User login
Doxorubicin, radiation doses predict heart risk in lymphoma survivors
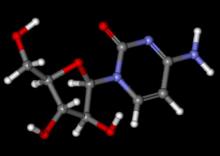
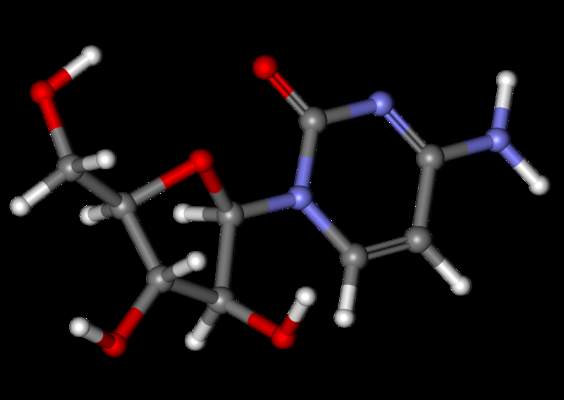
Adult lymphoma survivors who were treated with autologous hematopoietic stem-cell transplantation had a greater than sixfold increased risk of left ventricular systolic dysfunction compared with controls, according to a study published online in the Journal of Clinical Oncology.
Among 274 adult survivors of Hodgkin or non-Hodgkin lymphoma, 16% had left ventricular systolic dysfunction (LVSD): 11% had overt heart failure (HF) and 5% had asymptomatic LVSD, defined as a left ventricular ejection fraction of less than 50%.Heart symptoms were significantly associated with exposure to doxorubicin at a cumulative dose of 300 mg/m2 or more and with cardiac radiation therapy of more than 30 Gy. Recognizing these patient risk factors allows for more intensive follow-up with the goal of “identification and early treatment of asymptomatic LVSD [which] may prevent the development of HF,” wrote Dr. Klaus Murbraech of Oslo University Hospital and his colleagues (J. Clin. Oncol. 2015 July 13 [doi:10.1200/JCO.2015.60.8125]).
The investigators observed no association between lower-dose cardiac radiation therapy and LVSD. There was only a marginally significant association between the presence of two or more traditional cardiovascular disease risk factors and LVSD.
The cross-sectional multicenter cohort study is the first to assess the prevalence of LVSD, according to Dr. Murbraech and his colleagues. The study included adult survivors of Hodgkin or non-Hodgkin lymphoma, median age 56 years, who underwent autologous stem-cell transplants in Norway from 1987 to 2008. The median observation time was 13 years (range, 4-34 years). The control group consisted of initially healthy patients in an echocardiographic follow-up study. Controls were matched to patients based on age, sex, systolic blood pressure, and body mass index.
The study was supported by the South-Eastern Norway Regional Health Authority and Extrastiftelsen. Dr. Murbraech reported having no disclosures.
Adult lymphoma survivors who were treated with autologous hematopoietic stem-cell transplantation had a greater than sixfold increased risk of left ventricular systolic dysfunction compared with controls, according to a study published online in the Journal of Clinical Oncology.
Among 274 adult survivors of Hodgkin or non-Hodgkin lymphoma, 16% had left ventricular systolic dysfunction (LVSD): 11% had overt heart failure (HF) and 5% had asymptomatic LVSD, defined as a left ventricular ejection fraction of less than 50%.Heart symptoms were significantly associated with exposure to doxorubicin at a cumulative dose of 300 mg/m2 or more and with cardiac radiation therapy of more than 30 Gy. Recognizing these patient risk factors allows for more intensive follow-up with the goal of “identification and early treatment of asymptomatic LVSD [which] may prevent the development of HF,” wrote Dr. Klaus Murbraech of Oslo University Hospital and his colleagues (J. Clin. Oncol. 2015 July 13 [doi:10.1200/JCO.2015.60.8125]).
The investigators observed no association between lower-dose cardiac radiation therapy and LVSD. There was only a marginally significant association between the presence of two or more traditional cardiovascular disease risk factors and LVSD.
The cross-sectional multicenter cohort study is the first to assess the prevalence of LVSD, according to Dr. Murbraech and his colleagues. The study included adult survivors of Hodgkin or non-Hodgkin lymphoma, median age 56 years, who underwent autologous stem-cell transplants in Norway from 1987 to 2008. The median observation time was 13 years (range, 4-34 years). The control group consisted of initially healthy patients in an echocardiographic follow-up study. Controls were matched to patients based on age, sex, systolic blood pressure, and body mass index.
The study was supported by the South-Eastern Norway Regional Health Authority and Extrastiftelsen. Dr. Murbraech reported having no disclosures.
Adult lymphoma survivors who were treated with autologous hematopoietic stem-cell transplantation had a greater than sixfold increased risk of left ventricular systolic dysfunction compared with controls, according to a study published online in the Journal of Clinical Oncology.
Among 274 adult survivors of Hodgkin or non-Hodgkin lymphoma, 16% had left ventricular systolic dysfunction (LVSD): 11% had overt heart failure (HF) and 5% had asymptomatic LVSD, defined as a left ventricular ejection fraction of less than 50%.Heart symptoms were significantly associated with exposure to doxorubicin at a cumulative dose of 300 mg/m2 or more and with cardiac radiation therapy of more than 30 Gy. Recognizing these patient risk factors allows for more intensive follow-up with the goal of “identification and early treatment of asymptomatic LVSD [which] may prevent the development of HF,” wrote Dr. Klaus Murbraech of Oslo University Hospital and his colleagues (J. Clin. Oncol. 2015 July 13 [doi:10.1200/JCO.2015.60.8125]).
The investigators observed no association between lower-dose cardiac radiation therapy and LVSD. There was only a marginally significant association between the presence of two or more traditional cardiovascular disease risk factors and LVSD.
The cross-sectional multicenter cohort study is the first to assess the prevalence of LVSD, according to Dr. Murbraech and his colleagues. The study included adult survivors of Hodgkin or non-Hodgkin lymphoma, median age 56 years, who underwent autologous stem-cell transplants in Norway from 1987 to 2008. The median observation time was 13 years (range, 4-34 years). The control group consisted of initially healthy patients in an echocardiographic follow-up study. Controls were matched to patients based on age, sex, systolic blood pressure, and body mass index.
The study was supported by the South-Eastern Norway Regional Health Authority and Extrastiftelsen. Dr. Murbraech reported having no disclosures.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Lymphoma survivors treated with autologous hematopoietic stem-cell transplantation (auto-HSC) had a significantly higher risk of left ventricular systolic dysfunction than did controls.
Major finding: Treatment with at least 300 mg/m2 cumulative of doxorubicin and with over 30 Gy of cardiac radiation therapy were independent risk factors for LVSD.
Data source: A cross-sectional multicenter cohort study of 274 Hodgkin or non-Hodgkin lymphoma survivors.
Disclosures: Supported by the South-Eastern Norway Regional Health Authority and Extrastiftelsen. Dr. Murbraech reported having no disclosures.
ASCO updates guideline on CSFs
The American Society of Clinical Oncology (ASCO) has updated its clinical practice guideline on hematopoietic colony-stimulating factors (CSFs).
The guideline includes recommendations on the use of CSFs in the context of lymphoma, solid tumor malignancies, pediatric leukemia, and hematopoietic stem cell transplant.
There are no recommendations pertaining to adults with acute myeloid leukemia or myelodysplastic syndromes.
ASCO’s previous guideline on CSFs was issued in 2006. For the update, an ASCO expert panel conducted a formal systematic review of relevant articles from the medical literature published from October 2005 through September 2014.
Key recommendations from the resulting guideline are as follows.
Pegfilgrastim, filgrastim, tbo-filgrastim, and filgrastim-sndz (and other biosimilars, as they become available) can be used for the prevention of treatment-related febrile neutropenia.
For patients with lymphomas or solid tumors, primary prophylaxis with a CSF should be given during all cycles of chemotherapy in patients who have an approximately 20% or higher risk for febrile neutropenia on the basis of patient-, disease-, and treatment-related factors.
However, clinicians should also consider using chemotherapy regimens that do not require CSF administration but are as effective as regimens that do require a CSF.
Patients with lymphomas or solid tumors should receive secondary febrile neutropenia prophylaxis with a CSF if they experienced a neutropenic complication from a previous cycle of chemotherapy (for which they did not receive primary prophylaxis) when a reduced dose or treatment delay may compromise disease-free survival, overall survival, or treatment outcome.
However, the guideline also says that, in many clinical situations, dose reductions or delays may be a reasonable alternative.
CSFs should not be routinely used for patients with neutropenia who are afebrile or as adjunctive treatment with antibiotic therapy for patients with fever and neutropenia.
Dose-dense regimens with CSF support should only be used within an appropriately designed clinical trial or if use of the regimen is supported by convincing efficacy data. The guideline says that, for non-Hodgkin lymphoma, data on the value of dose-dense regimens with CSF support are limited and conflicting.
In the context of transplant, CSFs may be used alone, after chemotherapy, or in combination with plerixafor to mobilize peripheral blood stem cells. To reduce the duration of severe neutropenia, CSFs should be administered after autologous stem cell transplant and may be administered after allogeneic stem cell transplant.
CSFs should be avoided in patients receiving concomitant chemotherapy and radiation, particularly involving the mediastinum. CSFs may be considered in patients receiving radiation alone if the clinician expects prolonged treatment delays due to neutropenia.
Patients who are exposed to lethal doses of total-body radiotherapy, but not doses high enough to lead to certain death resulting from injury to other organs, should promptly receive CSFs or pegylated granulocyte CSFs.
Clinicians should consider prophylactic CSF for patients with diffuse aggressive lymphoma who are 65 or older and are receiving curative chemotherapy (R-CHOP), particularly if they have comorbidities.
The guideline also says the use of CSFs in pediatric patients will almost always be guided by clinical protocols. But CSFs should not be used in pediatric patients with nonrelapsed acute lymphoblastic leukemia or nonrelapsed acute myeloid leukemia who do not have an infection.
For more details, see the complete guideline. ASCO said it encourages feedback on its guidelines from oncologists, practitioners, and patients through the ASCO Guidelines Wiki. ![]()

The American Society of Clinical Oncology (ASCO) has updated its clinical practice guideline on hematopoietic colony-stimulating factors (CSFs).
The guideline includes recommendations on the use of CSFs in the context of lymphoma, solid tumor malignancies, pediatric leukemia, and hematopoietic stem cell transplant.
There are no recommendations pertaining to adults with acute myeloid leukemia or myelodysplastic syndromes.
ASCO’s previous guideline on CSFs was issued in 2006. For the update, an ASCO expert panel conducted a formal systematic review of relevant articles from the medical literature published from October 2005 through September 2014.
Key recommendations from the resulting guideline are as follows.
Pegfilgrastim, filgrastim, tbo-filgrastim, and filgrastim-sndz (and other biosimilars, as they become available) can be used for the prevention of treatment-related febrile neutropenia.
For patients with lymphomas or solid tumors, primary prophylaxis with a CSF should be given during all cycles of chemotherapy in patients who have an approximately 20% or higher risk for febrile neutropenia on the basis of patient-, disease-, and treatment-related factors.
However, clinicians should also consider using chemotherapy regimens that do not require CSF administration but are as effective as regimens that do require a CSF.
Patients with lymphomas or solid tumors should receive secondary febrile neutropenia prophylaxis with a CSF if they experienced a neutropenic complication from a previous cycle of chemotherapy (for which they did not receive primary prophylaxis) when a reduced dose or treatment delay may compromise disease-free survival, overall survival, or treatment outcome.
However, the guideline also says that, in many clinical situations, dose reductions or delays may be a reasonable alternative.
CSFs should not be routinely used for patients with neutropenia who are afebrile or as adjunctive treatment with antibiotic therapy for patients with fever and neutropenia.
Dose-dense regimens with CSF support should only be used within an appropriately designed clinical trial or if use of the regimen is supported by convincing efficacy data. The guideline says that, for non-Hodgkin lymphoma, data on the value of dose-dense regimens with CSF support are limited and conflicting.
In the context of transplant, CSFs may be used alone, after chemotherapy, or in combination with plerixafor to mobilize peripheral blood stem cells. To reduce the duration of severe neutropenia, CSFs should be administered after autologous stem cell transplant and may be administered after allogeneic stem cell transplant.
CSFs should be avoided in patients receiving concomitant chemotherapy and radiation, particularly involving the mediastinum. CSFs may be considered in patients receiving radiation alone if the clinician expects prolonged treatment delays due to neutropenia.
Patients who are exposed to lethal doses of total-body radiotherapy, but not doses high enough to lead to certain death resulting from injury to other organs, should promptly receive CSFs or pegylated granulocyte CSFs.
Clinicians should consider prophylactic CSF for patients with diffuse aggressive lymphoma who are 65 or older and are receiving curative chemotherapy (R-CHOP), particularly if they have comorbidities.
The guideline also says the use of CSFs in pediatric patients will almost always be guided by clinical protocols. But CSFs should not be used in pediatric patients with nonrelapsed acute lymphoblastic leukemia or nonrelapsed acute myeloid leukemia who do not have an infection.
For more details, see the complete guideline. ASCO said it encourages feedback on its guidelines from oncologists, practitioners, and patients through the ASCO Guidelines Wiki. ![]()

The American Society of Clinical Oncology (ASCO) has updated its clinical practice guideline on hematopoietic colony-stimulating factors (CSFs).
The guideline includes recommendations on the use of CSFs in the context of lymphoma, solid tumor malignancies, pediatric leukemia, and hematopoietic stem cell transplant.
There are no recommendations pertaining to adults with acute myeloid leukemia or myelodysplastic syndromes.
ASCO’s previous guideline on CSFs was issued in 2006. For the update, an ASCO expert panel conducted a formal systematic review of relevant articles from the medical literature published from October 2005 through September 2014.
Key recommendations from the resulting guideline are as follows.
Pegfilgrastim, filgrastim, tbo-filgrastim, and filgrastim-sndz (and other biosimilars, as they become available) can be used for the prevention of treatment-related febrile neutropenia.
For patients with lymphomas or solid tumors, primary prophylaxis with a CSF should be given during all cycles of chemotherapy in patients who have an approximately 20% or higher risk for febrile neutropenia on the basis of patient-, disease-, and treatment-related factors.
However, clinicians should also consider using chemotherapy regimens that do not require CSF administration but are as effective as regimens that do require a CSF.
Patients with lymphomas or solid tumors should receive secondary febrile neutropenia prophylaxis with a CSF if they experienced a neutropenic complication from a previous cycle of chemotherapy (for which they did not receive primary prophylaxis) when a reduced dose or treatment delay may compromise disease-free survival, overall survival, or treatment outcome.
However, the guideline also says that, in many clinical situations, dose reductions or delays may be a reasonable alternative.
CSFs should not be routinely used for patients with neutropenia who are afebrile or as adjunctive treatment with antibiotic therapy for patients with fever and neutropenia.
Dose-dense regimens with CSF support should only be used within an appropriately designed clinical trial or if use of the regimen is supported by convincing efficacy data. The guideline says that, for non-Hodgkin lymphoma, data on the value of dose-dense regimens with CSF support are limited and conflicting.
In the context of transplant, CSFs may be used alone, after chemotherapy, or in combination with plerixafor to mobilize peripheral blood stem cells. To reduce the duration of severe neutropenia, CSFs should be administered after autologous stem cell transplant and may be administered after allogeneic stem cell transplant.
CSFs should be avoided in patients receiving concomitant chemotherapy and radiation, particularly involving the mediastinum. CSFs may be considered in patients receiving radiation alone if the clinician expects prolonged treatment delays due to neutropenia.
Patients who are exposed to lethal doses of total-body radiotherapy, but not doses high enough to lead to certain death resulting from injury to other organs, should promptly receive CSFs or pegylated granulocyte CSFs.
Clinicians should consider prophylactic CSF for patients with diffuse aggressive lymphoma who are 65 or older and are receiving curative chemotherapy (R-CHOP), particularly if they have comorbidities.
The guideline also says the use of CSFs in pediatric patients will almost always be guided by clinical protocols. But CSFs should not be used in pediatric patients with nonrelapsed acute lymphoblastic leukemia or nonrelapsed acute myeloid leukemia who do not have an infection.
For more details, see the complete guideline. ASCO said it encourages feedback on its guidelines from oncologists, practitioners, and patients through the ASCO Guidelines Wiki. ![]()
Adopting may be more difficult for cancer survivors

by Vera Kratochvil
Cancer survivors may face more challenges when trying to adopt a child than individuals without a history of cancer, according to a new study.
Investigators found the sizable upfront costs associated with adoption and requirements regarding a prospective parent’s health could work against cancer survivors trying to adopt.
However, the study also suggested that birth mothers might be receptive to cancer survivors as adoptive parents.
The research was published in Cancer.
Little is known about the rate at which cancer survivors successfully adopt a child or about their experiences during the adoption process. So Gwendolyn Quinn, PhD, of the Moffitt Cancer Center in Tampa, Florida, and her colleagues conducted a study to gain some insight.
The investigators asked oncology nurses who were participating in a training program to conduct interviews with adoption agencies. Seventy-seven nurses across 15 states provided summaries of their interviews.
The nurses reported that adoption fees ranged from $3000 to $75,000. They noted that the upfront costs of adoption could deter cancer survivors who already have “a huge financial burden” due to treatment costs.
Not all of the adoption agencies contacted kept records on whether prospective adoptive parents were cancer survivors. But agencies that did track this reported an average of 10 former cancer patients a year seeking adoption.
A few agencies reported that a cancer history in an adoptive parent could be discouraging for a birth mother. But most reported the opposite—that birth mothers might feel confident in choosing a parent who has overcome hardships and has an appreciation for life.
Agencies usually required prospective parents to provide a letter from a physician regarding their health and medical history. In some cases, agencies required cancer survivors to be disease-free for 5 years before they could adopt a child.
In addition, international adoptions had greater restrictions for prospective parents with a cancer history (compared to US adoptions).
Dr Quinn said these are potentially discriminatory practices akin to restricting employment opportunities for people with disabilities.
“[P]erhaps this data will bring to light the need for policy revisions in adoption processes . . . ,” she added. ![]()

by Vera Kratochvil
Cancer survivors may face more challenges when trying to adopt a child than individuals without a history of cancer, according to a new study.
Investigators found the sizable upfront costs associated with adoption and requirements regarding a prospective parent’s health could work against cancer survivors trying to adopt.
However, the study also suggested that birth mothers might be receptive to cancer survivors as adoptive parents.
The research was published in Cancer.
Little is known about the rate at which cancer survivors successfully adopt a child or about their experiences during the adoption process. So Gwendolyn Quinn, PhD, of the Moffitt Cancer Center in Tampa, Florida, and her colleagues conducted a study to gain some insight.
The investigators asked oncology nurses who were participating in a training program to conduct interviews with adoption agencies. Seventy-seven nurses across 15 states provided summaries of their interviews.
The nurses reported that adoption fees ranged from $3000 to $75,000. They noted that the upfront costs of adoption could deter cancer survivors who already have “a huge financial burden” due to treatment costs.
Not all of the adoption agencies contacted kept records on whether prospective adoptive parents were cancer survivors. But agencies that did track this reported an average of 10 former cancer patients a year seeking adoption.
A few agencies reported that a cancer history in an adoptive parent could be discouraging for a birth mother. But most reported the opposite—that birth mothers might feel confident in choosing a parent who has overcome hardships and has an appreciation for life.
Agencies usually required prospective parents to provide a letter from a physician regarding their health and medical history. In some cases, agencies required cancer survivors to be disease-free for 5 years before they could adopt a child.
In addition, international adoptions had greater restrictions for prospective parents with a cancer history (compared to US adoptions).
Dr Quinn said these are potentially discriminatory practices akin to restricting employment opportunities for people with disabilities.
“[P]erhaps this data will bring to light the need for policy revisions in adoption processes . . . ,” she added. ![]()

by Vera Kratochvil
Cancer survivors may face more challenges when trying to adopt a child than individuals without a history of cancer, according to a new study.
Investigators found the sizable upfront costs associated with adoption and requirements regarding a prospective parent’s health could work against cancer survivors trying to adopt.
However, the study also suggested that birth mothers might be receptive to cancer survivors as adoptive parents.
The research was published in Cancer.
Little is known about the rate at which cancer survivors successfully adopt a child or about their experiences during the adoption process. So Gwendolyn Quinn, PhD, of the Moffitt Cancer Center in Tampa, Florida, and her colleagues conducted a study to gain some insight.
The investigators asked oncology nurses who were participating in a training program to conduct interviews with adoption agencies. Seventy-seven nurses across 15 states provided summaries of their interviews.
The nurses reported that adoption fees ranged from $3000 to $75,000. They noted that the upfront costs of adoption could deter cancer survivors who already have “a huge financial burden” due to treatment costs.
Not all of the adoption agencies contacted kept records on whether prospective adoptive parents were cancer survivors. But agencies that did track this reported an average of 10 former cancer patients a year seeking adoption.
A few agencies reported that a cancer history in an adoptive parent could be discouraging for a birth mother. But most reported the opposite—that birth mothers might feel confident in choosing a parent who has overcome hardships and has an appreciation for life.
Agencies usually required prospective parents to provide a letter from a physician regarding their health and medical history. In some cases, agencies required cancer survivors to be disease-free for 5 years before they could adopt a child.
In addition, international adoptions had greater restrictions for prospective parents with a cancer history (compared to US adoptions).
Dr Quinn said these are potentially discriminatory practices akin to restricting employment opportunities for people with disabilities.
“[P]erhaps this data will bring to light the need for policy revisions in adoption processes . . . ,” she added. ![]()
Ibrutinib approved to treat WM in EU

The European Commission has granted marketing authorization for ibrutinib (Imbruvica) to treat Waldenstrom’s macroglobulinemia (WM).
The Bruton’s tyrosine kinase inhibitor is now approved to treat adults with WM who have received at least one prior therapy or as first-line treatment for patients considered unsuitable for chemo-immunotherapy.
Ibrutinib is the first therapy approved specifically for WM in the European Union (EU). The approval applies to all 28 EU member states, plus Iceland, Norway, and Liechtenstein.
Ibrutinib is already approved to treat WM in the US. The drug is also approved in the EU, the US, and other countries to treat chronic lymphocytic leukemia and mantle cell lymphoma.
Janssen-Cilag International NV holds the marketing authorization for ibrutinib in Europe, and its affiliates market the drug in Europe and the rest of the world. In the US, ibrutinib is under joint development by Pharmacyclics and Janssen Biotech, Inc.
Phase 2 study
The European Commission’s approval of ibrutinib was based on a multicenter, phase 2 study in which researchers tested the drug (given at 420 mg once daily) in 63 patients with previously treated WM.
Initial data showed an overall response rate of 87.3% in patients who received the drug for a median of 11.7 months.
Updated results from the study were published in NEJM in April. After a median treatment duration of 19.1 months, the overall response rate was 91%.
At 24 months, the estimated rate of progression-free survival was 69%, and the estimated rate of overall survival was 95%.
The most common grade 2-4 adverse events were neutropenia (22%) and thrombocytopenia (14%). Ibrutinib-related neutropenia and thrombocytopenia were reversible but required a dose reduction in 3 patients and treatment discontinuation in 4 patients.
Grade 2 or higher bleeding events occurred in 4 patients, and there were 15 infections considered possibly related to ibrutinib.
Treatment-related atrial fibrillation (AFib) occurred in 3 patients, all of whom had a prior history of paroxysmal AFib. AFib resolved when treatment was withheld, and all 3 patients were able to continue on therapy per protocol without an additional event. ![]()

The European Commission has granted marketing authorization for ibrutinib (Imbruvica) to treat Waldenstrom’s macroglobulinemia (WM).
The Bruton’s tyrosine kinase inhibitor is now approved to treat adults with WM who have received at least one prior therapy or as first-line treatment for patients considered unsuitable for chemo-immunotherapy.
Ibrutinib is the first therapy approved specifically for WM in the European Union (EU). The approval applies to all 28 EU member states, plus Iceland, Norway, and Liechtenstein.
Ibrutinib is already approved to treat WM in the US. The drug is also approved in the EU, the US, and other countries to treat chronic lymphocytic leukemia and mantle cell lymphoma.
Janssen-Cilag International NV holds the marketing authorization for ibrutinib in Europe, and its affiliates market the drug in Europe and the rest of the world. In the US, ibrutinib is under joint development by Pharmacyclics and Janssen Biotech, Inc.
Phase 2 study
The European Commission’s approval of ibrutinib was based on a multicenter, phase 2 study in which researchers tested the drug (given at 420 mg once daily) in 63 patients with previously treated WM.
Initial data showed an overall response rate of 87.3% in patients who received the drug for a median of 11.7 months.
Updated results from the study were published in NEJM in April. After a median treatment duration of 19.1 months, the overall response rate was 91%.
At 24 months, the estimated rate of progression-free survival was 69%, and the estimated rate of overall survival was 95%.
The most common grade 2-4 adverse events were neutropenia (22%) and thrombocytopenia (14%). Ibrutinib-related neutropenia and thrombocytopenia were reversible but required a dose reduction in 3 patients and treatment discontinuation in 4 patients.
Grade 2 or higher bleeding events occurred in 4 patients, and there were 15 infections considered possibly related to ibrutinib.
Treatment-related atrial fibrillation (AFib) occurred in 3 patients, all of whom had a prior history of paroxysmal AFib. AFib resolved when treatment was withheld, and all 3 patients were able to continue on therapy per protocol without an additional event. ![]()

The European Commission has granted marketing authorization for ibrutinib (Imbruvica) to treat Waldenstrom’s macroglobulinemia (WM).
The Bruton’s tyrosine kinase inhibitor is now approved to treat adults with WM who have received at least one prior therapy or as first-line treatment for patients considered unsuitable for chemo-immunotherapy.
Ibrutinib is the first therapy approved specifically for WM in the European Union (EU). The approval applies to all 28 EU member states, plus Iceland, Norway, and Liechtenstein.
Ibrutinib is already approved to treat WM in the US. The drug is also approved in the EU, the US, and other countries to treat chronic lymphocytic leukemia and mantle cell lymphoma.
Janssen-Cilag International NV holds the marketing authorization for ibrutinib in Europe, and its affiliates market the drug in Europe and the rest of the world. In the US, ibrutinib is under joint development by Pharmacyclics and Janssen Biotech, Inc.
Phase 2 study
The European Commission’s approval of ibrutinib was based on a multicenter, phase 2 study in which researchers tested the drug (given at 420 mg once daily) in 63 patients with previously treated WM.
Initial data showed an overall response rate of 87.3% in patients who received the drug for a median of 11.7 months.
Updated results from the study were published in NEJM in April. After a median treatment duration of 19.1 months, the overall response rate was 91%.
At 24 months, the estimated rate of progression-free survival was 69%, and the estimated rate of overall survival was 95%.
The most common grade 2-4 adverse events were neutropenia (22%) and thrombocytopenia (14%). Ibrutinib-related neutropenia and thrombocytopenia were reversible but required a dose reduction in 3 patients and treatment discontinuation in 4 patients.
Grade 2 or higher bleeding events occurred in 4 patients, and there were 15 infections considered possibly related to ibrutinib.
Treatment-related atrial fibrillation (AFib) occurred in 3 patients, all of whom had a prior history of paroxysmal AFib. AFib resolved when treatment was withheld, and all 3 patients were able to continue on therapy per protocol without an additional event. ![]()
Older cancer patients under-utilize advanced care planning

Photo courtesy of NCI
and Mathews Media Group
Survey results suggest the use of aggressive treatment at the end of life is on the rise among older cancer patients, and these patients often fail to employ advanced care planning measures.
Researchers reviewed nearly 2000 surveys of people whose loved ones died of cancer and found that, from 2000 to 2012, there was a 51% increase in reports that patients received “all care possible” at the end of life.
There was a 22% increase in power-of-attorney assignment over the same period, but the use of living wills and discussions about end-of-life preferences decreased slightly.
“Although more cancer patients are assigning power-of-attorney privileges to someone they know and trust to make their medical decisions when they can’t, this practice may be the least helpful among advanced care planning tactics because it may be least associated with treatment intensity at the end of life,” said Amol Narang, MD, of The Johns Hopkins Hospital in Baltimore, Maryland.
Dr Narang and his colleagues described this research in JAMA Oncology.
The team analyzed survey data from 1985 next-of-kin surrogates of cancer patients. The patients were older than 50 years of age, had taken part in the Health and Retirement Study, and died between 2000 and 2012.
The data included in-depth “exit” interviews conducted with the surrogates after a patient died. Seventy-nine percent of exit survey respondents said they were the primary decision-maker in the patient’s medical care.
The data showed a significant increase in power-of-attorney assignments, from 52% in 2000 to 74% in 2012 (P=0.03). But the use of living wills decreased slightly, from 49% to 40% (P=0.63), as did discussions about end-of-life preferences, which fell from 68% to 60% (P=0.62).
“We found that many cancer patients still do not communicate their preferences for end-of-life care, despite the potential benefits to patients’ quality of life and caregiver bereavement,” Dr Narang said.
Survey results also suggested a significant increase in the percentage of patients who received “all care possible” at the end of life, from 7% in 2000 to 58% in 2012 (P=0.004). But there was no significant change in the rates of terminal hospitalizations, which fell from 29% to 27% (P=0.70).
The researchers found that granting power-of-attorney privileges significantly decreased the odds that patients would die in the hospital as opposed to hospice or their homes (adjusted odds ratio [AOR]=0.70, P<0.05). However, granting power of attorney was not associated with a significant change in limiting or withholding treatment at the end of life (AOR=1.52).
On the other hand, patients who created living wills or had end-of-life discussions were significantly more likely than their peers to limit or withhold certain treatments. The AOR was 2.51 for living wills (P<0.001) and 1.93 for end-of-life discussions (P=0.002).
The researchers said they observed the same trends regardless of who completed the exit survey.
Dr Narang noted that this study had its limitations. The survey questions were subjective, answers could have been hampered by a respondent’s lapse in memory, and answers could be biased by a respondent’s desire to meet social norms.
“But we were looking at trends over time,” he said, “so respondents’ bias would not likely change over time.” ![]()

Photo courtesy of NCI
and Mathews Media Group
Survey results suggest the use of aggressive treatment at the end of life is on the rise among older cancer patients, and these patients often fail to employ advanced care planning measures.
Researchers reviewed nearly 2000 surveys of people whose loved ones died of cancer and found that, from 2000 to 2012, there was a 51% increase in reports that patients received “all care possible” at the end of life.
There was a 22% increase in power-of-attorney assignment over the same period, but the use of living wills and discussions about end-of-life preferences decreased slightly.
“Although more cancer patients are assigning power-of-attorney privileges to someone they know and trust to make their medical decisions when they can’t, this practice may be the least helpful among advanced care planning tactics because it may be least associated with treatment intensity at the end of life,” said Amol Narang, MD, of The Johns Hopkins Hospital in Baltimore, Maryland.
Dr Narang and his colleagues described this research in JAMA Oncology.
The team analyzed survey data from 1985 next-of-kin surrogates of cancer patients. The patients were older than 50 years of age, had taken part in the Health and Retirement Study, and died between 2000 and 2012.
The data included in-depth “exit” interviews conducted with the surrogates after a patient died. Seventy-nine percent of exit survey respondents said they were the primary decision-maker in the patient’s medical care.
The data showed a significant increase in power-of-attorney assignments, from 52% in 2000 to 74% in 2012 (P=0.03). But the use of living wills decreased slightly, from 49% to 40% (P=0.63), as did discussions about end-of-life preferences, which fell from 68% to 60% (P=0.62).
“We found that many cancer patients still do not communicate their preferences for end-of-life care, despite the potential benefits to patients’ quality of life and caregiver bereavement,” Dr Narang said.
Survey results also suggested a significant increase in the percentage of patients who received “all care possible” at the end of life, from 7% in 2000 to 58% in 2012 (P=0.004). But there was no significant change in the rates of terminal hospitalizations, which fell from 29% to 27% (P=0.70).
The researchers found that granting power-of-attorney privileges significantly decreased the odds that patients would die in the hospital as opposed to hospice or their homes (adjusted odds ratio [AOR]=0.70, P<0.05). However, granting power of attorney was not associated with a significant change in limiting or withholding treatment at the end of life (AOR=1.52).
On the other hand, patients who created living wills or had end-of-life discussions were significantly more likely than their peers to limit or withhold certain treatments. The AOR was 2.51 for living wills (P<0.001) and 1.93 for end-of-life discussions (P=0.002).
The researchers said they observed the same trends regardless of who completed the exit survey.
Dr Narang noted that this study had its limitations. The survey questions were subjective, answers could have been hampered by a respondent’s lapse in memory, and answers could be biased by a respondent’s desire to meet social norms.
“But we were looking at trends over time,” he said, “so respondents’ bias would not likely change over time.” ![]()

Photo courtesy of NCI
and Mathews Media Group
Survey results suggest the use of aggressive treatment at the end of life is on the rise among older cancer patients, and these patients often fail to employ advanced care planning measures.
Researchers reviewed nearly 2000 surveys of people whose loved ones died of cancer and found that, from 2000 to 2012, there was a 51% increase in reports that patients received “all care possible” at the end of life.
There was a 22% increase in power-of-attorney assignment over the same period, but the use of living wills and discussions about end-of-life preferences decreased slightly.
“Although more cancer patients are assigning power-of-attorney privileges to someone they know and trust to make their medical decisions when they can’t, this practice may be the least helpful among advanced care planning tactics because it may be least associated with treatment intensity at the end of life,” said Amol Narang, MD, of The Johns Hopkins Hospital in Baltimore, Maryland.
Dr Narang and his colleagues described this research in JAMA Oncology.
The team analyzed survey data from 1985 next-of-kin surrogates of cancer patients. The patients were older than 50 years of age, had taken part in the Health and Retirement Study, and died between 2000 and 2012.
The data included in-depth “exit” interviews conducted with the surrogates after a patient died. Seventy-nine percent of exit survey respondents said they were the primary decision-maker in the patient’s medical care.
The data showed a significant increase in power-of-attorney assignments, from 52% in 2000 to 74% in 2012 (P=0.03). But the use of living wills decreased slightly, from 49% to 40% (P=0.63), as did discussions about end-of-life preferences, which fell from 68% to 60% (P=0.62).
“We found that many cancer patients still do not communicate their preferences for end-of-life care, despite the potential benefits to patients’ quality of life and caregiver bereavement,” Dr Narang said.
Survey results also suggested a significant increase in the percentage of patients who received “all care possible” at the end of life, from 7% in 2000 to 58% in 2012 (P=0.004). But there was no significant change in the rates of terminal hospitalizations, which fell from 29% to 27% (P=0.70).
The researchers found that granting power-of-attorney privileges significantly decreased the odds that patients would die in the hospital as opposed to hospice or their homes (adjusted odds ratio [AOR]=0.70, P<0.05). However, granting power of attorney was not associated with a significant change in limiting or withholding treatment at the end of life (AOR=1.52).
On the other hand, patients who created living wills or had end-of-life discussions were significantly more likely than their peers to limit or withhold certain treatments. The AOR was 2.51 for living wills (P<0.001) and 1.93 for end-of-life discussions (P=0.002).
The researchers said they observed the same trends regardless of who completed the exit survey.
Dr Narang noted that this study had its limitations. The survey questions were subjective, answers could have been hampered by a respondent’s lapse in memory, and answers could be biased by a respondent’s desire to meet social norms.
“But we were looking at trends over time,” he said, “so respondents’ bias would not likely change over time.” ![]()
How B-cell lymphoma evades NK cells

Image by Joshua Strokes
Researchers say they have determined how lymphoma cells evade natural killer (NK) cells, and this discovery has revealed potential solutions to the problem.
The team found that NK-cell activation and a second, “triggering” event are both necessary for NK cells to exhibit cytotoxicity in the presence of B-cell lymphoma.
Previous research demonstrated this 2-step process in vitro. Now, researchers have shown that it occurs in vivo.
Dr Ralph Mocikat, of Helmholtz Zentrum München in Munich, Germany, and his colleagues described this research in the European Journal of Immunology.
The team conducted experiments using transplantable tumors, a λ-myc-transgenic model of endogenously arising lymphoma that mimics human Burkitt lymphoma, and mice deficient in the NK group 2 D (NKG2D) receptor.
The experiments showed that NK cells could eliminate lymphoma cells after receiving 2 signals. The first was NK-cell activation, which gave rise to IFN-γ expression.
The researchers found that NK cells could be activated in the presence of MHC class Ilow tumor cells or by injecting bone marrow-derived dendritic cells. Previous research had shown that interleukin 2 (IL-2) and IL-15 could activate NK cells.
The second step involved the NKG2D receptor and its ligands. NKG2D ligands are located on the surface of tumor cells and bind to NK cells. The researchers found that, if these ligands are down-regulated, the NK cells cannot carry out cytotoxic activity.
However, the team found they could increase NKG2D ligand levels. They introduced bortezomib to the tumor cell line 291 and saw a roughly 4-fold increase in NKG2D ligand levels.
“Our results show that the transfer of NK cells is a possible strategic option to treat B-cell lymphoma,” Dr Mocikat said. “According to our findings, this therapeutic approach can be optimized when transferred NK cells are already activated in vitro prior to their injection, thus bypassing the missing activation potential in the tumor microenvironment. An additional injection of IFN-γ or of antibodies against IL-10 could further support the immune activity.” ![]()

Image by Joshua Strokes
Researchers say they have determined how lymphoma cells evade natural killer (NK) cells, and this discovery has revealed potential solutions to the problem.
The team found that NK-cell activation and a second, “triggering” event are both necessary for NK cells to exhibit cytotoxicity in the presence of B-cell lymphoma.
Previous research demonstrated this 2-step process in vitro. Now, researchers have shown that it occurs in vivo.
Dr Ralph Mocikat, of Helmholtz Zentrum München in Munich, Germany, and his colleagues described this research in the European Journal of Immunology.
The team conducted experiments using transplantable tumors, a λ-myc-transgenic model of endogenously arising lymphoma that mimics human Burkitt lymphoma, and mice deficient in the NK group 2 D (NKG2D) receptor.
The experiments showed that NK cells could eliminate lymphoma cells after receiving 2 signals. The first was NK-cell activation, which gave rise to IFN-γ expression.
The researchers found that NK cells could be activated in the presence of MHC class Ilow tumor cells or by injecting bone marrow-derived dendritic cells. Previous research had shown that interleukin 2 (IL-2) and IL-15 could activate NK cells.
The second step involved the NKG2D receptor and its ligands. NKG2D ligands are located on the surface of tumor cells and bind to NK cells. The researchers found that, if these ligands are down-regulated, the NK cells cannot carry out cytotoxic activity.
However, the team found they could increase NKG2D ligand levels. They introduced bortezomib to the tumor cell line 291 and saw a roughly 4-fold increase in NKG2D ligand levels.
“Our results show that the transfer of NK cells is a possible strategic option to treat B-cell lymphoma,” Dr Mocikat said. “According to our findings, this therapeutic approach can be optimized when transferred NK cells are already activated in vitro prior to their injection, thus bypassing the missing activation potential in the tumor microenvironment. An additional injection of IFN-γ or of antibodies against IL-10 could further support the immune activity.” ![]()

Image by Joshua Strokes
Researchers say they have determined how lymphoma cells evade natural killer (NK) cells, and this discovery has revealed potential solutions to the problem.
The team found that NK-cell activation and a second, “triggering” event are both necessary for NK cells to exhibit cytotoxicity in the presence of B-cell lymphoma.
Previous research demonstrated this 2-step process in vitro. Now, researchers have shown that it occurs in vivo.
Dr Ralph Mocikat, of Helmholtz Zentrum München in Munich, Germany, and his colleagues described this research in the European Journal of Immunology.
The team conducted experiments using transplantable tumors, a λ-myc-transgenic model of endogenously arising lymphoma that mimics human Burkitt lymphoma, and mice deficient in the NK group 2 D (NKG2D) receptor.
The experiments showed that NK cells could eliminate lymphoma cells after receiving 2 signals. The first was NK-cell activation, which gave rise to IFN-γ expression.
The researchers found that NK cells could be activated in the presence of MHC class Ilow tumor cells or by injecting bone marrow-derived dendritic cells. Previous research had shown that interleukin 2 (IL-2) and IL-15 could activate NK cells.
The second step involved the NKG2D receptor and its ligands. NKG2D ligands are located on the surface of tumor cells and bind to NK cells. The researchers found that, if these ligands are down-regulated, the NK cells cannot carry out cytotoxic activity.
However, the team found they could increase NKG2D ligand levels. They introduced bortezomib to the tumor cell line 291 and saw a roughly 4-fold increase in NKG2D ligand levels.
“Our results show that the transfer of NK cells is a possible strategic option to treat B-cell lymphoma,” Dr Mocikat said. “According to our findings, this therapeutic approach can be optimized when transferred NK cells are already activated in vitro prior to their injection, thus bypassing the missing activation potential in the tumor microenvironment. An additional injection of IFN-γ or of antibodies against IL-10 could further support the immune activity.” ![]()
AYAs with cancer receive aggressive EOL care

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()

patient and her father
Photo by Rhoda Baer
In a retrospective study, a majority of adolescents and young adults (AYAs) with terminal cancer received aggressive end-of-life (EOL) care.
Investigators looked at the use of intensive care, emergency room visits, chemotherapy use, and hospitalization among more than 600 AYAs with cancer who were treated at Kaiser Permanente in California.
Nearly 70% of patients made use of at least one of these measures in the last month of their life.
The investigators noted that their findings, which were published in JAMA Oncology, may not reflect care for the wider US population. But the study does suggest a need for more research into whether this
pattern reflects AYA cancer patients’ preferences for EOL care.
“A young person facing the end of life is a particularly difficult issue,” said study author Jennifer Mack, MD, MPH, of the Dana-Farber Cancer Institute in Boston, Massachusetts.
“While use of aggressive measures might be an informed decision by young people who would do anything they could to live longer, some interventions come with a cost, which is a poorer quality of life. This study raises questions about what kind of care they’re getting and how we can get them to the best quality of life at the end of their lives.”
The study included 633 patients, ages 15 to 39, who died of cancer between 2001 and 2010. The patients, who received care at Kaiser Permanente Southern California, had either been diagnosed with stage IV cancer or had a recurrence of stage I-III cancer. An initial review of a subset of 111 patients showed that death had been anticipated in 98% of cases.
The most common cancer diagnosis was gastrointestinal cancer (17%), while other common diagnoses were breast cancer (15%), genitourinary cancers (11%), leukemia (14%), and lymphoma (10%).
The investigators measured the use of 4 aggressive treatment measures—intensive care, emergency room visits, chemotherapy, and hospitalization—in patients’ last month of life.
Overall, 68% of patients (449/663) received at least one of these medically intensive EOL care measures. Eleven percent of patients (72/663) received chemotherapy, 22% (144/663) were admitted to the intensive care unit, 22% (147/663) had more than one emergency department visit, and 62% (413/663) were hospitalized.
Rates of hospitalization were higher among patients diagnosed with stage IV disease (66%) than among patients with stage I to III disease—66% and 58%, respectively (P=0.04).
The percentage of patients who received at least one medically intensive EOL care measure was higher in the stage IV cohort as well—71% and 63%, respectively (P=0.03). But there were no significant differences between the cohorts with regard to the other measures.
The investigators said these findings suggest the need to better understand EOL care preferences and decision-making in AYAs with cancer.
“We should think about talking with younger patients earlier about their prognoses, identifying their preferences, and working with them to deliver care that reflects those preferences,” Dr Mack said. “It may be that aggressive care is what they want, but they may end up on this pathway without thoughtful conversation and may be without recognition that they are dying.” ![]()
Combo shows potential for treating PEL

reviewing an image
Photo from the University
of Southern California
Immunomodulatory drugs (IMiDs) and BRD4 inhibitors hold promise for treating primary effusion lymphoma (PEL), according to preclinical research.
The IMiDs lenalidomide and pomalidomide had antiproliferative effects in PEL cell lines, and lenalidomide exhibited synergistic cytotoxicity with the BRD4 inhibitors JQ-1, IBET151, and PFI-1.
In mouse models of PEL, treatment with lenalidomide and JQ-1 significantly prolonged survival.
Preet Chaudhary, MD, PhD, of the University of Southern California, Los Angeles, and his colleagues reported these results in Oncogene.
The researchers first tested lenalidomide and pomalidomide in 9 PEL cell lines. Both drugs exhibited antiproliferative effects, at clinically achievable doses, in 6 of the cell lines (BC-3, BCBL-1, JSC-1, VG-1, UMPEL-1, and UMPEL-3).
The IMiDs displayed activity in the remaining 3 cells lines (BC-1, BCP-1, and APK-1) as well, but only at high doses (ranging from 2.6 μM to 10 μM for lenalidomide and from 226 nM to 744 nM for pomalidomide).
Looking into the mechanism of IMiDs in PEL, the researchers found these drugs induce G1 cell-cycle arrest without inducing Kaposi’s sarcoma-associated herpes virus lytic cycle reactivation.
Further investigation revealed that interferon α, β, and γ are cytotoxic to PEL, but they are not essential for the antiproliferative effects of IMiDs. On the other hand, cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1 are essential.
The researchers also found that knocking down MYC enhanced IMiD activity. This led them to BRD4 inhibitors, which are known to target MYC transcription.
The team tested lenalidomide in combination with JQ-1, IBET151, and PFI-1. The IMiD was “highly synergistic” with all 3 BRD4 inhibitors in BC-3 and BCBL-1 cell lines.
In both cell lines, treatment with lenalidomide and JQ-1 significantly decreased the expression of MYC and IRF4 at protein and mRNA levels. The treatment also resulted in G1 cell cycle arrest, cleavage of poly (ADP-ribose) polymerase, and apoptosis.
So the researchers tested this combination in a mouse model of PEL (NOD-SCID mice injected with BC-3 cells). And they observed a significant improvement in median survival for mice that received combination treatment, compared to lenalidomide or JQ-1 alone—51 days, 35 days, and 42 days, respectively (P≤0.01).![]()

reviewing an image
Photo from the University
of Southern California
Immunomodulatory drugs (IMiDs) and BRD4 inhibitors hold promise for treating primary effusion lymphoma (PEL), according to preclinical research.
The IMiDs lenalidomide and pomalidomide had antiproliferative effects in PEL cell lines, and lenalidomide exhibited synergistic cytotoxicity with the BRD4 inhibitors JQ-1, IBET151, and PFI-1.
In mouse models of PEL, treatment with lenalidomide and JQ-1 significantly prolonged survival.
Preet Chaudhary, MD, PhD, of the University of Southern California, Los Angeles, and his colleagues reported these results in Oncogene.
The researchers first tested lenalidomide and pomalidomide in 9 PEL cell lines. Both drugs exhibited antiproliferative effects, at clinically achievable doses, in 6 of the cell lines (BC-3, BCBL-1, JSC-1, VG-1, UMPEL-1, and UMPEL-3).
The IMiDs displayed activity in the remaining 3 cells lines (BC-1, BCP-1, and APK-1) as well, but only at high doses (ranging from 2.6 μM to 10 μM for lenalidomide and from 226 nM to 744 nM for pomalidomide).
Looking into the mechanism of IMiDs in PEL, the researchers found these drugs induce G1 cell-cycle arrest without inducing Kaposi’s sarcoma-associated herpes virus lytic cycle reactivation.
Further investigation revealed that interferon α, β, and γ are cytotoxic to PEL, but they are not essential for the antiproliferative effects of IMiDs. On the other hand, cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1 are essential.
The researchers also found that knocking down MYC enhanced IMiD activity. This led them to BRD4 inhibitors, which are known to target MYC transcription.
The team tested lenalidomide in combination with JQ-1, IBET151, and PFI-1. The IMiD was “highly synergistic” with all 3 BRD4 inhibitors in BC-3 and BCBL-1 cell lines.
In both cell lines, treatment with lenalidomide and JQ-1 significantly decreased the expression of MYC and IRF4 at protein and mRNA levels. The treatment also resulted in G1 cell cycle arrest, cleavage of poly (ADP-ribose) polymerase, and apoptosis.
So the researchers tested this combination in a mouse model of PEL (NOD-SCID mice injected with BC-3 cells). And they observed a significant improvement in median survival for mice that received combination treatment, compared to lenalidomide or JQ-1 alone—51 days, 35 days, and 42 days, respectively (P≤0.01).![]()

reviewing an image
Photo from the University
of Southern California
Immunomodulatory drugs (IMiDs) and BRD4 inhibitors hold promise for treating primary effusion lymphoma (PEL), according to preclinical research.
The IMiDs lenalidomide and pomalidomide had antiproliferative effects in PEL cell lines, and lenalidomide exhibited synergistic cytotoxicity with the BRD4 inhibitors JQ-1, IBET151, and PFI-1.
In mouse models of PEL, treatment with lenalidomide and JQ-1 significantly prolonged survival.
Preet Chaudhary, MD, PhD, of the University of Southern California, Los Angeles, and his colleagues reported these results in Oncogene.
The researchers first tested lenalidomide and pomalidomide in 9 PEL cell lines. Both drugs exhibited antiproliferative effects, at clinically achievable doses, in 6 of the cell lines (BC-3, BCBL-1, JSC-1, VG-1, UMPEL-1, and UMPEL-3).
The IMiDs displayed activity in the remaining 3 cells lines (BC-1, BCP-1, and APK-1) as well, but only at high doses (ranging from 2.6 μM to 10 μM for lenalidomide and from 226 nM to 744 nM for pomalidomide).
Looking into the mechanism of IMiDs in PEL, the researchers found these drugs induce G1 cell-cycle arrest without inducing Kaposi’s sarcoma-associated herpes virus lytic cycle reactivation.
Further investigation revealed that interferon α, β, and γ are cytotoxic to PEL, but they are not essential for the antiproliferative effects of IMiDs. On the other hand, cereblon-dependent suppression of IRF4 and rapid degradation of IKZF1 are essential.
The researchers also found that knocking down MYC enhanced IMiD activity. This led them to BRD4 inhibitors, which are known to target MYC transcription.
The team tested lenalidomide in combination with JQ-1, IBET151, and PFI-1. The IMiD was “highly synergistic” with all 3 BRD4 inhibitors in BC-3 and BCBL-1 cell lines.
In both cell lines, treatment with lenalidomide and JQ-1 significantly decreased the expression of MYC and IRF4 at protein and mRNA levels. The treatment also resulted in G1 cell cycle arrest, cleavage of poly (ADP-ribose) polymerase, and apoptosis.
So the researchers tested this combination in a mouse model of PEL (NOD-SCID mice injected with BC-3 cells). And they observed a significant improvement in median survival for mice that received combination treatment, compared to lenalidomide or JQ-1 alone—51 days, 35 days, and 42 days, respectively (P≤0.01).
Early progression predicts overall survival in FL

Photo by Rhoda Baer
The goal for many cancer patients is to reach the 5-year mark without progression, but a new study suggests 2 years might be a more appropriate goal for patients with follicular lymphoma (FL).
Previous research indicated that about 20% of FL patients relapse within 2 years of treatment.
Now, researchers have found these patients have a significantly worse 5-year survival rate than patients who make it past the 2-year mark without progressing.
Carla Casulo, MD, of the University of Rochester in New York, and her colleagues recounted these findings in the Journal of Clinical Oncology.
The team analyzed data from 588 patients with stage 2-4 FL who received first-line therapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
These patients could be separated into 2 groups: those with disease progression within 2 years of diagnosis (19%, n=110) and those who did not progress within the 2-year period (71%, n=420).
Eight percent of patients (n=46) were lost to follow-up, and 2% (n=12) died of causes other than progression within the 2-year period.
At 2 years, the overall survival (OS) rate was 68% in the early progression group and 97% in the group that did not progress. At 5 years, OS rates were 50% and 90%, respectively.
In unadjusted Cox regression analysis, early progression was associated with lower OS (hazard ratio[HR]=7.17). The same was true after the researchers adjusted for FLIPI score (HR=6.44).
The team observed similar results in a validation cohort of 147 FL patients who received first-line R-CHOP. At 2 years, the OS rate was 64% in the early progression group and 98% in the group that did not progress. At 5 years, OS rates were 34% and 94%, respectively.
Again, in an unadjusted analysis, early progression was associated with lower OS (HR=20.0). And this trend was maintained after the researchers adjusted for FLIPI score (HR=19.8).
The researchers also found that, for patients in the early progression group, clinical factors that were predictive of inferior OS were age, ECOG performance score, nodal sites, and disease stage. For the group that did not progress within 2 years, clinical factors that were predictive of OS were age and extranodal sites.
In a Cox regression analysis that encompassed these factors and early progression, only early progression, age, and ECOG performance scores remained significantly predictive of OS.
“[W]e have confirmed that all relapsed patients are not equal and therefore should not be approached the same at diagnosis, nor at the time of relapse, in terms of therapies,” Dr Casulo said.
“It will be critical to predict who is most likely to relapse early. We believe that targeted sequencing or gene-expression profiling will be important to understanding how to improve the outcomes of this group.”

Photo by Rhoda Baer
The goal for many cancer patients is to reach the 5-year mark without progression, but a new study suggests 2 years might be a more appropriate goal for patients with follicular lymphoma (FL).
Previous research indicated that about 20% of FL patients relapse within 2 years of treatment.
Now, researchers have found these patients have a significantly worse 5-year survival rate than patients who make it past the 2-year mark without progressing.
Carla Casulo, MD, of the University of Rochester in New York, and her colleagues recounted these findings in the Journal of Clinical Oncology.
The team analyzed data from 588 patients with stage 2-4 FL who received first-line therapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
These patients could be separated into 2 groups: those with disease progression within 2 years of diagnosis (19%, n=110) and those who did not progress within the 2-year period (71%, n=420).
Eight percent of patients (n=46) were lost to follow-up, and 2% (n=12) died of causes other than progression within the 2-year period.
At 2 years, the overall survival (OS) rate was 68% in the early progression group and 97% in the group that did not progress. At 5 years, OS rates were 50% and 90%, respectively.
In unadjusted Cox regression analysis, early progression was associated with lower OS (hazard ratio[HR]=7.17). The same was true after the researchers adjusted for FLIPI score (HR=6.44).
The team observed similar results in a validation cohort of 147 FL patients who received first-line R-CHOP. At 2 years, the OS rate was 64% in the early progression group and 98% in the group that did not progress. At 5 years, OS rates were 34% and 94%, respectively.
Again, in an unadjusted analysis, early progression was associated with lower OS (HR=20.0). And this trend was maintained after the researchers adjusted for FLIPI score (HR=19.8).
The researchers also found that, for patients in the early progression group, clinical factors that were predictive of inferior OS were age, ECOG performance score, nodal sites, and disease stage. For the group that did not progress within 2 years, clinical factors that were predictive of OS were age and extranodal sites.
In a Cox regression analysis that encompassed these factors and early progression, only early progression, age, and ECOG performance scores remained significantly predictive of OS.
“[W]e have confirmed that all relapsed patients are not equal and therefore should not be approached the same at diagnosis, nor at the time of relapse, in terms of therapies,” Dr Casulo said.
“It will be critical to predict who is most likely to relapse early. We believe that targeted sequencing or gene-expression profiling will be important to understanding how to improve the outcomes of this group.”

Photo by Rhoda Baer
The goal for many cancer patients is to reach the 5-year mark without progression, but a new study suggests 2 years might be a more appropriate goal for patients with follicular lymphoma (FL).
Previous research indicated that about 20% of FL patients relapse within 2 years of treatment.
Now, researchers have found these patients have a significantly worse 5-year survival rate than patients who make it past the 2-year mark without progressing.
Carla Casulo, MD, of the University of Rochester in New York, and her colleagues recounted these findings in the Journal of Clinical Oncology.
The team analyzed data from 588 patients with stage 2-4 FL who received first-line therapy with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).
These patients could be separated into 2 groups: those with disease progression within 2 years of diagnosis (19%, n=110) and those who did not progress within the 2-year period (71%, n=420).
Eight percent of patients (n=46) were lost to follow-up, and 2% (n=12) died of causes other than progression within the 2-year period.
At 2 years, the overall survival (OS) rate was 68% in the early progression group and 97% in the group that did not progress. At 5 years, OS rates were 50% and 90%, respectively.
In unadjusted Cox regression analysis, early progression was associated with lower OS (hazard ratio[HR]=7.17). The same was true after the researchers adjusted for FLIPI score (HR=6.44).
The team observed similar results in a validation cohort of 147 FL patients who received first-line R-CHOP. At 2 years, the OS rate was 64% in the early progression group and 98% in the group that did not progress. At 5 years, OS rates were 34% and 94%, respectively.
Again, in an unadjusted analysis, early progression was associated with lower OS (HR=20.0). And this trend was maintained after the researchers adjusted for FLIPI score (HR=19.8).
The researchers also found that, for patients in the early progression group, clinical factors that were predictive of inferior OS were age, ECOG performance score, nodal sites, and disease stage. For the group that did not progress within 2 years, clinical factors that were predictive of OS were age and extranodal sites.
In a Cox regression analysis that encompassed these factors and early progression, only early progression, age, and ECOG performance scores remained significantly predictive of OS.
“[W]e have confirmed that all relapsed patients are not equal and therefore should not be approached the same at diagnosis, nor at the time of relapse, in terms of therapies,” Dr Casulo said.
“It will be critical to predict who is most likely to relapse early. We believe that targeted sequencing or gene-expression profiling will be important to understanding how to improve the outcomes of this group.”
RBAC500 safe, effective for elderly patients with mantle cell lymphoma
Reducing the dose of cytarabine from 800 mg/m2 to 500 mg/m2 allowed a regimen of rituximab, bendamustine, and cytarabine to be safely administered as first-line therapy to elderly patients who had mantle cell lymphoma and were not candidates for autologous stem cell transplant, according to Dr. Carlo Visco of the San Bortolo Hospital in Vicenza, Italy.
“Hematologic toxicity was substantially reduced, compared to the earlier study, Dr. Visco said, calling the R-BAC500 regimen “a highly effective treatment” for patients with mantle cell lymphoma.
Speaking at the at the International Congress on Malignant Lymphoma in Lugano, Switzerland, Dr. Visco noted the “encouraging results, but high hematologic toxicity” seen in a previous study that employed the higher cytarabine dose. In that previous study, transient grades 3-4 thrombocytopenia occurred in 76% of cycles.
In an attempt to reduce hematologic toxicity, the Fondazione Italiana Linfomi designed a phase II trial in which the cytarabine dose was lowered to 500 mg/m2 (R-BAC500). The administration schedule of cytarabine (on days 2-4) and the other components of the original regimen (rituximab, 375 mg/m2, on day 1 and bendamustine, 70 mg/m2, on days 2 and 3) remained unchanged.
The 57 study subjects, median age 71, had newly diagnosed mantle cell lymphoma, and were not eligible for autologous transplant as determined by the comprehensive geriatric assessment; 75% of the patients were males and 91% had Ann Arbor stage III/IV disease.
The Mantle Cell International Prognostic Index (MIPI) was low in 15%, intermediate in 40%, and high in 45%; 9% had the blastoid variant of the disease.
The primary endpoints were complete remission rate, as measured by 18-fluorodeoxyglucose–PET, according to Cheson criteria 2007, and safety. Secondary endpoints included molecular response rate, progression-free survival, and overall survival.
The overall response rate was 96%, and the complete remission rate was 93%. The molecular response rate at the end of treatment was 76% on peripheral blood and 55% on bone marrow samples. With a median follow-up of 18 months, the projected 2-year progression-free survival was 83%, and the overall survival was 91% without maintenance therapy.
Nearly all patients, 53 of 57, received at least four cycles of therapy, and 36 had six cycles. Treatment was discontinued because of toxicity (primarily hematologic) in 15 patients. Only one patient discontinued because of progressive disease.
Grade 3 or 4 neutropenia and thrombocytopenia were observed in about half of administered cycles. Febrile neutropenia occurred in 6%. Extrahematologic toxicity was mainly cardiac (5%).
BR is a commonly used regimen for older, less fit patients with MCL. Inclusion of high dose cytarabine appears to be beneficial n younger patients with MCL, particularly in induction pre-SCT. The FIL has been investigating intermediate doses of cytarabine combined, rather than alternating, with BR. This phase 2 study utilized cytarabine 500 mg/m2 daily x 3 with BR (slightly lower than standard dose bendamustine). The patient population was older with predominantly intermediate-high MIPI, yet results were impressive, particularly the PET negative rate of 93% and marrow MRD negative rate of 55%. Follow-up is short, but remissions do appear durable. Concerns are the high number of patients unable to complete planned therapy, the high rate of grade 3 and 4 cytopenias, and the frequency of visits required for close blood count monitoring and blood product support.
BR is a commonly used regimen for older, less fit patients with MCL. Inclusion of high dose cytarabine appears to be beneficial n younger patients with MCL, particularly in induction pre-SCT. The FIL has been investigating intermediate doses of cytarabine combined, rather than alternating, with BR. This phase 2 study utilized cytarabine 500 mg/m2 daily x 3 with BR (slightly lower than standard dose bendamustine). The patient population was older with predominantly intermediate-high MIPI, yet results were impressive, particularly the PET negative rate of 93% and marrow MRD negative rate of 55%. Follow-up is short, but remissions do appear durable. Concerns are the high number of patients unable to complete planned therapy, the high rate of grade 3 and 4 cytopenias, and the frequency of visits required for close blood count monitoring and blood product support.
BR is a commonly used regimen for older, less fit patients with MCL. Inclusion of high dose cytarabine appears to be beneficial n younger patients with MCL, particularly in induction pre-SCT. The FIL has been investigating intermediate doses of cytarabine combined, rather than alternating, with BR. This phase 2 study utilized cytarabine 500 mg/m2 daily x 3 with BR (slightly lower than standard dose bendamustine). The patient population was older with predominantly intermediate-high MIPI, yet results were impressive, particularly the PET negative rate of 93% and marrow MRD negative rate of 55%. Follow-up is short, but remissions do appear durable. Concerns are the high number of patients unable to complete planned therapy, the high rate of grade 3 and 4 cytopenias, and the frequency of visits required for close blood count monitoring and blood product support.
Reducing the dose of cytarabine from 800 mg/m2 to 500 mg/m2 allowed a regimen of rituximab, bendamustine, and cytarabine to be safely administered as first-line therapy to elderly patients who had mantle cell lymphoma and were not candidates for autologous stem cell transplant, according to Dr. Carlo Visco of the San Bortolo Hospital in Vicenza, Italy.
“Hematologic toxicity was substantially reduced, compared to the earlier study, Dr. Visco said, calling the R-BAC500 regimen “a highly effective treatment” for patients with mantle cell lymphoma.
Speaking at the at the International Congress on Malignant Lymphoma in Lugano, Switzerland, Dr. Visco noted the “encouraging results, but high hematologic toxicity” seen in a previous study that employed the higher cytarabine dose. In that previous study, transient grades 3-4 thrombocytopenia occurred in 76% of cycles.
In an attempt to reduce hematologic toxicity, the Fondazione Italiana Linfomi designed a phase II trial in which the cytarabine dose was lowered to 500 mg/m2 (R-BAC500). The administration schedule of cytarabine (on days 2-4) and the other components of the original regimen (rituximab, 375 mg/m2, on day 1 and bendamustine, 70 mg/m2, on days 2 and 3) remained unchanged.
The 57 study subjects, median age 71, had newly diagnosed mantle cell lymphoma, and were not eligible for autologous transplant as determined by the comprehensive geriatric assessment; 75% of the patients were males and 91% had Ann Arbor stage III/IV disease.
The Mantle Cell International Prognostic Index (MIPI) was low in 15%, intermediate in 40%, and high in 45%; 9% had the blastoid variant of the disease.
The primary endpoints were complete remission rate, as measured by 18-fluorodeoxyglucose–PET, according to Cheson criteria 2007, and safety. Secondary endpoints included molecular response rate, progression-free survival, and overall survival.
The overall response rate was 96%, and the complete remission rate was 93%. The molecular response rate at the end of treatment was 76% on peripheral blood and 55% on bone marrow samples. With a median follow-up of 18 months, the projected 2-year progression-free survival was 83%, and the overall survival was 91% without maintenance therapy.
Nearly all patients, 53 of 57, received at least four cycles of therapy, and 36 had six cycles. Treatment was discontinued because of toxicity (primarily hematologic) in 15 patients. Only one patient discontinued because of progressive disease.
Grade 3 or 4 neutropenia and thrombocytopenia were observed in about half of administered cycles. Febrile neutropenia occurred in 6%. Extrahematologic toxicity was mainly cardiac (5%).
Reducing the dose of cytarabine from 800 mg/m2 to 500 mg/m2 allowed a regimen of rituximab, bendamustine, and cytarabine to be safely administered as first-line therapy to elderly patients who had mantle cell lymphoma and were not candidates for autologous stem cell transplant, according to Dr. Carlo Visco of the San Bortolo Hospital in Vicenza, Italy.
“Hematologic toxicity was substantially reduced, compared to the earlier study, Dr. Visco said, calling the R-BAC500 regimen “a highly effective treatment” for patients with mantle cell lymphoma.
Speaking at the at the International Congress on Malignant Lymphoma in Lugano, Switzerland, Dr. Visco noted the “encouraging results, but high hematologic toxicity” seen in a previous study that employed the higher cytarabine dose. In that previous study, transient grades 3-4 thrombocytopenia occurred in 76% of cycles.
In an attempt to reduce hematologic toxicity, the Fondazione Italiana Linfomi designed a phase II trial in which the cytarabine dose was lowered to 500 mg/m2 (R-BAC500). The administration schedule of cytarabine (on days 2-4) and the other components of the original regimen (rituximab, 375 mg/m2, on day 1 and bendamustine, 70 mg/m2, on days 2 and 3) remained unchanged.
The 57 study subjects, median age 71, had newly diagnosed mantle cell lymphoma, and were not eligible for autologous transplant as determined by the comprehensive geriatric assessment; 75% of the patients were males and 91% had Ann Arbor stage III/IV disease.
The Mantle Cell International Prognostic Index (MIPI) was low in 15%, intermediate in 40%, and high in 45%; 9% had the blastoid variant of the disease.
The primary endpoints were complete remission rate, as measured by 18-fluorodeoxyglucose–PET, according to Cheson criteria 2007, and safety. Secondary endpoints included molecular response rate, progression-free survival, and overall survival.
The overall response rate was 96%, and the complete remission rate was 93%. The molecular response rate at the end of treatment was 76% on peripheral blood and 55% on bone marrow samples. With a median follow-up of 18 months, the projected 2-year progression-free survival was 83%, and the overall survival was 91% without maintenance therapy.
Nearly all patients, 53 of 57, received at least four cycles of therapy, and 36 had six cycles. Treatment was discontinued because of toxicity (primarily hematologic) in 15 patients. Only one patient discontinued because of progressive disease.
Grade 3 or 4 neutropenia and thrombocytopenia were observed in about half of administered cycles. Febrile neutropenia occurred in 6%. Extrahematologic toxicity was mainly cardiac (5%).
FROM 13-ICML
Key clinical point: Reducing the dose of cytarabine from 800 mg/m2 to 500 mg/m2 allowed a regimen of rituximab, bendamustine, and cytarabine to be safely administered as first-line therapy to elderly patients with mantle cell lymphoma.
Major finding: Nearly all patients, 53 of 57, received at least four cycles of therapy, and 36 had six cycles. Treatment was discontinued because of toxicity (primarily hematologic) in 15 patients.
Data source: 57 study subjects, median age 71, who had newly diagnosed mantle cell lymphoma and were not eligible for autologous transplant as determined by the comprehensive geriatric assessment.
Disclosures: The trial was conducted by the Fondazione Italiana Linfomi. There were no relevant financial disclosures.