User login
For MD-IQ use only
New ESC cardio-oncology guideline aims to reduce cardiotoxicity
BARCELONA – Cardiovascular disease risk factors, as well as established disease, in patients undergoing cancer therapy can be safely managed to minimize cancer therapy–related cardiovascular toxicity (CVR-CVT), conclude the first cardio-oncology guidelines from the European Society of Cardiology.
The guidelines were presented at the annual congress of the European Society of Cardiology and published simultaneously in the European Heart Journal.
Guideline cochair Alexander R. Lyon, MD, PhD, told this news organization that the aim of the guideline was to “personalize the decision-making of a patient with cancer who has cardiovascular disease or is at risk of developing it from their treatment ... because it’s not one size fits all.”
because how you manage someone who’s at high risk is going to be different” than managing someone who is at moderate or low risk, he said.
“We’re doing a lot of surveillance because one of the big advantages of cardio-oncology is we know when someone is about to get treated,” Dr. Lyon, from the National Heart and Lung Institute, Imperial College London, and Cardio-Oncology Service, Royal Brompton Hospital, London, said.
“You don’t know in nature when someone’s going to have an acute myocardial infarction or acute viral myocarditis, but we do know when they’re coming into an oncology clinic to get an infusion of chemotherapy or tablets,” he noted.
The guidelines offer recommendations so that patients can “have their treatment safely and minimize interruptions.”
“We know these cancer therapies work; we’re here to get the best of both worlds” by minimizing cardiotoxicity, Dr. Lyon said.
Steady decline in cancer-related mortality
The guidelines note that since the 1990s there has been a “steady decline in cancer-related mortality, mirrored by a steady increase in cancer survival,” and the result is that “treatment-related side effects have gained more significance.”
Dr. Lyon said that between 2011 and 2021, there was a fivefold increase in the number of new referrals of cancer patients with cardiological consequences to his institution.
He said that one of main drivers is modifiable factors, such as smoking, obesity, and inactivity, which increase the risk for both cancer and cardiovascular disease.
“Allied to that, there’s been an improvement in treating cardiovascular diseases in people in their 40s, 50s, and 60s, so they’re surviving their heart failure, myocardial infarction, atrial fibrillation to develop cancers in later life.”
Combined with the aging population, the result is that “not only are many more people being diagnosed with cancer, because they’re living longer, but they have all these pre-existing heart risk factors, whether as confirmed disease or just the risk factors associated with that,” he said.
Another aspect is that many of the newer, targeted cancer therapies confer a cardiovascular risk.
Dr. Lyon said that the “most famous one” is trastuzumab, a monoclonal antibody that is used to treat HER2-positive breast cancer but that also causes left ventricular impairment “in about 15%-20% of the women taking it and can cause severe heart failure if it is missed.”
That, he continued, was the “forerunner of designer, targeted therapies,” and the subsequent “explosion” in the availability of modern cancer therapies has included many that confer cardiac issues.
The final reason for the greater interest in cardio-oncology, Dr. Lyon added, is the increasing awareness in oncology and hematology teams of the potential for cardiac problems among their patients.
“We have been reaching out to our oncology and hematology colleagues over the last 5-10 years to explain we’re here to help. We’re not here to stop their treatments, we’re here to support them.”
Presenting the guidelines, cochair Teresa López-Fernández, MD, cardiology department, La Paz University Hospital, IdiPAZ Research Institute, Madrid, said that the “spectrum of CVR-CVT presentations” includes arterial hypertension, cardiac arrhythmias, coronary artery disease, heart failure, and myocarditis.
She explained that cytotoxic cancer therapies are associated with an increased risk for cardiac toxicity that is most acute during the treatment phase but is not entirely diminished once it is over, then typically accumulates during long-term follow-up.
Crucially, the impact of cancer therapy on cardiovascular risk is dependent on several factors, such as patient age, cancer history, pre-existing cardiovascular risk factors or cardiovascular disease, and previous cardiotoxic cancer therapy.
There are nevertheless a number of potential strategies to reduce the risk for cardiac toxicity, including primary and secondary prevention prior to the start of cancer therapy and early CVR-CVT management during treatment, as well as cardiovascular risk assessment in the first year after treatment completion and cancer-survivorship programs.
To those ends, Dr. López-Fernández said the guidelines incorporate 272 new recommendations that cover the entire cardio-oncology care pathway, beginning with cardiovascular risk stratification before anticancer therapy.
They offer a risk-assessment checklist and make a series of recommendations for patients to be treated with potentially cardiotoxic drugs, such as anthracyclines, as well as recommendations on cardiac imaging.
The guidelines provide a range of recommendations for primary and secondary cancer therapy–related cardiovascular toxicity prevention, including minimization of the use of cardiotoxic drugs and the use of angiotensin-converting-enzyme inhibitors, angiotensin-receptor blockers, beta blockers, and statins for primary prevention.
They establish CVR-CVT monitoring protocols across the gamut of cancer therapies, from HER-targeted therapies, through immune checkpoint inhibitors, Bruton tyrosine kinase, CDK4/6, EGFR, VEGF, and ALK inhibitors, and androgen-deprivation and endocrine therapies, to the more novel CAR-T-cell therapies.
A section on radiotherapy-induced cardiovascular toxicity has its own protocol for the establishment of an individual’s mean heart dose of radiation or the amount of radiation exposure to the heart during treatment.
Next, Dr. Lyon looked at recommendations for the management of cardiovascular disease and cancer therapy–related cardiovascular toxicity in patients receiving anticancer treatment.
He underlined that treatment decisions should consider the cancer and cardiovascular symptom burden, the cancer prognosis, the requirements for cancer treatment, including alternative options, drug-drug interactions, and patient preferences.
Dr. Lyon highlighted the algorithms designed to aid the management of cardiac dysfunction related to anthracycline chemotherapy, HER2-targeted therapy, and immune checkpoint inhibitors, as well as QTc-prolonging anticancer drugs.
In the first 12 months after the completions of therapy, there are a number of risk factors for future cardiovascular disease, he continued.
These include high and very high baseline cardiovascular toxicity risk, anticancer treatments known to have a high risk for long-term cardiovascular complications, such as doxorubicin and radiotherapy, and moderate or severe CTR-CVT during anticancer treatment.
Over the long term, the guidelines recommend that surveillance in asymptomatic cancer survivors range from an annual cardiovascular risk assessment in low-risk patients to patient education and cardiovascular risk factor optimization, alongside regular transthoracic echocardiography in high-risk groups.
Finally, Dr. Lyon said the guidelines turn their attention to special populations, such as patients with cardiac masses and tumors, those with carcinoid heart disease, pregnant women receiving cancer therapy, as well as those with cardiac implantable electronic devices undergoing radiotherapy.
The guidelines were developed by the task force on cardio-oncology of the ESC, in collaboration with the European Hematology Association, the European Society for Therapeutic Radiology and Oncology, and the International Cardio-Oncology Society. Dr. Lyon declares relationships with Akcea, Takeda Pharmaceuticals, Pfizer, GlaxoSmithKline, AstraZeneca, Novartis, Ferring Pharmaceuticals, Heartfelt Technologies, Brainstorm, and Myocardial Solutions. Dr. López-Fernández declares relationships with Daiichi Sankyo, Almirall Spain, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
A version of this article first appeared on Medscape.com.
BARCELONA – Cardiovascular disease risk factors, as well as established disease, in patients undergoing cancer therapy can be safely managed to minimize cancer therapy–related cardiovascular toxicity (CVR-CVT), conclude the first cardio-oncology guidelines from the European Society of Cardiology.
The guidelines were presented at the annual congress of the European Society of Cardiology and published simultaneously in the European Heart Journal.
Guideline cochair Alexander R. Lyon, MD, PhD, told this news organization that the aim of the guideline was to “personalize the decision-making of a patient with cancer who has cardiovascular disease or is at risk of developing it from their treatment ... because it’s not one size fits all.”
because how you manage someone who’s at high risk is going to be different” than managing someone who is at moderate or low risk, he said.
“We’re doing a lot of surveillance because one of the big advantages of cardio-oncology is we know when someone is about to get treated,” Dr. Lyon, from the National Heart and Lung Institute, Imperial College London, and Cardio-Oncology Service, Royal Brompton Hospital, London, said.
“You don’t know in nature when someone’s going to have an acute myocardial infarction or acute viral myocarditis, but we do know when they’re coming into an oncology clinic to get an infusion of chemotherapy or tablets,” he noted.
The guidelines offer recommendations so that patients can “have their treatment safely and minimize interruptions.”
“We know these cancer therapies work; we’re here to get the best of both worlds” by minimizing cardiotoxicity, Dr. Lyon said.
Steady decline in cancer-related mortality
The guidelines note that since the 1990s there has been a “steady decline in cancer-related mortality, mirrored by a steady increase in cancer survival,” and the result is that “treatment-related side effects have gained more significance.”
Dr. Lyon said that between 2011 and 2021, there was a fivefold increase in the number of new referrals of cancer patients with cardiological consequences to his institution.
He said that one of main drivers is modifiable factors, such as smoking, obesity, and inactivity, which increase the risk for both cancer and cardiovascular disease.
“Allied to that, there’s been an improvement in treating cardiovascular diseases in people in their 40s, 50s, and 60s, so they’re surviving their heart failure, myocardial infarction, atrial fibrillation to develop cancers in later life.”
Combined with the aging population, the result is that “not only are many more people being diagnosed with cancer, because they’re living longer, but they have all these pre-existing heart risk factors, whether as confirmed disease or just the risk factors associated with that,” he said.
Another aspect is that many of the newer, targeted cancer therapies confer a cardiovascular risk.
Dr. Lyon said that the “most famous one” is trastuzumab, a monoclonal antibody that is used to treat HER2-positive breast cancer but that also causes left ventricular impairment “in about 15%-20% of the women taking it and can cause severe heart failure if it is missed.”
That, he continued, was the “forerunner of designer, targeted therapies,” and the subsequent “explosion” in the availability of modern cancer therapies has included many that confer cardiac issues.
The final reason for the greater interest in cardio-oncology, Dr. Lyon added, is the increasing awareness in oncology and hematology teams of the potential for cardiac problems among their patients.
“We have been reaching out to our oncology and hematology colleagues over the last 5-10 years to explain we’re here to help. We’re not here to stop their treatments, we’re here to support them.”
Presenting the guidelines, cochair Teresa López-Fernández, MD, cardiology department, La Paz University Hospital, IdiPAZ Research Institute, Madrid, said that the “spectrum of CVR-CVT presentations” includes arterial hypertension, cardiac arrhythmias, coronary artery disease, heart failure, and myocarditis.
She explained that cytotoxic cancer therapies are associated with an increased risk for cardiac toxicity that is most acute during the treatment phase but is not entirely diminished once it is over, then typically accumulates during long-term follow-up.
Crucially, the impact of cancer therapy on cardiovascular risk is dependent on several factors, such as patient age, cancer history, pre-existing cardiovascular risk factors or cardiovascular disease, and previous cardiotoxic cancer therapy.
There are nevertheless a number of potential strategies to reduce the risk for cardiac toxicity, including primary and secondary prevention prior to the start of cancer therapy and early CVR-CVT management during treatment, as well as cardiovascular risk assessment in the first year after treatment completion and cancer-survivorship programs.
To those ends, Dr. López-Fernández said the guidelines incorporate 272 new recommendations that cover the entire cardio-oncology care pathway, beginning with cardiovascular risk stratification before anticancer therapy.
They offer a risk-assessment checklist and make a series of recommendations for patients to be treated with potentially cardiotoxic drugs, such as anthracyclines, as well as recommendations on cardiac imaging.
The guidelines provide a range of recommendations for primary and secondary cancer therapy–related cardiovascular toxicity prevention, including minimization of the use of cardiotoxic drugs and the use of angiotensin-converting-enzyme inhibitors, angiotensin-receptor blockers, beta blockers, and statins for primary prevention.
They establish CVR-CVT monitoring protocols across the gamut of cancer therapies, from HER-targeted therapies, through immune checkpoint inhibitors, Bruton tyrosine kinase, CDK4/6, EGFR, VEGF, and ALK inhibitors, and androgen-deprivation and endocrine therapies, to the more novel CAR-T-cell therapies.
A section on radiotherapy-induced cardiovascular toxicity has its own protocol for the establishment of an individual’s mean heart dose of radiation or the amount of radiation exposure to the heart during treatment.
Next, Dr. Lyon looked at recommendations for the management of cardiovascular disease and cancer therapy–related cardiovascular toxicity in patients receiving anticancer treatment.
He underlined that treatment decisions should consider the cancer and cardiovascular symptom burden, the cancer prognosis, the requirements for cancer treatment, including alternative options, drug-drug interactions, and patient preferences.
Dr. Lyon highlighted the algorithms designed to aid the management of cardiac dysfunction related to anthracycline chemotherapy, HER2-targeted therapy, and immune checkpoint inhibitors, as well as QTc-prolonging anticancer drugs.
In the first 12 months after the completions of therapy, there are a number of risk factors for future cardiovascular disease, he continued.
These include high and very high baseline cardiovascular toxicity risk, anticancer treatments known to have a high risk for long-term cardiovascular complications, such as doxorubicin and radiotherapy, and moderate or severe CTR-CVT during anticancer treatment.
Over the long term, the guidelines recommend that surveillance in asymptomatic cancer survivors range from an annual cardiovascular risk assessment in low-risk patients to patient education and cardiovascular risk factor optimization, alongside regular transthoracic echocardiography in high-risk groups.
Finally, Dr. Lyon said the guidelines turn their attention to special populations, such as patients with cardiac masses and tumors, those with carcinoid heart disease, pregnant women receiving cancer therapy, as well as those with cardiac implantable electronic devices undergoing radiotherapy.
The guidelines were developed by the task force on cardio-oncology of the ESC, in collaboration with the European Hematology Association, the European Society for Therapeutic Radiology and Oncology, and the International Cardio-Oncology Society. Dr. Lyon declares relationships with Akcea, Takeda Pharmaceuticals, Pfizer, GlaxoSmithKline, AstraZeneca, Novartis, Ferring Pharmaceuticals, Heartfelt Technologies, Brainstorm, and Myocardial Solutions. Dr. López-Fernández declares relationships with Daiichi Sankyo, Almirall Spain, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
A version of this article first appeared on Medscape.com.
BARCELONA – Cardiovascular disease risk factors, as well as established disease, in patients undergoing cancer therapy can be safely managed to minimize cancer therapy–related cardiovascular toxicity (CVR-CVT), conclude the first cardio-oncology guidelines from the European Society of Cardiology.
The guidelines were presented at the annual congress of the European Society of Cardiology and published simultaneously in the European Heart Journal.
Guideline cochair Alexander R. Lyon, MD, PhD, told this news organization that the aim of the guideline was to “personalize the decision-making of a patient with cancer who has cardiovascular disease or is at risk of developing it from their treatment ... because it’s not one size fits all.”
because how you manage someone who’s at high risk is going to be different” than managing someone who is at moderate or low risk, he said.
“We’re doing a lot of surveillance because one of the big advantages of cardio-oncology is we know when someone is about to get treated,” Dr. Lyon, from the National Heart and Lung Institute, Imperial College London, and Cardio-Oncology Service, Royal Brompton Hospital, London, said.
“You don’t know in nature when someone’s going to have an acute myocardial infarction or acute viral myocarditis, but we do know when they’re coming into an oncology clinic to get an infusion of chemotherapy or tablets,” he noted.
The guidelines offer recommendations so that patients can “have their treatment safely and minimize interruptions.”
“We know these cancer therapies work; we’re here to get the best of both worlds” by minimizing cardiotoxicity, Dr. Lyon said.
Steady decline in cancer-related mortality
The guidelines note that since the 1990s there has been a “steady decline in cancer-related mortality, mirrored by a steady increase in cancer survival,” and the result is that “treatment-related side effects have gained more significance.”
Dr. Lyon said that between 2011 and 2021, there was a fivefold increase in the number of new referrals of cancer patients with cardiological consequences to his institution.
He said that one of main drivers is modifiable factors, such as smoking, obesity, and inactivity, which increase the risk for both cancer and cardiovascular disease.
“Allied to that, there’s been an improvement in treating cardiovascular diseases in people in their 40s, 50s, and 60s, so they’re surviving their heart failure, myocardial infarction, atrial fibrillation to develop cancers in later life.”
Combined with the aging population, the result is that “not only are many more people being diagnosed with cancer, because they’re living longer, but they have all these pre-existing heart risk factors, whether as confirmed disease or just the risk factors associated with that,” he said.
Another aspect is that many of the newer, targeted cancer therapies confer a cardiovascular risk.
Dr. Lyon said that the “most famous one” is trastuzumab, a monoclonal antibody that is used to treat HER2-positive breast cancer but that also causes left ventricular impairment “in about 15%-20% of the women taking it and can cause severe heart failure if it is missed.”
That, he continued, was the “forerunner of designer, targeted therapies,” and the subsequent “explosion” in the availability of modern cancer therapies has included many that confer cardiac issues.
The final reason for the greater interest in cardio-oncology, Dr. Lyon added, is the increasing awareness in oncology and hematology teams of the potential for cardiac problems among their patients.
“We have been reaching out to our oncology and hematology colleagues over the last 5-10 years to explain we’re here to help. We’re not here to stop their treatments, we’re here to support them.”
Presenting the guidelines, cochair Teresa López-Fernández, MD, cardiology department, La Paz University Hospital, IdiPAZ Research Institute, Madrid, said that the “spectrum of CVR-CVT presentations” includes arterial hypertension, cardiac arrhythmias, coronary artery disease, heart failure, and myocarditis.
She explained that cytotoxic cancer therapies are associated with an increased risk for cardiac toxicity that is most acute during the treatment phase but is not entirely diminished once it is over, then typically accumulates during long-term follow-up.
Crucially, the impact of cancer therapy on cardiovascular risk is dependent on several factors, such as patient age, cancer history, pre-existing cardiovascular risk factors or cardiovascular disease, and previous cardiotoxic cancer therapy.
There are nevertheless a number of potential strategies to reduce the risk for cardiac toxicity, including primary and secondary prevention prior to the start of cancer therapy and early CVR-CVT management during treatment, as well as cardiovascular risk assessment in the first year after treatment completion and cancer-survivorship programs.
To those ends, Dr. López-Fernández said the guidelines incorporate 272 new recommendations that cover the entire cardio-oncology care pathway, beginning with cardiovascular risk stratification before anticancer therapy.
They offer a risk-assessment checklist and make a series of recommendations for patients to be treated with potentially cardiotoxic drugs, such as anthracyclines, as well as recommendations on cardiac imaging.
The guidelines provide a range of recommendations for primary and secondary cancer therapy–related cardiovascular toxicity prevention, including minimization of the use of cardiotoxic drugs and the use of angiotensin-converting-enzyme inhibitors, angiotensin-receptor blockers, beta blockers, and statins for primary prevention.
They establish CVR-CVT monitoring protocols across the gamut of cancer therapies, from HER-targeted therapies, through immune checkpoint inhibitors, Bruton tyrosine kinase, CDK4/6, EGFR, VEGF, and ALK inhibitors, and androgen-deprivation and endocrine therapies, to the more novel CAR-T-cell therapies.
A section on radiotherapy-induced cardiovascular toxicity has its own protocol for the establishment of an individual’s mean heart dose of radiation or the amount of radiation exposure to the heart during treatment.
Next, Dr. Lyon looked at recommendations for the management of cardiovascular disease and cancer therapy–related cardiovascular toxicity in patients receiving anticancer treatment.
He underlined that treatment decisions should consider the cancer and cardiovascular symptom burden, the cancer prognosis, the requirements for cancer treatment, including alternative options, drug-drug interactions, and patient preferences.
Dr. Lyon highlighted the algorithms designed to aid the management of cardiac dysfunction related to anthracycline chemotherapy, HER2-targeted therapy, and immune checkpoint inhibitors, as well as QTc-prolonging anticancer drugs.
In the first 12 months after the completions of therapy, there are a number of risk factors for future cardiovascular disease, he continued.
These include high and very high baseline cardiovascular toxicity risk, anticancer treatments known to have a high risk for long-term cardiovascular complications, such as doxorubicin and radiotherapy, and moderate or severe CTR-CVT during anticancer treatment.
Over the long term, the guidelines recommend that surveillance in asymptomatic cancer survivors range from an annual cardiovascular risk assessment in low-risk patients to patient education and cardiovascular risk factor optimization, alongside regular transthoracic echocardiography in high-risk groups.
Finally, Dr. Lyon said the guidelines turn their attention to special populations, such as patients with cardiac masses and tumors, those with carcinoid heart disease, pregnant women receiving cancer therapy, as well as those with cardiac implantable electronic devices undergoing radiotherapy.
The guidelines were developed by the task force on cardio-oncology of the ESC, in collaboration with the European Hematology Association, the European Society for Therapeutic Radiology and Oncology, and the International Cardio-Oncology Society. Dr. Lyon declares relationships with Akcea, Takeda Pharmaceuticals, Pfizer, GlaxoSmithKline, AstraZeneca, Novartis, Ferring Pharmaceuticals, Heartfelt Technologies, Brainstorm, and Myocardial Solutions. Dr. López-Fernández declares relationships with Daiichi Sankyo, Almirall Spain, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
A version of this article first appeared on Medscape.com.
Bias and other barriers to HSCT access
For example, at the June 5 plenary session of the American Society of Clinical Oncology, Paul Richardson, MD, presented results of the DETERMINATION trial. More than 40,000 attendees heard his message that, in patients with newly diagnosed multiple myeloma (MM), up-front high-dose melphalan with autologous hematopoietic stem cell transplant (HSCT) support is associated with a significantly longer median progression-free survival of 67 months, compared with 46 months for patients randomized to delayed transplantation. The 5-year overall survival is similar for both arms.
While I and many of my colleagues in the field of transplantation used this data to strongly encourage MM patients to undergo HSCT as consolidation of their initial remission, others – including many investigators on the DETERMINATION trial – reached a starkly different conclusion. They suggested that delaying transplant was a valid option, since no survival benefit was observed.
Bias, when defined as a prejudice in favor of or against a specific treatment on the part of physicians and patients, has not been carefully studied in the realm of cellular therapies. However, physician and patient perceptions or misperceptions about the value or toxicity of a specific therapy are probably major drivers of whether a patient is referred for and accepts a particular form of treatment. In my specialization, that would mean either a stem cell transplant or other forms of cell therapy.
As with other medical procedures, in my field there are significant disparities in the use of transplantation among patients of different racial, ethnic, and age groups. Rates of both auto- and allo-HSCT are significantly higher for Whites than for African Americans. Hispanic patients have the lowest rates of utilization of auto-HSCT. Patients over the age of 60 have an eightfold risk of nonreferral to an HSCT center. Obviously, these nonreferrals reduce access to HSCT for older patients, particularly if they are seen at nonacademic centers.
One must question whether these disparities are caused by the physicians not believing in the value of transplantation, or simply not understanding its value? Or do they just lack the time to refer patients to a transplant center?
Socioeconomic factors, insurance status, age, and psychosocial characteristics all impact access to HSCT, yet some older patients with fewer economic resources and less insurance coverage still undergo the procedure. Is that because their physicians spent time educating these patients about the potential value of this treatment? Is it because the physicians went the extra mile to get these patients access to HSCT?
Physician preference also plays a significant role in whether a patient receives an allo-HSCT for acute myeloid leukemia and myelodysplastic syndrome. In a large survey of hematologists and oncologists performed by Pidala and colleagues, half of those surveyed agreed with the statement: “I feel the risk (morbidity and mortality) after HSCT is very high.” Most indicated that they “feel outcomes of unrelated donor HCT are much worse than matched sibling HCT.”
More importantly, more than one-third of those surveyed agreed that, “because of the high risks of allogeneic HSCT, I refer only after failure of conventional chemotherapy.” They voiced this opinion despite the fact that mortality rates after HSCT have been reduced significantly. With modern techniques, outcomes of unrelated donors are as good as with sibling donor transplants, and national guidelines strongly recommend that patients get referred before they become refractory to chemotherapy.
What can we do about this problem? Obviously, physician and provider education is important, but primary care physicians and general oncologists are already bombarded daily with new information. Relatively rare conditions like those we treat simply may not get their attention.
Personally, I think one of the most effective ways to overcome bias among physicians would be to target patients through a direct advertising campaign and public service announcements. Only by getting the attention of patients can they be directed to current, accurate information.
This solution could reduce the impact of physician biases or misperceptions and provide patients with greater access to lifesaving cell therapies.
Dr. Giralt is deputy division head of the division of hematologic malignancies at Memorial Sloan Kettering Cancer Center in New York.
For example, at the June 5 plenary session of the American Society of Clinical Oncology, Paul Richardson, MD, presented results of the DETERMINATION trial. More than 40,000 attendees heard his message that, in patients with newly diagnosed multiple myeloma (MM), up-front high-dose melphalan with autologous hematopoietic stem cell transplant (HSCT) support is associated with a significantly longer median progression-free survival of 67 months, compared with 46 months for patients randomized to delayed transplantation. The 5-year overall survival is similar for both arms.
While I and many of my colleagues in the field of transplantation used this data to strongly encourage MM patients to undergo HSCT as consolidation of their initial remission, others – including many investigators on the DETERMINATION trial – reached a starkly different conclusion. They suggested that delaying transplant was a valid option, since no survival benefit was observed.
Bias, when defined as a prejudice in favor of or against a specific treatment on the part of physicians and patients, has not been carefully studied in the realm of cellular therapies. However, physician and patient perceptions or misperceptions about the value or toxicity of a specific therapy are probably major drivers of whether a patient is referred for and accepts a particular form of treatment. In my specialization, that would mean either a stem cell transplant or other forms of cell therapy.
As with other medical procedures, in my field there are significant disparities in the use of transplantation among patients of different racial, ethnic, and age groups. Rates of both auto- and allo-HSCT are significantly higher for Whites than for African Americans. Hispanic patients have the lowest rates of utilization of auto-HSCT. Patients over the age of 60 have an eightfold risk of nonreferral to an HSCT center. Obviously, these nonreferrals reduce access to HSCT for older patients, particularly if they are seen at nonacademic centers.
One must question whether these disparities are caused by the physicians not believing in the value of transplantation, or simply not understanding its value? Or do they just lack the time to refer patients to a transplant center?
Socioeconomic factors, insurance status, age, and psychosocial characteristics all impact access to HSCT, yet some older patients with fewer economic resources and less insurance coverage still undergo the procedure. Is that because their physicians spent time educating these patients about the potential value of this treatment? Is it because the physicians went the extra mile to get these patients access to HSCT?
Physician preference also plays a significant role in whether a patient receives an allo-HSCT for acute myeloid leukemia and myelodysplastic syndrome. In a large survey of hematologists and oncologists performed by Pidala and colleagues, half of those surveyed agreed with the statement: “I feel the risk (morbidity and mortality) after HSCT is very high.” Most indicated that they “feel outcomes of unrelated donor HCT are much worse than matched sibling HCT.”
More importantly, more than one-third of those surveyed agreed that, “because of the high risks of allogeneic HSCT, I refer only after failure of conventional chemotherapy.” They voiced this opinion despite the fact that mortality rates after HSCT have been reduced significantly. With modern techniques, outcomes of unrelated donors are as good as with sibling donor transplants, and national guidelines strongly recommend that patients get referred before they become refractory to chemotherapy.
What can we do about this problem? Obviously, physician and provider education is important, but primary care physicians and general oncologists are already bombarded daily with new information. Relatively rare conditions like those we treat simply may not get their attention.
Personally, I think one of the most effective ways to overcome bias among physicians would be to target patients through a direct advertising campaign and public service announcements. Only by getting the attention of patients can they be directed to current, accurate information.
This solution could reduce the impact of physician biases or misperceptions and provide patients with greater access to lifesaving cell therapies.
Dr. Giralt is deputy division head of the division of hematologic malignancies at Memorial Sloan Kettering Cancer Center in New York.
For example, at the June 5 plenary session of the American Society of Clinical Oncology, Paul Richardson, MD, presented results of the DETERMINATION trial. More than 40,000 attendees heard his message that, in patients with newly diagnosed multiple myeloma (MM), up-front high-dose melphalan with autologous hematopoietic stem cell transplant (HSCT) support is associated with a significantly longer median progression-free survival of 67 months, compared with 46 months for patients randomized to delayed transplantation. The 5-year overall survival is similar for both arms.
While I and many of my colleagues in the field of transplantation used this data to strongly encourage MM patients to undergo HSCT as consolidation of their initial remission, others – including many investigators on the DETERMINATION trial – reached a starkly different conclusion. They suggested that delaying transplant was a valid option, since no survival benefit was observed.
Bias, when defined as a prejudice in favor of or against a specific treatment on the part of physicians and patients, has not been carefully studied in the realm of cellular therapies. However, physician and patient perceptions or misperceptions about the value or toxicity of a specific therapy are probably major drivers of whether a patient is referred for and accepts a particular form of treatment. In my specialization, that would mean either a stem cell transplant or other forms of cell therapy.
As with other medical procedures, in my field there are significant disparities in the use of transplantation among patients of different racial, ethnic, and age groups. Rates of both auto- and allo-HSCT are significantly higher for Whites than for African Americans. Hispanic patients have the lowest rates of utilization of auto-HSCT. Patients over the age of 60 have an eightfold risk of nonreferral to an HSCT center. Obviously, these nonreferrals reduce access to HSCT for older patients, particularly if they are seen at nonacademic centers.
One must question whether these disparities are caused by the physicians not believing in the value of transplantation, or simply not understanding its value? Or do they just lack the time to refer patients to a transplant center?
Socioeconomic factors, insurance status, age, and psychosocial characteristics all impact access to HSCT, yet some older patients with fewer economic resources and less insurance coverage still undergo the procedure. Is that because their physicians spent time educating these patients about the potential value of this treatment? Is it because the physicians went the extra mile to get these patients access to HSCT?
Physician preference also plays a significant role in whether a patient receives an allo-HSCT for acute myeloid leukemia and myelodysplastic syndrome. In a large survey of hematologists and oncologists performed by Pidala and colleagues, half of those surveyed agreed with the statement: “I feel the risk (morbidity and mortality) after HSCT is very high.” Most indicated that they “feel outcomes of unrelated donor HCT are much worse than matched sibling HCT.”
More importantly, more than one-third of those surveyed agreed that, “because of the high risks of allogeneic HSCT, I refer only after failure of conventional chemotherapy.” They voiced this opinion despite the fact that mortality rates after HSCT have been reduced significantly. With modern techniques, outcomes of unrelated donors are as good as with sibling donor transplants, and national guidelines strongly recommend that patients get referred before they become refractory to chemotherapy.
What can we do about this problem? Obviously, physician and provider education is important, but primary care physicians and general oncologists are already bombarded daily with new information. Relatively rare conditions like those we treat simply may not get their attention.
Personally, I think one of the most effective ways to overcome bias among physicians would be to target patients through a direct advertising campaign and public service announcements. Only by getting the attention of patients can they be directed to current, accurate information.
This solution could reduce the impact of physician biases or misperceptions and provide patients with greater access to lifesaving cell therapies.
Dr. Giralt is deputy division head of the division of hematologic malignancies at Memorial Sloan Kettering Cancer Center in New York.
Real medical news: Many teens trust fake medical news
The kids aren’t alright (at identifying fake news online)
If there’s one thing today’s teenagers are good at, it’s the Internet. What with their TokTiks, Fortnights, and memes whose lifespans are measured in milliseconds, it’s only natural that a contingent of people who have never known a world where the Internet wasn’t omnipresent would be highly skilled at navigating the dense, labyrinthine virtual world and the many falsehoods contained within.
Ladies and gentlemen, we’ve been duped, bamboozled, and smeckledorfed. New research from Slovakia suggests the opposite, in fact: Teenagers are just as bad as the rest of us, if not worse, at distinguishing between fake and real online health messaging.
For the study, 300 teenagers aged 16-19 years old were shown a group of messages about the health-promoting effects of fruits and vegetables; these messages were either false, true and neutral, or true with some sort of editing (a clickbait title or grammar mistakes) to mask their trustworthiness. Just under half of the subjects identified and trusted the true neutral messages over fake messages, while 41% couldn’t tell the difference and 11% trusted the fake messages more. In addition, they couldn’t tell the difference between fake and true messages when the content seemed plausible.
In a bit of good news, teenagers were just as likely to trust the edited true messages as the true neutral ones, except in instances when the edited message had a clickbait title. They were much less likely to trust those.
Based on their subjects’ rather poor performance, the study authors suggested teenagers go through health literacy and media literacy training, as well as develop their analytical and scientific reasoning. The LOTME staff rather suspects the study authors have never met a teenager. The only thing teenagers are going to get out of health literacy training is fodder for memes to put up on Myspace. Myspace is still a thing, right? We’re not old, we swear.
Can a computer help deliver babies?
Delivering babies can be a complicated business. Most doctors and midwives rely on their years of experience and training to make certain decisions for mothers in labor, but an artificial intelligence (AI) algorithm could make the entire process easier and safer.
Researchers from the Mayo Clinic recently reported that using an AI to analyze women’s labor patterns was very successful in determining whether a vaginal or cesarean delivery was appropriate.
They examined over 700 factors and over 66,000 deliveries from the National Institute of Child Health and Human Development’s multicenter Consortium on Safe Labor database to produce a risk-prediction model that may “provide an alternative to conventional labor charts and promote individualization of clinical decisions using baseline and labor characteristics of each patient,” they said in a written statement from the clinic.
It is hoped that the AI will reduce the risk of possible complications and the costs associated with maternal mortality. The AI also could be a significant tool for doctors and midwives in rural areas to determine when a patient needs to be moved to a location with a higher level of care.
“We believe the algorithm will work in real time, meaning every input of new data during an expectant woman’s labor automatically recalculates the risk of adverse outcome,” said senior author Abimbola Famuyide, MD, of the Mayo Clinic.
If it all works out, many lives and dollars could be saved, thanks to science.
Democracy, meet COVID-19
Everywhere you look, it seems, someone is trying to keep someone else from doing something: Don’t carry a gun. Don’t get an abortion. Don’t drive so fast. Don’t inhale that whipped cream. Don’t get a vaccine. Don’t put that in your mouth.
One of the biggies these days is voting rights. Some people are trying to prevent other people from voting. But why? Well, turns out that turnout can be bad for your health … at least during a worldwide pandemic event.
The evidence for that claim comes from researchers who examined the Italian national constitutional referendum conducted in September 2020 along with elections for assembly representatives in 7 of the country’s 20 regions and for mayors in about 12% of municipalities. The combination mattered: Voter turnout was higher in the municipalities that voted for both the referendum and local elections (69%), compared with municipalities voting only for the referendum (47%), the investigators reported in the Journal of Economic Behavior & Organization.
Also occurring in September of 2020 was, as we mentioned, a worldwide pandemic event. You may have heard about it.
The investigators considered the differences in election turnout between the various municipalities and compared them with new weekly COVID-19 infections at the municipality level. “Our model shows that something as fundamental as casting a vote can come at a cost,” investigator Giuseppe Moscelli, PhD, of the University of Surrey (England) said in a written statement.
What was the cost? Each 1% increase in turnout, they found, amounted to an average 1.1% increase in COVID infections after the elections.
See? More people voting means more COVID, which is bad. Which brings us to today’s lesson in people preventing other people from doing something. Don’t let COVID win. Stay in your house and never come out. And get that smeckledorf out of your mouth. You don’t know where it’s been.
The kids aren’t alright (at identifying fake news online)
If there’s one thing today’s teenagers are good at, it’s the Internet. What with their TokTiks, Fortnights, and memes whose lifespans are measured in milliseconds, it’s only natural that a contingent of people who have never known a world where the Internet wasn’t omnipresent would be highly skilled at navigating the dense, labyrinthine virtual world and the many falsehoods contained within.
Ladies and gentlemen, we’ve been duped, bamboozled, and smeckledorfed. New research from Slovakia suggests the opposite, in fact: Teenagers are just as bad as the rest of us, if not worse, at distinguishing between fake and real online health messaging.
For the study, 300 teenagers aged 16-19 years old were shown a group of messages about the health-promoting effects of fruits and vegetables; these messages were either false, true and neutral, or true with some sort of editing (a clickbait title or grammar mistakes) to mask their trustworthiness. Just under half of the subjects identified and trusted the true neutral messages over fake messages, while 41% couldn’t tell the difference and 11% trusted the fake messages more. In addition, they couldn’t tell the difference between fake and true messages when the content seemed plausible.
In a bit of good news, teenagers were just as likely to trust the edited true messages as the true neutral ones, except in instances when the edited message had a clickbait title. They were much less likely to trust those.
Based on their subjects’ rather poor performance, the study authors suggested teenagers go through health literacy and media literacy training, as well as develop their analytical and scientific reasoning. The LOTME staff rather suspects the study authors have never met a teenager. The only thing teenagers are going to get out of health literacy training is fodder for memes to put up on Myspace. Myspace is still a thing, right? We’re not old, we swear.
Can a computer help deliver babies?
Delivering babies can be a complicated business. Most doctors and midwives rely on their years of experience and training to make certain decisions for mothers in labor, but an artificial intelligence (AI) algorithm could make the entire process easier and safer.
Researchers from the Mayo Clinic recently reported that using an AI to analyze women’s labor patterns was very successful in determining whether a vaginal or cesarean delivery was appropriate.
They examined over 700 factors and over 66,000 deliveries from the National Institute of Child Health and Human Development’s multicenter Consortium on Safe Labor database to produce a risk-prediction model that may “provide an alternative to conventional labor charts and promote individualization of clinical decisions using baseline and labor characteristics of each patient,” they said in a written statement from the clinic.
It is hoped that the AI will reduce the risk of possible complications and the costs associated with maternal mortality. The AI also could be a significant tool for doctors and midwives in rural areas to determine when a patient needs to be moved to a location with a higher level of care.
“We believe the algorithm will work in real time, meaning every input of new data during an expectant woman’s labor automatically recalculates the risk of adverse outcome,” said senior author Abimbola Famuyide, MD, of the Mayo Clinic.
If it all works out, many lives and dollars could be saved, thanks to science.
Democracy, meet COVID-19
Everywhere you look, it seems, someone is trying to keep someone else from doing something: Don’t carry a gun. Don’t get an abortion. Don’t drive so fast. Don’t inhale that whipped cream. Don’t get a vaccine. Don’t put that in your mouth.
One of the biggies these days is voting rights. Some people are trying to prevent other people from voting. But why? Well, turns out that turnout can be bad for your health … at least during a worldwide pandemic event.
The evidence for that claim comes from researchers who examined the Italian national constitutional referendum conducted in September 2020 along with elections for assembly representatives in 7 of the country’s 20 regions and for mayors in about 12% of municipalities. The combination mattered: Voter turnout was higher in the municipalities that voted for both the referendum and local elections (69%), compared with municipalities voting only for the referendum (47%), the investigators reported in the Journal of Economic Behavior & Organization.
Also occurring in September of 2020 was, as we mentioned, a worldwide pandemic event. You may have heard about it.
The investigators considered the differences in election turnout between the various municipalities and compared them with new weekly COVID-19 infections at the municipality level. “Our model shows that something as fundamental as casting a vote can come at a cost,” investigator Giuseppe Moscelli, PhD, of the University of Surrey (England) said in a written statement.
What was the cost? Each 1% increase in turnout, they found, amounted to an average 1.1% increase in COVID infections after the elections.
See? More people voting means more COVID, which is bad. Which brings us to today’s lesson in people preventing other people from doing something. Don’t let COVID win. Stay in your house and never come out. And get that smeckledorf out of your mouth. You don’t know where it’s been.
The kids aren’t alright (at identifying fake news online)
If there’s one thing today’s teenagers are good at, it’s the Internet. What with their TokTiks, Fortnights, and memes whose lifespans are measured in milliseconds, it’s only natural that a contingent of people who have never known a world where the Internet wasn’t omnipresent would be highly skilled at navigating the dense, labyrinthine virtual world and the many falsehoods contained within.
Ladies and gentlemen, we’ve been duped, bamboozled, and smeckledorfed. New research from Slovakia suggests the opposite, in fact: Teenagers are just as bad as the rest of us, if not worse, at distinguishing between fake and real online health messaging.
For the study, 300 teenagers aged 16-19 years old were shown a group of messages about the health-promoting effects of fruits and vegetables; these messages were either false, true and neutral, or true with some sort of editing (a clickbait title or grammar mistakes) to mask their trustworthiness. Just under half of the subjects identified and trusted the true neutral messages over fake messages, while 41% couldn’t tell the difference and 11% trusted the fake messages more. In addition, they couldn’t tell the difference between fake and true messages when the content seemed plausible.
In a bit of good news, teenagers were just as likely to trust the edited true messages as the true neutral ones, except in instances when the edited message had a clickbait title. They were much less likely to trust those.
Based on their subjects’ rather poor performance, the study authors suggested teenagers go through health literacy and media literacy training, as well as develop their analytical and scientific reasoning. The LOTME staff rather suspects the study authors have never met a teenager. The only thing teenagers are going to get out of health literacy training is fodder for memes to put up on Myspace. Myspace is still a thing, right? We’re not old, we swear.
Can a computer help deliver babies?
Delivering babies can be a complicated business. Most doctors and midwives rely on their years of experience and training to make certain decisions for mothers in labor, but an artificial intelligence (AI) algorithm could make the entire process easier and safer.
Researchers from the Mayo Clinic recently reported that using an AI to analyze women’s labor patterns was very successful in determining whether a vaginal or cesarean delivery was appropriate.
They examined over 700 factors and over 66,000 deliveries from the National Institute of Child Health and Human Development’s multicenter Consortium on Safe Labor database to produce a risk-prediction model that may “provide an alternative to conventional labor charts and promote individualization of clinical decisions using baseline and labor characteristics of each patient,” they said in a written statement from the clinic.
It is hoped that the AI will reduce the risk of possible complications and the costs associated with maternal mortality. The AI also could be a significant tool for doctors and midwives in rural areas to determine when a patient needs to be moved to a location with a higher level of care.
“We believe the algorithm will work in real time, meaning every input of new data during an expectant woman’s labor automatically recalculates the risk of adverse outcome,” said senior author Abimbola Famuyide, MD, of the Mayo Clinic.
If it all works out, many lives and dollars could be saved, thanks to science.
Democracy, meet COVID-19
Everywhere you look, it seems, someone is trying to keep someone else from doing something: Don’t carry a gun. Don’t get an abortion. Don’t drive so fast. Don’t inhale that whipped cream. Don’t get a vaccine. Don’t put that in your mouth.
One of the biggies these days is voting rights. Some people are trying to prevent other people from voting. But why? Well, turns out that turnout can be bad for your health … at least during a worldwide pandemic event.
The evidence for that claim comes from researchers who examined the Italian national constitutional referendum conducted in September 2020 along with elections for assembly representatives in 7 of the country’s 20 regions and for mayors in about 12% of municipalities. The combination mattered: Voter turnout was higher in the municipalities that voted for both the referendum and local elections (69%), compared with municipalities voting only for the referendum (47%), the investigators reported in the Journal of Economic Behavior & Organization.
Also occurring in September of 2020 was, as we mentioned, a worldwide pandemic event. You may have heard about it.
The investigators considered the differences in election turnout between the various municipalities and compared them with new weekly COVID-19 infections at the municipality level. “Our model shows that something as fundamental as casting a vote can come at a cost,” investigator Giuseppe Moscelli, PhD, of the University of Surrey (England) said in a written statement.
What was the cost? Each 1% increase in turnout, they found, amounted to an average 1.1% increase in COVID infections after the elections.
See? More people voting means more COVID, which is bad. Which brings us to today’s lesson in people preventing other people from doing something. Don’t let COVID win. Stay in your house and never come out. And get that smeckledorf out of your mouth. You don’t know where it’s been.
Many young kids with COVID may show no symptoms
BY WILL PASS
Just 14% of adults who tested positive for SARS-CoV-2 were asymptomatic, versus 37% of children aged 0-4 years, in the paper. This raises concern that parents, childcare providers, and preschools may be underestimating infection in seemingly healthy young kids who have been exposed to COVID, wrote lead author Ruth A. Karron, MD, and colleagues in JAMA Network Open.
Methods
The new research involved 690 individuals from 175 households in Maryland who were monitored closely between November 2020 and October 2021. Every week for 8 months, participants completed online symptom checks and underwent PCR testing using nasal swabs, with symptomatic individuals submitting additional swabs for analysis.
“What was different about our study [compared with previous studies] was the intensity of our collection, and the fact that we collected specimens from asymptomatic people,” said Dr. Karron, a pediatrician and professor in the department of international health, Johns Hopkins University, Baltimore, in an interview. “You shed more virus earlier in the infection than later, and the fact that we were sampling every single week meant that we could pick up those early infections.”
The study also stands out for its focus on young children, Dr. Karron said. Enrollment required all households to have at least one child aged 0-4 years, so 256 out of 690 participants (37.1%) were in this youngest age group. The remainder of the population consisted of 100 older children aged 5-17 years (14.5%) and 334 adults aged 18-74 years (48.4%).
Children 4 and under more than twice as likely to be asymptomatic
By the end of the study, 51 participants had tested positive for SARS-CoV-2, among whom 14 had no symptoms. A closer look showed that children 0-4 years of age who contracted COVID were more than twice as likely to be asymptomatic as infected adults (36.8% vs. 14.3%).
The relationship between symptoms and viral load also differed between adults and young children.
While adults with high viral loads – suggesting greater contagiousness – typically had more severe COVID symptoms, no correlation was found in young kids, meaning children with mild or no symptoms could still be highly contagious.
Dr. Karron said these findings should help parents and other stakeholders make better-informed decisions based on known risks. She recommended testing young, asymptomatic children for COVID if they have been exposed to infected individuals, then acting accordingly based on the results.
“If a family is infected with the virus, and the 2-year-old is asymptomatic, and people are thinking about a visit to elderly grandparents who may be frail, one shouldn’t assume that the 2-year-old is uninfected,” Dr. Karron said. “That child should be tested along with other family members.”
Testing should also be considered for young children exposed to COVID at childcare facilities, she added.
But not every expert consulted for this piece shared these opinions of Dr. Karron.
“I question whether that effort is worth it,” said Dean Blumberg, MD, professor and chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, Calif.
He noted that recent Food and Drug Administration guidance for COVID testing calls for three negative at-home antigen tests to confirm lack of infection.
“That would take 4 days to get those tests done,” he said. “So, it’s a lot of testing. It’s a lot of record keeping, it’s inconvenient, it’s uncomfortable to be tested, and I just question whether it’s worth that effort.”
Applicability of findings to today questioned
Dr. Blumberg also questioned whether the study, which was completed almost a year ago, reflects the current pandemic landscape.
“At the time this study was done, it was predominantly Delta [variant instead of Omicron],” Dr. Blumberg said. “The other issue [with the study] is that … most of the children didn’t have preexisting immunity, so you have to take that into account.”
Preexisting immunity – whether from exposure or vaccination – could lower viral loads, so asymptomatic children today really could be less contagious than they were when the study was done, according to Dr. Blumberg. Kids without symptoms are also less likely to spread the virus, because they aren’t coughing or sneezing, he added.
Sara R. Kim, MD, and Janet A. Englund, MD, of the Seattle Children’s Research Institute, University of Washington, said it’s challenging to know how applicable the findings are, although they sided more with the investigators than Dr. Blumberg.
“Given the higher rate of transmissibility and infectivity of the Omicron variant, it is difficult to make direct associations between findings reported during this study period and those present in the current era during which the Omicron variant is circulating,” they wrote in an accompanying editorial. “However, the higher rates of asymptomatic infection observed among children in this study are likely to be consistent with those observed for current and future viral variants.”
Although the experts offered different interpretations of the findings, they shared similar perspectives on vaccination.
“The most important thing that parents can do is get their kids vaccinated, be vaccinated themselves, and have everybody in the household vaccinated and up to date for all doses that are indicated,” Dr. Blumberg said.
Dr. Karron noted that vaccination will be increasingly important in the coming months.
“Summer is ending; school is starting,” she said. “We’re going to be in large groups indoors again very soon. To keep young children safe, I think it’s really important for them to get vaccinated.”
The study was funded by the CDC. The investigators disclosed no other relationships. Dr. Englund disclosed relationships with AstraZeneca, GlaxoSmithKline, Merck, and others. Dr. Kim and Dr. Blumberg disclosed no relevant conflicts of interest.
BY WILL PASS
Just 14% of adults who tested positive for SARS-CoV-2 were asymptomatic, versus 37% of children aged 0-4 years, in the paper. This raises concern that parents, childcare providers, and preschools may be underestimating infection in seemingly healthy young kids who have been exposed to COVID, wrote lead author Ruth A. Karron, MD, and colleagues in JAMA Network Open.
Methods
The new research involved 690 individuals from 175 households in Maryland who were monitored closely between November 2020 and October 2021. Every week for 8 months, participants completed online symptom checks and underwent PCR testing using nasal swabs, with symptomatic individuals submitting additional swabs for analysis.
“What was different about our study [compared with previous studies] was the intensity of our collection, and the fact that we collected specimens from asymptomatic people,” said Dr. Karron, a pediatrician and professor in the department of international health, Johns Hopkins University, Baltimore, in an interview. “You shed more virus earlier in the infection than later, and the fact that we were sampling every single week meant that we could pick up those early infections.”
The study also stands out for its focus on young children, Dr. Karron said. Enrollment required all households to have at least one child aged 0-4 years, so 256 out of 690 participants (37.1%) were in this youngest age group. The remainder of the population consisted of 100 older children aged 5-17 years (14.5%) and 334 adults aged 18-74 years (48.4%).
Children 4 and under more than twice as likely to be asymptomatic
By the end of the study, 51 participants had tested positive for SARS-CoV-2, among whom 14 had no symptoms. A closer look showed that children 0-4 years of age who contracted COVID were more than twice as likely to be asymptomatic as infected adults (36.8% vs. 14.3%).
The relationship between symptoms and viral load also differed between adults and young children.
While adults with high viral loads – suggesting greater contagiousness – typically had more severe COVID symptoms, no correlation was found in young kids, meaning children with mild or no symptoms could still be highly contagious.
Dr. Karron said these findings should help parents and other stakeholders make better-informed decisions based on known risks. She recommended testing young, asymptomatic children for COVID if they have been exposed to infected individuals, then acting accordingly based on the results.
“If a family is infected with the virus, and the 2-year-old is asymptomatic, and people are thinking about a visit to elderly grandparents who may be frail, one shouldn’t assume that the 2-year-old is uninfected,” Dr. Karron said. “That child should be tested along with other family members.”
Testing should also be considered for young children exposed to COVID at childcare facilities, she added.
But not every expert consulted for this piece shared these opinions of Dr. Karron.
“I question whether that effort is worth it,” said Dean Blumberg, MD, professor and chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, Calif.
He noted that recent Food and Drug Administration guidance for COVID testing calls for three negative at-home antigen tests to confirm lack of infection.
“That would take 4 days to get those tests done,” he said. “So, it’s a lot of testing. It’s a lot of record keeping, it’s inconvenient, it’s uncomfortable to be tested, and I just question whether it’s worth that effort.”
Applicability of findings to today questioned
Dr. Blumberg also questioned whether the study, which was completed almost a year ago, reflects the current pandemic landscape.
“At the time this study was done, it was predominantly Delta [variant instead of Omicron],” Dr. Blumberg said. “The other issue [with the study] is that … most of the children didn’t have preexisting immunity, so you have to take that into account.”
Preexisting immunity – whether from exposure or vaccination – could lower viral loads, so asymptomatic children today really could be less contagious than they were when the study was done, according to Dr. Blumberg. Kids without symptoms are also less likely to spread the virus, because they aren’t coughing or sneezing, he added.
Sara R. Kim, MD, and Janet A. Englund, MD, of the Seattle Children’s Research Institute, University of Washington, said it’s challenging to know how applicable the findings are, although they sided more with the investigators than Dr. Blumberg.
“Given the higher rate of transmissibility and infectivity of the Omicron variant, it is difficult to make direct associations between findings reported during this study period and those present in the current era during which the Omicron variant is circulating,” they wrote in an accompanying editorial. “However, the higher rates of asymptomatic infection observed among children in this study are likely to be consistent with those observed for current and future viral variants.”
Although the experts offered different interpretations of the findings, they shared similar perspectives on vaccination.
“The most important thing that parents can do is get their kids vaccinated, be vaccinated themselves, and have everybody in the household vaccinated and up to date for all doses that are indicated,” Dr. Blumberg said.
Dr. Karron noted that vaccination will be increasingly important in the coming months.
“Summer is ending; school is starting,” she said. “We’re going to be in large groups indoors again very soon. To keep young children safe, I think it’s really important for them to get vaccinated.”
The study was funded by the CDC. The investigators disclosed no other relationships. Dr. Englund disclosed relationships with AstraZeneca, GlaxoSmithKline, Merck, and others. Dr. Kim and Dr. Blumberg disclosed no relevant conflicts of interest.
BY WILL PASS
Just 14% of adults who tested positive for SARS-CoV-2 were asymptomatic, versus 37% of children aged 0-4 years, in the paper. This raises concern that parents, childcare providers, and preschools may be underestimating infection in seemingly healthy young kids who have been exposed to COVID, wrote lead author Ruth A. Karron, MD, and colleagues in JAMA Network Open.
Methods
The new research involved 690 individuals from 175 households in Maryland who were monitored closely between November 2020 and October 2021. Every week for 8 months, participants completed online symptom checks and underwent PCR testing using nasal swabs, with symptomatic individuals submitting additional swabs for analysis.
“What was different about our study [compared with previous studies] was the intensity of our collection, and the fact that we collected specimens from asymptomatic people,” said Dr. Karron, a pediatrician and professor in the department of international health, Johns Hopkins University, Baltimore, in an interview. “You shed more virus earlier in the infection than later, and the fact that we were sampling every single week meant that we could pick up those early infections.”
The study also stands out for its focus on young children, Dr. Karron said. Enrollment required all households to have at least one child aged 0-4 years, so 256 out of 690 participants (37.1%) were in this youngest age group. The remainder of the population consisted of 100 older children aged 5-17 years (14.5%) and 334 adults aged 18-74 years (48.4%).
Children 4 and under more than twice as likely to be asymptomatic
By the end of the study, 51 participants had tested positive for SARS-CoV-2, among whom 14 had no symptoms. A closer look showed that children 0-4 years of age who contracted COVID were more than twice as likely to be asymptomatic as infected adults (36.8% vs. 14.3%).
The relationship between symptoms and viral load also differed between adults and young children.
While adults with high viral loads – suggesting greater contagiousness – typically had more severe COVID symptoms, no correlation was found in young kids, meaning children with mild or no symptoms could still be highly contagious.
Dr. Karron said these findings should help parents and other stakeholders make better-informed decisions based on known risks. She recommended testing young, asymptomatic children for COVID if they have been exposed to infected individuals, then acting accordingly based on the results.
“If a family is infected with the virus, and the 2-year-old is asymptomatic, and people are thinking about a visit to elderly grandparents who may be frail, one shouldn’t assume that the 2-year-old is uninfected,” Dr. Karron said. “That child should be tested along with other family members.”
Testing should also be considered for young children exposed to COVID at childcare facilities, she added.
But not every expert consulted for this piece shared these opinions of Dr. Karron.
“I question whether that effort is worth it,” said Dean Blumberg, MD, professor and chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, Calif.
He noted that recent Food and Drug Administration guidance for COVID testing calls for three negative at-home antigen tests to confirm lack of infection.
“That would take 4 days to get those tests done,” he said. “So, it’s a lot of testing. It’s a lot of record keeping, it’s inconvenient, it’s uncomfortable to be tested, and I just question whether it’s worth that effort.”
Applicability of findings to today questioned
Dr. Blumberg also questioned whether the study, which was completed almost a year ago, reflects the current pandemic landscape.
“At the time this study was done, it was predominantly Delta [variant instead of Omicron],” Dr. Blumberg said. “The other issue [with the study] is that … most of the children didn’t have preexisting immunity, so you have to take that into account.”
Preexisting immunity – whether from exposure or vaccination – could lower viral loads, so asymptomatic children today really could be less contagious than they were when the study was done, according to Dr. Blumberg. Kids without symptoms are also less likely to spread the virus, because they aren’t coughing or sneezing, he added.
Sara R. Kim, MD, and Janet A. Englund, MD, of the Seattle Children’s Research Institute, University of Washington, said it’s challenging to know how applicable the findings are, although they sided more with the investigators than Dr. Blumberg.
“Given the higher rate of transmissibility and infectivity of the Omicron variant, it is difficult to make direct associations between findings reported during this study period and those present in the current era during which the Omicron variant is circulating,” they wrote in an accompanying editorial. “However, the higher rates of asymptomatic infection observed among children in this study are likely to be consistent with those observed for current and future viral variants.”
Although the experts offered different interpretations of the findings, they shared similar perspectives on vaccination.
“The most important thing that parents can do is get their kids vaccinated, be vaccinated themselves, and have everybody in the household vaccinated and up to date for all doses that are indicated,” Dr. Blumberg said.
Dr. Karron noted that vaccination will be increasingly important in the coming months.
“Summer is ending; school is starting,” she said. “We’re going to be in large groups indoors again very soon. To keep young children safe, I think it’s really important for them to get vaccinated.”
The study was funded by the CDC. The investigators disclosed no other relationships. Dr. Englund disclosed relationships with AstraZeneca, GlaxoSmithKline, Merck, and others. Dr. Kim and Dr. Blumberg disclosed no relevant conflicts of interest.
FROM JAMA NETWORK OPEN
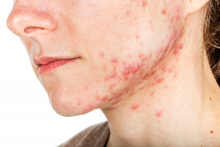
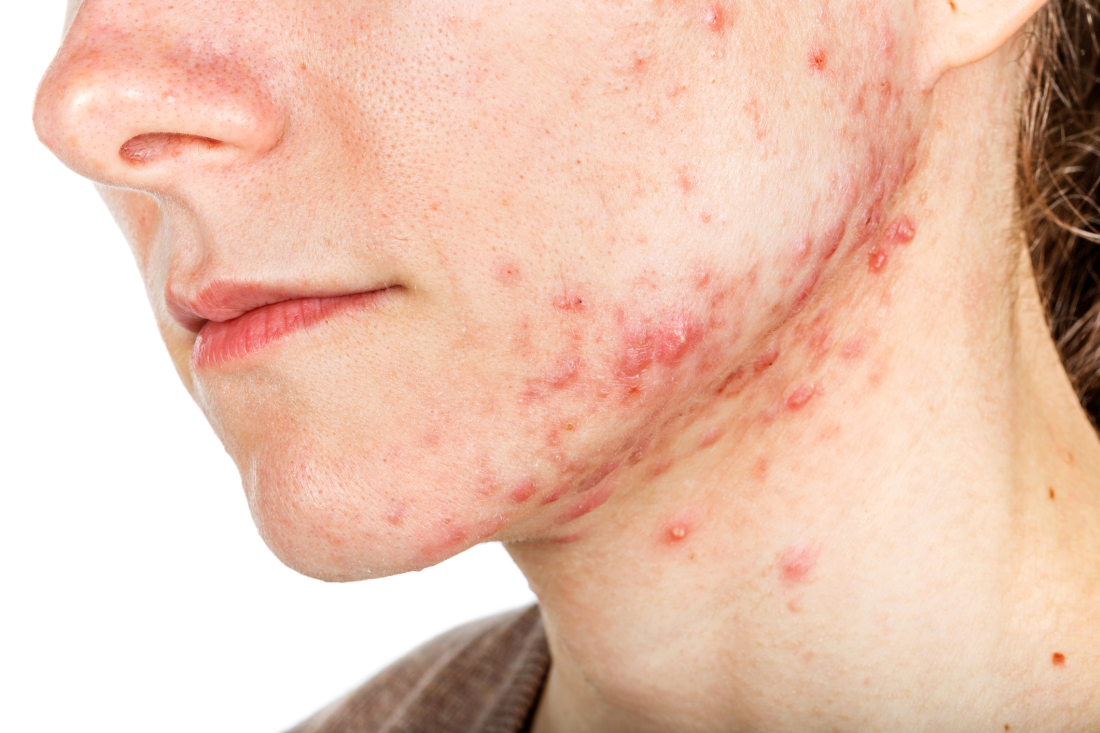
Hormonal therapy a safe, long term option for older women with recalcitrant acne
PORTLAND, ORE. – During her dermatology residency training at the University of California, Irvine, Medical Center, Jenny Murase, MD, remembers hearing a colleague say that her most angry patients of the day were adult women with recalcitrant acne who present to the clinic with questions like, “My skin has been clear my whole life! What’s going on?”
Such . In fact, 82% fail multiple courses of systemic antibiotics and 32% relapse after using isotretinoin, Dr. Murase, director of medical dermatology consultative services and patch testing at the Palo Alto Foundation Medical Group, said at the annual meeting of the Pacific Dermatologic Association.
In her clinical experience, hormonal therapy is a safe long-term option for recalcitrant acne in postmenarcheal females over the age of 14. “Although oral antibiotics are going to be superior to hormonal therapy in the first month or two, when you get to about six months, they have equivalent efficacy,” she said.
Telltale signs of acne associated with androgen excess include the development of nodulocystic papules along the jawline and small comedones over the forehead. Female patients with acne may request that labs be ordered to check their hormone levels, but that often is not necessary, according to Dr. Murase, who is also associate clinical professor of dermatology at the University of California, San Francisco. “There aren’t strict guidelines to indicate when you should perform hormonal testing, but warning signs that warrant further evaluation include hirsutism, androgenetic alopecia, virilization, infertility, oligomenorrhea or amenorrhea, and sudden onset of severe acne. The most common situation that warrants hormonal testing is polycystic ovary syndrome (PCOS).”
When there is a strong suspicion for hyperandrogenism, essential labs include free and total testosterone. Free testosterone is commonly elevated in patients with PCOS and total testosterone levels over 200 ng/dL is suggestive of an ovarian tumor. Other essential labs include 17-hyydroxyprogesterone (values greater than 200 ng/dL indicate congenital adrenal hyperplasia), and dehydroepiandrosterone sulfate (DHEA-S); levels over 8,000 mcg/dL indicate an adrenal tumor, while levels in the 4,000-8,000 mcg/dL range indicate congenital adrenal hyperplasia.
Helpful lab tests to consider include the ratio of luteinizing hormone to follicle-stimulating hormone; a 3:1 ratio or greater is suggestive for PCOS. “Ordering a prolactin level can also help, especially if patients are describing issues with headaches, which could indicate a pituitary tumor,” Dr. Murase added. Measuring sex hormone binding globulin (SHBG) levels can also be helpful. “If a patient has been on oral contraceptives for a long time, it increases their SHBG,” which, in older women, she said, “is inversely related to the development of type 2 diabetes.”
All labs for hyperandrogenism should be performed early in the morning on day 3 of the patient’s menstrual cycle. “If patients are on some kind of hormonal therapy, they need to be off of it for at least 6 weeks in order for you get a relevant test,” she said. Other relevant labs to consider include fasting glucose and lipids, cortisol, and thyroid-stimulating hormone.
Oral contraceptives
Estrogen contained in oral contraceptives (OCs) provides the most benefit to acne patients. “It reduces sebum production, decreases free testosterone and DHEA-S by stimulating SHBG synthesis in the liver, inhibits 5-alpha-reductase, which decreases peripheral testosterone conversion, and it decreases the production of ovarian and adrenal androgens,” Dr. Murase explained. “On average, you can get about 40%-70% reduction of lesion count, which is pretty good.”
Progestins with low androgenetic activity are the most helpful for acne, including norgestimate, desogestrel, and drospirenone. FDA-approved OC options include Ortho Tri-Cyclen, EstroStep, Yaz, and Beyaz. None has data showing superior efficacy.
No Pap smear or pelvic exam is required when prescribing OCs, but the risk of clotting should be discussed with patients. According to Dr. Murase, the risk of deep vein thrombosis (DVT) at baseline is about 1 per 10,000 woman-years, while the risk of DVT after 1 year on an OC is 3.4 per 10,000 years.
“This is a very mild increased risk that we’re talking about, but it is relevant in smokers, in those with hypertension, and in those who are diabetic,” she said. As for the risk of cancer associated with the use of OCs, a large collaborative study found a relative risk of 1.24 for developing breast cancer (not dose or duration related), but a risk reduction for endometrial, colorectal, and ovarian cancer.
The most common side effects associated with OCs are unscheduled bleeding, nausea, breast tenderness, and possible weight gain. Concomitant antibiotics can be used, with the exception of CYP3A4 inducers, such as rifampin. “That’s the main antibiotic we have to worry about that could affect the efficacy of the birth control pill,” she said. “It accounts for about three-quarters of pregnancies on antibiotics.”
Tetracyclines do not appear to increase the rate of birth defects with incidental first-trimester exposure, and data are reassuring but “tetracycline should be stopped within the first trimester as soon as the patient discovers she is pregnant,” Dr. Murase said.
Contraindications for OCs include being pregnant or breastfeeding; history of stroke, venous thromboembolism, or MI; history of smoking and being over age 35; uncontrolled hypertension; migraines with focal symptoms/aura; current or past breast cancer; hypercholesterolemia; diabetes with end-organ damage or having diabetes over age 35; liver issues such as a tumor, viral hepatitis, or cirrhosis; and a history of major surgery with prolonged immobilization.
Spironolactone
Another treatment option is spironolactone, a potassium-sparing diuretic that blocks aldosterone at a dose of 25 mg/day. At doses of 50-100 mg/day, it blocks androgen. “It can be used in combination with an oral contraceptive, with the rates of efficacy reported to range between 33% and 85%,” Dr. Murase said.
Spironolactone can also reduce hirsutism, improve androgenetic alopecia, and lower blood pressure by about 5 mm Hg systolic and 2.5 mm Hg diastolic. Dr. Murase usually checks blood pressure in patients, and “only if they’re really low I’ll talk about the potential for postural hypotension and the fact that you can get a little bit dizzy when going from a position of lying down to standing up.” Potassium levels should be checked at baseline and 4 weeks in patients older than age 46, in those with cardiac and/or renal disease, or in those on concomitant drospirenone or a third-generation progestin.
Spironolactone is classified as a pregnancy category D drug that could compromise the genital development of a male fetus. “So the onus is on us as providers to have the conversation with our patient,” she said. “If you’re putting a patient on spironolactone and they are of child-bearing age, you need to make sure that you’ve had the conversation with them about the fact that they should not get pregnant while on the medicine.”
Spironolactone also has a boxed warning citing the development of benign tumors in animal studies. That warning is based on studies in rats at doses of 10-150 mg/kg per day, “which is an extremely high dose and would never be given in humans,” said Dr. Murase, who has coauthored CME content regarding the safety of dermatologic medications in pregnancy and lactation.
In humans, there has been no evidence of the development of benign tumors associated with spironolactone therapy, and “there has been a decreased risk of prostate cancer and no association with its use and the development of breast, ovarian, bladder, kidney, gastric, or esophageal cancer,” she said.
Dr. Murase noted that during pregnancy, first-line oral antibiotics include amoxicillin for acne rosacea and cefadroxil for acne vulgaris. Macrolides are a second-line choice because of an increase in atrial/ventricular septal defects and pyloric stenosis that have been reported with first-trimester exposure.
“Erythromycin is the preferred choice over azithromycin and clarithromycin because it has the most data, [but] erythromycin estolate has been associated with increased AST levels in the second trimester,” she said. “It occurs in about 10% of cases and is reversible. Erythromycin base and erythromycin ethylsuccinate do not have this risk, and those are preferable.”
Dr. Murase disclosed that she has been a paid speaker of unbranded medical content for Regeneron and UCB. She is also a member of the advisory board for Leo Pharma, Eli Lilly, UCB, and Genzyme/Sanofi.
PORTLAND, ORE. – During her dermatology residency training at the University of California, Irvine, Medical Center, Jenny Murase, MD, remembers hearing a colleague say that her most angry patients of the day were adult women with recalcitrant acne who present to the clinic with questions like, “My skin has been clear my whole life! What’s going on?”
Such . In fact, 82% fail multiple courses of systemic antibiotics and 32% relapse after using isotretinoin, Dr. Murase, director of medical dermatology consultative services and patch testing at the Palo Alto Foundation Medical Group, said at the annual meeting of the Pacific Dermatologic Association.
In her clinical experience, hormonal therapy is a safe long-term option for recalcitrant acne in postmenarcheal females over the age of 14. “Although oral antibiotics are going to be superior to hormonal therapy in the first month or two, when you get to about six months, they have equivalent efficacy,” she said.
Telltale signs of acne associated with androgen excess include the development of nodulocystic papules along the jawline and small comedones over the forehead. Female patients with acne may request that labs be ordered to check their hormone levels, but that often is not necessary, according to Dr. Murase, who is also associate clinical professor of dermatology at the University of California, San Francisco. “There aren’t strict guidelines to indicate when you should perform hormonal testing, but warning signs that warrant further evaluation include hirsutism, androgenetic alopecia, virilization, infertility, oligomenorrhea or amenorrhea, and sudden onset of severe acne. The most common situation that warrants hormonal testing is polycystic ovary syndrome (PCOS).”
When there is a strong suspicion for hyperandrogenism, essential labs include free and total testosterone. Free testosterone is commonly elevated in patients with PCOS and total testosterone levels over 200 ng/dL is suggestive of an ovarian tumor. Other essential labs include 17-hyydroxyprogesterone (values greater than 200 ng/dL indicate congenital adrenal hyperplasia), and dehydroepiandrosterone sulfate (DHEA-S); levels over 8,000 mcg/dL indicate an adrenal tumor, while levels in the 4,000-8,000 mcg/dL range indicate congenital adrenal hyperplasia.
Helpful lab tests to consider include the ratio of luteinizing hormone to follicle-stimulating hormone; a 3:1 ratio or greater is suggestive for PCOS. “Ordering a prolactin level can also help, especially if patients are describing issues with headaches, which could indicate a pituitary tumor,” Dr. Murase added. Measuring sex hormone binding globulin (SHBG) levels can also be helpful. “If a patient has been on oral contraceptives for a long time, it increases their SHBG,” which, in older women, she said, “is inversely related to the development of type 2 diabetes.”
All labs for hyperandrogenism should be performed early in the morning on day 3 of the patient’s menstrual cycle. “If patients are on some kind of hormonal therapy, they need to be off of it for at least 6 weeks in order for you get a relevant test,” she said. Other relevant labs to consider include fasting glucose and lipids, cortisol, and thyroid-stimulating hormone.
Oral contraceptives
Estrogen contained in oral contraceptives (OCs) provides the most benefit to acne patients. “It reduces sebum production, decreases free testosterone and DHEA-S by stimulating SHBG synthesis in the liver, inhibits 5-alpha-reductase, which decreases peripheral testosterone conversion, and it decreases the production of ovarian and adrenal androgens,” Dr. Murase explained. “On average, you can get about 40%-70% reduction of lesion count, which is pretty good.”
Progestins with low androgenetic activity are the most helpful for acne, including norgestimate, desogestrel, and drospirenone. FDA-approved OC options include Ortho Tri-Cyclen, EstroStep, Yaz, and Beyaz. None has data showing superior efficacy.
No Pap smear or pelvic exam is required when prescribing OCs, but the risk of clotting should be discussed with patients. According to Dr. Murase, the risk of deep vein thrombosis (DVT) at baseline is about 1 per 10,000 woman-years, while the risk of DVT after 1 year on an OC is 3.4 per 10,000 years.
“This is a very mild increased risk that we’re talking about, but it is relevant in smokers, in those with hypertension, and in those who are diabetic,” she said. As for the risk of cancer associated with the use of OCs, a large collaborative study found a relative risk of 1.24 for developing breast cancer (not dose or duration related), but a risk reduction for endometrial, colorectal, and ovarian cancer.
The most common side effects associated with OCs are unscheduled bleeding, nausea, breast tenderness, and possible weight gain. Concomitant antibiotics can be used, with the exception of CYP3A4 inducers, such as rifampin. “That’s the main antibiotic we have to worry about that could affect the efficacy of the birth control pill,” she said. “It accounts for about three-quarters of pregnancies on antibiotics.”
Tetracyclines do not appear to increase the rate of birth defects with incidental first-trimester exposure, and data are reassuring but “tetracycline should be stopped within the first trimester as soon as the patient discovers she is pregnant,” Dr. Murase said.
Contraindications for OCs include being pregnant or breastfeeding; history of stroke, venous thromboembolism, or MI; history of smoking and being over age 35; uncontrolled hypertension; migraines with focal symptoms/aura; current or past breast cancer; hypercholesterolemia; diabetes with end-organ damage or having diabetes over age 35; liver issues such as a tumor, viral hepatitis, or cirrhosis; and a history of major surgery with prolonged immobilization.
Spironolactone
Another treatment option is spironolactone, a potassium-sparing diuretic that blocks aldosterone at a dose of 25 mg/day. At doses of 50-100 mg/day, it blocks androgen. “It can be used in combination with an oral contraceptive, with the rates of efficacy reported to range between 33% and 85%,” Dr. Murase said.
Spironolactone can also reduce hirsutism, improve androgenetic alopecia, and lower blood pressure by about 5 mm Hg systolic and 2.5 mm Hg diastolic. Dr. Murase usually checks blood pressure in patients, and “only if they’re really low I’ll talk about the potential for postural hypotension and the fact that you can get a little bit dizzy when going from a position of lying down to standing up.” Potassium levels should be checked at baseline and 4 weeks in patients older than age 46, in those with cardiac and/or renal disease, or in those on concomitant drospirenone or a third-generation progestin.
Spironolactone is classified as a pregnancy category D drug that could compromise the genital development of a male fetus. “So the onus is on us as providers to have the conversation with our patient,” she said. “If you’re putting a patient on spironolactone and they are of child-bearing age, you need to make sure that you’ve had the conversation with them about the fact that they should not get pregnant while on the medicine.”
Spironolactone also has a boxed warning citing the development of benign tumors in animal studies. That warning is based on studies in rats at doses of 10-150 mg/kg per day, “which is an extremely high dose and would never be given in humans,” said Dr. Murase, who has coauthored CME content regarding the safety of dermatologic medications in pregnancy and lactation.
In humans, there has been no evidence of the development of benign tumors associated with spironolactone therapy, and “there has been a decreased risk of prostate cancer and no association with its use and the development of breast, ovarian, bladder, kidney, gastric, or esophageal cancer,” she said.
Dr. Murase noted that during pregnancy, first-line oral antibiotics include amoxicillin for acne rosacea and cefadroxil for acne vulgaris. Macrolides are a second-line choice because of an increase in atrial/ventricular septal defects and pyloric stenosis that have been reported with first-trimester exposure.
“Erythromycin is the preferred choice over azithromycin and clarithromycin because it has the most data, [but] erythromycin estolate has been associated with increased AST levels in the second trimester,” she said. “It occurs in about 10% of cases and is reversible. Erythromycin base and erythromycin ethylsuccinate do not have this risk, and those are preferable.”
Dr. Murase disclosed that she has been a paid speaker of unbranded medical content for Regeneron and UCB. She is also a member of the advisory board for Leo Pharma, Eli Lilly, UCB, and Genzyme/Sanofi.
PORTLAND, ORE. – During her dermatology residency training at the University of California, Irvine, Medical Center, Jenny Murase, MD, remembers hearing a colleague say that her most angry patients of the day were adult women with recalcitrant acne who present to the clinic with questions like, “My skin has been clear my whole life! What’s going on?”
Such . In fact, 82% fail multiple courses of systemic antibiotics and 32% relapse after using isotretinoin, Dr. Murase, director of medical dermatology consultative services and patch testing at the Palo Alto Foundation Medical Group, said at the annual meeting of the Pacific Dermatologic Association.
In her clinical experience, hormonal therapy is a safe long-term option for recalcitrant acne in postmenarcheal females over the age of 14. “Although oral antibiotics are going to be superior to hormonal therapy in the first month or two, when you get to about six months, they have equivalent efficacy,” she said.
Telltale signs of acne associated with androgen excess include the development of nodulocystic papules along the jawline and small comedones over the forehead. Female patients with acne may request that labs be ordered to check their hormone levels, but that often is not necessary, according to Dr. Murase, who is also associate clinical professor of dermatology at the University of California, San Francisco. “There aren’t strict guidelines to indicate when you should perform hormonal testing, but warning signs that warrant further evaluation include hirsutism, androgenetic alopecia, virilization, infertility, oligomenorrhea or amenorrhea, and sudden onset of severe acne. The most common situation that warrants hormonal testing is polycystic ovary syndrome (PCOS).”
When there is a strong suspicion for hyperandrogenism, essential labs include free and total testosterone. Free testosterone is commonly elevated in patients with PCOS and total testosterone levels over 200 ng/dL is suggestive of an ovarian tumor. Other essential labs include 17-hyydroxyprogesterone (values greater than 200 ng/dL indicate congenital adrenal hyperplasia), and dehydroepiandrosterone sulfate (DHEA-S); levels over 8,000 mcg/dL indicate an adrenal tumor, while levels in the 4,000-8,000 mcg/dL range indicate congenital adrenal hyperplasia.
Helpful lab tests to consider include the ratio of luteinizing hormone to follicle-stimulating hormone; a 3:1 ratio or greater is suggestive for PCOS. “Ordering a prolactin level can also help, especially if patients are describing issues with headaches, which could indicate a pituitary tumor,” Dr. Murase added. Measuring sex hormone binding globulin (SHBG) levels can also be helpful. “If a patient has been on oral contraceptives for a long time, it increases their SHBG,” which, in older women, she said, “is inversely related to the development of type 2 diabetes.”
All labs for hyperandrogenism should be performed early in the morning on day 3 of the patient’s menstrual cycle. “If patients are on some kind of hormonal therapy, they need to be off of it for at least 6 weeks in order for you get a relevant test,” she said. Other relevant labs to consider include fasting glucose and lipids, cortisol, and thyroid-stimulating hormone.
Oral contraceptives
Estrogen contained in oral contraceptives (OCs) provides the most benefit to acne patients. “It reduces sebum production, decreases free testosterone and DHEA-S by stimulating SHBG synthesis in the liver, inhibits 5-alpha-reductase, which decreases peripheral testosterone conversion, and it decreases the production of ovarian and adrenal androgens,” Dr. Murase explained. “On average, you can get about 40%-70% reduction of lesion count, which is pretty good.”
Progestins with low androgenetic activity are the most helpful for acne, including norgestimate, desogestrel, and drospirenone. FDA-approved OC options include Ortho Tri-Cyclen, EstroStep, Yaz, and Beyaz. None has data showing superior efficacy.
No Pap smear or pelvic exam is required when prescribing OCs, but the risk of clotting should be discussed with patients. According to Dr. Murase, the risk of deep vein thrombosis (DVT) at baseline is about 1 per 10,000 woman-years, while the risk of DVT after 1 year on an OC is 3.4 per 10,000 years.
“This is a very mild increased risk that we’re talking about, but it is relevant in smokers, in those with hypertension, and in those who are diabetic,” she said. As for the risk of cancer associated with the use of OCs, a large collaborative study found a relative risk of 1.24 for developing breast cancer (not dose or duration related), but a risk reduction for endometrial, colorectal, and ovarian cancer.
The most common side effects associated with OCs are unscheduled bleeding, nausea, breast tenderness, and possible weight gain. Concomitant antibiotics can be used, with the exception of CYP3A4 inducers, such as rifampin. “That’s the main antibiotic we have to worry about that could affect the efficacy of the birth control pill,” she said. “It accounts for about three-quarters of pregnancies on antibiotics.”
Tetracyclines do not appear to increase the rate of birth defects with incidental first-trimester exposure, and data are reassuring but “tetracycline should be stopped within the first trimester as soon as the patient discovers she is pregnant,” Dr. Murase said.
Contraindications for OCs include being pregnant or breastfeeding; history of stroke, venous thromboembolism, or MI; history of smoking and being over age 35; uncontrolled hypertension; migraines with focal symptoms/aura; current or past breast cancer; hypercholesterolemia; diabetes with end-organ damage or having diabetes over age 35; liver issues such as a tumor, viral hepatitis, or cirrhosis; and a history of major surgery with prolonged immobilization.
Spironolactone
Another treatment option is spironolactone, a potassium-sparing diuretic that blocks aldosterone at a dose of 25 mg/day. At doses of 50-100 mg/day, it blocks androgen. “It can be used in combination with an oral contraceptive, with the rates of efficacy reported to range between 33% and 85%,” Dr. Murase said.
Spironolactone can also reduce hirsutism, improve androgenetic alopecia, and lower blood pressure by about 5 mm Hg systolic and 2.5 mm Hg diastolic. Dr. Murase usually checks blood pressure in patients, and “only if they’re really low I’ll talk about the potential for postural hypotension and the fact that you can get a little bit dizzy when going from a position of lying down to standing up.” Potassium levels should be checked at baseline and 4 weeks in patients older than age 46, in those with cardiac and/or renal disease, or in those on concomitant drospirenone or a third-generation progestin.
Spironolactone is classified as a pregnancy category D drug that could compromise the genital development of a male fetus. “So the onus is on us as providers to have the conversation with our patient,” she said. “If you’re putting a patient on spironolactone and they are of child-bearing age, you need to make sure that you’ve had the conversation with them about the fact that they should not get pregnant while on the medicine.”
Spironolactone also has a boxed warning citing the development of benign tumors in animal studies. That warning is based on studies in rats at doses of 10-150 mg/kg per day, “which is an extremely high dose and would never be given in humans,” said Dr. Murase, who has coauthored CME content regarding the safety of dermatologic medications in pregnancy and lactation.
In humans, there has been no evidence of the development of benign tumors associated with spironolactone therapy, and “there has been a decreased risk of prostate cancer and no association with its use and the development of breast, ovarian, bladder, kidney, gastric, or esophageal cancer,” she said.
Dr. Murase noted that during pregnancy, first-line oral antibiotics include amoxicillin for acne rosacea and cefadroxil for acne vulgaris. Macrolides are a second-line choice because of an increase in atrial/ventricular septal defects and pyloric stenosis that have been reported with first-trimester exposure.
“Erythromycin is the preferred choice over azithromycin and clarithromycin because it has the most data, [but] erythromycin estolate has been associated with increased AST levels in the second trimester,” she said. “It occurs in about 10% of cases and is reversible. Erythromycin base and erythromycin ethylsuccinate do not have this risk, and those are preferable.”
Dr. Murase disclosed that she has been a paid speaker of unbranded medical content for Regeneron and UCB. She is also a member of the advisory board for Leo Pharma, Eli Lilly, UCB, and Genzyme/Sanofi.
AT PDA 2022
Inhaled, systemic steroids linked to changes in brain structure
New research links the use of glucocorticoids with changes in white matter microstructure – which may explain the development of anxiety, depression, and other neuropsychiatric side effects related to these drugs, investigators say.
Results from a cross-sectional study showed that use of both systemic and inhaled glucocorticoids was associated with widespread reductions in fractional anisotropy (FA) and increases in mean diffusivity.
Glucocorticoids have “a whole catalogue” of adverse events, and effects on brain structure “adds to the list,” co-investigator Onno C. Meijer, PhD, professor of molecular neuroendocrinology of corticosteroids, department of medicine, Leiden University Medical Center, the Netherlands, told this news organization.
The findings should encourage clinicians to consider whether doses they are prescribing are too high, said Dr. Meijer. He added that the negative effect of glucocorticoids on the brain was also found in those using inhalers, such as patients with asthma.
The findings were published online in the BMJ Open.
Serious side effects
Glucocorticoids, a class of synthetic steroids with immunosuppressive properties, are prescribed for a wide range of conditions, including rheumatoid arthritis and asthma.
However, they are also associated with potentially serious metabolic, cardiovascular, and musculoskeletal side effects as well as neuropsychiatric side effects such as depression, mania, and cognitive impairment.
About 1 in 3 patients exposed to “quite a lot of these drugs” will experience neuropsychiatric symptoms, Dr. Meijer said.
Most previous studies that investigated effects from high levels of glucocorticoids on brain structure have been small and involved selected populations, such as those with Cushing disease.
The new study included participants from the UK Biobank, a large population-based cohort. Participants had undergone imaging and did not have a history of psychiatric disease – although they could have conditions associated with glucocorticoid use, including anxiety, depression, mania, or delirium.
The analysis included 222 patients using oral or parenteral glucocorticoids at the time of imaging (systemic group), 557 using inhaled glucocorticoids, and 24,106 not using glucocorticoids (the control group).
Inhaled steroids target the lungs, whereas a steroid in pill form “travels in the blood and reaches each and every organ and cell in the body and typically requires higher doses,” Dr. Meijer noted.
The groups were similar with respect to sex, education, and smoking status. However, the systemic glucocorticoid group was slightly older (mean age, 66.1 years vs. 63.3 years for inhaled glucocorticoid users and 63.5 years for the control group).
In addition to age, researchers adjusted for sex, education level, head position in the scanner, head size, assessment center, and year of imaging.
Imaging analyses
Imaging analyses showed systemic glucocorticoid use was associated with reduced global FA (adjusted mean difference, -3.7e-3; 95% confidence interval, -6.4e-3 to 1.0e-3), and reductions in regional FA in the body and genu of the corpus callosum versus the control group.
Inhaled glucocorticoid use was associated with reduced global FA (AMD, -2.3e-3; 95% CI, -4.0e-3 to -5.7e-4), and lower FA in the splenium of the corpus callosum and the cingulum of the hippocampus.
Global mean diffusivity was higher in systemic glucocorticoid users (AMD, 7.2e-6; 95% CI, 3.2e-6 to 1.1e-5) and inhaled glucocorticoid users (AMD, 2.7e-6; 95% CI, 1.7e-7 to 5.2e-6), compared with the control group.
The effects of glucocorticoids on white matter were “pervasive,” and the “most important finding” of the study, Dr. Meijer said. “We were impressed by the fact white matter is so sensitive to these drugs.”
He noted that it is likely that functional connectivity between brain regions is affected by use of glucocorticoids. “You could say communication between brain regions is probably somewhat impaired or challenged,” he said.
Subgroup analyses among participants using glucocorticoids chronically, defined as reported at two consecutive visits, suggested a potential dose-dependent or duration-dependent effect of glucocorticoids on white matter microstructure.
Systemic glucocorticoid use was also associated with an increase in total and grey matter volume of the caudate nucleus.
In addition, there was a significant association between inhaled glucocorticoid use and decreased grey matter volume of the amygdala, which Dr. Meijer said was surprising because studies have shown that glucocorticoids “can drive amygdala big time.”
Move away from ‘one dose for all’?
Another surprise was that the results showed no hippocampal volume differences with steroid use, Dr. Meijer noted.
The modest association between glucocorticoid use and brain volumes could indicate that white matter integrity is more sensitive to glucocorticoids than is grey matter volume, “at least at the structural level,” he said.
He added that longer use or higher doses may be necessary to also induce volumetric changes.
Participants also completed a questionnaire to assess mood over the previous 2 weeks. Systemic glucocorticoid users had more depressive symptoms, disinterest, tenseness/restlessness, and tiredness/lethargy, compared with the control group. Inhaled glucocorticoid users only reported more tiredness/lethargy.
The investigators note that mood-related effects could be linked to the condition for which glucocorticoids were prescribed: for example, rheumatoid arthritis or chronic obstructive pulmonary disease.
In terms of cognition, systemic glucocorticoid users performed significantly worse on the symbol digit substitution task, compared with participants in the control group.
In light of these findings, pharmaceutical companies that make inhaled corticosteroids “should perhaps find out if glucocorticoids can be dosed by kilogram body weight rather than simply one dose fits all,” which is currently the case, Dr. Meijer said.
Impressive, but several limitations
Commenting on the findings, E. Sherwood Brown, MD, PhD, Distinguished Chair in Psychiatric Research and professor and vice chair for clinical research, department of psychiatry, The University of Texas Southwestern Medical Center, Dallas, called the study sample size “impressive.”
In addition, the study is the first to look at systemic as well as inhaled corticosteroids, said Dr. Brown, who was not involved with the research. He noted that previously, there had been only case reports of psychiatric symptoms with inhaled corticosteroids.
That results are in the same direction but greater with systemic, compared with inhaled corticosteroids, is “particularly interesting” because this might suggest dose-dependent effects, Dr. Brown said.
He noted that cognitive differences were also only observed with systemic corticosteroids.
Some study observations, such as smaller amygdala volume with inhaled but not systemic corticosteroids, “are harder to understand,” said Dr. Brown.
However, he pointed out some study limitations. For example, data were apparently unavailable for verbal and declarative memory test data, despite corticosteroids probably affecting the hippocampus and causing memory changes.
Other drawbacks were that the dose and duration of corticosteroid use, as well as the medical histories of study participants, were not available, Dr. Brown said.
No study funding was reported. Dr. Meijer has received research grants and honorariums from Corcept Therapeutics and a speakers’ fee from Ipsen. Dr. Brown is on an advisory board for Sage Pharmaceuticals, which is developing neurosteroids (not corticosteroids) for mood disorders. He is also on a Medscape advisory board related to bipolar disorder.
A version of this article first appeared on Medscape.com.
New research links the use of glucocorticoids with changes in white matter microstructure – which may explain the development of anxiety, depression, and other neuropsychiatric side effects related to these drugs, investigators say.
Results from a cross-sectional study showed that use of both systemic and inhaled glucocorticoids was associated with widespread reductions in fractional anisotropy (FA) and increases in mean diffusivity.
Glucocorticoids have “a whole catalogue” of adverse events, and effects on brain structure “adds to the list,” co-investigator Onno C. Meijer, PhD, professor of molecular neuroendocrinology of corticosteroids, department of medicine, Leiden University Medical Center, the Netherlands, told this news organization.
The findings should encourage clinicians to consider whether doses they are prescribing are too high, said Dr. Meijer. He added that the negative effect of glucocorticoids on the brain was also found in those using inhalers, such as patients with asthma.
The findings were published online in the BMJ Open.
Serious side effects
Glucocorticoids, a class of synthetic steroids with immunosuppressive properties, are prescribed for a wide range of conditions, including rheumatoid arthritis and asthma.
However, they are also associated with potentially serious metabolic, cardiovascular, and musculoskeletal side effects as well as neuropsychiatric side effects such as depression, mania, and cognitive impairment.
About 1 in 3 patients exposed to “quite a lot of these drugs” will experience neuropsychiatric symptoms, Dr. Meijer said.
Most previous studies that investigated effects from high levels of glucocorticoids on brain structure have been small and involved selected populations, such as those with Cushing disease.
The new study included participants from the UK Biobank, a large population-based cohort. Participants had undergone imaging and did not have a history of psychiatric disease – although they could have conditions associated with glucocorticoid use, including anxiety, depression, mania, or delirium.
The analysis included 222 patients using oral or parenteral glucocorticoids at the time of imaging (systemic group), 557 using inhaled glucocorticoids, and 24,106 not using glucocorticoids (the control group).
Inhaled steroids target the lungs, whereas a steroid in pill form “travels in the blood and reaches each and every organ and cell in the body and typically requires higher doses,” Dr. Meijer noted.
The groups were similar with respect to sex, education, and smoking status. However, the systemic glucocorticoid group was slightly older (mean age, 66.1 years vs. 63.3 years for inhaled glucocorticoid users and 63.5 years for the control group).
In addition to age, researchers adjusted for sex, education level, head position in the scanner, head size, assessment center, and year of imaging.
Imaging analyses
Imaging analyses showed systemic glucocorticoid use was associated with reduced global FA (adjusted mean difference, -3.7e-3; 95% confidence interval, -6.4e-3 to 1.0e-3), and reductions in regional FA in the body and genu of the corpus callosum versus the control group.
Inhaled glucocorticoid use was associated with reduced global FA (AMD, -2.3e-3; 95% CI, -4.0e-3 to -5.7e-4), and lower FA in the splenium of the corpus callosum and the cingulum of the hippocampus.
Global mean diffusivity was higher in systemic glucocorticoid users (AMD, 7.2e-6; 95% CI, 3.2e-6 to 1.1e-5) and inhaled glucocorticoid users (AMD, 2.7e-6; 95% CI, 1.7e-7 to 5.2e-6), compared with the control group.
The effects of glucocorticoids on white matter were “pervasive,” and the “most important finding” of the study, Dr. Meijer said. “We were impressed by the fact white matter is so sensitive to these drugs.”
He noted that it is likely that functional connectivity between brain regions is affected by use of glucocorticoids. “You could say communication between brain regions is probably somewhat impaired or challenged,” he said.
Subgroup analyses among participants using glucocorticoids chronically, defined as reported at two consecutive visits, suggested a potential dose-dependent or duration-dependent effect of glucocorticoids on white matter microstructure.
Systemic glucocorticoid use was also associated with an increase in total and grey matter volume of the caudate nucleus.
In addition, there was a significant association between inhaled glucocorticoid use and decreased grey matter volume of the amygdala, which Dr. Meijer said was surprising because studies have shown that glucocorticoids “can drive amygdala big time.”
Move away from ‘one dose for all’?
Another surprise was that the results showed no hippocampal volume differences with steroid use, Dr. Meijer noted.
The modest association between glucocorticoid use and brain volumes could indicate that white matter integrity is more sensitive to glucocorticoids than is grey matter volume, “at least at the structural level,” he said.
He added that longer use or higher doses may be necessary to also induce volumetric changes.
Participants also completed a questionnaire to assess mood over the previous 2 weeks. Systemic glucocorticoid users had more depressive symptoms, disinterest, tenseness/restlessness, and tiredness/lethargy, compared with the control group. Inhaled glucocorticoid users only reported more tiredness/lethargy.
The investigators note that mood-related effects could be linked to the condition for which glucocorticoids were prescribed: for example, rheumatoid arthritis or chronic obstructive pulmonary disease.
In terms of cognition, systemic glucocorticoid users performed significantly worse on the symbol digit substitution task, compared with participants in the control group.
In light of these findings, pharmaceutical companies that make inhaled corticosteroids “should perhaps find out if glucocorticoids can be dosed by kilogram body weight rather than simply one dose fits all,” which is currently the case, Dr. Meijer said.
Impressive, but several limitations
Commenting on the findings, E. Sherwood Brown, MD, PhD, Distinguished Chair in Psychiatric Research and professor and vice chair for clinical research, department of psychiatry, The University of Texas Southwestern Medical Center, Dallas, called the study sample size “impressive.”
In addition, the study is the first to look at systemic as well as inhaled corticosteroids, said Dr. Brown, who was not involved with the research. He noted that previously, there had been only case reports of psychiatric symptoms with inhaled corticosteroids.
That results are in the same direction but greater with systemic, compared with inhaled corticosteroids, is “particularly interesting” because this might suggest dose-dependent effects, Dr. Brown said.
He noted that cognitive differences were also only observed with systemic corticosteroids.
Some study observations, such as smaller amygdala volume with inhaled but not systemic corticosteroids, “are harder to understand,” said Dr. Brown.
However, he pointed out some study limitations. For example, data were apparently unavailable for verbal and declarative memory test data, despite corticosteroids probably affecting the hippocampus and causing memory changes.
Other drawbacks were that the dose and duration of corticosteroid use, as well as the medical histories of study participants, were not available, Dr. Brown said.
No study funding was reported. Dr. Meijer has received research grants and honorariums from Corcept Therapeutics and a speakers’ fee from Ipsen. Dr. Brown is on an advisory board for Sage Pharmaceuticals, which is developing neurosteroids (not corticosteroids) for mood disorders. He is also on a Medscape advisory board related to bipolar disorder.
A version of this article first appeared on Medscape.com.
New research links the use of glucocorticoids with changes in white matter microstructure – which may explain the development of anxiety, depression, and other neuropsychiatric side effects related to these drugs, investigators say.
Results from a cross-sectional study showed that use of both systemic and inhaled glucocorticoids was associated with widespread reductions in fractional anisotropy (FA) and increases in mean diffusivity.
Glucocorticoids have “a whole catalogue” of adverse events, and effects on brain structure “adds to the list,” co-investigator Onno C. Meijer, PhD, professor of molecular neuroendocrinology of corticosteroids, department of medicine, Leiden University Medical Center, the Netherlands, told this news organization.
The findings should encourage clinicians to consider whether doses they are prescribing are too high, said Dr. Meijer. He added that the negative effect of glucocorticoids on the brain was also found in those using inhalers, such as patients with asthma.
The findings were published online in the BMJ Open.
Serious side effects
Glucocorticoids, a class of synthetic steroids with immunosuppressive properties, are prescribed for a wide range of conditions, including rheumatoid arthritis and asthma.
However, they are also associated with potentially serious metabolic, cardiovascular, and musculoskeletal side effects as well as neuropsychiatric side effects such as depression, mania, and cognitive impairment.
About 1 in 3 patients exposed to “quite a lot of these drugs” will experience neuropsychiatric symptoms, Dr. Meijer said.
Most previous studies that investigated effects from high levels of glucocorticoids on brain structure have been small and involved selected populations, such as those with Cushing disease.
The new study included participants from the UK Biobank, a large population-based cohort. Participants had undergone imaging and did not have a history of psychiatric disease – although they could have conditions associated with glucocorticoid use, including anxiety, depression, mania, or delirium.
The analysis included 222 patients using oral or parenteral glucocorticoids at the time of imaging (systemic group), 557 using inhaled glucocorticoids, and 24,106 not using glucocorticoids (the control group).
Inhaled steroids target the lungs, whereas a steroid in pill form “travels in the blood and reaches each and every organ and cell in the body and typically requires higher doses,” Dr. Meijer noted.
The groups were similar with respect to sex, education, and smoking status. However, the systemic glucocorticoid group was slightly older (mean age, 66.1 years vs. 63.3 years for inhaled glucocorticoid users and 63.5 years for the control group).
In addition to age, researchers adjusted for sex, education level, head position in the scanner, head size, assessment center, and year of imaging.
Imaging analyses
Imaging analyses showed systemic glucocorticoid use was associated with reduced global FA (adjusted mean difference, -3.7e-3; 95% confidence interval, -6.4e-3 to 1.0e-3), and reductions in regional FA in the body and genu of the corpus callosum versus the control group.
Inhaled glucocorticoid use was associated with reduced global FA (AMD, -2.3e-3; 95% CI, -4.0e-3 to -5.7e-4), and lower FA in the splenium of the corpus callosum and the cingulum of the hippocampus.
Global mean diffusivity was higher in systemic glucocorticoid users (AMD, 7.2e-6; 95% CI, 3.2e-6 to 1.1e-5) and inhaled glucocorticoid users (AMD, 2.7e-6; 95% CI, 1.7e-7 to 5.2e-6), compared with the control group.
The effects of glucocorticoids on white matter were “pervasive,” and the “most important finding” of the study, Dr. Meijer said. “We were impressed by the fact white matter is so sensitive to these drugs.”
He noted that it is likely that functional connectivity between brain regions is affected by use of glucocorticoids. “You could say communication between brain regions is probably somewhat impaired or challenged,” he said.
Subgroup analyses among participants using glucocorticoids chronically, defined as reported at two consecutive visits, suggested a potential dose-dependent or duration-dependent effect of glucocorticoids on white matter microstructure.
Systemic glucocorticoid use was also associated with an increase in total and grey matter volume of the caudate nucleus.
In addition, there was a significant association between inhaled glucocorticoid use and decreased grey matter volume of the amygdala, which Dr. Meijer said was surprising because studies have shown that glucocorticoids “can drive amygdala big time.”
Move away from ‘one dose for all’?
Another surprise was that the results showed no hippocampal volume differences with steroid use, Dr. Meijer noted.
The modest association between glucocorticoid use and brain volumes could indicate that white matter integrity is more sensitive to glucocorticoids than is grey matter volume, “at least at the structural level,” he said.
He added that longer use or higher doses may be necessary to also induce volumetric changes.
Participants also completed a questionnaire to assess mood over the previous 2 weeks. Systemic glucocorticoid users had more depressive symptoms, disinterest, tenseness/restlessness, and tiredness/lethargy, compared with the control group. Inhaled glucocorticoid users only reported more tiredness/lethargy.
The investigators note that mood-related effects could be linked to the condition for which glucocorticoids were prescribed: for example, rheumatoid arthritis or chronic obstructive pulmonary disease.
In terms of cognition, systemic glucocorticoid users performed significantly worse on the symbol digit substitution task, compared with participants in the control group.
In light of these findings, pharmaceutical companies that make inhaled corticosteroids “should perhaps find out if glucocorticoids can be dosed by kilogram body weight rather than simply one dose fits all,” which is currently the case, Dr. Meijer said.
Impressive, but several limitations
Commenting on the findings, E. Sherwood Brown, MD, PhD, Distinguished Chair in Psychiatric Research and professor and vice chair for clinical research, department of psychiatry, The University of Texas Southwestern Medical Center, Dallas, called the study sample size “impressive.”
In addition, the study is the first to look at systemic as well as inhaled corticosteroids, said Dr. Brown, who was not involved with the research. He noted that previously, there had been only case reports of psychiatric symptoms with inhaled corticosteroids.
That results are in the same direction but greater with systemic, compared with inhaled corticosteroids, is “particularly interesting” because this might suggest dose-dependent effects, Dr. Brown said.
He noted that cognitive differences were also only observed with systemic corticosteroids.
Some study observations, such as smaller amygdala volume with inhaled but not systemic corticosteroids, “are harder to understand,” said Dr. Brown.
However, he pointed out some study limitations. For example, data were apparently unavailable for verbal and declarative memory test data, despite corticosteroids probably affecting the hippocampus and causing memory changes.
Other drawbacks were that the dose and duration of corticosteroid use, as well as the medical histories of study participants, were not available, Dr. Brown said.
No study funding was reported. Dr. Meijer has received research grants and honorariums from Corcept Therapeutics and a speakers’ fee from Ipsen. Dr. Brown is on an advisory board for Sage Pharmaceuticals, which is developing neurosteroids (not corticosteroids) for mood disorders. He is also on a Medscape advisory board related to bipolar disorder.
A version of this article first appeared on Medscape.com.
FROM BMJ OPEN
Majority of muscle symptoms with statins not caused by treatment
In the vast majority of people who experience muscle pain or weakness while taking a statin, those symptoms are not related to the statin, a new individual patient data meta-analysis of randomized controlled trials shows.
The Cholesterol Trialists Collaboration meta-analysis examined 19 large randomized double-blind trials that compared statin therapy with placebo and involved almost 124,000 patients.
“Our results show that, in people who experience muscle symptoms in the first year of taking a statin, those symptoms are actually due to the statin in only 1 of 15 of those people. For the other 14 of the 15 people who experience muscle symptoms in the first year of taking a statin, that muscle pain is not due to the statin,” lead investigator Colin Baigent, MD, said.
After the first year, there was no difference in muscle symptoms between patients taking a statin or those taking placebo.
Dr. Baigent, who is director of the Population Health Research Unit at the University of Oxford (England), presented the data on Aug. 29 at the European Society of Cardiology 2022 Congress.
It was also simultaneously published online in The Lancet.
Dr. Baigent explained that statins very rarely cause serious muscle adverse effects with biochemical evidence of cellular damage, such as myopathy (which occurs in less than 1 in 10,000 patients per year) and rhabdomyolysis (which occurs in about 0.2 per 10,000 patients per year).
The effect of statins on other less serious muscle symptoms without biochemical evidence of cellular damage is less clear, but misinformation about the risks have arisen from nonrandomized studies, with social media and press reports suggesting that the risk for muscle symptoms with statins is extremely common, Dr. Baigent said.
In response to this, the Cholesterol Trialists Collaboration put together a new program of data collection, validation, and analysis to provide reliable information from large double-blind randomized trials that are free from bias and confounding.
“Overall, when we look at all these data, we find there is about a 3% relative increase in the risks of experiencing muscle pain or weakness with a statin versus with placebo,” Dr. Baigent reported.
Muscle pain or weakness was reported by 16,835 of 62,028 patients taking a statin, (27.1%), compared with 16,446 of 61,912 patients taking placebo (26.6%), for a rate ratio of 1.03 (95% confidence interval, 1.01-1.06).
In absolute terms, the results show a rate of 166 reports of muscle symptoms per 1,000 patient-years in those taking a statin, compared with 155 per 1,000-patient-years in those taking placebo in the first year. This gives a rate ratio of 1.07 and an excess of 11 cases of muscle pain or weakness per 1,000 patients in the first year of statin therapy.
“The very small excess of muscle symptoms in the statin patients were generally mild, with most patients able to continue treatment,” Dr. Baigent added.
After the first year, the rate of muscle pain or weakness was exactly the same in the statin and placebo groups, at 50 per 1,000 patient-years.
“Therefore, for the vast majority of people who experience muscle pain or weakness on a statin, those symptoms are not due to the statin itself. It is due to something else, which could be ageing, thyroid disease, or exercise,” Dr. Baigent said. “After the first year of taking a statin, there is no excess risk of muscle pain or weakness at all.”
“To summarize, the excess risk of muscle pain or weakness with statin use is tiny, and almost nonexistent after the first year,” he added.
“Muscle pain is very common in the general population, and it was very common in both patients taking a statin and those given placebo in these randomized trials. We can only detect a difference by looking at all the data combined in this enormous study. And we now know for sure that over 90% of cases of muscle symptoms experienced by people taking a statin are not due to the statin.”
The researchers also looked at statin intensity and found that the more intense statins tend to cause slightly more muscle pain. “There was also some evidence, although this was not very clear, that the muscle pain with the more intensive statins may persist for longer than 1 year,” Dr. Baigent said.
But in terms of different moderate-intensity and high-intensity statins, there was no evidence of differences in muscle pain between the individual statin brands, he added.
Better patient information needed
Dr. Baigent called for better information in statin package inserts about the real risk for muscle symptoms with these drugs.
“We need to do a better job of communicating the real risk of muscle symptom to patients who are taking statins and to their doctors. At the moment, doctors often stop statins if patients complain of muscle pain, but our data show that in 14 out of 15 times, they would be wrong for doing that. Stopping the statin is nearly always a mistake,” he commented.
“At present, the package inserts include a whole load of rubbish from observational studies, which are completely unreliable,” he added. “This is of no value to patients. They go through this information and find several symptoms they are experiencing, which they attribute to the drugs. We really need to divide up the information into the evidence that we really know for sure and then the more speculative stuff.”
Dr. Baigent also highlighted the large benefits of statins, compared with the small risk for muscle symptoms.
“While statins may cause 11 patients per 1,000 to experience some mild muscle pain in the first year of taking these drugs, and this was reduced to none in subsequent years, statins, when used for the primary prevention of cardiovascular disease, prevent 25 cardiovascular events per 1,000 patients every year they are taken. And for secondary prevention this rises to 50 events prevented per 1,000 patients each year,” he noted.
The individual participant data meta-analysis involved 23 trials with information on almost 155,000 patients. All trials included at least 1,000 patients and at least 2 years of scheduled treatment. Adverse-event data were collected for all individual participants in 19 large randomized double-blind trials comparing statin therapy with placebo (123,940 patients) and in four randomized double-blind trials comparing more-intensive with less-intensive statin therapy (30,724 patients).
In the four trials of more-intensive versus less-intensive statin therapy, high-intensity regimens (atorvastatin 40-80 mg daily or rosuvastatin 20-40 mg daily) resulted in a larger relative increase in the rate of muscle pain or weakness than moderate-intensity regimens, with rate ratios of 1.08 (95% CI, 1.04-1.13) and 1.02 (95% CI, 1.00-1.05), respectively.
‘Reassuring information’
Discussant of the study at the ESC Hotline session, Erin Bohula, MD, Brigham and Women’s Hospital, Boston, said this new analysis had many strengths and used a rigorous approach to look at the issue of muscle symptoms with statins.
She pointed out some challenges, including the fact that the definition of adverse muscle events has changed over time and differed in the various trials, with heterogeneous data capture across trials. “So, this was a Herculean task to harmonize this very complicated dataset.”
Dr. Bohula concluded: “I think this is a very significant undertaking, resulting in a rich dataset that enhances our understanding of muscle symptoms related to statin use. The take-home for me is that muscle symptoms are a common complaint in the general population but are very rarely attributable to statins. This is very reassuring to me, and I hope it is reassuring to patients and can help us encourage them with adherence, given the clear cardiovascular benefits of statins.”
Chair of the ESC Hotline session at which the study was presented, Gabriel Steg, MD, Hôpital Bichat, Paris, asked whether some statin patients who experienced muscle symptoms with the drugs in active run-in periods in the trials may have been excluded from the main trials, so that this information might not have been captured, but Dr. Baigent replied that they also examined those data, which had been accounted for in the analysis.
“That’s really good news,” Dr. Steg commented. “This study is going to be one more tool in our response to statin skeptics and I think, as such, this work is a really a service to public health.”
The meta-analysis was funded by the British Heart Foundation, the U.K. Medical Research Council, and the Australian National Health and Medical Research Council.
A version of this article first appeared on Medscape.com.
In the vast majority of people who experience muscle pain or weakness while taking a statin, those symptoms are not related to the statin, a new individual patient data meta-analysis of randomized controlled trials shows.
The Cholesterol Trialists Collaboration meta-analysis examined 19 large randomized double-blind trials that compared statin therapy with placebo and involved almost 124,000 patients.
“Our results show that, in people who experience muscle symptoms in the first year of taking a statin, those symptoms are actually due to the statin in only 1 of 15 of those people. For the other 14 of the 15 people who experience muscle symptoms in the first year of taking a statin, that muscle pain is not due to the statin,” lead investigator Colin Baigent, MD, said.
After the first year, there was no difference in muscle symptoms between patients taking a statin or those taking placebo.
Dr. Baigent, who is director of the Population Health Research Unit at the University of Oxford (England), presented the data on Aug. 29 at the European Society of Cardiology 2022 Congress.
It was also simultaneously published online in The Lancet.
Dr. Baigent explained that statins very rarely cause serious muscle adverse effects with biochemical evidence of cellular damage, such as myopathy (which occurs in less than 1 in 10,000 patients per year) and rhabdomyolysis (which occurs in about 0.2 per 10,000 patients per year).
The effect of statins on other less serious muscle symptoms without biochemical evidence of cellular damage is less clear, but misinformation about the risks have arisen from nonrandomized studies, with social media and press reports suggesting that the risk for muscle symptoms with statins is extremely common, Dr. Baigent said.
In response to this, the Cholesterol Trialists Collaboration put together a new program of data collection, validation, and analysis to provide reliable information from large double-blind randomized trials that are free from bias and confounding.
“Overall, when we look at all these data, we find there is about a 3% relative increase in the risks of experiencing muscle pain or weakness with a statin versus with placebo,” Dr. Baigent reported.
Muscle pain or weakness was reported by 16,835 of 62,028 patients taking a statin, (27.1%), compared with 16,446 of 61,912 patients taking placebo (26.6%), for a rate ratio of 1.03 (95% confidence interval, 1.01-1.06).
In absolute terms, the results show a rate of 166 reports of muscle symptoms per 1,000 patient-years in those taking a statin, compared with 155 per 1,000-patient-years in those taking placebo in the first year. This gives a rate ratio of 1.07 and an excess of 11 cases of muscle pain or weakness per 1,000 patients in the first year of statin therapy.
“The very small excess of muscle symptoms in the statin patients were generally mild, with most patients able to continue treatment,” Dr. Baigent added.
After the first year, the rate of muscle pain or weakness was exactly the same in the statin and placebo groups, at 50 per 1,000 patient-years.
“Therefore, for the vast majority of people who experience muscle pain or weakness on a statin, those symptoms are not due to the statin itself. It is due to something else, which could be ageing, thyroid disease, or exercise,” Dr. Baigent said. “After the first year of taking a statin, there is no excess risk of muscle pain or weakness at all.”
“To summarize, the excess risk of muscle pain or weakness with statin use is tiny, and almost nonexistent after the first year,” he added.
“Muscle pain is very common in the general population, and it was very common in both patients taking a statin and those given placebo in these randomized trials. We can only detect a difference by looking at all the data combined in this enormous study. And we now know for sure that over 90% of cases of muscle symptoms experienced by people taking a statin are not due to the statin.”
The researchers also looked at statin intensity and found that the more intense statins tend to cause slightly more muscle pain. “There was also some evidence, although this was not very clear, that the muscle pain with the more intensive statins may persist for longer than 1 year,” Dr. Baigent said.
But in terms of different moderate-intensity and high-intensity statins, there was no evidence of differences in muscle pain between the individual statin brands, he added.
Better patient information needed
Dr. Baigent called for better information in statin package inserts about the real risk for muscle symptoms with these drugs.
“We need to do a better job of communicating the real risk of muscle symptom to patients who are taking statins and to their doctors. At the moment, doctors often stop statins if patients complain of muscle pain, but our data show that in 14 out of 15 times, they would be wrong for doing that. Stopping the statin is nearly always a mistake,” he commented.
“At present, the package inserts include a whole load of rubbish from observational studies, which are completely unreliable,” he added. “This is of no value to patients. They go through this information and find several symptoms they are experiencing, which they attribute to the drugs. We really need to divide up the information into the evidence that we really know for sure and then the more speculative stuff.”
Dr. Baigent also highlighted the large benefits of statins, compared with the small risk for muscle symptoms.
“While statins may cause 11 patients per 1,000 to experience some mild muscle pain in the first year of taking these drugs, and this was reduced to none in subsequent years, statins, when used for the primary prevention of cardiovascular disease, prevent 25 cardiovascular events per 1,000 patients every year they are taken. And for secondary prevention this rises to 50 events prevented per 1,000 patients each year,” he noted.
The individual participant data meta-analysis involved 23 trials with information on almost 155,000 patients. All trials included at least 1,000 patients and at least 2 years of scheduled treatment. Adverse-event data were collected for all individual participants in 19 large randomized double-blind trials comparing statin therapy with placebo (123,940 patients) and in four randomized double-blind trials comparing more-intensive with less-intensive statin therapy (30,724 patients).
In the four trials of more-intensive versus less-intensive statin therapy, high-intensity regimens (atorvastatin 40-80 mg daily or rosuvastatin 20-40 mg daily) resulted in a larger relative increase in the rate of muscle pain or weakness than moderate-intensity regimens, with rate ratios of 1.08 (95% CI, 1.04-1.13) and 1.02 (95% CI, 1.00-1.05), respectively.
‘Reassuring information’
Discussant of the study at the ESC Hotline session, Erin Bohula, MD, Brigham and Women’s Hospital, Boston, said this new analysis had many strengths and used a rigorous approach to look at the issue of muscle symptoms with statins.
She pointed out some challenges, including the fact that the definition of adverse muscle events has changed over time and differed in the various trials, with heterogeneous data capture across trials. “So, this was a Herculean task to harmonize this very complicated dataset.”
Dr. Bohula concluded: “I think this is a very significant undertaking, resulting in a rich dataset that enhances our understanding of muscle symptoms related to statin use. The take-home for me is that muscle symptoms are a common complaint in the general population but are very rarely attributable to statins. This is very reassuring to me, and I hope it is reassuring to patients and can help us encourage them with adherence, given the clear cardiovascular benefits of statins.”
Chair of the ESC Hotline session at which the study was presented, Gabriel Steg, MD, Hôpital Bichat, Paris, asked whether some statin patients who experienced muscle symptoms with the drugs in active run-in periods in the trials may have been excluded from the main trials, so that this information might not have been captured, but Dr. Baigent replied that they also examined those data, which had been accounted for in the analysis.
“That’s really good news,” Dr. Steg commented. “This study is going to be one more tool in our response to statin skeptics and I think, as such, this work is a really a service to public health.”
The meta-analysis was funded by the British Heart Foundation, the U.K. Medical Research Council, and the Australian National Health and Medical Research Council.
A version of this article first appeared on Medscape.com.
In the vast majority of people who experience muscle pain or weakness while taking a statin, those symptoms are not related to the statin, a new individual patient data meta-analysis of randomized controlled trials shows.
The Cholesterol Trialists Collaboration meta-analysis examined 19 large randomized double-blind trials that compared statin therapy with placebo and involved almost 124,000 patients.
“Our results show that, in people who experience muscle symptoms in the first year of taking a statin, those symptoms are actually due to the statin in only 1 of 15 of those people. For the other 14 of the 15 people who experience muscle symptoms in the first year of taking a statin, that muscle pain is not due to the statin,” lead investigator Colin Baigent, MD, said.
After the first year, there was no difference in muscle symptoms between patients taking a statin or those taking placebo.
Dr. Baigent, who is director of the Population Health Research Unit at the University of Oxford (England), presented the data on Aug. 29 at the European Society of Cardiology 2022 Congress.
It was also simultaneously published online in The Lancet.
Dr. Baigent explained that statins very rarely cause serious muscle adverse effects with biochemical evidence of cellular damage, such as myopathy (which occurs in less than 1 in 10,000 patients per year) and rhabdomyolysis (which occurs in about 0.2 per 10,000 patients per year).
The effect of statins on other less serious muscle symptoms without biochemical evidence of cellular damage is less clear, but misinformation about the risks have arisen from nonrandomized studies, with social media and press reports suggesting that the risk for muscle symptoms with statins is extremely common, Dr. Baigent said.
In response to this, the Cholesterol Trialists Collaboration put together a new program of data collection, validation, and analysis to provide reliable information from large double-blind randomized trials that are free from bias and confounding.
“Overall, when we look at all these data, we find there is about a 3% relative increase in the risks of experiencing muscle pain or weakness with a statin versus with placebo,” Dr. Baigent reported.
Muscle pain or weakness was reported by 16,835 of 62,028 patients taking a statin, (27.1%), compared with 16,446 of 61,912 patients taking placebo (26.6%), for a rate ratio of 1.03 (95% confidence interval, 1.01-1.06).
In absolute terms, the results show a rate of 166 reports of muscle symptoms per 1,000 patient-years in those taking a statin, compared with 155 per 1,000-patient-years in those taking placebo in the first year. This gives a rate ratio of 1.07 and an excess of 11 cases of muscle pain or weakness per 1,000 patients in the first year of statin therapy.
“The very small excess of muscle symptoms in the statin patients were generally mild, with most patients able to continue treatment,” Dr. Baigent added.
After the first year, the rate of muscle pain or weakness was exactly the same in the statin and placebo groups, at 50 per 1,000 patient-years.
“Therefore, for the vast majority of people who experience muscle pain or weakness on a statin, those symptoms are not due to the statin itself. It is due to something else, which could be ageing, thyroid disease, or exercise,” Dr. Baigent said. “After the first year of taking a statin, there is no excess risk of muscle pain or weakness at all.”
“To summarize, the excess risk of muscle pain or weakness with statin use is tiny, and almost nonexistent after the first year,” he added.
“Muscle pain is very common in the general population, and it was very common in both patients taking a statin and those given placebo in these randomized trials. We can only detect a difference by looking at all the data combined in this enormous study. And we now know for sure that over 90% of cases of muscle symptoms experienced by people taking a statin are not due to the statin.”
The researchers also looked at statin intensity and found that the more intense statins tend to cause slightly more muscle pain. “There was also some evidence, although this was not very clear, that the muscle pain with the more intensive statins may persist for longer than 1 year,” Dr. Baigent said.
But in terms of different moderate-intensity and high-intensity statins, there was no evidence of differences in muscle pain between the individual statin brands, he added.
Better patient information needed
Dr. Baigent called for better information in statin package inserts about the real risk for muscle symptoms with these drugs.
“We need to do a better job of communicating the real risk of muscle symptom to patients who are taking statins and to their doctors. At the moment, doctors often stop statins if patients complain of muscle pain, but our data show that in 14 out of 15 times, they would be wrong for doing that. Stopping the statin is nearly always a mistake,” he commented.
“At present, the package inserts include a whole load of rubbish from observational studies, which are completely unreliable,” he added. “This is of no value to patients. They go through this information and find several symptoms they are experiencing, which they attribute to the drugs. We really need to divide up the information into the evidence that we really know for sure and then the more speculative stuff.”
Dr. Baigent also highlighted the large benefits of statins, compared with the small risk for muscle symptoms.
“While statins may cause 11 patients per 1,000 to experience some mild muscle pain in the first year of taking these drugs, and this was reduced to none in subsequent years, statins, when used for the primary prevention of cardiovascular disease, prevent 25 cardiovascular events per 1,000 patients every year they are taken. And for secondary prevention this rises to 50 events prevented per 1,000 patients each year,” he noted.
The individual participant data meta-analysis involved 23 trials with information on almost 155,000 patients. All trials included at least 1,000 patients and at least 2 years of scheduled treatment. Adverse-event data were collected for all individual participants in 19 large randomized double-blind trials comparing statin therapy with placebo (123,940 patients) and in four randomized double-blind trials comparing more-intensive with less-intensive statin therapy (30,724 patients).
In the four trials of more-intensive versus less-intensive statin therapy, high-intensity regimens (atorvastatin 40-80 mg daily or rosuvastatin 20-40 mg daily) resulted in a larger relative increase in the rate of muscle pain or weakness than moderate-intensity regimens, with rate ratios of 1.08 (95% CI, 1.04-1.13) and 1.02 (95% CI, 1.00-1.05), respectively.
‘Reassuring information’
Discussant of the study at the ESC Hotline session, Erin Bohula, MD, Brigham and Women’s Hospital, Boston, said this new analysis had many strengths and used a rigorous approach to look at the issue of muscle symptoms with statins.
She pointed out some challenges, including the fact that the definition of adverse muscle events has changed over time and differed in the various trials, with heterogeneous data capture across trials. “So, this was a Herculean task to harmonize this very complicated dataset.”
Dr. Bohula concluded: “I think this is a very significant undertaking, resulting in a rich dataset that enhances our understanding of muscle symptoms related to statin use. The take-home for me is that muscle symptoms are a common complaint in the general population but are very rarely attributable to statins. This is very reassuring to me, and I hope it is reassuring to patients and can help us encourage them with adherence, given the clear cardiovascular benefits of statins.”
Chair of the ESC Hotline session at which the study was presented, Gabriel Steg, MD, Hôpital Bichat, Paris, asked whether some statin patients who experienced muscle symptoms with the drugs in active run-in periods in the trials may have been excluded from the main trials, so that this information might not have been captured, but Dr. Baigent replied that they also examined those data, which had been accounted for in the analysis.
“That’s really good news,” Dr. Steg commented. “This study is going to be one more tool in our response to statin skeptics and I think, as such, this work is a really a service to public health.”
The meta-analysis was funded by the British Heart Foundation, the U.K. Medical Research Council, and the Australian National Health and Medical Research Council.
A version of this article first appeared on Medscape.com.
FROM ESC CONGRESS 2022
Online tool IDs people with genetic mutations linked to cancer
(PGVs) in a diverse spectrum of cancer susceptibility genes.
The PREMMplus online tool was developed and validated by researchers at the Dana-Farber Cancer Institute, Boston using three cohorts involving more than 30,000 individuals who had undergone multigene hereditary cancer risk testing.
The study was published online in the Journal of Clinical Oncology.
“Our findings show that PREMMplus has the potential to change the model by which patients and family members are referred for genetic testing and counseling,” senior author Sapna Syngal, MD, MPH, with Dana-Farber/Brigham and Women’s Hospital, Boston, said in an institution news release.
Traditionally, when there is concern about a family cancer history, the individual is referred to a genetics clinic, where a counselor takes a complete family history.
“At a time when there’s a shortage of genetic counselors, PREMMplus can help streamline risk assessment and ensure that their time can be focused on where they’re most needed – helping people understand the results of genetic testing and the options available when a cancer-susceptibility gene is found,” Dr. Syngal says.
Online tool
The tool uses clinical data (age, sex, ethnicity, and personal/family history of 18 cancers) to determine an individual’s likelihood of harboring a PGV in 19 cancer susceptibility genes.
A PREMMplus score of 2.5% or greater had a 89%-94% sensitivity and > 97% negative predictive value (NPV) for identifying individuals with PGVs in 11 well-defined “category A” high-penetrance cancer risk genes: APC, BRCA1, BRCA2, CDH1, EPCAM, MLH1, MSH2, MSH6, biallelic MUTYH, PMS2, and TP53.
These PGVs “represent diverse types of inherited cancer risk for which there are established risk-reduction guidelines,” the study team says. Cancers associated with these PGVs include breast, ovarian, colorectal, pancreatic, and prostate cancer, as well as those that make up Lynch syndrome.
The ability of PREMMplus to identify individuals with PGVs in “moderate-penetrance” cancer risk genes (such as CHEK2 and ATM) was somewhat reduced but was still “quite strong” (84%-90% sensitivity and > 93% NPV), the study team reports.
In an interview, Dr. Syngal said her ultimate vision of this online tool is that it will be adapted into the electronic medical record (EMR).
“Through the EMR, it might somehow get pushed out to people before an oncology or primary care appointment or before a mammography or colonoscopy. Then by the time they come in, the doctor or nurse practitioner has the information and can refer them for genetic testing if appropriate,” Dr. Syngal explained.
The tool is not currently available for routine clinical use. The goal is to make it available online in a couple of months.
Dr. Syngal said two versions will be available. One will be a user-friendly version that can be filled out directly by patients and that will tell whether someone passes the threshold of needing genetic testing. The patient would then take that information to their primary care doctor.
With the second version, the doctor and patient would fill out the information together during an office visit.
PREMMplus would be free for the individual patient or provider.
“What we hope is that hospital systems will use it and that insurance companies will also use it as a way to say who needs testing and who to approve for testing,” Dr. Syngal told this news organization.
“For a hospital system or a genetic testing company, for example, that wants to integrate it into their direct-to-consumer platform, they would have to take out a license from Dana-Farber, and cost would be negotiated with each entity based on how they’re going to use it,” Dr. Syngal said.
Funding for the research was provided by the National Institutes of Health. A complete list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
(PGVs) in a diverse spectrum of cancer susceptibility genes.
The PREMMplus online tool was developed and validated by researchers at the Dana-Farber Cancer Institute, Boston using three cohorts involving more than 30,000 individuals who had undergone multigene hereditary cancer risk testing.
The study was published online in the Journal of Clinical Oncology.
“Our findings show that PREMMplus has the potential to change the model by which patients and family members are referred for genetic testing and counseling,” senior author Sapna Syngal, MD, MPH, with Dana-Farber/Brigham and Women’s Hospital, Boston, said in an institution news release.
Traditionally, when there is concern about a family cancer history, the individual is referred to a genetics clinic, where a counselor takes a complete family history.
“At a time when there’s a shortage of genetic counselors, PREMMplus can help streamline risk assessment and ensure that their time can be focused on where they’re most needed – helping people understand the results of genetic testing and the options available when a cancer-susceptibility gene is found,” Dr. Syngal says.
Online tool
The tool uses clinical data (age, sex, ethnicity, and personal/family history of 18 cancers) to determine an individual’s likelihood of harboring a PGV in 19 cancer susceptibility genes.
A PREMMplus score of 2.5% or greater had a 89%-94% sensitivity and > 97% negative predictive value (NPV) for identifying individuals with PGVs in 11 well-defined “category A” high-penetrance cancer risk genes: APC, BRCA1, BRCA2, CDH1, EPCAM, MLH1, MSH2, MSH6, biallelic MUTYH, PMS2, and TP53.
These PGVs “represent diverse types of inherited cancer risk for which there are established risk-reduction guidelines,” the study team says. Cancers associated with these PGVs include breast, ovarian, colorectal, pancreatic, and prostate cancer, as well as those that make up Lynch syndrome.
The ability of PREMMplus to identify individuals with PGVs in “moderate-penetrance” cancer risk genes (such as CHEK2 and ATM) was somewhat reduced but was still “quite strong” (84%-90% sensitivity and > 93% NPV), the study team reports.
In an interview, Dr. Syngal said her ultimate vision of this online tool is that it will be adapted into the electronic medical record (EMR).
“Through the EMR, it might somehow get pushed out to people before an oncology or primary care appointment or before a mammography or colonoscopy. Then by the time they come in, the doctor or nurse practitioner has the information and can refer them for genetic testing if appropriate,” Dr. Syngal explained.
The tool is not currently available for routine clinical use. The goal is to make it available online in a couple of months.
Dr. Syngal said two versions will be available. One will be a user-friendly version that can be filled out directly by patients and that will tell whether someone passes the threshold of needing genetic testing. The patient would then take that information to their primary care doctor.
With the second version, the doctor and patient would fill out the information together during an office visit.
PREMMplus would be free for the individual patient or provider.
“What we hope is that hospital systems will use it and that insurance companies will also use it as a way to say who needs testing and who to approve for testing,” Dr. Syngal told this news organization.
“For a hospital system or a genetic testing company, for example, that wants to integrate it into their direct-to-consumer platform, they would have to take out a license from Dana-Farber, and cost would be negotiated with each entity based on how they’re going to use it,” Dr. Syngal said.
Funding for the research was provided by the National Institutes of Health. A complete list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
(PGVs) in a diverse spectrum of cancer susceptibility genes.
The PREMMplus online tool was developed and validated by researchers at the Dana-Farber Cancer Institute, Boston using three cohorts involving more than 30,000 individuals who had undergone multigene hereditary cancer risk testing.
The study was published online in the Journal of Clinical Oncology.
“Our findings show that PREMMplus has the potential to change the model by which patients and family members are referred for genetic testing and counseling,” senior author Sapna Syngal, MD, MPH, with Dana-Farber/Brigham and Women’s Hospital, Boston, said in an institution news release.
Traditionally, when there is concern about a family cancer history, the individual is referred to a genetics clinic, where a counselor takes a complete family history.
“At a time when there’s a shortage of genetic counselors, PREMMplus can help streamline risk assessment and ensure that their time can be focused on where they’re most needed – helping people understand the results of genetic testing and the options available when a cancer-susceptibility gene is found,” Dr. Syngal says.
Online tool
The tool uses clinical data (age, sex, ethnicity, and personal/family history of 18 cancers) to determine an individual’s likelihood of harboring a PGV in 19 cancer susceptibility genes.
A PREMMplus score of 2.5% or greater had a 89%-94% sensitivity and > 97% negative predictive value (NPV) for identifying individuals with PGVs in 11 well-defined “category A” high-penetrance cancer risk genes: APC, BRCA1, BRCA2, CDH1, EPCAM, MLH1, MSH2, MSH6, biallelic MUTYH, PMS2, and TP53.
These PGVs “represent diverse types of inherited cancer risk for which there are established risk-reduction guidelines,” the study team says. Cancers associated with these PGVs include breast, ovarian, colorectal, pancreatic, and prostate cancer, as well as those that make up Lynch syndrome.
The ability of PREMMplus to identify individuals with PGVs in “moderate-penetrance” cancer risk genes (such as CHEK2 and ATM) was somewhat reduced but was still “quite strong” (84%-90% sensitivity and > 93% NPV), the study team reports.
In an interview, Dr. Syngal said her ultimate vision of this online tool is that it will be adapted into the electronic medical record (EMR).
“Through the EMR, it might somehow get pushed out to people before an oncology or primary care appointment or before a mammography or colonoscopy. Then by the time they come in, the doctor or nurse practitioner has the information and can refer them for genetic testing if appropriate,” Dr. Syngal explained.
The tool is not currently available for routine clinical use. The goal is to make it available online in a couple of months.
Dr. Syngal said two versions will be available. One will be a user-friendly version that can be filled out directly by patients and that will tell whether someone passes the threshold of needing genetic testing. The patient would then take that information to their primary care doctor.
With the second version, the doctor and patient would fill out the information together during an office visit.
PREMMplus would be free for the individual patient or provider.
“What we hope is that hospital systems will use it and that insurance companies will also use it as a way to say who needs testing and who to approve for testing,” Dr. Syngal told this news organization.
“For a hospital system or a genetic testing company, for example, that wants to integrate it into their direct-to-consumer platform, they would have to take out a license from Dana-Farber, and cost would be negotiated with each entity based on how they’re going to use it,” Dr. Syngal said.
Funding for the research was provided by the National Institutes of Health. A complete list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
Intera Oncology recalls hepatic artery infusion pumps for possible life-threatening issue
Although no injuries or deaths related to the pump malfunction have been reported yet, the U.S. Food and Drug Administration has deemed the recall Class I, the most serious category that indicates the device could cause injury or death.
Intera Oncology initiated the recall in July following reports from clinicians that the pumps, which are implanted to deliver chemotherapy to treat liver tumors, were delivering medications faster than expected. A fast flow rate can lead to life-threatening hematologic toxicity, neurotoxicity, or death. It also means patients will run out of medication too soon, potentially leading to disease progression or death.
The FDA notice states the company has advised customers to continue to monitor flow rate as per standard refill procedure as well as monitor for liver toxicity to adjust dosing as per standard protocols.
The company also said to consider pump replacement if altered flow can’t be adequately managed by dosing adjustments or having patients come in for medication refills and to verify the flow rate sooner than every 2 weeks if the pump appears to be flowing more than 15% outside its labeled specification.
Questions about the recall can be directed to Intera Oncology at (800) 660-2660 or support@interaoncol.
A version of this article first appeared on Medscape.com.
Although no injuries or deaths related to the pump malfunction have been reported yet, the U.S. Food and Drug Administration has deemed the recall Class I, the most serious category that indicates the device could cause injury or death.
Intera Oncology initiated the recall in July following reports from clinicians that the pumps, which are implanted to deliver chemotherapy to treat liver tumors, were delivering medications faster than expected. A fast flow rate can lead to life-threatening hematologic toxicity, neurotoxicity, or death. It also means patients will run out of medication too soon, potentially leading to disease progression or death.
The FDA notice states the company has advised customers to continue to monitor flow rate as per standard refill procedure as well as monitor for liver toxicity to adjust dosing as per standard protocols.
The company also said to consider pump replacement if altered flow can’t be adequately managed by dosing adjustments or having patients come in for medication refills and to verify the flow rate sooner than every 2 weeks if the pump appears to be flowing more than 15% outside its labeled specification.
Questions about the recall can be directed to Intera Oncology at (800) 660-2660 or support@interaoncol.
A version of this article first appeared on Medscape.com.
Although no injuries or deaths related to the pump malfunction have been reported yet, the U.S. Food and Drug Administration has deemed the recall Class I, the most serious category that indicates the device could cause injury or death.
Intera Oncology initiated the recall in July following reports from clinicians that the pumps, which are implanted to deliver chemotherapy to treat liver tumors, were delivering medications faster than expected. A fast flow rate can lead to life-threatening hematologic toxicity, neurotoxicity, or death. It also means patients will run out of medication too soon, potentially leading to disease progression or death.
The FDA notice states the company has advised customers to continue to monitor flow rate as per standard refill procedure as well as monitor for liver toxicity to adjust dosing as per standard protocols.
The company also said to consider pump replacement if altered flow can’t be adequately managed by dosing adjustments or having patients come in for medication refills and to verify the flow rate sooner than every 2 weeks if the pump appears to be flowing more than 15% outside its labeled specification.
Questions about the recall can be directed to Intera Oncology at (800) 660-2660 or support@interaoncol.
A version of this article first appeared on Medscape.com.
Question 2
Q2. Correct answer: D. Treatment for recurrent or metastatic disease is imatinib.
Rationale
This patient has a gastrointestinal stromal tumor (GIST) of the stomach. GISTs are the most common mesenchymal tumor found in the stomach. Gastric GISTs have a better prognosis than those found in the small intestine. GISTs are often found incidentally but can cause symptoms such as bleeding due to ulceration. Pathology of a GIST shows spindle cells that stain positive for CD117 and harbor KIT mutations. Malignant potential and decreased survival are associated with size more than 2 cm and high mitotic index (more than 5/50 high power field). Endoscopic ultrasound with tissue sampling is the preferred diagnostic technique. High-risk features include lobulated or irregular borders, invasion into adjacent structures and heterogeneity. Fine needle aspirate may be suboptimal, and core biopsy is an acceptable alternative. Resection is indicated for lesions that are symptomatic, size more than 2 cm or high-risk EUS features. Lesions less than 2 cm without high-risk features can be surveyed by EUS annually. Endoscopic resection might be possible for small lesions but should be done in specialized centers. Metastatic or recurrent lesions are treated with imatinib.
Reference
ASGE Standards of Practice Committee. Gastrointest Endosc. 2015 Jul;82(1):1-8.
Q2. Correct answer: D. Treatment for recurrent or metastatic disease is imatinib.
Rationale
This patient has a gastrointestinal stromal tumor (GIST) of the stomach. GISTs are the most common mesenchymal tumor found in the stomach. Gastric GISTs have a better prognosis than those found in the small intestine. GISTs are often found incidentally but can cause symptoms such as bleeding due to ulceration. Pathology of a GIST shows spindle cells that stain positive for CD117 and harbor KIT mutations. Malignant potential and decreased survival are associated with size more than 2 cm and high mitotic index (more than 5/50 high power field). Endoscopic ultrasound with tissue sampling is the preferred diagnostic technique. High-risk features include lobulated or irregular borders, invasion into adjacent structures and heterogeneity. Fine needle aspirate may be suboptimal, and core biopsy is an acceptable alternative. Resection is indicated for lesions that are symptomatic, size more than 2 cm or high-risk EUS features. Lesions less than 2 cm without high-risk features can be surveyed by EUS annually. Endoscopic resection might be possible for small lesions but should be done in specialized centers. Metastatic or recurrent lesions are treated with imatinib.
Reference
ASGE Standards of Practice Committee. Gastrointest Endosc. 2015 Jul;82(1):1-8.
Q2. Correct answer: D. Treatment for recurrent or metastatic disease is imatinib.
Rationale
This patient has a gastrointestinal stromal tumor (GIST) of the stomach. GISTs are the most common mesenchymal tumor found in the stomach. Gastric GISTs have a better prognosis than those found in the small intestine. GISTs are often found incidentally but can cause symptoms such as bleeding due to ulceration. Pathology of a GIST shows spindle cells that stain positive for CD117 and harbor KIT mutations. Malignant potential and decreased survival are associated with size more than 2 cm and high mitotic index (more than 5/50 high power field). Endoscopic ultrasound with tissue sampling is the preferred diagnostic technique. High-risk features include lobulated or irregular borders, invasion into adjacent structures and heterogeneity. Fine needle aspirate may be suboptimal, and core biopsy is an acceptable alternative. Resection is indicated for lesions that are symptomatic, size more than 2 cm or high-risk EUS features. Lesions less than 2 cm without high-risk features can be surveyed by EUS annually. Endoscopic resection might be possible for small lesions but should be done in specialized centers. Metastatic or recurrent lesions are treated with imatinib.
Reference
ASGE Standards of Practice Committee. Gastrointest Endosc. 2015 Jul;82(1):1-8.
Q2. A 65-year-old man undergoes upper endoscopy for epigastric discomfort. The exam results are normal, except for a 3-cm submucosal mass in the body of the stomach. Endoscopic ultrasound shows that the mass arises from the fourth layer of the stomach wall. CT of the abdomen confirms the solid gastric mass with several small lesions in the liver concerning for metastatic disease. Biopsy of the mass shows CD117-positive spindle cells.