User login
Local hospitals still have a role in treating severe stroke
a new study has shown.
In the RACECAT trial, functional outcomes were similar for patients suspected of having a large-vessel occlusion stroke who were located in areas not currently served by a comprehensive stroke center, whether they were first taken to a local primary stroke center or whether they were transported over a longer distance to a comprehensive center.
“Under the particular conditions in our study where we had a very well-organized system, a ‘mothership’ transfer protocol for patients with suspected large-vessel occlusion has not proven superior over the ‘drip-and-ship’ protocol, but the opposite is also true,” lead investigator Marc Ribo, MD, concluded.
Dr. Ribo, assistant professor of neurology at Hospital Vall d’Hebron, Barcelona, presented the RACECAT results at the European Stroke Organisation–World Stroke Organisation (ESO-WSO) Conference 2020.
Dr. Ribo said in an interview that there is a feeling among the stroke community that patients with a suspected large-vessel occlusion should be transferred directly to a comprehensive stroke center capable of performing endovascular thrombectomy, even if there is a nearer, smaller primary stroke center where patients are usually taken first for thrombolysis.
“Many stroke neurologists believe we are losing time by sending patients with severe stroke to a local hospital and that we should skip this step, but this is controversial area,” he commented. “Our findings suggest that we should not automatically bypass local stroke centers.”
Dr. Ribo pointed out that the local centers performed very well in the study, with very fast “in/out” times for patients who were subsequently transferred for thrombectomy.
“On the basis of our results, we recommend that if a local stroke center can perform well like ours did – if they are within the time indicators for treating and transferring patients – then they should keep receiving these patients. But if they are not performing well in this regard, then they should probably be bypassed,” he commented.
The RACECAT trial was well received by stroke experts at an ESO-WSO 2020 press conference at which it was discussed.
Stefan Kiechl, MD, Medical University Innsbruck (Austria), described the trial as “outstanding,” adding: “It has addressed a very important question. It is a big achievement in stroke medicine.”
Patrik Michel, MD, Lausanne (Switzerland) University Hospital, said that “this is a very important and highly sophisticated trial in terms of design and execution. The message is that it doesn’t matter which pathway is used, but it is important to have a well-organized network with highly trained paramedics.”
RACECAT
The RACECAT trial was conducted in the Catalonia region of Spain. Twenty-seven hospitals participated, including 7 comprehensive stroke centers and 20 local stroke centers.
The trial included stroke patients with suspected large-vessel occlusion stroke, as determined on the basis of evaluation by paramedics using the criteria of a Rapid Arterial Occlusion Evaluation (RACE) scale score above 4 and on the basis of a call to a vascular neurologist. For inclusion in the study, patients had to be in a geographical area not served by a comprehensive stroke center and to have an estimated arrival time to a comprehensive center of less than 7 hours from symptom onset in order that thrombectomy would be possible.
Of 7,475 stroke code patients evaluated, 1,401 met the inclusion criteria and were randomly assigned to be transferred to a local hospital or to a comprehensive stroke center farther away.
Baseline characteristics were similar between the two groups. The patients had severe strokes with an average National Institutes of Health Stroke Scale score of 17. It was later confirmed that 46% of the patients enrolled in the study had a large-vessel occlusion stroke.
Results showed that time from symptom onset to hospital arrival was 142 minutes for those taken to a local center and 216 minutes for those taken to a comprehensive stroke center. Of those taken to a local hospital, 86% arrived within 4 hours of symptom onset and so were potential candidates for thrombolysis, compared with 76% of those taken to a comprehensive center.
Of the patients taken to a local hospital, 60% were given thrombolysis versus 43% of those taken immediately to a comprehensive center. On the other hand, 50% of patients who were taken directly to a comprehensive center underwent thrombectomy, compared with 40% who were first taken to a local center.
For patients who received thrombolysis, time to tissue plasminogen activator administration was 120 minutes for those treated at a local hospital versus 155 minutes for those taken directly to a comprehensive center.
For patients who received thrombectomy, time from symptom onset to groin puncture was 270 minutes if they were first taken to a local hospital and were then transferred, versus 214 minutes for those taken directly to the comprehensive center.
The primary efficacy endpoint was functional outcome using Modified Rankin Scale (mRS) shift analysis at 90 days for ischemic stroke patients. This showed a “completely flat” result, Dr. Ribo reported, with an adjusted hazard ratio of 1.029 for patients taken to a comprehensive center in comparison with those taken to a local center.
“There was absolutely no trend towards benefit in one group over the other,” he said.
What about hemorrhagic stroke?
The study also evaluated functional outcomes for the whole population enrolled. “If we make the decision just based on thrombectomy-eligible patients, we may harm the rest of the patients, so we did this study to look at the whole population of severe stroke patients,” Dr. Ribo said.
Of the study population, 25% of patients were found to have had a hemorrhagic stroke.
“The problem is, at the prehospital level, it is impossible to know if a patient is having a large-vessel occlusion ischemic stroke or a hemorrhagic stroke,” Dr. Ribo explained. “We have to make a decision for the whole population, and while a longer transport time to get to a comprehensive stroke center might help a patient with a large-vessel occlusion ischemic stroke, it might not be so appropriate for patients with a hemorrhagic stroke who need to have their blood pressure stabilized as soon as possible.”
For the whole population, the mRS shift analysis at 90 days was also neutral, with an aHR of 0.965.
When considering only patients with hemorrhagic stroke, the adjusted hazard ratio for the mRS shift analysis at 90 days was 1.216, which was still nonsignificant (95% confidence interval, 0.864-1.709). This included a nonsignificant increase in mortality among those taken directly to a comprehensive center.
“If we had better tools for a certain diagnosis in the field, then we could consider taking large-vessel occlusion ischemic stroke patients to a comprehensive center and hemorrhagic stroke patients to the local stroke center, but so far, we don’t have this option apart from a few places using mobile stroke units with CT scanners,” Dr. Ribo noted.
Transfer times to comprehensive centers in the study ranged from 30 minutes to 2.5 hours. “There might well be a difference in outcomes for short and long transfers, and we may be able to offer different transfer protocols in these different situations, and we are looking at that, but the study was only stopped in June, and we haven’t had a chance to analyze those results yet,” Dr. Ribo added.
Complications during transport occurred in 0.5% of those taken to a local hospital and in 1% of those taken directly to a comprehensive center. “We were concerned about complications with longer transfers, but these numbers are quite low. Intubations were very low – just one patient taken to a local center, versus three or four in the longer transfer group,” he added.
For both local and comprehensive centers, treatment times were impressive in the study. For local hospitals, the average in/out time was just 60 minutes for patients who went to a comprehensive center; for patients receiving thrombolysis, the average door to needle time was around 30 minutes.
Time to thrombectomy in the comprehensive center for patients transferred from a local hospital was also very fast, with an average door to groin puncture time of less than 40 minutes. “This shows we have a very well-oiled system,” Dr. Ribo said.
“There is always going to be a balance between a quicker time to thrombolysis by taking a patient to the closest hospital but a quicker time to thrombectomy if patients are taken straight to the comprehensive center,” he concluded. “But in our system, where we are achieving fast treatment and transfer times, our results show that patients had timely access to reperfusion therapies regardless of transfer protocol, and under these circumstances, it is fine for the emergency services to take stroke patients to the closest stroke center.”
Results applicable elsewhere?
During the discussion at an ESO-WSO 2020 press conference, other experts pointed out that the Catalonia group is a leader in this field, being the pioneers of the RACE score used in this study for paramedics to identify suspected large-vessel occlusions. This led to questions about the applicability of the results.
“The performance by paramedics was very good using the RACE scale, and the performance times were very impressive. Are these results applicable elsewhere?” Dr. Kiechl asked.
Dr. Ribo said the combination of the RACE score and a call with a vascular neurologist was of “great value” in identifying appropriate patients. Half of the patients selected in this way for the trial were confirmed to have a large-vessel occlusion. “That is a good result,” he added.
He noted that the performance of the local hospitals improved dramatically during the study. “They had an incentive to work on their times. They could have lost most of their stroke patients if their results came out worse. We told them they had an opportunity to show that they have a role in treating these patients, and they took that opportunity.”
Dr. Ribo said there were lessons here for those involved in acute stroke care. “When creating stroke transfer policies in local networks, the performances of individual centers need to be taken into account. If primary stroke centers are motivated and can work in a well-coordinated way and perform to within the recommended times, then they can keep receiving stroke code patients. This should be possible in most developed countries.”
Noting that the in/out time of 60 minutes at local hospitals was “very impressive,” Dr. Kiechl asked how such fast times were achieved.
Dr. Ribo responded that, to a great extent, this was because of ambulance staff. “We have trained the paramedics to anticipate a second transfer after delivering the patient to the local hospital so they can prepare for this rather than waiting for a second call.”
Dr. Ribo pointed out that there were other advantages in taking patients to local centers first. “For those that do not need to be transferred on, they will be closer to relatives. It is very difficult for the family if the patient is hundreds of miles away. And there may be a cost advantage. We did look at costs, but haven’t got that data yet.”
He said: “If local stroke centers do not treat so many stroke code patients, they will lose their expertise, and that will be detrimental to the remaining patients who are taken there. We want to try to maintain a good standard of stroke care across a decent spread of hospitals—not just a couple of major comprehensive centers,” he added.
Commenting on the study, Jesse Dawson, MD, University of Glasgow, who was chair of the plenary session at which the study was presented, said: “RACECAT is very interesting but needs a lot of thought to dissect. My takeaway is that we know that time to reperfusion is key, and we need to get these times as low as possible, but we don’t need to chase a particular care pathway. Thus, if your country/geography suits ‘drip and ship’ better, this is acceptable. If direct to endovascular is possible or you are close to such a center, then this is ideal. But within those paradigms, be as fast as possible.”
He added that results of the subgroups with regard to transfer time will be helpful.
The RACECAT study was funded by Fundacio Ictus Malaltia Vascular.
A version of this article originally appeared on Medscape.com.
a new study has shown.
In the RACECAT trial, functional outcomes were similar for patients suspected of having a large-vessel occlusion stroke who were located in areas not currently served by a comprehensive stroke center, whether they were first taken to a local primary stroke center or whether they were transported over a longer distance to a comprehensive center.
“Under the particular conditions in our study where we had a very well-organized system, a ‘mothership’ transfer protocol for patients with suspected large-vessel occlusion has not proven superior over the ‘drip-and-ship’ protocol, but the opposite is also true,” lead investigator Marc Ribo, MD, concluded.
Dr. Ribo, assistant professor of neurology at Hospital Vall d’Hebron, Barcelona, presented the RACECAT results at the European Stroke Organisation–World Stroke Organisation (ESO-WSO) Conference 2020.
Dr. Ribo said in an interview that there is a feeling among the stroke community that patients with a suspected large-vessel occlusion should be transferred directly to a comprehensive stroke center capable of performing endovascular thrombectomy, even if there is a nearer, smaller primary stroke center where patients are usually taken first for thrombolysis.
“Many stroke neurologists believe we are losing time by sending patients with severe stroke to a local hospital and that we should skip this step, but this is controversial area,” he commented. “Our findings suggest that we should not automatically bypass local stroke centers.”
Dr. Ribo pointed out that the local centers performed very well in the study, with very fast “in/out” times for patients who were subsequently transferred for thrombectomy.
“On the basis of our results, we recommend that if a local stroke center can perform well like ours did – if they are within the time indicators for treating and transferring patients – then they should keep receiving these patients. But if they are not performing well in this regard, then they should probably be bypassed,” he commented.
The RACECAT trial was well received by stroke experts at an ESO-WSO 2020 press conference at which it was discussed.
Stefan Kiechl, MD, Medical University Innsbruck (Austria), described the trial as “outstanding,” adding: “It has addressed a very important question. It is a big achievement in stroke medicine.”
Patrik Michel, MD, Lausanne (Switzerland) University Hospital, said that “this is a very important and highly sophisticated trial in terms of design and execution. The message is that it doesn’t matter which pathway is used, but it is important to have a well-organized network with highly trained paramedics.”
RACECAT
The RACECAT trial was conducted in the Catalonia region of Spain. Twenty-seven hospitals participated, including 7 comprehensive stroke centers and 20 local stroke centers.
The trial included stroke patients with suspected large-vessel occlusion stroke, as determined on the basis of evaluation by paramedics using the criteria of a Rapid Arterial Occlusion Evaluation (RACE) scale score above 4 and on the basis of a call to a vascular neurologist. For inclusion in the study, patients had to be in a geographical area not served by a comprehensive stroke center and to have an estimated arrival time to a comprehensive center of less than 7 hours from symptom onset in order that thrombectomy would be possible.
Of 7,475 stroke code patients evaluated, 1,401 met the inclusion criteria and were randomly assigned to be transferred to a local hospital or to a comprehensive stroke center farther away.
Baseline characteristics were similar between the two groups. The patients had severe strokes with an average National Institutes of Health Stroke Scale score of 17. It was later confirmed that 46% of the patients enrolled in the study had a large-vessel occlusion stroke.
Results showed that time from symptom onset to hospital arrival was 142 minutes for those taken to a local center and 216 minutes for those taken to a comprehensive stroke center. Of those taken to a local hospital, 86% arrived within 4 hours of symptom onset and so were potential candidates for thrombolysis, compared with 76% of those taken to a comprehensive center.
Of the patients taken to a local hospital, 60% were given thrombolysis versus 43% of those taken immediately to a comprehensive center. On the other hand, 50% of patients who were taken directly to a comprehensive center underwent thrombectomy, compared with 40% who were first taken to a local center.
For patients who received thrombolysis, time to tissue plasminogen activator administration was 120 minutes for those treated at a local hospital versus 155 minutes for those taken directly to a comprehensive center.
For patients who received thrombectomy, time from symptom onset to groin puncture was 270 minutes if they were first taken to a local hospital and were then transferred, versus 214 minutes for those taken directly to the comprehensive center.
The primary efficacy endpoint was functional outcome using Modified Rankin Scale (mRS) shift analysis at 90 days for ischemic stroke patients. This showed a “completely flat” result, Dr. Ribo reported, with an adjusted hazard ratio of 1.029 for patients taken to a comprehensive center in comparison with those taken to a local center.
“There was absolutely no trend towards benefit in one group over the other,” he said.
What about hemorrhagic stroke?
The study also evaluated functional outcomes for the whole population enrolled. “If we make the decision just based on thrombectomy-eligible patients, we may harm the rest of the patients, so we did this study to look at the whole population of severe stroke patients,” Dr. Ribo said.
Of the study population, 25% of patients were found to have had a hemorrhagic stroke.
“The problem is, at the prehospital level, it is impossible to know if a patient is having a large-vessel occlusion ischemic stroke or a hemorrhagic stroke,” Dr. Ribo explained. “We have to make a decision for the whole population, and while a longer transport time to get to a comprehensive stroke center might help a patient with a large-vessel occlusion ischemic stroke, it might not be so appropriate for patients with a hemorrhagic stroke who need to have their blood pressure stabilized as soon as possible.”
For the whole population, the mRS shift analysis at 90 days was also neutral, with an aHR of 0.965.
When considering only patients with hemorrhagic stroke, the adjusted hazard ratio for the mRS shift analysis at 90 days was 1.216, which was still nonsignificant (95% confidence interval, 0.864-1.709). This included a nonsignificant increase in mortality among those taken directly to a comprehensive center.
“If we had better tools for a certain diagnosis in the field, then we could consider taking large-vessel occlusion ischemic stroke patients to a comprehensive center and hemorrhagic stroke patients to the local stroke center, but so far, we don’t have this option apart from a few places using mobile stroke units with CT scanners,” Dr. Ribo noted.
Transfer times to comprehensive centers in the study ranged from 30 minutes to 2.5 hours. “There might well be a difference in outcomes for short and long transfers, and we may be able to offer different transfer protocols in these different situations, and we are looking at that, but the study was only stopped in June, and we haven’t had a chance to analyze those results yet,” Dr. Ribo added.
Complications during transport occurred in 0.5% of those taken to a local hospital and in 1% of those taken directly to a comprehensive center. “We were concerned about complications with longer transfers, but these numbers are quite low. Intubations were very low – just one patient taken to a local center, versus three or four in the longer transfer group,” he added.
For both local and comprehensive centers, treatment times were impressive in the study. For local hospitals, the average in/out time was just 60 minutes for patients who went to a comprehensive center; for patients receiving thrombolysis, the average door to needle time was around 30 minutes.
Time to thrombectomy in the comprehensive center for patients transferred from a local hospital was also very fast, with an average door to groin puncture time of less than 40 minutes. “This shows we have a very well-oiled system,” Dr. Ribo said.
“There is always going to be a balance between a quicker time to thrombolysis by taking a patient to the closest hospital but a quicker time to thrombectomy if patients are taken straight to the comprehensive center,” he concluded. “But in our system, where we are achieving fast treatment and transfer times, our results show that patients had timely access to reperfusion therapies regardless of transfer protocol, and under these circumstances, it is fine for the emergency services to take stroke patients to the closest stroke center.”
Results applicable elsewhere?
During the discussion at an ESO-WSO 2020 press conference, other experts pointed out that the Catalonia group is a leader in this field, being the pioneers of the RACE score used in this study for paramedics to identify suspected large-vessel occlusions. This led to questions about the applicability of the results.
“The performance by paramedics was very good using the RACE scale, and the performance times were very impressive. Are these results applicable elsewhere?” Dr. Kiechl asked.
Dr. Ribo said the combination of the RACE score and a call with a vascular neurologist was of “great value” in identifying appropriate patients. Half of the patients selected in this way for the trial were confirmed to have a large-vessel occlusion. “That is a good result,” he added.
He noted that the performance of the local hospitals improved dramatically during the study. “They had an incentive to work on their times. They could have lost most of their stroke patients if their results came out worse. We told them they had an opportunity to show that they have a role in treating these patients, and they took that opportunity.”
Dr. Ribo said there were lessons here for those involved in acute stroke care. “When creating stroke transfer policies in local networks, the performances of individual centers need to be taken into account. If primary stroke centers are motivated and can work in a well-coordinated way and perform to within the recommended times, then they can keep receiving stroke code patients. This should be possible in most developed countries.”
Noting that the in/out time of 60 minutes at local hospitals was “very impressive,” Dr. Kiechl asked how such fast times were achieved.
Dr. Ribo responded that, to a great extent, this was because of ambulance staff. “We have trained the paramedics to anticipate a second transfer after delivering the patient to the local hospital so they can prepare for this rather than waiting for a second call.”
Dr. Ribo pointed out that there were other advantages in taking patients to local centers first. “For those that do not need to be transferred on, they will be closer to relatives. It is very difficult for the family if the patient is hundreds of miles away. And there may be a cost advantage. We did look at costs, but haven’t got that data yet.”
He said: “If local stroke centers do not treat so many stroke code patients, they will lose their expertise, and that will be detrimental to the remaining patients who are taken there. We want to try to maintain a good standard of stroke care across a decent spread of hospitals—not just a couple of major comprehensive centers,” he added.
Commenting on the study, Jesse Dawson, MD, University of Glasgow, who was chair of the plenary session at which the study was presented, said: “RACECAT is very interesting but needs a lot of thought to dissect. My takeaway is that we know that time to reperfusion is key, and we need to get these times as low as possible, but we don’t need to chase a particular care pathway. Thus, if your country/geography suits ‘drip and ship’ better, this is acceptable. If direct to endovascular is possible or you are close to such a center, then this is ideal. But within those paradigms, be as fast as possible.”
He added that results of the subgroups with regard to transfer time will be helpful.
The RACECAT study was funded by Fundacio Ictus Malaltia Vascular.
A version of this article originally appeared on Medscape.com.
a new study has shown.
In the RACECAT trial, functional outcomes were similar for patients suspected of having a large-vessel occlusion stroke who were located in areas not currently served by a comprehensive stroke center, whether they were first taken to a local primary stroke center or whether they were transported over a longer distance to a comprehensive center.
“Under the particular conditions in our study where we had a very well-organized system, a ‘mothership’ transfer protocol for patients with suspected large-vessel occlusion has not proven superior over the ‘drip-and-ship’ protocol, but the opposite is also true,” lead investigator Marc Ribo, MD, concluded.
Dr. Ribo, assistant professor of neurology at Hospital Vall d’Hebron, Barcelona, presented the RACECAT results at the European Stroke Organisation–World Stroke Organisation (ESO-WSO) Conference 2020.
Dr. Ribo said in an interview that there is a feeling among the stroke community that patients with a suspected large-vessel occlusion should be transferred directly to a comprehensive stroke center capable of performing endovascular thrombectomy, even if there is a nearer, smaller primary stroke center where patients are usually taken first for thrombolysis.
“Many stroke neurologists believe we are losing time by sending patients with severe stroke to a local hospital and that we should skip this step, but this is controversial area,” he commented. “Our findings suggest that we should not automatically bypass local stroke centers.”
Dr. Ribo pointed out that the local centers performed very well in the study, with very fast “in/out” times for patients who were subsequently transferred for thrombectomy.
“On the basis of our results, we recommend that if a local stroke center can perform well like ours did – if they are within the time indicators for treating and transferring patients – then they should keep receiving these patients. But if they are not performing well in this regard, then they should probably be bypassed,” he commented.
The RACECAT trial was well received by stroke experts at an ESO-WSO 2020 press conference at which it was discussed.
Stefan Kiechl, MD, Medical University Innsbruck (Austria), described the trial as “outstanding,” adding: “It has addressed a very important question. It is a big achievement in stroke medicine.”
Patrik Michel, MD, Lausanne (Switzerland) University Hospital, said that “this is a very important and highly sophisticated trial in terms of design and execution. The message is that it doesn’t matter which pathway is used, but it is important to have a well-organized network with highly trained paramedics.”
RACECAT
The RACECAT trial was conducted in the Catalonia region of Spain. Twenty-seven hospitals participated, including 7 comprehensive stroke centers and 20 local stroke centers.
The trial included stroke patients with suspected large-vessel occlusion stroke, as determined on the basis of evaluation by paramedics using the criteria of a Rapid Arterial Occlusion Evaluation (RACE) scale score above 4 and on the basis of a call to a vascular neurologist. For inclusion in the study, patients had to be in a geographical area not served by a comprehensive stroke center and to have an estimated arrival time to a comprehensive center of less than 7 hours from symptom onset in order that thrombectomy would be possible.
Of 7,475 stroke code patients evaluated, 1,401 met the inclusion criteria and were randomly assigned to be transferred to a local hospital or to a comprehensive stroke center farther away.
Baseline characteristics were similar between the two groups. The patients had severe strokes with an average National Institutes of Health Stroke Scale score of 17. It was later confirmed that 46% of the patients enrolled in the study had a large-vessel occlusion stroke.
Results showed that time from symptom onset to hospital arrival was 142 minutes for those taken to a local center and 216 minutes for those taken to a comprehensive stroke center. Of those taken to a local hospital, 86% arrived within 4 hours of symptom onset and so were potential candidates for thrombolysis, compared with 76% of those taken to a comprehensive center.
Of the patients taken to a local hospital, 60% were given thrombolysis versus 43% of those taken immediately to a comprehensive center. On the other hand, 50% of patients who were taken directly to a comprehensive center underwent thrombectomy, compared with 40% who were first taken to a local center.
For patients who received thrombolysis, time to tissue plasminogen activator administration was 120 minutes for those treated at a local hospital versus 155 minutes for those taken directly to a comprehensive center.
For patients who received thrombectomy, time from symptom onset to groin puncture was 270 minutes if they were first taken to a local hospital and were then transferred, versus 214 minutes for those taken directly to the comprehensive center.
The primary efficacy endpoint was functional outcome using Modified Rankin Scale (mRS) shift analysis at 90 days for ischemic stroke patients. This showed a “completely flat” result, Dr. Ribo reported, with an adjusted hazard ratio of 1.029 for patients taken to a comprehensive center in comparison with those taken to a local center.
“There was absolutely no trend towards benefit in one group over the other,” he said.
What about hemorrhagic stroke?
The study also evaluated functional outcomes for the whole population enrolled. “If we make the decision just based on thrombectomy-eligible patients, we may harm the rest of the patients, so we did this study to look at the whole population of severe stroke patients,” Dr. Ribo said.
Of the study population, 25% of patients were found to have had a hemorrhagic stroke.
“The problem is, at the prehospital level, it is impossible to know if a patient is having a large-vessel occlusion ischemic stroke or a hemorrhagic stroke,” Dr. Ribo explained. “We have to make a decision for the whole population, and while a longer transport time to get to a comprehensive stroke center might help a patient with a large-vessel occlusion ischemic stroke, it might not be so appropriate for patients with a hemorrhagic stroke who need to have their blood pressure stabilized as soon as possible.”
For the whole population, the mRS shift analysis at 90 days was also neutral, with an aHR of 0.965.
When considering only patients with hemorrhagic stroke, the adjusted hazard ratio for the mRS shift analysis at 90 days was 1.216, which was still nonsignificant (95% confidence interval, 0.864-1.709). This included a nonsignificant increase in mortality among those taken directly to a comprehensive center.
“If we had better tools for a certain diagnosis in the field, then we could consider taking large-vessel occlusion ischemic stroke patients to a comprehensive center and hemorrhagic stroke patients to the local stroke center, but so far, we don’t have this option apart from a few places using mobile stroke units with CT scanners,” Dr. Ribo noted.
Transfer times to comprehensive centers in the study ranged from 30 minutes to 2.5 hours. “There might well be a difference in outcomes for short and long transfers, and we may be able to offer different transfer protocols in these different situations, and we are looking at that, but the study was only stopped in June, and we haven’t had a chance to analyze those results yet,” Dr. Ribo added.
Complications during transport occurred in 0.5% of those taken to a local hospital and in 1% of those taken directly to a comprehensive center. “We were concerned about complications with longer transfers, but these numbers are quite low. Intubations were very low – just one patient taken to a local center, versus three or four in the longer transfer group,” he added.
For both local and comprehensive centers, treatment times were impressive in the study. For local hospitals, the average in/out time was just 60 minutes for patients who went to a comprehensive center; for patients receiving thrombolysis, the average door to needle time was around 30 minutes.
Time to thrombectomy in the comprehensive center for patients transferred from a local hospital was also very fast, with an average door to groin puncture time of less than 40 minutes. “This shows we have a very well-oiled system,” Dr. Ribo said.
“There is always going to be a balance between a quicker time to thrombolysis by taking a patient to the closest hospital but a quicker time to thrombectomy if patients are taken straight to the comprehensive center,” he concluded. “But in our system, where we are achieving fast treatment and transfer times, our results show that patients had timely access to reperfusion therapies regardless of transfer protocol, and under these circumstances, it is fine for the emergency services to take stroke patients to the closest stroke center.”
Results applicable elsewhere?
During the discussion at an ESO-WSO 2020 press conference, other experts pointed out that the Catalonia group is a leader in this field, being the pioneers of the RACE score used in this study for paramedics to identify suspected large-vessel occlusions. This led to questions about the applicability of the results.
“The performance by paramedics was very good using the RACE scale, and the performance times were very impressive. Are these results applicable elsewhere?” Dr. Kiechl asked.
Dr. Ribo said the combination of the RACE score and a call with a vascular neurologist was of “great value” in identifying appropriate patients. Half of the patients selected in this way for the trial were confirmed to have a large-vessel occlusion. “That is a good result,” he added.
He noted that the performance of the local hospitals improved dramatically during the study. “They had an incentive to work on their times. They could have lost most of their stroke patients if their results came out worse. We told them they had an opportunity to show that they have a role in treating these patients, and they took that opportunity.”
Dr. Ribo said there were lessons here for those involved in acute stroke care. “When creating stroke transfer policies in local networks, the performances of individual centers need to be taken into account. If primary stroke centers are motivated and can work in a well-coordinated way and perform to within the recommended times, then they can keep receiving stroke code patients. This should be possible in most developed countries.”
Noting that the in/out time of 60 minutes at local hospitals was “very impressive,” Dr. Kiechl asked how such fast times were achieved.
Dr. Ribo responded that, to a great extent, this was because of ambulance staff. “We have trained the paramedics to anticipate a second transfer after delivering the patient to the local hospital so they can prepare for this rather than waiting for a second call.”
Dr. Ribo pointed out that there were other advantages in taking patients to local centers first. “For those that do not need to be transferred on, they will be closer to relatives. It is very difficult for the family if the patient is hundreds of miles away. And there may be a cost advantage. We did look at costs, but haven’t got that data yet.”
He said: “If local stroke centers do not treat so many stroke code patients, they will lose their expertise, and that will be detrimental to the remaining patients who are taken there. We want to try to maintain a good standard of stroke care across a decent spread of hospitals—not just a couple of major comprehensive centers,” he added.
Commenting on the study, Jesse Dawson, MD, University of Glasgow, who was chair of the plenary session at which the study was presented, said: “RACECAT is very interesting but needs a lot of thought to dissect. My takeaway is that we know that time to reperfusion is key, and we need to get these times as low as possible, but we don’t need to chase a particular care pathway. Thus, if your country/geography suits ‘drip and ship’ better, this is acceptable. If direct to endovascular is possible or you are close to such a center, then this is ideal. But within those paradigms, be as fast as possible.”
He added that results of the subgroups with regard to transfer time will be helpful.
The RACECAT study was funded by Fundacio Ictus Malaltia Vascular.
A version of this article originally appeared on Medscape.com.
FROM ESO-WSO 2020
What imaging can disclose about suspected stroke and its Tx
Stroke ranks second behind heart disease as the leading cause of mortality worldwide, accounting for 1 of every 19 deaths,1 and remains a serious cause of morbidity. Best practices in stroke diagnosis and management can seem elusive to front-line clinicians, for 2 reasons: the rate of proliferation and nuance in stroke medicine and the fact that the typical scope of primary care practice exists apart from much of the diagnostic tools and management schema provided in stroke centers.2 In this article, we describe and update the diagnosis of stroke and review imaging modalities, their nuances, and their application in practice.
Diagnosis of acute stroke
Acute stroke is diagnosed upon observation of new neurologic deficits and congruent neuroimaging. Some updated definitions favor a silent form of cerebral ischemia manifested by imaging pathology only; this form is not discussed in this article. Although there are several characteristically distinct stroke syndromes, there is no way to clinically distinguish ischemic pathology from hemorrhagic pathology.
Some common symptoms that should prompt evaluation for stroke are part of the American Stroke Association FAST mnemonic designed to promote public health awareness3-5:
f ace droopinga rm weaknesss peech difficultyt ime to call 911.
Other commonly reported stroke symptoms include unilateral weakness or numbness, confusion, word-finding difficulty, visual problems, difficulty ambulating, dizziness, loss of balance or coordination, and thunderclap headache. A stroke should also be considered in the presence of any new focal neurologic deficit.3,4
Stroke patients should be triaged by emergency medical services using a stroke screening scale, such as BE-FAST5 (a modification of FAST that adds balance and eye assessments); the Los Angeles Prehospital Stroke Screen (LAPSS)6,7; the Rapid Arterial oCclusion Evaluation (RACE)8; and the Cincinnati Prehospital Stroke Severity Scale (CP-SSS)9,10 (see “Stroke screening scales for early identification and triage"). Studies have not found that any single prehospital stroke scale is superior to the others for reliably predicting large-vessel occlusion; therefore, prehospital assessment is typically based on practice patterns in a given locale.11 A patient (or family member or caregiver) who seeks your care for stroke symptoms should be told to call 911 and get emergency transport to a health care facility that can capably administer intravenous (IV) thrombolysis.a
SIDEBAR
Stroke screening scales for early identification and triage
National Institutes of Health Stroke Scale
www.stroke.nih.gov/resources/scale.htm
FAST
www.stroke.org/en/help-and-support/resource-library/fast-materials
BE-FAST
www.ahajournals.org/doi/10.1161/STROKEAHA.116.015169
Los Angeles Prehospital Stroke Screen (LAPSS)
http://stroke.ucla.edu/workfiles/prehospital-screen.pdf
Rapid Artery Occlusion Evaluation (RACE)
www.mdcalc.com/rapid-arterial-occlusion-evaluation-race-scale-stroke
Cincinnati Prehospital Stroke Severity Scale (CP-SSS)
https://www.mdcalc.com/cincinnati-prehospital-stroke-severity-scale-cp-sss
First responders should elicit “last-known-normal” time; this critical information can aid in diagnosis and drive therapeutic options, especially if patients are unaccompanied at time of transport to a higher echelon of care. A point-of-care blood glucose test should be performed by emergency medical staff, with dextrose administered for a level < 45 mg/dL. Establishing IV access for fluids, medications, and contrast can be considered if it does not delay transport. A 12-lead electrocardiogram can also be considered, again, as long as it does not delay transport to a facility capable of providing definitive therapy. Notification by emergency services staff before arrival and transport of the patient to such a facility is the essential element of prehospital care, and should be prioritized above ancillary testing beyond the stroke assessment.14
Guidelines recommend use of the National Institutes of Health Stroke Scale (NIHSS; www.stroke.nih.gov/resources/scale.htm) for clinical evaluation upon arrival at the ED.15 Although no scale has been identified that can reliably predict large-vessel occlusion amenable to endovascular therapy (EVT), no other score has been found to outperform the NIHSS in achieving meaningful patient outcomes.16 Furthermore, NIHSS has been validated to track clinical changes in response to therapy, is widely utilized, and is free.
Continue to: A criticism of the NIHSS...
A criticism of the NIHSS is its bias toward left-hemispheric ischemic pathology.17 NIHSS includes 11 questions on a scale of 0 to 42; typically, a score < 4 is associated with a higher chance of a positive clinical outcome.18 There is no minimum or maximum NIHSS score that precludes treatment with thrombolysis or EVT.
Other commonly used scores in acute stroke include disability assessments. The modified Rankin scale, which is used most often, features a score of 0 (symptom-free) to 6 (death). A modified Rankin scale score of 0 or 1 is considered an indication of a favorable outcome after stroke.19 Note that these functional scores are not always part of an acute assessment but can be done early in the clinical course to gauge the response to treatment, and are collected for stroke-center certification.
Imaging modalities
Imaging is recommended within 20 minutes of arrival in the ED in a stroke patient who might be a candidate for thrombolysis or thrombectomy.3 There, imaging modalities commonly performed are noncontrast-enhanced head computed tomography (NCHCT); computed tomography (CT) angiography, with or without perfusion; and diffusion-weighted magnetic resonance imaging (MRI).20,21 In addition, more highly specialized imaging modalities are available for the evaluation of the stroke patient in specific, often limited, circumstances. All these modalities are described below and compared in the TABLE,20,21 using the ACR Appropriateness Criteria (of the American College of Radiology),21 which are guidelines for appropriate imaging of stroke, based on a clinical complaint. Separate recommendations and appraisals are offered by the most recent American Heart Association/American Stroke Association (AHA/ASA) guideline.3
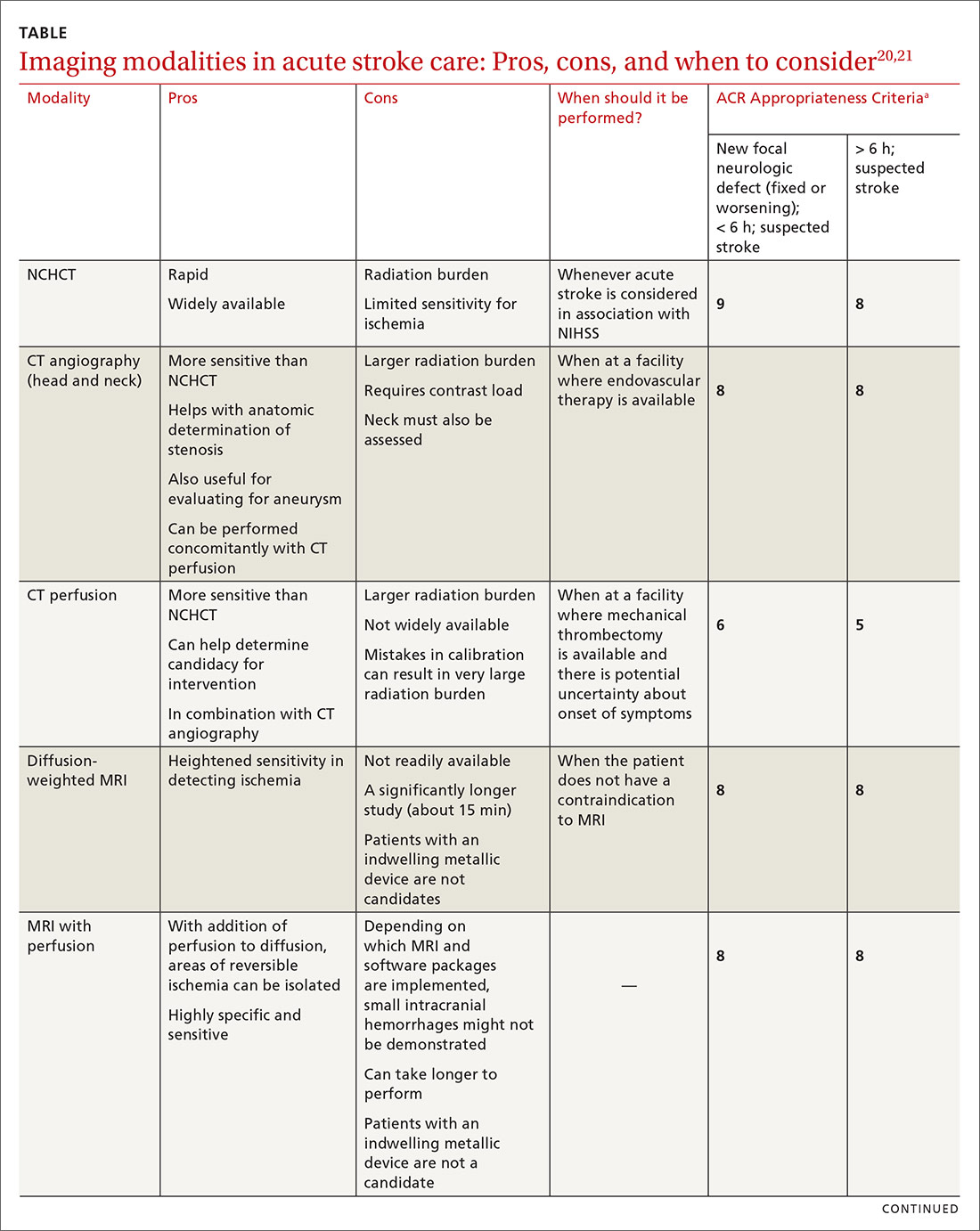
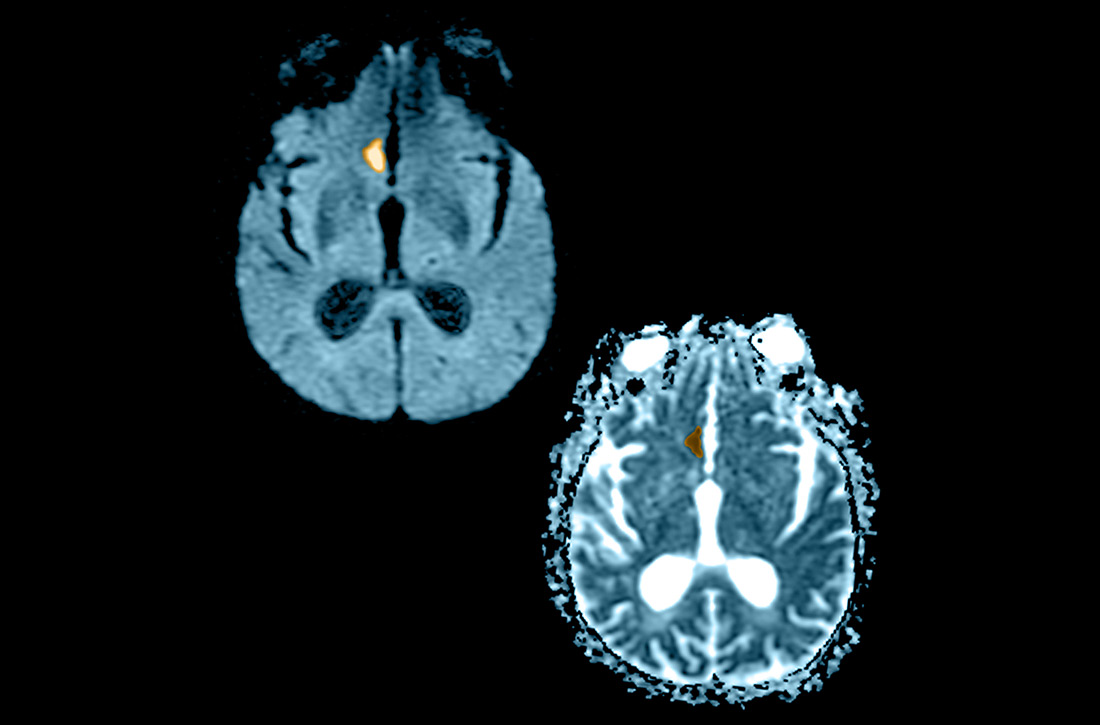
NCHCT. This study should be performed within 20 minutes after arrival at the ED because it provides rapid assessment of intracerebral hemorrhage, can effectively corroborate the diagnosis of some stroke mimickers, and identifies some candidates for EVT or thrombolysis3,21,22 (typically, the decision to proceed with EVT is based on adjunct imaging studies discussed in a bit). Evaluation for intracerebral hemorrhage is required prior to administering thrombolysis. Ischemic changes can be seen with variable specificity and sensitivity on NCHCT, depending on how much time has passed since the original insult. In all historical trials, CT was the only imaging modality used in the diagnosis of acute ischemic stroke (AIS) that suggested benefit from IV thrombolysis.23-25
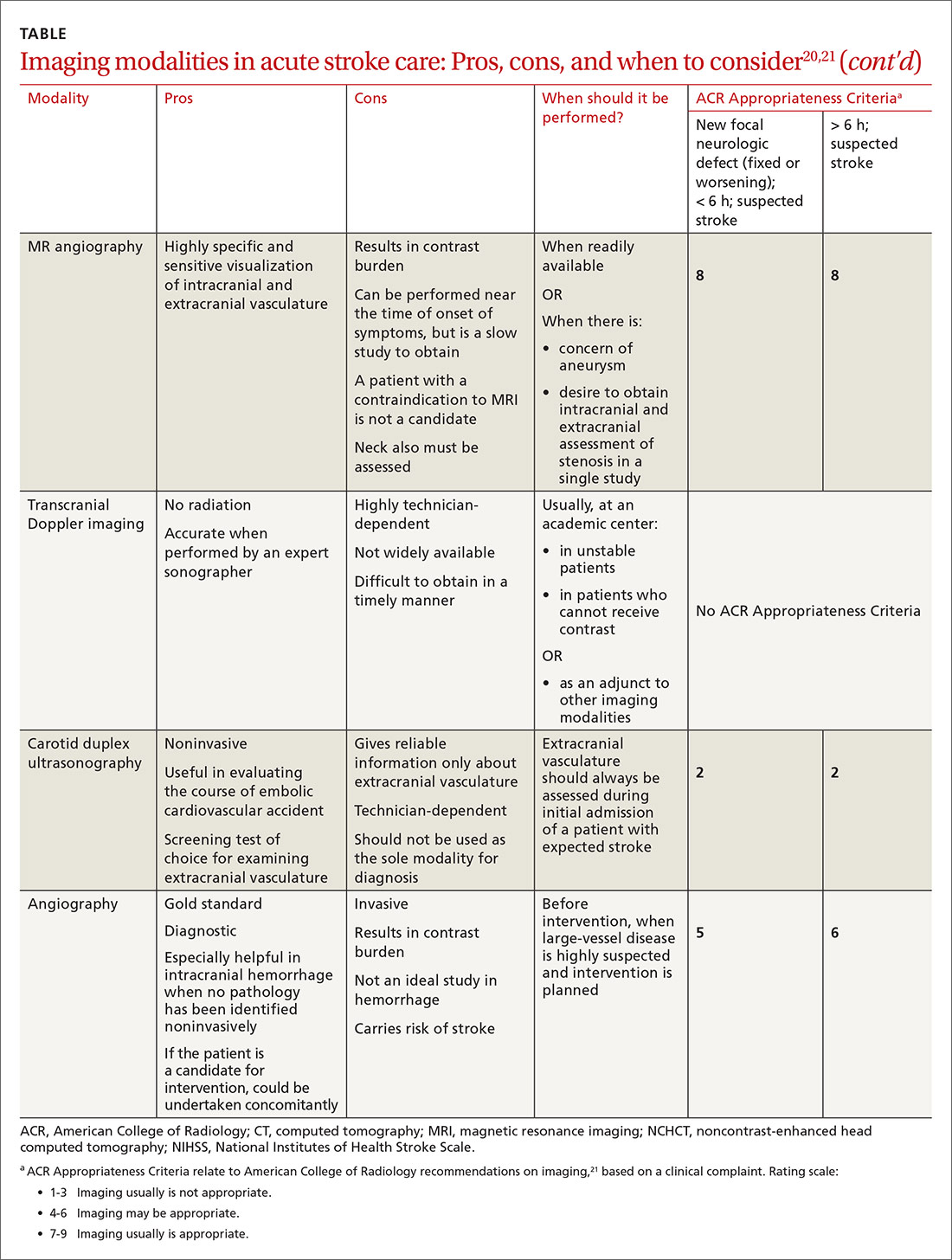
Acute, subacute, and chronic changes can be seen on NCHCT, although the modality has limited sensitivity for identifying AIS (ie, approximately 75% within 6 hours after the original insult):
- Acute findings on NCHCT include intracellular edema, which causes loss of the gray matter–white matter interface and effacement of the cortical sulci. This occurs as a result of increased cellular uptake of water in response to ischemia and cell death, resulting in a decreased density of tissue (hypoattenuation) in affected areas.
- Subacute changes appear in the 2- to 5-day window, including vasogenic edema with greater mass effect, hypoattenuation, and well-defined margins.3,20,21
- Chronic vascular findings on NCHCT include loss of brain tissue and hypoattenuation.
Continue to: NCHCT is typically performed...
NCHCT is typically performed in advance of other adjunct imaging modalities.3,20,21 Baseline NCHCT can be performed on patients with advanced kidney disease and those who have an indwelling metallic device.
CT angiography is performed with timed contrast, providing a 3-dimensional representation of the cerebral vasculature; the entire intracranial and extracranial vasculature, including the aortic arch, can be mapped in approximately 60 seconds. CT angiography is sensitive in identifying areas of stenosis > 50% and identifies clinically significant areas of stenosis up to approximately 90% of the time.26 For this reason, it is particularly helpful in identifying candidates for an interventional strategy beyond pharmacotherapeutic thrombolysis. In addition, CT angiography can visualize aneurysmal dilation and dissection, and help with the planning of interventions—specifically, the confident administration of thrombolysis or more specific planning for target lesions and EVT.
It also can help identify a host of vascular phenomena, such as arteriovenous malformations, Moyamoya disease (progressive arterial blockage within the basal ganglia and compensatory microvascularization), and some vasculopathies.20,27 In intracranial hemorrhage, CT with angiography can help evaluate for structural malformations and identify patients at risk of hematoma expansion.22
CT perfusion. Many stroke centers will perform a CT perfusion study,28 which encompasses as many as 3 different CT sequences:
- NCHCT
- vertex-to-arch angiography with contrast bolus
- administration of contrast and capture of a dynamic sequence through 1 or 2 slabs of tissue, allowing for the generation of maps of cerebral blood flow (CBF), mean transit time (MTT), and cerebral blood volume (CBV) of the entire cerebral vasculature.
The interplay of these 3 sequences drives characterization of lesions (ie, CBF = CBV/MTT). An infarct is characterized by low CBF, low CBV, and elevated MTT. In penumbral tissue, MTT is elevated but CBF is slightly decreased and CBV is normal or increased. Using CT perfusion, areas throughout the ischemic penumbra can be surveyed for favorable interventional characteristics.20,29
Continue to: A CT perfusion study adds...
A CT perfusion study adds at least 60 seconds to NCHCT. This modality can be useful in planning interventions and for stratifying appropriateness of reperfusion strategies in strokes of unknown duration.3,30 CT perfusion can be performed on any multidetector CT scan but (1) requires specialized software and expertise to interpret and (2) subjects the patient to a significant radiation dose, which, if incorrectly administered, can be considerably higher than intended.20,26,27
Diffusion-weighted MRI. This is the most sensitive study for demonstrating early ischemic changes; however, limitations include lack of availability, contraindication in patients with metallic indwelling implants, and duration of the study—although, at some stroke centers, diffusion-weighted MRI can be performed in ≤ 10 minutes.
MRI and NCHCT have comparable sensitivity in detecting intracranial hemorrhage. MRI is likely more sensitive in identifying areas of microhemorrhage: In diffusion-weighted MRI, the sensitivity of stroke detection increases to > 95%.31 The modality relies on the comparable movement of water through damaged vs normal neuronal tissue. Diffusion-weighted MRI does not require administration of concomitant contrast, which can be a benefit in patients who are allergic to gadolinium-based contrast agents or have advanced kidney disease that precludes the use of contrast. It typically does not result in adequate characterization of extracranial vasculature.
Other MRI modalities. These MRI extensions include magnetic resonance (MR) perfusion and MR angiography. Whereas diffusion-weighted MRI (discussed above) offers the most rapid and sensitive evaluation for ischemia, fluid-attenuated inversion recovery (FLAIR) imaging has been utilized as a comparator to isolated diffusion-weighted MRI to help determine stroke duration. FLAIR signal positivity typically occurs 6 to 24 hours after the initial insult but is negative in stroke that occurred < 3 hours earlier.32
MRI is limited, in terms of availability and increased study duration, especially when it comes to timely administration of thrombolysis. A benefit of this modality is less radiation and, as noted, superior sensitivity for ischemia. Diffusion-weighted MRI combined with MR perfusion analysis can help isolate areas of the ischemic penumbra. MR perfusion is performed for a similar reason as CT perfusion, although logistical execution across those modalities is significantly different. Considerations for choosing MR perfusion or CT perfusion should be made on an individual basis and based on available local resources and accepted local practice patterns.26
Continue to: In the subacute setting...
In the subacute setting, MR perfusion and MR angiography of the head and the neck are often performed to identify stenosis, dissection, and more subtle mimickers of cerebrovascular accident not ascertained on initial CT evaluation. These studies are typically performed well outside the window for thrombolysis or intervention.26 No guidelines specifically direct or recommend this practice pattern. The superior sensitivity and cerebral blood flow mapping of MR perfusion and MR angiography might be useful for validating a suspected diagnosis of ischemic stroke and providing phenotypic information about AIS events.
Transcranial Doppler imaging relies on bony windows to assess intracranial vascular flow, velocity, direction, and reactivity. This information can be utilized to diagnose stenosis or occlusion. This modality is principally used to evaluate for stenosis in the anterior circulation (sensitivity, 70%-90%; specificity, 90%-95%).20 Evaluation of the basilar, vertebral, and internal carotid arteries is less accurate (sensitivity, 55%-80%).20 Transcranial Doppler imaging is also used to assess for cerebral vasospasm after subarachnoid hemorrhage, monitor sickle cell disease patients’ overall risk for ischemic stroke, and augment thrombolysis. It is limited by the availability of an expert technician, and therefore is typically reserved for unstable patients or those who cannot receive contrast.20
Carotid duplex ultrasonography. A dynamic study such as duplex ultrasonography can be strongly considered for flow imaging of the extracranial carotids to evaluate for stenosis. Indications for carotid stenting or endarterectomy include 50% to 79% occlusion of the carotid artery on the same side as a recent transient ischemic attack or AIS. Carotid stenosis > 80% warrants consideration for intervention independent of a recent cerebrovascular accident. Interventions are typically performed 2 to 14 days after stroke.33 Although this study is of limited utility in the hyperacute setting, it is recommended within 24 hours after nondisabling stroke in the carotid territory, when (1) the patient is otherwise a candidate for a surgical or procedural intervention to address the stenosis and (2) none of the aforementioned studies that focus on neck vasculature have been performed.
Conventional (digital subtraction) angiography is the gold standard for mapping cerebrovascular disease because it is dynamic and highly accurate. It is, however, typically limited by the number of required personnel, its invasive nature, and the requirement for IV contrast. This study is performed during intra-arterial intervention techniques, including stent retrieval and intra-arterial thrombolysis.26
Impact of imaging on treatment
Imaging helps determine the cause and some characteristics of stroke, both of which can help determine therapy. Strokes can be broadly subcategorized as hemorrhagic or ischemic; recent studies suggest that 87% are ischemic.34 Knowledge of the historic details of the event, the patient (eg, known atrial fibrillation, anticoagulant use, history of falls), and findings on imaging can contribute to determine the cause of AIS, and can facilitate communication and consultation between the primary care physician and inpatient teams.35
Continue to: Best practices for stroke treatment...
Best practices for stroke treatment are based on the cause of the event.3 To identify the likely cause, the aforementioned characteristics are incorporated into one of the scoring systems, which seek to clarify either the cause or the phenotypic appearance of the AIS, which helps direct further testing and treatment. (The ASCOD36 and TOAST37 classification schemes are commonly used phenotypic and causative classifications, respectively.) Several (not all) of the broad phenotypic imaging patterns, with myriad clinical manifestations, are reviewed below. They include:
- Embolic stroke, which, classically, involves end circulation and therefore has cortical involvement. Typically, these originate from the heart or large extracranial arteries, and higher rates of atrial fibrillation and hypercoagulable states are implicated.
- Thrombotic stroke, which, typically, is from large vessels or small vessels, and occurs as a result of atherosclerosis. These strokes are more common at the origins or bifurcations of vessels. Symptoms of thrombotic stroke classically wax and wane slightly more frequently. Lacunar strokes are typically from thrombotic causes, although there are rare episodes of an embolic source contributing to a lacunar stroke syndrome.38
There is evidence for using MRI discrepancies between diffusion-weighted and FLAIR imaging to time AIS findings in so-called wake-up strokes.39 The rationale is that strokes < 4.5 hours old can be identified because they would have abnormal diffusion imaging components but normal findings with FLAIR. When these criteria were utilized in considering whether to treat with thrombolysis, there was a statistically significant improvement in 90-day modified Rankin scale (odds ratio = 1.61; 95% confidence interval, 1.09-2.36), but also an increased probability of death and intracerebral hemorrhage.39
A recent multicenter, randomized, open-label trial, with blinded outcomes assessment, showcased the efficacy of thrombectomy as an adjunct when ischemic brain territory was identified without frank infarction, as ascertained by CT perfusion within the anterior circulation. This trial showed that thrombectomy could be performed as long as 16 hours after the patient was last well-appearing and still result in an improved outcome with favorable imaging characteristics (on the modified Rankin scale, an ordinal score of 4 with medical therapy and an ordinal score of 3 with EVT [odds ratio = 2.77; 95% confidence interval, 1.63-4.70]).29 A 2018 multicenter, prospective, randomized trial with blinded assessment of endpoints extended this idea, demonstrating that, when there was mismatch of the clinical deficit (ie, high NIHSS score) and infarct volume (measured on diffusion-weighted MRI or CT perfusion), thrombectomy as late as 24 hours after the patient was last known to be well was beneficial for lesions in the anterior circulation—specifically, the intracranial internal carotid artery or the proximal middle cerebral artery.40
a Whether local emergency departments (EDs) should be bypassed in favor of a specialized stroke center is the subject of debate. The 2019 American Heart Association/American Stroke Association guidelines note the AHA’s Mission: Lifeline Stroke EMS algorithm, which bypasses the nearest ED in feared cases of large-vessel occlusion if travel to a comprehensive stroke center can be accomplished within 30 minutes of arrival at the scene. This is based on expert consensus.3,12,13
CORRESPONDENCE
Brian Ford, MD, 4301 Jones Bridge Road, Bethesda, MD; brian.ford@usuhs.edu.
1. Benjamin EJ, Virani SS, Callaway CW, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation. 2018;137:e67-e492.
2. Darves B. Collaboration key to post-stroke follow-up. ACP Internist. October 2009. https://acpinternist.org/archives/2009/10/stroke.htm. Accessed September 22, 2020.
3. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50e344-e418.
4. Sacco RL, Kasner SE, Broderick JP, et al; American Heart Association Stroke Council, Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular and Stroke Nursing; Council on Epidemiology and Prevention; Council on Peripheral Vascular Disease; Council on Nutrition, Physical Activity and Metabolism An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:2064-2089.
5. Aroor S, Singh R, Goldstein LB. BE-FAST (Balance, Eyes, Face, Arm, Speech, Time): Reducing the proportion of strokes missed using the FAST mnemonic. 2017;48:479-481.
6. Kidwell CS, Starkman S, Eckstein M, et al. Identifying stroke in the field. Prospective validation of the Los Angeles prehospital stroke screen (LAPSS). Stroke. 2000;31:71-76.
7. Llanes JN, Kidwell CS, Starkman S, et al. The Los Angeles Motor Scale (LAMS): a new measure to characterize stroke severity in the field. Prehosp Emerg Care. 2004;8:46-50.
8. Pérez de la Ossa N, Carrera D, Gorchs M, et al. Design and validation of a prehospital stroke scale to predict large arterial occlusion: the rapid arterial occlusion evaluation scale. Stroke. 2014;45:87-91.
9. Katz BS, McMullan JT, Sucharew H, et al. Design and validation of a prehospital scale to predict stroke severity: Cincinnati Prehospital Stroke Severity Scale. Stroke. 2015;466:1508-1512.
10. Kummer BR, et al. External validation of the Cincinnati Prehospital Stroke Severity Scale. J Stroke Cerebrovasc Dis. 2016;25:1270-1274.
11. Beume L-A, Hieber M, Kaller CP, et al. Large vessel occlusion in acute stroke. Stroke. 2018;49:2323-2329.
12. Man S, Zhao X, Uchino K, et al. Comparison of acute ischemic stroke care and outcomes between comprehensive stroke centers and primary stroke centers in the United States. Circ Cardiovasc Qual Outcomes. 2018;11:e004512.
13. American Heart Association (Mission: Lifeline—Stroke). Emergency medical services acute stroke routing. 2020. www.heart.org/-/media/files/professional/quality-improvement/mission-lifeline/2_25_2020/ds15698-qi-ems-algorithm_update-2142020.pdf?la=en. Accessed October 8, 2020.
14. Glober NK, Sporer KA, Guluma KZ, et al. Acute stroke: current evidence-based recommendations for prehospital care. West J Emerg Med. 2016;17:104-128.
15. NIH stroke scale. Bethesda, MD: National Institute of Neurological Disorders and Stroke, National Institutes of Health. www.stroke.nih.gov/resources/scale.htm. Accessed October 10, 2020.
16. Smith EE, Kent DM, Bulsara KR, et al; . Accuracy of prediction instruments for diagnosing large vessel occlusion in individuals with suspected stroke: a systematic review for the 2018 guidelines for the early management of patients with acute ischemic stroke. Stroke. 2018;49:e111-e122.
17. Woo D, Broderick JP, Kothari RU, et al. Does the National Institutes of Health Stroke Scale favor left hemisphere strokes? NINDS t-PA Stroke Study Group. Stroke. 1999;30:2355-2359.
18. Adams HP Jr, Davis PH, Leira EC, et al. Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology. 1999;53:126-131.
19. Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007;38:1091-1096.
20. Birenbaum D, Bancroft LW, Felsberg GJ. Imaging in acute stroke. West J Emerg Med. 2011;12:67-76.
21. Salmela MB, Mortazavi S, Jagadeesan BD, et al. ACR Appropriateness Criteria® Cerebrovascular Disease. J Am Coll Radiol. 2017;14:S34-S61.
22. Hemphill JC 3rd, Greenberg SM, Anderson CS, et al; American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46:2032-60.
23. Hacke W, Kaste M, Fieschi C, et al. Intravenous thrombolysis with recombinant tissue plasminogen activator for acute hemispheric stroke. The European Cooperative Acute Stroke Study (ECASS). JAMA. 1995;274:1017-1025.
24. The Tissue plasminogen activator for acute ischemic stroke. N Engl J Med, 1995;333:1581-1587.
25. Albers GW, Clark WM, Madden KP, et al. ATLANTIS trial: results for patients treated within 3 hours of stroke onset. Alteplase Thrombolysis for Acute Noninterventional Therapy in Ischemic Stroke. Stroke. 2002;33:493-495.
26. Khan R, Nael K, Erly W. Acute stroke imaging: what clinicians need to know. Am J Med. 2013;126:379-386.
27. Latchaw RE, Alberts MJ, Lev MH, et al; . Recommendations for managing of acute ischemic stroke: a scientific statement from the American Heart Association. Stroke. 2009;40:3646-3678.
28. Vagal A, Meganathan K, Kleindorfer DO, et al. Increasing use of computed tomographic perfusion and computed tomographic angiograms in acute ischemic stroke from 2006 to 2010. Stroke. 2014;45:1029-1034.
29. Albers GW, Marks MP, Kemp S, et al; DEFUSE 3 Investigators. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med. 2018;378:708-718.
30. Demeestere J, Wouters A, Christensen S, et al. Review of perfusion imaging in acute ischemic stroke: from time to tissue. Stroke. 2020;51:1017-1024.
31. Chalela JA, Kidwell CS, Nentwich LM, et al, Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: a prospective comparison. Lancet. 2007;369:293-298.
32. Aoki J, Kimura K, Iguchi Y, et al. FLAIR can estimate the onset time in acute ischemic stroke patients. J Neurol Sci. 2010;293:39-44.
33. Wabnitz AM, Turan TN. Symptomatic carotid artery stenosis: surgery, stenting, or medical therapy? Curr Treat Options Cardiovasc Med. 2017;19:62.
34. Muir KW, Santosh C. Imaging of acute stroke and transient ischaemic attack. J Neurol Neurosurg Psychiatry. 2005;76(suppl 3):iii19-iii28.
35. Cameron JI, Tsoi C, Marsella A.Optimizing stroke systems of care by enhancing transitions across care environments. Stroke. 2008;39:2637-2643.
36. Amarenco P, Bogousslavsky J, Caplan LR, et al. The ASCOD phenotyping of ischemic stroke (updated ASCO phenotyping). Cerebrovasc Dis. 2013;36:1-5.
37. Adams HP Jr, Bendixen BH, Kappelle LJ. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993;24:35-41.
38. Cacciatore A, Russo LS Jr. Lacunar infarction as an embolic complication of cardiac and arch angiography. Stroke. 1991;22:1603-1605.
39. Thomalla G, Simonsen CZ, Boutitie F, et al; WAKE-UP Investigators. MRI-guided thrombolysis for stroke with unknown time of onset. N Engl J Med. 2018;379:611-622.
40. Nogueira RG, Jadhav AP, Haussen DC, et al; DAWN Trial Investigators. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med. 2018;378:11-21.
Stroke ranks second behind heart disease as the leading cause of mortality worldwide, accounting for 1 of every 19 deaths,1 and remains a serious cause of morbidity. Best practices in stroke diagnosis and management can seem elusive to front-line clinicians, for 2 reasons: the rate of proliferation and nuance in stroke medicine and the fact that the typical scope of primary care practice exists apart from much of the diagnostic tools and management schema provided in stroke centers.2 In this article, we describe and update the diagnosis of stroke and review imaging modalities, their nuances, and their application in practice.
Diagnosis of acute stroke
Acute stroke is diagnosed upon observation of new neurologic deficits and congruent neuroimaging. Some updated definitions favor a silent form of cerebral ischemia manifested by imaging pathology only; this form is not discussed in this article. Although there are several characteristically distinct stroke syndromes, there is no way to clinically distinguish ischemic pathology from hemorrhagic pathology.
Some common symptoms that should prompt evaluation for stroke are part of the American Stroke Association FAST mnemonic designed to promote public health awareness3-5:
f ace droopinga rm weaknesss peech difficultyt ime to call 911.
Other commonly reported stroke symptoms include unilateral weakness or numbness, confusion, word-finding difficulty, visual problems, difficulty ambulating, dizziness, loss of balance or coordination, and thunderclap headache. A stroke should also be considered in the presence of any new focal neurologic deficit.3,4
Stroke patients should be triaged by emergency medical services using a stroke screening scale, such as BE-FAST5 (a modification of FAST that adds balance and eye assessments); the Los Angeles Prehospital Stroke Screen (LAPSS)6,7; the Rapid Arterial oCclusion Evaluation (RACE)8; and the Cincinnati Prehospital Stroke Severity Scale (CP-SSS)9,10 (see “Stroke screening scales for early identification and triage"). Studies have not found that any single prehospital stroke scale is superior to the others for reliably predicting large-vessel occlusion; therefore, prehospital assessment is typically based on practice patterns in a given locale.11 A patient (or family member or caregiver) who seeks your care for stroke symptoms should be told to call 911 and get emergency transport to a health care facility that can capably administer intravenous (IV) thrombolysis.a
SIDEBAR
Stroke screening scales for early identification and triage
National Institutes of Health Stroke Scale
www.stroke.nih.gov/resources/scale.htm
FAST
www.stroke.org/en/help-and-support/resource-library/fast-materials
BE-FAST
www.ahajournals.org/doi/10.1161/STROKEAHA.116.015169
Los Angeles Prehospital Stroke Screen (LAPSS)
http://stroke.ucla.edu/workfiles/prehospital-screen.pdf
Rapid Artery Occlusion Evaluation (RACE)
www.mdcalc.com/rapid-arterial-occlusion-evaluation-race-scale-stroke
Cincinnati Prehospital Stroke Severity Scale (CP-SSS)
https://www.mdcalc.com/cincinnati-prehospital-stroke-severity-scale-cp-sss
First responders should elicit “last-known-normal” time; this critical information can aid in diagnosis and drive therapeutic options, especially if patients are unaccompanied at time of transport to a higher echelon of care. A point-of-care blood glucose test should be performed by emergency medical staff, with dextrose administered for a level < 45 mg/dL. Establishing IV access for fluids, medications, and contrast can be considered if it does not delay transport. A 12-lead electrocardiogram can also be considered, again, as long as it does not delay transport to a facility capable of providing definitive therapy. Notification by emergency services staff before arrival and transport of the patient to such a facility is the essential element of prehospital care, and should be prioritized above ancillary testing beyond the stroke assessment.14
Guidelines recommend use of the National Institutes of Health Stroke Scale (NIHSS; www.stroke.nih.gov/resources/scale.htm) for clinical evaluation upon arrival at the ED.15 Although no scale has been identified that can reliably predict large-vessel occlusion amenable to endovascular therapy (EVT), no other score has been found to outperform the NIHSS in achieving meaningful patient outcomes.16 Furthermore, NIHSS has been validated to track clinical changes in response to therapy, is widely utilized, and is free.
Continue to: A criticism of the NIHSS...
A criticism of the NIHSS is its bias toward left-hemispheric ischemic pathology.17 NIHSS includes 11 questions on a scale of 0 to 42; typically, a score < 4 is associated with a higher chance of a positive clinical outcome.18 There is no minimum or maximum NIHSS score that precludes treatment with thrombolysis or EVT.
Other commonly used scores in acute stroke include disability assessments. The modified Rankin scale, which is used most often, features a score of 0 (symptom-free) to 6 (death). A modified Rankin scale score of 0 or 1 is considered an indication of a favorable outcome after stroke.19 Note that these functional scores are not always part of an acute assessment but can be done early in the clinical course to gauge the response to treatment, and are collected for stroke-center certification.
Imaging modalities
Imaging is recommended within 20 minutes of arrival in the ED in a stroke patient who might be a candidate for thrombolysis or thrombectomy.3 There, imaging modalities commonly performed are noncontrast-enhanced head computed tomography (NCHCT); computed tomography (CT) angiography, with or without perfusion; and diffusion-weighted magnetic resonance imaging (MRI).20,21 In addition, more highly specialized imaging modalities are available for the evaluation of the stroke patient in specific, often limited, circumstances. All these modalities are described below and compared in the TABLE,20,21 using the ACR Appropriateness Criteria (of the American College of Radiology),21 which are guidelines for appropriate imaging of stroke, based on a clinical complaint. Separate recommendations and appraisals are offered by the most recent American Heart Association/American Stroke Association (AHA/ASA) guideline.3

NCHCT. This study should be performed within 20 minutes after arrival at the ED because it provides rapid assessment of intracerebral hemorrhage, can effectively corroborate the diagnosis of some stroke mimickers, and identifies some candidates for EVT or thrombolysis3,21,22 (typically, the decision to proceed with EVT is based on adjunct imaging studies discussed in a bit). Evaluation for intracerebral hemorrhage is required prior to administering thrombolysis. Ischemic changes can be seen with variable specificity and sensitivity on NCHCT, depending on how much time has passed since the original insult. In all historical trials, CT was the only imaging modality used in the diagnosis of acute ischemic stroke (AIS) that suggested benefit from IV thrombolysis.23-25

Acute, subacute, and chronic changes can be seen on NCHCT, although the modality has limited sensitivity for identifying AIS (ie, approximately 75% within 6 hours after the original insult):
- Acute findings on NCHCT include intracellular edema, which causes loss of the gray matter–white matter interface and effacement of the cortical sulci. This occurs as a result of increased cellular uptake of water in response to ischemia and cell death, resulting in a decreased density of tissue (hypoattenuation) in affected areas.
- Subacute changes appear in the 2- to 5-day window, including vasogenic edema with greater mass effect, hypoattenuation, and well-defined margins.3,20,21
- Chronic vascular findings on NCHCT include loss of brain tissue and hypoattenuation.
Continue to: NCHCT is typically performed...
NCHCT is typically performed in advance of other adjunct imaging modalities.3,20,21 Baseline NCHCT can be performed on patients with advanced kidney disease and those who have an indwelling metallic device.
CT angiography is performed with timed contrast, providing a 3-dimensional representation of the cerebral vasculature; the entire intracranial and extracranial vasculature, including the aortic arch, can be mapped in approximately 60 seconds. CT angiography is sensitive in identifying areas of stenosis > 50% and identifies clinically significant areas of stenosis up to approximately 90% of the time.26 For this reason, it is particularly helpful in identifying candidates for an interventional strategy beyond pharmacotherapeutic thrombolysis. In addition, CT angiography can visualize aneurysmal dilation and dissection, and help with the planning of interventions—specifically, the confident administration of thrombolysis or more specific planning for target lesions and EVT.
It also can help identify a host of vascular phenomena, such as arteriovenous malformations, Moyamoya disease (progressive arterial blockage within the basal ganglia and compensatory microvascularization), and some vasculopathies.20,27 In intracranial hemorrhage, CT with angiography can help evaluate for structural malformations and identify patients at risk of hematoma expansion.22
CT perfusion. Many stroke centers will perform a CT perfusion study,28 which encompasses as many as 3 different CT sequences:
- NCHCT
- vertex-to-arch angiography with contrast bolus
- administration of contrast and capture of a dynamic sequence through 1 or 2 slabs of tissue, allowing for the generation of maps of cerebral blood flow (CBF), mean transit time (MTT), and cerebral blood volume (CBV) of the entire cerebral vasculature.
The interplay of these 3 sequences drives characterization of lesions (ie, CBF = CBV/MTT). An infarct is characterized by low CBF, low CBV, and elevated MTT. In penumbral tissue, MTT is elevated but CBF is slightly decreased and CBV is normal or increased. Using CT perfusion, areas throughout the ischemic penumbra can be surveyed for favorable interventional characteristics.20,29
Continue to: A CT perfusion study adds...
A CT perfusion study adds at least 60 seconds to NCHCT. This modality can be useful in planning interventions and for stratifying appropriateness of reperfusion strategies in strokes of unknown duration.3,30 CT perfusion can be performed on any multidetector CT scan but (1) requires specialized software and expertise to interpret and (2) subjects the patient to a significant radiation dose, which, if incorrectly administered, can be considerably higher than intended.20,26,27
Diffusion-weighted MRI. This is the most sensitive study for demonstrating early ischemic changes; however, limitations include lack of availability, contraindication in patients with metallic indwelling implants, and duration of the study—although, at some stroke centers, diffusion-weighted MRI can be performed in ≤ 10 minutes.
MRI and NCHCT have comparable sensitivity in detecting intracranial hemorrhage. MRI is likely more sensitive in identifying areas of microhemorrhage: In diffusion-weighted MRI, the sensitivity of stroke detection increases to > 95%.31 The modality relies on the comparable movement of water through damaged vs normal neuronal tissue. Diffusion-weighted MRI does not require administration of concomitant contrast, which can be a benefit in patients who are allergic to gadolinium-based contrast agents or have advanced kidney disease that precludes the use of contrast. It typically does not result in adequate characterization of extracranial vasculature.
Other MRI modalities. These MRI extensions include magnetic resonance (MR) perfusion and MR angiography. Whereas diffusion-weighted MRI (discussed above) offers the most rapid and sensitive evaluation for ischemia, fluid-attenuated inversion recovery (FLAIR) imaging has been utilized as a comparator to isolated diffusion-weighted MRI to help determine stroke duration. FLAIR signal positivity typically occurs 6 to 24 hours after the initial insult but is negative in stroke that occurred < 3 hours earlier.32
MRI is limited, in terms of availability and increased study duration, especially when it comes to timely administration of thrombolysis. A benefit of this modality is less radiation and, as noted, superior sensitivity for ischemia. Diffusion-weighted MRI combined with MR perfusion analysis can help isolate areas of the ischemic penumbra. MR perfusion is performed for a similar reason as CT perfusion, although logistical execution across those modalities is significantly different. Considerations for choosing MR perfusion or CT perfusion should be made on an individual basis and based on available local resources and accepted local practice patterns.26
Continue to: In the subacute setting...
In the subacute setting, MR perfusion and MR angiography of the head and the neck are often performed to identify stenosis, dissection, and more subtle mimickers of cerebrovascular accident not ascertained on initial CT evaluation. These studies are typically performed well outside the window for thrombolysis or intervention.26 No guidelines specifically direct or recommend this practice pattern. The superior sensitivity and cerebral blood flow mapping of MR perfusion and MR angiography might be useful for validating a suspected diagnosis of ischemic stroke and providing phenotypic information about AIS events.
Transcranial Doppler imaging relies on bony windows to assess intracranial vascular flow, velocity, direction, and reactivity. This information can be utilized to diagnose stenosis or occlusion. This modality is principally used to evaluate for stenosis in the anterior circulation (sensitivity, 70%-90%; specificity, 90%-95%).20 Evaluation of the basilar, vertebral, and internal carotid arteries is less accurate (sensitivity, 55%-80%).20 Transcranial Doppler imaging is also used to assess for cerebral vasospasm after subarachnoid hemorrhage, monitor sickle cell disease patients’ overall risk for ischemic stroke, and augment thrombolysis. It is limited by the availability of an expert technician, and therefore is typically reserved for unstable patients or those who cannot receive contrast.20
Carotid duplex ultrasonography. A dynamic study such as duplex ultrasonography can be strongly considered for flow imaging of the extracranial carotids to evaluate for stenosis. Indications for carotid stenting or endarterectomy include 50% to 79% occlusion of the carotid artery on the same side as a recent transient ischemic attack or AIS. Carotid stenosis > 80% warrants consideration for intervention independent of a recent cerebrovascular accident. Interventions are typically performed 2 to 14 days after stroke.33 Although this study is of limited utility in the hyperacute setting, it is recommended within 24 hours after nondisabling stroke in the carotid territory, when (1) the patient is otherwise a candidate for a surgical or procedural intervention to address the stenosis and (2) none of the aforementioned studies that focus on neck vasculature have been performed.
Conventional (digital subtraction) angiography is the gold standard for mapping cerebrovascular disease because it is dynamic and highly accurate. It is, however, typically limited by the number of required personnel, its invasive nature, and the requirement for IV contrast. This study is performed during intra-arterial intervention techniques, including stent retrieval and intra-arterial thrombolysis.26
Impact of imaging on treatment
Imaging helps determine the cause and some characteristics of stroke, both of which can help determine therapy. Strokes can be broadly subcategorized as hemorrhagic or ischemic; recent studies suggest that 87% are ischemic.34 Knowledge of the historic details of the event, the patient (eg, known atrial fibrillation, anticoagulant use, history of falls), and findings on imaging can contribute to determine the cause of AIS, and can facilitate communication and consultation between the primary care physician and inpatient teams.35
Continue to: Best practices for stroke treatment...
Best practices for stroke treatment are based on the cause of the event.3 To identify the likely cause, the aforementioned characteristics are incorporated into one of the scoring systems, which seek to clarify either the cause or the phenotypic appearance of the AIS, which helps direct further testing and treatment. (The ASCOD36 and TOAST37 classification schemes are commonly used phenotypic and causative classifications, respectively.) Several (not all) of the broad phenotypic imaging patterns, with myriad clinical manifestations, are reviewed below. They include:
- Embolic stroke, which, classically, involves end circulation and therefore has cortical involvement. Typically, these originate from the heart or large extracranial arteries, and higher rates of atrial fibrillation and hypercoagulable states are implicated.
- Thrombotic stroke, which, typically, is from large vessels or small vessels, and occurs as a result of atherosclerosis. These strokes are more common at the origins or bifurcations of vessels. Symptoms of thrombotic stroke classically wax and wane slightly more frequently. Lacunar strokes are typically from thrombotic causes, although there are rare episodes of an embolic source contributing to a lacunar stroke syndrome.38
There is evidence for using MRI discrepancies between diffusion-weighted and FLAIR imaging to time AIS findings in so-called wake-up strokes.39 The rationale is that strokes < 4.5 hours old can be identified because they would have abnormal diffusion imaging components but normal findings with FLAIR. When these criteria were utilized in considering whether to treat with thrombolysis, there was a statistically significant improvement in 90-day modified Rankin scale (odds ratio = 1.61; 95% confidence interval, 1.09-2.36), but also an increased probability of death and intracerebral hemorrhage.39
A recent multicenter, randomized, open-label trial, with blinded outcomes assessment, showcased the efficacy of thrombectomy as an adjunct when ischemic brain territory was identified without frank infarction, as ascertained by CT perfusion within the anterior circulation. This trial showed that thrombectomy could be performed as long as 16 hours after the patient was last well-appearing and still result in an improved outcome with favorable imaging characteristics (on the modified Rankin scale, an ordinal score of 4 with medical therapy and an ordinal score of 3 with EVT [odds ratio = 2.77; 95% confidence interval, 1.63-4.70]).29 A 2018 multicenter, prospective, randomized trial with blinded assessment of endpoints extended this idea, demonstrating that, when there was mismatch of the clinical deficit (ie, high NIHSS score) and infarct volume (measured on diffusion-weighted MRI or CT perfusion), thrombectomy as late as 24 hours after the patient was last known to be well was beneficial for lesions in the anterior circulation—specifically, the intracranial internal carotid artery or the proximal middle cerebral artery.40
a Whether local emergency departments (EDs) should be bypassed in favor of a specialized stroke center is the subject of debate. The 2019 American Heart Association/American Stroke Association guidelines note the AHA’s Mission: Lifeline Stroke EMS algorithm, which bypasses the nearest ED in feared cases of large-vessel occlusion if travel to a comprehensive stroke center can be accomplished within 30 minutes of arrival at the scene. This is based on expert consensus.3,12,13
CORRESPONDENCE
Brian Ford, MD, 4301 Jones Bridge Road, Bethesda, MD; brian.ford@usuhs.edu.
Stroke ranks second behind heart disease as the leading cause of mortality worldwide, accounting for 1 of every 19 deaths,1 and remains a serious cause of morbidity. Best practices in stroke diagnosis and management can seem elusive to front-line clinicians, for 2 reasons: the rate of proliferation and nuance in stroke medicine and the fact that the typical scope of primary care practice exists apart from much of the diagnostic tools and management schema provided in stroke centers.2 In this article, we describe and update the diagnosis of stroke and review imaging modalities, their nuances, and their application in practice.
Diagnosis of acute stroke
Acute stroke is diagnosed upon observation of new neurologic deficits and congruent neuroimaging. Some updated definitions favor a silent form of cerebral ischemia manifested by imaging pathology only; this form is not discussed in this article. Although there are several characteristically distinct stroke syndromes, there is no way to clinically distinguish ischemic pathology from hemorrhagic pathology.
Some common symptoms that should prompt evaluation for stroke are part of the American Stroke Association FAST mnemonic designed to promote public health awareness3-5:
f ace droopinga rm weaknesss peech difficultyt ime to call 911.
Other commonly reported stroke symptoms include unilateral weakness or numbness, confusion, word-finding difficulty, visual problems, difficulty ambulating, dizziness, loss of balance or coordination, and thunderclap headache. A stroke should also be considered in the presence of any new focal neurologic deficit.3,4
Stroke patients should be triaged by emergency medical services using a stroke screening scale, such as BE-FAST5 (a modification of FAST that adds balance and eye assessments); the Los Angeles Prehospital Stroke Screen (LAPSS)6,7; the Rapid Arterial oCclusion Evaluation (RACE)8; and the Cincinnati Prehospital Stroke Severity Scale (CP-SSS)9,10 (see “Stroke screening scales for early identification and triage"). Studies have not found that any single prehospital stroke scale is superior to the others for reliably predicting large-vessel occlusion; therefore, prehospital assessment is typically based on practice patterns in a given locale.11 A patient (or family member or caregiver) who seeks your care for stroke symptoms should be told to call 911 and get emergency transport to a health care facility that can capably administer intravenous (IV) thrombolysis.a
SIDEBAR
Stroke screening scales for early identification and triage
National Institutes of Health Stroke Scale
www.stroke.nih.gov/resources/scale.htm
FAST
www.stroke.org/en/help-and-support/resource-library/fast-materials
BE-FAST
www.ahajournals.org/doi/10.1161/STROKEAHA.116.015169
Los Angeles Prehospital Stroke Screen (LAPSS)
http://stroke.ucla.edu/workfiles/prehospital-screen.pdf
Rapid Artery Occlusion Evaluation (RACE)
www.mdcalc.com/rapid-arterial-occlusion-evaluation-race-scale-stroke
Cincinnati Prehospital Stroke Severity Scale (CP-SSS)
https://www.mdcalc.com/cincinnati-prehospital-stroke-severity-scale-cp-sss
First responders should elicit “last-known-normal” time; this critical information can aid in diagnosis and drive therapeutic options, especially if patients are unaccompanied at time of transport to a higher echelon of care. A point-of-care blood glucose test should be performed by emergency medical staff, with dextrose administered for a level < 45 mg/dL. Establishing IV access for fluids, medications, and contrast can be considered if it does not delay transport. A 12-lead electrocardiogram can also be considered, again, as long as it does not delay transport to a facility capable of providing definitive therapy. Notification by emergency services staff before arrival and transport of the patient to such a facility is the essential element of prehospital care, and should be prioritized above ancillary testing beyond the stroke assessment.14
Guidelines recommend use of the National Institutes of Health Stroke Scale (NIHSS; www.stroke.nih.gov/resources/scale.htm) for clinical evaluation upon arrival at the ED.15 Although no scale has been identified that can reliably predict large-vessel occlusion amenable to endovascular therapy (EVT), no other score has been found to outperform the NIHSS in achieving meaningful patient outcomes.16 Furthermore, NIHSS has been validated to track clinical changes in response to therapy, is widely utilized, and is free.
Continue to: A criticism of the NIHSS...
A criticism of the NIHSS is its bias toward left-hemispheric ischemic pathology.17 NIHSS includes 11 questions on a scale of 0 to 42; typically, a score < 4 is associated with a higher chance of a positive clinical outcome.18 There is no minimum or maximum NIHSS score that precludes treatment with thrombolysis or EVT.
Other commonly used scores in acute stroke include disability assessments. The modified Rankin scale, which is used most often, features a score of 0 (symptom-free) to 6 (death). A modified Rankin scale score of 0 or 1 is considered an indication of a favorable outcome after stroke.19 Note that these functional scores are not always part of an acute assessment but can be done early in the clinical course to gauge the response to treatment, and are collected for stroke-center certification.
Imaging modalities
Imaging is recommended within 20 minutes of arrival in the ED in a stroke patient who might be a candidate for thrombolysis or thrombectomy.3 There, imaging modalities commonly performed are noncontrast-enhanced head computed tomography (NCHCT); computed tomography (CT) angiography, with or without perfusion; and diffusion-weighted magnetic resonance imaging (MRI).20,21 In addition, more highly specialized imaging modalities are available for the evaluation of the stroke patient in specific, often limited, circumstances. All these modalities are described below and compared in the TABLE,20,21 using the ACR Appropriateness Criteria (of the American College of Radiology),21 which are guidelines for appropriate imaging of stroke, based on a clinical complaint. Separate recommendations and appraisals are offered by the most recent American Heart Association/American Stroke Association (AHA/ASA) guideline.3

NCHCT. This study should be performed within 20 minutes after arrival at the ED because it provides rapid assessment of intracerebral hemorrhage, can effectively corroborate the diagnosis of some stroke mimickers, and identifies some candidates for EVT or thrombolysis3,21,22 (typically, the decision to proceed with EVT is based on adjunct imaging studies discussed in a bit). Evaluation for intracerebral hemorrhage is required prior to administering thrombolysis. Ischemic changes can be seen with variable specificity and sensitivity on NCHCT, depending on how much time has passed since the original insult. In all historical trials, CT was the only imaging modality used in the diagnosis of acute ischemic stroke (AIS) that suggested benefit from IV thrombolysis.23-25

Acute, subacute, and chronic changes can be seen on NCHCT, although the modality has limited sensitivity for identifying AIS (ie, approximately 75% within 6 hours after the original insult):
- Acute findings on NCHCT include intracellular edema, which causes loss of the gray matter–white matter interface and effacement of the cortical sulci. This occurs as a result of increased cellular uptake of water in response to ischemia and cell death, resulting in a decreased density of tissue (hypoattenuation) in affected areas.
- Subacute changes appear in the 2- to 5-day window, including vasogenic edema with greater mass effect, hypoattenuation, and well-defined margins.3,20,21
- Chronic vascular findings on NCHCT include loss of brain tissue and hypoattenuation.
Continue to: NCHCT is typically performed...
NCHCT is typically performed in advance of other adjunct imaging modalities.3,20,21 Baseline NCHCT can be performed on patients with advanced kidney disease and those who have an indwelling metallic device.
CT angiography is performed with timed contrast, providing a 3-dimensional representation of the cerebral vasculature; the entire intracranial and extracranial vasculature, including the aortic arch, can be mapped in approximately 60 seconds. CT angiography is sensitive in identifying areas of stenosis > 50% and identifies clinically significant areas of stenosis up to approximately 90% of the time.26 For this reason, it is particularly helpful in identifying candidates for an interventional strategy beyond pharmacotherapeutic thrombolysis. In addition, CT angiography can visualize aneurysmal dilation and dissection, and help with the planning of interventions—specifically, the confident administration of thrombolysis or more specific planning for target lesions and EVT.
It also can help identify a host of vascular phenomena, such as arteriovenous malformations, Moyamoya disease (progressive arterial blockage within the basal ganglia and compensatory microvascularization), and some vasculopathies.20,27 In intracranial hemorrhage, CT with angiography can help evaluate for structural malformations and identify patients at risk of hematoma expansion.22
CT perfusion. Many stroke centers will perform a CT perfusion study,28 which encompasses as many as 3 different CT sequences:
- NCHCT
- vertex-to-arch angiography with contrast bolus
- administration of contrast and capture of a dynamic sequence through 1 or 2 slabs of tissue, allowing for the generation of maps of cerebral blood flow (CBF), mean transit time (MTT), and cerebral blood volume (CBV) of the entire cerebral vasculature.
The interplay of these 3 sequences drives characterization of lesions (ie, CBF = CBV/MTT). An infarct is characterized by low CBF, low CBV, and elevated MTT. In penumbral tissue, MTT is elevated but CBF is slightly decreased and CBV is normal or increased. Using CT perfusion, areas throughout the ischemic penumbra can be surveyed for favorable interventional characteristics.20,29
Continue to: A CT perfusion study adds...
A CT perfusion study adds at least 60 seconds to NCHCT. This modality can be useful in planning interventions and for stratifying appropriateness of reperfusion strategies in strokes of unknown duration.3,30 CT perfusion can be performed on any multidetector CT scan but (1) requires specialized software and expertise to interpret and (2) subjects the patient to a significant radiation dose, which, if incorrectly administered, can be considerably higher than intended.20,26,27
Diffusion-weighted MRI. This is the most sensitive study for demonstrating early ischemic changes; however, limitations include lack of availability, contraindication in patients with metallic indwelling implants, and duration of the study—although, at some stroke centers, diffusion-weighted MRI can be performed in ≤ 10 minutes.
MRI and NCHCT have comparable sensitivity in detecting intracranial hemorrhage. MRI is likely more sensitive in identifying areas of microhemorrhage: In diffusion-weighted MRI, the sensitivity of stroke detection increases to > 95%.31 The modality relies on the comparable movement of water through damaged vs normal neuronal tissue. Diffusion-weighted MRI does not require administration of concomitant contrast, which can be a benefit in patients who are allergic to gadolinium-based contrast agents or have advanced kidney disease that precludes the use of contrast. It typically does not result in adequate characterization of extracranial vasculature.
Other MRI modalities. These MRI extensions include magnetic resonance (MR) perfusion and MR angiography. Whereas diffusion-weighted MRI (discussed above) offers the most rapid and sensitive evaluation for ischemia, fluid-attenuated inversion recovery (FLAIR) imaging has been utilized as a comparator to isolated diffusion-weighted MRI to help determine stroke duration. FLAIR signal positivity typically occurs 6 to 24 hours after the initial insult but is negative in stroke that occurred < 3 hours earlier.32
MRI is limited, in terms of availability and increased study duration, especially when it comes to timely administration of thrombolysis. A benefit of this modality is less radiation and, as noted, superior sensitivity for ischemia. Diffusion-weighted MRI combined with MR perfusion analysis can help isolate areas of the ischemic penumbra. MR perfusion is performed for a similar reason as CT perfusion, although logistical execution across those modalities is significantly different. Considerations for choosing MR perfusion or CT perfusion should be made on an individual basis and based on available local resources and accepted local practice patterns.26
Continue to: In the subacute setting...
In the subacute setting, MR perfusion and MR angiography of the head and the neck are often performed to identify stenosis, dissection, and more subtle mimickers of cerebrovascular accident not ascertained on initial CT evaluation. These studies are typically performed well outside the window for thrombolysis or intervention.26 No guidelines specifically direct or recommend this practice pattern. The superior sensitivity and cerebral blood flow mapping of MR perfusion and MR angiography might be useful for validating a suspected diagnosis of ischemic stroke and providing phenotypic information about AIS events.
Transcranial Doppler imaging relies on bony windows to assess intracranial vascular flow, velocity, direction, and reactivity. This information can be utilized to diagnose stenosis or occlusion. This modality is principally used to evaluate for stenosis in the anterior circulation (sensitivity, 70%-90%; specificity, 90%-95%).20 Evaluation of the basilar, vertebral, and internal carotid arteries is less accurate (sensitivity, 55%-80%).20 Transcranial Doppler imaging is also used to assess for cerebral vasospasm after subarachnoid hemorrhage, monitor sickle cell disease patients’ overall risk for ischemic stroke, and augment thrombolysis. It is limited by the availability of an expert technician, and therefore is typically reserved for unstable patients or those who cannot receive contrast.20
Carotid duplex ultrasonography. A dynamic study such as duplex ultrasonography can be strongly considered for flow imaging of the extracranial carotids to evaluate for stenosis. Indications for carotid stenting or endarterectomy include 50% to 79% occlusion of the carotid artery on the same side as a recent transient ischemic attack or AIS. Carotid stenosis > 80% warrants consideration for intervention independent of a recent cerebrovascular accident. Interventions are typically performed 2 to 14 days after stroke.33 Although this study is of limited utility in the hyperacute setting, it is recommended within 24 hours after nondisabling stroke in the carotid territory, when (1) the patient is otherwise a candidate for a surgical or procedural intervention to address the stenosis and (2) none of the aforementioned studies that focus on neck vasculature have been performed.
Conventional (digital subtraction) angiography is the gold standard for mapping cerebrovascular disease because it is dynamic and highly accurate. It is, however, typically limited by the number of required personnel, its invasive nature, and the requirement for IV contrast. This study is performed during intra-arterial intervention techniques, including stent retrieval and intra-arterial thrombolysis.26
Impact of imaging on treatment
Imaging helps determine the cause and some characteristics of stroke, both of which can help determine therapy. Strokes can be broadly subcategorized as hemorrhagic or ischemic; recent studies suggest that 87% are ischemic.34 Knowledge of the historic details of the event, the patient (eg, known atrial fibrillation, anticoagulant use, history of falls), and findings on imaging can contribute to determine the cause of AIS, and can facilitate communication and consultation between the primary care physician and inpatient teams.35
Continue to: Best practices for stroke treatment...
Best practices for stroke treatment are based on the cause of the event.3 To identify the likely cause, the aforementioned characteristics are incorporated into one of the scoring systems, which seek to clarify either the cause or the phenotypic appearance of the AIS, which helps direct further testing and treatment. (The ASCOD36 and TOAST37 classification schemes are commonly used phenotypic and causative classifications, respectively.) Several (not all) of the broad phenotypic imaging patterns, with myriad clinical manifestations, are reviewed below. They include:
- Embolic stroke, which, classically, involves end circulation and therefore has cortical involvement. Typically, these originate from the heart or large extracranial arteries, and higher rates of atrial fibrillation and hypercoagulable states are implicated.
- Thrombotic stroke, which, typically, is from large vessels or small vessels, and occurs as a result of atherosclerosis. These strokes are more common at the origins or bifurcations of vessels. Symptoms of thrombotic stroke classically wax and wane slightly more frequently. Lacunar strokes are typically from thrombotic causes, although there are rare episodes of an embolic source contributing to a lacunar stroke syndrome.38
There is evidence for using MRI discrepancies between diffusion-weighted and FLAIR imaging to time AIS findings in so-called wake-up strokes.39 The rationale is that strokes < 4.5 hours old can be identified because they would have abnormal diffusion imaging components but normal findings with FLAIR. When these criteria were utilized in considering whether to treat with thrombolysis, there was a statistically significant improvement in 90-day modified Rankin scale (odds ratio = 1.61; 95% confidence interval, 1.09-2.36), but also an increased probability of death and intracerebral hemorrhage.39
A recent multicenter, randomized, open-label trial, with blinded outcomes assessment, showcased the efficacy of thrombectomy as an adjunct when ischemic brain territory was identified without frank infarction, as ascertained by CT perfusion within the anterior circulation. This trial showed that thrombectomy could be performed as long as 16 hours after the patient was last well-appearing and still result in an improved outcome with favorable imaging characteristics (on the modified Rankin scale, an ordinal score of 4 with medical therapy and an ordinal score of 3 with EVT [odds ratio = 2.77; 95% confidence interval, 1.63-4.70]).29 A 2018 multicenter, prospective, randomized trial with blinded assessment of endpoints extended this idea, demonstrating that, when there was mismatch of the clinical deficit (ie, high NIHSS score) and infarct volume (measured on diffusion-weighted MRI or CT perfusion), thrombectomy as late as 24 hours after the patient was last known to be well was beneficial for lesions in the anterior circulation—specifically, the intracranial internal carotid artery or the proximal middle cerebral artery.40
a Whether local emergency departments (EDs) should be bypassed in favor of a specialized stroke center is the subject of debate. The 2019 American Heart Association/American Stroke Association guidelines note the AHA’s Mission: Lifeline Stroke EMS algorithm, which bypasses the nearest ED in feared cases of large-vessel occlusion if travel to a comprehensive stroke center can be accomplished within 30 minutes of arrival at the scene. This is based on expert consensus.3,12,13
CORRESPONDENCE
Brian Ford, MD, 4301 Jones Bridge Road, Bethesda, MD; brian.ford@usuhs.edu.
1. Benjamin EJ, Virani SS, Callaway CW, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation. 2018;137:e67-e492.
2. Darves B. Collaboration key to post-stroke follow-up. ACP Internist. October 2009. https://acpinternist.org/archives/2009/10/stroke.htm. Accessed September 22, 2020.
3. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50e344-e418.
4. Sacco RL, Kasner SE, Broderick JP, et al; American Heart Association Stroke Council, Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular and Stroke Nursing; Council on Epidemiology and Prevention; Council on Peripheral Vascular Disease; Council on Nutrition, Physical Activity and Metabolism An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:2064-2089.
5. Aroor S, Singh R, Goldstein LB. BE-FAST (Balance, Eyes, Face, Arm, Speech, Time): Reducing the proportion of strokes missed using the FAST mnemonic. 2017;48:479-481.
6. Kidwell CS, Starkman S, Eckstein M, et al. Identifying stroke in the field. Prospective validation of the Los Angeles prehospital stroke screen (LAPSS). Stroke. 2000;31:71-76.
7. Llanes JN, Kidwell CS, Starkman S, et al. The Los Angeles Motor Scale (LAMS): a new measure to characterize stroke severity in the field. Prehosp Emerg Care. 2004;8:46-50.
8. Pérez de la Ossa N, Carrera D, Gorchs M, et al. Design and validation of a prehospital stroke scale to predict large arterial occlusion: the rapid arterial occlusion evaluation scale. Stroke. 2014;45:87-91.
9. Katz BS, McMullan JT, Sucharew H, et al. Design and validation of a prehospital scale to predict stroke severity: Cincinnati Prehospital Stroke Severity Scale. Stroke. 2015;466:1508-1512.
10. Kummer BR, et al. External validation of the Cincinnati Prehospital Stroke Severity Scale. J Stroke Cerebrovasc Dis. 2016;25:1270-1274.
11. Beume L-A, Hieber M, Kaller CP, et al. Large vessel occlusion in acute stroke. Stroke. 2018;49:2323-2329.
12. Man S, Zhao X, Uchino K, et al. Comparison of acute ischemic stroke care and outcomes between comprehensive stroke centers and primary stroke centers in the United States. Circ Cardiovasc Qual Outcomes. 2018;11:e004512.
13. American Heart Association (Mission: Lifeline—Stroke). Emergency medical services acute stroke routing. 2020. www.heart.org/-/media/files/professional/quality-improvement/mission-lifeline/2_25_2020/ds15698-qi-ems-algorithm_update-2142020.pdf?la=en. Accessed October 8, 2020.
14. Glober NK, Sporer KA, Guluma KZ, et al. Acute stroke: current evidence-based recommendations for prehospital care. West J Emerg Med. 2016;17:104-128.
15. NIH stroke scale. Bethesda, MD: National Institute of Neurological Disorders and Stroke, National Institutes of Health. www.stroke.nih.gov/resources/scale.htm. Accessed October 10, 2020.
16. Smith EE, Kent DM, Bulsara KR, et al; . Accuracy of prediction instruments for diagnosing large vessel occlusion in individuals with suspected stroke: a systematic review for the 2018 guidelines for the early management of patients with acute ischemic stroke. Stroke. 2018;49:e111-e122.
17. Woo D, Broderick JP, Kothari RU, et al. Does the National Institutes of Health Stroke Scale favor left hemisphere strokes? NINDS t-PA Stroke Study Group. Stroke. 1999;30:2355-2359.
18. Adams HP Jr, Davis PH, Leira EC, et al. Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology. 1999;53:126-131.
19. Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007;38:1091-1096.
20. Birenbaum D, Bancroft LW, Felsberg GJ. Imaging in acute stroke. West J Emerg Med. 2011;12:67-76.
21. Salmela MB, Mortazavi S, Jagadeesan BD, et al. ACR Appropriateness Criteria® Cerebrovascular Disease. J Am Coll Radiol. 2017;14:S34-S61.
22. Hemphill JC 3rd, Greenberg SM, Anderson CS, et al; American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46:2032-60.
23. Hacke W, Kaste M, Fieschi C, et al. Intravenous thrombolysis with recombinant tissue plasminogen activator for acute hemispheric stroke. The European Cooperative Acute Stroke Study (ECASS). JAMA. 1995;274:1017-1025.
24. The Tissue plasminogen activator for acute ischemic stroke. N Engl J Med, 1995;333:1581-1587.
25. Albers GW, Clark WM, Madden KP, et al. ATLANTIS trial: results for patients treated within 3 hours of stroke onset. Alteplase Thrombolysis for Acute Noninterventional Therapy in Ischemic Stroke. Stroke. 2002;33:493-495.
26. Khan R, Nael K, Erly W. Acute stroke imaging: what clinicians need to know. Am J Med. 2013;126:379-386.
27. Latchaw RE, Alberts MJ, Lev MH, et al; . Recommendations for managing of acute ischemic stroke: a scientific statement from the American Heart Association. Stroke. 2009;40:3646-3678.
28. Vagal A, Meganathan K, Kleindorfer DO, et al. Increasing use of computed tomographic perfusion and computed tomographic angiograms in acute ischemic stroke from 2006 to 2010. Stroke. 2014;45:1029-1034.
29. Albers GW, Marks MP, Kemp S, et al; DEFUSE 3 Investigators. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med. 2018;378:708-718.
30. Demeestere J, Wouters A, Christensen S, et al. Review of perfusion imaging in acute ischemic stroke: from time to tissue. Stroke. 2020;51:1017-1024.
31. Chalela JA, Kidwell CS, Nentwich LM, et al, Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: a prospective comparison. Lancet. 2007;369:293-298.
32. Aoki J, Kimura K, Iguchi Y, et al. FLAIR can estimate the onset time in acute ischemic stroke patients. J Neurol Sci. 2010;293:39-44.
33. Wabnitz AM, Turan TN. Symptomatic carotid artery stenosis: surgery, stenting, or medical therapy? Curr Treat Options Cardiovasc Med. 2017;19:62.
34. Muir KW, Santosh C. Imaging of acute stroke and transient ischaemic attack. J Neurol Neurosurg Psychiatry. 2005;76(suppl 3):iii19-iii28.
35. Cameron JI, Tsoi C, Marsella A.Optimizing stroke systems of care by enhancing transitions across care environments. Stroke. 2008;39:2637-2643.
36. Amarenco P, Bogousslavsky J, Caplan LR, et al. The ASCOD phenotyping of ischemic stroke (updated ASCO phenotyping). Cerebrovasc Dis. 2013;36:1-5.
37. Adams HP Jr, Bendixen BH, Kappelle LJ. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993;24:35-41.
38. Cacciatore A, Russo LS Jr. Lacunar infarction as an embolic complication of cardiac and arch angiography. Stroke. 1991;22:1603-1605.
39. Thomalla G, Simonsen CZ, Boutitie F, et al; WAKE-UP Investigators. MRI-guided thrombolysis for stroke with unknown time of onset. N Engl J Med. 2018;379:611-622.
40. Nogueira RG, Jadhav AP, Haussen DC, et al; DAWN Trial Investigators. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med. 2018;378:11-21.
1. Benjamin EJ, Virani SS, Callaway CW, et al; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2018 update: a report from the American Heart Association. Circulation. 2018;137:e67-e492.
2. Darves B. Collaboration key to post-stroke follow-up. ACP Internist. October 2009. https://acpinternist.org/archives/2009/10/stroke.htm. Accessed September 22, 2020.
3. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50e344-e418.
4. Sacco RL, Kasner SE, Broderick JP, et al; American Heart Association Stroke Council, Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular Radiology and Intervention; Council on Cardiovascular and Stroke Nursing; Council on Epidemiology and Prevention; Council on Peripheral Vascular Disease; Council on Nutrition, Physical Activity and Metabolism An updated definition of stroke for the 21st century: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:2064-2089.
5. Aroor S, Singh R, Goldstein LB. BE-FAST (Balance, Eyes, Face, Arm, Speech, Time): Reducing the proportion of strokes missed using the FAST mnemonic. 2017;48:479-481.
6. Kidwell CS, Starkman S, Eckstein M, et al. Identifying stroke in the field. Prospective validation of the Los Angeles prehospital stroke screen (LAPSS). Stroke. 2000;31:71-76.
7. Llanes JN, Kidwell CS, Starkman S, et al. The Los Angeles Motor Scale (LAMS): a new measure to characterize stroke severity in the field. Prehosp Emerg Care. 2004;8:46-50.
8. Pérez de la Ossa N, Carrera D, Gorchs M, et al. Design and validation of a prehospital stroke scale to predict large arterial occlusion: the rapid arterial occlusion evaluation scale. Stroke. 2014;45:87-91.
9. Katz BS, McMullan JT, Sucharew H, et al. Design and validation of a prehospital scale to predict stroke severity: Cincinnati Prehospital Stroke Severity Scale. Stroke. 2015;466:1508-1512.
10. Kummer BR, et al. External validation of the Cincinnati Prehospital Stroke Severity Scale. J Stroke Cerebrovasc Dis. 2016;25:1270-1274.
11. Beume L-A, Hieber M, Kaller CP, et al. Large vessel occlusion in acute stroke. Stroke. 2018;49:2323-2329.
12. Man S, Zhao X, Uchino K, et al. Comparison of acute ischemic stroke care and outcomes between comprehensive stroke centers and primary stroke centers in the United States. Circ Cardiovasc Qual Outcomes. 2018;11:e004512.
13. American Heart Association (Mission: Lifeline—Stroke). Emergency medical services acute stroke routing. 2020. www.heart.org/-/media/files/professional/quality-improvement/mission-lifeline/2_25_2020/ds15698-qi-ems-algorithm_update-2142020.pdf?la=en. Accessed October 8, 2020.
14. Glober NK, Sporer KA, Guluma KZ, et al. Acute stroke: current evidence-based recommendations for prehospital care. West J Emerg Med. 2016;17:104-128.
15. NIH stroke scale. Bethesda, MD: National Institute of Neurological Disorders and Stroke, National Institutes of Health. www.stroke.nih.gov/resources/scale.htm. Accessed October 10, 2020.
16. Smith EE, Kent DM, Bulsara KR, et al; . Accuracy of prediction instruments for diagnosing large vessel occlusion in individuals with suspected stroke: a systematic review for the 2018 guidelines for the early management of patients with acute ischemic stroke. Stroke. 2018;49:e111-e122.
17. Woo D, Broderick JP, Kothari RU, et al. Does the National Institutes of Health Stroke Scale favor left hemisphere strokes? NINDS t-PA Stroke Study Group. Stroke. 1999;30:2355-2359.
18. Adams HP Jr, Davis PH, Leira EC, et al. Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology. 1999;53:126-131.
19. Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007;38:1091-1096.
20. Birenbaum D, Bancroft LW, Felsberg GJ. Imaging in acute stroke. West J Emerg Med. 2011;12:67-76.
21. Salmela MB, Mortazavi S, Jagadeesan BD, et al. ACR Appropriateness Criteria® Cerebrovascular Disease. J Am Coll Radiol. 2017;14:S34-S61.
22. Hemphill JC 3rd, Greenberg SM, Anderson CS, et al; American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46:2032-60.
23. Hacke W, Kaste M, Fieschi C, et al. Intravenous thrombolysis with recombinant tissue plasminogen activator for acute hemispheric stroke. The European Cooperative Acute Stroke Study (ECASS). JAMA. 1995;274:1017-1025.
24. The Tissue plasminogen activator for acute ischemic stroke. N Engl J Med, 1995;333:1581-1587.
25. Albers GW, Clark WM, Madden KP, et al. ATLANTIS trial: results for patients treated within 3 hours of stroke onset. Alteplase Thrombolysis for Acute Noninterventional Therapy in Ischemic Stroke. Stroke. 2002;33:493-495.
26. Khan R, Nael K, Erly W. Acute stroke imaging: what clinicians need to know. Am J Med. 2013;126:379-386.
27. Latchaw RE, Alberts MJ, Lev MH, et al; . Recommendations for managing of acute ischemic stroke: a scientific statement from the American Heart Association. Stroke. 2009;40:3646-3678.
28. Vagal A, Meganathan K, Kleindorfer DO, et al. Increasing use of computed tomographic perfusion and computed tomographic angiograms in acute ischemic stroke from 2006 to 2010. Stroke. 2014;45:1029-1034.
29. Albers GW, Marks MP, Kemp S, et al; DEFUSE 3 Investigators. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med. 2018;378:708-718.
30. Demeestere J, Wouters A, Christensen S, et al. Review of perfusion imaging in acute ischemic stroke: from time to tissue. Stroke. 2020;51:1017-1024.
31. Chalela JA, Kidwell CS, Nentwich LM, et al, Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: a prospective comparison. Lancet. 2007;369:293-298.
32. Aoki J, Kimura K, Iguchi Y, et al. FLAIR can estimate the onset time in acute ischemic stroke patients. J Neurol Sci. 2010;293:39-44.
33. Wabnitz AM, Turan TN. Symptomatic carotid artery stenosis: surgery, stenting, or medical therapy? Curr Treat Options Cardiovasc Med. 2017;19:62.
34. Muir KW, Santosh C. Imaging of acute stroke and transient ischaemic attack. J Neurol Neurosurg Psychiatry. 2005;76(suppl 3):iii19-iii28.
35. Cameron JI, Tsoi C, Marsella A.Optimizing stroke systems of care by enhancing transitions across care environments. Stroke. 2008;39:2637-2643.
36. Amarenco P, Bogousslavsky J, Caplan LR, et al. The ASCOD phenotyping of ischemic stroke (updated ASCO phenotyping). Cerebrovasc Dis. 2013;36:1-5.
37. Adams HP Jr, Bendixen BH, Kappelle LJ. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993;24:35-41.
38. Cacciatore A, Russo LS Jr. Lacunar infarction as an embolic complication of cardiac and arch angiography. Stroke. 1991;22:1603-1605.
39. Thomalla G, Simonsen CZ, Boutitie F, et al; WAKE-UP Investigators. MRI-guided thrombolysis for stroke with unknown time of onset. N Engl J Med. 2018;379:611-622.
40. Nogueira RG, Jadhav AP, Haussen DC, et al; DAWN Trial Investigators. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med. 2018;378:11-21.
How to assess and relieve that perplexing rashless itch
Pruritus, defined as a sensation that induces a desire to scratch1 and classified as acute or chronic (lasting > 6 weeks),2 is one of the most common complaints among primary care patients: Approximately 1% of ambulatory visits in the United States are linked to pruritus.3
Chronic pruritus impairs quality of life; its impact has been compared to that of chronic pain.4 Treatment should therefore be instituted promptly. Although this condition might appear benign, chronic pruritus can be a symptom of a serious condition, as we describe here. When persistent pruritus is refractory to treatment, systemic causes should be fully explored.
In this article, we discuss the pathogenesis and management of pruritus without skin eruption in the adult nonpregnant patient. We also present practice recommendations to help you determine whether your patient’s pruritus is indicative of a serious systemic condition.
An incomplete understanding of the pathophysiology of pruritus
The pathophysiology of pruritus is not fully understood. It is generally recognized, however, that pruritus starts in the peripheral nerves located in the dermal–epidermal junction of the skin.5 The sensation is then transmitted along unmyelinated slow-conducting C fibers to the dorsal horn of the spinal cord.5,6 There are 2 types of C fibers that transmit the itch impulse6: A histamine-dependent type and a non-histamine-dependent type, which might explain why pruritus can be refractory to antihistamine treatment.6
Once the itch impulse has moved from the spinal cord, it travels along the spinothalamic tract up to the contralateral thalamus.1 From there, the impulse ascends to the cerebral cortex.1 In the cortex, the impulse triggers multiple areas of the brain, such as those responsible for sensation, motor function, reward, memory, and emotion.7
Several chemical mediators have been found to be peripheral and central inducers of pruritus: histamine, endogenous opioids, substance P, and serotonin.2 There are indications that certain receptors, such as mu-opioid receptors and kappa-opioid receptors, are key contributors to itch as well.2
A diverse etiology
The International Forum for the Study of Itch (IFSI) has established 6 main categories of causes of pruritus(TABLE 1)2:
- dermatologic
- systemic
- neurologic
- psychogenic
- mixed
- other.
Continue to: In this review...
In this review, we focus on the work-up and management of 3 of those categories: systemic, neurologic, and psychogenic causes of pruritus.
Systemic causes
Research has shown that 14% to 24% of patients who seek the care of a dermatologist for chronic itch without skin lesions have a systemic illness.8
Renal disease. Approximately 40% of patients with end-stage renal disease who are on hemodialysis or peritoneal dialysis have uremic pruritus.2 The itch is mostly generalized but can be pronounced on the back. For most patients, the itch is worse at night, causing a major impact on quality of life.6
Liver disease. In hepatic disease, there is often impairment in the secretion of bile, which can lead to cholestatic pruritus.2 This condition commonly affects the hands and feet first; later, it becomes generalized.2 Cholestatic pruritus can be elicited by tight-fitting clothing. Relief is not achieved by scratching.9 This type of itch effects 70% of patients with primary biliary cirrhosis and 15% of patients with hepatitis C infection.9
Hematologic disorders. Pruritus is a hallmark symptom of polycythemia rubra vera. Almost 50% of patients with this disorder report pruritus that occurs after exposure to water9; aquagenic pruritus can precede the formal diagnosis of polycythemia rubra vera by years.2 It has been speculated that platelet aggregation in this disorder leads to release of serotonin and histamine, which, in turn, causes itch.9
Continue to: Endocrine disorders
Endocrine disorders. Approximately 4% to 11% of patients with thyrotoxicosis have pruritus.1 It has been suggested that vasodilation, increased skin temperature, and a decreased itch threshold from untreated Graves disease might be inciting factors.
Malignancy. In generalized chronic pruritus without a known cause, strongly consider the likelihood of underlying malignancy8,10; for 10% of these patients, their chronic pruritus is a paraneoplastic sign. Paraneoplastic pruritus is characterized as an itch that predates clinical onset, or occurs early in the course, of a malignancy.9 The condition is most strongly linked to cancers of the liver, gallbladder, biliary tract, hematologic system, and skin.11
Chronic pruritus affects 30% of patients with Hodgkin lymphoma.9 General pruritus can precede this diagnosis by months, even years.1 In Hodgkin lymphoma patients who are in remission, a return of pruritic symptoms can be a harbinger of recurrence.9
Neurologic causes
A recent study found that 8% to 15% of patients referred to a dermatology clinic for chronic pruritus without skin eruption had underlying neurologic pathology.12 Although the specific mechanisms of neuropathic itch are still poorly understood, it has been theorized that the itch emanates from neuronal damage, which can come from peripheral or central nervous system lesions.9
Brachioradial pruritus. There are divergent theories about the etiology of brachioradial pruritus. One hypothesis is that the condition is caused by cervical nerve-root impingement at the level of C5-C8 that leads to nerve damage2; another is that chronic exposure to sunlight causes injury to peripheral cutaneous nerves.2 Brachioradial pruritus is localized to the dorsolateral forearm; it can also involve the neck, back, shoulder, upper arm, and chest, unilaterally and bilaterally. This pruritus can be intermittent and become worse upon exposure to sunlight.2
Continue to: Notalgia paresthetica
Notalgia paresthetica. This condition might also cause neuropathic pruritus as a consequence of nerve impingement. The itch of notalgia paresthesia is located on the skin, medial to the scapular border on the upper or mid-back.2 It has been postulated that the itch is caused by nerve entrapment of the posterior rami of spinal nerves arising from T2-T6.9 However, another theory suggests that the itch is caused by damage to peripheral nerves.9 The itch of notalgia paresthetica can wax and wane.2
Poststroke pruritus. Brain lesions, most often caused by stroke, can cause neuropathic itch. One of the best-known syndromes related to poststroke itch is Wallenberg syndrome (ischemia from a lateral medullary infarction), which typically presents with itch, thermalgic hypoesthesia of the face, cerebellar dysfunction, nausea, and vomiting.7
Shingles. More than one-half of patients who develop postherpetic neuralgia as a consequence of a herpes zoster infection also develop neuropathic pruritus.9 It is thought that postherpetic pruritus shares a comparable pathophysiology with postherpetic neuralgia, in which neurons involved in itch stimuli become damaged.7
Diabetes mellitus. Pruritus from diabetes can be classified as systemic or neuropathic. Diabetes is one of the most common causes of small-fiber polyneuropathy, which can cause neuropathic pruritus.13
Multiple sclerosis. Central nervous system lesions that affect sensory pathways can lead to neuropathic itch in multiple sclerosis. Patients can have severe episodes of generalized pruritus. It has been hypothesized that the neuropathic itch in multiple sclerosis is induced by activation of artificial synapses in demyelinated areas.2
Continue to: Psychogenic pruritus
Psychogenic pruritus
Chronic pruritus can be a comorbidity of psychiatric illness. A retrospective study found that pruritus occurs in 32% to 42% of psychiatric inpatients.14 Depression, anxiety, bipolar disorders, obsessive–compulsive disorders, somatoform disorders, psychosis, and substance abuse all have a strong link to psychogenic excoriation.15 Psychogenic excoriation, which can cause secondary skin lesions, occurs in psychiatric patients who excessively pick and scratch normal skin because they perceive an itch sensation or have a delusion of infestation.2 Affected skin can be marked by scattered crusted lesions (FIGURE) anywhere on the body that the patient can reach—most commonly, the extremities.2
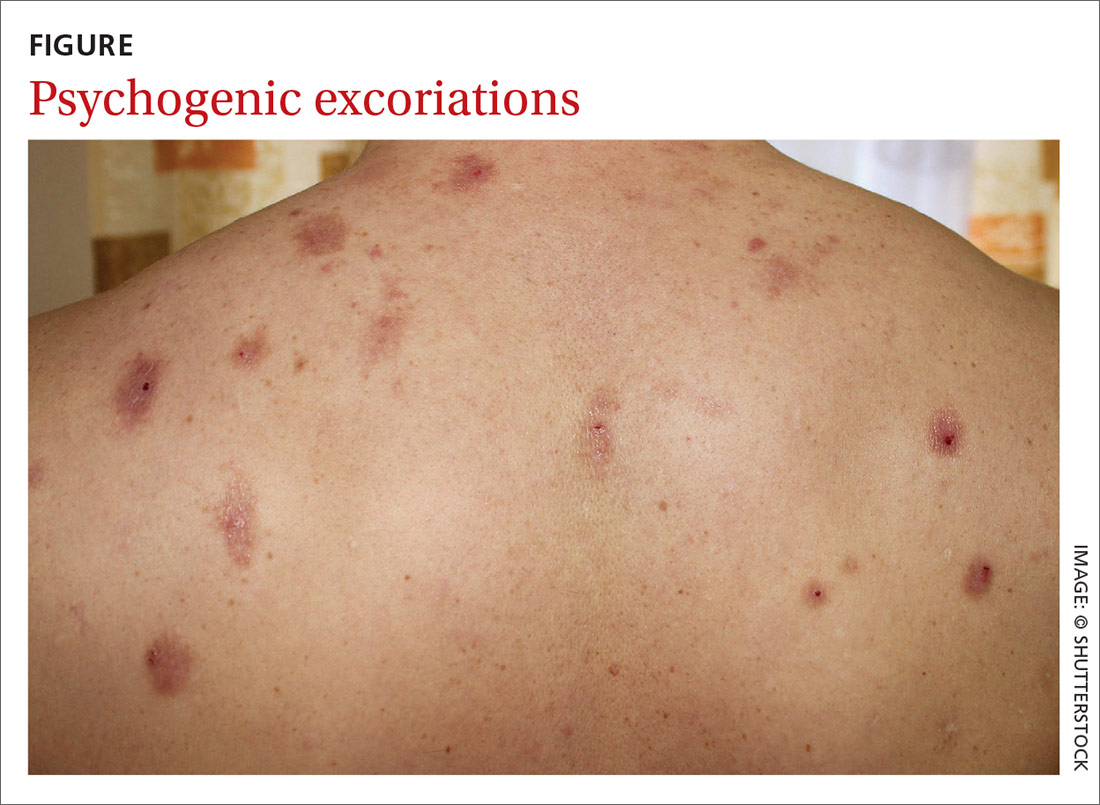
Delusion of infestation. Patients with a delusion of infestation have a strong belief that their body is infected by some kind of insect or microorganism.16 Before a diagnosis of delusion of infestation can be made, other organic causes must be excluded, including withdrawal from such substances as cocaine, amphetamines, and alcohol.16 Patients with a delusion of infestation can have, and maintain, a symptomatic response with continuing use of an atypical antipsychotic agent, including risperidone and olanzapine.17
Evaluation and diagnostic work-up
A thorough medical history, review of systems, medication review, social history, and family history are important when evaluating a patient with chronic pruritus.18 These items can be valuable in formulating a differential diagnosis, even before a physical examination.
Physical examination. The physical exam should include detailed inspection of the entire skin and hair18; such a comprehensive physical exam can determine whether the source of the itch is cutaneous.7 This, in turn, can help further narrow the differential diagnosis. It is crucial that the physical exam include palpation of the liver, spleen, lymph nodes, and thyroid for organomegaly,8 which could indicate a serious systemic condition, such as lymphoma.
The ice-pack sign—in which an ice pack is applied to the pruritic area, the patient experiences immediate relief of pruritus, and the itch returns soon after the ice pack is removed—is considered pathognomonic for brachioradial pruritus.19
Continue to: Chronic pruritus with abnormal findings...
Chronic pruritus with abnormal findings on the physical exam should prompt an initial work-up.18 Also consider an initial work-up for a patient with chronic pruritus whose symptom has not been relieved with conservative treatment.18
Laboratory testing. The initial laboratory work-up could include any of the following evaluations: complete blood count, measurement of thyroid-stimulating hormone, comprehensive metabolic panel (liver function, renal function, and the serum glucose level) and the erythrocyte sedimentation rate (TABLE 2).18 If warranted by the evaluation and physical exam, blood work can also include serologic studies for human immunodeficiency virus infection and hepatitis.17
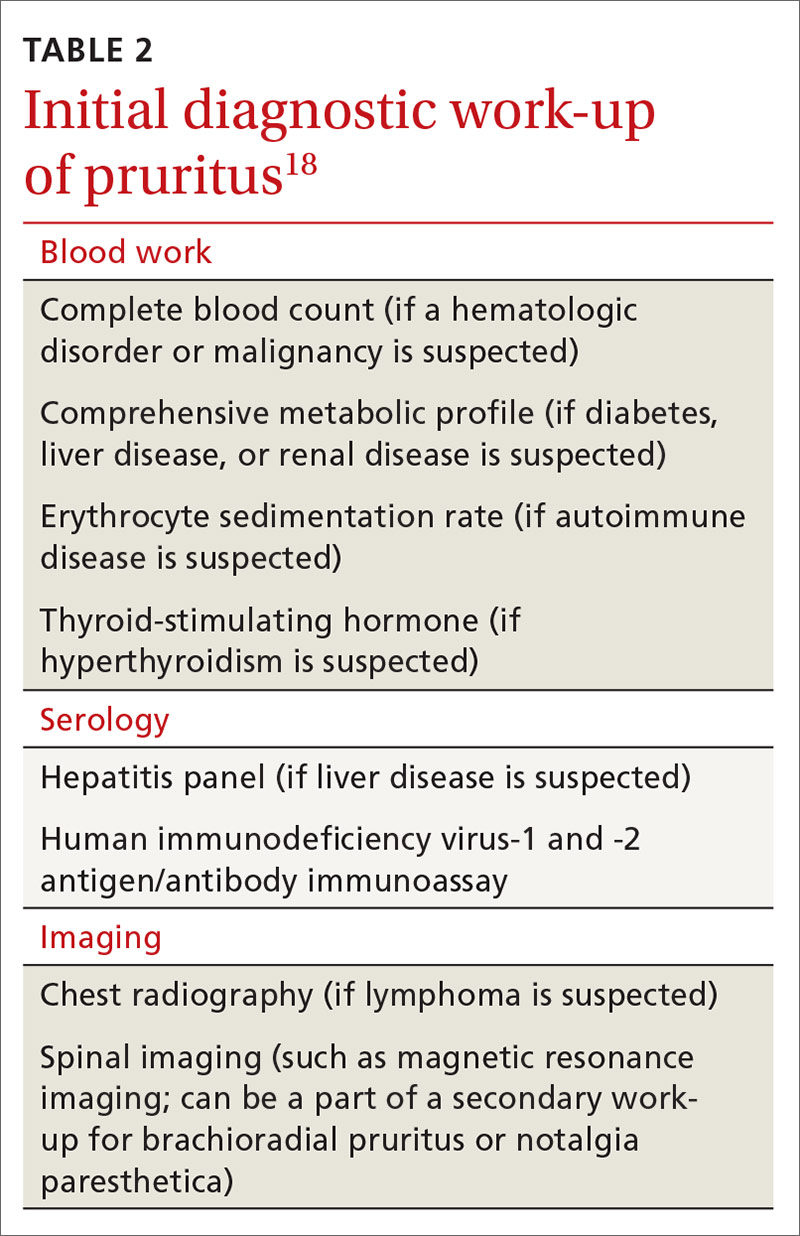
Imaging. Chest radiography should be performed if there is suspicion of malignancy, such as lymphoma.7 Although brachioradial pruritus and notalgia paresthetica have been postulated to be caused by impingement of spinal nerves, obtaining spinal imaging, such as magnetic resonance imaging, as part of the initial work-up is not recommended; because spinal images might not show evidence of spinal disease, obtaining spinal imaging is not a requirement before treating brachioradial pruritus and notalgia paresthetica. Do consider spinal imaging, however, for patients in whom brachioradial pruritus or notalgia paresthetica is suspected and conservative treatment has not produced a response.
Treatment: Nondrug approaches, topicals, systemic agents
Start conservatively. Treatment of pruritus should begin with behavior modification and nonpharmacotherapeutic options (TABLE 38). Educate the patient that scratching might cause secondary skin lesions; empowering them with that knowledge is sometimes enough to help break the scratching cycling—especially if the patient combines behavior modification with proper skin hydration with an emollient. To prevent secondary skin lesions through involuntary scratching, consider recommending that lesions be covered with an occlusive dressing or protective clothing.13

Stress has been shown to make chronic itch worse; therefore, stress-reduction activities, such as exercise, meditation, and yoga, might be helpful.20 For patients in whom pruritus has a psychological component, referral to a psychiatrist or psychologist might be therapeutic.
Continue to: When a patient complains...
When a patient complains of severe pruritus at first presentation, consider pharmacotherapy in conjunction with nonpharmacotherapeutic options. Several of the more effective topical therapies for pruritusa are listed in TABLE 4.20 Well-known systemic agents for this purpose are reviewed below and listed in TABLE 5.7
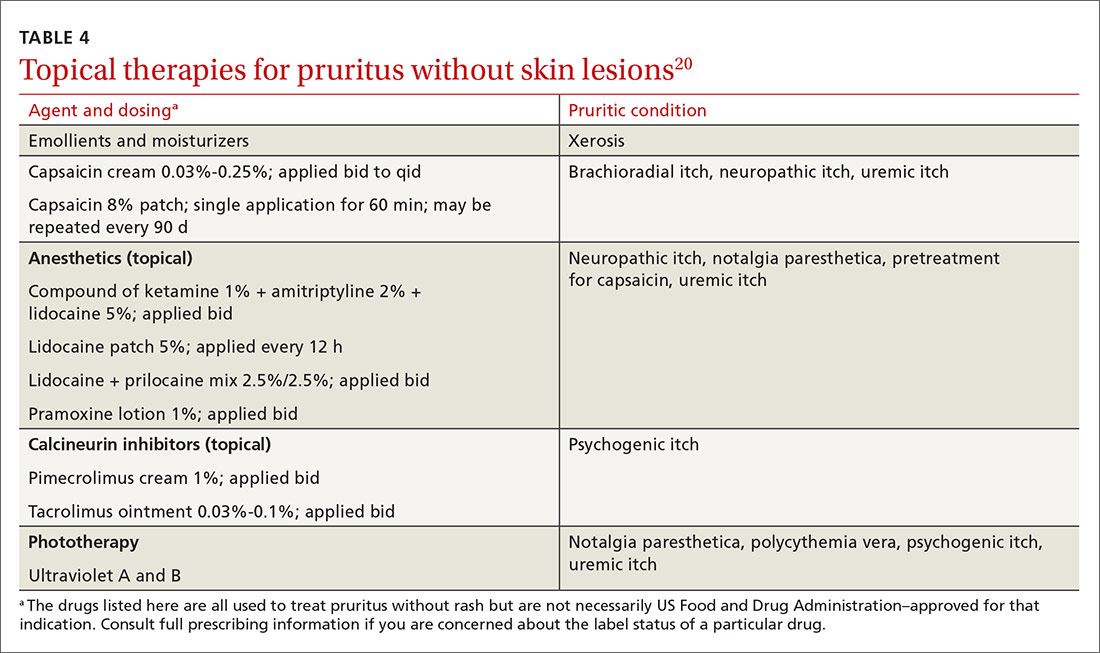
Systemic treatment
Antihistamines. A staple in the treatment of pruritus for many years, antihistamines are not effective for all causes; however, they are effective in treating paraneoplastic pruritus.20 First-generation antihistamines, with their sedating effect, can be useful for patients who experience generalized pruritus at night.20

Anticonvulsants. Gabapentin and pregabalin are analogs of the neurotransmitter gamma-aminobutyric acid.20 This drug class is helpful in neuropathic pruritus specifically caused by impingements, such as brachioradial pruritus and notalgia paresthetica.20 In addition, of all systemic therapies used to treat uremic pruritus, gabapentin has, in clinical trials, most consistently been found effective for uremic pruritus.6 (Note: Use renal dosing of gabapentin in patients with renal failure.)
Antidepressants. Selective serotonin reuptake inhibitors (SSRIs; eg, fluvoxamine, paroxetine, and sertraline) might cause itch to subside by increasing the serotonin level, which, in turn, works to decrease inflammatory substances that cause itch.7 SSRIs have been used to treat patients with psychogenic pruritus, cholestatic pruritus, and paraneoplastic pruritus.7
Tricyclic antidepressants (eg, amitriptyline and doxepin) lessen the itch by antagonizing histamine receptors and through anticholinergic mechanisms. Tricyclics are best used in the treatment of psychogenic and nocturnal itch.7
Continue to: Mirtazapine...
Mirtazapine, a tetracyclic antidepressant, works in patients with uremic pruritus, psychogenic pruritus, cholestatic pruritus, and paraneoplastic pruritus.1
Substance P antagonist. Aprepitant, a neurokinin receptor I antagonist, is a newer agent that inhibits binding of the itch mediator substance P to the neurokinin receptor. The drug has been found helpful in patients with drug-induced, paraneoplastic, and brachioradial pruritus.7
Opioid-receptor agents. Naltrexone, as a mu opioid-receptor antagonist, has shown promise as a treatment for uremic pruritus and cholestatic pruritus. Nalfurafine, a kappa opioid-receptor agonist, is emerging as a possible therapy for uremic pruritus.7
Bile-acid sequestrants. A few small studies have shown that treatment with a bile-acid sequestrant, such as cholestyramine and ursodiol, induces moderate improvement in symptoms in patients with cholestatic pruritus.21
CORRESPONDENCE
Matasha Russell, MD, Department of Family and Community Medicine, The University of Texas Health Science Center at Houston, McGovern Medical School, 6431 Fannin Street, JJL 324, Houston, TX 77030; Matasha.L.Russell@uth.tmc.edu.
1. Tarikci N, E, S, et al. Pruritus in systemic diseases: a review of etiological factors and new treatment modalities. ScientificWorldJournal. 2015;2015:803752.
2. Yosipovitch G, Bernhard JD. Clinical practice. Chronic pruritus. N Engl J Med. 2013;368:1625-1634.
3. Silverberg JI, Kantor RW, Dalal P. A comprehensive conceptual model of the experience of chronic itch in adults. Am J Clin Dermatol. 2018;19:759-769.
4. Matterne U, Apfelbacher CJ, Vogelgsang L, et al. Incidence and determinants of chronic pruritus: a population based cohort study. Acta Derm Venereol. 2013;93:532-537.
5. Moses S. Pruritus. Am Fam Physician. 2003;68:1135-1142.
6. Combs SA, Teixeira JP, Germain MJ. Pruritus in kidney disease. Semin Nephrol. 2015;35:383-391.
7. Shevchenko A, Valdes-Rodriguez R, Yosipovitch G. Causes, pathophysiology, and treatment of pruritus in the mature patient. Clin Dermatol. 2018;36:140-151.
8. Reamy BV, Bunt C. A diagnostic approach to pruritus. Am Fam Physician. 2011;84:195-202.
9. M. Current concepts of pathophysiology, epidemiology and classification of pruritus. Srp Arh Celok Lek. 2014;142:106-112.
10. Fett N, Haynes K, Propert KJ, et al. Five-year malignancy incidence in patients with chronic pruritus: a population-based cohort study aimed at limiting unnecessary screening practices. J Am Acad Dermatol. 2014;70:651-658.
11. Larson VA, Tang O, S, et al. Association between itch and cancer in 16,925 patients with pruritus: experience at a tertiary care center. J Am Acad Dermatol. 2019;80:931-937.
12. Rosen JD, Fostini AC, Chan YH, et al. Cross-sectional study of clinical distinctions between neuropathic and inflammatory pruritus. J Am Acad Dermatol. 2018;79:1143-1144.
13. Oaklander AL. Neuropathic itch. Semin Cutan Med Surg. 2011;30:87-92.
14. Ferm I, Sterner M, Wallengren J. Somatic and psychiatric comorbidity in patients with chronic pruritus. Acta Derm Venereol. 2010;90:395-400.
15. Jafferany M, Davari ME. Itch and psyche: psychiatric aspects of pruritus. Int J Dermatol. 2019;58:3-23.
16. Koo J, Lebwohl A. Psychodermatology: the mind and skin connection. Am Fam Physician. 2001;64:1873-1878.
17. Bewley AP, Lepping P, Freudenmann RW, et al. Delusional parasitosis: time to call it delusional infestation. Br J Dermatol.2010;163:1-2.
18. Clerc C-J, Misery L. A literature review of senile pruritus: from diagnosis to treatment. Acta Derm Venereol. 2017;97:433-440.
19. Bernhard JD, Bordeaux JS. Medical pearl: the ice-pack sign in brachioradial pruritus. J Am Acad Dermatol. 2005;52:1073.
20. Sanders KM, Nattkemper LA, Yosipovitch G. Advances in understanding itching and scratching: a new era of targeted treatments [version 1]. F1000Res. 2016;5 F1000 Faculty Rev–2042.
21. Hegade VS, Kendrick SFW, Dobbins RL, et al. Effect of ileal bile acid transporter inhibitor GSK2330672 on pruritus in primary biliary cholangitis: a double-blind, randomised, placebo-controlled, crossover, phase 2a study. Lancet. 2017;389:1114-1123.
Pruritus, defined as a sensation that induces a desire to scratch1 and classified as acute or chronic (lasting > 6 weeks),2 is one of the most common complaints among primary care patients: Approximately 1% of ambulatory visits in the United States are linked to pruritus.3
Chronic pruritus impairs quality of life; its impact has been compared to that of chronic pain.4 Treatment should therefore be instituted promptly. Although this condition might appear benign, chronic pruritus can be a symptom of a serious condition, as we describe here. When persistent pruritus is refractory to treatment, systemic causes should be fully explored.
In this article, we discuss the pathogenesis and management of pruritus without skin eruption in the adult nonpregnant patient. We also present practice recommendations to help you determine whether your patient’s pruritus is indicative of a serious systemic condition.

An incomplete understanding of the pathophysiology of pruritus
The pathophysiology of pruritus is not fully understood. It is generally recognized, however, that pruritus starts in the peripheral nerves located in the dermal–epidermal junction of the skin.5 The sensation is then transmitted along unmyelinated slow-conducting C fibers to the dorsal horn of the spinal cord.5,6 There are 2 types of C fibers that transmit the itch impulse6: A histamine-dependent type and a non-histamine-dependent type, which might explain why pruritus can be refractory to antihistamine treatment.6
Once the itch impulse has moved from the spinal cord, it travels along the spinothalamic tract up to the contralateral thalamus.1 From there, the impulse ascends to the cerebral cortex.1 In the cortex, the impulse triggers multiple areas of the brain, such as those responsible for sensation, motor function, reward, memory, and emotion.7
Several chemical mediators have been found to be peripheral and central inducers of pruritus: histamine, endogenous opioids, substance P, and serotonin.2 There are indications that certain receptors, such as mu-opioid receptors and kappa-opioid receptors, are key contributors to itch as well.2

A diverse etiology
The International Forum for the Study of Itch (IFSI) has established 6 main categories of causes of pruritus(TABLE 1)2:
- dermatologic
- systemic
- neurologic
- psychogenic
- mixed
- other.
Continue to: In this review...
In this review, we focus on the work-up and management of 3 of those categories: systemic, neurologic, and psychogenic causes of pruritus.
Systemic causes
Research has shown that 14% to 24% of patients who seek the care of a dermatologist for chronic itch without skin lesions have a systemic illness.8
Renal disease. Approximately 40% of patients with end-stage renal disease who are on hemodialysis or peritoneal dialysis have uremic pruritus.2 The itch is mostly generalized but can be pronounced on the back. For most patients, the itch is worse at night, causing a major impact on quality of life.6
Liver disease. In hepatic disease, there is often impairment in the secretion of bile, which can lead to cholestatic pruritus.2 This condition commonly affects the hands and feet first; later, it becomes generalized.2 Cholestatic pruritus can be elicited by tight-fitting clothing. Relief is not achieved by scratching.9 This type of itch effects 70% of patients with primary biliary cirrhosis and 15% of patients with hepatitis C infection.9
Hematologic disorders. Pruritus is a hallmark symptom of polycythemia rubra vera. Almost 50% of patients with this disorder report pruritus that occurs after exposure to water9; aquagenic pruritus can precede the formal diagnosis of polycythemia rubra vera by years.2 It has been speculated that platelet aggregation in this disorder leads to release of serotonin and histamine, which, in turn, causes itch.9
Continue to: Endocrine disorders
Endocrine disorders. Approximately 4% to 11% of patients with thyrotoxicosis have pruritus.1 It has been suggested that vasodilation, increased skin temperature, and a decreased itch threshold from untreated Graves disease might be inciting factors.
Malignancy. In generalized chronic pruritus without a known cause, strongly consider the likelihood of underlying malignancy8,10; for 10% of these patients, their chronic pruritus is a paraneoplastic sign. Paraneoplastic pruritus is characterized as an itch that predates clinical onset, or occurs early in the course, of a malignancy.9 The condition is most strongly linked to cancers of the liver, gallbladder, biliary tract, hematologic system, and skin.11
Chronic pruritus affects 30% of patients with Hodgkin lymphoma.9 General pruritus can precede this diagnosis by months, even years.1 In Hodgkin lymphoma patients who are in remission, a return of pruritic symptoms can be a harbinger of recurrence.9
Neurologic causes
A recent study found that 8% to 15% of patients referred to a dermatology clinic for chronic pruritus without skin eruption had underlying neurologic pathology.12 Although the specific mechanisms of neuropathic itch are still poorly understood, it has been theorized that the itch emanates from neuronal damage, which can come from peripheral or central nervous system lesions.9
Brachioradial pruritus. There are divergent theories about the etiology of brachioradial pruritus. One hypothesis is that the condition is caused by cervical nerve-root impingement at the level of C5-C8 that leads to nerve damage2; another is that chronic exposure to sunlight causes injury to peripheral cutaneous nerves.2 Brachioradial pruritus is localized to the dorsolateral forearm; it can also involve the neck, back, shoulder, upper arm, and chest, unilaterally and bilaterally. This pruritus can be intermittent and become worse upon exposure to sunlight.2
Continue to: Notalgia paresthetica
Notalgia paresthetica. This condition might also cause neuropathic pruritus as a consequence of nerve impingement. The itch of notalgia paresthesia is located on the skin, medial to the scapular border on the upper or mid-back.2 It has been postulated that the itch is caused by nerve entrapment of the posterior rami of spinal nerves arising from T2-T6.9 However, another theory suggests that the itch is caused by damage to peripheral nerves.9 The itch of notalgia paresthetica can wax and wane.2
Poststroke pruritus. Brain lesions, most often caused by stroke, can cause neuropathic itch. One of the best-known syndromes related to poststroke itch is Wallenberg syndrome (ischemia from a lateral medullary infarction), which typically presents with itch, thermalgic hypoesthesia of the face, cerebellar dysfunction, nausea, and vomiting.7
Shingles. More than one-half of patients who develop postherpetic neuralgia as a consequence of a herpes zoster infection also develop neuropathic pruritus.9 It is thought that postherpetic pruritus shares a comparable pathophysiology with postherpetic neuralgia, in which neurons involved in itch stimuli become damaged.7
Diabetes mellitus. Pruritus from diabetes can be classified as systemic or neuropathic. Diabetes is one of the most common causes of small-fiber polyneuropathy, which can cause neuropathic pruritus.13
Multiple sclerosis. Central nervous system lesions that affect sensory pathways can lead to neuropathic itch in multiple sclerosis. Patients can have severe episodes of generalized pruritus. It has been hypothesized that the neuropathic itch in multiple sclerosis is induced by activation of artificial synapses in demyelinated areas.2
Continue to: Psychogenic pruritus
Psychogenic pruritus
Chronic pruritus can be a comorbidity of psychiatric illness. A retrospective study found that pruritus occurs in 32% to 42% of psychiatric inpatients.14 Depression, anxiety, bipolar disorders, obsessive–compulsive disorders, somatoform disorders, psychosis, and substance abuse all have a strong link to psychogenic excoriation.15 Psychogenic excoriation, which can cause secondary skin lesions, occurs in psychiatric patients who excessively pick and scratch normal skin because they perceive an itch sensation or have a delusion of infestation.2 Affected skin can be marked by scattered crusted lesions (FIGURE) anywhere on the body that the patient can reach—most commonly, the extremities.2

Delusion of infestation. Patients with a delusion of infestation have a strong belief that their body is infected by some kind of insect or microorganism.16 Before a diagnosis of delusion of infestation can be made, other organic causes must be excluded, including withdrawal from such substances as cocaine, amphetamines, and alcohol.16 Patients with a delusion of infestation can have, and maintain, a symptomatic response with continuing use of an atypical antipsychotic agent, including risperidone and olanzapine.17
Evaluation and diagnostic work-up
A thorough medical history, review of systems, medication review, social history, and family history are important when evaluating a patient with chronic pruritus.18 These items can be valuable in formulating a differential diagnosis, even before a physical examination.
Physical examination. The physical exam should include detailed inspection of the entire skin and hair18; such a comprehensive physical exam can determine whether the source of the itch is cutaneous.7 This, in turn, can help further narrow the differential diagnosis. It is crucial that the physical exam include palpation of the liver, spleen, lymph nodes, and thyroid for organomegaly,8 which could indicate a serious systemic condition, such as lymphoma.
The ice-pack sign—in which an ice pack is applied to the pruritic area, the patient experiences immediate relief of pruritus, and the itch returns soon after the ice pack is removed—is considered pathognomonic for brachioradial pruritus.19
Continue to: Chronic pruritus with abnormal findings...
Chronic pruritus with abnormal findings on the physical exam should prompt an initial work-up.18 Also consider an initial work-up for a patient with chronic pruritus whose symptom has not been relieved with conservative treatment.18
Laboratory testing. The initial laboratory work-up could include any of the following evaluations: complete blood count, measurement of thyroid-stimulating hormone, comprehensive metabolic panel (liver function, renal function, and the serum glucose level) and the erythrocyte sedimentation rate (TABLE 2).18 If warranted by the evaluation and physical exam, blood work can also include serologic studies for human immunodeficiency virus infection and hepatitis.17

Imaging. Chest radiography should be performed if there is suspicion of malignancy, such as lymphoma.7 Although brachioradial pruritus and notalgia paresthetica have been postulated to be caused by impingement of spinal nerves, obtaining spinal imaging, such as magnetic resonance imaging, as part of the initial work-up is not recommended; because spinal images might not show evidence of spinal disease, obtaining spinal imaging is not a requirement before treating brachioradial pruritus and notalgia paresthetica. Do consider spinal imaging, however, for patients in whom brachioradial pruritus or notalgia paresthetica is suspected and conservative treatment has not produced a response.
Treatment: Nondrug approaches, topicals, systemic agents
Start conservatively. Treatment of pruritus should begin with behavior modification and nonpharmacotherapeutic options (TABLE 38). Educate the patient that scratching might cause secondary skin lesions; empowering them with that knowledge is sometimes enough to help break the scratching cycling—especially if the patient combines behavior modification with proper skin hydration with an emollient. To prevent secondary skin lesions through involuntary scratching, consider recommending that lesions be covered with an occlusive dressing or protective clothing.13

Stress has been shown to make chronic itch worse; therefore, stress-reduction activities, such as exercise, meditation, and yoga, might be helpful.20 For patients in whom pruritus has a psychological component, referral to a psychiatrist or psychologist might be therapeutic.
Continue to: When a patient complains...
When a patient complains of severe pruritus at first presentation, consider pharmacotherapy in conjunction with nonpharmacotherapeutic options. Several of the more effective topical therapies for pruritusa are listed in TABLE 4.20 Well-known systemic agents for this purpose are reviewed below and listed in TABLE 5.7

Systemic treatment
Antihistamines. A staple in the treatment of pruritus for many years, antihistamines are not effective for all causes; however, they are effective in treating paraneoplastic pruritus.20 First-generation antihistamines, with their sedating effect, can be useful for patients who experience generalized pruritus at night.20

Anticonvulsants. Gabapentin and pregabalin are analogs of the neurotransmitter gamma-aminobutyric acid.20 This drug class is helpful in neuropathic pruritus specifically caused by impingements, such as brachioradial pruritus and notalgia paresthetica.20 In addition, of all systemic therapies used to treat uremic pruritus, gabapentin has, in clinical trials, most consistently been found effective for uremic pruritus.6 (Note: Use renal dosing of gabapentin in patients with renal failure.)
Antidepressants. Selective serotonin reuptake inhibitors (SSRIs; eg, fluvoxamine, paroxetine, and sertraline) might cause itch to subside by increasing the serotonin level, which, in turn, works to decrease inflammatory substances that cause itch.7 SSRIs have been used to treat patients with psychogenic pruritus, cholestatic pruritus, and paraneoplastic pruritus.7
Tricyclic antidepressants (eg, amitriptyline and doxepin) lessen the itch by antagonizing histamine receptors and through anticholinergic mechanisms. Tricyclics are best used in the treatment of psychogenic and nocturnal itch.7
Continue to: Mirtazapine...
Mirtazapine, a tetracyclic antidepressant, works in patients with uremic pruritus, psychogenic pruritus, cholestatic pruritus, and paraneoplastic pruritus.1
Substance P antagonist. Aprepitant, a neurokinin receptor I antagonist, is a newer agent that inhibits binding of the itch mediator substance P to the neurokinin receptor. The drug has been found helpful in patients with drug-induced, paraneoplastic, and brachioradial pruritus.7
Opioid-receptor agents. Naltrexone, as a mu opioid-receptor antagonist, has shown promise as a treatment for uremic pruritus and cholestatic pruritus. Nalfurafine, a kappa opioid-receptor agonist, is emerging as a possible therapy for uremic pruritus.7
Bile-acid sequestrants. A few small studies have shown that treatment with a bile-acid sequestrant, such as cholestyramine and ursodiol, induces moderate improvement in symptoms in patients with cholestatic pruritus.21
CORRESPONDENCE
Matasha Russell, MD, Department of Family and Community Medicine, The University of Texas Health Science Center at Houston, McGovern Medical School, 6431 Fannin Street, JJL 324, Houston, TX 77030; Matasha.L.Russell@uth.tmc.edu.
Pruritus, defined as a sensation that induces a desire to scratch1 and classified as acute or chronic (lasting > 6 weeks),2 is one of the most common complaints among primary care patients: Approximately 1% of ambulatory visits in the United States are linked to pruritus.3
Chronic pruritus impairs quality of life; its impact has been compared to that of chronic pain.4 Treatment should therefore be instituted promptly. Although this condition might appear benign, chronic pruritus can be a symptom of a serious condition, as we describe here. When persistent pruritus is refractory to treatment, systemic causes should be fully explored.
In this article, we discuss the pathogenesis and management of pruritus without skin eruption in the adult nonpregnant patient. We also present practice recommendations to help you determine whether your patient’s pruritus is indicative of a serious systemic condition.

An incomplete understanding of the pathophysiology of pruritus
The pathophysiology of pruritus is not fully understood. It is generally recognized, however, that pruritus starts in the peripheral nerves located in the dermal–epidermal junction of the skin.5 The sensation is then transmitted along unmyelinated slow-conducting C fibers to the dorsal horn of the spinal cord.5,6 There are 2 types of C fibers that transmit the itch impulse6: A histamine-dependent type and a non-histamine-dependent type, which might explain why pruritus can be refractory to antihistamine treatment.6
Once the itch impulse has moved from the spinal cord, it travels along the spinothalamic tract up to the contralateral thalamus.1 From there, the impulse ascends to the cerebral cortex.1 In the cortex, the impulse triggers multiple areas of the brain, such as those responsible for sensation, motor function, reward, memory, and emotion.7
Several chemical mediators have been found to be peripheral and central inducers of pruritus: histamine, endogenous opioids, substance P, and serotonin.2 There are indications that certain receptors, such as mu-opioid receptors and kappa-opioid receptors, are key contributors to itch as well.2

A diverse etiology
The International Forum for the Study of Itch (IFSI) has established 6 main categories of causes of pruritus(TABLE 1)2:
- dermatologic
- systemic
- neurologic
- psychogenic
- mixed
- other.
Continue to: In this review...
In this review, we focus on the work-up and management of 3 of those categories: systemic, neurologic, and psychogenic causes of pruritus.
Systemic causes
Research has shown that 14% to 24% of patients who seek the care of a dermatologist for chronic itch without skin lesions have a systemic illness.8
Renal disease. Approximately 40% of patients with end-stage renal disease who are on hemodialysis or peritoneal dialysis have uremic pruritus.2 The itch is mostly generalized but can be pronounced on the back. For most patients, the itch is worse at night, causing a major impact on quality of life.6
Liver disease. In hepatic disease, there is often impairment in the secretion of bile, which can lead to cholestatic pruritus.2 This condition commonly affects the hands and feet first; later, it becomes generalized.2 Cholestatic pruritus can be elicited by tight-fitting clothing. Relief is not achieved by scratching.9 This type of itch effects 70% of patients with primary biliary cirrhosis and 15% of patients with hepatitis C infection.9
Hematologic disorders. Pruritus is a hallmark symptom of polycythemia rubra vera. Almost 50% of patients with this disorder report pruritus that occurs after exposure to water9; aquagenic pruritus can precede the formal diagnosis of polycythemia rubra vera by years.2 It has been speculated that platelet aggregation in this disorder leads to release of serotonin and histamine, which, in turn, causes itch.9
Continue to: Endocrine disorders
Endocrine disorders. Approximately 4% to 11% of patients with thyrotoxicosis have pruritus.1 It has been suggested that vasodilation, increased skin temperature, and a decreased itch threshold from untreated Graves disease might be inciting factors.
Malignancy. In generalized chronic pruritus without a known cause, strongly consider the likelihood of underlying malignancy8,10; for 10% of these patients, their chronic pruritus is a paraneoplastic sign. Paraneoplastic pruritus is characterized as an itch that predates clinical onset, or occurs early in the course, of a malignancy.9 The condition is most strongly linked to cancers of the liver, gallbladder, biliary tract, hematologic system, and skin.11
Chronic pruritus affects 30% of patients with Hodgkin lymphoma.9 General pruritus can precede this diagnosis by months, even years.1 In Hodgkin lymphoma patients who are in remission, a return of pruritic symptoms can be a harbinger of recurrence.9
Neurologic causes
A recent study found that 8% to 15% of patients referred to a dermatology clinic for chronic pruritus without skin eruption had underlying neurologic pathology.12 Although the specific mechanisms of neuropathic itch are still poorly understood, it has been theorized that the itch emanates from neuronal damage, which can come from peripheral or central nervous system lesions.9
Brachioradial pruritus. There are divergent theories about the etiology of brachioradial pruritus. One hypothesis is that the condition is caused by cervical nerve-root impingement at the level of C5-C8 that leads to nerve damage2; another is that chronic exposure to sunlight causes injury to peripheral cutaneous nerves.2 Brachioradial pruritus is localized to the dorsolateral forearm; it can also involve the neck, back, shoulder, upper arm, and chest, unilaterally and bilaterally. This pruritus can be intermittent and become worse upon exposure to sunlight.2
Continue to: Notalgia paresthetica
Notalgia paresthetica. This condition might also cause neuropathic pruritus as a consequence of nerve impingement. The itch of notalgia paresthesia is located on the skin, medial to the scapular border on the upper or mid-back.2 It has been postulated that the itch is caused by nerve entrapment of the posterior rami of spinal nerves arising from T2-T6.9 However, another theory suggests that the itch is caused by damage to peripheral nerves.9 The itch of notalgia paresthetica can wax and wane.2
Poststroke pruritus. Brain lesions, most often caused by stroke, can cause neuropathic itch. One of the best-known syndromes related to poststroke itch is Wallenberg syndrome (ischemia from a lateral medullary infarction), which typically presents with itch, thermalgic hypoesthesia of the face, cerebellar dysfunction, nausea, and vomiting.7
Shingles. More than one-half of patients who develop postherpetic neuralgia as a consequence of a herpes zoster infection also develop neuropathic pruritus.9 It is thought that postherpetic pruritus shares a comparable pathophysiology with postherpetic neuralgia, in which neurons involved in itch stimuli become damaged.7
Diabetes mellitus. Pruritus from diabetes can be classified as systemic or neuropathic. Diabetes is one of the most common causes of small-fiber polyneuropathy, which can cause neuropathic pruritus.13
Multiple sclerosis. Central nervous system lesions that affect sensory pathways can lead to neuropathic itch in multiple sclerosis. Patients can have severe episodes of generalized pruritus. It has been hypothesized that the neuropathic itch in multiple sclerosis is induced by activation of artificial synapses in demyelinated areas.2
Continue to: Psychogenic pruritus
Psychogenic pruritus
Chronic pruritus can be a comorbidity of psychiatric illness. A retrospective study found that pruritus occurs in 32% to 42% of psychiatric inpatients.14 Depression, anxiety, bipolar disorders, obsessive–compulsive disorders, somatoform disorders, psychosis, and substance abuse all have a strong link to psychogenic excoriation.15 Psychogenic excoriation, which can cause secondary skin lesions, occurs in psychiatric patients who excessively pick and scratch normal skin because they perceive an itch sensation or have a delusion of infestation.2 Affected skin can be marked by scattered crusted lesions (FIGURE) anywhere on the body that the patient can reach—most commonly, the extremities.2

Delusion of infestation. Patients with a delusion of infestation have a strong belief that their body is infected by some kind of insect or microorganism.16 Before a diagnosis of delusion of infestation can be made, other organic causes must be excluded, including withdrawal from such substances as cocaine, amphetamines, and alcohol.16 Patients with a delusion of infestation can have, and maintain, a symptomatic response with continuing use of an atypical antipsychotic agent, including risperidone and olanzapine.17
Evaluation and diagnostic work-up
A thorough medical history, review of systems, medication review, social history, and family history are important when evaluating a patient with chronic pruritus.18 These items can be valuable in formulating a differential diagnosis, even before a physical examination.
Physical examination. The physical exam should include detailed inspection of the entire skin and hair18; such a comprehensive physical exam can determine whether the source of the itch is cutaneous.7 This, in turn, can help further narrow the differential diagnosis. It is crucial that the physical exam include palpation of the liver, spleen, lymph nodes, and thyroid for organomegaly,8 which could indicate a serious systemic condition, such as lymphoma.
The ice-pack sign—in which an ice pack is applied to the pruritic area, the patient experiences immediate relief of pruritus, and the itch returns soon after the ice pack is removed—is considered pathognomonic for brachioradial pruritus.19
Continue to: Chronic pruritus with abnormal findings...
Chronic pruritus with abnormal findings on the physical exam should prompt an initial work-up.18 Also consider an initial work-up for a patient with chronic pruritus whose symptom has not been relieved with conservative treatment.18
Laboratory testing. The initial laboratory work-up could include any of the following evaluations: complete blood count, measurement of thyroid-stimulating hormone, comprehensive metabolic panel (liver function, renal function, and the serum glucose level) and the erythrocyte sedimentation rate (TABLE 2).18 If warranted by the evaluation and physical exam, blood work can also include serologic studies for human immunodeficiency virus infection and hepatitis.17

Imaging. Chest radiography should be performed if there is suspicion of malignancy, such as lymphoma.7 Although brachioradial pruritus and notalgia paresthetica have been postulated to be caused by impingement of spinal nerves, obtaining spinal imaging, such as magnetic resonance imaging, as part of the initial work-up is not recommended; because spinal images might not show evidence of spinal disease, obtaining spinal imaging is not a requirement before treating brachioradial pruritus and notalgia paresthetica. Do consider spinal imaging, however, for patients in whom brachioradial pruritus or notalgia paresthetica is suspected and conservative treatment has not produced a response.
Treatment: Nondrug approaches, topicals, systemic agents
Start conservatively. Treatment of pruritus should begin with behavior modification and nonpharmacotherapeutic options (TABLE 38). Educate the patient that scratching might cause secondary skin lesions; empowering them with that knowledge is sometimes enough to help break the scratching cycling—especially if the patient combines behavior modification with proper skin hydration with an emollient. To prevent secondary skin lesions through involuntary scratching, consider recommending that lesions be covered with an occlusive dressing or protective clothing.13

Stress has been shown to make chronic itch worse; therefore, stress-reduction activities, such as exercise, meditation, and yoga, might be helpful.20 For patients in whom pruritus has a psychological component, referral to a psychiatrist or psychologist might be therapeutic.
Continue to: When a patient complains...
When a patient complains of severe pruritus at first presentation, consider pharmacotherapy in conjunction with nonpharmacotherapeutic options. Several of the more effective topical therapies for pruritusa are listed in TABLE 4.20 Well-known systemic agents for this purpose are reviewed below and listed in TABLE 5.7

Systemic treatment
Antihistamines. A staple in the treatment of pruritus for many years, antihistamines are not effective for all causes; however, they are effective in treating paraneoplastic pruritus.20 First-generation antihistamines, with their sedating effect, can be useful for patients who experience generalized pruritus at night.20

Anticonvulsants. Gabapentin and pregabalin are analogs of the neurotransmitter gamma-aminobutyric acid.20 This drug class is helpful in neuropathic pruritus specifically caused by impingements, such as brachioradial pruritus and notalgia paresthetica.20 In addition, of all systemic therapies used to treat uremic pruritus, gabapentin has, in clinical trials, most consistently been found effective for uremic pruritus.6 (Note: Use renal dosing of gabapentin in patients with renal failure.)
Antidepressants. Selective serotonin reuptake inhibitors (SSRIs; eg, fluvoxamine, paroxetine, and sertraline) might cause itch to subside by increasing the serotonin level, which, in turn, works to decrease inflammatory substances that cause itch.7 SSRIs have been used to treat patients with psychogenic pruritus, cholestatic pruritus, and paraneoplastic pruritus.7
Tricyclic antidepressants (eg, amitriptyline and doxepin) lessen the itch by antagonizing histamine receptors and through anticholinergic mechanisms. Tricyclics are best used in the treatment of psychogenic and nocturnal itch.7
Continue to: Mirtazapine...
Mirtazapine, a tetracyclic antidepressant, works in patients with uremic pruritus, psychogenic pruritus, cholestatic pruritus, and paraneoplastic pruritus.1
Substance P antagonist. Aprepitant, a neurokinin receptor I antagonist, is a newer agent that inhibits binding of the itch mediator substance P to the neurokinin receptor. The drug has been found helpful in patients with drug-induced, paraneoplastic, and brachioradial pruritus.7
Opioid-receptor agents. Naltrexone, as a mu opioid-receptor antagonist, has shown promise as a treatment for uremic pruritus and cholestatic pruritus. Nalfurafine, a kappa opioid-receptor agonist, is emerging as a possible therapy for uremic pruritus.7
Bile-acid sequestrants. A few small studies have shown that treatment with a bile-acid sequestrant, such as cholestyramine and ursodiol, induces moderate improvement in symptoms in patients with cholestatic pruritus.21
CORRESPONDENCE
Matasha Russell, MD, Department of Family and Community Medicine, The University of Texas Health Science Center at Houston, McGovern Medical School, 6431 Fannin Street, JJL 324, Houston, TX 77030; Matasha.L.Russell@uth.tmc.edu.
1. Tarikci N, E, S, et al. Pruritus in systemic diseases: a review of etiological factors and new treatment modalities. ScientificWorldJournal. 2015;2015:803752.
2. Yosipovitch G, Bernhard JD. Clinical practice. Chronic pruritus. N Engl J Med. 2013;368:1625-1634.
3. Silverberg JI, Kantor RW, Dalal P. A comprehensive conceptual model of the experience of chronic itch in adults. Am J Clin Dermatol. 2018;19:759-769.
4. Matterne U, Apfelbacher CJ, Vogelgsang L, et al. Incidence and determinants of chronic pruritus: a population based cohort study. Acta Derm Venereol. 2013;93:532-537.
5. Moses S. Pruritus. Am Fam Physician. 2003;68:1135-1142.
6. Combs SA, Teixeira JP, Germain MJ. Pruritus in kidney disease. Semin Nephrol. 2015;35:383-391.
7. Shevchenko A, Valdes-Rodriguez R, Yosipovitch G. Causes, pathophysiology, and treatment of pruritus in the mature patient. Clin Dermatol. 2018;36:140-151.
8. Reamy BV, Bunt C. A diagnostic approach to pruritus. Am Fam Physician. 2011;84:195-202.
9. M. Current concepts of pathophysiology, epidemiology and classification of pruritus. Srp Arh Celok Lek. 2014;142:106-112.
10. Fett N, Haynes K, Propert KJ, et al. Five-year malignancy incidence in patients with chronic pruritus: a population-based cohort study aimed at limiting unnecessary screening practices. J Am Acad Dermatol. 2014;70:651-658.
11. Larson VA, Tang O, S, et al. Association between itch and cancer in 16,925 patients with pruritus: experience at a tertiary care center. J Am Acad Dermatol. 2019;80:931-937.
12. Rosen JD, Fostini AC, Chan YH, et al. Cross-sectional study of clinical distinctions between neuropathic and inflammatory pruritus. J Am Acad Dermatol. 2018;79:1143-1144.
13. Oaklander AL. Neuropathic itch. Semin Cutan Med Surg. 2011;30:87-92.
14. Ferm I, Sterner M, Wallengren J. Somatic and psychiatric comorbidity in patients with chronic pruritus. Acta Derm Venereol. 2010;90:395-400.
15. Jafferany M, Davari ME. Itch and psyche: psychiatric aspects of pruritus. Int J Dermatol. 2019;58:3-23.
16. Koo J, Lebwohl A. Psychodermatology: the mind and skin connection. Am Fam Physician. 2001;64:1873-1878.
17. Bewley AP, Lepping P, Freudenmann RW, et al. Delusional parasitosis: time to call it delusional infestation. Br J Dermatol.2010;163:1-2.
18. Clerc C-J, Misery L. A literature review of senile pruritus: from diagnosis to treatment. Acta Derm Venereol. 2017;97:433-440.
19. Bernhard JD, Bordeaux JS. Medical pearl: the ice-pack sign in brachioradial pruritus. J Am Acad Dermatol. 2005;52:1073.
20. Sanders KM, Nattkemper LA, Yosipovitch G. Advances in understanding itching and scratching: a new era of targeted treatments [version 1]. F1000Res. 2016;5 F1000 Faculty Rev–2042.
21. Hegade VS, Kendrick SFW, Dobbins RL, et al. Effect of ileal bile acid transporter inhibitor GSK2330672 on pruritus in primary biliary cholangitis: a double-blind, randomised, placebo-controlled, crossover, phase 2a study. Lancet. 2017;389:1114-1123.
1. Tarikci N, E, S, et al. Pruritus in systemic diseases: a review of etiological factors and new treatment modalities. ScientificWorldJournal. 2015;2015:803752.
2. Yosipovitch G, Bernhard JD. Clinical practice. Chronic pruritus. N Engl J Med. 2013;368:1625-1634.
3. Silverberg JI, Kantor RW, Dalal P. A comprehensive conceptual model of the experience of chronic itch in adults. Am J Clin Dermatol. 2018;19:759-769.
4. Matterne U, Apfelbacher CJ, Vogelgsang L, et al. Incidence and determinants of chronic pruritus: a population based cohort study. Acta Derm Venereol. 2013;93:532-537.
5. Moses S. Pruritus. Am Fam Physician. 2003;68:1135-1142.
6. Combs SA, Teixeira JP, Germain MJ. Pruritus in kidney disease. Semin Nephrol. 2015;35:383-391.
7. Shevchenko A, Valdes-Rodriguez R, Yosipovitch G. Causes, pathophysiology, and treatment of pruritus in the mature patient. Clin Dermatol. 2018;36:140-151.
8. Reamy BV, Bunt C. A diagnostic approach to pruritus. Am Fam Physician. 2011;84:195-202.
9. M. Current concepts of pathophysiology, epidemiology and classification of pruritus. Srp Arh Celok Lek. 2014;142:106-112.
10. Fett N, Haynes K, Propert KJ, et al. Five-year malignancy incidence in patients with chronic pruritus: a population-based cohort study aimed at limiting unnecessary screening practices. J Am Acad Dermatol. 2014;70:651-658.
11. Larson VA, Tang O, S, et al. Association between itch and cancer in 16,925 patients with pruritus: experience at a tertiary care center. J Am Acad Dermatol. 2019;80:931-937.
12. Rosen JD, Fostini AC, Chan YH, et al. Cross-sectional study of clinical distinctions between neuropathic and inflammatory pruritus. J Am Acad Dermatol. 2018;79:1143-1144.
13. Oaklander AL. Neuropathic itch. Semin Cutan Med Surg. 2011;30:87-92.
14. Ferm I, Sterner M, Wallengren J. Somatic and psychiatric comorbidity in patients with chronic pruritus. Acta Derm Venereol. 2010;90:395-400.
15. Jafferany M, Davari ME. Itch and psyche: psychiatric aspects of pruritus. Int J Dermatol. 2019;58:3-23.
16. Koo J, Lebwohl A. Psychodermatology: the mind and skin connection. Am Fam Physician. 2001;64:1873-1878.
17. Bewley AP, Lepping P, Freudenmann RW, et al. Delusional parasitosis: time to call it delusional infestation. Br J Dermatol.2010;163:1-2.
18. Clerc C-J, Misery L. A literature review of senile pruritus: from diagnosis to treatment. Acta Derm Venereol. 2017;97:433-440.
19. Bernhard JD, Bordeaux JS. Medical pearl: the ice-pack sign in brachioradial pruritus. J Am Acad Dermatol. 2005;52:1073.
20. Sanders KM, Nattkemper LA, Yosipovitch G. Advances in understanding itching and scratching: a new era of targeted treatments [version 1]. F1000Res. 2016;5 F1000 Faculty Rev–2042.
21. Hegade VS, Kendrick SFW, Dobbins RL, et al. Effect of ileal bile acid transporter inhibitor GSK2330672 on pruritus in primary biliary cholangitis: a double-blind, randomised, placebo-controlled, crossover, phase 2a study. Lancet. 2017;389:1114-1123.
PRACTICE RECOMMENDATIONS
› Undertake a diagnostic work-up for systemic causes of pruritus in patients who have a chronic, generalized itch and abnormal findings on physical examination. C
› Prescribe gabapentin for its effectiveness in treating pruritus caused by uremic and neurologic itch. B
› Consider prescribing one of the bile-acid sequestrants in patients with cholestatic pruritus because these agents can provide moderate relief of the symptom. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Increasing ear pain and headache
A previously healthy 12-year-old boy with normal development presented to his primary care physician (PCP) with a 1-week history of moderate ear pain. He was given a diagnosis of acute otitis media (AOM) and prescribed a 7-day course of amoxicillin. Although the child’s history was otherwise unremarkable, the mother reported that she’d had a deep venous thrombosis and pulmonary embolism a year earlier.
The boy continued to experience intermittent left ear pain 2 weeks after completing his antibiotic treatment, leading the PCP to refer him to an otolaryngologist. An examination by the otolaryngologist revealed a cloudy, bulging tympanic membrane. The patient was prescribed amoxicillin/clavulanate and ofloxacin ear drops.
Two days later, he was admitted to the emergency department (ED) due to worsening left ear pain and a new-onset left-sided headache. His left tympanic membrane was normal, with no tenderness or erythema of the mastoid. His vital signs were normal. He was afebrile and discharged home.
A week later, he returned to the ED with worsening ear pain and severe persistent headache, which was localized in the left occipital, left frontal, and retro-orbital regions. He denied light or sound sensitivity, nausea, vomiting, or increased lacrimation. He was tearful on examination due to the pain. He had no meningismus and normal fundi. A neurologic examination was nonlateralizing. Laboratory tests showed a normal complete blood count but increased C-reactive protein at 113 mg/dL (normal, < 0.3 mg/dL) and an erythrocyte sedimentation rate of 88 mm/hr (normal, 0-20 mm/hr).
Magnetic resonance imaging was ordered (FIGURES 1A and 1B), and Neurosurgery and Otolaryngology were consulted.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Acute mastoiditis with epidural abscess
The contrast-enhanced cranial MRI scan (FIGURE 1A) revealed a case of acute mastoiditis with fluid in the left mastoid (blue arrow) and a large epidural abscess in the left posterior fossa (green arrow). The normal right mastoid was air-filled (yellow arrow). The T2-weighted MRI scan (FIGURE 1B) showed mild dilatation of the lateral ventricles (blue arrow) secondary to compression on the fourth ventricle by mass effect from the epidural abscess.
Acute mastoiditis—a complication of AOM—is an inflammatory process of mastoid air cells, which are contiguous to the middle ear cleft. In one large study of 61,783 inpatient children admitted with AOM, acute mastoiditis was reported as the most common complication in 1505 (2.4%) of the cases.1 The 2000-2012 national estimated incidence rate of pediatric mastoiditis has ranged from a high of 2.7 per 100,000 population in 2006 to a low of 1.8 per 100,000 in 2012.2 Clinical features of mastoiditis include localized mastoid tenderness, swelling, erythema, fluctuance, protrusion of the auricle, and ear pain.3
The clinical presentation of epidural abscess can be subtle with fever, headache, neck pain, and changes in mental status developing over several days.1 Focal deficits and seizures are relatively uncommon. In a review of 308 children with acute mastoiditis (3 with an epidural abscess), high-grade fever and high absolute neutrophil count and C-reactive protein levels were associated with the development of complications of mastoiditis, including hearing loss, sinus venous thrombosis, intracranial abscess, and cranial nerve palsies.4
Venous sinus thrombosis was part of the differential
When we were caring for this patient, the differential diagnosis included a cranial extension of AOM. Venous sinus thrombosis was also considered, given the family history of a hypercoagulable state. The patient did not have any features suggesting primary headache syndromes, such as migraine, tension type, or cluster headache.
The differential for a patient complaining of ear pain also includes postauricular lymphadenopathy, mumps, periauricular cellulitis (with and without otitis externa), perichondritis of the auricle, and tumors involving the mastoid bone.4
Continue to: Imaging and treatment
Imaging and treatment
Imaging of temporal bone is not recommended to make a diagnosis of mastoiditis in children with characteristic clinical findings. When imaging is needed, contrast-enhanced computed tomography (CT) is best to help visualize changes in temporal bone. If intracranial complications are suspected, cranial MRI with contrast or cranial CT with contrast can be ordered (depending on availability).5
Conservative management with intravenous antimicrobial therapy and middle ear drainage with myringotomy is indicated for a child with uncomplicated acute or subacute mastoiditis. For patients with suppurative extracranial or intracranial complications, aggressive surgical management is needed.5
Treatment for this patient included craniotomy, evacuation of the epidural abscess, and mastoidectomy. A culture obtained from the abscess showed Streptococcus intermedius. He was treated with broad-spectrum antibiotics, including ceftriaxone, vancomycin, and metronidazole. Within a week of surgery, he was discharged from the hospital and continued antibiotic treatment for 6 weeks via a peripherally inserted central catheter line.
1. Lavin JM, Rusher T, Shah RK. Complications of pediatric otitis media. Otolaryngol Head Neck Surg. 2016;154:366-370.
2. King LM, Bartoces M, Hersh AL, et al. National incidence of pediatric mastoiditis in the United States, 2000-2012: creating a baseline for public health surveillance. Pediatr Infect Dis J. 2019;38:e14-e16.
3. Pang LH, Barakate MS, Havas TE. Mastoiditis in a paediatric population: a review of 11 years’ experience in management. Int J Pediatr Otorhinolaryngol. 2009;73:1520.
4. Bilavsky E, Yarden-Bilavsky H, Samra Z, et al. Clinical, laboratory, and microbiological differences between children with simple or complicated mastoiditis. Int J Pediatr Otorhinolaryngol. 2009;73:1270-1273.
5. Chesney J, Black A, Choo D. What is the best practice for acute mastoiditis in children? Laryngoscope. 2014;124:1057-1059.
A previously healthy 12-year-old boy with normal development presented to his primary care physician (PCP) with a 1-week history of moderate ear pain. He was given a diagnosis of acute otitis media (AOM) and prescribed a 7-day course of amoxicillin. Although the child’s history was otherwise unremarkable, the mother reported that she’d had a deep venous thrombosis and pulmonary embolism a year earlier.
The boy continued to experience intermittent left ear pain 2 weeks after completing his antibiotic treatment, leading the PCP to refer him to an otolaryngologist. An examination by the otolaryngologist revealed a cloudy, bulging tympanic membrane. The patient was prescribed amoxicillin/clavulanate and ofloxacin ear drops.
Two days later, he was admitted to the emergency department (ED) due to worsening left ear pain and a new-onset left-sided headache. His left tympanic membrane was normal, with no tenderness or erythema of the mastoid. His vital signs were normal. He was afebrile and discharged home.
A week later, he returned to the ED with worsening ear pain and severe persistent headache, which was localized in the left occipital, left frontal, and retro-orbital regions. He denied light or sound sensitivity, nausea, vomiting, or increased lacrimation. He was tearful on examination due to the pain. He had no meningismus and normal fundi. A neurologic examination was nonlateralizing. Laboratory tests showed a normal complete blood count but increased C-reactive protein at 113 mg/dL (normal, < 0.3 mg/dL) and an erythrocyte sedimentation rate of 88 mm/hr (normal, 0-20 mm/hr).
Magnetic resonance imaging was ordered (FIGURES 1A and 1B), and Neurosurgery and Otolaryngology were consulted.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Acute mastoiditis with epidural abscess
The contrast-enhanced cranial MRI scan (FIGURE 1A) revealed a case of acute mastoiditis with fluid in the left mastoid (blue arrow) and a large epidural abscess in the left posterior fossa (green arrow). The normal right mastoid was air-filled (yellow arrow). The T2-weighted MRI scan (FIGURE 1B) showed mild dilatation of the lateral ventricles (blue arrow) secondary to compression on the fourth ventricle by mass effect from the epidural abscess.
Acute mastoiditis—a complication of AOM—is an inflammatory process of mastoid air cells, which are contiguous to the middle ear cleft. In one large study of 61,783 inpatient children admitted with AOM, acute mastoiditis was reported as the most common complication in 1505 (2.4%) of the cases.1 The 2000-2012 national estimated incidence rate of pediatric mastoiditis has ranged from a high of 2.7 per 100,000 population in 2006 to a low of 1.8 per 100,000 in 2012.2 Clinical features of mastoiditis include localized mastoid tenderness, swelling, erythema, fluctuance, protrusion of the auricle, and ear pain.3
The clinical presentation of epidural abscess can be subtle with fever, headache, neck pain, and changes in mental status developing over several days.1 Focal deficits and seizures are relatively uncommon. In a review of 308 children with acute mastoiditis (3 with an epidural abscess), high-grade fever and high absolute neutrophil count and C-reactive protein levels were associated with the development of complications of mastoiditis, including hearing loss, sinus venous thrombosis, intracranial abscess, and cranial nerve palsies.4
Venous sinus thrombosis was part of the differential
When we were caring for this patient, the differential diagnosis included a cranial extension of AOM. Venous sinus thrombosis was also considered, given the family history of a hypercoagulable state. The patient did not have any features suggesting primary headache syndromes, such as migraine, tension type, or cluster headache.
The differential for a patient complaining of ear pain also includes postauricular lymphadenopathy, mumps, periauricular cellulitis (with and without otitis externa), perichondritis of the auricle, and tumors involving the mastoid bone.4
Continue to: Imaging and treatment
Imaging and treatment
Imaging of temporal bone is not recommended to make a diagnosis of mastoiditis in children with characteristic clinical findings. When imaging is needed, contrast-enhanced computed tomography (CT) is best to help visualize changes in temporal bone. If intracranial complications are suspected, cranial MRI with contrast or cranial CT with contrast can be ordered (depending on availability).5
Conservative management with intravenous antimicrobial therapy and middle ear drainage with myringotomy is indicated for a child with uncomplicated acute or subacute mastoiditis. For patients with suppurative extracranial or intracranial complications, aggressive surgical management is needed.5
Treatment for this patient included craniotomy, evacuation of the epidural abscess, and mastoidectomy. A culture obtained from the abscess showed Streptococcus intermedius. He was treated with broad-spectrum antibiotics, including ceftriaxone, vancomycin, and metronidazole. Within a week of surgery, he was discharged from the hospital and continued antibiotic treatment for 6 weeks via a peripherally inserted central catheter line.
A previously healthy 12-year-old boy with normal development presented to his primary care physician (PCP) with a 1-week history of moderate ear pain. He was given a diagnosis of acute otitis media (AOM) and prescribed a 7-day course of amoxicillin. Although the child’s history was otherwise unremarkable, the mother reported that she’d had a deep venous thrombosis and pulmonary embolism a year earlier.
The boy continued to experience intermittent left ear pain 2 weeks after completing his antibiotic treatment, leading the PCP to refer him to an otolaryngologist. An examination by the otolaryngologist revealed a cloudy, bulging tympanic membrane. The patient was prescribed amoxicillin/clavulanate and ofloxacin ear drops.
Two days later, he was admitted to the emergency department (ED) due to worsening left ear pain and a new-onset left-sided headache. His left tympanic membrane was normal, with no tenderness or erythema of the mastoid. His vital signs were normal. He was afebrile and discharged home.
A week later, he returned to the ED with worsening ear pain and severe persistent headache, which was localized in the left occipital, left frontal, and retro-orbital regions. He denied light or sound sensitivity, nausea, vomiting, or increased lacrimation. He was tearful on examination due to the pain. He had no meningismus and normal fundi. A neurologic examination was nonlateralizing. Laboratory tests showed a normal complete blood count but increased C-reactive protein at 113 mg/dL (normal, < 0.3 mg/dL) and an erythrocyte sedimentation rate of 88 mm/hr (normal, 0-20 mm/hr).
Magnetic resonance imaging was ordered (FIGURES 1A and 1B), and Neurosurgery and Otolaryngology were consulted.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Dx: Acute mastoiditis with epidural abscess
The contrast-enhanced cranial MRI scan (FIGURE 1A) revealed a case of acute mastoiditis with fluid in the left mastoid (blue arrow) and a large epidural abscess in the left posterior fossa (green arrow). The normal right mastoid was air-filled (yellow arrow). The T2-weighted MRI scan (FIGURE 1B) showed mild dilatation of the lateral ventricles (blue arrow) secondary to compression on the fourth ventricle by mass effect from the epidural abscess.
Acute mastoiditis—a complication of AOM—is an inflammatory process of mastoid air cells, which are contiguous to the middle ear cleft. In one large study of 61,783 inpatient children admitted with AOM, acute mastoiditis was reported as the most common complication in 1505 (2.4%) of the cases.1 The 2000-2012 national estimated incidence rate of pediatric mastoiditis has ranged from a high of 2.7 per 100,000 population in 2006 to a low of 1.8 per 100,000 in 2012.2 Clinical features of mastoiditis include localized mastoid tenderness, swelling, erythema, fluctuance, protrusion of the auricle, and ear pain.3
The clinical presentation of epidural abscess can be subtle with fever, headache, neck pain, and changes in mental status developing over several days.1 Focal deficits and seizures are relatively uncommon. In a review of 308 children with acute mastoiditis (3 with an epidural abscess), high-grade fever and high absolute neutrophil count and C-reactive protein levels were associated with the development of complications of mastoiditis, including hearing loss, sinus venous thrombosis, intracranial abscess, and cranial nerve palsies.4
Venous sinus thrombosis was part of the differential
When we were caring for this patient, the differential diagnosis included a cranial extension of AOM. Venous sinus thrombosis was also considered, given the family history of a hypercoagulable state. The patient did not have any features suggesting primary headache syndromes, such as migraine, tension type, or cluster headache.
The differential for a patient complaining of ear pain also includes postauricular lymphadenopathy, mumps, periauricular cellulitis (with and without otitis externa), perichondritis of the auricle, and tumors involving the mastoid bone.4
Continue to: Imaging and treatment
Imaging and treatment
Imaging of temporal bone is not recommended to make a diagnosis of mastoiditis in children with characteristic clinical findings. When imaging is needed, contrast-enhanced computed tomography (CT) is best to help visualize changes in temporal bone. If intracranial complications are suspected, cranial MRI with contrast or cranial CT with contrast can be ordered (depending on availability).5
Conservative management with intravenous antimicrobial therapy and middle ear drainage with myringotomy is indicated for a child with uncomplicated acute or subacute mastoiditis. For patients with suppurative extracranial or intracranial complications, aggressive surgical management is needed.5
Treatment for this patient included craniotomy, evacuation of the epidural abscess, and mastoidectomy. A culture obtained from the abscess showed Streptococcus intermedius. He was treated with broad-spectrum antibiotics, including ceftriaxone, vancomycin, and metronidazole. Within a week of surgery, he was discharged from the hospital and continued antibiotic treatment for 6 weeks via a peripherally inserted central catheter line.
1. Lavin JM, Rusher T, Shah RK. Complications of pediatric otitis media. Otolaryngol Head Neck Surg. 2016;154:366-370.
2. King LM, Bartoces M, Hersh AL, et al. National incidence of pediatric mastoiditis in the United States, 2000-2012: creating a baseline for public health surveillance. Pediatr Infect Dis J. 2019;38:e14-e16.
3. Pang LH, Barakate MS, Havas TE. Mastoiditis in a paediatric population: a review of 11 years’ experience in management. Int J Pediatr Otorhinolaryngol. 2009;73:1520.
4. Bilavsky E, Yarden-Bilavsky H, Samra Z, et al. Clinical, laboratory, and microbiological differences between children with simple or complicated mastoiditis. Int J Pediatr Otorhinolaryngol. 2009;73:1270-1273.
5. Chesney J, Black A, Choo D. What is the best practice for acute mastoiditis in children? Laryngoscope. 2014;124:1057-1059.
1. Lavin JM, Rusher T, Shah RK. Complications of pediatric otitis media. Otolaryngol Head Neck Surg. 2016;154:366-370.
2. King LM, Bartoces M, Hersh AL, et al. National incidence of pediatric mastoiditis in the United States, 2000-2012: creating a baseline for public health surveillance. Pediatr Infect Dis J. 2019;38:e14-e16.
3. Pang LH, Barakate MS, Havas TE. Mastoiditis in a paediatric population: a review of 11 years’ experience in management. Int J Pediatr Otorhinolaryngol. 2009;73:1520.
4. Bilavsky E, Yarden-Bilavsky H, Samra Z, et al. Clinical, laboratory, and microbiological differences between children with simple or complicated mastoiditis. Int J Pediatr Otorhinolaryngol. 2009;73:1270-1273.
5. Chesney J, Black A, Choo D. What is the best practice for acute mastoiditis in children? Laryngoscope. 2014;124:1057-1059.
Gene-replacement therapy shows promise in X-linked myotubular myopathy
, according to research presented at the 2020 CNS-ICNA Conjoint Meeting, which was held virtually this year. The treatment also appears to improve patients’ motor function significantly and help them to achieve motor milestones.
The results come from a phase 1/2 study of two doses of AT132. Three of 17 patients who received the higher dose had fatal liver dysfunction. The researchers are investigating these cases and will communicate their findings.
X-linked myotubular myopathy is a rare and often fatal neuromuscular disease. Mutations in MTM1, which encodes the myotubularin enzyme that is required for the development and function of skeletal muscle, cause the disease, which affects about one in 50,000 to one in 40,000 newborn boys. The disease is associated with profound muscle weakness and impairment of neuromuscular and respiratory function. Patients with X-linked myotubular myopathy achieve motor milestones much later or not at all, and most require a ventilator or a feeding tube. The mortality by age 18 months is approximately 50%.
The ASPIRO trial
Investigators theorized that muscle tissue would be an appropriate therapeutic target because it does not display dystrophic or inflammatory changes in most patients. They identified adeno-associated virus AAV8 as a potential carrier for gene therapy, since it targets skeletal muscle effectively.
Nancy L. Kuntz, MD, an attending physician at Ann and Robert H. Lurie Children’s Hospital of Chicago, and colleagues conducted the ASPIRO trial to examine AT132 as a potential treatment for X-linked myotubular myopathy. Eligible patients were younger than 5 years or had previously enrolled in a natural history study of the disease, required ventilator support at baseline, and had no clinically significant underlying liver disease. Patients were randomly assigned to 1 × 1014 vg/kg of AAT132, 3 × 1014 vg/kg of AT132, or delayed treatment. Participants assigned to delayed treatment served as the study’s control group.
The study’s primary end points were safety and change in hours of daily ventilator support from baseline to week 24 after dosing. The investigators also examined a respiratory endpoint (i.e., maximal inspiratory pressure [MIP]) and neuromuscular endpoints (i.e., motor milestones, CHOP INTEND score, and muscle biopsy).
Treatment improved respiratory function
As of July 28, Dr. Kuntz and colleagues had enrolled 23 patients in the trial. Six participants received the lower dose of therapy, and 17 received the higher dose. Median age was 1.7 years for the low-dose group and 2.6 years for the high-dose group.
Patients assigned to receive the higher dose of therapy received treatment more recently than the low-dose group, and not all of the former have reached 48 weeks since treatment, said Dr. Kuntz. Fewer efficacy data are thus available for the high-dose group.
Each dose of AT132 was associated with a significantly greater decrease from baseline in least squares mean daily hours of ventilator dependence, compared with the control condition. At week 48, the mean reduction was approximately 19 hours/day for patients receiving 1 × 1014 vg/kg of AAT132 and approximately 13 hours per day for patients receiving 3 × 1014 vg/kg of AT132. The investigators did not perform a statistical comparison of the two doses because of differing protocols for ventilator weaning between groups. All six patients who received the lower dose achieved ventilator independence, as did one patient who received the higher dose.
In addition, all treated patients had significantly greater increases from baseline in least squares mean MIP, compared with controls. The mean increase was 45.7 cmH2O for the low-dose group, 46.1 cmH2O for the high-dose group, and −8.0 cmH2O for controls.
Before treatment, most patients had not achieved any of the motor milestones that investigators assessed. After treatment, five of six patients receiving the low dose achieved independent walking, as did one in 10 patients receiving the high dose. No controls achieved this milestone. Treated patients also had significantly greater increases from baseline in least squares mean CHOP INTEND scores, compared with controls. At least at one time point, five of six patients receiving the low dose, six of 10 patients receiving the high dose, and one control patient achieved the mean score observed in healthy infants.
Patients in both treatment arms had improvements in muscle pathology at weeks 24 and 48, including improvements in organelle localization and fiber size. In addition, patients in both treatment arms had continued detectable vector copies and myotubularin protein expression at both time points.
Deaths under investigation
In the low-dose group, one patient had four serious treatment-emergent adverse events, and in the high-dose group, eight patients had 27 serious treatment-emergent adverse events. The three patients in the high-dose group who developed fatal liver dysfunction were among the older, heavier patients in the study and, consequently, received among the highest total doses of treatment. These patients had evidence of likely preexisting intrahepatic cholestasis.
“This clinical trial is on hold pending discussions between regulatory agencies and the study sponsor regarding additional recruitment and the duration of follow-up,” said Dr. Kuntz.
Audentes Therapeutics, which is developing AT132, funded the trial. Dr. Kuntz had no conflicts of interest.
SOURCE: Bönnemann CG et al. CNS-ICNA 2020, Abstract P.62.
, according to research presented at the 2020 CNS-ICNA Conjoint Meeting, which was held virtually this year. The treatment also appears to improve patients’ motor function significantly and help them to achieve motor milestones.
The results come from a phase 1/2 study of two doses of AT132. Three of 17 patients who received the higher dose had fatal liver dysfunction. The researchers are investigating these cases and will communicate their findings.
X-linked myotubular myopathy is a rare and often fatal neuromuscular disease. Mutations in MTM1, which encodes the myotubularin enzyme that is required for the development and function of skeletal muscle, cause the disease, which affects about one in 50,000 to one in 40,000 newborn boys. The disease is associated with profound muscle weakness and impairment of neuromuscular and respiratory function. Patients with X-linked myotubular myopathy achieve motor milestones much later or not at all, and most require a ventilator or a feeding tube. The mortality by age 18 months is approximately 50%.
The ASPIRO trial
Investigators theorized that muscle tissue would be an appropriate therapeutic target because it does not display dystrophic or inflammatory changes in most patients. They identified adeno-associated virus AAV8 as a potential carrier for gene therapy, since it targets skeletal muscle effectively.
Nancy L. Kuntz, MD, an attending physician at Ann and Robert H. Lurie Children’s Hospital of Chicago, and colleagues conducted the ASPIRO trial to examine AT132 as a potential treatment for X-linked myotubular myopathy. Eligible patients were younger than 5 years or had previously enrolled in a natural history study of the disease, required ventilator support at baseline, and had no clinically significant underlying liver disease. Patients were randomly assigned to 1 × 1014 vg/kg of AAT132, 3 × 1014 vg/kg of AT132, or delayed treatment. Participants assigned to delayed treatment served as the study’s control group.
The study’s primary end points were safety and change in hours of daily ventilator support from baseline to week 24 after dosing. The investigators also examined a respiratory endpoint (i.e., maximal inspiratory pressure [MIP]) and neuromuscular endpoints (i.e., motor milestones, CHOP INTEND score, and muscle biopsy).
Treatment improved respiratory function
As of July 28, Dr. Kuntz and colleagues had enrolled 23 patients in the trial. Six participants received the lower dose of therapy, and 17 received the higher dose. Median age was 1.7 years for the low-dose group and 2.6 years for the high-dose group.
Patients assigned to receive the higher dose of therapy received treatment more recently than the low-dose group, and not all of the former have reached 48 weeks since treatment, said Dr. Kuntz. Fewer efficacy data are thus available for the high-dose group.
Each dose of AT132 was associated with a significantly greater decrease from baseline in least squares mean daily hours of ventilator dependence, compared with the control condition. At week 48, the mean reduction was approximately 19 hours/day for patients receiving 1 × 1014 vg/kg of AAT132 and approximately 13 hours per day for patients receiving 3 × 1014 vg/kg of AT132. The investigators did not perform a statistical comparison of the two doses because of differing protocols for ventilator weaning between groups. All six patients who received the lower dose achieved ventilator independence, as did one patient who received the higher dose.
In addition, all treated patients had significantly greater increases from baseline in least squares mean MIP, compared with controls. The mean increase was 45.7 cmH2O for the low-dose group, 46.1 cmH2O for the high-dose group, and −8.0 cmH2O for controls.
Before treatment, most patients had not achieved any of the motor milestones that investigators assessed. After treatment, five of six patients receiving the low dose achieved independent walking, as did one in 10 patients receiving the high dose. No controls achieved this milestone. Treated patients also had significantly greater increases from baseline in least squares mean CHOP INTEND scores, compared with controls. At least at one time point, five of six patients receiving the low dose, six of 10 patients receiving the high dose, and one control patient achieved the mean score observed in healthy infants.
Patients in both treatment arms had improvements in muscle pathology at weeks 24 and 48, including improvements in organelle localization and fiber size. In addition, patients in both treatment arms had continued detectable vector copies and myotubularin protein expression at both time points.
Deaths under investigation
In the low-dose group, one patient had four serious treatment-emergent adverse events, and in the high-dose group, eight patients had 27 serious treatment-emergent adverse events. The three patients in the high-dose group who developed fatal liver dysfunction were among the older, heavier patients in the study and, consequently, received among the highest total doses of treatment. These patients had evidence of likely preexisting intrahepatic cholestasis.
“This clinical trial is on hold pending discussions between regulatory agencies and the study sponsor regarding additional recruitment and the duration of follow-up,” said Dr. Kuntz.
Audentes Therapeutics, which is developing AT132, funded the trial. Dr. Kuntz had no conflicts of interest.
SOURCE: Bönnemann CG et al. CNS-ICNA 2020, Abstract P.62.
, according to research presented at the 2020 CNS-ICNA Conjoint Meeting, which was held virtually this year. The treatment also appears to improve patients’ motor function significantly and help them to achieve motor milestones.
The results come from a phase 1/2 study of two doses of AT132. Three of 17 patients who received the higher dose had fatal liver dysfunction. The researchers are investigating these cases and will communicate their findings.
X-linked myotubular myopathy is a rare and often fatal neuromuscular disease. Mutations in MTM1, which encodes the myotubularin enzyme that is required for the development and function of skeletal muscle, cause the disease, which affects about one in 50,000 to one in 40,000 newborn boys. The disease is associated with profound muscle weakness and impairment of neuromuscular and respiratory function. Patients with X-linked myotubular myopathy achieve motor milestones much later or not at all, and most require a ventilator or a feeding tube. The mortality by age 18 months is approximately 50%.
The ASPIRO trial
Investigators theorized that muscle tissue would be an appropriate therapeutic target because it does not display dystrophic or inflammatory changes in most patients. They identified adeno-associated virus AAV8 as a potential carrier for gene therapy, since it targets skeletal muscle effectively.
Nancy L. Kuntz, MD, an attending physician at Ann and Robert H. Lurie Children’s Hospital of Chicago, and colleagues conducted the ASPIRO trial to examine AT132 as a potential treatment for X-linked myotubular myopathy. Eligible patients were younger than 5 years or had previously enrolled in a natural history study of the disease, required ventilator support at baseline, and had no clinically significant underlying liver disease. Patients were randomly assigned to 1 × 1014 vg/kg of AAT132, 3 × 1014 vg/kg of AT132, or delayed treatment. Participants assigned to delayed treatment served as the study’s control group.
The study’s primary end points were safety and change in hours of daily ventilator support from baseline to week 24 after dosing. The investigators also examined a respiratory endpoint (i.e., maximal inspiratory pressure [MIP]) and neuromuscular endpoints (i.e., motor milestones, CHOP INTEND score, and muscle biopsy).
Treatment improved respiratory function
As of July 28, Dr. Kuntz and colleagues had enrolled 23 patients in the trial. Six participants received the lower dose of therapy, and 17 received the higher dose. Median age was 1.7 years for the low-dose group and 2.6 years for the high-dose group.
Patients assigned to receive the higher dose of therapy received treatment more recently than the low-dose group, and not all of the former have reached 48 weeks since treatment, said Dr. Kuntz. Fewer efficacy data are thus available for the high-dose group.
Each dose of AT132 was associated with a significantly greater decrease from baseline in least squares mean daily hours of ventilator dependence, compared with the control condition. At week 48, the mean reduction was approximately 19 hours/day for patients receiving 1 × 1014 vg/kg of AAT132 and approximately 13 hours per day for patients receiving 3 × 1014 vg/kg of AT132. The investigators did not perform a statistical comparison of the two doses because of differing protocols for ventilator weaning between groups. All six patients who received the lower dose achieved ventilator independence, as did one patient who received the higher dose.
In addition, all treated patients had significantly greater increases from baseline in least squares mean MIP, compared with controls. The mean increase was 45.7 cmH2O for the low-dose group, 46.1 cmH2O for the high-dose group, and −8.0 cmH2O for controls.
Before treatment, most patients had not achieved any of the motor milestones that investigators assessed. After treatment, five of six patients receiving the low dose achieved independent walking, as did one in 10 patients receiving the high dose. No controls achieved this milestone. Treated patients also had significantly greater increases from baseline in least squares mean CHOP INTEND scores, compared with controls. At least at one time point, five of six patients receiving the low dose, six of 10 patients receiving the high dose, and one control patient achieved the mean score observed in healthy infants.
Patients in both treatment arms had improvements in muscle pathology at weeks 24 and 48, including improvements in organelle localization and fiber size. In addition, patients in both treatment arms had continued detectable vector copies and myotubularin protein expression at both time points.
Deaths under investigation
In the low-dose group, one patient had four serious treatment-emergent adverse events, and in the high-dose group, eight patients had 27 serious treatment-emergent adverse events. The three patients in the high-dose group who developed fatal liver dysfunction were among the older, heavier patients in the study and, consequently, received among the highest total doses of treatment. These patients had evidence of likely preexisting intrahepatic cholestasis.
“This clinical trial is on hold pending discussions between regulatory agencies and the study sponsor regarding additional recruitment and the duration of follow-up,” said Dr. Kuntz.
Audentes Therapeutics, which is developing AT132, funded the trial. Dr. Kuntz had no conflicts of interest.
SOURCE: Bönnemann CG et al. CNS-ICNA 2020, Abstract P.62.
FROM CNS-ICNA 2020
Blood test for Alzheimer’s disease comes to the clinic
, according to C2N Diagnostics, the company behind the test’s development. The availability of the noninvasive, easily administered test is being called a milestone in the early detection and diagnosis of Alzheimer’s disease.
The blood test “introduces a new option for patients, families, and the medical community that have eagerly awaited innovative tools to address Alzheimer’s troubling problems,” Joel B. Braunstein, MD, MBA, CEO of C2N Diagnostics, said in a press release.
“This is really an important advance,” said Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation (ADDF), which partially funded the development of the test, in a separate press release.
“You can now walk into your doctor’s office to get a blood test to help detect Alzheimer’s disease,” said Dr. Fillit. “This test answers a critical need for less costly and accessible diagnostic testing in memory and dementia care.”
A word of caution
However, Maria C. Carrillo, PhD, chief science officer, Alzheimer’s Association, highlighted the need for caution. The test is “very new,” experts have only “limited information” about it, and it is only available by prescription from a healthcare provider for patients with cognitive impairment, said Dr. Carrillo.
“The test is not [Food and Drug Administration] approved and it does not, on its own, diagnose Alzheimer’s disease,” added Dr. Carrillo. “Without FDA review, healthcare providers lack the agency’s guidance for how to use it when making decisions about a person’s health or treatment.”
Dr. Carrillo also noted that the test has only been studied in a limited number of individuals and that few data are available regarding underrepresented populations.
“As a result, it is not clear how accurate or generalizable the results are for all individuals and populations,” she noted.
Another factor to consider, said Dr. Carrillo, is that the test is not covered by insurance, including Medicare and Medicaid.
How it works
The test (PrecivityAD) is for use in patients with cognitive impairment. It requires a very small blood sample – as little as a teaspoon – from the patient’s forearm. The physician sends the sample to C2N Diagnostic’s specialized laboratory, where it is analyzed using mass spectrometry to measure concentrations of amyloid beta 42 and 40 and to detect the presence of apolipoprotein E isoforms.
The lab report, which is sent to the patient’s physician, details biomarker levels and provides an overall combined score, known as the Amyloid Probability Score, to assess the likelihood of low, intermediate, or high levels of amyloid plaque in the brain.
The company reports that, on the basis of data from 686 patients older than 60 years who had subjective cognitive impairment or dementia, the test correctly identified brain amyloid plaque status, as determined by quantitative amyloid positron-emission tomography (PET) scans, in 86% of the patients. In the analysis, the area under the curve for the receiver operating characteristic was 0.88.
The company notes that the test, the results of which require interpretation by a health care provider, is an important new tool to aid physicians in the evaluation process.
The new blood test is currently available in 45 states, the District of Columbia, and Puerto Rico.
C2N Diagnostics is moving ahead with development of a brain health panel to detect multiple blood-based markers for Alzheimer’s disease to aid in disease staging, treatment monitoring, and differential diagnosis.
The ADDF believes the path to approval of treatments of Alzheimer’s disease starts with a better diagnosis, Dr. Fillit said in his organization’s press release.
“Investing in biomarker research has been a core goal for the ADDF because reliable, accessible, and affordable biomarkers for Alzheimer’s diagnosis are critical to our ability to find drugs to prevent, slow, and even cure the disease. Our funding helped bring the first PET scan to market and now has helped bring the first blood test to market,” he said.
In addition to the ADDF, the National Institutes of Health, the GHR Foundation, and the BrightFocus Foundation contributed funding for the development of the amyloid blood test.
A version of this article originally appeared on Medscape.com.
, according to C2N Diagnostics, the company behind the test’s development. The availability of the noninvasive, easily administered test is being called a milestone in the early detection and diagnosis of Alzheimer’s disease.
The blood test “introduces a new option for patients, families, and the medical community that have eagerly awaited innovative tools to address Alzheimer’s troubling problems,” Joel B. Braunstein, MD, MBA, CEO of C2N Diagnostics, said in a press release.
“This is really an important advance,” said Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation (ADDF), which partially funded the development of the test, in a separate press release.
“You can now walk into your doctor’s office to get a blood test to help detect Alzheimer’s disease,” said Dr. Fillit. “This test answers a critical need for less costly and accessible diagnostic testing in memory and dementia care.”
A word of caution
However, Maria C. Carrillo, PhD, chief science officer, Alzheimer’s Association, highlighted the need for caution. The test is “very new,” experts have only “limited information” about it, and it is only available by prescription from a healthcare provider for patients with cognitive impairment, said Dr. Carrillo.
“The test is not [Food and Drug Administration] approved and it does not, on its own, diagnose Alzheimer’s disease,” added Dr. Carrillo. “Without FDA review, healthcare providers lack the agency’s guidance for how to use it when making decisions about a person’s health or treatment.”
Dr. Carrillo also noted that the test has only been studied in a limited number of individuals and that few data are available regarding underrepresented populations.
“As a result, it is not clear how accurate or generalizable the results are for all individuals and populations,” she noted.
Another factor to consider, said Dr. Carrillo, is that the test is not covered by insurance, including Medicare and Medicaid.
How it works
The test (PrecivityAD) is for use in patients with cognitive impairment. It requires a very small blood sample – as little as a teaspoon – from the patient’s forearm. The physician sends the sample to C2N Diagnostic’s specialized laboratory, where it is analyzed using mass spectrometry to measure concentrations of amyloid beta 42 and 40 and to detect the presence of apolipoprotein E isoforms.
The lab report, which is sent to the patient’s physician, details biomarker levels and provides an overall combined score, known as the Amyloid Probability Score, to assess the likelihood of low, intermediate, or high levels of amyloid plaque in the brain.
The company reports that, on the basis of data from 686 patients older than 60 years who had subjective cognitive impairment or dementia, the test correctly identified brain amyloid plaque status, as determined by quantitative amyloid positron-emission tomography (PET) scans, in 86% of the patients. In the analysis, the area under the curve for the receiver operating characteristic was 0.88.
The company notes that the test, the results of which require interpretation by a health care provider, is an important new tool to aid physicians in the evaluation process.
The new blood test is currently available in 45 states, the District of Columbia, and Puerto Rico.
C2N Diagnostics is moving ahead with development of a brain health panel to detect multiple blood-based markers for Alzheimer’s disease to aid in disease staging, treatment monitoring, and differential diagnosis.
The ADDF believes the path to approval of treatments of Alzheimer’s disease starts with a better diagnosis, Dr. Fillit said in his organization’s press release.
“Investing in biomarker research has been a core goal for the ADDF because reliable, accessible, and affordable biomarkers for Alzheimer’s diagnosis are critical to our ability to find drugs to prevent, slow, and even cure the disease. Our funding helped bring the first PET scan to market and now has helped bring the first blood test to market,” he said.
In addition to the ADDF, the National Institutes of Health, the GHR Foundation, and the BrightFocus Foundation contributed funding for the development of the amyloid blood test.
A version of this article originally appeared on Medscape.com.
, according to C2N Diagnostics, the company behind the test’s development. The availability of the noninvasive, easily administered test is being called a milestone in the early detection and diagnosis of Alzheimer’s disease.
The blood test “introduces a new option for patients, families, and the medical community that have eagerly awaited innovative tools to address Alzheimer’s troubling problems,” Joel B. Braunstein, MD, MBA, CEO of C2N Diagnostics, said in a press release.
“This is really an important advance,” said Howard Fillit, MD, founding executive director and chief science officer of the Alzheimer’s Drug Discovery Foundation (ADDF), which partially funded the development of the test, in a separate press release.
“You can now walk into your doctor’s office to get a blood test to help detect Alzheimer’s disease,” said Dr. Fillit. “This test answers a critical need for less costly and accessible diagnostic testing in memory and dementia care.”
A word of caution
However, Maria C. Carrillo, PhD, chief science officer, Alzheimer’s Association, highlighted the need for caution. The test is “very new,” experts have only “limited information” about it, and it is only available by prescription from a healthcare provider for patients with cognitive impairment, said Dr. Carrillo.
“The test is not [Food and Drug Administration] approved and it does not, on its own, diagnose Alzheimer’s disease,” added Dr. Carrillo. “Without FDA review, healthcare providers lack the agency’s guidance for how to use it when making decisions about a person’s health or treatment.”
Dr. Carrillo also noted that the test has only been studied in a limited number of individuals and that few data are available regarding underrepresented populations.
“As a result, it is not clear how accurate or generalizable the results are for all individuals and populations,” she noted.
Another factor to consider, said Dr. Carrillo, is that the test is not covered by insurance, including Medicare and Medicaid.
How it works
The test (PrecivityAD) is for use in patients with cognitive impairment. It requires a very small blood sample – as little as a teaspoon – from the patient’s forearm. The physician sends the sample to C2N Diagnostic’s specialized laboratory, where it is analyzed using mass spectrometry to measure concentrations of amyloid beta 42 and 40 and to detect the presence of apolipoprotein E isoforms.
The lab report, which is sent to the patient’s physician, details biomarker levels and provides an overall combined score, known as the Amyloid Probability Score, to assess the likelihood of low, intermediate, or high levels of amyloid plaque in the brain.
The company reports that, on the basis of data from 686 patients older than 60 years who had subjective cognitive impairment or dementia, the test correctly identified brain amyloid plaque status, as determined by quantitative amyloid positron-emission tomography (PET) scans, in 86% of the patients. In the analysis, the area under the curve for the receiver operating characteristic was 0.88.
The company notes that the test, the results of which require interpretation by a health care provider, is an important new tool to aid physicians in the evaluation process.
The new blood test is currently available in 45 states, the District of Columbia, and Puerto Rico.
C2N Diagnostics is moving ahead with development of a brain health panel to detect multiple blood-based markers for Alzheimer’s disease to aid in disease staging, treatment monitoring, and differential diagnosis.
The ADDF believes the path to approval of treatments of Alzheimer’s disease starts with a better diagnosis, Dr. Fillit said in his organization’s press release.
“Investing in biomarker research has been a core goal for the ADDF because reliable, accessible, and affordable biomarkers for Alzheimer’s diagnosis are critical to our ability to find drugs to prevent, slow, and even cure the disease. Our funding helped bring the first PET scan to market and now has helped bring the first blood test to market,” he said.
In addition to the ADDF, the National Institutes of Health, the GHR Foundation, and the BrightFocus Foundation contributed funding for the development of the amyloid blood test.
A version of this article originally appeared on Medscape.com.
A skin test for Parkinson’s disease diagnosis?
a new study suggests. For the study, researchers used a chemical assay to detect clumping of the protein alpha-synuclein, a hallmark of Parkinson’s disease, in autopsy skin samples taken from patients who had Parkinson’s disease confirmed by brain pathology and from controls without the disease. The test showed a high degree of sensitivity and specificity for the diagnosis of Parkinson’s disease.
The study was published online in Movement Disorders.
“This test has a lot of promise,” said senior author Anumantha Kanthasamy, PhD, professor of biomedical sciences at Iowa State University in Ames. “At present there are no peripheral biomarkers for Parkinson’s disease. The current diagnosis is just based on symptoms, and the symptoms can be similar to many other neurological diseases,” he added. “It can take many years to establish a correct diagnosis and the accuracy is low even with experienced neurologists.”
If the current results can be replicated in samples from live patients and in those with very early stages of Parkinson’s disease, a skin test could allow early diagnosis and the possibility of starting preventive treatments to slow disease progression before symptoms develop too severely, the researchers suggest.
Sensitive and specific test
The blinded study used a seeding assay – used previously to detect misfolded proteins in prion diseases – to analyze 50 skin samples provided by the Arizona Study of Aging and Neurodegenerative Disorders/Brain and Body Donation Program based at Banner Sun Health Research Institute in Sun City.
Half of the skin samples came from patients with Parkinson’s disease and half came from people without neurologic disease. The protein assay correctly diagnosed 24 out of 25 patients with Parkinson’s disease and only one of the 25 controls had the protein clumping.
“At present, the only way to definitely diagnose Parkinson’s disease is on autopsy – by the detection of alpha-synuclein clumps [Lewy bodies] in the brain,” commented Charles Adler, MD, professor of neurology at Mayo Clinic Arizona in Scottsdale and a coinvestigator of the study. “In our research, we have also seen clumping of alpha-synuclein in many other organs including submandibular gland, colon, skin, heart, and stomach, but in terms of access, the skin is probably the easiest source.”
In this study, “we found this seeding assay for alpha-synuclein clumps to be extremely sensitive and specific in the diagnosis of Parkinson’s disease,” he added. “This is very valuable data as we have samples from patients with autopsy-validated Parkinson’s disease.”
A reliable biomarker?
The researchers are now starting a study in living patients with funding from the National Institutes of Health in which they will repeat the process comparing skin samples from patients with clinically diagnosed Parkinson’s disease and controls.
“We need to know whether analyzing alpha-synuclein clumping in skin biopsies from live patients with Parkinson’s disease would serve as a reliable biomarker for disease progression. Will clumping of this protein in skin samples increase over time and does it correspond with disease progression?” Dr. Adler said.
In future they are also hoping to test individuals who have not yet developed Parkinson’s disease but may have some prodromal type symptoms and to test whether this assay could measure a treatment effect of drug therapy.
Dr. Adler noted that they are currently conducting an autopsy study of skin samples from individuals who did not have clinical Parkinson’s disease when alive but in whom Lewy bodies have been found postmortem.
“This suggests that the disease pathology starts before Parkinson’s symptoms develop, and in the future, if we can diagnose Parkinson’s disease earlier then we may be able to stop progression,” he said.
“There is a long list of compounds that have been studied to try and slow progression but haven’t shown benefits, but by the time patients develop symptoms they already have significant disease and [have] lost most of their dopamine neurons,” he added. “If we could backtrack by 10 years, then these drugs may well make a difference.”
Dr. Adler also noted that currently more advanced patients may undergo invasive procedures such as deep brain stimulation or surgery. “It is of utmost importance that they have an accurate diagnosis before being subjected to such procedures.”
In addition, he pointed out that an accurate test would help the drug development process. “It is vitally important to enroll patients with an accurate diagnosis in clinical trials of new drugs. At present, a large percentage of patients in these trials may not actually have Parkinson’s disease, which makes it very difficult to show a treatment effect.”
Important step, but preliminary
Commenting on the research, James Beck, PhD, chief scientific officer of the Parkinson’s Foundation, said the study “is an important step toward the creation of a new way to potentially diagnose Parkinson’s disease.”
But he cautioned that this is a preliminary study. “To really confirm the possibility of using this approach for diagnosing Parkinson’s disease, a larger study will be necessary. And it will be important to test this in a population with early disease – the most difficult group to accurately diagnose.”
Also commenting on the findings, Beate Ritz, MD, PhD, an epidemiologist at UCLA Fielding School of Public Health in Los Angeles, who is part of a team also working on ways to measure abnormal alpha-synuclein to diagnose Parkinson’s disease, described the current study of skin samples as “pretty nifty.”
“Their research shows clearly that they can distinguish between patients with Parkinson’s disease and controls in this way,” she said. “The big advantage of this study is that they have brain pathology, so they know exactly which individuals had Parkinson’s disease.”
Dr. Ritz is working with Gal Bitan, PhD, from the UCLA Brain Research Institute on a potential blood test to measure abnormal alpha-synuclein.
Dr. Ritz explained that it is not possible to measure alpha-synuclein pathology in regular blood samples as it is expressed normally in red blood cells, but they are measuring the protein and its more toxic phosphorylated form from exosomes, which contain the waste discarded by cells using technology that determines the origin of these exosomes.
“Alpha-synuclein itself is not a problem. It is the way it misfolds that causes toxicity and disrupts the workings of the cell,” Dr. Ritz added. “In Parkinson’s disease, it is particularly toxic to dopaminergic neurons, and in multiple system atrophy, it is toxic to glial cells, so if we can identify the source of the protein then that could be helpful.”
The study was funded by the National Institutes of Health and the US Army Medical Research Materiel Command. The study authors, Dr. Beck, and Dr. Ritz have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
a new study suggests. For the study, researchers used a chemical assay to detect clumping of the protein alpha-synuclein, a hallmark of Parkinson’s disease, in autopsy skin samples taken from patients who had Parkinson’s disease confirmed by brain pathology and from controls without the disease. The test showed a high degree of sensitivity and specificity for the diagnosis of Parkinson’s disease.
The study was published online in Movement Disorders.
“This test has a lot of promise,” said senior author Anumantha Kanthasamy, PhD, professor of biomedical sciences at Iowa State University in Ames. “At present there are no peripheral biomarkers for Parkinson’s disease. The current diagnosis is just based on symptoms, and the symptoms can be similar to many other neurological diseases,” he added. “It can take many years to establish a correct diagnosis and the accuracy is low even with experienced neurologists.”
If the current results can be replicated in samples from live patients and in those with very early stages of Parkinson’s disease, a skin test could allow early diagnosis and the possibility of starting preventive treatments to slow disease progression before symptoms develop too severely, the researchers suggest.
Sensitive and specific test
The blinded study used a seeding assay – used previously to detect misfolded proteins in prion diseases – to analyze 50 skin samples provided by the Arizona Study of Aging and Neurodegenerative Disorders/Brain and Body Donation Program based at Banner Sun Health Research Institute in Sun City.
Half of the skin samples came from patients with Parkinson’s disease and half came from people without neurologic disease. The protein assay correctly diagnosed 24 out of 25 patients with Parkinson’s disease and only one of the 25 controls had the protein clumping.
“At present, the only way to definitely diagnose Parkinson’s disease is on autopsy – by the detection of alpha-synuclein clumps [Lewy bodies] in the brain,” commented Charles Adler, MD, professor of neurology at Mayo Clinic Arizona in Scottsdale and a coinvestigator of the study. “In our research, we have also seen clumping of alpha-synuclein in many other organs including submandibular gland, colon, skin, heart, and stomach, but in terms of access, the skin is probably the easiest source.”
In this study, “we found this seeding assay for alpha-synuclein clumps to be extremely sensitive and specific in the diagnosis of Parkinson’s disease,” he added. “This is very valuable data as we have samples from patients with autopsy-validated Parkinson’s disease.”
A reliable biomarker?
The researchers are now starting a study in living patients with funding from the National Institutes of Health in which they will repeat the process comparing skin samples from patients with clinically diagnosed Parkinson’s disease and controls.
“We need to know whether analyzing alpha-synuclein clumping in skin biopsies from live patients with Parkinson’s disease would serve as a reliable biomarker for disease progression. Will clumping of this protein in skin samples increase over time and does it correspond with disease progression?” Dr. Adler said.
In future they are also hoping to test individuals who have not yet developed Parkinson’s disease but may have some prodromal type symptoms and to test whether this assay could measure a treatment effect of drug therapy.
Dr. Adler noted that they are currently conducting an autopsy study of skin samples from individuals who did not have clinical Parkinson’s disease when alive but in whom Lewy bodies have been found postmortem.
“This suggests that the disease pathology starts before Parkinson’s symptoms develop, and in the future, if we can diagnose Parkinson’s disease earlier then we may be able to stop progression,” he said.
“There is a long list of compounds that have been studied to try and slow progression but haven’t shown benefits, but by the time patients develop symptoms they already have significant disease and [have] lost most of their dopamine neurons,” he added. “If we could backtrack by 10 years, then these drugs may well make a difference.”
Dr. Adler also noted that currently more advanced patients may undergo invasive procedures such as deep brain stimulation or surgery. “It is of utmost importance that they have an accurate diagnosis before being subjected to such procedures.”
In addition, he pointed out that an accurate test would help the drug development process. “It is vitally important to enroll patients with an accurate diagnosis in clinical trials of new drugs. At present, a large percentage of patients in these trials may not actually have Parkinson’s disease, which makes it very difficult to show a treatment effect.”
Important step, but preliminary
Commenting on the research, James Beck, PhD, chief scientific officer of the Parkinson’s Foundation, said the study “is an important step toward the creation of a new way to potentially diagnose Parkinson’s disease.”
But he cautioned that this is a preliminary study. “To really confirm the possibility of using this approach for diagnosing Parkinson’s disease, a larger study will be necessary. And it will be important to test this in a population with early disease – the most difficult group to accurately diagnose.”
Also commenting on the findings, Beate Ritz, MD, PhD, an epidemiologist at UCLA Fielding School of Public Health in Los Angeles, who is part of a team also working on ways to measure abnormal alpha-synuclein to diagnose Parkinson’s disease, described the current study of skin samples as “pretty nifty.”
“Their research shows clearly that they can distinguish between patients with Parkinson’s disease and controls in this way,” she said. “The big advantage of this study is that they have brain pathology, so they know exactly which individuals had Parkinson’s disease.”
Dr. Ritz is working with Gal Bitan, PhD, from the UCLA Brain Research Institute on a potential blood test to measure abnormal alpha-synuclein.
Dr. Ritz explained that it is not possible to measure alpha-synuclein pathology in regular blood samples as it is expressed normally in red blood cells, but they are measuring the protein and its more toxic phosphorylated form from exosomes, which contain the waste discarded by cells using technology that determines the origin of these exosomes.
“Alpha-synuclein itself is not a problem. It is the way it misfolds that causes toxicity and disrupts the workings of the cell,” Dr. Ritz added. “In Parkinson’s disease, it is particularly toxic to dopaminergic neurons, and in multiple system atrophy, it is toxic to glial cells, so if we can identify the source of the protein then that could be helpful.”
The study was funded by the National Institutes of Health and the US Army Medical Research Materiel Command. The study authors, Dr. Beck, and Dr. Ritz have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
a new study suggests. For the study, researchers used a chemical assay to detect clumping of the protein alpha-synuclein, a hallmark of Parkinson’s disease, in autopsy skin samples taken from patients who had Parkinson’s disease confirmed by brain pathology and from controls without the disease. The test showed a high degree of sensitivity and specificity for the diagnosis of Parkinson’s disease.
The study was published online in Movement Disorders.
“This test has a lot of promise,” said senior author Anumantha Kanthasamy, PhD, professor of biomedical sciences at Iowa State University in Ames. “At present there are no peripheral biomarkers for Parkinson’s disease. The current diagnosis is just based on symptoms, and the symptoms can be similar to many other neurological diseases,” he added. “It can take many years to establish a correct diagnosis and the accuracy is low even with experienced neurologists.”
If the current results can be replicated in samples from live patients and in those with very early stages of Parkinson’s disease, a skin test could allow early diagnosis and the possibility of starting preventive treatments to slow disease progression before symptoms develop too severely, the researchers suggest.
Sensitive and specific test
The blinded study used a seeding assay – used previously to detect misfolded proteins in prion diseases – to analyze 50 skin samples provided by the Arizona Study of Aging and Neurodegenerative Disorders/Brain and Body Donation Program based at Banner Sun Health Research Institute in Sun City.
Half of the skin samples came from patients with Parkinson’s disease and half came from people without neurologic disease. The protein assay correctly diagnosed 24 out of 25 patients with Parkinson’s disease and only one of the 25 controls had the protein clumping.
“At present, the only way to definitely diagnose Parkinson’s disease is on autopsy – by the detection of alpha-synuclein clumps [Lewy bodies] in the brain,” commented Charles Adler, MD, professor of neurology at Mayo Clinic Arizona in Scottsdale and a coinvestigator of the study. “In our research, we have also seen clumping of alpha-synuclein in many other organs including submandibular gland, colon, skin, heart, and stomach, but in terms of access, the skin is probably the easiest source.”
In this study, “we found this seeding assay for alpha-synuclein clumps to be extremely sensitive and specific in the diagnosis of Parkinson’s disease,” he added. “This is very valuable data as we have samples from patients with autopsy-validated Parkinson’s disease.”
A reliable biomarker?
The researchers are now starting a study in living patients with funding from the National Institutes of Health in which they will repeat the process comparing skin samples from patients with clinically diagnosed Parkinson’s disease and controls.
“We need to know whether analyzing alpha-synuclein clumping in skin biopsies from live patients with Parkinson’s disease would serve as a reliable biomarker for disease progression. Will clumping of this protein in skin samples increase over time and does it correspond with disease progression?” Dr. Adler said.
In future they are also hoping to test individuals who have not yet developed Parkinson’s disease but may have some prodromal type symptoms and to test whether this assay could measure a treatment effect of drug therapy.
Dr. Adler noted that they are currently conducting an autopsy study of skin samples from individuals who did not have clinical Parkinson’s disease when alive but in whom Lewy bodies have been found postmortem.
“This suggests that the disease pathology starts before Parkinson’s symptoms develop, and in the future, if we can diagnose Parkinson’s disease earlier then we may be able to stop progression,” he said.
“There is a long list of compounds that have been studied to try and slow progression but haven’t shown benefits, but by the time patients develop symptoms they already have significant disease and [have] lost most of their dopamine neurons,” he added. “If we could backtrack by 10 years, then these drugs may well make a difference.”
Dr. Adler also noted that currently more advanced patients may undergo invasive procedures such as deep brain stimulation or surgery. “It is of utmost importance that they have an accurate diagnosis before being subjected to such procedures.”
In addition, he pointed out that an accurate test would help the drug development process. “It is vitally important to enroll patients with an accurate diagnosis in clinical trials of new drugs. At present, a large percentage of patients in these trials may not actually have Parkinson’s disease, which makes it very difficult to show a treatment effect.”
Important step, but preliminary
Commenting on the research, James Beck, PhD, chief scientific officer of the Parkinson’s Foundation, said the study “is an important step toward the creation of a new way to potentially diagnose Parkinson’s disease.”
But he cautioned that this is a preliminary study. “To really confirm the possibility of using this approach for diagnosing Parkinson’s disease, a larger study will be necessary. And it will be important to test this in a population with early disease – the most difficult group to accurately diagnose.”
Also commenting on the findings, Beate Ritz, MD, PhD, an epidemiologist at UCLA Fielding School of Public Health in Los Angeles, who is part of a team also working on ways to measure abnormal alpha-synuclein to diagnose Parkinson’s disease, described the current study of skin samples as “pretty nifty.”
“Their research shows clearly that they can distinguish between patients with Parkinson’s disease and controls in this way,” she said. “The big advantage of this study is that they have brain pathology, so they know exactly which individuals had Parkinson’s disease.”
Dr. Ritz is working with Gal Bitan, PhD, from the UCLA Brain Research Institute on a potential blood test to measure abnormal alpha-synuclein.
Dr. Ritz explained that it is not possible to measure alpha-synuclein pathology in regular blood samples as it is expressed normally in red blood cells, but they are measuring the protein and its more toxic phosphorylated form from exosomes, which contain the waste discarded by cells using technology that determines the origin of these exosomes.
“Alpha-synuclein itself is not a problem. It is the way it misfolds that causes toxicity and disrupts the workings of the cell,” Dr. Ritz added. “In Parkinson’s disease, it is particularly toxic to dopaminergic neurons, and in multiple system atrophy, it is toxic to glial cells, so if we can identify the source of the protein then that could be helpful.”
The study was funded by the National Institutes of Health and the US Army Medical Research Materiel Command. The study authors, Dr. Beck, and Dr. Ritz have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM MOVEMENT DISORDERS
‘Landmark’ study pushed detection of covert consciousness in TBI
Compelling advances in the ability to detect signs of consciousness in unconscious patients who have experienced traumatic brain injury (TBI) are leading to unprecedented changes in the field. There is now hope of improving outcomes and even sparing lives of patients who may otherwise have been mistakenly assessed as having no chance of recovery.
That research, published in the New England Journal of Medicine in June 2019, linked the promising signals of consciousness in comatose patients, detected only on imaging, with remarkable outcomes a year later.
“This was a landmark study,” said Brian L. Edlow, MD, in a presentation on the issue of covert consciousness at the virtual annual meeting of the American Neurological Association.
“Importantly, it is the first compelling evidence that early detection of covert consciousness also predicts 1-year outcomes in the Glasgow Outcome Scale Extended (GOSE), showing that covert consciousness in the ICU appears to be relevant for predicting long-term outcomes,” said Dr. Edlow, who is associate director of the Center for Neurotechnology and Neurorecovery, Massachusetts General Hospital, in Boston.
The researchers showed that 15% of unconscious patients with acute brain injury in the study exhibited significant brain activity on EEG in response to stimuli that included verbal commands such as envisioning that they are playing tennis.
Although other studies have shown similar effects with task-based stimuli, the New England Journal of Medicine study further showed that a year later, the patients who had shown signs of covert consciousness, also called “cognitive motor dissociation” (CMD), were significantly more likely to have a good functional outcome, said the study’s senior author, Jan Claassen, MD, director of critical care neurology at Columbia University, New York, who also presented at the ANA session.
“Importantly, a year later after injury, we found that 44% of patients with CMD and only 14% of non-CMD patients had a good functional outcome, defined as a GOSE score indicating a state where they can at least take care of themselves for 8 hours in a day,” he said.
“[Whether] these patients in a CMD state represent a parallel state or a transitory state on the road to recovery remains to be shown,” he said.
Jennifer Frontera, MD, a professor in the department of neurology at NYU Langone Health in New York and comoderator of the session, agreed that the research is “remarkable.”
“Also,” she said, “it is practical, since many could potentially apply and validate his algorithms, since EEG technology is portable and widely available.”
Research has ushered in a ‘sea change’ in neurocritical care
The research has helped push forward recommendations on the treatment of unconscious patients, Dr. Edlow said. “This has led to a sea change in our field just over the last 2 years, with multiple guidelines published suggesting that it may be time for us to consider incorporating task-based fMRI and EEG techniques into our clinical assessment of patients with disorders of consciousness,” Dr. Edlow said.
Among those updating their recommendations was the American Academy of Neurology, which revised guidelines on practice parameters for patients in a persistent vegetative state. Those guidelines had not been updated since 1995.
Although concluding that “no diagnostic assessment procedure had moderate or strong evidence for use,” the guidelines acknowledge that “it is possible that a positive electromyographic (EMG) response to command, EEG reactivity to sensory stimuli, laser-evoked potentials, and the Perturbational Complexity Index can distinguish a minimally conscious state from vegetative state/unresponsive wakefulness syndrome (VS/UWS).”
Earlier this year, the European Academy of Neurology followed suit with updated guidelines of its own. In the EAN guideline, the academy’s Panel on Coma, Disorders of Consciousness recommends that task-based fMRI, EEG, and other advanced assessments be performed as part of a composite assessment of consciousness and that a patient’s best performance or highest level of consciousness on any of those tests should be a reflection of their diagnosis, Dr. Edlow explained.
“What this means is that our field is moving toward a multimodal assessment of consciousness in the ICU as well as beyond, in the subacute to chronic setting, whereby the behavioral exam, advanced DG, and advanced MRI methods all also contribute to the diagnosis of consciousness,” he said.
The standard for assessment of disorders of consciousness is the Coma Recovery Scale–Revised, with a 25-item scale for diagnosis, prediction of outcome, and assessment of potential treatment efficacy.
But much uncertainty can remain despite the assessment, Dr. Claassen said. “Behavioral assessments of patients with acute brain injury are challenging because examinations fluctuate, and there’s variability between assessors,” he said. “Nevertheless, patients and their families demand guidance from us.”
Dr. Edlow pointed out that the largest study to date of the causes of death among patients with TBI in the ICU underscores the need for better assessments.
The study of more than 600 patients at six level l trauma centers in Canada showed that 70% of patients who died in the ICU from TBI did so as the result of the withdrawal of life-sustaining therapy. However, only about a half (57%) had an unreactive pupil, and only about a quarter (23.7%) had evidence of herniation on CT, findings that are commonly associated with a poor prognosis.
“What emerges from this is that the manner in which the clinicians communicated the prognosis to families was a primary determinant of decisions to withdraw life-sustaining therapy,” Dr. Edlow said.
Negative response not necessarily conclusive
Dr. Edlow added a word of caution that the science is still far from perfect. He noted that, for 25% of healthy patients who are given a motor imagery task, neuroimaging might not show a response, implying that the lack of a signal may not be conclusive.
He described the case of a patient who was comatose at the time she was scanned on day 3 after injury and who showed no responses to language, music, or motor imagery during the MRI, yet a year later, she was functionally independent, back in the workforce, and had very few residual symptoms from her trauma.
“So if a patient does not show a response, that does not prove the patient is not conscious, and it does not prove that the patient is likely to have a poor outcome,” Dr. Edlow said. Such cases underscore the need for more advances in understanding the inner workings of brain injury.
Dr. Edlow and his colleagues are embarking on a trial of the effects of intravenous methylphenidate in targeting the stimulation of dopaminergic circuits within the subcortical ascending arousal network in patients with severe brain injuries.
“The scientific premise of the trial is that personalized brain network mapping in the ICU can identify patients whose connectomes are amenable to neuromodulation,” Dr. Edlow and his colleague report in an article in Neurocritical Care.
The trial, called STIMPACT (Stimulant Therapy Targeted to Individualized Connectivity Maps to Promote ReACTivation of Consciousness), is part of the newly launched Connectome-based Clinical Trial Platform, which the authors describe as “a new paradigm for developing and testing targeted therapies that promote early recovery of consciousness in the ICU.”
Such efforts are essential, given the high stakes of TBI outcomes, Dr. Edlow said.
“Let’s be clear about the stakes of an incorrect prognosis,” he said. “If we’re overly pessimistic, then a patient who could have potential for meaningful recovery will likely die in our ICU. On the other hand, if we are overly optimistic, then a patient could end up in a vegetative or minimally conscious state that he or she may never have found to be acceptable,” he said.
Access to technologies a ‘civil right?’
Some ethicists in the field are recommending that patients be given access to the advanced techniques as a civil right, similar to the rights described in the Convention on the Rights of Persons With Disabilities, which was adopted by the United Nations in 2008, Dr. Edlow noted.
“So the question that we as clinicians are going to face moving forward from an ethical standpoint is, if we have access to these techniques, is it an ethical obligation to offer them now?” he said.
Dr. Edlow underscored the need to consider the reality that “there are profound issues relating to resource allocation and access to these advanced techniques, but we’re going to have to consider this together as we move forward.”
Dr. Edlow has received funding from the National Institutes of Health. Dr. Claassen is a minority shareholder with ICE Neurosystems. Dr. Frontera has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Compelling advances in the ability to detect signs of consciousness in unconscious patients who have experienced traumatic brain injury (TBI) are leading to unprecedented changes in the field. There is now hope of improving outcomes and even sparing lives of patients who may otherwise have been mistakenly assessed as having no chance of recovery.
That research, published in the New England Journal of Medicine in June 2019, linked the promising signals of consciousness in comatose patients, detected only on imaging, with remarkable outcomes a year later.
“This was a landmark study,” said Brian L. Edlow, MD, in a presentation on the issue of covert consciousness at the virtual annual meeting of the American Neurological Association.
“Importantly, it is the first compelling evidence that early detection of covert consciousness also predicts 1-year outcomes in the Glasgow Outcome Scale Extended (GOSE), showing that covert consciousness in the ICU appears to be relevant for predicting long-term outcomes,” said Dr. Edlow, who is associate director of the Center for Neurotechnology and Neurorecovery, Massachusetts General Hospital, in Boston.
The researchers showed that 15% of unconscious patients with acute brain injury in the study exhibited significant brain activity on EEG in response to stimuli that included verbal commands such as envisioning that they are playing tennis.
Although other studies have shown similar effects with task-based stimuli, the New England Journal of Medicine study further showed that a year later, the patients who had shown signs of covert consciousness, also called “cognitive motor dissociation” (CMD), were significantly more likely to have a good functional outcome, said the study’s senior author, Jan Claassen, MD, director of critical care neurology at Columbia University, New York, who also presented at the ANA session.
“Importantly, a year later after injury, we found that 44% of patients with CMD and only 14% of non-CMD patients had a good functional outcome, defined as a GOSE score indicating a state where they can at least take care of themselves for 8 hours in a day,” he said.
“[Whether] these patients in a CMD state represent a parallel state or a transitory state on the road to recovery remains to be shown,” he said.
Jennifer Frontera, MD, a professor in the department of neurology at NYU Langone Health in New York and comoderator of the session, agreed that the research is “remarkable.”
“Also,” she said, “it is practical, since many could potentially apply and validate his algorithms, since EEG technology is portable and widely available.”
Research has ushered in a ‘sea change’ in neurocritical care
The research has helped push forward recommendations on the treatment of unconscious patients, Dr. Edlow said. “This has led to a sea change in our field just over the last 2 years, with multiple guidelines published suggesting that it may be time for us to consider incorporating task-based fMRI and EEG techniques into our clinical assessment of patients with disorders of consciousness,” Dr. Edlow said.
Among those updating their recommendations was the American Academy of Neurology, which revised guidelines on practice parameters for patients in a persistent vegetative state. Those guidelines had not been updated since 1995.
Although concluding that “no diagnostic assessment procedure had moderate or strong evidence for use,” the guidelines acknowledge that “it is possible that a positive electromyographic (EMG) response to command, EEG reactivity to sensory stimuli, laser-evoked potentials, and the Perturbational Complexity Index can distinguish a minimally conscious state from vegetative state/unresponsive wakefulness syndrome (VS/UWS).”
Earlier this year, the European Academy of Neurology followed suit with updated guidelines of its own. In the EAN guideline, the academy’s Panel on Coma, Disorders of Consciousness recommends that task-based fMRI, EEG, and other advanced assessments be performed as part of a composite assessment of consciousness and that a patient’s best performance or highest level of consciousness on any of those tests should be a reflection of their diagnosis, Dr. Edlow explained.
“What this means is that our field is moving toward a multimodal assessment of consciousness in the ICU as well as beyond, in the subacute to chronic setting, whereby the behavioral exam, advanced DG, and advanced MRI methods all also contribute to the diagnosis of consciousness,” he said.
The standard for assessment of disorders of consciousness is the Coma Recovery Scale–Revised, with a 25-item scale for diagnosis, prediction of outcome, and assessment of potential treatment efficacy.
But much uncertainty can remain despite the assessment, Dr. Claassen said. “Behavioral assessments of patients with acute brain injury are challenging because examinations fluctuate, and there’s variability between assessors,” he said. “Nevertheless, patients and their families demand guidance from us.”
Dr. Edlow pointed out that the largest study to date of the causes of death among patients with TBI in the ICU underscores the need for better assessments.
The study of more than 600 patients at six level l trauma centers in Canada showed that 70% of patients who died in the ICU from TBI did so as the result of the withdrawal of life-sustaining therapy. However, only about a half (57%) had an unreactive pupil, and only about a quarter (23.7%) had evidence of herniation on CT, findings that are commonly associated with a poor prognosis.
“What emerges from this is that the manner in which the clinicians communicated the prognosis to families was a primary determinant of decisions to withdraw life-sustaining therapy,” Dr. Edlow said.
Negative response not necessarily conclusive
Dr. Edlow added a word of caution that the science is still far from perfect. He noted that, for 25% of healthy patients who are given a motor imagery task, neuroimaging might not show a response, implying that the lack of a signal may not be conclusive.
He described the case of a patient who was comatose at the time she was scanned on day 3 after injury and who showed no responses to language, music, or motor imagery during the MRI, yet a year later, she was functionally independent, back in the workforce, and had very few residual symptoms from her trauma.
“So if a patient does not show a response, that does not prove the patient is not conscious, and it does not prove that the patient is likely to have a poor outcome,” Dr. Edlow said. Such cases underscore the need for more advances in understanding the inner workings of brain injury.
Dr. Edlow and his colleagues are embarking on a trial of the effects of intravenous methylphenidate in targeting the stimulation of dopaminergic circuits within the subcortical ascending arousal network in patients with severe brain injuries.
“The scientific premise of the trial is that personalized brain network mapping in the ICU can identify patients whose connectomes are amenable to neuromodulation,” Dr. Edlow and his colleague report in an article in Neurocritical Care.
The trial, called STIMPACT (Stimulant Therapy Targeted to Individualized Connectivity Maps to Promote ReACTivation of Consciousness), is part of the newly launched Connectome-based Clinical Trial Platform, which the authors describe as “a new paradigm for developing and testing targeted therapies that promote early recovery of consciousness in the ICU.”
Such efforts are essential, given the high stakes of TBI outcomes, Dr. Edlow said.
“Let’s be clear about the stakes of an incorrect prognosis,” he said. “If we’re overly pessimistic, then a patient who could have potential for meaningful recovery will likely die in our ICU. On the other hand, if we are overly optimistic, then a patient could end up in a vegetative or minimally conscious state that he or she may never have found to be acceptable,” he said.
Access to technologies a ‘civil right?’
Some ethicists in the field are recommending that patients be given access to the advanced techniques as a civil right, similar to the rights described in the Convention on the Rights of Persons With Disabilities, which was adopted by the United Nations in 2008, Dr. Edlow noted.
“So the question that we as clinicians are going to face moving forward from an ethical standpoint is, if we have access to these techniques, is it an ethical obligation to offer them now?” he said.
Dr. Edlow underscored the need to consider the reality that “there are profound issues relating to resource allocation and access to these advanced techniques, but we’re going to have to consider this together as we move forward.”
Dr. Edlow has received funding from the National Institutes of Health. Dr. Claassen is a minority shareholder with ICE Neurosystems. Dr. Frontera has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Compelling advances in the ability to detect signs of consciousness in unconscious patients who have experienced traumatic brain injury (TBI) are leading to unprecedented changes in the field. There is now hope of improving outcomes and even sparing lives of patients who may otherwise have been mistakenly assessed as having no chance of recovery.
That research, published in the New England Journal of Medicine in June 2019, linked the promising signals of consciousness in comatose patients, detected only on imaging, with remarkable outcomes a year later.
“This was a landmark study,” said Brian L. Edlow, MD, in a presentation on the issue of covert consciousness at the virtual annual meeting of the American Neurological Association.
“Importantly, it is the first compelling evidence that early detection of covert consciousness also predicts 1-year outcomes in the Glasgow Outcome Scale Extended (GOSE), showing that covert consciousness in the ICU appears to be relevant for predicting long-term outcomes,” said Dr. Edlow, who is associate director of the Center for Neurotechnology and Neurorecovery, Massachusetts General Hospital, in Boston.
The researchers showed that 15% of unconscious patients with acute brain injury in the study exhibited significant brain activity on EEG in response to stimuli that included verbal commands such as envisioning that they are playing tennis.
Although other studies have shown similar effects with task-based stimuli, the New England Journal of Medicine study further showed that a year later, the patients who had shown signs of covert consciousness, also called “cognitive motor dissociation” (CMD), were significantly more likely to have a good functional outcome, said the study’s senior author, Jan Claassen, MD, director of critical care neurology at Columbia University, New York, who also presented at the ANA session.
“Importantly, a year later after injury, we found that 44% of patients with CMD and only 14% of non-CMD patients had a good functional outcome, defined as a GOSE score indicating a state where they can at least take care of themselves for 8 hours in a day,” he said.
“[Whether] these patients in a CMD state represent a parallel state or a transitory state on the road to recovery remains to be shown,” he said.
Jennifer Frontera, MD, a professor in the department of neurology at NYU Langone Health in New York and comoderator of the session, agreed that the research is “remarkable.”
“Also,” she said, “it is practical, since many could potentially apply and validate his algorithms, since EEG technology is portable and widely available.”
Research has ushered in a ‘sea change’ in neurocritical care
The research has helped push forward recommendations on the treatment of unconscious patients, Dr. Edlow said. “This has led to a sea change in our field just over the last 2 years, with multiple guidelines published suggesting that it may be time for us to consider incorporating task-based fMRI and EEG techniques into our clinical assessment of patients with disorders of consciousness,” Dr. Edlow said.
Among those updating their recommendations was the American Academy of Neurology, which revised guidelines on practice parameters for patients in a persistent vegetative state. Those guidelines had not been updated since 1995.
Although concluding that “no diagnostic assessment procedure had moderate or strong evidence for use,” the guidelines acknowledge that “it is possible that a positive electromyographic (EMG) response to command, EEG reactivity to sensory stimuli, laser-evoked potentials, and the Perturbational Complexity Index can distinguish a minimally conscious state from vegetative state/unresponsive wakefulness syndrome (VS/UWS).”
Earlier this year, the European Academy of Neurology followed suit with updated guidelines of its own. In the EAN guideline, the academy’s Panel on Coma, Disorders of Consciousness recommends that task-based fMRI, EEG, and other advanced assessments be performed as part of a composite assessment of consciousness and that a patient’s best performance or highest level of consciousness on any of those tests should be a reflection of their diagnosis, Dr. Edlow explained.
“What this means is that our field is moving toward a multimodal assessment of consciousness in the ICU as well as beyond, in the subacute to chronic setting, whereby the behavioral exam, advanced DG, and advanced MRI methods all also contribute to the diagnosis of consciousness,” he said.
The standard for assessment of disorders of consciousness is the Coma Recovery Scale–Revised, with a 25-item scale for diagnosis, prediction of outcome, and assessment of potential treatment efficacy.
But much uncertainty can remain despite the assessment, Dr. Claassen said. “Behavioral assessments of patients with acute brain injury are challenging because examinations fluctuate, and there’s variability between assessors,” he said. “Nevertheless, patients and their families demand guidance from us.”
Dr. Edlow pointed out that the largest study to date of the causes of death among patients with TBI in the ICU underscores the need for better assessments.
The study of more than 600 patients at six level l trauma centers in Canada showed that 70% of patients who died in the ICU from TBI did so as the result of the withdrawal of life-sustaining therapy. However, only about a half (57%) had an unreactive pupil, and only about a quarter (23.7%) had evidence of herniation on CT, findings that are commonly associated with a poor prognosis.
“What emerges from this is that the manner in which the clinicians communicated the prognosis to families was a primary determinant of decisions to withdraw life-sustaining therapy,” Dr. Edlow said.
Negative response not necessarily conclusive
Dr. Edlow added a word of caution that the science is still far from perfect. He noted that, for 25% of healthy patients who are given a motor imagery task, neuroimaging might not show a response, implying that the lack of a signal may not be conclusive.
He described the case of a patient who was comatose at the time she was scanned on day 3 after injury and who showed no responses to language, music, or motor imagery during the MRI, yet a year later, she was functionally independent, back in the workforce, and had very few residual symptoms from her trauma.
“So if a patient does not show a response, that does not prove the patient is not conscious, and it does not prove that the patient is likely to have a poor outcome,” Dr. Edlow said. Such cases underscore the need for more advances in understanding the inner workings of brain injury.
Dr. Edlow and his colleagues are embarking on a trial of the effects of intravenous methylphenidate in targeting the stimulation of dopaminergic circuits within the subcortical ascending arousal network in patients with severe brain injuries.
“The scientific premise of the trial is that personalized brain network mapping in the ICU can identify patients whose connectomes are amenable to neuromodulation,” Dr. Edlow and his colleague report in an article in Neurocritical Care.
The trial, called STIMPACT (Stimulant Therapy Targeted to Individualized Connectivity Maps to Promote ReACTivation of Consciousness), is part of the newly launched Connectome-based Clinical Trial Platform, which the authors describe as “a new paradigm for developing and testing targeted therapies that promote early recovery of consciousness in the ICU.”
Such efforts are essential, given the high stakes of TBI outcomes, Dr. Edlow said.
“Let’s be clear about the stakes of an incorrect prognosis,” he said. “If we’re overly pessimistic, then a patient who could have potential for meaningful recovery will likely die in our ICU. On the other hand, if we are overly optimistic, then a patient could end up in a vegetative or minimally conscious state that he or she may never have found to be acceptable,” he said.
Access to technologies a ‘civil right?’
Some ethicists in the field are recommending that patients be given access to the advanced techniques as a civil right, similar to the rights described in the Convention on the Rights of Persons With Disabilities, which was adopted by the United Nations in 2008, Dr. Edlow noted.
“So the question that we as clinicians are going to face moving forward from an ethical standpoint is, if we have access to these techniques, is it an ethical obligation to offer them now?” he said.
Dr. Edlow underscored the need to consider the reality that “there are profound issues relating to resource allocation and access to these advanced techniques, but we’re going to have to consider this together as we move forward.”
Dr. Edlow has received funding from the National Institutes of Health. Dr. Claassen is a minority shareholder with ICE Neurosystems. Dr. Frontera has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM ANA 2020
COVID-19 diagnosed on CTA scan in stroke patients
A routine scan used to evaluate some acute stroke patients can also detect SARS-CoV-2 infection in the upper lungs, a new study shows.
“As part of the stroke evaluation workup process, we were able to diagnose COVID-19 at the same time at no extra cost or additional workload,” lead author Charles Esenwa, MD, commented to Medscape Medical News. “This is an objective way to screen for COVID-19 in the acute stroke setting,” he added.
Esenwa is an assistant professor and a stroke neurologist at the Montefiore Medical Center/Albert Einstein College of Medicine in New York City.
He explained that, during the COVID-19 surge earlier this year, assessment of patients with severe acute stroke using computed tomography angiogram (CTA) scans – used to evaluate suitability for endovascular stroke therapy – also showed findings in the upper lung consistent with viral infection in some patients.
“We then assumed that these patients had COVID-19 and took extra precautions to keep them isolated and to protect staff involved in their care. It also allowed us to triage these patients more quickly than waiting for the COVID-19 swab test and arrange the most appropriate care for them,” Esenwa said.
The researchers have now gone back and analyzed their data on acute stroke patients who underwent CTA at their institution during the COVID-19 surge. They found that the changes identified in the lungs were highly specific for diagnosing SARS-CoV-2 infection.
The study was published online on Oct. 29 in Stroke.
“Stroke patients are normally screened for COVID-19 on hospitalization, but the swab test result can take several hours or longer to come back, and it is very useful for us to know if a patient could be infected,” Esenwa noted.
“When we do a CTA, we look at the blood vessels supplying the brain, but the scan also covers the top of the lung, as it starts at the aortic arch. We don’t normally look closely at that area, but we started to notice signs of active lung infection which could have been COVID-19,” he said. “For this paper, we went back to assess how accurate this approach actually was vs. the COVID-19 PCR test.”
The researchers report on 57 patients who presented to three Montefiore Health System hospitals in the Bronx, in New York City, with acute ischemic stroke and who underwent CTA of the head and neck in March and April 2020, the peak of the COVID-19 outbreak there. The patients also underwent PCR testing for COVID-19.
Results showed that 30 patients had a positive COVID-19 test result and that 27 had a negative result. Lung findings highly or very highly suspicious for COVID-19 pneumonia were identified during the CTA scan in 20 (67%) of the COVID-19–positive patients and in two (7%) of the COVID-19–negative patients.
These findings, when used in isolation, yielded a sensitivity of 0.67 and a specificity of 0.93. They had a positive predictive value of 0.19, a negative predictive value of 0.99, and accuracy of 0.92 for the diagnosis of COVID-19.
When apical lung assessment was combined with self-reported clinical symptoms of cough or dyspnea, sensitivity for the diagnosis of COVID-19 for patients presenting to the hospital for acute ischemic stroke increased to 0.83.
“We wondered whether looking at the whole lung would have found better results, but other studies which have done this actually found similar numbers to ours, so we think actually just looking at the top of the lungs, which can be seen in a stroke CTA, may be sufficient,” Esenwa said.
He emphasized the importance of establishing whether an acute stroke patient has COVID-19. “If we had a high suspicion of COVID-19 infection, we would take more precautions during any procedures, such as thrombectomy, and make sure to keep the patient isolated afterwards. It doesn’t necessarily affect the treatment given for stroke, but it affects the safety of the patients and everyone caring for them,” he commented.
Esenwa explained that intubation – which is sometime necessary during thrombectomy – can expose everyone in the room to aerosolized droplets. “So we would take much higher safety precautions if we thought the patient was COVID-19 positive,” he said.
“Early COVID-19 diagnosis also means patients can be given supportive treatment more quickly, admitted to ICU if appropriate, and we can all keep a close eye on pulmonary issues. So having that information is important in many ways,” he added.
Esenwa advises that any medical center that evaluates acute stroke patients for thrombectomy and is experiencing a COVID-19 surge can use this technique as a screening method for COVID-19.
He pointed out that the Montefiore Health System had a very high rate of COVID-19. That part of New York City was one of the worst hit areas of the world, and the CTA approach for identifying COVID-19 has been validated only in areas with such a high local incidence of COVID. If used in an area of lower prevalence, the accuracy would likely be less.
“We don’t know if this approach would work as well at times of low COVID-19 infection, where any lung findings would be more likely to be caused by other conditions, such as pneumonia due to other causes or congestive heart failure. So there would be more false positives,” Esenwa said.
“But when COVID-19 prevalence is high, the lung findings are much more likely to be a sign of COVID-19 infection. As COVID-19 numbers are now rising for a second time, it is likely to become a useful strategy again.”
The study was approved by the Albert Einstein College of Medicine/Montefiore Medical Center Institutional Review Board and had no external funding. Esenwa has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A routine scan used to evaluate some acute stroke patients can also detect SARS-CoV-2 infection in the upper lungs, a new study shows.
“As part of the stroke evaluation workup process, we were able to diagnose COVID-19 at the same time at no extra cost or additional workload,” lead author Charles Esenwa, MD, commented to Medscape Medical News. “This is an objective way to screen for COVID-19 in the acute stroke setting,” he added.
Esenwa is an assistant professor and a stroke neurologist at the Montefiore Medical Center/Albert Einstein College of Medicine in New York City.
He explained that, during the COVID-19 surge earlier this year, assessment of patients with severe acute stroke using computed tomography angiogram (CTA) scans – used to evaluate suitability for endovascular stroke therapy – also showed findings in the upper lung consistent with viral infection in some patients.
“We then assumed that these patients had COVID-19 and took extra precautions to keep them isolated and to protect staff involved in their care. It also allowed us to triage these patients more quickly than waiting for the COVID-19 swab test and arrange the most appropriate care for them,” Esenwa said.
The researchers have now gone back and analyzed their data on acute stroke patients who underwent CTA at their institution during the COVID-19 surge. They found that the changes identified in the lungs were highly specific for diagnosing SARS-CoV-2 infection.
The study was published online on Oct. 29 in Stroke.
“Stroke patients are normally screened for COVID-19 on hospitalization, but the swab test result can take several hours or longer to come back, and it is very useful for us to know if a patient could be infected,” Esenwa noted.
“When we do a CTA, we look at the blood vessels supplying the brain, but the scan also covers the top of the lung, as it starts at the aortic arch. We don’t normally look closely at that area, but we started to notice signs of active lung infection which could have been COVID-19,” he said. “For this paper, we went back to assess how accurate this approach actually was vs. the COVID-19 PCR test.”
The researchers report on 57 patients who presented to three Montefiore Health System hospitals in the Bronx, in New York City, with acute ischemic stroke and who underwent CTA of the head and neck in March and April 2020, the peak of the COVID-19 outbreak there. The patients also underwent PCR testing for COVID-19.
Results showed that 30 patients had a positive COVID-19 test result and that 27 had a negative result. Lung findings highly or very highly suspicious for COVID-19 pneumonia were identified during the CTA scan in 20 (67%) of the COVID-19–positive patients and in two (7%) of the COVID-19–negative patients.
These findings, when used in isolation, yielded a sensitivity of 0.67 and a specificity of 0.93. They had a positive predictive value of 0.19, a negative predictive value of 0.99, and accuracy of 0.92 for the diagnosis of COVID-19.
When apical lung assessment was combined with self-reported clinical symptoms of cough or dyspnea, sensitivity for the diagnosis of COVID-19 for patients presenting to the hospital for acute ischemic stroke increased to 0.83.
“We wondered whether looking at the whole lung would have found better results, but other studies which have done this actually found similar numbers to ours, so we think actually just looking at the top of the lungs, which can be seen in a stroke CTA, may be sufficient,” Esenwa said.
He emphasized the importance of establishing whether an acute stroke patient has COVID-19. “If we had a high suspicion of COVID-19 infection, we would take more precautions during any procedures, such as thrombectomy, and make sure to keep the patient isolated afterwards. It doesn’t necessarily affect the treatment given for stroke, but it affects the safety of the patients and everyone caring for them,” he commented.
Esenwa explained that intubation – which is sometime necessary during thrombectomy – can expose everyone in the room to aerosolized droplets. “So we would take much higher safety precautions if we thought the patient was COVID-19 positive,” he said.
“Early COVID-19 diagnosis also means patients can be given supportive treatment more quickly, admitted to ICU if appropriate, and we can all keep a close eye on pulmonary issues. So having that information is important in many ways,” he added.
Esenwa advises that any medical center that evaluates acute stroke patients for thrombectomy and is experiencing a COVID-19 surge can use this technique as a screening method for COVID-19.
He pointed out that the Montefiore Health System had a very high rate of COVID-19. That part of New York City was one of the worst hit areas of the world, and the CTA approach for identifying COVID-19 has been validated only in areas with such a high local incidence of COVID. If used in an area of lower prevalence, the accuracy would likely be less.
“We don’t know if this approach would work as well at times of low COVID-19 infection, where any lung findings would be more likely to be caused by other conditions, such as pneumonia due to other causes or congestive heart failure. So there would be more false positives,” Esenwa said.
“But when COVID-19 prevalence is high, the lung findings are much more likely to be a sign of COVID-19 infection. As COVID-19 numbers are now rising for a second time, it is likely to become a useful strategy again.”
The study was approved by the Albert Einstein College of Medicine/Montefiore Medical Center Institutional Review Board and had no external funding. Esenwa has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A routine scan used to evaluate some acute stroke patients can also detect SARS-CoV-2 infection in the upper lungs, a new study shows.
“As part of the stroke evaluation workup process, we were able to diagnose COVID-19 at the same time at no extra cost or additional workload,” lead author Charles Esenwa, MD, commented to Medscape Medical News. “This is an objective way to screen for COVID-19 in the acute stroke setting,” he added.
Esenwa is an assistant professor and a stroke neurologist at the Montefiore Medical Center/Albert Einstein College of Medicine in New York City.
He explained that, during the COVID-19 surge earlier this year, assessment of patients with severe acute stroke using computed tomography angiogram (CTA) scans – used to evaluate suitability for endovascular stroke therapy – also showed findings in the upper lung consistent with viral infection in some patients.
“We then assumed that these patients had COVID-19 and took extra precautions to keep them isolated and to protect staff involved in their care. It also allowed us to triage these patients more quickly than waiting for the COVID-19 swab test and arrange the most appropriate care for them,” Esenwa said.
The researchers have now gone back and analyzed their data on acute stroke patients who underwent CTA at their institution during the COVID-19 surge. They found that the changes identified in the lungs were highly specific for diagnosing SARS-CoV-2 infection.
The study was published online on Oct. 29 in Stroke.
“Stroke patients are normally screened for COVID-19 on hospitalization, but the swab test result can take several hours or longer to come back, and it is very useful for us to know if a patient could be infected,” Esenwa noted.
“When we do a CTA, we look at the blood vessels supplying the brain, but the scan also covers the top of the lung, as it starts at the aortic arch. We don’t normally look closely at that area, but we started to notice signs of active lung infection which could have been COVID-19,” he said. “For this paper, we went back to assess how accurate this approach actually was vs. the COVID-19 PCR test.”
The researchers report on 57 patients who presented to three Montefiore Health System hospitals in the Bronx, in New York City, with acute ischemic stroke and who underwent CTA of the head and neck in March and April 2020, the peak of the COVID-19 outbreak there. The patients also underwent PCR testing for COVID-19.
Results showed that 30 patients had a positive COVID-19 test result and that 27 had a negative result. Lung findings highly or very highly suspicious for COVID-19 pneumonia were identified during the CTA scan in 20 (67%) of the COVID-19–positive patients and in two (7%) of the COVID-19–negative patients.
These findings, when used in isolation, yielded a sensitivity of 0.67 and a specificity of 0.93. They had a positive predictive value of 0.19, a negative predictive value of 0.99, and accuracy of 0.92 for the diagnosis of COVID-19.
When apical lung assessment was combined with self-reported clinical symptoms of cough or dyspnea, sensitivity for the diagnosis of COVID-19 for patients presenting to the hospital for acute ischemic stroke increased to 0.83.
“We wondered whether looking at the whole lung would have found better results, but other studies which have done this actually found similar numbers to ours, so we think actually just looking at the top of the lungs, which can be seen in a stroke CTA, may be sufficient,” Esenwa said.
He emphasized the importance of establishing whether an acute stroke patient has COVID-19. “If we had a high suspicion of COVID-19 infection, we would take more precautions during any procedures, such as thrombectomy, and make sure to keep the patient isolated afterwards. It doesn’t necessarily affect the treatment given for stroke, but it affects the safety of the patients and everyone caring for them,” he commented.
Esenwa explained that intubation – which is sometime necessary during thrombectomy – can expose everyone in the room to aerosolized droplets. “So we would take much higher safety precautions if we thought the patient was COVID-19 positive,” he said.
“Early COVID-19 diagnosis also means patients can be given supportive treatment more quickly, admitted to ICU if appropriate, and we can all keep a close eye on pulmonary issues. So having that information is important in many ways,” he added.
Esenwa advises that any medical center that evaluates acute stroke patients for thrombectomy and is experiencing a COVID-19 surge can use this technique as a screening method for COVID-19.
He pointed out that the Montefiore Health System had a very high rate of COVID-19. That part of New York City was one of the worst hit areas of the world, and the CTA approach for identifying COVID-19 has been validated only in areas with such a high local incidence of COVID. If used in an area of lower prevalence, the accuracy would likely be less.
“We don’t know if this approach would work as well at times of low COVID-19 infection, where any lung findings would be more likely to be caused by other conditions, such as pneumonia due to other causes or congestive heart failure. So there would be more false positives,” Esenwa said.
“But when COVID-19 prevalence is high, the lung findings are much more likely to be a sign of COVID-19 infection. As COVID-19 numbers are now rising for a second time, it is likely to become a useful strategy again.”
The study was approved by the Albert Einstein College of Medicine/Montefiore Medical Center Institutional Review Board and had no external funding. Esenwa has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Inpatient opioid administration associated with postdischarge opioid use
Background: Efforts to reduce and monitor high-risk opioid prescribing have largely focused on outpatient prescribing with less empiric evaluation of inpatient administration. Little is known about the association of inpatient opioid administration and postdischarge opioid use.
Study design: Retrospective cohort.
Setting: 12 community and academic hospitals in Pennsylvania.
Synopsis: With electronic health record data from 2010-2014 to evaluate 148,068 opioid-naive patients aged 18 years and older, this study showed a relationship between inpatient opioid administration, specific patterns of inpatient opioid administration, and postdischarge opioid use. Specifically, inpatient opioid administration was associated with a 3.0% increase (95% CI, 2.8%-3.2%) in opioid use at 90 days post discharge. Additionally, inpatient opioid administration within 12 hours of hospital discharge was associated with a 3.6% increase (95% CI, 3.3%-3.9%) in opioid use at 90 days post discharge.
This observational study is prone to potential unmeasured confounders negating any clear causation. Rather, hospitalists should be aware of the increasing focus on inpatient opioid administration as it relates to outpatient opioid use, especially in the setting of the current opioid crisis.
Bottom line: Inpatient opioid administration and administration patterns are associated with 90-day postdischarge opioid use in opioid-naive patients.
Citation: Donohue JM et al. Patterns of opioid administration among opioid-naive inpatients and associations with postdischarge opioid use. Ann Intern Med. 2019 Jun 18:171(2):81-90.
Dr. Ledford is a hospitalist at Vanderbilt University Medical Center, Nashville, Tenn.
Background: Efforts to reduce and monitor high-risk opioid prescribing have largely focused on outpatient prescribing with less empiric evaluation of inpatient administration. Little is known about the association of inpatient opioid administration and postdischarge opioid use.
Study design: Retrospective cohort.
Setting: 12 community and academic hospitals in Pennsylvania.
Synopsis: With electronic health record data from 2010-2014 to evaluate 148,068 opioid-naive patients aged 18 years and older, this study showed a relationship between inpatient opioid administration, specific patterns of inpatient opioid administration, and postdischarge opioid use. Specifically, inpatient opioid administration was associated with a 3.0% increase (95% CI, 2.8%-3.2%) in opioid use at 90 days post discharge. Additionally, inpatient opioid administration within 12 hours of hospital discharge was associated with a 3.6% increase (95% CI, 3.3%-3.9%) in opioid use at 90 days post discharge.
This observational study is prone to potential unmeasured confounders negating any clear causation. Rather, hospitalists should be aware of the increasing focus on inpatient opioid administration as it relates to outpatient opioid use, especially in the setting of the current opioid crisis.
Bottom line: Inpatient opioid administration and administration patterns are associated with 90-day postdischarge opioid use in opioid-naive patients.
Citation: Donohue JM et al. Patterns of opioid administration among opioid-naive inpatients and associations with postdischarge opioid use. Ann Intern Med. 2019 Jun 18:171(2):81-90.
Dr. Ledford is a hospitalist at Vanderbilt University Medical Center, Nashville, Tenn.
Background: Efforts to reduce and monitor high-risk opioid prescribing have largely focused on outpatient prescribing with less empiric evaluation of inpatient administration. Little is known about the association of inpatient opioid administration and postdischarge opioid use.
Study design: Retrospective cohort.
Setting: 12 community and academic hospitals in Pennsylvania.
Synopsis: With electronic health record data from 2010-2014 to evaluate 148,068 opioid-naive patients aged 18 years and older, this study showed a relationship between inpatient opioid administration, specific patterns of inpatient opioid administration, and postdischarge opioid use. Specifically, inpatient opioid administration was associated with a 3.0% increase (95% CI, 2.8%-3.2%) in opioid use at 90 days post discharge. Additionally, inpatient opioid administration within 12 hours of hospital discharge was associated with a 3.6% increase (95% CI, 3.3%-3.9%) in opioid use at 90 days post discharge.
This observational study is prone to potential unmeasured confounders negating any clear causation. Rather, hospitalists should be aware of the increasing focus on inpatient opioid administration as it relates to outpatient opioid use, especially in the setting of the current opioid crisis.
Bottom line: Inpatient opioid administration and administration patterns are associated with 90-day postdischarge opioid use in opioid-naive patients.
Citation: Donohue JM et al. Patterns of opioid administration among opioid-naive inpatients and associations with postdischarge opioid use. Ann Intern Med. 2019 Jun 18:171(2):81-90.
Dr. Ledford is a hospitalist at Vanderbilt University Medical Center, Nashville, Tenn.