User login
Multiple myeloma rates rising fastest in East Asia
The global incidence of multiple myeloma rose by 126% from 1990 to 2016, with the largest regional increases occurring in East Asia and tropical Latin America, according to data from the Global Burden of Disease 2016 study.
East Asia (China, North Korea, and Taiwan) saw incident cases of multiple myeloma jump by 262% – up to 1.0 per 100,000 population – from 1990 to 2016, which was the largest increase among any of the 21 global regions; tropical Latin America’s 256% rise took its age-standardized incidence rate to 1.8 per 100,000. Worldwide, incidence of multiple myeloma was 2.1 cases per 100,000 in 2016, Andrew J. Cowan, MD, and his associates reported in JAMA Oncology.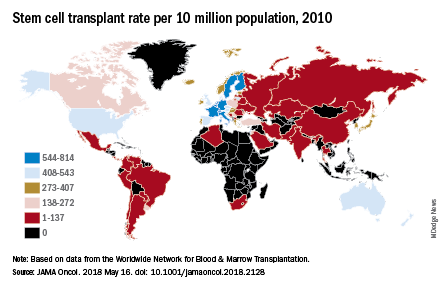
They also looked at treatment availability, with data on stem cell transplants for 2010 coming from the Worldwide Network for Blood & Marrow Transplantation (Lancet Haematol. 2015 Mar;2[3]:e91-100). The countries with the highest rates for all indications that year were Israel (814 per 10 million population), Italy (671), Germany (665), Sweden (625), and the Netherlands (614).
“Some regions of the world lack access to stem cell transplantation entirely, particularly sub-Saharan Africa (with the exception of South Africa),” wrote Dr. Cowan of the University of Washington, Seattle, and his associates.
The approval status of lenalidomide (Revlimid) and bortezomib (Velcade) in 2016 was used as a surrogate for availability of drug treatment: Lenalidomide had been approved in 73 countries out of 195 countries and territories and bortezomib in 103 countries. “On a global level, there are marked discrepancies in the availability of effective therapies. In addition to ensuring universal access to health care … it is imperative to at least ensure access to highly effective medications,” they wrote.
Dr. Cowan reported that he has received research funding from Janssen and AbbVie.
SOURCE: Cowan AJ et al. JAMA Oncol. 2018 May 16. doi: 10.1001/jamaoncol.2018.2128.
The global incidence of multiple myeloma rose by 126% from 1990 to 2016, with the largest regional increases occurring in East Asia and tropical Latin America, according to data from the Global Burden of Disease 2016 study.
East Asia (China, North Korea, and Taiwan) saw incident cases of multiple myeloma jump by 262% – up to 1.0 per 100,000 population – from 1990 to 2016, which was the largest increase among any of the 21 global regions; tropical Latin America’s 256% rise took its age-standardized incidence rate to 1.8 per 100,000. Worldwide, incidence of multiple myeloma was 2.1 cases per 100,000 in 2016, Andrew J. Cowan, MD, and his associates reported in JAMA Oncology.
They also looked at treatment availability, with data on stem cell transplants for 2010 coming from the Worldwide Network for Blood & Marrow Transplantation (Lancet Haematol. 2015 Mar;2[3]:e91-100). The countries with the highest rates for all indications that year were Israel (814 per 10 million population), Italy (671), Germany (665), Sweden (625), and the Netherlands (614).
“Some regions of the world lack access to stem cell transplantation entirely, particularly sub-Saharan Africa (with the exception of South Africa),” wrote Dr. Cowan of the University of Washington, Seattle, and his associates.
The approval status of lenalidomide (Revlimid) and bortezomib (Velcade) in 2016 was used as a surrogate for availability of drug treatment: Lenalidomide had been approved in 73 countries out of 195 countries and territories and bortezomib in 103 countries. “On a global level, there are marked discrepancies in the availability of effective therapies. In addition to ensuring universal access to health care … it is imperative to at least ensure access to highly effective medications,” they wrote.
Dr. Cowan reported that he has received research funding from Janssen and AbbVie.
SOURCE: Cowan AJ et al. JAMA Oncol. 2018 May 16. doi: 10.1001/jamaoncol.2018.2128.
The global incidence of multiple myeloma rose by 126% from 1990 to 2016, with the largest regional increases occurring in East Asia and tropical Latin America, according to data from the Global Burden of Disease 2016 study.
East Asia (China, North Korea, and Taiwan) saw incident cases of multiple myeloma jump by 262% – up to 1.0 per 100,000 population – from 1990 to 2016, which was the largest increase among any of the 21 global regions; tropical Latin America’s 256% rise took its age-standardized incidence rate to 1.8 per 100,000. Worldwide, incidence of multiple myeloma was 2.1 cases per 100,000 in 2016, Andrew J. Cowan, MD, and his associates reported in JAMA Oncology.
They also looked at treatment availability, with data on stem cell transplants for 2010 coming from the Worldwide Network for Blood & Marrow Transplantation (Lancet Haematol. 2015 Mar;2[3]:e91-100). The countries with the highest rates for all indications that year were Israel (814 per 10 million population), Italy (671), Germany (665), Sweden (625), and the Netherlands (614).
“Some regions of the world lack access to stem cell transplantation entirely, particularly sub-Saharan Africa (with the exception of South Africa),” wrote Dr. Cowan of the University of Washington, Seattle, and his associates.
The approval status of lenalidomide (Revlimid) and bortezomib (Velcade) in 2016 was used as a surrogate for availability of drug treatment: Lenalidomide had been approved in 73 countries out of 195 countries and territories and bortezomib in 103 countries. “On a global level, there are marked discrepancies in the availability of effective therapies. In addition to ensuring universal access to health care … it is imperative to at least ensure access to highly effective medications,” they wrote.
Dr. Cowan reported that he has received research funding from Janssen and AbbVie.
SOURCE: Cowan AJ et al. JAMA Oncol. 2018 May 16. doi: 10.1001/jamaoncol.2018.2128.
FROM JAMA ONCOLOGY
Regimen can improve DFS in newly diagnosed T-ALL
The addition of nelarabine can improve treatment outcomes for certain patients with T-cell acute lymphoblastic leukemia (T-ALL), according to a phase 3 trial.
Patients with newly diagnosed, intermediate- or high-risk T-ALL had a significant improvement in 4-year disease-free survival (DFS) if they received nelarabine in addition to chemotherapy and cranial irradiation.
The DFS benefit with nelarabine was significant for patients who received high-dose methotrexate but not for those who received escalating-dose methotrexate.
This study also included patients with T-cell lymphoblastic lymphoma (T-LL), and they did not experience an improvement in DFS with the addition of nelarabine.
Kimberly Dunsmore, MD, of Virginia Tech Carilion School of Medicine in Roanoke, presented these results in a press briefing in advance of the 2018 ASCO Annual Meeting. Additional results are scheduled to be presented at the meeting as abstract 10500.
This research was supported by the National Cancer Institute/National Institutes of Health and St. Baldrick’s Foundation. The researchers’ disclosures are listed with the abstract.
Patients and treatment
The trial enrolled 1895 patients, ages 1 to 30, who were newly diagnosed with T-ALL (94%) or T-LL (6%).
Patients received standard 4-drug induction chemotherapy, and 1307 of these patients were then randomized to 1 of 4 treatment arms.
Regardless of which arm they were randomized to, patients received an 11-drug chemotherapy regimen—the augmented Berlin-Frankfurt-Munster regimen. Intermediate- and high-risk patients in all 4 arms also received cranial irradiation.
In the first treatment arm, T-LL (n=58) and T-ALL (n=372) patients received escalating-dose methotrexate without leucovorin rescue and pegaspargase (C-MTX).
In the second treatment arm, patients with intermediate- and high-risk T-ALL (n=147) and T-LL (n=60) received C-MTX plus nelarabine (six 5-day courses at 650 mg/m2/day).
In the third arm, T-ALL patients (n=451) received high-dose methotrexate with leucovorin rescue (HD-MTX). T-LL patients were not eligible for this arm or the fourth treatment arm.
In the fourth arm, intermediate- and high-risk T-ALL patients (n=219) received HD-MTX and nelarabine (same schedule as above). This included 43 T-ALL patients who had induction failure and were assigned to this arm non-randomly.
Results
For T-ALL patients, the 4-year disease-free survival (DFS) rate was 84%, and the 4-year overall survival rate was 90%.
There was a significant improvement in DFS for T-ALL patients who received nelarabine compared to those who did not—89% and 83%, respectively (P=0.0332).
“Historically, about 80% of people [with T-ALL] live at least 4 years after being treated for their disease, but we felt we could and must do better,” Dr Dunsmore said. “Our trial shows that we could further increase survival rates by about 10%, which is very encouraging.”
Dr Dunsmore also noted that patients who received nelarabine had fewer central nervous system relapses.
Among T-ALL patients who received C-MTX, there was no significant difference in DFS for those who received nelarabine and those who did not—92% and 90%, respectively (P=0.3825).
However, for patients who received HD-MTX, the difference in DFS was significant. The DFS rate was 86% in patients who received nelarabine and 78% in those who did not (P=0.024).
For the T-ALL patients who failed induction and were assigned to HD-MTX and nelarabine, the 4-year DFS rate was 55%.
Patients with T-LL did not benefit from the addition of nelarabine. The 4-year DFS rate was 85% in the nelarabine recipients and 89% in non-recipients (P=0.2788).
There were no significant differences in overall toxicity or peripheral neurotoxicity between the treatment arms.
Dr Dunsmore said the next step with this research will be to examine the implications and potential benefits of using nelarabine in treatment protocols that do not include cranial radiation.
The addition of nelarabine can improve treatment outcomes for certain patients with T-cell acute lymphoblastic leukemia (T-ALL), according to a phase 3 trial.
Patients with newly diagnosed, intermediate- or high-risk T-ALL had a significant improvement in 4-year disease-free survival (DFS) if they received nelarabine in addition to chemotherapy and cranial irradiation.
The DFS benefit with nelarabine was significant for patients who received high-dose methotrexate but not for those who received escalating-dose methotrexate.
This study also included patients with T-cell lymphoblastic lymphoma (T-LL), and they did not experience an improvement in DFS with the addition of nelarabine.
Kimberly Dunsmore, MD, of Virginia Tech Carilion School of Medicine in Roanoke, presented these results in a press briefing in advance of the 2018 ASCO Annual Meeting. Additional results are scheduled to be presented at the meeting as abstract 10500.
This research was supported by the National Cancer Institute/National Institutes of Health and St. Baldrick’s Foundation. The researchers’ disclosures are listed with the abstract.
Patients and treatment
The trial enrolled 1895 patients, ages 1 to 30, who were newly diagnosed with T-ALL (94%) or T-LL (6%).
Patients received standard 4-drug induction chemotherapy, and 1307 of these patients were then randomized to 1 of 4 treatment arms.
Regardless of which arm they were randomized to, patients received an 11-drug chemotherapy regimen—the augmented Berlin-Frankfurt-Munster regimen. Intermediate- and high-risk patients in all 4 arms also received cranial irradiation.
In the first treatment arm, T-LL (n=58) and T-ALL (n=372) patients received escalating-dose methotrexate without leucovorin rescue and pegaspargase (C-MTX).
In the second treatment arm, patients with intermediate- and high-risk T-ALL (n=147) and T-LL (n=60) received C-MTX plus nelarabine (six 5-day courses at 650 mg/m2/day).
In the third arm, T-ALL patients (n=451) received high-dose methotrexate with leucovorin rescue (HD-MTX). T-LL patients were not eligible for this arm or the fourth treatment arm.
In the fourth arm, intermediate- and high-risk T-ALL patients (n=219) received HD-MTX and nelarabine (same schedule as above). This included 43 T-ALL patients who had induction failure and were assigned to this arm non-randomly.
Results
For T-ALL patients, the 4-year disease-free survival (DFS) rate was 84%, and the 4-year overall survival rate was 90%.
There was a significant improvement in DFS for T-ALL patients who received nelarabine compared to those who did not—89% and 83%, respectively (P=0.0332).
“Historically, about 80% of people [with T-ALL] live at least 4 years after being treated for their disease, but we felt we could and must do better,” Dr Dunsmore said. “Our trial shows that we could further increase survival rates by about 10%, which is very encouraging.”
Dr Dunsmore also noted that patients who received nelarabine had fewer central nervous system relapses.
Among T-ALL patients who received C-MTX, there was no significant difference in DFS for those who received nelarabine and those who did not—92% and 90%, respectively (P=0.3825).
However, for patients who received HD-MTX, the difference in DFS was significant. The DFS rate was 86% in patients who received nelarabine and 78% in those who did not (P=0.024).
For the T-ALL patients who failed induction and were assigned to HD-MTX and nelarabine, the 4-year DFS rate was 55%.
Patients with T-LL did not benefit from the addition of nelarabine. The 4-year DFS rate was 85% in the nelarabine recipients and 89% in non-recipients (P=0.2788).
There were no significant differences in overall toxicity or peripheral neurotoxicity between the treatment arms.
Dr Dunsmore said the next step with this research will be to examine the implications and potential benefits of using nelarabine in treatment protocols that do not include cranial radiation.
The addition of nelarabine can improve treatment outcomes for certain patients with T-cell acute lymphoblastic leukemia (T-ALL), according to a phase 3 trial.
Patients with newly diagnosed, intermediate- or high-risk T-ALL had a significant improvement in 4-year disease-free survival (DFS) if they received nelarabine in addition to chemotherapy and cranial irradiation.
The DFS benefit with nelarabine was significant for patients who received high-dose methotrexate but not for those who received escalating-dose methotrexate.
This study also included patients with T-cell lymphoblastic lymphoma (T-LL), and they did not experience an improvement in DFS with the addition of nelarabine.
Kimberly Dunsmore, MD, of Virginia Tech Carilion School of Medicine in Roanoke, presented these results in a press briefing in advance of the 2018 ASCO Annual Meeting. Additional results are scheduled to be presented at the meeting as abstract 10500.
This research was supported by the National Cancer Institute/National Institutes of Health and St. Baldrick’s Foundation. The researchers’ disclosures are listed with the abstract.
Patients and treatment
The trial enrolled 1895 patients, ages 1 to 30, who were newly diagnosed with T-ALL (94%) or T-LL (6%).
Patients received standard 4-drug induction chemotherapy, and 1307 of these patients were then randomized to 1 of 4 treatment arms.
Regardless of which arm they were randomized to, patients received an 11-drug chemotherapy regimen—the augmented Berlin-Frankfurt-Munster regimen. Intermediate- and high-risk patients in all 4 arms also received cranial irradiation.
In the first treatment arm, T-LL (n=58) and T-ALL (n=372) patients received escalating-dose methotrexate without leucovorin rescue and pegaspargase (C-MTX).
In the second treatment arm, patients with intermediate- and high-risk T-ALL (n=147) and T-LL (n=60) received C-MTX plus nelarabine (six 5-day courses at 650 mg/m2/day).
In the third arm, T-ALL patients (n=451) received high-dose methotrexate with leucovorin rescue (HD-MTX). T-LL patients were not eligible for this arm or the fourth treatment arm.
In the fourth arm, intermediate- and high-risk T-ALL patients (n=219) received HD-MTX and nelarabine (same schedule as above). This included 43 T-ALL patients who had induction failure and were assigned to this arm non-randomly.
Results
For T-ALL patients, the 4-year disease-free survival (DFS) rate was 84%, and the 4-year overall survival rate was 90%.
There was a significant improvement in DFS for T-ALL patients who received nelarabine compared to those who did not—89% and 83%, respectively (P=0.0332).
“Historically, about 80% of people [with T-ALL] live at least 4 years after being treated for their disease, but we felt we could and must do better,” Dr Dunsmore said. “Our trial shows that we could further increase survival rates by about 10%, which is very encouraging.”
Dr Dunsmore also noted that patients who received nelarabine had fewer central nervous system relapses.
Among T-ALL patients who received C-MTX, there was no significant difference in DFS for those who received nelarabine and those who did not—92% and 90%, respectively (P=0.3825).
However, for patients who received HD-MTX, the difference in DFS was significant. The DFS rate was 86% in patients who received nelarabine and 78% in those who did not (P=0.024).
For the T-ALL patients who failed induction and were assigned to HD-MTX and nelarabine, the 4-year DFS rate was 55%.
Patients with T-LL did not benefit from the addition of nelarabine. The 4-year DFS rate was 85% in the nelarabine recipients and 89% in non-recipients (P=0.2788).
There were no significant differences in overall toxicity or peripheral neurotoxicity between the treatment arms.
Dr Dunsmore said the next step with this research will be to examine the implications and potential benefits of using nelarabine in treatment protocols that do not include cranial radiation.
Adding bortezomib does not improve MCL outcomes
Bortezomib added to an alternating chemoimmunotherapy regimen did not improve time to treatment failure in patients with newly diagnosed mantle cell lymphoma (MCL), results of a phase 2 study have suggested.
Response rates and time to treatment failure were similar to what has been seen historically without the addition of bortezomib, according to study investigator Jorge E. Romaguera, MD, of the University of Texas MD Anderson Cancer Center, Houston, and his colleagues.
The phase 2 study included 95 patients with newly diagnosed MCL treated with alternating cycles of bortezomib added to rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (BzR-hyperCVAD) and bortezomib added to rituximab plus high-dose methotrexate and high-dose cytarabine (BzR-MA).
Of 87 patients evaluable for response, alternating BzR-hyperCVAD/BzR-MA resulted in an overall response rate of 100% and a complete response rate of 82%, Dr. Romaguera and his colleagues reported in the journal Cancer. At a median follow-up of 44 months, median time to treatment failure was 55 months, and median overall survival had not yet been reached, according to the report.
Dr. Romaguera and his coauthors compared these results with those from a previous study of alternating R-hyperCVAD/R-MA, in which the median time to treatment failure was 56.4 months. “This suggests that the addition of bortezomib does not improve the outcome,” they wrote in the current report.
Although more follow-up is needed, the landscape of MCL treatment is changing quickly, they added. In particular, lenalidomide and ibrutinib, already approved for relapsed/refractory MCL, are now being evaluated as part of first-line MCL regimens. “These drugs will offer strategies of either consolidation or maintenance after induction and will hopefully help continue to improve the duration of the initial response and the overall outcome,” the researchers wrote.
In the current phase 2 study, the fact that 100% of patients achieved complete response suggested that relapses come from minimal residual disease, which “has clearly become a clinical factor for the outcomes of patients with MCL and will likely become the next endpoint,” they wrote.
The researchers reported having no financial disclosures related to the study, which was supported by Takeda Oncology.
SOURCE: Romaguera JE et al. Cancer. 2018 May 3. doi: 10.1002/cncr.31361.
Bortezomib added to an alternating chemoimmunotherapy regimen did not improve time to treatment failure in patients with newly diagnosed mantle cell lymphoma (MCL), results of a phase 2 study have suggested.
Response rates and time to treatment failure were similar to what has been seen historically without the addition of bortezomib, according to study investigator Jorge E. Romaguera, MD, of the University of Texas MD Anderson Cancer Center, Houston, and his colleagues.
The phase 2 study included 95 patients with newly diagnosed MCL treated with alternating cycles of bortezomib added to rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (BzR-hyperCVAD) and bortezomib added to rituximab plus high-dose methotrexate and high-dose cytarabine (BzR-MA).
Of 87 patients evaluable for response, alternating BzR-hyperCVAD/BzR-MA resulted in an overall response rate of 100% and a complete response rate of 82%, Dr. Romaguera and his colleagues reported in the journal Cancer. At a median follow-up of 44 months, median time to treatment failure was 55 months, and median overall survival had not yet been reached, according to the report.
Dr. Romaguera and his coauthors compared these results with those from a previous study of alternating R-hyperCVAD/R-MA, in which the median time to treatment failure was 56.4 months. “This suggests that the addition of bortezomib does not improve the outcome,” they wrote in the current report.
Although more follow-up is needed, the landscape of MCL treatment is changing quickly, they added. In particular, lenalidomide and ibrutinib, already approved for relapsed/refractory MCL, are now being evaluated as part of first-line MCL regimens. “These drugs will offer strategies of either consolidation or maintenance after induction and will hopefully help continue to improve the duration of the initial response and the overall outcome,” the researchers wrote.
In the current phase 2 study, the fact that 100% of patients achieved complete response suggested that relapses come from minimal residual disease, which “has clearly become a clinical factor for the outcomes of patients with MCL and will likely become the next endpoint,” they wrote.
The researchers reported having no financial disclosures related to the study, which was supported by Takeda Oncology.
SOURCE: Romaguera JE et al. Cancer. 2018 May 3. doi: 10.1002/cncr.31361.
Bortezomib added to an alternating chemoimmunotherapy regimen did not improve time to treatment failure in patients with newly diagnosed mantle cell lymphoma (MCL), results of a phase 2 study have suggested.
Response rates and time to treatment failure were similar to what has been seen historically without the addition of bortezomib, according to study investigator Jorge E. Romaguera, MD, of the University of Texas MD Anderson Cancer Center, Houston, and his colleagues.
The phase 2 study included 95 patients with newly diagnosed MCL treated with alternating cycles of bortezomib added to rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (BzR-hyperCVAD) and bortezomib added to rituximab plus high-dose methotrexate and high-dose cytarabine (BzR-MA).
Of 87 patients evaluable for response, alternating BzR-hyperCVAD/BzR-MA resulted in an overall response rate of 100% and a complete response rate of 82%, Dr. Romaguera and his colleagues reported in the journal Cancer. At a median follow-up of 44 months, median time to treatment failure was 55 months, and median overall survival had not yet been reached, according to the report.
Dr. Romaguera and his coauthors compared these results with those from a previous study of alternating R-hyperCVAD/R-MA, in which the median time to treatment failure was 56.4 months. “This suggests that the addition of bortezomib does not improve the outcome,” they wrote in the current report.
Although more follow-up is needed, the landscape of MCL treatment is changing quickly, they added. In particular, lenalidomide and ibrutinib, already approved for relapsed/refractory MCL, are now being evaluated as part of first-line MCL regimens. “These drugs will offer strategies of either consolidation or maintenance after induction and will hopefully help continue to improve the duration of the initial response and the overall outcome,” the researchers wrote.
In the current phase 2 study, the fact that 100% of patients achieved complete response suggested that relapses come from minimal residual disease, which “has clearly become a clinical factor for the outcomes of patients with MCL and will likely become the next endpoint,” they wrote.
The researchers reported having no financial disclosures related to the study, which was supported by Takeda Oncology.
SOURCE: Romaguera JE et al. Cancer. 2018 May 3. doi: 10.1002/cncr.31361.
FROM CANCER
Key clinical point:
Major finding: Rates of overall and complete response were 100% and 82%, respectively, while time to treatment failure was 55 months.
Study details: A phase 2 trial that included 95 patients treated with alternating cycles of bortezomib added to rituximab plus hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (BzR-hyperCVAD) and bortezomib added to rituximab plus high-dose methotrexate and high-dose cytarabine (BzR-MA).
Disclosures: The study was supported by Takeda Oncology. The researchers reported having no financial disclosures related to the study.
Source: Romaguera JE et al. Cancer. 2018 May 3. doi: 10.1002/cncr.31361
Umbralisib has ‘distinct’ safety profile
Phase 1 trial results suggest umbralisib, a PI3Kδ/CK1ε inhibitor, can be safe and active in patients with relapsed or refractory B-cell malignancies.
Researchers said the safety profile of umbralisib “was distinct from that of other PI3Kδ inhibitors,” as it produced few immune-mediated adverse events (AEs).
Umbralisib also produced an objective response rate of 37% in the entire study cohort, 80% in patients with chronic lymphocytic leukemia (CLL), 53% in patients with follicular lymphoma (FL), and 31% in patients with diffuse large B-cell lymphoma (DLBCL).
These results were published in The Lancet Oncology. The study was sponsored by TG Therapeutics, Inc.
The trial enrolled 90 patients between January 17, 2013, and January 14, 2016.
There were 24 patients with CLL, 22 with FL, 16 with DLBCL, 11 with Hodgkin lymphoma, 6 with mantle cell lymphoma, 5 with marginal zone lymphoma, 3 with Waldenstrom’s macroglobulinemia, 2 with T-cell lymphoma, and 1 with hairy cell leukemia.
The median number of prior therapies was 3 (range, 2-5), and 49% of patients were refractory to previous therapy.
Treatment
Patients took umbralisib once daily in 28-day cycles until disease progression, unacceptable toxicity, or withdrawal of consent.
Initially, patients took the drug in a fasting state at doses of 50 mg, 100 mg, 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In April 2014, the researchers did a second dose-escalation with a micronized formulation of umbralisib, taken with food, at doses of 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In August, 2014, all patients who were still on the study transitioned to the 800 mg dose of the micronized formulation. This was the recommended phase 2 dose.
At the data cutoff in November 2016, 44 patients (49%) had received umbralisib for more than 6 cycles, and 23 (26%) had received the drug for more than 12 cycles. Thirteen patients (14%) were still taking umbralisib at the end of the study.
Most patients who stopped treatment did so because of disease progression (n=50, 56%) or AEs (n=9, 10%).
“We are pleased to have treated the first patient ever with umbralisib over 5 years ago and believe it has an important place in the treatment landscape for patients with hematologic malignancies,” said study author Howard A. Burris, MD, of the Sarah Cannon Research Institute in Nashville, Tennessee.
“Several patients from this phase 1 study are still on study today, approaching 5 years of continuous daily therapy, speaking to both the safety and efficacy profile of this unique agent.”
Safety
Dose-limiting toxicities (DLTs) occurred in 4 patients. One DLT was grade 3 maculopapular rash in a patient receiving the 800 mg dose of the initial formulation.
Another DLT was grade 3 hypokalemia in a patient receiving 1800 mg of the initial formulation. A third DLT was grade 3 fatigue, which occurred in 2 patients receiving 1800 mg of the micronized formulation.
Because of these toxicities, the maximum tolerated dose was 1200 mg of the micronized formulation.
The most common treatment-emergent AEs were diarrhea (43%), nausea (42%), and fatigue (31%). The most common grade 3/4 AEs were neutropenia (13%), anemia (9%), and thrombocytopenia (7%).
Serious AEs considered at least possibly related to umbralisib were pneumonia (3%), lung infection (1%), febrile neutropenia (1%), and colitis (2%).
Treatment discontinuation due to AEs considered at least possibly related to umbralisib occurred in 6 patients (7%). Two patients had grade 3 colitis, 2 had increased ALT/AST (grade 1 and grade 4), 1 had grade 2 diarrhea, and 1 had grade 3 fatigue.
There were no treatment-related deaths.
The researchers said the safety profile of umbralisib was distinct from that of other PI3Kδ inhibitors, as patients in this trial had fewer occurrences of autoimmune-like toxicities, such as colitis.
“Preclinically, umbralisib has a very unique profile, selectively inhibiting both PI3Kδ and CK1ε,” said study author Owen O’Connor, MD, PhD, of Columbia Presbyterian Medical Center in New York, New York.
“The clinical results in this paper support our thesis that the differentiated preclinical profile explains the differences seen in the clinic between umbralisib and the other PI3Kδ inhibitors.”
Response
The objective response rate was 37%, with 33 patients achieving a response and 3 patients having a complete response (CR).
Sixteen CLL patients responded (80%), all with partial responses (PRs). Four DLBCL patients responded (31%), all with PRs. And 9 FL patients responded (53%), 2 with CRs.
The remaining CR occurred in a Hodgkin lymphoma patient, and this was the only response in this patient group.
One patient with marginal zone lymphoma had a PR, as did 1 patient with mantle cell lymphoma. All other patients had stable disease or progressed.
The mean duration of response was 13.4 months in the CLL patients, 6.4 months in the DLBCL patients, and 9.3 months in the FL patients.
Phase 1 trial results suggest umbralisib, a PI3Kδ/CK1ε inhibitor, can be safe and active in patients with relapsed or refractory B-cell malignancies.
Researchers said the safety profile of umbralisib “was distinct from that of other PI3Kδ inhibitors,” as it produced few immune-mediated adverse events (AEs).
Umbralisib also produced an objective response rate of 37% in the entire study cohort, 80% in patients with chronic lymphocytic leukemia (CLL), 53% in patients with follicular lymphoma (FL), and 31% in patients with diffuse large B-cell lymphoma (DLBCL).
These results were published in The Lancet Oncology. The study was sponsored by TG Therapeutics, Inc.
The trial enrolled 90 patients between January 17, 2013, and January 14, 2016.
There were 24 patients with CLL, 22 with FL, 16 with DLBCL, 11 with Hodgkin lymphoma, 6 with mantle cell lymphoma, 5 with marginal zone lymphoma, 3 with Waldenstrom’s macroglobulinemia, 2 with T-cell lymphoma, and 1 with hairy cell leukemia.
The median number of prior therapies was 3 (range, 2-5), and 49% of patients were refractory to previous therapy.
Treatment
Patients took umbralisib once daily in 28-day cycles until disease progression, unacceptable toxicity, or withdrawal of consent.
Initially, patients took the drug in a fasting state at doses of 50 mg, 100 mg, 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In April 2014, the researchers did a second dose-escalation with a micronized formulation of umbralisib, taken with food, at doses of 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In August, 2014, all patients who were still on the study transitioned to the 800 mg dose of the micronized formulation. This was the recommended phase 2 dose.
At the data cutoff in November 2016, 44 patients (49%) had received umbralisib for more than 6 cycles, and 23 (26%) had received the drug for more than 12 cycles. Thirteen patients (14%) were still taking umbralisib at the end of the study.
Most patients who stopped treatment did so because of disease progression (n=50, 56%) or AEs (n=9, 10%).
“We are pleased to have treated the first patient ever with umbralisib over 5 years ago and believe it has an important place in the treatment landscape for patients with hematologic malignancies,” said study author Howard A. Burris, MD, of the Sarah Cannon Research Institute in Nashville, Tennessee.
“Several patients from this phase 1 study are still on study today, approaching 5 years of continuous daily therapy, speaking to both the safety and efficacy profile of this unique agent.”
Safety
Dose-limiting toxicities (DLTs) occurred in 4 patients. One DLT was grade 3 maculopapular rash in a patient receiving the 800 mg dose of the initial formulation.
Another DLT was grade 3 hypokalemia in a patient receiving 1800 mg of the initial formulation. A third DLT was grade 3 fatigue, which occurred in 2 patients receiving 1800 mg of the micronized formulation.
Because of these toxicities, the maximum tolerated dose was 1200 mg of the micronized formulation.
The most common treatment-emergent AEs were diarrhea (43%), nausea (42%), and fatigue (31%). The most common grade 3/4 AEs were neutropenia (13%), anemia (9%), and thrombocytopenia (7%).
Serious AEs considered at least possibly related to umbralisib were pneumonia (3%), lung infection (1%), febrile neutropenia (1%), and colitis (2%).
Treatment discontinuation due to AEs considered at least possibly related to umbralisib occurred in 6 patients (7%). Two patients had grade 3 colitis, 2 had increased ALT/AST (grade 1 and grade 4), 1 had grade 2 diarrhea, and 1 had grade 3 fatigue.
There were no treatment-related deaths.
The researchers said the safety profile of umbralisib was distinct from that of other PI3Kδ inhibitors, as patients in this trial had fewer occurrences of autoimmune-like toxicities, such as colitis.
“Preclinically, umbralisib has a very unique profile, selectively inhibiting both PI3Kδ and CK1ε,” said study author Owen O’Connor, MD, PhD, of Columbia Presbyterian Medical Center in New York, New York.
“The clinical results in this paper support our thesis that the differentiated preclinical profile explains the differences seen in the clinic between umbralisib and the other PI3Kδ inhibitors.”
Response
The objective response rate was 37%, with 33 patients achieving a response and 3 patients having a complete response (CR).
Sixteen CLL patients responded (80%), all with partial responses (PRs). Four DLBCL patients responded (31%), all with PRs. And 9 FL patients responded (53%), 2 with CRs.
The remaining CR occurred in a Hodgkin lymphoma patient, and this was the only response in this patient group.
One patient with marginal zone lymphoma had a PR, as did 1 patient with mantle cell lymphoma. All other patients had stable disease or progressed.
The mean duration of response was 13.4 months in the CLL patients, 6.4 months in the DLBCL patients, and 9.3 months in the FL patients.
Phase 1 trial results suggest umbralisib, a PI3Kδ/CK1ε inhibitor, can be safe and active in patients with relapsed or refractory B-cell malignancies.
Researchers said the safety profile of umbralisib “was distinct from that of other PI3Kδ inhibitors,” as it produced few immune-mediated adverse events (AEs).
Umbralisib also produced an objective response rate of 37% in the entire study cohort, 80% in patients with chronic lymphocytic leukemia (CLL), 53% in patients with follicular lymphoma (FL), and 31% in patients with diffuse large B-cell lymphoma (DLBCL).
These results were published in The Lancet Oncology. The study was sponsored by TG Therapeutics, Inc.
The trial enrolled 90 patients between January 17, 2013, and January 14, 2016.
There were 24 patients with CLL, 22 with FL, 16 with DLBCL, 11 with Hodgkin lymphoma, 6 with mantle cell lymphoma, 5 with marginal zone lymphoma, 3 with Waldenstrom’s macroglobulinemia, 2 with T-cell lymphoma, and 1 with hairy cell leukemia.
The median number of prior therapies was 3 (range, 2-5), and 49% of patients were refractory to previous therapy.
Treatment
Patients took umbralisib once daily in 28-day cycles until disease progression, unacceptable toxicity, or withdrawal of consent.
Initially, patients took the drug in a fasting state at doses of 50 mg, 100 mg, 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In April 2014, the researchers did a second dose-escalation with a micronized formulation of umbralisib, taken with food, at doses of 200 mg, 400 mg, 800 mg, 1200 mg, or 1800 mg.
In August, 2014, all patients who were still on the study transitioned to the 800 mg dose of the micronized formulation. This was the recommended phase 2 dose.
At the data cutoff in November 2016, 44 patients (49%) had received umbralisib for more than 6 cycles, and 23 (26%) had received the drug for more than 12 cycles. Thirteen patients (14%) were still taking umbralisib at the end of the study.
Most patients who stopped treatment did so because of disease progression (n=50, 56%) or AEs (n=9, 10%).
“We are pleased to have treated the first patient ever with umbralisib over 5 years ago and believe it has an important place in the treatment landscape for patients with hematologic malignancies,” said study author Howard A. Burris, MD, of the Sarah Cannon Research Institute in Nashville, Tennessee.
“Several patients from this phase 1 study are still on study today, approaching 5 years of continuous daily therapy, speaking to both the safety and efficacy profile of this unique agent.”
Safety
Dose-limiting toxicities (DLTs) occurred in 4 patients. One DLT was grade 3 maculopapular rash in a patient receiving the 800 mg dose of the initial formulation.
Another DLT was grade 3 hypokalemia in a patient receiving 1800 mg of the initial formulation. A third DLT was grade 3 fatigue, which occurred in 2 patients receiving 1800 mg of the micronized formulation.
Because of these toxicities, the maximum tolerated dose was 1200 mg of the micronized formulation.
The most common treatment-emergent AEs were diarrhea (43%), nausea (42%), and fatigue (31%). The most common grade 3/4 AEs were neutropenia (13%), anemia (9%), and thrombocytopenia (7%).
Serious AEs considered at least possibly related to umbralisib were pneumonia (3%), lung infection (1%), febrile neutropenia (1%), and colitis (2%).
Treatment discontinuation due to AEs considered at least possibly related to umbralisib occurred in 6 patients (7%). Two patients had grade 3 colitis, 2 had increased ALT/AST (grade 1 and grade 4), 1 had grade 2 diarrhea, and 1 had grade 3 fatigue.
There were no treatment-related deaths.
The researchers said the safety profile of umbralisib was distinct from that of other PI3Kδ inhibitors, as patients in this trial had fewer occurrences of autoimmune-like toxicities, such as colitis.
“Preclinically, umbralisib has a very unique profile, selectively inhibiting both PI3Kδ and CK1ε,” said study author Owen O’Connor, MD, PhD, of Columbia Presbyterian Medical Center in New York, New York.
“The clinical results in this paper support our thesis that the differentiated preclinical profile explains the differences seen in the clinic between umbralisib and the other PI3Kδ inhibitors.”
Response
The objective response rate was 37%, with 33 patients achieving a response and 3 patients having a complete response (CR).
Sixteen CLL patients responded (80%), all with partial responses (PRs). Four DLBCL patients responded (31%), all with PRs. And 9 FL patients responded (53%), 2 with CRs.
The remaining CR occurred in a Hodgkin lymphoma patient, and this was the only response in this patient group.
One patient with marginal zone lymphoma had a PR, as did 1 patient with mantle cell lymphoma. All other patients had stable disease or progressed.
The mean duration of response was 13.4 months in the CLL patients, 6.4 months in the DLBCL patients, and 9.3 months in the FL patients.
2017 notches up some landmark approvals
With advances in the understanding of cellular pathways, molecular genetics, and immunology, new drugs for cancer are being released at an increasing rate. A variety of novel agents have recently become available for use, generating excitement for patients and oncologists. Keeping track of all of these new agents is increasingly challenging. This brief review will summarize some of the newest drugs, their indications, and benefits (see related article).
Therapies by tumor
Breast cancer
CDK4/6 inhibitors. The CDK4/6 inhibitor palbociclib was approved in 2015 for the treatment of estrogen-positive, HER2-negative advanced breast cancer, and this year, two more drugs in this class – ribociclib and abemaciclib – were approved for the treatment of hormone receptor–positive breast cancer.
Ribociclib (Kisqali) 600 mg daily (3 weeks on, 1 week off) is approved for use in combination with an aromatase inhibitor. In the study on which the approval was based, there was a response rate of 53% for patients in the study group, compared with 37% for those who received aromatase inhibitor alone (progression-free survival (PFS), not reached vs 14.7 months for single-agent aromatase inhibitor).1 The occurrence of neutropenia seemed to be similar to that in patients receiving palbociclib. However, unlike with palbociclib, ribociclib requires ECG monitoring for QTc prolongation as well as monitoring of liver function tests.
Abemaciclib (Verzenio) has been approved in combination with fulvestrant as well as a monotherapy.2 PFS was 16.4 months for abemaciclib (150 mg bid in combination with fulvestrant), compared with 9.3 months for fulvestrant alone, with corresponding response rates of 48% and 21%. As monotherapy, abemaciclib 200 mg bid had a response rate of 20% with a duration of response of 8.6 months.
Tyrosine kinase inhibitors. The tyrosine kinase inhibitor neratinib (Nerlynx) was approved for extended adjuvant treatment of HER2-positive breast cancer after 1 year of adjuvant trastuzumab.3 Given at 240 mg (6 tablets) daily for a year, compared with a no-treatment control arm, it demonstrated an improvement in invasive disease-free survival (DFS) at 2 years from 91.9% to 94.2%, with no difference in overall survival yet noted. It is associated with diarrhea and also requires hepatic function monitoring.
Acute myelogenous leukemia
Multiple new agents were recently approved for use in acute myelogenous leukemia (AML), after decades of slow advance in new drug development.
Midostaurin (Rydapt) is an FLT3 inhibitor approved for use in combination with daunorubicin and Ara-C (cytosine arabinoside) for newly diagnosed AML with FLT3 mutations, which occur in about 30% of AML patients.4 It is given orally on days 8-21 at 50 mg bid with induction and consolidation.
In the study on which the approval was based, there was a 10% improvement in overall survival for this subset of AML patients who have a typically a worse prognosis. Event-free survival in patients in the study group was 8.2 months, compared with 3 months in the control arm patients, who did not receive the agent. The drug was also approved for aggressive systemic mastocytosis.
Enasidenib (Idhifa) has been approved for AML with an IDH2 mutation in the refractory/relapsed settings.5IDH2 mutations are present in about 20% of patients with AML. Given orally at 100 mg daily as a single agent, enasidenib was associated with a 19% complete remission rate. Patients need to be monitored for differentiation syndrome, somewhat similar to what is seen with ATRA with acute promyelocytic leukemia.
Liposomal daunorubicin and cytarabine (Vyxeos) was approved for newly diagnosed therapy- or myelodysplasia-related AML.6 This novel liposomal formulation combines two standard agents and is given intravenously on days 1, 3 and 5 over 90 minutes as daunorubicin 44 mg/m2 and cytarabine 100 mg/ m2. (For a second induction and in lower dose on consolidation cycles, it is given only on days 1 and 3). The liposomal formulation achieved a superior complete response rate compared with the standard 7+3 daunorubicin plus cytarabine regimen (38% vs 26%, respectively) and longer overall survival (9.6 versus 5.9 months) in these generally poor prognosis subsets.
Gemtuzumab ozogamicin (Mylotarg) was initially approved in 2000 but withdrawn from use in 2010 after trials failed to confirm benefit and demonstrated safety concerns. It has now been re-released in a lower dose and schedule from its original label.7 This immunoconjugate of an anti-CD33 bound to calicheamicin is approved for CD33-positive AML. Given at 3 mg/m2 on days 1, 4, and 7 in combination with standard daunorubicin–cytarabine induction chemotherapy, it improved event-free survival from 9.5 to 17.3 months. When administered as a single agent (6 mg/m2 on day 1 and 3 mg/m2 on day 8) in patients who were unable or unwilling to tolerate standard chemotherapy, it improved overall survival (4.9 months versus 3.6 months for best supportive care). As a single agent in relapsed AML, given at 3 mg/m2 days 1, 4, and 7 and followed by cytarabine consolidation, it was associated with a 26% complete response rate, with a median relapse-free survival of 11.6 months.
Ovarian/fallopian tube cancers
PARP inhibitors. For patients with ovarian/fallopian tube cancer, there are new indications and agents for PARP inhibition, including for patients with BRCA mutations (both somatic and germline) and those without BRCA mutations.
Olaparib (Lynparza) was previously approved only in a fourth-line setting for germline BRCA-mutated patients with advanced ovarian cancer, with a response rate of 34% with a median duration of 7.9 months. Given at 300 mg orally bid, it is now approved for use in maintenance in recurrence after response to platinum-based chemotherapy after 2 or more lines of therapy regardless of BRCA status. In this setting, progression-free survival increased to 8.4 months, compared with 4.8 months for placebo.8
Rubicarib (Rubraca) is approved for BRCA-mutated patients (either germline or somatic) with advanced ovarian cancer after two or more lines of chemotherapy.9 At 600 mg orally bid, results from phase 2 trials noted a 54% response rate, with a median duration of 9.2 months.
Niraparib (Zejula) is approved for use in maintenance in recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancers after platinum-based chemotherapy.10 In patients with germline BRCA mutations, niraparib at 300 mg orally daily resulted in a PFS of 21 months, compared with 5.5 months with placebo; PFS in patients with nongermline BRCA mutations was 9.3 versus 3.9 months, respectively.
Non-small cell lung cancer with EML-4 alk translocation
Crizotinib (Xalkori) has been the mainstay for treatment of for EML4-alk translocated non-small cell lung cancer. However, alectinib (Alcensa), previously for predominantly second-line use, seems more active than crizotinib in the first-line setting, particularly in the treatment and prevention of CNS metastases.
In addition, brigantinib (Alunbrig) has been approved for patients who are intolerant/refractory to crizotinib.11 At 90 mg once daily for 7 days, then escalating to 180 mg daily, it was noted to have a 50% response rate in crizotinib failures, including in the CNS.
Ceritinib (Zykadia) was approved at 750 mg once daily for EML4 alk positive NSCLC.12 In first line it had a response rate of 73% (versus 27% for chemotherapy) with a remission duration of 23.9 months (versus 11.1 months for chemotherapy).
Therapies by drug class
PD-1/PD-L1 antibodies
Anti-PD-1 antibodies nivolumab (Opdivo) and pembrolizumab (Keytruda) are widely used for a range of tumor types. Newer approvals for pembrolizumab are for adenocarcinoma of the stomach/gastro-esophageal junction with at least 1% PD-L1 expression, and in any tumor demonstrated to be MSI-high. Newer indications for nivolumab are for bladder cancer, MSI-high colon cancer, and for hepatoma previously treated with sorafenib. The anti-PD-L1 antibody atezolizumab (Tencentriq) is now approved for platinum-resistant metastatic lung cancer, in addition to platinum-ineligible and platinum-resistant urothelial cancer.
Avelumab (Bavencio) is an anti-PD-L1 approved for both Merkel cell and previously treated urothelial cancers at a dose of 10 mg/kg every 2 weeks.13 It demonstrated a 33% response rate for Merkel cell and a 16% response rate for urothelial cancer.
Durvalumab (Imfinzi) is another anti PD-L1 antibody approved at 10 mg/kg every 2 weeks for previously treated urothelial cancer with a 17% response rate (RR: PD-L1 high, 26%; low, 4%).14
PI3K kinase inhibitors
Copanlisib (Aliqopa) is a PI3K inhibitor approved for relapsed follicular lymphoma in patients who have progressed after two previous lines of therapy.15 It is a 60-mg, 1-hour infusion given on days 1, 8, and 15 every 28 days. In a phase 2 tria
BTK inhibitors
Acalabruitnib (Calquence) is approved for adults with previously treated mantle cell lymphoma. In a phase 2 trial at 100 mg orally bid, it achieved an 80% overall and 40% complete response rate.16 These response rates are higher than were seen for ibrutinib in its original phase 2 trial. The spectrum of toxicities seems similar to ibruitinib and includes bleeding, cytopenias, infection, and atrial fibrillation.
CD19 CAR-T cells
Perhaps the most exciting and novel new agents are genetically engineered autologous T cells. Tisagenlecleucel (Kymriah), a chimeric antigen receptor T cell (CART) that targets CD19 is approved for refractory B cell precursor acute lymphoblastic leukemia (in patients under 25 years) where the complete response rate was 83% (including patients with incomplete blood count recovery).17
Axicabtagene ciloleucel (aci-cel; Yescarta), also CD19-directed CART, is approved for adults with relapsed or refractory non-Hodgkin lymphoma after two lines of previous therapy (specifically large-cell lymphoma, primary mediastinal large B-cell lymphoma, and transformed follicular lymphoma). Response rate was 72% (complete, 51%; partial, 21%), with a median duration of response of 9.2 months.18
1. Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375(18):1738-1748.
2. Goetz MP, Toi M, Campone M, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35(32):3638-3646.
3. Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367-377.
4. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464.
5. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017 Aug 10;130(6):722-731.
6. Lancet JE, Rizzieri D, Schiller GJ, et al. Overall survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): subgroup analysis of a phase III study. http://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.7035. Published May 2017. Accessed November 20, 2017.
7. Appelbaum FR, Bernstein ID. Gemtuzumab ozogamicin for acute myeloid leukemia. http://www.bloodjournal.org/content/early/2017/10/11/blood-2017-09-797712?sso-checked=true. September 2017. Accessed November 20, 2017.
8. Kim G, Ison G, McKee AE, et al. FDA approval summary: olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clin Cancer Res. 2015;21:4257-4261.
9. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75-87.
10. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154-2164.
11. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: a randomized, multicenter phase II trial. J Clin Oncol. 2017;35(22):2490-2498.
12. Soria J-C, Tan DSW, MD, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917-929.
13. Apolo AB, Infante JR, Balmanoukian A et al. Avelumab, an anti–programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter, phase Ib study. J Clin Oncol. 2017;35(19):2117-2124.
14. Massard C, Gordon MS, Sharma S, et al. Safety and efficacy of durvalumab (MEDI4736), an anti–programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol. 2016;34(26):3119-3125.
15. Dreyling M, Morschhauser F, Bouabdallah K, et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol. 2017;28(9):2169-2178.
16. Wu J, Zhang M, Liu D. Acalabrutinib (ACP-196): a selective second-generation BTK inhibitor. https://jhoonline.biomedcentral.com/articles/10.1186/s13045-016-0250-9. Published March 9, 2016. Accessed November 20, 2017.
17. Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517.
18. Locke FL, Neelapu SS, Bartlett NL, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295.
With advances in the understanding of cellular pathways, molecular genetics, and immunology, new drugs for cancer are being released at an increasing rate. A variety of novel agents have recently become available for use, generating excitement for patients and oncologists. Keeping track of all of these new agents is increasingly challenging. This brief review will summarize some of the newest drugs, their indications, and benefits (see related article).
Therapies by tumor
Breast cancer
CDK4/6 inhibitors. The CDK4/6 inhibitor palbociclib was approved in 2015 for the treatment of estrogen-positive, HER2-negative advanced breast cancer, and this year, two more drugs in this class – ribociclib and abemaciclib – were approved for the treatment of hormone receptor–positive breast cancer.
Ribociclib (Kisqali) 600 mg daily (3 weeks on, 1 week off) is approved for use in combination with an aromatase inhibitor. In the study on which the approval was based, there was a response rate of 53% for patients in the study group, compared with 37% for those who received aromatase inhibitor alone (progression-free survival (PFS), not reached vs 14.7 months for single-agent aromatase inhibitor).1 The occurrence of neutropenia seemed to be similar to that in patients receiving palbociclib. However, unlike with palbociclib, ribociclib requires ECG monitoring for QTc prolongation as well as monitoring of liver function tests.
Abemaciclib (Verzenio) has been approved in combination with fulvestrant as well as a monotherapy.2 PFS was 16.4 months for abemaciclib (150 mg bid in combination with fulvestrant), compared with 9.3 months for fulvestrant alone, with corresponding response rates of 48% and 21%. As monotherapy, abemaciclib 200 mg bid had a response rate of 20% with a duration of response of 8.6 months.
Tyrosine kinase inhibitors. The tyrosine kinase inhibitor neratinib (Nerlynx) was approved for extended adjuvant treatment of HER2-positive breast cancer after 1 year of adjuvant trastuzumab.3 Given at 240 mg (6 tablets) daily for a year, compared with a no-treatment control arm, it demonstrated an improvement in invasive disease-free survival (DFS) at 2 years from 91.9% to 94.2%, with no difference in overall survival yet noted. It is associated with diarrhea and also requires hepatic function monitoring.
Acute myelogenous leukemia
Multiple new agents were recently approved for use in acute myelogenous leukemia (AML), after decades of slow advance in new drug development.
Midostaurin (Rydapt) is an FLT3 inhibitor approved for use in combination with daunorubicin and Ara-C (cytosine arabinoside) for newly diagnosed AML with FLT3 mutations, which occur in about 30% of AML patients.4 It is given orally on days 8-21 at 50 mg bid with induction and consolidation.
In the study on which the approval was based, there was a 10% improvement in overall survival for this subset of AML patients who have a typically a worse prognosis. Event-free survival in patients in the study group was 8.2 months, compared with 3 months in the control arm patients, who did not receive the agent. The drug was also approved for aggressive systemic mastocytosis.
Enasidenib (Idhifa) has been approved for AML with an IDH2 mutation in the refractory/relapsed settings.5IDH2 mutations are present in about 20% of patients with AML. Given orally at 100 mg daily as a single agent, enasidenib was associated with a 19% complete remission rate. Patients need to be monitored for differentiation syndrome, somewhat similar to what is seen with ATRA with acute promyelocytic leukemia.
Liposomal daunorubicin and cytarabine (Vyxeos) was approved for newly diagnosed therapy- or myelodysplasia-related AML.6 This novel liposomal formulation combines two standard agents and is given intravenously on days 1, 3 and 5 over 90 minutes as daunorubicin 44 mg/m2 and cytarabine 100 mg/ m2. (For a second induction and in lower dose on consolidation cycles, it is given only on days 1 and 3). The liposomal formulation achieved a superior complete response rate compared with the standard 7+3 daunorubicin plus cytarabine regimen (38% vs 26%, respectively) and longer overall survival (9.6 versus 5.9 months) in these generally poor prognosis subsets.
Gemtuzumab ozogamicin (Mylotarg) was initially approved in 2000 but withdrawn from use in 2010 after trials failed to confirm benefit and demonstrated safety concerns. It has now been re-released in a lower dose and schedule from its original label.7 This immunoconjugate of an anti-CD33 bound to calicheamicin is approved for CD33-positive AML. Given at 3 mg/m2 on days 1, 4, and 7 in combination with standard daunorubicin–cytarabine induction chemotherapy, it improved event-free survival from 9.5 to 17.3 months. When administered as a single agent (6 mg/m2 on day 1 and 3 mg/m2 on day 8) in patients who were unable or unwilling to tolerate standard chemotherapy, it improved overall survival (4.9 months versus 3.6 months for best supportive care). As a single agent in relapsed AML, given at 3 mg/m2 days 1, 4, and 7 and followed by cytarabine consolidation, it was associated with a 26% complete response rate, with a median relapse-free survival of 11.6 months.
Ovarian/fallopian tube cancers
PARP inhibitors. For patients with ovarian/fallopian tube cancer, there are new indications and agents for PARP inhibition, including for patients with BRCA mutations (both somatic and germline) and those without BRCA mutations.
Olaparib (Lynparza) was previously approved only in a fourth-line setting for germline BRCA-mutated patients with advanced ovarian cancer, with a response rate of 34% with a median duration of 7.9 months. Given at 300 mg orally bid, it is now approved for use in maintenance in recurrence after response to platinum-based chemotherapy after 2 or more lines of therapy regardless of BRCA status. In this setting, progression-free survival increased to 8.4 months, compared with 4.8 months for placebo.8
Rubicarib (Rubraca) is approved for BRCA-mutated patients (either germline or somatic) with advanced ovarian cancer after two or more lines of chemotherapy.9 At 600 mg orally bid, results from phase 2 trials noted a 54% response rate, with a median duration of 9.2 months.
Niraparib (Zejula) is approved for use in maintenance in recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancers after platinum-based chemotherapy.10 In patients with germline BRCA mutations, niraparib at 300 mg orally daily resulted in a PFS of 21 months, compared with 5.5 months with placebo; PFS in patients with nongermline BRCA mutations was 9.3 versus 3.9 months, respectively.
Non-small cell lung cancer with EML-4 alk translocation
Crizotinib (Xalkori) has been the mainstay for treatment of for EML4-alk translocated non-small cell lung cancer. However, alectinib (Alcensa), previously for predominantly second-line use, seems more active than crizotinib in the first-line setting, particularly in the treatment and prevention of CNS metastases.
In addition, brigantinib (Alunbrig) has been approved for patients who are intolerant/refractory to crizotinib.11 At 90 mg once daily for 7 days, then escalating to 180 mg daily, it was noted to have a 50% response rate in crizotinib failures, including in the CNS.
Ceritinib (Zykadia) was approved at 750 mg once daily for EML4 alk positive NSCLC.12 In first line it had a response rate of 73% (versus 27% for chemotherapy) with a remission duration of 23.9 months (versus 11.1 months for chemotherapy).
Therapies by drug class
PD-1/PD-L1 antibodies
Anti-PD-1 antibodies nivolumab (Opdivo) and pembrolizumab (Keytruda) are widely used for a range of tumor types. Newer approvals for pembrolizumab are for adenocarcinoma of the stomach/gastro-esophageal junction with at least 1% PD-L1 expression, and in any tumor demonstrated to be MSI-high. Newer indications for nivolumab are for bladder cancer, MSI-high colon cancer, and for hepatoma previously treated with sorafenib. The anti-PD-L1 antibody atezolizumab (Tencentriq) is now approved for platinum-resistant metastatic lung cancer, in addition to platinum-ineligible and platinum-resistant urothelial cancer.
Avelumab (Bavencio) is an anti-PD-L1 approved for both Merkel cell and previously treated urothelial cancers at a dose of 10 mg/kg every 2 weeks.13 It demonstrated a 33% response rate for Merkel cell and a 16% response rate for urothelial cancer.
Durvalumab (Imfinzi) is another anti PD-L1 antibody approved at 10 mg/kg every 2 weeks for previously treated urothelial cancer with a 17% response rate (RR: PD-L1 high, 26%; low, 4%).14
PI3K kinase inhibitors
Copanlisib (Aliqopa) is a PI3K inhibitor approved for relapsed follicular lymphoma in patients who have progressed after two previous lines of therapy.15 It is a 60-mg, 1-hour infusion given on days 1, 8, and 15 every 28 days. In a phase 2 tria
BTK inhibitors
Acalabruitnib (Calquence) is approved for adults with previously treated mantle cell lymphoma. In a phase 2 trial at 100 mg orally bid, it achieved an 80% overall and 40% complete response rate.16 These response rates are higher than were seen for ibrutinib in its original phase 2 trial. The spectrum of toxicities seems similar to ibruitinib and includes bleeding, cytopenias, infection, and atrial fibrillation.
CD19 CAR-T cells
Perhaps the most exciting and novel new agents are genetically engineered autologous T cells. Tisagenlecleucel (Kymriah), a chimeric antigen receptor T cell (CART) that targets CD19 is approved for refractory B cell precursor acute lymphoblastic leukemia (in patients under 25 years) where the complete response rate was 83% (including patients with incomplete blood count recovery).17
Axicabtagene ciloleucel (aci-cel; Yescarta), also CD19-directed CART, is approved for adults with relapsed or refractory non-Hodgkin lymphoma after two lines of previous therapy (specifically large-cell lymphoma, primary mediastinal large B-cell lymphoma, and transformed follicular lymphoma). Response rate was 72% (complete, 51%; partial, 21%), with a median duration of response of 9.2 months.18
With advances in the understanding of cellular pathways, molecular genetics, and immunology, new drugs for cancer are being released at an increasing rate. A variety of novel agents have recently become available for use, generating excitement for patients and oncologists. Keeping track of all of these new agents is increasingly challenging. This brief review will summarize some of the newest drugs, their indications, and benefits (see related article).
Therapies by tumor
Breast cancer
CDK4/6 inhibitors. The CDK4/6 inhibitor palbociclib was approved in 2015 for the treatment of estrogen-positive, HER2-negative advanced breast cancer, and this year, two more drugs in this class – ribociclib and abemaciclib – were approved for the treatment of hormone receptor–positive breast cancer.
Ribociclib (Kisqali) 600 mg daily (3 weeks on, 1 week off) is approved for use in combination with an aromatase inhibitor. In the study on which the approval was based, there was a response rate of 53% for patients in the study group, compared with 37% for those who received aromatase inhibitor alone (progression-free survival (PFS), not reached vs 14.7 months for single-agent aromatase inhibitor).1 The occurrence of neutropenia seemed to be similar to that in patients receiving palbociclib. However, unlike with palbociclib, ribociclib requires ECG monitoring for QTc prolongation as well as monitoring of liver function tests.
Abemaciclib (Verzenio) has been approved in combination with fulvestrant as well as a monotherapy.2 PFS was 16.4 months for abemaciclib (150 mg bid in combination with fulvestrant), compared with 9.3 months for fulvestrant alone, with corresponding response rates of 48% and 21%. As monotherapy, abemaciclib 200 mg bid had a response rate of 20% with a duration of response of 8.6 months.
Tyrosine kinase inhibitors. The tyrosine kinase inhibitor neratinib (Nerlynx) was approved for extended adjuvant treatment of HER2-positive breast cancer after 1 year of adjuvant trastuzumab.3 Given at 240 mg (6 tablets) daily for a year, compared with a no-treatment control arm, it demonstrated an improvement in invasive disease-free survival (DFS) at 2 years from 91.9% to 94.2%, with no difference in overall survival yet noted. It is associated with diarrhea and also requires hepatic function monitoring.
Acute myelogenous leukemia
Multiple new agents were recently approved for use in acute myelogenous leukemia (AML), after decades of slow advance in new drug development.
Midostaurin (Rydapt) is an FLT3 inhibitor approved for use in combination with daunorubicin and Ara-C (cytosine arabinoside) for newly diagnosed AML with FLT3 mutations, which occur in about 30% of AML patients.4 It is given orally on days 8-21 at 50 mg bid with induction and consolidation.
In the study on which the approval was based, there was a 10% improvement in overall survival for this subset of AML patients who have a typically a worse prognosis. Event-free survival in patients in the study group was 8.2 months, compared with 3 months in the control arm patients, who did not receive the agent. The drug was also approved for aggressive systemic mastocytosis.
Enasidenib (Idhifa) has been approved for AML with an IDH2 mutation in the refractory/relapsed settings.5IDH2 mutations are present in about 20% of patients with AML. Given orally at 100 mg daily as a single agent, enasidenib was associated with a 19% complete remission rate. Patients need to be monitored for differentiation syndrome, somewhat similar to what is seen with ATRA with acute promyelocytic leukemia.
Liposomal daunorubicin and cytarabine (Vyxeos) was approved for newly diagnosed therapy- or myelodysplasia-related AML.6 This novel liposomal formulation combines two standard agents and is given intravenously on days 1, 3 and 5 over 90 minutes as daunorubicin 44 mg/m2 and cytarabine 100 mg/ m2. (For a second induction and in lower dose on consolidation cycles, it is given only on days 1 and 3). The liposomal formulation achieved a superior complete response rate compared with the standard 7+3 daunorubicin plus cytarabine regimen (38% vs 26%, respectively) and longer overall survival (9.6 versus 5.9 months) in these generally poor prognosis subsets.
Gemtuzumab ozogamicin (Mylotarg) was initially approved in 2000 but withdrawn from use in 2010 after trials failed to confirm benefit and demonstrated safety concerns. It has now been re-released in a lower dose and schedule from its original label.7 This immunoconjugate of an anti-CD33 bound to calicheamicin is approved for CD33-positive AML. Given at 3 mg/m2 on days 1, 4, and 7 in combination with standard daunorubicin–cytarabine induction chemotherapy, it improved event-free survival from 9.5 to 17.3 months. When administered as a single agent (6 mg/m2 on day 1 and 3 mg/m2 on day 8) in patients who were unable or unwilling to tolerate standard chemotherapy, it improved overall survival (4.9 months versus 3.6 months for best supportive care). As a single agent in relapsed AML, given at 3 mg/m2 days 1, 4, and 7 and followed by cytarabine consolidation, it was associated with a 26% complete response rate, with a median relapse-free survival of 11.6 months.
Ovarian/fallopian tube cancers
PARP inhibitors. For patients with ovarian/fallopian tube cancer, there are new indications and agents for PARP inhibition, including for patients with BRCA mutations (both somatic and germline) and those without BRCA mutations.
Olaparib (Lynparza) was previously approved only in a fourth-line setting for germline BRCA-mutated patients with advanced ovarian cancer, with a response rate of 34% with a median duration of 7.9 months. Given at 300 mg orally bid, it is now approved for use in maintenance in recurrence after response to platinum-based chemotherapy after 2 or more lines of therapy regardless of BRCA status. In this setting, progression-free survival increased to 8.4 months, compared with 4.8 months for placebo.8
Rubicarib (Rubraca) is approved for BRCA-mutated patients (either germline or somatic) with advanced ovarian cancer after two or more lines of chemotherapy.9 At 600 mg orally bid, results from phase 2 trials noted a 54% response rate, with a median duration of 9.2 months.
Niraparib (Zejula) is approved for use in maintenance in recurrent epithelial ovarian, fallopian tube, and primary peritoneal cancers after platinum-based chemotherapy.10 In patients with germline BRCA mutations, niraparib at 300 mg orally daily resulted in a PFS of 21 months, compared with 5.5 months with placebo; PFS in patients with nongermline BRCA mutations was 9.3 versus 3.9 months, respectively.
Non-small cell lung cancer with EML-4 alk translocation
Crizotinib (Xalkori) has been the mainstay for treatment of for EML4-alk translocated non-small cell lung cancer. However, alectinib (Alcensa), previously for predominantly second-line use, seems more active than crizotinib in the first-line setting, particularly in the treatment and prevention of CNS metastases.
In addition, brigantinib (Alunbrig) has been approved for patients who are intolerant/refractory to crizotinib.11 At 90 mg once daily for 7 days, then escalating to 180 mg daily, it was noted to have a 50% response rate in crizotinib failures, including in the CNS.
Ceritinib (Zykadia) was approved at 750 mg once daily for EML4 alk positive NSCLC.12 In first line it had a response rate of 73% (versus 27% for chemotherapy) with a remission duration of 23.9 months (versus 11.1 months for chemotherapy).
Therapies by drug class
PD-1/PD-L1 antibodies
Anti-PD-1 antibodies nivolumab (Opdivo) and pembrolizumab (Keytruda) are widely used for a range of tumor types. Newer approvals for pembrolizumab are for adenocarcinoma of the stomach/gastro-esophageal junction with at least 1% PD-L1 expression, and in any tumor demonstrated to be MSI-high. Newer indications for nivolumab are for bladder cancer, MSI-high colon cancer, and for hepatoma previously treated with sorafenib. The anti-PD-L1 antibody atezolizumab (Tencentriq) is now approved for platinum-resistant metastatic lung cancer, in addition to platinum-ineligible and platinum-resistant urothelial cancer.
Avelumab (Bavencio) is an anti-PD-L1 approved for both Merkel cell and previously treated urothelial cancers at a dose of 10 mg/kg every 2 weeks.13 It demonstrated a 33% response rate for Merkel cell and a 16% response rate for urothelial cancer.
Durvalumab (Imfinzi) is another anti PD-L1 antibody approved at 10 mg/kg every 2 weeks for previously treated urothelial cancer with a 17% response rate (RR: PD-L1 high, 26%; low, 4%).14
PI3K kinase inhibitors
Copanlisib (Aliqopa) is a PI3K inhibitor approved for relapsed follicular lymphoma in patients who have progressed after two previous lines of therapy.15 It is a 60-mg, 1-hour infusion given on days 1, 8, and 15 every 28 days. In a phase 2 tria
BTK inhibitors
Acalabruitnib (Calquence) is approved for adults with previously treated mantle cell lymphoma. In a phase 2 trial at 100 mg orally bid, it achieved an 80% overall and 40% complete response rate.16 These response rates are higher than were seen for ibrutinib in its original phase 2 trial. The spectrum of toxicities seems similar to ibruitinib and includes bleeding, cytopenias, infection, and atrial fibrillation.
CD19 CAR-T cells
Perhaps the most exciting and novel new agents are genetically engineered autologous T cells. Tisagenlecleucel (Kymriah), a chimeric antigen receptor T cell (CART) that targets CD19 is approved for refractory B cell precursor acute lymphoblastic leukemia (in patients under 25 years) where the complete response rate was 83% (including patients with incomplete blood count recovery).17
Axicabtagene ciloleucel (aci-cel; Yescarta), also CD19-directed CART, is approved for adults with relapsed or refractory non-Hodgkin lymphoma after two lines of previous therapy (specifically large-cell lymphoma, primary mediastinal large B-cell lymphoma, and transformed follicular lymphoma). Response rate was 72% (complete, 51%; partial, 21%), with a median duration of response of 9.2 months.18
1. Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375(18):1738-1748.
2. Goetz MP, Toi M, Campone M, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35(32):3638-3646.
3. Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367-377.
4. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464.
5. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017 Aug 10;130(6):722-731.
6. Lancet JE, Rizzieri D, Schiller GJ, et al. Overall survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): subgroup analysis of a phase III study. http://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.7035. Published May 2017. Accessed November 20, 2017.
7. Appelbaum FR, Bernstein ID. Gemtuzumab ozogamicin for acute myeloid leukemia. http://www.bloodjournal.org/content/early/2017/10/11/blood-2017-09-797712?sso-checked=true. September 2017. Accessed November 20, 2017.
8. Kim G, Ison G, McKee AE, et al. FDA approval summary: olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clin Cancer Res. 2015;21:4257-4261.
9. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75-87.
10. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154-2164.
11. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: a randomized, multicenter phase II trial. J Clin Oncol. 2017;35(22):2490-2498.
12. Soria J-C, Tan DSW, MD, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917-929.
13. Apolo AB, Infante JR, Balmanoukian A et al. Avelumab, an anti–programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter, phase Ib study. J Clin Oncol. 2017;35(19):2117-2124.
14. Massard C, Gordon MS, Sharma S, et al. Safety and efficacy of durvalumab (MEDI4736), an anti–programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol. 2016;34(26):3119-3125.
15. Dreyling M, Morschhauser F, Bouabdallah K, et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol. 2017;28(9):2169-2178.
16. Wu J, Zhang M, Liu D. Acalabrutinib (ACP-196): a selective second-generation BTK inhibitor. https://jhoonline.biomedcentral.com/articles/10.1186/s13045-016-0250-9. Published March 9, 2016. Accessed November 20, 2017.
17. Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517.
18. Locke FL, Neelapu SS, Bartlett NL, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295.
1. Hortobagyi GN, Stemmer SM, Burris HA, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375(18):1738-1748.
2. Goetz MP, Toi M, Campone M, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35(32):3638-3646.
3. Chan A, Delaloge S, Holmes FA, et al. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2016;17(3):367-377.
4. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464.
5. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017 Aug 10;130(6):722-731.
6. Lancet JE, Rizzieri D, Schiller GJ, et al. Overall survival (OS) with CPX-351 versus 7+3 in older adults with newly diagnosed, therapy-related acute myeloid leukemia (tAML): subgroup analysis of a phase III study. http://ascopubs.org/doi/abs/10.1200/JCO.2017.35.15_suppl.7035. Published May 2017. Accessed November 20, 2017.
7. Appelbaum FR, Bernstein ID. Gemtuzumab ozogamicin for acute myeloid leukemia. http://www.bloodjournal.org/content/early/2017/10/11/blood-2017-09-797712?sso-checked=true. September 2017. Accessed November 20, 2017.
8. Kim G, Ison G, McKee AE, et al. FDA approval summary: olaparib monotherapy in patients with deleterious germline BRCA-mutated advanced ovarian cancer treated with three or more lines of chemotherapy. Clin Cancer Res. 2015;21:4257-4261.
9. Swisher EM, Lin KK, Oza AM, et al. Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 part 1): an international, multicentre, open-label, phase 2 trial. Lancet Oncol. 2017;18:75-87.
10. Mirza MR, Monk BJ, Herrstedt J, et al. Niraparib maintenance therapy in platinum-sensitive, recurrent ovarian cancer. N Engl J Med. 2016;375:2154-2164.
11. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: a randomized, multicenter phase II trial. J Clin Oncol. 2017;35(22):2490-2498.
12. Soria J-C, Tan DSW, MD, Chiari R, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. 2017;389(10072):917-929.
13. Apolo AB, Infante JR, Balmanoukian A et al. Avelumab, an anti–programmed death-ligand 1 antibody, in patients with refractory metastatic urothelial carcinoma: results from a multicenter, phase Ib study. J Clin Oncol. 2017;35(19):2117-2124.
14. Massard C, Gordon MS, Sharma S, et al. Safety and efficacy of durvalumab (MEDI4736), an anti–programmed cell death ligand-1 immune checkpoint inhibitor, in patients with advanced urothelial bladder cancer. J Clin Oncol. 2016;34(26):3119-3125.
15. Dreyling M, Morschhauser F, Bouabdallah K, et al. Phase II study of copanlisib, a PI3K inhibitor, in relapsed or refractory, indolent or aggressive lymphoma. Ann Oncol. 2017;28(9):2169-2178.
16. Wu J, Zhang M, Liu D. Acalabrutinib (ACP-196): a selective second-generation BTK inhibitor. https://jhoonline.biomedcentral.com/articles/10.1186/s13045-016-0250-9. Published March 9, 2016. Accessed November 20, 2017.
17. Maude SL, Frey N, Shaw PA, et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med. 2014;371(16):1507-1517.
18. Locke FL, Neelapu SS, Bartlett NL, et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol Ther. 2017;25(1):285-295.
Group calls on WHO to help fight HTLV-1
A group of scientists and activists are calling on the World Health Organization (WHO) to fight the spread of human T-cell leukemia virus subtype 1 (HTLV-1).
The group has written a letter to the WHO highlighting the global prevalence of HTLV-1 infection and recommending strategies to prevent the transmission of HTLV-1.
An abbreviated version of the letter was published in The Lancet. The full letter is available on the Global Virus Network (GVN)* website.
“Since my colleagues and I discovered HTLV-1 . . ., we have learned that this destructive and lethal virus is causing much devastation in communities with high prevalence,” said Robert C. Gallo, MD, co-founder and director of GVN and a professor at the University of Maryland School of Medicine in Baltimore.
“During the GVN meeting last September, I was astounded to learn of the hyper-endemic numbers in the Aboriginal population of Australia. HTLV-1 is endemic in other regions, including several islands of the Caribbean, and in countries such as Brazil, Iran, Japan, and Peru. We hope that the WHO will agree with us and begin to take action in promoting prevention strategies against HTLV-1.”
HTLV-1 prevalence
In their letter, Dr Gallo and his colleagues cite statistics of HTLV-1 prevalence around the world.
The authors note that, in a hospital-based cohort study conducted in Central Australia, 33.6% of Indigenous people tested HTLV-1 positive, with the incidence rate reaching 48.5% in older men.
Research has suggested that, in Brazil, the HTLV-1 prevalence rate is 1.8% in the general population, 1.3% in blood donors in certain regions, and 1.05% in pregnant women.
It is estimated that nearly 1 million people are HTLV-1-positive in Japan, and 850,000 to 1.7 million people in Nigeria are infected with the virus.
In Gabon, the HTLV-1 prevalence in adults is believed to be 5% to 10%. And in Central African Republic, 7% of older, female Pygmies in the southern region were found to be infected with HTLV-1.
Studies have indicated that 6.1% of the general population in Jamaica is positive for HTLV-1, and other Caribbean islands have similar prevalence rates.
The authors note that HTLV-1 and -2 have also been detected in non-endemic areas due to migration and sexual transmission.
It is estimated that 20,000 to 30,000 people are infected with HTLV-1 in the UK, and 10,000 to 25,000 people are infected in metropolitan France.
Other estimates suggest that 266,000 people are infected with HTLV-1 or -2 in the US, and 3600 people with HTLV-1 associated myelopathy/tropical spastic paraparesis (HAM/TSP) remain undiagnosed.
“The general neglect, globally, of the importance of HTLV-1 as a sexually transmitted infection that causes a range of debilitating inflammatory diseases does our patients, who request a sexual health screen, a disservice,” said Graham P. Taylor, MDMB, DSc, a professor at Imperial College London in the UK.
“It is also important to recognize the importance of mother-to-child transmission of HTLV-1 in the development of adult T-cell leukemia/lymphoma (ATLL) decades later. Despite the availability of highly sensitive and specific diagnostic tests for infection and a proven intervention, except for Japan, there are no antenatal screening programs. Evaluating the cost-effectiveness of such programs should now be a priority.”
“To prevent mother-to-child infection, the Japanese government has been offering HTLV-1 screening for all pregnant women without cost,” noted Yoshi Yamano, MD, PhD, a professor at St. Marianna University School of Medicine in Kawasaki, Japan.
“Taking a leadership role to promote research, it also provides grants for clinical trials and patient registries focused on ATLL and HAM/TSP.”
Preventing transmission
The letter outlines 5 strategies to prevent or reduce transmission of HTLV-1. The authors recommend:
- Protecting the sexually active population with routine HTLV-1 testing in sexual health clinics and through promotion of CMPC—Counsel & Monitor HTLV-1-positive patients, notify Partners, and promote Condom usage
- Protecting blood and organ donors and recipients by testing for HTLV, avoiding use of products that may be infected, and promoting CMPC
- Protecting mothers, babies, and fathers via routine antenatal care testing, advising HTLV-1-positive mothers against breastfeeding (when feasible), and promoting CMPC
- Protecting injectable drug users by promoting HTLV-1 testing, providing safe needles through needle exchange program, and promoting CMPC
- Supporting the general population and healthcare providers by providing access to an up-to-date WHO HTLV-1 Fact Sheet that can help healthcare providers diagnose HTLV-1 and related diseases and help patients protect themselves from HTLV-1.
“This virus has been underestimated since the time of its discovery perhaps because it is restricted to certain regions or because it is not terribly infectious,” said William Hall, MD, PhD, co-founder of GVN and a professor at the University College Dublin in Ireland.
“However, for decades, it has been known that HTLV-1 is highly carcinogenic and causes severe paralytic neurologic disease and immune disorders that can lead to bacterial infections. It is time that the WHO publicize prevention strategies against this devastating virus.”
*GVN is an international coalition of medical virologists dedicated to identifying, researching, fighting, and preventing current and emerging pandemic viruses that pose a threat to public health.
A group of scientists and activists are calling on the World Health Organization (WHO) to fight the spread of human T-cell leukemia virus subtype 1 (HTLV-1).
The group has written a letter to the WHO highlighting the global prevalence of HTLV-1 infection and recommending strategies to prevent the transmission of HTLV-1.
An abbreviated version of the letter was published in The Lancet. The full letter is available on the Global Virus Network (GVN)* website.
“Since my colleagues and I discovered HTLV-1 . . ., we have learned that this destructive and lethal virus is causing much devastation in communities with high prevalence,” said Robert C. Gallo, MD, co-founder and director of GVN and a professor at the University of Maryland School of Medicine in Baltimore.
“During the GVN meeting last September, I was astounded to learn of the hyper-endemic numbers in the Aboriginal population of Australia. HTLV-1 is endemic in other regions, including several islands of the Caribbean, and in countries such as Brazil, Iran, Japan, and Peru. We hope that the WHO will agree with us and begin to take action in promoting prevention strategies against HTLV-1.”
HTLV-1 prevalence
In their letter, Dr Gallo and his colleagues cite statistics of HTLV-1 prevalence around the world.
The authors note that, in a hospital-based cohort study conducted in Central Australia, 33.6% of Indigenous people tested HTLV-1 positive, with the incidence rate reaching 48.5% in older men.
Research has suggested that, in Brazil, the HTLV-1 prevalence rate is 1.8% in the general population, 1.3% in blood donors in certain regions, and 1.05% in pregnant women.
It is estimated that nearly 1 million people are HTLV-1-positive in Japan, and 850,000 to 1.7 million people in Nigeria are infected with the virus.
In Gabon, the HTLV-1 prevalence in adults is believed to be 5% to 10%. And in Central African Republic, 7% of older, female Pygmies in the southern region were found to be infected with HTLV-1.
Studies have indicated that 6.1% of the general population in Jamaica is positive for HTLV-1, and other Caribbean islands have similar prevalence rates.
The authors note that HTLV-1 and -2 have also been detected in non-endemic areas due to migration and sexual transmission.
It is estimated that 20,000 to 30,000 people are infected with HTLV-1 in the UK, and 10,000 to 25,000 people are infected in metropolitan France.
Other estimates suggest that 266,000 people are infected with HTLV-1 or -2 in the US, and 3600 people with HTLV-1 associated myelopathy/tropical spastic paraparesis (HAM/TSP) remain undiagnosed.
“The general neglect, globally, of the importance of HTLV-1 as a sexually transmitted infection that causes a range of debilitating inflammatory diseases does our patients, who request a sexual health screen, a disservice,” said Graham P. Taylor, MDMB, DSc, a professor at Imperial College London in the UK.
“It is also important to recognize the importance of mother-to-child transmission of HTLV-1 in the development of adult T-cell leukemia/lymphoma (ATLL) decades later. Despite the availability of highly sensitive and specific diagnostic tests for infection and a proven intervention, except for Japan, there are no antenatal screening programs. Evaluating the cost-effectiveness of such programs should now be a priority.”
“To prevent mother-to-child infection, the Japanese government has been offering HTLV-1 screening for all pregnant women without cost,” noted Yoshi Yamano, MD, PhD, a professor at St. Marianna University School of Medicine in Kawasaki, Japan.
“Taking a leadership role to promote research, it also provides grants for clinical trials and patient registries focused on ATLL and HAM/TSP.”
Preventing transmission
The letter outlines 5 strategies to prevent or reduce transmission of HTLV-1. The authors recommend:
- Protecting the sexually active population with routine HTLV-1 testing in sexual health clinics and through promotion of CMPC—Counsel & Monitor HTLV-1-positive patients, notify Partners, and promote Condom usage
- Protecting blood and organ donors and recipients by testing for HTLV, avoiding use of products that may be infected, and promoting CMPC
- Protecting mothers, babies, and fathers via routine antenatal care testing, advising HTLV-1-positive mothers against breastfeeding (when feasible), and promoting CMPC
- Protecting injectable drug users by promoting HTLV-1 testing, providing safe needles through needle exchange program, and promoting CMPC
- Supporting the general population and healthcare providers by providing access to an up-to-date WHO HTLV-1 Fact Sheet that can help healthcare providers diagnose HTLV-1 and related diseases and help patients protect themselves from HTLV-1.
“This virus has been underestimated since the time of its discovery perhaps because it is restricted to certain regions or because it is not terribly infectious,” said William Hall, MD, PhD, co-founder of GVN and a professor at the University College Dublin in Ireland.
“However, for decades, it has been known that HTLV-1 is highly carcinogenic and causes severe paralytic neurologic disease and immune disorders that can lead to bacterial infections. It is time that the WHO publicize prevention strategies against this devastating virus.”
*GVN is an international coalition of medical virologists dedicated to identifying, researching, fighting, and preventing current and emerging pandemic viruses that pose a threat to public health.
A group of scientists and activists are calling on the World Health Organization (WHO) to fight the spread of human T-cell leukemia virus subtype 1 (HTLV-1).
The group has written a letter to the WHO highlighting the global prevalence of HTLV-1 infection and recommending strategies to prevent the transmission of HTLV-1.
An abbreviated version of the letter was published in The Lancet. The full letter is available on the Global Virus Network (GVN)* website.
“Since my colleagues and I discovered HTLV-1 . . ., we have learned that this destructive and lethal virus is causing much devastation in communities with high prevalence,” said Robert C. Gallo, MD, co-founder and director of GVN and a professor at the University of Maryland School of Medicine in Baltimore.
“During the GVN meeting last September, I was astounded to learn of the hyper-endemic numbers in the Aboriginal population of Australia. HTLV-1 is endemic in other regions, including several islands of the Caribbean, and in countries such as Brazil, Iran, Japan, and Peru. We hope that the WHO will agree with us and begin to take action in promoting prevention strategies against HTLV-1.”
HTLV-1 prevalence
In their letter, Dr Gallo and his colleagues cite statistics of HTLV-1 prevalence around the world.
The authors note that, in a hospital-based cohort study conducted in Central Australia, 33.6% of Indigenous people tested HTLV-1 positive, with the incidence rate reaching 48.5% in older men.
Research has suggested that, in Brazil, the HTLV-1 prevalence rate is 1.8% in the general population, 1.3% in blood donors in certain regions, and 1.05% in pregnant women.
It is estimated that nearly 1 million people are HTLV-1-positive in Japan, and 850,000 to 1.7 million people in Nigeria are infected with the virus.
In Gabon, the HTLV-1 prevalence in adults is believed to be 5% to 10%. And in Central African Republic, 7% of older, female Pygmies in the southern region were found to be infected with HTLV-1.
Studies have indicated that 6.1% of the general population in Jamaica is positive for HTLV-1, and other Caribbean islands have similar prevalence rates.
The authors note that HTLV-1 and -2 have also been detected in non-endemic areas due to migration and sexual transmission.
It is estimated that 20,000 to 30,000 people are infected with HTLV-1 in the UK, and 10,000 to 25,000 people are infected in metropolitan France.
Other estimates suggest that 266,000 people are infected with HTLV-1 or -2 in the US, and 3600 people with HTLV-1 associated myelopathy/tropical spastic paraparesis (HAM/TSP) remain undiagnosed.
“The general neglect, globally, of the importance of HTLV-1 as a sexually transmitted infection that causes a range of debilitating inflammatory diseases does our patients, who request a sexual health screen, a disservice,” said Graham P. Taylor, MDMB, DSc, a professor at Imperial College London in the UK.
“It is also important to recognize the importance of mother-to-child transmission of HTLV-1 in the development of adult T-cell leukemia/lymphoma (ATLL) decades later. Despite the availability of highly sensitive and specific diagnostic tests for infection and a proven intervention, except for Japan, there are no antenatal screening programs. Evaluating the cost-effectiveness of such programs should now be a priority.”
“To prevent mother-to-child infection, the Japanese government has been offering HTLV-1 screening for all pregnant women without cost,” noted Yoshi Yamano, MD, PhD, a professor at St. Marianna University School of Medicine in Kawasaki, Japan.
“Taking a leadership role to promote research, it also provides grants for clinical trials and patient registries focused on ATLL and HAM/TSP.”
Preventing transmission
The letter outlines 5 strategies to prevent or reduce transmission of HTLV-1. The authors recommend:
- Protecting the sexually active population with routine HTLV-1 testing in sexual health clinics and through promotion of CMPC—Counsel & Monitor HTLV-1-positive patients, notify Partners, and promote Condom usage
- Protecting blood and organ donors and recipients by testing for HTLV, avoiding use of products that may be infected, and promoting CMPC
- Protecting mothers, babies, and fathers via routine antenatal care testing, advising HTLV-1-positive mothers against breastfeeding (when feasible), and promoting CMPC
- Protecting injectable drug users by promoting HTLV-1 testing, providing safe needles through needle exchange program, and promoting CMPC
- Supporting the general population and healthcare providers by providing access to an up-to-date WHO HTLV-1 Fact Sheet that can help healthcare providers diagnose HTLV-1 and related diseases and help patients protect themselves from HTLV-1.
“This virus has been underestimated since the time of its discovery perhaps because it is restricted to certain regions or because it is not terribly infectious,” said William Hall, MD, PhD, co-founder of GVN and a professor at the University College Dublin in Ireland.
“However, for decades, it has been known that HTLV-1 is highly carcinogenic and causes severe paralytic neurologic disease and immune disorders that can lead to bacterial infections. It is time that the WHO publicize prevention strategies against this devastating virus.”
*GVN is an international coalition of medical virologists dedicated to identifying, researching, fighting, and preventing current and emerging pandemic viruses that pose a threat to public health.
5-year data show deepening response with ibrutinib in CLL
Single-agent ibrutinib has had sustained efficacy and a rate of complete response that has increased over time, according to a 5-year follow-up report including 132 patients with chronic lymphocytic leukemia (CLL).
Efficacy has been maintained in both treatment-naive and relapsed/refractory CLL, despite the presence of high-risk genomic features in many patients, investigators reported in Blood.
Treatment has been well tolerated, and the occurrence of severe adverse events has diminished over time, according to Susan M. O’Brien, MD, of the Chao Family Comprehensive Cancer Center, University of California, Irvine, and her colleagues.
“The safety profile of ibrutinib over time remains acceptable and manageable, allowing almost one-half of the patients (48%) to be treated for more than 4 years and thus maximize response,” the investigators wrote.
The report was based on 5-year follow-up of patients with CLL who had been enrolled in a phase 1b/2 study (PCYC-1102) and an extension study (PCYC-1103). A total of 132 patients were evaluated, including 101 with relapsed/refractory disease and 31 who were treatment naive.
The overall response rate remained high, at 89% in this 5-year follow-up. Complete response rates increased over time, reaching 29% in treatment-naive patients and 10% in relapsed/refractory patients. In a previous report on 3-year follow-up for these patients, investigators reported complete response rates of 23% in the previously untreated group and 7% in the relapsed/refractory group. The new findings demonstrate “deepening of responses” with continued ibrutinib therapy, Dr. O’Brien and her coauthors wrote.
The 5-year rate of progression-free survival was 44% for relapsed/refractory patients and 92% for treatment-naive patients in this study. Median progression-free survival was 51 months for the relapsed/refractory cohort. “[Progression-free survival] with single-agent ibrutinib in [treatment-naive] patients appears particularly favorable, because the median has not been reached,” investigators wrote.
Adverse events that limited treatment were more frequent during the first year of treatment than in subsequent years, data show, while new onset of neutropenia and thrombocytopenia decreased over time.
The overall response rate was high even in patients with high-risk genetic features. Rates of overall response ranged from 79% to 97%, depending on genomic subgroup. That held true for relapsed/refractory CLL patients with del(17p), a significant negative prognostic factor for survival. In that group, the overall response rate was 79% and the duration of response was 31 months, representing “promising single-agent efficacy” in that group, investigators said.
Pharmacyclics, an AbbVie Company, provided funding for the study and writing support. The study was also supported by grants from the National Institutes of Health and a private foundation. Dr. O’Brien reported ties to AbbVie, Janssen, and Pharmacyclics. Other investigators reported financial relationships with various pharmaceutical companies.
SOURCE: O’Brien S et al. Blood. 2018;131(17):1910-9.
This report on the 5-year experience with single-agent ibrutinib in chronic lymphocytic leukemia (CLL) is a step forward in knowledge of the drug’s natural history, according to Jennifer R. Brown, MD, PhD.
“Ibrutinib data are starting to mature, but much opportunity for growth remains,” Dr. Brown said in an editorial.
One notable update in the report is that a median, progression-free survival has been reached in patients with relapsed/refractory CLL. The reported 51-month median progression-free survival is “strikingly good” in this cohort of patients with high-risk disease, and compares favorably to older regimens in similar patient populations, she said.
Regarding previously untreated CLL patients, it is notable that 45% of this cohort discontinued treatment, many apparently after year 4, Dr. Brown said. The finding that median duration on therapy was just about 5.5 years in the cohort could be “potentially quite useful” in counseling patients, if confirmed in a larger cohort, she added.
Given the discontinuation data, one unanswered question is the durability of remission after stopping ibrutinib in a “deep but probably not complete remission” versus continuing therapy.
“Further follow-up of patients who discontinue without disease progression, as well as systematic investigation of time-limited therapy, including novel likely combination approaches, is clearly warranted,” Dr. Brown said in her editorial.
Dr. Jennifer R. Brown is with the Dana-Farber Cancer Institute in Boston. These comments are adapted an accompanying editorial ( Blood. 2018;131:1880-2 ). Dr. Brown reported ties to Janssen, Pharmacyclics, AstraZeneca, Sun, Redx, Sunesis, Loxo, Gilead, TG Therapeutics, Verastem, and AbbVie.
This report on the 5-year experience with single-agent ibrutinib in chronic lymphocytic leukemia (CLL) is a step forward in knowledge of the drug’s natural history, according to Jennifer R. Brown, MD, PhD.
“Ibrutinib data are starting to mature, but much opportunity for growth remains,” Dr. Brown said in an editorial.
One notable update in the report is that a median, progression-free survival has been reached in patients with relapsed/refractory CLL. The reported 51-month median progression-free survival is “strikingly good” in this cohort of patients with high-risk disease, and compares favorably to older regimens in similar patient populations, she said.
Regarding previously untreated CLL patients, it is notable that 45% of this cohort discontinued treatment, many apparently after year 4, Dr. Brown said. The finding that median duration on therapy was just about 5.5 years in the cohort could be “potentially quite useful” in counseling patients, if confirmed in a larger cohort, she added.
Given the discontinuation data, one unanswered question is the durability of remission after stopping ibrutinib in a “deep but probably not complete remission” versus continuing therapy.
“Further follow-up of patients who discontinue without disease progression, as well as systematic investigation of time-limited therapy, including novel likely combination approaches, is clearly warranted,” Dr. Brown said in her editorial.
Dr. Jennifer R. Brown is with the Dana-Farber Cancer Institute in Boston. These comments are adapted an accompanying editorial ( Blood. 2018;131:1880-2 ). Dr. Brown reported ties to Janssen, Pharmacyclics, AstraZeneca, Sun, Redx, Sunesis, Loxo, Gilead, TG Therapeutics, Verastem, and AbbVie.
This report on the 5-year experience with single-agent ibrutinib in chronic lymphocytic leukemia (CLL) is a step forward in knowledge of the drug’s natural history, according to Jennifer R. Brown, MD, PhD.
“Ibrutinib data are starting to mature, but much opportunity for growth remains,” Dr. Brown said in an editorial.
One notable update in the report is that a median, progression-free survival has been reached in patients with relapsed/refractory CLL. The reported 51-month median progression-free survival is “strikingly good” in this cohort of patients with high-risk disease, and compares favorably to older regimens in similar patient populations, she said.
Regarding previously untreated CLL patients, it is notable that 45% of this cohort discontinued treatment, many apparently after year 4, Dr. Brown said. The finding that median duration on therapy was just about 5.5 years in the cohort could be “potentially quite useful” in counseling patients, if confirmed in a larger cohort, she added.
Given the discontinuation data, one unanswered question is the durability of remission after stopping ibrutinib in a “deep but probably not complete remission” versus continuing therapy.
“Further follow-up of patients who discontinue without disease progression, as well as systematic investigation of time-limited therapy, including novel likely combination approaches, is clearly warranted,” Dr. Brown said in her editorial.
Dr. Jennifer R. Brown is with the Dana-Farber Cancer Institute in Boston. These comments are adapted an accompanying editorial ( Blood. 2018;131:1880-2 ). Dr. Brown reported ties to Janssen, Pharmacyclics, AstraZeneca, Sun, Redx, Sunesis, Loxo, Gilead, TG Therapeutics, Verastem, and AbbVie.
Single-agent ibrutinib has had sustained efficacy and a rate of complete response that has increased over time, according to a 5-year follow-up report including 132 patients with chronic lymphocytic leukemia (CLL).
Efficacy has been maintained in both treatment-naive and relapsed/refractory CLL, despite the presence of high-risk genomic features in many patients, investigators reported in Blood.
Treatment has been well tolerated, and the occurrence of severe adverse events has diminished over time, according to Susan M. O’Brien, MD, of the Chao Family Comprehensive Cancer Center, University of California, Irvine, and her colleagues.
“The safety profile of ibrutinib over time remains acceptable and manageable, allowing almost one-half of the patients (48%) to be treated for more than 4 years and thus maximize response,” the investigators wrote.
The report was based on 5-year follow-up of patients with CLL who had been enrolled in a phase 1b/2 study (PCYC-1102) and an extension study (PCYC-1103). A total of 132 patients were evaluated, including 101 with relapsed/refractory disease and 31 who were treatment naive.
The overall response rate remained high, at 89% in this 5-year follow-up. Complete response rates increased over time, reaching 29% in treatment-naive patients and 10% in relapsed/refractory patients. In a previous report on 3-year follow-up for these patients, investigators reported complete response rates of 23% in the previously untreated group and 7% in the relapsed/refractory group. The new findings demonstrate “deepening of responses” with continued ibrutinib therapy, Dr. O’Brien and her coauthors wrote.
The 5-year rate of progression-free survival was 44% for relapsed/refractory patients and 92% for treatment-naive patients in this study. Median progression-free survival was 51 months for the relapsed/refractory cohort. “[Progression-free survival] with single-agent ibrutinib in [treatment-naive] patients appears particularly favorable, because the median has not been reached,” investigators wrote.
Adverse events that limited treatment were more frequent during the first year of treatment than in subsequent years, data show, while new onset of neutropenia and thrombocytopenia decreased over time.
The overall response rate was high even in patients with high-risk genetic features. Rates of overall response ranged from 79% to 97%, depending on genomic subgroup. That held true for relapsed/refractory CLL patients with del(17p), a significant negative prognostic factor for survival. In that group, the overall response rate was 79% and the duration of response was 31 months, representing “promising single-agent efficacy” in that group, investigators said.
Pharmacyclics, an AbbVie Company, provided funding for the study and writing support. The study was also supported by grants from the National Institutes of Health and a private foundation. Dr. O’Brien reported ties to AbbVie, Janssen, and Pharmacyclics. Other investigators reported financial relationships with various pharmaceutical companies.
SOURCE: O’Brien S et al. Blood. 2018;131(17):1910-9.
Single-agent ibrutinib has had sustained efficacy and a rate of complete response that has increased over time, according to a 5-year follow-up report including 132 patients with chronic lymphocytic leukemia (CLL).
Efficacy has been maintained in both treatment-naive and relapsed/refractory CLL, despite the presence of high-risk genomic features in many patients, investigators reported in Blood.
Treatment has been well tolerated, and the occurrence of severe adverse events has diminished over time, according to Susan M. O’Brien, MD, of the Chao Family Comprehensive Cancer Center, University of California, Irvine, and her colleagues.
“The safety profile of ibrutinib over time remains acceptable and manageable, allowing almost one-half of the patients (48%) to be treated for more than 4 years and thus maximize response,” the investigators wrote.
The report was based on 5-year follow-up of patients with CLL who had been enrolled in a phase 1b/2 study (PCYC-1102) and an extension study (PCYC-1103). A total of 132 patients were evaluated, including 101 with relapsed/refractory disease and 31 who were treatment naive.
The overall response rate remained high, at 89% in this 5-year follow-up. Complete response rates increased over time, reaching 29% in treatment-naive patients and 10% in relapsed/refractory patients. In a previous report on 3-year follow-up for these patients, investigators reported complete response rates of 23% in the previously untreated group and 7% in the relapsed/refractory group. The new findings demonstrate “deepening of responses” with continued ibrutinib therapy, Dr. O’Brien and her coauthors wrote.
The 5-year rate of progression-free survival was 44% for relapsed/refractory patients and 92% for treatment-naive patients in this study. Median progression-free survival was 51 months for the relapsed/refractory cohort. “[Progression-free survival] with single-agent ibrutinib in [treatment-naive] patients appears particularly favorable, because the median has not been reached,” investigators wrote.
Adverse events that limited treatment were more frequent during the first year of treatment than in subsequent years, data show, while new onset of neutropenia and thrombocytopenia decreased over time.
The overall response rate was high even in patients with high-risk genetic features. Rates of overall response ranged from 79% to 97%, depending on genomic subgroup. That held true for relapsed/refractory CLL patients with del(17p), a significant negative prognostic factor for survival. In that group, the overall response rate was 79% and the duration of response was 31 months, representing “promising single-agent efficacy” in that group, investigators said.
Pharmacyclics, an AbbVie Company, provided funding for the study and writing support. The study was also supported by grants from the National Institutes of Health and a private foundation. Dr. O’Brien reported ties to AbbVie, Janssen, and Pharmacyclics. Other investigators reported financial relationships with various pharmaceutical companies.
SOURCE: O’Brien S et al. Blood. 2018;131(17):1910-9.
FROM BLOOD
Key clinical point:
Major finding: The 5-year rate of progression-free survival was 44% for relapsed/refractory patients and 92% for treatment-naive patients.
Study details: Report on 5-year follow-up of 132 patients with CLL enrolled in a phase 1b/2 study (PCYC-1102) and an extension study (PCYC-1103).
Disclosures: Pharmacyclics, an AbbVie Company, provided funding for the study and writing support. The study was also supported by grants from the National Institutes of Health and a private foundation. The authors reported ties to Pharmacyclics and other companies.
Source: O’Brien S et al. Blood. 2018;131(17):1910-9.
Sorafenib Improves Survival for Patients With Rare Sarcomas
Promising results from a phase 3 trial could represent a paradigm shift in treatment of patients with desmoid tumors, say National Cancer Institute (NCI) researchers. Sorafenib tosylate extended progression-free survival (PFS) compared with that of placebo in 87 patients with desmoid tumors or aggressive fibromatosis (DT/DF).
Desmoid tumors or aggressive fibromatosis are rare sarcomas, usually found in the extremities or the abdomen and occasionally associated with familial adenomatous polyposis or Gardner syndrome. Effectiveness of current treatments—surgery, radiation, chemotherapy—is generally limited, says Jeff Abrams, MD, clinical director of NCI’s Division of Cancer Treatment and Diagnosis.
Sorafenib is a targeted treatment that interferes with the growth of cancer cells and new blood vessels that feed the tumors. It is approved for some patients with advanced kidney, liver, and thyroid cancers. It was chosen for the NCI trial because in earlier studies DT shrank in patients on sorafenib, and patients had fewer symptoms, including less pain.
Patients were assigned to receive sorafenib 400 mg/d or placebo. The trial was designed to assess improvement in PFS from 6 months for placebo to 15 months for sorafenib. Based on an interim analysis of the first 75 patients enrolled, the observed improvement in PFS exceeded the target. Patients on sorafenib were more likely to experience drug-related adverse effects, but those were generally not severe.
Treatment assignments have been disclosed to patients still receiving treatment and their physicians. Those who were receiving sorafenib can continue therapy, and those on placebo can switch to sorafenib.
Promising results from a phase 3 trial could represent a paradigm shift in treatment of patients with desmoid tumors, say National Cancer Institute (NCI) researchers. Sorafenib tosylate extended progression-free survival (PFS) compared with that of placebo in 87 patients with desmoid tumors or aggressive fibromatosis (DT/DF).
Desmoid tumors or aggressive fibromatosis are rare sarcomas, usually found in the extremities or the abdomen and occasionally associated with familial adenomatous polyposis or Gardner syndrome. Effectiveness of current treatments—surgery, radiation, chemotherapy—is generally limited, says Jeff Abrams, MD, clinical director of NCI’s Division of Cancer Treatment and Diagnosis.
Sorafenib is a targeted treatment that interferes with the growth of cancer cells and new blood vessels that feed the tumors. It is approved for some patients with advanced kidney, liver, and thyroid cancers. It was chosen for the NCI trial because in earlier studies DT shrank in patients on sorafenib, and patients had fewer symptoms, including less pain.
Patients were assigned to receive sorafenib 400 mg/d or placebo. The trial was designed to assess improvement in PFS from 6 months for placebo to 15 months for sorafenib. Based on an interim analysis of the first 75 patients enrolled, the observed improvement in PFS exceeded the target. Patients on sorafenib were more likely to experience drug-related adverse effects, but those were generally not severe.
Treatment assignments have been disclosed to patients still receiving treatment and their physicians. Those who were receiving sorafenib can continue therapy, and those on placebo can switch to sorafenib.
Promising results from a phase 3 trial could represent a paradigm shift in treatment of patients with desmoid tumors, say National Cancer Institute (NCI) researchers. Sorafenib tosylate extended progression-free survival (PFS) compared with that of placebo in 87 patients with desmoid tumors or aggressive fibromatosis (DT/DF).
Desmoid tumors or aggressive fibromatosis are rare sarcomas, usually found in the extremities or the abdomen and occasionally associated with familial adenomatous polyposis or Gardner syndrome. Effectiveness of current treatments—surgery, radiation, chemotherapy—is generally limited, says Jeff Abrams, MD, clinical director of NCI’s Division of Cancer Treatment and Diagnosis.
Sorafenib is a targeted treatment that interferes with the growth of cancer cells and new blood vessels that feed the tumors. It is approved for some patients with advanced kidney, liver, and thyroid cancers. It was chosen for the NCI trial because in earlier studies DT shrank in patients on sorafenib, and patients had fewer symptoms, including less pain.
Patients were assigned to receive sorafenib 400 mg/d or placebo. The trial was designed to assess improvement in PFS from 6 months for placebo to 15 months for sorafenib. Based on an interim analysis of the first 75 patients enrolled, the observed improvement in PFS exceeded the target. Patients on sorafenib were more likely to experience drug-related adverse effects, but those were generally not severe.
Treatment assignments have been disclosed to patients still receiving treatment and their physicians. Those who were receiving sorafenib can continue therapy, and those on placebo can switch to sorafenib.
Team identifies new prognostic factor for MF
New research suggests DNA sequencing can reveal which patients with early stage mycosis fungoides (MF) will develop aggressive disease.
By sequencing the T-cell receptor beta gene (TCRB) in skin biopsies from MF patients, investigators were able to measure the tumor clone frequency (TCF), or the percentage of all T cells that represent the MF clone.
The researchers found the TCF could predict progression-free survival (PFS) and overall survival (OS).
In fact, for patients with early stage MF, TCF was a stronger predictor of PFS than other established prognostic factors.
“While more work needs to be done, we think this approach has the potential to prospectively identify a subgroup of patients who are destined to develop aggressive, life-threatening disease and treat them in a more aggressive fashion with the intent to better manage and, ideally, cure their cancer,” said Thomas Kupper, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“At the same time, we believe we can provide reassurance to patients with a low-risk (low TCF) profile that they are likely to survive indefinitely with conventional conservative therapies. As a physician who has treated this disease for decades, I am excited to be involved with work that so directly and profoundly affects the care and management of these patients.”
Dr Kupper and his colleagues described this work in Science Translational Medicine.
The researchers had previously published a study showing that high-throughput sequencing of the TCRB gene was an accurate way to diagnose cutaneous T-cell lymphoma (CTCL), including MF.
With the current study, the investigators set out to determine if the method could be used to predict progression and survival in patients with CTCL.
The team sequenced TCRB in lesional skin biopsies from 309 CTCL patients, most of whom had MF. The discovery cohort had 208 patients (177 with MF), and the validation cohort had 101 patients (87 with MF).
The sequencing produced a snapshot of the TCRB genes from a large number of cells at the site of the lesion. And the researchers could use this to measure TCF.
The team tested the association of TCF with prognosis in all CTCL patients in the discovery cohort and found a TCF greater than 25% in the skin was significantly associated with reduced PFS (P<0.001) and OS (P<0.001). Results were similar in the validation cohort (P<0.001 for PFS and OS).
The investigators also found that TCF was significantly associated with PFS (P<0.001) and OS (P<0.001) in MF patients but not in patients with Sézary syndrome. The team noted that they did not assess the predictive value of TCF in the blood of Sézary patients.
In a multivariable analysis, TCF was still significantly associated with PFS (P<0.001) and OS (P<0.001) in the MF patients.
The researchers also assessed potential prognostic variables in the early stage MF patients and found a TCF greater than 25% was a stronger predictor of PFS than any other established prognostic factor. This includes disease stage, presence of plaques, elevated lactate dehydrogenase, age, large-cell transformation, and CLIPI score.
“In reviewing the results, 2 different patients could have identical-looking skin lesions, but one might have a TCF of 8%, and one would have a TCF of 40%,” Dr Kupper noted. “The latter patient was highly likely to progress—something we would never have been able to predict before this discovery.”
“The TCF was independent of how thick or thin the skin lesions were. Most importantly, compared to all other currently used means of trying to predict which patients would progress, TCF was by far the most sensitive and specific.”
The high-throughput sequencing in this study was performed using ImmunoSEQ, an assay developed by Adaptive Biotechnologies. The company did not sponsor the study, but company employees were involved in the research.
New research suggests DNA sequencing can reveal which patients with early stage mycosis fungoides (MF) will develop aggressive disease.
By sequencing the T-cell receptor beta gene (TCRB) in skin biopsies from MF patients, investigators were able to measure the tumor clone frequency (TCF), or the percentage of all T cells that represent the MF clone.
The researchers found the TCF could predict progression-free survival (PFS) and overall survival (OS).
In fact, for patients with early stage MF, TCF was a stronger predictor of PFS than other established prognostic factors.
“While more work needs to be done, we think this approach has the potential to prospectively identify a subgroup of patients who are destined to develop aggressive, life-threatening disease and treat them in a more aggressive fashion with the intent to better manage and, ideally, cure their cancer,” said Thomas Kupper, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“At the same time, we believe we can provide reassurance to patients with a low-risk (low TCF) profile that they are likely to survive indefinitely with conventional conservative therapies. As a physician who has treated this disease for decades, I am excited to be involved with work that so directly and profoundly affects the care and management of these patients.”
Dr Kupper and his colleagues described this work in Science Translational Medicine.
The researchers had previously published a study showing that high-throughput sequencing of the TCRB gene was an accurate way to diagnose cutaneous T-cell lymphoma (CTCL), including MF.
With the current study, the investigators set out to determine if the method could be used to predict progression and survival in patients with CTCL.
The team sequenced TCRB in lesional skin biopsies from 309 CTCL patients, most of whom had MF. The discovery cohort had 208 patients (177 with MF), and the validation cohort had 101 patients (87 with MF).
The sequencing produced a snapshot of the TCRB genes from a large number of cells at the site of the lesion. And the researchers could use this to measure TCF.
The team tested the association of TCF with prognosis in all CTCL patients in the discovery cohort and found a TCF greater than 25% in the skin was significantly associated with reduced PFS (P<0.001) and OS (P<0.001). Results were similar in the validation cohort (P<0.001 for PFS and OS).
The investigators also found that TCF was significantly associated with PFS (P<0.001) and OS (P<0.001) in MF patients but not in patients with Sézary syndrome. The team noted that they did not assess the predictive value of TCF in the blood of Sézary patients.
In a multivariable analysis, TCF was still significantly associated with PFS (P<0.001) and OS (P<0.001) in the MF patients.
The researchers also assessed potential prognostic variables in the early stage MF patients and found a TCF greater than 25% was a stronger predictor of PFS than any other established prognostic factor. This includes disease stage, presence of plaques, elevated lactate dehydrogenase, age, large-cell transformation, and CLIPI score.
“In reviewing the results, 2 different patients could have identical-looking skin lesions, but one might have a TCF of 8%, and one would have a TCF of 40%,” Dr Kupper noted. “The latter patient was highly likely to progress—something we would never have been able to predict before this discovery.”
“The TCF was independent of how thick or thin the skin lesions were. Most importantly, compared to all other currently used means of trying to predict which patients would progress, TCF was by far the most sensitive and specific.”
The high-throughput sequencing in this study was performed using ImmunoSEQ, an assay developed by Adaptive Biotechnologies. The company did not sponsor the study, but company employees were involved in the research.
New research suggests DNA sequencing can reveal which patients with early stage mycosis fungoides (MF) will develop aggressive disease.
By sequencing the T-cell receptor beta gene (TCRB) in skin biopsies from MF patients, investigators were able to measure the tumor clone frequency (TCF), or the percentage of all T cells that represent the MF clone.
The researchers found the TCF could predict progression-free survival (PFS) and overall survival (OS).
In fact, for patients with early stage MF, TCF was a stronger predictor of PFS than other established prognostic factors.
“While more work needs to be done, we think this approach has the potential to prospectively identify a subgroup of patients who are destined to develop aggressive, life-threatening disease and treat them in a more aggressive fashion with the intent to better manage and, ideally, cure their cancer,” said Thomas Kupper, MD, of Brigham and Women’s Hospital in Boston, Massachusetts.
“At the same time, we believe we can provide reassurance to patients with a low-risk (low TCF) profile that they are likely to survive indefinitely with conventional conservative therapies. As a physician who has treated this disease for decades, I am excited to be involved with work that so directly and profoundly affects the care and management of these patients.”
Dr Kupper and his colleagues described this work in Science Translational Medicine.
The researchers had previously published a study showing that high-throughput sequencing of the TCRB gene was an accurate way to diagnose cutaneous T-cell lymphoma (CTCL), including MF.
With the current study, the investigators set out to determine if the method could be used to predict progression and survival in patients with CTCL.
The team sequenced TCRB in lesional skin biopsies from 309 CTCL patients, most of whom had MF. The discovery cohort had 208 patients (177 with MF), and the validation cohort had 101 patients (87 with MF).
The sequencing produced a snapshot of the TCRB genes from a large number of cells at the site of the lesion. And the researchers could use this to measure TCF.
The team tested the association of TCF with prognosis in all CTCL patients in the discovery cohort and found a TCF greater than 25% in the skin was significantly associated with reduced PFS (P<0.001) and OS (P<0.001). Results were similar in the validation cohort (P<0.001 for PFS and OS).
The investigators also found that TCF was significantly associated with PFS (P<0.001) and OS (P<0.001) in MF patients but not in patients with Sézary syndrome. The team noted that they did not assess the predictive value of TCF in the blood of Sézary patients.
In a multivariable analysis, TCF was still significantly associated with PFS (P<0.001) and OS (P<0.001) in the MF patients.
The researchers also assessed potential prognostic variables in the early stage MF patients and found a TCF greater than 25% was a stronger predictor of PFS than any other established prognostic factor. This includes disease stage, presence of plaques, elevated lactate dehydrogenase, age, large-cell transformation, and CLIPI score.
“In reviewing the results, 2 different patients could have identical-looking skin lesions, but one might have a TCF of 8%, and one would have a TCF of 40%,” Dr Kupper noted. “The latter patient was highly likely to progress—something we would never have been able to predict before this discovery.”
“The TCF was independent of how thick or thin the skin lesions were. Most importantly, compared to all other currently used means of trying to predict which patients would progress, TCF was by far the most sensitive and specific.”
The high-throughput sequencing in this study was performed using ImmunoSEQ, an assay developed by Adaptive Biotechnologies. The company did not sponsor the study, but company employees were involved in the research.
‘Long-term benefits’ with nivolumab in cHL
Nivolumab can provide “long-term benefits” in patients with relapsed or refractory classical Hodgkin lymphoma (cHL), according to researchers.
Extended follow-up of the CheckMate-205 study showed that nearly 70% of patients responded to nivolumab.
The median duration of response was 17 months, and the median progression-free survival (PFS) was 15 months.
The most common drug-related AEs were fatigue, diarrhea, infusion-related reactions, rash, nausea, and pruritus.
Philippe Armand, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and his colleagues reported these results in the Journal of Clinical Oncology.
The study was sponsored by Bristol-Myers Squibb Company.
Patients
This phase 2 trial enrolled 243 adults with relapsed or refractory cHL who had undergone autologous hematopoietic stem cell transplant (auto-HSCT).
Their median age was 34 (range, 26-46), and 58% were male. Fifty-seven percent had stage IV disease, 20% had stage III, 21% had stage II, and 2% had stage I.
Patients had received a median of 4 prior therapies (range, 3-5). The median time from diagnosis to first nivolumab dose was 4.5 years (range, 2.4-7.6), and the median time from most recent auto-HSCT to first nivolumab dose was 2.0 years (range, 0.9-4.9).
The researchers divided patients into 3 cohorts according to exposure to brentuximab vedotin (BV):
- Cohort A was BV-naïve (n=63)
- Cohort B received BV only after auto-HSCT (n=80)
- Cohort C received BV before and/or after auto-HSCT (n=100).
Baseline characteristics were generally similar across the cohorts. However, cohort A had fewer prior treatments (median of 2 vs 4 in cohorts B and C).
Cohort B had the longest interval between diagnosis and first nivolumab dose (6.2 years vs 3.1 in cohort A and 3.5 in C) and between most recent auto-HSCT and first nivolumab dose (3.4 years vs 1 in cohort A and 1.7 in C).
Treatment
All patients received nivolumab at 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
In cohort C, patients who were in complete response (CR) for 1 year had to discontinue nivolumab, but they could resume treatment if they relapsed within 2 years.
A protocol amendment allowed patients to continue treatment despite progression if they had stable performance status and were deriving “perceived clinical benefit.”
At a median follow-up of 18 months, 40% percent of all patients were still on treatment, including 48% of patients in cohort A, 40% in cohort B, and 35% in cohort C.
Safety
The most common drug-related AEs were fatigue (23%), diarrhea (15%), infusion-related reactions (14%), rash (12%), nausea (10%), and pruritus (10%).
The most common grade 3/4 drug-related AEs were lipase increase (5%), neutropenia (3%), ALT increase (2%), AST increase (2%), and amylase increase (2%).
The most common serious drug-related AEs were infusion-related reactions (2%), pneumonitis (1%), pneumonia (1%), pleural effusion (1%), and pyrexia (1%).
Seven percent of patients discontinued treatment due to AEs. The most common of these were pneumonitis (2%) and autoimmune hepatitis (1%).
There were no deaths due to drug-related AEs.
Efficacy
The objective response rate was 69% overall, 65% in cohort A, 68% in cohort B, and 73% in cohort C.
CR was the best response for 16% of all patients, 29% of cohort A, 13% of cohort B, and 12% of cohort C.
The median duration of response was 17 months overall, 20 months for cohort A, 16 months for cohort B, 15 months for cohort C, and 20 months for patients who achieved a CR.
The researchers said responses were similar irrespective of BV treatment sequence.
The median PFS was 15 months for all patients, 18 months for cohort A, 15 months for cohort B, 12 months for cohort C, and 22 months for patients who achieved a CR.
The median overall survival was not reached in any of the cohorts.
The 12-month overall survival was 92% overall, 93% in cohort A, 95% in cohort B, 90% in cohort C, and 100% in patients who achieved a CR.
Subsequent HSCT
Forty-four patients proceeded to allogeneic HSCT after nivolumab, and the median post-HSCT follow-up was 5.5 months (range, 2.9-11.8).
At 6 months, the rate of transplant-related mortality was 13%, and the rate of disease progression was 7%.
The rate of grade 2-4 acute graft-vs-host disease (GVHD) was 30%, the rate of grade 3-4 acute GVHD was 20%, and the rate of chronic GVHD was 15%.
Nivolumab can provide “long-term benefits” in patients with relapsed or refractory classical Hodgkin lymphoma (cHL), according to researchers.
Extended follow-up of the CheckMate-205 study showed that nearly 70% of patients responded to nivolumab.
The median duration of response was 17 months, and the median progression-free survival (PFS) was 15 months.
The most common drug-related AEs were fatigue, diarrhea, infusion-related reactions, rash, nausea, and pruritus.
Philippe Armand, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and his colleagues reported these results in the Journal of Clinical Oncology.
The study was sponsored by Bristol-Myers Squibb Company.
Patients
This phase 2 trial enrolled 243 adults with relapsed or refractory cHL who had undergone autologous hematopoietic stem cell transplant (auto-HSCT).
Their median age was 34 (range, 26-46), and 58% were male. Fifty-seven percent had stage IV disease, 20% had stage III, 21% had stage II, and 2% had stage I.
Patients had received a median of 4 prior therapies (range, 3-5). The median time from diagnosis to first nivolumab dose was 4.5 years (range, 2.4-7.6), and the median time from most recent auto-HSCT to first nivolumab dose was 2.0 years (range, 0.9-4.9).
The researchers divided patients into 3 cohorts according to exposure to brentuximab vedotin (BV):
- Cohort A was BV-naïve (n=63)
- Cohort B received BV only after auto-HSCT (n=80)
- Cohort C received BV before and/or after auto-HSCT (n=100).
Baseline characteristics were generally similar across the cohorts. However, cohort A had fewer prior treatments (median of 2 vs 4 in cohorts B and C).
Cohort B had the longest interval between diagnosis and first nivolumab dose (6.2 years vs 3.1 in cohort A and 3.5 in C) and between most recent auto-HSCT and first nivolumab dose (3.4 years vs 1 in cohort A and 1.7 in C).
Treatment
All patients received nivolumab at 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
In cohort C, patients who were in complete response (CR) for 1 year had to discontinue nivolumab, but they could resume treatment if they relapsed within 2 years.
A protocol amendment allowed patients to continue treatment despite progression if they had stable performance status and were deriving “perceived clinical benefit.”
At a median follow-up of 18 months, 40% percent of all patients were still on treatment, including 48% of patients in cohort A, 40% in cohort B, and 35% in cohort C.
Safety
The most common drug-related AEs were fatigue (23%), diarrhea (15%), infusion-related reactions (14%), rash (12%), nausea (10%), and pruritus (10%).
The most common grade 3/4 drug-related AEs were lipase increase (5%), neutropenia (3%), ALT increase (2%), AST increase (2%), and amylase increase (2%).
The most common serious drug-related AEs were infusion-related reactions (2%), pneumonitis (1%), pneumonia (1%), pleural effusion (1%), and pyrexia (1%).
Seven percent of patients discontinued treatment due to AEs. The most common of these were pneumonitis (2%) and autoimmune hepatitis (1%).
There were no deaths due to drug-related AEs.
Efficacy
The objective response rate was 69% overall, 65% in cohort A, 68% in cohort B, and 73% in cohort C.
CR was the best response for 16% of all patients, 29% of cohort A, 13% of cohort B, and 12% of cohort C.
The median duration of response was 17 months overall, 20 months for cohort A, 16 months for cohort B, 15 months for cohort C, and 20 months for patients who achieved a CR.
The researchers said responses were similar irrespective of BV treatment sequence.
The median PFS was 15 months for all patients, 18 months for cohort A, 15 months for cohort B, 12 months for cohort C, and 22 months for patients who achieved a CR.
The median overall survival was not reached in any of the cohorts.
The 12-month overall survival was 92% overall, 93% in cohort A, 95% in cohort B, 90% in cohort C, and 100% in patients who achieved a CR.
Subsequent HSCT
Forty-four patients proceeded to allogeneic HSCT after nivolumab, and the median post-HSCT follow-up was 5.5 months (range, 2.9-11.8).
At 6 months, the rate of transplant-related mortality was 13%, and the rate of disease progression was 7%.
The rate of grade 2-4 acute graft-vs-host disease (GVHD) was 30%, the rate of grade 3-4 acute GVHD was 20%, and the rate of chronic GVHD was 15%.
Nivolumab can provide “long-term benefits” in patients with relapsed or refractory classical Hodgkin lymphoma (cHL), according to researchers.
Extended follow-up of the CheckMate-205 study showed that nearly 70% of patients responded to nivolumab.
The median duration of response was 17 months, and the median progression-free survival (PFS) was 15 months.
The most common drug-related AEs were fatigue, diarrhea, infusion-related reactions, rash, nausea, and pruritus.
Philippe Armand, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and his colleagues reported these results in the Journal of Clinical Oncology.
The study was sponsored by Bristol-Myers Squibb Company.
Patients
This phase 2 trial enrolled 243 adults with relapsed or refractory cHL who had undergone autologous hematopoietic stem cell transplant (auto-HSCT).
Their median age was 34 (range, 26-46), and 58% were male. Fifty-seven percent had stage IV disease, 20% had stage III, 21% had stage II, and 2% had stage I.
Patients had received a median of 4 prior therapies (range, 3-5). The median time from diagnosis to first nivolumab dose was 4.5 years (range, 2.4-7.6), and the median time from most recent auto-HSCT to first nivolumab dose was 2.0 years (range, 0.9-4.9).
The researchers divided patients into 3 cohorts according to exposure to brentuximab vedotin (BV):
- Cohort A was BV-naïve (n=63)
- Cohort B received BV only after auto-HSCT (n=80)
- Cohort C received BV before and/or after auto-HSCT (n=100).
Baseline characteristics were generally similar across the cohorts. However, cohort A had fewer prior treatments (median of 2 vs 4 in cohorts B and C).
Cohort B had the longest interval between diagnosis and first nivolumab dose (6.2 years vs 3.1 in cohort A and 3.5 in C) and between most recent auto-HSCT and first nivolumab dose (3.4 years vs 1 in cohort A and 1.7 in C).
Treatment
All patients received nivolumab at 3 mg/kg once every 2 weeks until disease progression or unacceptable toxicity.
In cohort C, patients who were in complete response (CR) for 1 year had to discontinue nivolumab, but they could resume treatment if they relapsed within 2 years.
A protocol amendment allowed patients to continue treatment despite progression if they had stable performance status and were deriving “perceived clinical benefit.”
At a median follow-up of 18 months, 40% percent of all patients were still on treatment, including 48% of patients in cohort A, 40% in cohort B, and 35% in cohort C.
Safety
The most common drug-related AEs were fatigue (23%), diarrhea (15%), infusion-related reactions (14%), rash (12%), nausea (10%), and pruritus (10%).
The most common grade 3/4 drug-related AEs were lipase increase (5%), neutropenia (3%), ALT increase (2%), AST increase (2%), and amylase increase (2%).
The most common serious drug-related AEs were infusion-related reactions (2%), pneumonitis (1%), pneumonia (1%), pleural effusion (1%), and pyrexia (1%).
Seven percent of patients discontinued treatment due to AEs. The most common of these were pneumonitis (2%) and autoimmune hepatitis (1%).
There were no deaths due to drug-related AEs.
Efficacy
The objective response rate was 69% overall, 65% in cohort A, 68% in cohort B, and 73% in cohort C.
CR was the best response for 16% of all patients, 29% of cohort A, 13% of cohort B, and 12% of cohort C.
The median duration of response was 17 months overall, 20 months for cohort A, 16 months for cohort B, 15 months for cohort C, and 20 months for patients who achieved a CR.
The researchers said responses were similar irrespective of BV treatment sequence.
The median PFS was 15 months for all patients, 18 months for cohort A, 15 months for cohort B, 12 months for cohort C, and 22 months for patients who achieved a CR.
The median overall survival was not reached in any of the cohorts.
The 12-month overall survival was 92% overall, 93% in cohort A, 95% in cohort B, 90% in cohort C, and 100% in patients who achieved a CR.
Subsequent HSCT
Forty-four patients proceeded to allogeneic HSCT after nivolumab, and the median post-HSCT follow-up was 5.5 months (range, 2.9-11.8).
At 6 months, the rate of transplant-related mortality was 13%, and the rate of disease progression was 7%.
The rate of grade 2-4 acute graft-vs-host disease (GVHD) was 30%, the rate of grade 3-4 acute GVHD was 20%, and the rate of chronic GVHD was 15%.