User login
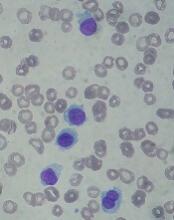
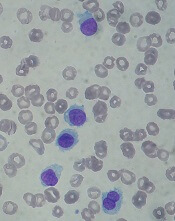
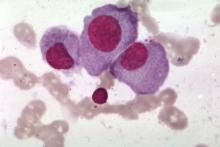
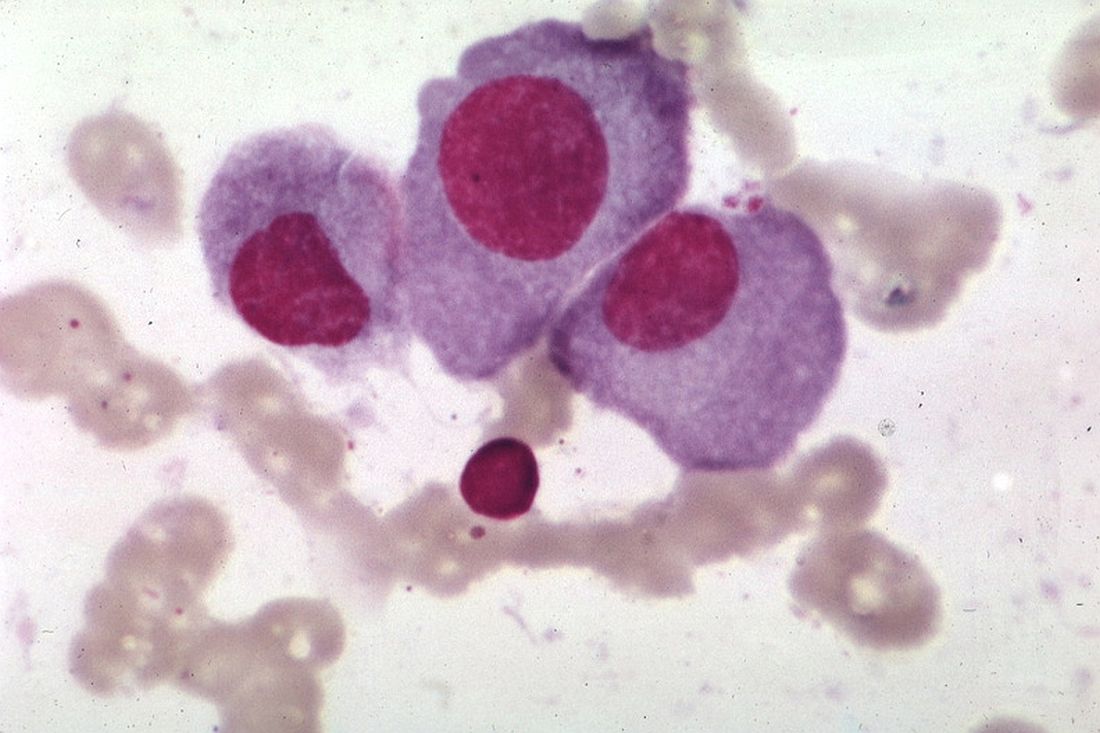
STORM trial shows response in penta-refractory myeloma
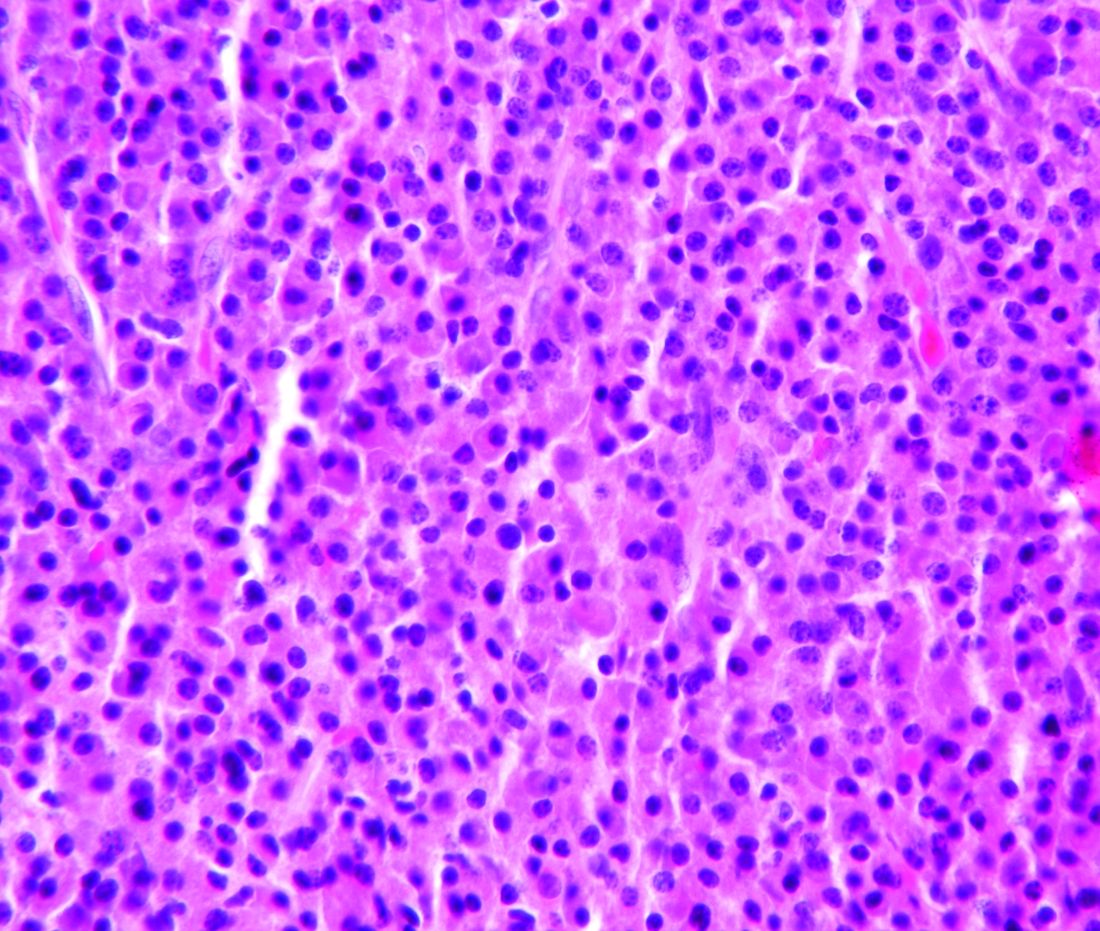
Treatment with selinexor and low-dose dexamethasone can provide a “meaningful clinical benefit” in patients with penta-refractory multiple myeloma, according to the principal investigator of the STORM trial.
Updated results from this phase 2 trial showed that selinexor and low-dose dexamethasone produced an overall response rate of 26.2% and a clinical benefit rate of 39.3%. The median progression-free survival was 3.7 months and the median overall survival was 8.6 months.
The trial’s principal investigator, Sundar Jagannath, MBBS, of the Icahn School of Medicine at Mount Sinai, New York, presented these results at the annual meeting of the Society of Hematologic Oncology.
“The additional phase 2b clinical results… are very encouraging for the patients suffering from penta-refractory multiple myeloma and their families,” Dr. Jagannath said in a statement. “Of particular significance, for the nearly 40% of patients who had a minimal response or better, the median survival was 15.6 months, which provided the opportunity for a meaningful clinical benefit for patients on the STORM [Selinexor Treatment of Refractory Myeloma] study.”
STORM (NCT02336815) included 122 patients with penta-refractory multiple myeloma. They had previously received bortezomib, carfilzomib, lenalidomide, pomalidomide, daratumumab, alkylating agents, and glucocorticoids. Their disease was refractory to glucocorticoids, at least one proteasome inhibitor, at least one immunomodulatory drug, daratumumab, and their most recent therapy.
The patients had received a median of seven prior treatment regimens. Their median age was 65 years, a little more than half were men, and more than half had high-risk cytogenetics. Patients received oral selinexor at 80 mg twice weekly plus dexamethasone at 20 mg twice weekly until disease progression.Two patients (1.6%) achieved stringent complete responses. They also had minimal residual disease negativity, one at the level of 1 x 10–6 and one at 1 x 10–4.
Very good partial responses were seen in 4.9% of patients, 19.7% had partial responses, 13.1% had minimal responses (MRs), and 39.3% had stable disease. Progressive disease occurred in 13.1% of patients; 8.2% were not evaluable for response.
The overall response rate (partial response or better) was 26.2%, the clinical benefit rate (MR or better) was 39.3%, and the disease control rate (stable disease or better) was 78.7%.
The median duration of response was 4.4 months. The median progression-free survival was 3.7 months overall, 4.6 months in patients with an MR or better, and 1.1 months in patients who had progressive disease or were not evaluable.
The median overall survival was 8.6 months for the entire cohort. Overall survival was 15.6 months in patients with an MR or better and 1.7 months in patients who had progressive disease or were not evaluable (P less than .0001).
The “most important” grade 3/4 adverse events, according to Dr. Jagannath, were thrombocytopenia (53.7%), anemia (29.3%), fatigue (22.8%), hyponatremia (16.3%), nausea (9.8%), diarrhea (6.5%), anorexia (3.3%), and emesis (3.3%). A total of 23 patients (19.5%) discontinued treatment because of a related adverse.
This study was sponsored by Karyopharm Therapeutics. Dr. Jagannath reported relationships with Karyopharm, Janssen, Celgene, Amgen, and GlaxoSmithKline.
SOURCE: Jagannath S et al. SOHO 2018, Abstract MM-255
Treatment with selinexor and low-dose dexamethasone can provide a “meaningful clinical benefit” in patients with penta-refractory multiple myeloma, according to the principal investigator of the STORM trial.
Updated results from this phase 2 trial showed that selinexor and low-dose dexamethasone produced an overall response rate of 26.2% and a clinical benefit rate of 39.3%. The median progression-free survival was 3.7 months and the median overall survival was 8.6 months.
The trial’s principal investigator, Sundar Jagannath, MBBS, of the Icahn School of Medicine at Mount Sinai, New York, presented these results at the annual meeting of the Society of Hematologic Oncology.
“The additional phase 2b clinical results… are very encouraging for the patients suffering from penta-refractory multiple myeloma and their families,” Dr. Jagannath said in a statement. “Of particular significance, for the nearly 40% of patients who had a minimal response or better, the median survival was 15.6 months, which provided the opportunity for a meaningful clinical benefit for patients on the STORM [Selinexor Treatment of Refractory Myeloma] study.”
STORM (NCT02336815) included 122 patients with penta-refractory multiple myeloma. They had previously received bortezomib, carfilzomib, lenalidomide, pomalidomide, daratumumab, alkylating agents, and glucocorticoids. Their disease was refractory to glucocorticoids, at least one proteasome inhibitor, at least one immunomodulatory drug, daratumumab, and their most recent therapy.
The patients had received a median of seven prior treatment regimens. Their median age was 65 years, a little more than half were men, and more than half had high-risk cytogenetics. Patients received oral selinexor at 80 mg twice weekly plus dexamethasone at 20 mg twice weekly until disease progression.Two patients (1.6%) achieved stringent complete responses. They also had minimal residual disease negativity, one at the level of 1 x 10–6 and one at 1 x 10–4.
Very good partial responses were seen in 4.9% of patients, 19.7% had partial responses, 13.1% had minimal responses (MRs), and 39.3% had stable disease. Progressive disease occurred in 13.1% of patients; 8.2% were not evaluable for response.
The overall response rate (partial response or better) was 26.2%, the clinical benefit rate (MR or better) was 39.3%, and the disease control rate (stable disease or better) was 78.7%.
The median duration of response was 4.4 months. The median progression-free survival was 3.7 months overall, 4.6 months in patients with an MR or better, and 1.1 months in patients who had progressive disease or were not evaluable.
The median overall survival was 8.6 months for the entire cohort. Overall survival was 15.6 months in patients with an MR or better and 1.7 months in patients who had progressive disease or were not evaluable (P less than .0001).
The “most important” grade 3/4 adverse events, according to Dr. Jagannath, were thrombocytopenia (53.7%), anemia (29.3%), fatigue (22.8%), hyponatremia (16.3%), nausea (9.8%), diarrhea (6.5%), anorexia (3.3%), and emesis (3.3%). A total of 23 patients (19.5%) discontinued treatment because of a related adverse.
This study was sponsored by Karyopharm Therapeutics. Dr. Jagannath reported relationships with Karyopharm, Janssen, Celgene, Amgen, and GlaxoSmithKline.
SOURCE: Jagannath S et al. SOHO 2018, Abstract MM-255
Treatment with selinexor and low-dose dexamethasone can provide a “meaningful clinical benefit” in patients with penta-refractory multiple myeloma, according to the principal investigator of the STORM trial.
Updated results from this phase 2 trial showed that selinexor and low-dose dexamethasone produced an overall response rate of 26.2% and a clinical benefit rate of 39.3%. The median progression-free survival was 3.7 months and the median overall survival was 8.6 months.
The trial’s principal investigator, Sundar Jagannath, MBBS, of the Icahn School of Medicine at Mount Sinai, New York, presented these results at the annual meeting of the Society of Hematologic Oncology.
“The additional phase 2b clinical results… are very encouraging for the patients suffering from penta-refractory multiple myeloma and their families,” Dr. Jagannath said in a statement. “Of particular significance, for the nearly 40% of patients who had a minimal response or better, the median survival was 15.6 months, which provided the opportunity for a meaningful clinical benefit for patients on the STORM [Selinexor Treatment of Refractory Myeloma] study.”
STORM (NCT02336815) included 122 patients with penta-refractory multiple myeloma. They had previously received bortezomib, carfilzomib, lenalidomide, pomalidomide, daratumumab, alkylating agents, and glucocorticoids. Their disease was refractory to glucocorticoids, at least one proteasome inhibitor, at least one immunomodulatory drug, daratumumab, and their most recent therapy.
The patients had received a median of seven prior treatment regimens. Their median age was 65 years, a little more than half were men, and more than half had high-risk cytogenetics. Patients received oral selinexor at 80 mg twice weekly plus dexamethasone at 20 mg twice weekly until disease progression.Two patients (1.6%) achieved stringent complete responses. They also had minimal residual disease negativity, one at the level of 1 x 10–6 and one at 1 x 10–4.
Very good partial responses were seen in 4.9% of patients, 19.7% had partial responses, 13.1% had minimal responses (MRs), and 39.3% had stable disease. Progressive disease occurred in 13.1% of patients; 8.2% were not evaluable for response.
The overall response rate (partial response or better) was 26.2%, the clinical benefit rate (MR or better) was 39.3%, and the disease control rate (stable disease or better) was 78.7%.
The median duration of response was 4.4 months. The median progression-free survival was 3.7 months overall, 4.6 months in patients with an MR or better, and 1.1 months in patients who had progressive disease or were not evaluable.
The median overall survival was 8.6 months for the entire cohort. Overall survival was 15.6 months in patients with an MR or better and 1.7 months in patients who had progressive disease or were not evaluable (P less than .0001).
The “most important” grade 3/4 adverse events, according to Dr. Jagannath, were thrombocytopenia (53.7%), anemia (29.3%), fatigue (22.8%), hyponatremia (16.3%), nausea (9.8%), diarrhea (6.5%), anorexia (3.3%), and emesis (3.3%). A total of 23 patients (19.5%) discontinued treatment because of a related adverse.
This study was sponsored by Karyopharm Therapeutics. Dr. Jagannath reported relationships with Karyopharm, Janssen, Celgene, Amgen, and GlaxoSmithKline.
SOURCE: Jagannath S et al. SOHO 2018, Abstract MM-255
FROM SOHO 2018
Key clinical point:
Major finding: The overall response rate was 26.2% and the clinical benefit rate was 39.3%.
Study details: A phase 2 trial of 122 patients with penta-refractory multiple myeloma.
Disclosures: This study was sponsored by Karyopharm Therapeutics. Dr. Jagannath reported relationships with Karyopharm, Janssen, Celgene, Amgen, and GlaxoSmithKline.
Source: Jagannath S et al. SOHO 2018, Abstract MM-255.
Be wary of watchful waiting in follicular lymphoma
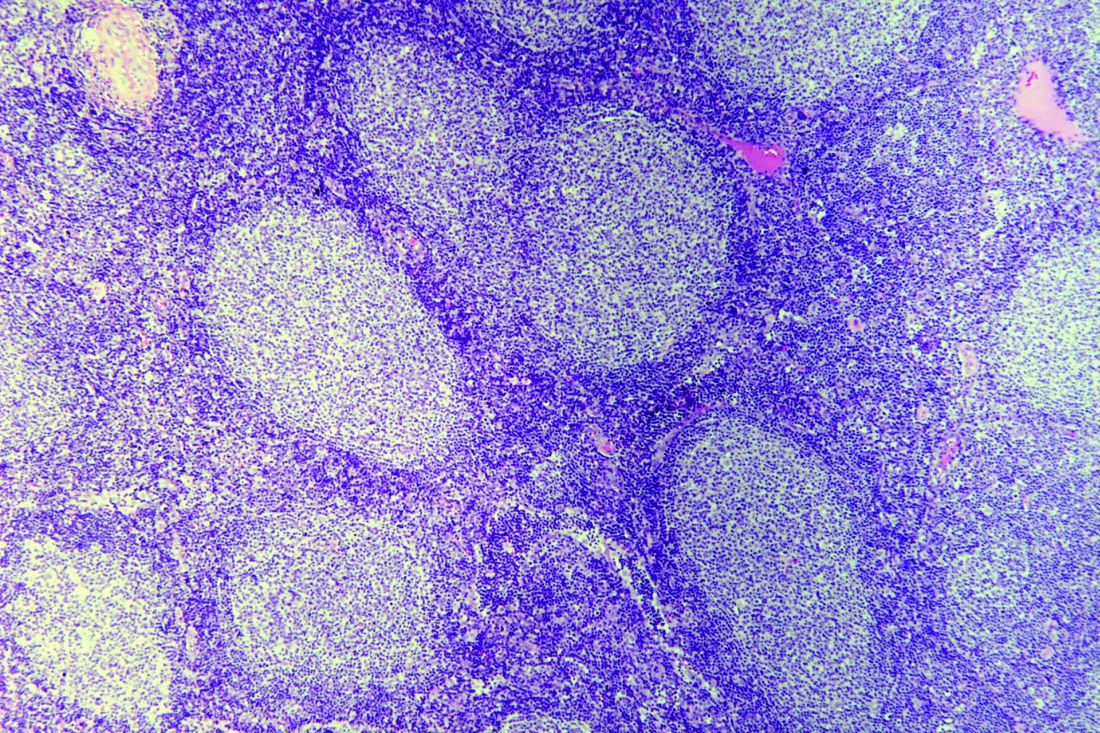
A substantial proportion of patients with follicular lymphoma managed with watchful waiting develop organ dysfunction or transformation that may negatively impact survival outcomes, results of a retrospective study suggest.
About one-quarter of patients managed with watchful waiting developed significant organ dysfunction or transformation at first progression over 8.2 years of follow-up.
Organ dysfunction and transformation were associated with significantly worse overall survival that could not be predicted based on baseline characteristics, the study authors reported in Clinical Lymphoma, Myeloma & Leukemia.
The study confirmed certain benefits of watchful waiting, including a low risk of progression and an “excellent” rate of overall survival, the investigators said.
However, the substantial rate of organ dysfunction and transformation in a subset of patients is “clinically meaningful for informed decision making,” reported Gwynivere A. Davies, MD, of the University of Calgary (Alta.), and her coauthors.
“While consenting patients to initial [watchful waiting], patients need to be informed about the risk for these adverse events, as well as receiving education and the need for close monitoring regarding symptoms that may indicate serious progression events,” Dr. Davies and her coauthors wrote.
Alternatively, rituximab chemotherapy, with or without rituximab maintenance, might be warranted for watchful waiting patients with clear disease progression before organ dysfunction or transformation events, despite not meeting high-tumor burden therapy indications.
The retrospective study included data from the Alberta Lymphoma Database on patients with grade 1-3a follicular lymphoma aged 18-70 years who were diagnosed between 1994 and 2011. Investigators identified 238 patients initially managed with watchful waiting, with a median age of 54.1 years at diagnosis. More than 80% were advanced stage.
Only 71% of these patients progressed, with a median time to progression of about 30 months and a 10-year survival rate from diagnosis of 81.2%, investigators said. However, 58 patients (24.4%) had organ dysfunction or transformation at the time of progression.
Those adverse outcomes significantly affected overall survival. The 10-year overall survival was 65.4% for patients with transformation at progression versus 83.2% for those without (P = .0017). Likewise, 10-year overall survival was 71.5% and 82.7%, respectively, for those with organ dysfunction at progression and those without (P = .028).
Investigators also looked at a comparison group of 236 follicular lymphoma patients managed with immediate rituximab chemotherapy. They found survival outcomes in that group were similar to those in the subgroup of 56 watchful waiting patients who received primarily rituximab-containing regimens at the time of organ dysfunction or transformation.
Taken together, the findings suggest management changes may be warranted for follicular lymphoma patients managed according to a watchful waiting strategy, the investigators wrote. “Consideration should be given to implementing standardized follow-up imaging, with early initiation of rituximab-based therapy if there is evidence of progression in an attempt to prevent these potentially clinically impactful events.”
Dr. Davies reported having no financial disclosures. Study coauthors reported disclosures related to Janssen, Gilead Sciences, Lundbeck, Roche, AbbVie, Amgen, Seattle Genetics, Bristol-Myers Squibb, Servier Laboratories, and Merck.
SOURCE: Davies GA et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 28. doi: 10.1016/j.clml.2018.08.015.
A substantial proportion of patients with follicular lymphoma managed with watchful waiting develop organ dysfunction or transformation that may negatively impact survival outcomes, results of a retrospective study suggest.
About one-quarter of patients managed with watchful waiting developed significant organ dysfunction or transformation at first progression over 8.2 years of follow-up.
Organ dysfunction and transformation were associated with significantly worse overall survival that could not be predicted based on baseline characteristics, the study authors reported in Clinical Lymphoma, Myeloma & Leukemia.
The study confirmed certain benefits of watchful waiting, including a low risk of progression and an “excellent” rate of overall survival, the investigators said.
However, the substantial rate of organ dysfunction and transformation in a subset of patients is “clinically meaningful for informed decision making,” reported Gwynivere A. Davies, MD, of the University of Calgary (Alta.), and her coauthors.
“While consenting patients to initial [watchful waiting], patients need to be informed about the risk for these adverse events, as well as receiving education and the need for close monitoring regarding symptoms that may indicate serious progression events,” Dr. Davies and her coauthors wrote.
Alternatively, rituximab chemotherapy, with or without rituximab maintenance, might be warranted for watchful waiting patients with clear disease progression before organ dysfunction or transformation events, despite not meeting high-tumor burden therapy indications.
The retrospective study included data from the Alberta Lymphoma Database on patients with grade 1-3a follicular lymphoma aged 18-70 years who were diagnosed between 1994 and 2011. Investigators identified 238 patients initially managed with watchful waiting, with a median age of 54.1 years at diagnosis. More than 80% were advanced stage.
Only 71% of these patients progressed, with a median time to progression of about 30 months and a 10-year survival rate from diagnosis of 81.2%, investigators said. However, 58 patients (24.4%) had organ dysfunction or transformation at the time of progression.
Those adverse outcomes significantly affected overall survival. The 10-year overall survival was 65.4% for patients with transformation at progression versus 83.2% for those without (P = .0017). Likewise, 10-year overall survival was 71.5% and 82.7%, respectively, for those with organ dysfunction at progression and those without (P = .028).
Investigators also looked at a comparison group of 236 follicular lymphoma patients managed with immediate rituximab chemotherapy. They found survival outcomes in that group were similar to those in the subgroup of 56 watchful waiting patients who received primarily rituximab-containing regimens at the time of organ dysfunction or transformation.
Taken together, the findings suggest management changes may be warranted for follicular lymphoma patients managed according to a watchful waiting strategy, the investigators wrote. “Consideration should be given to implementing standardized follow-up imaging, with early initiation of rituximab-based therapy if there is evidence of progression in an attempt to prevent these potentially clinically impactful events.”
Dr. Davies reported having no financial disclosures. Study coauthors reported disclosures related to Janssen, Gilead Sciences, Lundbeck, Roche, AbbVie, Amgen, Seattle Genetics, Bristol-Myers Squibb, Servier Laboratories, and Merck.
SOURCE: Davies GA et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 28. doi: 10.1016/j.clml.2018.08.015.
A substantial proportion of patients with follicular lymphoma managed with watchful waiting develop organ dysfunction or transformation that may negatively impact survival outcomes, results of a retrospective study suggest.
About one-quarter of patients managed with watchful waiting developed significant organ dysfunction or transformation at first progression over 8.2 years of follow-up.
Organ dysfunction and transformation were associated with significantly worse overall survival that could not be predicted based on baseline characteristics, the study authors reported in Clinical Lymphoma, Myeloma & Leukemia.
The study confirmed certain benefits of watchful waiting, including a low risk of progression and an “excellent” rate of overall survival, the investigators said.
However, the substantial rate of organ dysfunction and transformation in a subset of patients is “clinically meaningful for informed decision making,” reported Gwynivere A. Davies, MD, of the University of Calgary (Alta.), and her coauthors.
“While consenting patients to initial [watchful waiting], patients need to be informed about the risk for these adverse events, as well as receiving education and the need for close monitoring regarding symptoms that may indicate serious progression events,” Dr. Davies and her coauthors wrote.
Alternatively, rituximab chemotherapy, with or without rituximab maintenance, might be warranted for watchful waiting patients with clear disease progression before organ dysfunction or transformation events, despite not meeting high-tumor burden therapy indications.
The retrospective study included data from the Alberta Lymphoma Database on patients with grade 1-3a follicular lymphoma aged 18-70 years who were diagnosed between 1994 and 2011. Investigators identified 238 patients initially managed with watchful waiting, with a median age of 54.1 years at diagnosis. More than 80% were advanced stage.
Only 71% of these patients progressed, with a median time to progression of about 30 months and a 10-year survival rate from diagnosis of 81.2%, investigators said. However, 58 patients (24.4%) had organ dysfunction or transformation at the time of progression.
Those adverse outcomes significantly affected overall survival. The 10-year overall survival was 65.4% for patients with transformation at progression versus 83.2% for those without (P = .0017). Likewise, 10-year overall survival was 71.5% and 82.7%, respectively, for those with organ dysfunction at progression and those without (P = .028).
Investigators also looked at a comparison group of 236 follicular lymphoma patients managed with immediate rituximab chemotherapy. They found survival outcomes in that group were similar to those in the subgroup of 56 watchful waiting patients who received primarily rituximab-containing regimens at the time of organ dysfunction or transformation.
Taken together, the findings suggest management changes may be warranted for follicular lymphoma patients managed according to a watchful waiting strategy, the investigators wrote. “Consideration should be given to implementing standardized follow-up imaging, with early initiation of rituximab-based therapy if there is evidence of progression in an attempt to prevent these potentially clinically impactful events.”
Dr. Davies reported having no financial disclosures. Study coauthors reported disclosures related to Janssen, Gilead Sciences, Lundbeck, Roche, AbbVie, Amgen, Seattle Genetics, Bristol-Myers Squibb, Servier Laboratories, and Merck.
SOURCE: Davies GA et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 28. doi: 10.1016/j.clml.2018.08.015.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA
Key clinical point:
Major finding: A total of 58 patients (24.4%) had organ dysfunction or transformation at the time of progression and had worse survival outcomes, compared with patients who did not experience those events.
Study details: A retrospective study including data on 238 patients with grade 1-3a follicular lymphoma aged 18-70 years who were managed with watchful waiting.
Disclosures: Study authors reported disclosures related to Janssen, Gilead Sciences, Lundbeck, Roche, AbbVie, Amgen, Seattle Genetics, Bristol-Myers Squibb, Servier Laboratories, and Merck.
Source: Davies GA et al. Clin Lymphoma Myeloma Leuk. 2018 Aug 28. doi: 10.1016/j.clml.2018.08.015.
Pruritus linked to wide variety of cancers
A wide variety of hematologic, dermatologic, and solid organ malignancies are associated with pruritus, a large, single-center, retrospective study suggests.
Blacks with pruritus had a higher odds ratio of hematologic malignancies, among others, while whites had higher likelihood of liver, gastrointestinal, respiratory and gynecologic cancers, results of the study show.
The results by race help address a gap in the literature, according to Shawn G. Kwatra, MD, of Johns Hopkins University, Baltimore, and his coinvestigators.
“Little is known about the association between pruritus and malignancy among different ethnic groups,” Dr. Kwatra and his coauthors wrote in the Journal of the American Academy of Dermatology.
The study shows a stronger association with more types of malignancies than has been reported previously, according to the investigators.
“The main difference is that prior studies focused on diagnosis of malignancy after the onset of pruritus, while our study includes malignancies diagnosed on or after pruritus onset,” they wrote.
Retrospective data for the study, which came from the Johns Hopkins Health System, included 16,925 patients aged 18 years or older who presented with itching or pruritus between April 4, 2013 and Dec. 31, 2017.
Of those 16,925 patients, 2,903 were also diagnosed with a concomitant malignancy during that time period. Compared with patients with no itching diagnosis during that time period, the pruritus patients more likely to have a concomitant malignancy, with an OR of 5.76 (95% confidence interval, 5.53-6.00), Dr. Kwatra and his colleagues found.
Malignancies most strongly associated with pruritus included those of the skin, liver, gallbladder and biliary tract, and hematopoietic system.
Among hematologic malignancies, pruritus was most strongly linked to myeloid leukemia and primary cutaneous lymphoma, while among skin cancers, squamous cell carcinoma was most strongly linked.
Whites had higher odds of any malignancy versus blacks, according to investigators, with ORs of 6.12 (95% CI, 5.81-6.46) and 5.61 (95% CI, 5.21-6.04), respectively.
Blacks with pruritus had higher ORs for hematologic and soft tissue malignancies including those of the muscle, fat, and peripheral nerve, investigators said, while whites had higher ORs for skin and liver malignancies.
The investigators also looked at the prevalence of skin eruptions in patients with pruritus and malignancy. “Eruption is variable by malignancy type and points to differing underlying mechanisms of pruritus,” they reported.
The highest rates of skin eruption were in patients with myeloid leukemia at 66%, followed by bone cancers at 58%, lymphocytic leukemia at 57%, multiple myeloma at 53%, and bronchus at 53%. The lowest rates of skin eruption were in patients with gallbladder and biliary tract, colon, pancreas, and liver malignancies.
Dr. Kwatra reported that he is an advisory board member for Menlo Therapeutics and Trevi Therapeutics.
SOURCE: Kwatra SG et al. J Am Acad Dermatol. 2018 Sep 11. doi: 10.1016/j.jaad.2018.08.044.
A wide variety of hematologic, dermatologic, and solid organ malignancies are associated with pruritus, a large, single-center, retrospective study suggests.
Blacks with pruritus had a higher odds ratio of hematologic malignancies, among others, while whites had higher likelihood of liver, gastrointestinal, respiratory and gynecologic cancers, results of the study show.
The results by race help address a gap in the literature, according to Shawn G. Kwatra, MD, of Johns Hopkins University, Baltimore, and his coinvestigators.
“Little is known about the association between pruritus and malignancy among different ethnic groups,” Dr. Kwatra and his coauthors wrote in the Journal of the American Academy of Dermatology.
The study shows a stronger association with more types of malignancies than has been reported previously, according to the investigators.
“The main difference is that prior studies focused on diagnosis of malignancy after the onset of pruritus, while our study includes malignancies diagnosed on or after pruritus onset,” they wrote.
Retrospective data for the study, which came from the Johns Hopkins Health System, included 16,925 patients aged 18 years or older who presented with itching or pruritus between April 4, 2013 and Dec. 31, 2017.
Of those 16,925 patients, 2,903 were also diagnosed with a concomitant malignancy during that time period. Compared with patients with no itching diagnosis during that time period, the pruritus patients more likely to have a concomitant malignancy, with an OR of 5.76 (95% confidence interval, 5.53-6.00), Dr. Kwatra and his colleagues found.
Malignancies most strongly associated with pruritus included those of the skin, liver, gallbladder and biliary tract, and hematopoietic system.
Among hematologic malignancies, pruritus was most strongly linked to myeloid leukemia and primary cutaneous lymphoma, while among skin cancers, squamous cell carcinoma was most strongly linked.
Whites had higher odds of any malignancy versus blacks, according to investigators, with ORs of 6.12 (95% CI, 5.81-6.46) and 5.61 (95% CI, 5.21-6.04), respectively.
Blacks with pruritus had higher ORs for hematologic and soft tissue malignancies including those of the muscle, fat, and peripheral nerve, investigators said, while whites had higher ORs for skin and liver malignancies.
The investigators also looked at the prevalence of skin eruptions in patients with pruritus and malignancy. “Eruption is variable by malignancy type and points to differing underlying mechanisms of pruritus,” they reported.
The highest rates of skin eruption were in patients with myeloid leukemia at 66%, followed by bone cancers at 58%, lymphocytic leukemia at 57%, multiple myeloma at 53%, and bronchus at 53%. The lowest rates of skin eruption were in patients with gallbladder and biliary tract, colon, pancreas, and liver malignancies.
Dr. Kwatra reported that he is an advisory board member for Menlo Therapeutics and Trevi Therapeutics.
SOURCE: Kwatra SG et al. J Am Acad Dermatol. 2018 Sep 11. doi: 10.1016/j.jaad.2018.08.044.
A wide variety of hematologic, dermatologic, and solid organ malignancies are associated with pruritus, a large, single-center, retrospective study suggests.
Blacks with pruritus had a higher odds ratio of hematologic malignancies, among others, while whites had higher likelihood of liver, gastrointestinal, respiratory and gynecologic cancers, results of the study show.
The results by race help address a gap in the literature, according to Shawn G. Kwatra, MD, of Johns Hopkins University, Baltimore, and his coinvestigators.
“Little is known about the association between pruritus and malignancy among different ethnic groups,” Dr. Kwatra and his coauthors wrote in the Journal of the American Academy of Dermatology.
The study shows a stronger association with more types of malignancies than has been reported previously, according to the investigators.
“The main difference is that prior studies focused on diagnosis of malignancy after the onset of pruritus, while our study includes malignancies diagnosed on or after pruritus onset,” they wrote.
Retrospective data for the study, which came from the Johns Hopkins Health System, included 16,925 patients aged 18 years or older who presented with itching or pruritus between April 4, 2013 and Dec. 31, 2017.
Of those 16,925 patients, 2,903 were also diagnosed with a concomitant malignancy during that time period. Compared with patients with no itching diagnosis during that time period, the pruritus patients more likely to have a concomitant malignancy, with an OR of 5.76 (95% confidence interval, 5.53-6.00), Dr. Kwatra and his colleagues found.
Malignancies most strongly associated with pruritus included those of the skin, liver, gallbladder and biliary tract, and hematopoietic system.
Among hematologic malignancies, pruritus was most strongly linked to myeloid leukemia and primary cutaneous lymphoma, while among skin cancers, squamous cell carcinoma was most strongly linked.
Whites had higher odds of any malignancy versus blacks, according to investigators, with ORs of 6.12 (95% CI, 5.81-6.46) and 5.61 (95% CI, 5.21-6.04), respectively.
Blacks with pruritus had higher ORs for hematologic and soft tissue malignancies including those of the muscle, fat, and peripheral nerve, investigators said, while whites had higher ORs for skin and liver malignancies.
The investigators also looked at the prevalence of skin eruptions in patients with pruritus and malignancy. “Eruption is variable by malignancy type and points to differing underlying mechanisms of pruritus,” they reported.
The highest rates of skin eruption were in patients with myeloid leukemia at 66%, followed by bone cancers at 58%, lymphocytic leukemia at 57%, multiple myeloma at 53%, and bronchus at 53%. The lowest rates of skin eruption were in patients with gallbladder and biliary tract, colon, pancreas, and liver malignancies.
Dr. Kwatra reported that he is an advisory board member for Menlo Therapeutics and Trevi Therapeutics.
SOURCE: Kwatra SG et al. J Am Acad Dermatol. 2018 Sep 11. doi: 10.1016/j.jaad.2018.08.044.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point:
Major finding: Blacks with pruritus had higher odds ratios for hematologic and soft tissue malignancies, while whites had higher ORs for skin and liver malignancies.
Study details: A retrospective study of 16,925 adults with itching or pruritus seen at a tertiary care center.
Disclosures: Dr. Kwatra reported serving as an advisory board member for Menlo Therapeutics and Trevi Therapeutics.
Source: Kwatra SG et al. J Am Acad Dermatol. 2018 Sep 11. doi: 10.1016/j.jaad.2018.08.044.
Evaluation of Acute Toxicity in Treating Pelvic Lymph Nodes With Prostate Boost With Hypofractionated Simultaneous Integrated Boost (SIB) Using Volumetric Arc Therapy (VMAT)
Purpose: It is conventional to treat pelvic lymph nodes, followed by prostate boost in a sequential manner, requiring 8-9 weeks to complete therapy. In the last several years there have been several randomized studies of implementing moderate hypofractionated radiotherapy in prostate cancer to shorten the treatment time, which has proven to be non-inferior to conventional treatment. The purpose of this retrospective study is to evaluate the acute toxicities of hypofractionated SIB radiotherapy in treating the lymph nodes with prostate boost.
Methods: Between 2015 and 2017, twenty five high risk prostate cancer patients received pelvic node radiotherapy with prostate boost in 25 fractions with SIB technique with neo-adjuvant and concurrent hormone therapy to 50 Gy/25 fractions at 2 Gy/fraction to pelvic nodes and prostate boost for a total of 67.5 - 75 Gy at 2.7 to 3.0 Gy/fraction. We followed QUNTAC dose-volume constraints for the rectum, bladder and bowel. All these patients received long-term hormone therapy.
Results: The median age was 66 years (range 57-81 years). There were 6 stage II C, 7 III A, 15 III C, and 1 stage IV A. All patients were restaged as per American Joint Committee on Cancer 8th edition. Gleason Score: 6 (1), 7 (4+3) (4), 8 (5), and 9-10 (15). The average PSA was 17.2 ng/mL with a range of 5.6 to 51.92 ng/mL, and average number of positive cores was 74%. These factors put the majority of patients into the very high risk group. The median follow up was 24 months. The majority of the patients tolerated treatment well. Grade 0 genitourinary (GU) toxicity occurred
in 8 (33%) patients, grade II 16 (67%) patients, one patient had a Foley catheter during treatment, a majority of patients were on alpha blockers either before, during or post radiotherapy. Grade 0 gastrointestinal (GI) toxicity occurred in 21 (84%) patients, grade 1 in one, and grade II in 3 (12%) patients. There were no grade 3 or 4 GU or GI toxicities.
Conclusions: Simultaneous integrated boost with VMAT is well tolerated in treating pelvic nodes and prostate boost, without any major acute toxicities. This technique is used in mostly for very high risk localized prostate cancer patients, reducing number of fractions from conventional sequential treatment.
Purpose: It is conventional to treat pelvic lymph nodes, followed by prostate boost in a sequential manner, requiring 8-9 weeks to complete therapy. In the last several years there have been several randomized studies of implementing moderate hypofractionated radiotherapy in prostate cancer to shorten the treatment time, which has proven to be non-inferior to conventional treatment. The purpose of this retrospective study is to evaluate the acute toxicities of hypofractionated SIB radiotherapy in treating the lymph nodes with prostate boost.
Methods: Between 2015 and 2017, twenty five high risk prostate cancer patients received pelvic node radiotherapy with prostate boost in 25 fractions with SIB technique with neo-adjuvant and concurrent hormone therapy to 50 Gy/25 fractions at 2 Gy/fraction to pelvic nodes and prostate boost for a total of 67.5 - 75 Gy at 2.7 to 3.0 Gy/fraction. We followed QUNTAC dose-volume constraints for the rectum, bladder and bowel. All these patients received long-term hormone therapy.
Results: The median age was 66 years (range 57-81 years). There were 6 stage II C, 7 III A, 15 III C, and 1 stage IV A. All patients were restaged as per American Joint Committee on Cancer 8th edition. Gleason Score: 6 (1), 7 (4+3) (4), 8 (5), and 9-10 (15). The average PSA was 17.2 ng/mL with a range of 5.6 to 51.92 ng/mL, and average number of positive cores was 74%. These factors put the majority of patients into the very high risk group. The median follow up was 24 months. The majority of the patients tolerated treatment well. Grade 0 genitourinary (GU) toxicity occurred
in 8 (33%) patients, grade II 16 (67%) patients, one patient had a Foley catheter during treatment, a majority of patients were on alpha blockers either before, during or post radiotherapy. Grade 0 gastrointestinal (GI) toxicity occurred in 21 (84%) patients, grade 1 in one, and grade II in 3 (12%) patients. There were no grade 3 or 4 GU or GI toxicities.
Conclusions: Simultaneous integrated boost with VMAT is well tolerated in treating pelvic nodes and prostate boost, without any major acute toxicities. This technique is used in mostly for very high risk localized prostate cancer patients, reducing number of fractions from conventional sequential treatment.
Purpose: It is conventional to treat pelvic lymph nodes, followed by prostate boost in a sequential manner, requiring 8-9 weeks to complete therapy. In the last several years there have been several randomized studies of implementing moderate hypofractionated radiotherapy in prostate cancer to shorten the treatment time, which has proven to be non-inferior to conventional treatment. The purpose of this retrospective study is to evaluate the acute toxicities of hypofractionated SIB radiotherapy in treating the lymph nodes with prostate boost.
Methods: Between 2015 and 2017, twenty five high risk prostate cancer patients received pelvic node radiotherapy with prostate boost in 25 fractions with SIB technique with neo-adjuvant and concurrent hormone therapy to 50 Gy/25 fractions at 2 Gy/fraction to pelvic nodes and prostate boost for a total of 67.5 - 75 Gy at 2.7 to 3.0 Gy/fraction. We followed QUNTAC dose-volume constraints for the rectum, bladder and bowel. All these patients received long-term hormone therapy.
Results: The median age was 66 years (range 57-81 years). There were 6 stage II C, 7 III A, 15 III C, and 1 stage IV A. All patients were restaged as per American Joint Committee on Cancer 8th edition. Gleason Score: 6 (1), 7 (4+3) (4), 8 (5), and 9-10 (15). The average PSA was 17.2 ng/mL with a range of 5.6 to 51.92 ng/mL, and average number of positive cores was 74%. These factors put the majority of patients into the very high risk group. The median follow up was 24 months. The majority of the patients tolerated treatment well. Grade 0 genitourinary (GU) toxicity occurred
in 8 (33%) patients, grade II 16 (67%) patients, one patient had a Foley catheter during treatment, a majority of patients were on alpha blockers either before, during or post radiotherapy. Grade 0 gastrointestinal (GI) toxicity occurred in 21 (84%) patients, grade 1 in one, and grade II in 3 (12%) patients. There were no grade 3 or 4 GU or GI toxicities.
Conclusions: Simultaneous integrated boost with VMAT is well tolerated in treating pelvic nodes and prostate boost, without any major acute toxicities. This technique is used in mostly for very high risk localized prostate cancer patients, reducing number of fractions from conventional sequential treatment.
A Case of Systemic Mastocytosis With Associated Clonal Hematological Non-Mast Cell Lineage Disease at VA Pittsburgh Healthcare System
Introduction: Systemic mastocytosis (SM) is a rare myeloid neoplasm that is caused by accumulation of abnormal mast cells in the bone marrow, liver, spleen, and skin. The KIT D816V mutation encodes a constitutively activated receptor tyrosine kinase that drives disease pathogenesis. We present a case of systemic mastocytosis with associated clonal hematological non-mast cell disease (SM-AHNMD).
Background: A 71-year-old man presented with anemia, thrombocytopenia, absolute monocyte count of 2,000-4,000 and weight loss in August 2016. A CT showed splenomegaly and lymphadenopathy. Bone marrow biopsy revealed positive CD117 mast cells, CD34 myeloblasts and reticulin fibrosis consistent with SM. Immunohistochemistry confirmed the neoplastic cells were positive for CD25, but negative for CD2. PCR analysis revealed KIT D816V point mutation. Serum tryptase was 295 ug/L (normal 2.2-13.2). He was started on imatinib mesylate. However, his anemia, thrombocytopenia and splenomegaly worsened. He developed bilateral femoral neck fractures in April 2017. Imatinib was discontinued. He underwent bilateral hip hemiarthroplasty. Histology was consistent with SM (positive CD25 and CD117) with dysplastic megakaryocytes and increased monocytosis. By WHO classification he has SM-AHNMD with chronic myelomonocytic leukemia. He was started on cladribine for 4 cycles with good response in splenomegaly, anemia and thrombocytopenia, but he developed leukocytosis. Serum tryptase initially decreased to 141 but then rose to 243. Midostaurin 100 mg orally twice a day was initiated in December 2017. His cytopenia and splenomegaly improved. In March 2018 he was admitted for sigmoid colon obstruction due to inflammation or mass and underwent diverting loop ileostomy. Biopsy could not be performed. His serum tryptase decreased to 178 but increased to 275 in June 2018. He continues on midostaurin.
Discussion: SM-AHNMD constitutes approximately 40% of all SM with poor prognosis. SM is resistance to imatinib because of KIT D816V mutation. Cladribine has some activity. Midostaurin inhibits non-mutant and mutant KIT D816V with 58% response rate and median overall survival of 20 months. Our patient has a good response to both drugs.
Conclusions: Clinicians should be able to diagnose and treat SM. Cladribine and midostaurin are active drugs for SM.
Introduction: Systemic mastocytosis (SM) is a rare myeloid neoplasm that is caused by accumulation of abnormal mast cells in the bone marrow, liver, spleen, and skin. The KIT D816V mutation encodes a constitutively activated receptor tyrosine kinase that drives disease pathogenesis. We present a case of systemic mastocytosis with associated clonal hematological non-mast cell disease (SM-AHNMD).
Background: A 71-year-old man presented with anemia, thrombocytopenia, absolute monocyte count of 2,000-4,000 and weight loss in August 2016. A CT showed splenomegaly and lymphadenopathy. Bone marrow biopsy revealed positive CD117 mast cells, CD34 myeloblasts and reticulin fibrosis consistent with SM. Immunohistochemistry confirmed the neoplastic cells were positive for CD25, but negative for CD2. PCR analysis revealed KIT D816V point mutation. Serum tryptase was 295 ug/L (normal 2.2-13.2). He was started on imatinib mesylate. However, his anemia, thrombocytopenia and splenomegaly worsened. He developed bilateral femoral neck fractures in April 2017. Imatinib was discontinued. He underwent bilateral hip hemiarthroplasty. Histology was consistent with SM (positive CD25 and CD117) with dysplastic megakaryocytes and increased monocytosis. By WHO classification he has SM-AHNMD with chronic myelomonocytic leukemia. He was started on cladribine for 4 cycles with good response in splenomegaly, anemia and thrombocytopenia, but he developed leukocytosis. Serum tryptase initially decreased to 141 but then rose to 243. Midostaurin 100 mg orally twice a day was initiated in December 2017. His cytopenia and splenomegaly improved. In March 2018 he was admitted for sigmoid colon obstruction due to inflammation or mass and underwent diverting loop ileostomy. Biopsy could not be performed. His serum tryptase decreased to 178 but increased to 275 in June 2018. He continues on midostaurin.
Discussion: SM-AHNMD constitutes approximately 40% of all SM with poor prognosis. SM is resistance to imatinib because of KIT D816V mutation. Cladribine has some activity. Midostaurin inhibits non-mutant and mutant KIT D816V with 58% response rate and median overall survival of 20 months. Our patient has a good response to both drugs.
Conclusions: Clinicians should be able to diagnose and treat SM. Cladribine and midostaurin are active drugs for SM.
Introduction: Systemic mastocytosis (SM) is a rare myeloid neoplasm that is caused by accumulation of abnormal mast cells in the bone marrow, liver, spleen, and skin. The KIT D816V mutation encodes a constitutively activated receptor tyrosine kinase that drives disease pathogenesis. We present a case of systemic mastocytosis with associated clonal hematological non-mast cell disease (SM-AHNMD).
Background: A 71-year-old man presented with anemia, thrombocytopenia, absolute monocyte count of 2,000-4,000 and weight loss in August 2016. A CT showed splenomegaly and lymphadenopathy. Bone marrow biopsy revealed positive CD117 mast cells, CD34 myeloblasts and reticulin fibrosis consistent with SM. Immunohistochemistry confirmed the neoplastic cells were positive for CD25, but negative for CD2. PCR analysis revealed KIT D816V point mutation. Serum tryptase was 295 ug/L (normal 2.2-13.2). He was started on imatinib mesylate. However, his anemia, thrombocytopenia and splenomegaly worsened. He developed bilateral femoral neck fractures in April 2017. Imatinib was discontinued. He underwent bilateral hip hemiarthroplasty. Histology was consistent with SM (positive CD25 and CD117) with dysplastic megakaryocytes and increased monocytosis. By WHO classification he has SM-AHNMD with chronic myelomonocytic leukemia. He was started on cladribine for 4 cycles with good response in splenomegaly, anemia and thrombocytopenia, but he developed leukocytosis. Serum tryptase initially decreased to 141 but then rose to 243. Midostaurin 100 mg orally twice a day was initiated in December 2017. His cytopenia and splenomegaly improved. In March 2018 he was admitted for sigmoid colon obstruction due to inflammation or mass and underwent diverting loop ileostomy. Biopsy could not be performed. His serum tryptase decreased to 178 but increased to 275 in June 2018. He continues on midostaurin.
Discussion: SM-AHNMD constitutes approximately 40% of all SM with poor prognosis. SM is resistance to imatinib because of KIT D816V mutation. Cladribine has some activity. Midostaurin inhibits non-mutant and mutant KIT D816V with 58% response rate and median overall survival of 20 months. Our patient has a good response to both drugs.
Conclusions: Clinicians should be able to diagnose and treat SM. Cladribine and midostaurin are active drugs for SM.
Translocation T(11;14): Not Always Mantle Cell Lymphoma!
Background: The translocation t(11;14)(q13;q32) typically considered a hallmark of mantle cell lymphoma(MCL), has also been implicated in some cases of non-MCL lymphoproliferative disorders. Although uncommon, it has been reported in 2-5% of chronic lymphocytic leukemia (CLL) cases. Most of the cases identified have been observed mostly in relapsed CLL. This genetic aberration can be considered a significant prognostic indicator for CLL. t(11;14) positive CLL at the time of diagnosis has been rarely reported. We describe a case of a patient
diagnosed with CLL who was positive for this genetic abnormality.
Case Report: A 64-year-old white male presented with absolute lymphocytosis of 7 years. Lymphocyte immunophenotype detected a CD5(+) CD10(-) CD23(+) CD38(-) CD43(+) FMC7(partial dim) kappa-restricted B-cell population consistent with CLL. CT chest, abdomen, pelvis showed mildly prominent mediastinal and hilar lymph nodes only and was thus classified as Rai stage I. Peripheral FISH came back positive for t(11:14), cyclin D1-IgH translocation. His EPO, Jak2 and BCR-ABL mutation were all negative (done for mild erythrocytosis). Immunoglobulin and SPEP were negative. UPEP showed high Kappa/Lambda ratio. Although this tumor carries t(11;14) (q13;q32) translocation, immunostaining for BCL-1 was negative. It is possible that the gene is not expressed. Based on the staining and the clinical presentation, MCL was excluded. Per NCCN guidelines, patient is receiving clinical monitoring for stage I CLL.
Conclusions: Translocations involving the immunoglobulin genes are commonly identified. Uncommon genomic abnormalities in CLL should be recognized as significant independent predictors of disease progression and survival. It is important to recognize cases of CLL with t(11;14) translocation to achieve risk-adapted treatment strategies, which might be required to treat such patients.
Background: The translocation t(11;14)(q13;q32) typically considered a hallmark of mantle cell lymphoma(MCL), has also been implicated in some cases of non-MCL lymphoproliferative disorders. Although uncommon, it has been reported in 2-5% of chronic lymphocytic leukemia (CLL) cases. Most of the cases identified have been observed mostly in relapsed CLL. This genetic aberration can be considered a significant prognostic indicator for CLL. t(11;14) positive CLL at the time of diagnosis has been rarely reported. We describe a case of a patient
diagnosed with CLL who was positive for this genetic abnormality.
Case Report: A 64-year-old white male presented with absolute lymphocytosis of 7 years. Lymphocyte immunophenotype detected a CD5(+) CD10(-) CD23(+) CD38(-) CD43(+) FMC7(partial dim) kappa-restricted B-cell population consistent with CLL. CT chest, abdomen, pelvis showed mildly prominent mediastinal and hilar lymph nodes only and was thus classified as Rai stage I. Peripheral FISH came back positive for t(11:14), cyclin D1-IgH translocation. His EPO, Jak2 and BCR-ABL mutation were all negative (done for mild erythrocytosis). Immunoglobulin and SPEP were negative. UPEP showed high Kappa/Lambda ratio. Although this tumor carries t(11;14) (q13;q32) translocation, immunostaining for BCL-1 was negative. It is possible that the gene is not expressed. Based on the staining and the clinical presentation, MCL was excluded. Per NCCN guidelines, patient is receiving clinical monitoring for stage I CLL.
Conclusions: Translocations involving the immunoglobulin genes are commonly identified. Uncommon genomic abnormalities in CLL should be recognized as significant independent predictors of disease progression and survival. It is important to recognize cases of CLL with t(11;14) translocation to achieve risk-adapted treatment strategies, which might be required to treat such patients.
Background: The translocation t(11;14)(q13;q32) typically considered a hallmark of mantle cell lymphoma(MCL), has also been implicated in some cases of non-MCL lymphoproliferative disorders. Although uncommon, it has been reported in 2-5% of chronic lymphocytic leukemia (CLL) cases. Most of the cases identified have been observed mostly in relapsed CLL. This genetic aberration can be considered a significant prognostic indicator for CLL. t(11;14) positive CLL at the time of diagnosis has been rarely reported. We describe a case of a patient
diagnosed with CLL who was positive for this genetic abnormality.
Case Report: A 64-year-old white male presented with absolute lymphocytosis of 7 years. Lymphocyte immunophenotype detected a CD5(+) CD10(-) CD23(+) CD38(-) CD43(+) FMC7(partial dim) kappa-restricted B-cell population consistent with CLL. CT chest, abdomen, pelvis showed mildly prominent mediastinal and hilar lymph nodes only and was thus classified as Rai stage I. Peripheral FISH came back positive for t(11:14), cyclin D1-IgH translocation. His EPO, Jak2 and BCR-ABL mutation were all negative (done for mild erythrocytosis). Immunoglobulin and SPEP were negative. UPEP showed high Kappa/Lambda ratio. Although this tumor carries t(11;14) (q13;q32) translocation, immunostaining for BCL-1 was negative. It is possible that the gene is not expressed. Based on the staining and the clinical presentation, MCL was excluded. Per NCCN guidelines, patient is receiving clinical monitoring for stage I CLL.
Conclusions: Translocations involving the immunoglobulin genes are commonly identified. Uncommon genomic abnormalities in CLL should be recognized as significant independent predictors of disease progression and survival. It is important to recognize cases of CLL with t(11;14) translocation to achieve risk-adapted treatment strategies, which might be required to treat such patients.
FDA approves drug for hairy cell leukemia
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
The U.S. Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a Boxed Warning noting that the drug poses risks of capillary leak syndrome (CLS) and hemolytic uremic syndrome (HUS). Treatment with moxetumomab pasudotox should be delayed or discontinued in patients who develop CLS and discontinued in patients with HUS.
The FDA granted the application for moxetumomab pasudotox fast track and priority review designations, and the drug received orphan drug designation from the FDA.
The agency granted the approval of moxetumomab pasudotox to AstraZeneca Pharmaceuticals based on results from a phase 3 trial (NCT01829711).
Data from this study were presented at the 2018 ASCO Annual Meeting (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75% (60/80), the complete response (CR) rate was 41% (33/80), and the durable CR rate was 30% (24/80). Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most frequent treatment-related adverse events (AEs) were nausea (28%), peripheral edema (26%), headache (21%), and pyrexia (20%). Other treatment-related AEs included infections (8%) and neutropenia (3%).
Treatment-related AEs that led to discontinuation included HUS (5%), CLS (3%), and increased blood creatinine (3%).
In all, seven patients (9%) had CLS, and seven (9%) had HUS. This includes four (5%) patients who had both. CLS and HUS proved manageable and reversible.
There were three deaths in this trial, but none of them were considered treatment-related.
FDA approves new hairy cell leukemia drug
The Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a boxed warning noting that the drug poses risks of capillary leak syndrome and hemolytic uremic syndrome. Other serious warnings include the risk of decreased renal function, infusion-related reactions, and electrolyte abnormalities.
The FDA granted the application for moxetumomab pasudotox fast track, priority review, and an orphan drug designation.
The agency approved AstraZeneca’s moxetumomab pasudotox based on results from a phase 3 trial (NCT01829711). Data from this study were presented at the 2018 annual meeting of the American Society of Clinical Oncology (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75%, the complete response (CR) rate was 41%, and the durable CR rate was 30%. Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most common treatment-related adverse events (AEs) were nausea, peripheral edema, headache, and pyrexia. Other treatment-related AEs included infections and neutropenia.
Treatment-related AEs that led to discontinuation included capillary leak syndrome, hemolytic uremic syndrome, and increased blood creatinine.
There were three deaths in this trial, but none of them were considered treatment related.
The Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a boxed warning noting that the drug poses risks of capillary leak syndrome and hemolytic uremic syndrome. Other serious warnings include the risk of decreased renal function, infusion-related reactions, and electrolyte abnormalities.
The FDA granted the application for moxetumomab pasudotox fast track, priority review, and an orphan drug designation.
The agency approved AstraZeneca’s moxetumomab pasudotox based on results from a phase 3 trial (NCT01829711). Data from this study were presented at the 2018 annual meeting of the American Society of Clinical Oncology (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75%, the complete response (CR) rate was 41%, and the durable CR rate was 30%. Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most common treatment-related adverse events (AEs) were nausea, peripheral edema, headache, and pyrexia. Other treatment-related AEs included infections and neutropenia.
Treatment-related AEs that led to discontinuation included capillary leak syndrome, hemolytic uremic syndrome, and increased blood creatinine.
There were three deaths in this trial, but none of them were considered treatment related.
The Food and Drug Administration (FDA) has approved moxetumomab pasudotox-tdfk (Lumoxiti), a CD22-directed cytotoxin, to treat hairy cell leukemia (HCL).
Moxetumomab pasudotox is approved to treat adults with relapsed or refractory HCL who have received at least two prior systemic therapies, including treatment with a purine nucleoside analog.
The prescribing information for moxetumomab pasudotox includes a boxed warning noting that the drug poses risks of capillary leak syndrome and hemolytic uremic syndrome. Other serious warnings include the risk of decreased renal function, infusion-related reactions, and electrolyte abnormalities.
The FDA granted the application for moxetumomab pasudotox fast track, priority review, and an orphan drug designation.
The agency approved AstraZeneca’s moxetumomab pasudotox based on results from a phase 3 trial (NCT01829711). Data from this study were presented at the 2018 annual meeting of the American Society of Clinical Oncology (abstract 7004).
The trial included 80 patients with relapsed or refractory HCL who had received at least two prior lines of therapy.
At a median of 16.7 months of follow-up, the objective response rate was 75%, the complete response (CR) rate was 41%, and the durable CR rate was 30%. Durable CR was defined as CR with hematologic remission for more than 180 days.
Most patients with a CR achieved minimal residual disease negativity (82%; 27/33).
The median duration of response was not reached, nor was the median progression-free survival.
The most common treatment-related adverse events (AEs) were nausea, peripheral edema, headache, and pyrexia. Other treatment-related AEs included infections and neutropenia.
Treatment-related AEs that led to discontinuation included capillary leak syndrome, hemolytic uremic syndrome, and increased blood creatinine.
There were three deaths in this trial, but none of them were considered treatment related.
New U.S. cancer cases may exceed 2.3 million by 2035
The American Association for Cancer Research (AACR) has released its annual Cancer Progress Report, detailing recent advances in the fight against cancer and calling on elected officials to address the challenges that remain.
The AACR Cancer Progress Report 2018 lists the 22 new approvals for cancer treatments that have occurred during the last 12 months, including 12 therapies approved to treat hematologic malignancies.
However, the report also notes that cancer continues to pose immense public health challenges in the United States.
The estimated number of new cancer cases for 2018 is 1,735,350, and the estimated number of cancer deaths is 609,640.
The number of new cancer cases is predicted to increase to 2,387,304 in 2035. This is due, in large part, to the rising number of people age 65 and older, according to the report.
With this in mind, the AACR is calling on elected officials to:
Maintain “robust, sustained, and predictable growth” of the National Institutes of Health (NIH) budget, increasing it at least $2 billion in fiscal year (FY) 2019, for a total funding level of at least $39.1 billion.
Make sure the $711 million in funding provided through the 21st Century Cures Act for targeted initiatives—including the National Cancer Moonshot—“is fully appropriated in FY 2019 and is supplemental to the healthy increase for the NIH’s base budget.”
Raise the Food and Drug Administration’s base budget in FY 2019 to $3.1 billion—a $308 million increase above its FY 2018 level—to secure support for regulatory science and speed the development of medical products that are safe and effective.
Provide the Centers for Disease Control and Prevention’s Cancer Prevention and Control Programs with total funding of at least $517 million. This would include funding for “comprehensive cancer control, cancer registries, and screening and awareness programs for specific cancers.”
The American Association for Cancer Research (AACR) has released its annual Cancer Progress Report, detailing recent advances in the fight against cancer and calling on elected officials to address the challenges that remain.
The AACR Cancer Progress Report 2018 lists the 22 new approvals for cancer treatments that have occurred during the last 12 months, including 12 therapies approved to treat hematologic malignancies.
However, the report also notes that cancer continues to pose immense public health challenges in the United States.
The estimated number of new cancer cases for 2018 is 1,735,350, and the estimated number of cancer deaths is 609,640.
The number of new cancer cases is predicted to increase to 2,387,304 in 2035. This is due, in large part, to the rising number of people age 65 and older, according to the report.
With this in mind, the AACR is calling on elected officials to:
Maintain “robust, sustained, and predictable growth” of the National Institutes of Health (NIH) budget, increasing it at least $2 billion in fiscal year (FY) 2019, for a total funding level of at least $39.1 billion.
Make sure the $711 million in funding provided through the 21st Century Cures Act for targeted initiatives—including the National Cancer Moonshot—“is fully appropriated in FY 2019 and is supplemental to the healthy increase for the NIH’s base budget.”
Raise the Food and Drug Administration’s base budget in FY 2019 to $3.1 billion—a $308 million increase above its FY 2018 level—to secure support for regulatory science and speed the development of medical products that are safe and effective.
Provide the Centers for Disease Control and Prevention’s Cancer Prevention and Control Programs with total funding of at least $517 million. This would include funding for “comprehensive cancer control, cancer registries, and screening and awareness programs for specific cancers.”
The American Association for Cancer Research (AACR) has released its annual Cancer Progress Report, detailing recent advances in the fight against cancer and calling on elected officials to address the challenges that remain.
The AACR Cancer Progress Report 2018 lists the 22 new approvals for cancer treatments that have occurred during the last 12 months, including 12 therapies approved to treat hematologic malignancies.
However, the report also notes that cancer continues to pose immense public health challenges in the United States.
The estimated number of new cancer cases for 2018 is 1,735,350, and the estimated number of cancer deaths is 609,640.
The number of new cancer cases is predicted to increase to 2,387,304 in 2035. This is due, in large part, to the rising number of people age 65 and older, according to the report.
With this in mind, the AACR is calling on elected officials to:
Maintain “robust, sustained, and predictable growth” of the National Institutes of Health (NIH) budget, increasing it at least $2 billion in fiscal year (FY) 2019, for a total funding level of at least $39.1 billion.
Make sure the $711 million in funding provided through the 21st Century Cures Act for targeted initiatives—including the National Cancer Moonshot—“is fully appropriated in FY 2019 and is supplemental to the healthy increase for the NIH’s base budget.”
Raise the Food and Drug Administration’s base budget in FY 2019 to $3.1 billion—a $308 million increase above its FY 2018 level—to secure support for regulatory science and speed the development of medical products that are safe and effective.
Provide the Centers for Disease Control and Prevention’s Cancer Prevention and Control Programs with total funding of at least $517 million. This would include funding for “comprehensive cancer control, cancer registries, and screening and awareness programs for specific cancers.”
Novel AKT inhibitor active against MM cells
A novel inhibitor of AKT pathway signaling showed significant cytotoxic activity in mouse models and in human cells isolated from patients with primary or relapsed multiple myeloma (MM), investigators reported.
The experimental agent, labeled HS1793, is a derivative of the naturally occurring antioxidant compound resveratrol. In preclinical studies, HS1793 was shown to offer “great promise in eliminating MM cells and improving therapeutic responses in primary and relapsed/refractory MM patients,” according to Jin Han, MD, PhD, of Inje University in Busan, South Korea, and colleagues.
In a series of experiments, described in the journal Cancer Letters, the investigators demonstrated that HS1793 decreased AKT signaling to induce mitochondria-mediated cell death in multiple myeloma cells, and was cytotoxic and specific for myeloma cells in a mouse model of human metastatic myeloma, and in samples of human multiple myeloma cells.
When activated, AKT promotes oncogenesis by in turn activating other downstream pathways involved in proliferation or survival of malignant cells.
“AKT is frequently activated in MM cells and the incidence of AKT activation correlates positively with disease activity,” the authors noted.
They first screened 400 compounds, and narrowed in on resveratrol analogs, eventually choosing HS1793 as the most promising candidate.
This first experiment found evidence that suggested that the compound inhibits AKT activation by interfering with the interaction between AKT and its promoter HSP90.
They then showed in human MM cell lines that the antimyeloma action of HS1793 appeared to be from a dose-dependent effect that allowed for mitochondria-mediated programmed cell death.
In a separate series of experiments, they found that the inhibition by HS1793 of AKT/HSP90 interaction results in cell death by suppressing nuclear factor kappa–B (NF-KB) pathway signaling. The investigators had previously reported that a different compound, an inhibitor of spindle protein kinesin, induced MM cell death via inhibition of NF-KB signaling.
Next, the investigators showed that HS1793-induced cell death was caused by the direct inhibition of AKT that in turn suppressed NF-KB activation.
Finally, they showed in a mouse model of multiple myeloma metastatic to bone that HS1793 “dramatically decreased” lytic skull and femur lesions in treated mice, compared with mice treated with a vehicle placebo, and increased survival of the mice that received the AKT inhibitor.
They also showed that HS1793 was cytotoxic to multiple myeloma cells but not to normal plasma cells isolated from patients with MM.
“Given that HS1793 treatment specifically induced the death of primary and relapsed MM cells, HS1793 offers excellent translational potential as a novel MM therapy,” they wrote.
The study was supported by grants from the Korean government. The researchers reported having no potential conflicts of interest.
SOURCE: Song IS et al. Cancer Lett. 2018;432:205-15.
A novel inhibitor of AKT pathway signaling showed significant cytotoxic activity in mouse models and in human cells isolated from patients with primary or relapsed multiple myeloma (MM), investigators reported.
The experimental agent, labeled HS1793, is a derivative of the naturally occurring antioxidant compound resveratrol. In preclinical studies, HS1793 was shown to offer “great promise in eliminating MM cells and improving therapeutic responses in primary and relapsed/refractory MM patients,” according to Jin Han, MD, PhD, of Inje University in Busan, South Korea, and colleagues.
In a series of experiments, described in the journal Cancer Letters, the investigators demonstrated that HS1793 decreased AKT signaling to induce mitochondria-mediated cell death in multiple myeloma cells, and was cytotoxic and specific for myeloma cells in a mouse model of human metastatic myeloma, and in samples of human multiple myeloma cells.
When activated, AKT promotes oncogenesis by in turn activating other downstream pathways involved in proliferation or survival of malignant cells.
“AKT is frequently activated in MM cells and the incidence of AKT activation correlates positively with disease activity,” the authors noted.
They first screened 400 compounds, and narrowed in on resveratrol analogs, eventually choosing HS1793 as the most promising candidate.
This first experiment found evidence that suggested that the compound inhibits AKT activation by interfering with the interaction between AKT and its promoter HSP90.
They then showed in human MM cell lines that the antimyeloma action of HS1793 appeared to be from a dose-dependent effect that allowed for mitochondria-mediated programmed cell death.
In a separate series of experiments, they found that the inhibition by HS1793 of AKT/HSP90 interaction results in cell death by suppressing nuclear factor kappa–B (NF-KB) pathway signaling. The investigators had previously reported that a different compound, an inhibitor of spindle protein kinesin, induced MM cell death via inhibition of NF-KB signaling.
Next, the investigators showed that HS1793-induced cell death was caused by the direct inhibition of AKT that in turn suppressed NF-KB activation.
Finally, they showed in a mouse model of multiple myeloma metastatic to bone that HS1793 “dramatically decreased” lytic skull and femur lesions in treated mice, compared with mice treated with a vehicle placebo, and increased survival of the mice that received the AKT inhibitor.
They also showed that HS1793 was cytotoxic to multiple myeloma cells but not to normal plasma cells isolated from patients with MM.
“Given that HS1793 treatment specifically induced the death of primary and relapsed MM cells, HS1793 offers excellent translational potential as a novel MM therapy,” they wrote.
The study was supported by grants from the Korean government. The researchers reported having no potential conflicts of interest.
SOURCE: Song IS et al. Cancer Lett. 2018;432:205-15.
A novel inhibitor of AKT pathway signaling showed significant cytotoxic activity in mouse models and in human cells isolated from patients with primary or relapsed multiple myeloma (MM), investigators reported.
The experimental agent, labeled HS1793, is a derivative of the naturally occurring antioxidant compound resveratrol. In preclinical studies, HS1793 was shown to offer “great promise in eliminating MM cells and improving therapeutic responses in primary and relapsed/refractory MM patients,” according to Jin Han, MD, PhD, of Inje University in Busan, South Korea, and colleagues.
In a series of experiments, described in the journal Cancer Letters, the investigators demonstrated that HS1793 decreased AKT signaling to induce mitochondria-mediated cell death in multiple myeloma cells, and was cytotoxic and specific for myeloma cells in a mouse model of human metastatic myeloma, and in samples of human multiple myeloma cells.
When activated, AKT promotes oncogenesis by in turn activating other downstream pathways involved in proliferation or survival of malignant cells.
“AKT is frequently activated in MM cells and the incidence of AKT activation correlates positively with disease activity,” the authors noted.
They first screened 400 compounds, and narrowed in on resveratrol analogs, eventually choosing HS1793 as the most promising candidate.
This first experiment found evidence that suggested that the compound inhibits AKT activation by interfering with the interaction between AKT and its promoter HSP90.
They then showed in human MM cell lines that the antimyeloma action of HS1793 appeared to be from a dose-dependent effect that allowed for mitochondria-mediated programmed cell death.
In a separate series of experiments, they found that the inhibition by HS1793 of AKT/HSP90 interaction results in cell death by suppressing nuclear factor kappa–B (NF-KB) pathway signaling. The investigators had previously reported that a different compound, an inhibitor of spindle protein kinesin, induced MM cell death via inhibition of NF-KB signaling.
Next, the investigators showed that HS1793-induced cell death was caused by the direct inhibition of AKT that in turn suppressed NF-KB activation.
Finally, they showed in a mouse model of multiple myeloma metastatic to bone that HS1793 “dramatically decreased” lytic skull and femur lesions in treated mice, compared with mice treated with a vehicle placebo, and increased survival of the mice that received the AKT inhibitor.
They also showed that HS1793 was cytotoxic to multiple myeloma cells but not to normal plasma cells isolated from patients with MM.
“Given that HS1793 treatment specifically induced the death of primary and relapsed MM cells, HS1793 offers excellent translational potential as a novel MM therapy,” they wrote.
The study was supported by grants from the Korean government. The researchers reported having no potential conflicts of interest.
SOURCE: Song IS et al. Cancer Lett. 2018;432:205-15.
FROM CANCER LETTERS
Key clinical point:
Major finding: HS1793 showed significant multiple myeloma cytotoxicity in mouse models and in human cells isolated from patients with primary or relapsed/refractory myeloma.
Study details: Preclinical investigations in cell lines, murine models, and isolated human multiple myeloma cells.
Disclosures: The study was supported by grants from the Korean government. The researchers reported having no potential conflicts of interest.
Source: Song IS et al. Cancer Lett. 2018:432:205-15.