User login
For MD-IQ use only
Spironolactone safe, effective option for women with hidradenitis suppurativa
CARLSBAD, CALIF. –
Those are the key findings from a single-center retrospective study that Jennifer L. Hsiao, MD, and colleagues presented during a poster session at the annual symposium of the California Society of Dermatology & Dermatologic Surgery.
In an interview after the meeting, Dr. Hsiao, a dermatologist who directs the hidradenitis suppurativa clinic at the University of Southern California, Los Angeles, said that hormones are thought to play a role in HS pathogenesis given the typical HS symptom onset around puberty and fluctuations in disease activity with menses (typically premenstrual flares) and pregnancy. “Spironolactone, an anti-androgenic agent, is used to treat HS in women; however, there is a paucity of data on the efficacy of spironolactone for HS and whether certain patient characteristics may influence treatment response,” she told this news organization. “This study is unique in that we contribute to existing literature regarding spironolactone efficacy in HS and we also investigate whether the presence of menstrual HS flares or polycystic ovarian syndrome influences the likelihood of response to spironolactone.”
For the analysis, Dr. Hsiao and colleagues retrospectively reviewed the medical records of 53 adult women with HS who were prescribed spironolactone and who received care at USC’s HS clinic between January 2015 and December 2021. They collected data on demographics, comorbidities, HS medications, treatment response at 3 and 6 months, as well as adverse events. They also evaluated physician-assessed response to treatment when available.
The mean age of patients was 31 years, 37% were White, 30.4% were Black, 21.7% were Hispanic, 6.5% were Asian, and the remainder were biracial. The mean age at HS diagnosis was 25.1 years and the three most common comorbidities were acne (50.9%), obesity (45.3%), and anemia (37.7%). As for menstrual history, 56.6% had perimenstrual HS flares and 37.7% had irregular menstrual cycles. The top three classes of concomitant medications were antibiotics (58.5%), oral contraceptives (50.9%), and other birth control methods (18.9%).
The mean spironolactone dose was 104 mg/day; 84.1% of the women experienced improvement of HS 3 months after starting the drug, while 81.8% had improvement of their HS 6 months after starting the drug. The researchers also found that 56.6% of women had documented perimenstrual HS flares and 7.5% had PCOS.
“Spironolactone is often thought of as a helpful medication to consider if a patient reports having HS flares around menses or features of PCOS,” Dr. Hsiao said. However, she added, “our study found that there was no statistically significant difference in the response to spironolactone based on the presence of premenstrual flares or concomitant PCOS.” She said that spironolactone may be used as an adjunct therapeutic option in patients with more severe disease in addition to other medical and surgical therapies for HS. “Combining different treatment options that target different pathophysiologic factors is usually required to achieve adequate disease control in HS,” she said.
Dr. Hsiao acknowledged certain limitations of the study, including its single-center design and small sample size. “A confounding variable is that some patients were on other medications in addition to spironolactone, which may have influenced treatment outcomes,” she noted. “Larger prospective studies are needed to identify optimal dosing for spironolactone therapy in HS as well as predictors of treatment response.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, said that with only one FDA-approved systemic medication for the management of HS (adalimumab), “we off-label bandits must be creative to curtail the incredibly painful impact this chronic, destructive inflammatory disease can have on our patients.”
“The evidence supporting our approaches, whether it be antibiotics, immunomodulators, or in this case, antihormonal therapies, is limited, so more data is always welcome,” said Dr. Friedman, who was not involved with the study. “One very interesting point raised by the authors, one I share with my trainees frequently from my own experience, is that regardless of menstrual cycle abnormalities, spironolactone can be impactful. This is important to remember, in that overt signs of hormonal influences is not a requisite for the use or effectiveness of antihormonal therapy.”
Dr. Hsiao disclosed that she is a member of board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Novartis, UCB, as a speaker for AbbVie, and as an investigator for Amgen, Boehringer Ingelheim, and Incyte. Dr. Friedman reported having no relevant financial disclosures.
CARLSBAD, CALIF. –
Those are the key findings from a single-center retrospective study that Jennifer L. Hsiao, MD, and colleagues presented during a poster session at the annual symposium of the California Society of Dermatology & Dermatologic Surgery.
In an interview after the meeting, Dr. Hsiao, a dermatologist who directs the hidradenitis suppurativa clinic at the University of Southern California, Los Angeles, said that hormones are thought to play a role in HS pathogenesis given the typical HS symptom onset around puberty and fluctuations in disease activity with menses (typically premenstrual flares) and pregnancy. “Spironolactone, an anti-androgenic agent, is used to treat HS in women; however, there is a paucity of data on the efficacy of spironolactone for HS and whether certain patient characteristics may influence treatment response,” she told this news organization. “This study is unique in that we contribute to existing literature regarding spironolactone efficacy in HS and we also investigate whether the presence of menstrual HS flares or polycystic ovarian syndrome influences the likelihood of response to spironolactone.”
For the analysis, Dr. Hsiao and colleagues retrospectively reviewed the medical records of 53 adult women with HS who were prescribed spironolactone and who received care at USC’s HS clinic between January 2015 and December 2021. They collected data on demographics, comorbidities, HS medications, treatment response at 3 and 6 months, as well as adverse events. They also evaluated physician-assessed response to treatment when available.
The mean age of patients was 31 years, 37% were White, 30.4% were Black, 21.7% were Hispanic, 6.5% were Asian, and the remainder were biracial. The mean age at HS diagnosis was 25.1 years and the three most common comorbidities were acne (50.9%), obesity (45.3%), and anemia (37.7%). As for menstrual history, 56.6% had perimenstrual HS flares and 37.7% had irregular menstrual cycles. The top three classes of concomitant medications were antibiotics (58.5%), oral contraceptives (50.9%), and other birth control methods (18.9%).
The mean spironolactone dose was 104 mg/day; 84.1% of the women experienced improvement of HS 3 months after starting the drug, while 81.8% had improvement of their HS 6 months after starting the drug. The researchers also found that 56.6% of women had documented perimenstrual HS flares and 7.5% had PCOS.
“Spironolactone is often thought of as a helpful medication to consider if a patient reports having HS flares around menses or features of PCOS,” Dr. Hsiao said. However, she added, “our study found that there was no statistically significant difference in the response to spironolactone based on the presence of premenstrual flares or concomitant PCOS.” She said that spironolactone may be used as an adjunct therapeutic option in patients with more severe disease in addition to other medical and surgical therapies for HS. “Combining different treatment options that target different pathophysiologic factors is usually required to achieve adequate disease control in HS,” she said.
Dr. Hsiao acknowledged certain limitations of the study, including its single-center design and small sample size. “A confounding variable is that some patients were on other medications in addition to spironolactone, which may have influenced treatment outcomes,” she noted. “Larger prospective studies are needed to identify optimal dosing for spironolactone therapy in HS as well as predictors of treatment response.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, said that with only one FDA-approved systemic medication for the management of HS (adalimumab), “we off-label bandits must be creative to curtail the incredibly painful impact this chronic, destructive inflammatory disease can have on our patients.”
“The evidence supporting our approaches, whether it be antibiotics, immunomodulators, or in this case, antihormonal therapies, is limited, so more data is always welcome,” said Dr. Friedman, who was not involved with the study. “One very interesting point raised by the authors, one I share with my trainees frequently from my own experience, is that regardless of menstrual cycle abnormalities, spironolactone can be impactful. This is important to remember, in that overt signs of hormonal influences is not a requisite for the use or effectiveness of antihormonal therapy.”
Dr. Hsiao disclosed that she is a member of board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Novartis, UCB, as a speaker for AbbVie, and as an investigator for Amgen, Boehringer Ingelheim, and Incyte. Dr. Friedman reported having no relevant financial disclosures.
CARLSBAD, CALIF. –
Those are the key findings from a single-center retrospective study that Jennifer L. Hsiao, MD, and colleagues presented during a poster session at the annual symposium of the California Society of Dermatology & Dermatologic Surgery.
In an interview after the meeting, Dr. Hsiao, a dermatologist who directs the hidradenitis suppurativa clinic at the University of Southern California, Los Angeles, said that hormones are thought to play a role in HS pathogenesis given the typical HS symptom onset around puberty and fluctuations in disease activity with menses (typically premenstrual flares) and pregnancy. “Spironolactone, an anti-androgenic agent, is used to treat HS in women; however, there is a paucity of data on the efficacy of spironolactone for HS and whether certain patient characteristics may influence treatment response,” she told this news organization. “This study is unique in that we contribute to existing literature regarding spironolactone efficacy in HS and we also investigate whether the presence of menstrual HS flares or polycystic ovarian syndrome influences the likelihood of response to spironolactone.”
For the analysis, Dr. Hsiao and colleagues retrospectively reviewed the medical records of 53 adult women with HS who were prescribed spironolactone and who received care at USC’s HS clinic between January 2015 and December 2021. They collected data on demographics, comorbidities, HS medications, treatment response at 3 and 6 months, as well as adverse events. They also evaluated physician-assessed response to treatment when available.
The mean age of patients was 31 years, 37% were White, 30.4% were Black, 21.7% were Hispanic, 6.5% were Asian, and the remainder were biracial. The mean age at HS diagnosis was 25.1 years and the three most common comorbidities were acne (50.9%), obesity (45.3%), and anemia (37.7%). As for menstrual history, 56.6% had perimenstrual HS flares and 37.7% had irregular menstrual cycles. The top three classes of concomitant medications were antibiotics (58.5%), oral contraceptives (50.9%), and other birth control methods (18.9%).
The mean spironolactone dose was 104 mg/day; 84.1% of the women experienced improvement of HS 3 months after starting the drug, while 81.8% had improvement of their HS 6 months after starting the drug. The researchers also found that 56.6% of women had documented perimenstrual HS flares and 7.5% had PCOS.
“Spironolactone is often thought of as a helpful medication to consider if a patient reports having HS flares around menses or features of PCOS,” Dr. Hsiao said. However, she added, “our study found that there was no statistically significant difference in the response to spironolactone based on the presence of premenstrual flares or concomitant PCOS.” She said that spironolactone may be used as an adjunct therapeutic option in patients with more severe disease in addition to other medical and surgical therapies for HS. “Combining different treatment options that target different pathophysiologic factors is usually required to achieve adequate disease control in HS,” she said.
Dr. Hsiao acknowledged certain limitations of the study, including its single-center design and small sample size. “A confounding variable is that some patients were on other medications in addition to spironolactone, which may have influenced treatment outcomes,” she noted. “Larger prospective studies are needed to identify optimal dosing for spironolactone therapy in HS as well as predictors of treatment response.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, said that with only one FDA-approved systemic medication for the management of HS (adalimumab), “we off-label bandits must be creative to curtail the incredibly painful impact this chronic, destructive inflammatory disease can have on our patients.”
“The evidence supporting our approaches, whether it be antibiotics, immunomodulators, or in this case, antihormonal therapies, is limited, so more data is always welcome,” said Dr. Friedman, who was not involved with the study. “One very interesting point raised by the authors, one I share with my trainees frequently from my own experience, is that regardless of menstrual cycle abnormalities, spironolactone can be impactful. This is important to remember, in that overt signs of hormonal influences is not a requisite for the use or effectiveness of antihormonal therapy.”
Dr. Hsiao disclosed that she is a member of board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Novartis, UCB, as a speaker for AbbVie, and as an investigator for Amgen, Boehringer Ingelheim, and Incyte. Dr. Friedman reported having no relevant financial disclosures.
AT CALDERM 2023
Federal Health Care Data Trends 2023
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner, highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Limb Loss and Prostheses
- Neurology
- Cardiology
- Mental Health
- Diabetes
- Rheumatoid Arthritis
- Respiratory illnesses
- Women's Health
- HPV and Related Cancers
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner, highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Limb Loss and Prostheses
- Neurology
- Cardiology
- Mental Health
- Diabetes
- Rheumatoid Arthritis
- Respiratory illnesses
- Women's Health
- HPV and Related Cancers
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner, highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Limb Loss and Prostheses
- Neurology
- Cardiology
- Mental Health
- Diabetes
- Rheumatoid Arthritis
- Respiratory illnesses
- Women's Health
- HPV and Related Cancers
Video-Based Coaching for Dermatology Resident Surgical Education
To the Editor:
Video-based coaching (VBC) involves a surgeon recording a surgery and then reviewing the video with a surgical coach; it is a form of education that is gaining popularity among surgical specialties.1 Video-based education is underutilized in dermatology residency training.2 We conducted a pilot study at our dermatology residency program to evaluate the efficacy and feasibility of VBC.
The University of Texas at Austin Dell Medical School institutional review board approved this study. All 4 first-year dermatology residents were recruited to participate in this study. Participants filled out a prestudy survey assessing their surgical experience, confidence in performing surgery, and attitudes on VBC. Participants used a head-mounted point-of-view camera to record themselves performing a wide local excision on the trunk or extremities of a live human patient. Participants then reviewed the recording on their own and scored themselves using the Objective Structured Assessment of Technical Skills (OSATS) scoring table (scored from 1 to 5, with 5 being the highest possible score for each element), which is a validated tool for assessing surgical skills (eTable 1).3 Given that there were no assistants participating in the surgery, this element of the OSATS scoring table was excluded, making a maximum possible score of 30 and a minimum possible score of 6. After scoring themselves, participants then had a 1-on-1 coaching session with a fellowship-trained dermatologic surgeon (M.F. or T.H.) via online teleconferencing.
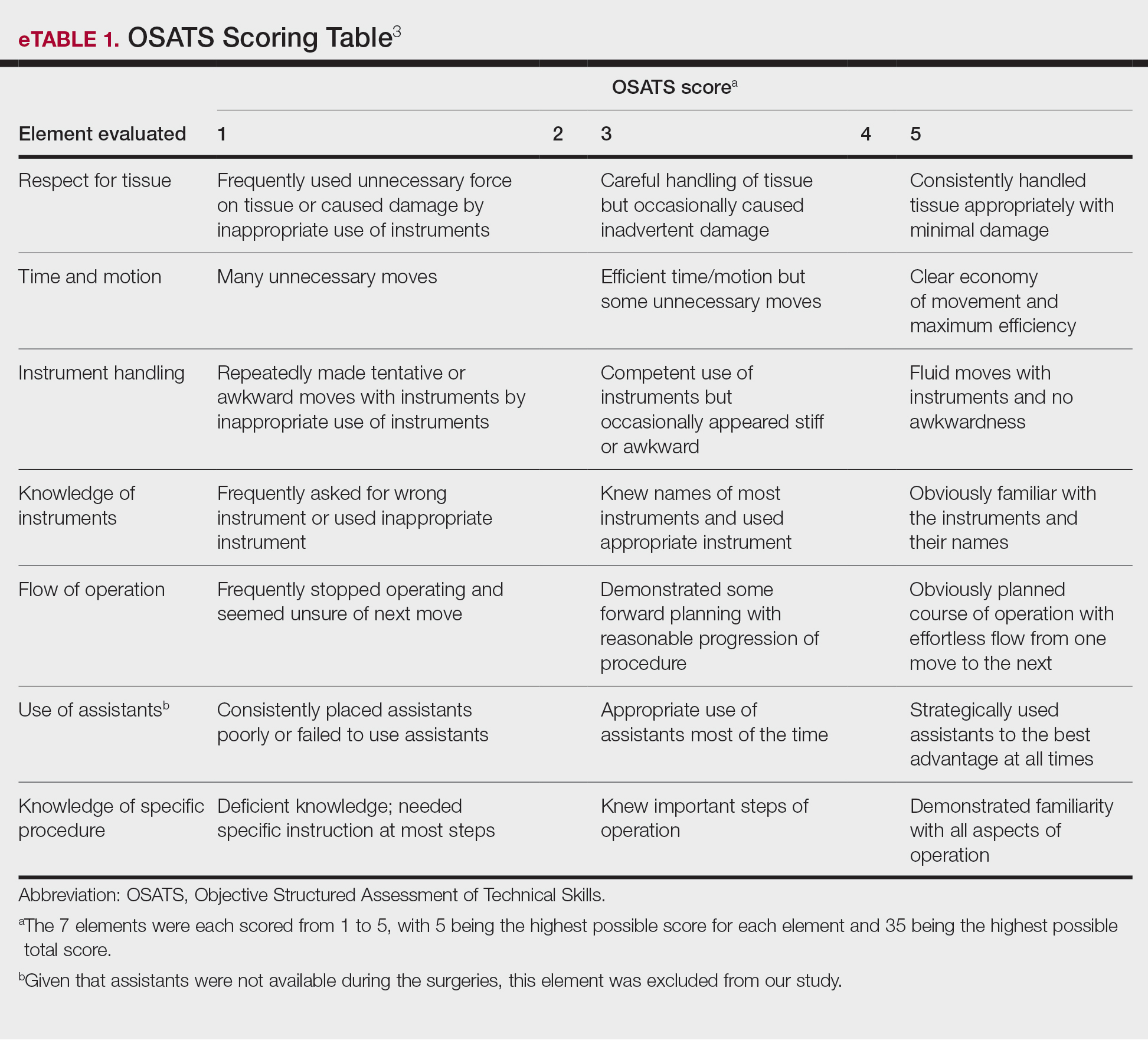
During the coaching session, participants and coaches reviewed the video. The surgical coaches also scored the residents using the OSATS, then residents and coaches discussed how the resident could improve using the OSATS scores as a guide. The residents then completed a poststudy survey assessing their surgical experience, confidence in performing surgery, and attitudes on VBC. Descriptive statistics were reported.
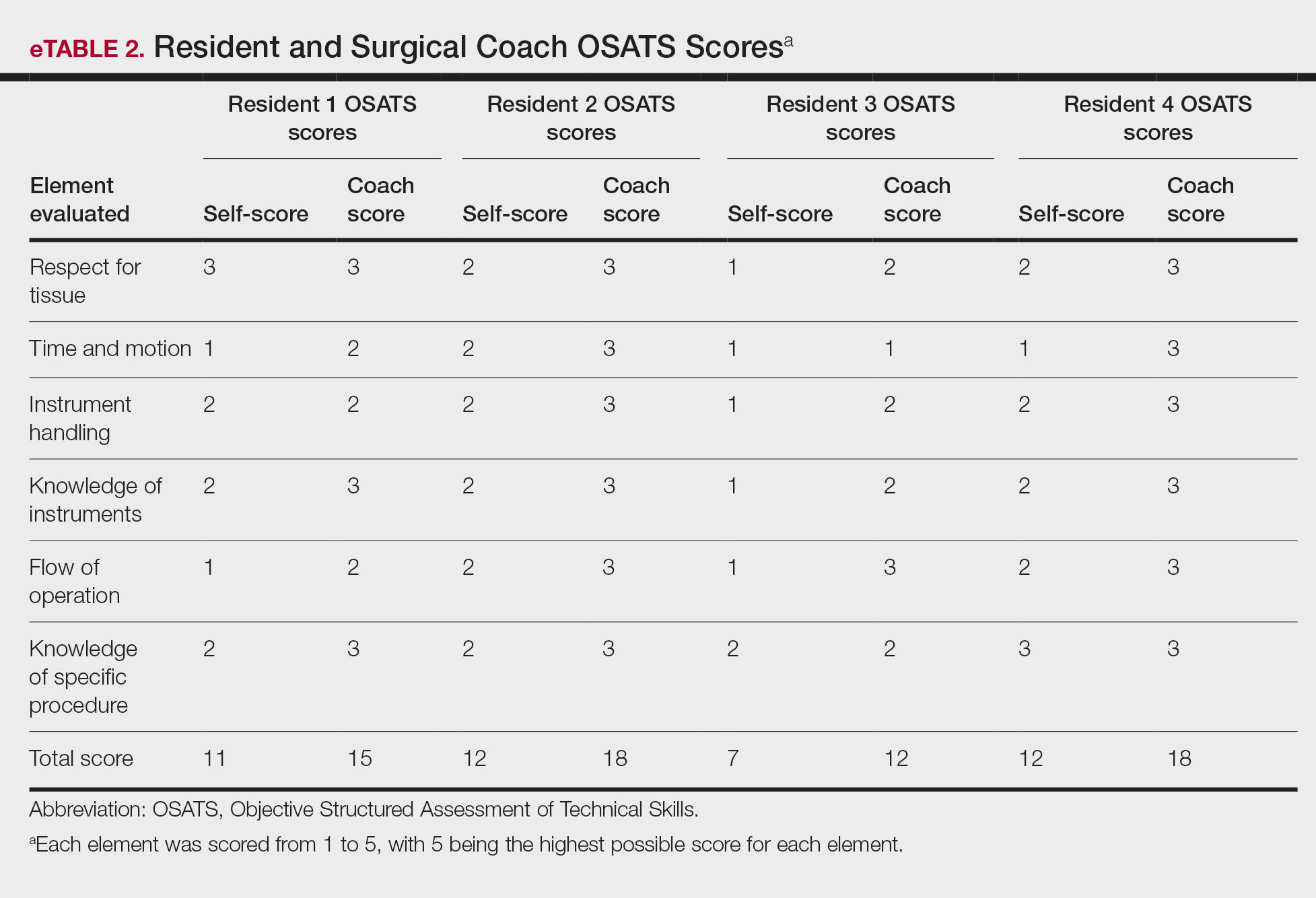
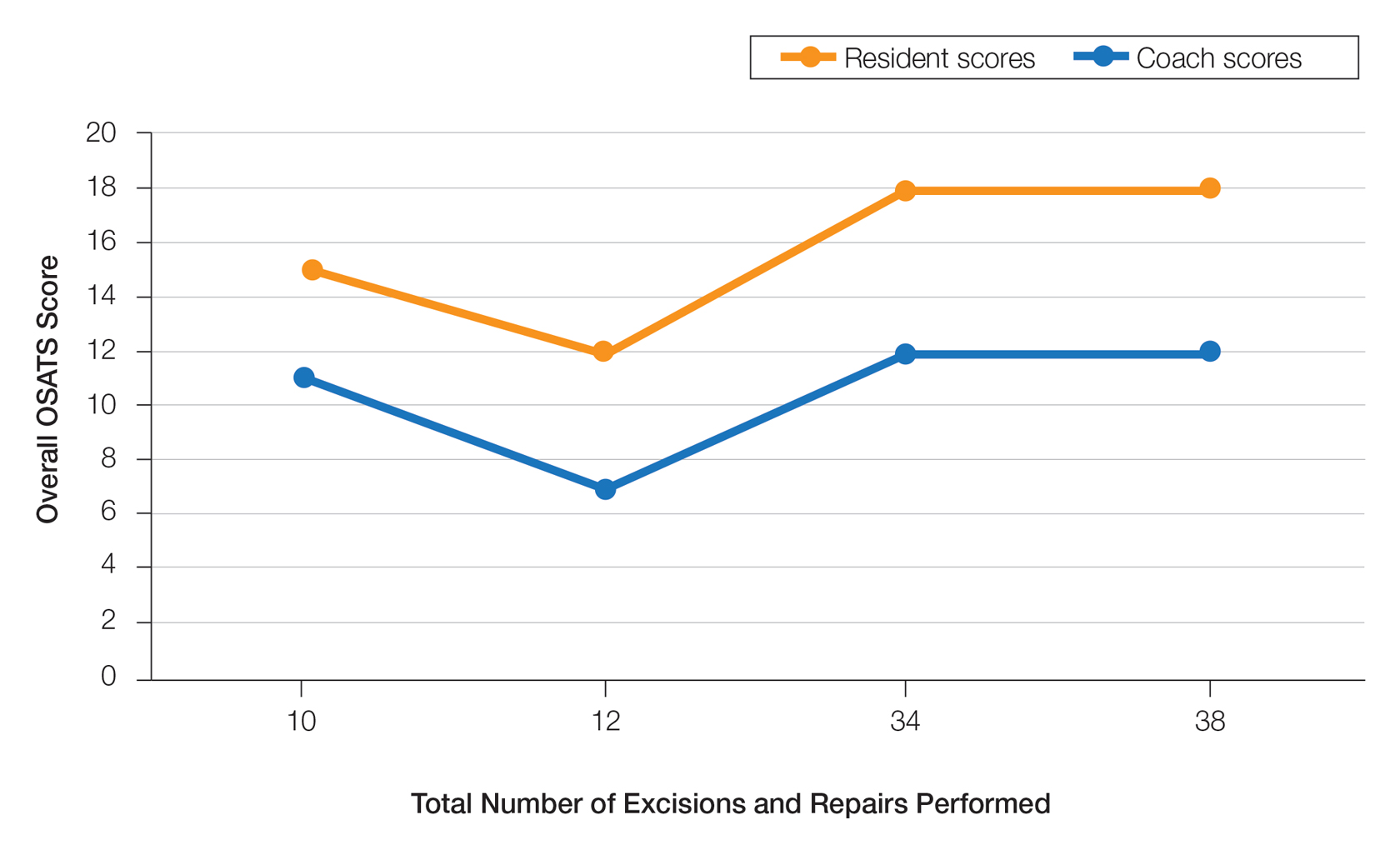
On average, residents spent 31.3 minutes reviewing their own surgeries and scoring themselves. The average time for a coaching session, which included time spent scoring, was 13.8 minutes. Residents scored themselves lower than the surgical coaches did by an average of 5.25 points (eTable 2). Residents gave themselves an average total score of 10.5, while their respective surgical coaches gave the residents an average score of 15.75. There was a trend of residents with greater surgical experience having higher OSATS scores (Figure). After the coaching session, 3 of 4 residents reported that they felt more confident in their surgical skills. All residents felt more confident in assessing their surgical skills and felt that VBC was an effective teaching measure. All residents agreed that VBC should be continued as part of their residency training.
Video-based coaching has the potential to provide several benefits for dermatology trainees. Because receiving feedback intraoperatively often can be distracting and incomplete, video review can instead allow the surgeon to focus on performing the surgery and then later focus on learning while reviewing the video.1,4 Feedback also can be more comprehensive and delivered without concern for time constraints or disturbing clinic flow as well as without the additional concern of the patient overhearing comments and feedback.3 Although independent video review in the absence of coaching can lead to improvement in surgical skills, the addition of VBC provides even greater potential educational benefit.4 During the COVID-19 pandemic, VBC allowed coaches to provide feedback without additional exposures. We utilized dermatologic surgery faculty as coaches, but this format of training also would apply to general dermatology faculty.
Another goal of VBC is to enhance a trainee’s ability to perform self-directed learning, which requires accurate self-assessment.4 Accurately assessing one’s own strengths empowers a trainee to act with appropriate confidence, while understanding one’s own weaknesses allows a trainee to effectively balance confidence and caution in daily practice.5 Interestingly, in our study all residents scored themselves lower than surgical coaches, but with 1 coaching session, the residents subsequently reported greater surgical confidence.
Time constraints can be a potential barrier to surgical coaching.4 Our study demonstrates that VBC requires minimal time investment. Increasing the speed of video playback allowed for efficient evaluation of resident surgeries without compromising the coach’s ability to provide comprehensive feedback. Our feedback sessions were performed virtually, which allowed for ease of scheduling between trainees and coaches.
Our pilot study demonstrated that VBC is relatively easy to implement in a dermatology residency training setting, leveraging relatively low-cost technologies and allowing for a means of learning that residents felt was effective. Video-based coaching requires minimal time investment from both trainees and coaches and has the potential to enhance surgical confidence. Our current study is limited by its small sample size. Future studies should include follow-up recordings and assess the efficacy of VBC in enhancing surgical skills.
- Greenberg CC, Dombrowski J, Dimick JB. Video-based surgical coaching: an emerging approach to performance improvement. JAMA Surg. 2016;151:282-283.
- Dai J, Bordeaux JS, Miller CJ, et al. Assessing surgical training and deliberate practice methods in dermatology residency: a survey of dermatology program directors. Dermatol Surg. 2016;42:977-984.
- Chitgopeker P, Sidey K, Aronson A, et al. Surgical skills video-based assessment tool for dermatology residents: a prospective pilot study. J Am Acad Dermatol. 2020;83:614-616.
- Bull NB, Silverman CD, Bonrath EM. Targeted surgical coaching can improve operative self-assessment ability: a single-blinded nonrandomized trial. Surgery. 2020;167:308-313.
- Eva KW, Regehr G. Self-assessment in the health professions: a reformulation and research agenda. Acad Med. 2005;80(10 suppl):S46-S54.
To the Editor:
Video-based coaching (VBC) involves a surgeon recording a surgery and then reviewing the video with a surgical coach; it is a form of education that is gaining popularity among surgical specialties.1 Video-based education is underutilized in dermatology residency training.2 We conducted a pilot study at our dermatology residency program to evaluate the efficacy and feasibility of VBC.
The University of Texas at Austin Dell Medical School institutional review board approved this study. All 4 first-year dermatology residents were recruited to participate in this study. Participants filled out a prestudy survey assessing their surgical experience, confidence in performing surgery, and attitudes on VBC. Participants used a head-mounted point-of-view camera to record themselves performing a wide local excision on the trunk or extremities of a live human patient. Participants then reviewed the recording on their own and scored themselves using the Objective Structured Assessment of Technical Skills (OSATS) scoring table (scored from 1 to 5, with 5 being the highest possible score for each element), which is a validated tool for assessing surgical skills (eTable 1).3 Given that there were no assistants participating in the surgery, this element of the OSATS scoring table was excluded, making a maximum possible score of 30 and a minimum possible score of 6. After scoring themselves, participants then had a 1-on-1 coaching session with a fellowship-trained dermatologic surgeon (M.F. or T.H.) via online teleconferencing.

During the coaching session, participants and coaches reviewed the video. The surgical coaches also scored the residents using the OSATS, then residents and coaches discussed how the resident could improve using the OSATS scores as a guide. The residents then completed a poststudy survey assessing their surgical experience, confidence in performing surgery, and attitudes on VBC. Descriptive statistics were reported.

On average, residents spent 31.3 minutes reviewing their own surgeries and scoring themselves. The average time for a coaching session, which included time spent scoring, was 13.8 minutes. Residents scored themselves lower than the surgical coaches did by an average of 5.25 points (eTable 2). Residents gave themselves an average total score of 10.5, while their respective surgical coaches gave the residents an average score of 15.75. There was a trend of residents with greater surgical experience having higher OSATS scores (Figure). After the coaching session, 3 of 4 residents reported that they felt more confident in their surgical skills. All residents felt more confident in assessing their surgical skills and felt that VBC was an effective teaching measure. All residents agreed that VBC should be continued as part of their residency training.

Video-based coaching has the potential to provide several benefits for dermatology trainees. Because receiving feedback intraoperatively often can be distracting and incomplete, video review can instead allow the surgeon to focus on performing the surgery and then later focus on learning while reviewing the video.1,4 Feedback also can be more comprehensive and delivered without concern for time constraints or disturbing clinic flow as well as without the additional concern of the patient overhearing comments and feedback.3 Although independent video review in the absence of coaching can lead to improvement in surgical skills, the addition of VBC provides even greater potential educational benefit.4 During the COVID-19 pandemic, VBC allowed coaches to provide feedback without additional exposures. We utilized dermatologic surgery faculty as coaches, but this format of training also would apply to general dermatology faculty.
Another goal of VBC is to enhance a trainee’s ability to perform self-directed learning, which requires accurate self-assessment.4 Accurately assessing one’s own strengths empowers a trainee to act with appropriate confidence, while understanding one’s own weaknesses allows a trainee to effectively balance confidence and caution in daily practice.5 Interestingly, in our study all residents scored themselves lower than surgical coaches, but with 1 coaching session, the residents subsequently reported greater surgical confidence.
Time constraints can be a potential barrier to surgical coaching.4 Our study demonstrates that VBC requires minimal time investment. Increasing the speed of video playback allowed for efficient evaluation of resident surgeries without compromising the coach’s ability to provide comprehensive feedback. Our feedback sessions were performed virtually, which allowed for ease of scheduling between trainees and coaches.
Our pilot study demonstrated that VBC is relatively easy to implement in a dermatology residency training setting, leveraging relatively low-cost technologies and allowing for a means of learning that residents felt was effective. Video-based coaching requires minimal time investment from both trainees and coaches and has the potential to enhance surgical confidence. Our current study is limited by its small sample size. Future studies should include follow-up recordings and assess the efficacy of VBC in enhancing surgical skills.
To the Editor:
Video-based coaching (VBC) involves a surgeon recording a surgery and then reviewing the video with a surgical coach; it is a form of education that is gaining popularity among surgical specialties.1 Video-based education is underutilized in dermatology residency training.2 We conducted a pilot study at our dermatology residency program to evaluate the efficacy and feasibility of VBC.
The University of Texas at Austin Dell Medical School institutional review board approved this study. All 4 first-year dermatology residents were recruited to participate in this study. Participants filled out a prestudy survey assessing their surgical experience, confidence in performing surgery, and attitudes on VBC. Participants used a head-mounted point-of-view camera to record themselves performing a wide local excision on the trunk or extremities of a live human patient. Participants then reviewed the recording on their own and scored themselves using the Objective Structured Assessment of Technical Skills (OSATS) scoring table (scored from 1 to 5, with 5 being the highest possible score for each element), which is a validated tool for assessing surgical skills (eTable 1).3 Given that there were no assistants participating in the surgery, this element of the OSATS scoring table was excluded, making a maximum possible score of 30 and a minimum possible score of 6. After scoring themselves, participants then had a 1-on-1 coaching session with a fellowship-trained dermatologic surgeon (M.F. or T.H.) via online teleconferencing.

During the coaching session, participants and coaches reviewed the video. The surgical coaches also scored the residents using the OSATS, then residents and coaches discussed how the resident could improve using the OSATS scores as a guide. The residents then completed a poststudy survey assessing their surgical experience, confidence in performing surgery, and attitudes on VBC. Descriptive statistics were reported.

On average, residents spent 31.3 minutes reviewing their own surgeries and scoring themselves. The average time for a coaching session, which included time spent scoring, was 13.8 minutes. Residents scored themselves lower than the surgical coaches did by an average of 5.25 points (eTable 2). Residents gave themselves an average total score of 10.5, while their respective surgical coaches gave the residents an average score of 15.75. There was a trend of residents with greater surgical experience having higher OSATS scores (Figure). After the coaching session, 3 of 4 residents reported that they felt more confident in their surgical skills. All residents felt more confident in assessing their surgical skills and felt that VBC was an effective teaching measure. All residents agreed that VBC should be continued as part of their residency training.

Video-based coaching has the potential to provide several benefits for dermatology trainees. Because receiving feedback intraoperatively often can be distracting and incomplete, video review can instead allow the surgeon to focus on performing the surgery and then later focus on learning while reviewing the video.1,4 Feedback also can be more comprehensive and delivered without concern for time constraints or disturbing clinic flow as well as without the additional concern of the patient overhearing comments and feedback.3 Although independent video review in the absence of coaching can lead to improvement in surgical skills, the addition of VBC provides even greater potential educational benefit.4 During the COVID-19 pandemic, VBC allowed coaches to provide feedback without additional exposures. We utilized dermatologic surgery faculty as coaches, but this format of training also would apply to general dermatology faculty.
Another goal of VBC is to enhance a trainee’s ability to perform self-directed learning, which requires accurate self-assessment.4 Accurately assessing one’s own strengths empowers a trainee to act with appropriate confidence, while understanding one’s own weaknesses allows a trainee to effectively balance confidence and caution in daily practice.5 Interestingly, in our study all residents scored themselves lower than surgical coaches, but with 1 coaching session, the residents subsequently reported greater surgical confidence.
Time constraints can be a potential barrier to surgical coaching.4 Our study demonstrates that VBC requires minimal time investment. Increasing the speed of video playback allowed for efficient evaluation of resident surgeries without compromising the coach’s ability to provide comprehensive feedback. Our feedback sessions were performed virtually, which allowed for ease of scheduling between trainees and coaches.
Our pilot study demonstrated that VBC is relatively easy to implement in a dermatology residency training setting, leveraging relatively low-cost technologies and allowing for a means of learning that residents felt was effective. Video-based coaching requires minimal time investment from both trainees and coaches and has the potential to enhance surgical confidence. Our current study is limited by its small sample size. Future studies should include follow-up recordings and assess the efficacy of VBC in enhancing surgical skills.
- Greenberg CC, Dombrowski J, Dimick JB. Video-based surgical coaching: an emerging approach to performance improvement. JAMA Surg. 2016;151:282-283.
- Dai J, Bordeaux JS, Miller CJ, et al. Assessing surgical training and deliberate practice methods in dermatology residency: a survey of dermatology program directors. Dermatol Surg. 2016;42:977-984.
- Chitgopeker P, Sidey K, Aronson A, et al. Surgical skills video-based assessment tool for dermatology residents: a prospective pilot study. J Am Acad Dermatol. 2020;83:614-616.
- Bull NB, Silverman CD, Bonrath EM. Targeted surgical coaching can improve operative self-assessment ability: a single-blinded nonrandomized trial. Surgery. 2020;167:308-313.
- Eva KW, Regehr G. Self-assessment in the health professions: a reformulation and research agenda. Acad Med. 2005;80(10 suppl):S46-S54.
- Greenberg CC, Dombrowski J, Dimick JB. Video-based surgical coaching: an emerging approach to performance improvement. JAMA Surg. 2016;151:282-283.
- Dai J, Bordeaux JS, Miller CJ, et al. Assessing surgical training and deliberate practice methods in dermatology residency: a survey of dermatology program directors. Dermatol Surg. 2016;42:977-984.
- Chitgopeker P, Sidey K, Aronson A, et al. Surgical skills video-based assessment tool for dermatology residents: a prospective pilot study. J Am Acad Dermatol. 2020;83:614-616.
- Bull NB, Silverman CD, Bonrath EM. Targeted surgical coaching can improve operative self-assessment ability: a single-blinded nonrandomized trial. Surgery. 2020;167:308-313.
- Eva KW, Regehr G. Self-assessment in the health professions: a reformulation and research agenda. Acad Med. 2005;80(10 suppl):S46-S54.
PRACTICE POINTS
- Video-based coaching (VBC) for surgical procedures is an up-and-coming form of medical education that allows a “coach” to provide thoughtful and in-depth feedback while reviewing a recording with the surgeon in a private setting. This format has potential utility in teaching dermatology resident surgeons being coached by a dermatology faculty member.
- We performed a pilot study demonstrating that VBC can be performed easily with a minimal time investment for both the surgeon and the coach. Dermatology residents not only felt that VBC was an effective teaching method but also should become a formal part of their education.
Should children know the severity of their disease? AAP weighs in with report
When children have a serious illness, some families choose not to disclose the severity to them, reasoning that knowing the extent of the illness may take away their hope. Deciding whether to tell children or adolescents about the seriousness of their disease is a complex judgment and can pose legal, ethical, and moral challenges for parents and care providers.
Default should be inclusion
The American Academy of Pediatrics (AAP) recommends in a new clinical report that the default should be to include children in conversations about their illness in a developmentally appropriate way, to the extent parents are comfortable.
The report, written by Sara Taub, MD and Robert Macauley, MD, MDiv, both in the department of pediatrics at Oregon Health & Science University in Portland, on behalf of the AAP Committee on Bioethics, was published online and appears in the October issue of Pediatrics.
“Rather than taking away hope, as some may fear, this approach of openness may create a space for children to ask their questions, share their concerns, and set goals that are appropriate to the circumstances,” the authors wrote in a press release.
The report offers strategies based on ethical, historical, legal, and cultural considerations when discussing what to share with a child or adolescent.
Some of the AAP’s other recommendations include the following:
- If the parents request nondisclosure, the first response should be seeking to understand why they prefer that stance. The care team members should also explain their position to parents.
- If there is no consensus on disclosure, establishing what each party believes is the minimum information that should be shared is important.
- Additional resources to navigate disagreement may be helpful, such as hospital ethics committees, mediators and patient advocates.
- Conversations with the family should be documented in the medical record.
Children may know more than you think
Dr. Taub said that even very young children may know more about their disease than adults believe.
“Without disclosure,” she said, “as children hear the conversations around them, they glean partial information and may weave together stories that are more frightening than reality.”
Sometimes families and the care team disagree on disclosure and for that scenario, the report offers guidance in finding middle ground.
For instance, when pediatricians feel ethically obligated to share information when parents oppose sharing, “pediatricians can reframe the discussion from whether information should be shared with the patient to what information will be communicated, how, and by whom,” the authors said in a press release.
Should you tell 15-year-old paraplegia is likely?
The authors give a case example of a 15-year-old whose spinal tumor likely will lead to paraplegia within weeks. Very few treatment options are available.
The parents ask the care team to avoid any discussions with the child about prognosis, reasoning that the news will be crushing and it’s better to deal with it if or when it happens.
The care team, however, feels compelled to find out about specific activities important to the child that may no longer be feasible with paraplegia.
The parents cite the child’s love of soccer and desire to see the Statue of Liberty. With that information and keeping the parents’ wishes in mind, the team reframes the conversation with the child in terms of goals, acknowledging that mobility may be more difficult in the future.
That conversation leads the child and the family to discuss moving up the trip to New York they had planned.
Guidance where there has been little
Timothy Joos, MD, MPH, a pediatrician who practices at a community health center in Seattle, who was not part of the recommendation team, said he was glad to see the AAP issue advice on a complex topic for which there is little practical guidance.
The authors’ case examples were “heart-tugging,” he said, and will help pediatricians work through their own scenarios.
Dr. Joos agreed with the overall premise that the default should be sharing the information.
“One of the foundations of medicine is truthfulness and openness and if we depart from that, we really have to have a good reason,” Dr. Joos said.
He said that since lying to patients should be nonnegotiable for any physician, it may help to talk with the parents first before answering an inquisitive patient’s questions and then have all parties gather for a discussion.
The authors note that AAP’s clinical reports are written by medical experts and reflect the latest evidence. The reports go through several rounds of peer review before they can be approved by the AAP board of directors.
The authors and Dr. Joos report no relevant financial relationships.
When children have a serious illness, some families choose not to disclose the severity to them, reasoning that knowing the extent of the illness may take away their hope. Deciding whether to tell children or adolescents about the seriousness of their disease is a complex judgment and can pose legal, ethical, and moral challenges for parents and care providers.
Default should be inclusion
The American Academy of Pediatrics (AAP) recommends in a new clinical report that the default should be to include children in conversations about their illness in a developmentally appropriate way, to the extent parents are comfortable.
The report, written by Sara Taub, MD and Robert Macauley, MD, MDiv, both in the department of pediatrics at Oregon Health & Science University in Portland, on behalf of the AAP Committee on Bioethics, was published online and appears in the October issue of Pediatrics.
“Rather than taking away hope, as some may fear, this approach of openness may create a space for children to ask their questions, share their concerns, and set goals that are appropriate to the circumstances,” the authors wrote in a press release.
The report offers strategies based on ethical, historical, legal, and cultural considerations when discussing what to share with a child or adolescent.
Some of the AAP’s other recommendations include the following:
- If the parents request nondisclosure, the first response should be seeking to understand why they prefer that stance. The care team members should also explain their position to parents.
- If there is no consensus on disclosure, establishing what each party believes is the minimum information that should be shared is important.
- Additional resources to navigate disagreement may be helpful, such as hospital ethics committees, mediators and patient advocates.
- Conversations with the family should be documented in the medical record.
Children may know more than you think
Dr. Taub said that even very young children may know more about their disease than adults believe.
“Without disclosure,” she said, “as children hear the conversations around them, they glean partial information and may weave together stories that are more frightening than reality.”
Sometimes families and the care team disagree on disclosure and for that scenario, the report offers guidance in finding middle ground.
For instance, when pediatricians feel ethically obligated to share information when parents oppose sharing, “pediatricians can reframe the discussion from whether information should be shared with the patient to what information will be communicated, how, and by whom,” the authors said in a press release.
Should you tell 15-year-old paraplegia is likely?
The authors give a case example of a 15-year-old whose spinal tumor likely will lead to paraplegia within weeks. Very few treatment options are available.
The parents ask the care team to avoid any discussions with the child about prognosis, reasoning that the news will be crushing and it’s better to deal with it if or when it happens.
The care team, however, feels compelled to find out about specific activities important to the child that may no longer be feasible with paraplegia.
The parents cite the child’s love of soccer and desire to see the Statue of Liberty. With that information and keeping the parents’ wishes in mind, the team reframes the conversation with the child in terms of goals, acknowledging that mobility may be more difficult in the future.
That conversation leads the child and the family to discuss moving up the trip to New York they had planned.
Guidance where there has been little
Timothy Joos, MD, MPH, a pediatrician who practices at a community health center in Seattle, who was not part of the recommendation team, said he was glad to see the AAP issue advice on a complex topic for which there is little practical guidance.
The authors’ case examples were “heart-tugging,” he said, and will help pediatricians work through their own scenarios.
Dr. Joos agreed with the overall premise that the default should be sharing the information.
“One of the foundations of medicine is truthfulness and openness and if we depart from that, we really have to have a good reason,” Dr. Joos said.
He said that since lying to patients should be nonnegotiable for any physician, it may help to talk with the parents first before answering an inquisitive patient’s questions and then have all parties gather for a discussion.
The authors note that AAP’s clinical reports are written by medical experts and reflect the latest evidence. The reports go through several rounds of peer review before they can be approved by the AAP board of directors.
The authors and Dr. Joos report no relevant financial relationships.
When children have a serious illness, some families choose not to disclose the severity to them, reasoning that knowing the extent of the illness may take away their hope. Deciding whether to tell children or adolescents about the seriousness of their disease is a complex judgment and can pose legal, ethical, and moral challenges for parents and care providers.
Default should be inclusion
The American Academy of Pediatrics (AAP) recommends in a new clinical report that the default should be to include children in conversations about their illness in a developmentally appropriate way, to the extent parents are comfortable.
The report, written by Sara Taub, MD and Robert Macauley, MD, MDiv, both in the department of pediatrics at Oregon Health & Science University in Portland, on behalf of the AAP Committee on Bioethics, was published online and appears in the October issue of Pediatrics.
“Rather than taking away hope, as some may fear, this approach of openness may create a space for children to ask their questions, share their concerns, and set goals that are appropriate to the circumstances,” the authors wrote in a press release.
The report offers strategies based on ethical, historical, legal, and cultural considerations when discussing what to share with a child or adolescent.
Some of the AAP’s other recommendations include the following:
- If the parents request nondisclosure, the first response should be seeking to understand why they prefer that stance. The care team members should also explain their position to parents.
- If there is no consensus on disclosure, establishing what each party believes is the minimum information that should be shared is important.
- Additional resources to navigate disagreement may be helpful, such as hospital ethics committees, mediators and patient advocates.
- Conversations with the family should be documented in the medical record.
Children may know more than you think
Dr. Taub said that even very young children may know more about their disease than adults believe.
“Without disclosure,” she said, “as children hear the conversations around them, they glean partial information and may weave together stories that are more frightening than reality.”
Sometimes families and the care team disagree on disclosure and for that scenario, the report offers guidance in finding middle ground.
For instance, when pediatricians feel ethically obligated to share information when parents oppose sharing, “pediatricians can reframe the discussion from whether information should be shared with the patient to what information will be communicated, how, and by whom,” the authors said in a press release.
Should you tell 15-year-old paraplegia is likely?
The authors give a case example of a 15-year-old whose spinal tumor likely will lead to paraplegia within weeks. Very few treatment options are available.
The parents ask the care team to avoid any discussions with the child about prognosis, reasoning that the news will be crushing and it’s better to deal with it if or when it happens.
The care team, however, feels compelled to find out about specific activities important to the child that may no longer be feasible with paraplegia.
The parents cite the child’s love of soccer and desire to see the Statue of Liberty. With that information and keeping the parents’ wishes in mind, the team reframes the conversation with the child in terms of goals, acknowledging that mobility may be more difficult in the future.
That conversation leads the child and the family to discuss moving up the trip to New York they had planned.
Guidance where there has been little
Timothy Joos, MD, MPH, a pediatrician who practices at a community health center in Seattle, who was not part of the recommendation team, said he was glad to see the AAP issue advice on a complex topic for which there is little practical guidance.
The authors’ case examples were “heart-tugging,” he said, and will help pediatricians work through their own scenarios.
Dr. Joos agreed with the overall premise that the default should be sharing the information.
“One of the foundations of medicine is truthfulness and openness and if we depart from that, we really have to have a good reason,” Dr. Joos said.
He said that since lying to patients should be nonnegotiable for any physician, it may help to talk with the parents first before answering an inquisitive patient’s questions and then have all parties gather for a discussion.
The authors note that AAP’s clinical reports are written by medical experts and reflect the latest evidence. The reports go through several rounds of peer review before they can be approved by the AAP board of directors.
The authors and Dr. Joos report no relevant financial relationships.
From Pediatrics
Cost concerns lead to cancer treatment delays, nonadherence
TOPLINE:
METHODOLOGY:
- Navigating the complexities of insurance coverage is difficult for cancer patients, and the clinical impact of managing these intricacies remains unclear.
- To understand the issue, investigators surveyed 510 insured cancer patients in the United States about how often they estimate out-of-pocket costs for medications, doctors’ visits, and lab tests and scans, as well as how often they ask their insurance company to help them understand their coverage and how often they appeal coverage decisions.
- The team then correlated the answers with how often patients reported postponing or skipping doctors’ appointments and lab tests and how often they delayed filling prescriptions or skipped doses.
- Breast, colorectal, lung, and prostate cancer were the most common diagnoses among respondents.
TAKEAWAY:
- Overall, 55% of participants said they “never” or “rarely” engaged in any insurance-related cost tasks. The most frequently performed administrative tasks included finding out the cost before filling a prescription (28%) or before undergoing lab tests or scans (20%), as well as estimating the cost before agreeing to a treatment (20%), asking an insurance company for help understanding coverage (18%), or appealing a denial (17%).
- After adjusting for age, race/ethnicity, education, and monthly out-of-pocket costs, participants who engaged in any cost task were 18% more likely to experience treatment delays or forgo care.
- Every additional cost task or increase in frequency of a cost task was associated with 32% higher frequency of treatment delay or nonadherence.
- Age, race, and monthly out-of-pocket costs were more strongly associated with treatment delays/nonadherence than cost-task burden. Younger patients and Black patients were more likely than others to experience cost-related delays/nonadherence.
IN PRACTICE:
- “Reductions to administrative burden on patients, whether through patient-level education interventions, the adaptation of hospital-based navigation programs, or policy-focused changes to insurance systems, will be crucial” for helping patients with cancer to overcome administrative burdens and improve access to care, the authors said.
SOURCE:
- The study, led by Meredith Doherty, PhD, of the University of Pennsylvania, Philadelphia, was published in Cancer Epidemiology, Biomarkers and Prevention.
LIMITATIONS:
The survey was voluntary, which raises the possibility of self-selection bias. Recall bias may also have occurred, particularly among patients farther out from diagnosis and treatment. The investigators did not include uninsured patients and did not stratify patients by insurance type, and they did not measure or account for health care literacy.
DISCLOSURES:
The study was funded by the American Cancer Society. The investigators have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Navigating the complexities of insurance coverage is difficult for cancer patients, and the clinical impact of managing these intricacies remains unclear.
- To understand the issue, investigators surveyed 510 insured cancer patients in the United States about how often they estimate out-of-pocket costs for medications, doctors’ visits, and lab tests and scans, as well as how often they ask their insurance company to help them understand their coverage and how often they appeal coverage decisions.
- The team then correlated the answers with how often patients reported postponing or skipping doctors’ appointments and lab tests and how often they delayed filling prescriptions or skipped doses.
- Breast, colorectal, lung, and prostate cancer were the most common diagnoses among respondents.
TAKEAWAY:
- Overall, 55% of participants said they “never” or “rarely” engaged in any insurance-related cost tasks. The most frequently performed administrative tasks included finding out the cost before filling a prescription (28%) or before undergoing lab tests or scans (20%), as well as estimating the cost before agreeing to a treatment (20%), asking an insurance company for help understanding coverage (18%), or appealing a denial (17%).
- After adjusting for age, race/ethnicity, education, and monthly out-of-pocket costs, participants who engaged in any cost task were 18% more likely to experience treatment delays or forgo care.
- Every additional cost task or increase in frequency of a cost task was associated with 32% higher frequency of treatment delay or nonadherence.
- Age, race, and monthly out-of-pocket costs were more strongly associated with treatment delays/nonadherence than cost-task burden. Younger patients and Black patients were more likely than others to experience cost-related delays/nonadherence.
IN PRACTICE:
- “Reductions to administrative burden on patients, whether through patient-level education interventions, the adaptation of hospital-based navigation programs, or policy-focused changes to insurance systems, will be crucial” for helping patients with cancer to overcome administrative burdens and improve access to care, the authors said.
SOURCE:
- The study, led by Meredith Doherty, PhD, of the University of Pennsylvania, Philadelphia, was published in Cancer Epidemiology, Biomarkers and Prevention.
LIMITATIONS:
The survey was voluntary, which raises the possibility of self-selection bias. Recall bias may also have occurred, particularly among patients farther out from diagnosis and treatment. The investigators did not include uninsured patients and did not stratify patients by insurance type, and they did not measure or account for health care literacy.
DISCLOSURES:
The study was funded by the American Cancer Society. The investigators have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Navigating the complexities of insurance coverage is difficult for cancer patients, and the clinical impact of managing these intricacies remains unclear.
- To understand the issue, investigators surveyed 510 insured cancer patients in the United States about how often they estimate out-of-pocket costs for medications, doctors’ visits, and lab tests and scans, as well as how often they ask their insurance company to help them understand their coverage and how often they appeal coverage decisions.
- The team then correlated the answers with how often patients reported postponing or skipping doctors’ appointments and lab tests and how often they delayed filling prescriptions or skipped doses.
- Breast, colorectal, lung, and prostate cancer were the most common diagnoses among respondents.
TAKEAWAY:
- Overall, 55% of participants said they “never” or “rarely” engaged in any insurance-related cost tasks. The most frequently performed administrative tasks included finding out the cost before filling a prescription (28%) or before undergoing lab tests or scans (20%), as well as estimating the cost before agreeing to a treatment (20%), asking an insurance company for help understanding coverage (18%), or appealing a denial (17%).
- After adjusting for age, race/ethnicity, education, and monthly out-of-pocket costs, participants who engaged in any cost task were 18% more likely to experience treatment delays or forgo care.
- Every additional cost task or increase in frequency of a cost task was associated with 32% higher frequency of treatment delay or nonadherence.
- Age, race, and monthly out-of-pocket costs were more strongly associated with treatment delays/nonadherence than cost-task burden. Younger patients and Black patients were more likely than others to experience cost-related delays/nonadherence.
IN PRACTICE:
- “Reductions to administrative burden on patients, whether through patient-level education interventions, the adaptation of hospital-based navigation programs, or policy-focused changes to insurance systems, will be crucial” for helping patients with cancer to overcome administrative burdens and improve access to care, the authors said.
SOURCE:
- The study, led by Meredith Doherty, PhD, of the University of Pennsylvania, Philadelphia, was published in Cancer Epidemiology, Biomarkers and Prevention.
LIMITATIONS:
The survey was voluntary, which raises the possibility of self-selection bias. Recall bias may also have occurred, particularly among patients farther out from diagnosis and treatment. The investigators did not include uninsured patients and did not stratify patients by insurance type, and they did not measure or account for health care literacy.
DISCLOSURES:
The study was funded by the American Cancer Society. The investigators have disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM CANCER EPIDEMIOLOGY, BIOMARKERS AND PREVENTION
These adverse events linked to improved cancer prognosis
TOPLINE:
.
METHODOLOGY:
- Emerging evidence suggests that the presence of cutaneous immune-related adverse events may be linked with favorable outcomes among patients with cancer who receive ICIs.
- Researchers conducted a systematic review and meta-analysis that included 23 studies and a total of 22,749 patients with cancer who received ICI treatment; studies compared outcomes among patients with and those without cutaneous immune-related adverse events.
- The major outcomes evaluated in the analysis were overall survival and progression-free survival (PFS); subgroup analyses assessed cutaneous immune-related adverse event type, cancer type, and other factors.
TAKEAWAY:
- The occurrence of cutaneous immune-related adverse events was associated with improved PFS (hazard ratio, 0.52; P < .001) and overall survival (HR, 0.61; P < .001).
- In the subgroup analysis, patients with eczematous (HR, 0.69), lichenoid or lichen planus–like skin lesions (HR, 0.51), pruritus without rash (HR, 0.70), psoriasis (HR, 0.63), or vitiligo (HR, 0.30) demonstrated a significant overall survival advantage. Vitiligo was the only adverse event associated with a PFS advantage (HR, 0.28).
- Among patients with melanoma, analyses revealed a significant association between the incidence of cutaneous immune-related adverse events and improved overall survival (HR, 0.51) and PFS (HR, 0.45). The authors highlighted similar findings among patients with non–small cell lung cancer (HR, 0.50 for overall survival and 0.61 for PFS).
IN PRACTICE:
“These data suggest that [cutaneous immune-related adverse events] may have useful prognostic value in ICI treatment,” the authors concluded.
SOURCE:
The analysis, led by Fei Wang, MD, Zhong Da Hospital, School of Medicine, Southeast University, Nanjing, China, was published online in JAMA Dermatology.
LIMITATIONS:
Most of the data came from retrospective studies, and there were limited data on specific patient subgroups. The Egger tests, used to assess potential publication bias in meta-analyses, revealed publication bias.
DISCLOSURES:
No disclosures were reported. The study was supported by a grant from the Postgraduate Research and Practice Innovation Program of Jiangsu Province.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Emerging evidence suggests that the presence of cutaneous immune-related adverse events may be linked with favorable outcomes among patients with cancer who receive ICIs.
- Researchers conducted a systematic review and meta-analysis that included 23 studies and a total of 22,749 patients with cancer who received ICI treatment; studies compared outcomes among patients with and those without cutaneous immune-related adverse events.
- The major outcomes evaluated in the analysis were overall survival and progression-free survival (PFS); subgroup analyses assessed cutaneous immune-related adverse event type, cancer type, and other factors.
TAKEAWAY:
- The occurrence of cutaneous immune-related adverse events was associated with improved PFS (hazard ratio, 0.52; P < .001) and overall survival (HR, 0.61; P < .001).
- In the subgroup analysis, patients with eczematous (HR, 0.69), lichenoid or lichen planus–like skin lesions (HR, 0.51), pruritus without rash (HR, 0.70), psoriasis (HR, 0.63), or vitiligo (HR, 0.30) demonstrated a significant overall survival advantage. Vitiligo was the only adverse event associated with a PFS advantage (HR, 0.28).
- Among patients with melanoma, analyses revealed a significant association between the incidence of cutaneous immune-related adverse events and improved overall survival (HR, 0.51) and PFS (HR, 0.45). The authors highlighted similar findings among patients with non–small cell lung cancer (HR, 0.50 for overall survival and 0.61 for PFS).
IN PRACTICE:
“These data suggest that [cutaneous immune-related adverse events] may have useful prognostic value in ICI treatment,” the authors concluded.
SOURCE:
The analysis, led by Fei Wang, MD, Zhong Da Hospital, School of Medicine, Southeast University, Nanjing, China, was published online in JAMA Dermatology.
LIMITATIONS:
Most of the data came from retrospective studies, and there were limited data on specific patient subgroups. The Egger tests, used to assess potential publication bias in meta-analyses, revealed publication bias.
DISCLOSURES:
No disclosures were reported. The study was supported by a grant from the Postgraduate Research and Practice Innovation Program of Jiangsu Province.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Emerging evidence suggests that the presence of cutaneous immune-related adverse events may be linked with favorable outcomes among patients with cancer who receive ICIs.
- Researchers conducted a systematic review and meta-analysis that included 23 studies and a total of 22,749 patients with cancer who received ICI treatment; studies compared outcomes among patients with and those without cutaneous immune-related adverse events.
- The major outcomes evaluated in the analysis were overall survival and progression-free survival (PFS); subgroup analyses assessed cutaneous immune-related adverse event type, cancer type, and other factors.
TAKEAWAY:
- The occurrence of cutaneous immune-related adverse events was associated with improved PFS (hazard ratio, 0.52; P < .001) and overall survival (HR, 0.61; P < .001).
- In the subgroup analysis, patients with eczematous (HR, 0.69), lichenoid or lichen planus–like skin lesions (HR, 0.51), pruritus without rash (HR, 0.70), psoriasis (HR, 0.63), or vitiligo (HR, 0.30) demonstrated a significant overall survival advantage. Vitiligo was the only adverse event associated with a PFS advantage (HR, 0.28).
- Among patients with melanoma, analyses revealed a significant association between the incidence of cutaneous immune-related adverse events and improved overall survival (HR, 0.51) and PFS (HR, 0.45). The authors highlighted similar findings among patients with non–small cell lung cancer (HR, 0.50 for overall survival and 0.61 for PFS).
IN PRACTICE:
“These data suggest that [cutaneous immune-related adverse events] may have useful prognostic value in ICI treatment,” the authors concluded.
SOURCE:
The analysis, led by Fei Wang, MD, Zhong Da Hospital, School of Medicine, Southeast University, Nanjing, China, was published online in JAMA Dermatology.
LIMITATIONS:
Most of the data came from retrospective studies, and there were limited data on specific patient subgroups. The Egger tests, used to assess potential publication bias in meta-analyses, revealed publication bias.
DISCLOSURES:
No disclosures were reported. The study was supported by a grant from the Postgraduate Research and Practice Innovation Program of Jiangsu Province.
A version of this article first appeared on Medscape.com.
Lack of medical device tracking leaves patients vulnerable
.
As a result of this siloing of information, patients are not getting the expected benefits of a regulation finalized over a decade ago by the U.S. Food and Drug Administration.
In 2013, the agency ordered companies to include unique device identifiers (UDIs) in plain-text and barcode format on some device labels, starting with implanted devices that are considered life-sustaining. The FDA said that tracking of UDI information would speed detection of complications linked to devices.
But identifiers are rarely on devices. At the time of the regulation creation, the FDA also said it expected this data would be integrated into EHRs. But only a few pioneer organizations such as Duke University and Mercy Health have so far attempted to track any UDI data in an organized way, researchers say.
Richard J. Kovacs, MD, the chief medical officer of the American College of Cardiology, contrasted the lack of useful implementation of UDI data with the speedy transfers of information that happen routinely in other industries. For example, employees of car rental agencies use handheld devices to gather detailed information about the vehicles being returned.
“But if you go to an emergency room with a medical device in your body, no one knows what it is or where it came from or anything about it,” Dr. Kovacs said in an interview.
Many physicians with expertise in device research have pushed for years to have insurers like Medicare require identification information on medical claims.
Even researchers face multiple obstacles in trying to investigate how well UDIs have been incorporated into EHRs and outcomes tied to certain devices.
In August, a Harvard team published a study in JAMA Internal Medicine, attempting to analyze the risks of endovascular aortic repair (EVAR) devices. They reported an 11.6% risk for serious blood leaks with AFX Endovascular AAA System aneurysm devices, more than double the 5.7% risk estimated for competing products. The team selected EVAR devices for the study due in part to their known safety concerns. Endologix, the maker of the devices, declined to comment for this story.
The Harvard team used data from the Veterans Affairs health system, which is considered more well organized than most other health systems. But UDI information was found for only 19 of the 13,941 patients whose records were studied. In those cases, only partial information was included.
The researchers developed natural language processing tools, which they used to scrounge clinical notes for information about which devices patients received.
Using this method isn’t feasible for most clinicians, given that records from independent hospitals might not provide this kind of data and descriptions to search, according to the authors of an editorial accompanying the paper. Those researchers urged Congress to pass a law mandating inclusion of UDIs for all devices on claims forms as a condition for reimbursement by federal health care programs.
Setback for advocates
The movement toward UDI suffered a setback in June.
An influential, but little known federal advisory panel, the National Committee on Vital Health Statistics (NCVHS), opted to not recommend use of this information in claims, saying the FDA should consider the matter further.
Gaining an NCVHS recommendation would have been a win, said Sen. Elizabeth Warren (D-MA), Sen. Charles E. Grassley (R-IA), and Rep. Bill Pascrell Jr. (D-NJ), in a December 2022 letter to the panel.
Including UDI data would let researchers track patients’ interactions with a health system and could be used to establish population-level correlations between a particular device and a long-term outcome or side effect, the lawmakers said.
That view had the support of at least one major maker of devices, Cook Group, which sells products for a variety of specialties, including cardiology.
In a comment to NCVHS, Cook urged for the inclusion identifiers in Medicare claims.
“While some have argued that the UDI is better suited for inclusion in the electronic health records, Cook believes this argument sets up a false choice between the two,” wrote Stephen L. Ferguson, JD, the chairman of Cook’s board. “Inclusion of the UDI in both electronic health records and claims forms will lead to a more robust system of real-world data.”
In contrast, AdvaMed, the trade group for device makers, told the NCVHS that it did not support adding the information to payment claims submissions, instead just supporting the inclusion in EHRs.
Dr. Kovacs of the ACC said one potential drawback to more transparency could be challenges in interpreting reports of complications in certain cases, at least initially. Reports about a flaw or even a suspected flaw in a device might lead patients to become concerned about their implanted devices, potentially registering unfounded complaints.
But this concern can be addressed through using “scientific rigor and safeguards” and is outweighed by the potential safety benefits for patients, Dr. Kovacs said.
Patients should ask health care systems to track and share information about their implanted devices, Dr. Kovacs suggested.
“I feel it would be my right to demand that that device information follows my electronic medical record, so that it’s readily available to anyone who’s taking care of me,” Dr. Kovacs said. “They would know what it is that’s in me, whether it’s a lens in my eye or a prosthesis in my hip or a highly complicated implantable cardiac electronic device.”
The Harvard study was supported by the FDA and National Institutes of Health. Authors of the study reported receiving fees from the FDA, Burroughs Wellcome Fund, and Harvard-MIT Center for Regulatory Science outside the submitted work. No other disclosures were reported. Authors of the editorial reported past and present connections with F-Prime Capital, FDA, Johnson & Johnson, the Medical Devices Innovation Consortium; the Agency for Healthcare Research and Quality; the National Heart, Lung, and Blood Institute; and Arnold Ventures, as well being an expert witness at in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against Biogen. Authors of the Viewpoint reported past and present connections with the National Evaluation System for Health Technology Coordinating Center (NESTcc), which is part of the Medical Device Innovation Consortium (MDIC); AIM North America UDI Advisory Committee, Mass General Brigham, Arnold Ventures; the Institute for Clinical and Economic Review California Technology Assessment Forum; Yale University, Johnson & Johnson, FD, Agency for Healthcare Research and Quality; the National Heart, Lung, and Blood Institute of the National Institutes of Health; as well as having been an expert witness in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against.
A version of this article first appeared on Medscape.com.
.
As a result of this siloing of information, patients are not getting the expected benefits of a regulation finalized over a decade ago by the U.S. Food and Drug Administration.
In 2013, the agency ordered companies to include unique device identifiers (UDIs) in plain-text and barcode format on some device labels, starting with implanted devices that are considered life-sustaining. The FDA said that tracking of UDI information would speed detection of complications linked to devices.
But identifiers are rarely on devices. At the time of the regulation creation, the FDA also said it expected this data would be integrated into EHRs. But only a few pioneer organizations such as Duke University and Mercy Health have so far attempted to track any UDI data in an organized way, researchers say.
Richard J. Kovacs, MD, the chief medical officer of the American College of Cardiology, contrasted the lack of useful implementation of UDI data with the speedy transfers of information that happen routinely in other industries. For example, employees of car rental agencies use handheld devices to gather detailed information about the vehicles being returned.
“But if you go to an emergency room with a medical device in your body, no one knows what it is or where it came from or anything about it,” Dr. Kovacs said in an interview.
Many physicians with expertise in device research have pushed for years to have insurers like Medicare require identification information on medical claims.
Even researchers face multiple obstacles in trying to investigate how well UDIs have been incorporated into EHRs and outcomes tied to certain devices.
In August, a Harvard team published a study in JAMA Internal Medicine, attempting to analyze the risks of endovascular aortic repair (EVAR) devices. They reported an 11.6% risk for serious blood leaks with AFX Endovascular AAA System aneurysm devices, more than double the 5.7% risk estimated for competing products. The team selected EVAR devices for the study due in part to their known safety concerns. Endologix, the maker of the devices, declined to comment for this story.
The Harvard team used data from the Veterans Affairs health system, which is considered more well organized than most other health systems. But UDI information was found for only 19 of the 13,941 patients whose records were studied. In those cases, only partial information was included.
The researchers developed natural language processing tools, which they used to scrounge clinical notes for information about which devices patients received.
Using this method isn’t feasible for most clinicians, given that records from independent hospitals might not provide this kind of data and descriptions to search, according to the authors of an editorial accompanying the paper. Those researchers urged Congress to pass a law mandating inclusion of UDIs for all devices on claims forms as a condition for reimbursement by federal health care programs.
Setback for advocates
The movement toward UDI suffered a setback in June.
An influential, but little known federal advisory panel, the National Committee on Vital Health Statistics (NCVHS), opted to not recommend use of this information in claims, saying the FDA should consider the matter further.
Gaining an NCVHS recommendation would have been a win, said Sen. Elizabeth Warren (D-MA), Sen. Charles E. Grassley (R-IA), and Rep. Bill Pascrell Jr. (D-NJ), in a December 2022 letter to the panel.
Including UDI data would let researchers track patients’ interactions with a health system and could be used to establish population-level correlations between a particular device and a long-term outcome or side effect, the lawmakers said.
That view had the support of at least one major maker of devices, Cook Group, which sells products for a variety of specialties, including cardiology.
In a comment to NCVHS, Cook urged for the inclusion identifiers in Medicare claims.
“While some have argued that the UDI is better suited for inclusion in the electronic health records, Cook believes this argument sets up a false choice between the two,” wrote Stephen L. Ferguson, JD, the chairman of Cook’s board. “Inclusion of the UDI in both electronic health records and claims forms will lead to a more robust system of real-world data.”
In contrast, AdvaMed, the trade group for device makers, told the NCVHS that it did not support adding the information to payment claims submissions, instead just supporting the inclusion in EHRs.
Dr. Kovacs of the ACC said one potential drawback to more transparency could be challenges in interpreting reports of complications in certain cases, at least initially. Reports about a flaw or even a suspected flaw in a device might lead patients to become concerned about their implanted devices, potentially registering unfounded complaints.
But this concern can be addressed through using “scientific rigor and safeguards” and is outweighed by the potential safety benefits for patients, Dr. Kovacs said.
Patients should ask health care systems to track and share information about their implanted devices, Dr. Kovacs suggested.
“I feel it would be my right to demand that that device information follows my electronic medical record, so that it’s readily available to anyone who’s taking care of me,” Dr. Kovacs said. “They would know what it is that’s in me, whether it’s a lens in my eye or a prosthesis in my hip or a highly complicated implantable cardiac electronic device.”
The Harvard study was supported by the FDA and National Institutes of Health. Authors of the study reported receiving fees from the FDA, Burroughs Wellcome Fund, and Harvard-MIT Center for Regulatory Science outside the submitted work. No other disclosures were reported. Authors of the editorial reported past and present connections with F-Prime Capital, FDA, Johnson & Johnson, the Medical Devices Innovation Consortium; the Agency for Healthcare Research and Quality; the National Heart, Lung, and Blood Institute; and Arnold Ventures, as well being an expert witness at in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against Biogen. Authors of the Viewpoint reported past and present connections with the National Evaluation System for Health Technology Coordinating Center (NESTcc), which is part of the Medical Device Innovation Consortium (MDIC); AIM North America UDI Advisory Committee, Mass General Brigham, Arnold Ventures; the Institute for Clinical and Economic Review California Technology Assessment Forum; Yale University, Johnson & Johnson, FD, Agency for Healthcare Research and Quality; the National Heart, Lung, and Blood Institute of the National Institutes of Health; as well as having been an expert witness in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against.
A version of this article first appeared on Medscape.com.
.
As a result of this siloing of information, patients are not getting the expected benefits of a regulation finalized over a decade ago by the U.S. Food and Drug Administration.
In 2013, the agency ordered companies to include unique device identifiers (UDIs) in plain-text and barcode format on some device labels, starting with implanted devices that are considered life-sustaining. The FDA said that tracking of UDI information would speed detection of complications linked to devices.
But identifiers are rarely on devices. At the time of the regulation creation, the FDA also said it expected this data would be integrated into EHRs. But only a few pioneer organizations such as Duke University and Mercy Health have so far attempted to track any UDI data in an organized way, researchers say.
Richard J. Kovacs, MD, the chief medical officer of the American College of Cardiology, contrasted the lack of useful implementation of UDI data with the speedy transfers of information that happen routinely in other industries. For example, employees of car rental agencies use handheld devices to gather detailed information about the vehicles being returned.
“But if you go to an emergency room with a medical device in your body, no one knows what it is or where it came from or anything about it,” Dr. Kovacs said in an interview.
Many physicians with expertise in device research have pushed for years to have insurers like Medicare require identification information on medical claims.
Even researchers face multiple obstacles in trying to investigate how well UDIs have been incorporated into EHRs and outcomes tied to certain devices.
In August, a Harvard team published a study in JAMA Internal Medicine, attempting to analyze the risks of endovascular aortic repair (EVAR) devices. They reported an 11.6% risk for serious blood leaks with AFX Endovascular AAA System aneurysm devices, more than double the 5.7% risk estimated for competing products. The team selected EVAR devices for the study due in part to their known safety concerns. Endologix, the maker of the devices, declined to comment for this story.
The Harvard team used data from the Veterans Affairs health system, which is considered more well organized than most other health systems. But UDI information was found for only 19 of the 13,941 patients whose records were studied. In those cases, only partial information was included.
The researchers developed natural language processing tools, which they used to scrounge clinical notes for information about which devices patients received.
Using this method isn’t feasible for most clinicians, given that records from independent hospitals might not provide this kind of data and descriptions to search, according to the authors of an editorial accompanying the paper. Those researchers urged Congress to pass a law mandating inclusion of UDIs for all devices on claims forms as a condition for reimbursement by federal health care programs.
Setback for advocates
The movement toward UDI suffered a setback in June.
An influential, but little known federal advisory panel, the National Committee on Vital Health Statistics (NCVHS), opted to not recommend use of this information in claims, saying the FDA should consider the matter further.
Gaining an NCVHS recommendation would have been a win, said Sen. Elizabeth Warren (D-MA), Sen. Charles E. Grassley (R-IA), and Rep. Bill Pascrell Jr. (D-NJ), in a December 2022 letter to the panel.
Including UDI data would let researchers track patients’ interactions with a health system and could be used to establish population-level correlations between a particular device and a long-term outcome or side effect, the lawmakers said.
That view had the support of at least one major maker of devices, Cook Group, which sells products for a variety of specialties, including cardiology.
In a comment to NCVHS, Cook urged for the inclusion identifiers in Medicare claims.
“While some have argued that the UDI is better suited for inclusion in the electronic health records, Cook believes this argument sets up a false choice between the two,” wrote Stephen L. Ferguson, JD, the chairman of Cook’s board. “Inclusion of the UDI in both electronic health records and claims forms will lead to a more robust system of real-world data.”
In contrast, AdvaMed, the trade group for device makers, told the NCVHS that it did not support adding the information to payment claims submissions, instead just supporting the inclusion in EHRs.
Dr. Kovacs of the ACC said one potential drawback to more transparency could be challenges in interpreting reports of complications in certain cases, at least initially. Reports about a flaw or even a suspected flaw in a device might lead patients to become concerned about their implanted devices, potentially registering unfounded complaints.
But this concern can be addressed through using “scientific rigor and safeguards” and is outweighed by the potential safety benefits for patients, Dr. Kovacs said.
Patients should ask health care systems to track and share information about their implanted devices, Dr. Kovacs suggested.
“I feel it would be my right to demand that that device information follows my electronic medical record, so that it’s readily available to anyone who’s taking care of me,” Dr. Kovacs said. “They would know what it is that’s in me, whether it’s a lens in my eye or a prosthesis in my hip or a highly complicated implantable cardiac electronic device.”
The Harvard study was supported by the FDA and National Institutes of Health. Authors of the study reported receiving fees from the FDA, Burroughs Wellcome Fund, and Harvard-MIT Center for Regulatory Science outside the submitted work. No other disclosures were reported. Authors of the editorial reported past and present connections with F-Prime Capital, FDA, Johnson & Johnson, the Medical Devices Innovation Consortium; the Agency for Healthcare Research and Quality; the National Heart, Lung, and Blood Institute; and Arnold Ventures, as well being an expert witness at in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against Biogen. Authors of the Viewpoint reported past and present connections with the National Evaluation System for Health Technology Coordinating Center (NESTcc), which is part of the Medical Device Innovation Consortium (MDIC); AIM North America UDI Advisory Committee, Mass General Brigham, Arnold Ventures; the Institute for Clinical and Economic Review California Technology Assessment Forum; Yale University, Johnson & Johnson, FD, Agency for Healthcare Research and Quality; the National Heart, Lung, and Blood Institute of the National Institutes of Health; as well as having been an expert witness in a qui tam suit alleging violations of the False Claims Act and Anti-Kickback Statute against.
A version of this article first appeared on Medscape.com.
Does continuity of care affect outcomes after arthroplasty?
according to new research.
In a retrospective cohort study that included more than 45,000 surgeries, the likelihood of visiting the ED within 90 days was 40% lower for patients who had unbroken continuity of primary care before knee replacement and 35% lower for patients who had unbroken continuity of primary care before hip replacement, compared with those who had several primary care clinicians. Continuity of primary care was defined as having one primary care clinician in the 3 years before surgery.
The findings highlight the critical need for improved access to primary care in Canada, lead author Lynn Lethbridge, from the department of surgery at Dalhousie University, Halifax, N.S., said in an interview. “The primary care providers have been aware that continuity of care matters, but I’m not sure that surgeons are,” she said. “If they become aware, the surgical team could develop strategies to help prevent somebody from coming back to the ED by discussing primary care provisions during the presurgical consultation.”
The study was published online in the Canadian Journal of Surgery.
Discontinuity a disadvantage
“There’s been a lot of news for the last few years here in Nova Scotia and all across Canada on the difficulties of getting and keeping a regular primary care provider, and there has also been a lot of coverage about overcrowded emergency departments and efforts to reduce the need for emergency care,” said Ms. Lethbridge. “The number of joint replacement procedures is expected to rise in the coming years, and we wanted to know if there was an association between not having a regular primary care provider and the chance of returning to the ED after joint replacement, so we undertook the study.”
The researchers accessed data on all patients in Nova Scotia who underwent nonemergency hip and knee procedures from 2005 to 2020. The study outcome was any ED visit within 90 days after discharge.
The investigators looked at 3 years of primary care history before surgery and calculated the Modified Modified Continuity Index (MMCI), which represents the number of primary care clinicians adjusted for the total number of visits, for each patient. The maximum value of the MMCI is 1, meaning that the patient saw the same doctor throughout the period examined. Lower scores mean that the patient saw different health care providers during that period.
Of the patients who underwent the 28,574 knee procedures included in the analysis, 13.9% had an ED visit within 90 days. Of the patients who underwent the 16,767 hip procedures included in the analysis, 13.5% had an ED visit within 90 days.
For patients undergoing knee procedures, the mean MMCI was 0.868, and 10.7% of patients had a perfect MMCI score of 1. For patients undergoing hip replacement, the mean MMCI was 0.864, and 13.5% had an MMCI of 1.
After controlling for confounders such as age, comorbidities, distance to hospital, and neighborhood income, the researchers found a statistically significant negative association between greater continuity of care and the probability of an ED visit.
A knee replacement patient with an MMCI score of 1 had a 90-day ED visit probability of 12.8%. A patient who had visited multiple primary care clinicians before surgery had a probability of 15.2%.
‘Quarterbacks’ needed
Commenting on the findings, Harman Chaudhry, MD, MSc, an orthopedic surgeon at Sunnybrook Health Science Center, Toronto, said: “Their main message quantified what many of us in health care already sense is true: That patients do benefit when there’s somebody, specifically a primary care provider, to quarterback their health care, look over the entirety of their care, and really know the patient.
“These are elective procedures, and joint replacement is a very siloed and episodic instance of care where we, as surgeons in hospitals, think we can control a lot of the presurgical and postsurgical care and really optimize patients,” he added. “But it’s clear that despite that, patients are still benefiting from having somebody who has known them for a longer period of time.”
The longstanding shortage of family physicians in Canada has worsened, especially since the beginning of the pandemic. The shortage means that many people are denied access to a family doctor of their own, said Dr. Chaudhry, who was not involved in the research.
“In Canada, there’s a large chunk of the population that is having difficulty accessing a primary care physician, so those patients have created their own primary care experience, taking advantage of walk-in clinics, perhaps urgent care centers, EDs when they need emergency care, and locum [tenens] physicians that may come into the community once in a while. And so, they have created their own primary care experience, but there is not a single person or single medical home where they’ve been seen for a period of years,” he said.
“Many of the patients in this study who were in the ED had nobody that the surgeon could communicate with or send his notes to,” Dr. Chaudhry concluded.
No source of funding for this study was reported. Ms. Lethbridge and Dr. Chaudhry reported no relevant financial relationships. One coauthor received travel support from Stryker and DePuy Synthes, has stock or stock options in Stryker, and participated in a data safety monitoring board for Hip Innovation Technology.
A version of this article first appeared on Medscape.com.
according to new research.
In a retrospective cohort study that included more than 45,000 surgeries, the likelihood of visiting the ED within 90 days was 40% lower for patients who had unbroken continuity of primary care before knee replacement and 35% lower for patients who had unbroken continuity of primary care before hip replacement, compared with those who had several primary care clinicians. Continuity of primary care was defined as having one primary care clinician in the 3 years before surgery.
The findings highlight the critical need for improved access to primary care in Canada, lead author Lynn Lethbridge, from the department of surgery at Dalhousie University, Halifax, N.S., said in an interview. “The primary care providers have been aware that continuity of care matters, but I’m not sure that surgeons are,” she said. “If they become aware, the surgical team could develop strategies to help prevent somebody from coming back to the ED by discussing primary care provisions during the presurgical consultation.”
The study was published online in the Canadian Journal of Surgery.
Discontinuity a disadvantage
“There’s been a lot of news for the last few years here in Nova Scotia and all across Canada on the difficulties of getting and keeping a regular primary care provider, and there has also been a lot of coverage about overcrowded emergency departments and efforts to reduce the need for emergency care,” said Ms. Lethbridge. “The number of joint replacement procedures is expected to rise in the coming years, and we wanted to know if there was an association between not having a regular primary care provider and the chance of returning to the ED after joint replacement, so we undertook the study.”
The researchers accessed data on all patients in Nova Scotia who underwent nonemergency hip and knee procedures from 2005 to 2020. The study outcome was any ED visit within 90 days after discharge.
The investigators looked at 3 years of primary care history before surgery and calculated the Modified Modified Continuity Index (MMCI), which represents the number of primary care clinicians adjusted for the total number of visits, for each patient. The maximum value of the MMCI is 1, meaning that the patient saw the same doctor throughout the period examined. Lower scores mean that the patient saw different health care providers during that period.
Of the patients who underwent the 28,574 knee procedures included in the analysis, 13.9% had an ED visit within 90 days. Of the patients who underwent the 16,767 hip procedures included in the analysis, 13.5% had an ED visit within 90 days.
For patients undergoing knee procedures, the mean MMCI was 0.868, and 10.7% of patients had a perfect MMCI score of 1. For patients undergoing hip replacement, the mean MMCI was 0.864, and 13.5% had an MMCI of 1.
After controlling for confounders such as age, comorbidities, distance to hospital, and neighborhood income, the researchers found a statistically significant negative association between greater continuity of care and the probability of an ED visit.
A knee replacement patient with an MMCI score of 1 had a 90-day ED visit probability of 12.8%. A patient who had visited multiple primary care clinicians before surgery had a probability of 15.2%.
‘Quarterbacks’ needed
Commenting on the findings, Harman Chaudhry, MD, MSc, an orthopedic surgeon at Sunnybrook Health Science Center, Toronto, said: “Their main message quantified what many of us in health care already sense is true: That patients do benefit when there’s somebody, specifically a primary care provider, to quarterback their health care, look over the entirety of their care, and really know the patient.
“These are elective procedures, and joint replacement is a very siloed and episodic instance of care where we, as surgeons in hospitals, think we can control a lot of the presurgical and postsurgical care and really optimize patients,” he added. “But it’s clear that despite that, patients are still benefiting from having somebody who has known them for a longer period of time.”
The longstanding shortage of family physicians in Canada has worsened, especially since the beginning of the pandemic. The shortage means that many people are denied access to a family doctor of their own, said Dr. Chaudhry, who was not involved in the research.
“In Canada, there’s a large chunk of the population that is having difficulty accessing a primary care physician, so those patients have created their own primary care experience, taking advantage of walk-in clinics, perhaps urgent care centers, EDs when they need emergency care, and locum [tenens] physicians that may come into the community once in a while. And so, they have created their own primary care experience, but there is not a single person or single medical home where they’ve been seen for a period of years,” he said.
“Many of the patients in this study who were in the ED had nobody that the surgeon could communicate with or send his notes to,” Dr. Chaudhry concluded.
No source of funding for this study was reported. Ms. Lethbridge and Dr. Chaudhry reported no relevant financial relationships. One coauthor received travel support from Stryker and DePuy Synthes, has stock or stock options in Stryker, and participated in a data safety monitoring board for Hip Innovation Technology.
A version of this article first appeared on Medscape.com.
according to new research.
In a retrospective cohort study that included more than 45,000 surgeries, the likelihood of visiting the ED within 90 days was 40% lower for patients who had unbroken continuity of primary care before knee replacement and 35% lower for patients who had unbroken continuity of primary care before hip replacement, compared with those who had several primary care clinicians. Continuity of primary care was defined as having one primary care clinician in the 3 years before surgery.
The findings highlight the critical need for improved access to primary care in Canada, lead author Lynn Lethbridge, from the department of surgery at Dalhousie University, Halifax, N.S., said in an interview. “The primary care providers have been aware that continuity of care matters, but I’m not sure that surgeons are,” she said. “If they become aware, the surgical team could develop strategies to help prevent somebody from coming back to the ED by discussing primary care provisions during the presurgical consultation.”
The study was published online in the Canadian Journal of Surgery.
Discontinuity a disadvantage
“There’s been a lot of news for the last few years here in Nova Scotia and all across Canada on the difficulties of getting and keeping a regular primary care provider, and there has also been a lot of coverage about overcrowded emergency departments and efforts to reduce the need for emergency care,” said Ms. Lethbridge. “The number of joint replacement procedures is expected to rise in the coming years, and we wanted to know if there was an association between not having a regular primary care provider and the chance of returning to the ED after joint replacement, so we undertook the study.”
The researchers accessed data on all patients in Nova Scotia who underwent nonemergency hip and knee procedures from 2005 to 2020. The study outcome was any ED visit within 90 days after discharge.
The investigators looked at 3 years of primary care history before surgery and calculated the Modified Modified Continuity Index (MMCI), which represents the number of primary care clinicians adjusted for the total number of visits, for each patient. The maximum value of the MMCI is 1, meaning that the patient saw the same doctor throughout the period examined. Lower scores mean that the patient saw different health care providers during that period.
Of the patients who underwent the 28,574 knee procedures included in the analysis, 13.9% had an ED visit within 90 days. Of the patients who underwent the 16,767 hip procedures included in the analysis, 13.5% had an ED visit within 90 days.
For patients undergoing knee procedures, the mean MMCI was 0.868, and 10.7% of patients had a perfect MMCI score of 1. For patients undergoing hip replacement, the mean MMCI was 0.864, and 13.5% had an MMCI of 1.
After controlling for confounders such as age, comorbidities, distance to hospital, and neighborhood income, the researchers found a statistically significant negative association between greater continuity of care and the probability of an ED visit.
A knee replacement patient with an MMCI score of 1 had a 90-day ED visit probability of 12.8%. A patient who had visited multiple primary care clinicians before surgery had a probability of 15.2%.
‘Quarterbacks’ needed
Commenting on the findings, Harman Chaudhry, MD, MSc, an orthopedic surgeon at Sunnybrook Health Science Center, Toronto, said: “Their main message quantified what many of us in health care already sense is true: That patients do benefit when there’s somebody, specifically a primary care provider, to quarterback their health care, look over the entirety of their care, and really know the patient.
“These are elective procedures, and joint replacement is a very siloed and episodic instance of care where we, as surgeons in hospitals, think we can control a lot of the presurgical and postsurgical care and really optimize patients,” he added. “But it’s clear that despite that, patients are still benefiting from having somebody who has known them for a longer period of time.”
The longstanding shortage of family physicians in Canada has worsened, especially since the beginning of the pandemic. The shortage means that many people are denied access to a family doctor of their own, said Dr. Chaudhry, who was not involved in the research.
“In Canada, there’s a large chunk of the population that is having difficulty accessing a primary care physician, so those patients have created their own primary care experience, taking advantage of walk-in clinics, perhaps urgent care centers, EDs when they need emergency care, and locum [tenens] physicians that may come into the community once in a while. And so, they have created their own primary care experience, but there is not a single person or single medical home where they’ve been seen for a period of years,” he said.
“Many of the patients in this study who were in the ED had nobody that the surgeon could communicate with or send his notes to,” Dr. Chaudhry concluded.
No source of funding for this study was reported. Ms. Lethbridge and Dr. Chaudhry reported no relevant financial relationships. One coauthor received travel support from Stryker and DePuy Synthes, has stock or stock options in Stryker, and participated in a data safety monitoring board for Hip Innovation Technology.
A version of this article first appeared on Medscape.com.
FROM THE CANADIAN JOURNAL OF SURGERY
Hidradenitis suppurativa experts reach consensus on treatment outcome measures
TOPLINE:
Hidradenitis suppurativa (HS) experts collaborated to reach consensus on a core set of outcome measures, with the intent of improving the management of HS in clinical practice.
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
Hidradenitis suppurativa (HS) experts collaborated to reach consensus on a core set of outcome measures, with the intent of improving the management of HS in clinical practice.
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
Hidradenitis suppurativa (HS) experts collaborated to reach consensus on a core set of outcome measures, with the intent of improving the management of HS in clinical practice.
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Hidradenitis suppurativa experts reach consensus on treatment outcome measures
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Participants in the study were 55 HS experts from the HiSTORIC group (dermatologists, internists, surgeons, and nurses) and 24 patient research partners.
- The group identified clinician- and patient-reported HS outcome measures in the literature, then participated in an online item reduction survey, followed by an electronic Delphi survey to reach consensus on which measures should be used in clinical practice. Consensus was defined as at least 67% of participants agreeing/strongly agreeing or disagreeing/strongly disagreeing about the use of a measure in clinical practice.
- The initial literature search yielded 11 HS studies with clinician-reported outcome measures and 12 with patient-reported outcomes; of these, eight and five, respectively, were included in the final reduction survey.
TAKEAWAY:
- The group reached consensus on two HS outcome measures for use in clinical practice: the HS Investigator Global Assessment (HS-IGA) score, a clinician-reported outcome measure selected by the HS experts, and the HS Quality of Life (HiSQOL) score, a patient-reported outcome measure selected by the patient research partners.
- The HS-IGA score uses a number between 0 and 5 based on the sum of abscesses, inflammatory and noninflammatory nodules, and tunnels in regions of the upper or lower body.
- The HiSQOL, a disease-specific quality-of-life measure for adults with HS, is designed to capture unique features of HS, including symptoms (such as pain, itch, odor, and drainage) and psychosocial outcomes and activities that may be affected by the disease.
IN PRACTICE:
“The intent of these recommendations is to provide an objective framework with both clinician and patient input that can facilitate bidirectional discussion, trust building, and decision making on the current treatment strategy and the need to adjust or escalate treatment in an appropriate time frame,” the authors wrote.
SOURCE:
The study was published online in JAMA Dermatology. The lead author was Nicole Mastacouris, MS, and the corresponding author was Amit Garg, MD, both of Northwell Health, New Hyde Park, N.Y.
LIMITATIONS:
The consensus results may have been affected by variations in HS management by region. Neither measure has been studied in clinical practice, and practice variability may limit their implementation.
DISCLOSURES:
The study was supported by grants from UCB and AbbVie. Ms. Mastacouris had no financial disclosures. Dr. Garg disclosed grant support from AbbVie and UCB during the conduct of the study, as well as personal fees from AbbVie, UCB, Aclaris Therapeutics, Anaptys Bio, Aristea Therapeutics, Boehringer Ingelheim, Bristol-Myers Squibb, Incyte, Insmed, Janssen, Novartis, Pfizer, Sonoma Biotherapeutics, Union Therapeutics, Ventyx Biosciences, and Viela Biosciences during the conduct of the study; Dr. Garg also holds patents for HS-IGA and HiSQOL. Many other coauthors disclosed relationships with multiple companies, including AbbVie and UCB, and some also disclosed patents, including patents for HiSQOL and HS Area and Severity Index.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY