User login
VIDEO: Venetoclax shows early good results in multiple myeloma
SAN DIEGO – Venetoclax has shown preliminary good results as an investigational monotherapy in patients with relapsed/refractory multiple myeloma, based on phase I data reported at the annual meeting of the American Society of Hematology.
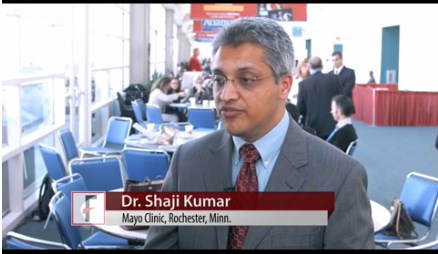
Shaji Kumar, MD, of the Mayo Clinic, Rochester, Minn., reported that venetoclax monotherapy had anti-myeloma activity in a dose-finding study among patients treated with a median of five previous therapies. As would be expected, the best responses to the small-molecule BCL-2 inhibitor were seen primarily in patients with t(11;14) chromosomal aberrations and high BCL-2, low BCL-XL and low MCL-1 expression levels.
In a video interview, Dr. Kumar discussed the results of this early-stage research as well as ongoing studies that are beginning to examine venetoclax in combination regimens for multiple myeloma.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Venetoclax is approved for the treatment of patients with chronic lymphocytic leukemia with 17p deletion who have received at least one prior treatment. Dr. Kumar receives research funding from Abbvie, the maker of venetoclax (Venclexta), and is a consultant to and receives research funding from several other drug companies.
mdales@frontlinemedcom.com
On Twitter @maryjodales
SAN DIEGO – Venetoclax has shown preliminary good results as an investigational monotherapy in patients with relapsed/refractory multiple myeloma, based on phase I data reported at the annual meeting of the American Society of Hematology.
Shaji Kumar, MD, of the Mayo Clinic, Rochester, Minn., reported that venetoclax monotherapy had anti-myeloma activity in a dose-finding study among patients treated with a median of five previous therapies. As would be expected, the best responses to the small-molecule BCL-2 inhibitor were seen primarily in patients with t(11;14) chromosomal aberrations and high BCL-2, low BCL-XL and low MCL-1 expression levels.
In a video interview, Dr. Kumar discussed the results of this early-stage research as well as ongoing studies that are beginning to examine venetoclax in combination regimens for multiple myeloma.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Venetoclax is approved for the treatment of patients with chronic lymphocytic leukemia with 17p deletion who have received at least one prior treatment. Dr. Kumar receives research funding from Abbvie, the maker of venetoclax (Venclexta), and is a consultant to and receives research funding from several other drug companies.
mdales@frontlinemedcom.com
On Twitter @maryjodales
SAN DIEGO – Venetoclax has shown preliminary good results as an investigational monotherapy in patients with relapsed/refractory multiple myeloma, based on phase I data reported at the annual meeting of the American Society of Hematology.
Shaji Kumar, MD, of the Mayo Clinic, Rochester, Minn., reported that venetoclax monotherapy had anti-myeloma activity in a dose-finding study among patients treated with a median of five previous therapies. As would be expected, the best responses to the small-molecule BCL-2 inhibitor were seen primarily in patients with t(11;14) chromosomal aberrations and high BCL-2, low BCL-XL and low MCL-1 expression levels.
In a video interview, Dr. Kumar discussed the results of this early-stage research as well as ongoing studies that are beginning to examine venetoclax in combination regimens for multiple myeloma.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Venetoclax is approved for the treatment of patients with chronic lymphocytic leukemia with 17p deletion who have received at least one prior treatment. Dr. Kumar receives research funding from Abbvie, the maker of venetoclax (Venclexta), and is a consultant to and receives research funding from several other drug companies.
mdales@frontlinemedcom.com
On Twitter @maryjodales
AT ASH 2016
FDA grants priority review to sBLA for pembrolizumab

Photo courtesy of Merck
The US Food and Drug Administration (FDA) has granted priority review to the supplemental biologics license application (sBLA) for pembrolizumab (Keytruda®) as a treatment for patients with refractory classical Hodgkin lymphoma (cHL) and for cHL patients who have relapsed after 3 or more prior lines of therapy.
The sBLA will be reviewed under the FDA’s accelerated approval program. The target action date is March 15, 2017.
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the antitumor immune response.
The drug, which is being developed by Merck, already has FDA approval as a treatment for melanoma, lung cancer, and head and neck cancer.
Pembrolizumab also has breakthrough therapy designation as a treatment for relapsed/refractory cHL.
The current sBLA for pembrolizumab is seeking approval for the drug at a fixed dose of 200 mg, administered intravenously every 3 weeks.
This is the first application for regulatory approval of pembrolizumab in a hematologic malignancy.
The sBLA is supported by data from the phase 1 KEYNOTE-013 trial and the phase 2 KEYNOTE-087 trial.
Results from KEYNOTE-013 (in cHL patients) were presented at the 2014 ASH Annual Meeting, and results from KEYNOTE-087 were presented at the 2016 ASCO Annual Meeting. ![]()

Photo courtesy of Merck
The US Food and Drug Administration (FDA) has granted priority review to the supplemental biologics license application (sBLA) for pembrolizumab (Keytruda®) as a treatment for patients with refractory classical Hodgkin lymphoma (cHL) and for cHL patients who have relapsed after 3 or more prior lines of therapy.
The sBLA will be reviewed under the FDA’s accelerated approval program. The target action date is March 15, 2017.
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the antitumor immune response.
The drug, which is being developed by Merck, already has FDA approval as a treatment for melanoma, lung cancer, and head and neck cancer.
Pembrolizumab also has breakthrough therapy designation as a treatment for relapsed/refractory cHL.
The current sBLA for pembrolizumab is seeking approval for the drug at a fixed dose of 200 mg, administered intravenously every 3 weeks.
This is the first application for regulatory approval of pembrolizumab in a hematologic malignancy.
The sBLA is supported by data from the phase 1 KEYNOTE-013 trial and the phase 2 KEYNOTE-087 trial.
Results from KEYNOTE-013 (in cHL patients) were presented at the 2014 ASH Annual Meeting, and results from KEYNOTE-087 were presented at the 2016 ASCO Annual Meeting. ![]()

Photo courtesy of Merck
The US Food and Drug Administration (FDA) has granted priority review to the supplemental biologics license application (sBLA) for pembrolizumab (Keytruda®) as a treatment for patients with refractory classical Hodgkin lymphoma (cHL) and for cHL patients who have relapsed after 3 or more prior lines of therapy.
The sBLA will be reviewed under the FDA’s accelerated approval program. The target action date is March 15, 2017.
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the antitumor immune response.
The drug, which is being developed by Merck, already has FDA approval as a treatment for melanoma, lung cancer, and head and neck cancer.
Pembrolizumab also has breakthrough therapy designation as a treatment for relapsed/refractory cHL.
The current sBLA for pembrolizumab is seeking approval for the drug at a fixed dose of 200 mg, administered intravenously every 3 weeks.
This is the first application for regulatory approval of pembrolizumab in a hematologic malignancy.
The sBLA is supported by data from the phase 1 KEYNOTE-013 trial and the phase 2 KEYNOTE-087 trial.
Results from KEYNOTE-013 (in cHL patients) were presented at the 2014 ASH Annual Meeting, and results from KEYNOTE-087 were presented at the 2016 ASCO Annual Meeting. ![]()
NCCN releases new guidelines for cancer patients

Nausea and Vomiting
©NCCN® 2016
The National Comprehensive Cancer Network (NCCN) has released new educational materials designed to help cancer patients combat nausea and vomiting.
The NCCN Guidelines for Patients® for Nausea and Vomiting and NCCN Quick Guide™ for Nausea and Vomiting are the first patient resources from NCCN to focus specifically on supportive care.
The resources are available on NCCN.org/patients and via the NCCN Patient Guides for Cancer mobile app.
NCCN Guidelines for Patients are patient-friendly translations of the NCCN Clinical Practice Guidelines in Oncology. Each resource features guidance from US cancer centers designed to help people living with cancer talk with their physicians about the best treatment options for their disease.
NCCN Quick Guide™ sheets are 1-page summaries of key points in the patient guidelines. They include elements such as “questions to ask your doctor,” a glossary of terms, and medical illustrations of anatomy, tests, and treatments.
The NCCN Guidelines for Patients for Nausea and Vomiting:
- Explain how these side effects are related to cancer treatment
- List cancer treatments that can cause nausea and vomiting
- Detail methods of preventing and treating these side effects
- Outline methods of coping with nausea and vomiting
- Provide a list of resources for information and support.
“At NCCN, our mission is to improve the lives of patients with cancer, and we are excited to be able to provide the information that will help patients better understand this common side effect of cancer treatment,” said Marcie R. Reeder, executive director of the NCCN Foundation.
“The NCCN Guidelines for Patients for Nausea and Vomiting are the first of a highly anticipated library of supportive care resources that provide patients with the same information their doctors use.” ![]()

Nausea and Vomiting
©NCCN® 2016
The National Comprehensive Cancer Network (NCCN) has released new educational materials designed to help cancer patients combat nausea and vomiting.
The NCCN Guidelines for Patients® for Nausea and Vomiting and NCCN Quick Guide™ for Nausea and Vomiting are the first patient resources from NCCN to focus specifically on supportive care.
The resources are available on NCCN.org/patients and via the NCCN Patient Guides for Cancer mobile app.
NCCN Guidelines for Patients are patient-friendly translations of the NCCN Clinical Practice Guidelines in Oncology. Each resource features guidance from US cancer centers designed to help people living with cancer talk with their physicians about the best treatment options for their disease.
NCCN Quick Guide™ sheets are 1-page summaries of key points in the patient guidelines. They include elements such as “questions to ask your doctor,” a glossary of terms, and medical illustrations of anatomy, tests, and treatments.
The NCCN Guidelines for Patients for Nausea and Vomiting:
- Explain how these side effects are related to cancer treatment
- List cancer treatments that can cause nausea and vomiting
- Detail methods of preventing and treating these side effects
- Outline methods of coping with nausea and vomiting
- Provide a list of resources for information and support.
“At NCCN, our mission is to improve the lives of patients with cancer, and we are excited to be able to provide the information that will help patients better understand this common side effect of cancer treatment,” said Marcie R. Reeder, executive director of the NCCN Foundation.
“The NCCN Guidelines for Patients for Nausea and Vomiting are the first of a highly anticipated library of supportive care resources that provide patients with the same information their doctors use.” ![]()

Nausea and Vomiting
©NCCN® 2016
The National Comprehensive Cancer Network (NCCN) has released new educational materials designed to help cancer patients combat nausea and vomiting.
The NCCN Guidelines for Patients® for Nausea and Vomiting and NCCN Quick Guide™ for Nausea and Vomiting are the first patient resources from NCCN to focus specifically on supportive care.
The resources are available on NCCN.org/patients and via the NCCN Patient Guides for Cancer mobile app.
NCCN Guidelines for Patients are patient-friendly translations of the NCCN Clinical Practice Guidelines in Oncology. Each resource features guidance from US cancer centers designed to help people living with cancer talk with their physicians about the best treatment options for their disease.
NCCN Quick Guide™ sheets are 1-page summaries of key points in the patient guidelines. They include elements such as “questions to ask your doctor,” a glossary of terms, and medical illustrations of anatomy, tests, and treatments.
The NCCN Guidelines for Patients for Nausea and Vomiting:
- Explain how these side effects are related to cancer treatment
- List cancer treatments that can cause nausea and vomiting
- Detail methods of preventing and treating these side effects
- Outline methods of coping with nausea and vomiting
- Provide a list of resources for information and support.
“At NCCN, our mission is to improve the lives of patients with cancer, and we are excited to be able to provide the information that will help patients better understand this common side effect of cancer treatment,” said Marcie R. Reeder, executive director of the NCCN Foundation.
“The NCCN Guidelines for Patients for Nausea and Vomiting are the first of a highly anticipated library of supportive care resources that provide patients with the same information their doctors use.” ![]()
NICE recommends taking ibrutinib off CDF

Photo courtesy of Janssen
The National Institute for Health and Care Excellence (NICE) has issued a final appraisal determination recommending that ibrutinib come off the Cancer Drugs Fund (CDF) and be made available through the National Health Service (NHS).
Ibrutinib is a Bruton’s tyrosine kinase inhibitor approved by the European Commission treat patients with chronic lymphocytic leukemia (CLL).
NICE is recommending that ibrutinib be made available through the NHS for previously treated CLL patients and untreated CLL patients who have 17p deletion or TP53 mutation.
This means patients will no longer have to apply to the CDF to obtain ibrutinib. The CDF is money the English government sets aside to pay for cancer drugs that haven’t been approved by NICE and aren’t available within the NHS.
Though certain NICE products and services are provided to Wales, Scotland, and Northern Ireland, the governments of these countries do not have a CDF
or similar program.
Following the decision to reform the CDF earlier this year, NICE began to reappraise all drugs in the CDF in April.
New recommendation
NICE previously said it could not recommend ibrutinib for routine NHS use. However, Janssen, the company that makes ibrutinib, agreed to reduce the
price of the drug for the NHS. Because of the discount, an independent appraisal committee was able to deem ibrutinib cost-effective.
The list price for a single tablet of ibrutinib (140 mg) is £51.10 (excluding tax). The cost of a year’s course of ibrutinib treatment is £55,954.50 (excluding tax).
The discount the NHS will receive is confidential. The Department of Health said the cost of ibrutinib will not constitute an excessive administrative burden on the NHS.
NICE’s final appraisal determination on ibrutinib is now with consultees who have the opportunity to appeal against it. If there is no appeal, or an appeal is not upheld, the final appraisal determination is issued by NICE as a guidance.
Once NICE issues a final guidance on ibrutinib, the NHS must make the drug available within 3 months. ![]()

Photo courtesy of Janssen
The National Institute for Health and Care Excellence (NICE) has issued a final appraisal determination recommending that ibrutinib come off the Cancer Drugs Fund (CDF) and be made available through the National Health Service (NHS).
Ibrutinib is a Bruton’s tyrosine kinase inhibitor approved by the European Commission treat patients with chronic lymphocytic leukemia (CLL).
NICE is recommending that ibrutinib be made available through the NHS for previously treated CLL patients and untreated CLL patients who have 17p deletion or TP53 mutation.
This means patients will no longer have to apply to the CDF to obtain ibrutinib. The CDF is money the English government sets aside to pay for cancer drugs that haven’t been approved by NICE and aren’t available within the NHS.
Though certain NICE products and services are provided to Wales, Scotland, and Northern Ireland, the governments of these countries do not have a CDF
or similar program.
Following the decision to reform the CDF earlier this year, NICE began to reappraise all drugs in the CDF in April.
New recommendation
NICE previously said it could not recommend ibrutinib for routine NHS use. However, Janssen, the company that makes ibrutinib, agreed to reduce the
price of the drug for the NHS. Because of the discount, an independent appraisal committee was able to deem ibrutinib cost-effective.
The list price for a single tablet of ibrutinib (140 mg) is £51.10 (excluding tax). The cost of a year’s course of ibrutinib treatment is £55,954.50 (excluding tax).
The discount the NHS will receive is confidential. The Department of Health said the cost of ibrutinib will not constitute an excessive administrative burden on the NHS.
NICE’s final appraisal determination on ibrutinib is now with consultees who have the opportunity to appeal against it. If there is no appeal, or an appeal is not upheld, the final appraisal determination is issued by NICE as a guidance.
Once NICE issues a final guidance on ibrutinib, the NHS must make the drug available within 3 months. ![]()

Photo courtesy of Janssen
The National Institute for Health and Care Excellence (NICE) has issued a final appraisal determination recommending that ibrutinib come off the Cancer Drugs Fund (CDF) and be made available through the National Health Service (NHS).
Ibrutinib is a Bruton’s tyrosine kinase inhibitor approved by the European Commission treat patients with chronic lymphocytic leukemia (CLL).
NICE is recommending that ibrutinib be made available through the NHS for previously treated CLL patients and untreated CLL patients who have 17p deletion or TP53 mutation.
This means patients will no longer have to apply to the CDF to obtain ibrutinib. The CDF is money the English government sets aside to pay for cancer drugs that haven’t been approved by NICE and aren’t available within the NHS.
Though certain NICE products and services are provided to Wales, Scotland, and Northern Ireland, the governments of these countries do not have a CDF
or similar program.
Following the decision to reform the CDF earlier this year, NICE began to reappraise all drugs in the CDF in April.
New recommendation
NICE previously said it could not recommend ibrutinib for routine NHS use. However, Janssen, the company that makes ibrutinib, agreed to reduce the
price of the drug for the NHS. Because of the discount, an independent appraisal committee was able to deem ibrutinib cost-effective.
The list price for a single tablet of ibrutinib (140 mg) is £51.10 (excluding tax). The cost of a year’s course of ibrutinib treatment is £55,954.50 (excluding tax).
The discount the NHS will receive is confidential. The Department of Health said the cost of ibrutinib will not constitute an excessive administrative burden on the NHS.
NICE’s final appraisal determination on ibrutinib is now with consultees who have the opportunity to appeal against it. If there is no appeal, or an appeal is not upheld, the final appraisal determination is issued by NICE as a guidance.
Once NICE issues a final guidance on ibrutinib, the NHS must make the drug available within 3 months. ![]()
Tazemetostat receives fast track designation for DLBCL

The US Food and Drug Administration (FDA) has granted fast track designation for tazemetostat as a treatment for patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) with EZH2 activating mutations.
Tazemetostat inhibits EZH2, a histone methyltransferase that appears to play a role in the growth and proliferation of a number of cancers, including DLBCL.
Tazemetostat is being developed by Epizyme, Inc.
The FDA’s fast track program is designed to facilitate the development and expedite the review of products intended to treat or prevent serious or life-threatening conditions and address unmet medical need.
Through the FDA’s fast track program, a product may be eligible for priority review. In addition, the company developing the product may be allowed to submit sections of the biologic license application or new drug application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings and written communications with the FDA.
Tazemetostat trials
Tazemetostat is under investigation as monotherapy and in combination with other agents as a treatment for multiple cancers.
Results from a phase 1 study suggested tazemetostat monotherapy can produce durable responses in patients with advanced non-Hodgkin lymphomas, including DLBCL. The study was presented at the 2015 ASH Annual Meeting.
Now, Epizyme is conducting a phase 2 study of tazemetostat monotherapy in adults with relapsed or refractory DLBCL or follicular lymphoma.
Tazemetostat is also being evaluated in 2 combination studies in patients with DLBCL.
In a phase 1b/2 trial, researchers are investigating tazemetostat in combination with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) as a front-line treatment for patients with DLBCL.
In a phase 1b study, researchers are evaluating tazemetostat in combination with atezolizumab, an anti-PD-L1 immunotherapy, in patients with relapsed and refractory DLBCL. ![]()

The US Food and Drug Administration (FDA) has granted fast track designation for tazemetostat as a treatment for patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) with EZH2 activating mutations.
Tazemetostat inhibits EZH2, a histone methyltransferase that appears to play a role in the growth and proliferation of a number of cancers, including DLBCL.
Tazemetostat is being developed by Epizyme, Inc.
The FDA’s fast track program is designed to facilitate the development and expedite the review of products intended to treat or prevent serious or life-threatening conditions and address unmet medical need.
Through the FDA’s fast track program, a product may be eligible for priority review. In addition, the company developing the product may be allowed to submit sections of the biologic license application or new drug application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings and written communications with the FDA.
Tazemetostat trials
Tazemetostat is under investigation as monotherapy and in combination with other agents as a treatment for multiple cancers.
Results from a phase 1 study suggested tazemetostat monotherapy can produce durable responses in patients with advanced non-Hodgkin lymphomas, including DLBCL. The study was presented at the 2015 ASH Annual Meeting.
Now, Epizyme is conducting a phase 2 study of tazemetostat monotherapy in adults with relapsed or refractory DLBCL or follicular lymphoma.
Tazemetostat is also being evaluated in 2 combination studies in patients with DLBCL.
In a phase 1b/2 trial, researchers are investigating tazemetostat in combination with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) as a front-line treatment for patients with DLBCL.
In a phase 1b study, researchers are evaluating tazemetostat in combination with atezolizumab, an anti-PD-L1 immunotherapy, in patients with relapsed and refractory DLBCL. ![]()

The US Food and Drug Administration (FDA) has granted fast track designation for tazemetostat as a treatment for patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) with EZH2 activating mutations.
Tazemetostat inhibits EZH2, a histone methyltransferase that appears to play a role in the growth and proliferation of a number of cancers, including DLBCL.
Tazemetostat is being developed by Epizyme, Inc.
The FDA’s fast track program is designed to facilitate the development and expedite the review of products intended to treat or prevent serious or life-threatening conditions and address unmet medical need.
Through the FDA’s fast track program, a product may be eligible for priority review. In addition, the company developing the product may be allowed to submit sections of the biologic license application or new drug application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings and written communications with the FDA.
Tazemetostat trials
Tazemetostat is under investigation as monotherapy and in combination with other agents as a treatment for multiple cancers.
Results from a phase 1 study suggested tazemetostat monotherapy can produce durable responses in patients with advanced non-Hodgkin lymphomas, including DLBCL. The study was presented at the 2015 ASH Annual Meeting.
Now, Epizyme is conducting a phase 2 study of tazemetostat monotherapy in adults with relapsed or refractory DLBCL or follicular lymphoma.
Tazemetostat is also being evaluated in 2 combination studies in patients with DLBCL.
In a phase 1b/2 trial, researchers are investigating tazemetostat in combination with R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone) as a front-line treatment for patients with DLBCL.
In a phase 1b study, researchers are evaluating tazemetostat in combination with atezolizumab, an anti-PD-L1 immunotherapy, in patients with relapsed and refractory DLBCL. ![]()
Worse outcomes for double-hit lymphomas after ASCT
Patients with double-hit lymphomas (DHLs) and double-expressor lymphomas (DELs) have inferior outcomes after undergoing autologous stem cell transplantation (ASCT), according to a new study published in the Journal of Clinical Oncology.
The worst outcomes were observed in patients with concurrent DELs and DHLs, and this “supports the concept that the double-hit/double-expressor biology appears to render DLBCL resistant to and less likely to be cured by chemotherapy,” write Alex Herrera, MD, an oncologist at the City of Hope, Duarte, Calif., and his colleagues.
But that said, a significant proportion of patients with relapsed/refractory DEL did experience durable remissions following ASCT, particularly those with isolated DEL without DHL.
This suggests that “the presence of DEL alone should not be considered a contraindication to ASCT,” the authors wrote (J Clin Oncol. 2016 Oct. 24. doi: 10.1200/JCO.2016.68.2740).
DHLs and DELs are subtypes of diffuse large B-cell lymphoma (DLBCL), and while they are associated with poor outcomes after standard chemoimmunotherapy, data remain limited as to outcomes of patients with relapsed or refractory disease who undergo ASCT.
The retrospective multicenter study included 117 patients with chemotherapy-sensitive relapsed/refractory DLBCL who underwent ASCT and had archival tumor material available. DEL with MYC/BCL2 coexpression was observed in 52 patients (44%) while 15 patients expressed MYC-R (13%), of whom 12 (10%) had DHL.
The median follow-up time was 45 months for survivors, and the 4-year progression-free survival (PFS) and overall survival (OS) were 54% for the entire cohort.
The 4-year PFS and OS in patients with DHL was worse as compared to those without DHL; 28% vs. 57% (P = .013), and 25% vs. 66% (P less than .001), respectively.
Those with DHL had poorer PFS (28%) and OS (25%), compared with patients with DEL but not DHL (PFS, 53% and OS, 61%) as well as patients with neither DEL nor DHL (PFS, 60% and OS, 70%; three-way P value for PFS, P = .013; OS, P = .002).
Patients with concurrent DEL and DHL had the poorest outcome, with a 4-year PFS of 0%.
After researchers adjusted for clinical characteristics, the only factors that remained significantly associated with PFS were DEL (hazard ratio, 1.8; P = .035) and DHL (HR, 2.9; P = .009). Factors that were significantly associated with OS were DHL (HR, 3.4; P = .004) and remission status at ASCT (HR for partial response, 2.4; P = .007).
Overall, patients with DHL were less likely to achieve a complete response following salvage therapy, and those with DEL and patients with DHL had a shorter time to relapse after induction therapy.
“Although some patients with relapsed/refractory DHL had long-term remission after ASCT (isolated DHL without DEL), the low survival rate in this group argues that alternative transplantation strategies, including allogeneic hematopoietic stem cell transplantation or peri-ASCT relapse prevention strategies should be studied,” they concluded.
Recognizing that the majority of patients with double-hit or double-expressor lymphoma will relapse after R-CHOP, this study evaluates the efficacy of autologous stem cell transplantation as a salvage modality. This is a carefully conducted, albeit retrospective, analysis of patients with relapsed or refractory double-hit or double-expressor lymphoma undergoing autologous hematopoietic stem cell transplantation (auto-HCT) at two high-volume institutions.
Although it is perhaps not surprising that double-hit and double-expressor phenotypes confer inferior outcomes, it is worth examining these issues in some detail. The first issue is that the authors have defined categories that are not recognized by the World Health Organization, but are routinely seen in clinical practice.
For example, it might be assumed that all patients with double-hit lymphoma will have MYC/BCL2 protein expression and, therefore, also have double-expressor lymphoma, but some patients with double-hit lymphoma do not have protein expression of MYC/BCL2 and these patients may have better outcomes than patients whose tumors display both double-hit and double-expressor characteristics.
A second caveat to interpreting the results is that the study population does not reflect the true denominator of all patients with relapsed diffuse large B-cell lymphoma, because only chemotherapy-sensitive patients undergoing auto-HCT were included.
So, should patients with relapsed double-hit and double-expressor lymphoma be offered auto-HCT? What are the alternatives to auto-HCT? Unfortunately, there are no clear answers to these questions, although the surprisingly excellent outcomes for patients without either of these features (70% long-term survival) suggest that there is a group of patients for whom auto-HCT remains an effective and standard tool. For double-hit and double-expressor lymphoma, a clinical trial based on specific biologic changes in individual patients is the ideal but is far from reality at this point.
Overall, despite being a retrospective series with a high attrition rate based on tissue availability, the central review of pathology, uniform assessment of double-hit and double-expressor features, and mature follow-up of 45 months makes this a thought-provoking and timely paper.
Sonali M. Smith, MD, is from the University of Chicago, and has disclosed a consulting or advisory role with Genentech, Seattle Genetics, TG Therapeutics, Gilead Sciences, Immunogenix, Pharmacyclics, NanoString Technologies, Genmab, Juno Therapeutics, Abbvie, and Portola Pharmaceuticals. These remarks were taken from the editorial accompanying Dr. Herrara’s report (J Clin Oncol. 2016 Oct 17. doi: 10.1200/JCO.2016.70.0625).
Recognizing that the majority of patients with double-hit or double-expressor lymphoma will relapse after R-CHOP, this study evaluates the efficacy of autologous stem cell transplantation as a salvage modality. This is a carefully conducted, albeit retrospective, analysis of patients with relapsed or refractory double-hit or double-expressor lymphoma undergoing autologous hematopoietic stem cell transplantation (auto-HCT) at two high-volume institutions.
Although it is perhaps not surprising that double-hit and double-expressor phenotypes confer inferior outcomes, it is worth examining these issues in some detail. The first issue is that the authors have defined categories that are not recognized by the World Health Organization, but are routinely seen in clinical practice.
For example, it might be assumed that all patients with double-hit lymphoma will have MYC/BCL2 protein expression and, therefore, also have double-expressor lymphoma, but some patients with double-hit lymphoma do not have protein expression of MYC/BCL2 and these patients may have better outcomes than patients whose tumors display both double-hit and double-expressor characteristics.
A second caveat to interpreting the results is that the study population does not reflect the true denominator of all patients with relapsed diffuse large B-cell lymphoma, because only chemotherapy-sensitive patients undergoing auto-HCT were included.
So, should patients with relapsed double-hit and double-expressor lymphoma be offered auto-HCT? What are the alternatives to auto-HCT? Unfortunately, there are no clear answers to these questions, although the surprisingly excellent outcomes for patients without either of these features (70% long-term survival) suggest that there is a group of patients for whom auto-HCT remains an effective and standard tool. For double-hit and double-expressor lymphoma, a clinical trial based on specific biologic changes in individual patients is the ideal but is far from reality at this point.
Overall, despite being a retrospective series with a high attrition rate based on tissue availability, the central review of pathology, uniform assessment of double-hit and double-expressor features, and mature follow-up of 45 months makes this a thought-provoking and timely paper.
Sonali M. Smith, MD, is from the University of Chicago, and has disclosed a consulting or advisory role with Genentech, Seattle Genetics, TG Therapeutics, Gilead Sciences, Immunogenix, Pharmacyclics, NanoString Technologies, Genmab, Juno Therapeutics, Abbvie, and Portola Pharmaceuticals. These remarks were taken from the editorial accompanying Dr. Herrara’s report (J Clin Oncol. 2016 Oct 17. doi: 10.1200/JCO.2016.70.0625).
Recognizing that the majority of patients with double-hit or double-expressor lymphoma will relapse after R-CHOP, this study evaluates the efficacy of autologous stem cell transplantation as a salvage modality. This is a carefully conducted, albeit retrospective, analysis of patients with relapsed or refractory double-hit or double-expressor lymphoma undergoing autologous hematopoietic stem cell transplantation (auto-HCT) at two high-volume institutions.
Although it is perhaps not surprising that double-hit and double-expressor phenotypes confer inferior outcomes, it is worth examining these issues in some detail. The first issue is that the authors have defined categories that are not recognized by the World Health Organization, but are routinely seen in clinical practice.
For example, it might be assumed that all patients with double-hit lymphoma will have MYC/BCL2 protein expression and, therefore, also have double-expressor lymphoma, but some patients with double-hit lymphoma do not have protein expression of MYC/BCL2 and these patients may have better outcomes than patients whose tumors display both double-hit and double-expressor characteristics.
A second caveat to interpreting the results is that the study population does not reflect the true denominator of all patients with relapsed diffuse large B-cell lymphoma, because only chemotherapy-sensitive patients undergoing auto-HCT were included.
So, should patients with relapsed double-hit and double-expressor lymphoma be offered auto-HCT? What are the alternatives to auto-HCT? Unfortunately, there are no clear answers to these questions, although the surprisingly excellent outcomes for patients without either of these features (70% long-term survival) suggest that there is a group of patients for whom auto-HCT remains an effective and standard tool. For double-hit and double-expressor lymphoma, a clinical trial based on specific biologic changes in individual patients is the ideal but is far from reality at this point.
Overall, despite being a retrospective series with a high attrition rate based on tissue availability, the central review of pathology, uniform assessment of double-hit and double-expressor features, and mature follow-up of 45 months makes this a thought-provoking and timely paper.
Sonali M. Smith, MD, is from the University of Chicago, and has disclosed a consulting or advisory role with Genentech, Seattle Genetics, TG Therapeutics, Gilead Sciences, Immunogenix, Pharmacyclics, NanoString Technologies, Genmab, Juno Therapeutics, Abbvie, and Portola Pharmaceuticals. These remarks were taken from the editorial accompanying Dr. Herrara’s report (J Clin Oncol. 2016 Oct 17. doi: 10.1200/JCO.2016.70.0625).
Patients with double-hit lymphomas (DHLs) and double-expressor lymphomas (DELs) have inferior outcomes after undergoing autologous stem cell transplantation (ASCT), according to a new study published in the Journal of Clinical Oncology.
The worst outcomes were observed in patients with concurrent DELs and DHLs, and this “supports the concept that the double-hit/double-expressor biology appears to render DLBCL resistant to and less likely to be cured by chemotherapy,” write Alex Herrera, MD, an oncologist at the City of Hope, Duarte, Calif., and his colleagues.
But that said, a significant proportion of patients with relapsed/refractory DEL did experience durable remissions following ASCT, particularly those with isolated DEL without DHL.
This suggests that “the presence of DEL alone should not be considered a contraindication to ASCT,” the authors wrote (J Clin Oncol. 2016 Oct. 24. doi: 10.1200/JCO.2016.68.2740).
DHLs and DELs are subtypes of diffuse large B-cell lymphoma (DLBCL), and while they are associated with poor outcomes after standard chemoimmunotherapy, data remain limited as to outcomes of patients with relapsed or refractory disease who undergo ASCT.
The retrospective multicenter study included 117 patients with chemotherapy-sensitive relapsed/refractory DLBCL who underwent ASCT and had archival tumor material available. DEL with MYC/BCL2 coexpression was observed in 52 patients (44%) while 15 patients expressed MYC-R (13%), of whom 12 (10%) had DHL.
The median follow-up time was 45 months for survivors, and the 4-year progression-free survival (PFS) and overall survival (OS) were 54% for the entire cohort.
The 4-year PFS and OS in patients with DHL was worse as compared to those without DHL; 28% vs. 57% (P = .013), and 25% vs. 66% (P less than .001), respectively.
Those with DHL had poorer PFS (28%) and OS (25%), compared with patients with DEL but not DHL (PFS, 53% and OS, 61%) as well as patients with neither DEL nor DHL (PFS, 60% and OS, 70%; three-way P value for PFS, P = .013; OS, P = .002).
Patients with concurrent DEL and DHL had the poorest outcome, with a 4-year PFS of 0%.
After researchers adjusted for clinical characteristics, the only factors that remained significantly associated with PFS were DEL (hazard ratio, 1.8; P = .035) and DHL (HR, 2.9; P = .009). Factors that were significantly associated with OS were DHL (HR, 3.4; P = .004) and remission status at ASCT (HR for partial response, 2.4; P = .007).
Overall, patients with DHL were less likely to achieve a complete response following salvage therapy, and those with DEL and patients with DHL had a shorter time to relapse after induction therapy.
“Although some patients with relapsed/refractory DHL had long-term remission after ASCT (isolated DHL without DEL), the low survival rate in this group argues that alternative transplantation strategies, including allogeneic hematopoietic stem cell transplantation or peri-ASCT relapse prevention strategies should be studied,” they concluded.
Patients with double-hit lymphomas (DHLs) and double-expressor lymphomas (DELs) have inferior outcomes after undergoing autologous stem cell transplantation (ASCT), according to a new study published in the Journal of Clinical Oncology.
The worst outcomes were observed in patients with concurrent DELs and DHLs, and this “supports the concept that the double-hit/double-expressor biology appears to render DLBCL resistant to and less likely to be cured by chemotherapy,” write Alex Herrera, MD, an oncologist at the City of Hope, Duarte, Calif., and his colleagues.
But that said, a significant proportion of patients with relapsed/refractory DEL did experience durable remissions following ASCT, particularly those with isolated DEL without DHL.
This suggests that “the presence of DEL alone should not be considered a contraindication to ASCT,” the authors wrote (J Clin Oncol. 2016 Oct. 24. doi: 10.1200/JCO.2016.68.2740).
DHLs and DELs are subtypes of diffuse large B-cell lymphoma (DLBCL), and while they are associated with poor outcomes after standard chemoimmunotherapy, data remain limited as to outcomes of patients with relapsed or refractory disease who undergo ASCT.
The retrospective multicenter study included 117 patients with chemotherapy-sensitive relapsed/refractory DLBCL who underwent ASCT and had archival tumor material available. DEL with MYC/BCL2 coexpression was observed in 52 patients (44%) while 15 patients expressed MYC-R (13%), of whom 12 (10%) had DHL.
The median follow-up time was 45 months for survivors, and the 4-year progression-free survival (PFS) and overall survival (OS) were 54% for the entire cohort.
The 4-year PFS and OS in patients with DHL was worse as compared to those without DHL; 28% vs. 57% (P = .013), and 25% vs. 66% (P less than .001), respectively.
Those with DHL had poorer PFS (28%) and OS (25%), compared with patients with DEL but not DHL (PFS, 53% and OS, 61%) as well as patients with neither DEL nor DHL (PFS, 60% and OS, 70%; three-way P value for PFS, P = .013; OS, P = .002).
Patients with concurrent DEL and DHL had the poorest outcome, with a 4-year PFS of 0%.
After researchers adjusted for clinical characteristics, the only factors that remained significantly associated with PFS were DEL (hazard ratio, 1.8; P = .035) and DHL (HR, 2.9; P = .009). Factors that were significantly associated with OS were DHL (HR, 3.4; P = .004) and remission status at ASCT (HR for partial response, 2.4; P = .007).
Overall, patients with DHL were less likely to achieve a complete response following salvage therapy, and those with DEL and patients with DHL had a shorter time to relapse after induction therapy.
“Although some patients with relapsed/refractory DHL had long-term remission after ASCT (isolated DHL without DEL), the low survival rate in this group argues that alternative transplantation strategies, including allogeneic hematopoietic stem cell transplantation or peri-ASCT relapse prevention strategies should be studied,” they concluded.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point:
Major finding: The 4-year progression-free survival in patients with DEL vs. non-DEL was 48% versus 59% (P = .049), and the 4-year OS was 56% vs. 67% (P = .10).
Data source: Retrospective, multicenter study that included 117 patients with relapsed/refractory aggressive B-cell non-Hodgkin lymphoma who underwent ASCT.
Disclosures: The study was funded by a Conquer Cancer Foundation/ASCO Young Investigator Award and National Cancer Institute Grants; the Dana-Farber Cancer Institute Award Fund for Collaborative Research Initiatives in Hematologic Oncology; the Harold and Virginia Lash/David Lash Fund for Lymphoma Research; and NCI Grant No. P30CA033572 for work performed in the COH Pathology Core. Dr. Herrera reports receiving research funding from Seattle Genetics, Pharmacyclics, Genentech, Immune Design, and Sequenta, and received travel, accommodations, and expenses from Bristol-Myers Squibb. Several coauthors also report relationships with industry.
EC approves nivolumab for relapsed/refractory cHL

Photo from Business Wire
The European Commission (EC) has approved nivolumab (Opdivo) for the treatment of adults with relapsed or refractory classical Hodgkin lymphoma (cHL) who have already received an autologous hematopoietic stem cell transplant (auto-HSCT) and treatment with brentuximab vedotin (BV).
Nivolumab is the first PD-1 inhibitor approved in the European Economic Area as a treatment for a hematologic malignancy.
The EC previously approved nivolumab to treat advanced melanoma, non-small cell lung cancer, and renal cell carcinoma. In Europe, nivolumab is marketed by Bristol-Myers Squibb.
Trials in cHL
The EC’s approval of nivolumab in cHL is based on an integrated analysis of data from 2 trials—the phase 1 CheckMate -039 trial and the phase 2 CheckMate -205 trial.
In CheckMate -039, researchers evaluated nivolumab in patients with cHL, non-Hodgkin lymphoma, and multiple myeloma. Results from this trial were presented at the 13th International Congress on Malignant Lymphoma in June 2015.
In CheckMate -205, researchers are evaluating nivolumab in 4 cohorts of cHL patients. Cohort A includes patients who previously received auto-HSCT and were BV-naïve at enrollment (n=63). Cohort B includes patients who previously received auto-HSCT followed by BV (n=80).
Cohort C includes patients who previously received BV before and/or after auto-HSCT (n=100). And cohort D, which is currently enrolling, is an evaluation of nivolumab in combination with chemotherapy in newly diagnosed, advanced-stage cHL patients who are treatment-naïve (n=50).
Results from cohort B were presented at the 21st Congress of the European Hematology Association in June 2016. Results from cohort C were presented at the 10th International Symposium on Hodgkin Lymphoma last month.
Integrated analysis
The analysis included cHL patients from CheckMate -205 and -039 who had received auto-HSCT and BV.
In the efficacy population (n=95), the objective response rate was 66%. The percentage of patients with a complete response was 6%. Twenty-three percent of patients had stable disease.
The median time to response was 2.0 months (range, 0.7-11.1), and the median duration of response was 13.1 months (range, 0.0+, 23.1+). At 12 months, the progression-free survival rate was 57%.
The safety of nivolumab in cHL was evaluated in 263 patients from CheckMate -205 (n=240) and CheckMate -039 (n=23). Serious adverse events (AEs) occurred in 21% of these patients.
The most common serious AEs (reported in at least 1% of patients) were infusion-related reactions, pneumonia, pleural effusion, pyrexia, rash, and pneumonitis.
The most common AEs (reported in at least 20% of patients) were fatigue (32%), upper respiratory tract infection (28%), pyrexia (24%), diarrhea (23%), and cough (22%).
Twenty-three percent of patients had a dose delay resulting from an AE, and 4.2% of patients discontinued treatment due to AEs.
Forty patients went on to allogeneic HSCT after nivolumab, and 6 of these patients died from complications of the transplant. The 40 patients had a median follow-up from allogeneic HSCT of 2.9 months (range, 0-22).
Because of these deaths, the US Food and Drug Administration asked Bristol-Myers Squibb to study the safety of allogeneic HSCT after nivolumab. ![]()

Photo from Business Wire
The European Commission (EC) has approved nivolumab (Opdivo) for the treatment of adults with relapsed or refractory classical Hodgkin lymphoma (cHL) who have already received an autologous hematopoietic stem cell transplant (auto-HSCT) and treatment with brentuximab vedotin (BV).
Nivolumab is the first PD-1 inhibitor approved in the European Economic Area as a treatment for a hematologic malignancy.
The EC previously approved nivolumab to treat advanced melanoma, non-small cell lung cancer, and renal cell carcinoma. In Europe, nivolumab is marketed by Bristol-Myers Squibb.
Trials in cHL
The EC’s approval of nivolumab in cHL is based on an integrated analysis of data from 2 trials—the phase 1 CheckMate -039 trial and the phase 2 CheckMate -205 trial.
In CheckMate -039, researchers evaluated nivolumab in patients with cHL, non-Hodgkin lymphoma, and multiple myeloma. Results from this trial were presented at the 13th International Congress on Malignant Lymphoma in June 2015.
In CheckMate -205, researchers are evaluating nivolumab in 4 cohorts of cHL patients. Cohort A includes patients who previously received auto-HSCT and were BV-naïve at enrollment (n=63). Cohort B includes patients who previously received auto-HSCT followed by BV (n=80).
Cohort C includes patients who previously received BV before and/or after auto-HSCT (n=100). And cohort D, which is currently enrolling, is an evaluation of nivolumab in combination with chemotherapy in newly diagnosed, advanced-stage cHL patients who are treatment-naïve (n=50).
Results from cohort B were presented at the 21st Congress of the European Hematology Association in June 2016. Results from cohort C were presented at the 10th International Symposium on Hodgkin Lymphoma last month.
Integrated analysis
The analysis included cHL patients from CheckMate -205 and -039 who had received auto-HSCT and BV.
In the efficacy population (n=95), the objective response rate was 66%. The percentage of patients with a complete response was 6%. Twenty-three percent of patients had stable disease.
The median time to response was 2.0 months (range, 0.7-11.1), and the median duration of response was 13.1 months (range, 0.0+, 23.1+). At 12 months, the progression-free survival rate was 57%.
The safety of nivolumab in cHL was evaluated in 263 patients from CheckMate -205 (n=240) and CheckMate -039 (n=23). Serious adverse events (AEs) occurred in 21% of these patients.
The most common serious AEs (reported in at least 1% of patients) were infusion-related reactions, pneumonia, pleural effusion, pyrexia, rash, and pneumonitis.
The most common AEs (reported in at least 20% of patients) were fatigue (32%), upper respiratory tract infection (28%), pyrexia (24%), diarrhea (23%), and cough (22%).
Twenty-three percent of patients had a dose delay resulting from an AE, and 4.2% of patients discontinued treatment due to AEs.
Forty patients went on to allogeneic HSCT after nivolumab, and 6 of these patients died from complications of the transplant. The 40 patients had a median follow-up from allogeneic HSCT of 2.9 months (range, 0-22).
Because of these deaths, the US Food and Drug Administration asked Bristol-Myers Squibb to study the safety of allogeneic HSCT after nivolumab. ![]()

Photo from Business Wire
The European Commission (EC) has approved nivolumab (Opdivo) for the treatment of adults with relapsed or refractory classical Hodgkin lymphoma (cHL) who have already received an autologous hematopoietic stem cell transplant (auto-HSCT) and treatment with brentuximab vedotin (BV).
Nivolumab is the first PD-1 inhibitor approved in the European Economic Area as a treatment for a hematologic malignancy.
The EC previously approved nivolumab to treat advanced melanoma, non-small cell lung cancer, and renal cell carcinoma. In Europe, nivolumab is marketed by Bristol-Myers Squibb.
Trials in cHL
The EC’s approval of nivolumab in cHL is based on an integrated analysis of data from 2 trials—the phase 1 CheckMate -039 trial and the phase 2 CheckMate -205 trial.
In CheckMate -039, researchers evaluated nivolumab in patients with cHL, non-Hodgkin lymphoma, and multiple myeloma. Results from this trial were presented at the 13th International Congress on Malignant Lymphoma in June 2015.
In CheckMate -205, researchers are evaluating nivolumab in 4 cohorts of cHL patients. Cohort A includes patients who previously received auto-HSCT and were BV-naïve at enrollment (n=63). Cohort B includes patients who previously received auto-HSCT followed by BV (n=80).
Cohort C includes patients who previously received BV before and/or after auto-HSCT (n=100). And cohort D, which is currently enrolling, is an evaluation of nivolumab in combination with chemotherapy in newly diagnosed, advanced-stage cHL patients who are treatment-naïve (n=50).
Results from cohort B were presented at the 21st Congress of the European Hematology Association in June 2016. Results from cohort C were presented at the 10th International Symposium on Hodgkin Lymphoma last month.
Integrated analysis
The analysis included cHL patients from CheckMate -205 and -039 who had received auto-HSCT and BV.
In the efficacy population (n=95), the objective response rate was 66%. The percentage of patients with a complete response was 6%. Twenty-three percent of patients had stable disease.
The median time to response was 2.0 months (range, 0.7-11.1), and the median duration of response was 13.1 months (range, 0.0+, 23.1+). At 12 months, the progression-free survival rate was 57%.
The safety of nivolumab in cHL was evaluated in 263 patients from CheckMate -205 (n=240) and CheckMate -039 (n=23). Serious adverse events (AEs) occurred in 21% of these patients.
The most common serious AEs (reported in at least 1% of patients) were infusion-related reactions, pneumonia, pleural effusion, pyrexia, rash, and pneumonitis.
The most common AEs (reported in at least 20% of patients) were fatigue (32%), upper respiratory tract infection (28%), pyrexia (24%), diarrhea (23%), and cough (22%).
Twenty-three percent of patients had a dose delay resulting from an AE, and 4.2% of patients discontinued treatment due to AEs.
Forty patients went on to allogeneic HSCT after nivolumab, and 6 of these patients died from complications of the transplant. The 40 patients had a median follow-up from allogeneic HSCT of 2.9 months (range, 0-22).
Because of these deaths, the US Food and Drug Administration asked Bristol-Myers Squibb to study the safety of allogeneic HSCT after nivolumab. ![]()
A look at HIV-related cancers: incidence, screening, and stem transplantation
HIV-related lymphoma rate remains sky-high despite ART
Key clinical point ART has had a major impact on the incidence of HIV-related non-Hodgkin lymphoma but no effect on Hodgkin lymphoma. Major finding The overall incidence of non-Hodgkin lymphoma in HIV-positive adults on ART is 287 cases per 100,000 person-years, varying by location and route of HIV acquisition. Data source A longitudinal analysis of non-Hodgkin lymphoma incidence in more than 210,000 HIV-infected adults on combination ART on 4 continents. Disclosures The European Union and the National Institutes of Health. The presenter reported having no financial conflicts of interest.
Click on the PDF icon at the top of this introduction to read the full article.
HIV-related lymphoma rate remains sky-high despite ART
Key clinical point ART has had a major impact on the incidence of HIV-related non-Hodgkin lymphoma but no effect on Hodgkin lymphoma. Major finding The overall incidence of non-Hodgkin lymphoma in HIV-positive adults on ART is 287 cases per 100,000 person-years, varying by location and route of HIV acquisition. Data source A longitudinal analysis of non-Hodgkin lymphoma incidence in more than 210,000 HIV-infected adults on combination ART on 4 continents. Disclosures The European Union and the National Institutes of Health. The presenter reported having no financial conflicts of interest.
Click on the PDF icon at the top of this introduction to read the full article.
HIV-related lymphoma rate remains sky-high despite ART
Key clinical point ART has had a major impact on the incidence of HIV-related non-Hodgkin lymphoma but no effect on Hodgkin lymphoma. Major finding The overall incidence of non-Hodgkin lymphoma in HIV-positive adults on ART is 287 cases per 100,000 person-years, varying by location and route of HIV acquisition. Data source A longitudinal analysis of non-Hodgkin lymphoma incidence in more than 210,000 HIV-infected adults on combination ART on 4 continents. Disclosures The European Union and the National Institutes of Health. The presenter reported having no financial conflicts of interest.
Click on the PDF icon at the top of this introduction to read the full article.
Split-dose R-CHOP: a new approach to administer cytotoxic chemo-immunotherapy to elderly patients with DLBCL
Background Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma. It is challenging to deliver standard rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) chemotherapy in the very elderly or elderly with comorbidities because of age-related changes in metabolism and performance.
Objectives To describe outcomes of a unique approach to the delivery of standard R-CHOP chemotherapy in split-doses for the treatment of elderly DLBCL patients.
Methods We performed a single center, retrospective analysis of all patients with DLBCL treated with split-dose R-CHOP during January 2007-April 2015. The patients received R-CHOP at a 50% dose reduction on days 1 and 15 of each 28-day cycle (split dose), with full dose rituximab on day 1 for up to 6 cycles. The total amount of chemotherapy delivered during each 28-day cycle of split-dose R-CHOP was equivalent to the cumulative dose in each 21-day cycle of standard R-CHOP.
Results We identified 22 patients who had been treated with split-dose R-CHOP (median age, 81 years). 10 patients had a Charlson Comorbidity Index score of 2 or more, and 13 were aged 80 or older. 12 patients completed their prescribed treatments, and 10 required further de-escalation or early termination owing to toxicity. All of the patients who completed therapy were in a complete remission at the end of treatment. The median overall survival for the entire cohort was 47 months, and median progression-free survival was 43 months.
Limitations Retrospective, single institution study, small cohort Conclusions Split-dose R-CHOP allowed administration of curative-intent therapy in an elderly population with encouraging outcomes.
Conclusions Split-dose R-CHOP allowed administration of curative-intent therapy in an elderly population with encouraging outcomes.
Funding/sponsorship Cancer Center Research Training Program, NCI 5-T32 CA09615-25 (fellowship funding for Dr Shah).
Click on the PDF icon at the top of this introduction to read the full article.
Background Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma. It is challenging to deliver standard rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) chemotherapy in the very elderly or elderly with comorbidities because of age-related changes in metabolism and performance.
Objectives To describe outcomes of a unique approach to the delivery of standard R-CHOP chemotherapy in split-doses for the treatment of elderly DLBCL patients.
Methods We performed a single center, retrospective analysis of all patients with DLBCL treated with split-dose R-CHOP during January 2007-April 2015. The patients received R-CHOP at a 50% dose reduction on days 1 and 15 of each 28-day cycle (split dose), with full dose rituximab on day 1 for up to 6 cycles. The total amount of chemotherapy delivered during each 28-day cycle of split-dose R-CHOP was equivalent to the cumulative dose in each 21-day cycle of standard R-CHOP.
Results We identified 22 patients who had been treated with split-dose R-CHOP (median age, 81 years). 10 patients had a Charlson Comorbidity Index score of 2 or more, and 13 were aged 80 or older. 12 patients completed their prescribed treatments, and 10 required further de-escalation or early termination owing to toxicity. All of the patients who completed therapy were in a complete remission at the end of treatment. The median overall survival for the entire cohort was 47 months, and median progression-free survival was 43 months.
Limitations Retrospective, single institution study, small cohort Conclusions Split-dose R-CHOP allowed administration of curative-intent therapy in an elderly population with encouraging outcomes.
Conclusions Split-dose R-CHOP allowed administration of curative-intent therapy in an elderly population with encouraging outcomes.
Funding/sponsorship Cancer Center Research Training Program, NCI 5-T32 CA09615-25 (fellowship funding for Dr Shah).
Click on the PDF icon at the top of this introduction to read the full article.
Background Diffuse large B-cell lymphoma (DLBCL) is the most common subtype of non-Hodgkin lymphoma. It is challenging to deliver standard rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) chemotherapy in the very elderly or elderly with comorbidities because of age-related changes in metabolism and performance.
Objectives To describe outcomes of a unique approach to the delivery of standard R-CHOP chemotherapy in split-doses for the treatment of elderly DLBCL patients.
Methods We performed a single center, retrospective analysis of all patients with DLBCL treated with split-dose R-CHOP during January 2007-April 2015. The patients received R-CHOP at a 50% dose reduction on days 1 and 15 of each 28-day cycle (split dose), with full dose rituximab on day 1 for up to 6 cycles. The total amount of chemotherapy delivered during each 28-day cycle of split-dose R-CHOP was equivalent to the cumulative dose in each 21-day cycle of standard R-CHOP.
Results We identified 22 patients who had been treated with split-dose R-CHOP (median age, 81 years). 10 patients had a Charlson Comorbidity Index score of 2 or more, and 13 were aged 80 or older. 12 patients completed their prescribed treatments, and 10 required further de-escalation or early termination owing to toxicity. All of the patients who completed therapy were in a complete remission at the end of treatment. The median overall survival for the entire cohort was 47 months, and median progression-free survival was 43 months.
Limitations Retrospective, single institution study, small cohort Conclusions Split-dose R-CHOP allowed administration of curative-intent therapy in an elderly population with encouraging outcomes.
Conclusions Split-dose R-CHOP allowed administration of curative-intent therapy in an elderly population with encouraging outcomes.
Funding/sponsorship Cancer Center Research Training Program, NCI 5-T32 CA09615-25 (fellowship funding for Dr Shah).
Click on the PDF icon at the top of this introduction to read the full article.
Outcome of tumor lysis syndrome in pediatric patients with hematologic malignancies – a single-center experience from Pakistan
Background Tumor lysis syndrome (TLS) is serious complication of anticancer chemotherapy, leading to substantial morbidity and mortality in adults and pediatric patients.
Objective To report the incidence and outcomes of TLS in pediatric patients with hematologic malignancies at a center in Pakistan.
Methods Retrospective chart review of 317 pediatric patients with hematologic malignancies during January 2008-December 2013. Demographic features and clinical and laboratory parameters of TLS, with immediate and 6-month outcomes were determined using a semi-structured questionnaire.
Results Median age at diagnosis was 9 years, with the 79.2% patients being male. Laboratory TLS was present in 36 patients (11.4%), with 27 (8.5%) developing clinical TLS and 13 (4.1%) requiring intensive care support. Hyperphosphatemia was the most frequent metabolic abnormality (14.2%), followed by hypocalcemia (13.9%), hyperuricemia (12.6%), and hyperkalemia (1.3%). 45 patients (14.2%) developed acute kidney injury (AKI). Patients who developed TLS had a signficantly higher white blood cell count at initiation of chemotherapy (142.0 x 109/L [SD, 173.1] vs 31.5 x 109/L [SD, 58.0]; P = .01) and a higher incidence of AKI (58.3% vs 8.5% of patients; P < .001).
Limitations Retrospective design of study, high rate of loss to follow-up, and unavailability of lactate dehydrogenase levels in a majority of patients.
Conclusion The incidence of TLS pediatric hematologic malignancies was 11.4% at our center. The main cause of death was sepsis. Hyperphosphatemia was the common metabolic derangement and hyperkalemia was the least common. TLS warrants intensive supportive care to prevent further morbidity and decrease mortality.
Click on the PDF icon at the top of this introduction to read the full article.
Background Tumor lysis syndrome (TLS) is serious complication of anticancer chemotherapy, leading to substantial morbidity and mortality in adults and pediatric patients.
Objective To report the incidence and outcomes of TLS in pediatric patients with hematologic malignancies at a center in Pakistan.
Methods Retrospective chart review of 317 pediatric patients with hematologic malignancies during January 2008-December 2013. Demographic features and clinical and laboratory parameters of TLS, with immediate and 6-month outcomes were determined using a semi-structured questionnaire.
Results Median age at diagnosis was 9 years, with the 79.2% patients being male. Laboratory TLS was present in 36 patients (11.4%), with 27 (8.5%) developing clinical TLS and 13 (4.1%) requiring intensive care support. Hyperphosphatemia was the most frequent metabolic abnormality (14.2%), followed by hypocalcemia (13.9%), hyperuricemia (12.6%), and hyperkalemia (1.3%). 45 patients (14.2%) developed acute kidney injury (AKI). Patients who developed TLS had a signficantly higher white blood cell count at initiation of chemotherapy (142.0 x 109/L [SD, 173.1] vs 31.5 x 109/L [SD, 58.0]; P = .01) and a higher incidence of AKI (58.3% vs 8.5% of patients; P < .001).
Limitations Retrospective design of study, high rate of loss to follow-up, and unavailability of lactate dehydrogenase levels in a majority of patients.
Conclusion The incidence of TLS pediatric hematologic malignancies was 11.4% at our center. The main cause of death was sepsis. Hyperphosphatemia was the common metabolic derangement and hyperkalemia was the least common. TLS warrants intensive supportive care to prevent further morbidity and decrease mortality.
Click on the PDF icon at the top of this introduction to read the full article.
Background Tumor lysis syndrome (TLS) is serious complication of anticancer chemotherapy, leading to substantial morbidity and mortality in adults and pediatric patients.
Objective To report the incidence and outcomes of TLS in pediatric patients with hematologic malignancies at a center in Pakistan.
Methods Retrospective chart review of 317 pediatric patients with hematologic malignancies during January 2008-December 2013. Demographic features and clinical and laboratory parameters of TLS, with immediate and 6-month outcomes were determined using a semi-structured questionnaire.
Results Median age at diagnosis was 9 years, with the 79.2% patients being male. Laboratory TLS was present in 36 patients (11.4%), with 27 (8.5%) developing clinical TLS and 13 (4.1%) requiring intensive care support. Hyperphosphatemia was the most frequent metabolic abnormality (14.2%), followed by hypocalcemia (13.9%), hyperuricemia (12.6%), and hyperkalemia (1.3%). 45 patients (14.2%) developed acute kidney injury (AKI). Patients who developed TLS had a signficantly higher white blood cell count at initiation of chemotherapy (142.0 x 109/L [SD, 173.1] vs 31.5 x 109/L [SD, 58.0]; P = .01) and a higher incidence of AKI (58.3% vs 8.5% of patients; P < .001).
Limitations Retrospective design of study, high rate of loss to follow-up, and unavailability of lactate dehydrogenase levels in a majority of patients.
Conclusion The incidence of TLS pediatric hematologic malignancies was 11.4% at our center. The main cause of death was sepsis. Hyperphosphatemia was the common metabolic derangement and hyperkalemia was the least common. TLS warrants intensive supportive care to prevent further morbidity and decrease mortality.
Click on the PDF icon at the top of this introduction to read the full article.