User login
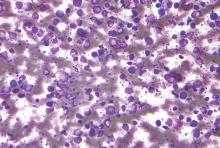
Study links radon and hematologic cancers in women

New research suggests there is a significant positive association between high levels of residential radon and the risk of hematologic malignancies in women.
The study is the first prospective, population-based study of residential radon exposure and hematologic malignancy risk.
Therefore, the researchers caution that it requires replication to better understand the association and whether it truly differs by sex.
Lauren Teras, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues conducted this study and reported the results in Environmental Research.
Radon is a naturally occurring byproduct of the decay of radium and is a known human lung carcinogen. It is the second-leading cause of lung cancer in the US.
Modeling studies have shown that radon delivers a non-negligible dose of alpha radiation to the bone marrow and therefore could increase the risk of hematologic malignancies. However, studies investigating the link between radon and hematologic malignancies have produced inconsistent results.
For the current study, Dr Teras and her colleagues used data from the American Cancer Society Cancer Prevention Study-II Nutrition Cohort to examine the association between county-level residential radon exposure and the risk of hematologic cancer.
The analysis included 140,652 participants, including 3019 who had hematologic malignancies during 19 years of follow-up (1992 to 2011).
The researchers found that women living in counties with the highest mean radon concentration (> 148 Bq/m3) had a significantly higher risk of developing a hematologic malignancy than women living in counties with the lowest radon levels (< 74 Bq/m3).
The adjusted hazard ratio (adjusted for age, race, family history of hematologic malignancy, etc.) was 1.63 (P=0.0010).
The researchers also found evidence of a dose-response relationship, with an adjusted hazard ratio of 1.38 (P=0.001).
The team said there was evidence of a positive exposure-response relationship between radon concentration and the risk of all lymphoid malignancy subtypes in women. But the highest risk was observed for follicular lymphoma, with an adjusted hazard ratio of 2.74 (P=0.02).
On the other hand, there was a non-significant inverse association between radon and myeloid leukemias in women.
There was no association between hematologic malignancy and radon exposure among the men.
The researchers said a possible explanation for this finding is that men may have a higher baseline risk of hematologic malignancy, possibly because of more exposure to occupational or other risk factors, which would reduce the impact of any additional risk from residential radon.
In women, who have a smaller baseline risk, residential radon exposure might be a larger contributor to overall risk.
Another reason for the sex difference observed in this study may be that the women of this generation spent more time in their homes, so they had more residential exposure than men.
“The overall lifetime risk of hematological cancers in the United States is about 2%, so even a 60% relative increase would still mean a relatively small absolute risk,” Dr Teras noted.
“Nonetheless, radon is already associated with lung cancer, and if other studies confirm the link to blood cancers, we think it would warrant strengthened public health efforts to mitigate residential radon risks.” ![]()

New research suggests there is a significant positive association between high levels of residential radon and the risk of hematologic malignancies in women.
The study is the first prospective, population-based study of residential radon exposure and hematologic malignancy risk.
Therefore, the researchers caution that it requires replication to better understand the association and whether it truly differs by sex.
Lauren Teras, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues conducted this study and reported the results in Environmental Research.
Radon is a naturally occurring byproduct of the decay of radium and is a known human lung carcinogen. It is the second-leading cause of lung cancer in the US.
Modeling studies have shown that radon delivers a non-negligible dose of alpha radiation to the bone marrow and therefore could increase the risk of hematologic malignancies. However, studies investigating the link between radon and hematologic malignancies have produced inconsistent results.
For the current study, Dr Teras and her colleagues used data from the American Cancer Society Cancer Prevention Study-II Nutrition Cohort to examine the association between county-level residential radon exposure and the risk of hematologic cancer.
The analysis included 140,652 participants, including 3019 who had hematologic malignancies during 19 years of follow-up (1992 to 2011).
The researchers found that women living in counties with the highest mean radon concentration (> 148 Bq/m3) had a significantly higher risk of developing a hematologic malignancy than women living in counties with the lowest radon levels (< 74 Bq/m3).
The adjusted hazard ratio (adjusted for age, race, family history of hematologic malignancy, etc.) was 1.63 (P=0.0010).
The researchers also found evidence of a dose-response relationship, with an adjusted hazard ratio of 1.38 (P=0.001).
The team said there was evidence of a positive exposure-response relationship between radon concentration and the risk of all lymphoid malignancy subtypes in women. But the highest risk was observed for follicular lymphoma, with an adjusted hazard ratio of 2.74 (P=0.02).
On the other hand, there was a non-significant inverse association between radon and myeloid leukemias in women.
There was no association between hematologic malignancy and radon exposure among the men.
The researchers said a possible explanation for this finding is that men may have a higher baseline risk of hematologic malignancy, possibly because of more exposure to occupational or other risk factors, which would reduce the impact of any additional risk from residential radon.
In women, who have a smaller baseline risk, residential radon exposure might be a larger contributor to overall risk.
Another reason for the sex difference observed in this study may be that the women of this generation spent more time in their homes, so they had more residential exposure than men.
“The overall lifetime risk of hematological cancers in the United States is about 2%, so even a 60% relative increase would still mean a relatively small absolute risk,” Dr Teras noted.
“Nonetheless, radon is already associated with lung cancer, and if other studies confirm the link to blood cancers, we think it would warrant strengthened public health efforts to mitigate residential radon risks.” ![]()

New research suggests there is a significant positive association between high levels of residential radon and the risk of hematologic malignancies in women.
The study is the first prospective, population-based study of residential radon exposure and hematologic malignancy risk.
Therefore, the researchers caution that it requires replication to better understand the association and whether it truly differs by sex.
Lauren Teras, PhD, of the American Cancer Society in Atlanta, Georgia, and her colleagues conducted this study and reported the results in Environmental Research.
Radon is a naturally occurring byproduct of the decay of radium and is a known human lung carcinogen. It is the second-leading cause of lung cancer in the US.
Modeling studies have shown that radon delivers a non-negligible dose of alpha radiation to the bone marrow and therefore could increase the risk of hematologic malignancies. However, studies investigating the link between radon and hematologic malignancies have produced inconsistent results.
For the current study, Dr Teras and her colleagues used data from the American Cancer Society Cancer Prevention Study-II Nutrition Cohort to examine the association between county-level residential radon exposure and the risk of hematologic cancer.
The analysis included 140,652 participants, including 3019 who had hematologic malignancies during 19 years of follow-up (1992 to 2011).
The researchers found that women living in counties with the highest mean radon concentration (> 148 Bq/m3) had a significantly higher risk of developing a hematologic malignancy than women living in counties with the lowest radon levels (< 74 Bq/m3).
The adjusted hazard ratio (adjusted for age, race, family history of hematologic malignancy, etc.) was 1.63 (P=0.0010).
The researchers also found evidence of a dose-response relationship, with an adjusted hazard ratio of 1.38 (P=0.001).
The team said there was evidence of a positive exposure-response relationship between radon concentration and the risk of all lymphoid malignancy subtypes in women. But the highest risk was observed for follicular lymphoma, with an adjusted hazard ratio of 2.74 (P=0.02).
On the other hand, there was a non-significant inverse association between radon and myeloid leukemias in women.
There was no association between hematologic malignancy and radon exposure among the men.
The researchers said a possible explanation for this finding is that men may have a higher baseline risk of hematologic malignancy, possibly because of more exposure to occupational or other risk factors, which would reduce the impact of any additional risk from residential radon.
In women, who have a smaller baseline risk, residential radon exposure might be a larger contributor to overall risk.
Another reason for the sex difference observed in this study may be that the women of this generation spent more time in their homes, so they had more residential exposure than men.
“The overall lifetime risk of hematological cancers in the United States is about 2%, so even a 60% relative increase would still mean a relatively small absolute risk,” Dr Teras noted.
“Nonetheless, radon is already associated with lung cancer, and if other studies confirm the link to blood cancers, we think it would warrant strengthened public health efforts to mitigate residential radon risks.” ![]()
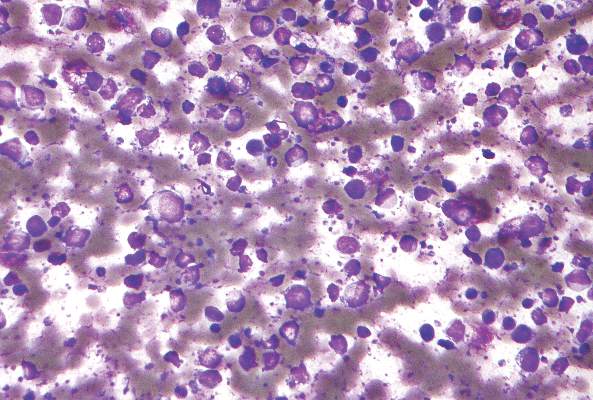
CHMP recommends approving drug to treat FL

The European Medicine Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended that obinutuzumab (Gazyvaro), an anti-CD20 monoclonal antibody, be approved for use in patients with follicular lymphoma (FL).
The recommended indication is for obinutuzumab to be given first in combination with bendamustine and then as maintenance therapy in FL patients who did not respond to, progressed during, or progressed up to 6 months after treatment with rituximab or a rituximab-containing regimen.
Based on the CHMP’s recommendation, a final decision regarding the approval of obinutuzumab in FL is expected from the European Commission in the coming months.
Obinutuzumab is already approved in the European Union for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia and comorbidities that make them unsuitable for full-dose fludarabine-based therapy.
Obinutuzumab is being developed by Roche.
GADOLIN trial
The CHMP’s recommendation to approve obinutuzumab in FL is based on results from the phase 3 GADOLIN trial. The study included 413 patients with rituximab-refractory non-Hodgkin lymphoma, including 321 patients with FL, 46 with marginal zone lymphoma, and 28 with small lymphocytic lymphoma.
The patients were randomized to receive bendamustine alone (control arm) or a combination of bendamustine and obinutuzumab followed by obinutuzumab maintenance (every 2 months for 2 years or until progression).
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC). The secondary endpoints were PFS assessed by investigator review, best overall response, complete response (CR), partial response (PR), duration of response, overall survival, and safety profile.
Among patients with FL, the obinutuzumab regimen improved PFS compared to bendamustine alone, as assessed by IRC (hazard ratio [HR]=0.48, P<0.0001). The median PFS was not reached in patients receiving the obinutuzumab regimen but was 13.8 months in those receiving bendamustine alone.
Investigator-assessed PFS was consistent with IRC-assessed PFS. Investigators said the median PFS with the obinutuzumab regimen was more than double that with bendamustine alone—29.2 months vs 13.7 months (HR=0.48, P<0.0001).
The best overall response for patients receiving the obinutuzumab regimen was 78.7% (15.5% CR, 63.2% PR), compared to 74.7% for those receiving bendamustine alone (18.7% CR, 56% PR), as assessed by the IRC.
The median duration of response was not reached for patients receiving the obinutuzumab regimen and was 11.6 months for those receiving bendamustine alone.
The median overall survival has not yet been reached in either study arm.
The most common grade 3/4 adverse events observed in patients receiving the obinutuzumab regimen were neutropenia (33%), infusion reactions (11%), and thrombocytopenia (10%).
The most common adverse events of any grade were infusion reactions (69%), neutropenia (35%), nausea (54%), fatigue (39%), cough (26%), diarrhea (27%), constipation (19%), fever (18%), thrombocytopenia (15%), vomiting (22%), upper respiratory tract infection (13%), decreased appetite (18%), joint or muscle pain (12%), sinusitis (12%), anemia (12%), general weakness (11%), and urinary tract infection (10%). ![]()

The European Medicine Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended that obinutuzumab (Gazyvaro), an anti-CD20 monoclonal antibody, be approved for use in patients with follicular lymphoma (FL).
The recommended indication is for obinutuzumab to be given first in combination with bendamustine and then as maintenance therapy in FL patients who did not respond to, progressed during, or progressed up to 6 months after treatment with rituximab or a rituximab-containing regimen.
Based on the CHMP’s recommendation, a final decision regarding the approval of obinutuzumab in FL is expected from the European Commission in the coming months.
Obinutuzumab is already approved in the European Union for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia and comorbidities that make them unsuitable for full-dose fludarabine-based therapy.
Obinutuzumab is being developed by Roche.
GADOLIN trial
The CHMP’s recommendation to approve obinutuzumab in FL is based on results from the phase 3 GADOLIN trial. The study included 413 patients with rituximab-refractory non-Hodgkin lymphoma, including 321 patients with FL, 46 with marginal zone lymphoma, and 28 with small lymphocytic lymphoma.
The patients were randomized to receive bendamustine alone (control arm) or a combination of bendamustine and obinutuzumab followed by obinutuzumab maintenance (every 2 months for 2 years or until progression).
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC). The secondary endpoints were PFS assessed by investigator review, best overall response, complete response (CR), partial response (PR), duration of response, overall survival, and safety profile.
Among patients with FL, the obinutuzumab regimen improved PFS compared to bendamustine alone, as assessed by IRC (hazard ratio [HR]=0.48, P<0.0001). The median PFS was not reached in patients receiving the obinutuzumab regimen but was 13.8 months in those receiving bendamustine alone.
Investigator-assessed PFS was consistent with IRC-assessed PFS. Investigators said the median PFS with the obinutuzumab regimen was more than double that with bendamustine alone—29.2 months vs 13.7 months (HR=0.48, P<0.0001).
The best overall response for patients receiving the obinutuzumab regimen was 78.7% (15.5% CR, 63.2% PR), compared to 74.7% for those receiving bendamustine alone (18.7% CR, 56% PR), as assessed by the IRC.
The median duration of response was not reached for patients receiving the obinutuzumab regimen and was 11.6 months for those receiving bendamustine alone.
The median overall survival has not yet been reached in either study arm.
The most common grade 3/4 adverse events observed in patients receiving the obinutuzumab regimen were neutropenia (33%), infusion reactions (11%), and thrombocytopenia (10%).
The most common adverse events of any grade were infusion reactions (69%), neutropenia (35%), nausea (54%), fatigue (39%), cough (26%), diarrhea (27%), constipation (19%), fever (18%), thrombocytopenia (15%), vomiting (22%), upper respiratory tract infection (13%), decreased appetite (18%), joint or muscle pain (12%), sinusitis (12%), anemia (12%), general weakness (11%), and urinary tract infection (10%). ![]()

The European Medicine Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended that obinutuzumab (Gazyvaro), an anti-CD20 monoclonal antibody, be approved for use in patients with follicular lymphoma (FL).
The recommended indication is for obinutuzumab to be given first in combination with bendamustine and then as maintenance therapy in FL patients who did not respond to, progressed during, or progressed up to 6 months after treatment with rituximab or a rituximab-containing regimen.
Based on the CHMP’s recommendation, a final decision regarding the approval of obinutuzumab in FL is expected from the European Commission in the coming months.
Obinutuzumab is already approved in the European Union for use in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia and comorbidities that make them unsuitable for full-dose fludarabine-based therapy.
Obinutuzumab is being developed by Roche.
GADOLIN trial
The CHMP’s recommendation to approve obinutuzumab in FL is based on results from the phase 3 GADOLIN trial. The study included 413 patients with rituximab-refractory non-Hodgkin lymphoma, including 321 patients with FL, 46 with marginal zone lymphoma, and 28 with small lymphocytic lymphoma.
The patients were randomized to receive bendamustine alone (control arm) or a combination of bendamustine and obinutuzumab followed by obinutuzumab maintenance (every 2 months for 2 years or until progression).
The primary endpoint of the study was progression-free survival (PFS), as assessed by an independent review committee (IRC). The secondary endpoints were PFS assessed by investigator review, best overall response, complete response (CR), partial response (PR), duration of response, overall survival, and safety profile.
Among patients with FL, the obinutuzumab regimen improved PFS compared to bendamustine alone, as assessed by IRC (hazard ratio [HR]=0.48, P<0.0001). The median PFS was not reached in patients receiving the obinutuzumab regimen but was 13.8 months in those receiving bendamustine alone.
Investigator-assessed PFS was consistent with IRC-assessed PFS. Investigators said the median PFS with the obinutuzumab regimen was more than double that with bendamustine alone—29.2 months vs 13.7 months (HR=0.48, P<0.0001).
The best overall response for patients receiving the obinutuzumab regimen was 78.7% (15.5% CR, 63.2% PR), compared to 74.7% for those receiving bendamustine alone (18.7% CR, 56% PR), as assessed by the IRC.
The median duration of response was not reached for patients receiving the obinutuzumab regimen and was 11.6 months for those receiving bendamustine alone.
The median overall survival has not yet been reached in either study arm.
The most common grade 3/4 adverse events observed in patients receiving the obinutuzumab regimen were neutropenia (33%), infusion reactions (11%), and thrombocytopenia (10%).
The most common adverse events of any grade were infusion reactions (69%), neutropenia (35%), nausea (54%), fatigue (39%), cough (26%), diarrhea (27%), constipation (19%), fever (18%), thrombocytopenia (15%), vomiting (22%), upper respiratory tract infection (13%), decreased appetite (18%), joint or muscle pain (12%), sinusitis (12%), anemia (12%), general weakness (11%), and urinary tract infection (10%). ![]()
Calcineurin-targeted therapies eyed for multiple myeloma
Treating multiple myeloma cells with panobinostat and FK506 reduced their viability by inhibiting expression of the PPP3CA catalytic subunit of calcineurin, according to researchers.
“The development of new calcineurin-targeted therapies, which inhibit PPP3CA–NF-kappaB signaling by small molecules, is expected to profoundly improve the treatment of multiple myeloma. This will overcome drug resistance and improve osteolytic lesions in a wide range of patients, including those receiving reduced intensity–conditioned allogeneic stem cell transplantation, who may be treated with panobinostat and FK506,” wrote Dr. Yoichi Imai of Tokyo Women’s Medical University in Japan, and his associates (JCI Insight 2016 doi: 10.1172/jci.insight.85061). .
Adding panobinostat to bortezomib and dexamethasone has been shown to improve progression-free survival in relapsed and refractory multiple myeloma, the researchers noted. Other studies have linked calcineurin activation to the pathogenesis of T-cell cancers, and calcineurin inhibition seems to be involved in defective B-cell activation, they added.
Eight candidate oncogenes were identified by use of the Gene Expression Omnibus. PPP3CA was expressed at significantly higher levels in multiple myeloma cells from patients with stage III disease compared with those with stage I disease (P = .016). Furthermore, levels of serum lactate dehydrogenase correlated with PPP3CA expression and with poor overall and progression-free survival. Patients with high PPP3CA expression also had high levels of alpha4 integrins, which mediate resistance to bortezomib and conventional chemotherapy, the researchers noted.
When multiple myeloma cells were exposed to either panobinostat or a control agent, PPP3CA dropped in the panobinostat-treated cells. Adding the proteasome inhibitor lactacystin to the mixture counteracted this effect, “supporting the possibility that PPP3CA expression was reduced through protein degradation by panobinostat,” they said.
Levels of PPP3CA expression were lower in multiple myeloma cells that were cotreated with an HDAC inhibitor (panobinostat or ACY-1215) and the immunosuppressive agent FK506 than in multiple myeloma cells that were exposed only to an HDAC inhibitor. In addition, the combination regimen blocked the formation of osteoclasts, which are involved in osteolytic lesions, the researchers noted.
Also, significantly higher PPP3CA expression, which correlated with worse progression-free survival, was noted in bortezomib-resistant patients, compared with bortezomib-sensitive patients.
“The cytotoxic effect exerted by panobinostat on CD20+ cells was subtle, while the addition of FK506 did not increase their viability,” the researchers reported. “Moreover, development of T and B lineage cells was normal in PPP3CA-deficient mice, and panobinostat did not compromise donor lymphocyte reconstitution in a mouse BM transplantation model. These results suggest that calcineurin-targeting therapy exerts an antimyeloma effect without inducing significant side effects in normal lymphoid systems.”
The Japan Society for the Promotion of Science, the Takeda Science Foundation, the International Myeloma Foundation, and the Japan Leukemia Research Fund funded the study. The investigators had no disclosures.
Treating multiple myeloma cells with panobinostat and FK506 reduced their viability by inhibiting expression of the PPP3CA catalytic subunit of calcineurin, according to researchers.
“The development of new calcineurin-targeted therapies, which inhibit PPP3CA–NF-kappaB signaling by small molecules, is expected to profoundly improve the treatment of multiple myeloma. This will overcome drug resistance and improve osteolytic lesions in a wide range of patients, including those receiving reduced intensity–conditioned allogeneic stem cell transplantation, who may be treated with panobinostat and FK506,” wrote Dr. Yoichi Imai of Tokyo Women’s Medical University in Japan, and his associates (JCI Insight 2016 doi: 10.1172/jci.insight.85061). .
Adding panobinostat to bortezomib and dexamethasone has been shown to improve progression-free survival in relapsed and refractory multiple myeloma, the researchers noted. Other studies have linked calcineurin activation to the pathogenesis of T-cell cancers, and calcineurin inhibition seems to be involved in defective B-cell activation, they added.
Eight candidate oncogenes were identified by use of the Gene Expression Omnibus. PPP3CA was expressed at significantly higher levels in multiple myeloma cells from patients with stage III disease compared with those with stage I disease (P = .016). Furthermore, levels of serum lactate dehydrogenase correlated with PPP3CA expression and with poor overall and progression-free survival. Patients with high PPP3CA expression also had high levels of alpha4 integrins, which mediate resistance to bortezomib and conventional chemotherapy, the researchers noted.
When multiple myeloma cells were exposed to either panobinostat or a control agent, PPP3CA dropped in the panobinostat-treated cells. Adding the proteasome inhibitor lactacystin to the mixture counteracted this effect, “supporting the possibility that PPP3CA expression was reduced through protein degradation by panobinostat,” they said.
Levels of PPP3CA expression were lower in multiple myeloma cells that were cotreated with an HDAC inhibitor (panobinostat or ACY-1215) and the immunosuppressive agent FK506 than in multiple myeloma cells that were exposed only to an HDAC inhibitor. In addition, the combination regimen blocked the formation of osteoclasts, which are involved in osteolytic lesions, the researchers noted.
Also, significantly higher PPP3CA expression, which correlated with worse progression-free survival, was noted in bortezomib-resistant patients, compared with bortezomib-sensitive patients.
“The cytotoxic effect exerted by panobinostat on CD20+ cells was subtle, while the addition of FK506 did not increase their viability,” the researchers reported. “Moreover, development of T and B lineage cells was normal in PPP3CA-deficient mice, and panobinostat did not compromise donor lymphocyte reconstitution in a mouse BM transplantation model. These results suggest that calcineurin-targeting therapy exerts an antimyeloma effect without inducing significant side effects in normal lymphoid systems.”
The Japan Society for the Promotion of Science, the Takeda Science Foundation, the International Myeloma Foundation, and the Japan Leukemia Research Fund funded the study. The investigators had no disclosures.
Treating multiple myeloma cells with panobinostat and FK506 reduced their viability by inhibiting expression of the PPP3CA catalytic subunit of calcineurin, according to researchers.
“The development of new calcineurin-targeted therapies, which inhibit PPP3CA–NF-kappaB signaling by small molecules, is expected to profoundly improve the treatment of multiple myeloma. This will overcome drug resistance and improve osteolytic lesions in a wide range of patients, including those receiving reduced intensity–conditioned allogeneic stem cell transplantation, who may be treated with panobinostat and FK506,” wrote Dr. Yoichi Imai of Tokyo Women’s Medical University in Japan, and his associates (JCI Insight 2016 doi: 10.1172/jci.insight.85061). .
Adding panobinostat to bortezomib and dexamethasone has been shown to improve progression-free survival in relapsed and refractory multiple myeloma, the researchers noted. Other studies have linked calcineurin activation to the pathogenesis of T-cell cancers, and calcineurin inhibition seems to be involved in defective B-cell activation, they added.
Eight candidate oncogenes were identified by use of the Gene Expression Omnibus. PPP3CA was expressed at significantly higher levels in multiple myeloma cells from patients with stage III disease compared with those with stage I disease (P = .016). Furthermore, levels of serum lactate dehydrogenase correlated with PPP3CA expression and with poor overall and progression-free survival. Patients with high PPP3CA expression also had high levels of alpha4 integrins, which mediate resistance to bortezomib and conventional chemotherapy, the researchers noted.
When multiple myeloma cells were exposed to either panobinostat or a control agent, PPP3CA dropped in the panobinostat-treated cells. Adding the proteasome inhibitor lactacystin to the mixture counteracted this effect, “supporting the possibility that PPP3CA expression was reduced through protein degradation by panobinostat,” they said.
Levels of PPP3CA expression were lower in multiple myeloma cells that were cotreated with an HDAC inhibitor (panobinostat or ACY-1215) and the immunosuppressive agent FK506 than in multiple myeloma cells that were exposed only to an HDAC inhibitor. In addition, the combination regimen blocked the formation of osteoclasts, which are involved in osteolytic lesions, the researchers noted.
Also, significantly higher PPP3CA expression, which correlated with worse progression-free survival, was noted in bortezomib-resistant patients, compared with bortezomib-sensitive patients.
“The cytotoxic effect exerted by panobinostat on CD20+ cells was subtle, while the addition of FK506 did not increase their viability,” the researchers reported. “Moreover, development of T and B lineage cells was normal in PPP3CA-deficient mice, and panobinostat did not compromise donor lymphocyte reconstitution in a mouse BM transplantation model. These results suggest that calcineurin-targeting therapy exerts an antimyeloma effect without inducing significant side effects in normal lymphoid systems.”
The Japan Society for the Promotion of Science, the Takeda Science Foundation, the International Myeloma Foundation, and the Japan Leukemia Research Fund funded the study. The investigators had no disclosures.
FROM JCI INSIGHT
Key clinical point: Treating multiple myeloma cells with panobinostat and FK506 reduced their viability by inhibiting expression of the PPP3CA catalytic subunit of calcineurin.
Major finding: PPP3CA was associated with MM cell viability and osteoclast formation, and was degraded by panobinostat through HDAC inhibition.
Data source: An in vitro and in vivo laboratory study of MM in human and mouse models.
Disclosures: The Japan Society for the Promotion of Science, the Takeda Science Foundation, the International Myeloma Foundation, and the Japan Leukemia Research Fund funded the study. The investigators had no disclosures.
Cancer diagnosis linked to mental health disorders

A recent cancer diagnosis is associated with an increased risk for mental health disorders and increased use of psychiatric medications, according to a large, nationwide study conducted in Sweden.
Overall, there was an increased risk of mental health disorders from 10 months before a cancer diagnosis that peaked during the first week after diagnosis and decreased after that, although the risk remained elevated at 10 years after diagnosis.
In addition, there was an increased use of psychiatric medications from 1 month before cancer diagnosis that peaked at about 3 months after diagnosis and remained elevated 2 years after diagnosis.
Donghao Lu, MD, of the Karolinska Institutet in Stockholm, Sweden and colleagues conducted this study and reported the results in JAMA Oncology.
The study included 304,118 patients with cancer and 3,041,174 cancer-free individuals randomly selected from the Swedish population for comparison.
The researchers investigated changes in risk for several common and potentially stress-related mental disorders—including depression, anxiety, substance abuse, somatoform/conversion disorder, and stress reaction/adjustment disorder—from the cancer diagnostic workup through to post-diagnosis.
They found the relative rate for all of the mental disorders studied started to increase from 10 months before cancer diagnosis, with a hazard ratio [HR] of 1.1 (95%CI, 1.1-1.2).
The rate peaked during the first week after diagnosis, with an HR of 6.7 (95%CI, 6.1-7.4). It decreased rapidly thereafter but was still elevated 10 years after diagnosis, with an HR of 1.1 (95%CI, 1.1-1.2).
The rate elevation was clear for all of the main cancers, including hematologic malignancies, except for nonmelanoma skin cancer.
Among the cancer patients, the mental disorder with the highest cumulative incidence was depression. This was followed by anxiety and stress reaction/adjustment disorder.
When compared to controls, the cancer patients had a higher cumulative incidence of most of the mental disorders. The exception was somatoform/conversion disorder.
The researchers also examined the use of psychiatric medications for patients with cancer to assess milder mental health conditions and symptoms.
The team found an increased use of psychiatric medications in cancer patients compared to controls, from 1 month before diagnosis—12.2% vs 11.7% (P=0.04)—that peaked at about 3 months after diagnosis—18.1% vs 11.9% (P<0.001)—and was still elevated 2 years after diagnosis—15.4% vs 12.7% (P<0.001).
The researchers said the results of this study support the existing guidelines of integrating psychological management into cancer care and call for extended vigilance for multiple mental disorders starting from the time of the cancer diagnostic workup. ![]()

A recent cancer diagnosis is associated with an increased risk for mental health disorders and increased use of psychiatric medications, according to a large, nationwide study conducted in Sweden.
Overall, there was an increased risk of mental health disorders from 10 months before a cancer diagnosis that peaked during the first week after diagnosis and decreased after that, although the risk remained elevated at 10 years after diagnosis.
In addition, there was an increased use of psychiatric medications from 1 month before cancer diagnosis that peaked at about 3 months after diagnosis and remained elevated 2 years after diagnosis.
Donghao Lu, MD, of the Karolinska Institutet in Stockholm, Sweden and colleagues conducted this study and reported the results in JAMA Oncology.
The study included 304,118 patients with cancer and 3,041,174 cancer-free individuals randomly selected from the Swedish population for comparison.
The researchers investigated changes in risk for several common and potentially stress-related mental disorders—including depression, anxiety, substance abuse, somatoform/conversion disorder, and stress reaction/adjustment disorder—from the cancer diagnostic workup through to post-diagnosis.
They found the relative rate for all of the mental disorders studied started to increase from 10 months before cancer diagnosis, with a hazard ratio [HR] of 1.1 (95%CI, 1.1-1.2).
The rate peaked during the first week after diagnosis, with an HR of 6.7 (95%CI, 6.1-7.4). It decreased rapidly thereafter but was still elevated 10 years after diagnosis, with an HR of 1.1 (95%CI, 1.1-1.2).
The rate elevation was clear for all of the main cancers, including hematologic malignancies, except for nonmelanoma skin cancer.
Among the cancer patients, the mental disorder with the highest cumulative incidence was depression. This was followed by anxiety and stress reaction/adjustment disorder.
When compared to controls, the cancer patients had a higher cumulative incidence of most of the mental disorders. The exception was somatoform/conversion disorder.
The researchers also examined the use of psychiatric medications for patients with cancer to assess milder mental health conditions and symptoms.
The team found an increased use of psychiatric medications in cancer patients compared to controls, from 1 month before diagnosis—12.2% vs 11.7% (P=0.04)—that peaked at about 3 months after diagnosis—18.1% vs 11.9% (P<0.001)—and was still elevated 2 years after diagnosis—15.4% vs 12.7% (P<0.001).
The researchers said the results of this study support the existing guidelines of integrating psychological management into cancer care and call for extended vigilance for multiple mental disorders starting from the time of the cancer diagnostic workup. ![]()

A recent cancer diagnosis is associated with an increased risk for mental health disorders and increased use of psychiatric medications, according to a large, nationwide study conducted in Sweden.
Overall, there was an increased risk of mental health disorders from 10 months before a cancer diagnosis that peaked during the first week after diagnosis and decreased after that, although the risk remained elevated at 10 years after diagnosis.
In addition, there was an increased use of psychiatric medications from 1 month before cancer diagnosis that peaked at about 3 months after diagnosis and remained elevated 2 years after diagnosis.
Donghao Lu, MD, of the Karolinska Institutet in Stockholm, Sweden and colleagues conducted this study and reported the results in JAMA Oncology.
The study included 304,118 patients with cancer and 3,041,174 cancer-free individuals randomly selected from the Swedish population for comparison.
The researchers investigated changes in risk for several common and potentially stress-related mental disorders—including depression, anxiety, substance abuse, somatoform/conversion disorder, and stress reaction/adjustment disorder—from the cancer diagnostic workup through to post-diagnosis.
They found the relative rate for all of the mental disorders studied started to increase from 10 months before cancer diagnosis, with a hazard ratio [HR] of 1.1 (95%CI, 1.1-1.2).
The rate peaked during the first week after diagnosis, with an HR of 6.7 (95%CI, 6.1-7.4). It decreased rapidly thereafter but was still elevated 10 years after diagnosis, with an HR of 1.1 (95%CI, 1.1-1.2).
The rate elevation was clear for all of the main cancers, including hematologic malignancies, except for nonmelanoma skin cancer.
Among the cancer patients, the mental disorder with the highest cumulative incidence was depression. This was followed by anxiety and stress reaction/adjustment disorder.
When compared to controls, the cancer patients had a higher cumulative incidence of most of the mental disorders. The exception was somatoform/conversion disorder.
The researchers also examined the use of psychiatric medications for patients with cancer to assess milder mental health conditions and symptoms.
The team found an increased use of psychiatric medications in cancer patients compared to controls, from 1 month before diagnosis—12.2% vs 11.7% (P=0.04)—that peaked at about 3 months after diagnosis—18.1% vs 11.9% (P<0.001)—and was still elevated 2 years after diagnosis—15.4% vs 12.7% (P<0.001).
The researchers said the results of this study support the existing guidelines of integrating psychological management into cancer care and call for extended vigilance for multiple mental disorders starting from the time of the cancer diagnostic workup. ![]()
Costs for orally administered cancer drugs on the rise

Photo courtesy of the CDC
New orally administered cancer drugs are much more expensive in their first year on the market than such drugs launched about 15 years ago, according to a study published in JAMA Oncology.
The research showed that a month of treatment with orally administered cancer drugs introduced in 2014 was, on average, 6 times more expensive at launch than monthly treatment costs for such drugs introduced in 2000, after adjusting for inflation.
In addition, most existing therapies had substantial price increases from the time they were launched to 2014.
“The major trend here is that these products are just getting more expensive over time,” said study author Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
For this study, Dr Dusetzina evaluated what commercial health insurance companies and patients paid for prescription fills—before rebates and discounts—for 32 orally administered cancer drugs from 2000 to 2014. The information came from the TruvenHealth MarketScan Commercial Claims and Encounters database.
The data showed that orally administered drugs approved in 2000 cost an average of $1869 (95% CI, $1648-$2121) per month, compared to $11,325 (95% CI, $10 989-$11 671) for those approved in 2014.
When Dr Dusetzina compared changes in spending by year from a product’s launch to 2014, she observed increases in most of the drugs studied.
The drugs with the largest increases in monthly spending were thalidomide, which increased from $1869 to $7564 ($5695) and imatinib, which increased from $3346 to $8479 ($5133).
However, 2 drugs showed decreases in mean monthly spending between their launch and 2014. Monthly spending for lenalidomide decreased from $10,109 to $9640 ($469), and monthly spending for vorinostat decreased from $9755 to $7592 ($2163).
Dr Dusetzina pointed out that the amount patients pay for these drugs depends on their healthcare benefits. However, the high prices are being passed along to patients more and more, potentially affecting the patients’ access to these drugs.
“Patients are increasingly taking on the burden of paying for these high-cost specialty drugs as plans move toward use of higher deductibles and co-insurance—where a patient will pay a percentage of the drug cost rather than a flat copay,” Dr Dusetzina said.
She noted that while this study did account for payments by commercial health plans, it did not account for spending by Medicaid and Medicare, which may differ. In addition, only the products that were dispensed and reimbursed by commercial health plans were included, which may have excluded rarely used or recently approved products. ![]()

Photo courtesy of the CDC
New orally administered cancer drugs are much more expensive in their first year on the market than such drugs launched about 15 years ago, according to a study published in JAMA Oncology.
The research showed that a month of treatment with orally administered cancer drugs introduced in 2014 was, on average, 6 times more expensive at launch than monthly treatment costs for such drugs introduced in 2000, after adjusting for inflation.
In addition, most existing therapies had substantial price increases from the time they were launched to 2014.
“The major trend here is that these products are just getting more expensive over time,” said study author Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
For this study, Dr Dusetzina evaluated what commercial health insurance companies and patients paid for prescription fills—before rebates and discounts—for 32 orally administered cancer drugs from 2000 to 2014. The information came from the TruvenHealth MarketScan Commercial Claims and Encounters database.
The data showed that orally administered drugs approved in 2000 cost an average of $1869 (95% CI, $1648-$2121) per month, compared to $11,325 (95% CI, $10 989-$11 671) for those approved in 2014.
When Dr Dusetzina compared changes in spending by year from a product’s launch to 2014, she observed increases in most of the drugs studied.
The drugs with the largest increases in monthly spending were thalidomide, which increased from $1869 to $7564 ($5695) and imatinib, which increased from $3346 to $8479 ($5133).
However, 2 drugs showed decreases in mean monthly spending between their launch and 2014. Monthly spending for lenalidomide decreased from $10,109 to $9640 ($469), and monthly spending for vorinostat decreased from $9755 to $7592 ($2163).
Dr Dusetzina pointed out that the amount patients pay for these drugs depends on their healthcare benefits. However, the high prices are being passed along to patients more and more, potentially affecting the patients’ access to these drugs.
“Patients are increasingly taking on the burden of paying for these high-cost specialty drugs as plans move toward use of higher deductibles and co-insurance—where a patient will pay a percentage of the drug cost rather than a flat copay,” Dr Dusetzina said.
She noted that while this study did account for payments by commercial health plans, it did not account for spending by Medicaid and Medicare, which may differ. In addition, only the products that were dispensed and reimbursed by commercial health plans were included, which may have excluded rarely used or recently approved products. ![]()

Photo courtesy of the CDC
New orally administered cancer drugs are much more expensive in their first year on the market than such drugs launched about 15 years ago, according to a study published in JAMA Oncology.
The research showed that a month of treatment with orally administered cancer drugs introduced in 2014 was, on average, 6 times more expensive at launch than monthly treatment costs for such drugs introduced in 2000, after adjusting for inflation.
In addition, most existing therapies had substantial price increases from the time they were launched to 2014.
“The major trend here is that these products are just getting more expensive over time,” said study author Stacie Dusetzina, PhD, of the University of North Carolina at Chapel Hill.
For this study, Dr Dusetzina evaluated what commercial health insurance companies and patients paid for prescription fills—before rebates and discounts—for 32 orally administered cancer drugs from 2000 to 2014. The information came from the TruvenHealth MarketScan Commercial Claims and Encounters database.
The data showed that orally administered drugs approved in 2000 cost an average of $1869 (95% CI, $1648-$2121) per month, compared to $11,325 (95% CI, $10 989-$11 671) for those approved in 2014.
When Dr Dusetzina compared changes in spending by year from a product’s launch to 2014, she observed increases in most of the drugs studied.
The drugs with the largest increases in monthly spending were thalidomide, which increased from $1869 to $7564 ($5695) and imatinib, which increased from $3346 to $8479 ($5133).
However, 2 drugs showed decreases in mean monthly spending between their launch and 2014. Monthly spending for lenalidomide decreased from $10,109 to $9640 ($469), and monthly spending for vorinostat decreased from $9755 to $7592 ($2163).
Dr Dusetzina pointed out that the amount patients pay for these drugs depends on their healthcare benefits. However, the high prices are being passed along to patients more and more, potentially affecting the patients’ access to these drugs.
“Patients are increasingly taking on the burden of paying for these high-cost specialty drugs as plans move toward use of higher deductibles and co-insurance—where a patient will pay a percentage of the drug cost rather than a flat copay,” Dr Dusetzina said.
She noted that while this study did account for payments by commercial health plans, it did not account for spending by Medicaid and Medicare, which may differ. In addition, only the products that were dispensed and reimbursed by commercial health plans were included, which may have excluded rarely used or recently approved products. ![]()
Low transformation rate in nodular lymphocyte–predominant Hodgkin lymphoma
Fewer than 8% of cases of nodular lymphocyte–predominant Hodgkin lymphoma (NLPHL) transformed to diffuse large B-cell lymphoma (DLBCL), based on a large prospective single-center study with long-term follow-up.
This rate was lower than the risk of transformation reported for transformed follicular lymphoma or chronic lymphocytic leukemia, according to Dr. Saad Kenderian and his associates at the Mayo Clinic, Rochester, Minn. Transformation was significantly associated with splenic involvement at presentation and with prior chemotherapy exposure, but did not worsen overall survival, they added.
“To our knowledge, this cohort represents the largest analysis to date of consecutive patients with NLPHL,” they said.
The study comprised 222 patients with newly diagnosed NLPHL who were treated at Mayo Clinic between 1970 and 2011. Median age at diagnosis was 40 years, and two-thirds of patients were men. The median follow-up period was 16 years (Blood 2016;12:1960-6. doi: 10.1182/blood-2015-08-665505).
During follow up, 17 cases (7.6%) transformed to DLBCL, for a transformation rate of 0.74 cases for every 100 patient-years, the investigators said. Median time to transformation was 35 months (range, 6-268 months). Predictors of transformation included any prior chemotherapy exposure (P = .04) and splenic involvement (P = .03). The rates of 40-year freedom from transformation were 87% when there was no splenic involvement and 21% when the spleen was involved, and were 87% if radiation therapy was used as a single modality compared with 77% in patients treated with prior chemotherapy or chemoradiation.
Five-year overall survival was 76% in patients with transformed disease, which was similar to overall survival among patients whose disease did not transform to DLBCL, the researchers noted.
Other studies of NLPHL have reported anywhere from a 2% to a 17% transformation rate, but those studies had smaller sample sizes, shorter follow-up periods, and less rigorous enrollment criteria and methods to confirm transformation, the investigators noted. “The finding of splenic involvement as a risk factor for transformation was reported by previous investigators. Interestingly, the association between exposure to prior chemotherapy and reduced freedom from transformation has not been reported in the past, but it has been observed in other low-grade lymphoma studies,” they added. “In contrast to follicular lymphoma, transformed NLPHL is not associated with an adverse impact on OS, suggesting a possibly different biology of transformation.”
The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
Kenderian et al. report a lower rate of transformation (7.6%) to diffuse large B-cell lymphoma for patients with nodular lymphocyte–predominant Hodgkin lymphoma compared with other series and found that transformation did not have a negative impact on overall survival. Reassuringly, even if transformation occurs, it is generally at a low rate. Also, these patients do well with additional treatment and do not have worse overall survival. At the MD Anderson Cancer Center, we have used a regimen based on R-CHOP and have not seen transformations. But only through large cooperative clinical trials can we determine whether R-CHOP or other more novel regimens are actually superior to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or rituximab (R)-ABVD for patients at high risk of transformation.
Dr. Michelle Fanale is at the University of Texas MD Anderson Cancer Center, Houston. She had no disclosures. These comments are from her editorial (Blood 2016;1927:1946-7 doi: 10.1182/blood-2016-03-699108).
Kenderian et al. report a lower rate of transformation (7.6%) to diffuse large B-cell lymphoma for patients with nodular lymphocyte–predominant Hodgkin lymphoma compared with other series and found that transformation did not have a negative impact on overall survival. Reassuringly, even if transformation occurs, it is generally at a low rate. Also, these patients do well with additional treatment and do not have worse overall survival. At the MD Anderson Cancer Center, we have used a regimen based on R-CHOP and have not seen transformations. But only through large cooperative clinical trials can we determine whether R-CHOP or other more novel regimens are actually superior to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or rituximab (R)-ABVD for patients at high risk of transformation.
Dr. Michelle Fanale is at the University of Texas MD Anderson Cancer Center, Houston. She had no disclosures. These comments are from her editorial (Blood 2016;1927:1946-7 doi: 10.1182/blood-2016-03-699108).
Kenderian et al. report a lower rate of transformation (7.6%) to diffuse large B-cell lymphoma for patients with nodular lymphocyte–predominant Hodgkin lymphoma compared with other series and found that transformation did not have a negative impact on overall survival. Reassuringly, even if transformation occurs, it is generally at a low rate. Also, these patients do well with additional treatment and do not have worse overall survival. At the MD Anderson Cancer Center, we have used a regimen based on R-CHOP and have not seen transformations. But only through large cooperative clinical trials can we determine whether R-CHOP or other more novel regimens are actually superior to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) or rituximab (R)-ABVD for patients at high risk of transformation.
Dr. Michelle Fanale is at the University of Texas MD Anderson Cancer Center, Houston. She had no disclosures. These comments are from her editorial (Blood 2016;1927:1946-7 doi: 10.1182/blood-2016-03-699108).
Fewer than 8% of cases of nodular lymphocyte–predominant Hodgkin lymphoma (NLPHL) transformed to diffuse large B-cell lymphoma (DLBCL), based on a large prospective single-center study with long-term follow-up.
This rate was lower than the risk of transformation reported for transformed follicular lymphoma or chronic lymphocytic leukemia, according to Dr. Saad Kenderian and his associates at the Mayo Clinic, Rochester, Minn. Transformation was significantly associated with splenic involvement at presentation and with prior chemotherapy exposure, but did not worsen overall survival, they added.
“To our knowledge, this cohort represents the largest analysis to date of consecutive patients with NLPHL,” they said.
The study comprised 222 patients with newly diagnosed NLPHL who were treated at Mayo Clinic between 1970 and 2011. Median age at diagnosis was 40 years, and two-thirds of patients were men. The median follow-up period was 16 years (Blood 2016;12:1960-6. doi: 10.1182/blood-2015-08-665505).
During follow up, 17 cases (7.6%) transformed to DLBCL, for a transformation rate of 0.74 cases for every 100 patient-years, the investigators said. Median time to transformation was 35 months (range, 6-268 months). Predictors of transformation included any prior chemotherapy exposure (P = .04) and splenic involvement (P = .03). The rates of 40-year freedom from transformation were 87% when there was no splenic involvement and 21% when the spleen was involved, and were 87% if radiation therapy was used as a single modality compared with 77% in patients treated with prior chemotherapy or chemoradiation.
Five-year overall survival was 76% in patients with transformed disease, which was similar to overall survival among patients whose disease did not transform to DLBCL, the researchers noted.
Other studies of NLPHL have reported anywhere from a 2% to a 17% transformation rate, but those studies had smaller sample sizes, shorter follow-up periods, and less rigorous enrollment criteria and methods to confirm transformation, the investigators noted. “The finding of splenic involvement as a risk factor for transformation was reported by previous investigators. Interestingly, the association between exposure to prior chemotherapy and reduced freedom from transformation has not been reported in the past, but it has been observed in other low-grade lymphoma studies,” they added. “In contrast to follicular lymphoma, transformed NLPHL is not associated with an adverse impact on OS, suggesting a possibly different biology of transformation.”
The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
Fewer than 8% of cases of nodular lymphocyte–predominant Hodgkin lymphoma (NLPHL) transformed to diffuse large B-cell lymphoma (DLBCL), based on a large prospective single-center study with long-term follow-up.
This rate was lower than the risk of transformation reported for transformed follicular lymphoma or chronic lymphocytic leukemia, according to Dr. Saad Kenderian and his associates at the Mayo Clinic, Rochester, Minn. Transformation was significantly associated with splenic involvement at presentation and with prior chemotherapy exposure, but did not worsen overall survival, they added.
“To our knowledge, this cohort represents the largest analysis to date of consecutive patients with NLPHL,” they said.
The study comprised 222 patients with newly diagnosed NLPHL who were treated at Mayo Clinic between 1970 and 2011. Median age at diagnosis was 40 years, and two-thirds of patients were men. The median follow-up period was 16 years (Blood 2016;12:1960-6. doi: 10.1182/blood-2015-08-665505).
During follow up, 17 cases (7.6%) transformed to DLBCL, for a transformation rate of 0.74 cases for every 100 patient-years, the investigators said. Median time to transformation was 35 months (range, 6-268 months). Predictors of transformation included any prior chemotherapy exposure (P = .04) and splenic involvement (P = .03). The rates of 40-year freedom from transformation were 87% when there was no splenic involvement and 21% when the spleen was involved, and were 87% if radiation therapy was used as a single modality compared with 77% in patients treated with prior chemotherapy or chemoradiation.
Five-year overall survival was 76% in patients with transformed disease, which was similar to overall survival among patients whose disease did not transform to DLBCL, the researchers noted.
Other studies of NLPHL have reported anywhere from a 2% to a 17% transformation rate, but those studies had smaller sample sizes, shorter follow-up periods, and less rigorous enrollment criteria and methods to confirm transformation, the investigators noted. “The finding of splenic involvement as a risk factor for transformation was reported by previous investigators. Interestingly, the association between exposure to prior chemotherapy and reduced freedom from transformation has not been reported in the past, but it has been observed in other low-grade lymphoma studies,” they added. “In contrast to follicular lymphoma, transformed NLPHL is not associated with an adverse impact on OS, suggesting a possibly different biology of transformation.”
The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
FROM BLOOD
Key clinical point: The risk of transformation to diffuse large B-cell lymphoma is low in patients with nodular lymphocyte–predominant Hodgkin lymphoma.
Major finding: Only 7.6% of cases transformed over a median of 16 years of follow-up, and transformation did not worsen overall survival.
Data source: A prospective single-center study of 222 consecutive adults with NLPHL.
Disclosures: The research was partially supported by Lymphoma SPORE and the Predolin Foundation. The investigators had no disclosures.
Multiple myeloma: newly approved drugs forge paradigm shift toward chronic disease
The pace of drug development for multiple myeloma was dizzying in 2015, with 5 regulatory approvals for the treatment of relapsed/refractory disease, 3 in a single month. As we stand on the brink of another paradigm shift in the management of this disease, we discuss the new classes of drugs and how they are shaping standard of care with the potential to make multiple myeloma a chronic disease.
Click on the PDF icon at the top of this introduction to read the full article.
The pace of drug development for multiple myeloma was dizzying in 2015, with 5 regulatory approvals for the treatment of relapsed/refractory disease, 3 in a single month. As we stand on the brink of another paradigm shift in the management of this disease, we discuss the new classes of drugs and how they are shaping standard of care with the potential to make multiple myeloma a chronic disease.
Click on the PDF icon at the top of this introduction to read the full article.
The pace of drug development for multiple myeloma was dizzying in 2015, with 5 regulatory approvals for the treatment of relapsed/refractory disease, 3 in a single month. As we stand on the brink of another paradigm shift in the management of this disease, we discuss the new classes of drugs and how they are shaping standard of care with the potential to make multiple myeloma a chronic disease.
Click on the PDF icon at the top of this introduction to read the full article.
Childhood cancer risk linked to mother’s birthplace

Photo by Nina Matthews
New research suggests a mother’s birthplace may affect the risk of certain cancers for Hispanic children.
The study showed that children of Hispanic mothers who were not born in the US had lower risks of brain cancers, neuroblastoma, and Wilms tumor, when compared to children of US-born Hispanic mothers and non-Hispanic white mothers born in the US.
However, all Hispanic children, regardless of where their mothers were born, had higher risks of acute leukemias and Hodgkin lymphoma but a lower risk of non-Hodgkin lymphoma (NHL).
Julia E. Heck, PhD, of the University of California, Los Angeles, and her colleagues reported these findings in JAMA Pediatrics.
The researchers used California birth records to identify children born from 1983 through 2011. Information on cancer cases came from California Cancer Registry records from 1988 to 2012.
The team restricted their analysis to children of US-born white, US-born Hispanic, and non-US-born Hispanic mothers. The study included 13,666 cases of children diagnosed with cancer before the age of 6 and 15,513,718 children who served as control subjects.
To assess the hazard ratios (HRs) for various cancers, the researchers used children of non-Hispanic white mothers as a reference (HR=1.00) and compared them to the children of non-US-born Hispanic mothers and US-born Hispanic mothers.
For children of non-US-born Hispanic mothers, the HR was 0.50 for glioma, 0.43 for astrocytoma, 0.47 for neuroblastoma, and 0.70 for Wilms tumor. For children of US-born Hispanic mothers, the HR was 0.71 for glioma, 0.62 for astrocytoma, 0.66 for neuroblastoma, and 0.88 for Wilms tumor.
When compared to non-Hispanic white children, Hispanic children had an increased risk of acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), and Hodgkin lymphoma but lower risks of NHL and Burkitt lymphoma.
For children of US-born Hispanic mothers, the HR was 1.20 for ALL, 1.28 for AML, 2.49 for Hodgkin lymphoma, 0.79 for NHL, and 0.69 for Burkitt lymphoma.
For children of non-US-born Hispanic mothers, the HR was 1.06 for ALL, 1.05 for AML, 2.35 for Hodgkin lymphoma, 0.76 for NHL, and 0.73 for Burkitt lymphoma.
The researchers said the differences observed between children of US-born and non-US-born Hispanic mothers may be explained by lifestyle differences and varying environmental exposures.
These factors may explain the differences in cancer incidence between Hispanic children and white children as well, but the differences may also be a result of genetic variation and infection exposures early in life. ![]()

Photo by Nina Matthews
New research suggests a mother’s birthplace may affect the risk of certain cancers for Hispanic children.
The study showed that children of Hispanic mothers who were not born in the US had lower risks of brain cancers, neuroblastoma, and Wilms tumor, when compared to children of US-born Hispanic mothers and non-Hispanic white mothers born in the US.
However, all Hispanic children, regardless of where their mothers were born, had higher risks of acute leukemias and Hodgkin lymphoma but a lower risk of non-Hodgkin lymphoma (NHL).
Julia E. Heck, PhD, of the University of California, Los Angeles, and her colleagues reported these findings in JAMA Pediatrics.
The researchers used California birth records to identify children born from 1983 through 2011. Information on cancer cases came from California Cancer Registry records from 1988 to 2012.
The team restricted their analysis to children of US-born white, US-born Hispanic, and non-US-born Hispanic mothers. The study included 13,666 cases of children diagnosed with cancer before the age of 6 and 15,513,718 children who served as control subjects.
To assess the hazard ratios (HRs) for various cancers, the researchers used children of non-Hispanic white mothers as a reference (HR=1.00) and compared them to the children of non-US-born Hispanic mothers and US-born Hispanic mothers.
For children of non-US-born Hispanic mothers, the HR was 0.50 for glioma, 0.43 for astrocytoma, 0.47 for neuroblastoma, and 0.70 for Wilms tumor. For children of US-born Hispanic mothers, the HR was 0.71 for glioma, 0.62 for astrocytoma, 0.66 for neuroblastoma, and 0.88 for Wilms tumor.
When compared to non-Hispanic white children, Hispanic children had an increased risk of acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), and Hodgkin lymphoma but lower risks of NHL and Burkitt lymphoma.
For children of US-born Hispanic mothers, the HR was 1.20 for ALL, 1.28 for AML, 2.49 for Hodgkin lymphoma, 0.79 for NHL, and 0.69 for Burkitt lymphoma.
For children of non-US-born Hispanic mothers, the HR was 1.06 for ALL, 1.05 for AML, 2.35 for Hodgkin lymphoma, 0.76 for NHL, and 0.73 for Burkitt lymphoma.
The researchers said the differences observed between children of US-born and non-US-born Hispanic mothers may be explained by lifestyle differences and varying environmental exposures.
These factors may explain the differences in cancer incidence between Hispanic children and white children as well, but the differences may also be a result of genetic variation and infection exposures early in life. ![]()

Photo by Nina Matthews
New research suggests a mother’s birthplace may affect the risk of certain cancers for Hispanic children.
The study showed that children of Hispanic mothers who were not born in the US had lower risks of brain cancers, neuroblastoma, and Wilms tumor, when compared to children of US-born Hispanic mothers and non-Hispanic white mothers born in the US.
However, all Hispanic children, regardless of where their mothers were born, had higher risks of acute leukemias and Hodgkin lymphoma but a lower risk of non-Hodgkin lymphoma (NHL).
Julia E. Heck, PhD, of the University of California, Los Angeles, and her colleagues reported these findings in JAMA Pediatrics.
The researchers used California birth records to identify children born from 1983 through 2011. Information on cancer cases came from California Cancer Registry records from 1988 to 2012.
The team restricted their analysis to children of US-born white, US-born Hispanic, and non-US-born Hispanic mothers. The study included 13,666 cases of children diagnosed with cancer before the age of 6 and 15,513,718 children who served as control subjects.
To assess the hazard ratios (HRs) for various cancers, the researchers used children of non-Hispanic white mothers as a reference (HR=1.00) and compared them to the children of non-US-born Hispanic mothers and US-born Hispanic mothers.
For children of non-US-born Hispanic mothers, the HR was 0.50 for glioma, 0.43 for astrocytoma, 0.47 for neuroblastoma, and 0.70 for Wilms tumor. For children of US-born Hispanic mothers, the HR was 0.71 for glioma, 0.62 for astrocytoma, 0.66 for neuroblastoma, and 0.88 for Wilms tumor.
When compared to non-Hispanic white children, Hispanic children had an increased risk of acute lymphoblastic leukemia (ALL), acute myeloid leukemia (AML), and Hodgkin lymphoma but lower risks of NHL and Burkitt lymphoma.
For children of US-born Hispanic mothers, the HR was 1.20 for ALL, 1.28 for AML, 2.49 for Hodgkin lymphoma, 0.79 for NHL, and 0.69 for Burkitt lymphoma.
For children of non-US-born Hispanic mothers, the HR was 1.06 for ALL, 1.05 for AML, 2.35 for Hodgkin lymphoma, 0.76 for NHL, and 0.73 for Burkitt lymphoma.
The researchers said the differences observed between children of US-born and non-US-born Hispanic mothers may be explained by lifestyle differences and varying environmental exposures.
These factors may explain the differences in cancer incidence between Hispanic children and white children as well, but the differences may also be a result of genetic variation and infection exposures early in life. ![]()
Bortezomib-based regimen + transplant increased progression-free survival in primary plasma cell leukemia
In a prospective study of 40 patients with primary plasma cell leukemia, upfront autotransplantation followed by allotransplant for younger patients and by consolidation/maintenance for older patients was associated with a median overall survival of 36.3 months and a median progression-free survival of 15.1 months.
Patients with this aggressive form of multiple myeloma received a regimen that combined standard chemotherapy, a proteasome inhibitor, high-dose melphalan followed by autologous stem cell transplantation, and allogeneic transplantation or immunomodulatory drugs, reported Dr. Bruno Royer of University Hospital in Amiens, France, and his associates.
Induction therapy consisted of four 21-day cycles: Cycles 1 and 3 included subcutaneous bortezomib, intravenous pegylated doxorubicin, and oral dexamethasone; cycles 2 and 4 included subcutaneous bortezomib, oral cyclophosphamide, and oral dexamethasone. Of 39 patients – one patient died 24 hours after study inclusion – 35 completed the four cycles. The overall response rate to induction was 69%: 10% of patients had a complete response and 26% had a very good partial response. Of 27 responding patients, 25 underwent high-dose melphalan followed by autologous stem cell transplantation.
The high response rates allowed 16 patients who were younger than 66 years and had an HLA-matched donor to then receive high-dose melphalan followed by autologous stem cell transplantation followed by consolidation with either an reduced-intensity conditioning allograft or a second high-dose melphalan followed by autologous stem cell transplantation and subsequent maintenance with lenalidomide, bortezomib, and dexamethasone for 1 year, the researchers said (J Clin Oncol. 2016 Apr 25. doi: 10.1200/JCO.2015.63.1929).
A total of 20% of patients had a complete response to the entire treatment protocol, 13% had a stringent complete response, 26% had a very good partial response, 5% had stable disease, and 5% had progressive disease. Thirteen patients died of progressive disease and four died of infections, including three that occurred during induction or after allograft.
This is only the second prospective trial in patients with primary plasma cell leukemia, an aggressive form of multiple myeloma that accounts for 2%-4% of cases, the researchers said. Future prospective trials should seek to optimize induction with newer combinations, such as carfilzomib, lenalidomide, dexamethasone, or monoclonal anti-CD38 antibodies. Also, optimizing the stem cell conditioning procedure and the postallograft immunomodulation may further benefit younger patients.
Dr. Royer reported receiving honoraria from Amgen and having served as a consultant or advisor for Octapharma Plasma. Fifteen coinvestigators also reported financial relationships with a number of pharmaceutical companies.
The study by Dr. Royer and associates is the first prospective trial to confirm that bortezomib-based regimens combined with a transplantation program may be effective and feasible in a significant proportion of patients with primary plasma cell leukemia. Response to induction therapy, however, was not remarkable; thus, although both cyclophosphamide and doxorubicin have demonstrated efficacy in primary plasma cell leukemia, the introduction of lenalidomide and/or incorporation of newer agents such as pomalidomide, carfilzomib, or daratumumab could hopefully optimize the induction phase and increase the rate and quality of response in future studies.
Hopefully, sequential phases of induction therapy, multiple transplantations (if applicable), further consolidation, and maintenance should ensure rapid disease control and reduction of early deaths from initial complications, a contrasting of clonal evolution that may induce drug resistance, and activity on residual disease by decreasing the risk of relapse. Feasibility of these approaches, however, may be limited, especially for older and frail patients who are unable to tolerate intensive induction or prolonged treatments. Personalized therapies with acceptable toxicities should be considered for these patients.
Dr. Pellegrino Musto is at Referral Cancer Center of Basilicata, Rionero in Vulture, Italy. He reported receiving honoraria from Celgene, Janssen-Cilag, Novartis, Sanofi, and Bristol-Myers Squibb. These comments are from an editorial (J Clin Oncol. 2016 Apr 25. doi: 10.1200/JCO.2016.66.6115) that accompanied the published study.
The study by Dr. Royer and associates is the first prospective trial to confirm that bortezomib-based regimens combined with a transplantation program may be effective and feasible in a significant proportion of patients with primary plasma cell leukemia. Response to induction therapy, however, was not remarkable; thus, although both cyclophosphamide and doxorubicin have demonstrated efficacy in primary plasma cell leukemia, the introduction of lenalidomide and/or incorporation of newer agents such as pomalidomide, carfilzomib, or daratumumab could hopefully optimize the induction phase and increase the rate and quality of response in future studies.
Hopefully, sequential phases of induction therapy, multiple transplantations (if applicable), further consolidation, and maintenance should ensure rapid disease control and reduction of early deaths from initial complications, a contrasting of clonal evolution that may induce drug resistance, and activity on residual disease by decreasing the risk of relapse. Feasibility of these approaches, however, may be limited, especially for older and frail patients who are unable to tolerate intensive induction or prolonged treatments. Personalized therapies with acceptable toxicities should be considered for these patients.
Dr. Pellegrino Musto is at Referral Cancer Center of Basilicata, Rionero in Vulture, Italy. He reported receiving honoraria from Celgene, Janssen-Cilag, Novartis, Sanofi, and Bristol-Myers Squibb. These comments are from an editorial (J Clin Oncol. 2016 Apr 25. doi: 10.1200/JCO.2016.66.6115) that accompanied the published study.
The study by Dr. Royer and associates is the first prospective trial to confirm that bortezomib-based regimens combined with a transplantation program may be effective and feasible in a significant proportion of patients with primary plasma cell leukemia. Response to induction therapy, however, was not remarkable; thus, although both cyclophosphamide and doxorubicin have demonstrated efficacy in primary plasma cell leukemia, the introduction of lenalidomide and/or incorporation of newer agents such as pomalidomide, carfilzomib, or daratumumab could hopefully optimize the induction phase and increase the rate and quality of response in future studies.
Hopefully, sequential phases of induction therapy, multiple transplantations (if applicable), further consolidation, and maintenance should ensure rapid disease control and reduction of early deaths from initial complications, a contrasting of clonal evolution that may induce drug resistance, and activity on residual disease by decreasing the risk of relapse. Feasibility of these approaches, however, may be limited, especially for older and frail patients who are unable to tolerate intensive induction or prolonged treatments. Personalized therapies with acceptable toxicities should be considered for these patients.
Dr. Pellegrino Musto is at Referral Cancer Center of Basilicata, Rionero in Vulture, Italy. He reported receiving honoraria from Celgene, Janssen-Cilag, Novartis, Sanofi, and Bristol-Myers Squibb. These comments are from an editorial (J Clin Oncol. 2016 Apr 25. doi: 10.1200/JCO.2016.66.6115) that accompanied the published study.
In a prospective study of 40 patients with primary plasma cell leukemia, upfront autotransplantation followed by allotransplant for younger patients and by consolidation/maintenance for older patients was associated with a median overall survival of 36.3 months and a median progression-free survival of 15.1 months.
Patients with this aggressive form of multiple myeloma received a regimen that combined standard chemotherapy, a proteasome inhibitor, high-dose melphalan followed by autologous stem cell transplantation, and allogeneic transplantation or immunomodulatory drugs, reported Dr. Bruno Royer of University Hospital in Amiens, France, and his associates.
Induction therapy consisted of four 21-day cycles: Cycles 1 and 3 included subcutaneous bortezomib, intravenous pegylated doxorubicin, and oral dexamethasone; cycles 2 and 4 included subcutaneous bortezomib, oral cyclophosphamide, and oral dexamethasone. Of 39 patients – one patient died 24 hours after study inclusion – 35 completed the four cycles. The overall response rate to induction was 69%: 10% of patients had a complete response and 26% had a very good partial response. Of 27 responding patients, 25 underwent high-dose melphalan followed by autologous stem cell transplantation.
The high response rates allowed 16 patients who were younger than 66 years and had an HLA-matched donor to then receive high-dose melphalan followed by autologous stem cell transplantation followed by consolidation with either an reduced-intensity conditioning allograft or a second high-dose melphalan followed by autologous stem cell transplantation and subsequent maintenance with lenalidomide, bortezomib, and dexamethasone for 1 year, the researchers said (J Clin Oncol. 2016 Apr 25. doi: 10.1200/JCO.2015.63.1929).
A total of 20% of patients had a complete response to the entire treatment protocol, 13% had a stringent complete response, 26% had a very good partial response, 5% had stable disease, and 5% had progressive disease. Thirteen patients died of progressive disease and four died of infections, including three that occurred during induction or after allograft.
This is only the second prospective trial in patients with primary plasma cell leukemia, an aggressive form of multiple myeloma that accounts for 2%-4% of cases, the researchers said. Future prospective trials should seek to optimize induction with newer combinations, such as carfilzomib, lenalidomide, dexamethasone, or monoclonal anti-CD38 antibodies. Also, optimizing the stem cell conditioning procedure and the postallograft immunomodulation may further benefit younger patients.
Dr. Royer reported receiving honoraria from Amgen and having served as a consultant or advisor for Octapharma Plasma. Fifteen coinvestigators also reported financial relationships with a number of pharmaceutical companies.
In a prospective study of 40 patients with primary plasma cell leukemia, upfront autotransplantation followed by allotransplant for younger patients and by consolidation/maintenance for older patients was associated with a median overall survival of 36.3 months and a median progression-free survival of 15.1 months.
Patients with this aggressive form of multiple myeloma received a regimen that combined standard chemotherapy, a proteasome inhibitor, high-dose melphalan followed by autologous stem cell transplantation, and allogeneic transplantation or immunomodulatory drugs, reported Dr. Bruno Royer of University Hospital in Amiens, France, and his associates.
Induction therapy consisted of four 21-day cycles: Cycles 1 and 3 included subcutaneous bortezomib, intravenous pegylated doxorubicin, and oral dexamethasone; cycles 2 and 4 included subcutaneous bortezomib, oral cyclophosphamide, and oral dexamethasone. Of 39 patients – one patient died 24 hours after study inclusion – 35 completed the four cycles. The overall response rate to induction was 69%: 10% of patients had a complete response and 26% had a very good partial response. Of 27 responding patients, 25 underwent high-dose melphalan followed by autologous stem cell transplantation.
The high response rates allowed 16 patients who were younger than 66 years and had an HLA-matched donor to then receive high-dose melphalan followed by autologous stem cell transplantation followed by consolidation with either an reduced-intensity conditioning allograft or a second high-dose melphalan followed by autologous stem cell transplantation and subsequent maintenance with lenalidomide, bortezomib, and dexamethasone for 1 year, the researchers said (J Clin Oncol. 2016 Apr 25. doi: 10.1200/JCO.2015.63.1929).
A total of 20% of patients had a complete response to the entire treatment protocol, 13% had a stringent complete response, 26% had a very good partial response, 5% had stable disease, and 5% had progressive disease. Thirteen patients died of progressive disease and four died of infections, including three that occurred during induction or after allograft.
This is only the second prospective trial in patients with primary plasma cell leukemia, an aggressive form of multiple myeloma that accounts for 2%-4% of cases, the researchers said. Future prospective trials should seek to optimize induction with newer combinations, such as carfilzomib, lenalidomide, dexamethasone, or monoclonal anti-CD38 antibodies. Also, optimizing the stem cell conditioning procedure and the postallograft immunomodulation may further benefit younger patients.
Dr. Royer reported receiving honoraria from Amgen and having served as a consultant or advisor for Octapharma Plasma. Fifteen coinvestigators also reported financial relationships with a number of pharmaceutical companies.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Progression-free survival was improved in patients who had primary plasma cell leukemia and underwent four cycles of induction; high-dose melphalan followed by autologous stem cell transplantation; consolidation with either a reduced-intensity conditioning allograft or a second high-dose melphalan followed by autologous stem cell transplantation; and subsequent maintenance with lenalidomide, bortezomib, and dexamethasone for 1 year.
Major finding: Median overall survival was 36.3 months and median progression-free survival was 15.1 months.
Data source: A prospective phase II study of 40 adults with newly diagnosed primary plasma cell leukemia.
Disclosures: Dr. Royer reported receiving honoraria from Amgen and having served as a consultant or advisor for Octapharma Plasma. Fifteen coinvestigators also reported financial relationships with various drug companies.
NIH stops production at 2 facilities

Photo by Daniel Sone
The National Institutes of Health (NIH) has suspended production in 2 of its facilities—a National Cancer Institute (NCI) laboratory engaged in the production of cell therapies and a National Institute of Mental Health facility producing positron emission tomography (PET) materials.
Last year, an inspection by the US Food and Drug Administration revealed problems with facilities, equipment, procedures, and training in the NIH Clinical Center Pharmaceutical Development Section (PDS), which is responsible for managing investigational drugs.
So the NIH closed the sterile production unit of the PDS and hired 2 companies specializing in quality assurance for manufacturing and compounding—Working Buildings and Clinical IQ—to evaluate all NIH facilities producing sterile or infused products for administration to research participants.
This review is still underway, and preliminary findings have identified facilities not in compliance with quality and safety standards, and not suitable for the production of sterile or infused products.
As a result, the NIH suspended production in the aforementioned facilities manufacturing cell therapy and PET materials.
The NIH said there is no evidence that any patients have been harmed, but a rigorous clinical review will be conducted. And the NIH will not enroll new patients in affected trials until the issues are resolved.
The NCI facility produces cell therapies in cooperation with Kite Pharma, Inc. The company and the NCI are advancing multiple clinical trials under Cooperative Research and Development Agreements for the treatment of hematologic malignancies and solid tumors.
Patients currently enrolled in ongoing NCI trials of cell therapy will continue to receive treatment, but no new patients will be enrolled until the review is complete.
And Kite Pharma said its 4 trials of the chimeric antigen receptor T-cell therapy KTE-C19 will continue. This includes:
ZUMA-1—KTE-C19 in patients with refractory, aggressive non-Hodgkin lymphoma
ZUMA-2—KTE-C19 in patients with relapsed/refractory mantle cell lymphoma
ZUMA-3—KTE-C19 in adults with relapsed/refractory B-precursor acute lymphoblastic leukemia
ZUMA-4—KTE-C19 in pediatric and adolescent patients with relapsed/refractory B-precursor acute lymphoblastic leukemia.
The company stressed that the review of the NCI’s manufacturing facilities is not related to KTE-C19 or Kite’s manufacturing capabilities. ![]()

Photo by Daniel Sone
The National Institutes of Health (NIH) has suspended production in 2 of its facilities—a National Cancer Institute (NCI) laboratory engaged in the production of cell therapies and a National Institute of Mental Health facility producing positron emission tomography (PET) materials.
Last year, an inspection by the US Food and Drug Administration revealed problems with facilities, equipment, procedures, and training in the NIH Clinical Center Pharmaceutical Development Section (PDS), which is responsible for managing investigational drugs.
So the NIH closed the sterile production unit of the PDS and hired 2 companies specializing in quality assurance for manufacturing and compounding—Working Buildings and Clinical IQ—to evaluate all NIH facilities producing sterile or infused products for administration to research participants.
This review is still underway, and preliminary findings have identified facilities not in compliance with quality and safety standards, and not suitable for the production of sterile or infused products.
As a result, the NIH suspended production in the aforementioned facilities manufacturing cell therapy and PET materials.
The NIH said there is no evidence that any patients have been harmed, but a rigorous clinical review will be conducted. And the NIH will not enroll new patients in affected trials until the issues are resolved.
The NCI facility produces cell therapies in cooperation with Kite Pharma, Inc. The company and the NCI are advancing multiple clinical trials under Cooperative Research and Development Agreements for the treatment of hematologic malignancies and solid tumors.
Patients currently enrolled in ongoing NCI trials of cell therapy will continue to receive treatment, but no new patients will be enrolled until the review is complete.
And Kite Pharma said its 4 trials of the chimeric antigen receptor T-cell therapy KTE-C19 will continue. This includes:
ZUMA-1—KTE-C19 in patients with refractory, aggressive non-Hodgkin lymphoma
ZUMA-2—KTE-C19 in patients with relapsed/refractory mantle cell lymphoma
ZUMA-3—KTE-C19 in adults with relapsed/refractory B-precursor acute lymphoblastic leukemia
ZUMA-4—KTE-C19 in pediatric and adolescent patients with relapsed/refractory B-precursor acute lymphoblastic leukemia.
The company stressed that the review of the NCI’s manufacturing facilities is not related to KTE-C19 or Kite’s manufacturing capabilities. ![]()

Photo by Daniel Sone
The National Institutes of Health (NIH) has suspended production in 2 of its facilities—a National Cancer Institute (NCI) laboratory engaged in the production of cell therapies and a National Institute of Mental Health facility producing positron emission tomography (PET) materials.
Last year, an inspection by the US Food and Drug Administration revealed problems with facilities, equipment, procedures, and training in the NIH Clinical Center Pharmaceutical Development Section (PDS), which is responsible for managing investigational drugs.
So the NIH closed the sterile production unit of the PDS and hired 2 companies specializing in quality assurance for manufacturing and compounding—Working Buildings and Clinical IQ—to evaluate all NIH facilities producing sterile or infused products for administration to research participants.
This review is still underway, and preliminary findings have identified facilities not in compliance with quality and safety standards, and not suitable for the production of sterile or infused products.
As a result, the NIH suspended production in the aforementioned facilities manufacturing cell therapy and PET materials.
The NIH said there is no evidence that any patients have been harmed, but a rigorous clinical review will be conducted. And the NIH will not enroll new patients in affected trials until the issues are resolved.
The NCI facility produces cell therapies in cooperation with Kite Pharma, Inc. The company and the NCI are advancing multiple clinical trials under Cooperative Research and Development Agreements for the treatment of hematologic malignancies and solid tumors.
Patients currently enrolled in ongoing NCI trials of cell therapy will continue to receive treatment, but no new patients will be enrolled until the review is complete.
And Kite Pharma said its 4 trials of the chimeric antigen receptor T-cell therapy KTE-C19 will continue. This includes:
ZUMA-1—KTE-C19 in patients with refractory, aggressive non-Hodgkin lymphoma
ZUMA-2—KTE-C19 in patients with relapsed/refractory mantle cell lymphoma
ZUMA-3—KTE-C19 in adults with relapsed/refractory B-precursor acute lymphoblastic leukemia
ZUMA-4—KTE-C19 in pediatric and adolescent patients with relapsed/refractory B-precursor acute lymphoblastic leukemia.
The company stressed that the review of the NCI’s manufacturing facilities is not related to KTE-C19 or Kite’s manufacturing capabilities. ![]()