User login
Estimating insulin resistance may help predict stroke, death in T2D
Calculating the estimated glucose disposal rate (eGDR) as a proxy for the level of insulin resistance may be useful way to determine if someone with type 2 diabetes (T2D) is at risk for having a first stroke, Swedish researchers have found.
In a large population-based study, the lower the eGDR score went, the higher the risk for having a first stroke became.
The eGDR score was also predictive of the chance of dying from any or a cardiovascular cause, Alexander Zabala, MD, reported at the annual meeting of the European Association for the Study of Diabetes (Abstract OP 01-4).
The link between insulin resistance and an increased risk for stroke has been known for some time, and not just in people with T2D. However, the current way of determining insulin resistance is not suitable for widespread practice.
“The goal standard technique for measuring insulin resistance is the euglycemic clamp method,” said Dr. Zabala, an internal medical resident at Södersjukhuset hospital and researcher at the Karolinska Institutet in Stockholm.
“For that reason, [the eGDR], a method based on readily available clinical factors – waist circumference, hypertension, and glycosylated hemoglobin was developed,” he explained. Body mass index can also be used in place of waist circumference, he qualified.
The eGDR has already been proven to be very precise in people with type 1 diabetes, said Dr. Zabala, and could be an “excellent tool to measure insulin resistance in a large patient population.”
Investigating the link between eGDR and first stroke risk
The aim of the study he presented was to see if changes in the eGDR were associated with changes in the risk of someone with T2D experiencing a first stroke, or dying from a cardiovascular or other cause.
An observational cohort was formed by first considering data on all adult patients with T2D who were logged in the Swedish National Diabetes Registry (NDR) during 2004-2016. Then anyone with a history of stroke, or with any missing data on the clinical variables needed to calculate the eGDR, were excluded.
This resulted in an overall population of 104,697 individuals, aged a mean of 63 years, who had developed T2D at around the age of 59 years. About 44% of the study population were women. The mean eGDR for the whole population was 5.6 mg/kg per min.
The study subjects were grouped according to four eGDR levels: 24,706 were in the lowest quartile of eGDR (less than 4 mg/kg per min), signifying the highest level of insulin resistance, and 18,762 were in the upper quartile of eGDR (greater than 8 mg/kg per min), signifying the lowest level of insulin resistance. The middle two groups had an eGDR between 4 and 6 mg/kg per min (40,187), and 6 and 8 mg/kg/min (21,042).
Data from the NDR were then combined with the Swedish Cause of Death register, the Swedish In-patient Care Diagnoses registry, and the Longitudinal Database for Health Insurance and Labour Market Studies (LISA) to determine the rates of stroke, ischemic stroke, hemorrhagic stroke, all-cause mortality, and cardiovascular mortality.
Increasing insulin resistance ups risk for stroke, death
After a median follow-up of 5.6 years, 4% (4,201) of the study population had had a stroke.
“We clearly see an increased occurrence of first-time stroke in the group with the lowest eGDR, indicating worst insulin resistance, in comparison with the group with the highest eGDR, indicating less insulin resistance,” Dr. Zabala reported.
After adjustment for potential confounding factors, including age at baseline, gender, diabetes duration, among other variables, the risk for stroke was lowest in those with a high eGDR value and highest for those with a low eGDR value.
Using individuals with the lowest eGDR (less than 4 mg/kg per min) and thus greatest risk of stroke as the reference, adjusted hazard ratios (aHR) for first-time stroke were: 0.60, 0.68, and 0.77 for those with an eGDR of greater than 8, 6-8, and 4-6 mg/kg per min, respectively.
The corresponding values for risk of ischemic stroke were 0.55, 0.68, and 0.75. Regarding hemorrhagic stroke, there was no statistically significant correlation between eGDR levels and stroke occurrence. This was due to the small number of cases recorded.
As for all-cause and cardiovascular mortality, a similar pattern was seen, with higher rates of death linked to increasing insulin resistance. Adjusted hazard ratios according to increasing insulin resistance (decreasing eGDR scores) for all-cause death were 0.68, 0.75, and 0.82 and for cardiovascular mortality were 0.65, 0.75, and 0.82.
A sensitivity analysis, using BMI instead of waist circumference to calculate the eGDR, showed a similar pattern, and “interestingly, a correlation between eGDR levels and risk of hemorrhagic stroke.” Dr. Zabala said.
Limitations and take-homes
Of course, this is an observational cohort study, so no conclusions on causality can be made and there are no data on the use of anti-diabetic treatments specifically. But there are strengths such as covering almost all adults with T2D in Sweden and a relatively long-follow-up time.
The findings suggest that “eGDR, which may reflect insulin resistance may be a useful risk marker for stroke and death in people with type 2 diabetes,” said Dr. Zabala.
“You had a very large cohort, and that certainly makes your results very valid,” observed Peter Novodvorsky, MUDr. (Hons), PhD, MRCP, a consultant diabetologist in Trenčín, Slovakia.
Dr. Novodvorsky, who chaired the session, picked up on the lack of information about how many people were taking newer diabetes drugs, such as the glucagon-like peptide 1 receptor antagonists and sodium glucose-lowering transport 2 inhibitors.
“As we all know, these might have protective effects which are not necessarily related to the glucose lowering or insulin resistance-lowering” effects, so could have influenced the results. In terms of how practical the eGDR is for clinical practice, Dr. Zabala observed in a press release: “eGDR could be used to help T2D patients better understand and manage their risk of stroke and death.
“It could also be of importance in research. In this era of personalized medicine, better stratification of type 2 diabetes patients will help optimize clinical trials and further vital research into treatment, diagnosis, care and prevention.”
The research was a collaboration between the Karolinska Institutet, Gothenburg University and the Swedish National Diabetes Registry. Dr. Zabala and coauthors reported having no conflicts of interest.
Calculating the estimated glucose disposal rate (eGDR) as a proxy for the level of insulin resistance may be useful way to determine if someone with type 2 diabetes (T2D) is at risk for having a first stroke, Swedish researchers have found.
In a large population-based study, the lower the eGDR score went, the higher the risk for having a first stroke became.
The eGDR score was also predictive of the chance of dying from any or a cardiovascular cause, Alexander Zabala, MD, reported at the annual meeting of the European Association for the Study of Diabetes (Abstract OP 01-4).
The link between insulin resistance and an increased risk for stroke has been known for some time, and not just in people with T2D. However, the current way of determining insulin resistance is not suitable for widespread practice.
“The goal standard technique for measuring insulin resistance is the euglycemic clamp method,” said Dr. Zabala, an internal medical resident at Södersjukhuset hospital and researcher at the Karolinska Institutet in Stockholm.
“For that reason, [the eGDR], a method based on readily available clinical factors – waist circumference, hypertension, and glycosylated hemoglobin was developed,” he explained. Body mass index can also be used in place of waist circumference, he qualified.
The eGDR has already been proven to be very precise in people with type 1 diabetes, said Dr. Zabala, and could be an “excellent tool to measure insulin resistance in a large patient population.”
Investigating the link between eGDR and first stroke risk
The aim of the study he presented was to see if changes in the eGDR were associated with changes in the risk of someone with T2D experiencing a first stroke, or dying from a cardiovascular or other cause.
An observational cohort was formed by first considering data on all adult patients with T2D who were logged in the Swedish National Diabetes Registry (NDR) during 2004-2016. Then anyone with a history of stroke, or with any missing data on the clinical variables needed to calculate the eGDR, were excluded.
This resulted in an overall population of 104,697 individuals, aged a mean of 63 years, who had developed T2D at around the age of 59 years. About 44% of the study population were women. The mean eGDR for the whole population was 5.6 mg/kg per min.
The study subjects were grouped according to four eGDR levels: 24,706 were in the lowest quartile of eGDR (less than 4 mg/kg per min), signifying the highest level of insulin resistance, and 18,762 were in the upper quartile of eGDR (greater than 8 mg/kg per min), signifying the lowest level of insulin resistance. The middle two groups had an eGDR between 4 and 6 mg/kg per min (40,187), and 6 and 8 mg/kg/min (21,042).
Data from the NDR were then combined with the Swedish Cause of Death register, the Swedish In-patient Care Diagnoses registry, and the Longitudinal Database for Health Insurance and Labour Market Studies (LISA) to determine the rates of stroke, ischemic stroke, hemorrhagic stroke, all-cause mortality, and cardiovascular mortality.
Increasing insulin resistance ups risk for stroke, death
After a median follow-up of 5.6 years, 4% (4,201) of the study population had had a stroke.
“We clearly see an increased occurrence of first-time stroke in the group with the lowest eGDR, indicating worst insulin resistance, in comparison with the group with the highest eGDR, indicating less insulin resistance,” Dr. Zabala reported.
After adjustment for potential confounding factors, including age at baseline, gender, diabetes duration, among other variables, the risk for stroke was lowest in those with a high eGDR value and highest for those with a low eGDR value.
Using individuals with the lowest eGDR (less than 4 mg/kg per min) and thus greatest risk of stroke as the reference, adjusted hazard ratios (aHR) for first-time stroke were: 0.60, 0.68, and 0.77 for those with an eGDR of greater than 8, 6-8, and 4-6 mg/kg per min, respectively.
The corresponding values for risk of ischemic stroke were 0.55, 0.68, and 0.75. Regarding hemorrhagic stroke, there was no statistically significant correlation between eGDR levels and stroke occurrence. This was due to the small number of cases recorded.
As for all-cause and cardiovascular mortality, a similar pattern was seen, with higher rates of death linked to increasing insulin resistance. Adjusted hazard ratios according to increasing insulin resistance (decreasing eGDR scores) for all-cause death were 0.68, 0.75, and 0.82 and for cardiovascular mortality were 0.65, 0.75, and 0.82.
A sensitivity analysis, using BMI instead of waist circumference to calculate the eGDR, showed a similar pattern, and “interestingly, a correlation between eGDR levels and risk of hemorrhagic stroke.” Dr. Zabala said.
Limitations and take-homes
Of course, this is an observational cohort study, so no conclusions on causality can be made and there are no data on the use of anti-diabetic treatments specifically. But there are strengths such as covering almost all adults with T2D in Sweden and a relatively long-follow-up time.
The findings suggest that “eGDR, which may reflect insulin resistance may be a useful risk marker for stroke and death in people with type 2 diabetes,” said Dr. Zabala.
“You had a very large cohort, and that certainly makes your results very valid,” observed Peter Novodvorsky, MUDr. (Hons), PhD, MRCP, a consultant diabetologist in Trenčín, Slovakia.
Dr. Novodvorsky, who chaired the session, picked up on the lack of information about how many people were taking newer diabetes drugs, such as the glucagon-like peptide 1 receptor antagonists and sodium glucose-lowering transport 2 inhibitors.
“As we all know, these might have protective effects which are not necessarily related to the glucose lowering or insulin resistance-lowering” effects, so could have influenced the results. In terms of how practical the eGDR is for clinical practice, Dr. Zabala observed in a press release: “eGDR could be used to help T2D patients better understand and manage their risk of stroke and death.
“It could also be of importance in research. In this era of personalized medicine, better stratification of type 2 diabetes patients will help optimize clinical trials and further vital research into treatment, diagnosis, care and prevention.”
The research was a collaboration between the Karolinska Institutet, Gothenburg University and the Swedish National Diabetes Registry. Dr. Zabala and coauthors reported having no conflicts of interest.
Calculating the estimated glucose disposal rate (eGDR) as a proxy for the level of insulin resistance may be useful way to determine if someone with type 2 diabetes (T2D) is at risk for having a first stroke, Swedish researchers have found.
In a large population-based study, the lower the eGDR score went, the higher the risk for having a first stroke became.
The eGDR score was also predictive of the chance of dying from any or a cardiovascular cause, Alexander Zabala, MD, reported at the annual meeting of the European Association for the Study of Diabetes (Abstract OP 01-4).
The link between insulin resistance and an increased risk for stroke has been known for some time, and not just in people with T2D. However, the current way of determining insulin resistance is not suitable for widespread practice.
“The goal standard technique for measuring insulin resistance is the euglycemic clamp method,” said Dr. Zabala, an internal medical resident at Södersjukhuset hospital and researcher at the Karolinska Institutet in Stockholm.
“For that reason, [the eGDR], a method based on readily available clinical factors – waist circumference, hypertension, and glycosylated hemoglobin was developed,” he explained. Body mass index can also be used in place of waist circumference, he qualified.
The eGDR has already been proven to be very precise in people with type 1 diabetes, said Dr. Zabala, and could be an “excellent tool to measure insulin resistance in a large patient population.”
Investigating the link between eGDR and first stroke risk
The aim of the study he presented was to see if changes in the eGDR were associated with changes in the risk of someone with T2D experiencing a first stroke, or dying from a cardiovascular or other cause.
An observational cohort was formed by first considering data on all adult patients with T2D who were logged in the Swedish National Diabetes Registry (NDR) during 2004-2016. Then anyone with a history of stroke, or with any missing data on the clinical variables needed to calculate the eGDR, were excluded.
This resulted in an overall population of 104,697 individuals, aged a mean of 63 years, who had developed T2D at around the age of 59 years. About 44% of the study population were women. The mean eGDR for the whole population was 5.6 mg/kg per min.
The study subjects were grouped according to four eGDR levels: 24,706 were in the lowest quartile of eGDR (less than 4 mg/kg per min), signifying the highest level of insulin resistance, and 18,762 were in the upper quartile of eGDR (greater than 8 mg/kg per min), signifying the lowest level of insulin resistance. The middle two groups had an eGDR between 4 and 6 mg/kg per min (40,187), and 6 and 8 mg/kg/min (21,042).
Data from the NDR were then combined with the Swedish Cause of Death register, the Swedish In-patient Care Diagnoses registry, and the Longitudinal Database for Health Insurance and Labour Market Studies (LISA) to determine the rates of stroke, ischemic stroke, hemorrhagic stroke, all-cause mortality, and cardiovascular mortality.
Increasing insulin resistance ups risk for stroke, death
After a median follow-up of 5.6 years, 4% (4,201) of the study population had had a stroke.
“We clearly see an increased occurrence of first-time stroke in the group with the lowest eGDR, indicating worst insulin resistance, in comparison with the group with the highest eGDR, indicating less insulin resistance,” Dr. Zabala reported.
After adjustment for potential confounding factors, including age at baseline, gender, diabetes duration, among other variables, the risk for stroke was lowest in those with a high eGDR value and highest for those with a low eGDR value.
Using individuals with the lowest eGDR (less than 4 mg/kg per min) and thus greatest risk of stroke as the reference, adjusted hazard ratios (aHR) for first-time stroke were: 0.60, 0.68, and 0.77 for those with an eGDR of greater than 8, 6-8, and 4-6 mg/kg per min, respectively.
The corresponding values for risk of ischemic stroke were 0.55, 0.68, and 0.75. Regarding hemorrhagic stroke, there was no statistically significant correlation between eGDR levels and stroke occurrence. This was due to the small number of cases recorded.
As for all-cause and cardiovascular mortality, a similar pattern was seen, with higher rates of death linked to increasing insulin resistance. Adjusted hazard ratios according to increasing insulin resistance (decreasing eGDR scores) for all-cause death were 0.68, 0.75, and 0.82 and for cardiovascular mortality were 0.65, 0.75, and 0.82.
A sensitivity analysis, using BMI instead of waist circumference to calculate the eGDR, showed a similar pattern, and “interestingly, a correlation between eGDR levels and risk of hemorrhagic stroke.” Dr. Zabala said.
Limitations and take-homes
Of course, this is an observational cohort study, so no conclusions on causality can be made and there are no data on the use of anti-diabetic treatments specifically. But there are strengths such as covering almost all adults with T2D in Sweden and a relatively long-follow-up time.
The findings suggest that “eGDR, which may reflect insulin resistance may be a useful risk marker for stroke and death in people with type 2 diabetes,” said Dr. Zabala.
“You had a very large cohort, and that certainly makes your results very valid,” observed Peter Novodvorsky, MUDr. (Hons), PhD, MRCP, a consultant diabetologist in Trenčín, Slovakia.
Dr. Novodvorsky, who chaired the session, picked up on the lack of information about how many people were taking newer diabetes drugs, such as the glucagon-like peptide 1 receptor antagonists and sodium glucose-lowering transport 2 inhibitors.
“As we all know, these might have protective effects which are not necessarily related to the glucose lowering or insulin resistance-lowering” effects, so could have influenced the results. In terms of how practical the eGDR is for clinical practice, Dr. Zabala observed in a press release: “eGDR could be used to help T2D patients better understand and manage their risk of stroke and death.
“It could also be of importance in research. In this era of personalized medicine, better stratification of type 2 diabetes patients will help optimize clinical trials and further vital research into treatment, diagnosis, care and prevention.”
The research was a collaboration between the Karolinska Institutet, Gothenburg University and the Swedish National Diabetes Registry. Dr. Zabala and coauthors reported having no conflicts of interest.
FROM EASD 2021
DIY nerve stimulation effective in episodic migraine
results from a phase 3 study show.
This is great news for headache patients who want to explore nondrug treatment options, said study investigator Deena E. Kuruvilla, MD, neurologist and headache specialist at the Westport Headache Institute, Connecticut.
She added that such devices “aren’t always part of the conversation when we’re discussing preventive and acute treatments with our patients. Making this a regular part of the conversation might be helpful to patients.”
The findings were presented at ANA 2021: 146th Annual Meeting of the American Neurological Association (ANA), which was held online.
A key therapeutic target
The randomized, double-blind trial compared E-TNS with sham stimulation for the acute treatment of migraine.
The E-TNS device (Verum Cefaly Abortive Program) stimulates the supraorbital nerve in the forehead. “This nerve is a branch of the trigeminal nerve, which is thought to be the key player in migraine pathophysiology,” Dr. Kuruvilla noted.
The device has been cleared by the U.S. Food and Drug Administration for acute and preventive treatment of migraine.
During a run-in period before randomization, patients were asked to keep a detailed headache diary and to become comfortable using the trial device to treat an acute migraine attack at home.
The study enrolled 538 adult patients at 10 centers. The patients were aged 18 to 65 years, and they had been having episodic migraines, with or without aura, for at least a year. The participants had to have received a migraine diagnosis before age 50, and they had to be experiencing an attack of migraine 2 to 8 days per month.
The patients used the device only for a migraine of at least moderate intensity that was accompanied by at least one migraine-associated symptom, such as photophobia, phonophobia, or nausea. They were asked not to take rescue medication prior to or during a therapy session.
Study participants applied either neurostimulation or sham stimulation for a continuous 2-hour period within 4 hours of a migraine attack over the 2-month study period.
The two primary endpoints were pain freedom and freedom from the most bothersome migraine-associated symptoms at 2 hours.
Compared to sham treatment, active stimulation was more effective in achieving pain freedom (P = .043) and freedom from the most bothersome migraine-associated symptom (P = .001) at 2 hours.
“So the study did meet both primary endpoints with statistical significance,” said Dr. Kuruvilla.
The five secondary endpoints included pain relief at 2 hours; absence of all migraine-associated symptoms at 2 hours; use of rescue medication within 24 hours; sustained pain freedom at 24 hours; and sustained pain relief at 24 hours.
All but one of these endpoints reached statistical significance, showing superiority for the active intervention. The only exception was in regard to use of rescue medication.
The most common adverse event (AE) was forehead paresthesia, discomfort, or burning, which was more common in the active-treatment group than in the sham-treatment group (P = .009). There were four cases of nausea or vomiting in the active-treatment group and none in the sham-treatment group. There were no serious AEs.
Available over the counter
Both moderators of the headache poster tour that featured this study – Justin C. McArthur, MBBS, from Johns Hopkins University, Baltimore, and Steven Galetta, MD, from NYU Grossman School of Medicine – praised the presentation.
Dr. Galetta questioned whether patients were receiving preventive therapies. Dr. Kuruvilla said that the patients were allowed to enter the trial while taking preventive therapies, including antiepileptic treatments, blood pressure medications, and antidepressants, but that they had to be receiving stable doses.
The investigators didn’t distinguish between participants who were taking preventive therapies and those who weren’t, she said. “The aim was really to look at acute treatment for migraine,” and patients taking such medication “had been stable on their regimen for a pretty prolonged period of time.”
Dr. McArthur asked about the origin of the nausea some patients experienced.
It was difficult to determine whether the nausea was an aspect of an individual patient’s migraine attack or was an effect of the stimulation, said Dr. Kuruvilla. She noted that some patients found the vibrating sensation from the device uncomfortable and that nausea could be associated with pain at the site.
The device costs $300 to $400 (U.S.) and is available over the counter.
Dr. Kuruvilla is a consultant for Cefaly, Neurolief, Theranica, Now What Media, and Kx Advisors. She is on the speakers bureau for AbbVie/Allergan, Amgen/Novartis, Lilly, the American Headache Society, Biohaven, and CME meeting, and she is on an advisory board at AbbVie/Allergan, Lilly, Theranica, and Amgen/Novartis. She is editor and associate editor of Healthline and is an author for WebMD/Medscape, Healthline.
A version of this article first appeared on Medscape.com.
results from a phase 3 study show.
This is great news for headache patients who want to explore nondrug treatment options, said study investigator Deena E. Kuruvilla, MD, neurologist and headache specialist at the Westport Headache Institute, Connecticut.
She added that such devices “aren’t always part of the conversation when we’re discussing preventive and acute treatments with our patients. Making this a regular part of the conversation might be helpful to patients.”
The findings were presented at ANA 2021: 146th Annual Meeting of the American Neurological Association (ANA), which was held online.
A key therapeutic target
The randomized, double-blind trial compared E-TNS with sham stimulation for the acute treatment of migraine.
The E-TNS device (Verum Cefaly Abortive Program) stimulates the supraorbital nerve in the forehead. “This nerve is a branch of the trigeminal nerve, which is thought to be the key player in migraine pathophysiology,” Dr. Kuruvilla noted.
The device has been cleared by the U.S. Food and Drug Administration for acute and preventive treatment of migraine.
During a run-in period before randomization, patients were asked to keep a detailed headache diary and to become comfortable using the trial device to treat an acute migraine attack at home.
The study enrolled 538 adult patients at 10 centers. The patients were aged 18 to 65 years, and they had been having episodic migraines, with or without aura, for at least a year. The participants had to have received a migraine diagnosis before age 50, and they had to be experiencing an attack of migraine 2 to 8 days per month.
The patients used the device only for a migraine of at least moderate intensity that was accompanied by at least one migraine-associated symptom, such as photophobia, phonophobia, or nausea. They were asked not to take rescue medication prior to or during a therapy session.
Study participants applied either neurostimulation or sham stimulation for a continuous 2-hour period within 4 hours of a migraine attack over the 2-month study period.
The two primary endpoints were pain freedom and freedom from the most bothersome migraine-associated symptoms at 2 hours.
Compared to sham treatment, active stimulation was more effective in achieving pain freedom (P = .043) and freedom from the most bothersome migraine-associated symptom (P = .001) at 2 hours.
“So the study did meet both primary endpoints with statistical significance,” said Dr. Kuruvilla.
The five secondary endpoints included pain relief at 2 hours; absence of all migraine-associated symptoms at 2 hours; use of rescue medication within 24 hours; sustained pain freedom at 24 hours; and sustained pain relief at 24 hours.
All but one of these endpoints reached statistical significance, showing superiority for the active intervention. The only exception was in regard to use of rescue medication.
The most common adverse event (AE) was forehead paresthesia, discomfort, or burning, which was more common in the active-treatment group than in the sham-treatment group (P = .009). There were four cases of nausea or vomiting in the active-treatment group and none in the sham-treatment group. There were no serious AEs.
Available over the counter
Both moderators of the headache poster tour that featured this study – Justin C. McArthur, MBBS, from Johns Hopkins University, Baltimore, and Steven Galetta, MD, from NYU Grossman School of Medicine – praised the presentation.
Dr. Galetta questioned whether patients were receiving preventive therapies. Dr. Kuruvilla said that the patients were allowed to enter the trial while taking preventive therapies, including antiepileptic treatments, blood pressure medications, and antidepressants, but that they had to be receiving stable doses.
The investigators didn’t distinguish between participants who were taking preventive therapies and those who weren’t, she said. “The aim was really to look at acute treatment for migraine,” and patients taking such medication “had been stable on their regimen for a pretty prolonged period of time.”
Dr. McArthur asked about the origin of the nausea some patients experienced.
It was difficult to determine whether the nausea was an aspect of an individual patient’s migraine attack or was an effect of the stimulation, said Dr. Kuruvilla. She noted that some patients found the vibrating sensation from the device uncomfortable and that nausea could be associated with pain at the site.
The device costs $300 to $400 (U.S.) and is available over the counter.
Dr. Kuruvilla is a consultant for Cefaly, Neurolief, Theranica, Now What Media, and Kx Advisors. She is on the speakers bureau for AbbVie/Allergan, Amgen/Novartis, Lilly, the American Headache Society, Biohaven, and CME meeting, and she is on an advisory board at AbbVie/Allergan, Lilly, Theranica, and Amgen/Novartis. She is editor and associate editor of Healthline and is an author for WebMD/Medscape, Healthline.
A version of this article first appeared on Medscape.com.
results from a phase 3 study show.
This is great news for headache patients who want to explore nondrug treatment options, said study investigator Deena E. Kuruvilla, MD, neurologist and headache specialist at the Westport Headache Institute, Connecticut.
She added that such devices “aren’t always part of the conversation when we’re discussing preventive and acute treatments with our patients. Making this a regular part of the conversation might be helpful to patients.”
The findings were presented at ANA 2021: 146th Annual Meeting of the American Neurological Association (ANA), which was held online.
A key therapeutic target
The randomized, double-blind trial compared E-TNS with sham stimulation for the acute treatment of migraine.
The E-TNS device (Verum Cefaly Abortive Program) stimulates the supraorbital nerve in the forehead. “This nerve is a branch of the trigeminal nerve, which is thought to be the key player in migraine pathophysiology,” Dr. Kuruvilla noted.
The device has been cleared by the U.S. Food and Drug Administration for acute and preventive treatment of migraine.
During a run-in period before randomization, patients were asked to keep a detailed headache diary and to become comfortable using the trial device to treat an acute migraine attack at home.
The study enrolled 538 adult patients at 10 centers. The patients were aged 18 to 65 years, and they had been having episodic migraines, with or without aura, for at least a year. The participants had to have received a migraine diagnosis before age 50, and they had to be experiencing an attack of migraine 2 to 8 days per month.
The patients used the device only for a migraine of at least moderate intensity that was accompanied by at least one migraine-associated symptom, such as photophobia, phonophobia, or nausea. They were asked not to take rescue medication prior to or during a therapy session.
Study participants applied either neurostimulation or sham stimulation for a continuous 2-hour period within 4 hours of a migraine attack over the 2-month study period.
The two primary endpoints were pain freedom and freedom from the most bothersome migraine-associated symptoms at 2 hours.
Compared to sham treatment, active stimulation was more effective in achieving pain freedom (P = .043) and freedom from the most bothersome migraine-associated symptom (P = .001) at 2 hours.
“So the study did meet both primary endpoints with statistical significance,” said Dr. Kuruvilla.
The five secondary endpoints included pain relief at 2 hours; absence of all migraine-associated symptoms at 2 hours; use of rescue medication within 24 hours; sustained pain freedom at 24 hours; and sustained pain relief at 24 hours.
All but one of these endpoints reached statistical significance, showing superiority for the active intervention. The only exception was in regard to use of rescue medication.
The most common adverse event (AE) was forehead paresthesia, discomfort, or burning, which was more common in the active-treatment group than in the sham-treatment group (P = .009). There were four cases of nausea or vomiting in the active-treatment group and none in the sham-treatment group. There were no serious AEs.
Available over the counter
Both moderators of the headache poster tour that featured this study – Justin C. McArthur, MBBS, from Johns Hopkins University, Baltimore, and Steven Galetta, MD, from NYU Grossman School of Medicine – praised the presentation.
Dr. Galetta questioned whether patients were receiving preventive therapies. Dr. Kuruvilla said that the patients were allowed to enter the trial while taking preventive therapies, including antiepileptic treatments, blood pressure medications, and antidepressants, but that they had to be receiving stable doses.
The investigators didn’t distinguish between participants who were taking preventive therapies and those who weren’t, she said. “The aim was really to look at acute treatment for migraine,” and patients taking such medication “had been stable on their regimen for a pretty prolonged period of time.”
Dr. McArthur asked about the origin of the nausea some patients experienced.
It was difficult to determine whether the nausea was an aspect of an individual patient’s migraine attack or was an effect of the stimulation, said Dr. Kuruvilla. She noted that some patients found the vibrating sensation from the device uncomfortable and that nausea could be associated with pain at the site.
The device costs $300 to $400 (U.S.) and is available over the counter.
Dr. Kuruvilla is a consultant for Cefaly, Neurolief, Theranica, Now What Media, and Kx Advisors. She is on the speakers bureau for AbbVie/Allergan, Amgen/Novartis, Lilly, the American Headache Society, Biohaven, and CME meeting, and she is on an advisory board at AbbVie/Allergan, Lilly, Theranica, and Amgen/Novartis. She is editor and associate editor of Healthline and is an author for WebMD/Medscape, Healthline.
A version of this article first appeared on Medscape.com.
FROM ANA
FDA approves combo pill for severe, acute pain
enough to require an opioid analgesic and for which alternative treatments fail to provide adequate pain relief.
Celecoxib is a nonsteroidal anti-inflammatory drug and tramadol is an opioid agonist. Seglentis contains 56 mg of celecoxib and 44 mg of tramadol.
“The unique co-crystal formulation of Seglentis provides effective pain relief via a multimodal approach,” Craig A. Sponseller, MD, chief medical officer of Kowa Pharmaceuticals America, said in a news release.
Esteve Pharmaceuticals has entered into an agreement with Kowa Pharmaceuticals America to commercialize the pain medicine in the United States, with a launch planned for early 2022.
“Seglentis uses four different and complementary mechanisms of analgesia and offers healthcare providers an important option to treat acute pain in adults that is severe enough to require opioid treatment and for which alternative treatments are inadequate,” Dr. Sponseller said.
Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses, the FDA will require a Risk Evaluation and Mitigation Strategy (REMS) for Seglentis.
The label states that the drug should be initiated as two tablets every 12 hours as needed and should be prescribed for the shortest duration consistent with individual patient treatment goals.
Patients should be monitored for respiratory depression, especially within the first 24 to 72 hours of initiating therapy with Seglentis.
Prescribers should discuss naloxone (Narcan) with patients and consider prescribing the opioid antagonist naloxone based on the patient’s risk factors for overdose.
Full prescribing information is available online.
A version of this article was first published on Medscape.com.
enough to require an opioid analgesic and for which alternative treatments fail to provide adequate pain relief.
Celecoxib is a nonsteroidal anti-inflammatory drug and tramadol is an opioid agonist. Seglentis contains 56 mg of celecoxib and 44 mg of tramadol.
“The unique co-crystal formulation of Seglentis provides effective pain relief via a multimodal approach,” Craig A. Sponseller, MD, chief medical officer of Kowa Pharmaceuticals America, said in a news release.
Esteve Pharmaceuticals has entered into an agreement with Kowa Pharmaceuticals America to commercialize the pain medicine in the United States, with a launch planned for early 2022.
“Seglentis uses four different and complementary mechanisms of analgesia and offers healthcare providers an important option to treat acute pain in adults that is severe enough to require opioid treatment and for which alternative treatments are inadequate,” Dr. Sponseller said.
Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses, the FDA will require a Risk Evaluation and Mitigation Strategy (REMS) for Seglentis.
The label states that the drug should be initiated as two tablets every 12 hours as needed and should be prescribed for the shortest duration consistent with individual patient treatment goals.
Patients should be monitored for respiratory depression, especially within the first 24 to 72 hours of initiating therapy with Seglentis.
Prescribers should discuss naloxone (Narcan) with patients and consider prescribing the opioid antagonist naloxone based on the patient’s risk factors for overdose.
Full prescribing information is available online.
A version of this article was first published on Medscape.com.
enough to require an opioid analgesic and for which alternative treatments fail to provide adequate pain relief.
Celecoxib is a nonsteroidal anti-inflammatory drug and tramadol is an opioid agonist. Seglentis contains 56 mg of celecoxib and 44 mg of tramadol.
“The unique co-crystal formulation of Seglentis provides effective pain relief via a multimodal approach,” Craig A. Sponseller, MD, chief medical officer of Kowa Pharmaceuticals America, said in a news release.
Esteve Pharmaceuticals has entered into an agreement with Kowa Pharmaceuticals America to commercialize the pain medicine in the United States, with a launch planned for early 2022.
“Seglentis uses four different and complementary mechanisms of analgesia and offers healthcare providers an important option to treat acute pain in adults that is severe enough to require opioid treatment and for which alternative treatments are inadequate,” Dr. Sponseller said.
Because of the risks of addiction, abuse, and misuse with opioids, even at recommended doses, the FDA will require a Risk Evaluation and Mitigation Strategy (REMS) for Seglentis.
The label states that the drug should be initiated as two tablets every 12 hours as needed and should be prescribed for the shortest duration consistent with individual patient treatment goals.
Patients should be monitored for respiratory depression, especially within the first 24 to 72 hours of initiating therapy with Seglentis.
Prescribers should discuss naloxone (Narcan) with patients and consider prescribing the opioid antagonist naloxone based on the patient’s risk factors for overdose.
Full prescribing information is available online.
A version of this article was first published on Medscape.com.
Art therapy linked to slowed Parkinson’s progression
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Adding art therapy to standard drug treatment in Parkinson’s disease (PD) not only improves severity of both motor and nonmotor symptoms, but also slows rates of disease progression, new research suggests.
Fifty PD patients were randomly assigned to receive either art therapy, including sculpting and drawing, plus drug therapy or drug therapy alone, and followed up over 12 months.
Patients receiving combined therapy experienced improvements in symptoms, depression, and cognitive scores, and had reduced tremor and daytime sleepiness. They were also substantially less likely to experience disease progression.
“The use of art therapy can reduce the severity of motor and nonmotor manifestations of Parkinson’s disease,” said study investigator Iryna Khubetova, MD, PhD, head of the neurology department, Odessa (Ukraine) Regional Clinical Hospital.
she added.
The findings were presented at the virtual congress of the European College of Neuropsychopharmacology.
A promising approach
Dr. Khubetova told this news organization that offering art therapy to PD patients was “very affordable,” especially as professional artists “provided materials for painting and other art supplies free of charge.”
“We hope this approach is very promising and would be widely adopted.”
She suggested the positive effect of art therapy could be related to “activating the brain’s reward neural network.”
This may be via improved visual attention acting on visuospatial mechanisms and emotional drive, with “activation of the medial orbitofrontal cortex, ventral striatum, and other structures.”
The researchers note PD, a “multisystem progressive neurodegenerative disease,” is among the three most common neurological disorders, with an incidence of 100-150 cases per 100,000 people.
They also note that nonpharmacologic approaches are “widely used” as an adjunct to drug therapy and as part of an “integrated approach” to disease management.
To examine the clinical efficacy of art therapy, the team recruited patients with PD who had preserved facility for independent movement, defined as stages 1-2.5 on the Hoehn and Yahr scale.
Patients were randomly assigned to art therapy sessions alongside standard drug therapy or to standard drug therapy alone. The art therapy included sculpting, free drawing, and coloring patterns.
Multiple benefits
Participants were assessed at baseline and at 6 and 12 months with the Unified Parkinson Disease Rating Scale (UPDRS), the Beck Depression Inventory, the Montreal Cognitive Assessment, and the Pegboard Test of finger dexterity.
Fifty patients were included in the study, with 30 assigned to standard drug therapy alone and 20 to the combined intervention. Participants had a mean age of 57.8 years, and 46% were women.
Over the study period, investigators found patients assigned to art therapy plus drug treatment had improved mood, as well as decreased daytime sleeping, reduced tremor, and a decrease in anxiety and fear intensity.
Between baseline and the 6- and 12-month assessments, patients in the combined therapy group showed improvements in scores on all of the questionnaires, and on the Pegboard Test. In contrast, scores were either stable or worsened in the standard drug therapy–alone group.
The team notes that there was also a marked difference in rates of disease progression, defined as a change on the Hoehn and Yahr scale of at least 0.5 points, between the two groups.
Only two (10%) patients in the combined drug and art therapy progressed over the study period, compared with 10 (33%) in the control group (P = .05).
The findings complement those of a recent study conducted by Alberto Cucca, MD, of the Fresco Institute for Parkinson’s and Movement Disorders, New York University, and colleagues.
Eighteen patients took part in the prospective, open-label trial. They were assessed before and after 20 sessions of art therapy on a range of measures.
Results revealed that following the art therapy, patients had improvements in the Navon Test (which assesses visual neglect, eye tracking, and UPDRS scores), as well as significantly increased functional connectivity levels in the visual cortex on resting-state functional MRI.
Many benefits, no side effects
Rebecca Gilbert, MD, PhD, vice president and chief scientific officer of the American Parkinson Disease Association, who was not involved in either study, told this news organization that the idea of art therapy for patients with Parkinson’s is “very reasonable.”
She highlighted that “people with Parkinson’s have many issues with their visuospatial abilities,” as well as their depth and distance perception, and so “enhancing that aspect could potentially be very beneficial.”
“So I’m hopeful that it’s a really good avenue to explore, and the preliminary data are very exciting.”
Dr. Gilbert also highlighted that the “wonderful” aspect of art therapy is that there are “so many benefits and not really any side effects.” Patients can “take the meds … and then enhance that with various therapies, and this would be an additional option.”
Another notable aspect of art therapy is the “social element” and the sense of “camaraderie,” although that has “to be teased out from the benefits you would get from the actual art therapy.”
Finally, Dr. Gilbert pointed out that the difference between the current trial and Dr. Cucca’s trial is the presence of a control group.
“Of course, it’s not blinded, because you know whether you got therapy or not … but that extra element of being able to compare with a group that didn’t get the treatment gives it a little more weight in terms of the field.”
No funding was declared. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ECNP 2021
New antimigraine drugs linked with less risk for adverse events
“[T]he lack of cardiovascular risks of these new classes of migraine-specific treatments may provide alternative treatment options for individuals for whom currently available acute treatments have failed or for those with cardiovascular contraindications,” write lead author Chun-Pai Yang, MD, PhD, of Taichung (Taiwan) Veterans General Hospital and colleagues, in the paper, published online in JAMA Network Open.
Methods
The new study compared the outcomes for acute migraine management using the ditan, lasmiditan (a 5-hydroxytryptamine [5HT]1F–receptor agonist), and the two gepants, rimegepant, and ubrogepant (calcitonin gene–related peptide [CGRP] antagonists), with standard triptan (selective 5-HT1B/1D–receptor agonist) therapy.
The researchers evaluated 64 double-blind randomized clinical trials which included 46,442 patients, the majority of whom (74%-87%) were women with an age range of 36-43 years.
The primary outcome evaluated was the odds ratio for freedom from pain at 2 hours after a single dose and secondary outcomes were the OR for pain relief at 2 hours following a dose, as well as any adverse events.
Results
Dr. Yang and colleagues found that virtually all medications with widespread clinical use, regardless of class, were associated with higher ORs for pain freedom when compared with placebo.
Compared to ditan and gepant agents, however, triptans were associated with significantly higher ORs for pain freedom. The odds ratio ranges were 1.72-3.40 for lasmiditan, 1.58-3.13 for rimegepant, and 1.54-3.05 for ubrogepant.
With respect to pain relief at 2 hours, while all medications were more effective than placebo, triptans were associated with higher ORs when compared with the other drug classes: lasmiditan (range: OR, 1.46; 95% confidence interval, 1.09-1.96 to OR, 3.31; 95% CI, 2.41-4.55), rimegepant (range: OR, 1.33; 95% CI, 1.01-1.76 to OR, 3.01; 95% CI, 2.33-3.88), and ubrogepant (range: OR, 1.38; 95% CI, 1.02-1.88 to OR, 3.13; 95% CI, 2.35-4.15)
When assessing tolerability, the researchers found that overall, triptans were associated with the higher ORs for any adverse events (AE) with a trend of dose-response relationship. Lasmiditan (in the ditan class) was associated with the highest risk for AEs among all treatments. Most of the AEs were mild to moderate and included chest pain, tightness, heaviness, and pressure.
Dr. Yang and colleagues note that, “although these two new classes of antimigraine drugs may not be as efficacious as triptans, these novel abortive agents without cardiovascular risks might offer an alternative to current specific migraine treatments for patients at risk of cardiovascular disease.”
Balancing efficacy and tolerability
“When choosing an acute medication for a patient there is always a balance between efficacy and tolerability,” headache specialist and associate director of North Shore Headache and Spine Lauren Natbony, MD, said in an interview.
“A medication can only be effective if a patient is able to tolerate it and will actually use it,” Dr. Natbony said.
With respect to the current review, Dr. Natbony pointed out, “response to acute therapy can differ between migraine attacks and may be based on variables not controlled for, such as how early in an attack the medication was taken, associated symptoms such as nausea that may make oral medications less efficacious, etc.”
The authors acknowledge that the focus on short-term responses and AEs after a single dose is a limitation of the study. They also pointed out what they considered to be a strength of the study, which was its network meta-analysis design. According to the authors, this design allowed for “multiple direct and indirect comparisons, ranking the efficacy and safety of individual pharmacologic interventions and providing more precise estimates than those of RCTs and traditional meta-analysis.”
Funding for this study was provided through grants from the Ministry of Science and Technology, Taiwan; the Brain Research Center; and National Yang Ming Chiao Tung University.
Dr. Yang has received personal fees and grants from various pharmaceutical companies. He has also received grants from the Taiwan Ministry of Technology and Science, the Brain Research Center, National Yang Ming Chiao Tung University, and Taipei Veterans General Hospital outside the submitted work. The other authors and Dr. Natbony disclosed no relevant financial relationships.
“[T]he lack of cardiovascular risks of these new classes of migraine-specific treatments may provide alternative treatment options for individuals for whom currently available acute treatments have failed or for those with cardiovascular contraindications,” write lead author Chun-Pai Yang, MD, PhD, of Taichung (Taiwan) Veterans General Hospital and colleagues, in the paper, published online in JAMA Network Open.
Methods
The new study compared the outcomes for acute migraine management using the ditan, lasmiditan (a 5-hydroxytryptamine [5HT]1F–receptor agonist), and the two gepants, rimegepant, and ubrogepant (calcitonin gene–related peptide [CGRP] antagonists), with standard triptan (selective 5-HT1B/1D–receptor agonist) therapy.
The researchers evaluated 64 double-blind randomized clinical trials which included 46,442 patients, the majority of whom (74%-87%) were women with an age range of 36-43 years.
The primary outcome evaluated was the odds ratio for freedom from pain at 2 hours after a single dose and secondary outcomes were the OR for pain relief at 2 hours following a dose, as well as any adverse events.
Results
Dr. Yang and colleagues found that virtually all medications with widespread clinical use, regardless of class, were associated with higher ORs for pain freedom when compared with placebo.
Compared to ditan and gepant agents, however, triptans were associated with significantly higher ORs for pain freedom. The odds ratio ranges were 1.72-3.40 for lasmiditan, 1.58-3.13 for rimegepant, and 1.54-3.05 for ubrogepant.
With respect to pain relief at 2 hours, while all medications were more effective than placebo, triptans were associated with higher ORs when compared with the other drug classes: lasmiditan (range: OR, 1.46; 95% confidence interval, 1.09-1.96 to OR, 3.31; 95% CI, 2.41-4.55), rimegepant (range: OR, 1.33; 95% CI, 1.01-1.76 to OR, 3.01; 95% CI, 2.33-3.88), and ubrogepant (range: OR, 1.38; 95% CI, 1.02-1.88 to OR, 3.13; 95% CI, 2.35-4.15)
When assessing tolerability, the researchers found that overall, triptans were associated with the higher ORs for any adverse events (AE) with a trend of dose-response relationship. Lasmiditan (in the ditan class) was associated with the highest risk for AEs among all treatments. Most of the AEs were mild to moderate and included chest pain, tightness, heaviness, and pressure.
Dr. Yang and colleagues note that, “although these two new classes of antimigraine drugs may not be as efficacious as triptans, these novel abortive agents without cardiovascular risks might offer an alternative to current specific migraine treatments for patients at risk of cardiovascular disease.”
Balancing efficacy and tolerability
“When choosing an acute medication for a patient there is always a balance between efficacy and tolerability,” headache specialist and associate director of North Shore Headache and Spine Lauren Natbony, MD, said in an interview.
“A medication can only be effective if a patient is able to tolerate it and will actually use it,” Dr. Natbony said.
With respect to the current review, Dr. Natbony pointed out, “response to acute therapy can differ between migraine attacks and may be based on variables not controlled for, such as how early in an attack the medication was taken, associated symptoms such as nausea that may make oral medications less efficacious, etc.”
The authors acknowledge that the focus on short-term responses and AEs after a single dose is a limitation of the study. They also pointed out what they considered to be a strength of the study, which was its network meta-analysis design. According to the authors, this design allowed for “multiple direct and indirect comparisons, ranking the efficacy and safety of individual pharmacologic interventions and providing more precise estimates than those of RCTs and traditional meta-analysis.”
Funding for this study was provided through grants from the Ministry of Science and Technology, Taiwan; the Brain Research Center; and National Yang Ming Chiao Tung University.
Dr. Yang has received personal fees and grants from various pharmaceutical companies. He has also received grants from the Taiwan Ministry of Technology and Science, the Brain Research Center, National Yang Ming Chiao Tung University, and Taipei Veterans General Hospital outside the submitted work. The other authors and Dr. Natbony disclosed no relevant financial relationships.
“[T]he lack of cardiovascular risks of these new classes of migraine-specific treatments may provide alternative treatment options for individuals for whom currently available acute treatments have failed or for those with cardiovascular contraindications,” write lead author Chun-Pai Yang, MD, PhD, of Taichung (Taiwan) Veterans General Hospital and colleagues, in the paper, published online in JAMA Network Open.
Methods
The new study compared the outcomes for acute migraine management using the ditan, lasmiditan (a 5-hydroxytryptamine [5HT]1F–receptor agonist), and the two gepants, rimegepant, and ubrogepant (calcitonin gene–related peptide [CGRP] antagonists), with standard triptan (selective 5-HT1B/1D–receptor agonist) therapy.
The researchers evaluated 64 double-blind randomized clinical trials which included 46,442 patients, the majority of whom (74%-87%) were women with an age range of 36-43 years.
The primary outcome evaluated was the odds ratio for freedom from pain at 2 hours after a single dose and secondary outcomes were the OR for pain relief at 2 hours following a dose, as well as any adverse events.
Results
Dr. Yang and colleagues found that virtually all medications with widespread clinical use, regardless of class, were associated with higher ORs for pain freedom when compared with placebo.
Compared to ditan and gepant agents, however, triptans were associated with significantly higher ORs for pain freedom. The odds ratio ranges were 1.72-3.40 for lasmiditan, 1.58-3.13 for rimegepant, and 1.54-3.05 for ubrogepant.
With respect to pain relief at 2 hours, while all medications were more effective than placebo, triptans were associated with higher ORs when compared with the other drug classes: lasmiditan (range: OR, 1.46; 95% confidence interval, 1.09-1.96 to OR, 3.31; 95% CI, 2.41-4.55), rimegepant (range: OR, 1.33; 95% CI, 1.01-1.76 to OR, 3.01; 95% CI, 2.33-3.88), and ubrogepant (range: OR, 1.38; 95% CI, 1.02-1.88 to OR, 3.13; 95% CI, 2.35-4.15)
When assessing tolerability, the researchers found that overall, triptans were associated with the higher ORs for any adverse events (AE) with a trend of dose-response relationship. Lasmiditan (in the ditan class) was associated with the highest risk for AEs among all treatments. Most of the AEs were mild to moderate and included chest pain, tightness, heaviness, and pressure.
Dr. Yang and colleagues note that, “although these two new classes of antimigraine drugs may not be as efficacious as triptans, these novel abortive agents without cardiovascular risks might offer an alternative to current specific migraine treatments for patients at risk of cardiovascular disease.”
Balancing efficacy and tolerability
“When choosing an acute medication for a patient there is always a balance between efficacy and tolerability,” headache specialist and associate director of North Shore Headache and Spine Lauren Natbony, MD, said in an interview.
“A medication can only be effective if a patient is able to tolerate it and will actually use it,” Dr. Natbony said.
With respect to the current review, Dr. Natbony pointed out, “response to acute therapy can differ between migraine attacks and may be based on variables not controlled for, such as how early in an attack the medication was taken, associated symptoms such as nausea that may make oral medications less efficacious, etc.”
The authors acknowledge that the focus on short-term responses and AEs after a single dose is a limitation of the study. They also pointed out what they considered to be a strength of the study, which was its network meta-analysis design. According to the authors, this design allowed for “multiple direct and indirect comparisons, ranking the efficacy and safety of individual pharmacologic interventions and providing more precise estimates than those of RCTs and traditional meta-analysis.”
Funding for this study was provided through grants from the Ministry of Science and Technology, Taiwan; the Brain Research Center; and National Yang Ming Chiao Tung University.
Dr. Yang has received personal fees and grants from various pharmaceutical companies. He has also received grants from the Taiwan Ministry of Technology and Science, the Brain Research Center, National Yang Ming Chiao Tung University, and Taipei Veterans General Hospital outside the submitted work. The other authors and Dr. Natbony disclosed no relevant financial relationships.
FROM JAMA NETWORK OPEN
Therapeutic homework adherence improves tics in Tourette’s disorder
Homework adherence between behavior therapy sessions is a significant predictor of therapeutic improvement in patients with Tourette’s disorder (TD), a study of 119 youth and adults suggests.
The assigning of “homework” to be completed between sessions – often used in cognitive-behavioral therapy – has been shown to reinforce learning but has not been well studied in TD.
“Understanding the relationship between homework adherence and therapeutic improvement from behavior therapy for TD may offer new insights for enhancing tic severity reductions achieved during this evidence-based treatment,” wrote Joey Ka-Yee Essoe, PhD, of the department of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, and colleagues.
To conduct the study, published in Behaviour Research and Therapy, the researchers recruited 70 youth and 49 adults with TD, ranging in age from 9 to 67 years, who underwent treatment at a single center. The average age was 21 years, and 80 participants were male. Treatment response was based on the Clinical Global Impressions of Improvement scale (CGI-I). Participants were assessed at baseline for tic severity and received eight sessions over 10 weeks. During those sessions, they were taught to perform a competing response to inhibit the expression of a tic when the tic or urge was detected.
Participants received homework at each weekly therapy session; most consisted of three to four practice sessions of about 30 minutes per week. Therapists reviewed the homework at the following session and adapted as needed to improve tic reduction skills.
After eight sessions of behavior therapy, overall greater homework adherence significantly predicted reduced tic severity and therapeutic improvement. However, early homework adherence predicted therapeutic improvement in youth, while late homework adherence predicted it in adults.
Overall,
However, homework adherence dipped midway through treatment in youth and showed a linear decline in adults, the researchers noted.
Among youth, baseline predictors of early homework adherence included lower levels of hyperactivity/impulsivity and caregiver strain. Among adults, baseline predictors of early homework adherence included lower anger scores, less social disability, and greater work disability.
The study findings were limited by several factors, including the absence of complete data on baseline predictors of homework adherence, reliance on a single measure of tic severity and improvement, and reliance on therapists’ reports of homework adherence, the researchers noted.
Future research should include objective measures of homework adherence, such as time-stamped videos, and different strategies may be needed for youth vs. adults, they added.
“Strategies that optimize homework adherence may enhance the efficacy of behavioral therapy, lead to greater tic severity reductions, and higher treatment response rates,” Dr. Essoe and colleagues wrote.
The study was supported by the Tourette Association of America, the National Institute of Mental Health, the American Academy of Neurology, and the American Psychological Foundation.
Homework adherence between behavior therapy sessions is a significant predictor of therapeutic improvement in patients with Tourette’s disorder (TD), a study of 119 youth and adults suggests.
The assigning of “homework” to be completed between sessions – often used in cognitive-behavioral therapy – has been shown to reinforce learning but has not been well studied in TD.
“Understanding the relationship between homework adherence and therapeutic improvement from behavior therapy for TD may offer new insights for enhancing tic severity reductions achieved during this evidence-based treatment,” wrote Joey Ka-Yee Essoe, PhD, of the department of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, and colleagues.
To conduct the study, published in Behaviour Research and Therapy, the researchers recruited 70 youth and 49 adults with TD, ranging in age from 9 to 67 years, who underwent treatment at a single center. The average age was 21 years, and 80 participants were male. Treatment response was based on the Clinical Global Impressions of Improvement scale (CGI-I). Participants were assessed at baseline for tic severity and received eight sessions over 10 weeks. During those sessions, they were taught to perform a competing response to inhibit the expression of a tic when the tic or urge was detected.
Participants received homework at each weekly therapy session; most consisted of three to four practice sessions of about 30 minutes per week. Therapists reviewed the homework at the following session and adapted as needed to improve tic reduction skills.
After eight sessions of behavior therapy, overall greater homework adherence significantly predicted reduced tic severity and therapeutic improvement. However, early homework adherence predicted therapeutic improvement in youth, while late homework adherence predicted it in adults.
Overall,
However, homework adherence dipped midway through treatment in youth and showed a linear decline in adults, the researchers noted.
Among youth, baseline predictors of early homework adherence included lower levels of hyperactivity/impulsivity and caregiver strain. Among adults, baseline predictors of early homework adherence included lower anger scores, less social disability, and greater work disability.
The study findings were limited by several factors, including the absence of complete data on baseline predictors of homework adherence, reliance on a single measure of tic severity and improvement, and reliance on therapists’ reports of homework adherence, the researchers noted.
Future research should include objective measures of homework adherence, such as time-stamped videos, and different strategies may be needed for youth vs. adults, they added.
“Strategies that optimize homework adherence may enhance the efficacy of behavioral therapy, lead to greater tic severity reductions, and higher treatment response rates,” Dr. Essoe and colleagues wrote.
The study was supported by the Tourette Association of America, the National Institute of Mental Health, the American Academy of Neurology, and the American Psychological Foundation.
Homework adherence between behavior therapy sessions is a significant predictor of therapeutic improvement in patients with Tourette’s disorder (TD), a study of 119 youth and adults suggests.
The assigning of “homework” to be completed between sessions – often used in cognitive-behavioral therapy – has been shown to reinforce learning but has not been well studied in TD.
“Understanding the relationship between homework adherence and therapeutic improvement from behavior therapy for TD may offer new insights for enhancing tic severity reductions achieved during this evidence-based treatment,” wrote Joey Ka-Yee Essoe, PhD, of the department of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, and colleagues.
To conduct the study, published in Behaviour Research and Therapy, the researchers recruited 70 youth and 49 adults with TD, ranging in age from 9 to 67 years, who underwent treatment at a single center. The average age was 21 years, and 80 participants were male. Treatment response was based on the Clinical Global Impressions of Improvement scale (CGI-I). Participants were assessed at baseline for tic severity and received eight sessions over 10 weeks. During those sessions, they were taught to perform a competing response to inhibit the expression of a tic when the tic or urge was detected.
Participants received homework at each weekly therapy session; most consisted of three to four practice sessions of about 30 minutes per week. Therapists reviewed the homework at the following session and adapted as needed to improve tic reduction skills.
After eight sessions of behavior therapy, overall greater homework adherence significantly predicted reduced tic severity and therapeutic improvement. However, early homework adherence predicted therapeutic improvement in youth, while late homework adherence predicted it in adults.
Overall,
However, homework adherence dipped midway through treatment in youth and showed a linear decline in adults, the researchers noted.
Among youth, baseline predictors of early homework adherence included lower levels of hyperactivity/impulsivity and caregiver strain. Among adults, baseline predictors of early homework adherence included lower anger scores, less social disability, and greater work disability.
The study findings were limited by several factors, including the absence of complete data on baseline predictors of homework adherence, reliance on a single measure of tic severity and improvement, and reliance on therapists’ reports of homework adherence, the researchers noted.
Future research should include objective measures of homework adherence, such as time-stamped videos, and different strategies may be needed for youth vs. adults, they added.
“Strategies that optimize homework adherence may enhance the efficacy of behavioral therapy, lead to greater tic severity reductions, and higher treatment response rates,” Dr. Essoe and colleagues wrote.
The study was supported by the Tourette Association of America, the National Institute of Mental Health, the American Academy of Neurology, and the American Psychological Foundation.
FROM BEHAVIOUR RESEARCH & THERAPY
Pandemic survey: Forty-six percent of pediatric headache patients got worse
, a newly released survey finds. But some actually found the pandemic era to be less stressful since they were tightly wound and could more easily control their home environments, a researcher said.
“We need to be very mindful of the connections between school and home environments – and social situations – and how they impact headache frequency,” said Marc DiSabella, DO, a pediatric neurologist at Children’s National Hospital/George Washington University, Washington. He is coauthor of a poster presented at the 50th annual meeting of the Child Neurology Society.
Dr. DiSabella and colleagues launched the survey to understand what headache patients were experiencing during the pandemic. They expected that “things were going to go really terrible in terms of headaches – or things would go great, and then things would crash when we had to reintegrate into society,” he said in an interview.
The team surveyed 113 pediatric patients who were evaluated at the hospital’s headache clinic between summer 2020 and winter 2021. Most of the patients were female (60%) and were aged 12-17 years (63%). Twenty-one percent were younger than 12 and 16% were older than 17. Chronic migraine (37%) was the most common diagnosis, followed by migraine with aura (22%), migraine without aura (19%), and new daily persistent headache (15%).
Nearly half (46%) of patients said their headaches had worsened during the pandemic. Many also reported more anxiety (55%), worsened mood (48%) and more stress (55%).
Dr. DiSabella said it’s especially notable that nearly two-thirds of those surveyed reported they were exercising less during the pandemic. Research has suggested that exercise and proper diet/sleep are crucial to improving headaches in kids, he said, and the survey findings suggest that exercise may be especially important. “Engaging in physical activity changes their pain threshold,” he said.
The researchers also reported that 60% of those surveyed said they looked at screens more than 6 hours per day. According to Dr. DiSabella, high screen use may not be worrisome from a headache perspective. “We have another study in publication that shows there’s not a clear association between frequency of screen use and headache intensity,” he said.
The survey doesn’t examine what has happened in recent weeks as schools have reopened. Anecdotally, Dr. DiSabella said some patients with migraine are feeling the stress of returning to normal routines. “They tend to be type A perfectionists and do well when they’re in control of their environment,” he said. “Now they’ve lost the control they had at home and are being put back into a stressful environment.”
Pandemic effects mixed
Commenting on the study, child neurologist Andrew D. Hershey, MD, PhD, of Cincinnati Children’s Hospital Medical Center, questioned the finding that many children suffered from more headaches during the pandemic. In his experience, “headaches were overall better when [children] were doing virtual learning,” he said in an interview. “We had fewer admissions, ED visits declined, and patients were maintaining better healthy habits. Some did express anxiety about not seeing friends, but were accommodating by doing this remotely.”
He added: “Since their return, kids are back to the same sleep deprivation issue since schools start too early, and they have more difficulty treating headaches acutely since they have to go to the nurse’s office [to do so]. They self-report a higher degree of stress and anxiety.”
On the other hand, Jack Gladstein, MD, a child neurologist at the University of Maryland, Baltimore, said in an interview that most of his patients suffered more headaches during the pandemic, although a small number with social anxiety thrived because they got to stay at home.
He agreed with Dr. DiSabella about the value of exercise. “At every visit we remind our youngsters with migraine to eat breakfast, exercise, get regular sleep, and drink fluids,” he said.
No study funding was reported. The study authors, Dr. Hershey, and Dr. Gladstein reported no disclosures.
, a newly released survey finds. But some actually found the pandemic era to be less stressful since they were tightly wound and could more easily control their home environments, a researcher said.
“We need to be very mindful of the connections between school and home environments – and social situations – and how they impact headache frequency,” said Marc DiSabella, DO, a pediatric neurologist at Children’s National Hospital/George Washington University, Washington. He is coauthor of a poster presented at the 50th annual meeting of the Child Neurology Society.
Dr. DiSabella and colleagues launched the survey to understand what headache patients were experiencing during the pandemic. They expected that “things were going to go really terrible in terms of headaches – or things would go great, and then things would crash when we had to reintegrate into society,” he said in an interview.
The team surveyed 113 pediatric patients who were evaluated at the hospital’s headache clinic between summer 2020 and winter 2021. Most of the patients were female (60%) and were aged 12-17 years (63%). Twenty-one percent were younger than 12 and 16% were older than 17. Chronic migraine (37%) was the most common diagnosis, followed by migraine with aura (22%), migraine without aura (19%), and new daily persistent headache (15%).
Nearly half (46%) of patients said their headaches had worsened during the pandemic. Many also reported more anxiety (55%), worsened mood (48%) and more stress (55%).
Dr. DiSabella said it’s especially notable that nearly two-thirds of those surveyed reported they were exercising less during the pandemic. Research has suggested that exercise and proper diet/sleep are crucial to improving headaches in kids, he said, and the survey findings suggest that exercise may be especially important. “Engaging in physical activity changes their pain threshold,” he said.
The researchers also reported that 60% of those surveyed said they looked at screens more than 6 hours per day. According to Dr. DiSabella, high screen use may not be worrisome from a headache perspective. “We have another study in publication that shows there’s not a clear association between frequency of screen use and headache intensity,” he said.
The survey doesn’t examine what has happened in recent weeks as schools have reopened. Anecdotally, Dr. DiSabella said some patients with migraine are feeling the stress of returning to normal routines. “They tend to be type A perfectionists and do well when they’re in control of their environment,” he said. “Now they’ve lost the control they had at home and are being put back into a stressful environment.”
Pandemic effects mixed
Commenting on the study, child neurologist Andrew D. Hershey, MD, PhD, of Cincinnati Children’s Hospital Medical Center, questioned the finding that many children suffered from more headaches during the pandemic. In his experience, “headaches were overall better when [children] were doing virtual learning,” he said in an interview. “We had fewer admissions, ED visits declined, and patients were maintaining better healthy habits. Some did express anxiety about not seeing friends, but were accommodating by doing this remotely.”
He added: “Since their return, kids are back to the same sleep deprivation issue since schools start too early, and they have more difficulty treating headaches acutely since they have to go to the nurse’s office [to do so]. They self-report a higher degree of stress and anxiety.”
On the other hand, Jack Gladstein, MD, a child neurologist at the University of Maryland, Baltimore, said in an interview that most of his patients suffered more headaches during the pandemic, although a small number with social anxiety thrived because they got to stay at home.
He agreed with Dr. DiSabella about the value of exercise. “At every visit we remind our youngsters with migraine to eat breakfast, exercise, get regular sleep, and drink fluids,” he said.
No study funding was reported. The study authors, Dr. Hershey, and Dr. Gladstein reported no disclosures.
, a newly released survey finds. But some actually found the pandemic era to be less stressful since they were tightly wound and could more easily control their home environments, a researcher said.
“We need to be very mindful of the connections between school and home environments – and social situations – and how they impact headache frequency,” said Marc DiSabella, DO, a pediatric neurologist at Children’s National Hospital/George Washington University, Washington. He is coauthor of a poster presented at the 50th annual meeting of the Child Neurology Society.
Dr. DiSabella and colleagues launched the survey to understand what headache patients were experiencing during the pandemic. They expected that “things were going to go really terrible in terms of headaches – or things would go great, and then things would crash when we had to reintegrate into society,” he said in an interview.
The team surveyed 113 pediatric patients who were evaluated at the hospital’s headache clinic between summer 2020 and winter 2021. Most of the patients were female (60%) and were aged 12-17 years (63%). Twenty-one percent were younger than 12 and 16% were older than 17. Chronic migraine (37%) was the most common diagnosis, followed by migraine with aura (22%), migraine without aura (19%), and new daily persistent headache (15%).
Nearly half (46%) of patients said their headaches had worsened during the pandemic. Many also reported more anxiety (55%), worsened mood (48%) and more stress (55%).
Dr. DiSabella said it’s especially notable that nearly two-thirds of those surveyed reported they were exercising less during the pandemic. Research has suggested that exercise and proper diet/sleep are crucial to improving headaches in kids, he said, and the survey findings suggest that exercise may be especially important. “Engaging in physical activity changes their pain threshold,” he said.
The researchers also reported that 60% of those surveyed said they looked at screens more than 6 hours per day. According to Dr. DiSabella, high screen use may not be worrisome from a headache perspective. “We have another study in publication that shows there’s not a clear association between frequency of screen use and headache intensity,” he said.
The survey doesn’t examine what has happened in recent weeks as schools have reopened. Anecdotally, Dr. DiSabella said some patients with migraine are feeling the stress of returning to normal routines. “They tend to be type A perfectionists and do well when they’re in control of their environment,” he said. “Now they’ve lost the control they had at home and are being put back into a stressful environment.”
Pandemic effects mixed
Commenting on the study, child neurologist Andrew D. Hershey, MD, PhD, of Cincinnati Children’s Hospital Medical Center, questioned the finding that many children suffered from more headaches during the pandemic. In his experience, “headaches were overall better when [children] were doing virtual learning,” he said in an interview. “We had fewer admissions, ED visits declined, and patients were maintaining better healthy habits. Some did express anxiety about not seeing friends, but were accommodating by doing this remotely.”
He added: “Since their return, kids are back to the same sleep deprivation issue since schools start too early, and they have more difficulty treating headaches acutely since they have to go to the nurse’s office [to do so]. They self-report a higher degree of stress and anxiety.”
On the other hand, Jack Gladstein, MD, a child neurologist at the University of Maryland, Baltimore, said in an interview that most of his patients suffered more headaches during the pandemic, although a small number with social anxiety thrived because they got to stay at home.
He agreed with Dr. DiSabella about the value of exercise. “At every visit we remind our youngsters with migraine to eat breakfast, exercise, get regular sleep, and drink fluids,” he said.
No study funding was reported. The study authors, Dr. Hershey, and Dr. Gladstein reported no disclosures.
FROM CNS 2021
‘Fascinating’ link between Alzheimer’s and COVID-19
The findings could lead to new treatment targets to slow progression and severity of both diseases.
Investigators found that a single genetic variant in the oligoadenylate synthetase 1 (OAS1) gene increases the risk for AD and that related variants in the same gene increase the likelihood of severe COVID-19 outcomes.
“These findings may allow us to identify new drug targets to slow progression of both diseases and reduce their severity,” Dervis Salih, PhD, senior research associate, UK Dementia Research Institute, University College London, said in an interview.
“Our work also suggests new approaches to treat both diseases with the same drugs,” Dr. Salih added.
The study was published online Oct. 7 in Brain.
Shared genetic network
The OAS1 gene is expressed in microglia, a type of immune cell that makes up around 10% of all cells in the brain.
In earlier work, investigators found evidence suggesting a link between the OAS1 gene and AD, but the function of the gene in microglia was unknown.
To further investigate the gene’s link to AD, they sequenced genetic data from 2,547 people – half with AD, and half without.
The genotyping analysis confirmed that the single-nucleotide polymorphism (SNP) rs1131454 within OAS1 is significantly associated with AD.
Given that the same OAS1 locus has recently been linked with severe COVID-19 outcomes, the researchers investigated four variants on the OAS1 gene.
Results indicate that SNPs within OAS1 associated with AD also show linkage to SNP variants associated with critical illness in COVID-19.
The rs1131454 (risk allele A) and rs4766676 (risk allele T) are associated with AD, and rs10735079 (risk allele A) and rs6489867 (risk allele T) are associated with critical illness with COVID-19, the investigators reported. All of these risk alleles dampen expression of OAS1.
“This study also provides strong new evidence that interferon signaling by the innate immune system plays a substantial role in the progression of Alzheimer’s,” said Dr. Salih.
“Identifying this shared genetic network in innate immune cells will allow us with future work to identify new biomarkers to track disease progression and also predict disease risk better for both disorders,” he added.
‘Fascinating’ link
In a statement from the UK nonprofit organization, Science Media Center, Kenneth Baillie, MBChB, with the University of Edinburgh, said this study builds on a discovery he and his colleagues made last year that OAS1 variants are associated with severe COVID-19.
“In the ISARIC4C study, we recently found that this is probably due to a change in the way cell membranes detect viruses, but this mechanism doesn’t explain the fascinating association with Alzheimer’s disease reported in this new work,” Dr. Baillie said.
“It is often the case that the same gene can have different roles in different parts of the body. Importantly, it doesn’t mean that having COVID-19 has any effect on your risk of Alzheimer’s,” he added.
Also weighing in on the new study, Jonathan Schott, MD, professor of neurology, University College London, noted that dementia is the “main preexisting health condition associated with COVID-19 mortality, accounting for about one in four deaths from COVID-19 between March and June 2020.
“While some of this excessive mortality may relate to people with dementia being overrepresented in care homes, which were particularly hard hit by the pandemic, or due to general increased vulnerability to infections, there have been questions as to whether there are common factors that might increase susceptibility both to developing dementia and to dying from COVID-19,” Dr. Schott explained.
This “elegant paper” provides evidence for the latter, “suggesting a common genetic mechanism both for Alzheimer’s disease and for severe COVID-19 infection,” Dr. Schott said.
“The identification of a genetic risk factor and elucidation of inflammatory pathways through which it may increase risk has important implications for our understanding of both diseases, with potential implications for novel treatments,” he added.
The study was funded by the UK Dementia Research Institute. The authors have disclosed no relevant financial relationships. Dr. Schott serves as chief medical officer for Alzheimer’s Research UK and is clinical adviser to the UK Dementia Research Institute. Dr. Baillie has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings could lead to new treatment targets to slow progression and severity of both diseases.
Investigators found that a single genetic variant in the oligoadenylate synthetase 1 (OAS1) gene increases the risk for AD and that related variants in the same gene increase the likelihood of severe COVID-19 outcomes.
“These findings may allow us to identify new drug targets to slow progression of both diseases and reduce their severity,” Dervis Salih, PhD, senior research associate, UK Dementia Research Institute, University College London, said in an interview.
“Our work also suggests new approaches to treat both diseases with the same drugs,” Dr. Salih added.
The study was published online Oct. 7 in Brain.
Shared genetic network
The OAS1 gene is expressed in microglia, a type of immune cell that makes up around 10% of all cells in the brain.
In earlier work, investigators found evidence suggesting a link between the OAS1 gene and AD, but the function of the gene in microglia was unknown.
To further investigate the gene’s link to AD, they sequenced genetic data from 2,547 people – half with AD, and half without.
The genotyping analysis confirmed that the single-nucleotide polymorphism (SNP) rs1131454 within OAS1 is significantly associated with AD.
Given that the same OAS1 locus has recently been linked with severe COVID-19 outcomes, the researchers investigated four variants on the OAS1 gene.
Results indicate that SNPs within OAS1 associated with AD also show linkage to SNP variants associated with critical illness in COVID-19.
The rs1131454 (risk allele A) and rs4766676 (risk allele T) are associated with AD, and rs10735079 (risk allele A) and rs6489867 (risk allele T) are associated with critical illness with COVID-19, the investigators reported. All of these risk alleles dampen expression of OAS1.
“This study also provides strong new evidence that interferon signaling by the innate immune system plays a substantial role in the progression of Alzheimer’s,” said Dr. Salih.
“Identifying this shared genetic network in innate immune cells will allow us with future work to identify new biomarkers to track disease progression and also predict disease risk better for both disorders,” he added.
‘Fascinating’ link
In a statement from the UK nonprofit organization, Science Media Center, Kenneth Baillie, MBChB, with the University of Edinburgh, said this study builds on a discovery he and his colleagues made last year that OAS1 variants are associated with severe COVID-19.
“In the ISARIC4C study, we recently found that this is probably due to a change in the way cell membranes detect viruses, but this mechanism doesn’t explain the fascinating association with Alzheimer’s disease reported in this new work,” Dr. Baillie said.
“It is often the case that the same gene can have different roles in different parts of the body. Importantly, it doesn’t mean that having COVID-19 has any effect on your risk of Alzheimer’s,” he added.
Also weighing in on the new study, Jonathan Schott, MD, professor of neurology, University College London, noted that dementia is the “main preexisting health condition associated with COVID-19 mortality, accounting for about one in four deaths from COVID-19 between March and June 2020.
“While some of this excessive mortality may relate to people with dementia being overrepresented in care homes, which were particularly hard hit by the pandemic, or due to general increased vulnerability to infections, there have been questions as to whether there are common factors that might increase susceptibility both to developing dementia and to dying from COVID-19,” Dr. Schott explained.
This “elegant paper” provides evidence for the latter, “suggesting a common genetic mechanism both for Alzheimer’s disease and for severe COVID-19 infection,” Dr. Schott said.
“The identification of a genetic risk factor and elucidation of inflammatory pathways through which it may increase risk has important implications for our understanding of both diseases, with potential implications for novel treatments,” he added.
The study was funded by the UK Dementia Research Institute. The authors have disclosed no relevant financial relationships. Dr. Schott serves as chief medical officer for Alzheimer’s Research UK and is clinical adviser to the UK Dementia Research Institute. Dr. Baillie has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings could lead to new treatment targets to slow progression and severity of both diseases.
Investigators found that a single genetic variant in the oligoadenylate synthetase 1 (OAS1) gene increases the risk for AD and that related variants in the same gene increase the likelihood of severe COVID-19 outcomes.
“These findings may allow us to identify new drug targets to slow progression of both diseases and reduce their severity,” Dervis Salih, PhD, senior research associate, UK Dementia Research Institute, University College London, said in an interview.
“Our work also suggests new approaches to treat both diseases with the same drugs,” Dr. Salih added.
The study was published online Oct. 7 in Brain.
Shared genetic network
The OAS1 gene is expressed in microglia, a type of immune cell that makes up around 10% of all cells in the brain.
In earlier work, investigators found evidence suggesting a link between the OAS1 gene and AD, but the function of the gene in microglia was unknown.
To further investigate the gene’s link to AD, they sequenced genetic data from 2,547 people – half with AD, and half without.
The genotyping analysis confirmed that the single-nucleotide polymorphism (SNP) rs1131454 within OAS1 is significantly associated with AD.
Given that the same OAS1 locus has recently been linked with severe COVID-19 outcomes, the researchers investigated four variants on the OAS1 gene.
Results indicate that SNPs within OAS1 associated with AD also show linkage to SNP variants associated with critical illness in COVID-19.
The rs1131454 (risk allele A) and rs4766676 (risk allele T) are associated with AD, and rs10735079 (risk allele A) and rs6489867 (risk allele T) are associated with critical illness with COVID-19, the investigators reported. All of these risk alleles dampen expression of OAS1.
“This study also provides strong new evidence that interferon signaling by the innate immune system plays a substantial role in the progression of Alzheimer’s,” said Dr. Salih.
“Identifying this shared genetic network in innate immune cells will allow us with future work to identify new biomarkers to track disease progression and also predict disease risk better for both disorders,” he added.
‘Fascinating’ link
In a statement from the UK nonprofit organization, Science Media Center, Kenneth Baillie, MBChB, with the University of Edinburgh, said this study builds on a discovery he and his colleagues made last year that OAS1 variants are associated with severe COVID-19.
“In the ISARIC4C study, we recently found that this is probably due to a change in the way cell membranes detect viruses, but this mechanism doesn’t explain the fascinating association with Alzheimer’s disease reported in this new work,” Dr. Baillie said.
“It is often the case that the same gene can have different roles in different parts of the body. Importantly, it doesn’t mean that having COVID-19 has any effect on your risk of Alzheimer’s,” he added.
Also weighing in on the new study, Jonathan Schott, MD, professor of neurology, University College London, noted that dementia is the “main preexisting health condition associated with COVID-19 mortality, accounting for about one in four deaths from COVID-19 between March and June 2020.
“While some of this excessive mortality may relate to people with dementia being overrepresented in care homes, which were particularly hard hit by the pandemic, or due to general increased vulnerability to infections, there have been questions as to whether there are common factors that might increase susceptibility both to developing dementia and to dying from COVID-19,” Dr. Schott explained.
This “elegant paper” provides evidence for the latter, “suggesting a common genetic mechanism both for Alzheimer’s disease and for severe COVID-19 infection,” Dr. Schott said.
“The identification of a genetic risk factor and elucidation of inflammatory pathways through which it may increase risk has important implications for our understanding of both diseases, with potential implications for novel treatments,” he added.
The study was funded by the UK Dementia Research Institute. The authors have disclosed no relevant financial relationships. Dr. Schott serves as chief medical officer for Alzheimer’s Research UK and is clinical adviser to the UK Dementia Research Institute. Dr. Baillie has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Epidiolex plus THC lowers seizures in pediatric epilepsy
the component of cannabis that makes people high in larger quantities, researchers reported.
“THC can contribute to seizure control and mitigation some of the side effects of CBD,” said study coauthor and Austin, Tex., child neurologist Karen Keough, MD, in an interview. Dr. Keough and colleagues presented their findings at the 50th annual meeting of the Child Neurology Society.
In a landmark move, the Food and Drug Administration approved Epidiolex in 2018 for the treatment of seizures in two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome. The agency had never before approved a drug with a purified ingredient derived from marijuana.
CBD, the active ingredient in Epidiolex, is nonpsychoactive. The use in medicine of THC, the main driver of marijuana’s ability to make people stoned, is much more controversial.
Dr. Keough said she had treated 60-70 children with CBD, at the same strength as in Epidiolex (100 mg), and 5 mg of THC before the drug was approved. “I was seeing some very impressive results, and some became seizure free who’d always been refractory,” she said.
When the Epidiolex became available, she said, some patients transitioned to it and stopped taking THC. According to her, some patients fared well. But others immediately experienced worse seizures, she said, and some developed side effects to Epidiolex in the absence of THC, such as agitation and appetite suppression.
Combination therapy
For the new study, a retrospective, unblinded cohort analysis, Dr. Keough and colleagues tracked patients who received various doses of CBD, in some cases as Epidiolex, and various doses of THC prescribed by the Texas Original Compassionate Cultivation dispensary, where she serves as chief medical officer.
The initial number of patients was 212; 135 consented to review and 10 were excluded for various reasons leaving a total of 74 subjects in the study. The subjects, whose median age at the start of the study was 12 years (range, 2-25 years), were tracked from 2018 to2021. Just over half (55%) were male, and they remained on the regimen for a median of 805 days (range, 400-1,141).
Of the 74 subjects, 45.9% had a reduction of seizures of more than 75%, and 20.3% had a reduction of 50%-75%. Only 4.1% saw their seizures worsen.
The THC doses varied from none to more than 12 mg/day; CBD doses varied from none to more than 26 mg/kg per day. O the 74 patients, 18 saw their greatest seizure reduction from baseline when they received no THC; 12 saw their greatest seizure reduction from baseline when they received 0-2 mg/kg per day of CBD.
Still controversial
Did the patients get high? In some cases they did, Dr. Keough said. However, “a lot of these patients are either too young or too cognitively limited to describe whether they’re feeling intoxicated. That’s one of the many reasons why this is so controversial. You have to go into this with eyes wide open. We’re working in an environment with limited information as to what an intoxicating dose is for a small kid.”
However, she said, it seems clear that “THC can enhance the effect of CBD in children with epilepsy” and reduce CBD side effects. It’s not surprising that the substances work differently since they interact with brain cells in different ways, she said.
For neurologists, she said, “the challenge is to find a reliable source of THC that you can count on and verify so you aren’t overdosing the patients.”
University of Saskatchewan, Saskatoon, child neurologist and cannabinoid researcher Richard Huntsman, MD, who’s familiar with the study findings, said in an interview that they “provide another strong signal that the addition of THC provides benefit, at least in some patients.”
But it’s still unclear “why some children respond best in regards to seizure reduction and side effect profile with combination CBD:THC therapy, and others seemed to do better with CBD alone,” he said. Also unknown: “the ideal THC:CBD ratio that allows optimal seizure control while preventing the potential harmful effects of THC.”
As for the future, he said, “as we are just scratching the surface of our knowledge about the use of cannabis-based therapies in children with neurological disorders, I suspect that the use of these therapies will expand over time.”
No study funding is reported. Dr. Keough disclosed serving as chief medical officer of Texas Original Compassionate Cultivation. Dr. Huntsman disclosed serving as lead investigator of the Cannabidiol in Children with Refractory Epileptic Encephalopathy study and serving on the boards of the Cannabinoid Research Initiative of Saskatchewan (University of Saskatchewan) and Canadian Childhood Cannabinoid Clinical Trials Consortium. He is also cochair of Health Canada’s Scientific Advisory Committee on Cannabinoids for Health Purposes.
the component of cannabis that makes people high in larger quantities, researchers reported.
“THC can contribute to seizure control and mitigation some of the side effects of CBD,” said study coauthor and Austin, Tex., child neurologist Karen Keough, MD, in an interview. Dr. Keough and colleagues presented their findings at the 50th annual meeting of the Child Neurology Society.
In a landmark move, the Food and Drug Administration approved Epidiolex in 2018 for the treatment of seizures in two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome. The agency had never before approved a drug with a purified ingredient derived from marijuana.
CBD, the active ingredient in Epidiolex, is nonpsychoactive. The use in medicine of THC, the main driver of marijuana’s ability to make people stoned, is much more controversial.
Dr. Keough said she had treated 60-70 children with CBD, at the same strength as in Epidiolex (100 mg), and 5 mg of THC before the drug was approved. “I was seeing some very impressive results, and some became seizure free who’d always been refractory,” she said.
When the Epidiolex became available, she said, some patients transitioned to it and stopped taking THC. According to her, some patients fared well. But others immediately experienced worse seizures, she said, and some developed side effects to Epidiolex in the absence of THC, such as agitation and appetite suppression.
Combination therapy
For the new study, a retrospective, unblinded cohort analysis, Dr. Keough and colleagues tracked patients who received various doses of CBD, in some cases as Epidiolex, and various doses of THC prescribed by the Texas Original Compassionate Cultivation dispensary, where she serves as chief medical officer.
The initial number of patients was 212; 135 consented to review and 10 were excluded for various reasons leaving a total of 74 subjects in the study. The subjects, whose median age at the start of the study was 12 years (range, 2-25 years), were tracked from 2018 to2021. Just over half (55%) were male, and they remained on the regimen for a median of 805 days (range, 400-1,141).
Of the 74 subjects, 45.9% had a reduction of seizures of more than 75%, and 20.3% had a reduction of 50%-75%. Only 4.1% saw their seizures worsen.
The THC doses varied from none to more than 12 mg/day; CBD doses varied from none to more than 26 mg/kg per day. O the 74 patients, 18 saw their greatest seizure reduction from baseline when they received no THC; 12 saw their greatest seizure reduction from baseline when they received 0-2 mg/kg per day of CBD.
Still controversial
Did the patients get high? In some cases they did, Dr. Keough said. However, “a lot of these patients are either too young or too cognitively limited to describe whether they’re feeling intoxicated. That’s one of the many reasons why this is so controversial. You have to go into this with eyes wide open. We’re working in an environment with limited information as to what an intoxicating dose is for a small kid.”
However, she said, it seems clear that “THC can enhance the effect of CBD in children with epilepsy” and reduce CBD side effects. It’s not surprising that the substances work differently since they interact with brain cells in different ways, she said.
For neurologists, she said, “the challenge is to find a reliable source of THC that you can count on and verify so you aren’t overdosing the patients.”
University of Saskatchewan, Saskatoon, child neurologist and cannabinoid researcher Richard Huntsman, MD, who’s familiar with the study findings, said in an interview that they “provide another strong signal that the addition of THC provides benefit, at least in some patients.”
But it’s still unclear “why some children respond best in regards to seizure reduction and side effect profile with combination CBD:THC therapy, and others seemed to do better with CBD alone,” he said. Also unknown: “the ideal THC:CBD ratio that allows optimal seizure control while preventing the potential harmful effects of THC.”
As for the future, he said, “as we are just scratching the surface of our knowledge about the use of cannabis-based therapies in children with neurological disorders, I suspect that the use of these therapies will expand over time.”
No study funding is reported. Dr. Keough disclosed serving as chief medical officer of Texas Original Compassionate Cultivation. Dr. Huntsman disclosed serving as lead investigator of the Cannabidiol in Children with Refractory Epileptic Encephalopathy study and serving on the boards of the Cannabinoid Research Initiative of Saskatchewan (University of Saskatchewan) and Canadian Childhood Cannabinoid Clinical Trials Consortium. He is also cochair of Health Canada’s Scientific Advisory Committee on Cannabinoids for Health Purposes.
the component of cannabis that makes people high in larger quantities, researchers reported.
“THC can contribute to seizure control and mitigation some of the side effects of CBD,” said study coauthor and Austin, Tex., child neurologist Karen Keough, MD, in an interview. Dr. Keough and colleagues presented their findings at the 50th annual meeting of the Child Neurology Society.
In a landmark move, the Food and Drug Administration approved Epidiolex in 2018 for the treatment of seizures in two rare forms of epilepsy, Lennox-Gastaut syndrome and Dravet syndrome. The agency had never before approved a drug with a purified ingredient derived from marijuana.
CBD, the active ingredient in Epidiolex, is nonpsychoactive. The use in medicine of THC, the main driver of marijuana’s ability to make people stoned, is much more controversial.
Dr. Keough said she had treated 60-70 children with CBD, at the same strength as in Epidiolex (100 mg), and 5 mg of THC before the drug was approved. “I was seeing some very impressive results, and some became seizure free who’d always been refractory,” she said.
When the Epidiolex became available, she said, some patients transitioned to it and stopped taking THC. According to her, some patients fared well. But others immediately experienced worse seizures, she said, and some developed side effects to Epidiolex in the absence of THC, such as agitation and appetite suppression.
Combination therapy
For the new study, a retrospective, unblinded cohort analysis, Dr. Keough and colleagues tracked patients who received various doses of CBD, in some cases as Epidiolex, and various doses of THC prescribed by the Texas Original Compassionate Cultivation dispensary, where she serves as chief medical officer.
The initial number of patients was 212; 135 consented to review and 10 were excluded for various reasons leaving a total of 74 subjects in the study. The subjects, whose median age at the start of the study was 12 years (range, 2-25 years), were tracked from 2018 to2021. Just over half (55%) were male, and they remained on the regimen for a median of 805 days (range, 400-1,141).
Of the 74 subjects, 45.9% had a reduction of seizures of more than 75%, and 20.3% had a reduction of 50%-75%. Only 4.1% saw their seizures worsen.
The THC doses varied from none to more than 12 mg/day; CBD doses varied from none to more than 26 mg/kg per day. O the 74 patients, 18 saw their greatest seizure reduction from baseline when they received no THC; 12 saw their greatest seizure reduction from baseline when they received 0-2 mg/kg per day of CBD.
Still controversial
Did the patients get high? In some cases they did, Dr. Keough said. However, “a lot of these patients are either too young or too cognitively limited to describe whether they’re feeling intoxicated. That’s one of the many reasons why this is so controversial. You have to go into this with eyes wide open. We’re working in an environment with limited information as to what an intoxicating dose is for a small kid.”
However, she said, it seems clear that “THC can enhance the effect of CBD in children with epilepsy” and reduce CBD side effects. It’s not surprising that the substances work differently since they interact with brain cells in different ways, she said.
For neurologists, she said, “the challenge is to find a reliable source of THC that you can count on and verify so you aren’t overdosing the patients.”
University of Saskatchewan, Saskatoon, child neurologist and cannabinoid researcher Richard Huntsman, MD, who’s familiar with the study findings, said in an interview that they “provide another strong signal that the addition of THC provides benefit, at least in some patients.”
But it’s still unclear “why some children respond best in regards to seizure reduction and side effect profile with combination CBD:THC therapy, and others seemed to do better with CBD alone,” he said. Also unknown: “the ideal THC:CBD ratio that allows optimal seizure control while preventing the potential harmful effects of THC.”
As for the future, he said, “as we are just scratching the surface of our knowledge about the use of cannabis-based therapies in children with neurological disorders, I suspect that the use of these therapies will expand over time.”
No study funding is reported. Dr. Keough disclosed serving as chief medical officer of Texas Original Compassionate Cultivation. Dr. Huntsman disclosed serving as lead investigator of the Cannabidiol in Children with Refractory Epileptic Encephalopathy study and serving on the boards of the Cannabinoid Research Initiative of Saskatchewan (University of Saskatchewan) and Canadian Childhood Cannabinoid Clinical Trials Consortium. He is also cochair of Health Canada’s Scientific Advisory Committee on Cannabinoids for Health Purposes.
FROM CNS 2021
Is genetic testing valuable in the clinical management of epilepsy?
, new research shows.
Results of a survey that included more than 400 patients showed that positive findings from genetic testing helped guide clinical management in 50% of cases and improved patient outcomes in 75%. In addition, the findings were applicable to both children and adults.
“Fifty percent of the time the physicians reported that, yes, receiving the genetic diagnosis did change how they managed the patients,” reported co-investigator Dianalee McKnight, PhD, director of medical affairs at Invitae, a medical genetic testing company headquartered in San Francisco. In 81.3% of cases, providers reported they changed clinical management within 3 months of receiving the genetic results, she added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
Test results can be practice-changing
Nearly 50% of positive genetic test results in epilepsy patients can help guide clinical management, Dr. McKnight noted. However, information on how physicians use genetic information in decision-making has been limited, prompting her conduct the survey.
A total of 1,567 physicians with 3,572 patients who had a definitive diagnosis of epilepsy were contacted. A total of 170 (10.8%) clinicians provided completed and eligible surveys on 429 patients with epilepsy.
The patient cohort comprised mostly children, with nearly 50 adults, which Dr. McKnight said is typical of the population receiving genetic testing in clinical practice.
She reported that genetic testing results prompted clinicians to make medication changes about 50% of the time. Other changes included specialist referral or to a clinical trial, monitoring for other neurological disease, and recommendations for dietary change or for surgery.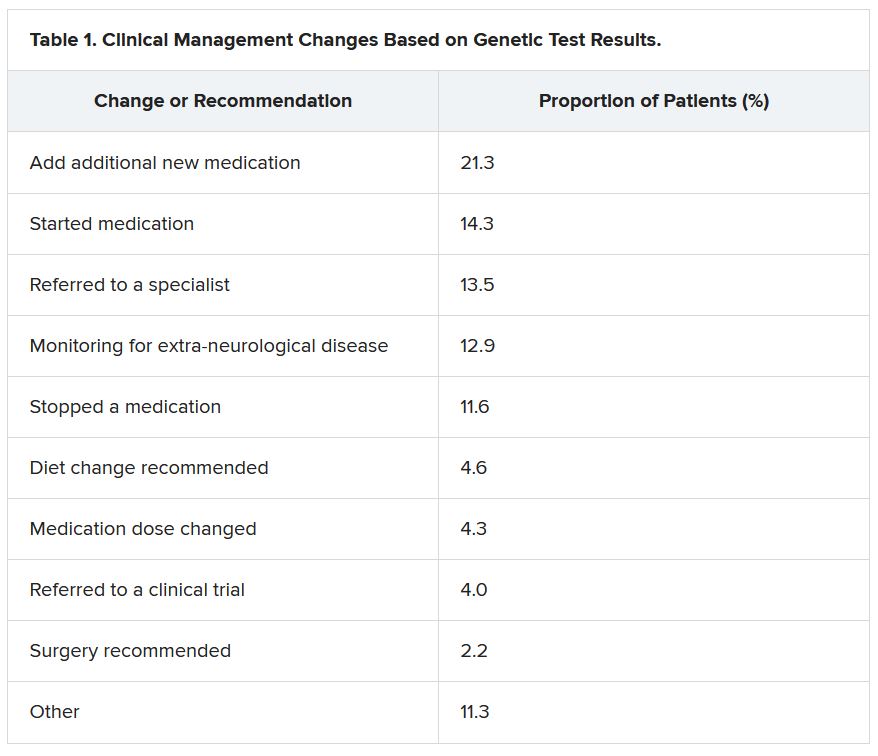
“Of the physicians who changed treatment, 75% reported there were positive outcomes for the patients,” Dr. McKnight told meeting attendees. “Most common was a reduction or a complete elimination of seizures, and that was reported in 65% of the cases.”
In many cases, the changes resulted in clinical improvements.
“There were 64 individuals who were having daily seizures before the genetic testing,” Dr. McKnight reported via email. “After receiving the genetic diagnosis and modifying their treatment, their physicians reported that 26% of individuals had complete seizure control and 46% of individuals had reduced seizure frequency to either weekly (20%), monthly (20%) or annually (6%).”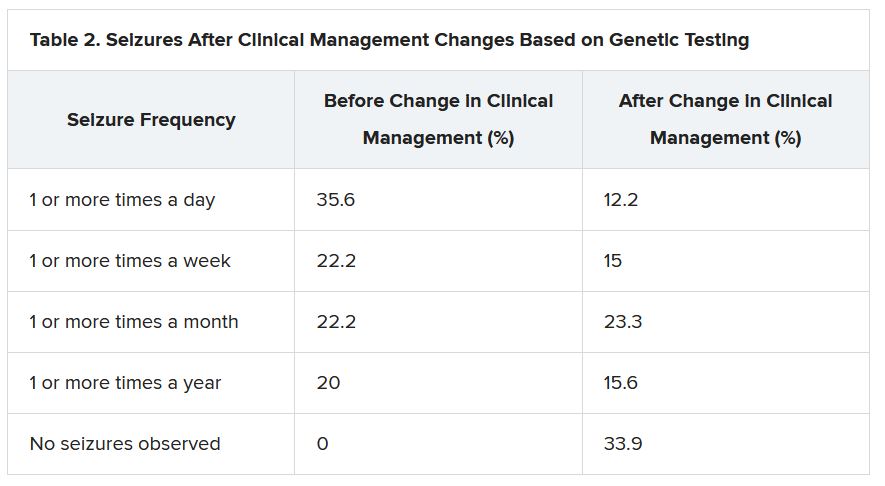
The best seizure control after modifying disease management occurred among children. Although the changes were not as dramatic for adults, they trended toward lower seizure frequency.
“It is still pretty significant that adults can receive genetic testing later in life and still have benefit in controlling their seizures,” Dr. McKnight said.
Twenty-three percent of patients showed improvement in behavior, development, academics, or movement issues, while 6% experienced reduced medication side effects.
Dr. McKnight also explored reasons for physicians not making changes to clinical management of patients based on the genetic results. The most common reason was that management was already consistent with the results (47.3%), followed by the results not being informative (26.1%), the results possibly being useful for future treatments in development (19.0%), or other or unknown reasons (7.6%).
Besides direct health and quality of life benefits from better seizure control, Dr. McKnight cited previous economic studies showing lower health care costs.
“It looked like an individual who has good seizure control will incur about 14,000 U.S. dollars a year compared with an individual with pretty poor seizure control, where it can be closer to 23,000 U.S. dollars a year,” Dr. McKnight said. This is mainly attributed to reduced hospitalizations and emergency department visits.
Dr. McKnight noted that currently there is no cost of genetic testing to the patient, the hospital, or insurers. Pharmaceutical companies, she said, sponsor the testing to potentially gather patients for clinical drug trials in development. However, patients remain completely anonymous.
Physicians who wish to have patient samples tested agree that the companies may contact them to ask if any of their patients with positive genetic test results would like to participate in a trial.
Dr. McKnight noted that genetic testing can be considered actionable in the clinic, helping to guide clinical decision-making and potentially leading to better outcomes. Going forward, she suggested performing large case-controlled studies “of individuals with the same genetic etiology ... to really find a true causation or correlation.”
Growing influence of genetic testing
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center in Columbus, noted that the study highlights the value of gene testing in improving outcomes in patients with epilepsy, particularly the pediatric population.
He said the findings make him optimistic about the potential of genetic testing in adult patients – with at least one caveat.
“The limitation is that if we do find some mutation, we don’t know what to do with that. That’s definitely one challenge. And we see that more often in the adult patient population,” said Dr. Singh, who was not involved with the research.
He noted that there is a small group of genetic mutations when, found in adults, may dramatically alter treatment.
For example, he noted that if there is a gene mutation related to mTOR pathways, that could provide a future target because there are already medications that target this pathway.
Genetic testing may also be useful in cases where patients have normal brain imaging and poor response to standard treatment or in cases where patients have congenital abnormalities such as intellectual impairment or facial dysmorphic features and a co-morbid seizure disorder, he said.
Dr. Singh noted that he has often found genetic testing impractical because “if I order DNA testing right now, it will take 4 months for me to get the results. I cannot wait 4 months for the results to come back” to adjust treatment.
Dr. McKnight is an employee of and a shareholder in Invitae, which funded the study. Dr. Singh has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows.
Results of a survey that included more than 400 patients showed that positive findings from genetic testing helped guide clinical management in 50% of cases and improved patient outcomes in 75%. In addition, the findings were applicable to both children and adults.
“Fifty percent of the time the physicians reported that, yes, receiving the genetic diagnosis did change how they managed the patients,” reported co-investigator Dianalee McKnight, PhD, director of medical affairs at Invitae, a medical genetic testing company headquartered in San Francisco. In 81.3% of cases, providers reported they changed clinical management within 3 months of receiving the genetic results, she added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
Test results can be practice-changing
Nearly 50% of positive genetic test results in epilepsy patients can help guide clinical management, Dr. McKnight noted. However, information on how physicians use genetic information in decision-making has been limited, prompting her conduct the survey.
A total of 1,567 physicians with 3,572 patients who had a definitive diagnosis of epilepsy were contacted. A total of 170 (10.8%) clinicians provided completed and eligible surveys on 429 patients with epilepsy.
The patient cohort comprised mostly children, with nearly 50 adults, which Dr. McKnight said is typical of the population receiving genetic testing in clinical practice.
She reported that genetic testing results prompted clinicians to make medication changes about 50% of the time. Other changes included specialist referral or to a clinical trial, monitoring for other neurological disease, and recommendations for dietary change or for surgery.
“Of the physicians who changed treatment, 75% reported there were positive outcomes for the patients,” Dr. McKnight told meeting attendees. “Most common was a reduction or a complete elimination of seizures, and that was reported in 65% of the cases.”
In many cases, the changes resulted in clinical improvements.
“There were 64 individuals who were having daily seizures before the genetic testing,” Dr. McKnight reported via email. “After receiving the genetic diagnosis and modifying their treatment, their physicians reported that 26% of individuals had complete seizure control and 46% of individuals had reduced seizure frequency to either weekly (20%), monthly (20%) or annually (6%).”
The best seizure control after modifying disease management occurred among children. Although the changes were not as dramatic for adults, they trended toward lower seizure frequency.
“It is still pretty significant that adults can receive genetic testing later in life and still have benefit in controlling their seizures,” Dr. McKnight said.
Twenty-three percent of patients showed improvement in behavior, development, academics, or movement issues, while 6% experienced reduced medication side effects.
Dr. McKnight also explored reasons for physicians not making changes to clinical management of patients based on the genetic results. The most common reason was that management was already consistent with the results (47.3%), followed by the results not being informative (26.1%), the results possibly being useful for future treatments in development (19.0%), or other or unknown reasons (7.6%).
Besides direct health and quality of life benefits from better seizure control, Dr. McKnight cited previous economic studies showing lower health care costs.
“It looked like an individual who has good seizure control will incur about 14,000 U.S. dollars a year compared with an individual with pretty poor seizure control, where it can be closer to 23,000 U.S. dollars a year,” Dr. McKnight said. This is mainly attributed to reduced hospitalizations and emergency department visits.
Dr. McKnight noted that currently there is no cost of genetic testing to the patient, the hospital, or insurers. Pharmaceutical companies, she said, sponsor the testing to potentially gather patients for clinical drug trials in development. However, patients remain completely anonymous.
Physicians who wish to have patient samples tested agree that the companies may contact them to ask if any of their patients with positive genetic test results would like to participate in a trial.
Dr. McKnight noted that genetic testing can be considered actionable in the clinic, helping to guide clinical decision-making and potentially leading to better outcomes. Going forward, she suggested performing large case-controlled studies “of individuals with the same genetic etiology ... to really find a true causation or correlation.”
Growing influence of genetic testing
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center in Columbus, noted that the study highlights the value of gene testing in improving outcomes in patients with epilepsy, particularly the pediatric population.
He said the findings make him optimistic about the potential of genetic testing in adult patients – with at least one caveat.
“The limitation is that if we do find some mutation, we don’t know what to do with that. That’s definitely one challenge. And we see that more often in the adult patient population,” said Dr. Singh, who was not involved with the research.
He noted that there is a small group of genetic mutations when, found in adults, may dramatically alter treatment.
For example, he noted that if there is a gene mutation related to mTOR pathways, that could provide a future target because there are already medications that target this pathway.
Genetic testing may also be useful in cases where patients have normal brain imaging and poor response to standard treatment or in cases where patients have congenital abnormalities such as intellectual impairment or facial dysmorphic features and a co-morbid seizure disorder, he said.
Dr. Singh noted that he has often found genetic testing impractical because “if I order DNA testing right now, it will take 4 months for me to get the results. I cannot wait 4 months for the results to come back” to adjust treatment.
Dr. McKnight is an employee of and a shareholder in Invitae, which funded the study. Dr. Singh has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research shows.
Results of a survey that included more than 400 patients showed that positive findings from genetic testing helped guide clinical management in 50% of cases and improved patient outcomes in 75%. In addition, the findings were applicable to both children and adults.
“Fifty percent of the time the physicians reported that, yes, receiving the genetic diagnosis did change how they managed the patients,” reported co-investigator Dianalee McKnight, PhD, director of medical affairs at Invitae, a medical genetic testing company headquartered in San Francisco. In 81.3% of cases, providers reported they changed clinical management within 3 months of receiving the genetic results, she added.
The findings were presented at the 2021 World Congress of Neurology (WCN).
Test results can be practice-changing
Nearly 50% of positive genetic test results in epilepsy patients can help guide clinical management, Dr. McKnight noted. However, information on how physicians use genetic information in decision-making has been limited, prompting her conduct the survey.
A total of 1,567 physicians with 3,572 patients who had a definitive diagnosis of epilepsy were contacted. A total of 170 (10.8%) clinicians provided completed and eligible surveys on 429 patients with epilepsy.
The patient cohort comprised mostly children, with nearly 50 adults, which Dr. McKnight said is typical of the population receiving genetic testing in clinical practice.
She reported that genetic testing results prompted clinicians to make medication changes about 50% of the time. Other changes included specialist referral or to a clinical trial, monitoring for other neurological disease, and recommendations for dietary change or for surgery.
“Of the physicians who changed treatment, 75% reported there were positive outcomes for the patients,” Dr. McKnight told meeting attendees. “Most common was a reduction or a complete elimination of seizures, and that was reported in 65% of the cases.”
In many cases, the changes resulted in clinical improvements.
“There were 64 individuals who were having daily seizures before the genetic testing,” Dr. McKnight reported via email. “After receiving the genetic diagnosis and modifying their treatment, their physicians reported that 26% of individuals had complete seizure control and 46% of individuals had reduced seizure frequency to either weekly (20%), monthly (20%) or annually (6%).”
The best seizure control after modifying disease management occurred among children. Although the changes were not as dramatic for adults, they trended toward lower seizure frequency.
“It is still pretty significant that adults can receive genetic testing later in life and still have benefit in controlling their seizures,” Dr. McKnight said.
Twenty-three percent of patients showed improvement in behavior, development, academics, or movement issues, while 6% experienced reduced medication side effects.
Dr. McKnight also explored reasons for physicians not making changes to clinical management of patients based on the genetic results. The most common reason was that management was already consistent with the results (47.3%), followed by the results not being informative (26.1%), the results possibly being useful for future treatments in development (19.0%), or other or unknown reasons (7.6%).
Besides direct health and quality of life benefits from better seizure control, Dr. McKnight cited previous economic studies showing lower health care costs.
“It looked like an individual who has good seizure control will incur about 14,000 U.S. dollars a year compared with an individual with pretty poor seizure control, where it can be closer to 23,000 U.S. dollars a year,” Dr. McKnight said. This is mainly attributed to reduced hospitalizations and emergency department visits.
Dr. McKnight noted that currently there is no cost of genetic testing to the patient, the hospital, or insurers. Pharmaceutical companies, she said, sponsor the testing to potentially gather patients for clinical drug trials in development. However, patients remain completely anonymous.
Physicians who wish to have patient samples tested agree that the companies may contact them to ask if any of their patients with positive genetic test results would like to participate in a trial.
Dr. McKnight noted that genetic testing can be considered actionable in the clinic, helping to guide clinical decision-making and potentially leading to better outcomes. Going forward, she suggested performing large case-controlled studies “of individuals with the same genetic etiology ... to really find a true causation or correlation.”
Growing influence of genetic testing
Commenting on the findings, Jaysingh Singh, MD, co-director of the Epilepsy Surgery Center at the Ohio State University Wexner Medical Center in Columbus, noted that the study highlights the value of gene testing in improving outcomes in patients with epilepsy, particularly the pediatric population.
He said the findings make him optimistic about the potential of genetic testing in adult patients – with at least one caveat.
“The limitation is that if we do find some mutation, we don’t know what to do with that. That’s definitely one challenge. And we see that more often in the adult patient population,” said Dr. Singh, who was not involved with the research.
He noted that there is a small group of genetic mutations when, found in adults, may dramatically alter treatment.
For example, he noted that if there is a gene mutation related to mTOR pathways, that could provide a future target because there are already medications that target this pathway.
Genetic testing may also be useful in cases where patients have normal brain imaging and poor response to standard treatment or in cases where patients have congenital abnormalities such as intellectual impairment or facial dysmorphic features and a co-morbid seizure disorder, he said.
Dr. Singh noted that he has often found genetic testing impractical because “if I order DNA testing right now, it will take 4 months for me to get the results. I cannot wait 4 months for the results to come back” to adjust treatment.
Dr. McKnight is an employee of and a shareholder in Invitae, which funded the study. Dr. Singh has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
From WCN 2021