User login
For MD-IQ use only
Doc releases song after racist massacre in Buffalo
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Physician-musician Cleveland Francis, MD, responded to the recent mass shooting in Buffalo, New York, which left 10 dead, in the only way he knew how. He wrote and recorded a song to honor the victims as “a plea to the other side to recognize us as people,” the Black cardiologist told this news organization.
He couldn’t sleep after the shooting, and “this song was just in my head.” In the 1990s, Dr. Francis took a 3-year sabbatical from medicine to perform and tour as a country singer. He leveraged his Nashville connections to get “Buffalo” produced and recorded.
Acclaimed artist James Threalkill created the accompanying art, titled “The Heavenly Escort of the Buffalo 10,” after listening to a scratch demo.
Dr. Francis doesn’t want people to overlook the massacre as just another gun violence incident because this was “overt hate-crime racism,” he said.
According to the affidavit submitted by FBI agent Christopher J. Dlugokinski, the suspect’s “motive for the mass shooting was to prevent Black people from replacing White people and eliminating the White race, and to inspire others to commit similar attacks.”
Dr. Francis views the Buffalo shooting as distinct from cases like the murder of George Floyd that involved crime or police. It immediately made him think of the Mother Emanuel Church shooting in Charleston, South Carolina. “Having a black skin is now a death warrant,” he said.
The song is also an appeal for White people to fight racism. Dr. Francis is concerned about young men caught up in white supremacy and suggests that we be more alert to children or grandchildren who disconnect from their families, spend time on the dark web, and access guns. The lyrics deliberately don’t mention guns because Dr. Francis wanted to stay out of that debate. “I just sang: ‘What else do I have to do to prove to you that I’m human too?’ ”
Despite his country credentials, Dr. Francis wrote “Buffalo” as a Gospel song because that genre “connects with Black people more and because that civil rights movement was through the church with Dr. Martin Luther King,” he explained. Although he sings all styles of music, the song is performed by Nashville-based singer Michael Lusk so that it’s not a “Cleve Francis thing,” he said, referring to his stage name.
Songwriter Norman Kerner collaborated on the song. The music was produced and recorded by David Thein and mixed by Bob Bullock of Nashville, who Dr. Francis had worked with when he was an artist on Capitol Records.
They sent the video and artwork to the Mayor of Buffalo, Byron Brown, but have yet to hear back. Dr. Francis hopes it could be part of their healing, noting that some people used the song in their Juneteenth celebrations.
The Louisiana native grew up during segregation and was one of two Black students in the Medical College of Virginia class of 1973. After completing his cardiology fellowship, no one would hire him, so Dr. Francis set up his own practice in Northern Virginia. He now works at Inova Heart and Vascular Institute in Alexandria, Va. He remains optimistic about race relations in America and would love a Black pop or Gospel star to record “Buffalo” and bring it to a wider audience.
Dr. Francis is a regular blogger for Medscape. His contribution to country music is recognized in the National Museum of African American History and Culture in Washington, DC. You can find more of his music on YouTube.
A version of this article first appeared on Medscape.com.
Mosquitoes and the vicious circle that’s gone viral
These viruses want mosquitoes with good taste
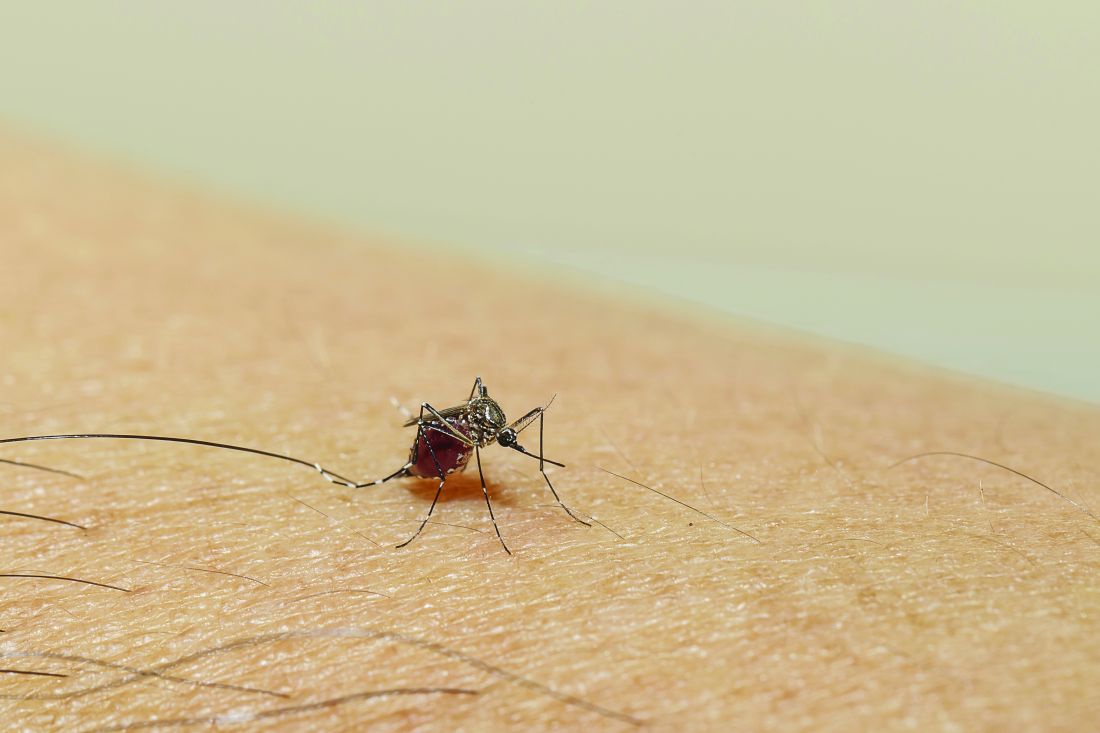
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
These viruses want mosquitoes with good taste
Taste can be a pretty subjective sense. Not everyone agrees on what tastes good and what tastes bad. Most people would agree that freshly baked cookies taste good, but what about lima beans? And what about mosquitoes? What tastes good to a mosquito?
The answer? Blood. Blood tastes good to a mosquito. That really wasn’t a very hard question, was it? You did know the answer, didn’t you? They don’t care about cookies, and they certainly don’t care about lima beans. It’s blood that they love.
That brings us back to subjectivity, because it is possible for blood to taste even better. The secret ingredient is dengue … and Zika.
A study just published in Cell demonstrates that mice infected with dengue and Zika viruses release a volatile compound called acetophenone. “We found that flavivirus [like dengue and Zika] can utilize the increased release of acetophenone to help itself achieve its lifecycles more effectively by making their hosts more attractive to mosquito vectors,” senior author Gong Cheng of Tsinghua University, Beijing, said in a written statement.
How do they do it? The viruses, he explained, promote the proliferation of acetophenone-producing skin bacteria. “As a result, some bacteria overreplicate and produce more acetophenone. Suddenly, these sick individuals smell as delicious to mosquitoes as a tray of freshly baked cookies to a group of five-year-old children,” the statement said.
And how do you stop a group of tiny, flying 5-year-olds? That’s right, with acne medication. Really? You knew that one but not the blood one before? The investigators fed isotretinoin to the infected mice, which led to reduced acetophenone release from skin bacteria and made the animals no more attractive to the mosquitoes than their uninfected counterparts.
The investigators are planning to take the next step – feeding isotretinoin to people with dengue and Zika – having gotten the official fictional taste-test approval of celebrity chef Gordon Ramsay, who said, “You’re going to feed this #$^% to sick people? ARE YOU &%*$@#& KIDDING ME?”
Okay, so maybe approval isn’t quite the right word.
Welcome to bladders of the rich and famous!
Don’t you hate it when you’re driving out to your multimillion-dollar second home in the Hamptons and traffic is so bad you absolutely have to find a place to “rest” along the way? But wouldn’t you know it, there just isn’t anywhere to stop! Geez, how do we live?
That’s where David Shusterman, MD, a urologist in New York City and a true American hero, comes in. He’s identified a market and positioned himself as the king of both bladder surgery and “bladder Botox” for the wealthy New Yorkers who regularly make long journeys from the city out to their second homes in the Hamptons. Traffic has increased dramatically on Long Island roads in recent years, and the journey can now taking upward of 4 hours. Some people just can’t make it that long without a bathroom break, and there are very few places to stop along the way.
Dr. Shusterman understands the plight of the Hamptons vacationer, as he told Insider.com: “I can’t tell you how many arguments I personally get into – I’ve lost three friends because I’m the driver and refuse to stop for them.” A tragedy worthy of Shakespeare himself.
During the summer season, Dr. Shusterman performs about 10 prostate artery embolizations a week, an hour-long procedure that shrinks the prostate, which is great for 50- to 60-year-old men with enlarged prostates that cause more frequent bathroom trips. He also performs Botox injections into the bladder once or twice a week for women, which reduces the need to urinate for roughly 6 months. The perfect amount of time to get them through the summer season.
These procedures are sometimes covered by insurance but can cost as much as $20,000 if paid out of pocket. That’s a lot of money to us, but if you’re the sort of person who has a second home in the Hamptons, $20,000 is chump change, especially if it means you won’t have to go 2 entire minutes out of your way to use a gas-station bathroom. Then again, having seen a more than a few gas-station bathrooms in our time, maybe they have a point.
Ditch the apples. Go for the avocados
We’ve all heard about “an apple a day,” but instead of apples you might want to go with avocados.
Avocados are generally thought to be a healthy fat. A study just published in the Journal of the American Heart Association proves that they actually don’t do anything for your waistline but will work wonders on your cholesterol level. The study involved 923 participants who were considered overweight/obese split into two groups: One was asked to consume an avocado a day, and the other continued their usual diets and were asked to consume fewer than two avocados a month.
At the end of the 6 months, the researchers found total cholesterol decreased by an additional 2.9 mg/dL and LDL cholesterol by 2.5 mg/dL in those who ate one avocado every day, compared with the usual-diet group. And even though avocados have a lot of calories, there was no clinical evidence that it impacted weight gain or any cardiometabolic risk factors, according to a statement from Penn State University.
Avocados, then, can be considered a guilt-free food. The findings from this study suggest it can give a substantial boost to your overall quality of diet, in turn lessening your risk of developing type 2 diabetes and some cancers, Kristina Peterson, PhD, assistant professor of nutritional sciences at Texas Tech University, said in the statement.
So get creative with your avocado recipes. You can only eat so much guacamole.
Your nose knows a good friend for you
You’ve probably noticed how dogs sniff other dogs and people before becoming friends. It would be pretty comical if people did the same thing, right? Just walked up to strangers and started sniffing them like dogs?
Well, apparently humans do go by smell when it comes to making friends, and they prefer people who smell like them. Maybe you’ve noticed that your friends look like you, share your values, and think the same way as you. You’re probably right, seeing as previous research has pointed to this.
For the current study, done to show how smell affects human behavior, researchers recruited people who befriended each other quickly, before knowing much about each other. They assumed that the relationships between these same-sex, nonromantic “click friends” relied more on physiological traits, including smell. After collecting samples from the click friends, researchers used an eNose to scan chemical signatures. In another experiment, human volunteers sniffed samples to determine if any were similar. Both experiments showed that click friends had more similar smells than pairs of random people.
“This is not to say that we act like goats or shrews – humans likely rely on other, far more dominant cues in their social decision-making. Nevertheless, our study’s results do suggest that our nose plays a bigger role than previously thought in our choice of friends,” said senior author Noam Sobel, PhD, of the Weizmann Institute of Science in Rehovot, Israel.
Lead author Inbal Ravreby, a graduate student at the institute, put it this way: “These results imply that, as the saying goes, there is chemistry in social chemistry.”
Bored? Change the world or read a book
A weekend, for most of us in solo practice, doesn’t really signify time off from work. It just means we’re not seeing patients at the office.
There’s always business stuff to do (like payroll and paying bills), legal cases to review, the never-ending forms for a million things, and all the other stuff there never seems to be enough time to do on weekdays.
So this weekend I started attacking the pile after dinner on Friday and found myself done by Saturday afternoon. Which is rare, usually I spend the better part of a weekend at my desk.
And then, unexpectedly faced with an empty desk, I found myself wondering what to do.
Boredom is one of the odder human conditions. Certainly, there are more ways to waste time now than there ever have been. TV, Netflix, phone games, TikTok, books, just to name a few.
But do we always have to be entertained? Many great scientists have said that world-changing ideas have come to them when they weren’t working, such as while showering or riding to work. Leo Szilard was crossing a London street in 1933 when he suddenly saw how a nuclear chain reaction would be self-sustaining once initiated. (Fortunately, he wasn’t hit by a car in the process.)
But I’m not Szilard. So I rationalized a reason not to exercise and sat on the couch with a book.
The remarkable human brain doesn’t shut down easily. With nothing else to do, most other mammals tend to doze off. But not us. It’s always on, trying to think of the next goal, the next move, the next whatever.
Having nothing to do sounds like a great idea, until you have nothing to do. It may be fine for a few days, but after a while you realize there’s only so long you can stare at the waves or mountains before your mind turns back to “what’s next.”
This isn’t a bad thing. Being bored is probably constructive. Without realizing it we use it to form new ideas and start new plans.
Maybe this is why we’re here. The mind that keeps working is a powerful tool, driving us forward in all walks of life. Perhaps it’s this feature that pushed the development of intelligence further and led us to form civilizations.
Perhaps it’s the real reason we keep moving forward.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
A weekend, for most of us in solo practice, doesn’t really signify time off from work. It just means we’re not seeing patients at the office.
There’s always business stuff to do (like payroll and paying bills), legal cases to review, the never-ending forms for a million things, and all the other stuff there never seems to be enough time to do on weekdays.
So this weekend I started attacking the pile after dinner on Friday and found myself done by Saturday afternoon. Which is rare, usually I spend the better part of a weekend at my desk.
And then, unexpectedly faced with an empty desk, I found myself wondering what to do.
Boredom is one of the odder human conditions. Certainly, there are more ways to waste time now than there ever have been. TV, Netflix, phone games, TikTok, books, just to name a few.
But do we always have to be entertained? Many great scientists have said that world-changing ideas have come to them when they weren’t working, such as while showering or riding to work. Leo Szilard was crossing a London street in 1933 when he suddenly saw how a nuclear chain reaction would be self-sustaining once initiated. (Fortunately, he wasn’t hit by a car in the process.)
But I’m not Szilard. So I rationalized a reason not to exercise and sat on the couch with a book.
The remarkable human brain doesn’t shut down easily. With nothing else to do, most other mammals tend to doze off. But not us. It’s always on, trying to think of the next goal, the next move, the next whatever.
Having nothing to do sounds like a great idea, until you have nothing to do. It may be fine for a few days, but after a while you realize there’s only so long you can stare at the waves or mountains before your mind turns back to “what’s next.”
This isn’t a bad thing. Being bored is probably constructive. Without realizing it we use it to form new ideas and start new plans.
Maybe this is why we’re here. The mind that keeps working is a powerful tool, driving us forward in all walks of life. Perhaps it’s this feature that pushed the development of intelligence further and led us to form civilizations.
Perhaps it’s the real reason we keep moving forward.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
A weekend, for most of us in solo practice, doesn’t really signify time off from work. It just means we’re not seeing patients at the office.
There’s always business stuff to do (like payroll and paying bills), legal cases to review, the never-ending forms for a million things, and all the other stuff there never seems to be enough time to do on weekdays.
So this weekend I started attacking the pile after dinner on Friday and found myself done by Saturday afternoon. Which is rare, usually I spend the better part of a weekend at my desk.
And then, unexpectedly faced with an empty desk, I found myself wondering what to do.
Boredom is one of the odder human conditions. Certainly, there are more ways to waste time now than there ever have been. TV, Netflix, phone games, TikTok, books, just to name a few.
But do we always have to be entertained? Many great scientists have said that world-changing ideas have come to them when they weren’t working, such as while showering or riding to work. Leo Szilard was crossing a London street in 1933 when he suddenly saw how a nuclear chain reaction would be self-sustaining once initiated. (Fortunately, he wasn’t hit by a car in the process.)
But I’m not Szilard. So I rationalized a reason not to exercise and sat on the couch with a book.
The remarkable human brain doesn’t shut down easily. With nothing else to do, most other mammals tend to doze off. But not us. It’s always on, trying to think of the next goal, the next move, the next whatever.
Having nothing to do sounds like a great idea, until you have nothing to do. It may be fine for a few days, but after a while you realize there’s only so long you can stare at the waves or mountains before your mind turns back to “what’s next.”
This isn’t a bad thing. Being bored is probably constructive. Without realizing it we use it to form new ideas and start new plans.
Maybe this is why we’re here. The mind that keeps working is a powerful tool, driving us forward in all walks of life. Perhaps it’s this feature that pushed the development of intelligence further and led us to form civilizations.
Perhaps it’s the real reason we keep moving forward.
Dr. Block has a solo neurology practice in Scottsdale, Ariz.
Book Review: Quality improvement in mental health care
Sunil Khushalani and Antonio DePaolo,
“Transforming Mental Healthcare: Applying Performance Improvement Methods to Mental Healthcare”
(London: Routledge, Taylor & Francis, 2022)
Since the publication of our book, “Lean Behavioral Health: The Kings County Hospital Story” (Oxford, England: Oxford University Press, 2014) almost a decade ago, “Transforming Mental Healthcare” is the first major book published about the use of a system for quality improvement across the health care continuum. That it has taken this long is probably surprising to those of us who have spent careers on trying to improve what is universally described as a system that is “broken” and in need of a major overhaul.
Every news cycle that reports mass violence typically spends a good bit of time talking about the failures of the mental health care system. One important lesson I learned when taking over the beleaguered Kings County (N.Y.) psychiatry service in 2009 (a department that has made extraordinary improvements over the years and is now exclaimed by the U.S. Department of Justice as “a model program”), is that the employees on the front line are often erroneously blamed for such failures.
The failure is systemic and usually starts at the top of the table of organization, not at the bottom. Dr. Khushalani and Dr. DePaolo have produced an excellent volume that should be purchased by every mental health care CEO and given “with thanks” to the local leaders overseeing the direct care of some of our nation’s most vulnerable patient populations.
The first part of “Transforming Mental Healthcare” provides an excellent overview of the current state of our mental health care system and its too numerous to name problems. This section could be a primer for all our legislators so their eyes can be opened to the failures on the ground that require their help in correcting. Many of the “failures” of our mental health care are societal failures – lack of affordable housing, access to care, reimbursement for care, gun access, etc. – and cannot be “fixed” by providers of care. Such problems are societal problems that call for societal and governmental solutions, and not only at the local level but from coast to coast.
The remainder of this easy to read and follow text provides many rich resources for the deliverers of mental health care. (e.g., plan-do-act, standard work, and A3 thinking).
The closing section focuses on leadership and culture – often overlooked to the detriment of any organization that doesn’t pay close attention to supporting both. Culture is cultivated and nourished by the organization’s leaders. Culture empowers staff to become problem solvers and agents of improvement. Empowered staff support and enrich their culture. Together a workplace that brings out the best of all its people is created, and burnout is held at bay.
“Transforming Mental Healthcare: Applying Performance Improvement Methods to Mental Healthcare” is a welcome and essential addition to the current morass, which is our mental health care delivery system, an oasis in the desert from which perhaps the lotus flower can emerge.
Dr. Merlino is emeritus professor of psychiatry, SUNY Downstate College of Medicine, Rhinebeck, N.Y., and formerly director of psychiatry at Kings County Hospital Center, Brooklyn, NY. He is the coauthor of “Lean Behavioral Health: The Kings County Hospital Story.” .
Sunil Khushalani and Antonio DePaolo,
“Transforming Mental Healthcare: Applying Performance Improvement Methods to Mental Healthcare”
(London: Routledge, Taylor & Francis, 2022)
Since the publication of our book, “Lean Behavioral Health: The Kings County Hospital Story” (Oxford, England: Oxford University Press, 2014) almost a decade ago, “Transforming Mental Healthcare” is the first major book published about the use of a system for quality improvement across the health care continuum. That it has taken this long is probably surprising to those of us who have spent careers on trying to improve what is universally described as a system that is “broken” and in need of a major overhaul.
Every news cycle that reports mass violence typically spends a good bit of time talking about the failures of the mental health care system. One important lesson I learned when taking over the beleaguered Kings County (N.Y.) psychiatry service in 2009 (a department that has made extraordinary improvements over the years and is now exclaimed by the U.S. Department of Justice as “a model program”), is that the employees on the front line are often erroneously blamed for such failures.
The failure is systemic and usually starts at the top of the table of organization, not at the bottom. Dr. Khushalani and Dr. DePaolo have produced an excellent volume that should be purchased by every mental health care CEO and given “with thanks” to the local leaders overseeing the direct care of some of our nation’s most vulnerable patient populations.
The first part of “Transforming Mental Healthcare” provides an excellent overview of the current state of our mental health care system and its too numerous to name problems. This section could be a primer for all our legislators so their eyes can be opened to the failures on the ground that require their help in correcting. Many of the “failures” of our mental health care are societal failures – lack of affordable housing, access to care, reimbursement for care, gun access, etc. – and cannot be “fixed” by providers of care. Such problems are societal problems that call for societal and governmental solutions, and not only at the local level but from coast to coast.
The remainder of this easy to read and follow text provides many rich resources for the deliverers of mental health care. (e.g., plan-do-act, standard work, and A3 thinking).
The closing section focuses on leadership and culture – often overlooked to the detriment of any organization that doesn’t pay close attention to supporting both. Culture is cultivated and nourished by the organization’s leaders. Culture empowers staff to become problem solvers and agents of improvement. Empowered staff support and enrich their culture. Together a workplace that brings out the best of all its people is created, and burnout is held at bay.
“Transforming Mental Healthcare: Applying Performance Improvement Methods to Mental Healthcare” is a welcome and essential addition to the current morass, which is our mental health care delivery system, an oasis in the desert from which perhaps the lotus flower can emerge.
Dr. Merlino is emeritus professor of psychiatry, SUNY Downstate College of Medicine, Rhinebeck, N.Y., and formerly director of psychiatry at Kings County Hospital Center, Brooklyn, NY. He is the coauthor of “Lean Behavioral Health: The Kings County Hospital Story.” .
Sunil Khushalani and Antonio DePaolo,
“Transforming Mental Healthcare: Applying Performance Improvement Methods to Mental Healthcare”
(London: Routledge, Taylor & Francis, 2022)
Since the publication of our book, “Lean Behavioral Health: The Kings County Hospital Story” (Oxford, England: Oxford University Press, 2014) almost a decade ago, “Transforming Mental Healthcare” is the first major book published about the use of a system for quality improvement across the health care continuum. That it has taken this long is probably surprising to those of us who have spent careers on trying to improve what is universally described as a system that is “broken” and in need of a major overhaul.
Every news cycle that reports mass violence typically spends a good bit of time talking about the failures of the mental health care system. One important lesson I learned when taking over the beleaguered Kings County (N.Y.) psychiatry service in 2009 (a department that has made extraordinary improvements over the years and is now exclaimed by the U.S. Department of Justice as “a model program”), is that the employees on the front line are often erroneously blamed for such failures.
The failure is systemic and usually starts at the top of the table of organization, not at the bottom. Dr. Khushalani and Dr. DePaolo have produced an excellent volume that should be purchased by every mental health care CEO and given “with thanks” to the local leaders overseeing the direct care of some of our nation’s most vulnerable patient populations.
The first part of “Transforming Mental Healthcare” provides an excellent overview of the current state of our mental health care system and its too numerous to name problems. This section could be a primer for all our legislators so their eyes can be opened to the failures on the ground that require their help in correcting. Many of the “failures” of our mental health care are societal failures – lack of affordable housing, access to care, reimbursement for care, gun access, etc. – and cannot be “fixed” by providers of care. Such problems are societal problems that call for societal and governmental solutions, and not only at the local level but from coast to coast.
The remainder of this easy to read and follow text provides many rich resources for the deliverers of mental health care. (e.g., plan-do-act, standard work, and A3 thinking).
The closing section focuses on leadership and culture – often overlooked to the detriment of any organization that doesn’t pay close attention to supporting both. Culture is cultivated and nourished by the organization’s leaders. Culture empowers staff to become problem solvers and agents of improvement. Empowered staff support and enrich their culture. Together a workplace that brings out the best of all its people is created, and burnout is held at bay.
“Transforming Mental Healthcare: Applying Performance Improvement Methods to Mental Healthcare” is a welcome and essential addition to the current morass, which is our mental health care delivery system, an oasis in the desert from which perhaps the lotus flower can emerge.
Dr. Merlino is emeritus professor of psychiatry, SUNY Downstate College of Medicine, Rhinebeck, N.Y., and formerly director of psychiatry at Kings County Hospital Center, Brooklyn, NY. He is the coauthor of “Lean Behavioral Health: The Kings County Hospital Story.” .
Doctors still overprescribing fluoroquinolones despite risks
When Amy Moser had a simple urinary tract infection in her late 20s, her doctor prescribed Cipro, a powerful antibiotic used to treat anthrax and some of the most fearsome bacterial infections.
Nearly 2 weeks after she finished her treatment, her left kneecap dislocated while she was trying on a swimsuit at a retail store. Shortly afterward, she had painful ligament ruptures in her wrists, then her shoulder dislocated, followed by three Achilles tendon tears.
“That’s when I fell apart,” says Ms. Moser, a Phoenix health blogger and book author. “From that moment on, for almost the next 2.5 years consistently, I had new tendon tears every few weeks.”
Ms. Moser’s doctors had no answer for what was causing her injuries, all of which required surgical fixes. A married mother of three, she was otherwise healthy and fit. So, after her third Achilles tear, she turned to the FDA’s website for answers. There, she found many warnings about side effects of Cipro, Levaquin, and other so-called fluoroquinolones, including risks for tendon and ligament injuries.
“When all the ruptures started to happen, my doctor kept asking me if I’d ever taken Levaquin, and every time I was like, ‘No.’ So I did what all doctors don’t want you to do: I Googled ‘Levaquin,’ ” she recalls.
Her search led to FDA warnings and articles about the possibility of tendon and ligament ruptures with fluroquinolones.
“That was the first time I’d ever even heard that word ‘fluroquinolones,’ and I found Cipro on that list ... and I realized that I’d just been prescribed that before everything started,” she says.
That was 12 years ago. Since then, the FDA has issued more warnings about fluoroquinolone risks. In that time, Ms. Moser, now 40, has had more than 30 surgeries to correct tendon ruptures and injuries, including a double-knee replacement this year.
“I am in chronic pain all the time,” she says. “I am chronically injured. I have a lot of tears that I’ve not fixed because they’re very complicated, and I don’t know if the rest of my body can handle the strain of recovering from those surgeries.”
Ms. Moser’s is hardly an isolated case. Since the 1980s, more than 60,000 patients have reported hundreds of thousands of serious events linked to fluoroquinolones to the FDA, including 6,575 reports of deaths.
The most common side effects were tendon rupture, as well as neurological and psychiatric symptoms. But experts estimate only 1%-10% of such events are reported to the FDA. That suggests that fluoroquinolones might have harmed hundreds of thousands of people in the United States alone, says Charles Bennett, MD, a hematologist at the University of South Carolina’s College of Pharmacy, Columbia.
Yet despite the many patient reports and FDA warnings on dangerous side effects, better treated with less risky antibiotics.
“There probably is overprescription by primary care doctors for urinary tract infections and respiratory infections, when there could be alternatives that are safer to use,” says Amesh Adalja, MD, an infectious disease specialist and senior scholar with the Johns Hopkins Center for Health Security.
“I would say that’s probably the case in the outpatient setting, not necessarily in the hospital setting or among infectious disease doctors ... but I think it’s important to say there are still some judicious uses of fluoroquinolones,” he says. “However, there probably is a lot of injudicious use of fluoroquinolones along with many other antibiotics in the primary care setting.”
FDA warnings on fluoroquinolones
Fluoroquinolones are a class of broad-spectrum antibiotics used for decades to treat certain bacterial infections.
FDA-approved fluoroquinolones include ciprofloxacin (Cipro), ciprofloxacin extended-release tablets, delafloxacin (Baxdela), gemifloxacin (Factive) levofloxacin (Levaquin), moxifloxacin (Avelox), and ofloxacin (Floxin). More than 60 generic versions of these brand-name medicines are also on the market, making them among the most prescribed antibiotics in the U.S.
Over the past 2 decades, a wide range of physical and mental health side effects have been tied to fluoroquinolones. As a result of these “adverse event reports” and research published in medical literature, the FDA has required an escalating series of warnings and safety labeling changes for doctors who prescribe these drugs.
- In 2008, the FDA first added a “black box” warning to fluoroquinolones, citing an increased risk of tendinitis and tendon rupture in patients prescribed these meds.
- In 2011, the agency required the warning label to include risks of worsening symptoms for those with myasthenia gravis, a chronic autoimmune disease that causes muscle weakness, vision problems, and speech problems.
- In 2013, regulators required updated labels noting the potential for irreversible peripheral neuropathy (serious nerve damage).
- In 2016, the FDA issued its strongest warning against the use of such antibiotics for simple bacterial infections – such as uncomplicated urinary tract infections (UTIs), acute sinusitis, and acute bronchitis – saying the “association of fluoroquinolones with disabling and potentially permanent side effects involving tendons, muscles, joints, nerves and the central nervous system ... outweighs the benefits for patients.”
- And in 2018, regulators required safety labeling changes to include warnings about the risks of aortic aneurysm – a life-threatening enlargement of the main vessel that delivers blood to the body – as well as mental health side effects and serious blood sugar disturbances.
But FDA regulators have stopped short of barring fluoroquinolone use in the treatment of bacterial infections, citing the benefits for certain conditions.
“For some patients, the benefits of fluoroquinolones may continue to outweigh the risks for treatment of serious bacterial infections, such as pneumonia or intra-abdominal infections,” said former FDA Commissioner Scott Gottlieb, MD, “but there are other serious, known risks associated with these strong antibiotics that must be carefully weighed when considering their use.”
In December 2021, a study published in the journal JAMA Network Open found the FDA’s warnings may have helped lower prescribing of the drugs in Medicare patients. But not all doctors have been responsive to those warnings, researchers found.
“An overall decline in change over time and an immediate change in fluoroquinolone prescribing was observed after the 2016 FDA warning,” the authors concluded. “Certain physicians, such as primary care physicians, were more responsive to FDA warnings than others. ... Findings of this study suggest that identifying the association of physician and organizational characteristics with fluoroquinolone prescribing practices could help in developing mechanisms for improving de-adoption.”
Some critics say the FDA should do more to spotlight the dangers of fluoroquinolones and require doctors and patients to sign checklist consent forms to show they are aware of the potential side effects of these drugs.
Rachel Brummert, a patient advocate who sits on an FDA consumer advisory board, believes the FDA needs to improve its communication to doctors on fluoroquinolone risks and get tougher with those who continue to inappropriately prescribe the drugs.
“I think there needs to be a system in place, where if something comes down from the FDA about a drug, the physician has to sign off on it, the patient has to sign off on it and mark that they understand that there are these ‘black box’ warnings,” says Ms. Brummert, 52, a representative on the FDA’s Medical Devices Advisory Committee.
As an example, she points to Australia’s medical laws requiring doctors and patients to sign a checklist before any fluoroquinolone prescription is approved.
“When a physician prescribes a fluoroquinolone antibiotic, there’s a checklist – does the patient have an infection, is it a simple infection, do they have allergies?” she notes. “And you can’t even get the prescription out – it won’t even print out, it won’t go into the system – unless you check all of the boxes. But we don’t do that here. We don’t have that type of system right now.”
Ms. Brummert says such a system might have prevented the harm from taking Levaquin her doctor prescribed for a suspected sinus infection in 2006.
Soon after she began taking the antibiotic, she ruptured her Achilles tendon, requiring surgery. By 2009, she’d had three ruptures, each needing surgical fixes. To date, she’s had more than 30 surgeries to correct tendon ruptures. She’s also had seizures, blood pressure issues, depression, chronic pain, and memory problems she attributes to taking Levaquin.
As it turns out, her doctor misdiagnosed her condition – a misstep that would have been averted with a system like Australia’s, which requires doctors to verify the presence of a bacterial infection through a simple test before prescribing a fluoroquinolone.
“When I got the Levaquin, it was for a suspected sinus infection that it turned out I didn’t even have in the first place,” she notes. “So, I took the Levaquin basically for nothing. But what I would have asked my doctor had I known is: ‘Why should I take something so strong for so simple an infection?’
“It seems common sense to me now that you don’t prescribe something that can kill anthrax for a simple sinus infection. It’s like an atom bomb killing a mosquito. I agree that there are uses for these drugs, but they are being overprescribed. And so, here I am 16 years later – I’m still rupturing, I’m still having surgery, and I’m still in pain – all for something I didn’t even need medicine for in the first place.”
Should guidelines be stronger?
So, why are so many doctors continuing to prescribe fluoroquinolones for simple infections? Dr. Adalja and other experts say several things are at work.
For one thing, Dr. Adalja notes, fluoroquinolones are broad-spectrum antibiotics that are effective against dangerous germs, including “gram-negative” bacterial infections, and are “100% bioavailable.” That means they are as effective when given in pill form as they are if put directly into a vein. So they can be used in an outpatient setting or to allow a patient to be discharged from a hospital sooner because they don’t need an IV to receive treatment.
“There are still some uses for these drugs because they are so bioavailable, and I think that drives some of the use, and those are legitimate uses, knowing that there are risks when you do it,” he says. “But no drug is without risks, and you have to weigh risks and benefits – that’s what medicine is about: deciding what the best drug is for a patient.”
But Dr. Adalja says the overprescription of fluoroquinolones is part of the larger trend of antibiotic overuse. That is driving up antibiotic resistance, which in turn is another thing leading doctors to turn to Cipro and other fluoroquinolones after other drugs have proven ineffective.
“You can’t separate this from the fact that 80% of antibiotic prescriptions in the outpatient setting are probably illegitimate or not warranted,” he notes. “And because fluoroquinolones are highly effective drugs against certain pathogens, they are the go-to [drug] for many people who are prescribing antibiotics.”
That’s why patients should be wary whenever a doctor prescribes a fluoroquinolone, or any drug to treat a suspected infection, he says.
“Any time a patient is getting prescribed an antibiotic by a physician, they should ask: ‘Do I really need this antibiotic?’ That should be the first question they ask,” he advises. “And if they’re getting a fluoroquinolone, they may want to ask: ‘Is this the best antibiotic for me?’ ”
What you can do
Ms. Brummert and Ms. Moser say they are sharing their stories to raise awareness of the dangers of fluoroquinolones.
Ms. Moser has published a book on her experiences, “The Magnificent Story of a Lame Author,” and provides a wealth of consumer resources on her blog: Mountains and Mustard Seeds.
“As much as I hate what has happened to me, it has put me in a place where I am glad that I can inform other patients,” she says.
Ms. Brummert supplements her advocacy work as an FDA adviser with useful materials she provides on her website: Drugwatch.com.
“Pain into purpose – that’s what I call it,” she says. “I can’t change what happened to me, but I can warn others.”
The upshot for patients?
- the FDA’s Drug Safety Communication on Fluoroquinolones online to learn more about the risks and benefits of these powerful antibiotics.
- If you believe you’ve been harmed by fluoroquinolones, MedWatch website to report your experiences.
Ms. Brummert also advises patients to ask 12 critical questions of any doctor who wants to prescribe a fluoroquinolone, including the following listed on her website:
- For what condition is this medication prescribed, and is there another drug specific to my condition?
- What are the risks associated with this medication, and do the benefits outweigh them?
- Will this medication interact with my other drugs and/or other health conditions?
- What are the “boxed” warnings for this medication, and where can I report adverse events?
“I would also do my own research,” she says. “I wouldn’t just take a prescription from a physician and just say, ‘OK, doctor knows best.’ ”
Ms. Moser agrees that you have to be your own patient advocate and not simply take a doctor’s advice on any medical issue without having a deeper conversation.
“I’ve had arguments with doctors who legitimately did not believe me when I told them what happened to me,” she says. “And I actually told them, ‘Go get your Physicians’ Desk Reference [for prescription drugs]’ and they opened the book in front of me and read the warnings. Obviously, they had not been keeping up with the added warnings. So, I do think that doctors do need to be better informed.”
“So, yes, it’s the FDA’s responsibility, but it is also the doctors’ responsibility to make sure that they’re watching out for the side effects and they’re reporting them when their patients come up with them and making those connections.”
A version of this article first appeared on WebMD.com.
When Amy Moser had a simple urinary tract infection in her late 20s, her doctor prescribed Cipro, a powerful antibiotic used to treat anthrax and some of the most fearsome bacterial infections.
Nearly 2 weeks after she finished her treatment, her left kneecap dislocated while she was trying on a swimsuit at a retail store. Shortly afterward, she had painful ligament ruptures in her wrists, then her shoulder dislocated, followed by three Achilles tendon tears.
“That’s when I fell apart,” says Ms. Moser, a Phoenix health blogger and book author. “From that moment on, for almost the next 2.5 years consistently, I had new tendon tears every few weeks.”
Ms. Moser’s doctors had no answer for what was causing her injuries, all of which required surgical fixes. A married mother of three, she was otherwise healthy and fit. So, after her third Achilles tear, she turned to the FDA’s website for answers. There, she found many warnings about side effects of Cipro, Levaquin, and other so-called fluoroquinolones, including risks for tendon and ligament injuries.
“When all the ruptures started to happen, my doctor kept asking me if I’d ever taken Levaquin, and every time I was like, ‘No.’ So I did what all doctors don’t want you to do: I Googled ‘Levaquin,’ ” she recalls.
Her search led to FDA warnings and articles about the possibility of tendon and ligament ruptures with fluroquinolones.
“That was the first time I’d ever even heard that word ‘fluroquinolones,’ and I found Cipro on that list ... and I realized that I’d just been prescribed that before everything started,” she says.
That was 12 years ago. Since then, the FDA has issued more warnings about fluoroquinolone risks. In that time, Ms. Moser, now 40, has had more than 30 surgeries to correct tendon ruptures and injuries, including a double-knee replacement this year.
“I am in chronic pain all the time,” she says. “I am chronically injured. I have a lot of tears that I’ve not fixed because they’re very complicated, and I don’t know if the rest of my body can handle the strain of recovering from those surgeries.”
Ms. Moser’s is hardly an isolated case. Since the 1980s, more than 60,000 patients have reported hundreds of thousands of serious events linked to fluoroquinolones to the FDA, including 6,575 reports of deaths.
The most common side effects were tendon rupture, as well as neurological and psychiatric symptoms. But experts estimate only 1%-10% of such events are reported to the FDA. That suggests that fluoroquinolones might have harmed hundreds of thousands of people in the United States alone, says Charles Bennett, MD, a hematologist at the University of South Carolina’s College of Pharmacy, Columbia.
Yet despite the many patient reports and FDA warnings on dangerous side effects, better treated with less risky antibiotics.
“There probably is overprescription by primary care doctors for urinary tract infections and respiratory infections, when there could be alternatives that are safer to use,” says Amesh Adalja, MD, an infectious disease specialist and senior scholar with the Johns Hopkins Center for Health Security.
“I would say that’s probably the case in the outpatient setting, not necessarily in the hospital setting or among infectious disease doctors ... but I think it’s important to say there are still some judicious uses of fluoroquinolones,” he says. “However, there probably is a lot of injudicious use of fluoroquinolones along with many other antibiotics in the primary care setting.”
FDA warnings on fluoroquinolones
Fluoroquinolones are a class of broad-spectrum antibiotics used for decades to treat certain bacterial infections.
FDA-approved fluoroquinolones include ciprofloxacin (Cipro), ciprofloxacin extended-release tablets, delafloxacin (Baxdela), gemifloxacin (Factive) levofloxacin (Levaquin), moxifloxacin (Avelox), and ofloxacin (Floxin). More than 60 generic versions of these brand-name medicines are also on the market, making them among the most prescribed antibiotics in the U.S.
Over the past 2 decades, a wide range of physical and mental health side effects have been tied to fluoroquinolones. As a result of these “adverse event reports” and research published in medical literature, the FDA has required an escalating series of warnings and safety labeling changes for doctors who prescribe these drugs.
- In 2008, the FDA first added a “black box” warning to fluoroquinolones, citing an increased risk of tendinitis and tendon rupture in patients prescribed these meds.
- In 2011, the agency required the warning label to include risks of worsening symptoms for those with myasthenia gravis, a chronic autoimmune disease that causes muscle weakness, vision problems, and speech problems.
- In 2013, regulators required updated labels noting the potential for irreversible peripheral neuropathy (serious nerve damage).
- In 2016, the FDA issued its strongest warning against the use of such antibiotics for simple bacterial infections – such as uncomplicated urinary tract infections (UTIs), acute sinusitis, and acute bronchitis – saying the “association of fluoroquinolones with disabling and potentially permanent side effects involving tendons, muscles, joints, nerves and the central nervous system ... outweighs the benefits for patients.”
- And in 2018, regulators required safety labeling changes to include warnings about the risks of aortic aneurysm – a life-threatening enlargement of the main vessel that delivers blood to the body – as well as mental health side effects and serious blood sugar disturbances.
But FDA regulators have stopped short of barring fluoroquinolone use in the treatment of bacterial infections, citing the benefits for certain conditions.
“For some patients, the benefits of fluoroquinolones may continue to outweigh the risks for treatment of serious bacterial infections, such as pneumonia or intra-abdominal infections,” said former FDA Commissioner Scott Gottlieb, MD, “but there are other serious, known risks associated with these strong antibiotics that must be carefully weighed when considering their use.”
In December 2021, a study published in the journal JAMA Network Open found the FDA’s warnings may have helped lower prescribing of the drugs in Medicare patients. But not all doctors have been responsive to those warnings, researchers found.
“An overall decline in change over time and an immediate change in fluoroquinolone prescribing was observed after the 2016 FDA warning,” the authors concluded. “Certain physicians, such as primary care physicians, were more responsive to FDA warnings than others. ... Findings of this study suggest that identifying the association of physician and organizational characteristics with fluoroquinolone prescribing practices could help in developing mechanisms for improving de-adoption.”
Some critics say the FDA should do more to spotlight the dangers of fluoroquinolones and require doctors and patients to sign checklist consent forms to show they are aware of the potential side effects of these drugs.
Rachel Brummert, a patient advocate who sits on an FDA consumer advisory board, believes the FDA needs to improve its communication to doctors on fluoroquinolone risks and get tougher with those who continue to inappropriately prescribe the drugs.
“I think there needs to be a system in place, where if something comes down from the FDA about a drug, the physician has to sign off on it, the patient has to sign off on it and mark that they understand that there are these ‘black box’ warnings,” says Ms. Brummert, 52, a representative on the FDA’s Medical Devices Advisory Committee.
As an example, she points to Australia’s medical laws requiring doctors and patients to sign a checklist before any fluoroquinolone prescription is approved.
“When a physician prescribes a fluoroquinolone antibiotic, there’s a checklist – does the patient have an infection, is it a simple infection, do they have allergies?” she notes. “And you can’t even get the prescription out – it won’t even print out, it won’t go into the system – unless you check all of the boxes. But we don’t do that here. We don’t have that type of system right now.”
Ms. Brummert says such a system might have prevented the harm from taking Levaquin her doctor prescribed for a suspected sinus infection in 2006.
Soon after she began taking the antibiotic, she ruptured her Achilles tendon, requiring surgery. By 2009, she’d had three ruptures, each needing surgical fixes. To date, she’s had more than 30 surgeries to correct tendon ruptures. She’s also had seizures, blood pressure issues, depression, chronic pain, and memory problems she attributes to taking Levaquin.
As it turns out, her doctor misdiagnosed her condition – a misstep that would have been averted with a system like Australia’s, which requires doctors to verify the presence of a bacterial infection through a simple test before prescribing a fluoroquinolone.
“When I got the Levaquin, it was for a suspected sinus infection that it turned out I didn’t even have in the first place,” she notes. “So, I took the Levaquin basically for nothing. But what I would have asked my doctor had I known is: ‘Why should I take something so strong for so simple an infection?’
“It seems common sense to me now that you don’t prescribe something that can kill anthrax for a simple sinus infection. It’s like an atom bomb killing a mosquito. I agree that there are uses for these drugs, but they are being overprescribed. And so, here I am 16 years later – I’m still rupturing, I’m still having surgery, and I’m still in pain – all for something I didn’t even need medicine for in the first place.”
Should guidelines be stronger?
So, why are so many doctors continuing to prescribe fluoroquinolones for simple infections? Dr. Adalja and other experts say several things are at work.
For one thing, Dr. Adalja notes, fluoroquinolones are broad-spectrum antibiotics that are effective against dangerous germs, including “gram-negative” bacterial infections, and are “100% bioavailable.” That means they are as effective when given in pill form as they are if put directly into a vein. So they can be used in an outpatient setting or to allow a patient to be discharged from a hospital sooner because they don’t need an IV to receive treatment.
“There are still some uses for these drugs because they are so bioavailable, and I think that drives some of the use, and those are legitimate uses, knowing that there are risks when you do it,” he says. “But no drug is without risks, and you have to weigh risks and benefits – that’s what medicine is about: deciding what the best drug is for a patient.”
But Dr. Adalja says the overprescription of fluoroquinolones is part of the larger trend of antibiotic overuse. That is driving up antibiotic resistance, which in turn is another thing leading doctors to turn to Cipro and other fluoroquinolones after other drugs have proven ineffective.
“You can’t separate this from the fact that 80% of antibiotic prescriptions in the outpatient setting are probably illegitimate or not warranted,” he notes. “And because fluoroquinolones are highly effective drugs against certain pathogens, they are the go-to [drug] for many people who are prescribing antibiotics.”
That’s why patients should be wary whenever a doctor prescribes a fluoroquinolone, or any drug to treat a suspected infection, he says.
“Any time a patient is getting prescribed an antibiotic by a physician, they should ask: ‘Do I really need this antibiotic?’ That should be the first question they ask,” he advises. “And if they’re getting a fluoroquinolone, they may want to ask: ‘Is this the best antibiotic for me?’ ”
What you can do
Ms. Brummert and Ms. Moser say they are sharing their stories to raise awareness of the dangers of fluoroquinolones.
Ms. Moser has published a book on her experiences, “The Magnificent Story of a Lame Author,” and provides a wealth of consumer resources on her blog: Mountains and Mustard Seeds.
“As much as I hate what has happened to me, it has put me in a place where I am glad that I can inform other patients,” she says.
Ms. Brummert supplements her advocacy work as an FDA adviser with useful materials she provides on her website: Drugwatch.com.
“Pain into purpose – that’s what I call it,” she says. “I can’t change what happened to me, but I can warn others.”
The upshot for patients?
- the FDA’s Drug Safety Communication on Fluoroquinolones online to learn more about the risks and benefits of these powerful antibiotics.
- If you believe you’ve been harmed by fluoroquinolones, MedWatch website to report your experiences.
Ms. Brummert also advises patients to ask 12 critical questions of any doctor who wants to prescribe a fluoroquinolone, including the following listed on her website:
- For what condition is this medication prescribed, and is there another drug specific to my condition?
- What are the risks associated with this medication, and do the benefits outweigh them?
- Will this medication interact with my other drugs and/or other health conditions?
- What are the “boxed” warnings for this medication, and where can I report adverse events?
“I would also do my own research,” she says. “I wouldn’t just take a prescription from a physician and just say, ‘OK, doctor knows best.’ ”
Ms. Moser agrees that you have to be your own patient advocate and not simply take a doctor’s advice on any medical issue without having a deeper conversation.
“I’ve had arguments with doctors who legitimately did not believe me when I told them what happened to me,” she says. “And I actually told them, ‘Go get your Physicians’ Desk Reference [for prescription drugs]’ and they opened the book in front of me and read the warnings. Obviously, they had not been keeping up with the added warnings. So, I do think that doctors do need to be better informed.”
“So, yes, it’s the FDA’s responsibility, but it is also the doctors’ responsibility to make sure that they’re watching out for the side effects and they’re reporting them when their patients come up with them and making those connections.”
A version of this article first appeared on WebMD.com.
When Amy Moser had a simple urinary tract infection in her late 20s, her doctor prescribed Cipro, a powerful antibiotic used to treat anthrax and some of the most fearsome bacterial infections.
Nearly 2 weeks after she finished her treatment, her left kneecap dislocated while she was trying on a swimsuit at a retail store. Shortly afterward, she had painful ligament ruptures in her wrists, then her shoulder dislocated, followed by three Achilles tendon tears.
“That’s when I fell apart,” says Ms. Moser, a Phoenix health blogger and book author. “From that moment on, for almost the next 2.5 years consistently, I had new tendon tears every few weeks.”
Ms. Moser’s doctors had no answer for what was causing her injuries, all of which required surgical fixes. A married mother of three, she was otherwise healthy and fit. So, after her third Achilles tear, she turned to the FDA’s website for answers. There, she found many warnings about side effects of Cipro, Levaquin, and other so-called fluoroquinolones, including risks for tendon and ligament injuries.
“When all the ruptures started to happen, my doctor kept asking me if I’d ever taken Levaquin, and every time I was like, ‘No.’ So I did what all doctors don’t want you to do: I Googled ‘Levaquin,’ ” she recalls.
Her search led to FDA warnings and articles about the possibility of tendon and ligament ruptures with fluroquinolones.
“That was the first time I’d ever even heard that word ‘fluroquinolones,’ and I found Cipro on that list ... and I realized that I’d just been prescribed that before everything started,” she says.
That was 12 years ago. Since then, the FDA has issued more warnings about fluoroquinolone risks. In that time, Ms. Moser, now 40, has had more than 30 surgeries to correct tendon ruptures and injuries, including a double-knee replacement this year.
“I am in chronic pain all the time,” she says. “I am chronically injured. I have a lot of tears that I’ve not fixed because they’re very complicated, and I don’t know if the rest of my body can handle the strain of recovering from those surgeries.”
Ms. Moser’s is hardly an isolated case. Since the 1980s, more than 60,000 patients have reported hundreds of thousands of serious events linked to fluoroquinolones to the FDA, including 6,575 reports of deaths.
The most common side effects were tendon rupture, as well as neurological and psychiatric symptoms. But experts estimate only 1%-10% of such events are reported to the FDA. That suggests that fluoroquinolones might have harmed hundreds of thousands of people in the United States alone, says Charles Bennett, MD, a hematologist at the University of South Carolina’s College of Pharmacy, Columbia.
Yet despite the many patient reports and FDA warnings on dangerous side effects, better treated with less risky antibiotics.
“There probably is overprescription by primary care doctors for urinary tract infections and respiratory infections, when there could be alternatives that are safer to use,” says Amesh Adalja, MD, an infectious disease specialist and senior scholar with the Johns Hopkins Center for Health Security.
“I would say that’s probably the case in the outpatient setting, not necessarily in the hospital setting or among infectious disease doctors ... but I think it’s important to say there are still some judicious uses of fluoroquinolones,” he says. “However, there probably is a lot of injudicious use of fluoroquinolones along with many other antibiotics in the primary care setting.”
FDA warnings on fluoroquinolones
Fluoroquinolones are a class of broad-spectrum antibiotics used for decades to treat certain bacterial infections.
FDA-approved fluoroquinolones include ciprofloxacin (Cipro), ciprofloxacin extended-release tablets, delafloxacin (Baxdela), gemifloxacin (Factive) levofloxacin (Levaquin), moxifloxacin (Avelox), and ofloxacin (Floxin). More than 60 generic versions of these brand-name medicines are also on the market, making them among the most prescribed antibiotics in the U.S.
Over the past 2 decades, a wide range of physical and mental health side effects have been tied to fluoroquinolones. As a result of these “adverse event reports” and research published in medical literature, the FDA has required an escalating series of warnings and safety labeling changes for doctors who prescribe these drugs.
- In 2008, the FDA first added a “black box” warning to fluoroquinolones, citing an increased risk of tendinitis and tendon rupture in patients prescribed these meds.
- In 2011, the agency required the warning label to include risks of worsening symptoms for those with myasthenia gravis, a chronic autoimmune disease that causes muscle weakness, vision problems, and speech problems.
- In 2013, regulators required updated labels noting the potential for irreversible peripheral neuropathy (serious nerve damage).
- In 2016, the FDA issued its strongest warning against the use of such antibiotics for simple bacterial infections – such as uncomplicated urinary tract infections (UTIs), acute sinusitis, and acute bronchitis – saying the “association of fluoroquinolones with disabling and potentially permanent side effects involving tendons, muscles, joints, nerves and the central nervous system ... outweighs the benefits for patients.”
- And in 2018, regulators required safety labeling changes to include warnings about the risks of aortic aneurysm – a life-threatening enlargement of the main vessel that delivers blood to the body – as well as mental health side effects and serious blood sugar disturbances.
But FDA regulators have stopped short of barring fluoroquinolone use in the treatment of bacterial infections, citing the benefits for certain conditions.
“For some patients, the benefits of fluoroquinolones may continue to outweigh the risks for treatment of serious bacterial infections, such as pneumonia or intra-abdominal infections,” said former FDA Commissioner Scott Gottlieb, MD, “but there are other serious, known risks associated with these strong antibiotics that must be carefully weighed when considering their use.”
In December 2021, a study published in the journal JAMA Network Open found the FDA’s warnings may have helped lower prescribing of the drugs in Medicare patients. But not all doctors have been responsive to those warnings, researchers found.
“An overall decline in change over time and an immediate change in fluoroquinolone prescribing was observed after the 2016 FDA warning,” the authors concluded. “Certain physicians, such as primary care physicians, were more responsive to FDA warnings than others. ... Findings of this study suggest that identifying the association of physician and organizational characteristics with fluoroquinolone prescribing practices could help in developing mechanisms for improving de-adoption.”
Some critics say the FDA should do more to spotlight the dangers of fluoroquinolones and require doctors and patients to sign checklist consent forms to show they are aware of the potential side effects of these drugs.
Rachel Brummert, a patient advocate who sits on an FDA consumer advisory board, believes the FDA needs to improve its communication to doctors on fluoroquinolone risks and get tougher with those who continue to inappropriately prescribe the drugs.
“I think there needs to be a system in place, where if something comes down from the FDA about a drug, the physician has to sign off on it, the patient has to sign off on it and mark that they understand that there are these ‘black box’ warnings,” says Ms. Brummert, 52, a representative on the FDA’s Medical Devices Advisory Committee.
As an example, she points to Australia’s medical laws requiring doctors and patients to sign a checklist before any fluoroquinolone prescription is approved.
“When a physician prescribes a fluoroquinolone antibiotic, there’s a checklist – does the patient have an infection, is it a simple infection, do they have allergies?” she notes. “And you can’t even get the prescription out – it won’t even print out, it won’t go into the system – unless you check all of the boxes. But we don’t do that here. We don’t have that type of system right now.”
Ms. Brummert says such a system might have prevented the harm from taking Levaquin her doctor prescribed for a suspected sinus infection in 2006.
Soon after she began taking the antibiotic, she ruptured her Achilles tendon, requiring surgery. By 2009, she’d had three ruptures, each needing surgical fixes. To date, she’s had more than 30 surgeries to correct tendon ruptures. She’s also had seizures, blood pressure issues, depression, chronic pain, and memory problems she attributes to taking Levaquin.
As it turns out, her doctor misdiagnosed her condition – a misstep that would have been averted with a system like Australia’s, which requires doctors to verify the presence of a bacterial infection through a simple test before prescribing a fluoroquinolone.
“When I got the Levaquin, it was for a suspected sinus infection that it turned out I didn’t even have in the first place,” she notes. “So, I took the Levaquin basically for nothing. But what I would have asked my doctor had I known is: ‘Why should I take something so strong for so simple an infection?’
“It seems common sense to me now that you don’t prescribe something that can kill anthrax for a simple sinus infection. It’s like an atom bomb killing a mosquito. I agree that there are uses for these drugs, but they are being overprescribed. And so, here I am 16 years later – I’m still rupturing, I’m still having surgery, and I’m still in pain – all for something I didn’t even need medicine for in the first place.”
Should guidelines be stronger?
So, why are so many doctors continuing to prescribe fluoroquinolones for simple infections? Dr. Adalja and other experts say several things are at work.
For one thing, Dr. Adalja notes, fluoroquinolones are broad-spectrum antibiotics that are effective against dangerous germs, including “gram-negative” bacterial infections, and are “100% bioavailable.” That means they are as effective when given in pill form as they are if put directly into a vein. So they can be used in an outpatient setting or to allow a patient to be discharged from a hospital sooner because they don’t need an IV to receive treatment.
“There are still some uses for these drugs because they are so bioavailable, and I think that drives some of the use, and those are legitimate uses, knowing that there are risks when you do it,” he says. “But no drug is without risks, and you have to weigh risks and benefits – that’s what medicine is about: deciding what the best drug is for a patient.”
But Dr. Adalja says the overprescription of fluoroquinolones is part of the larger trend of antibiotic overuse. That is driving up antibiotic resistance, which in turn is another thing leading doctors to turn to Cipro and other fluoroquinolones after other drugs have proven ineffective.
“You can’t separate this from the fact that 80% of antibiotic prescriptions in the outpatient setting are probably illegitimate or not warranted,” he notes. “And because fluoroquinolones are highly effective drugs against certain pathogens, they are the go-to [drug] for many people who are prescribing antibiotics.”
That’s why patients should be wary whenever a doctor prescribes a fluoroquinolone, or any drug to treat a suspected infection, he says.
“Any time a patient is getting prescribed an antibiotic by a physician, they should ask: ‘Do I really need this antibiotic?’ That should be the first question they ask,” he advises. “And if they’re getting a fluoroquinolone, they may want to ask: ‘Is this the best antibiotic for me?’ ”
What you can do
Ms. Brummert and Ms. Moser say they are sharing their stories to raise awareness of the dangers of fluoroquinolones.
Ms. Moser has published a book on her experiences, “The Magnificent Story of a Lame Author,” and provides a wealth of consumer resources on her blog: Mountains and Mustard Seeds.
“As much as I hate what has happened to me, it has put me in a place where I am glad that I can inform other patients,” she says.
Ms. Brummert supplements her advocacy work as an FDA adviser with useful materials she provides on her website: Drugwatch.com.
“Pain into purpose – that’s what I call it,” she says. “I can’t change what happened to me, but I can warn others.”
The upshot for patients?
- the FDA’s Drug Safety Communication on Fluoroquinolones online to learn more about the risks and benefits of these powerful antibiotics.
- If you believe you’ve been harmed by fluoroquinolones, MedWatch website to report your experiences.
Ms. Brummert also advises patients to ask 12 critical questions of any doctor who wants to prescribe a fluoroquinolone, including the following listed on her website:
- For what condition is this medication prescribed, and is there another drug specific to my condition?
- What are the risks associated with this medication, and do the benefits outweigh them?
- Will this medication interact with my other drugs and/or other health conditions?
- What are the “boxed” warnings for this medication, and where can I report adverse events?
“I would also do my own research,” she says. “I wouldn’t just take a prescription from a physician and just say, ‘OK, doctor knows best.’ ”
Ms. Moser agrees that you have to be your own patient advocate and not simply take a doctor’s advice on any medical issue without having a deeper conversation.
“I’ve had arguments with doctors who legitimately did not believe me when I told them what happened to me,” she says. “And I actually told them, ‘Go get your Physicians’ Desk Reference [for prescription drugs]’ and they opened the book in front of me and read the warnings. Obviously, they had not been keeping up with the added warnings. So, I do think that doctors do need to be better informed.”
“So, yes, it’s the FDA’s responsibility, but it is also the doctors’ responsibility to make sure that they’re watching out for the side effects and they’re reporting them when their patients come up with them and making those connections.”
A version of this article first appeared on WebMD.com.
What is palliative care and what’s new in practicing this type of medicine?
The World Health Organization defines palliative care as “an approach that improves the quality of life of patients (adults and children) and their families who are facing problems associated with life-threatening illness. It prevents and relieves suffering through the early identification, correct assessment, and treatment of pain and other problems, whether physical, psychosocial or spiritual.”1
The common misperception is that palliative care is only for those at end of life or only in the advanced stages of their illness. However, palliative care is ideally most helpful following individuals from diagnosis through their illness trajectory. Another misperception is that palliative care and hospice are the same thing. Though all hospice is palliative care, all palliative care is not hospice. Both palliative care and hospice provide care for individuals facing a serious illness and focus on the same philosophy of care, but palliative care can be initiated at any stage of illness, even if the goal is to pursue curative and life-prolonging therapies/interventions.
In contrast, hospice is considered for those who are at the end of life and are usually not pursuing life-prolonging therapies or interventions, instead focusing on comfort, symptom management, and optimization of quality of life.
Though there is a growing need for palliative care, there is a shortage of specialist palliative care providers. Much of the palliative care needs can be met by all providers who can offer basic symptom management, identification surrounding goals of care and discussions of advance care planning, and understanding of illness/prognosis and treatment options, which is called primary palliative care.2 In fact, two-thirds of patients with a serious illness other than cancer prefer discussion of end-of-life care or advance care planning with their primary care providers.3
Referral to specialty palliative care should be considered when there are more complexities to symptom/pain management and goals of care/end of life, transition to hospice, or complex communication dynamics.4
Though specialty palliative care was shown to be more comprehensive, both primary palliative care and specialty palliative care have led to improvements in the quality of life in individuals living with serious illness.5 Early integration of palliative care into routine care has been shown to improve symptom burden, mood, quality of life, survival, and health care costs.6
Updates in alternative and complementary therapies to palliative care
There are several alternative and complementary therapies to palliative care, including cannabis and psychedelics. These therapies are becoming or may become a familiar part of medical therapies that are listed in a patient’s history as part of their medical regimen, especially as more states continue to legalize and/or decriminalize the use of these alternative therapies for recreational or medicinal use.
Both cannabis and psychedelics have a longstanding history of therapeutic and holistic use. Cannabis has been used to manage symptoms such as pain since the 16th and 17th century.7 In palliative care, more patients may turn to various forms of cannabis as a source of relief from symptoms and suffering as their focus shifts more to quality of life.
Even with the increasing popularity of the use of cannabis among seriously ill patients, there is still a lack of evidence of the benefits of medical cannabis use in palliative care, and there is a lack of standardization of type of cannabis used and state regulations regarding their use.7
A recent systematic review found that despite the reported positive treatment effects of cannabis in palliative care, the results of the studies were conflicting. This highlights the need for further high-quality research to determine whether cannabis products are an effective treatment in palliative care patients.8
One limitation to note is that the majority of the included studies focused on cannabis use in patients with cancer for cancer-related symptoms. Few studies included patients with other serious conditions.
Psychedelics
There is evidence that psychedelic assisted therapy (PAT) is a safe and effective treatment for individuals with refractory depression, posttraumatic stress disorder, and substance use disorder.9 Plus, there have been ample studies providing support that PAT improves symptoms such as refractory anxiety/depression, demoralization, and existential distress in seriously ill patients, thus improving their quality of life and overall well-being.9
Nine U.S. cities and the State of Oregon have decriminalized or legalized the psychedelic psilocybin, based on the medical benefits patients have experienced evidenced from using it.10
In light of the increasing interest in PAT, Dr. Ira Byock provided the following points on what “all clinicians should know as they enter this uncharted territory”:
- Psychedelics have been around for a long time.
- Psychedelic-assisted therapies’ therapeutic effects are experiential.
- There are a variety of terms for specific categories of psychedelic compounds.
- Some palliative care teams are already caring for patients who undergo psychedelic experiences.
- Use of psychedelics should be well-observed by a skilled clinician with expertise.
I am hoping this provides a general refresher on palliative care and an overview of updates to alternative and complementary therapies for patients living with serious illness.9
Dr. Kang is a geriatrician and palliative care provider at the University of Washington, Seattle in the division of geriatrics and gerontology. She has no conflicts related to the content of this piece.
References
1. World Health Organization. Palliative care. 2020 Aug 5..
2. Weissman DE and Meier DE. Identifying patients in need of a palliative care assessment in the hospital setting a consensus report from the center to advance palliative care. J Palliat Med. 2011;14(1):17-23.
3. Sherry D et al. Is primary care physician involvement associated with earlier advance care planning? A study of patients in an academic primary care setting. J Palliat Med. 2022;25(1):75-80.
4. Quill TE and Abernethy AP. Generalist plus specialist palliative care-creating a more sustainable model. N Engl J Med. 2013;368:1173-75.
5. Ernecoff NC et al. Comparing specialty and primary palliative care interventions: Analysis of a systematic review. J Palliat Med. 2020;23(3):389-96.
6. Temmel JS et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2011;363:733-42.
7. Kogan M and Sexton M. Medical cannabis: A new old tool for palliative care. J Altern Complement Med . 2020 Sep;26(9):776-8.
8. Doppen M et al. Cannabis in palliative care: A systematic review of the current evidence. J Pain Symptom Manage. 2022 Jun 12;S0885-3924(22)00760-6.
9. Byock I. Psychedelics for serious illness: Five things clinicians need to know. The Center to Advance Palliative Care. Psychedelics for Serious Illness, Palliative in Practice, Center to Advance Palliative Care (capc.org). June 13, 2022.
10. Marks M. A strategy for rescheduling psilocybin. Scientific American. Oct. 11, 2021.
The World Health Organization defines palliative care as “an approach that improves the quality of life of patients (adults and children) and their families who are facing problems associated with life-threatening illness. It prevents and relieves suffering through the early identification, correct assessment, and treatment of pain and other problems, whether physical, psychosocial or spiritual.”1
The common misperception is that palliative care is only for those at end of life or only in the advanced stages of their illness. However, palliative care is ideally most helpful following individuals from diagnosis through their illness trajectory. Another misperception is that palliative care and hospice are the same thing. Though all hospice is palliative care, all palliative care is not hospice. Both palliative care and hospice provide care for individuals facing a serious illness and focus on the same philosophy of care, but palliative care can be initiated at any stage of illness, even if the goal is to pursue curative and life-prolonging therapies/interventions.
In contrast, hospice is considered for those who are at the end of life and are usually not pursuing life-prolonging therapies or interventions, instead focusing on comfort, symptom management, and optimization of quality of life.
Though there is a growing need for palliative care, there is a shortage of specialist palliative care providers. Much of the palliative care needs can be met by all providers who can offer basic symptom management, identification surrounding goals of care and discussions of advance care planning, and understanding of illness/prognosis and treatment options, which is called primary palliative care.2 In fact, two-thirds of patients with a serious illness other than cancer prefer discussion of end-of-life care or advance care planning with their primary care providers.3
Referral to specialty palliative care should be considered when there are more complexities to symptom/pain management and goals of care/end of life, transition to hospice, or complex communication dynamics.4
Though specialty palliative care was shown to be more comprehensive, both primary palliative care and specialty palliative care have led to improvements in the quality of life in individuals living with serious illness.5 Early integration of palliative care into routine care has been shown to improve symptom burden, mood, quality of life, survival, and health care costs.6
Updates in alternative and complementary therapies to palliative care
There are several alternative and complementary therapies to palliative care, including cannabis and psychedelics. These therapies are becoming or may become a familiar part of medical therapies that are listed in a patient’s history as part of their medical regimen, especially as more states continue to legalize and/or decriminalize the use of these alternative therapies for recreational or medicinal use.
Both cannabis and psychedelics have a longstanding history of therapeutic and holistic use. Cannabis has been used to manage symptoms such as pain since the 16th and 17th century.7 In palliative care, more patients may turn to various forms of cannabis as a source of relief from symptoms and suffering as their focus shifts more to quality of life.
Even with the increasing popularity of the use of cannabis among seriously ill patients, there is still a lack of evidence of the benefits of medical cannabis use in palliative care, and there is a lack of standardization of type of cannabis used and state regulations regarding their use.7
A recent systematic review found that despite the reported positive treatment effects of cannabis in palliative care, the results of the studies were conflicting. This highlights the need for further high-quality research to determine whether cannabis products are an effective treatment in palliative care patients.8
One limitation to note is that the majority of the included studies focused on cannabis use in patients with cancer for cancer-related symptoms. Few studies included patients with other serious conditions.
Psychedelics
There is evidence that psychedelic assisted therapy (PAT) is a safe and effective treatment for individuals with refractory depression, posttraumatic stress disorder, and substance use disorder.9 Plus, there have been ample studies providing support that PAT improves symptoms such as refractory anxiety/depression, demoralization, and existential distress in seriously ill patients, thus improving their quality of life and overall well-being.9
Nine U.S. cities and the State of Oregon have decriminalized or legalized the psychedelic psilocybin, based on the medical benefits patients have experienced evidenced from using it.10
In light of the increasing interest in PAT, Dr. Ira Byock provided the following points on what “all clinicians should know as they enter this uncharted territory”:
- Psychedelics have been around for a long time.
- Psychedelic-assisted therapies’ therapeutic effects are experiential.
- There are a variety of terms for specific categories of psychedelic compounds.
- Some palliative care teams are already caring for patients who undergo psychedelic experiences.
- Use of psychedelics should be well-observed by a skilled clinician with expertise.
I am hoping this provides a general refresher on palliative care and an overview of updates to alternative and complementary therapies for patients living with serious illness.9
Dr. Kang is a geriatrician and palliative care provider at the University of Washington, Seattle in the division of geriatrics and gerontology. She has no conflicts related to the content of this piece.
References
1. World Health Organization. Palliative care. 2020 Aug 5..
2. Weissman DE and Meier DE. Identifying patients in need of a palliative care assessment in the hospital setting a consensus report from the center to advance palliative care. J Palliat Med. 2011;14(1):17-23.
3. Sherry D et al. Is primary care physician involvement associated with earlier advance care planning? A study of patients in an academic primary care setting. J Palliat Med. 2022;25(1):75-80.
4. Quill TE and Abernethy AP. Generalist plus specialist palliative care-creating a more sustainable model. N Engl J Med. 2013;368:1173-75.
5. Ernecoff NC et al. Comparing specialty and primary palliative care interventions: Analysis of a systematic review. J Palliat Med. 2020;23(3):389-96.
6. Temmel JS et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2011;363:733-42.
7. Kogan M and Sexton M. Medical cannabis: A new old tool for palliative care. J Altern Complement Med . 2020 Sep;26(9):776-8.
8. Doppen M et al. Cannabis in palliative care: A systematic review of the current evidence. J Pain Symptom Manage. 2022 Jun 12;S0885-3924(22)00760-6.
9. Byock I. Psychedelics for serious illness: Five things clinicians need to know. The Center to Advance Palliative Care. Psychedelics for Serious Illness, Palliative in Practice, Center to Advance Palliative Care (capc.org). June 13, 2022.
10. Marks M. A strategy for rescheduling psilocybin. Scientific American. Oct. 11, 2021.
The World Health Organization defines palliative care as “an approach that improves the quality of life of patients (adults and children) and their families who are facing problems associated with life-threatening illness. It prevents and relieves suffering through the early identification, correct assessment, and treatment of pain and other problems, whether physical, psychosocial or spiritual.”1
The common misperception is that palliative care is only for those at end of life or only in the advanced stages of their illness. However, palliative care is ideally most helpful following individuals from diagnosis through their illness trajectory. Another misperception is that palliative care and hospice are the same thing. Though all hospice is palliative care, all palliative care is not hospice. Both palliative care and hospice provide care for individuals facing a serious illness and focus on the same philosophy of care, but palliative care can be initiated at any stage of illness, even if the goal is to pursue curative and life-prolonging therapies/interventions.
In contrast, hospice is considered for those who are at the end of life and are usually not pursuing life-prolonging therapies or interventions, instead focusing on comfort, symptom management, and optimization of quality of life.
Though there is a growing need for palliative care, there is a shortage of specialist palliative care providers. Much of the palliative care needs can be met by all providers who can offer basic symptom management, identification surrounding goals of care and discussions of advance care planning, and understanding of illness/prognosis and treatment options, which is called primary palliative care.2 In fact, two-thirds of patients with a serious illness other than cancer prefer discussion of end-of-life care or advance care planning with their primary care providers.3
Referral to specialty palliative care should be considered when there are more complexities to symptom/pain management and goals of care/end of life, transition to hospice, or complex communication dynamics.4
Though specialty palliative care was shown to be more comprehensive, both primary palliative care and specialty palliative care have led to improvements in the quality of life in individuals living with serious illness.5 Early integration of palliative care into routine care has been shown to improve symptom burden, mood, quality of life, survival, and health care costs.6
Updates in alternative and complementary therapies to palliative care
There are several alternative and complementary therapies to palliative care, including cannabis and psychedelics. These therapies are becoming or may become a familiar part of medical therapies that are listed in a patient’s history as part of their medical regimen, especially as more states continue to legalize and/or decriminalize the use of these alternative therapies for recreational or medicinal use.
Both cannabis and psychedelics have a longstanding history of therapeutic and holistic use. Cannabis has been used to manage symptoms such as pain since the 16th and 17th century.7 In palliative care, more patients may turn to various forms of cannabis as a source of relief from symptoms and suffering as their focus shifts more to quality of life.
Even with the increasing popularity of the use of cannabis among seriously ill patients, there is still a lack of evidence of the benefits of medical cannabis use in palliative care, and there is a lack of standardization of type of cannabis used and state regulations regarding their use.7
A recent systematic review found that despite the reported positive treatment effects of cannabis in palliative care, the results of the studies were conflicting. This highlights the need for further high-quality research to determine whether cannabis products are an effective treatment in palliative care patients.8
One limitation to note is that the majority of the included studies focused on cannabis use in patients with cancer for cancer-related symptoms. Few studies included patients with other serious conditions.
Psychedelics
There is evidence that psychedelic assisted therapy (PAT) is a safe and effective treatment for individuals with refractory depression, posttraumatic stress disorder, and substance use disorder.9 Plus, there have been ample studies providing support that PAT improves symptoms such as refractory anxiety/depression, demoralization, and existential distress in seriously ill patients, thus improving their quality of life and overall well-being.9
Nine U.S. cities and the State of Oregon have decriminalized or legalized the psychedelic psilocybin, based on the medical benefits patients have experienced evidenced from using it.10
In light of the increasing interest in PAT, Dr. Ira Byock provided the following points on what “all clinicians should know as they enter this uncharted territory”:
- Psychedelics have been around for a long time.
- Psychedelic-assisted therapies’ therapeutic effects are experiential.
- There are a variety of terms for specific categories of psychedelic compounds.
- Some palliative care teams are already caring for patients who undergo psychedelic experiences.
- Use of psychedelics should be well-observed by a skilled clinician with expertise.
I am hoping this provides a general refresher on palliative care and an overview of updates to alternative and complementary therapies for patients living with serious illness.9
Dr. Kang is a geriatrician and palliative care provider at the University of Washington, Seattle in the division of geriatrics and gerontology. She has no conflicts related to the content of this piece.
References
1. World Health Organization. Palliative care. 2020 Aug 5..
2. Weissman DE and Meier DE. Identifying patients in need of a palliative care assessment in the hospital setting a consensus report from the center to advance palliative care. J Palliat Med. 2011;14(1):17-23.
3. Sherry D et al. Is primary care physician involvement associated with earlier advance care planning? A study of patients in an academic primary care setting. J Palliat Med. 2022;25(1):75-80.
4. Quill TE and Abernethy AP. Generalist plus specialist palliative care-creating a more sustainable model. N Engl J Med. 2013;368:1173-75.
5. Ernecoff NC et al. Comparing specialty and primary palliative care interventions: Analysis of a systematic review. J Palliat Med. 2020;23(3):389-96.
6. Temmel JS et al. Early palliative care for patients with metastatic non–small-cell lung cancer. N Engl J Med. 2011;363:733-42.
7. Kogan M and Sexton M. Medical cannabis: A new old tool for palliative care. J Altern Complement Med . 2020 Sep;26(9):776-8.
8. Doppen M et al. Cannabis in palliative care: A systematic review of the current evidence. J Pain Symptom Manage. 2022 Jun 12;S0885-3924(22)00760-6.
9. Byock I. Psychedelics for serious illness: Five things clinicians need to know. The Center to Advance Palliative Care. Psychedelics for Serious Illness, Palliative in Practice, Center to Advance Palliative Care (capc.org). June 13, 2022.
10. Marks M. A strategy for rescheduling psilocybin. Scientific American. Oct. 11, 2021.
Can bone density scans help predict dementia risk?
, new research suggests.
In an analysis of more than 900 study participants, women in their 70s with more advanced abdominal aortic calcification (AAC) seen on lateral spine images during dual-energy x-ray absorptiometry (DXA) had a two- to fourfold higher risk for late-life dementia than those with low AAC.
This finding was independent of cardiovascular risk factors and apolipoprotein E (APOE ) genotype.
“While these results are exciting, we now need to undertake further large screening studies in older men and women using this approach to show that the findings are generalizable to older men and can identify people with greater cognitive decline,” coinvestigator Marc Sim, PhD, Edith Cowan University, Joondalup, Australia, said in an interview.
“This will hopefully open the door to studies of early disease-modifying interventions,” Sim said.
The findings were published online in The Lancet Regional Health – Western Pacific.
AAC and cognition
Late-life dementia occurring after age 80 is increasingly common because of both vascular and nonvascular risk factors.
Two recent studies in middle-aged and older men and women showed that AAC identified on bone densitometry was associated with poorer cognition, suggesting it may be related to cognitive decline and increased dementia risk.
This provided the rationale for the current study, Dr. Sim noted.
The researchers assessed AAC using DXA lateral spine images captured in the late 1990s in a prospective cohort of 958 older women who were participating in an osteoporosis study.
AAC was classified into established low, moderate, and extensive categories. At baseline, all women were aged 70 and older, and 45% had low AAC, 36% had moderate AAC, and 19% had extensive AAC.
Over 14.5 years, 150 women (15.7%) had a late-life hospitalization and/or died.
Improved risk prediction
Results showed that, compared with women who had low AAC, women with moderate and extensive AAC were more likely to experience late-life dementia hospitalization (9.3% low, 15.5% moderate, and 18.3% extensive) and death (2.8%, 8.3%, and 9.4%, respectively).
After multivariable adjustment, women with moderate AAC had a two- and threefold increased relative risk for late-life dementia hospitalization or death, compared with their peers who had low AAC.
Women with extensive AAC had a two- and fourfold increase in the adjusted relative risk for late-life dementia hospitalization or death.
“To our knowledge this is the first time it has been shown that AAC from these scans is related to late-life dementia,” Dr. Sim said.
“We demonstrated that AAC improved risk prediction in addition to cardiovascular risk factors and APOE genotype, a genetic risk factor for Alzheimer’s disease, the major form of dementia,” he added.
Dr. Sim noted “these additional lateral spine images” can be taken at the same time that hip and spine bone density tests are done.
“This provides an opportunity to identify AAC in large numbers of people,” he said.
He cautioned, however, that further studies with detailed dementia-related phenotypes, brain imaging, and measures of cognition are needed to confirm whether AAC will add value to dementia risk prediction.
‘Not surprising’
Commenting on the findings for this article, Claire Sexton, DPhil, senior director of scientific programs and outreach at the Alzheimer’s Association, Chicago, noted that AAC is a marker of atherosclerosis and is associated with vascular health outcomes.
Therefore, it is “not surprising it would be associated with dementia too. There’s been previous research linking atherosclerosis and Alzheimer’s disease,” Dr. Sexton said.
“What’s novel about this research is that it’s looking at AAC specifically, which can be identified through a relatively simple test that is already in widespread use,” she added.
Dr. Sexton noted that “much more research” is now needed in larger, more diverse populations in order to better understand the link between AAC and dementia – and whether bone density testing may be an appropriate dementia-screening tool.
“The good news is vascular conditions like atherosclerosis can be managed through lifestyle changes like eating a healthy diet and getting regular exercise. And research tells us what’s good for the heart is good for the brain,” Dr. Sexton said.
The study was funded by Kidney Health Australia, Healthway Health Promotion Foundation of Western Australia, Sir Charles Gairdner Hospital Research Advisory Committee Grant, and the National Health and Medical Research Council of Australia. Dr. Sim and Dr. Sexton have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
In an analysis of more than 900 study participants, women in their 70s with more advanced abdominal aortic calcification (AAC) seen on lateral spine images during dual-energy x-ray absorptiometry (DXA) had a two- to fourfold higher risk for late-life dementia than those with low AAC.
This finding was independent of cardiovascular risk factors and apolipoprotein E (APOE ) genotype.
“While these results are exciting, we now need to undertake further large screening studies in older men and women using this approach to show that the findings are generalizable to older men and can identify people with greater cognitive decline,” coinvestigator Marc Sim, PhD, Edith Cowan University, Joondalup, Australia, said in an interview.
“This will hopefully open the door to studies of early disease-modifying interventions,” Sim said.
The findings were published online in The Lancet Regional Health – Western Pacific.
AAC and cognition
Late-life dementia occurring after age 80 is increasingly common because of both vascular and nonvascular risk factors.
Two recent studies in middle-aged and older men and women showed that AAC identified on bone densitometry was associated with poorer cognition, suggesting it may be related to cognitive decline and increased dementia risk.
This provided the rationale for the current study, Dr. Sim noted.
The researchers assessed AAC using DXA lateral spine images captured in the late 1990s in a prospective cohort of 958 older women who were participating in an osteoporosis study.
AAC was classified into established low, moderate, and extensive categories. At baseline, all women were aged 70 and older, and 45% had low AAC, 36% had moderate AAC, and 19% had extensive AAC.
Over 14.5 years, 150 women (15.7%) had a late-life hospitalization and/or died.
Improved risk prediction
Results showed that, compared with women who had low AAC, women with moderate and extensive AAC were more likely to experience late-life dementia hospitalization (9.3% low, 15.5% moderate, and 18.3% extensive) and death (2.8%, 8.3%, and 9.4%, respectively).
After multivariable adjustment, women with moderate AAC had a two- and threefold increased relative risk for late-life dementia hospitalization or death, compared with their peers who had low AAC.
Women with extensive AAC had a two- and fourfold increase in the adjusted relative risk for late-life dementia hospitalization or death.
“To our knowledge this is the first time it has been shown that AAC from these scans is related to late-life dementia,” Dr. Sim said.
“We demonstrated that AAC improved risk prediction in addition to cardiovascular risk factors and APOE genotype, a genetic risk factor for Alzheimer’s disease, the major form of dementia,” he added.
Dr. Sim noted “these additional lateral spine images” can be taken at the same time that hip and spine bone density tests are done.
“This provides an opportunity to identify AAC in large numbers of people,” he said.
He cautioned, however, that further studies with detailed dementia-related phenotypes, brain imaging, and measures of cognition are needed to confirm whether AAC will add value to dementia risk prediction.
‘Not surprising’
Commenting on the findings for this article, Claire Sexton, DPhil, senior director of scientific programs and outreach at the Alzheimer’s Association, Chicago, noted that AAC is a marker of atherosclerosis and is associated with vascular health outcomes.
Therefore, it is “not surprising it would be associated with dementia too. There’s been previous research linking atherosclerosis and Alzheimer’s disease,” Dr. Sexton said.
“What’s novel about this research is that it’s looking at AAC specifically, which can be identified through a relatively simple test that is already in widespread use,” she added.
Dr. Sexton noted that “much more research” is now needed in larger, more diverse populations in order to better understand the link between AAC and dementia – and whether bone density testing may be an appropriate dementia-screening tool.
“The good news is vascular conditions like atherosclerosis can be managed through lifestyle changes like eating a healthy diet and getting regular exercise. And research tells us what’s good for the heart is good for the brain,” Dr. Sexton said.
The study was funded by Kidney Health Australia, Healthway Health Promotion Foundation of Western Australia, Sir Charles Gairdner Hospital Research Advisory Committee Grant, and the National Health and Medical Research Council of Australia. Dr. Sim and Dr. Sexton have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests.
In an analysis of more than 900 study participants, women in their 70s with more advanced abdominal aortic calcification (AAC) seen on lateral spine images during dual-energy x-ray absorptiometry (DXA) had a two- to fourfold higher risk for late-life dementia than those with low AAC.
This finding was independent of cardiovascular risk factors and apolipoprotein E (APOE ) genotype.
“While these results are exciting, we now need to undertake further large screening studies in older men and women using this approach to show that the findings are generalizable to older men and can identify people with greater cognitive decline,” coinvestigator Marc Sim, PhD, Edith Cowan University, Joondalup, Australia, said in an interview.
“This will hopefully open the door to studies of early disease-modifying interventions,” Sim said.
The findings were published online in The Lancet Regional Health – Western Pacific.
AAC and cognition
Late-life dementia occurring after age 80 is increasingly common because of both vascular and nonvascular risk factors.
Two recent studies in middle-aged and older men and women showed that AAC identified on bone densitometry was associated with poorer cognition, suggesting it may be related to cognitive decline and increased dementia risk.
This provided the rationale for the current study, Dr. Sim noted.
The researchers assessed AAC using DXA lateral spine images captured in the late 1990s in a prospective cohort of 958 older women who were participating in an osteoporosis study.
AAC was classified into established low, moderate, and extensive categories. At baseline, all women were aged 70 and older, and 45% had low AAC, 36% had moderate AAC, and 19% had extensive AAC.
Over 14.5 years, 150 women (15.7%) had a late-life hospitalization and/or died.
Improved risk prediction
Results showed that, compared with women who had low AAC, women with moderate and extensive AAC were more likely to experience late-life dementia hospitalization (9.3% low, 15.5% moderate, and 18.3% extensive) and death (2.8%, 8.3%, and 9.4%, respectively).
After multivariable adjustment, women with moderate AAC had a two- and threefold increased relative risk for late-life dementia hospitalization or death, compared with their peers who had low AAC.
Women with extensive AAC had a two- and fourfold increase in the adjusted relative risk for late-life dementia hospitalization or death.
“To our knowledge this is the first time it has been shown that AAC from these scans is related to late-life dementia,” Dr. Sim said.
“We demonstrated that AAC improved risk prediction in addition to cardiovascular risk factors and APOE genotype, a genetic risk factor for Alzheimer’s disease, the major form of dementia,” he added.
Dr. Sim noted “these additional lateral spine images” can be taken at the same time that hip and spine bone density tests are done.
“This provides an opportunity to identify AAC in large numbers of people,” he said.
He cautioned, however, that further studies with detailed dementia-related phenotypes, brain imaging, and measures of cognition are needed to confirm whether AAC will add value to dementia risk prediction.
‘Not surprising’
Commenting on the findings for this article, Claire Sexton, DPhil, senior director of scientific programs and outreach at the Alzheimer’s Association, Chicago, noted that AAC is a marker of atherosclerosis and is associated with vascular health outcomes.
Therefore, it is “not surprising it would be associated with dementia too. There’s been previous research linking atherosclerosis and Alzheimer’s disease,” Dr. Sexton said.
“What’s novel about this research is that it’s looking at AAC specifically, which can be identified through a relatively simple test that is already in widespread use,” she added.
Dr. Sexton noted that “much more research” is now needed in larger, more diverse populations in order to better understand the link between AAC and dementia – and whether bone density testing may be an appropriate dementia-screening tool.
“The good news is vascular conditions like atherosclerosis can be managed through lifestyle changes like eating a healthy diet and getting regular exercise. And research tells us what’s good for the heart is good for the brain,” Dr. Sexton said.
The study was funded by Kidney Health Australia, Healthway Health Promotion Foundation of Western Australia, Sir Charles Gairdner Hospital Research Advisory Committee Grant, and the National Health and Medical Research Council of Australia. Dr. Sim and Dr. Sexton have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE LANCET REGIONAL HEALTH – WESTERN PACIFIC
Pharmacist-Assisted Varenicline Tobacco Cessation Treatment for Veterans
Tobacco smoking remains the leading cause of preventable disease and death in the United States, accounting for more than 480,000 deaths annually.1 An estimated 50.6 million US adults (20.8%) identify as tobacco users, with even higher rates among veterans (29.2%).2,3 Tobacco use is estimated to cost the US more than $300 billion annually in direct and indirect medical costs.4 According to a 2015 report, more than two-thirds of adult smokers reported a desire to quit, while only 7.5% reported successfully quitting in the past year.5 According to that same report, only 57.2% of smokers who had seen a health professional in the past year reported receiving advice to quit.5 This statistic is unfortunate, as interventions that combine behavioral and pharmacologic support can drastically increase tobacco cessation rates compared with self-help materials or no treatment.6
Currently, 7 first-line medications (5 nicotine, 2 nonnicotine) have been shown to increase long-term smoking abstinence rates. Varenicline was approved by the US Food and Drug Administration (FDA) in 2006 for use in adults as an aid to smoking cessation treatment. As a partial agonist of the α4β2 nicotinic acetylcholine receptor, varenicline’s mechanism of action is believed to involve reduction of nicotine’s rewarding capacity.7 Varenicline not only aids in complete tobacco cessation but also has been found to be effective for reducing cigarette consumption among smokers not yet willing or able to make a quit attempt.8 Furthermore, varenicline has demonstrated efficacy among users of smokeless tobacco in achieving continuous abstinence.9
Widespread adoption of varenicline into clinical practice was perhaps slowed by early concerns of psychiatric complications, prompting the FDA to issue a boxed warning for risk of serious neuropsychiatric events. This boxed warning was removed in 2016 in response to publication of the Evaluating Adverse Events in a Global Smoking Cessation Study (EAGLES). In this randomized controlled trial of more than 8000 participants, among whom 50.5% had a psychiatric disorder determined to be stable, varenicline significantly increased rates of continuous tobacco cessation compared with bupropion or the nicotine patch without an increased risk of neuropsychiatric events.10 This study underscored not only the safety of varenicline, but also its superiority over other first-line cessation products. The most recently published clinical practice guidelines recommend varenicline as a first-line agent for helping patients achieve long-term smoking cessation.11,12
Pharmacists are uniquely positioned to provide tobacco cessation interventions given their medication expertise and accessibility to the public. Indeed, multiple studies have demonstrated the effectiveness of pharmacist-led interventions on tobacco cessation.13-15 As of 2019, only 12 states had statutes or regulations addressing pharmacist prescribing of tobacco cessation aids without a collaborative practice agreement or local standing order.16 Until recently, most of these states limited pharmacists’ prescriptive authority to
Within the US Department of Veterans Affairs (VA), the clinical pharmacy specialist (CPS) is credentialed as an advanced practitioner with authority to independently manage patient medication therapy for a variety of diseases specified under a scope of practice. Although CPSs have provided tobacco cessation services for years, expansion of their scope to include varenicline did not occur until June 26, 2019, at the Southern Arizona VA Health Care System (SAVAHCS). All VA prescribers must follow the same criteria for prescribing varenicline. Unless previously trialed on varenicline, patients must have failed an appropriate trial of first-line agents (NRT, bupropion, or combination therapy) or have a contraindication to use of these first-line therapies before varenicline can be considered. Exclusions to therapy would include history of serious hypersensitivity to varenicline; suicidal intent, plan, or attempt within the past 12 months; current substance use disorder other than nicotine (unless varenicline recommended or prescribed by mental health professional); or unstable mental health disorder.18
The purpose of this study was to evaluate the efficacy and safety of CPS management of varenicline compared with other clinicians. We hope that this study provides insight regarding how the expansion of CPS scope to include prescriptive authority for varenicline has affected patient outcomes.
Methods
This retrospective chart review was conducted using SAVAHCS electronic health records. This study was granted approval by the institutional review board and the research and development committee at SAVAHCS. Data were obtained through the Computerized Patient Record System from the information provided by the pharmacist informatics department and was recorded electronically on a secure Microsoft Excel spreadsheet.
To be eligible for this study, patients must have been aged ≥ 18 years with a varenicline prescription between July 1, 2019, and July 31, 2020. Patients were excluded if tobacco cessation was managed by community-based (non-VA) clincians or if there was a lack of documentation of tobacco use at baseline and after at least 12 weeks of varenicline therapy. Sample size was not designed to achieve statistical power. Potential patients were queried by a pharmacist specializing in clinical informatics. All patients meeting initial inclusion criteria were then screened individually to evaluate for exclusion criteria.
Data collected included baseline age, sex, race, type of tobacco use (cigarettes, smokeless, both), mean daily tobacco use, prespecified comorbidities (depression, anxiety, or other psychiatric condition), and previous cessation medications prescribed (NRT, bupropion, and previous trials of varenicline).
The primary outcomes were reduction in tobacco use calculated as change at 12 weeks from baseline (and 24 weeks if available), continuous abstinence at 12 weeks (and 24 weeks if available), adherence to varenicline therapy measured by proportion of days covered (days covered by refills during the measurement period divided by days between the first fill and the end of the measurement period), and time to first follow-up in days. For safety evaluation, charts were reviewed for documented adverse events (AEs) in the health record. These AEs were categorized as follows: gastrointestinal, mood disturbance, sleep disturbance, headache, seizures, allergy, or other.
Statistical analyses regarding veteran baseline characteristics were descriptive in nature. χ2 test was used to analyze differences in complete cessation rates and AEs, whereas a Student t test was used to compare reductions of tobacco use, proportion of days covered (ie, adherence), and time to first follow-up. An α of .05 was used to determine significance.
Results
From the initial search, 255 charts met general inclusion criteria. After chart review, only 50 patients from the CPS group and 93 patients from the other clinician group met criteria to be included (Figure 1). The CPS group included pharmacists specializing in ambulatory care and outpatient mental health. The other clinician group was composed primarily of primary care practitioners, psychiatrists, and pulmonologists.
Overall, baseline characteristics were similar between the groups (Table 1). In the overall study population, the mean age was 57.5 years, 90% of patients were male, and 99% of patients were cigarette smokers. Baseline mean (SD) tobacco use was similar between the groups: 14.5 (10.8) vs 14.8 (8.6) cigarettes daily for the CPS and other clinician group, respectively.
While there was a significant reduction in daily cigarette use for both groups at 12 and 24 weeks (Figure 2), there was no mean (SD) between-group difference found among those patients prescribed varenicline by a CPS compared with other clinicians: -7.9 (10.4) vs -5.4 (9.8) cigarettes daily, respectively (P = .15) (Table 2). Change in tobacco use at 24 weeks and rates of complete tobacco abstinence were also not statistically significant between prescriber groups. Adherence (as evidenced by refill data) was higher in the CPS group than in the other clinician group (42% vs 31%, respectively; P = .01). There was also a significant difference in time to first follow-up; patients whose varenicline therapy was managed by a CPS had a mean (SD) follow-up time of 52 (66) vs 163 (110) days when patients were managed by other clinicians (P < .001). AEs were documented in 42% of patients in the CPS group compared with 23% of patients in the other clinician group (Table 3). The most reported AEs were gastrointestinal, as well as mood and sleep disturbances.
Discussion
The results of this single center study suggest that management of varenicline by CPSs is associated with similar reductions in tobacco use and abstinence rates compared with management by other clinicians. These results provide evidence that CPS management of varenicline may be as safe and effective as management by other clinicians.
Adherence rates (reported as proportion of days covered when assessing varenicline refill data) were higher on average among patients managed by a CPS compared with patients managed by other clinicians. However, this outcome may not be as reflective of adherence as initially intended, given delays in follow-up (see limitations section). Time to first follow-up was drastically different between the groups, with much sooner follow-up by CPSs compared with other clinicians. Despite similar tobacco cessation rates between groups, more frequent follow-up by CPSs helps to assess patient barriers to cessation, adherence to therapy, and AEs with varenicline. A higher percentage of AEs were documented within the CPS group that could be attributed to disparities in documentation rather than true rates of AEs. While rates of AEs were initially intended to serve as the primary safety outcome, they may instead reflect pharmacists’ diligence in monitoring and documenting tolerability of medication therapy.
Limitations
Several limitations to this study should be noted. First, the data collected were only as detailed as the extent to which prescribers documented tobacco use, previous cessation trials, and AEs; thus, various data points are likely missing within this study that could impact the results presented. In line with lack of documentation, delays in follow-up (ie, annual primary care visits) sorely undermined proportion of days covered, making these data less indicative of true medication adherence. Furthermore, this study did not account for concurrent therapies, such as combination varenicline and nicotine gum/lozenges, or behavioral treatment strategies like cessation classes.
Another limitation was that some primary care practitioners prescribed varenicline but then referred these patients to a CPS for tobacco cessation follow-up. Per the study’s protocol, these patients were included within the other clinician group, which could have brought results closer to the null. Finally, the timing of this chart review (July 1, 2019, to July 31, 2020) intersects with the start of the COVID-19 pandemic, presenting a possible confounding factor if patients’ quit attempts were hindered by the stress and isolation of the pandemic.19 All pharmacist visits during the pandemic were conducted by telephone, which may have affected results.
Conclusions
In this study of veterans receiving varenicline, management by CPSs resulted in similar reductions of tobacco use and rates of complete abstinence compared with management by other clinicians. Pharmacist management was associated with greater adherence and shorter time to first follow-up compared with other clinicians. Additional research is needed to fully characterize the impact of pharmacist management of varenicline, justify expansion of clinical pharmacist scope of practice, and ultimately enhance patient outcomes regarding tobacco cessation.
It would be interesting to see more studies outside of the VA system to determine the impact of pharmacist management of varenicline for a more heterogenous patient population. At some point, a prospective controlled trial should be conducted to overcome the various confounding factors that limit the results of retrospective chart reviews
Acknowledgments
This article was prepared, and research was conducted with resources and the use of facilities at the Southern Arizona Veterans Affairs Health Care System in Tucson.
1. Centers for Disease Control and Prevention. Current cigarette smoking among adults in the United States. Updated March 17, 2022. Accessed May 31, 2022. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/adult_data/cig_smoking/index.htm 2. Cornelius ME, Wang TW, Jamal A, Loretan CG, Neff LJ. Tobacco product use among adults – United States, 2019. MMWR Morb Mortal Wkly Rep. 2020;69(46):1736-1742. doi:10.15585/mmwr.mm6946a4
3. Odani S, Agaku IT, Graffunder CM, Tynan MA, Armour BS. Tobacco product use among military veterans – United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2018;67(1):7-12. doi:10.15585/mmwr.mm6701a2
4. Hall W, Doran C. How much can the USA reduce health care costs by reducing smoking? PLoS Med. 2016;13(5):e1002021. doi:10.1371/journal.pmed.1002021.
5. Centers for Disease Control and Prevention. Smoking cessation: fast facts. Updated March 21, 2022. Accessed June 1, 2022. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/cessation/smoking-cessation-fast-facts/index.html
6. US Public Health Service Office of the Surgeon General; National Center for Chronic Disease Prevention and Health Promotion (US) Office on Smoking and Health. Chapter 6, Interventions for smoking cessation and treatments for nicotine dependence. In: Smoking Cessation: A Report of the Surgeon General [Internet]. Washington, DC: US Department of Health and Human Services; 2020. Accessed June 1, 2022. https://www.ncbi.nlm.nih.gov/books/NBK555596
7. Rollema H, Chambers LK, Coe JW, et al. Pharmacological profile of the α4β2 nicotinic acetylcholine receptor partial agonist varenicline, an effective smoking cessation aid. Neuropharmacology. 2007;52(3):985-994. doi:10.1016/j.neuropharm.2006.10.016
8. Ebbert JO, Hughes JR, West RJ, et al. Effect of varenicline on smoking cessation through smoking reduction: a randomized clinical trial. JAMA. 2015;313(7):687-694. doi:10.1001/jama.2015.280
9. Fagerström K, Gilljam H, Metcalfe M, Tonstad S, Messig M. Stopping smokeless tobacco with varenicline: randomised double blind placebo controlled trial. BMJ. 2010;341:c6549. doi:10.1136/bmj.c6549
10. Anthenelli RM, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): a double-blind, randomised, placebo-controlled clinical trial. Lancet. 2016;387(10037):2507-2520. doi:10.1016/S0140-6736(16)30272-0
11. Barua RS, Rigotti NA, Benowitz NL, et al. 2018 ACC expert consensus decision pathway on tobacco cessation treatment: a report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2018;72(25):3332-3365. doi:10.1016/j.jacc.2018.10.027
12. Leone FT, Zhang Y, Evers-Casey S, et al. Initiating pharmacologic treatment in tobacco-dependent adults. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;202(2):e5-e31. doi:10.1164/rccm.202005-1982ST
13. Saba M, Diep J, Saini B, Dhippayom T. Meta-analysis of the effectiveness of smoking cessation interventions in community pharmacy. J Clin Pharm Ther. 2014;39(3):240-247. doi:10.1111/jcpt.12131
14. Augustine JM, Taylor AM, Pelger M, Schiefer D, Warholak TL. Smoking quit rates among patients receiving pharmacist-provided pharmacotherapy and telephonic smoking cessation counseling. J Am Pharm Assoc. 2016;56(2):129-136. doi:10.1016/j.japh.2016.02.001
15. Dent LA, Harris KJ, Noonan CW. Tobacco interventions delivered by pharmacists: a summary and systematic review. Pharmacotherapy. 2007;27(7):1040-1051. doi:10.1592/phco.27.7.1040
16. National Alliance of State Pharmacy Associations. Pharmacist prescribing: tobacco cessation aids. February 10, 2021. Accessed June 1, 2022. https://naspa.us/resource/tobacco-cessation
17. Shen X, Bachyrycz A, Anderson JR, Tinker D, Raisch DW. Quitting patterns and predictors of success among participants in a tobacco cessation program provided by pharmacists in New Mexico. J Manag Care Spec Pharm. 2014;20(6):579-587. doi:10.18553/jmcp.2014.20.6.579
18. VA Center for Medication Safety, Tobacco Use Cessation Technical Advisory Group, Public Health Strategic Healthcare Group, VA Pharmacy Benefits Management Services, VISN Pharmacist Executives, and Medical Advisory Panel. Varenicline criteria for prescribing. 2008. Updated July 2011. Accessed June 9, 2022. https://www.healthquality.va.gov/tuc/VareniclineCriteriaforPrescribing.pdf
19. Jaklevic MC. COVID-19 and the “lost year” for smokers trying to quit. JAMA. 2021;325(19):1929-1930. doi:10.1001/jama.2021.5601
Tobacco smoking remains the leading cause of preventable disease and death in the United States, accounting for more than 480,000 deaths annually.1 An estimated 50.6 million US adults (20.8%) identify as tobacco users, with even higher rates among veterans (29.2%).2,3 Tobacco use is estimated to cost the US more than $300 billion annually in direct and indirect medical costs.4 According to a 2015 report, more than two-thirds of adult smokers reported a desire to quit, while only 7.5% reported successfully quitting in the past year.5 According to that same report, only 57.2% of smokers who had seen a health professional in the past year reported receiving advice to quit.5 This statistic is unfortunate, as interventions that combine behavioral and pharmacologic support can drastically increase tobacco cessation rates compared with self-help materials or no treatment.6
Currently, 7 first-line medications (5 nicotine, 2 nonnicotine) have been shown to increase long-term smoking abstinence rates. Varenicline was approved by the US Food and Drug Administration (FDA) in 2006 for use in adults as an aid to smoking cessation treatment. As a partial agonist of the α4β2 nicotinic acetylcholine receptor, varenicline’s mechanism of action is believed to involve reduction of nicotine’s rewarding capacity.7 Varenicline not only aids in complete tobacco cessation but also has been found to be effective for reducing cigarette consumption among smokers not yet willing or able to make a quit attempt.8 Furthermore, varenicline has demonstrated efficacy among users of smokeless tobacco in achieving continuous abstinence.9
Widespread adoption of varenicline into clinical practice was perhaps slowed by early concerns of psychiatric complications, prompting the FDA to issue a boxed warning for risk of serious neuropsychiatric events. This boxed warning was removed in 2016 in response to publication of the Evaluating Adverse Events in a Global Smoking Cessation Study (EAGLES). In this randomized controlled trial of more than 8000 participants, among whom 50.5% had a psychiatric disorder determined to be stable, varenicline significantly increased rates of continuous tobacco cessation compared with bupropion or the nicotine patch without an increased risk of neuropsychiatric events.10 This study underscored not only the safety of varenicline, but also its superiority over other first-line cessation products. The most recently published clinical practice guidelines recommend varenicline as a first-line agent for helping patients achieve long-term smoking cessation.11,12
Pharmacists are uniquely positioned to provide tobacco cessation interventions given their medication expertise and accessibility to the public. Indeed, multiple studies have demonstrated the effectiveness of pharmacist-led interventions on tobacco cessation.13-15 As of 2019, only 12 states had statutes or regulations addressing pharmacist prescribing of tobacco cessation aids without a collaborative practice agreement or local standing order.16 Until recently, most of these states limited pharmacists’ prescriptive authority to
Within the US Department of Veterans Affairs (VA), the clinical pharmacy specialist (CPS) is credentialed as an advanced practitioner with authority to independently manage patient medication therapy for a variety of diseases specified under a scope of practice. Although CPSs have provided tobacco cessation services for years, expansion of their scope to include varenicline did not occur until June 26, 2019, at the Southern Arizona VA Health Care System (SAVAHCS). All VA prescribers must follow the same criteria for prescribing varenicline. Unless previously trialed on varenicline, patients must have failed an appropriate trial of first-line agents (NRT, bupropion, or combination therapy) or have a contraindication to use of these first-line therapies before varenicline can be considered. Exclusions to therapy would include history of serious hypersensitivity to varenicline; suicidal intent, plan, or attempt within the past 12 months; current substance use disorder other than nicotine (unless varenicline recommended or prescribed by mental health professional); or unstable mental health disorder.18
The purpose of this study was to evaluate the efficacy and safety of CPS management of varenicline compared with other clinicians. We hope that this study provides insight regarding how the expansion of CPS scope to include prescriptive authority for varenicline has affected patient outcomes.
Methods
This retrospective chart review was conducted using SAVAHCS electronic health records. This study was granted approval by the institutional review board and the research and development committee at SAVAHCS. Data were obtained through the Computerized Patient Record System from the information provided by the pharmacist informatics department and was recorded electronically on a secure Microsoft Excel spreadsheet.
To be eligible for this study, patients must have been aged ≥ 18 years with a varenicline prescription between July 1, 2019, and July 31, 2020. Patients were excluded if tobacco cessation was managed by community-based (non-VA) clincians or if there was a lack of documentation of tobacco use at baseline and after at least 12 weeks of varenicline therapy. Sample size was not designed to achieve statistical power. Potential patients were queried by a pharmacist specializing in clinical informatics. All patients meeting initial inclusion criteria were then screened individually to evaluate for exclusion criteria.
Data collected included baseline age, sex, race, type of tobacco use (cigarettes, smokeless, both), mean daily tobacco use, prespecified comorbidities (depression, anxiety, or other psychiatric condition), and previous cessation medications prescribed (NRT, bupropion, and previous trials of varenicline).
The primary outcomes were reduction in tobacco use calculated as change at 12 weeks from baseline (and 24 weeks if available), continuous abstinence at 12 weeks (and 24 weeks if available), adherence to varenicline therapy measured by proportion of days covered (days covered by refills during the measurement period divided by days between the first fill and the end of the measurement period), and time to first follow-up in days. For safety evaluation, charts were reviewed for documented adverse events (AEs) in the health record. These AEs were categorized as follows: gastrointestinal, mood disturbance, sleep disturbance, headache, seizures, allergy, or other.
Statistical analyses regarding veteran baseline characteristics were descriptive in nature. χ2 test was used to analyze differences in complete cessation rates and AEs, whereas a Student t test was used to compare reductions of tobacco use, proportion of days covered (ie, adherence), and time to first follow-up. An α of .05 was used to determine significance.
Results
From the initial search, 255 charts met general inclusion criteria. After chart review, only 50 patients from the CPS group and 93 patients from the other clinician group met criteria to be included (Figure 1). The CPS group included pharmacists specializing in ambulatory care and outpatient mental health. The other clinician group was composed primarily of primary care practitioners, psychiatrists, and pulmonologists.
Overall, baseline characteristics were similar between the groups (Table 1). In the overall study population, the mean age was 57.5 years, 90% of patients were male, and 99% of patients were cigarette smokers. Baseline mean (SD) tobacco use was similar between the groups: 14.5 (10.8) vs 14.8 (8.6) cigarettes daily for the CPS and other clinician group, respectively.
While there was a significant reduction in daily cigarette use for both groups at 12 and 24 weeks (Figure 2), there was no mean (SD) between-group difference found among those patients prescribed varenicline by a CPS compared with other clinicians: -7.9 (10.4) vs -5.4 (9.8) cigarettes daily, respectively (P = .15) (Table 2). Change in tobacco use at 24 weeks and rates of complete tobacco abstinence were also not statistically significant between prescriber groups. Adherence (as evidenced by refill data) was higher in the CPS group than in the other clinician group (42% vs 31%, respectively; P = .01). There was also a significant difference in time to first follow-up; patients whose varenicline therapy was managed by a CPS had a mean (SD) follow-up time of 52 (66) vs 163 (110) days when patients were managed by other clinicians (P < .001). AEs were documented in 42% of patients in the CPS group compared with 23% of patients in the other clinician group (Table 3). The most reported AEs were gastrointestinal, as well as mood and sleep disturbances.
Discussion
The results of this single center study suggest that management of varenicline by CPSs is associated with similar reductions in tobacco use and abstinence rates compared with management by other clinicians. These results provide evidence that CPS management of varenicline may be as safe and effective as management by other clinicians.
Adherence rates (reported as proportion of days covered when assessing varenicline refill data) were higher on average among patients managed by a CPS compared with patients managed by other clinicians. However, this outcome may not be as reflective of adherence as initially intended, given delays in follow-up (see limitations section). Time to first follow-up was drastically different between the groups, with much sooner follow-up by CPSs compared with other clinicians. Despite similar tobacco cessation rates between groups, more frequent follow-up by CPSs helps to assess patient barriers to cessation, adherence to therapy, and AEs with varenicline. A higher percentage of AEs were documented within the CPS group that could be attributed to disparities in documentation rather than true rates of AEs. While rates of AEs were initially intended to serve as the primary safety outcome, they may instead reflect pharmacists’ diligence in monitoring and documenting tolerability of medication therapy.
Limitations
Several limitations to this study should be noted. First, the data collected were only as detailed as the extent to which prescribers documented tobacco use, previous cessation trials, and AEs; thus, various data points are likely missing within this study that could impact the results presented. In line with lack of documentation, delays in follow-up (ie, annual primary care visits) sorely undermined proportion of days covered, making these data less indicative of true medication adherence. Furthermore, this study did not account for concurrent therapies, such as combination varenicline and nicotine gum/lozenges, or behavioral treatment strategies like cessation classes.
Another limitation was that some primary care practitioners prescribed varenicline but then referred these patients to a CPS for tobacco cessation follow-up. Per the study’s protocol, these patients were included within the other clinician group, which could have brought results closer to the null. Finally, the timing of this chart review (July 1, 2019, to July 31, 2020) intersects with the start of the COVID-19 pandemic, presenting a possible confounding factor if patients’ quit attempts were hindered by the stress and isolation of the pandemic.19 All pharmacist visits during the pandemic were conducted by telephone, which may have affected results.
Conclusions
In this study of veterans receiving varenicline, management by CPSs resulted in similar reductions of tobacco use and rates of complete abstinence compared with management by other clinicians. Pharmacist management was associated with greater adherence and shorter time to first follow-up compared with other clinicians. Additional research is needed to fully characterize the impact of pharmacist management of varenicline, justify expansion of clinical pharmacist scope of practice, and ultimately enhance patient outcomes regarding tobacco cessation.
It would be interesting to see more studies outside of the VA system to determine the impact of pharmacist management of varenicline for a more heterogenous patient population. At some point, a prospective controlled trial should be conducted to overcome the various confounding factors that limit the results of retrospective chart reviews
Acknowledgments
This article was prepared, and research was conducted with resources and the use of facilities at the Southern Arizona Veterans Affairs Health Care System in Tucson.
Tobacco smoking remains the leading cause of preventable disease and death in the United States, accounting for more than 480,000 deaths annually.1 An estimated 50.6 million US adults (20.8%) identify as tobacco users, with even higher rates among veterans (29.2%).2,3 Tobacco use is estimated to cost the US more than $300 billion annually in direct and indirect medical costs.4 According to a 2015 report, more than two-thirds of adult smokers reported a desire to quit, while only 7.5% reported successfully quitting in the past year.5 According to that same report, only 57.2% of smokers who had seen a health professional in the past year reported receiving advice to quit.5 This statistic is unfortunate, as interventions that combine behavioral and pharmacologic support can drastically increase tobacco cessation rates compared with self-help materials or no treatment.6
Currently, 7 first-line medications (5 nicotine, 2 nonnicotine) have been shown to increase long-term smoking abstinence rates. Varenicline was approved by the US Food and Drug Administration (FDA) in 2006 for use in adults as an aid to smoking cessation treatment. As a partial agonist of the α4β2 nicotinic acetylcholine receptor, varenicline’s mechanism of action is believed to involve reduction of nicotine’s rewarding capacity.7 Varenicline not only aids in complete tobacco cessation but also has been found to be effective for reducing cigarette consumption among smokers not yet willing or able to make a quit attempt.8 Furthermore, varenicline has demonstrated efficacy among users of smokeless tobacco in achieving continuous abstinence.9
Widespread adoption of varenicline into clinical practice was perhaps slowed by early concerns of psychiatric complications, prompting the FDA to issue a boxed warning for risk of serious neuropsychiatric events. This boxed warning was removed in 2016 in response to publication of the Evaluating Adverse Events in a Global Smoking Cessation Study (EAGLES). In this randomized controlled trial of more than 8000 participants, among whom 50.5% had a psychiatric disorder determined to be stable, varenicline significantly increased rates of continuous tobacco cessation compared with bupropion or the nicotine patch without an increased risk of neuropsychiatric events.10 This study underscored not only the safety of varenicline, but also its superiority over other first-line cessation products. The most recently published clinical practice guidelines recommend varenicline as a first-line agent for helping patients achieve long-term smoking cessation.11,12
Pharmacists are uniquely positioned to provide tobacco cessation interventions given their medication expertise and accessibility to the public. Indeed, multiple studies have demonstrated the effectiveness of pharmacist-led interventions on tobacco cessation.13-15 As of 2019, only 12 states had statutes or regulations addressing pharmacist prescribing of tobacco cessation aids without a collaborative practice agreement or local standing order.16 Until recently, most of these states limited pharmacists’ prescriptive authority to
Within the US Department of Veterans Affairs (VA), the clinical pharmacy specialist (CPS) is credentialed as an advanced practitioner with authority to independently manage patient medication therapy for a variety of diseases specified under a scope of practice. Although CPSs have provided tobacco cessation services for years, expansion of their scope to include varenicline did not occur until June 26, 2019, at the Southern Arizona VA Health Care System (SAVAHCS). All VA prescribers must follow the same criteria for prescribing varenicline. Unless previously trialed on varenicline, patients must have failed an appropriate trial of first-line agents (NRT, bupropion, or combination therapy) or have a contraindication to use of these first-line therapies before varenicline can be considered. Exclusions to therapy would include history of serious hypersensitivity to varenicline; suicidal intent, plan, or attempt within the past 12 months; current substance use disorder other than nicotine (unless varenicline recommended or prescribed by mental health professional); or unstable mental health disorder.18
The purpose of this study was to evaluate the efficacy and safety of CPS management of varenicline compared with other clinicians. We hope that this study provides insight regarding how the expansion of CPS scope to include prescriptive authority for varenicline has affected patient outcomes.
Methods
This retrospective chart review was conducted using SAVAHCS electronic health records. This study was granted approval by the institutional review board and the research and development committee at SAVAHCS. Data were obtained through the Computerized Patient Record System from the information provided by the pharmacist informatics department and was recorded electronically on a secure Microsoft Excel spreadsheet.
To be eligible for this study, patients must have been aged ≥ 18 years with a varenicline prescription between July 1, 2019, and July 31, 2020. Patients were excluded if tobacco cessation was managed by community-based (non-VA) clincians or if there was a lack of documentation of tobacco use at baseline and after at least 12 weeks of varenicline therapy. Sample size was not designed to achieve statistical power. Potential patients were queried by a pharmacist specializing in clinical informatics. All patients meeting initial inclusion criteria were then screened individually to evaluate for exclusion criteria.
Data collected included baseline age, sex, race, type of tobacco use (cigarettes, smokeless, both), mean daily tobacco use, prespecified comorbidities (depression, anxiety, or other psychiatric condition), and previous cessation medications prescribed (NRT, bupropion, and previous trials of varenicline).
The primary outcomes were reduction in tobacco use calculated as change at 12 weeks from baseline (and 24 weeks if available), continuous abstinence at 12 weeks (and 24 weeks if available), adherence to varenicline therapy measured by proportion of days covered (days covered by refills during the measurement period divided by days between the first fill and the end of the measurement period), and time to first follow-up in days. For safety evaluation, charts were reviewed for documented adverse events (AEs) in the health record. These AEs were categorized as follows: gastrointestinal, mood disturbance, sleep disturbance, headache, seizures, allergy, or other.
Statistical analyses regarding veteran baseline characteristics were descriptive in nature. χ2 test was used to analyze differences in complete cessation rates and AEs, whereas a Student t test was used to compare reductions of tobacco use, proportion of days covered (ie, adherence), and time to first follow-up. An α of .05 was used to determine significance.
Results
From the initial search, 255 charts met general inclusion criteria. After chart review, only 50 patients from the CPS group and 93 patients from the other clinician group met criteria to be included (Figure 1). The CPS group included pharmacists specializing in ambulatory care and outpatient mental health. The other clinician group was composed primarily of primary care practitioners, psychiatrists, and pulmonologists.
Overall, baseline characteristics were similar between the groups (Table 1). In the overall study population, the mean age was 57.5 years, 90% of patients were male, and 99% of patients were cigarette smokers. Baseline mean (SD) tobacco use was similar between the groups: 14.5 (10.8) vs 14.8 (8.6) cigarettes daily for the CPS and other clinician group, respectively.
While there was a significant reduction in daily cigarette use for both groups at 12 and 24 weeks (Figure 2), there was no mean (SD) between-group difference found among those patients prescribed varenicline by a CPS compared with other clinicians: -7.9 (10.4) vs -5.4 (9.8) cigarettes daily, respectively (P = .15) (Table 2). Change in tobacco use at 24 weeks and rates of complete tobacco abstinence were also not statistically significant between prescriber groups. Adherence (as evidenced by refill data) was higher in the CPS group than in the other clinician group (42% vs 31%, respectively; P = .01). There was also a significant difference in time to first follow-up; patients whose varenicline therapy was managed by a CPS had a mean (SD) follow-up time of 52 (66) vs 163 (110) days when patients were managed by other clinicians (P < .001). AEs were documented in 42% of patients in the CPS group compared with 23% of patients in the other clinician group (Table 3). The most reported AEs were gastrointestinal, as well as mood and sleep disturbances.
Discussion
The results of this single center study suggest that management of varenicline by CPSs is associated with similar reductions in tobacco use and abstinence rates compared with management by other clinicians. These results provide evidence that CPS management of varenicline may be as safe and effective as management by other clinicians.
Adherence rates (reported as proportion of days covered when assessing varenicline refill data) were higher on average among patients managed by a CPS compared with patients managed by other clinicians. However, this outcome may not be as reflective of adherence as initially intended, given delays in follow-up (see limitations section). Time to first follow-up was drastically different between the groups, with much sooner follow-up by CPSs compared with other clinicians. Despite similar tobacco cessation rates between groups, more frequent follow-up by CPSs helps to assess patient barriers to cessation, adherence to therapy, and AEs with varenicline. A higher percentage of AEs were documented within the CPS group that could be attributed to disparities in documentation rather than true rates of AEs. While rates of AEs were initially intended to serve as the primary safety outcome, they may instead reflect pharmacists’ diligence in monitoring and documenting tolerability of medication therapy.
Limitations
Several limitations to this study should be noted. First, the data collected were only as detailed as the extent to which prescribers documented tobacco use, previous cessation trials, and AEs; thus, various data points are likely missing within this study that could impact the results presented. In line with lack of documentation, delays in follow-up (ie, annual primary care visits) sorely undermined proportion of days covered, making these data less indicative of true medication adherence. Furthermore, this study did not account for concurrent therapies, such as combination varenicline and nicotine gum/lozenges, or behavioral treatment strategies like cessation classes.
Another limitation was that some primary care practitioners prescribed varenicline but then referred these patients to a CPS for tobacco cessation follow-up. Per the study’s protocol, these patients were included within the other clinician group, which could have brought results closer to the null. Finally, the timing of this chart review (July 1, 2019, to July 31, 2020) intersects with the start of the COVID-19 pandemic, presenting a possible confounding factor if patients’ quit attempts were hindered by the stress and isolation of the pandemic.19 All pharmacist visits during the pandemic were conducted by telephone, which may have affected results.
Conclusions
In this study of veterans receiving varenicline, management by CPSs resulted in similar reductions of tobacco use and rates of complete abstinence compared with management by other clinicians. Pharmacist management was associated with greater adherence and shorter time to first follow-up compared with other clinicians. Additional research is needed to fully characterize the impact of pharmacist management of varenicline, justify expansion of clinical pharmacist scope of practice, and ultimately enhance patient outcomes regarding tobacco cessation.
It would be interesting to see more studies outside of the VA system to determine the impact of pharmacist management of varenicline for a more heterogenous patient population. At some point, a prospective controlled trial should be conducted to overcome the various confounding factors that limit the results of retrospective chart reviews
Acknowledgments
This article was prepared, and research was conducted with resources and the use of facilities at the Southern Arizona Veterans Affairs Health Care System in Tucson.
1. Centers for Disease Control and Prevention. Current cigarette smoking among adults in the United States. Updated March 17, 2022. Accessed May 31, 2022. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/adult_data/cig_smoking/index.htm 2. Cornelius ME, Wang TW, Jamal A, Loretan CG, Neff LJ. Tobacco product use among adults – United States, 2019. MMWR Morb Mortal Wkly Rep. 2020;69(46):1736-1742. doi:10.15585/mmwr.mm6946a4
3. Odani S, Agaku IT, Graffunder CM, Tynan MA, Armour BS. Tobacco product use among military veterans – United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2018;67(1):7-12. doi:10.15585/mmwr.mm6701a2
4. Hall W, Doran C. How much can the USA reduce health care costs by reducing smoking? PLoS Med. 2016;13(5):e1002021. doi:10.1371/journal.pmed.1002021.
5. Centers for Disease Control and Prevention. Smoking cessation: fast facts. Updated March 21, 2022. Accessed June 1, 2022. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/cessation/smoking-cessation-fast-facts/index.html
6. US Public Health Service Office of the Surgeon General; National Center for Chronic Disease Prevention and Health Promotion (US) Office on Smoking and Health. Chapter 6, Interventions for smoking cessation and treatments for nicotine dependence. In: Smoking Cessation: A Report of the Surgeon General [Internet]. Washington, DC: US Department of Health and Human Services; 2020. Accessed June 1, 2022. https://www.ncbi.nlm.nih.gov/books/NBK555596
7. Rollema H, Chambers LK, Coe JW, et al. Pharmacological profile of the α4β2 nicotinic acetylcholine receptor partial agonist varenicline, an effective smoking cessation aid. Neuropharmacology. 2007;52(3):985-994. doi:10.1016/j.neuropharm.2006.10.016
8. Ebbert JO, Hughes JR, West RJ, et al. Effect of varenicline on smoking cessation through smoking reduction: a randomized clinical trial. JAMA. 2015;313(7):687-694. doi:10.1001/jama.2015.280
9. Fagerström K, Gilljam H, Metcalfe M, Tonstad S, Messig M. Stopping smokeless tobacco with varenicline: randomised double blind placebo controlled trial. BMJ. 2010;341:c6549. doi:10.1136/bmj.c6549
10. Anthenelli RM, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): a double-blind, randomised, placebo-controlled clinical trial. Lancet. 2016;387(10037):2507-2520. doi:10.1016/S0140-6736(16)30272-0
11. Barua RS, Rigotti NA, Benowitz NL, et al. 2018 ACC expert consensus decision pathway on tobacco cessation treatment: a report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2018;72(25):3332-3365. doi:10.1016/j.jacc.2018.10.027
12. Leone FT, Zhang Y, Evers-Casey S, et al. Initiating pharmacologic treatment in tobacco-dependent adults. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;202(2):e5-e31. doi:10.1164/rccm.202005-1982ST
13. Saba M, Diep J, Saini B, Dhippayom T. Meta-analysis of the effectiveness of smoking cessation interventions in community pharmacy. J Clin Pharm Ther. 2014;39(3):240-247. doi:10.1111/jcpt.12131
14. Augustine JM, Taylor AM, Pelger M, Schiefer D, Warholak TL. Smoking quit rates among patients receiving pharmacist-provided pharmacotherapy and telephonic smoking cessation counseling. J Am Pharm Assoc. 2016;56(2):129-136. doi:10.1016/j.japh.2016.02.001
15. Dent LA, Harris KJ, Noonan CW. Tobacco interventions delivered by pharmacists: a summary and systematic review. Pharmacotherapy. 2007;27(7):1040-1051. doi:10.1592/phco.27.7.1040
16. National Alliance of State Pharmacy Associations. Pharmacist prescribing: tobacco cessation aids. February 10, 2021. Accessed June 1, 2022. https://naspa.us/resource/tobacco-cessation
17. Shen X, Bachyrycz A, Anderson JR, Tinker D, Raisch DW. Quitting patterns and predictors of success among participants in a tobacco cessation program provided by pharmacists in New Mexico. J Manag Care Spec Pharm. 2014;20(6):579-587. doi:10.18553/jmcp.2014.20.6.579
18. VA Center for Medication Safety, Tobacco Use Cessation Technical Advisory Group, Public Health Strategic Healthcare Group, VA Pharmacy Benefits Management Services, VISN Pharmacist Executives, and Medical Advisory Panel. Varenicline criteria for prescribing. 2008. Updated July 2011. Accessed June 9, 2022. https://www.healthquality.va.gov/tuc/VareniclineCriteriaforPrescribing.pdf
19. Jaklevic MC. COVID-19 and the “lost year” for smokers trying to quit. JAMA. 2021;325(19):1929-1930. doi:10.1001/jama.2021.5601
1. Centers for Disease Control and Prevention. Current cigarette smoking among adults in the United States. Updated March 17, 2022. Accessed May 31, 2022. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/adult_data/cig_smoking/index.htm 2. Cornelius ME, Wang TW, Jamal A, Loretan CG, Neff LJ. Tobacco product use among adults – United States, 2019. MMWR Morb Mortal Wkly Rep. 2020;69(46):1736-1742. doi:10.15585/mmwr.mm6946a4
3. Odani S, Agaku IT, Graffunder CM, Tynan MA, Armour BS. Tobacco product use among military veterans – United States, 2010-2015. MMWR Morb Mortal Wkly Rep. 2018;67(1):7-12. doi:10.15585/mmwr.mm6701a2
4. Hall W, Doran C. How much can the USA reduce health care costs by reducing smoking? PLoS Med. 2016;13(5):e1002021. doi:10.1371/journal.pmed.1002021.
5. Centers for Disease Control and Prevention. Smoking cessation: fast facts. Updated March 21, 2022. Accessed June 1, 2022. https://www.cdc.gov/tobacco/data_statistics/fact_sheets/cessation/smoking-cessation-fast-facts/index.html
6. US Public Health Service Office of the Surgeon General; National Center for Chronic Disease Prevention and Health Promotion (US) Office on Smoking and Health. Chapter 6, Interventions for smoking cessation and treatments for nicotine dependence. In: Smoking Cessation: A Report of the Surgeon General [Internet]. Washington, DC: US Department of Health and Human Services; 2020. Accessed June 1, 2022. https://www.ncbi.nlm.nih.gov/books/NBK555596
7. Rollema H, Chambers LK, Coe JW, et al. Pharmacological profile of the α4β2 nicotinic acetylcholine receptor partial agonist varenicline, an effective smoking cessation aid. Neuropharmacology. 2007;52(3):985-994. doi:10.1016/j.neuropharm.2006.10.016
8. Ebbert JO, Hughes JR, West RJ, et al. Effect of varenicline on smoking cessation through smoking reduction: a randomized clinical trial. JAMA. 2015;313(7):687-694. doi:10.1001/jama.2015.280
9. Fagerström K, Gilljam H, Metcalfe M, Tonstad S, Messig M. Stopping smokeless tobacco with varenicline: randomised double blind placebo controlled trial. BMJ. 2010;341:c6549. doi:10.1136/bmj.c6549
10. Anthenelli RM, Benowitz NL, West R, et al. Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): a double-blind, randomised, placebo-controlled clinical trial. Lancet. 2016;387(10037):2507-2520. doi:10.1016/S0140-6736(16)30272-0
11. Barua RS, Rigotti NA, Benowitz NL, et al. 2018 ACC expert consensus decision pathway on tobacco cessation treatment: a report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2018;72(25):3332-3365. doi:10.1016/j.jacc.2018.10.027
12. Leone FT, Zhang Y, Evers-Casey S, et al. Initiating pharmacologic treatment in tobacco-dependent adults. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. 2020;202(2):e5-e31. doi:10.1164/rccm.202005-1982ST
13. Saba M, Diep J, Saini B, Dhippayom T. Meta-analysis of the effectiveness of smoking cessation interventions in community pharmacy. J Clin Pharm Ther. 2014;39(3):240-247. doi:10.1111/jcpt.12131
14. Augustine JM, Taylor AM, Pelger M, Schiefer D, Warholak TL. Smoking quit rates among patients receiving pharmacist-provided pharmacotherapy and telephonic smoking cessation counseling. J Am Pharm Assoc. 2016;56(2):129-136. doi:10.1016/j.japh.2016.02.001
15. Dent LA, Harris KJ, Noonan CW. Tobacco interventions delivered by pharmacists: a summary and systematic review. Pharmacotherapy. 2007;27(7):1040-1051. doi:10.1592/phco.27.7.1040
16. National Alliance of State Pharmacy Associations. Pharmacist prescribing: tobacco cessation aids. February 10, 2021. Accessed June 1, 2022. https://naspa.us/resource/tobacco-cessation
17. Shen X, Bachyrycz A, Anderson JR, Tinker D, Raisch DW. Quitting patterns and predictors of success among participants in a tobacco cessation program provided by pharmacists in New Mexico. J Manag Care Spec Pharm. 2014;20(6):579-587. doi:10.18553/jmcp.2014.20.6.579
18. VA Center for Medication Safety, Tobacco Use Cessation Technical Advisory Group, Public Health Strategic Healthcare Group, VA Pharmacy Benefits Management Services, VISN Pharmacist Executives, and Medical Advisory Panel. Varenicline criteria for prescribing. 2008. Updated July 2011. Accessed June 9, 2022. https://www.healthquality.va.gov/tuc/VareniclineCriteriaforPrescribing.pdf
19. Jaklevic MC. COVID-19 and the “lost year” for smokers trying to quit. JAMA. 2021;325(19):1929-1930. doi:10.1001/jama.2021.5601
Appropriateness of Pharmacologic Thromboprophylaxis Prescribing Based on Padua Score Among Inpatient Veterans
Venous thromboembolism (VTE) presents as deep venous thromboembolism (DVT) or pulmonary embolism (PE). VTE is the third most common vascular disease and a leading cardiovascular complication.1,2 Hospitalized patients are at increased risk of developing VTE due to multiple factors such as inflammatory processes from acute illness, recent surgery or trauma leading to hypercoagulable states, and prolonged periods of immobilization.3 Additional risk factors for complications include presence of malignancy, obesity, and prior history of VTE. About half of VTE cases in the community setting occur as a result of a hospital admission for recent or ongoing acute illness or surgery.1 Hospitalized patients are often categorized as high risk for VTE, and this risk may persist postdischarge.4
The risk of hospital-associated VTE may be mitigated with either mechanical or pharmacologic thromboprophylaxis.5 Risk assessment models (RAMs), such as Padua Prediction Score (PPS) and IMPROVEDD, have been developed to assist in evaluating hospitalized patients’ risk of VTE and need for pharmacologic thromboprophylaxis (Table 1).1,5 The PPS is externally validated and can assist clinicians in VTE risk assessment when integrated into clinical decision making.6 Patients with a PPS ≥ 4 are deemed high risk for VTE, and pharmacologic thromboprophylaxis is indicated as long as the patient is not at high risk for bleeding. IMPROVEDD added D-dimer as an additional risk factor to IMPROVE and was validated in 2017 to help predict the risk of symptomatic VTE in acutely ill patients hospitalized for up to 77 days.7 IMPROVEDD scores ≥ 2 identify patients at high risk for symptomatic VTE through 77 days hospitalization, while scores ≥ 4 identify patients who may qualify for extended thromboprophylaxis.7 Despite their utility, RAMs may not be used appropriately within clinical practice, and whether patients should receive extended-duration thromboprophylaxis postdischarge and for how long is debatable.5
VTE events contribute to increased health care spending, morbidity, and mortality, thus it is imperative to evaluate current hospital practices with respect to appropriate prescribing of pharmacologic thromboprophylaxis.8 Appropriately identifying high-risk patients and prescribing pharmacologic thromboprophylaxis to limit preventable VTEs is essential. Conversely, it is important to withhold pharmacologic thromboprophylaxis from those deemed low risk to limit bleeding complications.9 Health care professionals must be good stewards of anticoagulant prescribing when implementing these tools along with clinical knowledge to weigh the risks vs benefits to promote medication safety and prevent further complications.10This quality improvement project aimed to evaluate if VTE thromboprophylaxis was appropriately given or withheld in hospitalized medical patients based on PPS calculated upon admission using a link to an online calculator embedded within an admission order set. Additionally, this study aimed to characterize patients readmitted for VTE within 45 days postdischarge to generate hypotheses for future stu
Methods
This was an observational, retrospective cohort study that took place at the US Department of Veterans Affairs (VA) Tennessee Valley Healthcare System (TVHS). TVHS is a multisite health care system with campuses in Nashville and Murfreesboro. Clinical pharmacists employed at the study site and the primary research investigators designed this study and oversaw its execution. The study was reviewed and deemed exempt as a quality improvement study by the TVHS Institutional Review Board.
This study included adult veterans aged ≥ 18 years admitted to a general medicine floor or the medical intensive care unit between June 1, 2017, and June 30, 2020. Patients were excluded if they were on chronic therapeutic anticoagulation prior to their index hospitalization, required therapeutic anticoagulation on admission for index hospitalization (ie, acute coronary syndrome requiring a heparin drip), or were bedded within the surgical intensive care unit. All patients admitted to the TVHS within the prespecified date range were extracted from the electronic health record. A second subset of patients meeting inclusion criteria and readmitted for VTE within 45 days of index hospitalization with International Classification of Diseases, Tenth Revision (ICD-10) descriptions including thrombosis or embolism were extracted for review of a secondary endpoint. Patients with preexisting clots, history of prior DVT or PE, or history of portal vein thrombosis were not reviewed.
The primary endpoint was the percentage of patients for whom pharmacologic thromboprophylaxis was appropriately initiated or withheld based on a PPS calculated upon admission (Table 2). PPS was chosen for review as it is the only RAM currently used at TVHS. Secondary endpoints were the percentage of patients with documented rationale for ordering thromboprophylaxis when not indicated, based on PPS, or withholding despite indication as well as the number of patients readmitted to TVHS for VTE within 45 days of discharge with IMPROVEDD scores ≥ 4 and < 4 (eAppendix available at doi:10.12788/fp.0291). The primary investigators performed a manual health record review of all patients meeting inclusion criteria. Descriptive statistics were used given this was a quality improvement study, therefore, sample size and power calculations were not necessary. Data were stored in Microsoft Excel spreadsheets that were encrypted and password protected. To maintain security of personal health information, all study files were kept on the TVHS internal network, and access was limited to the research investigators.
Results
Two hundred fifty patients meeting inclusion criteria were randomly selected for review for the primary endpoint. Of the patients reviewed for the primary endpoint, 118 had a PPS < 4 and 132 a PPS ≥ 4 (Figure). Pharmacologic thromboprophylaxis was inappropriately given or withheld based on their PPS for 91 (36.4%) patients. This included 58 (49.2%) patients in the low-risk group (PPS < 4) who had thromboprophylaxis inappropriately given and 33 (25.0%) patients in the high-risk group (PPS ≥ 4) who had thromboprophylaxis inappropriately withheld. Of the 58 patients with a PPS < 4 who were given prophylaxis, only 2 (3.4%) patients had documented rationale as to why anticoagulation was administered. Of the 132 patients with a PPS ≥ 4, 44 patients had thromboprophylaxis withheld. Eleven (8.3%) patients had thromboprophylaxis appropriately withheld due to presence or concern for bleeding. Commonly documented rationale for inappropriately withholding thromboprophylaxis when indicated included use of sequential compression devices (40.9%), pancytopenia (18.2%), dual antiplatelet therapy (9.1%), or patient was ambulatory (4.5%).
A secondary endpoint characterized patients at highest risk for developing a VTE after hospitalization for an acute illness. Seventy patients were readmitted within 45 days of discharge from the index hospitalization with ICD descriptions for embolism or thrombosis. Only 15 of those patients were readmitted with a newly diagnosed VTE not previously identified; 14 (93.3%) had a PPS ≥ 4 upon index admission and 10 (66.7%) appropriately received pharmacologic prophylaxis within 24 hours of admission. Of the 15 patients, 3 (20.0%) did not receive pharmacologic thromboprophylaxis within 24 hours of admission and 1 (6.7%) received thromboprophylaxis despite having a PPS < 4.
Looking at IMPROVEDD scores for the 15 patients at the index hospitalization discharge, 1 (6.7%) patient had an IMPROVEDD score < 2, 11 (73.3%) patients had IMPROVEDD scores ≥ 2, and 3 (20.0%) patients had IMPROVEDD scores ≥ 4. Two of the patients with IMPROVEDD scores ≥ 4 had a history of VTE and were aged > 60 years. Of the 15 patients reviewed, 7 had a diagnosis of cancer, and 3 were actively undergoing chemotherapy.
Discussion
PPS is the RAM embedded in our system’s order set, which identifies hospitalized medical patients at risk for VTE.6 In the original study that validated PPS, the results suggested that implementation of preventive measures during hospitalization in patients labeled as having high thrombotic risk confers longstanding protection against thromboembolic complications in comparison with untreated patients.6 However, PPS must be used consistently and appropriately to realize this benefit. Our results showed that pharmacologic thromboprophylaxis is frequently inappropriately given or withheld despite the incorporation of a RAM in an admission order set, suggesting there is a significant gap between written policy and actual practice. More than one-third of patients had thromboprophylaxis given or withheld inappropriately according to the PPS calculated manually on review. With this, there is concern for over- and underprescribing of thromboprophylaxis, which increases the risk of adverse events. Overprescribing can lead to unnecessary bleeding complications, whereas underprescribing can lead to preventable VTE.
One issue identified during this study was the need for a user-friendly interface. The PPS calculator currently embedded in our admission order set is a hyperlink to an online calculator. This is time consuming and cumbersome for clinicians tending to a high volume of patients, which may cause them to overlook the calculator and estimate risk based on clinician judgement. Noted areas for improvement regarding thromboprophylaxis during inpatient admissions include the failure to implement or adhere to risk stratification protocols, lack of appropriate assessment for thromboprophylaxis, and the overutilization of pharmacologic thromboprophylaxis in low-risk patients.11
Certain patients develop a VTE postdischarge despite efforts at prevention during their index hospitalization, which led us to explore our secondary endpoint looking at readmissions. Regarding thromboprophylaxis postdischarge, the duration of therapy is an area of current debate.5 Extended-duration thromboprophylaxis is defined as anticoagulation prescribed beyond hospitalization for up to 42 days total.1,12 To date, there have been 5 clinical trials to evaluate the utility of extended-duration thromboprophylaxis in hospitalized medically ill patients. While routine use is not recommended by the 2018 American Society of Hematology guidelines for management of VTE, more recent data suggest certain medically ill patients may derive benefit from extended-duration thromboprophylaxis.4 The IMPROVEDD score aimed to address this need, which is why it was calculated on index discharge for our patients readmitted within 45 days. Research is still needed to identify such patients and RAMs for capturing these subpopulations.1,11
Our secondary endpoint sought to characterize patients at highest risk for developing a VTE postdischarge. Of the 15 patients reviewed, 7 had a diagnosis of cancer and 3 were actively undergoing chemotherapy. With that, the Khorana Risk Score may have been a more appropriate RAM for some given the Khorana score is validated in ambulatory patients undergoing chemotherapy. D-dimer was only collected for 1 of the 15 patients, therefore, VTE risk could have been underestimated with the IMPROVEDD scores calculated. More than 75% of patients readmitted for VTE appropriately received thromboprophylaxis on index admission yet still went on to develop a VTE. It is essential to increase clinician awareness about hospital-acquired and postdischarge VTE. In line with guidance from the North American Thrombosis Forum, extended-duration thromboprophylaxis should be thoughtfully considered in high-risk patients.5 Pathways, including follow-up, are needed to implement postdischarge thromboprophylaxis when appropriate
Limitations
There were some inherent limitations to this study with its retrospective nature and small sample size. Data extraction was limited to health records within the VA, so there is a chance relevant history could be missed via incomplete documentation. Thus, our results could be an underestimation of postdischarge VTE prevalence if patients sought medical attention outside of the VA. Given this study was a retrospective chart review, data collection was limited to what was explicitly documented in the chart. Rationale for giving thromboprophylaxis when not indicated or holding when indicated may have been underestimated if clinicians did not document thoroughly in the electronic health record. Last, for the secondary endpoint reviewing the IMPROVEDD score, a D-dimer was not consistently obtained on admission, which could lead to underestimation of risk.
Conclusions
The results of this study showed that more than one-third of patients admitted to our facility within the prespecified timeframe had pharmacologic thromboprophylaxis inappropriately given or withheld according to a PPS manually calculated on admission. The PPS calculator currently embedded within our admission order set is not being utilized appropriately or consistently in clinical practice. Additionally, results from the secondary endpoint looking at IMPROVEDD scores highlight an unmet need for thromboprophylaxis at discharge. Pathways are needed to implement postdischarge thromboprophylaxis when appropriate for patients at highest thromboembolic risk.
1. Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2(22):3198-3225. doi:10.1182/bloodadvances.2018022954
2. Heit JA. Epidemiology of venous thromboembolism. Nat Rev Cardiol. 2015;12(8):464-474. doi:10.1038/nrcardio.2015.83
3. Turpie AG, Chin BS, Lip GY. Venous thromboembolism: pathophysiology, clinical features, and prevention. BMJ. 2002;325(7369):887-890. doi:10.1136/bmj.325.7369.887
4. Bajaj NS, Vaduganathan M, Qamar A, et al. Extended prophylaxis for venous thromboembolism after hospitalization for medical illness: A trial sequential and cumulative meta-analysis. Cannegieter SC, ed. PLoS Med. 2019;16(4):e1002797. doi:10.1371/journal.pmed.1002797
5. Barkoudah E, Piazza G, Hecht TEH, et al. Extended venous thromboembolism prophylaxis in medically ill patients: an NATF anticoagulation action initiative. Am J Med. 2020;133 (suppl 1):1-27. doi:10.1016/j.amjmed.2019.12.001
6. Barbar S, Noventa F, Rossetto V, et al. A risk assessment model for the identification of hospitalized medical patients at risk for venous thromboembolism: the Padua Prediction Score. J Thromb Haemost. 2010;8(11):2450-7. doi:10.1111/j.1538-7836.2010.04044.x
7. Gibson CM, Spyropoulos AC, Cohen AT, et al. The IMPROVEDD VTE risk score: incorporation of D-dimer into the IMPROVE score to improve venous thromboembolism risk stratification. TH Open. 2017;1(1):e56-e65. doi:10.1055/s-0037-1603929
8. ISTH Steering Committee for World Thrombosis Day. Thrombosis: a major contributor to global disease burden. Thromb Res. 2014;134(5):931-938. doi:10.1016/j.thromres.2014.08.014
9. Pavon JM, Sloane RJ, Pieper CF, et al. Poor adherence to risk stratification guidelines results in overuse of venous thromboembolism prophylaxis in hospitalized older adults. J Hosp Med. 2018;13(6):403-404. doi:10.12788/jhm.2916
10. Core elements of anticoagulation stewardship programs. Anticoagulation Forum. 2019. Accessed June 6, 2022. https://acforum-excellence.org/Resource-Center/resource_files/-2019-09-18-110254.pdf
11. Core elements of anticoagulation stewardship programs administrative oversight gap analysis: hospital and skilled nursing facilities. Anticoagulation Forum. 2019. Accessed June 6, 2022. https://acforum.org/web/downloads/ACF%20Gap%20Analysis%20Report.pdf
12. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e278S-e325S. doi:10.1378/chest.11-2404
Venous thromboembolism (VTE) presents as deep venous thromboembolism (DVT) or pulmonary embolism (PE). VTE is the third most common vascular disease and a leading cardiovascular complication.1,2 Hospitalized patients are at increased risk of developing VTE due to multiple factors such as inflammatory processes from acute illness, recent surgery or trauma leading to hypercoagulable states, and prolonged periods of immobilization.3 Additional risk factors for complications include presence of malignancy, obesity, and prior history of VTE. About half of VTE cases in the community setting occur as a result of a hospital admission for recent or ongoing acute illness or surgery.1 Hospitalized patients are often categorized as high risk for VTE, and this risk may persist postdischarge.4
The risk of hospital-associated VTE may be mitigated with either mechanical or pharmacologic thromboprophylaxis.5 Risk assessment models (RAMs), such as Padua Prediction Score (PPS) and IMPROVEDD, have been developed to assist in evaluating hospitalized patients’ risk of VTE and need for pharmacologic thromboprophylaxis (Table 1).1,5 The PPS is externally validated and can assist clinicians in VTE risk assessment when integrated into clinical decision making.6 Patients with a PPS ≥ 4 are deemed high risk for VTE, and pharmacologic thromboprophylaxis is indicated as long as the patient is not at high risk for bleeding. IMPROVEDD added D-dimer as an additional risk factor to IMPROVE and was validated in 2017 to help predict the risk of symptomatic VTE in acutely ill patients hospitalized for up to 77 days.7 IMPROVEDD scores ≥ 2 identify patients at high risk for symptomatic VTE through 77 days hospitalization, while scores ≥ 4 identify patients who may qualify for extended thromboprophylaxis.7 Despite their utility, RAMs may not be used appropriately within clinical practice, and whether patients should receive extended-duration thromboprophylaxis postdischarge and for how long is debatable.5
VTE events contribute to increased health care spending, morbidity, and mortality, thus it is imperative to evaluate current hospital practices with respect to appropriate prescribing of pharmacologic thromboprophylaxis.8 Appropriately identifying high-risk patients and prescribing pharmacologic thromboprophylaxis to limit preventable VTEs is essential. Conversely, it is important to withhold pharmacologic thromboprophylaxis from those deemed low risk to limit bleeding complications.9 Health care professionals must be good stewards of anticoagulant prescribing when implementing these tools along with clinical knowledge to weigh the risks vs benefits to promote medication safety and prevent further complications.10This quality improvement project aimed to evaluate if VTE thromboprophylaxis was appropriately given or withheld in hospitalized medical patients based on PPS calculated upon admission using a link to an online calculator embedded within an admission order set. Additionally, this study aimed to characterize patients readmitted for VTE within 45 days postdischarge to generate hypotheses for future stu
Methods
This was an observational, retrospective cohort study that took place at the US Department of Veterans Affairs (VA) Tennessee Valley Healthcare System (TVHS). TVHS is a multisite health care system with campuses in Nashville and Murfreesboro. Clinical pharmacists employed at the study site and the primary research investigators designed this study and oversaw its execution. The study was reviewed and deemed exempt as a quality improvement study by the TVHS Institutional Review Board.
This study included adult veterans aged ≥ 18 years admitted to a general medicine floor or the medical intensive care unit between June 1, 2017, and June 30, 2020. Patients were excluded if they were on chronic therapeutic anticoagulation prior to their index hospitalization, required therapeutic anticoagulation on admission for index hospitalization (ie, acute coronary syndrome requiring a heparin drip), or were bedded within the surgical intensive care unit. All patients admitted to the TVHS within the prespecified date range were extracted from the electronic health record. A second subset of patients meeting inclusion criteria and readmitted for VTE within 45 days of index hospitalization with International Classification of Diseases, Tenth Revision (ICD-10) descriptions including thrombosis or embolism were extracted for review of a secondary endpoint. Patients with preexisting clots, history of prior DVT or PE, or history of portal vein thrombosis were not reviewed.
The primary endpoint was the percentage of patients for whom pharmacologic thromboprophylaxis was appropriately initiated or withheld based on a PPS calculated upon admission (Table 2). PPS was chosen for review as it is the only RAM currently used at TVHS. Secondary endpoints were the percentage of patients with documented rationale for ordering thromboprophylaxis when not indicated, based on PPS, or withholding despite indication as well as the number of patients readmitted to TVHS for VTE within 45 days of discharge with IMPROVEDD scores ≥ 4 and < 4 (eAppendix available at doi:10.12788/fp.0291). The primary investigators performed a manual health record review of all patients meeting inclusion criteria. Descriptive statistics were used given this was a quality improvement study, therefore, sample size and power calculations were not necessary. Data were stored in Microsoft Excel spreadsheets that were encrypted and password protected. To maintain security of personal health information, all study files were kept on the TVHS internal network, and access was limited to the research investigators.
Results
Two hundred fifty patients meeting inclusion criteria were randomly selected for review for the primary endpoint. Of the patients reviewed for the primary endpoint, 118 had a PPS < 4 and 132 a PPS ≥ 4 (Figure). Pharmacologic thromboprophylaxis was inappropriately given or withheld based on their PPS for 91 (36.4%) patients. This included 58 (49.2%) patients in the low-risk group (PPS < 4) who had thromboprophylaxis inappropriately given and 33 (25.0%) patients in the high-risk group (PPS ≥ 4) who had thromboprophylaxis inappropriately withheld. Of the 58 patients with a PPS < 4 who were given prophylaxis, only 2 (3.4%) patients had documented rationale as to why anticoagulation was administered. Of the 132 patients with a PPS ≥ 4, 44 patients had thromboprophylaxis withheld. Eleven (8.3%) patients had thromboprophylaxis appropriately withheld due to presence or concern for bleeding. Commonly documented rationale for inappropriately withholding thromboprophylaxis when indicated included use of sequential compression devices (40.9%), pancytopenia (18.2%), dual antiplatelet therapy (9.1%), or patient was ambulatory (4.5%).
A secondary endpoint characterized patients at highest risk for developing a VTE after hospitalization for an acute illness. Seventy patients were readmitted within 45 days of discharge from the index hospitalization with ICD descriptions for embolism or thrombosis. Only 15 of those patients were readmitted with a newly diagnosed VTE not previously identified; 14 (93.3%) had a PPS ≥ 4 upon index admission and 10 (66.7%) appropriately received pharmacologic prophylaxis within 24 hours of admission. Of the 15 patients, 3 (20.0%) did not receive pharmacologic thromboprophylaxis within 24 hours of admission and 1 (6.7%) received thromboprophylaxis despite having a PPS < 4.
Looking at IMPROVEDD scores for the 15 patients at the index hospitalization discharge, 1 (6.7%) patient had an IMPROVEDD score < 2, 11 (73.3%) patients had IMPROVEDD scores ≥ 2, and 3 (20.0%) patients had IMPROVEDD scores ≥ 4. Two of the patients with IMPROVEDD scores ≥ 4 had a history of VTE and were aged > 60 years. Of the 15 patients reviewed, 7 had a diagnosis of cancer, and 3 were actively undergoing chemotherapy.
Discussion
PPS is the RAM embedded in our system’s order set, which identifies hospitalized medical patients at risk for VTE.6 In the original study that validated PPS, the results suggested that implementation of preventive measures during hospitalization in patients labeled as having high thrombotic risk confers longstanding protection against thromboembolic complications in comparison with untreated patients.6 However, PPS must be used consistently and appropriately to realize this benefit. Our results showed that pharmacologic thromboprophylaxis is frequently inappropriately given or withheld despite the incorporation of a RAM in an admission order set, suggesting there is a significant gap between written policy and actual practice. More than one-third of patients had thromboprophylaxis given or withheld inappropriately according to the PPS calculated manually on review. With this, there is concern for over- and underprescribing of thromboprophylaxis, which increases the risk of adverse events. Overprescribing can lead to unnecessary bleeding complications, whereas underprescribing can lead to preventable VTE.
One issue identified during this study was the need for a user-friendly interface. The PPS calculator currently embedded in our admission order set is a hyperlink to an online calculator. This is time consuming and cumbersome for clinicians tending to a high volume of patients, which may cause them to overlook the calculator and estimate risk based on clinician judgement. Noted areas for improvement regarding thromboprophylaxis during inpatient admissions include the failure to implement or adhere to risk stratification protocols, lack of appropriate assessment for thromboprophylaxis, and the overutilization of pharmacologic thromboprophylaxis in low-risk patients.11
Certain patients develop a VTE postdischarge despite efforts at prevention during their index hospitalization, which led us to explore our secondary endpoint looking at readmissions. Regarding thromboprophylaxis postdischarge, the duration of therapy is an area of current debate.5 Extended-duration thromboprophylaxis is defined as anticoagulation prescribed beyond hospitalization for up to 42 days total.1,12 To date, there have been 5 clinical trials to evaluate the utility of extended-duration thromboprophylaxis in hospitalized medically ill patients. While routine use is not recommended by the 2018 American Society of Hematology guidelines for management of VTE, more recent data suggest certain medically ill patients may derive benefit from extended-duration thromboprophylaxis.4 The IMPROVEDD score aimed to address this need, which is why it was calculated on index discharge for our patients readmitted within 45 days. Research is still needed to identify such patients and RAMs for capturing these subpopulations.1,11
Our secondary endpoint sought to characterize patients at highest risk for developing a VTE postdischarge. Of the 15 patients reviewed, 7 had a diagnosis of cancer and 3 were actively undergoing chemotherapy. With that, the Khorana Risk Score may have been a more appropriate RAM for some given the Khorana score is validated in ambulatory patients undergoing chemotherapy. D-dimer was only collected for 1 of the 15 patients, therefore, VTE risk could have been underestimated with the IMPROVEDD scores calculated. More than 75% of patients readmitted for VTE appropriately received thromboprophylaxis on index admission yet still went on to develop a VTE. It is essential to increase clinician awareness about hospital-acquired and postdischarge VTE. In line with guidance from the North American Thrombosis Forum, extended-duration thromboprophylaxis should be thoughtfully considered in high-risk patients.5 Pathways, including follow-up, are needed to implement postdischarge thromboprophylaxis when appropriate
Limitations
There were some inherent limitations to this study with its retrospective nature and small sample size. Data extraction was limited to health records within the VA, so there is a chance relevant history could be missed via incomplete documentation. Thus, our results could be an underestimation of postdischarge VTE prevalence if patients sought medical attention outside of the VA. Given this study was a retrospective chart review, data collection was limited to what was explicitly documented in the chart. Rationale for giving thromboprophylaxis when not indicated or holding when indicated may have been underestimated if clinicians did not document thoroughly in the electronic health record. Last, for the secondary endpoint reviewing the IMPROVEDD score, a D-dimer was not consistently obtained on admission, which could lead to underestimation of risk.
Conclusions
The results of this study showed that more than one-third of patients admitted to our facility within the prespecified timeframe had pharmacologic thromboprophylaxis inappropriately given or withheld according to a PPS manually calculated on admission. The PPS calculator currently embedded within our admission order set is not being utilized appropriately or consistently in clinical practice. Additionally, results from the secondary endpoint looking at IMPROVEDD scores highlight an unmet need for thromboprophylaxis at discharge. Pathways are needed to implement postdischarge thromboprophylaxis when appropriate for patients at highest thromboembolic risk.
Venous thromboembolism (VTE) presents as deep venous thromboembolism (DVT) or pulmonary embolism (PE). VTE is the third most common vascular disease and a leading cardiovascular complication.1,2 Hospitalized patients are at increased risk of developing VTE due to multiple factors such as inflammatory processes from acute illness, recent surgery or trauma leading to hypercoagulable states, and prolonged periods of immobilization.3 Additional risk factors for complications include presence of malignancy, obesity, and prior history of VTE. About half of VTE cases in the community setting occur as a result of a hospital admission for recent or ongoing acute illness or surgery.1 Hospitalized patients are often categorized as high risk for VTE, and this risk may persist postdischarge.4
The risk of hospital-associated VTE may be mitigated with either mechanical or pharmacologic thromboprophylaxis.5 Risk assessment models (RAMs), such as Padua Prediction Score (PPS) and IMPROVEDD, have been developed to assist in evaluating hospitalized patients’ risk of VTE and need for pharmacologic thromboprophylaxis (Table 1).1,5 The PPS is externally validated and can assist clinicians in VTE risk assessment when integrated into clinical decision making.6 Patients with a PPS ≥ 4 are deemed high risk for VTE, and pharmacologic thromboprophylaxis is indicated as long as the patient is not at high risk for bleeding. IMPROVEDD added D-dimer as an additional risk factor to IMPROVE and was validated in 2017 to help predict the risk of symptomatic VTE in acutely ill patients hospitalized for up to 77 days.7 IMPROVEDD scores ≥ 2 identify patients at high risk for symptomatic VTE through 77 days hospitalization, while scores ≥ 4 identify patients who may qualify for extended thromboprophylaxis.7 Despite their utility, RAMs may not be used appropriately within clinical practice, and whether patients should receive extended-duration thromboprophylaxis postdischarge and for how long is debatable.5
VTE events contribute to increased health care spending, morbidity, and mortality, thus it is imperative to evaluate current hospital practices with respect to appropriate prescribing of pharmacologic thromboprophylaxis.8 Appropriately identifying high-risk patients and prescribing pharmacologic thromboprophylaxis to limit preventable VTEs is essential. Conversely, it is important to withhold pharmacologic thromboprophylaxis from those deemed low risk to limit bleeding complications.9 Health care professionals must be good stewards of anticoagulant prescribing when implementing these tools along with clinical knowledge to weigh the risks vs benefits to promote medication safety and prevent further complications.10This quality improvement project aimed to evaluate if VTE thromboprophylaxis was appropriately given or withheld in hospitalized medical patients based on PPS calculated upon admission using a link to an online calculator embedded within an admission order set. Additionally, this study aimed to characterize patients readmitted for VTE within 45 days postdischarge to generate hypotheses for future stu
Methods
This was an observational, retrospective cohort study that took place at the US Department of Veterans Affairs (VA) Tennessee Valley Healthcare System (TVHS). TVHS is a multisite health care system with campuses in Nashville and Murfreesboro. Clinical pharmacists employed at the study site and the primary research investigators designed this study and oversaw its execution. The study was reviewed and deemed exempt as a quality improvement study by the TVHS Institutional Review Board.
This study included adult veterans aged ≥ 18 years admitted to a general medicine floor or the medical intensive care unit between June 1, 2017, and June 30, 2020. Patients were excluded if they were on chronic therapeutic anticoagulation prior to their index hospitalization, required therapeutic anticoagulation on admission for index hospitalization (ie, acute coronary syndrome requiring a heparin drip), or were bedded within the surgical intensive care unit. All patients admitted to the TVHS within the prespecified date range were extracted from the electronic health record. A second subset of patients meeting inclusion criteria and readmitted for VTE within 45 days of index hospitalization with International Classification of Diseases, Tenth Revision (ICD-10) descriptions including thrombosis or embolism were extracted for review of a secondary endpoint. Patients with preexisting clots, history of prior DVT or PE, or history of portal vein thrombosis were not reviewed.
The primary endpoint was the percentage of patients for whom pharmacologic thromboprophylaxis was appropriately initiated or withheld based on a PPS calculated upon admission (Table 2). PPS was chosen for review as it is the only RAM currently used at TVHS. Secondary endpoints were the percentage of patients with documented rationale for ordering thromboprophylaxis when not indicated, based on PPS, or withholding despite indication as well as the number of patients readmitted to TVHS for VTE within 45 days of discharge with IMPROVEDD scores ≥ 4 and < 4 (eAppendix available at doi:10.12788/fp.0291). The primary investigators performed a manual health record review of all patients meeting inclusion criteria. Descriptive statistics were used given this was a quality improvement study, therefore, sample size and power calculations were not necessary. Data were stored in Microsoft Excel spreadsheets that were encrypted and password protected. To maintain security of personal health information, all study files were kept on the TVHS internal network, and access was limited to the research investigators.
Results
Two hundred fifty patients meeting inclusion criteria were randomly selected for review for the primary endpoint. Of the patients reviewed for the primary endpoint, 118 had a PPS < 4 and 132 a PPS ≥ 4 (Figure). Pharmacologic thromboprophylaxis was inappropriately given or withheld based on their PPS for 91 (36.4%) patients. This included 58 (49.2%) patients in the low-risk group (PPS < 4) who had thromboprophylaxis inappropriately given and 33 (25.0%) patients in the high-risk group (PPS ≥ 4) who had thromboprophylaxis inappropriately withheld. Of the 58 patients with a PPS < 4 who were given prophylaxis, only 2 (3.4%) patients had documented rationale as to why anticoagulation was administered. Of the 132 patients with a PPS ≥ 4, 44 patients had thromboprophylaxis withheld. Eleven (8.3%) patients had thromboprophylaxis appropriately withheld due to presence or concern for bleeding. Commonly documented rationale for inappropriately withholding thromboprophylaxis when indicated included use of sequential compression devices (40.9%), pancytopenia (18.2%), dual antiplatelet therapy (9.1%), or patient was ambulatory (4.5%).
A secondary endpoint characterized patients at highest risk for developing a VTE after hospitalization for an acute illness. Seventy patients were readmitted within 45 days of discharge from the index hospitalization with ICD descriptions for embolism or thrombosis. Only 15 of those patients were readmitted with a newly diagnosed VTE not previously identified; 14 (93.3%) had a PPS ≥ 4 upon index admission and 10 (66.7%) appropriately received pharmacologic prophylaxis within 24 hours of admission. Of the 15 patients, 3 (20.0%) did not receive pharmacologic thromboprophylaxis within 24 hours of admission and 1 (6.7%) received thromboprophylaxis despite having a PPS < 4.
Looking at IMPROVEDD scores for the 15 patients at the index hospitalization discharge, 1 (6.7%) patient had an IMPROVEDD score < 2, 11 (73.3%) patients had IMPROVEDD scores ≥ 2, and 3 (20.0%) patients had IMPROVEDD scores ≥ 4. Two of the patients with IMPROVEDD scores ≥ 4 had a history of VTE and were aged > 60 years. Of the 15 patients reviewed, 7 had a diagnosis of cancer, and 3 were actively undergoing chemotherapy.
Discussion
PPS is the RAM embedded in our system’s order set, which identifies hospitalized medical patients at risk for VTE.6 In the original study that validated PPS, the results suggested that implementation of preventive measures during hospitalization in patients labeled as having high thrombotic risk confers longstanding protection against thromboembolic complications in comparison with untreated patients.6 However, PPS must be used consistently and appropriately to realize this benefit. Our results showed that pharmacologic thromboprophylaxis is frequently inappropriately given or withheld despite the incorporation of a RAM in an admission order set, suggesting there is a significant gap between written policy and actual practice. More than one-third of patients had thromboprophylaxis given or withheld inappropriately according to the PPS calculated manually on review. With this, there is concern for over- and underprescribing of thromboprophylaxis, which increases the risk of adverse events. Overprescribing can lead to unnecessary bleeding complications, whereas underprescribing can lead to preventable VTE.
One issue identified during this study was the need for a user-friendly interface. The PPS calculator currently embedded in our admission order set is a hyperlink to an online calculator. This is time consuming and cumbersome for clinicians tending to a high volume of patients, which may cause them to overlook the calculator and estimate risk based on clinician judgement. Noted areas for improvement regarding thromboprophylaxis during inpatient admissions include the failure to implement or adhere to risk stratification protocols, lack of appropriate assessment for thromboprophylaxis, and the overutilization of pharmacologic thromboprophylaxis in low-risk patients.11
Certain patients develop a VTE postdischarge despite efforts at prevention during their index hospitalization, which led us to explore our secondary endpoint looking at readmissions. Regarding thromboprophylaxis postdischarge, the duration of therapy is an area of current debate.5 Extended-duration thromboprophylaxis is defined as anticoagulation prescribed beyond hospitalization for up to 42 days total.1,12 To date, there have been 5 clinical trials to evaluate the utility of extended-duration thromboprophylaxis in hospitalized medically ill patients. While routine use is not recommended by the 2018 American Society of Hematology guidelines for management of VTE, more recent data suggest certain medically ill patients may derive benefit from extended-duration thromboprophylaxis.4 The IMPROVEDD score aimed to address this need, which is why it was calculated on index discharge for our patients readmitted within 45 days. Research is still needed to identify such patients and RAMs for capturing these subpopulations.1,11
Our secondary endpoint sought to characterize patients at highest risk for developing a VTE postdischarge. Of the 15 patients reviewed, 7 had a diagnosis of cancer and 3 were actively undergoing chemotherapy. With that, the Khorana Risk Score may have been a more appropriate RAM for some given the Khorana score is validated in ambulatory patients undergoing chemotherapy. D-dimer was only collected for 1 of the 15 patients, therefore, VTE risk could have been underestimated with the IMPROVEDD scores calculated. More than 75% of patients readmitted for VTE appropriately received thromboprophylaxis on index admission yet still went on to develop a VTE. It is essential to increase clinician awareness about hospital-acquired and postdischarge VTE. In line with guidance from the North American Thrombosis Forum, extended-duration thromboprophylaxis should be thoughtfully considered in high-risk patients.5 Pathways, including follow-up, are needed to implement postdischarge thromboprophylaxis when appropriate
Limitations
There were some inherent limitations to this study with its retrospective nature and small sample size. Data extraction was limited to health records within the VA, so there is a chance relevant history could be missed via incomplete documentation. Thus, our results could be an underestimation of postdischarge VTE prevalence if patients sought medical attention outside of the VA. Given this study was a retrospective chart review, data collection was limited to what was explicitly documented in the chart. Rationale for giving thromboprophylaxis when not indicated or holding when indicated may have been underestimated if clinicians did not document thoroughly in the electronic health record. Last, for the secondary endpoint reviewing the IMPROVEDD score, a D-dimer was not consistently obtained on admission, which could lead to underestimation of risk.
Conclusions
The results of this study showed that more than one-third of patients admitted to our facility within the prespecified timeframe had pharmacologic thromboprophylaxis inappropriately given or withheld according to a PPS manually calculated on admission. The PPS calculator currently embedded within our admission order set is not being utilized appropriately or consistently in clinical practice. Additionally, results from the secondary endpoint looking at IMPROVEDD scores highlight an unmet need for thromboprophylaxis at discharge. Pathways are needed to implement postdischarge thromboprophylaxis when appropriate for patients at highest thromboembolic risk.
1. Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2(22):3198-3225. doi:10.1182/bloodadvances.2018022954
2. Heit JA. Epidemiology of venous thromboembolism. Nat Rev Cardiol. 2015;12(8):464-474. doi:10.1038/nrcardio.2015.83
3. Turpie AG, Chin BS, Lip GY. Venous thromboembolism: pathophysiology, clinical features, and prevention. BMJ. 2002;325(7369):887-890. doi:10.1136/bmj.325.7369.887
4. Bajaj NS, Vaduganathan M, Qamar A, et al. Extended prophylaxis for venous thromboembolism after hospitalization for medical illness: A trial sequential and cumulative meta-analysis. Cannegieter SC, ed. PLoS Med. 2019;16(4):e1002797. doi:10.1371/journal.pmed.1002797
5. Barkoudah E, Piazza G, Hecht TEH, et al. Extended venous thromboembolism prophylaxis in medically ill patients: an NATF anticoagulation action initiative. Am J Med. 2020;133 (suppl 1):1-27. doi:10.1016/j.amjmed.2019.12.001
6. Barbar S, Noventa F, Rossetto V, et al. A risk assessment model for the identification of hospitalized medical patients at risk for venous thromboembolism: the Padua Prediction Score. J Thromb Haemost. 2010;8(11):2450-7. doi:10.1111/j.1538-7836.2010.04044.x
7. Gibson CM, Spyropoulos AC, Cohen AT, et al. The IMPROVEDD VTE risk score: incorporation of D-dimer into the IMPROVE score to improve venous thromboembolism risk stratification. TH Open. 2017;1(1):e56-e65. doi:10.1055/s-0037-1603929
8. ISTH Steering Committee for World Thrombosis Day. Thrombosis: a major contributor to global disease burden. Thromb Res. 2014;134(5):931-938. doi:10.1016/j.thromres.2014.08.014
9. Pavon JM, Sloane RJ, Pieper CF, et al. Poor adherence to risk stratification guidelines results in overuse of venous thromboembolism prophylaxis in hospitalized older adults. J Hosp Med. 2018;13(6):403-404. doi:10.12788/jhm.2916
10. Core elements of anticoagulation stewardship programs. Anticoagulation Forum. 2019. Accessed June 6, 2022. https://acforum-excellence.org/Resource-Center/resource_files/-2019-09-18-110254.pdf
11. Core elements of anticoagulation stewardship programs administrative oversight gap analysis: hospital and skilled nursing facilities. Anticoagulation Forum. 2019. Accessed June 6, 2022. https://acforum.org/web/downloads/ACF%20Gap%20Analysis%20Report.pdf
12. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e278S-e325S. doi:10.1378/chest.11-2404
1. Schünemann HJ, Cushman M, Burnett AE, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv. 2018;2(22):3198-3225. doi:10.1182/bloodadvances.2018022954
2. Heit JA. Epidemiology of venous thromboembolism. Nat Rev Cardiol. 2015;12(8):464-474. doi:10.1038/nrcardio.2015.83
3. Turpie AG, Chin BS, Lip GY. Venous thromboembolism: pathophysiology, clinical features, and prevention. BMJ. 2002;325(7369):887-890. doi:10.1136/bmj.325.7369.887
4. Bajaj NS, Vaduganathan M, Qamar A, et al. Extended prophylaxis for venous thromboembolism after hospitalization for medical illness: A trial sequential and cumulative meta-analysis. Cannegieter SC, ed. PLoS Med. 2019;16(4):e1002797. doi:10.1371/journal.pmed.1002797
5. Barkoudah E, Piazza G, Hecht TEH, et al. Extended venous thromboembolism prophylaxis in medically ill patients: an NATF anticoagulation action initiative. Am J Med. 2020;133 (suppl 1):1-27. doi:10.1016/j.amjmed.2019.12.001
6. Barbar S, Noventa F, Rossetto V, et al. A risk assessment model for the identification of hospitalized medical patients at risk for venous thromboembolism: the Padua Prediction Score. J Thromb Haemost. 2010;8(11):2450-7. doi:10.1111/j.1538-7836.2010.04044.x
7. Gibson CM, Spyropoulos AC, Cohen AT, et al. The IMPROVEDD VTE risk score: incorporation of D-dimer into the IMPROVE score to improve venous thromboembolism risk stratification. TH Open. 2017;1(1):e56-e65. doi:10.1055/s-0037-1603929
8. ISTH Steering Committee for World Thrombosis Day. Thrombosis: a major contributor to global disease burden. Thromb Res. 2014;134(5):931-938. doi:10.1016/j.thromres.2014.08.014
9. Pavon JM, Sloane RJ, Pieper CF, et al. Poor adherence to risk stratification guidelines results in overuse of venous thromboembolism prophylaxis in hospitalized older adults. J Hosp Med. 2018;13(6):403-404. doi:10.12788/jhm.2916
10. Core elements of anticoagulation stewardship programs. Anticoagulation Forum. 2019. Accessed June 6, 2022. https://acforum-excellence.org/Resource-Center/resource_files/-2019-09-18-110254.pdf
11. Core elements of anticoagulation stewardship programs administrative oversight gap analysis: hospital and skilled nursing facilities. Anticoagulation Forum. 2019. Accessed June 6, 2022. https://acforum.org/web/downloads/ACF%20Gap%20Analysis%20Report.pdf
12. Falck-Ytter Y, Francis CW, Johanson NA, et al. Prevention of VTE in orthopedic surgery patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl 2):e278S-e325S. doi:10.1378/chest.11-2404
Cannabis use causes spike in ED visits
Cannabis users had a 22% increased risk of an emergency department (ED) visit or hospitalization compared to nonusers, as determined from data from more than 30,000 individuals.
Although cannabis contains compounds similar to tobacco, “data published on the association between cannabis smoking and airways health have been contradictory,” and whether smoking cannabis increases a user’s risk of developing acute respiratory illness remains unclear, wrote Nicholas T. Vozoris, MD, of the University of Toronto, and colleagues.
In a study published in BMJ Open Respiratory Research, the investigators reviewed national health records data from 35,114 individuals aged 12-65 years for the period January 2009 to December 2015. Of these persons, 4,807 of the 6,425 who reported cannabis use in the past year were matched with 10,395 never-users who served as controls. The mean age of the study population at the index date was 35 years, and 42% were women; demographics were similar between users and control persons.
Overall, the odds of respiratory-related emergency department visits or hospitalizations were not significantly different between the cannabis users and the control persons (3.6% vs. 3.9%; odds ratio, 0.91). However, cannabis users had significantly greater odds of all-cause ED visits or hospitalizations (30.0% vs. 26.0%; OR, 1.22). All-cause mortality was 0.2% for both groups.
Respiratory problems were the second-highest reason for all-cause visits, the researchers noted. The lack of a difference in respiratory-related visits between cannabis users and nonusers conflicts somewhat with previous studies on this topic, which were limited, the researchers noted in their discussion.
The negative results also might stem from factors for which the researchers could not adjust, including insufficient cannabis smoke exposure among users in the study population, noninhalational cannabis use, which is less likely to have a respiratory effect, and possible secondhand exposure among control persons.
“It is also possible that our analysis might have been insufficiently powered to detect a significant signal with respect to the primary outcome,” they noted.
However, after the researchers controlled for multiple variables, the risk of an equally important morbidity outcome, all-cause ED visits or hospitalizations, was significantly greater among cannabis users than among control individuals, and respiratory reasons were the second most common cause for ED visits and hospitalizations in the all-cause outcome, they emphasized.
The study findings were limited by several factors, including the retrospective and observational design and the inability to control for all confounding variables, the researchers noted. Other limitations include the use of self-reports and potential for bias, the inability to perform dose-response analysis, and the high number of infrequent cannabis users in the study population.
However, the results suggest that cannabis use is associated with an increased risk of serious health events and should be discouraged, although more research is needed to confirm the current study findings, they concluded.
Consider range of causes for cannabis emergency visits
“With growing numbers of states legalizing recreational use of cannabis, it’s important to understand whether cannabis use is associated with increased emergency department visits,” Robert D. Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, told this news organization.
Previous studies have shown an association between increased ED visits and cannabis use in states, especially with edibles, where cannabis is legal, and “the current study reinforces the elevated risk of ED visits along with hospitalizations,” he said.
“While the researchers found no increased risk of respiratory-related complaints among users compared to the general population, there was an associated increase in ED visits and hospitalizations, which is important to understand,” said Dr. Glatter, who was not involved in the study.
“While this observational study found that the incidence of respiratory complaints was not significantly different among frequent users of cannabis, the increased odds that cannabis users would require evaluation in the emergency room or even hospitalization was still apparent even after the investigators controlled for such factors as use of alcohol, tobacco, illicit drug use, or other mental health–related disorders,” Dr. Glatter noted.
“That said, it’s a bit surprising that with the continued popularity of vaping, especially among teens, there was still not any appreciable or significant increase in respiratory complaints observed. Beyond this finding, I was not surprised by the overall conclusions of the current study, as we continue to see an elevated number of patients presenting to the ED with adverse events related to cannabis use.”
Dr. Glatter noted that “the majority of patients we see in the ED are associated with use of edibles, since it takes longer for the person to feel the effects, leading the user to consume more of the product up front, with delayed effects lasting up to 12 hours. This is what gets people into trouble and leads to toxicity of cannabis, or ‘overdoses,’ “ he explained.
When consuming edible cannabis products, “[p]eople need to begin at low dosages and not take additional gummies up front, since it can take up to 2 or even 3 hours in some cases to feel the initial effects. With the drug’s effects lasting up to 12 hours, it’s especially important to avoid operating any motor vehicles, bicycles, or scooters, since reaction time is impaired, as well as overall judgment, balance, and fine motor skills,” Dr. Glatter said.
Cannabis can land users in the ED for a range of reasons, said Dr. Glatter. “According to the study, 15% of the emergency room visits and hospitalizations were due to acute trauma, 14% due to respiratory issues, and 13% to gastrointestinal illnesses. These effects were seen in first-time users but not those with chronic use, according to the study inclusion criteria.”
Cannabis use could result in physical injuries through “impaired judgment, coordination, combined with an altered state of consciousness or generalized drowsiness, that could contribute to an increase in motor vehicle collisions, along with an increased risk for falls leading to lacerations, fractures, contusions, or bruising,” said Dr. Glatter. “Cannabis may also lead to an altered sense of perception related to interactions with others, resulting in feelings of anxiety or restlessness culminating in physical altercations and other injuries.”
The current study indicates the need for understanding the potential physical and psychological effects of cannabis use, he said.
“Additional research is needed to better understand the relative percentage cases related to edibles vs. inhalation presenting to the ED,” he noted. “There is no question that edibles continue to present significant dangers for those who don’t read labels or remain poorly informed regarding their dosing as a result of delayed onset and longer duration,” he said. To help reduce risk of toxicity, the concept of a “high lasting 12-15 hours, as with edibles, as opposed to 3-4 hours from inhalation must be clearly stated on packaging and better communicated with users, as the toxicity with edibles is more often from lack of prior knowledge about onset of effects related to dosing.”
In addition, the “potential for psychosis to develop with more chronic cannabis use, along with cannabinoid hyperemesis syndrome should be on every clinician’s radar,” Dr. Glatter emphasized.
“The bottom line is that as more states legalize the use of cannabis, it’s vital to also implement comprehensive public education efforts to provide users with the reported risks associated with not only inhalation (vaping or flower) but also edibles, which account for an increasingly greater percentage of ED visits and associated adverse effects,” he said.
The study was supported by the Lung Association–Ontario, as well as by grants from the Ontario Ministry of Health and the Ministry of Long-Term Care. The researchers and Dr. Glatter have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cannabis users had a 22% increased risk of an emergency department (ED) visit or hospitalization compared to nonusers, as determined from data from more than 30,000 individuals.
Although cannabis contains compounds similar to tobacco, “data published on the association between cannabis smoking and airways health have been contradictory,” and whether smoking cannabis increases a user’s risk of developing acute respiratory illness remains unclear, wrote Nicholas T. Vozoris, MD, of the University of Toronto, and colleagues.
In a study published in BMJ Open Respiratory Research, the investigators reviewed national health records data from 35,114 individuals aged 12-65 years for the period January 2009 to December 2015. Of these persons, 4,807 of the 6,425 who reported cannabis use in the past year were matched with 10,395 never-users who served as controls. The mean age of the study population at the index date was 35 years, and 42% were women; demographics were similar between users and control persons.
Overall, the odds of respiratory-related emergency department visits or hospitalizations were not significantly different between the cannabis users and the control persons (3.6% vs. 3.9%; odds ratio, 0.91). However, cannabis users had significantly greater odds of all-cause ED visits or hospitalizations (30.0% vs. 26.0%; OR, 1.22). All-cause mortality was 0.2% for both groups.
Respiratory problems were the second-highest reason for all-cause visits, the researchers noted. The lack of a difference in respiratory-related visits between cannabis users and nonusers conflicts somewhat with previous studies on this topic, which were limited, the researchers noted in their discussion.
The negative results also might stem from factors for which the researchers could not adjust, including insufficient cannabis smoke exposure among users in the study population, noninhalational cannabis use, which is less likely to have a respiratory effect, and possible secondhand exposure among control persons.
“It is also possible that our analysis might have been insufficiently powered to detect a significant signal with respect to the primary outcome,” they noted.
However, after the researchers controlled for multiple variables, the risk of an equally important morbidity outcome, all-cause ED visits or hospitalizations, was significantly greater among cannabis users than among control individuals, and respiratory reasons were the second most common cause for ED visits and hospitalizations in the all-cause outcome, they emphasized.
The study findings were limited by several factors, including the retrospective and observational design and the inability to control for all confounding variables, the researchers noted. Other limitations include the use of self-reports and potential for bias, the inability to perform dose-response analysis, and the high number of infrequent cannabis users in the study population.
However, the results suggest that cannabis use is associated with an increased risk of serious health events and should be discouraged, although more research is needed to confirm the current study findings, they concluded.
Consider range of causes for cannabis emergency visits
“With growing numbers of states legalizing recreational use of cannabis, it’s important to understand whether cannabis use is associated with increased emergency department visits,” Robert D. Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, told this news organization.
Previous studies have shown an association between increased ED visits and cannabis use in states, especially with edibles, where cannabis is legal, and “the current study reinforces the elevated risk of ED visits along with hospitalizations,” he said.
“While the researchers found no increased risk of respiratory-related complaints among users compared to the general population, there was an associated increase in ED visits and hospitalizations, which is important to understand,” said Dr. Glatter, who was not involved in the study.
“While this observational study found that the incidence of respiratory complaints was not significantly different among frequent users of cannabis, the increased odds that cannabis users would require evaluation in the emergency room or even hospitalization was still apparent even after the investigators controlled for such factors as use of alcohol, tobacco, illicit drug use, or other mental health–related disorders,” Dr. Glatter noted.
“That said, it’s a bit surprising that with the continued popularity of vaping, especially among teens, there was still not any appreciable or significant increase in respiratory complaints observed. Beyond this finding, I was not surprised by the overall conclusions of the current study, as we continue to see an elevated number of patients presenting to the ED with adverse events related to cannabis use.”
Dr. Glatter noted that “the majority of patients we see in the ED are associated with use of edibles, since it takes longer for the person to feel the effects, leading the user to consume more of the product up front, with delayed effects lasting up to 12 hours. This is what gets people into trouble and leads to toxicity of cannabis, or ‘overdoses,’ “ he explained.
When consuming edible cannabis products, “[p]eople need to begin at low dosages and not take additional gummies up front, since it can take up to 2 or even 3 hours in some cases to feel the initial effects. With the drug’s effects lasting up to 12 hours, it’s especially important to avoid operating any motor vehicles, bicycles, or scooters, since reaction time is impaired, as well as overall judgment, balance, and fine motor skills,” Dr. Glatter said.
Cannabis can land users in the ED for a range of reasons, said Dr. Glatter. “According to the study, 15% of the emergency room visits and hospitalizations were due to acute trauma, 14% due to respiratory issues, and 13% to gastrointestinal illnesses. These effects were seen in first-time users but not those with chronic use, according to the study inclusion criteria.”
Cannabis use could result in physical injuries through “impaired judgment, coordination, combined with an altered state of consciousness or generalized drowsiness, that could contribute to an increase in motor vehicle collisions, along with an increased risk for falls leading to lacerations, fractures, contusions, or bruising,” said Dr. Glatter. “Cannabis may also lead to an altered sense of perception related to interactions with others, resulting in feelings of anxiety or restlessness culminating in physical altercations and other injuries.”
The current study indicates the need for understanding the potential physical and psychological effects of cannabis use, he said.
“Additional research is needed to better understand the relative percentage cases related to edibles vs. inhalation presenting to the ED,” he noted. “There is no question that edibles continue to present significant dangers for those who don’t read labels or remain poorly informed regarding their dosing as a result of delayed onset and longer duration,” he said. To help reduce risk of toxicity, the concept of a “high lasting 12-15 hours, as with edibles, as opposed to 3-4 hours from inhalation must be clearly stated on packaging and better communicated with users, as the toxicity with edibles is more often from lack of prior knowledge about onset of effects related to dosing.”
In addition, the “potential for psychosis to develop with more chronic cannabis use, along with cannabinoid hyperemesis syndrome should be on every clinician’s radar,” Dr. Glatter emphasized.
“The bottom line is that as more states legalize the use of cannabis, it’s vital to also implement comprehensive public education efforts to provide users with the reported risks associated with not only inhalation (vaping or flower) but also edibles, which account for an increasingly greater percentage of ED visits and associated adverse effects,” he said.
The study was supported by the Lung Association–Ontario, as well as by grants from the Ontario Ministry of Health and the Ministry of Long-Term Care. The researchers and Dr. Glatter have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cannabis users had a 22% increased risk of an emergency department (ED) visit or hospitalization compared to nonusers, as determined from data from more than 30,000 individuals.
Although cannabis contains compounds similar to tobacco, “data published on the association between cannabis smoking and airways health have been contradictory,” and whether smoking cannabis increases a user’s risk of developing acute respiratory illness remains unclear, wrote Nicholas T. Vozoris, MD, of the University of Toronto, and colleagues.
In a study published in BMJ Open Respiratory Research, the investigators reviewed national health records data from 35,114 individuals aged 12-65 years for the period January 2009 to December 2015. Of these persons, 4,807 of the 6,425 who reported cannabis use in the past year were matched with 10,395 never-users who served as controls. The mean age of the study population at the index date was 35 years, and 42% were women; demographics were similar between users and control persons.
Overall, the odds of respiratory-related emergency department visits or hospitalizations were not significantly different between the cannabis users and the control persons (3.6% vs. 3.9%; odds ratio, 0.91). However, cannabis users had significantly greater odds of all-cause ED visits or hospitalizations (30.0% vs. 26.0%; OR, 1.22). All-cause mortality was 0.2% for both groups.
Respiratory problems were the second-highest reason for all-cause visits, the researchers noted. The lack of a difference in respiratory-related visits between cannabis users and nonusers conflicts somewhat with previous studies on this topic, which were limited, the researchers noted in their discussion.
The negative results also might stem from factors for which the researchers could not adjust, including insufficient cannabis smoke exposure among users in the study population, noninhalational cannabis use, which is less likely to have a respiratory effect, and possible secondhand exposure among control persons.
“It is also possible that our analysis might have been insufficiently powered to detect a significant signal with respect to the primary outcome,” they noted.
However, after the researchers controlled for multiple variables, the risk of an equally important morbidity outcome, all-cause ED visits or hospitalizations, was significantly greater among cannabis users than among control individuals, and respiratory reasons were the second most common cause for ED visits and hospitalizations in the all-cause outcome, they emphasized.
The study findings were limited by several factors, including the retrospective and observational design and the inability to control for all confounding variables, the researchers noted. Other limitations include the use of self-reports and potential for bias, the inability to perform dose-response analysis, and the high number of infrequent cannabis users in the study population.
However, the results suggest that cannabis use is associated with an increased risk of serious health events and should be discouraged, although more research is needed to confirm the current study findings, they concluded.
Consider range of causes for cannabis emergency visits
“With growing numbers of states legalizing recreational use of cannabis, it’s important to understand whether cannabis use is associated with increased emergency department visits,” Robert D. Glatter, MD, an emergency medicine physician at Lenox Hill Hospital, New York, told this news organization.
Previous studies have shown an association between increased ED visits and cannabis use in states, especially with edibles, where cannabis is legal, and “the current study reinforces the elevated risk of ED visits along with hospitalizations,” he said.
“While the researchers found no increased risk of respiratory-related complaints among users compared to the general population, there was an associated increase in ED visits and hospitalizations, which is important to understand,” said Dr. Glatter, who was not involved in the study.
“While this observational study found that the incidence of respiratory complaints was not significantly different among frequent users of cannabis, the increased odds that cannabis users would require evaluation in the emergency room or even hospitalization was still apparent even after the investigators controlled for such factors as use of alcohol, tobacco, illicit drug use, or other mental health–related disorders,” Dr. Glatter noted.
“That said, it’s a bit surprising that with the continued popularity of vaping, especially among teens, there was still not any appreciable or significant increase in respiratory complaints observed. Beyond this finding, I was not surprised by the overall conclusions of the current study, as we continue to see an elevated number of patients presenting to the ED with adverse events related to cannabis use.”
Dr. Glatter noted that “the majority of patients we see in the ED are associated with use of edibles, since it takes longer for the person to feel the effects, leading the user to consume more of the product up front, with delayed effects lasting up to 12 hours. This is what gets people into trouble and leads to toxicity of cannabis, or ‘overdoses,’ “ he explained.
When consuming edible cannabis products, “[p]eople need to begin at low dosages and not take additional gummies up front, since it can take up to 2 or even 3 hours in some cases to feel the initial effects. With the drug’s effects lasting up to 12 hours, it’s especially important to avoid operating any motor vehicles, bicycles, or scooters, since reaction time is impaired, as well as overall judgment, balance, and fine motor skills,” Dr. Glatter said.
Cannabis can land users in the ED for a range of reasons, said Dr. Glatter. “According to the study, 15% of the emergency room visits and hospitalizations were due to acute trauma, 14% due to respiratory issues, and 13% to gastrointestinal illnesses. These effects were seen in first-time users but not those with chronic use, according to the study inclusion criteria.”
Cannabis use could result in physical injuries through “impaired judgment, coordination, combined with an altered state of consciousness or generalized drowsiness, that could contribute to an increase in motor vehicle collisions, along with an increased risk for falls leading to lacerations, fractures, contusions, or bruising,” said Dr. Glatter. “Cannabis may also lead to an altered sense of perception related to interactions with others, resulting in feelings of anxiety or restlessness culminating in physical altercations and other injuries.”
The current study indicates the need for understanding the potential physical and psychological effects of cannabis use, he said.
“Additional research is needed to better understand the relative percentage cases related to edibles vs. inhalation presenting to the ED,” he noted. “There is no question that edibles continue to present significant dangers for those who don’t read labels or remain poorly informed regarding their dosing as a result of delayed onset and longer duration,” he said. To help reduce risk of toxicity, the concept of a “high lasting 12-15 hours, as with edibles, as opposed to 3-4 hours from inhalation must be clearly stated on packaging and better communicated with users, as the toxicity with edibles is more often from lack of prior knowledge about onset of effects related to dosing.”
In addition, the “potential for psychosis to develop with more chronic cannabis use, along with cannabinoid hyperemesis syndrome should be on every clinician’s radar,” Dr. Glatter emphasized.
“The bottom line is that as more states legalize the use of cannabis, it’s vital to also implement comprehensive public education efforts to provide users with the reported risks associated with not only inhalation (vaping or flower) but also edibles, which account for an increasingly greater percentage of ED visits and associated adverse effects,” he said.
The study was supported by the Lung Association–Ontario, as well as by grants from the Ontario Ministry of Health and the Ministry of Long-Term Care. The researchers and Dr. Glatter have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.