User login
Report details progress, obstacles in cancer research and care
Deaths from cancer are on the decline in the US, but new cases of cancer are on the rise, according to the 7th annual American Association for Cancer Research (AACR) Cancer Progress Report.
The data suggest the cancer death rate declined by 35% from 1991 to 2014 for children and by 25% for adults, a reduction that translates to 2.1 million cancer deaths avoided.
However, 600,920 people in the US are projected to die from cancer in 2017.
And the number of new cancer cases is predicted to rise from 1.7 million in 2017 to 2.3 million in 2030.
The report also estimates there will be 62,130 new cases of leukemia in 2017 and 24,500 leukemia deaths this year.
This includes:
- 5970 cases of acute lymphocytic leukemia and 1440 deaths
- 20,110 cases of chronic lymphocytic leukemia and 4660 deaths
- 21,380 cases of acute myeloid leukemia (AML) and 10,590 deaths
- 8950 cases of chronic myeloid leukemia and 1080 deaths.
The estimate for lymphomas is 80,500 new cases and 21,210 deaths.
This includes:
- 8260 cases of Hodgkin lymphoma (HL) and 1070 deaths
- 72,240 cases of non-Hodgkin lymphoma and 20,140 deaths.
The estimate for myeloma is 30,280 new cases and 12,590 deaths.
The report says the estimated new cases of cancer are based on cancer incidence rates from 49 states and the District of Columbia from 1995 through 2013, as reported by the North American Association of Central Cancer Registries. This represents about 98% of the US population.
The estimated deaths are based on US mortality data from 1997 through 2013, taken from the National Center for Health Statistics of the Centers for Disease Control and Prevention.
Drug approvals
The AACR report notes that, between August 1, 2016, and July 31, 2017, the US Food and Drug Administration (FDA) approved new uses for 15 anticancer agents, 9 of which had no previous FDA approval.
Five of the agents are immunotherapies, which the report dubs “revolutionary treatments that are increasing survival and improving quality of life for patients.”
Among the recently approved therapies are 3 used for hematology indications:
- Ibrutinib (Imbruvica), approved to treat patients with relapsed/refractory marginal zone lymphoma who require systemic therapy and have received at least 1 prior anti-CD20-based therapy
- Midostaurin (Rydapt), approved as monotherapy for adults with advanced systemic mastocytosis and for use in combination with standard cytarabine and daunorubicin induction, followed by cytarabine consolidation, in adults with newly diagnosed AML who are FLT3 mutation-positive, as detected by an FDA-approved test.
- Pembrolizumab (Keytruda), approved to treat adult and pediatric patients with refractory classical HL or those with classical HL who have relapsed after 3 or more prior lines of therapy.
Disparities and costs
The AACR report points out that advances against cancer have not benefited everyone equally, and cancer health disparities are some of the most pressing challenges.
Among the disparities listed is the fact that adolescents and young adults (ages 15 to 39) with AML have a 5-year relative survival rate that is 22% lower than that of children (ages 1 to 14) with AML.
And Hispanic children are 24% more likely to develop leukemia than non-Hispanic children.
Another concern mentioned in the report is the cost of cancer care. The direct medical costs of cancer care in 2014 were estimated to be nearly $87.6 billion. This number does not include the indirect costs of lost productivity due to cancer-related morbidity and mortality.
With this in mind, the AACR is calling for a $2 billion increase in funding for the National Institutes of Health in fiscal year 2018, for a total funding level of $36.2 billion.
The AACR also recommends an $80 million increase in the FDA budget, bringing it to $2.8 billion for fiscal year 2018. ![]()
Deaths from cancer are on the decline in the US, but new cases of cancer are on the rise, according to the 7th annual American Association for Cancer Research (AACR) Cancer Progress Report.
The data suggest the cancer death rate declined by 35% from 1991 to 2014 for children and by 25% for adults, a reduction that translates to 2.1 million cancer deaths avoided.
However, 600,920 people in the US are projected to die from cancer in 2017.
And the number of new cancer cases is predicted to rise from 1.7 million in 2017 to 2.3 million in 2030.
The report also estimates there will be 62,130 new cases of leukemia in 2017 and 24,500 leukemia deaths this year.
This includes:
- 5970 cases of acute lymphocytic leukemia and 1440 deaths
- 20,110 cases of chronic lymphocytic leukemia and 4660 deaths
- 21,380 cases of acute myeloid leukemia (AML) and 10,590 deaths
- 8950 cases of chronic myeloid leukemia and 1080 deaths.
The estimate for lymphomas is 80,500 new cases and 21,210 deaths.
This includes:
- 8260 cases of Hodgkin lymphoma (HL) and 1070 deaths
- 72,240 cases of non-Hodgkin lymphoma and 20,140 deaths.
The estimate for myeloma is 30,280 new cases and 12,590 deaths.
The report says the estimated new cases of cancer are based on cancer incidence rates from 49 states and the District of Columbia from 1995 through 2013, as reported by the North American Association of Central Cancer Registries. This represents about 98% of the US population.
The estimated deaths are based on US mortality data from 1997 through 2013, taken from the National Center for Health Statistics of the Centers for Disease Control and Prevention.
Drug approvals
The AACR report notes that, between August 1, 2016, and July 31, 2017, the US Food and Drug Administration (FDA) approved new uses for 15 anticancer agents, 9 of which had no previous FDA approval.
Five of the agents are immunotherapies, which the report dubs “revolutionary treatments that are increasing survival and improving quality of life for patients.”
Among the recently approved therapies are 3 used for hematology indications:
- Ibrutinib (Imbruvica), approved to treat patients with relapsed/refractory marginal zone lymphoma who require systemic therapy and have received at least 1 prior anti-CD20-based therapy
- Midostaurin (Rydapt), approved as monotherapy for adults with advanced systemic mastocytosis and for use in combination with standard cytarabine and daunorubicin induction, followed by cytarabine consolidation, in adults with newly diagnosed AML who are FLT3 mutation-positive, as detected by an FDA-approved test.
- Pembrolizumab (Keytruda), approved to treat adult and pediatric patients with refractory classical HL or those with classical HL who have relapsed after 3 or more prior lines of therapy.
Disparities and costs
The AACR report points out that advances against cancer have not benefited everyone equally, and cancer health disparities are some of the most pressing challenges.
Among the disparities listed is the fact that adolescents and young adults (ages 15 to 39) with AML have a 5-year relative survival rate that is 22% lower than that of children (ages 1 to 14) with AML.
And Hispanic children are 24% more likely to develop leukemia than non-Hispanic children.
Another concern mentioned in the report is the cost of cancer care. The direct medical costs of cancer care in 2014 were estimated to be nearly $87.6 billion. This number does not include the indirect costs of lost productivity due to cancer-related morbidity and mortality.
With this in mind, the AACR is calling for a $2 billion increase in funding for the National Institutes of Health in fiscal year 2018, for a total funding level of $36.2 billion.
The AACR also recommends an $80 million increase in the FDA budget, bringing it to $2.8 billion for fiscal year 2018. ![]()
Deaths from cancer are on the decline in the US, but new cases of cancer are on the rise, according to the 7th annual American Association for Cancer Research (AACR) Cancer Progress Report.
The data suggest the cancer death rate declined by 35% from 1991 to 2014 for children and by 25% for adults, a reduction that translates to 2.1 million cancer deaths avoided.
However, 600,920 people in the US are projected to die from cancer in 2017.
And the number of new cancer cases is predicted to rise from 1.7 million in 2017 to 2.3 million in 2030.
The report also estimates there will be 62,130 new cases of leukemia in 2017 and 24,500 leukemia deaths this year.
This includes:
- 5970 cases of acute lymphocytic leukemia and 1440 deaths
- 20,110 cases of chronic lymphocytic leukemia and 4660 deaths
- 21,380 cases of acute myeloid leukemia (AML) and 10,590 deaths
- 8950 cases of chronic myeloid leukemia and 1080 deaths.
The estimate for lymphomas is 80,500 new cases and 21,210 deaths.
This includes:
- 8260 cases of Hodgkin lymphoma (HL) and 1070 deaths
- 72,240 cases of non-Hodgkin lymphoma and 20,140 deaths.
The estimate for myeloma is 30,280 new cases and 12,590 deaths.
The report says the estimated new cases of cancer are based on cancer incidence rates from 49 states and the District of Columbia from 1995 through 2013, as reported by the North American Association of Central Cancer Registries. This represents about 98% of the US population.
The estimated deaths are based on US mortality data from 1997 through 2013, taken from the National Center for Health Statistics of the Centers for Disease Control and Prevention.
Drug approvals
The AACR report notes that, between August 1, 2016, and July 31, 2017, the US Food and Drug Administration (FDA) approved new uses for 15 anticancer agents, 9 of which had no previous FDA approval.
Five of the agents are immunotherapies, which the report dubs “revolutionary treatments that are increasing survival and improving quality of life for patients.”
Among the recently approved therapies are 3 used for hematology indications:
- Ibrutinib (Imbruvica), approved to treat patients with relapsed/refractory marginal zone lymphoma who require systemic therapy and have received at least 1 prior anti-CD20-based therapy
- Midostaurin (Rydapt), approved as monotherapy for adults with advanced systemic mastocytosis and for use in combination with standard cytarabine and daunorubicin induction, followed by cytarabine consolidation, in adults with newly diagnosed AML who are FLT3 mutation-positive, as detected by an FDA-approved test.
- Pembrolizumab (Keytruda), approved to treat adult and pediatric patients with refractory classical HL or those with classical HL who have relapsed after 3 or more prior lines of therapy.
Disparities and costs
The AACR report points out that advances against cancer have not benefited everyone equally, and cancer health disparities are some of the most pressing challenges.
Among the disparities listed is the fact that adolescents and young adults (ages 15 to 39) with AML have a 5-year relative survival rate that is 22% lower than that of children (ages 1 to 14) with AML.
And Hispanic children are 24% more likely to develop leukemia than non-Hispanic children.
Another concern mentioned in the report is the cost of cancer care. The direct medical costs of cancer care in 2014 were estimated to be nearly $87.6 billion. This number does not include the indirect costs of lost productivity due to cancer-related morbidity and mortality.
With this in mind, the AACR is calling for a $2 billion increase in funding for the National Institutes of Health in fiscal year 2018, for a total funding level of $36.2 billion.
The AACR also recommends an $80 million increase in the FDA budget, bringing it to $2.8 billion for fiscal year 2018. ![]()
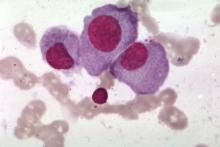
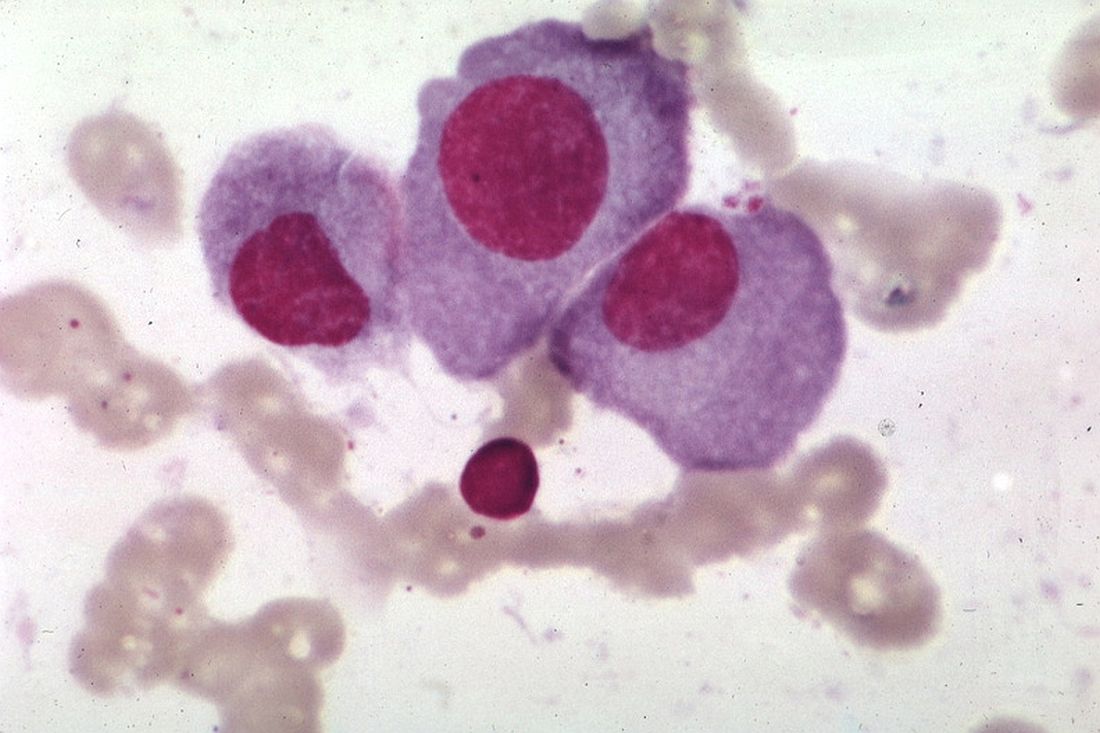
KW-2478 examined as novel add-on therapy for myeloma
Bortezomib and KW-2478, a novel nonansamycin heat shock protein 90 (Hsp90) inhibitor, had modest activity and was well tolerated in an open-label phase 1/2 study of patients with relapsed or refractory multiple myeloma.
The objective response rate in 79 evaluable patients treated with the combination was 39.2%, and the clinical benefit rate was 51.9%. Median progression-free survival was 6.7 months, and median duration of response was 5.5 months, according to a report by Jamie Cavenagh, MD, of St. Bartholomew’s Hospital, West Smithfield, London, and colleagues. The results were published online in the British Journal of Cancer.
KW-2478 showed synergistic antitumor activity with bortezomib in preclinical in vitro and in vivo studies, and the recommended phase 2 intravenous dosage of 175 mg/m2, along with 1.3 mg/m2 of bortezomib on days 1, 4, 8, and 11 of each 21-day cycle (up to eight cycles) in the current study was determined by phase 1 dose escalation; the maximum tolerated dose was not reached.
Although the antimyeloma activity of this novel treatment combination was relatively modest, the tolerability and apparent lack of overlapping toxicity suggest it deserves further exploration, including with alternate dosing schedules and combinations, for the treatment of relapsed/refractory multiple myeloma, the investigators concluded, noting that KW-2478 should also be studied in combination with other antimyeloma agents.
Kyowa Kirin Pharmaceutical Development Inc. funded the study. Dr. Cavenagh reported having no disclosures. Several other authors reported employment by the sponsor or by Kyowa Hakko Kirin Co. Ltd. Another, K. Yong, MD, reported receiving support from the National Institute for Health Research.
Bortezomib and KW-2478, a novel nonansamycin heat shock protein 90 (Hsp90) inhibitor, had modest activity and was well tolerated in an open-label phase 1/2 study of patients with relapsed or refractory multiple myeloma.
The objective response rate in 79 evaluable patients treated with the combination was 39.2%, and the clinical benefit rate was 51.9%. Median progression-free survival was 6.7 months, and median duration of response was 5.5 months, according to a report by Jamie Cavenagh, MD, of St. Bartholomew’s Hospital, West Smithfield, London, and colleagues. The results were published online in the British Journal of Cancer.
KW-2478 showed synergistic antitumor activity with bortezomib in preclinical in vitro and in vivo studies, and the recommended phase 2 intravenous dosage of 175 mg/m2, along with 1.3 mg/m2 of bortezomib on days 1, 4, 8, and 11 of each 21-day cycle (up to eight cycles) in the current study was determined by phase 1 dose escalation; the maximum tolerated dose was not reached.
Although the antimyeloma activity of this novel treatment combination was relatively modest, the tolerability and apparent lack of overlapping toxicity suggest it deserves further exploration, including with alternate dosing schedules and combinations, for the treatment of relapsed/refractory multiple myeloma, the investigators concluded, noting that KW-2478 should also be studied in combination with other antimyeloma agents.
Kyowa Kirin Pharmaceutical Development Inc. funded the study. Dr. Cavenagh reported having no disclosures. Several other authors reported employment by the sponsor or by Kyowa Hakko Kirin Co. Ltd. Another, K. Yong, MD, reported receiving support from the National Institute for Health Research.
Bortezomib and KW-2478, a novel nonansamycin heat shock protein 90 (Hsp90) inhibitor, had modest activity and was well tolerated in an open-label phase 1/2 study of patients with relapsed or refractory multiple myeloma.
The objective response rate in 79 evaluable patients treated with the combination was 39.2%, and the clinical benefit rate was 51.9%. Median progression-free survival was 6.7 months, and median duration of response was 5.5 months, according to a report by Jamie Cavenagh, MD, of St. Bartholomew’s Hospital, West Smithfield, London, and colleagues. The results were published online in the British Journal of Cancer.
KW-2478 showed synergistic antitumor activity with bortezomib in preclinical in vitro and in vivo studies, and the recommended phase 2 intravenous dosage of 175 mg/m2, along with 1.3 mg/m2 of bortezomib on days 1, 4, 8, and 11 of each 21-day cycle (up to eight cycles) in the current study was determined by phase 1 dose escalation; the maximum tolerated dose was not reached.
Although the antimyeloma activity of this novel treatment combination was relatively modest, the tolerability and apparent lack of overlapping toxicity suggest it deserves further exploration, including with alternate dosing schedules and combinations, for the treatment of relapsed/refractory multiple myeloma, the investigators concluded, noting that KW-2478 should also be studied in combination with other antimyeloma agents.
Kyowa Kirin Pharmaceutical Development Inc. funded the study. Dr. Cavenagh reported having no disclosures. Several other authors reported employment by the sponsor or by Kyowa Hakko Kirin Co. Ltd. Another, K. Yong, MD, reported receiving support from the National Institute for Health Research.
FROM THE BRITISH JOURNAL OF CANCER
Key clinical point:
Major finding: The objective response rate was 39.2%, and the clinical benefit rate was 51.9%.
Data source: An open-label phase 1/2 study of 95 patients.
Disclosures: Kyowa Kirin Pharmaceutical Development Inc. funded the study. Dr. Cavenagh reported having no disclosures. Several other authors reported employment by the sponsor or by Kyowa Hakko Kirin Co. Ltd. Another, K. Yong, MD, reported receiving support from the National Institute for Health Research.
Biosimilar matches rituximab in large follicular lymphoma trial
MADRID – The biosimilar GP2013 met the criteria for therapeutic equivalence to rituximab in a large comparator trial of previously untreated patients with aggressive follicular lymphoma.
Based on results presented at the European Society for Medical Oncology Congress, “there is absolutely no difference in the objective response rates for GP2013 and rituximab,” the primary endpoint of the trial, reported Wojciech Jurczak, MD, PhD, head of lymphoma, department of hematology, Jagiellonian University, Krakow, Poland.
Dr. Jurczak characterized the double-blind, randomized phase 3 trial ASSIST_FL (NCT01419665) as the “largest biosimilar trial in hematology.” For the study, 629 patients with previously untreated, advanced-stage follicular lymphoma were randomized to GP2013 and the conventional regimen of cyclophosphamide, vincristine, and prednisone (G-CVP) or rituximab and the same three-agent regimen (R-CVP). Each regimen was given for eight cycles followed by maintenance monotherapy with the assigned monoclonal antibody for up to 2 years.
The primary endpoint, objective response rate (ORR), was 87.1% and 87.5% for G-CVP and R-CVP, respectively. There were no differences in ORR among the subgroups evaluated, which included patients aged 60 years and older vs. younger patients, presence or absence of bulky disease, high or low FLIPI (Follicular Lymphoma International Prognostic Index) score, gender, and the geographic region where treatment was given.
Safety was a secondary endpoint evaluated at the end of eight cycles and again after a year of maintenance therapy. The proportion of patients with any adverse event and the proportion with grade 3 or greater adverse events were not statistically different at any time point. There were also no significant differences in any of the other secondary endpoints evaluated, which included pharmacokinetic, pharmacodynamic, and immune measures.
The results are consistent with those of a related randomized bioequivalence trial comparing GP2013 and rituximab in 312 patients with rheumatoid arthritis (Ann Rheum Dis. 2017;76:1598-1602). The primary endpoint in that trial was area-under-the-curve serum concentration time.
The two studies are mutually reinforcing, and “the ASSIST_FL trial ends the story. We have the totality of evidence that GP2013 can be considered a biosimilar,” said Dr. Jurczak, who anticipates “major price differences” for this agent relative to rituximab.
“We may, based on the results of the follicular lymphoma trial, use the biosimilar in all registered indications for rituximab,” said Dr. Jurczak. This is also the conclusion of the European Medicine Agency, which approved this agent in June 2017 for all rituximab indications.
In explaining the process for approval of biosimilars, Dr. Jurczak emphasized that demonstrating bioequivalence is not the same as the approval process for a new therapeutic agent, for which regulatory agents require a demonstration of efficacy on a meaningful clinical endpoint, such as progression-free survival. For biosimilars, it is not necessary to show clinical benefits. Biosimilars must demonstrate the same biological activity, and ORR is considered an acceptable measure.
The ESMO-invited discussant, Michele Ghielmini, MD, PhD, medical director at the Oncology Institute of Southern Switzerland, Bellinzona, agreed. “We can reasonably speculate that (when rituximab and the biosimilar are associated with the same) response rate, they will lead to the same clinical benefits.”
MADRID – The biosimilar GP2013 met the criteria for therapeutic equivalence to rituximab in a large comparator trial of previously untreated patients with aggressive follicular lymphoma.
Based on results presented at the European Society for Medical Oncology Congress, “there is absolutely no difference in the objective response rates for GP2013 and rituximab,” the primary endpoint of the trial, reported Wojciech Jurczak, MD, PhD, head of lymphoma, department of hematology, Jagiellonian University, Krakow, Poland.
Dr. Jurczak characterized the double-blind, randomized phase 3 trial ASSIST_FL (NCT01419665) as the “largest biosimilar trial in hematology.” For the study, 629 patients with previously untreated, advanced-stage follicular lymphoma were randomized to GP2013 and the conventional regimen of cyclophosphamide, vincristine, and prednisone (G-CVP) or rituximab and the same three-agent regimen (R-CVP). Each regimen was given for eight cycles followed by maintenance monotherapy with the assigned monoclonal antibody for up to 2 years.
The primary endpoint, objective response rate (ORR), was 87.1% and 87.5% for G-CVP and R-CVP, respectively. There were no differences in ORR among the subgroups evaluated, which included patients aged 60 years and older vs. younger patients, presence or absence of bulky disease, high or low FLIPI (Follicular Lymphoma International Prognostic Index) score, gender, and the geographic region where treatment was given.
Safety was a secondary endpoint evaluated at the end of eight cycles and again after a year of maintenance therapy. The proportion of patients with any adverse event and the proportion with grade 3 or greater adverse events were not statistically different at any time point. There were also no significant differences in any of the other secondary endpoints evaluated, which included pharmacokinetic, pharmacodynamic, and immune measures.
The results are consistent with those of a related randomized bioequivalence trial comparing GP2013 and rituximab in 312 patients with rheumatoid arthritis (Ann Rheum Dis. 2017;76:1598-1602). The primary endpoint in that trial was area-under-the-curve serum concentration time.
The two studies are mutually reinforcing, and “the ASSIST_FL trial ends the story. We have the totality of evidence that GP2013 can be considered a biosimilar,” said Dr. Jurczak, who anticipates “major price differences” for this agent relative to rituximab.
“We may, based on the results of the follicular lymphoma trial, use the biosimilar in all registered indications for rituximab,” said Dr. Jurczak. This is also the conclusion of the European Medicine Agency, which approved this agent in June 2017 for all rituximab indications.
In explaining the process for approval of biosimilars, Dr. Jurczak emphasized that demonstrating bioequivalence is not the same as the approval process for a new therapeutic agent, for which regulatory agents require a demonstration of efficacy on a meaningful clinical endpoint, such as progression-free survival. For biosimilars, it is not necessary to show clinical benefits. Biosimilars must demonstrate the same biological activity, and ORR is considered an acceptable measure.
The ESMO-invited discussant, Michele Ghielmini, MD, PhD, medical director at the Oncology Institute of Southern Switzerland, Bellinzona, agreed. “We can reasonably speculate that (when rituximab and the biosimilar are associated with the same) response rate, they will lead to the same clinical benefits.”
MADRID – The biosimilar GP2013 met the criteria for therapeutic equivalence to rituximab in a large comparator trial of previously untreated patients with aggressive follicular lymphoma.
Based on results presented at the European Society for Medical Oncology Congress, “there is absolutely no difference in the objective response rates for GP2013 and rituximab,” the primary endpoint of the trial, reported Wojciech Jurczak, MD, PhD, head of lymphoma, department of hematology, Jagiellonian University, Krakow, Poland.
Dr. Jurczak characterized the double-blind, randomized phase 3 trial ASSIST_FL (NCT01419665) as the “largest biosimilar trial in hematology.” For the study, 629 patients with previously untreated, advanced-stage follicular lymphoma were randomized to GP2013 and the conventional regimen of cyclophosphamide, vincristine, and prednisone (G-CVP) or rituximab and the same three-agent regimen (R-CVP). Each regimen was given for eight cycles followed by maintenance monotherapy with the assigned monoclonal antibody for up to 2 years.
The primary endpoint, objective response rate (ORR), was 87.1% and 87.5% for G-CVP and R-CVP, respectively. There were no differences in ORR among the subgroups evaluated, which included patients aged 60 years and older vs. younger patients, presence or absence of bulky disease, high or low FLIPI (Follicular Lymphoma International Prognostic Index) score, gender, and the geographic region where treatment was given.
Safety was a secondary endpoint evaluated at the end of eight cycles and again after a year of maintenance therapy. The proportion of patients with any adverse event and the proportion with grade 3 or greater adverse events were not statistically different at any time point. There were also no significant differences in any of the other secondary endpoints evaluated, which included pharmacokinetic, pharmacodynamic, and immune measures.
The results are consistent with those of a related randomized bioequivalence trial comparing GP2013 and rituximab in 312 patients with rheumatoid arthritis (Ann Rheum Dis. 2017;76:1598-1602). The primary endpoint in that trial was area-under-the-curve serum concentration time.
The two studies are mutually reinforcing, and “the ASSIST_FL trial ends the story. We have the totality of evidence that GP2013 can be considered a biosimilar,” said Dr. Jurczak, who anticipates “major price differences” for this agent relative to rituximab.
“We may, based on the results of the follicular lymphoma trial, use the biosimilar in all registered indications for rituximab,” said Dr. Jurczak. This is also the conclusion of the European Medicine Agency, which approved this agent in June 2017 for all rituximab indications.
In explaining the process for approval of biosimilars, Dr. Jurczak emphasized that demonstrating bioequivalence is not the same as the approval process for a new therapeutic agent, for which regulatory agents require a demonstration of efficacy on a meaningful clinical endpoint, such as progression-free survival. For biosimilars, it is not necessary to show clinical benefits. Biosimilars must demonstrate the same biological activity, and ORR is considered an acceptable measure.
The ESMO-invited discussant, Michele Ghielmini, MD, PhD, medical director at the Oncology Institute of Southern Switzerland, Bellinzona, agreed. “We can reasonably speculate that (when rituximab and the biosimilar are associated with the same) response rate, they will lead to the same clinical benefits.”
AT ESMO 2017
Key clinical point:
Major finding: The objective response rates were 87.1% and 87.5% for G-CVP and R-CVP.
Data source: Double-blind, multicenter randomized trial of 629 patients with previously untreated, advanced-stage follicular lymphoma.
Disclosures: Dr. Jurczak reported financial relationships with Sandoz.
Biosimilar deemed equivalent to reference drug in FL
MADRID—The biosimilar GP2013 has demonstrated equivalence to its reference drug rituximab in patients with previously untreated, advanced-stage follicular lymphoma (FL), according to researchers.
Treatment with GP2013 plus cyclophosphamide, vincristine, and prednisone (CVP) produced a similar overall response rate (ORR) as rituximab plus CVP in the phase 3 ASSIST-FL trial.
Survival rates were also similar between the treatment arms, as were adverse events (AEs).
Results from this study were published in The Lancet Haematology and presented at ESMO 2017 Congress (abstract 994O).
The study was funded by Hexal AG, a Sandoz company (part of the Novartis group), which is marketing GP2013 as Rixathon in Europe.
Patients and treatment
The trial included 629 patients with previously untreated, advanced-stage FL. They were randomized to receive 8 cycles of GP2013-CVP (n=314) or rituximab-CVP (n=315). Responders in either arm could receive monotherapy maintenance for up to 2 years.
The mean age was 57.5 in the GP2013 arm and 56.4 in the rituximab arm. Fifty-eight percent and 54% of patients, respectively, were female.
Fifty-seven percent of patients in the GP2013 arm and 56% in the rituximab arm had an ECOG performance status of 0. Forty percent and 39%, respectively, had a status of 1. Two percent and 4%, respectively, had a status of 2. (For the remaining 1% of patients in each arm, data on performance status were missing.)
Patients had an Ann Arbor stage of III—46% in the GP2013 arm and 43% in the GP2013 arm—or IV—54% in the GP2013 arm and 57% in the rituximab arm.
Fifty-six percent of patients in each arm were high-risk according to FLIPI. Thirty-four percent in the GP2013 arm and 33% in the rituximab arm were intermediate-risk. Ten percent and 11%, respectively, were low-risk.
Fourteen percent of patients in the GP2013 arm and 18% in the rituximab arm had bulky disease. Fifteen percent and 13%, respectively, had splenic involvement.
ORR and survival
The patients had a median follow-up of 23.8 months. The primary efficacy endpoint was equivalence in ORR, defined by a 95% confidence interval (CI) with a margin of ± 12% standard deviation.
The primary endpoint was met, as the ORR was 87% in the GP2013 arm and 88% in the rituximab arm, with a difference of –0.40% (95% CI –5.94%, 5.14%).
The complete response rate was 15% in the GP2013 arm and 13% in the rituximab arm. The partial response rates were 72% and 74%, respectively.
The median progression-free survival and overall survival have not been reached. However, the progression-free survival rate was 70% in the GP2013 arm and 76% in the rituximab arm (hazard ratio [HR]=1.31; 95% CI 1.02, 1.69).
The overall survival rate was 93% in the GP2013 arm and 91% in the rituximab arm (HR=0.77; 95% CI 0.49, 1.22).
Safety
During the combination phase, the incidence of AEs was 93% in the GP2013 arm and 91% in the rituximab arm. The incidence of serious AEs was 23% and 20%, respectively.
The most frequent AEs (in the GP2013 and rituximab arms, respectively) were neutropenia (26% and 30%), constipation (22% and 20%), and nausea (16% and 13%). The most common grade 3/4 AE was neutropenia (18% and 21%).
There were 11 deaths reported during the combination phase—4 in the GP2013 arm and 7 in the rituximab arm.
Three deaths in the GP2013 arm (sudden death, septic shock, and respiratory failure) and 2 deaths in the rituximab arm (multiple organ dysfunction syndrome and sepsis) were suspected to be related to study treatment.
During the maintenance phase, the incidence of AEs was 63% in the GP2013 arm and 57% in the rituximab arm. The incidence of serious AEs was 6% and 4%, respectively.
The most frequent AEs (in the GP2013 and rituximab arms, respectively) were infections and infestations (20% and 27%), neutropenia (10% and 6%), cough (9% and 6%), and upper respiratory tract infection (3% and 6%). The most common grade 3/4 AE was neutropenia (7% and 4%).
There were 4 deaths reported during the maintenance phase, 2 in each treatment arm. ![]()
MADRID—The biosimilar GP2013 has demonstrated equivalence to its reference drug rituximab in patients with previously untreated, advanced-stage follicular lymphoma (FL), according to researchers.
Treatment with GP2013 plus cyclophosphamide, vincristine, and prednisone (CVP) produced a similar overall response rate (ORR) as rituximab plus CVP in the phase 3 ASSIST-FL trial.
Survival rates were also similar between the treatment arms, as were adverse events (AEs).
Results from this study were published in The Lancet Haematology and presented at ESMO 2017 Congress (abstract 994O).
The study was funded by Hexal AG, a Sandoz company (part of the Novartis group), which is marketing GP2013 as Rixathon in Europe.
Patients and treatment
The trial included 629 patients with previously untreated, advanced-stage FL. They were randomized to receive 8 cycles of GP2013-CVP (n=314) or rituximab-CVP (n=315). Responders in either arm could receive monotherapy maintenance for up to 2 years.
The mean age was 57.5 in the GP2013 arm and 56.4 in the rituximab arm. Fifty-eight percent and 54% of patients, respectively, were female.
Fifty-seven percent of patients in the GP2013 arm and 56% in the rituximab arm had an ECOG performance status of 0. Forty percent and 39%, respectively, had a status of 1. Two percent and 4%, respectively, had a status of 2. (For the remaining 1% of patients in each arm, data on performance status were missing.)
Patients had an Ann Arbor stage of III—46% in the GP2013 arm and 43% in the GP2013 arm—or IV—54% in the GP2013 arm and 57% in the rituximab arm.
Fifty-six percent of patients in each arm were high-risk according to FLIPI. Thirty-four percent in the GP2013 arm and 33% in the rituximab arm were intermediate-risk. Ten percent and 11%, respectively, were low-risk.
Fourteen percent of patients in the GP2013 arm and 18% in the rituximab arm had bulky disease. Fifteen percent and 13%, respectively, had splenic involvement.
ORR and survival
The patients had a median follow-up of 23.8 months. The primary efficacy endpoint was equivalence in ORR, defined by a 95% confidence interval (CI) with a margin of ± 12% standard deviation.
The primary endpoint was met, as the ORR was 87% in the GP2013 arm and 88% in the rituximab arm, with a difference of –0.40% (95% CI –5.94%, 5.14%).
The complete response rate was 15% in the GP2013 arm and 13% in the rituximab arm. The partial response rates were 72% and 74%, respectively.
The median progression-free survival and overall survival have not been reached. However, the progression-free survival rate was 70% in the GP2013 arm and 76% in the rituximab arm (hazard ratio [HR]=1.31; 95% CI 1.02, 1.69).
The overall survival rate was 93% in the GP2013 arm and 91% in the rituximab arm (HR=0.77; 95% CI 0.49, 1.22).
Safety
During the combination phase, the incidence of AEs was 93% in the GP2013 arm and 91% in the rituximab arm. The incidence of serious AEs was 23% and 20%, respectively.
The most frequent AEs (in the GP2013 and rituximab arms, respectively) were neutropenia (26% and 30%), constipation (22% and 20%), and nausea (16% and 13%). The most common grade 3/4 AE was neutropenia (18% and 21%).
There were 11 deaths reported during the combination phase—4 in the GP2013 arm and 7 in the rituximab arm.
Three deaths in the GP2013 arm (sudden death, septic shock, and respiratory failure) and 2 deaths in the rituximab arm (multiple organ dysfunction syndrome and sepsis) were suspected to be related to study treatment.
During the maintenance phase, the incidence of AEs was 63% in the GP2013 arm and 57% in the rituximab arm. The incidence of serious AEs was 6% and 4%, respectively.
The most frequent AEs (in the GP2013 and rituximab arms, respectively) were infections and infestations (20% and 27%), neutropenia (10% and 6%), cough (9% and 6%), and upper respiratory tract infection (3% and 6%). The most common grade 3/4 AE was neutropenia (7% and 4%).
There were 4 deaths reported during the maintenance phase, 2 in each treatment arm. ![]()
MADRID—The biosimilar GP2013 has demonstrated equivalence to its reference drug rituximab in patients with previously untreated, advanced-stage follicular lymphoma (FL), according to researchers.
Treatment with GP2013 plus cyclophosphamide, vincristine, and prednisone (CVP) produced a similar overall response rate (ORR) as rituximab plus CVP in the phase 3 ASSIST-FL trial.
Survival rates were also similar between the treatment arms, as were adverse events (AEs).
Results from this study were published in The Lancet Haematology and presented at ESMO 2017 Congress (abstract 994O).
The study was funded by Hexal AG, a Sandoz company (part of the Novartis group), which is marketing GP2013 as Rixathon in Europe.
Patients and treatment
The trial included 629 patients with previously untreated, advanced-stage FL. They were randomized to receive 8 cycles of GP2013-CVP (n=314) or rituximab-CVP (n=315). Responders in either arm could receive monotherapy maintenance for up to 2 years.
The mean age was 57.5 in the GP2013 arm and 56.4 in the rituximab arm. Fifty-eight percent and 54% of patients, respectively, were female.
Fifty-seven percent of patients in the GP2013 arm and 56% in the rituximab arm had an ECOG performance status of 0. Forty percent and 39%, respectively, had a status of 1. Two percent and 4%, respectively, had a status of 2. (For the remaining 1% of patients in each arm, data on performance status were missing.)
Patients had an Ann Arbor stage of III—46% in the GP2013 arm and 43% in the GP2013 arm—or IV—54% in the GP2013 arm and 57% in the rituximab arm.
Fifty-six percent of patients in each arm were high-risk according to FLIPI. Thirty-four percent in the GP2013 arm and 33% in the rituximab arm were intermediate-risk. Ten percent and 11%, respectively, were low-risk.
Fourteen percent of patients in the GP2013 arm and 18% in the rituximab arm had bulky disease. Fifteen percent and 13%, respectively, had splenic involvement.
ORR and survival
The patients had a median follow-up of 23.8 months. The primary efficacy endpoint was equivalence in ORR, defined by a 95% confidence interval (CI) with a margin of ± 12% standard deviation.
The primary endpoint was met, as the ORR was 87% in the GP2013 arm and 88% in the rituximab arm, with a difference of –0.40% (95% CI –5.94%, 5.14%).
The complete response rate was 15% in the GP2013 arm and 13% in the rituximab arm. The partial response rates were 72% and 74%, respectively.
The median progression-free survival and overall survival have not been reached. However, the progression-free survival rate was 70% in the GP2013 arm and 76% in the rituximab arm (hazard ratio [HR]=1.31; 95% CI 1.02, 1.69).
The overall survival rate was 93% in the GP2013 arm and 91% in the rituximab arm (HR=0.77; 95% CI 0.49, 1.22).
Safety
During the combination phase, the incidence of AEs was 93% in the GP2013 arm and 91% in the rituximab arm. The incidence of serious AEs was 23% and 20%, respectively.
The most frequent AEs (in the GP2013 and rituximab arms, respectively) were neutropenia (26% and 30%), constipation (22% and 20%), and nausea (16% and 13%). The most common grade 3/4 AE was neutropenia (18% and 21%).
There were 11 deaths reported during the combination phase—4 in the GP2013 arm and 7 in the rituximab arm.
Three deaths in the GP2013 arm (sudden death, septic shock, and respiratory failure) and 2 deaths in the rituximab arm (multiple organ dysfunction syndrome and sepsis) were suspected to be related to study treatment.
During the maintenance phase, the incidence of AEs was 63% in the GP2013 arm and 57% in the rituximab arm. The incidence of serious AEs was 6% and 4%, respectively.
The most frequent AEs (in the GP2013 and rituximab arms, respectively) were infections and infestations (20% and 27%), neutropenia (10% and 6%), cough (9% and 6%), and upper respiratory tract infection (3% and 6%). The most common grade 3/4 AE was neutropenia (7% and 4%).
There were 4 deaths reported during the maintenance phase, 2 in each treatment arm. ![]()
Study: Many cancer patients don’t understand clinical trials
MADRID—Results of a nationwide study suggest many cancer patients in Ireland don’t understand key aspects of clinical trial methodology.
Most of the patients surveyed, which included individuals who had participated in a clinical trial, did not understand the concepts of randomization or equipoise.
“Over half of previous medical trial participants and 73% of those who had never been on a cancer clinical trial did not understand that, in a randomized trial, the treatment given was decided by chance,” said study investigator Catherine Kelly, MB BCh, of Mater Misericordiae University Hospital in Dublin, Ireland.
“We also found that most patients did not understand clinical equipoise—the fact that no one knows which treatment is best. Surprisingly, this was more marked in previous clinical trial participants, 60% of whom believed that their doctor would know which study arm was best.”
Dr Kelly and her colleagues presented these findings at the ESMO 2017 Congress (abstract 1465P_PR).
The researchers surveyed 1090 adult cancer patients treated at 1 of 14 participating oncology centers across Ireland.
The patients’ median age was 60 (range, 50-69), 64.4% were female, and 66% were diagnosed between 2014 and 2016. The most common cancer types were breast (31.4%), colorectal (15.6%), hematologic (12.6%), genitourinary (11.6%), and lung (6.8%).
The patients filled out anonymized questionnaires in which they were asked to evaluate statements about clinical trials. The patients had to determine whether a statement is true or false, or they could indicate that they didn’t know an answer.
A majority of the patients (82.3%) said they understood what a medical or cancer clinical trial is. And 27.8% of patients said they had previously participated in a cancer trial.
However, many patients didn’t know when clinical trials may be an option. Twenty-two percent of patients said it is true that “clinical trials are only used when standard treatments have not worked,” and 26.6% said they didn’t know if this statement is true or false.
Roughly a third (33.5%) of patients said it is true that, in a randomized trial, treatment is decided by chance, but 41.4% of patients said this is false, and 25% said they didn’t know.
More than half of patients (56.5%) said their doctor would know which treatment was superior in a clinical trial, and 23.2% of patients said they didn’t know if their doctor would know.
About 61% of all patients said their doctor would make sure they received the superior treatment in a clinical trial. An even greater percentage—63.6%—of patients who had previously participated in a clinical trial said the same.
“To provide informed consent when participating in a trial, patients need to understand these key concepts, and doctors explaining them well is essential to alleviating any fears that might prevent patients from participating,” Dr Kelly said.
“Doctors have a responsibility to properly inform their patients in this regard because they are the ones patients trust the most. As we analyze the data further, we will be able to offer physicians a more detailed picture of the questions patients need answered and the factors that influence their decision-making according to age group, cancer type, educational background, and other demographics.”
Funding for this research was provided to Cancer Trials Ireland by Amgen, Abbvie, Bayor, and Inveva. ![]()
MADRID—Results of a nationwide study suggest many cancer patients in Ireland don’t understand key aspects of clinical trial methodology.
Most of the patients surveyed, which included individuals who had participated in a clinical trial, did not understand the concepts of randomization or equipoise.
“Over half of previous medical trial participants and 73% of those who had never been on a cancer clinical trial did not understand that, in a randomized trial, the treatment given was decided by chance,” said study investigator Catherine Kelly, MB BCh, of Mater Misericordiae University Hospital in Dublin, Ireland.
“We also found that most patients did not understand clinical equipoise—the fact that no one knows which treatment is best. Surprisingly, this was more marked in previous clinical trial participants, 60% of whom believed that their doctor would know which study arm was best.”
Dr Kelly and her colleagues presented these findings at the ESMO 2017 Congress (abstract 1465P_PR).
The researchers surveyed 1090 adult cancer patients treated at 1 of 14 participating oncology centers across Ireland.
The patients’ median age was 60 (range, 50-69), 64.4% were female, and 66% were diagnosed between 2014 and 2016. The most common cancer types were breast (31.4%), colorectal (15.6%), hematologic (12.6%), genitourinary (11.6%), and lung (6.8%).
The patients filled out anonymized questionnaires in which they were asked to evaluate statements about clinical trials. The patients had to determine whether a statement is true or false, or they could indicate that they didn’t know an answer.
A majority of the patients (82.3%) said they understood what a medical or cancer clinical trial is. And 27.8% of patients said they had previously participated in a cancer trial.
However, many patients didn’t know when clinical trials may be an option. Twenty-two percent of patients said it is true that “clinical trials are only used when standard treatments have not worked,” and 26.6% said they didn’t know if this statement is true or false.
Roughly a third (33.5%) of patients said it is true that, in a randomized trial, treatment is decided by chance, but 41.4% of patients said this is false, and 25% said they didn’t know.
More than half of patients (56.5%) said their doctor would know which treatment was superior in a clinical trial, and 23.2% of patients said they didn’t know if their doctor would know.
About 61% of all patients said their doctor would make sure they received the superior treatment in a clinical trial. An even greater percentage—63.6%—of patients who had previously participated in a clinical trial said the same.
“To provide informed consent when participating in a trial, patients need to understand these key concepts, and doctors explaining them well is essential to alleviating any fears that might prevent patients from participating,” Dr Kelly said.
“Doctors have a responsibility to properly inform their patients in this regard because they are the ones patients trust the most. As we analyze the data further, we will be able to offer physicians a more detailed picture of the questions patients need answered and the factors that influence their decision-making according to age group, cancer type, educational background, and other demographics.”
Funding for this research was provided to Cancer Trials Ireland by Amgen, Abbvie, Bayor, and Inveva. ![]()
MADRID—Results of a nationwide study suggest many cancer patients in Ireland don’t understand key aspects of clinical trial methodology.
Most of the patients surveyed, which included individuals who had participated in a clinical trial, did not understand the concepts of randomization or equipoise.
“Over half of previous medical trial participants and 73% of those who had never been on a cancer clinical trial did not understand that, in a randomized trial, the treatment given was decided by chance,” said study investigator Catherine Kelly, MB BCh, of Mater Misericordiae University Hospital in Dublin, Ireland.
“We also found that most patients did not understand clinical equipoise—the fact that no one knows which treatment is best. Surprisingly, this was more marked in previous clinical trial participants, 60% of whom believed that their doctor would know which study arm was best.”
Dr Kelly and her colleagues presented these findings at the ESMO 2017 Congress (abstract 1465P_PR).
The researchers surveyed 1090 adult cancer patients treated at 1 of 14 participating oncology centers across Ireland.
The patients’ median age was 60 (range, 50-69), 64.4% were female, and 66% were diagnosed between 2014 and 2016. The most common cancer types were breast (31.4%), colorectal (15.6%), hematologic (12.6%), genitourinary (11.6%), and lung (6.8%).
The patients filled out anonymized questionnaires in which they were asked to evaluate statements about clinical trials. The patients had to determine whether a statement is true or false, or they could indicate that they didn’t know an answer.
A majority of the patients (82.3%) said they understood what a medical or cancer clinical trial is. And 27.8% of patients said they had previously participated in a cancer trial.
However, many patients didn’t know when clinical trials may be an option. Twenty-two percent of patients said it is true that “clinical trials are only used when standard treatments have not worked,” and 26.6% said they didn’t know if this statement is true or false.
Roughly a third (33.5%) of patients said it is true that, in a randomized trial, treatment is decided by chance, but 41.4% of patients said this is false, and 25% said they didn’t know.
More than half of patients (56.5%) said their doctor would know which treatment was superior in a clinical trial, and 23.2% of patients said they didn’t know if their doctor would know.
About 61% of all patients said their doctor would make sure they received the superior treatment in a clinical trial. An even greater percentage—63.6%—of patients who had previously participated in a clinical trial said the same.
“To provide informed consent when participating in a trial, patients need to understand these key concepts, and doctors explaining them well is essential to alleviating any fears that might prevent patients from participating,” Dr Kelly said.
“Doctors have a responsibility to properly inform their patients in this regard because they are the ones patients trust the most. As we analyze the data further, we will be able to offer physicians a more detailed picture of the questions patients need answered and the factors that influence their decision-making according to age group, cancer type, educational background, and other demographics.”
Funding for this research was provided to Cancer Trials Ireland by Amgen, Abbvie, Bayor, and Inveva. ![]()
CCSs have higher burden of chronic conditions
Adult survivors of childhood cancer have a greater cumulative burden of chronic health conditions than the general public, according to research published in The Lancet.
The study showed that, by age 50, childhood cancer survivors (CCSs) had experienced, on average, 17.1 chronic health conditions, and matched control subjects had experienced 9.2.
“The cumulative burden of chronic disease revealed in this analysis, along with the complexity and severity of chronic conditions some survivors experience, found childhood cancer survivors to be a vulnerable, medically complex population,” said study author Nickhill Bhakta, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
For this study, Dr Bhakta and his colleagues assessed the lifelong impact of 168 chronic health conditions—such as hepatic, thyroid, ocular, and reproductive disorders—on CCSs and control subjects.
The 3010 evaluable CCSs had survived 10 years or longer from their initial cancer diagnosis and were 18 years or older as of June 30, 2015. The 272 controls had no history of pediatric cancer and were matched to CCSs by age and sex.
At age 50, the cumulative incidence of chronic health conditions (of any grade) was 99.9% in CCSs and 96.0% in controls (P<0.0001). The cumulative incidence of grade 3 to 5 chronic health conditions was 96.0% and 84.9%, respectively (P<0.0001).
The cumulative burden for CCSs was 17.1 chronic health conditions, including 4.7 that were grade 3 to 5. For controls, the cumulative burden was 9.2 chronic health conditions, including 2.3 that were grade 3 to 5 (P<0.0001 for both comparisons).
The researchers said second neoplasms, spinal disorders, and pulmonary disease were major contributors to the excess total cumulative burden observed in CCSs. However, there was “notable heterogeneity” in burden according to the patients’ primary cancer diagnosis.
For instance, growth hormone deficiency was in the top 10th percentile of chronic health conditions for survivors of acute lymphoblastic leukemia but not for controls.
And pulmonary function deficits were in the top 10th percentile for survivors of acute myeloid leukemia and Hodgkin lymphoma but not for controls or survivors of acute lymphoblastic leukemia or non-Hodgkin lymphoma.
“This study found that the average childhood cancer survivor has a cumulative burden of chronic disease that requires a significant time investment by healthcare providers to disentangle and manage—time that community providers are unlikely to have,” Dr Bhakta said.
“The results suggest that childhood cancer survivors may benefit from the integrated, specialized healthcare delivery that is being tried for individuals infected with HIV or those with other complex, chronic health problems.” ![]()
Adult survivors of childhood cancer have a greater cumulative burden of chronic health conditions than the general public, according to research published in The Lancet.
The study showed that, by age 50, childhood cancer survivors (CCSs) had experienced, on average, 17.1 chronic health conditions, and matched control subjects had experienced 9.2.
“The cumulative burden of chronic disease revealed in this analysis, along with the complexity and severity of chronic conditions some survivors experience, found childhood cancer survivors to be a vulnerable, medically complex population,” said study author Nickhill Bhakta, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
For this study, Dr Bhakta and his colleagues assessed the lifelong impact of 168 chronic health conditions—such as hepatic, thyroid, ocular, and reproductive disorders—on CCSs and control subjects.
The 3010 evaluable CCSs had survived 10 years or longer from their initial cancer diagnosis and were 18 years or older as of June 30, 2015. The 272 controls had no history of pediatric cancer and were matched to CCSs by age and sex.
At age 50, the cumulative incidence of chronic health conditions (of any grade) was 99.9% in CCSs and 96.0% in controls (P<0.0001). The cumulative incidence of grade 3 to 5 chronic health conditions was 96.0% and 84.9%, respectively (P<0.0001).
The cumulative burden for CCSs was 17.1 chronic health conditions, including 4.7 that were grade 3 to 5. For controls, the cumulative burden was 9.2 chronic health conditions, including 2.3 that were grade 3 to 5 (P<0.0001 for both comparisons).
The researchers said second neoplasms, spinal disorders, and pulmonary disease were major contributors to the excess total cumulative burden observed in CCSs. However, there was “notable heterogeneity” in burden according to the patients’ primary cancer diagnosis.
For instance, growth hormone deficiency was in the top 10th percentile of chronic health conditions for survivors of acute lymphoblastic leukemia but not for controls.
And pulmonary function deficits were in the top 10th percentile for survivors of acute myeloid leukemia and Hodgkin lymphoma but not for controls or survivors of acute lymphoblastic leukemia or non-Hodgkin lymphoma.
“This study found that the average childhood cancer survivor has a cumulative burden of chronic disease that requires a significant time investment by healthcare providers to disentangle and manage—time that community providers are unlikely to have,” Dr Bhakta said.
“The results suggest that childhood cancer survivors may benefit from the integrated, specialized healthcare delivery that is being tried for individuals infected with HIV or those with other complex, chronic health problems.” ![]()
Adult survivors of childhood cancer have a greater cumulative burden of chronic health conditions than the general public, according to research published in The Lancet.
The study showed that, by age 50, childhood cancer survivors (CCSs) had experienced, on average, 17.1 chronic health conditions, and matched control subjects had experienced 9.2.
“The cumulative burden of chronic disease revealed in this analysis, along with the complexity and severity of chronic conditions some survivors experience, found childhood cancer survivors to be a vulnerable, medically complex population,” said study author Nickhill Bhakta, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
For this study, Dr Bhakta and his colleagues assessed the lifelong impact of 168 chronic health conditions—such as hepatic, thyroid, ocular, and reproductive disorders—on CCSs and control subjects.
The 3010 evaluable CCSs had survived 10 years or longer from their initial cancer diagnosis and were 18 years or older as of June 30, 2015. The 272 controls had no history of pediatric cancer and were matched to CCSs by age and sex.
At age 50, the cumulative incidence of chronic health conditions (of any grade) was 99.9% in CCSs and 96.0% in controls (P<0.0001). The cumulative incidence of grade 3 to 5 chronic health conditions was 96.0% and 84.9%, respectively (P<0.0001).
The cumulative burden for CCSs was 17.1 chronic health conditions, including 4.7 that were grade 3 to 5. For controls, the cumulative burden was 9.2 chronic health conditions, including 2.3 that were grade 3 to 5 (P<0.0001 for both comparisons).
The researchers said second neoplasms, spinal disorders, and pulmonary disease were major contributors to the excess total cumulative burden observed in CCSs. However, there was “notable heterogeneity” in burden according to the patients’ primary cancer diagnosis.
For instance, growth hormone deficiency was in the top 10th percentile of chronic health conditions for survivors of acute lymphoblastic leukemia but not for controls.
And pulmonary function deficits were in the top 10th percentile for survivors of acute myeloid leukemia and Hodgkin lymphoma but not for controls or survivors of acute lymphoblastic leukemia or non-Hodgkin lymphoma.
“This study found that the average childhood cancer survivor has a cumulative burden of chronic disease that requires a significant time investment by healthcare providers to disentangle and manage—time that community providers are unlikely to have,” Dr Bhakta said.
“The results suggest that childhood cancer survivors may benefit from the integrated, specialized healthcare delivery that is being tried for individuals infected with HIV or those with other complex, chronic health problems.” ![]()
FDA places full, partial holds on durvalumab trials
The US Food and Drug Administration (FDA) has placed a partial clinical hold on 5 trials and a full clinical hold on 1 trial of the anti-PD-L1 antibody durvalumab (Imfinzi™).
In these trials, researchers are testing durvalumab in combination with immunomodulatory and chemotherapy agents in patients with multiple myeloma (MM) and lymphomas.
At present, no new patients can be enrolled in any of the 6 trials.
Patients enrolled in the trials on partial clinical hold can remain on treatment if they are receiving clinical benefit.
Patients enrolled in the trial on full clinical hold will discontinue the study treatment.
The FDA’s decision to place these trials on hold is related to risks identified in trials studying another anti-PD-1 agent, pembrolizumab, in MM patients.
Data from the pembrolizumab trials indicate the risks outweigh the benefits when PD-1/PD-L1 treatment is given to MM patients in combination with dexamethasone and pomalidomide or lenalidomide.
In addition, there may be an unfavorable risk-benefit ratio for MM patients receiving PD-1/PD-L1 treatments alone or in other combinations.
With this in mind, the FDA placed the MEDI4736-MM-002 trial on full clinical hold.
MEDI4736-MM-002 is a phase 1b study designed to determine the recommended dose and regimen of durvalumab in combination with lenalidomide, with and without low-dose dexamethasone, in patients with newly diagnosed MM.
The FDA also placed the following trials on partial clinical hold:
- MEDI4736-MM-001: A phase 1b study to determine the recommended dose and regimen of durvalumab either as monotherapy or in combination with pomalidomide, with or without low-dose dexamethasone, in patients with relapsed and refractory MM
- MEDI4736-MM-003: A phase 2 study to determine the safety and efficacy of the combination of durvalumab and daratumumab in patients with relapsed and refractory MM
- MEDI4736-MM-005: A phase 2 study to determine the efficacy of the combination of durvalumab plus daratumumab in patients with relapsed and refractory MM who have progressed while on a current treatment regimen containing daratumumab
- MEDI4736-NHL-001: A phase 1/2 study to assess the safety and tolerability of durvalumab as monotherapy and in combination therapy in patients with lymphomas, including chronic lymphocytic leukemia. The only arm in this trial for which enrollment is suspended is the arm with the durvalumab, lenalidomide, and rituximab combination.
- MEDI4736-DLBCL-001: A phase 2 study to evaluate the safety and clinical activity of durvalumab in combination with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) or with lenalidomide plus R-CHOP in patients with previously untreated, high-risk diffuse large B-cell lymphoma.
The trials that will continue to enroll are:
- MEDI4736-MDS-001: A phase 2 study evaluating the efficacy and safety of subcutaneous azacitidine in combination with durvalumab in previously untreated patients with higher-risk myelodysplastic syndromes or in elderly (≥65 years) acute myeloid leukemia patients not eligible for hematopoietic stem cell transplant
- CC-486-MDS-006: A phase 2 study to evaluate the efficacy and safety of CC-486 alone or in combination with durvalumab in patients with myelodysplastic syndromes who fail to achieve an objective response to treatment with azacitidine for injection or decitabine.
Durvalumab is being developed by Celgene Corporation and MedImmune, the global biologics research and development arm of AstraZeneca.
The use of durvalumab in combination with other agents for the treatment of patients with hematologic malignancies is not approved by the FDA, and the safety and efficacy of those combinations has not been established.
Durvalumab has accelerated approval from the FDA to treat patients with locally advanced or metastatic urothelial carcinoma. ![]()
The US Food and Drug Administration (FDA) has placed a partial clinical hold on 5 trials and a full clinical hold on 1 trial of the anti-PD-L1 antibody durvalumab (Imfinzi™).
In these trials, researchers are testing durvalumab in combination with immunomodulatory and chemotherapy agents in patients with multiple myeloma (MM) and lymphomas.
At present, no new patients can be enrolled in any of the 6 trials.
Patients enrolled in the trials on partial clinical hold can remain on treatment if they are receiving clinical benefit.
Patients enrolled in the trial on full clinical hold will discontinue the study treatment.
The FDA’s decision to place these trials on hold is related to risks identified in trials studying another anti-PD-1 agent, pembrolizumab, in MM patients.
Data from the pembrolizumab trials indicate the risks outweigh the benefits when PD-1/PD-L1 treatment is given to MM patients in combination with dexamethasone and pomalidomide or lenalidomide.
In addition, there may be an unfavorable risk-benefit ratio for MM patients receiving PD-1/PD-L1 treatments alone or in other combinations.
With this in mind, the FDA placed the MEDI4736-MM-002 trial on full clinical hold.
MEDI4736-MM-002 is a phase 1b study designed to determine the recommended dose and regimen of durvalumab in combination with lenalidomide, with and without low-dose dexamethasone, in patients with newly diagnosed MM.
The FDA also placed the following trials on partial clinical hold:
- MEDI4736-MM-001: A phase 1b study to determine the recommended dose and regimen of durvalumab either as monotherapy or in combination with pomalidomide, with or without low-dose dexamethasone, in patients with relapsed and refractory MM
- MEDI4736-MM-003: A phase 2 study to determine the safety and efficacy of the combination of durvalumab and daratumumab in patients with relapsed and refractory MM
- MEDI4736-MM-005: A phase 2 study to determine the efficacy of the combination of durvalumab plus daratumumab in patients with relapsed and refractory MM who have progressed while on a current treatment regimen containing daratumumab
- MEDI4736-NHL-001: A phase 1/2 study to assess the safety and tolerability of durvalumab as monotherapy and in combination therapy in patients with lymphomas, including chronic lymphocytic leukemia. The only arm in this trial for which enrollment is suspended is the arm with the durvalumab, lenalidomide, and rituximab combination.
- MEDI4736-DLBCL-001: A phase 2 study to evaluate the safety and clinical activity of durvalumab in combination with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) or with lenalidomide plus R-CHOP in patients with previously untreated, high-risk diffuse large B-cell lymphoma.
The trials that will continue to enroll are:
- MEDI4736-MDS-001: A phase 2 study evaluating the efficacy and safety of subcutaneous azacitidine in combination with durvalumab in previously untreated patients with higher-risk myelodysplastic syndromes or in elderly (≥65 years) acute myeloid leukemia patients not eligible for hematopoietic stem cell transplant
- CC-486-MDS-006: A phase 2 study to evaluate the efficacy and safety of CC-486 alone or in combination with durvalumab in patients with myelodysplastic syndromes who fail to achieve an objective response to treatment with azacitidine for injection or decitabine.
Durvalumab is being developed by Celgene Corporation and MedImmune, the global biologics research and development arm of AstraZeneca.
The use of durvalumab in combination with other agents for the treatment of patients with hematologic malignancies is not approved by the FDA, and the safety and efficacy of those combinations has not been established.
Durvalumab has accelerated approval from the FDA to treat patients with locally advanced or metastatic urothelial carcinoma. ![]()
The US Food and Drug Administration (FDA) has placed a partial clinical hold on 5 trials and a full clinical hold on 1 trial of the anti-PD-L1 antibody durvalumab (Imfinzi™).
In these trials, researchers are testing durvalumab in combination with immunomodulatory and chemotherapy agents in patients with multiple myeloma (MM) and lymphomas.
At present, no new patients can be enrolled in any of the 6 trials.
Patients enrolled in the trials on partial clinical hold can remain on treatment if they are receiving clinical benefit.
Patients enrolled in the trial on full clinical hold will discontinue the study treatment.
The FDA’s decision to place these trials on hold is related to risks identified in trials studying another anti-PD-1 agent, pembrolizumab, in MM patients.
Data from the pembrolizumab trials indicate the risks outweigh the benefits when PD-1/PD-L1 treatment is given to MM patients in combination with dexamethasone and pomalidomide or lenalidomide.
In addition, there may be an unfavorable risk-benefit ratio for MM patients receiving PD-1/PD-L1 treatments alone or in other combinations.
With this in mind, the FDA placed the MEDI4736-MM-002 trial on full clinical hold.
MEDI4736-MM-002 is a phase 1b study designed to determine the recommended dose and regimen of durvalumab in combination with lenalidomide, with and without low-dose dexamethasone, in patients with newly diagnosed MM.
The FDA also placed the following trials on partial clinical hold:
- MEDI4736-MM-001: A phase 1b study to determine the recommended dose and regimen of durvalumab either as monotherapy or in combination with pomalidomide, with or without low-dose dexamethasone, in patients with relapsed and refractory MM
- MEDI4736-MM-003: A phase 2 study to determine the safety and efficacy of the combination of durvalumab and daratumumab in patients with relapsed and refractory MM
- MEDI4736-MM-005: A phase 2 study to determine the efficacy of the combination of durvalumab plus daratumumab in patients with relapsed and refractory MM who have progressed while on a current treatment regimen containing daratumumab
- MEDI4736-NHL-001: A phase 1/2 study to assess the safety and tolerability of durvalumab as monotherapy and in combination therapy in patients with lymphomas, including chronic lymphocytic leukemia. The only arm in this trial for which enrollment is suspended is the arm with the durvalumab, lenalidomide, and rituximab combination.
- MEDI4736-DLBCL-001: A phase 2 study to evaluate the safety and clinical activity of durvalumab in combination with rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) or with lenalidomide plus R-CHOP in patients with previously untreated, high-risk diffuse large B-cell lymphoma.
The trials that will continue to enroll are:
- MEDI4736-MDS-001: A phase 2 study evaluating the efficacy and safety of subcutaneous azacitidine in combination with durvalumab in previously untreated patients with higher-risk myelodysplastic syndromes or in elderly (≥65 years) acute myeloid leukemia patients not eligible for hematopoietic stem cell transplant
- CC-486-MDS-006: A phase 2 study to evaluate the efficacy and safety of CC-486 alone or in combination with durvalumab in patients with myelodysplastic syndromes who fail to achieve an objective response to treatment with azacitidine for injection or decitabine.
Durvalumab is being developed by Celgene Corporation and MedImmune, the global biologics research and development arm of AstraZeneca.
The use of durvalumab in combination with other agents for the treatment of patients with hematologic malignancies is not approved by the FDA, and the safety and efficacy of those combinations has not been established.
Durvalumab has accelerated approval from the FDA to treat patients with locally advanced or metastatic urothelial carcinoma. ![]()
FDA grants orphan designation to product for CMV
The US Food and Drug Administration (FDA) has granted orphan drug designation to ATA230 for the treatment of cytomegalovirus (CMV) viremia and disease in immunocompromised patients.
ATA230 is an allogeneic, cytotoxic T-lymphocyte (CTL) product targeting antigens expressed by CMV.
The product is under investigation in phase 2 trials of patients with CMV viremia and disease who are refractory or resistant to antiviral treatment.
Atara Biotherapeutics, Inc., the company developing ATA230, said it will evaluate development plans for this therapy with the FDA and other global health authorities after beginning phase 3 studies of another product, ATA129.
The company said it decided to prioritize ATA129, which is being developed to treat patients with Epstein-Barr-virus-associated post-transplant lymphoproliferative disorder.
Phase 2 trial of ATA230
Researchers reported phase 2 results with ATA230 at the 2016 ASH Annual Meeting.
The data encompassed 15 patients with documented CMV mutations conferring resistance to antiviral therapies. The patients had received a median of 3 prior therapies.
Eleven of the 15 patients (73.3%) responded to ATA230, 6 with complete responses and 5 with partial responses.
At 6 months, the overall survival was 72.7% in responders and 25% in non-responders.
Within the 6 months of follow-up, 1 of the 11 responders died of CMV, and 3 of the 4 non-responders died of CMV.
Adverse events occurred in 6 patients. One grade 3 event and 1 grade 4 event were considered possibly related to ATA230.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ATA230 for the treatment of cytomegalovirus (CMV) viremia and disease in immunocompromised patients.
ATA230 is an allogeneic, cytotoxic T-lymphocyte (CTL) product targeting antigens expressed by CMV.
The product is under investigation in phase 2 trials of patients with CMV viremia and disease who are refractory or resistant to antiviral treatment.
Atara Biotherapeutics, Inc., the company developing ATA230, said it will evaluate development plans for this therapy with the FDA and other global health authorities after beginning phase 3 studies of another product, ATA129.
The company said it decided to prioritize ATA129, which is being developed to treat patients with Epstein-Barr-virus-associated post-transplant lymphoproliferative disorder.
Phase 2 trial of ATA230
Researchers reported phase 2 results with ATA230 at the 2016 ASH Annual Meeting.
The data encompassed 15 patients with documented CMV mutations conferring resistance to antiviral therapies. The patients had received a median of 3 prior therapies.
Eleven of the 15 patients (73.3%) responded to ATA230, 6 with complete responses and 5 with partial responses.
At 6 months, the overall survival was 72.7% in responders and 25% in non-responders.
Within the 6 months of follow-up, 1 of the 11 responders died of CMV, and 3 of the 4 non-responders died of CMV.
Adverse events occurred in 6 patients. One grade 3 event and 1 grade 4 event were considered possibly related to ATA230.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ATA230 for the treatment of cytomegalovirus (CMV) viremia and disease in immunocompromised patients.
ATA230 is an allogeneic, cytotoxic T-lymphocyte (CTL) product targeting antigens expressed by CMV.
The product is under investigation in phase 2 trials of patients with CMV viremia and disease who are refractory or resistant to antiviral treatment.
Atara Biotherapeutics, Inc., the company developing ATA230, said it will evaluate development plans for this therapy with the FDA and other global health authorities after beginning phase 3 studies of another product, ATA129.
The company said it decided to prioritize ATA129, which is being developed to treat patients with Epstein-Barr-virus-associated post-transplant lymphoproliferative disorder.
Phase 2 trial of ATA230
Researchers reported phase 2 results with ATA230 at the 2016 ASH Annual Meeting.
The data encompassed 15 patients with documented CMV mutations conferring resistance to antiviral therapies. The patients had received a median of 3 prior therapies.
Eleven of the 15 patients (73.3%) responded to ATA230, 6 with complete responses and 5 with partial responses.
At 6 months, the overall survival was 72.7% in responders and 25% in non-responders.
Within the 6 months of follow-up, 1 of the 11 responders died of CMV, and 3 of the 4 non-responders died of CMV.
Adverse events occurred in 6 patients. One grade 3 event and 1 grade 4 event were considered possibly related to ATA230.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
FDA grants orphan designation to product for GVHD
The US Food and Drug Administration (FDA) has granted orphan drug designation to ApoGraft™ as prophylaxis for acute and chronic graft-versus-host disease (GVHD) in transplant recipients.
ApoGraft is a mobilized peripheral blood cell product collected via apheresis from a matched, related donor. The product is exposed to the apoptotic mediator Fas ligand prior to transplantation.
ApoGraft was designed to eliminate immune responses after transplantation of foreign cells and tissues.
ApoGraft is being developed by Cellect Biotechnology Ltd.
The company is testing ApoGraft as acute GVHD prophylaxis in a phase 1/2 trial.
The trial is currently enrolling patients with hemato-oncology disorders who are eligible for allogeneic, HLA-matched hematopoietic stem cell transplant (HSCT).
The study is expected to have 4 cohorts, each consisting of 3 patients.
The difference between the cohorts is the amount of apoptotic mediator Fas ligand (APO010) to which the graft is exposed during incubation prior to ApoGraft transplantation and HSCT:
- 10 ng/mL APO010 in Cohort 1
- 25 ng/mL APO010 in Cohort 2
- 50 ng/mL APO010 in Cohort 3
- 100 ng/mL APO010 in Cohort 4.
The study is expected to progress from one cohort to the next based on an independent data safety monitoring board review and analysis of safety data.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ApoGraft™ as prophylaxis for acute and chronic graft-versus-host disease (GVHD) in transplant recipients.
ApoGraft is a mobilized peripheral blood cell product collected via apheresis from a matched, related donor. The product is exposed to the apoptotic mediator Fas ligand prior to transplantation.
ApoGraft was designed to eliminate immune responses after transplantation of foreign cells and tissues.
ApoGraft is being developed by Cellect Biotechnology Ltd.
The company is testing ApoGraft as acute GVHD prophylaxis in a phase 1/2 trial.
The trial is currently enrolling patients with hemato-oncology disorders who are eligible for allogeneic, HLA-matched hematopoietic stem cell transplant (HSCT).
The study is expected to have 4 cohorts, each consisting of 3 patients.
The difference between the cohorts is the amount of apoptotic mediator Fas ligand (APO010) to which the graft is exposed during incubation prior to ApoGraft transplantation and HSCT:
- 10 ng/mL APO010 in Cohort 1
- 25 ng/mL APO010 in Cohort 2
- 50 ng/mL APO010 in Cohort 3
- 100 ng/mL APO010 in Cohort 4.
The study is expected to progress from one cohort to the next based on an independent data safety monitoring board review and analysis of safety data.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation to ApoGraft™ as prophylaxis for acute and chronic graft-versus-host disease (GVHD) in transplant recipients.
ApoGraft is a mobilized peripheral blood cell product collected via apheresis from a matched, related donor. The product is exposed to the apoptotic mediator Fas ligand prior to transplantation.
ApoGraft was designed to eliminate immune responses after transplantation of foreign cells and tissues.
ApoGraft is being developed by Cellect Biotechnology Ltd.
The company is testing ApoGraft as acute GVHD prophylaxis in a phase 1/2 trial.
The trial is currently enrolling patients with hemato-oncology disorders who are eligible for allogeneic, HLA-matched hematopoietic stem cell transplant (HSCT).
The study is expected to have 4 cohorts, each consisting of 3 patients.
The difference between the cohorts is the amount of apoptotic mediator Fas ligand (APO010) to which the graft is exposed during incubation prior to ApoGraft transplantation and HSCT:
- 10 ng/mL APO010 in Cohort 1
- 25 ng/mL APO010 in Cohort 2
- 50 ng/mL APO010 in Cohort 3
- 100 ng/mL APO010 in Cohort 4.
The study is expected to progress from one cohort to the next based on an independent data safety monitoring board review and analysis of safety data.
About orphan designation
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
Nanocarriers could treat leukemia, lymphoma and improve HSCT
Researchers say they have created nanoparticles loaded with messenger RNA (mRNA) that can give cells the ability to fight cancers and other diseases.
To use these freeze-dried nanocarriers, the team added water and introduced the resulting mixture to cells.
The nanocarriers were able to target T cells and hematopoietic stem cells (HSCs), delivering mRNA directly to the cells and triggering short-term gene expression.
The T cells were then able to fight leukemia and lymphoma in vitro and in vivo. And the HSCs demonstrated improvements in growth and regenerative potential.
Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington, and his colleagues described this research in Nature Communications.
“We developed a nanocarrier that binds and condenses synthetic mRNA and protects it from degradation,” Dr Stephan said.
The researchers surrounded the nanocarrier with a negatively charged envelope with a targeting ligand attached to the surface so the carrier homes and binds to a particular cell type. When this happens, the cell engulfs the carrier, which can be loaded with different types of manmade mRNA.
The researchers mixed the freeze-dried nanocarriers with water and samples of cells. Within 4 hours, cells started showing signs that editing had taken effect.
The team noted that boosters can be given if needed. And the nanocarriers are made from a dissolving biomaterial, so they are removed from the body like other cell waste.
Testing the carriers
Dr Stephan and his colleagues tested their nanocarriers in 3 ways.
First, the researchers tested nanoparticles carrying a gene-editing tool to T cells that snipped out their natural T-cell receptors and was paired with genes encoding a chimeric antigen receptor (CAR).
The resulting CAR T cells maintained their ability to proliferate and successfully eliminated leukemia cells.
Next, the researchers tested nanocarriers targeted to CAR T cells and containing foxo1 mRNA. This prompted the T cells to develop into a type of memory cell with enhanced antitumor activity.
The team found these CAR T cells induced “substantial disease regression” and prolonged survival in a mouse model of B-cell lymphoma.
Finally, the researchers tested nanocarriers targeted to HSCs. The carriers were equipped with mRNA that “induced key regulators of self-renewal,” accelerating the growth and regenerative potential of the HSCs in vitro.
Future possibilities
Dr Stephan and his colleagues noted that these nanocarriers are built on existing technology and can be used by individuals without knowledge of nanotechnology. Therefore, the team hopes the nanocarriers will be an off-the-shelf way for cell-therapy engineers to develop new approaches to treat diseases.
The researchers believe the nanocarriers could replace electroporation, a multistep cell-manufacturing technique that requires specialized equipment and clean rooms. The team noted that up to 60 times more cells survive the introduction of the nanocarriers than survive electroporation.
“You can imagine taking the nanoparticles, injecting them into a patient, and then you don’t have to culture cells at all anymore,” Dr Stephan said.
He is now looking for commercial partners to move the technology toward additional applications and into clinical trials.
Researchers say they have created nanoparticles loaded with messenger RNA (mRNA) that can give cells the ability to fight cancers and other diseases.
To use these freeze-dried nanocarriers, the team added water and introduced the resulting mixture to cells.
The nanocarriers were able to target T cells and hematopoietic stem cells (HSCs), delivering mRNA directly to the cells and triggering short-term gene expression.
The T cells were then able to fight leukemia and lymphoma in vitro and in vivo. And the HSCs demonstrated improvements in growth and regenerative potential.
Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington, and his colleagues described this research in Nature Communications.
“We developed a nanocarrier that binds and condenses synthetic mRNA and protects it from degradation,” Dr Stephan said.
The researchers surrounded the nanocarrier with a negatively charged envelope with a targeting ligand attached to the surface so the carrier homes and binds to a particular cell type. When this happens, the cell engulfs the carrier, which can be loaded with different types of manmade mRNA.
The researchers mixed the freeze-dried nanocarriers with water and samples of cells. Within 4 hours, cells started showing signs that editing had taken effect.
The team noted that boosters can be given if needed. And the nanocarriers are made from a dissolving biomaterial, so they are removed from the body like other cell waste.
Testing the carriers
Dr Stephan and his colleagues tested their nanocarriers in 3 ways.
First, the researchers tested nanoparticles carrying a gene-editing tool to T cells that snipped out their natural T-cell receptors and was paired with genes encoding a chimeric antigen receptor (CAR).
The resulting CAR T cells maintained their ability to proliferate and successfully eliminated leukemia cells.
Next, the researchers tested nanocarriers targeted to CAR T cells and containing foxo1 mRNA. This prompted the T cells to develop into a type of memory cell with enhanced antitumor activity.
The team found these CAR T cells induced “substantial disease regression” and prolonged survival in a mouse model of B-cell lymphoma.
Finally, the researchers tested nanocarriers targeted to HSCs. The carriers were equipped with mRNA that “induced key regulators of self-renewal,” accelerating the growth and regenerative potential of the HSCs in vitro.
Future possibilities
Dr Stephan and his colleagues noted that these nanocarriers are built on existing technology and can be used by individuals without knowledge of nanotechnology. Therefore, the team hopes the nanocarriers will be an off-the-shelf way for cell-therapy engineers to develop new approaches to treat diseases.
The researchers believe the nanocarriers could replace electroporation, a multistep cell-manufacturing technique that requires specialized equipment and clean rooms. The team noted that up to 60 times more cells survive the introduction of the nanocarriers than survive electroporation.
“You can imagine taking the nanoparticles, injecting them into a patient, and then you don’t have to culture cells at all anymore,” Dr Stephan said.
He is now looking for commercial partners to move the technology toward additional applications and into clinical trials.
Researchers say they have created nanoparticles loaded with messenger RNA (mRNA) that can give cells the ability to fight cancers and other diseases.
To use these freeze-dried nanocarriers, the team added water and introduced the resulting mixture to cells.
The nanocarriers were able to target T cells and hematopoietic stem cells (HSCs), delivering mRNA directly to the cells and triggering short-term gene expression.
The T cells were then able to fight leukemia and lymphoma in vitro and in vivo. And the HSCs demonstrated improvements in growth and regenerative potential.
Matthias Stephan, MD, PhD, of Fred Hutchinson Cancer Research Center in Seattle, Washington, and his colleagues described this research in Nature Communications.
“We developed a nanocarrier that binds and condenses synthetic mRNA and protects it from degradation,” Dr Stephan said.
The researchers surrounded the nanocarrier with a negatively charged envelope with a targeting ligand attached to the surface so the carrier homes and binds to a particular cell type. When this happens, the cell engulfs the carrier, which can be loaded with different types of manmade mRNA.
The researchers mixed the freeze-dried nanocarriers with water and samples of cells. Within 4 hours, cells started showing signs that editing had taken effect.
The team noted that boosters can be given if needed. And the nanocarriers are made from a dissolving biomaterial, so they are removed from the body like other cell waste.
Testing the carriers
Dr Stephan and his colleagues tested their nanocarriers in 3 ways.
First, the researchers tested nanoparticles carrying a gene-editing tool to T cells that snipped out their natural T-cell receptors and was paired with genes encoding a chimeric antigen receptor (CAR).
The resulting CAR T cells maintained their ability to proliferate and successfully eliminated leukemia cells.
Next, the researchers tested nanocarriers targeted to CAR T cells and containing foxo1 mRNA. This prompted the T cells to develop into a type of memory cell with enhanced antitumor activity.
The team found these CAR T cells induced “substantial disease regression” and prolonged survival in a mouse model of B-cell lymphoma.
Finally, the researchers tested nanocarriers targeted to HSCs. The carriers were equipped with mRNA that “induced key regulators of self-renewal,” accelerating the growth and regenerative potential of the HSCs in vitro.
Future possibilities
Dr Stephan and his colleagues noted that these nanocarriers are built on existing technology and can be used by individuals without knowledge of nanotechnology. Therefore, the team hopes the nanocarriers will be an off-the-shelf way for cell-therapy engineers to develop new approaches to treat diseases.
The researchers believe the nanocarriers could replace electroporation, a multistep cell-manufacturing technique that requires specialized equipment and clean rooms. The team noted that up to 60 times more cells survive the introduction of the nanocarriers than survive electroporation.
“You can imagine taking the nanoparticles, injecting them into a patient, and then you don’t have to culture cells at all anymore,” Dr Stephan said.
He is now looking for commercial partners to move the technology toward additional applications and into clinical trials.