User login
Refinements Advance Standard Therapies for Squamous Cell Carcinoma
ORLANDO – Despite a paucity of recently approved treatments for squamous cell carcinoma, you can use study findings from the past few years to refine your clinical approach, Dr. Suzanne Olbricht said.
"It would be nice if I had some new treatments to discuss, but I don't. So let's go through some recent studies," Dr. Olbricht said at the annual meeting of the Florida Society of Dermatologic Surgeons.
Photodynamic Therapy
Investigators, for example, found methyl aminolevulinate photodynamic therapy (MAL-PDT) superior to 5-fluorouracil and cryosurgery for squamous cell carcinoma in situ. In addition, ophthalmologists might start using more imiquimod to treat periorbital skin cancers based on outcomes of a recent case series, Dr. Olbricht said. Other findings support promise for the oral medications cetuximab and capecitabine.
The prospective, randomized study that bolsters use of MAL-PDT included 225 patients with biopsy-confirmed squamous cell carcinoma in situ (Arch. Dermatol. 2006;142:729-35). Researchers found an 80% complete response rate in the MAL-PDT–treated patients at 12 months, compared with a 69% rate with 5-fluorouracil and a 67% rate in patients treated with cryotherapy.
"MAL-PDT was judged [to have the] most acceptable cosmetic outcome," Dr. Olbricht said. She added that 5-fluorouracil is "still a good therapy if some patients prefer to do things at home."
This is a "large study that is worth thinking about," said Dr. Olbricht, chair of the department of dermatology at the Lahey Clinic in Burlington, Mass., and a member of the dermatology faculty at Harvard Medical School, Boston.
Photodynamic therapy (PDT) was associated with a 72% complete response in a smaller study of 30 patients with recurrent tumors of the head and neck (J. Drugs Dermatol. 2010;9:122-6).
These investigators described PDT as effective, tolerable, and associated with good cosmetic results for recurrent squamous cell carcinoma or basal cell carcinoma. One concern, Dr. Olbricht said, is "they did not describe how they did the PDT."
Imiquimod Therapy
Another noteworthy study supports imiquimod for periorbital skin lesions (Orbit 2010;29:83-7). Two patients in this case series had basal cell carcinoma of the eyelid, one had actinic keratosis, and another had intraepidermal squamous cell carcinoma (Bowen’s disease). A fifth patient presented with concomitant squamous cell carcinoma and intraepidermal squamous cell carcinoma. All tumors regressed with a clinical cure at 6-9 months.
"It’s a short follow-up, but your ophthalmologic colleagues will be using imiquimod," Dr. Olbricht said.
Cetuximab and Capecitabine
Oral cetuximab (Erbitux, Imclone) was associated with a complete clinical response for a patient with organ metastases from cutaneous squamous cell carcinoma (Dermatol. Surg. 2010;36:2069-74). Keep two caveats in mind with a cetuximab treatment strategy, Dr. Olbricht said. First, it's expensive – costing approximately $30,000 for an 8-week series of infusions. Second, cetuximab is effective only for skin cancer patients who do not have a KRAS mutation.
Another treatment showing promise is capecitabine (Xeloda, Hoffmann La Roche), an oral 5-fluorouracil precursor. New lesion development was halted within 6 months for three patients with a history of more than one new squamous cell carcinoma occurrence per month for more than 3 years (Dermatol. Surg. 2009;35:1657-72). Dr. Olbricht noted that hand and foot erythema occurred, which "is supposedly well known with this oral medication."
A meeting attendee asked how Dr. Olbricht manages patients with chronic, multiple squamous cell carcinomas. "I probably have six or seven people for years who come in at least once a month for skin cancer," she replied. "I try to take care of what they have at that moment."
For example, she recently treated a man whose biggest clinical concern is skin cancer 7 years post kidney transplant. "We sent 12 specimens to pathology. We did two Mohs procedures on his face, two excisions on his legs, and the rest were probably electrodesiccation and curettage on his trunk."
"I know I would make more money if I treated each lesion at individual sessions ... but I think this is best approach to this kind of patient," Dr. Olbricht said.
Dr. Olbricht said she had no relevant disclosures.
ORLANDO – Despite a paucity of recently approved treatments for squamous cell carcinoma, you can use study findings from the past few years to refine your clinical approach, Dr. Suzanne Olbricht said.
"It would be nice if I had some new treatments to discuss, but I don't. So let's go through some recent studies," Dr. Olbricht said at the annual meeting of the Florida Society of Dermatologic Surgeons.
Photodynamic Therapy
Investigators, for example, found methyl aminolevulinate photodynamic therapy (MAL-PDT) superior to 5-fluorouracil and cryosurgery for squamous cell carcinoma in situ. In addition, ophthalmologists might start using more imiquimod to treat periorbital skin cancers based on outcomes of a recent case series, Dr. Olbricht said. Other findings support promise for the oral medications cetuximab and capecitabine.
The prospective, randomized study that bolsters use of MAL-PDT included 225 patients with biopsy-confirmed squamous cell carcinoma in situ (Arch. Dermatol. 2006;142:729-35). Researchers found an 80% complete response rate in the MAL-PDT–treated patients at 12 months, compared with a 69% rate with 5-fluorouracil and a 67% rate in patients treated with cryotherapy.
"MAL-PDT was judged [to have the] most acceptable cosmetic outcome," Dr. Olbricht said. She added that 5-fluorouracil is "still a good therapy if some patients prefer to do things at home."
This is a "large study that is worth thinking about," said Dr. Olbricht, chair of the department of dermatology at the Lahey Clinic in Burlington, Mass., and a member of the dermatology faculty at Harvard Medical School, Boston.
Photodynamic therapy (PDT) was associated with a 72% complete response in a smaller study of 30 patients with recurrent tumors of the head and neck (J. Drugs Dermatol. 2010;9:122-6).
These investigators described PDT as effective, tolerable, and associated with good cosmetic results for recurrent squamous cell carcinoma or basal cell carcinoma. One concern, Dr. Olbricht said, is "they did not describe how they did the PDT."
Imiquimod Therapy
Another noteworthy study supports imiquimod for periorbital skin lesions (Orbit 2010;29:83-7). Two patients in this case series had basal cell carcinoma of the eyelid, one had actinic keratosis, and another had intraepidermal squamous cell carcinoma (Bowen’s disease). A fifth patient presented with concomitant squamous cell carcinoma and intraepidermal squamous cell carcinoma. All tumors regressed with a clinical cure at 6-9 months.
"It’s a short follow-up, but your ophthalmologic colleagues will be using imiquimod," Dr. Olbricht said.
Cetuximab and Capecitabine
Oral cetuximab (Erbitux, Imclone) was associated with a complete clinical response for a patient with organ metastases from cutaneous squamous cell carcinoma (Dermatol. Surg. 2010;36:2069-74). Keep two caveats in mind with a cetuximab treatment strategy, Dr. Olbricht said. First, it's expensive – costing approximately $30,000 for an 8-week series of infusions. Second, cetuximab is effective only for skin cancer patients who do not have a KRAS mutation.
Another treatment showing promise is capecitabine (Xeloda, Hoffmann La Roche), an oral 5-fluorouracil precursor. New lesion development was halted within 6 months for three patients with a history of more than one new squamous cell carcinoma occurrence per month for more than 3 years (Dermatol. Surg. 2009;35:1657-72). Dr. Olbricht noted that hand and foot erythema occurred, which "is supposedly well known with this oral medication."
A meeting attendee asked how Dr. Olbricht manages patients with chronic, multiple squamous cell carcinomas. "I probably have six or seven people for years who come in at least once a month for skin cancer," she replied. "I try to take care of what they have at that moment."
For example, she recently treated a man whose biggest clinical concern is skin cancer 7 years post kidney transplant. "We sent 12 specimens to pathology. We did two Mohs procedures on his face, two excisions on his legs, and the rest were probably electrodesiccation and curettage on his trunk."
"I know I would make more money if I treated each lesion at individual sessions ... but I think this is best approach to this kind of patient," Dr. Olbricht said.
Dr. Olbricht said she had no relevant disclosures.
ORLANDO – Despite a paucity of recently approved treatments for squamous cell carcinoma, you can use study findings from the past few years to refine your clinical approach, Dr. Suzanne Olbricht said.
"It would be nice if I had some new treatments to discuss, but I don't. So let's go through some recent studies," Dr. Olbricht said at the annual meeting of the Florida Society of Dermatologic Surgeons.
Photodynamic Therapy
Investigators, for example, found methyl aminolevulinate photodynamic therapy (MAL-PDT) superior to 5-fluorouracil and cryosurgery for squamous cell carcinoma in situ. In addition, ophthalmologists might start using more imiquimod to treat periorbital skin cancers based on outcomes of a recent case series, Dr. Olbricht said. Other findings support promise for the oral medications cetuximab and capecitabine.
The prospective, randomized study that bolsters use of MAL-PDT included 225 patients with biopsy-confirmed squamous cell carcinoma in situ (Arch. Dermatol. 2006;142:729-35). Researchers found an 80% complete response rate in the MAL-PDT–treated patients at 12 months, compared with a 69% rate with 5-fluorouracil and a 67% rate in patients treated with cryotherapy.
"MAL-PDT was judged [to have the] most acceptable cosmetic outcome," Dr. Olbricht said. She added that 5-fluorouracil is "still a good therapy if some patients prefer to do things at home."
This is a "large study that is worth thinking about," said Dr. Olbricht, chair of the department of dermatology at the Lahey Clinic in Burlington, Mass., and a member of the dermatology faculty at Harvard Medical School, Boston.
Photodynamic therapy (PDT) was associated with a 72% complete response in a smaller study of 30 patients with recurrent tumors of the head and neck (J. Drugs Dermatol. 2010;9:122-6).
These investigators described PDT as effective, tolerable, and associated with good cosmetic results for recurrent squamous cell carcinoma or basal cell carcinoma. One concern, Dr. Olbricht said, is "they did not describe how they did the PDT."
Imiquimod Therapy
Another noteworthy study supports imiquimod for periorbital skin lesions (Orbit 2010;29:83-7). Two patients in this case series had basal cell carcinoma of the eyelid, one had actinic keratosis, and another had intraepidermal squamous cell carcinoma (Bowen’s disease). A fifth patient presented with concomitant squamous cell carcinoma and intraepidermal squamous cell carcinoma. All tumors regressed with a clinical cure at 6-9 months.
"It’s a short follow-up, but your ophthalmologic colleagues will be using imiquimod," Dr. Olbricht said.
Cetuximab and Capecitabine
Oral cetuximab (Erbitux, Imclone) was associated with a complete clinical response for a patient with organ metastases from cutaneous squamous cell carcinoma (Dermatol. Surg. 2010;36:2069-74). Keep two caveats in mind with a cetuximab treatment strategy, Dr. Olbricht said. First, it's expensive – costing approximately $30,000 for an 8-week series of infusions. Second, cetuximab is effective only for skin cancer patients who do not have a KRAS mutation.
Another treatment showing promise is capecitabine (Xeloda, Hoffmann La Roche), an oral 5-fluorouracil precursor. New lesion development was halted within 6 months for three patients with a history of more than one new squamous cell carcinoma occurrence per month for more than 3 years (Dermatol. Surg. 2009;35:1657-72). Dr. Olbricht noted that hand and foot erythema occurred, which "is supposedly well known with this oral medication."
A meeting attendee asked how Dr. Olbricht manages patients with chronic, multiple squamous cell carcinomas. "I probably have six or seven people for years who come in at least once a month for skin cancer," she replied. "I try to take care of what they have at that moment."
For example, she recently treated a man whose biggest clinical concern is skin cancer 7 years post kidney transplant. "We sent 12 specimens to pathology. We did two Mohs procedures on his face, two excisions on his legs, and the rest were probably electrodesiccation and curettage on his trunk."
"I know I would make more money if I treated each lesion at individual sessions ... but I think this is best approach to this kind of patient," Dr. Olbricht said.
Dr. Olbricht said she had no relevant disclosures.
EXPERT OPINION FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGIC SURGEONS
High-Risk Skin Cancer Patients May Benefit from Mohs + Radiation
SAN DIEGO – Aggressive skin cancers can recur, spread, and kill – and these lesions deserve special consideration after even an apparently successful Mohs procedure.
"Mohs is especially well suited to identify the rare patient who has a high-risk profile for local recurrence or metastasis," Dr. David Hodgens said at a meeting sponsored by the American Society for Mohs Surgery. "We can work together to develop radiotherapy techniques that treat both the primary site and the nodal basins to minimize toxicity and give the patient a better chance of a cure."
The Mohs surgeon is unlikely to see many patients who will need postoperative radiotherapy, said Dr. Hodgens, director of radiation oncology at Scripps Memorial Hospital, La Jolla, Calif. "Keep in mind the vast majority of patients will not need this – but be on the lookout for those who might."
Any skin cancer – squamous or basal cell carcinoma, Merkel cell tumor, melanoma or angiosarcoma – can be increasingly dangerous if certain risk factors are present, Dr. Hodgens said. Risk factors for local recurrence or metastatic spread include a lesion size greater than 2 cm; depth greater then 4 mm or a Clark level IV-V; location on the ear or lip, or within a scar; or perineural or periosteal invasion. Immunosuppressed patients are at particularly high risk, as are those with an already recurrent lesion. "A lesion that has recurred after prior treatment is one of the greatest risks of further recurrence or metastatic spread," he stressed.
Electron beam radiation is the best treatment for high-risk skin cancers. "Electrons are essential in treating the postoperative Mohs patient. If your radiation oncologist is not using electrons, find one who is," he said.
High-energy x-rays are also used, but mostly to cover a nodal basin where spread is suspected. But electrons have a unique property that allows a narrow beam to deliver 90% of its energy to a prespecified depth and then very rapidly decrease that energy as the beam goes deeper into the tissue. "Electrons provide penetration to a desired depth without the through transmission of x-rays," Dr. Hodgens said.
Post-Mohs radiation calls for a large treatment field that encompasses the entire surgical field and generous margins. "We try to get at least 2 cm around the surgical bed," he said. Patients typically receive daily treatments for 5-7 weeks, with 1.8-2 Gy/day to the primary site. The total radiation dose is usually around 50-60 Gy.
"We treat them as seriously as we do any of our patients with breast, lung, or brain tumors," Dr. Hodgens said. "They have the potential to die of their skin cancer. This is a long aggressive course of treatment designed to give the best chance of cure."
X-rays can be added to treat the associated nodal basin, if nodal spread is a concern. "The toxicity of adding nodal radiation is minimal," Dr. Hodgens said. "It’s very well tolerated, with mostly just mild erythema. It’s really an easy thing to do and very worthwhile."
There are few studies published on postoperative radiation for strict indications after well-performed Mohs surgery. Most recently, a Greek study of 315 patients found that post-Mohs radiation was associated with a 92% reduced risk of recurrence (Eur. J. Cancer 2010;46:1563-72).
Last year, an Australian study of 118 patients with cutaneous head or neck cancers found that surgery plus radiotherapy improved survival rates, even among those with perineural involvement – a very poor prognostic factor. The 5-year local control rates were 90% for those with histologically proven perineural involvement and 57% for those with symptoms of perineural involvement (Head Neck 2009;31:604-10)
Dr. Hodgens had no financial disclosures.
SAN DIEGO – Aggressive skin cancers can recur, spread, and kill – and these lesions deserve special consideration after even an apparently successful Mohs procedure.
"Mohs is especially well suited to identify the rare patient who has a high-risk profile for local recurrence or metastasis," Dr. David Hodgens said at a meeting sponsored by the American Society for Mohs Surgery. "We can work together to develop radiotherapy techniques that treat both the primary site and the nodal basins to minimize toxicity and give the patient a better chance of a cure."
The Mohs surgeon is unlikely to see many patients who will need postoperative radiotherapy, said Dr. Hodgens, director of radiation oncology at Scripps Memorial Hospital, La Jolla, Calif. "Keep in mind the vast majority of patients will not need this – but be on the lookout for those who might."
Any skin cancer – squamous or basal cell carcinoma, Merkel cell tumor, melanoma or angiosarcoma – can be increasingly dangerous if certain risk factors are present, Dr. Hodgens said. Risk factors for local recurrence or metastatic spread include a lesion size greater than 2 cm; depth greater then 4 mm or a Clark level IV-V; location on the ear or lip, or within a scar; or perineural or periosteal invasion. Immunosuppressed patients are at particularly high risk, as are those with an already recurrent lesion. "A lesion that has recurred after prior treatment is one of the greatest risks of further recurrence or metastatic spread," he stressed.
Electron beam radiation is the best treatment for high-risk skin cancers. "Electrons are essential in treating the postoperative Mohs patient. If your radiation oncologist is not using electrons, find one who is," he said.
High-energy x-rays are also used, but mostly to cover a nodal basin where spread is suspected. But electrons have a unique property that allows a narrow beam to deliver 90% of its energy to a prespecified depth and then very rapidly decrease that energy as the beam goes deeper into the tissue. "Electrons provide penetration to a desired depth without the through transmission of x-rays," Dr. Hodgens said.
Post-Mohs radiation calls for a large treatment field that encompasses the entire surgical field and generous margins. "We try to get at least 2 cm around the surgical bed," he said. Patients typically receive daily treatments for 5-7 weeks, with 1.8-2 Gy/day to the primary site. The total radiation dose is usually around 50-60 Gy.
"We treat them as seriously as we do any of our patients with breast, lung, or brain tumors," Dr. Hodgens said. "They have the potential to die of their skin cancer. This is a long aggressive course of treatment designed to give the best chance of cure."
X-rays can be added to treat the associated nodal basin, if nodal spread is a concern. "The toxicity of adding nodal radiation is minimal," Dr. Hodgens said. "It’s very well tolerated, with mostly just mild erythema. It’s really an easy thing to do and very worthwhile."
There are few studies published on postoperative radiation for strict indications after well-performed Mohs surgery. Most recently, a Greek study of 315 patients found that post-Mohs radiation was associated with a 92% reduced risk of recurrence (Eur. J. Cancer 2010;46:1563-72).
Last year, an Australian study of 118 patients with cutaneous head or neck cancers found that surgery plus radiotherapy improved survival rates, even among those with perineural involvement – a very poor prognostic factor. The 5-year local control rates were 90% for those with histologically proven perineural involvement and 57% for those with symptoms of perineural involvement (Head Neck 2009;31:604-10)
Dr. Hodgens had no financial disclosures.
SAN DIEGO – Aggressive skin cancers can recur, spread, and kill – and these lesions deserve special consideration after even an apparently successful Mohs procedure.
"Mohs is especially well suited to identify the rare patient who has a high-risk profile for local recurrence or metastasis," Dr. David Hodgens said at a meeting sponsored by the American Society for Mohs Surgery. "We can work together to develop radiotherapy techniques that treat both the primary site and the nodal basins to minimize toxicity and give the patient a better chance of a cure."
The Mohs surgeon is unlikely to see many patients who will need postoperative radiotherapy, said Dr. Hodgens, director of radiation oncology at Scripps Memorial Hospital, La Jolla, Calif. "Keep in mind the vast majority of patients will not need this – but be on the lookout for those who might."
Any skin cancer – squamous or basal cell carcinoma, Merkel cell tumor, melanoma or angiosarcoma – can be increasingly dangerous if certain risk factors are present, Dr. Hodgens said. Risk factors for local recurrence or metastatic spread include a lesion size greater than 2 cm; depth greater then 4 mm or a Clark level IV-V; location on the ear or lip, or within a scar; or perineural or periosteal invasion. Immunosuppressed patients are at particularly high risk, as are those with an already recurrent lesion. "A lesion that has recurred after prior treatment is one of the greatest risks of further recurrence or metastatic spread," he stressed.
Electron beam radiation is the best treatment for high-risk skin cancers. "Electrons are essential in treating the postoperative Mohs patient. If your radiation oncologist is not using electrons, find one who is," he said.
High-energy x-rays are also used, but mostly to cover a nodal basin where spread is suspected. But electrons have a unique property that allows a narrow beam to deliver 90% of its energy to a prespecified depth and then very rapidly decrease that energy as the beam goes deeper into the tissue. "Electrons provide penetration to a desired depth without the through transmission of x-rays," Dr. Hodgens said.
Post-Mohs radiation calls for a large treatment field that encompasses the entire surgical field and generous margins. "We try to get at least 2 cm around the surgical bed," he said. Patients typically receive daily treatments for 5-7 weeks, with 1.8-2 Gy/day to the primary site. The total radiation dose is usually around 50-60 Gy.
"We treat them as seriously as we do any of our patients with breast, lung, or brain tumors," Dr. Hodgens said. "They have the potential to die of their skin cancer. This is a long aggressive course of treatment designed to give the best chance of cure."
X-rays can be added to treat the associated nodal basin, if nodal spread is a concern. "The toxicity of adding nodal radiation is minimal," Dr. Hodgens said. "It’s very well tolerated, with mostly just mild erythema. It’s really an easy thing to do and very worthwhile."
There are few studies published on postoperative radiation for strict indications after well-performed Mohs surgery. Most recently, a Greek study of 315 patients found that post-Mohs radiation was associated with a 92% reduced risk of recurrence (Eur. J. Cancer 2010;46:1563-72).
Last year, an Australian study of 118 patients with cutaneous head or neck cancers found that surgery plus radiotherapy improved survival rates, even among those with perineural involvement – a very poor prognostic factor. The 5-year local control rates were 90% for those with histologically proven perineural involvement and 57% for those with symptoms of perineural involvement (Head Neck 2009;31:604-10)
Dr. Hodgens had no financial disclosures.
EXPERT ANALYSIS FROM A MEETING SPONSORED BY THE AMERICAN SOCIETY FOR MOHS SURGERY
Immunostaining Advances Up Melanocyte ID Efficiency
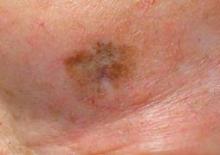
ORLANDO – Mohs surgery is an appropriate choice for lentigo maligna patients because advances in immunostaining allow easier and more efficient identification of melanocytes on frozen sections, said Dr. Basil S. Cherpelis.
Recent literature shows the recommended 5-mm margin for melanoma is often inadequate, Dr. Cherpelis said at the Orlando Dermatology Aesthetic and Clinical conference.
The margins needed for successful treatment of lentigo maligna vary – 5 mm is enough for some melanomas, while others might need a 1-cm margin.
"It makes sense to use a method of intraoperative margin control," said Dr. Cherpelis, of the Moffitt Cancer Center at the University of South Florida in Tampa. "That’s what gives you the flexibility to adjust your margins based on the melanoma that you happen to be treating."
One option for intraoperative margin control is traditional frozen sections, Dr. Cherpelis said. But frozen sections offer a limited view of the margin, and they pose the problem of freeze artifacts, which can make the distinction between melanocytes and keratinocytes difficult. A second option is the square or perimeter method, but this method involves a 1-2 week wait for complete results, which is inconvenient for doctors and patients.
A third option is Mohs surgery. Literature shows that Mohs is effective for melanoma, with the benefits of entire visualization and same-day reconstruction, but it is not widely used, Dr. Cherpelis said. One reason: Melanomas on permanent sections have halos around them, making identification easy, but frozen sections have similar halos around keratinocytes and melanocytes.
Using immunostaining as part of Mohs surgery can solve the identification problem, but until recently immunostain protocols could take at least an hour.
Dr. Cherpelis and his colleagues have streamlined the process.
"We have been able to shorten our protocols down to 19 minutes for MART-1 [melanoma-associated antigen recognized by T cells] and 35 minutes for MITF [microphthalmia-associated transcription factor]," Dr. Cherpelis said (Derm. Surg. 2009;35:207-13; Am. J. Dermatopath. 2010; 32:319-25, respectively). The protocols make Mohs surgery easier because the immunostain helps clinicians easily identify melanocytes.
MART-1 (a cytoplasmic stain) is the most common immunostain, but some data suggest it can falsely label keratinocytes in inflamed skin or in pigmented actinic keratoses, Dr. Cherpelis said. It also can cause pseudo-confluence (the appearance of touching melanocytes). By contrast, MITF is a nuclear stain, so it does not have the problem of pseudo-confluence.
Dr. Cherpelis said he had no relevant financial disclosures.
ORLANDO – Mohs surgery is an appropriate choice for lentigo maligna patients because advances in immunostaining allow easier and more efficient identification of melanocytes on frozen sections, said Dr. Basil S. Cherpelis.
Recent literature shows the recommended 5-mm margin for melanoma is often inadequate, Dr. Cherpelis said at the Orlando Dermatology Aesthetic and Clinical conference.
The margins needed for successful treatment of lentigo maligna vary – 5 mm is enough for some melanomas, while others might need a 1-cm margin.
"It makes sense to use a method of intraoperative margin control," said Dr. Cherpelis, of the Moffitt Cancer Center at the University of South Florida in Tampa. "That’s what gives you the flexibility to adjust your margins based on the melanoma that you happen to be treating."
One option for intraoperative margin control is traditional frozen sections, Dr. Cherpelis said. But frozen sections offer a limited view of the margin, and they pose the problem of freeze artifacts, which can make the distinction between melanocytes and keratinocytes difficult. A second option is the square or perimeter method, but this method involves a 1-2 week wait for complete results, which is inconvenient for doctors and patients.
A third option is Mohs surgery. Literature shows that Mohs is effective for melanoma, with the benefits of entire visualization and same-day reconstruction, but it is not widely used, Dr. Cherpelis said. One reason: Melanomas on permanent sections have halos around them, making identification easy, but frozen sections have similar halos around keratinocytes and melanocytes.
Using immunostaining as part of Mohs surgery can solve the identification problem, but until recently immunostain protocols could take at least an hour.
Dr. Cherpelis and his colleagues have streamlined the process.
"We have been able to shorten our protocols down to 19 minutes for MART-1 [melanoma-associated antigen recognized by T cells] and 35 minutes for MITF [microphthalmia-associated transcription factor]," Dr. Cherpelis said (Derm. Surg. 2009;35:207-13; Am. J. Dermatopath. 2010; 32:319-25, respectively). The protocols make Mohs surgery easier because the immunostain helps clinicians easily identify melanocytes.
MART-1 (a cytoplasmic stain) is the most common immunostain, but some data suggest it can falsely label keratinocytes in inflamed skin or in pigmented actinic keratoses, Dr. Cherpelis said. It also can cause pseudo-confluence (the appearance of touching melanocytes). By contrast, MITF is a nuclear stain, so it does not have the problem of pseudo-confluence.
Dr. Cherpelis said he had no relevant financial disclosures.
ORLANDO – Mohs surgery is an appropriate choice for lentigo maligna patients because advances in immunostaining allow easier and more efficient identification of melanocytes on frozen sections, said Dr. Basil S. Cherpelis.
Recent literature shows the recommended 5-mm margin for melanoma is often inadequate, Dr. Cherpelis said at the Orlando Dermatology Aesthetic and Clinical conference.
The margins needed for successful treatment of lentigo maligna vary – 5 mm is enough for some melanomas, while others might need a 1-cm margin.
"It makes sense to use a method of intraoperative margin control," said Dr. Cherpelis, of the Moffitt Cancer Center at the University of South Florida in Tampa. "That’s what gives you the flexibility to adjust your margins based on the melanoma that you happen to be treating."
One option for intraoperative margin control is traditional frozen sections, Dr. Cherpelis said. But frozen sections offer a limited view of the margin, and they pose the problem of freeze artifacts, which can make the distinction between melanocytes and keratinocytes difficult. A second option is the square or perimeter method, but this method involves a 1-2 week wait for complete results, which is inconvenient for doctors and patients.
A third option is Mohs surgery. Literature shows that Mohs is effective for melanoma, with the benefits of entire visualization and same-day reconstruction, but it is not widely used, Dr. Cherpelis said. One reason: Melanomas on permanent sections have halos around them, making identification easy, but frozen sections have similar halos around keratinocytes and melanocytes.
Using immunostaining as part of Mohs surgery can solve the identification problem, but until recently immunostain protocols could take at least an hour.
Dr. Cherpelis and his colleagues have streamlined the process.
"We have been able to shorten our protocols down to 19 minutes for MART-1 [melanoma-associated antigen recognized by T cells] and 35 minutes for MITF [microphthalmia-associated transcription factor]," Dr. Cherpelis said (Derm. Surg. 2009;35:207-13; Am. J. Dermatopath. 2010; 32:319-25, respectively). The protocols make Mohs surgery easier because the immunostain helps clinicians easily identify melanocytes.
MART-1 (a cytoplasmic stain) is the most common immunostain, but some data suggest it can falsely label keratinocytes in inflamed skin or in pigmented actinic keratoses, Dr. Cherpelis said. It also can cause pseudo-confluence (the appearance of touching melanocytes). By contrast, MITF is a nuclear stain, so it does not have the problem of pseudo-confluence.
Dr. Cherpelis said he had no relevant financial disclosures.
EXPERT ANALYSIS FROM THE ORLANDO DERMATOLOGY AESTHETIC & CLINICAL CONFERENCE
Cosmetic Outcomes of Treatments for Actinic Keratoses: An Emerging Endpoint of Therapy
Melanoma Vaccine Gets Orphan Designation
The Food and Drug Administration has awarded orphan drug designation to BioSante Pharmaceuticals' melanoma cancer vaccine.
The vaccine will be used to treat stage IIb-IV melanoma, says the company, in a statement. The Lincolnshire, Ill.–based BioSante estimates that 68,000 people were diagnosed with melanoma in 2010, and that 8,700 died. The FDA grants orphan status for products intended to treat fewer than 200,000 patients in the United States. According to the agency, makers of orphan products are eligible for tax credits and marketing incentives, but they still have to submit the same efficacy and safety data required of other products. Orphan status does not change the timetable for approval.
The BioSante vaccine is in early development at the Johns Hopkins Sidney Kimmel Comprehensive Cancer Center. It is a "GVAX" vaccine. According to a BioSante statement, it uses cell lines that are genetically modified to secrete granulocyte-macrophage colony–stimulating factor (GM-CSF). The cells are then irradiated to prevent further cell division. The GVAX model is also being used in BioSante products in development for pancreatic cancer, acute myeloid leukemia, and chronic myeloid leukemia.
The Food and Drug Administration has awarded orphan drug designation to BioSante Pharmaceuticals' melanoma cancer vaccine.
The vaccine will be used to treat stage IIb-IV melanoma, says the company, in a statement. The Lincolnshire, Ill.–based BioSante estimates that 68,000 people were diagnosed with melanoma in 2010, and that 8,700 died. The FDA grants orphan status for products intended to treat fewer than 200,000 patients in the United States. According to the agency, makers of orphan products are eligible for tax credits and marketing incentives, but they still have to submit the same efficacy and safety data required of other products. Orphan status does not change the timetable for approval.
The BioSante vaccine is in early development at the Johns Hopkins Sidney Kimmel Comprehensive Cancer Center. It is a "GVAX" vaccine. According to a BioSante statement, it uses cell lines that are genetically modified to secrete granulocyte-macrophage colony–stimulating factor (GM-CSF). The cells are then irradiated to prevent further cell division. The GVAX model is also being used in BioSante products in development for pancreatic cancer, acute myeloid leukemia, and chronic myeloid leukemia.
The Food and Drug Administration has awarded orphan drug designation to BioSante Pharmaceuticals' melanoma cancer vaccine.
The vaccine will be used to treat stage IIb-IV melanoma, says the company, in a statement. The Lincolnshire, Ill.–based BioSante estimates that 68,000 people were diagnosed with melanoma in 2010, and that 8,700 died. The FDA grants orphan status for products intended to treat fewer than 200,000 patients in the United States. According to the agency, makers of orphan products are eligible for tax credits and marketing incentives, but they still have to submit the same efficacy and safety data required of other products. Orphan status does not change the timetable for approval.
The BioSante vaccine is in early development at the Johns Hopkins Sidney Kimmel Comprehensive Cancer Center. It is a "GVAX" vaccine. According to a BioSante statement, it uses cell lines that are genetically modified to secrete granulocyte-macrophage colony–stimulating factor (GM-CSF). The cells are then irradiated to prevent further cell division. The GVAX model is also being used in BioSante products in development for pancreatic cancer, acute myeloid leukemia, and chronic myeloid leukemia.
Sunless Tanners
Coloring the skin with dihydroxyacetone to create an artificial tan was discovered serendipitously in the 1950s by Eva Wittgenstein, whose patients were taking DHA orally. She observed that when patients regurgitated the DHA, pigmented spots remained on the skin (Science 1960;132:894-5).
The change in pigmentation results from the interaction of DHA with amino acids in the stratum corneum (Br. J. Dermatol. 2003;149:332-40).
Although the first product brought to market to exploit this new knowledge in 1959 met with initial success, it fell into disuse because of the poor cosmetic results (J. Am. Acad. Dermatol. 2003;49:1096-106; South. Med. J. 2005;98:1192-5).
Public awareness of sunless tanning has grown in recent years, however, and has been met with formulations that afford a much improved cosmetic performance, with DHA remaining as the main active ingredient in sunless-tanning agents (South. Med. J. 2005;98:1192-5; Am. J. Clin. Dermatol. 2002;3:317-8).
This column will briefly review the role of sunless-tanning lotions and sprays in dermatology, and will try to place recommendations to patients about sunless tanners in the context of sunscreens and tanning attitudes.
Sunless tanners have been used, with varying degrees of popularity, for 50 years; recent evidence suggests that the use of these products has increased in recent years. A long track record of usage and research indicates that sunless-tanning compounds are safe (J. Environ. Pathol. Toxicol. Oncol. 1984;5:349-51; Am. J. Clin. Dermatol. 2002;3:317-8; South. Med. J. 2005;98:1192-5; J. Am. Acad. Dermatol. 2007;56:387-90.) However, there is some debate as to whether the use of these products renders users more inclined to stay in the sun longer. Also, ongoing research has raised some safety issues regarding DHA.
Chemical Protection
In 2004, Petersen et al. investigated the effects of DHA on cell survival and proliferation of a human keratinocyte cell line, HaCaT. Significant genotoxic activity was identified in these cultured cells, as researchers noted dose- and time-dependent morphologic alterations as well as cytoplasmic budding and cell detachment in DHA-treated cells, and apoptosis and marked declines in proliferation 1 day after DHA exposure. Interestingly, the development of DNA strand breaks was prevented via preincubation with antioxidants. The authors suggested that their findings raised concerns about the long-term use of treating the skin with DHA-containing formulations (Mutat. Res. 2004;560:173-86).
A small study by Faurschou et al. showed that DHA provided human skin with modest UVB protection, with a 5% DHA cream applied three times or a 20% DHA cream applied once yielding an effect similar to an SPF 1.6. They acknowledged that most commercial DHA products contain 3%-5% DHA and offer scant protection. A potential advantage of sunless tanners, if they offer protection, suggested the authors, would be that no spots would be left unattended since such gaps would be conspicuous for lack of browning (Arch. Dermatol. 2004;140:886-7).
Howe, Reed, and Dellavalle contended that DHA in self-tanning agents provided enough UVA protection to warrant use as an adjunct to daily sunscreen use for broad-band protection (J. Am. Acad. Dermatol. 2008;58:894). The researchers referenced a 1975 study in which 3% DHA augmented sunlight tolerance among five patients with UVA-sensitive dermatoses (Dermatologica 1975;150:346-51; J. Am. Acad. Dermatol. 2008;58:894).
In 2009, Choquenet et al. assessed the sun protection potency of self-tanners and foundations using an in vitro method to identify indicators such as sun protection factor (SPF), UVA protection factor (PF-UVA), and UVB/UVA ratio. Using seven amino acids in the corneal cells of the epidermis, the investigators identified SPFs virtually equivalent to zero (approximately SPF 2). Conversely, they found foundations to exhibit satisfactory photostability and a non-negligible SPF, noting that such products are safe given their typically once-daily application (J. Dermatol. 2009;36:587-91).
Attitudes and Behavior
Users of sunless-tanning products were found, in a survey of 2,005 randomly selected South Australian adults published in 2001, to be more likely to use sunscreen but less likely to wear a hat or other sun-protective clothing. Sunburns were also linked to the use of sunless tanners in this study (Med. J. Aust. 2001;174:75-8; J. Am. Acad. Dermatol. 2007;56:387-90).
In 2005, Mahler et al. conducted a randomized, controlled trial with 1-month follow-up of 146 Southern California college students to study the impact of UV photoaging photographs and data on the sun protection plans and behaviors of young adults, and whether sunless-tanning lotion might play a role in sun-protective behavior. Of the 146 volunteers, 91.1% completed the "surprise" 1-month follow-up. The intervention consisted of volunteers being shown a UV facial photo and short video illustrating the etiology and results of photoaging. The investigators found that the UV photographic intervention was successful insofar as significantly better sun protection strategies and behaviors were employed by the test group, compared with controls. In addition, they noted that those who used sunless-tanning lotion were more likely to exhibit extensive sun-protective behavior, compared with individuals who received the intervention alone. The authors suggested that such interventions represented a cost-effective approach that might yield behaviors with long-term health benefits in the form of a lower incidence of skin cancer (Arch. Dermatol. 2005;141:373-80).
Later that year, Sheehan and Lesher reported on their anonymous survey of 121 adults (107 women and 14 men) who received spray-on sunless-tanning treatments between February and May 2004. Most reported that sunless tanning had not or would not affect their use of sunscreen or time spent outdoors. Interestingly, though, 73% of the respondents who had used tanning beds indicated that they had reduced or would reduce their use of indoor tanning. The researchers concluded that practitioners should recommend sunless tanning to their patients who use conventional tanning beds as a way to lower their solar exposure and risk of skin cancer (South. Med. J. 2005;98:1192-5).
In 2006, Brooks et al. reported on their July 2004, greater Boston, cross-sectional survey of 448 nonrandomly selected people aged 18-30 years on the subjects of artificial tanning products, recent history of sunburns, and tanning-bed use. During the preceding year, 22% of respondents had used sunless-tanning products and the same percentage hadn’t but would consider using them in the next year. Users of these lotions were more likely to have been severe burners and, after controlling for skin type, the investigators found that previous and potential users were more likely to have experienced sunburns during the summer and to have used tanning beds than were respondents who had neither used nor planned to use sunless tanners. The researchers concluded that the option of sunless tanning did not seem to lower the incidence of sunburn or the use of tanning beds. Further, they urged caution in offering broad support for artificial tanning products, even though looking for alternatives to UV exposure is to be encouraged (J. Am. Acad. Dermatol. 2006;54:1060-6).
A 2007 study using data from 5,491 completed interviews as part of the National Cancer Institute’s HINTS (Health Information National Trends Survey) 2005 set out to determine the prevalence of sunless-tanning formulation use and to understand the relationship between sunless tanning, indoor tanning, and sun-protective behavior. In so doing, the investigators found that the use of sunless-tanning products was relatively rare, with an estimated 11% of U.S. adults claiming to have used such products in the previous year. Of this group, 13% used sunless tanners more than 25 times; 12% used them 11-24 times; 35% used them 3-10 times; and 40% used them just once or twice. Users and exclusive users were more likely to be women, well educated, and living in the West (J. Am. Acad. Dermatol. 2007;56:387-90).
Recent Cause for Pause
In 2008, Jung et al. used the electron spin resonance spectroscopy–based radical sun protection factor, which measures free-radical reactions in skin biopsies during UV exposure and can determine the protective effect of UV filters and sunscreens, to analyze three different self-tanning agents, including DHA. Noting that the reaction of the reducing sugars in such products and amino acids (Maillard reaction) in the skin layer engendered Amadori products that created free radicals during UV irradiation, the investigators reported that in DHA-treated skin, more than 180% additional radicals were produced during sun exposure, compared with untreated skin. They concluded that the use of self-tanners necessitated reducing solar exposure duration (Spectrochim. Acta A. Mol. Biomol Spectrosc. 2008;69:1423-8). Consequently, DHA users would be well advised to wait at least 24 hours before solar exposure following application of sunless lotion or spray (Dermatol. Clin. 2009;27:149-54).
Conclusion
In addition to the need to continually persuade patients to engage in skin-protective behavior – namely, limiting sun exposure, avoiding peak-time exposures, wearing wide-brimmed hats and clothing with SPF value, and using sunscreens (preferably those containing avobenzone, Mexoryl, zinc oxide, or titanium dioxide) every day – it is incumbent upon dermatologists to disabuse our patients of the idea that untanned skin is less attractive or, conversely, that tans should be equated with attractive skin. Although healthy messages (that there is no such thing as a healthy tan, and that a tan indicates skin damage) are slowly spreading, sunless tanners appear to be a medically and cosmetically acceptable intermediary step, far surpassing the patently detrimental effects of indoor tanning. That said, patients should be advised to refrain from solar exposure for a full 24 hours after applying sunless tanners. Furthermore, much more research is necessary to truly establish that DHA delivers even modest protection from UVA or UVB. Accordingly, patients should be counseled that sunless tanners do not suffice as a base, nor do they justify extended solar exposure, and that sunscreens should also be applied.
Coloring the skin with dihydroxyacetone to create an artificial tan was discovered serendipitously in the 1950s by Eva Wittgenstein, whose patients were taking DHA orally. She observed that when patients regurgitated the DHA, pigmented spots remained on the skin (Science 1960;132:894-5).
The change in pigmentation results from the interaction of DHA with amino acids in the stratum corneum (Br. J. Dermatol. 2003;149:332-40).
Although the first product brought to market to exploit this new knowledge in 1959 met with initial success, it fell into disuse because of the poor cosmetic results (J. Am. Acad. Dermatol. 2003;49:1096-106; South. Med. J. 2005;98:1192-5).
Public awareness of sunless tanning has grown in recent years, however, and has been met with formulations that afford a much improved cosmetic performance, with DHA remaining as the main active ingredient in sunless-tanning agents (South. Med. J. 2005;98:1192-5; Am. J. Clin. Dermatol. 2002;3:317-8).
This column will briefly review the role of sunless-tanning lotions and sprays in dermatology, and will try to place recommendations to patients about sunless tanners in the context of sunscreens and tanning attitudes.
Sunless tanners have been used, with varying degrees of popularity, for 50 years; recent evidence suggests that the use of these products has increased in recent years. A long track record of usage and research indicates that sunless-tanning compounds are safe (J. Environ. Pathol. Toxicol. Oncol. 1984;5:349-51; Am. J. Clin. Dermatol. 2002;3:317-8; South. Med. J. 2005;98:1192-5; J. Am. Acad. Dermatol. 2007;56:387-90.) However, there is some debate as to whether the use of these products renders users more inclined to stay in the sun longer. Also, ongoing research has raised some safety issues regarding DHA.
Chemical Protection
In 2004, Petersen et al. investigated the effects of DHA on cell survival and proliferation of a human keratinocyte cell line, HaCaT. Significant genotoxic activity was identified in these cultured cells, as researchers noted dose- and time-dependent morphologic alterations as well as cytoplasmic budding and cell detachment in DHA-treated cells, and apoptosis and marked declines in proliferation 1 day after DHA exposure. Interestingly, the development of DNA strand breaks was prevented via preincubation with antioxidants. The authors suggested that their findings raised concerns about the long-term use of treating the skin with DHA-containing formulations (Mutat. Res. 2004;560:173-86).
A small study by Faurschou et al. showed that DHA provided human skin with modest UVB protection, with a 5% DHA cream applied three times or a 20% DHA cream applied once yielding an effect similar to an SPF 1.6. They acknowledged that most commercial DHA products contain 3%-5% DHA and offer scant protection. A potential advantage of sunless tanners, if they offer protection, suggested the authors, would be that no spots would be left unattended since such gaps would be conspicuous for lack of browning (Arch. Dermatol. 2004;140:886-7).
Howe, Reed, and Dellavalle contended that DHA in self-tanning agents provided enough UVA protection to warrant use as an adjunct to daily sunscreen use for broad-band protection (J. Am. Acad. Dermatol. 2008;58:894). The researchers referenced a 1975 study in which 3% DHA augmented sunlight tolerance among five patients with UVA-sensitive dermatoses (Dermatologica 1975;150:346-51; J. Am. Acad. Dermatol. 2008;58:894).
In 2009, Choquenet et al. assessed the sun protection potency of self-tanners and foundations using an in vitro method to identify indicators such as sun protection factor (SPF), UVA protection factor (PF-UVA), and UVB/UVA ratio. Using seven amino acids in the corneal cells of the epidermis, the investigators identified SPFs virtually equivalent to zero (approximately SPF 2). Conversely, they found foundations to exhibit satisfactory photostability and a non-negligible SPF, noting that such products are safe given their typically once-daily application (J. Dermatol. 2009;36:587-91).
Attitudes and Behavior
Users of sunless-tanning products were found, in a survey of 2,005 randomly selected South Australian adults published in 2001, to be more likely to use sunscreen but less likely to wear a hat or other sun-protective clothing. Sunburns were also linked to the use of sunless tanners in this study (Med. J. Aust. 2001;174:75-8; J. Am. Acad. Dermatol. 2007;56:387-90).
In 2005, Mahler et al. conducted a randomized, controlled trial with 1-month follow-up of 146 Southern California college students to study the impact of UV photoaging photographs and data on the sun protection plans and behaviors of young adults, and whether sunless-tanning lotion might play a role in sun-protective behavior. Of the 146 volunteers, 91.1% completed the "surprise" 1-month follow-up. The intervention consisted of volunteers being shown a UV facial photo and short video illustrating the etiology and results of photoaging. The investigators found that the UV photographic intervention was successful insofar as significantly better sun protection strategies and behaviors were employed by the test group, compared with controls. In addition, they noted that those who used sunless-tanning lotion were more likely to exhibit extensive sun-protective behavior, compared with individuals who received the intervention alone. The authors suggested that such interventions represented a cost-effective approach that might yield behaviors with long-term health benefits in the form of a lower incidence of skin cancer (Arch. Dermatol. 2005;141:373-80).
Later that year, Sheehan and Lesher reported on their anonymous survey of 121 adults (107 women and 14 men) who received spray-on sunless-tanning treatments between February and May 2004. Most reported that sunless tanning had not or would not affect their use of sunscreen or time spent outdoors. Interestingly, though, 73% of the respondents who had used tanning beds indicated that they had reduced or would reduce their use of indoor tanning. The researchers concluded that practitioners should recommend sunless tanning to their patients who use conventional tanning beds as a way to lower their solar exposure and risk of skin cancer (South. Med. J. 2005;98:1192-5).
In 2006, Brooks et al. reported on their July 2004, greater Boston, cross-sectional survey of 448 nonrandomly selected people aged 18-30 years on the subjects of artificial tanning products, recent history of sunburns, and tanning-bed use. During the preceding year, 22% of respondents had used sunless-tanning products and the same percentage hadn’t but would consider using them in the next year. Users of these lotions were more likely to have been severe burners and, after controlling for skin type, the investigators found that previous and potential users were more likely to have experienced sunburns during the summer and to have used tanning beds than were respondents who had neither used nor planned to use sunless tanners. The researchers concluded that the option of sunless tanning did not seem to lower the incidence of sunburn or the use of tanning beds. Further, they urged caution in offering broad support for artificial tanning products, even though looking for alternatives to UV exposure is to be encouraged (J. Am. Acad. Dermatol. 2006;54:1060-6).
A 2007 study using data from 5,491 completed interviews as part of the National Cancer Institute’s HINTS (Health Information National Trends Survey) 2005 set out to determine the prevalence of sunless-tanning formulation use and to understand the relationship between sunless tanning, indoor tanning, and sun-protective behavior. In so doing, the investigators found that the use of sunless-tanning products was relatively rare, with an estimated 11% of U.S. adults claiming to have used such products in the previous year. Of this group, 13% used sunless tanners more than 25 times; 12% used them 11-24 times; 35% used them 3-10 times; and 40% used them just once or twice. Users and exclusive users were more likely to be women, well educated, and living in the West (J. Am. Acad. Dermatol. 2007;56:387-90).
Recent Cause for Pause
In 2008, Jung et al. used the electron spin resonance spectroscopy–based radical sun protection factor, which measures free-radical reactions in skin biopsies during UV exposure and can determine the protective effect of UV filters and sunscreens, to analyze three different self-tanning agents, including DHA. Noting that the reaction of the reducing sugars in such products and amino acids (Maillard reaction) in the skin layer engendered Amadori products that created free radicals during UV irradiation, the investigators reported that in DHA-treated skin, more than 180% additional radicals were produced during sun exposure, compared with untreated skin. They concluded that the use of self-tanners necessitated reducing solar exposure duration (Spectrochim. Acta A. Mol. Biomol Spectrosc. 2008;69:1423-8). Consequently, DHA users would be well advised to wait at least 24 hours before solar exposure following application of sunless lotion or spray (Dermatol. Clin. 2009;27:149-54).
Conclusion
In addition to the need to continually persuade patients to engage in skin-protective behavior – namely, limiting sun exposure, avoiding peak-time exposures, wearing wide-brimmed hats and clothing with SPF value, and using sunscreens (preferably those containing avobenzone, Mexoryl, zinc oxide, or titanium dioxide) every day – it is incumbent upon dermatologists to disabuse our patients of the idea that untanned skin is less attractive or, conversely, that tans should be equated with attractive skin. Although healthy messages (that there is no such thing as a healthy tan, and that a tan indicates skin damage) are slowly spreading, sunless tanners appear to be a medically and cosmetically acceptable intermediary step, far surpassing the patently detrimental effects of indoor tanning. That said, patients should be advised to refrain from solar exposure for a full 24 hours after applying sunless tanners. Furthermore, much more research is necessary to truly establish that DHA delivers even modest protection from UVA or UVB. Accordingly, patients should be counseled that sunless tanners do not suffice as a base, nor do they justify extended solar exposure, and that sunscreens should also be applied.
Coloring the skin with dihydroxyacetone to create an artificial tan was discovered serendipitously in the 1950s by Eva Wittgenstein, whose patients were taking DHA orally. She observed that when patients regurgitated the DHA, pigmented spots remained on the skin (Science 1960;132:894-5).
The change in pigmentation results from the interaction of DHA with amino acids in the stratum corneum (Br. J. Dermatol. 2003;149:332-40).
Although the first product brought to market to exploit this new knowledge in 1959 met with initial success, it fell into disuse because of the poor cosmetic results (J. Am. Acad. Dermatol. 2003;49:1096-106; South. Med. J. 2005;98:1192-5).
Public awareness of sunless tanning has grown in recent years, however, and has been met with formulations that afford a much improved cosmetic performance, with DHA remaining as the main active ingredient in sunless-tanning agents (South. Med. J. 2005;98:1192-5; Am. J. Clin. Dermatol. 2002;3:317-8).
This column will briefly review the role of sunless-tanning lotions and sprays in dermatology, and will try to place recommendations to patients about sunless tanners in the context of sunscreens and tanning attitudes.
Sunless tanners have been used, with varying degrees of popularity, for 50 years; recent evidence suggests that the use of these products has increased in recent years. A long track record of usage and research indicates that sunless-tanning compounds are safe (J. Environ. Pathol. Toxicol. Oncol. 1984;5:349-51; Am. J. Clin. Dermatol. 2002;3:317-8; South. Med. J. 2005;98:1192-5; J. Am. Acad. Dermatol. 2007;56:387-90.) However, there is some debate as to whether the use of these products renders users more inclined to stay in the sun longer. Also, ongoing research has raised some safety issues regarding DHA.
Chemical Protection
In 2004, Petersen et al. investigated the effects of DHA on cell survival and proliferation of a human keratinocyte cell line, HaCaT. Significant genotoxic activity was identified in these cultured cells, as researchers noted dose- and time-dependent morphologic alterations as well as cytoplasmic budding and cell detachment in DHA-treated cells, and apoptosis and marked declines in proliferation 1 day after DHA exposure. Interestingly, the development of DNA strand breaks was prevented via preincubation with antioxidants. The authors suggested that their findings raised concerns about the long-term use of treating the skin with DHA-containing formulations (Mutat. Res. 2004;560:173-86).
A small study by Faurschou et al. showed that DHA provided human skin with modest UVB protection, with a 5% DHA cream applied three times or a 20% DHA cream applied once yielding an effect similar to an SPF 1.6. They acknowledged that most commercial DHA products contain 3%-5% DHA and offer scant protection. A potential advantage of sunless tanners, if they offer protection, suggested the authors, would be that no spots would be left unattended since such gaps would be conspicuous for lack of browning (Arch. Dermatol. 2004;140:886-7).
Howe, Reed, and Dellavalle contended that DHA in self-tanning agents provided enough UVA protection to warrant use as an adjunct to daily sunscreen use for broad-band protection (J. Am. Acad. Dermatol. 2008;58:894). The researchers referenced a 1975 study in which 3% DHA augmented sunlight tolerance among five patients with UVA-sensitive dermatoses (Dermatologica 1975;150:346-51; J. Am. Acad. Dermatol. 2008;58:894).
In 2009, Choquenet et al. assessed the sun protection potency of self-tanners and foundations using an in vitro method to identify indicators such as sun protection factor (SPF), UVA protection factor (PF-UVA), and UVB/UVA ratio. Using seven amino acids in the corneal cells of the epidermis, the investigators identified SPFs virtually equivalent to zero (approximately SPF 2). Conversely, they found foundations to exhibit satisfactory photostability and a non-negligible SPF, noting that such products are safe given their typically once-daily application (J. Dermatol. 2009;36:587-91).
Attitudes and Behavior
Users of sunless-tanning products were found, in a survey of 2,005 randomly selected South Australian adults published in 2001, to be more likely to use sunscreen but less likely to wear a hat or other sun-protective clothing. Sunburns were also linked to the use of sunless tanners in this study (Med. J. Aust. 2001;174:75-8; J. Am. Acad. Dermatol. 2007;56:387-90).
In 2005, Mahler et al. conducted a randomized, controlled trial with 1-month follow-up of 146 Southern California college students to study the impact of UV photoaging photographs and data on the sun protection plans and behaviors of young adults, and whether sunless-tanning lotion might play a role in sun-protective behavior. Of the 146 volunteers, 91.1% completed the "surprise" 1-month follow-up. The intervention consisted of volunteers being shown a UV facial photo and short video illustrating the etiology and results of photoaging. The investigators found that the UV photographic intervention was successful insofar as significantly better sun protection strategies and behaviors were employed by the test group, compared with controls. In addition, they noted that those who used sunless-tanning lotion were more likely to exhibit extensive sun-protective behavior, compared with individuals who received the intervention alone. The authors suggested that such interventions represented a cost-effective approach that might yield behaviors with long-term health benefits in the form of a lower incidence of skin cancer (Arch. Dermatol. 2005;141:373-80).
Later that year, Sheehan and Lesher reported on their anonymous survey of 121 adults (107 women and 14 men) who received spray-on sunless-tanning treatments between February and May 2004. Most reported that sunless tanning had not or would not affect their use of sunscreen or time spent outdoors. Interestingly, though, 73% of the respondents who had used tanning beds indicated that they had reduced or would reduce their use of indoor tanning. The researchers concluded that practitioners should recommend sunless tanning to their patients who use conventional tanning beds as a way to lower their solar exposure and risk of skin cancer (South. Med. J. 2005;98:1192-5).
In 2006, Brooks et al. reported on their July 2004, greater Boston, cross-sectional survey of 448 nonrandomly selected people aged 18-30 years on the subjects of artificial tanning products, recent history of sunburns, and tanning-bed use. During the preceding year, 22% of respondents had used sunless-tanning products and the same percentage hadn’t but would consider using them in the next year. Users of these lotions were more likely to have been severe burners and, after controlling for skin type, the investigators found that previous and potential users were more likely to have experienced sunburns during the summer and to have used tanning beds than were respondents who had neither used nor planned to use sunless tanners. The researchers concluded that the option of sunless tanning did not seem to lower the incidence of sunburn or the use of tanning beds. Further, they urged caution in offering broad support for artificial tanning products, even though looking for alternatives to UV exposure is to be encouraged (J. Am. Acad. Dermatol. 2006;54:1060-6).
A 2007 study using data from 5,491 completed interviews as part of the National Cancer Institute’s HINTS (Health Information National Trends Survey) 2005 set out to determine the prevalence of sunless-tanning formulation use and to understand the relationship between sunless tanning, indoor tanning, and sun-protective behavior. In so doing, the investigators found that the use of sunless-tanning products was relatively rare, with an estimated 11% of U.S. adults claiming to have used such products in the previous year. Of this group, 13% used sunless tanners more than 25 times; 12% used them 11-24 times; 35% used them 3-10 times; and 40% used them just once or twice. Users and exclusive users were more likely to be women, well educated, and living in the West (J. Am. Acad. Dermatol. 2007;56:387-90).
Recent Cause for Pause
In 2008, Jung et al. used the electron spin resonance spectroscopy–based radical sun protection factor, which measures free-radical reactions in skin biopsies during UV exposure and can determine the protective effect of UV filters and sunscreens, to analyze three different self-tanning agents, including DHA. Noting that the reaction of the reducing sugars in such products and amino acids (Maillard reaction) in the skin layer engendered Amadori products that created free radicals during UV irradiation, the investigators reported that in DHA-treated skin, more than 180% additional radicals were produced during sun exposure, compared with untreated skin. They concluded that the use of self-tanners necessitated reducing solar exposure duration (Spectrochim. Acta A. Mol. Biomol Spectrosc. 2008;69:1423-8). Consequently, DHA users would be well advised to wait at least 24 hours before solar exposure following application of sunless lotion or spray (Dermatol. Clin. 2009;27:149-54).
Conclusion
In addition to the need to continually persuade patients to engage in skin-protective behavior – namely, limiting sun exposure, avoiding peak-time exposures, wearing wide-brimmed hats and clothing with SPF value, and using sunscreens (preferably those containing avobenzone, Mexoryl, zinc oxide, or titanium dioxide) every day – it is incumbent upon dermatologists to disabuse our patients of the idea that untanned skin is less attractive or, conversely, that tans should be equated with attractive skin. Although healthy messages (that there is no such thing as a healthy tan, and that a tan indicates skin damage) are slowly spreading, sunless tanners appear to be a medically and cosmetically acceptable intermediary step, far surpassing the patently detrimental effects of indoor tanning. That said, patients should be advised to refrain from solar exposure for a full 24 hours after applying sunless tanners. Furthermore, much more research is necessary to truly establish that DHA delivers even modest protection from UVA or UVB. Accordingly, patients should be counseled that sunless tanners do not suffice as a base, nor do they justify extended solar exposure, and that sunscreens should also be applied.
Cancer Breakthrough Pain in the Presence of Cancer-Related Chronic Pain: Fact versus Perceptions of Health-Care Providers and Patients
Review
Michelle I. Rhiner RN, MSN, ACHPN![]()
![]()
| Referred to by: | The Challenges of Treating Patients with Cancer Pain The Journal of Supportive Oncology, Volume 8, Issue 6, November-December 2010, Pages 239-240, Sloan Beth Karver, Jessalyn H. Berger | |
| Referred to by: | A Conceptual Solution to Improve the Management of Cancer-Related Breakthrough Pain The Journal of Supportive Oncology, Volume 8, Issue 6, November-December 2010, Page 241, Wendy Ledesma, Toby C. Campbell | |
Abstract
Cancer breakthrough pain is a flare in pain that “breaks through” well-controlled persistent cancer pain. Although the condition is highly prevalent, the concept of cancer breakthrough pain is not well understood and is therefore underdiagnosed and undertreated. The purpose of this review is to examine the roles the health-care practitioner and patient/family caregiver play in the undertreatment of breakthrough pain. A lack of technical knowledge about pain management and pain assessment, attitudes about opioid addiction, and regulatory guidelines influence the manner in which opioids are prescribed. Patients harbor a variety of fears and misconceptions, such as opioid addiction, tolerance, side effects, and the meaning of pain, which can create a barrier to effective communication with their health-care provider regarding their cancer pain management and specifically their breakthrough pain. Identifying these issues gives health-care professionals and patients an opportunity to develop strategies that can improve the treatment of cancer breakthrough pain.
Article Outline
Adapted from Payne R.10
| CATEGORY OF PAIN | CHARACTERISTICS |
|---|---|
| Neuropathic | Caused by structural changes in the central nervous system and the peripheral nervous system; described as tingling, burning, or shooting in nature |
| Visceral | Deep cramping and tearing pain that may originate in internal organs |
| Somatic | Requires skeletal involvement; described as constant throbbing and aching that increases with movement |
A significant number of patients suffering from cancer experience these pain flares. In a single-population evaluation of cancer pain (n = 159), it was reported that of those patients with continuous pain, 57 (75%) experienced cancer BTP. More than half (54%) of those patients who experienced cancer pain reported it being related to particular activities, while a little over one-quarter of patients (26%) experienced pain idiopathic in nature and 16% of patients experienced end-of-dose pain.3 In a survey of 545 patients with cancer experiencing fluctuations in pain conducted by the American Pain Foundation, 96% of these patients experienced episodes of cancer BTP at least once a month, more than 70% experienced cancer BTP episodes at least once a week, and more than one-fifth (22%) experienced cancer BTP more than once a day.11
The inadequate treatment of cancer pain is an issue with far-reaching implications; therefore, the purpose of this review is to examine the roles that health-care providers, patients, and family caregivers play in the undertreatment of cancer BTP.
Defining Cancer BTP
Despite the reported prevalence of the condition, questions continue to surround the definition of cancer BTP. The earliest clinical accounts of cancer BTP describe it as a greater than moderate temporary flare in pain that occurs on a baseline of moderate pain in patients receiving opioids for cancer pain.8 Presently, descriptions of cancer BTP include statements that flares may occur in the presence of stable persistent pain (regardless of the treatment)12 having pain levels ranging from moderate to severe, with onset depending on the subtype of pain.13
Some controversy centers on the term “breakthrough pain.” The American-English term “breakthrough pain” does not have an exact translation to other languages. Physicians in Europe may use the term “episodic” or “transient” to describe these fluctuations in cancer pain. However, these terms do not capture the idea that the pain is above and beyond persistent pain control (eg, “breaks through baseline analgesia”). Furthermore, if the term “breakthrough pain” is used, it may be limited to descriptions of pain that occur at the end of the dosing cycle.14 Finally, there are physicians in some countries who do not view exacerbation of cancer pain as a separate clinical entity. Instead, they view spikes of pain as a predictable or normal element of cancer pain.14
In spite of the questions surrounding cancer BTP, persistent pain must be controlled before management of cancer BTP can proceed. In addition, repeated episodes of cancer BTP may indicate that baseline pain has not been properly assessed and not adequately managed.10 Therefore, cancer BTP and persistent cancer pain should be assessed independently as they are separate clinical conditions.
Treatment Approaches to Persistent Cancer Pain versus Cancer BTP
Persistent cancer pain requires around-the-clock (ATC) treatment with therapeutic agents that both maximize outcomes and minimize risks. ATC treatment allows maintenance of drug concentrations and prevents peaks and troughs resulting in increased risk of toxicity and lack of efficacy. Opioids such as morphine, oxycodone, methadone, and fentanyl are used extensively for the treatment of cancer pain as they produce an analgesic effect at a minimum dose and are easily titrated.15 While opioids are administrated orally per World Health Organization recommendations,15 they may also be administered rectally, intravenously, subcutaneously, intramuscularly, and transdermally. Clinical trials are being conducted to examine the administration of opioids through inhalation.16 Methadone is an example of a long-acting opioid that has been used to treat persistent pain.17 Pharmaceutically long-acting drugs such as sustained-release formulations of morphine, oxycodone, oxymorphone, and hydromorphone have been used to treat cancer pain.18 Adjuvant medications may be coadministered with opioids to treat symptoms that occur concurrently with cancer pain and to augment the analgesic effect. Although few clinical trials have evaluated the efficacy of adjuvants in patients with cancer, local anesthetics (eg, lidocaine), nonsteroidal anti-inflammatory drugs (eg, aspirin, naproxen, and ibuprofen), nonopioid analgesics (eg, acetaminophen), antidepressants (eg, amitriptyline, duloxetine, and venlafaxine), and anticonvulsants (eg, gabapentin, pregabalin, valproate, and lamotrigine) have been used to supplement opioid therapy.12
A number of physical and cognitive–behavioral interventions may be used in addition to pharmacological treatments to alleviate some of the pain symptoms experienced in patients with persistent cancer pain. In most patients, heating pads or ice packs may be used to relieve pain and reduce swelling; however, neither heat nor cold should be used on irradiated tissue, and caution should be used when using ice packs on patients with peripheral vascular disease.19 Exercise may be started or continued to improve physical conditioning. While tumor masses should not be manipulated, other techniques requiring physical stimulation, such as massage, pressure, and vibration, may also be used in the treatment of persistent pain. Either moving an immobile patient or temporarily restricting movement can prevent or alleviate pain. Acupuncture is another treatment that may be used to treat persistent cancer pain. Psychosocial interventions include hypnosis, use of relaxation techniques, biofeedback, and cognitive distraction.19
Orally administered, short-acting or rapid-acting opioids are used to treat cancer BTP episodes. However, the pharmacokinetic profiles of oral opioids may not match the onset and duration of some cancer BTP episodes.20 An analgesic agent used to treat cancer BTP should match the temporal characteristics of cancer BTP, be easily titrated to higher or lower doses if needed, and, if used appropriately in opioid-tolerant patients, not be associated with undue adverse effects.
Oral transmucosal delivery of opioids is an option for these challenges. Three formulations of the opioid fentanyl are available for transmucosal delivery: oral transmucosal fentanyl citrate (OTFC),21 fentanyl buccal tablets (FBTs),22 and fentanyl buccal soluble film (FBSF).23 OTFC is a fentanyl lozenge that has demonstrated an analgesic effect within 15 minutes of administration. Despite its rapid onset of action, the amount of fentanyl administered with the OTFC lozenge depends on the education provided to the patient on the use of this medication and the ability of the patient to actively use this product.21 While FBT (a tablet that utilizes an effervescent reaction to improve absorption) does not require substantial patient participation, its use has been associated with application-site side effects.22 Like OTFC and FBT, FBSF offers a rapid onset of action but does not require active patient participation and has minimal oral adverse side effects.23 These oral transmucosal fentanyl products should be administered only to opioid-tolerant patients, to avoid the risk of life-threatening respiratory depression.
Physician-Related Factors in Clinical Inertia
Three elements shape physicians' perceptions of the severity of cancer BTP and influence prescribing practices: technical knowledge, attitudes concerning use of opioids, and regulatory restrictions.[24], [25], [26] and [27]
The medical training of most physicians may not include courses in pain management. Instead, practitioners' education is a result of inpatient experience during the postgraduate years and is generally geared toward management of acute injuries, postoperative pain, and cardiovascular events such as myocardial infarctions.28 More evidence of this lack of knowledge among physicians is presented in a survey of British Columbian physicians.29 Among those doctors who responded, only 32% were aware of the dose that would produce an equal analgesic effect when a switch between morphine and acetaminophen is required. Furthermore, only 55% of physicians correctly stated that doses of opioids for cancer BTP should be 10% of the total daily dose and administered every 1 or 2 hours as needed per National Comprehensive Cancer Network guidelines. These findings suggest that when patients need stronger analgesics, physicians may not be skilled in converting to a more potent opioid or calculating a dose for cancer BTP management.29 Indiscriminate polypharmacy presents additional complications, whereby coadministration of multiple opioids exposes patients to an increased risk of toxicity and distorts the clinician's interpretation of outcomes.30
In addition to training, the communication skills of the practitioner play a role in effective treatment of cancer BTP and persistent cancer pain. Discussions regarding treatment can significantly influence a patient's decision to use opioids. In an interview study conducted during a cancer pain management trial, patients were reportedly occasionally “suspicious about the idea of choice” in the use of opioids.31 These patients favored thorough discussions of pain treatment options with physicians who were knowledgeable and confident about the use of opioids. The likelihood of patients participating in a pain management trial also increased if the physician stated that a lower dose of opioid would be used initially and treatment would cease if side effects developed.
Health-care providers' attitudes about the side effects of opioid treatment also impact treatment of persistent cancer pain and cancer BTP.32 In addition, although clinical studies do not support the assertion that patients may become addicted to opioids used to treat acute and persistent cancer pain, the belief that these patients are at risk for addiction prevents some health-care providers from prescribing opioids.33 The perceived relationship between opioid tolerance and addiction is one of the sources of confusion. One of the complications of opioid therapy is pharmacological tolerance, the loss of analgesic effect. Tolerance to an opioid may be determined genetically or acquired after metabolic changes, changes to receptors, or by learned behavior.33 The result of tolerance is decreasing pain relief, in spite of persistent doses over time.[34] and [35] Pharmacological tolerance is associated with incomplete cross-tolerance to other opioids. In contrast, addiction is a psychological syndrome characterized by uncontrolled and persistent use of the drug despite harm.[34], [35] and [36] Physical dependence on opioids is characterized by a withdrawal syndrome that occurs when the dose is stopped or decreased suddenly or an antagonist is administered.36 Pharmacological dependence is not synonymous with addiction; pharmacological tolerance occurs with many commonly prescribed drugs (such as nitrates) and does not indicate addiction.34
The prevalence of opioid addiction among a population of patients with cancer pain varies. In a meta-review of opioid addiction studies, authors reported a 0%–7.7% prevalence rate of addiction in patients with cancer depending on the population studied and the diagnostic measure used.37 This corresponds with the rate of addiction prevalent in the population at large—it does not support a cause-and-effect relationship between opioid administration and addiction.
Assessing and treating both persistent pain and cancer BTP in special populations such as the elderly and pediatric patients understandably presents a number of concerns for physicians. Although preclinical studies show that aging often correlates with increased pain sensitivity,[38], [39], [40] and [41] pain is often underdiagnosed in older patients.42 Age bias often shapes physicians' attitudes toward pain in the elderly. In a survey of 386 physicians to examine the attitudes, knowledge, and psychological factors that contribute to pain management decisions, about 31% believed that older patients were less likely to report pain than patients who were younger. The lack of studies designed to assess the efficacy of pain treatments in elderly patients and the unavailability of meta-analyses and systemic reviews to investigate use of opioids in the elderly are two reasons physicians are often hesitant in treating elderly patients with opioids.38 Another reason for the inadequate treatment of pain in the elderly is concern surrounding drug–drug interactions and uncontrolled side effects resulting from polypharmacy and variability in the patient population.43 The aging process results in impaired kidney and liver function and changes in body composition (including increases in body fat and changes in protein binding), which lead to alterations in drug distribution, metabolism, and elimination. Aging affects receptor responses and substrate intake in older individuals, and thus, the biological processes of increasing age influence the pharmacodynamics of drugs. Most elderly patients experience multiple comorbidities that require medications in addition to treatments for persistent pain and cancer BTP. It is not always possible to predict the drug–drug interactions and side effects that will often result from concomitant medications in this patient population.38 Finally, cognitive impairment and the inability to effectively communicate by the elderly make it difficult for physicians to properly assess pain in this patient population.44
Cancer BTP in children is often insufficiently treated because it is rarely assessed and poorly investigated. Additional research in this area would be useful in determining the true degree of risk associated with the use of opioids in children with cancer.[45] and [46]
Finally, practitioners working within the constraints of stringent laws and guidelines surrounding the allowed use of opioids may be reluctant to prescribe opioids given concerns over repercussions or uncertainty regarding appropriate protocols and documentation for use.[12], [47] and [48] Since cancer BTP episodes are not always properly assessed or recognized as separate clinical entities, opioids may not be prescribed in dosages sufficient to treat flares of persistent pain or more appropriate opioids that would best match the cancer BTP experience may not be considered.30 Risk evaluation and mitigation strategies49 and programs that require extensive documentation (such as writing prescriptions in triplicate29) may prevent health-care providers from prescribing opioids to patients experiencing cancer BTP. Physicians may also be hesitant to prescribe opioids because of the time and effort required for reimbursement by managed health-care companies.50
Patient-Related Factors in Clinical Inertia
Patient perceptions and beliefs about pain medication are similar to those of clinicians in some respects. In a study of patients in not-for-profit community hospitals and outpatients, both groups (27% of inpatients and 37% of outpatients) expressed a fear of addiction to pain medication.51 These patients also expressed the belief that medication should be saved until pain gets worse and that medication may interfere with daily activities.51 Like doctors, patients expected concerns about the side effects of pain medications including drowsiness, constipation, nausea, and difficulty breathing. Unlike doctors, a significant number of these patients also expressed concern about the cost of medication.51
Treatment decisions may be based on a number of factors including the patient's understanding of the diagnosis and confidence in the effectiveness of treatments.52 Patients may believe that pain is part of the cancer diagnosis and is to be expected. Some patients believe that use of opioids signifies that the “end of life” is near. They may believe that side effects related to opioids are unavoidable and the burden of the use of opioids outweighs the benefits.53
Another factor to consider when examining patient barriers to effective pain medication is the inability to effectively communicate pain to health-care professionals. The health-care setting may play a role in effectively describing pain. Patients in hospitals have the benefit of health-care providers being available to ask questions about their pain experience, which aids in describing the pain experience. Outpatients, on the other hand, may have difficulty in explaining their level of pain to family members and health-care providers.51 Patients may also be reluctant to describe their pain to family members or health-care professionals for fear of causing distress or appearing to be “weak”.54
Summary and Considerations
Many health-care professionals are not aware of the dual components of cancer pain: persistent pain and cancer BTP. This lack of understanding results in underdiagnosis and undertreatment of persistent cancer pain and cancer BTP, which can have significant repercussions on the patient–physician relationship. In a pan-European survey to examine the treatment of cancer pain, many patients reported feeling that clinicians did not prioritize the treatment of cancer pain highly enough; instead, treatment of cancer was given greater emphasis. The consequence of the lack of time committed to pain assessment and discussions geared toward the treatment options was that patients felt that their quality of life was not important to clinicians, their pain was not appreciated, and the physician did not know how to treat their pain.55
Several steps may be taken to close the gap between patients and physicians in the treatment of cancer BTP. Continuing education of physicians and other health-care professionals about the importance of persistent cancer pain and cancer BTP is essential to overcoming this obstacle.10 Properly prescribing opioids as treatments for cancer BTP and adherence to analgesic guidelines can prevent the undertreatment of this condition.56 Proper documentation of doses previously prescribed, side effects observed after dosing, and the severity of pain experienced may decrease the risk of adverse events.57
Improved pain management by practitioners can be achieved through knowledge and application of a number of validated pain assessment tools (Table 2). The patient self-report (in the form of a pain diary) is one of the most reliable methods of assessing persistent pain and cancer BTP; however, physicians must be careful to ensure accurate documentation of pain flares in patients who experience mental impairment or do not understand the concept of cancer BTP.10 Also, physical examination and appropriate tests are required to determine the pathophysiology of pain.12 In addition to the patient diary and the physical examination, there are a number of tools available to assess persistent pain and cancer BTP (Table 2).[58], [59] and [60]
| ASSESSMENT | |
|---|---|
| Unidimensional scales | Numerical Rating Scale (NRS) |
| Visual Analogue Scale (VAS) | |
| Verbal Rating Scale | |
| Wong Baker FACES Pain Rating Scale | |
| Colored Visual Analogue Scale | |
| Coping Strategies Questionnaire | |
| Multidimensional scales | Brief Pain Inventory |
| McGill Pain Questionnaire |
Pain assessments may be unidimensional or multidimensional. Unidimensional assessments such as the Visual Analogue Scale and the Numerical Rating Scale60 offer simple measurements for changes in pain intensity,61 are easy to use, and require minimal health-care provider involvement. Multidimensional pain assessment instruments like the Brief Pain Inventory and the McGill Pain Questionnaire, on the other hand, consider not only pain intensity but also dimensions such as changes or fluctuations in pain, treatments, location of pain, physical descriptions of pain sensations, emotions and feelings related to pain, duration of pain, and history of pain.61
An additional resource to utilize would be the pain knowledge of nurses. Performance-based testing to examine the deficiencies in assessment and management of cancer show that hospice-care nurses were more proficient in assessing pain intensity and pain location than resident and family physicians.[62], [63] and [64] Nurses also have the opportunity to spend more time with the patient and to assess response to treatment plans.56
Conclusions
Cancer BTP is underdiagnosed and undertreated due to a number of patient and health-care provider–related factors. By identifying misconceptions; providing education to health-care providers, patients, and family caregivers; and accessing all available resources to improve pain diagnosis and management, steps can be taken to ensure that patients experiencing persistent cancer pain and cancer BTP are properly treated.
References1
1 A. Caraceni, C. Martini and E. Zecca et al., Breakthrough pain characteristics and syndromes in patients with cancer pain: An international survey, Palliat Med 18 (2004), pp. 177–183. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (93)
2 R.K. Portenoy, D. Payne and P. Jacobsen, Breakthrough pain: characteristics and impact in patients with cancer pain, Pain 81 (1999), pp. 129–134. Article |

3 T. Gutgsell, D. Walsh, D.S. Zhukovsky, F. Gonzales and R. Lagman, A prospective study of the pathophysiology and clinical characteristics of pain in a palliative medicine population, Am J Hosp Palliat Care 20 (2003), pp. 140–148. View Record in Scopus | Cited By in Scopus (16)
4 S.S. Hwang, V.T. Chang and B. Kasimis, Cancer breakthrough pain characteristics and responses to treatment at a VA medical center, Pain 101 (2003), pp. 55–64. Article |

5 K.B. Svendsen, S. Andersen and S. Arnason et al., Breakthrough pain in malignant and non-malignant diseases: a review of prevalence, characteristics and mechanisms, Eur J Pain 9 (2005), pp. 195–206. Article |

6 M.P. Davis and D. Walsh, Epidemiology of cancer pain and factors influencing poor pain control, Am J Hosp Palliat Care 21 (2004), pp. 137–142. View Record in Scopus | Cited By in Scopus (32)
7 D.A. Fishbain, Pharmacotherapeutic management of breakthrough pain in patients with chronic persistent pain, Am J Manag Care 14 (suppl 1) (2008), pp. S123–S128. View Record in Scopus | Cited By in Scopus (5)
8 R.K. Portenoy and N.A. Hagen, Breakthrough pain: definition, prevalence and characteristics, Pain 41 (1990), pp. 273–281. Abstract |

9 Shoemaker S, Bruns D, Portenoy RK. Characteristics and impact of breakthrough pain (BTP) in noncancer and cancer-related chronic pain managed by clinicians who are not pain specialists. Presented at the American Academy of Pain Medicine 25th Annual Meeting, Honolulu, HI, January 28–31, 2009.
10 R. Payne, Recognition and diagnosis of breakthrough pain, Pain Med 8 (suppl 1) (2007), pp. S3–S7. View Record in Scopus | Cited By in Scopus (25)
11 American Pain Foundation, Breakthrough Cancer Pain Survey Fact Sheet http://www.painfoundation.org/learn/programs/spotlight-on-cancer-pain/breakthrough-pain/btcp-fact-sheet.pdf 2009.
12 National Cancer Institute, Pain (PDQ®) Health Professional Version 2010 http://www.cancer.gov/cancertopics/pdq/supportivecare/pain/HealthProfessional Accessed January 29, 2010.
13 J. Abrahm, A Physician's Guide to Pain and Symptom Management in Cancer Patients (2nd ed.), Johns Hopkins University Press, Baltimore (2005).
14 S.M. Colleau, The significance of breakthrough pain in cancer, Cancer Pain Release (1999), p. 12.
15 World Health Organization, Cancer Pain Relief (2nd ed.), World Health Organization, Geneva (1996).
16 S.J. Farr and B.A. Otulana, Pulmonary delivery of opioids as pain therapeutics, Adv Drug Deliv Rev 58 (2006), pp. 1076–1088. Article |

17 C.M. Terpening and W.M. Johnson, Methadone as an analgesic: a review of the risks and benefits, W V Med J 103 (2007), pp. 14–18. View Record in Scopus | Cited By in Scopus (10)
18 G. Zeppetella, Opioids for cancer breakthrough pain: a pilot study reporting patient assessment of time to meaningful pain relief, J Pain Symptom Manage 35 (2008), pp. 563–567. Article |

19 National Cancer Institute, Pain (PDR®) Health Professional Version 2009 http://www.cancer.gov/cancertopics/pdq/supportivecare/pain/HealthProfessional Accessed December 1, 2009.
20 National Comprehensive Cancer Network (NCCN), National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology, Adult Cancer Pain http://www.nccn.org/professionals/physician_gls/f_guidelines.asp Accessed May 10, 2010.
21 , Actiq (oral transmucosal fentanyl citrate lozenge) prescribing information Cepahlon, Inc.: Salt Lake City, UT http://www.actiq.com/pdf/actiq_package_insert_4_5_07.pdf Accessed May 21, 2010.
22 , Fentora (fentanyl buccal tablet) prescribing information Cephalon, Inc.: Frazer, PA http://www.fentora.com/pdfs/pdf100_prescribing_info.pdf Accessed May 21, 2010.
23 , Onsolis (fentanyl buccal soluble film) prescribing information Meda Pharmaceuticals, Inc.: Somerset, NJ http://onsolisfocus.com/pdf/pi.pdf Accessed May 21, 2010.
24 F. Brennan, D.B. Carr and M. Cousins, Pain management: a fundamental human right, Anesth Analg 105 (2007), pp. 205–221. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (109)
25 T.E. Elliott and B.A. Elliott, Physician attitudes and beliefs about use of morphine for cancer pain, J Pain Symptom Manage 7 (1992), pp. 141–148. Abstract |

26 B.A. Rich, An ethical analysis of the barriers to effective pain management, Camb Q Healthc Ethics 9 (2000), pp. 54–70. View Record in Scopus | Cited By in Scopus (27)
27 K.L. Sees and H.W. Clark, Opioid use in the treatment of chronic pain: assessment of addiction, J Pain Symptom Manage 8 (1993), pp. 257–264. Article |

28 J.E. Mortimer and N.L. Bartlett, Assessment of knowledge about cancer pain management by physicians in training, J Pain Symptom Manage 14 (1997), pp. 21–28. Abstract |

29 R. Gallagher, P. Hawley and W. Yeomans, A survey of cancer pain management knowledge and attitudes of British Columbian physicians, Pain Res Manag 9 (2004), pp. 188–194. View Record in Scopus | Cited By in Scopus (17)
30 B.G. Jenkins, P.H. Tuffin, C.L. Choo and S.A. Schug, Opioid prescribing: an assessment using quality statements, J Clin Pharm Ther 30 (2005), pp. 597–602. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (7)
31 C.M. Reid, R. Gooberman-Hill and G.W. Hanks, Opioid analgesics for cancer pain: symptom control for the living or comfort for the dying?: A qualitative study to investigate the factors influencing the decision to accept morphine for pain caused by cancer, Ann Oncol 19 (2008), pp. 44–48. View Record in Scopus | Cited By in Scopus (18)
32 R. Jacobsen, P. Sjogren, C. Moldrup and L. Christrup, Physician-related barriers to cancer pain management with opioid analgesics: a systematic review, J Opioid Manag 3 (2007), pp. 207–214. View Record in Scopus | Cited By in Scopus (18)
33 R. Benyamin, A.M. Trescot and S. Datta et al., Opioid complications and side effects, Pain Physician 11 (2008), pp. S105–S120. View Record in Scopus | Cited By in Scopus (86)
34 K. Bell and A. Salmon, Pain, physical dependence and pseudoaddiction: redefining addiction for “nice” people?, Int J Drug Policy 20 (2009), pp. 170–178. Article |

35 E.O. Dumas and G.M. Pollack, Opioid tolerance development: a pharmacokinetic/pharmacodynamic perspective, AAPS J 10 (2008), pp. 537–551. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (5)
36 American Pain Society, Definitions Related to the Use of Opioids for the Treatment of Pain http://www.ampainsoc.org/advocacy/opioids2.htm Accessed March 23, 2010.
37 J. Hojsted and P. Sjogren, Addiction to opioids in chronic pain patients: a literature review, Eur J Pain 11 (2007), pp. 490–518. Article |

38 O.H. Wilder-Smith, Opioid use in the elderly, Eur J Pain 9 (2005), pp. 137–140. Article |

39 D. Jourdan, S. Boghossian and A. Alloui et al., Age-related changes in nociception and effect of morphine in the Lou rat, Eur J Pain 4 (2000), pp. 291–300. Abstract |

40 E. Kramer and R.J. Bodnar, Age-related decrements in morphine analgesia: a parametric analysis, Neurobiol Aging 7 (1986), pp. 185–191. Abstract |

41 M.A. Smith and J.D. Gray, Age-related differences in sensitivity to the antinociceptive effects of opioids in male rats: Influence of nociceptive intensity and intrinsic efficacy at the mu receptor, Psychopharmacology (Berl) 156 (2001), pp. 445–453. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (26)
42 F.M. Gloth 3rd, Pain management in older adults: prevention and treatment, J Am Geriatr Soc 49 (2001), pp. 188–199. Full Text via CrossRef
43 S.M. Weinstein, L.F. Laux and J.I. Thornby et al., Physicians' attitudes toward pain and the use of opioid analgesics: results of a survey from the Texas Cancer Pain Initiative, South Med J 93 (2000), pp. 479–487. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (94)
44 B. Collett, S. O'Mahoney, P. Schofield, S.J. Closs and J. Potter, The assessment of pain in older people, Clin Med 7 (2007), pp. 496–500. View Record in Scopus | Cited By in Scopus (2)
45 R.A. Morrison, Update on sickle cell disease: incidence of addiction and choice of opioid in pain management, Pediatr Nurs 17 (1991), p. 503. View Record in Scopus | Cited By in Scopus (6)
46 C.H. Pegelow, Survey of pain management therapy provided for children with sickle cell disease, Clin Pediatr (Phila) 31 (1992), pp. 211–214. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (46)
47 C. Wright, S. Schnoll and D. Bernstein, Risk evaluation and mitigation strategies for drugs with abuse liability: public interest, special interest, conflicts of interest, and the industry perspective, Ann N Y Acad Sci 1141 (2008), pp. 284–303. View Record in Scopus | Cited By in Scopus (0)
48 M. Zenz, T. Zenz, M. Tryba and M. Strumpf, Severe undertreatment of cancer pain: a 3-year survey of the German situation, J Pain Symptom Manage 10 (1995), pp. 187–191. Abstract |

49 C. Wright, S. Schnoll and D. Bernstein, Risk evaluation and mitigation strategies for drug with abuse liability: public interest, special interest, conflicts of interest and industry perspective, Ann N Y Acad Sci 1141 (2008), pp. 284–303. View Record in Scopus | Cited By in Scopus (6)
50 D. Nemecek, Improving access to buprenorphine: A managed care company overcomes several obstacles to make this treatment more available, Behav Healthc 27 (2007), pp. 40–42. View Record in Scopus | Cited By in Scopus (1)
51 C.C. Corizzo, M.C. Baker and G.C. Henkelmann, Assessment of patient satisfaction with pain management in small community inpatient and outpatient settings, Oncol Nurs Forum 27 (2000), pp. 1279–1286. View Record in Scopus | Cited By in Scopus (10)
52 R. Sapir, R. Catane and B. Kaufman et al., Cancer patient expectations of and communication with oncologists and oncology nurses: the experience of an integrated oncology and palliative care service, Support Care Cancer 8 (2000), pp. 458–463. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (37)
53 K. Forbes, Opioids: beliefs and myths, J Pain Palliat Care Pharmacother 20 (2006), pp. 33–35. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (8)
54 P.E. Berry and S.E. Ward, Barriers to pain management in hospice: a study of family caregivers, Hosp J 10 (1995), pp. 19–33. View Record in Scopus | Cited By in Scopus (45)
55 H. Breivik, N. Cherny and B. Collett et al., Cancer-related pain: a pan-European survey of prevalence, treatment, and patient attitudes, Ann Oncol 20 (2009), pp. 1420–1433. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (36)
56 S.H. Neo, E.C. Loh and W.H. Koo, An audit of morphine prescribing in a hospice, Singapore Med J 42 (2001), pp. 417–419. View Record in Scopus | Cited By in Scopus (8)
57 M. Weber and C. Huber, Documentation of severe pain, opioid doses, and opioid-related side effects in outpatients with cancer: a retrospective study, J Pain Symptom Manage 17 (1999), pp. 49–54. Article |

58 F. De Conno, A. Caraceni and A. Gamba et al., Pain measurement in cancer patients: a comparison of six methods, Pain 57 (1994), pp. 161–166. Abstract |

59 G.D. Hakonsen, S. Hudson and T. Loennechen, Design and validation of a medication assessment tool for cancer pain management, Pharm World Sci 28 (2006), pp. 342–351. View Record in Scopus | Cited By in Scopus (5)
60 A. Caraceni, N. Cherny and R. Fainsinger et al., Pain measurement tools and methods in clinical research in palliative care: recommendations of an Expert Working Group of the European Association of Palliative Care, J Pain Symptom Manage 23 (2002), pp. 239–255. Article |

61 J.C. Holen, M.J. Hjermstad and J.H. Loge et al., Pain assessment tools: is the content appropriate for use in palliative care?, J Pain Symptom Manage 32 (2006), pp. 567–580. Article |
62 P.A. Sloan, M.B. Donnelly, B. Vanderveer, M. Delomas, R.W. Schwartz and D.A. Sloan, Cancer pain education among family physicians, J Pain Symptom Manage 14 (1997), pp. 74–81. Abstract |
63 P.A. Sloan, B.L. Vanderveer, J.S. Snapp, M. Johnson and D.A. Sloan, Cancer pain assessment and management recommendations by hospice nurses: University of Kentucky, Lexington, Kentucky, J Pain Symptom Manage 18 (1999), pp. 103–110. Article |
64 C.T. Furstenberg, T.A. Ahles and M.B. Whedon et al., Knowledge and attitudes of health-care providers toward cancer pain management: a comparison of physicians, nurses, and pharmacists in the state of New Hampshire, J Pain Symptom Manage 15 (1998), pp. 335–349. Article |

Vitae
Dr. Rhiner is from Pallgesia Associates, Alta Loma, California.
Dr. von Gunten is Provost, Institute for Palliative Medicine at San Diego Hospice, San Diego, California.
Review
Michelle I. Rhiner RN, MSN, ACHPN![]()
![]()
| Referred to by: | The Challenges of Treating Patients with Cancer Pain The Journal of Supportive Oncology, Volume 8, Issue 6, November-December 2010, Pages 239-240, Sloan Beth Karver, Jessalyn H. Berger | |
| Referred to by: | A Conceptual Solution to Improve the Management of Cancer-Related Breakthrough Pain The Journal of Supportive Oncology, Volume 8, Issue 6, November-December 2010, Page 241, Wendy Ledesma, Toby C. Campbell | |
Abstract
Cancer breakthrough pain is a flare in pain that “breaks through” well-controlled persistent cancer pain. Although the condition is highly prevalent, the concept of cancer breakthrough pain is not well understood and is therefore underdiagnosed and undertreated. The purpose of this review is to examine the roles the health-care practitioner and patient/family caregiver play in the undertreatment of breakthrough pain. A lack of technical knowledge about pain management and pain assessment, attitudes about opioid addiction, and regulatory guidelines influence the manner in which opioids are prescribed. Patients harbor a variety of fears and misconceptions, such as opioid addiction, tolerance, side effects, and the meaning of pain, which can create a barrier to effective communication with their health-care provider regarding their cancer pain management and specifically their breakthrough pain. Identifying these issues gives health-care professionals and patients an opportunity to develop strategies that can improve the treatment of cancer breakthrough pain.
Article Outline
Adapted from Payne R.10
| CATEGORY OF PAIN | CHARACTERISTICS |
|---|---|
| Neuropathic | Caused by structural changes in the central nervous system and the peripheral nervous system; described as tingling, burning, or shooting in nature |
| Visceral | Deep cramping and tearing pain that may originate in internal organs |
| Somatic | Requires skeletal involvement; described as constant throbbing and aching that increases with movement |
A significant number of patients suffering from cancer experience these pain flares. In a single-population evaluation of cancer pain (n = 159), it was reported that of those patients with continuous pain, 57 (75%) experienced cancer BTP. More than half (54%) of those patients who experienced cancer pain reported it being related to particular activities, while a little over one-quarter of patients (26%) experienced pain idiopathic in nature and 16% of patients experienced end-of-dose pain.3 In a survey of 545 patients with cancer experiencing fluctuations in pain conducted by the American Pain Foundation, 96% of these patients experienced episodes of cancer BTP at least once a month, more than 70% experienced cancer BTP episodes at least once a week, and more than one-fifth (22%) experienced cancer BTP more than once a day.11
The inadequate treatment of cancer pain is an issue with far-reaching implications; therefore, the purpose of this review is to examine the roles that health-care providers, patients, and family caregivers play in the undertreatment of cancer BTP.
Defining Cancer BTP
Despite the reported prevalence of the condition, questions continue to surround the definition of cancer BTP. The earliest clinical accounts of cancer BTP describe it as a greater than moderate temporary flare in pain that occurs on a baseline of moderate pain in patients receiving opioids for cancer pain.8 Presently, descriptions of cancer BTP include statements that flares may occur in the presence of stable persistent pain (regardless of the treatment)12 having pain levels ranging from moderate to severe, with onset depending on the subtype of pain.13
Some controversy centers on the term “breakthrough pain.” The American-English term “breakthrough pain” does not have an exact translation to other languages. Physicians in Europe may use the term “episodic” or “transient” to describe these fluctuations in cancer pain. However, these terms do not capture the idea that the pain is above and beyond persistent pain control (eg, “breaks through baseline analgesia”). Furthermore, if the term “breakthrough pain” is used, it may be limited to descriptions of pain that occur at the end of the dosing cycle.14 Finally, there are physicians in some countries who do not view exacerbation of cancer pain as a separate clinical entity. Instead, they view spikes of pain as a predictable or normal element of cancer pain.14
In spite of the questions surrounding cancer BTP, persistent pain must be controlled before management of cancer BTP can proceed. In addition, repeated episodes of cancer BTP may indicate that baseline pain has not been properly assessed and not adequately managed.10 Therefore, cancer BTP and persistent cancer pain should be assessed independently as they are separate clinical conditions.
Treatment Approaches to Persistent Cancer Pain versus Cancer BTP
Persistent cancer pain requires around-the-clock (ATC) treatment with therapeutic agents that both maximize outcomes and minimize risks. ATC treatment allows maintenance of drug concentrations and prevents peaks and troughs resulting in increased risk of toxicity and lack of efficacy. Opioids such as morphine, oxycodone, methadone, and fentanyl are used extensively for the treatment of cancer pain as they produce an analgesic effect at a minimum dose and are easily titrated.15 While opioids are administrated orally per World Health Organization recommendations,15 they may also be administered rectally, intravenously, subcutaneously, intramuscularly, and transdermally. Clinical trials are being conducted to examine the administration of opioids through inhalation.16 Methadone is an example of a long-acting opioid that has been used to treat persistent pain.17 Pharmaceutically long-acting drugs such as sustained-release formulations of morphine, oxycodone, oxymorphone, and hydromorphone have been used to treat cancer pain.18 Adjuvant medications may be coadministered with opioids to treat symptoms that occur concurrently with cancer pain and to augment the analgesic effect. Although few clinical trials have evaluated the efficacy of adjuvants in patients with cancer, local anesthetics (eg, lidocaine), nonsteroidal anti-inflammatory drugs (eg, aspirin, naproxen, and ibuprofen), nonopioid analgesics (eg, acetaminophen), antidepressants (eg, amitriptyline, duloxetine, and venlafaxine), and anticonvulsants (eg, gabapentin, pregabalin, valproate, and lamotrigine) have been used to supplement opioid therapy.12
A number of physical and cognitive–behavioral interventions may be used in addition to pharmacological treatments to alleviate some of the pain symptoms experienced in patients with persistent cancer pain. In most patients, heating pads or ice packs may be used to relieve pain and reduce swelling; however, neither heat nor cold should be used on irradiated tissue, and caution should be used when using ice packs on patients with peripheral vascular disease.19 Exercise may be started or continued to improve physical conditioning. While tumor masses should not be manipulated, other techniques requiring physical stimulation, such as massage, pressure, and vibration, may also be used in the treatment of persistent pain. Either moving an immobile patient or temporarily restricting movement can prevent or alleviate pain. Acupuncture is another treatment that may be used to treat persistent cancer pain. Psychosocial interventions include hypnosis, use of relaxation techniques, biofeedback, and cognitive distraction.19
Orally administered, short-acting or rapid-acting opioids are used to treat cancer BTP episodes. However, the pharmacokinetic profiles of oral opioids may not match the onset and duration of some cancer BTP episodes.20 An analgesic agent used to treat cancer BTP should match the temporal characteristics of cancer BTP, be easily titrated to higher or lower doses if needed, and, if used appropriately in opioid-tolerant patients, not be associated with undue adverse effects.
Oral transmucosal delivery of opioids is an option for these challenges. Three formulations of the opioid fentanyl are available for transmucosal delivery: oral transmucosal fentanyl citrate (OTFC),21 fentanyl buccal tablets (FBTs),22 and fentanyl buccal soluble film (FBSF).23 OTFC is a fentanyl lozenge that has demonstrated an analgesic effect within 15 minutes of administration. Despite its rapid onset of action, the amount of fentanyl administered with the OTFC lozenge depends on the education provided to the patient on the use of this medication and the ability of the patient to actively use this product.21 While FBT (a tablet that utilizes an effervescent reaction to improve absorption) does not require substantial patient participation, its use has been associated with application-site side effects.22 Like OTFC and FBT, FBSF offers a rapid onset of action but does not require active patient participation and has minimal oral adverse side effects.23 These oral transmucosal fentanyl products should be administered only to opioid-tolerant patients, to avoid the risk of life-threatening respiratory depression.
Physician-Related Factors in Clinical Inertia
Three elements shape physicians' perceptions of the severity of cancer BTP and influence prescribing practices: technical knowledge, attitudes concerning use of opioids, and regulatory restrictions.[24], [25], [26] and [27]
The medical training of most physicians may not include courses in pain management. Instead, practitioners' education is a result of inpatient experience during the postgraduate years and is generally geared toward management of acute injuries, postoperative pain, and cardiovascular events such as myocardial infarctions.28 More evidence of this lack of knowledge among physicians is presented in a survey of British Columbian physicians.29 Among those doctors who responded, only 32% were aware of the dose that would produce an equal analgesic effect when a switch between morphine and acetaminophen is required. Furthermore, only 55% of physicians correctly stated that doses of opioids for cancer BTP should be 10% of the total daily dose and administered every 1 or 2 hours as needed per National Comprehensive Cancer Network guidelines. These findings suggest that when patients need stronger analgesics, physicians may not be skilled in converting to a more potent opioid or calculating a dose for cancer BTP management.29 Indiscriminate polypharmacy presents additional complications, whereby coadministration of multiple opioids exposes patients to an increased risk of toxicity and distorts the clinician's interpretation of outcomes.30
In addition to training, the communication skills of the practitioner play a role in effective treatment of cancer BTP and persistent cancer pain. Discussions regarding treatment can significantly influence a patient's decision to use opioids. In an interview study conducted during a cancer pain management trial, patients were reportedly occasionally “suspicious about the idea of choice” in the use of opioids.31 These patients favored thorough discussions of pain treatment options with physicians who were knowledgeable and confident about the use of opioids. The likelihood of patients participating in a pain management trial also increased if the physician stated that a lower dose of opioid would be used initially and treatment would cease if side effects developed.
Health-care providers' attitudes about the side effects of opioid treatment also impact treatment of persistent cancer pain and cancer BTP.32 In addition, although clinical studies do not support the assertion that patients may become addicted to opioids used to treat acute and persistent cancer pain, the belief that these patients are at risk for addiction prevents some health-care providers from prescribing opioids.33 The perceived relationship between opioid tolerance and addiction is one of the sources of confusion. One of the complications of opioid therapy is pharmacological tolerance, the loss of analgesic effect. Tolerance to an opioid may be determined genetically or acquired after metabolic changes, changes to receptors, or by learned behavior.33 The result of tolerance is decreasing pain relief, in spite of persistent doses over time.[34] and [35] Pharmacological tolerance is associated with incomplete cross-tolerance to other opioids. In contrast, addiction is a psychological syndrome characterized by uncontrolled and persistent use of the drug despite harm.[34], [35] and [36] Physical dependence on opioids is characterized by a withdrawal syndrome that occurs when the dose is stopped or decreased suddenly or an antagonist is administered.36 Pharmacological dependence is not synonymous with addiction; pharmacological tolerance occurs with many commonly prescribed drugs (such as nitrates) and does not indicate addiction.34
The prevalence of opioid addiction among a population of patients with cancer pain varies. In a meta-review of opioid addiction studies, authors reported a 0%–7.7% prevalence rate of addiction in patients with cancer depending on the population studied and the diagnostic measure used.37 This corresponds with the rate of addiction prevalent in the population at large—it does not support a cause-and-effect relationship between opioid administration and addiction.
Assessing and treating both persistent pain and cancer BTP in special populations such as the elderly and pediatric patients understandably presents a number of concerns for physicians. Although preclinical studies show that aging often correlates with increased pain sensitivity,[38], [39], [40] and [41] pain is often underdiagnosed in older patients.42 Age bias often shapes physicians' attitudes toward pain in the elderly. In a survey of 386 physicians to examine the attitudes, knowledge, and psychological factors that contribute to pain management decisions, about 31% believed that older patients were less likely to report pain than patients who were younger. The lack of studies designed to assess the efficacy of pain treatments in elderly patients and the unavailability of meta-analyses and systemic reviews to investigate use of opioids in the elderly are two reasons physicians are often hesitant in treating elderly patients with opioids.38 Another reason for the inadequate treatment of pain in the elderly is concern surrounding drug–drug interactions and uncontrolled side effects resulting from polypharmacy and variability in the patient population.43 The aging process results in impaired kidney and liver function and changes in body composition (including increases in body fat and changes in protein binding), which lead to alterations in drug distribution, metabolism, and elimination. Aging affects receptor responses and substrate intake in older individuals, and thus, the biological processes of increasing age influence the pharmacodynamics of drugs. Most elderly patients experience multiple comorbidities that require medications in addition to treatments for persistent pain and cancer BTP. It is not always possible to predict the drug–drug interactions and side effects that will often result from concomitant medications in this patient population.38 Finally, cognitive impairment and the inability to effectively communicate by the elderly make it difficult for physicians to properly assess pain in this patient population.44
Cancer BTP in children is often insufficiently treated because it is rarely assessed and poorly investigated. Additional research in this area would be useful in determining the true degree of risk associated with the use of opioids in children with cancer.[45] and [46]
Finally, practitioners working within the constraints of stringent laws and guidelines surrounding the allowed use of opioids may be reluctant to prescribe opioids given concerns over repercussions or uncertainty regarding appropriate protocols and documentation for use.[12], [47] and [48] Since cancer BTP episodes are not always properly assessed or recognized as separate clinical entities, opioids may not be prescribed in dosages sufficient to treat flares of persistent pain or more appropriate opioids that would best match the cancer BTP experience may not be considered.30 Risk evaluation and mitigation strategies49 and programs that require extensive documentation (such as writing prescriptions in triplicate29) may prevent health-care providers from prescribing opioids to patients experiencing cancer BTP. Physicians may also be hesitant to prescribe opioids because of the time and effort required for reimbursement by managed health-care companies.50
Patient-Related Factors in Clinical Inertia
Patient perceptions and beliefs about pain medication are similar to those of clinicians in some respects. In a study of patients in not-for-profit community hospitals and outpatients, both groups (27% of inpatients and 37% of outpatients) expressed a fear of addiction to pain medication.51 These patients also expressed the belief that medication should be saved until pain gets worse and that medication may interfere with daily activities.51 Like doctors, patients expected concerns about the side effects of pain medications including drowsiness, constipation, nausea, and difficulty breathing. Unlike doctors, a significant number of these patients also expressed concern about the cost of medication.51
Treatment decisions may be based on a number of factors including the patient's understanding of the diagnosis and confidence in the effectiveness of treatments.52 Patients may believe that pain is part of the cancer diagnosis and is to be expected. Some patients believe that use of opioids signifies that the “end of life” is near. They may believe that side effects related to opioids are unavoidable and the burden of the use of opioids outweighs the benefits.53
Another factor to consider when examining patient barriers to effective pain medication is the inability to effectively communicate pain to health-care professionals. The health-care setting may play a role in effectively describing pain. Patients in hospitals have the benefit of health-care providers being available to ask questions about their pain experience, which aids in describing the pain experience. Outpatients, on the other hand, may have difficulty in explaining their level of pain to family members and health-care providers.51 Patients may also be reluctant to describe their pain to family members or health-care professionals for fear of causing distress or appearing to be “weak”.54
Summary and Considerations
Many health-care professionals are not aware of the dual components of cancer pain: persistent pain and cancer BTP. This lack of understanding results in underdiagnosis and undertreatment of persistent cancer pain and cancer BTP, which can have significant repercussions on the patient–physician relationship. In a pan-European survey to examine the treatment of cancer pain, many patients reported feeling that clinicians did not prioritize the treatment of cancer pain highly enough; instead, treatment of cancer was given greater emphasis. The consequence of the lack of time committed to pain assessment and discussions geared toward the treatment options was that patients felt that their quality of life was not important to clinicians, their pain was not appreciated, and the physician did not know how to treat their pain.55
Several steps may be taken to close the gap between patients and physicians in the treatment of cancer BTP. Continuing education of physicians and other health-care professionals about the importance of persistent cancer pain and cancer BTP is essential to overcoming this obstacle.10 Properly prescribing opioids as treatments for cancer BTP and adherence to analgesic guidelines can prevent the undertreatment of this condition.56 Proper documentation of doses previously prescribed, side effects observed after dosing, and the severity of pain experienced may decrease the risk of adverse events.57
Improved pain management by practitioners can be achieved through knowledge and application of a number of validated pain assessment tools (Table 2). The patient self-report (in the form of a pain diary) is one of the most reliable methods of assessing persistent pain and cancer BTP; however, physicians must be careful to ensure accurate documentation of pain flares in patients who experience mental impairment or do not understand the concept of cancer BTP.10 Also, physical examination and appropriate tests are required to determine the pathophysiology of pain.12 In addition to the patient diary and the physical examination, there are a number of tools available to assess persistent pain and cancer BTP (Table 2).[58], [59] and [60]
| ASSESSMENT | |
|---|---|
| Unidimensional scales | Numerical Rating Scale (NRS) |
| Visual Analogue Scale (VAS) | |
| Verbal Rating Scale | |
| Wong Baker FACES Pain Rating Scale | |
| Colored Visual Analogue Scale | |
| Coping Strategies Questionnaire | |
| Multidimensional scales | Brief Pain Inventory |
| McGill Pain Questionnaire |
Pain assessments may be unidimensional or multidimensional. Unidimensional assessments such as the Visual Analogue Scale and the Numerical Rating Scale60 offer simple measurements for changes in pain intensity,61 are easy to use, and require minimal health-care provider involvement. Multidimensional pain assessment instruments like the Brief Pain Inventory and the McGill Pain Questionnaire, on the other hand, consider not only pain intensity but also dimensions such as changes or fluctuations in pain, treatments, location of pain, physical descriptions of pain sensations, emotions and feelings related to pain, duration of pain, and history of pain.61
An additional resource to utilize would be the pain knowledge of nurses. Performance-based testing to examine the deficiencies in assessment and management of cancer show that hospice-care nurses were more proficient in assessing pain intensity and pain location than resident and family physicians.[62], [63] and [64] Nurses also have the opportunity to spend more time with the patient and to assess response to treatment plans.56
Conclusions
Cancer BTP is underdiagnosed and undertreated due to a number of patient and health-care provider–related factors. By identifying misconceptions; providing education to health-care providers, patients, and family caregivers; and accessing all available resources to improve pain diagnosis and management, steps can be taken to ensure that patients experiencing persistent cancer pain and cancer BTP are properly treated.
References1
1 A. Caraceni, C. Martini and E. Zecca et al., Breakthrough pain characteristics and syndromes in patients with cancer pain: An international survey, Palliat Med 18 (2004), pp. 177–183. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (93)
2 R.K. Portenoy, D. Payne and P. Jacobsen, Breakthrough pain: characteristics and impact in patients with cancer pain, Pain 81 (1999), pp. 129–134. Article |
3 T. Gutgsell, D. Walsh, D.S. Zhukovsky, F. Gonzales and R. Lagman, A prospective study of the pathophysiology and clinical characteristics of pain in a palliative medicine population, Am J Hosp Palliat Care 20 (2003), pp. 140–148. View Record in Scopus | Cited By in Scopus (16)
4 S.S. Hwang, V.T. Chang and B. Kasimis, Cancer breakthrough pain characteristics and responses to treatment at a VA medical center, Pain 101 (2003), pp. 55–64. Article |
5 K.B. Svendsen, S. Andersen and S. Arnason et al., Breakthrough pain in malignant and non-malignant diseases: a review of prevalence, characteristics and mechanisms, Eur J Pain 9 (2005), pp. 195–206. Article |
6 M.P. Davis and D. Walsh, Epidemiology of cancer pain and factors influencing poor pain control, Am J Hosp Palliat Care 21 (2004), pp. 137–142. View Record in Scopus | Cited By in Scopus (32)
7 D.A. Fishbain, Pharmacotherapeutic management of breakthrough pain in patients with chronic persistent pain, Am J Manag Care 14 (suppl 1) (2008), pp. S123–S128. View Record in Scopus | Cited By in Scopus (5)
8 R.K. Portenoy and N.A. Hagen, Breakthrough pain: definition, prevalence and characteristics, Pain 41 (1990), pp. 273–281. Abstract |
9 Shoemaker S, Bruns D, Portenoy RK. Characteristics and impact of breakthrough pain (BTP) in noncancer and cancer-related chronic pain managed by clinicians who are not pain specialists. Presented at the American Academy of Pain Medicine 25th Annual Meeting, Honolulu, HI, January 28–31, 2009.
10 R. Payne, Recognition and diagnosis of breakthrough pain, Pain Med 8 (suppl 1) (2007), pp. S3–S7. View Record in Scopus | Cited By in Scopus (25)
11 American Pain Foundation, Breakthrough Cancer Pain Survey Fact Sheet http://www.painfoundation.org/learn/programs/spotlight-on-cancer-pain/breakthrough-pain/btcp-fact-sheet.pdf 2009.
12 National Cancer Institute, Pain (PDQ®) Health Professional Version 2010 http://www.cancer.gov/cancertopics/pdq/supportivecare/pain/HealthProfessional Accessed January 29, 2010.
13 J. Abrahm, A Physician's Guide to Pain and Symptom Management in Cancer Patients (2nd ed.), Johns Hopkins University Press, Baltimore (2005).
14 S.M. Colleau, The significance of breakthrough pain in cancer, Cancer Pain Release (1999), p. 12.
15 World Health Organization, Cancer Pain Relief (2nd ed.), World Health Organization, Geneva (1996).
16 S.J. Farr and B.A. Otulana, Pulmonary delivery of opioids as pain therapeutics, Adv Drug Deliv Rev 58 (2006), pp. 1076–1088. Article |
17 C.M. Terpening and W.M. Johnson, Methadone as an analgesic: a review of the risks and benefits, W V Med J 103 (2007), pp. 14–18. View Record in Scopus | Cited By in Scopus (10)
18 G. Zeppetella, Opioids for cancer breakthrough pain: a pilot study reporting patient assessment of time to meaningful pain relief, J Pain Symptom Manage 35 (2008), pp. 563–567. Article |
19 National Cancer Institute, Pain (PDR®) Health Professional Version 2009 http://www.cancer.gov/cancertopics/pdq/supportivecare/pain/HealthProfessional Accessed December 1, 2009.
20 National Comprehensive Cancer Network (NCCN), National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology, Adult Cancer Pain http://www.nccn.org/professionals/physician_gls/f_guidelines.asp Accessed May 10, 2010.
21 , Actiq (oral transmucosal fentanyl citrate lozenge) prescribing information Cepahlon, Inc.: Salt Lake City, UT http://www.actiq.com/pdf/actiq_package_insert_4_5_07.pdf Accessed May 21, 2010.
22 , Fentora (fentanyl buccal tablet) prescribing information Cephalon, Inc.: Frazer, PA http://www.fentora.com/pdfs/pdf100_prescribing_info.pdf Accessed May 21, 2010.
23 , Onsolis (fentanyl buccal soluble film) prescribing information Meda Pharmaceuticals, Inc.: Somerset, NJ http://onsolisfocus.com/pdf/pi.pdf Accessed May 21, 2010.
24 F. Brennan, D.B. Carr and M. Cousins, Pain management: a fundamental human right, Anesth Analg 105 (2007), pp. 205–221. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (109)
25 T.E. Elliott and B.A. Elliott, Physician attitudes and beliefs about use of morphine for cancer pain, J Pain Symptom Manage 7 (1992), pp. 141–148. Abstract |
26 B.A. Rich, An ethical analysis of the barriers to effective pain management, Camb Q Healthc Ethics 9 (2000), pp. 54–70. View Record in Scopus | Cited By in Scopus (27)
27 K.L. Sees and H.W. Clark, Opioid use in the treatment of chronic pain: assessment of addiction, J Pain Symptom Manage 8 (1993), pp. 257–264. Article |
28 J.E. Mortimer and N.L. Bartlett, Assessment of knowledge about cancer pain management by physicians in training, J Pain Symptom Manage 14 (1997), pp. 21–28. Abstract |
29 R. Gallagher, P. Hawley and W. Yeomans, A survey of cancer pain management knowledge and attitudes of British Columbian physicians, Pain Res Manag 9 (2004), pp. 188–194. View Record in Scopus | Cited By in Scopus (17)
30 B.G. Jenkins, P.H. Tuffin, C.L. Choo and S.A. Schug, Opioid prescribing: an assessment using quality statements, J Clin Pharm Ther 30 (2005), pp. 597–602. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (7)
31 C.M. Reid, R. Gooberman-Hill and G.W. Hanks, Opioid analgesics for cancer pain: symptom control for the living or comfort for the dying?: A qualitative study to investigate the factors influencing the decision to accept morphine for pain caused by cancer, Ann Oncol 19 (2008), pp. 44–48. View Record in Scopus | Cited By in Scopus (18)
32 R. Jacobsen, P. Sjogren, C. Moldrup and L. Christrup, Physician-related barriers to cancer pain management with opioid analgesics: a systematic review, J Opioid Manag 3 (2007), pp. 207–214. View Record in Scopus | Cited By in Scopus (18)
33 R. Benyamin, A.M. Trescot and S. Datta et al., Opioid complications and side effects, Pain Physician 11 (2008), pp. S105–S120. View Record in Scopus | Cited By in Scopus (86)
34 K. Bell and A. Salmon, Pain, physical dependence and pseudoaddiction: redefining addiction for “nice” people?, Int J Drug Policy 20 (2009), pp. 170–178. Article |
35 E.O. Dumas and G.M. Pollack, Opioid tolerance development: a pharmacokinetic/pharmacodynamic perspective, AAPS J 10 (2008), pp. 537–551. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (5)
36 American Pain Society, Definitions Related to the Use of Opioids for the Treatment of Pain http://www.ampainsoc.org/advocacy/opioids2.htm Accessed March 23, 2010.
37 J. Hojsted and P. Sjogren, Addiction to opioids in chronic pain patients: a literature review, Eur J Pain 11 (2007), pp. 490–518. Article |
38 O.H. Wilder-Smith, Opioid use in the elderly, Eur J Pain 9 (2005), pp. 137–140. Article |
39 D. Jourdan, S. Boghossian and A. Alloui et al., Age-related changes in nociception and effect of morphine in the Lou rat, Eur J Pain 4 (2000), pp. 291–300. Abstract |
40 E. Kramer and R.J. Bodnar, Age-related decrements in morphine analgesia: a parametric analysis, Neurobiol Aging 7 (1986), pp. 185–191. Abstract |
41 M.A. Smith and J.D. Gray, Age-related differences in sensitivity to the antinociceptive effects of opioids in male rats: Influence of nociceptive intensity and intrinsic efficacy at the mu receptor, Psychopharmacology (Berl) 156 (2001), pp. 445–453. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (26)
42 F.M. Gloth 3rd, Pain management in older adults: prevention and treatment, J Am Geriatr Soc 49 (2001), pp. 188–199. Full Text via CrossRef
43 S.M. Weinstein, L.F. Laux and J.I. Thornby et al., Physicians' attitudes toward pain and the use of opioid analgesics: results of a survey from the Texas Cancer Pain Initiative, South Med J 93 (2000), pp. 479–487. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (94)
44 B. Collett, S. O'Mahoney, P. Schofield, S.J. Closs and J. Potter, The assessment of pain in older people, Clin Med 7 (2007), pp. 496–500. View Record in Scopus | Cited By in Scopus (2)
45 R.A. Morrison, Update on sickle cell disease: incidence of addiction and choice of opioid in pain management, Pediatr Nurs 17 (1991), p. 503. View Record in Scopus | Cited By in Scopus (6)
46 C.H. Pegelow, Survey of pain management therapy provided for children with sickle cell disease, Clin Pediatr (Phila) 31 (1992), pp. 211–214. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (46)
47 C. Wright, S. Schnoll and D. Bernstein, Risk evaluation and mitigation strategies for drugs with abuse liability: public interest, special interest, conflicts of interest, and the industry perspective, Ann N Y Acad Sci 1141 (2008), pp. 284–303. View Record in Scopus | Cited By in Scopus (0)
48 M. Zenz, T. Zenz, M. Tryba and M. Strumpf, Severe undertreatment of cancer pain: a 3-year survey of the German situation, J Pain Symptom Manage 10 (1995), pp. 187–191. Abstract |
49 C. Wright, S. Schnoll and D. Bernstein, Risk evaluation and mitigation strategies for drug with abuse liability: public interest, special interest, conflicts of interest and industry perspective, Ann N Y Acad Sci 1141 (2008), pp. 284–303. View Record in Scopus | Cited By in Scopus (6)
50 D. Nemecek, Improving access to buprenorphine: A managed care company overcomes several obstacles to make this treatment more available, Behav Healthc 27 (2007), pp. 40–42. View Record in Scopus | Cited By in Scopus (1)
51 C.C. Corizzo, M.C. Baker and G.C. Henkelmann, Assessment of patient satisfaction with pain management in small community inpatient and outpatient settings, Oncol Nurs Forum 27 (2000), pp. 1279–1286. View Record in Scopus | Cited By in Scopus (10)
52 R. Sapir, R. Catane and B. Kaufman et al., Cancer patient expectations of and communication with oncologists and oncology nurses: the experience of an integrated oncology and palliative care service, Support Care Cancer 8 (2000), pp. 458–463. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (37)
53 K. Forbes, Opioids: beliefs and myths, J Pain Palliat Care Pharmacother 20 (2006), pp. 33–35. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (8)
54 P.E. Berry and S.E. Ward, Barriers to pain management in hospice: a study of family caregivers, Hosp J 10 (1995), pp. 19–33. View Record in Scopus | Cited By in Scopus (45)
55 H. Breivik, N. Cherny and B. Collett et al., Cancer-related pain: a pan-European survey of prevalence, treatment, and patient attitudes, Ann Oncol 20 (2009), pp. 1420–1433. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (36)
56 S.H. Neo, E.C. Loh and W.H. Koo, An audit of morphine prescribing in a hospice, Singapore Med J 42 (2001), pp. 417–419. View Record in Scopus | Cited By in Scopus (8)
57 M. Weber and C. Huber, Documentation of severe pain, opioid doses, and opioid-related side effects in outpatients with cancer: a retrospective study, J Pain Symptom Manage 17 (1999), pp. 49–54. Article |
58 F. De Conno, A. Caraceni and A. Gamba et al., Pain measurement in cancer patients: a comparison of six methods, Pain 57 (1994), pp. 161–166. Abstract |
59 G.D. Hakonsen, S. Hudson and T. Loennechen, Design and validation of a medication assessment tool for cancer pain management, Pharm World Sci 28 (2006), pp. 342–351. View Record in Scopus | Cited By in Scopus (5)
60 A. Caraceni, N. Cherny and R. Fainsinger et al., Pain measurement tools and methods in clinical research in palliative care: recommendations of an Expert Working Group of the European Association of Palliative Care, J Pain Symptom Manage 23 (2002), pp. 239–255. Article |
61 J.C. Holen, M.J. Hjermstad and J.H. Loge et al., Pain assessment tools: is the content appropriate for use in palliative care?, J Pain Symptom Manage 32 (2006), pp. 567–580. Article |
62 P.A. Sloan, M.B. Donnelly, B. Vanderveer, M. Delomas, R.W. Schwartz and D.A. Sloan, Cancer pain education among family physicians, J Pain Symptom Manage 14 (1997), pp. 74–81. Abstract |
63 P.A. Sloan, B.L. Vanderveer, J.S. Snapp, M. Johnson and D.A. Sloan, Cancer pain assessment and management recommendations by hospice nurses: University of Kentucky, Lexington, Kentucky, J Pain Symptom Manage 18 (1999), pp. 103–110. Article |
64 C.T. Furstenberg, T.A. Ahles and M.B. Whedon et al., Knowledge and attitudes of health-care providers toward cancer pain management: a comparison of physicians, nurses, and pharmacists in the state of New Hampshire, J Pain Symptom Manage 15 (1998), pp. 335–349. Article |

Vitae
Dr. Rhiner is from Pallgesia Associates, Alta Loma, California.
Dr. von Gunten is Provost, Institute for Palliative Medicine at San Diego Hospice, San Diego, California.
Review
Michelle I. Rhiner RN, MSN, ACHPN![]()
![]()
| Referred to by: | The Challenges of Treating Patients with Cancer Pain The Journal of Supportive Oncology, Volume 8, Issue 6, November-December 2010, Pages 239-240, Sloan Beth Karver, Jessalyn H. Berger | |
| Referred to by: | A Conceptual Solution to Improve the Management of Cancer-Related Breakthrough Pain The Journal of Supportive Oncology, Volume 8, Issue 6, November-December 2010, Page 241, Wendy Ledesma, Toby C. Campbell | |
Abstract
Cancer breakthrough pain is a flare in pain that “breaks through” well-controlled persistent cancer pain. Although the condition is highly prevalent, the concept of cancer breakthrough pain is not well understood and is therefore underdiagnosed and undertreated. The purpose of this review is to examine the roles the health-care practitioner and patient/family caregiver play in the undertreatment of breakthrough pain. A lack of technical knowledge about pain management and pain assessment, attitudes about opioid addiction, and regulatory guidelines influence the manner in which opioids are prescribed. Patients harbor a variety of fears and misconceptions, such as opioid addiction, tolerance, side effects, and the meaning of pain, which can create a barrier to effective communication with their health-care provider regarding their cancer pain management and specifically their breakthrough pain. Identifying these issues gives health-care professionals and patients an opportunity to develop strategies that can improve the treatment of cancer breakthrough pain.
Article Outline
Adapted from Payne R.10
| CATEGORY OF PAIN | CHARACTERISTICS |
|---|---|
| Neuropathic | Caused by structural changes in the central nervous system and the peripheral nervous system; described as tingling, burning, or shooting in nature |
| Visceral | Deep cramping and tearing pain that may originate in internal organs |
| Somatic | Requires skeletal involvement; described as constant throbbing and aching that increases with movement |
A significant number of patients suffering from cancer experience these pain flares. In a single-population evaluation of cancer pain (n = 159), it was reported that of those patients with continuous pain, 57 (75%) experienced cancer BTP. More than half (54%) of those patients who experienced cancer pain reported it being related to particular activities, while a little over one-quarter of patients (26%) experienced pain idiopathic in nature and 16% of patients experienced end-of-dose pain.3 In a survey of 545 patients with cancer experiencing fluctuations in pain conducted by the American Pain Foundation, 96% of these patients experienced episodes of cancer BTP at least once a month, more than 70% experienced cancer BTP episodes at least once a week, and more than one-fifth (22%) experienced cancer BTP more than once a day.11
The inadequate treatment of cancer pain is an issue with far-reaching implications; therefore, the purpose of this review is to examine the roles that health-care providers, patients, and family caregivers play in the undertreatment of cancer BTP.
Defining Cancer BTP
Despite the reported prevalence of the condition, questions continue to surround the definition of cancer BTP. The earliest clinical accounts of cancer BTP describe it as a greater than moderate temporary flare in pain that occurs on a baseline of moderate pain in patients receiving opioids for cancer pain.8 Presently, descriptions of cancer BTP include statements that flares may occur in the presence of stable persistent pain (regardless of the treatment)12 having pain levels ranging from moderate to severe, with onset depending on the subtype of pain.13
Some controversy centers on the term “breakthrough pain.” The American-English term “breakthrough pain” does not have an exact translation to other languages. Physicians in Europe may use the term “episodic” or “transient” to describe these fluctuations in cancer pain. However, these terms do not capture the idea that the pain is above and beyond persistent pain control (eg, “breaks through baseline analgesia”). Furthermore, if the term “breakthrough pain” is used, it may be limited to descriptions of pain that occur at the end of the dosing cycle.14 Finally, there are physicians in some countries who do not view exacerbation of cancer pain as a separate clinical entity. Instead, they view spikes of pain as a predictable or normal element of cancer pain.14
In spite of the questions surrounding cancer BTP, persistent pain must be controlled before management of cancer BTP can proceed. In addition, repeated episodes of cancer BTP may indicate that baseline pain has not been properly assessed and not adequately managed.10 Therefore, cancer BTP and persistent cancer pain should be assessed independently as they are separate clinical conditions.
Treatment Approaches to Persistent Cancer Pain versus Cancer BTP
Persistent cancer pain requires around-the-clock (ATC) treatment with therapeutic agents that both maximize outcomes and minimize risks. ATC treatment allows maintenance of drug concentrations and prevents peaks and troughs resulting in increased risk of toxicity and lack of efficacy. Opioids such as morphine, oxycodone, methadone, and fentanyl are used extensively for the treatment of cancer pain as they produce an analgesic effect at a minimum dose and are easily titrated.15 While opioids are administrated orally per World Health Organization recommendations,15 they may also be administered rectally, intravenously, subcutaneously, intramuscularly, and transdermally. Clinical trials are being conducted to examine the administration of opioids through inhalation.16 Methadone is an example of a long-acting opioid that has been used to treat persistent pain.17 Pharmaceutically long-acting drugs such as sustained-release formulations of morphine, oxycodone, oxymorphone, and hydromorphone have been used to treat cancer pain.18 Adjuvant medications may be coadministered with opioids to treat symptoms that occur concurrently with cancer pain and to augment the analgesic effect. Although few clinical trials have evaluated the efficacy of adjuvants in patients with cancer, local anesthetics (eg, lidocaine), nonsteroidal anti-inflammatory drugs (eg, aspirin, naproxen, and ibuprofen), nonopioid analgesics (eg, acetaminophen), antidepressants (eg, amitriptyline, duloxetine, and venlafaxine), and anticonvulsants (eg, gabapentin, pregabalin, valproate, and lamotrigine) have been used to supplement opioid therapy.12
A number of physical and cognitive–behavioral interventions may be used in addition to pharmacological treatments to alleviate some of the pain symptoms experienced in patients with persistent cancer pain. In most patients, heating pads or ice packs may be used to relieve pain and reduce swelling; however, neither heat nor cold should be used on irradiated tissue, and caution should be used when using ice packs on patients with peripheral vascular disease.19 Exercise may be started or continued to improve physical conditioning. While tumor masses should not be manipulated, other techniques requiring physical stimulation, such as massage, pressure, and vibration, may also be used in the treatment of persistent pain. Either moving an immobile patient or temporarily restricting movement can prevent or alleviate pain. Acupuncture is another treatment that may be used to treat persistent cancer pain. Psychosocial interventions include hypnosis, use of relaxation techniques, biofeedback, and cognitive distraction.19
Orally administered, short-acting or rapid-acting opioids are used to treat cancer BTP episodes. However, the pharmacokinetic profiles of oral opioids may not match the onset and duration of some cancer BTP episodes.20 An analgesic agent used to treat cancer BTP should match the temporal characteristics of cancer BTP, be easily titrated to higher or lower doses if needed, and, if used appropriately in opioid-tolerant patients, not be associated with undue adverse effects.
Oral transmucosal delivery of opioids is an option for these challenges. Three formulations of the opioid fentanyl are available for transmucosal delivery: oral transmucosal fentanyl citrate (OTFC),21 fentanyl buccal tablets (FBTs),22 and fentanyl buccal soluble film (FBSF).23 OTFC is a fentanyl lozenge that has demonstrated an analgesic effect within 15 minutes of administration. Despite its rapid onset of action, the amount of fentanyl administered with the OTFC lozenge depends on the education provided to the patient on the use of this medication and the ability of the patient to actively use this product.21 While FBT (a tablet that utilizes an effervescent reaction to improve absorption) does not require substantial patient participation, its use has been associated with application-site side effects.22 Like OTFC and FBT, FBSF offers a rapid onset of action but does not require active patient participation and has minimal oral adverse side effects.23 These oral transmucosal fentanyl products should be administered only to opioid-tolerant patients, to avoid the risk of life-threatening respiratory depression.
Physician-Related Factors in Clinical Inertia
Three elements shape physicians' perceptions of the severity of cancer BTP and influence prescribing practices: technical knowledge, attitudes concerning use of opioids, and regulatory restrictions.[24], [25], [26] and [27]
The medical training of most physicians may not include courses in pain management. Instead, practitioners' education is a result of inpatient experience during the postgraduate years and is generally geared toward management of acute injuries, postoperative pain, and cardiovascular events such as myocardial infarctions.28 More evidence of this lack of knowledge among physicians is presented in a survey of British Columbian physicians.29 Among those doctors who responded, only 32% were aware of the dose that would produce an equal analgesic effect when a switch between morphine and acetaminophen is required. Furthermore, only 55% of physicians correctly stated that doses of opioids for cancer BTP should be 10% of the total daily dose and administered every 1 or 2 hours as needed per National Comprehensive Cancer Network guidelines. These findings suggest that when patients need stronger analgesics, physicians may not be skilled in converting to a more potent opioid or calculating a dose for cancer BTP management.29 Indiscriminate polypharmacy presents additional complications, whereby coadministration of multiple opioids exposes patients to an increased risk of toxicity and distorts the clinician's interpretation of outcomes.30
In addition to training, the communication skills of the practitioner play a role in effective treatment of cancer BTP and persistent cancer pain. Discussions regarding treatment can significantly influence a patient's decision to use opioids. In an interview study conducted during a cancer pain management trial, patients were reportedly occasionally “suspicious about the idea of choice” in the use of opioids.31 These patients favored thorough discussions of pain treatment options with physicians who were knowledgeable and confident about the use of opioids. The likelihood of patients participating in a pain management trial also increased if the physician stated that a lower dose of opioid would be used initially and treatment would cease if side effects developed.
Health-care providers' attitudes about the side effects of opioid treatment also impact treatment of persistent cancer pain and cancer BTP.32 In addition, although clinical studies do not support the assertion that patients may become addicted to opioids used to treat acute and persistent cancer pain, the belief that these patients are at risk for addiction prevents some health-care providers from prescribing opioids.33 The perceived relationship between opioid tolerance and addiction is one of the sources of confusion. One of the complications of opioid therapy is pharmacological tolerance, the loss of analgesic effect. Tolerance to an opioid may be determined genetically or acquired after metabolic changes, changes to receptors, or by learned behavior.33 The result of tolerance is decreasing pain relief, in spite of persistent doses over time.[34] and [35] Pharmacological tolerance is associated with incomplete cross-tolerance to other opioids. In contrast, addiction is a psychological syndrome characterized by uncontrolled and persistent use of the drug despite harm.[34], [35] and [36] Physical dependence on opioids is characterized by a withdrawal syndrome that occurs when the dose is stopped or decreased suddenly or an antagonist is administered.36 Pharmacological dependence is not synonymous with addiction; pharmacological tolerance occurs with many commonly prescribed drugs (such as nitrates) and does not indicate addiction.34
The prevalence of opioid addiction among a population of patients with cancer pain varies. In a meta-review of opioid addiction studies, authors reported a 0%–7.7% prevalence rate of addiction in patients with cancer depending on the population studied and the diagnostic measure used.37 This corresponds with the rate of addiction prevalent in the population at large—it does not support a cause-and-effect relationship between opioid administration and addiction.
Assessing and treating both persistent pain and cancer BTP in special populations such as the elderly and pediatric patients understandably presents a number of concerns for physicians. Although preclinical studies show that aging often correlates with increased pain sensitivity,[38], [39], [40] and [41] pain is often underdiagnosed in older patients.42 Age bias often shapes physicians' attitudes toward pain in the elderly. In a survey of 386 physicians to examine the attitudes, knowledge, and psychological factors that contribute to pain management decisions, about 31% believed that older patients were less likely to report pain than patients who were younger. The lack of studies designed to assess the efficacy of pain treatments in elderly patients and the unavailability of meta-analyses and systemic reviews to investigate use of opioids in the elderly are two reasons physicians are often hesitant in treating elderly patients with opioids.38 Another reason for the inadequate treatment of pain in the elderly is concern surrounding drug–drug interactions and uncontrolled side effects resulting from polypharmacy and variability in the patient population.43 The aging process results in impaired kidney and liver function and changes in body composition (including increases in body fat and changes in protein binding), which lead to alterations in drug distribution, metabolism, and elimination. Aging affects receptor responses and substrate intake in older individuals, and thus, the biological processes of increasing age influence the pharmacodynamics of drugs. Most elderly patients experience multiple comorbidities that require medications in addition to treatments for persistent pain and cancer BTP. It is not always possible to predict the drug–drug interactions and side effects that will often result from concomitant medications in this patient population.38 Finally, cognitive impairment and the inability to effectively communicate by the elderly make it difficult for physicians to properly assess pain in this patient population.44
Cancer BTP in children is often insufficiently treated because it is rarely assessed and poorly investigated. Additional research in this area would be useful in determining the true degree of risk associated with the use of opioids in children with cancer.[45] and [46]
Finally, practitioners working within the constraints of stringent laws and guidelines surrounding the allowed use of opioids may be reluctant to prescribe opioids given concerns over repercussions or uncertainty regarding appropriate protocols and documentation for use.[12], [47] and [48] Since cancer BTP episodes are not always properly assessed or recognized as separate clinical entities, opioids may not be prescribed in dosages sufficient to treat flares of persistent pain or more appropriate opioids that would best match the cancer BTP experience may not be considered.30 Risk evaluation and mitigation strategies49 and programs that require extensive documentation (such as writing prescriptions in triplicate29) may prevent health-care providers from prescribing opioids to patients experiencing cancer BTP. Physicians may also be hesitant to prescribe opioids because of the time and effort required for reimbursement by managed health-care companies.50
Patient-Related Factors in Clinical Inertia
Patient perceptions and beliefs about pain medication are similar to those of clinicians in some respects. In a study of patients in not-for-profit community hospitals and outpatients, both groups (27% of inpatients and 37% of outpatients) expressed a fear of addiction to pain medication.51 These patients also expressed the belief that medication should be saved until pain gets worse and that medication may interfere with daily activities.51 Like doctors, patients expected concerns about the side effects of pain medications including drowsiness, constipation, nausea, and difficulty breathing. Unlike doctors, a significant number of these patients also expressed concern about the cost of medication.51
Treatment decisions may be based on a number of factors including the patient's understanding of the diagnosis and confidence in the effectiveness of treatments.52 Patients may believe that pain is part of the cancer diagnosis and is to be expected. Some patients believe that use of opioids signifies that the “end of life” is near. They may believe that side effects related to opioids are unavoidable and the burden of the use of opioids outweighs the benefits.53
Another factor to consider when examining patient barriers to effective pain medication is the inability to effectively communicate pain to health-care professionals. The health-care setting may play a role in effectively describing pain. Patients in hospitals have the benefit of health-care providers being available to ask questions about their pain experience, which aids in describing the pain experience. Outpatients, on the other hand, may have difficulty in explaining their level of pain to family members and health-care providers.51 Patients may also be reluctant to describe their pain to family members or health-care professionals for fear of causing distress or appearing to be “weak”.54
Summary and Considerations
Many health-care professionals are not aware of the dual components of cancer pain: persistent pain and cancer BTP. This lack of understanding results in underdiagnosis and undertreatment of persistent cancer pain and cancer BTP, which can have significant repercussions on the patient–physician relationship. In a pan-European survey to examine the treatment of cancer pain, many patients reported feeling that clinicians did not prioritize the treatment of cancer pain highly enough; instead, treatment of cancer was given greater emphasis. The consequence of the lack of time committed to pain assessment and discussions geared toward the treatment options was that patients felt that their quality of life was not important to clinicians, their pain was not appreciated, and the physician did not know how to treat their pain.55
Several steps may be taken to close the gap between patients and physicians in the treatment of cancer BTP. Continuing education of physicians and other health-care professionals about the importance of persistent cancer pain and cancer BTP is essential to overcoming this obstacle.10 Properly prescribing opioids as treatments for cancer BTP and adherence to analgesic guidelines can prevent the undertreatment of this condition.56 Proper documentation of doses previously prescribed, side effects observed after dosing, and the severity of pain experienced may decrease the risk of adverse events.57
Improved pain management by practitioners can be achieved through knowledge and application of a number of validated pain assessment tools (Table 2). The patient self-report (in the form of a pain diary) is one of the most reliable methods of assessing persistent pain and cancer BTP; however, physicians must be careful to ensure accurate documentation of pain flares in patients who experience mental impairment or do not understand the concept of cancer BTP.10 Also, physical examination and appropriate tests are required to determine the pathophysiology of pain.12 In addition to the patient diary and the physical examination, there are a number of tools available to assess persistent pain and cancer BTP (Table 2).[58], [59] and [60]
| ASSESSMENT | |
|---|---|
| Unidimensional scales | Numerical Rating Scale (NRS) |
| Visual Analogue Scale (VAS) | |
| Verbal Rating Scale | |
| Wong Baker FACES Pain Rating Scale | |
| Colored Visual Analogue Scale | |
| Coping Strategies Questionnaire | |
| Multidimensional scales | Brief Pain Inventory |
| McGill Pain Questionnaire |
Pain assessments may be unidimensional or multidimensional. Unidimensional assessments such as the Visual Analogue Scale and the Numerical Rating Scale60 offer simple measurements for changes in pain intensity,61 are easy to use, and require minimal health-care provider involvement. Multidimensional pain assessment instruments like the Brief Pain Inventory and the McGill Pain Questionnaire, on the other hand, consider not only pain intensity but also dimensions such as changes or fluctuations in pain, treatments, location of pain, physical descriptions of pain sensations, emotions and feelings related to pain, duration of pain, and history of pain.61
An additional resource to utilize would be the pain knowledge of nurses. Performance-based testing to examine the deficiencies in assessment and management of cancer show that hospice-care nurses were more proficient in assessing pain intensity and pain location than resident and family physicians.[62], [63] and [64] Nurses also have the opportunity to spend more time with the patient and to assess response to treatment plans.56
Conclusions
Cancer BTP is underdiagnosed and undertreated due to a number of patient and health-care provider–related factors. By identifying misconceptions; providing education to health-care providers, patients, and family caregivers; and accessing all available resources to improve pain diagnosis and management, steps can be taken to ensure that patients experiencing persistent cancer pain and cancer BTP are properly treated.
References1
1 A. Caraceni, C. Martini and E. Zecca et al., Breakthrough pain characteristics and syndromes in patients with cancer pain: An international survey, Palliat Med 18 (2004), pp. 177–183. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (93)
2 R.K. Portenoy, D. Payne and P. Jacobsen, Breakthrough pain: characteristics and impact in patients with cancer pain, Pain 81 (1999), pp. 129–134. Article |
3 T. Gutgsell, D. Walsh, D.S. Zhukovsky, F. Gonzales and R. Lagman, A prospective study of the pathophysiology and clinical characteristics of pain in a palliative medicine population, Am J Hosp Palliat Care 20 (2003), pp. 140–148. View Record in Scopus | Cited By in Scopus (16)
4 S.S. Hwang, V.T. Chang and B. Kasimis, Cancer breakthrough pain characteristics and responses to treatment at a VA medical center, Pain 101 (2003), pp. 55–64. Article |
5 K.B. Svendsen, S. Andersen and S. Arnason et al., Breakthrough pain in malignant and non-malignant diseases: a review of prevalence, characteristics and mechanisms, Eur J Pain 9 (2005), pp. 195–206. Article |
6 M.P. Davis and D. Walsh, Epidemiology of cancer pain and factors influencing poor pain control, Am J Hosp Palliat Care 21 (2004), pp. 137–142. View Record in Scopus | Cited By in Scopus (32)
7 D.A. Fishbain, Pharmacotherapeutic management of breakthrough pain in patients with chronic persistent pain, Am J Manag Care 14 (suppl 1) (2008), pp. S123–S128. View Record in Scopus | Cited By in Scopus (5)
8 R.K. Portenoy and N.A. Hagen, Breakthrough pain: definition, prevalence and characteristics, Pain 41 (1990), pp. 273–281. Abstract |
9 Shoemaker S, Bruns D, Portenoy RK. Characteristics and impact of breakthrough pain (BTP) in noncancer and cancer-related chronic pain managed by clinicians who are not pain specialists. Presented at the American Academy of Pain Medicine 25th Annual Meeting, Honolulu, HI, January 28–31, 2009.
10 R. Payne, Recognition and diagnosis of breakthrough pain, Pain Med 8 (suppl 1) (2007), pp. S3–S7. View Record in Scopus | Cited By in Scopus (25)
11 American Pain Foundation, Breakthrough Cancer Pain Survey Fact Sheet http://www.painfoundation.org/learn/programs/spotlight-on-cancer-pain/breakthrough-pain/btcp-fact-sheet.pdf 2009.
12 National Cancer Institute, Pain (PDQ®) Health Professional Version 2010 http://www.cancer.gov/cancertopics/pdq/supportivecare/pain/HealthProfessional Accessed January 29, 2010.
13 J. Abrahm, A Physician's Guide to Pain and Symptom Management in Cancer Patients (2nd ed.), Johns Hopkins University Press, Baltimore (2005).
14 S.M. Colleau, The significance of breakthrough pain in cancer, Cancer Pain Release (1999), p. 12.
15 World Health Organization, Cancer Pain Relief (2nd ed.), World Health Organization, Geneva (1996).
16 S.J. Farr and B.A. Otulana, Pulmonary delivery of opioids as pain therapeutics, Adv Drug Deliv Rev 58 (2006), pp. 1076–1088. Article |
17 C.M. Terpening and W.M. Johnson, Methadone as an analgesic: a review of the risks and benefits, W V Med J 103 (2007), pp. 14–18. View Record in Scopus | Cited By in Scopus (10)
18 G. Zeppetella, Opioids for cancer breakthrough pain: a pilot study reporting patient assessment of time to meaningful pain relief, J Pain Symptom Manage 35 (2008), pp. 563–567. Article |
19 National Cancer Institute, Pain (PDR®) Health Professional Version 2009 http://www.cancer.gov/cancertopics/pdq/supportivecare/pain/HealthProfessional Accessed December 1, 2009.
20 National Comprehensive Cancer Network (NCCN), National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology, Adult Cancer Pain http://www.nccn.org/professionals/physician_gls/f_guidelines.asp Accessed May 10, 2010.
21 , Actiq (oral transmucosal fentanyl citrate lozenge) prescribing information Cepahlon, Inc.: Salt Lake City, UT http://www.actiq.com/pdf/actiq_package_insert_4_5_07.pdf Accessed May 21, 2010.
22 , Fentora (fentanyl buccal tablet) prescribing information Cephalon, Inc.: Frazer, PA http://www.fentora.com/pdfs/pdf100_prescribing_info.pdf Accessed May 21, 2010.
23 , Onsolis (fentanyl buccal soluble film) prescribing information Meda Pharmaceuticals, Inc.: Somerset, NJ http://onsolisfocus.com/pdf/pi.pdf Accessed May 21, 2010.
24 F. Brennan, D.B. Carr and M. Cousins, Pain management: a fundamental human right, Anesth Analg 105 (2007), pp. 205–221. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (109)
25 T.E. Elliott and B.A. Elliott, Physician attitudes and beliefs about use of morphine for cancer pain, J Pain Symptom Manage 7 (1992), pp. 141–148. Abstract |
26 B.A. Rich, An ethical analysis of the barriers to effective pain management, Camb Q Healthc Ethics 9 (2000), pp. 54–70. View Record in Scopus | Cited By in Scopus (27)
27 K.L. Sees and H.W. Clark, Opioid use in the treatment of chronic pain: assessment of addiction, J Pain Symptom Manage 8 (1993), pp. 257–264. Article |
28 J.E. Mortimer and N.L. Bartlett, Assessment of knowledge about cancer pain management by physicians in training, J Pain Symptom Manage 14 (1997), pp. 21–28. Abstract |
29 R. Gallagher, P. Hawley and W. Yeomans, A survey of cancer pain management knowledge and attitudes of British Columbian physicians, Pain Res Manag 9 (2004), pp. 188–194. View Record in Scopus | Cited By in Scopus (17)
30 B.G. Jenkins, P.H. Tuffin, C.L. Choo and S.A. Schug, Opioid prescribing: an assessment using quality statements, J Clin Pharm Ther 30 (2005), pp. 597–602. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (7)
31 C.M. Reid, R. Gooberman-Hill and G.W. Hanks, Opioid analgesics for cancer pain: symptom control for the living or comfort for the dying?: A qualitative study to investigate the factors influencing the decision to accept morphine for pain caused by cancer, Ann Oncol 19 (2008), pp. 44–48. View Record in Scopus | Cited By in Scopus (18)
32 R. Jacobsen, P. Sjogren, C. Moldrup and L. Christrup, Physician-related barriers to cancer pain management with opioid analgesics: a systematic review, J Opioid Manag 3 (2007), pp. 207–214. View Record in Scopus | Cited By in Scopus (18)
33 R. Benyamin, A.M. Trescot and S. Datta et al., Opioid complications and side effects, Pain Physician 11 (2008), pp. S105–S120. View Record in Scopus | Cited By in Scopus (86)
34 K. Bell and A. Salmon, Pain, physical dependence and pseudoaddiction: redefining addiction for “nice” people?, Int J Drug Policy 20 (2009), pp. 170–178. Article |
35 E.O. Dumas and G.M. Pollack, Opioid tolerance development: a pharmacokinetic/pharmacodynamic perspective, AAPS J 10 (2008), pp. 537–551. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (5)
36 American Pain Society, Definitions Related to the Use of Opioids for the Treatment of Pain http://www.ampainsoc.org/advocacy/opioids2.htm Accessed March 23, 2010.
37 J. Hojsted and P. Sjogren, Addiction to opioids in chronic pain patients: a literature review, Eur J Pain 11 (2007), pp. 490–518. Article |
38 O.H. Wilder-Smith, Opioid use in the elderly, Eur J Pain 9 (2005), pp. 137–140. Article |
39 D. Jourdan, S. Boghossian and A. Alloui et al., Age-related changes in nociception and effect of morphine in the Lou rat, Eur J Pain 4 (2000), pp. 291–300. Abstract |
40 E. Kramer and R.J. Bodnar, Age-related decrements in morphine analgesia: a parametric analysis, Neurobiol Aging 7 (1986), pp. 185–191. Abstract |
41 M.A. Smith and J.D. Gray, Age-related differences in sensitivity to the antinociceptive effects of opioids in male rats: Influence of nociceptive intensity and intrinsic efficacy at the mu receptor, Psychopharmacology (Berl) 156 (2001), pp. 445–453. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (26)
42 F.M. Gloth 3rd, Pain management in older adults: prevention and treatment, J Am Geriatr Soc 49 (2001), pp. 188–199. Full Text via CrossRef
43 S.M. Weinstein, L.F. Laux and J.I. Thornby et al., Physicians' attitudes toward pain and the use of opioid analgesics: results of a survey from the Texas Cancer Pain Initiative, South Med J 93 (2000), pp. 479–487. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (94)
44 B. Collett, S. O'Mahoney, P. Schofield, S.J. Closs and J. Potter, The assessment of pain in older people, Clin Med 7 (2007), pp. 496–500. View Record in Scopus | Cited By in Scopus (2)
45 R.A. Morrison, Update on sickle cell disease: incidence of addiction and choice of opioid in pain management, Pediatr Nurs 17 (1991), p. 503. View Record in Scopus | Cited By in Scopus (6)
46 C.H. Pegelow, Survey of pain management therapy provided for children with sickle cell disease, Clin Pediatr (Phila) 31 (1992), pp. 211–214. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (46)
47 C. Wright, S. Schnoll and D. Bernstein, Risk evaluation and mitigation strategies for drugs with abuse liability: public interest, special interest, conflicts of interest, and the industry perspective, Ann N Y Acad Sci 1141 (2008), pp. 284–303. View Record in Scopus | Cited By in Scopus (0)
48 M. Zenz, T. Zenz, M. Tryba and M. Strumpf, Severe undertreatment of cancer pain: a 3-year survey of the German situation, J Pain Symptom Manage 10 (1995), pp. 187–191. Abstract |
49 C. Wright, S. Schnoll and D. Bernstein, Risk evaluation and mitigation strategies for drug with abuse liability: public interest, special interest, conflicts of interest and industry perspective, Ann N Y Acad Sci 1141 (2008), pp. 284–303. View Record in Scopus | Cited By in Scopus (6)
50 D. Nemecek, Improving access to buprenorphine: A managed care company overcomes several obstacles to make this treatment more available, Behav Healthc 27 (2007), pp. 40–42. View Record in Scopus | Cited By in Scopus (1)
51 C.C. Corizzo, M.C. Baker and G.C. Henkelmann, Assessment of patient satisfaction with pain management in small community inpatient and outpatient settings, Oncol Nurs Forum 27 (2000), pp. 1279–1286. View Record in Scopus | Cited By in Scopus (10)
52 R. Sapir, R. Catane and B. Kaufman et al., Cancer patient expectations of and communication with oncologists and oncology nurses: the experience of an integrated oncology and palliative care service, Support Care Cancer 8 (2000), pp. 458–463. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (37)
53 K. Forbes, Opioids: beliefs and myths, J Pain Palliat Care Pharmacother 20 (2006), pp. 33–35. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (8)
54 P.E. Berry and S.E. Ward, Barriers to pain management in hospice: a study of family caregivers, Hosp J 10 (1995), pp. 19–33. View Record in Scopus | Cited By in Scopus (45)
55 H. Breivik, N. Cherny and B. Collett et al., Cancer-related pain: a pan-European survey of prevalence, treatment, and patient attitudes, Ann Oncol 20 (2009), pp. 1420–1433. Full Text via CrossRef | View Record in Scopus | Cited By in Scopus (36)
56 S.H. Neo, E.C. Loh and W.H. Koo, An audit of morphine prescribing in a hospice, Singapore Med J 42 (2001), pp. 417–419. View Record in Scopus | Cited By in Scopus (8)
57 M. Weber and C. Huber, Documentation of severe pain, opioid doses, and opioid-related side effects in outpatients with cancer: a retrospective study, J Pain Symptom Manage 17 (1999), pp. 49–54. Article |
58 F. De Conno, A. Caraceni and A. Gamba et al., Pain measurement in cancer patients: a comparison of six methods, Pain 57 (1994), pp. 161–166. Abstract |
59 G.D. Hakonsen, S. Hudson and T. Loennechen, Design and validation of a medication assessment tool for cancer pain management, Pharm World Sci 28 (2006), pp. 342–351. View Record in Scopus | Cited By in Scopus (5)
60 A. Caraceni, N. Cherny and R. Fainsinger et al., Pain measurement tools and methods in clinical research in palliative care: recommendations of an Expert Working Group of the European Association of Palliative Care, J Pain Symptom Manage 23 (2002), pp. 239–255. Article |
61 J.C. Holen, M.J. Hjermstad and J.H. Loge et al., Pain assessment tools: is the content appropriate for use in palliative care?, J Pain Symptom Manage 32 (2006), pp. 567–580. Article |
62 P.A. Sloan, M.B. Donnelly, B. Vanderveer, M. Delomas, R.W. Schwartz and D.A. Sloan, Cancer pain education among family physicians, J Pain Symptom Manage 14 (1997), pp. 74–81. Abstract |
63 P.A. Sloan, B.L. Vanderveer, J.S. Snapp, M. Johnson and D.A. Sloan, Cancer pain assessment and management recommendations by hospice nurses: University of Kentucky, Lexington, Kentucky, J Pain Symptom Manage 18 (1999), pp. 103–110. Article |
64 C.T. Furstenberg, T.A. Ahles and M.B. Whedon et al., Knowledge and attitudes of health-care providers toward cancer pain management: a comparison of physicians, nurses, and pharmacists in the state of New Hampshire, J Pain Symptom Manage 15 (1998), pp. 335–349. Article |

Vitae
Dr. Rhiner is from Pallgesia Associates, Alta Loma, California.
Dr. von Gunten is Provost, Institute for Palliative Medicine at San Diego Hospice, San Diego, California.
Cancer breakthrough pain is a flare in pain that “breaks through” well-controlled persistent cancer pain. Although the condition is highly prevalent, the concept of cancer breakthrough pain is not well understood and is therefore underdiagnosed and undertreated. The purpose of this review is to examine the roles the health-care practitioner and patient/family caregiver play in the undertreatment of breakthrough pain. A lack of technical knowledge about pain management and pain assessment, attitudes about opioid addiction, and regulatory guidelines influence the manner in which opioids are prescribed. Patients harbor a variety of fears and misconceptions, such as opioid addiction, tolerance, side effects, and the meaning of pain, which can create a barrier to effective communication with their health-care provider regarding their cancer pain management and specifically their breakthrough pain. Identifying these issues gives health-care professionals and patients an opportunity to develop strategies that can improve the treatment of cancer breakthrough pain.
AAD Maintains Vitamin D Recommendation in New Update
The American Academy of Dermatology still recommends vitamin D come from a healthy diet and supplements, not from unprotected skin exposure to ultraviolet radiation, according to their newly updated position statement.
The update comes in response to the Institute of Medicine's (IOM) recent review of existing data on possible roles for vitamin D status in certain types of cancers, and neurologic, infectious, autoimmune, and cardiovascular diseases. In the report, the IOM called existing evidence "inconsistent, inconclusive as to causality, and insufficient to inform nutritional requirement." However, the organization found that the evidence does support a role for vitamin D in bone health.
"The IOM's review of the scientific evidence about vitamin D supports the Academy's long-standing recommendation on safe ways to get this important vitamin – through a healthy diet which incorporates foods naturally rich in vitamin D, vitamin D–fortified foods and beverages, and vitamin D supplements," AAD president Dr. William D. James noted in a statement.
In particular, the statement reinforces the idea that unprotected UV exposure to the sun or indoor tanning devices is a known risk factor for the development of skin cancer. There is no scientifically validated, safe threshold level of UV exposure from the sun or indoor tanning devices that allows for maximal vitamin D synthesis without increasing skin cancer risk.
The Academy recommends a comprehensive photoprotective regimen, including the regular and proper use of a broad-spectrum sunscreen to protect against skin cancer.
The statement also includes new IOM Recommended Dietary Allowances (RDA) for calcium and vitamin D intake. Of note, these age-based values were derived assuming minimal or no sun exposure, given inconsistent contributions of sunlight to vitamin D in the population and also because of the risk of cancer associated with sun exposure.
Adequate vitamin D was defined by the IOM as blood levels of at least 20 ng/mL as measured in the United States (50 nmol/L as measured in Canada).
Physicians should discuss options for obtaining sufficient dietary or supplementary sources of vitamin D with patients having concerned about their vitamin D levels.
The American Academy of Dermatology still recommends vitamin D come from a healthy diet and supplements, not from unprotected skin exposure to ultraviolet radiation, according to their newly updated position statement.
The update comes in response to the Institute of Medicine's (IOM) recent review of existing data on possible roles for vitamin D status in certain types of cancers, and neurologic, infectious, autoimmune, and cardiovascular diseases. In the report, the IOM called existing evidence "inconsistent, inconclusive as to causality, and insufficient to inform nutritional requirement." However, the organization found that the evidence does support a role for vitamin D in bone health.
"The IOM's review of the scientific evidence about vitamin D supports the Academy's long-standing recommendation on safe ways to get this important vitamin – through a healthy diet which incorporates foods naturally rich in vitamin D, vitamin D–fortified foods and beverages, and vitamin D supplements," AAD president Dr. William D. James noted in a statement.
In particular, the statement reinforces the idea that unprotected UV exposure to the sun or indoor tanning devices is a known risk factor for the development of skin cancer. There is no scientifically validated, safe threshold level of UV exposure from the sun or indoor tanning devices that allows for maximal vitamin D synthesis without increasing skin cancer risk.
The Academy recommends a comprehensive photoprotective regimen, including the regular and proper use of a broad-spectrum sunscreen to protect against skin cancer.
The statement also includes new IOM Recommended Dietary Allowances (RDA) for calcium and vitamin D intake. Of note, these age-based values were derived assuming minimal or no sun exposure, given inconsistent contributions of sunlight to vitamin D in the population and also because of the risk of cancer associated with sun exposure.
Adequate vitamin D was defined by the IOM as blood levels of at least 20 ng/mL as measured in the United States (50 nmol/L as measured in Canada).
Physicians should discuss options for obtaining sufficient dietary or supplementary sources of vitamin D with patients having concerned about their vitamin D levels.
The American Academy of Dermatology still recommends vitamin D come from a healthy diet and supplements, not from unprotected skin exposure to ultraviolet radiation, according to their newly updated position statement.
The update comes in response to the Institute of Medicine's (IOM) recent review of existing data on possible roles for vitamin D status in certain types of cancers, and neurologic, infectious, autoimmune, and cardiovascular diseases. In the report, the IOM called existing evidence "inconsistent, inconclusive as to causality, and insufficient to inform nutritional requirement." However, the organization found that the evidence does support a role for vitamin D in bone health.
"The IOM's review of the scientific evidence about vitamin D supports the Academy's long-standing recommendation on safe ways to get this important vitamin – through a healthy diet which incorporates foods naturally rich in vitamin D, vitamin D–fortified foods and beverages, and vitamin D supplements," AAD president Dr. William D. James noted in a statement.
In particular, the statement reinforces the idea that unprotected UV exposure to the sun or indoor tanning devices is a known risk factor for the development of skin cancer. There is no scientifically validated, safe threshold level of UV exposure from the sun or indoor tanning devices that allows for maximal vitamin D synthesis without increasing skin cancer risk.
The Academy recommends a comprehensive photoprotective regimen, including the regular and proper use of a broad-spectrum sunscreen to protect against skin cancer.
The statement also includes new IOM Recommended Dietary Allowances (RDA) for calcium and vitamin D intake. Of note, these age-based values were derived assuming minimal or no sun exposure, given inconsistent contributions of sunlight to vitamin D in the population and also because of the risk of cancer associated with sun exposure.
Adequate vitamin D was defined by the IOM as blood levels of at least 20 ng/mL as measured in the United States (50 nmol/L as measured in Canada).
Physicians should discuss options for obtaining sufficient dietary or supplementary sources of vitamin D with patients having concerned about their vitamin D levels.
FROM THE AMERICAN ACADEMY OF DERMATOLOGY
Time Constraints Limit Full-Body Skin Exams
A majority of dermatologists, internists, and family physicians routinely perform full-body examinations for skin cancer – but time constraints and patient reluctance are common barriers to better exam rates, according to a report in the January issue of the Archives of Dermatology.
A total of 81% of dermatologists, 56% of internists, and 60% of family physicians in a nationally representative sample reported that they routinely perform the skin cancer examinations.
All three groups of physicians also identified barriers to performing skin examinations more often, but the barriers varied by specialty, said Susan A. Oliveria, Sc.D., of Memorial Sloan-Kettering Cancer Center’s dermatology service, New York, and her associates (Arch. Dermatol. 2011;147:39-44).
The investigators assessed skin cancer screening practices because little is known about the subject, and a better understanding both of the obstacles and of the facilitating factors could help improve primary and secondary screening.
To examine the issue, the researchers used data from a survey to which 1,669 physicians in group or solo private practice responded. The respondents included 679 dermatologists, 431 internists, and 559 family physicians. Most reported that they saw 200-600 patients per month, and most were aged 41-60 years.
Approximately 54% of internists and family physicians reported that time constraints prevented them from doing full-body skin exams more frequently, compared with only 31% of dermatologists. Half of the internists and family physicians also said that "competing comorbidities" often took priority over skin exams, compared with only 16% of dermatologists.
In contrast, significantly more dermatologists (44%) identified patient embarrassment or reluctance as a barrier to performing full-body skin exams, compared with internists (33%) or family physicians (31%). That is likely because most patients see a dermatologist for an isolated skin condition such as a wart or rash, the investigators noted, and do not expect to undress for a full-body exam.
In contrast, patients expect to undress for an internist or family physician, because disrobing for pelvic or rectal exams often is part of their annual physical examinations. Moreover, primary care physicians "have years to build up relationships with their patients," the study authors explained, while dermatologists typically do not.
To overcome that barrier, "dermatologists could educate their patients about the role of a full-body skin examination in a routine visit by providing them with written material to read and establishing a comforting physician-patient relationship," Dr. Oliveria and her colleagues said.
All the physicians reported that they were more likely to screen for skin cancer in patients who had one or more risk factors for the disease. Even so, only 87% of dermatologists, 65% of internists, and 70% of family physicians reported that they performed full-body exams in most of their high-risk patients.
Another common reason for performing a full-body skin cancer screen was the same for all three specialties: having a patient ask to have such an exam or to have a suspicious mole checked. That shows that it is important to maintain public education programs encouraging patients to request skin exams or mole checks, the researchers said.
Fewer internists (56%) and family physicians (54%) than dermatologists (78%) reported that their skill and expertise at performing skin exams and diagnosing skin cancer facilitated their screening practices. That finding suggests that enhanced dermatologic training during medical school and continuing medical education programs would be beneficial, the investigators added.
The National Cancer Institute supported the study. No financial conflicts of interest were reported.
A majority of dermatologists, internists, and family physicians routinely perform full-body examinations for skin cancer – but time constraints and patient reluctance are common barriers to better exam rates, according to a report in the January issue of the Archives of Dermatology.
A total of 81% of dermatologists, 56% of internists, and 60% of family physicians in a nationally representative sample reported that they routinely perform the skin cancer examinations.
All three groups of physicians also identified barriers to performing skin examinations more often, but the barriers varied by specialty, said Susan A. Oliveria, Sc.D., of Memorial Sloan-Kettering Cancer Center’s dermatology service, New York, and her associates (Arch. Dermatol. 2011;147:39-44).
The investigators assessed skin cancer screening practices because little is known about the subject, and a better understanding both of the obstacles and of the facilitating factors could help improve primary and secondary screening.
To examine the issue, the researchers used data from a survey to which 1,669 physicians in group or solo private practice responded. The respondents included 679 dermatologists, 431 internists, and 559 family physicians. Most reported that they saw 200-600 patients per month, and most were aged 41-60 years.
Approximately 54% of internists and family physicians reported that time constraints prevented them from doing full-body skin exams more frequently, compared with only 31% of dermatologists. Half of the internists and family physicians also said that "competing comorbidities" often took priority over skin exams, compared with only 16% of dermatologists.
In contrast, significantly more dermatologists (44%) identified patient embarrassment or reluctance as a barrier to performing full-body skin exams, compared with internists (33%) or family physicians (31%). That is likely because most patients see a dermatologist for an isolated skin condition such as a wart or rash, the investigators noted, and do not expect to undress for a full-body exam.
In contrast, patients expect to undress for an internist or family physician, because disrobing for pelvic or rectal exams often is part of their annual physical examinations. Moreover, primary care physicians "have years to build up relationships with their patients," the study authors explained, while dermatologists typically do not.
To overcome that barrier, "dermatologists could educate their patients about the role of a full-body skin examination in a routine visit by providing them with written material to read and establishing a comforting physician-patient relationship," Dr. Oliveria and her colleagues said.
All the physicians reported that they were more likely to screen for skin cancer in patients who had one or more risk factors for the disease. Even so, only 87% of dermatologists, 65% of internists, and 70% of family physicians reported that they performed full-body exams in most of their high-risk patients.
Another common reason for performing a full-body skin cancer screen was the same for all three specialties: having a patient ask to have such an exam or to have a suspicious mole checked. That shows that it is important to maintain public education programs encouraging patients to request skin exams or mole checks, the researchers said.
Fewer internists (56%) and family physicians (54%) than dermatologists (78%) reported that their skill and expertise at performing skin exams and diagnosing skin cancer facilitated their screening practices. That finding suggests that enhanced dermatologic training during medical school and continuing medical education programs would be beneficial, the investigators added.
The National Cancer Institute supported the study. No financial conflicts of interest were reported.
A majority of dermatologists, internists, and family physicians routinely perform full-body examinations for skin cancer – but time constraints and patient reluctance are common barriers to better exam rates, according to a report in the January issue of the Archives of Dermatology.
A total of 81% of dermatologists, 56% of internists, and 60% of family physicians in a nationally representative sample reported that they routinely perform the skin cancer examinations.
All three groups of physicians also identified barriers to performing skin examinations more often, but the barriers varied by specialty, said Susan A. Oliveria, Sc.D., of Memorial Sloan-Kettering Cancer Center’s dermatology service, New York, and her associates (Arch. Dermatol. 2011;147:39-44).
The investigators assessed skin cancer screening practices because little is known about the subject, and a better understanding both of the obstacles and of the facilitating factors could help improve primary and secondary screening.
To examine the issue, the researchers used data from a survey to which 1,669 physicians in group or solo private practice responded. The respondents included 679 dermatologists, 431 internists, and 559 family physicians. Most reported that they saw 200-600 patients per month, and most were aged 41-60 years.
Approximately 54% of internists and family physicians reported that time constraints prevented them from doing full-body skin exams more frequently, compared with only 31% of dermatologists. Half of the internists and family physicians also said that "competing comorbidities" often took priority over skin exams, compared with only 16% of dermatologists.
In contrast, significantly more dermatologists (44%) identified patient embarrassment or reluctance as a barrier to performing full-body skin exams, compared with internists (33%) or family physicians (31%). That is likely because most patients see a dermatologist for an isolated skin condition such as a wart or rash, the investigators noted, and do not expect to undress for a full-body exam.
In contrast, patients expect to undress for an internist or family physician, because disrobing for pelvic or rectal exams often is part of their annual physical examinations. Moreover, primary care physicians "have years to build up relationships with their patients," the study authors explained, while dermatologists typically do not.
To overcome that barrier, "dermatologists could educate their patients about the role of a full-body skin examination in a routine visit by providing them with written material to read and establishing a comforting physician-patient relationship," Dr. Oliveria and her colleagues said.
All the physicians reported that they were more likely to screen for skin cancer in patients who had one or more risk factors for the disease. Even so, only 87% of dermatologists, 65% of internists, and 70% of family physicians reported that they performed full-body exams in most of their high-risk patients.
Another common reason for performing a full-body skin cancer screen was the same for all three specialties: having a patient ask to have such an exam or to have a suspicious mole checked. That shows that it is important to maintain public education programs encouraging patients to request skin exams or mole checks, the researchers said.
Fewer internists (56%) and family physicians (54%) than dermatologists (78%) reported that their skill and expertise at performing skin exams and diagnosing skin cancer facilitated their screening practices. That finding suggests that enhanced dermatologic training during medical school and continuing medical education programs would be beneficial, the investigators added.
The National Cancer Institute supported the study. No financial conflicts of interest were reported.
FROM THE ARCHIVES OF DERMATOLOGY
Major Finding: 81% of dermatologists, 56% of internists, and 60% of family physicians report that they routinely perform full-body examinations for skin cancer.
Data Source: Survey of a representative sample of 679 dermatologists, 431 internists, and 559 family physicians.
Disclosures: The National Cancer Institute–supported the study. No financial conflicts of interest were reported.
Experts Prefer Mohs for Lentigo Maligna
ORLANDO - Standard surgical margins often are inadequate for lentigo maligna; instead, perimeter excision techniques or Mohs surgery is preferable because these approaches consistently provide high cure rates, according to Dr. Basil S. Cherpelis.
Lentigo maligna is a subtype of melanoma in situ, characterized as an overgrowth of atypical melanocytes with the potential to become lentigo maligna melanoma. "I think of it as the problem child of the melanoma family," Dr. Cherpelis said at the annual meeting of the Florida Society of Dermatologic Surgeons.
"What margin should I use, and if I am going to do Mohs, how can I see margins on frozen sections?" Intraoperative margin control is recommended, but recent literature suggests that standard 5-mm margins are frequently inadequate (Clin. Plast. Surg. 2010;37:35-46; Int. J. Dermatol. 2010;49:482-91).
In general, traditional frozen sections permit only 1% of margins to be assessed. For that reason, Dr. Cherpelis said he prefers permanent processing or Mohs surgery for these patients. With permanent processing, a surgeon leaves a central island of tissue and removes peripheral strips. Excised samples sent to a lab for evaluation typically take several days for results.
"Permanent processing has excellent cure rates. But on the downside, a patient has to wait for pathology results, which can be inconvenient," said Dr. Cherpelis, a cutaneous oncologist at Moffitt Cancer Center at the University of South Florida in Tampa.
In contrast, Mohs micrographic excision and repair are performed on the same day. Because the entire margin is visualized, cure rates are "excellent," Dr. Cherpelis said.
As to why Mohs isn’t more widely used, one possible reason is that it can be difficult to distinguish melanocytes from keratinocytes on frozen section, he said.
Immunohistostains to the rescue: Melanoma antigen recognized by T cells (MART-1) and microphthalmia-associated transcription factor (MITF) staining each has a role.
"MART-1 is a valuable immunostain for melanoma in situ on photodamaged skin," said Dr. L. Frank Glass, a dermatopathologist at Moffitt Cancer Center. MART-1 on frozen sections can provide the same data as permanent processing. "But MART-1 is not a magic bullet," he added. "There can be false positives."
In addition, expertise and teamwork are required with immunohistostains, Dr. Glass said. "The right MART-1 staining takes a technical staff to do it right."
MITF works in frozen sections and produces results similar to permanent sections. Clinicians can use this nuclear immunostain to quantify melanocytes and assess other parameters that distinguish melanoma in situ from solar lentigines or skin with chronic sun damage, for example.
"MITF is particularly useful in combination with MART-1," Dr. Glass said.
Instead of dendritic processes that overlap and look like confluence, MITF reveals discreet melanocytic cells, Dr. Glass said. "You can feel more comfortable calling a case negative with MITF – if it shows no abnormal cytology – in the same patient where the MART-1 shows clumping."
"The bottom line is we are looking for 100% specificity for melanoma," Dr. Glass said. He and his colleagues also are developing cutoff values, based on melanocyte diameter and density, to distinguish melanoma in situ from solar lentigo or sun-damaged skin.
Dr. Cherpelis and Dr. Glass said they had no relevant financial disclosures.
ORLANDO - Standard surgical margins often are inadequate for lentigo maligna; instead, perimeter excision techniques or Mohs surgery is preferable because these approaches consistently provide high cure rates, according to Dr. Basil S. Cherpelis.
Lentigo maligna is a subtype of melanoma in situ, characterized as an overgrowth of atypical melanocytes with the potential to become lentigo maligna melanoma. "I think of it as the problem child of the melanoma family," Dr. Cherpelis said at the annual meeting of the Florida Society of Dermatologic Surgeons.
"What margin should I use, and if I am going to do Mohs, how can I see margins on frozen sections?" Intraoperative margin control is recommended, but recent literature suggests that standard 5-mm margins are frequently inadequate (Clin. Plast. Surg. 2010;37:35-46; Int. J. Dermatol. 2010;49:482-91).
In general, traditional frozen sections permit only 1% of margins to be assessed. For that reason, Dr. Cherpelis said he prefers permanent processing or Mohs surgery for these patients. With permanent processing, a surgeon leaves a central island of tissue and removes peripheral strips. Excised samples sent to a lab for evaluation typically take several days for results.
"Permanent processing has excellent cure rates. But on the downside, a patient has to wait for pathology results, which can be inconvenient," said Dr. Cherpelis, a cutaneous oncologist at Moffitt Cancer Center at the University of South Florida in Tampa.
In contrast, Mohs micrographic excision and repair are performed on the same day. Because the entire margin is visualized, cure rates are "excellent," Dr. Cherpelis said.
As to why Mohs isn’t more widely used, one possible reason is that it can be difficult to distinguish melanocytes from keratinocytes on frozen section, he said.
Immunohistostains to the rescue: Melanoma antigen recognized by T cells (MART-1) and microphthalmia-associated transcription factor (MITF) staining each has a role.
"MART-1 is a valuable immunostain for melanoma in situ on photodamaged skin," said Dr. L. Frank Glass, a dermatopathologist at Moffitt Cancer Center. MART-1 on frozen sections can provide the same data as permanent processing. "But MART-1 is not a magic bullet," he added. "There can be false positives."
In addition, expertise and teamwork are required with immunohistostains, Dr. Glass said. "The right MART-1 staining takes a technical staff to do it right."
MITF works in frozen sections and produces results similar to permanent sections. Clinicians can use this nuclear immunostain to quantify melanocytes and assess other parameters that distinguish melanoma in situ from solar lentigines or skin with chronic sun damage, for example.
"MITF is particularly useful in combination with MART-1," Dr. Glass said.
Instead of dendritic processes that overlap and look like confluence, MITF reveals discreet melanocytic cells, Dr. Glass said. "You can feel more comfortable calling a case negative with MITF – if it shows no abnormal cytology – in the same patient where the MART-1 shows clumping."
"The bottom line is we are looking for 100% specificity for melanoma," Dr. Glass said. He and his colleagues also are developing cutoff values, based on melanocyte diameter and density, to distinguish melanoma in situ from solar lentigo or sun-damaged skin.
Dr. Cherpelis and Dr. Glass said they had no relevant financial disclosures.
ORLANDO - Standard surgical margins often are inadequate for lentigo maligna; instead, perimeter excision techniques or Mohs surgery is preferable because these approaches consistently provide high cure rates, according to Dr. Basil S. Cherpelis.
Lentigo maligna is a subtype of melanoma in situ, characterized as an overgrowth of atypical melanocytes with the potential to become lentigo maligna melanoma. "I think of it as the problem child of the melanoma family," Dr. Cherpelis said at the annual meeting of the Florida Society of Dermatologic Surgeons.
"What margin should I use, and if I am going to do Mohs, how can I see margins on frozen sections?" Intraoperative margin control is recommended, but recent literature suggests that standard 5-mm margins are frequently inadequate (Clin. Plast. Surg. 2010;37:35-46; Int. J. Dermatol. 2010;49:482-91).
In general, traditional frozen sections permit only 1% of margins to be assessed. For that reason, Dr. Cherpelis said he prefers permanent processing or Mohs surgery for these patients. With permanent processing, a surgeon leaves a central island of tissue and removes peripheral strips. Excised samples sent to a lab for evaluation typically take several days for results.
"Permanent processing has excellent cure rates. But on the downside, a patient has to wait for pathology results, which can be inconvenient," said Dr. Cherpelis, a cutaneous oncologist at Moffitt Cancer Center at the University of South Florida in Tampa.
In contrast, Mohs micrographic excision and repair are performed on the same day. Because the entire margin is visualized, cure rates are "excellent," Dr. Cherpelis said.
As to why Mohs isn’t more widely used, one possible reason is that it can be difficult to distinguish melanocytes from keratinocytes on frozen section, he said.
Immunohistostains to the rescue: Melanoma antigen recognized by T cells (MART-1) and microphthalmia-associated transcription factor (MITF) staining each has a role.
"MART-1 is a valuable immunostain for melanoma in situ on photodamaged skin," said Dr. L. Frank Glass, a dermatopathologist at Moffitt Cancer Center. MART-1 on frozen sections can provide the same data as permanent processing. "But MART-1 is not a magic bullet," he added. "There can be false positives."
In addition, expertise and teamwork are required with immunohistostains, Dr. Glass said. "The right MART-1 staining takes a technical staff to do it right."
MITF works in frozen sections and produces results similar to permanent sections. Clinicians can use this nuclear immunostain to quantify melanocytes and assess other parameters that distinguish melanoma in situ from solar lentigines or skin with chronic sun damage, for example.
"MITF is particularly useful in combination with MART-1," Dr. Glass said.
Instead of dendritic processes that overlap and look like confluence, MITF reveals discreet melanocytic cells, Dr. Glass said. "You can feel more comfortable calling a case negative with MITF – if it shows no abnormal cytology – in the same patient where the MART-1 shows clumping."
"The bottom line is we are looking for 100% specificity for melanoma," Dr. Glass said. He and his colleagues also are developing cutoff values, based on melanocyte diameter and density, to distinguish melanoma in situ from solar lentigo or sun-damaged skin.
Dr. Cherpelis and Dr. Glass said they had no relevant financial disclosures.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE FLORIDA SOCIETY OF DERMATOLOGIC SURGEONS