hartford Healthcare Ayer Neuroscience Institute Headache Center, West Hartford, Conn.
These mechanisms are like a bait-and-switch for the brain, said Britany Klenofsky, MD, assistant professor of neurology, Icahn School of Medicine at Mount Sinai, New York. “You’re trying to stimulate pain somewhere else [on the body] to tell the brain to protect itself and release [the neurotransmitter] serotonin,” she said. “You do this by putting the device on your arm, an area that’s away from the head where the pain is actively occurring, turning the device on, and increasing the stimulation to a nearly painful stimulus.”
This pseudo pain prompts the brain to release serotonin, the feel-good hormone along with norepinephrine and noradrenaline. The device works best when it’s used as soon as a migraine starts, so patients should hook up Nerivio within the first 20-30 minutes of onset of pain, said Dr. Grosberg, who was an investigator on the double-blind treatment study that led to FDA clearance. If patients wait too long, the device may not work.
This is why as soon as Ms. Montgomery feels a migraine aura (there are six types of migraine auras, including visual changes and muscle weakness) that occurs right before a migraine strikes, she puts the device armband on her upper arm and launches its smartphone app. Then she turns on the device for a 45-minute treatment, which begins with what she characterizes as tingling and vibration sensations on her arm. She turns up the intensity of the sensations, which are mild electric currents, until they are well-felt but not painful.
Ms. Montgomery said she can use the device and multitask since there’s no need for her to lie down or sit in a darkened room. And since it is worn on the arm, she can wear it under a shirtsleeve while working or out in public without anyone noticing. She also uses the app’s migraine diary and guided meditation to help reduce the anxiety that often accompanies her migraines.
The device is approved for adolescents and adults and can be used for both episodic and chronic migraines. From an efficacy standpoint, the device provides relief about as well as a commonly used pharmaceutical class of drugs, triptans. About 37% of people with episodic migraine achieved complete freedom from pain 2 hours after their treatment. In addition, about two-thirds of people reported pain relief after 2 hours, which is better success than people find with many prescription and nonprescription drugs.
A separate study looked at acute treatment for chronic migraine sufferers and found nearly 60% of people using the device found relief and 21% said they were pain-free after 2 hours. Almost two-thirds of those who experienced pain relief were pain-free 24 hours after the treatment.
Finding the perfect patient
There are other FDA-cleared noninvasive devices to treat migraines. One device, CEFALY, is an external trigeminal nerve stimulation device that sits on the forehead. Another device, SpringTMS, uses transcranial magnetic stimulation on the back of the head. A third option, the gammaCore Sapphire, is placed on the neck to stimulate the vagus nerve. All three have been cleared by the FDA to work as preventive and acute treatments for migraine.
Theranica, the company that developed Nerivio, is trying to boost use of the device by allowing patients to get a prescription via telehealth visits with a physician.
The company, as well as the companies behind the other neuromodulation devices, are marketing their treatments to children ages 12 and up since nonpharmacologic options are often preferable for parents, said Thomas Berk, MD, a clinical associate professor in the division of headache at NYU Langone Health in New York.
Dr. Berk said the devices could be appealing for those people who don’t want or can’t take medication, such as pregnant women or those who don’t respond well to drugs. “[They] could also be used by somebody who needs something in addition to a medication,” he said.
For now, people like Ms. Montgomery say they are happy to have another tool in their migraine arsenal. “Overall, I’m taking less medication because I haven’t had to have my Ubrelvy refilled as often as I used to,” she said. “It’s really helped me manage changes and stresses in my life.”
A version of this article first appeared on Medscape.com.










