User login
For MD-IQ use only
Dressing in blue
On the first Friday in March, it has become an annual tradition to dress in blue to promote colorectal cancer awareness. Twitter feeds are filled with photos of members of our gastroenterology community (sometimes entire endoscopy units!) swathed in various shades of blue. This tradition was started in the mid-2000’s by a patient diagnosed with early-onset colorectal cancer who planned a fund raiser at her daughter’s elementary school where students were encouraged to wear a blue outfit and make a $1 donation to support awareness of this deadly but preventable cancer. What was once a local effort has now grown into a national phenomenon, and a powerful opportunity for the medical community to educate patients, friends, and family regarding risk factors for colorectal cancer and the importance of timely and effective screening. But while raising awareness is vital, it is only an initial step in the complex process of optimizing delivery of screening services and improving cancer outcomes through prevention and early detection.
In this month’s issue of GIHN, we report on a study from Cancer demonstrating the effectiveness of Spanish-speaking patient navigators in boosting colorectal cancer screening rates among Hispanic patients. We also highlight a quality improvement initiative at a large academic medical center demonstrating the impact of an electronic “primer” message delivered through the patient portal on screening completion rates in a mailed fecal immunochemical test outreach program. Finally, in this month’s Practice Management Toolbox column, Dr. Brill and Dr. Lieberman advise us on how to prepare for upcoming coverage changes impacting screening colonoscopy – a result of AGA’s tireless efforts to eliminate financial barriers impeding access to colorectal cancer screening.
As always, thank you for being a dedicated reader and please stay safe out there. Better days are ahead.
Megan A. Adams, MD, JD, MSc
Editor in Chief
On the first Friday in March, it has become an annual tradition to dress in blue to promote colorectal cancer awareness. Twitter feeds are filled with photos of members of our gastroenterology community (sometimes entire endoscopy units!) swathed in various shades of blue. This tradition was started in the mid-2000’s by a patient diagnosed with early-onset colorectal cancer who planned a fund raiser at her daughter’s elementary school where students were encouraged to wear a blue outfit and make a $1 donation to support awareness of this deadly but preventable cancer. What was once a local effort has now grown into a national phenomenon, and a powerful opportunity for the medical community to educate patients, friends, and family regarding risk factors for colorectal cancer and the importance of timely and effective screening. But while raising awareness is vital, it is only an initial step in the complex process of optimizing delivery of screening services and improving cancer outcomes through prevention and early detection.
In this month’s issue of GIHN, we report on a study from Cancer demonstrating the effectiveness of Spanish-speaking patient navigators in boosting colorectal cancer screening rates among Hispanic patients. We also highlight a quality improvement initiative at a large academic medical center demonstrating the impact of an electronic “primer” message delivered through the patient portal on screening completion rates in a mailed fecal immunochemical test outreach program. Finally, in this month’s Practice Management Toolbox column, Dr. Brill and Dr. Lieberman advise us on how to prepare for upcoming coverage changes impacting screening colonoscopy – a result of AGA’s tireless efforts to eliminate financial barriers impeding access to colorectal cancer screening.
As always, thank you for being a dedicated reader and please stay safe out there. Better days are ahead.
Megan A. Adams, MD, JD, MSc
Editor in Chief
On the first Friday in March, it has become an annual tradition to dress in blue to promote colorectal cancer awareness. Twitter feeds are filled with photos of members of our gastroenterology community (sometimes entire endoscopy units!) swathed in various shades of blue. This tradition was started in the mid-2000’s by a patient diagnosed with early-onset colorectal cancer who planned a fund raiser at her daughter’s elementary school where students were encouraged to wear a blue outfit and make a $1 donation to support awareness of this deadly but preventable cancer. What was once a local effort has now grown into a national phenomenon, and a powerful opportunity for the medical community to educate patients, friends, and family regarding risk factors for colorectal cancer and the importance of timely and effective screening. But while raising awareness is vital, it is only an initial step in the complex process of optimizing delivery of screening services and improving cancer outcomes through prevention and early detection.
In this month’s issue of GIHN, we report on a study from Cancer demonstrating the effectiveness of Spanish-speaking patient navigators in boosting colorectal cancer screening rates among Hispanic patients. We also highlight a quality improvement initiative at a large academic medical center demonstrating the impact of an electronic “primer” message delivered through the patient portal on screening completion rates in a mailed fecal immunochemical test outreach program. Finally, in this month’s Practice Management Toolbox column, Dr. Brill and Dr. Lieberman advise us on how to prepare for upcoming coverage changes impacting screening colonoscopy – a result of AGA’s tireless efforts to eliminate financial barriers impeding access to colorectal cancer screening.
As always, thank you for being a dedicated reader and please stay safe out there. Better days are ahead.
Megan A. Adams, MD, JD, MSc
Editor in Chief
Autism spectrum disorder: Keys to early detection and accurate diagnosis
FIRST OF 2 PARTS
Autism spectrum disorder (ASD) is a complex, heterogenous neurodevelopmental disorder with genetic and environmental underpinnings, and an onset early in life.1-9 It affects social communication, cognition, and sensory-motor domains, and manifests as deficits in social reciprocity, repetitive behavior, restricted range of interests, and sensory sensitivities.6,10-14 In recent years, the prevalence of ASD has been increasing.3,6,10 A large percentage of individuals with ASD experience significant social deficits in adulthood,10 which often leads to isolation, depressive symptoms, and poor occupational and relationship functioning.15,16 Interventions in early childhood can result in significant and lasting changes in outcome and in functioning of individuals with ASD.
This article provides an update on various aspects of ASD diagnosis, with the goal of equipping clinicians with knowledge to help make an accurate ASD diagnosis at an early stage. Part 1 focuses on early detection and diagnosis, while Part 2 will describe treatment strategies.
Benefits of early detection
Substantial research has established that early intervention confers substantial benefits for outcomes among children with ASD.2,3,5,6,9,13,14,16-22 Earlier age of intervention correlates with greater developmental gain and symptom reduction.21,23 The atypical neural development responsible for ASD likely occurs much earlier than the behavioral manifestations of this disorder, which implies that there is a crucial period to intervene before behavioral features emerge.1 This necessitates early recognition of ASD,9,17 and the need for further research to find novel ways to detect ASD earlier.
In the United States, children with ASD are diagnosed with the disorder on average between age 3 and 4 years.6,24 However, evidence suggests there may be a prodromal phase for ASD during the first several months of life, wherein infants and toddlers exhibit developmentally inadequate communication and social skills and/or unusual behaviors.18 Behavioral signs suggestive of ASD may be evident as early as infancy, and commonly earlier than age 18 months.1,17,19 Problems with sleeping and eating may be evident in early childhood.19 Deficits in joint attention may be evident as early as age 6 months to 8 months. Research suggests that a diagnosis of ASD by trained, expert professionals is likely to be accurate at the age of 2, and even as early as 18 months.6,24
In a prospective study, Anderson et al25 found that 9% of children who were diagnosed with ASD at age 2 no longer met the diagnostic criteria for ASD by adulthood.6 Those who no longer met ASD criteria were more likely to have received early intervention, had a verbal IQ ≥70, and had experienced a larger decrease in repetitive behaviors between ages 2 and 3, compared with other youth in this study who had a verbal IQ ≥70. One of the limitations of this study was a small sample size (85 participants); larger, randomized studies are needed to replicate these findings.25
Continue to: Characteristics of ASD...
Characteristics of ASD
Table 16,8,10,13,15,26-29 outlines various characteristics of ASD, which may manifest in varying degrees among children with the condition.
Speech/language. Speech helps to facilitate bonding between parents and an infant by offering a soothing, pleasurable, and reinforcing experience.30 More than 50% of children with ASD have language delays or deficits that persist throughout adulthood.13 The extent of these language deficits varies; in general, the more severe the speech/language deficits, the more severe the long-term symptoms.13 Language deficits in young children with ASD tend to be of both the expressive and receptive type, with onset in infancy, which suggests that neural processes predate the emergence of behavioral symptoms of ASD, and also that early language deficits/delays could be a marker for or indicator of future risk of ASD.13 Individuals with ASD also have been noted to have limitations in orienting or attending to human voices.13,30
Facial recognition. Evidence has linked ASD with deficits in facial recognition that emerge in the first few months of life.2 Earlier studies have found that lack of attention to others’ faces was the strongest distinguishing factor between 1-year-olds with ASD and typically developing 1-year-olds.2,31 A recent study that used EEG to compare facial emotion recognition in boys with ASD vs typically developing boys found that boys with ASD exhibited significantly lower sensitivity to angry and fearful faces.27
Other features. A 2020 study (N = 37) found that compared with typically developing children, those with ASD show less “interactional synchrony’’ (a dynamic process in which the timing of children and caregivers’ behaviors [specifically, vocalizations and movements] become mutually coordinated) with both familiar and unfamiliar adults.32 These researchers concluded that impairment in interactional synchrony may be linked to social communication deficits in ASD.32
A recent study (N = 98) evaluated “sluggish cognitive tempo” in 3 groups of children: children with attention-deficit/hyperactivity disorder (ADHD), children with ASD, and children with both ADHD and ASD.33 It found that children with ASD exhibited sluggish cognitive tempo at levels similar to those of the other 2 groups, and indicated that sluggish cognitive tempo may be linked with “social and global impairment above and beyond” the impairment associated with ASD.
Understanding early aberrations in neurobiologic processes in ASD can help develop biomarkers for early recognition of ASD, as well as guide the development of targeted interventions and treatments (Box1-3,7-9,12,13,30,35-39).
Box
Compared with individuals who do not have autism spectrum disorder (ASD), individuals with ASD exhibit anatomical differences in the brain that can be seen on MRI.9,35 Brain regions affected in ASD include the frontal gyrus, temporal gyrus, cingulate gyrus, postcentral gyrus, precuneus, caudate, and hippocampus.9 Some studies have found anomalous structural neural characteristics in infants, such as in the uncinate fasciculus, that correlated with later joint attention challenges, while others have found aberrations in the corpus callosum(responsible for transfer of procedural learning between the hemispheres, and oculomotor response)and internal capsule (responsible for sensorimotor function, as well as other functions) in children with ASD.12
Widespread white matter anomalies have been noted in ASD.12,35,36 In a 2-year longitudinal study that used diffusion tensor imaging, Li et al35 found that preschool children with ASD experience overgrowth of the uncinate fasciculus, which is one of the brain regions implicated in socioemotional processing, and concluded that this overgrowth correlated with ASD severity.35 Andrews et al37 used diffusion-weighted MRI to examine white matter in 127 preschool children. They found that compared with typically developing children, children with ASD exhibited altered white matter microstructure.37
Research suggests that developing representations of the reward value of social stimuli may be challenging for children with ASD.2 Abrams et al30 used resting-state functional brain MRI to evaluate children with typical development and children with highfunctioning, “verbally fluent” ASD. They found that the children with ASD exhibited lower functional connectivity between voice-specific left hemisphere posterior superior temporal sulcus and areas representing the reward circuitry.30 This study also found that children with ASD had underconnectivity between the right hemisphere posterior superior temporal sulcus (which deals with speech prosody) and areas known for emotion-linked associative learning, the orbitofrontal cortex and amygdala.30 These findings are thought to align with the social motivation theory of ASD.13,30,38
The extent of underconnectivity between these systems was found to determine the severity of communication challenges in high-functioning children with ASD.30 One MRI study observed lower gray matter volume in the voice-selective bilateral superior temporal sulcus in children age approximately 9 to 11 years with ASD.39
Neural systems responsible for facial recognition (particularly the right fusiform gyrus and other brain areas) have been shown to exist or begin “very early in life,” which suggests that impaired face recognition may be an early marker of ASD.2 In addition to problems with visual scanning, preferential attention to (and visual sensitivity to) biological motion is a forerunner for the development of social interactions in infants, specifically in regard to being able to detect and recognize emotion, which is considered vital for attachment.7,8 Impaired biological motion perception has been found in very young children with ASD.7,8 This presents an important avenue/potential biomarker for further research to better understand neurobiologic processes underlying atypical development at an earlier age.3,8
Early neural biomarkers for ASD
Nonlinear EEG values may serve as an early neurobiomarker for detecting ASD in young children.1 Because it is relatively inexpensive and convenient, EEG may be highly useful for detecting ASD.1 A study that compared EEG results of 99 infants who had siblings with ASD and 89 low-risk controls from age 3 months to 36 months found that nonlinear EEG measurements predicted with high accuracy later diagnosis of ASD, and were strongly correlated with later Autism Diagnostic Observation Schedule scores.1
Continue to: A complex differential diagnosis...
A complex differential diagnosis
The differential diagnosis of ASD warrants careful attention and consideration to rule out other developmental and psychiatric conditions.
Intellectual disability (ID). DSM-5 diagnostic criteria for ASD necessitate that disturbances are not better explained by ID or global developmental delay and that deficits should exceed impairment consistent with the level of intellectual disability.28 Still, ASD is often overdiagnosed in children with ID.28 Research suggests phenotypic and genetic overlap between ID and ASD.28 Social functioning is often impaired in patients with ID; the greater the severity of ID, the greater the degree of social deficits.28 In approximately 30% of cases, ASD and ID are comorbid.6 This overlap and comorbidity can pose a challenge, particularly due to the inherent complexities involved in assessment and differentiation.28 When ID is present in ASD, there is a greater degree of social-communication deficits.6 It may be difficult to assess for ASD symptoms in children with severe ID.28 Although there is no minimum age or developmental level below which ASD should not be diagnosed, some studies have started to use minimum criteria for diagnosis, such as a nonverbal mental age of 18 months.28,40 Commonly used tests for ASD have much lower specificity when used for children with nonverbal age <15 months.28 It would make sense, then, that the presence of ID might significantly affect the results of these diagnostic tests.28
Other conditions that need to be ruled out include language disorders, hearing loss, rare genetic neurodevelopmental disorders (eg, Fragile X syndrome,3 Rett syndrome6), childhood-onset schizophrenia, obsessive-compulsive disorder, attachment disorders, and other conditions.18 ASD may be overdiagnosed in children with genetic disorders such as Angelman syndrome.41 In a systematic review, Moss and Howlin42 recommended caution when evaluating ASD-like behavioral symptoms in children with genetic syndromes and severe ID. On the other hand, some research has observed that individuals with Fragile X syndrome may exhibit symptoms that meet criteria for ASD.6,43 McDuffie et al43 used the Autism Diagnostic Interview-Revised (ADI-R) to compare boys with Fragile X syndrome who also met criteria for ASD with boys with nonsyndromic ASD. Those in the former group had lesser impairment in social smiling, offering, showing, and nonverbal gestures, but had more complex mannerisms, compared with boys in the latter group.43
Milder manifestations of ASD may be more challenging to diagnose,1 particularly in children age <3 and those with above-average cognition.6 Generally, in the case of a patient with ASD, parents find that the child did not have a period of typical development, or unusual behaviors were evident early on.17
ASD can be comorbid with ADHD. The presence of ADHD may mask or delay the diagnosis of ASD in children.6 In children with both ASD and ADHD, studies have found greater reduction in social and adaptive functioning compared with children with ADHD alone.44
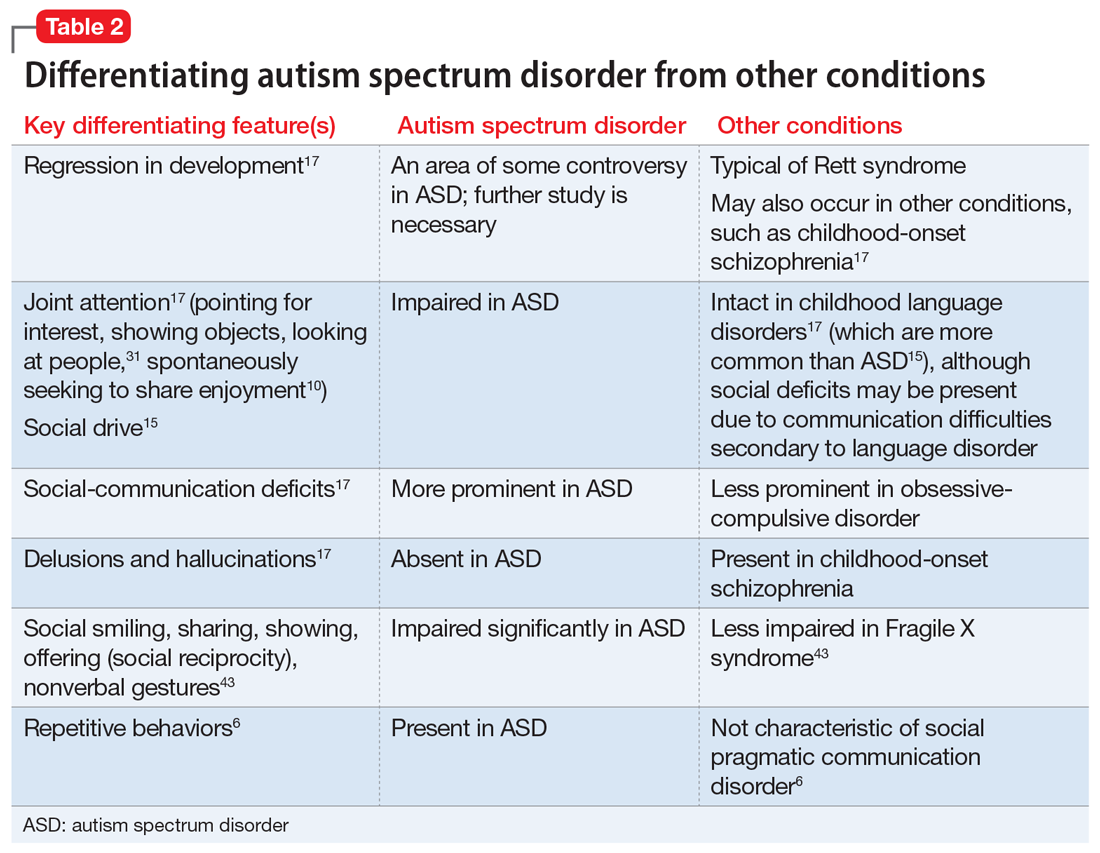
Table 26,10,15,17,31,43 highlights some of the features that can be used to distinguish ASD from other conditions.
Continue to: Screening and diagnosis...
Screening and diagnosis
A medical workup is the first step to rule out other potential conditions that could be masquerading as ASD.17 Obtain a comprehensive history from parents/caregivers, particularly regarding social, behavioral, movement, sensory, and developmental aspects. In addition, audiologic testing is an essential step. Consider genetic testing, particularly if any dysmorphic features and/or ID are present, both of which confer additional risk for a genetic syndrome.6 A physical exam to detect any neurologic anomalies, organ dysfunction, and body dysmorphic features should be conducted.6
The Modified Checklist for Autism in Toddlers–Revised (MCHAT-R) is a commonly used, validated parental screening survey for ASD.5,6 Research has shown that this survey has <50% specificity.5A recent American Academy of Pediatrics Clinical Report recommended universal screening for ASD at pediatric visits at age 18 months and at 24 months, in addition to developmental screening for all children at routine pediatric visits at age 9, 18, and 30 months.6,19
Screening tools such as the Modified Checklist for Autism in Toddlers with Follow-Up (M-CHAT/F) can be integrated into routine primary health care. In a large (N = 25,999) study, Guthrie et al45 used M-CHAT/F to conduct universal, primary care–based screening in young children. They found that the positive predictive value of M-CHAT/F was lower among girls, children of color, and those from lower-income households. There is a need for development of screening tools with higher accuracy and sensitivity for identifying young children with ASD regardless of their ethnic or socioeconomic background, and also for children older than 30 months.5,6,45
Definitive diagnosis of ASD is ideally done by a multidisciplinary team46 using established gold standard measures such as the ADOS (Autism Diagnostic Observation Schedule) and ADI-R.47 Such multidisciplinary teams usually include a child psychiatrist, child psychologist, speech therapist, occupational therapist, school educator, and developmental pediatrician. However, because there are long wait times to receive this type of diagnosis in the United States,6 in the interest of not missing the critical window of early intervention, physicians who suspect a patient may have ASD should refer the child and family for appropriate educational and behavioral interventions as early as possible, rather than waiting for definitive testing.6
ADI-R has limitations in distinguishing ASD from other conditions, especially in very young children, and particularly in distinguishing ASD from childhood-onset schizophrenia.47 Similarly, ADOS, which is a semi-structured, standardized, observation assessment tool, also has limitations, including generating false-positive results, which can make it difficult to distinguish children and adolescents with developmental disabilities from those with ASD.47 However, in combination, these 2 tools are generally efficacious.47 Further research is warranted to develop and fine-tune definitive diagnostic tools with greater sensitivity and specificity.
A newer measure—the Autism Parent Screen for Infants (APSI) questionnaire—has been shown to be effective in detecting early signs predictive of ASD in high-risk infants (eg, siblings of children with ASD), and has potential as an early screening tool.48,49
Part 2 of this article will review nonpharmacologic and pharmacologic treatments for patients with ASD.
1. Bosl WJ, Tager-Flusberg H, Nelson CA. EEG analytics for early detection of autism spectrum disorder: a data-driven approach. Sci Rep. 2018;8(1):6828. doi:10.1038/s41598-018-24318-x
2. Dawson G, Carver L, Meltzoff AN, et al. Neural correlates of face and object recognition in young children with autism spectrum disorder, developmental delay, and typical development. Child Dev. 2002;73(3):700-717. doi:10.1111/1467-8624.00433
3. Frye RE, Vassall S, Kaur G, et al. Emerging biomarkers in autism spectrum disorder: a systematic review. Ann Transl Med. 2019;7(23):792. doi:10.21037/atm.2019.11.5
4. Gordon I, Vander Wyk BC, Bennett RH, et al. Oxytocin enhances brain function in children with autism. Proc Natl Acad Sci U S A. 2013;110(52):20953-20958. doi:10.1073/pnas.1312857110
5. Hicks SD, Carpenter RL, Wagner KE, et al. Saliva microRNA differentiates children with autism from peers with typical and atypical development. J Am Acad Child Adolesc Psychiatry. 2020;59(2):296-308.
6. Hyman SL, Levy SE, Myers SM, et al; Council on Children with Disabilities, Section on Developmental and Behavioral Pediatrics. Identification, evaluation, and management of children with autism spectrum disorder. Pediatrics. 2020;145(1):e20193447. doi:10.1542/peds.2019-3447
7. Kaiser MD, Hudac CM, Shultz S, et al. Neural signatures of autism. Proc Natl Acad Sci U S A. 2010;107(49):21223-1228. doi:10.1073/pnas.1010412107
8. Klin A, Lin DJ, Gorrindo P, et al. Two-year-olds with autism orient to non-social contingencies rather than biological motion. Nature. 2009;459(7244):257-261. doi:10.1038/nature07868
9. Chen T, Chen Y, Yuan M, et al. Towards developing a practical artificial intelligence tool for diagnosing and evaluating autism spectrum disorder: a study using multicenter ABIDE II datasets. JMIR Med Inform. 2020;8(5):e15767. doi:10.2196/15767
10. Maglione MA, Gans D, Das L, et al; Technical Expert Panel, & HRSA Autism Intervention Research – Behavioral (AIR‐B) Network. Nonmedical interventions for children with ASD: recommended guidelines and further research needs. Pediatrics. 2012;30(Suppl 2), S169-S178.
11. Monz BU, Houghton R, Law K, et al. Treatment patterns in children with autism in the United States. Autism Res. 2019;12(3):5170-526. doi:10.1002/aur.2070
12. Shukla DK, Keehn B, Lincoln AJ, et al. White matter compromise of callosal and subcortical fiber tracts in children with autism spectrum disorder: a diffusion tensor imaging study. J Am Acad Child Adolesc Psychiatry. 2010;49(12):1269-1278.e12782. doi:10.1016/j.jaac.2010.08.018
13. Sperdin HF, Schaer M. Aberrant development of speech processing in young children with autism: new insights from neuroimaging biomarkers. Front Neurosci. 2016;10:393. doi: 10.3389/fnins.2016.00393
14. Zwaigenbaum L, Brian JA, Ip A. Early detection for autism spectrum disorder in young children. Paediatr Child Health. 2019;24(7):424-443. doi:10.1093/pch/pxz119
15. Simms MD, Jin XM. Autism, language disorder, and social (pragmatic) communication disorder: DSM-V and differential diagnoses. Pediatr Rev. 2015;36(8):355-363. doi:10.1542/pir.36-8-355
16. Su Maw S, Haga C. Effectiveness of cognitive, developmental, and behavioural interventions for autism spectrum disorder in preschool-aged children: a systematic review and meta-analysis. Heliyon. 2018;4(9):e00763. doi:10.1016/j.heliyon.2018.e00763
17. Volkmar F, Siegel M, Woodbury-Smith M, et al. Practice parameter for the assessment and treatment of children and adolescents with autism spectrum disorder. J Am Acad Child Adolesc Psychiatry, 2014;53(2):237-257.
18. Landa RJ. Efficacy of early interventions for infants and young children with, and at risk for, autism spectrum disorders. Int Rev Psychiatry. 2018;30(1):25-39. doi:10.1080/09540261.2018.1432574
19. Lipkin PH, Macias MM; Council on Children with Disabilities, Section on Developmental and Behavioral Pediatrics. Promoting optimal development: identifying infants and young children with developmental disorders through developmental surveillance and screening. Pediatrics. 2020;145(1)e20193449. doi:10.1542/peds.2019-3449
20. Pickles A, Le Couteur A, Leadbitter K, et al. Parent-mediated social communication therapy for young children with autism (PACT): long-term follow-up of a randomised controlled trial. Lancet. 2016;388:2501-2509.
21. Rogers SJ, Estes A, Lord C, et al. Effects of a brief early start Denver model (ESDM)-based parent intervention on toddlers at risk for autism spectrum disorders: a randomized controlled trial. J Am Acad Child Adolesc Psychiatry. 2012;51(10):1052-1065. doi:10.1016/j.jaac.2012.08.003
22. Schreibman L, Dawson G, Stahmer AC, et al. Naturalistic developmental behavioral interventions: empirically validated treatments for autism spectrum disorder. J Autism Dev Disord. 2015;45(8):2411-2428. doi:10.1007/s10803-015-2407-8
23. Mundy P. A review of joint attention and social-cognitive brain systems in typical development and autism spectrum disorder. Eur J Neurosci. 2018;47(6):497-514.
24. Zwaigenbaum L, Bryson SE, Brian J, et al. Stability of diagnostic assessment for autism spectrum disorder between 18 and 36 months in a high-risk cohort. Autism Res. 2016;9(7):790-800. doi:10.1002/aur.1585
25. Anderson DK, Liang JW, Lord C. Predicting young adult outcome among more and less cognitively able individuals with autism spectrum disorders. J Child Psychol Psychiatry. 2014;55(5):485-494. doi:10.1111/jcpp.12178
26. Jones W, Carr K, Klin A. Absence of preferential looking to the eyes of approaching adults predicts level of social disability in 2-year-old toddlers with autism spectrum disorder. Arch Gen Psychiatry. 2008;65(8):946-954. doi:10.1001/archpsyc.65.8.946
27. Van der Donck S, Dzhelyova M, Vettori S, et al. Rapid neural categorization of angry and fearful faces is specifically impaired in boys with autism spectrum disorder. J Child Psychol Psychiatry. 2020;61(9):1019-1029. doi:10.1111/jcpp.13201
28. Thurm A, Farmer C, Salzman E, et al. State of the field: differentiating intellectual disability from autism spectrum disorder. Front Psychiatry. 2019;10:526. doi:10.3389/fpsyt.2019.00526
29. Kuno-Fujita A, Iwabuchi T, Wakusawa K, et al. Sensory processing patterns and fusiform activity during face processing in autism spectrum disorder. Autism Res. 2020;13(5):741-750. doi: 10.1002/aur.2283
30. Abrams DA, Lynch CJ, Cheng KM, et al. Underconnectivity between voice-selective cortex and reward circuitry in children with autism. Proc Natl Acad Sci U S A. 2013;110(29):12060-12065. doi:10.1073/pnas.1302982110
31. Osterling J, Dawson G. Early recognition of children with autism: a study of first birthday home videotapes. J Autism Dev Disord. 1994;24(3):247-257.
32. Zampella CJ, Csumitta KD, Simon E, et al. Interactional synchrony and its association with social and communication ability in children with and without autism spectrum disorder. J Autism Dev Disord. 2020;50(9):3195-3206. doi:10.1007/s10803-020-04412-8
33. McFayden T, Jarrett MA, White SW, et al. Sluggish cognitive tempo in autism spectrum disorder, ADHD, and their comorbidity: implications for impairment. J Clin Child Adolesc Psychol. 2020:1-8. doi:10.1080/15374416.2020.1716365
34. Baribeau DA, Vigod S, Pullenayegum E, et al. Repetitive behavior severity as an early indicator of risk for elevated anxiety symptoms in autism spectrum disorder. J Am Acad Child Adolesc Psychiatry. 2020;59(7):890-899.e3. doi:10.1016/j.jaac.2019.08.478
35. Li Y, Zhou Z, Chang C, et al. Anomalies in uncinate fasciculus development and social defects in preschoolers with autism spectrum disorder. BMC Psychiatry. 2019;19(1):399. doi:10.1186/s12888-019-2391-1
36. Payabvash S, Palacios EM, Owen JP, et al. White matter connectome edge density in children with autism spectrum disorders: potential imaging biomarkers using machine-learning models. Brain Connect. 2019;9(2):209-220. doi:10.1089/brain.2018.0658
37. Andrews DS, Lee JK, Solomon M, et al. A diffusion-weighted imaging tract-based spatial statistics study of autism spectrum disorder in preschool-aged children. J Neurodev Disord. 2019;11(1):32. doi:10.1186/s11689-019-9291-z
38. Chevallier C, Kohls G, Troiani V, et al. The social motivation theory of autism. Trends Cogn Sci. 2012;16(4):231-239. doi:10.1016/j.tics.2012.02.007
39. Boddaert N, Chabane N, Gervais H, et al. Superior temporal sulcus anatomical abnormalities in childhood autism: a voxel-based morphometry MRI study. Neuroimage. 2004;23(1):364-369. doi:10.1016/j.neuroimage.2004.06.016
40. Lord C, Petkova E, Hus V, et al. A multisite study of the clinical diagnosis of different autism spectrum disorders. Arch Gen Psychiatry. 2012;69(3):306-313. doi:10.1001/archgenpsychiatry.2011.148
41. Trillingsgaard A, ØStergaard JR. Autism in Angelman syndrome: an exploration of comorbidity. Autism. 2004;8(2):163-174.
42. Moss J, Howlin P. Autism spectrum disorders in genetic syndromes: implications for diagnosis, intervention and understanding the wider autism spectrum disorder population. J Intellect Disabil Res. 2009;53(10):852-873. doi:10.1111/j.1365-2788.2009.01197.x
43. McDuffie A, Thurman AJ, Hagerman RJ, et al. Symptoms of autism in males with Fragile X syndrome: a comparison to nonsyndromic ASD using current ADI-R scores. J Autism Dev Disord. 2015;45(7):1925-1937. doi:10.1007/s10803-013-2013-6
44. Ashwood KL, Tye C, Azadi B, et al. Brief report: adaptive functioning in children with ASD, ADHD and ASD + ADHD. J Autism Dev Disord. 2015;45(7):2235-4222. doi:10.1007/s10803-014-2352-y
45. Guthrie W, Wallis K, Bennett A, et al. Accuracy of autism screening in a large pediatric network. Pediatrics. 2019;144(4): e20183963. doi:10.1542/peds.2018-3963
46. Brian JA, Zwaigenbaum L, Ip A. Standards of diagnostic assessment for autism spectrum disorder. Paediatr Child Health. 2019;24(7):444-460. doi:10.1093/pch/pxz117
47. Frigaux A, Evrard R, Lighezzolo-Alnot J. ADI-R and ADOS and the differential diagnosis of autism spectrum disorders: interests, limits and openings. Encephale. 2019;45(5):441-448. doi:10.1016/j.encep.2019.07.002
48. Sacrey LR, Zwaigenbaum L, Bryson S, et al. Screening for behavioral signs of autism spectrum disorder in 9-month-old infant siblings. J Autism Dev Disord. 2021;51(3):839-848. doi:10.1007/s10803-020-04371-0
49. Sacrey LR, Bryson S, Zwaigenbaum L, et al. The autism parent screen for infants: predicting risk of autism spectrum disorder based on parent-reported behavior observed at 6-24 months of age. Autism. 2018;22(3):322-334
FIRST OF 2 PARTS
Autism spectrum disorder (ASD) is a complex, heterogenous neurodevelopmental disorder with genetic and environmental underpinnings, and an onset early in life.1-9 It affects social communication, cognition, and sensory-motor domains, and manifests as deficits in social reciprocity, repetitive behavior, restricted range of interests, and sensory sensitivities.6,10-14 In recent years, the prevalence of ASD has been increasing.3,6,10 A large percentage of individuals with ASD experience significant social deficits in adulthood,10 which often leads to isolation, depressive symptoms, and poor occupational and relationship functioning.15,16 Interventions in early childhood can result in significant and lasting changes in outcome and in functioning of individuals with ASD.
This article provides an update on various aspects of ASD diagnosis, with the goal of equipping clinicians with knowledge to help make an accurate ASD diagnosis at an early stage. Part 1 focuses on early detection and diagnosis, while Part 2 will describe treatment strategies.
Benefits of early detection
Substantial research has established that early intervention confers substantial benefits for outcomes among children with ASD.2,3,5,6,9,13,14,16-22 Earlier age of intervention correlates with greater developmental gain and symptom reduction.21,23 The atypical neural development responsible for ASD likely occurs much earlier than the behavioral manifestations of this disorder, which implies that there is a crucial period to intervene before behavioral features emerge.1 This necessitates early recognition of ASD,9,17 and the need for further research to find novel ways to detect ASD earlier.
In the United States, children with ASD are diagnosed with the disorder on average between age 3 and 4 years.6,24 However, evidence suggests there may be a prodromal phase for ASD during the first several months of life, wherein infants and toddlers exhibit developmentally inadequate communication and social skills and/or unusual behaviors.18 Behavioral signs suggestive of ASD may be evident as early as infancy, and commonly earlier than age 18 months.1,17,19 Problems with sleeping and eating may be evident in early childhood.19 Deficits in joint attention may be evident as early as age 6 months to 8 months. Research suggests that a diagnosis of ASD by trained, expert professionals is likely to be accurate at the age of 2, and even as early as 18 months.6,24
In a prospective study, Anderson et al25 found that 9% of children who were diagnosed with ASD at age 2 no longer met the diagnostic criteria for ASD by adulthood.6 Those who no longer met ASD criteria were more likely to have received early intervention, had a verbal IQ ≥70, and had experienced a larger decrease in repetitive behaviors between ages 2 and 3, compared with other youth in this study who had a verbal IQ ≥70. One of the limitations of this study was a small sample size (85 participants); larger, randomized studies are needed to replicate these findings.25
Continue to: Characteristics of ASD...
Characteristics of ASD
Table 16,8,10,13,15,26-29 outlines various characteristics of ASD, which may manifest in varying degrees among children with the condition.
Speech/language. Speech helps to facilitate bonding between parents and an infant by offering a soothing, pleasurable, and reinforcing experience.30 More than 50% of children with ASD have language delays or deficits that persist throughout adulthood.13 The extent of these language deficits varies; in general, the more severe the speech/language deficits, the more severe the long-term symptoms.13 Language deficits in young children with ASD tend to be of both the expressive and receptive type, with onset in infancy, which suggests that neural processes predate the emergence of behavioral symptoms of ASD, and also that early language deficits/delays could be a marker for or indicator of future risk of ASD.13 Individuals with ASD also have been noted to have limitations in orienting or attending to human voices.13,30
Facial recognition. Evidence has linked ASD with deficits in facial recognition that emerge in the first few months of life.2 Earlier studies have found that lack of attention to others’ faces was the strongest distinguishing factor between 1-year-olds with ASD and typically developing 1-year-olds.2,31 A recent study that used EEG to compare facial emotion recognition in boys with ASD vs typically developing boys found that boys with ASD exhibited significantly lower sensitivity to angry and fearful faces.27
Other features. A 2020 study (N = 37) found that compared with typically developing children, those with ASD show less “interactional synchrony’’ (a dynamic process in which the timing of children and caregivers’ behaviors [specifically, vocalizations and movements] become mutually coordinated) with both familiar and unfamiliar adults.32 These researchers concluded that impairment in interactional synchrony may be linked to social communication deficits in ASD.32
A recent study (N = 98) evaluated “sluggish cognitive tempo” in 3 groups of children: children with attention-deficit/hyperactivity disorder (ADHD), children with ASD, and children with both ADHD and ASD.33 It found that children with ASD exhibited sluggish cognitive tempo at levels similar to those of the other 2 groups, and indicated that sluggish cognitive tempo may be linked with “social and global impairment above and beyond” the impairment associated with ASD.
Understanding early aberrations in neurobiologic processes in ASD can help develop biomarkers for early recognition of ASD, as well as guide the development of targeted interventions and treatments (Box1-3,7-9,12,13,30,35-39).
Box
Compared with individuals who do not have autism spectrum disorder (ASD), individuals with ASD exhibit anatomical differences in the brain that can be seen on MRI.9,35 Brain regions affected in ASD include the frontal gyrus, temporal gyrus, cingulate gyrus, postcentral gyrus, precuneus, caudate, and hippocampus.9 Some studies have found anomalous structural neural characteristics in infants, such as in the uncinate fasciculus, that correlated with later joint attention challenges, while others have found aberrations in the corpus callosum(responsible for transfer of procedural learning between the hemispheres, and oculomotor response)and internal capsule (responsible for sensorimotor function, as well as other functions) in children with ASD.12
Widespread white matter anomalies have been noted in ASD.12,35,36 In a 2-year longitudinal study that used diffusion tensor imaging, Li et al35 found that preschool children with ASD experience overgrowth of the uncinate fasciculus, which is one of the brain regions implicated in socioemotional processing, and concluded that this overgrowth correlated with ASD severity.35 Andrews et al37 used diffusion-weighted MRI to examine white matter in 127 preschool children. They found that compared with typically developing children, children with ASD exhibited altered white matter microstructure.37
Research suggests that developing representations of the reward value of social stimuli may be challenging for children with ASD.2 Abrams et al30 used resting-state functional brain MRI to evaluate children with typical development and children with highfunctioning, “verbally fluent” ASD. They found that the children with ASD exhibited lower functional connectivity between voice-specific left hemisphere posterior superior temporal sulcus and areas representing the reward circuitry.30 This study also found that children with ASD had underconnectivity between the right hemisphere posterior superior temporal sulcus (which deals with speech prosody) and areas known for emotion-linked associative learning, the orbitofrontal cortex and amygdala.30 These findings are thought to align with the social motivation theory of ASD.13,30,38
The extent of underconnectivity between these systems was found to determine the severity of communication challenges in high-functioning children with ASD.30 One MRI study observed lower gray matter volume in the voice-selective bilateral superior temporal sulcus in children age approximately 9 to 11 years with ASD.39
Neural systems responsible for facial recognition (particularly the right fusiform gyrus and other brain areas) have been shown to exist or begin “very early in life,” which suggests that impaired face recognition may be an early marker of ASD.2 In addition to problems with visual scanning, preferential attention to (and visual sensitivity to) biological motion is a forerunner for the development of social interactions in infants, specifically in regard to being able to detect and recognize emotion, which is considered vital for attachment.7,8 Impaired biological motion perception has been found in very young children with ASD.7,8 This presents an important avenue/potential biomarker for further research to better understand neurobiologic processes underlying atypical development at an earlier age.3,8
Early neural biomarkers for ASD
Nonlinear EEG values may serve as an early neurobiomarker for detecting ASD in young children.1 Because it is relatively inexpensive and convenient, EEG may be highly useful for detecting ASD.1 A study that compared EEG results of 99 infants who had siblings with ASD and 89 low-risk controls from age 3 months to 36 months found that nonlinear EEG measurements predicted with high accuracy later diagnosis of ASD, and were strongly correlated with later Autism Diagnostic Observation Schedule scores.1
Continue to: A complex differential diagnosis...
A complex differential diagnosis
The differential diagnosis of ASD warrants careful attention and consideration to rule out other developmental and psychiatric conditions.
Intellectual disability (ID). DSM-5 diagnostic criteria for ASD necessitate that disturbances are not better explained by ID or global developmental delay and that deficits should exceed impairment consistent with the level of intellectual disability.28 Still, ASD is often overdiagnosed in children with ID.28 Research suggests phenotypic and genetic overlap between ID and ASD.28 Social functioning is often impaired in patients with ID; the greater the severity of ID, the greater the degree of social deficits.28 In approximately 30% of cases, ASD and ID are comorbid.6 This overlap and comorbidity can pose a challenge, particularly due to the inherent complexities involved in assessment and differentiation.28 When ID is present in ASD, there is a greater degree of social-communication deficits.6 It may be difficult to assess for ASD symptoms in children with severe ID.28 Although there is no minimum age or developmental level below which ASD should not be diagnosed, some studies have started to use minimum criteria for diagnosis, such as a nonverbal mental age of 18 months.28,40 Commonly used tests for ASD have much lower specificity when used for children with nonverbal age <15 months.28 It would make sense, then, that the presence of ID might significantly affect the results of these diagnostic tests.28
Other conditions that need to be ruled out include language disorders, hearing loss, rare genetic neurodevelopmental disorders (eg, Fragile X syndrome,3 Rett syndrome6), childhood-onset schizophrenia, obsessive-compulsive disorder, attachment disorders, and other conditions.18 ASD may be overdiagnosed in children with genetic disorders such as Angelman syndrome.41 In a systematic review, Moss and Howlin42 recommended caution when evaluating ASD-like behavioral symptoms in children with genetic syndromes and severe ID. On the other hand, some research has observed that individuals with Fragile X syndrome may exhibit symptoms that meet criteria for ASD.6,43 McDuffie et al43 used the Autism Diagnostic Interview-Revised (ADI-R) to compare boys with Fragile X syndrome who also met criteria for ASD with boys with nonsyndromic ASD. Those in the former group had lesser impairment in social smiling, offering, showing, and nonverbal gestures, but had more complex mannerisms, compared with boys in the latter group.43
Milder manifestations of ASD may be more challenging to diagnose,1 particularly in children age <3 and those with above-average cognition.6 Generally, in the case of a patient with ASD, parents find that the child did not have a period of typical development, or unusual behaviors were evident early on.17
ASD can be comorbid with ADHD. The presence of ADHD may mask or delay the diagnosis of ASD in children.6 In children with both ASD and ADHD, studies have found greater reduction in social and adaptive functioning compared with children with ADHD alone.44
Table 26,10,15,17,31,43 highlights some of the features that can be used to distinguish ASD from other conditions.

Continue to: Screening and diagnosis...
Screening and diagnosis
A medical workup is the first step to rule out other potential conditions that could be masquerading as ASD.17 Obtain a comprehensive history from parents/caregivers, particularly regarding social, behavioral, movement, sensory, and developmental aspects. In addition, audiologic testing is an essential step. Consider genetic testing, particularly if any dysmorphic features and/or ID are present, both of which confer additional risk for a genetic syndrome.6 A physical exam to detect any neurologic anomalies, organ dysfunction, and body dysmorphic features should be conducted.6
The Modified Checklist for Autism in Toddlers–Revised (MCHAT-R) is a commonly used, validated parental screening survey for ASD.5,6 Research has shown that this survey has <50% specificity.5A recent American Academy of Pediatrics Clinical Report recommended universal screening for ASD at pediatric visits at age 18 months and at 24 months, in addition to developmental screening for all children at routine pediatric visits at age 9, 18, and 30 months.6,19
Screening tools such as the Modified Checklist for Autism in Toddlers with Follow-Up (M-CHAT/F) can be integrated into routine primary health care. In a large (N = 25,999) study, Guthrie et al45 used M-CHAT/F to conduct universal, primary care–based screening in young children. They found that the positive predictive value of M-CHAT/F was lower among girls, children of color, and those from lower-income households. There is a need for development of screening tools with higher accuracy and sensitivity for identifying young children with ASD regardless of their ethnic or socioeconomic background, and also for children older than 30 months.5,6,45
Definitive diagnosis of ASD is ideally done by a multidisciplinary team46 using established gold standard measures such as the ADOS (Autism Diagnostic Observation Schedule) and ADI-R.47 Such multidisciplinary teams usually include a child psychiatrist, child psychologist, speech therapist, occupational therapist, school educator, and developmental pediatrician. However, because there are long wait times to receive this type of diagnosis in the United States,6 in the interest of not missing the critical window of early intervention, physicians who suspect a patient may have ASD should refer the child and family for appropriate educational and behavioral interventions as early as possible, rather than waiting for definitive testing.6
ADI-R has limitations in distinguishing ASD from other conditions, especially in very young children, and particularly in distinguishing ASD from childhood-onset schizophrenia.47 Similarly, ADOS, which is a semi-structured, standardized, observation assessment tool, also has limitations, including generating false-positive results, which can make it difficult to distinguish children and adolescents with developmental disabilities from those with ASD.47 However, in combination, these 2 tools are generally efficacious.47 Further research is warranted to develop and fine-tune definitive diagnostic tools with greater sensitivity and specificity.
A newer measure—the Autism Parent Screen for Infants (APSI) questionnaire—has been shown to be effective in detecting early signs predictive of ASD in high-risk infants (eg, siblings of children with ASD), and has potential as an early screening tool.48,49
Part 2 of this article will review nonpharmacologic and pharmacologic treatments for patients with ASD.
FIRST OF 2 PARTS
Autism spectrum disorder (ASD) is a complex, heterogenous neurodevelopmental disorder with genetic and environmental underpinnings, and an onset early in life.1-9 It affects social communication, cognition, and sensory-motor domains, and manifests as deficits in social reciprocity, repetitive behavior, restricted range of interests, and sensory sensitivities.6,10-14 In recent years, the prevalence of ASD has been increasing.3,6,10 A large percentage of individuals with ASD experience significant social deficits in adulthood,10 which often leads to isolation, depressive symptoms, and poor occupational and relationship functioning.15,16 Interventions in early childhood can result in significant and lasting changes in outcome and in functioning of individuals with ASD.
This article provides an update on various aspects of ASD diagnosis, with the goal of equipping clinicians with knowledge to help make an accurate ASD diagnosis at an early stage. Part 1 focuses on early detection and diagnosis, while Part 2 will describe treatment strategies.
Benefits of early detection
Substantial research has established that early intervention confers substantial benefits for outcomes among children with ASD.2,3,5,6,9,13,14,16-22 Earlier age of intervention correlates with greater developmental gain and symptom reduction.21,23 The atypical neural development responsible for ASD likely occurs much earlier than the behavioral manifestations of this disorder, which implies that there is a crucial period to intervene before behavioral features emerge.1 This necessitates early recognition of ASD,9,17 and the need for further research to find novel ways to detect ASD earlier.
In the United States, children with ASD are diagnosed with the disorder on average between age 3 and 4 years.6,24 However, evidence suggests there may be a prodromal phase for ASD during the first several months of life, wherein infants and toddlers exhibit developmentally inadequate communication and social skills and/or unusual behaviors.18 Behavioral signs suggestive of ASD may be evident as early as infancy, and commonly earlier than age 18 months.1,17,19 Problems with sleeping and eating may be evident in early childhood.19 Deficits in joint attention may be evident as early as age 6 months to 8 months. Research suggests that a diagnosis of ASD by trained, expert professionals is likely to be accurate at the age of 2, and even as early as 18 months.6,24
In a prospective study, Anderson et al25 found that 9% of children who were diagnosed with ASD at age 2 no longer met the diagnostic criteria for ASD by adulthood.6 Those who no longer met ASD criteria were more likely to have received early intervention, had a verbal IQ ≥70, and had experienced a larger decrease in repetitive behaviors between ages 2 and 3, compared with other youth in this study who had a verbal IQ ≥70. One of the limitations of this study was a small sample size (85 participants); larger, randomized studies are needed to replicate these findings.25
Continue to: Characteristics of ASD...
Characteristics of ASD
Table 16,8,10,13,15,26-29 outlines various characteristics of ASD, which may manifest in varying degrees among children with the condition.
Speech/language. Speech helps to facilitate bonding between parents and an infant by offering a soothing, pleasurable, and reinforcing experience.30 More than 50% of children with ASD have language delays or deficits that persist throughout adulthood.13 The extent of these language deficits varies; in general, the more severe the speech/language deficits, the more severe the long-term symptoms.13 Language deficits in young children with ASD tend to be of both the expressive and receptive type, with onset in infancy, which suggests that neural processes predate the emergence of behavioral symptoms of ASD, and also that early language deficits/delays could be a marker for or indicator of future risk of ASD.13 Individuals with ASD also have been noted to have limitations in orienting or attending to human voices.13,30
Facial recognition. Evidence has linked ASD with deficits in facial recognition that emerge in the first few months of life.2 Earlier studies have found that lack of attention to others’ faces was the strongest distinguishing factor between 1-year-olds with ASD and typically developing 1-year-olds.2,31 A recent study that used EEG to compare facial emotion recognition in boys with ASD vs typically developing boys found that boys with ASD exhibited significantly lower sensitivity to angry and fearful faces.27
Other features. A 2020 study (N = 37) found that compared with typically developing children, those with ASD show less “interactional synchrony’’ (a dynamic process in which the timing of children and caregivers’ behaviors [specifically, vocalizations and movements] become mutually coordinated) with both familiar and unfamiliar adults.32 These researchers concluded that impairment in interactional synchrony may be linked to social communication deficits in ASD.32
A recent study (N = 98) evaluated “sluggish cognitive tempo” in 3 groups of children: children with attention-deficit/hyperactivity disorder (ADHD), children with ASD, and children with both ADHD and ASD.33 It found that children with ASD exhibited sluggish cognitive tempo at levels similar to those of the other 2 groups, and indicated that sluggish cognitive tempo may be linked with “social and global impairment above and beyond” the impairment associated with ASD.
Understanding early aberrations in neurobiologic processes in ASD can help develop biomarkers for early recognition of ASD, as well as guide the development of targeted interventions and treatments (Box1-3,7-9,12,13,30,35-39).
Box
Compared with individuals who do not have autism spectrum disorder (ASD), individuals with ASD exhibit anatomical differences in the brain that can be seen on MRI.9,35 Brain regions affected in ASD include the frontal gyrus, temporal gyrus, cingulate gyrus, postcentral gyrus, precuneus, caudate, and hippocampus.9 Some studies have found anomalous structural neural characteristics in infants, such as in the uncinate fasciculus, that correlated with later joint attention challenges, while others have found aberrations in the corpus callosum(responsible for transfer of procedural learning between the hemispheres, and oculomotor response)and internal capsule (responsible for sensorimotor function, as well as other functions) in children with ASD.12
Widespread white matter anomalies have been noted in ASD.12,35,36 In a 2-year longitudinal study that used diffusion tensor imaging, Li et al35 found that preschool children with ASD experience overgrowth of the uncinate fasciculus, which is one of the brain regions implicated in socioemotional processing, and concluded that this overgrowth correlated with ASD severity.35 Andrews et al37 used diffusion-weighted MRI to examine white matter in 127 preschool children. They found that compared with typically developing children, children with ASD exhibited altered white matter microstructure.37
Research suggests that developing representations of the reward value of social stimuli may be challenging for children with ASD.2 Abrams et al30 used resting-state functional brain MRI to evaluate children with typical development and children with highfunctioning, “verbally fluent” ASD. They found that the children with ASD exhibited lower functional connectivity between voice-specific left hemisphere posterior superior temporal sulcus and areas representing the reward circuitry.30 This study also found that children with ASD had underconnectivity between the right hemisphere posterior superior temporal sulcus (which deals with speech prosody) and areas known for emotion-linked associative learning, the orbitofrontal cortex and amygdala.30 These findings are thought to align with the social motivation theory of ASD.13,30,38
The extent of underconnectivity between these systems was found to determine the severity of communication challenges in high-functioning children with ASD.30 One MRI study observed lower gray matter volume in the voice-selective bilateral superior temporal sulcus in children age approximately 9 to 11 years with ASD.39
Neural systems responsible for facial recognition (particularly the right fusiform gyrus and other brain areas) have been shown to exist or begin “very early in life,” which suggests that impaired face recognition may be an early marker of ASD.2 In addition to problems with visual scanning, preferential attention to (and visual sensitivity to) biological motion is a forerunner for the development of social interactions in infants, specifically in regard to being able to detect and recognize emotion, which is considered vital for attachment.7,8 Impaired biological motion perception has been found in very young children with ASD.7,8 This presents an important avenue/potential biomarker for further research to better understand neurobiologic processes underlying atypical development at an earlier age.3,8
Early neural biomarkers for ASD
Nonlinear EEG values may serve as an early neurobiomarker for detecting ASD in young children.1 Because it is relatively inexpensive and convenient, EEG may be highly useful for detecting ASD.1 A study that compared EEG results of 99 infants who had siblings with ASD and 89 low-risk controls from age 3 months to 36 months found that nonlinear EEG measurements predicted with high accuracy later diagnosis of ASD, and were strongly correlated with later Autism Diagnostic Observation Schedule scores.1
Continue to: A complex differential diagnosis...
A complex differential diagnosis
The differential diagnosis of ASD warrants careful attention and consideration to rule out other developmental and psychiatric conditions.
Intellectual disability (ID). DSM-5 diagnostic criteria for ASD necessitate that disturbances are not better explained by ID or global developmental delay and that deficits should exceed impairment consistent with the level of intellectual disability.28 Still, ASD is often overdiagnosed in children with ID.28 Research suggests phenotypic and genetic overlap between ID and ASD.28 Social functioning is often impaired in patients with ID; the greater the severity of ID, the greater the degree of social deficits.28 In approximately 30% of cases, ASD and ID are comorbid.6 This overlap and comorbidity can pose a challenge, particularly due to the inherent complexities involved in assessment and differentiation.28 When ID is present in ASD, there is a greater degree of social-communication deficits.6 It may be difficult to assess for ASD symptoms in children with severe ID.28 Although there is no minimum age or developmental level below which ASD should not be diagnosed, some studies have started to use minimum criteria for diagnosis, such as a nonverbal mental age of 18 months.28,40 Commonly used tests for ASD have much lower specificity when used for children with nonverbal age <15 months.28 It would make sense, then, that the presence of ID might significantly affect the results of these diagnostic tests.28
Other conditions that need to be ruled out include language disorders, hearing loss, rare genetic neurodevelopmental disorders (eg, Fragile X syndrome,3 Rett syndrome6), childhood-onset schizophrenia, obsessive-compulsive disorder, attachment disorders, and other conditions.18 ASD may be overdiagnosed in children with genetic disorders such as Angelman syndrome.41 In a systematic review, Moss and Howlin42 recommended caution when evaluating ASD-like behavioral symptoms in children with genetic syndromes and severe ID. On the other hand, some research has observed that individuals with Fragile X syndrome may exhibit symptoms that meet criteria for ASD.6,43 McDuffie et al43 used the Autism Diagnostic Interview-Revised (ADI-R) to compare boys with Fragile X syndrome who also met criteria for ASD with boys with nonsyndromic ASD. Those in the former group had lesser impairment in social smiling, offering, showing, and nonverbal gestures, but had more complex mannerisms, compared with boys in the latter group.43
Milder manifestations of ASD may be more challenging to diagnose,1 particularly in children age <3 and those with above-average cognition.6 Generally, in the case of a patient with ASD, parents find that the child did not have a period of typical development, or unusual behaviors were evident early on.17
ASD can be comorbid with ADHD. The presence of ADHD may mask or delay the diagnosis of ASD in children.6 In children with both ASD and ADHD, studies have found greater reduction in social and adaptive functioning compared with children with ADHD alone.44
Table 26,10,15,17,31,43 highlights some of the features that can be used to distinguish ASD from other conditions.

Continue to: Screening and diagnosis...
Screening and diagnosis
A medical workup is the first step to rule out other potential conditions that could be masquerading as ASD.17 Obtain a comprehensive history from parents/caregivers, particularly regarding social, behavioral, movement, sensory, and developmental aspects. In addition, audiologic testing is an essential step. Consider genetic testing, particularly if any dysmorphic features and/or ID are present, both of which confer additional risk for a genetic syndrome.6 A physical exam to detect any neurologic anomalies, organ dysfunction, and body dysmorphic features should be conducted.6
The Modified Checklist for Autism in Toddlers–Revised (MCHAT-R) is a commonly used, validated parental screening survey for ASD.5,6 Research has shown that this survey has <50% specificity.5A recent American Academy of Pediatrics Clinical Report recommended universal screening for ASD at pediatric visits at age 18 months and at 24 months, in addition to developmental screening for all children at routine pediatric visits at age 9, 18, and 30 months.6,19
Screening tools such as the Modified Checklist for Autism in Toddlers with Follow-Up (M-CHAT/F) can be integrated into routine primary health care. In a large (N = 25,999) study, Guthrie et al45 used M-CHAT/F to conduct universal, primary care–based screening in young children. They found that the positive predictive value of M-CHAT/F was lower among girls, children of color, and those from lower-income households. There is a need for development of screening tools with higher accuracy and sensitivity for identifying young children with ASD regardless of their ethnic or socioeconomic background, and also for children older than 30 months.5,6,45
Definitive diagnosis of ASD is ideally done by a multidisciplinary team46 using established gold standard measures such as the ADOS (Autism Diagnostic Observation Schedule) and ADI-R.47 Such multidisciplinary teams usually include a child psychiatrist, child psychologist, speech therapist, occupational therapist, school educator, and developmental pediatrician. However, because there are long wait times to receive this type of diagnosis in the United States,6 in the interest of not missing the critical window of early intervention, physicians who suspect a patient may have ASD should refer the child and family for appropriate educational and behavioral interventions as early as possible, rather than waiting for definitive testing.6
ADI-R has limitations in distinguishing ASD from other conditions, especially in very young children, and particularly in distinguishing ASD from childhood-onset schizophrenia.47 Similarly, ADOS, which is a semi-structured, standardized, observation assessment tool, also has limitations, including generating false-positive results, which can make it difficult to distinguish children and adolescents with developmental disabilities from those with ASD.47 However, in combination, these 2 tools are generally efficacious.47 Further research is warranted to develop and fine-tune definitive diagnostic tools with greater sensitivity and specificity.
A newer measure—the Autism Parent Screen for Infants (APSI) questionnaire—has been shown to be effective in detecting early signs predictive of ASD in high-risk infants (eg, siblings of children with ASD), and has potential as an early screening tool.48,49
Part 2 of this article will review nonpharmacologic and pharmacologic treatments for patients with ASD.
1. Bosl WJ, Tager-Flusberg H, Nelson CA. EEG analytics for early detection of autism spectrum disorder: a data-driven approach. Sci Rep. 2018;8(1):6828. doi:10.1038/s41598-018-24318-x
2. Dawson G, Carver L, Meltzoff AN, et al. Neural correlates of face and object recognition in young children with autism spectrum disorder, developmental delay, and typical development. Child Dev. 2002;73(3):700-717. doi:10.1111/1467-8624.00433
3. Frye RE, Vassall S, Kaur G, et al. Emerging biomarkers in autism spectrum disorder: a systematic review. Ann Transl Med. 2019;7(23):792. doi:10.21037/atm.2019.11.5
4. Gordon I, Vander Wyk BC, Bennett RH, et al. Oxytocin enhances brain function in children with autism. Proc Natl Acad Sci U S A. 2013;110(52):20953-20958. doi:10.1073/pnas.1312857110
5. Hicks SD, Carpenter RL, Wagner KE, et al. Saliva microRNA differentiates children with autism from peers with typical and atypical development. J Am Acad Child Adolesc Psychiatry. 2020;59(2):296-308.
6. Hyman SL, Levy SE, Myers SM, et al; Council on Children with Disabilities, Section on Developmental and Behavioral Pediatrics. Identification, evaluation, and management of children with autism spectrum disorder. Pediatrics. 2020;145(1):e20193447. doi:10.1542/peds.2019-3447
7. Kaiser MD, Hudac CM, Shultz S, et al. Neural signatures of autism. Proc Natl Acad Sci U S A. 2010;107(49):21223-1228. doi:10.1073/pnas.1010412107
8. Klin A, Lin DJ, Gorrindo P, et al. Two-year-olds with autism orient to non-social contingencies rather than biological motion. Nature. 2009;459(7244):257-261. doi:10.1038/nature07868
9. Chen T, Chen Y, Yuan M, et al. Towards developing a practical artificial intelligence tool for diagnosing and evaluating autism spectrum disorder: a study using multicenter ABIDE II datasets. JMIR Med Inform. 2020;8(5):e15767. doi:10.2196/15767
10. Maglione MA, Gans D, Das L, et al; Technical Expert Panel, & HRSA Autism Intervention Research – Behavioral (AIR‐B) Network. Nonmedical interventions for children with ASD: recommended guidelines and further research needs. Pediatrics. 2012;30(Suppl 2), S169-S178.
11. Monz BU, Houghton R, Law K, et al. Treatment patterns in children with autism in the United States. Autism Res. 2019;12(3):5170-526. doi:10.1002/aur.2070
12. Shukla DK, Keehn B, Lincoln AJ, et al. White matter compromise of callosal and subcortical fiber tracts in children with autism spectrum disorder: a diffusion tensor imaging study. J Am Acad Child Adolesc Psychiatry. 2010;49(12):1269-1278.e12782. doi:10.1016/j.jaac.2010.08.018
13. Sperdin HF, Schaer M. Aberrant development of speech processing in young children with autism: new insights from neuroimaging biomarkers. Front Neurosci. 2016;10:393. doi: 10.3389/fnins.2016.00393
14. Zwaigenbaum L, Brian JA, Ip A. Early detection for autism spectrum disorder in young children. Paediatr Child Health. 2019;24(7):424-443. doi:10.1093/pch/pxz119
15. Simms MD, Jin XM. Autism, language disorder, and social (pragmatic) communication disorder: DSM-V and differential diagnoses. Pediatr Rev. 2015;36(8):355-363. doi:10.1542/pir.36-8-355
16. Su Maw S, Haga C. Effectiveness of cognitive, developmental, and behavioural interventions for autism spectrum disorder in preschool-aged children: a systematic review and meta-analysis. Heliyon. 2018;4(9):e00763. doi:10.1016/j.heliyon.2018.e00763
17. Volkmar F, Siegel M, Woodbury-Smith M, et al. Practice parameter for the assessment and treatment of children and adolescents with autism spectrum disorder. J Am Acad Child Adolesc Psychiatry, 2014;53(2):237-257.
18. Landa RJ. Efficacy of early interventions for infants and young children with, and at risk for, autism spectrum disorders. Int Rev Psychiatry. 2018;30(1):25-39. doi:10.1080/09540261.2018.1432574
19. Lipkin PH, Macias MM; Council on Children with Disabilities, Section on Developmental and Behavioral Pediatrics. Promoting optimal development: identifying infants and young children with developmental disorders through developmental surveillance and screening. Pediatrics. 2020;145(1)e20193449. doi:10.1542/peds.2019-3449
20. Pickles A, Le Couteur A, Leadbitter K, et al. Parent-mediated social communication therapy for young children with autism (PACT): long-term follow-up of a randomised controlled trial. Lancet. 2016;388:2501-2509.
21. Rogers SJ, Estes A, Lord C, et al. Effects of a brief early start Denver model (ESDM)-based parent intervention on toddlers at risk for autism spectrum disorders: a randomized controlled trial. J Am Acad Child Adolesc Psychiatry. 2012;51(10):1052-1065. doi:10.1016/j.jaac.2012.08.003
22. Schreibman L, Dawson G, Stahmer AC, et al. Naturalistic developmental behavioral interventions: empirically validated treatments for autism spectrum disorder. J Autism Dev Disord. 2015;45(8):2411-2428. doi:10.1007/s10803-015-2407-8
23. Mundy P. A review of joint attention and social-cognitive brain systems in typical development and autism spectrum disorder. Eur J Neurosci. 2018;47(6):497-514.
24. Zwaigenbaum L, Bryson SE, Brian J, et al. Stability of diagnostic assessment for autism spectrum disorder between 18 and 36 months in a high-risk cohort. Autism Res. 2016;9(7):790-800. doi:10.1002/aur.1585
25. Anderson DK, Liang JW, Lord C. Predicting young adult outcome among more and less cognitively able individuals with autism spectrum disorders. J Child Psychol Psychiatry. 2014;55(5):485-494. doi:10.1111/jcpp.12178
26. Jones W, Carr K, Klin A. Absence of preferential looking to the eyes of approaching adults predicts level of social disability in 2-year-old toddlers with autism spectrum disorder. Arch Gen Psychiatry. 2008;65(8):946-954. doi:10.1001/archpsyc.65.8.946
27. Van der Donck S, Dzhelyova M, Vettori S, et al. Rapid neural categorization of angry and fearful faces is specifically impaired in boys with autism spectrum disorder. J Child Psychol Psychiatry. 2020;61(9):1019-1029. doi:10.1111/jcpp.13201
28. Thurm A, Farmer C, Salzman E, et al. State of the field: differentiating intellectual disability from autism spectrum disorder. Front Psychiatry. 2019;10:526. doi:10.3389/fpsyt.2019.00526
29. Kuno-Fujita A, Iwabuchi T, Wakusawa K, et al. Sensory processing patterns and fusiform activity during face processing in autism spectrum disorder. Autism Res. 2020;13(5):741-750. doi: 10.1002/aur.2283
30. Abrams DA, Lynch CJ, Cheng KM, et al. Underconnectivity between voice-selective cortex and reward circuitry in children with autism. Proc Natl Acad Sci U S A. 2013;110(29):12060-12065. doi:10.1073/pnas.1302982110
31. Osterling J, Dawson G. Early recognition of children with autism: a study of first birthday home videotapes. J Autism Dev Disord. 1994;24(3):247-257.
32. Zampella CJ, Csumitta KD, Simon E, et al. Interactional synchrony and its association with social and communication ability in children with and without autism spectrum disorder. J Autism Dev Disord. 2020;50(9):3195-3206. doi:10.1007/s10803-020-04412-8
33. McFayden T, Jarrett MA, White SW, et al. Sluggish cognitive tempo in autism spectrum disorder, ADHD, and their comorbidity: implications for impairment. J Clin Child Adolesc Psychol. 2020:1-8. doi:10.1080/15374416.2020.1716365
34. Baribeau DA, Vigod S, Pullenayegum E, et al. Repetitive behavior severity as an early indicator of risk for elevated anxiety symptoms in autism spectrum disorder. J Am Acad Child Adolesc Psychiatry. 2020;59(7):890-899.e3. doi:10.1016/j.jaac.2019.08.478
35. Li Y, Zhou Z, Chang C, et al. Anomalies in uncinate fasciculus development and social defects in preschoolers with autism spectrum disorder. BMC Psychiatry. 2019;19(1):399. doi:10.1186/s12888-019-2391-1
36. Payabvash S, Palacios EM, Owen JP, et al. White matter connectome edge density in children with autism spectrum disorders: potential imaging biomarkers using machine-learning models. Brain Connect. 2019;9(2):209-220. doi:10.1089/brain.2018.0658
37. Andrews DS, Lee JK, Solomon M, et al. A diffusion-weighted imaging tract-based spatial statistics study of autism spectrum disorder in preschool-aged children. J Neurodev Disord. 2019;11(1):32. doi:10.1186/s11689-019-9291-z
38. Chevallier C, Kohls G, Troiani V, et al. The social motivation theory of autism. Trends Cogn Sci. 2012;16(4):231-239. doi:10.1016/j.tics.2012.02.007
39. Boddaert N, Chabane N, Gervais H, et al. Superior temporal sulcus anatomical abnormalities in childhood autism: a voxel-based morphometry MRI study. Neuroimage. 2004;23(1):364-369. doi:10.1016/j.neuroimage.2004.06.016
40. Lord C, Petkova E, Hus V, et al. A multisite study of the clinical diagnosis of different autism spectrum disorders. Arch Gen Psychiatry. 2012;69(3):306-313. doi:10.1001/archgenpsychiatry.2011.148
41. Trillingsgaard A, ØStergaard JR. Autism in Angelman syndrome: an exploration of comorbidity. Autism. 2004;8(2):163-174.
42. Moss J, Howlin P. Autism spectrum disorders in genetic syndromes: implications for diagnosis, intervention and understanding the wider autism spectrum disorder population. J Intellect Disabil Res. 2009;53(10):852-873. doi:10.1111/j.1365-2788.2009.01197.x
43. McDuffie A, Thurman AJ, Hagerman RJ, et al. Symptoms of autism in males with Fragile X syndrome: a comparison to nonsyndromic ASD using current ADI-R scores. J Autism Dev Disord. 2015;45(7):1925-1937. doi:10.1007/s10803-013-2013-6
44. Ashwood KL, Tye C, Azadi B, et al. Brief report: adaptive functioning in children with ASD, ADHD and ASD + ADHD. J Autism Dev Disord. 2015;45(7):2235-4222. doi:10.1007/s10803-014-2352-y
45. Guthrie W, Wallis K, Bennett A, et al. Accuracy of autism screening in a large pediatric network. Pediatrics. 2019;144(4): e20183963. doi:10.1542/peds.2018-3963
46. Brian JA, Zwaigenbaum L, Ip A. Standards of diagnostic assessment for autism spectrum disorder. Paediatr Child Health. 2019;24(7):444-460. doi:10.1093/pch/pxz117
47. Frigaux A, Evrard R, Lighezzolo-Alnot J. ADI-R and ADOS and the differential diagnosis of autism spectrum disorders: interests, limits and openings. Encephale. 2019;45(5):441-448. doi:10.1016/j.encep.2019.07.002
48. Sacrey LR, Zwaigenbaum L, Bryson S, et al. Screening for behavioral signs of autism spectrum disorder in 9-month-old infant siblings. J Autism Dev Disord. 2021;51(3):839-848. doi:10.1007/s10803-020-04371-0
49. Sacrey LR, Bryson S, Zwaigenbaum L, et al. The autism parent screen for infants: predicting risk of autism spectrum disorder based on parent-reported behavior observed at 6-24 months of age. Autism. 2018;22(3):322-334
1. Bosl WJ, Tager-Flusberg H, Nelson CA. EEG analytics for early detection of autism spectrum disorder: a data-driven approach. Sci Rep. 2018;8(1):6828. doi:10.1038/s41598-018-24318-x
2. Dawson G, Carver L, Meltzoff AN, et al. Neural correlates of face and object recognition in young children with autism spectrum disorder, developmental delay, and typical development. Child Dev. 2002;73(3):700-717. doi:10.1111/1467-8624.00433
3. Frye RE, Vassall S, Kaur G, et al. Emerging biomarkers in autism spectrum disorder: a systematic review. Ann Transl Med. 2019;7(23):792. doi:10.21037/atm.2019.11.5
4. Gordon I, Vander Wyk BC, Bennett RH, et al. Oxytocin enhances brain function in children with autism. Proc Natl Acad Sci U S A. 2013;110(52):20953-20958. doi:10.1073/pnas.1312857110
5. Hicks SD, Carpenter RL, Wagner KE, et al. Saliva microRNA differentiates children with autism from peers with typical and atypical development. J Am Acad Child Adolesc Psychiatry. 2020;59(2):296-308.
6. Hyman SL, Levy SE, Myers SM, et al; Council on Children with Disabilities, Section on Developmental and Behavioral Pediatrics. Identification, evaluation, and management of children with autism spectrum disorder. Pediatrics. 2020;145(1):e20193447. doi:10.1542/peds.2019-3447
7. Kaiser MD, Hudac CM, Shultz S, et al. Neural signatures of autism. Proc Natl Acad Sci U S A. 2010;107(49):21223-1228. doi:10.1073/pnas.1010412107
8. Klin A, Lin DJ, Gorrindo P, et al. Two-year-olds with autism orient to non-social contingencies rather than biological motion. Nature. 2009;459(7244):257-261. doi:10.1038/nature07868
9. Chen T, Chen Y, Yuan M, et al. Towards developing a practical artificial intelligence tool for diagnosing and evaluating autism spectrum disorder: a study using multicenter ABIDE II datasets. JMIR Med Inform. 2020;8(5):e15767. doi:10.2196/15767
10. Maglione MA, Gans D, Das L, et al; Technical Expert Panel, & HRSA Autism Intervention Research – Behavioral (AIR‐B) Network. Nonmedical interventions for children with ASD: recommended guidelines and further research needs. Pediatrics. 2012;30(Suppl 2), S169-S178.
11. Monz BU, Houghton R, Law K, et al. Treatment patterns in children with autism in the United States. Autism Res. 2019;12(3):5170-526. doi:10.1002/aur.2070
12. Shukla DK, Keehn B, Lincoln AJ, et al. White matter compromise of callosal and subcortical fiber tracts in children with autism spectrum disorder: a diffusion tensor imaging study. J Am Acad Child Adolesc Psychiatry. 2010;49(12):1269-1278.e12782. doi:10.1016/j.jaac.2010.08.018
13. Sperdin HF, Schaer M. Aberrant development of speech processing in young children with autism: new insights from neuroimaging biomarkers. Front Neurosci. 2016;10:393. doi: 10.3389/fnins.2016.00393
14. Zwaigenbaum L, Brian JA, Ip A. Early detection for autism spectrum disorder in young children. Paediatr Child Health. 2019;24(7):424-443. doi:10.1093/pch/pxz119
15. Simms MD, Jin XM. Autism, language disorder, and social (pragmatic) communication disorder: DSM-V and differential diagnoses. Pediatr Rev. 2015;36(8):355-363. doi:10.1542/pir.36-8-355
16. Su Maw S, Haga C. Effectiveness of cognitive, developmental, and behavioural interventions for autism spectrum disorder in preschool-aged children: a systematic review and meta-analysis. Heliyon. 2018;4(9):e00763. doi:10.1016/j.heliyon.2018.e00763
17. Volkmar F, Siegel M, Woodbury-Smith M, et al. Practice parameter for the assessment and treatment of children and adolescents with autism spectrum disorder. J Am Acad Child Adolesc Psychiatry, 2014;53(2):237-257.
18. Landa RJ. Efficacy of early interventions for infants and young children with, and at risk for, autism spectrum disorders. Int Rev Psychiatry. 2018;30(1):25-39. doi:10.1080/09540261.2018.1432574
19. Lipkin PH, Macias MM; Council on Children with Disabilities, Section on Developmental and Behavioral Pediatrics. Promoting optimal development: identifying infants and young children with developmental disorders through developmental surveillance and screening. Pediatrics. 2020;145(1)e20193449. doi:10.1542/peds.2019-3449
20. Pickles A, Le Couteur A, Leadbitter K, et al. Parent-mediated social communication therapy for young children with autism (PACT): long-term follow-up of a randomised controlled trial. Lancet. 2016;388:2501-2509.
21. Rogers SJ, Estes A, Lord C, et al. Effects of a brief early start Denver model (ESDM)-based parent intervention on toddlers at risk for autism spectrum disorders: a randomized controlled trial. J Am Acad Child Adolesc Psychiatry. 2012;51(10):1052-1065. doi:10.1016/j.jaac.2012.08.003
22. Schreibman L, Dawson G, Stahmer AC, et al. Naturalistic developmental behavioral interventions: empirically validated treatments for autism spectrum disorder. J Autism Dev Disord. 2015;45(8):2411-2428. doi:10.1007/s10803-015-2407-8
23. Mundy P. A review of joint attention and social-cognitive brain systems in typical development and autism spectrum disorder. Eur J Neurosci. 2018;47(6):497-514.
24. Zwaigenbaum L, Bryson SE, Brian J, et al. Stability of diagnostic assessment for autism spectrum disorder between 18 and 36 months in a high-risk cohort. Autism Res. 2016;9(7):790-800. doi:10.1002/aur.1585
25. Anderson DK, Liang JW, Lord C. Predicting young adult outcome among more and less cognitively able individuals with autism spectrum disorders. J Child Psychol Psychiatry. 2014;55(5):485-494. doi:10.1111/jcpp.12178
26. Jones W, Carr K, Klin A. Absence of preferential looking to the eyes of approaching adults predicts level of social disability in 2-year-old toddlers with autism spectrum disorder. Arch Gen Psychiatry. 2008;65(8):946-954. doi:10.1001/archpsyc.65.8.946
27. Van der Donck S, Dzhelyova M, Vettori S, et al. Rapid neural categorization of angry and fearful faces is specifically impaired in boys with autism spectrum disorder. J Child Psychol Psychiatry. 2020;61(9):1019-1029. doi:10.1111/jcpp.13201
28. Thurm A, Farmer C, Salzman E, et al. State of the field: differentiating intellectual disability from autism spectrum disorder. Front Psychiatry. 2019;10:526. doi:10.3389/fpsyt.2019.00526
29. Kuno-Fujita A, Iwabuchi T, Wakusawa K, et al. Sensory processing patterns and fusiform activity during face processing in autism spectrum disorder. Autism Res. 2020;13(5):741-750. doi: 10.1002/aur.2283
30. Abrams DA, Lynch CJ, Cheng KM, et al. Underconnectivity between voice-selective cortex and reward circuitry in children with autism. Proc Natl Acad Sci U S A. 2013;110(29):12060-12065. doi:10.1073/pnas.1302982110
31. Osterling J, Dawson G. Early recognition of children with autism: a study of first birthday home videotapes. J Autism Dev Disord. 1994;24(3):247-257.
32. Zampella CJ, Csumitta KD, Simon E, et al. Interactional synchrony and its association with social and communication ability in children with and without autism spectrum disorder. J Autism Dev Disord. 2020;50(9):3195-3206. doi:10.1007/s10803-020-04412-8
33. McFayden T, Jarrett MA, White SW, et al. Sluggish cognitive tempo in autism spectrum disorder, ADHD, and their comorbidity: implications for impairment. J Clin Child Adolesc Psychol. 2020:1-8. doi:10.1080/15374416.2020.1716365
34. Baribeau DA, Vigod S, Pullenayegum E, et al. Repetitive behavior severity as an early indicator of risk for elevated anxiety symptoms in autism spectrum disorder. J Am Acad Child Adolesc Psychiatry. 2020;59(7):890-899.e3. doi:10.1016/j.jaac.2019.08.478
35. Li Y, Zhou Z, Chang C, et al. Anomalies in uncinate fasciculus development and social defects in preschoolers with autism spectrum disorder. BMC Psychiatry. 2019;19(1):399. doi:10.1186/s12888-019-2391-1
36. Payabvash S, Palacios EM, Owen JP, et al. White matter connectome edge density in children with autism spectrum disorders: potential imaging biomarkers using machine-learning models. Brain Connect. 2019;9(2):209-220. doi:10.1089/brain.2018.0658
37. Andrews DS, Lee JK, Solomon M, et al. A diffusion-weighted imaging tract-based spatial statistics study of autism spectrum disorder in preschool-aged children. J Neurodev Disord. 2019;11(1):32. doi:10.1186/s11689-019-9291-z
38. Chevallier C, Kohls G, Troiani V, et al. The social motivation theory of autism. Trends Cogn Sci. 2012;16(4):231-239. doi:10.1016/j.tics.2012.02.007
39. Boddaert N, Chabane N, Gervais H, et al. Superior temporal sulcus anatomical abnormalities in childhood autism: a voxel-based morphometry MRI study. Neuroimage. 2004;23(1):364-369. doi:10.1016/j.neuroimage.2004.06.016
40. Lord C, Petkova E, Hus V, et al. A multisite study of the clinical diagnosis of different autism spectrum disorders. Arch Gen Psychiatry. 2012;69(3):306-313. doi:10.1001/archgenpsychiatry.2011.148
41. Trillingsgaard A, ØStergaard JR. Autism in Angelman syndrome: an exploration of comorbidity. Autism. 2004;8(2):163-174.
42. Moss J, Howlin P. Autism spectrum disorders in genetic syndromes: implications for diagnosis, intervention and understanding the wider autism spectrum disorder population. J Intellect Disabil Res. 2009;53(10):852-873. doi:10.1111/j.1365-2788.2009.01197.x
43. McDuffie A, Thurman AJ, Hagerman RJ, et al. Symptoms of autism in males with Fragile X syndrome: a comparison to nonsyndromic ASD using current ADI-R scores. J Autism Dev Disord. 2015;45(7):1925-1937. doi:10.1007/s10803-013-2013-6
44. Ashwood KL, Tye C, Azadi B, et al. Brief report: adaptive functioning in children with ASD, ADHD and ASD + ADHD. J Autism Dev Disord. 2015;45(7):2235-4222. doi:10.1007/s10803-014-2352-y
45. Guthrie W, Wallis K, Bennett A, et al. Accuracy of autism screening in a large pediatric network. Pediatrics. 2019;144(4): e20183963. doi:10.1542/peds.2018-3963
46. Brian JA, Zwaigenbaum L, Ip A. Standards of diagnostic assessment for autism spectrum disorder. Paediatr Child Health. 2019;24(7):444-460. doi:10.1093/pch/pxz117
47. Frigaux A, Evrard R, Lighezzolo-Alnot J. ADI-R and ADOS and the differential diagnosis of autism spectrum disorders: interests, limits and openings. Encephale. 2019;45(5):441-448. doi:10.1016/j.encep.2019.07.002
48. Sacrey LR, Zwaigenbaum L, Bryson S, et al. Screening for behavioral signs of autism spectrum disorder in 9-month-old infant siblings. J Autism Dev Disord. 2021;51(3):839-848. doi:10.1007/s10803-020-04371-0
49. Sacrey LR, Bryson S, Zwaigenbaum L, et al. The autism parent screen for infants: predicting risk of autism spectrum disorder based on parent-reported behavior observed at 6-24 months of age. Autism. 2018;22(3):322-334
Differentiating pediatric schizotypal disorder from schizophrenia and autism
Schizotypal disorder is a complex condition that is characterized by cognitive-perceptual impairments, oddness, disorganization, and interpersonal difficulties. It often is unrecognized or underdiagnosed. In DSM-5, schizotypal disorder is categorized a personality disorder, but it is also considered part of the schizophrenia spectrum disorders.1 The diagnostic criteria for schizotypal disorder are outlined in the Table.1,2

Although schizotypal disorder has a lifetime prevalence of approximately 4% in the general population of the United States,2 it can present during childhood or adolescence and may be overlooked in the differential diagnosis for psychotic symptoms in pediatric patients.3 Schizotypal disorder of childhood (SDC) can present with significant overlap with several pediatric diagnoses, including schizophrenia spectrum disorders and autism spectrum disorder (ASD), all of which may include psychotic symptoms and difficulties in interpersonal relationships. This overlap, combined with the lack of awareness of schizotypal disorder, can pose a diagnostic challenge. Better recognition of SDC could result in earlier and more effective treatment. In this article, we provide tips for differentiating SDC from childhood-onset schizophrenia and from ASD.
Differentiating SDC from schizophrenia
SDC may be mistaken for childhood-onset schizophrenia due to its perceptual disturbances (which may be interpreted as visual or auditory hallucinations), bizarre fantasies (which may be mistaken for overt delusions), paranoia, and odd behavior. Two ways to distinguish SDC from childhood schizophrenia are by clinical course and by severity of negative psychotic symptoms.
SDC tends to have an overall stable clinical course,4 with patients experiencing periods of time when they exhibit a more normal mental status complemented by fluctuations in symptom severity, which are exacerbated by stressors and followed by a return to baseline.3 SDC psychotic symptoms are predominantly positive, and patients typically do not demonstrate negative features beyond social difficulties. Childhood-onset schizophrenia is typically progressive and disabling, with worsening severity over time, and is much more likely to incorporate prominent negative symptoms.3
Differentiating SDC from ASD
SDC also demonstrates considerable diagnostic overlap with ASD, especially with regards to inappropriate affect; odd thinking, behavior, and speech; and social difficulties. Further complicating the diagnosis, ASD and SDC are comorbid in approximately 40% of ASD cases.3,5 The Melbourne Assessment of Schizotypy in Kids demonstrates validity in diagnosing schizotypal disorder in patients with comorbid ASD.5,6 For clinicians without easy access to advanced testing, 2 ways to distinguish SDC from ASD are the content of the odd behavior and thoughts, and the patient’s reaction to social deficits.
In SDC, odd behavior and thoughts most often revolve around daydreaming and a focus on “elaborate inner fantasies.”3,6 Unlike in ASD, in patients with SDC, behaviors don’t typically involve stereotyped mannerisms, the patient is unlikely to have rigid interests (apart from their fantasies), and there is not a particular focus on detail in the external world.3,6 Notably, imaginary companions are common in SDC; children with ASD are less likely to have an imaginary companion compared with children with SDC or those with no psychiatric diagnosis.6 Patients with SDC have social difficulties (often due to social anxiety stemming from their paranoia) but usually seek out interaction and are bothered by alienation, while patients with ASD may have less interest in social engagement.6
1. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. American Psychiatric Association; 2013.
2. Pulay AJ, Stinson FS, Dawson DA, et al. Prevalence, correlates, disability, and comorbidity of DSM-IV schizotypal personality disorder: results from the wave 2 national epidemiologic survey on alcohol and related conditions. Prim Care Companion J Clin Psychiatry. 2009;11(2):53-67. doi:10.4088/pcc.08m00679
3. Tonge BJ, Testa R, Díaz-Arteche C, et al. Schizotypal disorder in children—a neglected diagnosis. Schizophrenia Bulletin Open. 2020;1(1):sgaa048. doi:10.1093/schizbullopen/sgaa048
4. Asarnow JR. Childhood-onset schizotypal disorder: a follow-up study and comparison with childhood-onset schizophrenia. J Child Adolesc Psychopharmacol. 2005;15(3):395-402.
5. Jones HP, Testa RR, Ross N, et al. The Melbourne Assessment of Schizotypy in Kids: a useful measure of childhood schizotypal personality disorder. Biomed Res Int. 2015;2015:635732. doi:10.1155/2015/635732
6. Poletti M, Raballo A. Childhood schizotypal features vs. high-functioning autism spectrum disorder: developmental overlaps and phenomenological differences. Schizophr Res. 2020;223:53-58. doi:10.1016/j.schres.2020.09.027
Schizotypal disorder is a complex condition that is characterized by cognitive-perceptual impairments, oddness, disorganization, and interpersonal difficulties. It often is unrecognized or underdiagnosed. In DSM-5, schizotypal disorder is categorized a personality disorder, but it is also considered part of the schizophrenia spectrum disorders.1 The diagnostic criteria for schizotypal disorder are outlined in the Table.1,2

Although schizotypal disorder has a lifetime prevalence of approximately 4% in the general population of the United States,2 it can present during childhood or adolescence and may be overlooked in the differential diagnosis for psychotic symptoms in pediatric patients.3 Schizotypal disorder of childhood (SDC) can present with significant overlap with several pediatric diagnoses, including schizophrenia spectrum disorders and autism spectrum disorder (ASD), all of which may include psychotic symptoms and difficulties in interpersonal relationships. This overlap, combined with the lack of awareness of schizotypal disorder, can pose a diagnostic challenge. Better recognition of SDC could result in earlier and more effective treatment. In this article, we provide tips for differentiating SDC from childhood-onset schizophrenia and from ASD.
Differentiating SDC from schizophrenia
SDC may be mistaken for childhood-onset schizophrenia due to its perceptual disturbances (which may be interpreted as visual or auditory hallucinations), bizarre fantasies (which may be mistaken for overt delusions), paranoia, and odd behavior. Two ways to distinguish SDC from childhood schizophrenia are by clinical course and by severity of negative psychotic symptoms.
SDC tends to have an overall stable clinical course,4 with patients experiencing periods of time when they exhibit a more normal mental status complemented by fluctuations in symptom severity, which are exacerbated by stressors and followed by a return to baseline.3 SDC psychotic symptoms are predominantly positive, and patients typically do not demonstrate negative features beyond social difficulties. Childhood-onset schizophrenia is typically progressive and disabling, with worsening severity over time, and is much more likely to incorporate prominent negative symptoms.3
Differentiating SDC from ASD
SDC also demonstrates considerable diagnostic overlap with ASD, especially with regards to inappropriate affect; odd thinking, behavior, and speech; and social difficulties. Further complicating the diagnosis, ASD and SDC are comorbid in approximately 40% of ASD cases.3,5 The Melbourne Assessment of Schizotypy in Kids demonstrates validity in diagnosing schizotypal disorder in patients with comorbid ASD.5,6 For clinicians without easy access to advanced testing, 2 ways to distinguish SDC from ASD are the content of the odd behavior and thoughts, and the patient’s reaction to social deficits.
In SDC, odd behavior and thoughts most often revolve around daydreaming and a focus on “elaborate inner fantasies.”3,6 Unlike in ASD, in patients with SDC, behaviors don’t typically involve stereotyped mannerisms, the patient is unlikely to have rigid interests (apart from their fantasies), and there is not a particular focus on detail in the external world.3,6 Notably, imaginary companions are common in SDC; children with ASD are less likely to have an imaginary companion compared with children with SDC or those with no psychiatric diagnosis.6 Patients with SDC have social difficulties (often due to social anxiety stemming from their paranoia) but usually seek out interaction and are bothered by alienation, while patients with ASD may have less interest in social engagement.6
Schizotypal disorder is a complex condition that is characterized by cognitive-perceptual impairments, oddness, disorganization, and interpersonal difficulties. It often is unrecognized or underdiagnosed. In DSM-5, schizotypal disorder is categorized a personality disorder, but it is also considered part of the schizophrenia spectrum disorders.1 The diagnostic criteria for schizotypal disorder are outlined in the Table.1,2

Although schizotypal disorder has a lifetime prevalence of approximately 4% in the general population of the United States,2 it can present during childhood or adolescence and may be overlooked in the differential diagnosis for psychotic symptoms in pediatric patients.3 Schizotypal disorder of childhood (SDC) can present with significant overlap with several pediatric diagnoses, including schizophrenia spectrum disorders and autism spectrum disorder (ASD), all of which may include psychotic symptoms and difficulties in interpersonal relationships. This overlap, combined with the lack of awareness of schizotypal disorder, can pose a diagnostic challenge. Better recognition of SDC could result in earlier and more effective treatment. In this article, we provide tips for differentiating SDC from childhood-onset schizophrenia and from ASD.
Differentiating SDC from schizophrenia
SDC may be mistaken for childhood-onset schizophrenia due to its perceptual disturbances (which may be interpreted as visual or auditory hallucinations), bizarre fantasies (which may be mistaken for overt delusions), paranoia, and odd behavior. Two ways to distinguish SDC from childhood schizophrenia are by clinical course and by severity of negative psychotic symptoms.
SDC tends to have an overall stable clinical course,4 with patients experiencing periods of time when they exhibit a more normal mental status complemented by fluctuations in symptom severity, which are exacerbated by stressors and followed by a return to baseline.3 SDC psychotic symptoms are predominantly positive, and patients typically do not demonstrate negative features beyond social difficulties. Childhood-onset schizophrenia is typically progressive and disabling, with worsening severity over time, and is much more likely to incorporate prominent negative symptoms.3
Differentiating SDC from ASD
SDC also demonstrates considerable diagnostic overlap with ASD, especially with regards to inappropriate affect; odd thinking, behavior, and speech; and social difficulties. Further complicating the diagnosis, ASD and SDC are comorbid in approximately 40% of ASD cases.3,5 The Melbourne Assessment of Schizotypy in Kids demonstrates validity in diagnosing schizotypal disorder in patients with comorbid ASD.5,6 For clinicians without easy access to advanced testing, 2 ways to distinguish SDC from ASD are the content of the odd behavior and thoughts, and the patient’s reaction to social deficits.
In SDC, odd behavior and thoughts most often revolve around daydreaming and a focus on “elaborate inner fantasies.”3,6 Unlike in ASD, in patients with SDC, behaviors don’t typically involve stereotyped mannerisms, the patient is unlikely to have rigid interests (apart from their fantasies), and there is not a particular focus on detail in the external world.3,6 Notably, imaginary companions are common in SDC; children with ASD are less likely to have an imaginary companion compared with children with SDC or those with no psychiatric diagnosis.6 Patients with SDC have social difficulties (often due to social anxiety stemming from their paranoia) but usually seek out interaction and are bothered by alienation, while patients with ASD may have less interest in social engagement.6
1. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. American Psychiatric Association; 2013.
2. Pulay AJ, Stinson FS, Dawson DA, et al. Prevalence, correlates, disability, and comorbidity of DSM-IV schizotypal personality disorder: results from the wave 2 national epidemiologic survey on alcohol and related conditions. Prim Care Companion J Clin Psychiatry. 2009;11(2):53-67. doi:10.4088/pcc.08m00679
3. Tonge BJ, Testa R, Díaz-Arteche C, et al. Schizotypal disorder in children—a neglected diagnosis. Schizophrenia Bulletin Open. 2020;1(1):sgaa048. doi:10.1093/schizbullopen/sgaa048
4. Asarnow JR. Childhood-onset schizotypal disorder: a follow-up study and comparison with childhood-onset schizophrenia. J Child Adolesc Psychopharmacol. 2005;15(3):395-402.
5. Jones HP, Testa RR, Ross N, et al. The Melbourne Assessment of Schizotypy in Kids: a useful measure of childhood schizotypal personality disorder. Biomed Res Int. 2015;2015:635732. doi:10.1155/2015/635732
6. Poletti M, Raballo A. Childhood schizotypal features vs. high-functioning autism spectrum disorder: developmental overlaps and phenomenological differences. Schizophr Res. 2020;223:53-58. doi:10.1016/j.schres.2020.09.027
1. Diagnostic and Statistical Manual of Mental Disorders: DSM-5. 5th ed. American Psychiatric Association; 2013.
2. Pulay AJ, Stinson FS, Dawson DA, et al. Prevalence, correlates, disability, and comorbidity of DSM-IV schizotypal personality disorder: results from the wave 2 national epidemiologic survey on alcohol and related conditions. Prim Care Companion J Clin Psychiatry. 2009;11(2):53-67. doi:10.4088/pcc.08m00679
3. Tonge BJ, Testa R, Díaz-Arteche C, et al. Schizotypal disorder in children—a neglected diagnosis. Schizophrenia Bulletin Open. 2020;1(1):sgaa048. doi:10.1093/schizbullopen/sgaa048
4. Asarnow JR. Childhood-onset schizotypal disorder: a follow-up study and comparison with childhood-onset schizophrenia. J Child Adolesc Psychopharmacol. 2005;15(3):395-402.
5. Jones HP, Testa RR, Ross N, et al. The Melbourne Assessment of Schizotypy in Kids: a useful measure of childhood schizotypal personality disorder. Biomed Res Int. 2015;2015:635732. doi:10.1155/2015/635732
6. Poletti M, Raballo A. Childhood schizotypal features vs. high-functioning autism spectrum disorder: developmental overlaps and phenomenological differences. Schizophr Res. 2020;223:53-58. doi:10.1016/j.schres.2020.09.027
How to say ‘no’ to inappropriate patient requests
Although we may want to say “yes” when our patients ask us for certain medications, work excuses, etc, often it is more appropriate to say “no” because the conditions do not support those requests. Saying no to a patient usually is not a comfortable experience, but we should not say yes to avoid hurting their feelings, damaging our rapport with them, or having them post potential negative reviews about us. For many of us, saying no is a skill that does not come naturally. For some, bluntly telling a patient no may work, but this approach is more likely to be ineffective. At the same time, saying no in an equivocal manner may weaken our patients’ confidence in us and could be displeasing for both our patients and us.1,2
We should say no in an “effective, professional manner that fosters good patient care and preserves the therapeutic relationship, while supporting physician well-being.”1 In this article, I provide practical tips for saying no to inappropriate patient requests in an emphatic manner so that we can feel more empowered and less uncomfortable.
Acknowledge and analyze your discomfort.
Before saying no, recognize that you are feeling uncomfortable with your patient’s inappropriate request. This uncomfortable feeling is a probable cue that there is likely no appropriate context for their request, ie, saying yes would be poor medical care, illegal, against policy, etc.1,3 In most cases, you should be able to identify the reason(s) your patient’s request feels inappropriate and uncomfortable.
Gather information and provide an explanation.
Ask your patient for more information about their request so you can determine if there are any underlying factors and if any additional information is needed.3 Once you decide to say no, explain why. Your explanation should be brief, because lengthy explanations might create room for debate (which could be exhausting and/or time-consuming), lead to giving in to their inappropriate request, and/or lead them to become more frustrated and misunderstood.1
Be empathetic, and re-establish rapport.
After declining a patient’s request, you may have to use empathy to re-establish rapport if it has been damaged. After being told no, your patient may feel frustrated or powerless. Acknowledge their feelings with statements such as “I know this is not want you wanted to hear” or “I can see you are irritated.”Accept your patient’s negative emotions, rather than minimizing them or trying to fix them.1,3
1. Kane M, Chambliss ML. Getting to no: how to respond to inappropriate patient requests. Fam Prac Manag. 2018;25(1):25-30.
2. Paterniti DA, Facher TL, Cipri CS, et al. Getting to “no”: strategies primary care physicians use to deny patient requests. Arch Intern Med. 2010;170(4):381-388.
3. Huben-Kearney A. Just say no to certain patient requests—and here’s how. Psychiatric News. 2021;56(2):13.
Although we may want to say “yes” when our patients ask us for certain medications, work excuses, etc, often it is more appropriate to say “no” because the conditions do not support those requests. Saying no to a patient usually is not a comfortable experience, but we should not say yes to avoid hurting their feelings, damaging our rapport with them, or having them post potential negative reviews about us. For many of us, saying no is a skill that does not come naturally. For some, bluntly telling a patient no may work, but this approach is more likely to be ineffective. At the same time, saying no in an equivocal manner may weaken our patients’ confidence in us and could be displeasing for both our patients and us.1,2
We should say no in an “effective, professional manner that fosters good patient care and preserves the therapeutic relationship, while supporting physician well-being.”1 In this article, I provide practical tips for saying no to inappropriate patient requests in an emphatic manner so that we can feel more empowered and less uncomfortable.
Acknowledge and analyze your discomfort.
Before saying no, recognize that you are feeling uncomfortable with your patient’s inappropriate request. This uncomfortable feeling is a probable cue that there is likely no appropriate context for their request, ie, saying yes would be poor medical care, illegal, against policy, etc.1,3 In most cases, you should be able to identify the reason(s) your patient’s request feels inappropriate and uncomfortable.
Gather information and provide an explanation.
Ask your patient for more information about their request so you can determine if there are any underlying factors and if any additional information is needed.3 Once you decide to say no, explain why. Your explanation should be brief, because lengthy explanations might create room for debate (which could be exhausting and/or time-consuming), lead to giving in to their inappropriate request, and/or lead them to become more frustrated and misunderstood.1
Be empathetic, and re-establish rapport.
After declining a patient’s request, you may have to use empathy to re-establish rapport if it has been damaged. After being told no, your patient may feel frustrated or powerless. Acknowledge their feelings with statements such as “I know this is not want you wanted to hear” or “I can see you are irritated.”Accept your patient’s negative emotions, rather than minimizing them or trying to fix them.1,3
Although we may want to say “yes” when our patients ask us for certain medications, work excuses, etc, often it is more appropriate to say “no” because the conditions do not support those requests. Saying no to a patient usually is not a comfortable experience, but we should not say yes to avoid hurting their feelings, damaging our rapport with them, or having them post potential negative reviews about us. For many of us, saying no is a skill that does not come naturally. For some, bluntly telling a patient no may work, but this approach is more likely to be ineffective. At the same time, saying no in an equivocal manner may weaken our patients’ confidence in us and could be displeasing for both our patients and us.1,2
We should say no in an “effective, professional manner that fosters good patient care and preserves the therapeutic relationship, while supporting physician well-being.”1 In this article, I provide practical tips for saying no to inappropriate patient requests in an emphatic manner so that we can feel more empowered and less uncomfortable.
Acknowledge and analyze your discomfort.
Before saying no, recognize that you are feeling uncomfortable with your patient’s inappropriate request. This uncomfortable feeling is a probable cue that there is likely no appropriate context for their request, ie, saying yes would be poor medical care, illegal, against policy, etc.1,3 In most cases, you should be able to identify the reason(s) your patient’s request feels inappropriate and uncomfortable.
Gather information and provide an explanation.
Ask your patient for more information about their request so you can determine if there are any underlying factors and if any additional information is needed.3 Once you decide to say no, explain why. Your explanation should be brief, because lengthy explanations might create room for debate (which could be exhausting and/or time-consuming), lead to giving in to their inappropriate request, and/or lead them to become more frustrated and misunderstood.1
Be empathetic, and re-establish rapport.
After declining a patient’s request, you may have to use empathy to re-establish rapport if it has been damaged. After being told no, your patient may feel frustrated or powerless. Acknowledge their feelings with statements such as “I know this is not want you wanted to hear” or “I can see you are irritated.”Accept your patient’s negative emotions, rather than minimizing them or trying to fix them.1,3
1. Kane M, Chambliss ML. Getting to no: how to respond to inappropriate patient requests. Fam Prac Manag. 2018;25(1):25-30.
2. Paterniti DA, Facher TL, Cipri CS, et al. Getting to “no”: strategies primary care physicians use to deny patient requests. Arch Intern Med. 2010;170(4):381-388.
3. Huben-Kearney A. Just say no to certain patient requests—and here’s how. Psychiatric News. 2021;56(2):13.
1. Kane M, Chambliss ML. Getting to no: how to respond to inappropriate patient requests. Fam Prac Manag. 2018;25(1):25-30.
2. Paterniti DA, Facher TL, Cipri CS, et al. Getting to “no”: strategies primary care physicians use to deny patient requests. Arch Intern Med. 2010;170(4):381-388.
3. Huben-Kearney A. Just say no to certain patient requests—and here’s how. Psychiatric News. 2021;56(2):13.
Closing your practice: What to consider
Closing your practice can be a stressful experience, and it requires careful planning. The process requires numerous steps, such as informing your staff, notifying your patients, closing accounts with your vendors and suppliers, storing medical records, and following applicable federal and state laws for dissolving your practice.1,2 Many of these steps may require consulting with an attorney, an accountant, and your malpractice insurance carrier.1,2 Although the recommendations I provide in this article are not exhaustive, when faced with closing your practice, be sure to consider the following factors.
Notify staff and patients.
Select a date to close your practice that will allow you to stop taking new patients, provides adequate leeway for your staff to find new employment and for you to hire temporary staff if needed, ensures you meet your obligations to your staff, such as payroll, and gives you time to set up appropriate continuity of care for your patients. In addition to verbally notifying your patients of your practice’s closing, inform them in writing (whether hand-delivered or via certified mail with return receipt) of the date of the practice’s closure, reason for the closure, cancellation of scheduled appointments after the closure date, referral options, and how they can obtain a copy of their medical records.1,2 Make sure your patients have an adequate supply of their medications before the closure.
Notify other parties.
Inform all suppliers, vendors, contracted service providers, insurance broker(s) for your practice, and payers (including Medicare and Medicaid, if applicable) of your intent to close your practice.1,2 Provide payers with a forwarding address to send payments that resolve after your practice closes, and request final invoices from vendors and suppliers so you can close your accounts with them. If you don’t own the building in which your practice is located, notify the building management in accordance with the provisions of your lease.1,2 Give cancellation notices to utilities and ancillary services (eg, labs, imaging facilities) to which you refer your patients, and notify facilities where you are credentialed and have admitting privileges.1,2 Inform your state medical licensing board, your state’s controlled substance division, and the Drug Enforcement Administration, because these agencies have requirements regarding changing the status of your medical license (if you decide to retire), continuing or surrendering your state and federal controlled substance registration, and disposal of prescription medications and prescription pads.1,2 Contact your local post office and delivery services with your change of address.
Address other considerations.
Set up a medical record retention and destruction plan in accordance with state and federal regulations, arrange for the safe storage for both paper and electronic medical records, and make sure storage facilities have experience handling confidential, Health Insurance Portability and Accountability Act (HIPAA)-sensitive patient information.1,2 In addition, establish a process for permanently deleting all HIPAA-sensitive patient information from any equipment that you don’t intend to keep.1,2
1. Funicelli AM. Risk management checklist when closing your practice. Psychiatric News. 2020;55(23):11.
2. American Academy of Family Physicians. Closing your practice checklist. Accessed January 21, 2022. https://www.aafp.org/dam/AAFP/documents/practice_management/admin_staffing/ClosingPracticeChecklist.pdf
Closing your practice can be a stressful experience, and it requires careful planning. The process requires numerous steps, such as informing your staff, notifying your patients, closing accounts with your vendors and suppliers, storing medical records, and following applicable federal and state laws for dissolving your practice.1,2 Many of these steps may require consulting with an attorney, an accountant, and your malpractice insurance carrier.1,2 Although the recommendations I provide in this article are not exhaustive, when faced with closing your practice, be sure to consider the following factors.
Notify staff and patients.
Select a date to close your practice that will allow you to stop taking new patients, provides adequate leeway for your staff to find new employment and for you to hire temporary staff if needed, ensures you meet your obligations to your staff, such as payroll, and gives you time to set up appropriate continuity of care for your patients. In addition to verbally notifying your patients of your practice’s closing, inform them in writing (whether hand-delivered or via certified mail with return receipt) of the date of the practice’s closure, reason for the closure, cancellation of scheduled appointments after the closure date, referral options, and how they can obtain a copy of their medical records.1,2 Make sure your patients have an adequate supply of their medications before the closure.
Notify other parties.
Inform all suppliers, vendors, contracted service providers, insurance broker(s) for your practice, and payers (including Medicare and Medicaid, if applicable) of your intent to close your practice.1,2 Provide payers with a forwarding address to send payments that resolve after your practice closes, and request final invoices from vendors and suppliers so you can close your accounts with them. If you don’t own the building in which your practice is located, notify the building management in accordance with the provisions of your lease.1,2 Give cancellation notices to utilities and ancillary services (eg, labs, imaging facilities) to which you refer your patients, and notify facilities where you are credentialed and have admitting privileges.1,2 Inform your state medical licensing board, your state’s controlled substance division, and the Drug Enforcement Administration, because these agencies have requirements regarding changing the status of your medical license (if you decide to retire), continuing or surrendering your state and federal controlled substance registration, and disposal of prescription medications and prescription pads.1,2 Contact your local post office and delivery services with your change of address.
Address other considerations.
Set up a medical record retention and destruction plan in accordance with state and federal regulations, arrange for the safe storage for both paper and electronic medical records, and make sure storage facilities have experience handling confidential, Health Insurance Portability and Accountability Act (HIPAA)-sensitive patient information.1,2 In addition, establish a process for permanently deleting all HIPAA-sensitive patient information from any equipment that you don’t intend to keep.1,2
Closing your practice can be a stressful experience, and it requires careful planning. The process requires numerous steps, such as informing your staff, notifying your patients, closing accounts with your vendors and suppliers, storing medical records, and following applicable federal and state laws for dissolving your practice.1,2 Many of these steps may require consulting with an attorney, an accountant, and your malpractice insurance carrier.1,2 Although the recommendations I provide in this article are not exhaustive, when faced with closing your practice, be sure to consider the following factors.
Notify staff and patients.
Select a date to close your practice that will allow you to stop taking new patients, provides adequate leeway for your staff to find new employment and for you to hire temporary staff if needed, ensures you meet your obligations to your staff, such as payroll, and gives you time to set up appropriate continuity of care for your patients. In addition to verbally notifying your patients of your practice’s closing, inform them in writing (whether hand-delivered or via certified mail with return receipt) of the date of the practice’s closure, reason for the closure, cancellation of scheduled appointments after the closure date, referral options, and how they can obtain a copy of their medical records.1,2 Make sure your patients have an adequate supply of their medications before the closure.
Notify other parties.
Inform all suppliers, vendors, contracted service providers, insurance broker(s) for your practice, and payers (including Medicare and Medicaid, if applicable) of your intent to close your practice.1,2 Provide payers with a forwarding address to send payments that resolve after your practice closes, and request final invoices from vendors and suppliers so you can close your accounts with them. If you don’t own the building in which your practice is located, notify the building management in accordance with the provisions of your lease.1,2 Give cancellation notices to utilities and ancillary services (eg, labs, imaging facilities) to which you refer your patients, and notify facilities where you are credentialed and have admitting privileges.1,2 Inform your state medical licensing board, your state’s controlled substance division, and the Drug Enforcement Administration, because these agencies have requirements regarding changing the status of your medical license (if you decide to retire), continuing or surrendering your state and federal controlled substance registration, and disposal of prescription medications and prescription pads.1,2 Contact your local post office and delivery services with your change of address.
Address other considerations.
Set up a medical record retention and destruction plan in accordance with state and federal regulations, arrange for the safe storage for both paper and electronic medical records, and make sure storage facilities have experience handling confidential, Health Insurance Portability and Accountability Act (HIPAA)-sensitive patient information.1,2 In addition, establish a process for permanently deleting all HIPAA-sensitive patient information from any equipment that you don’t intend to keep.1,2
1. Funicelli AM. Risk management checklist when closing your practice. Psychiatric News. 2020;55(23):11.
2. American Academy of Family Physicians. Closing your practice checklist. Accessed January 21, 2022. https://www.aafp.org/dam/AAFP/documents/practice_management/admin_staffing/ClosingPracticeChecklist.pdf
1. Funicelli AM. Risk management checklist when closing your practice. Psychiatric News. 2020;55(23):11.
2. American Academy of Family Physicians. Closing your practice checklist. Accessed January 21, 2022. https://www.aafp.org/dam/AAFP/documents/practice_management/admin_staffing/ClosingPracticeChecklist.pdf
What's your diagnosis?
Mucous membrane pemphigoid with esophageal web stricture.
Additional laboratory examination showed that his serum anti-BP180 antibody level was high (11.7 U/mL; normal range, <9.0 U/mL). Biopsy specimens taken from the laryngopharyngeal erosion showed subepithelial blister formation and it was consistent with pemphigoid pathologically (Figure D). He did not have cutaneous lesions and was diagnosed with mucous membrane pemphigoid (MMP). After performing endoscopic dilation, prednisolone (20 mg/d) was administered orally. Three months after starting the prednisolone treatment, follow-up endoscopy showed improvements of the laryngopharyngeal erosions (Figure E) and esophageal blister on the web. However, esophageal narrowing remained, and thus endoscopic balloon dilation was performed (Figure F-H). Three months after the dilation, the narrowing improved (Figure I).
MMP is an autoimmune blistering disease that induces the formation of mucous membrane subepithelial bullae. Basement membrane zone components such as collagen XVII (also known as BP180) are targets of autoantibodies in MMP. Symptomatic esophageal involvement affects 5.4% of patients with MMP and dysphagia is the most frequent symptom.1 Endoscopic findings include erosion, web stricture, subepithelial hematomas, and scars.2,3 Endoscopic dilation is sometimes necessary for the treatment of severe esophageal strictures.1
References
1. Zehou O et al. Br J Dermatol. 2017 Oct;177(4):1074-85.
2. Sallout H et al. Gastrointest Endosc. 2000 Sep;52(3):429-33.
3. Gaspar R et al. Gastrointest Endosc. 2017 Aug;86(2):400-2.
Mucous membrane pemphigoid with esophageal web stricture.
Additional laboratory examination showed that his serum anti-BP180 antibody level was high (11.7 U/mL; normal range, <9.0 U/mL). Biopsy specimens taken from the laryngopharyngeal erosion showed subepithelial blister formation and it was consistent with pemphigoid pathologically (Figure D). He did not have cutaneous lesions and was diagnosed with mucous membrane pemphigoid (MMP). After performing endoscopic dilation, prednisolone (20 mg/d) was administered orally. Three months after starting the prednisolone treatment, follow-up endoscopy showed improvements of the laryngopharyngeal erosions (Figure E) and esophageal blister on the web. However, esophageal narrowing remained, and thus endoscopic balloon dilation was performed (Figure F-H). Three months after the dilation, the narrowing improved (Figure I).

MMP is an autoimmune blistering disease that induces the formation of mucous membrane subepithelial bullae. Basement membrane zone components such as collagen XVII (also known as BP180) are targets of autoantibodies in MMP. Symptomatic esophageal involvement affects 5.4% of patients with MMP and dysphagia is the most frequent symptom.1 Endoscopic findings include erosion, web stricture, subepithelial hematomas, and scars.2,3 Endoscopic dilation is sometimes necessary for the treatment of severe esophageal strictures.1
References
1. Zehou O et al. Br J Dermatol. 2017 Oct;177(4):1074-85.
2. Sallout H et al. Gastrointest Endosc. 2000 Sep;52(3):429-33.
3. Gaspar R et al. Gastrointest Endosc. 2017 Aug;86(2):400-2.
Mucous membrane pemphigoid with esophageal web stricture.
Additional laboratory examination showed that his serum anti-BP180 antibody level was high (11.7 U/mL; normal range, <9.0 U/mL). Biopsy specimens taken from the laryngopharyngeal erosion showed subepithelial blister formation and it was consistent with pemphigoid pathologically (Figure D). He did not have cutaneous lesions and was diagnosed with mucous membrane pemphigoid (MMP). After performing endoscopic dilation, prednisolone (20 mg/d) was administered orally. Three months after starting the prednisolone treatment, follow-up endoscopy showed improvements of the laryngopharyngeal erosions (Figure E) and esophageal blister on the web. However, esophageal narrowing remained, and thus endoscopic balloon dilation was performed (Figure F-H). Three months after the dilation, the narrowing improved (Figure I).

MMP is an autoimmune blistering disease that induces the formation of mucous membrane subepithelial bullae. Basement membrane zone components such as collagen XVII (also known as BP180) are targets of autoantibodies in MMP. Symptomatic esophageal involvement affects 5.4% of patients with MMP and dysphagia is the most frequent symptom.1 Endoscopic findings include erosion, web stricture, subepithelial hematomas, and scars.2,3 Endoscopic dilation is sometimes necessary for the treatment of severe esophageal strictures.1
References
1. Zehou O et al. Br J Dermatol. 2017 Oct;177(4):1074-85.
2. Sallout H et al. Gastrointest Endosc. 2000 Sep;52(3):429-33.
3. Gaspar R et al. Gastrointest Endosc. 2017 Aug;86(2):400-2.
A 70-year-old man with a history of rectal cancer was referred to our clinic for chronic dysphagia and odynophagia. He did not have fevers or an allergic history. Physical examination was unremarkable except for multiple erosions in the oral cavity. Upper gastrointestinal endoscopy revealed multiple erosions in the palate and laryngopharynx (Figure A), a web stricture in the cervical esophagus (Figure B), and multiple scars in the thoracic esophagus.
Laboratory examination showed normal results including a normal white blood cell count (8010/mcL; eosinophils 360/mcL), hemoglobin level (14.0 g/dL), mean corpuscular volume (97.8 fL), serum iron level (140 mcg/dL), and ferritin level (50.5 mg/L). His dysphagia gradually worsened and he finally could not take pills nor solid food. Two weeks after the first endoscopy, a second endoscopic examination was performed and it showed exacerbation of esophageal stricture and appearance of a bloody blister (Figure C).
What is the diagnosis?
Previously published in Gastroenterology
The art of saying 'I don't know'
My wife and I had been married for 3 or 4 years when I became aware that a small cluster of my new in-laws had taken to referring to me as “Dr. I-don’t-know.” It wasn’t hard to figure out how I had earned this potentially derogatory moniker. As the only physician in the family it was not unusual for me to be peppered with medical questions. Most were unanswerable, at least by me. For example, “Will, how does aspirin work?” – which in the 1970s wasn’t something covered in medical school. Other questions were asked in a context that made it clear my answer was going to be so far removed from the preformed opinion of the questioner that Thanksgiving dinner didn’t feel like an appropriate occasion for my answer. “I don’t think I really know,” seemed to make the most sense.
In those early growing years of my outpatient general practice my in-laws weren’t the only people who must have thought of me as “Dr. I-don’t-know.” My training took place in well-thought-of teaching hospitals and during my senior residency and military tour I did enough moonlighting that by the time I entered private practice I had logged a lot of hours in the trenches. But, there were still a ton of things I didn’t know.
You probably remember how those first few years on the outside of the ivory towers felt with no one handy to ask. Even if there was someone a phone call away you didn’t want to appear as incompetent as you were by telling the patient or family that you needed to call the department head at your training program.
So, what did you do? You called. But you developed some clever language that could buy you time while you called your old mentor or hit the books. There was no Internet. Generally, that script would start with some version of “I don’t know, but ...”
As time passed and you gained more experience there were fewer questions and situations in which you needed to admit you were a few clues short of the answer. However, still, many times a day, you honestly didn’t know the answer. Hopefully, over time, you had perfected your delivery so that revealing your ignorance wasn’t driving patients away.
The art of saying “I don’t know” boils down to what you say after the “but” and how you say it. As long as you have offered a plan to find the answer and demonstrate that you are concerned about the patient, your ignorance will be tolerated and maybe even be appreciated.
“I don’t know the answer to that question, but let’s see if we can find a solution” could cover it. If finding that solution is going to require more time than the office visit allows – which it usually doesn’t – the promise of a timely response and a commitment to keep that promise is an absolute requirement. Repeated failure to keep promises is one of the quickest ways to sour a patient-doctor relationship and a potential practice killer.
I think my in-laws no longer refer to me as Dr. I-don’t-know. At least they have refrained from asking me any medical questions. But, my reputation as a physician unafraid to admit his ignorance continues here in town. Occasionally I encounter a parent of a former patient who fondly recalls my willingness to say “I don’t know.” If we had a family crest I would like it to include the motto “Ignoramus Sed Pertinet” (We don’t know but we care).
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
The headline on this article was updated on 2/28/22.
My wife and I had been married for 3 or 4 years when I became aware that a small cluster of my new in-laws had taken to referring to me as “Dr. I-don’t-know.” It wasn’t hard to figure out how I had earned this potentially derogatory moniker. As the only physician in the family it was not unusual for me to be peppered with medical questions. Most were unanswerable, at least by me. For example, “Will, how does aspirin work?” – which in the 1970s wasn’t something covered in medical school. Other questions were asked in a context that made it clear my answer was going to be so far removed from the preformed opinion of the questioner that Thanksgiving dinner didn’t feel like an appropriate occasion for my answer. “I don’t think I really know,” seemed to make the most sense.
In those early growing years of my outpatient general practice my in-laws weren’t the only people who must have thought of me as “Dr. I-don’t-know.” My training took place in well-thought-of teaching hospitals and during my senior residency and military tour I did enough moonlighting that by the time I entered private practice I had logged a lot of hours in the trenches. But, there were still a ton of things I didn’t know.
You probably remember how those first few years on the outside of the ivory towers felt with no one handy to ask. Even if there was someone a phone call away you didn’t want to appear as incompetent as you were by telling the patient or family that you needed to call the department head at your training program.
So, what did you do? You called. But you developed some clever language that could buy you time while you called your old mentor or hit the books. There was no Internet. Generally, that script would start with some version of “I don’t know, but ...”
As time passed and you gained more experience there were fewer questions and situations in which you needed to admit you were a few clues short of the answer. However, still, many times a day, you honestly didn’t know the answer. Hopefully, over time, you had perfected your delivery so that revealing your ignorance wasn’t driving patients away.
The art of saying “I don’t know” boils down to what you say after the “but” and how you say it. As long as you have offered a plan to find the answer and demonstrate that you are concerned about the patient, your ignorance will be tolerated and maybe even be appreciated.
“I don’t know the answer to that question, but let’s see if we can find a solution” could cover it. If finding that solution is going to require more time than the office visit allows – which it usually doesn’t – the promise of a timely response and a commitment to keep that promise is an absolute requirement. Repeated failure to keep promises is one of the quickest ways to sour a patient-doctor relationship and a potential practice killer.
I think my in-laws no longer refer to me as Dr. I-don’t-know. At least they have refrained from asking me any medical questions. But, my reputation as a physician unafraid to admit his ignorance continues here in town. Occasionally I encounter a parent of a former patient who fondly recalls my willingness to say “I don’t know.” If we had a family crest I would like it to include the motto “Ignoramus Sed Pertinet” (We don’t know but we care).
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
The headline on this article was updated on 2/28/22.
My wife and I had been married for 3 or 4 years when I became aware that a small cluster of my new in-laws had taken to referring to me as “Dr. I-don’t-know.” It wasn’t hard to figure out how I had earned this potentially derogatory moniker. As the only physician in the family it was not unusual for me to be peppered with medical questions. Most were unanswerable, at least by me. For example, “Will, how does aspirin work?” – which in the 1970s wasn’t something covered in medical school. Other questions were asked in a context that made it clear my answer was going to be so far removed from the preformed opinion of the questioner that Thanksgiving dinner didn’t feel like an appropriate occasion for my answer. “I don’t think I really know,” seemed to make the most sense.
In those early growing years of my outpatient general practice my in-laws weren’t the only people who must have thought of me as “Dr. I-don’t-know.” My training took place in well-thought-of teaching hospitals and during my senior residency and military tour I did enough moonlighting that by the time I entered private practice I had logged a lot of hours in the trenches. But, there were still a ton of things I didn’t know.
You probably remember how those first few years on the outside of the ivory towers felt with no one handy to ask. Even if there was someone a phone call away you didn’t want to appear as incompetent as you were by telling the patient or family that you needed to call the department head at your training program.
So, what did you do? You called. But you developed some clever language that could buy you time while you called your old mentor or hit the books. There was no Internet. Generally, that script would start with some version of “I don’t know, but ...”
As time passed and you gained more experience there were fewer questions and situations in which you needed to admit you were a few clues short of the answer. However, still, many times a day, you honestly didn’t know the answer. Hopefully, over time, you had perfected your delivery so that revealing your ignorance wasn’t driving patients away.
The art of saying “I don’t know” boils down to what you say after the “but” and how you say it. As long as you have offered a plan to find the answer and demonstrate that you are concerned about the patient, your ignorance will be tolerated and maybe even be appreciated.
“I don’t know the answer to that question, but let’s see if we can find a solution” could cover it. If finding that solution is going to require more time than the office visit allows – which it usually doesn’t – the promise of a timely response and a commitment to keep that promise is an absolute requirement. Repeated failure to keep promises is one of the quickest ways to sour a patient-doctor relationship and a potential practice killer.
I think my in-laws no longer refer to me as Dr. I-don’t-know. At least they have refrained from asking me any medical questions. But, my reputation as a physician unafraid to admit his ignorance continues here in town. Occasionally I encounter a parent of a former patient who fondly recalls my willingness to say “I don’t know.” If we had a family crest I would like it to include the motto “Ignoramus Sed Pertinet” (We don’t know but we care).
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
The headline on this article was updated on 2/28/22.
Why challenging patients can trigger resentment
I have a secret. It’s one I think many physicians and nurses share. Sometimes, when I’m stretched too thin — overbooked, hungry, tired, fielding yet another appeal to an insurance company in the middle of a clinic day —
As soon as this happens, I feel immediate guilt. These are the worst moments of my day. Why the heck would I resent my patients? They’re the entire reason I’m there. I wouldn’t be a physician without patients to care for. I became a physician, and completed subspecialty training, to help patients. People.
Recently, I started thinking more about this emotion of resentment. What exactly is it, and where does it come from? Is what I’m feeling actually resentment? Or is it something else?
Two books I’ve recently read have helped me explore the complicated emotion of resentment and how it might play a role in burnout for both physicians and nurses.
First, Brené Brown’s most recent book, Atlas of the Heart: Mapping Meaningful Connection and the Language of Human Experience, provides a roadmap for 87 of our human emotions. (That’s right — 87!)
One emotion of the 87 that she shares has been a particular struggle for her has been our good old friend, resentment.
In her book, Dr Brown shares that she initially considered resentment to belong to the anger family of emotion. As I read this, I agreed. When I feel resentful, I associate that with feeling angry.
But she then writes about her discovery that resentment, in fact, belongs to the envy family. She explains how this discovery shook her world. I had to close the book for a moment at this point.
Wait a minute, I thought. If resentment is in the envy family, why do we (physicians) often find ourselves resenting patients who take up our time? What are we envious of?
I took some time to think about how this might be true. Could it be that I’m envious they have the time I don’t have? I want to have all the time in the world to answer their questions, but the reality is I don’t.
Or maybe it’s because sometimes I feel the patient is expecting me to offer them something more than is available. A cure when there might be none.
But is this actually true? Or is this my unrealistic expectation of myself?
Here’s how Brené Brown defines resentment in her book: “Resentment is the feeling of frustration, judgment, anger, ‘better than,’ and/or hidden envy related to perceived unfairness or injustice. It’s an emotion that we often experience when we fail to set boundaries or ask for what we need, or when expectations let us down because they were based on things we can’t control, like what other people think, what they feel, or how they’re going to react.”
Wow, I thought, Healthcare checks all of these boxes.
- Perceived unfairness of work schedules? Check.
- Perceived injustice? Of course — we see that in our dealings with insurance company denials every day.
But those are both extrinsic. What about the intrinsic factors she’s calling us out on here?
- Do we, as physicians, fail to set boundaries?
- Do we fail to ask for what we need?
Hard yes and yes. (Do we even know, as physicians, what our own boundaries are?)
And the last one:
- Do our expectations of how our clinic day will go let us down every day because they’re based on things we can’t control?
My brain had to repeat the critical parts of that: Expectations let us down when they’re based on things we can’t control.
But wait, my brain argued back; I’m the physician, I thought I was supposed to get to control things.
Next, the revelation: Could it be that a key to experiencing less resentment is accepting how much control we don’t have in a typical day?
And a corollary: How much does resentment factor into burnout? (To read more on my personal journey with burnout, see this piece).
It so happens that around this same time, I was reading another excellent book, Changing How We Think About Difficult Patients: A Guide for Physicians and Healthcare Professionals, by Joan Naidorf, DO.
Dr Naidorf is an emergency medicine physician of 30 years who wrote the book to “provid[e] insight and tools to manage our negative thoughts about difficult patients” and help “beleaguered colleagues…return to their benevolent guiding principles and find more enjoyment in their vitally important careers.”
As I read Dr Naidorf’s book, I thus did so with the mindset of wanting to further understand for myself where this specific emotion of resentment toward our “difficult” patients could come from and how to best understand it in order to get past it.
Dr. Naidorf writes, “Challenging patients will never stop appearing… You cannot change them or control them—the only person you can control is you.”
I wondered how much the resentment we might involuntarily feel at being asked to see a “difficult” patient has nothing to do with the patient but everything to do with it making us feel not in control of the situation.
Dr. Naidorf also writes, “Negative thoughts about challenging patients can cause, in otherwise capable clinicians, a sense of inadequacy and incompetence.”
Do we perhaps resent our challenging patients because of the negative thoughts they sometimes trigger in us? If so, how does this relate to envy, as Dr. Brown asserts resentment is tied to? Is it triggering us to feel inadequate?
“[Difficult patients] often make us question ourselves,” Dr. Naidorf writes, “and we need to feel comfortable with the answers.”
Again, the discrepancy between expectations and reality creates the negative emotion.
Or, as Dr. Naidorf writes, “What if you could stop judging others so harshly and accept them exactly as they are?”
Hmmm, I thought, then the cessation of harsh judgment and implementation of acceptance would have to apply to us too. The elusive concept of self-compassion.
Maybe the resentment/envy comes from us not allowing ourselves to behave in this way because to do so would allow too much vulnerability. Something most of us were conditioned to avoid to survive medical training.
Dr. Brown also writes about an “aha” moment she had in her struggle to understand resentment. “I’m not mad because you’re resting. I’m mad because I’m so bone tired and I want to rest. But, unlike you, I’m going to pretend that I don’t need to.”
I felt all too seen in that passage. Could it be my old nemesis, perfectionism, creeping its way back in? Is resentment the ugly stepsister to perfectionism?
Perhaps challenging patients can engender resentment because they make us feel like we’re not living up to our own unrealistic expectations. And in that case, we need to change our unrealistic expectations for ourselves.
Dr Naidorf’s book explores much more on the complex matter of what makes a “difficult” patient, but I chose to focus here only on the resentment piece as a tie-in to Dr. Brown’s book. I highly recommend both books for further reading to help physicians and nurses navigate the complex emotions our jobs can trigger.
Most importantly, recognizing that we have these transient negative emotions does not make us bad people or healthcare professionals. It only makes us human.
Dr. Lycette is medical director, Providence Oncology and Hematology Care Clinic, Seaside, Ore. She has disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
I have a secret. It’s one I think many physicians and nurses share. Sometimes, when I’m stretched too thin — overbooked, hungry, tired, fielding yet another appeal to an insurance company in the middle of a clinic day —
As soon as this happens, I feel immediate guilt. These are the worst moments of my day. Why the heck would I resent my patients? They’re the entire reason I’m there. I wouldn’t be a physician without patients to care for. I became a physician, and completed subspecialty training, to help patients. People.
Recently, I started thinking more about this emotion of resentment. What exactly is it, and where does it come from? Is what I’m feeling actually resentment? Or is it something else?
Two books I’ve recently read have helped me explore the complicated emotion of resentment and how it might play a role in burnout for both physicians and nurses.
First, Brené Brown’s most recent book, Atlas of the Heart: Mapping Meaningful Connection and the Language of Human Experience, provides a roadmap for 87 of our human emotions. (That’s right — 87!)
One emotion of the 87 that she shares has been a particular struggle for her has been our good old friend, resentment.
In her book, Dr Brown shares that she initially considered resentment to belong to the anger family of emotion. As I read this, I agreed. When I feel resentful, I associate that with feeling angry.
But she then writes about her discovery that resentment, in fact, belongs to the envy family. She explains how this discovery shook her world. I had to close the book for a moment at this point.
Wait a minute, I thought. If resentment is in the envy family, why do we (physicians) often find ourselves resenting patients who take up our time? What are we envious of?
I took some time to think about how this might be true. Could it be that I’m envious they have the time I don’t have? I want to have all the time in the world to answer their questions, but the reality is I don’t.
Or maybe it’s because sometimes I feel the patient is expecting me to offer them something more than is available. A cure when there might be none.
But is this actually true? Or is this my unrealistic expectation of myself?
Here’s how Brené Brown defines resentment in her book: “Resentment is the feeling of frustration, judgment, anger, ‘better than,’ and/or hidden envy related to perceived unfairness or injustice. It’s an emotion that we often experience when we fail to set boundaries or ask for what we need, or when expectations let us down because they were based on things we can’t control, like what other people think, what they feel, or how they’re going to react.”
Wow, I thought, Healthcare checks all of these boxes.
- Perceived unfairness of work schedules? Check.
- Perceived injustice? Of course — we see that in our dealings with insurance company denials every day.
But those are both extrinsic. What about the intrinsic factors she’s calling us out on here?
- Do we, as physicians, fail to set boundaries?
- Do we fail to ask for what we need?
Hard yes and yes. (Do we even know, as physicians, what our own boundaries are?)
And the last one:
- Do our expectations of how our clinic day will go let us down every day because they’re based on things we can’t control?
My brain had to repeat the critical parts of that: Expectations let us down when they’re based on things we can’t control.
But wait, my brain argued back; I’m the physician, I thought I was supposed to get to control things.
Next, the revelation: Could it be that a key to experiencing less resentment is accepting how much control we don’t have in a typical day?
And a corollary: How much does resentment factor into burnout? (To read more on my personal journey with burnout, see this piece).
It so happens that around this same time, I was reading another excellent book, Changing How We Think About Difficult Patients: A Guide for Physicians and Healthcare Professionals, by Joan Naidorf, DO.
Dr Naidorf is an emergency medicine physician of 30 years who wrote the book to “provid[e] insight and tools to manage our negative thoughts about difficult patients” and help “beleaguered colleagues…return to their benevolent guiding principles and find more enjoyment in their vitally important careers.”
As I read Dr Naidorf’s book, I thus did so with the mindset of wanting to further understand for myself where this specific emotion of resentment toward our “difficult” patients could come from and how to best understand it in order to get past it.
Dr. Naidorf writes, “Challenging patients will never stop appearing… You cannot change them or control them—the only person you can control is you.”
I wondered how much the resentment we might involuntarily feel at being asked to see a “difficult” patient has nothing to do with the patient but everything to do with it making us feel not in control of the situation.
Dr. Naidorf also writes, “Negative thoughts about challenging patients can cause, in otherwise capable clinicians, a sense of inadequacy and incompetence.”
Do we perhaps resent our challenging patients because of the negative thoughts they sometimes trigger in us? If so, how does this relate to envy, as Dr. Brown asserts resentment is tied to? Is it triggering us to feel inadequate?
“[Difficult patients] often make us question ourselves,” Dr. Naidorf writes, “and we need to feel comfortable with the answers.”
Again, the discrepancy between expectations and reality creates the negative emotion.
Or, as Dr. Naidorf writes, “What if you could stop judging others so harshly and accept them exactly as they are?”
Hmmm, I thought, then the cessation of harsh judgment and implementation of acceptance would have to apply to us too. The elusive concept of self-compassion.
Maybe the resentment/envy comes from us not allowing ourselves to behave in this way because to do so would allow too much vulnerability. Something most of us were conditioned to avoid to survive medical training.
Dr. Brown also writes about an “aha” moment she had in her struggle to understand resentment. “I’m not mad because you’re resting. I’m mad because I’m so bone tired and I want to rest. But, unlike you, I’m going to pretend that I don’t need to.”
I felt all too seen in that passage. Could it be my old nemesis, perfectionism, creeping its way back in? Is resentment the ugly stepsister to perfectionism?
Perhaps challenging patients can engender resentment because they make us feel like we’re not living up to our own unrealistic expectations. And in that case, we need to change our unrealistic expectations for ourselves.
Dr Naidorf’s book explores much more on the complex matter of what makes a “difficult” patient, but I chose to focus here only on the resentment piece as a tie-in to Dr. Brown’s book. I highly recommend both books for further reading to help physicians and nurses navigate the complex emotions our jobs can trigger.
Most importantly, recognizing that we have these transient negative emotions does not make us bad people or healthcare professionals. It only makes us human.
Dr. Lycette is medical director, Providence Oncology and Hematology Care Clinic, Seaside, Ore. She has disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
I have a secret. It’s one I think many physicians and nurses share. Sometimes, when I’m stretched too thin — overbooked, hungry, tired, fielding yet another appeal to an insurance company in the middle of a clinic day —
As soon as this happens, I feel immediate guilt. These are the worst moments of my day. Why the heck would I resent my patients? They’re the entire reason I’m there. I wouldn’t be a physician without patients to care for. I became a physician, and completed subspecialty training, to help patients. People.
Recently, I started thinking more about this emotion of resentment. What exactly is it, and where does it come from? Is what I’m feeling actually resentment? Or is it something else?
Two books I’ve recently read have helped me explore the complicated emotion of resentment and how it might play a role in burnout for both physicians and nurses.
First, Brené Brown’s most recent book, Atlas of the Heart: Mapping Meaningful Connection and the Language of Human Experience, provides a roadmap for 87 of our human emotions. (That’s right — 87!)
One emotion of the 87 that she shares has been a particular struggle for her has been our good old friend, resentment.
In her book, Dr Brown shares that she initially considered resentment to belong to the anger family of emotion. As I read this, I agreed. When I feel resentful, I associate that with feeling angry.
But she then writes about her discovery that resentment, in fact, belongs to the envy family. She explains how this discovery shook her world. I had to close the book for a moment at this point.
Wait a minute, I thought. If resentment is in the envy family, why do we (physicians) often find ourselves resenting patients who take up our time? What are we envious of?
I took some time to think about how this might be true. Could it be that I’m envious they have the time I don’t have? I want to have all the time in the world to answer their questions, but the reality is I don’t.
Or maybe it’s because sometimes I feel the patient is expecting me to offer them something more than is available. A cure when there might be none.
But is this actually true? Or is this my unrealistic expectation of myself?
Here’s how Brené Brown defines resentment in her book: “Resentment is the feeling of frustration, judgment, anger, ‘better than,’ and/or hidden envy related to perceived unfairness or injustice. It’s an emotion that we often experience when we fail to set boundaries or ask for what we need, or when expectations let us down because they were based on things we can’t control, like what other people think, what they feel, or how they’re going to react.”
Wow, I thought, Healthcare checks all of these boxes.
- Perceived unfairness of work schedules? Check.
- Perceived injustice? Of course — we see that in our dealings with insurance company denials every day.
But those are both extrinsic. What about the intrinsic factors she’s calling us out on here?
- Do we, as physicians, fail to set boundaries?
- Do we fail to ask for what we need?
Hard yes and yes. (Do we even know, as physicians, what our own boundaries are?)
And the last one:
- Do our expectations of how our clinic day will go let us down every day because they’re based on things we can’t control?
My brain had to repeat the critical parts of that: Expectations let us down when they’re based on things we can’t control.
But wait, my brain argued back; I’m the physician, I thought I was supposed to get to control things.
Next, the revelation: Could it be that a key to experiencing less resentment is accepting how much control we don’t have in a typical day?
And a corollary: How much does resentment factor into burnout? (To read more on my personal journey with burnout, see this piece).
It so happens that around this same time, I was reading another excellent book, Changing How We Think About Difficult Patients: A Guide for Physicians and Healthcare Professionals, by Joan Naidorf, DO.
Dr Naidorf is an emergency medicine physician of 30 years who wrote the book to “provid[e] insight and tools to manage our negative thoughts about difficult patients” and help “beleaguered colleagues…return to their benevolent guiding principles and find more enjoyment in their vitally important careers.”
As I read Dr Naidorf’s book, I thus did so with the mindset of wanting to further understand for myself where this specific emotion of resentment toward our “difficult” patients could come from and how to best understand it in order to get past it.
Dr. Naidorf writes, “Challenging patients will never stop appearing… You cannot change them or control them—the only person you can control is you.”
I wondered how much the resentment we might involuntarily feel at being asked to see a “difficult” patient has nothing to do with the patient but everything to do with it making us feel not in control of the situation.
Dr. Naidorf also writes, “Negative thoughts about challenging patients can cause, in otherwise capable clinicians, a sense of inadequacy and incompetence.”
Do we perhaps resent our challenging patients because of the negative thoughts they sometimes trigger in us? If so, how does this relate to envy, as Dr. Brown asserts resentment is tied to? Is it triggering us to feel inadequate?
“[Difficult patients] often make us question ourselves,” Dr. Naidorf writes, “and we need to feel comfortable with the answers.”
Again, the discrepancy between expectations and reality creates the negative emotion.
Or, as Dr. Naidorf writes, “What if you could stop judging others so harshly and accept them exactly as they are?”
Hmmm, I thought, then the cessation of harsh judgment and implementation of acceptance would have to apply to us too. The elusive concept of self-compassion.
Maybe the resentment/envy comes from us not allowing ourselves to behave in this way because to do so would allow too much vulnerability. Something most of us were conditioned to avoid to survive medical training.
Dr. Brown also writes about an “aha” moment she had in her struggle to understand resentment. “I’m not mad because you’re resting. I’m mad because I’m so bone tired and I want to rest. But, unlike you, I’m going to pretend that I don’t need to.”
I felt all too seen in that passage. Could it be my old nemesis, perfectionism, creeping its way back in? Is resentment the ugly stepsister to perfectionism?
Perhaps challenging patients can engender resentment because they make us feel like we’re not living up to our own unrealistic expectations. And in that case, we need to change our unrealistic expectations for ourselves.
Dr Naidorf’s book explores much more on the complex matter of what makes a “difficult” patient, but I chose to focus here only on the resentment piece as a tie-in to Dr. Brown’s book. I highly recommend both books for further reading to help physicians and nurses navigate the complex emotions our jobs can trigger.
Most importantly, recognizing that we have these transient negative emotions does not make us bad people or healthcare professionals. It only makes us human.
Dr. Lycette is medical director, Providence Oncology and Hematology Care Clinic, Seaside, Ore. She has disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Infectious disease pop quiz: Clinical challenge #16 for the ObGyn
What is the best test for the diagnosis of acute hepatitis A infection?
Continue to the answer...
The single best test for the diagnosis of acute hepatitis A infection is detection of immunoglobulin M (IgM)–specific antibody to the virus.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
What is the best test for the diagnosis of acute hepatitis A infection?
Continue to the answer...
The single best test for the diagnosis of acute hepatitis A infection is detection of immunoglobulin M (IgM)–specific antibody to the virus.
What is the best test for the diagnosis of acute hepatitis A infection?
Continue to the answer...
The single best test for the diagnosis of acute hepatitis A infection is detection of immunoglobulin M (IgM)–specific antibody to the virus.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
- Duff P. Maternal and perinatal infections: bacterial. In: Landon MB, Galan HL, Jauniaux ERM, et al. Gabbe’s Obstetrics: Normal and Problem Pregnancies. 8th ed. Elsevier; 2021:1124-1146.
- Duff P. Maternal and fetal infections. In: Resnik R, Lockwood CJ, Moore TJ, et al. Creasy & Resnik’s Maternal-Fetal Medicine: Principles and Practice. 8th ed. Elsevier; 2019:862-919.
Ukrainian physicians ‘ready to die for their freedom’
Nasogastric tubes. Foley catheter kits. Hydrogel anti-burn bandages and transfusion bags. Heparin, atropine, tramadol.
These items are just a few of some two dozen critical medical supplies that physicians in Ukraine desperately need, according to Leo Wolansky, MD, a Ukrainian-American radiologist and president of the Ukrainian Medical Association of North America (UMANA).
Dr. Wolansky founded a teaching program with an organization called Friends of Radiology in Ukraine in 1996 and has been running courses for specialists there ever since. He last visited the country in 2019, before the COVID-19 pandemic, but has remained in contact with his medical colleagues by phone and email. Over the weekend of Feb. 26-27, UMANA held a fundraiser for Ukraine, raising more than $17,000.
Question: Where is your family from, and do you have relatives in the country now?
Dr. Wolansky: My family is from two different parts of Ukraine. My mother was from central Ukraine. Her father, Ivan Sharyj, was part of the students’ militia that fought at the famous battle of Kruty in 1918. Four hundred Ukrainian militia fought against 5,000 professional Russian soldiers and were massacred. He later wrote the first eye-witness account. Afterwards, he had the opportunity to flee Ukraine but chose to stay under a pseudonym. Eventually, during Stalin’s purges [1929-1933], the regime found him, arrested him, tortured him, and executed him. My mother was seven when she saw her father arrested, never to return home. My father was from Western Ukraine, which did not have a long history of Russian occupation. His mother’s family was very patriotic; her first cousin, Stepan Vytvytskyi, eventually became the president of Ukraine in exile from 1955-1964.
I have second and more distant cousins in Kyiv. My wife has first cousins in Western Ukraine. They and my doctor colleagues are suffering greatly but are ready to die for their freedom.
Question: The Russian invasion of Ukraine has put tremendous stress on the Ukrainian people, including the country’s medical professionals. How do doctors in these kinds of situations handle casualties they can’t prevent? How do they work around that sense that everything is out of their control?
Dr. Wolansky: A lot of infrastructural things are being disrupted; there are limitations that you wouldn’t normally encounter. Ukraine has been developing a lot of sophisticated medical technology, but it still has room to grow. Under these circumstances, when there are bombs going off and transportation is being disrupted, it creates very new and significant obstacles to surmount. It still has not risen to massive casualties, and we can just pray that it does not, but in times of war, a very different kind of medicine is practiced.
But remember, Ukraine has been at war since 2014, when Russia took Crimea and invaded the Eastern provinces. The doctors there are not unfamiliar with war injuries. At our conferences in Ukraine, I have seen radiological presentations of injuries sustained in war – gunshots, fractures, and amputations – as well as other kinds of traumatic injuries. You’re going for a kind of more emergent treatment: to transfuse, to maintain peoples’ blood pressure, put bandages on, sterilize and sanitize wounds to prevent infections. I imagine there will be many field hospitals set up between now and the next few weeks to deal with the acute injuries.
Question: Ukraine has struggled with high rates of HIV and multidrug-resistant tuberculosis, as well as a lack of resources for treating patients with mental illness. Meanwhile, the country has had more that 5 million cases of COVID-19 and an estimated 112,000 deaths from the disease. Are you concerned about an exacerbation of infection rates, including of COVID, particularly among refugees and those who become homeless?
Dr. Wolansky: Because COVID ran pretty rampant in Ukraine, I think that – at a high cost – there is a level of natural immunity in the population. And the weather is going to be getting warmer soon, and respiratory viruses are cyclic in nature, so I don’t know if that’s going to be a big complicating factor. However, people get sick all the time, and the prognosis for them is going to be much worse than it otherwise might be. If you have a heart attack, your chances were way better when the roads were clear and people weren’t shooting at you.
Right now, it’s very regional where the infrastructure is being destroyed. The West, where I used to go, is in much better shape than the East because it has not been the focus of Russian attacks. But Kyiv could turn into a very big humanitarian crisis very quickly if there’s no electricity, no water. All sorts of medical conditions could be greatly exacerbated, and some new health crises could arise from water contamination, bombs causing buildings to collapse, and other problems. Whatever the illness is, it’s going to be harder to take care of it.
Questions: Doctors Without Borders announced that it was suspending its operations in Ukraine because of the invasion – missions that included HIV care in Severodonetsk, tuberculosis care in Zhytomyr, and improving health care access in Donetsk in eastern Ukraine, according to the aid group. What do doctors in Ukraine need most acutely now, other than peace?
Dr. Wolansky: Obviously, money is valuable, and military protection, which would prevent additional damage to their infrastructure. One thing that bears mentioning. There’s been a fair amount of coverage of this, but I’ve witnessed it first-hand: The Ukrainian people are fiercely patriotic, and there’s really no way their spirit can be conquered. The USSR invaded Afghanistan, and after years of thinking they were in command, they left because they could no longer take the guerilla warfare and the constant sniper attacks. Ukraine’s population is many times larger than Afghanistan’s; there’s no way they can be subdued. And remember, the Ukrainian people have been free for 30 years – generations of young people have known no other way of life. They are not going to give that up.
A version of this article first appeared on Medscape.com.
Nasogastric tubes. Foley catheter kits. Hydrogel anti-burn bandages and transfusion bags. Heparin, atropine, tramadol.
These items are just a few of some two dozen critical medical supplies that physicians in Ukraine desperately need, according to Leo Wolansky, MD, a Ukrainian-American radiologist and president of the Ukrainian Medical Association of North America (UMANA).
Dr. Wolansky founded a teaching program with an organization called Friends of Radiology in Ukraine in 1996 and has been running courses for specialists there ever since. He last visited the country in 2019, before the COVID-19 pandemic, but has remained in contact with his medical colleagues by phone and email. Over the weekend of Feb. 26-27, UMANA held a fundraiser for Ukraine, raising more than $17,000.
Question: Where is your family from, and do you have relatives in the country now?
Dr. Wolansky: My family is from two different parts of Ukraine. My mother was from central Ukraine. Her father, Ivan Sharyj, was part of the students’ militia that fought at the famous battle of Kruty in 1918. Four hundred Ukrainian militia fought against 5,000 professional Russian soldiers and were massacred. He later wrote the first eye-witness account. Afterwards, he had the opportunity to flee Ukraine but chose to stay under a pseudonym. Eventually, during Stalin’s purges [1929-1933], the regime found him, arrested him, tortured him, and executed him. My mother was seven when she saw her father arrested, never to return home. My father was from Western Ukraine, which did not have a long history of Russian occupation. His mother’s family was very patriotic; her first cousin, Stepan Vytvytskyi, eventually became the president of Ukraine in exile from 1955-1964.
I have second and more distant cousins in Kyiv. My wife has first cousins in Western Ukraine. They and my doctor colleagues are suffering greatly but are ready to die for their freedom.
Question: The Russian invasion of Ukraine has put tremendous stress on the Ukrainian people, including the country’s medical professionals. How do doctors in these kinds of situations handle casualties they can’t prevent? How do they work around that sense that everything is out of their control?
Dr. Wolansky: A lot of infrastructural things are being disrupted; there are limitations that you wouldn’t normally encounter. Ukraine has been developing a lot of sophisticated medical technology, but it still has room to grow. Under these circumstances, when there are bombs going off and transportation is being disrupted, it creates very new and significant obstacles to surmount. It still has not risen to massive casualties, and we can just pray that it does not, but in times of war, a very different kind of medicine is practiced.
But remember, Ukraine has been at war since 2014, when Russia took Crimea and invaded the Eastern provinces. The doctors there are not unfamiliar with war injuries. At our conferences in Ukraine, I have seen radiological presentations of injuries sustained in war – gunshots, fractures, and amputations – as well as other kinds of traumatic injuries. You’re going for a kind of more emergent treatment: to transfuse, to maintain peoples’ blood pressure, put bandages on, sterilize and sanitize wounds to prevent infections. I imagine there will be many field hospitals set up between now and the next few weeks to deal with the acute injuries.
Question: Ukraine has struggled with high rates of HIV and multidrug-resistant tuberculosis, as well as a lack of resources for treating patients with mental illness. Meanwhile, the country has had more that 5 million cases of COVID-19 and an estimated 112,000 deaths from the disease. Are you concerned about an exacerbation of infection rates, including of COVID, particularly among refugees and those who become homeless?
Dr. Wolansky: Because COVID ran pretty rampant in Ukraine, I think that – at a high cost – there is a level of natural immunity in the population. And the weather is going to be getting warmer soon, and respiratory viruses are cyclic in nature, so I don’t know if that’s going to be a big complicating factor. However, people get sick all the time, and the prognosis for them is going to be much worse than it otherwise might be. If you have a heart attack, your chances were way better when the roads were clear and people weren’t shooting at you.
Right now, it’s very regional where the infrastructure is being destroyed. The West, where I used to go, is in much better shape than the East because it has not been the focus of Russian attacks. But Kyiv could turn into a very big humanitarian crisis very quickly if there’s no electricity, no water. All sorts of medical conditions could be greatly exacerbated, and some new health crises could arise from water contamination, bombs causing buildings to collapse, and other problems. Whatever the illness is, it’s going to be harder to take care of it.
Questions: Doctors Without Borders announced that it was suspending its operations in Ukraine because of the invasion – missions that included HIV care in Severodonetsk, tuberculosis care in Zhytomyr, and improving health care access in Donetsk in eastern Ukraine, according to the aid group. What do doctors in Ukraine need most acutely now, other than peace?
Dr. Wolansky: Obviously, money is valuable, and military protection, which would prevent additional damage to their infrastructure. One thing that bears mentioning. There’s been a fair amount of coverage of this, but I’ve witnessed it first-hand: The Ukrainian people are fiercely patriotic, and there’s really no way their spirit can be conquered. The USSR invaded Afghanistan, and after years of thinking they were in command, they left because they could no longer take the guerilla warfare and the constant sniper attacks. Ukraine’s population is many times larger than Afghanistan’s; there’s no way they can be subdued. And remember, the Ukrainian people have been free for 30 years – generations of young people have known no other way of life. They are not going to give that up.
A version of this article first appeared on Medscape.com.
Nasogastric tubes. Foley catheter kits. Hydrogel anti-burn bandages and transfusion bags. Heparin, atropine, tramadol.
These items are just a few of some two dozen critical medical supplies that physicians in Ukraine desperately need, according to Leo Wolansky, MD, a Ukrainian-American radiologist and president of the Ukrainian Medical Association of North America (UMANA).
Dr. Wolansky founded a teaching program with an organization called Friends of Radiology in Ukraine in 1996 and has been running courses for specialists there ever since. He last visited the country in 2019, before the COVID-19 pandemic, but has remained in contact with his medical colleagues by phone and email. Over the weekend of Feb. 26-27, UMANA held a fundraiser for Ukraine, raising more than $17,000.
Question: Where is your family from, and do you have relatives in the country now?
Dr. Wolansky: My family is from two different parts of Ukraine. My mother was from central Ukraine. Her father, Ivan Sharyj, was part of the students’ militia that fought at the famous battle of Kruty in 1918. Four hundred Ukrainian militia fought against 5,000 professional Russian soldiers and were massacred. He later wrote the first eye-witness account. Afterwards, he had the opportunity to flee Ukraine but chose to stay under a pseudonym. Eventually, during Stalin’s purges [1929-1933], the regime found him, arrested him, tortured him, and executed him. My mother was seven when she saw her father arrested, never to return home. My father was from Western Ukraine, which did not have a long history of Russian occupation. His mother’s family was very patriotic; her first cousin, Stepan Vytvytskyi, eventually became the president of Ukraine in exile from 1955-1964.
I have second and more distant cousins in Kyiv. My wife has first cousins in Western Ukraine. They and my doctor colleagues are suffering greatly but are ready to die for their freedom.
Question: The Russian invasion of Ukraine has put tremendous stress on the Ukrainian people, including the country’s medical professionals. How do doctors in these kinds of situations handle casualties they can’t prevent? How do they work around that sense that everything is out of their control?
Dr. Wolansky: A lot of infrastructural things are being disrupted; there are limitations that you wouldn’t normally encounter. Ukraine has been developing a lot of sophisticated medical technology, but it still has room to grow. Under these circumstances, when there are bombs going off and transportation is being disrupted, it creates very new and significant obstacles to surmount. It still has not risen to massive casualties, and we can just pray that it does not, but in times of war, a very different kind of medicine is practiced.
But remember, Ukraine has been at war since 2014, when Russia took Crimea and invaded the Eastern provinces. The doctors there are not unfamiliar with war injuries. At our conferences in Ukraine, I have seen radiological presentations of injuries sustained in war – gunshots, fractures, and amputations – as well as other kinds of traumatic injuries. You’re going for a kind of more emergent treatment: to transfuse, to maintain peoples’ blood pressure, put bandages on, sterilize and sanitize wounds to prevent infections. I imagine there will be many field hospitals set up between now and the next few weeks to deal with the acute injuries.
Question: Ukraine has struggled with high rates of HIV and multidrug-resistant tuberculosis, as well as a lack of resources for treating patients with mental illness. Meanwhile, the country has had more that 5 million cases of COVID-19 and an estimated 112,000 deaths from the disease. Are you concerned about an exacerbation of infection rates, including of COVID, particularly among refugees and those who become homeless?
Dr. Wolansky: Because COVID ran pretty rampant in Ukraine, I think that – at a high cost – there is a level of natural immunity in the population. And the weather is going to be getting warmer soon, and respiratory viruses are cyclic in nature, so I don’t know if that’s going to be a big complicating factor. However, people get sick all the time, and the prognosis for them is going to be much worse than it otherwise might be. If you have a heart attack, your chances were way better when the roads were clear and people weren’t shooting at you.
Right now, it’s very regional where the infrastructure is being destroyed. The West, where I used to go, is in much better shape than the East because it has not been the focus of Russian attacks. But Kyiv could turn into a very big humanitarian crisis very quickly if there’s no electricity, no water. All sorts of medical conditions could be greatly exacerbated, and some new health crises could arise from water contamination, bombs causing buildings to collapse, and other problems. Whatever the illness is, it’s going to be harder to take care of it.
Questions: Doctors Without Borders announced that it was suspending its operations in Ukraine because of the invasion – missions that included HIV care in Severodonetsk, tuberculosis care in Zhytomyr, and improving health care access in Donetsk in eastern Ukraine, according to the aid group. What do doctors in Ukraine need most acutely now, other than peace?
Dr. Wolansky: Obviously, money is valuable, and military protection, which would prevent additional damage to their infrastructure. One thing that bears mentioning. There’s been a fair amount of coverage of this, but I’ve witnessed it first-hand: The Ukrainian people are fiercely patriotic, and there’s really no way their spirit can be conquered. The USSR invaded Afghanistan, and after years of thinking they were in command, they left because they could no longer take the guerilla warfare and the constant sniper attacks. Ukraine’s population is many times larger than Afghanistan’s; there’s no way they can be subdued. And remember, the Ukrainian people have been free for 30 years – generations of young people have known no other way of life. They are not going to give that up.
A version of this article first appeared on Medscape.com.







