User login
Bariatric surgery may up risk for epilepsy
Analyzing health records, investigators compared almost 17,000 patients who had undergone bariatric surgery with more than 620,000 individuals with obesity who had not undergone the surgery.
During a minimum 3-year follow-up period, the surgery group had a 45% higher risk of developing epilepsy than the nonsurgery group. Moreover, patients who had a stroke after their bariatric surgery were 14 times more likely to develop epilepsy than those who did not have a stroke.
“When considering having bariatric surgery, people should talk to their doctors about the benefits and risks,” senior investigator Jorge Burneo, MD, professor of neurology, biostatistics, and epidemiology and endowed chair in epilepsy at Western University, London, told this news organization.
“While there are many health benefits of weight loss, our findings suggest that epilepsy is a long-term risk of bariatric surgery for weight loss,” Dr. Burneo said.
The findings were published online in Neurology.
Unrecognized risk factor?
Bariatric surgery has become more common as global rates of obesity have increased. The surgery has been shown to reduce the risk for serious obesity-related conditions, the researchers note.
However, “in addition to the positive outcomes of bariatric surgery, several long-term neurological complications have also been identified,” they write.
One previous study reported increased epilepsy risk following gastric bypass. Those findings “suggest that bariatric surgery may be an unrecognized epilepsy risk factor; however, this possible association has not been thoroughly explored,” write the investigators.
Dr. Burneo said he conducted the study because he has seen patients with epilepsy in his clinic who were “without risk factors, with normal MRIs, who shared the history of having bariatric surgery before the development of epilepsy.”
The researchers’ primary objective was to “assess whether epilepsy risk is elevated following bariatric surgery for weight loss relative to a nonsurgical cohort of patients who are obese,” he noted.
The study used linked administrative health databases in Ontario, Canada. Patients were accrued from July 1, 2010, to Dec. 31, 2016, and were followed until Dec. 31, 2019. The analysis included 639,472 participants, 2.7% of whom had undergone bariatric surgery.
The “exposed” cohort consisted of all Ontario residents aged 18 years or older who had undergone bariatric surgery during the 6-year period (n = 16,958; 65.1% women; mean age, 47.4 years), while the “unexposed” cohort consisted of patients hospitalized with a diagnosis of obesity who had not undergone bariatric surgery (n = 622,514; 62.8% women; mean age, 47.6 years).
Patients with a history of seizures, epilepsy, epilepsy risk factors, prior brain surgery, psychiatric disorders, or drug or alcohol abuse/dependence were excluded from the analysis.
The researchers collected data on patients’ sociodemographic characteristics at the index date, as well as Charlson Comorbidity Index scores during the 2 years prior to index, and data regarding several specific comorbidities, such as diabetes mellitus, hypertension, sleep apnea, depression/anxiety, and cardiovascular factors.
The exposed and unexposed cohorts were followed for a median period of 5.8 and 5.9 person-years, respectively.
‘Unclear’ mechanisms
Before weighting, 0.4% of participants in the exposed cohort (n = 73) developed epilepsy, versus 0.2% of participants in the unexposed cohort (n = 1,260) by the end of the follow-up period.
In the weighted cohorts, there were 50.1 epilepsy diagnoses per 100,000 person-years, versus 34.1 per 100,000 person-years (rate difference, 16 per 100,000 person-years).
The multivariable analysis of the weighted cohort showed the hazard ratio for epilepsy cases that were associated with bariatric surgery was 1.45 (95% confidence interval, 1.35-1.56), after adjusting for sleep apnea and including stroke as a time-varying covariate.
Having a stroke during the follow-up period increased epilepsy 14-fold in the exposed cohort (HR, 14.03; 95% CI, 4.25-46.25).
The investigators note that they were unable to measure obesity status or body mass index throughout the study and that some obesity-related comorbidities “may affect epilepsy risk.”
In addition, Dr. Burneo reported that the study did not investigate potential causes and mechanisms of the association between bariatric surgery and epilepsy risk.
Hypotheses “include potential nutritional deficiencies, receipt of general anesthesia, or other unclear causes,” he said.
“Future research should investigate epilepsy as a potential long-term complication of bariatric surgery, exploring the possible effects of this procedure,” Dr. Burneo added.
Risk-benefit discussion
In a comment, Jacqueline French, MD, professor of neurology at NYU Grossman School of Medicine, and director of NYU’s Epilepsy Study Consortium, said she was “not 100% surprised by the findings” because she has seen in her clinical practice “a number of patients who developed epilepsy after bariatric surgery or had a history of bariatric surgery at the time they developed epilepsy.”
On the other hand, she has also seen patients who did not have a history of bariatric surgery and who developed epilepsy.
“I’m unable to tell if there is an association, although I’ve had it at the back of my head as a thought and wondered about it,” said Dr. French, who is also the chief medical and innovation officer at the Epilepsy Foundation. She was not involved with the study.
She noted that possible mechanisms underlying the association are that gastric bypass surgery leads to a “significant alteration” in nutrient absorption. Moreover, “we now know that the microbiome is associated with epilepsy” and that changes occur in the gut microbiome after bariatric surgery, Dr. French said.
There are two take-home messages for practicing clinicians, she added.
“Although the risk [of developing epilepsy] is very low, it should be presented as part of the risks and benefits to patients considering bariatric surgery,” she said.
“It’s equally important to follow up on the potential differences in these patients who go on to develop epilepsy following bariatric surgery,” said Dr. French. “Is there a certain metabolic profile or some nutrient previously absorbed that now is not absorbed that might predispose people to risk?”
This would be “enormously important to know because it might not just pertain to these people but to a whole other cohort of people who develop epilepsy,” Dr. French concluded.
The study was funded by the Ontario Ministry of Health and Ministry of Long-Term Care and by the Jack Cowin Endowed Chair in Epilepsy Research at Western University. Dr. Burneo holds the Jack Cowin Endowed Chair in Epilepsy Research at Western University. The other investigators and Dr. French have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Analyzing health records, investigators compared almost 17,000 patients who had undergone bariatric surgery with more than 620,000 individuals with obesity who had not undergone the surgery.
During a minimum 3-year follow-up period, the surgery group had a 45% higher risk of developing epilepsy than the nonsurgery group. Moreover, patients who had a stroke after their bariatric surgery were 14 times more likely to develop epilepsy than those who did not have a stroke.
“When considering having bariatric surgery, people should talk to their doctors about the benefits and risks,” senior investigator Jorge Burneo, MD, professor of neurology, biostatistics, and epidemiology and endowed chair in epilepsy at Western University, London, told this news organization.
“While there are many health benefits of weight loss, our findings suggest that epilepsy is a long-term risk of bariatric surgery for weight loss,” Dr. Burneo said.
The findings were published online in Neurology.
Unrecognized risk factor?
Bariatric surgery has become more common as global rates of obesity have increased. The surgery has been shown to reduce the risk for serious obesity-related conditions, the researchers note.
However, “in addition to the positive outcomes of bariatric surgery, several long-term neurological complications have also been identified,” they write.
One previous study reported increased epilepsy risk following gastric bypass. Those findings “suggest that bariatric surgery may be an unrecognized epilepsy risk factor; however, this possible association has not been thoroughly explored,” write the investigators.
Dr. Burneo said he conducted the study because he has seen patients with epilepsy in his clinic who were “without risk factors, with normal MRIs, who shared the history of having bariatric surgery before the development of epilepsy.”
The researchers’ primary objective was to “assess whether epilepsy risk is elevated following bariatric surgery for weight loss relative to a nonsurgical cohort of patients who are obese,” he noted.
The study used linked administrative health databases in Ontario, Canada. Patients were accrued from July 1, 2010, to Dec. 31, 2016, and were followed until Dec. 31, 2019. The analysis included 639,472 participants, 2.7% of whom had undergone bariatric surgery.
The “exposed” cohort consisted of all Ontario residents aged 18 years or older who had undergone bariatric surgery during the 6-year period (n = 16,958; 65.1% women; mean age, 47.4 years), while the “unexposed” cohort consisted of patients hospitalized with a diagnosis of obesity who had not undergone bariatric surgery (n = 622,514; 62.8% women; mean age, 47.6 years).
Patients with a history of seizures, epilepsy, epilepsy risk factors, prior brain surgery, psychiatric disorders, or drug or alcohol abuse/dependence were excluded from the analysis.
The researchers collected data on patients’ sociodemographic characteristics at the index date, as well as Charlson Comorbidity Index scores during the 2 years prior to index, and data regarding several specific comorbidities, such as diabetes mellitus, hypertension, sleep apnea, depression/anxiety, and cardiovascular factors.
The exposed and unexposed cohorts were followed for a median period of 5.8 and 5.9 person-years, respectively.
‘Unclear’ mechanisms
Before weighting, 0.4% of participants in the exposed cohort (n = 73) developed epilepsy, versus 0.2% of participants in the unexposed cohort (n = 1,260) by the end of the follow-up period.
In the weighted cohorts, there were 50.1 epilepsy diagnoses per 100,000 person-years, versus 34.1 per 100,000 person-years (rate difference, 16 per 100,000 person-years).
The multivariable analysis of the weighted cohort showed the hazard ratio for epilepsy cases that were associated with bariatric surgery was 1.45 (95% confidence interval, 1.35-1.56), after adjusting for sleep apnea and including stroke as a time-varying covariate.
Having a stroke during the follow-up period increased epilepsy 14-fold in the exposed cohort (HR, 14.03; 95% CI, 4.25-46.25).
The investigators note that they were unable to measure obesity status or body mass index throughout the study and that some obesity-related comorbidities “may affect epilepsy risk.”
In addition, Dr. Burneo reported that the study did not investigate potential causes and mechanisms of the association between bariatric surgery and epilepsy risk.
Hypotheses “include potential nutritional deficiencies, receipt of general anesthesia, or other unclear causes,” he said.
“Future research should investigate epilepsy as a potential long-term complication of bariatric surgery, exploring the possible effects of this procedure,” Dr. Burneo added.
Risk-benefit discussion
In a comment, Jacqueline French, MD, professor of neurology at NYU Grossman School of Medicine, and director of NYU’s Epilepsy Study Consortium, said she was “not 100% surprised by the findings” because she has seen in her clinical practice “a number of patients who developed epilepsy after bariatric surgery or had a history of bariatric surgery at the time they developed epilepsy.”
On the other hand, she has also seen patients who did not have a history of bariatric surgery and who developed epilepsy.
“I’m unable to tell if there is an association, although I’ve had it at the back of my head as a thought and wondered about it,” said Dr. French, who is also the chief medical and innovation officer at the Epilepsy Foundation. She was not involved with the study.
She noted that possible mechanisms underlying the association are that gastric bypass surgery leads to a “significant alteration” in nutrient absorption. Moreover, “we now know that the microbiome is associated with epilepsy” and that changes occur in the gut microbiome after bariatric surgery, Dr. French said.
There are two take-home messages for practicing clinicians, she added.
“Although the risk [of developing epilepsy] is very low, it should be presented as part of the risks and benefits to patients considering bariatric surgery,” she said.
“It’s equally important to follow up on the potential differences in these patients who go on to develop epilepsy following bariatric surgery,” said Dr. French. “Is there a certain metabolic profile or some nutrient previously absorbed that now is not absorbed that might predispose people to risk?”
This would be “enormously important to know because it might not just pertain to these people but to a whole other cohort of people who develop epilepsy,” Dr. French concluded.
The study was funded by the Ontario Ministry of Health and Ministry of Long-Term Care and by the Jack Cowin Endowed Chair in Epilepsy Research at Western University. Dr. Burneo holds the Jack Cowin Endowed Chair in Epilepsy Research at Western University. The other investigators and Dr. French have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Analyzing health records, investigators compared almost 17,000 patients who had undergone bariatric surgery with more than 620,000 individuals with obesity who had not undergone the surgery.
During a minimum 3-year follow-up period, the surgery group had a 45% higher risk of developing epilepsy than the nonsurgery group. Moreover, patients who had a stroke after their bariatric surgery were 14 times more likely to develop epilepsy than those who did not have a stroke.
“When considering having bariatric surgery, people should talk to their doctors about the benefits and risks,” senior investigator Jorge Burneo, MD, professor of neurology, biostatistics, and epidemiology and endowed chair in epilepsy at Western University, London, told this news organization.
“While there are many health benefits of weight loss, our findings suggest that epilepsy is a long-term risk of bariatric surgery for weight loss,” Dr. Burneo said.
The findings were published online in Neurology.
Unrecognized risk factor?
Bariatric surgery has become more common as global rates of obesity have increased. The surgery has been shown to reduce the risk for serious obesity-related conditions, the researchers note.
However, “in addition to the positive outcomes of bariatric surgery, several long-term neurological complications have also been identified,” they write.
One previous study reported increased epilepsy risk following gastric bypass. Those findings “suggest that bariatric surgery may be an unrecognized epilepsy risk factor; however, this possible association has not been thoroughly explored,” write the investigators.
Dr. Burneo said he conducted the study because he has seen patients with epilepsy in his clinic who were “without risk factors, with normal MRIs, who shared the history of having bariatric surgery before the development of epilepsy.”
The researchers’ primary objective was to “assess whether epilepsy risk is elevated following bariatric surgery for weight loss relative to a nonsurgical cohort of patients who are obese,” he noted.
The study used linked administrative health databases in Ontario, Canada. Patients were accrued from July 1, 2010, to Dec. 31, 2016, and were followed until Dec. 31, 2019. The analysis included 639,472 participants, 2.7% of whom had undergone bariatric surgery.
The “exposed” cohort consisted of all Ontario residents aged 18 years or older who had undergone bariatric surgery during the 6-year period (n = 16,958; 65.1% women; mean age, 47.4 years), while the “unexposed” cohort consisted of patients hospitalized with a diagnosis of obesity who had not undergone bariatric surgery (n = 622,514; 62.8% women; mean age, 47.6 years).
Patients with a history of seizures, epilepsy, epilepsy risk factors, prior brain surgery, psychiatric disorders, or drug or alcohol abuse/dependence were excluded from the analysis.
The researchers collected data on patients’ sociodemographic characteristics at the index date, as well as Charlson Comorbidity Index scores during the 2 years prior to index, and data regarding several specific comorbidities, such as diabetes mellitus, hypertension, sleep apnea, depression/anxiety, and cardiovascular factors.
The exposed and unexposed cohorts were followed for a median period of 5.8 and 5.9 person-years, respectively.
‘Unclear’ mechanisms
Before weighting, 0.4% of participants in the exposed cohort (n = 73) developed epilepsy, versus 0.2% of participants in the unexposed cohort (n = 1,260) by the end of the follow-up period.
In the weighted cohorts, there were 50.1 epilepsy diagnoses per 100,000 person-years, versus 34.1 per 100,000 person-years (rate difference, 16 per 100,000 person-years).
The multivariable analysis of the weighted cohort showed the hazard ratio for epilepsy cases that were associated with bariatric surgery was 1.45 (95% confidence interval, 1.35-1.56), after adjusting for sleep apnea and including stroke as a time-varying covariate.
Having a stroke during the follow-up period increased epilepsy 14-fold in the exposed cohort (HR, 14.03; 95% CI, 4.25-46.25).
The investigators note that they were unable to measure obesity status or body mass index throughout the study and that some obesity-related comorbidities “may affect epilepsy risk.”
In addition, Dr. Burneo reported that the study did not investigate potential causes and mechanisms of the association between bariatric surgery and epilepsy risk.
Hypotheses “include potential nutritional deficiencies, receipt of general anesthesia, or other unclear causes,” he said.
“Future research should investigate epilepsy as a potential long-term complication of bariatric surgery, exploring the possible effects of this procedure,” Dr. Burneo added.
Risk-benefit discussion
In a comment, Jacqueline French, MD, professor of neurology at NYU Grossman School of Medicine, and director of NYU’s Epilepsy Study Consortium, said she was “not 100% surprised by the findings” because she has seen in her clinical practice “a number of patients who developed epilepsy after bariatric surgery or had a history of bariatric surgery at the time they developed epilepsy.”
On the other hand, she has also seen patients who did not have a history of bariatric surgery and who developed epilepsy.
“I’m unable to tell if there is an association, although I’ve had it at the back of my head as a thought and wondered about it,” said Dr. French, who is also the chief medical and innovation officer at the Epilepsy Foundation. She was not involved with the study.
She noted that possible mechanisms underlying the association are that gastric bypass surgery leads to a “significant alteration” in nutrient absorption. Moreover, “we now know that the microbiome is associated with epilepsy” and that changes occur in the gut microbiome after bariatric surgery, Dr. French said.
There are two take-home messages for practicing clinicians, she added.
“Although the risk [of developing epilepsy] is very low, it should be presented as part of the risks and benefits to patients considering bariatric surgery,” she said.
“It’s equally important to follow up on the potential differences in these patients who go on to develop epilepsy following bariatric surgery,” said Dr. French. “Is there a certain metabolic profile or some nutrient previously absorbed that now is not absorbed that might predispose people to risk?”
This would be “enormously important to know because it might not just pertain to these people but to a whole other cohort of people who develop epilepsy,” Dr. French concluded.
The study was funded by the Ontario Ministry of Health and Ministry of Long-Term Care and by the Jack Cowin Endowed Chair in Epilepsy Research at Western University. Dr. Burneo holds the Jack Cowin Endowed Chair in Epilepsy Research at Western University. The other investigators and Dr. French have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NEUROLOGY
High Risk of Long COVID Neurologic Sequelae in Veterans
We now know that the effects of COVID-19 don’t always end when the infection seems over. Long COVID—the postacute sequelae—can encompass a wide range of extrapulmonary organ dysfunctions. Most studies on COVID-19 have had follow-ups of 6 months or less with a narrow selection of neurologic outcomes, say Evan Xu, Yan Xie, PhD, and Ziyad Al-Aly, MD of the US Department of Veterans Affairs (VA) St. Louis Health Care System in Missouri. The 12-month study of 11,652,484 people published in Nature Medicine sounds an alert: Get ready to care for more patients with long-term, even chronic, neurologic disorders from migraine to stroke.
The researchers “leveraged the breadth and depth” of the VA’s national health care databases to build 3 groups: 154,068 people who survived the first 30 days of COVID-19; 5,638,795 VA users with no evidence of COVID-19 infection; and 5,859,621 VA users during 2017 (ie, prepandemic). Altogether, the groups corresponded to 14,064,985 person-years of follow-up.
The findings, which the researchers termed robust, revealed substantial risks and burdens beyond the first 30 days of COVID-19 infection, including “an array of neurologic disorders spanning several disease categories.”
Patients were at greater risk for stroke (both ischemic and hemorrhagic), cognition and memory disorders, peripheral nervous system disorders, episodic disorders like migraine and seizures, extrapyramidal and movement disorders, mental health disorders, musculoskeletal disorders, sensory disorders, Guillain-Barré syndrome, and encephalitis or encephalopathy.
The researchers estimated the hazard ratio of any neurological sequelae as 1.42. The risks were elevated even in people who did not require hospitalization during acute COVID-19 and increased according to the care setting of the acute phase of the disease from nonhospitalized to hospitalized and admitted to intensive care.
“Given the colossal scale of the pandemic,” the researchers say, governments and health systems should consider these findings when devising policy for continued management and developing plans for a postpandemic world. Some of the disorders they report on, they note, “are serious chronic conditions that will impact some people for a lifetime.” They point to 2 key findings: first, regardless of age, people with COVID-19 had a higher risk of all the neurologic outcomes examined, and second, the analyses suggest that the effects on risk were stronger in younger adults.
“The effects of these disorders on younger lives are profound and cannot be overstated,” the researchers say. Equally troubling, they note, is the stronger effect of COVID-19 on mental health, musculoskeletal, and episodic disorders in older adults, “highlighting their vulnerability” to these disorders following COVID-19 infection.
“It is imperative,” the researchers conclude, “that we recognize the enormous challenges posed by long COVID and all its downstream long-term consequences” and design capacity planning and clinical care pathways to address the needs of people who make it past the acute phase of COVID-19
We now know that the effects of COVID-19 don’t always end when the infection seems over. Long COVID—the postacute sequelae—can encompass a wide range of extrapulmonary organ dysfunctions. Most studies on COVID-19 have had follow-ups of 6 months or less with a narrow selection of neurologic outcomes, say Evan Xu, Yan Xie, PhD, and Ziyad Al-Aly, MD of the US Department of Veterans Affairs (VA) St. Louis Health Care System in Missouri. The 12-month study of 11,652,484 people published in Nature Medicine sounds an alert: Get ready to care for more patients with long-term, even chronic, neurologic disorders from migraine to stroke.
The researchers “leveraged the breadth and depth” of the VA’s national health care databases to build 3 groups: 154,068 people who survived the first 30 days of COVID-19; 5,638,795 VA users with no evidence of COVID-19 infection; and 5,859,621 VA users during 2017 (ie, prepandemic). Altogether, the groups corresponded to 14,064,985 person-years of follow-up.
The findings, which the researchers termed robust, revealed substantial risks and burdens beyond the first 30 days of COVID-19 infection, including “an array of neurologic disorders spanning several disease categories.”
Patients were at greater risk for stroke (both ischemic and hemorrhagic), cognition and memory disorders, peripheral nervous system disorders, episodic disorders like migraine and seizures, extrapyramidal and movement disorders, mental health disorders, musculoskeletal disorders, sensory disorders, Guillain-Barré syndrome, and encephalitis or encephalopathy.
The researchers estimated the hazard ratio of any neurological sequelae as 1.42. The risks were elevated even in people who did not require hospitalization during acute COVID-19 and increased according to the care setting of the acute phase of the disease from nonhospitalized to hospitalized and admitted to intensive care.
“Given the colossal scale of the pandemic,” the researchers say, governments and health systems should consider these findings when devising policy for continued management and developing plans for a postpandemic world. Some of the disorders they report on, they note, “are serious chronic conditions that will impact some people for a lifetime.” They point to 2 key findings: first, regardless of age, people with COVID-19 had a higher risk of all the neurologic outcomes examined, and second, the analyses suggest that the effects on risk were stronger in younger adults.
“The effects of these disorders on younger lives are profound and cannot be overstated,” the researchers say. Equally troubling, they note, is the stronger effect of COVID-19 on mental health, musculoskeletal, and episodic disorders in older adults, “highlighting their vulnerability” to these disorders following COVID-19 infection.
“It is imperative,” the researchers conclude, “that we recognize the enormous challenges posed by long COVID and all its downstream long-term consequences” and design capacity planning and clinical care pathways to address the needs of people who make it past the acute phase of COVID-19
We now know that the effects of COVID-19 don’t always end when the infection seems over. Long COVID—the postacute sequelae—can encompass a wide range of extrapulmonary organ dysfunctions. Most studies on COVID-19 have had follow-ups of 6 months or less with a narrow selection of neurologic outcomes, say Evan Xu, Yan Xie, PhD, and Ziyad Al-Aly, MD of the US Department of Veterans Affairs (VA) St. Louis Health Care System in Missouri. The 12-month study of 11,652,484 people published in Nature Medicine sounds an alert: Get ready to care for more patients with long-term, even chronic, neurologic disorders from migraine to stroke.
The researchers “leveraged the breadth and depth” of the VA’s national health care databases to build 3 groups: 154,068 people who survived the first 30 days of COVID-19; 5,638,795 VA users with no evidence of COVID-19 infection; and 5,859,621 VA users during 2017 (ie, prepandemic). Altogether, the groups corresponded to 14,064,985 person-years of follow-up.
The findings, which the researchers termed robust, revealed substantial risks and burdens beyond the first 30 days of COVID-19 infection, including “an array of neurologic disorders spanning several disease categories.”
Patients were at greater risk for stroke (both ischemic and hemorrhagic), cognition and memory disorders, peripheral nervous system disorders, episodic disorders like migraine and seizures, extrapyramidal and movement disorders, mental health disorders, musculoskeletal disorders, sensory disorders, Guillain-Barré syndrome, and encephalitis or encephalopathy.
The researchers estimated the hazard ratio of any neurological sequelae as 1.42. The risks were elevated even in people who did not require hospitalization during acute COVID-19 and increased according to the care setting of the acute phase of the disease from nonhospitalized to hospitalized and admitted to intensive care.
“Given the colossal scale of the pandemic,” the researchers say, governments and health systems should consider these findings when devising policy for continued management and developing plans for a postpandemic world. Some of the disorders they report on, they note, “are serious chronic conditions that will impact some people for a lifetime.” They point to 2 key findings: first, regardless of age, people with COVID-19 had a higher risk of all the neurologic outcomes examined, and second, the analyses suggest that the effects on risk were stronger in younger adults.
“The effects of these disorders on younger lives are profound and cannot be overstated,” the researchers say. Equally troubling, they note, is the stronger effect of COVID-19 on mental health, musculoskeletal, and episodic disorders in older adults, “highlighting their vulnerability” to these disorders following COVID-19 infection.
“It is imperative,” the researchers conclude, “that we recognize the enormous challenges posed by long COVID and all its downstream long-term consequences” and design capacity planning and clinical care pathways to address the needs of people who make it past the acute phase of COVID-19
Using SNRIs to prevent migraines in patients with depression
Ms. D, age 45, has major depressive disorder (MDD), generalized anxiety disorder (GAD), migraines, and hypertension. At a follow-up visit, she says she has been under a lot of stress at work in the past several months and feels her antidepressant is not working well for her depression or anxiety. Ms. D notes that lately she has had more frequent migraines, occurring approximately 4 times per month during the past 3 months. She describes a severe throbbing frontal pain that occurs primarily on the left side of her head, but sometimes on the right side. Ms. D says she experiences nausea, vomiting, and photophobia during these migraine episodes. The migraines last up to 12 hours, but often resolve with sumatriptan 50 mg as needed.
Ms. D takes fluoxetine 60 mg/d for depression and anxiety, lisinopril 20 mg/d for hypertension, as well as a women’s multivitamin and vitamin D3 daily. She has not tried other antidepressants and misses doses of her medications about once every other week. Her blood pressure is 125/80 mm Hg; heart rate is 80 beats per minute; and temperature is 37° C. Ms. D’s treatment team is considering switching her to a medication that can act as preventative therapy for migraines while also treating her depression and anxiety.
Migraine is a chronic, disabling neurovascular disorder that affects approximately 15% of the United States population.1 It is the second-leading disabling condition worldwide and may negatively affect social, family, personal, academic, and occupational domains.2 Migraine is often characterized by throbbing pain, is frequently unilateral, and may last 24 to 72 hours.3 It may occur with or without aura and can be associated with nausea, vomiting, or sensitivity to light.3 Episodic migraines occur <15 days a month, while chronic migraines occur ≥15 days a month.4
Many psychiatric, neurologic, vascular, and cardiac comorbidities are more prevalent in individuals who experience migraine headaches compared to the general population. Common psychiatric comorbidities found in patients with migraines are depression, bipolar disorder, GAD, panic disorder, and posttraumatic stress disorder5; MDD is the most common.4 A person who experiences migraine headaches is 2 to 4 times more likely to develop MDD than one who does not experience migraine headaches.4
First-line treatments for preventing migraine including divalproex, topiramate, metoprolol, propranolol, and timolol.6 However, for some patients with migraines and comorbid depression or anxiety, an antidepressant may be an option. This article briefly reviews the evidence for using antidepressants that have been studied for their ability to decrease migraine frequency.
Antidepressants that can prevent migraine
Tricyclic antidepressants (TCAs) are second- or third-line options for migraine prevention.6 While TCAs have proven to be effective for preventing migraines, many patients are unable to tolerate their adverse effects (ie, anticholinergic effects, sedation).7 TCAs may be more appealing for younger patients, who may be less bothered by anticholinergic burden, or those who have difficulty sleeping.
Serotonin-norepinephrine reuptake inhibitors (SNRIs). There has been growing interest in understanding the potential utility of SNRIs as a preventative treatment for migraines. Research has found that SNRIs are as effective as TCAs for preventing migraines and also more tolerable in terms of adverse effects.7 SNRIs such as venlafaxine and duloxetine are currently prescribed off-label to prevent migraines despite a lack of FDA approval for this indication.8
Continue to: Understanding the safety and efficacy...
Understanding the safety and efficacy of SNRIs as preventative treatment for episodic migraines is useful, particularly for patients with comorbid depression. The Table8-17 details clinical information related to SNRI use.
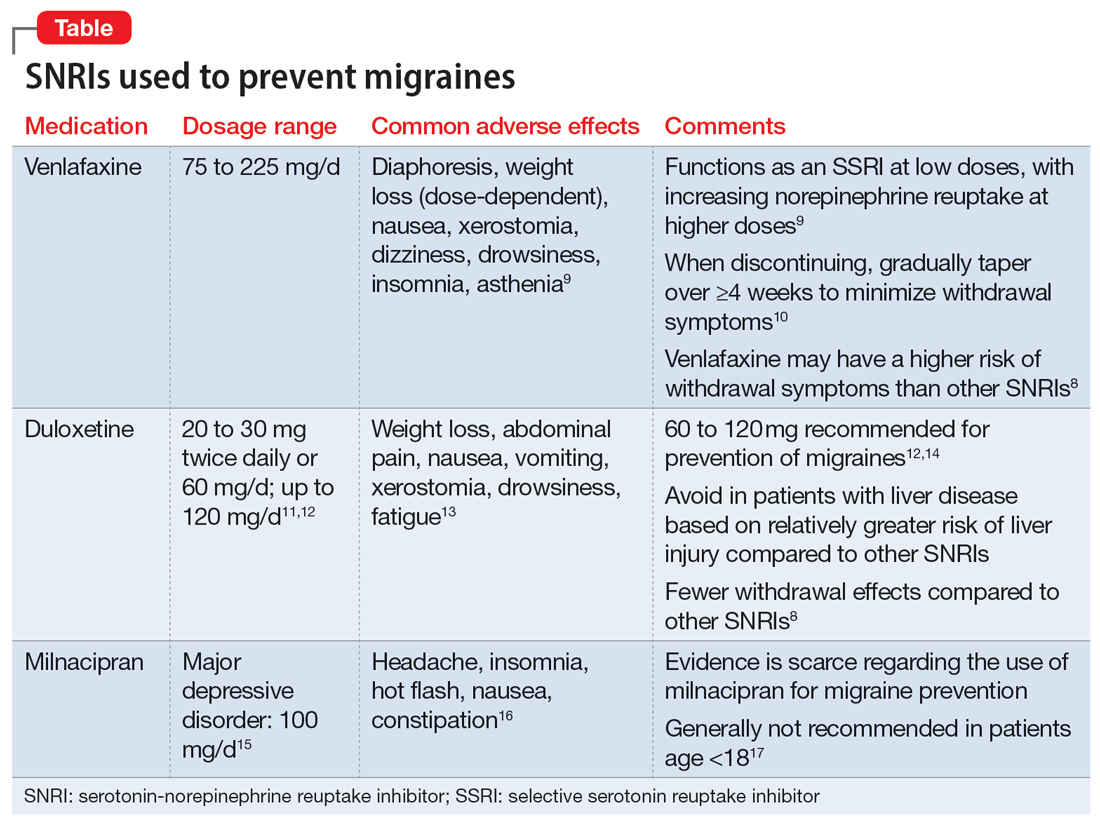
Duloxetine has demonstrated efficacy in preventing migraines in patients with comorbid depression.8 In a 2019 study, Kisler et al14 found that duloxetine 60 mg/d for 7 weeks was more effective for migraine prophylaxis than placebo as measured by the percentage of self-estimated migraine improvement by each patient compared to pretreatment levels (duloxetine: 52.3% ± 30.4%; placebo: 26.0% ± 27.3%; P = .001).
Venlafaxine has also demonstrated efficacy for preventing migraines in patients with comorbid depression.8 One study demonstrated a significant decrease in headaches per month with the use of venlafaxine 150 mg/d compared to placebo.18 Adelman et al19 found a reduction in migraine headaches per month (16.1 to 11.1, P < .0001) in patients who took venlafaxine for an average of 6 months with a mean dose of 150 mg/d. In a study of patients who did not have a mood disorder, Tarlaci20 found that venlafaxine reduced migraine headache independent of its antidepressant action.
Though milnacipran has not been studied as extensively as other SNRIs, evidence suggests it reduces the incidence of headaches and migraines, especially among episodic migraine patients. Although it has an equipotent effect on both serotonin and norepinephrine (NE) reuptake, milnacipran has a greater NE effect compared to other SNRIs approved for treating mood disorders. A prospective, single-arm study by Engel et al21 found a significant (P < .005) reduction from baseline in all headache and migraine days per month with the use of milnacipran 100 mg/d over the course of 3 months. The number of headache days per month was reduced by 4.2 compared to baseline. This same study reported improved functionality and reduced use of acute and symptomatic medications overall due to the decrease in headaches and migraines.21
In addition to demonstrating that certain SNRIs can effectively prevent migraine, some evidence suggests certain patients may benefit from the opportunity to decrease pill burden by using a single medication to treat both depression and migraine.22 Duloxetine may be preferred for patients who struggle with adherence (such as Ms. D) due to its relatively lower incidence of withdrawal symptoms compared to venlafaxine.8
CASE CONTINUED
Ms. D’s psychiatrist concludes she would be an appropriate candidate for treatment with an SNRI due to her history of MDD and chronic migraines. Because Ms. D expresses some difficulty remembering to take her medications, the psychiatrist recommends duloxetine because it is less likely to produce withdrawal symptoms compared to venlafaxine. To decrease pill burden, fluoxetine 60 mg is stopped with no taper due to its long half-life, and duloxetine is started at 30 mg/d, with a planned increase to 60 mg/d after 1 to 2 weeks as tolerated to target both mood and migraine prophylaxis. Duloxetine will not interact with Ms. D’s current medication regimen, including lisinopril, women’s multivitamin, or vitamin D3. The psychiatrist discusses the importance of medication adherence to improve her conditions effectively and safely. Ms. D’s heart rate and blood pressure will continue to be monitored.
Related Resources
- Leo RJ, Khalid K. Antidepressants for chronic pain. Current Psychiatry. 2019;18(2):8-16,21-22.
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
Drug Brand Names
Divalproex • Depakote
Duloxetine • Cymbalta
Fluoxetine • Prozac
Lisinopril • Zestril, Prinivil
Milnacipran • Savella
Sumatriptan • Imitrex
Topiramate • Topamax
Venlafaxine • Effexor
1. Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58(4):496-505. doi:10.1111/head.13281
2. GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi:10.1016/S1474-4422(18)30322-3
3. Goadsby PJ, Lipton RB, Ferrari MD. Migraine--current understanding and treatment. N Engl J Med. 2002;346(4):257-270. doi:10.1056/NEJMra010917
4. Amoozegar F. Depression comorbidity in migraine. Int Rev Psychiatry. 2017;29(5):504-515. doi:10.1080/09540261.2017.1326882
5. Burch RC, Buse DC, Lipton RB. Migraine: epidemiology, burden, and comorbidity. Neurol Clin. 2019;37(4):631-649. doi:10.1016/j.ncl.2019.06.001
6. Ha H, Gonzalez A. Migraine headache prophylaxis. Am Fam Physician. 2019;99(1):17-24.
7. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine (Baltimore). 2017;96(22):e6989. doi:10.1097/MD.0000000000006989
8. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
9. Venlafaxine. Lexicomp. 2021. http://online.lexi.com/
10. Ogle NR, Akkerman SR. Guidance for the discontinuation or switching of antidepressant therapies in adults. J Pharm Pract. 2013;26(4):389-396. doi:10.1177/0897190012467210
11. Duloxetine [package insert]. Indianapolis, IN: Eli Lilly and Company; 2004.
12. Young WB, Bradley KC, Anjum MW, et al. Duloxetine prophylaxis for episodic migraine in persons without depression: a prospective study. Headache. 2013;53(9):1430-1437.
13. Duloxetine. Lexicomp. 2021. http://online.lexi.com/
14. Kisler LB, Weissman-Fogel I, Coghill RC, et al. Individualization of migraine prevention: a randomized controlled trial of psychophysical-based prediction of duloxetine efficacy. Clin J Pain. 2019;35(9):753-765.
15. Mansuy L. Antidepressant therapy with milnacipran and venlafaxine. Neuropsychiatr Dis Treat. 2010;6 (Suppl I):17-22.
16. Milnacipran. Lexicomp. 2021. http://online.lexi.com/
17. Milnacipran. MedlinePlus. Updated January 22, 2022. Accessed August 19, 2022. https://medlineplus.gov/druginfo/meds/a609016.html
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144-152. doi:10.1111/j.1526-4610.2005.05029.x
19. Adelman LC, Adelman JU, Von Seggern R, et al. Venlafaxine extended release (XR) for the prophylaxis of migraine and tension-type headache: a retrospective study in a clinical setting. Headache. 2000;40(7):572-580. doi:10.1046/j.1526-4610.2000.00089.x
20. Tarlaci S. Escitalopram and venlafaxine for the prophylaxis of migraine headache without mood disorders. Clin Neuropharmacol. 2009;32(5):254-258. doi:10.1097/WNF.0b013e3181a8c84f
21. Engel ER, Kudrow D, Rapoport AM. A prospective, open-label study of milnacipran in the prevention of headache in patients with episodic or chronic migraine. Neurol Sci. 2014;35(3):429-435. doi:10.1007/s10072-013-1536-0
22. Baumgartner A, Drame K, Geutjens S, et al. Does the polypill improve patient adherence compared to its individual formulations? A systematic review. Pharmaceutics. 2020;12(2):190.
Ms. D, age 45, has major depressive disorder (MDD), generalized anxiety disorder (GAD), migraines, and hypertension. At a follow-up visit, she says she has been under a lot of stress at work in the past several months and feels her antidepressant is not working well for her depression or anxiety. Ms. D notes that lately she has had more frequent migraines, occurring approximately 4 times per month during the past 3 months. She describes a severe throbbing frontal pain that occurs primarily on the left side of her head, but sometimes on the right side. Ms. D says she experiences nausea, vomiting, and photophobia during these migraine episodes. The migraines last up to 12 hours, but often resolve with sumatriptan 50 mg as needed.
Ms. D takes fluoxetine 60 mg/d for depression and anxiety, lisinopril 20 mg/d for hypertension, as well as a women’s multivitamin and vitamin D3 daily. She has not tried other antidepressants and misses doses of her medications about once every other week. Her blood pressure is 125/80 mm Hg; heart rate is 80 beats per minute; and temperature is 37° C. Ms. D’s treatment team is considering switching her to a medication that can act as preventative therapy for migraines while also treating her depression and anxiety.
Migraine is a chronic, disabling neurovascular disorder that affects approximately 15% of the United States population.1 It is the second-leading disabling condition worldwide and may negatively affect social, family, personal, academic, and occupational domains.2 Migraine is often characterized by throbbing pain, is frequently unilateral, and may last 24 to 72 hours.3 It may occur with or without aura and can be associated with nausea, vomiting, or sensitivity to light.3 Episodic migraines occur <15 days a month, while chronic migraines occur ≥15 days a month.4
Many psychiatric, neurologic, vascular, and cardiac comorbidities are more prevalent in individuals who experience migraine headaches compared to the general population. Common psychiatric comorbidities found in patients with migraines are depression, bipolar disorder, GAD, panic disorder, and posttraumatic stress disorder5; MDD is the most common.4 A person who experiences migraine headaches is 2 to 4 times more likely to develop MDD than one who does not experience migraine headaches.4
First-line treatments for preventing migraine including divalproex, topiramate, metoprolol, propranolol, and timolol.6 However, for some patients with migraines and comorbid depression or anxiety, an antidepressant may be an option. This article briefly reviews the evidence for using antidepressants that have been studied for their ability to decrease migraine frequency.
Antidepressants that can prevent migraine
Tricyclic antidepressants (TCAs) are second- or third-line options for migraine prevention.6 While TCAs have proven to be effective for preventing migraines, many patients are unable to tolerate their adverse effects (ie, anticholinergic effects, sedation).7 TCAs may be more appealing for younger patients, who may be less bothered by anticholinergic burden, or those who have difficulty sleeping.
Serotonin-norepinephrine reuptake inhibitors (SNRIs). There has been growing interest in understanding the potential utility of SNRIs as a preventative treatment for migraines. Research has found that SNRIs are as effective as TCAs for preventing migraines and also more tolerable in terms of adverse effects.7 SNRIs such as venlafaxine and duloxetine are currently prescribed off-label to prevent migraines despite a lack of FDA approval for this indication.8
Continue to: Understanding the safety and efficacy...
Understanding the safety and efficacy of SNRIs as preventative treatment for episodic migraines is useful, particularly for patients with comorbid depression. The Table8-17 details clinical information related to SNRI use.

Duloxetine has demonstrated efficacy in preventing migraines in patients with comorbid depression.8 In a 2019 study, Kisler et al14 found that duloxetine 60 mg/d for 7 weeks was more effective for migraine prophylaxis than placebo as measured by the percentage of self-estimated migraine improvement by each patient compared to pretreatment levels (duloxetine: 52.3% ± 30.4%; placebo: 26.0% ± 27.3%; P = .001).
Venlafaxine has also demonstrated efficacy for preventing migraines in patients with comorbid depression.8 One study demonstrated a significant decrease in headaches per month with the use of venlafaxine 150 mg/d compared to placebo.18 Adelman et al19 found a reduction in migraine headaches per month (16.1 to 11.1, P < .0001) in patients who took venlafaxine for an average of 6 months with a mean dose of 150 mg/d. In a study of patients who did not have a mood disorder, Tarlaci20 found that venlafaxine reduced migraine headache independent of its antidepressant action.
Though milnacipran has not been studied as extensively as other SNRIs, evidence suggests it reduces the incidence of headaches and migraines, especially among episodic migraine patients. Although it has an equipotent effect on both serotonin and norepinephrine (NE) reuptake, milnacipran has a greater NE effect compared to other SNRIs approved for treating mood disorders. A prospective, single-arm study by Engel et al21 found a significant (P < .005) reduction from baseline in all headache and migraine days per month with the use of milnacipran 100 mg/d over the course of 3 months. The number of headache days per month was reduced by 4.2 compared to baseline. This same study reported improved functionality and reduced use of acute and symptomatic medications overall due to the decrease in headaches and migraines.21
In addition to demonstrating that certain SNRIs can effectively prevent migraine, some evidence suggests certain patients may benefit from the opportunity to decrease pill burden by using a single medication to treat both depression and migraine.22 Duloxetine may be preferred for patients who struggle with adherence (such as Ms. D) due to its relatively lower incidence of withdrawal symptoms compared to venlafaxine.8
CASE CONTINUED
Ms. D’s psychiatrist concludes she would be an appropriate candidate for treatment with an SNRI due to her history of MDD and chronic migraines. Because Ms. D expresses some difficulty remembering to take her medications, the psychiatrist recommends duloxetine because it is less likely to produce withdrawal symptoms compared to venlafaxine. To decrease pill burden, fluoxetine 60 mg is stopped with no taper due to its long half-life, and duloxetine is started at 30 mg/d, with a planned increase to 60 mg/d after 1 to 2 weeks as tolerated to target both mood and migraine prophylaxis. Duloxetine will not interact with Ms. D’s current medication regimen, including lisinopril, women’s multivitamin, or vitamin D3. The psychiatrist discusses the importance of medication adherence to improve her conditions effectively and safely. Ms. D’s heart rate and blood pressure will continue to be monitored.
Related Resources
- Leo RJ, Khalid K. Antidepressants for chronic pain. Current Psychiatry. 2019;18(2):8-16,21-22.
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
Drug Brand Names
Divalproex • Depakote
Duloxetine • Cymbalta
Fluoxetine • Prozac
Lisinopril • Zestril, Prinivil
Milnacipran • Savella
Sumatriptan • Imitrex
Topiramate • Topamax
Venlafaxine • Effexor
Ms. D, age 45, has major depressive disorder (MDD), generalized anxiety disorder (GAD), migraines, and hypertension. At a follow-up visit, she says she has been under a lot of stress at work in the past several months and feels her antidepressant is not working well for her depression or anxiety. Ms. D notes that lately she has had more frequent migraines, occurring approximately 4 times per month during the past 3 months. She describes a severe throbbing frontal pain that occurs primarily on the left side of her head, but sometimes on the right side. Ms. D says she experiences nausea, vomiting, and photophobia during these migraine episodes. The migraines last up to 12 hours, but often resolve with sumatriptan 50 mg as needed.
Ms. D takes fluoxetine 60 mg/d for depression and anxiety, lisinopril 20 mg/d for hypertension, as well as a women’s multivitamin and vitamin D3 daily. She has not tried other antidepressants and misses doses of her medications about once every other week. Her blood pressure is 125/80 mm Hg; heart rate is 80 beats per minute; and temperature is 37° C. Ms. D’s treatment team is considering switching her to a medication that can act as preventative therapy for migraines while also treating her depression and anxiety.
Migraine is a chronic, disabling neurovascular disorder that affects approximately 15% of the United States population.1 It is the second-leading disabling condition worldwide and may negatively affect social, family, personal, academic, and occupational domains.2 Migraine is often characterized by throbbing pain, is frequently unilateral, and may last 24 to 72 hours.3 It may occur with or without aura and can be associated with nausea, vomiting, or sensitivity to light.3 Episodic migraines occur <15 days a month, while chronic migraines occur ≥15 days a month.4
Many psychiatric, neurologic, vascular, and cardiac comorbidities are more prevalent in individuals who experience migraine headaches compared to the general population. Common psychiatric comorbidities found in patients with migraines are depression, bipolar disorder, GAD, panic disorder, and posttraumatic stress disorder5; MDD is the most common.4 A person who experiences migraine headaches is 2 to 4 times more likely to develop MDD than one who does not experience migraine headaches.4
First-line treatments for preventing migraine including divalproex, topiramate, metoprolol, propranolol, and timolol.6 However, for some patients with migraines and comorbid depression or anxiety, an antidepressant may be an option. This article briefly reviews the evidence for using antidepressants that have been studied for their ability to decrease migraine frequency.
Antidepressants that can prevent migraine
Tricyclic antidepressants (TCAs) are second- or third-line options for migraine prevention.6 While TCAs have proven to be effective for preventing migraines, many patients are unable to tolerate their adverse effects (ie, anticholinergic effects, sedation).7 TCAs may be more appealing for younger patients, who may be less bothered by anticholinergic burden, or those who have difficulty sleeping.
Serotonin-norepinephrine reuptake inhibitors (SNRIs). There has been growing interest in understanding the potential utility of SNRIs as a preventative treatment for migraines. Research has found that SNRIs are as effective as TCAs for preventing migraines and also more tolerable in terms of adverse effects.7 SNRIs such as venlafaxine and duloxetine are currently prescribed off-label to prevent migraines despite a lack of FDA approval for this indication.8
Continue to: Understanding the safety and efficacy...
Understanding the safety and efficacy of SNRIs as preventative treatment for episodic migraines is useful, particularly for patients with comorbid depression. The Table8-17 details clinical information related to SNRI use.

Duloxetine has demonstrated efficacy in preventing migraines in patients with comorbid depression.8 In a 2019 study, Kisler et al14 found that duloxetine 60 mg/d for 7 weeks was more effective for migraine prophylaxis than placebo as measured by the percentage of self-estimated migraine improvement by each patient compared to pretreatment levels (duloxetine: 52.3% ± 30.4%; placebo: 26.0% ± 27.3%; P = .001).
Venlafaxine has also demonstrated efficacy for preventing migraines in patients with comorbid depression.8 One study demonstrated a significant decrease in headaches per month with the use of venlafaxine 150 mg/d compared to placebo.18 Adelman et al19 found a reduction in migraine headaches per month (16.1 to 11.1, P < .0001) in patients who took venlafaxine for an average of 6 months with a mean dose of 150 mg/d. In a study of patients who did not have a mood disorder, Tarlaci20 found that venlafaxine reduced migraine headache independent of its antidepressant action.
Though milnacipran has not been studied as extensively as other SNRIs, evidence suggests it reduces the incidence of headaches and migraines, especially among episodic migraine patients. Although it has an equipotent effect on both serotonin and norepinephrine (NE) reuptake, milnacipran has a greater NE effect compared to other SNRIs approved for treating mood disorders. A prospective, single-arm study by Engel et al21 found a significant (P < .005) reduction from baseline in all headache and migraine days per month with the use of milnacipran 100 mg/d over the course of 3 months. The number of headache days per month was reduced by 4.2 compared to baseline. This same study reported improved functionality and reduced use of acute and symptomatic medications overall due to the decrease in headaches and migraines.21
In addition to demonstrating that certain SNRIs can effectively prevent migraine, some evidence suggests certain patients may benefit from the opportunity to decrease pill burden by using a single medication to treat both depression and migraine.22 Duloxetine may be preferred for patients who struggle with adherence (such as Ms. D) due to its relatively lower incidence of withdrawal symptoms compared to venlafaxine.8
CASE CONTINUED
Ms. D’s psychiatrist concludes she would be an appropriate candidate for treatment with an SNRI due to her history of MDD and chronic migraines. Because Ms. D expresses some difficulty remembering to take her medications, the psychiatrist recommends duloxetine because it is less likely to produce withdrawal symptoms compared to venlafaxine. To decrease pill burden, fluoxetine 60 mg is stopped with no taper due to its long half-life, and duloxetine is started at 30 mg/d, with a planned increase to 60 mg/d after 1 to 2 weeks as tolerated to target both mood and migraine prophylaxis. Duloxetine will not interact with Ms. D’s current medication regimen, including lisinopril, women’s multivitamin, or vitamin D3. The psychiatrist discusses the importance of medication adherence to improve her conditions effectively and safely. Ms. D’s heart rate and blood pressure will continue to be monitored.
Related Resources
- Leo RJ, Khalid K. Antidepressants for chronic pain. Current Psychiatry. 2019;18(2):8-16,21-22.
- Williams AM, Knox ED. When to prescribe antidepressants to treat comorbid depression and pain disorders. Current Psychiatry. 2017;16(1):55-58.
Drug Brand Names
Divalproex • Depakote
Duloxetine • Cymbalta
Fluoxetine • Prozac
Lisinopril • Zestril, Prinivil
Milnacipran • Savella
Sumatriptan • Imitrex
Topiramate • Topamax
Venlafaxine • Effexor
1. Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58(4):496-505. doi:10.1111/head.13281
2. GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi:10.1016/S1474-4422(18)30322-3
3. Goadsby PJ, Lipton RB, Ferrari MD. Migraine--current understanding and treatment. N Engl J Med. 2002;346(4):257-270. doi:10.1056/NEJMra010917
4. Amoozegar F. Depression comorbidity in migraine. Int Rev Psychiatry. 2017;29(5):504-515. doi:10.1080/09540261.2017.1326882
5. Burch RC, Buse DC, Lipton RB. Migraine: epidemiology, burden, and comorbidity. Neurol Clin. 2019;37(4):631-649. doi:10.1016/j.ncl.2019.06.001
6. Ha H, Gonzalez A. Migraine headache prophylaxis. Am Fam Physician. 2019;99(1):17-24.
7. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine (Baltimore). 2017;96(22):e6989. doi:10.1097/MD.0000000000006989
8. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
9. Venlafaxine. Lexicomp. 2021. http://online.lexi.com/
10. Ogle NR, Akkerman SR. Guidance for the discontinuation or switching of antidepressant therapies in adults. J Pharm Pract. 2013;26(4):389-396. doi:10.1177/0897190012467210
11. Duloxetine [package insert]. Indianapolis, IN: Eli Lilly and Company; 2004.
12. Young WB, Bradley KC, Anjum MW, et al. Duloxetine prophylaxis for episodic migraine in persons without depression: a prospective study. Headache. 2013;53(9):1430-1437.
13. Duloxetine. Lexicomp. 2021. http://online.lexi.com/
14. Kisler LB, Weissman-Fogel I, Coghill RC, et al. Individualization of migraine prevention: a randomized controlled trial of psychophysical-based prediction of duloxetine efficacy. Clin J Pain. 2019;35(9):753-765.
15. Mansuy L. Antidepressant therapy with milnacipran and venlafaxine. Neuropsychiatr Dis Treat. 2010;6 (Suppl I):17-22.
16. Milnacipran. Lexicomp. 2021. http://online.lexi.com/
17. Milnacipran. MedlinePlus. Updated January 22, 2022. Accessed August 19, 2022. https://medlineplus.gov/druginfo/meds/a609016.html
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144-152. doi:10.1111/j.1526-4610.2005.05029.x
19. Adelman LC, Adelman JU, Von Seggern R, et al. Venlafaxine extended release (XR) for the prophylaxis of migraine and tension-type headache: a retrospective study in a clinical setting. Headache. 2000;40(7):572-580. doi:10.1046/j.1526-4610.2000.00089.x
20. Tarlaci S. Escitalopram and venlafaxine for the prophylaxis of migraine headache without mood disorders. Clin Neuropharmacol. 2009;32(5):254-258. doi:10.1097/WNF.0b013e3181a8c84f
21. Engel ER, Kudrow D, Rapoport AM. A prospective, open-label study of milnacipran in the prevention of headache in patients with episodic or chronic migraine. Neurol Sci. 2014;35(3):429-435. doi:10.1007/s10072-013-1536-0
22. Baumgartner A, Drame K, Geutjens S, et al. Does the polypill improve patient adherence compared to its individual formulations? A systematic review. Pharmaceutics. 2020;12(2):190.
1. Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58(4):496-505. doi:10.1111/head.13281
2. GBD 2016 Headache Collaborators. Global, regional, and national burden of migraine and tension-type headache, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018;17(11):954-976. doi:10.1016/S1474-4422(18)30322-3
3. Goadsby PJ, Lipton RB, Ferrari MD. Migraine--current understanding and treatment. N Engl J Med. 2002;346(4):257-270. doi:10.1056/NEJMra010917
4. Amoozegar F. Depression comorbidity in migraine. Int Rev Psychiatry. 2017;29(5):504-515. doi:10.1080/09540261.2017.1326882
5. Burch RC, Buse DC, Lipton RB. Migraine: epidemiology, burden, and comorbidity. Neurol Clin. 2019;37(4):631-649. doi:10.1016/j.ncl.2019.06.001
6. Ha H, Gonzalez A. Migraine headache prophylaxis. Am Fam Physician. 2019;99(1):17-24.
7. Xu XM, Liu Y, Dong MX, et al. Tricyclic antidepressants for preventing migraine in adults. Medicine (Baltimore). 2017;96(22):e6989. doi:10.1097/MD.0000000000006989
8. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
9. Venlafaxine. Lexicomp. 2021. http://online.lexi.com/
10. Ogle NR, Akkerman SR. Guidance for the discontinuation or switching of antidepressant therapies in adults. J Pharm Pract. 2013;26(4):389-396. doi:10.1177/0897190012467210
11. Duloxetine [package insert]. Indianapolis, IN: Eli Lilly and Company; 2004.
12. Young WB, Bradley KC, Anjum MW, et al. Duloxetine prophylaxis for episodic migraine in persons without depression: a prospective study. Headache. 2013;53(9):1430-1437.
13. Duloxetine. Lexicomp. 2021. http://online.lexi.com/
14. Kisler LB, Weissman-Fogel I, Coghill RC, et al. Individualization of migraine prevention: a randomized controlled trial of psychophysical-based prediction of duloxetine efficacy. Clin J Pain. 2019;35(9):753-765.
15. Mansuy L. Antidepressant therapy with milnacipran and venlafaxine. Neuropsychiatr Dis Treat. 2010;6 (Suppl I):17-22.
16. Milnacipran. Lexicomp. 2021. http://online.lexi.com/
17. Milnacipran. MedlinePlus. Updated January 22, 2022. Accessed August 19, 2022. https://medlineplus.gov/druginfo/meds/a609016.html
18. Ozyalcin SN, Talu GK, Kiziltan E, et al. The efficacy and safety of venlafaxine in the prophylaxis of migraine. Headache. 2005;45(2):144-152. doi:10.1111/j.1526-4610.2005.05029.x
19. Adelman LC, Adelman JU, Von Seggern R, et al. Venlafaxine extended release (XR) for the prophylaxis of migraine and tension-type headache: a retrospective study in a clinical setting. Headache. 2000;40(7):572-580. doi:10.1046/j.1526-4610.2000.00089.x
20. Tarlaci S. Escitalopram and venlafaxine for the prophylaxis of migraine headache without mood disorders. Clin Neuropharmacol. 2009;32(5):254-258. doi:10.1097/WNF.0b013e3181a8c84f
21. Engel ER, Kudrow D, Rapoport AM. A prospective, open-label study of milnacipran in the prevention of headache in patients with episodic or chronic migraine. Neurol Sci. 2014;35(3):429-435. doi:10.1007/s10072-013-1536-0
22. Baumgartner A, Drame K, Geutjens S, et al. Does the polypill improve patient adherence compared to its individual formulations? A systematic review. Pharmaceutics. 2020;12(2):190.
Racial disparities in preventive services use seen among patients with spina bifida or cerebral palsy
Black adults also had lower odds of having a bone density screening, compared with White adults. Plus, comorbidities were highest among the Black patients, according to the paper, which was published in Annals of Family Medicine.
Elham Mahmoudi, PhD, and her coauthors examined private insurance claims from 11,635 patients with cerebral palsy (CP) or spina bifida over ten years from 2007 to 2017. The researchers analyzed comorbidities and compared the rates of different psychological, cardiometabolic, and musculoskeletal conditions among these patients.
Only 23% of Hispanic participants and 18% of Black participants attended an annual wellness visit, compared with 32% of the White participants.
Only 1% of Black and 2% of White participants received any bone density screening (odds ratio = 0.54, 95% confidence interval [CI], 0.31-0.95), a service that is essential for catching a patient’s potential risk for osteoporosis and fractures.
According to the researchers, patients accessed services such as bone density scans, cholesterol assessments, diabetes screenings, and annual wellness visits less than recommended for people with those chronic conditions.
“People with spina bifida and cerebral palsy have complex care needs. We know through our work that chronic conditions are much higher among them compared with adults without disabilities,” Dr. Mahmoudi, associate professor in the department of family medicine at University of Michigan, Ann Arbor, said in an interview. “I was surprised to see even with private insurance, the rate of using preventative services is so low among White people and minority populations.”
Comorbidities highest in Black participants
Black adults had the highest comorbidity score of 2.5, and Hispanic adults had the lowest comorbidity score of 1.8. For White adults in the study, the comorbidity score was 2.0.
Osteoporosis, a common concern for people with spina bifida or cerebral palsy, was detected in around 4% of all participants. Osteoarthritis was detected in 13.38% of Black participants, versus 8.53% of Hispanic participants and 11.09% of White participants.
Diabetes and hypertension were more common among Black participants than among Hispanic and White participants. The percentages of Black patients with hypertension and diabetes were 16.5% and 39.89%, respectively. Among the Hispanic and White adults, the percentages with hypertension were 22.3% and 28.2%, respectively, according to the paper.
Disparities in access
Jamil Paden, racial and health equity manager at the Christopher and Dana Reeve Foundation, said getting access to literature, transportation, tables, chairs, weigh scales, and imaging equipment that accommodate the needs of people with disabilities are some of the biggest challenges for people with disabilities who are trying to receive care.
“It’s not a one size fits all, we have to recognize that if someone doesn’t see themselves in a particular place, then it makes it more challenging for them to feel comfortable speaking up and saying things about their health, which would prevent a person from saying something early on,” Mr. Paden said in an interview. “That particular issue will continue to grow and become more of a health risk, or health challenge down the line.”
Mr. Paden emphasized intersections between class, race, and circumstances which can, together, make health care less equitable for people with disabilities, especially in underserved communities and communities of color. He urged health care providers to distance their practices from a “one size fits all” approach to treatment and engage in their patients’ individual lives and communities.
“It’s not enough to just say, Hey, you have a disability. So let me treat your disability ... You have to recognize that although a patient may have a dire diagnosis, they also are a person of color, and they have to navigate different aspects of life from their counterparts,” he said.
Dr. Mahmoudi said patient and provider understanding of the disability is often lacking. She recommended advocating for patients, noting that giving both patients and providers the tools to further educate themselves and apply that to their regular visits is a good first step.
“Just having access to a facility doesn’t mean they will get the services they need. Preventative services that are recommended for people with disabilities differ from the general population. Providers should be educated about that and the patient needs to be educated about that,” she added.
“Patients who do not approach clinicians get lost in the system. Maybe many facilities are not disability friendly, or they need health literacy. If they don’t know they are at risk for osteoporosis, for example, then they won’t ask,” Dr. Mahmoudi said.
The study was funded by The National Institute on Disability, Independent Living, and Rehabilitation Research. Dr. Mahmoudi and Mr. Paden report no relevant financial relationships.
Black adults also had lower odds of having a bone density screening, compared with White adults. Plus, comorbidities were highest among the Black patients, according to the paper, which was published in Annals of Family Medicine.
Elham Mahmoudi, PhD, and her coauthors examined private insurance claims from 11,635 patients with cerebral palsy (CP) or spina bifida over ten years from 2007 to 2017. The researchers analyzed comorbidities and compared the rates of different psychological, cardiometabolic, and musculoskeletal conditions among these patients.
Only 23% of Hispanic participants and 18% of Black participants attended an annual wellness visit, compared with 32% of the White participants.
Only 1% of Black and 2% of White participants received any bone density screening (odds ratio = 0.54, 95% confidence interval [CI], 0.31-0.95), a service that is essential for catching a patient’s potential risk for osteoporosis and fractures.
According to the researchers, patients accessed services such as bone density scans, cholesterol assessments, diabetes screenings, and annual wellness visits less than recommended for people with those chronic conditions.
“People with spina bifida and cerebral palsy have complex care needs. We know through our work that chronic conditions are much higher among them compared with adults without disabilities,” Dr. Mahmoudi, associate professor in the department of family medicine at University of Michigan, Ann Arbor, said in an interview. “I was surprised to see even with private insurance, the rate of using preventative services is so low among White people and minority populations.”
Comorbidities highest in Black participants
Black adults had the highest comorbidity score of 2.5, and Hispanic adults had the lowest comorbidity score of 1.8. For White adults in the study, the comorbidity score was 2.0.
Osteoporosis, a common concern for people with spina bifida or cerebral palsy, was detected in around 4% of all participants. Osteoarthritis was detected in 13.38% of Black participants, versus 8.53% of Hispanic participants and 11.09% of White participants.
Diabetes and hypertension were more common among Black participants than among Hispanic and White participants. The percentages of Black patients with hypertension and diabetes were 16.5% and 39.89%, respectively. Among the Hispanic and White adults, the percentages with hypertension were 22.3% and 28.2%, respectively, according to the paper.
Disparities in access
Jamil Paden, racial and health equity manager at the Christopher and Dana Reeve Foundation, said getting access to literature, transportation, tables, chairs, weigh scales, and imaging equipment that accommodate the needs of people with disabilities are some of the biggest challenges for people with disabilities who are trying to receive care.
“It’s not a one size fits all, we have to recognize that if someone doesn’t see themselves in a particular place, then it makes it more challenging for them to feel comfortable speaking up and saying things about their health, which would prevent a person from saying something early on,” Mr. Paden said in an interview. “That particular issue will continue to grow and become more of a health risk, or health challenge down the line.”
Mr. Paden emphasized intersections between class, race, and circumstances which can, together, make health care less equitable for people with disabilities, especially in underserved communities and communities of color. He urged health care providers to distance their practices from a “one size fits all” approach to treatment and engage in their patients’ individual lives and communities.
“It’s not enough to just say, Hey, you have a disability. So let me treat your disability ... You have to recognize that although a patient may have a dire diagnosis, they also are a person of color, and they have to navigate different aspects of life from their counterparts,” he said.
Dr. Mahmoudi said patient and provider understanding of the disability is often lacking. She recommended advocating for patients, noting that giving both patients and providers the tools to further educate themselves and apply that to their regular visits is a good first step.
“Just having access to a facility doesn’t mean they will get the services they need. Preventative services that are recommended for people with disabilities differ from the general population. Providers should be educated about that and the patient needs to be educated about that,” she added.
“Patients who do not approach clinicians get lost in the system. Maybe many facilities are not disability friendly, or they need health literacy. If they don’t know they are at risk for osteoporosis, for example, then they won’t ask,” Dr. Mahmoudi said.
The study was funded by The National Institute on Disability, Independent Living, and Rehabilitation Research. Dr. Mahmoudi and Mr. Paden report no relevant financial relationships.
Black adults also had lower odds of having a bone density screening, compared with White adults. Plus, comorbidities were highest among the Black patients, according to the paper, which was published in Annals of Family Medicine.
Elham Mahmoudi, PhD, and her coauthors examined private insurance claims from 11,635 patients with cerebral palsy (CP) or spina bifida over ten years from 2007 to 2017. The researchers analyzed comorbidities and compared the rates of different psychological, cardiometabolic, and musculoskeletal conditions among these patients.
Only 23% of Hispanic participants and 18% of Black participants attended an annual wellness visit, compared with 32% of the White participants.
Only 1% of Black and 2% of White participants received any bone density screening (odds ratio = 0.54, 95% confidence interval [CI], 0.31-0.95), a service that is essential for catching a patient’s potential risk for osteoporosis and fractures.
According to the researchers, patients accessed services such as bone density scans, cholesterol assessments, diabetes screenings, and annual wellness visits less than recommended for people with those chronic conditions.
“People with spina bifida and cerebral palsy have complex care needs. We know through our work that chronic conditions are much higher among them compared with adults without disabilities,” Dr. Mahmoudi, associate professor in the department of family medicine at University of Michigan, Ann Arbor, said in an interview. “I was surprised to see even with private insurance, the rate of using preventative services is so low among White people and minority populations.”
Comorbidities highest in Black participants
Black adults had the highest comorbidity score of 2.5, and Hispanic adults had the lowest comorbidity score of 1.8. For White adults in the study, the comorbidity score was 2.0.
Osteoporosis, a common concern for people with spina bifida or cerebral palsy, was detected in around 4% of all participants. Osteoarthritis was detected in 13.38% of Black participants, versus 8.53% of Hispanic participants and 11.09% of White participants.
Diabetes and hypertension were more common among Black participants than among Hispanic and White participants. The percentages of Black patients with hypertension and diabetes were 16.5% and 39.89%, respectively. Among the Hispanic and White adults, the percentages with hypertension were 22.3% and 28.2%, respectively, according to the paper.
Disparities in access
Jamil Paden, racial and health equity manager at the Christopher and Dana Reeve Foundation, said getting access to literature, transportation, tables, chairs, weigh scales, and imaging equipment that accommodate the needs of people with disabilities are some of the biggest challenges for people with disabilities who are trying to receive care.
“It’s not a one size fits all, we have to recognize that if someone doesn’t see themselves in a particular place, then it makes it more challenging for them to feel comfortable speaking up and saying things about their health, which would prevent a person from saying something early on,” Mr. Paden said in an interview. “That particular issue will continue to grow and become more of a health risk, or health challenge down the line.”
Mr. Paden emphasized intersections between class, race, and circumstances which can, together, make health care less equitable for people with disabilities, especially in underserved communities and communities of color. He urged health care providers to distance their practices from a “one size fits all” approach to treatment and engage in their patients’ individual lives and communities.
“It’s not enough to just say, Hey, you have a disability. So let me treat your disability ... You have to recognize that although a patient may have a dire diagnosis, they also are a person of color, and they have to navigate different aspects of life from their counterparts,” he said.
Dr. Mahmoudi said patient and provider understanding of the disability is often lacking. She recommended advocating for patients, noting that giving both patients and providers the tools to further educate themselves and apply that to their regular visits is a good first step.
“Just having access to a facility doesn’t mean they will get the services they need. Preventative services that are recommended for people with disabilities differ from the general population. Providers should be educated about that and the patient needs to be educated about that,” she added.
“Patients who do not approach clinicians get lost in the system. Maybe many facilities are not disability friendly, or they need health literacy. If they don’t know they are at risk for osteoporosis, for example, then they won’t ask,” Dr. Mahmoudi said.
The study was funded by The National Institute on Disability, Independent Living, and Rehabilitation Research. Dr. Mahmoudi and Mr. Paden report no relevant financial relationships.
FROM ANNALS OF FAMILY MEDICINE
ALS drug gets FDA panel thumbs-up after rare second look
In a rare second review of a new drug application,
By a vote of 7-2, the FDA Peripheral and Central Nervous System Drugs Advisory Committee reversed course on AMX0035 (Amylyx Pharmaceuticals), a combination of sodium phenylbutyrate and taurursodiol.
The panel previously voted 6-4 to reject the drug, ruling that data provided by Amylyx had failed to demonstrate that the survival benefit reported in the only clinical trial of AMX0035 so far was a direct result of the drug.
This time, two panelists who previously voted no were swayed by the drug maker’s new analysis of previously presented research, more than 1,300 public comments in support of the drug, supportive testimony from ALS patients and clinicians, and assurances from company executives that Amylyx would pull the drug from the market if results of an ongoing phase 3 clinical trial show the drug doesn’t work.
“As in March, today we have to have an internal dialogue between our scientific scrutiny and clinical compassion,” said Liana G. Apostolova, MD, from Indiana University, Indianapolis, who originally voted against the application.
“Today I also saw additional confirmatory evidence that was not unequivocally persuasive but was nonetheless reassuring,” Dr. Apostolova said. “Because of that I am voting in support of AMX0035.”
A rare second chance
ALS (Lou Gehrig’s disease) is a progressive, fatal neurodegenerative disease affecting nerve cells in the brain and spinal cord that causes loss of motor control. It is rare, affecting about 30,000 people in the United States with another 5,000 new cases diagnosed each year. Most people with the disease die within 2 years of diagnosis.
The FDA has approved two therapies for ALS, but both have limited efficacy.
Typically, FDA approval requires two large studies or one study with a “very persuasive” effect on survival.
Amylyx’s application is based on a single study, the multicenter, two-phase CENTAUR trial. In that trial, 137 people with ALS received AMX0035 or placebo for 24 weeks.
Researchers found that patients receiving AMX0035 had a 25% slower decline in function, compared with the those taking placebo. A change of 20% or more is considered clinically meaningful.
The investigators also found a statistically significant median difference of 4.8 months in time to death, first hospitalization, or tracheostomy/permanent assisted ventilation in the group originally assigned to receive AMX0035 compared with the group originally assigned to receive placebo (hazard ratio, 0.62; P = .023).
In the panel’s previous vote against the drug application, members cited several issues with the study, concluding that it did not offer persuasive or robust evidence of efficacy. They also cited missing data assumptions in the primary analysis, issues of randomization and imbalances in concomitant use of riluzole and edaravone, the two FDA-approved drugs for ALS.
The FDA later requested additional information from Amylyx, delayed its final ruling on the new drug application to Sept. 29, and called for a second review meeting – a virtually unheard-of move.
An FDA review posted in advance of the meeting Sept. 29 had hinted at a different outcome. In that report, regulators said new data from Amylyx were not “sufficiently independent or persuasive” to establish effectiveness.
However, FDA officials in the meeting stressed the importance of considering unmet medical need in ALS in the panel’s decision-making process.
“Recognizing the substantial unmet medical need in ALS, we feel that it is important that the committee is afforded the opportunity to consider this new information, along with the information presented at the prior meeting, in that context,” Billy Dunn, MD, director of the FDA Office of Neuroscience, said during the meeting.
Panelists heard additional data that Amylyx claims confirms the results of the CENTAUR study, including new analyses of the previously submitted survival data and new data from that study and an open-label extension.
They also provided new information on a biomarker data from a phase 2 study of AMX0035 to treat Alzheimer’s disease.
“I think we note the limitations of the analyses, but we still haven’t taken it off the table that they could be considered as confirmatory evidence and that’s why we’re here today,” said Teresa Buracchio, MD, director of the division of neurology for the FDA.
Two members of the panel who voted no in March stuck with that position at the Sept. 29 meeting.
“Unfortunately, I don’t believe the new evidence we’ve reviewed, while promising, combined with that prior evidence, constitutes substantial evidence of effectiveness,” said panelist Caleb Alexander, MD, a professor of epidemiology and medicine at the Johns Hopkins University Center for Drug Safety and Effectiveness, Baltimore.
Dr. Alexander, who also voted no in March, said that post hoc data presented at the meeting were not enough to assuage concerns that led him and others to reject the drug in March.
A challenging situation
Amylyx is currently leading the 48-week international, phase 3, placebo-controlled PHOENIX clinical trial of AMX0035. The study has enrolled about half of its 600-patient target.
“Undoubtedly, the results of the phase 3 study would be highly informative for a regulatory decision on the current ... review for AMX0035,” said Emily Freilich, MD, of the FDA.
However, results aren’t expected until late 2023 or early 2024, which “places the agency in a challenging situation of potentially making a regulatory decision that may not be subsequently confirmed by the results of the ongoing study.”
In June, Amylyx received conditional approval in Canada for the drug, but final approval depends on the outcome of the PHOENIX trial. The FDA does not offer a conditional approval track.
“If AMX0035 is not approved now, the FDA anticipated decision will likely happen in 2025, underscoring the critical importance of today’s outcome,” said Tammy Sarnelli, MPAHC, global head of Regulatory Affairs for Amylyx Pharmaceuticals.
If the FDA were to approve AMX0035 and results from the PHOENIX trial ultimately fail to prove efficacy, Justin Klee, co-CEO and cofounder of Amylyx Pharmaceuticals, said the company would withdraw the drug.
“To be clear, if PHOENIX is not successful, we will do what is right for patients, which includes voluntarily removing the product from the market,” Mr. Klee said.
Regardless of the company’s decision, FDA officials noted that the agency does have the ability to recall a drug from the market if studies show that it no longer meets requirements for approval.
“The FDA, with all due respect, significantly understates the complexity and likelihood of their pulling a product from the market,” Dr. Alexander said. “Whether or not they can ultimately pull a product from the market is no substitute for the evidentiary thresholds that are required for market access.”
A version of this article first appeared on Medscape.com.
In a rare second review of a new drug application,
By a vote of 7-2, the FDA Peripheral and Central Nervous System Drugs Advisory Committee reversed course on AMX0035 (Amylyx Pharmaceuticals), a combination of sodium phenylbutyrate and taurursodiol.
The panel previously voted 6-4 to reject the drug, ruling that data provided by Amylyx had failed to demonstrate that the survival benefit reported in the only clinical trial of AMX0035 so far was a direct result of the drug.
This time, two panelists who previously voted no were swayed by the drug maker’s new analysis of previously presented research, more than 1,300 public comments in support of the drug, supportive testimony from ALS patients and clinicians, and assurances from company executives that Amylyx would pull the drug from the market if results of an ongoing phase 3 clinical trial show the drug doesn’t work.
“As in March, today we have to have an internal dialogue between our scientific scrutiny and clinical compassion,” said Liana G. Apostolova, MD, from Indiana University, Indianapolis, who originally voted against the application.
“Today I also saw additional confirmatory evidence that was not unequivocally persuasive but was nonetheless reassuring,” Dr. Apostolova said. “Because of that I am voting in support of AMX0035.”
A rare second chance
ALS (Lou Gehrig’s disease) is a progressive, fatal neurodegenerative disease affecting nerve cells in the brain and spinal cord that causes loss of motor control. It is rare, affecting about 30,000 people in the United States with another 5,000 new cases diagnosed each year. Most people with the disease die within 2 years of diagnosis.
The FDA has approved two therapies for ALS, but both have limited efficacy.
Typically, FDA approval requires two large studies or one study with a “very persuasive” effect on survival.
Amylyx’s application is based on a single study, the multicenter, two-phase CENTAUR trial. In that trial, 137 people with ALS received AMX0035 or placebo for 24 weeks.
Researchers found that patients receiving AMX0035 had a 25% slower decline in function, compared with the those taking placebo. A change of 20% or more is considered clinically meaningful.
The investigators also found a statistically significant median difference of 4.8 months in time to death, first hospitalization, or tracheostomy/permanent assisted ventilation in the group originally assigned to receive AMX0035 compared with the group originally assigned to receive placebo (hazard ratio, 0.62; P = .023).
In the panel’s previous vote against the drug application, members cited several issues with the study, concluding that it did not offer persuasive or robust evidence of efficacy. They also cited missing data assumptions in the primary analysis, issues of randomization and imbalances in concomitant use of riluzole and edaravone, the two FDA-approved drugs for ALS.
The FDA later requested additional information from Amylyx, delayed its final ruling on the new drug application to Sept. 29, and called for a second review meeting – a virtually unheard-of move.
An FDA review posted in advance of the meeting Sept. 29 had hinted at a different outcome. In that report, regulators said new data from Amylyx were not “sufficiently independent or persuasive” to establish effectiveness.
However, FDA officials in the meeting stressed the importance of considering unmet medical need in ALS in the panel’s decision-making process.
“Recognizing the substantial unmet medical need in ALS, we feel that it is important that the committee is afforded the opportunity to consider this new information, along with the information presented at the prior meeting, in that context,” Billy Dunn, MD, director of the FDA Office of Neuroscience, said during the meeting.
Panelists heard additional data that Amylyx claims confirms the results of the CENTAUR study, including new analyses of the previously submitted survival data and new data from that study and an open-label extension.
They also provided new information on a biomarker data from a phase 2 study of AMX0035 to treat Alzheimer’s disease.
“I think we note the limitations of the analyses, but we still haven’t taken it off the table that they could be considered as confirmatory evidence and that’s why we’re here today,” said Teresa Buracchio, MD, director of the division of neurology for the FDA.
Two members of the panel who voted no in March stuck with that position at the Sept. 29 meeting.
“Unfortunately, I don’t believe the new evidence we’ve reviewed, while promising, combined with that prior evidence, constitutes substantial evidence of effectiveness,” said panelist Caleb Alexander, MD, a professor of epidemiology and medicine at the Johns Hopkins University Center for Drug Safety and Effectiveness, Baltimore.
Dr. Alexander, who also voted no in March, said that post hoc data presented at the meeting were not enough to assuage concerns that led him and others to reject the drug in March.
A challenging situation
Amylyx is currently leading the 48-week international, phase 3, placebo-controlled PHOENIX clinical trial of AMX0035. The study has enrolled about half of its 600-patient target.
“Undoubtedly, the results of the phase 3 study would be highly informative for a regulatory decision on the current ... review for AMX0035,” said Emily Freilich, MD, of the FDA.
However, results aren’t expected until late 2023 or early 2024, which “places the agency in a challenging situation of potentially making a regulatory decision that may not be subsequently confirmed by the results of the ongoing study.”
In June, Amylyx received conditional approval in Canada for the drug, but final approval depends on the outcome of the PHOENIX trial. The FDA does not offer a conditional approval track.
“If AMX0035 is not approved now, the FDA anticipated decision will likely happen in 2025, underscoring the critical importance of today’s outcome,” said Tammy Sarnelli, MPAHC, global head of Regulatory Affairs for Amylyx Pharmaceuticals.
If the FDA were to approve AMX0035 and results from the PHOENIX trial ultimately fail to prove efficacy, Justin Klee, co-CEO and cofounder of Amylyx Pharmaceuticals, said the company would withdraw the drug.
“To be clear, if PHOENIX is not successful, we will do what is right for patients, which includes voluntarily removing the product from the market,” Mr. Klee said.
Regardless of the company’s decision, FDA officials noted that the agency does have the ability to recall a drug from the market if studies show that it no longer meets requirements for approval.
“The FDA, with all due respect, significantly understates the complexity and likelihood of their pulling a product from the market,” Dr. Alexander said. “Whether or not they can ultimately pull a product from the market is no substitute for the evidentiary thresholds that are required for market access.”
A version of this article first appeared on Medscape.com.
In a rare second review of a new drug application,
By a vote of 7-2, the FDA Peripheral and Central Nervous System Drugs Advisory Committee reversed course on AMX0035 (Amylyx Pharmaceuticals), a combination of sodium phenylbutyrate and taurursodiol.
The panel previously voted 6-4 to reject the drug, ruling that data provided by Amylyx had failed to demonstrate that the survival benefit reported in the only clinical trial of AMX0035 so far was a direct result of the drug.
This time, two panelists who previously voted no were swayed by the drug maker’s new analysis of previously presented research, more than 1,300 public comments in support of the drug, supportive testimony from ALS patients and clinicians, and assurances from company executives that Amylyx would pull the drug from the market if results of an ongoing phase 3 clinical trial show the drug doesn’t work.
“As in March, today we have to have an internal dialogue between our scientific scrutiny and clinical compassion,” said Liana G. Apostolova, MD, from Indiana University, Indianapolis, who originally voted against the application.
“Today I also saw additional confirmatory evidence that was not unequivocally persuasive but was nonetheless reassuring,” Dr. Apostolova said. “Because of that I am voting in support of AMX0035.”
A rare second chance
ALS (Lou Gehrig’s disease) is a progressive, fatal neurodegenerative disease affecting nerve cells in the brain and spinal cord that causes loss of motor control. It is rare, affecting about 30,000 people in the United States with another 5,000 new cases diagnosed each year. Most people with the disease die within 2 years of diagnosis.
The FDA has approved two therapies for ALS, but both have limited efficacy.
Typically, FDA approval requires two large studies or one study with a “very persuasive” effect on survival.
Amylyx’s application is based on a single study, the multicenter, two-phase CENTAUR trial. In that trial, 137 people with ALS received AMX0035 or placebo for 24 weeks.
Researchers found that patients receiving AMX0035 had a 25% slower decline in function, compared with the those taking placebo. A change of 20% or more is considered clinically meaningful.
The investigators also found a statistically significant median difference of 4.8 months in time to death, first hospitalization, or tracheostomy/permanent assisted ventilation in the group originally assigned to receive AMX0035 compared with the group originally assigned to receive placebo (hazard ratio, 0.62; P = .023).
In the panel’s previous vote against the drug application, members cited several issues with the study, concluding that it did not offer persuasive or robust evidence of efficacy. They also cited missing data assumptions in the primary analysis, issues of randomization and imbalances in concomitant use of riluzole and edaravone, the two FDA-approved drugs for ALS.
The FDA later requested additional information from Amylyx, delayed its final ruling on the new drug application to Sept. 29, and called for a second review meeting – a virtually unheard-of move.
An FDA review posted in advance of the meeting Sept. 29 had hinted at a different outcome. In that report, regulators said new data from Amylyx were not “sufficiently independent or persuasive” to establish effectiveness.
However, FDA officials in the meeting stressed the importance of considering unmet medical need in ALS in the panel’s decision-making process.
“Recognizing the substantial unmet medical need in ALS, we feel that it is important that the committee is afforded the opportunity to consider this new information, along with the information presented at the prior meeting, in that context,” Billy Dunn, MD, director of the FDA Office of Neuroscience, said during the meeting.
Panelists heard additional data that Amylyx claims confirms the results of the CENTAUR study, including new analyses of the previously submitted survival data and new data from that study and an open-label extension.
They also provided new information on a biomarker data from a phase 2 study of AMX0035 to treat Alzheimer’s disease.
“I think we note the limitations of the analyses, but we still haven’t taken it off the table that they could be considered as confirmatory evidence and that’s why we’re here today,” said Teresa Buracchio, MD, director of the division of neurology for the FDA.
Two members of the panel who voted no in March stuck with that position at the Sept. 29 meeting.
“Unfortunately, I don’t believe the new evidence we’ve reviewed, while promising, combined with that prior evidence, constitutes substantial evidence of effectiveness,” said panelist Caleb Alexander, MD, a professor of epidemiology and medicine at the Johns Hopkins University Center for Drug Safety and Effectiveness, Baltimore.
Dr. Alexander, who also voted no in March, said that post hoc data presented at the meeting were not enough to assuage concerns that led him and others to reject the drug in March.
A challenging situation
Amylyx is currently leading the 48-week international, phase 3, placebo-controlled PHOENIX clinical trial of AMX0035. The study has enrolled about half of its 600-patient target.
“Undoubtedly, the results of the phase 3 study would be highly informative for a regulatory decision on the current ... review for AMX0035,” said Emily Freilich, MD, of the FDA.
However, results aren’t expected until late 2023 or early 2024, which “places the agency in a challenging situation of potentially making a regulatory decision that may not be subsequently confirmed by the results of the ongoing study.”
In June, Amylyx received conditional approval in Canada for the drug, but final approval depends on the outcome of the PHOENIX trial. The FDA does not offer a conditional approval track.
“If AMX0035 is not approved now, the FDA anticipated decision will likely happen in 2025, underscoring the critical importance of today’s outcome,” said Tammy Sarnelli, MPAHC, global head of Regulatory Affairs for Amylyx Pharmaceuticals.
If the FDA were to approve AMX0035 and results from the PHOENIX trial ultimately fail to prove efficacy, Justin Klee, co-CEO and cofounder of Amylyx Pharmaceuticals, said the company would withdraw the drug.
“To be clear, if PHOENIX is not successful, we will do what is right for patients, which includes voluntarily removing the product from the market,” Mr. Klee said.
Regardless of the company’s decision, FDA officials noted that the agency does have the ability to recall a drug from the market if studies show that it no longer meets requirements for approval.
“The FDA, with all due respect, significantly understates the complexity and likelihood of their pulling a product from the market,” Dr. Alexander said. “Whether or not they can ultimately pull a product from the market is no substitute for the evidentiary thresholds that are required for market access.”
A version of this article first appeared on Medscape.com.
Positive top-line phase 3 data for lecanemab in early Alzheimer’s
compared with placebo and decreased amyloid levels in the brain of adults enrolled in a phase 3 trial.
The Clarity AD trial included 1,795 adults with early AD and confirmed amyloid pathology in the brain. Treatment consisted of lecanemab 10 mg/kg biweekly or matching placebo.
Treatment with lecanemab met the primary endpoint, reducing clinical decline on the global cognitive and functional scale, the Clinical Dementia Rating–Sum of Boxes (CDR-SB), at 18 months by 27%, compared with placebo, with a treatment difference in the score change of –0.45 (P = .00005), the companies reported.
Starting as early as 6 months, across all time points, treatment with lecanemab yielded highly statistically significant changes in CDR-SB from baseline, compared with placebo (all P < .01).
The study also met all key secondary endpoints with highly statistically significant results, compared with placebo (P < .01).
Key secondary endpoints, in comparison with placebo, were change from baseline at 18 months in amyloid levels in the brain measured by amyloid PET, the AD Assessment Scale–cognitive subscale14 (ADAS-cog14), the AD Composite Score (ADCOMS), and the AD Cooperative Study–Activities of Daily Living Scale for Mild Cognitive Impairment (ADCS MCI-ADL).
Imaging abnormalities within expectations
Overall, rates of amyloid-related imaging abnormalities (ARIA) related to lecanemab were “within expectations,” the companies said.
The incidence of ARIA related to edema (ARIA-E) was 12.5% in the lecanemab group and 1.7% in the placebo group.
The incidence of symptomatic ARIA-E was 2.8% and 0.0%, respectively, and the rate of cerebral hemorrhage (ARIA-H) was 17.0% and 8.7%. The total incidence of ARIA (ARIA-E and/or ARIA-H) was 21.3% in the lecanemab group and 9.3% in the placebo group.
Full results of the Clarity AD trial will be presented in November at the Clinical Trials on Alzheimer’s Congress.
Incremental benefit
Responding to the findings, the Alzheimer’s Association said in a statement that it “enthusiastically welcomes” the positive findings. It noted that these are “the most encouraging results in clinical trials treating the underlying causes of Alzheimer’s to date.
“For people in the earliest stages of Alzheimer’s, this treatment has the potential to change the course of the disease in a clinically meaningful way. These results indicate lecanemab may give people more time at or near their full abilities to participate in daily life, remain independent and make future health care decisions,” the Alzheimer’s Association added.
Also weighing in, Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, said in a release that “the combination of the biomarker change – reduced amyloid – plus slowing of cognitive decline in this study is encouraging news for the 57 million patients around the world living with Alzheimer’s.
“However, amyloid-clearing drugs will provide an incremental benefit at best, and there is still a pressing need for the next generation of drugs focused on other targets based on our knowledge of the biology of aging,” Dr. Fillit cautioned.
“We are optimistic about the future as many of these drugs are in development, with 75% of drugs in the pipeline now targeting nonamyloid pathways of neurodegeneration,” he added.
In July 2022, the Food and Drug Administration accepted Eisai’s biologics license application for lecanemab under the accelerated approval pathway and granted priority review. Lecanemab has a prescription Drugs User Fee Act action date of Jan. 6, 2023.
A version of this article first appeared on Medscape.com.
compared with placebo and decreased amyloid levels in the brain of adults enrolled in a phase 3 trial.
The Clarity AD trial included 1,795 adults with early AD and confirmed amyloid pathology in the brain. Treatment consisted of lecanemab 10 mg/kg biweekly or matching placebo.
Treatment with lecanemab met the primary endpoint, reducing clinical decline on the global cognitive and functional scale, the Clinical Dementia Rating–Sum of Boxes (CDR-SB), at 18 months by 27%, compared with placebo, with a treatment difference in the score change of –0.45 (P = .00005), the companies reported.
Starting as early as 6 months, across all time points, treatment with lecanemab yielded highly statistically significant changes in CDR-SB from baseline, compared with placebo (all P < .01).
The study also met all key secondary endpoints with highly statistically significant results, compared with placebo (P < .01).
Key secondary endpoints, in comparison with placebo, were change from baseline at 18 months in amyloid levels in the brain measured by amyloid PET, the AD Assessment Scale–cognitive subscale14 (ADAS-cog14), the AD Composite Score (ADCOMS), and the AD Cooperative Study–Activities of Daily Living Scale for Mild Cognitive Impairment (ADCS MCI-ADL).
Imaging abnormalities within expectations
Overall, rates of amyloid-related imaging abnormalities (ARIA) related to lecanemab were “within expectations,” the companies said.
The incidence of ARIA related to edema (ARIA-E) was 12.5% in the lecanemab group and 1.7% in the placebo group.
The incidence of symptomatic ARIA-E was 2.8% and 0.0%, respectively, and the rate of cerebral hemorrhage (ARIA-H) was 17.0% and 8.7%. The total incidence of ARIA (ARIA-E and/or ARIA-H) was 21.3% in the lecanemab group and 9.3% in the placebo group.
Full results of the Clarity AD trial will be presented in November at the Clinical Trials on Alzheimer’s Congress.
Incremental benefit
Responding to the findings, the Alzheimer’s Association said in a statement that it “enthusiastically welcomes” the positive findings. It noted that these are “the most encouraging results in clinical trials treating the underlying causes of Alzheimer’s to date.
“For people in the earliest stages of Alzheimer’s, this treatment has the potential to change the course of the disease in a clinically meaningful way. These results indicate lecanemab may give people more time at or near their full abilities to participate in daily life, remain independent and make future health care decisions,” the Alzheimer’s Association added.
Also weighing in, Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, said in a release that “the combination of the biomarker change – reduced amyloid – plus slowing of cognitive decline in this study is encouraging news for the 57 million patients around the world living with Alzheimer’s.
“However, amyloid-clearing drugs will provide an incremental benefit at best, and there is still a pressing need for the next generation of drugs focused on other targets based on our knowledge of the biology of aging,” Dr. Fillit cautioned.
“We are optimistic about the future as many of these drugs are in development, with 75% of drugs in the pipeline now targeting nonamyloid pathways of neurodegeneration,” he added.
In July 2022, the Food and Drug Administration accepted Eisai’s biologics license application for lecanemab under the accelerated approval pathway and granted priority review. Lecanemab has a prescription Drugs User Fee Act action date of Jan. 6, 2023.
A version of this article first appeared on Medscape.com.
compared with placebo and decreased amyloid levels in the brain of adults enrolled in a phase 3 trial.
The Clarity AD trial included 1,795 adults with early AD and confirmed amyloid pathology in the brain. Treatment consisted of lecanemab 10 mg/kg biweekly or matching placebo.
Treatment with lecanemab met the primary endpoint, reducing clinical decline on the global cognitive and functional scale, the Clinical Dementia Rating–Sum of Boxes (CDR-SB), at 18 months by 27%, compared with placebo, with a treatment difference in the score change of –0.45 (P = .00005), the companies reported.
Starting as early as 6 months, across all time points, treatment with lecanemab yielded highly statistically significant changes in CDR-SB from baseline, compared with placebo (all P < .01).
The study also met all key secondary endpoints with highly statistically significant results, compared with placebo (P < .01).
Key secondary endpoints, in comparison with placebo, were change from baseline at 18 months in amyloid levels in the brain measured by amyloid PET, the AD Assessment Scale–cognitive subscale14 (ADAS-cog14), the AD Composite Score (ADCOMS), and the AD Cooperative Study–Activities of Daily Living Scale for Mild Cognitive Impairment (ADCS MCI-ADL).
Imaging abnormalities within expectations
Overall, rates of amyloid-related imaging abnormalities (ARIA) related to lecanemab were “within expectations,” the companies said.
The incidence of ARIA related to edema (ARIA-E) was 12.5% in the lecanemab group and 1.7% in the placebo group.
The incidence of symptomatic ARIA-E was 2.8% and 0.0%, respectively, and the rate of cerebral hemorrhage (ARIA-H) was 17.0% and 8.7%. The total incidence of ARIA (ARIA-E and/or ARIA-H) was 21.3% in the lecanemab group and 9.3% in the placebo group.
Full results of the Clarity AD trial will be presented in November at the Clinical Trials on Alzheimer’s Congress.
Incremental benefit
Responding to the findings, the Alzheimer’s Association said in a statement that it “enthusiastically welcomes” the positive findings. It noted that these are “the most encouraging results in clinical trials treating the underlying causes of Alzheimer’s to date.
“For people in the earliest stages of Alzheimer’s, this treatment has the potential to change the course of the disease in a clinically meaningful way. These results indicate lecanemab may give people more time at or near their full abilities to participate in daily life, remain independent and make future health care decisions,” the Alzheimer’s Association added.
Also weighing in, Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, said in a release that “the combination of the biomarker change – reduced amyloid – plus slowing of cognitive decline in this study is encouraging news for the 57 million patients around the world living with Alzheimer’s.
“However, amyloid-clearing drugs will provide an incremental benefit at best, and there is still a pressing need for the next generation of drugs focused on other targets based on our knowledge of the biology of aging,” Dr. Fillit cautioned.
“We are optimistic about the future as many of these drugs are in development, with 75% of drugs in the pipeline now targeting nonamyloid pathways of neurodegeneration,” he added.
In July 2022, the Food and Drug Administration accepted Eisai’s biologics license application for lecanemab under the accelerated approval pathway and granted priority review. Lecanemab has a prescription Drugs User Fee Act action date of Jan. 6, 2023.
A version of this article first appeared on Medscape.com.
Understanding of developmental language disorder in children
Developmental language disorder (DLD) is characterized by receptive or expressive language difficulties or both. Children with the neurodevelopmental condition “struggle to comprehend and use their native language for no obvious reason,” said the authors of a new study. This leads to problems with grammar, vocabulary, and holding conversations, and in turn an increased risk of “difficulties when learning to read, underachieving academically, being unemployed, and facing social and mental health challenges.”
The condition is common and estimated to affect 7% of children – approximately two in every classroom – but is “underrecognized” said the authors.
Saloni Krishnan, PhD, reader at Royal Holloway, University of London, who led the study as a research fellow at the University of Oxford, England, explained: “DLD is a relatively unknown and understudied condition, unlike better known neurodevelopmental conditions such as ADHD, dyslexia, or autism.”
It is suspected that children with DLD may have differences in areas of the brain involved with learning habits and rules. “Although we know that DLD does not result from gross neural lesions, we still do not have a clear picture of how brain anatomy differs in children with DLD,” the authors highlighted.
Language learning difficulties linked to brain differences
For their study, published in eLife, researchers used an MRI technique called multiparameter mapping (MPM) to investigate microstructural neural differences in children with DLD. The technique measures the properties of brain tissue and is particularly useful for measuring the amounts of myelin.
“Understanding the neural basis of DLD is particularly challenging given the developmental nature of the disorder, as well as the lack of animal models for understanding language,” explained the authors. However, they pointed out that MPM allows an “unparalleled in vivo method” to investigate microstructural neural changes in children with DLD.
Kate Watkins, PhD, professor of cognitive neuroscience at the University of Oxford and senior author, said: “This type of scan tells us more about the makeup or composition of the brain tissue in different areas.”
As part of the Oxford Brain Organisation in Language Development (OxBOLD) study, the researchers recruited and tested 175 children between the ages of 10 and 15 years. Subsequently, 56 children with typical language development and 33 children with DLD were scanned using MPM.
The researchers compared the two groups and found that children with DLD have less myelin in parts of the brain responsible for speaking, listening, and learning rules and habits.
Specifically, maps of magnetization transfer saturation (MTsat) – which index myelin – in children with DLD showed reductions in MTsat values in the caudate nucleus bilaterally, and in the left ventral sensorimotor cortex and Heschl’s gyrus.
“Our findings using this protocol suggest that the caudate nucleus, as well as regions in the wider speech and language network, show alterations in myelin in children with DLD,” explained the authors.
“Given myelin’s role in enabling fast and reliable communication in the brain, reduced myelin content may explain why children with DLD struggle with speech and language processing,” they highlighted.
Significant advance in DLD understanding
The study findings established changes in striatal and cortical myelin as a “neural basis for DLD,” explained the journal editor, who highlighted that this was a “significant advance” in the understanding of DLD. “These brain differences may explain the poorer language outcomes in this group,” the authors said.
The findings “strongly point” to a role for the striatum in the development of DLD, and this role is likely to be in the “learning of habits and sequences,” the authors said.
They pointed out, however, that myelin patterns can change over development, and that myelination can be observed after successful training. “It is important to assess whether these differences in myelin persist over development in DLD, and if they can be targeted through training using behavioral interventions,” they emphasized.
Professor Watkins commented: “The findings might help us understand the pathways involved at a biological level and ultimately allow us to explain why children with DLD have problems with language learning.”
A spokesperson for the RADLD (Raising Awareness of Developmental Language Disorder) organization, commented: “Developmental language disorder has long been understood to have a neurological basis; however, these differences in the brain development have received limited attention in research.” It added that utilizing new technology helps to better understand the “potential neurological differences” experienced by people with DLD.
More studies are needed to determine if these brain differences cause language problems and how or if experiencing language difficulties could cause these changes in the brain, explained the authors. They hoped that further research may help scientists find new treatments that target these brain differences.
Funding was provided by UK Research and Innovation, Wellcome Trust. The authors declared no competing interests.
A version of this article first appeared on MedscapeUK.
Developmental language disorder (DLD) is characterized by receptive or expressive language difficulties or both. Children with the neurodevelopmental condition “struggle to comprehend and use their native language for no obvious reason,” said the authors of a new study. This leads to problems with grammar, vocabulary, and holding conversations, and in turn an increased risk of “difficulties when learning to read, underachieving academically, being unemployed, and facing social and mental health challenges.”
The condition is common and estimated to affect 7% of children – approximately two in every classroom – but is “underrecognized” said the authors.
Saloni Krishnan, PhD, reader at Royal Holloway, University of London, who led the study as a research fellow at the University of Oxford, England, explained: “DLD is a relatively unknown and understudied condition, unlike better known neurodevelopmental conditions such as ADHD, dyslexia, or autism.”
It is suspected that children with DLD may have differences in areas of the brain involved with learning habits and rules. “Although we know that DLD does not result from gross neural lesions, we still do not have a clear picture of how brain anatomy differs in children with DLD,” the authors highlighted.
Language learning difficulties linked to brain differences
For their study, published in eLife, researchers used an MRI technique called multiparameter mapping (MPM) to investigate microstructural neural differences in children with DLD. The technique measures the properties of brain tissue and is particularly useful for measuring the amounts of myelin.
“Understanding the neural basis of DLD is particularly challenging given the developmental nature of the disorder, as well as the lack of animal models for understanding language,” explained the authors. However, they pointed out that MPM allows an “unparalleled in vivo method” to investigate microstructural neural changes in children with DLD.
Kate Watkins, PhD, professor of cognitive neuroscience at the University of Oxford and senior author, said: “This type of scan tells us more about the makeup or composition of the brain tissue in different areas.”
As part of the Oxford Brain Organisation in Language Development (OxBOLD) study, the researchers recruited and tested 175 children between the ages of 10 and 15 years. Subsequently, 56 children with typical language development and 33 children with DLD were scanned using MPM.
The researchers compared the two groups and found that children with DLD have less myelin in parts of the brain responsible for speaking, listening, and learning rules and habits.
Specifically, maps of magnetization transfer saturation (MTsat) – which index myelin – in children with DLD showed reductions in MTsat values in the caudate nucleus bilaterally, and in the left ventral sensorimotor cortex and Heschl’s gyrus.
“Our findings using this protocol suggest that the caudate nucleus, as well as regions in the wider speech and language network, show alterations in myelin in children with DLD,” explained the authors.
“Given myelin’s role in enabling fast and reliable communication in the brain, reduced myelin content may explain why children with DLD struggle with speech and language processing,” they highlighted.
Significant advance in DLD understanding
The study findings established changes in striatal and cortical myelin as a “neural basis for DLD,” explained the journal editor, who highlighted that this was a “significant advance” in the understanding of DLD. “These brain differences may explain the poorer language outcomes in this group,” the authors said.
The findings “strongly point” to a role for the striatum in the development of DLD, and this role is likely to be in the “learning of habits and sequences,” the authors said.
They pointed out, however, that myelin patterns can change over development, and that myelination can be observed after successful training. “It is important to assess whether these differences in myelin persist over development in DLD, and if they can be targeted through training using behavioral interventions,” they emphasized.
Professor Watkins commented: “The findings might help us understand the pathways involved at a biological level and ultimately allow us to explain why children with DLD have problems with language learning.”
A spokesperson for the RADLD (Raising Awareness of Developmental Language Disorder) organization, commented: “Developmental language disorder has long been understood to have a neurological basis; however, these differences in the brain development have received limited attention in research.” It added that utilizing new technology helps to better understand the “potential neurological differences” experienced by people with DLD.
More studies are needed to determine if these brain differences cause language problems and how or if experiencing language difficulties could cause these changes in the brain, explained the authors. They hoped that further research may help scientists find new treatments that target these brain differences.
Funding was provided by UK Research and Innovation, Wellcome Trust. The authors declared no competing interests.
A version of this article first appeared on MedscapeUK.
Developmental language disorder (DLD) is characterized by receptive or expressive language difficulties or both. Children with the neurodevelopmental condition “struggle to comprehend and use their native language for no obvious reason,” said the authors of a new study. This leads to problems with grammar, vocabulary, and holding conversations, and in turn an increased risk of “difficulties when learning to read, underachieving academically, being unemployed, and facing social and mental health challenges.”
The condition is common and estimated to affect 7% of children – approximately two in every classroom – but is “underrecognized” said the authors.
Saloni Krishnan, PhD, reader at Royal Holloway, University of London, who led the study as a research fellow at the University of Oxford, England, explained: “DLD is a relatively unknown and understudied condition, unlike better known neurodevelopmental conditions such as ADHD, dyslexia, or autism.”
It is suspected that children with DLD may have differences in areas of the brain involved with learning habits and rules. “Although we know that DLD does not result from gross neural lesions, we still do not have a clear picture of how brain anatomy differs in children with DLD,” the authors highlighted.
Language learning difficulties linked to brain differences
For their study, published in eLife, researchers used an MRI technique called multiparameter mapping (MPM) to investigate microstructural neural differences in children with DLD. The technique measures the properties of brain tissue and is particularly useful for measuring the amounts of myelin.
“Understanding the neural basis of DLD is particularly challenging given the developmental nature of the disorder, as well as the lack of animal models for understanding language,” explained the authors. However, they pointed out that MPM allows an “unparalleled in vivo method” to investigate microstructural neural changes in children with DLD.
Kate Watkins, PhD, professor of cognitive neuroscience at the University of Oxford and senior author, said: “This type of scan tells us more about the makeup or composition of the brain tissue in different areas.”
As part of the Oxford Brain Organisation in Language Development (OxBOLD) study, the researchers recruited and tested 175 children between the ages of 10 and 15 years. Subsequently, 56 children with typical language development and 33 children with DLD were scanned using MPM.
The researchers compared the two groups and found that children with DLD have less myelin in parts of the brain responsible for speaking, listening, and learning rules and habits.
Specifically, maps of magnetization transfer saturation (MTsat) – which index myelin – in children with DLD showed reductions in MTsat values in the caudate nucleus bilaterally, and in the left ventral sensorimotor cortex and Heschl’s gyrus.
“Our findings using this protocol suggest that the caudate nucleus, as well as regions in the wider speech and language network, show alterations in myelin in children with DLD,” explained the authors.
“Given myelin’s role in enabling fast and reliable communication in the brain, reduced myelin content may explain why children with DLD struggle with speech and language processing,” they highlighted.
Significant advance in DLD understanding
The study findings established changes in striatal and cortical myelin as a “neural basis for DLD,” explained the journal editor, who highlighted that this was a “significant advance” in the understanding of DLD. “These brain differences may explain the poorer language outcomes in this group,” the authors said.
The findings “strongly point” to a role for the striatum in the development of DLD, and this role is likely to be in the “learning of habits and sequences,” the authors said.
They pointed out, however, that myelin patterns can change over development, and that myelination can be observed after successful training. “It is important to assess whether these differences in myelin persist over development in DLD, and if they can be targeted through training using behavioral interventions,” they emphasized.
Professor Watkins commented: “The findings might help us understand the pathways involved at a biological level and ultimately allow us to explain why children with DLD have problems with language learning.”
A spokesperson for the RADLD (Raising Awareness of Developmental Language Disorder) organization, commented: “Developmental language disorder has long been understood to have a neurological basis; however, these differences in the brain development have received limited attention in research.” It added that utilizing new technology helps to better understand the “potential neurological differences” experienced by people with DLD.
More studies are needed to determine if these brain differences cause language problems and how or if experiencing language difficulties could cause these changes in the brain, explained the authors. They hoped that further research may help scientists find new treatments that target these brain differences.
Funding was provided by UK Research and Innovation, Wellcome Trust. The authors declared no competing interests.
A version of this article first appeared on MedscapeUK.
Not just a bad dream: Nightmares may predict dementia
Results from a large cohort study showed that healthy middle-aged adults who had bad dreams at least once a week were four times more likely to experience cognitive decline over the following decade, and older adults were twice as likely to be diagnosed with dementia, compared with peers who never had bad dreams.
Frequent nightmares may “identify people who are at high risk of developing dementia in the future, several years or decades before the characteristic memory and thinking problems emerge,” study investigator Abidemi Otaiku, BMBS, University of Birmingham, England, said in an interview.
“This would be the optimum time for doctors to intervene to try and slow down or prevent dementia from developing,” Dr. Otaiku said.
The findings were published online in The Lancet journal eClinicalMedicine).
Distressing dreams
Distressing dreams have been previously associated with faster cognitive decline and increased dementia risk in patients with Parkinson’s disease (PD), but whether the same holds for individuals from the general population without PD is unknown.
To investigate, Dr. Otaiku examined data from three community-based cohorts in the United States. This included 605 middle-aged adults (aged 35-64 years) who were followed for up to 13 years and 2,600 adults aged 79 and older who were followed for up to 7 years. All were considered cognitively normal at baseline.
The prevalence of frequent distressing dreams, defined as occurring “once a week or more,” was higher in the older cohort compared with the middle-aged cohort (6.9% vs. 6.0%, respectively).
This is in line with other research that showed distressing dreams remain relatively stable throughout early adulthood and then progressively increase in prevalence from middle to older adulthood.
After adjustment for all covariates, a higher frequency of distressing dreams was linearly and statistically significantly associated with a higher risk for cognitive decline in middle-aged adults (P = .016) and a higher risk for dementia in older adults (P = .001).
In the fully adjusted model, compared with middle-aged adults who never had bad dreams, those who reported having one or more bad dreams weekly had a fourfold risk for cognitive decline (adjusted odds ratio [aOR], 3.99; 95% confidence interval [CI], 1.07-14.85).
Older adults who had one or more bad dreams weekly had a greater than twofold increased risk for developing dementia (aOR, 2.21; 95% CI, 1.35-3.62).
Early days
In sex-stratified analyses, distressing dreams were strongly and statistically significantly associated with cognitive decline and dementia in men, but were only weakly and nonsignificantly associated with cognitive decline and dementia in women.
Dr. Otaiku said he suspects some individuals in the preclinical phase of dementia have “subtle neurodegeneration occurring over time in the right frontal lobe: the area of the brain that helps to downregulate negative emotions whilst we are awake, and also whilst we are dreaming.”
This could result in “depression and anxiety in the day, and nightmares and bad dreams during the night,” he said.
It is possible that treatment for frequent nightmares may help to slow cognitive decline and delay or prevent dementia, Dr. Otaiku added.
He noted that prazosin is used to treat nightmares and has been shown to prevent memory decline and reduce amyloid B generation in preclinical studies of Alzheimer’s disease.
“This is an exciting prospect [but] it is still early days and we will need research to see whether treating nightmares might help to reduce dementia risk down the line,” Dr. Otaiku said.
Credible research
In an interview regarding these findings, Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, said: “This is credible research consistent with the idea that sleep disturbances may be a risk factor or warning sign of cognitive decline.”
She added that “what’s novel here” is the researchers examined distressing dreams – not more physical sleep disturbances and disorders such as insomnia or apnea.
“However, nightmares can disturb sleep in the same way these disorders do by waking people up in the middle of the night,” said Dr. Carrillo, who was not involved with the study.
“Previous research has pointed to nightmares being indicative of potential changes in the brain that can precede other dementias like Parkinson’s disease. More research is needed to tease out what exactly is happening in the brain during nightmares that may be contributing to this increased risk,” she said.
Dr. Carrillo noted that “getting good sleep” is important for overall health, which includes brain health.
“The good news is there are treatments – both drug and nondrug – that can help address sleep disturbances,” she added.
This study received no external funding. Dr. Otaiku and Dr. Carrillo have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results from a large cohort study showed that healthy middle-aged adults who had bad dreams at least once a week were four times more likely to experience cognitive decline over the following decade, and older adults were twice as likely to be diagnosed with dementia, compared with peers who never had bad dreams.
Frequent nightmares may “identify people who are at high risk of developing dementia in the future, several years or decades before the characteristic memory and thinking problems emerge,” study investigator Abidemi Otaiku, BMBS, University of Birmingham, England, said in an interview.
“This would be the optimum time for doctors to intervene to try and slow down or prevent dementia from developing,” Dr. Otaiku said.
The findings were published online in The Lancet journal eClinicalMedicine).
Distressing dreams
Distressing dreams have been previously associated with faster cognitive decline and increased dementia risk in patients with Parkinson’s disease (PD), but whether the same holds for individuals from the general population without PD is unknown.
To investigate, Dr. Otaiku examined data from three community-based cohorts in the United States. This included 605 middle-aged adults (aged 35-64 years) who were followed for up to 13 years and 2,600 adults aged 79 and older who were followed for up to 7 years. All were considered cognitively normal at baseline.
The prevalence of frequent distressing dreams, defined as occurring “once a week or more,” was higher in the older cohort compared with the middle-aged cohort (6.9% vs. 6.0%, respectively).
This is in line with other research that showed distressing dreams remain relatively stable throughout early adulthood and then progressively increase in prevalence from middle to older adulthood.
After adjustment for all covariates, a higher frequency of distressing dreams was linearly and statistically significantly associated with a higher risk for cognitive decline in middle-aged adults (P = .016) and a higher risk for dementia in older adults (P = .001).
In the fully adjusted model, compared with middle-aged adults who never had bad dreams, those who reported having one or more bad dreams weekly had a fourfold risk for cognitive decline (adjusted odds ratio [aOR], 3.99; 95% confidence interval [CI], 1.07-14.85).
Older adults who had one or more bad dreams weekly had a greater than twofold increased risk for developing dementia (aOR, 2.21; 95% CI, 1.35-3.62).
Early days
In sex-stratified analyses, distressing dreams were strongly and statistically significantly associated with cognitive decline and dementia in men, but were only weakly and nonsignificantly associated with cognitive decline and dementia in women.
Dr. Otaiku said he suspects some individuals in the preclinical phase of dementia have “subtle neurodegeneration occurring over time in the right frontal lobe: the area of the brain that helps to downregulate negative emotions whilst we are awake, and also whilst we are dreaming.”
This could result in “depression and anxiety in the day, and nightmares and bad dreams during the night,” he said.
It is possible that treatment for frequent nightmares may help to slow cognitive decline and delay or prevent dementia, Dr. Otaiku added.
He noted that prazosin is used to treat nightmares and has been shown to prevent memory decline and reduce amyloid B generation in preclinical studies of Alzheimer’s disease.
“This is an exciting prospect [but] it is still early days and we will need research to see whether treating nightmares might help to reduce dementia risk down the line,” Dr. Otaiku said.
Credible research
In an interview regarding these findings, Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, said: “This is credible research consistent with the idea that sleep disturbances may be a risk factor or warning sign of cognitive decline.”
She added that “what’s novel here” is the researchers examined distressing dreams – not more physical sleep disturbances and disorders such as insomnia or apnea.
“However, nightmares can disturb sleep in the same way these disorders do by waking people up in the middle of the night,” said Dr. Carrillo, who was not involved with the study.
“Previous research has pointed to nightmares being indicative of potential changes in the brain that can precede other dementias like Parkinson’s disease. More research is needed to tease out what exactly is happening in the brain during nightmares that may be contributing to this increased risk,” she said.
Dr. Carrillo noted that “getting good sleep” is important for overall health, which includes brain health.
“The good news is there are treatments – both drug and nondrug – that can help address sleep disturbances,” she added.
This study received no external funding. Dr. Otaiku and Dr. Carrillo have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results from a large cohort study showed that healthy middle-aged adults who had bad dreams at least once a week were four times more likely to experience cognitive decline over the following decade, and older adults were twice as likely to be diagnosed with dementia, compared with peers who never had bad dreams.
Frequent nightmares may “identify people who are at high risk of developing dementia in the future, several years or decades before the characteristic memory and thinking problems emerge,” study investigator Abidemi Otaiku, BMBS, University of Birmingham, England, said in an interview.
“This would be the optimum time for doctors to intervene to try and slow down or prevent dementia from developing,” Dr. Otaiku said.
The findings were published online in The Lancet journal eClinicalMedicine).
Distressing dreams
Distressing dreams have been previously associated with faster cognitive decline and increased dementia risk in patients with Parkinson’s disease (PD), but whether the same holds for individuals from the general population without PD is unknown.
To investigate, Dr. Otaiku examined data from three community-based cohorts in the United States. This included 605 middle-aged adults (aged 35-64 years) who were followed for up to 13 years and 2,600 adults aged 79 and older who were followed for up to 7 years. All were considered cognitively normal at baseline.
The prevalence of frequent distressing dreams, defined as occurring “once a week or more,” was higher in the older cohort compared with the middle-aged cohort (6.9% vs. 6.0%, respectively).
This is in line with other research that showed distressing dreams remain relatively stable throughout early adulthood and then progressively increase in prevalence from middle to older adulthood.
After adjustment for all covariates, a higher frequency of distressing dreams was linearly and statistically significantly associated with a higher risk for cognitive decline in middle-aged adults (P = .016) and a higher risk for dementia in older adults (P = .001).
In the fully adjusted model, compared with middle-aged adults who never had bad dreams, those who reported having one or more bad dreams weekly had a fourfold risk for cognitive decline (adjusted odds ratio [aOR], 3.99; 95% confidence interval [CI], 1.07-14.85).
Older adults who had one or more bad dreams weekly had a greater than twofold increased risk for developing dementia (aOR, 2.21; 95% CI, 1.35-3.62).
Early days
In sex-stratified analyses, distressing dreams were strongly and statistically significantly associated with cognitive decline and dementia in men, but were only weakly and nonsignificantly associated with cognitive decline and dementia in women.
Dr. Otaiku said he suspects some individuals in the preclinical phase of dementia have “subtle neurodegeneration occurring over time in the right frontal lobe: the area of the brain that helps to downregulate negative emotions whilst we are awake, and also whilst we are dreaming.”
This could result in “depression and anxiety in the day, and nightmares and bad dreams during the night,” he said.
It is possible that treatment for frequent nightmares may help to slow cognitive decline and delay or prevent dementia, Dr. Otaiku added.
He noted that prazosin is used to treat nightmares and has been shown to prevent memory decline and reduce amyloid B generation in preclinical studies of Alzheimer’s disease.
“This is an exciting prospect [but] it is still early days and we will need research to see whether treating nightmares might help to reduce dementia risk down the line,” Dr. Otaiku said.
Credible research
In an interview regarding these findings, Maria C. Carrillo, PhD, chief science officer for the Alzheimer’s Association, said: “This is credible research consistent with the idea that sleep disturbances may be a risk factor or warning sign of cognitive decline.”
She added that “what’s novel here” is the researchers examined distressing dreams – not more physical sleep disturbances and disorders such as insomnia or apnea.
“However, nightmares can disturb sleep in the same way these disorders do by waking people up in the middle of the night,” said Dr. Carrillo, who was not involved with the study.
“Previous research has pointed to nightmares being indicative of potential changes in the brain that can precede other dementias like Parkinson’s disease. More research is needed to tease out what exactly is happening in the brain during nightmares that may be contributing to this increased risk,” she said.
Dr. Carrillo noted that “getting good sleep” is important for overall health, which includes brain health.
“The good news is there are treatments – both drug and nondrug – that can help address sleep disturbances,” she added.
This study received no external funding. Dr. Otaiku and Dr. Carrillo have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECLINICALMEDICINE
COVID vaccination does not appear to worsen symptoms of Parkinson’s disease
Nonmotor symptoms seemed to improve after SARS-CoV-2 vaccination, although the investigators could not verify a causal relationship.
Vaccination programs should continue for patients with Parkinson’s disease, they said, reporting their clinical results at the International Congress of Parkinson’s Disease and Movement Disorders.
The International Parkinson and Movement Disorder Society has recommended vaccining patients with Parkinson’s disease. “All approved mRNA-based and viral vector vaccines are not expected to interact with Parkinson’s disease, but patients [still] report concern with regard to the benefits, risks, and safeness in Parkinson’s disease,” Mayela Rodríguez-Violante, MD, MSc, and colleagues wrote in an abstract of their findings.
Social isolation may be contributing to these beliefs and concerns, though this is inconclusive.
Investigators from Mexico City conducted a retrospective study of patients with Parkinson’s disease to see how COVID-19 vaccination affected motor and nonmotor symptoms. They enlisted 60 patients (66.7% were male; aged 65.7 ± 11.35 years) who received either a vector-viral vaccine (Vaxzevria Coronavirus) or an mRNA vaccine (BNT162b2).
A Wilcoxon signed-rank test assessed scale differences before and after vaccination, measuring motor involvement (Unified Parkinson’s Disease Rating Scale), nonmotor involvement (Non-Motor Rating Scale [NMSS]), cognitive impairment (Montreal Cognitive Assessment), and quality of life (8-item Parkinson’s Disease Questionnaire index).
Investigators found no significant difference between scales, although they did notice a marked improvement in non-motor symptoms.
“The main takeaway is that vaccination against COVID-19 does not appear to worsen motor or nonmotor symptoms in persons with Parkinson’s disease. The benefits outweigh the risks,” said Dr. Rodríguez-Violante, the study’s lead author and a movement disorder specialist at the National Institute of Neurology and Neurosurgery, Mexico City.
Next steps are to increase the sample size to see if it’s possible to have a similar number in terms of type of vaccine, said Dr. Rodríguez-Violante. “Also, the data presented refers to primary series doses so booster effects will also be studied.”
Few studies have looked at vaccines and their possible effects on this patient population. However, a 2021 study of 181 patients with Parkinson’s disease reported that 2 (1.1%) had adverse effects after receiving the BNT162b2 mRNA vaccine. One of the patients, a 61-year-old woman with a decade-long history of Parkinson’s disease, developed severe, continuous, generalized dyskinesia 6 hours after a first dose of vaccine. The second patient was 79 years old and had Parkinson’s disease for 5 years. She developed fever, confusion, delusions, and continuous severe dyskinesia for 3 days following her vaccination.
“This highlights that there is a variability in the response triggered by the vaccine that might likely depend on individual immunological profiles … clinicians should be aware of this possibility and monitor their patients after they receive their vaccination,” Roberto Erro, MD, PhD and colleagues wrote in the Movement Disorders journal.
Nonmotor symptoms seemed to improve after SARS-CoV-2 vaccination, although the investigators could not verify a causal relationship.
Vaccination programs should continue for patients with Parkinson’s disease, they said, reporting their clinical results at the International Congress of Parkinson’s Disease and Movement Disorders.
The International Parkinson and Movement Disorder Society has recommended vaccining patients with Parkinson’s disease. “All approved mRNA-based and viral vector vaccines are not expected to interact with Parkinson’s disease, but patients [still] report concern with regard to the benefits, risks, and safeness in Parkinson’s disease,” Mayela Rodríguez-Violante, MD, MSc, and colleagues wrote in an abstract of their findings.
Social isolation may be contributing to these beliefs and concerns, though this is inconclusive.
Investigators from Mexico City conducted a retrospective study of patients with Parkinson’s disease to see how COVID-19 vaccination affected motor and nonmotor symptoms. They enlisted 60 patients (66.7% were male; aged 65.7 ± 11.35 years) who received either a vector-viral vaccine (Vaxzevria Coronavirus) or an mRNA vaccine (BNT162b2).
A Wilcoxon signed-rank test assessed scale differences before and after vaccination, measuring motor involvement (Unified Parkinson’s Disease Rating Scale), nonmotor involvement (Non-Motor Rating Scale [NMSS]), cognitive impairment (Montreal Cognitive Assessment), and quality of life (8-item Parkinson’s Disease Questionnaire index).
Investigators found no significant difference between scales, although they did notice a marked improvement in non-motor symptoms.
“The main takeaway is that vaccination against COVID-19 does not appear to worsen motor or nonmotor symptoms in persons with Parkinson’s disease. The benefits outweigh the risks,” said Dr. Rodríguez-Violante, the study’s lead author and a movement disorder specialist at the National Institute of Neurology and Neurosurgery, Mexico City.
Next steps are to increase the sample size to see if it’s possible to have a similar number in terms of type of vaccine, said Dr. Rodríguez-Violante. “Also, the data presented refers to primary series doses so booster effects will also be studied.”
Few studies have looked at vaccines and their possible effects on this patient population. However, a 2021 study of 181 patients with Parkinson’s disease reported that 2 (1.1%) had adverse effects after receiving the BNT162b2 mRNA vaccine. One of the patients, a 61-year-old woman with a decade-long history of Parkinson’s disease, developed severe, continuous, generalized dyskinesia 6 hours after a first dose of vaccine. The second patient was 79 years old and had Parkinson’s disease for 5 years. She developed fever, confusion, delusions, and continuous severe dyskinesia for 3 days following her vaccination.
“This highlights that there is a variability in the response triggered by the vaccine that might likely depend on individual immunological profiles … clinicians should be aware of this possibility and monitor their patients after they receive their vaccination,” Roberto Erro, MD, PhD and colleagues wrote in the Movement Disorders journal.
Nonmotor symptoms seemed to improve after SARS-CoV-2 vaccination, although the investigators could not verify a causal relationship.
Vaccination programs should continue for patients with Parkinson’s disease, they said, reporting their clinical results at the International Congress of Parkinson’s Disease and Movement Disorders.
The International Parkinson and Movement Disorder Society has recommended vaccining patients with Parkinson’s disease. “All approved mRNA-based and viral vector vaccines are not expected to interact with Parkinson’s disease, but patients [still] report concern with regard to the benefits, risks, and safeness in Parkinson’s disease,” Mayela Rodríguez-Violante, MD, MSc, and colleagues wrote in an abstract of their findings.
Social isolation may be contributing to these beliefs and concerns, though this is inconclusive.
Investigators from Mexico City conducted a retrospective study of patients with Parkinson’s disease to see how COVID-19 vaccination affected motor and nonmotor symptoms. They enlisted 60 patients (66.7% were male; aged 65.7 ± 11.35 years) who received either a vector-viral vaccine (Vaxzevria Coronavirus) or an mRNA vaccine (BNT162b2).
A Wilcoxon signed-rank test assessed scale differences before and after vaccination, measuring motor involvement (Unified Parkinson’s Disease Rating Scale), nonmotor involvement (Non-Motor Rating Scale [NMSS]), cognitive impairment (Montreal Cognitive Assessment), and quality of life (8-item Parkinson’s Disease Questionnaire index).
Investigators found no significant difference between scales, although they did notice a marked improvement in non-motor symptoms.
“The main takeaway is that vaccination against COVID-19 does not appear to worsen motor or nonmotor symptoms in persons with Parkinson’s disease. The benefits outweigh the risks,” said Dr. Rodríguez-Violante, the study’s lead author and a movement disorder specialist at the National Institute of Neurology and Neurosurgery, Mexico City.
Next steps are to increase the sample size to see if it’s possible to have a similar number in terms of type of vaccine, said Dr. Rodríguez-Violante. “Also, the data presented refers to primary series doses so booster effects will also be studied.”
Few studies have looked at vaccines and their possible effects on this patient population. However, a 2021 study of 181 patients with Parkinson’s disease reported that 2 (1.1%) had adverse effects after receiving the BNT162b2 mRNA vaccine. One of the patients, a 61-year-old woman with a decade-long history of Parkinson’s disease, developed severe, continuous, generalized dyskinesia 6 hours after a first dose of vaccine. The second patient was 79 years old and had Parkinson’s disease for 5 years. She developed fever, confusion, delusions, and continuous severe dyskinesia for 3 days following her vaccination.
“This highlights that there is a variability in the response triggered by the vaccine that might likely depend on individual immunological profiles … clinicians should be aware of this possibility and monitor their patients after they receive their vaccination,” Roberto Erro, MD, PhD and colleagues wrote in the Movement Disorders journal.
FROM MDS 2022
A history of head trauma may predict Parkinson’s disease progression
, new research suggests.
In a longitudinal online study, among patients with Parkinson’s disease who had a history of head injury, motor impairment developed 25% faster and cognitive impairment developed 45% faster than among those without such a history.
In addition, severe head injuries were associated with an even more rapid onset of impairment. The results give weight to the idea that “it’s head injuries themselves” prior to the development of Parkinson’s disease that might exacerbate motor and cognitive symptoms, said study investigator Ethan Brown, MD, assistant professor, Weill Institute of Neurosciences, department of neurology, University of California, San Francisco.
The findings emphasize the importance of “doing everything we can” to prevent falls and head injuries for patients with Parkinson’s disease, Dr. Brown said.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Reverse causality concerns
Head injury is a risk factor for Parkinson’s disease, but its relationship to Parkinson’s disease progression is not well established. “There has always been this concern in Parkinson’s disease that maybe it’s problems with motor impairment that lead to head injuries, so reverse causality is an issue,” said Dr. Brown. “We wanted to look at whether risk factors we know relate to the development of Parkinson’s disease can also have a bearing on its progression,” he added.
The analysis was part of the online Fox Insight study that is evaluating motor and nonmotor symptoms in individuals with and those without Parkinson’s disease. The study included participants who had completed questionnaires on such things as head trauma.
The study included 1,065 patients (47% women; mean age, 63 years) with Parkinson’s disease who reported having had a head injury at least 5 years prior to their diagnosis. Among the participants, the mean duration of Parkinson’s disease was 7.5 years.
The investigators employed a 5-year lag time in their study to exclude head injuries caused by early motor dysfunction, they noted. “We wanted to look at people who had these head injuries we think might be part of the cause of Parkinson’s disease as opposed to a result of them,” Dr. Brown said.
In this head injury group, 51% had received one head injury, 28% had received two injuries, and 22% had received more than two injuries.
The study also included 1,457 participants (56% women; mean age, 65 years) with Parkinson’s disease who had not had a head injury prior to their diagnosis. Of these patients, the mean time with a Parkinson’s disease diagnosis was 8 years.
Dr. Brown noted that the age and sex distribution of the study group was “probably representative” of the general Parkinson’s disease population. However, because the participants had to be able to go online and complete questionnaires, it is unlikely that, among these patients, Parkinson’s disease was far advanced, he said.
The investigators adjusted for age, sex, years of education, and Parkinson’s disease duration.
Two-hit hypothesis?
The researchers compared time from diagnosis to the development of significant motor impairment, such as the need for assistance with walking, and cognitive impairment, such as having a score of less than 43 on the Penn Daily Activities Questionnaire.
They also examined the role of more severe head injuries. In the head injury group, over half (54%) had had a severe head injury, including 543 who had lost consciousness and others who had suffered a fracture or had had a seizure.
Results showed that the adjusted hazard ratio for developing motor impairment among those with a head injury, compared with those who had not had a head injury was 1.24 (95% confidence interval, 1.01-1.53; P = .037). For severe injuries, the aHR for motor impairment was 1.44 (95% CI, 1.13-1.83; P = .003).
For cognitive impairment, the aHR for those with versus without head injuries was 1.45 (95% CI, 1.14-1.86; P = .003); and for severe injuries, the aHR was 1.49 (95% CI, 1.11-2.0; P = .008).
Aside from severity, the researchers did not examine subgroups. However, Dr. Brown reported that his team would like to stratify results by sex and other variables in the future.
He noted that various mechanisms may explain why Parkinson’s disease progression is faster for patients who have a history of head injury, compared with others. Chronic inflammation due to the injury and “co-pathology” might play some role, he said. He noted that head injuries are associated with cognitive impairment in other conditions, including Alzheimer’s disease.
There is also the “two hit” hypothesis, Dr. Brown said. “A head injury could cause such broad damage that once people develop Parkinson’s disease, it’s harder for them to compensate.”
Dr. Brown also noted there might have been a “higher magnitude” of a difference between groups had the study captured participants with more severe symptoms.
‘Provocative’ findings
Michael S. Okun, MD, medical advisor at the Parkinson’s Foundation and professor and director at the Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the new data are “provocative.”
“The idea that a head injury may be important in predicting how quickly and how severely deficits will manifest could be important to the treating clinician,” said Dr. Okun, who was not involved with the research.
He noted that the results suggest clinicians should elicit more information from patients about head trauma. “They should be seeking more than a binary ‘yes or no’ answer to head injury when questioning patients,” he added.
Dr. Okun reiterated that head injury is a “known and important risk factor” not only for Parkinson’s disease but also for other neurodegenerative diseases. “It’s important to counsel patients about the association,” he said.
The study was supported by the Michael J. Fox Foundation. Dr. Brown reports having received grant support from the Michael J. Fox Foundation. Dr. Okun has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
In a longitudinal online study, among patients with Parkinson’s disease who had a history of head injury, motor impairment developed 25% faster and cognitive impairment developed 45% faster than among those without such a history.
In addition, severe head injuries were associated with an even more rapid onset of impairment. The results give weight to the idea that “it’s head injuries themselves” prior to the development of Parkinson’s disease that might exacerbate motor and cognitive symptoms, said study investigator Ethan Brown, MD, assistant professor, Weill Institute of Neurosciences, department of neurology, University of California, San Francisco.
The findings emphasize the importance of “doing everything we can” to prevent falls and head injuries for patients with Parkinson’s disease, Dr. Brown said.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Reverse causality concerns
Head injury is a risk factor for Parkinson’s disease, but its relationship to Parkinson’s disease progression is not well established. “There has always been this concern in Parkinson’s disease that maybe it’s problems with motor impairment that lead to head injuries, so reverse causality is an issue,” said Dr. Brown. “We wanted to look at whether risk factors we know relate to the development of Parkinson’s disease can also have a bearing on its progression,” he added.
The analysis was part of the online Fox Insight study that is evaluating motor and nonmotor symptoms in individuals with and those without Parkinson’s disease. The study included participants who had completed questionnaires on such things as head trauma.
The study included 1,065 patients (47% women; mean age, 63 years) with Parkinson’s disease who reported having had a head injury at least 5 years prior to their diagnosis. Among the participants, the mean duration of Parkinson’s disease was 7.5 years.
The investigators employed a 5-year lag time in their study to exclude head injuries caused by early motor dysfunction, they noted. “We wanted to look at people who had these head injuries we think might be part of the cause of Parkinson’s disease as opposed to a result of them,” Dr. Brown said.
In this head injury group, 51% had received one head injury, 28% had received two injuries, and 22% had received more than two injuries.
The study also included 1,457 participants (56% women; mean age, 65 years) with Parkinson’s disease who had not had a head injury prior to their diagnosis. Of these patients, the mean time with a Parkinson’s disease diagnosis was 8 years.
Dr. Brown noted that the age and sex distribution of the study group was “probably representative” of the general Parkinson’s disease population. However, because the participants had to be able to go online and complete questionnaires, it is unlikely that, among these patients, Parkinson’s disease was far advanced, he said.
The investigators adjusted for age, sex, years of education, and Parkinson’s disease duration.
Two-hit hypothesis?
The researchers compared time from diagnosis to the development of significant motor impairment, such as the need for assistance with walking, and cognitive impairment, such as having a score of less than 43 on the Penn Daily Activities Questionnaire.
They also examined the role of more severe head injuries. In the head injury group, over half (54%) had had a severe head injury, including 543 who had lost consciousness and others who had suffered a fracture or had had a seizure.
Results showed that the adjusted hazard ratio for developing motor impairment among those with a head injury, compared with those who had not had a head injury was 1.24 (95% confidence interval, 1.01-1.53; P = .037). For severe injuries, the aHR for motor impairment was 1.44 (95% CI, 1.13-1.83; P = .003).
For cognitive impairment, the aHR for those with versus without head injuries was 1.45 (95% CI, 1.14-1.86; P = .003); and for severe injuries, the aHR was 1.49 (95% CI, 1.11-2.0; P = .008).
Aside from severity, the researchers did not examine subgroups. However, Dr. Brown reported that his team would like to stratify results by sex and other variables in the future.
He noted that various mechanisms may explain why Parkinson’s disease progression is faster for patients who have a history of head injury, compared with others. Chronic inflammation due to the injury and “co-pathology” might play some role, he said. He noted that head injuries are associated with cognitive impairment in other conditions, including Alzheimer’s disease.
There is also the “two hit” hypothesis, Dr. Brown said. “A head injury could cause such broad damage that once people develop Parkinson’s disease, it’s harder for them to compensate.”
Dr. Brown also noted there might have been a “higher magnitude” of a difference between groups had the study captured participants with more severe symptoms.
‘Provocative’ findings
Michael S. Okun, MD, medical advisor at the Parkinson’s Foundation and professor and director at the Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the new data are “provocative.”
“The idea that a head injury may be important in predicting how quickly and how severely deficits will manifest could be important to the treating clinician,” said Dr. Okun, who was not involved with the research.
He noted that the results suggest clinicians should elicit more information from patients about head trauma. “They should be seeking more than a binary ‘yes or no’ answer to head injury when questioning patients,” he added.
Dr. Okun reiterated that head injury is a “known and important risk factor” not only for Parkinson’s disease but also for other neurodegenerative diseases. “It’s important to counsel patients about the association,” he said.
The study was supported by the Michael J. Fox Foundation. Dr. Brown reports having received grant support from the Michael J. Fox Foundation. Dr. Okun has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
In a longitudinal online study, among patients with Parkinson’s disease who had a history of head injury, motor impairment developed 25% faster and cognitive impairment developed 45% faster than among those without such a history.
In addition, severe head injuries were associated with an even more rapid onset of impairment. The results give weight to the idea that “it’s head injuries themselves” prior to the development of Parkinson’s disease that might exacerbate motor and cognitive symptoms, said study investigator Ethan Brown, MD, assistant professor, Weill Institute of Neurosciences, department of neurology, University of California, San Francisco.
The findings emphasize the importance of “doing everything we can” to prevent falls and head injuries for patients with Parkinson’s disease, Dr. Brown said.
The findings were presented at the International Congress of Parkinson’s Disease and Movement Disorders.
Reverse causality concerns
Head injury is a risk factor for Parkinson’s disease, but its relationship to Parkinson’s disease progression is not well established. “There has always been this concern in Parkinson’s disease that maybe it’s problems with motor impairment that lead to head injuries, so reverse causality is an issue,” said Dr. Brown. “We wanted to look at whether risk factors we know relate to the development of Parkinson’s disease can also have a bearing on its progression,” he added.
The analysis was part of the online Fox Insight study that is evaluating motor and nonmotor symptoms in individuals with and those without Parkinson’s disease. The study included participants who had completed questionnaires on such things as head trauma.
The study included 1,065 patients (47% women; mean age, 63 years) with Parkinson’s disease who reported having had a head injury at least 5 years prior to their diagnosis. Among the participants, the mean duration of Parkinson’s disease was 7.5 years.
The investigators employed a 5-year lag time in their study to exclude head injuries caused by early motor dysfunction, they noted. “We wanted to look at people who had these head injuries we think might be part of the cause of Parkinson’s disease as opposed to a result of them,” Dr. Brown said.
In this head injury group, 51% had received one head injury, 28% had received two injuries, and 22% had received more than two injuries.
The study also included 1,457 participants (56% women; mean age, 65 years) with Parkinson’s disease who had not had a head injury prior to their diagnosis. Of these patients, the mean time with a Parkinson’s disease diagnosis was 8 years.
Dr. Brown noted that the age and sex distribution of the study group was “probably representative” of the general Parkinson’s disease population. However, because the participants had to be able to go online and complete questionnaires, it is unlikely that, among these patients, Parkinson’s disease was far advanced, he said.
The investigators adjusted for age, sex, years of education, and Parkinson’s disease duration.
Two-hit hypothesis?
The researchers compared time from diagnosis to the development of significant motor impairment, such as the need for assistance with walking, and cognitive impairment, such as having a score of less than 43 on the Penn Daily Activities Questionnaire.
They also examined the role of more severe head injuries. In the head injury group, over half (54%) had had a severe head injury, including 543 who had lost consciousness and others who had suffered a fracture or had had a seizure.
Results showed that the adjusted hazard ratio for developing motor impairment among those with a head injury, compared with those who had not had a head injury was 1.24 (95% confidence interval, 1.01-1.53; P = .037). For severe injuries, the aHR for motor impairment was 1.44 (95% CI, 1.13-1.83; P = .003).
For cognitive impairment, the aHR for those with versus without head injuries was 1.45 (95% CI, 1.14-1.86; P = .003); and for severe injuries, the aHR was 1.49 (95% CI, 1.11-2.0; P = .008).
Aside from severity, the researchers did not examine subgroups. However, Dr. Brown reported that his team would like to stratify results by sex and other variables in the future.
He noted that various mechanisms may explain why Parkinson’s disease progression is faster for patients who have a history of head injury, compared with others. Chronic inflammation due to the injury and “co-pathology” might play some role, he said. He noted that head injuries are associated with cognitive impairment in other conditions, including Alzheimer’s disease.
There is also the “two hit” hypothesis, Dr. Brown said. “A head injury could cause such broad damage that once people develop Parkinson’s disease, it’s harder for them to compensate.”
Dr. Brown also noted there might have been a “higher magnitude” of a difference between groups had the study captured participants with more severe symptoms.
‘Provocative’ findings
Michael S. Okun, MD, medical advisor at the Parkinson’s Foundation and professor and director at the Norman Fixel Institute for Neurological Diseases, University of Florida, Gainesville, said the new data are “provocative.”
“The idea that a head injury may be important in predicting how quickly and how severely deficits will manifest could be important to the treating clinician,” said Dr. Okun, who was not involved with the research.
He noted that the results suggest clinicians should elicit more information from patients about head trauma. “They should be seeking more than a binary ‘yes or no’ answer to head injury when questioning patients,” he added.
Dr. Okun reiterated that head injury is a “known and important risk factor” not only for Parkinson’s disease but also for other neurodegenerative diseases. “It’s important to counsel patients about the association,” he said.
The study was supported by the Michael J. Fox Foundation. Dr. Brown reports having received grant support from the Michael J. Fox Foundation. Dr. Okun has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From MDS 2022