User login
For MD-IQ use only
A valuable learning experience
It was a pleasure to serve as editor-in-chief (EIC) of GI & Hepatology News from 2011 to 2016. As the second EIC of the newspaper, I was preceded by Dr. Charles Lightdale – big shoes to fill! I was fortunate to attract a strong group of associate editors who covered many key areas of interest for the paper’s readership. With the enthusiastic support of American Gastroenterological Association staff members, we published once-monthly and received generally positive feedback from readers – predominantly U.S.-based AGA members.
Serving as EIC was also a learning opportunity for me. A number of potentially newsworthy items were brought to my attention – some of which I would not otherwise have seen. Although not all were of direct relevance to the readership, I believe that most of those we published were of value.
One rewarding aspect of the editorship was the opportunity to liaise with those experts from whom I solicited commentaries on some of our featured items. These busy individuals were consistently generous with their time and expertise, and I believe that their contributions added to the paper’s overall appeal.
I initiated the inclusion of two DDSEP questions per edition, and am pleased that this feature continues. One less successful venture was the attempt at a Correspondence section, which ultimately proved too cumbersome to maintain.
I congratulate the AGA on the 15th anniversary of GIHN, and I wish the current EIC, Dr. Megan Adams, and her editorial colleagues continued success in providing this benefit to AGA members.
Colin W. Howden, MD, AGAF, is professor emeritus in the division of gastroenterology, department of medicine, at the University of Tennessee, Memphis. He is a consultant for Allakos, Ironwood, Phathom, and RedHill Biopharma. He is a member of speakers’ bureaus for Alnylam, RedHill Biopharma, and Sanofi/Genzyme. He owns stock in Antibe Therapeutics.
It was a pleasure to serve as editor-in-chief (EIC) of GI & Hepatology News from 2011 to 2016. As the second EIC of the newspaper, I was preceded by Dr. Charles Lightdale – big shoes to fill! I was fortunate to attract a strong group of associate editors who covered many key areas of interest for the paper’s readership. With the enthusiastic support of American Gastroenterological Association staff members, we published once-monthly and received generally positive feedback from readers – predominantly U.S.-based AGA members.
Serving as EIC was also a learning opportunity for me. A number of potentially newsworthy items were brought to my attention – some of which I would not otherwise have seen. Although not all were of direct relevance to the readership, I believe that most of those we published were of value.
One rewarding aspect of the editorship was the opportunity to liaise with those experts from whom I solicited commentaries on some of our featured items. These busy individuals were consistently generous with their time and expertise, and I believe that their contributions added to the paper’s overall appeal.
I initiated the inclusion of two DDSEP questions per edition, and am pleased that this feature continues. One less successful venture was the attempt at a Correspondence section, which ultimately proved too cumbersome to maintain.
I congratulate the AGA on the 15th anniversary of GIHN, and I wish the current EIC, Dr. Megan Adams, and her editorial colleagues continued success in providing this benefit to AGA members.
Colin W. Howden, MD, AGAF, is professor emeritus in the division of gastroenterology, department of medicine, at the University of Tennessee, Memphis. He is a consultant for Allakos, Ironwood, Phathom, and RedHill Biopharma. He is a member of speakers’ bureaus for Alnylam, RedHill Biopharma, and Sanofi/Genzyme. He owns stock in Antibe Therapeutics.
It was a pleasure to serve as editor-in-chief (EIC) of GI & Hepatology News from 2011 to 2016. As the second EIC of the newspaper, I was preceded by Dr. Charles Lightdale – big shoes to fill! I was fortunate to attract a strong group of associate editors who covered many key areas of interest for the paper’s readership. With the enthusiastic support of American Gastroenterological Association staff members, we published once-monthly and received generally positive feedback from readers – predominantly U.S.-based AGA members.
Serving as EIC was also a learning opportunity for me. A number of potentially newsworthy items were brought to my attention – some of which I would not otherwise have seen. Although not all were of direct relevance to the readership, I believe that most of those we published were of value.
One rewarding aspect of the editorship was the opportunity to liaise with those experts from whom I solicited commentaries on some of our featured items. These busy individuals were consistently generous with their time and expertise, and I believe that their contributions added to the paper’s overall appeal.
I initiated the inclusion of two DDSEP questions per edition, and am pleased that this feature continues. One less successful venture was the attempt at a Correspondence section, which ultimately proved too cumbersome to maintain.
I congratulate the AGA on the 15th anniversary of GIHN, and I wish the current EIC, Dr. Megan Adams, and her editorial colleagues continued success in providing this benefit to AGA members.
Colin W. Howden, MD, AGAF, is professor emeritus in the division of gastroenterology, department of medicine, at the University of Tennessee, Memphis. He is a consultant for Allakos, Ironwood, Phathom, and RedHill Biopharma. He is a member of speakers’ bureaus for Alnylam, RedHill Biopharma, and Sanofi/Genzyme. He owns stock in Antibe Therapeutics.
A bittersweet farewell
Dear colleagues,
It is with bittersweet sentiments that I introduce my last issue of The New Gastroenterologist as its Editor-in-Chief. As I reflect on my time as EIC, I am immensely grateful to the AGA for this opportunity and am proud of the journal’s continuing evolution.
To begin with this issue’s content, our “In Focus” clinical feature reviews nonalcoholic fatty liver disease (NAFLD) and is written by Dr. Naga Chalasani and Dr. Eduardo Vilar-Gomez (Indiana University). This is an excellent, comprehensive piece that details the diagnosis of NAFLD and a multifaceted management approach including dietary and lifestyle modifications, pharmacotherapy, and surgical options.
Learning endoscopy is hard, but so is teaching it. Dr. Navin Kumar (Brigham and Women’s Hospital) lends tangible advice to faculty on how to optimize their endoscopy teaching skills.
Our short clinical review for this quarter, brought to you by Dr. Grace Kim and Dr. Uzma Siddiqui (University of Chicago), offers a helpful discussion on appropriate endoscopic management of duodenal and ampullary adenomas.
Statistical concepts can often be difficult to understand; Dr. Manol Jovani (University of Kentucky) provides a useful, practical explanation of effect modification.
The AGA editorial fellowship is a wonderful opportunity and we are fortunate to have two current fellows share their experience in this issue. Dr. Judy Trieu (Loyola University Chicago) discusses her experience with Clinical Gastroenterology and Hepatology and Dr. Helenie Kefalakes (Hannover Medical School – Germany) reports on her time with Gastroenterology.
Ethics manifests itself in gastroenterology in many ways, and I am happy to have introduced a case-based ethics series to our publication. For my last issue, Dr. Ariel Sims (University of Chicago) and I discuss a case of repeated deliberate foreign body ingestion and the ethical considerations for us as endoscopists.
It can be difficult to navigate the many options that exist within the realm of disability insurance. Dr. Trevor Smith (Advanced EyeCare Professionals) reviews the reasons to obtain disability insurance, how to apply and what to look for in a policy.
Lastly, the DHPA Private Practice Perspectives article for this issue is written by Dr. Marc Sonenshine (Atlanta Gastroenterology Associates) who shares his practice’s innovative approach to implementing a formalized mentorship program for physicians early in their career.
As my editorship comes to a close, I would be remiss not to thank several key people who have been instrumental in the last 3 years. First, Dr. Gautham Reddy, an important mentor of mine and former program director who sent me this opportunity and encouraged me to apply. In addition, I am grateful to the chief of our Section at the University of Chicago, Dr. David Rubin, for his collaboration and support. Ryan Farrell, the managing editor of TNG, has been nothing short of amazing to work with and is the true backbone of our publication. I would also like to thank the staff of our publisher, Frontline Medical Communications, especially Kathy Scarbeck and Christopher Palmer, as well as the EIC and former EIC of our parent publication, GI & Hepatology News, Dr. Megan Adams and Dr. John Allen. Finally, thank you to our readers, whose continued interest has made TNG a success.
Taken together, my experience as EIC of TNG for the last 3 years has been unparalleled. The opportunity to translate the questions, challenges, and experiences of early-career gastroenterologists into written pieces that would be shared with the international community is one I truly never thought I would find within a career in medicine. I am excited for the future and growth of TNG in the hands of a new EIC.
If you have interest in contributing or have ideas for future TNG topics, please contact me (vijayarao@medicine.bsd.uchicago.edu) or Ryan Farrell (rfarrell@gastro.org), managing editor of TNG.
Respectfully,
Vijaya L. Rao, MD
Editor-in-Chief
Dear colleagues,
It is with bittersweet sentiments that I introduce my last issue of The New Gastroenterologist as its Editor-in-Chief. As I reflect on my time as EIC, I am immensely grateful to the AGA for this opportunity and am proud of the journal’s continuing evolution.
To begin with this issue’s content, our “In Focus” clinical feature reviews nonalcoholic fatty liver disease (NAFLD) and is written by Dr. Naga Chalasani and Dr. Eduardo Vilar-Gomez (Indiana University). This is an excellent, comprehensive piece that details the diagnosis of NAFLD and a multifaceted management approach including dietary and lifestyle modifications, pharmacotherapy, and surgical options.
Learning endoscopy is hard, but so is teaching it. Dr. Navin Kumar (Brigham and Women’s Hospital) lends tangible advice to faculty on how to optimize their endoscopy teaching skills.
Our short clinical review for this quarter, brought to you by Dr. Grace Kim and Dr. Uzma Siddiqui (University of Chicago), offers a helpful discussion on appropriate endoscopic management of duodenal and ampullary adenomas.
Statistical concepts can often be difficult to understand; Dr. Manol Jovani (University of Kentucky) provides a useful, practical explanation of effect modification.
The AGA editorial fellowship is a wonderful opportunity and we are fortunate to have two current fellows share their experience in this issue. Dr. Judy Trieu (Loyola University Chicago) discusses her experience with Clinical Gastroenterology and Hepatology and Dr. Helenie Kefalakes (Hannover Medical School – Germany) reports on her time with Gastroenterology.
Ethics manifests itself in gastroenterology in many ways, and I am happy to have introduced a case-based ethics series to our publication. For my last issue, Dr. Ariel Sims (University of Chicago) and I discuss a case of repeated deliberate foreign body ingestion and the ethical considerations for us as endoscopists.
It can be difficult to navigate the many options that exist within the realm of disability insurance. Dr. Trevor Smith (Advanced EyeCare Professionals) reviews the reasons to obtain disability insurance, how to apply and what to look for in a policy.
Lastly, the DHPA Private Practice Perspectives article for this issue is written by Dr. Marc Sonenshine (Atlanta Gastroenterology Associates) who shares his practice’s innovative approach to implementing a formalized mentorship program for physicians early in their career.
As my editorship comes to a close, I would be remiss not to thank several key people who have been instrumental in the last 3 years. First, Dr. Gautham Reddy, an important mentor of mine and former program director who sent me this opportunity and encouraged me to apply. In addition, I am grateful to the chief of our Section at the University of Chicago, Dr. David Rubin, for his collaboration and support. Ryan Farrell, the managing editor of TNG, has been nothing short of amazing to work with and is the true backbone of our publication. I would also like to thank the staff of our publisher, Frontline Medical Communications, especially Kathy Scarbeck and Christopher Palmer, as well as the EIC and former EIC of our parent publication, GI & Hepatology News, Dr. Megan Adams and Dr. John Allen. Finally, thank you to our readers, whose continued interest has made TNG a success.
Taken together, my experience as EIC of TNG for the last 3 years has been unparalleled. The opportunity to translate the questions, challenges, and experiences of early-career gastroenterologists into written pieces that would be shared with the international community is one I truly never thought I would find within a career in medicine. I am excited for the future and growth of TNG in the hands of a new EIC.
If you have interest in contributing or have ideas for future TNG topics, please contact me (vijayarao@medicine.bsd.uchicago.edu) or Ryan Farrell (rfarrell@gastro.org), managing editor of TNG.
Respectfully,
Vijaya L. Rao, MD
Editor-in-Chief
Dear colleagues,
It is with bittersweet sentiments that I introduce my last issue of The New Gastroenterologist as its Editor-in-Chief. As I reflect on my time as EIC, I am immensely grateful to the AGA for this opportunity and am proud of the journal’s continuing evolution.
To begin with this issue’s content, our “In Focus” clinical feature reviews nonalcoholic fatty liver disease (NAFLD) and is written by Dr. Naga Chalasani and Dr. Eduardo Vilar-Gomez (Indiana University). This is an excellent, comprehensive piece that details the diagnosis of NAFLD and a multifaceted management approach including dietary and lifestyle modifications, pharmacotherapy, and surgical options.
Learning endoscopy is hard, but so is teaching it. Dr. Navin Kumar (Brigham and Women’s Hospital) lends tangible advice to faculty on how to optimize their endoscopy teaching skills.
Our short clinical review for this quarter, brought to you by Dr. Grace Kim and Dr. Uzma Siddiqui (University of Chicago), offers a helpful discussion on appropriate endoscopic management of duodenal and ampullary adenomas.
Statistical concepts can often be difficult to understand; Dr. Manol Jovani (University of Kentucky) provides a useful, practical explanation of effect modification.
The AGA editorial fellowship is a wonderful opportunity and we are fortunate to have two current fellows share their experience in this issue. Dr. Judy Trieu (Loyola University Chicago) discusses her experience with Clinical Gastroenterology and Hepatology and Dr. Helenie Kefalakes (Hannover Medical School – Germany) reports on her time with Gastroenterology.
Ethics manifests itself in gastroenterology in many ways, and I am happy to have introduced a case-based ethics series to our publication. For my last issue, Dr. Ariel Sims (University of Chicago) and I discuss a case of repeated deliberate foreign body ingestion and the ethical considerations for us as endoscopists.
It can be difficult to navigate the many options that exist within the realm of disability insurance. Dr. Trevor Smith (Advanced EyeCare Professionals) reviews the reasons to obtain disability insurance, how to apply and what to look for in a policy.
Lastly, the DHPA Private Practice Perspectives article for this issue is written by Dr. Marc Sonenshine (Atlanta Gastroenterology Associates) who shares his practice’s innovative approach to implementing a formalized mentorship program for physicians early in their career.
As my editorship comes to a close, I would be remiss not to thank several key people who have been instrumental in the last 3 years. First, Dr. Gautham Reddy, an important mentor of mine and former program director who sent me this opportunity and encouraged me to apply. In addition, I am grateful to the chief of our Section at the University of Chicago, Dr. David Rubin, for his collaboration and support. Ryan Farrell, the managing editor of TNG, has been nothing short of amazing to work with and is the true backbone of our publication. I would also like to thank the staff of our publisher, Frontline Medical Communications, especially Kathy Scarbeck and Christopher Palmer, as well as the EIC and former EIC of our parent publication, GI & Hepatology News, Dr. Megan Adams and Dr. John Allen. Finally, thank you to our readers, whose continued interest has made TNG a success.
Taken together, my experience as EIC of TNG for the last 3 years has been unparalleled. The opportunity to translate the questions, challenges, and experiences of early-career gastroenterologists into written pieces that would be shared with the international community is one I truly never thought I would find within a career in medicine. I am excited for the future and growth of TNG in the hands of a new EIC.
If you have interest in contributing or have ideas for future TNG topics, please contact me (vijayarao@medicine.bsd.uchicago.edu) or Ryan Farrell (rfarrell@gastro.org), managing editor of TNG.
Respectfully,
Vijaya L. Rao, MD
Editor-in-Chief
Don’t let insurance policies burden GI practices
Join us at AGA Advocacy Day on Thursday, Sept. 22, 2022, to virtually meet with your members of Congress to urge them to rein in insurance policies like prior authorization and step therapy.
If GI providers don’t have a seat at the table and engage with lawmakers, these decisions will be influenced by payers and other parties that do not have your or your patients’ best interests at heart.
AGA Advocacy Day is held shortly before the end of the fiscal year – prime time to educate policymakers and their staff about your everyday challenges and the reality of GI patient care in your state. We will also discuss the need for robust federal funding for GI research and the devastating impact that Medicare cuts could have on your practice.
Register today and AGA will take care of the rest, including scheduling your meetings and providing comprehensive advocacy training. Now more than ever, your voice needs to be heard on Capitol Hill.
Join us at AGA Advocacy Day on Thursday, Sept. 22, 2022, to virtually meet with your members of Congress to urge them to rein in insurance policies like prior authorization and step therapy.
If GI providers don’t have a seat at the table and engage with lawmakers, these decisions will be influenced by payers and other parties that do not have your or your patients’ best interests at heart.
AGA Advocacy Day is held shortly before the end of the fiscal year – prime time to educate policymakers and their staff about your everyday challenges and the reality of GI patient care in your state. We will also discuss the need for robust federal funding for GI research and the devastating impact that Medicare cuts could have on your practice.
Register today and AGA will take care of the rest, including scheduling your meetings and providing comprehensive advocacy training. Now more than ever, your voice needs to be heard on Capitol Hill.
Join us at AGA Advocacy Day on Thursday, Sept. 22, 2022, to virtually meet with your members of Congress to urge them to rein in insurance policies like prior authorization and step therapy.
If GI providers don’t have a seat at the table and engage with lawmakers, these decisions will be influenced by payers and other parties that do not have your or your patients’ best interests at heart.
AGA Advocacy Day is held shortly before the end of the fiscal year – prime time to educate policymakers and their staff about your everyday challenges and the reality of GI patient care in your state. We will also discuss the need for robust federal funding for GI research and the devastating impact that Medicare cuts could have on your practice.
Register today and AGA will take care of the rest, including scheduling your meetings and providing comprehensive advocacy training. Now more than ever, your voice needs to be heard on Capitol Hill.
You can make a difference
The American Gastroenterological Association Research Foundation is the charitable arm of the AGA and plays an important role in medical research by providing grants to young scientists at a critical time in their career.
“I am beyond excited to be able to continue my journey as a young physician-scientist with the funding support. I truly understand that my career would not be possible without those who generously support scientific research. I am humbled at the opportunity to contribute to something larger than myself. As such, I am extremely grateful to the AGA Research Foundation and its donors who continue to support this vision of a future where suffering related to digestive diseases has been eliminated,” said Brian A. Sullivan, MD, a 2021 AGA Research Scholar Award recipient, who is a physician-scientist who aspires to lead a collaborative research program and develop more effective strategies for colorectal cancer prevention.
In the past decade alone, we’ve witnessed seminal work in colorectal cancer genetics and a renaissance in the understanding of inflammatory bowel syndrome and the gut microbiome. However, continued progress in advancing the treatment and cure of digestive diseases is at risk because of cuts in government spending. Without help from other funding sources, young investigators are struggling to continue their research, build their research portfolio, and obtain federal funding.
Your contribution makes a difference
With donations from AGA members, we can provide young researchers with a secure, ongoing stable source of funding that drives advancement in the diagnosis, treatment and cure of digestive diseases. Everyone benefits from GI research developed by dedicated investigators.
“I donated to the AGA Research Foundation to ensure the vitality of our specialty, and to fund the research of future generations of gastroenterologists. Funding from organizations like the AGA Research Foundation is crucial for young scientists and gastroenterologists to launch their careers. At the start of my career, I received two AGA research awards. As a grateful recipient of such funding, I felt it was my turn to support the mission of the organization that I regard as my academic home away from home institution,” said Michael Camilleri, MD, AGAF, chair of the AGA Research Foundation and AGA Past President.
Many breakthroughs have been achieved through gastroenterological and hepatological research over the past century, forming the basis of the modern medical practice. Join fellow AGA members by contributing to this tradition of discovery.
Make a tax-deductible donation to the AGA Research Foundation at www.gastro.org/donate or by mail to 4930 Del Ray Avenue, Bethesda, MD 20814.
Learn more about the AGA Research Foundation at https://foundation.gastro.org.
The American Gastroenterological Association Research Foundation is the charitable arm of the AGA and plays an important role in medical research by providing grants to young scientists at a critical time in their career.
“I am beyond excited to be able to continue my journey as a young physician-scientist with the funding support. I truly understand that my career would not be possible without those who generously support scientific research. I am humbled at the opportunity to contribute to something larger than myself. As such, I am extremely grateful to the AGA Research Foundation and its donors who continue to support this vision of a future where suffering related to digestive diseases has been eliminated,” said Brian A. Sullivan, MD, a 2021 AGA Research Scholar Award recipient, who is a physician-scientist who aspires to lead a collaborative research program and develop more effective strategies for colorectal cancer prevention.
In the past decade alone, we’ve witnessed seminal work in colorectal cancer genetics and a renaissance in the understanding of inflammatory bowel syndrome and the gut microbiome. However, continued progress in advancing the treatment and cure of digestive diseases is at risk because of cuts in government spending. Without help from other funding sources, young investigators are struggling to continue their research, build their research portfolio, and obtain federal funding.
Your contribution makes a difference
With donations from AGA members, we can provide young researchers with a secure, ongoing stable source of funding that drives advancement in the diagnosis, treatment and cure of digestive diseases. Everyone benefits from GI research developed by dedicated investigators.
“I donated to the AGA Research Foundation to ensure the vitality of our specialty, and to fund the research of future generations of gastroenterologists. Funding from organizations like the AGA Research Foundation is crucial for young scientists and gastroenterologists to launch their careers. At the start of my career, I received two AGA research awards. As a grateful recipient of such funding, I felt it was my turn to support the mission of the organization that I regard as my academic home away from home institution,” said Michael Camilleri, MD, AGAF, chair of the AGA Research Foundation and AGA Past President.
Many breakthroughs have been achieved through gastroenterological and hepatological research over the past century, forming the basis of the modern medical practice. Join fellow AGA members by contributing to this tradition of discovery.
Make a tax-deductible donation to the AGA Research Foundation at www.gastro.org/donate or by mail to 4930 Del Ray Avenue, Bethesda, MD 20814.
Learn more about the AGA Research Foundation at https://foundation.gastro.org.
The American Gastroenterological Association Research Foundation is the charitable arm of the AGA and plays an important role in medical research by providing grants to young scientists at a critical time in their career.
“I am beyond excited to be able to continue my journey as a young physician-scientist with the funding support. I truly understand that my career would not be possible without those who generously support scientific research. I am humbled at the opportunity to contribute to something larger than myself. As such, I am extremely grateful to the AGA Research Foundation and its donors who continue to support this vision of a future where suffering related to digestive diseases has been eliminated,” said Brian A. Sullivan, MD, a 2021 AGA Research Scholar Award recipient, who is a physician-scientist who aspires to lead a collaborative research program and develop more effective strategies for colorectal cancer prevention.
In the past decade alone, we’ve witnessed seminal work in colorectal cancer genetics and a renaissance in the understanding of inflammatory bowel syndrome and the gut microbiome. However, continued progress in advancing the treatment and cure of digestive diseases is at risk because of cuts in government spending. Without help from other funding sources, young investigators are struggling to continue their research, build their research portfolio, and obtain federal funding.
Your contribution makes a difference
With donations from AGA members, we can provide young researchers with a secure, ongoing stable source of funding that drives advancement in the diagnosis, treatment and cure of digestive diseases. Everyone benefits from GI research developed by dedicated investigators.
“I donated to the AGA Research Foundation to ensure the vitality of our specialty, and to fund the research of future generations of gastroenterologists. Funding from organizations like the AGA Research Foundation is crucial for young scientists and gastroenterologists to launch their careers. At the start of my career, I received two AGA research awards. As a grateful recipient of such funding, I felt it was my turn to support the mission of the organization that I regard as my academic home away from home institution,” said Michael Camilleri, MD, AGAF, chair of the AGA Research Foundation and AGA Past President.
Many breakthroughs have been achieved through gastroenterological and hepatological research over the past century, forming the basis of the modern medical practice. Join fellow AGA members by contributing to this tradition of discovery.
Make a tax-deductible donation to the AGA Research Foundation at www.gastro.org/donate or by mail to 4930 Del Ray Avenue, Bethesda, MD 20814.
Learn more about the AGA Research Foundation at https://foundation.gastro.org.
What's your diagnosis?
Pancreatic adenocarcinoma arising from main duct intraductal papillary mucinous neoplasm with inadvertent main pancreatic duct stenting.
The FNA was positive for carcinoma with abundant mucin, which, taken together with the imaging findings, was indicative of pancreatic adenocarcinoma arising from main duct intraductal papillary mucinous neoplasm (M-IPMN).
The post-endoscopic retrograde cholangiopancreatography (ERCP) CT revealed inadvertent placement of the fully covered self-expanding metallic stent (fcSEMS) within the main pancreatic duct (MPD) stricture and persistent common bile duct (CBD) obstruction. On post hoc review of the fluoroscopic and cross-sectional imaging, it became evident that the massively dilated MPD was mistaken during ERCP for the CBD and left hepatic duct (Figure F). In addition, the patient also had several cysts within the liver (compatible with incidental polycystic liver disease), which further complicated real-time image interpretation.
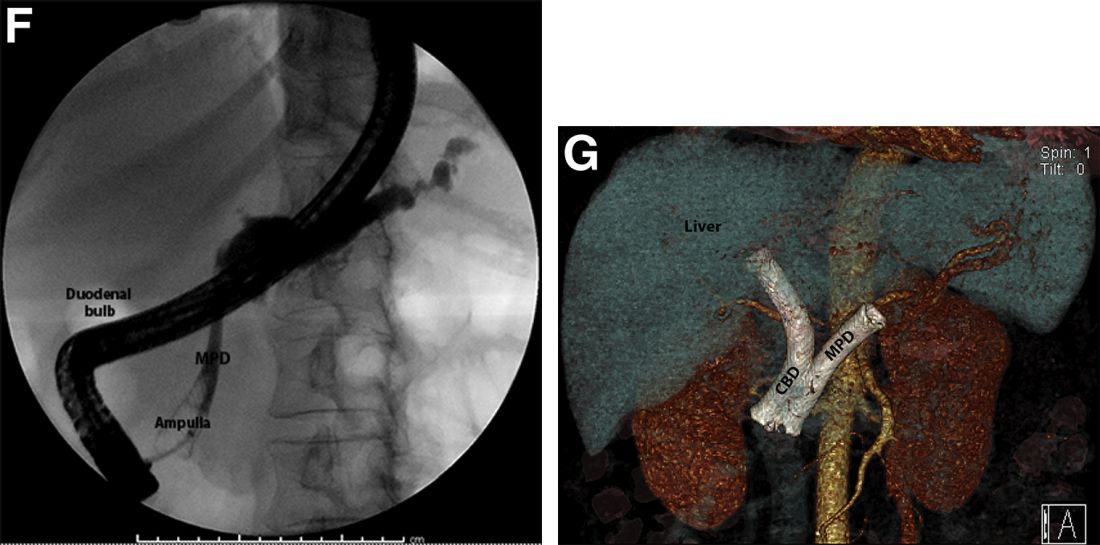
Based on multidisciplinary discussion, the precedent of a prior series of successful palliative MPD stenting in the setting of adenocarcinoma,1 and the notable improvement in the patient's steatorrhea and abdominal pain, the initially placed fcSEMS was left in situ across the MPD stricture, and a second fcSEMS was successfully deployed across the CBD stricture (Figure G), resulting in prompt improvement in serum liver tests. The patient was thereafter initiated on palliative chemotherapy with gemcitabine and abraxane and has maintained clinically stable disease for the last 9 months.
M-IPMN is a premalignant condition in which endoscopy plays an important role. In our patient, because of anatomic and morphologic abnormalities, including the massive dilation of the MPD and severe distal biliary compression in the context of an obstructing pancreatic head mass arising from M-IPMN, initial deployment of the fcSEMS occurred unwittingly into the MPD. Little is known about the impact of fcSEMS in the MPD in patients with pancreatic adenocarcinoma, although in select cases, alleviation of pain caused by MPD obstruction and improvement in quality of life have been reported.2,3 In the case of our patient, fcSEMS placement in the MPD indeed led to symptomatic relief as manifested by a decrease in both diarrhea and pain and an increase in appetite; the addition of a fcSEMS in the CBD led to serum liver test normalization and permitted the initiation of chemotherapy. Further studies are needed to examine the outcomes of palliative MPD stenting in patients with obstructing pancreatic malignancies as well as the epidemiology and biology of M-IPMN and associated pancreatic adenocarcinoma in minority populations.
References
1. Tham TC et al. Am J Gastroenterol. 2000 Apr;95(4):956-60.
2. Grimm IS, Baron TH. Gastroenterology. 2015 Jul;149(1):20-2.
3. Wehrmann T et al. Eur J Gastroenterol Hepatol. 2005 Dec;17(12):1395-400.
Pancreatic adenocarcinoma arising from main duct intraductal papillary mucinous neoplasm with inadvertent main pancreatic duct stenting.
The FNA was positive for carcinoma with abundant mucin, which, taken together with the imaging findings, was indicative of pancreatic adenocarcinoma arising from main duct intraductal papillary mucinous neoplasm (M-IPMN).
The post-endoscopic retrograde cholangiopancreatography (ERCP) CT revealed inadvertent placement of the fully covered self-expanding metallic stent (fcSEMS) within the main pancreatic duct (MPD) stricture and persistent common bile duct (CBD) obstruction. On post hoc review of the fluoroscopic and cross-sectional imaging, it became evident that the massively dilated MPD was mistaken during ERCP for the CBD and left hepatic duct (Figure F). In addition, the patient also had several cysts within the liver (compatible with incidental polycystic liver disease), which further complicated real-time image interpretation.

Based on multidisciplinary discussion, the precedent of a prior series of successful palliative MPD stenting in the setting of adenocarcinoma,1 and the notable improvement in the patient's steatorrhea and abdominal pain, the initially placed fcSEMS was left in situ across the MPD stricture, and a second fcSEMS was successfully deployed across the CBD stricture (Figure G), resulting in prompt improvement in serum liver tests. The patient was thereafter initiated on palliative chemotherapy with gemcitabine and abraxane and has maintained clinically stable disease for the last 9 months.
M-IPMN is a premalignant condition in which endoscopy plays an important role. In our patient, because of anatomic and morphologic abnormalities, including the massive dilation of the MPD and severe distal biliary compression in the context of an obstructing pancreatic head mass arising from M-IPMN, initial deployment of the fcSEMS occurred unwittingly into the MPD. Little is known about the impact of fcSEMS in the MPD in patients with pancreatic adenocarcinoma, although in select cases, alleviation of pain caused by MPD obstruction and improvement in quality of life have been reported.2,3 In the case of our patient, fcSEMS placement in the MPD indeed led to symptomatic relief as manifested by a decrease in both diarrhea and pain and an increase in appetite; the addition of a fcSEMS in the CBD led to serum liver test normalization and permitted the initiation of chemotherapy. Further studies are needed to examine the outcomes of palliative MPD stenting in patients with obstructing pancreatic malignancies as well as the epidemiology and biology of M-IPMN and associated pancreatic adenocarcinoma in minority populations.
References
1. Tham TC et al. Am J Gastroenterol. 2000 Apr;95(4):956-60.
2. Grimm IS, Baron TH. Gastroenterology. 2015 Jul;149(1):20-2.
3. Wehrmann T et al. Eur J Gastroenterol Hepatol. 2005 Dec;17(12):1395-400.
Pancreatic adenocarcinoma arising from main duct intraductal papillary mucinous neoplasm with inadvertent main pancreatic duct stenting.
The FNA was positive for carcinoma with abundant mucin, which, taken together with the imaging findings, was indicative of pancreatic adenocarcinoma arising from main duct intraductal papillary mucinous neoplasm (M-IPMN).
The post-endoscopic retrograde cholangiopancreatography (ERCP) CT revealed inadvertent placement of the fully covered self-expanding metallic stent (fcSEMS) within the main pancreatic duct (MPD) stricture and persistent common bile duct (CBD) obstruction. On post hoc review of the fluoroscopic and cross-sectional imaging, it became evident that the massively dilated MPD was mistaken during ERCP for the CBD and left hepatic duct (Figure F). In addition, the patient also had several cysts within the liver (compatible with incidental polycystic liver disease), which further complicated real-time image interpretation.

Based on multidisciplinary discussion, the precedent of a prior series of successful palliative MPD stenting in the setting of adenocarcinoma,1 and the notable improvement in the patient's steatorrhea and abdominal pain, the initially placed fcSEMS was left in situ across the MPD stricture, and a second fcSEMS was successfully deployed across the CBD stricture (Figure G), resulting in prompt improvement in serum liver tests. The patient was thereafter initiated on palliative chemotherapy with gemcitabine and abraxane and has maintained clinically stable disease for the last 9 months.
M-IPMN is a premalignant condition in which endoscopy plays an important role. In our patient, because of anatomic and morphologic abnormalities, including the massive dilation of the MPD and severe distal biliary compression in the context of an obstructing pancreatic head mass arising from M-IPMN, initial deployment of the fcSEMS occurred unwittingly into the MPD. Little is known about the impact of fcSEMS in the MPD in patients with pancreatic adenocarcinoma, although in select cases, alleviation of pain caused by MPD obstruction and improvement in quality of life have been reported.2,3 In the case of our patient, fcSEMS placement in the MPD indeed led to symptomatic relief as manifested by a decrease in both diarrhea and pain and an increase in appetite; the addition of a fcSEMS in the CBD led to serum liver test normalization and permitted the initiation of chemotherapy. Further studies are needed to examine the outcomes of palliative MPD stenting in patients with obstructing pancreatic malignancies as well as the epidemiology and biology of M-IPMN and associated pancreatic adenocarcinoma in minority populations.
References
1. Tham TC et al. Am J Gastroenterol. 2000 Apr;95(4):956-60.
2. Grimm IS, Baron TH. Gastroenterology. 2015 Jul;149(1):20-2.
3. Wehrmann T et al. Eur J Gastroenterol Hepatol. 2005 Dec;17(12):1395-400.
A 69-year-old Filipino American woman presented with increasing epigastralgia, worsening appetite, jaundice, and oily diarrhea over the course of 3 months. Her past medical history consisted of diabetes, hypertension, hyperlipidemia, and osteopenia being managed with metformin, losartan, and atorvastatin, respectively.
Physical examination revealed she was thin (body mass index, 22 kg/m2) and jaundiced with moderate tenderness to epigastric palpation and 1+ peripheral pitting edema. Laboratory tests were significant for normal complete blood count and elevated alanine aminotransferase (113 U/L), alkaline phosphatase (235 U/L), bilirubin (7.3 mg/dL), international normalized ratio (1.3), and carbohydrate antigen 19-9 (7886 U/L). A CT scan of the abdomen revealed severe extrahepatic and intrahepatic ductal dilation, with a common bile duct (CBD) and main pancreatic duct (MPD) diameter of 2.5 and 1.7 cm, respectively, as well an infiltrating, malignant-appearing, 4.5-cm spheroid mass in the head of the pancreas (Figure A). The mass involved the superior mesenteric vein at the portal confluence and encased >50% of the superior mesenteric artery.

To further characterize these findings, magnetic resonance cholangiopancreatography was performed, which additionally revealed multifocal cysts throughout the liver ranging from 0.5 to 5.0 cm in greatest diameter, as seen on maximal intensity projection algorithm (Figure B).
The patient was referred for same-session endoscopic ultrasound examination with fine needle aspiration (FNA) and endoscopic retrograde cholangiopancreatography (ERCP) for further diagnosis and treatment. Endoscopic ultrasound demonstrated a large, hypoechoic mass in the pancreatic head with severe CBD and MPD dilation proximally, corresponding with the cross-sectional imaging findings; FNA was performed. ERCP demonstrated a long, distal CBD stricture and what appeared to be nonopacification of the right hepatic ductal system; a 10 × 60-mm fully covered self-expanding metallic stent (fcSEMS) was placed across the stricture (Figure C, D). Over the subsequent 3 days, the patient's diarrhea resolved and epigastralgia improved; however, serum liver tests did not downtrend, thus prompting repeat imaging (Figure E).
Based on the patient's clinical history, cross-sectional imaging findings, and only partial response to therapeutic ERCP, what are the patient's likely diagnoses?
Becoming an AGA committee chair as an early-career physician
One of the cornerstones of member engagement within the American Gastroenterological Association is its committees, which provide a platform for AGA members to network, effect change at the institutional level, and obtain leadership positions. For many within the AGA, exposure to these committees occurs during training. Both of us were first introduced to the possibility of serving on an AGA committee by faculty members at our institution. Each year, applications for available committee positions open in the fall and are due on Nov. 1. While you can be nominated by other members, self-nomination is common and encouraged. Truthfully, neither of us was quite certain what committee membership would entail. However, we both applied to several committees because we knew that it would be an excellent opportunity to network with leading gastroenterologists across the country and to have the ability to become involved in key AGA programs.
We were selected to serve 2-year terms as trainee members on the Government Affairs and Publication Committees, respectively, which gave us a deeper understanding of how an organization with both a full-time professional staff and group of volunteer members functions. A unique feature of the AGA is its Trainee and Early Career (TEC) Committee, which mainly comprises trainee members who serve on other committees. By virtue of our roles with the other committees, we also became full-fledged members of the TEC committee, which is dedicated to enhancing the experience for trainees and those who are within 5 years of graduation.
One of the most innovative programs developed by the TEC committee is Career Development Workshops, which is a webinar series focused on important topics not covered in fellowship, such as different career paths, personal finance, and how to increase the number of underrepresented individuals in the field. The predecessor of the Career Development Workshops was the in-person Regional Practice Skills Workshop, and we both took on the responsibility of planning and organizing separate workshops. For one of us (Stephanie), that involved enlisting our fellowship program to host the event. For the other (Peter), it meant collaborating with our local gastroenterology society to cosponsor the workshop. It was extremely rewarding to organize the workshops, which allowed us to work closely with AGA staff and local gastroenterology faculty, as well as our peers, to bring the events to fruition. For both of us, the success of the Regional Practice Skills Workshop was one of the highlights of our tenure on the TEC committee.
Although we were not aware of it at the time, volunteering to plan a workshop and assisting with other projects and subcommittees were signs of enthusiasm and leadership that the AGA recognized and valued. Our advice on becoming a committee chair is to not only show an interest in committee projects but also to turn that interest into action. A committee member who is strongly interested in a leadership position cannot expect to transition into that role by being a “silent but present” member. You need to do more than just show up. You should actively participate in projects, engage in discussions, and devote your time and energy to ensure the success of committee programs. However, you should also make sure to have sufficient bandwidth to make meaningful contributions to each project and not commit to tasks that you cannot complete. To set yourself apart on a committee, it is important to be actively engaged and committed to a project (or two) that allows for professional growth and visibility. Ideally, you will become an integral part of a committee that sparks your drive to serve.
Applying to become a committee chair follows the same process and timeline as for any other committee position, and you can be nominated or self-nominate. Although previous experience on that specific committee is not a prerequisite for most chair positions, having previously served on any AGA committee or task force is generally required. Successful applicants serve for 1 year as chair-elect, during which they work closely with the outgoing chair and staff to ensure a smooth transition when their 3-year term as chair officially begins in June.
Each committee has a guiding mission statement and a staff liaison who provides institutional knowledge and logistical support. However, the committee members, and especially the chair, have considerable latitude to develop and implement new initiatives or retire old ones. The entire committee meets twice per year, once in September in Washington, D.C., and once at DDW. Between the meetings, working groups are formed to move the various programs forward. In addition to the Career Development Workshops, the TEC committee organizes the Young Delegates program (which allows any AGA member to volunteer on small, time-limited projects), a symposium at DDW focused on trainee and early career issues, and a networking event at DDW. Moreover, we collaborate with other committees and provide input from the perspective of younger members on larger initiatives such as the AGA Equity Project and Career Compass.
As chair, we lead the twice-yearly meetings as well as the working groups. We strongly encourage all committee members to participate on at least one working group, which develops leadership skills and provides the opportunity to moderate sessions for the Career Development Workshops and DDW symposium. Moreover, we solicit feedback on ways to improve current programming, start new initiatives, and work with other committees that the TEC committee members are part of. Trainees and early career members are seen as a key constituency group within the AGA, and we take the responsibility of increasing the value of membership for this group seriously.
As early-career physicians ourselves, we also view the chance to serve as a committee chair as a great career development opportunity. It allows us to expand our professional networks, help shape an organization that is a leading voice and advocate for digestive health, and meet the needs of young members who are the future of the AGA.
There is no doubt that all of you have achieved amazing things on the way to becoming a trainee or early career professional in the competitive fields of gastroenterology and hepatology. The AGA is constantly looking for bright, motivated individuals to serve as volunteers and future leaders. Our experience shows that with a bit of persistence to get in the door – through Young Delegates or a committee – along with lots of hard work along the way, you will be in a great position to rise through the ranks and help lead an organization at the vanguard of our field.
Dr. Liang is assistant professor of medicine and population health, New York University Langone Health, and a staff physician at VA New York Harbor Health Care System. Dr. Pointer is a founder and managing partner of Digestive and Liver Health Specialists. She is on staff as a clinical gastroenterologist at Tristar Hendersonville (Tenn.) Medical Center. They have no conflicts of interest.
One of the cornerstones of member engagement within the American Gastroenterological Association is its committees, which provide a platform for AGA members to network, effect change at the institutional level, and obtain leadership positions. For many within the AGA, exposure to these committees occurs during training. Both of us were first introduced to the possibility of serving on an AGA committee by faculty members at our institution. Each year, applications for available committee positions open in the fall and are due on Nov. 1. While you can be nominated by other members, self-nomination is common and encouraged. Truthfully, neither of us was quite certain what committee membership would entail. However, we both applied to several committees because we knew that it would be an excellent opportunity to network with leading gastroenterologists across the country and to have the ability to become involved in key AGA programs.
We were selected to serve 2-year terms as trainee members on the Government Affairs and Publication Committees, respectively, which gave us a deeper understanding of how an organization with both a full-time professional staff and group of volunteer members functions. A unique feature of the AGA is its Trainee and Early Career (TEC) Committee, which mainly comprises trainee members who serve on other committees. By virtue of our roles with the other committees, we also became full-fledged members of the TEC committee, which is dedicated to enhancing the experience for trainees and those who are within 5 years of graduation.
One of the most innovative programs developed by the TEC committee is Career Development Workshops, which is a webinar series focused on important topics not covered in fellowship, such as different career paths, personal finance, and how to increase the number of underrepresented individuals in the field. The predecessor of the Career Development Workshops was the in-person Regional Practice Skills Workshop, and we both took on the responsibility of planning and organizing separate workshops. For one of us (Stephanie), that involved enlisting our fellowship program to host the event. For the other (Peter), it meant collaborating with our local gastroenterology society to cosponsor the workshop. It was extremely rewarding to organize the workshops, which allowed us to work closely with AGA staff and local gastroenterology faculty, as well as our peers, to bring the events to fruition. For both of us, the success of the Regional Practice Skills Workshop was one of the highlights of our tenure on the TEC committee.
Although we were not aware of it at the time, volunteering to plan a workshop and assisting with other projects and subcommittees were signs of enthusiasm and leadership that the AGA recognized and valued. Our advice on becoming a committee chair is to not only show an interest in committee projects but also to turn that interest into action. A committee member who is strongly interested in a leadership position cannot expect to transition into that role by being a “silent but present” member. You need to do more than just show up. You should actively participate in projects, engage in discussions, and devote your time and energy to ensure the success of committee programs. However, you should also make sure to have sufficient bandwidth to make meaningful contributions to each project and not commit to tasks that you cannot complete. To set yourself apart on a committee, it is important to be actively engaged and committed to a project (or two) that allows for professional growth and visibility. Ideally, you will become an integral part of a committee that sparks your drive to serve.
Applying to become a committee chair follows the same process and timeline as for any other committee position, and you can be nominated or self-nominate. Although previous experience on that specific committee is not a prerequisite for most chair positions, having previously served on any AGA committee or task force is generally required. Successful applicants serve for 1 year as chair-elect, during which they work closely with the outgoing chair and staff to ensure a smooth transition when their 3-year term as chair officially begins in June.
Each committee has a guiding mission statement and a staff liaison who provides institutional knowledge and logistical support. However, the committee members, and especially the chair, have considerable latitude to develop and implement new initiatives or retire old ones. The entire committee meets twice per year, once in September in Washington, D.C., and once at DDW. Between the meetings, working groups are formed to move the various programs forward. In addition to the Career Development Workshops, the TEC committee organizes the Young Delegates program (which allows any AGA member to volunteer on small, time-limited projects), a symposium at DDW focused on trainee and early career issues, and a networking event at DDW. Moreover, we collaborate with other committees and provide input from the perspective of younger members on larger initiatives such as the AGA Equity Project and Career Compass.
As chair, we lead the twice-yearly meetings as well as the working groups. We strongly encourage all committee members to participate on at least one working group, which develops leadership skills and provides the opportunity to moderate sessions for the Career Development Workshops and DDW symposium. Moreover, we solicit feedback on ways to improve current programming, start new initiatives, and work with other committees that the TEC committee members are part of. Trainees and early career members are seen as a key constituency group within the AGA, and we take the responsibility of increasing the value of membership for this group seriously.
As early-career physicians ourselves, we also view the chance to serve as a committee chair as a great career development opportunity. It allows us to expand our professional networks, help shape an organization that is a leading voice and advocate for digestive health, and meet the needs of young members who are the future of the AGA.
There is no doubt that all of you have achieved amazing things on the way to becoming a trainee or early career professional in the competitive fields of gastroenterology and hepatology. The AGA is constantly looking for bright, motivated individuals to serve as volunteers and future leaders. Our experience shows that with a bit of persistence to get in the door – through Young Delegates or a committee – along with lots of hard work along the way, you will be in a great position to rise through the ranks and help lead an organization at the vanguard of our field.
Dr. Liang is assistant professor of medicine and population health, New York University Langone Health, and a staff physician at VA New York Harbor Health Care System. Dr. Pointer is a founder and managing partner of Digestive and Liver Health Specialists. She is on staff as a clinical gastroenterologist at Tristar Hendersonville (Tenn.) Medical Center. They have no conflicts of interest.
One of the cornerstones of member engagement within the American Gastroenterological Association is its committees, which provide a platform for AGA members to network, effect change at the institutional level, and obtain leadership positions. For many within the AGA, exposure to these committees occurs during training. Both of us were first introduced to the possibility of serving on an AGA committee by faculty members at our institution. Each year, applications for available committee positions open in the fall and are due on Nov. 1. While you can be nominated by other members, self-nomination is common and encouraged. Truthfully, neither of us was quite certain what committee membership would entail. However, we both applied to several committees because we knew that it would be an excellent opportunity to network with leading gastroenterologists across the country and to have the ability to become involved in key AGA programs.
We were selected to serve 2-year terms as trainee members on the Government Affairs and Publication Committees, respectively, which gave us a deeper understanding of how an organization with both a full-time professional staff and group of volunteer members functions. A unique feature of the AGA is its Trainee and Early Career (TEC) Committee, which mainly comprises trainee members who serve on other committees. By virtue of our roles with the other committees, we also became full-fledged members of the TEC committee, which is dedicated to enhancing the experience for trainees and those who are within 5 years of graduation.
One of the most innovative programs developed by the TEC committee is Career Development Workshops, which is a webinar series focused on important topics not covered in fellowship, such as different career paths, personal finance, and how to increase the number of underrepresented individuals in the field. The predecessor of the Career Development Workshops was the in-person Regional Practice Skills Workshop, and we both took on the responsibility of planning and organizing separate workshops. For one of us (Stephanie), that involved enlisting our fellowship program to host the event. For the other (Peter), it meant collaborating with our local gastroenterology society to cosponsor the workshop. It was extremely rewarding to organize the workshops, which allowed us to work closely with AGA staff and local gastroenterology faculty, as well as our peers, to bring the events to fruition. For both of us, the success of the Regional Practice Skills Workshop was one of the highlights of our tenure on the TEC committee.
Although we were not aware of it at the time, volunteering to plan a workshop and assisting with other projects and subcommittees were signs of enthusiasm and leadership that the AGA recognized and valued. Our advice on becoming a committee chair is to not only show an interest in committee projects but also to turn that interest into action. A committee member who is strongly interested in a leadership position cannot expect to transition into that role by being a “silent but present” member. You need to do more than just show up. You should actively participate in projects, engage in discussions, and devote your time and energy to ensure the success of committee programs. However, you should also make sure to have sufficient bandwidth to make meaningful contributions to each project and not commit to tasks that you cannot complete. To set yourself apart on a committee, it is important to be actively engaged and committed to a project (or two) that allows for professional growth and visibility. Ideally, you will become an integral part of a committee that sparks your drive to serve.
Applying to become a committee chair follows the same process and timeline as for any other committee position, and you can be nominated or self-nominate. Although previous experience on that specific committee is not a prerequisite for most chair positions, having previously served on any AGA committee or task force is generally required. Successful applicants serve for 1 year as chair-elect, during which they work closely with the outgoing chair and staff to ensure a smooth transition when their 3-year term as chair officially begins in June.
Each committee has a guiding mission statement and a staff liaison who provides institutional knowledge and logistical support. However, the committee members, and especially the chair, have considerable latitude to develop and implement new initiatives or retire old ones. The entire committee meets twice per year, once in September in Washington, D.C., and once at DDW. Between the meetings, working groups are formed to move the various programs forward. In addition to the Career Development Workshops, the TEC committee organizes the Young Delegates program (which allows any AGA member to volunteer on small, time-limited projects), a symposium at DDW focused on trainee and early career issues, and a networking event at DDW. Moreover, we collaborate with other committees and provide input from the perspective of younger members on larger initiatives such as the AGA Equity Project and Career Compass.
As chair, we lead the twice-yearly meetings as well as the working groups. We strongly encourage all committee members to participate on at least one working group, which develops leadership skills and provides the opportunity to moderate sessions for the Career Development Workshops and DDW symposium. Moreover, we solicit feedback on ways to improve current programming, start new initiatives, and work with other committees that the TEC committee members are part of. Trainees and early career members are seen as a key constituency group within the AGA, and we take the responsibility of increasing the value of membership for this group seriously.
As early-career physicians ourselves, we also view the chance to serve as a committee chair as a great career development opportunity. It allows us to expand our professional networks, help shape an organization that is a leading voice and advocate for digestive health, and meet the needs of young members who are the future of the AGA.
There is no doubt that all of you have achieved amazing things on the way to becoming a trainee or early career professional in the competitive fields of gastroenterology and hepatology. The AGA is constantly looking for bright, motivated individuals to serve as volunteers and future leaders. Our experience shows that with a bit of persistence to get in the door – through Young Delegates or a committee – along with lots of hard work along the way, you will be in a great position to rise through the ranks and help lead an organization at the vanguard of our field.
Dr. Liang is assistant professor of medicine and population health, New York University Langone Health, and a staff physician at VA New York Harbor Health Care System. Dr. Pointer is a founder and managing partner of Digestive and Liver Health Specialists. She is on staff as a clinical gastroenterologist at Tristar Hendersonville (Tenn.) Medical Center. They have no conflicts of interest.
Guideline advises against depression screening in pregnancy
The Canadian Task Force on Preventive Health Care recommends against the routine screening of all pregnant and postpartum women for depression using a standard questionnaire, according to its new guideline.
The basis for its position is the lack of evidence that such screening “adds value beyond discussions about overall wellbeing, depression, anxiety, and mood that are currently a part of established perinatal clinical care.
“We should not be using a one-size-fits all approach,” lead author Eddy Lang, MD, professor and head of emergency medicine at the Cumming School of Medicine, University of Calgary (Alta.), told this news organization.
Instead, the task force emphasizes regular clinical care, including asking patients about their wellbeing and support systems. The task force categorizes the recommendation as conditional and as having very low-certainty evidence.
The recommendation was published in CMAJ.
One randomized study
The task force is an independent panel of clinicians and scientists that makes recommendations on primary and secondary prevention in primary care. A working group of five members of the task force developed this recommendation with scientific support from Public Health Agency of Canada staff.
In its research, the task force found only one study that showed a benefit of routine depression screening in this population. This study was a randomized controlled trial conducted in Hong Kong. Researchers evaluated 462 postpartum women who were randomly assigned to receive screening with the Edinburgh Postnatal Depression Scale (EPDS) or no screening 2 months post partum.
“We found the effect of screening in this study to be very uncertain for the important outcomes of interest,” said Dr. Lang.
“These included parent-child stress, marital stress, and the number of infant hospital admissions. The effects of screening on all of these outcomes were very uncertain, mainly because it was such a small trial,” he said.
The task force also assessed how pregnant and postpartum women feel about being screened. What these women most wanted was a good relationship with a trusted primary care provider who would initiate discussions about their mental health in a caring atmosphere.
“Although they told us they liked the idea of universal screening, they admitted to their family doctors that they actually preferred to be asked about their wellbeing, [to be asked] how things were going at home, and [to have] a discussion about their mental health and wellbeing, rather than a formal screening process. They felt a discussion about depression with a primary health care provider during the pregnancy and postpartum period is critical,” said Dr. Lang.
Thus, the task force recommends “against instrument-based depression screening using a questionnaire with cutoff score to distinguish ‘screen positive’ and ‘screen negative’ administered to all individuals during pregnancy and the postpartum period (up to 1 year after childbirth).”
Screening remains common
“There’s a lot of uncertainty in the scientific community about whether it’s a good idea to administer a screening test to all pregnant and postpartum women to determine in a systematic way if they might be suffering from depression,” said Dr. Lang.
The task force recommended against screening for depression among perinatal or postpartum women in 2013, but screening is still performed in many provinces, said Dr. Lang.
Dr. Lang emphasized that the recommendation does not apply to usual care, in which the provider asks questions about and discusses a patient’s mental health and proceeds on the basis of their clinical judgment; nor does it apply to diagnostic pathways in which the clinician suspects that the individual may have depression and tests her accordingly.
“What we are saying in our recommendation is that all clinicians should ask about a patient’s wellbeing, about their mood, their anxiety, and these questions are an important part of the clinical assessment of pregnant and postpartum women. But we’re also saying the usefulness of doing so with a questionnaire and using a cutoff score on the questionnaire to decide who needs further assessment or possibly treatment is unproven by the research,” Dr. Lang said.
A growing problem
For Diane Francoeur, MD, CEO of the Society of Obstetricians and Gynecologists of Canada, this is all well and good, but the reality is that such screening is better than nothing.
Quebec is the only Canadian province that conducts universal screening for all pregnant and postpartum women, Dr. Francoeur said in an interview. She was not part of the task force.
“I agree that it should be more than one approach, but the problem is that there is such a shortage of resources. There are many issues that can arise when you follow a woman during her pregnancy,” she said.
Dr. Francoeur said that COVID-19 has been particularly tough on women, including pregnant and postpartum women, who are the most vulnerable.
“Especially during the COVID era, it was astonishing how women were not doing well. Their stress level was so high. We need to have a specific approach dedicated to prenatal mental health, because it’s a problem that is bigger than it used to be,” she said.
Violence against women has increased considerably since the beginning of the COVID-19 pandemic, said Dr. Francoeur. “Many more women have been killed by their partners. We have never seen anything like this before, and I hope we will never see this again,” she said.
“Help was more available a few years ago, but now, it’s really hard if and when you need to have a quick consultation with a specialist and the woman is really depressed. It can take forever. So, it’s okay to screen, but then, what’s next? Who is going to be there to take these women and help them? And we don’t have the answer,” Dr. Francoeur said.
Pregnant and postpartum women who suffer from depression need more than pills, she added. “We reassure them and treat their depression pharmacologically, but it’s also a time to give appropriate support and help them through the pregnancy and get well prepared to receive their newborn, because, as we now know, that first year of life is really important for the child, and the mom needs to be supported.”
Funding for the Canadian Task Force on Preventive Health Care is provided by the Public Health Agency of Canada. Dr. Lang and Dr. Francoeur reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Canadian Task Force on Preventive Health Care recommends against the routine screening of all pregnant and postpartum women for depression using a standard questionnaire, according to its new guideline.
The basis for its position is the lack of evidence that such screening “adds value beyond discussions about overall wellbeing, depression, anxiety, and mood that are currently a part of established perinatal clinical care.
“We should not be using a one-size-fits all approach,” lead author Eddy Lang, MD, professor and head of emergency medicine at the Cumming School of Medicine, University of Calgary (Alta.), told this news organization.
Instead, the task force emphasizes regular clinical care, including asking patients about their wellbeing and support systems. The task force categorizes the recommendation as conditional and as having very low-certainty evidence.
The recommendation was published in CMAJ.
One randomized study
The task force is an independent panel of clinicians and scientists that makes recommendations on primary and secondary prevention in primary care. A working group of five members of the task force developed this recommendation with scientific support from Public Health Agency of Canada staff.
In its research, the task force found only one study that showed a benefit of routine depression screening in this population. This study was a randomized controlled trial conducted in Hong Kong. Researchers evaluated 462 postpartum women who were randomly assigned to receive screening with the Edinburgh Postnatal Depression Scale (EPDS) or no screening 2 months post partum.
“We found the effect of screening in this study to be very uncertain for the important outcomes of interest,” said Dr. Lang.
“These included parent-child stress, marital stress, and the number of infant hospital admissions. The effects of screening on all of these outcomes were very uncertain, mainly because it was such a small trial,” he said.
The task force also assessed how pregnant and postpartum women feel about being screened. What these women most wanted was a good relationship with a trusted primary care provider who would initiate discussions about their mental health in a caring atmosphere.
“Although they told us they liked the idea of universal screening, they admitted to their family doctors that they actually preferred to be asked about their wellbeing, [to be asked] how things were going at home, and [to have] a discussion about their mental health and wellbeing, rather than a formal screening process. They felt a discussion about depression with a primary health care provider during the pregnancy and postpartum period is critical,” said Dr. Lang.
Thus, the task force recommends “against instrument-based depression screening using a questionnaire with cutoff score to distinguish ‘screen positive’ and ‘screen negative’ administered to all individuals during pregnancy and the postpartum period (up to 1 year after childbirth).”
Screening remains common
“There’s a lot of uncertainty in the scientific community about whether it’s a good idea to administer a screening test to all pregnant and postpartum women to determine in a systematic way if they might be suffering from depression,” said Dr. Lang.
The task force recommended against screening for depression among perinatal or postpartum women in 2013, but screening is still performed in many provinces, said Dr. Lang.
Dr. Lang emphasized that the recommendation does not apply to usual care, in which the provider asks questions about and discusses a patient’s mental health and proceeds on the basis of their clinical judgment; nor does it apply to diagnostic pathways in which the clinician suspects that the individual may have depression and tests her accordingly.
“What we are saying in our recommendation is that all clinicians should ask about a patient’s wellbeing, about their mood, their anxiety, and these questions are an important part of the clinical assessment of pregnant and postpartum women. But we’re also saying the usefulness of doing so with a questionnaire and using a cutoff score on the questionnaire to decide who needs further assessment or possibly treatment is unproven by the research,” Dr. Lang said.
A growing problem
For Diane Francoeur, MD, CEO of the Society of Obstetricians and Gynecologists of Canada, this is all well and good, but the reality is that such screening is better than nothing.
Quebec is the only Canadian province that conducts universal screening for all pregnant and postpartum women, Dr. Francoeur said in an interview. She was not part of the task force.
“I agree that it should be more than one approach, but the problem is that there is such a shortage of resources. There are many issues that can arise when you follow a woman during her pregnancy,” she said.
Dr. Francoeur said that COVID-19 has been particularly tough on women, including pregnant and postpartum women, who are the most vulnerable.
“Especially during the COVID era, it was astonishing how women were not doing well. Their stress level was so high. We need to have a specific approach dedicated to prenatal mental health, because it’s a problem that is bigger than it used to be,” she said.
Violence against women has increased considerably since the beginning of the COVID-19 pandemic, said Dr. Francoeur. “Many more women have been killed by their partners. We have never seen anything like this before, and I hope we will never see this again,” she said.
“Help was more available a few years ago, but now, it’s really hard if and when you need to have a quick consultation with a specialist and the woman is really depressed. It can take forever. So, it’s okay to screen, but then, what’s next? Who is going to be there to take these women and help them? And we don’t have the answer,” Dr. Francoeur said.
Pregnant and postpartum women who suffer from depression need more than pills, she added. “We reassure them and treat their depression pharmacologically, but it’s also a time to give appropriate support and help them through the pregnancy and get well prepared to receive their newborn, because, as we now know, that first year of life is really important for the child, and the mom needs to be supported.”
Funding for the Canadian Task Force on Preventive Health Care is provided by the Public Health Agency of Canada. Dr. Lang and Dr. Francoeur reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The Canadian Task Force on Preventive Health Care recommends against the routine screening of all pregnant and postpartum women for depression using a standard questionnaire, according to its new guideline.
The basis for its position is the lack of evidence that such screening “adds value beyond discussions about overall wellbeing, depression, anxiety, and mood that are currently a part of established perinatal clinical care.
“We should not be using a one-size-fits all approach,” lead author Eddy Lang, MD, professor and head of emergency medicine at the Cumming School of Medicine, University of Calgary (Alta.), told this news organization.
Instead, the task force emphasizes regular clinical care, including asking patients about their wellbeing and support systems. The task force categorizes the recommendation as conditional and as having very low-certainty evidence.
The recommendation was published in CMAJ.
One randomized study
The task force is an independent panel of clinicians and scientists that makes recommendations on primary and secondary prevention in primary care. A working group of five members of the task force developed this recommendation with scientific support from Public Health Agency of Canada staff.
In its research, the task force found only one study that showed a benefit of routine depression screening in this population. This study was a randomized controlled trial conducted in Hong Kong. Researchers evaluated 462 postpartum women who were randomly assigned to receive screening with the Edinburgh Postnatal Depression Scale (EPDS) or no screening 2 months post partum.
“We found the effect of screening in this study to be very uncertain for the important outcomes of interest,” said Dr. Lang.
“These included parent-child stress, marital stress, and the number of infant hospital admissions. The effects of screening on all of these outcomes were very uncertain, mainly because it was such a small trial,” he said.
The task force also assessed how pregnant and postpartum women feel about being screened. What these women most wanted was a good relationship with a trusted primary care provider who would initiate discussions about their mental health in a caring atmosphere.
“Although they told us they liked the idea of universal screening, they admitted to their family doctors that they actually preferred to be asked about their wellbeing, [to be asked] how things were going at home, and [to have] a discussion about their mental health and wellbeing, rather than a formal screening process. They felt a discussion about depression with a primary health care provider during the pregnancy and postpartum period is critical,” said Dr. Lang.
Thus, the task force recommends “against instrument-based depression screening using a questionnaire with cutoff score to distinguish ‘screen positive’ and ‘screen negative’ administered to all individuals during pregnancy and the postpartum period (up to 1 year after childbirth).”
Screening remains common
“There’s a lot of uncertainty in the scientific community about whether it’s a good idea to administer a screening test to all pregnant and postpartum women to determine in a systematic way if they might be suffering from depression,” said Dr. Lang.
The task force recommended against screening for depression among perinatal or postpartum women in 2013, but screening is still performed in many provinces, said Dr. Lang.
Dr. Lang emphasized that the recommendation does not apply to usual care, in which the provider asks questions about and discusses a patient’s mental health and proceeds on the basis of their clinical judgment; nor does it apply to diagnostic pathways in which the clinician suspects that the individual may have depression and tests her accordingly.
“What we are saying in our recommendation is that all clinicians should ask about a patient’s wellbeing, about their mood, their anxiety, and these questions are an important part of the clinical assessment of pregnant and postpartum women. But we’re also saying the usefulness of doing so with a questionnaire and using a cutoff score on the questionnaire to decide who needs further assessment or possibly treatment is unproven by the research,” Dr. Lang said.
A growing problem
For Diane Francoeur, MD, CEO of the Society of Obstetricians and Gynecologists of Canada, this is all well and good, but the reality is that such screening is better than nothing.
Quebec is the only Canadian province that conducts universal screening for all pregnant and postpartum women, Dr. Francoeur said in an interview. She was not part of the task force.
“I agree that it should be more than one approach, but the problem is that there is such a shortage of resources. There are many issues that can arise when you follow a woman during her pregnancy,” she said.
Dr. Francoeur said that COVID-19 has been particularly tough on women, including pregnant and postpartum women, who are the most vulnerable.
“Especially during the COVID era, it was astonishing how women were not doing well. Their stress level was so high. We need to have a specific approach dedicated to prenatal mental health, because it’s a problem that is bigger than it used to be,” she said.
Violence against women has increased considerably since the beginning of the COVID-19 pandemic, said Dr. Francoeur. “Many more women have been killed by their partners. We have never seen anything like this before, and I hope we will never see this again,” she said.
“Help was more available a few years ago, but now, it’s really hard if and when you need to have a quick consultation with a specialist and the woman is really depressed. It can take forever. So, it’s okay to screen, but then, what’s next? Who is going to be there to take these women and help them? And we don’t have the answer,” Dr. Francoeur said.
Pregnant and postpartum women who suffer from depression need more than pills, she added. “We reassure them and treat their depression pharmacologically, but it’s also a time to give appropriate support and help them through the pregnancy and get well prepared to receive their newborn, because, as we now know, that first year of life is really important for the child, and the mom needs to be supported.”
Funding for the Canadian Task Force on Preventive Health Care is provided by the Public Health Agency of Canada. Dr. Lang and Dr. Francoeur reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CMAJ
Life and death decisions: What keeps oncologists up at night
It was 2 a.m. And Rebecca Shatsky, MD, could not sleep.
The breast oncologist was thinking about a patient of hers with metastatic cancer.
The patient’s disease had been asymptomatic for some time. Then without warning, her cancer suddenly exploded. Her bone marrow was failing, and her liver was not far behind.
Dr. Shatsky had a treatment plan ready to go but still, she felt uneasy.
“I had to be honest with her that I didn’t know if this plan would work,” says Dr. Shatsky, a medical oncologist at University of California, San Diego (UCSD).
That night, after visiting the patient in the hospital, Dr. Shatsky lay awake going over her next move, making sure it was the right one and hoping it would help keep the disease at bay.
“It’s so much pressure when someone is depending on you to make life or death decisions,” Dr. Shatsky said.
And in the quiet hours of night, these concerns grow louder.
Dr. Shatsky is not alone.
“There’s no off button,” says Aaron Goodman, MD, a hematologist at UCSD Health who goes by “Papa Heme” on Twitter. “I’m always thinking about my patients. Constantly.”
The public rarely gets a glimpse of these private moments. On occasion, oncologists will share a personal story, but more often, insights come from broad research on the ethical, emotional, and psychological toll of practicing medicine.
Many oncologists carry this baggage home with them because they have no other option.
“There is simply no time to process the weight of the day when I’ve got seven more patients who need my full attention before lunch,” Mark Lewis, MD, director, department of gastrointestinal oncology, Intermountain Healthcare, Salt Lake City, Utah. “That is why my processing happens outside of the office, when my brain can be quiet.”
What am I missing?
Dr. Goodman recognizes the gravity of each decision he makes. He pores over every detail of a patient’s scans, lab results, history, and symptoms.
But no matter how many times he checks and rechecks, one question nags at him: What am I missing?
For Dr. Goodman, this exhaustive level of attention is worth it.
“When errors are made, it’s someone’s life,” Dr. Goodman said. “Nothing would have prepared me for this responsibility. Until it lies on you, it’s impossible to understand how much trust patients put into us.”
That trust becomes most apparent for Dr. Goodman when facing a decision about how to treat a patient with acute myeloid leukemia who’s in remission.
Give more chemotherapy to root out the leukemia cells still lurking in the body, and the patient faces a high risk of the cancer returning. Pick stem cell transplant, and the chance of being cured goes up significantly, but the patient could also die within 100 days of the transplant.
“All together, the data show I’m helping patients with a transplant, but for the individual, I could be causing harm. Someone could be living less because of a decision I made,” Dr. Goodman said.
For patients with advanced cancer, oncologists may need to think several moves ahead. Mapping out a patient’s treatment options can feel like a game of chess. Dr. Shatsky is always trying to anticipate how the tumor will behave, what is driving it, and how lifestyle factors may influence a patient’s response in the present and the future.
“It is a mind game,” she says. “Like in chess, I try to outsmart my opponent. But with advanced cancer, there are not necessarily clear-cut guidelines or one way to manage the disease, and I have to do the best I can with drugs I have.”
That’s the art of oncology: Balancing the many knowns and unknowns of a person’s cancer alongside the toxicities of treatment and a patient’s hopes and goals.
Throughout the year, Don Dizon, MD, will see a number of patients with advanced disease. In these instances, the question he often wrestles with is if the patient can’t be cured, whether more treatment will just cause greater harm.
Dr. Dizon recently faced this dilemma with an older patient with metastatic disease who had not done well with an initial treatment regimen. After outlining the risks for more chemotherapy, he explained one option would be to forgo it and simply treat her symptoms.
“It’s an impossible choice,” says Dr. Dizon, director of women’s cancers at Lifespan Cancer Institute and director of medical oncology at Rhode Island Hospital, Providence.
Chemotherapy can provide symptom relief, but it can also be toxic – and patients may be so frail, they can die from more therapy.
“I told my patient, if in your heart, you want to try more therapy, that’s okay. But it’s also okay if you don’t,” Dr. Dizon recalled.
Her response: “You’re supposed to give me the answer.”
However, for patients approaching the end of life, there often is no right answer.
“It’s part of the discomfort you live with as a patient and oncologist, and when I leave the clinic, that’s one thing that follows me home,” Dr. Dizon said. “At the end of the day, I need to look in the mirror and know I did the best I could.”
The difficult conversation
Every Sunday, Dr. Lewis feels the weight of the week ahead. He and his wife, a pediatrician, call it the “Sunday scaries.”
It’s when Dr. Lewis begins thinking about the delicate conversations to come, rehearsing how he’s going to share the news that a person has advanced cancer or that a cancer, once in remission, has returned.
“Before the pandemic, I had 36 people come to a visit where I delivered some very heavy news and it became a Greek chorus of sobbing,” he recalls.
For every oncologist, delivering bad news is an integral part of the job. But after spending months, sometimes years, with a patient and the family, Dr. Lewis knows how to take the temperature of the room – who will likely prefer a more blunt style and who might need a gentler touch.
“The longer you know a patient and family, the better you can gauge the best approach,” Dr. Lewis said. “And for some, you know it’ll be complete devastation no matter what.”
When Jennifer Lycette, MD, prepares for a difficult conversation, she’ll run down all the possible ways it could go. Sometimes her brain will get stuck in a loop, cycling through the different trajectories on repeat.
“For years, I didn’t know how to cope with that,” said Dr. Lycette, medical director at Providence Oncology and Hematology Care Clinic in Seaside, Ore. “I wasn’t taught the tools to cope with that in my medical training. It took midcareer professional coaching that I sought out on my own to learn to remind myself that no matter what the person says, I have the experience and skill set to handle what comes next and to simply be present in the moment with the patient.”
The question that now sits with Dr. Lycette hours after a visit is what she could have done better. She knows from experience how important it is to choose her words carefully.
Early in her career, Dr. Lycette had a patient with stage IV cancer who wanted to know more about the death process. Because most people ask about pain, she assured him that he likely wouldn’t experience too much pain with his type of cancer.
“It will probably be like falling asleep,” said Dr. Lycette, hoping she was offering comfort. “When I saw him next, he told me he hadn’t slept.”
He was afraid that if he did, he wouldn’t wake up.
In that moment, Dr. Lycette realized the power that her words carry and the importance of trying to understand the inner lives of her patients.
Life outside the clinic
Sometimes an oncologist’s late-night ruminations have little to do with cancer itself.
Manali Patel, MD, finds herself worrying if her patients will have enough to eat and whether she will be able to help.
“I was up at 3 a.m. one morning, thinking about how we’re going to fund a project for patients from low-income households who we discovered were experiencing severe food insecurity – what grants we need, what foundations we can work with,” said Dr. Patel, a medical oncologist at Stanford Hospital and Clinics and the VA Palo Alto Health Care System in California.
The past few years of the pandemic have added a new layer of worry for Dr. Patel.
“I don’t want my patients to die from a preventable virus when they’ve already been through so much suffering,” Dr. Patel said.
This thought feeds worries about how her actions outside the clinic could unintentionally harm her patients. Should she go to a big medical conference? A family gathering? The grocery store?
“There are some places you can’t avoid, but these decisions have caused a lot of strife for me,” she said. “The health and safety of our patients – that’s in our wheelhouse – but so many of the policies are outside of our control.”
The inevitable losses and the wins
For patients with metastatic disease, eventually the treatment options will run out.
Dr. Shatsky likes to be up front with patients about that reality: “There will come a day when I will tell you there’s nothing more I can do, and you need to trust that I’m being honest with you and that’s the truth.”
For Dr. Goodman, the devastation that bad news brings patients and families is glaring. He knows there will be no more normalcy in their lives.
“I see a lot of suffering, but I know the suffering happens regardless of whether I see it or not,” Dr. Goodman said.
That’s why holding on to the victories can be so important. Dr. Goodman recalled a young patient who came to him with a 20-cm tumor and is now cured. “Had I not met that individual and done what I had done, he’d be dead, but now he’s going to live his life,” Dr. Goodman said. “But I don’t wake up at 2 a.m. thinking about that.”
Dr. Shatsky gets a lot of joy from the wins – the patients who do really well, the times when she can help a friend or colleagues – and those moments go a long way to outweigh the hurt, worry, and workload.
When dealing with so much gray, “the wins are important, knowing you can make a difference is important,” Dr. Dizon said.
And there’s a delicate balance.
“I think patients want an oncologist who cares and is genuinely invested in their outcomes but not someone who is so sad all the time,” Dr. Lewis said. “When I lose a patient, I still grieve each loss, but I can’t mourn every patient’s death like it’s a family member. Otherwise, I’d break.”
What would you do if you had terminal cancer?
Dr. Dizon recalled how a friend handled the news. She went home and made dinner, he said.
Ultimately, she lived for many years. She saw her kids get married, met her first grandchild, and had time to prepare, something not everyone gets the chance to do.
That’s why it’s important to “do what you normally do as long as you can,” Dr. Dizon said. “Live your life.”
A version of this article first appeared on Medscape.com.
It was 2 a.m. And Rebecca Shatsky, MD, could not sleep.
The breast oncologist was thinking about a patient of hers with metastatic cancer.
The patient’s disease had been asymptomatic for some time. Then without warning, her cancer suddenly exploded. Her bone marrow was failing, and her liver was not far behind.
Dr. Shatsky had a treatment plan ready to go but still, she felt uneasy.
“I had to be honest with her that I didn’t know if this plan would work,” says Dr. Shatsky, a medical oncologist at University of California, San Diego (UCSD).
That night, after visiting the patient in the hospital, Dr. Shatsky lay awake going over her next move, making sure it was the right one and hoping it would help keep the disease at bay.
“It’s so much pressure when someone is depending on you to make life or death decisions,” Dr. Shatsky said.
And in the quiet hours of night, these concerns grow louder.
Dr. Shatsky is not alone.
“There’s no off button,” says Aaron Goodman, MD, a hematologist at UCSD Health who goes by “Papa Heme” on Twitter. “I’m always thinking about my patients. Constantly.”
The public rarely gets a glimpse of these private moments. On occasion, oncologists will share a personal story, but more often, insights come from broad research on the ethical, emotional, and psychological toll of practicing medicine.
Many oncologists carry this baggage home with them because they have no other option.
“There is simply no time to process the weight of the day when I’ve got seven more patients who need my full attention before lunch,” Mark Lewis, MD, director, department of gastrointestinal oncology, Intermountain Healthcare, Salt Lake City, Utah. “That is why my processing happens outside of the office, when my brain can be quiet.”
What am I missing?
Dr. Goodman recognizes the gravity of each decision he makes. He pores over every detail of a patient’s scans, lab results, history, and symptoms.
But no matter how many times he checks and rechecks, one question nags at him: What am I missing?
For Dr. Goodman, this exhaustive level of attention is worth it.
“When errors are made, it’s someone’s life,” Dr. Goodman said. “Nothing would have prepared me for this responsibility. Until it lies on you, it’s impossible to understand how much trust patients put into us.”
That trust becomes most apparent for Dr. Goodman when facing a decision about how to treat a patient with acute myeloid leukemia who’s in remission.
Give more chemotherapy to root out the leukemia cells still lurking in the body, and the patient faces a high risk of the cancer returning. Pick stem cell transplant, and the chance of being cured goes up significantly, but the patient could also die within 100 days of the transplant.
“All together, the data show I’m helping patients with a transplant, but for the individual, I could be causing harm. Someone could be living less because of a decision I made,” Dr. Goodman said.
For patients with advanced cancer, oncologists may need to think several moves ahead. Mapping out a patient’s treatment options can feel like a game of chess. Dr. Shatsky is always trying to anticipate how the tumor will behave, what is driving it, and how lifestyle factors may influence a patient’s response in the present and the future.
“It is a mind game,” she says. “Like in chess, I try to outsmart my opponent. But with advanced cancer, there are not necessarily clear-cut guidelines or one way to manage the disease, and I have to do the best I can with drugs I have.”
That’s the art of oncology: Balancing the many knowns and unknowns of a person’s cancer alongside the toxicities of treatment and a patient’s hopes and goals.
Throughout the year, Don Dizon, MD, will see a number of patients with advanced disease. In these instances, the question he often wrestles with is if the patient can’t be cured, whether more treatment will just cause greater harm.
Dr. Dizon recently faced this dilemma with an older patient with metastatic disease who had not done well with an initial treatment regimen. After outlining the risks for more chemotherapy, he explained one option would be to forgo it and simply treat her symptoms.
“It’s an impossible choice,” says Dr. Dizon, director of women’s cancers at Lifespan Cancer Institute and director of medical oncology at Rhode Island Hospital, Providence.
Chemotherapy can provide symptom relief, but it can also be toxic – and patients may be so frail, they can die from more therapy.
“I told my patient, if in your heart, you want to try more therapy, that’s okay. But it’s also okay if you don’t,” Dr. Dizon recalled.
Her response: “You’re supposed to give me the answer.”
However, for patients approaching the end of life, there often is no right answer.
“It’s part of the discomfort you live with as a patient and oncologist, and when I leave the clinic, that’s one thing that follows me home,” Dr. Dizon said. “At the end of the day, I need to look in the mirror and know I did the best I could.”
The difficult conversation
Every Sunday, Dr. Lewis feels the weight of the week ahead. He and his wife, a pediatrician, call it the “Sunday scaries.”
It’s when Dr. Lewis begins thinking about the delicate conversations to come, rehearsing how he’s going to share the news that a person has advanced cancer or that a cancer, once in remission, has returned.
“Before the pandemic, I had 36 people come to a visit where I delivered some very heavy news and it became a Greek chorus of sobbing,” he recalls.
For every oncologist, delivering bad news is an integral part of the job. But after spending months, sometimes years, with a patient and the family, Dr. Lewis knows how to take the temperature of the room – who will likely prefer a more blunt style and who might need a gentler touch.
“The longer you know a patient and family, the better you can gauge the best approach,” Dr. Lewis said. “And for some, you know it’ll be complete devastation no matter what.”
When Jennifer Lycette, MD, prepares for a difficult conversation, she’ll run down all the possible ways it could go. Sometimes her brain will get stuck in a loop, cycling through the different trajectories on repeat.
“For years, I didn’t know how to cope with that,” said Dr. Lycette, medical director at Providence Oncology and Hematology Care Clinic in Seaside, Ore. “I wasn’t taught the tools to cope with that in my medical training. It took midcareer professional coaching that I sought out on my own to learn to remind myself that no matter what the person says, I have the experience and skill set to handle what comes next and to simply be present in the moment with the patient.”
The question that now sits with Dr. Lycette hours after a visit is what she could have done better. She knows from experience how important it is to choose her words carefully.
Early in her career, Dr. Lycette had a patient with stage IV cancer who wanted to know more about the death process. Because most people ask about pain, she assured him that he likely wouldn’t experience too much pain with his type of cancer.
“It will probably be like falling asleep,” said Dr. Lycette, hoping she was offering comfort. “When I saw him next, he told me he hadn’t slept.”
He was afraid that if he did, he wouldn’t wake up.
In that moment, Dr. Lycette realized the power that her words carry and the importance of trying to understand the inner lives of her patients.
Life outside the clinic
Sometimes an oncologist’s late-night ruminations have little to do with cancer itself.
Manali Patel, MD, finds herself worrying if her patients will have enough to eat and whether she will be able to help.
“I was up at 3 a.m. one morning, thinking about how we’re going to fund a project for patients from low-income households who we discovered were experiencing severe food insecurity – what grants we need, what foundations we can work with,” said Dr. Patel, a medical oncologist at Stanford Hospital and Clinics and the VA Palo Alto Health Care System in California.
The past few years of the pandemic have added a new layer of worry for Dr. Patel.
“I don’t want my patients to die from a preventable virus when they’ve already been through so much suffering,” Dr. Patel said.
This thought feeds worries about how her actions outside the clinic could unintentionally harm her patients. Should she go to a big medical conference? A family gathering? The grocery store?
“There are some places you can’t avoid, but these decisions have caused a lot of strife for me,” she said. “The health and safety of our patients – that’s in our wheelhouse – but so many of the policies are outside of our control.”
The inevitable losses and the wins
For patients with metastatic disease, eventually the treatment options will run out.
Dr. Shatsky likes to be up front with patients about that reality: “There will come a day when I will tell you there’s nothing more I can do, and you need to trust that I’m being honest with you and that’s the truth.”
For Dr. Goodman, the devastation that bad news brings patients and families is glaring. He knows there will be no more normalcy in their lives.
“I see a lot of suffering, but I know the suffering happens regardless of whether I see it or not,” Dr. Goodman said.
That’s why holding on to the victories can be so important. Dr. Goodman recalled a young patient who came to him with a 20-cm tumor and is now cured. “Had I not met that individual and done what I had done, he’d be dead, but now he’s going to live his life,” Dr. Goodman said. “But I don’t wake up at 2 a.m. thinking about that.”
Dr. Shatsky gets a lot of joy from the wins – the patients who do really well, the times when she can help a friend or colleagues – and those moments go a long way to outweigh the hurt, worry, and workload.
When dealing with so much gray, “the wins are important, knowing you can make a difference is important,” Dr. Dizon said.
And there’s a delicate balance.
“I think patients want an oncologist who cares and is genuinely invested in their outcomes but not someone who is so sad all the time,” Dr. Lewis said. “When I lose a patient, I still grieve each loss, but I can’t mourn every patient’s death like it’s a family member. Otherwise, I’d break.”
What would you do if you had terminal cancer?
Dr. Dizon recalled how a friend handled the news. She went home and made dinner, he said.
Ultimately, she lived for many years. She saw her kids get married, met her first grandchild, and had time to prepare, something not everyone gets the chance to do.
That’s why it’s important to “do what you normally do as long as you can,” Dr. Dizon said. “Live your life.”
A version of this article first appeared on Medscape.com.
It was 2 a.m. And Rebecca Shatsky, MD, could not sleep.
The breast oncologist was thinking about a patient of hers with metastatic cancer.
The patient’s disease had been asymptomatic for some time. Then without warning, her cancer suddenly exploded. Her bone marrow was failing, and her liver was not far behind.
Dr. Shatsky had a treatment plan ready to go but still, she felt uneasy.
“I had to be honest with her that I didn’t know if this plan would work,” says Dr. Shatsky, a medical oncologist at University of California, San Diego (UCSD).
That night, after visiting the patient in the hospital, Dr. Shatsky lay awake going over her next move, making sure it was the right one and hoping it would help keep the disease at bay.
“It’s so much pressure when someone is depending on you to make life or death decisions,” Dr. Shatsky said.
And in the quiet hours of night, these concerns grow louder.
Dr. Shatsky is not alone.
“There’s no off button,” says Aaron Goodman, MD, a hematologist at UCSD Health who goes by “Papa Heme” on Twitter. “I’m always thinking about my patients. Constantly.”
The public rarely gets a glimpse of these private moments. On occasion, oncologists will share a personal story, but more often, insights come from broad research on the ethical, emotional, and psychological toll of practicing medicine.
Many oncologists carry this baggage home with them because they have no other option.
“There is simply no time to process the weight of the day when I’ve got seven more patients who need my full attention before lunch,” Mark Lewis, MD, director, department of gastrointestinal oncology, Intermountain Healthcare, Salt Lake City, Utah. “That is why my processing happens outside of the office, when my brain can be quiet.”
What am I missing?
Dr. Goodman recognizes the gravity of each decision he makes. He pores over every detail of a patient’s scans, lab results, history, and symptoms.
But no matter how many times he checks and rechecks, one question nags at him: What am I missing?
For Dr. Goodman, this exhaustive level of attention is worth it.
“When errors are made, it’s someone’s life,” Dr. Goodman said. “Nothing would have prepared me for this responsibility. Until it lies on you, it’s impossible to understand how much trust patients put into us.”
That trust becomes most apparent for Dr. Goodman when facing a decision about how to treat a patient with acute myeloid leukemia who’s in remission.
Give more chemotherapy to root out the leukemia cells still lurking in the body, and the patient faces a high risk of the cancer returning. Pick stem cell transplant, and the chance of being cured goes up significantly, but the patient could also die within 100 days of the transplant.
“All together, the data show I’m helping patients with a transplant, but for the individual, I could be causing harm. Someone could be living less because of a decision I made,” Dr. Goodman said.
For patients with advanced cancer, oncologists may need to think several moves ahead. Mapping out a patient’s treatment options can feel like a game of chess. Dr. Shatsky is always trying to anticipate how the tumor will behave, what is driving it, and how lifestyle factors may influence a patient’s response in the present and the future.
“It is a mind game,” she says. “Like in chess, I try to outsmart my opponent. But with advanced cancer, there are not necessarily clear-cut guidelines or one way to manage the disease, and I have to do the best I can with drugs I have.”
That’s the art of oncology: Balancing the many knowns and unknowns of a person’s cancer alongside the toxicities of treatment and a patient’s hopes and goals.
Throughout the year, Don Dizon, MD, will see a number of patients with advanced disease. In these instances, the question he often wrestles with is if the patient can’t be cured, whether more treatment will just cause greater harm.
Dr. Dizon recently faced this dilemma with an older patient with metastatic disease who had not done well with an initial treatment regimen. After outlining the risks for more chemotherapy, he explained one option would be to forgo it and simply treat her symptoms.
“It’s an impossible choice,” says Dr. Dizon, director of women’s cancers at Lifespan Cancer Institute and director of medical oncology at Rhode Island Hospital, Providence.
Chemotherapy can provide symptom relief, but it can also be toxic – and patients may be so frail, they can die from more therapy.
“I told my patient, if in your heart, you want to try more therapy, that’s okay. But it’s also okay if you don’t,” Dr. Dizon recalled.
Her response: “You’re supposed to give me the answer.”
However, for patients approaching the end of life, there often is no right answer.
“It’s part of the discomfort you live with as a patient and oncologist, and when I leave the clinic, that’s one thing that follows me home,” Dr. Dizon said. “At the end of the day, I need to look in the mirror and know I did the best I could.”
The difficult conversation
Every Sunday, Dr. Lewis feels the weight of the week ahead. He and his wife, a pediatrician, call it the “Sunday scaries.”
It’s when Dr. Lewis begins thinking about the delicate conversations to come, rehearsing how he’s going to share the news that a person has advanced cancer or that a cancer, once in remission, has returned.
“Before the pandemic, I had 36 people come to a visit where I delivered some very heavy news and it became a Greek chorus of sobbing,” he recalls.
For every oncologist, delivering bad news is an integral part of the job. But after spending months, sometimes years, with a patient and the family, Dr. Lewis knows how to take the temperature of the room – who will likely prefer a more blunt style and who might need a gentler touch.
“The longer you know a patient and family, the better you can gauge the best approach,” Dr. Lewis said. “And for some, you know it’ll be complete devastation no matter what.”
When Jennifer Lycette, MD, prepares for a difficult conversation, she’ll run down all the possible ways it could go. Sometimes her brain will get stuck in a loop, cycling through the different trajectories on repeat.
“For years, I didn’t know how to cope with that,” said Dr. Lycette, medical director at Providence Oncology and Hematology Care Clinic in Seaside, Ore. “I wasn’t taught the tools to cope with that in my medical training. It took midcareer professional coaching that I sought out on my own to learn to remind myself that no matter what the person says, I have the experience and skill set to handle what comes next and to simply be present in the moment with the patient.”
The question that now sits with Dr. Lycette hours after a visit is what she could have done better. She knows from experience how important it is to choose her words carefully.
Early in her career, Dr. Lycette had a patient with stage IV cancer who wanted to know more about the death process. Because most people ask about pain, she assured him that he likely wouldn’t experience too much pain with his type of cancer.
“It will probably be like falling asleep,” said Dr. Lycette, hoping she was offering comfort. “When I saw him next, he told me he hadn’t slept.”
He was afraid that if he did, he wouldn’t wake up.
In that moment, Dr. Lycette realized the power that her words carry and the importance of trying to understand the inner lives of her patients.
Life outside the clinic
Sometimes an oncologist’s late-night ruminations have little to do with cancer itself.
Manali Patel, MD, finds herself worrying if her patients will have enough to eat and whether she will be able to help.
“I was up at 3 a.m. one morning, thinking about how we’re going to fund a project for patients from low-income households who we discovered were experiencing severe food insecurity – what grants we need, what foundations we can work with,” said Dr. Patel, a medical oncologist at Stanford Hospital and Clinics and the VA Palo Alto Health Care System in California.
The past few years of the pandemic have added a new layer of worry for Dr. Patel.
“I don’t want my patients to die from a preventable virus when they’ve already been through so much suffering,” Dr. Patel said.
This thought feeds worries about how her actions outside the clinic could unintentionally harm her patients. Should she go to a big medical conference? A family gathering? The grocery store?
“There are some places you can’t avoid, but these decisions have caused a lot of strife for me,” she said. “The health and safety of our patients – that’s in our wheelhouse – but so many of the policies are outside of our control.”
The inevitable losses and the wins
For patients with metastatic disease, eventually the treatment options will run out.
Dr. Shatsky likes to be up front with patients about that reality: “There will come a day when I will tell you there’s nothing more I can do, and you need to trust that I’m being honest with you and that’s the truth.”
For Dr. Goodman, the devastation that bad news brings patients and families is glaring. He knows there will be no more normalcy in their lives.
“I see a lot of suffering, but I know the suffering happens regardless of whether I see it or not,” Dr. Goodman said.
That’s why holding on to the victories can be so important. Dr. Goodman recalled a young patient who came to him with a 20-cm tumor and is now cured. “Had I not met that individual and done what I had done, he’d be dead, but now he’s going to live his life,” Dr. Goodman said. “But I don’t wake up at 2 a.m. thinking about that.”
Dr. Shatsky gets a lot of joy from the wins – the patients who do really well, the times when she can help a friend or colleagues – and those moments go a long way to outweigh the hurt, worry, and workload.
When dealing with so much gray, “the wins are important, knowing you can make a difference is important,” Dr. Dizon said.
And there’s a delicate balance.
“I think patients want an oncologist who cares and is genuinely invested in their outcomes but not someone who is so sad all the time,” Dr. Lewis said. “When I lose a patient, I still grieve each loss, but I can’t mourn every patient’s death like it’s a family member. Otherwise, I’d break.”
What would you do if you had terminal cancer?
Dr. Dizon recalled how a friend handled the news. She went home and made dinner, he said.
Ultimately, she lived for many years. She saw her kids get married, met her first grandchild, and had time to prepare, something not everyone gets the chance to do.
That’s why it’s important to “do what you normally do as long as you can,” Dr. Dizon said. “Live your life.”
A version of this article first appeared on Medscape.com.
Rituximab for Acquired Hemophilia A in the Setting of Bullous Pemphigoid
To the Editor:
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by the formation of antihemidesmosomal antibodies, resulting in tense bullae concentrated on the extremities and trunk that often are preceded by a pruritic urticarial phase.1 A rare complication of BP is the subsequent development of acquired hemophilia A. We report a case of BP with associated factor VIII–neutralizing antibodies in a patient who improved with prednisone and rituximab therapy.
A 78-year-old woman presented with red-orange pruritic plaques on the right heel that spread to involve the arms and legs, abdomen, and trunk with new-onset bullae over the course of 2 weeks (Figure 1). Dermatology was consulted, and a diagnosis of BP was confirmed via biopsy and direct immunofluorescence.
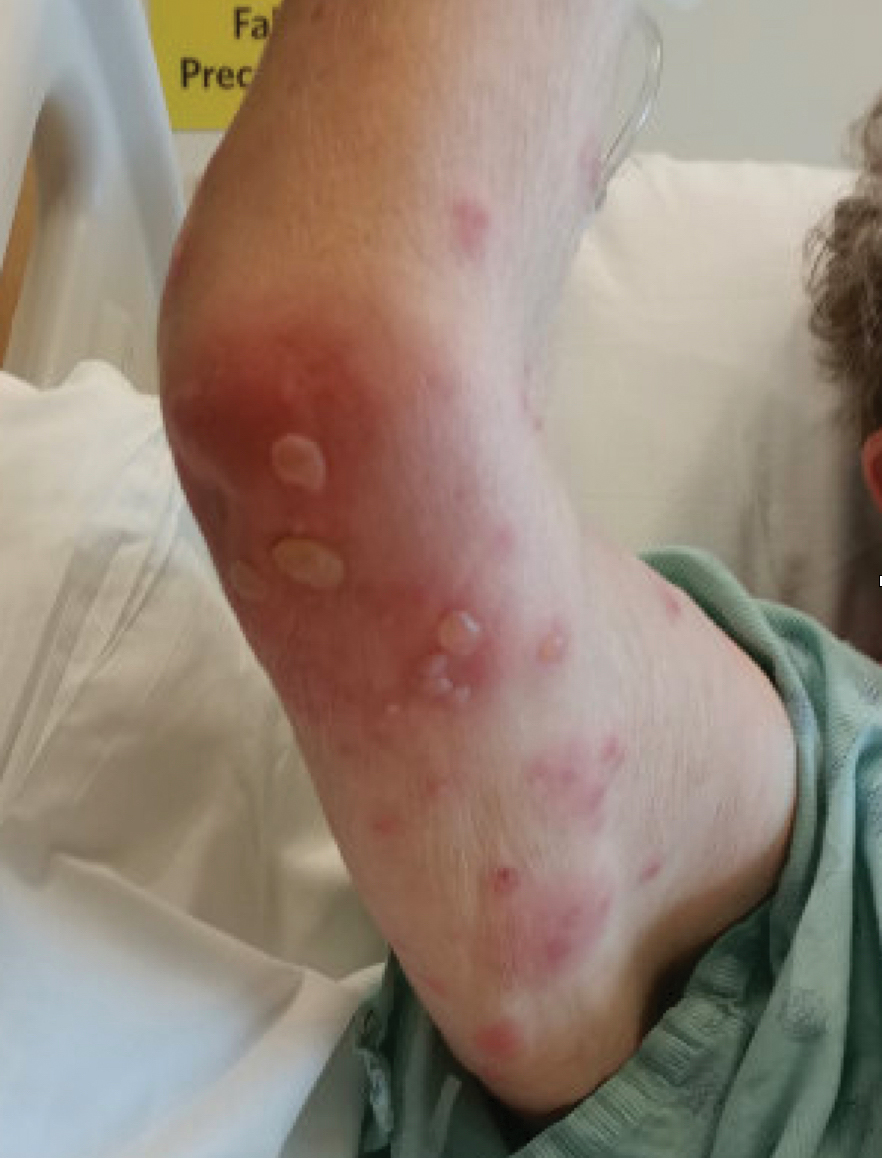
Despite treatment with prednisone 40 mg/d and clobetasol ointment 0.05%, she continued to develop extensive cutaneous bullae and new hemorrhagic bullae on the buccal mucosae (Figure 2), necessitating hospital admission. She clinically improved after prednisone was increased to 60 mg/d and mycophenolate mofetil 500 mg twice daily was added; however, she returned 8 days after discharge from the hospital with altered mental status, new-onset hematomas of the abdomen and right leg, and a hemoglobin level of 5.8 g/dL (reference range, 14.0–17.5 g/dL). Activated prothrombin time was prolonged without correction on mixing studies, raising concern for coagulation factor inhibition. Factor VIII activity was diminished to 9% and then 1% three days later. Mycophenolate mofetil was discontinued, and the patient was acutely stabilized with blood transfusions, intravenous immunoglobulin, tranexamic acid, and aminocaproic acid. Rituximab was initiated at 1000 mg and then administered again 2 weeks later. At 7-week follow-up, coagulation studies normalized, and there was no evidence of blistering dermatosis on examination.
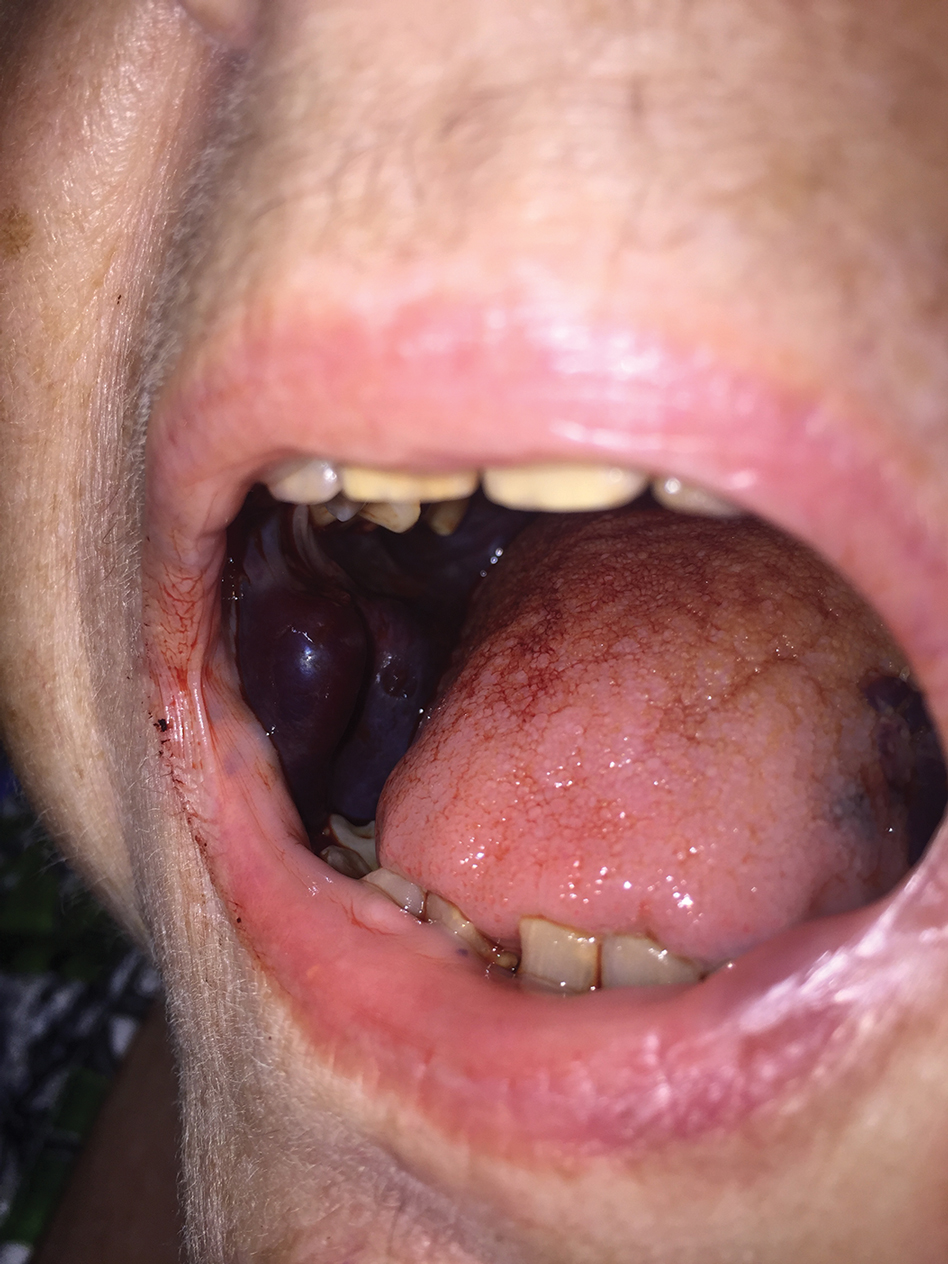
Bullous pemphigoid generally is seen in patients older than 60 years, and the incidence increases with age. The disease course follows formation of IgG antibodies against BP180 or BP230, leading to localized activation of the complement cascade at the basement membrane zone.1 Medications, vaccinations, UV radiation, and burns have been implicated in disease induction.2
Identification of antihemidesmosomal antibodies on lesional biopsy via direct immunofluorescence is the gold standard for diagnosis, though indirect antibodies measured via enzyme-linked immunosorbent assay may provide information regarding disease severity.1 Patients with milder disease may be treated with topical corticosteroids, doxycycline, and nicotinamide; however, severe disease requires treatment with systemic glucocorticoids and steroid-sparing agents.3 Rituximab initially was approved by the US Food and Drug Administration for the treatment of pemphigus vulgaris, and mounting evidence for the use of rituximab in BP is promising. Although data are limited to retrospective studies, rituximab has shown notable remission rates and steroid-sparing effects in those with moderate to severe BP.4
Acquired hemophilia A (AHA) is caused by the production of IgG autoantibodies, which block physiologic interactions between factor VIII and factor IX, phospholipids, and von Willebrand factor.5 Acquired hemophilia A often is diagnosed by prolonged activated prothrombin time and decreased factor VIII activity after a previously unaffected patient develops severe bleeding. Treatment involves re-establishing hemostasis and the use of corticosteroids and immunosuppressive agents to diminish autoantibody production.4
Bullous pemphigoid–associated AHA likely is due to antigenic similarity between BP180 and factor VIII, leading to concomitant neutralization of factor VIII with the production of BP-associated autoantibodies.5 Bullous pemphigoid–associated AHA has been reported with manifestations of bleeding concurrent with or after the development of dermatologic disease. Rituximab use has been reported with clinical efficacy in several cases, including our patient.6 Continued hematologic monitoring is recommended, as recurrences are common within the first 2 years.5
- Bağcı IS, Horváth ON, Ruzicka T, et al. Bullous pemphigoid. Autoimmun Rev. 2017;16:445-455.
- Schiavo AL, Ruocco E, Brancaccio G, et al. Bullous pemphigoid: etiology, pathogenesis, and inducing factors: facts and controversies. Clin Dermatol 2013;31:391-399.
- Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-332.
- Cho Y, Chu C, Wang L. First-line combination therapy with rituximab and corticosteroids provides a high complete remission rate in moderate-to-severe bullous pemphigoid. Br J Dermatol. 2015;173:302-304.
- Zdziarska J, Musial J. Acquired hemophilia A: an underdiagnosed severe bleeding disorder. Pol Arch Med Wewn. 2014;124:200-206.
- Binet Q, Lambert C, Sacré L, et al. Successful management of acquired hemophilia associated with bullous pemphigoid: a case report and review of the literature [published online March 28, 2017]. Case Rep Hematol. 2017;2017:2057019.
To the Editor:
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by the formation of antihemidesmosomal antibodies, resulting in tense bullae concentrated on the extremities and trunk that often are preceded by a pruritic urticarial phase.1 A rare complication of BP is the subsequent development of acquired hemophilia A. We report a case of BP with associated factor VIII–neutralizing antibodies in a patient who improved with prednisone and rituximab therapy.
A 78-year-old woman presented with red-orange pruritic plaques on the right heel that spread to involve the arms and legs, abdomen, and trunk with new-onset bullae over the course of 2 weeks (Figure 1). Dermatology was consulted, and a diagnosis of BP was confirmed via biopsy and direct immunofluorescence.

Despite treatment with prednisone 40 mg/d and clobetasol ointment 0.05%, she continued to develop extensive cutaneous bullae and new hemorrhagic bullae on the buccal mucosae (Figure 2), necessitating hospital admission. She clinically improved after prednisone was increased to 60 mg/d and mycophenolate mofetil 500 mg twice daily was added; however, she returned 8 days after discharge from the hospital with altered mental status, new-onset hematomas of the abdomen and right leg, and a hemoglobin level of 5.8 g/dL (reference range, 14.0–17.5 g/dL). Activated prothrombin time was prolonged without correction on mixing studies, raising concern for coagulation factor inhibition. Factor VIII activity was diminished to 9% and then 1% three days later. Mycophenolate mofetil was discontinued, and the patient was acutely stabilized with blood transfusions, intravenous immunoglobulin, tranexamic acid, and aminocaproic acid. Rituximab was initiated at 1000 mg and then administered again 2 weeks later. At 7-week follow-up, coagulation studies normalized, and there was no evidence of blistering dermatosis on examination.

Bullous pemphigoid generally is seen in patients older than 60 years, and the incidence increases with age. The disease course follows formation of IgG antibodies against BP180 or BP230, leading to localized activation of the complement cascade at the basement membrane zone.1 Medications, vaccinations, UV radiation, and burns have been implicated in disease induction.2
Identification of antihemidesmosomal antibodies on lesional biopsy via direct immunofluorescence is the gold standard for diagnosis, though indirect antibodies measured via enzyme-linked immunosorbent assay may provide information regarding disease severity.1 Patients with milder disease may be treated with topical corticosteroids, doxycycline, and nicotinamide; however, severe disease requires treatment with systemic glucocorticoids and steroid-sparing agents.3 Rituximab initially was approved by the US Food and Drug Administration for the treatment of pemphigus vulgaris, and mounting evidence for the use of rituximab in BP is promising. Although data are limited to retrospective studies, rituximab has shown notable remission rates and steroid-sparing effects in those with moderate to severe BP.4
Acquired hemophilia A (AHA) is caused by the production of IgG autoantibodies, which block physiologic interactions between factor VIII and factor IX, phospholipids, and von Willebrand factor.5 Acquired hemophilia A often is diagnosed by prolonged activated prothrombin time and decreased factor VIII activity after a previously unaffected patient develops severe bleeding. Treatment involves re-establishing hemostasis and the use of corticosteroids and immunosuppressive agents to diminish autoantibody production.4
Bullous pemphigoid–associated AHA likely is due to antigenic similarity between BP180 and factor VIII, leading to concomitant neutralization of factor VIII with the production of BP-associated autoantibodies.5 Bullous pemphigoid–associated AHA has been reported with manifestations of bleeding concurrent with or after the development of dermatologic disease. Rituximab use has been reported with clinical efficacy in several cases, including our patient.6 Continued hematologic monitoring is recommended, as recurrences are common within the first 2 years.5
To the Editor:
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by the formation of antihemidesmosomal antibodies, resulting in tense bullae concentrated on the extremities and trunk that often are preceded by a pruritic urticarial phase.1 A rare complication of BP is the subsequent development of acquired hemophilia A. We report a case of BP with associated factor VIII–neutralizing antibodies in a patient who improved with prednisone and rituximab therapy.
A 78-year-old woman presented with red-orange pruritic plaques on the right heel that spread to involve the arms and legs, abdomen, and trunk with new-onset bullae over the course of 2 weeks (Figure 1). Dermatology was consulted, and a diagnosis of BP was confirmed via biopsy and direct immunofluorescence.

Despite treatment with prednisone 40 mg/d and clobetasol ointment 0.05%, she continued to develop extensive cutaneous bullae and new hemorrhagic bullae on the buccal mucosae (Figure 2), necessitating hospital admission. She clinically improved after prednisone was increased to 60 mg/d and mycophenolate mofetil 500 mg twice daily was added; however, she returned 8 days after discharge from the hospital with altered mental status, new-onset hematomas of the abdomen and right leg, and a hemoglobin level of 5.8 g/dL (reference range, 14.0–17.5 g/dL). Activated prothrombin time was prolonged without correction on mixing studies, raising concern for coagulation factor inhibition. Factor VIII activity was diminished to 9% and then 1% three days later. Mycophenolate mofetil was discontinued, and the patient was acutely stabilized with blood transfusions, intravenous immunoglobulin, tranexamic acid, and aminocaproic acid. Rituximab was initiated at 1000 mg and then administered again 2 weeks later. At 7-week follow-up, coagulation studies normalized, and there was no evidence of blistering dermatosis on examination.

Bullous pemphigoid generally is seen in patients older than 60 years, and the incidence increases with age. The disease course follows formation of IgG antibodies against BP180 or BP230, leading to localized activation of the complement cascade at the basement membrane zone.1 Medications, vaccinations, UV radiation, and burns have been implicated in disease induction.2
Identification of antihemidesmosomal antibodies on lesional biopsy via direct immunofluorescence is the gold standard for diagnosis, though indirect antibodies measured via enzyme-linked immunosorbent assay may provide information regarding disease severity.1 Patients with milder disease may be treated with topical corticosteroids, doxycycline, and nicotinamide; however, severe disease requires treatment with systemic glucocorticoids and steroid-sparing agents.3 Rituximab initially was approved by the US Food and Drug Administration for the treatment of pemphigus vulgaris, and mounting evidence for the use of rituximab in BP is promising. Although data are limited to retrospective studies, rituximab has shown notable remission rates and steroid-sparing effects in those with moderate to severe BP.4
Acquired hemophilia A (AHA) is caused by the production of IgG autoantibodies, which block physiologic interactions between factor VIII and factor IX, phospholipids, and von Willebrand factor.5 Acquired hemophilia A often is diagnosed by prolonged activated prothrombin time and decreased factor VIII activity after a previously unaffected patient develops severe bleeding. Treatment involves re-establishing hemostasis and the use of corticosteroids and immunosuppressive agents to diminish autoantibody production.4
Bullous pemphigoid–associated AHA likely is due to antigenic similarity between BP180 and factor VIII, leading to concomitant neutralization of factor VIII with the production of BP-associated autoantibodies.5 Bullous pemphigoid–associated AHA has been reported with manifestations of bleeding concurrent with or after the development of dermatologic disease. Rituximab use has been reported with clinical efficacy in several cases, including our patient.6 Continued hematologic monitoring is recommended, as recurrences are common within the first 2 years.5
- Bağcı IS, Horváth ON, Ruzicka T, et al. Bullous pemphigoid. Autoimmun Rev. 2017;16:445-455.
- Schiavo AL, Ruocco E, Brancaccio G, et al. Bullous pemphigoid: etiology, pathogenesis, and inducing factors: facts and controversies. Clin Dermatol 2013;31:391-399.
- Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-332.
- Cho Y, Chu C, Wang L. First-line combination therapy with rituximab and corticosteroids provides a high complete remission rate in moderate-to-severe bullous pemphigoid. Br J Dermatol. 2015;173:302-304.
- Zdziarska J, Musial J. Acquired hemophilia A: an underdiagnosed severe bleeding disorder. Pol Arch Med Wewn. 2014;124:200-206.
- Binet Q, Lambert C, Sacré L, et al. Successful management of acquired hemophilia associated with bullous pemphigoid: a case report and review of the literature [published online March 28, 2017]. Case Rep Hematol. 2017;2017:2057019.
- Bağcı IS, Horváth ON, Ruzicka T, et al. Bullous pemphigoid. Autoimmun Rev. 2017;16:445-455.
- Schiavo AL, Ruocco E, Brancaccio G, et al. Bullous pemphigoid: etiology, pathogenesis, and inducing factors: facts and controversies. Clin Dermatol 2013;31:391-399.
- Schmidt E, Zillikens D. Pemphigoid diseases. Lancet. 2013;381:320-332.
- Cho Y, Chu C, Wang L. First-line combination therapy with rituximab and corticosteroids provides a high complete remission rate in moderate-to-severe bullous pemphigoid. Br J Dermatol. 2015;173:302-304.
- Zdziarska J, Musial J. Acquired hemophilia A: an underdiagnosed severe bleeding disorder. Pol Arch Med Wewn. 2014;124:200-206.
- Binet Q, Lambert C, Sacré L, et al. Successful management of acquired hemophilia associated with bullous pemphigoid: a case report and review of the literature [published online March 28, 2017]. Case Rep Hematol. 2017;2017:2057019.
Practice Points
- Physicians must be aware of the potential for acquired hemophilia A in patients with bullous pemphigoid (BP).
- Rituximab is an effective therapy for BP and should be considered for patients in this cohort.
Coming to a pill near you: The exercise molecule
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.
Exercise in a pill? Sign us up
You just got home from a long shift and you know you should go to the gym, but the bed is calling and you just answered. We know sometimes we have to make sacrifices in the name of fitness, but there just aren’t enough hours in the day. Unless our prayers have been answered. There could be a pill that has the benefits of working out without having to work out.
In a study published in Nature, investigators reported that they have identified a molecule made during exercise and used it on mice, which took in less food after being given the pill, which may open doors to understanding how exercise affects hunger.
In the first part of the study, the researchers found the molecule, known as Lac-Phe – which is synthesized from lactate and phenylalanine – in the blood plasma of mice after they had run on a treadmill.
The investigators then gave a Lac-Phe supplement to mice on high-fat diets and found that their food intake was about 50% of what other mice were eating. The supplement also improved their glucose tolerance.
Because the research also found Lac-Phe in humans who exercised, they hope that this pill will be in our future. “Our next steps include finding more details about how Lac-Phe mediates its effects in the body, including the brain,” Yong Xu, MD, of Baylor College of Medicine, Houston, said in a written statement. “Our goal is to learn to modulate this exercise pathway for therapeutic interventions.”
As always, we are rooting for you, science!
Gonorrhea and grandparents: A match made in prehistoric heaven
*Editorial note: LOTME takes no responsibility for any unfortunate imagery the reader may have experienced from the above headline.
Old people are the greatest. Back pains, cognitive decline, aches in all the diodes down your left side, there’s nothing quite like your golden years. Notably, however, humans are one of the few animals who experience true old age, as most creatures are adapted to maximize reproductive potential. As such, living past menopause is rare in the animal kingdom.
This is where the “grandmother hypothesis” comes in: Back in Ye Olde Stone Age, women who lived into old age could provide child care for younger women, because human babies require a lot more time and attention than other animal offspring. But how did humans end up living so long? Enter a group of Californian researchers, who believe they have an answer. It was gonorrhea.
When compared with the chimpanzee genome (as well as with Neanderthals and Denisovans, our closest ancestors), humans have a unique mutated version of the CD33 gene that lacks a sugar-binding site; the standard version uses the sugar-binding site to protect against autoimmune response in the body, but that same site actually suppresses the brain’s ability to clear away damaged brain cells and amyloid, which eventually leads to diseases like dementia. The mutated version allows microglia (brain immune cells) to attack and clear out this unwanted material. People with higher levels of this mutated CD33 variant actually have higher protection against Alzheimer’s.
Interestingly, gonorrhea bacteria are coated in the same sugar that standard CD33 receptors bind to, thus allowing them to bypass the body’s immune system. According to the researchers, the mutated CD33 version likely emerged as a protection against gonorrhea, depriving the bacteria of their “molecular mimicry” abilities. In one of life’s happy accidents, it turned out this mutation also protects against age-related diseases, thus allowing humans with the mutation to live longer. Obviously, this was a good thing, and we ran with it until the modern day. Now we have senior citizens climbing Everest, and all our politicians keep on politicking into their 70s and 80s ... well, everything has its drawbacks.
Parents raise a glass to children’s food addiction
There can be something pretty addicting about processed foods. Have you ever eaten just one french fry? Or taken just one cookie? If so, your willpower is incredible. For many of us, it can be a struggle to stop.
A recent study from the University of Michigan, which considered the existence of an eating phenotype, suggests our parents’ habits could be to blame.
By administering a series of questionnaires that inquired about food addiction, alcohol use disorders, cannabis use disorder, nicotine/e-cigarette dependence, and their family tree, investigators found that participants with a “paternal history of problematic alcohol use” had higher risk of food addiction but not obesity.
Apparently about one in five people display a clinically significant addiction to highly processed foods. It was noted that foods like ice cream, pizza, and french fries have high amounts of refined carbs and fats, which could trigger an addictive response.
Lindzey Hoover, a graduate student at the university who was the study’s lead author, noted that living in an environment where these foods are cheap and accessible can be really challenging for those with a family history of addiction. The investigators suggested that public health approaches, like restriction of other substances and marketing to kids, should be put in place for highly processed foods.
Maybe french fries should come with a warning label.
A prescription for America’s traffic problems
Nostalgia is a funny thing. Do you ever feel nostalgic about things that really weren’t very pleasant in the first place? Take, for instance, the morning commute. Here in the Washington area, more than 2 years into the COVID era, the traffic is still not what it used to be … and we kind of miss it.
Nah, not really. That was just a way to get everyone thinking about driving, because AAA has something of an explanation for the situation out there on the highways and byways of America. It’s drugs. No, not those kinds of drugs. This time it’s prescription drugs that are the problem. Well, part of the problem, anyway.
AAA did a survey last summer and found that nearly 50% of drivers “used one or more potentially impairing medications in the past 30 days. … The proportion of those choosing to drive is higher among those taking multiple medications.” How much higher? More than 63% of those with two or more prescriptions were driving within 2 hours of taking at least one of those meds, as were 71% of those taking three or more.
The 2,657 respondents also were asked about the types of potentially impairing drugs they were taking: 61% of those using antidepressants had been on the road within 2 hours of use at least once in the past 30 days, as had 73% of those taking an amphetamine, AAA said.
So there you have it. That guy in the BMW who’s been tailgating you for the last 3 miles? He may be a jerk, but there’s a good chance he’s a jerk with a prescription … or two … or three.