User login
Nanoparticle-based vaccine could prevent EBV

Photo by Sakura Midori
Researchers say they have developed a nanoparticle-based vaccine against Epstein-Barr virus (EBV) that can induce potent neutralizing antibodies in mice and monkeys.
These results suggest that using a structure-based vaccine design and self-assembling nanoparticles to deliver a viral protein that prompts an immune response could be a promising approach for developing an EBV vaccine for humans.
Most efforts to develop a preventive EBV vaccine have focused on glycoprotein 350 (gp350), a molecule on the surface of EBV that helps the virus attach to B cells. EBV gp350 is thought to be a key target for antibodies capable of preventing viral infection.
Previously, researchers showed that vaccinating monkeys with gp350 protected the animals from developing lymphomas after exposure to a high dose of EBV.
However, in the only large human trial of an experimental EBV vaccine conducted to date, the EBV gp350 vaccine did not prevent EBV infection, although it did reduce the rate of infectious mononucleosis by 78%.
With this in mind, Masaru Kanekiyo, DVM, PhD, of the National Institutes of Health in Bethesda, Maryland, and his colleagues set out to create a better vaccine.
They described their work in a paper published in Cell.
The team designed a nanoparticle-based vaccine that expressed the cell-binding portion of gp350. In tests, the experimental vaccine induced potent neutralizing antibodies in both mice and cynomolgus macaques (Macaca fascicularis).
In fact, compared with soluble gp350, the nanoparticle-based vaccine induced 10- to 100-fold higher levels of neutralizing antibodies in mice.
The researchers believe the nanoparticle vaccine design could be used to create or redesign vaccines against other pathogens as well. ![]()

Photo by Sakura Midori
Researchers say they have developed a nanoparticle-based vaccine against Epstein-Barr virus (EBV) that can induce potent neutralizing antibodies in mice and monkeys.
These results suggest that using a structure-based vaccine design and self-assembling nanoparticles to deliver a viral protein that prompts an immune response could be a promising approach for developing an EBV vaccine for humans.
Most efforts to develop a preventive EBV vaccine have focused on glycoprotein 350 (gp350), a molecule on the surface of EBV that helps the virus attach to B cells. EBV gp350 is thought to be a key target for antibodies capable of preventing viral infection.
Previously, researchers showed that vaccinating monkeys with gp350 protected the animals from developing lymphomas after exposure to a high dose of EBV.
However, in the only large human trial of an experimental EBV vaccine conducted to date, the EBV gp350 vaccine did not prevent EBV infection, although it did reduce the rate of infectious mononucleosis by 78%.
With this in mind, Masaru Kanekiyo, DVM, PhD, of the National Institutes of Health in Bethesda, Maryland, and his colleagues set out to create a better vaccine.
They described their work in a paper published in Cell.
The team designed a nanoparticle-based vaccine that expressed the cell-binding portion of gp350. In tests, the experimental vaccine induced potent neutralizing antibodies in both mice and cynomolgus macaques (Macaca fascicularis).
In fact, compared with soluble gp350, the nanoparticle-based vaccine induced 10- to 100-fold higher levels of neutralizing antibodies in mice.
The researchers believe the nanoparticle vaccine design could be used to create or redesign vaccines against other pathogens as well. ![]()

Photo by Sakura Midori
Researchers say they have developed a nanoparticle-based vaccine against Epstein-Barr virus (EBV) that can induce potent neutralizing antibodies in mice and monkeys.
These results suggest that using a structure-based vaccine design and self-assembling nanoparticles to deliver a viral protein that prompts an immune response could be a promising approach for developing an EBV vaccine for humans.
Most efforts to develop a preventive EBV vaccine have focused on glycoprotein 350 (gp350), a molecule on the surface of EBV that helps the virus attach to B cells. EBV gp350 is thought to be a key target for antibodies capable of preventing viral infection.
Previously, researchers showed that vaccinating monkeys with gp350 protected the animals from developing lymphomas after exposure to a high dose of EBV.
However, in the only large human trial of an experimental EBV vaccine conducted to date, the EBV gp350 vaccine did not prevent EBV infection, although it did reduce the rate of infectious mononucleosis by 78%.
With this in mind, Masaru Kanekiyo, DVM, PhD, of the National Institutes of Health in Bethesda, Maryland, and his colleagues set out to create a better vaccine.
They described their work in a paper published in Cell.
The team designed a nanoparticle-based vaccine that expressed the cell-binding portion of gp350. In tests, the experimental vaccine induced potent neutralizing antibodies in both mice and cynomolgus macaques (Macaca fascicularis).
In fact, compared with soluble gp350, the nanoparticle-based vaccine induced 10- to 100-fold higher levels of neutralizing antibodies in mice.
The researchers believe the nanoparticle vaccine design could be used to create or redesign vaccines against other pathogens as well. ![]()
Tool that lets patients report AEs proves reliable

receiving chemotherapy
Photo by Rhoda Baer
Results of a multicenter study indicate that a tool cancer patients can use to report adverse events (AEs) is as accurate as other, established patient-reported and clinical measures.
The tool is the National Cancer Institute’s Patient Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE).
Study investigators were able to validate 119 of 124 PRO-CTCAE questions against 2 established measurement tools.
The 5 questions that were not validated could not be evaluated due to underrepresentation in the study population.
This research was published in JAMA Oncology.
“In most cancer clinical trials, information on side effects is collected by providers who have limited time with their patients, and current patient questionnaires are limited in scope and depth,” said study author Amylou Dueck, PhD, of the Mayo Clinic in Scottsdale, Arizona.
“PRO-CTCAE is a library of items for patients to directly report on the level of each of their symptoms, to enhance the reporting of side effects in cancer clinical trials, which is normally based on information from providers. The study itself is unprecedented, as more than 100 distinct questions about symptomatic adverse events were validated simultaneously.”
To assess the PRO-CTCAE, Dr Dueck and her colleagues recruited 975 cancer patients from 9 clinical practices across the US, including 7 cancer centers.
The patients had a range of cancers and were undergoing outpatient chemotherapy and/or radiation therapy. The investigators said these participants reflected the geographic, ethnic, racial, and economic diversity in cancer clinical trials.
The patients were asked to fill out the PRO-CTCAE questionnaire before appointments. The investigators then compared patient reports to clinician-reported Eastern Cooperative Oncology Group (ECOG) performance status and the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire (QLQ-C30).
A majority of patients completed items on the PRO-CTCAE questionnaire at their first visit (96.4%, 940/975) and second visit (90.6%, 852/940).
Most patients (99.8%, 938/940) reported having at least 1 symptomatic AE, with 81.7% (768/940) reporting at least 1 AE as frequent, severe, and/or interfering “quite a bit” with daily activities.
To gauge the accuracy of the PRO-CTCAE, the investigators assessed construct validity, test-retest reliability, and responsiveness of PRO-CTCAE items.
Construct validity
The investigators explained that construct validity reflects the association between a new measurement tool and an established measure.
Construct validity is often investigated through convergent validity, which determines whether the new tool moves in the same direction as an established instrument, and known-groups validity, which determines whether the tool can distinguish between groups of patients who are thought to be distinct.
When the investigators considered all QLQ-C30 functioning/global scales, they found that all 124 items on the PRO-CTCAE questionnaire were associated in the expected direction with 1 or more scales. One hundred and fourteen of the PRO-CTCAE items demonstrated a meaningful correlation (Pearson r≥0.1), and 111 of them were statistically significant (P<0.05 for all).
Scores for 94 of 124 PRO-CTCAE items were higher among patients with an ECOG performance status of 2 to 4 (17.1% of patients) than among patients with a score of 0 to 1. The difference was significant for 58 of the items (P<0.05 for all).
Test-retest reliability and responsiveness
The investigators said they estimated test-retest reliability using the intraclass correlation coefficient (ICC), based on a 1-way analysis of variance model with an ICC of 0.7 or greater interpreted as high.
Test-retest reliability was 0.7 or greater for 36 of 49 prespecified PRO-CTCAE items. The median ICC was 0.76 [range, 0.53-0.96).
The investigators assessed the responsiveness of PRO-CTCAE items by comparing any change from the first visit to the second visit in 27 items that were selected a priori.
Correlations between PRO-CTCAE item changes and corresponding QLQ-C30 scale changes were significant for all 27 items (P≤0.006 for all).
“This is a landmark study demonstrating that meaningful information about adverse events can be elicited from patients themselves, which is a major step for advancing the patient-centeredness of clinical trials,” said study author Ethan Basch, MD, of the Lineberger Cancer Center of the University of North Carolina in Chapel Hill. ![]()

receiving chemotherapy
Photo by Rhoda Baer
Results of a multicenter study indicate that a tool cancer patients can use to report adverse events (AEs) is as accurate as other, established patient-reported and clinical measures.
The tool is the National Cancer Institute’s Patient Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE).
Study investigators were able to validate 119 of 124 PRO-CTCAE questions against 2 established measurement tools.
The 5 questions that were not validated could not be evaluated due to underrepresentation in the study population.
This research was published in JAMA Oncology.
“In most cancer clinical trials, information on side effects is collected by providers who have limited time with their patients, and current patient questionnaires are limited in scope and depth,” said study author Amylou Dueck, PhD, of the Mayo Clinic in Scottsdale, Arizona.
“PRO-CTCAE is a library of items for patients to directly report on the level of each of their symptoms, to enhance the reporting of side effects in cancer clinical trials, which is normally based on information from providers. The study itself is unprecedented, as more than 100 distinct questions about symptomatic adverse events were validated simultaneously.”
To assess the PRO-CTCAE, Dr Dueck and her colleagues recruited 975 cancer patients from 9 clinical practices across the US, including 7 cancer centers.
The patients had a range of cancers and were undergoing outpatient chemotherapy and/or radiation therapy. The investigators said these participants reflected the geographic, ethnic, racial, and economic diversity in cancer clinical trials.
The patients were asked to fill out the PRO-CTCAE questionnaire before appointments. The investigators then compared patient reports to clinician-reported Eastern Cooperative Oncology Group (ECOG) performance status and the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire (QLQ-C30).
A majority of patients completed items on the PRO-CTCAE questionnaire at their first visit (96.4%, 940/975) and second visit (90.6%, 852/940).
Most patients (99.8%, 938/940) reported having at least 1 symptomatic AE, with 81.7% (768/940) reporting at least 1 AE as frequent, severe, and/or interfering “quite a bit” with daily activities.
To gauge the accuracy of the PRO-CTCAE, the investigators assessed construct validity, test-retest reliability, and responsiveness of PRO-CTCAE items.
Construct validity
The investigators explained that construct validity reflects the association between a new measurement tool and an established measure.
Construct validity is often investigated through convergent validity, which determines whether the new tool moves in the same direction as an established instrument, and known-groups validity, which determines whether the tool can distinguish between groups of patients who are thought to be distinct.
When the investigators considered all QLQ-C30 functioning/global scales, they found that all 124 items on the PRO-CTCAE questionnaire were associated in the expected direction with 1 or more scales. One hundred and fourteen of the PRO-CTCAE items demonstrated a meaningful correlation (Pearson r≥0.1), and 111 of them were statistically significant (P<0.05 for all).
Scores for 94 of 124 PRO-CTCAE items were higher among patients with an ECOG performance status of 2 to 4 (17.1% of patients) than among patients with a score of 0 to 1. The difference was significant for 58 of the items (P<0.05 for all).
Test-retest reliability and responsiveness
The investigators said they estimated test-retest reliability using the intraclass correlation coefficient (ICC), based on a 1-way analysis of variance model with an ICC of 0.7 or greater interpreted as high.
Test-retest reliability was 0.7 or greater for 36 of 49 prespecified PRO-CTCAE items. The median ICC was 0.76 [range, 0.53-0.96).
The investigators assessed the responsiveness of PRO-CTCAE items by comparing any change from the first visit to the second visit in 27 items that were selected a priori.
Correlations between PRO-CTCAE item changes and corresponding QLQ-C30 scale changes were significant for all 27 items (P≤0.006 for all).
“This is a landmark study demonstrating that meaningful information about adverse events can be elicited from patients themselves, which is a major step for advancing the patient-centeredness of clinical trials,” said study author Ethan Basch, MD, of the Lineberger Cancer Center of the University of North Carolina in Chapel Hill. ![]()

receiving chemotherapy
Photo by Rhoda Baer
Results of a multicenter study indicate that a tool cancer patients can use to report adverse events (AEs) is as accurate as other, established patient-reported and clinical measures.
The tool is the National Cancer Institute’s Patient Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE).
Study investigators were able to validate 119 of 124 PRO-CTCAE questions against 2 established measurement tools.
The 5 questions that were not validated could not be evaluated due to underrepresentation in the study population.
This research was published in JAMA Oncology.
“In most cancer clinical trials, information on side effects is collected by providers who have limited time with their patients, and current patient questionnaires are limited in scope and depth,” said study author Amylou Dueck, PhD, of the Mayo Clinic in Scottsdale, Arizona.
“PRO-CTCAE is a library of items for patients to directly report on the level of each of their symptoms, to enhance the reporting of side effects in cancer clinical trials, which is normally based on information from providers. The study itself is unprecedented, as more than 100 distinct questions about symptomatic adverse events were validated simultaneously.”
To assess the PRO-CTCAE, Dr Dueck and her colleagues recruited 975 cancer patients from 9 clinical practices across the US, including 7 cancer centers.
The patients had a range of cancers and were undergoing outpatient chemotherapy and/or radiation therapy. The investigators said these participants reflected the geographic, ethnic, racial, and economic diversity in cancer clinical trials.
The patients were asked to fill out the PRO-CTCAE questionnaire before appointments. The investigators then compared patient reports to clinician-reported Eastern Cooperative Oncology Group (ECOG) performance status and the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire (QLQ-C30).
A majority of patients completed items on the PRO-CTCAE questionnaire at their first visit (96.4%, 940/975) and second visit (90.6%, 852/940).
Most patients (99.8%, 938/940) reported having at least 1 symptomatic AE, with 81.7% (768/940) reporting at least 1 AE as frequent, severe, and/or interfering “quite a bit” with daily activities.
To gauge the accuracy of the PRO-CTCAE, the investigators assessed construct validity, test-retest reliability, and responsiveness of PRO-CTCAE items.
Construct validity
The investigators explained that construct validity reflects the association between a new measurement tool and an established measure.
Construct validity is often investigated through convergent validity, which determines whether the new tool moves in the same direction as an established instrument, and known-groups validity, which determines whether the tool can distinguish between groups of patients who are thought to be distinct.
When the investigators considered all QLQ-C30 functioning/global scales, they found that all 124 items on the PRO-CTCAE questionnaire were associated in the expected direction with 1 or more scales. One hundred and fourteen of the PRO-CTCAE items demonstrated a meaningful correlation (Pearson r≥0.1), and 111 of them were statistically significant (P<0.05 for all).
Scores for 94 of 124 PRO-CTCAE items were higher among patients with an ECOG performance status of 2 to 4 (17.1% of patients) than among patients with a score of 0 to 1. The difference was significant for 58 of the items (P<0.05 for all).
Test-retest reliability and responsiveness
The investigators said they estimated test-retest reliability using the intraclass correlation coefficient (ICC), based on a 1-way analysis of variance model with an ICC of 0.7 or greater interpreted as high.
Test-retest reliability was 0.7 or greater for 36 of 49 prespecified PRO-CTCAE items. The median ICC was 0.76 [range, 0.53-0.96).
The investigators assessed the responsiveness of PRO-CTCAE items by comparing any change from the first visit to the second visit in 27 items that were selected a priori.
Correlations between PRO-CTCAE item changes and corresponding QLQ-C30 scale changes were significant for all 27 items (P≤0.006 for all).
“This is a landmark study demonstrating that meaningful information about adverse events can be elicited from patients themselves, which is a major step for advancing the patient-centeredness of clinical trials,” said study author Ethan Basch, MD, of the Lineberger Cancer Center of the University of North Carolina in Chapel Hill. ![]()
New prognostic model for follicular lymphoma

Photo courtesy of NIH
A newly developed prognostic model can identify follicular lymphoma (FL) patients at the highest risk for treatment failure, according to researchers.
To create this model, called m7-FLIPI, the team combined the Follicular Lymphoma International Prognostic Index (FLIPI), Eastern Cooperative Oncology Group (ECOG) performance status, and the mutation status of 7 genes—EZH2, ARID1A, MEF2B, EP300, FOXO1, CREBBP, and CARD11.
The researchers said this is the first prognostic model for FL that accounts for both clinical factors and genetic mutations.
They described the creation and testing of the model in The Lancet Oncology.
“We set out to determine, at the time of diagnosis, which patients’ disease will have sustained responses after treatment and whether new genetic data could help inform which patients are at risk for developing progressive lymphoma so clinicians would be able to offer these high-risk patients more effective therapies,” said Randy Gascoyne, MD, of the British Columbia Cancer Agency in Vancouver, Canada.
He and his colleagues created the m7-FLIPI by conducting a retrospective analysis of genetic mutations and clinical risk factors in 2 cohorts of patients with symptomatic, advanced stage, or bulky FL grade 1, 2, or 3A.
The patients had a biopsy specimen collected 12 months or less before they began first-line treatment with an immunochemotherapy regimen containing rituximab.
Training cohort
The training cohort consisted of 151 FL patients who received R-CHOP. The median follow-up for these patients was 7.7 years.
When the researchers applied the m7-FLIPI to this cohort, they found 28% of patients (43/151) were defined as high-risk, with a 5-year failure-free survival (FFS) rate of 38.29%.
And 72% of patients (108/151) were defined as low-risk, with a 5-year FFS of 77.21%. The hazard ratio was 4.14 (P<0.0001).
The positive predictive value for 5-year FFS was 64%, and the negative predictive value was 78%. The m7-FLIPI outperformed a prognostic model of only gene mutations and the FLIPI-2.
Validation cohort
The validation cohort consisted of 107 patients who received R-CVP. The median follow-up for these patients was 6.7 years.
When the researchers applied the m7-FLIPI to this cohort, they found that 22% of patients (24/107) were defined as high-risk, with a 5-year FFS of 25%.
And 78% of patients (83/107) were defined as low-risk, with a 5-year FFS of 68.24%. The hazard ratio was 3.58 (P<0.0001).
The positive predictive value for 5-year FFS was 72%, and the negative predictive value was 68%. The m7-FLIPI outperformed the FLIPI alone and the FLIPI combined with ECOG performance status.
Overall survival
Although the m7-FLIPI was designed specifically for FFS, the researchers also tested its prognostic utility for overall survival (OS).
In the training cohort, high-risk disease according to the m7-FLIPI was associated with a 5-year OS of 65.25%, compared to 89.98% for low-risk disease (P=0.00031).
In the validation cohort, 5-year OS was 41.67% for patients with high-risk disease and 84.01% for patients with low-risk disease (P<0.0001). In both cohorts, the m7-FLIPI outperformed the FLIPI alone.
Based on these results, the researchers believe the m7-FLIPI could be utilized in a clinical setting to test all new FL patients at diagnosis and identify patients who harbor the most aggressive disease.
“The m7-FLIPI could be extremely significant for the medical community,” Dr Gascoyne said, “changing the story for high-risk patients who are currently destined to not respond well to standard treatment.” ![]()

Photo courtesy of NIH
A newly developed prognostic model can identify follicular lymphoma (FL) patients at the highest risk for treatment failure, according to researchers.
To create this model, called m7-FLIPI, the team combined the Follicular Lymphoma International Prognostic Index (FLIPI), Eastern Cooperative Oncology Group (ECOG) performance status, and the mutation status of 7 genes—EZH2, ARID1A, MEF2B, EP300, FOXO1, CREBBP, and CARD11.
The researchers said this is the first prognostic model for FL that accounts for both clinical factors and genetic mutations.
They described the creation and testing of the model in The Lancet Oncology.
“We set out to determine, at the time of diagnosis, which patients’ disease will have sustained responses after treatment and whether new genetic data could help inform which patients are at risk for developing progressive lymphoma so clinicians would be able to offer these high-risk patients more effective therapies,” said Randy Gascoyne, MD, of the British Columbia Cancer Agency in Vancouver, Canada.
He and his colleagues created the m7-FLIPI by conducting a retrospective analysis of genetic mutations and clinical risk factors in 2 cohorts of patients with symptomatic, advanced stage, or bulky FL grade 1, 2, or 3A.
The patients had a biopsy specimen collected 12 months or less before they began first-line treatment with an immunochemotherapy regimen containing rituximab.
Training cohort
The training cohort consisted of 151 FL patients who received R-CHOP. The median follow-up for these patients was 7.7 years.
When the researchers applied the m7-FLIPI to this cohort, they found 28% of patients (43/151) were defined as high-risk, with a 5-year failure-free survival (FFS) rate of 38.29%.
And 72% of patients (108/151) were defined as low-risk, with a 5-year FFS of 77.21%. The hazard ratio was 4.14 (P<0.0001).
The positive predictive value for 5-year FFS was 64%, and the negative predictive value was 78%. The m7-FLIPI outperformed a prognostic model of only gene mutations and the FLIPI-2.
Validation cohort
The validation cohort consisted of 107 patients who received R-CVP. The median follow-up for these patients was 6.7 years.
When the researchers applied the m7-FLIPI to this cohort, they found that 22% of patients (24/107) were defined as high-risk, with a 5-year FFS of 25%.
And 78% of patients (83/107) were defined as low-risk, with a 5-year FFS of 68.24%. The hazard ratio was 3.58 (P<0.0001).
The positive predictive value for 5-year FFS was 72%, and the negative predictive value was 68%. The m7-FLIPI outperformed the FLIPI alone and the FLIPI combined with ECOG performance status.
Overall survival
Although the m7-FLIPI was designed specifically for FFS, the researchers also tested its prognostic utility for overall survival (OS).
In the training cohort, high-risk disease according to the m7-FLIPI was associated with a 5-year OS of 65.25%, compared to 89.98% for low-risk disease (P=0.00031).
In the validation cohort, 5-year OS was 41.67% for patients with high-risk disease and 84.01% for patients with low-risk disease (P<0.0001). In both cohorts, the m7-FLIPI outperformed the FLIPI alone.
Based on these results, the researchers believe the m7-FLIPI could be utilized in a clinical setting to test all new FL patients at diagnosis and identify patients who harbor the most aggressive disease.
“The m7-FLIPI could be extremely significant for the medical community,” Dr Gascoyne said, “changing the story for high-risk patients who are currently destined to not respond well to standard treatment.” ![]()

Photo courtesy of NIH
A newly developed prognostic model can identify follicular lymphoma (FL) patients at the highest risk for treatment failure, according to researchers.
To create this model, called m7-FLIPI, the team combined the Follicular Lymphoma International Prognostic Index (FLIPI), Eastern Cooperative Oncology Group (ECOG) performance status, and the mutation status of 7 genes—EZH2, ARID1A, MEF2B, EP300, FOXO1, CREBBP, and CARD11.
The researchers said this is the first prognostic model for FL that accounts for both clinical factors and genetic mutations.
They described the creation and testing of the model in The Lancet Oncology.
“We set out to determine, at the time of diagnosis, which patients’ disease will have sustained responses after treatment and whether new genetic data could help inform which patients are at risk for developing progressive lymphoma so clinicians would be able to offer these high-risk patients more effective therapies,” said Randy Gascoyne, MD, of the British Columbia Cancer Agency in Vancouver, Canada.
He and his colleagues created the m7-FLIPI by conducting a retrospective analysis of genetic mutations and clinical risk factors in 2 cohorts of patients with symptomatic, advanced stage, or bulky FL grade 1, 2, or 3A.
The patients had a biopsy specimen collected 12 months or less before they began first-line treatment with an immunochemotherapy regimen containing rituximab.
Training cohort
The training cohort consisted of 151 FL patients who received R-CHOP. The median follow-up for these patients was 7.7 years.
When the researchers applied the m7-FLIPI to this cohort, they found 28% of patients (43/151) were defined as high-risk, with a 5-year failure-free survival (FFS) rate of 38.29%.
And 72% of patients (108/151) were defined as low-risk, with a 5-year FFS of 77.21%. The hazard ratio was 4.14 (P<0.0001).
The positive predictive value for 5-year FFS was 64%, and the negative predictive value was 78%. The m7-FLIPI outperformed a prognostic model of only gene mutations and the FLIPI-2.
Validation cohort
The validation cohort consisted of 107 patients who received R-CVP. The median follow-up for these patients was 6.7 years.
When the researchers applied the m7-FLIPI to this cohort, they found that 22% of patients (24/107) were defined as high-risk, with a 5-year FFS of 25%.
And 78% of patients (83/107) were defined as low-risk, with a 5-year FFS of 68.24%. The hazard ratio was 3.58 (P<0.0001).
The positive predictive value for 5-year FFS was 72%, and the negative predictive value was 68%. The m7-FLIPI outperformed the FLIPI alone and the FLIPI combined with ECOG performance status.
Overall survival
Although the m7-FLIPI was designed specifically for FFS, the researchers also tested its prognostic utility for overall survival (OS).
In the training cohort, high-risk disease according to the m7-FLIPI was associated with a 5-year OS of 65.25%, compared to 89.98% for low-risk disease (P=0.00031).
In the validation cohort, 5-year OS was 41.67% for patients with high-risk disease and 84.01% for patients with low-risk disease (P<0.0001). In both cohorts, the m7-FLIPI outperformed the FLIPI alone.
Based on these results, the researchers believe the m7-FLIPI could be utilized in a clinical setting to test all new FL patients at diagnosis and identify patients who harbor the most aggressive disease.
“The m7-FLIPI could be extremely significant for the medical community,” Dr Gascoyne said, “changing the story for high-risk patients who are currently destined to not respond well to standard treatment.” ![]()
How CLL evades the immune system

Photo courtesy of
Monash University
A study published in Leukemia has revealed a mechanism by which chronic lymphocytic leukemia (CLL) evades the immune system.
“It turns out that cancer cells are very good at sabotaging the immune system, using various tricks that confuse immune cells and ‘smoke screens’ preventing immune cells from recognizing the cancer,” said study author Fabienne Mackay, PhD, of Monash University in Melbourne, Victoria, Australia.
She and her colleagues believe they have determined exactly how CLL confuses the immune system and devised a way to stop it without destroying the patient’s immune system.
The team noted that B cells rely on the protein BAFF to survive. And each B cell has 3 different kinds of receptors that detect the presence of BAFF in the blood—TACI, BAFF-R, and BCMA.
The researchers discovered that, in CLL patients, the TACI receptors of cancerous B cells over-produce interleukin-10 (IL-10), which tricks the immune system into thinking nothing is wrong, allowing CLL to thrive undetected.
“We found that, when the receptor called TACI was blocked, it prevented the secretion of IL-10 without eliminating normal B cells,” Dr Mackay said. “Without IL-10, the tumor can no longer keep the immune system at bay, which means the patient’s immune system can be ‘kick-started’ again to fight infections and cancers.”
“This is very exciting because it means that B cells stay alive and well to do their job in the immune system fighting other infections. It also means the over-production of IL-10 is stopped, and the CLL cells are now exposed to immune cells specialized in fighting cancers.”
Dr Mackay said her team’s discovery may be relevant for cancers other than CLL and could change the way they are treated.
“The best weapon we have for fighting cancer is the immune system itself,” Dr Mackay noted. “It can sense the presence of an infection but also the emergence of a cancer.” ![]()

Photo courtesy of
Monash University
A study published in Leukemia has revealed a mechanism by which chronic lymphocytic leukemia (CLL) evades the immune system.
“It turns out that cancer cells are very good at sabotaging the immune system, using various tricks that confuse immune cells and ‘smoke screens’ preventing immune cells from recognizing the cancer,” said study author Fabienne Mackay, PhD, of Monash University in Melbourne, Victoria, Australia.
She and her colleagues believe they have determined exactly how CLL confuses the immune system and devised a way to stop it without destroying the patient’s immune system.
The team noted that B cells rely on the protein BAFF to survive. And each B cell has 3 different kinds of receptors that detect the presence of BAFF in the blood—TACI, BAFF-R, and BCMA.
The researchers discovered that, in CLL patients, the TACI receptors of cancerous B cells over-produce interleukin-10 (IL-10), which tricks the immune system into thinking nothing is wrong, allowing CLL to thrive undetected.
“We found that, when the receptor called TACI was blocked, it prevented the secretion of IL-10 without eliminating normal B cells,” Dr Mackay said. “Without IL-10, the tumor can no longer keep the immune system at bay, which means the patient’s immune system can be ‘kick-started’ again to fight infections and cancers.”
“This is very exciting because it means that B cells stay alive and well to do their job in the immune system fighting other infections. It also means the over-production of IL-10 is stopped, and the CLL cells are now exposed to immune cells specialized in fighting cancers.”
Dr Mackay said her team’s discovery may be relevant for cancers other than CLL and could change the way they are treated.
“The best weapon we have for fighting cancer is the immune system itself,” Dr Mackay noted. “It can sense the presence of an infection but also the emergence of a cancer.” ![]()

Photo courtesy of
Monash University
A study published in Leukemia has revealed a mechanism by which chronic lymphocytic leukemia (CLL) evades the immune system.
“It turns out that cancer cells are very good at sabotaging the immune system, using various tricks that confuse immune cells and ‘smoke screens’ preventing immune cells from recognizing the cancer,” said study author Fabienne Mackay, PhD, of Monash University in Melbourne, Victoria, Australia.
She and her colleagues believe they have determined exactly how CLL confuses the immune system and devised a way to stop it without destroying the patient’s immune system.
The team noted that B cells rely on the protein BAFF to survive. And each B cell has 3 different kinds of receptors that detect the presence of BAFF in the blood—TACI, BAFF-R, and BCMA.
The researchers discovered that, in CLL patients, the TACI receptors of cancerous B cells over-produce interleukin-10 (IL-10), which tricks the immune system into thinking nothing is wrong, allowing CLL to thrive undetected.
“We found that, when the receptor called TACI was blocked, it prevented the secretion of IL-10 without eliminating normal B cells,” Dr Mackay said. “Without IL-10, the tumor can no longer keep the immune system at bay, which means the patient’s immune system can be ‘kick-started’ again to fight infections and cancers.”
“This is very exciting because it means that B cells stay alive and well to do their job in the immune system fighting other infections. It also means the over-production of IL-10 is stopped, and the CLL cells are now exposed to immune cells specialized in fighting cancers.”
Dr Mackay said her team’s discovery may be relevant for cancers other than CLL and could change the way they are treated.
“The best weapon we have for fighting cancer is the immune system itself,” Dr Mackay noted. “It can sense the presence of an infection but also the emergence of a cancer.” ![]()
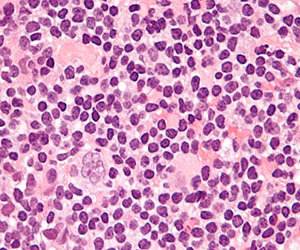
Mutations may contribute to CTCL

mycosis fungoides
Researchers have identified 15 mutations that may drive cutaneous T-cell lymphoma (CTCL).
The team sequenced normal and cancerous samples from 73 patients with mycosis fungoides or Sézary syndrome.
This revealed recurrent alterations in the TNFR2 pathway, as well as mutations in phosphoinositide 3-kinase (PI3K)-related genes, NF-κB pathway genes, and other genes that regulate T-cell survival and proliferation.
Specifically, the researchers identified TNFRSF1B point mutations, TNFRSF1B gains, CTLA4-CD28 fusions, a TRAF3 deletion, and mutations in NFAT5, TEC, PIK3CD, PIK3R6, PIK3CG, PIK3R5, PIK3R4, VAV1, MALT1, CD28, and ITK.
Paul Khavari, MD, PhD, of Stanford University in California, and his colleagues conducted this research and described their findings in a letter to Nature Genetics.
TNFR2 mutations
The researchers noted that the most frequent recurrent point mutation they identified occurred at codon 377 of TNFRSF1B (5%; 4/73), resulting in a recurrent TNFR2 Thr377Ile mutant.
TNFR2 is a receptor that regulates T-cell signaling pathways, and the mutation locked the receptor into an always-on state, preventing the T-cell-survival pathway from shutting down.
Previous studies showed that patients with increased TNFR2 in their bloodstream had more aggressive forms of CTCL that were more likely to return quickly after treatment.
This led Dr Khavari and his colleagues to look at the other patients’ DNA to see if duplications could account for both the elevated levels in the blood and increased signaling to activate the T-cell-survival pathway. The team found that 10 of the patients had TNFRSF1B gains.
In total, TNFRSF1B was altered in 18% of patients (13/73), by point mutation or gain (both in 1 patient). The researchers said this suggests a potential role of oncogenic TNFR2 signaling in the development of CTCL.
The team uncovered evidence to support this role by growing cells in the lab with either the point mutation or the gain. Their experiment showed the T-cell-survival pathway was more active in these cells than in normal cells.
Now, the researchers are working to incorporate the mutations they identified into the DNA of mice to study the mutated genes’ effects and the actions of drugs on those genes. ![]()

mycosis fungoides
Researchers have identified 15 mutations that may drive cutaneous T-cell lymphoma (CTCL).
The team sequenced normal and cancerous samples from 73 patients with mycosis fungoides or Sézary syndrome.
This revealed recurrent alterations in the TNFR2 pathway, as well as mutations in phosphoinositide 3-kinase (PI3K)-related genes, NF-κB pathway genes, and other genes that regulate T-cell survival and proliferation.
Specifically, the researchers identified TNFRSF1B point mutations, TNFRSF1B gains, CTLA4-CD28 fusions, a TRAF3 deletion, and mutations in NFAT5, TEC, PIK3CD, PIK3R6, PIK3CG, PIK3R5, PIK3R4, VAV1, MALT1, CD28, and ITK.
Paul Khavari, MD, PhD, of Stanford University in California, and his colleagues conducted this research and described their findings in a letter to Nature Genetics.
TNFR2 mutations
The researchers noted that the most frequent recurrent point mutation they identified occurred at codon 377 of TNFRSF1B (5%; 4/73), resulting in a recurrent TNFR2 Thr377Ile mutant.
TNFR2 is a receptor that regulates T-cell signaling pathways, and the mutation locked the receptor into an always-on state, preventing the T-cell-survival pathway from shutting down.
Previous studies showed that patients with increased TNFR2 in their bloodstream had more aggressive forms of CTCL that were more likely to return quickly after treatment.
This led Dr Khavari and his colleagues to look at the other patients’ DNA to see if duplications could account for both the elevated levels in the blood and increased signaling to activate the T-cell-survival pathway. The team found that 10 of the patients had TNFRSF1B gains.
In total, TNFRSF1B was altered in 18% of patients (13/73), by point mutation or gain (both in 1 patient). The researchers said this suggests a potential role of oncogenic TNFR2 signaling in the development of CTCL.
The team uncovered evidence to support this role by growing cells in the lab with either the point mutation or the gain. Their experiment showed the T-cell-survival pathway was more active in these cells than in normal cells.
Now, the researchers are working to incorporate the mutations they identified into the DNA of mice to study the mutated genes’ effects and the actions of drugs on those genes. ![]()

mycosis fungoides
Researchers have identified 15 mutations that may drive cutaneous T-cell lymphoma (CTCL).
The team sequenced normal and cancerous samples from 73 patients with mycosis fungoides or Sézary syndrome.
This revealed recurrent alterations in the TNFR2 pathway, as well as mutations in phosphoinositide 3-kinase (PI3K)-related genes, NF-κB pathway genes, and other genes that regulate T-cell survival and proliferation.
Specifically, the researchers identified TNFRSF1B point mutations, TNFRSF1B gains, CTLA4-CD28 fusions, a TRAF3 deletion, and mutations in NFAT5, TEC, PIK3CD, PIK3R6, PIK3CG, PIK3R5, PIK3R4, VAV1, MALT1, CD28, and ITK.
Paul Khavari, MD, PhD, of Stanford University in California, and his colleagues conducted this research and described their findings in a letter to Nature Genetics.
TNFR2 mutations
The researchers noted that the most frequent recurrent point mutation they identified occurred at codon 377 of TNFRSF1B (5%; 4/73), resulting in a recurrent TNFR2 Thr377Ile mutant.
TNFR2 is a receptor that regulates T-cell signaling pathways, and the mutation locked the receptor into an always-on state, preventing the T-cell-survival pathway from shutting down.
Previous studies showed that patients with increased TNFR2 in their bloodstream had more aggressive forms of CTCL that were more likely to return quickly after treatment.
This led Dr Khavari and his colleagues to look at the other patients’ DNA to see if duplications could account for both the elevated levels in the blood and increased signaling to activate the T-cell-survival pathway. The team found that 10 of the patients had TNFRSF1B gains.
In total, TNFRSF1B was altered in 18% of patients (13/73), by point mutation or gain (both in 1 patient). The researchers said this suggests a potential role of oncogenic TNFR2 signaling in the development of CTCL.
The team uncovered evidence to support this role by growing cells in the lab with either the point mutation or the gain. Their experiment showed the T-cell-survival pathway was more active in these cells than in normal cells.
Now, the researchers are working to incorporate the mutations they identified into the DNA of mice to study the mutated genes’ effects and the actions of drugs on those genes. ![]()
How religion affects well-being in cancer patients

Photo by Petr Kratochvil
Three meta-analyses shed new light on the role religion and spirituality play in cancer patients’ mental, social, and physical well-being.
The analyses, published in Cancer, indicate that religion and spirituality have significant associations with patients’ health.
But investigators observed wide variability among studies with regard to how different dimensions of religion and spirituality relate to different aspects of health.
In the first analysis, the investigators focused on physical health. Patients reporting greater overall religiousness and spirituality reported better physical health, greater ability to perform their usual daily tasks, and fewer physical symptoms of cancer and treatment.
“These relationships were particularly strong in patients who experienced greater emotional aspects of religion and spirituality, including a sense of meaning and purpose in life as well as a connection to a source larger than oneself,” said study author Heather Jim, PhD, of the Moffitt Cancer Center in Tampa, Florida.
Dr Jim noted that patients who reported greater cognitive aspects of religion and spirituality, such as the ability to integrate the cancer into their religious or spiritual beliefs, also reported better physical health. However, physical health was not related to behavioral aspects of religion and spiritualty, such as church attendance, prayer, or meditation.
In the second analysis, the investigators examined patients’ mental health. The team discovered that emotional aspects of religion and spirituality were more strongly associated with positive mental health than behavioral or cognitive aspects of religion and spirituality.
“Spiritual well-being was, unsurprisingly, associated with less anxiety, depression, or distress,” said study author John Salsman, PhD, of Wake Forest School of Medicine in Winston-Salem, North Carolina.
“Also, greater levels of spiritual distress and a sense of disconnectedness with God or a religious community was associated with greater psychological distress or poorer emotional well-being.”
The third analysis pertained to social health, or patients’ capacity to retain social roles and relationships in the face of illness. Religion and spirituality, as well as each of its dimensions, had modest but reliable links with social health.
“When we took a closer look, we found that patients with stronger spiritual well-being, more benign images of God (such as perceptions of a benevolent God rather than an angry or distant God), or stronger beliefs (such as convictions that a personal God can be called upon for assistance) reported better social health,” said study author Allen Sherman, PhD, of the University of Arkansas for Medical Sciences in Little Rock. “In contrast, those who struggled with their faith fared more poorly.”
The investigators believe future research should focus on how relationships between religious or spiritual involvement and health change over time and whether support services designed to enhance particular aspects of religion and spirituality in interested patients might help improve their well-being.
“In addition, some patients struggle with the religious or spiritual significance of their cancer, which is normal,” Dr Jim said. “How they resolve their struggle may impact their health, but more research is needed to better understand and support these patients.” ![]()

Photo by Petr Kratochvil
Three meta-analyses shed new light on the role religion and spirituality play in cancer patients’ mental, social, and physical well-being.
The analyses, published in Cancer, indicate that religion and spirituality have significant associations with patients’ health.
But investigators observed wide variability among studies with regard to how different dimensions of religion and spirituality relate to different aspects of health.
In the first analysis, the investigators focused on physical health. Patients reporting greater overall religiousness and spirituality reported better physical health, greater ability to perform their usual daily tasks, and fewer physical symptoms of cancer and treatment.
“These relationships were particularly strong in patients who experienced greater emotional aspects of religion and spirituality, including a sense of meaning and purpose in life as well as a connection to a source larger than oneself,” said study author Heather Jim, PhD, of the Moffitt Cancer Center in Tampa, Florida.
Dr Jim noted that patients who reported greater cognitive aspects of religion and spirituality, such as the ability to integrate the cancer into their religious or spiritual beliefs, also reported better physical health. However, physical health was not related to behavioral aspects of religion and spiritualty, such as church attendance, prayer, or meditation.
In the second analysis, the investigators examined patients’ mental health. The team discovered that emotional aspects of religion and spirituality were more strongly associated with positive mental health than behavioral or cognitive aspects of religion and spirituality.
“Spiritual well-being was, unsurprisingly, associated with less anxiety, depression, or distress,” said study author John Salsman, PhD, of Wake Forest School of Medicine in Winston-Salem, North Carolina.
“Also, greater levels of spiritual distress and a sense of disconnectedness with God or a religious community was associated with greater psychological distress or poorer emotional well-being.”
The third analysis pertained to social health, or patients’ capacity to retain social roles and relationships in the face of illness. Religion and spirituality, as well as each of its dimensions, had modest but reliable links with social health.
“When we took a closer look, we found that patients with stronger spiritual well-being, more benign images of God (such as perceptions of a benevolent God rather than an angry or distant God), or stronger beliefs (such as convictions that a personal God can be called upon for assistance) reported better social health,” said study author Allen Sherman, PhD, of the University of Arkansas for Medical Sciences in Little Rock. “In contrast, those who struggled with their faith fared more poorly.”
The investigators believe future research should focus on how relationships between religious or spiritual involvement and health change over time and whether support services designed to enhance particular aspects of religion and spirituality in interested patients might help improve their well-being.
“In addition, some patients struggle with the religious or spiritual significance of their cancer, which is normal,” Dr Jim said. “How they resolve their struggle may impact their health, but more research is needed to better understand and support these patients.” ![]()

Photo by Petr Kratochvil
Three meta-analyses shed new light on the role religion and spirituality play in cancer patients’ mental, social, and physical well-being.
The analyses, published in Cancer, indicate that religion and spirituality have significant associations with patients’ health.
But investigators observed wide variability among studies with regard to how different dimensions of religion and spirituality relate to different aspects of health.
In the first analysis, the investigators focused on physical health. Patients reporting greater overall religiousness and spirituality reported better physical health, greater ability to perform their usual daily tasks, and fewer physical symptoms of cancer and treatment.
“These relationships were particularly strong in patients who experienced greater emotional aspects of religion and spirituality, including a sense of meaning and purpose in life as well as a connection to a source larger than oneself,” said study author Heather Jim, PhD, of the Moffitt Cancer Center in Tampa, Florida.
Dr Jim noted that patients who reported greater cognitive aspects of religion and spirituality, such as the ability to integrate the cancer into their religious or spiritual beliefs, also reported better physical health. However, physical health was not related to behavioral aspects of religion and spiritualty, such as church attendance, prayer, or meditation.
In the second analysis, the investigators examined patients’ mental health. The team discovered that emotional aspects of religion and spirituality were more strongly associated with positive mental health than behavioral or cognitive aspects of religion and spirituality.
“Spiritual well-being was, unsurprisingly, associated with less anxiety, depression, or distress,” said study author John Salsman, PhD, of Wake Forest School of Medicine in Winston-Salem, North Carolina.
“Also, greater levels of spiritual distress and a sense of disconnectedness with God or a religious community was associated with greater psychological distress or poorer emotional well-being.”
The third analysis pertained to social health, or patients’ capacity to retain social roles and relationships in the face of illness. Religion and spirituality, as well as each of its dimensions, had modest but reliable links with social health.
“When we took a closer look, we found that patients with stronger spiritual well-being, more benign images of God (such as perceptions of a benevolent God rather than an angry or distant God), or stronger beliefs (such as convictions that a personal God can be called upon for assistance) reported better social health,” said study author Allen Sherman, PhD, of the University of Arkansas for Medical Sciences in Little Rock. “In contrast, those who struggled with their faith fared more poorly.”
The investigators believe future research should focus on how relationships between religious or spiritual involvement and health change over time and whether support services designed to enhance particular aspects of religion and spirituality in interested patients might help improve their well-being.
“In addition, some patients struggle with the religious or spiritual significance of their cancer, which is normal,” Dr Jim said. “How they resolve their struggle may impact their health, but more research is needed to better understand and support these patients.” ![]()
Lymph2Cx assay identifies outcomes in DLBCL
In patients with diffuse large B-cell lymphoma (DLBCL) treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), the Lymph2Cx assay was able to separate the cohort into groups with significantly different outcomes based on cell of origin (COO), investigators reported online in the Journal of Clinical Oncology.
In pairwise multivariable analyses, COO was associated with outcomes that were independent of International Prognostic Index score (IPI) and MYC/BCL2 immunohistochemistry (IHC).
Gene expression profiling of DLBCL has provided classification into two distinct subtypes: germinal center B-cell–like (GCB) and activated B-cell–like (ABC) subtypes. Using the cell of origin classification can define subgroups with distinct biology ad pathogenesis, as well as identify patient groups with different outcomes after treatment. Improvements in technology have allowed for the use of formalin-fixed paraffin-embedded tissue (FFPET) biopsies for more reliable gene expression profiling.
“The size of the study cohort allowed exploration of the prognostic value of COO in comparison with other prognostic tools,” wrote Dr. David W. Scott of the British Columbia Cancer Agency, Vancouver, and his colleagues (J Clin Oncol. 2015 Aug 3. doi: 10.1200/JCO.2014.60.2383).“Although the IPI remains the most powerful tool for risk stratification, COO assignment provides additional prognostic information, particularly evident in the intermediate IPI score group,” the authors noted.
The consistency and reproducibility of COO assignment using the Lymph2Cx assay was evaluated in a large patient cohort treated with R-CHOP therapy, and the relationship between COO, MYC/BCL2 dual expression, and IPI score with respect to defining prognosis in patients with DLBCL was also investigated.
Reproducibility of COO assignment using the Lymph2Cx assay was tested using repeated sampling within tumor biopsies and changes in reagent lots, and concordance of COO calls across the two reagent lots was 100%.
The COO was then determined in 344 patients with de novo DLBCL who were treated with R-CHOP at a single center. MYC and BCL2 protein expression was assessed using immunohistochemistry on tissue microarrays, and the median follow-up of living patients was 6.5 years (range, 0.75-13.2 years).
COO was a prognostic biomarker in the patient cohort. Those with activated B-cell–like DLBCL had significantly inferior outcomes as compared to patients with germinal center B-cell–like DLBCL (log-rank P less than .001 for time to progression, progression-free survival, disease-specific survival, and overall survival).
When reviewing the relationship between COO, IPI score, and MYC/BCL2 IHC, pairwise multivariable analyses demonstrated that the prognostic impact of COO is independent of IPI score. The prognostic value added by COO was most notable when patients with intermediate IPI scores were evaluated (ABC vs. GCB: 5-year time to progression, 53% v 74%; log-rank P = .003).
Both COO and MYC/BCL2 IHC identified high-risk groups that were similar in size (32% vs. 31%) with comparable outcomes (5-year time to progression, 51% vs. 51%). In pairwise multivariable analyses that included COO and MYC/BCL2 IHC as variables, COO remained significant, thus showing that COO had prognostic power “beyond that conferred merely by enrichment of MYC-positive/BCL2-positive patient cases of the ABC subtype,” wrote the authors.
When COO, IPI, and MYC/BCL2 IHC were all included in multivariable analyses, COO remained significantly associated with time to progression and progression-free survival.
“We anticipate that over the next few years, with the emergence of agents with selective activity in ABC or GCB DLBCL, the determination of COO will become part of the foundation for optimal patient care,” the authors concluded.
In patients with diffuse large B-cell lymphoma (DLBCL) treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), the Lymph2Cx assay was able to separate the cohort into groups with significantly different outcomes based on cell of origin (COO), investigators reported online in the Journal of Clinical Oncology.
In pairwise multivariable analyses, COO was associated with outcomes that were independent of International Prognostic Index score (IPI) and MYC/BCL2 immunohistochemistry (IHC).
Gene expression profiling of DLBCL has provided classification into two distinct subtypes: germinal center B-cell–like (GCB) and activated B-cell–like (ABC) subtypes. Using the cell of origin classification can define subgroups with distinct biology ad pathogenesis, as well as identify patient groups with different outcomes after treatment. Improvements in technology have allowed for the use of formalin-fixed paraffin-embedded tissue (FFPET) biopsies for more reliable gene expression profiling.
“The size of the study cohort allowed exploration of the prognostic value of COO in comparison with other prognostic tools,” wrote Dr. David W. Scott of the British Columbia Cancer Agency, Vancouver, and his colleagues (J Clin Oncol. 2015 Aug 3. doi: 10.1200/JCO.2014.60.2383).“Although the IPI remains the most powerful tool for risk stratification, COO assignment provides additional prognostic information, particularly evident in the intermediate IPI score group,” the authors noted.
The consistency and reproducibility of COO assignment using the Lymph2Cx assay was evaluated in a large patient cohort treated with R-CHOP therapy, and the relationship between COO, MYC/BCL2 dual expression, and IPI score with respect to defining prognosis in patients with DLBCL was also investigated.
Reproducibility of COO assignment using the Lymph2Cx assay was tested using repeated sampling within tumor biopsies and changes in reagent lots, and concordance of COO calls across the two reagent lots was 100%.
The COO was then determined in 344 patients with de novo DLBCL who were treated with R-CHOP at a single center. MYC and BCL2 protein expression was assessed using immunohistochemistry on tissue microarrays, and the median follow-up of living patients was 6.5 years (range, 0.75-13.2 years).
COO was a prognostic biomarker in the patient cohort. Those with activated B-cell–like DLBCL had significantly inferior outcomes as compared to patients with germinal center B-cell–like DLBCL (log-rank P less than .001 for time to progression, progression-free survival, disease-specific survival, and overall survival).
When reviewing the relationship between COO, IPI score, and MYC/BCL2 IHC, pairwise multivariable analyses demonstrated that the prognostic impact of COO is independent of IPI score. The prognostic value added by COO was most notable when patients with intermediate IPI scores were evaluated (ABC vs. GCB: 5-year time to progression, 53% v 74%; log-rank P = .003).
Both COO and MYC/BCL2 IHC identified high-risk groups that were similar in size (32% vs. 31%) with comparable outcomes (5-year time to progression, 51% vs. 51%). In pairwise multivariable analyses that included COO and MYC/BCL2 IHC as variables, COO remained significant, thus showing that COO had prognostic power “beyond that conferred merely by enrichment of MYC-positive/BCL2-positive patient cases of the ABC subtype,” wrote the authors.
When COO, IPI, and MYC/BCL2 IHC were all included in multivariable analyses, COO remained significantly associated with time to progression and progression-free survival.
“We anticipate that over the next few years, with the emergence of agents with selective activity in ABC or GCB DLBCL, the determination of COO will become part of the foundation for optimal patient care,” the authors concluded.
In patients with diffuse large B-cell lymphoma (DLBCL) treated with rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), the Lymph2Cx assay was able to separate the cohort into groups with significantly different outcomes based on cell of origin (COO), investigators reported online in the Journal of Clinical Oncology.
In pairwise multivariable analyses, COO was associated with outcomes that were independent of International Prognostic Index score (IPI) and MYC/BCL2 immunohistochemistry (IHC).
Gene expression profiling of DLBCL has provided classification into two distinct subtypes: germinal center B-cell–like (GCB) and activated B-cell–like (ABC) subtypes. Using the cell of origin classification can define subgroups with distinct biology ad pathogenesis, as well as identify patient groups with different outcomes after treatment. Improvements in technology have allowed for the use of formalin-fixed paraffin-embedded tissue (FFPET) biopsies for more reliable gene expression profiling.
“The size of the study cohort allowed exploration of the prognostic value of COO in comparison with other prognostic tools,” wrote Dr. David W. Scott of the British Columbia Cancer Agency, Vancouver, and his colleagues (J Clin Oncol. 2015 Aug 3. doi: 10.1200/JCO.2014.60.2383).“Although the IPI remains the most powerful tool for risk stratification, COO assignment provides additional prognostic information, particularly evident in the intermediate IPI score group,” the authors noted.
The consistency and reproducibility of COO assignment using the Lymph2Cx assay was evaluated in a large patient cohort treated with R-CHOP therapy, and the relationship between COO, MYC/BCL2 dual expression, and IPI score with respect to defining prognosis in patients with DLBCL was also investigated.
Reproducibility of COO assignment using the Lymph2Cx assay was tested using repeated sampling within tumor biopsies and changes in reagent lots, and concordance of COO calls across the two reagent lots was 100%.
The COO was then determined in 344 patients with de novo DLBCL who were treated with R-CHOP at a single center. MYC and BCL2 protein expression was assessed using immunohistochemistry on tissue microarrays, and the median follow-up of living patients was 6.5 years (range, 0.75-13.2 years).
COO was a prognostic biomarker in the patient cohort. Those with activated B-cell–like DLBCL had significantly inferior outcomes as compared to patients with germinal center B-cell–like DLBCL (log-rank P less than .001 for time to progression, progression-free survival, disease-specific survival, and overall survival).
When reviewing the relationship between COO, IPI score, and MYC/BCL2 IHC, pairwise multivariable analyses demonstrated that the prognostic impact of COO is independent of IPI score. The prognostic value added by COO was most notable when patients with intermediate IPI scores were evaluated (ABC vs. GCB: 5-year time to progression, 53% v 74%; log-rank P = .003).
Both COO and MYC/BCL2 IHC identified high-risk groups that were similar in size (32% vs. 31%) with comparable outcomes (5-year time to progression, 51% vs. 51%). In pairwise multivariable analyses that included COO and MYC/BCL2 IHC as variables, COO remained significant, thus showing that COO had prognostic power “beyond that conferred merely by enrichment of MYC-positive/BCL2-positive patient cases of the ABC subtype,” wrote the authors.
When COO, IPI, and MYC/BCL2 IHC were all included in multivariable analyses, COO remained significantly associated with time to progression and progression-free survival.
“We anticipate that over the next few years, with the emergence of agents with selective activity in ABC or GCB DLBCL, the determination of COO will become part of the foundation for optimal patient care,” the authors concluded.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: The Lymph2Cx is an accurate assay for DLBCL cell of origin assignment, and identified groups with significantly different outcomes after treatment with R-CHOP.
Major finding: The Lymph2Cx assay provided concordant COO calls in 100% of 83 formalin-fixed paraffin-embedded tissue biopsies, and COO was associated with significantly different outcomes independent of IPI score and MYC/BCL2 immunohistochemistry
Data source: The Lymph2Cx assay was compared with the IPI) score and MYC/BCL2 coexpression status in FFPET biopsies from 344 patients with DLBCL uniformly treated with R-CHOP.
Disclosures: The study was supported by the Terry Fox Foundation and by the British Columbia Cancer Foundation. Dr. Scott is potentially named inventor on pending patent on use of gene expression profiling to assign COO in DLBCL. Dr. Scott and several coauthors also report financial relationships with industry.
Topical gel appears safe, effective in CTCL
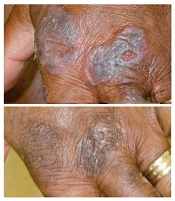
treatment (top) and 16 weeks
after treatment began
Photo from Penn Medicine
Results of a phase 1 trial suggest a topical gel can prompt regression of both treated and untreated lesions in patients with
early stage cutaneous T-cell lymphoma (CTCL).
Of the 12 patients who received the treatment, resiquimod gel, 75% had a significant improvement in treated lesions.
And 92% of patients had a more than 50% improvement in body surface area involvement, which included untreated lesions. Two patients experienced complete disease clearance.
Adverse events associated with resiquimod were largely limited to the skin, although 2 patients had transient, low-grade fever. Five patients developed superficial skin erosions that healed when treatment was stopped and did not reappear after treatment began again.
Alain Rook, MD, of the University of Pennsylvania in Philadelphia, and his colleagues reported these results in Blood.
“The results of the trial suggest that resiquimod is safely and effectively absorbed into the skin and, beyond diminishing treated lesions, also enhances the immune response, leading to healing of even untreated lesions,” Dr Rook said.
“To our knowledge, this is the first topical therapy that can clear untreated lesions and lead to complete remission in some patients.”
Treatment and response
Dr Rook and his colleagues tested resiquimod gel in 12 patients who had previously undergone an average of 6 treatments for early stage CTCL.
The patients applied specified doses of resiquimod (0.03% or 0.06%) to select skin lesions for 16 weeks. Some patients using the 0.06% dose had complete clearance of all malignant cells after 8 weeks.
By the final evaluation, treated lesions had significantly improved in 75% of patients, and 30% of patients had complete clearance of all treated lesions.
Resiquimod also improved untreated lesions, resulting in more than 50% improvement in body surface area involvement for 92% of patients.
Two participants, one of whom had been living with CTCL for more than 15 years without responding to treatment, experienced complete eradication of the disease.
Malignant cell analysis
The researchers used high-throughput sequencing to detect malignant cells in patient samples. The technique could identify a single malignant cell among 100,000 healthy cells.
The team analyzed DNA from biopsies of the same lesion before treatment and 8 weeks after treatment began.
They observed a significant reduction of malignant T cells in 9 of 10 patients tested, 3 of whom had complete eradication of the malignant population and 1 of whom had a 99.6% reduction.
Adverse events
Adverse events associated with resiquimod were all grade 1 and were primarily related to local skin irritation. There were no serious adverse events.
Five of 8 patients receiving resiquimod at the 0.06% dose developed superficial skin erosions at some sites of treatment. These erosions healed completely within a week of stopping treatment and did not recur with re-initiation of treatment.
Two patients receiving the 0.06% dose experienced 2 days of low-grade fever (less than 100.50 F) when treatment began.
“Overall, lesions responded far better to topical resiquimod than they have with other topical therapies, including some potent topical steroids and topical chemotherapy, and [resiquimod] was extremely well tolerated by patients,” Dr Rook said.
“Building upon previous research, our study suggests resiquimod might be useful in combination with other therapies in the treatment of more advanced CTCL. Further research with larger participant populations is needed to determine the best approach and application for these patients.” ![]()

treatment (top) and 16 weeks
after treatment began
Photo from Penn Medicine
Results of a phase 1 trial suggest a topical gel can prompt regression of both treated and untreated lesions in patients with
early stage cutaneous T-cell lymphoma (CTCL).
Of the 12 patients who received the treatment, resiquimod gel, 75% had a significant improvement in treated lesions.
And 92% of patients had a more than 50% improvement in body surface area involvement, which included untreated lesions. Two patients experienced complete disease clearance.
Adverse events associated with resiquimod were largely limited to the skin, although 2 patients had transient, low-grade fever. Five patients developed superficial skin erosions that healed when treatment was stopped and did not reappear after treatment began again.
Alain Rook, MD, of the University of Pennsylvania in Philadelphia, and his colleagues reported these results in Blood.
“The results of the trial suggest that resiquimod is safely and effectively absorbed into the skin and, beyond diminishing treated lesions, also enhances the immune response, leading to healing of even untreated lesions,” Dr Rook said.
“To our knowledge, this is the first topical therapy that can clear untreated lesions and lead to complete remission in some patients.”
Treatment and response
Dr Rook and his colleagues tested resiquimod gel in 12 patients who had previously undergone an average of 6 treatments for early stage CTCL.
The patients applied specified doses of resiquimod (0.03% or 0.06%) to select skin lesions for 16 weeks. Some patients using the 0.06% dose had complete clearance of all malignant cells after 8 weeks.
By the final evaluation, treated lesions had significantly improved in 75% of patients, and 30% of patients had complete clearance of all treated lesions.
Resiquimod also improved untreated lesions, resulting in more than 50% improvement in body surface area involvement for 92% of patients.
Two participants, one of whom had been living with CTCL for more than 15 years without responding to treatment, experienced complete eradication of the disease.
Malignant cell analysis
The researchers used high-throughput sequencing to detect malignant cells in patient samples. The technique could identify a single malignant cell among 100,000 healthy cells.
The team analyzed DNA from biopsies of the same lesion before treatment and 8 weeks after treatment began.
They observed a significant reduction of malignant T cells in 9 of 10 patients tested, 3 of whom had complete eradication of the malignant population and 1 of whom had a 99.6% reduction.
Adverse events
Adverse events associated with resiquimod were all grade 1 and were primarily related to local skin irritation. There were no serious adverse events.
Five of 8 patients receiving resiquimod at the 0.06% dose developed superficial skin erosions at some sites of treatment. These erosions healed completely within a week of stopping treatment and did not recur with re-initiation of treatment.
Two patients receiving the 0.06% dose experienced 2 days of low-grade fever (less than 100.50 F) when treatment began.
“Overall, lesions responded far better to topical resiquimod than they have with other topical therapies, including some potent topical steroids and topical chemotherapy, and [resiquimod] was extremely well tolerated by patients,” Dr Rook said.
“Building upon previous research, our study suggests resiquimod might be useful in combination with other therapies in the treatment of more advanced CTCL. Further research with larger participant populations is needed to determine the best approach and application for these patients.” ![]()

treatment (top) and 16 weeks
after treatment began
Photo from Penn Medicine
Results of a phase 1 trial suggest a topical gel can prompt regression of both treated and untreated lesions in patients with
early stage cutaneous T-cell lymphoma (CTCL).
Of the 12 patients who received the treatment, resiquimod gel, 75% had a significant improvement in treated lesions.
And 92% of patients had a more than 50% improvement in body surface area involvement, which included untreated lesions. Two patients experienced complete disease clearance.
Adverse events associated with resiquimod were largely limited to the skin, although 2 patients had transient, low-grade fever. Five patients developed superficial skin erosions that healed when treatment was stopped and did not reappear after treatment began again.
Alain Rook, MD, of the University of Pennsylvania in Philadelphia, and his colleagues reported these results in Blood.
“The results of the trial suggest that resiquimod is safely and effectively absorbed into the skin and, beyond diminishing treated lesions, also enhances the immune response, leading to healing of even untreated lesions,” Dr Rook said.
“To our knowledge, this is the first topical therapy that can clear untreated lesions and lead to complete remission in some patients.”
Treatment and response
Dr Rook and his colleagues tested resiquimod gel in 12 patients who had previously undergone an average of 6 treatments for early stage CTCL.
The patients applied specified doses of resiquimod (0.03% or 0.06%) to select skin lesions for 16 weeks. Some patients using the 0.06% dose had complete clearance of all malignant cells after 8 weeks.
By the final evaluation, treated lesions had significantly improved in 75% of patients, and 30% of patients had complete clearance of all treated lesions.
Resiquimod also improved untreated lesions, resulting in more than 50% improvement in body surface area involvement for 92% of patients.
Two participants, one of whom had been living with CTCL for more than 15 years without responding to treatment, experienced complete eradication of the disease.
Malignant cell analysis
The researchers used high-throughput sequencing to detect malignant cells in patient samples. The technique could identify a single malignant cell among 100,000 healthy cells.
The team analyzed DNA from biopsies of the same lesion before treatment and 8 weeks after treatment began.
They observed a significant reduction of malignant T cells in 9 of 10 patients tested, 3 of whom had complete eradication of the malignant population and 1 of whom had a 99.6% reduction.
Adverse events
Adverse events associated with resiquimod were all grade 1 and were primarily related to local skin irritation. There were no serious adverse events.
Five of 8 patients receiving resiquimod at the 0.06% dose developed superficial skin erosions at some sites of treatment. These erosions healed completely within a week of stopping treatment and did not recur with re-initiation of treatment.
Two patients receiving the 0.06% dose experienced 2 days of low-grade fever (less than 100.50 F) when treatment began.
“Overall, lesions responded far better to topical resiquimod than they have with other topical therapies, including some potent topical steroids and topical chemotherapy, and [resiquimod] was extremely well tolerated by patients,” Dr Rook said.
“Building upon previous research, our study suggests resiquimod might be useful in combination with other therapies in the treatment of more advanced CTCL. Further research with larger participant populations is needed to determine the best approach and application for these patients.”
Treatment of choice for NLPHL? Involved-field RT alone
Involved-field radiotherapy alone should be the treatment of choice for stage 1A nodular lymphocyte-predominant Hodgkin lymphoma because it yields similar survival outcomes but fewer toxic effects than other therapies, investigators reported online Aug. 3 in Journal of Clinical Oncology.
Few studies have examined the long-term outcomes of patients who have this generally indolent malignancy, even though late treatment-associated effects are their major cause of death, said Dr. Dennis A. Eichenauer and his associates at University Hospital Cologne and University Hospital Münster, both in Germany.
To assess long-term outcomes in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL), the investigators analyzed data for 256 patients who participated in seven prospective randomized clinical trials comparing various treatments during a 21-year period, who were followed for a median of 91 months. The study participants’ median age at diagnosis was 39 years (range, 16-75 years), and all achieved remission with therapy. They were categorized by treatment type: 108 received involved-field radiotherapy (IF-RT) alone; 49 received extended-field radiotherapy (EF-RT) alone; 72 received combined modality treatment involving various combinations of doxorubicin, bleomycin, vinblastine, dacarbazine, IF-RT, EF-RT, and/or rituximab; and 27 received rituximab alone.
At 8 years, progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, which are nonsignificant differences. Overall survival was 99.0%, 95.7%, and 98.6%, respectively, which also are nonsignificant differences. Rituximab alone was markedly less effective than the other treatments, with a 4-year progression-free survival of only 81.0%. “Compared with patients who received IF-RT, patients treated with rituximab had a hazard ratio of 4.99 for relapse,” Dr. Eichenauer and his associates wrote (J Clin Oncol. 2015 Aug 3 [doi: 10.1200/JCO. 2014.60.4363]).
Only 3.7% of patients treated with IF-RT developed a second malignancy during follow-up, compared with 11.1% of those treated with combined modalities, 6.1% of those treated with EF-RT, and 7.4% of those treated with rituximab. The rate of relapse was markedly higher with rituximab than with the other treatments.
The main study findings are twofold. First, IF-RT was at least as effective as other treatments in controlling NLPHL, was less toxic acutely, and carried similar or reduced risks of late adverse effects such as relapse and second malignancies. It should be considered the first-line treatment of choice. Second, rituximab alone should not be used routinely in these patients because it yields poorer survival outcomes and a higher relapse rate. “However, it might represent an option for individual patients, such as young women with abdominal disease, to avoid gonadotoxic effects of radiotherapy,” the investigators said.
Involved-field radiotherapy alone should be the treatment of choice for stage 1A nodular lymphocyte-predominant Hodgkin lymphoma because it yields similar survival outcomes but fewer toxic effects than other therapies, investigators reported online Aug. 3 in Journal of Clinical Oncology.
Few studies have examined the long-term outcomes of patients who have this generally indolent malignancy, even though late treatment-associated effects are their major cause of death, said Dr. Dennis A. Eichenauer and his associates at University Hospital Cologne and University Hospital Münster, both in Germany.
To assess long-term outcomes in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL), the investigators analyzed data for 256 patients who participated in seven prospective randomized clinical trials comparing various treatments during a 21-year period, who were followed for a median of 91 months. The study participants’ median age at diagnosis was 39 years (range, 16-75 years), and all achieved remission with therapy. They were categorized by treatment type: 108 received involved-field radiotherapy (IF-RT) alone; 49 received extended-field radiotherapy (EF-RT) alone; 72 received combined modality treatment involving various combinations of doxorubicin, bleomycin, vinblastine, dacarbazine, IF-RT, EF-RT, and/or rituximab; and 27 received rituximab alone.
At 8 years, progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, which are nonsignificant differences. Overall survival was 99.0%, 95.7%, and 98.6%, respectively, which also are nonsignificant differences. Rituximab alone was markedly less effective than the other treatments, with a 4-year progression-free survival of only 81.0%. “Compared with patients who received IF-RT, patients treated with rituximab had a hazard ratio of 4.99 for relapse,” Dr. Eichenauer and his associates wrote (J Clin Oncol. 2015 Aug 3 [doi: 10.1200/JCO. 2014.60.4363]).
Only 3.7% of patients treated with IF-RT developed a second malignancy during follow-up, compared with 11.1% of those treated with combined modalities, 6.1% of those treated with EF-RT, and 7.4% of those treated with rituximab. The rate of relapse was markedly higher with rituximab than with the other treatments.
The main study findings are twofold. First, IF-RT was at least as effective as other treatments in controlling NLPHL, was less toxic acutely, and carried similar or reduced risks of late adverse effects such as relapse and second malignancies. It should be considered the first-line treatment of choice. Second, rituximab alone should not be used routinely in these patients because it yields poorer survival outcomes and a higher relapse rate. “However, it might represent an option for individual patients, such as young women with abdominal disease, to avoid gonadotoxic effects of radiotherapy,” the investigators said.
Involved-field radiotherapy alone should be the treatment of choice for stage 1A nodular lymphocyte-predominant Hodgkin lymphoma because it yields similar survival outcomes but fewer toxic effects than other therapies, investigators reported online Aug. 3 in Journal of Clinical Oncology.
Few studies have examined the long-term outcomes of patients who have this generally indolent malignancy, even though late treatment-associated effects are their major cause of death, said Dr. Dennis A. Eichenauer and his associates at University Hospital Cologne and University Hospital Münster, both in Germany.
To assess long-term outcomes in nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL), the investigators analyzed data for 256 patients who participated in seven prospective randomized clinical trials comparing various treatments during a 21-year period, who were followed for a median of 91 months. The study participants’ median age at diagnosis was 39 years (range, 16-75 years), and all achieved remission with therapy. They were categorized by treatment type: 108 received involved-field radiotherapy (IF-RT) alone; 49 received extended-field radiotherapy (EF-RT) alone; 72 received combined modality treatment involving various combinations of doxorubicin, bleomycin, vinblastine, dacarbazine, IF-RT, EF-RT, and/or rituximab; and 27 received rituximab alone.
At 8 years, progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, which are nonsignificant differences. Overall survival was 99.0%, 95.7%, and 98.6%, respectively, which also are nonsignificant differences. Rituximab alone was markedly less effective than the other treatments, with a 4-year progression-free survival of only 81.0%. “Compared with patients who received IF-RT, patients treated with rituximab had a hazard ratio of 4.99 for relapse,” Dr. Eichenauer and his associates wrote (J Clin Oncol. 2015 Aug 3 [doi: 10.1200/JCO. 2014.60.4363]).
Only 3.7% of patients treated with IF-RT developed a second malignancy during follow-up, compared with 11.1% of those treated with combined modalities, 6.1% of those treated with EF-RT, and 7.4% of those treated with rituximab. The rate of relapse was markedly higher with rituximab than with the other treatments.
The main study findings are twofold. First, IF-RT was at least as effective as other treatments in controlling NLPHL, was less toxic acutely, and carried similar or reduced risks of late adverse effects such as relapse and second malignancies. It should be considered the first-line treatment of choice. Second, rituximab alone should not be used routinely in these patients because it yields poorer survival outcomes and a higher relapse rate. “However, it might represent an option for individual patients, such as young women with abdominal disease, to avoid gonadotoxic effects of radiotherapy,” the investigators said.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Involved-field radiotherapy alone yields similar survival but fewer toxic effects than do other treatments for stage 1A nodular lymphocyte–predominant Hodgkin lymphoma.
Major finding: Progression-free survival was 91.9% with IF-RT, 84.3% with EF-RT, and 88.5% with combined modalities, and overall survival was 99.0%, 95.7%, and 98.6%, respectively.
Data source: A retrospective analysis of outcomes of seven German prospective clinical trials involving 256 patients followed for a median of 91 months.
Disclosures: This study was supported by University Hospital Cologne and University Hospital Münster. Dr. Eichenauer reported having no relevant financial disclosures; his associates reported ties to Amgen, Takeda, Novartis, Gilead, and Takeda/Millennium.
Drug gets orphan designation for CTCL

The European Commission has granted orphan drug designation to synthetic hypericin, the active pharmaceutical ingredient in SGX301, for the treatment of cutaneous T-cell lymphoma (CTCL).
SGX301 is a first-in-class, photodynamic therapy utilizing safe, visible light for activation. Synthetic hypericin is a potent photosensitizer that is topically applied to skin lesions and activated by visible fluorescent light 16 to 24 hours later.
This treatment approach is intended to prevent the secondary malignancies that may occur following chemotherapy or photodynamic therapies that are dependent on ultraviolet exposure.
Combined with photoactivation, hypericin has demonstrated significant antiproliferative effects on activated, normal human lymphoid cells and inhibited the growth of malignant T cells isolated from CTCL patients.
Topical hypericin has also proven safe in a phase 1 study of healthy volunteers.
In a phase 2 trial of patients with CTCL (mycosis fungoides only) or psoriasis, topical hypericin conferred a significant improvement over placebo. Among CTCL patients, the treatment prompted a response rate of 58.3%, compared to an 8.3% response rate for placebo (P≤0.04).
Topical hypericin was also well tolerated in this trial. There were no deaths or serious adverse events related to the treatment. However, there were reports of mild to moderate burning, itching, erythema, and pruritus at the application site.
Soligenix, Inc., the company developing SGX301, is currently working with CTCL centers, the National Organization for Rare Disorders, and the Cutaneous Lymphoma Foundation to begin a 120-subject phase 3 trial of SGX301.
About orphan designation
The European Commission grants orphan designation to medicines designed to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons in the European Union and has no satisfactory treatment available.
In addition to a 10-year period of marketing exclusivity after product approval, orphan drug designation provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase, as well as direct access to the centralized authorization procedure.
SGX301 has both orphan designation and fast track designation from the US Food and Drug Administration for the first-line treatment of CTCL.

The European Commission has granted orphan drug designation to synthetic hypericin, the active pharmaceutical ingredient in SGX301, for the treatment of cutaneous T-cell lymphoma (CTCL).
SGX301 is a first-in-class, photodynamic therapy utilizing safe, visible light for activation. Synthetic hypericin is a potent photosensitizer that is topically applied to skin lesions and activated by visible fluorescent light 16 to 24 hours later.
This treatment approach is intended to prevent the secondary malignancies that may occur following chemotherapy or photodynamic therapies that are dependent on ultraviolet exposure.
Combined with photoactivation, hypericin has demonstrated significant antiproliferative effects on activated, normal human lymphoid cells and inhibited the growth of malignant T cells isolated from CTCL patients.
Topical hypericin has also proven safe in a phase 1 study of healthy volunteers.
In a phase 2 trial of patients with CTCL (mycosis fungoides only) or psoriasis, topical hypericin conferred a significant improvement over placebo. Among CTCL patients, the treatment prompted a response rate of 58.3%, compared to an 8.3% response rate for placebo (P≤0.04).
Topical hypericin was also well tolerated in this trial. There were no deaths or serious adverse events related to the treatment. However, there were reports of mild to moderate burning, itching, erythema, and pruritus at the application site.
Soligenix, Inc., the company developing SGX301, is currently working with CTCL centers, the National Organization for Rare Disorders, and the Cutaneous Lymphoma Foundation to begin a 120-subject phase 3 trial of SGX301.
About orphan designation
The European Commission grants orphan designation to medicines designed to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons in the European Union and has no satisfactory treatment available.
In addition to a 10-year period of marketing exclusivity after product approval, orphan drug designation provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase, as well as direct access to the centralized authorization procedure.
SGX301 has both orphan designation and fast track designation from the US Food and Drug Administration for the first-line treatment of CTCL.

The European Commission has granted orphan drug designation to synthetic hypericin, the active pharmaceutical ingredient in SGX301, for the treatment of cutaneous T-cell lymphoma (CTCL).
SGX301 is a first-in-class, photodynamic therapy utilizing safe, visible light for activation. Synthetic hypericin is a potent photosensitizer that is topically applied to skin lesions and activated by visible fluorescent light 16 to 24 hours later.
This treatment approach is intended to prevent the secondary malignancies that may occur following chemotherapy or photodynamic therapies that are dependent on ultraviolet exposure.
Combined with photoactivation, hypericin has demonstrated significant antiproliferative effects on activated, normal human lymphoid cells and inhibited the growth of malignant T cells isolated from CTCL patients.
Topical hypericin has also proven safe in a phase 1 study of healthy volunteers.
In a phase 2 trial of patients with CTCL (mycosis fungoides only) or psoriasis, topical hypericin conferred a significant improvement over placebo. Among CTCL patients, the treatment prompted a response rate of 58.3%, compared to an 8.3% response rate for placebo (P≤0.04).
Topical hypericin was also well tolerated in this trial. There were no deaths or serious adverse events related to the treatment. However, there were reports of mild to moderate burning, itching, erythema, and pruritus at the application site.
Soligenix, Inc., the company developing SGX301, is currently working with CTCL centers, the National Organization for Rare Disorders, and the Cutaneous Lymphoma Foundation to begin a 120-subject phase 3 trial of SGX301.
About orphan designation
The European Commission grants orphan designation to medicines designed to treat a life-threatening or chronically debilitating condition that affects no more than 5 in 10,000 persons in the European Union and has no satisfactory treatment available.
In addition to a 10-year period of marketing exclusivity after product approval, orphan drug designation provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase, as well as direct access to the centralized authorization procedure.
SGX301 has both orphan designation and fast track designation from the US Food and Drug Administration for the first-line treatment of CTCL.