User login
Metabolic tumor volume predicts outcome in follicular lymphoma
The total metabolic tumor volume, as quantified on PET scanning at the time that follicular lymphoma is diagnosed, is a strong independent predictor of treatment response and patient outcome, according to a report published online in Journal of Clinical Oncology.
Until now, no study has specifically examined the prognostic possibilities of PET-derived total metabolic tumor volume (TMTV) for this malignancy, either on its own or in combination with any of several existing prognostic indices. Those tools use a variety of surrogates to estimate tumor burden. Now that PET is recommended at diagnosis for all cases of follicular lymphoma and anatomic CT data are also available, it is much easier to estimate total tumor burden than it was when those indices were developed, said Michel Meignan, MD, PhD, of Hôpital Henri Mondor, Crétiel (France) and his associates.
It is crucial to identify patients likely to have a poor response to standard treatment, both to spare them the considerable adverse effects of that treatment and to select them for alternative first-line approaches. Even though patient survival has improved markedly during the past decade with the introduction of combined treatment using rituximab plus chemotherapy, approximately 20% of patients still show disease progression within 2 years, and the 5-year overall survival is only 50%, the investigators noted.
To assess the prognostic value of TMTV as assessed by PET, they pooled data from three multicenter prospective studies involving 185 patients with either a high tumor burden or advanced-stage follicular lymphoma. These participants were followed for a median of 63.5 months at 56 medical centers in France, Belgium, Australia, and Italy.
A TMTV threshold of 510 cm3 was found to have the optimal sensitivity (0.46), specificity (0.83), positive predictive value (0.67), and negative predictive value (0.67) for predicting both progression-free and overall survival. The 30% of patients who had a TMTV greater than that cutoff point had markedly inferior 5-year progression-free survival (less than 3 years), while the 70% who had a smaller TMTV had median progression-free survival of more than 6 years, Dr. Meignan and his associates said (J Clin Oncol. 2016 Aug 22. doi:10.1200/JCO.2016.66.9440).
Combining TMTV with other prognostic measures improved predictions even further. Patients who had both a high TMTV and an intermediate to high score on the Follicular Lymphoma International Prognostic Index 2 showed extremely poor outcomes, with a median progression-free survival of only 19 months. “This population can no longer be characterized as having an indolent lymphoma,” the investigators said.
No sponsor or funding source was cited for this study. Dr. Meignan reported receiving fees for travel and expenses from Roche; his associates reported ties to numerous industry sources.
The total metabolic tumor volume, as quantified on PET scanning at the time that follicular lymphoma is diagnosed, is a strong independent predictor of treatment response and patient outcome, according to a report published online in Journal of Clinical Oncology.
Until now, no study has specifically examined the prognostic possibilities of PET-derived total metabolic tumor volume (TMTV) for this malignancy, either on its own or in combination with any of several existing prognostic indices. Those tools use a variety of surrogates to estimate tumor burden. Now that PET is recommended at diagnosis for all cases of follicular lymphoma and anatomic CT data are also available, it is much easier to estimate total tumor burden than it was when those indices were developed, said Michel Meignan, MD, PhD, of Hôpital Henri Mondor, Crétiel (France) and his associates.
It is crucial to identify patients likely to have a poor response to standard treatment, both to spare them the considerable adverse effects of that treatment and to select them for alternative first-line approaches. Even though patient survival has improved markedly during the past decade with the introduction of combined treatment using rituximab plus chemotherapy, approximately 20% of patients still show disease progression within 2 years, and the 5-year overall survival is only 50%, the investigators noted.
To assess the prognostic value of TMTV as assessed by PET, they pooled data from three multicenter prospective studies involving 185 patients with either a high tumor burden or advanced-stage follicular lymphoma. These participants were followed for a median of 63.5 months at 56 medical centers in France, Belgium, Australia, and Italy.
A TMTV threshold of 510 cm3 was found to have the optimal sensitivity (0.46), specificity (0.83), positive predictive value (0.67), and negative predictive value (0.67) for predicting both progression-free and overall survival. The 30% of patients who had a TMTV greater than that cutoff point had markedly inferior 5-year progression-free survival (less than 3 years), while the 70% who had a smaller TMTV had median progression-free survival of more than 6 years, Dr. Meignan and his associates said (J Clin Oncol. 2016 Aug 22. doi:10.1200/JCO.2016.66.9440).
Combining TMTV with other prognostic measures improved predictions even further. Patients who had both a high TMTV and an intermediate to high score on the Follicular Lymphoma International Prognostic Index 2 showed extremely poor outcomes, with a median progression-free survival of only 19 months. “This population can no longer be characterized as having an indolent lymphoma,” the investigators said.
No sponsor or funding source was cited for this study. Dr. Meignan reported receiving fees for travel and expenses from Roche; his associates reported ties to numerous industry sources.
The total metabolic tumor volume, as quantified on PET scanning at the time that follicular lymphoma is diagnosed, is a strong independent predictor of treatment response and patient outcome, according to a report published online in Journal of Clinical Oncology.
Until now, no study has specifically examined the prognostic possibilities of PET-derived total metabolic tumor volume (TMTV) for this malignancy, either on its own or in combination with any of several existing prognostic indices. Those tools use a variety of surrogates to estimate tumor burden. Now that PET is recommended at diagnosis for all cases of follicular lymphoma and anatomic CT data are also available, it is much easier to estimate total tumor burden than it was when those indices were developed, said Michel Meignan, MD, PhD, of Hôpital Henri Mondor, Crétiel (France) and his associates.
It is crucial to identify patients likely to have a poor response to standard treatment, both to spare them the considerable adverse effects of that treatment and to select them for alternative first-line approaches. Even though patient survival has improved markedly during the past decade with the introduction of combined treatment using rituximab plus chemotherapy, approximately 20% of patients still show disease progression within 2 years, and the 5-year overall survival is only 50%, the investigators noted.
To assess the prognostic value of TMTV as assessed by PET, they pooled data from three multicenter prospective studies involving 185 patients with either a high tumor burden or advanced-stage follicular lymphoma. These participants were followed for a median of 63.5 months at 56 medical centers in France, Belgium, Australia, and Italy.
A TMTV threshold of 510 cm3 was found to have the optimal sensitivity (0.46), specificity (0.83), positive predictive value (0.67), and negative predictive value (0.67) for predicting both progression-free and overall survival. The 30% of patients who had a TMTV greater than that cutoff point had markedly inferior 5-year progression-free survival (less than 3 years), while the 70% who had a smaller TMTV had median progression-free survival of more than 6 years, Dr. Meignan and his associates said (J Clin Oncol. 2016 Aug 22. doi:10.1200/JCO.2016.66.9440).
Combining TMTV with other prognostic measures improved predictions even further. Patients who had both a high TMTV and an intermediate to high score on the Follicular Lymphoma International Prognostic Index 2 showed extremely poor outcomes, with a median progression-free survival of only 19 months. “This population can no longer be characterized as having an indolent lymphoma,” the investigators said.
No sponsor or funding source was cited for this study. Dr. Meignan reported receiving fees for travel and expenses from Roche; his associates reported ties to numerous industry sources.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: At diagnosis, the total metabolic tumor volume of follicular lymphoma predicts treatment response and patient outcome.
Major finding: A TMTV threshold of 510 cm3 was found to have the optimal sensitivity (0.46), specificity (0.83), positive predictive value (0.67), and negative predictive value (0.67) for predicting both progression-free and overall survival.
Data source: A pooled analysis of three multicenter prospective studies involving 185 patients with a high burden of disease.
Disclosures: No sponsor or funding source was cited for this study. Dr. Meignan reported receiving fees for travel and expenses from Roche; his associates reported ties to numerous industry sources.
New treatment option for relapsed/refractory NHL

Results of the phase 3 GADOLIN trial have revealed a new treatment option for patients with relapsed/refractory non-Hodgkin lymphoma (NHL), according to researchers.
The trial showed that obinutuzumab plus bendamustine, followed by obinutuzumab maintenance, can ward off disease progression in NHL patients who have relapsed after rituximab-based
therapy or stopped responding to it.
The obinutuzumab regimen significantly improved progression-free survival (PFS) when compared to bendamustine alone (without maintenance).
However, there was no significant difference between the 2 treatment arms with regard to overall survival (OS).
Still, the trial was stopped before its protocol-specified final analysis because of the PFS benefit in the obinutuzumab arm.
Laurie Sehn, MD, of the BC Cancer Agency in Vancouver, British Columbia, Canada, and her colleagues reported the results of this trial in The Lancet. The research was funded by F. Hoffmann-La Roche Ltd.
Patients and treatment
GADOLIN enrolled patients with relapsed/refractory follicular lymphoma (FL), marginal zone lymphoma (MZL), small lymphocytic lymphoma (SLL), and Waldenstrom’s macroglobulinemia (WM).
The patients were randomized to receive one of the following treatments:
- bendamustine alone (120 mg/m2/day on days 1 and 2 for up to six 28-day cycles)
- bendamustine (90 mg/m2/day on days 1 and 2 for up to six 28-day cycles) plus obinutuzumab (1000 mg on days 1, 8, and 15 for cycle 1, followed by 1 dose for up to six 28-day cycles), followed by obinutuzumab maintenance (1000 mg every 2 months for 2 years or until progression).
The investigators said baseline characteristics were well-balanced between the treatment arms. Patients in both arms had received a median of 2 prior treatments, and the median time from last treatment was about 4 months.
Of the 194 patients randomized to the obinutuzumab arm, 155 had FL, 27 had MZL, and 12 had SLL. Of the 202 patients randomized to the bendamustine (control) arm, 166 had FL, 19 had MZL, 16 had SLL, and 1 had WM.
Ultimately, 156 patients completed induction in the obinutuzumab arm, as did 129 patients in the control arm. Thirty-six patients completed maintenance with obinutuzumab, and 46 were still receiving maintenance at the time of analysis.
The median follow-up was 21.9 months in the obinutuzumab arm and 20.3 months in the control arm.
Safety
Nearly all patients in both arms experienced at least 1 adverse event (AE).
Grade 3-5 AEs occurred in 68% of patients in the obinutuzumab arm and 62% in the control arm. The most frequent of these were neutropenia (33% vs 26%), thrombocytopenia (11% vs 16%), anemia (8% vs 10%), and infusion-related reactions (11% vs 6%).
Serious AEs occurred in 38% of patients in the obinutuzumab arm and 33% in the control arm. The most common were febrile neutropenia (4% vs 3%), infusion-related reactions (4% vs 2%), and pneumonia (3% vs 5%).
Response
According to an independent review committee, the overall response rate at the end of induction was 69% in the obinutuzumab arm and 63% in the control arm. The complete response rates were 11% and 12%, respectively.
The median duration of response was not reached in the obinutuzumab arm and was 13.2 months in the control arm.
Survival
The median PFS was not reached in the obinutuzumab arm and was 14.9 months in the control arm (P=0.0001), according to the independent review committee.
According to investigators, the median PFS was 29.2 months and 14 months, respectively (P<0.0001).
At last follow-up, the median OS had not been reached in either arm (P=0.40).
There were 34 deaths in the obinutuzumab arm and 41 in the control arm (18% and 20%, respectively). Most patients died of disease progression (65% and 71%, respectively).
Twelve patients in each arm (6%) died of AEs. Three of these deaths were treatment-related in the obinutuzumab arm (acute myeloid leukemia, vascular pseudoaneurysm, and pseudomonal sepsis).
Five of the 12 AE deaths in the control arm were treatment-related (sepsis, 2 cases of leukemia, and 2 cases of Pneumocystis jirovecii pneumonia).
Taking these results together, the investigators said the obinutuzumab regimen had a manageable toxicity profile, and it produced a “clinically meaningful and significant” improvement in PFS when compared to bendamustine alone. ![]()

Results of the phase 3 GADOLIN trial have revealed a new treatment option for patients with relapsed/refractory non-Hodgkin lymphoma (NHL), according to researchers.
The trial showed that obinutuzumab plus bendamustine, followed by obinutuzumab maintenance, can ward off disease progression in NHL patients who have relapsed after rituximab-based
therapy or stopped responding to it.
The obinutuzumab regimen significantly improved progression-free survival (PFS) when compared to bendamustine alone (without maintenance).
However, there was no significant difference between the 2 treatment arms with regard to overall survival (OS).
Still, the trial was stopped before its protocol-specified final analysis because of the PFS benefit in the obinutuzumab arm.
Laurie Sehn, MD, of the BC Cancer Agency in Vancouver, British Columbia, Canada, and her colleagues reported the results of this trial in The Lancet. The research was funded by F. Hoffmann-La Roche Ltd.
Patients and treatment
GADOLIN enrolled patients with relapsed/refractory follicular lymphoma (FL), marginal zone lymphoma (MZL), small lymphocytic lymphoma (SLL), and Waldenstrom’s macroglobulinemia (WM).
The patients were randomized to receive one of the following treatments:
- bendamustine alone (120 mg/m2/day on days 1 and 2 for up to six 28-day cycles)
- bendamustine (90 mg/m2/day on days 1 and 2 for up to six 28-day cycles) plus obinutuzumab (1000 mg on days 1, 8, and 15 for cycle 1, followed by 1 dose for up to six 28-day cycles), followed by obinutuzumab maintenance (1000 mg every 2 months for 2 years or until progression).
The investigators said baseline characteristics were well-balanced between the treatment arms. Patients in both arms had received a median of 2 prior treatments, and the median time from last treatment was about 4 months.
Of the 194 patients randomized to the obinutuzumab arm, 155 had FL, 27 had MZL, and 12 had SLL. Of the 202 patients randomized to the bendamustine (control) arm, 166 had FL, 19 had MZL, 16 had SLL, and 1 had WM.
Ultimately, 156 patients completed induction in the obinutuzumab arm, as did 129 patients in the control arm. Thirty-six patients completed maintenance with obinutuzumab, and 46 were still receiving maintenance at the time of analysis.
The median follow-up was 21.9 months in the obinutuzumab arm and 20.3 months in the control arm.
Safety
Nearly all patients in both arms experienced at least 1 adverse event (AE).
Grade 3-5 AEs occurred in 68% of patients in the obinutuzumab arm and 62% in the control arm. The most frequent of these were neutropenia (33% vs 26%), thrombocytopenia (11% vs 16%), anemia (8% vs 10%), and infusion-related reactions (11% vs 6%).
Serious AEs occurred in 38% of patients in the obinutuzumab arm and 33% in the control arm. The most common were febrile neutropenia (4% vs 3%), infusion-related reactions (4% vs 2%), and pneumonia (3% vs 5%).
Response
According to an independent review committee, the overall response rate at the end of induction was 69% in the obinutuzumab arm and 63% in the control arm. The complete response rates were 11% and 12%, respectively.
The median duration of response was not reached in the obinutuzumab arm and was 13.2 months in the control arm.
Survival
The median PFS was not reached in the obinutuzumab arm and was 14.9 months in the control arm (P=0.0001), according to the independent review committee.
According to investigators, the median PFS was 29.2 months and 14 months, respectively (P<0.0001).
At last follow-up, the median OS had not been reached in either arm (P=0.40).
There were 34 deaths in the obinutuzumab arm and 41 in the control arm (18% and 20%, respectively). Most patients died of disease progression (65% and 71%, respectively).
Twelve patients in each arm (6%) died of AEs. Three of these deaths were treatment-related in the obinutuzumab arm (acute myeloid leukemia, vascular pseudoaneurysm, and pseudomonal sepsis).
Five of the 12 AE deaths in the control arm were treatment-related (sepsis, 2 cases of leukemia, and 2 cases of Pneumocystis jirovecii pneumonia).
Taking these results together, the investigators said the obinutuzumab regimen had a manageable toxicity profile, and it produced a “clinically meaningful and significant” improvement in PFS when compared to bendamustine alone. ![]()

Results of the phase 3 GADOLIN trial have revealed a new treatment option for patients with relapsed/refractory non-Hodgkin lymphoma (NHL), according to researchers.
The trial showed that obinutuzumab plus bendamustine, followed by obinutuzumab maintenance, can ward off disease progression in NHL patients who have relapsed after rituximab-based
therapy or stopped responding to it.
The obinutuzumab regimen significantly improved progression-free survival (PFS) when compared to bendamustine alone (without maintenance).
However, there was no significant difference between the 2 treatment arms with regard to overall survival (OS).
Still, the trial was stopped before its protocol-specified final analysis because of the PFS benefit in the obinutuzumab arm.
Laurie Sehn, MD, of the BC Cancer Agency in Vancouver, British Columbia, Canada, and her colleagues reported the results of this trial in The Lancet. The research was funded by F. Hoffmann-La Roche Ltd.
Patients and treatment
GADOLIN enrolled patients with relapsed/refractory follicular lymphoma (FL), marginal zone lymphoma (MZL), small lymphocytic lymphoma (SLL), and Waldenstrom’s macroglobulinemia (WM).
The patients were randomized to receive one of the following treatments:
- bendamustine alone (120 mg/m2/day on days 1 and 2 for up to six 28-day cycles)
- bendamustine (90 mg/m2/day on days 1 and 2 for up to six 28-day cycles) plus obinutuzumab (1000 mg on days 1, 8, and 15 for cycle 1, followed by 1 dose for up to six 28-day cycles), followed by obinutuzumab maintenance (1000 mg every 2 months for 2 years or until progression).
The investigators said baseline characteristics were well-balanced between the treatment arms. Patients in both arms had received a median of 2 prior treatments, and the median time from last treatment was about 4 months.
Of the 194 patients randomized to the obinutuzumab arm, 155 had FL, 27 had MZL, and 12 had SLL. Of the 202 patients randomized to the bendamustine (control) arm, 166 had FL, 19 had MZL, 16 had SLL, and 1 had WM.
Ultimately, 156 patients completed induction in the obinutuzumab arm, as did 129 patients in the control arm. Thirty-six patients completed maintenance with obinutuzumab, and 46 were still receiving maintenance at the time of analysis.
The median follow-up was 21.9 months in the obinutuzumab arm and 20.3 months in the control arm.
Safety
Nearly all patients in both arms experienced at least 1 adverse event (AE).
Grade 3-5 AEs occurred in 68% of patients in the obinutuzumab arm and 62% in the control arm. The most frequent of these were neutropenia (33% vs 26%), thrombocytopenia (11% vs 16%), anemia (8% vs 10%), and infusion-related reactions (11% vs 6%).
Serious AEs occurred in 38% of patients in the obinutuzumab arm and 33% in the control arm. The most common were febrile neutropenia (4% vs 3%), infusion-related reactions (4% vs 2%), and pneumonia (3% vs 5%).
Response
According to an independent review committee, the overall response rate at the end of induction was 69% in the obinutuzumab arm and 63% in the control arm. The complete response rates were 11% and 12%, respectively.
The median duration of response was not reached in the obinutuzumab arm and was 13.2 months in the control arm.
Survival
The median PFS was not reached in the obinutuzumab arm and was 14.9 months in the control arm (P=0.0001), according to the independent review committee.
According to investigators, the median PFS was 29.2 months and 14 months, respectively (P<0.0001).
At last follow-up, the median OS had not been reached in either arm (P=0.40).
There were 34 deaths in the obinutuzumab arm and 41 in the control arm (18% and 20%, respectively). Most patients died of disease progression (65% and 71%, respectively).
Twelve patients in each arm (6%) died of AEs. Three of these deaths were treatment-related in the obinutuzumab arm (acute myeloid leukemia, vascular pseudoaneurysm, and pseudomonal sepsis).
Five of the 12 AE deaths in the control arm were treatment-related (sepsis, 2 cases of leukemia, and 2 cases of Pneumocystis jirovecii pneumonia).
Taking these results together, the investigators said the obinutuzumab regimen had a manageable toxicity profile, and it produced a “clinically meaningful and significant” improvement in PFS when compared to bendamustine alone. ![]()
Predicting drug’s efficacy in relapsed DLBCL

The histone deacetylase inhibitor panobinostat can produce durable responses in certain patients with relapsed diffuse large B-cell lymphoma (DLBCL), according to a phase 2 trial.
Though less than 30% of the patients in this trial responded to treatment, the median duration of response was 14.5 months, and the longest ongoing response has lasted more than 3.5 years.
In addition, researchers found the presence of mutations in MEF2B and levels of circulating tumor DNA (ctDNA) could be used to predict response.
Sarit Assouline, MD, of Jewish General Hospital in Montreal, Quebec, Canada, and her colleagues reported these findings in Blood.
“DLBCL is one of the more common forms of lymphoma and is highly aggressive,” Dr Assouline noted. “Following relapse, there are no effective standards of treatment, and life expectancy averages 6 months.”
“Our challenge is to identify new biomarkers and target specific mutations in order to improve the prognosis. While many clinical trials simply report on how patients respond to a therapy, we devised this study to reveal the mechanisms on which the therapy works in order to understand which patients would benefit.”
The trial enrolled 40 patients with relapsed or refractory de novo (n=27) or transformed (n=13) DLBCL. The patients’ median age was 59.8 (range, 28.9-78.6). They received a median of 3 prior therapies (range, 1-9), and 75% of patients (n=30) were refractory to their most recent therapy.
The patients received panobinostat at 30 mg three times a week, with rituximab (n=19) or without (n=21).
Adverse events
The most common grade 3 or higher adverse events (AEs) that were thought to be related to panobinostat (with and without rituximab, respectively) were thrombocytopenia (68% and 71%) and neutropenia (11% and 24%).
Twenty-three patients (58%) required dose reductions, and 22 (55%) required dose interruptions. All of these were due to AEs, and there was no difference between patients who received rituximab and those who did not.
All 19 serious AEs were a result of disease progression.
Response
Twenty-eight percent of the patients (11/40) responded to treatment, and receiving rituximab did not increase the likelihood of response. Six patients (29%) responded to panobinostat alone, and 5 (26%) responded to panobinostat and rituximab.
There were 7 complete responses—5 of them among patients who received panobinostat alone. And there were 4 partial responses—3 among the patients who received panobinostat plus rituximab.
The median duration of response was 14.5 months overall, 9.4 months among patients who received panobinostat plus rituximab, and it was not reached among patients who received panobinostat alone.
At the data cutoff, 6 of the 11 responders had not progressed. The longest response had lasted more than 43 months and was ongoing at the cutoff point.
Predicting response
The researchers performed genome and exome sequencing in relapsed tumor biopsies and quantified ctDNA in serial plasma samples.
They found that mutations in MEF2B were significantly associated with response to panobinostat. The likelihood ratio for response was 3.67 for patients with MEF2B mutations.
On the other hand, increased levels of ctDNA were strongly associated with treatment failure. A significant increase in ctDNA at day 15 after treatment initiation could predict a lack of response with a sensitivity of 71.4% and a specificity of 100%.
“This trial has generated considerable data regarding methodology for processing samples from a clinical study, the genetic mutations associated with DLBCL and how they evolve over time, on ctDNA, and mechanisms of resistance to histone deacetylase inhibitors,” Dr Assouline concluded.
Novartis provided panobinostat and financial support for this trial, and Hoffman-LaRoche supplied rituximab. ![]()

The histone deacetylase inhibitor panobinostat can produce durable responses in certain patients with relapsed diffuse large B-cell lymphoma (DLBCL), according to a phase 2 trial.
Though less than 30% of the patients in this trial responded to treatment, the median duration of response was 14.5 months, and the longest ongoing response has lasted more than 3.5 years.
In addition, researchers found the presence of mutations in MEF2B and levels of circulating tumor DNA (ctDNA) could be used to predict response.
Sarit Assouline, MD, of Jewish General Hospital in Montreal, Quebec, Canada, and her colleagues reported these findings in Blood.
“DLBCL is one of the more common forms of lymphoma and is highly aggressive,” Dr Assouline noted. “Following relapse, there are no effective standards of treatment, and life expectancy averages 6 months.”
“Our challenge is to identify new biomarkers and target specific mutations in order to improve the prognosis. While many clinical trials simply report on how patients respond to a therapy, we devised this study to reveal the mechanisms on which the therapy works in order to understand which patients would benefit.”
The trial enrolled 40 patients with relapsed or refractory de novo (n=27) or transformed (n=13) DLBCL. The patients’ median age was 59.8 (range, 28.9-78.6). They received a median of 3 prior therapies (range, 1-9), and 75% of patients (n=30) were refractory to their most recent therapy.
The patients received panobinostat at 30 mg three times a week, with rituximab (n=19) or without (n=21).
Adverse events
The most common grade 3 or higher adverse events (AEs) that were thought to be related to panobinostat (with and without rituximab, respectively) were thrombocytopenia (68% and 71%) and neutropenia (11% and 24%).
Twenty-three patients (58%) required dose reductions, and 22 (55%) required dose interruptions. All of these were due to AEs, and there was no difference between patients who received rituximab and those who did not.
All 19 serious AEs were a result of disease progression.
Response
Twenty-eight percent of the patients (11/40) responded to treatment, and receiving rituximab did not increase the likelihood of response. Six patients (29%) responded to panobinostat alone, and 5 (26%) responded to panobinostat and rituximab.
There were 7 complete responses—5 of them among patients who received panobinostat alone. And there were 4 partial responses—3 among the patients who received panobinostat plus rituximab.
The median duration of response was 14.5 months overall, 9.4 months among patients who received panobinostat plus rituximab, and it was not reached among patients who received panobinostat alone.
At the data cutoff, 6 of the 11 responders had not progressed. The longest response had lasted more than 43 months and was ongoing at the cutoff point.
Predicting response
The researchers performed genome and exome sequencing in relapsed tumor biopsies and quantified ctDNA in serial plasma samples.
They found that mutations in MEF2B were significantly associated with response to panobinostat. The likelihood ratio for response was 3.67 for patients with MEF2B mutations.
On the other hand, increased levels of ctDNA were strongly associated with treatment failure. A significant increase in ctDNA at day 15 after treatment initiation could predict a lack of response with a sensitivity of 71.4% and a specificity of 100%.
“This trial has generated considerable data regarding methodology for processing samples from a clinical study, the genetic mutations associated with DLBCL and how they evolve over time, on ctDNA, and mechanisms of resistance to histone deacetylase inhibitors,” Dr Assouline concluded.
Novartis provided panobinostat and financial support for this trial, and Hoffman-LaRoche supplied rituximab. ![]()

The histone deacetylase inhibitor panobinostat can produce durable responses in certain patients with relapsed diffuse large B-cell lymphoma (DLBCL), according to a phase 2 trial.
Though less than 30% of the patients in this trial responded to treatment, the median duration of response was 14.5 months, and the longest ongoing response has lasted more than 3.5 years.
In addition, researchers found the presence of mutations in MEF2B and levels of circulating tumor DNA (ctDNA) could be used to predict response.
Sarit Assouline, MD, of Jewish General Hospital in Montreal, Quebec, Canada, and her colleagues reported these findings in Blood.
“DLBCL is one of the more common forms of lymphoma and is highly aggressive,” Dr Assouline noted. “Following relapse, there are no effective standards of treatment, and life expectancy averages 6 months.”
“Our challenge is to identify new biomarkers and target specific mutations in order to improve the prognosis. While many clinical trials simply report on how patients respond to a therapy, we devised this study to reveal the mechanisms on which the therapy works in order to understand which patients would benefit.”
The trial enrolled 40 patients with relapsed or refractory de novo (n=27) or transformed (n=13) DLBCL. The patients’ median age was 59.8 (range, 28.9-78.6). They received a median of 3 prior therapies (range, 1-9), and 75% of patients (n=30) were refractory to their most recent therapy.
The patients received panobinostat at 30 mg three times a week, with rituximab (n=19) or without (n=21).
Adverse events
The most common grade 3 or higher adverse events (AEs) that were thought to be related to panobinostat (with and without rituximab, respectively) were thrombocytopenia (68% and 71%) and neutropenia (11% and 24%).
Twenty-three patients (58%) required dose reductions, and 22 (55%) required dose interruptions. All of these were due to AEs, and there was no difference between patients who received rituximab and those who did not.
All 19 serious AEs were a result of disease progression.
Response
Twenty-eight percent of the patients (11/40) responded to treatment, and receiving rituximab did not increase the likelihood of response. Six patients (29%) responded to panobinostat alone, and 5 (26%) responded to panobinostat and rituximab.
There were 7 complete responses—5 of them among patients who received panobinostat alone. And there were 4 partial responses—3 among the patients who received panobinostat plus rituximab.
The median duration of response was 14.5 months overall, 9.4 months among patients who received panobinostat plus rituximab, and it was not reached among patients who received panobinostat alone.
At the data cutoff, 6 of the 11 responders had not progressed. The longest response had lasted more than 43 months and was ongoing at the cutoff point.
Predicting response
The researchers performed genome and exome sequencing in relapsed tumor biopsies and quantified ctDNA in serial plasma samples.
They found that mutations in MEF2B were significantly associated with response to panobinostat. The likelihood ratio for response was 3.67 for patients with MEF2B mutations.
On the other hand, increased levels of ctDNA were strongly associated with treatment failure. A significant increase in ctDNA at day 15 after treatment initiation could predict a lack of response with a sensitivity of 71.4% and a specificity of 100%.
“This trial has generated considerable data regarding methodology for processing samples from a clinical study, the genetic mutations associated with DLBCL and how they evolve over time, on ctDNA, and mechanisms of resistance to histone deacetylase inhibitors,” Dr Assouline concluded.
Novartis provided panobinostat and financial support for this trial, and Hoffman-LaRoche supplied rituximab. ![]()
Cancer survivors have ‘normal’ sex lives, survey says

receiving treatment
Photo by Rhoda Baer
A new study suggests cancer survivors and the general population have comparable sex lives, although cancer survivors don’t realize it.
According to a survey of more than 6500 people, cancer survivors over the age of 49 have just as much sex and similar levels of sexual function as individuals of the same age who never had cancer.
However, the cancer survivors were more likely to report being dissatisfied with their sex lives.
“We hope our findings will put cancer survivors’ concerns to rest—showing that they are just as sexually active and function just as well as others their age,” said Sarah Jackson, PhD, of University College London in the UK.
“The next stage of our research will look at why cancer patients feel less satisfied with their sex lives.”
Dr Jackson and her colleagues reported their current findings in Cancer.
The researchers set out to explore differences in sexual activity and function, as well as concerns about sex, between cancer survivors and cancer-free controls in a population-based study.
The team surveyed 3708 women (341 cancer survivors and 3367 controls) and 2982 men (220 cancer survivors and 2762 controls) aged 50 and older. Male and female cancer survivors were significantly older than controls (P<0.001 for both) and reported more comorbidities (P=0.003 for both).
Frequency
There were no significant differences in levels of sexual activity between cancer survivors and controls of either sex.
Among women, 58.2% of cancer survivors and 55.5% of controls reported having any sexual activity in the last year. Among men, the rates were 76.0% and 78.5%, respectively.
Overall, about half of the people surveyed reported having “frequent” sexual intercourse, which was defined as 2 to 3 times a month or more.
This included 49.1% of female cancer survivors, 50.1% of female controls, 49% of male cancer survivors, and 48% of male controls.
Function
The incidence of sexual problems was similar in cancer survivors and controls—both male and female.
For example, around a third of the women said they had problems becoming aroused (31.4% of cancer survivors and 31.8% of controls), and about 40% of the men had erectile dysfunction (40.3% of cancer survivors and 39.3% of controls).
Satisfaction
Despite similar levels of sexual activity and function, cancer survivors were more likely than controls to report feeling dissatisfied with their sex lives.
Among the women, 18.2% of cancer survivors and 11.8% of controls reported dissatisfaction (P=0.034). Among the men, the rates were 30.9% and 19.8%, respectively (P=0.023).
In addition, female cancer survivors were more likely to be concerned about their libido than female controls—10.2% and 7.1%, respectively (P=0.006). But there was no significant difference for the men.
Time from cancer diagnosis
The researchers also found the amount of time from cancer diagnosis was a factor affecting sexual function and concern among women but not men.
Females diagnosed with cancer less than 5 years from the time they were surveyed were more likely than female controls to report difficulty becoming aroused (55.4% and 31.8%, respectively, P=0.016) and achieving orgasm (60.6% and 28.3%, respectively, P<0.001).
The recently diagnosed females were also more likely than controls to be concerned about sexual desire (14.8% and 7.1%, respectively, P=0.007) and orgasmic experience (17.6% and 7.1%, respectively, P=0.042).
“Although some cancer treatments are known to impact on sexual function, this study suggests that the majority of cancer patients have similar sexual function and activity as the general population,” said Martin Ledwick, of Cancer Research UK, which sponsored this study.
“However, cancer patients in the study were more likely to be dissatisfied with their sex lives . . . . This highlights the need for health professionals to make sure they talk about sex with all patients—not just the ones whose sexual function is likely to be affected by their cancer or its treatment.” ![]()

receiving treatment
Photo by Rhoda Baer
A new study suggests cancer survivors and the general population have comparable sex lives, although cancer survivors don’t realize it.
According to a survey of more than 6500 people, cancer survivors over the age of 49 have just as much sex and similar levels of sexual function as individuals of the same age who never had cancer.
However, the cancer survivors were more likely to report being dissatisfied with their sex lives.
“We hope our findings will put cancer survivors’ concerns to rest—showing that they are just as sexually active and function just as well as others their age,” said Sarah Jackson, PhD, of University College London in the UK.
“The next stage of our research will look at why cancer patients feel less satisfied with their sex lives.”
Dr Jackson and her colleagues reported their current findings in Cancer.
The researchers set out to explore differences in sexual activity and function, as well as concerns about sex, between cancer survivors and cancer-free controls in a population-based study.
The team surveyed 3708 women (341 cancer survivors and 3367 controls) and 2982 men (220 cancer survivors and 2762 controls) aged 50 and older. Male and female cancer survivors were significantly older than controls (P<0.001 for both) and reported more comorbidities (P=0.003 for both).
Frequency
There were no significant differences in levels of sexual activity between cancer survivors and controls of either sex.
Among women, 58.2% of cancer survivors and 55.5% of controls reported having any sexual activity in the last year. Among men, the rates were 76.0% and 78.5%, respectively.
Overall, about half of the people surveyed reported having “frequent” sexual intercourse, which was defined as 2 to 3 times a month or more.
This included 49.1% of female cancer survivors, 50.1% of female controls, 49% of male cancer survivors, and 48% of male controls.
Function
The incidence of sexual problems was similar in cancer survivors and controls—both male and female.
For example, around a third of the women said they had problems becoming aroused (31.4% of cancer survivors and 31.8% of controls), and about 40% of the men had erectile dysfunction (40.3% of cancer survivors and 39.3% of controls).
Satisfaction
Despite similar levels of sexual activity and function, cancer survivors were more likely than controls to report feeling dissatisfied with their sex lives.
Among the women, 18.2% of cancer survivors and 11.8% of controls reported dissatisfaction (P=0.034). Among the men, the rates were 30.9% and 19.8%, respectively (P=0.023).
In addition, female cancer survivors were more likely to be concerned about their libido than female controls—10.2% and 7.1%, respectively (P=0.006). But there was no significant difference for the men.
Time from cancer diagnosis
The researchers also found the amount of time from cancer diagnosis was a factor affecting sexual function and concern among women but not men.
Females diagnosed with cancer less than 5 years from the time they were surveyed were more likely than female controls to report difficulty becoming aroused (55.4% and 31.8%, respectively, P=0.016) and achieving orgasm (60.6% and 28.3%, respectively, P<0.001).
The recently diagnosed females were also more likely than controls to be concerned about sexual desire (14.8% and 7.1%, respectively, P=0.007) and orgasmic experience (17.6% and 7.1%, respectively, P=0.042).
“Although some cancer treatments are known to impact on sexual function, this study suggests that the majority of cancer patients have similar sexual function and activity as the general population,” said Martin Ledwick, of Cancer Research UK, which sponsored this study.
“However, cancer patients in the study were more likely to be dissatisfied with their sex lives . . . . This highlights the need for health professionals to make sure they talk about sex with all patients—not just the ones whose sexual function is likely to be affected by their cancer or its treatment.” ![]()

receiving treatment
Photo by Rhoda Baer
A new study suggests cancer survivors and the general population have comparable sex lives, although cancer survivors don’t realize it.
According to a survey of more than 6500 people, cancer survivors over the age of 49 have just as much sex and similar levels of sexual function as individuals of the same age who never had cancer.
However, the cancer survivors were more likely to report being dissatisfied with their sex lives.
“We hope our findings will put cancer survivors’ concerns to rest—showing that they are just as sexually active and function just as well as others their age,” said Sarah Jackson, PhD, of University College London in the UK.
“The next stage of our research will look at why cancer patients feel less satisfied with their sex lives.”
Dr Jackson and her colleagues reported their current findings in Cancer.
The researchers set out to explore differences in sexual activity and function, as well as concerns about sex, between cancer survivors and cancer-free controls in a population-based study.
The team surveyed 3708 women (341 cancer survivors and 3367 controls) and 2982 men (220 cancer survivors and 2762 controls) aged 50 and older. Male and female cancer survivors were significantly older than controls (P<0.001 for both) and reported more comorbidities (P=0.003 for both).
Frequency
There were no significant differences in levels of sexual activity between cancer survivors and controls of either sex.
Among women, 58.2% of cancer survivors and 55.5% of controls reported having any sexual activity in the last year. Among men, the rates were 76.0% and 78.5%, respectively.
Overall, about half of the people surveyed reported having “frequent” sexual intercourse, which was defined as 2 to 3 times a month or more.
This included 49.1% of female cancer survivors, 50.1% of female controls, 49% of male cancer survivors, and 48% of male controls.
Function
The incidence of sexual problems was similar in cancer survivors and controls—both male and female.
For example, around a third of the women said they had problems becoming aroused (31.4% of cancer survivors and 31.8% of controls), and about 40% of the men had erectile dysfunction (40.3% of cancer survivors and 39.3% of controls).
Satisfaction
Despite similar levels of sexual activity and function, cancer survivors were more likely than controls to report feeling dissatisfied with their sex lives.
Among the women, 18.2% of cancer survivors and 11.8% of controls reported dissatisfaction (P=0.034). Among the men, the rates were 30.9% and 19.8%, respectively (P=0.023).
In addition, female cancer survivors were more likely to be concerned about their libido than female controls—10.2% and 7.1%, respectively (P=0.006). But there was no significant difference for the men.
Time from cancer diagnosis
The researchers also found the amount of time from cancer diagnosis was a factor affecting sexual function and concern among women but not men.
Females diagnosed with cancer less than 5 years from the time they were surveyed were more likely than female controls to report difficulty becoming aroused (55.4% and 31.8%, respectively, P=0.016) and achieving orgasm (60.6% and 28.3%, respectively, P<0.001).
The recently diagnosed females were also more likely than controls to be concerned about sexual desire (14.8% and 7.1%, respectively, P=0.007) and orgasmic experience (17.6% and 7.1%, respectively, P=0.042).
“Although some cancer treatments are known to impact on sexual function, this study suggests that the majority of cancer patients have similar sexual function and activity as the general population,” said Martin Ledwick, of Cancer Research UK, which sponsored this study.
“However, cancer patients in the study were more likely to be dissatisfied with their sex lives . . . . This highlights the need for health professionals to make sure they talk about sex with all patients—not just the ones whose sexual function is likely to be affected by their cancer or its treatment.” ![]()
Music may alleviate cancer patients’ symptoms

Photo by Lars Frantzen
Results of a systematic review suggest music can help alleviate symptoms of anxiety, pain, and fatigue in cancer patients.
The review included more than 50 studies investigating the impact of music therapy—a personalized music experience offered by trained music therapists—and music medicine—listening to pre-recorded music provided by a doctor or nurse—on psychological and physical outcomes in people with cancer.
“We found that music therapy interventions specifically help improve patients’ quality of life,” said study author Joke Bradt, PhD, of Drexel University in Philadelphia, Pennsylvania.
“These are important findings, as these outcomes play an important role in patients’ overall well-being.”
Dr Bradt and her colleagues reported their findings in Cochrane Database of Systematic Reviews.
The researchers examined 52 trials including 3731 cancer patients. The music interventions were classified as music therapy in 23 of the trials and as music medicine in 29 trials.
Analyses suggested that both types of music interventions positively impacted patients. The interventions had a moderate-to-strong effect on anxiety, a strong effect on pain reduction, and a small-to-moderate effect on fatigue.
Small reductions in heart and respiratory rates, as well as lowered blood pressure, were linked to the interventions as well.
In addition, the researchers observed a moderate increase in patients’ quality of life with music therapy but not music medicine.
The team could not determine the effect of music interventions on depression due to the low quality of evidence. And there was no evidence that the interventions improve mood, distress, or physical functioning, but there were few trials investigating these outcomes.
Similarly, the researchers said they could not draw any conclusions about the effect of music interventions on immunologic functioning, coping, resilience, or communication because there were not enough trials evaluating these outcomes.
Still, the researchers hope music interventions will become more widely used, in light of the potential benefits to cancer patients.
“We hope that the findings of this review will encourage healthcare providers in medical settings to seriously consider the use of music therapy in the psychosocial care of people with cancer,” Dr Bradt said. ![]()

Photo by Lars Frantzen
Results of a systematic review suggest music can help alleviate symptoms of anxiety, pain, and fatigue in cancer patients.
The review included more than 50 studies investigating the impact of music therapy—a personalized music experience offered by trained music therapists—and music medicine—listening to pre-recorded music provided by a doctor or nurse—on psychological and physical outcomes in people with cancer.
“We found that music therapy interventions specifically help improve patients’ quality of life,” said study author Joke Bradt, PhD, of Drexel University in Philadelphia, Pennsylvania.
“These are important findings, as these outcomes play an important role in patients’ overall well-being.”
Dr Bradt and her colleagues reported their findings in Cochrane Database of Systematic Reviews.
The researchers examined 52 trials including 3731 cancer patients. The music interventions were classified as music therapy in 23 of the trials and as music medicine in 29 trials.
Analyses suggested that both types of music interventions positively impacted patients. The interventions had a moderate-to-strong effect on anxiety, a strong effect on pain reduction, and a small-to-moderate effect on fatigue.
Small reductions in heart and respiratory rates, as well as lowered blood pressure, were linked to the interventions as well.
In addition, the researchers observed a moderate increase in patients’ quality of life with music therapy but not music medicine.
The team could not determine the effect of music interventions on depression due to the low quality of evidence. And there was no evidence that the interventions improve mood, distress, or physical functioning, but there were few trials investigating these outcomes.
Similarly, the researchers said they could not draw any conclusions about the effect of music interventions on immunologic functioning, coping, resilience, or communication because there were not enough trials evaluating these outcomes.
Still, the researchers hope music interventions will become more widely used, in light of the potential benefits to cancer patients.
“We hope that the findings of this review will encourage healthcare providers in medical settings to seriously consider the use of music therapy in the psychosocial care of people with cancer,” Dr Bradt said. ![]()

Photo by Lars Frantzen
Results of a systematic review suggest music can help alleviate symptoms of anxiety, pain, and fatigue in cancer patients.
The review included more than 50 studies investigating the impact of music therapy—a personalized music experience offered by trained music therapists—and music medicine—listening to pre-recorded music provided by a doctor or nurse—on psychological and physical outcomes in people with cancer.
“We found that music therapy interventions specifically help improve patients’ quality of life,” said study author Joke Bradt, PhD, of Drexel University in Philadelphia, Pennsylvania.
“These are important findings, as these outcomes play an important role in patients’ overall well-being.”
Dr Bradt and her colleagues reported their findings in Cochrane Database of Systematic Reviews.
The researchers examined 52 trials including 3731 cancer patients. The music interventions were classified as music therapy in 23 of the trials and as music medicine in 29 trials.
Analyses suggested that both types of music interventions positively impacted patients. The interventions had a moderate-to-strong effect on anxiety, a strong effect on pain reduction, and a small-to-moderate effect on fatigue.
Small reductions in heart and respiratory rates, as well as lowered blood pressure, were linked to the interventions as well.
In addition, the researchers observed a moderate increase in patients’ quality of life with music therapy but not music medicine.
The team could not determine the effect of music interventions on depression due to the low quality of evidence. And there was no evidence that the interventions improve mood, distress, or physical functioning, but there were few trials investigating these outcomes.
Similarly, the researchers said they could not draw any conclusions about the effect of music interventions on immunologic functioning, coping, resilience, or communication because there were not enough trials evaluating these outcomes.
Still, the researchers hope music interventions will become more widely used, in light of the potential benefits to cancer patients.
“We hope that the findings of this review will encourage healthcare providers in medical settings to seriously consider the use of music therapy in the psychosocial care of people with cancer,” Dr Bradt said. ![]()
Cancer trends shifting in HIV-positive patients
DURBAN, SOUTH AFRICA – The rates of the AIDS-defining cancers Kaposi’s sarcoma and non-Hodgkin’s lymphoma have plummeted in the antiretroviral era, yet they are still the two top cancers in terms of cumulative incidence in HIV-infected patients by age 75, Benigno Rodriguez, MD, said at the 21st International AIDS Conference.
Lung cancer also remains a major concern in the HIV-infected population. Each of these three cancers carries about a 1 in 25 lifetime risk to age 75, the estimated lifespan of HIV-positive patients on combination antiretroviral therapy (ART), according to Dr. Rodriguez of Case Western Reserve University in Cleveland.
ART has resulted in a marked change in cancer trends among HIV-infected patients. The incidence of AIDS-defining cancers has decreased “massively,” Dr. Rodriguez observed; but because ART has extended the lifespan, patients are now living long enough to get other cancers. And they do so at a higher rate than that of the general population because of their impaired immune function, higher rate of risk factors such as smoking, and greater prevalence of oncogenic viral coinfections such as hepatitis B and C and Epstein-Barr virus.
The increase in the incidence and risk of colorectal, liver, and anal cancers among HIV-positive individuals since the introduction of combination ART can largely be explained by the population’s longer exposure to risk due to increasing survival, he said.
Dr. Rodriguez presented highlights of an analysis of cancer trends in North America during 1996-2009. The study was based on 86,620 HIV-infected persons with 475,660 person-years of follow-up and 196,987 subjects without HIV infection and with more than 1.8 million person-years of follow-up. Trends over time were assessed for nine cancers: Kaposi’s sarcoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and melanoma, as well as anal, lung, colorectal, liver, and oropharyngeal cancers (Ann Intern Med. 2015;163:507-18).
Examining trends in three periods – 1996-1999, 2000-2004, and 2005-2009 – the investigators looked at the impact of ART over time on rates of AIDS-related and non–AIDS-related cancers in HIV-infected patients and compared them to results in the general HIV-uninfected population.
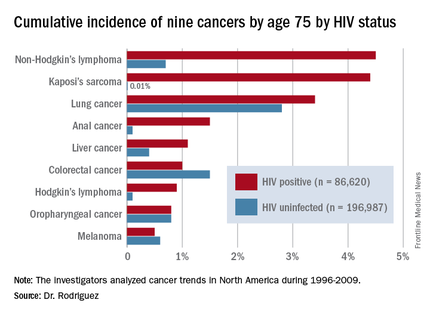
By conducting a competing risk analysis, Dr. Rodriguez and his coworkers were able to estimate the cumulative lifetime risk of the various cancers by age 75, a metric that provides readily understandable information for counseling HIV-infected patients about their long-term cancer risk.
The measure “is more intuitive than using incidence rates,” Dr. Rodriguez said. In a study of 1,578 HIV-infected patients who received the hepatitis B vaccine, for example, those patients whose immune function did not to respond to the vaccine were more likely to be among the 6% of patients who subsequently developed cancer during up to 20 years of follow-up.
The findings on cancer trends in the ART era have clinical implications for cancer screening and prevention in HIV-infected patients. The high rates of smoking and lung cancer in this population make HIV-positive smokers a logical target for lung cancer screening. The rising risk of colorectal cancer – the cumulative lifetime risk to age 75 was 0.4% in 1996-1999, 0.7% in 2000-2004, and 1.3% in 2005-2009 – suggests a need for increased colorectal cancer screening in the older HIV-positive population.
Early and sustained HIV suppression with combination ART remains the only known method of preventing AIDS-defining cancers. Dr. Rodriguez and his coinvestigators in the Centers for AIDS Research Network of Integrated Clinical Systems demonstrated the crucial role of suppressing HIV in a study of 6,036 HIV-infected patients who started on ART and were followed for more than 21,000 person-years. Compared with HIV-infected patients with a 3-month lagged HIV viremia of no more than 50 copies/mL, patients’ risk of developing non-Hodgkin’s lymphoma was 2.1-fold greater if their 3-month lagged HIV viremia was 51-500 copies/mL and 3.56-fold greater if it exceeded 500 copies/mL (Clin Infect Dis. 2014 Jun;58[11]:1599-606).
The study on cancer trends over time was funded by the National Institutes of Health. Dr. Rodriguez reported receiving honoraria from Gilead Sciences.
DURBAN, SOUTH AFRICA – The rates of the AIDS-defining cancers Kaposi’s sarcoma and non-Hodgkin’s lymphoma have plummeted in the antiretroviral era, yet they are still the two top cancers in terms of cumulative incidence in HIV-infected patients by age 75, Benigno Rodriguez, MD, said at the 21st International AIDS Conference.
Lung cancer also remains a major concern in the HIV-infected population. Each of these three cancers carries about a 1 in 25 lifetime risk to age 75, the estimated lifespan of HIV-positive patients on combination antiretroviral therapy (ART), according to Dr. Rodriguez of Case Western Reserve University in Cleveland.
ART has resulted in a marked change in cancer trends among HIV-infected patients. The incidence of AIDS-defining cancers has decreased “massively,” Dr. Rodriguez observed; but because ART has extended the lifespan, patients are now living long enough to get other cancers. And they do so at a higher rate than that of the general population because of their impaired immune function, higher rate of risk factors such as smoking, and greater prevalence of oncogenic viral coinfections such as hepatitis B and C and Epstein-Barr virus.
The increase in the incidence and risk of colorectal, liver, and anal cancers among HIV-positive individuals since the introduction of combination ART can largely be explained by the population’s longer exposure to risk due to increasing survival, he said.
Dr. Rodriguez presented highlights of an analysis of cancer trends in North America during 1996-2009. The study was based on 86,620 HIV-infected persons with 475,660 person-years of follow-up and 196,987 subjects without HIV infection and with more than 1.8 million person-years of follow-up. Trends over time were assessed for nine cancers: Kaposi’s sarcoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and melanoma, as well as anal, lung, colorectal, liver, and oropharyngeal cancers (Ann Intern Med. 2015;163:507-18).
Examining trends in three periods – 1996-1999, 2000-2004, and 2005-2009 – the investigators looked at the impact of ART over time on rates of AIDS-related and non–AIDS-related cancers in HIV-infected patients and compared them to results in the general HIV-uninfected population.

By conducting a competing risk analysis, Dr. Rodriguez and his coworkers were able to estimate the cumulative lifetime risk of the various cancers by age 75, a metric that provides readily understandable information for counseling HIV-infected patients about their long-term cancer risk.
The measure “is more intuitive than using incidence rates,” Dr. Rodriguez said. In a study of 1,578 HIV-infected patients who received the hepatitis B vaccine, for example, those patients whose immune function did not to respond to the vaccine were more likely to be among the 6% of patients who subsequently developed cancer during up to 20 years of follow-up.
The findings on cancer trends in the ART era have clinical implications for cancer screening and prevention in HIV-infected patients. The high rates of smoking and lung cancer in this population make HIV-positive smokers a logical target for lung cancer screening. The rising risk of colorectal cancer – the cumulative lifetime risk to age 75 was 0.4% in 1996-1999, 0.7% in 2000-2004, and 1.3% in 2005-2009 – suggests a need for increased colorectal cancer screening in the older HIV-positive population.
Early and sustained HIV suppression with combination ART remains the only known method of preventing AIDS-defining cancers. Dr. Rodriguez and his coinvestigators in the Centers for AIDS Research Network of Integrated Clinical Systems demonstrated the crucial role of suppressing HIV in a study of 6,036 HIV-infected patients who started on ART and were followed for more than 21,000 person-years. Compared with HIV-infected patients with a 3-month lagged HIV viremia of no more than 50 copies/mL, patients’ risk of developing non-Hodgkin’s lymphoma was 2.1-fold greater if their 3-month lagged HIV viremia was 51-500 copies/mL and 3.56-fold greater if it exceeded 500 copies/mL (Clin Infect Dis. 2014 Jun;58[11]:1599-606).
The study on cancer trends over time was funded by the National Institutes of Health. Dr. Rodriguez reported receiving honoraria from Gilead Sciences.
DURBAN, SOUTH AFRICA – The rates of the AIDS-defining cancers Kaposi’s sarcoma and non-Hodgkin’s lymphoma have plummeted in the antiretroviral era, yet they are still the two top cancers in terms of cumulative incidence in HIV-infected patients by age 75, Benigno Rodriguez, MD, said at the 21st International AIDS Conference.
Lung cancer also remains a major concern in the HIV-infected population. Each of these three cancers carries about a 1 in 25 lifetime risk to age 75, the estimated lifespan of HIV-positive patients on combination antiretroviral therapy (ART), according to Dr. Rodriguez of Case Western Reserve University in Cleveland.
ART has resulted in a marked change in cancer trends among HIV-infected patients. The incidence of AIDS-defining cancers has decreased “massively,” Dr. Rodriguez observed; but because ART has extended the lifespan, patients are now living long enough to get other cancers. And they do so at a higher rate than that of the general population because of their impaired immune function, higher rate of risk factors such as smoking, and greater prevalence of oncogenic viral coinfections such as hepatitis B and C and Epstein-Barr virus.
The increase in the incidence and risk of colorectal, liver, and anal cancers among HIV-positive individuals since the introduction of combination ART can largely be explained by the population’s longer exposure to risk due to increasing survival, he said.
Dr. Rodriguez presented highlights of an analysis of cancer trends in North America during 1996-2009. The study was based on 86,620 HIV-infected persons with 475,660 person-years of follow-up and 196,987 subjects without HIV infection and with more than 1.8 million person-years of follow-up. Trends over time were assessed for nine cancers: Kaposi’s sarcoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and melanoma, as well as anal, lung, colorectal, liver, and oropharyngeal cancers (Ann Intern Med. 2015;163:507-18).
Examining trends in three periods – 1996-1999, 2000-2004, and 2005-2009 – the investigators looked at the impact of ART over time on rates of AIDS-related and non–AIDS-related cancers in HIV-infected patients and compared them to results in the general HIV-uninfected population.

By conducting a competing risk analysis, Dr. Rodriguez and his coworkers were able to estimate the cumulative lifetime risk of the various cancers by age 75, a metric that provides readily understandable information for counseling HIV-infected patients about their long-term cancer risk.
The measure “is more intuitive than using incidence rates,” Dr. Rodriguez said. In a study of 1,578 HIV-infected patients who received the hepatitis B vaccine, for example, those patients whose immune function did not to respond to the vaccine were more likely to be among the 6% of patients who subsequently developed cancer during up to 20 years of follow-up.
The findings on cancer trends in the ART era have clinical implications for cancer screening and prevention in HIV-infected patients. The high rates of smoking and lung cancer in this population make HIV-positive smokers a logical target for lung cancer screening. The rising risk of colorectal cancer – the cumulative lifetime risk to age 75 was 0.4% in 1996-1999, 0.7% in 2000-2004, and 1.3% in 2005-2009 – suggests a need for increased colorectal cancer screening in the older HIV-positive population.
Early and sustained HIV suppression with combination ART remains the only known method of preventing AIDS-defining cancers. Dr. Rodriguez and his coinvestigators in the Centers for AIDS Research Network of Integrated Clinical Systems demonstrated the crucial role of suppressing HIV in a study of 6,036 HIV-infected patients who started on ART and were followed for more than 21,000 person-years. Compared with HIV-infected patients with a 3-month lagged HIV viremia of no more than 50 copies/mL, patients’ risk of developing non-Hodgkin’s lymphoma was 2.1-fold greater if their 3-month lagged HIV viremia was 51-500 copies/mL and 3.56-fold greater if it exceeded 500 copies/mL (Clin Infect Dis. 2014 Jun;58[11]:1599-606).
The study on cancer trends over time was funded by the National Institutes of Health. Dr. Rodriguez reported receiving honoraria from Gilead Sciences.
AT AIDS 2016
Key clinical point: HIV-infected persons in North America have roughly a 1 in 25 cumulative lifetime risk of developing lung cancer, Kaposi’s sarcoma, or non-Hodgkin’s lymphoma.
Major finding: The cumulative lifetime risk of developing non-Hodgkin’s lymphoma in HIV-infected patients on combination antiretroviral therapy is sevenfold greater than the risk in the general population.
Data source: A competing risk analysis for nine cancers based upon 86,620 HIV-infected persons followed for 475,660 person-years and 196,987 subjects not infected with HIV and with more than 1.8 million person-years of follow-up.
Disclosures: The National Institutes of Health funded the study. The presenter reported receiving honoraria from Gilead Sciences.
Drug can worsen allo-HSCT outcomes in ATLL

Photo by Chad McNeeley
Results of a large, retrospective study suggest that receiving mogamulizumab before allogeneic hematopoietic stem cell transplant (allo-HSCT) can worsen outcomes in patients with adult T-cell leukemia/lymphoma (ATLL).
Patients who received mogamulizumab had a higher risk of grade 3/4 acute graft-versus-host disease (GVHD), a higher incidence of nonrelapse mortality, and worse overall survival than patients who did not take the drug.
Researchers reported these findings in the Journal of Clinical Oncology.
Previous research suggested that pre-HSCT mogamulizumab can produce adverse effects, but these studies had small patient numbers. Researchers have suggested the adverse effects may occur because mogamulizumab depletes regulatory T cells for several months, but there has been no direct evidence supporting this idea.
To investigate the issue, Shigeo Fuji, MD, of National Cancer Center Hospital in Tokyo, Japan, and colleagues assessed the impact of pre-HSCT mogamulizumab in a large group of ATLL patients undergoing allo-HSCT.
The study included 996 patients age 70 and younger. Patients had aggressive ATLL diagnosed between 2000 and 2013. They received intensive chemotherapy as first-line treatment.
Eighty-two of the patients received mogamulizumab before HSCT, with a median of 45 days from the last mogamulizumab treatment to allo-HSCT.
Pre-HSCT mogamulizumab was associated with an increased risk of grade 3/4 acute GVHD, with a relative risk of 1.80 (P<0.01).
Patients who received mogamulizumab were also more likely to be refractory to the systemic corticosteroids given to treat acute GVHD, with a relative risk of 2.09 (P<0.01).
The 1-year cumulative incidence of nonrelapse mortality was significantly higher among patients who received mogamulizumab than among those who did not—43.7% and 25.1%, respectively (P<0.01).
And the probability of 1-year overall survival was significantly lower in patients who received mogamulizumab than in those who did not—32.3% and 49.4%, respectively (P<0.01).
The researchers noted that outcomes were particularly poor when patients received mogamulizumab within less than 50 days of allo-HSCT.
The team said this study appears to confirm that pre-HSCT mogamulizumab significantly worsens clinical outcomes, mainly because of an increased risk of severe/corticosteroid-refractory acute GVHD. And the results support the idea that the drug depletes regulatory T cells.
The researchers concluded that mogamulizumab should be used with caution in ATLL patients who are eligible for allo-HSCT. And the possibility of intensifying GVHD prophylaxis in patients who do receive pre-HSCT mogamulizumab should be explored. ![]()

Photo by Chad McNeeley
Results of a large, retrospective study suggest that receiving mogamulizumab before allogeneic hematopoietic stem cell transplant (allo-HSCT) can worsen outcomes in patients with adult T-cell leukemia/lymphoma (ATLL).
Patients who received mogamulizumab had a higher risk of grade 3/4 acute graft-versus-host disease (GVHD), a higher incidence of nonrelapse mortality, and worse overall survival than patients who did not take the drug.
Researchers reported these findings in the Journal of Clinical Oncology.
Previous research suggested that pre-HSCT mogamulizumab can produce adverse effects, but these studies had small patient numbers. Researchers have suggested the adverse effects may occur because mogamulizumab depletes regulatory T cells for several months, but there has been no direct evidence supporting this idea.
To investigate the issue, Shigeo Fuji, MD, of National Cancer Center Hospital in Tokyo, Japan, and colleagues assessed the impact of pre-HSCT mogamulizumab in a large group of ATLL patients undergoing allo-HSCT.
The study included 996 patients age 70 and younger. Patients had aggressive ATLL diagnosed between 2000 and 2013. They received intensive chemotherapy as first-line treatment.
Eighty-two of the patients received mogamulizumab before HSCT, with a median of 45 days from the last mogamulizumab treatment to allo-HSCT.
Pre-HSCT mogamulizumab was associated with an increased risk of grade 3/4 acute GVHD, with a relative risk of 1.80 (P<0.01).
Patients who received mogamulizumab were also more likely to be refractory to the systemic corticosteroids given to treat acute GVHD, with a relative risk of 2.09 (P<0.01).
The 1-year cumulative incidence of nonrelapse mortality was significantly higher among patients who received mogamulizumab than among those who did not—43.7% and 25.1%, respectively (P<0.01).
And the probability of 1-year overall survival was significantly lower in patients who received mogamulizumab than in those who did not—32.3% and 49.4%, respectively (P<0.01).
The researchers noted that outcomes were particularly poor when patients received mogamulizumab within less than 50 days of allo-HSCT.
The team said this study appears to confirm that pre-HSCT mogamulizumab significantly worsens clinical outcomes, mainly because of an increased risk of severe/corticosteroid-refractory acute GVHD. And the results support the idea that the drug depletes regulatory T cells.
The researchers concluded that mogamulizumab should be used with caution in ATLL patients who are eligible for allo-HSCT. And the possibility of intensifying GVHD prophylaxis in patients who do receive pre-HSCT mogamulizumab should be explored. ![]()

Photo by Chad McNeeley
Results of a large, retrospective study suggest that receiving mogamulizumab before allogeneic hematopoietic stem cell transplant (allo-HSCT) can worsen outcomes in patients with adult T-cell leukemia/lymphoma (ATLL).
Patients who received mogamulizumab had a higher risk of grade 3/4 acute graft-versus-host disease (GVHD), a higher incidence of nonrelapse mortality, and worse overall survival than patients who did not take the drug.
Researchers reported these findings in the Journal of Clinical Oncology.
Previous research suggested that pre-HSCT mogamulizumab can produce adverse effects, but these studies had small patient numbers. Researchers have suggested the adverse effects may occur because mogamulizumab depletes regulatory T cells for several months, but there has been no direct evidence supporting this idea.
To investigate the issue, Shigeo Fuji, MD, of National Cancer Center Hospital in Tokyo, Japan, and colleagues assessed the impact of pre-HSCT mogamulizumab in a large group of ATLL patients undergoing allo-HSCT.
The study included 996 patients age 70 and younger. Patients had aggressive ATLL diagnosed between 2000 and 2013. They received intensive chemotherapy as first-line treatment.
Eighty-two of the patients received mogamulizumab before HSCT, with a median of 45 days from the last mogamulizumab treatment to allo-HSCT.
Pre-HSCT mogamulizumab was associated with an increased risk of grade 3/4 acute GVHD, with a relative risk of 1.80 (P<0.01).
Patients who received mogamulizumab were also more likely to be refractory to the systemic corticosteroids given to treat acute GVHD, with a relative risk of 2.09 (P<0.01).
The 1-year cumulative incidence of nonrelapse mortality was significantly higher among patients who received mogamulizumab than among those who did not—43.7% and 25.1%, respectively (P<0.01).
And the probability of 1-year overall survival was significantly lower in patients who received mogamulizumab than in those who did not—32.3% and 49.4%, respectively (P<0.01).
The researchers noted that outcomes were particularly poor when patients received mogamulizumab within less than 50 days of allo-HSCT.
The team said this study appears to confirm that pre-HSCT mogamulizumab significantly worsens clinical outcomes, mainly because of an increased risk of severe/corticosteroid-refractory acute GVHD. And the results support the idea that the drug depletes regulatory T cells.
The researchers concluded that mogamulizumab should be used with caution in ATLL patients who are eligible for allo-HSCT. And the possibility of intensifying GVHD prophylaxis in patients who do receive pre-HSCT mogamulizumab should be explored. ![]()
Drug no longer in development for DLBCL, other cancers

ProNAi Therapeutics recently announced its decision to stop development of PNT2258, a drug designed to treat cancers characterized by overexpression of BCL2.
In June, the company suspended development of PNT2258, closing enrollment in a phase 2 trial of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) and a phase 2 trial of patients with Richter’s transformation.
Now, ProNAi has said it does not plan to resume development of the drug.
“[N]o further investment in PNT2258 or the underlying DNAi platform by ProNAi is contemplated, and the company subsequently has closed its research facility based in Plymouth, Michigan, which supported these programs,” the company said in a statement.
About PNT2258
PNT2258 consists of a single-stranded, 24-base DNAi oligonucleotide known as PNT100 that is encapsulated in lipid nanoparticles.
The DNAi technology platform is based on a discovery that single-stranded DNA oligonucleotides can interact with genomic DNA to interfere with oncogenes. PNT100 DNAi is designed to target a genetic regulatory region associated with BCL2.
Last March, PNT2258 was granted orphan drug designation from the US Food and Drug Administration for the treatment of DLBCL.
ProNAi initially suspended the development of PNT2258 in June, following a review of interim data from the phase 2 Wolverine trial. The company said the drug produced “modest efficacy” in this trial, but it seemed the data were not “robust enough” to justify continued development of PNT2258.
“We have decided to suspend development of PNT2258 pending further review of these data in order to determine next steps for both this asset and the DNAi platform,” Nick Glover, president and CEO of ProNAi, said at the time.
The Wolverine trial was designed to evaluate the safety and efficacy of PNT2258 monotherapy in 61 patients with relapsed/refractory DLBCL.
ProNAi reported interim safety and efficacy data as of April 25, 2016, for the first 37 subjects enrolled. The response rate was 8.1% overall (n=37) and 15.8% in the response-evaluable subgroup (n=19).
Subjects were considered response-evaluable if they met the amended eligibility criteria—a performance status of 0 to 1, 1 to 3 prior systemic treatment regimens, and receipt of at least 8 doses of PNT2258 within 35 days of starting therapy.
PNT2258 was also being evaluated in patients with Richter’s transformation in the phase 2 Brighton study. In June, ProNAi said it had enrolled 5 subjects in this study, and 4 had discontinued. No responses were observed.
“On the basis of these interim assessments, we have decided to close the Wolverine and Brighton studies to further enrollment of new subjects,” Barbara Klencke, chief development officer of ProNAi, said at the time.
PNT2258 was evaluated in 2 prior studies as well. In a phase 1 study (NCT01191775), PNT2258 was given to 22 subjects with advanced solid tumors. The drug was considered well tolerated at doses ranging from 1 mg/m2 through 150 mg/m2.
A pilot study of PNT2258 (NCT01733238) enrolled 13 subjects with relapsed/refractory B-cell non-Hodgkin lymphoma. Responses were observed in subjects with DLBCL and those with follicular lymphoma.
Six subjects were progression-free at 12 months, and progression-free survival extended to 2 years and beyond in 4 subjects. The majority of the adverse events were grade 1 or 2. ![]()

ProNAi Therapeutics recently announced its decision to stop development of PNT2258, a drug designed to treat cancers characterized by overexpression of BCL2.
In June, the company suspended development of PNT2258, closing enrollment in a phase 2 trial of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) and a phase 2 trial of patients with Richter’s transformation.
Now, ProNAi has said it does not plan to resume development of the drug.
“[N]o further investment in PNT2258 or the underlying DNAi platform by ProNAi is contemplated, and the company subsequently has closed its research facility based in Plymouth, Michigan, which supported these programs,” the company said in a statement.
About PNT2258
PNT2258 consists of a single-stranded, 24-base DNAi oligonucleotide known as PNT100 that is encapsulated in lipid nanoparticles.
The DNAi technology platform is based on a discovery that single-stranded DNA oligonucleotides can interact with genomic DNA to interfere with oncogenes. PNT100 DNAi is designed to target a genetic regulatory region associated with BCL2.
Last March, PNT2258 was granted orphan drug designation from the US Food and Drug Administration for the treatment of DLBCL.
ProNAi initially suspended the development of PNT2258 in June, following a review of interim data from the phase 2 Wolverine trial. The company said the drug produced “modest efficacy” in this trial, but it seemed the data were not “robust enough” to justify continued development of PNT2258.
“We have decided to suspend development of PNT2258 pending further review of these data in order to determine next steps for both this asset and the DNAi platform,” Nick Glover, president and CEO of ProNAi, said at the time.
The Wolverine trial was designed to evaluate the safety and efficacy of PNT2258 monotherapy in 61 patients with relapsed/refractory DLBCL.
ProNAi reported interim safety and efficacy data as of April 25, 2016, for the first 37 subjects enrolled. The response rate was 8.1% overall (n=37) and 15.8% in the response-evaluable subgroup (n=19).
Subjects were considered response-evaluable if they met the amended eligibility criteria—a performance status of 0 to 1, 1 to 3 prior systemic treatment regimens, and receipt of at least 8 doses of PNT2258 within 35 days of starting therapy.
PNT2258 was also being evaluated in patients with Richter’s transformation in the phase 2 Brighton study. In June, ProNAi said it had enrolled 5 subjects in this study, and 4 had discontinued. No responses were observed.
“On the basis of these interim assessments, we have decided to close the Wolverine and Brighton studies to further enrollment of new subjects,” Barbara Klencke, chief development officer of ProNAi, said at the time.
PNT2258 was evaluated in 2 prior studies as well. In a phase 1 study (NCT01191775), PNT2258 was given to 22 subjects with advanced solid tumors. The drug was considered well tolerated at doses ranging from 1 mg/m2 through 150 mg/m2.
A pilot study of PNT2258 (NCT01733238) enrolled 13 subjects with relapsed/refractory B-cell non-Hodgkin lymphoma. Responses were observed in subjects with DLBCL and those with follicular lymphoma.
Six subjects were progression-free at 12 months, and progression-free survival extended to 2 years and beyond in 4 subjects. The majority of the adverse events were grade 1 or 2. ![]()

ProNAi Therapeutics recently announced its decision to stop development of PNT2258, a drug designed to treat cancers characterized by overexpression of BCL2.
In June, the company suspended development of PNT2258, closing enrollment in a phase 2 trial of patients with relapsed/refractory diffuse large B-cell lymphoma (DLBCL) and a phase 2 trial of patients with Richter’s transformation.
Now, ProNAi has said it does not plan to resume development of the drug.
“[N]o further investment in PNT2258 or the underlying DNAi platform by ProNAi is contemplated, and the company subsequently has closed its research facility based in Plymouth, Michigan, which supported these programs,” the company said in a statement.
About PNT2258
PNT2258 consists of a single-stranded, 24-base DNAi oligonucleotide known as PNT100 that is encapsulated in lipid nanoparticles.
The DNAi technology platform is based on a discovery that single-stranded DNA oligonucleotides can interact with genomic DNA to interfere with oncogenes. PNT100 DNAi is designed to target a genetic regulatory region associated with BCL2.
Last March, PNT2258 was granted orphan drug designation from the US Food and Drug Administration for the treatment of DLBCL.
ProNAi initially suspended the development of PNT2258 in June, following a review of interim data from the phase 2 Wolverine trial. The company said the drug produced “modest efficacy” in this trial, but it seemed the data were not “robust enough” to justify continued development of PNT2258.
“We have decided to suspend development of PNT2258 pending further review of these data in order to determine next steps for both this asset and the DNAi platform,” Nick Glover, president and CEO of ProNAi, said at the time.
The Wolverine trial was designed to evaluate the safety and efficacy of PNT2258 monotherapy in 61 patients with relapsed/refractory DLBCL.
ProNAi reported interim safety and efficacy data as of April 25, 2016, for the first 37 subjects enrolled. The response rate was 8.1% overall (n=37) and 15.8% in the response-evaluable subgroup (n=19).
Subjects were considered response-evaluable if they met the amended eligibility criteria—a performance status of 0 to 1, 1 to 3 prior systemic treatment regimens, and receipt of at least 8 doses of PNT2258 within 35 days of starting therapy.
PNT2258 was also being evaluated in patients with Richter’s transformation in the phase 2 Brighton study. In June, ProNAi said it had enrolled 5 subjects in this study, and 4 had discontinued. No responses were observed.
“On the basis of these interim assessments, we have decided to close the Wolverine and Brighton studies to further enrollment of new subjects,” Barbara Klencke, chief development officer of ProNAi, said at the time.
PNT2258 was evaluated in 2 prior studies as well. In a phase 1 study (NCT01191775), PNT2258 was given to 22 subjects with advanced solid tumors. The drug was considered well tolerated at doses ranging from 1 mg/m2 through 150 mg/m2.
A pilot study of PNT2258 (NCT01733238) enrolled 13 subjects with relapsed/refractory B-cell non-Hodgkin lymphoma. Responses were observed in subjects with DLBCL and those with follicular lymphoma.
Six subjects were progression-free at 12 months, and progression-free survival extended to 2 years and beyond in 4 subjects. The majority of the adverse events were grade 1 or 2. ![]()
Pulmonary complications affect cancer survivors long-term

Photo by Bill Branson
A new study suggests that survivors of childhood cancer may be plagued by pulmonary complications related to treatment well into their adult lives.
The research indicated that the cumulative incidence of pulmonary complications continues to increase up to 25 years from a patient’s initial cancer diagnosis.
In addition, platinum-based chemotherapy and higher doses of radiation were linked to an increased risk of death from pulmonary causes.
Andrew C. Dietz, MD, of Children’s Hospital Los Angeles in California, and his colleagues reported these findings in Cancer.
The researchers analyzed data from 20,690 five-year cancer survivors who participated in the Childhood Cancer Survivor Study to determine the incidence of death from pulmonary causes.
The team also assessed the incidence of various pulmonary complications in 14,316 of those cancer survivors (who completed a baseline survey and/or 1 of 2 follow-up surveys years later), comparing the results to those seen in a control group of 4027 cancer survivor siblings.
About 34% of the 14,316 cancer survivors had been diagnosed with acute leukemia, and about 21% were diagnosed with Hodgkin or non-Hodgkin lymphoma.
The cancer survivors’ median age at diagnosis was 7 (range, 0-21), and their median age at evaluation was 32 (range, 6-59). The median time from diagnosis was 25 years (range, 5-39).
Compared with controls, cancer survivors were more likely to be male, black, and Hispanic. Cancer survivors were slightly younger and more likely to report a history of congestive heart failure, but they were less likely to be overweight/obese or have ever smoked at the time of the baseline survey.
Results
By age 45, the cumulative incidence of any pulmonary condition was 29.6% among cancer survivors and 26.5% among controls (P=0.001).
The cancer survivors were more likely than controls to report chronic cough (rate ratio [RR]=1.6), the need for extra oxygen (RR=1.8), lung fibrosis (RR=3.5), and recurrent pneumonia (RR=2.0).
Among cancer survivors, the risk of asthma was significantly associated with exposure to asparaginase. Chronic cough was significantly associated with chest wall or lung surgery, anthracyclines, hydroxyurea, and lung radiation doses ≥15 Gy.
Emphysema was significantly associated with lomustine. The need for extra oxygen was significantly associated with hematopoietic stem cell transplant, chest wall or lung surgery, and lung radiation doses ≥10 Gy.
Lung fibrosis was significantly associated with chest wall or lung surgery, asparaginase, platinum-based chemotherapy, and lung radiation doses ≥10 Gy. Recurrent pneumonia was significantly associated with lung radiation doses ≥15 Gy.
The standardized mortality ratio for death from pulmonary causes among all eligible cancer survivors (n=20,690) was 5.9. Pulmonary death was significantly associated with exposure to platinum-based agents and lung radiation doses ≥10 Gy.
“This study adds to our understanding of specific, long-term risks to pulmonary health for survivors of childhood cancer and will help refine guidelines for appropriate screening, health surveillance, and counseling,” said study author Daniel A. Mulrooney, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
He added that this knowledge could potentially contribute to the design and testing of better, targeted interventions to decrease adverse pulmonary events in this population. ![]()

Photo by Bill Branson
A new study suggests that survivors of childhood cancer may be plagued by pulmonary complications related to treatment well into their adult lives.
The research indicated that the cumulative incidence of pulmonary complications continues to increase up to 25 years from a patient’s initial cancer diagnosis.
In addition, platinum-based chemotherapy and higher doses of radiation were linked to an increased risk of death from pulmonary causes.
Andrew C. Dietz, MD, of Children’s Hospital Los Angeles in California, and his colleagues reported these findings in Cancer.
The researchers analyzed data from 20,690 five-year cancer survivors who participated in the Childhood Cancer Survivor Study to determine the incidence of death from pulmonary causes.
The team also assessed the incidence of various pulmonary complications in 14,316 of those cancer survivors (who completed a baseline survey and/or 1 of 2 follow-up surveys years later), comparing the results to those seen in a control group of 4027 cancer survivor siblings.
About 34% of the 14,316 cancer survivors had been diagnosed with acute leukemia, and about 21% were diagnosed with Hodgkin or non-Hodgkin lymphoma.
The cancer survivors’ median age at diagnosis was 7 (range, 0-21), and their median age at evaluation was 32 (range, 6-59). The median time from diagnosis was 25 years (range, 5-39).
Compared with controls, cancer survivors were more likely to be male, black, and Hispanic. Cancer survivors were slightly younger and more likely to report a history of congestive heart failure, but they were less likely to be overweight/obese or have ever smoked at the time of the baseline survey.
Results
By age 45, the cumulative incidence of any pulmonary condition was 29.6% among cancer survivors and 26.5% among controls (P=0.001).
The cancer survivors were more likely than controls to report chronic cough (rate ratio [RR]=1.6), the need for extra oxygen (RR=1.8), lung fibrosis (RR=3.5), and recurrent pneumonia (RR=2.0).
Among cancer survivors, the risk of asthma was significantly associated with exposure to asparaginase. Chronic cough was significantly associated with chest wall or lung surgery, anthracyclines, hydroxyurea, and lung radiation doses ≥15 Gy.
Emphysema was significantly associated with lomustine. The need for extra oxygen was significantly associated with hematopoietic stem cell transplant, chest wall or lung surgery, and lung radiation doses ≥10 Gy.
Lung fibrosis was significantly associated with chest wall or lung surgery, asparaginase, platinum-based chemotherapy, and lung radiation doses ≥10 Gy. Recurrent pneumonia was significantly associated with lung radiation doses ≥15 Gy.
The standardized mortality ratio for death from pulmonary causes among all eligible cancer survivors (n=20,690) was 5.9. Pulmonary death was significantly associated with exposure to platinum-based agents and lung radiation doses ≥10 Gy.
“This study adds to our understanding of specific, long-term risks to pulmonary health for survivors of childhood cancer and will help refine guidelines for appropriate screening, health surveillance, and counseling,” said study author Daniel A. Mulrooney, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
He added that this knowledge could potentially contribute to the design and testing of better, targeted interventions to decrease adverse pulmonary events in this population. ![]()

Photo by Bill Branson
A new study suggests that survivors of childhood cancer may be plagued by pulmonary complications related to treatment well into their adult lives.
The research indicated that the cumulative incidence of pulmonary complications continues to increase up to 25 years from a patient’s initial cancer diagnosis.
In addition, platinum-based chemotherapy and higher doses of radiation were linked to an increased risk of death from pulmonary causes.
Andrew C. Dietz, MD, of Children’s Hospital Los Angeles in California, and his colleagues reported these findings in Cancer.
The researchers analyzed data from 20,690 five-year cancer survivors who participated in the Childhood Cancer Survivor Study to determine the incidence of death from pulmonary causes.
The team also assessed the incidence of various pulmonary complications in 14,316 of those cancer survivors (who completed a baseline survey and/or 1 of 2 follow-up surveys years later), comparing the results to those seen in a control group of 4027 cancer survivor siblings.
About 34% of the 14,316 cancer survivors had been diagnosed with acute leukemia, and about 21% were diagnosed with Hodgkin or non-Hodgkin lymphoma.
The cancer survivors’ median age at diagnosis was 7 (range, 0-21), and their median age at evaluation was 32 (range, 6-59). The median time from diagnosis was 25 years (range, 5-39).
Compared with controls, cancer survivors were more likely to be male, black, and Hispanic. Cancer survivors were slightly younger and more likely to report a history of congestive heart failure, but they were less likely to be overweight/obese or have ever smoked at the time of the baseline survey.
Results
By age 45, the cumulative incidence of any pulmonary condition was 29.6% among cancer survivors and 26.5% among controls (P=0.001).
The cancer survivors were more likely than controls to report chronic cough (rate ratio [RR]=1.6), the need for extra oxygen (RR=1.8), lung fibrosis (RR=3.5), and recurrent pneumonia (RR=2.0).
Among cancer survivors, the risk of asthma was significantly associated with exposure to asparaginase. Chronic cough was significantly associated with chest wall or lung surgery, anthracyclines, hydroxyurea, and lung radiation doses ≥15 Gy.
Emphysema was significantly associated with lomustine. The need for extra oxygen was significantly associated with hematopoietic stem cell transplant, chest wall or lung surgery, and lung radiation doses ≥10 Gy.
Lung fibrosis was significantly associated with chest wall or lung surgery, asparaginase, platinum-based chemotherapy, and lung radiation doses ≥10 Gy. Recurrent pneumonia was significantly associated with lung radiation doses ≥15 Gy.
The standardized mortality ratio for death from pulmonary causes among all eligible cancer survivors (n=20,690) was 5.9. Pulmonary death was significantly associated with exposure to platinum-based agents and lung radiation doses ≥10 Gy.
“This study adds to our understanding of specific, long-term risks to pulmonary health for survivors of childhood cancer and will help refine guidelines for appropriate screening, health surveillance, and counseling,” said study author Daniel A. Mulrooney, MD, of St. Jude Children’s Research Hospital in Memphis, Tennessee.
He added that this knowledge could potentially contribute to the design and testing of better, targeted interventions to decrease adverse pulmonary events in this population.
Research provides new insight into CLL

New research explains how an inherited genetic variant associated with an increased risk of chronic lymphocytic leukemia (CLL) helps cancer cells survive.
Previous research showed that DNA variations at 15q15.1 are linked with an increased risk of CLL.
With the current study, researchers believe they have identified the causal variant at 15q15.1 and determined the mechanism by which it influences tumorigenesis.
Richard Houlston, MD, PhD, of The Institute of Cancer Research in London, UK, and his colleagues conducted this research and detailed the results in Cell Reports.
The researchers said the single nucleotide polymorphism rs539846 underlies the 15q15.1 CLL risk locus.
And the rs539846-A risk allele interferes with BCL-2 modifying factor (BMF), which normally works to produce pro-apoptotic signals.
This interference makes it harder for the protein RELA to “flip on” the activity of BMF and reduces levels of the pro-apoptotic signals, allowing CLL cells to sidestep self-destruction.
“Although many significant risk variants for this type of leukemia have been identified, the biological mechanisms through which these variants affect leukemia development have been less well studied,” Dr Houlston said.
“This study highlights the importance of cell-death-inducing proteins such as BMF in controlling CLL development and could help in the design of new drugs to treat this disease.”
In addition, Dr Houlston and his colleagues said their findings complement work from phase 1 and phase 2 trials of venetoclax (formerly ABT-199) in CLL.
The trials suggested that venetoclax mimics pro-apoptotic proteins by targeting the pro-survival BCL-2 pathway. In this way, the drug can produce anticancer effects in CLL patients.
Dr Houlston and his colleagues believe their discovery could provide important insight into how venetoclax and similar drugs work, which could optimize the drugs’ use.

New research explains how an inherited genetic variant associated with an increased risk of chronic lymphocytic leukemia (CLL) helps cancer cells survive.
Previous research showed that DNA variations at 15q15.1 are linked with an increased risk of CLL.
With the current study, researchers believe they have identified the causal variant at 15q15.1 and determined the mechanism by which it influences tumorigenesis.
Richard Houlston, MD, PhD, of The Institute of Cancer Research in London, UK, and his colleagues conducted this research and detailed the results in Cell Reports.
The researchers said the single nucleotide polymorphism rs539846 underlies the 15q15.1 CLL risk locus.
And the rs539846-A risk allele interferes with BCL-2 modifying factor (BMF), which normally works to produce pro-apoptotic signals.
This interference makes it harder for the protein RELA to “flip on” the activity of BMF and reduces levels of the pro-apoptotic signals, allowing CLL cells to sidestep self-destruction.
“Although many significant risk variants for this type of leukemia have been identified, the biological mechanisms through which these variants affect leukemia development have been less well studied,” Dr Houlston said.
“This study highlights the importance of cell-death-inducing proteins such as BMF in controlling CLL development and could help in the design of new drugs to treat this disease.”
In addition, Dr Houlston and his colleagues said their findings complement work from phase 1 and phase 2 trials of venetoclax (formerly ABT-199) in CLL.
The trials suggested that venetoclax mimics pro-apoptotic proteins by targeting the pro-survival BCL-2 pathway. In this way, the drug can produce anticancer effects in CLL patients.
Dr Houlston and his colleagues believe their discovery could provide important insight into how venetoclax and similar drugs work, which could optimize the drugs’ use.

New research explains how an inherited genetic variant associated with an increased risk of chronic lymphocytic leukemia (CLL) helps cancer cells survive.
Previous research showed that DNA variations at 15q15.1 are linked with an increased risk of CLL.
With the current study, researchers believe they have identified the causal variant at 15q15.1 and determined the mechanism by which it influences tumorigenesis.
Richard Houlston, MD, PhD, of The Institute of Cancer Research in London, UK, and his colleagues conducted this research and detailed the results in Cell Reports.
The researchers said the single nucleotide polymorphism rs539846 underlies the 15q15.1 CLL risk locus.
And the rs539846-A risk allele interferes with BCL-2 modifying factor (BMF), which normally works to produce pro-apoptotic signals.
This interference makes it harder for the protein RELA to “flip on” the activity of BMF and reduces levels of the pro-apoptotic signals, allowing CLL cells to sidestep self-destruction.
“Although many significant risk variants for this type of leukemia have been identified, the biological mechanisms through which these variants affect leukemia development have been less well studied,” Dr Houlston said.
“This study highlights the importance of cell-death-inducing proteins such as BMF in controlling CLL development and could help in the design of new drugs to treat this disease.”
In addition, Dr Houlston and his colleagues said their findings complement work from phase 1 and phase 2 trials of venetoclax (formerly ABT-199) in CLL.
The trials suggested that venetoclax mimics pro-apoptotic proteins by targeting the pro-survival BCL-2 pathway. In this way, the drug can produce anticancer effects in CLL patients.
Dr Houlston and his colleagues believe their discovery could provide important insight into how venetoclax and similar drugs work, which could optimize the drugs’ use.

