User login
The new obesity breakthrough drugs
This article was originally published December 10 on Medscape editor-in-chief Eric Topol’s Substack ”Ground Truths.”
– achieving a substantial amount of weight loss without serious side effects. Many attempts to get there now fill a graveyard of failed drugs, such as fen-phen in the 1990s when a single small study of this drug combination in 121 people unleashed millions of prescriptions, some leading to serious heart valve lesions that resulted in withdrawal of the drug in 1995. The drug rimonabant, an endocannabinoid receptor blocker (think of blocking the munchies after marijuana) looked encouraging in randomized trials. However, subsequently, in a trial that I led of nearly 19,000 participants in 42 countries around the world, there was a significant excess of depression, neuropsychiatric side-effects and suicidal ideation which spelled the end of that drug’s life.
In the United States, where there had not been an antiobesity drug approved by the Food and Drug Administration since 2014, Wegovy (semaglutide), a once-weekly injection was approved in June 2021. The same drug, at a lower dose, is known as Ozempic (as in O-O-O, Ozempic, the ubiquitous commercial that you undoubtedly hear and see on TV) and had already been approved in January 2020 for improving glucose regulation in diabetes. The next drug on fast track at FDA to be imminently approved is tirzepatide (Mounjaro) following its approval for diabetes in May 2022. It is noteworthy that the discovery of these drugs for weight loss was serendipitous: they were being developed for improving glucose regulation and unexpectedly were found to achieve significant weight reduction.
Both semaglutide and tirzepatide underwent randomized, placebo-controlled trials for obesity, with marked reduction of weight as shown below. Tirzepatide at dose of 10-15 mg per week achieved greater than 20% body weight reduction. Semaglutide at a dose of 2.4 mg achieved about 17% reduction. These per cent changes in body weight are 7-9 fold more than seen with placebo (2%-3% reduction). Note: these levels of percent body-weight reduction resemble what is typically achieved with the different types of bariatric surgery, such as gastric bypass.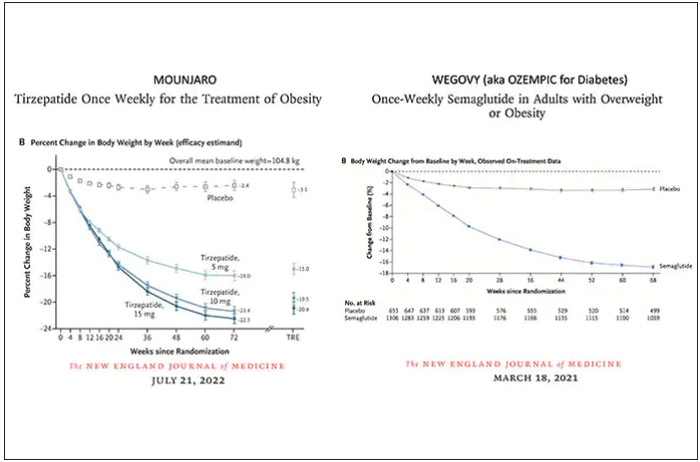
Another way to present the data for the two trials is shown here, with an edge for tirzepatide at high (10-15 mg) doses, extending to greater than 25% body-weight reduction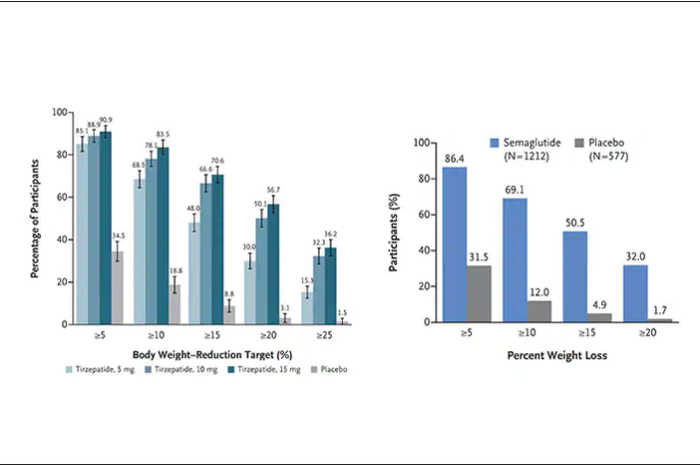
The results with semaglutide were extended to teens in a randomized trial (as shown below), and a similar trial with tirzepatide is in progress.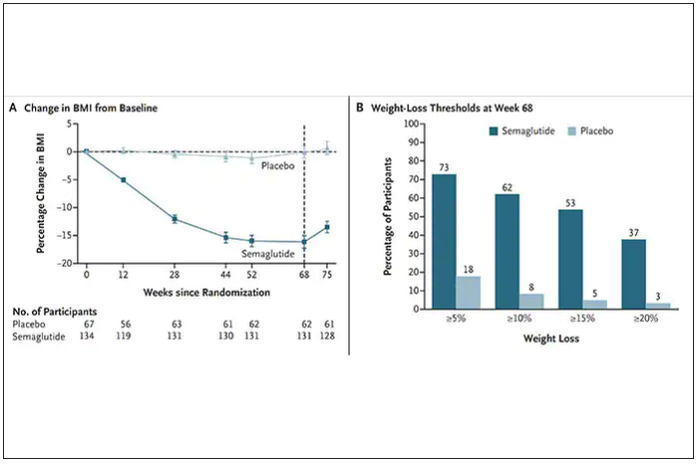
How do these drugs work?
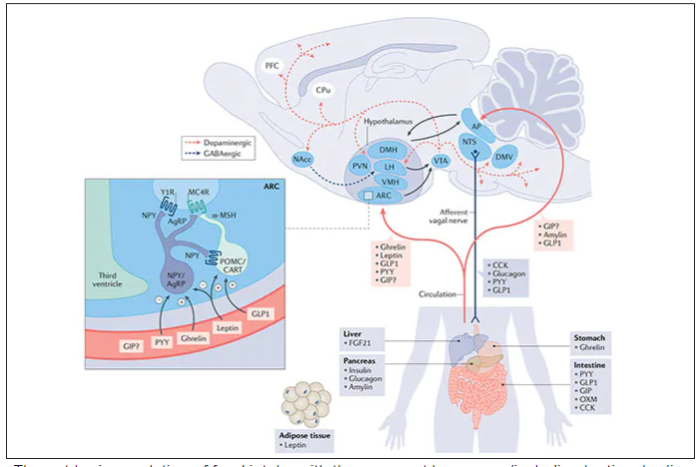
These are peptides in the class of incretins, mimicking gut hormones that are secreted after food intake which stimulate insulin secretion.
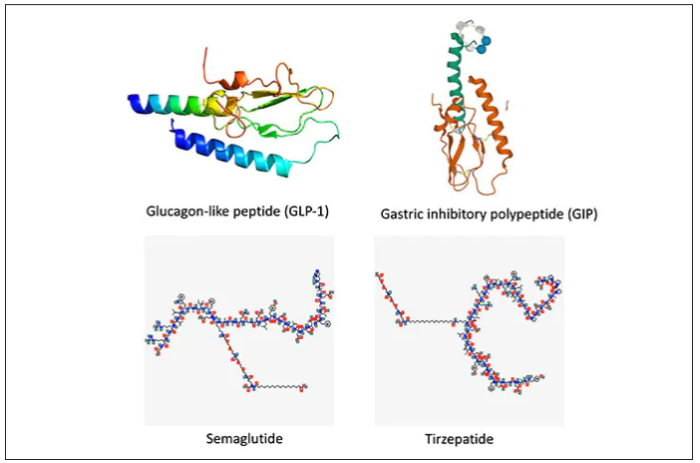
These two drugs have in common long half-lives (about 5 days), which affords once-weekly dosing, but have different mechanisms of action. Semaglutide activates (an agonist) the glucagonlike peptide–1 receptor, while tirzepatide is in a new class of dual agonists: It activates (mimics) both the GLP-1 receptor and GIP receptors (Gastric inhibit polypeptide is also known as glucose-dependent insulinotropic polypeptide.) The potency of activation for tirzepatide is fivefold more for GIPR than GLP1. As seen below, there are body wide effects that include the brain, liver, pancreas, stomach, intestine, skeletal muscle and fat tissue. While their mode of action is somewhat different, their clinical effects are overlapping, which include enhancing satiety, delaying gastric emptying, increasing insulin and its sensitivity, decreasing glucagon, and, of course, reducing high glucose levels. The overlap extends to side effects of nausea, vomiting, abdominal pain, constipation and diarrhea. Yet only 4%-6% of participants discontinued the drug in these trials, mostly owing to these GI side effects (and 1%-2% in the placebo group discontinued the study drug for the same reasons).
In randomized trials among people with type 2 diabetes, the drugs achieved hemoglobin A1c reduction of at least an absolute 2 percentage points which led to their FDA approvals (For semaglutide in January 2020, and for tirzepatide in May 2022). The edge that tirzepatide has exhibited for weight-loss reduction may be related to its dual agonist role, but the enhancement via GIP receptor activation is not fully resolved (as seen below with GIP? designation). The Amgen drug in development (AMG-133) has a marked weight loss effect but inhibits GIP rather than mimics it, clouding our precise understanding of the mechanism.
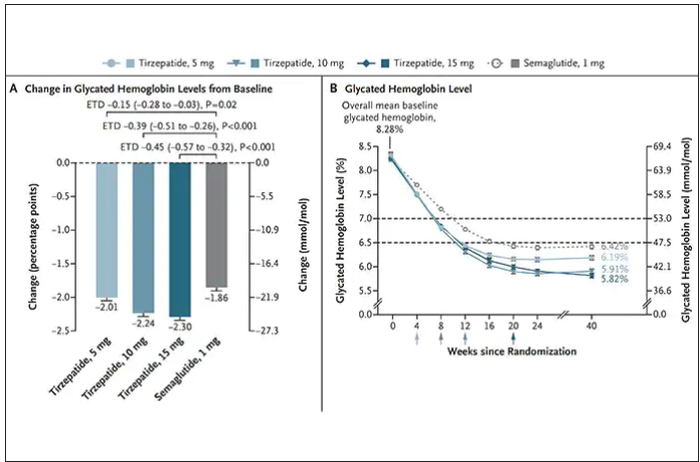
Nevertheless, when the two drugs were directly compared in a randomized trial for improving glucose regulation, tirzepatide was superior to semaglutide, as shown below. Of note, both drugs achieved very favorable effects on lipids, reducing triglycerides and LDL cholesterol and raising HDL cholesterol, along with reduction of blood pressure, an outgrowth of the indirect effect of weight reduction and direct metabolic effects of the drugs.
While there has been a concern about other side effects besides the GI ones noted above, review of all the trials to date in these classes of medication do not reinforce a risk of acute pancreatitis. Other rare side effects that have been noted with these drugs include allergic reactions, gallstones (which can occur with a large amount of weight loss), and potential of medullary thyroid cancer (so far only documented in rats, not people), which is why they are contraindicated in people with Type 2 multiple endocrine neoplasia syndrome.
How they are given and practical considerations
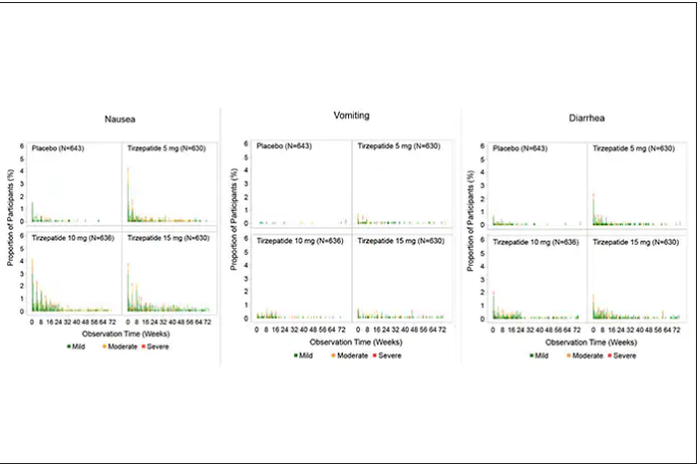
For semaglutide, which has FDA approval, the indication is a body mass index of 30 kg/m2 or greater than 27 and a weight-related medical condition (such as hypertension, hypercholesterolemia, or diabetes). To reduce the GI side effects, which mainly occur in the early dose escalation period, semaglutide is given in increasing doses by a prefilled pen by self-injection under the skin (abdomen, thigh, or arm) starting at 0.25 mg for a month and gradual increases each month reaching the maximum dose of 2.4 mg at month 5. The FDA label for dosing of tirzepatide has not been provided yet but in the weight loss trial there was a similar dose escalation from 2.5 mg up to 15 mg by month 5. The escalation is essential to reduce the frequent GI side effects, such as seen below in the tirzepatide trial.
Semaglutide is very expensive, about $1,500 per month, and not covered by Medicare. There are manufacturer starter coupons from Novo Nordisk, but that is just for the first month. These drugs have to be taken for a year to 18 months to have their full effect and without changes in lifestyle that are durable, it is likely that weight will be regained after stopping them.
What does this mean?
More than 650 million adults and 340 million children aged 5-18 are obese. The global obesity epidemic has been relentless, worsening each year, and a driver of “diabesity,” the combined dual epidemic. We now have a breakthrough class of drugs that can achieve profound weight loss equivalent to bariatric surgery, along with the side benefits of reducing cardiovascular risk factors (hypertension and hyperlipidemia), improving glucose regulation, reversing fatty liver, and the many detrimental long-term effects of obesity such as osteoarthritis and various cancers. That, in itself, is remarkable. Revolutionary.
But the downsides are also obvious. Self-injections, even though they are once a week, are not palatable for many. We have seen far more of these injectables in recent years such as the proprotein convertase subtilisin/kexin type 9 inhibitors for hypercholesterolemia or the tumor necrosis factor blockers for autoimmune conditions. That still will not make them a popular item for such an enormous population of potential users.
That brings me to Rybelsus, the oral form of semaglutide, which is approved for glucose regulation improvement but not obesity. It effects for weight loss have been modest, compared with Wegovy (5 to 8 pounds for the 7- and 14-mg dose, respectively). But the potential for the very high efficacy of an injectable to be achievable via a pill represents an important path going forward—it could help markedly reduce the cost and uptake.
The problem of discontinuation of the drugs is big, since there are limited data and the likelihood is that the weight will be regained unless there are substantial changes in lifestyle. We know how hard it is to durably achieve such changes, along with the undesirability (and uncertainty with respect to unknown side effects) of having to take injectable drugs for many years, no less the cost of doing that.
The cost of these drugs will clearly and profoundly exacerbate inequities, since they are eminently affordable by the rich, but the need is extreme among the indigent. We’ve already seen celebrities take Wegovy for weight loss who are not obese, a window into how these drugs can and will be used without supportive data. As one physician recently observed, “Other than Viagra and Botox, I’ve seen no other medication so quickly become part of modern culture’s social vernacular.” Already there are concerns that such use is preventing access to the drugs for those who qualify and need them.
There are multiple agents in the class under development which should help increase competition and reduce cost, but they will remain expensive. There is private insurance reimbursement, often with a significant copay, for people who tightly fit the inclusion criteria. Eventual coverage by Medicare will markedly expand their use, and we can expect cost-effectiveness studies to be published showing how much saving there is for the drugs compared with bariatric surgery or not achieving the weight loss. But that doesn’t change the cost at the societal level. Even as we’ve seen with generics, which will ultimately be available, the alleviation of the cost problem isn’t what we’d hoped.
This is not unlike the recent triumphs of gene therapy, as in $3.5 million for a cure of hemophilia that just got FDA approval, but instead of a rare disease we are talking about the most common medical condition in the world. We finally get across the long sought after (what many would qualify as miraculous) goal line, but the economics collide with the uptake and real benefit.
These concerns can’t be put aside in the health inequity-laden world we live in, that will unquestionably be exacerbated. However, we cannot miss that this represents one of the most important, biggest medical breakthroughs in history. This may signify the end or marked reduction in the need for bariatric surgery. These drugs will likely become some of the most prescribed of all medications in the upcoming years. While there are many drawbacks, we shouldn’t miss such an extraordinary advance in medicine – the first real, potent and safe treatment of obesity.
Thanks for reading Ground Truths. I hope you will share these posts and subscribe, to be sure you don’t miss them.
Dr. Topol is director, Scripps Translational Science Institute; executive vice president and professor of molecular medicine at The Scripps Research Institute and senior consultant, division of cardiovascular diseases, at the Scripps Clinic, both in La Jolla, Calif. He disclosed relevant financial relationships with Dexcom, Illumina, Molecular Stethoscope, Walgreens, Quest Diagnostics, MyoKardia, and National Institutes of Health. A version of this article first appeared on Medscape.com.
This article was originally published December 10 on Medscape editor-in-chief Eric Topol’s Substack ”Ground Truths.”
– achieving a substantial amount of weight loss without serious side effects. Many attempts to get there now fill a graveyard of failed drugs, such as fen-phen in the 1990s when a single small study of this drug combination in 121 people unleashed millions of prescriptions, some leading to serious heart valve lesions that resulted in withdrawal of the drug in 1995. The drug rimonabant, an endocannabinoid receptor blocker (think of blocking the munchies after marijuana) looked encouraging in randomized trials. However, subsequently, in a trial that I led of nearly 19,000 participants in 42 countries around the world, there was a significant excess of depression, neuropsychiatric side-effects and suicidal ideation which spelled the end of that drug’s life.
In the United States, where there had not been an antiobesity drug approved by the Food and Drug Administration since 2014, Wegovy (semaglutide), a once-weekly injection was approved in June 2021. The same drug, at a lower dose, is known as Ozempic (as in O-O-O, Ozempic, the ubiquitous commercial that you undoubtedly hear and see on TV) and had already been approved in January 2020 for improving glucose regulation in diabetes. The next drug on fast track at FDA to be imminently approved is tirzepatide (Mounjaro) following its approval for diabetes in May 2022. It is noteworthy that the discovery of these drugs for weight loss was serendipitous: they were being developed for improving glucose regulation and unexpectedly were found to achieve significant weight reduction.
Both semaglutide and tirzepatide underwent randomized, placebo-controlled trials for obesity, with marked reduction of weight as shown below. Tirzepatide at dose of 10-15 mg per week achieved greater than 20% body weight reduction. Semaglutide at a dose of 2.4 mg achieved about 17% reduction. These per cent changes in body weight are 7-9 fold more than seen with placebo (2%-3% reduction). Note: these levels of percent body-weight reduction resemble what is typically achieved with the different types of bariatric surgery, such as gastric bypass.
Another way to present the data for the two trials is shown here, with an edge for tirzepatide at high (10-15 mg) doses, extending to greater than 25% body-weight reduction
The results with semaglutide were extended to teens in a randomized trial (as shown below), and a similar trial with tirzepatide is in progress.
How do these drugs work?
These are peptides in the class of incretins, mimicking gut hormones that are secreted after food intake which stimulate insulin secretion.

These two drugs have in common long half-lives (about 5 days), which affords once-weekly dosing, but have different mechanisms of action. Semaglutide activates (an agonist) the glucagonlike peptide–1 receptor, while tirzepatide is in a new class of dual agonists: It activates (mimics) both the GLP-1 receptor and GIP receptors (Gastric inhibit polypeptide is also known as glucose-dependent insulinotropic polypeptide.) The potency of activation for tirzepatide is fivefold more for GIPR than GLP1. As seen below, there are body wide effects that include the brain, liver, pancreas, stomach, intestine, skeletal muscle and fat tissue. While their mode of action is somewhat different, their clinical effects are overlapping, which include enhancing satiety, delaying gastric emptying, increasing insulin and its sensitivity, decreasing glucagon, and, of course, reducing high glucose levels. The overlap extends to side effects of nausea, vomiting, abdominal pain, constipation and diarrhea. Yet only 4%-6% of participants discontinued the drug in these trials, mostly owing to these GI side effects (and 1%-2% in the placebo group discontinued the study drug for the same reasons).
In randomized trials among people with type 2 diabetes, the drugs achieved hemoglobin A1c reduction of at least an absolute 2 percentage points which led to their FDA approvals (For semaglutide in January 2020, and for tirzepatide in May 2022). The edge that tirzepatide has exhibited for weight-loss reduction may be related to its dual agonist role, but the enhancement via GIP receptor activation is not fully resolved (as seen below with GIP? designation). The Amgen drug in development (AMG-133) has a marked weight loss effect but inhibits GIP rather than mimics it, clouding our precise understanding of the mechanism.

Nevertheless, when the two drugs were directly compared in a randomized trial for improving glucose regulation, tirzepatide was superior to semaglutide, as shown below. Of note, both drugs achieved very favorable effects on lipids, reducing triglycerides and LDL cholesterol and raising HDL cholesterol, along with reduction of blood pressure, an outgrowth of the indirect effect of weight reduction and direct metabolic effects of the drugs.

While there has been a concern about other side effects besides the GI ones noted above, review of all the trials to date in these classes of medication do not reinforce a risk of acute pancreatitis. Other rare side effects that have been noted with these drugs include allergic reactions, gallstones (which can occur with a large amount of weight loss), and potential of medullary thyroid cancer (so far only documented in rats, not people), which is why they are contraindicated in people with Type 2 multiple endocrine neoplasia syndrome.
How they are given and practical considerations
For semaglutide, which has FDA approval, the indication is a body mass index of 30 kg/m2 or greater than 27 and a weight-related medical condition (such as hypertension, hypercholesterolemia, or diabetes). To reduce the GI side effects, which mainly occur in the early dose escalation period, semaglutide is given in increasing doses by a prefilled pen by self-injection under the skin (abdomen, thigh, or arm) starting at 0.25 mg for a month and gradual increases each month reaching the maximum dose of 2.4 mg at month 5. The FDA label for dosing of tirzepatide has not been provided yet but in the weight loss trial there was a similar dose escalation from 2.5 mg up to 15 mg by month 5. The escalation is essential to reduce the frequent GI side effects, such as seen below in the tirzepatide trial.

Semaglutide is very expensive, about $1,500 per month, and not covered by Medicare. There are manufacturer starter coupons from Novo Nordisk, but that is just for the first month. These drugs have to be taken for a year to 18 months to have their full effect and without changes in lifestyle that are durable, it is likely that weight will be regained after stopping them.
What does this mean?
More than 650 million adults and 340 million children aged 5-18 are obese. The global obesity epidemic has been relentless, worsening each year, and a driver of “diabesity,” the combined dual epidemic. We now have a breakthrough class of drugs that can achieve profound weight loss equivalent to bariatric surgery, along with the side benefits of reducing cardiovascular risk factors (hypertension and hyperlipidemia), improving glucose regulation, reversing fatty liver, and the many detrimental long-term effects of obesity such as osteoarthritis and various cancers. That, in itself, is remarkable. Revolutionary.
But the downsides are also obvious. Self-injections, even though they are once a week, are not palatable for many. We have seen far more of these injectables in recent years such as the proprotein convertase subtilisin/kexin type 9 inhibitors for hypercholesterolemia or the tumor necrosis factor blockers for autoimmune conditions. That still will not make them a popular item for such an enormous population of potential users.
That brings me to Rybelsus, the oral form of semaglutide, which is approved for glucose regulation improvement but not obesity. It effects for weight loss have been modest, compared with Wegovy (5 to 8 pounds for the 7- and 14-mg dose, respectively). But the potential for the very high efficacy of an injectable to be achievable via a pill represents an important path going forward—it could help markedly reduce the cost and uptake.
The problem of discontinuation of the drugs is big, since there are limited data and the likelihood is that the weight will be regained unless there are substantial changes in lifestyle. We know how hard it is to durably achieve such changes, along with the undesirability (and uncertainty with respect to unknown side effects) of having to take injectable drugs for many years, no less the cost of doing that.
The cost of these drugs will clearly and profoundly exacerbate inequities, since they are eminently affordable by the rich, but the need is extreme among the indigent. We’ve already seen celebrities take Wegovy for weight loss who are not obese, a window into how these drugs can and will be used without supportive data. As one physician recently observed, “Other than Viagra and Botox, I’ve seen no other medication so quickly become part of modern culture’s social vernacular.” Already there are concerns that such use is preventing access to the drugs for those who qualify and need them.
There are multiple agents in the class under development which should help increase competition and reduce cost, but they will remain expensive. There is private insurance reimbursement, often with a significant copay, for people who tightly fit the inclusion criteria. Eventual coverage by Medicare will markedly expand their use, and we can expect cost-effectiveness studies to be published showing how much saving there is for the drugs compared with bariatric surgery or not achieving the weight loss. But that doesn’t change the cost at the societal level. Even as we’ve seen with generics, which will ultimately be available, the alleviation of the cost problem isn’t what we’d hoped.
This is not unlike the recent triumphs of gene therapy, as in $3.5 million for a cure of hemophilia that just got FDA approval, but instead of a rare disease we are talking about the most common medical condition in the world. We finally get across the long sought after (what many would qualify as miraculous) goal line, but the economics collide with the uptake and real benefit.
These concerns can’t be put aside in the health inequity-laden world we live in, that will unquestionably be exacerbated. However, we cannot miss that this represents one of the most important, biggest medical breakthroughs in history. This may signify the end or marked reduction in the need for bariatric surgery. These drugs will likely become some of the most prescribed of all medications in the upcoming years. While there are many drawbacks, we shouldn’t miss such an extraordinary advance in medicine – the first real, potent and safe treatment of obesity.
Thanks for reading Ground Truths. I hope you will share these posts and subscribe, to be sure you don’t miss them.
Dr. Topol is director, Scripps Translational Science Institute; executive vice president and professor of molecular medicine at The Scripps Research Institute and senior consultant, division of cardiovascular diseases, at the Scripps Clinic, both in La Jolla, Calif. He disclosed relevant financial relationships with Dexcom, Illumina, Molecular Stethoscope, Walgreens, Quest Diagnostics, MyoKardia, and National Institutes of Health. A version of this article first appeared on Medscape.com.
This article was originally published December 10 on Medscape editor-in-chief Eric Topol’s Substack ”Ground Truths.”
– achieving a substantial amount of weight loss without serious side effects. Many attempts to get there now fill a graveyard of failed drugs, such as fen-phen in the 1990s when a single small study of this drug combination in 121 people unleashed millions of prescriptions, some leading to serious heart valve lesions that resulted in withdrawal of the drug in 1995. The drug rimonabant, an endocannabinoid receptor blocker (think of blocking the munchies after marijuana) looked encouraging in randomized trials. However, subsequently, in a trial that I led of nearly 19,000 participants in 42 countries around the world, there was a significant excess of depression, neuropsychiatric side-effects and suicidal ideation which spelled the end of that drug’s life.
In the United States, where there had not been an antiobesity drug approved by the Food and Drug Administration since 2014, Wegovy (semaglutide), a once-weekly injection was approved in June 2021. The same drug, at a lower dose, is known as Ozempic (as in O-O-O, Ozempic, the ubiquitous commercial that you undoubtedly hear and see on TV) and had already been approved in January 2020 for improving glucose regulation in diabetes. The next drug on fast track at FDA to be imminently approved is tirzepatide (Mounjaro) following its approval for diabetes in May 2022. It is noteworthy that the discovery of these drugs for weight loss was serendipitous: they were being developed for improving glucose regulation and unexpectedly were found to achieve significant weight reduction.
Both semaglutide and tirzepatide underwent randomized, placebo-controlled trials for obesity, with marked reduction of weight as shown below. Tirzepatide at dose of 10-15 mg per week achieved greater than 20% body weight reduction. Semaglutide at a dose of 2.4 mg achieved about 17% reduction. These per cent changes in body weight are 7-9 fold more than seen with placebo (2%-3% reduction). Note: these levels of percent body-weight reduction resemble what is typically achieved with the different types of bariatric surgery, such as gastric bypass.
Another way to present the data for the two trials is shown here, with an edge for tirzepatide at high (10-15 mg) doses, extending to greater than 25% body-weight reduction
The results with semaglutide were extended to teens in a randomized trial (as shown below), and a similar trial with tirzepatide is in progress.
How do these drugs work?
These are peptides in the class of incretins, mimicking gut hormones that are secreted after food intake which stimulate insulin secretion.

These two drugs have in common long half-lives (about 5 days), which affords once-weekly dosing, but have different mechanisms of action. Semaglutide activates (an agonist) the glucagonlike peptide–1 receptor, while tirzepatide is in a new class of dual agonists: It activates (mimics) both the GLP-1 receptor and GIP receptors (Gastric inhibit polypeptide is also known as glucose-dependent insulinotropic polypeptide.) The potency of activation for tirzepatide is fivefold more for GIPR than GLP1. As seen below, there are body wide effects that include the brain, liver, pancreas, stomach, intestine, skeletal muscle and fat tissue. While their mode of action is somewhat different, their clinical effects are overlapping, which include enhancing satiety, delaying gastric emptying, increasing insulin and its sensitivity, decreasing glucagon, and, of course, reducing high glucose levels. The overlap extends to side effects of nausea, vomiting, abdominal pain, constipation and diarrhea. Yet only 4%-6% of participants discontinued the drug in these trials, mostly owing to these GI side effects (and 1%-2% in the placebo group discontinued the study drug for the same reasons).
In randomized trials among people with type 2 diabetes, the drugs achieved hemoglobin A1c reduction of at least an absolute 2 percentage points which led to their FDA approvals (For semaglutide in January 2020, and for tirzepatide in May 2022). The edge that tirzepatide has exhibited for weight-loss reduction may be related to its dual agonist role, but the enhancement via GIP receptor activation is not fully resolved (as seen below with GIP? designation). The Amgen drug in development (AMG-133) has a marked weight loss effect but inhibits GIP rather than mimics it, clouding our precise understanding of the mechanism.

Nevertheless, when the two drugs were directly compared in a randomized trial for improving glucose regulation, tirzepatide was superior to semaglutide, as shown below. Of note, both drugs achieved very favorable effects on lipids, reducing triglycerides and LDL cholesterol and raising HDL cholesterol, along with reduction of blood pressure, an outgrowth of the indirect effect of weight reduction and direct metabolic effects of the drugs.

While there has been a concern about other side effects besides the GI ones noted above, review of all the trials to date in these classes of medication do not reinforce a risk of acute pancreatitis. Other rare side effects that have been noted with these drugs include allergic reactions, gallstones (which can occur with a large amount of weight loss), and potential of medullary thyroid cancer (so far only documented in rats, not people), which is why they are contraindicated in people with Type 2 multiple endocrine neoplasia syndrome.
How they are given and practical considerations
For semaglutide, which has FDA approval, the indication is a body mass index of 30 kg/m2 or greater than 27 and a weight-related medical condition (such as hypertension, hypercholesterolemia, or diabetes). To reduce the GI side effects, which mainly occur in the early dose escalation period, semaglutide is given in increasing doses by a prefilled pen by self-injection under the skin (abdomen, thigh, or arm) starting at 0.25 mg for a month and gradual increases each month reaching the maximum dose of 2.4 mg at month 5. The FDA label for dosing of tirzepatide has not been provided yet but in the weight loss trial there was a similar dose escalation from 2.5 mg up to 15 mg by month 5. The escalation is essential to reduce the frequent GI side effects, such as seen below in the tirzepatide trial.

Semaglutide is very expensive, about $1,500 per month, and not covered by Medicare. There are manufacturer starter coupons from Novo Nordisk, but that is just for the first month. These drugs have to be taken for a year to 18 months to have their full effect and without changes in lifestyle that are durable, it is likely that weight will be regained after stopping them.
What does this mean?
More than 650 million adults and 340 million children aged 5-18 are obese. The global obesity epidemic has been relentless, worsening each year, and a driver of “diabesity,” the combined dual epidemic. We now have a breakthrough class of drugs that can achieve profound weight loss equivalent to bariatric surgery, along with the side benefits of reducing cardiovascular risk factors (hypertension and hyperlipidemia), improving glucose regulation, reversing fatty liver, and the many detrimental long-term effects of obesity such as osteoarthritis and various cancers. That, in itself, is remarkable. Revolutionary.
But the downsides are also obvious. Self-injections, even though they are once a week, are not palatable for many. We have seen far more of these injectables in recent years such as the proprotein convertase subtilisin/kexin type 9 inhibitors for hypercholesterolemia or the tumor necrosis factor blockers for autoimmune conditions. That still will not make them a popular item for such an enormous population of potential users.
That brings me to Rybelsus, the oral form of semaglutide, which is approved for glucose regulation improvement but not obesity. It effects for weight loss have been modest, compared with Wegovy (5 to 8 pounds for the 7- and 14-mg dose, respectively). But the potential for the very high efficacy of an injectable to be achievable via a pill represents an important path going forward—it could help markedly reduce the cost and uptake.
The problem of discontinuation of the drugs is big, since there are limited data and the likelihood is that the weight will be regained unless there are substantial changes in lifestyle. We know how hard it is to durably achieve such changes, along with the undesirability (and uncertainty with respect to unknown side effects) of having to take injectable drugs for many years, no less the cost of doing that.
The cost of these drugs will clearly and profoundly exacerbate inequities, since they are eminently affordable by the rich, but the need is extreme among the indigent. We’ve already seen celebrities take Wegovy for weight loss who are not obese, a window into how these drugs can and will be used without supportive data. As one physician recently observed, “Other than Viagra and Botox, I’ve seen no other medication so quickly become part of modern culture’s social vernacular.” Already there are concerns that such use is preventing access to the drugs for those who qualify and need them.
There are multiple agents in the class under development which should help increase competition and reduce cost, but they will remain expensive. There is private insurance reimbursement, often with a significant copay, for people who tightly fit the inclusion criteria. Eventual coverage by Medicare will markedly expand their use, and we can expect cost-effectiveness studies to be published showing how much saving there is for the drugs compared with bariatric surgery or not achieving the weight loss. But that doesn’t change the cost at the societal level. Even as we’ve seen with generics, which will ultimately be available, the alleviation of the cost problem isn’t what we’d hoped.
This is not unlike the recent triumphs of gene therapy, as in $3.5 million for a cure of hemophilia that just got FDA approval, but instead of a rare disease we are talking about the most common medical condition in the world. We finally get across the long sought after (what many would qualify as miraculous) goal line, but the economics collide with the uptake and real benefit.
These concerns can’t be put aside in the health inequity-laden world we live in, that will unquestionably be exacerbated. However, we cannot miss that this represents one of the most important, biggest medical breakthroughs in history. This may signify the end or marked reduction in the need for bariatric surgery. These drugs will likely become some of the most prescribed of all medications in the upcoming years. While there are many drawbacks, we shouldn’t miss such an extraordinary advance in medicine – the first real, potent and safe treatment of obesity.
Thanks for reading Ground Truths. I hope you will share these posts and subscribe, to be sure you don’t miss them.
Dr. Topol is director, Scripps Translational Science Institute; executive vice president and professor of molecular medicine at The Scripps Research Institute and senior consultant, division of cardiovascular diseases, at the Scripps Clinic, both in La Jolla, Calif. He disclosed relevant financial relationships with Dexcom, Illumina, Molecular Stethoscope, Walgreens, Quest Diagnostics, MyoKardia, and National Institutes of Health. A version of this article first appeared on Medscape.com.
Does paying people to lose weight work?
It denies the impact of the thousands of genes and dozens of hormones involved in our individual levels of hunger, cravings, and fullness. It denies the torrential current of our ultraprocessed and calorific food environment. It denies the constant push of food advertising and the role food has taken on as the star of even the smallest of events and celebrations. It denies the role of food as a seminal pleasure in a world that, even for those possessing great degrees of privilege is challenging, let alone for those facing tremendous and varied difficulties. And of course, it upholds the hateful notion that, if people just wanted it badly enough, they’d manage their weight, the corollary of which is that people with obesity are unmotivated and lazy.
Yet the notion that, if people want it badly enough, they’d make it happen, is incredibly commonplace. It’s so commonplace that NBC aired their prime-time televised reality show The Biggest Loser from 2004 through 2016, featuring people with obesity competing for a $500,000 prize during a 30-week–long orgy of fat-shaming, victim-blaming, hugely restrictive eating, and injury. It’s also so commonplace that studies are still being conducted exploring the impact of paying people to lose weight.
The most recent of these – “Effectiveness of Goal-Directed and Outcome-Based Financial Incentives for Weight Loss in Primary Care Patients With Obesity Living in Socioeconomically Disadvantaged Neighborhoods: A Randomized Clinical Trial” – examined the effects of randomly assigning participants whose annual household incomes were less than $40,000 to either a free year of Weight Watchers and the provisions of basic weight loss advice (exercise, track your food, eat healthfully, et cetera) or to an incentivized program that would see them earning up to $750 over 6 months, with dollars being awarded for such things as attendance in education sessions, keeping a food diary, recording their weight, and obtaining a certain amount of exercise or for weight loss.
Resultswise – though you might not have gathered it from the conclusion of the paper, which states that incentives were more effective at 12 months – the average incentivized participant lost roughly 6 pounds more than those given only resources. It should also be mentioned that over half of the incentivized group did not complete the study.
That these sorts of studies are still being conducted is depressing. Medicine and academia need to actively stop promoting harmful stereotypes when it comes to the genesis of a chronic noncommunicable disease that is not caused by a lack of desire, needing the right incentive, but is rather caused by the interaction of millions of years of evolution during extreme dietary insecurity with a modern-day food environment and culture that constantly offers, provides, and encourages consumption. This is especially true now that there are effective antiobesity medications whose success underwrites the notion that it’s physiology, rather than a lack of wanting it enough, that gets in the way of sustained success.
A version of this article first appeared on Medscape.com.
It denies the impact of the thousands of genes and dozens of hormones involved in our individual levels of hunger, cravings, and fullness. It denies the torrential current of our ultraprocessed and calorific food environment. It denies the constant push of food advertising and the role food has taken on as the star of even the smallest of events and celebrations. It denies the role of food as a seminal pleasure in a world that, even for those possessing great degrees of privilege is challenging, let alone for those facing tremendous and varied difficulties. And of course, it upholds the hateful notion that, if people just wanted it badly enough, they’d manage their weight, the corollary of which is that people with obesity are unmotivated and lazy.
Yet the notion that, if people want it badly enough, they’d make it happen, is incredibly commonplace. It’s so commonplace that NBC aired their prime-time televised reality show The Biggest Loser from 2004 through 2016, featuring people with obesity competing for a $500,000 prize during a 30-week–long orgy of fat-shaming, victim-blaming, hugely restrictive eating, and injury. It’s also so commonplace that studies are still being conducted exploring the impact of paying people to lose weight.
The most recent of these – “Effectiveness of Goal-Directed and Outcome-Based Financial Incentives for Weight Loss in Primary Care Patients With Obesity Living in Socioeconomically Disadvantaged Neighborhoods: A Randomized Clinical Trial” – examined the effects of randomly assigning participants whose annual household incomes were less than $40,000 to either a free year of Weight Watchers and the provisions of basic weight loss advice (exercise, track your food, eat healthfully, et cetera) or to an incentivized program that would see them earning up to $750 over 6 months, with dollars being awarded for such things as attendance in education sessions, keeping a food diary, recording their weight, and obtaining a certain amount of exercise or for weight loss.
Resultswise – though you might not have gathered it from the conclusion of the paper, which states that incentives were more effective at 12 months – the average incentivized participant lost roughly 6 pounds more than those given only resources. It should also be mentioned that over half of the incentivized group did not complete the study.
That these sorts of studies are still being conducted is depressing. Medicine and academia need to actively stop promoting harmful stereotypes when it comes to the genesis of a chronic noncommunicable disease that is not caused by a lack of desire, needing the right incentive, but is rather caused by the interaction of millions of years of evolution during extreme dietary insecurity with a modern-day food environment and culture that constantly offers, provides, and encourages consumption. This is especially true now that there are effective antiobesity medications whose success underwrites the notion that it’s physiology, rather than a lack of wanting it enough, that gets in the way of sustained success.
A version of this article first appeared on Medscape.com.
It denies the impact of the thousands of genes and dozens of hormones involved in our individual levels of hunger, cravings, and fullness. It denies the torrential current of our ultraprocessed and calorific food environment. It denies the constant push of food advertising and the role food has taken on as the star of even the smallest of events and celebrations. It denies the role of food as a seminal pleasure in a world that, even for those possessing great degrees of privilege is challenging, let alone for those facing tremendous and varied difficulties. And of course, it upholds the hateful notion that, if people just wanted it badly enough, they’d manage their weight, the corollary of which is that people with obesity are unmotivated and lazy.
Yet the notion that, if people want it badly enough, they’d make it happen, is incredibly commonplace. It’s so commonplace that NBC aired their prime-time televised reality show The Biggest Loser from 2004 through 2016, featuring people with obesity competing for a $500,000 prize during a 30-week–long orgy of fat-shaming, victim-blaming, hugely restrictive eating, and injury. It’s also so commonplace that studies are still being conducted exploring the impact of paying people to lose weight.
The most recent of these – “Effectiveness of Goal-Directed and Outcome-Based Financial Incentives for Weight Loss in Primary Care Patients With Obesity Living in Socioeconomically Disadvantaged Neighborhoods: A Randomized Clinical Trial” – examined the effects of randomly assigning participants whose annual household incomes were less than $40,000 to either a free year of Weight Watchers and the provisions of basic weight loss advice (exercise, track your food, eat healthfully, et cetera) or to an incentivized program that would see them earning up to $750 over 6 months, with dollars being awarded for such things as attendance in education sessions, keeping a food diary, recording their weight, and obtaining a certain amount of exercise or for weight loss.
Resultswise – though you might not have gathered it from the conclusion of the paper, which states that incentives were more effective at 12 months – the average incentivized participant lost roughly 6 pounds more than those given only resources. It should also be mentioned that over half of the incentivized group did not complete the study.
That these sorts of studies are still being conducted is depressing. Medicine and academia need to actively stop promoting harmful stereotypes when it comes to the genesis of a chronic noncommunicable disease that is not caused by a lack of desire, needing the right incentive, but is rather caused by the interaction of millions of years of evolution during extreme dietary insecurity with a modern-day food environment and culture that constantly offers, provides, and encourages consumption. This is especially true now that there are effective antiobesity medications whose success underwrites the notion that it’s physiology, rather than a lack of wanting it enough, that gets in the way of sustained success.
A version of this article first appeared on Medscape.com.
Low-carb, high-fat, calorie-unrestricted diet improves type 2 diabetes
This was true regardless of an individual’s calorie intake, in the randomized controlled trial published in the Annals of Internal Medicine.
Patients with T2D who ate a low-carb, high-fat diet (LCHF) lost more weight and saw greater improvements in both glycemic control and insulin resistance than those who ate a high-carb, low-fat diet (HCLF), reported lead author Camilla Dalby Hansen, MD, of University of Southern Denmark, Odense, and colleagues, suggesting that this is an effective, nonpharmaceutical treatment option for T2D.
The trial enrolled 185 patients with T2D, for whom low-calorie diets are often recommended to induce weight loss and improve glycemic control.
The trouble with this common recommendation, the investigators wrote, is that it induces hunger, so few patients stick to it.
“Therefore, calorie-unrestricted diets may be a better alternative to achieve long-term maintenance,” Dr. Hansen and colleagues wrote, noting that this approach “is not widely investigated.”
Study methods and results
In the new study, participants were randomized in a 2:1 ratio to follow the LCHF or HCLF diet for 6 months, with no restriction on calorie intake. Patients were evaluated at baseline, 3 months, 6 months, and 9 months (3 months after discontinuation). Parameters included glycemic control, serum lipid levels, and metabolic markers. The final analysis included 165 patients.
While patients in both groups lost weight, those in the LCHF group lost, on average, about 8 pounds more than the HCLF group, a significant difference. While the LCHF diet was associated with greater improvements in glycemic control (HbA1c) than the HCLF diet, it also led to slightly greater increases in LDL levels. In both groups, HDL levels increased, and triglycerides decreased, without significant differences between groups.
The above changes were not sustained 3 months after finishing the diet.
“I believe we have sufficient data to include LCHF as one of the diet options for people with type 2 diabetes,” Dr. Hansen said in a written comment, considering all available data.
Although the diet did lead to significant clinical benefits, she predicted that some patients would still struggle with adherence in the real world.
“The LCHF diet can be difficult for some people to follow,” Dr. Hansen said. “It is a bit more expensive, and it can be difficult to comply to in social gatherings, simply because our society is not suited for this type of diet.”
The magic of unrestricted calories
Jay H. Shubrook, DO, diabetologist and professor at Touro University of California, Vallejo, offered a similar view.
“When you start to fiddle with the diet, it affects not only the person, but all the people they eat with, because eating is a communal experience,” Dr. Shubrook said, in an interview.
Still, he said the present study is “a big deal,” because T2D is a “noncommunicable pandemic,” and “anything we could do that disrupts this process is very important.”
While some may struggle to follow the LCHF diet, Dr. Shubrook predicted better long-term adherence than the low-calorie diet usually recommended.
“What’s magic about this study is because it wasn’t calorie restricted, I think it made it a little bit more flexible for people to continue,” Dr. Shubrook said.
He added that he thinks patients will need a fair amount of coaching and education about food choices in order to lose weight on a diet without calorie restrictions.
Not the first study of its kind
In a written comment, Jeff Volek, PhD, RD, professor at the Ohio State University, Columbus, called the present study “another important piece of work, demonstrating yet again, that a low-carbohydrate eating pattern is superior to a high-carbohydrate approach in people with insulin resistance.”
Yet Dr. Volek, who has conducted numerous studies on low-carbohydrate diets, also said there is “little here that is new or surprising.”
He went on to admonish Dr. Hansen and colleagues for failing to recognize those who have already broken ground in this area.
“Unfortunately, these authors do not give credit to the many researchers who have published extensively on low-carbohydrate diets in the past, and instead make claims about being the first to study a calorie unrestricted low-carb diet in individuals with T2D, which is clearly not the case,” Dr. Volek said. “There is a large body of literature showing similar findings with better control over diet, larger cohorts, longer follow-up, and more comprehensive biomarker assessment.”
He noted that data supporting low-carb diets for T2D have been sufficient since at least 2019, when the American Diabetes Association updated their guidance on the subject.
Citing a paper published in Diabetes Care, he said, “Low-carbohydrate eating patterns, especially very-low-carbohydrate eating patterns, have been shown to reduce A1C and the need for antihyperglycemic medications.”
The study was funded by Novo Nordisk Foundation, Danish Diabetes Academy, Odense University Hospital, and others. The investigators disclosed additional relationships with Eli Lilly, Amgen, UCB, and others. Dr. Shubrook disclosed relationships with Abbot, AstraZeneca, Bayer, and others.
This was true regardless of an individual’s calorie intake, in the randomized controlled trial published in the Annals of Internal Medicine.
Patients with T2D who ate a low-carb, high-fat diet (LCHF) lost more weight and saw greater improvements in both glycemic control and insulin resistance than those who ate a high-carb, low-fat diet (HCLF), reported lead author Camilla Dalby Hansen, MD, of University of Southern Denmark, Odense, and colleagues, suggesting that this is an effective, nonpharmaceutical treatment option for T2D.
The trial enrolled 185 patients with T2D, for whom low-calorie diets are often recommended to induce weight loss and improve glycemic control.
The trouble with this common recommendation, the investigators wrote, is that it induces hunger, so few patients stick to it.
“Therefore, calorie-unrestricted diets may be a better alternative to achieve long-term maintenance,” Dr. Hansen and colleagues wrote, noting that this approach “is not widely investigated.”
Study methods and results
In the new study, participants were randomized in a 2:1 ratio to follow the LCHF or HCLF diet for 6 months, with no restriction on calorie intake. Patients were evaluated at baseline, 3 months, 6 months, and 9 months (3 months after discontinuation). Parameters included glycemic control, serum lipid levels, and metabolic markers. The final analysis included 165 patients.
While patients in both groups lost weight, those in the LCHF group lost, on average, about 8 pounds more than the HCLF group, a significant difference. While the LCHF diet was associated with greater improvements in glycemic control (HbA1c) than the HCLF diet, it also led to slightly greater increases in LDL levels. In both groups, HDL levels increased, and triglycerides decreased, without significant differences between groups.
The above changes were not sustained 3 months after finishing the diet.
“I believe we have sufficient data to include LCHF as one of the diet options for people with type 2 diabetes,” Dr. Hansen said in a written comment, considering all available data.
Although the diet did lead to significant clinical benefits, she predicted that some patients would still struggle with adherence in the real world.
“The LCHF diet can be difficult for some people to follow,” Dr. Hansen said. “It is a bit more expensive, and it can be difficult to comply to in social gatherings, simply because our society is not suited for this type of diet.”
The magic of unrestricted calories
Jay H. Shubrook, DO, diabetologist and professor at Touro University of California, Vallejo, offered a similar view.
“When you start to fiddle with the diet, it affects not only the person, but all the people they eat with, because eating is a communal experience,” Dr. Shubrook said, in an interview.
Still, he said the present study is “a big deal,” because T2D is a “noncommunicable pandemic,” and “anything we could do that disrupts this process is very important.”
While some may struggle to follow the LCHF diet, Dr. Shubrook predicted better long-term adherence than the low-calorie diet usually recommended.
“What’s magic about this study is because it wasn’t calorie restricted, I think it made it a little bit more flexible for people to continue,” Dr. Shubrook said.
He added that he thinks patients will need a fair amount of coaching and education about food choices in order to lose weight on a diet without calorie restrictions.
Not the first study of its kind
In a written comment, Jeff Volek, PhD, RD, professor at the Ohio State University, Columbus, called the present study “another important piece of work, demonstrating yet again, that a low-carbohydrate eating pattern is superior to a high-carbohydrate approach in people with insulin resistance.”
Yet Dr. Volek, who has conducted numerous studies on low-carbohydrate diets, also said there is “little here that is new or surprising.”
He went on to admonish Dr. Hansen and colleagues for failing to recognize those who have already broken ground in this area.
“Unfortunately, these authors do not give credit to the many researchers who have published extensively on low-carbohydrate diets in the past, and instead make claims about being the first to study a calorie unrestricted low-carb diet in individuals with T2D, which is clearly not the case,” Dr. Volek said. “There is a large body of literature showing similar findings with better control over diet, larger cohorts, longer follow-up, and more comprehensive biomarker assessment.”
He noted that data supporting low-carb diets for T2D have been sufficient since at least 2019, when the American Diabetes Association updated their guidance on the subject.
Citing a paper published in Diabetes Care, he said, “Low-carbohydrate eating patterns, especially very-low-carbohydrate eating patterns, have been shown to reduce A1C and the need for antihyperglycemic medications.”
The study was funded by Novo Nordisk Foundation, Danish Diabetes Academy, Odense University Hospital, and others. The investigators disclosed additional relationships with Eli Lilly, Amgen, UCB, and others. Dr. Shubrook disclosed relationships with Abbot, AstraZeneca, Bayer, and others.
This was true regardless of an individual’s calorie intake, in the randomized controlled trial published in the Annals of Internal Medicine.
Patients with T2D who ate a low-carb, high-fat diet (LCHF) lost more weight and saw greater improvements in both glycemic control and insulin resistance than those who ate a high-carb, low-fat diet (HCLF), reported lead author Camilla Dalby Hansen, MD, of University of Southern Denmark, Odense, and colleagues, suggesting that this is an effective, nonpharmaceutical treatment option for T2D.
The trial enrolled 185 patients with T2D, for whom low-calorie diets are often recommended to induce weight loss and improve glycemic control.
The trouble with this common recommendation, the investigators wrote, is that it induces hunger, so few patients stick to it.
“Therefore, calorie-unrestricted diets may be a better alternative to achieve long-term maintenance,” Dr. Hansen and colleagues wrote, noting that this approach “is not widely investigated.”
Study methods and results
In the new study, participants were randomized in a 2:1 ratio to follow the LCHF or HCLF diet for 6 months, with no restriction on calorie intake. Patients were evaluated at baseline, 3 months, 6 months, and 9 months (3 months after discontinuation). Parameters included glycemic control, serum lipid levels, and metabolic markers. The final analysis included 165 patients.
While patients in both groups lost weight, those in the LCHF group lost, on average, about 8 pounds more than the HCLF group, a significant difference. While the LCHF diet was associated with greater improvements in glycemic control (HbA1c) than the HCLF diet, it also led to slightly greater increases in LDL levels. In both groups, HDL levels increased, and triglycerides decreased, without significant differences between groups.
The above changes were not sustained 3 months after finishing the diet.
“I believe we have sufficient data to include LCHF as one of the diet options for people with type 2 diabetes,” Dr. Hansen said in a written comment, considering all available data.
Although the diet did lead to significant clinical benefits, she predicted that some patients would still struggle with adherence in the real world.
“The LCHF diet can be difficult for some people to follow,” Dr. Hansen said. “It is a bit more expensive, and it can be difficult to comply to in social gatherings, simply because our society is not suited for this type of diet.”
The magic of unrestricted calories
Jay H. Shubrook, DO, diabetologist and professor at Touro University of California, Vallejo, offered a similar view.
“When you start to fiddle with the diet, it affects not only the person, but all the people they eat with, because eating is a communal experience,” Dr. Shubrook said, in an interview.
Still, he said the present study is “a big deal,” because T2D is a “noncommunicable pandemic,” and “anything we could do that disrupts this process is very important.”
While some may struggle to follow the LCHF diet, Dr. Shubrook predicted better long-term adherence than the low-calorie diet usually recommended.
“What’s magic about this study is because it wasn’t calorie restricted, I think it made it a little bit more flexible for people to continue,” Dr. Shubrook said.
He added that he thinks patients will need a fair amount of coaching and education about food choices in order to lose weight on a diet without calorie restrictions.
Not the first study of its kind
In a written comment, Jeff Volek, PhD, RD, professor at the Ohio State University, Columbus, called the present study “another important piece of work, demonstrating yet again, that a low-carbohydrate eating pattern is superior to a high-carbohydrate approach in people with insulin resistance.”
Yet Dr. Volek, who has conducted numerous studies on low-carbohydrate diets, also said there is “little here that is new or surprising.”
He went on to admonish Dr. Hansen and colleagues for failing to recognize those who have already broken ground in this area.
“Unfortunately, these authors do not give credit to the many researchers who have published extensively on low-carbohydrate diets in the past, and instead make claims about being the first to study a calorie unrestricted low-carb diet in individuals with T2D, which is clearly not the case,” Dr. Volek said. “There is a large body of literature showing similar findings with better control over diet, larger cohorts, longer follow-up, and more comprehensive biomarker assessment.”
He noted that data supporting low-carb diets for T2D have been sufficient since at least 2019, when the American Diabetes Association updated their guidance on the subject.
Citing a paper published in Diabetes Care, he said, “Low-carbohydrate eating patterns, especially very-low-carbohydrate eating patterns, have been shown to reduce A1C and the need for antihyperglycemic medications.”
The study was funded by Novo Nordisk Foundation, Danish Diabetes Academy, Odense University Hospital, and others. The investigators disclosed additional relationships with Eli Lilly, Amgen, UCB, and others. Dr. Shubrook disclosed relationships with Abbot, AstraZeneca, Bayer, and others.
FROM ANNALS OF INTERNAL MEDICINE
Cold water immersion can have benefits
according to researchers from the Arctic University of Norway and the University Hospital of North Norway.
What to know
- Immersion in cold water has a major impact on the body. It elevates the heart rate and has positive effects on brown adipose tissue, a type of “good” body fat that is activated by cold and may protect against and cardiovascular disease.
- Exposure to cold water or cold air also appears to increase the production of the protein adiponectin by adipose tissue. Adiponectin plays a key role in protecting against , diabetes, and other diseases.
- Repeated cold-water immersions by inexperienced as well as experienced swimmers during the winter months significantly increased sensitivity and decreased insulin concentrations.
- Numerous health and well-being claims from regular exposure to the cold, such as weight loss, better mental health, and increased libido, may be explained by other factors, including an active lifestyle, trained stress handling, and social interactions, as well as a positive mindset.
- Those seeking to voluntarily practice cold-water immersion need to be educated about possible health risks associated with taking a dip in icy water, which include the consequences of hypothermia, and of heart and lung problems, which are often related to the shock from the cold.
This is a summary of the article, “Health effects of voluntary exposure to cold water – a continuing subject of debate,” published by the International Journal of Circumpolar Health.
A version of this article first appeared on Medscape.com.
according to researchers from the Arctic University of Norway and the University Hospital of North Norway.
What to know
- Immersion in cold water has a major impact on the body. It elevates the heart rate and has positive effects on brown adipose tissue, a type of “good” body fat that is activated by cold and may protect against and cardiovascular disease.
- Exposure to cold water or cold air also appears to increase the production of the protein adiponectin by adipose tissue. Adiponectin plays a key role in protecting against , diabetes, and other diseases.
- Repeated cold-water immersions by inexperienced as well as experienced swimmers during the winter months significantly increased sensitivity and decreased insulin concentrations.
- Numerous health and well-being claims from regular exposure to the cold, such as weight loss, better mental health, and increased libido, may be explained by other factors, including an active lifestyle, trained stress handling, and social interactions, as well as a positive mindset.
- Those seeking to voluntarily practice cold-water immersion need to be educated about possible health risks associated with taking a dip in icy water, which include the consequences of hypothermia, and of heart and lung problems, which are often related to the shock from the cold.
This is a summary of the article, “Health effects of voluntary exposure to cold water – a continuing subject of debate,” published by the International Journal of Circumpolar Health.
A version of this article first appeared on Medscape.com.
according to researchers from the Arctic University of Norway and the University Hospital of North Norway.
What to know
- Immersion in cold water has a major impact on the body. It elevates the heart rate and has positive effects on brown adipose tissue, a type of “good” body fat that is activated by cold and may protect against and cardiovascular disease.
- Exposure to cold water or cold air also appears to increase the production of the protein adiponectin by adipose tissue. Adiponectin plays a key role in protecting against , diabetes, and other diseases.
- Repeated cold-water immersions by inexperienced as well as experienced swimmers during the winter months significantly increased sensitivity and decreased insulin concentrations.
- Numerous health and well-being claims from regular exposure to the cold, such as weight loss, better mental health, and increased libido, may be explained by other factors, including an active lifestyle, trained stress handling, and social interactions, as well as a positive mindset.
- Those seeking to voluntarily practice cold-water immersion need to be educated about possible health risks associated with taking a dip in icy water, which include the consequences of hypothermia, and of heart and lung problems, which are often related to the shock from the cold.
This is a summary of the article, “Health effects of voluntary exposure to cold water – a continuing subject of debate,” published by the International Journal of Circumpolar Health.
A version of this article first appeared on Medscape.com.
FROM THE INTERNATIONAL JOURNAL OF CIRCUMPOLAR HEALTH
Everyone wins when losers get paid
Bribery really is the solution to all of life’s problems
Breaking news: The United States has a bit of an obesity epidemic. Okay, maybe not so breaking news. But it’s a problem we’ve been struggling with for a very long time. Part of the issue is that there really is no secret to weight loss. Pretty much anything can work if you’re committed. The millions of diets floating around are testament to this idea.
The problem of losing weight is amplified if you don’t rake in the big bucks. Lower-income individuals often can’t afford healthy superfoods, and they’re often too busy to spend time at classes, exercising, or following programs. A group of researchers at New York University has offered up an alternate solution to encourage weight loss in low-income people: Pay them.
Specifically, pay them for losing weight. A reward, if you will. The researchers recruited several hundred lower-income people and split them into three groups. All participants received a free 1-year membership to a gym and weight-loss program, as well as food journals and fitness devices, but one group received payment (on average, about $300 overall) for attending meetings, exercising a certain amount every week, or weighing themselves twice a week. About 40% of people in this group lost 5% of their body weight after 6 months, twice as many as in the group that did not receive payment for performing these tasks.
The big winners, however, were those in the third group. They also received the free stuff, but the researchers offered them a more simple and direct bribe: Lose 5% of your weight over 6 months and we’ll pay you. The reward? About $450 on average, and it worked very well, with half this group losing the weight after 6 months. That said, after a year something like a fifth of this group put the weight back on, bringing them in line with the group that was paid to perform tasks. Still, both groups outperformed the control group, which received no money.
The takeaway from this research is pretty obvious. Pay people a fair price to do something, and they’ll do it. This is a lesson that has absolutely no relevance in the modern world. Nope, none whatsoever. We all receive completely fair wages. We all have plenty of money to pay for things. Everything is fine.
More green space, less medicine
Have you heard of the 3-30-300 rule? Proposed by urban forester Cecil Konijnendijk, it’s become the rule of thumb for urban planners and other foresters into getting more green space in populated areas. A recent study has found that people who lived within this 3-30-300 rule had better mental health and less medication use.
If you’re not an urban forester, however, you may not know what the 3-30-300 rule is. But it’s pretty simple, people should be able to see at least three trees from their home, have 30% tree canopy in their neighborhood, and have 300 Spartans to defend against the Persian army.
We may have made that last one up. It’s actually have a green space or park within 300 meters of your home.
In the new study, only 4.7% of people surveyed lived in an area that followed all three rules. About 62% of the surveyed lived with a green space at least 300 meters away, 43% had at least three trees within 15 meters from their home, and a rather pitiful 9% had adequate tree canopy coverage in their neighborhood.
Greater adherence to the 3-30-300 rule was associated with fewer visits to the psychologist, with 8.3% of the participants reporting a psychologist visit in the last year. The data come from a sample of a little over 3,000 Barcelona residents aged 15-97 who were randomly selected to participate in the Barcelona Public Health Agency Survey.
“There is an urgent need to provide citizens with more green space,” said Mark Nieuwenhuijsen, lead author of the study. “We may need to tear out asphalt and plant more trees, which would not only improve health, but also reduce heat island effects and contribute to carbon capture.”
The main goal and message is that more green space is good for everyone. So if you’re feeling a little overwhelmed, take a breather and sit somewhere green. Or call those 300 Spartans and get them to start knocking some buildings down.
Said the toilet to the engineer: Do you hear what I hear?
A mythical hero’s journey took Dorothy along the yellow brick road to find the Wizard of Oz. Huckleberry Finn used a raft to float down the Mississippi River. Luke Skywalker did most of his traveling between planets. For the rest of us, the journey may be just a bit shorter.
Also a bit less heroic. Unless, of course, you’re prepping for a colonoscopy. Yup, we’re headed to the toilet, but not just any toilet. This toilet was the subject of a presentation at the annual meeting of the Acoustical Society of America, titled “The feces thesis: Using machine learning to detect diarrhea,” and that presentation was the hero’s journey of Maia Gatlin, PhD, a research engineer at the Georgia Institute of Technology.
She and her team attached a noninvasive microphone sensor to a toilet, and now they can identify bowel diseases without collecting any identifiable information.
The audio sample of an excretion event is “transformed into a spectrogram, which essentially captures the sound in an image. Different events produce different features in the audio and the spectrogram. For example, urination creates a consistent tone, while defecation may have a singular tone. In contrast, diarrhea is more random,” they explained in the written statement.
They used a machine learning algorithm to classify each spectrogram based on its features. “The algorithm’s performance was tested against data with and without background noises to make sure it was learning the right sound features, regardless of the sensor’s environment,” Dr. Gatlin and associates wrote.
Their goal is to use the toilet sensor in areas where cholera is common to prevent the spread of disease. After that, who knows? “Perhaps someday, our algorithm can be used with existing in-home smart devices to monitor one’s own bowel movements and health!” she suggested.
That would be a heroic toilet indeed.
Bribery really is the solution to all of life’s problems
Breaking news: The United States has a bit of an obesity epidemic. Okay, maybe not so breaking news. But it’s a problem we’ve been struggling with for a very long time. Part of the issue is that there really is no secret to weight loss. Pretty much anything can work if you’re committed. The millions of diets floating around are testament to this idea.
The problem of losing weight is amplified if you don’t rake in the big bucks. Lower-income individuals often can’t afford healthy superfoods, and they’re often too busy to spend time at classes, exercising, or following programs. A group of researchers at New York University has offered up an alternate solution to encourage weight loss in low-income people: Pay them.
Specifically, pay them for losing weight. A reward, if you will. The researchers recruited several hundred lower-income people and split them into three groups. All participants received a free 1-year membership to a gym and weight-loss program, as well as food journals and fitness devices, but one group received payment (on average, about $300 overall) for attending meetings, exercising a certain amount every week, or weighing themselves twice a week. About 40% of people in this group lost 5% of their body weight after 6 months, twice as many as in the group that did not receive payment for performing these tasks.
The big winners, however, were those in the third group. They also received the free stuff, but the researchers offered them a more simple and direct bribe: Lose 5% of your weight over 6 months and we’ll pay you. The reward? About $450 on average, and it worked very well, with half this group losing the weight after 6 months. That said, after a year something like a fifth of this group put the weight back on, bringing them in line with the group that was paid to perform tasks. Still, both groups outperformed the control group, which received no money.
The takeaway from this research is pretty obvious. Pay people a fair price to do something, and they’ll do it. This is a lesson that has absolutely no relevance in the modern world. Nope, none whatsoever. We all receive completely fair wages. We all have plenty of money to pay for things. Everything is fine.
More green space, less medicine
Have you heard of the 3-30-300 rule? Proposed by urban forester Cecil Konijnendijk, it’s become the rule of thumb for urban planners and other foresters into getting more green space in populated areas. A recent study has found that people who lived within this 3-30-300 rule had better mental health and less medication use.
If you’re not an urban forester, however, you may not know what the 3-30-300 rule is. But it’s pretty simple, people should be able to see at least three trees from their home, have 30% tree canopy in their neighborhood, and have 300 Spartans to defend against the Persian army.
We may have made that last one up. It’s actually have a green space or park within 300 meters of your home.
In the new study, only 4.7% of people surveyed lived in an area that followed all three rules. About 62% of the surveyed lived with a green space at least 300 meters away, 43% had at least three trees within 15 meters from their home, and a rather pitiful 9% had adequate tree canopy coverage in their neighborhood.
Greater adherence to the 3-30-300 rule was associated with fewer visits to the psychologist, with 8.3% of the participants reporting a psychologist visit in the last year. The data come from a sample of a little over 3,000 Barcelona residents aged 15-97 who were randomly selected to participate in the Barcelona Public Health Agency Survey.
“There is an urgent need to provide citizens with more green space,” said Mark Nieuwenhuijsen, lead author of the study. “We may need to tear out asphalt and plant more trees, which would not only improve health, but also reduce heat island effects and contribute to carbon capture.”
The main goal and message is that more green space is good for everyone. So if you’re feeling a little overwhelmed, take a breather and sit somewhere green. Or call those 300 Spartans and get them to start knocking some buildings down.
Said the toilet to the engineer: Do you hear what I hear?
A mythical hero’s journey took Dorothy along the yellow brick road to find the Wizard of Oz. Huckleberry Finn used a raft to float down the Mississippi River. Luke Skywalker did most of his traveling between planets. For the rest of us, the journey may be just a bit shorter.
Also a bit less heroic. Unless, of course, you’re prepping for a colonoscopy. Yup, we’re headed to the toilet, but not just any toilet. This toilet was the subject of a presentation at the annual meeting of the Acoustical Society of America, titled “The feces thesis: Using machine learning to detect diarrhea,” and that presentation was the hero’s journey of Maia Gatlin, PhD, a research engineer at the Georgia Institute of Technology.
She and her team attached a noninvasive microphone sensor to a toilet, and now they can identify bowel diseases without collecting any identifiable information.
The audio sample of an excretion event is “transformed into a spectrogram, which essentially captures the sound in an image. Different events produce different features in the audio and the spectrogram. For example, urination creates a consistent tone, while defecation may have a singular tone. In contrast, diarrhea is more random,” they explained in the written statement.
They used a machine learning algorithm to classify each spectrogram based on its features. “The algorithm’s performance was tested against data with and without background noises to make sure it was learning the right sound features, regardless of the sensor’s environment,” Dr. Gatlin and associates wrote.
Their goal is to use the toilet sensor in areas where cholera is common to prevent the spread of disease. After that, who knows? “Perhaps someday, our algorithm can be used with existing in-home smart devices to monitor one’s own bowel movements and health!” she suggested.
That would be a heroic toilet indeed.
Bribery really is the solution to all of life’s problems
Breaking news: The United States has a bit of an obesity epidemic. Okay, maybe not so breaking news. But it’s a problem we’ve been struggling with for a very long time. Part of the issue is that there really is no secret to weight loss. Pretty much anything can work if you’re committed. The millions of diets floating around are testament to this idea.
The problem of losing weight is amplified if you don’t rake in the big bucks. Lower-income individuals often can’t afford healthy superfoods, and they’re often too busy to spend time at classes, exercising, or following programs. A group of researchers at New York University has offered up an alternate solution to encourage weight loss in low-income people: Pay them.
Specifically, pay them for losing weight. A reward, if you will. The researchers recruited several hundred lower-income people and split them into three groups. All participants received a free 1-year membership to a gym and weight-loss program, as well as food journals and fitness devices, but one group received payment (on average, about $300 overall) for attending meetings, exercising a certain amount every week, or weighing themselves twice a week. About 40% of people in this group lost 5% of their body weight after 6 months, twice as many as in the group that did not receive payment for performing these tasks.
The big winners, however, were those in the third group. They also received the free stuff, but the researchers offered them a more simple and direct bribe: Lose 5% of your weight over 6 months and we’ll pay you. The reward? About $450 on average, and it worked very well, with half this group losing the weight after 6 months. That said, after a year something like a fifth of this group put the weight back on, bringing them in line with the group that was paid to perform tasks. Still, both groups outperformed the control group, which received no money.
The takeaway from this research is pretty obvious. Pay people a fair price to do something, and they’ll do it. This is a lesson that has absolutely no relevance in the modern world. Nope, none whatsoever. We all receive completely fair wages. We all have plenty of money to pay for things. Everything is fine.
More green space, less medicine
Have you heard of the 3-30-300 rule? Proposed by urban forester Cecil Konijnendijk, it’s become the rule of thumb for urban planners and other foresters into getting more green space in populated areas. A recent study has found that people who lived within this 3-30-300 rule had better mental health and less medication use.
If you’re not an urban forester, however, you may not know what the 3-30-300 rule is. But it’s pretty simple, people should be able to see at least three trees from their home, have 30% tree canopy in their neighborhood, and have 300 Spartans to defend against the Persian army.
We may have made that last one up. It’s actually have a green space or park within 300 meters of your home.
In the new study, only 4.7% of people surveyed lived in an area that followed all three rules. About 62% of the surveyed lived with a green space at least 300 meters away, 43% had at least three trees within 15 meters from their home, and a rather pitiful 9% had adequate tree canopy coverage in their neighborhood.
Greater adherence to the 3-30-300 rule was associated with fewer visits to the psychologist, with 8.3% of the participants reporting a psychologist visit in the last year. The data come from a sample of a little over 3,000 Barcelona residents aged 15-97 who were randomly selected to participate in the Barcelona Public Health Agency Survey.
“There is an urgent need to provide citizens with more green space,” said Mark Nieuwenhuijsen, lead author of the study. “We may need to tear out asphalt and plant more trees, which would not only improve health, but also reduce heat island effects and contribute to carbon capture.”
The main goal and message is that more green space is good for everyone. So if you’re feeling a little overwhelmed, take a breather and sit somewhere green. Or call those 300 Spartans and get them to start knocking some buildings down.
Said the toilet to the engineer: Do you hear what I hear?
A mythical hero’s journey took Dorothy along the yellow brick road to find the Wizard of Oz. Huckleberry Finn used a raft to float down the Mississippi River. Luke Skywalker did most of his traveling between planets. For the rest of us, the journey may be just a bit shorter.
Also a bit less heroic. Unless, of course, you’re prepping for a colonoscopy. Yup, we’re headed to the toilet, but not just any toilet. This toilet was the subject of a presentation at the annual meeting of the Acoustical Society of America, titled “The feces thesis: Using machine learning to detect diarrhea,” and that presentation was the hero’s journey of Maia Gatlin, PhD, a research engineer at the Georgia Institute of Technology.
She and her team attached a noninvasive microphone sensor to a toilet, and now they can identify bowel diseases without collecting any identifiable information.
The audio sample of an excretion event is “transformed into a spectrogram, which essentially captures the sound in an image. Different events produce different features in the audio and the spectrogram. For example, urination creates a consistent tone, while defecation may have a singular tone. In contrast, diarrhea is more random,” they explained in the written statement.
They used a machine learning algorithm to classify each spectrogram based on its features. “The algorithm’s performance was tested against data with and without background noises to make sure it was learning the right sound features, regardless of the sensor’s environment,” Dr. Gatlin and associates wrote.
Their goal is to use the toilet sensor in areas where cholera is common to prevent the spread of disease. After that, who knows? “Perhaps someday, our algorithm can be used with existing in-home smart devices to monitor one’s own bowel movements and health!” she suggested.
That would be a heroic toilet indeed.
Youths have strong opinions on language about body weight
With youth obesity on the rise – an estimated 1 in 5 youths are impacted by obesity, according to the Centers for Disease Control and Prevention – conversations about healthy weight are becoming more commonplace, not only in the pediatrician’s office, but at home, too. But the language we use around this sensitive topic is important, as youths are acutely aware that words have a direct impact on their mental health.
Sixteen-year-old Avery DiCocco of Northbrook, Ill., knows how vulnerable teenagers feel.
“I think it definitely matters the way that parents and doctors address weight,” she said. “You never know who may be insecure, and using negative words could go a lot further than they think with impacting self-esteem.”
A new study published in Pediatrics brings light to the words that parents (and providers) use when speaking to youths (ages 10-17) about their weight.
Researchers from the University of Connecticut Rudd Center for Food Policy & Health, Hartford, led an online survey of youths and their parents. Those who took part were asked about 27 terms related to body weight. Parents were asked to comment on their use of these words, while youths commented on the emotional response. The researchers said 1,936 parents and 2,032 adolescents were surveyed between September and December 2021.
Although results skewed toward the use of more positive words, such as “healthy weight,” over terms like “obese,” “fat” or “large,” there was variation across ethnicity, sexual orientation, and weight status. For example, it was noted in the study, funded by WW International, that preference for the word “curvy” was higher among Hispanic/Latino youths, sexual minority youths, and those with a body mass index in the 95th percentile, compared with their White, heterosexual, and lower-weight peers.
Words matter
In 2017, the American Academy of Pediatrics came out with a policy statement on weight stigma and the need for doctors to use more neutral language and less stigmatizing terms in practice when discussing weight among youths.
But one of the reasons this new study is important, said Gregory Germain, MD, associate chief of pediatrics at Yale New Haven (Conn.) Children’s Hospital, is that this study focuses on parents who interact with their kids much more often than a pediatrician who sees them a few times a year.
“Parental motivation, discussion, interaction on a consistent basis – that dialogue is so critical in kids with obesity,” said Germain, who stresses that all adults, coaches, and educators should consider this study as well.
“When we think about those detrimental impacts on mental health when more stigmatizing language is used, just us being more mindful in how we are talking to youth can make such a profound impact,” said Rebecca Kamody, PhD, a clinical psychologist at Yale University, with a research and clinical focus on eating and weight disorders.
“In essence, this is a low-hanging fruit intervention,” she said.
Dr. Kamody recommends we take a lesson from “cultural humility” in psychology to understand how to approach this with kids, calling for “the humbleness as parents or providers in asking someone what they want used to make it the safest place for a discussion with our youth.”
One piece of the puzzle
Dr. Germain and Dr. Kamody agreed that language in discussing this topic is important but that we need to recognize that this topic in general is extremely tricky.
For one, “there are these very real metabolic complications of having high weight at a young age,” said Kamody, who stresses the need for balance to make real change.
Dr. Germain agreed. “Finding a fine line between discussing obesity and not kicking your kid into disordered eating is important.”
The researchers also recognize the limits of an online study, where self-reporting parents may not want to admit using negative weight terminology, but certainly believe it’s a start in identifying some of the undesirable patterns that may be occurring when it comes to weight.
“The overarching message is a positive one, that with our preteens and teenage kids, we need to watch our language, to create a nonjudgmental and safe environment to discuss weight and any issue involved with taking care of themselves,” said Dr. Germain.
A version of this article first appeared on Medscape.com.
With youth obesity on the rise – an estimated 1 in 5 youths are impacted by obesity, according to the Centers for Disease Control and Prevention – conversations about healthy weight are becoming more commonplace, not only in the pediatrician’s office, but at home, too. But the language we use around this sensitive topic is important, as youths are acutely aware that words have a direct impact on their mental health.
Sixteen-year-old Avery DiCocco of Northbrook, Ill., knows how vulnerable teenagers feel.
“I think it definitely matters the way that parents and doctors address weight,” she said. “You never know who may be insecure, and using negative words could go a lot further than they think with impacting self-esteem.”
A new study published in Pediatrics brings light to the words that parents (and providers) use when speaking to youths (ages 10-17) about their weight.
Researchers from the University of Connecticut Rudd Center for Food Policy & Health, Hartford, led an online survey of youths and their parents. Those who took part were asked about 27 terms related to body weight. Parents were asked to comment on their use of these words, while youths commented on the emotional response. The researchers said 1,936 parents and 2,032 adolescents were surveyed between September and December 2021.
Although results skewed toward the use of more positive words, such as “healthy weight,” over terms like “obese,” “fat” or “large,” there was variation across ethnicity, sexual orientation, and weight status. For example, it was noted in the study, funded by WW International, that preference for the word “curvy” was higher among Hispanic/Latino youths, sexual minority youths, and those with a body mass index in the 95th percentile, compared with their White, heterosexual, and lower-weight peers.
Words matter
In 2017, the American Academy of Pediatrics came out with a policy statement on weight stigma and the need for doctors to use more neutral language and less stigmatizing terms in practice when discussing weight among youths.
But one of the reasons this new study is important, said Gregory Germain, MD, associate chief of pediatrics at Yale New Haven (Conn.) Children’s Hospital, is that this study focuses on parents who interact with their kids much more often than a pediatrician who sees them a few times a year.
“Parental motivation, discussion, interaction on a consistent basis – that dialogue is so critical in kids with obesity,” said Germain, who stresses that all adults, coaches, and educators should consider this study as well.
“When we think about those detrimental impacts on mental health when more stigmatizing language is used, just us being more mindful in how we are talking to youth can make such a profound impact,” said Rebecca Kamody, PhD, a clinical psychologist at Yale University, with a research and clinical focus on eating and weight disorders.
“In essence, this is a low-hanging fruit intervention,” she said.
Dr. Kamody recommends we take a lesson from “cultural humility” in psychology to understand how to approach this with kids, calling for “the humbleness as parents or providers in asking someone what they want used to make it the safest place for a discussion with our youth.”
One piece of the puzzle
Dr. Germain and Dr. Kamody agreed that language in discussing this topic is important but that we need to recognize that this topic in general is extremely tricky.
For one, “there are these very real metabolic complications of having high weight at a young age,” said Kamody, who stresses the need for balance to make real change.
Dr. Germain agreed. “Finding a fine line between discussing obesity and not kicking your kid into disordered eating is important.”
The researchers also recognize the limits of an online study, where self-reporting parents may not want to admit using negative weight terminology, but certainly believe it’s a start in identifying some of the undesirable patterns that may be occurring when it comes to weight.
“The overarching message is a positive one, that with our preteens and teenage kids, we need to watch our language, to create a nonjudgmental and safe environment to discuss weight and any issue involved with taking care of themselves,” said Dr. Germain.
A version of this article first appeared on Medscape.com.
With youth obesity on the rise – an estimated 1 in 5 youths are impacted by obesity, according to the Centers for Disease Control and Prevention – conversations about healthy weight are becoming more commonplace, not only in the pediatrician’s office, but at home, too. But the language we use around this sensitive topic is important, as youths are acutely aware that words have a direct impact on their mental health.
Sixteen-year-old Avery DiCocco of Northbrook, Ill., knows how vulnerable teenagers feel.
“I think it definitely matters the way that parents and doctors address weight,” she said. “You never know who may be insecure, and using negative words could go a lot further than they think with impacting self-esteem.”
A new study published in Pediatrics brings light to the words that parents (and providers) use when speaking to youths (ages 10-17) about their weight.
Researchers from the University of Connecticut Rudd Center for Food Policy & Health, Hartford, led an online survey of youths and their parents. Those who took part were asked about 27 terms related to body weight. Parents were asked to comment on their use of these words, while youths commented on the emotional response. The researchers said 1,936 parents and 2,032 adolescents were surveyed between September and December 2021.
Although results skewed toward the use of more positive words, such as “healthy weight,” over terms like “obese,” “fat” or “large,” there was variation across ethnicity, sexual orientation, and weight status. For example, it was noted in the study, funded by WW International, that preference for the word “curvy” was higher among Hispanic/Latino youths, sexual minority youths, and those with a body mass index in the 95th percentile, compared with their White, heterosexual, and lower-weight peers.
Words matter
In 2017, the American Academy of Pediatrics came out with a policy statement on weight stigma and the need for doctors to use more neutral language and less stigmatizing terms in practice when discussing weight among youths.
But one of the reasons this new study is important, said Gregory Germain, MD, associate chief of pediatrics at Yale New Haven (Conn.) Children’s Hospital, is that this study focuses on parents who interact with their kids much more often than a pediatrician who sees them a few times a year.
“Parental motivation, discussion, interaction on a consistent basis – that dialogue is so critical in kids with obesity,” said Germain, who stresses that all adults, coaches, and educators should consider this study as well.
“When we think about those detrimental impacts on mental health when more stigmatizing language is used, just us being more mindful in how we are talking to youth can make such a profound impact,” said Rebecca Kamody, PhD, a clinical psychologist at Yale University, with a research and clinical focus on eating and weight disorders.
“In essence, this is a low-hanging fruit intervention,” she said.
Dr. Kamody recommends we take a lesson from “cultural humility” in psychology to understand how to approach this with kids, calling for “the humbleness as parents or providers in asking someone what they want used to make it the safest place for a discussion with our youth.”
One piece of the puzzle
Dr. Germain and Dr. Kamody agreed that language in discussing this topic is important but that we need to recognize that this topic in general is extremely tricky.
For one, “there are these very real metabolic complications of having high weight at a young age,” said Kamody, who stresses the need for balance to make real change.
Dr. Germain agreed. “Finding a fine line between discussing obesity and not kicking your kid into disordered eating is important.”
The researchers also recognize the limits of an online study, where self-reporting parents may not want to admit using negative weight terminology, but certainly believe it’s a start in identifying some of the undesirable patterns that may be occurring when it comes to weight.
“The overarching message is a positive one, that with our preteens and teenage kids, we need to watch our language, to create a nonjudgmental and safe environment to discuss weight and any issue involved with taking care of themselves,” said Dr. Germain.
A version of this article first appeared on Medscape.com.
FROM PEDIATRICS
Green Mediterranean diet lowers visceral adipose tissue
according to a new analysis of the 18-month Dietary Intervention Randomized Controlled Trial Polyphenols Unprocessed (DIRECT-PLUS) trial.
The new results indicate that the green Mediterranean diet lowered visceral fat by twice as much as the standard Mediterranean diet (14% vs. 7%), reported Iris Shai, PhD, of Ben-Gurion University of the Negev in Be’er Sheva, Israel, and colleagues.
“This study may suggest an improved dietary protocol for treating visceral adiposity,” the authors wrote in their article, published recently in BMC Medicine.
“A healthy lifestyle is a strong basis for any weight-loss program. We learned from the results of our experiment that the quality of food is no less important than the number of calories consumed and the goal today is to understand the mechanisms of various nutrients, for example, positive ones such as the polyphenols, and negative ones such as empty carbohydrates and processed red meat, on the pace of fat cell differentiation and their aggregation in the viscera,” Dr. Shai said in a press release from Ben‐Gurion University.
“A 14% reduction in visceral fat is a dramatic achievement for making simple changes to your diet and lifestyle. Weight loss is an important goal only if it is accompanied by impressive results in reducing adipose tissue,” added coauthor Hila Zelicha, RD, PhD, also of Ben‐Gurion University of the Negev.
Previous randomized controlled trials have shown that dietary changes with a higher polyphenol content tend to produce better cardiometabolic outcomes and appear to mobilize particular ectopic fat depots, the researchers noted.
The main results of the DIRECT-PLUS trial were published in 2020 in Heart. Almost 300 participants with abdominal obesity/dyslipidemia were randomized to one of three diet groups (all accompanied by physical activity): standard healthy dietary guidelines (HDG), standard Mediterranean diet, and the so-called green Mediterranean diet. The mean age of participants was 51 years, and men comprised 88% of the study cohort.
Participants in both Mediterranean diet groups ate 28 grams/day of walnuts, which accounted for about 440 mg/day of polyphenols. Participants in the green Mediterranean group also ate 100 grams/day of frozen cubes of a Wolffia globosa (duckweed strain) plant green shake, and three to four cups/day of green tea, which contributed to consumption of 800 mg/day of polyphenols, and decreased red meat consumption.
Both the green and standard Mediterranean diet groups achieved similar weight loss (–6.2 kg and –5.4 kg) compared with the HDG group (–1.5 kg; P < .001). However, the green Mediterranean diet group had a greater reduction in waist circumference (–8.6 cm) than the standard Mediterranean diet group (–6.8 cm; P = .033) and HDG group (–4.3 cm; P < .001). Stratification by gender showed these differences were significant only among men.
Explaining the rationale for the study, the researchers noted that visceral adipose tissue accumulation is a key factor that differentiates metabolic healthy and unhealthy obese individuals, is closely related to the development of multiple cardiovascular risk factors, including hypertension, dyslipidemia, and type 2 diabetes, and is an independent marker of mortality.
Now, their latest data show the green Mediterranean diet group lost approximately twice as much visceral adipose tissue compared with the standard Mediterranean diet and HDG groups (−14.1%, −6.0%, and − 4.2%; P < .05 independent of weight loss, sex, waist circumference, or age).
Lower red meat consumption, greater dietary consumption of walnuts, Wolffia globosa, and green tea, increased urine urolithin A polyphenol, and elevated total plasma polyphenols were significantly associated with greater visceral adipose tissue loss (P < .05, multivariate models).
“A green Mediterranean diet enriched with polyphenols and decreased red meat consumption might serve as an improved version of the Mediterranean diet for targeted VAT reduction. Future studies are needed to explore the exact mechanisms of specific polyphenol-rich foods on visceral adiposity,” the study authors concluded.
A version of this article first appeared on Medscape.com.
according to a new analysis of the 18-month Dietary Intervention Randomized Controlled Trial Polyphenols Unprocessed (DIRECT-PLUS) trial.
The new results indicate that the green Mediterranean diet lowered visceral fat by twice as much as the standard Mediterranean diet (14% vs. 7%), reported Iris Shai, PhD, of Ben-Gurion University of the Negev in Be’er Sheva, Israel, and colleagues.
“This study may suggest an improved dietary protocol for treating visceral adiposity,” the authors wrote in their article, published recently in BMC Medicine.
“A healthy lifestyle is a strong basis for any weight-loss program. We learned from the results of our experiment that the quality of food is no less important than the number of calories consumed and the goal today is to understand the mechanisms of various nutrients, for example, positive ones such as the polyphenols, and negative ones such as empty carbohydrates and processed red meat, on the pace of fat cell differentiation and their aggregation in the viscera,” Dr. Shai said in a press release from Ben‐Gurion University.
“A 14% reduction in visceral fat is a dramatic achievement for making simple changes to your diet and lifestyle. Weight loss is an important goal only if it is accompanied by impressive results in reducing adipose tissue,” added coauthor Hila Zelicha, RD, PhD, also of Ben‐Gurion University of the Negev.
Previous randomized controlled trials have shown that dietary changes with a higher polyphenol content tend to produce better cardiometabolic outcomes and appear to mobilize particular ectopic fat depots, the researchers noted.
The main results of the DIRECT-PLUS trial were published in 2020 in Heart. Almost 300 participants with abdominal obesity/dyslipidemia were randomized to one of three diet groups (all accompanied by physical activity): standard healthy dietary guidelines (HDG), standard Mediterranean diet, and the so-called green Mediterranean diet. The mean age of participants was 51 years, and men comprised 88% of the study cohort.
Participants in both Mediterranean diet groups ate 28 grams/day of walnuts, which accounted for about 440 mg/day of polyphenols. Participants in the green Mediterranean group also ate 100 grams/day of frozen cubes of a Wolffia globosa (duckweed strain) plant green shake, and three to four cups/day of green tea, which contributed to consumption of 800 mg/day of polyphenols, and decreased red meat consumption.
Both the green and standard Mediterranean diet groups achieved similar weight loss (–6.2 kg and –5.4 kg) compared with the HDG group (–1.5 kg; P < .001). However, the green Mediterranean diet group had a greater reduction in waist circumference (–8.6 cm) than the standard Mediterranean diet group (–6.8 cm; P = .033) and HDG group (–4.3 cm; P < .001). Stratification by gender showed these differences were significant only among men.
Explaining the rationale for the study, the researchers noted that visceral adipose tissue accumulation is a key factor that differentiates metabolic healthy and unhealthy obese individuals, is closely related to the development of multiple cardiovascular risk factors, including hypertension, dyslipidemia, and type 2 diabetes, and is an independent marker of mortality.
Now, their latest data show the green Mediterranean diet group lost approximately twice as much visceral adipose tissue compared with the standard Mediterranean diet and HDG groups (−14.1%, −6.0%, and − 4.2%; P < .05 independent of weight loss, sex, waist circumference, or age).
Lower red meat consumption, greater dietary consumption of walnuts, Wolffia globosa, and green tea, increased urine urolithin A polyphenol, and elevated total plasma polyphenols were significantly associated with greater visceral adipose tissue loss (P < .05, multivariate models).
“A green Mediterranean diet enriched with polyphenols and decreased red meat consumption might serve as an improved version of the Mediterranean diet for targeted VAT reduction. Future studies are needed to explore the exact mechanisms of specific polyphenol-rich foods on visceral adiposity,” the study authors concluded.
A version of this article first appeared on Medscape.com.
according to a new analysis of the 18-month Dietary Intervention Randomized Controlled Trial Polyphenols Unprocessed (DIRECT-PLUS) trial.
The new results indicate that the green Mediterranean diet lowered visceral fat by twice as much as the standard Mediterranean diet (14% vs. 7%), reported Iris Shai, PhD, of Ben-Gurion University of the Negev in Be’er Sheva, Israel, and colleagues.
“This study may suggest an improved dietary protocol for treating visceral adiposity,” the authors wrote in their article, published recently in BMC Medicine.
“A healthy lifestyle is a strong basis for any weight-loss program. We learned from the results of our experiment that the quality of food is no less important than the number of calories consumed and the goal today is to understand the mechanisms of various nutrients, for example, positive ones such as the polyphenols, and negative ones such as empty carbohydrates and processed red meat, on the pace of fat cell differentiation and their aggregation in the viscera,” Dr. Shai said in a press release from Ben‐Gurion University.
“A 14% reduction in visceral fat is a dramatic achievement for making simple changes to your diet and lifestyle. Weight loss is an important goal only if it is accompanied by impressive results in reducing adipose tissue,” added coauthor Hila Zelicha, RD, PhD, also of Ben‐Gurion University of the Negev.
Previous randomized controlled trials have shown that dietary changes with a higher polyphenol content tend to produce better cardiometabolic outcomes and appear to mobilize particular ectopic fat depots, the researchers noted.
The main results of the DIRECT-PLUS trial were published in 2020 in Heart. Almost 300 participants with abdominal obesity/dyslipidemia were randomized to one of three diet groups (all accompanied by physical activity): standard healthy dietary guidelines (HDG), standard Mediterranean diet, and the so-called green Mediterranean diet. The mean age of participants was 51 years, and men comprised 88% of the study cohort.
Participants in both Mediterranean diet groups ate 28 grams/day of walnuts, which accounted for about 440 mg/day of polyphenols. Participants in the green Mediterranean group also ate 100 grams/day of frozen cubes of a Wolffia globosa (duckweed strain) plant green shake, and three to four cups/day of green tea, which contributed to consumption of 800 mg/day of polyphenols, and decreased red meat consumption.
Both the green and standard Mediterranean diet groups achieved similar weight loss (–6.2 kg and –5.4 kg) compared with the HDG group (–1.5 kg; P < .001). However, the green Mediterranean diet group had a greater reduction in waist circumference (–8.6 cm) than the standard Mediterranean diet group (–6.8 cm; P = .033) and HDG group (–4.3 cm; P < .001). Stratification by gender showed these differences were significant only among men.
Explaining the rationale for the study, the researchers noted that visceral adipose tissue accumulation is a key factor that differentiates metabolic healthy and unhealthy obese individuals, is closely related to the development of multiple cardiovascular risk factors, including hypertension, dyslipidemia, and type 2 diabetes, and is an independent marker of mortality.
Now, their latest data show the green Mediterranean diet group lost approximately twice as much visceral adipose tissue compared with the standard Mediterranean diet and HDG groups (−14.1%, −6.0%, and − 4.2%; P < .05 independent of weight loss, sex, waist circumference, or age).
Lower red meat consumption, greater dietary consumption of walnuts, Wolffia globosa, and green tea, increased urine urolithin A polyphenol, and elevated total plasma polyphenols were significantly associated with greater visceral adipose tissue loss (P < .05, multivariate models).
“A green Mediterranean diet enriched with polyphenols and decreased red meat consumption might serve as an improved version of the Mediterranean diet for targeted VAT reduction. Future studies are needed to explore the exact mechanisms of specific polyphenol-rich foods on visceral adiposity,” the study authors concluded.
A version of this article first appeared on Medscape.com.
FROM BMC MEDICINE
The TikTok trend that triggered a diabetes drug shortage
Weight loss advice is everywhere you look on social media, but one trend sweeping TikTok has led to shortages of an important diabetes drug.
Ozempic, a weekly injection that helps boost insulin sensitivity in people with type 2 diabetes, also suppresses appetite, which leads to weight loss. Stories of celebrities using the drug off-label to lose a few pounds have led to an explosion of interest. And now people with diabetes – people whose lives could be saved by the drug – are having trouble finding it.
Kim Kardashian and Elon Musk
In the spring, Kim Kardashian pulled off a dramatic weight loss to fit into Marilyn Monroe’s dress for the Met Gala. Soon rumors began to circulate that she’d used Ozempic to do it. Just this week, new Twitter owner Elon Musk tweeted about his own use of Ozempic and its sibling drug, Wegovy.
Variety dubbed Ozempic “the worst kept secret in Hollywood – especially given that its most enthusiastic users are not prediabetic and do not require the drug.” The rich and famous are spending $1,200 to $1,500 a month to get access.
As so often happens, high-profile use sparked a trend. Videos on TikTok hashtagged #ozempic have amassed more than 275 million views, and #ozempicweightloss has more than 110 million.
This raises concerns about who, exactly, is watching these videos, and what message they’re receiving.
“Forty-two percent of Americans have obesity, and even more have overweight. That’s affecting our younger people and our adolescents,” says Caroline Apovian, MD, codirector of the Center for Weight Management and Wellness at the Brigham and Women’s Hospital in Boston. “They’re looking at TikTok and other social media outlets for help.”
A new study shows how damaging this can be: Researchers analyzed 1,000 videos with nutrition, food, and weight-related hashtags, with over 1 billion views combined. They found that nearly all included messages glorifying weight loss and thinness.
At last, an effective weight-loss drug
Ozempic is Danish drug company Novo Nordisk’s brand name for semaglutide, which works by mimicking a naturally occurring hormone known as GLP-1. It travels to your brain and helps you feel full on less food. That leads to weight loss. In one 68-week study, semaglutide helped people lose an average of 15% of their body weight. But it’s not a miracle drug: You still have to change your eating habits and stay physically active.
The FDA approved Ozempic to treat people with type 2 diabetes in 2017. Four years later, Novo Nordisk received the green light for a higher-dose version meant specifically for people with obesity. Wegovy is approved for use only if you have a BMI of at least 27 with one or more weight-related ailments, or a BMI of 30 or more with none.
“These drugs are dominating my practice, because they’re so effective,” says Amanda Velazquez, MD, director of obesity medicine at Cedars-Sinai Medical Center in Los Angeles. The drug is considered safe, “so the majority of patients are good candidates.”
More demand than supply
As word spread about how well Ozempic and Wegovy worked, social media posts helped drive even more people to seek out the drugs. Now demand is outpacing the supply – according to the FDA, starter doses of Ozempic will have limited availability through January.
“In Hollywood, people are losing 10 pounds, getting it for $1,500 a month, and depleting stores for people who have such severe obesity that they have congestive heart failure and diabetes,” Dr. Apovian says. “These are people who are going to die, and you’re taking it away just for cosmetic weight loss. That is deplorable.”
In addition to huge demand, Wegovy also had a disruption in its supply chain. Right now, it isn’t available at all in lower doses, which is helping to spike off-label demand for Ozempic. Novo Nordisk expects to have these problems sorted out by the end of the year, with distribution following soon after.
The price of access
With a list price of $1,350 a month, Wegovy costs as much as many mortgages. And Medicaid, Medicare, and many insurance companies don’t cover it. Although obesity is a disease, the insurance industry treats weight loss as more of a vanity issue – so even if you could find the drug, you might not be able to afford it.
“We’re seeing that roughly half the prescriptions we write aren’t being covered,” Dr. Apovian says. “And for the half that are covered, we have to do prior authorization, which takes days, and it’s laborious.” In some instances, she says, insurance companies withdraw authorization after 3 months if they don’t see enough weight coming off.
It’s not like you can take Wegovy for 3 months, lose some weight, and expect it to stay off, either. The medication requires a real commitment, potentially for life. That’s because once the semaglutide leaves your system, your appetite returns. In one study, people regained two-thirds of the weight they’d lost within a year of stopping.
Many see a double standard in the insurance companies’ refusal to cover a drug that could prevent serious illness or death.
“They’re saying it’s not cost-effective to give the 42% of Americans who have a BMI over 30 Wegovy. Did they say this when statins came out?” Dr. Apovian says. “Why are they doing this with antiobesity agents? It’s the culture. The culture isn’t ready to adopt obesity as the disease that it is.”
Unpleasant side effects
Let’s assume you’re one of the lucky ones – your insurance covers Wegovy, and you can actually find some. You might discover that using it is no walk in the park. Common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea.
“The way we counteract that is to start very slowly at a low dose of these medications,” Dr. Apovian says. “We only go up when the patient doesn’t have nausea or it gets better.”
Elise Davenport was excited to try Wegovy. “I did my online research. I’m the type who’s interested in early adoption, tech gadgets and stuff,” says the 40-year-old technical writer. “I wanted to try it because I’d tried so many other things that failed, or hadn’t worked long-term.”
With a BMI over 30, Ms. Davenport qualified for the drug. She signed up for an online program that guaranteed insurance coverage and started taking it in October 2021. At first, the side effects were mild, just a touch of nausea and diarrhea. And the results were impressive. She found it easy to feel satisfied with smaller portions and lost her cravings for sugar and highly processed foods. The weight fell off, roughly 5 pounds a week.
It turns out, that’s too much, too fast. Dr. Apovian and Dr. Velazquez say their patients lose more like 2 pounds each week, with careful monitoring.
By early December, Ms. Davenport’s side effects were ramping up. Because of shortages in lower dosages, the online program wasn’t able to adjust hers right away. She felt nauseated all the time, bad enough that brushing her teeth made her vomit and she had to force herself to eat. Some weeks, she managed less than 500 calories a day. Her sleep patterns became erratic. And then her depression, which medication had kept under control for years, spiraled.
“I remember sitting on the floor of my bathroom crying, thinking I’d rather carry the extra weight,” she says. “I used to take a lot of enjoyment from food, and I had none of that anymore. It was such a joyless experience at that point.”
Eventually, her dosage was reduced and the symptoms let up, but her primary care doctor encouraged her to stop. By the time she did, in March, she’d lost 55 pounds. So far, she’s gained back about 10.
More than just weight loss
Even though Ms. Davenport’s experience wasn’t a good one, with better monitoring, she’d be willing to try again. For one thing, seeing how easy it was to eat less with medical help helped to undo years of shame.
“Our culture treats obesity like a moral failing. I realized I’d been made to feel that way by doctors and programs – that I wasn’t doing enough,” she says. “This drug made me realize there are legit physiological things going on in my body, things that are often excluded from the conversation.”
Dr. Apovian and Dr. Velazquez say their patients regularly discover similar things.
“Obesity is not a disease of willpower. Medications are not the easy way out,” Dr. Velazquez says. “This is a chronic, relapsing medical condition, and because of that, we should treat it how we treat diabetes, high blood pressure, all these other conditions. We’d never hold back medication for individuals coming in with high blood pressure, tell them to work on willpower and withhold drugs they’d qualify for.”
A version of this article first appeared on WebMD.com.
Weight loss advice is everywhere you look on social media, but one trend sweeping TikTok has led to shortages of an important diabetes drug.
Ozempic, a weekly injection that helps boost insulin sensitivity in people with type 2 diabetes, also suppresses appetite, which leads to weight loss. Stories of celebrities using the drug off-label to lose a few pounds have led to an explosion of interest. And now people with diabetes – people whose lives could be saved by the drug – are having trouble finding it.
Kim Kardashian and Elon Musk
In the spring, Kim Kardashian pulled off a dramatic weight loss to fit into Marilyn Monroe’s dress for the Met Gala. Soon rumors began to circulate that she’d used Ozempic to do it. Just this week, new Twitter owner Elon Musk tweeted about his own use of Ozempic and its sibling drug, Wegovy.
Variety dubbed Ozempic “the worst kept secret in Hollywood – especially given that its most enthusiastic users are not prediabetic and do not require the drug.” The rich and famous are spending $1,200 to $1,500 a month to get access.
As so often happens, high-profile use sparked a trend. Videos on TikTok hashtagged #ozempic have amassed more than 275 million views, and #ozempicweightloss has more than 110 million.
This raises concerns about who, exactly, is watching these videos, and what message they’re receiving.
“Forty-two percent of Americans have obesity, and even more have overweight. That’s affecting our younger people and our adolescents,” says Caroline Apovian, MD, codirector of the Center for Weight Management and Wellness at the Brigham and Women’s Hospital in Boston. “They’re looking at TikTok and other social media outlets for help.”
A new study shows how damaging this can be: Researchers analyzed 1,000 videos with nutrition, food, and weight-related hashtags, with over 1 billion views combined. They found that nearly all included messages glorifying weight loss and thinness.
At last, an effective weight-loss drug
Ozempic is Danish drug company Novo Nordisk’s brand name for semaglutide, which works by mimicking a naturally occurring hormone known as GLP-1. It travels to your brain and helps you feel full on less food. That leads to weight loss. In one 68-week study, semaglutide helped people lose an average of 15% of their body weight. But it’s not a miracle drug: You still have to change your eating habits and stay physically active.
The FDA approved Ozempic to treat people with type 2 diabetes in 2017. Four years later, Novo Nordisk received the green light for a higher-dose version meant specifically for people with obesity. Wegovy is approved for use only if you have a BMI of at least 27 with one or more weight-related ailments, or a BMI of 30 or more with none.
“These drugs are dominating my practice, because they’re so effective,” says Amanda Velazquez, MD, director of obesity medicine at Cedars-Sinai Medical Center in Los Angeles. The drug is considered safe, “so the majority of patients are good candidates.”
More demand than supply
As word spread about how well Ozempic and Wegovy worked, social media posts helped drive even more people to seek out the drugs. Now demand is outpacing the supply – according to the FDA, starter doses of Ozempic will have limited availability through January.
“In Hollywood, people are losing 10 pounds, getting it for $1,500 a month, and depleting stores for people who have such severe obesity that they have congestive heart failure and diabetes,” Dr. Apovian says. “These are people who are going to die, and you’re taking it away just for cosmetic weight loss. That is deplorable.”
In addition to huge demand, Wegovy also had a disruption in its supply chain. Right now, it isn’t available at all in lower doses, which is helping to spike off-label demand for Ozempic. Novo Nordisk expects to have these problems sorted out by the end of the year, with distribution following soon after.
The price of access
With a list price of $1,350 a month, Wegovy costs as much as many mortgages. And Medicaid, Medicare, and many insurance companies don’t cover it. Although obesity is a disease, the insurance industry treats weight loss as more of a vanity issue – so even if you could find the drug, you might not be able to afford it.
“We’re seeing that roughly half the prescriptions we write aren’t being covered,” Dr. Apovian says. “And for the half that are covered, we have to do prior authorization, which takes days, and it’s laborious.” In some instances, she says, insurance companies withdraw authorization after 3 months if they don’t see enough weight coming off.
It’s not like you can take Wegovy for 3 months, lose some weight, and expect it to stay off, either. The medication requires a real commitment, potentially for life. That’s because once the semaglutide leaves your system, your appetite returns. In one study, people regained two-thirds of the weight they’d lost within a year of stopping.
Many see a double standard in the insurance companies’ refusal to cover a drug that could prevent serious illness or death.
“They’re saying it’s not cost-effective to give the 42% of Americans who have a BMI over 30 Wegovy. Did they say this when statins came out?” Dr. Apovian says. “Why are they doing this with antiobesity agents? It’s the culture. The culture isn’t ready to adopt obesity as the disease that it is.”
Unpleasant side effects
Let’s assume you’re one of the lucky ones – your insurance covers Wegovy, and you can actually find some. You might discover that using it is no walk in the park. Common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea.
“The way we counteract that is to start very slowly at a low dose of these medications,” Dr. Apovian says. “We only go up when the patient doesn’t have nausea or it gets better.”
Elise Davenport was excited to try Wegovy. “I did my online research. I’m the type who’s interested in early adoption, tech gadgets and stuff,” says the 40-year-old technical writer. “I wanted to try it because I’d tried so many other things that failed, or hadn’t worked long-term.”
With a BMI over 30, Ms. Davenport qualified for the drug. She signed up for an online program that guaranteed insurance coverage and started taking it in October 2021. At first, the side effects were mild, just a touch of nausea and diarrhea. And the results were impressive. She found it easy to feel satisfied with smaller portions and lost her cravings for sugar and highly processed foods. The weight fell off, roughly 5 pounds a week.
It turns out, that’s too much, too fast. Dr. Apovian and Dr. Velazquez say their patients lose more like 2 pounds each week, with careful monitoring.
By early December, Ms. Davenport’s side effects were ramping up. Because of shortages in lower dosages, the online program wasn’t able to adjust hers right away. She felt nauseated all the time, bad enough that brushing her teeth made her vomit and she had to force herself to eat. Some weeks, she managed less than 500 calories a day. Her sleep patterns became erratic. And then her depression, which medication had kept under control for years, spiraled.
“I remember sitting on the floor of my bathroom crying, thinking I’d rather carry the extra weight,” she says. “I used to take a lot of enjoyment from food, and I had none of that anymore. It was such a joyless experience at that point.”
Eventually, her dosage was reduced and the symptoms let up, but her primary care doctor encouraged her to stop. By the time she did, in March, she’d lost 55 pounds. So far, she’s gained back about 10.
More than just weight loss
Even though Ms. Davenport’s experience wasn’t a good one, with better monitoring, she’d be willing to try again. For one thing, seeing how easy it was to eat less with medical help helped to undo years of shame.
“Our culture treats obesity like a moral failing. I realized I’d been made to feel that way by doctors and programs – that I wasn’t doing enough,” she says. “This drug made me realize there are legit physiological things going on in my body, things that are often excluded from the conversation.”
Dr. Apovian and Dr. Velazquez say their patients regularly discover similar things.
“Obesity is not a disease of willpower. Medications are not the easy way out,” Dr. Velazquez says. “This is a chronic, relapsing medical condition, and because of that, we should treat it how we treat diabetes, high blood pressure, all these other conditions. We’d never hold back medication for individuals coming in with high blood pressure, tell them to work on willpower and withhold drugs they’d qualify for.”
A version of this article first appeared on WebMD.com.
Weight loss advice is everywhere you look on social media, but one trend sweeping TikTok has led to shortages of an important diabetes drug.
Ozempic, a weekly injection that helps boost insulin sensitivity in people with type 2 diabetes, also suppresses appetite, which leads to weight loss. Stories of celebrities using the drug off-label to lose a few pounds have led to an explosion of interest. And now people with diabetes – people whose lives could be saved by the drug – are having trouble finding it.
Kim Kardashian and Elon Musk
In the spring, Kim Kardashian pulled off a dramatic weight loss to fit into Marilyn Monroe’s dress for the Met Gala. Soon rumors began to circulate that she’d used Ozempic to do it. Just this week, new Twitter owner Elon Musk tweeted about his own use of Ozempic and its sibling drug, Wegovy.
Variety dubbed Ozempic “the worst kept secret in Hollywood – especially given that its most enthusiastic users are not prediabetic and do not require the drug.” The rich and famous are spending $1,200 to $1,500 a month to get access.
As so often happens, high-profile use sparked a trend. Videos on TikTok hashtagged #ozempic have amassed more than 275 million views, and #ozempicweightloss has more than 110 million.
This raises concerns about who, exactly, is watching these videos, and what message they’re receiving.
“Forty-two percent of Americans have obesity, and even more have overweight. That’s affecting our younger people and our adolescents,” says Caroline Apovian, MD, codirector of the Center for Weight Management and Wellness at the Brigham and Women’s Hospital in Boston. “They’re looking at TikTok and other social media outlets for help.”
A new study shows how damaging this can be: Researchers analyzed 1,000 videos with nutrition, food, and weight-related hashtags, with over 1 billion views combined. They found that nearly all included messages glorifying weight loss and thinness.
At last, an effective weight-loss drug
Ozempic is Danish drug company Novo Nordisk’s brand name for semaglutide, which works by mimicking a naturally occurring hormone known as GLP-1. It travels to your brain and helps you feel full on less food. That leads to weight loss. In one 68-week study, semaglutide helped people lose an average of 15% of their body weight. But it’s not a miracle drug: You still have to change your eating habits and stay physically active.
The FDA approved Ozempic to treat people with type 2 diabetes in 2017. Four years later, Novo Nordisk received the green light for a higher-dose version meant specifically for people with obesity. Wegovy is approved for use only if you have a BMI of at least 27 with one or more weight-related ailments, or a BMI of 30 or more with none.
“These drugs are dominating my practice, because they’re so effective,” says Amanda Velazquez, MD, director of obesity medicine at Cedars-Sinai Medical Center in Los Angeles. The drug is considered safe, “so the majority of patients are good candidates.”
More demand than supply
As word spread about how well Ozempic and Wegovy worked, social media posts helped drive even more people to seek out the drugs. Now demand is outpacing the supply – according to the FDA, starter doses of Ozempic will have limited availability through January.
“In Hollywood, people are losing 10 pounds, getting it for $1,500 a month, and depleting stores for people who have such severe obesity that they have congestive heart failure and diabetes,” Dr. Apovian says. “These are people who are going to die, and you’re taking it away just for cosmetic weight loss. That is deplorable.”
In addition to huge demand, Wegovy also had a disruption in its supply chain. Right now, it isn’t available at all in lower doses, which is helping to spike off-label demand for Ozempic. Novo Nordisk expects to have these problems sorted out by the end of the year, with distribution following soon after.
The price of access
With a list price of $1,350 a month, Wegovy costs as much as many mortgages. And Medicaid, Medicare, and many insurance companies don’t cover it. Although obesity is a disease, the insurance industry treats weight loss as more of a vanity issue – so even if you could find the drug, you might not be able to afford it.
“We’re seeing that roughly half the prescriptions we write aren’t being covered,” Dr. Apovian says. “And for the half that are covered, we have to do prior authorization, which takes days, and it’s laborious.” In some instances, she says, insurance companies withdraw authorization after 3 months if they don’t see enough weight coming off.
It’s not like you can take Wegovy for 3 months, lose some weight, and expect it to stay off, either. The medication requires a real commitment, potentially for life. That’s because once the semaglutide leaves your system, your appetite returns. In one study, people regained two-thirds of the weight they’d lost within a year of stopping.
Many see a double standard in the insurance companies’ refusal to cover a drug that could prevent serious illness or death.
“They’re saying it’s not cost-effective to give the 42% of Americans who have a BMI over 30 Wegovy. Did they say this when statins came out?” Dr. Apovian says. “Why are they doing this with antiobesity agents? It’s the culture. The culture isn’t ready to adopt obesity as the disease that it is.”
Unpleasant side effects
Let’s assume you’re one of the lucky ones – your insurance covers Wegovy, and you can actually find some. You might discover that using it is no walk in the park. Common side effects include gastrointestinal issues like nausea, vomiting, and diarrhea.
“The way we counteract that is to start very slowly at a low dose of these medications,” Dr. Apovian says. “We only go up when the patient doesn’t have nausea or it gets better.”
Elise Davenport was excited to try Wegovy. “I did my online research. I’m the type who’s interested in early adoption, tech gadgets and stuff,” says the 40-year-old technical writer. “I wanted to try it because I’d tried so many other things that failed, or hadn’t worked long-term.”
With a BMI over 30, Ms. Davenport qualified for the drug. She signed up for an online program that guaranteed insurance coverage and started taking it in October 2021. At first, the side effects were mild, just a touch of nausea and diarrhea. And the results were impressive. She found it easy to feel satisfied with smaller portions and lost her cravings for sugar and highly processed foods. The weight fell off, roughly 5 pounds a week.
It turns out, that’s too much, too fast. Dr. Apovian and Dr. Velazquez say their patients lose more like 2 pounds each week, with careful monitoring.
By early December, Ms. Davenport’s side effects were ramping up. Because of shortages in lower dosages, the online program wasn’t able to adjust hers right away. She felt nauseated all the time, bad enough that brushing her teeth made her vomit and she had to force herself to eat. Some weeks, she managed less than 500 calories a day. Her sleep patterns became erratic. And then her depression, which medication had kept under control for years, spiraled.
“I remember sitting on the floor of my bathroom crying, thinking I’d rather carry the extra weight,” she says. “I used to take a lot of enjoyment from food, and I had none of that anymore. It was such a joyless experience at that point.”
Eventually, her dosage was reduced and the symptoms let up, but her primary care doctor encouraged her to stop. By the time she did, in March, she’d lost 55 pounds. So far, she’s gained back about 10.
More than just weight loss
Even though Ms. Davenport’s experience wasn’t a good one, with better monitoring, she’d be willing to try again. For one thing, seeing how easy it was to eat less with medical help helped to undo years of shame.
“Our culture treats obesity like a moral failing. I realized I’d been made to feel that way by doctors and programs – that I wasn’t doing enough,” she says. “This drug made me realize there are legit physiological things going on in my body, things that are often excluded from the conversation.”
Dr. Apovian and Dr. Velazquez say their patients regularly discover similar things.
“Obesity is not a disease of willpower. Medications are not the easy way out,” Dr. Velazquez says. “This is a chronic, relapsing medical condition, and because of that, we should treat it how we treat diabetes, high blood pressure, all these other conditions. We’d never hold back medication for individuals coming in with high blood pressure, tell them to work on willpower and withhold drugs they’d qualify for.”
A version of this article first appeared on WebMD.com.
Both potatoes and beans reduced insulin resistance, weight in controlled study
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
Low energy–density diets that are based either on potatoes or beans similarly reduced insulin resistance in adults with poor blood glucose control, according to a controlled feeding study in 36 individuals.
Potatoes have gotten a bad rap for their high glycemic index, but they have little fat and a low energy density, wrote the study investigators. In fact, “cooling of gelatinized potatoes generates appreciable levels of slowly digested starch (resistant starch type 3) and substantially lowers the blood glucose response that potatoes elicit.”
“There is a view that potatoes are a less healthy plant food, but there is very little empirical data from randomized trials to support this view,” senior investigator John P. Kirwan, PhD, said in an interview.
Dry beans and peas (known as pulses) also contain resistant starch that improves insulin sensitivity and glucose tolerance, and multiple studies support pulses as part of a low-glycemic diet to improve glucose control in adults, the researchers explained, but because the density of food often guides how much people eat, they hypothesized that potatoes could substitute for beans and provide similar glucose control benefits.
In a study published in the Journal of Medicinal Food, the researchers randomized 36 adults aged 18-60 years with insulin resistance to 8 weeks of a low energy–density diet (1 kcal/g) high in either potatoes or beans. The baseline body mass index ranged from 25 to 40 kg/m2. Insulin resistance was defined using the homeostatic model assessment of insulin resistance (HOMA-IR) with a score greater than 2.
The controlled diet consisted of 50%-55% carbohydrates, 30%-35% fats, and 15%-20% protein. Each meal in the potato group included a side of potatoes, and each meal in the bean group included a side of beans.
The primary outcome was the mean change in blood glucose concentration; the researchers also assessed weight loss.
A total of 14 individuals in the potato group and 17 in the bean group completed the study; but data from the 18 individuals in each group were included in an intent-to-treat analysis.
Among study completers, HOMA-IR in the bean group showed an average decrease of 1.4 from baseline (P = .02 ); a similar decrease of 1.3 occurred in the potato group (P < .05) with no significant difference between the two diets.
Overall compliance with both diets was roughly 88%. Body weight reductions were similar in both groups and significantly reduced from baseline over the study period, with average reductions in intent-to-treat analysis of 5.82 kg in the potato group and 4.0 kg in the bean group. BMI also was significantly reduced from baseline in both potato and bean groups (2.04 kg/m2 and 1.35 kg/m2, respectively). Although baseline differences were not significant, “BMI at baseline was higher and the reduction in response to the treatment was significantly greater in the potato diet compared with the bean diet,” the researchers noted. The effect on blood glucose response was not significantly different between the two groups or from baseline, they said.
The findings were limited by several factors including the small size, relatively short study period, and controlled nature of the study diet, the researchers noted. “The addition of a typical Western diet would have enhanced our understanding of the effect of low energy–dense diets on metabolic outcomes,” they noted in their discussion.
However, both diets led to a reduction in body weight, and the low energy density of both potato and bean diets promoted weight loss without affecting appetite or requiring calorie restriction, the researchers explained. Therefore, “this weight loss if sustained over time could have a substantial impact on body weight,” they said.
“We hypothesized that there would be equivalence between the potato and bean diet and this hypothesis proved to be correct,” said Dr. Kirwan, of the Pennington Biomedical Research Center, Baton Rouge, La., in an interview.
The take-home message for clinicians is that, though small, the study was very well-controlled, Dr. Kirwan emphasized. “Clinicians ought to consider the health benefits of the potato when it is cooked and served appropriately.”
Looking ahead, larger randomized controlled trials with additional control arms, longer time of at least 12 weeks, and different patient populations are needed, Dr. Kirwan added.
Findings mitigate food myths
The debate continues about whether there are foods that are “good” or “evil;” or foods that one “should not eat” or “should eat,” said Amy Rothberg, MD, associate professor of internal medicine and of nutritional sciences at the University of Michigan, Ann Arbor, in an interview.
“This study dispels the myth that incorporating a small portion of potato into the diet (although these are not potatoes that are fried, or are topped with cheese, bacon, sour cream, etc.) results in deleterious metabolic outcomes when compared to a diet that is comprised of beans (pulses) as part of a low energy–dense diet,” she explained.
“The diet in both groups was of low energy density, which has been shown to result in fewer calories consumed, weight loss, and improvement in insulin resistance,” so the similarity in results was not so surprising, said Dr. Rothberg.
For the clinical takeaway, Dr. Rothberg agreed with the study authors: “Clinicians may counsel their patients that they can still consume a small potato (with the caveat above regarding cooking methods and toppings) as part of a balanced meal so long as they are keeping their overall calories low and not exceeding their metabolic requirements based on body weight/BMI,” she said.
As for additional research, studies with a longer time frame and a larger and more diverse study population are needed, including populations with common insulin resistance comorbidities such as type 2 diabetes, fatty liver disease, and cardiovascular disease, Dr. Rothberg noted.
Consumer considerations, with caveats
The key message for consumers is that, “based on this very small study of short duration, consuming a small portion of potato as part of an overall balanced, low-energy diet did not produce adverse effects on glucose or insulin when compared to a diet of pulses known to have favorable effects on glucose and insulin,” Dr. Rothberg told this news organization. However, “consumers should note that, although the results from this small study are encouraging, it would be premature to extrapolate the findings from this study to other populations,” she said. Also, keep in mind that the study was supported in part by the Alliance for Potato Research, although the authors stated that none of the funders (Alliance for Potato Research and Education and the National Institutes of Health) had any role in the design, analysis, or writing of the article, she added.
The study was supported in part by the Alliance for Potato Research and Education and the National Institutes of Health, which funds the Louisiana Clinical and Translational Science Center. The researchers and Dr. Rothberg had no financial conflicts to disclose.
FROM THE JOURNAL OF MEDICINAL FOOD
Obesity tied to worse brain health in children
CHICAGO – Higher weight and body mass index (BMI) in preadolescents are linked with poor brain health, new research suggests.
Poor brain health has been linked with obesity in adults, but little has been known about the link in children.
Lead author Simone Kaltenhauser, a postgraduate research fellow in radiology and biomedical imaging at the Yale School of Medicine, New Haven, Conn., presented her findings at the annual meeting of the Radiological Society of North America.
To represent the national sociodemographic makeup, the researchers used baseline information from the Adolescent Brain Cognitive Development (ABCD) study, which included 11,878 children aged 9 and 10 years from 21 centers across the United States.
This study included 5,169 children (51.9% girls). Children who had had traumatic brain injury, eating disorders, neurodevelopmental problems, and psychiatric diseases were excluded from the final analysis.
The researchers analyzed information from structural MRI and resting-state functional MRI, which allowed them to measure brain activity by detecting changes in blood flow.
“We analyzed the average fractional anisotropy (FA), mean (MD), axial (AD) and radial diffusivity (RD), and neurite density (ND) of 35 white matter (WM) tracts; cortical thickness and surface of 68 regions; and functional connectivity of 91 predefined network correlations,” the authors explained.
They adjusted for age, sex, race/ethnicity, handedness, and socioeconomic status. They used linear models to determine associations between weight and BMI z-scores and the imaging metrics.
Loss of white matter integrity
Among children with obesity, there was pervasive loss of white matter integrity and neurite density, cortical gray matter thinning, and decreased connectivity within and between networks that have been associated with impulse control and reward-based decision-making.
“These changes persisted in a similar pattern also 2 years later,” she said.
A member of the audience asked whether a reverse relationship might be at work – that poor brain health might drive obesity rather than the other way around.
Ms. Kaltenhauser agreed that other factors could be driving the link and acknowledged as a limitation that they did not have access to genetic information on the children.
Information on the effects of overweight and obesity is critical, especially in the United States, where an estimated 1 in 5 children are obese.
Ms. Kaltenhauser said she hopes her study raises awareness of potential brain health consequences as well as the physical consequences of childhood obesity.
Senior author Sam Payabvash, MD, a neuroradiologist and assistant professor of radiology and biomedical imaging at the Yale School of Medicine, said in a press release that the study may help explain the findings from previous studies that show an association between higher BMI in children and poor cognitive functioning and academic performance.
“The longitudinal ABCD study gives us the opportunity to observe any changes that occur in children with higher weight and BMI z-scores,” Dr. Payabvash said. “We’ll need to watch over the next 6-10 years.”
Avenues for future research
Amit B. Desai, MD, a neuroradiologist with Mayo Clinic in Jacksonville, Fla., who was not part of the study, said that while the research demonstrates an association between brain structure and BMI and obesity, “there may be other lurking variables.”
“What’s happening at an earlier stage in life could be causing both,” he said.
He noted that he would like to see future studies involving children at even earlier ages, along with investigations of the role of genetics or socioeconomic factors.
Including older children would be interesting as well, he said.
“Myelination doesn’t complete until you’re in your late teens or early 20s, so there are structural changes happening in the brain much later on into adolescence and early adulthood,” Dr. Desai said.
It would be premature, he said, to conclude from these findings that if children have obesity, there must be something wrong with their brain.
He would like to see whether there are changes in this link if a child is obese early on and later has normal weight or if a child has normal weight early and then becomes obese.
“It’s definitely an eye-opening study, but [it] needs additional work to expand upon it,” he said.
Ms. Kaltenhauser and Dr. Desai report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHICAGO – Higher weight and body mass index (BMI) in preadolescents are linked with poor brain health, new research suggests.
Poor brain health has been linked with obesity in adults, but little has been known about the link in children.
Lead author Simone Kaltenhauser, a postgraduate research fellow in radiology and biomedical imaging at the Yale School of Medicine, New Haven, Conn., presented her findings at the annual meeting of the Radiological Society of North America.
To represent the national sociodemographic makeup, the researchers used baseline information from the Adolescent Brain Cognitive Development (ABCD) study, which included 11,878 children aged 9 and 10 years from 21 centers across the United States.
This study included 5,169 children (51.9% girls). Children who had had traumatic brain injury, eating disorders, neurodevelopmental problems, and psychiatric diseases were excluded from the final analysis.
The researchers analyzed information from structural MRI and resting-state functional MRI, which allowed them to measure brain activity by detecting changes in blood flow.
“We analyzed the average fractional anisotropy (FA), mean (MD), axial (AD) and radial diffusivity (RD), and neurite density (ND) of 35 white matter (WM) tracts; cortical thickness and surface of 68 regions; and functional connectivity of 91 predefined network correlations,” the authors explained.
They adjusted for age, sex, race/ethnicity, handedness, and socioeconomic status. They used linear models to determine associations between weight and BMI z-scores and the imaging metrics.
Loss of white matter integrity
Among children with obesity, there was pervasive loss of white matter integrity and neurite density, cortical gray matter thinning, and decreased connectivity within and between networks that have been associated with impulse control and reward-based decision-making.
“These changes persisted in a similar pattern also 2 years later,” she said.
A member of the audience asked whether a reverse relationship might be at work – that poor brain health might drive obesity rather than the other way around.
Ms. Kaltenhauser agreed that other factors could be driving the link and acknowledged as a limitation that they did not have access to genetic information on the children.
Information on the effects of overweight and obesity is critical, especially in the United States, where an estimated 1 in 5 children are obese.
Ms. Kaltenhauser said she hopes her study raises awareness of potential brain health consequences as well as the physical consequences of childhood obesity.
Senior author Sam Payabvash, MD, a neuroradiologist and assistant professor of radiology and biomedical imaging at the Yale School of Medicine, said in a press release that the study may help explain the findings from previous studies that show an association between higher BMI in children and poor cognitive functioning and academic performance.
“The longitudinal ABCD study gives us the opportunity to observe any changes that occur in children with higher weight and BMI z-scores,” Dr. Payabvash said. “We’ll need to watch over the next 6-10 years.”
Avenues for future research
Amit B. Desai, MD, a neuroradiologist with Mayo Clinic in Jacksonville, Fla., who was not part of the study, said that while the research demonstrates an association between brain structure and BMI and obesity, “there may be other lurking variables.”
“What’s happening at an earlier stage in life could be causing both,” he said.
He noted that he would like to see future studies involving children at even earlier ages, along with investigations of the role of genetics or socioeconomic factors.
Including older children would be interesting as well, he said.
“Myelination doesn’t complete until you’re in your late teens or early 20s, so there are structural changes happening in the brain much later on into adolescence and early adulthood,” Dr. Desai said.
It would be premature, he said, to conclude from these findings that if children have obesity, there must be something wrong with their brain.
He would like to see whether there are changes in this link if a child is obese early on and later has normal weight or if a child has normal weight early and then becomes obese.
“It’s definitely an eye-opening study, but [it] needs additional work to expand upon it,” he said.
Ms. Kaltenhauser and Dr. Desai report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
CHICAGO – Higher weight and body mass index (BMI) in preadolescents are linked with poor brain health, new research suggests.
Poor brain health has been linked with obesity in adults, but little has been known about the link in children.
Lead author Simone Kaltenhauser, a postgraduate research fellow in radiology and biomedical imaging at the Yale School of Medicine, New Haven, Conn., presented her findings at the annual meeting of the Radiological Society of North America.
To represent the national sociodemographic makeup, the researchers used baseline information from the Adolescent Brain Cognitive Development (ABCD) study, which included 11,878 children aged 9 and 10 years from 21 centers across the United States.
This study included 5,169 children (51.9% girls). Children who had had traumatic brain injury, eating disorders, neurodevelopmental problems, and psychiatric diseases were excluded from the final analysis.
The researchers analyzed information from structural MRI and resting-state functional MRI, which allowed them to measure brain activity by detecting changes in blood flow.
“We analyzed the average fractional anisotropy (FA), mean (MD), axial (AD) and radial diffusivity (RD), and neurite density (ND) of 35 white matter (WM) tracts; cortical thickness and surface of 68 regions; and functional connectivity of 91 predefined network correlations,” the authors explained.
They adjusted for age, sex, race/ethnicity, handedness, and socioeconomic status. They used linear models to determine associations between weight and BMI z-scores and the imaging metrics.
Loss of white matter integrity
Among children with obesity, there was pervasive loss of white matter integrity and neurite density, cortical gray matter thinning, and decreased connectivity within and between networks that have been associated with impulse control and reward-based decision-making.
“These changes persisted in a similar pattern also 2 years later,” she said.
A member of the audience asked whether a reverse relationship might be at work – that poor brain health might drive obesity rather than the other way around.
Ms. Kaltenhauser agreed that other factors could be driving the link and acknowledged as a limitation that they did not have access to genetic information on the children.
Information on the effects of overweight and obesity is critical, especially in the United States, where an estimated 1 in 5 children are obese.
Ms. Kaltenhauser said she hopes her study raises awareness of potential brain health consequences as well as the physical consequences of childhood obesity.
Senior author Sam Payabvash, MD, a neuroradiologist and assistant professor of radiology and biomedical imaging at the Yale School of Medicine, said in a press release that the study may help explain the findings from previous studies that show an association between higher BMI in children and poor cognitive functioning and academic performance.
“The longitudinal ABCD study gives us the opportunity to observe any changes that occur in children with higher weight and BMI z-scores,” Dr. Payabvash said. “We’ll need to watch over the next 6-10 years.”
Avenues for future research
Amit B. Desai, MD, a neuroradiologist with Mayo Clinic in Jacksonville, Fla., who was not part of the study, said that while the research demonstrates an association between brain structure and BMI and obesity, “there may be other lurking variables.”
“What’s happening at an earlier stage in life could be causing both,” he said.
He noted that he would like to see future studies involving children at even earlier ages, along with investigations of the role of genetics or socioeconomic factors.
Including older children would be interesting as well, he said.
“Myelination doesn’t complete until you’re in your late teens or early 20s, so there are structural changes happening in the brain much later on into adolescence and early adulthood,” Dr. Desai said.
It would be premature, he said, to conclude from these findings that if children have obesity, there must be something wrong with their brain.
He would like to see whether there are changes in this link if a child is obese early on and later has normal weight or if a child has normal weight early and then becomes obese.
“It’s definitely an eye-opening study, but [it] needs additional work to expand upon it,” he said.
Ms. Kaltenhauser and Dr. Desai report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT RSNA 2022





