User login
For MD-IQ use only
Time for mental stress testing in ANOCA?
, new results show.
Further analysis in the small study suggested that mental stress–induced myocardial ischemia (MSIMI) was not statistically related to coronary microvascular dysfunction (CMD).
“Since the findings do not support a correlation between MSIMI and CMD, which has been a widely accepted mechanistic explanation of ANOCA, routine mental stress testing in patients with ANOCA seems necessary,” researchers led by Qingshan Geng, MD, PhD, of Shenzhen People’s Hospital, Guangdong, China, conclude in a report published online in the Journal of the American College of Cardiology.
Dr. Geng said in an interview that the use of virtual reality devices to administer mental stress tests “ensures standardized experimental procedures, with each participant receiving an objectively equivalent level of stress load.
“The immersive experience provided by VR lowers the environmental requirements for the test,” he noted. “Furthermore, the application of VR reduces the workload of personnel responsible for inducing mental stress, simplifying the experimental process.”
The team also developed a mobile app that enables remote monitoring of participants’ visual experiences during PET/CT scans and facilitates communication, he added.
Mental stress testing and meds?
Both ANOCA and MSIMI in patients with coronary artery disease disproportionately affect women and are associated with poor cardiovascular prognosis, the researchers write.
“However, the role of MSIMI and the exact influence of mental stress in ANOCA have not previously been studied,” they point out.
For this investigation, 84 women with ANOCA and 42 age-matched controls underwent three mental stress challenges delivered via VR.
Tests included mental arithmetic, making a public speech describing a recent emotionally upsetting event, and a task-modified Stroop test, in which participants were asked to say the color in which the word appears, not the color that the word names. For example, if the word “yellow” appears in blue type, blue would be the correct answer.
An adenosine stress test was given 5-8 minutes after the mental stress challenges started, and cardiac PET/CT was used to examine myocardial blood flow and perfusion.
The investigators report that women with ANOCA had a much higher rate of MSIMI (42.9%), compared with control participants (one patient; 2.4%). They also had a higher proportion of coronary microvascular dysfunction (CMD; 24.6% vs. 8.6%), but the occurrence of MSIMI and CMD was not related, the authors note.
Consistent with previous studies, “we observed that CMD is more prevalent in ANOCA women than the age-matched healthy individuals. MSIMI rate, however, was notably higher than the rate of CMD in our female ANOCA population,” they write. “The lack of a significant association between MSIMI and CMD indicates the mechanisms of MSIMI cannot be well explained by the adenosine-induced CMD.”
Dr. Geng suggested that ANOCA patients may benefit from treatment with escitalopram.
“Compelling evidence” from the REMIT randomized, placebo-controlled trial validates the efficacy of the drug as an MSIMI treatment, he said.
Sample size too small?
Asked for comment on the findings, Viola Vaccarino, MD, PhD, Wilton Looney Distinguished Professor of Cardiovascular Research at Emory University’s Rollins School of Public Health and a professor in the university’s School of Medicine, Atlanta, said she disagreed with several aspects of this study and the investigators’ conclusions.
Although the study suggests that MSIMI is prevalent among women with ANOCA, “the sample size was too small to make any definite conclusions,” she said in an interview.
“In fact,” she said, “I do not agree with the authors’ conclusions that MSIMI and CMD were not related, based on the data presented, even though the P value was not significant.”
In addition, more research is needed before screening can be recommended, she said. “The effectiveness of this testing modality in this population should be demonstrated first.”
Furthermore, she added, “an established treatment for MSIMI has yet to be tested in large, controlled trials, which limits the potential clinical benefit that may result from this [screening] test.”
For now, to ameliorate potential MSIMI in women with ANOCA, Dr. Vaccarino recommends behavioral modalities or stress-reduction management techniques, including biofeedback, meditation, breathing exercises, and “just plain regular physical activity,” rather than the use of psychotropic medications.
Dr. Vaccarino’s team has a study underway that builds on earlier work involving more than 900 participants, which showed that MSIMI was significantly associated with an increased risk of cardiovascular death or nonfatal myocardial infarction (hazard ratio, 2.5).
The ongoing study, which investigates the link between emotional stress and heart disease in men and women, should be completed in about 3 years, she said.
Microvascular disease or spasm?
Leslie Cho, MD, chair of the American College of Cardiology’s Cardiovascular Disease in Women Committee, director of the Cleveland Clinic’s Women’s Cardiovascular Center, and professor of medicine at Cleveland Clinic Lerner School of Medicine and Case Western Reserve Medical School, commented on the mental stress–heart connection and mental stress testing for this article.
A “very big flaw” of the JACC study, she said, is that although PET testing can detect microvascular disease, it cannot detect microvascular spasm.
PET can show the coronary flow reserve, “which is a nice way to assess microvascular dysfunction,” she acknowledged, “but it really can’t tell microvascular spasm, because adenosine works in a different pathway than acetylcholine – and I think it’s important for people to have the right diagnosis.
“We do physiologic testing to distinguish the two conditions,” she noted. “We do the gold standard, which is the cath lab.”
“The problem with women with chest pain for years is that they get a stress test, they get a cath, and everything’s normal. Then they get blown off as anxious or whatever.”
Clinicians should conduct the gold standard workup – provocative physiologic testing – for these women who continue to have chest pain when results of other tests are negative, she said. “The test used to be very cumbersome, but today, we have systems that make it super easy to use and to distinguish microvascular disease and microvascular spasm.”
Importantly, she added, physiologic testing should be performed when women are off therapy – something that doesn’t always happen in the clinic.
Regarding treatment, she added, “if you’re having emotional stress, the answer is not another medicine. The answer is cognitive-behavioral therapy or another behavioral intervention to overcome anxiety.”
Tune in and advocate
What can clinicians do for women with ANOCA after testing reveals no significant coronary artery disease or microvascular spasms?
“Very often, it’s a matter of the doctor listening and responding to the patient,” Johanna Contreras, MD, a cardiologist at Mount Sinai Hospital, New York, said in an intereview.
In her practice, Dr. Contreras sees highly stressed women on a daily basis. Many of her patients are women from diverse racial/ethnic groups, often of lower socioeconomic status, who are heads of households, work more than one job, and experience other major stressors.
“My message to clinicians is: don’t give up on a woman just because you looked at the arteries and couldn’t find anything specific. If she keeps coming back with the same symptom, it’s important to address it,” she said. “Maybe it isn’t the symptom. Maybe she needs to talk about her situation, about the physiological and psychosocial factors contributing to the symptom that a test alone won’t reveal.”
Regarding cardiovascular spasms that are identified through physiologic testing, she said, “I don’t know that medications such as SSRIs [selective serotonin reuptake inhibitors] are going to change anything. But many things can be changed by listening or helping the patient to stop and think about her mental health.”
Following up with a referral to a therapist can help, she said; “Take away the mental health stigma by telling the patient that the referral is simply to help her cope.”
Dr. Contreras urges clinicians to be advocates for such patients. If an insurance company says it will cover only three therapy sessions, “tell them that three appointments are not enough” to address multiple issues.
“If we invest money in helping patients identify and cope with these issues, we are likely to get better long-term outcomes, rather than having that woman come into the emergency department with chest pain over and over and doing 20,000 tests that are going to show exactly the same thing,” Dr. Contreras concluded.
Dr. Geng’s study was supported by the High-Level Hospital Construction Project of Guangdong Provincial People’s Hospital, by a grant from Guangdong Provincial Bureau of Traditional Chinese Medicine, and by a grant from Guangdong Medical Science and Technology Research Foundation. The authors, Dr. Vaccarino, Dr. Contreras, and Dr. Cho report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
, new results show.
Further analysis in the small study suggested that mental stress–induced myocardial ischemia (MSIMI) was not statistically related to coronary microvascular dysfunction (CMD).
“Since the findings do not support a correlation between MSIMI and CMD, which has been a widely accepted mechanistic explanation of ANOCA, routine mental stress testing in patients with ANOCA seems necessary,” researchers led by Qingshan Geng, MD, PhD, of Shenzhen People’s Hospital, Guangdong, China, conclude in a report published online in the Journal of the American College of Cardiology.
Dr. Geng said in an interview that the use of virtual reality devices to administer mental stress tests “ensures standardized experimental procedures, with each participant receiving an objectively equivalent level of stress load.
“The immersive experience provided by VR lowers the environmental requirements for the test,” he noted. “Furthermore, the application of VR reduces the workload of personnel responsible for inducing mental stress, simplifying the experimental process.”
The team also developed a mobile app that enables remote monitoring of participants’ visual experiences during PET/CT scans and facilitates communication, he added.
Mental stress testing and meds?
Both ANOCA and MSIMI in patients with coronary artery disease disproportionately affect women and are associated with poor cardiovascular prognosis, the researchers write.
“However, the role of MSIMI and the exact influence of mental stress in ANOCA have not previously been studied,” they point out.
For this investigation, 84 women with ANOCA and 42 age-matched controls underwent three mental stress challenges delivered via VR.
Tests included mental arithmetic, making a public speech describing a recent emotionally upsetting event, and a task-modified Stroop test, in which participants were asked to say the color in which the word appears, not the color that the word names. For example, if the word “yellow” appears in blue type, blue would be the correct answer.
An adenosine stress test was given 5-8 minutes after the mental stress challenges started, and cardiac PET/CT was used to examine myocardial blood flow and perfusion.
The investigators report that women with ANOCA had a much higher rate of MSIMI (42.9%), compared with control participants (one patient; 2.4%). They also had a higher proportion of coronary microvascular dysfunction (CMD; 24.6% vs. 8.6%), but the occurrence of MSIMI and CMD was not related, the authors note.
Consistent with previous studies, “we observed that CMD is more prevalent in ANOCA women than the age-matched healthy individuals. MSIMI rate, however, was notably higher than the rate of CMD in our female ANOCA population,” they write. “The lack of a significant association between MSIMI and CMD indicates the mechanisms of MSIMI cannot be well explained by the adenosine-induced CMD.”
Dr. Geng suggested that ANOCA patients may benefit from treatment with escitalopram.
“Compelling evidence” from the REMIT randomized, placebo-controlled trial validates the efficacy of the drug as an MSIMI treatment, he said.
Sample size too small?
Asked for comment on the findings, Viola Vaccarino, MD, PhD, Wilton Looney Distinguished Professor of Cardiovascular Research at Emory University’s Rollins School of Public Health and a professor in the university’s School of Medicine, Atlanta, said she disagreed with several aspects of this study and the investigators’ conclusions.
Although the study suggests that MSIMI is prevalent among women with ANOCA, “the sample size was too small to make any definite conclusions,” she said in an interview.
“In fact,” she said, “I do not agree with the authors’ conclusions that MSIMI and CMD were not related, based on the data presented, even though the P value was not significant.”
In addition, more research is needed before screening can be recommended, she said. “The effectiveness of this testing modality in this population should be demonstrated first.”
Furthermore, she added, “an established treatment for MSIMI has yet to be tested in large, controlled trials, which limits the potential clinical benefit that may result from this [screening] test.”
For now, to ameliorate potential MSIMI in women with ANOCA, Dr. Vaccarino recommends behavioral modalities or stress-reduction management techniques, including biofeedback, meditation, breathing exercises, and “just plain regular physical activity,” rather than the use of psychotropic medications.
Dr. Vaccarino’s team has a study underway that builds on earlier work involving more than 900 participants, which showed that MSIMI was significantly associated with an increased risk of cardiovascular death or nonfatal myocardial infarction (hazard ratio, 2.5).
The ongoing study, which investigates the link between emotional stress and heart disease in men and women, should be completed in about 3 years, she said.
Microvascular disease or spasm?
Leslie Cho, MD, chair of the American College of Cardiology’s Cardiovascular Disease in Women Committee, director of the Cleveland Clinic’s Women’s Cardiovascular Center, and professor of medicine at Cleveland Clinic Lerner School of Medicine and Case Western Reserve Medical School, commented on the mental stress–heart connection and mental stress testing for this article.
A “very big flaw” of the JACC study, she said, is that although PET testing can detect microvascular disease, it cannot detect microvascular spasm.
PET can show the coronary flow reserve, “which is a nice way to assess microvascular dysfunction,” she acknowledged, “but it really can’t tell microvascular spasm, because adenosine works in a different pathway than acetylcholine – and I think it’s important for people to have the right diagnosis.
“We do physiologic testing to distinguish the two conditions,” she noted. “We do the gold standard, which is the cath lab.”
“The problem with women with chest pain for years is that they get a stress test, they get a cath, and everything’s normal. Then they get blown off as anxious or whatever.”
Clinicians should conduct the gold standard workup – provocative physiologic testing – for these women who continue to have chest pain when results of other tests are negative, she said. “The test used to be very cumbersome, but today, we have systems that make it super easy to use and to distinguish microvascular disease and microvascular spasm.”
Importantly, she added, physiologic testing should be performed when women are off therapy – something that doesn’t always happen in the clinic.
Regarding treatment, she added, “if you’re having emotional stress, the answer is not another medicine. The answer is cognitive-behavioral therapy or another behavioral intervention to overcome anxiety.”
Tune in and advocate
What can clinicians do for women with ANOCA after testing reveals no significant coronary artery disease or microvascular spasms?
“Very often, it’s a matter of the doctor listening and responding to the patient,” Johanna Contreras, MD, a cardiologist at Mount Sinai Hospital, New York, said in an intereview.
In her practice, Dr. Contreras sees highly stressed women on a daily basis. Many of her patients are women from diverse racial/ethnic groups, often of lower socioeconomic status, who are heads of households, work more than one job, and experience other major stressors.
“My message to clinicians is: don’t give up on a woman just because you looked at the arteries and couldn’t find anything specific. If she keeps coming back with the same symptom, it’s important to address it,” she said. “Maybe it isn’t the symptom. Maybe she needs to talk about her situation, about the physiological and psychosocial factors contributing to the symptom that a test alone won’t reveal.”
Regarding cardiovascular spasms that are identified through physiologic testing, she said, “I don’t know that medications such as SSRIs [selective serotonin reuptake inhibitors] are going to change anything. But many things can be changed by listening or helping the patient to stop and think about her mental health.”
Following up with a referral to a therapist can help, she said; “Take away the mental health stigma by telling the patient that the referral is simply to help her cope.”
Dr. Contreras urges clinicians to be advocates for such patients. If an insurance company says it will cover only three therapy sessions, “tell them that three appointments are not enough” to address multiple issues.
“If we invest money in helping patients identify and cope with these issues, we are likely to get better long-term outcomes, rather than having that woman come into the emergency department with chest pain over and over and doing 20,000 tests that are going to show exactly the same thing,” Dr. Contreras concluded.
Dr. Geng’s study was supported by the High-Level Hospital Construction Project of Guangdong Provincial People’s Hospital, by a grant from Guangdong Provincial Bureau of Traditional Chinese Medicine, and by a grant from Guangdong Medical Science and Technology Research Foundation. The authors, Dr. Vaccarino, Dr. Contreras, and Dr. Cho report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
, new results show.
Further analysis in the small study suggested that mental stress–induced myocardial ischemia (MSIMI) was not statistically related to coronary microvascular dysfunction (CMD).
“Since the findings do not support a correlation between MSIMI and CMD, which has been a widely accepted mechanistic explanation of ANOCA, routine mental stress testing in patients with ANOCA seems necessary,” researchers led by Qingshan Geng, MD, PhD, of Shenzhen People’s Hospital, Guangdong, China, conclude in a report published online in the Journal of the American College of Cardiology.
Dr. Geng said in an interview that the use of virtual reality devices to administer mental stress tests “ensures standardized experimental procedures, with each participant receiving an objectively equivalent level of stress load.
“The immersive experience provided by VR lowers the environmental requirements for the test,” he noted. “Furthermore, the application of VR reduces the workload of personnel responsible for inducing mental stress, simplifying the experimental process.”
The team also developed a mobile app that enables remote monitoring of participants’ visual experiences during PET/CT scans and facilitates communication, he added.
Mental stress testing and meds?
Both ANOCA and MSIMI in patients with coronary artery disease disproportionately affect women and are associated with poor cardiovascular prognosis, the researchers write.
“However, the role of MSIMI and the exact influence of mental stress in ANOCA have not previously been studied,” they point out.
For this investigation, 84 women with ANOCA and 42 age-matched controls underwent three mental stress challenges delivered via VR.
Tests included mental arithmetic, making a public speech describing a recent emotionally upsetting event, and a task-modified Stroop test, in which participants were asked to say the color in which the word appears, not the color that the word names. For example, if the word “yellow” appears in blue type, blue would be the correct answer.
An adenosine stress test was given 5-8 minutes after the mental stress challenges started, and cardiac PET/CT was used to examine myocardial blood flow and perfusion.
The investigators report that women with ANOCA had a much higher rate of MSIMI (42.9%), compared with control participants (one patient; 2.4%). They also had a higher proportion of coronary microvascular dysfunction (CMD; 24.6% vs. 8.6%), but the occurrence of MSIMI and CMD was not related, the authors note.
Consistent with previous studies, “we observed that CMD is more prevalent in ANOCA women than the age-matched healthy individuals. MSIMI rate, however, was notably higher than the rate of CMD in our female ANOCA population,” they write. “The lack of a significant association between MSIMI and CMD indicates the mechanisms of MSIMI cannot be well explained by the adenosine-induced CMD.”
Dr. Geng suggested that ANOCA patients may benefit from treatment with escitalopram.
“Compelling evidence” from the REMIT randomized, placebo-controlled trial validates the efficacy of the drug as an MSIMI treatment, he said.
Sample size too small?
Asked for comment on the findings, Viola Vaccarino, MD, PhD, Wilton Looney Distinguished Professor of Cardiovascular Research at Emory University’s Rollins School of Public Health and a professor in the university’s School of Medicine, Atlanta, said she disagreed with several aspects of this study and the investigators’ conclusions.
Although the study suggests that MSIMI is prevalent among women with ANOCA, “the sample size was too small to make any definite conclusions,” she said in an interview.
“In fact,” she said, “I do not agree with the authors’ conclusions that MSIMI and CMD were not related, based on the data presented, even though the P value was not significant.”
In addition, more research is needed before screening can be recommended, she said. “The effectiveness of this testing modality in this population should be demonstrated first.”
Furthermore, she added, “an established treatment for MSIMI has yet to be tested in large, controlled trials, which limits the potential clinical benefit that may result from this [screening] test.”
For now, to ameliorate potential MSIMI in women with ANOCA, Dr. Vaccarino recommends behavioral modalities or stress-reduction management techniques, including biofeedback, meditation, breathing exercises, and “just plain regular physical activity,” rather than the use of psychotropic medications.
Dr. Vaccarino’s team has a study underway that builds on earlier work involving more than 900 participants, which showed that MSIMI was significantly associated with an increased risk of cardiovascular death or nonfatal myocardial infarction (hazard ratio, 2.5).
The ongoing study, which investigates the link between emotional stress and heart disease in men and women, should be completed in about 3 years, she said.
Microvascular disease or spasm?
Leslie Cho, MD, chair of the American College of Cardiology’s Cardiovascular Disease in Women Committee, director of the Cleveland Clinic’s Women’s Cardiovascular Center, and professor of medicine at Cleveland Clinic Lerner School of Medicine and Case Western Reserve Medical School, commented on the mental stress–heart connection and mental stress testing for this article.
A “very big flaw” of the JACC study, she said, is that although PET testing can detect microvascular disease, it cannot detect microvascular spasm.
PET can show the coronary flow reserve, “which is a nice way to assess microvascular dysfunction,” she acknowledged, “but it really can’t tell microvascular spasm, because adenosine works in a different pathway than acetylcholine – and I think it’s important for people to have the right diagnosis.
“We do physiologic testing to distinguish the two conditions,” she noted. “We do the gold standard, which is the cath lab.”
“The problem with women with chest pain for years is that they get a stress test, they get a cath, and everything’s normal. Then they get blown off as anxious or whatever.”
Clinicians should conduct the gold standard workup – provocative physiologic testing – for these women who continue to have chest pain when results of other tests are negative, she said. “The test used to be very cumbersome, but today, we have systems that make it super easy to use and to distinguish microvascular disease and microvascular spasm.”
Importantly, she added, physiologic testing should be performed when women are off therapy – something that doesn’t always happen in the clinic.
Regarding treatment, she added, “if you’re having emotional stress, the answer is not another medicine. The answer is cognitive-behavioral therapy or another behavioral intervention to overcome anxiety.”
Tune in and advocate
What can clinicians do for women with ANOCA after testing reveals no significant coronary artery disease or microvascular spasms?
“Very often, it’s a matter of the doctor listening and responding to the patient,” Johanna Contreras, MD, a cardiologist at Mount Sinai Hospital, New York, said in an intereview.
In her practice, Dr. Contreras sees highly stressed women on a daily basis. Many of her patients are women from diverse racial/ethnic groups, often of lower socioeconomic status, who are heads of households, work more than one job, and experience other major stressors.
“My message to clinicians is: don’t give up on a woman just because you looked at the arteries and couldn’t find anything specific. If she keeps coming back with the same symptom, it’s important to address it,” she said. “Maybe it isn’t the symptom. Maybe she needs to talk about her situation, about the physiological and psychosocial factors contributing to the symptom that a test alone won’t reveal.”
Regarding cardiovascular spasms that are identified through physiologic testing, she said, “I don’t know that medications such as SSRIs [selective serotonin reuptake inhibitors] are going to change anything. But many things can be changed by listening or helping the patient to stop and think about her mental health.”
Following up with a referral to a therapist can help, she said; “Take away the mental health stigma by telling the patient that the referral is simply to help her cope.”
Dr. Contreras urges clinicians to be advocates for such patients. If an insurance company says it will cover only three therapy sessions, “tell them that three appointments are not enough” to address multiple issues.
“If we invest money in helping patients identify and cope with these issues, we are likely to get better long-term outcomes, rather than having that woman come into the emergency department with chest pain over and over and doing 20,000 tests that are going to show exactly the same thing,” Dr. Contreras concluded.
Dr. Geng’s study was supported by the High-Level Hospital Construction Project of Guangdong Provincial People’s Hospital, by a grant from Guangdong Provincial Bureau of Traditional Chinese Medicine, and by a grant from Guangdong Medical Science and Technology Research Foundation. The authors, Dr. Vaccarino, Dr. Contreras, and Dr. Cho report no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In defense of artificial sweeteners
More than 140 million Americans use artificial sweeteners, a habit driven by the irrefutable fact that excess sugar is harmful. But I’m continually amazed by alarmist headlines on the topic.
In May, the World Health Organization (WHO) released a report to support its “conditional recommendation” against the use of non-sugar sweeteners (NSS) for weight control. Despite the WHO’s goal “to provide evidence-informed guidance,” the report includes the disclaimer that “The recommendation is based on evidence of low certainty.”
Low certainty is an accurate descriptor for the findings of many of the 280-plus studies in the report. That the guidance does not apply to patients with diabetes was easily lost in the repeated mentions of the perceived dangers of these sugar alternatives.
The review included various table-top and beverage sweeteners, including acesulfame K, aspartame, saccharin, sucralose, stevia, and stevia derivatives. Low-calorie sugars and sugar alcohols such as erythritol were excluded.
The WHO looked at long- and short-term trials, randomized controlled trials (RCTs), prospective studies, and case-control studies measuring a wide range of endpoints, from dental caries to cancer. The report highlighted that some findings cannot be attributed directly to NSS use but may simply be due to their substitution for sugar. Differences in outcomes due to sex, ethnicity, and body weight status could not be assessed either. And the WHO conceded the possibility of reverse causation in observational studies wherein higher-risk individuals may consume more NSS.
Nonnutritive sweeteners are given little credit for weight loss. “A significant difference in body weight and BMI was only observed in trials that reported a reduction in energy intake ... rather than primarily by an inherent property of NSS that can modulate body weight (independently of energy intake),” the report reads. But isn’t the desired effect of using an artificial sweetener instead of table sugar that you lower your calorie intake?
The WHO noted that weight loss was not sustained – a finding in nearly every weight loss trial in history and something more attributable to human nature than the sweetener one chooses.
The document outlines that meta-analyses of prospective cohort studies show that higher intakes of NSS were associated with an increased risk for type 2 diabetes and elevated fasting glucose, while meta-analyses of randomized trials suggest no significant effect on “biomarkers used in the assessment and diagnosis of diabetes and insulin resistance, including fasting glucose, fasting insulin and hemoglobin A1c.”
Similar disparities are noted with cardiovascular risk. Prospective trials suggest an increased risk for CVD, including stroke and its precursor, hypertension; but again, the RCT data found no evidence to suggest a significant effect “on biomarkers used in the assessment and diagnosis of CVDs, including blood pressure, low-density lipoprotein cholesterol and other blood lipids.”
Splenda and stevia under fire
Predictably, some in the nonnutritive sweetener industry are incensed.
Ted Gelov, CEO of Heartland Food Products Group, maker of Splenda, responded in a press release, “Every few years now it seems I have to come to you and clarify misleading headlines ... Suggesting that sweeteners like Splenda cannot have long-term benefits is a disservice to healthcare providers, their patients, and all consumers.”
Splenda has been on the U.S. market since 1999, and Mr. Gelov reportedly uses three to eight packets daily in his coffee and tea.
I reached out to Heartland and they sent me an eight-page document consisting of over 50 statements, summaries, and clinical trials supporting the safety of artificial sweeteners, including sucralose, an ingredient in Splenda. In 2016, Mr. Gelov rebutted claims that sucralose was linked to cancer in Swiss male mice. These “dramatized headlines are based on one flawed study by an isolated Italian research laboratory, the Ramazzini Institute,” Mr. Gelov wrote.
Another recent headline was about the DNA-damaging effects of sucralose-6-acetate (S6A) seen in an in vitro study published in the Journal of Toxicology and Environmental Health. According to the authors, commercial sucralose samples contain up to 0.67% S6A, a manufacturing impurity.
Despite many reports linking this study to Splenda, Heartland wrote that “Splenda and its ingredients were never studied or tested in this research. We, and our suppliers, rigorously and routinely test and monitor for any impurities in our products. We can confirm that S6A is not present in Splenda Brand sucralose down to the lowest detection limit possible, which is .001% sensitivity level.”
F. Perry Wilson, MD, director of Clinical and Translational Research Accelerator at Yale and a regular contributor to this news organization, took to Twitter to put this study in context: “The human exposure equivalent to sucralose would be 60 packets per day,” he pointed out. And the blood levels of S6A with normal consumption would not “come close to the DNA damage threshold noted in the article.”
Perhaps the most concerning scientific data suggesting a link between artificial sweetener use and ill health is a Cleveland Clinic study showing an association between higher blood levels of erythritol and adverse cardiovascular outcomes such as heart attack, stroke, or death. The researchers also found that erythritol, which is found in stevia and some keto food products, made platelet activation and clot formation easier.
When I asked about these findings, Heartland stated, “The study was primarily conducted on patients who were at an elevated risk of cardiovascular events due to their advanced age, elevated body mass and presence of pre-existing health conditions ... the stated findings were only an association and cannot imply causation.”
The main conclusion I’ve drawn on the topic of artificial sweeteners is that a lot of resources were wasted in performing underpowered, poorly designed trials on compounds that are already generally regarded as safe (GRAS) by the FDA. The WHO “conditional guideline” is, by its own description, based on a plethora of “low certainty” to “very low certainty” evidence.
The monies to produce the WHO report and many of these trials would have been better spent educating the public on the difference between simple and complex carbohydrates; the inflammatory and disease-producing effects of excess sugars; and how to prevent, diagnose, and treat diabetes.
If more trials on artificial sweeteners are planned, they should be performed on people doing human things – which does not include ingesting 60 packets of any sweetener in a single day.
In my personal N-of-1 trial, consuming sugar makes me crave more, feel sluggish, and gain weight. I don’t believe that NSS alone will control my weight. But I’ll continue to drink two cups of stevia-laced coffee every morning, take walks, avoid alcohol, eat my vegetables, and hope for the best.
Dr. Walton-Shirley is a clinical cardiologist in Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
More than 140 million Americans use artificial sweeteners, a habit driven by the irrefutable fact that excess sugar is harmful. But I’m continually amazed by alarmist headlines on the topic.
In May, the World Health Organization (WHO) released a report to support its “conditional recommendation” against the use of non-sugar sweeteners (NSS) for weight control. Despite the WHO’s goal “to provide evidence-informed guidance,” the report includes the disclaimer that “The recommendation is based on evidence of low certainty.”
Low certainty is an accurate descriptor for the findings of many of the 280-plus studies in the report. That the guidance does not apply to patients with diabetes was easily lost in the repeated mentions of the perceived dangers of these sugar alternatives.
The review included various table-top and beverage sweeteners, including acesulfame K, aspartame, saccharin, sucralose, stevia, and stevia derivatives. Low-calorie sugars and sugar alcohols such as erythritol were excluded.
The WHO looked at long- and short-term trials, randomized controlled trials (RCTs), prospective studies, and case-control studies measuring a wide range of endpoints, from dental caries to cancer. The report highlighted that some findings cannot be attributed directly to NSS use but may simply be due to their substitution for sugar. Differences in outcomes due to sex, ethnicity, and body weight status could not be assessed either. And the WHO conceded the possibility of reverse causation in observational studies wherein higher-risk individuals may consume more NSS.
Nonnutritive sweeteners are given little credit for weight loss. “A significant difference in body weight and BMI was only observed in trials that reported a reduction in energy intake ... rather than primarily by an inherent property of NSS that can modulate body weight (independently of energy intake),” the report reads. But isn’t the desired effect of using an artificial sweetener instead of table sugar that you lower your calorie intake?
The WHO noted that weight loss was not sustained – a finding in nearly every weight loss trial in history and something more attributable to human nature than the sweetener one chooses.
The document outlines that meta-analyses of prospective cohort studies show that higher intakes of NSS were associated with an increased risk for type 2 diabetes and elevated fasting glucose, while meta-analyses of randomized trials suggest no significant effect on “biomarkers used in the assessment and diagnosis of diabetes and insulin resistance, including fasting glucose, fasting insulin and hemoglobin A1c.”
Similar disparities are noted with cardiovascular risk. Prospective trials suggest an increased risk for CVD, including stroke and its precursor, hypertension; but again, the RCT data found no evidence to suggest a significant effect “on biomarkers used in the assessment and diagnosis of CVDs, including blood pressure, low-density lipoprotein cholesterol and other blood lipids.”
Splenda and stevia under fire
Predictably, some in the nonnutritive sweetener industry are incensed.
Ted Gelov, CEO of Heartland Food Products Group, maker of Splenda, responded in a press release, “Every few years now it seems I have to come to you and clarify misleading headlines ... Suggesting that sweeteners like Splenda cannot have long-term benefits is a disservice to healthcare providers, their patients, and all consumers.”
Splenda has been on the U.S. market since 1999, and Mr. Gelov reportedly uses three to eight packets daily in his coffee and tea.
I reached out to Heartland and they sent me an eight-page document consisting of over 50 statements, summaries, and clinical trials supporting the safety of artificial sweeteners, including sucralose, an ingredient in Splenda. In 2016, Mr. Gelov rebutted claims that sucralose was linked to cancer in Swiss male mice. These “dramatized headlines are based on one flawed study by an isolated Italian research laboratory, the Ramazzini Institute,” Mr. Gelov wrote.
Another recent headline was about the DNA-damaging effects of sucralose-6-acetate (S6A) seen in an in vitro study published in the Journal of Toxicology and Environmental Health. According to the authors, commercial sucralose samples contain up to 0.67% S6A, a manufacturing impurity.
Despite many reports linking this study to Splenda, Heartland wrote that “Splenda and its ingredients were never studied or tested in this research. We, and our suppliers, rigorously and routinely test and monitor for any impurities in our products. We can confirm that S6A is not present in Splenda Brand sucralose down to the lowest detection limit possible, which is .001% sensitivity level.”
F. Perry Wilson, MD, director of Clinical and Translational Research Accelerator at Yale and a regular contributor to this news organization, took to Twitter to put this study in context: “The human exposure equivalent to sucralose would be 60 packets per day,” he pointed out. And the blood levels of S6A with normal consumption would not “come close to the DNA damage threshold noted in the article.”
Perhaps the most concerning scientific data suggesting a link between artificial sweetener use and ill health is a Cleveland Clinic study showing an association between higher blood levels of erythritol and adverse cardiovascular outcomes such as heart attack, stroke, or death. The researchers also found that erythritol, which is found in stevia and some keto food products, made platelet activation and clot formation easier.
When I asked about these findings, Heartland stated, “The study was primarily conducted on patients who were at an elevated risk of cardiovascular events due to their advanced age, elevated body mass and presence of pre-existing health conditions ... the stated findings were only an association and cannot imply causation.”
The main conclusion I’ve drawn on the topic of artificial sweeteners is that a lot of resources were wasted in performing underpowered, poorly designed trials on compounds that are already generally regarded as safe (GRAS) by the FDA. The WHO “conditional guideline” is, by its own description, based on a plethora of “low certainty” to “very low certainty” evidence.
The monies to produce the WHO report and many of these trials would have been better spent educating the public on the difference between simple and complex carbohydrates; the inflammatory and disease-producing effects of excess sugars; and how to prevent, diagnose, and treat diabetes.
If more trials on artificial sweeteners are planned, they should be performed on people doing human things – which does not include ingesting 60 packets of any sweetener in a single day.
In my personal N-of-1 trial, consuming sugar makes me crave more, feel sluggish, and gain weight. I don’t believe that NSS alone will control my weight. But I’ll continue to drink two cups of stevia-laced coffee every morning, take walks, avoid alcohol, eat my vegetables, and hope for the best.
Dr. Walton-Shirley is a clinical cardiologist in Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
More than 140 million Americans use artificial sweeteners, a habit driven by the irrefutable fact that excess sugar is harmful. But I’m continually amazed by alarmist headlines on the topic.
In May, the World Health Organization (WHO) released a report to support its “conditional recommendation” against the use of non-sugar sweeteners (NSS) for weight control. Despite the WHO’s goal “to provide evidence-informed guidance,” the report includes the disclaimer that “The recommendation is based on evidence of low certainty.”
Low certainty is an accurate descriptor for the findings of many of the 280-plus studies in the report. That the guidance does not apply to patients with diabetes was easily lost in the repeated mentions of the perceived dangers of these sugar alternatives.
The review included various table-top and beverage sweeteners, including acesulfame K, aspartame, saccharin, sucralose, stevia, and stevia derivatives. Low-calorie sugars and sugar alcohols such as erythritol were excluded.
The WHO looked at long- and short-term trials, randomized controlled trials (RCTs), prospective studies, and case-control studies measuring a wide range of endpoints, from dental caries to cancer. The report highlighted that some findings cannot be attributed directly to NSS use but may simply be due to their substitution for sugar. Differences in outcomes due to sex, ethnicity, and body weight status could not be assessed either. And the WHO conceded the possibility of reverse causation in observational studies wherein higher-risk individuals may consume more NSS.
Nonnutritive sweeteners are given little credit for weight loss. “A significant difference in body weight and BMI was only observed in trials that reported a reduction in energy intake ... rather than primarily by an inherent property of NSS that can modulate body weight (independently of energy intake),” the report reads. But isn’t the desired effect of using an artificial sweetener instead of table sugar that you lower your calorie intake?
The WHO noted that weight loss was not sustained – a finding in nearly every weight loss trial in history and something more attributable to human nature than the sweetener one chooses.
The document outlines that meta-analyses of prospective cohort studies show that higher intakes of NSS were associated with an increased risk for type 2 diabetes and elevated fasting glucose, while meta-analyses of randomized trials suggest no significant effect on “biomarkers used in the assessment and diagnosis of diabetes and insulin resistance, including fasting glucose, fasting insulin and hemoglobin A1c.”
Similar disparities are noted with cardiovascular risk. Prospective trials suggest an increased risk for CVD, including stroke and its precursor, hypertension; but again, the RCT data found no evidence to suggest a significant effect “on biomarkers used in the assessment and diagnosis of CVDs, including blood pressure, low-density lipoprotein cholesterol and other blood lipids.”
Splenda and stevia under fire
Predictably, some in the nonnutritive sweetener industry are incensed.
Ted Gelov, CEO of Heartland Food Products Group, maker of Splenda, responded in a press release, “Every few years now it seems I have to come to you and clarify misleading headlines ... Suggesting that sweeteners like Splenda cannot have long-term benefits is a disservice to healthcare providers, their patients, and all consumers.”
Splenda has been on the U.S. market since 1999, and Mr. Gelov reportedly uses three to eight packets daily in his coffee and tea.
I reached out to Heartland and they sent me an eight-page document consisting of over 50 statements, summaries, and clinical trials supporting the safety of artificial sweeteners, including sucralose, an ingredient in Splenda. In 2016, Mr. Gelov rebutted claims that sucralose was linked to cancer in Swiss male mice. These “dramatized headlines are based on one flawed study by an isolated Italian research laboratory, the Ramazzini Institute,” Mr. Gelov wrote.
Another recent headline was about the DNA-damaging effects of sucralose-6-acetate (S6A) seen in an in vitro study published in the Journal of Toxicology and Environmental Health. According to the authors, commercial sucralose samples contain up to 0.67% S6A, a manufacturing impurity.
Despite many reports linking this study to Splenda, Heartland wrote that “Splenda and its ingredients were never studied or tested in this research. We, and our suppliers, rigorously and routinely test and monitor for any impurities in our products. We can confirm that S6A is not present in Splenda Brand sucralose down to the lowest detection limit possible, which is .001% sensitivity level.”
F. Perry Wilson, MD, director of Clinical and Translational Research Accelerator at Yale and a regular contributor to this news organization, took to Twitter to put this study in context: “The human exposure equivalent to sucralose would be 60 packets per day,” he pointed out. And the blood levels of S6A with normal consumption would not “come close to the DNA damage threshold noted in the article.”
Perhaps the most concerning scientific data suggesting a link between artificial sweetener use and ill health is a Cleveland Clinic study showing an association between higher blood levels of erythritol and adverse cardiovascular outcomes such as heart attack, stroke, or death. The researchers also found that erythritol, which is found in stevia and some keto food products, made platelet activation and clot formation easier.
When I asked about these findings, Heartland stated, “The study was primarily conducted on patients who were at an elevated risk of cardiovascular events due to their advanced age, elevated body mass and presence of pre-existing health conditions ... the stated findings were only an association and cannot imply causation.”
The main conclusion I’ve drawn on the topic of artificial sweeteners is that a lot of resources were wasted in performing underpowered, poorly designed trials on compounds that are already generally regarded as safe (GRAS) by the FDA. The WHO “conditional guideline” is, by its own description, based on a plethora of “low certainty” to “very low certainty” evidence.
The monies to produce the WHO report and many of these trials would have been better spent educating the public on the difference between simple and complex carbohydrates; the inflammatory and disease-producing effects of excess sugars; and how to prevent, diagnose, and treat diabetes.
If more trials on artificial sweeteners are planned, they should be performed on people doing human things – which does not include ingesting 60 packets of any sweetener in a single day.
In my personal N-of-1 trial, consuming sugar makes me crave more, feel sluggish, and gain weight. I don’t believe that NSS alone will control my weight. But I’ll continue to drink two cups of stevia-laced coffee every morning, take walks, avoid alcohol, eat my vegetables, and hope for the best.
Dr. Walton-Shirley is a clinical cardiologist in Nashville, Tenn. She disclosed no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Harvard medical school sued over stolen body part scandal
Plaintiffs include relatives of people whose remains were allegedly stolen and sold. The lawsuit alleges that as many as 400 cadavers may have been trafficked in a multi-year scheme. Details were revealed in a June 13 indictment by the U.S. attorney for the Middle District of Pennsylvania.
“Medical schools like Harvard have a duty to ensure [donated remains] are handled properly and with decency and to ensure they are used for their intended purpose of scientific study,” attorney Jeff Catalano said in a statement.
“I do think Harvard has that duty,” said Arthur Caplan, PhD, director, Division of Medical Ethics, New York University. But, he added, “I will say there’s not much they can do when employees set out to systematically undermine them.”
The indictment alleges that from 2018 through August 2022, Harvard morgue manager Cedric Lodge stole dissected portions of donated cadavers, including heads, brains, skin, and bones, which were then sold by him and his wife, Denise Lodge, to Katrina Maclean, owner of Kat’s Creepy Creations, Peabody, Mass. Ms. Maclean allegedly sold human remains to Joshua Taylor and Jeremy Pauley, both Pennsylvania residents.
On occasion, Mr. Lodge allowed Ms. Maclean, Mr. Taylor, and others into the morgue to choose which parts they wanted, according to the indictment. Mr. Taylor, Ms. Maclean, and Denise Lodge are all named in the indictment. Mr. Pauley was charged separately.
They each face a maximum of 15 years in prison.
Ms. Maclean allegedly bought two dissected faces for $600 and shipped human skin to Mr. Pauley to be made into leather; Mr. Pauley then eventually shipped the “tanned human skin” back to Ms. Maclean, according to the indictment. Over a 3-year period, Mr. Taylor paid the Lodges some $37,000 for stolen human remains, the indictment charges.
Mr. Pauley also purchased human remains from Candace Chapman Scott, who stole them from her employer, a mortuary in Little Rock, Ark. The mortuary received remains for cremation from an area medical school, according to the indictment.
After being notified of the investigation in March, Harvard cooperated fully, the school said in a statement from George Q. Daley, MD, PhD, dean of the Faculty of Medicine.
“We are appalled to learn that something so disturbing could happen on our campus – a community dedicated to healing and serving others,” the statement said. “The reported incidents are a betrayal of HMS and, most importantly, each of the individuals who altruistically chose to will their bodies to HMS through the Anatomical Gift Program to advance medical education and research.”
The U.S. attorney thanked Harvard for its cooperation, saying that it “is also a victim here.”
Dr. Caplan, who also writes an ethics column for this news organization, agrees. The school was betrayed, he said.
“You expect professionalism, integrity on the part of your doctors, on the part of your technicians, on the part of your workforce,” he said. He noted that those expectations are explained in institutions’ codes of ethics and policies.
Harvard said Mr. Lodge had worked in the morgue since 1995 and that he took several leaves: from September 2021 to February 2022, and again starting February 14. The school terminated his employment on May 6.
His duties included intake of anatomic donors’ bodies. He coordinated embalming and oversaw the storage and movement of cadavers to and from teaching labs. When studies were complete, he prepared remains to be transported to and from the external crematorium and, when appropriate, for burial, according to a Harvard fact sheet for families.
The medical school has convened an outside expert panel to evaluate the Anatomical Gift Program and morgue policies and practices. The panel is expected to make its findings public at the end of the summer.
Dr. Caplan said he hoped the committee recommends unannounced audits of cadaver donation programs and medical tissue and bone suppliers, which could help expose illicit diversions. “You need to keep an eye, which no one seems to do because it’s a state issue and it’s not a priority, on that trade in body parts,” he said.
He believes other medical schools will reexamine their donation programs, especially given Harvard’s status.
“With a prominent place like that having this kind of problem, I can’t imagine there’s not a little bit of a scramble at a lot of the body programs to make sure that they know that things are as they should be,” Dr. Caplan said.
A version of this article first appeared on Medscape.com.
Plaintiffs include relatives of people whose remains were allegedly stolen and sold. The lawsuit alleges that as many as 400 cadavers may have been trafficked in a multi-year scheme. Details were revealed in a June 13 indictment by the U.S. attorney for the Middle District of Pennsylvania.
“Medical schools like Harvard have a duty to ensure [donated remains] are handled properly and with decency and to ensure they are used for their intended purpose of scientific study,” attorney Jeff Catalano said in a statement.
“I do think Harvard has that duty,” said Arthur Caplan, PhD, director, Division of Medical Ethics, New York University. But, he added, “I will say there’s not much they can do when employees set out to systematically undermine them.”
The indictment alleges that from 2018 through August 2022, Harvard morgue manager Cedric Lodge stole dissected portions of donated cadavers, including heads, brains, skin, and bones, which were then sold by him and his wife, Denise Lodge, to Katrina Maclean, owner of Kat’s Creepy Creations, Peabody, Mass. Ms. Maclean allegedly sold human remains to Joshua Taylor and Jeremy Pauley, both Pennsylvania residents.
On occasion, Mr. Lodge allowed Ms. Maclean, Mr. Taylor, and others into the morgue to choose which parts they wanted, according to the indictment. Mr. Taylor, Ms. Maclean, and Denise Lodge are all named in the indictment. Mr. Pauley was charged separately.
They each face a maximum of 15 years in prison.
Ms. Maclean allegedly bought two dissected faces for $600 and shipped human skin to Mr. Pauley to be made into leather; Mr. Pauley then eventually shipped the “tanned human skin” back to Ms. Maclean, according to the indictment. Over a 3-year period, Mr. Taylor paid the Lodges some $37,000 for stolen human remains, the indictment charges.
Mr. Pauley also purchased human remains from Candace Chapman Scott, who stole them from her employer, a mortuary in Little Rock, Ark. The mortuary received remains for cremation from an area medical school, according to the indictment.
After being notified of the investigation in March, Harvard cooperated fully, the school said in a statement from George Q. Daley, MD, PhD, dean of the Faculty of Medicine.
“We are appalled to learn that something so disturbing could happen on our campus – a community dedicated to healing and serving others,” the statement said. “The reported incidents are a betrayal of HMS and, most importantly, each of the individuals who altruistically chose to will their bodies to HMS through the Anatomical Gift Program to advance medical education and research.”
The U.S. attorney thanked Harvard for its cooperation, saying that it “is also a victim here.”
Dr. Caplan, who also writes an ethics column for this news organization, agrees. The school was betrayed, he said.
“You expect professionalism, integrity on the part of your doctors, on the part of your technicians, on the part of your workforce,” he said. He noted that those expectations are explained in institutions’ codes of ethics and policies.
Harvard said Mr. Lodge had worked in the morgue since 1995 and that he took several leaves: from September 2021 to February 2022, and again starting February 14. The school terminated his employment on May 6.
His duties included intake of anatomic donors’ bodies. He coordinated embalming and oversaw the storage and movement of cadavers to and from teaching labs. When studies were complete, he prepared remains to be transported to and from the external crematorium and, when appropriate, for burial, according to a Harvard fact sheet for families.
The medical school has convened an outside expert panel to evaluate the Anatomical Gift Program and morgue policies and practices. The panel is expected to make its findings public at the end of the summer.
Dr. Caplan said he hoped the committee recommends unannounced audits of cadaver donation programs and medical tissue and bone suppliers, which could help expose illicit diversions. “You need to keep an eye, which no one seems to do because it’s a state issue and it’s not a priority, on that trade in body parts,” he said.
He believes other medical schools will reexamine their donation programs, especially given Harvard’s status.
“With a prominent place like that having this kind of problem, I can’t imagine there’s not a little bit of a scramble at a lot of the body programs to make sure that they know that things are as they should be,” Dr. Caplan said.
A version of this article first appeared on Medscape.com.
Plaintiffs include relatives of people whose remains were allegedly stolen and sold. The lawsuit alleges that as many as 400 cadavers may have been trafficked in a multi-year scheme. Details were revealed in a June 13 indictment by the U.S. attorney for the Middle District of Pennsylvania.
“Medical schools like Harvard have a duty to ensure [donated remains] are handled properly and with decency and to ensure they are used for their intended purpose of scientific study,” attorney Jeff Catalano said in a statement.
“I do think Harvard has that duty,” said Arthur Caplan, PhD, director, Division of Medical Ethics, New York University. But, he added, “I will say there’s not much they can do when employees set out to systematically undermine them.”
The indictment alleges that from 2018 through August 2022, Harvard morgue manager Cedric Lodge stole dissected portions of donated cadavers, including heads, brains, skin, and bones, which were then sold by him and his wife, Denise Lodge, to Katrina Maclean, owner of Kat’s Creepy Creations, Peabody, Mass. Ms. Maclean allegedly sold human remains to Joshua Taylor and Jeremy Pauley, both Pennsylvania residents.
On occasion, Mr. Lodge allowed Ms. Maclean, Mr. Taylor, and others into the morgue to choose which parts they wanted, according to the indictment. Mr. Taylor, Ms. Maclean, and Denise Lodge are all named in the indictment. Mr. Pauley was charged separately.
They each face a maximum of 15 years in prison.
Ms. Maclean allegedly bought two dissected faces for $600 and shipped human skin to Mr. Pauley to be made into leather; Mr. Pauley then eventually shipped the “tanned human skin” back to Ms. Maclean, according to the indictment. Over a 3-year period, Mr. Taylor paid the Lodges some $37,000 for stolen human remains, the indictment charges.
Mr. Pauley also purchased human remains from Candace Chapman Scott, who stole them from her employer, a mortuary in Little Rock, Ark. The mortuary received remains for cremation from an area medical school, according to the indictment.
After being notified of the investigation in March, Harvard cooperated fully, the school said in a statement from George Q. Daley, MD, PhD, dean of the Faculty of Medicine.
“We are appalled to learn that something so disturbing could happen on our campus – a community dedicated to healing and serving others,” the statement said. “The reported incidents are a betrayal of HMS and, most importantly, each of the individuals who altruistically chose to will their bodies to HMS through the Anatomical Gift Program to advance medical education and research.”
The U.S. attorney thanked Harvard for its cooperation, saying that it “is also a victim here.”
Dr. Caplan, who also writes an ethics column for this news organization, agrees. The school was betrayed, he said.
“You expect professionalism, integrity on the part of your doctors, on the part of your technicians, on the part of your workforce,” he said. He noted that those expectations are explained in institutions’ codes of ethics and policies.
Harvard said Mr. Lodge had worked in the morgue since 1995 and that he took several leaves: from September 2021 to February 2022, and again starting February 14. The school terminated his employment on May 6.
His duties included intake of anatomic donors’ bodies. He coordinated embalming and oversaw the storage and movement of cadavers to and from teaching labs. When studies were complete, he prepared remains to be transported to and from the external crematorium and, when appropriate, for burial, according to a Harvard fact sheet for families.
The medical school has convened an outside expert panel to evaluate the Anatomical Gift Program and morgue policies and practices. The panel is expected to make its findings public at the end of the summer.
Dr. Caplan said he hoped the committee recommends unannounced audits of cadaver donation programs and medical tissue and bone suppliers, which could help expose illicit diversions. “You need to keep an eye, which no one seems to do because it’s a state issue and it’s not a priority, on that trade in body parts,” he said.
He believes other medical schools will reexamine their donation programs, especially given Harvard’s status.
“With a prominent place like that having this kind of problem, I can’t imagine there’s not a little bit of a scramble at a lot of the body programs to make sure that they know that things are as they should be,” Dr. Caplan said.
A version of this article first appeared on Medscape.com.
Product updates and reviews
REVIEW
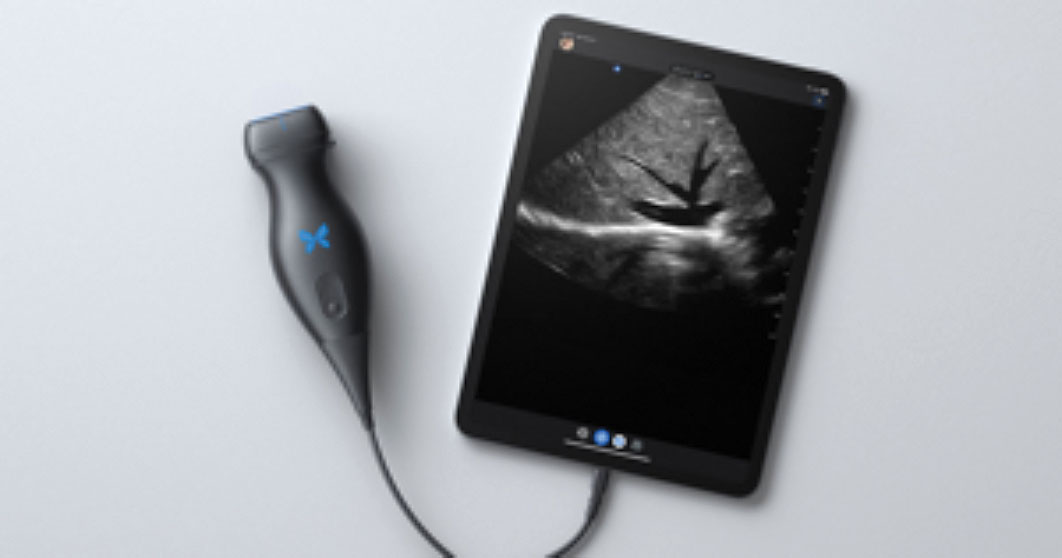
Butterfly iQ+: Offering day-to-day portable ultrasound tech
The Butterfly iQ+ app with an ultrasound probe and cable is available from Butterfly Network, Inc, in Guilford, Connecticut.
Background. It could be reasonably argued that ultrasonography has surpassed the speculum as the single most important tool in ObGyn. From its origins in 1949 with the pioneering work of George Ludwig using A-mode (amplitude-mode) ultrasound and the first publication of its use in pregnancy using B-mode (brightness-mode) ultrasound in Lancet in 1958 by Donald and colleagues, this technology has become so ingrained into ObGyn that it is often frustrating to practice comfortably without it. Thus, today, the biggest question facing most practitioners is not whether or not to have an ultrasound in their practice but which one to have.
Given the wide range of quality, functionality, and price within the ultrasound device space, choosing the right technology can feel as daunting as choosing the perfect restaurant in New York City. That said, when looking for entry-level ultrasound technology to address the day-to-day basic needs of your average ObGyn, the Butterfly iQ+ may be an easy choice.
Design/Functionality. The Butterfly iQ+ app does not come with a screen. Rather, the device is compatible with both iOS and Android systems and readily connects to a vast array of easily purchased devices, with either lightening or USB-C ports. In our office, we use an iPad mini. The probe is lightweight (309 g) and contains a rechargeable 2600 mAh lithium ion battery, so that its power source is independent of the device to which it is attached. The probe is a 2D array with 9000 micro-machined sensors. It allows for imaging using M-mode, B-mode, Color Doppler, Power Doppler, and Pulsed Wave Doppler. (I don’t know what the last two are or what they are used for, but they sound important.) It has a scan depth range of 1 cm to 30 cm. The downloadable Butterfly iQ+ app that has the software that makes the probe functional has more tools, controls, and presets than anyone could ever need. But that’s not all. The App has data encrypted HIPAA/HITECH-compliant Cloud-based connectivity that offers unlimited image storage, access to reports, and embedded CPT codes should billing capabilities be needed.
The true beauty of the Butterfly iQ+ is that the image quality is awesome and it is really easy to use. The software is mostly intuitive and takes only a minimal effort to learn. The device holds its charge more than adequately for a day in the office and the recharging process is fast and easy. When it comes to the device’s design and functionality–as a Capricorn–I am still looking for its flaws.
Innovation. The real innovations of the Butterfly iQ+ are its “ultrasound-on-a-chip”™ technology and its incorporation of a rechargeable battery into the probe. This combination allows for crystal clear imaging in a cordless, portable device. While most other similar technologies waste their time, technology, space, and cost on the screen, the Butterfly iQ+ punted on that challenge and put all their efforts into the probe and the software. It was a great choice.
Summary. In our office, the Butterfly iQ+ has changed the way we practice. Our trusty fetal dopplers are mostly gone, having been replaced by the Butterfly iQ+. At almost every prenatal visit, patients can now see their baby rather than just hear the heartbeat (and they can hear it too if they want by using the M-mode functionality on the device). Patients love it, and so do the doctors. Instead of just hearing heart beats, fetal position and quick fluid checks are now routine, so we think our care is actually a little better than it was. The Butterfly iQ+ is also great for confirming IUD locations after placement or when the strings are not visible. All-in-all, I love this product. Who doesn’t love butterflies?!
For more information, visit https://www.butterflynetwork.com
The views of the author are personal opinions and do not necessarily represent the views of OBG
References
- Kaproth-Joslin KA, Nicola R, Dogra VS. The History of US: from bats and boats to the bedside and beyond: RSNA centennial article. Radiographics. 2015;35:960-970.
- Donald I, MacVicar J, Brown TG. Investigation of abdominal masses by pulsed ultrasound. Lancet. 1958;1:1188-1195.
REVIEW
Butterfly iQ+: Offering day-to-day portable ultrasound tech
The Butterfly iQ+ app with an ultrasound probe and cable is available from Butterfly Network, Inc, in Guilford, Connecticut.
Background. It could be reasonably argued that ultrasonography has surpassed the speculum as the single most important tool in ObGyn. From its origins in 1949 with the pioneering work of George Ludwig using A-mode (amplitude-mode) ultrasound and the first publication of its use in pregnancy using B-mode (brightness-mode) ultrasound in Lancet in 1958 by Donald and colleagues, this technology has become so ingrained into ObGyn that it is often frustrating to practice comfortably without it. Thus, today, the biggest question facing most practitioners is not whether or not to have an ultrasound in their practice but which one to have.
Given the wide range of quality, functionality, and price within the ultrasound device space, choosing the right technology can feel as daunting as choosing the perfect restaurant in New York City. That said, when looking for entry-level ultrasound technology to address the day-to-day basic needs of your average ObGyn, the Butterfly iQ+ may be an easy choice.
Design/Functionality. The Butterfly iQ+ app does not come with a screen. Rather, the device is compatible with both iOS and Android systems and readily connects to a vast array of easily purchased devices, with either lightening or USB-C ports. In our office, we use an iPad mini. The probe is lightweight (309 g) and contains a rechargeable 2600 mAh lithium ion battery, so that its power source is independent of the device to which it is attached. The probe is a 2D array with 9000 micro-machined sensors. It allows for imaging using M-mode, B-mode, Color Doppler, Power Doppler, and Pulsed Wave Doppler. (I don’t know what the last two are or what they are used for, but they sound important.) It has a scan depth range of 1 cm to 30 cm. The downloadable Butterfly iQ+ app that has the software that makes the probe functional has more tools, controls, and presets than anyone could ever need. But that’s not all. The App has data encrypted HIPAA/HITECH-compliant Cloud-based connectivity that offers unlimited image storage, access to reports, and embedded CPT codes should billing capabilities be needed.
The true beauty of the Butterfly iQ+ is that the image quality is awesome and it is really easy to use. The software is mostly intuitive and takes only a minimal effort to learn. The device holds its charge more than adequately for a day in the office and the recharging process is fast and easy. When it comes to the device’s design and functionality–as a Capricorn–I am still looking for its flaws.
Innovation. The real innovations of the Butterfly iQ+ are its “ultrasound-on-a-chip”™ technology and its incorporation of a rechargeable battery into the probe. This combination allows for crystal clear imaging in a cordless, portable device. While most other similar technologies waste their time, technology, space, and cost on the screen, the Butterfly iQ+ punted on that challenge and put all their efforts into the probe and the software. It was a great choice.
Summary. In our office, the Butterfly iQ+ has changed the way we practice. Our trusty fetal dopplers are mostly gone, having been replaced by the Butterfly iQ+. At almost every prenatal visit, patients can now see their baby rather than just hear the heartbeat (and they can hear it too if they want by using the M-mode functionality on the device). Patients love it, and so do the doctors. Instead of just hearing heart beats, fetal position and quick fluid checks are now routine, so we think our care is actually a little better than it was. The Butterfly iQ+ is also great for confirming IUD locations after placement or when the strings are not visible. All-in-all, I love this product. Who doesn’t love butterflies?!
For more information, visit https://www.butterflynetwork.com
The views of the author are personal opinions and do not necessarily represent the views of OBG
References
- Kaproth-Joslin KA, Nicola R, Dogra VS. The History of US: from bats and boats to the bedside and beyond: RSNA centennial article. Radiographics. 2015;35:960-970.
- Donald I, MacVicar J, Brown TG. Investigation of abdominal masses by pulsed ultrasound. Lancet. 1958;1:1188-1195.
REVIEW
Butterfly iQ+: Offering day-to-day portable ultrasound tech
The Butterfly iQ+ app with an ultrasound probe and cable is available from Butterfly Network, Inc, in Guilford, Connecticut.
Background. It could be reasonably argued that ultrasonography has surpassed the speculum as the single most important tool in ObGyn. From its origins in 1949 with the pioneering work of George Ludwig using A-mode (amplitude-mode) ultrasound and the first publication of its use in pregnancy using B-mode (brightness-mode) ultrasound in Lancet in 1958 by Donald and colleagues, this technology has become so ingrained into ObGyn that it is often frustrating to practice comfortably without it. Thus, today, the biggest question facing most practitioners is not whether or not to have an ultrasound in their practice but which one to have.
Given the wide range of quality, functionality, and price within the ultrasound device space, choosing the right technology can feel as daunting as choosing the perfect restaurant in New York City. That said, when looking for entry-level ultrasound technology to address the day-to-day basic needs of your average ObGyn, the Butterfly iQ+ may be an easy choice.
Design/Functionality. The Butterfly iQ+ app does not come with a screen. Rather, the device is compatible with both iOS and Android systems and readily connects to a vast array of easily purchased devices, with either lightening or USB-C ports. In our office, we use an iPad mini. The probe is lightweight (309 g) and contains a rechargeable 2600 mAh lithium ion battery, so that its power source is independent of the device to which it is attached. The probe is a 2D array with 9000 micro-machined sensors. It allows for imaging using M-mode, B-mode, Color Doppler, Power Doppler, and Pulsed Wave Doppler. (I don’t know what the last two are or what they are used for, but they sound important.) It has a scan depth range of 1 cm to 30 cm. The downloadable Butterfly iQ+ app that has the software that makes the probe functional has more tools, controls, and presets than anyone could ever need. But that’s not all. The App has data encrypted HIPAA/HITECH-compliant Cloud-based connectivity that offers unlimited image storage, access to reports, and embedded CPT codes should billing capabilities be needed.
The true beauty of the Butterfly iQ+ is that the image quality is awesome and it is really easy to use. The software is mostly intuitive and takes only a minimal effort to learn. The device holds its charge more than adequately for a day in the office and the recharging process is fast and easy. When it comes to the device’s design and functionality–as a Capricorn–I am still looking for its flaws.
Innovation. The real innovations of the Butterfly iQ+ are its “ultrasound-on-a-chip”™ technology and its incorporation of a rechargeable battery into the probe. This combination allows for crystal clear imaging in a cordless, portable device. While most other similar technologies waste their time, technology, space, and cost on the screen, the Butterfly iQ+ punted on that challenge and put all their efforts into the probe and the software. It was a great choice.
Summary. In our office, the Butterfly iQ+ has changed the way we practice. Our trusty fetal dopplers are mostly gone, having been replaced by the Butterfly iQ+. At almost every prenatal visit, patients can now see their baby rather than just hear the heartbeat (and they can hear it too if they want by using the M-mode functionality on the device). Patients love it, and so do the doctors. Instead of just hearing heart beats, fetal position and quick fluid checks are now routine, so we think our care is actually a little better than it was. The Butterfly iQ+ is also great for confirming IUD locations after placement or when the strings are not visible. All-in-all, I love this product. Who doesn’t love butterflies?!
For more information, visit https://www.butterflynetwork.com
The views of the author are personal opinions and do not necessarily represent the views of OBG
References
- Kaproth-Joslin KA, Nicola R, Dogra VS. The History of US: from bats and boats to the bedside and beyond: RSNA centennial article. Radiographics. 2015;35:960-970.
- Donald I, MacVicar J, Brown TG. Investigation of abdominal masses by pulsed ultrasound. Lancet. 1958;1:1188-1195.
New cannabis laws, higher binge drinking rates linked
TOPLINE:
METHODOLOGY:
Among adolescents, binge drinking, defined as having five or more drinks for men and four or more drinks for women at one time, is associated with poor academic performance, sexual risk, and injury in the short term, as well as the development of alcohol use disorder and academic disengagement in the long term.
Current evidence regarding the association between recreational cannabis laws (RCLs) and binge drinking is limited.
States in which RCLs have been implemented include Colorado, Washington, Alaska, Oregon, Nevada, California, Massachusetts, and Vermont, as well as the District of Columbia.
The study included 817,359 people aged 12 and older who participated in the 2008-2019 National Survey on Drug Use and Health (NSDUH), a nationally representative survey of the U.S. population.
TAKEAWAY:
Overall, states that have not enacted cannabis laws showed consistently lower rates of binge drinking over time among all age groups.
In all states, there were substantial declines in reporting of past-month binge drinking in some age groups – from 17.5% (95% confidence interval, 16.9-18.2) in 2008 to 11.1% (10.4-11.8) in 2019 among those aged 12-20 and a drop from 43.7% (42.4-44.9) to 40.2% (39.1-41.1) among those aged 21-30.
There were overall increases in binge drinking in all states regardless of cannabis laws among individuals aged 31 and older. The most extensive increases were among people aged 31-40 (from 28.1% [95% CI, 26.6-29.6] to 33.3% [32.1-34.6]), followed by participants aged 51 and over (from 13.3% [95% CI, 12.2-14.4] to 16.8% [15.8-17.7]).
IN PRACTICE:
“Our findings support calls to reinforce health care providers’ discussions about alcohol use with older adults,” particularly in RCL states, the researchers write.
STUDY DETAILS:
The study was conducted out by Priscila Dib Gonçalves, PhD, department of epidemiology, Columbia University School of Public Health, New York, and colleagues. It was published in the International Journal of Drug Policy.
LIMITATIONS:
Alcohol-related measures, including binge drinking, were self-reported, which may introduce recall bias and underreporting. NSDUH binge drinking measures were not adjusted for sex differences from 2008 to 2014, which may result in underreporting of binge drinking in females before 2015. The researchers did not examine cannabis policy provisions, such as cultivation restrictions, pricing control, the tax imposed, and consumption restrictions.
DISCLOSURES:
The study received support from the National Institutes of Health, the National Institute on Drug Abuse, the National Center for Injury Prevention and Control, and the Centers for Disease Control and Prevention. The authors report no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
Among adolescents, binge drinking, defined as having five or more drinks for men and four or more drinks for women at one time, is associated with poor academic performance, sexual risk, and injury in the short term, as well as the development of alcohol use disorder and academic disengagement in the long term.
Current evidence regarding the association between recreational cannabis laws (RCLs) and binge drinking is limited.
States in which RCLs have been implemented include Colorado, Washington, Alaska, Oregon, Nevada, California, Massachusetts, and Vermont, as well as the District of Columbia.
The study included 817,359 people aged 12 and older who participated in the 2008-2019 National Survey on Drug Use and Health (NSDUH), a nationally representative survey of the U.S. population.
TAKEAWAY:
Overall, states that have not enacted cannabis laws showed consistently lower rates of binge drinking over time among all age groups.
In all states, there were substantial declines in reporting of past-month binge drinking in some age groups – from 17.5% (95% confidence interval, 16.9-18.2) in 2008 to 11.1% (10.4-11.8) in 2019 among those aged 12-20 and a drop from 43.7% (42.4-44.9) to 40.2% (39.1-41.1) among those aged 21-30.
There were overall increases in binge drinking in all states regardless of cannabis laws among individuals aged 31 and older. The most extensive increases were among people aged 31-40 (from 28.1% [95% CI, 26.6-29.6] to 33.3% [32.1-34.6]), followed by participants aged 51 and over (from 13.3% [95% CI, 12.2-14.4] to 16.8% [15.8-17.7]).
IN PRACTICE:
“Our findings support calls to reinforce health care providers’ discussions about alcohol use with older adults,” particularly in RCL states, the researchers write.
STUDY DETAILS:
The study was conducted out by Priscila Dib Gonçalves, PhD, department of epidemiology, Columbia University School of Public Health, New York, and colleagues. It was published in the International Journal of Drug Policy.
LIMITATIONS:
Alcohol-related measures, including binge drinking, were self-reported, which may introduce recall bias and underreporting. NSDUH binge drinking measures were not adjusted for sex differences from 2008 to 2014, which may result in underreporting of binge drinking in females before 2015. The researchers did not examine cannabis policy provisions, such as cultivation restrictions, pricing control, the tax imposed, and consumption restrictions.
DISCLOSURES:
The study received support from the National Institutes of Health, the National Institute on Drug Abuse, the National Center for Injury Prevention and Control, and the Centers for Disease Control and Prevention. The authors report no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
Among adolescents, binge drinking, defined as having five or more drinks for men and four or more drinks for women at one time, is associated with poor academic performance, sexual risk, and injury in the short term, as well as the development of alcohol use disorder and academic disengagement in the long term.
Current evidence regarding the association between recreational cannabis laws (RCLs) and binge drinking is limited.
States in which RCLs have been implemented include Colorado, Washington, Alaska, Oregon, Nevada, California, Massachusetts, and Vermont, as well as the District of Columbia.
The study included 817,359 people aged 12 and older who participated in the 2008-2019 National Survey on Drug Use and Health (NSDUH), a nationally representative survey of the U.S. population.
TAKEAWAY:
Overall, states that have not enacted cannabis laws showed consistently lower rates of binge drinking over time among all age groups.
In all states, there were substantial declines in reporting of past-month binge drinking in some age groups – from 17.5% (95% confidence interval, 16.9-18.2) in 2008 to 11.1% (10.4-11.8) in 2019 among those aged 12-20 and a drop from 43.7% (42.4-44.9) to 40.2% (39.1-41.1) among those aged 21-30.
There were overall increases in binge drinking in all states regardless of cannabis laws among individuals aged 31 and older. The most extensive increases were among people aged 31-40 (from 28.1% [95% CI, 26.6-29.6] to 33.3% [32.1-34.6]), followed by participants aged 51 and over (from 13.3% [95% CI, 12.2-14.4] to 16.8% [15.8-17.7]).
IN PRACTICE:
“Our findings support calls to reinforce health care providers’ discussions about alcohol use with older adults,” particularly in RCL states, the researchers write.
STUDY DETAILS:
The study was conducted out by Priscila Dib Gonçalves, PhD, department of epidemiology, Columbia University School of Public Health, New York, and colleagues. It was published in the International Journal of Drug Policy.
LIMITATIONS:
Alcohol-related measures, including binge drinking, were self-reported, which may introduce recall bias and underreporting. NSDUH binge drinking measures were not adjusted for sex differences from 2008 to 2014, which may result in underreporting of binge drinking in females before 2015. The researchers did not examine cannabis policy provisions, such as cultivation restrictions, pricing control, the tax imposed, and consumption restrictions.
DISCLOSURES:
The study received support from the National Institutes of Health, the National Institute on Drug Abuse, the National Center for Injury Prevention and Control, and the Centers for Disease Control and Prevention. The authors report no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Ruptured aneurysm turns MD couple into doctor-patient
Dr. Taylor Delgado: It was Saturday night, and we had just gone to bed. Suddenly, Ali sat up, and screamed, “My head!” She then became nonresponsive and had a seizure. I was in disbelief, but I also knew exactly what was happening. I called 911: “My wife is having a head bleed. I need an ambulance.” It was a bad connection, and they could barely understand me.
As I tried to carry Ali downstairs, she vomited. She still had rubber bands in her mouth from the jaw fracture that was a result of her accident just a month ago. I knew she needed an airway.
I grabbed a tracheostomy tube, but the opening over her trachea put in for the accident had since closed. I tried to push the tube through her neck, but it hurt her; her eyes opened.
I thought to myself: Maybe she doesn’t need it. This can wait until she gets to the hospital. I can’t do this to her. But she vomited again, and I knew what I had to do.
We were at the top of our stairs. I didn’t have a blade or any other equipment, just the tracheostomy tube with the dilator. I pushed hard, and she started fighting me. I had to hold her hands away with one arm. The tube popped in and she stared back at me in pain and fear.
I finally got her downstairs and called medical control at University Hospital of Cincinnati. I was able to speak with one of the attendings: “Ali’s aneurysm ruptured, and she just had a seizure. She has a GCS of 11 or 12. I replaced her tracheostomy tube. We’ll be there shortly.”
When I heard sirens come down our street, I carried Ali outside, but the sirens were from a firetruck. They likely assumed someone had fallen and had a head laceration. It was beyond deflating. I yelled incredulously: “We need an ambulance here now!”
When the ambulance finally arrived, they tried to tell me that I could not ride with them. Or if I did, I would have to sit up front. After arguing back and forth for a few seconds, I finally demanded: “This is medical control. This is MD-88, and this is my patient. I’m sitting in back with you. She needs four Zofran and two midazolam IV now.”
One month earlier ...
Dr. Alison Delgado: Taylor and I were both 4 months into our second year of residency, and we had been married for 5 months. I was a pediatric resident at Cincinnati Children’s Hospital. She was an emergency medicine resident at the University Hospital. I was having my first day off in a couple weeks, and she was working a shift in the emergency department. She was also a part of the flight crew that day. Second-year residents would go out to the scenes of accidents or to other hospitals to transport the patient back to their Level I trauma center via helicopter. The resident was the physician and considered the leader on these flights.
That afternoon, I went for a bicycle ride. About three-quarters of the way through my ride, I was struck by a car.
The EMS crew got to me fairly quickly. They intubated me at the scene and got me to the closest hospital. Immediately, the hospital realized my case was outside the scope of their care. They contacted University Hospital requesting that their flight crew come to transport me.
Dr. Taylor Delgado: At around 5:30 p.m. the day of my shift, the tones went out on the radio: “AirCare 1 and Pod Doc, you are requested for interhospital transfer, 27-year-old Jane Doe, GCS 5.” That was the only information given.
When we landed at the hospital, I walked in with my nurse. I was listening to the doctor’s report and doing my once over. The patient was a little bit bradycardic, heart rate in the 40s or 50s. Blood pressure was normal if not a little bit elevated. There was obvious facial trauma. The endotracheal tube in place.
She was covered with a blanket, but some of her clothing was visible. Suddenly, I recognized it. It was our cycling team’s kit. I thought, please don’t let it be Ali.
My flight nurse went out and called back to dispatch. “This is my doc’s wife. Dispatch the second helicopter!” She had to repeat herself a few times before they understood what was happening.
As Ali’s spouse, I couldn’t be the flight doctor. I didn’t care. I called medical control myself and told them: “This is Ali. We have to fly her. She has a head injury.” They said: “You can’t fly her.” I said: “We can’t delay her care. I have to fly her.” They said: “No, you can’t fly her.” I broke down. Devastated.
I went back into the room and looked at Ali. Her heart rate was dropping. My flight nurse was in the trauma bay with the emergency physician. We realized definitive care was being delayed because of my presence, which was an awful feeling to have. I think at that point we realized, you do nothing, or you act. So, we acted.
I told my flight nurse: “Let’s give her atropine to increase her heart rate.” I asked about sedation, and she hadn’t had anything. I spurted off some doses: “a hundred of fentanyl and five of midazolam.” My flight nurse actually administered smaller doses. She thought it was a bit aggressive, and she was correct. I was trying to maintain composure, but it was hard.
The emergency medicine physician volunteered to fly with her, so I called back medical control in desperation: “This doctor’s willing to fly. Let him take her.”
They told me apologetically, knowing my agony, that he was not trained to fly and therefore could not do so. I sat down in the ambulance bay crying, waiting for the second helicopter to arrive.
When we got Ali onto AirCare 2, my nurse then told me I couldn’t fly with her. I said, “I’m flying with her.” She said, “no, it’s not safe.” I said, “I’m not leaving her. I’ll sit in the front. What do you think I’m going to do? Jump out of the helicopter?” I think they realized there was no other option that I would agree to. I rode up front.
It was the fastest flight to the trauma center that I had ever experienced. They did a hot offload, meaning they didn’t even shut down the blades. We got her to the trauma center. And then it was a whole other layer of chaos.
Dr. Alison Delgado: Taylor’s presence may have delayed my transfer, but the University emergency department was prepped and waiting for me. Radiology was on hold, surgery and neurosurgery were there waiting. Everyone was in the trauma bay.
Dr. Taylor Delgado: My younger sister was a social worker in that emergency department, and she was on shift. She and my residency director went to CT with Ali. As the images from Ali’s CT scan showed up on the screens, everyone in the room gasped. She had a nonsurvivable head injury.
The AirCare 2 doctor collapsed into our director’s arms and cried: “She’s going to die tonight.” He responded: “I know. But we’ve got work to do.” Then he asked my sister how close she was with me. She told him we were extremely close. “Good, because we have to break the news that she’s going to die tonight.”
But the doctor never told me. I was in the consultation room. He came in and told me that she had a lot of bleeding around the brain, but he couldn’t find the words to tell me the true severity. He didn’t have to.
Dr. Alison Delgado: I was in a coma for 5 days. Shift by shift, they were amazed that I was still there. I had a broken jaw, broken vertebrae in my spine, a broken clavicle and sternum and contusions to my heart and lungs. I was later found to have a dissection of my carotid artery as well as an aneurysm to the carotid artery. These were both caused by the accident.
My jaw was wired shut and a tracheostomy was placed. They coiled the aneurysm and put a stent in the dissection. I was placed on dual antiplatelet therapy to prevent stent thrombosis.
When I initially woke from the coma during my hospital stay, I could not speak, but I remember being told why I was there. My first two thoughts were: Was it my fault? and I need to get back to work.
Two and a half weeks later, I was stable enough to go to an in-patient rehab facility.
I was very motivated. I made a lot of good progress, because Taylor was there with me. We looked through pictures, trying to jog my memory and help with my vocabulary. I’d look at a bird and know this is a flying animal but couldn’t think of the word bird. I couldn’t remember my mom’s name.
Dr. Taylor Delgado: She was becoming more fluent with her speech each day. Her right arm was working more normally. We started going on walks outside. Within 14 days she was discharged home.
When we left the rehab facility, I took a couple extra tracheostomy tubes and supplies, because I didn’t know how long Ali would have her trach. The emergency medicine person in me just thought, always have these things on hand.
A few days later, her ENT doctor decannulated her tracheostomy tube. In our minds, we were done.
The next night, she had the intracranial hemorrhage.
Return to the hospital ...
Dr. Taylor Delgado: The aneurysm they had coiled had ruptured. Ali had a recurrent subarachnoid hemorrhage and an intracranial hemorrhage, and she was still bleeding. So, they took her to IR to try to embolize it and accomplished as much as they possibly could.
She had hydrocephalus, the ventricles in her brain were enlarged. Normally, they would put in a drain, but they couldn’t because she was on aspirin and Plavix (clopidogrel). That would risk her having a bleed around that insertion site, which would cause a brain hemorrhage.
Dr. Alison Delgado: I was like a ticking time bomb. We knew I would have to have surgery as soon as possible to open my skull and clip the aneurysm. But I had to be on the Plavix and aspirin for at least 6 weeks before it would be considered safe to discontinue them. It was another 3 weeks before they could proceed with the surgery.
The second hospitalization was scarier than the first, because I was much more aware. I knew that I might not be able to return to my residency and do the thing I had dreamed of doing. There were risks of me becoming blind or paralyzed during the surgery. I might not even leave the hospital.
Dr. Taylor Delgado: It was mid-December by then, and my dad asked her, “Ali, what do you want for Christmas?” She looked at him deadpan and said, “normal brain.”
Dr. Alison Delgado: The surgery was successful. I went home a few days later. But I’d lost everything I had gained in rehabilitation. My speech was back to square one.
None of the doctors really expected me to go back to work. But from my standpoint, I thought, I could have died the day I was hit. I could have died when the aneurysm ruptured, or at any point along the way. But I’m here and I’m going back to work.
Dr. Taylor Delgado: In January, I went back to work and I had to fly on the helicopter. They were worried about how I would react. My flight director flew with me on my first shift. Our first flight was an inter-facility STEMI transfer. No big deal. The second one was a car accident outside of Batesville, Ind. We were in the back of the ambulance, and I looked at this woman. She was 27 years old, thin, with long hair. She looked exactly like Ali.
Ali flashed into my mind, and I was like, nope. Ali’s at home. She’s fine. This person is right here, right now. Do what you do. I intubated her in the helicopter. We gave her hypertonic saline. I started a blood transfusion. Afterward, my flight director came up to me and said: “You’re released back to full duty. That was the hardest test you could possibly have on your first day back flying, and you nailed it.”
Dr. Alison Delgado: I finished my residency in December of 2012 and passed my pediatric board exam on the first try, almost exactly 3 years after my accident.
The spring before I started medical school in 2005, I had won the Cincinnati Flying Pig marathon. In 2011, a few months after my accident, they invited us to be the starters of the race. When we stood at the starting line, I decided right then I was going to run this marathon again the next year. In spring 2012, I returned and finished in fourth place, beating my previous winning time by two minutes.
I have a different level of empathy for my patients now. I know what it’s like to be scared. I know what it’s like to not know if you’re going to leave the hospital. I’ve lived that. The process of writing my book was also cathartic for me. I told my story to try to give people hope.
Dr. Taylor Delgado: I have a tattoo on my wrist showing the date of Ali’s accident. The idea was to remind myself of what we’ve come through and everyone who went above and beyond. To show gratitude to them and remember everything that they did for us. It’s also to remember that every patient I see is somebody else’s Alison.
A version of this article first appeared on Medscape.com.
Dr. Taylor Delgado: It was Saturday night, and we had just gone to bed. Suddenly, Ali sat up, and screamed, “My head!” She then became nonresponsive and had a seizure. I was in disbelief, but I also knew exactly what was happening. I called 911: “My wife is having a head bleed. I need an ambulance.” It was a bad connection, and they could barely understand me.
As I tried to carry Ali downstairs, she vomited. She still had rubber bands in her mouth from the jaw fracture that was a result of her accident just a month ago. I knew she needed an airway.
I grabbed a tracheostomy tube, but the opening over her trachea put in for the accident had since closed. I tried to push the tube through her neck, but it hurt her; her eyes opened.
I thought to myself: Maybe she doesn’t need it. This can wait until she gets to the hospital. I can’t do this to her. But she vomited again, and I knew what I had to do.
We were at the top of our stairs. I didn’t have a blade or any other equipment, just the tracheostomy tube with the dilator. I pushed hard, and she started fighting me. I had to hold her hands away with one arm. The tube popped in and she stared back at me in pain and fear.
I finally got her downstairs and called medical control at University Hospital of Cincinnati. I was able to speak with one of the attendings: “Ali’s aneurysm ruptured, and she just had a seizure. She has a GCS of 11 or 12. I replaced her tracheostomy tube. We’ll be there shortly.”
When I heard sirens come down our street, I carried Ali outside, but the sirens were from a firetruck. They likely assumed someone had fallen and had a head laceration. It was beyond deflating. I yelled incredulously: “We need an ambulance here now!”
When the ambulance finally arrived, they tried to tell me that I could not ride with them. Or if I did, I would have to sit up front. After arguing back and forth for a few seconds, I finally demanded: “This is medical control. This is MD-88, and this is my patient. I’m sitting in back with you. She needs four Zofran and two midazolam IV now.”
One month earlier ...
Dr. Alison Delgado: Taylor and I were both 4 months into our second year of residency, and we had been married for 5 months. I was a pediatric resident at Cincinnati Children’s Hospital. She was an emergency medicine resident at the University Hospital. I was having my first day off in a couple weeks, and she was working a shift in the emergency department. She was also a part of the flight crew that day. Second-year residents would go out to the scenes of accidents or to other hospitals to transport the patient back to their Level I trauma center via helicopter. The resident was the physician and considered the leader on these flights.
That afternoon, I went for a bicycle ride. About three-quarters of the way through my ride, I was struck by a car.
The EMS crew got to me fairly quickly. They intubated me at the scene and got me to the closest hospital. Immediately, the hospital realized my case was outside the scope of their care. They contacted University Hospital requesting that their flight crew come to transport me.
Dr. Taylor Delgado: At around 5:30 p.m. the day of my shift, the tones went out on the radio: “AirCare 1 and Pod Doc, you are requested for interhospital transfer, 27-year-old Jane Doe, GCS 5.” That was the only information given.
When we landed at the hospital, I walked in with my nurse. I was listening to the doctor’s report and doing my once over. The patient was a little bit bradycardic, heart rate in the 40s or 50s. Blood pressure was normal if not a little bit elevated. There was obvious facial trauma. The endotracheal tube in place.
She was covered with a blanket, but some of her clothing was visible. Suddenly, I recognized it. It was our cycling team’s kit. I thought, please don’t let it be Ali.
My flight nurse went out and called back to dispatch. “This is my doc’s wife. Dispatch the second helicopter!” She had to repeat herself a few times before they understood what was happening.
As Ali’s spouse, I couldn’t be the flight doctor. I didn’t care. I called medical control myself and told them: “This is Ali. We have to fly her. She has a head injury.” They said: “You can’t fly her.” I said: “We can’t delay her care. I have to fly her.” They said: “No, you can’t fly her.” I broke down. Devastated.
I went back into the room and looked at Ali. Her heart rate was dropping. My flight nurse was in the trauma bay with the emergency physician. We realized definitive care was being delayed because of my presence, which was an awful feeling to have. I think at that point we realized, you do nothing, or you act. So, we acted.
I told my flight nurse: “Let’s give her atropine to increase her heart rate.” I asked about sedation, and she hadn’t had anything. I spurted off some doses: “a hundred of fentanyl and five of midazolam.” My flight nurse actually administered smaller doses. She thought it was a bit aggressive, and she was correct. I was trying to maintain composure, but it was hard.
The emergency medicine physician volunteered to fly with her, so I called back medical control in desperation: “This doctor’s willing to fly. Let him take her.”
They told me apologetically, knowing my agony, that he was not trained to fly and therefore could not do so. I sat down in the ambulance bay crying, waiting for the second helicopter to arrive.
When we got Ali onto AirCare 2, my nurse then told me I couldn’t fly with her. I said, “I’m flying with her.” She said, “no, it’s not safe.” I said, “I’m not leaving her. I’ll sit in the front. What do you think I’m going to do? Jump out of the helicopter?” I think they realized there was no other option that I would agree to. I rode up front.
It was the fastest flight to the trauma center that I had ever experienced. They did a hot offload, meaning they didn’t even shut down the blades. We got her to the trauma center. And then it was a whole other layer of chaos.
Dr. Alison Delgado: Taylor’s presence may have delayed my transfer, but the University emergency department was prepped and waiting for me. Radiology was on hold, surgery and neurosurgery were there waiting. Everyone was in the trauma bay.
Dr. Taylor Delgado: My younger sister was a social worker in that emergency department, and she was on shift. She and my residency director went to CT with Ali. As the images from Ali’s CT scan showed up on the screens, everyone in the room gasped. She had a nonsurvivable head injury.
The AirCare 2 doctor collapsed into our director’s arms and cried: “She’s going to die tonight.” He responded: “I know. But we’ve got work to do.” Then he asked my sister how close she was with me. She told him we were extremely close. “Good, because we have to break the news that she’s going to die tonight.”
But the doctor never told me. I was in the consultation room. He came in and told me that she had a lot of bleeding around the brain, but he couldn’t find the words to tell me the true severity. He didn’t have to.
Dr. Alison Delgado: I was in a coma for 5 days. Shift by shift, they were amazed that I was still there. I had a broken jaw, broken vertebrae in my spine, a broken clavicle and sternum and contusions to my heart and lungs. I was later found to have a dissection of my carotid artery as well as an aneurysm to the carotid artery. These were both caused by the accident.
My jaw was wired shut and a tracheostomy was placed. They coiled the aneurysm and put a stent in the dissection. I was placed on dual antiplatelet therapy to prevent stent thrombosis.
When I initially woke from the coma during my hospital stay, I could not speak, but I remember being told why I was there. My first two thoughts were: Was it my fault? and I need to get back to work.
Two and a half weeks later, I was stable enough to go to an in-patient rehab facility.
I was very motivated. I made a lot of good progress, because Taylor was there with me. We looked through pictures, trying to jog my memory and help with my vocabulary. I’d look at a bird and know this is a flying animal but couldn’t think of the word bird. I couldn’t remember my mom’s name.
Dr. Taylor Delgado: She was becoming more fluent with her speech each day. Her right arm was working more normally. We started going on walks outside. Within 14 days she was discharged home.
When we left the rehab facility, I took a couple extra tracheostomy tubes and supplies, because I didn’t know how long Ali would have her trach. The emergency medicine person in me just thought, always have these things on hand.
A few days later, her ENT doctor decannulated her tracheostomy tube. In our minds, we were done.
The next night, she had the intracranial hemorrhage.
Return to the hospital ...
Dr. Taylor Delgado: The aneurysm they had coiled had ruptured. Ali had a recurrent subarachnoid hemorrhage and an intracranial hemorrhage, and she was still bleeding. So, they took her to IR to try to embolize it and accomplished as much as they possibly could.
She had hydrocephalus, the ventricles in her brain were enlarged. Normally, they would put in a drain, but they couldn’t because she was on aspirin and Plavix (clopidogrel). That would risk her having a bleed around that insertion site, which would cause a brain hemorrhage.
Dr. Alison Delgado: I was like a ticking time bomb. We knew I would have to have surgery as soon as possible to open my skull and clip the aneurysm. But I had to be on the Plavix and aspirin for at least 6 weeks before it would be considered safe to discontinue them. It was another 3 weeks before they could proceed with the surgery.
The second hospitalization was scarier than the first, because I was much more aware. I knew that I might not be able to return to my residency and do the thing I had dreamed of doing. There were risks of me becoming blind or paralyzed during the surgery. I might not even leave the hospital.
Dr. Taylor Delgado: It was mid-December by then, and my dad asked her, “Ali, what do you want for Christmas?” She looked at him deadpan and said, “normal brain.”
Dr. Alison Delgado: The surgery was successful. I went home a few days later. But I’d lost everything I had gained in rehabilitation. My speech was back to square one.
None of the doctors really expected me to go back to work. But from my standpoint, I thought, I could have died the day I was hit. I could have died when the aneurysm ruptured, or at any point along the way. But I’m here and I’m going back to work.
Dr. Taylor Delgado: In January, I went back to work and I had to fly on the helicopter. They were worried about how I would react. My flight director flew with me on my first shift. Our first flight was an inter-facility STEMI transfer. No big deal. The second one was a car accident outside of Batesville, Ind. We were in the back of the ambulance, and I looked at this woman. She was 27 years old, thin, with long hair. She looked exactly like Ali.
Ali flashed into my mind, and I was like, nope. Ali’s at home. She’s fine. This person is right here, right now. Do what you do. I intubated her in the helicopter. We gave her hypertonic saline. I started a blood transfusion. Afterward, my flight director came up to me and said: “You’re released back to full duty. That was the hardest test you could possibly have on your first day back flying, and you nailed it.”
Dr. Alison Delgado: I finished my residency in December of 2012 and passed my pediatric board exam on the first try, almost exactly 3 years after my accident.
The spring before I started medical school in 2005, I had won the Cincinnati Flying Pig marathon. In 2011, a few months after my accident, they invited us to be the starters of the race. When we stood at the starting line, I decided right then I was going to run this marathon again the next year. In spring 2012, I returned and finished in fourth place, beating my previous winning time by two minutes.
I have a different level of empathy for my patients now. I know what it’s like to be scared. I know what it’s like to not know if you’re going to leave the hospital. I’ve lived that. The process of writing my book was also cathartic for me. I told my story to try to give people hope.
Dr. Taylor Delgado: I have a tattoo on my wrist showing the date of Ali’s accident. The idea was to remind myself of what we’ve come through and everyone who went above and beyond. To show gratitude to them and remember everything that they did for us. It’s also to remember that every patient I see is somebody else’s Alison.
A version of this article first appeared on Medscape.com.
Dr. Taylor Delgado: It was Saturday night, and we had just gone to bed. Suddenly, Ali sat up, and screamed, “My head!” She then became nonresponsive and had a seizure. I was in disbelief, but I also knew exactly what was happening. I called 911: “My wife is having a head bleed. I need an ambulance.” It was a bad connection, and they could barely understand me.
As I tried to carry Ali downstairs, she vomited. She still had rubber bands in her mouth from the jaw fracture that was a result of her accident just a month ago. I knew she needed an airway.
I grabbed a tracheostomy tube, but the opening over her trachea put in for the accident had since closed. I tried to push the tube through her neck, but it hurt her; her eyes opened.
I thought to myself: Maybe she doesn’t need it. This can wait until she gets to the hospital. I can’t do this to her. But she vomited again, and I knew what I had to do.
We were at the top of our stairs. I didn’t have a blade or any other equipment, just the tracheostomy tube with the dilator. I pushed hard, and she started fighting me. I had to hold her hands away with one arm. The tube popped in and she stared back at me in pain and fear.
I finally got her downstairs and called medical control at University Hospital of Cincinnati. I was able to speak with one of the attendings: “Ali’s aneurysm ruptured, and she just had a seizure. She has a GCS of 11 or 12. I replaced her tracheostomy tube. We’ll be there shortly.”
When I heard sirens come down our street, I carried Ali outside, but the sirens were from a firetruck. They likely assumed someone had fallen and had a head laceration. It was beyond deflating. I yelled incredulously: “We need an ambulance here now!”
When the ambulance finally arrived, they tried to tell me that I could not ride with them. Or if I did, I would have to sit up front. After arguing back and forth for a few seconds, I finally demanded: “This is medical control. This is MD-88, and this is my patient. I’m sitting in back with you. She needs four Zofran and two midazolam IV now.”
One month earlier ...
Dr. Alison Delgado: Taylor and I were both 4 months into our second year of residency, and we had been married for 5 months. I was a pediatric resident at Cincinnati Children’s Hospital. She was an emergency medicine resident at the University Hospital. I was having my first day off in a couple weeks, and she was working a shift in the emergency department. She was also a part of the flight crew that day. Second-year residents would go out to the scenes of accidents or to other hospitals to transport the patient back to their Level I trauma center via helicopter. The resident was the physician and considered the leader on these flights.
That afternoon, I went for a bicycle ride. About three-quarters of the way through my ride, I was struck by a car.
The EMS crew got to me fairly quickly. They intubated me at the scene and got me to the closest hospital. Immediately, the hospital realized my case was outside the scope of their care. They contacted University Hospital requesting that their flight crew come to transport me.
Dr. Taylor Delgado: At around 5:30 p.m. the day of my shift, the tones went out on the radio: “AirCare 1 and Pod Doc, you are requested for interhospital transfer, 27-year-old Jane Doe, GCS 5.” That was the only information given.
When we landed at the hospital, I walked in with my nurse. I was listening to the doctor’s report and doing my once over. The patient was a little bit bradycardic, heart rate in the 40s or 50s. Blood pressure was normal if not a little bit elevated. There was obvious facial trauma. The endotracheal tube in place.
She was covered with a blanket, but some of her clothing was visible. Suddenly, I recognized it. It was our cycling team’s kit. I thought, please don’t let it be Ali.
My flight nurse went out and called back to dispatch. “This is my doc’s wife. Dispatch the second helicopter!” She had to repeat herself a few times before they understood what was happening.
As Ali’s spouse, I couldn’t be the flight doctor. I didn’t care. I called medical control myself and told them: “This is Ali. We have to fly her. She has a head injury.” They said: “You can’t fly her.” I said: “We can’t delay her care. I have to fly her.” They said: “No, you can’t fly her.” I broke down. Devastated.
I went back into the room and looked at Ali. Her heart rate was dropping. My flight nurse was in the trauma bay with the emergency physician. We realized definitive care was being delayed because of my presence, which was an awful feeling to have. I think at that point we realized, you do nothing, or you act. So, we acted.
I told my flight nurse: “Let’s give her atropine to increase her heart rate.” I asked about sedation, and she hadn’t had anything. I spurted off some doses: “a hundred of fentanyl and five of midazolam.” My flight nurse actually administered smaller doses. She thought it was a bit aggressive, and she was correct. I was trying to maintain composure, but it was hard.
The emergency medicine physician volunteered to fly with her, so I called back medical control in desperation: “This doctor’s willing to fly. Let him take her.”
They told me apologetically, knowing my agony, that he was not trained to fly and therefore could not do so. I sat down in the ambulance bay crying, waiting for the second helicopter to arrive.
When we got Ali onto AirCare 2, my nurse then told me I couldn’t fly with her. I said, “I’m flying with her.” She said, “no, it’s not safe.” I said, “I’m not leaving her. I’ll sit in the front. What do you think I’m going to do? Jump out of the helicopter?” I think they realized there was no other option that I would agree to. I rode up front.
It was the fastest flight to the trauma center that I had ever experienced. They did a hot offload, meaning they didn’t even shut down the blades. We got her to the trauma center. And then it was a whole other layer of chaos.
Dr. Alison Delgado: Taylor’s presence may have delayed my transfer, but the University emergency department was prepped and waiting for me. Radiology was on hold, surgery and neurosurgery were there waiting. Everyone was in the trauma bay.
Dr. Taylor Delgado: My younger sister was a social worker in that emergency department, and she was on shift. She and my residency director went to CT with Ali. As the images from Ali’s CT scan showed up on the screens, everyone in the room gasped. She had a nonsurvivable head injury.
The AirCare 2 doctor collapsed into our director’s arms and cried: “She’s going to die tonight.” He responded: “I know. But we’ve got work to do.” Then he asked my sister how close she was with me. She told him we were extremely close. “Good, because we have to break the news that she’s going to die tonight.”
But the doctor never told me. I was in the consultation room. He came in and told me that she had a lot of bleeding around the brain, but he couldn’t find the words to tell me the true severity. He didn’t have to.
Dr. Alison Delgado: I was in a coma for 5 days. Shift by shift, they were amazed that I was still there. I had a broken jaw, broken vertebrae in my spine, a broken clavicle and sternum and contusions to my heart and lungs. I was later found to have a dissection of my carotid artery as well as an aneurysm to the carotid artery. These were both caused by the accident.
My jaw was wired shut and a tracheostomy was placed. They coiled the aneurysm and put a stent in the dissection. I was placed on dual antiplatelet therapy to prevent stent thrombosis.
When I initially woke from the coma during my hospital stay, I could not speak, but I remember being told why I was there. My first two thoughts were: Was it my fault? and I need to get back to work.
Two and a half weeks later, I was stable enough to go to an in-patient rehab facility.
I was very motivated. I made a lot of good progress, because Taylor was there with me. We looked through pictures, trying to jog my memory and help with my vocabulary. I’d look at a bird and know this is a flying animal but couldn’t think of the word bird. I couldn’t remember my mom’s name.
Dr. Taylor Delgado: She was becoming more fluent with her speech each day. Her right arm was working more normally. We started going on walks outside. Within 14 days she was discharged home.
When we left the rehab facility, I took a couple extra tracheostomy tubes and supplies, because I didn’t know how long Ali would have her trach. The emergency medicine person in me just thought, always have these things on hand.
A few days later, her ENT doctor decannulated her tracheostomy tube. In our minds, we were done.
The next night, she had the intracranial hemorrhage.
Return to the hospital ...
Dr. Taylor Delgado: The aneurysm they had coiled had ruptured. Ali had a recurrent subarachnoid hemorrhage and an intracranial hemorrhage, and she was still bleeding. So, they took her to IR to try to embolize it and accomplished as much as they possibly could.
She had hydrocephalus, the ventricles in her brain were enlarged. Normally, they would put in a drain, but they couldn’t because she was on aspirin and Plavix (clopidogrel). That would risk her having a bleed around that insertion site, which would cause a brain hemorrhage.
Dr. Alison Delgado: I was like a ticking time bomb. We knew I would have to have surgery as soon as possible to open my skull and clip the aneurysm. But I had to be on the Plavix and aspirin for at least 6 weeks before it would be considered safe to discontinue them. It was another 3 weeks before they could proceed with the surgery.
The second hospitalization was scarier than the first, because I was much more aware. I knew that I might not be able to return to my residency and do the thing I had dreamed of doing. There were risks of me becoming blind or paralyzed during the surgery. I might not even leave the hospital.
Dr. Taylor Delgado: It was mid-December by then, and my dad asked her, “Ali, what do you want for Christmas?” She looked at him deadpan and said, “normal brain.”
Dr. Alison Delgado: The surgery was successful. I went home a few days later. But I’d lost everything I had gained in rehabilitation. My speech was back to square one.
None of the doctors really expected me to go back to work. But from my standpoint, I thought, I could have died the day I was hit. I could have died when the aneurysm ruptured, or at any point along the way. But I’m here and I’m going back to work.
Dr. Taylor Delgado: In January, I went back to work and I had to fly on the helicopter. They were worried about how I would react. My flight director flew with me on my first shift. Our first flight was an inter-facility STEMI transfer. No big deal. The second one was a car accident outside of Batesville, Ind. We were in the back of the ambulance, and I looked at this woman. She was 27 years old, thin, with long hair. She looked exactly like Ali.
Ali flashed into my mind, and I was like, nope. Ali’s at home. She’s fine. This person is right here, right now. Do what you do. I intubated her in the helicopter. We gave her hypertonic saline. I started a blood transfusion. Afterward, my flight director came up to me and said: “You’re released back to full duty. That was the hardest test you could possibly have on your first day back flying, and you nailed it.”
Dr. Alison Delgado: I finished my residency in December of 2012 and passed my pediatric board exam on the first try, almost exactly 3 years after my accident.
The spring before I started medical school in 2005, I had won the Cincinnati Flying Pig marathon. In 2011, a few months after my accident, they invited us to be the starters of the race. When we stood at the starting line, I decided right then I was going to run this marathon again the next year. In spring 2012, I returned and finished in fourth place, beating my previous winning time by two minutes.
I have a different level of empathy for my patients now. I know what it’s like to be scared. I know what it’s like to not know if you’re going to leave the hospital. I’ve lived that. The process of writing my book was also cathartic for me. I told my story to try to give people hope.
Dr. Taylor Delgado: I have a tattoo on my wrist showing the date of Ali’s accident. The idea was to remind myself of what we’ve come through and everyone who went above and beyond. To show gratitude to them and remember everything that they did for us. It’s also to remember that every patient I see is somebody else’s Alison.
A version of this article first appeared on Medscape.com.
Severe strep infections rebound after pandemic lull
Severe infections caused by group A streptococcus bacteria are on the rise in countries around the world, including the United States, according to new data from the Centers for Disease Control and Prevention.
Group A strep bacteria usually cause mild illnesses like strep throat and scarlet fever. But they can also cause more severe diseases, like the flesh-eating disease necrotizing fasciitis and streptococcal toxic shock syndrome, known as invasive group A strep infections.
These infections fell by 25% during the COVID-19 pandemic and were especially low in children. The number of milder infections also dropped. But in 2022, severe infections came roaring back, particularly in children.
such as Colorado and Minnesota.
Now in 2023, invasive infections are high in children in some parts of the country, even after respiratory viruses like the flu and respiratory syncytial virus (RSV) decreased in those areas. Some parts of the country also saw high rates of invasive infections in older adults.
Less severe strep A infections in children have returned to levels similar to or higher than those seen in prepandemic years.
A similar postpandemic resurgence in invasive infections has also been seen in other countries, including Canada, the United Kingdom, France, and Denmark.
Strep A is a very common bacteria that causes only mild or no symptoms in most people, and severe infections are usually quite rare. They tend to affect the most vulnerable people: those who have another virus, multiple chronic conditions, or an open wound.
People should watch for fever, headaches, or confusion during a strep infection, which all might signal a more severe illness.
A version of this article first appeared on Medscape.com.
Severe infections caused by group A streptococcus bacteria are on the rise in countries around the world, including the United States, according to new data from the Centers for Disease Control and Prevention.
Group A strep bacteria usually cause mild illnesses like strep throat and scarlet fever. But they can also cause more severe diseases, like the flesh-eating disease necrotizing fasciitis and streptococcal toxic shock syndrome, known as invasive group A strep infections.
These infections fell by 25% during the COVID-19 pandemic and were especially low in children. The number of milder infections also dropped. But in 2022, severe infections came roaring back, particularly in children.
such as Colorado and Minnesota.
Now in 2023, invasive infections are high in children in some parts of the country, even after respiratory viruses like the flu and respiratory syncytial virus (RSV) decreased in those areas. Some parts of the country also saw high rates of invasive infections in older adults.
Less severe strep A infections in children have returned to levels similar to or higher than those seen in prepandemic years.
A similar postpandemic resurgence in invasive infections has also been seen in other countries, including Canada, the United Kingdom, France, and Denmark.
Strep A is a very common bacteria that causes only mild or no symptoms in most people, and severe infections are usually quite rare. They tend to affect the most vulnerable people: those who have another virus, multiple chronic conditions, or an open wound.
People should watch for fever, headaches, or confusion during a strep infection, which all might signal a more severe illness.
A version of this article first appeared on Medscape.com.
Severe infections caused by group A streptococcus bacteria are on the rise in countries around the world, including the United States, according to new data from the Centers for Disease Control and Prevention.
Group A strep bacteria usually cause mild illnesses like strep throat and scarlet fever. But they can also cause more severe diseases, like the flesh-eating disease necrotizing fasciitis and streptococcal toxic shock syndrome, known as invasive group A strep infections.
These infections fell by 25% during the COVID-19 pandemic and were especially low in children. The number of milder infections also dropped. But in 2022, severe infections came roaring back, particularly in children.
such as Colorado and Minnesota.
Now in 2023, invasive infections are high in children in some parts of the country, even after respiratory viruses like the flu and respiratory syncytial virus (RSV) decreased in those areas. Some parts of the country also saw high rates of invasive infections in older adults.
Less severe strep A infections in children have returned to levels similar to or higher than those seen in prepandemic years.
A similar postpandemic resurgence in invasive infections has also been seen in other countries, including Canada, the United Kingdom, France, and Denmark.
Strep A is a very common bacteria that causes only mild or no symptoms in most people, and severe infections are usually quite rare. They tend to affect the most vulnerable people: those who have another virus, multiple chronic conditions, or an open wound.
People should watch for fever, headaches, or confusion during a strep infection, which all might signal a more severe illness.
A version of this article first appeared on Medscape.com.
Low-calorie tastes sweeter with a little salt
Low-calorie tastes sweeter with a little salt
Diet and sugar-free foods and drinks seem like a good idea, but it’s hard to get past that strange aftertaste, right? It’s the calling card for the noncaloric aspartame- and stevia-containing sweeteners that we consume to make us feel like we can have the best of both worlds.
That weird lingering taste can be a total turn-off for some (raises hand), but researchers have found an almost facepalm solution to the not-so-sweet problem, and it’s salt.
Now, the concept of sweet and salty is not a far-fetched partnership when it comes to snack consumption (try M&Ms in your popcorn). The researchers at Almendra, a manufacturer of stevia sweeteners, put that iconic flavor pair to the test by adding mineral salts that have some nutritional value to lessen the effect of a stevia compound, rebaudioside A, found in noncaloric sweeteners.
The researchers added in magnesium chloride, calcium chloride, and potassium chloride separately to lessen rebaudioside A’s intensity, but they needed so much salt that it killed the sweet taste completely. A blend of the three mineral salts, however, reduced the lingering taste by 79% and improved the real sugar-like taste. The researchers tried this blend in reduced-calorie orange juice and a citrus-flavored soft drink, improving the taste in both.
The salty and sweet match comes in for the win once again. This time helping against the fight of obesity instead of making it worse.
Pseudomonas’ Achilles’ heel is more of an Achilles’ genetic switch
Today, on the long-awaited return of “Bacteria vs. the World,” we meet one of the rock stars of infectious disease.
LOTME: Through the use of imaginary technology, we’re talking to Pseudomonas aeruginosa. Thanks for joining us on such short notice, after Neisseria gonorrhoeae canceled at the last minute.
P. aeruginosa: No problem. I think we can all guess what that little devil is up to.
LOTME: Bacterial resistance to antibiotics is a huge problem for our species. What makes you so hard to fight?
P. aeruginosa: We’ve been trying to keep that a secret, actually, but now that researchers in Switzerland and Denmark seem to have figured it out, I guess it’s okay for me to spill the beans.
LOTME: Beans? What do beans have to do with it?
P. aeruginosa: Nothing, it’s just a colloquial expression that means I’m sharing previously private information.
LOTME: Sure, we knew that. Please, continue your spilling.
P. aeruginosa: The secret is … Well, let’s just say we were a little worried when the Clash released “Should I Stay or Should I Go” back in the 1980s.
LOTME: The Clash? Now we’re really confused.
P. aeruginosa: The answer to their question, “Should I stay or should I go? is yes. Successful invasion of a human is all about division of labor. “While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites,” is how the investigators described it. We can increase surface colonization by using a “job-sharing” process, they said, and even resist antibiotics because most of us remain in the protective biofilm.
LOTME: And they say you guys don’t have brains.
P. aeruginosa: But wait, there’s more. We don’t just divide the labor randomly. After the initial colonization we form two functionally distinct subpopulations. One has high levels of the bacterial signaling molecule c-di-GMP and stays put to work on the biofilm. The other group, with low levels of c-di-GMP, heads out to the surrounding tissue to continue the colonization. As project leader Urs Jenal put it, “By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
LOTME: Pretty clever stuff, for humans, anyway.
P. aeruginosa: We agree, but now that you know our secret, we can’t let you share it.
LOTME: Wait! The journal article’s already been published. Your secret is out. You can’t stop that by infecting me.
P. aeruginosa: True enough, but are you familiar with the fable of the scorpion and the frog? It’s our nature.
LOTME: Nooooo! N. gonorrhoeae wouldn’t have done this!
What a pain in the Butt
Businesses rise and businesses fall. We all know that one cursed location, that spot in town where we see businesses move in and close up in a matter of months. At the same time, though, there are also businesses that have been around as long as anyone can remember, pillars of the community.
Corydon, IN., likely has a few such long-lived shops, but it is officially down one 70-year-old family business as of late April, with the unfortunate passing of beloved local pharmacy Butt Drugs. Prescription pick-up in rear.
The business dates back to 1952, when it was founded as William H. Butt Drugs. We’re sure William Butt was never teased about his last name. Nope. No one would ever do that. After he passed the store to his children, it underwent a stint as Butt Rexall Drugs. When the shop was passed down to its third-generation and ultimately final owner, Katie Butt Beckort, she decided to simplify the name. Get right down to the bottom of things, as it were.
Butt Drugs was a popular spot, featuring an old-school soda fountain and themed souvenirs. According to Ms. Butt Beckort, people would come from miles away to buy “I love Butt Drugs” T-shirts, magnets, and so on. Yes, they knew perfectly well what they were sitting on.
So, if was such a hit, why did it close? Butt Drugs may have a hilarious name and merchandise to match, but the pharmacy portion of the pharmacy had been losing money for years. You know, the actual point of the business. As with so many things, we can blame it on the insurance companies. More than half the drugs that passed through Butt Drugs’ doors were sold at a loss, because the insurance companies refused to reimburse the store more than the wholesale price of the drug. Not even a good butt drug could clear up that financial diarrhea.
And so, we’ve lost Butt Drugs forever. Spicy food enthusiasts, coffee drinkers, and all patrons of Taco Bell, take a moment to reflect and mourn on what you’ve lost. No more Butt Drugs to relieve your suffering. A true kick in the butt indeed.
Low-calorie tastes sweeter with a little salt
Diet and sugar-free foods and drinks seem like a good idea, but it’s hard to get past that strange aftertaste, right? It’s the calling card for the noncaloric aspartame- and stevia-containing sweeteners that we consume to make us feel like we can have the best of both worlds.
That weird lingering taste can be a total turn-off for some (raises hand), but researchers have found an almost facepalm solution to the not-so-sweet problem, and it’s salt.
Now, the concept of sweet and salty is not a far-fetched partnership when it comes to snack consumption (try M&Ms in your popcorn). The researchers at Almendra, a manufacturer of stevia sweeteners, put that iconic flavor pair to the test by adding mineral salts that have some nutritional value to lessen the effect of a stevia compound, rebaudioside A, found in noncaloric sweeteners.
The researchers added in magnesium chloride, calcium chloride, and potassium chloride separately to lessen rebaudioside A’s intensity, but they needed so much salt that it killed the sweet taste completely. A blend of the three mineral salts, however, reduced the lingering taste by 79% and improved the real sugar-like taste. The researchers tried this blend in reduced-calorie orange juice and a citrus-flavored soft drink, improving the taste in both.
The salty and sweet match comes in for the win once again. This time helping against the fight of obesity instead of making it worse.
Pseudomonas’ Achilles’ heel is more of an Achilles’ genetic switch
Today, on the long-awaited return of “Bacteria vs. the World,” we meet one of the rock stars of infectious disease.
LOTME: Through the use of imaginary technology, we’re talking to Pseudomonas aeruginosa. Thanks for joining us on such short notice, after Neisseria gonorrhoeae canceled at the last minute.
P. aeruginosa: No problem. I think we can all guess what that little devil is up to.
LOTME: Bacterial resistance to antibiotics is a huge problem for our species. What makes you so hard to fight?
P. aeruginosa: We’ve been trying to keep that a secret, actually, but now that researchers in Switzerland and Denmark seem to have figured it out, I guess it’s okay for me to spill the beans.
LOTME: Beans? What do beans have to do with it?
P. aeruginosa: Nothing, it’s just a colloquial expression that means I’m sharing previously private information.
LOTME: Sure, we knew that. Please, continue your spilling.
P. aeruginosa: The secret is … Well, let’s just say we were a little worried when the Clash released “Should I Stay or Should I Go” back in the 1980s.
LOTME: The Clash? Now we’re really confused.
P. aeruginosa: The answer to their question, “Should I stay or should I go? is yes. Successful invasion of a human is all about division of labor. “While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites,” is how the investigators described it. We can increase surface colonization by using a “job-sharing” process, they said, and even resist antibiotics because most of us remain in the protective biofilm.
LOTME: And they say you guys don’t have brains.
P. aeruginosa: But wait, there’s more. We don’t just divide the labor randomly. After the initial colonization we form two functionally distinct subpopulations. One has high levels of the bacterial signaling molecule c-di-GMP and stays put to work on the biofilm. The other group, with low levels of c-di-GMP, heads out to the surrounding tissue to continue the colonization. As project leader Urs Jenal put it, “By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
LOTME: Pretty clever stuff, for humans, anyway.
P. aeruginosa: We agree, but now that you know our secret, we can’t let you share it.
LOTME: Wait! The journal article’s already been published. Your secret is out. You can’t stop that by infecting me.
P. aeruginosa: True enough, but are you familiar with the fable of the scorpion and the frog? It’s our nature.
LOTME: Nooooo! N. gonorrhoeae wouldn’t have done this!
What a pain in the Butt
Businesses rise and businesses fall. We all know that one cursed location, that spot in town where we see businesses move in and close up in a matter of months. At the same time, though, there are also businesses that have been around as long as anyone can remember, pillars of the community.
Corydon, IN., likely has a few such long-lived shops, but it is officially down one 70-year-old family business as of late April, with the unfortunate passing of beloved local pharmacy Butt Drugs. Prescription pick-up in rear.
The business dates back to 1952, when it was founded as William H. Butt Drugs. We’re sure William Butt was never teased about his last name. Nope. No one would ever do that. After he passed the store to his children, it underwent a stint as Butt Rexall Drugs. When the shop was passed down to its third-generation and ultimately final owner, Katie Butt Beckort, she decided to simplify the name. Get right down to the bottom of things, as it were.
Butt Drugs was a popular spot, featuring an old-school soda fountain and themed souvenirs. According to Ms. Butt Beckort, people would come from miles away to buy “I love Butt Drugs” T-shirts, magnets, and so on. Yes, they knew perfectly well what they were sitting on.
So, if was such a hit, why did it close? Butt Drugs may have a hilarious name and merchandise to match, but the pharmacy portion of the pharmacy had been losing money for years. You know, the actual point of the business. As with so many things, we can blame it on the insurance companies. More than half the drugs that passed through Butt Drugs’ doors were sold at a loss, because the insurance companies refused to reimburse the store more than the wholesale price of the drug. Not even a good butt drug could clear up that financial diarrhea.
And so, we’ve lost Butt Drugs forever. Spicy food enthusiasts, coffee drinkers, and all patrons of Taco Bell, take a moment to reflect and mourn on what you’ve lost. No more Butt Drugs to relieve your suffering. A true kick in the butt indeed.
Low-calorie tastes sweeter with a little salt
Diet and sugar-free foods and drinks seem like a good idea, but it’s hard to get past that strange aftertaste, right? It’s the calling card for the noncaloric aspartame- and stevia-containing sweeteners that we consume to make us feel like we can have the best of both worlds.
That weird lingering taste can be a total turn-off for some (raises hand), but researchers have found an almost facepalm solution to the not-so-sweet problem, and it’s salt.
Now, the concept of sweet and salty is not a far-fetched partnership when it comes to snack consumption (try M&Ms in your popcorn). The researchers at Almendra, a manufacturer of stevia sweeteners, put that iconic flavor pair to the test by adding mineral salts that have some nutritional value to lessen the effect of a stevia compound, rebaudioside A, found in noncaloric sweeteners.
The researchers added in magnesium chloride, calcium chloride, and potassium chloride separately to lessen rebaudioside A’s intensity, but they needed so much salt that it killed the sweet taste completely. A blend of the three mineral salts, however, reduced the lingering taste by 79% and improved the real sugar-like taste. The researchers tried this blend in reduced-calorie orange juice and a citrus-flavored soft drink, improving the taste in both.
The salty and sweet match comes in for the win once again. This time helping against the fight of obesity instead of making it worse.
Pseudomonas’ Achilles’ heel is more of an Achilles’ genetic switch
Today, on the long-awaited return of “Bacteria vs. the World,” we meet one of the rock stars of infectious disease.
LOTME: Through the use of imaginary technology, we’re talking to Pseudomonas aeruginosa. Thanks for joining us on such short notice, after Neisseria gonorrhoeae canceled at the last minute.
P. aeruginosa: No problem. I think we can all guess what that little devil is up to.
LOTME: Bacterial resistance to antibiotics is a huge problem for our species. What makes you so hard to fight?
P. aeruginosa: We’ve been trying to keep that a secret, actually, but now that researchers in Switzerland and Denmark seem to have figured it out, I guess it’s okay for me to spill the beans.
LOTME: Beans? What do beans have to do with it?
P. aeruginosa: Nothing, it’s just a colloquial expression that means I’m sharing previously private information.
LOTME: Sure, we knew that. Please, continue your spilling.
P. aeruginosa: The secret is … Well, let’s just say we were a little worried when the Clash released “Should I Stay or Should I Go” back in the 1980s.
LOTME: The Clash? Now we’re really confused.
P. aeruginosa: The answer to their question, “Should I stay or should I go? is yes. Successful invasion of a human is all about division of labor. “While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites,” is how the investigators described it. We can increase surface colonization by using a “job-sharing” process, they said, and even resist antibiotics because most of us remain in the protective biofilm.
LOTME: And they say you guys don’t have brains.
P. aeruginosa: But wait, there’s more. We don’t just divide the labor randomly. After the initial colonization we form two functionally distinct subpopulations. One has high levels of the bacterial signaling molecule c-di-GMP and stays put to work on the biofilm. The other group, with low levels of c-di-GMP, heads out to the surrounding tissue to continue the colonization. As project leader Urs Jenal put it, “By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
LOTME: Pretty clever stuff, for humans, anyway.
P. aeruginosa: We agree, but now that you know our secret, we can’t let you share it.
LOTME: Wait! The journal article’s already been published. Your secret is out. You can’t stop that by infecting me.
P. aeruginosa: True enough, but are you familiar with the fable of the scorpion and the frog? It’s our nature.
LOTME: Nooooo! N. gonorrhoeae wouldn’t have done this!
What a pain in the Butt
Businesses rise and businesses fall. We all know that one cursed location, that spot in town where we see businesses move in and close up in a matter of months. At the same time, though, there are also businesses that have been around as long as anyone can remember, pillars of the community.
Corydon, IN., likely has a few such long-lived shops, but it is officially down one 70-year-old family business as of late April, with the unfortunate passing of beloved local pharmacy Butt Drugs. Prescription pick-up in rear.
The business dates back to 1952, when it was founded as William H. Butt Drugs. We’re sure William Butt was never teased about his last name. Nope. No one would ever do that. After he passed the store to his children, it underwent a stint as Butt Rexall Drugs. When the shop was passed down to its third-generation and ultimately final owner, Katie Butt Beckort, she decided to simplify the name. Get right down to the bottom of things, as it were.
Butt Drugs was a popular spot, featuring an old-school soda fountain and themed souvenirs. According to Ms. Butt Beckort, people would come from miles away to buy “I love Butt Drugs” T-shirts, magnets, and so on. Yes, they knew perfectly well what they were sitting on.
So, if was such a hit, why did it close? Butt Drugs may have a hilarious name and merchandise to match, but the pharmacy portion of the pharmacy had been losing money for years. You know, the actual point of the business. As with so many things, we can blame it on the insurance companies. More than half the drugs that passed through Butt Drugs’ doors were sold at a loss, because the insurance companies refused to reimburse the store more than the wholesale price of the drug. Not even a good butt drug could clear up that financial diarrhea.
And so, we’ve lost Butt Drugs forever. Spicy food enthusiasts, coffee drinkers, and all patrons of Taco Bell, take a moment to reflect and mourn on what you’ve lost. No more Butt Drugs to relieve your suffering. A true kick in the butt indeed.
Should you have a chaperone in the exam room? Many say yes
This transcript has been edited for clarity.
I’m Art Caplan, PhD. I’m at the division of medical ethics at NYU’s Grossman School of Medicine.
, such as breasts, genitalia, and the perianal area.
In some institutions, there has been a movement toward saying a chaperone must be present, that it’s mandatory. I know that is true at Yale’s health care centers and clinics. Others do so when the patient requests it. An interesting situation sometimes occurs when the hospital or the clinic requires a chaperone but the patient says, “I don’t want a chaperone. I want my privacy. I want the gynecologist or the urologist only. I don’t want anyone else to be seeing me. I’m not comfortable with anyone other than the doctor in the room.”
Complicating this issue of when is a chaperone appropriate and when can it be refused, if ever, is the fact that the role of chaperone is ill defined. For example, there isn’t really agreement on who can be a chaperone. Could it be a medical student? Could it be a nurse? Could it be another doctor? Should it be someone who at least has finished nursing school or medical school? Can it be a patient representative? There are no standards about who can play the role.
Should the chaperone be available to be seen when they’re in the room? Should they stay behind a curtain or somewhere where they’re not, so to speak, intrusive into what’s going on in the exam room? Do they sit in a chair? Do they stand? How do they behave, if you will? There’s no agreement.
There’s still no agreement on the training that a chaperone should have. Do we charge them with trying to represent what’s going on with the patient or trying to protect the doctor against any accusations that are ill founded about inappropriate conduct? Are they supposed to do both? How do they obtain consent, if they do, from the patient undergoing an examination in a sensitive part of their body or one that they’re sensitive about?
This area really requires some hard thinking if you’re considering having chaperones present. I think there are some online courses that offer some training. I haven’t looked at them, but they might be worth a look to see if they make you more comfortable about getting a chaperone oriented. I think it’s probably important to set a policy saying a chaperone must always be present for these kinds of examinations and list them, or one can be requested no matter what is going on in terms of the kind of exam being conducted.
There needs to be some statement saying that you have permission to either accept them or refuse them – or you don’t. Should they always be present, for example, with patients who are minors, adolescents or children? Does that extend that far out where a guardian, parent, or someone has to give permission?
In this area, I think we can all understand why chaperones have come to the fore, including allegations of misconduct and inappropriate touching, and considering comfort levels of patients to just put them more at ease. It’s obvious that we haven’t, as a nation or a medical profession, thought it through to the degree to which we have to.
I’m certainly not anti-chaperone, and I believe that if patients are more comfortable having one present, or a doctor is more comfortable having one present, or if we all agree that there are certain patients – kids – where certain types of examinations require or ought to expect the chaperone to be present, that’s wonderful.
We’ve got to lay out the rights of the doctors. We’ve got to lay out the rights of the institutions. We’ve got to lay out the rights of the patients. We should agree on who these people are. We should agree on how they’re trained.
We’ve got some work ahead of us if we’re going to have chaperones become a standard part of the medical examination.
Dr. Kaplan reported conflicts of interest with the Franklin Institute, Tengion, Biogen Idec, Johnson & Johnson, and PriCara.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Art Caplan, PhD. I’m at the division of medical ethics at NYU’s Grossman School of Medicine.
, such as breasts, genitalia, and the perianal area.
In some institutions, there has been a movement toward saying a chaperone must be present, that it’s mandatory. I know that is true at Yale’s health care centers and clinics. Others do so when the patient requests it. An interesting situation sometimes occurs when the hospital or the clinic requires a chaperone but the patient says, “I don’t want a chaperone. I want my privacy. I want the gynecologist or the urologist only. I don’t want anyone else to be seeing me. I’m not comfortable with anyone other than the doctor in the room.”
Complicating this issue of when is a chaperone appropriate and when can it be refused, if ever, is the fact that the role of chaperone is ill defined. For example, there isn’t really agreement on who can be a chaperone. Could it be a medical student? Could it be a nurse? Could it be another doctor? Should it be someone who at least has finished nursing school or medical school? Can it be a patient representative? There are no standards about who can play the role.
Should the chaperone be available to be seen when they’re in the room? Should they stay behind a curtain or somewhere where they’re not, so to speak, intrusive into what’s going on in the exam room? Do they sit in a chair? Do they stand? How do they behave, if you will? There’s no agreement.
There’s still no agreement on the training that a chaperone should have. Do we charge them with trying to represent what’s going on with the patient or trying to protect the doctor against any accusations that are ill founded about inappropriate conduct? Are they supposed to do both? How do they obtain consent, if they do, from the patient undergoing an examination in a sensitive part of their body or one that they’re sensitive about?
This area really requires some hard thinking if you’re considering having chaperones present. I think there are some online courses that offer some training. I haven’t looked at them, but they might be worth a look to see if they make you more comfortable about getting a chaperone oriented. I think it’s probably important to set a policy saying a chaperone must always be present for these kinds of examinations and list them, or one can be requested no matter what is going on in terms of the kind of exam being conducted.
There needs to be some statement saying that you have permission to either accept them or refuse them – or you don’t. Should they always be present, for example, with patients who are minors, adolescents or children? Does that extend that far out where a guardian, parent, or someone has to give permission?
In this area, I think we can all understand why chaperones have come to the fore, including allegations of misconduct and inappropriate touching, and considering comfort levels of patients to just put them more at ease. It’s obvious that we haven’t, as a nation or a medical profession, thought it through to the degree to which we have to.
I’m certainly not anti-chaperone, and I believe that if patients are more comfortable having one present, or a doctor is more comfortable having one present, or if we all agree that there are certain patients – kids – where certain types of examinations require or ought to expect the chaperone to be present, that’s wonderful.
We’ve got to lay out the rights of the doctors. We’ve got to lay out the rights of the institutions. We’ve got to lay out the rights of the patients. We should agree on who these people are. We should agree on how they’re trained.
We’ve got some work ahead of us if we’re going to have chaperones become a standard part of the medical examination.
Dr. Kaplan reported conflicts of interest with the Franklin Institute, Tengion, Biogen Idec, Johnson & Johnson, and PriCara.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Art Caplan, PhD. I’m at the division of medical ethics at NYU’s Grossman School of Medicine.
, such as breasts, genitalia, and the perianal area.
In some institutions, there has been a movement toward saying a chaperone must be present, that it’s mandatory. I know that is true at Yale’s health care centers and clinics. Others do so when the patient requests it. An interesting situation sometimes occurs when the hospital or the clinic requires a chaperone but the patient says, “I don’t want a chaperone. I want my privacy. I want the gynecologist or the urologist only. I don’t want anyone else to be seeing me. I’m not comfortable with anyone other than the doctor in the room.”
Complicating this issue of when is a chaperone appropriate and when can it be refused, if ever, is the fact that the role of chaperone is ill defined. For example, there isn’t really agreement on who can be a chaperone. Could it be a medical student? Could it be a nurse? Could it be another doctor? Should it be someone who at least has finished nursing school or medical school? Can it be a patient representative? There are no standards about who can play the role.
Should the chaperone be available to be seen when they’re in the room? Should they stay behind a curtain or somewhere where they’re not, so to speak, intrusive into what’s going on in the exam room? Do they sit in a chair? Do they stand? How do they behave, if you will? There’s no agreement.
There’s still no agreement on the training that a chaperone should have. Do we charge them with trying to represent what’s going on with the patient or trying to protect the doctor against any accusations that are ill founded about inappropriate conduct? Are they supposed to do both? How do they obtain consent, if they do, from the patient undergoing an examination in a sensitive part of their body or one that they’re sensitive about?
This area really requires some hard thinking if you’re considering having chaperones present. I think there are some online courses that offer some training. I haven’t looked at them, but they might be worth a look to see if they make you more comfortable about getting a chaperone oriented. I think it’s probably important to set a policy saying a chaperone must always be present for these kinds of examinations and list them, or one can be requested no matter what is going on in terms of the kind of exam being conducted.
There needs to be some statement saying that you have permission to either accept them or refuse them – or you don’t. Should they always be present, for example, with patients who are minors, adolescents or children? Does that extend that far out where a guardian, parent, or someone has to give permission?
In this area, I think we can all understand why chaperones have come to the fore, including allegations of misconduct and inappropriate touching, and considering comfort levels of patients to just put them more at ease. It’s obvious that we haven’t, as a nation or a medical profession, thought it through to the degree to which we have to.
I’m certainly not anti-chaperone, and I believe that if patients are more comfortable having one present, or a doctor is more comfortable having one present, or if we all agree that there are certain patients – kids – where certain types of examinations require or ought to expect the chaperone to be present, that’s wonderful.
We’ve got to lay out the rights of the doctors. We’ve got to lay out the rights of the institutions. We’ve got to lay out the rights of the patients. We should agree on who these people are. We should agree on how they’re trained.
We’ve got some work ahead of us if we’re going to have chaperones become a standard part of the medical examination.
Dr. Kaplan reported conflicts of interest with the Franklin Institute, Tengion, Biogen Idec, Johnson & Johnson, and PriCara.
A version of this article first appeared on Medscape.com.
New tool uses nanotechnology to speed up diagnostic testing of infectious disease
A new tool promises to expedite detection of infectious disease, according to researchers from McGill University, Montreal.
The diagnostic platform, called The device was tested for several respiratory viruses and bacteria, including the H1N1 influenza virus and SARS-CoV-2. It achieved 95% accuracy at identifying COVID-19 and its variants in 48 human saliva samples.
“COVID was something that opened our eyes, and now we have to think more seriously about point-of-care diagnostics,” Sara Mahshid, PhD, assistant professor of biomedical engineering and Canada Research Chair in Nano-Biosensing Devices at McGill University, said in an interview. The technology could become important for a range of medical applications, especially in low-resource areas.
The development was detailed in an article in Nature Nanotechnology.
Nonclinical setting
The COVID-19 pandemic has demonstrated the need for fast and accurate testing that can be used outside of a clinical setting. The gold-standard diagnostic method is PCR testing, but its accuracy comes with a trade-off. PCR testing involves a lengthy protocol and requires a centralized testing facility.
With QolorEX, the investigators aimed to develop a new test that achieves the accuracy of PCR in an automated tool that can be used outside of a testing facility or hospital setting. Dr. Mahshid noted a particular need for a tool that could be used in congregate settings, such as airports, schools, or restaurants.
The device is compact enough to sit on a tabletop or bench and can be used easily in group settings, according to Dr. Mahshid. In the future, she hopes to further miniaturize the device to make it more scalable for widespread use.
Requiring only a saliva sample, the tool is easy to use. Unlike current COVID-19 rapid tests, which involve several steps, the system is automated and does not require manually mixing reagents. After collecting a sample, a user taps a button in a smartphone or computer application. The device handles the rest.
“We’re not chemists who understand how to mix these solutions,” Dr. Mahshid said. Avoiding those extra steps may reduce the false positives and false negatives caused by user error.
Fast results
QolorEX can return results in 13 minutes, like a rapid antigen test does. Like a PCR test, the device uses nucleic acid amplification. But PCR tests typically take much longer. The sample analysis alone takes 1.5-2 hours.
The new test accelerates the reaction by injecting light-excited “hot” electrons from the surface of a nanoplasmonic sensor. The device then uses imaging and a machine learning algorithm to quantify a color transformation that occurs when a pathogen is present.
The fast, reliable results make the system potentially appropriate for use in places such as airports. Previously, passengers had to wait 24 hours for a negative COVID test before boarding a plane. A device such as QolorEX would allow screening on site.
The ability of the tool to distinguish between bacterial and viral infections so quickly is “an application that is both important and extremely difficult to achieve,” according to Nikhil Bhalla, PhD, in a research briefing. Dr. Bhalla is a lecturer in electronic engineering at Ulster University, Belfast, Ireland.
The researchers hope that by delivering results quickly, the device will help reduce the spread of respiratory diseases and possibly save lives.
‘Sensitive and specific’
The primary benefit of the tool is its ability to return results quickly while having low false positive and false negative rates, according to Leyla Soleymani, PhD, of McMaster University, Hamilton, Ont. “It is hard to come by rapid tests that are both sensitive and specific, compared to PCR,” Dr. Soleymani told this news organization.
Although QolorEX was developed to detect COVID-19 and other infectious diseases, the uses of the device are not limited to the pathogens tested. The tool can be applied to a range of tests that currently use PCR technology. Dr. Mahshid and her team are considering several other applications of the technology, such as analyzing therapeutics for antimicrobial-resistant pathogens prioritized by the World Health Organization. The technology may also have potential for detecting cancer and bacterial infections, Dr. Mahshid said in an interview.
But to Dr. Soleymani, the most exciting application remains its use in diagnosing infectious diseases. She noted, however, that it’s unclear whether the price of the device will be too high for widespread home use. It may be more practical for family physician clinics and other facilities.
Before the device becomes commercially available, more testing is needed to validate the results, which are based on a limited number of samples that were available in a research setting.
The study was supported by the MI4 Emergency COVID-19 Research Funding, Natural Sciences and Engineering Research Council of Canada, Canadian Institutes of Health Research, Canada Foundation for Innovation, and McGill University. Dr. Mahshid and Dr. Soleymani reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A new tool promises to expedite detection of infectious disease, according to researchers from McGill University, Montreal.
The diagnostic platform, called The device was tested for several respiratory viruses and bacteria, including the H1N1 influenza virus and SARS-CoV-2. It achieved 95% accuracy at identifying COVID-19 and its variants in 48 human saliva samples.
“COVID was something that opened our eyes, and now we have to think more seriously about point-of-care diagnostics,” Sara Mahshid, PhD, assistant professor of biomedical engineering and Canada Research Chair in Nano-Biosensing Devices at McGill University, said in an interview. The technology could become important for a range of medical applications, especially in low-resource areas.
The development was detailed in an article in Nature Nanotechnology.
Nonclinical setting
The COVID-19 pandemic has demonstrated the need for fast and accurate testing that can be used outside of a clinical setting. The gold-standard diagnostic method is PCR testing, but its accuracy comes with a trade-off. PCR testing involves a lengthy protocol and requires a centralized testing facility.
With QolorEX, the investigators aimed to develop a new test that achieves the accuracy of PCR in an automated tool that can be used outside of a testing facility or hospital setting. Dr. Mahshid noted a particular need for a tool that could be used in congregate settings, such as airports, schools, or restaurants.
The device is compact enough to sit on a tabletop or bench and can be used easily in group settings, according to Dr. Mahshid. In the future, she hopes to further miniaturize the device to make it more scalable for widespread use.
Requiring only a saliva sample, the tool is easy to use. Unlike current COVID-19 rapid tests, which involve several steps, the system is automated and does not require manually mixing reagents. After collecting a sample, a user taps a button in a smartphone or computer application. The device handles the rest.
“We’re not chemists who understand how to mix these solutions,” Dr. Mahshid said. Avoiding those extra steps may reduce the false positives and false negatives caused by user error.
Fast results
QolorEX can return results in 13 minutes, like a rapid antigen test does. Like a PCR test, the device uses nucleic acid amplification. But PCR tests typically take much longer. The sample analysis alone takes 1.5-2 hours.
The new test accelerates the reaction by injecting light-excited “hot” electrons from the surface of a nanoplasmonic sensor. The device then uses imaging and a machine learning algorithm to quantify a color transformation that occurs when a pathogen is present.
The fast, reliable results make the system potentially appropriate for use in places such as airports. Previously, passengers had to wait 24 hours for a negative COVID test before boarding a plane. A device such as QolorEX would allow screening on site.
The ability of the tool to distinguish between bacterial and viral infections so quickly is “an application that is both important and extremely difficult to achieve,” according to Nikhil Bhalla, PhD, in a research briefing. Dr. Bhalla is a lecturer in electronic engineering at Ulster University, Belfast, Ireland.
The researchers hope that by delivering results quickly, the device will help reduce the spread of respiratory diseases and possibly save lives.
‘Sensitive and specific’
The primary benefit of the tool is its ability to return results quickly while having low false positive and false negative rates, according to Leyla Soleymani, PhD, of McMaster University, Hamilton, Ont. “It is hard to come by rapid tests that are both sensitive and specific, compared to PCR,” Dr. Soleymani told this news organization.
Although QolorEX was developed to detect COVID-19 and other infectious diseases, the uses of the device are not limited to the pathogens tested. The tool can be applied to a range of tests that currently use PCR technology. Dr. Mahshid and her team are considering several other applications of the technology, such as analyzing therapeutics for antimicrobial-resistant pathogens prioritized by the World Health Organization. The technology may also have potential for detecting cancer and bacterial infections, Dr. Mahshid said in an interview.
But to Dr. Soleymani, the most exciting application remains its use in diagnosing infectious diseases. She noted, however, that it’s unclear whether the price of the device will be too high for widespread home use. It may be more practical for family physician clinics and other facilities.
Before the device becomes commercially available, more testing is needed to validate the results, which are based on a limited number of samples that were available in a research setting.
The study was supported by the MI4 Emergency COVID-19 Research Funding, Natural Sciences and Engineering Research Council of Canada, Canadian Institutes of Health Research, Canada Foundation for Innovation, and McGill University. Dr. Mahshid and Dr. Soleymani reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
A new tool promises to expedite detection of infectious disease, according to researchers from McGill University, Montreal.
The diagnostic platform, called The device was tested for several respiratory viruses and bacteria, including the H1N1 influenza virus and SARS-CoV-2. It achieved 95% accuracy at identifying COVID-19 and its variants in 48 human saliva samples.
“COVID was something that opened our eyes, and now we have to think more seriously about point-of-care diagnostics,” Sara Mahshid, PhD, assistant professor of biomedical engineering and Canada Research Chair in Nano-Biosensing Devices at McGill University, said in an interview. The technology could become important for a range of medical applications, especially in low-resource areas.
The development was detailed in an article in Nature Nanotechnology.
Nonclinical setting
The COVID-19 pandemic has demonstrated the need for fast and accurate testing that can be used outside of a clinical setting. The gold-standard diagnostic method is PCR testing, but its accuracy comes with a trade-off. PCR testing involves a lengthy protocol and requires a centralized testing facility.
With QolorEX, the investigators aimed to develop a new test that achieves the accuracy of PCR in an automated tool that can be used outside of a testing facility or hospital setting. Dr. Mahshid noted a particular need for a tool that could be used in congregate settings, such as airports, schools, or restaurants.
The device is compact enough to sit on a tabletop or bench and can be used easily in group settings, according to Dr. Mahshid. In the future, she hopes to further miniaturize the device to make it more scalable for widespread use.
Requiring only a saliva sample, the tool is easy to use. Unlike current COVID-19 rapid tests, which involve several steps, the system is automated and does not require manually mixing reagents. After collecting a sample, a user taps a button in a smartphone or computer application. The device handles the rest.
“We’re not chemists who understand how to mix these solutions,” Dr. Mahshid said. Avoiding those extra steps may reduce the false positives and false negatives caused by user error.
Fast results
QolorEX can return results in 13 minutes, like a rapid antigen test does. Like a PCR test, the device uses nucleic acid amplification. But PCR tests typically take much longer. The sample analysis alone takes 1.5-2 hours.
The new test accelerates the reaction by injecting light-excited “hot” electrons from the surface of a nanoplasmonic sensor. The device then uses imaging and a machine learning algorithm to quantify a color transformation that occurs when a pathogen is present.
The fast, reliable results make the system potentially appropriate for use in places such as airports. Previously, passengers had to wait 24 hours for a negative COVID test before boarding a plane. A device such as QolorEX would allow screening on site.
The ability of the tool to distinguish between bacterial and viral infections so quickly is “an application that is both important and extremely difficult to achieve,” according to Nikhil Bhalla, PhD, in a research briefing. Dr. Bhalla is a lecturer in electronic engineering at Ulster University, Belfast, Ireland.
The researchers hope that by delivering results quickly, the device will help reduce the spread of respiratory diseases and possibly save lives.
‘Sensitive and specific’
The primary benefit of the tool is its ability to return results quickly while having low false positive and false negative rates, according to Leyla Soleymani, PhD, of McMaster University, Hamilton, Ont. “It is hard to come by rapid tests that are both sensitive and specific, compared to PCR,” Dr. Soleymani told this news organization.
Although QolorEX was developed to detect COVID-19 and other infectious diseases, the uses of the device are not limited to the pathogens tested. The tool can be applied to a range of tests that currently use PCR technology. Dr. Mahshid and her team are considering several other applications of the technology, such as analyzing therapeutics for antimicrobial-resistant pathogens prioritized by the World Health Organization. The technology may also have potential for detecting cancer and bacterial infections, Dr. Mahshid said in an interview.
But to Dr. Soleymani, the most exciting application remains its use in diagnosing infectious diseases. She noted, however, that it’s unclear whether the price of the device will be too high for widespread home use. It may be more practical for family physician clinics and other facilities.
Before the device becomes commercially available, more testing is needed to validate the results, which are based on a limited number of samples that were available in a research setting.
The study was supported by the MI4 Emergency COVID-19 Research Funding, Natural Sciences and Engineering Research Council of Canada, Canadian Institutes of Health Research, Canada Foundation for Innovation, and McGill University. Dr. Mahshid and Dr. Soleymani reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM NATURE NANOTECHNOLOGY