User login
Angiography can wait for cardiac arrest without ST-elevation
A protocol of immediate angiography provided no mortality benefit over a strategy or delayed or more selective angiography among patients resuscitated from out-of-hospital cardiac arrest and without ST-segment elevation, new randomized results show.
“Among patients with resuscitated out-of-hospital cardiac arrest of possible cardiac origin, with shockable and nonshockable arrest rhythm and no ST-elevation, a strategy of immediate, unselected coronary angiography was not found to be beneficial over a delayed and selective approach with regard to the 30-day risk of all-cause death,” concluded principal investigator Steffen Desch, MD, University of Leipzig (Germany) Heart Center.
The results support previous results of the Coronary Angiography after Cardiac Arrest (COACT) trial, in patients with shockable rhythms, which also showed no differences in clinical outcomes between immediate and delayed coronary angiography at both 90 days and 1 year, he noted.
“What the clinicians wanted to know is, is it really necessary to get up at 3 a.m. in the morning to perform a coronary angiography on these patients, and that’s certainly out,” Dr. Desch said in an interview. “So, there’s really no room for this strategy anymore. You can take your time and wait a day or 2.”
These findings, from the TOMAHAWK trial, were presented Aug. 29 at the annual congress of the European Society of Cardiology and simultaneously published online in the New England Journal of Medicine.
Larger group without ST-segment elevation
Prognosis after out-of-hospital cardiac arrest is extremely poor, with an overall survival rate of less than 10%, Dr. Desch noted. “Actually, only 20% make it to the hospital; the vast majority of these patients die out in the field, so there’s really a great need in improving treatment.”
Acute coronary syndrome accounts for up to 60% of out-of-hospital arrests in which a cardiac cause has been identified, the authors wrote in their report. ST-segment elevation on postresuscitation electrocardiography “has good positive predictive value” for acute coronary lesions triggering the arrest, but in the far larger subgroup of patients without ST-segment elevation, “the spectrum of underlying causes is considerably broader and includes both cardiac and noncardiac causes.”
In patients with myocardial infarction, early revascularization would prevent negative consequences of myocardial injury, but unselected early coronary angiography would put patients not having an MI at unnecessary risk for procedural complications or delay in the diagnosis of the actual cause of their arrest, they noted.
In this trial, the researchers randomly assigned 554 patients from 31 sites in Germany and Denmark who were successfully resuscitated after cardiac arrest of possible cardiac origin to immediate transfer for coronary angiography or to initial intensive care assessment with delayed or selective angiography after a minimum delay of at least 1 day.
In the end, the average delay in this arm was 2 days, Dr. Desch noted. If the clinical course indicated that a coronary cause was unlikely, angiography might not be performed at all in this group.
No patient had ST-segment elevation on postresuscitation electrocardiography. The primary endpoint was death from any cause at 30 days; secondary end points were death from any cause or severe neurologic deficit at 30 days.
Results showed that 95% of patients in the immediate angiography group actually underwent the procedure, compared with 62% of those in the delayed group, a finding that was “logical” given the study design, he said.
At 30 days, 54% of patients in the immediate angiography group and 46% in the delayed group had died, a nonsignificant difference (P = .06). Because the researchers had performed an interim analysis, Dr. Desch explained, the final P value for significance in this trial was not .05, but rather .034, to account for multiple comparisons.
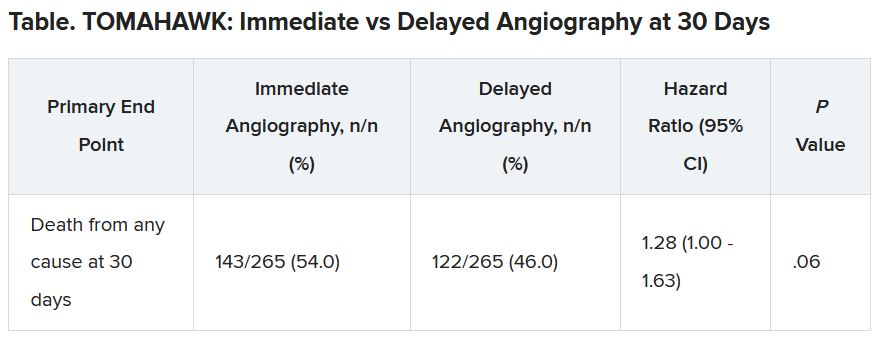
The secondary end point of death from any cause or severe neurologic deficit at 30 days “was actually nominally significant in favor of the delayed group,” he said. “So, this is not corrected for multiple testing, it’s just a hypothesis that’s in the room, but it’s certainly worthy of discussion that the immediate strategy might actually cause harm.”
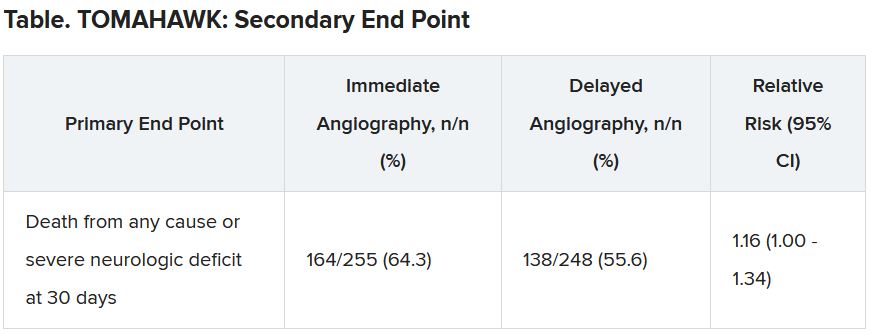
There was no difference between the groups in peak release of myocardial enzymes, or any other safety end points, including bleeding, stroke, or renal failure, Dr. Desch said.
Further analyses showed no large differences between subgroups, including age, diabetes, first monitored rhythm, confirmed MI as the trigger of the arrest, sex, and the time from cardiac arrest to the return of spontaneous circulation, he noted.
Opportunity to minimize harm
Discussant for the results during the presentation was Susanna Price, MBBS, PhD, Royal Brompton Hospital, London.
Dr. Price concluded: “What this means for me, is it gives me information that’s useful regarding the opportunity to minimize harm, which is a lot of what critical care is about, so we don’t necessarily now have to move these patients very acutely when they’ve just come in through the ED [emergency department]. It has implications for resource utilization, but also implications for mobilizing patients around the hospital during COVID-19.”
It’s also important to note that coronary angiography was still carried out in certain patients, “so we still have to have that dialogue with our interventional cardiologists for certain patients who may need to go to the cath lab, and what it should now allow us to do is give appropriate focus to how to manage these patients when they come in to the ED or to our ICUs [intensive care units],” she said.
Dr. Price added, though, that perhaps “the most important slide” in the presentation was that showing 90% of these patients had a witnessed cardiac arrest, “and yet a third of these patients, 168 of them, had no bystander CPR at all.”
She pointed to the “chain of survival” after cardiac arrest, of which Charles D. Deakin, MD, University Hospital Southampton (England), wrote that “not all links are equal.”
“Early recognition and calling for help, early CPR, early defibrillation where appropriate are very, very important, and we need to be addressing all of these, as well as what happens in the cath lab and after admission,” Dr. Price said.
This research was funded by the German Center for Cardiovascular Research. Dr. Desch and Dr. Price reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
A protocol of immediate angiography provided no mortality benefit over a strategy or delayed or more selective angiography among patients resuscitated from out-of-hospital cardiac arrest and without ST-segment elevation, new randomized results show.
“Among patients with resuscitated out-of-hospital cardiac arrest of possible cardiac origin, with shockable and nonshockable arrest rhythm and no ST-elevation, a strategy of immediate, unselected coronary angiography was not found to be beneficial over a delayed and selective approach with regard to the 30-day risk of all-cause death,” concluded principal investigator Steffen Desch, MD, University of Leipzig (Germany) Heart Center.
The results support previous results of the Coronary Angiography after Cardiac Arrest (COACT) trial, in patients with shockable rhythms, which also showed no differences in clinical outcomes between immediate and delayed coronary angiography at both 90 days and 1 year, he noted.
“What the clinicians wanted to know is, is it really necessary to get up at 3 a.m. in the morning to perform a coronary angiography on these patients, and that’s certainly out,” Dr. Desch said in an interview. “So, there’s really no room for this strategy anymore. You can take your time and wait a day or 2.”
These findings, from the TOMAHAWK trial, were presented Aug. 29 at the annual congress of the European Society of Cardiology and simultaneously published online in the New England Journal of Medicine.
Larger group without ST-segment elevation
Prognosis after out-of-hospital cardiac arrest is extremely poor, with an overall survival rate of less than 10%, Dr. Desch noted. “Actually, only 20% make it to the hospital; the vast majority of these patients die out in the field, so there’s really a great need in improving treatment.”
Acute coronary syndrome accounts for up to 60% of out-of-hospital arrests in which a cardiac cause has been identified, the authors wrote in their report. ST-segment elevation on postresuscitation electrocardiography “has good positive predictive value” for acute coronary lesions triggering the arrest, but in the far larger subgroup of patients without ST-segment elevation, “the spectrum of underlying causes is considerably broader and includes both cardiac and noncardiac causes.”
In patients with myocardial infarction, early revascularization would prevent negative consequences of myocardial injury, but unselected early coronary angiography would put patients not having an MI at unnecessary risk for procedural complications or delay in the diagnosis of the actual cause of their arrest, they noted.
In this trial, the researchers randomly assigned 554 patients from 31 sites in Germany and Denmark who were successfully resuscitated after cardiac arrest of possible cardiac origin to immediate transfer for coronary angiography or to initial intensive care assessment with delayed or selective angiography after a minimum delay of at least 1 day.
In the end, the average delay in this arm was 2 days, Dr. Desch noted. If the clinical course indicated that a coronary cause was unlikely, angiography might not be performed at all in this group.
No patient had ST-segment elevation on postresuscitation electrocardiography. The primary endpoint was death from any cause at 30 days; secondary end points were death from any cause or severe neurologic deficit at 30 days.
Results showed that 95% of patients in the immediate angiography group actually underwent the procedure, compared with 62% of those in the delayed group, a finding that was “logical” given the study design, he said.
At 30 days, 54% of patients in the immediate angiography group and 46% in the delayed group had died, a nonsignificant difference (P = .06). Because the researchers had performed an interim analysis, Dr. Desch explained, the final P value for significance in this trial was not .05, but rather .034, to account for multiple comparisons.

The secondary end point of death from any cause or severe neurologic deficit at 30 days “was actually nominally significant in favor of the delayed group,” he said. “So, this is not corrected for multiple testing, it’s just a hypothesis that’s in the room, but it’s certainly worthy of discussion that the immediate strategy might actually cause harm.”

There was no difference between the groups in peak release of myocardial enzymes, or any other safety end points, including bleeding, stroke, or renal failure, Dr. Desch said.
Further analyses showed no large differences between subgroups, including age, diabetes, first monitored rhythm, confirmed MI as the trigger of the arrest, sex, and the time from cardiac arrest to the return of spontaneous circulation, he noted.
Opportunity to minimize harm
Discussant for the results during the presentation was Susanna Price, MBBS, PhD, Royal Brompton Hospital, London.
Dr. Price concluded: “What this means for me, is it gives me information that’s useful regarding the opportunity to minimize harm, which is a lot of what critical care is about, so we don’t necessarily now have to move these patients very acutely when they’ve just come in through the ED [emergency department]. It has implications for resource utilization, but also implications for mobilizing patients around the hospital during COVID-19.”
It’s also important to note that coronary angiography was still carried out in certain patients, “so we still have to have that dialogue with our interventional cardiologists for certain patients who may need to go to the cath lab, and what it should now allow us to do is give appropriate focus to how to manage these patients when they come in to the ED or to our ICUs [intensive care units],” she said.
Dr. Price added, though, that perhaps “the most important slide” in the presentation was that showing 90% of these patients had a witnessed cardiac arrest, “and yet a third of these patients, 168 of them, had no bystander CPR at all.”
She pointed to the “chain of survival” after cardiac arrest, of which Charles D. Deakin, MD, University Hospital Southampton (England), wrote that “not all links are equal.”
“Early recognition and calling for help, early CPR, early defibrillation where appropriate are very, very important, and we need to be addressing all of these, as well as what happens in the cath lab and after admission,” Dr. Price said.
This research was funded by the German Center for Cardiovascular Research. Dr. Desch and Dr. Price reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
A protocol of immediate angiography provided no mortality benefit over a strategy or delayed or more selective angiography among patients resuscitated from out-of-hospital cardiac arrest and without ST-segment elevation, new randomized results show.
“Among patients with resuscitated out-of-hospital cardiac arrest of possible cardiac origin, with shockable and nonshockable arrest rhythm and no ST-elevation, a strategy of immediate, unselected coronary angiography was not found to be beneficial over a delayed and selective approach with regard to the 30-day risk of all-cause death,” concluded principal investigator Steffen Desch, MD, University of Leipzig (Germany) Heart Center.
The results support previous results of the Coronary Angiography after Cardiac Arrest (COACT) trial, in patients with shockable rhythms, which also showed no differences in clinical outcomes between immediate and delayed coronary angiography at both 90 days and 1 year, he noted.
“What the clinicians wanted to know is, is it really necessary to get up at 3 a.m. in the morning to perform a coronary angiography on these patients, and that’s certainly out,” Dr. Desch said in an interview. “So, there’s really no room for this strategy anymore. You can take your time and wait a day or 2.”
These findings, from the TOMAHAWK trial, were presented Aug. 29 at the annual congress of the European Society of Cardiology and simultaneously published online in the New England Journal of Medicine.
Larger group without ST-segment elevation
Prognosis after out-of-hospital cardiac arrest is extremely poor, with an overall survival rate of less than 10%, Dr. Desch noted. “Actually, only 20% make it to the hospital; the vast majority of these patients die out in the field, so there’s really a great need in improving treatment.”
Acute coronary syndrome accounts for up to 60% of out-of-hospital arrests in which a cardiac cause has been identified, the authors wrote in their report. ST-segment elevation on postresuscitation electrocardiography “has good positive predictive value” for acute coronary lesions triggering the arrest, but in the far larger subgroup of patients without ST-segment elevation, “the spectrum of underlying causes is considerably broader and includes both cardiac and noncardiac causes.”
In patients with myocardial infarction, early revascularization would prevent negative consequences of myocardial injury, but unselected early coronary angiography would put patients not having an MI at unnecessary risk for procedural complications or delay in the diagnosis of the actual cause of their arrest, they noted.
In this trial, the researchers randomly assigned 554 patients from 31 sites in Germany and Denmark who were successfully resuscitated after cardiac arrest of possible cardiac origin to immediate transfer for coronary angiography or to initial intensive care assessment with delayed or selective angiography after a minimum delay of at least 1 day.
In the end, the average delay in this arm was 2 days, Dr. Desch noted. If the clinical course indicated that a coronary cause was unlikely, angiography might not be performed at all in this group.
No patient had ST-segment elevation on postresuscitation electrocardiography. The primary endpoint was death from any cause at 30 days; secondary end points were death from any cause or severe neurologic deficit at 30 days.
Results showed that 95% of patients in the immediate angiography group actually underwent the procedure, compared with 62% of those in the delayed group, a finding that was “logical” given the study design, he said.
At 30 days, 54% of patients in the immediate angiography group and 46% in the delayed group had died, a nonsignificant difference (P = .06). Because the researchers had performed an interim analysis, Dr. Desch explained, the final P value for significance in this trial was not .05, but rather .034, to account for multiple comparisons.

The secondary end point of death from any cause or severe neurologic deficit at 30 days “was actually nominally significant in favor of the delayed group,” he said. “So, this is not corrected for multiple testing, it’s just a hypothesis that’s in the room, but it’s certainly worthy of discussion that the immediate strategy might actually cause harm.”

There was no difference between the groups in peak release of myocardial enzymes, or any other safety end points, including bleeding, stroke, or renal failure, Dr. Desch said.
Further analyses showed no large differences between subgroups, including age, diabetes, first monitored rhythm, confirmed MI as the trigger of the arrest, sex, and the time from cardiac arrest to the return of spontaneous circulation, he noted.
Opportunity to minimize harm
Discussant for the results during the presentation was Susanna Price, MBBS, PhD, Royal Brompton Hospital, London.
Dr. Price concluded: “What this means for me, is it gives me information that’s useful regarding the opportunity to minimize harm, which is a lot of what critical care is about, so we don’t necessarily now have to move these patients very acutely when they’ve just come in through the ED [emergency department]. It has implications for resource utilization, but also implications for mobilizing patients around the hospital during COVID-19.”
It’s also important to note that coronary angiography was still carried out in certain patients, “so we still have to have that dialogue with our interventional cardiologists for certain patients who may need to go to the cath lab, and what it should now allow us to do is give appropriate focus to how to manage these patients when they come in to the ED or to our ICUs [intensive care units],” she said.
Dr. Price added, though, that perhaps “the most important slide” in the presentation was that showing 90% of these patients had a witnessed cardiac arrest, “and yet a third of these patients, 168 of them, had no bystander CPR at all.”
She pointed to the “chain of survival” after cardiac arrest, of which Charles D. Deakin, MD, University Hospital Southampton (England), wrote that “not all links are equal.”
“Early recognition and calling for help, early CPR, early defibrillation where appropriate are very, very important, and we need to be addressing all of these, as well as what happens in the cath lab and after admission,” Dr. Price said.
This research was funded by the German Center for Cardiovascular Research. Dr. Desch and Dr. Price reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
Human brain patterns may help build a better AI system
new research suggests. “This work opens new opportunities to discover how the network organization of the brain optimizes cognitive capacity,” wrote researchers from The Neuro (Montreal Neurological Institute–Hospital) and the Quebec Artificial Intelligence Institute.
Senior investigator Bratislav Misic, PhD, said the research has potential clinical application for studying diseases of the brain, which is something his team is actively working on. “For example, using MRI techniques, we can measure different patterns of atrophy in neurodegenerative diseases such as Alzheimer’s disease,” he said.
“We can use these disease patterns from real patients to artificially lesion these connectomes and to ask how a particular disease causes a particular pattern of symptoms and cognitive deficits,” he added.
The findings were published online in Nature Machine Intelligence.
Unique approach
Using brain imaging data, the investigators reconstructed a human brain connectivity pattern and applied it to an artificial neural network. After training, the artificial neural network successfully performed a working memory task more flexibly and efficiently than other “benchmark” AI systems.
The researchers noted that their approach is unique because previous work on brain connectivity, also known as connectomics, has focused on describing brain organization without regard to how it actually functions.
Traditional artificial neural network have arbitrary structures that do not reflect how real brain networks are organized. Integrating brain connectomics into the construction of artificial neural network can reveal how the wiring of the brain supports specific cognitive skills, the investigators wrote.
“Up until now, if you look at how neural networks are constructed, the architectures that are used are very ad hoc and very problem specific,” Dr. Misic said. “But the connectomics revolution that’s happened in neuroscience over the past 20 years or so has given us the ability to really measure and trace out connection patterns in a variety of organisms, including the human brain.”
He noted that the researchers took wiring patterns of the real human brain and implemented it as an artificial neural network. They then “trained that network to perform a very simple cognitive task, and when you compare it to other benchmark architectures, it actually does better.”
This shows that there is “something fundamentally different about how the human brain is wired up and that the design principles that we can see in the human brain could be used to potentially build better artificial networks,” Dr. Misic concluded.
Funding for the research was provided by the Canada First Research Excellence Fund, awarded to McGill University for the Healthy Brains, Healthy Lives initiative, and by the Natural Sciences and Engineering Research Council of Canada, Fonds de Recherche du Quebec – Santé, the Canadian Institute for Advanced Research, Canada Research Chairs, Fonds de Recherche du Quebec – Nature et Technologies, and the Centre UNIQUE (Union of Neuroscience and Artificial Intelligence). The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. “This work opens new opportunities to discover how the network organization of the brain optimizes cognitive capacity,” wrote researchers from The Neuro (Montreal Neurological Institute–Hospital) and the Quebec Artificial Intelligence Institute.
Senior investigator Bratislav Misic, PhD, said the research has potential clinical application for studying diseases of the brain, which is something his team is actively working on. “For example, using MRI techniques, we can measure different patterns of atrophy in neurodegenerative diseases such as Alzheimer’s disease,” he said.
“We can use these disease patterns from real patients to artificially lesion these connectomes and to ask how a particular disease causes a particular pattern of symptoms and cognitive deficits,” he added.
The findings were published online in Nature Machine Intelligence.
Unique approach
Using brain imaging data, the investigators reconstructed a human brain connectivity pattern and applied it to an artificial neural network. After training, the artificial neural network successfully performed a working memory task more flexibly and efficiently than other “benchmark” AI systems.
The researchers noted that their approach is unique because previous work on brain connectivity, also known as connectomics, has focused on describing brain organization without regard to how it actually functions.
Traditional artificial neural network have arbitrary structures that do not reflect how real brain networks are organized. Integrating brain connectomics into the construction of artificial neural network can reveal how the wiring of the brain supports specific cognitive skills, the investigators wrote.
“Up until now, if you look at how neural networks are constructed, the architectures that are used are very ad hoc and very problem specific,” Dr. Misic said. “But the connectomics revolution that’s happened in neuroscience over the past 20 years or so has given us the ability to really measure and trace out connection patterns in a variety of organisms, including the human brain.”
He noted that the researchers took wiring patterns of the real human brain and implemented it as an artificial neural network. They then “trained that network to perform a very simple cognitive task, and when you compare it to other benchmark architectures, it actually does better.”
This shows that there is “something fundamentally different about how the human brain is wired up and that the design principles that we can see in the human brain could be used to potentially build better artificial networks,” Dr. Misic concluded.
Funding for the research was provided by the Canada First Research Excellence Fund, awarded to McGill University for the Healthy Brains, Healthy Lives initiative, and by the Natural Sciences and Engineering Research Council of Canada, Fonds de Recherche du Quebec – Santé, the Canadian Institute for Advanced Research, Canada Research Chairs, Fonds de Recherche du Quebec – Nature et Technologies, and the Centre UNIQUE (Union of Neuroscience and Artificial Intelligence). The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests. “This work opens new opportunities to discover how the network organization of the brain optimizes cognitive capacity,” wrote researchers from The Neuro (Montreal Neurological Institute–Hospital) and the Quebec Artificial Intelligence Institute.
Senior investigator Bratislav Misic, PhD, said the research has potential clinical application for studying diseases of the brain, which is something his team is actively working on. “For example, using MRI techniques, we can measure different patterns of atrophy in neurodegenerative diseases such as Alzheimer’s disease,” he said.
“We can use these disease patterns from real patients to artificially lesion these connectomes and to ask how a particular disease causes a particular pattern of symptoms and cognitive deficits,” he added.
The findings were published online in Nature Machine Intelligence.
Unique approach
Using brain imaging data, the investigators reconstructed a human brain connectivity pattern and applied it to an artificial neural network. After training, the artificial neural network successfully performed a working memory task more flexibly and efficiently than other “benchmark” AI systems.
The researchers noted that their approach is unique because previous work on brain connectivity, also known as connectomics, has focused on describing brain organization without regard to how it actually functions.
Traditional artificial neural network have arbitrary structures that do not reflect how real brain networks are organized. Integrating brain connectomics into the construction of artificial neural network can reveal how the wiring of the brain supports specific cognitive skills, the investigators wrote.
“Up until now, if you look at how neural networks are constructed, the architectures that are used are very ad hoc and very problem specific,” Dr. Misic said. “But the connectomics revolution that’s happened in neuroscience over the past 20 years or so has given us the ability to really measure and trace out connection patterns in a variety of organisms, including the human brain.”
He noted that the researchers took wiring patterns of the real human brain and implemented it as an artificial neural network. They then “trained that network to perform a very simple cognitive task, and when you compare it to other benchmark architectures, it actually does better.”
This shows that there is “something fundamentally different about how the human brain is wired up and that the design principles that we can see in the human brain could be used to potentially build better artificial networks,” Dr. Misic concluded.
Funding for the research was provided by the Canada First Research Excellence Fund, awarded to McGill University for the Healthy Brains, Healthy Lives initiative, and by the Natural Sciences and Engineering Research Council of Canada, Fonds de Recherche du Quebec – Santé, the Canadian Institute for Advanced Research, Canada Research Chairs, Fonds de Recherche du Quebec – Nature et Technologies, and the Centre UNIQUE (Union of Neuroscience and Artificial Intelligence). The investigators have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE MACHINE INTELLIGENCE
NIH to study COVID vaccine booster in people with autoimmune disease
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
In the wake of the Centers for Disease Control and Prevention’s recommendation for a third COVID-19 mRNA vaccine dose for immunocompromised people and the Food and Drug Administration’s authorization of the third dose, the according to an announcement.
The investigators of the trial, called COVID‐19 Booster Vaccine in Autoimmune Disease Non‐Responders, also want to determine if pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine.
The trial will specifically look at the effects of mycophenolate mofetil (MMF) or mycophenolic acid (MPA), and methotrexate (MTX), or receipt of B cell–depletion therapy such as rituximab within the past 12 months on immune response to a booster dose in people with systemic lupus erythematosus, rheumatoid arthritis, multiple sclerosis, systemic sclerosis, or pemphigus. They have to have either no serologic response to their initial COVID-19 vaccine regimen or a suboptimal response, defined as a Roche Elecsys Anti-SARS-CoV-2 S (RBD) result greater than or equal to 50 U/mL.
The results of studies conducted in solid-organ transplant recipients who take immunosuppressants showed that an extra dose of vaccine could improve the immune response to the vaccine in many of the individuals, which suggests that the same approach might work in people with autoimmune disease who need treatment with immunosuppressive drugs. Improving the immune response of people with autoimmune disease to COVID-19 vaccines is important because higher rates of severe COVID-19 and death have been reported in this group of patients than in the general population, and it is unclear whether this is attributable to the autoimmune disease, the immunosuppressive medications taken to treat it, or both.
The open-label trial, conducted by the NIAID-funded Autoimmunity Centers of Excellence, aims to enroll 600 people aged 18 years and older with those conditions at 15-20 sites in the United States.
Because medications commonly taken by people with these conditions have been associated with poorer immune responses to vaccines, the trial will randomize the following two cohorts to stop or continue taking their immunosuppressive medication(s) or stop them before and after the booster according to protocol:
- Cohort 1 includes people who are taking MMF or MPA, without additional B cell–depleting medications or MTX.
- Cohort 2 includes people who are taking MTX without additional B cell–depleting medications or MMF/MPA.
A third, nonrandomized cohort consists of people who have received B cell–depletion therapy within the past 12 months regardless of whether they are also taking MMF/MPA or MTX.
Besides the cohort-specific exclusions, other rheumatic disease medications, including biologics, are allowed in the groups.
The primary outcome of the trial is the proportion of participants who have a protective antibody response at week 4. Secondary outcomes will examine various antibody responses at intervals, changes in disease activity across autoimmune diseases, adverse events, and SARS-CoV-2 infections out to 48 weeks.
Study participants will be followed for a total of 13 months. Preliminary results are expected in November 2021, according to the National Institutes of Health.
The trial is being led by Judith James, MD, PhD; Meggan Mackay, MD, MS; Dinesh Khanna, MBBS, MSc; and Amit Bar-Or, MD.
Young Black and White athletes differ in how they recover from concussions
, according to a new study on racial differences in concussion recovery.
“The findings from this study provide novel evidence that the recovery experience following sport-related concussion likely differs between Black and White athletes, and understanding these differences may serve to provide better and more personalized intervention and management strategies,” wrote lead author Aaron M. Yengo-Kahn, MD, of Vanderbilt University Medical Center in Nashville, Tenn. The study was published in the Journal of Neurosurgery: Pediatrics.
To assess how postconcussion experiences and recovery time differ among young White and Black athletes, the researchers launched a retrospective cohort study of youths between the ages of 12 and 23 from the middle Tennessee, northern Alabama, and southern Kentucky regions who had been treated for sport-related concussion. Using data from the Vanderbilt Sports Concussion Center’s outcome registry, they examined the records of 247 student-athletes, 211 of whom were White and 36 of whom were Black.
The majority of the athletes were male – 58% of the White group and 78% of the Black group – and their average age across groups was roughly 16 years. Thirty-three percent of the Black athletes were on public insurance, compared with just 6% of the White athletes, and 41% of the Black athletes lived in low–median income areas while 55% of the White athletes lived in areas with a high median income. Approximately 90% of each group played contact sports.
The median time to symptom resolution was 21 days (interquartile range, 10.5-61.0) for White athletes but just 12.3 days (IQR, 6.8-28.0) for Black athletes. Multivariable regression confirmed that Black athletes reached asymptomatic status sooner than White athletes (hazard ratio, 1.497; 95% confidence interval, 1.014-2.209; P = .042). “The observed shorter symptom resolution among the Black athletes may be explained by a complex interplay among race, concussion knowledge, attitudes toward sport-related concussion, reporting behavior, and sociodemographic disparities,” the authors noted.
The median time until returning to school post injury was 2 school days (IQR, 0-5) for White athletes and 0 school days (IQR, 0-2) for Black athletes. After multivariable analysis, being Black was indeed associated with returning to school sooner, compared with being White (HR, 1.522; 95% CI, 1.02-2,27; P = .040). Being Black was also associated with being less likely to a report a change in daily activity post concussion (odds ratio, 0.368; 95% CI, 0.136-0.996; P = .049).
Adding race to research
To make headway toward understanding race’s impact on concussion research, the authors proposed three immediate steps: Work directly with schools instead of clinics or emergency departments, match the diversity of study cohorts with the racial makeup of the surrounding community, and consider race as a covariate during study design.
“In our work with concussions, there is very little reported on race or racism or how racism affects how patients are navigating these spaces,” said coauthor Jessica Wallace, PhD, of the department of health science at the University of Alabama in Tuscaloosa, Ala., in an interview. “But we have so many athletes at the youth level, adolescent level, even the collegiate level; it’s such a diverse array of patients. We need to have data representative of all of our groups so that we know where we need to be intentional about reducing disparities and closing gaps.”
Dr. Wallace, who recently authored a study on the underreporting of concussions among Black and White high school athletes, emphasized the need for concussion research to be a true collaboration across disciplines.
“I approach this work from this public health and athletic training lens, whereas a lot of my collaborators are in neurosurgery and neurology,” she said. “Moving forward, we as a scientific clinical community have to do interdisciplinary work and be very intentional about how we go about closing these gaps. We have to recognize that there are differences in knowledge and in care, and they’re unacceptable, and we have to work collaboratively in providing resources to communities equitably to decrease them.”
The authors acknowledged their study’s limitations, including the retrospective nature of the study, using zip codes to determine median household income, and an unbalanced number of White and Black athletes. They did add, however, that the ratio of participants “generally aligns with census data in the surrounding metropolitan and county areas.” That said, they also surmised that the scarcity of Black athletes could indicate a deeper disparity in health care system usage and asked future researchers to “consider enrolling athletes directly from schools rather than from within the concussion clinic only.”
Dr. Yengo-Kahn disclosed holding a compensated position on the scientific advisory board of BlinkTBI, but the authors noted that the company had no role in the study and its products were not used. No other conflicts of interest were reported.
, according to a new study on racial differences in concussion recovery.
“The findings from this study provide novel evidence that the recovery experience following sport-related concussion likely differs between Black and White athletes, and understanding these differences may serve to provide better and more personalized intervention and management strategies,” wrote lead author Aaron M. Yengo-Kahn, MD, of Vanderbilt University Medical Center in Nashville, Tenn. The study was published in the Journal of Neurosurgery: Pediatrics.
To assess how postconcussion experiences and recovery time differ among young White and Black athletes, the researchers launched a retrospective cohort study of youths between the ages of 12 and 23 from the middle Tennessee, northern Alabama, and southern Kentucky regions who had been treated for sport-related concussion. Using data from the Vanderbilt Sports Concussion Center’s outcome registry, they examined the records of 247 student-athletes, 211 of whom were White and 36 of whom were Black.
The majority of the athletes were male – 58% of the White group and 78% of the Black group – and their average age across groups was roughly 16 years. Thirty-three percent of the Black athletes were on public insurance, compared with just 6% of the White athletes, and 41% of the Black athletes lived in low–median income areas while 55% of the White athletes lived in areas with a high median income. Approximately 90% of each group played contact sports.
The median time to symptom resolution was 21 days (interquartile range, 10.5-61.0) for White athletes but just 12.3 days (IQR, 6.8-28.0) for Black athletes. Multivariable regression confirmed that Black athletes reached asymptomatic status sooner than White athletes (hazard ratio, 1.497; 95% confidence interval, 1.014-2.209; P = .042). “The observed shorter symptom resolution among the Black athletes may be explained by a complex interplay among race, concussion knowledge, attitudes toward sport-related concussion, reporting behavior, and sociodemographic disparities,” the authors noted.
The median time until returning to school post injury was 2 school days (IQR, 0-5) for White athletes and 0 school days (IQR, 0-2) for Black athletes. After multivariable analysis, being Black was indeed associated with returning to school sooner, compared with being White (HR, 1.522; 95% CI, 1.02-2,27; P = .040). Being Black was also associated with being less likely to a report a change in daily activity post concussion (odds ratio, 0.368; 95% CI, 0.136-0.996; P = .049).
Adding race to research
To make headway toward understanding race’s impact on concussion research, the authors proposed three immediate steps: Work directly with schools instead of clinics or emergency departments, match the diversity of study cohorts with the racial makeup of the surrounding community, and consider race as a covariate during study design.
“In our work with concussions, there is very little reported on race or racism or how racism affects how patients are navigating these spaces,” said coauthor Jessica Wallace, PhD, of the department of health science at the University of Alabama in Tuscaloosa, Ala., in an interview. “But we have so many athletes at the youth level, adolescent level, even the collegiate level; it’s such a diverse array of patients. We need to have data representative of all of our groups so that we know where we need to be intentional about reducing disparities and closing gaps.”
Dr. Wallace, who recently authored a study on the underreporting of concussions among Black and White high school athletes, emphasized the need for concussion research to be a true collaboration across disciplines.
“I approach this work from this public health and athletic training lens, whereas a lot of my collaborators are in neurosurgery and neurology,” she said. “Moving forward, we as a scientific clinical community have to do interdisciplinary work and be very intentional about how we go about closing these gaps. We have to recognize that there are differences in knowledge and in care, and they’re unacceptable, and we have to work collaboratively in providing resources to communities equitably to decrease them.”
The authors acknowledged their study’s limitations, including the retrospective nature of the study, using zip codes to determine median household income, and an unbalanced number of White and Black athletes. They did add, however, that the ratio of participants “generally aligns with census data in the surrounding metropolitan and county areas.” That said, they also surmised that the scarcity of Black athletes could indicate a deeper disparity in health care system usage and asked future researchers to “consider enrolling athletes directly from schools rather than from within the concussion clinic only.”
Dr. Yengo-Kahn disclosed holding a compensated position on the scientific advisory board of BlinkTBI, but the authors noted that the company had no role in the study and its products were not used. No other conflicts of interest were reported.
, according to a new study on racial differences in concussion recovery.
“The findings from this study provide novel evidence that the recovery experience following sport-related concussion likely differs between Black and White athletes, and understanding these differences may serve to provide better and more personalized intervention and management strategies,” wrote lead author Aaron M. Yengo-Kahn, MD, of Vanderbilt University Medical Center in Nashville, Tenn. The study was published in the Journal of Neurosurgery: Pediatrics.
To assess how postconcussion experiences and recovery time differ among young White and Black athletes, the researchers launched a retrospective cohort study of youths between the ages of 12 and 23 from the middle Tennessee, northern Alabama, and southern Kentucky regions who had been treated for sport-related concussion. Using data from the Vanderbilt Sports Concussion Center’s outcome registry, they examined the records of 247 student-athletes, 211 of whom were White and 36 of whom were Black.
The majority of the athletes were male – 58% of the White group and 78% of the Black group – and their average age across groups was roughly 16 years. Thirty-three percent of the Black athletes were on public insurance, compared with just 6% of the White athletes, and 41% of the Black athletes lived in low–median income areas while 55% of the White athletes lived in areas with a high median income. Approximately 90% of each group played contact sports.
The median time to symptom resolution was 21 days (interquartile range, 10.5-61.0) for White athletes but just 12.3 days (IQR, 6.8-28.0) for Black athletes. Multivariable regression confirmed that Black athletes reached asymptomatic status sooner than White athletes (hazard ratio, 1.497; 95% confidence interval, 1.014-2.209; P = .042). “The observed shorter symptom resolution among the Black athletes may be explained by a complex interplay among race, concussion knowledge, attitudes toward sport-related concussion, reporting behavior, and sociodemographic disparities,” the authors noted.
The median time until returning to school post injury was 2 school days (IQR, 0-5) for White athletes and 0 school days (IQR, 0-2) for Black athletes. After multivariable analysis, being Black was indeed associated with returning to school sooner, compared with being White (HR, 1.522; 95% CI, 1.02-2,27; P = .040). Being Black was also associated with being less likely to a report a change in daily activity post concussion (odds ratio, 0.368; 95% CI, 0.136-0.996; P = .049).
Adding race to research
To make headway toward understanding race’s impact on concussion research, the authors proposed three immediate steps: Work directly with schools instead of clinics or emergency departments, match the diversity of study cohorts with the racial makeup of the surrounding community, and consider race as a covariate during study design.
“In our work with concussions, there is very little reported on race or racism or how racism affects how patients are navigating these spaces,” said coauthor Jessica Wallace, PhD, of the department of health science at the University of Alabama in Tuscaloosa, Ala., in an interview. “But we have so many athletes at the youth level, adolescent level, even the collegiate level; it’s such a diverse array of patients. We need to have data representative of all of our groups so that we know where we need to be intentional about reducing disparities and closing gaps.”
Dr. Wallace, who recently authored a study on the underreporting of concussions among Black and White high school athletes, emphasized the need for concussion research to be a true collaboration across disciplines.
“I approach this work from this public health and athletic training lens, whereas a lot of my collaborators are in neurosurgery and neurology,” she said. “Moving forward, we as a scientific clinical community have to do interdisciplinary work and be very intentional about how we go about closing these gaps. We have to recognize that there are differences in knowledge and in care, and they’re unacceptable, and we have to work collaboratively in providing resources to communities equitably to decrease them.”
The authors acknowledged their study’s limitations, including the retrospective nature of the study, using zip codes to determine median household income, and an unbalanced number of White and Black athletes. They did add, however, that the ratio of participants “generally aligns with census data in the surrounding metropolitan and county areas.” That said, they also surmised that the scarcity of Black athletes could indicate a deeper disparity in health care system usage and asked future researchers to “consider enrolling athletes directly from schools rather than from within the concussion clinic only.”
Dr. Yengo-Kahn disclosed holding a compensated position on the scientific advisory board of BlinkTBI, but the authors noted that the company had no role in the study and its products were not used. No other conflicts of interest were reported.
FROM THE JOURNAL OF NEUROSURGERY: PEDIATRICS
Atogepant reduces migraine days: ADVANCE trial results published
full results from a phase 3 trial suggest.
AbbVie, the company developing the oral therapy, announced topline results of the ADVANCE trial of atogepant last year. Safety results were presented in April at the 2021 annual meeting of the American Academy of Neurology.
The full results were published online Aug. 19 in the New England Journal of Medicine ahead of the upcoming target action date of the U.S. Food and Drug Administration.
The multicenter study included nearly 900 patients who were randomly assigned to receive either placebo or one of three doses of atogepant for 12 weeks. The mean number of monthly migraine days decreased by about 4 for all three doses of the active treatment, compared with a reduction of 2.5 days with placebo.
“Overall, this study showed us that atogepant was safe and surprisingly seems to be pretty effective regardless of the dose,” said lead author Jessica Ailani, MD, director of MedStar Georgetown Headache Center and associate professor of neurology at Georgetown University, Washington.
All doses effective
The study included 873 patients with episodic migraine with or without aura. Patients who were not assigned to the placebo control group received either 10 mg, 30 mg, or 60 mg of atogepant once daily.
After a 4-week screening period, all patients received treatment for 12 weeks and then entered a 4-week safety follow-up period. In total, the participants completed eight scheduled clinical visits.
The mean reduction from baseline in the mean number of migraine days per month was 3.7 with the 10-mg dose of atogepant, 3.9 with the 30-mg dose, 4.2 with the 60-mg dose, and 2.5 with placebo. The differences between each active dose and placebo was statistically significant (P < .001).
Treatment with the CGRP inhibitor was also associated with a reduction in the mean number of headache days per month. The mean reduction from baseline was 3.9 days for the 10-mg dose, 4.0 days for the 30-mg dose, 4.2 days for the 60-mg dose, and 2.5 days for placebo (P < .001 for all comparisons with placebo).
In addition, for 55.6% of the 10-mg group, 58.7% of the 30-mg group, 60.8% of the 60-mg group, and 29.0% of the control group, there was a reduction of at least 50% in the 3-month average number of migraine days per month (P < .001 for each vs. placebo).
The most commonly reported adverse events (AEs) among patients who received atogepant were constipation (6.9%-7.7% across doses), nausea (4.4%-6.1%), and upper respiratory tract infection (1.4%-3.9%). Frequency of AEs did not differ between the active-treatment groups and the control group, and no relationships between AEs and atogepant dose were observed.
Multidose flexibility
“Side effects were pretty even across the board,” said Dr. Ailani. She noted that the reported AEs were expected because of atogepant’s mechanism of action. In addition, the rate of discontinuation in the study was low.
The proportion of participants who experienced a reduction in monthly migraine days of at least 50% grew as time passed. “By the end of this study, your chance of having a greater than 50% response is about 75%,” Dr. Ailani said.
“Imagine telling your patient, ‘You stick on this drug for 3 months, and I can almost guarantee you that you’re going to get better,’” she added.
Although the treatment has no drug-drug contraindications, drug-drug interactions may occur. “The availability of various doses would allow clinicians to adjust treatment to avoid potential drug-drug interactions,” said Dr. Ailani. “That multidose flexibility is very important.”
An FDA decision on atogepant could be made in the coming months. “I’m hopeful, as a clinician, that it is positive news, because we really have waited a long time for something like this,” Dr. Ailani said.
“You can easily identify patients who would do well on this medication,” she added.
In a different study of atogepant among patients with chronic migraine, there were recruitment delays because of the pandemic. That study is now almost complete, Dr. Ailani reported.
“Well-conducted study”
Commenting on the findings, Kathleen B. Digre, MD, chief of the division of headache and neuro-ophthalmology at the University of Utah Health, Salt Lake City, expressed enthusiasm for the experimental drug. “I’m excited to see another treatment modality for migraine,” said Dr. Digre, who was not involved with the research. “It was a very well-conducted study,” she added.
The treatment arms were almost identical in regard to disease severity, and all the doses showed an effect. Although the difference in reduction of monthly migraine days in comparison with placebo was numerically small, “for people who have frequent migraine, it’s important,” Dr. Digre said.
The results for atogepant should be viewed in a larger context, however. “Even though it’s a treatment that works better than placebo for well-matched controls, it may not be a medication that everybody’s going to respond to,” she noted. “And we can’t generalize it for some of the most disabled people, which is for chronic migraine,” she said.
It is significant that the study was published in the New England Journal of Medicine, Dr. Digre noted. “Sometimes migraine is dismissed as not important and not affecting people’s lives,” she said. “That makes me very happy to see migraine being taken seriously by our major journals.”
In addition, she noted that the prospects for FDA approval of atogepant seem favorable. “I’m hopeful that they will approve it, because it’s got a low side-effect profile, plus it’s effective.”
Migraine-specific preventive therapy has emerged only in the past few years. “I’m so excited to see this surge of preventive medicine for migraine,” Dr. Digre said. “It’s so important, because we see so many people who are disabled by migraine,” she added.
The study was funded by Allergan before atogepant was acquired by AbbVie. Dr. Ailani has received honoraria from AbbVie for consulting, has received compensation from Allergan and AbbVie for participating in a speakers’ bureau, and has received clinical trial grants from Allergan. Dr. Digre has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
full results from a phase 3 trial suggest.
AbbVie, the company developing the oral therapy, announced topline results of the ADVANCE trial of atogepant last year. Safety results were presented in April at the 2021 annual meeting of the American Academy of Neurology.
The full results were published online Aug. 19 in the New England Journal of Medicine ahead of the upcoming target action date of the U.S. Food and Drug Administration.
The multicenter study included nearly 900 patients who were randomly assigned to receive either placebo or one of three doses of atogepant for 12 weeks. The mean number of monthly migraine days decreased by about 4 for all three doses of the active treatment, compared with a reduction of 2.5 days with placebo.
“Overall, this study showed us that atogepant was safe and surprisingly seems to be pretty effective regardless of the dose,” said lead author Jessica Ailani, MD, director of MedStar Georgetown Headache Center and associate professor of neurology at Georgetown University, Washington.
All doses effective
The study included 873 patients with episodic migraine with or without aura. Patients who were not assigned to the placebo control group received either 10 mg, 30 mg, or 60 mg of atogepant once daily.
After a 4-week screening period, all patients received treatment for 12 weeks and then entered a 4-week safety follow-up period. In total, the participants completed eight scheduled clinical visits.
The mean reduction from baseline in the mean number of migraine days per month was 3.7 with the 10-mg dose of atogepant, 3.9 with the 30-mg dose, 4.2 with the 60-mg dose, and 2.5 with placebo. The differences between each active dose and placebo was statistically significant (P < .001).
Treatment with the CGRP inhibitor was also associated with a reduction in the mean number of headache days per month. The mean reduction from baseline was 3.9 days for the 10-mg dose, 4.0 days for the 30-mg dose, 4.2 days for the 60-mg dose, and 2.5 days for placebo (P < .001 for all comparisons with placebo).
In addition, for 55.6% of the 10-mg group, 58.7% of the 30-mg group, 60.8% of the 60-mg group, and 29.0% of the control group, there was a reduction of at least 50% in the 3-month average number of migraine days per month (P < .001 for each vs. placebo).
The most commonly reported adverse events (AEs) among patients who received atogepant were constipation (6.9%-7.7% across doses), nausea (4.4%-6.1%), and upper respiratory tract infection (1.4%-3.9%). Frequency of AEs did not differ between the active-treatment groups and the control group, and no relationships between AEs and atogepant dose were observed.
Multidose flexibility
“Side effects were pretty even across the board,” said Dr. Ailani. She noted that the reported AEs were expected because of atogepant’s mechanism of action. In addition, the rate of discontinuation in the study was low.
The proportion of participants who experienced a reduction in monthly migraine days of at least 50% grew as time passed. “By the end of this study, your chance of having a greater than 50% response is about 75%,” Dr. Ailani said.
“Imagine telling your patient, ‘You stick on this drug for 3 months, and I can almost guarantee you that you’re going to get better,’” she added.
Although the treatment has no drug-drug contraindications, drug-drug interactions may occur. “The availability of various doses would allow clinicians to adjust treatment to avoid potential drug-drug interactions,” said Dr. Ailani. “That multidose flexibility is very important.”
An FDA decision on atogepant could be made in the coming months. “I’m hopeful, as a clinician, that it is positive news, because we really have waited a long time for something like this,” Dr. Ailani said.
“You can easily identify patients who would do well on this medication,” she added.
In a different study of atogepant among patients with chronic migraine, there were recruitment delays because of the pandemic. That study is now almost complete, Dr. Ailani reported.
“Well-conducted study”
Commenting on the findings, Kathleen B. Digre, MD, chief of the division of headache and neuro-ophthalmology at the University of Utah Health, Salt Lake City, expressed enthusiasm for the experimental drug. “I’m excited to see another treatment modality for migraine,” said Dr. Digre, who was not involved with the research. “It was a very well-conducted study,” she added.
The treatment arms were almost identical in regard to disease severity, and all the doses showed an effect. Although the difference in reduction of monthly migraine days in comparison with placebo was numerically small, “for people who have frequent migraine, it’s important,” Dr. Digre said.
The results for atogepant should be viewed in a larger context, however. “Even though it’s a treatment that works better than placebo for well-matched controls, it may not be a medication that everybody’s going to respond to,” she noted. “And we can’t generalize it for some of the most disabled people, which is for chronic migraine,” she said.
It is significant that the study was published in the New England Journal of Medicine, Dr. Digre noted. “Sometimes migraine is dismissed as not important and not affecting people’s lives,” she said. “That makes me very happy to see migraine being taken seriously by our major journals.”
In addition, she noted that the prospects for FDA approval of atogepant seem favorable. “I’m hopeful that they will approve it, because it’s got a low side-effect profile, plus it’s effective.”
Migraine-specific preventive therapy has emerged only in the past few years. “I’m so excited to see this surge of preventive medicine for migraine,” Dr. Digre said. “It’s so important, because we see so many people who are disabled by migraine,” she added.
The study was funded by Allergan before atogepant was acquired by AbbVie. Dr. Ailani has received honoraria from AbbVie for consulting, has received compensation from Allergan and AbbVie for participating in a speakers’ bureau, and has received clinical trial grants from Allergan. Dr. Digre has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
full results from a phase 3 trial suggest.
AbbVie, the company developing the oral therapy, announced topline results of the ADVANCE trial of atogepant last year. Safety results were presented in April at the 2021 annual meeting of the American Academy of Neurology.
The full results were published online Aug. 19 in the New England Journal of Medicine ahead of the upcoming target action date of the U.S. Food and Drug Administration.
The multicenter study included nearly 900 patients who were randomly assigned to receive either placebo or one of three doses of atogepant for 12 weeks. The mean number of monthly migraine days decreased by about 4 for all three doses of the active treatment, compared with a reduction of 2.5 days with placebo.
“Overall, this study showed us that atogepant was safe and surprisingly seems to be pretty effective regardless of the dose,” said lead author Jessica Ailani, MD, director of MedStar Georgetown Headache Center and associate professor of neurology at Georgetown University, Washington.
All doses effective
The study included 873 patients with episodic migraine with or without aura. Patients who were not assigned to the placebo control group received either 10 mg, 30 mg, or 60 mg of atogepant once daily.
After a 4-week screening period, all patients received treatment for 12 weeks and then entered a 4-week safety follow-up period. In total, the participants completed eight scheduled clinical visits.
The mean reduction from baseline in the mean number of migraine days per month was 3.7 with the 10-mg dose of atogepant, 3.9 with the 30-mg dose, 4.2 with the 60-mg dose, and 2.5 with placebo. The differences between each active dose and placebo was statistically significant (P < .001).
Treatment with the CGRP inhibitor was also associated with a reduction in the mean number of headache days per month. The mean reduction from baseline was 3.9 days for the 10-mg dose, 4.0 days for the 30-mg dose, 4.2 days for the 60-mg dose, and 2.5 days for placebo (P < .001 for all comparisons with placebo).
In addition, for 55.6% of the 10-mg group, 58.7% of the 30-mg group, 60.8% of the 60-mg group, and 29.0% of the control group, there was a reduction of at least 50% in the 3-month average number of migraine days per month (P < .001 for each vs. placebo).
The most commonly reported adverse events (AEs) among patients who received atogepant were constipation (6.9%-7.7% across doses), nausea (4.4%-6.1%), and upper respiratory tract infection (1.4%-3.9%). Frequency of AEs did not differ between the active-treatment groups and the control group, and no relationships between AEs and atogepant dose were observed.
Multidose flexibility
“Side effects were pretty even across the board,” said Dr. Ailani. She noted that the reported AEs were expected because of atogepant’s mechanism of action. In addition, the rate of discontinuation in the study was low.
The proportion of participants who experienced a reduction in monthly migraine days of at least 50% grew as time passed. “By the end of this study, your chance of having a greater than 50% response is about 75%,” Dr. Ailani said.
“Imagine telling your patient, ‘You stick on this drug for 3 months, and I can almost guarantee you that you’re going to get better,’” she added.
Although the treatment has no drug-drug contraindications, drug-drug interactions may occur. “The availability of various doses would allow clinicians to adjust treatment to avoid potential drug-drug interactions,” said Dr. Ailani. “That multidose flexibility is very important.”
An FDA decision on atogepant could be made in the coming months. “I’m hopeful, as a clinician, that it is positive news, because we really have waited a long time for something like this,” Dr. Ailani said.
“You can easily identify patients who would do well on this medication,” she added.
In a different study of atogepant among patients with chronic migraine, there were recruitment delays because of the pandemic. That study is now almost complete, Dr. Ailani reported.
“Well-conducted study”
Commenting on the findings, Kathleen B. Digre, MD, chief of the division of headache and neuro-ophthalmology at the University of Utah Health, Salt Lake City, expressed enthusiasm for the experimental drug. “I’m excited to see another treatment modality for migraine,” said Dr. Digre, who was not involved with the research. “It was a very well-conducted study,” she added.
The treatment arms were almost identical in regard to disease severity, and all the doses showed an effect. Although the difference in reduction of monthly migraine days in comparison with placebo was numerically small, “for people who have frequent migraine, it’s important,” Dr. Digre said.
The results for atogepant should be viewed in a larger context, however. “Even though it’s a treatment that works better than placebo for well-matched controls, it may not be a medication that everybody’s going to respond to,” she noted. “And we can’t generalize it for some of the most disabled people, which is for chronic migraine,” she said.
It is significant that the study was published in the New England Journal of Medicine, Dr. Digre noted. “Sometimes migraine is dismissed as not important and not affecting people’s lives,” she said. “That makes me very happy to see migraine being taken seriously by our major journals.”
In addition, she noted that the prospects for FDA approval of atogepant seem favorable. “I’m hopeful that they will approve it, because it’s got a low side-effect profile, plus it’s effective.”
Migraine-specific preventive therapy has emerged only in the past few years. “I’m so excited to see this surge of preventive medicine for migraine,” Dr. Digre said. “It’s so important, because we see so many people who are disabled by migraine,” she added.
The study was funded by Allergan before atogepant was acquired by AbbVie. Dr. Ailani has received honoraria from AbbVie for consulting, has received compensation from Allergan and AbbVie for participating in a speakers’ bureau, and has received clinical trial grants from Allergan. Dr. Digre has reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Efficacy of gabapentin for treatment of alcohol use disorders
Background: Up to 30 million people in the United States meet criteria for alcohol use disorder. Gabapentin addresses symptoms of protracted withdrawal such as insomnia, irritability, difficulty with attention, dysphoria, and anxiety. It does that by acting on voltage-gated calcium channels and, in turn, influencing GABA and glutamate tone and activity.
Study design: Double-blind, placebo-controlled, randomized clinical trial.
Settings: Academic ambulatory setting at the Medical University of South Carolina.
Synopsis: A total of 96 community-recruited participants were randomly assigned to gabapentin and placebo arm then treated and followed for a total of 16 weeks. The gabapentin arm received gradual increments of gabapentin reaching up to 1,200 mg/day by day 5. The control group received placebo in blister packs. Individuals in the gabapentin arm, compared with those in the placebo arm, showed 18.6% (P = .02) more no heavy–drinking days, with a number needed to treat (NNT) of 5.4, and 13.8% (P = .04) more total abstinence days, with an NNT of 6.2. The prestudy high–alcohol withdrawal group in particular had significantly less relapse to heavy drinking (P = .02; NNT, 3.1) and more total abstinence (P = .03; NNT, 2.7) when treated with gabapentin.
A couple of study limitations were a significant noncompletion rate (30% in gabapentin arm and 39% in the placebo arm) and self-reported alcohol withdrawal symptoms prior to entry into the study.
Bottom line: Gabapentin helps in reducing drinking and maintaining alcohol abstinence in individuals with alcohol use disorder, especially those with high–alcohol withdrawal symptoms.
Citation: Anton RF et al. Efficacy of gabapentin for the treatment of alcohol use disorder in patients with alcohol withdrawal symptoms: A randomized clinical trial. JAMA Intern Med. 2020 Mar 9;180(5):728-36.
Dr. Gaddam is a hospitalist and assistant professor of medicine at UK HealthCare, Lexington, Ky.
Background: Up to 30 million people in the United States meet criteria for alcohol use disorder. Gabapentin addresses symptoms of protracted withdrawal such as insomnia, irritability, difficulty with attention, dysphoria, and anxiety. It does that by acting on voltage-gated calcium channels and, in turn, influencing GABA and glutamate tone and activity.
Study design: Double-blind, placebo-controlled, randomized clinical trial.
Settings: Academic ambulatory setting at the Medical University of South Carolina.
Synopsis: A total of 96 community-recruited participants were randomly assigned to gabapentin and placebo arm then treated and followed for a total of 16 weeks. The gabapentin arm received gradual increments of gabapentin reaching up to 1,200 mg/day by day 5. The control group received placebo in blister packs. Individuals in the gabapentin arm, compared with those in the placebo arm, showed 18.6% (P = .02) more no heavy–drinking days, with a number needed to treat (NNT) of 5.4, and 13.8% (P = .04) more total abstinence days, with an NNT of 6.2. The prestudy high–alcohol withdrawal group in particular had significantly less relapse to heavy drinking (P = .02; NNT, 3.1) and more total abstinence (P = .03; NNT, 2.7) when treated with gabapentin.
A couple of study limitations were a significant noncompletion rate (30% in gabapentin arm and 39% in the placebo arm) and self-reported alcohol withdrawal symptoms prior to entry into the study.
Bottom line: Gabapentin helps in reducing drinking and maintaining alcohol abstinence in individuals with alcohol use disorder, especially those with high–alcohol withdrawal symptoms.
Citation: Anton RF et al. Efficacy of gabapentin for the treatment of alcohol use disorder in patients with alcohol withdrawal symptoms: A randomized clinical trial. JAMA Intern Med. 2020 Mar 9;180(5):728-36.
Dr. Gaddam is a hospitalist and assistant professor of medicine at UK HealthCare, Lexington, Ky.
Background: Up to 30 million people in the United States meet criteria for alcohol use disorder. Gabapentin addresses symptoms of protracted withdrawal such as insomnia, irritability, difficulty with attention, dysphoria, and anxiety. It does that by acting on voltage-gated calcium channels and, in turn, influencing GABA and glutamate tone and activity.
Study design: Double-blind, placebo-controlled, randomized clinical trial.
Settings: Academic ambulatory setting at the Medical University of South Carolina.
Synopsis: A total of 96 community-recruited participants were randomly assigned to gabapentin and placebo arm then treated and followed for a total of 16 weeks. The gabapentin arm received gradual increments of gabapentin reaching up to 1,200 mg/day by day 5. The control group received placebo in blister packs. Individuals in the gabapentin arm, compared with those in the placebo arm, showed 18.6% (P = .02) more no heavy–drinking days, with a number needed to treat (NNT) of 5.4, and 13.8% (P = .04) more total abstinence days, with an NNT of 6.2. The prestudy high–alcohol withdrawal group in particular had significantly less relapse to heavy drinking (P = .02; NNT, 3.1) and more total abstinence (P = .03; NNT, 2.7) when treated with gabapentin.
A couple of study limitations were a significant noncompletion rate (30% in gabapentin arm and 39% in the placebo arm) and self-reported alcohol withdrawal symptoms prior to entry into the study.
Bottom line: Gabapentin helps in reducing drinking and maintaining alcohol abstinence in individuals with alcohol use disorder, especially those with high–alcohol withdrawal symptoms.
Citation: Anton RF et al. Efficacy of gabapentin for the treatment of alcohol use disorder in patients with alcohol withdrawal symptoms: A randomized clinical trial. JAMA Intern Med. 2020 Mar 9;180(5):728-36.
Dr. Gaddam is a hospitalist and assistant professor of medicine at UK HealthCare, Lexington, Ky.
New recommendations address ME/CFS diagnosis and management
New consensus recommendations address diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), with advice that may also be helpful for patients with lingering symptoms following acute COVID-19 infection.
The document was published online Aug. 25, 2021, in the Mayo Clinic Proceedings by the 23-member U.S. ME/CFS Clinician Coalition, headed by Lucinda Bateman, MD, of the Bateman Horne Center of Excellence, Salt Lake City. The document is the culmination of work that began with a summit held at the center in March 2018.
The target audience is both generalist and specialist health care providers. While ME/CFS is estimated to affect up to 2.5 million Americans, more than 90% are either undiagnosed or misdiagnosed with other conditions such as depression. And those who are diagnosed often receive inappropriate, outdated treatments such as psychotherapy and exercise prescriptions.
“Despite myalgic encephalomyelitis/chronic fatigue syndrome affecting millions of people worldwide, many clinicians lack the knowledge to appropriately diagnose or manage ME/CFS. Unfortunately, clinical guidance has been scarce, obsolete, or potentially harmful,” Dr. Bateman and colleagues wrote.
The urgency of appropriate recognition and management of ME/CFS has increased as growing numbers of people are exhibiting signs and symptoms of ME/CFS following acute COVID-19 infection. This isn’t surprising because the illness has long been linked to other infections, including Epstein-Barr virus, the authors noted.
The document covers the epidemiology, impact, and prognosis of ME/CFS, as well as etiology and pathophysiology. “Scientific studies demonstrate multiple dysfunctional organ systems, including neuro, immune, and metabolic, in ME/CFS. These findings are not explained merely by deconditioning,” document coauthor Lily Chu, MD, an independent consultant in Burlingame, Calif., said in an interview.
The document reviews the 2015 U.S. Institute of Medicine (now Academy of Medicine) diagnostic criteria that are now also recommended by the Centers for Disease Control and Prevention. They are based on four main symptoms: substantial reduction or impairment in the ability to engage in preillness levels of occupational, educational, social or personal activities for longer than 6 months; postexertional malaise, a worsening of all current symptoms, that patients often describe as a “crash”; unrefreshing sleep; and cognitive impairment and/or orthostatic intolerance.
“The new diagnostic criteria focusing on the key symptom of postexertional malaise rather than chronic fatigue, which is common in many conditions, may make the diagnostic process quicker and more accurate. Diagnosis now is both an inclusionary and not just exclusionary process, so it’s not necessary to eliminate all causes of fatigue. Diagnose patients who fit the criteria and be alert for it in people with persistent symptoms post COVID,” Dr. Chu said.
The document provides advice for taking a clinical history to obtain the information necessary for making the diagnosis, including use of laboratory testing to rule out other conditions. Physical exams, while they may not reveal specific abnormalities, may help in identifying comorbidities and ruling out alternative diagnoses.
A long list of nonpharmacologic and pharmacologic treatment and management approaches is offered for each of the individual core and common ME/CFS symptoms, including postexertional malaise, orthostatic intolerance, sleep issues, cognitive dysfunction and fatigue, immune dysfunction, pain, and gastrointestinal issues.
The document recommends against using the “outdated standard of care” cognitive-behavioral therapy and graded exercise therapy as primary treatments for the illness. Instead, the authors recommend teaching patients “pacing,” an individualized approach to energy conservation aimed at minimizing the frequency, duration, and severity of postexertional malaise.
Clinicians are also advised to assess patients’ daily living needs and provide support, including acquiring handicap placards, work or school accommodations, and disability benefits.
“There are things clinicians can do now to help patients even without a disease-modifying treatment. These are actions they are already familiar with and carry out for people with other chronic diseases, which often have limited treatment options as well. Don’t underestimate the importance and value of supportive care for patients.” Dr. Chu said.
The recommendations are based primarily on clinical expertise because there are very few randomized trials, and much of the evidence from other types of trials has been flawed, document coauthor Anthony L. Komaroff, MD, of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said in an interview.
“The sad reality is there aren’t very many large randomized clinical trials with this illness and so what a group of very experienced clinicians did was to gather their collective experience and report it as that. It’s largely uncontrolled experience, but from people who have seen a lot of patients, for what it’s worth to the medical community.”
Dr. Komaroff also advised that clinicians watch out for ME/CFS in patients with long COVID. “If we find that those called long COVID meet ME/CFS criteria, the reason for knowing that is that there are already some treatments that according to experienced clinicians are helpful for ME/CFS, and it would be perfectly appropriate to try some of them in long COVID, particularly the ones that have minimal adverse reactions.”
The guidelines project was supported by the Open Medicine Foundation. Dr. Komaroff reported receiving personal fees from Serimmune outside the submitted work. Dr. Chu has no disclosures.
New consensus recommendations address diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), with advice that may also be helpful for patients with lingering symptoms following acute COVID-19 infection.
The document was published online Aug. 25, 2021, in the Mayo Clinic Proceedings by the 23-member U.S. ME/CFS Clinician Coalition, headed by Lucinda Bateman, MD, of the Bateman Horne Center of Excellence, Salt Lake City. The document is the culmination of work that began with a summit held at the center in March 2018.
The target audience is both generalist and specialist health care providers. While ME/CFS is estimated to affect up to 2.5 million Americans, more than 90% are either undiagnosed or misdiagnosed with other conditions such as depression. And those who are diagnosed often receive inappropriate, outdated treatments such as psychotherapy and exercise prescriptions.
“Despite myalgic encephalomyelitis/chronic fatigue syndrome affecting millions of people worldwide, many clinicians lack the knowledge to appropriately diagnose or manage ME/CFS. Unfortunately, clinical guidance has been scarce, obsolete, or potentially harmful,” Dr. Bateman and colleagues wrote.
The urgency of appropriate recognition and management of ME/CFS has increased as growing numbers of people are exhibiting signs and symptoms of ME/CFS following acute COVID-19 infection. This isn’t surprising because the illness has long been linked to other infections, including Epstein-Barr virus, the authors noted.
The document covers the epidemiology, impact, and prognosis of ME/CFS, as well as etiology and pathophysiology. “Scientific studies demonstrate multiple dysfunctional organ systems, including neuro, immune, and metabolic, in ME/CFS. These findings are not explained merely by deconditioning,” document coauthor Lily Chu, MD, an independent consultant in Burlingame, Calif., said in an interview.
The document reviews the 2015 U.S. Institute of Medicine (now Academy of Medicine) diagnostic criteria that are now also recommended by the Centers for Disease Control and Prevention. They are based on four main symptoms: substantial reduction or impairment in the ability to engage in preillness levels of occupational, educational, social or personal activities for longer than 6 months; postexertional malaise, a worsening of all current symptoms, that patients often describe as a “crash”; unrefreshing sleep; and cognitive impairment and/or orthostatic intolerance.
“The new diagnostic criteria focusing on the key symptom of postexertional malaise rather than chronic fatigue, which is common in many conditions, may make the diagnostic process quicker and more accurate. Diagnosis now is both an inclusionary and not just exclusionary process, so it’s not necessary to eliminate all causes of fatigue. Diagnose patients who fit the criteria and be alert for it in people with persistent symptoms post COVID,” Dr. Chu said.
The document provides advice for taking a clinical history to obtain the information necessary for making the diagnosis, including use of laboratory testing to rule out other conditions. Physical exams, while they may not reveal specific abnormalities, may help in identifying comorbidities and ruling out alternative diagnoses.
A long list of nonpharmacologic and pharmacologic treatment and management approaches is offered for each of the individual core and common ME/CFS symptoms, including postexertional malaise, orthostatic intolerance, sleep issues, cognitive dysfunction and fatigue, immune dysfunction, pain, and gastrointestinal issues.
The document recommends against using the “outdated standard of care” cognitive-behavioral therapy and graded exercise therapy as primary treatments for the illness. Instead, the authors recommend teaching patients “pacing,” an individualized approach to energy conservation aimed at minimizing the frequency, duration, and severity of postexertional malaise.
Clinicians are also advised to assess patients’ daily living needs and provide support, including acquiring handicap placards, work or school accommodations, and disability benefits.
“There are things clinicians can do now to help patients even without a disease-modifying treatment. These are actions they are already familiar with and carry out for people with other chronic diseases, which often have limited treatment options as well. Don’t underestimate the importance and value of supportive care for patients.” Dr. Chu said.
The recommendations are based primarily on clinical expertise because there are very few randomized trials, and much of the evidence from other types of trials has been flawed, document coauthor Anthony L. Komaroff, MD, of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said in an interview.
“The sad reality is there aren’t very many large randomized clinical trials with this illness and so what a group of very experienced clinicians did was to gather their collective experience and report it as that. It’s largely uncontrolled experience, but from people who have seen a lot of patients, for what it’s worth to the medical community.”
Dr. Komaroff also advised that clinicians watch out for ME/CFS in patients with long COVID. “If we find that those called long COVID meet ME/CFS criteria, the reason for knowing that is that there are already some treatments that according to experienced clinicians are helpful for ME/CFS, and it would be perfectly appropriate to try some of them in long COVID, particularly the ones that have minimal adverse reactions.”
The guidelines project was supported by the Open Medicine Foundation. Dr. Komaroff reported receiving personal fees from Serimmune outside the submitted work. Dr. Chu has no disclosures.
New consensus recommendations address diagnosis and management of myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), with advice that may also be helpful for patients with lingering symptoms following acute COVID-19 infection.
The document was published online Aug. 25, 2021, in the Mayo Clinic Proceedings by the 23-member U.S. ME/CFS Clinician Coalition, headed by Lucinda Bateman, MD, of the Bateman Horne Center of Excellence, Salt Lake City. The document is the culmination of work that began with a summit held at the center in March 2018.
The target audience is both generalist and specialist health care providers. While ME/CFS is estimated to affect up to 2.5 million Americans, more than 90% are either undiagnosed or misdiagnosed with other conditions such as depression. And those who are diagnosed often receive inappropriate, outdated treatments such as psychotherapy and exercise prescriptions.
“Despite myalgic encephalomyelitis/chronic fatigue syndrome affecting millions of people worldwide, many clinicians lack the knowledge to appropriately diagnose or manage ME/CFS. Unfortunately, clinical guidance has been scarce, obsolete, or potentially harmful,” Dr. Bateman and colleagues wrote.
The urgency of appropriate recognition and management of ME/CFS has increased as growing numbers of people are exhibiting signs and symptoms of ME/CFS following acute COVID-19 infection. This isn’t surprising because the illness has long been linked to other infections, including Epstein-Barr virus, the authors noted.
The document covers the epidemiology, impact, and prognosis of ME/CFS, as well as etiology and pathophysiology. “Scientific studies demonstrate multiple dysfunctional organ systems, including neuro, immune, and metabolic, in ME/CFS. These findings are not explained merely by deconditioning,” document coauthor Lily Chu, MD, an independent consultant in Burlingame, Calif., said in an interview.
The document reviews the 2015 U.S. Institute of Medicine (now Academy of Medicine) diagnostic criteria that are now also recommended by the Centers for Disease Control and Prevention. They are based on four main symptoms: substantial reduction or impairment in the ability to engage in preillness levels of occupational, educational, social or personal activities for longer than 6 months; postexertional malaise, a worsening of all current symptoms, that patients often describe as a “crash”; unrefreshing sleep; and cognitive impairment and/or orthostatic intolerance.
“The new diagnostic criteria focusing on the key symptom of postexertional malaise rather than chronic fatigue, which is common in many conditions, may make the diagnostic process quicker and more accurate. Diagnosis now is both an inclusionary and not just exclusionary process, so it’s not necessary to eliminate all causes of fatigue. Diagnose patients who fit the criteria and be alert for it in people with persistent symptoms post COVID,” Dr. Chu said.
The document provides advice for taking a clinical history to obtain the information necessary for making the diagnosis, including use of laboratory testing to rule out other conditions. Physical exams, while they may not reveal specific abnormalities, may help in identifying comorbidities and ruling out alternative diagnoses.
A long list of nonpharmacologic and pharmacologic treatment and management approaches is offered for each of the individual core and common ME/CFS symptoms, including postexertional malaise, orthostatic intolerance, sleep issues, cognitive dysfunction and fatigue, immune dysfunction, pain, and gastrointestinal issues.
The document recommends against using the “outdated standard of care” cognitive-behavioral therapy and graded exercise therapy as primary treatments for the illness. Instead, the authors recommend teaching patients “pacing,” an individualized approach to energy conservation aimed at minimizing the frequency, duration, and severity of postexertional malaise.
Clinicians are also advised to assess patients’ daily living needs and provide support, including acquiring handicap placards, work or school accommodations, and disability benefits.
“There are things clinicians can do now to help patients even without a disease-modifying treatment. These are actions they are already familiar with and carry out for people with other chronic diseases, which often have limited treatment options as well. Don’t underestimate the importance and value of supportive care for patients.” Dr. Chu said.
The recommendations are based primarily on clinical expertise because there are very few randomized trials, and much of the evidence from other types of trials has been flawed, document coauthor Anthony L. Komaroff, MD, of Brigham and Women’s Hospital and Harvard Medical School, both in Boston, said in an interview.
“The sad reality is there aren’t very many large randomized clinical trials with this illness and so what a group of very experienced clinicians did was to gather their collective experience and report it as that. It’s largely uncontrolled experience, but from people who have seen a lot of patients, for what it’s worth to the medical community.”
Dr. Komaroff also advised that clinicians watch out for ME/CFS in patients with long COVID. “If we find that those called long COVID meet ME/CFS criteria, the reason for knowing that is that there are already some treatments that according to experienced clinicians are helpful for ME/CFS, and it would be perfectly appropriate to try some of them in long COVID, particularly the ones that have minimal adverse reactions.”
The guidelines project was supported by the Open Medicine Foundation. Dr. Komaroff reported receiving personal fees from Serimmune outside the submitted work. Dr. Chu has no disclosures.
FROM THE MAYO CLINIC PROCEEDINGS
Anxiety, inactivity linked to cognitive impairment in Parkinson’s
Parkinson’s disease patients who develop anxiety early in their disease are at risk for reduced physical activity, which promotes further anxiety and cognitive decline, data from nearly 500 individuals show.
Anxiety occurs in 20%-60% of Parkinson’s disease (PD) patients but often goes undiagnosed, wrote Jacob D. Jones, PhD, of California State University, San Bernardino, and colleagues.
“Anxiety can attenuate motivation to engage in physical activity leading to more anxiety and other negative cognitive outcomes,” although physical activity has been shown to improve cognitive function in PD patients, they said. However, physical activity as a mediator between anxiety and cognitive function in PD has not been well studied, they noted.
In a study published in Mental Health and Physical Activity Participants were followed for up to 5 years and completed neuropsychological tests, tests of motor severity, and self-reports on anxiety and physical activity. Anxiety was assessed using the State-Trait Anxiety Inventory-Trait (STAI-T) subscale. Physical activity was assessed using the Physical Activity Scale for the Elderly (PASE). Motor severity was assessed using the Unified Parkinson’s Disease Rating Scale-Part III (UPDRS). The average age of the participants was 61 years, 65% were men, and 96% were White.
Using a direct-effect model, the researchers found that individuals whose anxiety increased during the study period also showed signs of cognitive decline. A significant between-person effect showed that individuals who were generally more anxious also scored lower on cognitive tests over the 5-year study period.
In a mediation model computed with structural equation modeling, physical activity mediated the link between anxiety and cognition, most notably household activity.
“There was a significant within-person association between anxiety and household activities, meaning that individuals who became more anxious over the 5-year study also became less active in the home,” reported Dr. Jones and colleagues.
However, no significant indirect effect was noted regarding the between-person findings of the impact of physical activity on anxiety and cognitive decline. Although more severe anxiety was associated with less activity, cognitive performance was not associated with either type of physical activity.
The presence of a within-person effect “suggests that reductions in physical activity, specifically within the first 5 years of disease onset, may be detrimental to mental health,” the researchers emphasized. Given that the study population was newly diagnosed with PD “it is likely the within-person terms are more sensitive to changes in anxiety, physical activity, and cognition that are more directly the result of the PD process, as opposed to lifestyle/preexisting traits,” they said.
The study findings were limited by several factors, including the use of self-reports to measure physical activity, and the lack of granular information about the details of physical activity, the researchers noted. Another limitation was the inclusion of only newly diagnosed PD patients, which might limit generalizability.
“Future research is warranted to understand if other modes, intensities, or complexities of physical activity impact individuals with PD in a different manner in relation to cognition,” they said.
Dr. Jones and colleagues had no disclosures. The PPMI is supported by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including numerous pharmaceutical companies.
Parkinson’s disease patients who develop anxiety early in their disease are at risk for reduced physical activity, which promotes further anxiety and cognitive decline, data from nearly 500 individuals show.
Anxiety occurs in 20%-60% of Parkinson’s disease (PD) patients but often goes undiagnosed, wrote Jacob D. Jones, PhD, of California State University, San Bernardino, and colleagues.
“Anxiety can attenuate motivation to engage in physical activity leading to more anxiety and other negative cognitive outcomes,” although physical activity has been shown to improve cognitive function in PD patients, they said. However, physical activity as a mediator between anxiety and cognitive function in PD has not been well studied, they noted.
In a study published in Mental Health and Physical Activity Participants were followed for up to 5 years and completed neuropsychological tests, tests of motor severity, and self-reports on anxiety and physical activity. Anxiety was assessed using the State-Trait Anxiety Inventory-Trait (STAI-T) subscale. Physical activity was assessed using the Physical Activity Scale for the Elderly (PASE). Motor severity was assessed using the Unified Parkinson’s Disease Rating Scale-Part III (UPDRS). The average age of the participants was 61 years, 65% were men, and 96% were White.
Using a direct-effect model, the researchers found that individuals whose anxiety increased during the study period also showed signs of cognitive decline. A significant between-person effect showed that individuals who were generally more anxious also scored lower on cognitive tests over the 5-year study period.
In a mediation model computed with structural equation modeling, physical activity mediated the link between anxiety and cognition, most notably household activity.
“There was a significant within-person association between anxiety and household activities, meaning that individuals who became more anxious over the 5-year study also became less active in the home,” reported Dr. Jones and colleagues.
However, no significant indirect effect was noted regarding the between-person findings of the impact of physical activity on anxiety and cognitive decline. Although more severe anxiety was associated with less activity, cognitive performance was not associated with either type of physical activity.
The presence of a within-person effect “suggests that reductions in physical activity, specifically within the first 5 years of disease onset, may be detrimental to mental health,” the researchers emphasized. Given that the study population was newly diagnosed with PD “it is likely the within-person terms are more sensitive to changes in anxiety, physical activity, and cognition that are more directly the result of the PD process, as opposed to lifestyle/preexisting traits,” they said.
The study findings were limited by several factors, including the use of self-reports to measure physical activity, and the lack of granular information about the details of physical activity, the researchers noted. Another limitation was the inclusion of only newly diagnosed PD patients, which might limit generalizability.
“Future research is warranted to understand if other modes, intensities, or complexities of physical activity impact individuals with PD in a different manner in relation to cognition,” they said.
Dr. Jones and colleagues had no disclosures. The PPMI is supported by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including numerous pharmaceutical companies.
Parkinson’s disease patients who develop anxiety early in their disease are at risk for reduced physical activity, which promotes further anxiety and cognitive decline, data from nearly 500 individuals show.
Anxiety occurs in 20%-60% of Parkinson’s disease (PD) patients but often goes undiagnosed, wrote Jacob D. Jones, PhD, of California State University, San Bernardino, and colleagues.
“Anxiety can attenuate motivation to engage in physical activity leading to more anxiety and other negative cognitive outcomes,” although physical activity has been shown to improve cognitive function in PD patients, they said. However, physical activity as a mediator between anxiety and cognitive function in PD has not been well studied, they noted.
In a study published in Mental Health and Physical Activity Participants were followed for up to 5 years and completed neuropsychological tests, tests of motor severity, and self-reports on anxiety and physical activity. Anxiety was assessed using the State-Trait Anxiety Inventory-Trait (STAI-T) subscale. Physical activity was assessed using the Physical Activity Scale for the Elderly (PASE). Motor severity was assessed using the Unified Parkinson’s Disease Rating Scale-Part III (UPDRS). The average age of the participants was 61 years, 65% were men, and 96% were White.
Using a direct-effect model, the researchers found that individuals whose anxiety increased during the study period also showed signs of cognitive decline. A significant between-person effect showed that individuals who were generally more anxious also scored lower on cognitive tests over the 5-year study period.
In a mediation model computed with structural equation modeling, physical activity mediated the link between anxiety and cognition, most notably household activity.
“There was a significant within-person association between anxiety and household activities, meaning that individuals who became more anxious over the 5-year study also became less active in the home,” reported Dr. Jones and colleagues.
However, no significant indirect effect was noted regarding the between-person findings of the impact of physical activity on anxiety and cognitive decline. Although more severe anxiety was associated with less activity, cognitive performance was not associated with either type of physical activity.
The presence of a within-person effect “suggests that reductions in physical activity, specifically within the first 5 years of disease onset, may be detrimental to mental health,” the researchers emphasized. Given that the study population was newly diagnosed with PD “it is likely the within-person terms are more sensitive to changes in anxiety, physical activity, and cognition that are more directly the result of the PD process, as opposed to lifestyle/preexisting traits,” they said.
The study findings were limited by several factors, including the use of self-reports to measure physical activity, and the lack of granular information about the details of physical activity, the researchers noted. Another limitation was the inclusion of only newly diagnosed PD patients, which might limit generalizability.
“Future research is warranted to understand if other modes, intensities, or complexities of physical activity impact individuals with PD in a different manner in relation to cognition,” they said.
Dr. Jones and colleagues had no disclosures. The PPMI is supported by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including numerous pharmaceutical companies.
FROM MENTAL HEALTH AND PHYSICAL ACTIVITY
Stimulating jobs may help stave off dementia onset
Individuals with cognitively stimulating jobs are at a lower risk of developing dementia than their peers with less challenging employment, new research suggests.
Results from a large, multicohort study also showed an association between cognitive stimulation and lower levels of certain plasma proteins, providing possible clues on a protective biological mechanism.
“These new findings support the hypothesis that mental stimulation in adulthood may postpone the onset of dementia,” Mika Kivimäki, PhD, professor and director of the Whitehall II Study, department of epidemiology, University College London, said in an interview.
The results were published online Aug. 19, 2021, in the BMJ.
‘Work fast and hard’
Researchers assessed the association between workplace cognitive stimulation and dementia incidence in seven cohorts that included almost 108,000 men and women (mean age, 44.6 years). All were free of dementia at baseline.
Participants included civil servants, public sector employees, forestry workers, and others from the general working population.
Investigators separated the participants into three categories of workplace cognitive stimulation: “high,” which referred to both high job demand and high job control; “low,” which referred to low demands and low control; and “medium,” which referred to all other combinations of job demand and job control.
“Highly cognitively stimulating jobs require you to work fast and hard, learn new things, be creative, and have a high level of skill,” said Dr. Kivimäki.
The researchers controlled for low education, hypertension, smoking, obesity, depression, physical inactivity, diabetes, low social contact, excessive alcohol consumption, and traumatic brain injury. These represent 10 of the 12 dementia risk factors named by the 2020 Lancet Commission on Dementia Prevention as having convincing evidence, Dr. Kivimäki noted.
Although the investigators had no data on the other two risk factors of hearing loss and air pollution, these are unlikely to be confounding factors, he said.
Follow-up for incident dementia varied from 13.7 to 30.1 years, depending on the cohort, and was 16.7 years in the total patient population. The mean age at dementia onset was 71.2 years.
Benefits across the life course
Results showed that incident dementia per 10,000 person years was 7.3 in the low–cognitive stimulation group and 4.8 in the high-stimulation group, for a difference of 2.5.
“These differences were relatively small because the incidence of dementia in this relatively young population was low,” Dr. Kivimäki said.
Compared with those with low stimulation, the adjusted hazard ratio for dementia for this with high stimulation was 0.77 (95% CI, 0.65-0.92).
The results were similar for men and women, and for those younger and older than 60 years. However, the link between workplace cognitive stimulation appeared stronger for Alzheimer’s disease than for other dementias.
There also appeared to be additive effects of higher cognitive stimulation in both childhood, as indicated by higher educational attainment, and adulthood, based on work characteristics, said Dr. Kivimäki.
“These findings support the benefits of cognitive stimulation across the life course, with education leading to higher peak cognitive performance and cognitive stimulation at work lowering age-related cognitive decline,” he added.
The findings don’t seem to be the result of workers with cognitive impairment remaining in unchallenging jobs, he noted. Separate analyses showed lower dementia incidence even when 10 years or more separated the assessment of cognitive stimulation and the dementia diagnosis.
“This suggests that the findings are unlikely to be biased due to reverse causation,” Dr. Kivimäki said.
Possible mechanism
Findings were similar when the researchers assessed effect from job changes. “This is probably because people in highly stimulating jobs are more likely to change to another highly stimulating job than to a low-stimulating job,” said Dr. Kivimäki. “Similarly, people with less stimulating jobs are seldom able to change to a substantially more stimulating job.”
As a dementia risk factor, low workplace stimulation is comparable with high alcohol intake and physical inactivity, but is weaker than education, diabetes, smoking, hypertension, and obesity, Dr. Kivimäki noted.
When asked about individuals with less cognitively stimulating jobs who are enormously stimulated outside work, he said that “previous large-scale studies have failed to find evidence that leisure time cognitive activity would significantly reduce risk of dementia.”
To explore potential underlying mechanisms, the investigators examined almost 5,000 plasma proteins in more than 2,200 individuals from one cohort in the Whitehall II study. They found six proteins were significantly lower among participants with high versus low cognitive stimulation.
In another analysis that included more than 13,500 participants from the Whitehall and another cohort, higher levels of three of these plasma proteins were associated with increased dementia risk – or conversely, lower protein levels with lower dementia risk.
The findings suggest a “novel plausible explanation” for the link between workplace cognitive stimulation and dementia risk, said Dr. Kivimäki.
He noted that higher levels of certain proteins prevent brain cells from forming new connections.
‘Some of the most compelling evidence to date’
In an accompanying editorial, Serhiy Dekhtyar, PhD, assistant professor (Docent), Aging Research Center, Karolinska Institute, Stockholm, noted that the study is “an important piece of work” and “some of the most compelling evidence to date” on the role of occupational cognitive stimulation in dementia risk.
The large-scale investigation in multiple cohorts and contexts has “advanced the field” and could help “explain previously mixed findings in the literature,” Dekhtyar said in an interview.
Importantly, the researchers provide “an indication of biological mechanisms potentially connecting work mental stimulation and dementia,” he added.
However, Dr. Dekhtyar noted that the difference of 2.5 incident cases of dementia per 10,000 person years of follow-up between the low and high mental-stimulation groups “is not especially large” – although it is comparable with other established risk factors for dementia.
He suspects the effect size would have been larger had the follow-up for dementia been longer.
Dr. Dekhtyar also raised the possibility that “innate cognition” might affect both educational and occupational attainment, and the subsequent dementia risk.
“Without taking this into account, we may inadvertently conclude that education or occupational stimulation help differentially preserve cognition into late life – when in reality, it may be initial differences in cognitive ability that are preserved throughout life,” he concluded.
Funding sources for the study included Nordic Research Programme on Health and Welfare (NordForsk), Medical Research Council, Wellcome Trust, Academy of Finland, and Helsinki Institute of Life Science. Dr. Kivimäki has received support from NordForsk, the UK Medical Research Council, the Wellcome Trust, the Academy of Finland, and the Helsinki Institute of Life Science. Dr. Dekhtyar disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Individuals with cognitively stimulating jobs are at a lower risk of developing dementia than their peers with less challenging employment, new research suggests.
Results from a large, multicohort study also showed an association between cognitive stimulation and lower levels of certain plasma proteins, providing possible clues on a protective biological mechanism.
“These new findings support the hypothesis that mental stimulation in adulthood may postpone the onset of dementia,” Mika Kivimäki, PhD, professor and director of the Whitehall II Study, department of epidemiology, University College London, said in an interview.
The results were published online Aug. 19, 2021, in the BMJ.
‘Work fast and hard’
Researchers assessed the association between workplace cognitive stimulation and dementia incidence in seven cohorts that included almost 108,000 men and women (mean age, 44.6 years). All were free of dementia at baseline.
Participants included civil servants, public sector employees, forestry workers, and others from the general working population.
Investigators separated the participants into three categories of workplace cognitive stimulation: “high,” which referred to both high job demand and high job control; “low,” which referred to low demands and low control; and “medium,” which referred to all other combinations of job demand and job control.
“Highly cognitively stimulating jobs require you to work fast and hard, learn new things, be creative, and have a high level of skill,” said Dr. Kivimäki.
The researchers controlled for low education, hypertension, smoking, obesity, depression, physical inactivity, diabetes, low social contact, excessive alcohol consumption, and traumatic brain injury. These represent 10 of the 12 dementia risk factors named by the 2020 Lancet Commission on Dementia Prevention as having convincing evidence, Dr. Kivimäki noted.
Although the investigators had no data on the other two risk factors of hearing loss and air pollution, these are unlikely to be confounding factors, he said.
Follow-up for incident dementia varied from 13.7 to 30.1 years, depending on the cohort, and was 16.7 years in the total patient population. The mean age at dementia onset was 71.2 years.
Benefits across the life course
Results showed that incident dementia per 10,000 person years was 7.3 in the low–cognitive stimulation group and 4.8 in the high-stimulation group, for a difference of 2.5.
“These differences were relatively small because the incidence of dementia in this relatively young population was low,” Dr. Kivimäki said.
Compared with those with low stimulation, the adjusted hazard ratio for dementia for this with high stimulation was 0.77 (95% CI, 0.65-0.92).
The results were similar for men and women, and for those younger and older than 60 years. However, the link between workplace cognitive stimulation appeared stronger for Alzheimer’s disease than for other dementias.
There also appeared to be additive effects of higher cognitive stimulation in both childhood, as indicated by higher educational attainment, and adulthood, based on work characteristics, said Dr. Kivimäki.
“These findings support the benefits of cognitive stimulation across the life course, with education leading to higher peak cognitive performance and cognitive stimulation at work lowering age-related cognitive decline,” he added.
The findings don’t seem to be the result of workers with cognitive impairment remaining in unchallenging jobs, he noted. Separate analyses showed lower dementia incidence even when 10 years or more separated the assessment of cognitive stimulation and the dementia diagnosis.
“This suggests that the findings are unlikely to be biased due to reverse causation,” Dr. Kivimäki said.
Possible mechanism
Findings were similar when the researchers assessed effect from job changes. “This is probably because people in highly stimulating jobs are more likely to change to another highly stimulating job than to a low-stimulating job,” said Dr. Kivimäki. “Similarly, people with less stimulating jobs are seldom able to change to a substantially more stimulating job.”
As a dementia risk factor, low workplace stimulation is comparable with high alcohol intake and physical inactivity, but is weaker than education, diabetes, smoking, hypertension, and obesity, Dr. Kivimäki noted.
When asked about individuals with less cognitively stimulating jobs who are enormously stimulated outside work, he said that “previous large-scale studies have failed to find evidence that leisure time cognitive activity would significantly reduce risk of dementia.”
To explore potential underlying mechanisms, the investigators examined almost 5,000 plasma proteins in more than 2,200 individuals from one cohort in the Whitehall II study. They found six proteins were significantly lower among participants with high versus low cognitive stimulation.
In another analysis that included more than 13,500 participants from the Whitehall and another cohort, higher levels of three of these plasma proteins were associated with increased dementia risk – or conversely, lower protein levels with lower dementia risk.
The findings suggest a “novel plausible explanation” for the link between workplace cognitive stimulation and dementia risk, said Dr. Kivimäki.
He noted that higher levels of certain proteins prevent brain cells from forming new connections.
‘Some of the most compelling evidence to date’
In an accompanying editorial, Serhiy Dekhtyar, PhD, assistant professor (Docent), Aging Research Center, Karolinska Institute, Stockholm, noted that the study is “an important piece of work” and “some of the most compelling evidence to date” on the role of occupational cognitive stimulation in dementia risk.
The large-scale investigation in multiple cohorts and contexts has “advanced the field” and could help “explain previously mixed findings in the literature,” Dekhtyar said in an interview.
Importantly, the researchers provide “an indication of biological mechanisms potentially connecting work mental stimulation and dementia,” he added.
However, Dr. Dekhtyar noted that the difference of 2.5 incident cases of dementia per 10,000 person years of follow-up between the low and high mental-stimulation groups “is not especially large” – although it is comparable with other established risk factors for dementia.
He suspects the effect size would have been larger had the follow-up for dementia been longer.
Dr. Dekhtyar also raised the possibility that “innate cognition” might affect both educational and occupational attainment, and the subsequent dementia risk.
“Without taking this into account, we may inadvertently conclude that education or occupational stimulation help differentially preserve cognition into late life – when in reality, it may be initial differences in cognitive ability that are preserved throughout life,” he concluded.
Funding sources for the study included Nordic Research Programme on Health and Welfare (NordForsk), Medical Research Council, Wellcome Trust, Academy of Finland, and Helsinki Institute of Life Science. Dr. Kivimäki has received support from NordForsk, the UK Medical Research Council, the Wellcome Trust, the Academy of Finland, and the Helsinki Institute of Life Science. Dr. Dekhtyar disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Individuals with cognitively stimulating jobs are at a lower risk of developing dementia than their peers with less challenging employment, new research suggests.
Results from a large, multicohort study also showed an association between cognitive stimulation and lower levels of certain plasma proteins, providing possible clues on a protective biological mechanism.
“These new findings support the hypothesis that mental stimulation in adulthood may postpone the onset of dementia,” Mika Kivimäki, PhD, professor and director of the Whitehall II Study, department of epidemiology, University College London, said in an interview.
The results were published online Aug. 19, 2021, in the BMJ.
‘Work fast and hard’
Researchers assessed the association between workplace cognitive stimulation and dementia incidence in seven cohorts that included almost 108,000 men and women (mean age, 44.6 years). All were free of dementia at baseline.
Participants included civil servants, public sector employees, forestry workers, and others from the general working population.
Investigators separated the participants into three categories of workplace cognitive stimulation: “high,” which referred to both high job demand and high job control; “low,” which referred to low demands and low control; and “medium,” which referred to all other combinations of job demand and job control.
“Highly cognitively stimulating jobs require you to work fast and hard, learn new things, be creative, and have a high level of skill,” said Dr. Kivimäki.
The researchers controlled for low education, hypertension, smoking, obesity, depression, physical inactivity, diabetes, low social contact, excessive alcohol consumption, and traumatic brain injury. These represent 10 of the 12 dementia risk factors named by the 2020 Lancet Commission on Dementia Prevention as having convincing evidence, Dr. Kivimäki noted.
Although the investigators had no data on the other two risk factors of hearing loss and air pollution, these are unlikely to be confounding factors, he said.
Follow-up for incident dementia varied from 13.7 to 30.1 years, depending on the cohort, and was 16.7 years in the total patient population. The mean age at dementia onset was 71.2 years.
Benefits across the life course
Results showed that incident dementia per 10,000 person years was 7.3 in the low–cognitive stimulation group and 4.8 in the high-stimulation group, for a difference of 2.5.
“These differences were relatively small because the incidence of dementia in this relatively young population was low,” Dr. Kivimäki said.
Compared with those with low stimulation, the adjusted hazard ratio for dementia for this with high stimulation was 0.77 (95% CI, 0.65-0.92).
The results were similar for men and women, and for those younger and older than 60 years. However, the link between workplace cognitive stimulation appeared stronger for Alzheimer’s disease than for other dementias.
There also appeared to be additive effects of higher cognitive stimulation in both childhood, as indicated by higher educational attainment, and adulthood, based on work characteristics, said Dr. Kivimäki.
“These findings support the benefits of cognitive stimulation across the life course, with education leading to higher peak cognitive performance and cognitive stimulation at work lowering age-related cognitive decline,” he added.
The findings don’t seem to be the result of workers with cognitive impairment remaining in unchallenging jobs, he noted. Separate analyses showed lower dementia incidence even when 10 years or more separated the assessment of cognitive stimulation and the dementia diagnosis.
“This suggests that the findings are unlikely to be biased due to reverse causation,” Dr. Kivimäki said.
Possible mechanism
Findings were similar when the researchers assessed effect from job changes. “This is probably because people in highly stimulating jobs are more likely to change to another highly stimulating job than to a low-stimulating job,” said Dr. Kivimäki. “Similarly, people with less stimulating jobs are seldom able to change to a substantially more stimulating job.”
As a dementia risk factor, low workplace stimulation is comparable with high alcohol intake and physical inactivity, but is weaker than education, diabetes, smoking, hypertension, and obesity, Dr. Kivimäki noted.
When asked about individuals with less cognitively stimulating jobs who are enormously stimulated outside work, he said that “previous large-scale studies have failed to find evidence that leisure time cognitive activity would significantly reduce risk of dementia.”
To explore potential underlying mechanisms, the investigators examined almost 5,000 plasma proteins in more than 2,200 individuals from one cohort in the Whitehall II study. They found six proteins were significantly lower among participants with high versus low cognitive stimulation.
In another analysis that included more than 13,500 participants from the Whitehall and another cohort, higher levels of three of these plasma proteins were associated with increased dementia risk – or conversely, lower protein levels with lower dementia risk.
The findings suggest a “novel plausible explanation” for the link between workplace cognitive stimulation and dementia risk, said Dr. Kivimäki.
He noted that higher levels of certain proteins prevent brain cells from forming new connections.
‘Some of the most compelling evidence to date’
In an accompanying editorial, Serhiy Dekhtyar, PhD, assistant professor (Docent), Aging Research Center, Karolinska Institute, Stockholm, noted that the study is “an important piece of work” and “some of the most compelling evidence to date” on the role of occupational cognitive stimulation in dementia risk.
The large-scale investigation in multiple cohorts and contexts has “advanced the field” and could help “explain previously mixed findings in the literature,” Dekhtyar said in an interview.
Importantly, the researchers provide “an indication of biological mechanisms potentially connecting work mental stimulation and dementia,” he added.
However, Dr. Dekhtyar noted that the difference of 2.5 incident cases of dementia per 10,000 person years of follow-up between the low and high mental-stimulation groups “is not especially large” – although it is comparable with other established risk factors for dementia.
He suspects the effect size would have been larger had the follow-up for dementia been longer.
Dr. Dekhtyar also raised the possibility that “innate cognition” might affect both educational and occupational attainment, and the subsequent dementia risk.
“Without taking this into account, we may inadvertently conclude that education or occupational stimulation help differentially preserve cognition into late life – when in reality, it may be initial differences in cognitive ability that are preserved throughout life,” he concluded.
Funding sources for the study included Nordic Research Programme on Health and Welfare (NordForsk), Medical Research Council, Wellcome Trust, Academy of Finland, and Helsinki Institute of Life Science. Dr. Kivimäki has received support from NordForsk, the UK Medical Research Council, the Wellcome Trust, the Academy of Finland, and the Helsinki Institute of Life Science. Dr. Dekhtyar disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Headache seen affecting some pregnancy outcomes
, according to results from an observational study.
Migraine during pregnancy has been associated in previous studies with hypertensive pregnancy complications including preeclampsia; however, little is known about other headache types and their effects on pregnancy and birth outcomes.
For their research, published online July 20 in Cephalalgia, Isabella Neri, MD, PhD, and colleagues at Hospital Policlinico of Modena, Italy, looked at headache status for 515 consecutive pregnant women evaluated during their first trimester and followed through childbirth.
Altogether 224 women, or 43.5% of the cohort, were diagnosed with migraine without aura (n = 72), migraine with aura (n = 27), or tension-type headache (n = 125). The authors did not report on the severity or frequency of headaches.
Women with migraine with aura and tension-type headache saw higher rates of small-for-gestational-age infants (25.9% and 10.4% of births, respectively) compared with 5.5% for women without headache. Women presenting with tension-type headache saw elevated risk for small-for-gestational-age infants (odds ratio [OR] 4.19, P = .004) as did women with migraine with aura (OR 5.37, P = .02).
Admission to neonatal intensive care was significantly higher in all the headache groups. However, the authors found no statistically significant associations between headaches and any other perinatal outcome investigated in the study, including gestational diabetes, placental abruption, gestational hypertension, and preterm delivery.
A previous study conducted by the same research group had reported a relationship between migraine and gestational hypertension. The authors cited the small sample size of the migraine groups in the current study, “the diverse features of the population,” and the popularity of low-dose aspirin administration as potentially affecting that outcome.
Interpret findings with caution
Asked by this news organization to comment on the research, two headache neurologists praised Dr. Neri and colleagues’ research for focusing on an understudied topic – but also said that the results would not change their practice unless replicated in larger studies.
Elizabeth W. Loder, MD, MPH, chief emeritus of the division of headache at Brigham and Women’s Faulkner Hospital in Boston, urged caution in interpreting the findings, particularly with regard to tension-type headache. “This study adds to information suggesting that pregnancy complications probably are higher in women who have migraine with aura, and there’s biological plausibility for that,” Dr. Loder said. “Having aura means you may have some vascular abnormalities and things that logically might be associated with an increased risk of small-for-gestational age infants.” But the small size of the migraine-with-aura group in this study – 27 women – and the fact that other perinatal outcomes measured in the study did not reach significance, allows for the possibility that the small-for-gestational-age findings were due to chance, Dr. Loder noted.
With tension-type headache, a biological rationale for small-for-gestational-age risk is more elusive, Dr. Loder said. “I would want to see that association replicated in another study before I thought that I needed to warn women with tension-type headache about this potential outcome. There’s lot of uncertainty here about the magnitude of the risk.”
While Dr. Neri and colleagues described the instruments used in their study to diagnose migraine and migraine with aura, they did not explain how tension-type headache was diagnosed.
Tension-type headache, while common, is still not well characterized, Dr. Loder noted, and may represent a heterogeneous condition or the milder end of a biological continuum that includes migraine with aura. Also, the group in the study had a higher prevalence of smoking, and though the authors made statistical adjustments for smoking status, “smokers are systematically different than people who aren’t in other ways that could be associated with these outcomes,” Dr. Loder said.
While the authors of the study suggested that interventions might be indicated for women with tension-type headache in pregnancy, “showing an association doesn’t necessarily mean that intervening would make a difference” on pregnancy outcomes, Dr. Loder said.
Amaal J. Starling, MD, of the Mayo Clinic in Phoenix, Ariz., said in an interview that she, too, appreciated that this study looked at pregnancy outcomes in the setting of headache disorders. “Unfortunately even though headache disorders and especially migraine affect women so much, we still know very little about migraine in pregnancy,” she said.
Dr. Starling noted that many women with migraine are discouraged by their health care providers from becoming pregnant, because of the false belief that migraine cannot be managed in pregnancy. In her own practice, she said, she treats many patients with severe headache who become pregnant and who require pharmacological intervention during pregnancy.
This does not mean she regards headache in pregnancy as innocent. “I want patients to be on high alert for changes in headache symptoms in pregnancy. If someone has worsening of headache or migraine or aura in the setting of pregnancy, we consider that a red flag,” potentially indicating complications such as high blood pressure, gestational hypertension, or a blood clot.
Like Dr. Loder, Dr. Starling said she was not surprised by Dr. Neri and colleagues’ finding that migraine with aura might impact pregnancy outcomes. “We know that migraine with aura has a lot of vascular abnormalities that underlie the pathogenesis,” she said.
Dr. Starling found the findings related to tension-type headache less convincing, not least because the diagnostic criteria for tension-type headache was not made clear in the study. “I view this as an exploratory study that says maybe there’s a signal here. A larger epidemiological study would need to be done to confirm or refute this data,” Dr. Starling said. Until the findings can be replicated, “this study would not affect my clinical practice in any way.”
Dr. Neri and colleagues described no outside funding for their research or financial conflicts of interest. Dr. Starling has received consulting fees from pharmaceutical manufacturers but reported no disclosures relevant to the study discussed. Dr. Loder reported no financial conflicts of interest.
, according to results from an observational study.
Migraine during pregnancy has been associated in previous studies with hypertensive pregnancy complications including preeclampsia; however, little is known about other headache types and their effects on pregnancy and birth outcomes.
For their research, published online July 20 in Cephalalgia, Isabella Neri, MD, PhD, and colleagues at Hospital Policlinico of Modena, Italy, looked at headache status for 515 consecutive pregnant women evaluated during their first trimester and followed through childbirth.
Altogether 224 women, or 43.5% of the cohort, were diagnosed with migraine without aura (n = 72), migraine with aura (n = 27), or tension-type headache (n = 125). The authors did not report on the severity or frequency of headaches.
Women with migraine with aura and tension-type headache saw higher rates of small-for-gestational-age infants (25.9% and 10.4% of births, respectively) compared with 5.5% for women without headache. Women presenting with tension-type headache saw elevated risk for small-for-gestational-age infants (odds ratio [OR] 4.19, P = .004) as did women with migraine with aura (OR 5.37, P = .02).
Admission to neonatal intensive care was significantly higher in all the headache groups. However, the authors found no statistically significant associations between headaches and any other perinatal outcome investigated in the study, including gestational diabetes, placental abruption, gestational hypertension, and preterm delivery.
A previous study conducted by the same research group had reported a relationship between migraine and gestational hypertension. The authors cited the small sample size of the migraine groups in the current study, “the diverse features of the population,” and the popularity of low-dose aspirin administration as potentially affecting that outcome.
Interpret findings with caution
Asked by this news organization to comment on the research, two headache neurologists praised Dr. Neri and colleagues’ research for focusing on an understudied topic – but also said that the results would not change their practice unless replicated in larger studies.
Elizabeth W. Loder, MD, MPH, chief emeritus of the division of headache at Brigham and Women’s Faulkner Hospital in Boston, urged caution in interpreting the findings, particularly with regard to tension-type headache. “This study adds to information suggesting that pregnancy complications probably are higher in women who have migraine with aura, and there’s biological plausibility for that,” Dr. Loder said. “Having aura means you may have some vascular abnormalities and things that logically might be associated with an increased risk of small-for-gestational age infants.” But the small size of the migraine-with-aura group in this study – 27 women – and the fact that other perinatal outcomes measured in the study did not reach significance, allows for the possibility that the small-for-gestational-age findings were due to chance, Dr. Loder noted.
With tension-type headache, a biological rationale for small-for-gestational-age risk is more elusive, Dr. Loder said. “I would want to see that association replicated in another study before I thought that I needed to warn women with tension-type headache about this potential outcome. There’s lot of uncertainty here about the magnitude of the risk.”
While Dr. Neri and colleagues described the instruments used in their study to diagnose migraine and migraine with aura, they did not explain how tension-type headache was diagnosed.
Tension-type headache, while common, is still not well characterized, Dr. Loder noted, and may represent a heterogeneous condition or the milder end of a biological continuum that includes migraine with aura. Also, the group in the study had a higher prevalence of smoking, and though the authors made statistical adjustments for smoking status, “smokers are systematically different than people who aren’t in other ways that could be associated with these outcomes,” Dr. Loder said.
While the authors of the study suggested that interventions might be indicated for women with tension-type headache in pregnancy, “showing an association doesn’t necessarily mean that intervening would make a difference” on pregnancy outcomes, Dr. Loder said.
Amaal J. Starling, MD, of the Mayo Clinic in Phoenix, Ariz., said in an interview that she, too, appreciated that this study looked at pregnancy outcomes in the setting of headache disorders. “Unfortunately even though headache disorders and especially migraine affect women so much, we still know very little about migraine in pregnancy,” she said.
Dr. Starling noted that many women with migraine are discouraged by their health care providers from becoming pregnant, because of the false belief that migraine cannot be managed in pregnancy. In her own practice, she said, she treats many patients with severe headache who become pregnant and who require pharmacological intervention during pregnancy.
This does not mean she regards headache in pregnancy as innocent. “I want patients to be on high alert for changes in headache symptoms in pregnancy. If someone has worsening of headache or migraine or aura in the setting of pregnancy, we consider that a red flag,” potentially indicating complications such as high blood pressure, gestational hypertension, or a blood clot.
Like Dr. Loder, Dr. Starling said she was not surprised by Dr. Neri and colleagues’ finding that migraine with aura might impact pregnancy outcomes. “We know that migraine with aura has a lot of vascular abnormalities that underlie the pathogenesis,” she said.
Dr. Starling found the findings related to tension-type headache less convincing, not least because the diagnostic criteria for tension-type headache was not made clear in the study. “I view this as an exploratory study that says maybe there’s a signal here. A larger epidemiological study would need to be done to confirm or refute this data,” Dr. Starling said. Until the findings can be replicated, “this study would not affect my clinical practice in any way.”
Dr. Neri and colleagues described no outside funding for their research or financial conflicts of interest. Dr. Starling has received consulting fees from pharmaceutical manufacturers but reported no disclosures relevant to the study discussed. Dr. Loder reported no financial conflicts of interest.
, according to results from an observational study.
Migraine during pregnancy has been associated in previous studies with hypertensive pregnancy complications including preeclampsia; however, little is known about other headache types and their effects on pregnancy and birth outcomes.
For their research, published online July 20 in Cephalalgia, Isabella Neri, MD, PhD, and colleagues at Hospital Policlinico of Modena, Italy, looked at headache status for 515 consecutive pregnant women evaluated during their first trimester and followed through childbirth.
Altogether 224 women, or 43.5% of the cohort, were diagnosed with migraine without aura (n = 72), migraine with aura (n = 27), or tension-type headache (n = 125). The authors did not report on the severity or frequency of headaches.
Women with migraine with aura and tension-type headache saw higher rates of small-for-gestational-age infants (25.9% and 10.4% of births, respectively) compared with 5.5% for women without headache. Women presenting with tension-type headache saw elevated risk for small-for-gestational-age infants (odds ratio [OR] 4.19, P = .004) as did women with migraine with aura (OR 5.37, P = .02).
Admission to neonatal intensive care was significantly higher in all the headache groups. However, the authors found no statistically significant associations between headaches and any other perinatal outcome investigated in the study, including gestational diabetes, placental abruption, gestational hypertension, and preterm delivery.
A previous study conducted by the same research group had reported a relationship between migraine and gestational hypertension. The authors cited the small sample size of the migraine groups in the current study, “the diverse features of the population,” and the popularity of low-dose aspirin administration as potentially affecting that outcome.
Interpret findings with caution
Asked by this news organization to comment on the research, two headache neurologists praised Dr. Neri and colleagues’ research for focusing on an understudied topic – but also said that the results would not change their practice unless replicated in larger studies.
Elizabeth W. Loder, MD, MPH, chief emeritus of the division of headache at Brigham and Women’s Faulkner Hospital in Boston, urged caution in interpreting the findings, particularly with regard to tension-type headache. “This study adds to information suggesting that pregnancy complications probably are higher in women who have migraine with aura, and there’s biological plausibility for that,” Dr. Loder said. “Having aura means you may have some vascular abnormalities and things that logically might be associated with an increased risk of small-for-gestational age infants.” But the small size of the migraine-with-aura group in this study – 27 women – and the fact that other perinatal outcomes measured in the study did not reach significance, allows for the possibility that the small-for-gestational-age findings were due to chance, Dr. Loder noted.
With tension-type headache, a biological rationale for small-for-gestational-age risk is more elusive, Dr. Loder said. “I would want to see that association replicated in another study before I thought that I needed to warn women with tension-type headache about this potential outcome. There’s lot of uncertainty here about the magnitude of the risk.”
While Dr. Neri and colleagues described the instruments used in their study to diagnose migraine and migraine with aura, they did not explain how tension-type headache was diagnosed.
Tension-type headache, while common, is still not well characterized, Dr. Loder noted, and may represent a heterogeneous condition or the milder end of a biological continuum that includes migraine with aura. Also, the group in the study had a higher prevalence of smoking, and though the authors made statistical adjustments for smoking status, “smokers are systematically different than people who aren’t in other ways that could be associated with these outcomes,” Dr. Loder said.
While the authors of the study suggested that interventions might be indicated for women with tension-type headache in pregnancy, “showing an association doesn’t necessarily mean that intervening would make a difference” on pregnancy outcomes, Dr. Loder said.
Amaal J. Starling, MD, of the Mayo Clinic in Phoenix, Ariz., said in an interview that she, too, appreciated that this study looked at pregnancy outcomes in the setting of headache disorders. “Unfortunately even though headache disorders and especially migraine affect women so much, we still know very little about migraine in pregnancy,” she said.
Dr. Starling noted that many women with migraine are discouraged by their health care providers from becoming pregnant, because of the false belief that migraine cannot be managed in pregnancy. In her own practice, she said, she treats many patients with severe headache who become pregnant and who require pharmacological intervention during pregnancy.
This does not mean she regards headache in pregnancy as innocent. “I want patients to be on high alert for changes in headache symptoms in pregnancy. If someone has worsening of headache or migraine or aura in the setting of pregnancy, we consider that a red flag,” potentially indicating complications such as high blood pressure, gestational hypertension, or a blood clot.
Like Dr. Loder, Dr. Starling said she was not surprised by Dr. Neri and colleagues’ finding that migraine with aura might impact pregnancy outcomes. “We know that migraine with aura has a lot of vascular abnormalities that underlie the pathogenesis,” she said.
Dr. Starling found the findings related to tension-type headache less convincing, not least because the diagnostic criteria for tension-type headache was not made clear in the study. “I view this as an exploratory study that says maybe there’s a signal here. A larger epidemiological study would need to be done to confirm or refute this data,” Dr. Starling said. Until the findings can be replicated, “this study would not affect my clinical practice in any way.”
Dr. Neri and colleagues described no outside funding for their research or financial conflicts of interest. Dr. Starling has received consulting fees from pharmaceutical manufacturers but reported no disclosures relevant to the study discussed. Dr. Loder reported no financial conflicts of interest.
FROM CEPHALALGIA