User login
ACA exchanges limiting for patients with blood cancers, report suggests

Credit: CDC
A new report suggests that many health plans in the insurance exchanges mandated by the Affordable Care Act (ACA) will impose high out-of-pocket costs for patients with hematologic malignancies and provide limited access to specialty treatment centers.
Furthermore, although the plans analyzed appear to provide adequate coverage of hematology/oncology drugs, most require prior authorization.
In other words, the insurer must be notified and may not approve the purchase of a drug based on medical evidence or other criteria.
This report, “2014 Individual Exchange Policies in Four States: An Early Look for Patients with Blood Cancers,” was commissioned by the Leukemia & Lymphoma Society and prepared by Milliman, Inc.
It provides a look at the 2014 individual benefit designs, coverage benefits, and premiums for policies sold on 4 state health insurance exchanges—California, New York, Florida, and Texas—with a focus on items of interest for patients with hematologic malignancies.
“[W]hile many new rules under ACA make obtaining insurance easier for people with blood cancers, such as prohibiting companies from turning away patients with pre-existing conditions and eliminating lifetime coverage limitations, the Milliman report identifies several areas of concern that we want cancer patients to be aware of and policymakers to address,” said Mark Velleca, MD, PhD, chief policy and advocacy officer of the Leukemia & Lymphoma Society.
Premium costs
To compare monthly premium rates, the report’s authors captured rates for a 50-year-old non-smoker with an annual income of $90,000 residing in Houston, Los Angeles, Miami, or New York City.
They found considerable variation according to plan type and location, but overall, plans were cheapest in Houston. Monthly premiums for Houston ranged from $234 to $520. The range was $274 to $566 for Los Angeles, $277 to $635 for Miami, and $307 to $896 for New York.
The ranges reflect the costs according to plan type. Each insurer offers 4 different health plans: Platinum (about 10% cost-sharing), Gold (roughly 20%), Silver (roughly 30%), and Bronze (roughly 40%).
Cost-sharing
The authors noted that the lower-tier Bronze and Silver plans require significant cost-sharing for patients. The report revealed high deductibles in the health plans, sometimes nearly as high as the out-of-pocket ceiling.
Deductibles for the Silver and Bronze plans are often at least $2000 and $4000, respectively, for individuals. The maximum out-of-pocket limits set for 2014 are $6350 for an individual policy and $12,799 for a family policy.
Some insurers offer plans in some states with lower out-of-pocket limits. However, the out-of-pocket limit does not apply to non-covered drugs or treatment centers.
Drug coverage
When analyzing drug coverage, the authors decided to look at 3 drugs used to treat chronic myeloid leukemia—imatinib (Gleevec), nilotinib (Tasigna), and dasatinib (Sprycel)—and 5 drugs used to treat multiple myeloma—thalidomide (Thalomid), lenalidomide (Revlimid), pomalidomide (Pomalyst), cyclophosphamide (Cytoxan), and melphalan (Alkeran).
Most of the insurers require prior authorization for these drugs, but most of them cover all 3 chronic myeloid leukemia drugs and a majority of the myeloma drugs. Pomalyst and Cytoxan are often not covered, although most insurers do cover generic cyclophosphamide.
Network adequacy
Most of the insurers studied do not cover all NCI-designated cancer and transplant centers, and a few do not cover any of these centers. The authors said this could discourage patient enrollment in these plans or mean that a patient’s recommended treatment is not covered.
And since it is unlikely that any out-of-network expenses will count toward a patient’s out-of-pocket maximum, cancer patients could accumulate thousands of dollars of medical expenses and never reach their out-of-pocket maximum.
The authors did note, however, that satisfactory cancer care can be provided outside of NCI-designated cancer and transplant centers.
For more details, see the full report. ![]()

Credit: CDC
A new report suggests that many health plans in the insurance exchanges mandated by the Affordable Care Act (ACA) will impose high out-of-pocket costs for patients with hematologic malignancies and provide limited access to specialty treatment centers.
Furthermore, although the plans analyzed appear to provide adequate coverage of hematology/oncology drugs, most require prior authorization.
In other words, the insurer must be notified and may not approve the purchase of a drug based on medical evidence or other criteria.
This report, “2014 Individual Exchange Policies in Four States: An Early Look for Patients with Blood Cancers,” was commissioned by the Leukemia & Lymphoma Society and prepared by Milliman, Inc.
It provides a look at the 2014 individual benefit designs, coverage benefits, and premiums for policies sold on 4 state health insurance exchanges—California, New York, Florida, and Texas—with a focus on items of interest for patients with hematologic malignancies.
“[W]hile many new rules under ACA make obtaining insurance easier for people with blood cancers, such as prohibiting companies from turning away patients with pre-existing conditions and eliminating lifetime coverage limitations, the Milliman report identifies several areas of concern that we want cancer patients to be aware of and policymakers to address,” said Mark Velleca, MD, PhD, chief policy and advocacy officer of the Leukemia & Lymphoma Society.
Premium costs
To compare monthly premium rates, the report’s authors captured rates for a 50-year-old non-smoker with an annual income of $90,000 residing in Houston, Los Angeles, Miami, or New York City.
They found considerable variation according to plan type and location, but overall, plans were cheapest in Houston. Monthly premiums for Houston ranged from $234 to $520. The range was $274 to $566 for Los Angeles, $277 to $635 for Miami, and $307 to $896 for New York.
The ranges reflect the costs according to plan type. Each insurer offers 4 different health plans: Platinum (about 10% cost-sharing), Gold (roughly 20%), Silver (roughly 30%), and Bronze (roughly 40%).
Cost-sharing
The authors noted that the lower-tier Bronze and Silver plans require significant cost-sharing for patients. The report revealed high deductibles in the health plans, sometimes nearly as high as the out-of-pocket ceiling.
Deductibles for the Silver and Bronze plans are often at least $2000 and $4000, respectively, for individuals. The maximum out-of-pocket limits set for 2014 are $6350 for an individual policy and $12,799 for a family policy.
Some insurers offer plans in some states with lower out-of-pocket limits. However, the out-of-pocket limit does not apply to non-covered drugs or treatment centers.
Drug coverage
When analyzing drug coverage, the authors decided to look at 3 drugs used to treat chronic myeloid leukemia—imatinib (Gleevec), nilotinib (Tasigna), and dasatinib (Sprycel)—and 5 drugs used to treat multiple myeloma—thalidomide (Thalomid), lenalidomide (Revlimid), pomalidomide (Pomalyst), cyclophosphamide (Cytoxan), and melphalan (Alkeran).
Most of the insurers require prior authorization for these drugs, but most of them cover all 3 chronic myeloid leukemia drugs and a majority of the myeloma drugs. Pomalyst and Cytoxan are often not covered, although most insurers do cover generic cyclophosphamide.
Network adequacy
Most of the insurers studied do not cover all NCI-designated cancer and transplant centers, and a few do not cover any of these centers. The authors said this could discourage patient enrollment in these plans or mean that a patient’s recommended treatment is not covered.
And since it is unlikely that any out-of-network expenses will count toward a patient’s out-of-pocket maximum, cancer patients could accumulate thousands of dollars of medical expenses and never reach their out-of-pocket maximum.
The authors did note, however, that satisfactory cancer care can be provided outside of NCI-designated cancer and transplant centers.
For more details, see the full report. ![]()

Credit: CDC
A new report suggests that many health plans in the insurance exchanges mandated by the Affordable Care Act (ACA) will impose high out-of-pocket costs for patients with hematologic malignancies and provide limited access to specialty treatment centers.
Furthermore, although the plans analyzed appear to provide adequate coverage of hematology/oncology drugs, most require prior authorization.
In other words, the insurer must be notified and may not approve the purchase of a drug based on medical evidence or other criteria.
This report, “2014 Individual Exchange Policies in Four States: An Early Look for Patients with Blood Cancers,” was commissioned by the Leukemia & Lymphoma Society and prepared by Milliman, Inc.
It provides a look at the 2014 individual benefit designs, coverage benefits, and premiums for policies sold on 4 state health insurance exchanges—California, New York, Florida, and Texas—with a focus on items of interest for patients with hematologic malignancies.
“[W]hile many new rules under ACA make obtaining insurance easier for people with blood cancers, such as prohibiting companies from turning away patients with pre-existing conditions and eliminating lifetime coverage limitations, the Milliman report identifies several areas of concern that we want cancer patients to be aware of and policymakers to address,” said Mark Velleca, MD, PhD, chief policy and advocacy officer of the Leukemia & Lymphoma Society.
Premium costs
To compare monthly premium rates, the report’s authors captured rates for a 50-year-old non-smoker with an annual income of $90,000 residing in Houston, Los Angeles, Miami, or New York City.
They found considerable variation according to plan type and location, but overall, plans were cheapest in Houston. Monthly premiums for Houston ranged from $234 to $520. The range was $274 to $566 for Los Angeles, $277 to $635 for Miami, and $307 to $896 for New York.
The ranges reflect the costs according to plan type. Each insurer offers 4 different health plans: Platinum (about 10% cost-sharing), Gold (roughly 20%), Silver (roughly 30%), and Bronze (roughly 40%).
Cost-sharing
The authors noted that the lower-tier Bronze and Silver plans require significant cost-sharing for patients. The report revealed high deductibles in the health plans, sometimes nearly as high as the out-of-pocket ceiling.
Deductibles for the Silver and Bronze plans are often at least $2000 and $4000, respectively, for individuals. The maximum out-of-pocket limits set for 2014 are $6350 for an individual policy and $12,799 for a family policy.
Some insurers offer plans in some states with lower out-of-pocket limits. However, the out-of-pocket limit does not apply to non-covered drugs or treatment centers.
Drug coverage
When analyzing drug coverage, the authors decided to look at 3 drugs used to treat chronic myeloid leukemia—imatinib (Gleevec), nilotinib (Tasigna), and dasatinib (Sprycel)—and 5 drugs used to treat multiple myeloma—thalidomide (Thalomid), lenalidomide (Revlimid), pomalidomide (Pomalyst), cyclophosphamide (Cytoxan), and melphalan (Alkeran).
Most of the insurers require prior authorization for these drugs, but most of them cover all 3 chronic myeloid leukemia drugs and a majority of the myeloma drugs. Pomalyst and Cytoxan are often not covered, although most insurers do cover generic cyclophosphamide.
Network adequacy
Most of the insurers studied do not cover all NCI-designated cancer and transplant centers, and a few do not cover any of these centers. The authors said this could discourage patient enrollment in these plans or mean that a patient’s recommended treatment is not covered.
And since it is unlikely that any out-of-network expenses will count toward a patient’s out-of-pocket maximum, cancer patients could accumulate thousands of dollars of medical expenses and never reach their out-of-pocket maximum.
The authors did note, however, that satisfactory cancer care can be provided outside of NCI-designated cancer and transplant centers.
For more details, see the full report. ![]()
Compound active against a range of cancers
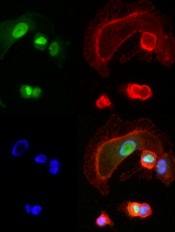
expressing NFAT3c-GFP
A little-studied chemical compound has “wide and potent” anticancer activity, investigators have reported in Cancer Cell.
The compound, BMH-21, works by inhibiting the RNA polymerase transcription pathway (Pol I), thereby preventing cancer cell communication and replication.
“Without this transcription machinery, cancer cells cannot function,” said study author Marikki Laiho, MD, PhD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
She and her colleagues homed in on BMH-21 by screening a library of chemical compounds thought to have potential for anticancer activity.
Specifically, the team looked at the compounds’ ability to interfere with transcription in the National Cancer Institute’s collection of 60 human tumor cell lines (known as NCI-60).
BMH-21 demonstrated activity against all 9 cancer types studied—leukemia and melanoma, as well as breast, CNS, colon, lung, ovarian, prostate, and renal cancers.
The drug also repressed tumor growth in mouse models of colon cancer and melanoma.
Additional analyses showed that BMH-21 inhibited Pol I transcription and caused disintegration of the nucleolus. The drug activated loss of the Pol I catalytic subunit RPA194, which led to disassembly of the Pol I holocomplex from the ribosomal DNA.
And the loss of RPA194, which was a result of increased proteasome-mediated turnover, was associated with decreased cancer cell viability.
Dr Laiho and her colleagues are continuing studies of BMH-21 in animal models to confirm the drug’s anticancer activity, identify any toxicities associated with the compound, and determine the optimal dose.
And because Pol I activity is frequently deregulated in cancers, the investigators believe BMH-21 could have therapeutic potential for many malignancies.
Dr Laiho is currently collaborating with experts in multiple myeloma, medullary thyroid cancer, and prostate cancer to explore the drug’s activity in these malignancies. ![]()

expressing NFAT3c-GFP
A little-studied chemical compound has “wide and potent” anticancer activity, investigators have reported in Cancer Cell.
The compound, BMH-21, works by inhibiting the RNA polymerase transcription pathway (Pol I), thereby preventing cancer cell communication and replication.
“Without this transcription machinery, cancer cells cannot function,” said study author Marikki Laiho, MD, PhD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
She and her colleagues homed in on BMH-21 by screening a library of chemical compounds thought to have potential for anticancer activity.
Specifically, the team looked at the compounds’ ability to interfere with transcription in the National Cancer Institute’s collection of 60 human tumor cell lines (known as NCI-60).
BMH-21 demonstrated activity against all 9 cancer types studied—leukemia and melanoma, as well as breast, CNS, colon, lung, ovarian, prostate, and renal cancers.
The drug also repressed tumor growth in mouse models of colon cancer and melanoma.
Additional analyses showed that BMH-21 inhibited Pol I transcription and caused disintegration of the nucleolus. The drug activated loss of the Pol I catalytic subunit RPA194, which led to disassembly of the Pol I holocomplex from the ribosomal DNA.
And the loss of RPA194, which was a result of increased proteasome-mediated turnover, was associated with decreased cancer cell viability.
Dr Laiho and her colleagues are continuing studies of BMH-21 in animal models to confirm the drug’s anticancer activity, identify any toxicities associated with the compound, and determine the optimal dose.
And because Pol I activity is frequently deregulated in cancers, the investigators believe BMH-21 could have therapeutic potential for many malignancies.
Dr Laiho is currently collaborating with experts in multiple myeloma, medullary thyroid cancer, and prostate cancer to explore the drug’s activity in these malignancies. ![]()

expressing NFAT3c-GFP
A little-studied chemical compound has “wide and potent” anticancer activity, investigators have reported in Cancer Cell.
The compound, BMH-21, works by inhibiting the RNA polymerase transcription pathway (Pol I), thereby preventing cancer cell communication and replication.
“Without this transcription machinery, cancer cells cannot function,” said study author Marikki Laiho, MD, PhD, of the Johns Hopkins University School of Medicine in Baltimore, Maryland.
She and her colleagues homed in on BMH-21 by screening a library of chemical compounds thought to have potential for anticancer activity.
Specifically, the team looked at the compounds’ ability to interfere with transcription in the National Cancer Institute’s collection of 60 human tumor cell lines (known as NCI-60).
BMH-21 demonstrated activity against all 9 cancer types studied—leukemia and melanoma, as well as breast, CNS, colon, lung, ovarian, prostate, and renal cancers.
The drug also repressed tumor growth in mouse models of colon cancer and melanoma.
Additional analyses showed that BMH-21 inhibited Pol I transcription and caused disintegration of the nucleolus. The drug activated loss of the Pol I catalytic subunit RPA194, which led to disassembly of the Pol I holocomplex from the ribosomal DNA.
And the loss of RPA194, which was a result of increased proteasome-mediated turnover, was associated with decreased cancer cell viability.
Dr Laiho and her colleagues are continuing studies of BMH-21 in animal models to confirm the drug’s anticancer activity, identify any toxicities associated with the compound, and determine the optimal dose.
And because Pol I activity is frequently deregulated in cancers, the investigators believe BMH-21 could have therapeutic potential for many malignancies.
Dr Laiho is currently collaborating with experts in multiple myeloma, medullary thyroid cancer, and prostate cancer to explore the drug’s activity in these malignancies. ![]()
Study reveals ‘widespread’ genetic heterogeneity in MM
different colored cells
Credit: Lauren Solomon
Results of a new study suggest the genetic landscape of multiple myeloma (MM) may be more complex than we thought.
The research revealed “widespread” heterogeneity in samples from more than 200 MM patients.
In some cases, a single patient had multiple mutations in the same pathway. And most of the patients harbored at least 3 detectable subclonal mutations.
The researchers said these findings, published in Cancer Cell, might explain why targeted therapies are not always effective in MM and why some patients relapse after treatment.
“What this new work shows us is that when we treat an individual patient with multiple myeloma, it’s possible that we’re not just looking at one disease, but at many,” said study author Todd Golub, MD, of the Dana-Farber Cancer Institute in Cambridge, Massachusetts.
“In the same person, there could be cancer cells with different genetic make-ups. These findings indicate a need to identify the extent of genetic diversity within a tumor as we move toward precision cancer medicine and genome-based diagnostics.”
Dr Golub and his colleagues studied samples from 203 MM patients and identified frequent mutations in genes known to play an important role in MM, including KRAS, NRAS, and BRAF.
But many of these telltale mutations were not present in all MM cells. Instead, they were often observed only in a subclonal population.
This suggests targeted therapies may have limitations in patients whose tumors are made up of these subclonal populations, the researchers said.
To explore the therapeutic implications of this research, the team performed follow-up experiments looking specifically at BRAF, a gene for which several inhibitors exist.
Previous studies indicated that roughly 4% of MM patients may have mutations in this gene. And a recent report on a single MM patient treated with drugs targeting BRAF showed promising results.
However, Dr Golub and his colleagues found evidence that treating a tumor harboring subclonal BRAF mutations with one of these agents may, at best, kill a fraction of the cells and, at worst, stimulate another cancer cell subpopulation to grow.
“There’s clearly potential for these drugs in some patients with multiple myeloma, but we show that there are also potential problems for others,” said study author Jens Lohr, MD, PhD, also of Dana-Farber.
“If a patient has a BRAF mutation in less than 100% of his cells, or if he has mutations in KRAS or NRAS at the same time, [it] may influence the response to an inhibitor.”
This suggests subclonal populations could be one of the reasons many patients suffer relapse after treatment, the researchers said.
“Matching the right drug to the right patient may not be as easy as finding a mutation and having a drug that targets it,” Dr Lohr said. “We have to keep this additional parameter of heterogeneity in mind and keep exploring what it means for therapy.” ![]()

different colored cells
Credit: Lauren Solomon
Results of a new study suggest the genetic landscape of multiple myeloma (MM) may be more complex than we thought.
The research revealed “widespread” heterogeneity in samples from more than 200 MM patients.
In some cases, a single patient had multiple mutations in the same pathway. And most of the patients harbored at least 3 detectable subclonal mutations.
The researchers said these findings, published in Cancer Cell, might explain why targeted therapies are not always effective in MM and why some patients relapse after treatment.
“What this new work shows us is that when we treat an individual patient with multiple myeloma, it’s possible that we’re not just looking at one disease, but at many,” said study author Todd Golub, MD, of the Dana-Farber Cancer Institute in Cambridge, Massachusetts.
“In the same person, there could be cancer cells with different genetic make-ups. These findings indicate a need to identify the extent of genetic diversity within a tumor as we move toward precision cancer medicine and genome-based diagnostics.”
Dr Golub and his colleagues studied samples from 203 MM patients and identified frequent mutations in genes known to play an important role in MM, including KRAS, NRAS, and BRAF.
But many of these telltale mutations were not present in all MM cells. Instead, they were often observed only in a subclonal population.
This suggests targeted therapies may have limitations in patients whose tumors are made up of these subclonal populations, the researchers said.
To explore the therapeutic implications of this research, the team performed follow-up experiments looking specifically at BRAF, a gene for which several inhibitors exist.
Previous studies indicated that roughly 4% of MM patients may have mutations in this gene. And a recent report on a single MM patient treated with drugs targeting BRAF showed promising results.
However, Dr Golub and his colleagues found evidence that treating a tumor harboring subclonal BRAF mutations with one of these agents may, at best, kill a fraction of the cells and, at worst, stimulate another cancer cell subpopulation to grow.
“There’s clearly potential for these drugs in some patients with multiple myeloma, but we show that there are also potential problems for others,” said study author Jens Lohr, MD, PhD, also of Dana-Farber.
“If a patient has a BRAF mutation in less than 100% of his cells, or if he has mutations in KRAS or NRAS at the same time, [it] may influence the response to an inhibitor.”
This suggests subclonal populations could be one of the reasons many patients suffer relapse after treatment, the researchers said.
“Matching the right drug to the right patient may not be as easy as finding a mutation and having a drug that targets it,” Dr Lohr said. “We have to keep this additional parameter of heterogeneity in mind and keep exploring what it means for therapy.” ![]()

different colored cells
Credit: Lauren Solomon
Results of a new study suggest the genetic landscape of multiple myeloma (MM) may be more complex than we thought.
The research revealed “widespread” heterogeneity in samples from more than 200 MM patients.
In some cases, a single patient had multiple mutations in the same pathway. And most of the patients harbored at least 3 detectable subclonal mutations.
The researchers said these findings, published in Cancer Cell, might explain why targeted therapies are not always effective in MM and why some patients relapse after treatment.
“What this new work shows us is that when we treat an individual patient with multiple myeloma, it’s possible that we’re not just looking at one disease, but at many,” said study author Todd Golub, MD, of the Dana-Farber Cancer Institute in Cambridge, Massachusetts.
“In the same person, there could be cancer cells with different genetic make-ups. These findings indicate a need to identify the extent of genetic diversity within a tumor as we move toward precision cancer medicine and genome-based diagnostics.”
Dr Golub and his colleagues studied samples from 203 MM patients and identified frequent mutations in genes known to play an important role in MM, including KRAS, NRAS, and BRAF.
But many of these telltale mutations were not present in all MM cells. Instead, they were often observed only in a subclonal population.
This suggests targeted therapies may have limitations in patients whose tumors are made up of these subclonal populations, the researchers said.
To explore the therapeutic implications of this research, the team performed follow-up experiments looking specifically at BRAF, a gene for which several inhibitors exist.
Previous studies indicated that roughly 4% of MM patients may have mutations in this gene. And a recent report on a single MM patient treated with drugs targeting BRAF showed promising results.
However, Dr Golub and his colleagues found evidence that treating a tumor harboring subclonal BRAF mutations with one of these agents may, at best, kill a fraction of the cells and, at worst, stimulate another cancer cell subpopulation to grow.
“There’s clearly potential for these drugs in some patients with multiple myeloma, but we show that there are also potential problems for others,” said study author Jens Lohr, MD, PhD, also of Dana-Farber.
“If a patient has a BRAF mutation in less than 100% of his cells, or if he has mutations in KRAS or NRAS at the same time, [it] may influence the response to an inhibitor.”
This suggests subclonal populations could be one of the reasons many patients suffer relapse after treatment, the researchers said.
“Matching the right drug to the right patient may not be as easy as finding a mutation and having a drug that targets it,” Dr Lohr said. “We have to keep this additional parameter of heterogeneity in mind and keep exploring what it means for therapy.” ![]()
Interventions can ease insomnia in cancer patients

Credit: RelaxingMusic
A new study suggests cancer patients struggling with insomnia can choose between 2 behavioral interventions to obtain relief: cognitive behavioral therapy for insomnia (CBT-I) and mindfulness-based stress reduction (MBSR).
CBT-I is the gold standard of care, but the research showed that MBSR can also help improve sleep for cancer patients.
CBT-I involves stimulus control, sleep restriction, cognitive therapy, and relaxation training. When combined, these strategies target and reduce sleep-related physiologic and cognitive arousal to re-establish restorative sleep.
MBSR provides patients with psychoeducation on the relationship between stress and health. It also employs meditation techniques and gentle yoga to support mindful awareness and help patients respond better to stress.
Previous research has shown that MBSR can reduce distress and improve psychological well-being in patients with cancer. But this is the first study to directly compare MBSR to CBT-I in cancer patients.
The results are published in the Journal of Clinical Oncology.
“Insomnia and disturbed sleep are significant problems that can affect approximately half of all cancer patients,” said lead study author Sheila Garland, PhD, of Abramson Cancer Center at the University of Pennsylvania in Philadelphia.
“If not properly addressed, sleep disturbances can negatively influence therapeutic and supportive care measures for these patients, so it’s critical that clinicians can offer patients reliable, effective, and tailored interventions.”
With this in mind, Dr Garland and her colleagues tested behavioral interventions for insomnia in 111 patients recruited from a cancer center in Calgary, Alberta, Canada. Patients were randomized to either a CBT-I program (n=47) or an MBSR program (n=64) for 8 weeks.
Thirty-two patients completed the CBT-I program, and 40 completed the MBSR program. The researchers assessed patients immediately after program completion (at 2 months) and at 5 months from baseline.
Immediately after completion, MBSR was less effective than CBT-I at improving insomnia severity (P=0.35). But at the 5-month follow-up point, MBSR proved noninferior to CBT-I (P=0.02).
Patients in the CBT-I group showed greater overall improvement in subjectively measured sleep onset latency, sleep efficiency, sleep quality, and dysfunctional sleep beliefs than patients in the MBSR group.
But both groups showed progressive improvement over time when it came to subjectively measured total sleep time, wake after sleep onset, stress, and mood disturbance.
“That MBSR can produce similar improvements to CBT-I and that both [interventions] can effectively reduce stress and mood disturbance expands the available treatment options for insomnia in cancer patients,” Dr Garland said.
“This study suggests that we should not apply a ‘one-size-fits-all model’ to the treatment of insomnia and emphasizes the need to individualize treatment based on patient characteristics and preferences.” ![]()

Credit: RelaxingMusic
A new study suggests cancer patients struggling with insomnia can choose between 2 behavioral interventions to obtain relief: cognitive behavioral therapy for insomnia (CBT-I) and mindfulness-based stress reduction (MBSR).
CBT-I is the gold standard of care, but the research showed that MBSR can also help improve sleep for cancer patients.
CBT-I involves stimulus control, sleep restriction, cognitive therapy, and relaxation training. When combined, these strategies target and reduce sleep-related physiologic and cognitive arousal to re-establish restorative sleep.
MBSR provides patients with psychoeducation on the relationship between stress and health. It also employs meditation techniques and gentle yoga to support mindful awareness and help patients respond better to stress.
Previous research has shown that MBSR can reduce distress and improve psychological well-being in patients with cancer. But this is the first study to directly compare MBSR to CBT-I in cancer patients.
The results are published in the Journal of Clinical Oncology.
“Insomnia and disturbed sleep are significant problems that can affect approximately half of all cancer patients,” said lead study author Sheila Garland, PhD, of Abramson Cancer Center at the University of Pennsylvania in Philadelphia.
“If not properly addressed, sleep disturbances can negatively influence therapeutic and supportive care measures for these patients, so it’s critical that clinicians can offer patients reliable, effective, and tailored interventions.”
With this in mind, Dr Garland and her colleagues tested behavioral interventions for insomnia in 111 patients recruited from a cancer center in Calgary, Alberta, Canada. Patients were randomized to either a CBT-I program (n=47) or an MBSR program (n=64) for 8 weeks.
Thirty-two patients completed the CBT-I program, and 40 completed the MBSR program. The researchers assessed patients immediately after program completion (at 2 months) and at 5 months from baseline.
Immediately after completion, MBSR was less effective than CBT-I at improving insomnia severity (P=0.35). But at the 5-month follow-up point, MBSR proved noninferior to CBT-I (P=0.02).
Patients in the CBT-I group showed greater overall improvement in subjectively measured sleep onset latency, sleep efficiency, sleep quality, and dysfunctional sleep beliefs than patients in the MBSR group.
But both groups showed progressive improvement over time when it came to subjectively measured total sleep time, wake after sleep onset, stress, and mood disturbance.
“That MBSR can produce similar improvements to CBT-I and that both [interventions] can effectively reduce stress and mood disturbance expands the available treatment options for insomnia in cancer patients,” Dr Garland said.
“This study suggests that we should not apply a ‘one-size-fits-all model’ to the treatment of insomnia and emphasizes the need to individualize treatment based on patient characteristics and preferences.” ![]()

Credit: RelaxingMusic
A new study suggests cancer patients struggling with insomnia can choose between 2 behavioral interventions to obtain relief: cognitive behavioral therapy for insomnia (CBT-I) and mindfulness-based stress reduction (MBSR).
CBT-I is the gold standard of care, but the research showed that MBSR can also help improve sleep for cancer patients.
CBT-I involves stimulus control, sleep restriction, cognitive therapy, and relaxation training. When combined, these strategies target and reduce sleep-related physiologic and cognitive arousal to re-establish restorative sleep.
MBSR provides patients with psychoeducation on the relationship between stress and health. It also employs meditation techniques and gentle yoga to support mindful awareness and help patients respond better to stress.
Previous research has shown that MBSR can reduce distress and improve psychological well-being in patients with cancer. But this is the first study to directly compare MBSR to CBT-I in cancer patients.
The results are published in the Journal of Clinical Oncology.
“Insomnia and disturbed sleep are significant problems that can affect approximately half of all cancer patients,” said lead study author Sheila Garland, PhD, of Abramson Cancer Center at the University of Pennsylvania in Philadelphia.
“If not properly addressed, sleep disturbances can negatively influence therapeutic and supportive care measures for these patients, so it’s critical that clinicians can offer patients reliable, effective, and tailored interventions.”
With this in mind, Dr Garland and her colleagues tested behavioral interventions for insomnia in 111 patients recruited from a cancer center in Calgary, Alberta, Canada. Patients were randomized to either a CBT-I program (n=47) or an MBSR program (n=64) for 8 weeks.
Thirty-two patients completed the CBT-I program, and 40 completed the MBSR program. The researchers assessed patients immediately after program completion (at 2 months) and at 5 months from baseline.
Immediately after completion, MBSR was less effective than CBT-I at improving insomnia severity (P=0.35). But at the 5-month follow-up point, MBSR proved noninferior to CBT-I (P=0.02).
Patients in the CBT-I group showed greater overall improvement in subjectively measured sleep onset latency, sleep efficiency, sleep quality, and dysfunctional sleep beliefs than patients in the MBSR group.
But both groups showed progressive improvement over time when it came to subjectively measured total sleep time, wake after sleep onset, stress, and mood disturbance.
“That MBSR can produce similar improvements to CBT-I and that both [interventions] can effectively reduce stress and mood disturbance expands the available treatment options for insomnia in cancer patients,” Dr Garland said.
“This study suggests that we should not apply a ‘one-size-fits-all model’ to the treatment of insomnia and emphasizes the need to individualize treatment based on patient characteristics and preferences.” ![]()
Leukemia is leading cause of cancer death among young Americans

receiving chemotherapy
Credit: Rhoda Baer
Leukemia is the leading cause of cancer death in the US for men under 40 and women aged 20 and younger, according to a report by the American Cancer Society.
Non-Hodgkin lymphoma (NHL) is also among the 5 leading causes of cancer death for men under 40 and for women age 80 and older.
These data appear in “Cancer Statistics, 2014,” a report published in CA: A Cancer Journal for Clinicians.
The report includes statistics on cancer incidence and death from 1975 to 2010, as well as projections for 2014.
In the latest data (from 2010), NHL was the fifth leading cause of cancer death for men under 20 and for women over 79. It was the fourth leading cause of cancer death for men ages 20 to 39.
And leukemia was the third leading cause of cancer death for women ages 20 to 39, in addition to being the leading cause of cancer death for women under 21 and men under 40.
However, of all cancer types, leukemia and NHL have seen the largest improvements in survival, according to data comparing 5-year survival rates between 1975-1977 and 2003-2009.
Five-year survival rates for leukemia were 34% for 1975-1977 and 59% for 2003-2009 (P<0.05). For NHL, 5-year survival rates were 47% for 1975-1977 and 71% for 2003-2009 (P<0.05).
Projections for 2014
The report authors took past data into account to make estimates on cancer incidence and death for 2014. They projected that 1,665,540 patients will be diagnosed with cancer this year, and 585,720 patients will die of cancer.
Roughly 79,990 patients will be diagnosed with lymphoma—9190 with Hodgkin lymphoma and 70,800 with NHL. Approximately 18,990 patients will die of NHL, and 1180 will die of Hodgkin lymphoma.
There will be 24,050 new cases of myeloma in 2014 and 11,090 myeloma deaths, the authors said.
This year will see 52,380 patients diagnosed with leukemias—6020 with acute lymphocytic leukemia (ALL), 15,720 with chronic lymphocytic leukemia (CLL), 18,860 with acute myeloid leukemia (AML), 5980 with chronic myeloid leukemia (CML), and 5800 with other types of leukemia.
And there will be 24,090 leukemia deaths—1440 from ALL, 4600 from CLL, 10,460 from AML, 810 from CML, and 6780 from other leukemias.
For more information, see the complete report. ![]()

receiving chemotherapy
Credit: Rhoda Baer
Leukemia is the leading cause of cancer death in the US for men under 40 and women aged 20 and younger, according to a report by the American Cancer Society.
Non-Hodgkin lymphoma (NHL) is also among the 5 leading causes of cancer death for men under 40 and for women age 80 and older.
These data appear in “Cancer Statistics, 2014,” a report published in CA: A Cancer Journal for Clinicians.
The report includes statistics on cancer incidence and death from 1975 to 2010, as well as projections for 2014.
In the latest data (from 2010), NHL was the fifth leading cause of cancer death for men under 20 and for women over 79. It was the fourth leading cause of cancer death for men ages 20 to 39.
And leukemia was the third leading cause of cancer death for women ages 20 to 39, in addition to being the leading cause of cancer death for women under 21 and men under 40.
However, of all cancer types, leukemia and NHL have seen the largest improvements in survival, according to data comparing 5-year survival rates between 1975-1977 and 2003-2009.
Five-year survival rates for leukemia were 34% for 1975-1977 and 59% for 2003-2009 (P<0.05). For NHL, 5-year survival rates were 47% for 1975-1977 and 71% for 2003-2009 (P<0.05).
Projections for 2014
The report authors took past data into account to make estimates on cancer incidence and death for 2014. They projected that 1,665,540 patients will be diagnosed with cancer this year, and 585,720 patients will die of cancer.
Roughly 79,990 patients will be diagnosed with lymphoma—9190 with Hodgkin lymphoma and 70,800 with NHL. Approximately 18,990 patients will die of NHL, and 1180 will die of Hodgkin lymphoma.
There will be 24,050 new cases of myeloma in 2014 and 11,090 myeloma deaths, the authors said.
This year will see 52,380 patients diagnosed with leukemias—6020 with acute lymphocytic leukemia (ALL), 15,720 with chronic lymphocytic leukemia (CLL), 18,860 with acute myeloid leukemia (AML), 5980 with chronic myeloid leukemia (CML), and 5800 with other types of leukemia.
And there will be 24,090 leukemia deaths—1440 from ALL, 4600 from CLL, 10,460 from AML, 810 from CML, and 6780 from other leukemias.
For more information, see the complete report. ![]()

receiving chemotherapy
Credit: Rhoda Baer
Leukemia is the leading cause of cancer death in the US for men under 40 and women aged 20 and younger, according to a report by the American Cancer Society.
Non-Hodgkin lymphoma (NHL) is also among the 5 leading causes of cancer death for men under 40 and for women age 80 and older.
These data appear in “Cancer Statistics, 2014,” a report published in CA: A Cancer Journal for Clinicians.
The report includes statistics on cancer incidence and death from 1975 to 2010, as well as projections for 2014.
In the latest data (from 2010), NHL was the fifth leading cause of cancer death for men under 20 and for women over 79. It was the fourth leading cause of cancer death for men ages 20 to 39.
And leukemia was the third leading cause of cancer death for women ages 20 to 39, in addition to being the leading cause of cancer death for women under 21 and men under 40.
However, of all cancer types, leukemia and NHL have seen the largest improvements in survival, according to data comparing 5-year survival rates between 1975-1977 and 2003-2009.
Five-year survival rates for leukemia were 34% for 1975-1977 and 59% for 2003-2009 (P<0.05). For NHL, 5-year survival rates were 47% for 1975-1977 and 71% for 2003-2009 (P<0.05).
Projections for 2014
The report authors took past data into account to make estimates on cancer incidence and death for 2014. They projected that 1,665,540 patients will be diagnosed with cancer this year, and 585,720 patients will die of cancer.
Roughly 79,990 patients will be diagnosed with lymphoma—9190 with Hodgkin lymphoma and 70,800 with NHL. Approximately 18,990 patients will die of NHL, and 1180 will die of Hodgkin lymphoma.
There will be 24,050 new cases of myeloma in 2014 and 11,090 myeloma deaths, the authors said.
This year will see 52,380 patients diagnosed with leukemias—6020 with acute lymphocytic leukemia (ALL), 15,720 with chronic lymphocytic leukemia (CLL), 18,860 with acute myeloid leukemia (AML), 5980 with chronic myeloid leukemia (CML), and 5800 with other types of leukemia.
And there will be 24,090 leukemia deaths—1440 from ALL, 4600 from CLL, 10,460 from AML, 810 from CML, and 6780 from other leukemias.
For more information, see the complete report. ![]()
Renal failure in multiple myeloma
This report details the case of a 65-year-old man who was diagnosed with multiple myeloma in 2006 and since 2009, has attempted to control the progression of his disease with the most powerful available treatment regimens, including bortezomib-based regimens, for both induction and consolidation therapy followed by autologous stem-cell transplants. Subsequently, because the patient was deemed treatment refractory, treatment with the newly approved carfilzomib was initiated. Coincidentally, the patient developed acute kidney injury, evidenced by tenfold rise in his creatinine levels, 2 weeks after the initiation of carfilzomib.
Click on the PDF icon at the top of this introduction to read the full article.
This report details the case of a 65-year-old man who was diagnosed with multiple myeloma in 2006 and since 2009, has attempted to control the progression of his disease with the most powerful available treatment regimens, including bortezomib-based regimens, for both induction and consolidation therapy followed by autologous stem-cell transplants. Subsequently, because the patient was deemed treatment refractory, treatment with the newly approved carfilzomib was initiated. Coincidentally, the patient developed acute kidney injury, evidenced by tenfold rise in his creatinine levels, 2 weeks after the initiation of carfilzomib.
Click on the PDF icon at the top of this introduction to read the full article.
This report details the case of a 65-year-old man who was diagnosed with multiple myeloma in 2006 and since 2009, has attempted to control the progression of his disease with the most powerful available treatment regimens, including bortezomib-based regimens, for both induction and consolidation therapy followed by autologous stem-cell transplants. Subsequently, because the patient was deemed treatment refractory, treatment with the newly approved carfilzomib was initiated. Coincidentally, the patient developed acute kidney injury, evidenced by tenfold rise in his creatinine levels, 2 weeks after the initiation of carfilzomib.
Click on the PDF icon at the top of this introduction to read the full article.
FDA approves pomalidomide for MM

Credit: Steven Harbour
The US Food and Drug Administration (FDA) has granted accelerated approval for the immunomodulatory agent pomalidomide (Pomalyst) to treat patients with advanced multiple myeloma (MM).
Continued FDA approval for the drug may be contingent upon verification and description of clinical benefit in confirmatory trials.
Pomalidomide is intended for use in combination with dexamethasone to treat MM patients who have received at least 2 prior
therapies (including lenalidomide and a proteasome inhibitor) and who experienced progression within 60 days of their last treatment.
Pomalidomide has demonstrated some efficacy in this patient population in a number of studies.
In a study published in Blood last year (PG Richardson et al.), pomalidomide elicited responses in MM patients who were refractory to lenalidomide, bortezomib, or both drugs.
In a study presented at ASH 2011 (abstract 634), pomalidomide did not fare as well when given alone to patients with refractory MM. However, combining the drug with low-dose dexamethasone significantly improved responses.
A study presented at ASH 2012 (LBA-6) built upon those findings, showing that pomalidomide plus low-dose dexamethasone was superior to high-dose dexamethasone in MM patients who were refractory to lenalidomide and bortezomib.
Common side effects observed with pomalidomide include neutropenia, anemia, thrombocytopenia, fatigue, weakness, constipation, diarrhea, upper respiratory tract infections, back pain, and fever.
In addition, pomalidomide has been shown to cause venous thromboembolism, as well as severe, life-threatening birth defects in pregnant women. The drug carries a boxed warning alerting patients and healthcare professionals to both of these risks.
Because of the embryo-fetal risk, pomalidomide is available only through the Pomalyst Risk Evaluation and Mitigation Strategy (REMS) Program. Prescribers must be certified with the program by enrolling and complying with the REMS requirements.
Patients must sign a patient-physician agreement form and comply with the REMS requirements. In particular, female patients who are not pregnant but can become pregnant must comply with the pregnancy testing and contraception requirements, and males must comply with contraception requirements.
Pharmacies must be certified with the Pomalyst REMS Program, must only dispense the drug to patients who are authorized to receive it, and must comply with REMS requirements. Both lenalidomide and thalidomide have similar REMS.
Pomalidomide is marketed by Celgene, which is based in Summit, New Jersey. ![]()

Credit: Steven Harbour
The US Food and Drug Administration (FDA) has granted accelerated approval for the immunomodulatory agent pomalidomide (Pomalyst) to treat patients with advanced multiple myeloma (MM).
Continued FDA approval for the drug may be contingent upon verification and description of clinical benefit in confirmatory trials.
Pomalidomide is intended for use in combination with dexamethasone to treat MM patients who have received at least 2 prior
therapies (including lenalidomide and a proteasome inhibitor) and who experienced progression within 60 days of their last treatment.
Pomalidomide has demonstrated some efficacy in this patient population in a number of studies.
In a study published in Blood last year (PG Richardson et al.), pomalidomide elicited responses in MM patients who were refractory to lenalidomide, bortezomib, or both drugs.
In a study presented at ASH 2011 (abstract 634), pomalidomide did not fare as well when given alone to patients with refractory MM. However, combining the drug with low-dose dexamethasone significantly improved responses.
A study presented at ASH 2012 (LBA-6) built upon those findings, showing that pomalidomide plus low-dose dexamethasone was superior to high-dose dexamethasone in MM patients who were refractory to lenalidomide and bortezomib.
Common side effects observed with pomalidomide include neutropenia, anemia, thrombocytopenia, fatigue, weakness, constipation, diarrhea, upper respiratory tract infections, back pain, and fever.
In addition, pomalidomide has been shown to cause venous thromboembolism, as well as severe, life-threatening birth defects in pregnant women. The drug carries a boxed warning alerting patients and healthcare professionals to both of these risks.
Because of the embryo-fetal risk, pomalidomide is available only through the Pomalyst Risk Evaluation and Mitigation Strategy (REMS) Program. Prescribers must be certified with the program by enrolling and complying with the REMS requirements.
Patients must sign a patient-physician agreement form and comply with the REMS requirements. In particular, female patients who are not pregnant but can become pregnant must comply with the pregnancy testing and contraception requirements, and males must comply with contraception requirements.
Pharmacies must be certified with the Pomalyst REMS Program, must only dispense the drug to patients who are authorized to receive it, and must comply with REMS requirements. Both lenalidomide and thalidomide have similar REMS.
Pomalidomide is marketed by Celgene, which is based in Summit, New Jersey. ![]()

Credit: Steven Harbour
The US Food and Drug Administration (FDA) has granted accelerated approval for the immunomodulatory agent pomalidomide (Pomalyst) to treat patients with advanced multiple myeloma (MM).
Continued FDA approval for the drug may be contingent upon verification and description of clinical benefit in confirmatory trials.
Pomalidomide is intended for use in combination with dexamethasone to treat MM patients who have received at least 2 prior
therapies (including lenalidomide and a proteasome inhibitor) and who experienced progression within 60 days of their last treatment.
Pomalidomide has demonstrated some efficacy in this patient population in a number of studies.
In a study published in Blood last year (PG Richardson et al.), pomalidomide elicited responses in MM patients who were refractory to lenalidomide, bortezomib, or both drugs.
In a study presented at ASH 2011 (abstract 634), pomalidomide did not fare as well when given alone to patients with refractory MM. However, combining the drug with low-dose dexamethasone significantly improved responses.
A study presented at ASH 2012 (LBA-6) built upon those findings, showing that pomalidomide plus low-dose dexamethasone was superior to high-dose dexamethasone in MM patients who were refractory to lenalidomide and bortezomib.
Common side effects observed with pomalidomide include neutropenia, anemia, thrombocytopenia, fatigue, weakness, constipation, diarrhea, upper respiratory tract infections, back pain, and fever.
In addition, pomalidomide has been shown to cause venous thromboembolism, as well as severe, life-threatening birth defects in pregnant women. The drug carries a boxed warning alerting patients and healthcare professionals to both of these risks.
Because of the embryo-fetal risk, pomalidomide is available only through the Pomalyst Risk Evaluation and Mitigation Strategy (REMS) Program. Prescribers must be certified with the program by enrolling and complying with the REMS requirements.
Patients must sign a patient-physician agreement form and comply with the REMS requirements. In particular, female patients who are not pregnant but can become pregnant must comply with the pregnancy testing and contraception requirements, and males must comply with contraception requirements.
Pharmacies must be certified with the Pomalyst REMS Program, must only dispense the drug to patients who are authorized to receive it, and must comply with REMS requirements. Both lenalidomide and thalidomide have similar REMS.
Pomalidomide is marketed by Celgene, which is based in Summit, New Jersey. ![]()
FDA grants drug accelerated approval for relapsed/refractory MM

The FDA has announced accelerated approval of carfilzomib (Kyprolis) as treatment for relapsed or refractory multiple myeloma (MM).
The drug is indicated for MM patients who have received at least 2 prior therapies, including bortezomib and an immunomodulatory agent, and have demonstrated disease progression on or within 60 days of completing their last treatment.
Carfilzomib was approved under the FDA’s accelerated approval program, which allows the agency to approve a drug based on data suggesting a clinical benefit. The drug’s maker is required to submit additional information after the approval to confirm that benefit.
Carfilzomib’s approval was based on efficacy data from a trial of 266 patients and safety data from 526 patients who received the drug.
The 266 patients had relapsed MM and had received at least 2 prior therapies, including bortezomib and an immunomodulatory agent (either thalidomide or lenalidomide). Patients received carfilzomib intravenously over a period of 2 to 10 minutes on 2 consecutive days a week for 3 weeks, followed by a 12-day rest period.
Patients received 20 mg/m2 at each dose in cycle 1 and 27 mg/m2 in subsequent cycles. They continued to receive treatment until their disease progressed, they developed unacceptable toxicity, or they completed 12 cycles.
Following treatment, the overall response rate was 22.9%. One patient achieved a complete response, 13 had very good partial responses, and 47 achieved partial responses. The median response duration was 7.8 months.
Researchers also evaluated carfilzomib’s safety in 526 patients with relapsed MM. Patients received a median of 4 treatment cycles and a median cumulative carfilzomib dose of 993.4 mg.
The most common adverse reactions—with an incidence of 30% or greater—were fatigue, anemia, nausea, thrombocytopenia, dyspnea, diarrhea, and pyrexia.
In addition, 45% of patients experienced serious adverse reactions. The most common were pneumonia, acute renal failure, pyrexia, and congestive heart failure.
Seven percent of patients (n=37) died on study. The most common causes of death, other than underlying disease, were cardiac (n=5), end-organ failure (n=4), and infection (n=4).
Carfilzomib will be marketed as Kyprolis by Onyx Pharmaceuticals. As a condition of the drug’s accelerated approval, Onyx is required to submit the complete analysis of an ongoing phase 3 trial comparing lenalidomide plus low-dose dexamethasone to lenalidomide, low-dose dexamethasone, and carfilzomib.
For more information on carfilzomib, consult the FDA website. ![]()

The FDA has announced accelerated approval of carfilzomib (Kyprolis) as treatment for relapsed or refractory multiple myeloma (MM).
The drug is indicated for MM patients who have received at least 2 prior therapies, including bortezomib and an immunomodulatory agent, and have demonstrated disease progression on or within 60 days of completing their last treatment.
Carfilzomib was approved under the FDA’s accelerated approval program, which allows the agency to approve a drug based on data suggesting a clinical benefit. The drug’s maker is required to submit additional information after the approval to confirm that benefit.
Carfilzomib’s approval was based on efficacy data from a trial of 266 patients and safety data from 526 patients who received the drug.
The 266 patients had relapsed MM and had received at least 2 prior therapies, including bortezomib and an immunomodulatory agent (either thalidomide or lenalidomide). Patients received carfilzomib intravenously over a period of 2 to 10 minutes on 2 consecutive days a week for 3 weeks, followed by a 12-day rest period.
Patients received 20 mg/m2 at each dose in cycle 1 and 27 mg/m2 in subsequent cycles. They continued to receive treatment until their disease progressed, they developed unacceptable toxicity, or they completed 12 cycles.
Following treatment, the overall response rate was 22.9%. One patient achieved a complete response, 13 had very good partial responses, and 47 achieved partial responses. The median response duration was 7.8 months.
Researchers also evaluated carfilzomib’s safety in 526 patients with relapsed MM. Patients received a median of 4 treatment cycles and a median cumulative carfilzomib dose of 993.4 mg.
The most common adverse reactions—with an incidence of 30% or greater—were fatigue, anemia, nausea, thrombocytopenia, dyspnea, diarrhea, and pyrexia.
In addition, 45% of patients experienced serious adverse reactions. The most common were pneumonia, acute renal failure, pyrexia, and congestive heart failure.
Seven percent of patients (n=37) died on study. The most common causes of death, other than underlying disease, were cardiac (n=5), end-organ failure (n=4), and infection (n=4).
Carfilzomib will be marketed as Kyprolis by Onyx Pharmaceuticals. As a condition of the drug’s accelerated approval, Onyx is required to submit the complete analysis of an ongoing phase 3 trial comparing lenalidomide plus low-dose dexamethasone to lenalidomide, low-dose dexamethasone, and carfilzomib.
For more information on carfilzomib, consult the FDA website. ![]()

The FDA has announced accelerated approval of carfilzomib (Kyprolis) as treatment for relapsed or refractory multiple myeloma (MM).
The drug is indicated for MM patients who have received at least 2 prior therapies, including bortezomib and an immunomodulatory agent, and have demonstrated disease progression on or within 60 days of completing their last treatment.
Carfilzomib was approved under the FDA’s accelerated approval program, which allows the agency to approve a drug based on data suggesting a clinical benefit. The drug’s maker is required to submit additional information after the approval to confirm that benefit.
Carfilzomib’s approval was based on efficacy data from a trial of 266 patients and safety data from 526 patients who received the drug.
The 266 patients had relapsed MM and had received at least 2 prior therapies, including bortezomib and an immunomodulatory agent (either thalidomide or lenalidomide). Patients received carfilzomib intravenously over a period of 2 to 10 minutes on 2 consecutive days a week for 3 weeks, followed by a 12-day rest period.
Patients received 20 mg/m2 at each dose in cycle 1 and 27 mg/m2 in subsequent cycles. They continued to receive treatment until their disease progressed, they developed unacceptable toxicity, or they completed 12 cycles.
Following treatment, the overall response rate was 22.9%. One patient achieved a complete response, 13 had very good partial responses, and 47 achieved partial responses. The median response duration was 7.8 months.
Researchers also evaluated carfilzomib’s safety in 526 patients with relapsed MM. Patients received a median of 4 treatment cycles and a median cumulative carfilzomib dose of 993.4 mg.
The most common adverse reactions—with an incidence of 30% or greater—were fatigue, anemia, nausea, thrombocytopenia, dyspnea, diarrhea, and pyrexia.
In addition, 45% of patients experienced serious adverse reactions. The most common were pneumonia, acute renal failure, pyrexia, and congestive heart failure.
Seven percent of patients (n=37) died on study. The most common causes of death, other than underlying disease, were cardiac (n=5), end-organ failure (n=4), and infection (n=4).
Carfilzomib will be marketed as Kyprolis by Onyx Pharmaceuticals. As a condition of the drug’s accelerated approval, Onyx is required to submit the complete analysis of an ongoing phase 3 trial comparing lenalidomide plus low-dose dexamethasone to lenalidomide, low-dose dexamethasone, and carfilzomib.
For more information on carfilzomib, consult the FDA website. ![]()
Pomalidomide and loDex achieve impressive results in MM

Convention Center, site of
the 53rd ASH Annual Meeting
Photo courtesy of ASH
SAN DIEGO—Impressive results have been achieved with pomalidomide and low-dose dexamethasone (loDex) in relapsed and refractory patients with multiple myeloma (MM), according to study investigators.
The combination achieved a 34% response rate, including a complete response of 1% and very good partial response of 10% in this population of MM patients refractory to lenalidomide, bortezomib, or both.
Paul Richardson, MD, of Dana-Farber Cancer Institute, reported the results of the phase 1/2 study, known as MM-002, at the 53rd ASH Annual Meeting (abstract 634).
The phase 1 portion of the trial had been reported previously. It established a maximum-tolerated dose of 4 mg daily of pomalidomide.
Dr Richardson called pomalidomide “arguably the most potent of all the IMiDs.” It has significant antimyeloma activity in vitro, promising activity as a single agent in patients with relapsed/refractory MM and efficacy when combined with loDex in patients previously treated with lenalidomide and/or bortezomib.
Dr Richardson pointed out that the investigators defined relapsed and refractory disease rigorously.
“Patients had to have progressed during or 60 days from last treatment, and this had to be confirmed with source documentation prior to study entry,” he said.
In addition, patients had to have measurable levels of M-paraprotein in serum or urine. They must have had 2 or more prior therapies that included 2 or more cycles of lenalidomide and 2 or more cycles of bortezomib, either separately or in combination.
Two hundred twenty-one patients were randomized, 108 to pomalidomide alone and 113 to pomalidomide plus loDex. The pomalidomide regimen was 4 mg daily for 3 weeks with 1 week off. The loDex consisted of 40 mg per week.
Patients were a median age of 63 years. Seventy-eight percent were refractory to lenalidomide, 71% to bortezomib, and 60% to both agents. They had a median of 5 prior therapies, range 2-13.
One hundred ninety-one patients were evaluable for response. They had a median number of 5 cycles of therapy (range, 1-17) and a median duration of 5 months of treatment.
The combination arm had a 34% overall response rate, including partial response or better, compared to 13% in the pomalidomide-alone arm.
Forty-five percent in the combination arm achieved a minor response, compared to 29% in the pomalidomide-alone arm.
And 81% of the patients overall achieved stable disease or better. Responses were rapid and appeared durable, Dr Richardson said.
“What was particularly reassuring and encouraging as well,” he said, “is to see a very similar rate of response for those patients who were refractory to lenalidomide and bortezomib together.” The overall response rate in those patients was 30%.
The median progression-free survival for patients on the combination was 4.7 months. For those on pomalidomide alone, it was 2.7 months.
And the median overall survival on the combination arm was 16.9 months. For those on pomalidomide alone, it was 14 months.
Patients who were refractory to both lenalidomide and bortezomib achieved progression-free survival approaching 4 months with the drug combination and 2 months for pomalidomide alone.
Neutropenia was the dominant grade 3-4 hematologic side effect, followed by thrombocytopenia and anemia. Dose reduction was required in a minority of patients.
The nonhematologic side effects were quite uncommon and included pneumonia and fatigue. No grade 3-4 peripheral neuropathy was reported.
The rate of thromboembolism was less than 5% in either arm. Anticoagulation with low-dose aspirin was required, however.
The investigators concluded that pomalidomide and loDex are synergistic in terms of response rate, and they were impressed that the consistent and durable responses occurred regardless of prior therapy or refractoriness.
On this basis, pomalidomide plus loDex is being investigated in phase 3 trials and as part of combination treatments, including bortezomib. ![]()

Convention Center, site of
the 53rd ASH Annual Meeting
Photo courtesy of ASH
SAN DIEGO—Impressive results have been achieved with pomalidomide and low-dose dexamethasone (loDex) in relapsed and refractory patients with multiple myeloma (MM), according to study investigators.
The combination achieved a 34% response rate, including a complete response of 1% and very good partial response of 10% in this population of MM patients refractory to lenalidomide, bortezomib, or both.
Paul Richardson, MD, of Dana-Farber Cancer Institute, reported the results of the phase 1/2 study, known as MM-002, at the 53rd ASH Annual Meeting (abstract 634).
The phase 1 portion of the trial had been reported previously. It established a maximum-tolerated dose of 4 mg daily of pomalidomide.
Dr Richardson called pomalidomide “arguably the most potent of all the IMiDs.” It has significant antimyeloma activity in vitro, promising activity as a single agent in patients with relapsed/refractory MM and efficacy when combined with loDex in patients previously treated with lenalidomide and/or bortezomib.
Dr Richardson pointed out that the investigators defined relapsed and refractory disease rigorously.
“Patients had to have progressed during or 60 days from last treatment, and this had to be confirmed with source documentation prior to study entry,” he said.
In addition, patients had to have measurable levels of M-paraprotein in serum or urine. They must have had 2 or more prior therapies that included 2 or more cycles of lenalidomide and 2 or more cycles of bortezomib, either separately or in combination.
Two hundred twenty-one patients were randomized, 108 to pomalidomide alone and 113 to pomalidomide plus loDex. The pomalidomide regimen was 4 mg daily for 3 weeks with 1 week off. The loDex consisted of 40 mg per week.
Patients were a median age of 63 years. Seventy-eight percent were refractory to lenalidomide, 71% to bortezomib, and 60% to both agents. They had a median of 5 prior therapies, range 2-13.
One hundred ninety-one patients were evaluable for response. They had a median number of 5 cycles of therapy (range, 1-17) and a median duration of 5 months of treatment.
The combination arm had a 34% overall response rate, including partial response or better, compared to 13% in the pomalidomide-alone arm.
Forty-five percent in the combination arm achieved a minor response, compared to 29% in the pomalidomide-alone arm.
And 81% of the patients overall achieved stable disease or better. Responses were rapid and appeared durable, Dr Richardson said.
“What was particularly reassuring and encouraging as well,” he said, “is to see a very similar rate of response for those patients who were refractory to lenalidomide and bortezomib together.” The overall response rate in those patients was 30%.
The median progression-free survival for patients on the combination was 4.7 months. For those on pomalidomide alone, it was 2.7 months.
And the median overall survival on the combination arm was 16.9 months. For those on pomalidomide alone, it was 14 months.
Patients who were refractory to both lenalidomide and bortezomib achieved progression-free survival approaching 4 months with the drug combination and 2 months for pomalidomide alone.
Neutropenia was the dominant grade 3-4 hematologic side effect, followed by thrombocytopenia and anemia. Dose reduction was required in a minority of patients.
The nonhematologic side effects were quite uncommon and included pneumonia and fatigue. No grade 3-4 peripheral neuropathy was reported.
The rate of thromboembolism was less than 5% in either arm. Anticoagulation with low-dose aspirin was required, however.
The investigators concluded that pomalidomide and loDex are synergistic in terms of response rate, and they were impressed that the consistent and durable responses occurred regardless of prior therapy or refractoriness.
On this basis, pomalidomide plus loDex is being investigated in phase 3 trials and as part of combination treatments, including bortezomib. ![]()

Convention Center, site of
the 53rd ASH Annual Meeting
Photo courtesy of ASH
SAN DIEGO—Impressive results have been achieved with pomalidomide and low-dose dexamethasone (loDex) in relapsed and refractory patients with multiple myeloma (MM), according to study investigators.
The combination achieved a 34% response rate, including a complete response of 1% and very good partial response of 10% in this population of MM patients refractory to lenalidomide, bortezomib, or both.
Paul Richardson, MD, of Dana-Farber Cancer Institute, reported the results of the phase 1/2 study, known as MM-002, at the 53rd ASH Annual Meeting (abstract 634).
The phase 1 portion of the trial had been reported previously. It established a maximum-tolerated dose of 4 mg daily of pomalidomide.
Dr Richardson called pomalidomide “arguably the most potent of all the IMiDs.” It has significant antimyeloma activity in vitro, promising activity as a single agent in patients with relapsed/refractory MM and efficacy when combined with loDex in patients previously treated with lenalidomide and/or bortezomib.
Dr Richardson pointed out that the investigators defined relapsed and refractory disease rigorously.
“Patients had to have progressed during or 60 days from last treatment, and this had to be confirmed with source documentation prior to study entry,” he said.
In addition, patients had to have measurable levels of M-paraprotein in serum or urine. They must have had 2 or more prior therapies that included 2 or more cycles of lenalidomide and 2 or more cycles of bortezomib, either separately or in combination.
Two hundred twenty-one patients were randomized, 108 to pomalidomide alone and 113 to pomalidomide plus loDex. The pomalidomide regimen was 4 mg daily for 3 weeks with 1 week off. The loDex consisted of 40 mg per week.
Patients were a median age of 63 years. Seventy-eight percent were refractory to lenalidomide, 71% to bortezomib, and 60% to both agents. They had a median of 5 prior therapies, range 2-13.
One hundred ninety-one patients were evaluable for response. They had a median number of 5 cycles of therapy (range, 1-17) and a median duration of 5 months of treatment.
The combination arm had a 34% overall response rate, including partial response or better, compared to 13% in the pomalidomide-alone arm.
Forty-five percent in the combination arm achieved a minor response, compared to 29% in the pomalidomide-alone arm.
And 81% of the patients overall achieved stable disease or better. Responses were rapid and appeared durable, Dr Richardson said.
“What was particularly reassuring and encouraging as well,” he said, “is to see a very similar rate of response for those patients who were refractory to lenalidomide and bortezomib together.” The overall response rate in those patients was 30%.
The median progression-free survival for patients on the combination was 4.7 months. For those on pomalidomide alone, it was 2.7 months.
And the median overall survival on the combination arm was 16.9 months. For those on pomalidomide alone, it was 14 months.
Patients who were refractory to both lenalidomide and bortezomib achieved progression-free survival approaching 4 months with the drug combination and 2 months for pomalidomide alone.
Neutropenia was the dominant grade 3-4 hematologic side effect, followed by thrombocytopenia and anemia. Dose reduction was required in a minority of patients.
The nonhematologic side effects were quite uncommon and included pneumonia and fatigue. No grade 3-4 peripheral neuropathy was reported.
The rate of thromboembolism was less than 5% in either arm. Anticoagulation with low-dose aspirin was required, however.
The investigators concluded that pomalidomide and loDex are synergistic in terms of response rate, and they were impressed that the consistent and durable responses occurred regardless of prior therapy or refractoriness.
On this basis, pomalidomide plus loDex is being investigated in phase 3 trials and as part of combination treatments, including bortezomib. ![]()
CTAs are promising therapeutic targets in MM
New York, NY—A new study suggests that cancer testis antigens (CTAs) should be therapeutically targeted in patients with multiple myeloma (MM).
The study revealed that CTAs are frequently expressed in newly diagnosed MM patients, the presence of certain CTAs can help predict poor survival, and MM patients experience spontaneous antibody responses to CTAs. Adam Cohen, MD, of Fox Chase Cancer Center in Philadelphia, presented this research at Lymphoma & Myeloma 2009, where it was deemed “the best myeloma abstract.”
Dr Cohen and his colleagues enrolled in their study 67 newly diagnosed MM patients. Patients received an induction regimen consisting of thalidomide, doxorubicin, and dexamethasone, and 54 patients went on to receive autologous stem cell transplant.
The researchers assessed CTA expression in cryopreserved pretreatment bone marrow plasma cells. Seventy-seven percent of patients had at least 1 CTA. MAGE-A3 was present in 52% of patients, SSX1 in 40%, CT7 in 29%, CT10 in 25%, NY-ESO1 in 21%, and SSX5 was expressed in 17% of patients. Twenty-nine percent of patients had 3 or more CTAs.
“So the main question was, what was the prognostic significance of these findings?” Dr Cohen said. “We looked at overall survival on the basis of the presence or absence of each of these antigens or based on the absolute number of antigens that were expressed. What we found were 2 antigens that really seemed to stand out, in terms of having prognostic significance.”
Patients who expressed MAGE-A3 or NY-ESO1 had worse overall survival (OS) than patients who expressed other CTAs. OS was a median of 66 months in patients with MAGE-A3 and 65 months in patients with NY-ESO1, while OS was not reached in the other patients.
The poor OS observed in patients with MAGE-A3 and NY-ESO1 was independent of disease stage, cytogenetic abnormalities, and response to induction therapy.
Dr Cohen and his colleagues then assessed pre- and post-treatment sera for antibody responses. Forty-six patients had sera available. Six patients had antibody responses to NY-ESO1. Of these patients, 2 also demonstrated responses to CT7, 1 had response to CT10, and 1 had response to SSX4.
“[A]ll these patients had immunity to NY-ESO1, but in 2 patients, number 30 and 54, there actually was no NY-ESO1 expression in their bone marrow,” Dr Cohen said. “[B]oth of these had extramedullary disease, and so the suggestion was that there may be an additional source of the NY-ESO1 antigen.”
This theory was supported by the fact that these 2 patients had soft tissue plasmacytomas. And the presence of NY-ESO1 antibody was significantly associated with soft tissue involvement, as 67% of NY-ESO1 antibody-positive patients had soft tissue plasmacytomas.
In addition, antibody response against NY-ESO1 was associated with poor OS. NY-ESO1 antibody-positive patients had an OS of 21 months, while OS was not reached in NY-ESO1 antibody-negative patients.
Dr Cohen presented these data at Lymphoma & Myeloma 2009, which took place October 22-24. ![]()
New York, NY—A new study suggests that cancer testis antigens (CTAs) should be therapeutically targeted in patients with multiple myeloma (MM).
The study revealed that CTAs are frequently expressed in newly diagnosed MM patients, the presence of certain CTAs can help predict poor survival, and MM patients experience spontaneous antibody responses to CTAs. Adam Cohen, MD, of Fox Chase Cancer Center in Philadelphia, presented this research at Lymphoma & Myeloma 2009, where it was deemed “the best myeloma abstract.”
Dr Cohen and his colleagues enrolled in their study 67 newly diagnosed MM patients. Patients received an induction regimen consisting of thalidomide, doxorubicin, and dexamethasone, and 54 patients went on to receive autologous stem cell transplant.
The researchers assessed CTA expression in cryopreserved pretreatment bone marrow plasma cells. Seventy-seven percent of patients had at least 1 CTA. MAGE-A3 was present in 52% of patients, SSX1 in 40%, CT7 in 29%, CT10 in 25%, NY-ESO1 in 21%, and SSX5 was expressed in 17% of patients. Twenty-nine percent of patients had 3 or more CTAs.
“So the main question was, what was the prognostic significance of these findings?” Dr Cohen said. “We looked at overall survival on the basis of the presence or absence of each of these antigens or based on the absolute number of antigens that were expressed. What we found were 2 antigens that really seemed to stand out, in terms of having prognostic significance.”
Patients who expressed MAGE-A3 or NY-ESO1 had worse overall survival (OS) than patients who expressed other CTAs. OS was a median of 66 months in patients with MAGE-A3 and 65 months in patients with NY-ESO1, while OS was not reached in the other patients.
The poor OS observed in patients with MAGE-A3 and NY-ESO1 was independent of disease stage, cytogenetic abnormalities, and response to induction therapy.
Dr Cohen and his colleagues then assessed pre- and post-treatment sera for antibody responses. Forty-six patients had sera available. Six patients had antibody responses to NY-ESO1. Of these patients, 2 also demonstrated responses to CT7, 1 had response to CT10, and 1 had response to SSX4.
“[A]ll these patients had immunity to NY-ESO1, but in 2 patients, number 30 and 54, there actually was no NY-ESO1 expression in their bone marrow,” Dr Cohen said. “[B]oth of these had extramedullary disease, and so the suggestion was that there may be an additional source of the NY-ESO1 antigen.”
This theory was supported by the fact that these 2 patients had soft tissue plasmacytomas. And the presence of NY-ESO1 antibody was significantly associated with soft tissue involvement, as 67% of NY-ESO1 antibody-positive patients had soft tissue plasmacytomas.
In addition, antibody response against NY-ESO1 was associated with poor OS. NY-ESO1 antibody-positive patients had an OS of 21 months, while OS was not reached in NY-ESO1 antibody-negative patients.
Dr Cohen presented these data at Lymphoma & Myeloma 2009, which took place October 22-24. ![]()
New York, NY—A new study suggests that cancer testis antigens (CTAs) should be therapeutically targeted in patients with multiple myeloma (MM).
The study revealed that CTAs are frequently expressed in newly diagnosed MM patients, the presence of certain CTAs can help predict poor survival, and MM patients experience spontaneous antibody responses to CTAs. Adam Cohen, MD, of Fox Chase Cancer Center in Philadelphia, presented this research at Lymphoma & Myeloma 2009, where it was deemed “the best myeloma abstract.”
Dr Cohen and his colleagues enrolled in their study 67 newly diagnosed MM patients. Patients received an induction regimen consisting of thalidomide, doxorubicin, and dexamethasone, and 54 patients went on to receive autologous stem cell transplant.
The researchers assessed CTA expression in cryopreserved pretreatment bone marrow plasma cells. Seventy-seven percent of patients had at least 1 CTA. MAGE-A3 was present in 52% of patients, SSX1 in 40%, CT7 in 29%, CT10 in 25%, NY-ESO1 in 21%, and SSX5 was expressed in 17% of patients. Twenty-nine percent of patients had 3 or more CTAs.
“So the main question was, what was the prognostic significance of these findings?” Dr Cohen said. “We looked at overall survival on the basis of the presence or absence of each of these antigens or based on the absolute number of antigens that were expressed. What we found were 2 antigens that really seemed to stand out, in terms of having prognostic significance.”
Patients who expressed MAGE-A3 or NY-ESO1 had worse overall survival (OS) than patients who expressed other CTAs. OS was a median of 66 months in patients with MAGE-A3 and 65 months in patients with NY-ESO1, while OS was not reached in the other patients.
The poor OS observed in patients with MAGE-A3 and NY-ESO1 was independent of disease stage, cytogenetic abnormalities, and response to induction therapy.
Dr Cohen and his colleagues then assessed pre- and post-treatment sera for antibody responses. Forty-six patients had sera available. Six patients had antibody responses to NY-ESO1. Of these patients, 2 also demonstrated responses to CT7, 1 had response to CT10, and 1 had response to SSX4.
“[A]ll these patients had immunity to NY-ESO1, but in 2 patients, number 30 and 54, there actually was no NY-ESO1 expression in their bone marrow,” Dr Cohen said. “[B]oth of these had extramedullary disease, and so the suggestion was that there may be an additional source of the NY-ESO1 antigen.”
This theory was supported by the fact that these 2 patients had soft tissue plasmacytomas. And the presence of NY-ESO1 antibody was significantly associated with soft tissue involvement, as 67% of NY-ESO1 antibody-positive patients had soft tissue plasmacytomas.
In addition, antibody response against NY-ESO1 was associated with poor OS. NY-ESO1 antibody-positive patients had an OS of 21 months, while OS was not reached in NY-ESO1 antibody-negative patients.
Dr Cohen presented these data at Lymphoma & Myeloma 2009, which took place October 22-24. ![]()