User login
Not your ordinary neuropathy
She has had a diagnosis of type 2 diabetes for the past 4 years. She initially presented with polyuria/polydipsia and a hemoglobin A1c level of 9.5. She has previously not tolerated metformin, and did not want to take any subsequent medications. She was seen 4 months ago and at that time had an A1c level of 12.5. She decided she wanted to really treat her diabetes as well as she could. She started consuming a low carbohydrate diet, restarted metformin and began using a continuous glucose monitor. She also started taking nighttime glargine insulin, and mealtime insulin apart. She reports she lost 20 pounds over the past 4 months, her blood sugars now run between 100-120 fasting, and up to 180 before meals. She has had a severe, sharp pain in both of her feet over the past month that is interfering with sleep and makes walking painful for her. An exam reveals hyperesthesia of both feet, and her A1c level is 7.5. What is the most likely cause of her neuropathic symptoms?
A. Vitamin B12 deficiency
B. Diabetic neuropathy
C. Insulin neuritis
D. Charcot-Marie-Tooth disease
The most likely cause
In this case, certainly considering vitamin B12 deficiency is reasonable. It is highly unlikely though, given the rapidity of onset of symptoms, and that the patient has been on metformin for a very short period of time. Chronic metformin use is associated with low B12 levels, and the American Diabetes Association has advised that regular monitoring of vitamin B12 levels should be done on patients who are on long-term metformin.1
Diabetic neuropathy is also unlikely, given the rapidity of symptoms in this patient. What is most likely in this patient is treatment-induced neuropathy (TIN), first described with the name “insulin neuritis”.
Research on TIN
Gibbons and colleagues evaluated 16 patients with diabetes with recent marked, rapid improvement in glycemic control who developed a sudden, painful neuropathy.2 All developed symptoms within 8 weeks of intensive glucose control, with 69% having autonomic dysfunction as well, and all developing worsening retinopathy.
Gibbons and Freeman did a retrospective study of patients referred to a diabetic neuropathy clinic over a 5-year period to try to understand how prevalent TIN is.3
A total of 954 patients were evaluated for diabetic neuropathy. Treatment induced neuropathy was defined as a painful neuropathy and/or autonomic dysfunction occurring within 8 weeks of intensified treatment and a drop of the A1c level greater than 2 over a 3-month period.
A total of 104 patients (10.9%) met the criteria for treatment induced neuropathy. Patients who had a decrease in A1c had a much greater chance of developing a painful or autonomic neuropathy than patients who had no change in A1c (P < .0001). The same patients had a much higher risk of developing retinopathy (P < .001). The greater the reduction in A1c, the greater the risk. Patients whose A1c decreased by 2%-3% over 3 months had an absolute risk of 20%, whereas those with a A1c decease of greater than 4% had an 80% absolute risk.
Siddique and colleagues reported on three cases with very different clinical presentations of TIN.4 One patient had an acute third nerve palsy, another patient had a lumbosacral radiculoplexus neuropathy, and the third patient presented with a diffuse painful sensory neuropathy and postural hypotension.
Most patients improve over time from their neuropathic symptoms, with better recovery in patients with type 1 diabetes.2
Pearl
Strongly consider treatment induced neuropathy in your patients with diabetes who present with acute painful neuropathy and/or autonomic dysfunction in the setting of rapid improvement of glucose control.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. American Diabetes Association. Diabetes Care. 2019 Jan;42(Suppl 1):S90-102.
2. Gibbons CH and Freeman R. Ann Neurol 2010; 67:534–41.
3. Gibbons CH and Freeman R. Brain. 2015;138:43-52.
4. Siddique N et al. Endocrinol Diabetes Metab Case Rep. 2020 Feb 26;2020:19-0140.
She has had a diagnosis of type 2 diabetes for the past 4 years. She initially presented with polyuria/polydipsia and a hemoglobin A1c level of 9.5. She has previously not tolerated metformin, and did not want to take any subsequent medications. She was seen 4 months ago and at that time had an A1c level of 12.5. She decided she wanted to really treat her diabetes as well as she could. She started consuming a low carbohydrate diet, restarted metformin and began using a continuous glucose monitor. She also started taking nighttime glargine insulin, and mealtime insulin apart. She reports she lost 20 pounds over the past 4 months, her blood sugars now run between 100-120 fasting, and up to 180 before meals. She has had a severe, sharp pain in both of her feet over the past month that is interfering with sleep and makes walking painful for her. An exam reveals hyperesthesia of both feet, and her A1c level is 7.5. What is the most likely cause of her neuropathic symptoms?
A. Vitamin B12 deficiency
B. Diabetic neuropathy
C. Insulin neuritis
D. Charcot-Marie-Tooth disease
The most likely cause
In this case, certainly considering vitamin B12 deficiency is reasonable. It is highly unlikely though, given the rapidity of onset of symptoms, and that the patient has been on metformin for a very short period of time. Chronic metformin use is associated with low B12 levels, and the American Diabetes Association has advised that regular monitoring of vitamin B12 levels should be done on patients who are on long-term metformin.1
Diabetic neuropathy is also unlikely, given the rapidity of symptoms in this patient. What is most likely in this patient is treatment-induced neuropathy (TIN), first described with the name “insulin neuritis”.
Research on TIN
Gibbons and colleagues evaluated 16 patients with diabetes with recent marked, rapid improvement in glycemic control who developed a sudden, painful neuropathy.2 All developed symptoms within 8 weeks of intensive glucose control, with 69% having autonomic dysfunction as well, and all developing worsening retinopathy.
Gibbons and Freeman did a retrospective study of patients referred to a diabetic neuropathy clinic over a 5-year period to try to understand how prevalent TIN is.3
A total of 954 patients were evaluated for diabetic neuropathy. Treatment induced neuropathy was defined as a painful neuropathy and/or autonomic dysfunction occurring within 8 weeks of intensified treatment and a drop of the A1c level greater than 2 over a 3-month period.
A total of 104 patients (10.9%) met the criteria for treatment induced neuropathy. Patients who had a decrease in A1c had a much greater chance of developing a painful or autonomic neuropathy than patients who had no change in A1c (P < .0001). The same patients had a much higher risk of developing retinopathy (P < .001). The greater the reduction in A1c, the greater the risk. Patients whose A1c decreased by 2%-3% over 3 months had an absolute risk of 20%, whereas those with a A1c decease of greater than 4% had an 80% absolute risk.
Siddique and colleagues reported on three cases with very different clinical presentations of TIN.4 One patient had an acute third nerve palsy, another patient had a lumbosacral radiculoplexus neuropathy, and the third patient presented with a diffuse painful sensory neuropathy and postural hypotension.
Most patients improve over time from their neuropathic symptoms, with better recovery in patients with type 1 diabetes.2
Pearl
Strongly consider treatment induced neuropathy in your patients with diabetes who present with acute painful neuropathy and/or autonomic dysfunction in the setting of rapid improvement of glucose control.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. American Diabetes Association. Diabetes Care. 2019 Jan;42(Suppl 1):S90-102.
2. Gibbons CH and Freeman R. Ann Neurol 2010; 67:534–41.
3. Gibbons CH and Freeman R. Brain. 2015;138:43-52.
4. Siddique N et al. Endocrinol Diabetes Metab Case Rep. 2020 Feb 26;2020:19-0140.
She has had a diagnosis of type 2 diabetes for the past 4 years. She initially presented with polyuria/polydipsia and a hemoglobin A1c level of 9.5. She has previously not tolerated metformin, and did not want to take any subsequent medications. She was seen 4 months ago and at that time had an A1c level of 12.5. She decided she wanted to really treat her diabetes as well as she could. She started consuming a low carbohydrate diet, restarted metformin and began using a continuous glucose monitor. She also started taking nighttime glargine insulin, and mealtime insulin apart. She reports she lost 20 pounds over the past 4 months, her blood sugars now run between 100-120 fasting, and up to 180 before meals. She has had a severe, sharp pain in both of her feet over the past month that is interfering with sleep and makes walking painful for her. An exam reveals hyperesthesia of both feet, and her A1c level is 7.5. What is the most likely cause of her neuropathic symptoms?
A. Vitamin B12 deficiency
B. Diabetic neuropathy
C. Insulin neuritis
D. Charcot-Marie-Tooth disease
The most likely cause
In this case, certainly considering vitamin B12 deficiency is reasonable. It is highly unlikely though, given the rapidity of onset of symptoms, and that the patient has been on metformin for a very short period of time. Chronic metformin use is associated with low B12 levels, and the American Diabetes Association has advised that regular monitoring of vitamin B12 levels should be done on patients who are on long-term metformin.1
Diabetic neuropathy is also unlikely, given the rapidity of symptoms in this patient. What is most likely in this patient is treatment-induced neuropathy (TIN), first described with the name “insulin neuritis”.
Research on TIN
Gibbons and colleagues evaluated 16 patients with diabetes with recent marked, rapid improvement in glycemic control who developed a sudden, painful neuropathy.2 All developed symptoms within 8 weeks of intensive glucose control, with 69% having autonomic dysfunction as well, and all developing worsening retinopathy.
Gibbons and Freeman did a retrospective study of patients referred to a diabetic neuropathy clinic over a 5-year period to try to understand how prevalent TIN is.3
A total of 954 patients were evaluated for diabetic neuropathy. Treatment induced neuropathy was defined as a painful neuropathy and/or autonomic dysfunction occurring within 8 weeks of intensified treatment and a drop of the A1c level greater than 2 over a 3-month period.
A total of 104 patients (10.9%) met the criteria for treatment induced neuropathy. Patients who had a decrease in A1c had a much greater chance of developing a painful or autonomic neuropathy than patients who had no change in A1c (P < .0001). The same patients had a much higher risk of developing retinopathy (P < .001). The greater the reduction in A1c, the greater the risk. Patients whose A1c decreased by 2%-3% over 3 months had an absolute risk of 20%, whereas those with a A1c decease of greater than 4% had an 80% absolute risk.
Siddique and colleagues reported on three cases with very different clinical presentations of TIN.4 One patient had an acute third nerve palsy, another patient had a lumbosacral radiculoplexus neuropathy, and the third patient presented with a diffuse painful sensory neuropathy and postural hypotension.
Most patients improve over time from their neuropathic symptoms, with better recovery in patients with type 1 diabetes.2
Pearl
Strongly consider treatment induced neuropathy in your patients with diabetes who present with acute painful neuropathy and/or autonomic dysfunction in the setting of rapid improvement of glucose control.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. American Diabetes Association. Diabetes Care. 2019 Jan;42(Suppl 1):S90-102.
2. Gibbons CH and Freeman R. Ann Neurol 2010; 67:534–41.
3. Gibbons CH and Freeman R. Brain. 2015;138:43-52.
4. Siddique N et al. Endocrinol Diabetes Metab Case Rep. 2020 Feb 26;2020:19-0140.
FDA approves diagnostic device for autism spectrum disorder
The Food and Drug Administration has approved marketing for a device that will help diagnose autism spectrum disorder (ASD) in children between the ages of 18 months and 5 years old who exhibit potential symptoms.
Cognoa ASD Diagnosis Aid is a machine learning–based software program that receives information from parents or caregivers, video analysts, and health care providers to assist physicians in evaluating whether a child is at risk of having autism.
Autism is a developmental disorder that can cause social, communication, and behavioral challenges, according to the Centers for Disease Control and Prevention. The disorder affects about 1 in 54 children. The disorder is difficult to diagnose because there isn’t a medical test to diagnose the it. Instead, physicians have to look at a child’s developmental history and behavior to make a diagnosis.
Many children are not diagnosed with ASD until later in childhood, which in some cases delays treatment and early intervention. ASD may be detected as early as 18 months, but the average age of diagnosis for ASD is 4.3 years, according to the FDA.
“[ASD] can delay a child’s physical, cognitive, and social development, including motor skill development, learning, communication, and interacting with others. The earlier ASD can be diagnosed, the more quickly intervention strategies and appropriate therapies can begin,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement. “Today’s marketing authorization provides a new tool for helping diagnose children with ASD.”
The safety and efficacy of the Cognoa ASD Diagnosis Aid was assessed in a study of 425 patients between the ages of 18 months and 5 years old. For the study, researchers compared the diagnostic assessments made by the device to those made by a panel of clinical experts who used the current standard ASD diagnostic process. The device diagnosed 32% of the children with either a “Positive for ASD” or a “Negative for ASD” result. Researchers found that the device matched the panel’s conclusions for 81% of the patients who received a positive diagnosis. For those who received a negative diagnosis, the device matched the panel’s conclusions for 98% of the patients. In addition, the device made an accurate ASD determination in 98.4% of patients with the condition and in 78.9% of patients without the condition.
Cognoa ASD Diagnosis Aid has three main components. One component includes a mobile app for caregivers to answer questions about the child’s behavioral problems and to upload videos of the child. The next component is a video analysis portal for specialists to view and analyze uploaded videos of patients. Another component is a portal for health care providers that allows them to enter answers to preloaded questions about behavior problems, track the information provided by parents, and review a report of the results.
After the machine learning–based device processes the information provided by parents and health care providers, it reports either a positive or a negative diagnosis. If there is insufficient information to make either a positive or a negative diagnosis, the ASD Diagnostic AID will report that no result can be generated.
Some of the risks associated with this device include misdiagnosis and delayed diagnosis of ASD because of a false-positive or false-negative result, or when no result is generated. Researchers said a false-positive result occurred in 15 out of 303 study subjects without ASD and a false-negative result occurred in 1 out of 122 study subjects with ASD.
The FDA emphasized that the device is indicated to aid physicians in the process of diagnosing ASD in children. This means it shouldn’t be treated as a standalone diagnostic device, but as an adjunct to the diagnostic process.
The Food and Drug Administration has approved marketing for a device that will help diagnose autism spectrum disorder (ASD) in children between the ages of 18 months and 5 years old who exhibit potential symptoms.
Cognoa ASD Diagnosis Aid is a machine learning–based software program that receives information from parents or caregivers, video analysts, and health care providers to assist physicians in evaluating whether a child is at risk of having autism.
Autism is a developmental disorder that can cause social, communication, and behavioral challenges, according to the Centers for Disease Control and Prevention. The disorder affects about 1 in 54 children. The disorder is difficult to diagnose because there isn’t a medical test to diagnose the it. Instead, physicians have to look at a child’s developmental history and behavior to make a diagnosis.
Many children are not diagnosed with ASD until later in childhood, which in some cases delays treatment and early intervention. ASD may be detected as early as 18 months, but the average age of diagnosis for ASD is 4.3 years, according to the FDA.
“[ASD] can delay a child’s physical, cognitive, and social development, including motor skill development, learning, communication, and interacting with others. The earlier ASD can be diagnosed, the more quickly intervention strategies and appropriate therapies can begin,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement. “Today’s marketing authorization provides a new tool for helping diagnose children with ASD.”
The safety and efficacy of the Cognoa ASD Diagnosis Aid was assessed in a study of 425 patients between the ages of 18 months and 5 years old. For the study, researchers compared the diagnostic assessments made by the device to those made by a panel of clinical experts who used the current standard ASD diagnostic process. The device diagnosed 32% of the children with either a “Positive for ASD” or a “Negative for ASD” result. Researchers found that the device matched the panel’s conclusions for 81% of the patients who received a positive diagnosis. For those who received a negative diagnosis, the device matched the panel’s conclusions for 98% of the patients. In addition, the device made an accurate ASD determination in 98.4% of patients with the condition and in 78.9% of patients without the condition.
Cognoa ASD Diagnosis Aid has three main components. One component includes a mobile app for caregivers to answer questions about the child’s behavioral problems and to upload videos of the child. The next component is a video analysis portal for specialists to view and analyze uploaded videos of patients. Another component is a portal for health care providers that allows them to enter answers to preloaded questions about behavior problems, track the information provided by parents, and review a report of the results.
After the machine learning–based device processes the information provided by parents and health care providers, it reports either a positive or a negative diagnosis. If there is insufficient information to make either a positive or a negative diagnosis, the ASD Diagnostic AID will report that no result can be generated.
Some of the risks associated with this device include misdiagnosis and delayed diagnosis of ASD because of a false-positive or false-negative result, or when no result is generated. Researchers said a false-positive result occurred in 15 out of 303 study subjects without ASD and a false-negative result occurred in 1 out of 122 study subjects with ASD.
The FDA emphasized that the device is indicated to aid physicians in the process of diagnosing ASD in children. This means it shouldn’t be treated as a standalone diagnostic device, but as an adjunct to the diagnostic process.
The Food and Drug Administration has approved marketing for a device that will help diagnose autism spectrum disorder (ASD) in children between the ages of 18 months and 5 years old who exhibit potential symptoms.
Cognoa ASD Diagnosis Aid is a machine learning–based software program that receives information from parents or caregivers, video analysts, and health care providers to assist physicians in evaluating whether a child is at risk of having autism.
Autism is a developmental disorder that can cause social, communication, and behavioral challenges, according to the Centers for Disease Control and Prevention. The disorder affects about 1 in 54 children. The disorder is difficult to diagnose because there isn’t a medical test to diagnose the it. Instead, physicians have to look at a child’s developmental history and behavior to make a diagnosis.
Many children are not diagnosed with ASD until later in childhood, which in some cases delays treatment and early intervention. ASD may be detected as early as 18 months, but the average age of diagnosis for ASD is 4.3 years, according to the FDA.
“[ASD] can delay a child’s physical, cognitive, and social development, including motor skill development, learning, communication, and interacting with others. The earlier ASD can be diagnosed, the more quickly intervention strategies and appropriate therapies can begin,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement. “Today’s marketing authorization provides a new tool for helping diagnose children with ASD.”
The safety and efficacy of the Cognoa ASD Diagnosis Aid was assessed in a study of 425 patients between the ages of 18 months and 5 years old. For the study, researchers compared the diagnostic assessments made by the device to those made by a panel of clinical experts who used the current standard ASD diagnostic process. The device diagnosed 32% of the children with either a “Positive for ASD” or a “Negative for ASD” result. Researchers found that the device matched the panel’s conclusions for 81% of the patients who received a positive diagnosis. For those who received a negative diagnosis, the device matched the panel’s conclusions for 98% of the patients. In addition, the device made an accurate ASD determination in 98.4% of patients with the condition and in 78.9% of patients without the condition.
Cognoa ASD Diagnosis Aid has three main components. One component includes a mobile app for caregivers to answer questions about the child’s behavioral problems and to upload videos of the child. The next component is a video analysis portal for specialists to view and analyze uploaded videos of patients. Another component is a portal for health care providers that allows them to enter answers to preloaded questions about behavior problems, track the information provided by parents, and review a report of the results.
After the machine learning–based device processes the information provided by parents and health care providers, it reports either a positive or a negative diagnosis. If there is insufficient information to make either a positive or a negative diagnosis, the ASD Diagnostic AID will report that no result can be generated.
Some of the risks associated with this device include misdiagnosis and delayed diagnosis of ASD because of a false-positive or false-negative result, or when no result is generated. Researchers said a false-positive result occurred in 15 out of 303 study subjects without ASD and a false-negative result occurred in 1 out of 122 study subjects with ASD.
The FDA emphasized that the device is indicated to aid physicians in the process of diagnosing ASD in children. This means it shouldn’t be treated as a standalone diagnostic device, but as an adjunct to the diagnostic process.
Differences in Palliative Care Delivery Among Adults With Cancer and With Terminal Noncancer Illness in Their Last Year of Life
Study Overview
Objective. To examine the patterns in palliative care delivery in the last year of life among adults with cancer compared with adults with a noncancer terminal diagnosis.
Design. Population-based cohort study in Ontario, Canada, using linked administrative and clinical databases. The study included all adults ages 18 and over who died of cancer or noncancer terminal illnesses and received physician-delivered palliative care that was initiated in the last year of life between January 2010 and December 2017. These palliative care services are identified through the use of claims fee codes by physicians that account for delivery of palliative care, such as symptom management and counseling, that are intended to be palliative rather than curative. Exclusion criteria include patients who had 2 or more palliative care service claims the year prior to the last year of life, which may indicate existing palliative care services rather than initiation of new palliative care services in the last year of life. Other patients who were excluded from the study had palliative care services initiated within 7 days of death, as it is less likely that services and support would be arranged prior to death given the short time frame. The types of noncancer illnesses included heart failure, chronic obstructive pulmonary disease, end-stage renal disease, cirrhosis, stroke, and dementia. For the comparison of palliative care services, types of illnesses were divided into cancer, chronic organ failure (heart failure, chronic pulmonary disease, end-stage renal disease, cirrhosis, or stroke), and dementia, as they may represent different trajectories of illnesses and needs.
Setting and participants. The study included 145 709 adults who died during the study period, among 351 941 adults who died from illnesses described above. Another 105 587 were excluded because there were no palliative care services before death, 48 525 were excluded because of existing palliative care services prior to the last year of life, and 44 164 were excluded because palliative care was initiated within 7 days of death. Among the study population included, 21 054 died of chronic organ failure, 14 033 died of dementia, and 110 622 died of cancer. The median age of the study population was 78 years, with an interquartile range of 67 to 86 years, and 50.7% were female. Approximately 12.8% of the study population reside in rural areas; median frailty score (hospital frailty risk score) among those who died of chronic organ failure was 10, and the score among those who died of dementia was 13. The frailty score among those who died of cancer was 3, indicating less frailty. Those who died of organ failure and dementia also had a high mean number of prescription medications (18 and 16, respectively) compared with those with cancer (11).
Main outcome measures. Study outcome measures include the timing of palliative care initiation (primary outcome), categorized into time frames of ≤ 30 days, 31 to 90 days, and > 90 days before death; location of initiation of palliative care services, categorized into clinic, home, hospital, subacute care, and case management; models of care, categorized as generalist, consultative, or specialist palliative care; total number of palliative care visits before death; and location of death. The models of palliative care delivery were categorized based on the proportion of palliative care fee codes claimed by physicians. Physicians whose annual billing included more than 10% of palliative care service codes were considered palliative care specialists. Using this designation, models of palliative care were categorized into those delivered by palliative care specialists, generalists (nonpalliative care specialists), or both.
Main results. The study found that the timing of palliative care initiation was earlier among those who died of cancer compared with those with organ failure or dementia (28.9% vs 15.9% and 15.3%, respectively). After adjustment, those who died of organ failure and those who died of dementia were less likely to have palliative care services initiated > 90 days prior to death (odds ratio [OR] 0.48 and 0.42, respectively) and between 31 to 90 days prior to death (OR 0.77 and 0.60, respectively), when compared with those who died of cancer (who served as the reference group). Regarding location of palliative care initiation, adults who died of cancer were less likely to have palliative care services initiated at home (14.5%) compared with those who died of organ failure (32.8%) or dementia (27.9%). Overall, those who died of cancer received more palliative care visits from initiation to death (median of 11 visits) compared with those who died oforgan failure (median 4 visits) and dementia (median 4 visits). Regarding models of palliative care delivery, a higher proportion of palliative care was delivered by palliative care specialists rather than generalists among cancer patients (72.9%) compared with those with organ failure (43.3%) or dementia (40.1%). The proportion of patients with cancer who died at home was 62.6%, which was higher than those with organ failure (53.3%) but lower than those with dementia (75%).
Conclusion. There are differences in the delivery of palliative care among patients with cancer and other noncancer terminal illnesses, including timing of initiation of palliative care services, location of services, number of visits, and delivery by types of practitioners of palliative care. Understanding these disparities and targeting them are potentially important steps to ensuring appropriate access to palliative care across settings and disease types.
Commentary
Palliative care improves the quality of life of patients with serious illnesses and reduces symptom burden, and results in better satisfaction and less burdensome care.1 Although palliative care approaches have been championed for cancer management, there is increasing evidence that palliative care also improves outcomes for patients with noncancer illnesses such as heart failure.2 This study highlights the differences in palliative care delivery for patients who have cancer and noncancer diagnoses, demonstrating that timing, location, and care delivery models differ among patients with different diagnoses. The finding that noncancer terminal illness often has later palliative care initiation is a significant one, as early palliative care has been associated with improved patient outcomes3; thus, efforts to initiate palliative care earlier in the course of illness may benefit these patients.
A particular challenge in determining when to initiate palliative care lies in predicting outcomes,4 particularly for different types of illnesses, which may have different trajectories of advancing disease and functional change. Recent research has tested novel prognostic approaches, such as using machine learning to generate mortality estimates and integrating them into clinical decision support.5 These approaches may have the potential to enhance palliative care delivery and may be adapted to be used in managing patients with noncancer illnesses as well. The study also found that patients with cancer were more likely to receive palliative care from specialists rather than generalists, although this could be due to how palliative care is integrated in hospitals, clinics, and systems of care that serve patients with cancer. Identifying approaches that yield better palliative care models and delivery may help to further enhance care for patients with noncancer illnesses.
Applications for Clinical Practice
Identifying differences in patterns of palliative care delivery among those with cancer and other diagnoses may be an important step towards identifying gaps and avenues to improve palliative care delivery. The underlying reasons for these differences could be targeted so that patients across settings and diagnoses may have equal access to palliative care to improve their symptoms and quality of life. Policy makers and health system leaders may consider learning from how palliative care has been integrated into oncology care, to help transform care delivery for other noncancer terminal illnesses. It may also involve broadening education to providers in different specialties, so that the value and importance of palliative care may be recognized beyond oncological care.
1. Kavalieratos D, Corbelli J, Zhang D, et al. Association Between Palliative Care and Patient and Caregiver Outcomes: A Systematic Review and Meta-analysis. JAMA. 2016;316(20):2104-2114.
2. Quinn KL, Stukel T, Stall NM, et al. Association between palliative care and healthcare outcomes among adults with terminal non-cancer illness: population based matched cohort study. BMJ. 2020;370:m2257.
3. Temel JS, Greer JA, Muzikansky A, et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2010;363:733-742.
4. White N, Reid F, Harris A, et al. A Systematic Review of Predictions of Survival in Palliative Care: How Accurate Are Clinicians and Who Are the Experts? PLoS One. 2016;11(8):e0161407.
5. Manz CR, Parikh RB, Small DS, et al. Effect of Integrating Machine Learning Mortality Estimates With Behavioral Nudges to Clinicians on Serious Illness Conversations Among Patients With Cancer: A Stepped-Wedge Cluster Randomized Clinical Trial. JAMA Oncol. 2020;6(12):e204759.
Study Overview
Objective. To examine the patterns in palliative care delivery in the last year of life among adults with cancer compared with adults with a noncancer terminal diagnosis.
Design. Population-based cohort study in Ontario, Canada, using linked administrative and clinical databases. The study included all adults ages 18 and over who died of cancer or noncancer terminal illnesses and received physician-delivered palliative care that was initiated in the last year of life between January 2010 and December 2017. These palliative care services are identified through the use of claims fee codes by physicians that account for delivery of palliative care, such as symptom management and counseling, that are intended to be palliative rather than curative. Exclusion criteria include patients who had 2 or more palliative care service claims the year prior to the last year of life, which may indicate existing palliative care services rather than initiation of new palliative care services in the last year of life. Other patients who were excluded from the study had palliative care services initiated within 7 days of death, as it is less likely that services and support would be arranged prior to death given the short time frame. The types of noncancer illnesses included heart failure, chronic obstructive pulmonary disease, end-stage renal disease, cirrhosis, stroke, and dementia. For the comparison of palliative care services, types of illnesses were divided into cancer, chronic organ failure (heart failure, chronic pulmonary disease, end-stage renal disease, cirrhosis, or stroke), and dementia, as they may represent different trajectories of illnesses and needs.
Setting and participants. The study included 145 709 adults who died during the study period, among 351 941 adults who died from illnesses described above. Another 105 587 were excluded because there were no palliative care services before death, 48 525 were excluded because of existing palliative care services prior to the last year of life, and 44 164 were excluded because palliative care was initiated within 7 days of death. Among the study population included, 21 054 died of chronic organ failure, 14 033 died of dementia, and 110 622 died of cancer. The median age of the study population was 78 years, with an interquartile range of 67 to 86 years, and 50.7% were female. Approximately 12.8% of the study population reside in rural areas; median frailty score (hospital frailty risk score) among those who died of chronic organ failure was 10, and the score among those who died of dementia was 13. The frailty score among those who died of cancer was 3, indicating less frailty. Those who died of organ failure and dementia also had a high mean number of prescription medications (18 and 16, respectively) compared with those with cancer (11).
Main outcome measures. Study outcome measures include the timing of palliative care initiation (primary outcome), categorized into time frames of ≤ 30 days, 31 to 90 days, and > 90 days before death; location of initiation of palliative care services, categorized into clinic, home, hospital, subacute care, and case management; models of care, categorized as generalist, consultative, or specialist palliative care; total number of palliative care visits before death; and location of death. The models of palliative care delivery were categorized based on the proportion of palliative care fee codes claimed by physicians. Physicians whose annual billing included more than 10% of palliative care service codes were considered palliative care specialists. Using this designation, models of palliative care were categorized into those delivered by palliative care specialists, generalists (nonpalliative care specialists), or both.
Main results. The study found that the timing of palliative care initiation was earlier among those who died of cancer compared with those with organ failure or dementia (28.9% vs 15.9% and 15.3%, respectively). After adjustment, those who died of organ failure and those who died of dementia were less likely to have palliative care services initiated > 90 days prior to death (odds ratio [OR] 0.48 and 0.42, respectively) and between 31 to 90 days prior to death (OR 0.77 and 0.60, respectively), when compared with those who died of cancer (who served as the reference group). Regarding location of palliative care initiation, adults who died of cancer were less likely to have palliative care services initiated at home (14.5%) compared with those who died of organ failure (32.8%) or dementia (27.9%). Overall, those who died of cancer received more palliative care visits from initiation to death (median of 11 visits) compared with those who died oforgan failure (median 4 visits) and dementia (median 4 visits). Regarding models of palliative care delivery, a higher proportion of palliative care was delivered by palliative care specialists rather than generalists among cancer patients (72.9%) compared with those with organ failure (43.3%) or dementia (40.1%). The proportion of patients with cancer who died at home was 62.6%, which was higher than those with organ failure (53.3%) but lower than those with dementia (75%).
Conclusion. There are differences in the delivery of palliative care among patients with cancer and other noncancer terminal illnesses, including timing of initiation of palliative care services, location of services, number of visits, and delivery by types of practitioners of palliative care. Understanding these disparities and targeting them are potentially important steps to ensuring appropriate access to palliative care across settings and disease types.
Commentary
Palliative care improves the quality of life of patients with serious illnesses and reduces symptom burden, and results in better satisfaction and less burdensome care.1 Although palliative care approaches have been championed for cancer management, there is increasing evidence that palliative care also improves outcomes for patients with noncancer illnesses such as heart failure.2 This study highlights the differences in palliative care delivery for patients who have cancer and noncancer diagnoses, demonstrating that timing, location, and care delivery models differ among patients with different diagnoses. The finding that noncancer terminal illness often has later palliative care initiation is a significant one, as early palliative care has been associated with improved patient outcomes3; thus, efforts to initiate palliative care earlier in the course of illness may benefit these patients.
A particular challenge in determining when to initiate palliative care lies in predicting outcomes,4 particularly for different types of illnesses, which may have different trajectories of advancing disease and functional change. Recent research has tested novel prognostic approaches, such as using machine learning to generate mortality estimates and integrating them into clinical decision support.5 These approaches may have the potential to enhance palliative care delivery and may be adapted to be used in managing patients with noncancer illnesses as well. The study also found that patients with cancer were more likely to receive palliative care from specialists rather than generalists, although this could be due to how palliative care is integrated in hospitals, clinics, and systems of care that serve patients with cancer. Identifying approaches that yield better palliative care models and delivery may help to further enhance care for patients with noncancer illnesses.
Applications for Clinical Practice
Identifying differences in patterns of palliative care delivery among those with cancer and other diagnoses may be an important step towards identifying gaps and avenues to improve palliative care delivery. The underlying reasons for these differences could be targeted so that patients across settings and diagnoses may have equal access to palliative care to improve their symptoms and quality of life. Policy makers and health system leaders may consider learning from how palliative care has been integrated into oncology care, to help transform care delivery for other noncancer terminal illnesses. It may also involve broadening education to providers in different specialties, so that the value and importance of palliative care may be recognized beyond oncological care.
Study Overview
Objective. To examine the patterns in palliative care delivery in the last year of life among adults with cancer compared with adults with a noncancer terminal diagnosis.
Design. Population-based cohort study in Ontario, Canada, using linked administrative and clinical databases. The study included all adults ages 18 and over who died of cancer or noncancer terminal illnesses and received physician-delivered palliative care that was initiated in the last year of life between January 2010 and December 2017. These palliative care services are identified through the use of claims fee codes by physicians that account for delivery of palliative care, such as symptom management and counseling, that are intended to be palliative rather than curative. Exclusion criteria include patients who had 2 or more palliative care service claims the year prior to the last year of life, which may indicate existing palliative care services rather than initiation of new palliative care services in the last year of life. Other patients who were excluded from the study had palliative care services initiated within 7 days of death, as it is less likely that services and support would be arranged prior to death given the short time frame. The types of noncancer illnesses included heart failure, chronic obstructive pulmonary disease, end-stage renal disease, cirrhosis, stroke, and dementia. For the comparison of palliative care services, types of illnesses were divided into cancer, chronic organ failure (heart failure, chronic pulmonary disease, end-stage renal disease, cirrhosis, or stroke), and dementia, as they may represent different trajectories of illnesses and needs.
Setting and participants. The study included 145 709 adults who died during the study period, among 351 941 adults who died from illnesses described above. Another 105 587 were excluded because there were no palliative care services before death, 48 525 were excluded because of existing palliative care services prior to the last year of life, and 44 164 were excluded because palliative care was initiated within 7 days of death. Among the study population included, 21 054 died of chronic organ failure, 14 033 died of dementia, and 110 622 died of cancer. The median age of the study population was 78 years, with an interquartile range of 67 to 86 years, and 50.7% were female. Approximately 12.8% of the study population reside in rural areas; median frailty score (hospital frailty risk score) among those who died of chronic organ failure was 10, and the score among those who died of dementia was 13. The frailty score among those who died of cancer was 3, indicating less frailty. Those who died of organ failure and dementia also had a high mean number of prescription medications (18 and 16, respectively) compared with those with cancer (11).
Main outcome measures. Study outcome measures include the timing of palliative care initiation (primary outcome), categorized into time frames of ≤ 30 days, 31 to 90 days, and > 90 days before death; location of initiation of palliative care services, categorized into clinic, home, hospital, subacute care, and case management; models of care, categorized as generalist, consultative, or specialist palliative care; total number of palliative care visits before death; and location of death. The models of palliative care delivery were categorized based on the proportion of palliative care fee codes claimed by physicians. Physicians whose annual billing included more than 10% of palliative care service codes were considered palliative care specialists. Using this designation, models of palliative care were categorized into those delivered by palliative care specialists, generalists (nonpalliative care specialists), or both.
Main results. The study found that the timing of palliative care initiation was earlier among those who died of cancer compared with those with organ failure or dementia (28.9% vs 15.9% and 15.3%, respectively). After adjustment, those who died of organ failure and those who died of dementia were less likely to have palliative care services initiated > 90 days prior to death (odds ratio [OR] 0.48 and 0.42, respectively) and between 31 to 90 days prior to death (OR 0.77 and 0.60, respectively), when compared with those who died of cancer (who served as the reference group). Regarding location of palliative care initiation, adults who died of cancer were less likely to have palliative care services initiated at home (14.5%) compared with those who died of organ failure (32.8%) or dementia (27.9%). Overall, those who died of cancer received more palliative care visits from initiation to death (median of 11 visits) compared with those who died oforgan failure (median 4 visits) and dementia (median 4 visits). Regarding models of palliative care delivery, a higher proportion of palliative care was delivered by palliative care specialists rather than generalists among cancer patients (72.9%) compared with those with organ failure (43.3%) or dementia (40.1%). The proportion of patients with cancer who died at home was 62.6%, which was higher than those with organ failure (53.3%) but lower than those with dementia (75%).
Conclusion. There are differences in the delivery of palliative care among patients with cancer and other noncancer terminal illnesses, including timing of initiation of palliative care services, location of services, number of visits, and delivery by types of practitioners of palliative care. Understanding these disparities and targeting them are potentially important steps to ensuring appropriate access to palliative care across settings and disease types.
Commentary
Palliative care improves the quality of life of patients with serious illnesses and reduces symptom burden, and results in better satisfaction and less burdensome care.1 Although palliative care approaches have been championed for cancer management, there is increasing evidence that palliative care also improves outcomes for patients with noncancer illnesses such as heart failure.2 This study highlights the differences in palliative care delivery for patients who have cancer and noncancer diagnoses, demonstrating that timing, location, and care delivery models differ among patients with different diagnoses. The finding that noncancer terminal illness often has later palliative care initiation is a significant one, as early palliative care has been associated with improved patient outcomes3; thus, efforts to initiate palliative care earlier in the course of illness may benefit these patients.
A particular challenge in determining when to initiate palliative care lies in predicting outcomes,4 particularly for different types of illnesses, which may have different trajectories of advancing disease and functional change. Recent research has tested novel prognostic approaches, such as using machine learning to generate mortality estimates and integrating them into clinical decision support.5 These approaches may have the potential to enhance palliative care delivery and may be adapted to be used in managing patients with noncancer illnesses as well. The study also found that patients with cancer were more likely to receive palliative care from specialists rather than generalists, although this could be due to how palliative care is integrated in hospitals, clinics, and systems of care that serve patients with cancer. Identifying approaches that yield better palliative care models and delivery may help to further enhance care for patients with noncancer illnesses.
Applications for Clinical Practice
Identifying differences in patterns of palliative care delivery among those with cancer and other diagnoses may be an important step towards identifying gaps and avenues to improve palliative care delivery. The underlying reasons for these differences could be targeted so that patients across settings and diagnoses may have equal access to palliative care to improve their symptoms and quality of life. Policy makers and health system leaders may consider learning from how palliative care has been integrated into oncology care, to help transform care delivery for other noncancer terminal illnesses. It may also involve broadening education to providers in different specialties, so that the value and importance of palliative care may be recognized beyond oncological care.
1. Kavalieratos D, Corbelli J, Zhang D, et al. Association Between Palliative Care and Patient and Caregiver Outcomes: A Systematic Review and Meta-analysis. JAMA. 2016;316(20):2104-2114.
2. Quinn KL, Stukel T, Stall NM, et al. Association between palliative care and healthcare outcomes among adults with terminal non-cancer illness: population based matched cohort study. BMJ. 2020;370:m2257.
3. Temel JS, Greer JA, Muzikansky A, et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2010;363:733-742.
4. White N, Reid F, Harris A, et al. A Systematic Review of Predictions of Survival in Palliative Care: How Accurate Are Clinicians and Who Are the Experts? PLoS One. 2016;11(8):e0161407.
5. Manz CR, Parikh RB, Small DS, et al. Effect of Integrating Machine Learning Mortality Estimates With Behavioral Nudges to Clinicians on Serious Illness Conversations Among Patients With Cancer: A Stepped-Wedge Cluster Randomized Clinical Trial. JAMA Oncol. 2020;6(12):e204759.
1. Kavalieratos D, Corbelli J, Zhang D, et al. Association Between Palliative Care and Patient and Caregiver Outcomes: A Systematic Review and Meta-analysis. JAMA. 2016;316(20):2104-2114.
2. Quinn KL, Stukel T, Stall NM, et al. Association between palliative care and healthcare outcomes among adults with terminal non-cancer illness: population based matched cohort study. BMJ. 2020;370:m2257.
3. Temel JS, Greer JA, Muzikansky A, et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2010;363:733-742.
4. White N, Reid F, Harris A, et al. A Systematic Review of Predictions of Survival in Palliative Care: How Accurate Are Clinicians and Who Are the Experts? PLoS One. 2016;11(8):e0161407.
5. Manz CR, Parikh RB, Small DS, et al. Effect of Integrating Machine Learning Mortality Estimates With Behavioral Nudges to Clinicians on Serious Illness Conversations Among Patients With Cancer: A Stepped-Wedge Cluster Randomized Clinical Trial. JAMA Oncol. 2020;6(12):e204759.
A Service Evaluation of Acute Neurological Patients Managed on Clinically Inappropriate Wards
From Western Sussex Hospitals NHS Foundation Trust, Physiotherapy Department, Chichester, UK (Richard J. Holmes), and Western Sussex Hospitals NHS Foundation Trust, Department of Occupational Therapy, Chichester, UK (Sophie Stratford).
Objective: Despite the benefits of early and frequent input from a neurologist, there is wide variation in the availability of this service, especially in district general hospitals, with many patients managed on clinically inappropriate wards. The purpose of this service evaluation was to explore the impact this had on patient care.
Methods: A retrospective service evaluation was undertaken at a National Health Service hospital by reviewing patient records over a 6-month period. Data related to demographics, processes within the patient’s care, and secondary complications were recorded. Findings were compared with those of stroke patients managed on a specialist stroke ward.
Results: A total of 63 patients were identified, with a mean age of 72 years. The mean length of stay was 25.9 days, with a readmission rate of 16.7%. Only 15.9% of patients were reviewed by a neurologist. There was a high rate of secondary complications, with a number of patients experiencing falls (11.1%), pressure ulcers (14.3%), and health care–acquired infections (33.3%) during their admission.
Conclusions: The lack of specialist input from a neurologist and the management of patients on clinically inappropriate wards may have negatively impacted length of stay, readmission rates, and the frequency of secondary complications.
Keywords: evaluation; clinical safety; neurology; patient-centered care; clinical outcomes; length of stay.
It is estimated that 10% of acute admissions to district general hospitals (DGHs) of the National Health Service (NHS) in the United Kingdom are due to a neurological problem other than stroke.1 In 2011, a joint report from the Royal College of Physicians and the Association of British Neurologists (ABN) recommended that all of these patients should be admitted under the care of a neurologist and be regularly reviewed by a neurologist during their admission.2 The rationale for this recommendation is clear. The involvement of a neurologist has been shown to improve accuracy of the diagnosis3 and significantly reduce length of stay.4,5 Studies have also shown that the involvement of a neurologist has led to a change in the management plan in as high as 79%6 to 89%3 of cases, suggesting that a high proportion of neurological patients not seen by a neurologist are being managed suboptimally.
Despite this, a recent ABN survey of acute neurology services found ongoing wide variations in the availability of this specialist care, with a large proportion of DGHs having limited or no access to a neurologist and very few having dedicated neurology beds.7 While it is recognized that services have been structured in response to the reduced numbers of neurologists within the United Kingdom,8 it is prudent to assess the impact that such services have on patient care.
With this in mind, we planned to evaluate the current provision of care provided to neurological patients in a real-world setting. This was conducted in the context of a neurology liaison service at a DGH with no dedicated neurology beds.
Methods
A retrospective service evaluation was undertaken at a DGH in the southeast of England. The NHS hospital has neurologists on site who provide diagnostic and therapeutic consultations on the wards, but there are no dedicated beds for patients with neurological conditions. Patients requiring neurosurgical input are referred to a tertiary neurosciences center.
Patients were selected from the neurotherapy database if they were referred into the service between August 1, 2019, and January 31, 2020. The neurotherapy database was used as this was the only source that held thorough data on this patient group and allowed for the identification of patients who were not referred into the neurologist’s service. Patients were included if they had a new neurological condition as their primary diagnosis or if they had an exacerbation of an already established neurological condition. If a patient was admitted with more than 1 neurological diagnosis then the primary diagnosis for the admission was to be used in the analysis, though this did not occur during this evaluation. Patients with a primary diagnosis of a stroke were included if they were not managed on the acute stroke ward. Those managed on the stroke ward were excluded so that an analysis of patients managed on wards that were deemed clinically inappropriate could be undertaken. Patients were not included if they had a pre-existing neurological condition (ie, dementia, multiple sclerosis) but were admitted due to a non-neurological cause such as a fall or infection. All patients who met the criteria were included.
A team member independently reviewed each set of patient notes. Demographic data extracted from the medical notes included the patient’s age (on admission), gender, and diagnosis. Medical, nursing, and therapy notes were reviewed to identify secondary complications that arose during the patient’s admission. The secondary complications reviewed were falls (defined as the patient unexpectedly coming to the ground or other lower level), health care–acquired infections (HAIs) (defined as any infection acquired during the hospital admission), and pressure ulcers (defined as injuries to the skin or underlying tissue during the hospital admission). Other details, obtained from the patient administration system, included the length of stay (days), the number of ward moves the patient experienced, the speciality of the consultant responsible for the patient’s care, the discharge destination, and whether the patient was readmitted for any cause within 30 days. All data collected were stored on a password-protected computer and no patient-identifiable data were included.
The results were collated using descriptive statistics. The χ2 test was used to compare categorical data between those patients who were and were not reviewed by a neurologist, and the Mann-Whitney U test was used to compare differences in the length of stay between these 2 groups.
No national data relating to this specific patient group were available within the literature. Therefore, to provide a comparator of neurological patients within the same hospital, data were collected on stroke patients managed on the stroke ward. This group was deemed most appropriate for comparison as they present with similar neurological symptoms but are cared for on a specialist ward. During the evaluation period, 284 stroke patients were admitted to the stroke ward. A sample of 75 patients was randomly selected using a random number generator, and the procedure for data collection was repeated. It was not appropriate to make direct comparative analysis on these 2 groups due to the inherent differences, but it was felt important to provide context with regards to what usual care was like on a specialist ward within the same hospital.
Ethical approval was not required as this was a service evaluation of routinely collected data within a single hospital site.
Results
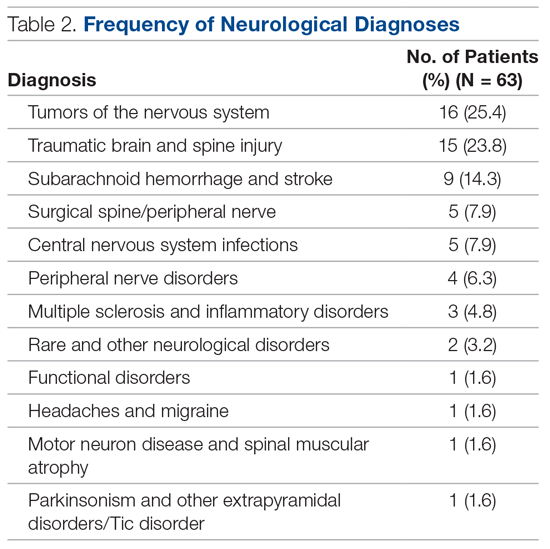
In total, 63 patients were identified: 26 females and 37 males. The median age of patients was 74 years (range, 39-92 years). These demographic details and comparisons to stroke patients managed on a specialist ward can be seen in Table 1. To quantify the range of diagnoses, the condition groups defined by GIRFT Neurology Methodology9 were used. The most common diagnoses were tumors of the nervous system (25.4%) and traumatic brain and spine injury (23.8%). The other conditions included in the analysis can be seen in Table 2.
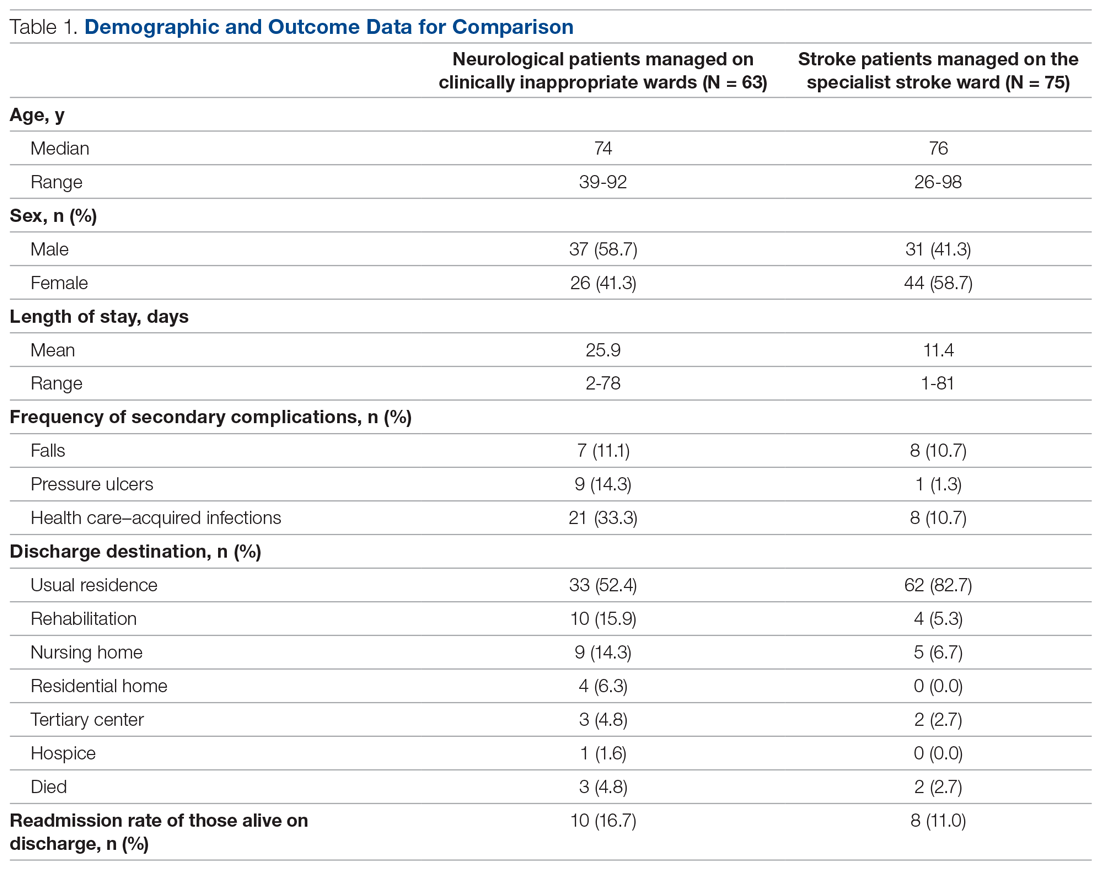
Despite having a neurological condition as their primary diagnosis, only 15.9% of patients were reviewed by a neurologist during their hospital admission. Patients were most commonly under the care of a geriatrician (60.3%), but they were also managed by orthopedics (12.6%), acute medicine (7.9%), respiratory (6.3%), cardiology (4.8%), gastroenterology (3.2%), and surgery (3.2%). One patient (1.6%) was managed by intensivists.
The average length of stay was 25.9 days (range, 2-78 days). This was more than double the average length of stay on the stroke ward (11.4 days) (Table 1) and the national average for patients with neurological conditions (9.78 days).10 During their stay, 33% had 2 or more ward moves, with 1 patient moving wards a total of 6 times. Just over half (52.4%) of the patients returned to their usual residence on discharge. The remainder were discharged to rehabilitation units (15.9%), nursing homes (14.3%), residential homes (6.3%), tertiary centers (4.8%), and hospice (1.6%). Unfortunately, 3 patients (4.8%) passed away. Of those still alive (n = 60), 16.7% were readmitted to the hospital within 30 days, compared to a readmission rate of 11% on the stroke ward. None of the patients who were readmitted were seen by a neurologist during their initial admission.
The frequency of secondary complications was reviewed as a measure of the multidisciplinary management of this patient group. It was noted that 11.1% had a fall on the ward, which was similar to a rate of 10.7% on the stroke ward. More striking was the fact that 14.3% of patients developed a pressure ulcer and 33.3% developed an HAI during their admission, compared with rates of 1.3% and 10.7%, respectively, on the stroke ward (Table 1).
There were no significant differences found in length of stay between those who were and were not reviewed by a neurologist (P = .73). This was also true for categorical data, whereby readmission rate (P = .13), frequency of falls (P = .22), frequency of pressure ulcers (P = .67), and HAIs (P = .81) all failed to show a significant difference between groups.
Discussion
The findings of this service evaluation show markedly poorer outcomes for neurological patients compared to stroke patients managed on a specialist stroke ward. It is suggested that these results are in part due to the lack of specialist input from a neurologist in the majority of cases and the fact that all were managed on clinically inappropriate wards. Only 15.9% of neurological patients were seen by a neurologist. This is a slight improvement compared to previous studies in DGHs that showed rates of 10%1 and 11%,11 but it is still a far cry from the goal of 100% set out in recommendations.2 In addition, the increased readmission rate may be suggestive of suboptimal management, especially given that none of those readmitted had been reviewed by a neurologist. There are undoubtedly other factors that may influence readmissions, such as comorbidities, the severity/complexity of the condition, and the strength of community services. However, the impact of a lack of input from a specialist should not be underestimated, and further evaluation of this factor (with confounding factors controlled) would be beneficial.
The result of an extended length of stay was also a predictable outcome based on previous evidence.4,5 With the potential for suboptimal management plans and inaccurate diagnoses, it is inevitable that the patient’s movement through the hospital system will be impeded. In our example, it is possible that the extended length of stay was influenced by the fact that patients included in the evaluation were managed on nonspecialist wards and a large proportion had multiple ward changes.
Given that the evidence clearly shows that stroke patients are most effectively managed by a multidisciplinary team (MDT) with specialist skills,12 it is likely that other neurological patients, who have similar multifactorial needs, would also benefit. The patients in our evaluation were cared for by nursing staff who lacked specific skills and experience in neurology. The allied health professionals involved were specialists in neurotherapy but were not based on the ward and not directly linked to the ward MDT. A review by Epstein found that the benefits of having a MDT, in any speciality, working together on a ward included improved communication, reduced adverse events, and a reduced length of stay.13 This lack of an effective MDT approach may provide some explanation as to why the average length of stay and the rates of some secondary complications were at such elevated levels.
A systematic review exploring the impact of patients admitted to clinically inappropriate wards in a range of specialities found that these patients were associated with worse outcomes.14 This is supported by our findings, in which a higher rate of pressure ulcers and HAIs were observed when compared to rates in the specialist stroke ward. Again, a potential explanation for this is the impact of patients being managed by clinicians who lack the specialist knowledge of the patient group and the risks they face. Another explanation could be due to the high number of ward moves the patients experienced. Blay et al found that ward moves increased length of stay and carried an associated clinical risk, with the odds of falls and HAIs increasing with each move.15 A case example of this is apparent within our analysis in that the patient who experienced 6 ward moves not only had the longest length of stay (78 days), but also developed a pressure ulcer and 2 HAIs during their admission.
This service evaluation had a number of limitations that should be considered when interpreting the results. First, despite including all patients who met the criteria within the stipulated time frame, the sample size was relatively small, making it difficult to identify consistent patterns of behavior within the data.
Furthermore, caution should be applied when interpreting the comparators used, as the patient groups are not equivalent. The use of comparison against a standard is not a prerequisite in a service evaluation of this nature, but comparators were included to help frame the context for the reader. As such, they should only be used in this way rather than to make any firm conclusions.
Finally, as the evaluation was limited to the use of routinely collected data, there are several variables, other than those reported, which may have influenced the results. For example, it was not possible to ascertain certain demographic details, such as body mass index and socioeconomic factors, nor lifestyle factors such as smoking status, alcohol consumption, and exercise levels, all of which could impact negatively on the outcomes of interest. Furthermore, data were not collected on follow-up services after discharge to evaluate whether these had any impact on readmission rates.
Conclusion
This service evaluation highlights the potential impact of managing neurological patients on clinically inappropriate wards with limited input from a neurologist. There is the potential to ameliorate these impacts by cohorting these patients in neurologist-led beds with a specialist MDT. While there are limitations in the design of our study, including the lack of a controlled comparison, the small sample size, and the fact that this is an evaluation of a single service, the negative impacts to patients are concerning and warrant further investigation.
Corresponding author: Richard J. Holmes, MSc, Physiotherapy Department, St. Richard’s Hospital, Chichester, West Sussex, PO19 6SE; richard.holmes8@nhs.net.
Financial disclosures: None.
1. Kanagaratnam M, Boodhoo A, MacDonald BK, Nitkunan A. Prevalence of acute neurology: a 2-week snapshot in a district general hospital. Clin Med (Lond). 2020;20(2):169-173.
2. Royal College of Physicians. Local adult neurology services for the next decade. Report of a working party. June 2011. Accessed October 29, 2020. https://www.mstrust.org.uk/sites/default/files/files/Local%20adult%20neurology%20services%20for%20the%20next%20decade.pdf
3. McColgan P, Carr AS, McCarron MO. The value of a liaison neurology service in a district general hospital. Postgrad Med J. 2011;87(1025):166-169.
4. Forbes R, Craig J, Callender M, Patterson V. Liaison neurology for acute medical admissions. Clin Med (Lond). 2004;4(3):290.
5. Craig J, Chua R, Russell C, et al. A cohort study of early neurological consultation by telemedicine on the care of neurological inpatients. J Neurol Neurosurg Psychiatry. 2004;75(7):1031-1035.
6. Ali E, Chaila E, Hutchinson M, Tubridy N. The ‘hidden work’ of a hospital neurologist: 1000 consults later. Eur J Neurol. 2010;17(4):e28-e32.
7. Association of British Neurologists. Acute Neurology services survey 2017. Accessed October 29, 2020. https://cdn.ymaws.com/www.theabn.org/resource/collection/219B4A48-4D25-4726-97AA-0EB6090769BE/ABN_2017_Acute_Neurology_Survey.pdf
8. Nitkunan A, Lawrence J, Reilly MM. Neurology Workforce Survey. January 28, 2020. Accessed October 28, 2020. https://cdn.ymaws.com/www.theabn.org/resource/collection/219B4A48-4D25-4726-97AA-0EB6090769BE/2020_ABN_Neurology_Workforce_Survey_2018-19_28_Jan_2020.pdf
9. Fuller G, Connolly M, Mummery C, Williams A. GIRT Neurology Methodology and Initial Summary of Regional Data. September 2019. Accessed October 26, 2020. https://gettingitrightfirsttime.co.uk/wp-content/uploads/2017/07/GIRFT-neurology-methodology-090919-FINAL.pdf
10. The Neurological Alliance. Neuro Numbers 2019. Accessed October 28, 2020. https://www.neural.org.uk/wp-content/uploads/2019/07/neuro-numbers-2019.pdf
11. Cai A, Brex P. A survey of acute neurology at a general hospital in the UK. Clin Med (Lond). 2010;10(6):642-643.
12. Langhorne P, Ramachandra S; Stroke Unit Trialists’ Collaboration. Organised inpatient (stroke unit) care for stroke: network meta-analysis. Cochrane Database Syst Rev. 2020;4(4):CD000197.
13. Epstein NE. Multidisciplinary in-hospital teams improve patient outcomes: A review. Surg Neurol Int. 2014;5(Suppl 7):S295-S303.
14. La Regina M, Guarneri F, Romano E, et al. What Quality and Safety of Care for Patients Admitted to Clinically Inappropriate Wards: a Systematic Review. J Gen Intern Med. 2019;34(7):1314-1321.
15. Blay N, Roche M, Duffield C, Xu X. Intrahospital transfers and adverse patient outcomes: An analysis of administrative health data. J Clin Nurs. 2017;26(23-24):4927-4935.
From Western Sussex Hospitals NHS Foundation Trust, Physiotherapy Department, Chichester, UK (Richard J. Holmes), and Western Sussex Hospitals NHS Foundation Trust, Department of Occupational Therapy, Chichester, UK (Sophie Stratford).
Objective: Despite the benefits of early and frequent input from a neurologist, there is wide variation in the availability of this service, especially in district general hospitals, with many patients managed on clinically inappropriate wards. The purpose of this service evaluation was to explore the impact this had on patient care.
Methods: A retrospective service evaluation was undertaken at a National Health Service hospital by reviewing patient records over a 6-month period. Data related to demographics, processes within the patient’s care, and secondary complications were recorded. Findings were compared with those of stroke patients managed on a specialist stroke ward.
Results: A total of 63 patients were identified, with a mean age of 72 years. The mean length of stay was 25.9 days, with a readmission rate of 16.7%. Only 15.9% of patients were reviewed by a neurologist. There was a high rate of secondary complications, with a number of patients experiencing falls (11.1%), pressure ulcers (14.3%), and health care–acquired infections (33.3%) during their admission.
Conclusions: The lack of specialist input from a neurologist and the management of patients on clinically inappropriate wards may have negatively impacted length of stay, readmission rates, and the frequency of secondary complications.
Keywords: evaluation; clinical safety; neurology; patient-centered care; clinical outcomes; length of stay.
It is estimated that 10% of acute admissions to district general hospitals (DGHs) of the National Health Service (NHS) in the United Kingdom are due to a neurological problem other than stroke.1 In 2011, a joint report from the Royal College of Physicians and the Association of British Neurologists (ABN) recommended that all of these patients should be admitted under the care of a neurologist and be regularly reviewed by a neurologist during their admission.2 The rationale for this recommendation is clear. The involvement of a neurologist has been shown to improve accuracy of the diagnosis3 and significantly reduce length of stay.4,5 Studies have also shown that the involvement of a neurologist has led to a change in the management plan in as high as 79%6 to 89%3 of cases, suggesting that a high proportion of neurological patients not seen by a neurologist are being managed suboptimally.
Despite this, a recent ABN survey of acute neurology services found ongoing wide variations in the availability of this specialist care, with a large proportion of DGHs having limited or no access to a neurologist and very few having dedicated neurology beds.7 While it is recognized that services have been structured in response to the reduced numbers of neurologists within the United Kingdom,8 it is prudent to assess the impact that such services have on patient care.
With this in mind, we planned to evaluate the current provision of care provided to neurological patients in a real-world setting. This was conducted in the context of a neurology liaison service at a DGH with no dedicated neurology beds.
Methods
A retrospective service evaluation was undertaken at a DGH in the southeast of England. The NHS hospital has neurologists on site who provide diagnostic and therapeutic consultations on the wards, but there are no dedicated beds for patients with neurological conditions. Patients requiring neurosurgical input are referred to a tertiary neurosciences center.
Patients were selected from the neurotherapy database if they were referred into the service between August 1, 2019, and January 31, 2020. The neurotherapy database was used as this was the only source that held thorough data on this patient group and allowed for the identification of patients who were not referred into the neurologist’s service. Patients were included if they had a new neurological condition as their primary diagnosis or if they had an exacerbation of an already established neurological condition. If a patient was admitted with more than 1 neurological diagnosis then the primary diagnosis for the admission was to be used in the analysis, though this did not occur during this evaluation. Patients with a primary diagnosis of a stroke were included if they were not managed on the acute stroke ward. Those managed on the stroke ward were excluded so that an analysis of patients managed on wards that were deemed clinically inappropriate could be undertaken. Patients were not included if they had a pre-existing neurological condition (ie, dementia, multiple sclerosis) but were admitted due to a non-neurological cause such as a fall or infection. All patients who met the criteria were included.
A team member independently reviewed each set of patient notes. Demographic data extracted from the medical notes included the patient’s age (on admission), gender, and diagnosis. Medical, nursing, and therapy notes were reviewed to identify secondary complications that arose during the patient’s admission. The secondary complications reviewed were falls (defined as the patient unexpectedly coming to the ground or other lower level), health care–acquired infections (HAIs) (defined as any infection acquired during the hospital admission), and pressure ulcers (defined as injuries to the skin or underlying tissue during the hospital admission). Other details, obtained from the patient administration system, included the length of stay (days), the number of ward moves the patient experienced, the speciality of the consultant responsible for the patient’s care, the discharge destination, and whether the patient was readmitted for any cause within 30 days. All data collected were stored on a password-protected computer and no patient-identifiable data were included.
The results were collated using descriptive statistics. The χ2 test was used to compare categorical data between those patients who were and were not reviewed by a neurologist, and the Mann-Whitney U test was used to compare differences in the length of stay between these 2 groups.
No national data relating to this specific patient group were available within the literature. Therefore, to provide a comparator of neurological patients within the same hospital, data were collected on stroke patients managed on the stroke ward. This group was deemed most appropriate for comparison as they present with similar neurological symptoms but are cared for on a specialist ward. During the evaluation period, 284 stroke patients were admitted to the stroke ward. A sample of 75 patients was randomly selected using a random number generator, and the procedure for data collection was repeated. It was not appropriate to make direct comparative analysis on these 2 groups due to the inherent differences, but it was felt important to provide context with regards to what usual care was like on a specialist ward within the same hospital.
Ethical approval was not required as this was a service evaluation of routinely collected data within a single hospital site.
Results
In total, 63 patients were identified: 26 females and 37 males. The median age of patients was 74 years (range, 39-92 years). These demographic details and comparisons to stroke patients managed on a specialist ward can be seen in Table 1. To quantify the range of diagnoses, the condition groups defined by GIRFT Neurology Methodology9 were used. The most common diagnoses were tumors of the nervous system (25.4%) and traumatic brain and spine injury (23.8%). The other conditions included in the analysis can be seen in Table 2.

Despite having a neurological condition as their primary diagnosis, only 15.9% of patients were reviewed by a neurologist during their hospital admission. Patients were most commonly under the care of a geriatrician (60.3%), but they were also managed by orthopedics (12.6%), acute medicine (7.9%), respiratory (6.3%), cardiology (4.8%), gastroenterology (3.2%), and surgery (3.2%). One patient (1.6%) was managed by intensivists.

The average length of stay was 25.9 days (range, 2-78 days). This was more than double the average length of stay on the stroke ward (11.4 days) (Table 1) and the national average for patients with neurological conditions (9.78 days).10 During their stay, 33% had 2 or more ward moves, with 1 patient moving wards a total of 6 times. Just over half (52.4%) of the patients returned to their usual residence on discharge. The remainder were discharged to rehabilitation units (15.9%), nursing homes (14.3%), residential homes (6.3%), tertiary centers (4.8%), and hospice (1.6%). Unfortunately, 3 patients (4.8%) passed away. Of those still alive (n = 60), 16.7% were readmitted to the hospital within 30 days, compared to a readmission rate of 11% on the stroke ward. None of the patients who were readmitted were seen by a neurologist during their initial admission.
The frequency of secondary complications was reviewed as a measure of the multidisciplinary management of this patient group. It was noted that 11.1% had a fall on the ward, which was similar to a rate of 10.7% on the stroke ward. More striking was the fact that 14.3% of patients developed a pressure ulcer and 33.3% developed an HAI during their admission, compared with rates of 1.3% and 10.7%, respectively, on the stroke ward (Table 1).
There were no significant differences found in length of stay between those who were and were not reviewed by a neurologist (P = .73). This was also true for categorical data, whereby readmission rate (P = .13), frequency of falls (P = .22), frequency of pressure ulcers (P = .67), and HAIs (P = .81) all failed to show a significant difference between groups.
Discussion
The findings of this service evaluation show markedly poorer outcomes for neurological patients compared to stroke patients managed on a specialist stroke ward. It is suggested that these results are in part due to the lack of specialist input from a neurologist in the majority of cases and the fact that all were managed on clinically inappropriate wards. Only 15.9% of neurological patients were seen by a neurologist. This is a slight improvement compared to previous studies in DGHs that showed rates of 10%1 and 11%,11 but it is still a far cry from the goal of 100% set out in recommendations.2 In addition, the increased readmission rate may be suggestive of suboptimal management, especially given that none of those readmitted had been reviewed by a neurologist. There are undoubtedly other factors that may influence readmissions, such as comorbidities, the severity/complexity of the condition, and the strength of community services. However, the impact of a lack of input from a specialist should not be underestimated, and further evaluation of this factor (with confounding factors controlled) would be beneficial.
The result of an extended length of stay was also a predictable outcome based on previous evidence.4,5 With the potential for suboptimal management plans and inaccurate diagnoses, it is inevitable that the patient’s movement through the hospital system will be impeded. In our example, it is possible that the extended length of stay was influenced by the fact that patients included in the evaluation were managed on nonspecialist wards and a large proportion had multiple ward changes.
Given that the evidence clearly shows that stroke patients are most effectively managed by a multidisciplinary team (MDT) with specialist skills,12 it is likely that other neurological patients, who have similar multifactorial needs, would also benefit. The patients in our evaluation were cared for by nursing staff who lacked specific skills and experience in neurology. The allied health professionals involved were specialists in neurotherapy but were not based on the ward and not directly linked to the ward MDT. A review by Epstein found that the benefits of having a MDT, in any speciality, working together on a ward included improved communication, reduced adverse events, and a reduced length of stay.13 This lack of an effective MDT approach may provide some explanation as to why the average length of stay and the rates of some secondary complications were at such elevated levels.
A systematic review exploring the impact of patients admitted to clinically inappropriate wards in a range of specialities found that these patients were associated with worse outcomes.14 This is supported by our findings, in which a higher rate of pressure ulcers and HAIs were observed when compared to rates in the specialist stroke ward. Again, a potential explanation for this is the impact of patients being managed by clinicians who lack the specialist knowledge of the patient group and the risks they face. Another explanation could be due to the high number of ward moves the patients experienced. Blay et al found that ward moves increased length of stay and carried an associated clinical risk, with the odds of falls and HAIs increasing with each move.15 A case example of this is apparent within our analysis in that the patient who experienced 6 ward moves not only had the longest length of stay (78 days), but also developed a pressure ulcer and 2 HAIs during their admission.
This service evaluation had a number of limitations that should be considered when interpreting the results. First, despite including all patients who met the criteria within the stipulated time frame, the sample size was relatively small, making it difficult to identify consistent patterns of behavior within the data.
Furthermore, caution should be applied when interpreting the comparators used, as the patient groups are not equivalent. The use of comparison against a standard is not a prerequisite in a service evaluation of this nature, but comparators were included to help frame the context for the reader. As such, they should only be used in this way rather than to make any firm conclusions.
Finally, as the evaluation was limited to the use of routinely collected data, there are several variables, other than those reported, which may have influenced the results. For example, it was not possible to ascertain certain demographic details, such as body mass index and socioeconomic factors, nor lifestyle factors such as smoking status, alcohol consumption, and exercise levels, all of which could impact negatively on the outcomes of interest. Furthermore, data were not collected on follow-up services after discharge to evaluate whether these had any impact on readmission rates.
Conclusion
This service evaluation highlights the potential impact of managing neurological patients on clinically inappropriate wards with limited input from a neurologist. There is the potential to ameliorate these impacts by cohorting these patients in neurologist-led beds with a specialist MDT. While there are limitations in the design of our study, including the lack of a controlled comparison, the small sample size, and the fact that this is an evaluation of a single service, the negative impacts to patients are concerning and warrant further investigation.
Corresponding author: Richard J. Holmes, MSc, Physiotherapy Department, St. Richard’s Hospital, Chichester, West Sussex, PO19 6SE; richard.holmes8@nhs.net.
Financial disclosures: None.
From Western Sussex Hospitals NHS Foundation Trust, Physiotherapy Department, Chichester, UK (Richard J. Holmes), and Western Sussex Hospitals NHS Foundation Trust, Department of Occupational Therapy, Chichester, UK (Sophie Stratford).
Objective: Despite the benefits of early and frequent input from a neurologist, there is wide variation in the availability of this service, especially in district general hospitals, with many patients managed on clinically inappropriate wards. The purpose of this service evaluation was to explore the impact this had on patient care.
Methods: A retrospective service evaluation was undertaken at a National Health Service hospital by reviewing patient records over a 6-month period. Data related to demographics, processes within the patient’s care, and secondary complications were recorded. Findings were compared with those of stroke patients managed on a specialist stroke ward.
Results: A total of 63 patients were identified, with a mean age of 72 years. The mean length of stay was 25.9 days, with a readmission rate of 16.7%. Only 15.9% of patients were reviewed by a neurologist. There was a high rate of secondary complications, with a number of patients experiencing falls (11.1%), pressure ulcers (14.3%), and health care–acquired infections (33.3%) during their admission.
Conclusions: The lack of specialist input from a neurologist and the management of patients on clinically inappropriate wards may have negatively impacted length of stay, readmission rates, and the frequency of secondary complications.
Keywords: evaluation; clinical safety; neurology; patient-centered care; clinical outcomes; length of stay.
It is estimated that 10% of acute admissions to district general hospitals (DGHs) of the National Health Service (NHS) in the United Kingdom are due to a neurological problem other than stroke.1 In 2011, a joint report from the Royal College of Physicians and the Association of British Neurologists (ABN) recommended that all of these patients should be admitted under the care of a neurologist and be regularly reviewed by a neurologist during their admission.2 The rationale for this recommendation is clear. The involvement of a neurologist has been shown to improve accuracy of the diagnosis3 and significantly reduce length of stay.4,5 Studies have also shown that the involvement of a neurologist has led to a change in the management plan in as high as 79%6 to 89%3 of cases, suggesting that a high proportion of neurological patients not seen by a neurologist are being managed suboptimally.
Despite this, a recent ABN survey of acute neurology services found ongoing wide variations in the availability of this specialist care, with a large proportion of DGHs having limited or no access to a neurologist and very few having dedicated neurology beds.7 While it is recognized that services have been structured in response to the reduced numbers of neurologists within the United Kingdom,8 it is prudent to assess the impact that such services have on patient care.
With this in mind, we planned to evaluate the current provision of care provided to neurological patients in a real-world setting. This was conducted in the context of a neurology liaison service at a DGH with no dedicated neurology beds.
Methods
A retrospective service evaluation was undertaken at a DGH in the southeast of England. The NHS hospital has neurologists on site who provide diagnostic and therapeutic consultations on the wards, but there are no dedicated beds for patients with neurological conditions. Patients requiring neurosurgical input are referred to a tertiary neurosciences center.
Patients were selected from the neurotherapy database if they were referred into the service between August 1, 2019, and January 31, 2020. The neurotherapy database was used as this was the only source that held thorough data on this patient group and allowed for the identification of patients who were not referred into the neurologist’s service. Patients were included if they had a new neurological condition as their primary diagnosis or if they had an exacerbation of an already established neurological condition. If a patient was admitted with more than 1 neurological diagnosis then the primary diagnosis for the admission was to be used in the analysis, though this did not occur during this evaluation. Patients with a primary diagnosis of a stroke were included if they were not managed on the acute stroke ward. Those managed on the stroke ward were excluded so that an analysis of patients managed on wards that were deemed clinically inappropriate could be undertaken. Patients were not included if they had a pre-existing neurological condition (ie, dementia, multiple sclerosis) but were admitted due to a non-neurological cause such as a fall or infection. All patients who met the criteria were included.
A team member independently reviewed each set of patient notes. Demographic data extracted from the medical notes included the patient’s age (on admission), gender, and diagnosis. Medical, nursing, and therapy notes were reviewed to identify secondary complications that arose during the patient’s admission. The secondary complications reviewed were falls (defined as the patient unexpectedly coming to the ground or other lower level), health care–acquired infections (HAIs) (defined as any infection acquired during the hospital admission), and pressure ulcers (defined as injuries to the skin or underlying tissue during the hospital admission). Other details, obtained from the patient administration system, included the length of stay (days), the number of ward moves the patient experienced, the speciality of the consultant responsible for the patient’s care, the discharge destination, and whether the patient was readmitted for any cause within 30 days. All data collected were stored on a password-protected computer and no patient-identifiable data were included.
The results were collated using descriptive statistics. The χ2 test was used to compare categorical data between those patients who were and were not reviewed by a neurologist, and the Mann-Whitney U test was used to compare differences in the length of stay between these 2 groups.
No national data relating to this specific patient group were available within the literature. Therefore, to provide a comparator of neurological patients within the same hospital, data were collected on stroke patients managed on the stroke ward. This group was deemed most appropriate for comparison as they present with similar neurological symptoms but are cared for on a specialist ward. During the evaluation period, 284 stroke patients were admitted to the stroke ward. A sample of 75 patients was randomly selected using a random number generator, and the procedure for data collection was repeated. It was not appropriate to make direct comparative analysis on these 2 groups due to the inherent differences, but it was felt important to provide context with regards to what usual care was like on a specialist ward within the same hospital.
Ethical approval was not required as this was a service evaluation of routinely collected data within a single hospital site.
Results
In total, 63 patients were identified: 26 females and 37 males. The median age of patients was 74 years (range, 39-92 years). These demographic details and comparisons to stroke patients managed on a specialist ward can be seen in Table 1. To quantify the range of diagnoses, the condition groups defined by GIRFT Neurology Methodology9 were used. The most common diagnoses were tumors of the nervous system (25.4%) and traumatic brain and spine injury (23.8%). The other conditions included in the analysis can be seen in Table 2.

Despite having a neurological condition as their primary diagnosis, only 15.9% of patients were reviewed by a neurologist during their hospital admission. Patients were most commonly under the care of a geriatrician (60.3%), but they were also managed by orthopedics (12.6%), acute medicine (7.9%), respiratory (6.3%), cardiology (4.8%), gastroenterology (3.2%), and surgery (3.2%). One patient (1.6%) was managed by intensivists.

The average length of stay was 25.9 days (range, 2-78 days). This was more than double the average length of stay on the stroke ward (11.4 days) (Table 1) and the national average for patients with neurological conditions (9.78 days).10 During their stay, 33% had 2 or more ward moves, with 1 patient moving wards a total of 6 times. Just over half (52.4%) of the patients returned to their usual residence on discharge. The remainder were discharged to rehabilitation units (15.9%), nursing homes (14.3%), residential homes (6.3%), tertiary centers (4.8%), and hospice (1.6%). Unfortunately, 3 patients (4.8%) passed away. Of those still alive (n = 60), 16.7% were readmitted to the hospital within 30 days, compared to a readmission rate of 11% on the stroke ward. None of the patients who were readmitted were seen by a neurologist during their initial admission.
The frequency of secondary complications was reviewed as a measure of the multidisciplinary management of this patient group. It was noted that 11.1% had a fall on the ward, which was similar to a rate of 10.7% on the stroke ward. More striking was the fact that 14.3% of patients developed a pressure ulcer and 33.3% developed an HAI during their admission, compared with rates of 1.3% and 10.7%, respectively, on the stroke ward (Table 1).
There were no significant differences found in length of stay between those who were and were not reviewed by a neurologist (P = .73). This was also true for categorical data, whereby readmission rate (P = .13), frequency of falls (P = .22), frequency of pressure ulcers (P = .67), and HAIs (P = .81) all failed to show a significant difference between groups.
Discussion
The findings of this service evaluation show markedly poorer outcomes for neurological patients compared to stroke patients managed on a specialist stroke ward. It is suggested that these results are in part due to the lack of specialist input from a neurologist in the majority of cases and the fact that all were managed on clinically inappropriate wards. Only 15.9% of neurological patients were seen by a neurologist. This is a slight improvement compared to previous studies in DGHs that showed rates of 10%1 and 11%,11 but it is still a far cry from the goal of 100% set out in recommendations.2 In addition, the increased readmission rate may be suggestive of suboptimal management, especially given that none of those readmitted had been reviewed by a neurologist. There are undoubtedly other factors that may influence readmissions, such as comorbidities, the severity/complexity of the condition, and the strength of community services. However, the impact of a lack of input from a specialist should not be underestimated, and further evaluation of this factor (with confounding factors controlled) would be beneficial.
The result of an extended length of stay was also a predictable outcome based on previous evidence.4,5 With the potential for suboptimal management plans and inaccurate diagnoses, it is inevitable that the patient’s movement through the hospital system will be impeded. In our example, it is possible that the extended length of stay was influenced by the fact that patients included in the evaluation were managed on nonspecialist wards and a large proportion had multiple ward changes.
Given that the evidence clearly shows that stroke patients are most effectively managed by a multidisciplinary team (MDT) with specialist skills,12 it is likely that other neurological patients, who have similar multifactorial needs, would also benefit. The patients in our evaluation were cared for by nursing staff who lacked specific skills and experience in neurology. The allied health professionals involved were specialists in neurotherapy but were not based on the ward and not directly linked to the ward MDT. A review by Epstein found that the benefits of having a MDT, in any speciality, working together on a ward included improved communication, reduced adverse events, and a reduced length of stay.13 This lack of an effective MDT approach may provide some explanation as to why the average length of stay and the rates of some secondary complications were at such elevated levels.
A systematic review exploring the impact of patients admitted to clinically inappropriate wards in a range of specialities found that these patients were associated with worse outcomes.14 This is supported by our findings, in which a higher rate of pressure ulcers and HAIs were observed when compared to rates in the specialist stroke ward. Again, a potential explanation for this is the impact of patients being managed by clinicians who lack the specialist knowledge of the patient group and the risks they face. Another explanation could be due to the high number of ward moves the patients experienced. Blay et al found that ward moves increased length of stay and carried an associated clinical risk, with the odds of falls and HAIs increasing with each move.15 A case example of this is apparent within our analysis in that the patient who experienced 6 ward moves not only had the longest length of stay (78 days), but also developed a pressure ulcer and 2 HAIs during their admission.
This service evaluation had a number of limitations that should be considered when interpreting the results. First, despite including all patients who met the criteria within the stipulated time frame, the sample size was relatively small, making it difficult to identify consistent patterns of behavior within the data.
Furthermore, caution should be applied when interpreting the comparators used, as the patient groups are not equivalent. The use of comparison against a standard is not a prerequisite in a service evaluation of this nature, but comparators were included to help frame the context for the reader. As such, they should only be used in this way rather than to make any firm conclusions.
Finally, as the evaluation was limited to the use of routinely collected data, there are several variables, other than those reported, which may have influenced the results. For example, it was not possible to ascertain certain demographic details, such as body mass index and socioeconomic factors, nor lifestyle factors such as smoking status, alcohol consumption, and exercise levels, all of which could impact negatively on the outcomes of interest. Furthermore, data were not collected on follow-up services after discharge to evaluate whether these had any impact on readmission rates.
Conclusion
This service evaluation highlights the potential impact of managing neurological patients on clinically inappropriate wards with limited input from a neurologist. There is the potential to ameliorate these impacts by cohorting these patients in neurologist-led beds with a specialist MDT. While there are limitations in the design of our study, including the lack of a controlled comparison, the small sample size, and the fact that this is an evaluation of a single service, the negative impacts to patients are concerning and warrant further investigation.
Corresponding author: Richard J. Holmes, MSc, Physiotherapy Department, St. Richard’s Hospital, Chichester, West Sussex, PO19 6SE; richard.holmes8@nhs.net.
Financial disclosures: None.
1. Kanagaratnam M, Boodhoo A, MacDonald BK, Nitkunan A. Prevalence of acute neurology: a 2-week snapshot in a district general hospital. Clin Med (Lond). 2020;20(2):169-173.
2. Royal College of Physicians. Local adult neurology services for the next decade. Report of a working party. June 2011. Accessed October 29, 2020. https://www.mstrust.org.uk/sites/default/files/files/Local%20adult%20neurology%20services%20for%20the%20next%20decade.pdf
3. McColgan P, Carr AS, McCarron MO. The value of a liaison neurology service in a district general hospital. Postgrad Med J. 2011;87(1025):166-169.
4. Forbes R, Craig J, Callender M, Patterson V. Liaison neurology for acute medical admissions. Clin Med (Lond). 2004;4(3):290.
5. Craig J, Chua R, Russell C, et al. A cohort study of early neurological consultation by telemedicine on the care of neurological inpatients. J Neurol Neurosurg Psychiatry. 2004;75(7):1031-1035.
6. Ali E, Chaila E, Hutchinson M, Tubridy N. The ‘hidden work’ of a hospital neurologist: 1000 consults later. Eur J Neurol. 2010;17(4):e28-e32.
7. Association of British Neurologists. Acute Neurology services survey 2017. Accessed October 29, 2020. https://cdn.ymaws.com/www.theabn.org/resource/collection/219B4A48-4D25-4726-97AA-0EB6090769BE/ABN_2017_Acute_Neurology_Survey.pdf
8. Nitkunan A, Lawrence J, Reilly MM. Neurology Workforce Survey. January 28, 2020. Accessed October 28, 2020. https://cdn.ymaws.com/www.theabn.org/resource/collection/219B4A48-4D25-4726-97AA-0EB6090769BE/2020_ABN_Neurology_Workforce_Survey_2018-19_28_Jan_2020.pdf
9. Fuller G, Connolly M, Mummery C, Williams A. GIRT Neurology Methodology and Initial Summary of Regional Data. September 2019. Accessed October 26, 2020. https://gettingitrightfirsttime.co.uk/wp-content/uploads/2017/07/GIRFT-neurology-methodology-090919-FINAL.pdf
10. The Neurological Alliance. Neuro Numbers 2019. Accessed October 28, 2020. https://www.neural.org.uk/wp-content/uploads/2019/07/neuro-numbers-2019.pdf
11. Cai A, Brex P. A survey of acute neurology at a general hospital in the UK. Clin Med (Lond). 2010;10(6):642-643.
12. Langhorne P, Ramachandra S; Stroke Unit Trialists’ Collaboration. Organised inpatient (stroke unit) care for stroke: network meta-analysis. Cochrane Database Syst Rev. 2020;4(4):CD000197.
13. Epstein NE. Multidisciplinary in-hospital teams improve patient outcomes: A review. Surg Neurol Int. 2014;5(Suppl 7):S295-S303.
14. La Regina M, Guarneri F, Romano E, et al. What Quality and Safety of Care for Patients Admitted to Clinically Inappropriate Wards: a Systematic Review. J Gen Intern Med. 2019;34(7):1314-1321.
15. Blay N, Roche M, Duffield C, Xu X. Intrahospital transfers and adverse patient outcomes: An analysis of administrative health data. J Clin Nurs. 2017;26(23-24):4927-4935.
1. Kanagaratnam M, Boodhoo A, MacDonald BK, Nitkunan A. Prevalence of acute neurology: a 2-week snapshot in a district general hospital. Clin Med (Lond). 2020;20(2):169-173.
2. Royal College of Physicians. Local adult neurology services for the next decade. Report of a working party. June 2011. Accessed October 29, 2020. https://www.mstrust.org.uk/sites/default/files/files/Local%20adult%20neurology%20services%20for%20the%20next%20decade.pdf
3. McColgan P, Carr AS, McCarron MO. The value of a liaison neurology service in a district general hospital. Postgrad Med J. 2011;87(1025):166-169.
4. Forbes R, Craig J, Callender M, Patterson V. Liaison neurology for acute medical admissions. Clin Med (Lond). 2004;4(3):290.
5. Craig J, Chua R, Russell C, et al. A cohort study of early neurological consultation by telemedicine on the care of neurological inpatients. J Neurol Neurosurg Psychiatry. 2004;75(7):1031-1035.
6. Ali E, Chaila E, Hutchinson M, Tubridy N. The ‘hidden work’ of a hospital neurologist: 1000 consults later. Eur J Neurol. 2010;17(4):e28-e32.
7. Association of British Neurologists. Acute Neurology services survey 2017. Accessed October 29, 2020. https://cdn.ymaws.com/www.theabn.org/resource/collection/219B4A48-4D25-4726-97AA-0EB6090769BE/ABN_2017_Acute_Neurology_Survey.pdf
8. Nitkunan A, Lawrence J, Reilly MM. Neurology Workforce Survey. January 28, 2020. Accessed October 28, 2020. https://cdn.ymaws.com/www.theabn.org/resource/collection/219B4A48-4D25-4726-97AA-0EB6090769BE/2020_ABN_Neurology_Workforce_Survey_2018-19_28_Jan_2020.pdf
9. Fuller G, Connolly M, Mummery C, Williams A. GIRT Neurology Methodology and Initial Summary of Regional Data. September 2019. Accessed October 26, 2020. https://gettingitrightfirsttime.co.uk/wp-content/uploads/2017/07/GIRFT-neurology-methodology-090919-FINAL.pdf
10. The Neurological Alliance. Neuro Numbers 2019. Accessed October 28, 2020. https://www.neural.org.uk/wp-content/uploads/2019/07/neuro-numbers-2019.pdf
11. Cai A, Brex P. A survey of acute neurology at a general hospital in the UK. Clin Med (Lond). 2010;10(6):642-643.
12. Langhorne P, Ramachandra S; Stroke Unit Trialists’ Collaboration. Organised inpatient (stroke unit) care for stroke: network meta-analysis. Cochrane Database Syst Rev. 2020;4(4):CD000197.
13. Epstein NE. Multidisciplinary in-hospital teams improve patient outcomes: A review. Surg Neurol Int. 2014;5(Suppl 7):S295-S303.
14. La Regina M, Guarneri F, Romano E, et al. What Quality and Safety of Care for Patients Admitted to Clinically Inappropriate Wards: a Systematic Review. J Gen Intern Med. 2019;34(7):1314-1321.
15. Blay N, Roche M, Duffield C, Xu X. Intrahospital transfers and adverse patient outcomes: An analysis of administrative health data. J Clin Nurs. 2017;26(23-24):4927-4935.
New AHA/ASA guideline on secondary stroke prevention
When possible, diagnostic tests to determine the cause of a first stroke or transient ischemic attack (TIA) should be completed within 48 hours after symptom onset, the American Heart Association/American Stroke Association said in an updated clinical practice guideline.
“It is critically important to understand the best ways to prevent another stroke once someone has had a stroke or a TIA,” Dawn O. Kleindorfer, MD, chair of the guideline writing group, said in a news release.
“If we can pinpoint the cause of the first stroke or TIA, we can tailor strategies to prevent a second stroke,” said Dr. Kleindorfer, professor and chair, department of neurology, University of Michigan, Ann Arbor.
The updated guideline was published online May 24, 2021, in Stroke.
“The secondary prevention of stroke guideline is one of the ASA’s ‘flagship’ guidelines, last updated in 2014,” Dr. Kleindorfer said.
The update includes “a number of changes to the writing and formatting of this guideline to make it easier for professionals to understand and locate information more quickly, ultimately greatly improving patient care and preventing more strokes in our patients,” she noted.
Let pathogenic subtype guide prevention
For patients who have survived a stroke or TIA, management of vascular risk factors, particularly hypertension, diabetes, cholesterol/triglyceride levels, and smoking cessation, are key secondary prevention tactics, the guideline said.
Limiting salt intake and/or following a heart-healthy Mediterranean diet is also advised, as is engaging in at least moderate-intensity aerobic activity for at least 10 minutes four times a week or vigorous-intensity aerobic activity for at least 20 minutes twice a week.
“Approximately 80% of strokes can be prevented by controlling blood pressure, eating a healthy diet, engaging in regular physical activity, not smoking and maintaining a healthy weight,” Amytis Towfighi, MD, vice chair of the guideline writing group and director of neurologic services, Los Angeles County Department of Health Services, noted in the release.
For health care professionals, the guideline said specific recommendations for secondary prevention often depend on the ischemic stroke/TIA subtype. “Therefore, new in this guideline is a section describing recommendations for the diagnostic workup after ischemic stroke, to define ischemic stroke pathogenesis (when possible), and to identify targets for treatment to reduce the risk of recurrent ischemic stroke. Recommendations are now segregated by pathogenetic subtype,” the guideline stated.
Among the recommendations:
- Use multidisciplinary care teams to personalize care for patients and employ shared decision-making with the patient to develop care plans that incorporate a patient’s wishes, goals, and concerns.
- Screen for and initiate anticoagulant drug therapy to reduce recurrent events.
- Prescribe antithrombotic therapy, including antiplatelets or anticoagulants, in the absence of contraindications. The guideline noted that the combination of antiplatelets and anticoagulation is typically not recommended for preventing second strokes and that dual antiplatelet therapy (DAPT) – taking along with a second medication to prevent blood clotting – is recommended in the short term and only for specific patients: those with early arriving minor stroke and high-risk TIA or severe symptomatic stenosis.
- Consider or carotid artery stenting for select patients with narrowing of carotid arteries.
- Aggressive medical management of risk factors and short-term DAPT are preferred for patients with severe intracranial stenosis thought to be the cause of first stroke or TIA.
- In some patients, it’s reasonable to consider percutaneous closure of .
The guideline is accompanied by a systematic review and meta-analysis regarding the benefits and risks of dual antiplatelet versus single antiplatelet therapy for secondary stroke prevention. The authors conclude that DAPT may be appropriate for select patients.
“Additional research is needed to determine: the optimal timing of starting treatment relative to the clinical event; the optimal duration of DAPT to maximize the risk-benefit ratio; whether additional populations excluded from POINT and CHANCE [two of the trials examined], such as those with major stroke, may also benefit from early DAPT; and whether certain genetic profiles eliminate the benefit of early DAPT,” concluded the reviewers, led by Devin Brown, MD, University of Michigan.
The guideline was prepared on behalf of and approved by the AHA Stroke Council’s Scientific Statements Oversight Committee on Clinical Practice Guidelines. The writing group included representatives from the AHA/ASA and the American Academy of Neurology. The guideline has been endorsed by the American Association of Neurological Surgeons/Congress of Neurological Surgeons and the Society of Vascular and Interventional Neurology. It has also been affirmed by the AAN as an educational tool for neurologists.
The research had no commercial funding.
A version of this article first appeared on Medscape.com.
When possible, diagnostic tests to determine the cause of a first stroke or transient ischemic attack (TIA) should be completed within 48 hours after symptom onset, the American Heart Association/American Stroke Association said in an updated clinical practice guideline.
“It is critically important to understand the best ways to prevent another stroke once someone has had a stroke or a TIA,” Dawn O. Kleindorfer, MD, chair of the guideline writing group, said in a news release.
“If we can pinpoint the cause of the first stroke or TIA, we can tailor strategies to prevent a second stroke,” said Dr. Kleindorfer, professor and chair, department of neurology, University of Michigan, Ann Arbor.
The updated guideline was published online May 24, 2021, in Stroke.
“The secondary prevention of stroke guideline is one of the ASA’s ‘flagship’ guidelines, last updated in 2014,” Dr. Kleindorfer said.
The update includes “a number of changes to the writing and formatting of this guideline to make it easier for professionals to understand and locate information more quickly, ultimately greatly improving patient care and preventing more strokes in our patients,” she noted.
Let pathogenic subtype guide prevention
For patients who have survived a stroke or TIA, management of vascular risk factors, particularly hypertension, diabetes, cholesterol/triglyceride levels, and smoking cessation, are key secondary prevention tactics, the guideline said.
Limiting salt intake and/or following a heart-healthy Mediterranean diet is also advised, as is engaging in at least moderate-intensity aerobic activity for at least 10 minutes four times a week or vigorous-intensity aerobic activity for at least 20 minutes twice a week.
“Approximately 80% of strokes can be prevented by controlling blood pressure, eating a healthy diet, engaging in regular physical activity, not smoking and maintaining a healthy weight,” Amytis Towfighi, MD, vice chair of the guideline writing group and director of neurologic services, Los Angeles County Department of Health Services, noted in the release.
For health care professionals, the guideline said specific recommendations for secondary prevention often depend on the ischemic stroke/TIA subtype. “Therefore, new in this guideline is a section describing recommendations for the diagnostic workup after ischemic stroke, to define ischemic stroke pathogenesis (when possible), and to identify targets for treatment to reduce the risk of recurrent ischemic stroke. Recommendations are now segregated by pathogenetic subtype,” the guideline stated.
Among the recommendations:
- Use multidisciplinary care teams to personalize care for patients and employ shared decision-making with the patient to develop care plans that incorporate a patient’s wishes, goals, and concerns.
- Screen for and initiate anticoagulant drug therapy to reduce recurrent events.
- Prescribe antithrombotic therapy, including antiplatelets or anticoagulants, in the absence of contraindications. The guideline noted that the combination of antiplatelets and anticoagulation is typically not recommended for preventing second strokes and that dual antiplatelet therapy (DAPT) – taking along with a second medication to prevent blood clotting – is recommended in the short term and only for specific patients: those with early arriving minor stroke and high-risk TIA or severe symptomatic stenosis.
- Consider or carotid artery stenting for select patients with narrowing of carotid arteries.
- Aggressive medical management of risk factors and short-term DAPT are preferred for patients with severe intracranial stenosis thought to be the cause of first stroke or TIA.
- In some patients, it’s reasonable to consider percutaneous closure of .
The guideline is accompanied by a systematic review and meta-analysis regarding the benefits and risks of dual antiplatelet versus single antiplatelet therapy for secondary stroke prevention. The authors conclude that DAPT may be appropriate for select patients.
“Additional research is needed to determine: the optimal timing of starting treatment relative to the clinical event; the optimal duration of DAPT to maximize the risk-benefit ratio; whether additional populations excluded from POINT and CHANCE [two of the trials examined], such as those with major stroke, may also benefit from early DAPT; and whether certain genetic profiles eliminate the benefit of early DAPT,” concluded the reviewers, led by Devin Brown, MD, University of Michigan.
The guideline was prepared on behalf of and approved by the AHA Stroke Council’s Scientific Statements Oversight Committee on Clinical Practice Guidelines. The writing group included representatives from the AHA/ASA and the American Academy of Neurology. The guideline has been endorsed by the American Association of Neurological Surgeons/Congress of Neurological Surgeons and the Society of Vascular and Interventional Neurology. It has also been affirmed by the AAN as an educational tool for neurologists.
The research had no commercial funding.
A version of this article first appeared on Medscape.com.
When possible, diagnostic tests to determine the cause of a first stroke or transient ischemic attack (TIA) should be completed within 48 hours after symptom onset, the American Heart Association/American Stroke Association said in an updated clinical practice guideline.
“It is critically important to understand the best ways to prevent another stroke once someone has had a stroke or a TIA,” Dawn O. Kleindorfer, MD, chair of the guideline writing group, said in a news release.
“If we can pinpoint the cause of the first stroke or TIA, we can tailor strategies to prevent a second stroke,” said Dr. Kleindorfer, professor and chair, department of neurology, University of Michigan, Ann Arbor.
The updated guideline was published online May 24, 2021, in Stroke.
“The secondary prevention of stroke guideline is one of the ASA’s ‘flagship’ guidelines, last updated in 2014,” Dr. Kleindorfer said.
The update includes “a number of changes to the writing and formatting of this guideline to make it easier for professionals to understand and locate information more quickly, ultimately greatly improving patient care and preventing more strokes in our patients,” she noted.
Let pathogenic subtype guide prevention
For patients who have survived a stroke or TIA, management of vascular risk factors, particularly hypertension, diabetes, cholesterol/triglyceride levels, and smoking cessation, are key secondary prevention tactics, the guideline said.
Limiting salt intake and/or following a heart-healthy Mediterranean diet is also advised, as is engaging in at least moderate-intensity aerobic activity for at least 10 minutes four times a week or vigorous-intensity aerobic activity for at least 20 minutes twice a week.
“Approximately 80% of strokes can be prevented by controlling blood pressure, eating a healthy diet, engaging in regular physical activity, not smoking and maintaining a healthy weight,” Amytis Towfighi, MD, vice chair of the guideline writing group and director of neurologic services, Los Angeles County Department of Health Services, noted in the release.
For health care professionals, the guideline said specific recommendations for secondary prevention often depend on the ischemic stroke/TIA subtype. “Therefore, new in this guideline is a section describing recommendations for the diagnostic workup after ischemic stroke, to define ischemic stroke pathogenesis (when possible), and to identify targets for treatment to reduce the risk of recurrent ischemic stroke. Recommendations are now segregated by pathogenetic subtype,” the guideline stated.
Among the recommendations:
- Use multidisciplinary care teams to personalize care for patients and employ shared decision-making with the patient to develop care plans that incorporate a patient’s wishes, goals, and concerns.
- Screen for and initiate anticoagulant drug therapy to reduce recurrent events.
- Prescribe antithrombotic therapy, including antiplatelets or anticoagulants, in the absence of contraindications. The guideline noted that the combination of antiplatelets and anticoagulation is typically not recommended for preventing second strokes and that dual antiplatelet therapy (DAPT) – taking along with a second medication to prevent blood clotting – is recommended in the short term and only for specific patients: those with early arriving minor stroke and high-risk TIA or severe symptomatic stenosis.
- Consider or carotid artery stenting for select patients with narrowing of carotid arteries.
- Aggressive medical management of risk factors and short-term DAPT are preferred for patients with severe intracranial stenosis thought to be the cause of first stroke or TIA.
- In some patients, it’s reasonable to consider percutaneous closure of .
The guideline is accompanied by a systematic review and meta-analysis regarding the benefits and risks of dual antiplatelet versus single antiplatelet therapy for secondary stroke prevention. The authors conclude that DAPT may be appropriate for select patients.
“Additional research is needed to determine: the optimal timing of starting treatment relative to the clinical event; the optimal duration of DAPT to maximize the risk-benefit ratio; whether additional populations excluded from POINT and CHANCE [two of the trials examined], such as those with major stroke, may also benefit from early DAPT; and whether certain genetic profiles eliminate the benefit of early DAPT,” concluded the reviewers, led by Devin Brown, MD, University of Michigan.
The guideline was prepared on behalf of and approved by the AHA Stroke Council’s Scientific Statements Oversight Committee on Clinical Practice Guidelines. The writing group included representatives from the AHA/ASA and the American Academy of Neurology. The guideline has been endorsed by the American Association of Neurological Surgeons/Congress of Neurological Surgeons and the Society of Vascular and Interventional Neurology. It has also been affirmed by the AAN as an educational tool for neurologists.
The research had no commercial funding.
A version of this article first appeared on Medscape.com.
Large vessel stroke linked to AstraZeneca COVID vaccine
The three cases (one of which was fatal) occurred in two women and one man in their 30s or 40s and involved blockages of the carotid and middle cerebral artery. Two of the three patients also had venous thrombosis involving the portal and cerebral venous system. All three also had extremely low platelet counts, confirmed antibodies to platelet factor 4, and raised D-dimer levels, all characteristic of the vaccine-induced immune thrombotic thrombocytopenia (VITT) reaction associated with the AstraZeneca vaccine.
They are described in detail in a letter published online on May 25 in the Journal of Neurology, Neurosurgery & Psychiatry
“These are [the] first detailed reports of arterial stroke believed to be caused by VITT after the AstraZeneca COVID vaccine, although stroke has been mentioned previously in the VITT data,” said senior author David Werring, PhD, FRCP.
“VITT has more commonly presented as CVST [Cerebral venous sinus thrombosis] which is stroke caused by a venous thrombosis; these cases are showing that it can also cause stroke caused by an arterial thrombosis,” explained Dr. Werring, professor of clinical neurology at the Stroke Research Centre, University College London.
“In patients who present with ischemic stroke, especially younger patients, and who have had the AstraZeneca vaccine within the past month, clinicians need to consider VITT as a possible cause, as there is a specific treatment needed for this syndrome,” he said.
Young patients presenting with ischemic stroke after receiving the AstraZeneca vaccine should urgently be evaluated for VITT with laboratory tests, including platelet count, D-dimers, fibrinogen, and anti-PF4 antibodies, the authors wrote, and then managed by a multidisciplinary team, including hematology, neurology, stroke, neurosurgery, and neuroradiology, for rapid access to treatments including intravenous immune globulin, methylprednisolone, plasmapheresis, and nonheparin anticoagulants such as fondaparinux, argatroban, or direct oral anticoagulants.
Dr. Werring noted that these reports do not add anything to the overall risk/benefit of the vaccine, as they are only describing three cases. “While VITT is very serious, the benefit of the vaccine still outweighs its risks,” he said. “Around 40% of patients hospitalized with COVID-19 experience some sort of thrombosis and about 1.5% have an ischemic stroke. Whereas latest figures from the U.K. estimate the incidence of VITT with the AstraZeneca vaccine of 1 in 50,000 to 1 in 100,000.
“Our report doesn’t suggest that VITT is more common than these latest figures estimate, but we are just drawing attention to an alternative presentation,” he added.
Three cases
The first patient in the current case series, a woman in her 30s, experienced an intermittent headache on the right side and around her eyes 6 days after the vaccine. Five days later, she awoke feeling drowsy and with weakness to her left face, arm, and leg.
Imaging revealed a blocked right middle cerebral artery with brain infarction and clots in the right portal vein. She underwent brain surgery to reduce the pressure in her skull, plasma removal and replacement, and received the anticoagulant fondaparinux, but she still unfortunately died.
The second patient, a woman in her late 30s, presented with headache, confusion, weakness in her left arm, and loss of vision on the left side 12 days after having received the vaccine. Imaging showed occlusion of both carotid arteries, as well as pulmonary embolism and a left cerebral venous sinus thrombosis.
Her platelet count increased following plasma removal and replacement and intravenous corticosteroids, and her condition improved after fondaparinux treatment.
The third patient, a man in his early 40s, presented 3 weeks after receiving his vaccination with problems speaking. Imaging showed a clot in the left middle cerebral artery, but there was no evidence of clots in the cerebral venous sinuses. He received a platelet and plasma transfusion, and fondaparinux, and remains stable.
High index of suspicion required
In a linked commentary, Hugh Markus, PhD, FRCP, professor of stroke medicine at the University of Cambridge, United Kingdom, wrote: “This report emphasizes that the immune mediated coagulopathy can also cause arterial thrombosis, including ischemic stroke, although venous thrombosis and especially cerebral venous sinus thrombosis appear more frequent.
“During the current period of COVID vaccination, a high index of suspicion is required to identify thrombotic episodes following vaccination,” he added. “However, it is important to remember that these side effects are rare and much less common than both cerebral venous thrombosis and ischemic stroke associated with COVID-19 infection itself.”
Risk/benefit unaltered
Several experts who commented on these reports for the Science Media Centre all agreed with Dr. Werring and Dr. Markus that these reports do not alter the current risk/benefit estimates with the vaccine.
Ian Douglas, PhD, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who sits on the U.K.’s Medicines and Healthcare Products Regulatory Agency’s Pharmacovigilance Expert Advisory Group, said: “The picture regarding the rare syndrome of blood clots combined with low platelet counts associated with the AstraZeneca vaccine is becoming clearer. Until now, the cases described have tended to involve clots in veins such as cerebral vein thrombosis. In this series of three case reports, we now have some evidence that the types of blood vessels affected include arteries as well as veins.”
“It’s important to stress that such cases remain very rare, and it’s certainly much rarer in people who have had the AstraZeneca vaccine than it is in people affected by COVID-19 itself,” Dr. Douglas emphasized.
“The description of the cases suggests the patients involved presented with the same kind of symptoms as already described in cases involving cerebral vein thrombosis, and they don’t suggest patients need to be on the alert for anything different,” he added.
“However, the emergence of details like this will help guide health professionals who may be faced with similar cases in future; the sooner such cases are recognized, the more chance they will quickly receive the right kind of treatment, hopefully leading to better outcomes.”
Will Lester, MBChB, PhD, consultant hematologist, University Hospitals Birmingham NHS Foundation Trust, said: “VITT remains a rare complication, and patients with a history of thrombosis, including stroke, should not consider themselves to be at any higher risk of this type of rare thrombosis after vaccination, and COVID infection itself is a significant risk for stroke and other types of thrombosis.”
Many countries have paused use of the AstraZeneca vaccine because of its link to the VITT syndrome or restricted its use to older people as the VITT reaction appears to be slightly more common in younger people. In the United Kingdom, the current recommendation is that individuals under 40 years of age should be offered an alternative to the AstraZeneca vaccine where possible.
A version of this article first appeared on Medscape.com.
The three cases (one of which was fatal) occurred in two women and one man in their 30s or 40s and involved blockages of the carotid and middle cerebral artery. Two of the three patients also had venous thrombosis involving the portal and cerebral venous system. All three also had extremely low platelet counts, confirmed antibodies to platelet factor 4, and raised D-dimer levels, all characteristic of the vaccine-induced immune thrombotic thrombocytopenia (VITT) reaction associated with the AstraZeneca vaccine.
They are described in detail in a letter published online on May 25 in the Journal of Neurology, Neurosurgery & Psychiatry
“These are [the] first detailed reports of arterial stroke believed to be caused by VITT after the AstraZeneca COVID vaccine, although stroke has been mentioned previously in the VITT data,” said senior author David Werring, PhD, FRCP.
“VITT has more commonly presented as CVST [Cerebral venous sinus thrombosis] which is stroke caused by a venous thrombosis; these cases are showing that it can also cause stroke caused by an arterial thrombosis,” explained Dr. Werring, professor of clinical neurology at the Stroke Research Centre, University College London.
“In patients who present with ischemic stroke, especially younger patients, and who have had the AstraZeneca vaccine within the past month, clinicians need to consider VITT as a possible cause, as there is a specific treatment needed for this syndrome,” he said.
Young patients presenting with ischemic stroke after receiving the AstraZeneca vaccine should urgently be evaluated for VITT with laboratory tests, including platelet count, D-dimers, fibrinogen, and anti-PF4 antibodies, the authors wrote, and then managed by a multidisciplinary team, including hematology, neurology, stroke, neurosurgery, and neuroradiology, for rapid access to treatments including intravenous immune globulin, methylprednisolone, plasmapheresis, and nonheparin anticoagulants such as fondaparinux, argatroban, or direct oral anticoagulants.
Dr. Werring noted that these reports do not add anything to the overall risk/benefit of the vaccine, as they are only describing three cases. “While VITT is very serious, the benefit of the vaccine still outweighs its risks,” he said. “Around 40% of patients hospitalized with COVID-19 experience some sort of thrombosis and about 1.5% have an ischemic stroke. Whereas latest figures from the U.K. estimate the incidence of VITT with the AstraZeneca vaccine of 1 in 50,000 to 1 in 100,000.
“Our report doesn’t suggest that VITT is more common than these latest figures estimate, but we are just drawing attention to an alternative presentation,” he added.
Three cases
The first patient in the current case series, a woman in her 30s, experienced an intermittent headache on the right side and around her eyes 6 days after the vaccine. Five days later, she awoke feeling drowsy and with weakness to her left face, arm, and leg.
Imaging revealed a blocked right middle cerebral artery with brain infarction and clots in the right portal vein. She underwent brain surgery to reduce the pressure in her skull, plasma removal and replacement, and received the anticoagulant fondaparinux, but she still unfortunately died.
The second patient, a woman in her late 30s, presented with headache, confusion, weakness in her left arm, and loss of vision on the left side 12 days after having received the vaccine. Imaging showed occlusion of both carotid arteries, as well as pulmonary embolism and a left cerebral venous sinus thrombosis.
Her platelet count increased following plasma removal and replacement and intravenous corticosteroids, and her condition improved after fondaparinux treatment.
The third patient, a man in his early 40s, presented 3 weeks after receiving his vaccination with problems speaking. Imaging showed a clot in the left middle cerebral artery, but there was no evidence of clots in the cerebral venous sinuses. He received a platelet and plasma transfusion, and fondaparinux, and remains stable.
High index of suspicion required
In a linked commentary, Hugh Markus, PhD, FRCP, professor of stroke medicine at the University of Cambridge, United Kingdom, wrote: “This report emphasizes that the immune mediated coagulopathy can also cause arterial thrombosis, including ischemic stroke, although venous thrombosis and especially cerebral venous sinus thrombosis appear more frequent.
“During the current period of COVID vaccination, a high index of suspicion is required to identify thrombotic episodes following vaccination,” he added. “However, it is important to remember that these side effects are rare and much less common than both cerebral venous thrombosis and ischemic stroke associated with COVID-19 infection itself.”
Risk/benefit unaltered
Several experts who commented on these reports for the Science Media Centre all agreed with Dr. Werring and Dr. Markus that these reports do not alter the current risk/benefit estimates with the vaccine.
Ian Douglas, PhD, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who sits on the U.K.’s Medicines and Healthcare Products Regulatory Agency’s Pharmacovigilance Expert Advisory Group, said: “The picture regarding the rare syndrome of blood clots combined with low platelet counts associated with the AstraZeneca vaccine is becoming clearer. Until now, the cases described have tended to involve clots in veins such as cerebral vein thrombosis. In this series of three case reports, we now have some evidence that the types of blood vessels affected include arteries as well as veins.”
“It’s important to stress that such cases remain very rare, and it’s certainly much rarer in people who have had the AstraZeneca vaccine than it is in people affected by COVID-19 itself,” Dr. Douglas emphasized.
“The description of the cases suggests the patients involved presented with the same kind of symptoms as already described in cases involving cerebral vein thrombosis, and they don’t suggest patients need to be on the alert for anything different,” he added.
“However, the emergence of details like this will help guide health professionals who may be faced with similar cases in future; the sooner such cases are recognized, the more chance they will quickly receive the right kind of treatment, hopefully leading to better outcomes.”
Will Lester, MBChB, PhD, consultant hematologist, University Hospitals Birmingham NHS Foundation Trust, said: “VITT remains a rare complication, and patients with a history of thrombosis, including stroke, should not consider themselves to be at any higher risk of this type of rare thrombosis after vaccination, and COVID infection itself is a significant risk for stroke and other types of thrombosis.”
Many countries have paused use of the AstraZeneca vaccine because of its link to the VITT syndrome or restricted its use to older people as the VITT reaction appears to be slightly more common in younger people. In the United Kingdom, the current recommendation is that individuals under 40 years of age should be offered an alternative to the AstraZeneca vaccine where possible.
A version of this article first appeared on Medscape.com.
The three cases (one of which was fatal) occurred in two women and one man in their 30s or 40s and involved blockages of the carotid and middle cerebral artery. Two of the three patients also had venous thrombosis involving the portal and cerebral venous system. All three also had extremely low platelet counts, confirmed antibodies to platelet factor 4, and raised D-dimer levels, all characteristic of the vaccine-induced immune thrombotic thrombocytopenia (VITT) reaction associated with the AstraZeneca vaccine.
They are described in detail in a letter published online on May 25 in the Journal of Neurology, Neurosurgery & Psychiatry
“These are [the] first detailed reports of arterial stroke believed to be caused by VITT after the AstraZeneca COVID vaccine, although stroke has been mentioned previously in the VITT data,” said senior author David Werring, PhD, FRCP.
“VITT has more commonly presented as CVST [Cerebral venous sinus thrombosis] which is stroke caused by a venous thrombosis; these cases are showing that it can also cause stroke caused by an arterial thrombosis,” explained Dr. Werring, professor of clinical neurology at the Stroke Research Centre, University College London.
“In patients who present with ischemic stroke, especially younger patients, and who have had the AstraZeneca vaccine within the past month, clinicians need to consider VITT as a possible cause, as there is a specific treatment needed for this syndrome,” he said.
Young patients presenting with ischemic stroke after receiving the AstraZeneca vaccine should urgently be evaluated for VITT with laboratory tests, including platelet count, D-dimers, fibrinogen, and anti-PF4 antibodies, the authors wrote, and then managed by a multidisciplinary team, including hematology, neurology, stroke, neurosurgery, and neuroradiology, for rapid access to treatments including intravenous immune globulin, methylprednisolone, plasmapheresis, and nonheparin anticoagulants such as fondaparinux, argatroban, or direct oral anticoagulants.
Dr. Werring noted that these reports do not add anything to the overall risk/benefit of the vaccine, as they are only describing three cases. “While VITT is very serious, the benefit of the vaccine still outweighs its risks,” he said. “Around 40% of patients hospitalized with COVID-19 experience some sort of thrombosis and about 1.5% have an ischemic stroke. Whereas latest figures from the U.K. estimate the incidence of VITT with the AstraZeneca vaccine of 1 in 50,000 to 1 in 100,000.
“Our report doesn’t suggest that VITT is more common than these latest figures estimate, but we are just drawing attention to an alternative presentation,” he added.
Three cases
The first patient in the current case series, a woman in her 30s, experienced an intermittent headache on the right side and around her eyes 6 days after the vaccine. Five days later, she awoke feeling drowsy and with weakness to her left face, arm, and leg.
Imaging revealed a blocked right middle cerebral artery with brain infarction and clots in the right portal vein. She underwent brain surgery to reduce the pressure in her skull, plasma removal and replacement, and received the anticoagulant fondaparinux, but she still unfortunately died.
The second patient, a woman in her late 30s, presented with headache, confusion, weakness in her left arm, and loss of vision on the left side 12 days after having received the vaccine. Imaging showed occlusion of both carotid arteries, as well as pulmonary embolism and a left cerebral venous sinus thrombosis.
Her platelet count increased following plasma removal and replacement and intravenous corticosteroids, and her condition improved after fondaparinux treatment.
The third patient, a man in his early 40s, presented 3 weeks after receiving his vaccination with problems speaking. Imaging showed a clot in the left middle cerebral artery, but there was no evidence of clots in the cerebral venous sinuses. He received a platelet and plasma transfusion, and fondaparinux, and remains stable.
High index of suspicion required
In a linked commentary, Hugh Markus, PhD, FRCP, professor of stroke medicine at the University of Cambridge, United Kingdom, wrote: “This report emphasizes that the immune mediated coagulopathy can also cause arterial thrombosis, including ischemic stroke, although venous thrombosis and especially cerebral venous sinus thrombosis appear more frequent.
“During the current period of COVID vaccination, a high index of suspicion is required to identify thrombotic episodes following vaccination,” he added. “However, it is important to remember that these side effects are rare and much less common than both cerebral venous thrombosis and ischemic stroke associated with COVID-19 infection itself.”
Risk/benefit unaltered
Several experts who commented on these reports for the Science Media Centre all agreed with Dr. Werring and Dr. Markus that these reports do not alter the current risk/benefit estimates with the vaccine.
Ian Douglas, PhD, professor of pharmacoepidemiology, London School of Hygiene & Tropical Medicine, who sits on the U.K.’s Medicines and Healthcare Products Regulatory Agency’s Pharmacovigilance Expert Advisory Group, said: “The picture regarding the rare syndrome of blood clots combined with low platelet counts associated with the AstraZeneca vaccine is becoming clearer. Until now, the cases described have tended to involve clots in veins such as cerebral vein thrombosis. In this series of three case reports, we now have some evidence that the types of blood vessels affected include arteries as well as veins.”
“It’s important to stress that such cases remain very rare, and it’s certainly much rarer in people who have had the AstraZeneca vaccine than it is in people affected by COVID-19 itself,” Dr. Douglas emphasized.
“The description of the cases suggests the patients involved presented with the same kind of symptoms as already described in cases involving cerebral vein thrombosis, and they don’t suggest patients need to be on the alert for anything different,” he added.
“However, the emergence of details like this will help guide health professionals who may be faced with similar cases in future; the sooner such cases are recognized, the more chance they will quickly receive the right kind of treatment, hopefully leading to better outcomes.”
Will Lester, MBChB, PhD, consultant hematologist, University Hospitals Birmingham NHS Foundation Trust, said: “VITT remains a rare complication, and patients with a history of thrombosis, including stroke, should not consider themselves to be at any higher risk of this type of rare thrombosis after vaccination, and COVID infection itself is a significant risk for stroke and other types of thrombosis.”
Many countries have paused use of the AstraZeneca vaccine because of its link to the VITT syndrome or restricted its use to older people as the VITT reaction appears to be slightly more common in younger people. In the United Kingdom, the current recommendation is that individuals under 40 years of age should be offered an alternative to the AstraZeneca vaccine where possible.
A version of this article first appeared on Medscape.com.
The end of happy hour? No safe level of alcohol for the brain
There is no safe amount of alcohol consumption for the brain; even moderate drinking adversely affects brain structure and function, according a British study of more 25,000 adults.
“This is one of the largest studies of alcohol and brain health to date,” Anya Topiwala, DPhil, University of Oxford (England), told this news organization.
“There have been previous claims the relationship between alcohol and brain health are J-shaped (ie., small amounts are protective), but we formally tested this and did not find it to be the case. In fact, we found that any level of alcohol was associated with poorer brain health, compared to no alcohol,” Dr. Topiwala added.
The study, which has not yet been peer reviewed, was published online May 12 in MedRxiv.
Global impact on the brain
Participants provided detailed information on their alcohol intake. The cohort included 691 never-drinkers, 617 former drinkers, and 24,069 current drinkers.
Median alcohol intake was 13.5 units (102 g) weekly. Almost half of the sample (48.2%) were drinking above current UK low-risk guidelines (14 units, 112 g weekly), but few were heavy drinkers (>50 units, 400 g weekly).
After adjusting for all known potential confounders and multiple comparisons, a higher volume of alcohol consumed per week was associated with lower gray matter in “almost all areas of the brain,” Dr. Topiwala said in an interview.
Alcohol consumption accounted for up to 0.8% of gray matter volume variance. “The size of the effect is small, albeit greater than any other modifiable risk factor. These brain changes have been previously linked to aging, poorer performance on memory changes, and dementia,” Dr. Topiwala said.
Widespread negative associations were also found between drinking alcohol and all the measures of white matter integrity that were assessed. There was a significant positive association between alcohol consumption and resting-state functional connectivity.
Higher blood pressure and body mass index “steepened” the negative associations between alcohol and brain health, and binge drinking had additive negative effects on brain structure beyond the absolute volume consumed.
There was no evidence that the risk for alcohol-related brain harm differs according to the type of alcohol consumed (wine, beer, or spirits).
A key limitation of the study is that the study population from the UK Biobank represents a sample that is healthier, better educated, and less deprived and is characterized by less ethnic diversity than the general population. “As with any observational study, we cannot infer causality from association,” the authors note.
What remains unclear, they say, is the duration of drinking needed to cause an effect on the brain. It may be that vulnerability is increased during periods of life in which dynamic brain changes occur, such as adolescence and older age.
They also note that some studies of alcohol-dependent individuals have suggested that at least some brain damage is reversible upon abstinence. Whether that is true for moderate drinkers is unknown.
On the basis of their findings, there is “no safe dose of alcohol for the brain,” Dr. Topiwala and colleagues conclude. They suggest that current low-risk drinking guidelines be revisited to take account of brain effects.
Experts weigh in
Several experts weighed in on the study in a statement from the nonprofit UK Science Media Center.
Paul Matthews, MD, head of the department of brain sciences, Imperial College London, noted that this “carefully performed preliminary report extends our earlier UK Dementia Research Institute study of a smaller group from same UK Biobank population also showing that even moderate drinking is associated with greater atrophy of the brain, as well as injury to the heart and liver.”
Dr. Matthews said the investigators’ conclusion that there is no safe threshold below which alcohol consumption has no toxic effects “echoes our own. We join with them in suggesting that current public health guidelines concerning alcohol consumption may need to be revisited.”
Rebecca Dewey, PhD, research fellow in neuroimaging, University of Nottingham (England), cautioned that “the degree to which very small changes in brain volume are harmful” is unknown.
“While there was no threshold under which alcohol consumption did not cause changes in the brain, there may a degree of brain volume difference that is irrelevant to brain health. We don’t know what these people’s brains looked like before they drank alcohol, so the brain may have learned to cope/compensate,” Dewey said.
Sadie Boniface, PhD, head of research at the Institute of Alcohol Studies and visiting researcher at King’s College London, said, “While we can’t yet say for sure whether there is ‘no safe level’ of alcohol regarding brain health at the moment, it has been known for decades that heavy drinking is bad for brain health.
“We also shouldn’t forget alcohol affects all parts of the body and there are multiple health risks. For example, it is already known there is ‘no safe level’ of alcohol consumption for the seven types of cancer caused by alcohol, as identified by the UK chief medical officers,” Dr. Boniface said.
The study was supported in part by the Wellcome Trust, Li Ka Shing Center for Health Information and Discovery, the National Institutes of Health, and the UK Medical Research Council. Dr. Topiwala, Dr. Boniface, Dr. Dewey, and Dr. Matthews have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
There is no safe amount of alcohol consumption for the brain; even moderate drinking adversely affects brain structure and function, according a British study of more 25,000 adults.
“This is one of the largest studies of alcohol and brain health to date,” Anya Topiwala, DPhil, University of Oxford (England), told this news organization.
“There have been previous claims the relationship between alcohol and brain health are J-shaped (ie., small amounts are protective), but we formally tested this and did not find it to be the case. In fact, we found that any level of alcohol was associated with poorer brain health, compared to no alcohol,” Dr. Topiwala added.
The study, which has not yet been peer reviewed, was published online May 12 in MedRxiv.
Global impact on the brain
Participants provided detailed information on their alcohol intake. The cohort included 691 never-drinkers, 617 former drinkers, and 24,069 current drinkers.
Median alcohol intake was 13.5 units (102 g) weekly. Almost half of the sample (48.2%) were drinking above current UK low-risk guidelines (14 units, 112 g weekly), but few were heavy drinkers (>50 units, 400 g weekly).
After adjusting for all known potential confounders and multiple comparisons, a higher volume of alcohol consumed per week was associated with lower gray matter in “almost all areas of the brain,” Dr. Topiwala said in an interview.
Alcohol consumption accounted for up to 0.8% of gray matter volume variance. “The size of the effect is small, albeit greater than any other modifiable risk factor. These brain changes have been previously linked to aging, poorer performance on memory changes, and dementia,” Dr. Topiwala said.
Widespread negative associations were also found between drinking alcohol and all the measures of white matter integrity that were assessed. There was a significant positive association between alcohol consumption and resting-state functional connectivity.
Higher blood pressure and body mass index “steepened” the negative associations between alcohol and brain health, and binge drinking had additive negative effects on brain structure beyond the absolute volume consumed.
There was no evidence that the risk for alcohol-related brain harm differs according to the type of alcohol consumed (wine, beer, or spirits).
A key limitation of the study is that the study population from the UK Biobank represents a sample that is healthier, better educated, and less deprived and is characterized by less ethnic diversity than the general population. “As with any observational study, we cannot infer causality from association,” the authors note.
What remains unclear, they say, is the duration of drinking needed to cause an effect on the brain. It may be that vulnerability is increased during periods of life in which dynamic brain changes occur, such as adolescence and older age.
They also note that some studies of alcohol-dependent individuals have suggested that at least some brain damage is reversible upon abstinence. Whether that is true for moderate drinkers is unknown.
On the basis of their findings, there is “no safe dose of alcohol for the brain,” Dr. Topiwala and colleagues conclude. They suggest that current low-risk drinking guidelines be revisited to take account of brain effects.
Experts weigh in
Several experts weighed in on the study in a statement from the nonprofit UK Science Media Center.
Paul Matthews, MD, head of the department of brain sciences, Imperial College London, noted that this “carefully performed preliminary report extends our earlier UK Dementia Research Institute study of a smaller group from same UK Biobank population also showing that even moderate drinking is associated with greater atrophy of the brain, as well as injury to the heart and liver.”
Dr. Matthews said the investigators’ conclusion that there is no safe threshold below which alcohol consumption has no toxic effects “echoes our own. We join with them in suggesting that current public health guidelines concerning alcohol consumption may need to be revisited.”
Rebecca Dewey, PhD, research fellow in neuroimaging, University of Nottingham (England), cautioned that “the degree to which very small changes in brain volume are harmful” is unknown.
“While there was no threshold under which alcohol consumption did not cause changes in the brain, there may a degree of brain volume difference that is irrelevant to brain health. We don’t know what these people’s brains looked like before they drank alcohol, so the brain may have learned to cope/compensate,” Dewey said.
Sadie Boniface, PhD, head of research at the Institute of Alcohol Studies and visiting researcher at King’s College London, said, “While we can’t yet say for sure whether there is ‘no safe level’ of alcohol regarding brain health at the moment, it has been known for decades that heavy drinking is bad for brain health.
“We also shouldn’t forget alcohol affects all parts of the body and there are multiple health risks. For example, it is already known there is ‘no safe level’ of alcohol consumption for the seven types of cancer caused by alcohol, as identified by the UK chief medical officers,” Dr. Boniface said.
The study was supported in part by the Wellcome Trust, Li Ka Shing Center for Health Information and Discovery, the National Institutes of Health, and the UK Medical Research Council. Dr. Topiwala, Dr. Boniface, Dr. Dewey, and Dr. Matthews have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
There is no safe amount of alcohol consumption for the brain; even moderate drinking adversely affects brain structure and function, according a British study of more 25,000 adults.
“This is one of the largest studies of alcohol and brain health to date,” Anya Topiwala, DPhil, University of Oxford (England), told this news organization.
“There have been previous claims the relationship between alcohol and brain health are J-shaped (ie., small amounts are protective), but we formally tested this and did not find it to be the case. In fact, we found that any level of alcohol was associated with poorer brain health, compared to no alcohol,” Dr. Topiwala added.
The study, which has not yet been peer reviewed, was published online May 12 in MedRxiv.
Global impact on the brain
Participants provided detailed information on their alcohol intake. The cohort included 691 never-drinkers, 617 former drinkers, and 24,069 current drinkers.
Median alcohol intake was 13.5 units (102 g) weekly. Almost half of the sample (48.2%) were drinking above current UK low-risk guidelines (14 units, 112 g weekly), but few were heavy drinkers (>50 units, 400 g weekly).
After adjusting for all known potential confounders and multiple comparisons, a higher volume of alcohol consumed per week was associated with lower gray matter in “almost all areas of the brain,” Dr. Topiwala said in an interview.
Alcohol consumption accounted for up to 0.8% of gray matter volume variance. “The size of the effect is small, albeit greater than any other modifiable risk factor. These brain changes have been previously linked to aging, poorer performance on memory changes, and dementia,” Dr. Topiwala said.
Widespread negative associations were also found between drinking alcohol and all the measures of white matter integrity that were assessed. There was a significant positive association between alcohol consumption and resting-state functional connectivity.
Higher blood pressure and body mass index “steepened” the negative associations between alcohol and brain health, and binge drinking had additive negative effects on brain structure beyond the absolute volume consumed.
There was no evidence that the risk for alcohol-related brain harm differs according to the type of alcohol consumed (wine, beer, or spirits).
A key limitation of the study is that the study population from the UK Biobank represents a sample that is healthier, better educated, and less deprived and is characterized by less ethnic diversity than the general population. “As with any observational study, we cannot infer causality from association,” the authors note.
What remains unclear, they say, is the duration of drinking needed to cause an effect on the brain. It may be that vulnerability is increased during periods of life in which dynamic brain changes occur, such as adolescence and older age.
They also note that some studies of alcohol-dependent individuals have suggested that at least some brain damage is reversible upon abstinence. Whether that is true for moderate drinkers is unknown.
On the basis of their findings, there is “no safe dose of alcohol for the brain,” Dr. Topiwala and colleagues conclude. They suggest that current low-risk drinking guidelines be revisited to take account of brain effects.
Experts weigh in
Several experts weighed in on the study in a statement from the nonprofit UK Science Media Center.
Paul Matthews, MD, head of the department of brain sciences, Imperial College London, noted that this “carefully performed preliminary report extends our earlier UK Dementia Research Institute study of a smaller group from same UK Biobank population also showing that even moderate drinking is associated with greater atrophy of the brain, as well as injury to the heart and liver.”
Dr. Matthews said the investigators’ conclusion that there is no safe threshold below which alcohol consumption has no toxic effects “echoes our own. We join with them in suggesting that current public health guidelines concerning alcohol consumption may need to be revisited.”
Rebecca Dewey, PhD, research fellow in neuroimaging, University of Nottingham (England), cautioned that “the degree to which very small changes in brain volume are harmful” is unknown.
“While there was no threshold under which alcohol consumption did not cause changes in the brain, there may a degree of brain volume difference that is irrelevant to brain health. We don’t know what these people’s brains looked like before they drank alcohol, so the brain may have learned to cope/compensate,” Dewey said.
Sadie Boniface, PhD, head of research at the Institute of Alcohol Studies and visiting researcher at King’s College London, said, “While we can’t yet say for sure whether there is ‘no safe level’ of alcohol regarding brain health at the moment, it has been known for decades that heavy drinking is bad for brain health.
“We also shouldn’t forget alcohol affects all parts of the body and there are multiple health risks. For example, it is already known there is ‘no safe level’ of alcohol consumption for the seven types of cancer caused by alcohol, as identified by the UK chief medical officers,” Dr. Boniface said.
The study was supported in part by the Wellcome Trust, Li Ka Shing Center for Health Information and Discovery, the National Institutes of Health, and the UK Medical Research Council. Dr. Topiwala, Dr. Boniface, Dr. Dewey, and Dr. Matthews have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Young adults with epilepsy face higher mental illness risks
Young adults with epilepsy experience higher rates of anxiety, depression, and suicidality, compared with their counterparts in the general population, a new study shows.
The findings, based on a study of 144 young adults with epilepsy (YAWE), was published recently in Epilepsy & Behavior.
“People with epilepsy (PWE) are at a significantly higher risk of experiencing mental health difficulties, compared with healthy controls and individuals with other [long-term conditions] such as asthma and diabetes,” according to Rachel Batchelor, MSc, and Michelle D. Taylor, PhD, of the University of London (England) in Surrey.
Young adulthood, which encompasses people aged 18-25 years, has been identified as “a peak age of onset for anxiety and depression,” but mental health in young adults with epilepsy in particular has not been well studied, they wrote.
The survey measured current mental health symptoms, including anxiety, depression, and suicidality, as well as sociodemographic and epilepsy-related factors, coping strategies, and social support (Epilepsy Behav. 2021 May;118:107911. doi: 10.1016/j.yebeh.2021.107911).
The average age of the respondents was 21.6 years, 61% were female, and 88% were of White British ethnicity. A total of 88 participants were single, 48 were in a relationship, and 8 were married or engaged. About one-third (38%) worked full-time, and 28.5% were full-time university students, 18.8% worked part-time, and 8.3% were unemployed and not students. The average age of seizure onset was 12.4 years.
Overall, 116 (80.6%) of the survey respondents met the criteria for anxiety, 110 (76.4%) for depression, and 51 (35.4%) for suicidality.
Ratings of all three of these conditions were significantly higher in females, compared with males, the researchers noted. Anxiety, depression, and suicidality also were rated higher for individuals who waited more than 1 year vs. less than 1 year for an epilepsy diagnosis from the time of seizure onset, for those suffering from anti-seizure medication side effects vs. no side effects, and for those with comorbid conditions vs. no comorbid conditions.
Avoidant-focused coping strategies were positively correlated with anxiety, depression, and suicidality, while problem-focused coping and meaning-focused coping were negatively correlated, the researchers said. In addition, those who reported greater levels of support from friends had lower rates of anxiety and depression, and those who reported greater levels of support from family had lower rates of suicidality.
The study findings were limited by several factors, including the relatively homogenous population, and the absence of data on current anxiety and depression medications and additional professional support, the researchers noted.
However, the results extend the research on mental health in people with epilepsy, and the study is the first known to focus on the young adult population with epilepsy, they said.
“The high rates of anxiety, depression, and suicidality underscore the need for better integration of mental health provision into epilepsy care,” the researchers wrote. “While it would be premature to base recommendations for treating anxiety, depression, and suicidality in YAWE on the current study, investigating the efficacy of psychological interventions (for example, [acceptance and commitment therapy], [compassion-focused therapy], peer support, and family-based [therapy]) designed to address the psychosocial variables shown to independently predict mental health outcomes in YAWE would be worthy future research avenues,” they concluded.
The study received no outside funding, and the researchers disclosed no financial conflicts.
Young adults with epilepsy experience higher rates of anxiety, depression, and suicidality, compared with their counterparts in the general population, a new study shows.
The findings, based on a study of 144 young adults with epilepsy (YAWE), was published recently in Epilepsy & Behavior.
“People with epilepsy (PWE) are at a significantly higher risk of experiencing mental health difficulties, compared with healthy controls and individuals with other [long-term conditions] such as asthma and diabetes,” according to Rachel Batchelor, MSc, and Michelle D. Taylor, PhD, of the University of London (England) in Surrey.
Young adulthood, which encompasses people aged 18-25 years, has been identified as “a peak age of onset for anxiety and depression,” but mental health in young adults with epilepsy in particular has not been well studied, they wrote.
The survey measured current mental health symptoms, including anxiety, depression, and suicidality, as well as sociodemographic and epilepsy-related factors, coping strategies, and social support (Epilepsy Behav. 2021 May;118:107911. doi: 10.1016/j.yebeh.2021.107911).
The average age of the respondents was 21.6 years, 61% were female, and 88% were of White British ethnicity. A total of 88 participants were single, 48 were in a relationship, and 8 were married or engaged. About one-third (38%) worked full-time, and 28.5% were full-time university students, 18.8% worked part-time, and 8.3% were unemployed and not students. The average age of seizure onset was 12.4 years.
Overall, 116 (80.6%) of the survey respondents met the criteria for anxiety, 110 (76.4%) for depression, and 51 (35.4%) for suicidality.
Ratings of all three of these conditions were significantly higher in females, compared with males, the researchers noted. Anxiety, depression, and suicidality also were rated higher for individuals who waited more than 1 year vs. less than 1 year for an epilepsy diagnosis from the time of seizure onset, for those suffering from anti-seizure medication side effects vs. no side effects, and for those with comorbid conditions vs. no comorbid conditions.
Avoidant-focused coping strategies were positively correlated with anxiety, depression, and suicidality, while problem-focused coping and meaning-focused coping were negatively correlated, the researchers said. In addition, those who reported greater levels of support from friends had lower rates of anxiety and depression, and those who reported greater levels of support from family had lower rates of suicidality.
The study findings were limited by several factors, including the relatively homogenous population, and the absence of data on current anxiety and depression medications and additional professional support, the researchers noted.
However, the results extend the research on mental health in people with epilepsy, and the study is the first known to focus on the young adult population with epilepsy, they said.
“The high rates of anxiety, depression, and suicidality underscore the need for better integration of mental health provision into epilepsy care,” the researchers wrote. “While it would be premature to base recommendations for treating anxiety, depression, and suicidality in YAWE on the current study, investigating the efficacy of psychological interventions (for example, [acceptance and commitment therapy], [compassion-focused therapy], peer support, and family-based [therapy]) designed to address the psychosocial variables shown to independently predict mental health outcomes in YAWE would be worthy future research avenues,” they concluded.
The study received no outside funding, and the researchers disclosed no financial conflicts.
Young adults with epilepsy experience higher rates of anxiety, depression, and suicidality, compared with their counterparts in the general population, a new study shows.
The findings, based on a study of 144 young adults with epilepsy (YAWE), was published recently in Epilepsy & Behavior.
“People with epilepsy (PWE) are at a significantly higher risk of experiencing mental health difficulties, compared with healthy controls and individuals with other [long-term conditions] such as asthma and diabetes,” according to Rachel Batchelor, MSc, and Michelle D. Taylor, PhD, of the University of London (England) in Surrey.
Young adulthood, which encompasses people aged 18-25 years, has been identified as “a peak age of onset for anxiety and depression,” but mental health in young adults with epilepsy in particular has not been well studied, they wrote.
The survey measured current mental health symptoms, including anxiety, depression, and suicidality, as well as sociodemographic and epilepsy-related factors, coping strategies, and social support (Epilepsy Behav. 2021 May;118:107911. doi: 10.1016/j.yebeh.2021.107911).
The average age of the respondents was 21.6 years, 61% were female, and 88% were of White British ethnicity. A total of 88 participants were single, 48 were in a relationship, and 8 were married or engaged. About one-third (38%) worked full-time, and 28.5% were full-time university students, 18.8% worked part-time, and 8.3% were unemployed and not students. The average age of seizure onset was 12.4 years.
Overall, 116 (80.6%) of the survey respondents met the criteria for anxiety, 110 (76.4%) for depression, and 51 (35.4%) for suicidality.
Ratings of all three of these conditions were significantly higher in females, compared with males, the researchers noted. Anxiety, depression, and suicidality also were rated higher for individuals who waited more than 1 year vs. less than 1 year for an epilepsy diagnosis from the time of seizure onset, for those suffering from anti-seizure medication side effects vs. no side effects, and for those with comorbid conditions vs. no comorbid conditions.
Avoidant-focused coping strategies were positively correlated with anxiety, depression, and suicidality, while problem-focused coping and meaning-focused coping were negatively correlated, the researchers said. In addition, those who reported greater levels of support from friends had lower rates of anxiety and depression, and those who reported greater levels of support from family had lower rates of suicidality.
The study findings were limited by several factors, including the relatively homogenous population, and the absence of data on current anxiety and depression medications and additional professional support, the researchers noted.
However, the results extend the research on mental health in people with epilepsy, and the study is the first known to focus on the young adult population with epilepsy, they said.
“The high rates of anxiety, depression, and suicidality underscore the need for better integration of mental health provision into epilepsy care,” the researchers wrote. “While it would be premature to base recommendations for treating anxiety, depression, and suicidality in YAWE on the current study, investigating the efficacy of psychological interventions (for example, [acceptance and commitment therapy], [compassion-focused therapy], peer support, and family-based [therapy]) designed to address the psychosocial variables shown to independently predict mental health outcomes in YAWE would be worthy future research avenues,” they concluded.
The study received no outside funding, and the researchers disclosed no financial conflicts.
FROM EPILEPSY & BEHAVIOR
Psychosis, depression tied to neurodegeneration in Parkinson’s
Depression and psychosis are significantly associated with neuronal loss and gliosis – but not with Lewy body scores – in Parkinson’s disease, data from analyses of the brains of 175 patients suggest.
Previous research has suggested a link between neuronal loss and depression in Parkinson’s disease (PD) but the impact of Lewy bodies has not been well studied, Nicole Mercado Fischer, MPH, of Johns Hopkins University, Baltimore, and colleagues wrote.
Evaluating Lewy body scores and neuronal loss/gliosis in the substantia nigra pars compacta (SN) and locus coeruleus (LC) could increase understanding of pathophysiology in PD, they said.
In a study published in the American Journal of Geriatric Psychiatry, the researchers analyzed the brains of 175 individuals with a primary diagnosis of PD.
A total of 98 participants had diagnoses of psychosis, 88 had depression, and 55 had anxiety. The average age of onset for PD was 62.4 years; 67.4% of the subjects were male, and 97.8% were White. The mean duration of illness was 16 years, and the average age at death was 78 years.
Psychosis was significantly associated with severe neuronal loss and gliosis in both the LC and SN (P = .048 and P = .042, respectively). Depression was significantly associated with severe neuronal loss in the SN (P = .042) but not in the LC. Anxiety was not associated with severe neuronal loss in either brain region. These results remained significant after a multivariate analysis, the researchers noted. However, Lewy body scores were not associated with any neuropsychiatric symptom, and severity of neuronal loss and gliosis was not correlated with Lewy body scores.
The study findings were limited by several factors, including the retrospective design and inability to collect pathology data for all patients, the researchers noted. Also, in some cases, the collection of clinical data and observation of brain tissue pathology took place years apart, and the researchers did not assess medication records.
However, the results were strengthened by the large sample size and “further support the notion that in vivo clinical symptoms of PD are either not caused by Lewy body pathology or that the relationship is confounded by the time of autopsy,” they said. and eventually by using new functional imaging techniques in vivo.”
The researchers had no financial conflicts to disclose. Two coauthors were supported in part by the National Institutes of Health.
Depression and psychosis are significantly associated with neuronal loss and gliosis – but not with Lewy body scores – in Parkinson’s disease, data from analyses of the brains of 175 patients suggest.
Previous research has suggested a link between neuronal loss and depression in Parkinson’s disease (PD) but the impact of Lewy bodies has not been well studied, Nicole Mercado Fischer, MPH, of Johns Hopkins University, Baltimore, and colleagues wrote.
Evaluating Lewy body scores and neuronal loss/gliosis in the substantia nigra pars compacta (SN) and locus coeruleus (LC) could increase understanding of pathophysiology in PD, they said.
In a study published in the American Journal of Geriatric Psychiatry, the researchers analyzed the brains of 175 individuals with a primary diagnosis of PD.
A total of 98 participants had diagnoses of psychosis, 88 had depression, and 55 had anxiety. The average age of onset for PD was 62.4 years; 67.4% of the subjects were male, and 97.8% were White. The mean duration of illness was 16 years, and the average age at death was 78 years.
Psychosis was significantly associated with severe neuronal loss and gliosis in both the LC and SN (P = .048 and P = .042, respectively). Depression was significantly associated with severe neuronal loss in the SN (P = .042) but not in the LC. Anxiety was not associated with severe neuronal loss in either brain region. These results remained significant after a multivariate analysis, the researchers noted. However, Lewy body scores were not associated with any neuropsychiatric symptom, and severity of neuronal loss and gliosis was not correlated with Lewy body scores.
The study findings were limited by several factors, including the retrospective design and inability to collect pathology data for all patients, the researchers noted. Also, in some cases, the collection of clinical data and observation of brain tissue pathology took place years apart, and the researchers did not assess medication records.
However, the results were strengthened by the large sample size and “further support the notion that in vivo clinical symptoms of PD are either not caused by Lewy body pathology or that the relationship is confounded by the time of autopsy,” they said. and eventually by using new functional imaging techniques in vivo.”
The researchers had no financial conflicts to disclose. Two coauthors were supported in part by the National Institutes of Health.
Depression and psychosis are significantly associated with neuronal loss and gliosis – but not with Lewy body scores – in Parkinson’s disease, data from analyses of the brains of 175 patients suggest.
Previous research has suggested a link between neuronal loss and depression in Parkinson’s disease (PD) but the impact of Lewy bodies has not been well studied, Nicole Mercado Fischer, MPH, of Johns Hopkins University, Baltimore, and colleagues wrote.
Evaluating Lewy body scores and neuronal loss/gliosis in the substantia nigra pars compacta (SN) and locus coeruleus (LC) could increase understanding of pathophysiology in PD, they said.
In a study published in the American Journal of Geriatric Psychiatry, the researchers analyzed the brains of 175 individuals with a primary diagnosis of PD.
A total of 98 participants had diagnoses of psychosis, 88 had depression, and 55 had anxiety. The average age of onset for PD was 62.4 years; 67.4% of the subjects were male, and 97.8% were White. The mean duration of illness was 16 years, and the average age at death was 78 years.
Psychosis was significantly associated with severe neuronal loss and gliosis in both the LC and SN (P = .048 and P = .042, respectively). Depression was significantly associated with severe neuronal loss in the SN (P = .042) but not in the LC. Anxiety was not associated with severe neuronal loss in either brain region. These results remained significant after a multivariate analysis, the researchers noted. However, Lewy body scores were not associated with any neuropsychiatric symptom, and severity of neuronal loss and gliosis was not correlated with Lewy body scores.
The study findings were limited by several factors, including the retrospective design and inability to collect pathology data for all patients, the researchers noted. Also, in some cases, the collection of clinical data and observation of brain tissue pathology took place years apart, and the researchers did not assess medication records.
However, the results were strengthened by the large sample size and “further support the notion that in vivo clinical symptoms of PD are either not caused by Lewy body pathology or that the relationship is confounded by the time of autopsy,” they said. and eventually by using new functional imaging techniques in vivo.”
The researchers had no financial conflicts to disclose. Two coauthors were supported in part by the National Institutes of Health.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
Healthy lifestyle can reduce dementia risk despite family history
Individuals at increased risk for dementia because of family history can reduce that risk by adopting healthy lifestyle behaviors, data from more than 300,000 adults aged 50-73 years suggest.
Having a parent or sibling with dementia can increase a person’s risk of developing dementia themselves by nearly 75%, compared with someone with no first-degree family history of dementia, according to Angelique Brellenthin, PhD, of Iowa State University, Ames, and colleagues.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting sponsored by the American Heart Association, the researchers reviewed information for 302,239 men and women who were enrolled in the U.K. Biobank, a population-based study of more than 500,000 individuals in the United Kingdom, between 2006 and 2010.
The study participants had no evidence of dementia at baseline, and completed questionnaires about family history and lifestyle. The questions included details about six healthy lifestyle behaviors: eating a healthy diet, engaging in at least 150 minutes of moderate to vigorous physical activity per week, sleeping 6-9 hours each night, drinking alcohol in moderation, not smoking, and maintaining a body mass index below the obese level (less than 30 kg/m2).
The researchers identified 1,698 participants (0.6%) who developed dementia over an average follow-up period of 8 years. Those with a family history (first-degree relative) of dementia had a 70% increased risk of dementia, compared with those who had no such family history.
Overall, individuals who engaged in all six healthy behaviors reduced their risk of dementia by about half, compared with those who engaged in two or fewer healthy behaviors. Engaging in three healthy behaviors reduced the risk of dementia by 30%, compared with engaging in two or fewer healthy behaviors, and this association held after controlling not only for family history of dementia, but also for other dementia risk factors such as age, sex, race, and education level, as well as high blood pressure, high cholesterol, and the presence of type 2 diabetes.
Similarly, among participants with a family history of dementia, those who engaged in three healthy lifestyle behaviors showed a 25%-35% reduction in dementia risk, compared with those who engaged in two or fewer healthy behaviors.
The study findings were limited by several factors including the inability to prove that lifestyle can cause or prevent dementia, only to show an association, the researchers noted. Also, the findings were limited by the reliance on self-reports, rather than genetic data, to confirm familial dementia.
However, the findings were strengthened by the large sample size, and the results suggest that a healthy lifestyle can impact cognitive health, and support the value of encouraging healthy behaviors in general, and especially among individuals with a family history of dementia, they said.
Small changes may promote prevention
The study is important now because, as the population ages, many individuals have a family member who has had dementia, said lead author Dr. Brellenthin, in an interview. “It’s important to understand how lifestyle behaviors affect the risk of dementia when it runs in families,” she said.
Dr. Brellenthin said she was surprised by some of the findings. “It was surprising to see that the risk of dementia was reduced with just three healthy behaviors [but was further reduced as you added more behaviors] compared to two or fewer behaviors. However, it was not surprising to see that these same lifestyle behaviors that tend to be good for the heart and body are also good for the brain.”
The evidence that following just three healthy behaviors can reduce the risk of dementia by 25%-35% for individuals with a familial history of dementia has clinical implications, Dr. Brellenthin said. “Many people are already following some of these behaviors like not smoking, so it might be possible to focus on adding just one more behavior, like getting enough sleep, and going from there.”
Commenting on the study, AHA President Mitchell S. V. Elkind, MD, said that the study “tells us that, yes, family history is important [in determining the risk of dementia], and much of that may be driven by genetic factors, but some of that impact can be mitigated or decreased by engaging in those important behaviors that we know are good to maintain brain health.
“The tricky thing, of course, is getting people to engage in these behaviors. That’s where a lot of work in the future will be: changing people’s behavior to become more healthy, and figuring out exactly which behaviors may be the easiest to engage in and be most likely to have public health impact,” added Dr. Elkind, professor of neurology and epidemiology at Columbia University and attending neurologist at New York–Presbyterian/Columbia University Irving Medical Center, New York.
The study received no outside funding, but the was research was conducted using the U.K. Biobank resources. The researchers had no financial conflicts to disclose.
Individuals at increased risk for dementia because of family history can reduce that risk by adopting healthy lifestyle behaviors, data from more than 300,000 adults aged 50-73 years suggest.
Having a parent or sibling with dementia can increase a person’s risk of developing dementia themselves by nearly 75%, compared with someone with no first-degree family history of dementia, according to Angelique Brellenthin, PhD, of Iowa State University, Ames, and colleagues.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting sponsored by the American Heart Association, the researchers reviewed information for 302,239 men and women who were enrolled in the U.K. Biobank, a population-based study of more than 500,000 individuals in the United Kingdom, between 2006 and 2010.
The study participants had no evidence of dementia at baseline, and completed questionnaires about family history and lifestyle. The questions included details about six healthy lifestyle behaviors: eating a healthy diet, engaging in at least 150 minutes of moderate to vigorous physical activity per week, sleeping 6-9 hours each night, drinking alcohol in moderation, not smoking, and maintaining a body mass index below the obese level (less than 30 kg/m2).
The researchers identified 1,698 participants (0.6%) who developed dementia over an average follow-up period of 8 years. Those with a family history (first-degree relative) of dementia had a 70% increased risk of dementia, compared with those who had no such family history.
Overall, individuals who engaged in all six healthy behaviors reduced their risk of dementia by about half, compared with those who engaged in two or fewer healthy behaviors. Engaging in three healthy behaviors reduced the risk of dementia by 30%, compared with engaging in two or fewer healthy behaviors, and this association held after controlling not only for family history of dementia, but also for other dementia risk factors such as age, sex, race, and education level, as well as high blood pressure, high cholesterol, and the presence of type 2 diabetes.
Similarly, among participants with a family history of dementia, those who engaged in three healthy lifestyle behaviors showed a 25%-35% reduction in dementia risk, compared with those who engaged in two or fewer healthy behaviors.
The study findings were limited by several factors including the inability to prove that lifestyle can cause or prevent dementia, only to show an association, the researchers noted. Also, the findings were limited by the reliance on self-reports, rather than genetic data, to confirm familial dementia.
However, the findings were strengthened by the large sample size, and the results suggest that a healthy lifestyle can impact cognitive health, and support the value of encouraging healthy behaviors in general, and especially among individuals with a family history of dementia, they said.
Small changes may promote prevention
The study is important now because, as the population ages, many individuals have a family member who has had dementia, said lead author Dr. Brellenthin, in an interview. “It’s important to understand how lifestyle behaviors affect the risk of dementia when it runs in families,” she said.
Dr. Brellenthin said she was surprised by some of the findings. “It was surprising to see that the risk of dementia was reduced with just three healthy behaviors [but was further reduced as you added more behaviors] compared to two or fewer behaviors. However, it was not surprising to see that these same lifestyle behaviors that tend to be good for the heart and body are also good for the brain.”
The evidence that following just three healthy behaviors can reduce the risk of dementia by 25%-35% for individuals with a familial history of dementia has clinical implications, Dr. Brellenthin said. “Many people are already following some of these behaviors like not smoking, so it might be possible to focus on adding just one more behavior, like getting enough sleep, and going from there.”
Commenting on the study, AHA President Mitchell S. V. Elkind, MD, said that the study “tells us that, yes, family history is important [in determining the risk of dementia], and much of that may be driven by genetic factors, but some of that impact can be mitigated or decreased by engaging in those important behaviors that we know are good to maintain brain health.
“The tricky thing, of course, is getting people to engage in these behaviors. That’s where a lot of work in the future will be: changing people’s behavior to become more healthy, and figuring out exactly which behaviors may be the easiest to engage in and be most likely to have public health impact,” added Dr. Elkind, professor of neurology and epidemiology at Columbia University and attending neurologist at New York–Presbyterian/Columbia University Irving Medical Center, New York.
The study received no outside funding, but the was research was conducted using the U.K. Biobank resources. The researchers had no financial conflicts to disclose.
Individuals at increased risk for dementia because of family history can reduce that risk by adopting healthy lifestyle behaviors, data from more than 300,000 adults aged 50-73 years suggest.
Having a parent or sibling with dementia can increase a person’s risk of developing dementia themselves by nearly 75%, compared with someone with no first-degree family history of dementia, according to Angelique Brellenthin, PhD, of Iowa State University, Ames, and colleagues.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting sponsored by the American Heart Association, the researchers reviewed information for 302,239 men and women who were enrolled in the U.K. Biobank, a population-based study of more than 500,000 individuals in the United Kingdom, between 2006 and 2010.
The study participants had no evidence of dementia at baseline, and completed questionnaires about family history and lifestyle. The questions included details about six healthy lifestyle behaviors: eating a healthy diet, engaging in at least 150 minutes of moderate to vigorous physical activity per week, sleeping 6-9 hours each night, drinking alcohol in moderation, not smoking, and maintaining a body mass index below the obese level (less than 30 kg/m2).
The researchers identified 1,698 participants (0.6%) who developed dementia over an average follow-up period of 8 years. Those with a family history (first-degree relative) of dementia had a 70% increased risk of dementia, compared with those who had no such family history.
Overall, individuals who engaged in all six healthy behaviors reduced their risk of dementia by about half, compared with those who engaged in two or fewer healthy behaviors. Engaging in three healthy behaviors reduced the risk of dementia by 30%, compared with engaging in two or fewer healthy behaviors, and this association held after controlling not only for family history of dementia, but also for other dementia risk factors such as age, sex, race, and education level, as well as high blood pressure, high cholesterol, and the presence of type 2 diabetes.
Similarly, among participants with a family history of dementia, those who engaged in three healthy lifestyle behaviors showed a 25%-35% reduction in dementia risk, compared with those who engaged in two or fewer healthy behaviors.
The study findings were limited by several factors including the inability to prove that lifestyle can cause or prevent dementia, only to show an association, the researchers noted. Also, the findings were limited by the reliance on self-reports, rather than genetic data, to confirm familial dementia.
However, the findings were strengthened by the large sample size, and the results suggest that a healthy lifestyle can impact cognitive health, and support the value of encouraging healthy behaviors in general, and especially among individuals with a family history of dementia, they said.
Small changes may promote prevention
The study is important now because, as the population ages, many individuals have a family member who has had dementia, said lead author Dr. Brellenthin, in an interview. “It’s important to understand how lifestyle behaviors affect the risk of dementia when it runs in families,” she said.
Dr. Brellenthin said she was surprised by some of the findings. “It was surprising to see that the risk of dementia was reduced with just three healthy behaviors [but was further reduced as you added more behaviors] compared to two or fewer behaviors. However, it was not surprising to see that these same lifestyle behaviors that tend to be good for the heart and body are also good for the brain.”
The evidence that following just three healthy behaviors can reduce the risk of dementia by 25%-35% for individuals with a familial history of dementia has clinical implications, Dr. Brellenthin said. “Many people are already following some of these behaviors like not smoking, so it might be possible to focus on adding just one more behavior, like getting enough sleep, and going from there.”
Commenting on the study, AHA President Mitchell S. V. Elkind, MD, said that the study “tells us that, yes, family history is important [in determining the risk of dementia], and much of that may be driven by genetic factors, but some of that impact can be mitigated or decreased by engaging in those important behaviors that we know are good to maintain brain health.
“The tricky thing, of course, is getting people to engage in these behaviors. That’s where a lot of work in the future will be: changing people’s behavior to become more healthy, and figuring out exactly which behaviors may be the easiest to engage in and be most likely to have public health impact,” added Dr. Elkind, professor of neurology and epidemiology at Columbia University and attending neurologist at New York–Presbyterian/Columbia University Irving Medical Center, New York.
The study received no outside funding, but the was research was conducted using the U.K. Biobank resources. The researchers had no financial conflicts to disclose.
FROM EPI/LIFESTYLE 2021










