User login
For MD-IQ use only
Abstracts from the Neurology Exchange 2023, a virtual event held September 19-21, 2023
Is low-molecular-weight heparin superior to aspirin for VTE prophylaxis?
ILLUSTRATIVE CASE
A 72-year-old man with well-controlled hypertension and chronic obstructive pulmonary disease is scheduled for right total hip arthroplasty (THA) due to severe arthritis. He will be admitted to the hospital overnight, and his orthopedic surgeon anticipates 2 to 3 days of inpatient recovery time. In addition to medical management of the patient’s comorbid conditions, the surgeon asks if you have any insight regarding VTE prophylaxis for this patient. Specifically, do you think aspirin is equal to LMWH for VTE prophylaxis?
All adults undergoing major orthopedic surgery are considered to be at high risk for postoperative VTE development, with those having lower-limb procedures at highest risk.2 Of the more than 2.2 million THAs and total knee arthroplasties (TKAs) performed in the United States between 2012 and 2020, 55% were primary TKAs and 39% primary THAs.3 The American College of Chest Physicians (ACCP) estimated a baseline 35-day risk for VTE of 4.3% in patients undergoing major orthopedic surgery.4 The highest VTE risk occurs during the first 7 to 14 days post surgery (1.8% for symptomatic deep vein thrombosis [DVT] and 1% for pulmonary embolism [PE]), with a slightly lower risk during the subsequent 15 to 35 days (1% for symptomatic DVT and 0.5% for PE).4
Aspirin’s low cost, availability, and ease of administration make it an attractive choice for VTE prevention in patients post THA and TKA surgery. The Pulmonary Embolism Prevention (PEP) trial evaluated 13,356 patients undergoing hip fracture repair and 4088 patients undergoing arthroplasty and found aspirin to be safe and effective in prevention of VTEs compared with placebo. The investigators concluded that “there is now good evidence for considering aspirin routinely in a wide range of surgical and medical groups at high risk of venous thromboembolism.”5 The PEP study, along with others, led to the emergence of aspirin monotherapy for VTE prophylaxis.
Current guidelines for perioperative VTE prophylaxis are based on American Society of Hematology (ASH) and ACCP recommendations. For patients undergoing THA or TKA, ASH suggests using aspirin or anticoagulants for VTE prophylaxis; when anticoagulants are used, they suggest using a direct oral anticoagulant (DOAC) over LMWH.6 The ASH guidelines are conditional recommendations based on very low certainty of effects, and the ASH panel recognized the need for further investigation with large, high-quality clinical trials.
The ACCP guidelines are clearer in recommending VTE prophylaxis vs no prophylaxis for major orthopedic surgeries and recommend the use of LMWH over other agents, including aspirin, DOACs, warfarin, and intermittent pneumatic compression (IPC) devices.4
Although prophylaxis is widely recommended to mitigate the elevated risk for VTE among patients undergoing orthopedic surgery, aspirin as monotherapy remains controversial.7 Many orthopedic surgeons prescribe aspirin as a sole VTE prophylaxis agent; however, this practice is not well supported by data from large, well-conducted, randomized trials or inferiority trials.2
STUDY SUMMARY
Aspirin did not meet the noninferiority criterion for postoperative VTE
The CRISTAL trial compared the use of aspirin vs LMWH (enoxaparin) for VTE prophylaxis in patients ages 18 years or older undergoing primary THA or TKA for osteoarthritis.1 This Australian study used a cluster-randomized, crossover, registry-nested, noninferiority trial design. Of note, in Australia, aspirin is formulated in 100-mg tablets, equivalent to the standard 81-mg low-dose tablet in the United States.
Continue to: Patients taking prescribed antiplatelet...
Patients taking prescribed antiplatelet medication for preexisting conditions (~20% of patients in each group) were allowed to continue antiplatelet therapy during the trial. Patients were excluded if they were receiving an anticoagulant prior to their procedure or had a medical contraindication to aspirin or enoxaparin.
Thirty-one hospital sites were randomly assigned a treatment protocol using either aspirin or enoxaparin. Once target patient enrollment was met with the initial assigned medication, the site switched to the second/other agent. This resulted in 5675 patients in the aspirin group and 4036 in the enoxaparin group enrolled between April 2019 and December 2020, with final follow-up in August 2021; of these, 259 in the aspirin group and 249 in the enoxaparin group were lost to follow-up, opted out, or died.
The aspirin group was given 100 mg PO daily and the enoxaparin group was given 40 mg SC daily (20 mg daily for patients weighing < 50 kg or with an estimated glomerular filtration rate < 30 mL/min/1.73 m2) for 35 days after THA and 14 days after TKA. Both treatment groups received IPC calf devices intraoperatively and postoperatively, and mobilization was offered on postoperative Day 0 or 1.
The primary outcome—development of symptomatic VTE within 90 days of the procedure—occurred in 187 (3.5%) patients in the aspirin group and 69 (1.8%) patients in the enoxaparin group (estimated difference = 1.97%; 95% CI, 0.54%-3.41%). This did not meet the noninferiority criterion for aspirin, based on an estimated assumed rate of 2% and a noninferiority margin of 1%, and in fact was statistically superior for enoxaparin (P = .007). There were no significant differences between the 2 groups in major bleeding or death within 90 days.1
WHAT’S NEW
Enoxaparin was significantly superior to aspirin for VTE prophylaxis
Although this study was designed as a noninferiority trial, analysis showed enoxaparin to be significantly superior for postoperative VTE prophylaxis compared with aspirin.
Continue to: CAVEATS
CAVEATS
Study aspirin dosing differed from US standard
This study showed significantly lower rates of symptomatic VTE in the enoxaparin group compared with the aspirin group; however, the majority of this difference was driven by rates of below-the-knee DVTs, which are clinically less relevant.8 Also, this trial used a 100-mg aspirin formulation, which is not available in the United States.
CHALLENGES TO IMPLEMENTATION
Aspirin is far cheaper and administered orally
Aspirin is significantly cheaper than enoxaparin, costing about $0.13 per dose (~$4 for 30 tablets at the 81-mg dose) vs roughly $9 per 40 mg/0.4 mL dose for enoxaparin.9 However, a cost-effectiveness analysis may be useful to determine (for example) whether the higher cost of enoxaparin may be offset by fewer DVTs and other sequelae. Lastly, LMWH is an injection, which some patients may refuse.
1. CRISTAL Study Group; Sidhu VS, Kelly TL, Pratt N, et al. Effect of aspirin vs enoxaparin on symptomatic venous thromboembolism in patients undergoing hip or knee arthroplasty: the CRISTAL randomized trial. JAMA. 2022;328:719-727. doi: 10.1001/jama.2022.13416
2. Douketis JD, Mithoowani S. Prevention of venous thromboembolism in adults undergoing hip fracture repair or hip or knee replacement. UpToDate. Updated January 25, 2023. Accessed May 24, 2023. www.uptodate.com/contents/prevention-of-venous-thromboembolism-in-adults-undergoing-hip-fracture-repair-or-hip-or-knee-replacement
3. Siddiqi A, Levine BR, Springer BD. Highlights of the 2021 American Joint Replacement Registry annual report. Arthroplast Today. 2022;13:205-207. doi: 10.1016/j.artd.2022.01.020
4. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 suppl):e278S-e325S. doi: 10.1378/chest.11-2404
5. Pulmonary Embolism Prevention (PEP) trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet. 2000;355:1295-1302. doi: 10.1016/S0140-6736(00)02110-3
6. Anderson DR, Morgano GP, Bennett C, et al. American Society of Hematology 2019 guidelines for management of venous thromboembolism: prevention of venous thromboembolism in surgical hospitalized patients. Blood Adv. 2019;3:3898-3944. doi: 10.1182/bloodadvances.2019000975
7. Matharu GS, Kunutsor SK, Judge A, et al. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180:376-384. doi: 10.1001/jamainternmed.2019.6108
8. Brett AS, Friedman RJ. Aspirin vs. enoxaparin for prophylaxis after hip or knee replacement. NEJM Journal Watch. September 15, 2022. Accessed May 24, 2023. www.jwatch.org/na55272/2022/09/15/aspirin-vs-enoxaparin-prophylaxis-after-hip-or-knee
9. Enoxaparin. GoodRx. Accessed August 7, 2023. www.goodrx.com/enoxaparin
ILLUSTRATIVE CASE
A 72-year-old man with well-controlled hypertension and chronic obstructive pulmonary disease is scheduled for right total hip arthroplasty (THA) due to severe arthritis. He will be admitted to the hospital overnight, and his orthopedic surgeon anticipates 2 to 3 days of inpatient recovery time. In addition to medical management of the patient’s comorbid conditions, the surgeon asks if you have any insight regarding VTE prophylaxis for this patient. Specifically, do you think aspirin is equal to LMWH for VTE prophylaxis?
All adults undergoing major orthopedic surgery are considered to be at high risk for postoperative VTE development, with those having lower-limb procedures at highest risk.2 Of the more than 2.2 million THAs and total knee arthroplasties (TKAs) performed in the United States between 2012 and 2020, 55% were primary TKAs and 39% primary THAs.3 The American College of Chest Physicians (ACCP) estimated a baseline 35-day risk for VTE of 4.3% in patients undergoing major orthopedic surgery.4 The highest VTE risk occurs during the first 7 to 14 days post surgery (1.8% for symptomatic deep vein thrombosis [DVT] and 1% for pulmonary embolism [PE]), with a slightly lower risk during the subsequent 15 to 35 days (1% for symptomatic DVT and 0.5% for PE).4
Aspirin’s low cost, availability, and ease of administration make it an attractive choice for VTE prevention in patients post THA and TKA surgery. The Pulmonary Embolism Prevention (PEP) trial evaluated 13,356 patients undergoing hip fracture repair and 4088 patients undergoing arthroplasty and found aspirin to be safe and effective in prevention of VTEs compared with placebo. The investigators concluded that “there is now good evidence for considering aspirin routinely in a wide range of surgical and medical groups at high risk of venous thromboembolism.”5 The PEP study, along with others, led to the emergence of aspirin monotherapy for VTE prophylaxis.
Current guidelines for perioperative VTE prophylaxis are based on American Society of Hematology (ASH) and ACCP recommendations. For patients undergoing THA or TKA, ASH suggests using aspirin or anticoagulants for VTE prophylaxis; when anticoagulants are used, they suggest using a direct oral anticoagulant (DOAC) over LMWH.6 The ASH guidelines are conditional recommendations based on very low certainty of effects, and the ASH panel recognized the need for further investigation with large, high-quality clinical trials.
The ACCP guidelines are clearer in recommending VTE prophylaxis vs no prophylaxis for major orthopedic surgeries and recommend the use of LMWH over other agents, including aspirin, DOACs, warfarin, and intermittent pneumatic compression (IPC) devices.4
Although prophylaxis is widely recommended to mitigate the elevated risk for VTE among patients undergoing orthopedic surgery, aspirin as monotherapy remains controversial.7 Many orthopedic surgeons prescribe aspirin as a sole VTE prophylaxis agent; however, this practice is not well supported by data from large, well-conducted, randomized trials or inferiority trials.2
STUDY SUMMARY
Aspirin did not meet the noninferiority criterion for postoperative VTE
The CRISTAL trial compared the use of aspirin vs LMWH (enoxaparin) for VTE prophylaxis in patients ages 18 years or older undergoing primary THA or TKA for osteoarthritis.1 This Australian study used a cluster-randomized, crossover, registry-nested, noninferiority trial design. Of note, in Australia, aspirin is formulated in 100-mg tablets, equivalent to the standard 81-mg low-dose tablet in the United States.
Continue to: Patients taking prescribed antiplatelet...
Patients taking prescribed antiplatelet medication for preexisting conditions (~20% of patients in each group) were allowed to continue antiplatelet therapy during the trial. Patients were excluded if they were receiving an anticoagulant prior to their procedure or had a medical contraindication to aspirin or enoxaparin.
Thirty-one hospital sites were randomly assigned a treatment protocol using either aspirin or enoxaparin. Once target patient enrollment was met with the initial assigned medication, the site switched to the second/other agent. This resulted in 5675 patients in the aspirin group and 4036 in the enoxaparin group enrolled between April 2019 and December 2020, with final follow-up in August 2021; of these, 259 in the aspirin group and 249 in the enoxaparin group were lost to follow-up, opted out, or died.
The aspirin group was given 100 mg PO daily and the enoxaparin group was given 40 mg SC daily (20 mg daily for patients weighing < 50 kg or with an estimated glomerular filtration rate < 30 mL/min/1.73 m2) for 35 days after THA and 14 days after TKA. Both treatment groups received IPC calf devices intraoperatively and postoperatively, and mobilization was offered on postoperative Day 0 or 1.
The primary outcome—development of symptomatic VTE within 90 days of the procedure—occurred in 187 (3.5%) patients in the aspirin group and 69 (1.8%) patients in the enoxaparin group (estimated difference = 1.97%; 95% CI, 0.54%-3.41%). This did not meet the noninferiority criterion for aspirin, based on an estimated assumed rate of 2% and a noninferiority margin of 1%, and in fact was statistically superior for enoxaparin (P = .007). There were no significant differences between the 2 groups in major bleeding or death within 90 days.1
WHAT’S NEW
Enoxaparin was significantly superior to aspirin for VTE prophylaxis
Although this study was designed as a noninferiority trial, analysis showed enoxaparin to be significantly superior for postoperative VTE prophylaxis compared with aspirin.
Continue to: CAVEATS
CAVEATS
Study aspirin dosing differed from US standard
This study showed significantly lower rates of symptomatic VTE in the enoxaparin group compared with the aspirin group; however, the majority of this difference was driven by rates of below-the-knee DVTs, which are clinically less relevant.8 Also, this trial used a 100-mg aspirin formulation, which is not available in the United States.
CHALLENGES TO IMPLEMENTATION
Aspirin is far cheaper and administered orally
Aspirin is significantly cheaper than enoxaparin, costing about $0.13 per dose (~$4 for 30 tablets at the 81-mg dose) vs roughly $9 per 40 mg/0.4 mL dose for enoxaparin.9 However, a cost-effectiveness analysis may be useful to determine (for example) whether the higher cost of enoxaparin may be offset by fewer DVTs and other sequelae. Lastly, LMWH is an injection, which some patients may refuse.
ILLUSTRATIVE CASE
A 72-year-old man with well-controlled hypertension and chronic obstructive pulmonary disease is scheduled for right total hip arthroplasty (THA) due to severe arthritis. He will be admitted to the hospital overnight, and his orthopedic surgeon anticipates 2 to 3 days of inpatient recovery time. In addition to medical management of the patient’s comorbid conditions, the surgeon asks if you have any insight regarding VTE prophylaxis for this patient. Specifically, do you think aspirin is equal to LMWH for VTE prophylaxis?
All adults undergoing major orthopedic surgery are considered to be at high risk for postoperative VTE development, with those having lower-limb procedures at highest risk.2 Of the more than 2.2 million THAs and total knee arthroplasties (TKAs) performed in the United States between 2012 and 2020, 55% were primary TKAs and 39% primary THAs.3 The American College of Chest Physicians (ACCP) estimated a baseline 35-day risk for VTE of 4.3% in patients undergoing major orthopedic surgery.4 The highest VTE risk occurs during the first 7 to 14 days post surgery (1.8% for symptomatic deep vein thrombosis [DVT] and 1% for pulmonary embolism [PE]), with a slightly lower risk during the subsequent 15 to 35 days (1% for symptomatic DVT and 0.5% for PE).4
Aspirin’s low cost, availability, and ease of administration make it an attractive choice for VTE prevention in patients post THA and TKA surgery. The Pulmonary Embolism Prevention (PEP) trial evaluated 13,356 patients undergoing hip fracture repair and 4088 patients undergoing arthroplasty and found aspirin to be safe and effective in prevention of VTEs compared with placebo. The investigators concluded that “there is now good evidence for considering aspirin routinely in a wide range of surgical and medical groups at high risk of venous thromboembolism.”5 The PEP study, along with others, led to the emergence of aspirin monotherapy for VTE prophylaxis.
Current guidelines for perioperative VTE prophylaxis are based on American Society of Hematology (ASH) and ACCP recommendations. For patients undergoing THA or TKA, ASH suggests using aspirin or anticoagulants for VTE prophylaxis; when anticoagulants are used, they suggest using a direct oral anticoagulant (DOAC) over LMWH.6 The ASH guidelines are conditional recommendations based on very low certainty of effects, and the ASH panel recognized the need for further investigation with large, high-quality clinical trials.
The ACCP guidelines are clearer in recommending VTE prophylaxis vs no prophylaxis for major orthopedic surgeries and recommend the use of LMWH over other agents, including aspirin, DOACs, warfarin, and intermittent pneumatic compression (IPC) devices.4
Although prophylaxis is widely recommended to mitigate the elevated risk for VTE among patients undergoing orthopedic surgery, aspirin as monotherapy remains controversial.7 Many orthopedic surgeons prescribe aspirin as a sole VTE prophylaxis agent; however, this practice is not well supported by data from large, well-conducted, randomized trials or inferiority trials.2
STUDY SUMMARY
Aspirin did not meet the noninferiority criterion for postoperative VTE
The CRISTAL trial compared the use of aspirin vs LMWH (enoxaparin) for VTE prophylaxis in patients ages 18 years or older undergoing primary THA or TKA for osteoarthritis.1 This Australian study used a cluster-randomized, crossover, registry-nested, noninferiority trial design. Of note, in Australia, aspirin is formulated in 100-mg tablets, equivalent to the standard 81-mg low-dose tablet in the United States.
Continue to: Patients taking prescribed antiplatelet...
Patients taking prescribed antiplatelet medication for preexisting conditions (~20% of patients in each group) were allowed to continue antiplatelet therapy during the trial. Patients were excluded if they were receiving an anticoagulant prior to their procedure or had a medical contraindication to aspirin or enoxaparin.
Thirty-one hospital sites were randomly assigned a treatment protocol using either aspirin or enoxaparin. Once target patient enrollment was met with the initial assigned medication, the site switched to the second/other agent. This resulted in 5675 patients in the aspirin group and 4036 in the enoxaparin group enrolled between April 2019 and December 2020, with final follow-up in August 2021; of these, 259 in the aspirin group and 249 in the enoxaparin group were lost to follow-up, opted out, or died.
The aspirin group was given 100 mg PO daily and the enoxaparin group was given 40 mg SC daily (20 mg daily for patients weighing < 50 kg or with an estimated glomerular filtration rate < 30 mL/min/1.73 m2) for 35 days after THA and 14 days after TKA. Both treatment groups received IPC calf devices intraoperatively and postoperatively, and mobilization was offered on postoperative Day 0 or 1.
The primary outcome—development of symptomatic VTE within 90 days of the procedure—occurred in 187 (3.5%) patients in the aspirin group and 69 (1.8%) patients in the enoxaparin group (estimated difference = 1.97%; 95% CI, 0.54%-3.41%). This did not meet the noninferiority criterion for aspirin, based on an estimated assumed rate of 2% and a noninferiority margin of 1%, and in fact was statistically superior for enoxaparin (P = .007). There were no significant differences between the 2 groups in major bleeding or death within 90 days.1
WHAT’S NEW
Enoxaparin was significantly superior to aspirin for VTE prophylaxis
Although this study was designed as a noninferiority trial, analysis showed enoxaparin to be significantly superior for postoperative VTE prophylaxis compared with aspirin.
Continue to: CAVEATS
CAVEATS
Study aspirin dosing differed from US standard
This study showed significantly lower rates of symptomatic VTE in the enoxaparin group compared with the aspirin group; however, the majority of this difference was driven by rates of below-the-knee DVTs, which are clinically less relevant.8 Also, this trial used a 100-mg aspirin formulation, which is not available in the United States.
CHALLENGES TO IMPLEMENTATION
Aspirin is far cheaper and administered orally
Aspirin is significantly cheaper than enoxaparin, costing about $0.13 per dose (~$4 for 30 tablets at the 81-mg dose) vs roughly $9 per 40 mg/0.4 mL dose for enoxaparin.9 However, a cost-effectiveness analysis may be useful to determine (for example) whether the higher cost of enoxaparin may be offset by fewer DVTs and other sequelae. Lastly, LMWH is an injection, which some patients may refuse.
1. CRISTAL Study Group; Sidhu VS, Kelly TL, Pratt N, et al. Effect of aspirin vs enoxaparin on symptomatic venous thromboembolism in patients undergoing hip or knee arthroplasty: the CRISTAL randomized trial. JAMA. 2022;328:719-727. doi: 10.1001/jama.2022.13416
2. Douketis JD, Mithoowani S. Prevention of venous thromboembolism in adults undergoing hip fracture repair or hip or knee replacement. UpToDate. Updated January 25, 2023. Accessed May 24, 2023. www.uptodate.com/contents/prevention-of-venous-thromboembolism-in-adults-undergoing-hip-fracture-repair-or-hip-or-knee-replacement
3. Siddiqi A, Levine BR, Springer BD. Highlights of the 2021 American Joint Replacement Registry annual report. Arthroplast Today. 2022;13:205-207. doi: 10.1016/j.artd.2022.01.020
4. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 suppl):e278S-e325S. doi: 10.1378/chest.11-2404
5. Pulmonary Embolism Prevention (PEP) trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet. 2000;355:1295-1302. doi: 10.1016/S0140-6736(00)02110-3
6. Anderson DR, Morgano GP, Bennett C, et al. American Society of Hematology 2019 guidelines for management of venous thromboembolism: prevention of venous thromboembolism in surgical hospitalized patients. Blood Adv. 2019;3:3898-3944. doi: 10.1182/bloodadvances.2019000975
7. Matharu GS, Kunutsor SK, Judge A, et al. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180:376-384. doi: 10.1001/jamainternmed.2019.6108
8. Brett AS, Friedman RJ. Aspirin vs. enoxaparin for prophylaxis after hip or knee replacement. NEJM Journal Watch. September 15, 2022. Accessed May 24, 2023. www.jwatch.org/na55272/2022/09/15/aspirin-vs-enoxaparin-prophylaxis-after-hip-or-knee
9. Enoxaparin. GoodRx. Accessed August 7, 2023. www.goodrx.com/enoxaparin
1. CRISTAL Study Group; Sidhu VS, Kelly TL, Pratt N, et al. Effect of aspirin vs enoxaparin on symptomatic venous thromboembolism in patients undergoing hip or knee arthroplasty: the CRISTAL randomized trial. JAMA. 2022;328:719-727. doi: 10.1001/jama.2022.13416
2. Douketis JD, Mithoowani S. Prevention of venous thromboembolism in adults undergoing hip fracture repair or hip or knee replacement. UpToDate. Updated January 25, 2023. Accessed May 24, 2023. www.uptodate.com/contents/prevention-of-venous-thromboembolism-in-adults-undergoing-hip-fracture-repair-or-hip-or-knee-replacement
3. Siddiqi A, Levine BR, Springer BD. Highlights of the 2021 American Joint Replacement Registry annual report. Arthroplast Today. 2022;13:205-207. doi: 10.1016/j.artd.2022.01.020
4. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2012;141(2 suppl):e278S-e325S. doi: 10.1378/chest.11-2404
5. Pulmonary Embolism Prevention (PEP) trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet. 2000;355:1295-1302. doi: 10.1016/S0140-6736(00)02110-3
6. Anderson DR, Morgano GP, Bennett C, et al. American Society of Hematology 2019 guidelines for management of venous thromboembolism: prevention of venous thromboembolism in surgical hospitalized patients. Blood Adv. 2019;3:3898-3944. doi: 10.1182/bloodadvances.2019000975
7. Matharu GS, Kunutsor SK, Judge A, et al. Clinical effectiveness and safety of aspirin for venous thromboembolism prophylaxis after total hip and knee replacement: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2020;180:376-384. doi: 10.1001/jamainternmed.2019.6108
8. Brett AS, Friedman RJ. Aspirin vs. enoxaparin for prophylaxis after hip or knee replacement. NEJM Journal Watch. September 15, 2022. Accessed May 24, 2023. www.jwatch.org/na55272/2022/09/15/aspirin-vs-enoxaparin-prophylaxis-after-hip-or-knee
9. Enoxaparin. GoodRx. Accessed August 7, 2023. www.goodrx.com/enoxaparin
PRACTICE CHANGER
Consider low-molecular-weight heparin (LMWH) rather than aspirin to prevent postoperative venous thromboembolism (VTE) in patients undergoing total hip or knee arthroplasty for osteoarthritis.
STRENGTH OF RECOMMENDATION
B: Based on a single cluster-randomized crossover trial.1
51-year-old woman • history of Graves disease • general fatigue, palpitations, and hand tremors • Dx?
THE CASE
A 51-year-old Japanese woman presented with fever, sore throat, and dyspnea of less than 1 day’s duration. Although she had developed general fatigue, palpitations, and tremors of the hands 2 months earlier, she had not sought medical care.
Her medical history included Graves disease, which had been diagnosed 13 years earlier. She reported that her only medication was methimazole 10 mg/d. She did not have any family history of endocrinopathies or hematologic diseases.
Physical examination revealed a body temperature of 99.7 °F; heart rate, 130 beats/min; blood pressure, 182/62 mm Hg; respiratory rate, 46 breaths/min; and oxygen saturation, 100% on room air. Pharyngeal erythema was seen. Lung sounds were clear. The patient had tremors in her hands, tenderness of the thyroid gland, and exophthalmos. No leg edema or jugular vein distension was seen.
Laboratory tests indicated hyperthyroidism, with a thyroid-stimulating hormone level < 0.01 µIU/mL (normal range, 0.5-5 µIU/mL); free T3 level, 4.87 pg/mL (normal range, 2.3-4.3 pg/mL); and free T4 level, 2.97 ng/dL (normal range, 0.9-1.7 ng/dL). The patient also had a white blood cell (WBC) count of 1020 cells/µL (normal range, 3500-9000 cells/µL) and neutrophil count of 5 cells/µL (normal range, 1500-6500 cells/µL).
Other blood cell counts were normal, and a chest x-ray did not reveal any abnormal findings. In addition, there was no evidence to suggest hematologic malignancies or congenital neutropenia.
THE DIAGNOSIS
Based on the patient’s low WBC and neutrophil counts, agranulocytosis due to antithyroid drug therapy was suspected; however, this diagnosis would be highly unusual in the context of a 13-year history of therapy. Further history taking revealed that, because of her lack of financial means, unstable living conditions, and lack of understanding of the necessity for medication adherence, the patient had not taken methimazole
In consideration of these factors, a diagnosis of exacerbation of hyperthyroidism and agranulocytosis (due to methimazole restart and upper respiratory infection) was made.
Continue to: DISCUSSION
DISCUSSION
Agranulocytosis is a severe adverse event of antithyroid agents and requires prompt diagnosis and treatment. In a 26-year study at one clinic, it occurred in approximately 0.4% of patients taking antithyroid agents.1 The possible mechanisms of agranulocytosis are the direct toxicity of drugs and immune-mediated responses.2 Older age, female sex, and some HLA genotypes are reported to be associated with susceptibility to agranulocytosis.2
Although the development of agranulocytosis tends to be dose related, a small dose of antithyroid agent can sometimes cause the condition.3,4 It usually occurs within the first 3 months of treatment initiation, but occasionally patients develop agranulocytosis after long-term therapy.5 Interruption and subsequent resumption of the same antithyroid drug treatment also can be a risk factor for agranulocytosis, as in this case.5
Treatment includes drug cessation, administration of broad-spectrum antibiotics if infection is suspected, and granulocyte-colony stimulating factor (G-CSF) therapy.5
Our patient was hospitalized, and methimazole was stopped immediately. Administration of potassium iodide 50 mg/d and G-CSF was started. Meropenem 3 g/d also was administered for neutropenic fever.
The patient’s condition improved, and her WBC count increased to 1640 cells/µL on Day 8 and 10,890 cells/µL on Day 9. G-CSF was stopped on Day 12 and meropenem on Day 13. Bone marrow aspiration was not performed because of improvement in lab values and her overall condition. Although monitoring of WBC count during methimazole therapy is controversial,5 we decided to routinely monitor this patient due to the possibility of drug cross-reactivity.
Continue to: Despite repeated explanations...
Despite repeated explanations that it was dangerous for a patient who had developed agranulocytosis to take another antithyroid medication, the patient refused surgical treatment or radioiodine ablation because of her financial situation. (While all Japanese citizens are covered by a national health insurance program, patients ages 6 to 70 years are required to pay approximately 30% of medical and pharmaceutical costs.) On Day 21, potassium iodide was stopped, and propylthiouracil 300 mg/d was administered with careful follow-up. Agranulocytosis did not recur.
Immediate problem solved, but what about the future?
During her hospital stay, the medical team spoke with the patient many times, during which she expressed anxiety about her health conditions and the difficulties that she had experienced in her life. The clinicians acknowledged her concerns and assured the patient of their continuing commitment to her well-being even after discharge. The patient also was advised that she should take her medication as prescribed and that if she had a fever or sore throat, she should stop the medication and seek medical care as soon as possible. The patient accepted the medical team’s advice and expressed hope for the future.
Conversations about medication adherence. In 1 survey, about 60% of patients taking antithyroid drugs were unfamiliar with the symptoms of agranulocytosis.6 To deliver safe and effective treatment and detect conditions such as agranulocytosis at an early stage, clinicians must communicate clearly with patients who have hyperthyroidism, providing sufficient explanation and ensuring understanding on the patient’s part.
Patients may be reluctant to provide the details of medication adherence.7 Although it is common for patients to need services for socioeconomic issues,8 health care professionals sometimes fail to adequately discuss these issues with patients, especially if the patients are marginalized and/or have lower economic status.9 Cases such as ours underscore the importance of improving clinicians’ awareness and sensitivity to patients’ socioeconomic challenges.10,11
Our patient received information about welfare and other government services from a medical social worker during her hospital stay. She also was informed that she could seek assistance from medical social workers in the future if needed.
Continue to: The patient was discharged...
The patient was discharged on Day 28. After discharge, she took propylthiouracil as prescribed (300 mg/d), and her Graves disease was well controlled. Outpatient follow-up visits were performed every 1 or 2 months. No adverse events of propylthiouracil were seen in the ensuing time.
THE TAKEAWAY
Patients with chronic conditions sometimes discontinue medications, and they may not talk about it with their medical team, especially if they have socioeconomic or other difficulties in their lives. Clinicians should consider medication nonadherence and its risk factors when patients with chronic conditions develop unexpected adverse events.
We thank Jane Charbonneau, DVM, from Edanz for doing an English-language review of a draft of this manuscript.
CORRESPONDENCE
Takuya Maejima, MD, Department of General Medicine and Primary Care, University of Tsukuba Hospital, 2-1-1 Amakubo, Tsukuba, Ibaraki 305-8576 Japan; t.maejima.2019@gmail.com
1. Tajiri J, Noguchi S. Antithyroid drug-induced agranulocytosis: special reference to normal white blood cell count agranulocytosis. Thyroid. 2004;14:459-462. doi: 10.1089/105072504323150787
2. Vicente N, Cardoso L, Barros L, et al. Antithyroid drug-induced agranulocytosis: state of the art on diagnosis and management. Drugs R D. 2017;17:91-96. doi: 10.1007/s40268-017-0172-1
3. Takata K, Kubota S, Fukata S, et al. Methimazole-induced agranulocytosis in patients with Graves’ disease is more frequent with an initial dose of 30 mg daily than with 15 mg daily. Thyroid. 2009;19:559-563. doi: 10.1089/thy.2008.0364
4. Tsuboi K, Ueshiba H, Shimojo M, et al. The relation of initial methimazole dose to the incidence of methimazole-induced agranulocytosis in patients with Graves’ disease. Endocr J. 2007;54:39-43. doi: 10.1507/endocrj.k05-068
5. Burch HB, Cooper DS. Management of Graves disease: a review. J Am Med Assoc. 2015;314:2544-2554. doi: 10.1001/jama.2015.16535
6. Robinson J, Richardson M, Hickey J, et al. Patient knowledge of antithyroid drug-induced agranulocytosis. Eur Thyroid J. 2014;3:245-251. doi: https://doi.org/10.1159/000367990
7. Kini V, Ho PM. Interventions to improve medication adherence: a review. J Am Med Assoc. 2018;320:2461-2473. doi: 10.1001/jama.2018.19271
8. Vest JR, Grannis SJ, Haut DP, et al. Using structured and unstructured data to identify patients’ need for services that address the social determinants of health. Int J Med Inform. 2017;107:101-106. doi: 10.1016/j.ijmedinf.2017.09.008
9. Willems S, De Maesschalck S, Deveugele M, et al. Socio-economic status of the patient and doctor-patient communication: does it make a difference? Patient Educ Couns. 2005;56:139-146. doi: 10.1016/j.pec.2004.02.011
10. The College of Family Physicians of Canada. Best advice: social determinants of health. Accessed September 15, 2023. https://patientsmedicalhome.ca/resources/best-advice-guides/best-advice-guide-social-determinants-health/
11. Hunter K, Thomson B. A scoping review of social determinants of health curricula in post-graduate medical education. Can Med Educ J. 2019;10:e61-e71. doi: 10.36834/cmej.61709
THE CASE
A 51-year-old Japanese woman presented with fever, sore throat, and dyspnea of less than 1 day’s duration. Although she had developed general fatigue, palpitations, and tremors of the hands 2 months earlier, she had not sought medical care.
Her medical history included Graves disease, which had been diagnosed 13 years earlier. She reported that her only medication was methimazole 10 mg/d. She did not have any family history of endocrinopathies or hematologic diseases.
Physical examination revealed a body temperature of 99.7 °F; heart rate, 130 beats/min; blood pressure, 182/62 mm Hg; respiratory rate, 46 breaths/min; and oxygen saturation, 100% on room air. Pharyngeal erythema was seen. Lung sounds were clear. The patient had tremors in her hands, tenderness of the thyroid gland, and exophthalmos. No leg edema or jugular vein distension was seen.
Laboratory tests indicated hyperthyroidism, with a thyroid-stimulating hormone level < 0.01 µIU/mL (normal range, 0.5-5 µIU/mL); free T3 level, 4.87 pg/mL (normal range, 2.3-4.3 pg/mL); and free T4 level, 2.97 ng/dL (normal range, 0.9-1.7 ng/dL). The patient also had a white blood cell (WBC) count of 1020 cells/µL (normal range, 3500-9000 cells/µL) and neutrophil count of 5 cells/µL (normal range, 1500-6500 cells/µL).
Other blood cell counts were normal, and a chest x-ray did not reveal any abnormal findings. In addition, there was no evidence to suggest hematologic malignancies or congenital neutropenia.
THE DIAGNOSIS
Based on the patient’s low WBC and neutrophil counts, agranulocytosis due to antithyroid drug therapy was suspected; however, this diagnosis would be highly unusual in the context of a 13-year history of therapy. Further history taking revealed that, because of her lack of financial means, unstable living conditions, and lack of understanding of the necessity for medication adherence, the patient had not taken methimazole
In consideration of these factors, a diagnosis of exacerbation of hyperthyroidism and agranulocytosis (due to methimazole restart and upper respiratory infection) was made.
Continue to: DISCUSSION
DISCUSSION
Agranulocytosis is a severe adverse event of antithyroid agents and requires prompt diagnosis and treatment. In a 26-year study at one clinic, it occurred in approximately 0.4% of patients taking antithyroid agents.1 The possible mechanisms of agranulocytosis are the direct toxicity of drugs and immune-mediated responses.2 Older age, female sex, and some HLA genotypes are reported to be associated with susceptibility to agranulocytosis.2
Although the development of agranulocytosis tends to be dose related, a small dose of antithyroid agent can sometimes cause the condition.3,4 It usually occurs within the first 3 months of treatment initiation, but occasionally patients develop agranulocytosis after long-term therapy.5 Interruption and subsequent resumption of the same antithyroid drug treatment also can be a risk factor for agranulocytosis, as in this case.5
Treatment includes drug cessation, administration of broad-spectrum antibiotics if infection is suspected, and granulocyte-colony stimulating factor (G-CSF) therapy.5
Our patient was hospitalized, and methimazole was stopped immediately. Administration of potassium iodide 50 mg/d and G-CSF was started. Meropenem 3 g/d also was administered for neutropenic fever.
The patient’s condition improved, and her WBC count increased to 1640 cells/µL on Day 8 and 10,890 cells/µL on Day 9. G-CSF was stopped on Day 12 and meropenem on Day 13. Bone marrow aspiration was not performed because of improvement in lab values and her overall condition. Although monitoring of WBC count during methimazole therapy is controversial,5 we decided to routinely monitor this patient due to the possibility of drug cross-reactivity.
Continue to: Despite repeated explanations...
Despite repeated explanations that it was dangerous for a patient who had developed agranulocytosis to take another antithyroid medication, the patient refused surgical treatment or radioiodine ablation because of her financial situation. (While all Japanese citizens are covered by a national health insurance program, patients ages 6 to 70 years are required to pay approximately 30% of medical and pharmaceutical costs.) On Day 21, potassium iodide was stopped, and propylthiouracil 300 mg/d was administered with careful follow-up. Agranulocytosis did not recur.
Immediate problem solved, but what about the future?
During her hospital stay, the medical team spoke with the patient many times, during which she expressed anxiety about her health conditions and the difficulties that she had experienced in her life. The clinicians acknowledged her concerns and assured the patient of their continuing commitment to her well-being even after discharge. The patient also was advised that she should take her medication as prescribed and that if she had a fever or sore throat, she should stop the medication and seek medical care as soon as possible. The patient accepted the medical team’s advice and expressed hope for the future.
Conversations about medication adherence. In 1 survey, about 60% of patients taking antithyroid drugs were unfamiliar with the symptoms of agranulocytosis.6 To deliver safe and effective treatment and detect conditions such as agranulocytosis at an early stage, clinicians must communicate clearly with patients who have hyperthyroidism, providing sufficient explanation and ensuring understanding on the patient’s part.
Patients may be reluctant to provide the details of medication adherence.7 Although it is common for patients to need services for socioeconomic issues,8 health care professionals sometimes fail to adequately discuss these issues with patients, especially if the patients are marginalized and/or have lower economic status.9 Cases such as ours underscore the importance of improving clinicians’ awareness and sensitivity to patients’ socioeconomic challenges.10,11
Our patient received information about welfare and other government services from a medical social worker during her hospital stay. She also was informed that she could seek assistance from medical social workers in the future if needed.
Continue to: The patient was discharged...
The patient was discharged on Day 28. After discharge, she took propylthiouracil as prescribed (300 mg/d), and her Graves disease was well controlled. Outpatient follow-up visits were performed every 1 or 2 months. No adverse events of propylthiouracil were seen in the ensuing time.
THE TAKEAWAY
Patients with chronic conditions sometimes discontinue medications, and they may not talk about it with their medical team, especially if they have socioeconomic or other difficulties in their lives. Clinicians should consider medication nonadherence and its risk factors when patients with chronic conditions develop unexpected adverse events.
We thank Jane Charbonneau, DVM, from Edanz for doing an English-language review of a draft of this manuscript.
CORRESPONDENCE
Takuya Maejima, MD, Department of General Medicine and Primary Care, University of Tsukuba Hospital, 2-1-1 Amakubo, Tsukuba, Ibaraki 305-8576 Japan; t.maejima.2019@gmail.com
THE CASE
A 51-year-old Japanese woman presented with fever, sore throat, and dyspnea of less than 1 day’s duration. Although she had developed general fatigue, palpitations, and tremors of the hands 2 months earlier, she had not sought medical care.
Her medical history included Graves disease, which had been diagnosed 13 years earlier. She reported that her only medication was methimazole 10 mg/d. She did not have any family history of endocrinopathies or hematologic diseases.
Physical examination revealed a body temperature of 99.7 °F; heart rate, 130 beats/min; blood pressure, 182/62 mm Hg; respiratory rate, 46 breaths/min; and oxygen saturation, 100% on room air. Pharyngeal erythema was seen. Lung sounds were clear. The patient had tremors in her hands, tenderness of the thyroid gland, and exophthalmos. No leg edema or jugular vein distension was seen.
Laboratory tests indicated hyperthyroidism, with a thyroid-stimulating hormone level < 0.01 µIU/mL (normal range, 0.5-5 µIU/mL); free T3 level, 4.87 pg/mL (normal range, 2.3-4.3 pg/mL); and free T4 level, 2.97 ng/dL (normal range, 0.9-1.7 ng/dL). The patient also had a white blood cell (WBC) count of 1020 cells/µL (normal range, 3500-9000 cells/µL) and neutrophil count of 5 cells/µL (normal range, 1500-6500 cells/µL).
Other blood cell counts were normal, and a chest x-ray did not reveal any abnormal findings. In addition, there was no evidence to suggest hematologic malignancies or congenital neutropenia.
THE DIAGNOSIS
Based on the patient’s low WBC and neutrophil counts, agranulocytosis due to antithyroid drug therapy was suspected; however, this diagnosis would be highly unusual in the context of a 13-year history of therapy. Further history taking revealed that, because of her lack of financial means, unstable living conditions, and lack of understanding of the necessity for medication adherence, the patient had not taken methimazole
In consideration of these factors, a diagnosis of exacerbation of hyperthyroidism and agranulocytosis (due to methimazole restart and upper respiratory infection) was made.
Continue to: DISCUSSION
DISCUSSION
Agranulocytosis is a severe adverse event of antithyroid agents and requires prompt diagnosis and treatment. In a 26-year study at one clinic, it occurred in approximately 0.4% of patients taking antithyroid agents.1 The possible mechanisms of agranulocytosis are the direct toxicity of drugs and immune-mediated responses.2 Older age, female sex, and some HLA genotypes are reported to be associated with susceptibility to agranulocytosis.2
Although the development of agranulocytosis tends to be dose related, a small dose of antithyroid agent can sometimes cause the condition.3,4 It usually occurs within the first 3 months of treatment initiation, but occasionally patients develop agranulocytosis after long-term therapy.5 Interruption and subsequent resumption of the same antithyroid drug treatment also can be a risk factor for agranulocytosis, as in this case.5
Treatment includes drug cessation, administration of broad-spectrum antibiotics if infection is suspected, and granulocyte-colony stimulating factor (G-CSF) therapy.5
Our patient was hospitalized, and methimazole was stopped immediately. Administration of potassium iodide 50 mg/d and G-CSF was started. Meropenem 3 g/d also was administered for neutropenic fever.
The patient’s condition improved, and her WBC count increased to 1640 cells/µL on Day 8 and 10,890 cells/µL on Day 9. G-CSF was stopped on Day 12 and meropenem on Day 13. Bone marrow aspiration was not performed because of improvement in lab values and her overall condition. Although monitoring of WBC count during methimazole therapy is controversial,5 we decided to routinely monitor this patient due to the possibility of drug cross-reactivity.
Continue to: Despite repeated explanations...
Despite repeated explanations that it was dangerous for a patient who had developed agranulocytosis to take another antithyroid medication, the patient refused surgical treatment or radioiodine ablation because of her financial situation. (While all Japanese citizens are covered by a national health insurance program, patients ages 6 to 70 years are required to pay approximately 30% of medical and pharmaceutical costs.) On Day 21, potassium iodide was stopped, and propylthiouracil 300 mg/d was administered with careful follow-up. Agranulocytosis did not recur.
Immediate problem solved, but what about the future?
During her hospital stay, the medical team spoke with the patient many times, during which she expressed anxiety about her health conditions and the difficulties that she had experienced in her life. The clinicians acknowledged her concerns and assured the patient of their continuing commitment to her well-being even after discharge. The patient also was advised that she should take her medication as prescribed and that if she had a fever or sore throat, she should stop the medication and seek medical care as soon as possible. The patient accepted the medical team’s advice and expressed hope for the future.
Conversations about medication adherence. In 1 survey, about 60% of patients taking antithyroid drugs were unfamiliar with the symptoms of agranulocytosis.6 To deliver safe and effective treatment and detect conditions such as agranulocytosis at an early stage, clinicians must communicate clearly with patients who have hyperthyroidism, providing sufficient explanation and ensuring understanding on the patient’s part.
Patients may be reluctant to provide the details of medication adherence.7 Although it is common for patients to need services for socioeconomic issues,8 health care professionals sometimes fail to adequately discuss these issues with patients, especially if the patients are marginalized and/or have lower economic status.9 Cases such as ours underscore the importance of improving clinicians’ awareness and sensitivity to patients’ socioeconomic challenges.10,11
Our patient received information about welfare and other government services from a medical social worker during her hospital stay. She also was informed that she could seek assistance from medical social workers in the future if needed.
Continue to: The patient was discharged...
The patient was discharged on Day 28. After discharge, she took propylthiouracil as prescribed (300 mg/d), and her Graves disease was well controlled. Outpatient follow-up visits were performed every 1 or 2 months. No adverse events of propylthiouracil were seen in the ensuing time.
THE TAKEAWAY
Patients with chronic conditions sometimes discontinue medications, and they may not talk about it with their medical team, especially if they have socioeconomic or other difficulties in their lives. Clinicians should consider medication nonadherence and its risk factors when patients with chronic conditions develop unexpected adverse events.
We thank Jane Charbonneau, DVM, from Edanz for doing an English-language review of a draft of this manuscript.
CORRESPONDENCE
Takuya Maejima, MD, Department of General Medicine and Primary Care, University of Tsukuba Hospital, 2-1-1 Amakubo, Tsukuba, Ibaraki 305-8576 Japan; t.maejima.2019@gmail.com
1. Tajiri J, Noguchi S. Antithyroid drug-induced agranulocytosis: special reference to normal white blood cell count agranulocytosis. Thyroid. 2004;14:459-462. doi: 10.1089/105072504323150787
2. Vicente N, Cardoso L, Barros L, et al. Antithyroid drug-induced agranulocytosis: state of the art on diagnosis and management. Drugs R D. 2017;17:91-96. doi: 10.1007/s40268-017-0172-1
3. Takata K, Kubota S, Fukata S, et al. Methimazole-induced agranulocytosis in patients with Graves’ disease is more frequent with an initial dose of 30 mg daily than with 15 mg daily. Thyroid. 2009;19:559-563. doi: 10.1089/thy.2008.0364
4. Tsuboi K, Ueshiba H, Shimojo M, et al. The relation of initial methimazole dose to the incidence of methimazole-induced agranulocytosis in patients with Graves’ disease. Endocr J. 2007;54:39-43. doi: 10.1507/endocrj.k05-068
5. Burch HB, Cooper DS. Management of Graves disease: a review. J Am Med Assoc. 2015;314:2544-2554. doi: 10.1001/jama.2015.16535
6. Robinson J, Richardson M, Hickey J, et al. Patient knowledge of antithyroid drug-induced agranulocytosis. Eur Thyroid J. 2014;3:245-251. doi: https://doi.org/10.1159/000367990
7. Kini V, Ho PM. Interventions to improve medication adherence: a review. J Am Med Assoc. 2018;320:2461-2473. doi: 10.1001/jama.2018.19271
8. Vest JR, Grannis SJ, Haut DP, et al. Using structured and unstructured data to identify patients’ need for services that address the social determinants of health. Int J Med Inform. 2017;107:101-106. doi: 10.1016/j.ijmedinf.2017.09.008
9. Willems S, De Maesschalck S, Deveugele M, et al. Socio-economic status of the patient and doctor-patient communication: does it make a difference? Patient Educ Couns. 2005;56:139-146. doi: 10.1016/j.pec.2004.02.011
10. The College of Family Physicians of Canada. Best advice: social determinants of health. Accessed September 15, 2023. https://patientsmedicalhome.ca/resources/best-advice-guides/best-advice-guide-social-determinants-health/
11. Hunter K, Thomson B. A scoping review of social determinants of health curricula in post-graduate medical education. Can Med Educ J. 2019;10:e61-e71. doi: 10.36834/cmej.61709
1. Tajiri J, Noguchi S. Antithyroid drug-induced agranulocytosis: special reference to normal white blood cell count agranulocytosis. Thyroid. 2004;14:459-462. doi: 10.1089/105072504323150787
2. Vicente N, Cardoso L, Barros L, et al. Antithyroid drug-induced agranulocytosis: state of the art on diagnosis and management. Drugs R D. 2017;17:91-96. doi: 10.1007/s40268-017-0172-1
3. Takata K, Kubota S, Fukata S, et al. Methimazole-induced agranulocytosis in patients with Graves’ disease is more frequent with an initial dose of 30 mg daily than with 15 mg daily. Thyroid. 2009;19:559-563. doi: 10.1089/thy.2008.0364
4. Tsuboi K, Ueshiba H, Shimojo M, et al. The relation of initial methimazole dose to the incidence of methimazole-induced agranulocytosis in patients with Graves’ disease. Endocr J. 2007;54:39-43. doi: 10.1507/endocrj.k05-068
5. Burch HB, Cooper DS. Management of Graves disease: a review. J Am Med Assoc. 2015;314:2544-2554. doi: 10.1001/jama.2015.16535
6. Robinson J, Richardson M, Hickey J, et al. Patient knowledge of antithyroid drug-induced agranulocytosis. Eur Thyroid J. 2014;3:245-251. doi: https://doi.org/10.1159/000367990
7. Kini V, Ho PM. Interventions to improve medication adherence: a review. J Am Med Assoc. 2018;320:2461-2473. doi: 10.1001/jama.2018.19271
8. Vest JR, Grannis SJ, Haut DP, et al. Using structured and unstructured data to identify patients’ need for services that address the social determinants of health. Int J Med Inform. 2017;107:101-106. doi: 10.1016/j.ijmedinf.2017.09.008
9. Willems S, De Maesschalck S, Deveugele M, et al. Socio-economic status of the patient and doctor-patient communication: does it make a difference? Patient Educ Couns. 2005;56:139-146. doi: 10.1016/j.pec.2004.02.011
10. The College of Family Physicians of Canada. Best advice: social determinants of health. Accessed September 15, 2023. https://patientsmedicalhome.ca/resources/best-advice-guides/best-advice-guide-social-determinants-health/
11. Hunter K, Thomson B. A scoping review of social determinants of health curricula in post-graduate medical education. Can Med Educ J. 2019;10:e61-e71. doi: 10.36834/cmej.61709
► History of Graves disease
► General fatigue, palpitations, and hand tremors
Getting PrEP to the patients who need it
More than 1.2 million Americans are living with HIV, and more than 30,000 new cases are diagnosed each year. While total incidence has declined since 2016, HIV remains a nationwide epidemic.1
Medications that prevent HIV acquisition, termed preexposure prophylaxis (PrEP), are an important tool to initiate in the primary care setting to reduce HIV transmission. However, while there are an estimated 1.2 million people eligible for PrEP, only 36% have received PrEP prescriptions.2 Several barriers that have impeded its widespread adoption include a lack of clinician knowledge and clinical resources for testing, high medication costs, and stigma around sexual health and intravenous (IV) drug use.
The value of PrEP
PrEP is chemoprophylaxis against the acquisition of HIV infection through the administration of an oral or injectable medication to people at risk for HIV. This practice began in the early 2000s, with the first oral regimen approved in 2012, and since has become an important tool in preventing HIV transmission.
When taken as prescribed, PrEP medications reduce the risk for acquiring HIV through sex by approximately 99% and can reduce the risk for acquiring HIV from injection drug use by approximately 74%.3 The US Preventive Services Task Force issued a Grade “A” recommendation to offer PrEP to people at high risk for HIV acquisition in June 2019 and reaffirmed it in a 2023 update.4
PrEP is notably distinct from postexposure prophylaxis (PEP), which is the administration of medication to prevent HIV infection after a possible exposure.
The available regimens
Regimens for PrEP include oral tablets or intramuscular (IM) injections.5 There are 3 PrEP regimens approved by the US Food and Drug Administration (FDA): tenofovir disoproxil fumarate/emtricitabine (Truvada), tenofovir alafenamide/emtricitabine (Descovy), and cabotegravir (Apretude).
Truvada is once-daily oral PrEP that was approved in 2012 and is now available in a generic formulation. Notable adverse effects of Truvada include a small negative impact on renal function and small reductions in bone mineral density; these have been noted in individual trials, but in meta-analyses such differences were not found to be statistically significant.6-8 The most common adverse effects of Truvada, experienced by up to 6% of patients, are gastrointestinal symptoms, fatigue, headache/dizziness, depression, and insomnia; most symptoms resolve within weeks.
Continue to: Descovy
Descovy is daily oral PrEP that was approved in 2019. Descovy is associated with increases in LDL and triglycerides but has less impact on renal and bone health.9 The most common adverse effect of Descovy, experienced by about 5% of patients, is diarrhea, followed by nausea.
Apretude was approved in 2021 and is a 600-mg IM injection given monthly for 2 months, then every 2 months (± 7 days). The advantages of Apretude are frequency and discreteness of dosing and the ability to use in patients with estimated creatinine clearance (eCrCl) > 15 mL/min. The most common adverse effects of Apretude are injection-site reactions, which occur in 30% to 80% of patients but are rarely significant enough to lead to discontinuation (< 2% of patients discontinue use due to injection-site reactions).10
Who should take PrEP?
The latest Centers for Disease Control and Prevention (CDC) guidelines recommend that all sexually active adults receive information about PrEP.5 Indications for PrEP are broad and summarized in the FIGURE.5
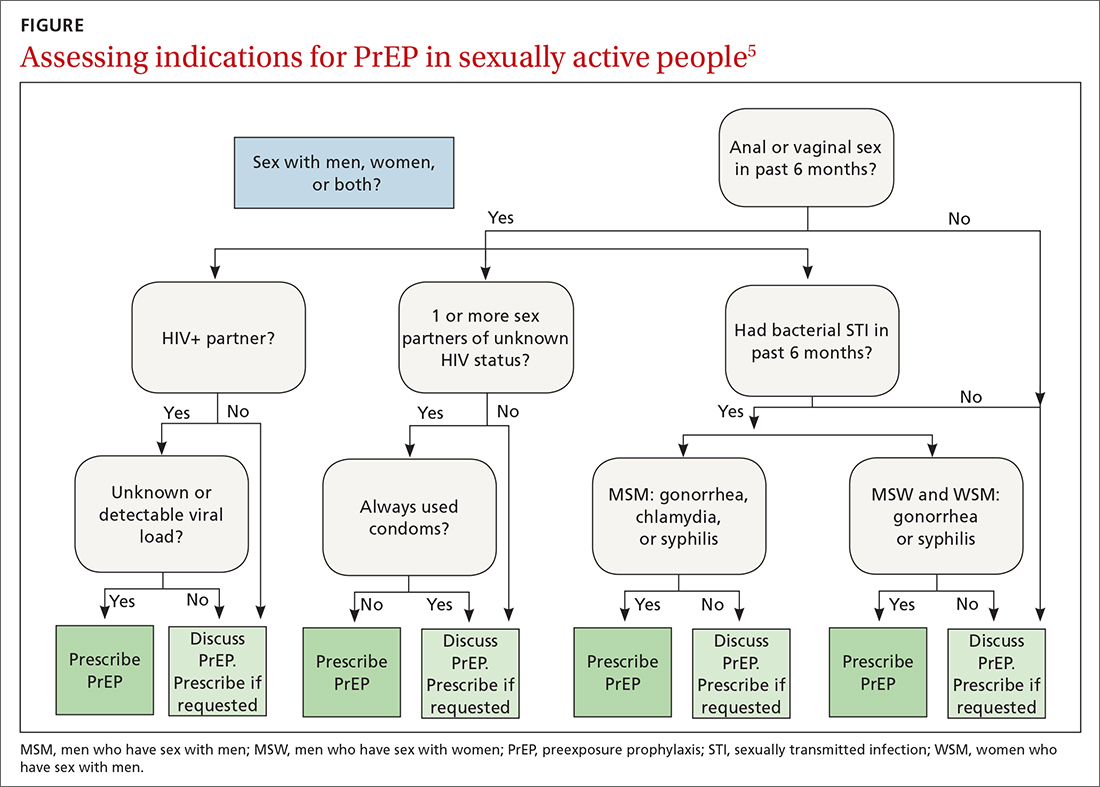
PrEP is indicated in patients who report sexual or injection drug use behaviors that place them at substantial ongoing risk for HIV exposure. Specific indications include patients with sexual partner(s) with unknown HIV status with whom they have inconsistent or no condom use, a history of bacterial sexually transmitted infection (STI) in the past 6 months, an HIV-positive sexual partner, or the sharing of injection drug equipment.
Hepatitis B infection is not a contraindication for PrEP use, but knowledge of infection status is essential. All current oral medications used for PrEP have activity against hepatitis B. Incomplete adherence to or abrupt discontinuation of oral PrEP could precipitate a hepatitis B flare. Hepatitis B surface antigen should be tested at the time of PrEP initiation, although PrEP can begin while testing is in process.
Continue to: How to use PrEP
How to use PrEP
At PrEP initiation, acute or chronic HIV infection must be excluded with a documented negative HIV antigen/antibody test within 1 week of prescribing PrEP.5 The CDC guidelines provide an updated HIV testing algorithm (www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf, p 30-31, Figures 4a and 4b), which considers whether patients have received PrEP recently.
Patients with recent high-risk exposures or symptoms of acute HIV at the time of desired PrEP initiation should have an HIV-1 viral load checked with negative results before PrEP is prescribed. Additional criteria for PrEP include weight > 35 kg; screening for hepatitis B virus infection; screening for drug interactions; and drug-specific eCrCl cutoffs of > 60 mL/min for Truvada, > 30 mL/min for Descovy, and > 15 mL/min for Apretude.5
Studies regarding time to medication effectiveness are limited. Pharmacokinetic studies of Truvada demonstrate sufficient drug concentrations should be present in peripheral blood mononuclear cells and rectal tissue within 7 days of initiation of oral dosing and around 20 days in vaginal tissue.
Of note, while expedited partner therapy is used as a harm-reduction strategy to treat the sexual partners of patients diagnosed with certain STIs, PrEP is not recommended to be used in this way.
Ongoing monitoring with PrEP. Once oral PrEP is started, STI risk assessment and HIV testing via 4th generation antibody/antigen test should be completed at least every 3 months. PrEP oral prescription refills should be limited to 3 months. For patients receiving IM PrEP (Apretude), HIV testing via viral load and antibody/antigen testing should be done at the time of each injection (every 2 months).5
Continue to: With oral PrEP...
With oral PrEP, renal function should be checked every 6 months in patients older than 50 years or those with eCrCl < 90 mL/min at initiation. For patients younger than 50 years with no baseline renal dysfunction, the latest guidelines now recommend monitoring every 12 months instead of 6 months.5
For patients on Descovy, a lipid panel is recommended at PrEP initiation and every 12 months. Testing for other STIs can be considered on this schedule, based on clinical assessment. The TABLE5 summarizes recommended monitoring for patients taking oral PrEP.
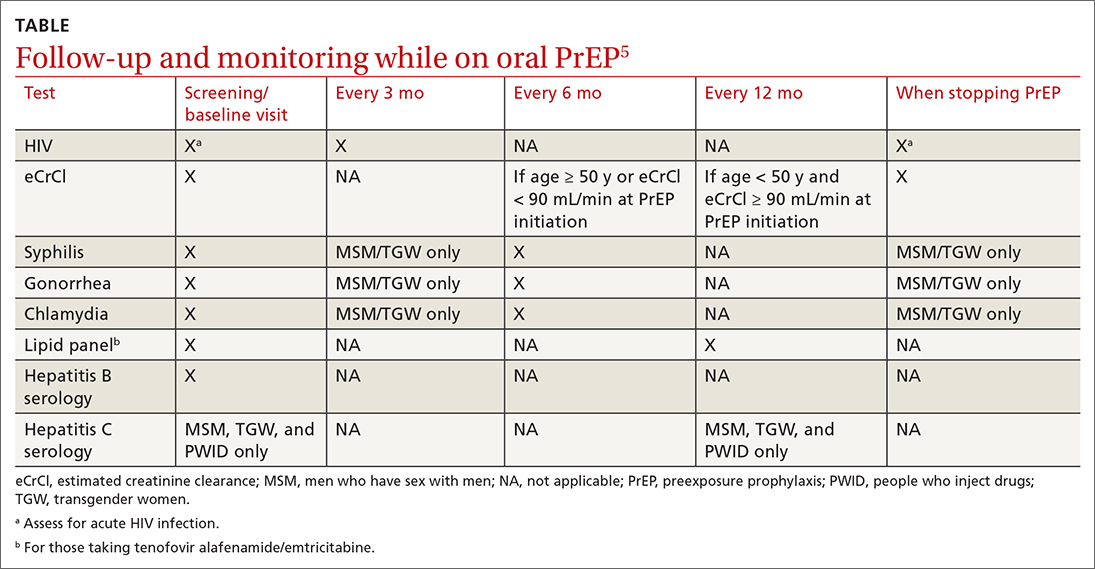
Recommended follow-up provides an opportunity to have frequent contact with a potentially high-risk population, and PrEP should be one part of a comprehensive HIV prevention and risk reduction plan. Many patients at high risk for HIV acquisition may benefit from frequent follow-up to address screening, referral, and treatment of substance use disorders, mental health conditions, and chronic medical conditions (including hepatitis C infection) and provide ongoing preventive health care.
Special uses of PrEP
Same-day PrEP. Starting PrEP on the day of the initial appointment may be appropriate based on patient risk factors and barriers to care, such as a high risk for contracting HIV before the subsequent appointment for a prescription of PrEP or an inability to return to the clinic in a timely fashion due to transportation or work constraints, or clinician availability. For these patients, assuming there is a low concern for acute or chronic HIV infection, PrEP can be initiated on the day of the initial visit.5
In these cases, point-of-care HIV and creatinine testing with same-day results should be completed. Antigen/antibody fingerstick testing or HIV-1 RNA test are preferred; oral fluid HIV testing should not be used for same-day PrEP due to its lower sensitivity for HIV detection. If same-day testing is unavailable, blood should be drawn at the visit so that HIV and creatinine testing can be completed as soon as possible.
Continue to: In addition to initial laboratory testing...
In addition to initial laboratory testing, clinics offering same-day PrEP should be able to provide: (1) assistance for patients to enroll in health insurance or a medication assistance program (eg, Ready, Set, PrEP) for those ineligible for insurance coverage, (2) rapid follow-up on all laboratory results with reliable patient contact information, and (3) follow-up appointments with clinicians able to prescribe and administer PrEP medications.
Off-label “on-demand” PrEP. An off-label treatment regimen for men who have sex with men (MSM) is termed “on-demand” PrEP or “2-1-1 PrEP” and is included in the CDC guidelines for consideration by clinicians.5 This alternative dosing schedule can be used for individuals who have sex less frequently and in a more planned fashion.
On-demand PrEP requires a patient to take 2 tablets of Truvada 2 to 24 hours before sex, followed by 1 tablet 24 hours and 1 tablet 48 hours after sexual activity. If a sexual act occurs at 48 hours, the patient should extend the daily dose for 48 additional hours, such that PrEP is always used daily for 48 hours after the last sex act.
This method has been studied with Truvada in MSM in Europe and Canada through the IPERGAY and PREVENIR trials and shown to have ≥ 86% efficacy in preventing HIV acquisition.11,12 The only US-based study showed lower efficacy; however, based on the currently available data, the International Antiviral Society-USA Panel has recommended it as an alternative regimen.13,14
PrEP via telehealth. Visits for PrEP initiation and continuation can be completed via telehealth.5 Patients then can complete necessary laboratory tests by going to a physical laboratory location or using mailed specimen kits in which they can self-collect urine, oral/rectal swabs, and fingerstick blood samples.
Continue to: PrEP use in specific populations
PrEP use in specific populations
Adolescents
Truvada, Descovy, and Apretude all are now approved for use in adolescents weighing ≥ 35 kg. Two important considerations when prescribing to this population are the effects of Truvada on bone health and the unique barriers to access.
In studies of adolescent MSM using Truvada for PrEP, bone mineral density declined, especially among those ages 15 to 19 years.15 As such, the clinical impact of decreased bone mineral density should be weighed against the risk for HIV acquisition; however, bone mineral density monitoring is not recommended in the current guidelines. CDC guidelines suggest considering Descovy for male adolescents given its potential lower impact on bone mineral density.5
Confidentiality and legal issues exist when prescribing PrEP to minors. In terms of parental/guardian involvement, clinicians who are prescribing PrEP for patients younger than 18 years should consult the CDC website for guidance on local and state regulations that govern prescribing and confidentiality (www.cdc.gov/hiv/policies/law/states/minors.html).
Insurance billing statements may lead to inadvertent disclosure of a minor’s decision to take PrEP to their legal guardian.16 Generic Truvada costs less than $100 for a 3-month supply when using goodrx.com, which may offer an alternative to insurance for medication payment.
Peripartum patients
The increased risk for HIV acquisition in the peripartum period for female patients is well documented.17 Guidelines recommend offering PrEP with Truvada to female patients at risk for conception, currently pregnant, or breastfeeding when that patient’s partner has HIV and the partner’s viral load is unknown or detectable. Descovy is not recommended for pregnant or breastfeeding patients.5 Cabotegravir-containing regimens (Apretude) have not been approved by the FDA for pregnant or breastfeeding patients.5
Continue to: Data on the impact of...
Data on the impact of Truvada for PrEP on fetal health are still emerging. A large study in Kenya showed no significant differences in preterm birth, low birth weight, or early infant growth, and a randomized, noninferiority trial in South Africa showed no association between Truvada for PrEP and preterm birth or the birth of small-for-gestational-age infants.18,19 There are no definitive studies of breastfeeding infants exposed to Truvada, but data from previous trials of breastfeeding mothers who were taking the individual components that are combined in the Truvada pill indicated there is minimal medication exposure to the infant.5
PrEP studies in the peripartum period to date have been conducted exclusively among cisgender women, and data do not yet reflect the experiences of transgender men, genderqueer people, and nonbinary individuals in the peripartum period.5
Transgender people
Transgender women should be strongly considered candidates for PrEP as they are at an extremely high risk for HIV acquisition. The most recent National HIV Behavioral Surveillance survey found that approximately 42% of transgender women were living with HIV.20 The survey revealed stark racial and ethnic disparities among transgender women living with HIV: 62% identified as Black/African American, compared with 35% Hispanic/Latina and 17% White.20
Transgender women report high rates of sexual assault, unprotected receptive anal sex, commercial sex work, homelessness, mental health disorders, and substance use, putting them at increased risk for HIV acquisition.21 However, transgender women are less likely to have discussed PrEP with a clinician, are less likely to be on PrEP even when interested in starting, and have higher rates of medication nonadherence compared with cisgender MSM.21,22 PrEP has not been found to decrease levels of feminizing hormones; however, studies are mixed as to whether feminizing hormones decrease Truvada concentrations in rectal mucosa, so clinicians should emphasize the importance of daily medication adherence.23
Transgender men have not been included in any PrEP trials, so no specific recommendations are available.
Continue to: Disparities in PrEP access and use exist
Disparities in PrEP access and use exist
The lifetime risk for HIV acquisition is 9% among White MSM, 50% among Black MSM, and 20% among Hispanic MSM.24 Despite this large disparity in disease burden, Black and Hispanic individuals are less likely to be aware of PrEP, have discussed PrEP with a health care professional, or used PrEP compared with their White counterparts.25 As a result, in 2020, PrEP coverage for eligible White individuals was 61%, while coverage among eligible Black and Hispanic/Latino individuals was just 8% and 14%, respectively.26
Surveillance data comparing male and female PrEP coverage reveal further disparities between the sexes, with PrEP coverage for eligible female-at-birth patients estimated to be 9% compared with 25.8% for male-at-birth patients.26 The gap between the risk for HIV infection and the access to and uptake of PrEP coverage is most pronounced among Black women, for whom the rate of new HIV diagnosis is > 10 times higher than it is for White women, but who have some of the lowest awareness and utilization rates of all demographics.27
The rural population at risk. Disparities in HIV awareness and PrEP use also exist between rural and urban populations, as well as by health insurance status. Rural areas have been shown to lag behind urban areas in PrEP awareness and use. Two potential explanations for this disparity are differences in HIV- and drug use–associated stigma and health insurance status. Greater stigma against drug use and HIV in rural areas has been associated with lower rates of PrEP use.28
Individuals younger than 65 years in rural areas are less likely to have private health insurance and more likely to be uninsured compared with their urban counterparts, which may impact access to clinicians knowledgeable about PrEP.29 Notably, MSM who live in states that have expanded Medicaid have higher rates of PrEP use compared with MSM living in states that have not expanded Medicaid.30
Health insurers in the United States are required to cover PrEP medication, clinician visits, and associated blood work with no patient cost-sharing, although implementation barriers such as prior authorizations still exist.
Conclusion
Family physicians are well positioned to identify patients at risk for HIV infection, prescribe PrEP, organize comprehensive follow-up care, and partner with their health systems and local communities to reduce barriers to care. Those who can leverage existing relationships with local health departments, school-based health clinics, congregate housing programs, LGBTQIA+ advocacy groups, harm-reduction coalitions, and other community-based organizations to raise PrEP awareness play a critical role in preventing HIV transmission and reducing health care disparities in their communities.
CORRESPONDENCE
Andrew V.A. Foley, MD, MPH, Erie Family Health Centers, 2418 W Division Street, Chicago, IL 60622; andrewvafoley@gmail.com
1. CDC. Estimated HIV incidence and prevalence in the United States 2017–2021. HIV Surveill Supplemental Rep. 2023;28. Accessed October 23, 2023. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-28-no-3/index.html
2. CDC. Core indicators for monitoring the Ending the HIV Epidemic initiative (preliminary data): National HIV Surveillance System data reported through March 2023; and preexposure prophylaxis (PrEP) data reported through December 2022. HIV Surveill Data Tables. 2023;4. Published June 2023. Accessed October 23, 2023. https://www.cdc.gov/hiv/library/reports/surveillance-data-tables/
3. CDC. Division of HIV Prevention, National Center for HIV, Viral Hepatitis, STD, and TB Prevention. PrEP effectiveness. Updated June 2022. Accessed October 23, 2023. https://www.cdc.gov/hiv/basics/prep/prep-effectiveness.html
4. US Preventive Services Task Force. Prevention of acquisition of HIV: preexposure prophylaxis. Final recommendation statement. August 22, 2023. Accessed October 23, 2023. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-human-immunodeficiency-virus-hiv-infection-pre-exposure-prophylaxis
5. CDC. Preexposure prophylaxis for the prevention of HIV infection in the United States—2021 update: a clinical practice guideline. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf
6. Mugwanya KK, Wyatt C, Celum C, et al. Changes in glomerular kidney function among HIV-1-uninfected men and women receiving emtricitabine-tenofovir disoproxil fumarate preexposure prophylaxis: a randomized clinical trial. JAMA Intern Med. 2015;175:246-254. doi: 10.1001/jamainternmed.2014.6786
7. Havens PL, Stephensen CB, Van Loan MD, et al. Decline in bone mass with tenofovir disoproxil fumarate/emtricitabine is associated with hormonal changes in the absence of renal impairment when used by HIV-uninfected adolescent boys and young men for HIV preexposure prophylaxis. Clin Infect Dis. 2017;64:317-325. doi: 10.1093/cid/ciw765
8. Pilkington V, Hill A, Hughes S, et al. How safe is TDF/FTC as PrEP? A systematic review and meta-analysis of the risk of adverse events in 13 randomised trials of PrEP. J Virus Erad. 2018;4:215-224.
9. Mayer KH, Molina JM, Thompson MA, et al. Emtricitabine and tenofovir alafenamide vs emtricitabine and tenofovir disoproxil fumarate for HIV pre-exposure prophylaxis (DISCOVER): primary results from a randomised, double-blind, multicentre, active-controlled, phase 3, non-inferiority trial. Lancet. 2020;396:239-254. doi: 10.1016/S0140-6736(20)31065-5
10. Liegeon G, Ghosn, J. Long-acting injectable cabotegravir for PrEP: a game-changer in HIV prevention. HIV Med. 2022;24:653-663. doi: 10.1111/hiv.13451
11. Molina JM, Capitant C, Spire B, et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N Engl J Med. 2015;373:2237-2246. doi: 10.1056/NEJMoa1506273
12. Molina JM, Ghosn J, Assoumou L, et al. Daily and on-demand HIV pre-exposure prophylaxis with emtricitabine and tenofovir disoproxil (ANRS PREVENIR): a prospective observational cohort study. Lancet HIV. 2022;9:e554-e562. doi: 10.1016/S2352-3018(22)00133-3
13. Dimitrov D, Moore JR, Wood D, et al. Predicted effectiveness of daily and nondaily preexposure prophylaxis for men who have sex with men based on sex and pill-taking patterns from the Human Immuno Virus Prevention Trials Network 067/ADAPT Study. Clin Infect Dis. 2020;71:249-255. doi: 10.1093/cid/ciz799
14. Saag MS, Gandhi RT, Hoy JF, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2020 recommendations of the International Antiviral Society-USA Panel. JAMA. 2020;324:1651-1669. doi: 10.1001/jama.2020.17025
15. Havens PL, Perumean-Chaney SE, Patki A, et al. Changes in bone mass after discontinuation of preexposure prophylaxis with tenofovir disoproxil fumarate/emtricitabine in young men who have sex with men: extension phase results of Adolescent Trials Network Protocols 110 and 113. Clin Infect Dis. 2020;70:687-691. doi: 10.1093/cid/ciz486
16. Neilan AM, Salvant Valentine S, Knopf AS. Case 27-2021: a 16-year-old boy seeking human immunodeficiency virus prophylaxis. N Engl J Med. 2021;385:1034-1041. doi: 10.1056/NEJMcpc1909626
17. Thomson KA, Hughes J, Baeten JM, et al. Increased risk of HIV acquisition among women throughout pregnancy and during the postpartum period: a prospective per-coital-act analysis among women with HIV-infected partners. J Infect Dis. 2018;218:16-25. doi: 10.1093/infdis/jiy113
18. Dettinger JC, Kinuthia J, Pintye J, et al. Perinatal outcomes following maternal pre-exposure prophylaxis (PrEP) use during pregnancy: results from a large PrEP implementation program in Kenya. J Int AIDS Soc. 2019;22:e25378. doi: 10.1002/jia2.25378
19. Moodley D, Lombard C, Govender V, et al. Pregnancy and neonatal safety outcomes of timing of initiation of daily oral tenofovir disoproxil fumarate and emtricitabine pre-exposure prophylaxis for HIV prevention (CAP016): an open-label, randomised, non-inferiority trial. Lancet HIV. 2023;10:e154-e163. doi: 10.1016/S2352-3018(22)00369-1
20. CDC. HIV Infection, Risk, Prevention, and Testing Behaviors Among Transgender Women—National HIV Behavioral Surveillance, 7 U.S. Cities, 2019–2020. HIV Surveillance Special Report 27. April 2021. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-special-report-number-27.pdf
21. Wilson EC, Turner CM, Arayasirikul S, et al. Disparities in the PrEP continuum for trans women compared to MSM in San Francisco, California: results from population-based cross-sectional behavioural surveillance studies. J Int AIDS Soc. 2020;23:e25539. doi: 10.1002/jia2.25539
22. Poteat T, Wirtz A, Malik M, et al. A gap between willingness and uptake: findings from mixed methods research on HIV prevention among Black and Latina transgender women. J Acquir Immune Defic Syndr. 2019;82:131-140. doi: 10.1097/QAI.0000000000002112
23. Cottrell ML, Prince HM, Schauer AP, et al. Decreased tenofovir diphosphate concentrations in a transgender female cohort: implications for human immunodeficiency virus preexposure prophylaxis. Clin Infect Dis. 2019;69:2201-2204. doi: 10.1093/cid/ciz290
24. Hess KL, Hu X, Lansky A, et al. Lifetime risk of a diagnosis of HIV infection in the United States. Ann Epidemiol. 2017;27:238-243. doi: 10.1016/j.annepidem.2017.02.003
25. Kanny D, Jeffries WL 4th, Chapin-Bardales J, et al. Racial/ethnic disparities in HIV preexposure prophylaxis among men who have sex with men—23 urban areas, 2017. MMWR Morb Mortal Wkly Rep. 2019;68:801-806. doi: 10.15585/mmwr.mm6837a2
26. CDC. Core indicators for monitoring the Ending the HIV Epidemic initiative (early release): National HIV Surveillance System data reported through December 2020; and preexposure prophylaxis (PrEP) data reported through September 2020. HIV Surveill Data Tables. 2021;2. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/library/reports/surveillance-data-tables/vol-2-no-2/cdc-hiv-surveillance-tables-vol-2-no-2.pdf
27. CDC. Diagnoses of HIV infection in the United States and dependent areas 2021: special focus profiles. Updated May 23, 2023. Accessed October 23, 2023. www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-34/content/special-focus-profiles.html
28. Walters SM, Frank D, Van Ham B, et al. PrEP care continuum engagement among persons who inject drugs: rural and urban differences in stigma and social infrastructure. AIDS Behav. 2021;26:1308-1320. doi: 10.1007/s10461-021-03488-2
29. Foutz J, Artiga S, Garfield R. The role of Medicaid in rural America [issue brief]. April 25, 2017. Accessed August 16, 2023. www.kff.org/medicaid/issue-brief/the-role-of-medicaid-in-rural-america/
30. Baugher AR, Finlayson T, Lewis R, et al. Health care coverage and preexposure prophylaxis (PrEP) use among men who have sex with men living in 22 US cities with vs without Medicaid expansion, 2017. Am J Public Health. 2021;111:743-751. doi: 10.2105/AJPH.2020.306035
More than 1.2 million Americans are living with HIV, and more than 30,000 new cases are diagnosed each year. While total incidence has declined since 2016, HIV remains a nationwide epidemic.1
Medications that prevent HIV acquisition, termed preexposure prophylaxis (PrEP), are an important tool to initiate in the primary care setting to reduce HIV transmission. However, while there are an estimated 1.2 million people eligible for PrEP, only 36% have received PrEP prescriptions.2 Several barriers that have impeded its widespread adoption include a lack of clinician knowledge and clinical resources for testing, high medication costs, and stigma around sexual health and intravenous (IV) drug use.
The value of PrEP
PrEP is chemoprophylaxis against the acquisition of HIV infection through the administration of an oral or injectable medication to people at risk for HIV. This practice began in the early 2000s, with the first oral regimen approved in 2012, and since has become an important tool in preventing HIV transmission.
When taken as prescribed, PrEP medications reduce the risk for acquiring HIV through sex by approximately 99% and can reduce the risk for acquiring HIV from injection drug use by approximately 74%.3 The US Preventive Services Task Force issued a Grade “A” recommendation to offer PrEP to people at high risk for HIV acquisition in June 2019 and reaffirmed it in a 2023 update.4
PrEP is notably distinct from postexposure prophylaxis (PEP), which is the administration of medication to prevent HIV infection after a possible exposure.
The available regimens
Regimens for PrEP include oral tablets or intramuscular (IM) injections.5 There are 3 PrEP regimens approved by the US Food and Drug Administration (FDA): tenofovir disoproxil fumarate/emtricitabine (Truvada), tenofovir alafenamide/emtricitabine (Descovy), and cabotegravir (Apretude).
Truvada is once-daily oral PrEP that was approved in 2012 and is now available in a generic formulation. Notable adverse effects of Truvada include a small negative impact on renal function and small reductions in bone mineral density; these have been noted in individual trials, but in meta-analyses such differences were not found to be statistically significant.6-8 The most common adverse effects of Truvada, experienced by up to 6% of patients, are gastrointestinal symptoms, fatigue, headache/dizziness, depression, and insomnia; most symptoms resolve within weeks.
Continue to: Descovy
Descovy is daily oral PrEP that was approved in 2019. Descovy is associated with increases in LDL and triglycerides but has less impact on renal and bone health.9 The most common adverse effect of Descovy, experienced by about 5% of patients, is diarrhea, followed by nausea.
Apretude was approved in 2021 and is a 600-mg IM injection given monthly for 2 months, then every 2 months (± 7 days). The advantages of Apretude are frequency and discreteness of dosing and the ability to use in patients with estimated creatinine clearance (eCrCl) > 15 mL/min. The most common adverse effects of Apretude are injection-site reactions, which occur in 30% to 80% of patients but are rarely significant enough to lead to discontinuation (< 2% of patients discontinue use due to injection-site reactions).10
Who should take PrEP?
The latest Centers for Disease Control and Prevention (CDC) guidelines recommend that all sexually active adults receive information about PrEP.5 Indications for PrEP are broad and summarized in the FIGURE.5

PrEP is indicated in patients who report sexual or injection drug use behaviors that place them at substantial ongoing risk for HIV exposure. Specific indications include patients with sexual partner(s) with unknown HIV status with whom they have inconsistent or no condom use, a history of bacterial sexually transmitted infection (STI) in the past 6 months, an HIV-positive sexual partner, or the sharing of injection drug equipment.
Hepatitis B infection is not a contraindication for PrEP use, but knowledge of infection status is essential. All current oral medications used for PrEP have activity against hepatitis B. Incomplete adherence to or abrupt discontinuation of oral PrEP could precipitate a hepatitis B flare. Hepatitis B surface antigen should be tested at the time of PrEP initiation, although PrEP can begin while testing is in process.
Continue to: How to use PrEP
How to use PrEP
At PrEP initiation, acute or chronic HIV infection must be excluded with a documented negative HIV antigen/antibody test within 1 week of prescribing PrEP.5 The CDC guidelines provide an updated HIV testing algorithm (www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf, p 30-31, Figures 4a and 4b), which considers whether patients have received PrEP recently.
Patients with recent high-risk exposures or symptoms of acute HIV at the time of desired PrEP initiation should have an HIV-1 viral load checked with negative results before PrEP is prescribed. Additional criteria for PrEP include weight > 35 kg; screening for hepatitis B virus infection; screening for drug interactions; and drug-specific eCrCl cutoffs of > 60 mL/min for Truvada, > 30 mL/min for Descovy, and > 15 mL/min for Apretude.5
Studies regarding time to medication effectiveness are limited. Pharmacokinetic studies of Truvada demonstrate sufficient drug concentrations should be present in peripheral blood mononuclear cells and rectal tissue within 7 days of initiation of oral dosing and around 20 days in vaginal tissue.
Of note, while expedited partner therapy is used as a harm-reduction strategy to treat the sexual partners of patients diagnosed with certain STIs, PrEP is not recommended to be used in this way.
Ongoing monitoring with PrEP. Once oral PrEP is started, STI risk assessment and HIV testing via 4th generation antibody/antigen test should be completed at least every 3 months. PrEP oral prescription refills should be limited to 3 months. For patients receiving IM PrEP (Apretude), HIV testing via viral load and antibody/antigen testing should be done at the time of each injection (every 2 months).5
Continue to: With oral PrEP...
With oral PrEP, renal function should be checked every 6 months in patients older than 50 years or those with eCrCl < 90 mL/min at initiation. For patients younger than 50 years with no baseline renal dysfunction, the latest guidelines now recommend monitoring every 12 months instead of 6 months.5
For patients on Descovy, a lipid panel is recommended at PrEP initiation and every 12 months. Testing for other STIs can be considered on this schedule, based on clinical assessment. The TABLE5 summarizes recommended monitoring for patients taking oral PrEP.

Recommended follow-up provides an opportunity to have frequent contact with a potentially high-risk population, and PrEP should be one part of a comprehensive HIV prevention and risk reduction plan. Many patients at high risk for HIV acquisition may benefit from frequent follow-up to address screening, referral, and treatment of substance use disorders, mental health conditions, and chronic medical conditions (including hepatitis C infection) and provide ongoing preventive health care.
Special uses of PrEP
Same-day PrEP. Starting PrEP on the day of the initial appointment may be appropriate based on patient risk factors and barriers to care, such as a high risk for contracting HIV before the subsequent appointment for a prescription of PrEP or an inability to return to the clinic in a timely fashion due to transportation or work constraints, or clinician availability. For these patients, assuming there is a low concern for acute or chronic HIV infection, PrEP can be initiated on the day of the initial visit.5
In these cases, point-of-care HIV and creatinine testing with same-day results should be completed. Antigen/antibody fingerstick testing or HIV-1 RNA test are preferred; oral fluid HIV testing should not be used for same-day PrEP due to its lower sensitivity for HIV detection. If same-day testing is unavailable, blood should be drawn at the visit so that HIV and creatinine testing can be completed as soon as possible.
Continue to: In addition to initial laboratory testing...
In addition to initial laboratory testing, clinics offering same-day PrEP should be able to provide: (1) assistance for patients to enroll in health insurance or a medication assistance program (eg, Ready, Set, PrEP) for those ineligible for insurance coverage, (2) rapid follow-up on all laboratory results with reliable patient contact information, and (3) follow-up appointments with clinicians able to prescribe and administer PrEP medications.
Off-label “on-demand” PrEP. An off-label treatment regimen for men who have sex with men (MSM) is termed “on-demand” PrEP or “2-1-1 PrEP” and is included in the CDC guidelines for consideration by clinicians.5 This alternative dosing schedule can be used for individuals who have sex less frequently and in a more planned fashion.
On-demand PrEP requires a patient to take 2 tablets of Truvada 2 to 24 hours before sex, followed by 1 tablet 24 hours and 1 tablet 48 hours after sexual activity. If a sexual act occurs at 48 hours, the patient should extend the daily dose for 48 additional hours, such that PrEP is always used daily for 48 hours after the last sex act.
This method has been studied with Truvada in MSM in Europe and Canada through the IPERGAY and PREVENIR trials and shown to have ≥ 86% efficacy in preventing HIV acquisition.11,12 The only US-based study showed lower efficacy; however, based on the currently available data, the International Antiviral Society-USA Panel has recommended it as an alternative regimen.13,14
PrEP via telehealth. Visits for PrEP initiation and continuation can be completed via telehealth.5 Patients then can complete necessary laboratory tests by going to a physical laboratory location or using mailed specimen kits in which they can self-collect urine, oral/rectal swabs, and fingerstick blood samples.
Continue to: PrEP use in specific populations
PrEP use in specific populations
Adolescents
Truvada, Descovy, and Apretude all are now approved for use in adolescents weighing ≥ 35 kg. Two important considerations when prescribing to this population are the effects of Truvada on bone health and the unique barriers to access.
In studies of adolescent MSM using Truvada for PrEP, bone mineral density declined, especially among those ages 15 to 19 years.15 As such, the clinical impact of decreased bone mineral density should be weighed against the risk for HIV acquisition; however, bone mineral density monitoring is not recommended in the current guidelines. CDC guidelines suggest considering Descovy for male adolescents given its potential lower impact on bone mineral density.5
Confidentiality and legal issues exist when prescribing PrEP to minors. In terms of parental/guardian involvement, clinicians who are prescribing PrEP for patients younger than 18 years should consult the CDC website for guidance on local and state regulations that govern prescribing and confidentiality (www.cdc.gov/hiv/policies/law/states/minors.html).
Insurance billing statements may lead to inadvertent disclosure of a minor’s decision to take PrEP to their legal guardian.16 Generic Truvada costs less than $100 for a 3-month supply when using goodrx.com, which may offer an alternative to insurance for medication payment.
Peripartum patients
The increased risk for HIV acquisition in the peripartum period for female patients is well documented.17 Guidelines recommend offering PrEP with Truvada to female patients at risk for conception, currently pregnant, or breastfeeding when that patient’s partner has HIV and the partner’s viral load is unknown or detectable. Descovy is not recommended for pregnant or breastfeeding patients.5 Cabotegravir-containing regimens (Apretude) have not been approved by the FDA for pregnant or breastfeeding patients.5
Continue to: Data on the impact of...
Data on the impact of Truvada for PrEP on fetal health are still emerging. A large study in Kenya showed no significant differences in preterm birth, low birth weight, or early infant growth, and a randomized, noninferiority trial in South Africa showed no association between Truvada for PrEP and preterm birth or the birth of small-for-gestational-age infants.18,19 There are no definitive studies of breastfeeding infants exposed to Truvada, but data from previous trials of breastfeeding mothers who were taking the individual components that are combined in the Truvada pill indicated there is minimal medication exposure to the infant.5
PrEP studies in the peripartum period to date have been conducted exclusively among cisgender women, and data do not yet reflect the experiences of transgender men, genderqueer people, and nonbinary individuals in the peripartum period.5
Transgender people
Transgender women should be strongly considered candidates for PrEP as they are at an extremely high risk for HIV acquisition. The most recent National HIV Behavioral Surveillance survey found that approximately 42% of transgender women were living with HIV.20 The survey revealed stark racial and ethnic disparities among transgender women living with HIV: 62% identified as Black/African American, compared with 35% Hispanic/Latina and 17% White.20
Transgender women report high rates of sexual assault, unprotected receptive anal sex, commercial sex work, homelessness, mental health disorders, and substance use, putting them at increased risk for HIV acquisition.21 However, transgender women are less likely to have discussed PrEP with a clinician, are less likely to be on PrEP even when interested in starting, and have higher rates of medication nonadherence compared with cisgender MSM.21,22 PrEP has not been found to decrease levels of feminizing hormones; however, studies are mixed as to whether feminizing hormones decrease Truvada concentrations in rectal mucosa, so clinicians should emphasize the importance of daily medication adherence.23
Transgender men have not been included in any PrEP trials, so no specific recommendations are available.
Continue to: Disparities in PrEP access and use exist
Disparities in PrEP access and use exist
The lifetime risk for HIV acquisition is 9% among White MSM, 50% among Black MSM, and 20% among Hispanic MSM.24 Despite this large disparity in disease burden, Black and Hispanic individuals are less likely to be aware of PrEP, have discussed PrEP with a health care professional, or used PrEP compared with their White counterparts.25 As a result, in 2020, PrEP coverage for eligible White individuals was 61%, while coverage among eligible Black and Hispanic/Latino individuals was just 8% and 14%, respectively.26
Surveillance data comparing male and female PrEP coverage reveal further disparities between the sexes, with PrEP coverage for eligible female-at-birth patients estimated to be 9% compared with 25.8% for male-at-birth patients.26 The gap between the risk for HIV infection and the access to and uptake of PrEP coverage is most pronounced among Black women, for whom the rate of new HIV diagnosis is > 10 times higher than it is for White women, but who have some of the lowest awareness and utilization rates of all demographics.27
The rural population at risk. Disparities in HIV awareness and PrEP use also exist between rural and urban populations, as well as by health insurance status. Rural areas have been shown to lag behind urban areas in PrEP awareness and use. Two potential explanations for this disparity are differences in HIV- and drug use–associated stigma and health insurance status. Greater stigma against drug use and HIV in rural areas has been associated with lower rates of PrEP use.28
Individuals younger than 65 years in rural areas are less likely to have private health insurance and more likely to be uninsured compared with their urban counterparts, which may impact access to clinicians knowledgeable about PrEP.29 Notably, MSM who live in states that have expanded Medicaid have higher rates of PrEP use compared with MSM living in states that have not expanded Medicaid.30
Health insurers in the United States are required to cover PrEP medication, clinician visits, and associated blood work with no patient cost-sharing, although implementation barriers such as prior authorizations still exist.
Conclusion
Family physicians are well positioned to identify patients at risk for HIV infection, prescribe PrEP, organize comprehensive follow-up care, and partner with their health systems and local communities to reduce barriers to care. Those who can leverage existing relationships with local health departments, school-based health clinics, congregate housing programs, LGBTQIA+ advocacy groups, harm-reduction coalitions, and other community-based organizations to raise PrEP awareness play a critical role in preventing HIV transmission and reducing health care disparities in their communities.
CORRESPONDENCE
Andrew V.A. Foley, MD, MPH, Erie Family Health Centers, 2418 W Division Street, Chicago, IL 60622; andrewvafoley@gmail.com
More than 1.2 million Americans are living with HIV, and more than 30,000 new cases are diagnosed each year. While total incidence has declined since 2016, HIV remains a nationwide epidemic.1
Medications that prevent HIV acquisition, termed preexposure prophylaxis (PrEP), are an important tool to initiate in the primary care setting to reduce HIV transmission. However, while there are an estimated 1.2 million people eligible for PrEP, only 36% have received PrEP prescriptions.2 Several barriers that have impeded its widespread adoption include a lack of clinician knowledge and clinical resources for testing, high medication costs, and stigma around sexual health and intravenous (IV) drug use.
The value of PrEP
PrEP is chemoprophylaxis against the acquisition of HIV infection through the administration of an oral or injectable medication to people at risk for HIV. This practice began in the early 2000s, with the first oral regimen approved in 2012, and since has become an important tool in preventing HIV transmission.
When taken as prescribed, PrEP medications reduce the risk for acquiring HIV through sex by approximately 99% and can reduce the risk for acquiring HIV from injection drug use by approximately 74%.3 The US Preventive Services Task Force issued a Grade “A” recommendation to offer PrEP to people at high risk for HIV acquisition in June 2019 and reaffirmed it in a 2023 update.4
PrEP is notably distinct from postexposure prophylaxis (PEP), which is the administration of medication to prevent HIV infection after a possible exposure.
The available regimens
Regimens for PrEP include oral tablets or intramuscular (IM) injections.5 There are 3 PrEP regimens approved by the US Food and Drug Administration (FDA): tenofovir disoproxil fumarate/emtricitabine (Truvada), tenofovir alafenamide/emtricitabine (Descovy), and cabotegravir (Apretude).
Truvada is once-daily oral PrEP that was approved in 2012 and is now available in a generic formulation. Notable adverse effects of Truvada include a small negative impact on renal function and small reductions in bone mineral density; these have been noted in individual trials, but in meta-analyses such differences were not found to be statistically significant.6-8 The most common adverse effects of Truvada, experienced by up to 6% of patients, are gastrointestinal symptoms, fatigue, headache/dizziness, depression, and insomnia; most symptoms resolve within weeks.
Continue to: Descovy
Descovy is daily oral PrEP that was approved in 2019. Descovy is associated with increases in LDL and triglycerides but has less impact on renal and bone health.9 The most common adverse effect of Descovy, experienced by about 5% of patients, is diarrhea, followed by nausea.
Apretude was approved in 2021 and is a 600-mg IM injection given monthly for 2 months, then every 2 months (± 7 days). The advantages of Apretude are frequency and discreteness of dosing and the ability to use in patients with estimated creatinine clearance (eCrCl) > 15 mL/min. The most common adverse effects of Apretude are injection-site reactions, which occur in 30% to 80% of patients but are rarely significant enough to lead to discontinuation (< 2% of patients discontinue use due to injection-site reactions).10
Who should take PrEP?
The latest Centers for Disease Control and Prevention (CDC) guidelines recommend that all sexually active adults receive information about PrEP.5 Indications for PrEP are broad and summarized in the FIGURE.5

PrEP is indicated in patients who report sexual or injection drug use behaviors that place them at substantial ongoing risk for HIV exposure. Specific indications include patients with sexual partner(s) with unknown HIV status with whom they have inconsistent or no condom use, a history of bacterial sexually transmitted infection (STI) in the past 6 months, an HIV-positive sexual partner, or the sharing of injection drug equipment.
Hepatitis B infection is not a contraindication for PrEP use, but knowledge of infection status is essential. All current oral medications used for PrEP have activity against hepatitis B. Incomplete adherence to or abrupt discontinuation of oral PrEP could precipitate a hepatitis B flare. Hepatitis B surface antigen should be tested at the time of PrEP initiation, although PrEP can begin while testing is in process.
Continue to: How to use PrEP
How to use PrEP
At PrEP initiation, acute or chronic HIV infection must be excluded with a documented negative HIV antigen/antibody test within 1 week of prescribing PrEP.5 The CDC guidelines provide an updated HIV testing algorithm (www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf, p 30-31, Figures 4a and 4b), which considers whether patients have received PrEP recently.
Patients with recent high-risk exposures or symptoms of acute HIV at the time of desired PrEP initiation should have an HIV-1 viral load checked with negative results before PrEP is prescribed. Additional criteria for PrEP include weight > 35 kg; screening for hepatitis B virus infection; screening for drug interactions; and drug-specific eCrCl cutoffs of > 60 mL/min for Truvada, > 30 mL/min for Descovy, and > 15 mL/min for Apretude.5
Studies regarding time to medication effectiveness are limited. Pharmacokinetic studies of Truvada demonstrate sufficient drug concentrations should be present in peripheral blood mononuclear cells and rectal tissue within 7 days of initiation of oral dosing and around 20 days in vaginal tissue.
Of note, while expedited partner therapy is used as a harm-reduction strategy to treat the sexual partners of patients diagnosed with certain STIs, PrEP is not recommended to be used in this way.
Ongoing monitoring with PrEP. Once oral PrEP is started, STI risk assessment and HIV testing via 4th generation antibody/antigen test should be completed at least every 3 months. PrEP oral prescription refills should be limited to 3 months. For patients receiving IM PrEP (Apretude), HIV testing via viral load and antibody/antigen testing should be done at the time of each injection (every 2 months).5
Continue to: With oral PrEP...
With oral PrEP, renal function should be checked every 6 months in patients older than 50 years or those with eCrCl < 90 mL/min at initiation. For patients younger than 50 years with no baseline renal dysfunction, the latest guidelines now recommend monitoring every 12 months instead of 6 months.5
For patients on Descovy, a lipid panel is recommended at PrEP initiation and every 12 months. Testing for other STIs can be considered on this schedule, based on clinical assessment. The TABLE5 summarizes recommended monitoring for patients taking oral PrEP.

Recommended follow-up provides an opportunity to have frequent contact with a potentially high-risk population, and PrEP should be one part of a comprehensive HIV prevention and risk reduction plan. Many patients at high risk for HIV acquisition may benefit from frequent follow-up to address screening, referral, and treatment of substance use disorders, mental health conditions, and chronic medical conditions (including hepatitis C infection) and provide ongoing preventive health care.
Special uses of PrEP
Same-day PrEP. Starting PrEP on the day of the initial appointment may be appropriate based on patient risk factors and barriers to care, such as a high risk for contracting HIV before the subsequent appointment for a prescription of PrEP or an inability to return to the clinic in a timely fashion due to transportation or work constraints, or clinician availability. For these patients, assuming there is a low concern for acute or chronic HIV infection, PrEP can be initiated on the day of the initial visit.5
In these cases, point-of-care HIV and creatinine testing with same-day results should be completed. Antigen/antibody fingerstick testing or HIV-1 RNA test are preferred; oral fluid HIV testing should not be used for same-day PrEP due to its lower sensitivity for HIV detection. If same-day testing is unavailable, blood should be drawn at the visit so that HIV and creatinine testing can be completed as soon as possible.
Continue to: In addition to initial laboratory testing...
In addition to initial laboratory testing, clinics offering same-day PrEP should be able to provide: (1) assistance for patients to enroll in health insurance or a medication assistance program (eg, Ready, Set, PrEP) for those ineligible for insurance coverage, (2) rapid follow-up on all laboratory results with reliable patient contact information, and (3) follow-up appointments with clinicians able to prescribe and administer PrEP medications.
Off-label “on-demand” PrEP. An off-label treatment regimen for men who have sex with men (MSM) is termed “on-demand” PrEP or “2-1-1 PrEP” and is included in the CDC guidelines for consideration by clinicians.5 This alternative dosing schedule can be used for individuals who have sex less frequently and in a more planned fashion.
On-demand PrEP requires a patient to take 2 tablets of Truvada 2 to 24 hours before sex, followed by 1 tablet 24 hours and 1 tablet 48 hours after sexual activity. If a sexual act occurs at 48 hours, the patient should extend the daily dose for 48 additional hours, such that PrEP is always used daily for 48 hours after the last sex act.
This method has been studied with Truvada in MSM in Europe and Canada through the IPERGAY and PREVENIR trials and shown to have ≥ 86% efficacy in preventing HIV acquisition.11,12 The only US-based study showed lower efficacy; however, based on the currently available data, the International Antiviral Society-USA Panel has recommended it as an alternative regimen.13,14
PrEP via telehealth. Visits for PrEP initiation and continuation can be completed via telehealth.5 Patients then can complete necessary laboratory tests by going to a physical laboratory location or using mailed specimen kits in which they can self-collect urine, oral/rectal swabs, and fingerstick blood samples.
Continue to: PrEP use in specific populations
PrEP use in specific populations
Adolescents
Truvada, Descovy, and Apretude all are now approved for use in adolescents weighing ≥ 35 kg. Two important considerations when prescribing to this population are the effects of Truvada on bone health and the unique barriers to access.
In studies of adolescent MSM using Truvada for PrEP, bone mineral density declined, especially among those ages 15 to 19 years.15 As such, the clinical impact of decreased bone mineral density should be weighed against the risk for HIV acquisition; however, bone mineral density monitoring is not recommended in the current guidelines. CDC guidelines suggest considering Descovy for male adolescents given its potential lower impact on bone mineral density.5
Confidentiality and legal issues exist when prescribing PrEP to minors. In terms of parental/guardian involvement, clinicians who are prescribing PrEP for patients younger than 18 years should consult the CDC website for guidance on local and state regulations that govern prescribing and confidentiality (www.cdc.gov/hiv/policies/law/states/minors.html).
Insurance billing statements may lead to inadvertent disclosure of a minor’s decision to take PrEP to their legal guardian.16 Generic Truvada costs less than $100 for a 3-month supply when using goodrx.com, which may offer an alternative to insurance for medication payment.
Peripartum patients
The increased risk for HIV acquisition in the peripartum period for female patients is well documented.17 Guidelines recommend offering PrEP with Truvada to female patients at risk for conception, currently pregnant, or breastfeeding when that patient’s partner has HIV and the partner’s viral load is unknown or detectable. Descovy is not recommended for pregnant or breastfeeding patients.5 Cabotegravir-containing regimens (Apretude) have not been approved by the FDA for pregnant or breastfeeding patients.5
Continue to: Data on the impact of...
Data on the impact of Truvada for PrEP on fetal health are still emerging. A large study in Kenya showed no significant differences in preterm birth, low birth weight, or early infant growth, and a randomized, noninferiority trial in South Africa showed no association between Truvada for PrEP and preterm birth or the birth of small-for-gestational-age infants.18,19 There are no definitive studies of breastfeeding infants exposed to Truvada, but data from previous trials of breastfeeding mothers who were taking the individual components that are combined in the Truvada pill indicated there is minimal medication exposure to the infant.5
PrEP studies in the peripartum period to date have been conducted exclusively among cisgender women, and data do not yet reflect the experiences of transgender men, genderqueer people, and nonbinary individuals in the peripartum period.5
Transgender people
Transgender women should be strongly considered candidates for PrEP as they are at an extremely high risk for HIV acquisition. The most recent National HIV Behavioral Surveillance survey found that approximately 42% of transgender women were living with HIV.20 The survey revealed stark racial and ethnic disparities among transgender women living with HIV: 62% identified as Black/African American, compared with 35% Hispanic/Latina and 17% White.20
Transgender women report high rates of sexual assault, unprotected receptive anal sex, commercial sex work, homelessness, mental health disorders, and substance use, putting them at increased risk for HIV acquisition.21 However, transgender women are less likely to have discussed PrEP with a clinician, are less likely to be on PrEP even when interested in starting, and have higher rates of medication nonadherence compared with cisgender MSM.21,22 PrEP has not been found to decrease levels of feminizing hormones; however, studies are mixed as to whether feminizing hormones decrease Truvada concentrations in rectal mucosa, so clinicians should emphasize the importance of daily medication adherence.23
Transgender men have not been included in any PrEP trials, so no specific recommendations are available.
Continue to: Disparities in PrEP access and use exist
Disparities in PrEP access and use exist
The lifetime risk for HIV acquisition is 9% among White MSM, 50% among Black MSM, and 20% among Hispanic MSM.24 Despite this large disparity in disease burden, Black and Hispanic individuals are less likely to be aware of PrEP, have discussed PrEP with a health care professional, or used PrEP compared with their White counterparts.25 As a result, in 2020, PrEP coverage for eligible White individuals was 61%, while coverage among eligible Black and Hispanic/Latino individuals was just 8% and 14%, respectively.26
Surveillance data comparing male and female PrEP coverage reveal further disparities between the sexes, with PrEP coverage for eligible female-at-birth patients estimated to be 9% compared with 25.8% for male-at-birth patients.26 The gap between the risk for HIV infection and the access to and uptake of PrEP coverage is most pronounced among Black women, for whom the rate of new HIV diagnosis is > 10 times higher than it is for White women, but who have some of the lowest awareness and utilization rates of all demographics.27
The rural population at risk. Disparities in HIV awareness and PrEP use also exist between rural and urban populations, as well as by health insurance status. Rural areas have been shown to lag behind urban areas in PrEP awareness and use. Two potential explanations for this disparity are differences in HIV- and drug use–associated stigma and health insurance status. Greater stigma against drug use and HIV in rural areas has been associated with lower rates of PrEP use.28
Individuals younger than 65 years in rural areas are less likely to have private health insurance and more likely to be uninsured compared with their urban counterparts, which may impact access to clinicians knowledgeable about PrEP.29 Notably, MSM who live in states that have expanded Medicaid have higher rates of PrEP use compared with MSM living in states that have not expanded Medicaid.30
Health insurers in the United States are required to cover PrEP medication, clinician visits, and associated blood work with no patient cost-sharing, although implementation barriers such as prior authorizations still exist.
Conclusion
Family physicians are well positioned to identify patients at risk for HIV infection, prescribe PrEP, organize comprehensive follow-up care, and partner with their health systems and local communities to reduce barriers to care. Those who can leverage existing relationships with local health departments, school-based health clinics, congregate housing programs, LGBTQIA+ advocacy groups, harm-reduction coalitions, and other community-based organizations to raise PrEP awareness play a critical role in preventing HIV transmission and reducing health care disparities in their communities.
CORRESPONDENCE
Andrew V.A. Foley, MD, MPH, Erie Family Health Centers, 2418 W Division Street, Chicago, IL 60622; andrewvafoley@gmail.com
1. CDC. Estimated HIV incidence and prevalence in the United States 2017–2021. HIV Surveill Supplemental Rep. 2023;28. Accessed October 23, 2023. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-28-no-3/index.html
2. CDC. Core indicators for monitoring the Ending the HIV Epidemic initiative (preliminary data): National HIV Surveillance System data reported through March 2023; and preexposure prophylaxis (PrEP) data reported through December 2022. HIV Surveill Data Tables. 2023;4. Published June 2023. Accessed October 23, 2023. https://www.cdc.gov/hiv/library/reports/surveillance-data-tables/
3. CDC. Division of HIV Prevention, National Center for HIV, Viral Hepatitis, STD, and TB Prevention. PrEP effectiveness. Updated June 2022. Accessed October 23, 2023. https://www.cdc.gov/hiv/basics/prep/prep-effectiveness.html
4. US Preventive Services Task Force. Prevention of acquisition of HIV: preexposure prophylaxis. Final recommendation statement. August 22, 2023. Accessed October 23, 2023. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-human-immunodeficiency-virus-hiv-infection-pre-exposure-prophylaxis
5. CDC. Preexposure prophylaxis for the prevention of HIV infection in the United States—2021 update: a clinical practice guideline. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf
6. Mugwanya KK, Wyatt C, Celum C, et al. Changes in glomerular kidney function among HIV-1-uninfected men and women receiving emtricitabine-tenofovir disoproxil fumarate preexposure prophylaxis: a randomized clinical trial. JAMA Intern Med. 2015;175:246-254. doi: 10.1001/jamainternmed.2014.6786
7. Havens PL, Stephensen CB, Van Loan MD, et al. Decline in bone mass with tenofovir disoproxil fumarate/emtricitabine is associated with hormonal changes in the absence of renal impairment when used by HIV-uninfected adolescent boys and young men for HIV preexposure prophylaxis. Clin Infect Dis. 2017;64:317-325. doi: 10.1093/cid/ciw765
8. Pilkington V, Hill A, Hughes S, et al. How safe is TDF/FTC as PrEP? A systematic review and meta-analysis of the risk of adverse events in 13 randomised trials of PrEP. J Virus Erad. 2018;4:215-224.
9. Mayer KH, Molina JM, Thompson MA, et al. Emtricitabine and tenofovir alafenamide vs emtricitabine and tenofovir disoproxil fumarate for HIV pre-exposure prophylaxis (DISCOVER): primary results from a randomised, double-blind, multicentre, active-controlled, phase 3, non-inferiority trial. Lancet. 2020;396:239-254. doi: 10.1016/S0140-6736(20)31065-5
10. Liegeon G, Ghosn, J. Long-acting injectable cabotegravir for PrEP: a game-changer in HIV prevention. HIV Med. 2022;24:653-663. doi: 10.1111/hiv.13451
11. Molina JM, Capitant C, Spire B, et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N Engl J Med. 2015;373:2237-2246. doi: 10.1056/NEJMoa1506273
12. Molina JM, Ghosn J, Assoumou L, et al. Daily and on-demand HIV pre-exposure prophylaxis with emtricitabine and tenofovir disoproxil (ANRS PREVENIR): a prospective observational cohort study. Lancet HIV. 2022;9:e554-e562. doi: 10.1016/S2352-3018(22)00133-3
13. Dimitrov D, Moore JR, Wood D, et al. Predicted effectiveness of daily and nondaily preexposure prophylaxis for men who have sex with men based on sex and pill-taking patterns from the Human Immuno Virus Prevention Trials Network 067/ADAPT Study. Clin Infect Dis. 2020;71:249-255. doi: 10.1093/cid/ciz799
14. Saag MS, Gandhi RT, Hoy JF, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2020 recommendations of the International Antiviral Society-USA Panel. JAMA. 2020;324:1651-1669. doi: 10.1001/jama.2020.17025
15. Havens PL, Perumean-Chaney SE, Patki A, et al. Changes in bone mass after discontinuation of preexposure prophylaxis with tenofovir disoproxil fumarate/emtricitabine in young men who have sex with men: extension phase results of Adolescent Trials Network Protocols 110 and 113. Clin Infect Dis. 2020;70:687-691. doi: 10.1093/cid/ciz486
16. Neilan AM, Salvant Valentine S, Knopf AS. Case 27-2021: a 16-year-old boy seeking human immunodeficiency virus prophylaxis. N Engl J Med. 2021;385:1034-1041. doi: 10.1056/NEJMcpc1909626
17. Thomson KA, Hughes J, Baeten JM, et al. Increased risk of HIV acquisition among women throughout pregnancy and during the postpartum period: a prospective per-coital-act analysis among women with HIV-infected partners. J Infect Dis. 2018;218:16-25. doi: 10.1093/infdis/jiy113
18. Dettinger JC, Kinuthia J, Pintye J, et al. Perinatal outcomes following maternal pre-exposure prophylaxis (PrEP) use during pregnancy: results from a large PrEP implementation program in Kenya. J Int AIDS Soc. 2019;22:e25378. doi: 10.1002/jia2.25378
19. Moodley D, Lombard C, Govender V, et al. Pregnancy and neonatal safety outcomes of timing of initiation of daily oral tenofovir disoproxil fumarate and emtricitabine pre-exposure prophylaxis for HIV prevention (CAP016): an open-label, randomised, non-inferiority trial. Lancet HIV. 2023;10:e154-e163. doi: 10.1016/S2352-3018(22)00369-1
20. CDC. HIV Infection, Risk, Prevention, and Testing Behaviors Among Transgender Women—National HIV Behavioral Surveillance, 7 U.S. Cities, 2019–2020. HIV Surveillance Special Report 27. April 2021. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-special-report-number-27.pdf
21. Wilson EC, Turner CM, Arayasirikul S, et al. Disparities in the PrEP continuum for trans women compared to MSM in San Francisco, California: results from population-based cross-sectional behavioural surveillance studies. J Int AIDS Soc. 2020;23:e25539. doi: 10.1002/jia2.25539
22. Poteat T, Wirtz A, Malik M, et al. A gap between willingness and uptake: findings from mixed methods research on HIV prevention among Black and Latina transgender women. J Acquir Immune Defic Syndr. 2019;82:131-140. doi: 10.1097/QAI.0000000000002112
23. Cottrell ML, Prince HM, Schauer AP, et al. Decreased tenofovir diphosphate concentrations in a transgender female cohort: implications for human immunodeficiency virus preexposure prophylaxis. Clin Infect Dis. 2019;69:2201-2204. doi: 10.1093/cid/ciz290
24. Hess KL, Hu X, Lansky A, et al. Lifetime risk of a diagnosis of HIV infection in the United States. Ann Epidemiol. 2017;27:238-243. doi: 10.1016/j.annepidem.2017.02.003
25. Kanny D, Jeffries WL 4th, Chapin-Bardales J, et al. Racial/ethnic disparities in HIV preexposure prophylaxis among men who have sex with men—23 urban areas, 2017. MMWR Morb Mortal Wkly Rep. 2019;68:801-806. doi: 10.15585/mmwr.mm6837a2
26. CDC. Core indicators for monitoring the Ending the HIV Epidemic initiative (early release): National HIV Surveillance System data reported through December 2020; and preexposure prophylaxis (PrEP) data reported through September 2020. HIV Surveill Data Tables. 2021;2. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/library/reports/surveillance-data-tables/vol-2-no-2/cdc-hiv-surveillance-tables-vol-2-no-2.pdf
27. CDC. Diagnoses of HIV infection in the United States and dependent areas 2021: special focus profiles. Updated May 23, 2023. Accessed October 23, 2023. www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-34/content/special-focus-profiles.html
28. Walters SM, Frank D, Van Ham B, et al. PrEP care continuum engagement among persons who inject drugs: rural and urban differences in stigma and social infrastructure. AIDS Behav. 2021;26:1308-1320. doi: 10.1007/s10461-021-03488-2
29. Foutz J, Artiga S, Garfield R. The role of Medicaid in rural America [issue brief]. April 25, 2017. Accessed August 16, 2023. www.kff.org/medicaid/issue-brief/the-role-of-medicaid-in-rural-america/
30. Baugher AR, Finlayson T, Lewis R, et al. Health care coverage and preexposure prophylaxis (PrEP) use among men who have sex with men living in 22 US cities with vs without Medicaid expansion, 2017. Am J Public Health. 2021;111:743-751. doi: 10.2105/AJPH.2020.306035
1. CDC. Estimated HIV incidence and prevalence in the United States 2017–2021. HIV Surveill Supplemental Rep. 2023;28. Accessed October 23, 2023. https://www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-28-no-3/index.html
2. CDC. Core indicators for monitoring the Ending the HIV Epidemic initiative (preliminary data): National HIV Surveillance System data reported through March 2023; and preexposure prophylaxis (PrEP) data reported through December 2022. HIV Surveill Data Tables. 2023;4. Published June 2023. Accessed October 23, 2023. https://www.cdc.gov/hiv/library/reports/surveillance-data-tables/
3. CDC. Division of HIV Prevention, National Center for HIV, Viral Hepatitis, STD, and TB Prevention. PrEP effectiveness. Updated June 2022. Accessed October 23, 2023. https://www.cdc.gov/hiv/basics/prep/prep-effectiveness.html
4. US Preventive Services Task Force. Prevention of acquisition of HIV: preexposure prophylaxis. Final recommendation statement. August 22, 2023. Accessed October 23, 2023. https://uspreventiveservicestaskforce.org/uspstf/recommendation/prevention-of-human-immunodeficiency-virus-hiv-infection-pre-exposure-prophylaxis
5. CDC. Preexposure prophylaxis for the prevention of HIV infection in the United States—2021 update: a clinical practice guideline. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2021.pdf
6. Mugwanya KK, Wyatt C, Celum C, et al. Changes in glomerular kidney function among HIV-1-uninfected men and women receiving emtricitabine-tenofovir disoproxil fumarate preexposure prophylaxis: a randomized clinical trial. JAMA Intern Med. 2015;175:246-254. doi: 10.1001/jamainternmed.2014.6786
7. Havens PL, Stephensen CB, Van Loan MD, et al. Decline in bone mass with tenofovir disoproxil fumarate/emtricitabine is associated with hormonal changes in the absence of renal impairment when used by HIV-uninfected adolescent boys and young men for HIV preexposure prophylaxis. Clin Infect Dis. 2017;64:317-325. doi: 10.1093/cid/ciw765
8. Pilkington V, Hill A, Hughes S, et al. How safe is TDF/FTC as PrEP? A systematic review and meta-analysis of the risk of adverse events in 13 randomised trials of PrEP. J Virus Erad. 2018;4:215-224.
9. Mayer KH, Molina JM, Thompson MA, et al. Emtricitabine and tenofovir alafenamide vs emtricitabine and tenofovir disoproxil fumarate for HIV pre-exposure prophylaxis (DISCOVER): primary results from a randomised, double-blind, multicentre, active-controlled, phase 3, non-inferiority trial. Lancet. 2020;396:239-254. doi: 10.1016/S0140-6736(20)31065-5
10. Liegeon G, Ghosn, J. Long-acting injectable cabotegravir for PrEP: a game-changer in HIV prevention. HIV Med. 2022;24:653-663. doi: 10.1111/hiv.13451
11. Molina JM, Capitant C, Spire B, et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N Engl J Med. 2015;373:2237-2246. doi: 10.1056/NEJMoa1506273
12. Molina JM, Ghosn J, Assoumou L, et al. Daily and on-demand HIV pre-exposure prophylaxis with emtricitabine and tenofovir disoproxil (ANRS PREVENIR): a prospective observational cohort study. Lancet HIV. 2022;9:e554-e562. doi: 10.1016/S2352-3018(22)00133-3
13. Dimitrov D, Moore JR, Wood D, et al. Predicted effectiveness of daily and nondaily preexposure prophylaxis for men who have sex with men based on sex and pill-taking patterns from the Human Immuno Virus Prevention Trials Network 067/ADAPT Study. Clin Infect Dis. 2020;71:249-255. doi: 10.1093/cid/ciz799
14. Saag MS, Gandhi RT, Hoy JF, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2020 recommendations of the International Antiviral Society-USA Panel. JAMA. 2020;324:1651-1669. doi: 10.1001/jama.2020.17025
15. Havens PL, Perumean-Chaney SE, Patki A, et al. Changes in bone mass after discontinuation of preexposure prophylaxis with tenofovir disoproxil fumarate/emtricitabine in young men who have sex with men: extension phase results of Adolescent Trials Network Protocols 110 and 113. Clin Infect Dis. 2020;70:687-691. doi: 10.1093/cid/ciz486
16. Neilan AM, Salvant Valentine S, Knopf AS. Case 27-2021: a 16-year-old boy seeking human immunodeficiency virus prophylaxis. N Engl J Med. 2021;385:1034-1041. doi: 10.1056/NEJMcpc1909626
17. Thomson KA, Hughes J, Baeten JM, et al. Increased risk of HIV acquisition among women throughout pregnancy and during the postpartum period: a prospective per-coital-act analysis among women with HIV-infected partners. J Infect Dis. 2018;218:16-25. doi: 10.1093/infdis/jiy113
18. Dettinger JC, Kinuthia J, Pintye J, et al. Perinatal outcomes following maternal pre-exposure prophylaxis (PrEP) use during pregnancy: results from a large PrEP implementation program in Kenya. J Int AIDS Soc. 2019;22:e25378. doi: 10.1002/jia2.25378
19. Moodley D, Lombard C, Govender V, et al. Pregnancy and neonatal safety outcomes of timing of initiation of daily oral tenofovir disoproxil fumarate and emtricitabine pre-exposure prophylaxis for HIV prevention (CAP016): an open-label, randomised, non-inferiority trial. Lancet HIV. 2023;10:e154-e163. doi: 10.1016/S2352-3018(22)00369-1
20. CDC. HIV Infection, Risk, Prevention, and Testing Behaviors Among Transgender Women—National HIV Behavioral Surveillance, 7 U.S. Cities, 2019–2020. HIV Surveillance Special Report 27. April 2021. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/library/reports/surveillance/cdc-hiv-surveillance-special-report-number-27.pdf
21. Wilson EC, Turner CM, Arayasirikul S, et al. Disparities in the PrEP continuum for trans women compared to MSM in San Francisco, California: results from population-based cross-sectional behavioural surveillance studies. J Int AIDS Soc. 2020;23:e25539. doi: 10.1002/jia2.25539
22. Poteat T, Wirtz A, Malik M, et al. A gap between willingness and uptake: findings from mixed methods research on HIV prevention among Black and Latina transgender women. J Acquir Immune Defic Syndr. 2019;82:131-140. doi: 10.1097/QAI.0000000000002112
23. Cottrell ML, Prince HM, Schauer AP, et al. Decreased tenofovir diphosphate concentrations in a transgender female cohort: implications for human immunodeficiency virus preexposure prophylaxis. Clin Infect Dis. 2019;69:2201-2204. doi: 10.1093/cid/ciz290
24. Hess KL, Hu X, Lansky A, et al. Lifetime risk of a diagnosis of HIV infection in the United States. Ann Epidemiol. 2017;27:238-243. doi: 10.1016/j.annepidem.2017.02.003
25. Kanny D, Jeffries WL 4th, Chapin-Bardales J, et al. Racial/ethnic disparities in HIV preexposure prophylaxis among men who have sex with men—23 urban areas, 2017. MMWR Morb Mortal Wkly Rep. 2019;68:801-806. doi: 10.15585/mmwr.mm6837a2
26. CDC. Core indicators for monitoring the Ending the HIV Epidemic initiative (early release): National HIV Surveillance System data reported through December 2020; and preexposure prophylaxis (PrEP) data reported through September 2020. HIV Surveill Data Tables. 2021;2. Accessed October 23, 2023. www.cdc.gov/hiv/pdf/library/reports/surveillance-data-tables/vol-2-no-2/cdc-hiv-surveillance-tables-vol-2-no-2.pdf
27. CDC. Diagnoses of HIV infection in the United States and dependent areas 2021: special focus profiles. Updated May 23, 2023. Accessed October 23, 2023. www.cdc.gov/hiv/library/reports/hiv-surveillance/vol-34/content/special-focus-profiles.html
28. Walters SM, Frank D, Van Ham B, et al. PrEP care continuum engagement among persons who inject drugs: rural and urban differences in stigma and social infrastructure. AIDS Behav. 2021;26:1308-1320. doi: 10.1007/s10461-021-03488-2
29. Foutz J, Artiga S, Garfield R. The role of Medicaid in rural America [issue brief]. April 25, 2017. Accessed August 16, 2023. www.kff.org/medicaid/issue-brief/the-role-of-medicaid-in-rural-america/
30. Baugher AR, Finlayson T, Lewis R, et al. Health care coverage and preexposure prophylaxis (PrEP) use among men who have sex with men living in 22 US cities with vs without Medicaid expansion, 2017. Am J Public Health. 2021;111:743-751. doi: 10.2105/AJPH.2020.306035
PRACTICE RECOMMENDATIONS
› Perform routine screening of patients for preexposure prophylaxis (PrEP) eligibility. B
› Prescribe oral or intramuscular PrEP for eligible patients after screening for HIV, other sexually transmitted infections, and hepatitis B, and establishing baseline renal function. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
The future of medicine is RNA
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
Every once in a while, medicine changes in a fundamental way, and we may not realize it while it’s happening. I wasn’t around in 1928 when Fleming discovered penicillin; or in 1953 when Watson, Crick, and Franklin characterized the double-helical structure of DNA.
But looking at medicine today, there are essentially two places where I think we will see, in retrospect, that we were at a fundamental turning point. One is artificial intelligence, which gets so much attention and hype that I will simply say yes, this will change things, stay tuned.
The other is a bit more obscure, but I suspect it may be just as impactful. That other thing is
I want to start with the idea that many diseases are, fundamentally, a problem of proteins. In some cases, like hypercholesterolemia, the body produces too much protein; in others, like hemophilia, too little.
When you think about disease this way, you realize that our current medications take effect late in the disease game. We have these molecules that try to block a protein from its receptor, prevent a protein from cleaving another protein, or increase the rate that a protein is broken down. It’s all distal to the fundamental problem: the production of the bad protein in the first place.
Enter small inhibitory RNAs, or siRNAs for short, discovered in 1998 by Andrew Fire and Craig Mello at UMass Worcester. The two won the Nobel prize in medicine just 8 years later; that’s a really short time, highlighting just how important this discovery was. In contrast, Karikó and Weissman won the Nobel for mRNA vaccines this year, after inventing them 18 years ago.
siRNAs are the body’s way of targeting proteins for destruction before they are ever created. About 20 base pairs long, siRNAs seek out a complementary target mRNA, attach to it, and call in a group of proteins to destroy it. With the target mRNA gone, no protein can be created.
You see where this is going, right? How does high cholesterol kill you? Proteins. How does Staphylococcus aureus kill you? Proteins. Even viruses can’t replicate if their RNA is prevented from being turned into proteins.
So, how do we use siRNAs? A new paper appearing in JAMA describes a fairly impressive use case.
The background here is that higher levels of lipoprotein(a), an LDL-like protein, are associated with cardiovascular disease, heart attack, and stroke. But unfortunately, statins really don’t have any effect on lipoprotein(a) levels. Neither does diet. Your lipoprotein(a) level seems to be more or less hard-coded genetically.
So, what if we stop the genetic machinery from working? Enter lepodisiran, a drug from Eli Lilly. Unlike so many other medications, which are usually found in nature, purified, and synthesized, lepodisiran was created from scratch. It’s not hard. Thanks to the Human Genome Project, we know the genetic code for lipoprotein(a), so inventing an siRNA to target it specifically is trivial. That’s one of the key features of siRNA – you don’t have to find a chemical that binds strongly to some protein receptor, and worry about the off-target effects and all that nonsense. You just pick a protein you want to suppress and you suppress it.
Okay, it’s not that simple. siRNA is broken down very quickly by the body, so it needs to be targeted to the organ of interest – in this case, the liver, since that is where lipoprotein(a) is synthesized. Lepodisiran is targeted to the liver by this special targeting label here.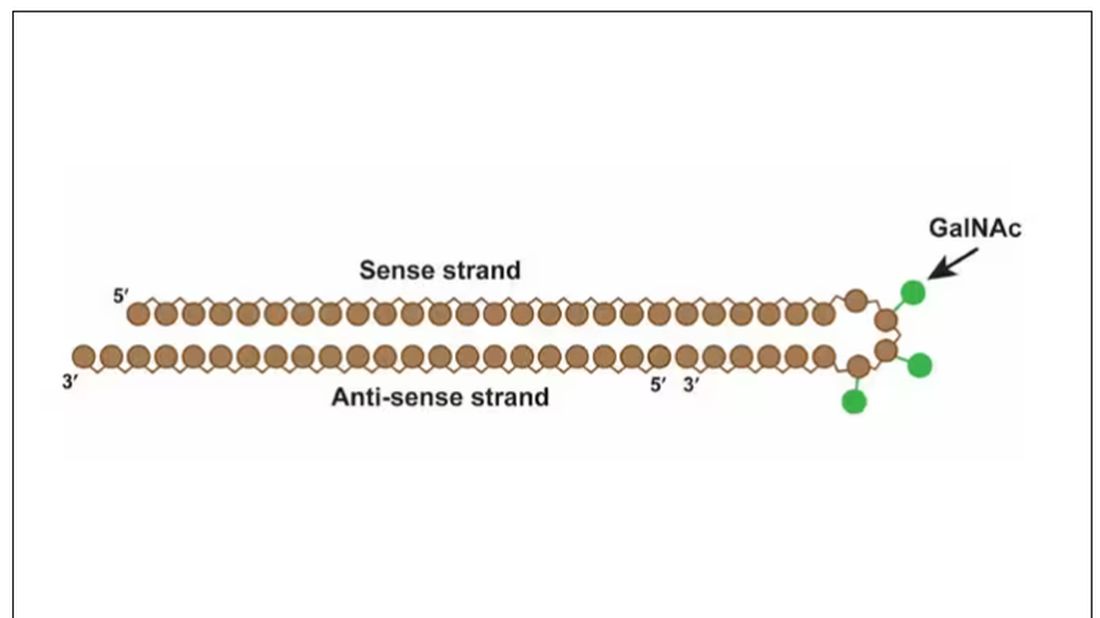
The report is a standard dose-escalation trial. Six patients, all with elevated lipoprotein(a) levels, were started with a 4-mg dose (two additional individuals got placebo). They were intensely monitored, spending 3 days in a research unit for multiple blood draws followed by weekly, and then biweekly outpatient visits. Once they had done well, the next group of six people received a higher dose (two more got placebo), and the process was repeated – six times total – until the highest dose, 608 mg, was reached.
This is an injection, of course; siRNA wouldn’t withstand the harshness of the digestive system. And it’s only one injection. You can see from the blood concentration curves that within about 48 hours, circulating lepodisiran was not detectable.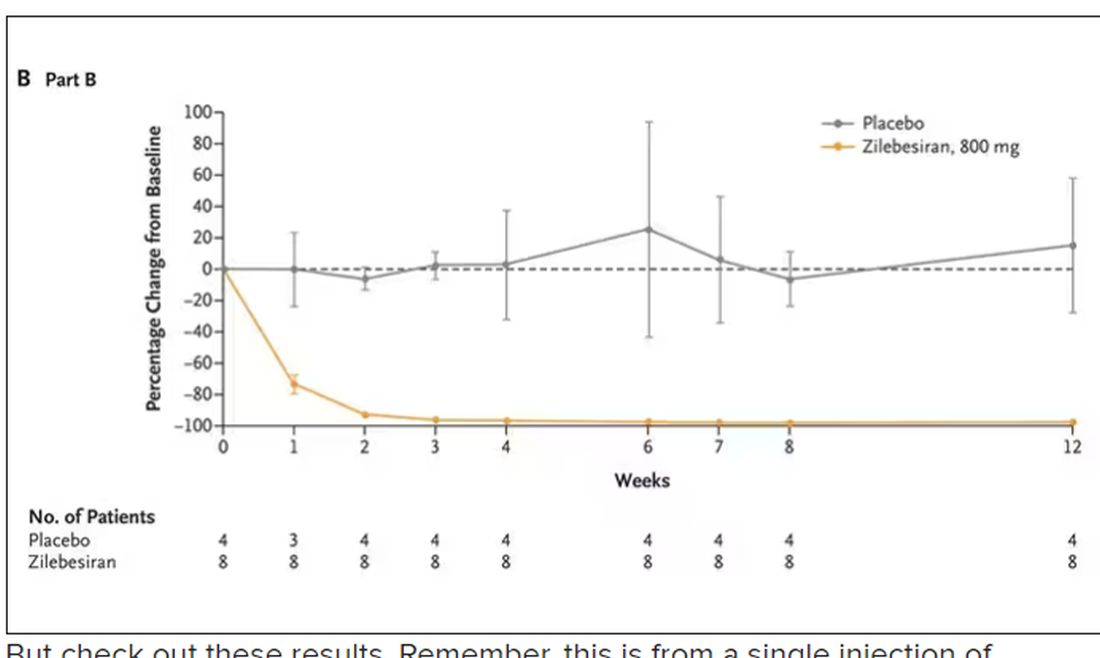
But check out these results. Remember, this is from a single injection of lepodisiran.
Lipoprotein(a) levels start to drop within a week of administration, and they stay down. In the higher-dose groups, levels are nearly undetectable a year after that injection.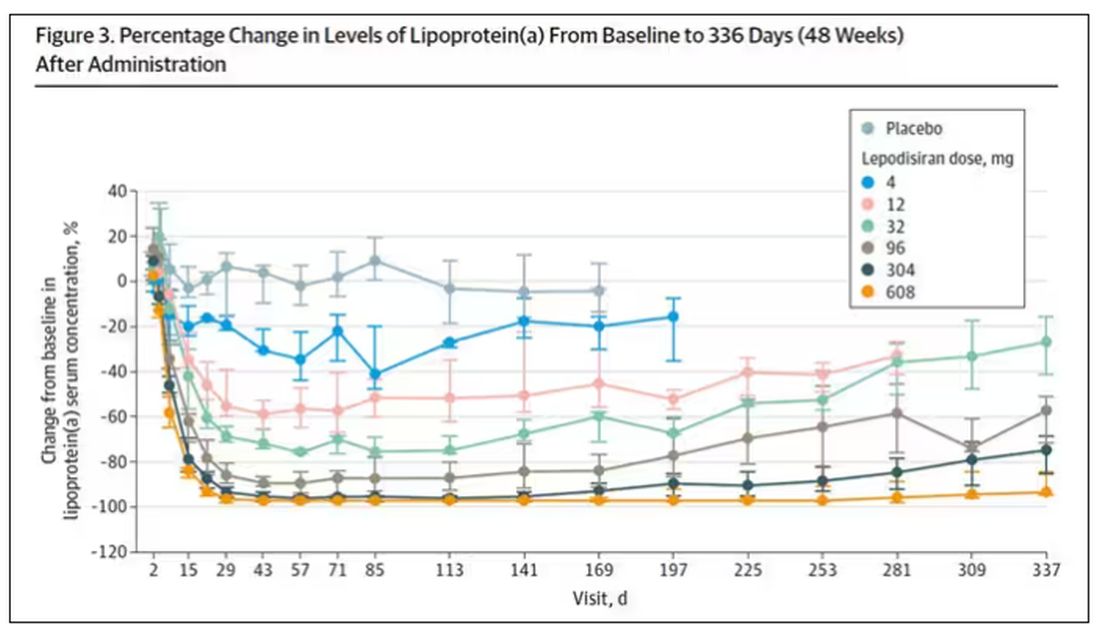
It was this graph that made me sit back and think that there might be something new under the sun. A single injection that can suppress protein synthesis for an entire year? If it really works, it changes the game.
Of course, this study wasn’t powered to look at important outcomes like heart attacks and strokes. It was primarily designed to assess safety, and the drug was pretty well tolerated, with similar rates of adverse events in the drug and placebo groups.
As crazy as it sounds, the real concern here might be that this drug is too good; is it safe to drop your lipoprotein(a) levels to zero for a year? I don’t know. But lower doses don’t have quite as strong an effect.
Trust me, these drugs are going to change things. They already are. In July, The New England Journal of Medicine published a study of zilebesiran, an siRNA that inhibits the production of angiotensinogen, to control blood pressure. Similar story: One injection led to a basically complete suppression of angiotensinogen and a sustained decrease in blood pressure.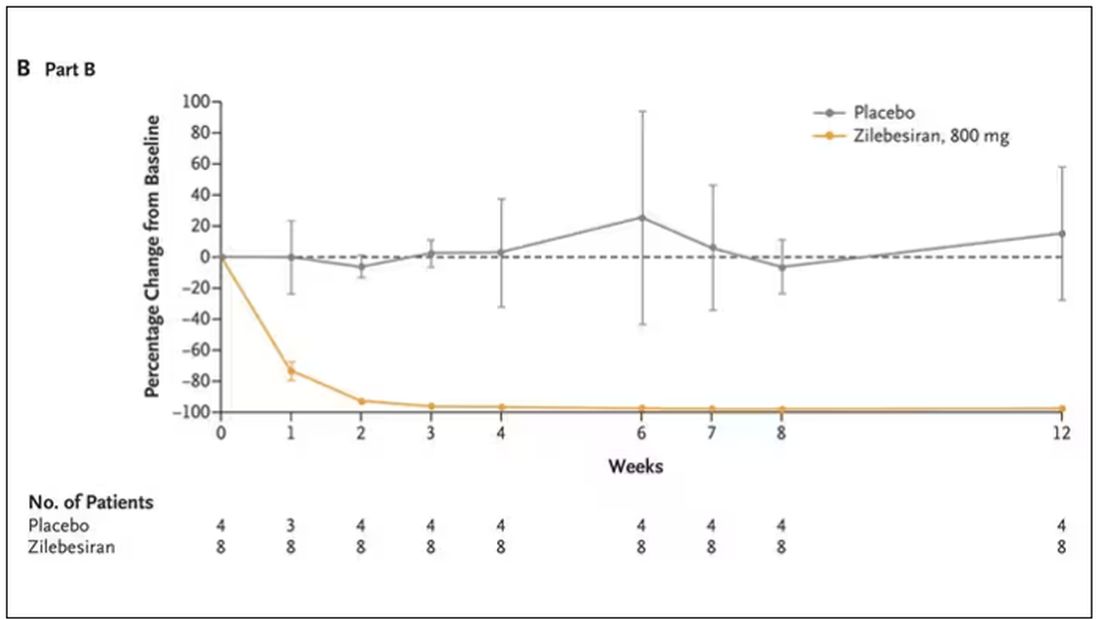
I’m not exaggerating when I say that there may come a time when you go to your doctor once a year, get your RNA shots, and don’t have to take any other medication from that point on. And that time may be, like, 5 years from now. It’s wild.
Seems to me that that rapid Nobel Prize was very well deserved.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships. This transcript has been edited for clarity.
A version of this article appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
Every once in a while, medicine changes in a fundamental way, and we may not realize it while it’s happening. I wasn’t around in 1928 when Fleming discovered penicillin; or in 1953 when Watson, Crick, and Franklin characterized the double-helical structure of DNA.
But looking at medicine today, there are essentially two places where I think we will see, in retrospect, that we were at a fundamental turning point. One is artificial intelligence, which gets so much attention and hype that I will simply say yes, this will change things, stay tuned.
The other is a bit more obscure, but I suspect it may be just as impactful. That other thing is
I want to start with the idea that many diseases are, fundamentally, a problem of proteins. In some cases, like hypercholesterolemia, the body produces too much protein; in others, like hemophilia, too little.
When you think about disease this way, you realize that our current medications take effect late in the disease game. We have these molecules that try to block a protein from its receptor, prevent a protein from cleaving another protein, or increase the rate that a protein is broken down. It’s all distal to the fundamental problem: the production of the bad protein in the first place.
Enter small inhibitory RNAs, or siRNAs for short, discovered in 1998 by Andrew Fire and Craig Mello at UMass Worcester. The two won the Nobel prize in medicine just 8 years later; that’s a really short time, highlighting just how important this discovery was. In contrast, Karikó and Weissman won the Nobel for mRNA vaccines this year, after inventing them 18 years ago.
siRNAs are the body’s way of targeting proteins for destruction before they are ever created. About 20 base pairs long, siRNAs seek out a complementary target mRNA, attach to it, and call in a group of proteins to destroy it. With the target mRNA gone, no protein can be created.
You see where this is going, right? How does high cholesterol kill you? Proteins. How does Staphylococcus aureus kill you? Proteins. Even viruses can’t replicate if their RNA is prevented from being turned into proteins.
So, how do we use siRNAs? A new paper appearing in JAMA describes a fairly impressive use case.
The background here is that higher levels of lipoprotein(a), an LDL-like protein, are associated with cardiovascular disease, heart attack, and stroke. But unfortunately, statins really don’t have any effect on lipoprotein(a) levels. Neither does diet. Your lipoprotein(a) level seems to be more or less hard-coded genetically.
So, what if we stop the genetic machinery from working? Enter lepodisiran, a drug from Eli Lilly. Unlike so many other medications, which are usually found in nature, purified, and synthesized, lepodisiran was created from scratch. It’s not hard. Thanks to the Human Genome Project, we know the genetic code for lipoprotein(a), so inventing an siRNA to target it specifically is trivial. That’s one of the key features of siRNA – you don’t have to find a chemical that binds strongly to some protein receptor, and worry about the off-target effects and all that nonsense. You just pick a protein you want to suppress and you suppress it.
Okay, it’s not that simple. siRNA is broken down very quickly by the body, so it needs to be targeted to the organ of interest – in this case, the liver, since that is where lipoprotein(a) is synthesized. Lepodisiran is targeted to the liver by this special targeting label here.
The report is a standard dose-escalation trial. Six patients, all with elevated lipoprotein(a) levels, were started with a 4-mg dose (two additional individuals got placebo). They were intensely monitored, spending 3 days in a research unit for multiple blood draws followed by weekly, and then biweekly outpatient visits. Once they had done well, the next group of six people received a higher dose (two more got placebo), and the process was repeated – six times total – until the highest dose, 608 mg, was reached.
This is an injection, of course; siRNA wouldn’t withstand the harshness of the digestive system. And it’s only one injection. You can see from the blood concentration curves that within about 48 hours, circulating lepodisiran was not detectable.
But check out these results. Remember, this is from a single injection of lepodisiran.
Lipoprotein(a) levels start to drop within a week of administration, and they stay down. In the higher-dose groups, levels are nearly undetectable a year after that injection.
It was this graph that made me sit back and think that there might be something new under the sun. A single injection that can suppress protein synthesis for an entire year? If it really works, it changes the game.
Of course, this study wasn’t powered to look at important outcomes like heart attacks and strokes. It was primarily designed to assess safety, and the drug was pretty well tolerated, with similar rates of adverse events in the drug and placebo groups.
As crazy as it sounds, the real concern here might be that this drug is too good; is it safe to drop your lipoprotein(a) levels to zero for a year? I don’t know. But lower doses don’t have quite as strong an effect.
Trust me, these drugs are going to change things. They already are. In July, The New England Journal of Medicine published a study of zilebesiran, an siRNA that inhibits the production of angiotensinogen, to control blood pressure. Similar story: One injection led to a basically complete suppression of angiotensinogen and a sustained decrease in blood pressure.
I’m not exaggerating when I say that there may come a time when you go to your doctor once a year, get your RNA shots, and don’t have to take any other medication from that point on. And that time may be, like, 5 years from now. It’s wild.
Seems to me that that rapid Nobel Prize was very well deserved.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships. This transcript has been edited for clarity.
A version of this article appeared on Medscape.com.
Welcome to Impact Factor, your weekly dose of commentary on a new medical study. I’m Dr F. Perry Wilson of the Yale School of Medicine.
Every once in a while, medicine changes in a fundamental way, and we may not realize it while it’s happening. I wasn’t around in 1928 when Fleming discovered penicillin; or in 1953 when Watson, Crick, and Franklin characterized the double-helical structure of DNA.
But looking at medicine today, there are essentially two places where I think we will see, in retrospect, that we were at a fundamental turning point. One is artificial intelligence, which gets so much attention and hype that I will simply say yes, this will change things, stay tuned.
The other is a bit more obscure, but I suspect it may be just as impactful. That other thing is
I want to start with the idea that many diseases are, fundamentally, a problem of proteins. In some cases, like hypercholesterolemia, the body produces too much protein; in others, like hemophilia, too little.
When you think about disease this way, you realize that our current medications take effect late in the disease game. We have these molecules that try to block a protein from its receptor, prevent a protein from cleaving another protein, or increase the rate that a protein is broken down. It’s all distal to the fundamental problem: the production of the bad protein in the first place.
Enter small inhibitory RNAs, or siRNAs for short, discovered in 1998 by Andrew Fire and Craig Mello at UMass Worcester. The two won the Nobel prize in medicine just 8 years later; that’s a really short time, highlighting just how important this discovery was. In contrast, Karikó and Weissman won the Nobel for mRNA vaccines this year, after inventing them 18 years ago.
siRNAs are the body’s way of targeting proteins for destruction before they are ever created. About 20 base pairs long, siRNAs seek out a complementary target mRNA, attach to it, and call in a group of proteins to destroy it. With the target mRNA gone, no protein can be created.
You see where this is going, right? How does high cholesterol kill you? Proteins. How does Staphylococcus aureus kill you? Proteins. Even viruses can’t replicate if their RNA is prevented from being turned into proteins.
So, how do we use siRNAs? A new paper appearing in JAMA describes a fairly impressive use case.
The background here is that higher levels of lipoprotein(a), an LDL-like protein, are associated with cardiovascular disease, heart attack, and stroke. But unfortunately, statins really don’t have any effect on lipoprotein(a) levels. Neither does diet. Your lipoprotein(a) level seems to be more or less hard-coded genetically.
So, what if we stop the genetic machinery from working? Enter lepodisiran, a drug from Eli Lilly. Unlike so many other medications, which are usually found in nature, purified, and synthesized, lepodisiran was created from scratch. It’s not hard. Thanks to the Human Genome Project, we know the genetic code for lipoprotein(a), so inventing an siRNA to target it specifically is trivial. That’s one of the key features of siRNA – you don’t have to find a chemical that binds strongly to some protein receptor, and worry about the off-target effects and all that nonsense. You just pick a protein you want to suppress and you suppress it.
Okay, it’s not that simple. siRNA is broken down very quickly by the body, so it needs to be targeted to the organ of interest – in this case, the liver, since that is where lipoprotein(a) is synthesized. Lepodisiran is targeted to the liver by this special targeting label here.
The report is a standard dose-escalation trial. Six patients, all with elevated lipoprotein(a) levels, were started with a 4-mg dose (two additional individuals got placebo). They were intensely monitored, spending 3 days in a research unit for multiple blood draws followed by weekly, and then biweekly outpatient visits. Once they had done well, the next group of six people received a higher dose (two more got placebo), and the process was repeated – six times total – until the highest dose, 608 mg, was reached.
This is an injection, of course; siRNA wouldn’t withstand the harshness of the digestive system. And it’s only one injection. You can see from the blood concentration curves that within about 48 hours, circulating lepodisiran was not detectable.
But check out these results. Remember, this is from a single injection of lepodisiran.
Lipoprotein(a) levels start to drop within a week of administration, and they stay down. In the higher-dose groups, levels are nearly undetectable a year after that injection.
It was this graph that made me sit back and think that there might be something new under the sun. A single injection that can suppress protein synthesis for an entire year? If it really works, it changes the game.
Of course, this study wasn’t powered to look at important outcomes like heart attacks and strokes. It was primarily designed to assess safety, and the drug was pretty well tolerated, with similar rates of adverse events in the drug and placebo groups.
As crazy as it sounds, the real concern here might be that this drug is too good; is it safe to drop your lipoprotein(a) levels to zero for a year? I don’t know. But lower doses don’t have quite as strong an effect.
Trust me, these drugs are going to change things. They already are. In July, The New England Journal of Medicine published a study of zilebesiran, an siRNA that inhibits the production of angiotensinogen, to control blood pressure. Similar story: One injection led to a basically complete suppression of angiotensinogen and a sustained decrease in blood pressure.
I’m not exaggerating when I say that there may come a time when you go to your doctor once a year, get your RNA shots, and don’t have to take any other medication from that point on. And that time may be, like, 5 years from now. It’s wild.
Seems to me that that rapid Nobel Prize was very well deserved.
Dr. F. Perry Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships. This transcript has been edited for clarity.
A version of this article appeared on Medscape.com.
Painless nodules on legs
A 34-YEAR-OLD MAN presented with a 6-month history of asymptomatic, progressively enlarging subcutaneous nodules over his bilateral lower legs. He denied any history of injury, and there was no bleeding or discharge. The patient had a history of Graves disease that had been treated with radioiodine therapy 2 years prior, followed by thyroxine replacement (150 mcg/d, 5 d/wk and 125 mcg/d, 2 d/wk). At the time of presentation, his thyroid function tests indicated subclinical hypothyroidism: free T4, 21.2 pmol/L (normal range, 11.8-24.6 pmol/L) and thyroid-stimulating hormone (TSH), 14.07 mIU/L (normal range, 0.27-4.2 mIU/L).
Examination revealed nontender, soft brown nodules over the bilateral shins, with minimal overlying lichenification (FIGURE 1). There was no peau d’orange (orange peel) appearance to suggest significant edema. A punch biopsy was performed.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pretibial myxedema
The patient’s history, paired with the results of the punch biopsy, were consistent with a diagnosis of pretibial myxedema, part of the triad of Graves disease along with thyroid ophthalmopathy and acropachy (soft-tissue swelling of the hands and clubbing of the fingers). Histopathologic findings revealed wide separation of collagen bundles throughout the entire reticular dermis without fibroplasia (FIGURE 2A). The spaces contained basophilic strands (FIGURE 2B), and the strands stained strongly positive on Alcian blue (FIGURE 2C), confirming the presence of dermal mucin. Widely separated collagen fibers and deposited mucin are indicative of pretibial myxedema. No granulomas or lymphoid proliferations were seen.
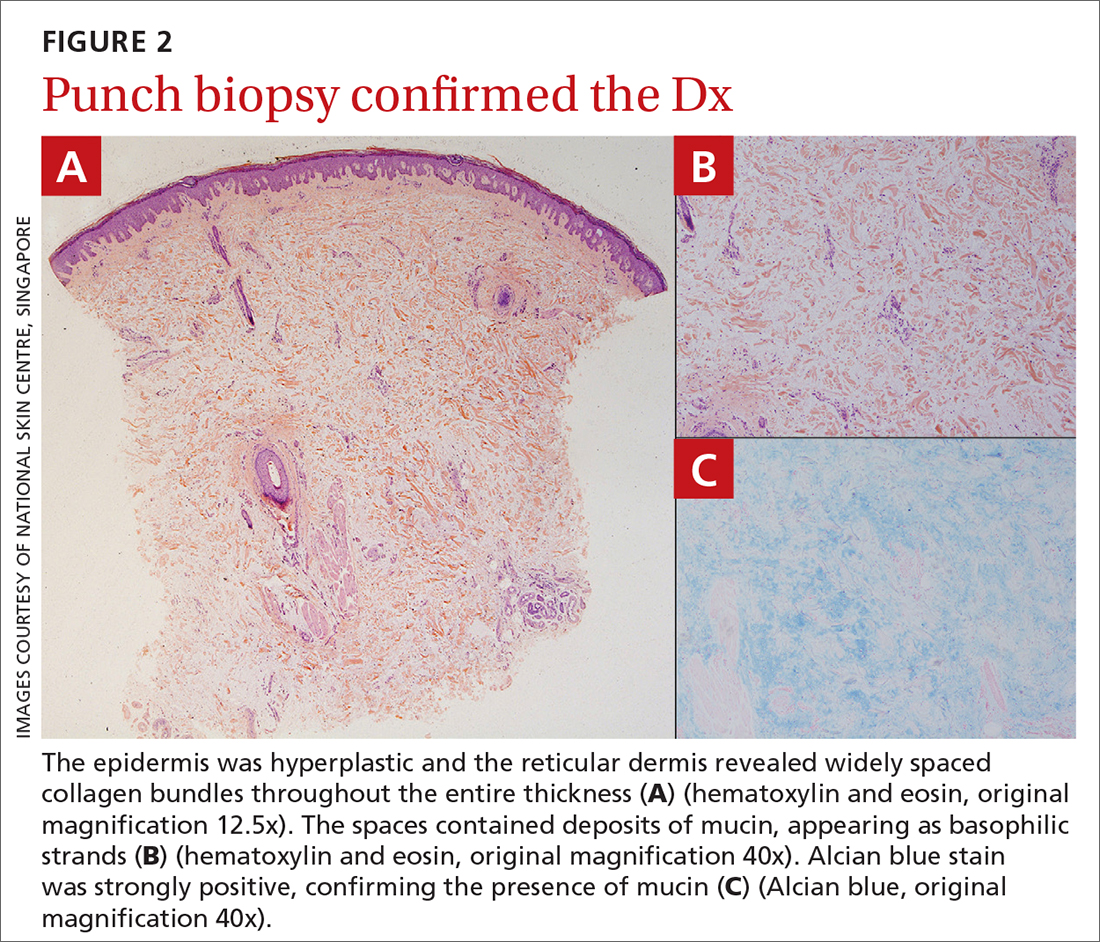
The pathogenesis of pretibial myxedema is widely postulated to be due to the stimulation of dermal fibroblasts by anti–TSH antibodies, causing overproduction of glycosaminoglycans and hyaluronic acid1 and obstructing lymphatic microcirculation, resulting in nonpitting edema.2
There are 5 distinct clinical variants of pretibial myxedema1,3:
- The diffuse form is the most common. It manifests on the lower leg with hard, nonpitting edema and cutaneous thickening.
- The plaque form manifests on the lower leg as well-demarcated erythematous or pigmented flat-topped lesions.
- The nodular form, which our patient had, typically manifests on the lower leg as well-demarcated erythematous, pigmented, or skin-colored raised, solid lesions. There may be 1 lesion or several.
- The mixed form manifests as 2 or more of the other variants.
- The elephantiasic form is the rarest and the most severe. There are widespread swollen nodules and plaques on the lower legs and/or arms.
A rare, late manifestation
Although pathognomonic for Graves disease, pretibial myxedema is a late manifestation that occurs in less than 5% of these patients.4 The most common site of involvement is the pretibial region, although less common sites include the face, arms, shoulders, abdomen, pinna, and the location of previous scars.4
While pretibial myxedema usually is associated with hyperthyroidism, it can occur after treatment (as was the case here), while the patient is in a euthyroid or hypothyroid state. Radioiodine therapy has been reported to be a trigger for pretibial myxedema in 1 case report, although the pathophysiology is not fully understood.5
Continue to: More serious conditions must be ruled out
More serious conditions must be ruled out
The differential for painless nodules includes cutaneous lymphoma and atypical infections of fungal or mycobacterial etiology.
Cutaneous lymphoma that manifests with leg tumors includes primary cutaneous anaplastic CD30+ large cell lymphoma (PCALCL) and primary cutaneous diffuse large B-cell lymphoma, leg type (PCDLBL-LT). The former may occur in young patients, whereas the latter tends to manifest in the elderly. Biopsy shows a neoplastic proliferation of atypical lymphocytes within the dermis,6 differing from our case.
Atypical infections may be detected through bacterial, mycobacterial, or fungal cultures, and may be accompanied by elevated inflammatory markers or other systemic symptoms of the infection, setting it apart from pretibial myxedema.
Treatment is simple and noninvasive
Pretibial myxedema is usually asymptomatic, with minimal morbidity. The nodular variant may resolve spontaneously; thus, therapeutic management often is reserved for severe cases or for those with cosmetic concerns. Treatment options include mid- to high-potency topical corticosteroids with an occlusive dressing for 1 to 2 weeks (or until resolution) or an intralesional triamcinolone injection (5-10 mg/mL, single or monthly until resolution), compression stockings, and pneumatic compression.2
This patient was treated with a single intralesional injection of triamcinolone 10 mg/mL. The nodules resolved within a month.
1. Thammarucha S, Sudtikoonaseth P. Nodular pretibial myxedema with Graves’ disease: a case report. Thai J Dermatol. 2021;37:30-36.
2. Singla M, Gupta A. Nodular thyroid dermopathy: not a hallmark of Graves’ disease. Am J Med. 2019;132:e521-e522. doi: 10.1016/j.amjmed.2018.11.004
3. Lan C, Wang Y, Zeng X, et al. Morphological diversity of pretibial myxedema and its mechanism of evolving process and outcome: a retrospective study of 216 cases. J Thyroid Res. 2016:2016:265217
4. doi: 10.1155/2016/2652174 4. Patil MM, Kamalanathan S, Sahoo J, et al. Pretibial myxedema. QJM. 2015;108:985. doi: 10.1093/qjmed/hcv136
5. Harvey RD, Metcalfe RA, Morteo C, et al. Acute pre-tibial myxoedema following radioiodine therapy for thyrotoxic Graves’ disease. Clin Endocrinol (Oxf). 1995;42:657-660. doi: 10.1111/j.1365-2265.1995.tb02695.x
6. Schukow C, Ahmed A. Dermatopathology, cutaneous lymphomas. StatPearls [Internet]. Updated February 16, 2023. Accessed October 23, 2023. www.ncbi.nlm.nih.gov/books/NBK589703/
A 34-YEAR-OLD MAN presented with a 6-month history of asymptomatic, progressively enlarging subcutaneous nodules over his bilateral lower legs. He denied any history of injury, and there was no bleeding or discharge. The patient had a history of Graves disease that had been treated with radioiodine therapy 2 years prior, followed by thyroxine replacement (150 mcg/d, 5 d/wk and 125 mcg/d, 2 d/wk). At the time of presentation, his thyroid function tests indicated subclinical hypothyroidism: free T4, 21.2 pmol/L (normal range, 11.8-24.6 pmol/L) and thyroid-stimulating hormone (TSH), 14.07 mIU/L (normal range, 0.27-4.2 mIU/L).
Examination revealed nontender, soft brown nodules over the bilateral shins, with minimal overlying lichenification (FIGURE 1). There was no peau d’orange (orange peel) appearance to suggest significant edema. A punch biopsy was performed.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pretibial myxedema
The patient’s history, paired with the results of the punch biopsy, were consistent with a diagnosis of pretibial myxedema, part of the triad of Graves disease along with thyroid ophthalmopathy and acropachy (soft-tissue swelling of the hands and clubbing of the fingers). Histopathologic findings revealed wide separation of collagen bundles throughout the entire reticular dermis without fibroplasia (FIGURE 2A). The spaces contained basophilic strands (FIGURE 2B), and the strands stained strongly positive on Alcian blue (FIGURE 2C), confirming the presence of dermal mucin. Widely separated collagen fibers and deposited mucin are indicative of pretibial myxedema. No granulomas or lymphoid proliferations were seen.

The pathogenesis of pretibial myxedema is widely postulated to be due to the stimulation of dermal fibroblasts by anti–TSH antibodies, causing overproduction of glycosaminoglycans and hyaluronic acid1 and obstructing lymphatic microcirculation, resulting in nonpitting edema.2
There are 5 distinct clinical variants of pretibial myxedema1,3:
- The diffuse form is the most common. It manifests on the lower leg with hard, nonpitting edema and cutaneous thickening.
- The plaque form manifests on the lower leg as well-demarcated erythematous or pigmented flat-topped lesions.
- The nodular form, which our patient had, typically manifests on the lower leg as well-demarcated erythematous, pigmented, or skin-colored raised, solid lesions. There may be 1 lesion or several.
- The mixed form manifests as 2 or more of the other variants.
- The elephantiasic form is the rarest and the most severe. There are widespread swollen nodules and plaques on the lower legs and/or arms.
A rare, late manifestation
Although pathognomonic for Graves disease, pretibial myxedema is a late manifestation that occurs in less than 5% of these patients.4 The most common site of involvement is the pretibial region, although less common sites include the face, arms, shoulders, abdomen, pinna, and the location of previous scars.4
While pretibial myxedema usually is associated with hyperthyroidism, it can occur after treatment (as was the case here), while the patient is in a euthyroid or hypothyroid state. Radioiodine therapy has been reported to be a trigger for pretibial myxedema in 1 case report, although the pathophysiology is not fully understood.5
Continue to: More serious conditions must be ruled out
More serious conditions must be ruled out
The differential for painless nodules includes cutaneous lymphoma and atypical infections of fungal or mycobacterial etiology.
Cutaneous lymphoma that manifests with leg tumors includes primary cutaneous anaplastic CD30+ large cell lymphoma (PCALCL) and primary cutaneous diffuse large B-cell lymphoma, leg type (PCDLBL-LT). The former may occur in young patients, whereas the latter tends to manifest in the elderly. Biopsy shows a neoplastic proliferation of atypical lymphocytes within the dermis,6 differing from our case.
Atypical infections may be detected through bacterial, mycobacterial, or fungal cultures, and may be accompanied by elevated inflammatory markers or other systemic symptoms of the infection, setting it apart from pretibial myxedema.
Treatment is simple and noninvasive
Pretibial myxedema is usually asymptomatic, with minimal morbidity. The nodular variant may resolve spontaneously; thus, therapeutic management often is reserved for severe cases or for those with cosmetic concerns. Treatment options include mid- to high-potency topical corticosteroids with an occlusive dressing for 1 to 2 weeks (or until resolution) or an intralesional triamcinolone injection (5-10 mg/mL, single or monthly until resolution), compression stockings, and pneumatic compression.2
This patient was treated with a single intralesional injection of triamcinolone 10 mg/mL. The nodules resolved within a month.
A 34-YEAR-OLD MAN presented with a 6-month history of asymptomatic, progressively enlarging subcutaneous nodules over his bilateral lower legs. He denied any history of injury, and there was no bleeding or discharge. The patient had a history of Graves disease that had been treated with radioiodine therapy 2 years prior, followed by thyroxine replacement (150 mcg/d, 5 d/wk and 125 mcg/d, 2 d/wk). At the time of presentation, his thyroid function tests indicated subclinical hypothyroidism: free T4, 21.2 pmol/L (normal range, 11.8-24.6 pmol/L) and thyroid-stimulating hormone (TSH), 14.07 mIU/L (normal range, 0.27-4.2 mIU/L).
Examination revealed nontender, soft brown nodules over the bilateral shins, with minimal overlying lichenification (FIGURE 1). There was no peau d’orange (orange peel) appearance to suggest significant edema. A punch biopsy was performed.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pretibial myxedema
The patient’s history, paired with the results of the punch biopsy, were consistent with a diagnosis of pretibial myxedema, part of the triad of Graves disease along with thyroid ophthalmopathy and acropachy (soft-tissue swelling of the hands and clubbing of the fingers). Histopathologic findings revealed wide separation of collagen bundles throughout the entire reticular dermis without fibroplasia (FIGURE 2A). The spaces contained basophilic strands (FIGURE 2B), and the strands stained strongly positive on Alcian blue (FIGURE 2C), confirming the presence of dermal mucin. Widely separated collagen fibers and deposited mucin are indicative of pretibial myxedema. No granulomas or lymphoid proliferations were seen.

The pathogenesis of pretibial myxedema is widely postulated to be due to the stimulation of dermal fibroblasts by anti–TSH antibodies, causing overproduction of glycosaminoglycans and hyaluronic acid1 and obstructing lymphatic microcirculation, resulting in nonpitting edema.2
There are 5 distinct clinical variants of pretibial myxedema1,3:
- The diffuse form is the most common. It manifests on the lower leg with hard, nonpitting edema and cutaneous thickening.
- The plaque form manifests on the lower leg as well-demarcated erythematous or pigmented flat-topped lesions.
- The nodular form, which our patient had, typically manifests on the lower leg as well-demarcated erythematous, pigmented, or skin-colored raised, solid lesions. There may be 1 lesion or several.
- The mixed form manifests as 2 or more of the other variants.
- The elephantiasic form is the rarest and the most severe. There are widespread swollen nodules and plaques on the lower legs and/or arms.
A rare, late manifestation
Although pathognomonic for Graves disease, pretibial myxedema is a late manifestation that occurs in less than 5% of these patients.4 The most common site of involvement is the pretibial region, although less common sites include the face, arms, shoulders, abdomen, pinna, and the location of previous scars.4
While pretibial myxedema usually is associated with hyperthyroidism, it can occur after treatment (as was the case here), while the patient is in a euthyroid or hypothyroid state. Radioiodine therapy has been reported to be a trigger for pretibial myxedema in 1 case report, although the pathophysiology is not fully understood.5
Continue to: More serious conditions must be ruled out
More serious conditions must be ruled out
The differential for painless nodules includes cutaneous lymphoma and atypical infections of fungal or mycobacterial etiology.
Cutaneous lymphoma that manifests with leg tumors includes primary cutaneous anaplastic CD30+ large cell lymphoma (PCALCL) and primary cutaneous diffuse large B-cell lymphoma, leg type (PCDLBL-LT). The former may occur in young patients, whereas the latter tends to manifest in the elderly. Biopsy shows a neoplastic proliferation of atypical lymphocytes within the dermis,6 differing from our case.
Atypical infections may be detected through bacterial, mycobacterial, or fungal cultures, and may be accompanied by elevated inflammatory markers or other systemic symptoms of the infection, setting it apart from pretibial myxedema.
Treatment is simple and noninvasive
Pretibial myxedema is usually asymptomatic, with minimal morbidity. The nodular variant may resolve spontaneously; thus, therapeutic management often is reserved for severe cases or for those with cosmetic concerns. Treatment options include mid- to high-potency topical corticosteroids with an occlusive dressing for 1 to 2 weeks (or until resolution) or an intralesional triamcinolone injection (5-10 mg/mL, single or monthly until resolution), compression stockings, and pneumatic compression.2
This patient was treated with a single intralesional injection of triamcinolone 10 mg/mL. The nodules resolved within a month.
1. Thammarucha S, Sudtikoonaseth P. Nodular pretibial myxedema with Graves’ disease: a case report. Thai J Dermatol. 2021;37:30-36.
2. Singla M, Gupta A. Nodular thyroid dermopathy: not a hallmark of Graves’ disease. Am J Med. 2019;132:e521-e522. doi: 10.1016/j.amjmed.2018.11.004
3. Lan C, Wang Y, Zeng X, et al. Morphological diversity of pretibial myxedema and its mechanism of evolving process and outcome: a retrospective study of 216 cases. J Thyroid Res. 2016:2016:265217
4. doi: 10.1155/2016/2652174 4. Patil MM, Kamalanathan S, Sahoo J, et al. Pretibial myxedema. QJM. 2015;108:985. doi: 10.1093/qjmed/hcv136
5. Harvey RD, Metcalfe RA, Morteo C, et al. Acute pre-tibial myxoedema following radioiodine therapy for thyrotoxic Graves’ disease. Clin Endocrinol (Oxf). 1995;42:657-660. doi: 10.1111/j.1365-2265.1995.tb02695.x
6. Schukow C, Ahmed A. Dermatopathology, cutaneous lymphomas. StatPearls [Internet]. Updated February 16, 2023. Accessed October 23, 2023. www.ncbi.nlm.nih.gov/books/NBK589703/
1. Thammarucha S, Sudtikoonaseth P. Nodular pretibial myxedema with Graves’ disease: a case report. Thai J Dermatol. 2021;37:30-36.
2. Singla M, Gupta A. Nodular thyroid dermopathy: not a hallmark of Graves’ disease. Am J Med. 2019;132:e521-e522. doi: 10.1016/j.amjmed.2018.11.004
3. Lan C, Wang Y, Zeng X, et al. Morphological diversity of pretibial myxedema and its mechanism of evolving process and outcome: a retrospective study of 216 cases. J Thyroid Res. 2016:2016:265217
4. doi: 10.1155/2016/2652174 4. Patil MM, Kamalanathan S, Sahoo J, et al. Pretibial myxedema. QJM. 2015;108:985. doi: 10.1093/qjmed/hcv136
5. Harvey RD, Metcalfe RA, Morteo C, et al. Acute pre-tibial myxoedema following radioiodine therapy for thyrotoxic Graves’ disease. Clin Endocrinol (Oxf). 1995;42:657-660. doi: 10.1111/j.1365-2265.1995.tb02695.x
6. Schukow C, Ahmed A. Dermatopathology, cutaneous lymphomas. StatPearls [Internet]. Updated February 16, 2023. Accessed October 23, 2023. www.ncbi.nlm.nih.gov/books/NBK589703/
Alpha-gal syndrome: Red meat is ‘just the beginning,’ expert says
ANAHEIM, CALIF. – allergist and immunologist Scott P. Commins, MD, PhD, told attendees at the annual meeting of the American College of Allergy, Asthma, and Immunology (ACAAI) annual meeting.
Dr. Commins, associate chief for allergy and immunology at the University of North Carolina at Chapel Hill, has made alpha-gal, a potentially fatal allergy, which, in the United States is tied to the bite of the Lone Star tick, his primary research focus.
Beyond red meat, “there are some people who are allergic to all things mammal,” he explained. Dairy products from mammals, medical devices made from mammalian products, vaccines and medicines that contain gelatin, and even commercial products such as perfumes and cosmetics may be behind an AGS reaction.
“The derived products from pigs and cows really find their way into a lot of our day-to-day products,” he said. “I try to keep an open mind about these exposures.”
Physicians should also be aware that “this can happen to kids,” said Dr. Commins. “It looks very similar to adults’ [AGS]. They can end up in the emergency department.”
He also had clinical advice about food challenges for AGS. He explained that there’s more alpha-gal in beef than in other red meats (including pork, venison, and lamb) with the exception of pork kidney. Pork kidney, he said, “has the most alpha-gal that we can find in the lab.”
Dr. Commins said he has stopped using beef for AGS food challenges and has switched to pork sausage patties with a high fat content microwaved in the clinic because they have less alpha-gal in general and he views them as safer.
Long delay in symptom onset
AGS symptoms typically take 2-6 hours to appear after eating red meat or being exposed to mammalian products, but Dr. Commins related a story about a patient he sent home who had very mild symptoms (some lower back itching) after he had spent the day at the clinic after a pork sausage food challenge for AGS.
The patient had returned home. Eight hours after the food challenge, his wife sent Dr. Commins a picture of her husband’s back, which was riddled with welts and was itching badly.
“I learned that if you’re going to do these food challenges, if there is a hint of symptoms at the clinic at 6 hours, keep them in the clinic, because it may really take that long to evolve,” Dr. Commins said.
One of the early signs he’s discovered is palmar erythema (redness and swelling of the hands).
Research has shown that AGS has been heavily concentrated in the Southeast, where Lone Star tick populations are clustered, but research has shown that from 2017 to 2022, it moved up the East Coast to the central United States and Upper Midwest.
“We are seeing increasing diagnoses of AGS in places that are not, perhaps, where we first thought this allergy existed,” said Dr. Commins. “Stay aware,” he cautioned.
The allergy is not exclusive to the United States, he noted. In Europe and Australia, for example, AGS is not thought to be tied to the Lone Star tick, which doesn’t inhabit those regions.
“It is a global phenomenon,” Dr. Commins said.
In August, the CDC alerted physicians to emerging cases of alpha-gal allergy after an article in Morbidity and Mortality Weekly Report indicated that health care providers have little knowledge about the allergy. Of the 1,500 health care providers surveyed, 42% had never heard of the syndrome, and another 35% were not confident in diagnosing or managing affected patients.
Matthew Lau, MD, an allergist with Kaiser Permanente in Honolulu who listened to Dr. Commins’ talk, told this news organization, “It’s important to raise awareness in primary care particularly, he said, as “allergists see only a fraction of the [AGS] patients.”
Allergists can help raise awareness
“Allergists have a role to alert the general community” and to drive more referrals, he said. That includes emergency departments, where physicians commonly see anaphylaxis.
Dr. Lau said he expects the incidence of AGS to increase, because global warming will likely lengthen warmer seasons and cause the geographic distribution to change.
Jay Lieberman, MD, a pediatric allergist at Le Bonheur Children’s Hospital in Memphis, Tenn., told this news organization, “There’s still a lot of confusion, and hearing from an expert like Dr. Commins helps tease out the not-obvious things about patients who are having more mild symptoms,” such as from allergy to dairy or medicines or vaccines that contain gelatin.
As a pediatric allergist, Dr. Lieberman said he sees less alpha-gal than his colleagues, but, he said, “On the adult side in Tennessee, it’s rampant.”
Dr. Commins, Dr. Lieberman, and Dr. Lau report no relevant financial relationships.
A version of this article appeared on Medscape.com.
ANAHEIM, CALIF. – allergist and immunologist Scott P. Commins, MD, PhD, told attendees at the annual meeting of the American College of Allergy, Asthma, and Immunology (ACAAI) annual meeting.
Dr. Commins, associate chief for allergy and immunology at the University of North Carolina at Chapel Hill, has made alpha-gal, a potentially fatal allergy, which, in the United States is tied to the bite of the Lone Star tick, his primary research focus.
Beyond red meat, “there are some people who are allergic to all things mammal,” he explained. Dairy products from mammals, medical devices made from mammalian products, vaccines and medicines that contain gelatin, and even commercial products such as perfumes and cosmetics may be behind an AGS reaction.
“The derived products from pigs and cows really find their way into a lot of our day-to-day products,” he said. “I try to keep an open mind about these exposures.”
Physicians should also be aware that “this can happen to kids,” said Dr. Commins. “It looks very similar to adults’ [AGS]. They can end up in the emergency department.”
He also had clinical advice about food challenges for AGS. He explained that there’s more alpha-gal in beef than in other red meats (including pork, venison, and lamb) with the exception of pork kidney. Pork kidney, he said, “has the most alpha-gal that we can find in the lab.”
Dr. Commins said he has stopped using beef for AGS food challenges and has switched to pork sausage patties with a high fat content microwaved in the clinic because they have less alpha-gal in general and he views them as safer.
Long delay in symptom onset
AGS symptoms typically take 2-6 hours to appear after eating red meat or being exposed to mammalian products, but Dr. Commins related a story about a patient he sent home who had very mild symptoms (some lower back itching) after he had spent the day at the clinic after a pork sausage food challenge for AGS.
The patient had returned home. Eight hours after the food challenge, his wife sent Dr. Commins a picture of her husband’s back, which was riddled with welts and was itching badly.
“I learned that if you’re going to do these food challenges, if there is a hint of symptoms at the clinic at 6 hours, keep them in the clinic, because it may really take that long to evolve,” Dr. Commins said.
One of the early signs he’s discovered is palmar erythema (redness and swelling of the hands).
Research has shown that AGS has been heavily concentrated in the Southeast, where Lone Star tick populations are clustered, but research has shown that from 2017 to 2022, it moved up the East Coast to the central United States and Upper Midwest.
“We are seeing increasing diagnoses of AGS in places that are not, perhaps, where we first thought this allergy existed,” said Dr. Commins. “Stay aware,” he cautioned.
The allergy is not exclusive to the United States, he noted. In Europe and Australia, for example, AGS is not thought to be tied to the Lone Star tick, which doesn’t inhabit those regions.
“It is a global phenomenon,” Dr. Commins said.
In August, the CDC alerted physicians to emerging cases of alpha-gal allergy after an article in Morbidity and Mortality Weekly Report indicated that health care providers have little knowledge about the allergy. Of the 1,500 health care providers surveyed, 42% had never heard of the syndrome, and another 35% were not confident in diagnosing or managing affected patients.
Matthew Lau, MD, an allergist with Kaiser Permanente in Honolulu who listened to Dr. Commins’ talk, told this news organization, “It’s important to raise awareness in primary care particularly, he said, as “allergists see only a fraction of the [AGS] patients.”
Allergists can help raise awareness
“Allergists have a role to alert the general community” and to drive more referrals, he said. That includes emergency departments, where physicians commonly see anaphylaxis.
Dr. Lau said he expects the incidence of AGS to increase, because global warming will likely lengthen warmer seasons and cause the geographic distribution to change.
Jay Lieberman, MD, a pediatric allergist at Le Bonheur Children’s Hospital in Memphis, Tenn., told this news organization, “There’s still a lot of confusion, and hearing from an expert like Dr. Commins helps tease out the not-obvious things about patients who are having more mild symptoms,” such as from allergy to dairy or medicines or vaccines that contain gelatin.
As a pediatric allergist, Dr. Lieberman said he sees less alpha-gal than his colleagues, but, he said, “On the adult side in Tennessee, it’s rampant.”
Dr. Commins, Dr. Lieberman, and Dr. Lau report no relevant financial relationships.
A version of this article appeared on Medscape.com.
ANAHEIM, CALIF. – allergist and immunologist Scott P. Commins, MD, PhD, told attendees at the annual meeting of the American College of Allergy, Asthma, and Immunology (ACAAI) annual meeting.
Dr. Commins, associate chief for allergy and immunology at the University of North Carolina at Chapel Hill, has made alpha-gal, a potentially fatal allergy, which, in the United States is tied to the bite of the Lone Star tick, his primary research focus.
Beyond red meat, “there are some people who are allergic to all things mammal,” he explained. Dairy products from mammals, medical devices made from mammalian products, vaccines and medicines that contain gelatin, and even commercial products such as perfumes and cosmetics may be behind an AGS reaction.
“The derived products from pigs and cows really find their way into a lot of our day-to-day products,” he said. “I try to keep an open mind about these exposures.”
Physicians should also be aware that “this can happen to kids,” said Dr. Commins. “It looks very similar to adults’ [AGS]. They can end up in the emergency department.”
He also had clinical advice about food challenges for AGS. He explained that there’s more alpha-gal in beef than in other red meats (including pork, venison, and lamb) with the exception of pork kidney. Pork kidney, he said, “has the most alpha-gal that we can find in the lab.”
Dr. Commins said he has stopped using beef for AGS food challenges and has switched to pork sausage patties with a high fat content microwaved in the clinic because they have less alpha-gal in general and he views them as safer.
Long delay in symptom onset
AGS symptoms typically take 2-6 hours to appear after eating red meat or being exposed to mammalian products, but Dr. Commins related a story about a patient he sent home who had very mild symptoms (some lower back itching) after he had spent the day at the clinic after a pork sausage food challenge for AGS.
The patient had returned home. Eight hours after the food challenge, his wife sent Dr. Commins a picture of her husband’s back, which was riddled with welts and was itching badly.
“I learned that if you’re going to do these food challenges, if there is a hint of symptoms at the clinic at 6 hours, keep them in the clinic, because it may really take that long to evolve,” Dr. Commins said.
One of the early signs he’s discovered is palmar erythema (redness and swelling of the hands).
Research has shown that AGS has been heavily concentrated in the Southeast, where Lone Star tick populations are clustered, but research has shown that from 2017 to 2022, it moved up the East Coast to the central United States and Upper Midwest.
“We are seeing increasing diagnoses of AGS in places that are not, perhaps, where we first thought this allergy existed,” said Dr. Commins. “Stay aware,” he cautioned.
The allergy is not exclusive to the United States, he noted. In Europe and Australia, for example, AGS is not thought to be tied to the Lone Star tick, which doesn’t inhabit those regions.
“It is a global phenomenon,” Dr. Commins said.
In August, the CDC alerted physicians to emerging cases of alpha-gal allergy after an article in Morbidity and Mortality Weekly Report indicated that health care providers have little knowledge about the allergy. Of the 1,500 health care providers surveyed, 42% had never heard of the syndrome, and another 35% were not confident in diagnosing or managing affected patients.
Matthew Lau, MD, an allergist with Kaiser Permanente in Honolulu who listened to Dr. Commins’ talk, told this news organization, “It’s important to raise awareness in primary care particularly, he said, as “allergists see only a fraction of the [AGS] patients.”
Allergists can help raise awareness
“Allergists have a role to alert the general community” and to drive more referrals, he said. That includes emergency departments, where physicians commonly see anaphylaxis.
Dr. Lau said he expects the incidence of AGS to increase, because global warming will likely lengthen warmer seasons and cause the geographic distribution to change.
Jay Lieberman, MD, a pediatric allergist at Le Bonheur Children’s Hospital in Memphis, Tenn., told this news organization, “There’s still a lot of confusion, and hearing from an expert like Dr. Commins helps tease out the not-obvious things about patients who are having more mild symptoms,” such as from allergy to dairy or medicines or vaccines that contain gelatin.
As a pediatric allergist, Dr. Lieberman said he sees less alpha-gal than his colleagues, but, he said, “On the adult side in Tennessee, it’s rampant.”
Dr. Commins, Dr. Lieberman, and Dr. Lau report no relevant financial relationships.
A version of this article appeared on Medscape.com.
A new standard for treatment of torus fractures of the wrist?
ILLUSTRATIVE CASE
A 9-year-old girl presents to your urgent care clinic after a fall while snowboarding for the first time. She reports falling forward onto her outstretched right hand and describes pain in her distal right forearm. She denies paresthesias, weakness, or lacerations. Physical examination reveals mild edema of the dorsal aspect of her distal right forearm and tenderness to palpation of the dorsal aspect of her distal radius. She denies tenderness to palpation of her ulna, anatomic snuffbox, hand, and elbow. Range of motion of the wrist is full on passive testing, but she declines active testing due to pain. Wrist radiographs reveal an uncomplicated torus fracture of the distal radius. Can immobilization with a soft bandage alone sufficiently treat this fracture?
Fractures of the distal radius are among the most common fractures of the upper extremity and commonly occur from a fall onto an outstretched hand.2 In the pediatric population, torus fractures, also known as buckle fractures, are the most common type of distal radius fracture, comprising an estimated 50% of pediatric wrist fractures.3,4 This is due to the presence of a
Pediatric torus fractures of the distal radius generally are treated with immobilization,2 traditionally through a
Despite common use of immobilization, torus fractures of the distal radius are anatomically stable, and displacement is unlikely to occur.7,8 As such, many studies have suggested that treatment of torus fractures with rigid immobilization in a cast or splint may not be necessary.9,10 However, a 2018 Cochrane review concluded that the quality of evidence illustrating similar recovery between treatments was low, leaving uncertainty as to the most appropriate management strategy.6 Less casting and follow-up imaging could have positive implications for patient satisfaction, health care–associated costs, and radiation exposure.10
This study, the Forearm Fracture Recovery in Children Evaluation (FORCE) trial, compared the traditional treatment of distal radius torus fractures with rigid immobilization to soft immobilization and immediate discharge.
STUDY SUMMARY
Providing quality evidence for a standard of care
FORCE was a randomized controlled equivalence trial (N = 965) across 23 emergency departments (EDs) in the United Kingdom that compared pain and function in pediatric patients with distal radius torus fractures treated with a soft bandage and immediate discharge vs rigid immobilization and routine follow-up.1 Patients included children ages 4 to 15 years presenting to the ED with a distal radius torus fracture, which was confirmed radiologically.
Patients with concomitant
Continue to: Patients were randomly assigned...
Patients were randomly assigned in a 1:1 ratio to receive treatment with either a soft bandage such as a gauze roller bandage (n = 489) or rigid immobilization (n = 476). For patients in the bandage group, a soft bandage was applied in the ED or provided for home application without planned clinical follow-up. Patients in the rigid immobilization group were treated in the ED with either a removable manufactured splint or a molded splint or cast, followed by the standard follow-up practice of the treating center. Patients in the soft bandage group were advised not to wear the bandage for more than 3 weeks. Blinding was not possible, but the treatment team did not take part in patient follow-up.
The primary outcome was change in pain 3 days after treatment, measured on the Wong-Baker FACES Pain Rating Scale (an ordinal assessment using 6 illustrated facial expressions translated to a numeric rating on a scale of 0-10, with higher scores indicating worse pain). This scale has an established minimum clinically important difference (MCID) value of 1 face (2 points).11 Per standard practice in equivalence trials, the equivalence margin was defined as half the MCID, with a value of 1.0 used in this study.
Secondary outcomes measured over the 6-week follow-up period included additional pain measurements using the Wong-Baker scale, measures of function and health-related quality of life, analgesia use, days of absence from school or childcare, complication rates, and patient satisfaction. This study used modified intention-to-treat and per-protocol analyses.
The mean age of participants was 9.6 years; 39% were girls and 61% were boys. In the bandage group, 94% opted to have the soft bandage applied in the ED, and 95% of the rigid immobilization group were treated with a removable wrist splint in the ED. At 3 days, pain scores improved by 3.2 points (standard deviation [SD] = 2.1) in the soft bandage group and 3.1 points (SD = 2.1) in the rigid immobilization group. The adjusted difference was –0.1 (95% CI, –0.37 to 0.17) in the intention-to-treat analysis and –0.06 (95% CI, –0.34 to 0.21) in the per-protocol analysis, which were both less than the predetermined equivalence margin. This equivalence margin also was met at all secondary time points (1 day, 7 days, 3 weeks, and 6 weeks after treatment) and in subgroup analysis of those 4 to 7 years and 8 to 15 years.
Use of any analgesia in the prior 24 hours was slightly higher in the soft bandage group on Day 1 (83% vs 78%; P = .04) and Day 3 (57% vs 51%; P = .05), but this difference was not seen on Day 7. Satisfaction, measured via a 7-point Likert scale (range from “extremely satisfied” to “extremely unsatisfied”), was slightly lower in the soft bandage group on Day 1 (median 2 [interquartile range = 1, 2] vs median 1 [interquartile range = 1, 2]; P < .0001) but was not different after 6 weeks. There were no measured differences in any other secondary outcomes, including function, quality of life, and complication rates.
Continue to: By the primary end point...
By the primary end point of 3 days, 36 patients (7%) in the soft bandage group returned to medical care requesting a change to rigid immobilization, compared with 1 patient (0.2%) in the rigid immobilization group declining intervention.
WHAT’S NEW
Equivalence in pain and function scores
This trial showed equivalence in pain at 3 days’ follow-up in children with distal radius torus fractures who were offered bandaging and then immediately discharged from the ED, compared with rigid immobilization and clinical follow-up. There were no significant differences in pain or function between groups during the 6 weeks following the initial injury. De-escalation of treatment offers an equivalent, resource-sparing alternative to traditional treatment of these fractures.
CAVEATS
Lack of masking likely introduced bias
There are no major caveats associated with managing distal radius torus fractures with a soft bandage and discharge from the ED, compared with the traditional treatment of rigid immobilization. However, bias was likely introduced in patient-reported outcomes due to the inability to mask patients and families to the treatment allocation. This may have led to overstating the severity of outcomes in the bandage group, given the strong preference for rigid immobilization, although equivalence was illustrated despite this potential bias.
CHALLENGES TO IMPLEMENTATION
Preferences may be difficult to change
Parents and clinicians demonstrated a preference for rigid immobilization, as shown in the imbalance in treatment crossovers, with 7% of children changing to the rigid immobilization group by the primary study end point of 3 days. The study authors hypothesized that crossovers may have been due to the perception by some parents that rigid immobilization is the gold standard of treatment, as well as clinicians’ seeking to escalate care for patients returning for follow-up. Policy and guideline changes, as well as physician efforts to educate patients on outcomes with soft bandage treatment, are likely to improve these misconceptions.
1. Perry DC, Achten J, Knight R, et al; FORCE Collaborators in collaboration with PERUKI. Immobilisation of torus fractures of the wrist in children (FORCE): a randomised controlled equivalence trial in the UK. Lancet. 2022;400:39-47. doi: 10.1016/S0140-6736(22)01015-7
2. Patel DS, Statuta SM, Ahmed N. Common fractures of the radius and ulna. Am Fam Physician. 2021;103:345-354.
3. Asokan A, Kheir N. Pediatric Torus Buckle Fracture. StatPearls Publishing; 2023.
4. Naranje SM, Erali RA, Warner WC Jr, et al. Epidemiology of pediatric fractures presenting to emergency departments in the United States. J Pediatr Orthop. 2016;36:e45-e48. doi: 10.1097/BPO.0000000000000595
5. Kennedy SA, Slobogean GP, Mulpuri K. Does degree of immobilization influence refracture rate in the forearm buckle fracture? J Pediatr Orthop B. 2010;19:77-81. doi: 10.1097/BPB.0b013e32832f067a
6. Handoll HHG, Elliott J, Iheozor-Ejiofor Z, et al. Interventions for treating wrist fractures in children. Cochrane Database Syst Rev. 2018;12:CD012470. doi: 10.1002/14651858.CD012470.pub2
7. Perry DC, Gibson P, Roland D, et al. What level of immobilisation is necessary for treatment of torus (buckle) fractures of the distal radius in children? BMJ. 2021;372:m4862. doi: 10.1136/bmj.m4862
8. Williams KG, Smith G, Luhmann SJ, et al. A randomized controlled trial of cast versus splint for distal radial buckle fracture: an evaluation of satisfaction, convenience, and preference. Pediatr Emerg Care. 2013;29:555-559. doi: 10.1097/PEC.0b013e31828e56fb
9. Jiang N, Cao ZH, Ma YF, et al. Management of pediatric forearm torus fractures: a systematic review and meta-analysis. Pediatr Emerg Care. 2016;32:773-778. doi: 10.1097/PEC.0000000000000579
10. Williams BA, Alvarado CA, Montoya-Williams DC, et al. Buckling down on torus fractures: has evolving evidence affected practice? J Child Orthop. 2018;12:123-128. doi: 10.1302/1863-2548.12.170122
11. Garra G, Singer AJ, Taira BR, et al. Validation of the Wong-Baker FACES Pain Rating Scale in pediatric emergency department patients. Acad Emerg Med. 2010;17:50-54. doi: 10.1111/j.1553-2712.2009.00620.x
ILLUSTRATIVE CASE
A 9-year-old girl presents to your urgent care clinic after a fall while snowboarding for the first time. She reports falling forward onto her outstretched right hand and describes pain in her distal right forearm. She denies paresthesias, weakness, or lacerations. Physical examination reveals mild edema of the dorsal aspect of her distal right forearm and tenderness to palpation of the dorsal aspect of her distal radius. She denies tenderness to palpation of her ulna, anatomic snuffbox, hand, and elbow. Range of motion of the wrist is full on passive testing, but she declines active testing due to pain. Wrist radiographs reveal an uncomplicated torus fracture of the distal radius. Can immobilization with a soft bandage alone sufficiently treat this fracture?
Fractures of the distal radius are among the most common fractures of the upper extremity and commonly occur from a fall onto an outstretched hand.2 In the pediatric population, torus fractures, also known as buckle fractures, are the most common type of distal radius fracture, comprising an estimated 50% of pediatric wrist fractures.3,4 This is due to the presence of a
Pediatric torus fractures of the distal radius generally are treated with immobilization,2 traditionally through a
Despite common use of immobilization, torus fractures of the distal radius are anatomically stable, and displacement is unlikely to occur.7,8 As such, many studies have suggested that treatment of torus fractures with rigid immobilization in a cast or splint may not be necessary.9,10 However, a 2018 Cochrane review concluded that the quality of evidence illustrating similar recovery between treatments was low, leaving uncertainty as to the most appropriate management strategy.6 Less casting and follow-up imaging could have positive implications for patient satisfaction, health care–associated costs, and radiation exposure.10
This study, the Forearm Fracture Recovery in Children Evaluation (FORCE) trial, compared the traditional treatment of distal radius torus fractures with rigid immobilization to soft immobilization and immediate discharge.
STUDY SUMMARY
Providing quality evidence for a standard of care
FORCE was a randomized controlled equivalence trial (N = 965) across 23 emergency departments (EDs) in the United Kingdom that compared pain and function in pediatric patients with distal radius torus fractures treated with a soft bandage and immediate discharge vs rigid immobilization and routine follow-up.1 Patients included children ages 4 to 15 years presenting to the ED with a distal radius torus fracture, which was confirmed radiologically.
Patients with concomitant
Continue to: Patients were randomly assigned...
Patients were randomly assigned in a 1:1 ratio to receive treatment with either a soft bandage such as a gauze roller bandage (n = 489) or rigid immobilization (n = 476). For patients in the bandage group, a soft bandage was applied in the ED or provided for home application without planned clinical follow-up. Patients in the rigid immobilization group were treated in the ED with either a removable manufactured splint or a molded splint or cast, followed by the standard follow-up practice of the treating center. Patients in the soft bandage group were advised not to wear the bandage for more than 3 weeks. Blinding was not possible, but the treatment team did not take part in patient follow-up.
The primary outcome was change in pain 3 days after treatment, measured on the Wong-Baker FACES Pain Rating Scale (an ordinal assessment using 6 illustrated facial expressions translated to a numeric rating on a scale of 0-10, with higher scores indicating worse pain). This scale has an established minimum clinically important difference (MCID) value of 1 face (2 points).11 Per standard practice in equivalence trials, the equivalence margin was defined as half the MCID, with a value of 1.0 used in this study.
Secondary outcomes measured over the 6-week follow-up period included additional pain measurements using the Wong-Baker scale, measures of function and health-related quality of life, analgesia use, days of absence from school or childcare, complication rates, and patient satisfaction. This study used modified intention-to-treat and per-protocol analyses.
The mean age of participants was 9.6 years; 39% were girls and 61% were boys. In the bandage group, 94% opted to have the soft bandage applied in the ED, and 95% of the rigid immobilization group were treated with a removable wrist splint in the ED. At 3 days, pain scores improved by 3.2 points (standard deviation [SD] = 2.1) in the soft bandage group and 3.1 points (SD = 2.1) in the rigid immobilization group. The adjusted difference was –0.1 (95% CI, –0.37 to 0.17) in the intention-to-treat analysis and –0.06 (95% CI, –0.34 to 0.21) in the per-protocol analysis, which were both less than the predetermined equivalence margin. This equivalence margin also was met at all secondary time points (1 day, 7 days, 3 weeks, and 6 weeks after treatment) and in subgroup analysis of those 4 to 7 years and 8 to 15 years.
Use of any analgesia in the prior 24 hours was slightly higher in the soft bandage group on Day 1 (83% vs 78%; P = .04) and Day 3 (57% vs 51%; P = .05), but this difference was not seen on Day 7. Satisfaction, measured via a 7-point Likert scale (range from “extremely satisfied” to “extremely unsatisfied”), was slightly lower in the soft bandage group on Day 1 (median 2 [interquartile range = 1, 2] vs median 1 [interquartile range = 1, 2]; P < .0001) but was not different after 6 weeks. There were no measured differences in any other secondary outcomes, including function, quality of life, and complication rates.
Continue to: By the primary end point...
By the primary end point of 3 days, 36 patients (7%) in the soft bandage group returned to medical care requesting a change to rigid immobilization, compared with 1 patient (0.2%) in the rigid immobilization group declining intervention.
WHAT’S NEW
Equivalence in pain and function scores
This trial showed equivalence in pain at 3 days’ follow-up in children with distal radius torus fractures who were offered bandaging and then immediately discharged from the ED, compared with rigid immobilization and clinical follow-up. There were no significant differences in pain or function between groups during the 6 weeks following the initial injury. De-escalation of treatment offers an equivalent, resource-sparing alternative to traditional treatment of these fractures.
CAVEATS
Lack of masking likely introduced bias
There are no major caveats associated with managing distal radius torus fractures with a soft bandage and discharge from the ED, compared with the traditional treatment of rigid immobilization. However, bias was likely introduced in patient-reported outcomes due to the inability to mask patients and families to the treatment allocation. This may have led to overstating the severity of outcomes in the bandage group, given the strong preference for rigid immobilization, although equivalence was illustrated despite this potential bias.
CHALLENGES TO IMPLEMENTATION
Preferences may be difficult to change
Parents and clinicians demonstrated a preference for rigid immobilization, as shown in the imbalance in treatment crossovers, with 7% of children changing to the rigid immobilization group by the primary study end point of 3 days. The study authors hypothesized that crossovers may have been due to the perception by some parents that rigid immobilization is the gold standard of treatment, as well as clinicians’ seeking to escalate care for patients returning for follow-up. Policy and guideline changes, as well as physician efforts to educate patients on outcomes with soft bandage treatment, are likely to improve these misconceptions.
ILLUSTRATIVE CASE
A 9-year-old girl presents to your urgent care clinic after a fall while snowboarding for the first time. She reports falling forward onto her outstretched right hand and describes pain in her distal right forearm. She denies paresthesias, weakness, or lacerations. Physical examination reveals mild edema of the dorsal aspect of her distal right forearm and tenderness to palpation of the dorsal aspect of her distal radius. She denies tenderness to palpation of her ulna, anatomic snuffbox, hand, and elbow. Range of motion of the wrist is full on passive testing, but she declines active testing due to pain. Wrist radiographs reveal an uncomplicated torus fracture of the distal radius. Can immobilization with a soft bandage alone sufficiently treat this fracture?
Fractures of the distal radius are among the most common fractures of the upper extremity and commonly occur from a fall onto an outstretched hand.2 In the pediatric population, torus fractures, also known as buckle fractures, are the most common type of distal radius fracture, comprising an estimated 50% of pediatric wrist fractures.3,4 This is due to the presence of a
Pediatric torus fractures of the distal radius generally are treated with immobilization,2 traditionally through a
Despite common use of immobilization, torus fractures of the distal radius are anatomically stable, and displacement is unlikely to occur.7,8 As such, many studies have suggested that treatment of torus fractures with rigid immobilization in a cast or splint may not be necessary.9,10 However, a 2018 Cochrane review concluded that the quality of evidence illustrating similar recovery between treatments was low, leaving uncertainty as to the most appropriate management strategy.6 Less casting and follow-up imaging could have positive implications for patient satisfaction, health care–associated costs, and radiation exposure.10
This study, the Forearm Fracture Recovery in Children Evaluation (FORCE) trial, compared the traditional treatment of distal radius torus fractures with rigid immobilization to soft immobilization and immediate discharge.
STUDY SUMMARY
Providing quality evidence for a standard of care
FORCE was a randomized controlled equivalence trial (N = 965) across 23 emergency departments (EDs) in the United Kingdom that compared pain and function in pediatric patients with distal radius torus fractures treated with a soft bandage and immediate discharge vs rigid immobilization and routine follow-up.1 Patients included children ages 4 to 15 years presenting to the ED with a distal radius torus fracture, which was confirmed radiologically.
Patients with concomitant
Continue to: Patients were randomly assigned...
Patients were randomly assigned in a 1:1 ratio to receive treatment with either a soft bandage such as a gauze roller bandage (n = 489) or rigid immobilization (n = 476). For patients in the bandage group, a soft bandage was applied in the ED or provided for home application without planned clinical follow-up. Patients in the rigid immobilization group were treated in the ED with either a removable manufactured splint or a molded splint or cast, followed by the standard follow-up practice of the treating center. Patients in the soft bandage group were advised not to wear the bandage for more than 3 weeks. Blinding was not possible, but the treatment team did not take part in patient follow-up.
The primary outcome was change in pain 3 days after treatment, measured on the Wong-Baker FACES Pain Rating Scale (an ordinal assessment using 6 illustrated facial expressions translated to a numeric rating on a scale of 0-10, with higher scores indicating worse pain). This scale has an established minimum clinically important difference (MCID) value of 1 face (2 points).11 Per standard practice in equivalence trials, the equivalence margin was defined as half the MCID, with a value of 1.0 used in this study.
Secondary outcomes measured over the 6-week follow-up period included additional pain measurements using the Wong-Baker scale, measures of function and health-related quality of life, analgesia use, days of absence from school or childcare, complication rates, and patient satisfaction. This study used modified intention-to-treat and per-protocol analyses.
The mean age of participants was 9.6 years; 39% were girls and 61% were boys. In the bandage group, 94% opted to have the soft bandage applied in the ED, and 95% of the rigid immobilization group were treated with a removable wrist splint in the ED. At 3 days, pain scores improved by 3.2 points (standard deviation [SD] = 2.1) in the soft bandage group and 3.1 points (SD = 2.1) in the rigid immobilization group. The adjusted difference was –0.1 (95% CI, –0.37 to 0.17) in the intention-to-treat analysis and –0.06 (95% CI, –0.34 to 0.21) in the per-protocol analysis, which were both less than the predetermined equivalence margin. This equivalence margin also was met at all secondary time points (1 day, 7 days, 3 weeks, and 6 weeks after treatment) and in subgroup analysis of those 4 to 7 years and 8 to 15 years.
Use of any analgesia in the prior 24 hours was slightly higher in the soft bandage group on Day 1 (83% vs 78%; P = .04) and Day 3 (57% vs 51%; P = .05), but this difference was not seen on Day 7. Satisfaction, measured via a 7-point Likert scale (range from “extremely satisfied” to “extremely unsatisfied”), was slightly lower in the soft bandage group on Day 1 (median 2 [interquartile range = 1, 2] vs median 1 [interquartile range = 1, 2]; P < .0001) but was not different after 6 weeks. There were no measured differences in any other secondary outcomes, including function, quality of life, and complication rates.
Continue to: By the primary end point...
By the primary end point of 3 days, 36 patients (7%) in the soft bandage group returned to medical care requesting a change to rigid immobilization, compared with 1 patient (0.2%) in the rigid immobilization group declining intervention.
WHAT’S NEW
Equivalence in pain and function scores
This trial showed equivalence in pain at 3 days’ follow-up in children with distal radius torus fractures who were offered bandaging and then immediately discharged from the ED, compared with rigid immobilization and clinical follow-up. There were no significant differences in pain or function between groups during the 6 weeks following the initial injury. De-escalation of treatment offers an equivalent, resource-sparing alternative to traditional treatment of these fractures.
CAVEATS
Lack of masking likely introduced bias
There are no major caveats associated with managing distal radius torus fractures with a soft bandage and discharge from the ED, compared with the traditional treatment of rigid immobilization. However, bias was likely introduced in patient-reported outcomes due to the inability to mask patients and families to the treatment allocation. This may have led to overstating the severity of outcomes in the bandage group, given the strong preference for rigid immobilization, although equivalence was illustrated despite this potential bias.
CHALLENGES TO IMPLEMENTATION
Preferences may be difficult to change
Parents and clinicians demonstrated a preference for rigid immobilization, as shown in the imbalance in treatment crossovers, with 7% of children changing to the rigid immobilization group by the primary study end point of 3 days. The study authors hypothesized that crossovers may have been due to the perception by some parents that rigid immobilization is the gold standard of treatment, as well as clinicians’ seeking to escalate care for patients returning for follow-up. Policy and guideline changes, as well as physician efforts to educate patients on outcomes with soft bandage treatment, are likely to improve these misconceptions.
1. Perry DC, Achten J, Knight R, et al; FORCE Collaborators in collaboration with PERUKI. Immobilisation of torus fractures of the wrist in children (FORCE): a randomised controlled equivalence trial in the UK. Lancet. 2022;400:39-47. doi: 10.1016/S0140-6736(22)01015-7
2. Patel DS, Statuta SM, Ahmed N. Common fractures of the radius and ulna. Am Fam Physician. 2021;103:345-354.
3. Asokan A, Kheir N. Pediatric Torus Buckle Fracture. StatPearls Publishing; 2023.
4. Naranje SM, Erali RA, Warner WC Jr, et al. Epidemiology of pediatric fractures presenting to emergency departments in the United States. J Pediatr Orthop. 2016;36:e45-e48. doi: 10.1097/BPO.0000000000000595
5. Kennedy SA, Slobogean GP, Mulpuri K. Does degree of immobilization influence refracture rate in the forearm buckle fracture? J Pediatr Orthop B. 2010;19:77-81. doi: 10.1097/BPB.0b013e32832f067a
6. Handoll HHG, Elliott J, Iheozor-Ejiofor Z, et al. Interventions for treating wrist fractures in children. Cochrane Database Syst Rev. 2018;12:CD012470. doi: 10.1002/14651858.CD012470.pub2
7. Perry DC, Gibson P, Roland D, et al. What level of immobilisation is necessary for treatment of torus (buckle) fractures of the distal radius in children? BMJ. 2021;372:m4862. doi: 10.1136/bmj.m4862
8. Williams KG, Smith G, Luhmann SJ, et al. A randomized controlled trial of cast versus splint for distal radial buckle fracture: an evaluation of satisfaction, convenience, and preference. Pediatr Emerg Care. 2013;29:555-559. doi: 10.1097/PEC.0b013e31828e56fb
9. Jiang N, Cao ZH, Ma YF, et al. Management of pediatric forearm torus fractures: a systematic review and meta-analysis. Pediatr Emerg Care. 2016;32:773-778. doi: 10.1097/PEC.0000000000000579
10. Williams BA, Alvarado CA, Montoya-Williams DC, et al. Buckling down on torus fractures: has evolving evidence affected practice? J Child Orthop. 2018;12:123-128. doi: 10.1302/1863-2548.12.170122
11. Garra G, Singer AJ, Taira BR, et al. Validation of the Wong-Baker FACES Pain Rating Scale in pediatric emergency department patients. Acad Emerg Med. 2010;17:50-54. doi: 10.1111/j.1553-2712.2009.00620.x
1. Perry DC, Achten J, Knight R, et al; FORCE Collaborators in collaboration with PERUKI. Immobilisation of torus fractures of the wrist in children (FORCE): a randomised controlled equivalence trial in the UK. Lancet. 2022;400:39-47. doi: 10.1016/S0140-6736(22)01015-7
2. Patel DS, Statuta SM, Ahmed N. Common fractures of the radius and ulna. Am Fam Physician. 2021;103:345-354.
3. Asokan A, Kheir N. Pediatric Torus Buckle Fracture. StatPearls Publishing; 2023.
4. Naranje SM, Erali RA, Warner WC Jr, et al. Epidemiology of pediatric fractures presenting to emergency departments in the United States. J Pediatr Orthop. 2016;36:e45-e48. doi: 10.1097/BPO.0000000000000595
5. Kennedy SA, Slobogean GP, Mulpuri K. Does degree of immobilization influence refracture rate in the forearm buckle fracture? J Pediatr Orthop B. 2010;19:77-81. doi: 10.1097/BPB.0b013e32832f067a
6. Handoll HHG, Elliott J, Iheozor-Ejiofor Z, et al. Interventions for treating wrist fractures in children. Cochrane Database Syst Rev. 2018;12:CD012470. doi: 10.1002/14651858.CD012470.pub2
7. Perry DC, Gibson P, Roland D, et al. What level of immobilisation is necessary for treatment of torus (buckle) fractures of the distal radius in children? BMJ. 2021;372:m4862. doi: 10.1136/bmj.m4862
8. Williams KG, Smith G, Luhmann SJ, et al. A randomized controlled trial of cast versus splint for distal radial buckle fracture: an evaluation of satisfaction, convenience, and preference. Pediatr Emerg Care. 2013;29:555-559. doi: 10.1097/PEC.0b013e31828e56fb
9. Jiang N, Cao ZH, Ma YF, et al. Management of pediatric forearm torus fractures: a systematic review and meta-analysis. Pediatr Emerg Care. 2016;32:773-778. doi: 10.1097/PEC.0000000000000579
10. Williams BA, Alvarado CA, Montoya-Williams DC, et al. Buckling down on torus fractures: has evolving evidence affected practice? J Child Orthop. 2018;12:123-128. doi: 10.1302/1863-2548.12.170122
11. Garra G, Singer AJ, Taira BR, et al. Validation of the Wong-Baker FACES Pain Rating Scale in pediatric emergency department patients. Acad Emerg Med. 2010;17:50-54. doi: 10.1111/j.1553-2712.2009.00620.x
PRACTICE CHANGER
For uncomplicated pediatric torus fractures of the distal radius, consider definitive management with soft bandage immobilization until pain resolution, rather than rigid immobilization and clinical follow-up.
STRENGTH OF RECOMMENDATION
B: Based on a single randomized controlled trial with patient-oriented outcomes.1
Perry DC, Achten J, Knight R, et al; FORCE Collaborators in collaboration with PERUKI. Immobilisation of torus fractures of the wrist in children (FORCE): a randomised controlled equivalence trial in the UK. Lancet. 2022;400:39-47. doi: 10.1016/S0140-6736(22)01015-7
An FP’s guide to caring for patients with seizure and epilepsy
Managing first-time seizures and epilepsy often requires consultation with a neurologist or epileptologist for diagnosis and subsequent management, including when medical treatment fails or in determining whether patients may benefit from surgery. However, given the high prevalence of epilepsy and even higher incidence of a single seizure, family physicians contribute significantly to the management of these patients. The main issues are managing a first-time seizure, making the diagnosis, establishing a treatment plan, and exploring triggers and mitigating factors.
Seizure vs epilepsy
All patients with epilepsy experience seizures, but not every person who experiences a seizure has (or will develop) epilepsy. Nearly 10% of the population has one seizure during their lifetime, whereas the risk for epilepsy is just 3%.1 Therefore, a first-time seizure may not herald epilepsy, defined as repetitive (≥ 2) unprovoked seizures more than 24 hours apart.2 Seizures can be provoked (acute symptomatic) or unprovoked; a clear distinction between these 2 occurrences—as well as between single and recurrent seizures—is critical for proper management. A close look at the circumstances of a first-time seizure is imperative to define the nature of the event and the possibility of further seizures before devising a treatment plan.
Provoked seizures are due to an acute brain insult such as toxic-metabolic disorders, concussion, alcohol withdrawal, an adverse effect of a medication or its withdrawal, or photic stimulation presumably by disrupting the brain’s metabolic homeostasis or integrity. The key factor is that provoked seizures always happen in close temporal association with an acute insult. A single provoked seizure happens each year in 29 to 39 individuals per 100,000.3 While these seizures typically occur singly, there is a small risk they may recur if the triggering insult persists or repeats.1 Therefore, more than 1 seizure per se may not indicate epilepsy.3
Unprovoked seizures reflect an underlying brain dysfunction. A single unprovoked seizure happens in 23 to 61 individuals per 100,000 per year, often in men in either younger or older age groups.3 Unprovoked seizures may occur only once or may recur (ie, evolve into epilepsy). The latter scenario happens in only about half of cases; the overall risk for a recurrent seizure within 2 years of a first seizure is estimated at 42% (24% to 65%, depending on the etiology and electroencephalogram [EEG] findings).4 More specifically, without treatment the relapse rate will be 36% at 1 year and 47% at 2 years.4 Further, a second unprovoked seizure, if untreated, would increase the risk for third and fourth seizures to 73% and 76%, respectively, within 4 years.3
Evaluating the first-time seizure
Ask the patient or observers about the circumstances of the event to differentiate provoked from unprovoked onset. For one thing, not all “spells” are seizures. The differential diagnoses may include syncope, psychogenic nonepileptic events, drug intoxication or withdrawal, migraine, panic attacks, sleep disorders (parasomnia), transient global amnesia, concussion, and transient ischemic attack. EEG, neuroimaging, and other relevant diagnostic tests often are needed (eg, electrocardiogram/echocardiogram/Holter monitoring to evaluate for syncope/cardiac arrhythmia). Clinically, syncopal episodes tend to be brief with rapid recovery and no confusion, speech problems, aura, or lateralizing signs such as hand posturing or lip smacking that are typical with focal seizures. However, cases of convulsive syncope can be challenging to assess without diagnostic tests.
True convulsive seizures do not have the variability in clinical signs seen with psychogenic nonepileptic events (eg, alternating body parts involved or direction of movements). Transient global amnesia is a rare condition with no established diagnostic test and is considered a diagnosis of exclusion, although bitemporal hyperintensities on magnetic resonance imaging (MRI) may appear 12 to 48 hours after the clinical episode.5 Blood work is needed in patients with medical issues treated with multiple medications to evaluate for metabolic derangements; otherwise, routine blood work provides minimal information in stable patients.
Region-specific causes. Neurocysticercosis is common in some regions, such as Latin America; therefore, attention should be paid to this aspect of patient history.
Continue to: Is it really a first-time seizure?
Is it really a first-time seizure? A “first,” usually dramatic, generalized tonic-clonic seizure that triggers the diagnostic work-up may not be the very first seizure. Evidence suggests that many patients have experienced prior undiagnosed seizures. Subtle prior events often missed include episodes of deja vu, transient feelings of fear or unusual smells, speech difficulties, staring spells, or myoclonic jerks.1 A routine EEG to record epileptiform discharges and a high-resolution brain MRI to rule out any intracranial pathology are indicated. However, if the EEG indicates a primary generalized (as opposed to focal-onset) epilepsy, a brain MRI may not be needed. If a routine EEG is unrevealing, long-term video-EEG monitoring may be needed to detect an abnormality.
Accuracy of EEG and MRI. Following a first unprovoked seizure, routine EEG to detect epileptiform discharges in adults has yielded a sensitivity of 17.3% and specificity of 94.7%. In evaluating children, these values are 57.8% and 69.6%, respectively.6 If results are equivocal, a 24-hour EEG can increase the likelihood of detecting epileptiform discharges to 89% of patients.7 Brain MRI may detect an abnormality in 12% to 14% of patients with newly diagnosed epilepsy, and in up to 80% of those with recurrent seizures.8 In confirming hippocampus sclerosis, MRI has demonstrated a sensitivity of 93% and specificity of 86%.9
When to treat a first-time seizure. Available data and prediction models identify risk factors that would help determine whether to start an antiseizure medication after a first unprovoked seizure:
Epilepsy diagnosis
The International League Against Epilepsy (ILAE) previously defined epilepsy as 2 unprovoked seizures more than 24 hours apart. However, a more recent ILAE task force modified this definition: even a single unprovoked seizure would be enough to diagnose epilepsy if there is high probability of further seizures—eg, in the presence of definitive epileptiform discharges on EEG or presence of a brain tumor or a remote brain insult on imaging, since such conditions induce an enduring predisposition to generate epileptic seizures. 2 Also, a single unprovoked seizure is enough to diagnose epilepsy if it is part of an epileptic syndrome such as juvenile myoclonic epilepsy. Further, a time limit was added to the definition—ie, epilepsy is considered resolved if a patient remains seizure free for 10 years without use of antiseizure medications during the past 5 years. However, given the multitude of variables and evidence, the task force acknowledged the need for individualized considerations. 2
Seizure classification
Classification of seizure type is based on the site of seizure onset and its spread pattern—ie, focal, generalized, or unknown onset.
Continue to: Focal-onset seizures
Focal-onset seizures originate “within networks limited to one hemisphere,” although possibly in more than 1 region (ie, multifocal, and presence or absence of loss of awareness). 12 Focal seizures may then be further classified into “motor onset” or “nonmotor onset” (eg, autonomic, emotional, sensory). 2
Generalized seizures are those “originating at some point within, and rapidly engaging, bilaterally distributed networks.” 13 Unlike focal-onset seizures, generalized seizures are not classified based on awareness, as most generalized seizures involve loss of awareness (absence) or total loss of consciousness (generalized tonic-clonic). They are instead categorized based on the presence of motor vs nonmotor features (eg, tonic-clonic, myoclonic, atonic). Epilepsy classification is quite dynamic and constantly updated based on new genetic, electroencephalographic, and neuroimaging discoveries.
Treatment of epilepsy
Antiseizure medications
Treatment with antiseizure medications (ASMs; formerly known as antiepileptic drugs ) is the mainstay of epilepsy management. Achieving efficacy (seizure freedom) and tolerability (minimal adverse effects) are the primary goals of treatment. Factors that should govern the selection of an ASM include the seizure type/epilepsy syndrome, adverse effect profile of the ASM, pharmacodynamic/pharmacokinetic considerations, and patient comorbidities.
The Standard and New Antiepileptic Drugs (SANAD I and II) trials provide data from direct, unblinded, and longitudinal comparisons of existing and new ASMs and their utility in different seizure types. In the SANAD I cohort of patients with generalized and unclassified epilepsies, valproate was superior to lamotrigine and topiramate for 12-month remission and treatment failure rates, respectively.14 However, valproate generally is avoided in women of childbearing age due its potential adverse effects during pregnancy. In focal epilepsies, lamotrigine was superior to carbamazepine, gabapentin, and topiramate with respect to treatment failure, and noninferior to carbamazepine for 12-month remission.15 In the SANAD II trial, levetiracetam was noninferior to valproate for incidence of adverse events in patients with generalized and unclassified epilepsies although was found to be neither more clinically effective nor more cost effective.16 For patients of childbearing potential with generalized and unclassified epilepsies, there is evidence to support the safe and effective use of levetiracetam.17 In focal epilepsies, lamotrigine was superior to levetiracetam and zonisamide with respect to treatment failures and adverse events and was noninferior to zonisamide for 12-month remission.18 In summary, levetiracetam and valproate (not to be used in women of childbearing potential) are considered appropriate first-line agents for generalized and unclassified epilepsies while lamotrigine is deemed an appropriate first-line agent for focal epilepsies (TABLE 119-28).
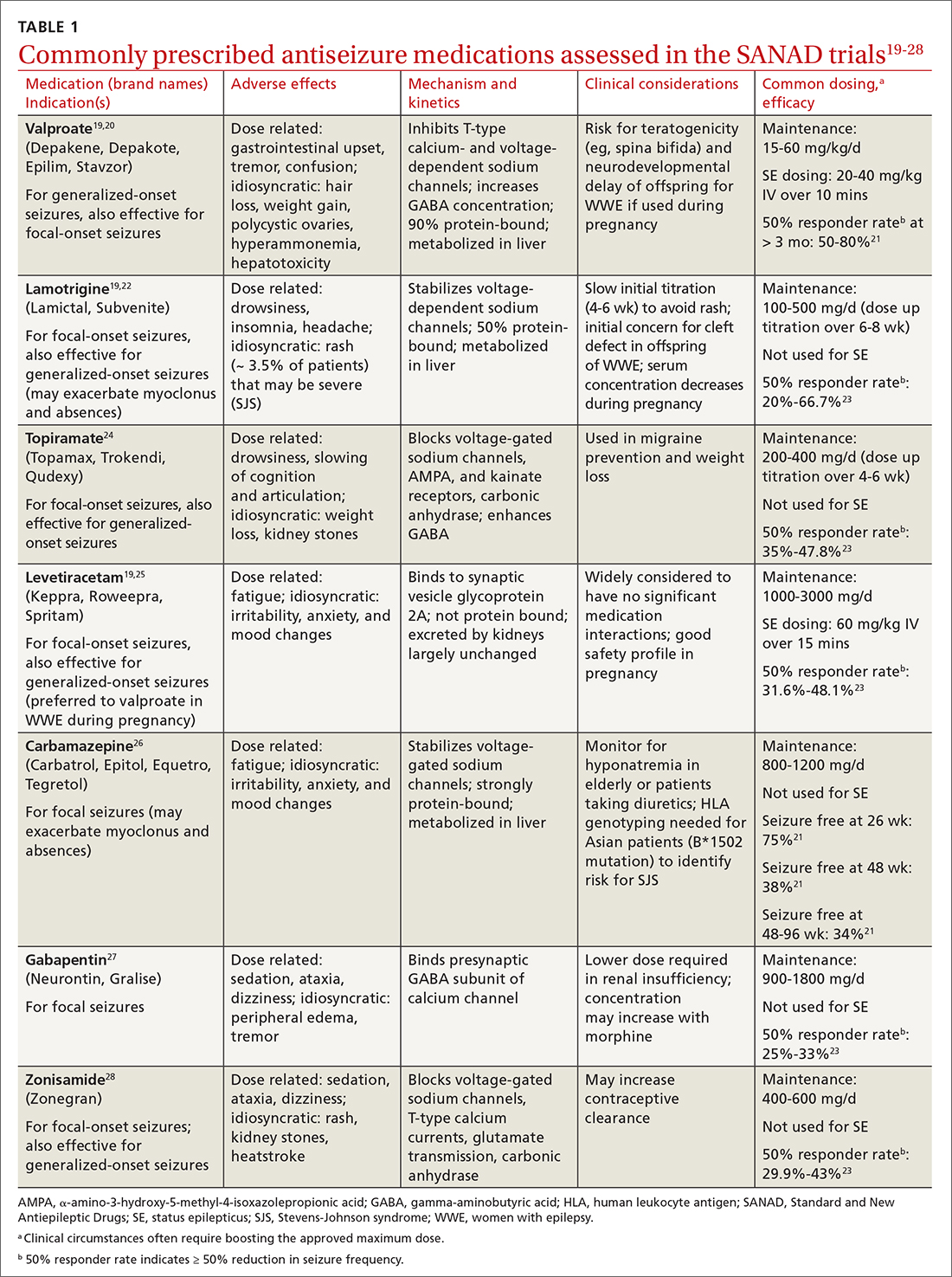
Drug level monitoring. It is standard practice to periodically monitor serum levels in patients taking first-generation ASMs such as phenytoin, carbamazepine, phenobarbital, and valproic acid because of their narrow therapeutic range and the potential for overdose or interaction with other medications or foods (eg, grapefruit juice may increase carbamazepine serum level by inhibiting CYP3A4, the enzyme that metabolizes the drug). Patients taking newer ASMs may not require regular serum level monitoring except during titration, with hepatic or renal dosing, when concomitantly used with estrogen-based oral contraceptives (eg, lamotrigine), before or during pregnancy, or when nonadherence is suspected.
Continue to: Can antiseizure treatment be stopped?
Can antiseizure treatment be stopped?
Current evidence favors continuing ASM therapy in patients whose seizures are under control, although the decision should be tailored to an individual’s circumstances. According to the 2021 American Academy of Neurology (AAN) guidelines, adults who have been seizure free for at least 2 years and discontinue ASMs are possibly still at higher risk for seizure recurrence in the long term (24-60 months), compared with those who continue treatment.29 On the other hand, for adults who have been seizure free for at least 12 months, ASM withdrawal may not increase their risk for status epilepticus, and there are insufficient data to support or refute an effect on mortality or quality of life with ASM withdrawal in this population. The decision to taper or maintain ASM therapy in seizure-free patients also should take into consideration other clinically relevant outcome measures such as the patient’s lifestyle and medication adverse effects. Therefore, this decision should be made after sufficient discussion with patients and their caregivers. (Information for patients can be found at: www.epilepsy.com/treatment/medicines/stopping-medication.)
For children, the AAN guideline panel recommends discussing with family the small risk (2%) for becoming medication resistant if seizures recur during or after ASM withdrawal. 29 For children who have been seizure free for 18 to 24 months, there is probably not a significant long-term (24-48 months) difference in seizure recurrence in those who taper ASMs vs those who do not. However, presence of epileptiform discharges on EEG before discontinuation of an ASM indicates increased risk for seizure recurrence. 29
Intractable (refractory) epilepsy
While most patients with epilepsy attain complete seizure control with appropriate drug therapy, approximately 30% continue to experience seizures (“drug-resistant” epilepsy, also termed intractable or refractory ). 30 In 2010, the ILAE defined drug-resistant epilepsy as “failure of adequate trials of two tolerated, appropriately chosen and used anti-epileptic drug schedules (whether as monotherapy or in combination) to achieve sustained seizure freedom” (defined as cessation of seizures for at least 3 times the longest pre-intervention inter-seizure interval or 12 months, whichever is longer). 21,31 It should be noted that drug withdrawal due to adverse effects is not counted as failure of that ASM. Recognition of drug-resistant epilepsy may prompt referral to an epileptologist who can consider rational combination drug therapy or surgical resection of the seizure focus, vagus nerve stimulation, electrical stimulation of the seizure focus, or deep brain (thalamic) stimulation.
Seizure triggers and mitigating factors
Epilepsy mostly affects patients during seizure episodes; however, the unpredictability of these events adds significantly to the burden of disease. There are no reliable methods for predicting seizure other than knowing of the several potential risks and recognizing and avoiding these triggers.
Noncompliance with antiseizure medications is a common seizure trigger affecting up to one-half of patients with epilepsy.32
Continue to: Medications
Medications may provoke seizures in susceptible individuals
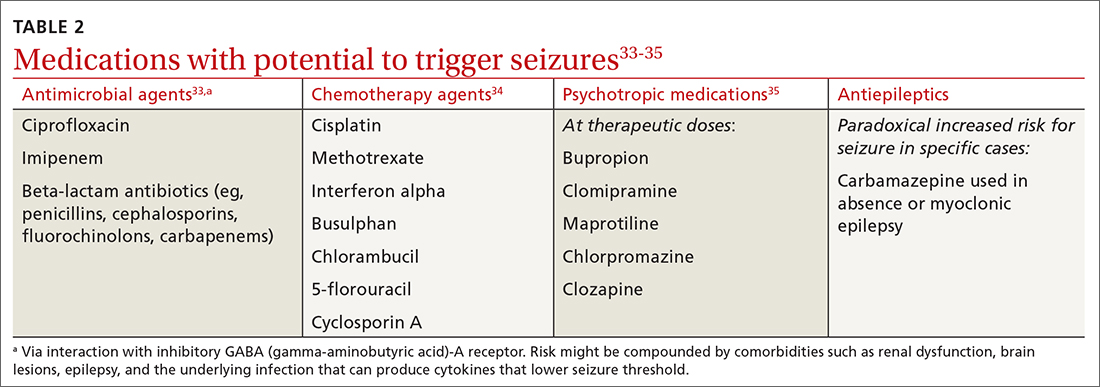
Sleep deprivation is a potential seizure trigger in people with epilepsy based on observational studies, case reports, patient surveys, and EEG-based studies, although data from randomized controlled studies are limited.36 The standard best practice is to encourage appropriate sleep hygiene, which involves getting at least 7 hours of sleep per night.37
Alcohol is a GABAergic substance like benzodiazepines with antiseizure effects. However, it acts as a potential precipitant of seizures in cases of withdrawal or acute intoxication, or when it leads to sleep disruption or nonadherence to antiseizure medications. Therefore, advise patients with alcohol use disorder to slowly taper consumption (best done through a support program) and avoid sudden withdrawal. However, complete abstinence from alcohol use is not often recommended except in special circumstances (eg, a history of alcohol-related seizures). Several studies have demonstrated that modest alcohol use (1-2 drinks per occasion) does not increase seizure frequency or significantly alter serum concentrations of commonly used ASMs.38
Cannabis and other substances. The 2 main biologically active components of marijuana are delta-9-tetrahydrocannibinol (THC), the main psychoactive constituent, and cannabidiol (CBD). Animal and human studies have demonstrated anticonvulsant properties of THC and CBD. But THC, in high amounts, can result in adverse cognitive effects and worsening seizures.39 A purified 98% oil-based CBD extract (Epidiolex) has been approved as an adjunctive treatment for certain medically refractory epilepsy syndromes in children and young adults—ie, Dravet syndrome, Lennox-Gastaut syndrome, and tuberous sclerosis complex syndrome.40 There are no reliable data on the effect of recreational use of marijuana on seizure control. Other illicit substances such as cocaine may lower seizure threshold by their stimulatory and disruptive effects on sleep, diet, and healthy routines.
Special clinical cases
Pregnancy and epilepsy
Despite the potential adverse effects of ASMs on fetal health, the current global consensus is to continue treatment during pregnancy, given that the potential harm of convulsive seizures outweighs the potential risks associated with in-utero exposure to ASMs. There is not enough evidence to indicate significant harm to the fetus caused by focal, absence, or myoclonic seizures. Low-dose folic acid is used to minimize the risks of ASMs during pregnancy.
Continue to: As the fetus develops...
As the fetus develops, there are changes in volume of ASM distribution, renal clearance, protein binding, and hepatic metabolism, which require checking serum levels at regular intervals and making dosage adjustments.
The ongoing study evaluating Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD)41 has led to multiple landmark studies guiding the choice of preferred ASMs during pregnancy in patients with epilepsy.42,43 This has culminated in today’s use of lamotrigine and levetiracetam as the 2 preferred agents (while avoiding valproate) in pregnant patients with epilepsy.44
Psychogenic nonepileptic seizures
A form of conversion disorder, psychogenic nonepileptic seizures (PNES) manifests as abnormal motor or behavioral events mimicking seizures but without associated epileptiform discharges on EEG. This is observed in 10% of patients seen in epilepsy clinics and even more often in those admitted to epilepsy monitoring units (25%-40%).45 Diagnosis of PNES requires EEG monitoring both for confirmation and for discernment from true epileptic seizures, in particular frontal lobe epilepsy that may clinically mimic PNES. PNES often is associated with underlying psychological tensions or comorbid conditions such as depression, anxiety, or traumatic life experiences. There is no treatment for PNES per se, and its management is focused on controlling any underlying psychological comorbidities that may not always be obvious. There is some evidence suggesting that these patients experience an innate inability to verbally express their emotions and instead subconsciously resort to psychosomatics to express them in a somatic dimension.46,47
Status epilepticus
Defined as prolonged seizures (> 5 min) or 2 consecutive seizures without regaining aware ness in between, status epilepticus (SE) is a potentially fatal condition. Subclinical nonconvulsive SE, especially in comatose patients, can be diagnosed only via EEG monitoring. Untreated SE may manifest as a diagnostic dilemma in unresponsive or critically ill patients and can increase the risk for mortality. 48
Febrile seizures
Febrile seizures affect 2% to 5% of children most often in the second year of life.49 The use of preventive antiseizure medication is not recommended; instead, the key is to investigate the underlying febrile illness. Lumbar puncture is indicated if there are signs and symptoms of meningitis (25% of children with bacterial meningitis present with seizures).49 Febrile seizures often are self-limited, but there is risk for SE in up to 15% of cases.50 If convulsive febrile seizures last longer than 5 minutes, initiate benzodiazepines followed by the standard protocol used for the management of SE.51
Continue to: Epilepsy as a spectrum disorder
Epilepsy as a spectrum disorder
The higher prevalence of comorbid cognitive and psychiatric conditions in patients with epilepsy, affecting about half of patients, 52 suggests that seizures may constitute only one aspect of a multifaceted disease that otherwise should be considered a spectrum disorder. Among such conditions are memory deficits, depression, and anxiety. Conversely, epilepsy is more common in patients with depression than in those without. 52
Social impact of epilepsy
Vehicle driving regulations. Patients with epilepsy are required to follow state law regarding driving restrictions. Different states have different rules and regulations about driving restrictions and reporting requirements (by patients or their physicians). Refer patients to the Department of Motor Vehicles (DMV) in their state of residence for up-to-date instructions.53 The Epilepsy Foundation (epilepsy.com) can serve as a resource for each state’s DMV website.
Employment assistance. Having epilepsy should not preclude patients from seeking employment and pursuing meaningful careers. The Americans with Disabilities Act (ADA) and the US Equal Employment Opportunity Commission (EEOC) forbid discrimination against qualified people with disabilities, including those with epilepsy, and require reasonable accommodations in the workplace (www.eeoc.gov/laws/guidance/epilepsy-workplace-and-ada).54
CORRESPONDENCE
Gholam K. Motamedi, MD, Department of Neurology, PHC 7, Georgetown University Hospital, 3800 Reservoir Road, NW, Washington, DC 20007; motamedi@georgetown.edu
1. Hauser WA, Annegers JF, Rocca WA. Descriptive epidemiology of epilepsy: contributions of population-based studies from Rochester, Minnesota. Mayo Clin Proc. 1996;71:576-586. doi: 10.4065/71.6.576
2. Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55:475-482. doi: 10.1111/epi.12550.
3. Hauser WA, Beghi E. First seizure definitions and worldwide incidence and mortality. Epilepsia. 2008;49:8-12. doi: 10.1111/j.1528-1167.2008.01443.x
4. Berg AT, Shinnar S. The risk of seizure recurrence following a first unprovoked seizure: a quantitative review. Neurology. 1991;41:965-972. doi: 10.1212/wnl.41.7.965
5. Ropper AH. Transient global amnesia. N Engl J Med. 2023;388:635-540. doi: 10.1056/NEJMra2213867
6. Bouma HK, Labos C, Gore GC, et al. The diagnostic accuracy of routine electroencephalography after a first unprovoked seizure. Eur J Neurol. 2016;23:455-463. doi: 10.1111/ene.12739
7. Narayanan JT, Labar DR, Schaul N. Latency to first spike in the EEG of epilepsy patients. Seizure. 2008;17:34-41. doi: 10.1016/j.seizure.2007.06.003
8. Salmenpera TM, Duncan JS. Imaging in epilepsy. J Neurol Neurosurg Psychiatry. 2005;76:iii2-iii10. doi: 10.1136/jnnp.2005.075135
9. Jackson GD, Berkovic SF, Tress , et al Hippocampal sclerosis can be reliably detected by magnetic resonance imaging. Neurology. 1990;40:1869-1875. doi: 10.1212/wnl.40.12.1869
10. Bonnett LJ, Kim, L, Johnson A, et al. Risk of seizure recurrence in people with single seizures and early epilepsy - model development and external validation. Seizure. 2022;94:26-32. doi: 10.1016/j.seizure.2021.11.007
11. Krumholz A, Wiebe S, Gronseth GS, et al. Evidence-based guideline: management of an unprovoked first seizure in adults: Report of the Guideline Development Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2015;84:1705-1713. doi: 10.1212/WNL.0000000000001487
12. Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE Commission for Classification and terminology. Epilepsia. 2017;58:522-530. doi: 10.1111/epi.13670
13. Berg AT, Berkovic SF, Brodie MJ, et al. Revised terminology and concepts for organization of seizures and epilepsy: report of the ILAE Commission on Classification and Terminology, 2005-2009. Epilepsia. 2010;51:676-685. doi: 10.1111/j.1528-1167.2010.02522.x
14. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of valproate, lamotrigine, or topiramate for generalized and unclassifiable epilepsy: an unblinded randomized controlled trial. Lancet. 2007;369:1016-1026. doi: 10.1016/S0140-6736(07)60461-9
15. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy: an unblinded randomized controlled trial. Lancet 2007;369:1000-1015. doi: 10.1016/S0140-6736(07)60460-7
16. Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of valproate versus levetiracetam for newly diagnosed generalized and unclassified epilepsy: an open-label, non-inferiority, multicentre, phase 4, randomized controlled trial. Lancet. 2021;397:1375-1386. doi: 10.1016/S0140-6736(21)00246-4
17. Mawhinney E, Craig J, Morrow J. Levetiracetam in pregnancy: results from the UK and Ireland epilepsy and pregnancy registers. Neurology. 2013;80:400-405.
18. Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of levetiracetam, zonisamide, or lamotrigine for newly diagnosed focal epilepsy: an open-label, non-inferiority, multicentre, phase 4, randomized controlled trial. Lancet. 2021;397:1363-1374. doi: 10.1016/S0140-6736(21)00247-6
19. Smith PE. Initial management of seizure in adults. N Engl J Med. 2021;385:251-263. doi: 10.1056/NEJMcp2024526
20. Depakene (valproic acid). Package insert. Abbott Laboratories; 2011. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2011/018081s046_18082s031lbl.pdf
21. Greenberg RG, Melloni C, Wu H, et al. Therapeutic index estimation of antiepileptic drugs: a systematic literature review approach. Clin Neuropharmacol. 2016;39:232-240. doi: 10.1097/WNF.0000000000000172
22. Lamictal (lamotrigine). Package insert. GlaxoSmithKline; 2009. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2009/020241s037s038,020764s030s031lbl.pdf
23. LaRoche SM, Helmers SL. The new antiepileptic drugs: scientific review. JAMA. 2004;291:605-614. doi: 10.1001/jama.291.5.605
24. Topamax (topiramate). Package insert. Janssen Pharmaceuticals, Inc. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2012/020844s041lbl.pdf
25. Keppra (levetiracetam). Package insert. UCB, Inc.; 2009. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2009/021035s078s080%2C021505s021s024lbl.pdf
26. Carbatrol (carbamazepine). Package insert. Shire US Inc; 2013. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2013/020712s032s035lbl.pdf
27.Neurontin (gabapentin). Package insert. Pfizer; 2017. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2017/020235s064_020882s047_021129s046lbl.pdf
28.Zonegran (zonisamide). Package insert. Eisai Inc; 2006. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2006/020789s019lbl.pdf
29.Gloss D, Paragon K, Pack A, et al. Antiseizure medication withdrawal in seizure-free patients: practice advisory update. Report of the AAN Guideline Subcommittee. Neurology. 2021;97:1072-1081. doi: 10.1212/WNL.0000000000012944
30.Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000:342:314-319. doi: 10.1056/NEJM200002033420503
31.Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia. 2010;51:1069-1077. doi: 10.1111/j.1528-1167.2009.02397.x
Compliance during treatment of epilepsy. Epilepsia 1988;29(suppl 2):S79-S84.
33.utter R, Rüegg S, Tschudin-Sutter S. Seizures as adverse events of antibiotic drugs: a systematic review. Neurology. 2015;13;85:1332-1341. doi: 10.1212/WNL.0000000000002023
34.Singh G, Rees JH, Sander JW. Seizures and epilepsy in oncological practice: causes, course, mechanisms and treatment. JNNP. 2007;78:342-349. doi: 10.1136/jnnp.2006.106211
35.Pisani F, Oteri G, Costa C., et al. Effects of psychotropic drugs on seizure threshold. Drug Safety. 2002;25:91-110.
36.Rossi KC, Joe J, Makhjia M, et al. Insufficient sleep, electroencephalogram activation, and seizure risk: re-evaluating the evidence. Ann Neurol. 2020;86:798-806. doi: 10.1002/ana.25710
37.Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843-844. doi: 10.5665/sleep.4716
38.Höppener RJ, Kuyer A, van der Lugt PJ. Epilepsy and alcohol: the influence of social alcohol intake on seizures and treatment in epilepsy. Epilepsia. 1983;24:459-471. doi: 10.1111/j.1528-1157.1983.tb04917.x
39.Keeler MH, Reifler CB. Grand mal convulsions subsequent to marijuana use. Case report. Dis Nerv Syst. 1967:28:474-475.
40.Epidiolex (cannabidiol). Package insert. Greenwich Biosciences Inc; 2018. Accessed September 27, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf
41.ClinicalTrials.gov. Maternal Outcomes and Neurodevelopmental Effects of Antiseizure Drugs (MONEAD). Accessed September 24, 2023. https://classic.clinicaltrials.gov/ct2/show/NCT01730170
42.Meador KJ, Baker GA, Finnell RH, et al. In utero antiepileptic drug exposure: fetal death and malformations. Neurology. 2006;67:407-412. doi: 10.1212/01.wnl.0000227919.81208.b2
43.Meador K, Reynolds MW, Crean S. Pregnancy outcomes in women with epilepsy: a systematic review and meta-analysis of published pregnancy registries and cohorts. Epilepsy Res. 2008;81:1-13. doi:10.1016/j.eplepsyres.2008.04.022
44.Marxer CA, Rüegg S, Rauch A review of the evidence on the risk of congenital malformations and neurodevelopmental disorders in association with antiseizure medications during pregnancy. Expert Opin Drug Saf. 2021;20:1487-1499. doi: 10.1080/14740338.2021.1943355
Asadi-Pooya AA, Sperling MR. Epidemiology of psychogenic nonepileptic seizures. Epilepsy Behav. 2015;46:60-65. doi: 10.1016/j.yebeh.2015.03.015
Evaluation and treatment of psychogenic nonepileptic seizures. Neurol Clin. 2022;40:799-820. doi: 10.1016/j.ncl.2022.03.017
47.Motamedi GK. Psychogenic nonepileptic seizures: a disconnect between body and mind. Epilepsy Behav. 2018;78:293-294. doi: 10.1016/j.yebeh.2017.10.016
, Nonconvulsive status epilepticus. Emerg Med Clin North Am. 2011;29:65-72. doi: 10.1016/j.emc.2010.08.006
doi: 10.1542/peds.2010-3318
Drug management for acute tonic-clonic convulsions including convulsive status epilepticus in children. Cochrane Database Sys Rev. 2018;1(1):CD001905. doi: 10.1002/14651858.CD001905.pub3
52.Jensen FE. Epilepsy as a spectrum disorder: implications from novel clinical and basic neuroscience. Epilepsia. 2011;52(suppl 1):1-6. doi: 10.1111/j.1528-1167.2010.02904.x
53.Kass JS, Rose RV. Driving and epilepsy: ethical, legal, and health care policy challenges. Continuum (Minneap Minn). 2019;25:537-542. doi: 10.1212/CON.0000000000000714
54.Troxell J. Epilepsy and employment: the Americans with Disabilities Act and its protections against employment discrimination. Med Law. 1997;16:375-384.
Managing first-time seizures and epilepsy often requires consultation with a neurologist or epileptologist for diagnosis and subsequent management, including when medical treatment fails or in determining whether patients may benefit from surgery. However, given the high prevalence of epilepsy and even higher incidence of a single seizure, family physicians contribute significantly to the management of these patients. The main issues are managing a first-time seizure, making the diagnosis, establishing a treatment plan, and exploring triggers and mitigating factors.
Seizure vs epilepsy
All patients with epilepsy experience seizures, but not every person who experiences a seizure has (or will develop) epilepsy. Nearly 10% of the population has one seizure during their lifetime, whereas the risk for epilepsy is just 3%.1 Therefore, a first-time seizure may not herald epilepsy, defined as repetitive (≥ 2) unprovoked seizures more than 24 hours apart.2 Seizures can be provoked (acute symptomatic) or unprovoked; a clear distinction between these 2 occurrences—as well as between single and recurrent seizures—is critical for proper management. A close look at the circumstances of a first-time seizure is imperative to define the nature of the event and the possibility of further seizures before devising a treatment plan.
Provoked seizures are due to an acute brain insult such as toxic-metabolic disorders, concussion, alcohol withdrawal, an adverse effect of a medication or its withdrawal, or photic stimulation presumably by disrupting the brain’s metabolic homeostasis or integrity. The key factor is that provoked seizures always happen in close temporal association with an acute insult. A single provoked seizure happens each year in 29 to 39 individuals per 100,000.3 While these seizures typically occur singly, there is a small risk they may recur if the triggering insult persists or repeats.1 Therefore, more than 1 seizure per se may not indicate epilepsy.3
Unprovoked seizures reflect an underlying brain dysfunction. A single unprovoked seizure happens in 23 to 61 individuals per 100,000 per year, often in men in either younger or older age groups.3 Unprovoked seizures may occur only once or may recur (ie, evolve into epilepsy). The latter scenario happens in only about half of cases; the overall risk for a recurrent seizure within 2 years of a first seizure is estimated at 42% (24% to 65%, depending on the etiology and electroencephalogram [EEG] findings).4 More specifically, without treatment the relapse rate will be 36% at 1 year and 47% at 2 years.4 Further, a second unprovoked seizure, if untreated, would increase the risk for third and fourth seizures to 73% and 76%, respectively, within 4 years.3
Evaluating the first-time seizure
Ask the patient or observers about the circumstances of the event to differentiate provoked from unprovoked onset. For one thing, not all “spells” are seizures. The differential diagnoses may include syncope, psychogenic nonepileptic events, drug intoxication or withdrawal, migraine, panic attacks, sleep disorders (parasomnia), transient global amnesia, concussion, and transient ischemic attack. EEG, neuroimaging, and other relevant diagnostic tests often are needed (eg, electrocardiogram/echocardiogram/Holter monitoring to evaluate for syncope/cardiac arrhythmia). Clinically, syncopal episodes tend to be brief with rapid recovery and no confusion, speech problems, aura, or lateralizing signs such as hand posturing or lip smacking that are typical with focal seizures. However, cases of convulsive syncope can be challenging to assess without diagnostic tests.
True convulsive seizures do not have the variability in clinical signs seen with psychogenic nonepileptic events (eg, alternating body parts involved or direction of movements). Transient global amnesia is a rare condition with no established diagnostic test and is considered a diagnosis of exclusion, although bitemporal hyperintensities on magnetic resonance imaging (MRI) may appear 12 to 48 hours after the clinical episode.5 Blood work is needed in patients with medical issues treated with multiple medications to evaluate for metabolic derangements; otherwise, routine blood work provides minimal information in stable patients.
Region-specific causes. Neurocysticercosis is common in some regions, such as Latin America; therefore, attention should be paid to this aspect of patient history.
Continue to: Is it really a first-time seizure?
Is it really a first-time seizure? A “first,” usually dramatic, generalized tonic-clonic seizure that triggers the diagnostic work-up may not be the very first seizure. Evidence suggests that many patients have experienced prior undiagnosed seizures. Subtle prior events often missed include episodes of deja vu, transient feelings of fear or unusual smells, speech difficulties, staring spells, or myoclonic jerks.1 A routine EEG to record epileptiform discharges and a high-resolution brain MRI to rule out any intracranial pathology are indicated. However, if the EEG indicates a primary generalized (as opposed to focal-onset) epilepsy, a brain MRI may not be needed. If a routine EEG is unrevealing, long-term video-EEG monitoring may be needed to detect an abnormality.
Accuracy of EEG and MRI. Following a first unprovoked seizure, routine EEG to detect epileptiform discharges in adults has yielded a sensitivity of 17.3% and specificity of 94.7%. In evaluating children, these values are 57.8% and 69.6%, respectively.6 If results are equivocal, a 24-hour EEG can increase the likelihood of detecting epileptiform discharges to 89% of patients.7 Brain MRI may detect an abnormality in 12% to 14% of patients with newly diagnosed epilepsy, and in up to 80% of those with recurrent seizures.8 In confirming hippocampus sclerosis, MRI has demonstrated a sensitivity of 93% and specificity of 86%.9
When to treat a first-time seizure. Available data and prediction models identify risk factors that would help determine whether to start an antiseizure medication after a first unprovoked seizure:
Epilepsy diagnosis
The International League Against Epilepsy (ILAE) previously defined epilepsy as 2 unprovoked seizures more than 24 hours apart. However, a more recent ILAE task force modified this definition: even a single unprovoked seizure would be enough to diagnose epilepsy if there is high probability of further seizures—eg, in the presence of definitive epileptiform discharges on EEG or presence of a brain tumor or a remote brain insult on imaging, since such conditions induce an enduring predisposition to generate epileptic seizures. 2 Also, a single unprovoked seizure is enough to diagnose epilepsy if it is part of an epileptic syndrome such as juvenile myoclonic epilepsy. Further, a time limit was added to the definition—ie, epilepsy is considered resolved if a patient remains seizure free for 10 years without use of antiseizure medications during the past 5 years. However, given the multitude of variables and evidence, the task force acknowledged the need for individualized considerations. 2
Seizure classification
Classification of seizure type is based on the site of seizure onset and its spread pattern—ie, focal, generalized, or unknown onset.
Continue to: Focal-onset seizures
Focal-onset seizures originate “within networks limited to one hemisphere,” although possibly in more than 1 region (ie, multifocal, and presence or absence of loss of awareness). 12 Focal seizures may then be further classified into “motor onset” or “nonmotor onset” (eg, autonomic, emotional, sensory). 2
Generalized seizures are those “originating at some point within, and rapidly engaging, bilaterally distributed networks.” 13 Unlike focal-onset seizures, generalized seizures are not classified based on awareness, as most generalized seizures involve loss of awareness (absence) or total loss of consciousness (generalized tonic-clonic). They are instead categorized based on the presence of motor vs nonmotor features (eg, tonic-clonic, myoclonic, atonic). Epilepsy classification is quite dynamic and constantly updated based on new genetic, electroencephalographic, and neuroimaging discoveries.
Treatment of epilepsy
Antiseizure medications
Treatment with antiseizure medications (ASMs; formerly known as antiepileptic drugs ) is the mainstay of epilepsy management. Achieving efficacy (seizure freedom) and tolerability (minimal adverse effects) are the primary goals of treatment. Factors that should govern the selection of an ASM include the seizure type/epilepsy syndrome, adverse effect profile of the ASM, pharmacodynamic/pharmacokinetic considerations, and patient comorbidities.
The Standard and New Antiepileptic Drugs (SANAD I and II) trials provide data from direct, unblinded, and longitudinal comparisons of existing and new ASMs and their utility in different seizure types. In the SANAD I cohort of patients with generalized and unclassified epilepsies, valproate was superior to lamotrigine and topiramate for 12-month remission and treatment failure rates, respectively.14 However, valproate generally is avoided in women of childbearing age due its potential adverse effects during pregnancy. In focal epilepsies, lamotrigine was superior to carbamazepine, gabapentin, and topiramate with respect to treatment failure, and noninferior to carbamazepine for 12-month remission.15 In the SANAD II trial, levetiracetam was noninferior to valproate for incidence of adverse events in patients with generalized and unclassified epilepsies although was found to be neither more clinically effective nor more cost effective.16 For patients of childbearing potential with generalized and unclassified epilepsies, there is evidence to support the safe and effective use of levetiracetam.17 In focal epilepsies, lamotrigine was superior to levetiracetam and zonisamide with respect to treatment failures and adverse events and was noninferior to zonisamide for 12-month remission.18 In summary, levetiracetam and valproate (not to be used in women of childbearing potential) are considered appropriate first-line agents for generalized and unclassified epilepsies while lamotrigine is deemed an appropriate first-line agent for focal epilepsies (TABLE 119-28).

Drug level monitoring. It is standard practice to periodically monitor serum levels in patients taking first-generation ASMs such as phenytoin, carbamazepine, phenobarbital, and valproic acid because of their narrow therapeutic range and the potential for overdose or interaction with other medications or foods (eg, grapefruit juice may increase carbamazepine serum level by inhibiting CYP3A4, the enzyme that metabolizes the drug). Patients taking newer ASMs may not require regular serum level monitoring except during titration, with hepatic or renal dosing, when concomitantly used with estrogen-based oral contraceptives (eg, lamotrigine), before or during pregnancy, or when nonadherence is suspected.
Continue to: Can antiseizure treatment be stopped?
Can antiseizure treatment be stopped?
Current evidence favors continuing ASM therapy in patients whose seizures are under control, although the decision should be tailored to an individual’s circumstances. According to the 2021 American Academy of Neurology (AAN) guidelines, adults who have been seizure free for at least 2 years and discontinue ASMs are possibly still at higher risk for seizure recurrence in the long term (24-60 months), compared with those who continue treatment.29 On the other hand, for adults who have been seizure free for at least 12 months, ASM withdrawal may not increase their risk for status epilepticus, and there are insufficient data to support or refute an effect on mortality or quality of life with ASM withdrawal in this population. The decision to taper or maintain ASM therapy in seizure-free patients also should take into consideration other clinically relevant outcome measures such as the patient’s lifestyle and medication adverse effects. Therefore, this decision should be made after sufficient discussion with patients and their caregivers. (Information for patients can be found at: www.epilepsy.com/treatment/medicines/stopping-medication.)
For children, the AAN guideline panel recommends discussing with family the small risk (2%) for becoming medication resistant if seizures recur during or after ASM withdrawal. 29 For children who have been seizure free for 18 to 24 months, there is probably not a significant long-term (24-48 months) difference in seizure recurrence in those who taper ASMs vs those who do not. However, presence of epileptiform discharges on EEG before discontinuation of an ASM indicates increased risk for seizure recurrence. 29
Intractable (refractory) epilepsy
While most patients with epilepsy attain complete seizure control with appropriate drug therapy, approximately 30% continue to experience seizures (“drug-resistant” epilepsy, also termed intractable or refractory ). 30 In 2010, the ILAE defined drug-resistant epilepsy as “failure of adequate trials of two tolerated, appropriately chosen and used anti-epileptic drug schedules (whether as monotherapy or in combination) to achieve sustained seizure freedom” (defined as cessation of seizures for at least 3 times the longest pre-intervention inter-seizure interval or 12 months, whichever is longer). 21,31 It should be noted that drug withdrawal due to adverse effects is not counted as failure of that ASM. Recognition of drug-resistant epilepsy may prompt referral to an epileptologist who can consider rational combination drug therapy or surgical resection of the seizure focus, vagus nerve stimulation, electrical stimulation of the seizure focus, or deep brain (thalamic) stimulation.
Seizure triggers and mitigating factors
Epilepsy mostly affects patients during seizure episodes; however, the unpredictability of these events adds significantly to the burden of disease. There are no reliable methods for predicting seizure other than knowing of the several potential risks and recognizing and avoiding these triggers.
Noncompliance with antiseizure medications is a common seizure trigger affecting up to one-half of patients with epilepsy.32
Continue to: Medications
Medications may provoke seizures in susceptible individuals

Sleep deprivation is a potential seizure trigger in people with epilepsy based on observational studies, case reports, patient surveys, and EEG-based studies, although data from randomized controlled studies are limited.36 The standard best practice is to encourage appropriate sleep hygiene, which involves getting at least 7 hours of sleep per night.37
Alcohol is a GABAergic substance like benzodiazepines with antiseizure effects. However, it acts as a potential precipitant of seizures in cases of withdrawal or acute intoxication, or when it leads to sleep disruption or nonadherence to antiseizure medications. Therefore, advise patients with alcohol use disorder to slowly taper consumption (best done through a support program) and avoid sudden withdrawal. However, complete abstinence from alcohol use is not often recommended except in special circumstances (eg, a history of alcohol-related seizures). Several studies have demonstrated that modest alcohol use (1-2 drinks per occasion) does not increase seizure frequency or significantly alter serum concentrations of commonly used ASMs.38
Cannabis and other substances. The 2 main biologically active components of marijuana are delta-9-tetrahydrocannibinol (THC), the main psychoactive constituent, and cannabidiol (CBD). Animal and human studies have demonstrated anticonvulsant properties of THC and CBD. But THC, in high amounts, can result in adverse cognitive effects and worsening seizures.39 A purified 98% oil-based CBD extract (Epidiolex) has been approved as an adjunctive treatment for certain medically refractory epilepsy syndromes in children and young adults—ie, Dravet syndrome, Lennox-Gastaut syndrome, and tuberous sclerosis complex syndrome.40 There are no reliable data on the effect of recreational use of marijuana on seizure control. Other illicit substances such as cocaine may lower seizure threshold by their stimulatory and disruptive effects on sleep, diet, and healthy routines.
Special clinical cases
Pregnancy and epilepsy
Despite the potential adverse effects of ASMs on fetal health, the current global consensus is to continue treatment during pregnancy, given that the potential harm of convulsive seizures outweighs the potential risks associated with in-utero exposure to ASMs. There is not enough evidence to indicate significant harm to the fetus caused by focal, absence, or myoclonic seizures. Low-dose folic acid is used to minimize the risks of ASMs during pregnancy.
Continue to: As the fetus develops...
As the fetus develops, there are changes in volume of ASM distribution, renal clearance, protein binding, and hepatic metabolism, which require checking serum levels at regular intervals and making dosage adjustments.
The ongoing study evaluating Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD)41 has led to multiple landmark studies guiding the choice of preferred ASMs during pregnancy in patients with epilepsy.42,43 This has culminated in today’s use of lamotrigine and levetiracetam as the 2 preferred agents (while avoiding valproate) in pregnant patients with epilepsy.44
Psychogenic nonepileptic seizures
A form of conversion disorder, psychogenic nonepileptic seizures (PNES) manifests as abnormal motor or behavioral events mimicking seizures but without associated epileptiform discharges on EEG. This is observed in 10% of patients seen in epilepsy clinics and even more often in those admitted to epilepsy monitoring units (25%-40%).45 Diagnosis of PNES requires EEG monitoring both for confirmation and for discernment from true epileptic seizures, in particular frontal lobe epilepsy that may clinically mimic PNES. PNES often is associated with underlying psychological tensions or comorbid conditions such as depression, anxiety, or traumatic life experiences. There is no treatment for PNES per se, and its management is focused on controlling any underlying psychological comorbidities that may not always be obvious. There is some evidence suggesting that these patients experience an innate inability to verbally express their emotions and instead subconsciously resort to psychosomatics to express them in a somatic dimension.46,47
Status epilepticus
Defined as prolonged seizures (> 5 min) or 2 consecutive seizures without regaining aware ness in between, status epilepticus (SE) is a potentially fatal condition. Subclinical nonconvulsive SE, especially in comatose patients, can be diagnosed only via EEG monitoring. Untreated SE may manifest as a diagnostic dilemma in unresponsive or critically ill patients and can increase the risk for mortality. 48
Febrile seizures
Febrile seizures affect 2% to 5% of children most often in the second year of life.49 The use of preventive antiseizure medication is not recommended; instead, the key is to investigate the underlying febrile illness. Lumbar puncture is indicated if there are signs and symptoms of meningitis (25% of children with bacterial meningitis present with seizures).49 Febrile seizures often are self-limited, but there is risk for SE in up to 15% of cases.50 If convulsive febrile seizures last longer than 5 minutes, initiate benzodiazepines followed by the standard protocol used for the management of SE.51
Continue to: Epilepsy as a spectrum disorder
Epilepsy as a spectrum disorder
The higher prevalence of comorbid cognitive and psychiatric conditions in patients with epilepsy, affecting about half of patients, 52 suggests that seizures may constitute only one aspect of a multifaceted disease that otherwise should be considered a spectrum disorder. Among such conditions are memory deficits, depression, and anxiety. Conversely, epilepsy is more common in patients with depression than in those without. 52
Social impact of epilepsy
Vehicle driving regulations. Patients with epilepsy are required to follow state law regarding driving restrictions. Different states have different rules and regulations about driving restrictions and reporting requirements (by patients or their physicians). Refer patients to the Department of Motor Vehicles (DMV) in their state of residence for up-to-date instructions.53 The Epilepsy Foundation (epilepsy.com) can serve as a resource for each state’s DMV website.
Employment assistance. Having epilepsy should not preclude patients from seeking employment and pursuing meaningful careers. The Americans with Disabilities Act (ADA) and the US Equal Employment Opportunity Commission (EEOC) forbid discrimination against qualified people with disabilities, including those with epilepsy, and require reasonable accommodations in the workplace (www.eeoc.gov/laws/guidance/epilepsy-workplace-and-ada).54
CORRESPONDENCE
Gholam K. Motamedi, MD, Department of Neurology, PHC 7, Georgetown University Hospital, 3800 Reservoir Road, NW, Washington, DC 20007; motamedi@georgetown.edu
Managing first-time seizures and epilepsy often requires consultation with a neurologist or epileptologist for diagnosis and subsequent management, including when medical treatment fails or in determining whether patients may benefit from surgery. However, given the high prevalence of epilepsy and even higher incidence of a single seizure, family physicians contribute significantly to the management of these patients. The main issues are managing a first-time seizure, making the diagnosis, establishing a treatment plan, and exploring triggers and mitigating factors.
Seizure vs epilepsy
All patients with epilepsy experience seizures, but not every person who experiences a seizure has (or will develop) epilepsy. Nearly 10% of the population has one seizure during their lifetime, whereas the risk for epilepsy is just 3%.1 Therefore, a first-time seizure may not herald epilepsy, defined as repetitive (≥ 2) unprovoked seizures more than 24 hours apart.2 Seizures can be provoked (acute symptomatic) or unprovoked; a clear distinction between these 2 occurrences—as well as between single and recurrent seizures—is critical for proper management. A close look at the circumstances of a first-time seizure is imperative to define the nature of the event and the possibility of further seizures before devising a treatment plan.
Provoked seizures are due to an acute brain insult such as toxic-metabolic disorders, concussion, alcohol withdrawal, an adverse effect of a medication or its withdrawal, or photic stimulation presumably by disrupting the brain’s metabolic homeostasis or integrity. The key factor is that provoked seizures always happen in close temporal association with an acute insult. A single provoked seizure happens each year in 29 to 39 individuals per 100,000.3 While these seizures typically occur singly, there is a small risk they may recur if the triggering insult persists or repeats.1 Therefore, more than 1 seizure per se may not indicate epilepsy.3
Unprovoked seizures reflect an underlying brain dysfunction. A single unprovoked seizure happens in 23 to 61 individuals per 100,000 per year, often in men in either younger or older age groups.3 Unprovoked seizures may occur only once or may recur (ie, evolve into epilepsy). The latter scenario happens in only about half of cases; the overall risk for a recurrent seizure within 2 years of a first seizure is estimated at 42% (24% to 65%, depending on the etiology and electroencephalogram [EEG] findings).4 More specifically, without treatment the relapse rate will be 36% at 1 year and 47% at 2 years.4 Further, a second unprovoked seizure, if untreated, would increase the risk for third and fourth seizures to 73% and 76%, respectively, within 4 years.3
Evaluating the first-time seizure
Ask the patient or observers about the circumstances of the event to differentiate provoked from unprovoked onset. For one thing, not all “spells” are seizures. The differential diagnoses may include syncope, psychogenic nonepileptic events, drug intoxication or withdrawal, migraine, panic attacks, sleep disorders (parasomnia), transient global amnesia, concussion, and transient ischemic attack. EEG, neuroimaging, and other relevant diagnostic tests often are needed (eg, electrocardiogram/echocardiogram/Holter monitoring to evaluate for syncope/cardiac arrhythmia). Clinically, syncopal episodes tend to be brief with rapid recovery and no confusion, speech problems, aura, or lateralizing signs such as hand posturing or lip smacking that are typical with focal seizures. However, cases of convulsive syncope can be challenging to assess without diagnostic tests.
True convulsive seizures do not have the variability in clinical signs seen with psychogenic nonepileptic events (eg, alternating body parts involved or direction of movements). Transient global amnesia is a rare condition with no established diagnostic test and is considered a diagnosis of exclusion, although bitemporal hyperintensities on magnetic resonance imaging (MRI) may appear 12 to 48 hours after the clinical episode.5 Blood work is needed in patients with medical issues treated with multiple medications to evaluate for metabolic derangements; otherwise, routine blood work provides minimal information in stable patients.
Region-specific causes. Neurocysticercosis is common in some regions, such as Latin America; therefore, attention should be paid to this aspect of patient history.
Continue to: Is it really a first-time seizure?
Is it really a first-time seizure? A “first,” usually dramatic, generalized tonic-clonic seizure that triggers the diagnostic work-up may not be the very first seizure. Evidence suggests that many patients have experienced prior undiagnosed seizures. Subtle prior events often missed include episodes of deja vu, transient feelings of fear or unusual smells, speech difficulties, staring spells, or myoclonic jerks.1 A routine EEG to record epileptiform discharges and a high-resolution brain MRI to rule out any intracranial pathology are indicated. However, if the EEG indicates a primary generalized (as opposed to focal-onset) epilepsy, a brain MRI may not be needed. If a routine EEG is unrevealing, long-term video-EEG monitoring may be needed to detect an abnormality.
Accuracy of EEG and MRI. Following a first unprovoked seizure, routine EEG to detect epileptiform discharges in adults has yielded a sensitivity of 17.3% and specificity of 94.7%. In evaluating children, these values are 57.8% and 69.6%, respectively.6 If results are equivocal, a 24-hour EEG can increase the likelihood of detecting epileptiform discharges to 89% of patients.7 Brain MRI may detect an abnormality in 12% to 14% of patients with newly diagnosed epilepsy, and in up to 80% of those with recurrent seizures.8 In confirming hippocampus sclerosis, MRI has demonstrated a sensitivity of 93% and specificity of 86%.9
When to treat a first-time seizure. Available data and prediction models identify risk factors that would help determine whether to start an antiseizure medication after a first unprovoked seizure:
Epilepsy diagnosis
The International League Against Epilepsy (ILAE) previously defined epilepsy as 2 unprovoked seizures more than 24 hours apart. However, a more recent ILAE task force modified this definition: even a single unprovoked seizure would be enough to diagnose epilepsy if there is high probability of further seizures—eg, in the presence of definitive epileptiform discharges on EEG or presence of a brain tumor or a remote brain insult on imaging, since such conditions induce an enduring predisposition to generate epileptic seizures. 2 Also, a single unprovoked seizure is enough to diagnose epilepsy if it is part of an epileptic syndrome such as juvenile myoclonic epilepsy. Further, a time limit was added to the definition—ie, epilepsy is considered resolved if a patient remains seizure free for 10 years without use of antiseizure medications during the past 5 years. However, given the multitude of variables and evidence, the task force acknowledged the need for individualized considerations. 2
Seizure classification
Classification of seizure type is based on the site of seizure onset and its spread pattern—ie, focal, generalized, or unknown onset.
Continue to: Focal-onset seizures
Focal-onset seizures originate “within networks limited to one hemisphere,” although possibly in more than 1 region (ie, multifocal, and presence or absence of loss of awareness). 12 Focal seizures may then be further classified into “motor onset” or “nonmotor onset” (eg, autonomic, emotional, sensory). 2
Generalized seizures are those “originating at some point within, and rapidly engaging, bilaterally distributed networks.” 13 Unlike focal-onset seizures, generalized seizures are not classified based on awareness, as most generalized seizures involve loss of awareness (absence) or total loss of consciousness (generalized tonic-clonic). They are instead categorized based on the presence of motor vs nonmotor features (eg, tonic-clonic, myoclonic, atonic). Epilepsy classification is quite dynamic and constantly updated based on new genetic, electroencephalographic, and neuroimaging discoveries.
Treatment of epilepsy
Antiseizure medications
Treatment with antiseizure medications (ASMs; formerly known as antiepileptic drugs ) is the mainstay of epilepsy management. Achieving efficacy (seizure freedom) and tolerability (minimal adverse effects) are the primary goals of treatment. Factors that should govern the selection of an ASM include the seizure type/epilepsy syndrome, adverse effect profile of the ASM, pharmacodynamic/pharmacokinetic considerations, and patient comorbidities.
The Standard and New Antiepileptic Drugs (SANAD I and II) trials provide data from direct, unblinded, and longitudinal comparisons of existing and new ASMs and their utility in different seizure types. In the SANAD I cohort of patients with generalized and unclassified epilepsies, valproate was superior to lamotrigine and topiramate for 12-month remission and treatment failure rates, respectively.14 However, valproate generally is avoided in women of childbearing age due its potential adverse effects during pregnancy. In focal epilepsies, lamotrigine was superior to carbamazepine, gabapentin, and topiramate with respect to treatment failure, and noninferior to carbamazepine for 12-month remission.15 In the SANAD II trial, levetiracetam was noninferior to valproate for incidence of adverse events in patients with generalized and unclassified epilepsies although was found to be neither more clinically effective nor more cost effective.16 For patients of childbearing potential with generalized and unclassified epilepsies, there is evidence to support the safe and effective use of levetiracetam.17 In focal epilepsies, lamotrigine was superior to levetiracetam and zonisamide with respect to treatment failures and adverse events and was noninferior to zonisamide for 12-month remission.18 In summary, levetiracetam and valproate (not to be used in women of childbearing potential) are considered appropriate first-line agents for generalized and unclassified epilepsies while lamotrigine is deemed an appropriate first-line agent for focal epilepsies (TABLE 119-28).

Drug level monitoring. It is standard practice to periodically monitor serum levels in patients taking first-generation ASMs such as phenytoin, carbamazepine, phenobarbital, and valproic acid because of their narrow therapeutic range and the potential for overdose or interaction with other medications or foods (eg, grapefruit juice may increase carbamazepine serum level by inhibiting CYP3A4, the enzyme that metabolizes the drug). Patients taking newer ASMs may not require regular serum level monitoring except during titration, with hepatic or renal dosing, when concomitantly used with estrogen-based oral contraceptives (eg, lamotrigine), before or during pregnancy, or when nonadherence is suspected.
Continue to: Can antiseizure treatment be stopped?
Can antiseizure treatment be stopped?
Current evidence favors continuing ASM therapy in patients whose seizures are under control, although the decision should be tailored to an individual’s circumstances. According to the 2021 American Academy of Neurology (AAN) guidelines, adults who have been seizure free for at least 2 years and discontinue ASMs are possibly still at higher risk for seizure recurrence in the long term (24-60 months), compared with those who continue treatment.29 On the other hand, for adults who have been seizure free for at least 12 months, ASM withdrawal may not increase their risk for status epilepticus, and there are insufficient data to support or refute an effect on mortality or quality of life with ASM withdrawal in this population. The decision to taper or maintain ASM therapy in seizure-free patients also should take into consideration other clinically relevant outcome measures such as the patient’s lifestyle and medication adverse effects. Therefore, this decision should be made after sufficient discussion with patients and their caregivers. (Information for patients can be found at: www.epilepsy.com/treatment/medicines/stopping-medication.)
For children, the AAN guideline panel recommends discussing with family the small risk (2%) for becoming medication resistant if seizures recur during or after ASM withdrawal. 29 For children who have been seizure free for 18 to 24 months, there is probably not a significant long-term (24-48 months) difference in seizure recurrence in those who taper ASMs vs those who do not. However, presence of epileptiform discharges on EEG before discontinuation of an ASM indicates increased risk for seizure recurrence. 29
Intractable (refractory) epilepsy
While most patients with epilepsy attain complete seizure control with appropriate drug therapy, approximately 30% continue to experience seizures (“drug-resistant” epilepsy, also termed intractable or refractory ). 30 In 2010, the ILAE defined drug-resistant epilepsy as “failure of adequate trials of two tolerated, appropriately chosen and used anti-epileptic drug schedules (whether as monotherapy or in combination) to achieve sustained seizure freedom” (defined as cessation of seizures for at least 3 times the longest pre-intervention inter-seizure interval or 12 months, whichever is longer). 21,31 It should be noted that drug withdrawal due to adverse effects is not counted as failure of that ASM. Recognition of drug-resistant epilepsy may prompt referral to an epileptologist who can consider rational combination drug therapy or surgical resection of the seizure focus, vagus nerve stimulation, electrical stimulation of the seizure focus, or deep brain (thalamic) stimulation.
Seizure triggers and mitigating factors
Epilepsy mostly affects patients during seizure episodes; however, the unpredictability of these events adds significantly to the burden of disease. There are no reliable methods for predicting seizure other than knowing of the several potential risks and recognizing and avoiding these triggers.
Noncompliance with antiseizure medications is a common seizure trigger affecting up to one-half of patients with epilepsy.32
Continue to: Medications
Medications may provoke seizures in susceptible individuals

Sleep deprivation is a potential seizure trigger in people with epilepsy based on observational studies, case reports, patient surveys, and EEG-based studies, although data from randomized controlled studies are limited.36 The standard best practice is to encourage appropriate sleep hygiene, which involves getting at least 7 hours of sleep per night.37
Alcohol is a GABAergic substance like benzodiazepines with antiseizure effects. However, it acts as a potential precipitant of seizures in cases of withdrawal or acute intoxication, or when it leads to sleep disruption or nonadherence to antiseizure medications. Therefore, advise patients with alcohol use disorder to slowly taper consumption (best done through a support program) and avoid sudden withdrawal. However, complete abstinence from alcohol use is not often recommended except in special circumstances (eg, a history of alcohol-related seizures). Several studies have demonstrated that modest alcohol use (1-2 drinks per occasion) does not increase seizure frequency or significantly alter serum concentrations of commonly used ASMs.38
Cannabis and other substances. The 2 main biologically active components of marijuana are delta-9-tetrahydrocannibinol (THC), the main psychoactive constituent, and cannabidiol (CBD). Animal and human studies have demonstrated anticonvulsant properties of THC and CBD. But THC, in high amounts, can result in adverse cognitive effects and worsening seizures.39 A purified 98% oil-based CBD extract (Epidiolex) has been approved as an adjunctive treatment for certain medically refractory epilepsy syndromes in children and young adults—ie, Dravet syndrome, Lennox-Gastaut syndrome, and tuberous sclerosis complex syndrome.40 There are no reliable data on the effect of recreational use of marijuana on seizure control. Other illicit substances such as cocaine may lower seizure threshold by their stimulatory and disruptive effects on sleep, diet, and healthy routines.
Special clinical cases
Pregnancy and epilepsy
Despite the potential adverse effects of ASMs on fetal health, the current global consensus is to continue treatment during pregnancy, given that the potential harm of convulsive seizures outweighs the potential risks associated with in-utero exposure to ASMs. There is not enough evidence to indicate significant harm to the fetus caused by focal, absence, or myoclonic seizures. Low-dose folic acid is used to minimize the risks of ASMs during pregnancy.
Continue to: As the fetus develops...
As the fetus develops, there are changes in volume of ASM distribution, renal clearance, protein binding, and hepatic metabolism, which require checking serum levels at regular intervals and making dosage adjustments.
The ongoing study evaluating Maternal Outcomes and Neurodevelopmental Effects of Antiepileptic Drugs (MONEAD)41 has led to multiple landmark studies guiding the choice of preferred ASMs during pregnancy in patients with epilepsy.42,43 This has culminated in today’s use of lamotrigine and levetiracetam as the 2 preferred agents (while avoiding valproate) in pregnant patients with epilepsy.44
Psychogenic nonepileptic seizures
A form of conversion disorder, psychogenic nonepileptic seizures (PNES) manifests as abnormal motor or behavioral events mimicking seizures but without associated epileptiform discharges on EEG. This is observed in 10% of patients seen in epilepsy clinics and even more often in those admitted to epilepsy monitoring units (25%-40%).45 Diagnosis of PNES requires EEG monitoring both for confirmation and for discernment from true epileptic seizures, in particular frontal lobe epilepsy that may clinically mimic PNES. PNES often is associated with underlying psychological tensions or comorbid conditions such as depression, anxiety, or traumatic life experiences. There is no treatment for PNES per se, and its management is focused on controlling any underlying psychological comorbidities that may not always be obvious. There is some evidence suggesting that these patients experience an innate inability to verbally express their emotions and instead subconsciously resort to psychosomatics to express them in a somatic dimension.46,47
Status epilepticus
Defined as prolonged seizures (> 5 min) or 2 consecutive seizures without regaining aware ness in between, status epilepticus (SE) is a potentially fatal condition. Subclinical nonconvulsive SE, especially in comatose patients, can be diagnosed only via EEG monitoring. Untreated SE may manifest as a diagnostic dilemma in unresponsive or critically ill patients and can increase the risk for mortality. 48
Febrile seizures
Febrile seizures affect 2% to 5% of children most often in the second year of life.49 The use of preventive antiseizure medication is not recommended; instead, the key is to investigate the underlying febrile illness. Lumbar puncture is indicated if there are signs and symptoms of meningitis (25% of children with bacterial meningitis present with seizures).49 Febrile seizures often are self-limited, but there is risk for SE in up to 15% of cases.50 If convulsive febrile seizures last longer than 5 minutes, initiate benzodiazepines followed by the standard protocol used for the management of SE.51
Continue to: Epilepsy as a spectrum disorder
Epilepsy as a spectrum disorder
The higher prevalence of comorbid cognitive and psychiatric conditions in patients with epilepsy, affecting about half of patients, 52 suggests that seizures may constitute only one aspect of a multifaceted disease that otherwise should be considered a spectrum disorder. Among such conditions are memory deficits, depression, and anxiety. Conversely, epilepsy is more common in patients with depression than in those without. 52
Social impact of epilepsy
Vehicle driving regulations. Patients with epilepsy are required to follow state law regarding driving restrictions. Different states have different rules and regulations about driving restrictions and reporting requirements (by patients or their physicians). Refer patients to the Department of Motor Vehicles (DMV) in their state of residence for up-to-date instructions.53 The Epilepsy Foundation (epilepsy.com) can serve as a resource for each state’s DMV website.
Employment assistance. Having epilepsy should not preclude patients from seeking employment and pursuing meaningful careers. The Americans with Disabilities Act (ADA) and the US Equal Employment Opportunity Commission (EEOC) forbid discrimination against qualified people with disabilities, including those with epilepsy, and require reasonable accommodations in the workplace (www.eeoc.gov/laws/guidance/epilepsy-workplace-and-ada).54
CORRESPONDENCE
Gholam K. Motamedi, MD, Department of Neurology, PHC 7, Georgetown University Hospital, 3800 Reservoir Road, NW, Washington, DC 20007; motamedi@georgetown.edu
1. Hauser WA, Annegers JF, Rocca WA. Descriptive epidemiology of epilepsy: contributions of population-based studies from Rochester, Minnesota. Mayo Clin Proc. 1996;71:576-586. doi: 10.4065/71.6.576
2. Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55:475-482. doi: 10.1111/epi.12550.
3. Hauser WA, Beghi E. First seizure definitions and worldwide incidence and mortality. Epilepsia. 2008;49:8-12. doi: 10.1111/j.1528-1167.2008.01443.x
4. Berg AT, Shinnar S. The risk of seizure recurrence following a first unprovoked seizure: a quantitative review. Neurology. 1991;41:965-972. doi: 10.1212/wnl.41.7.965
5. Ropper AH. Transient global amnesia. N Engl J Med. 2023;388:635-540. doi: 10.1056/NEJMra2213867
6. Bouma HK, Labos C, Gore GC, et al. The diagnostic accuracy of routine electroencephalography after a first unprovoked seizure. Eur J Neurol. 2016;23:455-463. doi: 10.1111/ene.12739
7. Narayanan JT, Labar DR, Schaul N. Latency to first spike in the EEG of epilepsy patients. Seizure. 2008;17:34-41. doi: 10.1016/j.seizure.2007.06.003
8. Salmenpera TM, Duncan JS. Imaging in epilepsy. J Neurol Neurosurg Psychiatry. 2005;76:iii2-iii10. doi: 10.1136/jnnp.2005.075135
9. Jackson GD, Berkovic SF, Tress , et al Hippocampal sclerosis can be reliably detected by magnetic resonance imaging. Neurology. 1990;40:1869-1875. doi: 10.1212/wnl.40.12.1869
10. Bonnett LJ, Kim, L, Johnson A, et al. Risk of seizure recurrence in people with single seizures and early epilepsy - model development and external validation. Seizure. 2022;94:26-32. doi: 10.1016/j.seizure.2021.11.007
11. Krumholz A, Wiebe S, Gronseth GS, et al. Evidence-based guideline: management of an unprovoked first seizure in adults: Report of the Guideline Development Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2015;84:1705-1713. doi: 10.1212/WNL.0000000000001487
12. Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE Commission for Classification and terminology. Epilepsia. 2017;58:522-530. doi: 10.1111/epi.13670
13. Berg AT, Berkovic SF, Brodie MJ, et al. Revised terminology and concepts for organization of seizures and epilepsy: report of the ILAE Commission on Classification and Terminology, 2005-2009. Epilepsia. 2010;51:676-685. doi: 10.1111/j.1528-1167.2010.02522.x
14. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of valproate, lamotrigine, or topiramate for generalized and unclassifiable epilepsy: an unblinded randomized controlled trial. Lancet. 2007;369:1016-1026. doi: 10.1016/S0140-6736(07)60461-9
15. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy: an unblinded randomized controlled trial. Lancet 2007;369:1000-1015. doi: 10.1016/S0140-6736(07)60460-7
16. Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of valproate versus levetiracetam for newly diagnosed generalized and unclassified epilepsy: an open-label, non-inferiority, multicentre, phase 4, randomized controlled trial. Lancet. 2021;397:1375-1386. doi: 10.1016/S0140-6736(21)00246-4
17. Mawhinney E, Craig J, Morrow J. Levetiracetam in pregnancy: results from the UK and Ireland epilepsy and pregnancy registers. Neurology. 2013;80:400-405.
18. Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of levetiracetam, zonisamide, or lamotrigine for newly diagnosed focal epilepsy: an open-label, non-inferiority, multicentre, phase 4, randomized controlled trial. Lancet. 2021;397:1363-1374. doi: 10.1016/S0140-6736(21)00247-6
19. Smith PE. Initial management of seizure in adults. N Engl J Med. 2021;385:251-263. doi: 10.1056/NEJMcp2024526
20. Depakene (valproic acid). Package insert. Abbott Laboratories; 2011. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2011/018081s046_18082s031lbl.pdf
21. Greenberg RG, Melloni C, Wu H, et al. Therapeutic index estimation of antiepileptic drugs: a systematic literature review approach. Clin Neuropharmacol. 2016;39:232-240. doi: 10.1097/WNF.0000000000000172
22. Lamictal (lamotrigine). Package insert. GlaxoSmithKline; 2009. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2009/020241s037s038,020764s030s031lbl.pdf
23. LaRoche SM, Helmers SL. The new antiepileptic drugs: scientific review. JAMA. 2004;291:605-614. doi: 10.1001/jama.291.5.605
24. Topamax (topiramate). Package insert. Janssen Pharmaceuticals, Inc. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2012/020844s041lbl.pdf
25. Keppra (levetiracetam). Package insert. UCB, Inc.; 2009. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2009/021035s078s080%2C021505s021s024lbl.pdf
26. Carbatrol (carbamazepine). Package insert. Shire US Inc; 2013. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2013/020712s032s035lbl.pdf
27.Neurontin (gabapentin). Package insert. Pfizer; 2017. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2017/020235s064_020882s047_021129s046lbl.pdf
28.Zonegran (zonisamide). Package insert. Eisai Inc; 2006. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2006/020789s019lbl.pdf
29.Gloss D, Paragon K, Pack A, et al. Antiseizure medication withdrawal in seizure-free patients: practice advisory update. Report of the AAN Guideline Subcommittee. Neurology. 2021;97:1072-1081. doi: 10.1212/WNL.0000000000012944
30.Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000:342:314-319. doi: 10.1056/NEJM200002033420503
31.Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia. 2010;51:1069-1077. doi: 10.1111/j.1528-1167.2009.02397.x
Compliance during treatment of epilepsy. Epilepsia 1988;29(suppl 2):S79-S84.
33.utter R, Rüegg S, Tschudin-Sutter S. Seizures as adverse events of antibiotic drugs: a systematic review. Neurology. 2015;13;85:1332-1341. doi: 10.1212/WNL.0000000000002023
34.Singh G, Rees JH, Sander JW. Seizures and epilepsy in oncological practice: causes, course, mechanisms and treatment. JNNP. 2007;78:342-349. doi: 10.1136/jnnp.2006.106211
35.Pisani F, Oteri G, Costa C., et al. Effects of psychotropic drugs on seizure threshold. Drug Safety. 2002;25:91-110.
36.Rossi KC, Joe J, Makhjia M, et al. Insufficient sleep, electroencephalogram activation, and seizure risk: re-evaluating the evidence. Ann Neurol. 2020;86:798-806. doi: 10.1002/ana.25710
37.Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843-844. doi: 10.5665/sleep.4716
38.Höppener RJ, Kuyer A, van der Lugt PJ. Epilepsy and alcohol: the influence of social alcohol intake on seizures and treatment in epilepsy. Epilepsia. 1983;24:459-471. doi: 10.1111/j.1528-1157.1983.tb04917.x
39.Keeler MH, Reifler CB. Grand mal convulsions subsequent to marijuana use. Case report. Dis Nerv Syst. 1967:28:474-475.
40.Epidiolex (cannabidiol). Package insert. Greenwich Biosciences Inc; 2018. Accessed September 27, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf
41.ClinicalTrials.gov. Maternal Outcomes and Neurodevelopmental Effects of Antiseizure Drugs (MONEAD). Accessed September 24, 2023. https://classic.clinicaltrials.gov/ct2/show/NCT01730170
42.Meador KJ, Baker GA, Finnell RH, et al. In utero antiepileptic drug exposure: fetal death and malformations. Neurology. 2006;67:407-412. doi: 10.1212/01.wnl.0000227919.81208.b2
43.Meador K, Reynolds MW, Crean S. Pregnancy outcomes in women with epilepsy: a systematic review and meta-analysis of published pregnancy registries and cohorts. Epilepsy Res. 2008;81:1-13. doi:10.1016/j.eplepsyres.2008.04.022
44.Marxer CA, Rüegg S, Rauch A review of the evidence on the risk of congenital malformations and neurodevelopmental disorders in association with antiseizure medications during pregnancy. Expert Opin Drug Saf. 2021;20:1487-1499. doi: 10.1080/14740338.2021.1943355
Asadi-Pooya AA, Sperling MR. Epidemiology of psychogenic nonepileptic seizures. Epilepsy Behav. 2015;46:60-65. doi: 10.1016/j.yebeh.2015.03.015
Evaluation and treatment of psychogenic nonepileptic seizures. Neurol Clin. 2022;40:799-820. doi: 10.1016/j.ncl.2022.03.017
47.Motamedi GK. Psychogenic nonepileptic seizures: a disconnect between body and mind. Epilepsy Behav. 2018;78:293-294. doi: 10.1016/j.yebeh.2017.10.016
, Nonconvulsive status epilepticus. Emerg Med Clin North Am. 2011;29:65-72. doi: 10.1016/j.emc.2010.08.006
doi: 10.1542/peds.2010-3318
Drug management for acute tonic-clonic convulsions including convulsive status epilepticus in children. Cochrane Database Sys Rev. 2018;1(1):CD001905. doi: 10.1002/14651858.CD001905.pub3
52.Jensen FE. Epilepsy as a spectrum disorder: implications from novel clinical and basic neuroscience. Epilepsia. 2011;52(suppl 1):1-6. doi: 10.1111/j.1528-1167.2010.02904.x
53.Kass JS, Rose RV. Driving and epilepsy: ethical, legal, and health care policy challenges. Continuum (Minneap Minn). 2019;25:537-542. doi: 10.1212/CON.0000000000000714
54.Troxell J. Epilepsy and employment: the Americans with Disabilities Act and its protections against employment discrimination. Med Law. 1997;16:375-384.
1. Hauser WA, Annegers JF, Rocca WA. Descriptive epidemiology of epilepsy: contributions of population-based studies from Rochester, Minnesota. Mayo Clin Proc. 1996;71:576-586. doi: 10.4065/71.6.576
2. Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia. 2014;55:475-482. doi: 10.1111/epi.12550.
3. Hauser WA, Beghi E. First seizure definitions and worldwide incidence and mortality. Epilepsia. 2008;49:8-12. doi: 10.1111/j.1528-1167.2008.01443.x
4. Berg AT, Shinnar S. The risk of seizure recurrence following a first unprovoked seizure: a quantitative review. Neurology. 1991;41:965-972. doi: 10.1212/wnl.41.7.965
5. Ropper AH. Transient global amnesia. N Engl J Med. 2023;388:635-540. doi: 10.1056/NEJMra2213867
6. Bouma HK, Labos C, Gore GC, et al. The diagnostic accuracy of routine electroencephalography after a first unprovoked seizure. Eur J Neurol. 2016;23:455-463. doi: 10.1111/ene.12739
7. Narayanan JT, Labar DR, Schaul N. Latency to first spike in the EEG of epilepsy patients. Seizure. 2008;17:34-41. doi: 10.1016/j.seizure.2007.06.003
8. Salmenpera TM, Duncan JS. Imaging in epilepsy. J Neurol Neurosurg Psychiatry. 2005;76:iii2-iii10. doi: 10.1136/jnnp.2005.075135
9. Jackson GD, Berkovic SF, Tress , et al Hippocampal sclerosis can be reliably detected by magnetic resonance imaging. Neurology. 1990;40:1869-1875. doi: 10.1212/wnl.40.12.1869
10. Bonnett LJ, Kim, L, Johnson A, et al. Risk of seizure recurrence in people with single seizures and early epilepsy - model development and external validation. Seizure. 2022;94:26-32. doi: 10.1016/j.seizure.2021.11.007
11. Krumholz A, Wiebe S, Gronseth GS, et al. Evidence-based guideline: management of an unprovoked first seizure in adults: Report of the Guideline Development Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2015;84:1705-1713. doi: 10.1212/WNL.0000000000001487
12. Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy: position paper of the ILAE Commission for Classification and terminology. Epilepsia. 2017;58:522-530. doi: 10.1111/epi.13670
13. Berg AT, Berkovic SF, Brodie MJ, et al. Revised terminology and concepts for organization of seizures and epilepsy: report of the ILAE Commission on Classification and Terminology, 2005-2009. Epilepsia. 2010;51:676-685. doi: 10.1111/j.1528-1167.2010.02522.x
14. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of valproate, lamotrigine, or topiramate for generalized and unclassifiable epilepsy: an unblinded randomized controlled trial. Lancet. 2007;369:1016-1026. doi: 10.1016/S0140-6736(07)60461-9
15. Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy: an unblinded randomized controlled trial. Lancet 2007;369:1000-1015. doi: 10.1016/S0140-6736(07)60460-7
16. Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of valproate versus levetiracetam for newly diagnosed generalized and unclassified epilepsy: an open-label, non-inferiority, multicentre, phase 4, randomized controlled trial. Lancet. 2021;397:1375-1386. doi: 10.1016/S0140-6736(21)00246-4
17. Mawhinney E, Craig J, Morrow J. Levetiracetam in pregnancy: results from the UK and Ireland epilepsy and pregnancy registers. Neurology. 2013;80:400-405.
18. Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of levetiracetam, zonisamide, or lamotrigine for newly diagnosed focal epilepsy: an open-label, non-inferiority, multicentre, phase 4, randomized controlled trial. Lancet. 2021;397:1363-1374. doi: 10.1016/S0140-6736(21)00247-6
19. Smith PE. Initial management of seizure in adults. N Engl J Med. 2021;385:251-263. doi: 10.1056/NEJMcp2024526
20. Depakene (valproic acid). Package insert. Abbott Laboratories; 2011. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2011/018081s046_18082s031lbl.pdf
21. Greenberg RG, Melloni C, Wu H, et al. Therapeutic index estimation of antiepileptic drugs: a systematic literature review approach. Clin Neuropharmacol. 2016;39:232-240. doi: 10.1097/WNF.0000000000000172
22. Lamictal (lamotrigine). Package insert. GlaxoSmithKline; 2009. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2009/020241s037s038,020764s030s031lbl.pdf
23. LaRoche SM, Helmers SL. The new antiepileptic drugs: scientific review. JAMA. 2004;291:605-614. doi: 10.1001/jama.291.5.605
24. Topamax (topiramate). Package insert. Janssen Pharmaceuticals, Inc. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2012/020844s041lbl.pdf
25. Keppra (levetiracetam). Package insert. UCB, Inc.; 2009. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2009/021035s078s080%2C021505s021s024lbl.pdf
26. Carbatrol (carbamazepine). Package insert. Shire US Inc; 2013. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2013/020712s032s035lbl.pdf
27.Neurontin (gabapentin). Package insert. Pfizer; 2017. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2017/020235s064_020882s047_021129s046lbl.pdf
28.Zonegran (zonisamide). Package insert. Eisai Inc; 2006. Accessed October 6, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2006/020789s019lbl.pdf
29.Gloss D, Paragon K, Pack A, et al. Antiseizure medication withdrawal in seizure-free patients: practice advisory update. Report of the AAN Guideline Subcommittee. Neurology. 2021;97:1072-1081. doi: 10.1212/WNL.0000000000012944
30.Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000:342:314-319. doi: 10.1056/NEJM200002033420503
31.Kwan P, Arzimanoglou A, Berg AT, et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia. 2010;51:1069-1077. doi: 10.1111/j.1528-1167.2009.02397.x
Compliance during treatment of epilepsy. Epilepsia 1988;29(suppl 2):S79-S84.
33.utter R, Rüegg S, Tschudin-Sutter S. Seizures as adverse events of antibiotic drugs: a systematic review. Neurology. 2015;13;85:1332-1341. doi: 10.1212/WNL.0000000000002023
34.Singh G, Rees JH, Sander JW. Seizures and epilepsy in oncological practice: causes, course, mechanisms and treatment. JNNP. 2007;78:342-349. doi: 10.1136/jnnp.2006.106211
35.Pisani F, Oteri G, Costa C., et al. Effects of psychotropic drugs on seizure threshold. Drug Safety. 2002;25:91-110.
36.Rossi KC, Joe J, Makhjia M, et al. Insufficient sleep, electroencephalogram activation, and seizure risk: re-evaluating the evidence. Ann Neurol. 2020;86:798-806. doi: 10.1002/ana.25710
37.Watson NF, Badr MS, Belenky G, et al. Recommended amount of sleep for a healthy adult: a Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. Sleep. 2015;38:843-844. doi: 10.5665/sleep.4716
38.Höppener RJ, Kuyer A, van der Lugt PJ. Epilepsy and alcohol: the influence of social alcohol intake on seizures and treatment in epilepsy. Epilepsia. 1983;24:459-471. doi: 10.1111/j.1528-1157.1983.tb04917.x
39.Keeler MH, Reifler CB. Grand mal convulsions subsequent to marijuana use. Case report. Dis Nerv Syst. 1967:28:474-475.
40.Epidiolex (cannabidiol). Package insert. Greenwich Biosciences Inc; 2018. Accessed September 27, 2023. www.accessdata.fda.gov/drugsatfda_docs/label/2018/210365lbl.pdf
41.ClinicalTrials.gov. Maternal Outcomes and Neurodevelopmental Effects of Antiseizure Drugs (MONEAD). Accessed September 24, 2023. https://classic.clinicaltrials.gov/ct2/show/NCT01730170
42.Meador KJ, Baker GA, Finnell RH, et al. In utero antiepileptic drug exposure: fetal death and malformations. Neurology. 2006;67:407-412. doi: 10.1212/01.wnl.0000227919.81208.b2
43.Meador K, Reynolds MW, Crean S. Pregnancy outcomes in women with epilepsy: a systematic review and meta-analysis of published pregnancy registries and cohorts. Epilepsy Res. 2008;81:1-13. doi:10.1016/j.eplepsyres.2008.04.022
44.Marxer CA, Rüegg S, Rauch A review of the evidence on the risk of congenital malformations and neurodevelopmental disorders in association with antiseizure medications during pregnancy. Expert Opin Drug Saf. 2021;20:1487-1499. doi: 10.1080/14740338.2021.1943355
Asadi-Pooya AA, Sperling MR. Epidemiology of psychogenic nonepileptic seizures. Epilepsy Behav. 2015;46:60-65. doi: 10.1016/j.yebeh.2015.03.015
Evaluation and treatment of psychogenic nonepileptic seizures. Neurol Clin. 2022;40:799-820. doi: 10.1016/j.ncl.2022.03.017
47.Motamedi GK. Psychogenic nonepileptic seizures: a disconnect between body and mind. Epilepsy Behav. 2018;78:293-294. doi: 10.1016/j.yebeh.2017.10.016
, Nonconvulsive status epilepticus. Emerg Med Clin North Am. 2011;29:65-72. doi: 10.1016/j.emc.2010.08.006
doi: 10.1542/peds.2010-3318
Drug management for acute tonic-clonic convulsions including convulsive status epilepticus in children. Cochrane Database Sys Rev. 2018;1(1):CD001905. doi: 10.1002/14651858.CD001905.pub3
52.Jensen FE. Epilepsy as a spectrum disorder: implications from novel clinical and basic neuroscience. Epilepsia. 2011;52(suppl 1):1-6. doi: 10.1111/j.1528-1167.2010.02904.x
53.Kass JS, Rose RV. Driving and epilepsy: ethical, legal, and health care policy challenges. Continuum (Minneap Minn). 2019;25:537-542. doi: 10.1212/CON.0000000000000714
54.Troxell J. Epilepsy and employment: the Americans with Disabilities Act and its protections against employment discrimination. Med Law. 1997;16:375-384.
PRACTICE RECOMMENDATIONS
› Consider treating a first-time seizure if electroencephalography shows particular epileptiform activity, if the neurologic exam or computerized tomography or magnetic resonance imaging results are abnormal, if the seizure is focal or nocturnal, or if there is a family history of seizures. A
› Consider valproate (except for women of childbearing age) and levetiracetam as first-line agents for generalized or unclassified epilepsy, and lamotrigine for focal epilepsies. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series