User login
Bone Metastasis: Concise Overview
Bone metastasis is a relatively common complication of cancer, often developing as they advance, especially in prostate cancer and breast cancer. Bone metastasis can profoundly affect patients’ daily activities and quality of life (QOL) due to severe pain and associated major complications. Prompt palliative therapy is required for symptomatic pain relief and prevention of the devastating complications of bone metastasis.
Epidemiology
Bone is the most common and preferred site for metastatic involvement of cancer. Advanced cancers frequently develop metastases to the bone during the later phases of cancer progression. At least 100,000 patients develop bone metastases every year, although the exact number of bone metastases is not known.1 Multiple myeloma (MM), breast cancer, and prostate cancer are responsible for up to 70% of bone metastases cases.2 Gastrointestinal cancers contribute least to bone metastases: < 15% of all cases.2
Related: Effective Treatment Options for Metastatic Pancreatic Cancer
The prognosis of bone metastases is generally poor, although it partly depends on the primary site of the original cancer and on the presence of any additional metastases to visceral organs. For example, it is known that survival times are longer for patients with primary prostate or breast cancer than for patients with lung cancer primary tumors.3,4
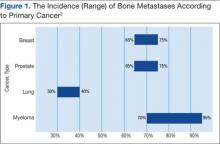
Prostate and breast cancers are the most common primary cancers of bone metastases. At postmortem studies, patients who died of prostate cancer or breast cancer revealed evidence of bone metastases in up to 75% of cases (Figure 1). Regardless of their survival expectancy, however, most patients with bone metastasis need immediate medical attention and active palliative therapy to prevent devastating complications related to bone metastasis, such as pathologic bone fractures and severe bone pain.
Clinical Features
Multiple Myeloma
Multiple myeloma is the second most common hematologic malignancy and is caused by an abnormal accumulation of clonal plasma cells in the bone marrow. Characteristic clinical manifestations include bony destruction and related features of bone pain, anemia (80% of cases), hypocalcemia, and renal dysfunction. Pathologic fractures, renal failure, or hyperviscosity syndrome often develops. More than 20,000 new patients are diagnosed with MM and about 11,000 patients in the U.S. die of MM every year. Multiple myeloma and is twice as likely to develop in men as it is in women. A large number of MM cases are under the care of VAMCs (about 10%-12% of all MM cases).7,8
Abnormal laboratory tests show an elevated total protein level in the blood and/or urine (Bence Jones proteinuria). Serum electrophoresis detects M-protein in about 80% to 90% of patients. Patients may also present with renal failure. The differential diagnosis includes other malignancies, such as metastatic carcinoma, lymphoma, leukemia, and monoclonal gammopathy.
Pathophysiology
Normal bone tissue is made up of 2 different types of cells: osteoblasts and osteoclasts. New bone is constantly being produced while old bone is broken down. When tumor cells invade bone, the cancer cells produce 1 of 2 distinct substances; as a result, either osteoclasts or osteoblasts are stimulated, depending on tumor type metastasized to the bone. The activated osteoclasts then dissolve the bone, weakening the bone (osteolytic phenomenon), and the osteoblasts stimulate bone formation, hardening the bone (osteoblastic or sclerotic process).
Diagnosis and Evaluation
The most important first step in evaluating bone metastasis in a patient is to take a thorough, careful medical history and perform a physical examination. The examination not only helps locate suspected sites of bone metastases, but also helps determine necessary diagnostic studies.
The radiographic appearance of bone metastasis can be classified into 4 groups: osteolytic, osteoblastic, osteoporotic, and mixed. Imaging characteristics of osteolytic lesions include the destruction/thinning of bone, whereas osteoblastic (osteosclerotic) lesions appear with excess deposition of new bones. In contrast to malignant osteolytic lesions, osteoporotic lesions look like faded bone without cortical destruction or increased density.
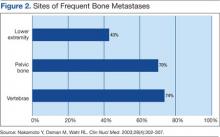
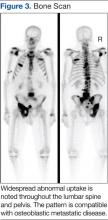
The main choice of imaging study for screening suspected bone metastases is usually the bone scan (Figure 3). Plain radiographs are not useful in the early detection of bone metastases, because bone lesions do not show up on plain films until 30% to 50% of the bone mineral is lost.5,9 Although most metastatic bone lesions represent a mixture of osteoblastic and -lytic processes, metastatic lesions of lung cancer and breast cancer are predominantly osteolytic in contrast to mainly osteoblastic lesions of prostate cancer metastases.10
The osteoblastic process of bone metastases is best demonstrated on a bone scan; however, a positive bone scan does not necessarily indicate bone metastases, because it is not highly specific of metastatic disease. Several benign bone lesions (such as osteoarthritis, traumatic injury, and Paget disease) also show positive readings. Magnetic resonance imaging (MRI) is not useful in screening for bone metastases, but it is better in assessing bone metastases compared with a bone scan, because it is more sensitive, especially for spinal lesions. The reported sensitivity of MRI is 91% to 100%, whereas bone scan sensitivity is only 62% to 85%.11,12
Even though the bone scan has been assumed to be the best imaging study for bone metastases, positron emission tomography (PET) scans can be more useful in detecting osteolytic bone metastases, as they can light up areas of increased metabolic activity. Positron emission tomography scans, however, are less sensitive for osteoblastic metastases. An additional advantage of PET scans is that they can be used for whole-body scanning/surveillance to rule out visceral involvement.
Published studies indicate that bone scans better detect sclerotic bone metastases and PET scans are superior in revealing osteolytic metastases.13-15 Furthermore, in contrast to bone scans, PET scans can identify additional lesions in addition to bone lesion. According to recent reports, PET provides higher sensitivity and specificity in demonstrating lytic and sclerotic metastases compared with that of the bone scan.16
Breast Cancer
The role of PET for breast cancer is controversial. A study by Lonneux and colleagues found that PET is highly sensitive in confirming distant metastasis from breast cancer, whereas researchers reported a similar sensitivity but higher specificity.17 Ohta and colleagues reported that PET and bone scan had identical sensitivity (77.7%), but PET was more specific than the bone scan (97.6% vs 80.9%, respectively).14 The study conclusion by Cook and colleagues was that PET is superior to bone scan in the detection of metastatic osteolytic bone lesions from breast cancer, whereas osteoblastic metastatic bone lesions from breast cancer are less likely to be demonstrated on a PET scan.18
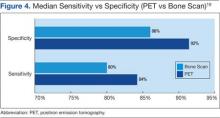
Houssami and Costelloe conducted a systematic review of 16 reported studies that comparatively tested the accuracy of imaging modalities for bone metastases in breast cancer.19 Sensitivity was generally similar between PET and bone scans in most studies reviewed. Four studies reported similar sensitivity but higher specificity for PET; the median specificity for PET and bone scan was 92% vs 85.5%, respectively (Figure 4).
Prostate Cancer
Prostate cancer is now established as the “classic” cancer for false-negative results on PET. Positron emission tomography does not perform well in the identification of osteoblastic skeletal metastases from prostate cancer. Yeh and colleagues reported only 18% positivity with PET.20 Interestingly, however, progressive metastatic prostate cancer showed a higher yield of 77% sensitivity with PET, perhaps because active osseous disease can be better picked up by PET scans.21
Related: Prostate Cancer Survivorship Care
Lung Cancer
For non-small cell lung cancer, both bone scan and PET showed a similar sensitivity for bone metastases detection, but the PET scan was more specific than the bone scan. Lung cancer often metastasizes to bone: up to 36% of patients at postmortem study. Lung cancer with bone metastases has a poor prognosis with median survival time typically measured in months. Most patients with bone metastases develop complications, such as severe pain, bone fracture, hypercalcemia, and spinal cord compression. Bone-targeted therapies play a greater role in the management of lung cancer patients, aiming for delaying disease progression and preserving QOL.22,23
Therapeutic Strategy and Management
Major morbidities associated with bone metastases include severe pain, hypercalcemia, bone fractures, spinal compression fractures, and cord or nerve root compression. This section reviews appropriate management techniques reported in the literature, particularly external beam radiation therapy.
Radiation Therapy
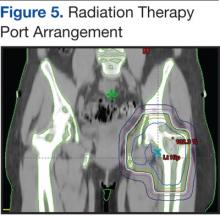
Pain is the most serious complication of bone metastases. Radiation therapy has been established as standard therapy and an effective pain palliation modality. Up to 80% of patients achieve partial pain relief, and > 33% of patients experience complete pain relief after radiation (Figure 5).24,25 Although a 3,000 cGy given over a 2-week period has been commonly used, a standard dose-fraction radiation treatment regimen has not been established.
The RTOG study was a randomized clinical study comparing various radiation schedules; 1,500 cGyin 1 week; vs 2,000 cGy in 1 week; vs 2,500 cGy in 1 week; vs 3,000 cGy in 2 weeks; or 4,050 cGy in 3 weeks. The conclusion was that local radiotherapy was an effective therapy for symptomatic and palliative therapy of bone metastases. Furthermore, low-dose radiotherapy was as good as various higher dose protracted courses of radiation treatments in terms of overall response rates (ORRs).24
Nearly 96% of patients eventually reported minimal pain relief to their palliative course of radiotherapy and experienced at least some pain relief within 4 weeks of radiation therapy. Complete pain relief was attained in 54% of patients regardless of the radiation dose-fraction schedules used. The median duration of complete pain response was about 12 weeks; > 70% of patients did not experience relapse of pain.26
Hartsell and colleagues investigated the efficacy of 800 cGy in a single fraction compared with 3,000 cGy in 10 fractions as part of a phase 3 randomized study of symptomatic therapy for pain palliation.27 The results showed 66% ORRs with similar complete and partial response rates (RRs) for both radiation groups. The complete RRs were 15% in the 800 cGy single-fraction arm vs 18% in the 3,000 cGy therapy arm, whereas partial RRs were 50% and 48% in the single vs the 3,000 cGy arms, respectively. However, there was a higher rate of retreatment for patients treated with the 800 cGy single-fraction radiotherapy. The 800 cGy single-fraction radiotherapy program seems rather popular in Canada and in European countries but is currently not widely used in the U.S.
Surgical Therapy
The surgical indications for managing bone metastases can vary, depending on disease location, surgeon’s preference, and patient’s overall disease status and related morbidities. Pain relief of fractured long bones (humerus, femur, or tibia) is crucial. The main goals of surgical intervention in these cases include the restoration of stability and functional mobility, pain control, and improving QOL. Weight-bearing bones (humerus/tibia) are especially at risk of bone fracture, and compromise of these is an indication of surgery. Postoperative external-beam radiation is recommended in most cases to eradicate residual microscopic disease or tumor progression.28
Radiopharmaceutical Therapy
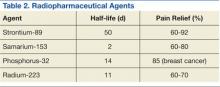
Bone-seeking radiopharmaceuticals are effective and have been widely used for pain palliation. The usual indications for radiopharmaceutical therapy include diffuse osteoblastic skeletal metastases demonstrated on bone scan, painful bone metastases not responding well to analgesics, and hormone-refractory metastatic prostate cancer. At present, strontium-89 (Sr-89), samarium-153 (Sm-153), phosphorus-32 (P-32), and radium 223 dichloride are radionuclides currently accepted as attractive therapeutic modalities for pain management (Table 2).
The clinical response is not immediate, and the average time to response is 1 to 2 weeks, but sometimes much longer. The main adverse reaction of systemic radiopharmaceutical therapy is myelotoxicity, such as thrombocytopenia and/or leukopenia. Occasionally, a so-called flare phenomenon of a transient pain increase may develop as well.29,30
Systemic Pharmacotherapy
Bisphosphonates are drugs commonly used to treat bone metastases. The benefits of bisphosphonate therapy are bone pain relief, the reduction of bone destruction, and the prevention of hypercalcemia and bone fractures. Bisphosphonates are typically more effective in osteolytic metastases and easily bind to bone, inhibiting bone resorption and increasing mineralization.31,32 Also, recent clinical studies suggest that bisphosphonates may inhibit tumor progression of bone metastases.
Related: Cancer Drugs Increase Rate of Preventable Hospital Admissions
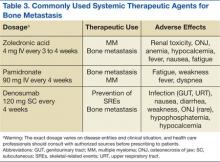
Zoledronic acid is currently one of the most potent bisphosphonates and is effective in most types of metastatic bone lesions.33 Denosumab, another drug, diminishes osteoclast activity, leading to decreased bone resorption and increased bone mass.34,35 Denosumab is useful in preventing complications as a result of bone metastases from solid tumors and has been recently approved by the FDA for treatment of postmenopausal osteoporosis and the prevention of skeletal-related events (SREs) in cancer patients with bone metastases.
Adverse Effects
Zoledronate and bisphosphonates in general are not recommended for patients with kidney disease, including hypocalcaemia and severe renal impairment. A rare but well-known complication of bisphosphonate administration is osteonecrosis of the jaw, which is somewhat more common in MM, especially after dental extractions. General nonspecific adverse effects include fatigue, anemia, muscle aches, fever, and/or edema in the feet or legs. Flulike symptoms and generalized bone discomfort can also be seen shortly after the first infusion (Table 3).
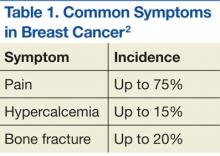
Breast Cancer
Bisphosphonates have been shown to effectively prevent SREs in breast cancer patients with bone metastases.36 For example, zoledronic acid is the most effective bisphosphonate and has been demonstrated to significantly delay the time to development of a first SRE, reducing the overall SRE rate by 43%.37
Lung Cancer
According to Rosen and colleagues, lung cancer patients with bone metastases who received zoledronic acid (4 mg every 3 weeks) experienced a 9% reduction in SREs, a relative delay in median time to a first SRE, and a significantly reduced incidence of SREs.37
Prostate Cancer
Zoledronic acid is the only bisphosphonate that proved effective in the treatment of prostate cancer patients with bone metastases. Zoledronic acid significantly reduced the risk of SREs (36%) and bone pain as well as delayed the median time to first SRE (nearly 6 months).38,39
Multiple Myeloma
Bisphosphonates are recommended for bone metastases to prevent new bone lesions. Studies have shown pamidronate (90 mg every 4 weeks) resulted in a 41% reduction in SREs at 9 months and a 25% reduction at 21 months.40,41 Oral clodronate, another agent, also significantly reduced SREs and pain in patients with MM.42
Conclusion
Metastatic cancer with bone metastases occurs as cancer advances and spreads to the bone from the primary site of the original solid cancer. Nearly 70% of patients with prostate and breast cancers and about 30% to 40% of patients with lung cancer develop bone metastases. In addition, up to 95% of MMs involve bone. The most frequent and important symptom of bone metastasis is pain. In addition, bone metastasis causes bone fractures, hypercalcemia, and spinal cord and nerve compression. Imaging studies, such as bone scans and PET studies, are useful tools in diagnosing bone metastases.
Therapeutic management of bone metastases is expanding and rapidly evolving. For better therapy outcomes, treatment should be both individualized and coordinated among the care team, including a medical oncologist, radiation oncologist, surgeon, and radiologist. Available therapeutic modalities include radiation therapy, radiopharmaceutical therapy, surgery, and systemic pharmacotherapy (zoledronate, pamidronate, and denosumab).
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225-249.
2. Cooleman RE. Metastatic bone disease: Clinical features, pathophysiology, and treatment strategies. Cancer Treat Rev. 2001;27(3):165-1763.
3. Hirabayashi H, Ebara S, Kinoshita T, et al. Clinical outcome and survival after palliative surgery for spinal metastases. Cancer. 2003;97(2):476-84.
4. van der Linden YM, Dijkstra SPDS, Vonk EJA, Marijnen CA, Leer JW; Dutch Bone Metastasis Study Group. Prediction of survival in patients with metastases in the spinal column. Cancer. 2005;103(2):320-328.
5. Coleman RE. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12(20, pt 2):6243S-6249S.
6. Body JJ. Metastatic bone disease: Clinical and therapeutic aspects. Bone. 1992;13(suppl 1):S57-S62.
7. Siegel RS, Ma J, Zou Z, Jermal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9-29.
8. National Cancer Institute. SEER stat fact sheets: Myeloma. National Cancer Institute Website. http://seer.cancer.gov/statfacts/html/mulmy.html. Accessed January 12, 2015.
9. Lentle BC, McGowan DG, Dierich H. Technetium-99M polyphosphate bone scanning in carcinoma of the prostate. Br J Urol. 1974;46(5):543-548.
10. Söderlund V. Radiological diagnosis of skeletal metastases. Eur Radiol. 1996;6(5):587-595.
11. Flickinger FW, Sanal SM. Bone marrow MRI: Techniques and accuracy for detecting breast cancer metastases. Magn Reson Imaging. 1994;12(6):829-35.
12. Hamaoka T, Madewell JE, Podoloff DA, Hortobagyi GN, Ueno NT. Bone imaging in metastatic breast cancer. J Clin Oncol. 2004;22(14):2942-2953.
13. Daldrup-Link HE, Franzius C, Link TM et al. Whole-body MR imaging for detection of bone metastases in children and young adults: Comparison with skeletal scintigraphy and FDG PET. AJR Am J Roentgenol. 2001;177(1):229-236.
14. Ohta M, Tokuda Y, Suzuki Y, et al. Whole body PET for the evaluation of bony metastases in patients with breast cancer: Comparison with 99Tcm-MDP bone scintigraphy. Nucl Med Commun. 2001;22(8):875-879.
15. Koolen BB, Vegt E, Rutgers EJ, et al. FDG-avid sclerotic bone metastases in breast cancer patients: A PET/CT case series. Ann Nucl Med. 2012;26(1):86-91.
16. Even-Sapir E, Metser U, Flusser G, et al. Assessment of malignant skeletal disease: Initial experience with 18F-fluoride PET/CT and comparison between 18F-fluoride PET and 18F-fluoride PET/CT. J Nucl Med. 2004;45(2):272-278.
17. Lonneux M, Borbath II, Berlière M, Kirkove C, Pauwels S. The place of whole-body PET FDG for the diagnosis of distant recurrence of breast cancer. Clin Positron Imaging. 2000;3(2):45-49.
18. Cook GJ, Houston S, Rubens R, Maisey MN, Fogelman I. Detection of bone metastases in breast cancer by 18FDG PET: Differing metabolic activity in osteoblastic and osteolytic lesions. J Clin Oncol. 1998;16(10):3375-3379.
19. Houssami N, Costelloe CM. Imaging bone metastases in breast cancer: Evidence on comparative test accuracy. Ann Oncol. 2012;23(4):834-843.
20. Yeh SD, Imbriaco M, Larson SM, et al. Detection of bony metastases of androgen-independent prostate cancer by PET-FDG. Nucl Med Biol. 1996;23(6):693-697.
21. Morris MJ, Akhurst T, Osman I, et al. Fluorinated deoxyglucose positron emission tomography imaging in progressive metastatic prostate cancer. Urology. 2002;59(6):913-918.
22. Rosen LS, Gordon D, Tchekmedyian S, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: A phase III, double-blind, randomized trial—the Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21(16):3150-3157.
23. Hillner BE, Ingle JN, Chlebowski RT, et al; American Society of Clinical Psychology. American Society of Clinical Oncology 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol. 2003;21(21):4042-4057.
24. Chow E, Harris K, Fan G, Tsao M, Size WM. Palliative radiotherapy trials for bone metastases: A systematic review. J Clin Oncol. 2007;25(11):1423-1436.
25. Wu JS, Wong R, Johnston M, Bezjak A, Whelan T; Cancer Care Ontario Practice Guidelines Initiative Supportive Care Group. Meta-analysis of dose-fractionation radiotherapy trials for the palliation of painful bone metastases. Int J Radiat Oncol Biol Phys. 2003;55(3):594-605.
26. Tong D, Gillick L, Hendrickson FR. The palliation of symptomatic osseous metastases. Final results of the study by the Radiation Therapy Oncology Group. Cancer. 1982;50(5):893-899.
27. Hartsell WF, Scott CB, Bruner DW, et al. Randomized trial of short- versus long-course radiotherapy for palliation of painful bone metastases. J Natl Cancer Inst. 2005;97(11):798-804.
28. Frassica DA. General principles of external beam radiation therapy for skeletal metastases. Clin Orthop Relat Res. 2003;415(suppl):S158-S164.
29. Silberstein EB. Systemic radiopharmaceutical therapy of painful osteoblastic metastases. Semin Radiat Oncol. 2000;10(3):240-249.
30. Neville-Webbe HL, Gnant M, Coleman RE. Potential anticancer properties of bisphosphonates. Semin Oncol. 2010;37(suppl 1):S53-S65.
31. Loftus LS, Edwards-Bennett S, Sokol GH. Systemic therapy for bone metastases. Cancer Control. 2012;19(2):145-153.
32. Rosen L, Harland SJ, Oosterlinck W. Broad clinical activity of zoledronic acid in osteolytic to osteoblastic bone lesions in patients with a broad range of solid tumors. Am J Clin Oncol. 2002;25(6)(suppl 1):S19-S24.
33. Fornier MN. Denosumab: Second chapter in controlling bone metastases or a new book? J Clin Oncol. 2010;28(35):5127-5131.
34. Mortimer JE, Pal SK. Safety considerations for use of bone-targeted agents in patients with cancer. Semin Oncol. 2010;37(suppl 1):S66-S72.
35. Pavlakis N, Schmidt R, Stockler M. Bisphosphonates for breast cancer. Cochrane Database Syst Rev. 2005;3:CD003474.
36. Kohno N, Aogi K, Minami H, et al. Zoledronic acid significantly reduces skeletal complications compared with placebo in Japanese women with bone metastases from breast cancer: A randomized, placebo-controlled trial. J Clin Oncol. 2005;23(15):3314-3321.
37. Rosen LS, Gordon D, Tchekmedyian NS, et al. Long-term efficacy and safety of zoledronic acid in the treatment of skeletal metastases in patients with nonsmall cell lung carcinoma and other solid tumors: A randomized, phase III, double-blind, placebo-controlled trial. Cancer. 2004;100(12):2613-2621.
38. Saad F, Gleason DM, Murray R, et al. Long-term efficacy of zoledronic acid for the prevention of skeletal complications in patients with metastatic hormone-refractory prostate cancer. J Natl Cancer Inst. 2004;96(11):879-882.
39. Saad F, Eastham J. Zoledronic acid improves clinical outcomes when administered before onset of bone pain in patients with prostate cancer. Urology. 2010;76(5):1175-1181.
40. Berenson JR, Lichtenstein A, Porter L, et al. Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. Myeloma Aredia Study Group. N Engl J Med. 1996;334(8):488-493.
41. Berenson JR, Lichtenstein A, Porter L, et al. Long-term pamidronate treatment of advanced multiple myeloma patients reduces skeletal events. Myeloma Aredia Study Group. J Clin Oncol. 1998;16(2):593-602.
42. Lahtinen R, Laakso M, Palva I, Virkkunen P, Elomaa I. Randomised, placebo-controlled multicentre trial of clodronate in multiple myeloma. Finnish Leukaemia Group. Lancet. 1992;340(8827):1049-1052.
Bone metastasis is a relatively common complication of cancer, often developing as they advance, especially in prostate cancer and breast cancer. Bone metastasis can profoundly affect patients’ daily activities and quality of life (QOL) due to severe pain and associated major complications. Prompt palliative therapy is required for symptomatic pain relief and prevention of the devastating complications of bone metastasis.
Epidemiology
Bone is the most common and preferred site for metastatic involvement of cancer. Advanced cancers frequently develop metastases to the bone during the later phases of cancer progression. At least 100,000 patients develop bone metastases every year, although the exact number of bone metastases is not known.1 Multiple myeloma (MM), breast cancer, and prostate cancer are responsible for up to 70% of bone metastases cases.2 Gastrointestinal cancers contribute least to bone metastases: < 15% of all cases.2
Related: Effective Treatment Options for Metastatic Pancreatic Cancer
The prognosis of bone metastases is generally poor, although it partly depends on the primary site of the original cancer and on the presence of any additional metastases to visceral organs. For example, it is known that survival times are longer for patients with primary prostate or breast cancer than for patients with lung cancer primary tumors.3,4
Prostate and breast cancers are the most common primary cancers of bone metastases. At postmortem studies, patients who died of prostate cancer or breast cancer revealed evidence of bone metastases in up to 75% of cases (Figure 1). Regardless of their survival expectancy, however, most patients with bone metastasis need immediate medical attention and active palliative therapy to prevent devastating complications related to bone metastasis, such as pathologic bone fractures and severe bone pain.
Clinical Features
Multiple Myeloma
Multiple myeloma is the second most common hematologic malignancy and is caused by an abnormal accumulation of clonal plasma cells in the bone marrow. Characteristic clinical manifestations include bony destruction and related features of bone pain, anemia (80% of cases), hypocalcemia, and renal dysfunction. Pathologic fractures, renal failure, or hyperviscosity syndrome often develops. More than 20,000 new patients are diagnosed with MM and about 11,000 patients in the U.S. die of MM every year. Multiple myeloma and is twice as likely to develop in men as it is in women. A large number of MM cases are under the care of VAMCs (about 10%-12% of all MM cases).7,8
Abnormal laboratory tests show an elevated total protein level in the blood and/or urine (Bence Jones proteinuria). Serum electrophoresis detects M-protein in about 80% to 90% of patients. Patients may also present with renal failure. The differential diagnosis includes other malignancies, such as metastatic carcinoma, lymphoma, leukemia, and monoclonal gammopathy.
Pathophysiology
Normal bone tissue is made up of 2 different types of cells: osteoblasts and osteoclasts. New bone is constantly being produced while old bone is broken down. When tumor cells invade bone, the cancer cells produce 1 of 2 distinct substances; as a result, either osteoclasts or osteoblasts are stimulated, depending on tumor type metastasized to the bone. The activated osteoclasts then dissolve the bone, weakening the bone (osteolytic phenomenon), and the osteoblasts stimulate bone formation, hardening the bone (osteoblastic or sclerotic process).
Diagnosis and Evaluation
The most important first step in evaluating bone metastasis in a patient is to take a thorough, careful medical history and perform a physical examination. The examination not only helps locate suspected sites of bone metastases, but also helps determine necessary diagnostic studies.
The radiographic appearance of bone metastasis can be classified into 4 groups: osteolytic, osteoblastic, osteoporotic, and mixed. Imaging characteristics of osteolytic lesions include the destruction/thinning of bone, whereas osteoblastic (osteosclerotic) lesions appear with excess deposition of new bones. In contrast to malignant osteolytic lesions, osteoporotic lesions look like faded bone without cortical destruction or increased density.
The main choice of imaging study for screening suspected bone metastases is usually the bone scan (Figure 3). Plain radiographs are not useful in the early detection of bone metastases, because bone lesions do not show up on plain films until 30% to 50% of the bone mineral is lost.5,9 Although most metastatic bone lesions represent a mixture of osteoblastic and -lytic processes, metastatic lesions of lung cancer and breast cancer are predominantly osteolytic in contrast to mainly osteoblastic lesions of prostate cancer metastases.10
The osteoblastic process of bone metastases is best demonstrated on a bone scan; however, a positive bone scan does not necessarily indicate bone metastases, because it is not highly specific of metastatic disease. Several benign bone lesions (such as osteoarthritis, traumatic injury, and Paget disease) also show positive readings. Magnetic resonance imaging (MRI) is not useful in screening for bone metastases, but it is better in assessing bone metastases compared with a bone scan, because it is more sensitive, especially for spinal lesions. The reported sensitivity of MRI is 91% to 100%, whereas bone scan sensitivity is only 62% to 85%.11,12
Even though the bone scan has been assumed to be the best imaging study for bone metastases, positron emission tomography (PET) scans can be more useful in detecting osteolytic bone metastases, as they can light up areas of increased metabolic activity. Positron emission tomography scans, however, are less sensitive for osteoblastic metastases. An additional advantage of PET scans is that they can be used for whole-body scanning/surveillance to rule out visceral involvement.
Published studies indicate that bone scans better detect sclerotic bone metastases and PET scans are superior in revealing osteolytic metastases.13-15 Furthermore, in contrast to bone scans, PET scans can identify additional lesions in addition to bone lesion. According to recent reports, PET provides higher sensitivity and specificity in demonstrating lytic and sclerotic metastases compared with that of the bone scan.16
Breast Cancer
The role of PET for breast cancer is controversial. A study by Lonneux and colleagues found that PET is highly sensitive in confirming distant metastasis from breast cancer, whereas researchers reported a similar sensitivity but higher specificity.17 Ohta and colleagues reported that PET and bone scan had identical sensitivity (77.7%), but PET was more specific than the bone scan (97.6% vs 80.9%, respectively).14 The study conclusion by Cook and colleagues was that PET is superior to bone scan in the detection of metastatic osteolytic bone lesions from breast cancer, whereas osteoblastic metastatic bone lesions from breast cancer are less likely to be demonstrated on a PET scan.18
Houssami and Costelloe conducted a systematic review of 16 reported studies that comparatively tested the accuracy of imaging modalities for bone metastases in breast cancer.19 Sensitivity was generally similar between PET and bone scans in most studies reviewed. Four studies reported similar sensitivity but higher specificity for PET; the median specificity for PET and bone scan was 92% vs 85.5%, respectively (Figure 4).
Prostate Cancer
Prostate cancer is now established as the “classic” cancer for false-negative results on PET. Positron emission tomography does not perform well in the identification of osteoblastic skeletal metastases from prostate cancer. Yeh and colleagues reported only 18% positivity with PET.20 Interestingly, however, progressive metastatic prostate cancer showed a higher yield of 77% sensitivity with PET, perhaps because active osseous disease can be better picked up by PET scans.21
Related: Prostate Cancer Survivorship Care
Lung Cancer
For non-small cell lung cancer, both bone scan and PET showed a similar sensitivity for bone metastases detection, but the PET scan was more specific than the bone scan. Lung cancer often metastasizes to bone: up to 36% of patients at postmortem study. Lung cancer with bone metastases has a poor prognosis with median survival time typically measured in months. Most patients with bone metastases develop complications, such as severe pain, bone fracture, hypercalcemia, and spinal cord compression. Bone-targeted therapies play a greater role in the management of lung cancer patients, aiming for delaying disease progression and preserving QOL.22,23
Therapeutic Strategy and Management
Major morbidities associated with bone metastases include severe pain, hypercalcemia, bone fractures, spinal compression fractures, and cord or nerve root compression. This section reviews appropriate management techniques reported in the literature, particularly external beam radiation therapy.
Radiation Therapy
Pain is the most serious complication of bone metastases. Radiation therapy has been established as standard therapy and an effective pain palliation modality. Up to 80% of patients achieve partial pain relief, and > 33% of patients experience complete pain relief after radiation (Figure 5).24,25 Although a 3,000 cGy given over a 2-week period has been commonly used, a standard dose-fraction radiation treatment regimen has not been established.
The RTOG study was a randomized clinical study comparing various radiation schedules; 1,500 cGyin 1 week; vs 2,000 cGy in 1 week; vs 2,500 cGy in 1 week; vs 3,000 cGy in 2 weeks; or 4,050 cGy in 3 weeks. The conclusion was that local radiotherapy was an effective therapy for symptomatic and palliative therapy of bone metastases. Furthermore, low-dose radiotherapy was as good as various higher dose protracted courses of radiation treatments in terms of overall response rates (ORRs).24
Nearly 96% of patients eventually reported minimal pain relief to their palliative course of radiotherapy and experienced at least some pain relief within 4 weeks of radiation therapy. Complete pain relief was attained in 54% of patients regardless of the radiation dose-fraction schedules used. The median duration of complete pain response was about 12 weeks; > 70% of patients did not experience relapse of pain.26
Hartsell and colleagues investigated the efficacy of 800 cGy in a single fraction compared with 3,000 cGy in 10 fractions as part of a phase 3 randomized study of symptomatic therapy for pain palliation.27 The results showed 66% ORRs with similar complete and partial response rates (RRs) for both radiation groups. The complete RRs were 15% in the 800 cGy single-fraction arm vs 18% in the 3,000 cGy therapy arm, whereas partial RRs were 50% and 48% in the single vs the 3,000 cGy arms, respectively. However, there was a higher rate of retreatment for patients treated with the 800 cGy single-fraction radiotherapy. The 800 cGy single-fraction radiotherapy program seems rather popular in Canada and in European countries but is currently not widely used in the U.S.
Surgical Therapy
The surgical indications for managing bone metastases can vary, depending on disease location, surgeon’s preference, and patient’s overall disease status and related morbidities. Pain relief of fractured long bones (humerus, femur, or tibia) is crucial. The main goals of surgical intervention in these cases include the restoration of stability and functional mobility, pain control, and improving QOL. Weight-bearing bones (humerus/tibia) are especially at risk of bone fracture, and compromise of these is an indication of surgery. Postoperative external-beam radiation is recommended in most cases to eradicate residual microscopic disease or tumor progression.28
Radiopharmaceutical Therapy
Bone-seeking radiopharmaceuticals are effective and have been widely used for pain palliation. The usual indications for radiopharmaceutical therapy include diffuse osteoblastic skeletal metastases demonstrated on bone scan, painful bone metastases not responding well to analgesics, and hormone-refractory metastatic prostate cancer. At present, strontium-89 (Sr-89), samarium-153 (Sm-153), phosphorus-32 (P-32), and radium 223 dichloride are radionuclides currently accepted as attractive therapeutic modalities for pain management (Table 2).
The clinical response is not immediate, and the average time to response is 1 to 2 weeks, but sometimes much longer. The main adverse reaction of systemic radiopharmaceutical therapy is myelotoxicity, such as thrombocytopenia and/or leukopenia. Occasionally, a so-called flare phenomenon of a transient pain increase may develop as well.29,30
Systemic Pharmacotherapy
Bisphosphonates are drugs commonly used to treat bone metastases. The benefits of bisphosphonate therapy are bone pain relief, the reduction of bone destruction, and the prevention of hypercalcemia and bone fractures. Bisphosphonates are typically more effective in osteolytic metastases and easily bind to bone, inhibiting bone resorption and increasing mineralization.31,32 Also, recent clinical studies suggest that bisphosphonates may inhibit tumor progression of bone metastases.
Related: Cancer Drugs Increase Rate of Preventable Hospital Admissions
Zoledronic acid is currently one of the most potent bisphosphonates and is effective in most types of metastatic bone lesions.33 Denosumab, another drug, diminishes osteoclast activity, leading to decreased bone resorption and increased bone mass.34,35 Denosumab is useful in preventing complications as a result of bone metastases from solid tumors and has been recently approved by the FDA for treatment of postmenopausal osteoporosis and the prevention of skeletal-related events (SREs) in cancer patients with bone metastases.
Adverse Effects
Zoledronate and bisphosphonates in general are not recommended for patients with kidney disease, including hypocalcaemia and severe renal impairment. A rare but well-known complication of bisphosphonate administration is osteonecrosis of the jaw, which is somewhat more common in MM, especially after dental extractions. General nonspecific adverse effects include fatigue, anemia, muscle aches, fever, and/or edema in the feet or legs. Flulike symptoms and generalized bone discomfort can also be seen shortly after the first infusion (Table 3).
Breast Cancer
Bisphosphonates have been shown to effectively prevent SREs in breast cancer patients with bone metastases.36 For example, zoledronic acid is the most effective bisphosphonate and has been demonstrated to significantly delay the time to development of a first SRE, reducing the overall SRE rate by 43%.37
Lung Cancer
According to Rosen and colleagues, lung cancer patients with bone metastases who received zoledronic acid (4 mg every 3 weeks) experienced a 9% reduction in SREs, a relative delay in median time to a first SRE, and a significantly reduced incidence of SREs.37
Prostate Cancer
Zoledronic acid is the only bisphosphonate that proved effective in the treatment of prostate cancer patients with bone metastases. Zoledronic acid significantly reduced the risk of SREs (36%) and bone pain as well as delayed the median time to first SRE (nearly 6 months).38,39
Multiple Myeloma
Bisphosphonates are recommended for bone metastases to prevent new bone lesions. Studies have shown pamidronate (90 mg every 4 weeks) resulted in a 41% reduction in SREs at 9 months and a 25% reduction at 21 months.40,41 Oral clodronate, another agent, also significantly reduced SREs and pain in patients with MM.42
Conclusion
Metastatic cancer with bone metastases occurs as cancer advances and spreads to the bone from the primary site of the original solid cancer. Nearly 70% of patients with prostate and breast cancers and about 30% to 40% of patients with lung cancer develop bone metastases. In addition, up to 95% of MMs involve bone. The most frequent and important symptom of bone metastasis is pain. In addition, bone metastasis causes bone fractures, hypercalcemia, and spinal cord and nerve compression. Imaging studies, such as bone scans and PET studies, are useful tools in diagnosing bone metastases.
Therapeutic management of bone metastases is expanding and rapidly evolving. For better therapy outcomes, treatment should be both individualized and coordinated among the care team, including a medical oncologist, radiation oncologist, surgeon, and radiologist. Available therapeutic modalities include radiation therapy, radiopharmaceutical therapy, surgery, and systemic pharmacotherapy (zoledronate, pamidronate, and denosumab).
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Bone metastasis is a relatively common complication of cancer, often developing as they advance, especially in prostate cancer and breast cancer. Bone metastasis can profoundly affect patients’ daily activities and quality of life (QOL) due to severe pain and associated major complications. Prompt palliative therapy is required for symptomatic pain relief and prevention of the devastating complications of bone metastasis.
Epidemiology
Bone is the most common and preferred site for metastatic involvement of cancer. Advanced cancers frequently develop metastases to the bone during the later phases of cancer progression. At least 100,000 patients develop bone metastases every year, although the exact number of bone metastases is not known.1 Multiple myeloma (MM), breast cancer, and prostate cancer are responsible for up to 70% of bone metastases cases.2 Gastrointestinal cancers contribute least to bone metastases: < 15% of all cases.2
Related: Effective Treatment Options for Metastatic Pancreatic Cancer
The prognosis of bone metastases is generally poor, although it partly depends on the primary site of the original cancer and on the presence of any additional metastases to visceral organs. For example, it is known that survival times are longer for patients with primary prostate or breast cancer than for patients with lung cancer primary tumors.3,4
Prostate and breast cancers are the most common primary cancers of bone metastases. At postmortem studies, patients who died of prostate cancer or breast cancer revealed evidence of bone metastases in up to 75% of cases (Figure 1). Regardless of their survival expectancy, however, most patients with bone metastasis need immediate medical attention and active palliative therapy to prevent devastating complications related to bone metastasis, such as pathologic bone fractures and severe bone pain.
Clinical Features
Multiple Myeloma
Multiple myeloma is the second most common hematologic malignancy and is caused by an abnormal accumulation of clonal plasma cells in the bone marrow. Characteristic clinical manifestations include bony destruction and related features of bone pain, anemia (80% of cases), hypocalcemia, and renal dysfunction. Pathologic fractures, renal failure, or hyperviscosity syndrome often develops. More than 20,000 new patients are diagnosed with MM and about 11,000 patients in the U.S. die of MM every year. Multiple myeloma and is twice as likely to develop in men as it is in women. A large number of MM cases are under the care of VAMCs (about 10%-12% of all MM cases).7,8
Abnormal laboratory tests show an elevated total protein level in the blood and/or urine (Bence Jones proteinuria). Serum electrophoresis detects M-protein in about 80% to 90% of patients. Patients may also present with renal failure. The differential diagnosis includes other malignancies, such as metastatic carcinoma, lymphoma, leukemia, and monoclonal gammopathy.
Pathophysiology
Normal bone tissue is made up of 2 different types of cells: osteoblasts and osteoclasts. New bone is constantly being produced while old bone is broken down. When tumor cells invade bone, the cancer cells produce 1 of 2 distinct substances; as a result, either osteoclasts or osteoblasts are stimulated, depending on tumor type metastasized to the bone. The activated osteoclasts then dissolve the bone, weakening the bone (osteolytic phenomenon), and the osteoblasts stimulate bone formation, hardening the bone (osteoblastic or sclerotic process).
Diagnosis and Evaluation
The most important first step in evaluating bone metastasis in a patient is to take a thorough, careful medical history and perform a physical examination. The examination not only helps locate suspected sites of bone metastases, but also helps determine necessary diagnostic studies.
The radiographic appearance of bone metastasis can be classified into 4 groups: osteolytic, osteoblastic, osteoporotic, and mixed. Imaging characteristics of osteolytic lesions include the destruction/thinning of bone, whereas osteoblastic (osteosclerotic) lesions appear with excess deposition of new bones. In contrast to malignant osteolytic lesions, osteoporotic lesions look like faded bone without cortical destruction or increased density.
The main choice of imaging study for screening suspected bone metastases is usually the bone scan (Figure 3). Plain radiographs are not useful in the early detection of bone metastases, because bone lesions do not show up on plain films until 30% to 50% of the bone mineral is lost.5,9 Although most metastatic bone lesions represent a mixture of osteoblastic and -lytic processes, metastatic lesions of lung cancer and breast cancer are predominantly osteolytic in contrast to mainly osteoblastic lesions of prostate cancer metastases.10
The osteoblastic process of bone metastases is best demonstrated on a bone scan; however, a positive bone scan does not necessarily indicate bone metastases, because it is not highly specific of metastatic disease. Several benign bone lesions (such as osteoarthritis, traumatic injury, and Paget disease) also show positive readings. Magnetic resonance imaging (MRI) is not useful in screening for bone metastases, but it is better in assessing bone metastases compared with a bone scan, because it is more sensitive, especially for spinal lesions. The reported sensitivity of MRI is 91% to 100%, whereas bone scan sensitivity is only 62% to 85%.11,12
Even though the bone scan has been assumed to be the best imaging study for bone metastases, positron emission tomography (PET) scans can be more useful in detecting osteolytic bone metastases, as they can light up areas of increased metabolic activity. Positron emission tomography scans, however, are less sensitive for osteoblastic metastases. An additional advantage of PET scans is that they can be used for whole-body scanning/surveillance to rule out visceral involvement.
Published studies indicate that bone scans better detect sclerotic bone metastases and PET scans are superior in revealing osteolytic metastases.13-15 Furthermore, in contrast to bone scans, PET scans can identify additional lesions in addition to bone lesion. According to recent reports, PET provides higher sensitivity and specificity in demonstrating lytic and sclerotic metastases compared with that of the bone scan.16
Breast Cancer
The role of PET for breast cancer is controversial. A study by Lonneux and colleagues found that PET is highly sensitive in confirming distant metastasis from breast cancer, whereas researchers reported a similar sensitivity but higher specificity.17 Ohta and colleagues reported that PET and bone scan had identical sensitivity (77.7%), but PET was more specific than the bone scan (97.6% vs 80.9%, respectively).14 The study conclusion by Cook and colleagues was that PET is superior to bone scan in the detection of metastatic osteolytic bone lesions from breast cancer, whereas osteoblastic metastatic bone lesions from breast cancer are less likely to be demonstrated on a PET scan.18
Houssami and Costelloe conducted a systematic review of 16 reported studies that comparatively tested the accuracy of imaging modalities for bone metastases in breast cancer.19 Sensitivity was generally similar between PET and bone scans in most studies reviewed. Four studies reported similar sensitivity but higher specificity for PET; the median specificity for PET and bone scan was 92% vs 85.5%, respectively (Figure 4).
Prostate Cancer
Prostate cancer is now established as the “classic” cancer for false-negative results on PET. Positron emission tomography does not perform well in the identification of osteoblastic skeletal metastases from prostate cancer. Yeh and colleagues reported only 18% positivity with PET.20 Interestingly, however, progressive metastatic prostate cancer showed a higher yield of 77% sensitivity with PET, perhaps because active osseous disease can be better picked up by PET scans.21
Related: Prostate Cancer Survivorship Care
Lung Cancer
For non-small cell lung cancer, both bone scan and PET showed a similar sensitivity for bone metastases detection, but the PET scan was more specific than the bone scan. Lung cancer often metastasizes to bone: up to 36% of patients at postmortem study. Lung cancer with bone metastases has a poor prognosis with median survival time typically measured in months. Most patients with bone metastases develop complications, such as severe pain, bone fracture, hypercalcemia, and spinal cord compression. Bone-targeted therapies play a greater role in the management of lung cancer patients, aiming for delaying disease progression and preserving QOL.22,23
Therapeutic Strategy and Management
Major morbidities associated with bone metastases include severe pain, hypercalcemia, bone fractures, spinal compression fractures, and cord or nerve root compression. This section reviews appropriate management techniques reported in the literature, particularly external beam radiation therapy.
Radiation Therapy
Pain is the most serious complication of bone metastases. Radiation therapy has been established as standard therapy and an effective pain palliation modality. Up to 80% of patients achieve partial pain relief, and > 33% of patients experience complete pain relief after radiation (Figure 5).24,25 Although a 3,000 cGy given over a 2-week period has been commonly used, a standard dose-fraction radiation treatment regimen has not been established.
The RTOG study was a randomized clinical study comparing various radiation schedules; 1,500 cGyin 1 week; vs 2,000 cGy in 1 week; vs 2,500 cGy in 1 week; vs 3,000 cGy in 2 weeks; or 4,050 cGy in 3 weeks. The conclusion was that local radiotherapy was an effective therapy for symptomatic and palliative therapy of bone metastases. Furthermore, low-dose radiotherapy was as good as various higher dose protracted courses of radiation treatments in terms of overall response rates (ORRs).24
Nearly 96% of patients eventually reported minimal pain relief to their palliative course of radiotherapy and experienced at least some pain relief within 4 weeks of radiation therapy. Complete pain relief was attained in 54% of patients regardless of the radiation dose-fraction schedules used. The median duration of complete pain response was about 12 weeks; > 70% of patients did not experience relapse of pain.26
Hartsell and colleagues investigated the efficacy of 800 cGy in a single fraction compared with 3,000 cGy in 10 fractions as part of a phase 3 randomized study of symptomatic therapy for pain palliation.27 The results showed 66% ORRs with similar complete and partial response rates (RRs) for both radiation groups. The complete RRs were 15% in the 800 cGy single-fraction arm vs 18% in the 3,000 cGy therapy arm, whereas partial RRs were 50% and 48% in the single vs the 3,000 cGy arms, respectively. However, there was a higher rate of retreatment for patients treated with the 800 cGy single-fraction radiotherapy. The 800 cGy single-fraction radiotherapy program seems rather popular in Canada and in European countries but is currently not widely used in the U.S.
Surgical Therapy
The surgical indications for managing bone metastases can vary, depending on disease location, surgeon’s preference, and patient’s overall disease status and related morbidities. Pain relief of fractured long bones (humerus, femur, or tibia) is crucial. The main goals of surgical intervention in these cases include the restoration of stability and functional mobility, pain control, and improving QOL. Weight-bearing bones (humerus/tibia) are especially at risk of bone fracture, and compromise of these is an indication of surgery. Postoperative external-beam radiation is recommended in most cases to eradicate residual microscopic disease or tumor progression.28
Radiopharmaceutical Therapy
Bone-seeking radiopharmaceuticals are effective and have been widely used for pain palliation. The usual indications for radiopharmaceutical therapy include diffuse osteoblastic skeletal metastases demonstrated on bone scan, painful bone metastases not responding well to analgesics, and hormone-refractory metastatic prostate cancer. At present, strontium-89 (Sr-89), samarium-153 (Sm-153), phosphorus-32 (P-32), and radium 223 dichloride are radionuclides currently accepted as attractive therapeutic modalities for pain management (Table 2).
The clinical response is not immediate, and the average time to response is 1 to 2 weeks, but sometimes much longer. The main adverse reaction of systemic radiopharmaceutical therapy is myelotoxicity, such as thrombocytopenia and/or leukopenia. Occasionally, a so-called flare phenomenon of a transient pain increase may develop as well.29,30
Systemic Pharmacotherapy
Bisphosphonates are drugs commonly used to treat bone metastases. The benefits of bisphosphonate therapy are bone pain relief, the reduction of bone destruction, and the prevention of hypercalcemia and bone fractures. Bisphosphonates are typically more effective in osteolytic metastases and easily bind to bone, inhibiting bone resorption and increasing mineralization.31,32 Also, recent clinical studies suggest that bisphosphonates may inhibit tumor progression of bone metastases.
Related: Cancer Drugs Increase Rate of Preventable Hospital Admissions
Zoledronic acid is currently one of the most potent bisphosphonates and is effective in most types of metastatic bone lesions.33 Denosumab, another drug, diminishes osteoclast activity, leading to decreased bone resorption and increased bone mass.34,35 Denosumab is useful in preventing complications as a result of bone metastases from solid tumors and has been recently approved by the FDA for treatment of postmenopausal osteoporosis and the prevention of skeletal-related events (SREs) in cancer patients with bone metastases.
Adverse Effects
Zoledronate and bisphosphonates in general are not recommended for patients with kidney disease, including hypocalcaemia and severe renal impairment. A rare but well-known complication of bisphosphonate administration is osteonecrosis of the jaw, which is somewhat more common in MM, especially after dental extractions. General nonspecific adverse effects include fatigue, anemia, muscle aches, fever, and/or edema in the feet or legs. Flulike symptoms and generalized bone discomfort can also be seen shortly after the first infusion (Table 3).
Breast Cancer
Bisphosphonates have been shown to effectively prevent SREs in breast cancer patients with bone metastases.36 For example, zoledronic acid is the most effective bisphosphonate and has been demonstrated to significantly delay the time to development of a first SRE, reducing the overall SRE rate by 43%.37
Lung Cancer
According to Rosen and colleagues, lung cancer patients with bone metastases who received zoledronic acid (4 mg every 3 weeks) experienced a 9% reduction in SREs, a relative delay in median time to a first SRE, and a significantly reduced incidence of SREs.37
Prostate Cancer
Zoledronic acid is the only bisphosphonate that proved effective in the treatment of prostate cancer patients with bone metastases. Zoledronic acid significantly reduced the risk of SREs (36%) and bone pain as well as delayed the median time to first SRE (nearly 6 months).38,39
Multiple Myeloma
Bisphosphonates are recommended for bone metastases to prevent new bone lesions. Studies have shown pamidronate (90 mg every 4 weeks) resulted in a 41% reduction in SREs at 9 months and a 25% reduction at 21 months.40,41 Oral clodronate, another agent, also significantly reduced SREs and pain in patients with MM.42
Conclusion
Metastatic cancer with bone metastases occurs as cancer advances and spreads to the bone from the primary site of the original solid cancer. Nearly 70% of patients with prostate and breast cancers and about 30% to 40% of patients with lung cancer develop bone metastases. In addition, up to 95% of MMs involve bone. The most frequent and important symptom of bone metastasis is pain. In addition, bone metastasis causes bone fractures, hypercalcemia, and spinal cord and nerve compression. Imaging studies, such as bone scans and PET studies, are useful tools in diagnosing bone metastases.
Therapeutic management of bone metastases is expanding and rapidly evolving. For better therapy outcomes, treatment should be both individualized and coordinated among the care team, including a medical oncologist, radiation oncologist, surgeon, and radiologist. Available therapeutic modalities include radiation therapy, radiopharmaceutical therapy, surgery, and systemic pharmacotherapy (zoledronate, pamidronate, and denosumab).
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review the complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
1. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225-249.
2. Cooleman RE. Metastatic bone disease: Clinical features, pathophysiology, and treatment strategies. Cancer Treat Rev. 2001;27(3):165-1763.
3. Hirabayashi H, Ebara S, Kinoshita T, et al. Clinical outcome and survival after palliative surgery for spinal metastases. Cancer. 2003;97(2):476-84.
4. van der Linden YM, Dijkstra SPDS, Vonk EJA, Marijnen CA, Leer JW; Dutch Bone Metastasis Study Group. Prediction of survival in patients with metastases in the spinal column. Cancer. 2005;103(2):320-328.
5. Coleman RE. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12(20, pt 2):6243S-6249S.
6. Body JJ. Metastatic bone disease: Clinical and therapeutic aspects. Bone. 1992;13(suppl 1):S57-S62.
7. Siegel RS, Ma J, Zou Z, Jermal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9-29.
8. National Cancer Institute. SEER stat fact sheets: Myeloma. National Cancer Institute Website. http://seer.cancer.gov/statfacts/html/mulmy.html. Accessed January 12, 2015.
9. Lentle BC, McGowan DG, Dierich H. Technetium-99M polyphosphate bone scanning in carcinoma of the prostate. Br J Urol. 1974;46(5):543-548.
10. Söderlund V. Radiological diagnosis of skeletal metastases. Eur Radiol. 1996;6(5):587-595.
11. Flickinger FW, Sanal SM. Bone marrow MRI: Techniques and accuracy for detecting breast cancer metastases. Magn Reson Imaging. 1994;12(6):829-35.
12. Hamaoka T, Madewell JE, Podoloff DA, Hortobagyi GN, Ueno NT. Bone imaging in metastatic breast cancer. J Clin Oncol. 2004;22(14):2942-2953.
13. Daldrup-Link HE, Franzius C, Link TM et al. Whole-body MR imaging for detection of bone metastases in children and young adults: Comparison with skeletal scintigraphy and FDG PET. AJR Am J Roentgenol. 2001;177(1):229-236.
14. Ohta M, Tokuda Y, Suzuki Y, et al. Whole body PET for the evaluation of bony metastases in patients with breast cancer: Comparison with 99Tcm-MDP bone scintigraphy. Nucl Med Commun. 2001;22(8):875-879.
15. Koolen BB, Vegt E, Rutgers EJ, et al. FDG-avid sclerotic bone metastases in breast cancer patients: A PET/CT case series. Ann Nucl Med. 2012;26(1):86-91.
16. Even-Sapir E, Metser U, Flusser G, et al. Assessment of malignant skeletal disease: Initial experience with 18F-fluoride PET/CT and comparison between 18F-fluoride PET and 18F-fluoride PET/CT. J Nucl Med. 2004;45(2):272-278.
17. Lonneux M, Borbath II, Berlière M, Kirkove C, Pauwels S. The place of whole-body PET FDG for the diagnosis of distant recurrence of breast cancer. Clin Positron Imaging. 2000;3(2):45-49.
18. Cook GJ, Houston S, Rubens R, Maisey MN, Fogelman I. Detection of bone metastases in breast cancer by 18FDG PET: Differing metabolic activity in osteoblastic and osteolytic lesions. J Clin Oncol. 1998;16(10):3375-3379.
19. Houssami N, Costelloe CM. Imaging bone metastases in breast cancer: Evidence on comparative test accuracy. Ann Oncol. 2012;23(4):834-843.
20. Yeh SD, Imbriaco M, Larson SM, et al. Detection of bony metastases of androgen-independent prostate cancer by PET-FDG. Nucl Med Biol. 1996;23(6):693-697.
21. Morris MJ, Akhurst T, Osman I, et al. Fluorinated deoxyglucose positron emission tomography imaging in progressive metastatic prostate cancer. Urology. 2002;59(6):913-918.
22. Rosen LS, Gordon D, Tchekmedyian S, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: A phase III, double-blind, randomized trial—the Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21(16):3150-3157.
23. Hillner BE, Ingle JN, Chlebowski RT, et al; American Society of Clinical Psychology. American Society of Clinical Oncology 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol. 2003;21(21):4042-4057.
24. Chow E, Harris K, Fan G, Tsao M, Size WM. Palliative radiotherapy trials for bone metastases: A systematic review. J Clin Oncol. 2007;25(11):1423-1436.
25. Wu JS, Wong R, Johnston M, Bezjak A, Whelan T; Cancer Care Ontario Practice Guidelines Initiative Supportive Care Group. Meta-analysis of dose-fractionation radiotherapy trials for the palliation of painful bone metastases. Int J Radiat Oncol Biol Phys. 2003;55(3):594-605.
26. Tong D, Gillick L, Hendrickson FR. The palliation of symptomatic osseous metastases. Final results of the study by the Radiation Therapy Oncology Group. Cancer. 1982;50(5):893-899.
27. Hartsell WF, Scott CB, Bruner DW, et al. Randomized trial of short- versus long-course radiotherapy for palliation of painful bone metastases. J Natl Cancer Inst. 2005;97(11):798-804.
28. Frassica DA. General principles of external beam radiation therapy for skeletal metastases. Clin Orthop Relat Res. 2003;415(suppl):S158-S164.
29. Silberstein EB. Systemic radiopharmaceutical therapy of painful osteoblastic metastases. Semin Radiat Oncol. 2000;10(3):240-249.
30. Neville-Webbe HL, Gnant M, Coleman RE. Potential anticancer properties of bisphosphonates. Semin Oncol. 2010;37(suppl 1):S53-S65.
31. Loftus LS, Edwards-Bennett S, Sokol GH. Systemic therapy for bone metastases. Cancer Control. 2012;19(2):145-153.
32. Rosen L, Harland SJ, Oosterlinck W. Broad clinical activity of zoledronic acid in osteolytic to osteoblastic bone lesions in patients with a broad range of solid tumors. Am J Clin Oncol. 2002;25(6)(suppl 1):S19-S24.
33. Fornier MN. Denosumab: Second chapter in controlling bone metastases or a new book? J Clin Oncol. 2010;28(35):5127-5131.
34. Mortimer JE, Pal SK. Safety considerations for use of bone-targeted agents in patients with cancer. Semin Oncol. 2010;37(suppl 1):S66-S72.
35. Pavlakis N, Schmidt R, Stockler M. Bisphosphonates for breast cancer. Cochrane Database Syst Rev. 2005;3:CD003474.
36. Kohno N, Aogi K, Minami H, et al. Zoledronic acid significantly reduces skeletal complications compared with placebo in Japanese women with bone metastases from breast cancer: A randomized, placebo-controlled trial. J Clin Oncol. 2005;23(15):3314-3321.
37. Rosen LS, Gordon D, Tchekmedyian NS, et al. Long-term efficacy and safety of zoledronic acid in the treatment of skeletal metastases in patients with nonsmall cell lung carcinoma and other solid tumors: A randomized, phase III, double-blind, placebo-controlled trial. Cancer. 2004;100(12):2613-2621.
38. Saad F, Gleason DM, Murray R, et al. Long-term efficacy of zoledronic acid for the prevention of skeletal complications in patients with metastatic hormone-refractory prostate cancer. J Natl Cancer Inst. 2004;96(11):879-882.
39. Saad F, Eastham J. Zoledronic acid improves clinical outcomes when administered before onset of bone pain in patients with prostate cancer. Urology. 2010;76(5):1175-1181.
40. Berenson JR, Lichtenstein A, Porter L, et al. Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. Myeloma Aredia Study Group. N Engl J Med. 1996;334(8):488-493.
41. Berenson JR, Lichtenstein A, Porter L, et al. Long-term pamidronate treatment of advanced multiple myeloma patients reduces skeletal events. Myeloma Aredia Study Group. J Clin Oncol. 1998;16(2):593-602.
42. Lahtinen R, Laakso M, Palva I, Virkkunen P, Elomaa I. Randomised, placebo-controlled multicentre trial of clodronate in multiple myeloma. Finnish Leukaemia Group. Lancet. 1992;340(8827):1049-1052.
1. Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225-249.
2. Cooleman RE. Metastatic bone disease: Clinical features, pathophysiology, and treatment strategies. Cancer Treat Rev. 2001;27(3):165-1763.
3. Hirabayashi H, Ebara S, Kinoshita T, et al. Clinical outcome and survival after palliative surgery for spinal metastases. Cancer. 2003;97(2):476-84.
4. van der Linden YM, Dijkstra SPDS, Vonk EJA, Marijnen CA, Leer JW; Dutch Bone Metastasis Study Group. Prediction of survival in patients with metastases in the spinal column. Cancer. 2005;103(2):320-328.
5. Coleman RE. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12(20, pt 2):6243S-6249S.
6. Body JJ. Metastatic bone disease: Clinical and therapeutic aspects. Bone. 1992;13(suppl 1):S57-S62.
7. Siegel RS, Ma J, Zou Z, Jermal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64(1):9-29.
8. National Cancer Institute. SEER stat fact sheets: Myeloma. National Cancer Institute Website. http://seer.cancer.gov/statfacts/html/mulmy.html. Accessed January 12, 2015.
9. Lentle BC, McGowan DG, Dierich H. Technetium-99M polyphosphate bone scanning in carcinoma of the prostate. Br J Urol. 1974;46(5):543-548.
10. Söderlund V. Radiological diagnosis of skeletal metastases. Eur Radiol. 1996;6(5):587-595.
11. Flickinger FW, Sanal SM. Bone marrow MRI: Techniques and accuracy for detecting breast cancer metastases. Magn Reson Imaging. 1994;12(6):829-35.
12. Hamaoka T, Madewell JE, Podoloff DA, Hortobagyi GN, Ueno NT. Bone imaging in metastatic breast cancer. J Clin Oncol. 2004;22(14):2942-2953.
13. Daldrup-Link HE, Franzius C, Link TM et al. Whole-body MR imaging for detection of bone metastases in children and young adults: Comparison with skeletal scintigraphy and FDG PET. AJR Am J Roentgenol. 2001;177(1):229-236.
14. Ohta M, Tokuda Y, Suzuki Y, et al. Whole body PET for the evaluation of bony metastases in patients with breast cancer: Comparison with 99Tcm-MDP bone scintigraphy. Nucl Med Commun. 2001;22(8):875-879.
15. Koolen BB, Vegt E, Rutgers EJ, et al. FDG-avid sclerotic bone metastases in breast cancer patients: A PET/CT case series. Ann Nucl Med. 2012;26(1):86-91.
16. Even-Sapir E, Metser U, Flusser G, et al. Assessment of malignant skeletal disease: Initial experience with 18F-fluoride PET/CT and comparison between 18F-fluoride PET and 18F-fluoride PET/CT. J Nucl Med. 2004;45(2):272-278.
17. Lonneux M, Borbath II, Berlière M, Kirkove C, Pauwels S. The place of whole-body PET FDG for the diagnosis of distant recurrence of breast cancer. Clin Positron Imaging. 2000;3(2):45-49.
18. Cook GJ, Houston S, Rubens R, Maisey MN, Fogelman I. Detection of bone metastases in breast cancer by 18FDG PET: Differing metabolic activity in osteoblastic and osteolytic lesions. J Clin Oncol. 1998;16(10):3375-3379.
19. Houssami N, Costelloe CM. Imaging bone metastases in breast cancer: Evidence on comparative test accuracy. Ann Oncol. 2012;23(4):834-843.
20. Yeh SD, Imbriaco M, Larson SM, et al. Detection of bony metastases of androgen-independent prostate cancer by PET-FDG. Nucl Med Biol. 1996;23(6):693-697.
21. Morris MJ, Akhurst T, Osman I, et al. Fluorinated deoxyglucose positron emission tomography imaging in progressive metastatic prostate cancer. Urology. 2002;59(6):913-918.
22. Rosen LS, Gordon D, Tchekmedyian S, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: A phase III, double-blind, randomized trial—the Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21(16):3150-3157.
23. Hillner BE, Ingle JN, Chlebowski RT, et al; American Society of Clinical Psychology. American Society of Clinical Oncology 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol. 2003;21(21):4042-4057.
24. Chow E, Harris K, Fan G, Tsao M, Size WM. Palliative radiotherapy trials for bone metastases: A systematic review. J Clin Oncol. 2007;25(11):1423-1436.
25. Wu JS, Wong R, Johnston M, Bezjak A, Whelan T; Cancer Care Ontario Practice Guidelines Initiative Supportive Care Group. Meta-analysis of dose-fractionation radiotherapy trials for the palliation of painful bone metastases. Int J Radiat Oncol Biol Phys. 2003;55(3):594-605.
26. Tong D, Gillick L, Hendrickson FR. The palliation of symptomatic osseous metastases. Final results of the study by the Radiation Therapy Oncology Group. Cancer. 1982;50(5):893-899.
27. Hartsell WF, Scott CB, Bruner DW, et al. Randomized trial of short- versus long-course radiotherapy for palliation of painful bone metastases. J Natl Cancer Inst. 2005;97(11):798-804.
28. Frassica DA. General principles of external beam radiation therapy for skeletal metastases. Clin Orthop Relat Res. 2003;415(suppl):S158-S164.
29. Silberstein EB. Systemic radiopharmaceutical therapy of painful osteoblastic metastases. Semin Radiat Oncol. 2000;10(3):240-249.
30. Neville-Webbe HL, Gnant M, Coleman RE. Potential anticancer properties of bisphosphonates. Semin Oncol. 2010;37(suppl 1):S53-S65.
31. Loftus LS, Edwards-Bennett S, Sokol GH. Systemic therapy for bone metastases. Cancer Control. 2012;19(2):145-153.
32. Rosen L, Harland SJ, Oosterlinck W. Broad clinical activity of zoledronic acid in osteolytic to osteoblastic bone lesions in patients with a broad range of solid tumors. Am J Clin Oncol. 2002;25(6)(suppl 1):S19-S24.
33. Fornier MN. Denosumab: Second chapter in controlling bone metastases or a new book? J Clin Oncol. 2010;28(35):5127-5131.
34. Mortimer JE, Pal SK. Safety considerations for use of bone-targeted agents in patients with cancer. Semin Oncol. 2010;37(suppl 1):S66-S72.
35. Pavlakis N, Schmidt R, Stockler M. Bisphosphonates for breast cancer. Cochrane Database Syst Rev. 2005;3:CD003474.
36. Kohno N, Aogi K, Minami H, et al. Zoledronic acid significantly reduces skeletal complications compared with placebo in Japanese women with bone metastases from breast cancer: A randomized, placebo-controlled trial. J Clin Oncol. 2005;23(15):3314-3321.
37. Rosen LS, Gordon D, Tchekmedyian NS, et al. Long-term efficacy and safety of zoledronic acid in the treatment of skeletal metastases in patients with nonsmall cell lung carcinoma and other solid tumors: A randomized, phase III, double-blind, placebo-controlled trial. Cancer. 2004;100(12):2613-2621.
38. Saad F, Gleason DM, Murray R, et al. Long-term efficacy of zoledronic acid for the prevention of skeletal complications in patients with metastatic hormone-refractory prostate cancer. J Natl Cancer Inst. 2004;96(11):879-882.
39. Saad F, Eastham J. Zoledronic acid improves clinical outcomes when administered before onset of bone pain in patients with prostate cancer. Urology. 2010;76(5):1175-1181.
40. Berenson JR, Lichtenstein A, Porter L, et al. Efficacy of pamidronate in reducing skeletal events in patients with advanced multiple myeloma. Myeloma Aredia Study Group. N Engl J Med. 1996;334(8):488-493.
41. Berenson JR, Lichtenstein A, Porter L, et al. Long-term pamidronate treatment of advanced multiple myeloma patients reduces skeletal events. Myeloma Aredia Study Group. J Clin Oncol. 1998;16(2):593-602.
42. Lahtinen R, Laakso M, Palva I, Virkkunen P, Elomaa I. Randomised, placebo-controlled multicentre trial of clodronate in multiple myeloma. Finnish Leukaemia Group. Lancet. 1992;340(8827):1049-1052.
Neural stem cells may fight chemo brain

Photo by Janet Stevens
Human neural stem cell treatments are showing promise for reversing learning and memory deficits after chemotherapy, according to an article in Cancer Research.
Investigators found that transplanting stem cells in rats a week after they completed a series of chemotherapy sessions restored a range of cognitive functions, as
measured a month later via behavioral testing.
In contrast, animals that did not receive stem cells showed significant learning and memory impairment.
“Our findings provide the first solid evidence that transplantation of human neural stem cells can be used to reverse chemotherapeutic-induced damage of healthy tissue in the brain,” said study author Charles Limoli, PhD, of the University of California, Irvine.
For this study, Dr Limoli and his colleagues transplanted adult neural stem cells into the brains of rats that had received cyclophosphamide.
The cells migrated throughout the hippocampus, where they survived and differentiated into multiple neural cell types. Additionally, these cells triggered the secretion of neurotrophic growth factors that helped rebuild wounded neurons.
The investigators also found that engrafted cells protected the host neurons, thereby preventing the loss or promoting the repair of damaged neurons and their finer structural elements, referred to as dendritic spines.
“This research suggests that stem cell therapies may one day be implemented in the clinic to provide relief to patients suffering from cognitive impairments incurred as a result of their cancer treatments,” Dr Limoli said. “While much work remains, a clinical trial analyzing the safety of such approaches may be possible within a few years.” ![]()

Photo by Janet Stevens
Human neural stem cell treatments are showing promise for reversing learning and memory deficits after chemotherapy, according to an article in Cancer Research.
Investigators found that transplanting stem cells in rats a week after they completed a series of chemotherapy sessions restored a range of cognitive functions, as
measured a month later via behavioral testing.
In contrast, animals that did not receive stem cells showed significant learning and memory impairment.
“Our findings provide the first solid evidence that transplantation of human neural stem cells can be used to reverse chemotherapeutic-induced damage of healthy tissue in the brain,” said study author Charles Limoli, PhD, of the University of California, Irvine.
For this study, Dr Limoli and his colleagues transplanted adult neural stem cells into the brains of rats that had received cyclophosphamide.
The cells migrated throughout the hippocampus, where they survived and differentiated into multiple neural cell types. Additionally, these cells triggered the secretion of neurotrophic growth factors that helped rebuild wounded neurons.
The investigators also found that engrafted cells protected the host neurons, thereby preventing the loss or promoting the repair of damaged neurons and their finer structural elements, referred to as dendritic spines.
“This research suggests that stem cell therapies may one day be implemented in the clinic to provide relief to patients suffering from cognitive impairments incurred as a result of their cancer treatments,” Dr Limoli said. “While much work remains, a clinical trial analyzing the safety of such approaches may be possible within a few years.” ![]()

Photo by Janet Stevens
Human neural stem cell treatments are showing promise for reversing learning and memory deficits after chemotherapy, according to an article in Cancer Research.
Investigators found that transplanting stem cells in rats a week after they completed a series of chemotherapy sessions restored a range of cognitive functions, as
measured a month later via behavioral testing.
In contrast, animals that did not receive stem cells showed significant learning and memory impairment.
“Our findings provide the first solid evidence that transplantation of human neural stem cells can be used to reverse chemotherapeutic-induced damage of healthy tissue in the brain,” said study author Charles Limoli, PhD, of the University of California, Irvine.
For this study, Dr Limoli and his colleagues transplanted adult neural stem cells into the brains of rats that had received cyclophosphamide.
The cells migrated throughout the hippocampus, where they survived and differentiated into multiple neural cell types. Additionally, these cells triggered the secretion of neurotrophic growth factors that helped rebuild wounded neurons.
The investigators also found that engrafted cells protected the host neurons, thereby preventing the loss or promoting the repair of damaged neurons and their finer structural elements, referred to as dendritic spines.
“This research suggests that stem cell therapies may one day be implemented in the clinic to provide relief to patients suffering from cognitive impairments incurred as a result of their cancer treatments,” Dr Limoli said. “While much work remains, a clinical trial analyzing the safety of such approaches may be possible within a few years.” ![]()
NICE rejects pomalidomide as MM treatment

In a final draft guidance, the UK’s National Institute for Health and Care Excellence (NICE) has said it cannot recommend pomalidomide (Imnovid) for the treatment of multiple myeloma (MM).
NICE recommends thalidomide for most MM patients as a first-line treatment and bortezomib for patients who are unable to receive thalidomide and those who fail first-line treatment.
For patients who have received 2 prior therapies, the agency recommends lenalidomide.
NICE considered pomalidomide for use in MM patients after their third or subsequent relapse.
The agency said it could not recommend pomalidomide, in combination with dexamethasone, within its marketing authorization, which is to treat adults with relapsed and refractory MM who have had at least 2 prior treatments, including lenalidomide and bortezomib, and whose disease has progressed on their last therapy.
“Unfortunately, we cannot recommend pomalidomide, as the analyses from Celgene, the company that markets pomalidomide, showed that the drug does not offer enough benefit to justify its high price,” said Sir Andrew Dillon, NICE chief executive.
NICE’s final draft guidance is now with consultees, who have the opportunity to appeal against it. Until NICE issues its final guidance, National Health Service (NHS) bodies should make decisions locally on the funding of specific treatments.
This draft guidance does not mean that patients currently taking pomalidomide will stop receiving it. They have the option to continue treatment until they and their clinicians consider it appropriate to stop.
And pomalidomide is recommended for use within NHS Scotland.
Insufficient evidence
The committee advising NICE was not able to judge with any confidence how much more effective pomalidomide was compared with current treatment options based on the evidence provided by Celgene before and after consultation.
However, bearing in mind the magnitude of the differences in the overall survival estimates between pomalidomide and high-dose dexamethasone in the phase 3 MM-003 trial, and all data presented to the committee for comparators, the committee was persuaded that pomalidomide extends life for at least 3 months, on average, when compared with standard NHS care.
Nevertheless, considering the incremental cost-effectiveness ratios, the committee concluded that, even with the end-of-life criteria met, the weighting that would have to be placed on the quality-adjusted life-years (QALYs) gained would be too high to consider pomalidomide a cost-effective use of NHS resources.
Also, the committee concluded that the uncertainty in the relative effectiveness of pomalidomide compared with established NHS practice would affect any weighting that could be placed on the QALYs gained.
All cost-per-QALY figures presented by Celgene were over £50,000 compared with bortezomib and over £70,000 compared with bendamustine plus thalidomide and dexamethasone. And the figures would further increase when a number of more realistic assumptions were included in the model, the committee said.
A pack of pomalidomide (21 tablets of 1 mg, 2 mg, 3 mg, or 4 mg) costs £8884. The recommended dosage of the drug is 4 mg once daily, taken on days 1 to 21 of repeated 28 day cycles. Treatment should continue until disease progression. ![]()

In a final draft guidance, the UK’s National Institute for Health and Care Excellence (NICE) has said it cannot recommend pomalidomide (Imnovid) for the treatment of multiple myeloma (MM).
NICE recommends thalidomide for most MM patients as a first-line treatment and bortezomib for patients who are unable to receive thalidomide and those who fail first-line treatment.
For patients who have received 2 prior therapies, the agency recommends lenalidomide.
NICE considered pomalidomide for use in MM patients after their third or subsequent relapse.
The agency said it could not recommend pomalidomide, in combination with dexamethasone, within its marketing authorization, which is to treat adults with relapsed and refractory MM who have had at least 2 prior treatments, including lenalidomide and bortezomib, and whose disease has progressed on their last therapy.
“Unfortunately, we cannot recommend pomalidomide, as the analyses from Celgene, the company that markets pomalidomide, showed that the drug does not offer enough benefit to justify its high price,” said Sir Andrew Dillon, NICE chief executive.
NICE’s final draft guidance is now with consultees, who have the opportunity to appeal against it. Until NICE issues its final guidance, National Health Service (NHS) bodies should make decisions locally on the funding of specific treatments.
This draft guidance does not mean that patients currently taking pomalidomide will stop receiving it. They have the option to continue treatment until they and their clinicians consider it appropriate to stop.
And pomalidomide is recommended for use within NHS Scotland.
Insufficient evidence
The committee advising NICE was not able to judge with any confidence how much more effective pomalidomide was compared with current treatment options based on the evidence provided by Celgene before and after consultation.
However, bearing in mind the magnitude of the differences in the overall survival estimates between pomalidomide and high-dose dexamethasone in the phase 3 MM-003 trial, and all data presented to the committee for comparators, the committee was persuaded that pomalidomide extends life for at least 3 months, on average, when compared with standard NHS care.
Nevertheless, considering the incremental cost-effectiveness ratios, the committee concluded that, even with the end-of-life criteria met, the weighting that would have to be placed on the quality-adjusted life-years (QALYs) gained would be too high to consider pomalidomide a cost-effective use of NHS resources.
Also, the committee concluded that the uncertainty in the relative effectiveness of pomalidomide compared with established NHS practice would affect any weighting that could be placed on the QALYs gained.
All cost-per-QALY figures presented by Celgene were over £50,000 compared with bortezomib and over £70,000 compared with bendamustine plus thalidomide and dexamethasone. And the figures would further increase when a number of more realistic assumptions were included in the model, the committee said.
A pack of pomalidomide (21 tablets of 1 mg, 2 mg, 3 mg, or 4 mg) costs £8884. The recommended dosage of the drug is 4 mg once daily, taken on days 1 to 21 of repeated 28 day cycles. Treatment should continue until disease progression. ![]()

In a final draft guidance, the UK’s National Institute for Health and Care Excellence (NICE) has said it cannot recommend pomalidomide (Imnovid) for the treatment of multiple myeloma (MM).
NICE recommends thalidomide for most MM patients as a first-line treatment and bortezomib for patients who are unable to receive thalidomide and those who fail first-line treatment.
For patients who have received 2 prior therapies, the agency recommends lenalidomide.
NICE considered pomalidomide for use in MM patients after their third or subsequent relapse.
The agency said it could not recommend pomalidomide, in combination with dexamethasone, within its marketing authorization, which is to treat adults with relapsed and refractory MM who have had at least 2 prior treatments, including lenalidomide and bortezomib, and whose disease has progressed on their last therapy.
“Unfortunately, we cannot recommend pomalidomide, as the analyses from Celgene, the company that markets pomalidomide, showed that the drug does not offer enough benefit to justify its high price,” said Sir Andrew Dillon, NICE chief executive.
NICE’s final draft guidance is now with consultees, who have the opportunity to appeal against it. Until NICE issues its final guidance, National Health Service (NHS) bodies should make decisions locally on the funding of specific treatments.
This draft guidance does not mean that patients currently taking pomalidomide will stop receiving it. They have the option to continue treatment until they and their clinicians consider it appropriate to stop.
And pomalidomide is recommended for use within NHS Scotland.
Insufficient evidence
The committee advising NICE was not able to judge with any confidence how much more effective pomalidomide was compared with current treatment options based on the evidence provided by Celgene before and after consultation.
However, bearing in mind the magnitude of the differences in the overall survival estimates between pomalidomide and high-dose dexamethasone in the phase 3 MM-003 trial, and all data presented to the committee for comparators, the committee was persuaded that pomalidomide extends life for at least 3 months, on average, when compared with standard NHS care.
Nevertheless, considering the incremental cost-effectiveness ratios, the committee concluded that, even with the end-of-life criteria met, the weighting that would have to be placed on the quality-adjusted life-years (QALYs) gained would be too high to consider pomalidomide a cost-effective use of NHS resources.
Also, the committee concluded that the uncertainty in the relative effectiveness of pomalidomide compared with established NHS practice would affect any weighting that could be placed on the QALYs gained.
All cost-per-QALY figures presented by Celgene were over £50,000 compared with bortezomib and over £70,000 compared with bendamustine plus thalidomide and dexamethasone. And the figures would further increase when a number of more realistic assumptions were included in the model, the committee said.
A pack of pomalidomide (21 tablets of 1 mg, 2 mg, 3 mg, or 4 mg) costs £8884. The recommended dosage of the drug is 4 mg once daily, taken on days 1 to 21 of repeated 28 day cycles. Treatment should continue until disease progression. ![]()
Cancer patients rarely make inappropriate requests, study shows

Photo courtesy of NIH
Although it makes sense that patient requests might drive physicians to practice defensive medicine, new research suggests that may not be the case with cancer patients.
The study, conducted at outpatient oncology centers, showed that patients rarely made clinically inappropriate requests.
Only 1% of more than 5000 patient-clinician encounters resulted in a clinically inappropriate request. And physicians rarely complied with these requests.
Keerthi Gogineni, MD, of the Hospital of the University of Pennsylvania in Philadelphia, and colleagues reported these findings in JAMA Oncology.
The researchers analyzed interviews with clinicians immediately after they visited with patients to assess whether a patient had made a request, the type of request made, and the clinical appropriateness of it.
The interviews were conducted at outpatient oncology facilities at 3 Philadelphia-area hospitals between October 2013 and June 2014.
The authors evaluated 5050 patient-clinician encounters involving 3624 patients and 60 clinicians. Most of the patients were women, and the most common cancer was hematologic.
Overall, 440 (8.7%) of the encounters included a patient demand or request, such as for imaging studies, treatments, or tests. And physicians complied with 365 (83%) of them.
Of all the patient-clinician encounters, 50 (1%) included a clinically inappropriate request. Clinicians complied with 7 of them. So, in 0.14% of encounters, clinicians ordered a test or treatment based on a clinically inappropriate request.
“At least in oncology, ‘demanding patients’ seem infrequent and may not account for a significant proportion of costs,” the researchers concluded.
In a related editorial, Anthony L. Back, MD, of the Seattle Cancer Care Alliance in Washington, wrote that inappropriate patient demands appear to be “more mythical than real.”
“[W]e have to stop blaming patients for being demanding,” he wrote. “In reality, it is hardly happening. The myth of the demanding patient is more about our own responses and how lackluster communication skills can contribute to difficult situations that stick in our throats and in our memories. And when we have calmed down enough to look up, we see that what is really happening between patients and physicians these days is something quite different.”
“It is possible that what the study by Gogineni et al documents is a point in the evolution of the patient-physician relationship when both sides recognize the complexity of cancer care belies a simple fix. Perhaps this ‘negative’ study is pointing to an important truth: that we need to redirect our attention from the myths that are distracting us.” ![]()

Photo courtesy of NIH
Although it makes sense that patient requests might drive physicians to practice defensive medicine, new research suggests that may not be the case with cancer patients.
The study, conducted at outpatient oncology centers, showed that patients rarely made clinically inappropriate requests.
Only 1% of more than 5000 patient-clinician encounters resulted in a clinically inappropriate request. And physicians rarely complied with these requests.
Keerthi Gogineni, MD, of the Hospital of the University of Pennsylvania in Philadelphia, and colleagues reported these findings in JAMA Oncology.
The researchers analyzed interviews with clinicians immediately after they visited with patients to assess whether a patient had made a request, the type of request made, and the clinical appropriateness of it.
The interviews were conducted at outpatient oncology facilities at 3 Philadelphia-area hospitals between October 2013 and June 2014.
The authors evaluated 5050 patient-clinician encounters involving 3624 patients and 60 clinicians. Most of the patients were women, and the most common cancer was hematologic.
Overall, 440 (8.7%) of the encounters included a patient demand or request, such as for imaging studies, treatments, or tests. And physicians complied with 365 (83%) of them.
Of all the patient-clinician encounters, 50 (1%) included a clinically inappropriate request. Clinicians complied with 7 of them. So, in 0.14% of encounters, clinicians ordered a test or treatment based on a clinically inappropriate request.
“At least in oncology, ‘demanding patients’ seem infrequent and may not account for a significant proportion of costs,” the researchers concluded.
In a related editorial, Anthony L. Back, MD, of the Seattle Cancer Care Alliance in Washington, wrote that inappropriate patient demands appear to be “more mythical than real.”
“[W]e have to stop blaming patients for being demanding,” he wrote. “In reality, it is hardly happening. The myth of the demanding patient is more about our own responses and how lackluster communication skills can contribute to difficult situations that stick in our throats and in our memories. And when we have calmed down enough to look up, we see that what is really happening between patients and physicians these days is something quite different.”
“It is possible that what the study by Gogineni et al documents is a point in the evolution of the patient-physician relationship when both sides recognize the complexity of cancer care belies a simple fix. Perhaps this ‘negative’ study is pointing to an important truth: that we need to redirect our attention from the myths that are distracting us.” ![]()

Photo courtesy of NIH
Although it makes sense that patient requests might drive physicians to practice defensive medicine, new research suggests that may not be the case with cancer patients.
The study, conducted at outpatient oncology centers, showed that patients rarely made clinically inappropriate requests.
Only 1% of more than 5000 patient-clinician encounters resulted in a clinically inappropriate request. And physicians rarely complied with these requests.
Keerthi Gogineni, MD, of the Hospital of the University of Pennsylvania in Philadelphia, and colleagues reported these findings in JAMA Oncology.
The researchers analyzed interviews with clinicians immediately after they visited with patients to assess whether a patient had made a request, the type of request made, and the clinical appropriateness of it.
The interviews were conducted at outpatient oncology facilities at 3 Philadelphia-area hospitals between October 2013 and June 2014.
The authors evaluated 5050 patient-clinician encounters involving 3624 patients and 60 clinicians. Most of the patients were women, and the most common cancer was hematologic.
Overall, 440 (8.7%) of the encounters included a patient demand or request, such as for imaging studies, treatments, or tests. And physicians complied with 365 (83%) of them.
Of all the patient-clinician encounters, 50 (1%) included a clinically inappropriate request. Clinicians complied with 7 of them. So, in 0.14% of encounters, clinicians ordered a test or treatment based on a clinically inappropriate request.
“At least in oncology, ‘demanding patients’ seem infrequent and may not account for a significant proportion of costs,” the researchers concluded.
In a related editorial, Anthony L. Back, MD, of the Seattle Cancer Care Alliance in Washington, wrote that inappropriate patient demands appear to be “more mythical than real.”
“[W]e have to stop blaming patients for being demanding,” he wrote. “In reality, it is hardly happening. The myth of the demanding patient is more about our own responses and how lackluster communication skills can contribute to difficult situations that stick in our throats and in our memories. And when we have calmed down enough to look up, we see that what is really happening between patients and physicians these days is something quite different.”
“It is possible that what the study by Gogineni et al documents is a point in the evolution of the patient-physician relationship when both sides recognize the complexity of cancer care belies a simple fix. Perhaps this ‘negative’ study is pointing to an important truth: that we need to redirect our attention from the myths that are distracting us.” ![]()
Gene variations tied to drug-related hearing loss

Photo by Peter Barta
New research has revealed inherited genetic variations associated with rapid hearing loss in young cancer patients who receive cisplatin.
The drug is used to treat a range of cancers and is known to pose a risk of severe hearing loss, but the risk factors involved are not completely understood.
Now, researchers have found that variations in the gene ACYP2 are associated with an increased risk of cisplatin-related hearing loss.
Jun J. Yang, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported this discovery in Nature Genetics.
The researchers checked the DNA of 238 young patients with brain tumors for more than 1.7 million common genetic variations. The team found that variations in ACYP2 were associated with as much as a 4-fold greater risk of cisplatin-related hearing loss.
The screening is among the first to survey the genetic landscape for clues to help explain why the risk of cisplatin-related hearing loss varies so widely among patients.
“This is an important first step in being able to pinpoint patients who are at higher risk of developing cisplatin toxicity and to learn how to better manage that risk,” said study author Clinton Stewart, PharmD, also of St Jude.
The researchers confirmed the association between the high-risk ACYP2 variants and cisplatin-related hearing loss in a separate group of 68 brain tumor patients. The association was independent of other risk factors for cisplatin-related hearing loss, including patient age and receipt of radiation therapy.
Twenty-four of the 306 patients in this study had at least one copy of the high-risk ACYP2 variant. All 24 patients had measurable hearing loss that occurred as early as weeks after beginning cisplatin therapy.
Overall, however, the ACYP2 variant explained a relatively small proportion of hearing damage. Just 12.4% of the 194 patients in this study with cisplatin-related hearing loss carried the ACYP2 variant.
“This suggests that other genes also contribute to the risk of hearing loss and are yet to be identified,” Dr Yang said. “Further research is needed to understand how the ACYP2 variations modify the risk . . . of cisplatin toxicity.”
Such studies could potentially lead to new medications to protect high-risk patients from cisplatin-related toxicity or help identify candidates for intensive monitoring of their hearing, Dr Stewart said. Early intervention could then be offered if problems are identified.
This study included patients enrolled in 1 of 3 trials designed by St Jude investigators for newly diagnosed pediatric brain tumors. The protocols involved similar treatment, including surgery to remove as much of the tumor as possible, followed by radiation, which was modified based on patient age and other risk factors.
The patients were scheduled to receive 4 rounds of cisplatin therapy. Patients’ hearing was tested before treatment began, after radiation therapy, after each round of chemotherapy, and then at regular standardized intervals. Analysis of the resulting data led to identification of ACYP2 and other variants.
“Our primary goal is to cure children with brain tumors, but we also have a duty to help patients survive with a high quality of life,” said Giles Robinson, MD, also of St Jude.
“Hearing loss can have a significant impact on a child’s quality of life, language development, and academic performance. There is no easy fix, but the more we know about the risk factors, the better we will understand how to use cisplatin.” ![]()

Photo by Peter Barta
New research has revealed inherited genetic variations associated with rapid hearing loss in young cancer patients who receive cisplatin.
The drug is used to treat a range of cancers and is known to pose a risk of severe hearing loss, but the risk factors involved are not completely understood.
Now, researchers have found that variations in the gene ACYP2 are associated with an increased risk of cisplatin-related hearing loss.
Jun J. Yang, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported this discovery in Nature Genetics.
The researchers checked the DNA of 238 young patients with brain tumors for more than 1.7 million common genetic variations. The team found that variations in ACYP2 were associated with as much as a 4-fold greater risk of cisplatin-related hearing loss.
The screening is among the first to survey the genetic landscape for clues to help explain why the risk of cisplatin-related hearing loss varies so widely among patients.
“This is an important first step in being able to pinpoint patients who are at higher risk of developing cisplatin toxicity and to learn how to better manage that risk,” said study author Clinton Stewart, PharmD, also of St Jude.
The researchers confirmed the association between the high-risk ACYP2 variants and cisplatin-related hearing loss in a separate group of 68 brain tumor patients. The association was independent of other risk factors for cisplatin-related hearing loss, including patient age and receipt of radiation therapy.
Twenty-four of the 306 patients in this study had at least one copy of the high-risk ACYP2 variant. All 24 patients had measurable hearing loss that occurred as early as weeks after beginning cisplatin therapy.
Overall, however, the ACYP2 variant explained a relatively small proportion of hearing damage. Just 12.4% of the 194 patients in this study with cisplatin-related hearing loss carried the ACYP2 variant.
“This suggests that other genes also contribute to the risk of hearing loss and are yet to be identified,” Dr Yang said. “Further research is needed to understand how the ACYP2 variations modify the risk . . . of cisplatin toxicity.”
Such studies could potentially lead to new medications to protect high-risk patients from cisplatin-related toxicity or help identify candidates for intensive monitoring of their hearing, Dr Stewart said. Early intervention could then be offered if problems are identified.
This study included patients enrolled in 1 of 3 trials designed by St Jude investigators for newly diagnosed pediatric brain tumors. The protocols involved similar treatment, including surgery to remove as much of the tumor as possible, followed by radiation, which was modified based on patient age and other risk factors.
The patients were scheduled to receive 4 rounds of cisplatin therapy. Patients’ hearing was tested before treatment began, after radiation therapy, after each round of chemotherapy, and then at regular standardized intervals. Analysis of the resulting data led to identification of ACYP2 and other variants.
“Our primary goal is to cure children with brain tumors, but we also have a duty to help patients survive with a high quality of life,” said Giles Robinson, MD, also of St Jude.
“Hearing loss can have a significant impact on a child’s quality of life, language development, and academic performance. There is no easy fix, but the more we know about the risk factors, the better we will understand how to use cisplatin.” ![]()

Photo by Peter Barta
New research has revealed inherited genetic variations associated with rapid hearing loss in young cancer patients who receive cisplatin.
The drug is used to treat a range of cancers and is known to pose a risk of severe hearing loss, but the risk factors involved are not completely understood.
Now, researchers have found that variations in the gene ACYP2 are associated with an increased risk of cisplatin-related hearing loss.
Jun J. Yang, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported this discovery in Nature Genetics.
The researchers checked the DNA of 238 young patients with brain tumors for more than 1.7 million common genetic variations. The team found that variations in ACYP2 were associated with as much as a 4-fold greater risk of cisplatin-related hearing loss.
The screening is among the first to survey the genetic landscape for clues to help explain why the risk of cisplatin-related hearing loss varies so widely among patients.
“This is an important first step in being able to pinpoint patients who are at higher risk of developing cisplatin toxicity and to learn how to better manage that risk,” said study author Clinton Stewart, PharmD, also of St Jude.
The researchers confirmed the association between the high-risk ACYP2 variants and cisplatin-related hearing loss in a separate group of 68 brain tumor patients. The association was independent of other risk factors for cisplatin-related hearing loss, including patient age and receipt of radiation therapy.
Twenty-four of the 306 patients in this study had at least one copy of the high-risk ACYP2 variant. All 24 patients had measurable hearing loss that occurred as early as weeks after beginning cisplatin therapy.
Overall, however, the ACYP2 variant explained a relatively small proportion of hearing damage. Just 12.4% of the 194 patients in this study with cisplatin-related hearing loss carried the ACYP2 variant.
“This suggests that other genes also contribute to the risk of hearing loss and are yet to be identified,” Dr Yang said. “Further research is needed to understand how the ACYP2 variations modify the risk . . . of cisplatin toxicity.”
Such studies could potentially lead to new medications to protect high-risk patients from cisplatin-related toxicity or help identify candidates for intensive monitoring of their hearing, Dr Stewart said. Early intervention could then be offered if problems are identified.
This study included patients enrolled in 1 of 3 trials designed by St Jude investigators for newly diagnosed pediatric brain tumors. The protocols involved similar treatment, including surgery to remove as much of the tumor as possible, followed by radiation, which was modified based on patient age and other risk factors.
The patients were scheduled to receive 4 rounds of cisplatin therapy. Patients’ hearing was tested before treatment began, after radiation therapy, after each round of chemotherapy, and then at regular standardized intervals. Analysis of the resulting data led to identification of ACYP2 and other variants.
“Our primary goal is to cure children with brain tumors, but we also have a duty to help patients survive with a high quality of life,” said Giles Robinson, MD, also of St Jude.
“Hearing loss can have a significant impact on a child’s quality of life, language development, and academic performance. There is no easy fix, but the more we know about the risk factors, the better we will understand how to use cisplatin.” ![]()
Signs may predict death in cancer patients

Researchers have identified 8 highly specific physical and cognitive signs that seem to be associated with imminent death in cancer patients.
The findings, published in Cancer, could offer clinicians the ability to better communicate with patients and their families.
The research might also help guide the medical team and caregivers when it comes to complex decision making, such as discontinuing tests and therapy, plans for hospital discharge, and hospice referral.
Previous studies in end-of-life care have focused on physicians prognosticating better. However, research on how to tell if a patient has entered the final days of life has been minimal, according to David Hui, MD, of the University of Texas MD Anderson Cancer Center in Houston.
“In the past, studies trying to understand the signs associated with impending death were conducted in people who were recognized as dying, so there’s a potential bias built into this model,” Dr Hui said.
“With our study, we observed a list of signs in patients from the time they were admitted to the palliative care unit. They were observed systematically, twice a day, without knowing if the patient would die or be discharged.”
Dr Hui and his colleagues observed 357 cancer patients, 57% of whom ultimately died. The researchers observed 52 physical and cognitive signs—identified by Dr Hui and his colleagues in previous research—twice a day from the patient’s admission to discharge or death.
Of those 52 signs, the 8 most highly associated with impending death within 3 days were:
- Nonreactive pupils
- Decreased response to verbal stimuli
- Decreased response to visual stimuli
- Inability to close eyelids
- Drooping of the nasolabial fold
- Neck hyperextension
- Grunting of vocal cords
- Upper gastrointestinal bleeding.
“When cancer patients reach the last days of life, this is an extremely emotional time for families; their stress levels cannot be understated,” Dr Hui said.
“Knowing when death is imminent would provide more information so caregivers can plan appropriately. For clinicians, having this information could help reassure families that we are providing the best care possible.”
Dr Hui stressed that this research is not yet practice-changing, but is an important step in understanding these 8 signs and their relation to impending death. In addition, the findings are only representative of imminent cancer death and should not be generalized to other causes of death.
Follow-up studies in different settings are planned. Dr Hui and his colleagues plan to look at the reliability of the identified signs, as well as evaluate this research in other countries and in the hospice setting. ![]()

Researchers have identified 8 highly specific physical and cognitive signs that seem to be associated with imminent death in cancer patients.
The findings, published in Cancer, could offer clinicians the ability to better communicate with patients and their families.
The research might also help guide the medical team and caregivers when it comes to complex decision making, such as discontinuing tests and therapy, plans for hospital discharge, and hospice referral.
Previous studies in end-of-life care have focused on physicians prognosticating better. However, research on how to tell if a patient has entered the final days of life has been minimal, according to David Hui, MD, of the University of Texas MD Anderson Cancer Center in Houston.
“In the past, studies trying to understand the signs associated with impending death were conducted in people who were recognized as dying, so there’s a potential bias built into this model,” Dr Hui said.
“With our study, we observed a list of signs in patients from the time they were admitted to the palliative care unit. They were observed systematically, twice a day, without knowing if the patient would die or be discharged.”
Dr Hui and his colleagues observed 357 cancer patients, 57% of whom ultimately died. The researchers observed 52 physical and cognitive signs—identified by Dr Hui and his colleagues in previous research—twice a day from the patient’s admission to discharge or death.
Of those 52 signs, the 8 most highly associated with impending death within 3 days were:
- Nonreactive pupils
- Decreased response to verbal stimuli
- Decreased response to visual stimuli
- Inability to close eyelids
- Drooping of the nasolabial fold
- Neck hyperextension
- Grunting of vocal cords
- Upper gastrointestinal bleeding.
“When cancer patients reach the last days of life, this is an extremely emotional time for families; their stress levels cannot be understated,” Dr Hui said.
“Knowing when death is imminent would provide more information so caregivers can plan appropriately. For clinicians, having this information could help reassure families that we are providing the best care possible.”
Dr Hui stressed that this research is not yet practice-changing, but is an important step in understanding these 8 signs and their relation to impending death. In addition, the findings are only representative of imminent cancer death and should not be generalized to other causes of death.
Follow-up studies in different settings are planned. Dr Hui and his colleagues plan to look at the reliability of the identified signs, as well as evaluate this research in other countries and in the hospice setting. ![]()

Researchers have identified 8 highly specific physical and cognitive signs that seem to be associated with imminent death in cancer patients.
The findings, published in Cancer, could offer clinicians the ability to better communicate with patients and their families.
The research might also help guide the medical team and caregivers when it comes to complex decision making, such as discontinuing tests and therapy, plans for hospital discharge, and hospice referral.
Previous studies in end-of-life care have focused on physicians prognosticating better. However, research on how to tell if a patient has entered the final days of life has been minimal, according to David Hui, MD, of the University of Texas MD Anderson Cancer Center in Houston.
“In the past, studies trying to understand the signs associated with impending death were conducted in people who were recognized as dying, so there’s a potential bias built into this model,” Dr Hui said.
“With our study, we observed a list of signs in patients from the time they were admitted to the palliative care unit. They were observed systematically, twice a day, without knowing if the patient would die or be discharged.”
Dr Hui and his colleagues observed 357 cancer patients, 57% of whom ultimately died. The researchers observed 52 physical and cognitive signs—identified by Dr Hui and his colleagues in previous research—twice a day from the patient’s admission to discharge or death.
Of those 52 signs, the 8 most highly associated with impending death within 3 days were:
- Nonreactive pupils
- Decreased response to verbal stimuli
- Decreased response to visual stimuli
- Inability to close eyelids
- Drooping of the nasolabial fold
- Neck hyperextension
- Grunting of vocal cords
- Upper gastrointestinal bleeding.
“When cancer patients reach the last days of life, this is an extremely emotional time for families; their stress levels cannot be understated,” Dr Hui said.
“Knowing when death is imminent would provide more information so caregivers can plan appropriately. For clinicians, having this information could help reassure families that we are providing the best care possible.”
Dr Hui stressed that this research is not yet practice-changing, but is an important step in understanding these 8 signs and their relation to impending death. In addition, the findings are only representative of imminent cancer death and should not be generalized to other causes of death.
Follow-up studies in different settings are planned. Dr Hui and his colleagues plan to look at the reliability of the identified signs, as well as evaluate this research in other countries and in the hospice setting. ![]()
Though costly, blood cancer drugs appear cost-effective

Photo by Bill Branson
A new analysis indicates that certain high-cost therapies for hematologic malignancies provide reasonable value for money spent.
Most cost-effectiveness ratios were lower than thresholds commonly used to establish cost-effectiveness in the US—$50,000 or $100,000 per quality-adjusted life year (QALY) gained.
The median cost-effectiveness ratio was highest for chronic myeloid leukemia (CML), at $55,000/QALY, and lowest for non-Hodgkin lymphoma (NHL), at $21,500/QALY.
Researchers presented these data in Blood.
“Given the increased discussion about the high cost of these treatments, we were somewhat surprised to discover that their cost-effectiveness ratios were lower than expected,” said study author Peter J. Neumann, ScD, of Tufts Medical Center in Boston.
“Our analysis had a small sample size and included both industry- and non-industry-funded studies. In addition, cost-effectiveness ratios may have changed over time as associated costs or benefits have changed. However, the study underscores that debates in healthcare should consider the value of breakthrough drugs and not just costs.”
With that issue in mind, Dr Neumann and his colleagues had conducted a systematic review of studies published between 1996 and 2012 that examined the cost utility of agents for hematologic malignancies. The cost utility of a drug was depicted as a ratio of a drug’s total cost per patient QALY gained.
The researchers identified 29 studies, 22 of which were industry-funded. Nine studies were conducted from a US perspective, 6 from the UK, 3 from Norway, 3 from Sweden, 2 from France, 1 from Canada, 1 from Finland, and 4 from “other” countries.
The team grouped studies according to malignancy—CML, chronic lymphocytic leukemia (CLL), NHL, and multiple myeloma (MM)—as well as by treatment—α interferon, alemtuzumab, bendamustine, bortezomib, dasatinib, imatinib, lenalidomide, rituximab alone or in combination, and thalidomide.
The studies reported 44 cost-effectiveness ratios, most concerning interventions for NHL (41%) or CML (30%). Most ratios pertained to rituximab (43%), α interferon (18%), or imatinib (16%), and the most common intervention-disease combination was rituximab (alone or in combination) for NHL (36%).
The median cost-effectiveness ratios fluctuated over time, rising from $35,000/QALY (1996-2002) to $52,000/QALY (2003-2006), then falling to $22,000/QALY (2007-2012).
The median cost-effectiveness ratio reported by industry-funded studies was lower ($26,000/QALY) than for non-industry-funded studies ($33,000/QALY).
Four cost-effectiveness ratios, 1 from an industry-funded study, exceeded $100,000/QALY. This included 2 studies of bortezomib in MM, 1 of α interferon in CML, and 1 of imatinib in CML.
The researchers said these results suggest that many new treatments for hematologic malignancies may confer reasonable value for money spent. The distribution of cost-effectiveness ratios is comparable to those for cancers overall and for other healthcare fields, they said.
This study was funded by internal resources at the Center for the Evaluation of Value and Risk in Health. The center receives funding from federal, private foundation, and pharmaceutical industry sources. ![]()

Photo by Bill Branson
A new analysis indicates that certain high-cost therapies for hematologic malignancies provide reasonable value for money spent.
Most cost-effectiveness ratios were lower than thresholds commonly used to establish cost-effectiveness in the US—$50,000 or $100,000 per quality-adjusted life year (QALY) gained.
The median cost-effectiveness ratio was highest for chronic myeloid leukemia (CML), at $55,000/QALY, and lowest for non-Hodgkin lymphoma (NHL), at $21,500/QALY.
Researchers presented these data in Blood.
“Given the increased discussion about the high cost of these treatments, we were somewhat surprised to discover that their cost-effectiveness ratios were lower than expected,” said study author Peter J. Neumann, ScD, of Tufts Medical Center in Boston.
“Our analysis had a small sample size and included both industry- and non-industry-funded studies. In addition, cost-effectiveness ratios may have changed over time as associated costs or benefits have changed. However, the study underscores that debates in healthcare should consider the value of breakthrough drugs and not just costs.”
With that issue in mind, Dr Neumann and his colleagues had conducted a systematic review of studies published between 1996 and 2012 that examined the cost utility of agents for hematologic malignancies. The cost utility of a drug was depicted as a ratio of a drug’s total cost per patient QALY gained.
The researchers identified 29 studies, 22 of which were industry-funded. Nine studies were conducted from a US perspective, 6 from the UK, 3 from Norway, 3 from Sweden, 2 from France, 1 from Canada, 1 from Finland, and 4 from “other” countries.
The team grouped studies according to malignancy—CML, chronic lymphocytic leukemia (CLL), NHL, and multiple myeloma (MM)—as well as by treatment—α interferon, alemtuzumab, bendamustine, bortezomib, dasatinib, imatinib, lenalidomide, rituximab alone or in combination, and thalidomide.
The studies reported 44 cost-effectiveness ratios, most concerning interventions for NHL (41%) or CML (30%). Most ratios pertained to rituximab (43%), α interferon (18%), or imatinib (16%), and the most common intervention-disease combination was rituximab (alone or in combination) for NHL (36%).
The median cost-effectiveness ratios fluctuated over time, rising from $35,000/QALY (1996-2002) to $52,000/QALY (2003-2006), then falling to $22,000/QALY (2007-2012).
The median cost-effectiveness ratio reported by industry-funded studies was lower ($26,000/QALY) than for non-industry-funded studies ($33,000/QALY).
Four cost-effectiveness ratios, 1 from an industry-funded study, exceeded $100,000/QALY. This included 2 studies of bortezomib in MM, 1 of α interferon in CML, and 1 of imatinib in CML.
The researchers said these results suggest that many new treatments for hematologic malignancies may confer reasonable value for money spent. The distribution of cost-effectiveness ratios is comparable to those for cancers overall and for other healthcare fields, they said.
This study was funded by internal resources at the Center for the Evaluation of Value and Risk in Health. The center receives funding from federal, private foundation, and pharmaceutical industry sources. ![]()

Photo by Bill Branson
A new analysis indicates that certain high-cost therapies for hematologic malignancies provide reasonable value for money spent.
Most cost-effectiveness ratios were lower than thresholds commonly used to establish cost-effectiveness in the US—$50,000 or $100,000 per quality-adjusted life year (QALY) gained.
The median cost-effectiveness ratio was highest for chronic myeloid leukemia (CML), at $55,000/QALY, and lowest for non-Hodgkin lymphoma (NHL), at $21,500/QALY.
Researchers presented these data in Blood.
“Given the increased discussion about the high cost of these treatments, we were somewhat surprised to discover that their cost-effectiveness ratios were lower than expected,” said study author Peter J. Neumann, ScD, of Tufts Medical Center in Boston.
“Our analysis had a small sample size and included both industry- and non-industry-funded studies. In addition, cost-effectiveness ratios may have changed over time as associated costs or benefits have changed. However, the study underscores that debates in healthcare should consider the value of breakthrough drugs and not just costs.”
With that issue in mind, Dr Neumann and his colleagues had conducted a systematic review of studies published between 1996 and 2012 that examined the cost utility of agents for hematologic malignancies. The cost utility of a drug was depicted as a ratio of a drug’s total cost per patient QALY gained.
The researchers identified 29 studies, 22 of which were industry-funded. Nine studies were conducted from a US perspective, 6 from the UK, 3 from Norway, 3 from Sweden, 2 from France, 1 from Canada, 1 from Finland, and 4 from “other” countries.
The team grouped studies according to malignancy—CML, chronic lymphocytic leukemia (CLL), NHL, and multiple myeloma (MM)—as well as by treatment—α interferon, alemtuzumab, bendamustine, bortezomib, dasatinib, imatinib, lenalidomide, rituximab alone or in combination, and thalidomide.
The studies reported 44 cost-effectiveness ratios, most concerning interventions for NHL (41%) or CML (30%). Most ratios pertained to rituximab (43%), α interferon (18%), or imatinib (16%), and the most common intervention-disease combination was rituximab (alone or in combination) for NHL (36%).
The median cost-effectiveness ratios fluctuated over time, rising from $35,000/QALY (1996-2002) to $52,000/QALY (2003-2006), then falling to $22,000/QALY (2007-2012).
The median cost-effectiveness ratio reported by industry-funded studies was lower ($26,000/QALY) than for non-industry-funded studies ($33,000/QALY).
Four cost-effectiveness ratios, 1 from an industry-funded study, exceeded $100,000/QALY. This included 2 studies of bortezomib in MM, 1 of α interferon in CML, and 1 of imatinib in CML.
The researchers said these results suggest that many new treatments for hematologic malignancies may confer reasonable value for money spent. The distribution of cost-effectiveness ratios is comparable to those for cancers overall and for other healthcare fields, they said.
This study was funded by internal resources at the Center for the Evaluation of Value and Risk in Health. The center receives funding from federal, private foundation, and pharmaceutical industry sources. ![]()
mAb shows ‘modest activity’ in rel/ref MM

Photo by Linda Bartlett
The monoclonal IgM antibody PAT-SM6 was “well-tolerated” and showed “modest clinical activity” in patients with relapsed or refractory multiple myeloma (MM), researchers reported in haematologica.
Adverse events occurred in all 12 patients enrolled in the phase 1/2a study, but most were considered unrelated to treatment.
A third of patients, all of whom had progressive disease upon study entry, achieved stable disease after receiving PAT-SM6. The remaining patients progressed.
Leo Rasche, MD, of University Hospital Wurzburg in Germany, and his colleagues conducted this study. It was funded, in part, by Patrys Limited, the company developing PAT-SM6.
The study included 12 heavily pretreated MM patients. They had a median age of 69.5 years and a long-standing history of MM (range, 3.25 to 15.75 years). They had received a median of 3.9 prior lines of therapy (range, 2-7).
Patients received 4 escalating doses of PAT-SM6, over a period of 2 weeks, via intravenous infusions at 0.3 mg/kg, 1 mg/kg, 3 mg/kg, and 6 mg/kg.
Safety data
There were 54 treatment-emergent adverse events in all 12 patients. However, there were no dose-limiting toxicities and no deaths. The maximum tolerated dose has not been reached.
More than 80% of the adverse events were of mild to moderate intensity. Two patients (16.6%) each experienced a single serious event. One patient had acute back pain, and one had a bile duct stone. Neither of these events was considered treatment-related.
Twenty-one adverse events were considered treatment-related. This included leukopenia (66.6%), neutropenia (50%), hypertension (16.6%), catheter-related thrombophlebitis (8.3%), injection site erythema (8.3%), slight headache (8.3%), C-reactive protein increase (8.3%), and hypertriglyceridemia (8.3%).
Efficacy data
Most patients progressed following treatment, but 4 (33.3%) had stable disease. The investigators noted that stable disease is not necessarily connected with a clinical benefit, so they analyzed the 4 patients in detail.
Patient 4, who received PAT-SM6 at 1 mg/kg, entered the study with high-risk disease. The patient had 13q deletion and 1q21 gain, had received 5 prior lines of therapy, and was refractory to novel agents, including pomalidomide and bortezomib.
The patient was treatment-free for 1 month prior to receiving PAT-SM6. During treatment, there were no symptoms of active myeloma, and the patient asked to continue salvage therapy 1 week after the end of the study.
Patient 7, who received PAT-SM6 at 3 mg/kg, had been diagnosed with MM for 15 years and had received 4 prior lines of therapy.
The patient’s treatment-free interval before receiving PAT-SM6 was 30 months. After PAT-SM6, the patient was therapy-free for 4.6 months.
Patient 10, who received PAT-SM6 at 6mg/kg, had 6 prior lines of therapy, including tandem autologous stem cell transplant and several polychemotherapeutic regimens.
The patient was therapy-free for 4 months prior to receiving PAT-SM6. The patient requested salvage therapy 1 month after receiving PAT-SM6.
Patient 11, who received PAT-SM6 at 6mg/kg, had 4 prior lines of therapy and was refractory to both lenalidomide and thalidomide.
The patient’s treatment-free interval prior to PAT-SM6 was 12 months. After PAT-SM6, the patient was therapy-free for 5.2 months.
The researchers said additional trials testing PAT-SM6 in combination with other MM therapies are planned. PAT-SM6 has received orphan drug designation for MM in the US and the European Union. ![]()

Photo by Linda Bartlett
The monoclonal IgM antibody PAT-SM6 was “well-tolerated” and showed “modest clinical activity” in patients with relapsed or refractory multiple myeloma (MM), researchers reported in haematologica.
Adverse events occurred in all 12 patients enrolled in the phase 1/2a study, but most were considered unrelated to treatment.
A third of patients, all of whom had progressive disease upon study entry, achieved stable disease after receiving PAT-SM6. The remaining patients progressed.
Leo Rasche, MD, of University Hospital Wurzburg in Germany, and his colleagues conducted this study. It was funded, in part, by Patrys Limited, the company developing PAT-SM6.
The study included 12 heavily pretreated MM patients. They had a median age of 69.5 years and a long-standing history of MM (range, 3.25 to 15.75 years). They had received a median of 3.9 prior lines of therapy (range, 2-7).
Patients received 4 escalating doses of PAT-SM6, over a period of 2 weeks, via intravenous infusions at 0.3 mg/kg, 1 mg/kg, 3 mg/kg, and 6 mg/kg.
Safety data
There were 54 treatment-emergent adverse events in all 12 patients. However, there were no dose-limiting toxicities and no deaths. The maximum tolerated dose has not been reached.
More than 80% of the adverse events were of mild to moderate intensity. Two patients (16.6%) each experienced a single serious event. One patient had acute back pain, and one had a bile duct stone. Neither of these events was considered treatment-related.
Twenty-one adverse events were considered treatment-related. This included leukopenia (66.6%), neutropenia (50%), hypertension (16.6%), catheter-related thrombophlebitis (8.3%), injection site erythema (8.3%), slight headache (8.3%), C-reactive protein increase (8.3%), and hypertriglyceridemia (8.3%).
Efficacy data
Most patients progressed following treatment, but 4 (33.3%) had stable disease. The investigators noted that stable disease is not necessarily connected with a clinical benefit, so they analyzed the 4 patients in detail.
Patient 4, who received PAT-SM6 at 1 mg/kg, entered the study with high-risk disease. The patient had 13q deletion and 1q21 gain, had received 5 prior lines of therapy, and was refractory to novel agents, including pomalidomide and bortezomib.
The patient was treatment-free for 1 month prior to receiving PAT-SM6. During treatment, there were no symptoms of active myeloma, and the patient asked to continue salvage therapy 1 week after the end of the study.
Patient 7, who received PAT-SM6 at 3 mg/kg, had been diagnosed with MM for 15 years and had received 4 prior lines of therapy.
The patient’s treatment-free interval before receiving PAT-SM6 was 30 months. After PAT-SM6, the patient was therapy-free for 4.6 months.
Patient 10, who received PAT-SM6 at 6mg/kg, had 6 prior lines of therapy, including tandem autologous stem cell transplant and several polychemotherapeutic regimens.
The patient was therapy-free for 4 months prior to receiving PAT-SM6. The patient requested salvage therapy 1 month after receiving PAT-SM6.
Patient 11, who received PAT-SM6 at 6mg/kg, had 4 prior lines of therapy and was refractory to both lenalidomide and thalidomide.
The patient’s treatment-free interval prior to PAT-SM6 was 12 months. After PAT-SM6, the patient was therapy-free for 5.2 months.
The researchers said additional trials testing PAT-SM6 in combination with other MM therapies are planned. PAT-SM6 has received orphan drug designation for MM in the US and the European Union. ![]()

Photo by Linda Bartlett
The monoclonal IgM antibody PAT-SM6 was “well-tolerated” and showed “modest clinical activity” in patients with relapsed or refractory multiple myeloma (MM), researchers reported in haematologica.
Adverse events occurred in all 12 patients enrolled in the phase 1/2a study, but most were considered unrelated to treatment.
A third of patients, all of whom had progressive disease upon study entry, achieved stable disease after receiving PAT-SM6. The remaining patients progressed.
Leo Rasche, MD, of University Hospital Wurzburg in Germany, and his colleagues conducted this study. It was funded, in part, by Patrys Limited, the company developing PAT-SM6.
The study included 12 heavily pretreated MM patients. They had a median age of 69.5 years and a long-standing history of MM (range, 3.25 to 15.75 years). They had received a median of 3.9 prior lines of therapy (range, 2-7).
Patients received 4 escalating doses of PAT-SM6, over a period of 2 weeks, via intravenous infusions at 0.3 mg/kg, 1 mg/kg, 3 mg/kg, and 6 mg/kg.
Safety data
There were 54 treatment-emergent adverse events in all 12 patients. However, there were no dose-limiting toxicities and no deaths. The maximum tolerated dose has not been reached.
More than 80% of the adverse events were of mild to moderate intensity. Two patients (16.6%) each experienced a single serious event. One patient had acute back pain, and one had a bile duct stone. Neither of these events was considered treatment-related.
Twenty-one adverse events were considered treatment-related. This included leukopenia (66.6%), neutropenia (50%), hypertension (16.6%), catheter-related thrombophlebitis (8.3%), injection site erythema (8.3%), slight headache (8.3%), C-reactive protein increase (8.3%), and hypertriglyceridemia (8.3%).
Efficacy data
Most patients progressed following treatment, but 4 (33.3%) had stable disease. The investigators noted that stable disease is not necessarily connected with a clinical benefit, so they analyzed the 4 patients in detail.
Patient 4, who received PAT-SM6 at 1 mg/kg, entered the study with high-risk disease. The patient had 13q deletion and 1q21 gain, had received 5 prior lines of therapy, and was refractory to novel agents, including pomalidomide and bortezomib.
The patient was treatment-free for 1 month prior to receiving PAT-SM6. During treatment, there were no symptoms of active myeloma, and the patient asked to continue salvage therapy 1 week after the end of the study.
Patient 7, who received PAT-SM6 at 3 mg/kg, had been diagnosed with MM for 15 years and had received 4 prior lines of therapy.
The patient’s treatment-free interval before receiving PAT-SM6 was 30 months. After PAT-SM6, the patient was therapy-free for 4.6 months.
Patient 10, who received PAT-SM6 at 6mg/kg, had 6 prior lines of therapy, including tandem autologous stem cell transplant and several polychemotherapeutic regimens.
The patient was therapy-free for 4 months prior to receiving PAT-SM6. The patient requested salvage therapy 1 month after receiving PAT-SM6.
Patient 11, who received PAT-SM6 at 6mg/kg, had 4 prior lines of therapy and was refractory to both lenalidomide and thalidomide.
The patient’s treatment-free interval prior to PAT-SM6 was 12 months. After PAT-SM6, the patient was therapy-free for 5.2 months.
The researchers said additional trials testing PAT-SM6 in combination with other MM therapies are planned. PAT-SM6 has received orphan drug designation for MM in the US and the European Union.
mAb prompts responses in pretreated MM

Credit: Linda Bartlett
Single-agent daratumumab can elicit responses in patients with multiple myeloma (MM) who have failed treatment with proteasome inhibitors and immunomodulatory agents (IMiDs), preliminary results of a phase 2 study suggest.
The study enrolled MM patients who have received at least 3 different lines of therapy, including both a proteasome inhibitor and an IMiD, and patients who are double-refractory to a proteasome inhibitor and an IMiD.
Daratumumab, an anti-CD38 monoclonal antibody, has breakthrough therapy designation from the US Food and Drug Administration to treat this patient population.
Genmab A/S, the company that discovered daratumumab and licensed it to Janssen Biotech, Inc. for development, announced results from the phase 2 trial (Sirius MMY2002) yesterday.
This 2-part study enrolled 124 MM patients. The goal of part 1 was to define an optimal daratumumab regimen going forward, and part 2 was an expansion based on the optimal regimen.
Patients were randomized to receive daratumumab at 8 mg/kg every 4 weeks continuously by intravenous infusion or at 16 mg/kg administered at weekly intervals for 8 weeks, then every 2 weeks for an additional 16 weeks, and every 4 weeks thereafter by intravenous infusion.
Genmab reported that the overall response rate was 29.2% in the 16 mg/kg dosing group. And the median duration of response was 7.4 months, as determined by an independent review committee.
Daratumumab also showed a manageable safety profile, according to Genmab. The company said the data will be discussed with health authorities at upcoming meetings, pending their agreement.

Credit: Linda Bartlett
Single-agent daratumumab can elicit responses in patients with multiple myeloma (MM) who have failed treatment with proteasome inhibitors and immunomodulatory agents (IMiDs), preliminary results of a phase 2 study suggest.
The study enrolled MM patients who have received at least 3 different lines of therapy, including both a proteasome inhibitor and an IMiD, and patients who are double-refractory to a proteasome inhibitor and an IMiD.
Daratumumab, an anti-CD38 monoclonal antibody, has breakthrough therapy designation from the US Food and Drug Administration to treat this patient population.
Genmab A/S, the company that discovered daratumumab and licensed it to Janssen Biotech, Inc. for development, announced results from the phase 2 trial (Sirius MMY2002) yesterday.
This 2-part study enrolled 124 MM patients. The goal of part 1 was to define an optimal daratumumab regimen going forward, and part 2 was an expansion based on the optimal regimen.
Patients were randomized to receive daratumumab at 8 mg/kg every 4 weeks continuously by intravenous infusion or at 16 mg/kg administered at weekly intervals for 8 weeks, then every 2 weeks for an additional 16 weeks, and every 4 weeks thereafter by intravenous infusion.
Genmab reported that the overall response rate was 29.2% in the 16 mg/kg dosing group. And the median duration of response was 7.4 months, as determined by an independent review committee.
Daratumumab also showed a manageable safety profile, according to Genmab. The company said the data will be discussed with health authorities at upcoming meetings, pending their agreement.

Credit: Linda Bartlett
Single-agent daratumumab can elicit responses in patients with multiple myeloma (MM) who have failed treatment with proteasome inhibitors and immunomodulatory agents (IMiDs), preliminary results of a phase 2 study suggest.
The study enrolled MM patients who have received at least 3 different lines of therapy, including both a proteasome inhibitor and an IMiD, and patients who are double-refractory to a proteasome inhibitor and an IMiD.
Daratumumab, an anti-CD38 monoclonal antibody, has breakthrough therapy designation from the US Food and Drug Administration to treat this patient population.
Genmab A/S, the company that discovered daratumumab and licensed it to Janssen Biotech, Inc. for development, announced results from the phase 2 trial (Sirius MMY2002) yesterday.
This 2-part study enrolled 124 MM patients. The goal of part 1 was to define an optimal daratumumab regimen going forward, and part 2 was an expansion based on the optimal regimen.
Patients were randomized to receive daratumumab at 8 mg/kg every 4 weeks continuously by intravenous infusion or at 16 mg/kg administered at weekly intervals for 8 weeks, then every 2 weeks for an additional 16 weeks, and every 4 weeks thereafter by intravenous infusion.
Genmab reported that the overall response rate was 29.2% in the 16 mg/kg dosing group. And the median duration of response was 7.4 months, as determined by an independent review committee.
Daratumumab also showed a manageable safety profile, according to Genmab. The company said the data will be discussed with health authorities at upcoming meetings, pending their agreement.
Discovery could help make Ras druggable

Credit: Jes Andersen/
University of Copenhagen
Researchers say they have discovered how Ras proteins find their proper place in cells, a finding that may aid the development of novel approaches to treat cancers.
The team noted that cancers develop if Ras proteins start to trigger misregulation, and Ras misregulates if it misses its correct location on the cell wall—the membrane.
What the researchers discovered is that Ras cannot reach its designated location if the membrane has the wrong shape.
“If the curvature of the cell is right, Ras goes to the right place,” said Dimitrios Stamou, PhD, of the University of Copenhagen in Denmark.
“If the membrane is too straight or too bent, it does not. And Ras is very much like any other worker. If it never finds the way to its workplace, it is not likely to get any work done.”
Dr Stamou and his colleagues described this discovery in Nature Chemical Biology.
Ras proteins are thought to be misregulated in upwards of 30% of all cancers. For 3 decades, researchers have been searching for ways to quell the killer protein.
Their lack of success has given Ras a reputation as the “undruggable cancer target,” but Dr Stamou believes we can change by moving in a new direction.
“If Ras goes off the rails because of changes in the curvature of the cell, perhaps we should target whatever changes the shape of the cell membrane,” he said.
Looking for a correlation between cell shape and Ras misregulation was unusual, even bordering on controversial, said study author Jannik Bruun Larsen, PhD, of the University of Copenhagen.
The researchers were investigating how Ras proteins attach themselves to the cell wall, and Dr Larsen tried to attach Ras to a variety of simulated cell membranes formed into small spheres or vesicles of varying sizes.
He found that Ras would attach more readily to smaller spheres, which were more curved than the large ones, and Dr Larsen started to see a pattern.
“For more than a decade, people thought that the constituents of the cell wall was the thing that controlled where Ras was localized,” Dr Larson said. “We have shown that at least one other aspect—namely, membrane curvature—governs where Ras ends up in the cell and is therefore likely to be a factor in cancer development.”
All of the research so far has been conducted in vitro. Dr Stamou said the next big challenge is to uncover how these effects play out in living systems.
“It will be 10 times more difficult to uncover these effects in living systems, but it needs to happen,” he said. “We have started, and we really hope others will follow. It may prove complicated to develop a drug that changes the shape of cells, but I am certain that the discovery of the shape/misregulation-correlation will at least lead to new ways to diagnose cancers.”

Credit: Jes Andersen/
University of Copenhagen
Researchers say they have discovered how Ras proteins find their proper place in cells, a finding that may aid the development of novel approaches to treat cancers.
The team noted that cancers develop if Ras proteins start to trigger misregulation, and Ras misregulates if it misses its correct location on the cell wall—the membrane.
What the researchers discovered is that Ras cannot reach its designated location if the membrane has the wrong shape.
“If the curvature of the cell is right, Ras goes to the right place,” said Dimitrios Stamou, PhD, of the University of Copenhagen in Denmark.
“If the membrane is too straight or too bent, it does not. And Ras is very much like any other worker. If it never finds the way to its workplace, it is not likely to get any work done.”
Dr Stamou and his colleagues described this discovery in Nature Chemical Biology.
Ras proteins are thought to be misregulated in upwards of 30% of all cancers. For 3 decades, researchers have been searching for ways to quell the killer protein.
Their lack of success has given Ras a reputation as the “undruggable cancer target,” but Dr Stamou believes we can change by moving in a new direction.
“If Ras goes off the rails because of changes in the curvature of the cell, perhaps we should target whatever changes the shape of the cell membrane,” he said.
Looking for a correlation between cell shape and Ras misregulation was unusual, even bordering on controversial, said study author Jannik Bruun Larsen, PhD, of the University of Copenhagen.
The researchers were investigating how Ras proteins attach themselves to the cell wall, and Dr Larsen tried to attach Ras to a variety of simulated cell membranes formed into small spheres or vesicles of varying sizes.
He found that Ras would attach more readily to smaller spheres, which were more curved than the large ones, and Dr Larsen started to see a pattern.
“For more than a decade, people thought that the constituents of the cell wall was the thing that controlled where Ras was localized,” Dr Larson said. “We have shown that at least one other aspect—namely, membrane curvature—governs where Ras ends up in the cell and is therefore likely to be a factor in cancer development.”
All of the research so far has been conducted in vitro. Dr Stamou said the next big challenge is to uncover how these effects play out in living systems.
“It will be 10 times more difficult to uncover these effects in living systems, but it needs to happen,” he said. “We have started, and we really hope others will follow. It may prove complicated to develop a drug that changes the shape of cells, but I am certain that the discovery of the shape/misregulation-correlation will at least lead to new ways to diagnose cancers.”

Credit: Jes Andersen/
University of Copenhagen
Researchers say they have discovered how Ras proteins find their proper place in cells, a finding that may aid the development of novel approaches to treat cancers.
The team noted that cancers develop if Ras proteins start to trigger misregulation, and Ras misregulates if it misses its correct location on the cell wall—the membrane.
What the researchers discovered is that Ras cannot reach its designated location if the membrane has the wrong shape.
“If the curvature of the cell is right, Ras goes to the right place,” said Dimitrios Stamou, PhD, of the University of Copenhagen in Denmark.
“If the membrane is too straight or too bent, it does not. And Ras is very much like any other worker. If it never finds the way to its workplace, it is not likely to get any work done.”
Dr Stamou and his colleagues described this discovery in Nature Chemical Biology.
Ras proteins are thought to be misregulated in upwards of 30% of all cancers. For 3 decades, researchers have been searching for ways to quell the killer protein.
Their lack of success has given Ras a reputation as the “undruggable cancer target,” but Dr Stamou believes we can change by moving in a new direction.
“If Ras goes off the rails because of changes in the curvature of the cell, perhaps we should target whatever changes the shape of the cell membrane,” he said.
Looking for a correlation between cell shape and Ras misregulation was unusual, even bordering on controversial, said study author Jannik Bruun Larsen, PhD, of the University of Copenhagen.
The researchers were investigating how Ras proteins attach themselves to the cell wall, and Dr Larsen tried to attach Ras to a variety of simulated cell membranes formed into small spheres or vesicles of varying sizes.
He found that Ras would attach more readily to smaller spheres, which were more curved than the large ones, and Dr Larsen started to see a pattern.
“For more than a decade, people thought that the constituents of the cell wall was the thing that controlled where Ras was localized,” Dr Larson said. “We have shown that at least one other aspect—namely, membrane curvature—governs where Ras ends up in the cell and is therefore likely to be a factor in cancer development.”
All of the research so far has been conducted in vitro. Dr Stamou said the next big challenge is to uncover how these effects play out in living systems.
“It will be 10 times more difficult to uncover these effects in living systems, but it needs to happen,” he said. “We have started, and we really hope others will follow. It may prove complicated to develop a drug that changes the shape of cells, but I am certain that the discovery of the shape/misregulation-correlation will at least lead to new ways to diagnose cancers.”