User login
Transplantation plus VRD tops VRD alone in myeloma
Adding high-dose chemotherapy and autologous stem cell transplantation to two cycles of bortezomib, lenalidomide, and dexamethasone (VRD) significantly improved progression-free survival in adults with newly diagnosed multiple myeloma, according to new analyses from the multicenter, randomized, phase III IFM 2009 Study.
Median PFS was 50 months in the transplantation group, versus 36 months when patients only received five cycles of VRD for consolidation (adjusted hazard ratio for disease progression or death, 0.65; 95% confidence interval, 0.53 to 0.80; P less than .001), reported Michel Attal, MD, of Institut Universitaire du Cancer de Toulouse-Oncopole in Toulouse, France, and his associates. “Transplantation was also associated with a higher rate of complete response, a lower rate of minimal residual disease detection, and a longer median time to progression,” they wrote (N Engl J Med. 2017 April 6. doi: 10.1056/NEJMoa1611750).
Transplantation did not improve 4-year overall survival (81% with transplantation plus VRD and 82% with VRD only), perhaps because VRD-only patients had the option of salvage transplantation if their disease progressed, the investigators said. In addition, grade 3 or 4 neutropenias were significantly more common with transplantation than otherwise (92% versus 47%), as were grade 3 or 4 gastrointestinal disorders (28% versus 7%) and infections (20% versus 9%). However, the groups did not significantly differ in terms of treatment-related deaths, second primary cancers, thromboembolic events, or peripheral neuropathy.
Before the advent of immunomodulatory drugs and proteasome inhibitors, several randomized trials showed that the benefits of high-dose chemotherapy with autologous stem cell transplantation outperformed conventional chemotherapy in multiple myeloma. For IFM 2009, 700 newly diagnosed patients aged up to 65 years received three 21-day cycles of VRD induction: bortezomib 1.3 mg/m2 on days 1, 4, 8, and 11; lenalidomide 25 mg on days 1-14; and dexamethasone 20 mg on days 1, 2, 4, 5, 8, 9, 11, and 12. Consolidation consisted of either five more VRD cycles with dexamethasone reduced to 10 mg/day, or high-dose (200 mg/m2) melphalan plus transplantation followed by two VRD cycles with the reduced dexamethasone dose. Both arms received 1 year of lenalidomide maintenance (10 mg/day for 3 months, followed by 15/mg day, depending on side effects).
Transplantation was associated with a significantly higher likelihood of complete response (59% versus 48%; P = .03) and undetectable minimal residual disease (79% versus 65%; P less than .001). Undetectable minimal residual disease, in turn, predicted longer PFS (adjusted hazard ratio, 0.30; P = .001). In contrast, age, sex, isotype of the monoclonal component, International Staging System disease stage, and cytogenetic risk profile did not significantly affect PFS among transplant patients.
The PFS benefit of transplantation “must be weighed against the increased risk of toxic effects associated with high-dose chemotherapy plus transplantation, especially since we found that later transplantation might be as effective as early transplantation in securing long- term survival,” the researchers said. “Our results suggest that the use of a combination therapy that incorporates newer proteasome inhibitors, next-generation immunomodulatory drugs, and potent monoclonal antibodies along with transplantation tailored according to minimal residual disease detection could further improve outcomes among adults up to 65 years of age who have multiple myeloma.”
Celgene, Janssen, the French Ministry of Health Programme Hospitalier de Recherche Clinique, and the French National Research Agency funded the study. Dr. Attal had no conflicts. Nine coinvestigators disclosed ties to Celgene, Janssen, and several other pharmaceutical companies. Two coinvestigators disclosed ownership or founder roles in OncoPep, one of whom disclosed patent licensure to OncoPep for a multipeptide vaccine. The other investigators had no conflicts.
In the IFM 2009 trial, the planned treatments were realistic. The combination of immunomodulatory drugs and proteasome inhibitors is associated with high response rates similar to those achieved with transplantation, whereas previous trials had used less effective chemotherapy regimens as comparators.
[Transplantation] resulted in a deeper and longer initial treatment response than did nontransplantation. However, the benefits of transplantation were more modest than some might have hoped, and it did not appear to be curative.
A question remains as to whether all patients with multiple myeloma should undergo immediate autologous stem-cell transplantation, since the use of delayed transplantation resulted in similar overall survival, with some patients not needing transplantation to date. The observed lack of a survival benefit for early transplantation was consistent with previous results that suggested salvage transplantation is an equalizer in this regard.
Charles A. Schiffer, MD, and Jeffrey A. Zonder, MD, are with Karmanos Cancer Institute, Wayne State University, Detroit, Mich. Dr. Zonder is a member of the editorial advisory board of Hematology News. Their comments were taken from an editorial (N Engl J Med. 2017 April 6. doi: 10.1056/NEJMe1700453).
In the IFM 2009 trial, the planned treatments were realistic. The combination of immunomodulatory drugs and proteasome inhibitors is associated with high response rates similar to those achieved with transplantation, whereas previous trials had used less effective chemotherapy regimens as comparators.
[Transplantation] resulted in a deeper and longer initial treatment response than did nontransplantation. However, the benefits of transplantation were more modest than some might have hoped, and it did not appear to be curative.
A question remains as to whether all patients with multiple myeloma should undergo immediate autologous stem-cell transplantation, since the use of delayed transplantation resulted in similar overall survival, with some patients not needing transplantation to date. The observed lack of a survival benefit for early transplantation was consistent with previous results that suggested salvage transplantation is an equalizer in this regard.
Charles A. Schiffer, MD, and Jeffrey A. Zonder, MD, are with Karmanos Cancer Institute, Wayne State University, Detroit, Mich. Dr. Zonder is a member of the editorial advisory board of Hematology News. Their comments were taken from an editorial (N Engl J Med. 2017 April 6. doi: 10.1056/NEJMe1700453).
In the IFM 2009 trial, the planned treatments were realistic. The combination of immunomodulatory drugs and proteasome inhibitors is associated with high response rates similar to those achieved with transplantation, whereas previous trials had used less effective chemotherapy regimens as comparators.
[Transplantation] resulted in a deeper and longer initial treatment response than did nontransplantation. However, the benefits of transplantation were more modest than some might have hoped, and it did not appear to be curative.
A question remains as to whether all patients with multiple myeloma should undergo immediate autologous stem-cell transplantation, since the use of delayed transplantation resulted in similar overall survival, with some patients not needing transplantation to date. The observed lack of a survival benefit for early transplantation was consistent with previous results that suggested salvage transplantation is an equalizer in this regard.
Charles A. Schiffer, MD, and Jeffrey A. Zonder, MD, are with Karmanos Cancer Institute, Wayne State University, Detroit, Mich. Dr. Zonder is a member of the editorial advisory board of Hematology News. Their comments were taken from an editorial (N Engl J Med. 2017 April 6. doi: 10.1056/NEJMe1700453).
Adding high-dose chemotherapy and autologous stem cell transplantation to two cycles of bortezomib, lenalidomide, and dexamethasone (VRD) significantly improved progression-free survival in adults with newly diagnosed multiple myeloma, according to new analyses from the multicenter, randomized, phase III IFM 2009 Study.
Median PFS was 50 months in the transplantation group, versus 36 months when patients only received five cycles of VRD for consolidation (adjusted hazard ratio for disease progression or death, 0.65; 95% confidence interval, 0.53 to 0.80; P less than .001), reported Michel Attal, MD, of Institut Universitaire du Cancer de Toulouse-Oncopole in Toulouse, France, and his associates. “Transplantation was also associated with a higher rate of complete response, a lower rate of minimal residual disease detection, and a longer median time to progression,” they wrote (N Engl J Med. 2017 April 6. doi: 10.1056/NEJMoa1611750).
Transplantation did not improve 4-year overall survival (81% with transplantation plus VRD and 82% with VRD only), perhaps because VRD-only patients had the option of salvage transplantation if their disease progressed, the investigators said. In addition, grade 3 or 4 neutropenias were significantly more common with transplantation than otherwise (92% versus 47%), as were grade 3 or 4 gastrointestinal disorders (28% versus 7%) and infections (20% versus 9%). However, the groups did not significantly differ in terms of treatment-related deaths, second primary cancers, thromboembolic events, or peripheral neuropathy.
Before the advent of immunomodulatory drugs and proteasome inhibitors, several randomized trials showed that the benefits of high-dose chemotherapy with autologous stem cell transplantation outperformed conventional chemotherapy in multiple myeloma. For IFM 2009, 700 newly diagnosed patients aged up to 65 years received three 21-day cycles of VRD induction: bortezomib 1.3 mg/m2 on days 1, 4, 8, and 11; lenalidomide 25 mg on days 1-14; and dexamethasone 20 mg on days 1, 2, 4, 5, 8, 9, 11, and 12. Consolidation consisted of either five more VRD cycles with dexamethasone reduced to 10 mg/day, or high-dose (200 mg/m2) melphalan plus transplantation followed by two VRD cycles with the reduced dexamethasone dose. Both arms received 1 year of lenalidomide maintenance (10 mg/day for 3 months, followed by 15/mg day, depending on side effects).
Transplantation was associated with a significantly higher likelihood of complete response (59% versus 48%; P = .03) and undetectable minimal residual disease (79% versus 65%; P less than .001). Undetectable minimal residual disease, in turn, predicted longer PFS (adjusted hazard ratio, 0.30; P = .001). In contrast, age, sex, isotype of the monoclonal component, International Staging System disease stage, and cytogenetic risk profile did not significantly affect PFS among transplant patients.
The PFS benefit of transplantation “must be weighed against the increased risk of toxic effects associated with high-dose chemotherapy plus transplantation, especially since we found that later transplantation might be as effective as early transplantation in securing long- term survival,” the researchers said. “Our results suggest that the use of a combination therapy that incorporates newer proteasome inhibitors, next-generation immunomodulatory drugs, and potent monoclonal antibodies along with transplantation tailored according to minimal residual disease detection could further improve outcomes among adults up to 65 years of age who have multiple myeloma.”
Celgene, Janssen, the French Ministry of Health Programme Hospitalier de Recherche Clinique, and the French National Research Agency funded the study. Dr. Attal had no conflicts. Nine coinvestigators disclosed ties to Celgene, Janssen, and several other pharmaceutical companies. Two coinvestigators disclosed ownership or founder roles in OncoPep, one of whom disclosed patent licensure to OncoPep for a multipeptide vaccine. The other investigators had no conflicts.
Adding high-dose chemotherapy and autologous stem cell transplantation to two cycles of bortezomib, lenalidomide, and dexamethasone (VRD) significantly improved progression-free survival in adults with newly diagnosed multiple myeloma, according to new analyses from the multicenter, randomized, phase III IFM 2009 Study.
Median PFS was 50 months in the transplantation group, versus 36 months when patients only received five cycles of VRD for consolidation (adjusted hazard ratio for disease progression or death, 0.65; 95% confidence interval, 0.53 to 0.80; P less than .001), reported Michel Attal, MD, of Institut Universitaire du Cancer de Toulouse-Oncopole in Toulouse, France, and his associates. “Transplantation was also associated with a higher rate of complete response, a lower rate of minimal residual disease detection, and a longer median time to progression,” they wrote (N Engl J Med. 2017 April 6. doi: 10.1056/NEJMoa1611750).
Transplantation did not improve 4-year overall survival (81% with transplantation plus VRD and 82% with VRD only), perhaps because VRD-only patients had the option of salvage transplantation if their disease progressed, the investigators said. In addition, grade 3 or 4 neutropenias were significantly more common with transplantation than otherwise (92% versus 47%), as were grade 3 or 4 gastrointestinal disorders (28% versus 7%) and infections (20% versus 9%). However, the groups did not significantly differ in terms of treatment-related deaths, second primary cancers, thromboembolic events, or peripheral neuropathy.
Before the advent of immunomodulatory drugs and proteasome inhibitors, several randomized trials showed that the benefits of high-dose chemotherapy with autologous stem cell transplantation outperformed conventional chemotherapy in multiple myeloma. For IFM 2009, 700 newly diagnosed patients aged up to 65 years received three 21-day cycles of VRD induction: bortezomib 1.3 mg/m2 on days 1, 4, 8, and 11; lenalidomide 25 mg on days 1-14; and dexamethasone 20 mg on days 1, 2, 4, 5, 8, 9, 11, and 12. Consolidation consisted of either five more VRD cycles with dexamethasone reduced to 10 mg/day, or high-dose (200 mg/m2) melphalan plus transplantation followed by two VRD cycles with the reduced dexamethasone dose. Both arms received 1 year of lenalidomide maintenance (10 mg/day for 3 months, followed by 15/mg day, depending on side effects).
Transplantation was associated with a significantly higher likelihood of complete response (59% versus 48%; P = .03) and undetectable minimal residual disease (79% versus 65%; P less than .001). Undetectable minimal residual disease, in turn, predicted longer PFS (adjusted hazard ratio, 0.30; P = .001). In contrast, age, sex, isotype of the monoclonal component, International Staging System disease stage, and cytogenetic risk profile did not significantly affect PFS among transplant patients.
The PFS benefit of transplantation “must be weighed against the increased risk of toxic effects associated with high-dose chemotherapy plus transplantation, especially since we found that later transplantation might be as effective as early transplantation in securing long- term survival,” the researchers said. “Our results suggest that the use of a combination therapy that incorporates newer proteasome inhibitors, next-generation immunomodulatory drugs, and potent monoclonal antibodies along with transplantation tailored according to minimal residual disease detection could further improve outcomes among adults up to 65 years of age who have multiple myeloma.”
Celgene, Janssen, the French Ministry of Health Programme Hospitalier de Recherche Clinique, and the French National Research Agency funded the study. Dr. Attal had no conflicts. Nine coinvestigators disclosed ties to Celgene, Janssen, and several other pharmaceutical companies. Two coinvestigators disclosed ownership or founder roles in OncoPep, one of whom disclosed patent licensure to OncoPep for a multipeptide vaccine. The other investigators had no conflicts.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: High-dose chemotherapy and autologous stem cell transplantation plus bortezomib, lenalidomide, and dexamethasone (VRD) outperformed consolidation with VRD only in adults with newly diagnosed multiple myeloma.
Major finding: Median progression-free survival was 50 months with transplantation plus VRD and 36 months with VRD only (adjusted hazard ratio for disease progression or death, 0.65; P less than .001).
Data source: A randomized, multicenter, open-label, phase III trial of 700 patients with multiple myeloma.
Disclosures: Celgene, Janssen, the French Ministry of Health Programme Hospitalier de Recherche Clinique, and the French National Research Agency funded the study. Dr. Attal had no conflicts. Nine coinvestigators disclosed ties to Celgene, Janssen, and several other pharmaceutical companies. Two coinvestigators disclosed ownership or founder roles in OncoPep, one of whom disclosed patent licensure to OncoPep for a multipeptide vaccine. The other investigators had no conflicts.
Inhibitor produces durable responses in rel/ref iNHL
WASHINGTON, DC—An investigational drug can produce durable responses and has a manageable safety profile in patients with relapsed or refractory indolent non-Hodgkin lymphoma (iNHL), according to researchers.
The drug is copanlisib, an intravenous pan-class I phosphatidylinositol-3-kinase (PI3K) inhibitor.
In the phase 2 CHRONOS-1 trial, copanlisib produced an objective response rate (ORR) of 59.2%, with a complete response (CR) rate of 12%, in patients with relapsed/refractory iNHL.
The median duration of response exceeded 98 weeks.
There were 3 deaths considered related to copanlisib, and the most common treatment-related adverse events (AEs) were transient hyperglycemia and hypertension.
These results were presented at the AACR Annual Meeting 2017 (abstract CT149). The study is supported by Bayer, the company developing copanlisib.
CHRONOS-1 included 141 patients with iNHL. Most (n=104) had follicular lymphoma (FL), 23 had marginal zone lymphoma (MZL), 8 had small lymphocytic lymphoma, and 6 had lymphoplasmacytoid/Waldenstrӧm’s macroglobulinemia.
All patients had relapsed after or were refractory to at least 2 prior lines of therapy, which included both rituximab and an alkylating agent.
For this study, the patients received 60 mg of intravenous copanlisib intermittently on days 1, 8, and 15 of a 28-day cycle.
At the time of analysis, the median duration of treatment was 22 weeks (range, 1-105), and 46 patients were still receiving copanlisib.
Efficacy
For the entire cohort, the ORR was 59.2%. Twelve percent of patients achieved a CR, 47.2% had a partial response (PR), 29.6% had stable disease, and 2.1% had progressive disease.
Among patients with FL, the ORR was 58.7%, the CR rate was 14.4%, and the PR rate was 44.2%.
Among patients with MZL, the ORR was 69.6%, with 8.7% of patients achieving a CR and 60.9% achieving a PR.
For the entire cohort, the estimated median duration of response was 687 days (range, 0-687). For patients with FL, it was 370 days (range, 0-687).
The estimated median progression-free survival was 340 days (range, 0-736), and the median overall survival had not been reached at the time of analysis.
Safety
The most common treatment-related AEs were transient hyperglycemia (all grades, 49%/grade 3+, 40%) and hypertension (all grades, 29%/grade 3+, 23%).
The researchers said other AEs of interest were neutropenia (all grades, 25%/grade 3+, 19%), diarrhea (all grades, 18%/grade 3+, 4%), lung infection (all grades, 14%/grade 3+, 11%), pneumonitis (all grades, 7%/grade 3+, 1.4%), and colitis (0.7%, all grade 3+).
Laboratory AEs of interest were alanine aminotransferase increase (all grades, 23%/grade 1, 19%) and aspartate aminotransferase increase (all grades, 28%/grade 1, 25%).
There were 2 non-fatal opportunistic infections.
There were 6 deaths, and 3 of them were considered related to copanlisib. These 3 deaths were due to lung infection, respiratory failure, and a thromboembolic event.
“[I]nhibition of the PI3K pathway has been shown to be an effective therapeutic strategy in treating indolent lymphomas . . .,” said study investigator Martin Dreyling, MD, of Klinikum der Universität München-Grosshadern in Munich, Germany.
“However, concerns exist about the safety of available oral PI3K inhibitors . . . . The results of CHRONOS-1 demonstrate that intermittent intravenous administration of copanlisib achieved durable efficacy with a manageable safety profile in this difficult-to-treat patient population.” ![]()
WASHINGTON, DC—An investigational drug can produce durable responses and has a manageable safety profile in patients with relapsed or refractory indolent non-Hodgkin lymphoma (iNHL), according to researchers.
The drug is copanlisib, an intravenous pan-class I phosphatidylinositol-3-kinase (PI3K) inhibitor.
In the phase 2 CHRONOS-1 trial, copanlisib produced an objective response rate (ORR) of 59.2%, with a complete response (CR) rate of 12%, in patients with relapsed/refractory iNHL.
The median duration of response exceeded 98 weeks.
There were 3 deaths considered related to copanlisib, and the most common treatment-related adverse events (AEs) were transient hyperglycemia and hypertension.
These results were presented at the AACR Annual Meeting 2017 (abstract CT149). The study is supported by Bayer, the company developing copanlisib.
CHRONOS-1 included 141 patients with iNHL. Most (n=104) had follicular lymphoma (FL), 23 had marginal zone lymphoma (MZL), 8 had small lymphocytic lymphoma, and 6 had lymphoplasmacytoid/Waldenstrӧm’s macroglobulinemia.
All patients had relapsed after or were refractory to at least 2 prior lines of therapy, which included both rituximab and an alkylating agent.
For this study, the patients received 60 mg of intravenous copanlisib intermittently on days 1, 8, and 15 of a 28-day cycle.
At the time of analysis, the median duration of treatment was 22 weeks (range, 1-105), and 46 patients were still receiving copanlisib.
Efficacy
For the entire cohort, the ORR was 59.2%. Twelve percent of patients achieved a CR, 47.2% had a partial response (PR), 29.6% had stable disease, and 2.1% had progressive disease.
Among patients with FL, the ORR was 58.7%, the CR rate was 14.4%, and the PR rate was 44.2%.
Among patients with MZL, the ORR was 69.6%, with 8.7% of patients achieving a CR and 60.9% achieving a PR.
For the entire cohort, the estimated median duration of response was 687 days (range, 0-687). For patients with FL, it was 370 days (range, 0-687).
The estimated median progression-free survival was 340 days (range, 0-736), and the median overall survival had not been reached at the time of analysis.
Safety
The most common treatment-related AEs were transient hyperglycemia (all grades, 49%/grade 3+, 40%) and hypertension (all grades, 29%/grade 3+, 23%).
The researchers said other AEs of interest were neutropenia (all grades, 25%/grade 3+, 19%), diarrhea (all grades, 18%/grade 3+, 4%), lung infection (all grades, 14%/grade 3+, 11%), pneumonitis (all grades, 7%/grade 3+, 1.4%), and colitis (0.7%, all grade 3+).
Laboratory AEs of interest were alanine aminotransferase increase (all grades, 23%/grade 1, 19%) and aspartate aminotransferase increase (all grades, 28%/grade 1, 25%).
There were 2 non-fatal opportunistic infections.
There were 6 deaths, and 3 of them were considered related to copanlisib. These 3 deaths were due to lung infection, respiratory failure, and a thromboembolic event.
“[I]nhibition of the PI3K pathway has been shown to be an effective therapeutic strategy in treating indolent lymphomas . . .,” said study investigator Martin Dreyling, MD, of Klinikum der Universität München-Grosshadern in Munich, Germany.
“However, concerns exist about the safety of available oral PI3K inhibitors . . . . The results of CHRONOS-1 demonstrate that intermittent intravenous administration of copanlisib achieved durable efficacy with a manageable safety profile in this difficult-to-treat patient population.” ![]()
WASHINGTON, DC—An investigational drug can produce durable responses and has a manageable safety profile in patients with relapsed or refractory indolent non-Hodgkin lymphoma (iNHL), according to researchers.
The drug is copanlisib, an intravenous pan-class I phosphatidylinositol-3-kinase (PI3K) inhibitor.
In the phase 2 CHRONOS-1 trial, copanlisib produced an objective response rate (ORR) of 59.2%, with a complete response (CR) rate of 12%, in patients with relapsed/refractory iNHL.
The median duration of response exceeded 98 weeks.
There were 3 deaths considered related to copanlisib, and the most common treatment-related adverse events (AEs) were transient hyperglycemia and hypertension.
These results were presented at the AACR Annual Meeting 2017 (abstract CT149). The study is supported by Bayer, the company developing copanlisib.
CHRONOS-1 included 141 patients with iNHL. Most (n=104) had follicular lymphoma (FL), 23 had marginal zone lymphoma (MZL), 8 had small lymphocytic lymphoma, and 6 had lymphoplasmacytoid/Waldenstrӧm’s macroglobulinemia.
All patients had relapsed after or were refractory to at least 2 prior lines of therapy, which included both rituximab and an alkylating agent.
For this study, the patients received 60 mg of intravenous copanlisib intermittently on days 1, 8, and 15 of a 28-day cycle.
At the time of analysis, the median duration of treatment was 22 weeks (range, 1-105), and 46 patients were still receiving copanlisib.
Efficacy
For the entire cohort, the ORR was 59.2%. Twelve percent of patients achieved a CR, 47.2% had a partial response (PR), 29.6% had stable disease, and 2.1% had progressive disease.
Among patients with FL, the ORR was 58.7%, the CR rate was 14.4%, and the PR rate was 44.2%.
Among patients with MZL, the ORR was 69.6%, with 8.7% of patients achieving a CR and 60.9% achieving a PR.
For the entire cohort, the estimated median duration of response was 687 days (range, 0-687). For patients with FL, it was 370 days (range, 0-687).
The estimated median progression-free survival was 340 days (range, 0-736), and the median overall survival had not been reached at the time of analysis.
Safety
The most common treatment-related AEs were transient hyperglycemia (all grades, 49%/grade 3+, 40%) and hypertension (all grades, 29%/grade 3+, 23%).
The researchers said other AEs of interest were neutropenia (all grades, 25%/grade 3+, 19%), diarrhea (all grades, 18%/grade 3+, 4%), lung infection (all grades, 14%/grade 3+, 11%), pneumonitis (all grades, 7%/grade 3+, 1.4%), and colitis (0.7%, all grade 3+).
Laboratory AEs of interest were alanine aminotransferase increase (all grades, 23%/grade 1, 19%) and aspartate aminotransferase increase (all grades, 28%/grade 1, 25%).
There were 2 non-fatal opportunistic infections.
There were 6 deaths, and 3 of them were considered related to copanlisib. These 3 deaths were due to lung infection, respiratory failure, and a thromboembolic event.
“[I]nhibition of the PI3K pathway has been shown to be an effective therapeutic strategy in treating indolent lymphomas . . .,” said study investigator Martin Dreyling, MD, of Klinikum der Universität München-Grosshadern in Munich, Germany.
“However, concerns exist about the safety of available oral PI3K inhibitors . . . . The results of CHRONOS-1 demonstrate that intermittent intravenous administration of copanlisib achieved durable efficacy with a manageable safety profile in this difficult-to-treat patient population.” ![]()
Half of patients retain response to CAR T-cell therapy
WASHINGTON, DC—Roughly half of patients who responded to chimeric antigen receptor (CAR) T-cell therapy in the ZUMA-1 trial have retained that response at a median follow-up exceeding 8 months.
The CAR T-cell therapy, axicabtagene ciloleucel (formerly KTE-C19), initially produced an objective response rate (ORR) of 82% in this trial of patients with relapsed/refractory B-cell non-Hodgkin lymphoma (NHL).
At a median follow-up of 8.7 months, 44% of all patients (53% of responders) are still in response, and 39% are in complete response (CR).
Thirteen percent of patients had grade 3 or higher cytokine release syndrome (CRS), and 28% had neurologic events.
There were 2 deaths related to axicabtagene ciloleucel.
Frederick L. Locke, MD, of Moffitt Cancer Center in Tampa, Florida, presented these updated results from ZUMA-1 at the AACR Annual Meeting 2017 (abstract CT019).
ZUMA-1 is sponsored by Kite Pharma but is also funded, in part, by the Leukemia and Lymphoma Society Therapy Acceleration Program.
Patients and treatment
The trial enrolled 111 patients, 101 of whom were successfully treated with axicabtagene ciloleucel. Seven patients could not be treated due to serious adverse events, 1 due to unavailable product, and 2 due to non-measurable disease.
Seventy-seven of the patients had diffuse large B-cell lymphoma (DLBCL), and 24 had transformed follicular lymphoma (TFL) or primary mediastinal B-cell lymphoma (PMBCL). Eighty-five percent of patients had stage III/IV disease.
Seventy-nine percent were refractory to chemotherapy and did not have a prior autologous stem cell transplant (auto-SCT). Twenty-one percent did undergo auto-SCT and relapsed within 12 months of the procedure.
Sixty-nine percent of patients had received 3 or more lines of prior therapy, and 54% were refractory to 2 consecutive lines of prior therapy.
For this study, the patients received a conditioning regimen of cyclophosphamide (500 mg/m2) and fludarabine (30 mg/m2) for 3 days.
Two days after the conditioning regimen was completed, patients received axicabtagene ciloleucel at a target dose of 2 × 106 CAR T cells/kg.
Efficacy
The following table shows overall response data, response data at 6 months, and ongoing responses at the primary analysis data cut-off.
| DLBCL (n=77) | TFL/PMBCL (n=24) | Combined (n=101) | ||||
| ORR (%) | CR (%) | ORR (%) | CR (%) | ORR (%) | CR (%) | |
| ORR | 82 | 49 | 83 | 71 | 82 | 54 |
| Month 6 | 36 | 31 | 54 | 50 | 41 | 36 |
| Ongoing | 36 | 31 | 67 | 63 | 44 | 39 |
The researchers said the ORR was generally consistent in key subgroups. The ORR was 83% in patients who were refractory to their second or greater line of therapy and 76% in patients who relapsed within 12 months of auto-SCT.
Overall, the median duration of response was 8.2 months. However, the median duration of response has not been reached for patients with a CR.
At a median follow-up of 8.7 months, the median overall survival has not been reached.
Safety
The most common grade 3 or higher adverse events included anemia (43%), neutropenia (39%), decreased neutrophil count (32%), febrile neutropenia (31%), decreased white blood cell count (29%), thrombocytopenia (24%), encephalopathy (21%), and decreased lymphocyte count (20%).
The incidence of grade 3 or higher CRS was 13%, and the incidence of neurologic events was 28%. These represent decreases from the interim analysis of ZUMA-1, when the rate of grade 3+ CRS was 18%, and the rate of neurological events was 34%.
“We believe the rates of CRS and neurologic events decreased over the course of the study as clinicians gained experience in the management of adverse events,” said Jeff Wiezorek, MD, senior vice-president of clinical development at Kite Pharma.
There were 3 deaths throughout the course of the trial that were not a result of disease progression.
Two deaths were deemed related to axicabtagene ciloleucel. One was a case of hemophagocytic lymphohistiocytosis. The other was cardiac arrest in the setting of CRS.
The third death was the result of a pulmonary embolism and was considered unrelated to axicabtagene ciloleucel. ![]()
WASHINGTON, DC—Roughly half of patients who responded to chimeric antigen receptor (CAR) T-cell therapy in the ZUMA-1 trial have retained that response at a median follow-up exceeding 8 months.
The CAR T-cell therapy, axicabtagene ciloleucel (formerly KTE-C19), initially produced an objective response rate (ORR) of 82% in this trial of patients with relapsed/refractory B-cell non-Hodgkin lymphoma (NHL).
At a median follow-up of 8.7 months, 44% of all patients (53% of responders) are still in response, and 39% are in complete response (CR).
Thirteen percent of patients had grade 3 or higher cytokine release syndrome (CRS), and 28% had neurologic events.
There were 2 deaths related to axicabtagene ciloleucel.
Frederick L. Locke, MD, of Moffitt Cancer Center in Tampa, Florida, presented these updated results from ZUMA-1 at the AACR Annual Meeting 2017 (abstract CT019).
ZUMA-1 is sponsored by Kite Pharma but is also funded, in part, by the Leukemia and Lymphoma Society Therapy Acceleration Program.
Patients and treatment
The trial enrolled 111 patients, 101 of whom were successfully treated with axicabtagene ciloleucel. Seven patients could not be treated due to serious adverse events, 1 due to unavailable product, and 2 due to non-measurable disease.
Seventy-seven of the patients had diffuse large B-cell lymphoma (DLBCL), and 24 had transformed follicular lymphoma (TFL) or primary mediastinal B-cell lymphoma (PMBCL). Eighty-five percent of patients had stage III/IV disease.
Seventy-nine percent were refractory to chemotherapy and did not have a prior autologous stem cell transplant (auto-SCT). Twenty-one percent did undergo auto-SCT and relapsed within 12 months of the procedure.
Sixty-nine percent of patients had received 3 or more lines of prior therapy, and 54% were refractory to 2 consecutive lines of prior therapy.
For this study, the patients received a conditioning regimen of cyclophosphamide (500 mg/m2) and fludarabine (30 mg/m2) for 3 days.
Two days after the conditioning regimen was completed, patients received axicabtagene ciloleucel at a target dose of 2 × 106 CAR T cells/kg.
Efficacy
The following table shows overall response data, response data at 6 months, and ongoing responses at the primary analysis data cut-off.
| DLBCL (n=77) | TFL/PMBCL (n=24) | Combined (n=101) | ||||
| ORR (%) | CR (%) | ORR (%) | CR (%) | ORR (%) | CR (%) | |
| ORR | 82 | 49 | 83 | 71 | 82 | 54 |
| Month 6 | 36 | 31 | 54 | 50 | 41 | 36 |
| Ongoing | 36 | 31 | 67 | 63 | 44 | 39 |
The researchers said the ORR was generally consistent in key subgroups. The ORR was 83% in patients who were refractory to their second or greater line of therapy and 76% in patients who relapsed within 12 months of auto-SCT.
Overall, the median duration of response was 8.2 months. However, the median duration of response has not been reached for patients with a CR.
At a median follow-up of 8.7 months, the median overall survival has not been reached.
Safety
The most common grade 3 or higher adverse events included anemia (43%), neutropenia (39%), decreased neutrophil count (32%), febrile neutropenia (31%), decreased white blood cell count (29%), thrombocytopenia (24%), encephalopathy (21%), and decreased lymphocyte count (20%).
The incidence of grade 3 or higher CRS was 13%, and the incidence of neurologic events was 28%. These represent decreases from the interim analysis of ZUMA-1, when the rate of grade 3+ CRS was 18%, and the rate of neurological events was 34%.
“We believe the rates of CRS and neurologic events decreased over the course of the study as clinicians gained experience in the management of adverse events,” said Jeff Wiezorek, MD, senior vice-president of clinical development at Kite Pharma.
There were 3 deaths throughout the course of the trial that were not a result of disease progression.
Two deaths were deemed related to axicabtagene ciloleucel. One was a case of hemophagocytic lymphohistiocytosis. The other was cardiac arrest in the setting of CRS.
The third death was the result of a pulmonary embolism and was considered unrelated to axicabtagene ciloleucel. ![]()
WASHINGTON, DC—Roughly half of patients who responded to chimeric antigen receptor (CAR) T-cell therapy in the ZUMA-1 trial have retained that response at a median follow-up exceeding 8 months.
The CAR T-cell therapy, axicabtagene ciloleucel (formerly KTE-C19), initially produced an objective response rate (ORR) of 82% in this trial of patients with relapsed/refractory B-cell non-Hodgkin lymphoma (NHL).
At a median follow-up of 8.7 months, 44% of all patients (53% of responders) are still in response, and 39% are in complete response (CR).
Thirteen percent of patients had grade 3 or higher cytokine release syndrome (CRS), and 28% had neurologic events.
There were 2 deaths related to axicabtagene ciloleucel.
Frederick L. Locke, MD, of Moffitt Cancer Center in Tampa, Florida, presented these updated results from ZUMA-1 at the AACR Annual Meeting 2017 (abstract CT019).
ZUMA-1 is sponsored by Kite Pharma but is also funded, in part, by the Leukemia and Lymphoma Society Therapy Acceleration Program.
Patients and treatment
The trial enrolled 111 patients, 101 of whom were successfully treated with axicabtagene ciloleucel. Seven patients could not be treated due to serious adverse events, 1 due to unavailable product, and 2 due to non-measurable disease.
Seventy-seven of the patients had diffuse large B-cell lymphoma (DLBCL), and 24 had transformed follicular lymphoma (TFL) or primary mediastinal B-cell lymphoma (PMBCL). Eighty-five percent of patients had stage III/IV disease.
Seventy-nine percent were refractory to chemotherapy and did not have a prior autologous stem cell transplant (auto-SCT). Twenty-one percent did undergo auto-SCT and relapsed within 12 months of the procedure.
Sixty-nine percent of patients had received 3 or more lines of prior therapy, and 54% were refractory to 2 consecutive lines of prior therapy.
For this study, the patients received a conditioning regimen of cyclophosphamide (500 mg/m2) and fludarabine (30 mg/m2) for 3 days.
Two days after the conditioning regimen was completed, patients received axicabtagene ciloleucel at a target dose of 2 × 106 CAR T cells/kg.
Efficacy
The following table shows overall response data, response data at 6 months, and ongoing responses at the primary analysis data cut-off.
| DLBCL (n=77) | TFL/PMBCL (n=24) | Combined (n=101) | ||||
| ORR (%) | CR (%) | ORR (%) | CR (%) | ORR (%) | CR (%) | |
| ORR | 82 | 49 | 83 | 71 | 82 | 54 |
| Month 6 | 36 | 31 | 54 | 50 | 41 | 36 |
| Ongoing | 36 | 31 | 67 | 63 | 44 | 39 |
The researchers said the ORR was generally consistent in key subgroups. The ORR was 83% in patients who were refractory to their second or greater line of therapy and 76% in patients who relapsed within 12 months of auto-SCT.
Overall, the median duration of response was 8.2 months. However, the median duration of response has not been reached for patients with a CR.
At a median follow-up of 8.7 months, the median overall survival has not been reached.
Safety
The most common grade 3 or higher adverse events included anemia (43%), neutropenia (39%), decreased neutrophil count (32%), febrile neutropenia (31%), decreased white blood cell count (29%), thrombocytopenia (24%), encephalopathy (21%), and decreased lymphocyte count (20%).
The incidence of grade 3 or higher CRS was 13%, and the incidence of neurologic events was 28%. These represent decreases from the interim analysis of ZUMA-1, when the rate of grade 3+ CRS was 18%, and the rate of neurological events was 34%.
“We believe the rates of CRS and neurologic events decreased over the course of the study as clinicians gained experience in the management of adverse events,” said Jeff Wiezorek, MD, senior vice-president of clinical development at Kite Pharma.
There were 3 deaths throughout the course of the trial that were not a result of disease progression.
Two deaths were deemed related to axicabtagene ciloleucel. One was a case of hemophagocytic lymphohistiocytosis. The other was cardiac arrest in the setting of CRS.
The third death was the result of a pulmonary embolism and was considered unrelated to axicabtagene ciloleucel. ![]()
New BTK inhibitor may overcome resistance in CLL
WASHINGTON, DC—Preclinical research suggests a second-generation BTK inhibitor may overcome the acquired resistance observed with its predecessor in patients with chronic lymphocytic leukemia (CLL).
Investigators found the non-covalent BTK inhibitor SNS-062 was unaffected by the BTK C481S mutation, which confers resistance to the first-generation BTK inhibitor ibrutinib.
“[A] subset of patients acquire resistance to ibrutinib, the current standard-of-care BTK inhibitor,” said Amy Johnson, PhD, of The Ohio State University in Columbus.
“A key resistance mechanism to covalent BTK inhibitors is a point mutation in the BTK active site, converting cysteine 481 to serine, or C481S.”
“In this study, we demonstrate that SNS-062, which binds non-covalently to BTK, is a potent inhibitor of BTK unaffected by the presence of the C481S mutation. These findings support clinical investigation of SNS-062 to address acquired resistance to covalent BTK inhibitors in patients.”
Dr Johnson and her colleagues presented these findings at the AACR Annual Meeting 2017 (abstract 1207).
SNS-062 is being developed by Sunesis Pharmaceuticals, Inc., and company investigators were involved in this research. But the study was sponsored by The Ohio State University.
For this study, Dr Johnson and her colleagues tested SNS-062 in primary CLL cells and X-linked agammaglobulinemia human cell lines.
The investigators found that SNS-062 inhibited BTK, decreased the expression of B-cell activation markers, and reduced CLL cell viability in a dose-dependent manner. And these effects were comparable to those observed with ibrutinib.
SNS-062 and ibrutinib demonstrated comparable activity against wild-type BTK. However, ibrutinib and another BTK inhibitor, acalabrutinib, were hindered by the BTK C481S mutation, while SNS-062 was not.
The investigators said SNS-062 was 6 times more potent than ibrutinib against C481S BTK and more than 640 times more potent than acalabrutinib.
The team also noted that SNS-062 exhibited high specificity, affecting a limited number of kinases outside the TEC kinase family.
Finally, the investigators found that SNS-062 diminished stromal cell protection in CLL cells, suggesting the drug can hinder protection from the tumor microenvironment. ![]()
WASHINGTON, DC—Preclinical research suggests a second-generation BTK inhibitor may overcome the acquired resistance observed with its predecessor in patients with chronic lymphocytic leukemia (CLL).
Investigators found the non-covalent BTK inhibitor SNS-062 was unaffected by the BTK C481S mutation, which confers resistance to the first-generation BTK inhibitor ibrutinib.
“[A] subset of patients acquire resistance to ibrutinib, the current standard-of-care BTK inhibitor,” said Amy Johnson, PhD, of The Ohio State University in Columbus.
“A key resistance mechanism to covalent BTK inhibitors is a point mutation in the BTK active site, converting cysteine 481 to serine, or C481S.”
“In this study, we demonstrate that SNS-062, which binds non-covalently to BTK, is a potent inhibitor of BTK unaffected by the presence of the C481S mutation. These findings support clinical investigation of SNS-062 to address acquired resistance to covalent BTK inhibitors in patients.”
Dr Johnson and her colleagues presented these findings at the AACR Annual Meeting 2017 (abstract 1207).
SNS-062 is being developed by Sunesis Pharmaceuticals, Inc., and company investigators were involved in this research. But the study was sponsored by The Ohio State University.
For this study, Dr Johnson and her colleagues tested SNS-062 in primary CLL cells and X-linked agammaglobulinemia human cell lines.
The investigators found that SNS-062 inhibited BTK, decreased the expression of B-cell activation markers, and reduced CLL cell viability in a dose-dependent manner. And these effects were comparable to those observed with ibrutinib.
SNS-062 and ibrutinib demonstrated comparable activity against wild-type BTK. However, ibrutinib and another BTK inhibitor, acalabrutinib, were hindered by the BTK C481S mutation, while SNS-062 was not.
The investigators said SNS-062 was 6 times more potent than ibrutinib against C481S BTK and more than 640 times more potent than acalabrutinib.
The team also noted that SNS-062 exhibited high specificity, affecting a limited number of kinases outside the TEC kinase family.
Finally, the investigators found that SNS-062 diminished stromal cell protection in CLL cells, suggesting the drug can hinder protection from the tumor microenvironment. ![]()
WASHINGTON, DC—Preclinical research suggests a second-generation BTK inhibitor may overcome the acquired resistance observed with its predecessor in patients with chronic lymphocytic leukemia (CLL).
Investigators found the non-covalent BTK inhibitor SNS-062 was unaffected by the BTK C481S mutation, which confers resistance to the first-generation BTK inhibitor ibrutinib.
“[A] subset of patients acquire resistance to ibrutinib, the current standard-of-care BTK inhibitor,” said Amy Johnson, PhD, of The Ohio State University in Columbus.
“A key resistance mechanism to covalent BTK inhibitors is a point mutation in the BTK active site, converting cysteine 481 to serine, or C481S.”
“In this study, we demonstrate that SNS-062, which binds non-covalently to BTK, is a potent inhibitor of BTK unaffected by the presence of the C481S mutation. These findings support clinical investigation of SNS-062 to address acquired resistance to covalent BTK inhibitors in patients.”
Dr Johnson and her colleagues presented these findings at the AACR Annual Meeting 2017 (abstract 1207).
SNS-062 is being developed by Sunesis Pharmaceuticals, Inc., and company investigators were involved in this research. But the study was sponsored by The Ohio State University.
For this study, Dr Johnson and her colleagues tested SNS-062 in primary CLL cells and X-linked agammaglobulinemia human cell lines.
The investigators found that SNS-062 inhibited BTK, decreased the expression of B-cell activation markers, and reduced CLL cell viability in a dose-dependent manner. And these effects were comparable to those observed with ibrutinib.
SNS-062 and ibrutinib demonstrated comparable activity against wild-type BTK. However, ibrutinib and another BTK inhibitor, acalabrutinib, were hindered by the BTK C481S mutation, while SNS-062 was not.
The investigators said SNS-062 was 6 times more potent than ibrutinib against C481S BTK and more than 640 times more potent than acalabrutinib.
The team also noted that SNS-062 exhibited high specificity, affecting a limited number of kinases outside the TEC kinase family.
Finally, the investigators found that SNS-062 diminished stromal cell protection in CLL cells, suggesting the drug can hinder protection from the tumor microenvironment. ![]()
ASCO addresses needs of SGMs with cancer
The American Society of Clinical Oncology (ASCO) has issued recommendations addressing the needs of sexual and gender minority (SGM) populations with cancer.
The recommendations are designed to focus attention on the challenges facing the SGM community—including discrimination and greater risk of anxiety and depression, resulting in disparate care—and concrete steps that can help minimize health disparities among SGM individuals.
The recommendations were published in a policy statement in the Journal of Clinical Oncology.
“Sexual and gender minorities face unique challenges related to cancer risk, discrimination, and other psychosocial issues,” said ASCO President Daniel F. Hayes, MD.
“Compounding these challenges is the fact that providers may have a lack of knowledge and sensitivity about the health risks and health needs facing their SGM patients.”
SGMs include individuals who are lesbian, gay, bisexual, transgender, and intersex (also referred to as those with differences in sex development).
ASCO’s policy statement notes that SGM populations bear a disproportionate cancer burden stemming from several factors, including:
- Lower rates of cancer screening, in part due to lower rates of insurance coverage, exclusion from traditional screening campaigns, and previous experience of discrimination in the healthcare system
- A hesitancy on the part of SGM patients to disclose their sexual orientation to providers due to a fear of stigmatization, which can create additional barriers to care.
ASCO’s statement calls for a coordinated effort to address health disparities among SGM populations, including:
- Increased patient access to culturally competent support services
- Expanded cancer prevention education for SGM individuals
- Robust policies prohibiting discrimination
- Adequate insurance coverage to meet the needs of SGM individuals affected by cancer
- Inclusion of SGM status as a required data element in cancer registries and clinical trials
- Increased focus on SGM populations in cancer research.
“Our objective was to raise awareness among oncology providers, patients, policy makers, and other stakeholders about the cancer care needs of SGM populations and the barriers that SGM individuals face in getting the highest-quality care,” said Jennifer J. Griggs, MD, lead author of the policy statement and a professor at the University of Michigan in Ann Arbor.
“To address these barriers, a coordinated effort is needed to enhance education for patients and providers, to improve outreach and support, and to encourage productive policy and legislative action.” ![]()
The American Society of Clinical Oncology (ASCO) has issued recommendations addressing the needs of sexual and gender minority (SGM) populations with cancer.
The recommendations are designed to focus attention on the challenges facing the SGM community—including discrimination and greater risk of anxiety and depression, resulting in disparate care—and concrete steps that can help minimize health disparities among SGM individuals.
The recommendations were published in a policy statement in the Journal of Clinical Oncology.
“Sexual and gender minorities face unique challenges related to cancer risk, discrimination, and other psychosocial issues,” said ASCO President Daniel F. Hayes, MD.
“Compounding these challenges is the fact that providers may have a lack of knowledge and sensitivity about the health risks and health needs facing their SGM patients.”
SGMs include individuals who are lesbian, gay, bisexual, transgender, and intersex (also referred to as those with differences in sex development).
ASCO’s policy statement notes that SGM populations bear a disproportionate cancer burden stemming from several factors, including:
- Lower rates of cancer screening, in part due to lower rates of insurance coverage, exclusion from traditional screening campaigns, and previous experience of discrimination in the healthcare system
- A hesitancy on the part of SGM patients to disclose their sexual orientation to providers due to a fear of stigmatization, which can create additional barriers to care.
ASCO’s statement calls for a coordinated effort to address health disparities among SGM populations, including:
- Increased patient access to culturally competent support services
- Expanded cancer prevention education for SGM individuals
- Robust policies prohibiting discrimination
- Adequate insurance coverage to meet the needs of SGM individuals affected by cancer
- Inclusion of SGM status as a required data element in cancer registries and clinical trials
- Increased focus on SGM populations in cancer research.
“Our objective was to raise awareness among oncology providers, patients, policy makers, and other stakeholders about the cancer care needs of SGM populations and the barriers that SGM individuals face in getting the highest-quality care,” said Jennifer J. Griggs, MD, lead author of the policy statement and a professor at the University of Michigan in Ann Arbor.
“To address these barriers, a coordinated effort is needed to enhance education for patients and providers, to improve outreach and support, and to encourage productive policy and legislative action.” ![]()
The American Society of Clinical Oncology (ASCO) has issued recommendations addressing the needs of sexual and gender minority (SGM) populations with cancer.
The recommendations are designed to focus attention on the challenges facing the SGM community—including discrimination and greater risk of anxiety and depression, resulting in disparate care—and concrete steps that can help minimize health disparities among SGM individuals.
The recommendations were published in a policy statement in the Journal of Clinical Oncology.
“Sexual and gender minorities face unique challenges related to cancer risk, discrimination, and other psychosocial issues,” said ASCO President Daniel F. Hayes, MD.
“Compounding these challenges is the fact that providers may have a lack of knowledge and sensitivity about the health risks and health needs facing their SGM patients.”
SGMs include individuals who are lesbian, gay, bisexual, transgender, and intersex (also referred to as those with differences in sex development).
ASCO’s policy statement notes that SGM populations bear a disproportionate cancer burden stemming from several factors, including:
- Lower rates of cancer screening, in part due to lower rates of insurance coverage, exclusion from traditional screening campaigns, and previous experience of discrimination in the healthcare system
- A hesitancy on the part of SGM patients to disclose their sexual orientation to providers due to a fear of stigmatization, which can create additional barriers to care.
ASCO’s statement calls for a coordinated effort to address health disparities among SGM populations, including:
- Increased patient access to culturally competent support services
- Expanded cancer prevention education for SGM individuals
- Robust policies prohibiting discrimination
- Adequate insurance coverage to meet the needs of SGM individuals affected by cancer
- Inclusion of SGM status as a required data element in cancer registries and clinical trials
- Increased focus on SGM populations in cancer research.
“Our objective was to raise awareness among oncology providers, patients, policy makers, and other stakeholders about the cancer care needs of SGM populations and the barriers that SGM individuals face in getting the highest-quality care,” said Jennifer J. Griggs, MD, lead author of the policy statement and a professor at the University of Michigan in Ann Arbor.
“To address these barriers, a coordinated effort is needed to enhance education for patients and providers, to improve outreach and support, and to encourage productive policy and legislative action.” ![]()
Report shows increase in blood cancer incidence and survival
A report on cancer in the US suggests the incidence of leukemia and myeloma has been on the rise in recent years, but the incidence of non-Hodgkin lymphoma (NHL) has been on the decline.
Meanwhile, annual death rates for leukemia and NHL have decreased, and annual death rates for myeloma have decreased in men but not in women.
Furthermore, patients with leukemia, NHL, and myeloma have seen a substantial improvement in 5-year survival rates in recent years relative to patients in the late 1970s.
These findings are part of the Annual Report to the Nation on the Status of Cancer, 1975-2014, which has been published in the Journal of the National Cancer Institute.
This report is released each year, but the current edition includes a special section focused on survival.
“While trends in death rates are the most commonly used measure to assess progress against cancer, survival trends are also an important measure to evaluate progress in improvement of cancer outcomes,” said Ahmedin Jemal, DVM, PhD, of the American Cancer Society.
“We last included a special section on cancer survival in 2004, and, as we found then, survival improved over time for almost all cancers at every stage of diagnosis.”
For the current report, researchers calculated the 5-year average annual percent changes (AAPCs) for 2009 to 2013 for cancer incidence and for 2010 to 2014 for cancer mortality.
Cancer incidence (2009-2013)
In women, the AAPC increased 1.5% for leukemia (P<0.05), decreased 0.5% for NHL (P<0.05), and increased 2.2% for myeloma (P<0.05).
In men, the AAPC increased 1.7% for leukemia (P<0.05), decreased 0.2% for NHL, and increased 2.8% for myeloma (P<0.05).
Cancer mortality (2010-2014)
In women, the AAPC decreased 1.2% for leukemia (P<0.05), decreased 2.2% for NHL (P<0.05), and increased 0.5% for myeloma.
In men, the AAPC decreased 1.0% for leukemia (P<0.05), decreased 2.0% for NHL (P<0.05), and decreased 0.9% for myeloma (P<0.05).
5-year survival
The researchers compared 5-year relative survival for cancers diagnosed from 1975 to 1977 and those diagnosed from 2006 to 2012.
The absolute percentage change over time (for both sexes combined) was 26.1% for NHL, 25.7% for myeloma, and 28.5% for leukemia.
Five-year survival for patients diagnosed in 1975-1977 was 46.5% for NHL, 24.6% for myeloma, and 34.2% for leukemia.
Five-year survival for patients diagnosed in 2006-2012 was 72.6% for NHL, 50.2% for myeloma, and 62.7% for leukemia. ![]()
A report on cancer in the US suggests the incidence of leukemia and myeloma has been on the rise in recent years, but the incidence of non-Hodgkin lymphoma (NHL) has been on the decline.
Meanwhile, annual death rates for leukemia and NHL have decreased, and annual death rates for myeloma have decreased in men but not in women.
Furthermore, patients with leukemia, NHL, and myeloma have seen a substantial improvement in 5-year survival rates in recent years relative to patients in the late 1970s.
These findings are part of the Annual Report to the Nation on the Status of Cancer, 1975-2014, which has been published in the Journal of the National Cancer Institute.
This report is released each year, but the current edition includes a special section focused on survival.
“While trends in death rates are the most commonly used measure to assess progress against cancer, survival trends are also an important measure to evaluate progress in improvement of cancer outcomes,” said Ahmedin Jemal, DVM, PhD, of the American Cancer Society.
“We last included a special section on cancer survival in 2004, and, as we found then, survival improved over time for almost all cancers at every stage of diagnosis.”
For the current report, researchers calculated the 5-year average annual percent changes (AAPCs) for 2009 to 2013 for cancer incidence and for 2010 to 2014 for cancer mortality.
Cancer incidence (2009-2013)
In women, the AAPC increased 1.5% for leukemia (P<0.05), decreased 0.5% for NHL (P<0.05), and increased 2.2% for myeloma (P<0.05).
In men, the AAPC increased 1.7% for leukemia (P<0.05), decreased 0.2% for NHL, and increased 2.8% for myeloma (P<0.05).
Cancer mortality (2010-2014)
In women, the AAPC decreased 1.2% for leukemia (P<0.05), decreased 2.2% for NHL (P<0.05), and increased 0.5% for myeloma.
In men, the AAPC decreased 1.0% for leukemia (P<0.05), decreased 2.0% for NHL (P<0.05), and decreased 0.9% for myeloma (P<0.05).
5-year survival
The researchers compared 5-year relative survival for cancers diagnosed from 1975 to 1977 and those diagnosed from 2006 to 2012.
The absolute percentage change over time (for both sexes combined) was 26.1% for NHL, 25.7% for myeloma, and 28.5% for leukemia.
Five-year survival for patients diagnosed in 1975-1977 was 46.5% for NHL, 24.6% for myeloma, and 34.2% for leukemia.
Five-year survival for patients diagnosed in 2006-2012 was 72.6% for NHL, 50.2% for myeloma, and 62.7% for leukemia. ![]()
A report on cancer in the US suggests the incidence of leukemia and myeloma has been on the rise in recent years, but the incidence of non-Hodgkin lymphoma (NHL) has been on the decline.
Meanwhile, annual death rates for leukemia and NHL have decreased, and annual death rates for myeloma have decreased in men but not in women.
Furthermore, patients with leukemia, NHL, and myeloma have seen a substantial improvement in 5-year survival rates in recent years relative to patients in the late 1970s.
These findings are part of the Annual Report to the Nation on the Status of Cancer, 1975-2014, which has been published in the Journal of the National Cancer Institute.
This report is released each year, but the current edition includes a special section focused on survival.
“While trends in death rates are the most commonly used measure to assess progress against cancer, survival trends are also an important measure to evaluate progress in improvement of cancer outcomes,” said Ahmedin Jemal, DVM, PhD, of the American Cancer Society.
“We last included a special section on cancer survival in 2004, and, as we found then, survival improved over time for almost all cancers at every stage of diagnosis.”
For the current report, researchers calculated the 5-year average annual percent changes (AAPCs) for 2009 to 2013 for cancer incidence and for 2010 to 2014 for cancer mortality.
Cancer incidence (2009-2013)
In women, the AAPC increased 1.5% for leukemia (P<0.05), decreased 0.5% for NHL (P<0.05), and increased 2.2% for myeloma (P<0.05).
In men, the AAPC increased 1.7% for leukemia (P<0.05), decreased 0.2% for NHL, and increased 2.8% for myeloma (P<0.05).
Cancer mortality (2010-2014)
In women, the AAPC decreased 1.2% for leukemia (P<0.05), decreased 2.2% for NHL (P<0.05), and increased 0.5% for myeloma.
In men, the AAPC decreased 1.0% for leukemia (P<0.05), decreased 2.0% for NHL (P<0.05), and decreased 0.9% for myeloma (P<0.05).
5-year survival
The researchers compared 5-year relative survival for cancers diagnosed from 1975 to 1977 and those diagnosed from 2006 to 2012.
The absolute percentage change over time (for both sexes combined) was 26.1% for NHL, 25.7% for myeloma, and 28.5% for leukemia.
Five-year survival for patients diagnosed in 1975-1977 was 46.5% for NHL, 24.6% for myeloma, and 34.2% for leukemia.
Five-year survival for patients diagnosed in 2006-2012 was 72.6% for NHL, 50.2% for myeloma, and 62.7% for leukemia. ![]()
Demystifying the diagnosis and classification of lymphoma: a guide to the hematopathologist’s galaxy
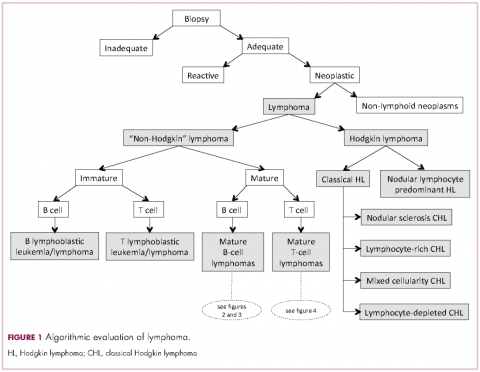
Lymphomas constitute a very heterogeneous group of neoplasms with diverse clinical presentations, prognoses, and responses to therapy. Approximately 80,500 new cases of lymphoma are expected to be diagnosed in the United States in 2017, of which about one quarter will lead to the death of the patient.1 Perhaps more so than any other group of neoplasms, the diagnosis of lymphoma involves the integration of a multiplicity of clinical, histologic and immunophenotypic findings and, on occasion, cytogenetic and molecular results as well. An accurate diagnosis of lymphoma, usually rendered by hematopathologists, allows hematologists/oncologists to treat patients appropriately. Herein we will describe a simplified approach to the diagnosis and classification of lymphomas (Figure 1).
Lymphoma classification
Lymphomas are clonal neoplasms characterized by the expansion of abnormal lymphoid cells that may develop in any organ but commonly involve lymph nodes. The fourth edition of the World Health Organization (WHO) Classification of Tumours of Haematopoietic and Lymphoid tissues, published in 2008, is the official and most current guideline used for diagnosis of lymphoid neoplasms.2 The WHO scheme classifies lymphomas according to the type of cell from which they are derived (mature and immature B cells, T cells, or natural killer (NK) cells, findings determined by their morphology and immunophenotype) and their clinical, cytogenetic, and/or molecular features. This official classification is currently being updated3 and is expected to be published in full in 2017, at which time it is anticipated to include definitions for more than 70 distinct neoplasms.
Lymphomas are broadly and informally classified as Hodgkin lymphomas (HLs) and non-Hodgkin lymphomas (NHLs), based on the differences these two groups show in their clinical presentation, treatment, prognosis, and proportion of neoplastic cells, among others. NHLs are by far the most common type of lymphomas, accounting for approximately 90% of all new cases of lymphoma in the United States and 70% worldwide.1,2 NHLs are a very heterogeneous group of B-, T-, or NK-cell neoplasms that, in turn, can also be informally subclassified as low-grade (or indolent) or high-grade (or aggressive) according to their predicted clinical behavior. HLs are comparatively rare, less heterogeneous, uniformly of B-cell origin and, in the case of classical Hodgkin lymphoma, highly curable.1,2 It is beyond the scope of this manuscript to outline the features of each of the >70 specific entities, but the reader is referred elsewhere for more detail and encouraged to become familiarized with the complexity, challenges, and beauty of lymphoma diagnosis.2,3
Biopsy procedure
A correct diagnosis begins with an adequate biopsy procedure. It is essential that biopsy specimens for lymphoma evaluation be submitted fresh and unfixed, because some crucial analyses such as flow cytometry or conventional cytogenetics can only be performed on fresh tissue. Indeed, it is important for the hematologist/oncologist and/or surgeon and/or interventional radiologist to converse with the hematopathologist prior to and even during some procedures to ensure the correct processing of the specimen. Also, it is important to limit the compression of the specimen and the excessive use of cauterization during the biopsy procedure, both of which cause artifacts that may render impossible the interpretation of the histopathologic findings.
Given that the diagnosis of lymphoma is based not only on the cytologic details of the lymphoma cells but also on the architectural pattern with which they infiltrate an organ, the larger the biopsy specimen, the easier it will be for a hematopathologist to identify the pattern. In addition, excisional biopsies frequently contain more diagnostic tissue than needle core biopsies and this provides pathologists with the option to submit tissue fragments for ancillary tests that require unfixed tissue as noted above. Needle core biopsies of lymph nodes are increasingly being used because of their association with fewer complications and lower cost than excisional biopsies. However, needle core biopsies provide only a glimpse of the pattern of infiltration and may not be completely representative of the architecture. Therefore, excisional lymph node biopsies of lymph nodes are preferred over needle core biopsies, recognizing that in the setting of deeply seated lymph nodes, needle core biopsies may be the only or the best surgical option.
Clinical presentation
Accurate diagnosis of lymphoma cannot take place in a vacuum. The hematopathologist’s initial approach to the diagnosis of lymphoid processes in tissue biopsies should begin with a thorough review of the clinical history, although some pathology laboratories may not have immediate access to this information. The hematopathologist should evaluate factors such as age, gender, location of the tumor, symptomatology, medications, serology, and prior history of malignancy, immunosuppression or immunodeficiency in every case. Other important but frequently omitted parts of the clinical history are the patient’s occupation, history of exposure to animals, and the presence of tattoos, which may be associated with certain reactive lymphadenopathies.
Histomorphologic evaluation
Despite the plethora of new and increasingly sophisticated tools, histologic and morphologic analysis still remains the cornerstone of diagnosis in hematopathology. However, for the characterization of an increasing number of reactive and neoplastic lymphoid processes, hematopathologists may also require immunophenotypic, molecular, and cytogenetic tests for an accurate diagnosis. Upon review of the clinical information, a microscopic evaluation of the tissue submitted for processing by the histology laboratory will be performed. The results of concurrent flow cytometric evaluation (performed on fresh unfixed material) should also be available in most if not all cases before the H&E-stained slides are available for review. Upon receipt of H&E-stained slides, the hematopathologist will evaluate the quality of the submitted specimen, since many diagnostic difficulties stem from suboptimal techniques related to the biopsy procedure, fixation, processing, cutting, or staining (Figure 1). If deemed suitable for accurate diagnosis, a search for signs of preservation or disruption of the organ that was biopsied will follow. The identification of certain morphologic patterns aids the hematopathologist in answering the first question: “what organ is this and is this consistent with what is indicated on the requisition?” This is usually immediately followed by “is this sufficient and adequate material for a diagnosis?” and “is there any normal architecture?” If the architecture is not normal, “is this alteration due to a reactive or a neoplastic process?” If neoplastic, “is it lymphoma or a non-hematolymphoid neoplasm?”
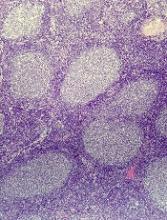
Both reactive and neoplastic processes have variably unique morphologic features that if properly recognized, guide the subsequent testing. However, some reactive and neoplastic processes can present with overlapping features, and even after extensive immunophenotypic evaluation and the performance of ancillary studies, it may not be possible to conclusively determine its nature. If the lymph node architecture is altered or effaced, the predominant pattern of infiltration (eg, nodular, diffuse, interfollicular, intrasinusoidal) and the degree of alteration of the normal architecture is evaluated, usually at low magnification. When the presence of an infiltrate is recognized, its components must be characterized. If the infiltrate is composed of a homogeneous expansion of lymphoid cells that disrupts or replaces the normal lymphoid architecture, a lymphoma will be suspected or diagnosed. The pattern of distribution of the cells along with their individual morphologic characteristics (ie, size, nuclear shape, chromatin configuration, nucleoli, amount and hue of cytoplasm) are key factors for the diagnosis and classification of the lymphoma that will guide subsequent testing. The immunophenotypic analysis (by immunohistochemistry, flow cytometry or a combination of both) may confirm the reactive or neoplastic nature of the process, and its subclassification. B-cell lymphomas, in particular have variable and distinctive histologic features: as a diffuse infiltrate of large mature lymphoid cells (eg, diffuse large B-cell lymphoma), an expansion of immature lymphoid cells (lymphoblastic lymphoma), and a nodular infiltrate of small, intermediate and/or mature large B cells (eg, follicular lymphoma).
Mature T-cell lymphomas may display similar histologic, features but they can be quite heterogeneous with an infiltrate composed of one predominant cell type or a mixture of small, medium-sized, and large atypical lymphoid cells (on occasion with abundant clear cytoplasm) and a variable number of eosinophils, plasma cells, macrophages (including granulomas), and B cells. HLs most commonly efface the lymph node architecture with a nodular or diffuse infiltrate variably composed of reactive lymphocytes, granulocytes, macrophages, and plasma cells and usually a minority of large neoplastic cells (Hodgkin/Reed-Sternberg cells and/or lymphocyte predominant cells).
Once the H&E-stained slides are evaluated and a diagnosis of lymphoma is suspected or established, the hematopathologist will attempt to determine whether it has mature or immature features, and whether low- or high-grade morphologic characteristics are present. The maturity of lymphoid cells is generally determined by the nature of the chromatin, which if “fine” and homogeneous (with or without a conspicuous nucleolus) will usually, but not always, be considered immature, whereas clumped, vesicular or hyperchromatic chromatin is generally, but not always, associated with maturity. If the chromatin displays immature features, the differential diagnosis will mainly include B- and T-lymphoblastic lymphomas, but also blastoid variants of mature neoplasm such as mantle cell lymphoma, and follicular lymphoma, as well as high-grade B-cell lymphomas. Features associated with low-grade lymphomas (eg, follicular lymphoma, small lymphocytic lymphoma/chronic lymphocytic leukemia, marginal zone lymphoma, lymphoplasmacytic lymphoma) include small cell morphology, mature chromatin, absence of a significant number of mitoses or apoptotic cells, and a low proliferation index as shown by immunohistochemistry for Ki67. High-grade lymphomas, such as lymphoblastic lymphoma, Burkitt lymphoma, or certain large B-cell lymphomas tend to show opposite features, and some of the mature entities are frequently associated with MYC rearrangements. Of note, immature lymphomas tend to be clinically high grade, but not all clinically high-grade lymphomas are immature. Conversely, the majority of low-grade lymphomas are usually mature.
Immunophenotypic evaluation
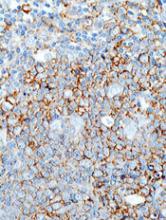
Immunophenotypic evaluation is essential because the lineage of lymphoma cells cannot be determined by morphology alone. The immunophenotype is the combination of proteins/markers (eg, CD20, CD3, TdT) expressed by cells. Usually, it is evaluated by immunohistochemistry and/or flow cytometry, which help determine the proportion of lymphoid cells that express a certain marker and its location and intensity within the cells. While immunohistochemistry is normally performed on formalin-fixed and paraffin-embedded tissue, flow cytometry can be evaluated only on fresh unfixed tissue. Flow cytometry has the advantage over immunohistochemistry of being faster and better at simultaneously identifying coexpression of multiple markers on multiple cell populations. However, certain markers can only be evaluated by immunohistochemistry.
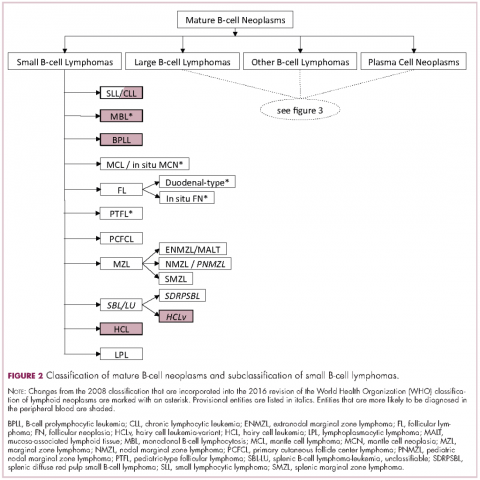
The immunophenotypic analysis will in most cases reveal whether the lymphomas is of B-, T- or NK-cell origin, and whether a lymphoma subtype associated immunophenotype is present. Typical pan B-cell antigens include PAX5, CD19, and CD79a (CD20 is less broadly expressed throughout B-cell differentiation, although it is usually evident in most mature B-cell lymphomas), and typical pan T-cell antigens include CD2, CD5, and CD7. The immature or mature nature of a lymphoma can also be confirmed by evaluation of the immunophenotype. Immature lymphomas commonly express one or more of TdT, CD10, or CD34; T-lymphoblastic lymphoma cells may also coexpress CD1a. The majority of NHLs and all HLs are derived from (or reflect) B cells at different stages of maturation. Mature B-cell lymphomas are the most common type of lymphoma and typically, but not always, express pan B-cell markers as well as surface membrane immunoglobulin, with the latter also most useful in assessing clonality via a determination of light chain restriction. Some mature B-cell lymphomas tend to acquire markers that are either never physiologically expressed by normal mature B cells (eg, cyclin D1 in mantle cell lymphoma, or BCL2 in germinal center B cells in follicular lymphoma) or only expressed in a minor fraction (eg, CD5 that is characteristically expressed in small lymphocytic and mantle cell lymphoma). The most common mature B-cell lymphomas include diffuse large B-cell lymphoma, follicular lymphoma, small lymphocytic lymphoma, mantle cell lymphoma, marginal zone lymphoma, Burkitt lymphoma, and lymphoplasmacytic lymphoma (Figures 2 and 3). Classical HLs are also lymphomas of B-cell origin that demonstrate diminished preservation of their B-cell immunophenotype (as evidenced by the dim expression of PAX5 but absence of most other pan B-cell antigens), expression of CD30, variable expression of CD15, and loss of CD45 (Figure 1). In contrast, nodular lymphocyte predominant HL shows a preserved B-cell immunophenotypic program and expression of CD45, typically without CD30 and CD15. Of note, the evaluation of the immunophenotype of the neoplastic cells in HL is routinely assessed by immunohistochemistry because most flow cytometry laboratories cannot reliably detect and characterize the low numbers of these cells.
Mature T-cell lymphomas generally express one or more T-cell markers, and tend to display a T-helper (CD4-positive) or cytotoxic (CD8-positive) immunophenotype and may show loss of markers expressed by most normal T-cells (eg, CD5, CD7; Figure 4). However, a subset of them may express markers not commonly detected in normal T cells, such as ALK. NK-cell lymphomas lack surface CD3 (expressing only cytoplasmic CD3) and CD5 but express some pan T-cell antigens (such as CD2 and CD7) as well as CD16 and/or CD56.
Patients with primary or acquired immune dysfunction are at risk for development of lymphoma and other less clearly defined lymphoproliferative disorders, the majority of which are associated with infection of the lymphoid cells with Epstein-Barr virus (EBV). Therefore, evaluation with chromogenic in situ hybridization for an EBV-encoded early RNA (EBER1) is routinely performed in these cases; it is thus essential that the hematopathologist be informed of the altered immune system of the patient. If lymphoma develops, they may be morphologically similar to those that appear in immunocompetent patients, which specifically in the post-transplant setting are known as monomorphic post-transplant lymphoproliferative disorders (PTLD). If the PTLD does not meet the criteria for any of the recognized types of lymphoma, it may be best characterized as a polymorphic PTLD.
Once the lineage (B-, T-, or NK-cell) of the mature lymphoma has been established, the sum (and on occasion the gestalt) of the clinical, morphologic, immunophenotypic and other findings will be considered for the subclassification of the neoplasm.
Cytogenetic and molecular evaluation
If the morphologic and immunophenotypic analysis is inconclusive or nondiagnostic, then molecular and/or cytogenetic testing may further aid in the characterization of the process. Some of available molecular tests include analyses for the rearrangements of the variable region of the immunoglobulin (IG) or T-cell receptor (TCR) genes and for mutations on specific genes. The identification of specific mutations not only confirms the clonal nature of the process but, on occasion, it may also help subclassify the lymphoma, whereas IG or TCR rearrangement studies are used to establish whether a lymphoid expansion is polyclonal or monoclonal. The molecular findings should not be evaluated in isolation, because not all monoclonal rearrangements are diagnostic of lymphoma, and not all lymphomas will show a monoclonal rearrangement. Other methodologies that can aid in the identification of a clonal process or specific genetic abnormalities include metaphase cytogenetics (karyotyping) and fluorescence in situ hybridization (FISH). If any cytogenetic abnormalities are found in sufficient numbers (and constitutional abnormalities are excluded), their identification indicates the presence of a clonal process. Also, some cytogenetic abnormalities are characteristic of certain lymphomas. However, they may be neither 100% diagnostically sensitive nor diagnostically specific, for example, the hallmark t(14;18)/IGH-BCL2 is not present in all follicular lymphomas and not all lymphomas with this translocation are follicular lymphomas. Whereas FISH is generally performed on a minimum of 200 cells, compared with typically 20 metaphase by “conventional” karyotyping, and is therefore considered to have higher analytical sensitivity, it evaluates only for the presence or absence of the abnormality being investigated with a given set of probes, and therefore other abnormalities, if present, will not be identified. The value of FISH cytogenetic studies is perhaps best illustrated in the need to diagnose double hit lymphomas, amongst other scenarios. The detection of certain mutations can aid in the diagnosis of certain lymphomas, such as MYD88 in lymphoplasmacytic lymphoma, prognosis of others, such as in follicular lymphoma and identify pathways that may be precisely therapeutically targeted.
Final remarks
The diagnosis of lymphoma can be complex and usually requires the hematopathologist to integrate multiple parameters. The classification of lymphomas is not static, and new entities or variants are continuously described, and the facets of well-known ones refined. While such changes are often to the chagrin of hematologists/oncologists and hematopathologists alike, we should embrace the incorporation of nascent and typically cool data into our practice, as more therapeutically relevant entities are molded.
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017 ;67(1):7-30.
2. Swerdlow SH, Campo E, Harris NL, et al, eds. WHO classification of tumours of haematopoietic and lymphoid tissues. In: Bosman FT, Jaffe ES, Lakhani SR, Ohgaki H, eds. World Health Organization Classification of Tumours. Lyon, France: IARC; 2008.
3. Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016 ;127(20):2375-2390.
Lymphomas constitute a very heterogeneous group of neoplasms with diverse clinical presentations, prognoses, and responses to therapy. Approximately 80,500 new cases of lymphoma are expected to be diagnosed in the United States in 2017, of which about one quarter will lead to the death of the patient.1 Perhaps more so than any other group of neoplasms, the diagnosis of lymphoma involves the integration of a multiplicity of clinical, histologic and immunophenotypic findings and, on occasion, cytogenetic and molecular results as well. An accurate diagnosis of lymphoma, usually rendered by hematopathologists, allows hematologists/oncologists to treat patients appropriately. Herein we will describe a simplified approach to the diagnosis and classification of lymphomas (Figure 1).
Lymphoma classification
Lymphomas are clonal neoplasms characterized by the expansion of abnormal lymphoid cells that may develop in any organ but commonly involve lymph nodes. The fourth edition of the World Health Organization (WHO) Classification of Tumours of Haematopoietic and Lymphoid tissues, published in 2008, is the official and most current guideline used for diagnosis of lymphoid neoplasms.2 The WHO scheme classifies lymphomas according to the type of cell from which they are derived (mature and immature B cells, T cells, or natural killer (NK) cells, findings determined by their morphology and immunophenotype) and their clinical, cytogenetic, and/or molecular features. This official classification is currently being updated3 and is expected to be published in full in 2017, at which time it is anticipated to include definitions for more than 70 distinct neoplasms.
Lymphomas are broadly and informally classified as Hodgkin lymphomas (HLs) and non-Hodgkin lymphomas (NHLs), based on the differences these two groups show in their clinical presentation, treatment, prognosis, and proportion of neoplastic cells, among others. NHLs are by far the most common type of lymphomas, accounting for approximately 90% of all new cases of lymphoma in the United States and 70% worldwide.1,2 NHLs are a very heterogeneous group of B-, T-, or NK-cell neoplasms that, in turn, can also be informally subclassified as low-grade (or indolent) or high-grade (or aggressive) according to their predicted clinical behavior. HLs are comparatively rare, less heterogeneous, uniformly of B-cell origin and, in the case of classical Hodgkin lymphoma, highly curable.1,2 It is beyond the scope of this manuscript to outline the features of each of the >70 specific entities, but the reader is referred elsewhere for more detail and encouraged to become familiarized with the complexity, challenges, and beauty of lymphoma diagnosis.2,3
Biopsy procedure
A correct diagnosis begins with an adequate biopsy procedure. It is essential that biopsy specimens for lymphoma evaluation be submitted fresh and unfixed, because some crucial analyses such as flow cytometry or conventional cytogenetics can only be performed on fresh tissue. Indeed, it is important for the hematologist/oncologist and/or surgeon and/or interventional radiologist to converse with the hematopathologist prior to and even during some procedures to ensure the correct processing of the specimen. Also, it is important to limit the compression of the specimen and the excessive use of cauterization during the biopsy procedure, both of which cause artifacts that may render impossible the interpretation of the histopathologic findings.
Given that the diagnosis of lymphoma is based not only on the cytologic details of the lymphoma cells but also on the architectural pattern with which they infiltrate an organ, the larger the biopsy specimen, the easier it will be for a hematopathologist to identify the pattern. In addition, excisional biopsies frequently contain more diagnostic tissue than needle core biopsies and this provides pathologists with the option to submit tissue fragments for ancillary tests that require unfixed tissue as noted above. Needle core biopsies of lymph nodes are increasingly being used because of their association with fewer complications and lower cost than excisional biopsies. However, needle core biopsies provide only a glimpse of the pattern of infiltration and may not be completely representative of the architecture. Therefore, excisional lymph node biopsies of lymph nodes are preferred over needle core biopsies, recognizing that in the setting of deeply seated lymph nodes, needle core biopsies may be the only or the best surgical option.
Clinical presentation
Accurate diagnosis of lymphoma cannot take place in a vacuum. The hematopathologist’s initial approach to the diagnosis of lymphoid processes in tissue biopsies should begin with a thorough review of the clinical history, although some pathology laboratories may not have immediate access to this information. The hematopathologist should evaluate factors such as age, gender, location of the tumor, symptomatology, medications, serology, and prior history of malignancy, immunosuppression or immunodeficiency in every case. Other important but frequently omitted parts of the clinical history are the patient’s occupation, history of exposure to animals, and the presence of tattoos, which may be associated with certain reactive lymphadenopathies.
Histomorphologic evaluation
Despite the plethora of new and increasingly sophisticated tools, histologic and morphologic analysis still remains the cornerstone of diagnosis in hematopathology. However, for the characterization of an increasing number of reactive and neoplastic lymphoid processes, hematopathologists may also require immunophenotypic, molecular, and cytogenetic tests for an accurate diagnosis. Upon review of the clinical information, a microscopic evaluation of the tissue submitted for processing by the histology laboratory will be performed. The results of concurrent flow cytometric evaluation (performed on fresh unfixed material) should also be available in most if not all cases before the H&E-stained slides are available for review. Upon receipt of H&E-stained slides, the hematopathologist will evaluate the quality of the submitted specimen, since many diagnostic difficulties stem from suboptimal techniques related to the biopsy procedure, fixation, processing, cutting, or staining (Figure 1). If deemed suitable for accurate diagnosis, a search for signs of preservation or disruption of the organ that was biopsied will follow. The identification of certain morphologic patterns aids the hematopathologist in answering the first question: “what organ is this and is this consistent with what is indicated on the requisition?” This is usually immediately followed by “is this sufficient and adequate material for a diagnosis?” and “is there any normal architecture?” If the architecture is not normal, “is this alteration due to a reactive or a neoplastic process?” If neoplastic, “is it lymphoma or a non-hematolymphoid neoplasm?”
Both reactive and neoplastic processes have variably unique morphologic features that if properly recognized, guide the subsequent testing. However, some reactive and neoplastic processes can present with overlapping features, and even after extensive immunophenotypic evaluation and the performance of ancillary studies, it may not be possible to conclusively determine its nature. If the lymph node architecture is altered or effaced, the predominant pattern of infiltration (eg, nodular, diffuse, interfollicular, intrasinusoidal) and the degree of alteration of the normal architecture is evaluated, usually at low magnification. When the presence of an infiltrate is recognized, its components must be characterized. If the infiltrate is composed of a homogeneous expansion of lymphoid cells that disrupts or replaces the normal lymphoid architecture, a lymphoma will be suspected or diagnosed. The pattern of distribution of the cells along with their individual morphologic characteristics (ie, size, nuclear shape, chromatin configuration, nucleoli, amount and hue of cytoplasm) are key factors for the diagnosis and classification of the lymphoma that will guide subsequent testing. The immunophenotypic analysis (by immunohistochemistry, flow cytometry or a combination of both) may confirm the reactive or neoplastic nature of the process, and its subclassification. B-cell lymphomas, in particular have variable and distinctive histologic features: as a diffuse infiltrate of large mature lymphoid cells (eg, diffuse large B-cell lymphoma), an expansion of immature lymphoid cells (lymphoblastic lymphoma), and a nodular infiltrate of small, intermediate and/or mature large B cells (eg, follicular lymphoma).
Mature T-cell lymphomas may display similar histologic, features but they can be quite heterogeneous with an infiltrate composed of one predominant cell type or a mixture of small, medium-sized, and large atypical lymphoid cells (on occasion with abundant clear cytoplasm) and a variable number of eosinophils, plasma cells, macrophages (including granulomas), and B cells. HLs most commonly efface the lymph node architecture with a nodular or diffuse infiltrate variably composed of reactive lymphocytes, granulocytes, macrophages, and plasma cells and usually a minority of large neoplastic cells (Hodgkin/Reed-Sternberg cells and/or lymphocyte predominant cells).
Once the H&E-stained slides are evaluated and a diagnosis of lymphoma is suspected or established, the hematopathologist will attempt to determine whether it has mature or immature features, and whether low- or high-grade morphologic characteristics are present. The maturity of lymphoid cells is generally determined by the nature of the chromatin, which if “fine” and homogeneous (with or without a conspicuous nucleolus) will usually, but not always, be considered immature, whereas clumped, vesicular or hyperchromatic chromatin is generally, but not always, associated with maturity. If the chromatin displays immature features, the differential diagnosis will mainly include B- and T-lymphoblastic lymphomas, but also blastoid variants of mature neoplasm such as mantle cell lymphoma, and follicular lymphoma, as well as high-grade B-cell lymphomas. Features associated with low-grade lymphomas (eg, follicular lymphoma, small lymphocytic lymphoma/chronic lymphocytic leukemia, marginal zone lymphoma, lymphoplasmacytic lymphoma) include small cell morphology, mature chromatin, absence of a significant number of mitoses or apoptotic cells, and a low proliferation index as shown by immunohistochemistry for Ki67. High-grade lymphomas, such as lymphoblastic lymphoma, Burkitt lymphoma, or certain large B-cell lymphomas tend to show opposite features, and some of the mature entities are frequently associated with MYC rearrangements. Of note, immature lymphomas tend to be clinically high grade, but not all clinically high-grade lymphomas are immature. Conversely, the majority of low-grade lymphomas are usually mature.
Immunophenotypic evaluation
Immunophenotypic evaluation is essential because the lineage of lymphoma cells cannot be determined by morphology alone. The immunophenotype is the combination of proteins/markers (eg, CD20, CD3, TdT) expressed by cells. Usually, it is evaluated by immunohistochemistry and/or flow cytometry, which help determine the proportion of lymphoid cells that express a certain marker and its location and intensity within the cells. While immunohistochemistry is normally performed on formalin-fixed and paraffin-embedded tissue, flow cytometry can be evaluated only on fresh unfixed tissue. Flow cytometry has the advantage over immunohistochemistry of being faster and better at simultaneously identifying coexpression of multiple markers on multiple cell populations. However, certain markers can only be evaluated by immunohistochemistry.
The immunophenotypic analysis will in most cases reveal whether the lymphomas is of B-, T- or NK-cell origin, and whether a lymphoma subtype associated immunophenotype is present. Typical pan B-cell antigens include PAX5, CD19, and CD79a (CD20 is less broadly expressed throughout B-cell differentiation, although it is usually evident in most mature B-cell lymphomas), and typical pan T-cell antigens include CD2, CD5, and CD7. The immature or mature nature of a lymphoma can also be confirmed by evaluation of the immunophenotype. Immature lymphomas commonly express one or more of TdT, CD10, or CD34; T-lymphoblastic lymphoma cells may also coexpress CD1a. The majority of NHLs and all HLs are derived from (or reflect) B cells at different stages of maturation. Mature B-cell lymphomas are the most common type of lymphoma and typically, but not always, express pan B-cell markers as well as surface membrane immunoglobulin, with the latter also most useful in assessing clonality via a determination of light chain restriction. Some mature B-cell lymphomas tend to acquire markers that are either never physiologically expressed by normal mature B cells (eg, cyclin D1 in mantle cell lymphoma, or BCL2 in germinal center B cells in follicular lymphoma) or only expressed in a minor fraction (eg, CD5 that is characteristically expressed in small lymphocytic and mantle cell lymphoma). The most common mature B-cell lymphomas include diffuse large B-cell lymphoma, follicular lymphoma, small lymphocytic lymphoma, mantle cell lymphoma, marginal zone lymphoma, Burkitt lymphoma, and lymphoplasmacytic lymphoma (Figures 2 and 3). Classical HLs are also lymphomas of B-cell origin that demonstrate diminished preservation of their B-cell immunophenotype (as evidenced by the dim expression of PAX5 but absence of most other pan B-cell antigens), expression of CD30, variable expression of CD15, and loss of CD45 (Figure 1). In contrast, nodular lymphocyte predominant HL shows a preserved B-cell immunophenotypic program and expression of CD45, typically without CD30 and CD15. Of note, the evaluation of the immunophenotype of the neoplastic cells in HL is routinely assessed by immunohistochemistry because most flow cytometry laboratories cannot reliably detect and characterize the low numbers of these cells.
Mature T-cell lymphomas generally express one or more T-cell markers, and tend to display a T-helper (CD4-positive) or cytotoxic (CD8-positive) immunophenotype and may show loss of markers expressed by most normal T-cells (eg, CD5, CD7; Figure 4). However, a subset of them may express markers not commonly detected in normal T cells, such as ALK. NK-cell lymphomas lack surface CD3 (expressing only cytoplasmic CD3) and CD5 but express some pan T-cell antigens (such as CD2 and CD7) as well as CD16 and/or CD56.
Patients with primary or acquired immune dysfunction are at risk for development of lymphoma and other less clearly defined lymphoproliferative disorders, the majority of which are associated with infection of the lymphoid cells with Epstein-Barr virus (EBV). Therefore, evaluation with chromogenic in situ hybridization for an EBV-encoded early RNA (EBER1) is routinely performed in these cases; it is thus essential that the hematopathologist be informed of the altered immune system of the patient. If lymphoma develops, they may be morphologically similar to those that appear in immunocompetent patients, which specifically in the post-transplant setting are known as monomorphic post-transplant lymphoproliferative disorders (PTLD). If the PTLD does not meet the criteria for any of the recognized types of lymphoma, it may be best characterized as a polymorphic PTLD.
Once the lineage (B-, T-, or NK-cell) of the mature lymphoma has been established, the sum (and on occasion the gestalt) of the clinical, morphologic, immunophenotypic and other findings will be considered for the subclassification of the neoplasm.
Cytogenetic and molecular evaluation
If the morphologic and immunophenotypic analysis is inconclusive or nondiagnostic, then molecular and/or cytogenetic testing may further aid in the characterization of the process. Some of available molecular tests include analyses for the rearrangements of the variable region of the immunoglobulin (IG) or T-cell receptor (TCR) genes and for mutations on specific genes. The identification of specific mutations not only confirms the clonal nature of the process but, on occasion, it may also help subclassify the lymphoma, whereas IG or TCR rearrangement studies are used to establish whether a lymphoid expansion is polyclonal or monoclonal. The molecular findings should not be evaluated in isolation, because not all monoclonal rearrangements are diagnostic of lymphoma, and not all lymphomas will show a monoclonal rearrangement. Other methodologies that can aid in the identification of a clonal process or specific genetic abnormalities include metaphase cytogenetics (karyotyping) and fluorescence in situ hybridization (FISH). If any cytogenetic abnormalities are found in sufficient numbers (and constitutional abnormalities are excluded), their identification indicates the presence of a clonal process. Also, some cytogenetic abnormalities are characteristic of certain lymphomas. However, they may be neither 100% diagnostically sensitive nor diagnostically specific, for example, the hallmark t(14;18)/IGH-BCL2 is not present in all follicular lymphomas and not all lymphomas with this translocation are follicular lymphomas. Whereas FISH is generally performed on a minimum of 200 cells, compared with typically 20 metaphase by “conventional” karyotyping, and is therefore considered to have higher analytical sensitivity, it evaluates only for the presence or absence of the abnormality being investigated with a given set of probes, and therefore other abnormalities, if present, will not be identified. The value of FISH cytogenetic studies is perhaps best illustrated in the need to diagnose double hit lymphomas, amongst other scenarios. The detection of certain mutations can aid in the diagnosis of certain lymphomas, such as MYD88 in lymphoplasmacytic lymphoma, prognosis of others, such as in follicular lymphoma and identify pathways that may be precisely therapeutically targeted.
Final remarks
The diagnosis of lymphoma can be complex and usually requires the hematopathologist to integrate multiple parameters. The classification of lymphomas is not static, and new entities or variants are continuously described, and the facets of well-known ones refined. While such changes are often to the chagrin of hematologists/oncologists and hematopathologists alike, we should embrace the incorporation of nascent and typically cool data into our practice, as more therapeutically relevant entities are molded.
Lymphomas constitute a very heterogeneous group of neoplasms with diverse clinical presentations, prognoses, and responses to therapy. Approximately 80,500 new cases of lymphoma are expected to be diagnosed in the United States in 2017, of which about one quarter will lead to the death of the patient.1 Perhaps more so than any other group of neoplasms, the diagnosis of lymphoma involves the integration of a multiplicity of clinical, histologic and immunophenotypic findings and, on occasion, cytogenetic and molecular results as well. An accurate diagnosis of lymphoma, usually rendered by hematopathologists, allows hematologists/oncologists to treat patients appropriately. Herein we will describe a simplified approach to the diagnosis and classification of lymphomas (Figure 1).
Lymphoma classification
Lymphomas are clonal neoplasms characterized by the expansion of abnormal lymphoid cells that may develop in any organ but commonly involve lymph nodes. The fourth edition of the World Health Organization (WHO) Classification of Tumours of Haematopoietic and Lymphoid tissues, published in 2008, is the official and most current guideline used for diagnosis of lymphoid neoplasms.2 The WHO scheme classifies lymphomas according to the type of cell from which they are derived (mature and immature B cells, T cells, or natural killer (NK) cells, findings determined by their morphology and immunophenotype) and their clinical, cytogenetic, and/or molecular features. This official classification is currently being updated3 and is expected to be published in full in 2017, at which time it is anticipated to include definitions for more than 70 distinct neoplasms.
Lymphomas are broadly and informally classified as Hodgkin lymphomas (HLs) and non-Hodgkin lymphomas (NHLs), based on the differences these two groups show in their clinical presentation, treatment, prognosis, and proportion of neoplastic cells, among others. NHLs are by far the most common type of lymphomas, accounting for approximately 90% of all new cases of lymphoma in the United States and 70% worldwide.1,2 NHLs are a very heterogeneous group of B-, T-, or NK-cell neoplasms that, in turn, can also be informally subclassified as low-grade (or indolent) or high-grade (or aggressive) according to their predicted clinical behavior. HLs are comparatively rare, less heterogeneous, uniformly of B-cell origin and, in the case of classical Hodgkin lymphoma, highly curable.1,2 It is beyond the scope of this manuscript to outline the features of each of the >70 specific entities, but the reader is referred elsewhere for more detail and encouraged to become familiarized with the complexity, challenges, and beauty of lymphoma diagnosis.2,3
Biopsy procedure
A correct diagnosis begins with an adequate biopsy procedure. It is essential that biopsy specimens for lymphoma evaluation be submitted fresh and unfixed, because some crucial analyses such as flow cytometry or conventional cytogenetics can only be performed on fresh tissue. Indeed, it is important for the hematologist/oncologist and/or surgeon and/or interventional radiologist to converse with the hematopathologist prior to and even during some procedures to ensure the correct processing of the specimen. Also, it is important to limit the compression of the specimen and the excessive use of cauterization during the biopsy procedure, both of which cause artifacts that may render impossible the interpretation of the histopathologic findings.
Given that the diagnosis of lymphoma is based not only on the cytologic details of the lymphoma cells but also on the architectural pattern with which they infiltrate an organ, the larger the biopsy specimen, the easier it will be for a hematopathologist to identify the pattern. In addition, excisional biopsies frequently contain more diagnostic tissue than needle core biopsies and this provides pathologists with the option to submit tissue fragments for ancillary tests that require unfixed tissue as noted above. Needle core biopsies of lymph nodes are increasingly being used because of their association with fewer complications and lower cost than excisional biopsies. However, needle core biopsies provide only a glimpse of the pattern of infiltration and may not be completely representative of the architecture. Therefore, excisional lymph node biopsies of lymph nodes are preferred over needle core biopsies, recognizing that in the setting of deeply seated lymph nodes, needle core biopsies may be the only or the best surgical option.
Clinical presentation
Accurate diagnosis of lymphoma cannot take place in a vacuum. The hematopathologist’s initial approach to the diagnosis of lymphoid processes in tissue biopsies should begin with a thorough review of the clinical history, although some pathology laboratories may not have immediate access to this information. The hematopathologist should evaluate factors such as age, gender, location of the tumor, symptomatology, medications, serology, and prior history of malignancy, immunosuppression or immunodeficiency in every case. Other important but frequently omitted parts of the clinical history are the patient’s occupation, history of exposure to animals, and the presence of tattoos, which may be associated with certain reactive lymphadenopathies.
Histomorphologic evaluation
Despite the plethora of new and increasingly sophisticated tools, histologic and morphologic analysis still remains the cornerstone of diagnosis in hematopathology. However, for the characterization of an increasing number of reactive and neoplastic lymphoid processes, hematopathologists may also require immunophenotypic, molecular, and cytogenetic tests for an accurate diagnosis. Upon review of the clinical information, a microscopic evaluation of the tissue submitted for processing by the histology laboratory will be performed. The results of concurrent flow cytometric evaluation (performed on fresh unfixed material) should also be available in most if not all cases before the H&E-stained slides are available for review. Upon receipt of H&E-stained slides, the hematopathologist will evaluate the quality of the submitted specimen, since many diagnostic difficulties stem from suboptimal techniques related to the biopsy procedure, fixation, processing, cutting, or staining (Figure 1). If deemed suitable for accurate diagnosis, a search for signs of preservation or disruption of the organ that was biopsied will follow. The identification of certain morphologic patterns aids the hematopathologist in answering the first question: “what organ is this and is this consistent with what is indicated on the requisition?” This is usually immediately followed by “is this sufficient and adequate material for a diagnosis?” and “is there any normal architecture?” If the architecture is not normal, “is this alteration due to a reactive or a neoplastic process?” If neoplastic, “is it lymphoma or a non-hematolymphoid neoplasm?”
Both reactive and neoplastic processes have variably unique morphologic features that if properly recognized, guide the subsequent testing. However, some reactive and neoplastic processes can present with overlapping features, and even after extensive immunophenotypic evaluation and the performance of ancillary studies, it may not be possible to conclusively determine its nature. If the lymph node architecture is altered or effaced, the predominant pattern of infiltration (eg, nodular, diffuse, interfollicular, intrasinusoidal) and the degree of alteration of the normal architecture is evaluated, usually at low magnification. When the presence of an infiltrate is recognized, its components must be characterized. If the infiltrate is composed of a homogeneous expansion of lymphoid cells that disrupts or replaces the normal lymphoid architecture, a lymphoma will be suspected or diagnosed. The pattern of distribution of the cells along with their individual morphologic characteristics (ie, size, nuclear shape, chromatin configuration, nucleoli, amount and hue of cytoplasm) are key factors for the diagnosis and classification of the lymphoma that will guide subsequent testing. The immunophenotypic analysis (by immunohistochemistry, flow cytometry or a combination of both) may confirm the reactive or neoplastic nature of the process, and its subclassification. B-cell lymphomas, in particular have variable and distinctive histologic features: as a diffuse infiltrate of large mature lymphoid cells (eg, diffuse large B-cell lymphoma), an expansion of immature lymphoid cells (lymphoblastic lymphoma), and a nodular infiltrate of small, intermediate and/or mature large B cells (eg, follicular lymphoma).
Mature T-cell lymphomas may display similar histologic, features but they can be quite heterogeneous with an infiltrate composed of one predominant cell type or a mixture of small, medium-sized, and large atypical lymphoid cells (on occasion with abundant clear cytoplasm) and a variable number of eosinophils, plasma cells, macrophages (including granulomas), and B cells. HLs most commonly efface the lymph node architecture with a nodular or diffuse infiltrate variably composed of reactive lymphocytes, granulocytes, macrophages, and plasma cells and usually a minority of large neoplastic cells (Hodgkin/Reed-Sternberg cells and/or lymphocyte predominant cells).
Once the H&E-stained slides are evaluated and a diagnosis of lymphoma is suspected or established, the hematopathologist will attempt to determine whether it has mature or immature features, and whether low- or high-grade morphologic characteristics are present. The maturity of lymphoid cells is generally determined by the nature of the chromatin, which if “fine” and homogeneous (with or without a conspicuous nucleolus) will usually, but not always, be considered immature, whereas clumped, vesicular or hyperchromatic chromatin is generally, but not always, associated with maturity. If the chromatin displays immature features, the differential diagnosis will mainly include B- and T-lymphoblastic lymphomas, but also blastoid variants of mature neoplasm such as mantle cell lymphoma, and follicular lymphoma, as well as high-grade B-cell lymphomas. Features associated with low-grade lymphomas (eg, follicular lymphoma, small lymphocytic lymphoma/chronic lymphocytic leukemia, marginal zone lymphoma, lymphoplasmacytic lymphoma) include small cell morphology, mature chromatin, absence of a significant number of mitoses or apoptotic cells, and a low proliferation index as shown by immunohistochemistry for Ki67. High-grade lymphomas, such as lymphoblastic lymphoma, Burkitt lymphoma, or certain large B-cell lymphomas tend to show opposite features, and some of the mature entities are frequently associated with MYC rearrangements. Of note, immature lymphomas tend to be clinically high grade, but not all clinically high-grade lymphomas are immature. Conversely, the majority of low-grade lymphomas are usually mature.
Immunophenotypic evaluation
Immunophenotypic evaluation is essential because the lineage of lymphoma cells cannot be determined by morphology alone. The immunophenotype is the combination of proteins/markers (eg, CD20, CD3, TdT) expressed by cells. Usually, it is evaluated by immunohistochemistry and/or flow cytometry, which help determine the proportion of lymphoid cells that express a certain marker and its location and intensity within the cells. While immunohistochemistry is normally performed on formalin-fixed and paraffin-embedded tissue, flow cytometry can be evaluated only on fresh unfixed tissue. Flow cytometry has the advantage over immunohistochemistry of being faster and better at simultaneously identifying coexpression of multiple markers on multiple cell populations. However, certain markers can only be evaluated by immunohistochemistry.
The immunophenotypic analysis will in most cases reveal whether the lymphomas is of B-, T- or NK-cell origin, and whether a lymphoma subtype associated immunophenotype is present. Typical pan B-cell antigens include PAX5, CD19, and CD79a (CD20 is less broadly expressed throughout B-cell differentiation, although it is usually evident in most mature B-cell lymphomas), and typical pan T-cell antigens include CD2, CD5, and CD7. The immature or mature nature of a lymphoma can also be confirmed by evaluation of the immunophenotype. Immature lymphomas commonly express one or more of TdT, CD10, or CD34; T-lymphoblastic lymphoma cells may also coexpress CD1a. The majority of NHLs and all HLs are derived from (or reflect) B cells at different stages of maturation. Mature B-cell lymphomas are the most common type of lymphoma and typically, but not always, express pan B-cell markers as well as surface membrane immunoglobulin, with the latter also most useful in assessing clonality via a determination of light chain restriction. Some mature B-cell lymphomas tend to acquire markers that are either never physiologically expressed by normal mature B cells (eg, cyclin D1 in mantle cell lymphoma, or BCL2 in germinal center B cells in follicular lymphoma) or only expressed in a minor fraction (eg, CD5 that is characteristically expressed in small lymphocytic and mantle cell lymphoma). The most common mature B-cell lymphomas include diffuse large B-cell lymphoma, follicular lymphoma, small lymphocytic lymphoma, mantle cell lymphoma, marginal zone lymphoma, Burkitt lymphoma, and lymphoplasmacytic lymphoma (Figures 2 and 3). Classical HLs are also lymphomas of B-cell origin that demonstrate diminished preservation of their B-cell immunophenotype (as evidenced by the dim expression of PAX5 but absence of most other pan B-cell antigens), expression of CD30, variable expression of CD15, and loss of CD45 (Figure 1). In contrast, nodular lymphocyte predominant HL shows a preserved B-cell immunophenotypic program and expression of CD45, typically without CD30 and CD15. Of note, the evaluation of the immunophenotype of the neoplastic cells in HL is routinely assessed by immunohistochemistry because most flow cytometry laboratories cannot reliably detect and characterize the low numbers of these cells.
Mature T-cell lymphomas generally express one or more T-cell markers, and tend to display a T-helper (CD4-positive) or cytotoxic (CD8-positive) immunophenotype and may show loss of markers expressed by most normal T-cells (eg, CD5, CD7; Figure 4). However, a subset of them may express markers not commonly detected in normal T cells, such as ALK. NK-cell lymphomas lack surface CD3 (expressing only cytoplasmic CD3) and CD5 but express some pan T-cell antigens (such as CD2 and CD7) as well as CD16 and/or CD56.
Patients with primary or acquired immune dysfunction are at risk for development of lymphoma and other less clearly defined lymphoproliferative disorders, the majority of which are associated with infection of the lymphoid cells with Epstein-Barr virus (EBV). Therefore, evaluation with chromogenic in situ hybridization for an EBV-encoded early RNA (EBER1) is routinely performed in these cases; it is thus essential that the hematopathologist be informed of the altered immune system of the patient. If lymphoma develops, they may be morphologically similar to those that appear in immunocompetent patients, which specifically in the post-transplant setting are known as monomorphic post-transplant lymphoproliferative disorders (PTLD). If the PTLD does not meet the criteria for any of the recognized types of lymphoma, it may be best characterized as a polymorphic PTLD.
Once the lineage (B-, T-, or NK-cell) of the mature lymphoma has been established, the sum (and on occasion the gestalt) of the clinical, morphologic, immunophenotypic and other findings will be considered for the subclassification of the neoplasm.
Cytogenetic and molecular evaluation
If the morphologic and immunophenotypic analysis is inconclusive or nondiagnostic, then molecular and/or cytogenetic testing may further aid in the characterization of the process. Some of available molecular tests include analyses for the rearrangements of the variable region of the immunoglobulin (IG) or T-cell receptor (TCR) genes and for mutations on specific genes. The identification of specific mutations not only confirms the clonal nature of the process but, on occasion, it may also help subclassify the lymphoma, whereas IG or TCR rearrangement studies are used to establish whether a lymphoid expansion is polyclonal or monoclonal. The molecular findings should not be evaluated in isolation, because not all monoclonal rearrangements are diagnostic of lymphoma, and not all lymphomas will show a monoclonal rearrangement. Other methodologies that can aid in the identification of a clonal process or specific genetic abnormalities include metaphase cytogenetics (karyotyping) and fluorescence in situ hybridization (FISH). If any cytogenetic abnormalities are found in sufficient numbers (and constitutional abnormalities are excluded), their identification indicates the presence of a clonal process. Also, some cytogenetic abnormalities are characteristic of certain lymphomas. However, they may be neither 100% diagnostically sensitive nor diagnostically specific, for example, the hallmark t(14;18)/IGH-BCL2 is not present in all follicular lymphomas and not all lymphomas with this translocation are follicular lymphomas. Whereas FISH is generally performed on a minimum of 200 cells, compared with typically 20 metaphase by “conventional” karyotyping, and is therefore considered to have higher analytical sensitivity, it evaluates only for the presence or absence of the abnormality being investigated with a given set of probes, and therefore other abnormalities, if present, will not be identified. The value of FISH cytogenetic studies is perhaps best illustrated in the need to diagnose double hit lymphomas, amongst other scenarios. The detection of certain mutations can aid in the diagnosis of certain lymphomas, such as MYD88 in lymphoplasmacytic lymphoma, prognosis of others, such as in follicular lymphoma and identify pathways that may be precisely therapeutically targeted.
Final remarks
The diagnosis of lymphoma can be complex and usually requires the hematopathologist to integrate multiple parameters. The classification of lymphomas is not static, and new entities or variants are continuously described, and the facets of well-known ones refined. While such changes are often to the chagrin of hematologists/oncologists and hematopathologists alike, we should embrace the incorporation of nascent and typically cool data into our practice, as more therapeutically relevant entities are molded.
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017 ;67(1):7-30.
2. Swerdlow SH, Campo E, Harris NL, et al, eds. WHO classification of tumours of haematopoietic and lymphoid tissues. In: Bosman FT, Jaffe ES, Lakhani SR, Ohgaki H, eds. World Health Organization Classification of Tumours. Lyon, France: IARC; 2008.
3. Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016 ;127(20):2375-2390.
1. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017 ;67(1):7-30.
2. Swerdlow SH, Campo E, Harris NL, et al, eds. WHO classification of tumours of haematopoietic and lymphoid tissues. In: Bosman FT, Jaffe ES, Lakhani SR, Ohgaki H, eds. World Health Organization Classification of Tumours. Lyon, France: IARC; 2008.
3. Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016 ;127(20):2375-2390.
FDA lifts partial clinical hold for some selinexor trials
The US Food and Drug Administration (FDA) has lifted the partial clinical hold on trials of selinexor (KPT-330) in patients with hematologic malignancies.
The partial hold, which was announced on March 10, was placed on all trials of the drug, including those in patients with solid tumor malignancies.
The hold meant that no new patients could be enrolled in selinexor trials.
Patients who were already enrolled and had stable disease or better could remain on selinexor therapy.
Now, the FDA has lifted the hold on trials of patients with hematologic malignancies, so new patients can be enrolled in these trials and begin receiving selinexor.
The FDA had placed the hold due to a lack of information in the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, noted that the hold was not the result of patient deaths or any new information regarding the safety profile of selinexor.
In response to the hold, Karyopharm amended the investigator’s brochure, updated informed consent documents, and submitted the documents to the FDA.
“The Karyopharm team worked diligently to update and submit the required documents to the FDA, which allowed the hematology division to expeditiously remove the partial clinical hold,” said Michael G. Kauffman, MD, PhD, chief executive officer of Karyopharm.
“We anticipate that the solid tumor divisions will follow suit shortly. Patient enrollment is again underway in our hematologic oncology studies. Our previously disclosed enrollment rates and timelines for both ongoing and planned trials are not expected to be materially impacted.”
About selinexor
Selinexor is a first-in-class, oral, selective inhibitor of nuclear export compound. The drug functions by inhibiting the nuclear export protein XPO1 (also called CRM1).
This leads to the accumulation of tumor suppressor proteins in the cell nucleus, which subsequently reinitiates and amplifies their tumor suppressor function. This is thought to prompt apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor.
The drug is currently being evaluated in clinical trials across multiple cancer indications, including in acute myeloid leukemia (SOPRA), in multiple myeloma in combination with low-dose dexamethasone (STORM) and backbone therapies (STOMP), as well as in diffuse large B-cell lymphoma (SADAL).
The US Food and Drug Administration (FDA) has lifted the partial clinical hold on trials of selinexor (KPT-330) in patients with hematologic malignancies.
The partial hold, which was announced on March 10, was placed on all trials of the drug, including those in patients with solid tumor malignancies.
The hold meant that no new patients could be enrolled in selinexor trials.
Patients who were already enrolled and had stable disease or better could remain on selinexor therapy.
Now, the FDA has lifted the hold on trials of patients with hematologic malignancies, so new patients can be enrolled in these trials and begin receiving selinexor.
The FDA had placed the hold due to a lack of information in the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, noted that the hold was not the result of patient deaths or any new information regarding the safety profile of selinexor.
In response to the hold, Karyopharm amended the investigator’s brochure, updated informed consent documents, and submitted the documents to the FDA.
“The Karyopharm team worked diligently to update and submit the required documents to the FDA, which allowed the hematology division to expeditiously remove the partial clinical hold,” said Michael G. Kauffman, MD, PhD, chief executive officer of Karyopharm.
“We anticipate that the solid tumor divisions will follow suit shortly. Patient enrollment is again underway in our hematologic oncology studies. Our previously disclosed enrollment rates and timelines for both ongoing and planned trials are not expected to be materially impacted.”
About selinexor
Selinexor is a first-in-class, oral, selective inhibitor of nuclear export compound. The drug functions by inhibiting the nuclear export protein XPO1 (also called CRM1).
This leads to the accumulation of tumor suppressor proteins in the cell nucleus, which subsequently reinitiates and amplifies their tumor suppressor function. This is thought to prompt apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor.
The drug is currently being evaluated in clinical trials across multiple cancer indications, including in acute myeloid leukemia (SOPRA), in multiple myeloma in combination with low-dose dexamethasone (STORM) and backbone therapies (STOMP), as well as in diffuse large B-cell lymphoma (SADAL).
The US Food and Drug Administration (FDA) has lifted the partial clinical hold on trials of selinexor (KPT-330) in patients with hematologic malignancies.
The partial hold, which was announced on March 10, was placed on all trials of the drug, including those in patients with solid tumor malignancies.
The hold meant that no new patients could be enrolled in selinexor trials.
Patients who were already enrolled and had stable disease or better could remain on selinexor therapy.
Now, the FDA has lifted the hold on trials of patients with hematologic malignancies, so new patients can be enrolled in these trials and begin receiving selinexor.
The FDA had placed the hold due to a lack of information in the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, noted that the hold was not the result of patient deaths or any new information regarding the safety profile of selinexor.
In response to the hold, Karyopharm amended the investigator’s brochure, updated informed consent documents, and submitted the documents to the FDA.
“The Karyopharm team worked diligently to update and submit the required documents to the FDA, which allowed the hematology division to expeditiously remove the partial clinical hold,” said Michael G. Kauffman, MD, PhD, chief executive officer of Karyopharm.
“We anticipate that the solid tumor divisions will follow suit shortly. Patient enrollment is again underway in our hematologic oncology studies. Our previously disclosed enrollment rates and timelines for both ongoing and planned trials are not expected to be materially impacted.”
About selinexor
Selinexor is a first-in-class, oral, selective inhibitor of nuclear export compound. The drug functions by inhibiting the nuclear export protein XPO1 (also called CRM1).
This leads to the accumulation of tumor suppressor proteins in the cell nucleus, which subsequently reinitiates and amplifies their tumor suppressor function. This is thought to prompt apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor.
The drug is currently being evaluated in clinical trials across multiple cancer indications, including in acute myeloid leukemia (SOPRA), in multiple myeloma in combination with low-dose dexamethasone (STORM) and backbone therapies (STOMP), as well as in diffuse large B-cell lymphoma (SADAL).
Phase 2 study of daratumumab in NHL won’t proceed
The phase 2 CARINA study of daratumumab in non-Hodgkin lymphoma (NHL) will not proceed to stage 2, according to Genmab A/S and Janssen Biotech, Inc.
In this study, researchers have been investigating daratumumab monotherapy in patients with relapsed or refractory follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), or mantle cell lymphoma (MCL).
Researchers planned to enroll up to 210 patients in this trial in 2 stages. Stage 1 was designed to provide a preliminary assessment of activity.
The goal of stage 2 was to further evaluate the safety and efficacy of daratumumab in the 3 patient groups.
Stage 2 will not proceed because a data review showed the FL and DLBCL cohorts did not reach the predefined futility thresholds, which were overall response rates of 50% and 30%, respectively. In the MCL cohort, the overall response rate was not evaluable due to slow recruitment.
The decision regarding this study has no impact on other ongoing or planned studies with daratumumab.
“While we hoped that daratumumab as a monotherapy could potentially provide a new treatment option in NHL patients with a high unmet medical need, the preliminary activity profile seen was not sufficient for the study to continue,” said Jan van de Winkel, PhD, chief executive officer of Genmab.
“Daratumumab is still being investigated in a number of indications, including multiple myeloma and other hematological cancers, such as NK/T-cell lymphoma and myelodysplastic syndrome, as well as in solid tumors.”
About daratumumab
Daratumumab is a human IgG1k monoclonal antibody that binds to the CD38 molecule.
In the US, daratumumab is approved for use in combination with lenalidomide and dexamethasone, or bortezomib and dexamethasone, for the treatment of patients with multiple myeloma who have received at least 1 prior therapy.
Daratumumab monotherapy is approved in the US for patients with multiple myeloma who have received at least 3 prior lines of therapy, including a proteasome inhibitor and an immunomodulatory agent, or who are double-refractory to a proteasome inhibitor and an immunomodulatory agent.
Daratumumab is being developed by Janssen Biotech, Inc. under an exclusive worldwide license from Genmab. ![]()
The phase 2 CARINA study of daratumumab in non-Hodgkin lymphoma (NHL) will not proceed to stage 2, according to Genmab A/S and Janssen Biotech, Inc.
In this study, researchers have been investigating daratumumab monotherapy in patients with relapsed or refractory follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), or mantle cell lymphoma (MCL).
Researchers planned to enroll up to 210 patients in this trial in 2 stages. Stage 1 was designed to provide a preliminary assessment of activity.
The goal of stage 2 was to further evaluate the safety and efficacy of daratumumab in the 3 patient groups.
Stage 2 will not proceed because a data review showed the FL and DLBCL cohorts did not reach the predefined futility thresholds, which were overall response rates of 50% and 30%, respectively. In the MCL cohort, the overall response rate was not evaluable due to slow recruitment.
The decision regarding this study has no impact on other ongoing or planned studies with daratumumab.
“While we hoped that daratumumab as a monotherapy could potentially provide a new treatment option in NHL patients with a high unmet medical need, the preliminary activity profile seen was not sufficient for the study to continue,” said Jan van de Winkel, PhD, chief executive officer of Genmab.
“Daratumumab is still being investigated in a number of indications, including multiple myeloma and other hematological cancers, such as NK/T-cell lymphoma and myelodysplastic syndrome, as well as in solid tumors.”
About daratumumab
Daratumumab is a human IgG1k monoclonal antibody that binds to the CD38 molecule.
In the US, daratumumab is approved for use in combination with lenalidomide and dexamethasone, or bortezomib and dexamethasone, for the treatment of patients with multiple myeloma who have received at least 1 prior therapy.
Daratumumab monotherapy is approved in the US for patients with multiple myeloma who have received at least 3 prior lines of therapy, including a proteasome inhibitor and an immunomodulatory agent, or who are double-refractory to a proteasome inhibitor and an immunomodulatory agent.
Daratumumab is being developed by Janssen Biotech, Inc. under an exclusive worldwide license from Genmab. ![]()
The phase 2 CARINA study of daratumumab in non-Hodgkin lymphoma (NHL) will not proceed to stage 2, according to Genmab A/S and Janssen Biotech, Inc.
In this study, researchers have been investigating daratumumab monotherapy in patients with relapsed or refractory follicular lymphoma (FL), diffuse large B-cell lymphoma (DLBCL), or mantle cell lymphoma (MCL).
Researchers planned to enroll up to 210 patients in this trial in 2 stages. Stage 1 was designed to provide a preliminary assessment of activity.
The goal of stage 2 was to further evaluate the safety and efficacy of daratumumab in the 3 patient groups.
Stage 2 will not proceed because a data review showed the FL and DLBCL cohorts did not reach the predefined futility thresholds, which were overall response rates of 50% and 30%, respectively. In the MCL cohort, the overall response rate was not evaluable due to slow recruitment.
The decision regarding this study has no impact on other ongoing or planned studies with daratumumab.
“While we hoped that daratumumab as a monotherapy could potentially provide a new treatment option in NHL patients with a high unmet medical need, the preliminary activity profile seen was not sufficient for the study to continue,” said Jan van de Winkel, PhD, chief executive officer of Genmab.
“Daratumumab is still being investigated in a number of indications, including multiple myeloma and other hematological cancers, such as NK/T-cell lymphoma and myelodysplastic syndrome, as well as in solid tumors.”
About daratumumab
Daratumumab is a human IgG1k monoclonal antibody that binds to the CD38 molecule.
In the US, daratumumab is approved for use in combination with lenalidomide and dexamethasone, or bortezomib and dexamethasone, for the treatment of patients with multiple myeloma who have received at least 1 prior therapy.
Daratumumab monotherapy is approved in the US for patients with multiple myeloma who have received at least 3 prior lines of therapy, including a proteasome inhibitor and an immunomodulatory agent, or who are double-refractory to a proteasome inhibitor and an immunomodulatory agent.
Daratumumab is being developed by Janssen Biotech, Inc. under an exclusive worldwide license from Genmab. ![]()
NCCN launches radiation therapy resource
The National Comprehensive Cancer Network® (NCCN®) recently launched the NCCN Radiation Therapy Compendium™, which provides a single access point for NCCN recommendations pertaining to radiation therapy.
The compendium provides guidance on all radiation therapy modalities recommended within NCCN guidelines, including intensity modulated radiation therapy, intra-operative radiation therapy, stereotactic radiosurgery/stereotactic body radiotherapy/stereotactic ablative radiotherapy, image-guided radiotherapy, low dose-rate brachytherapy/high dose-rate brachytherapy, radioisotope, and particle therapy.
“As a single source for all radiation therapy recommendations within the NCCN guidelines, the compendium benefits patients with cancer by assisting providers and payers in making evidence-based treatment and coverage decisions,” said Robert W. Carlson, MD, chief executive officer of NCCN.
The NCCN Radiation Therapy Compendium™ includes recommendations for the following 24 cancer types:
Acute myeloid leukemia
Anal cancer
B-cell lymphomas
Bladder cancer
Breast cancer
Chronic lymphocytic leukemia/small lymphoblastic lymphoma
Colon cancer
Hepatobiliary cancers
Kidney cancer
Malignant pleural mesothelioma
Melanoma
Multiple myeloma
Neuroendocrine tumors
Non-small cell lung cancer
Occult primary cancer
Pancreatic adenocarcinoma
Penile cancer
Primary cutaneous B-cell lymphomas
Prostate cancer
Rectal cancer
Small cell lung cancer
Soft tissue sarcoma
T-cell lymphomas
Testicular cancer
NCCN said additional cancer types will be published on a rolling basis over the coming months. ![]()
The National Comprehensive Cancer Network® (NCCN®) recently launched the NCCN Radiation Therapy Compendium™, which provides a single access point for NCCN recommendations pertaining to radiation therapy.
The compendium provides guidance on all radiation therapy modalities recommended within NCCN guidelines, including intensity modulated radiation therapy, intra-operative radiation therapy, stereotactic radiosurgery/stereotactic body radiotherapy/stereotactic ablative radiotherapy, image-guided radiotherapy, low dose-rate brachytherapy/high dose-rate brachytherapy, radioisotope, and particle therapy.
“As a single source for all radiation therapy recommendations within the NCCN guidelines, the compendium benefits patients with cancer by assisting providers and payers in making evidence-based treatment and coverage decisions,” said Robert W. Carlson, MD, chief executive officer of NCCN.
The NCCN Radiation Therapy Compendium™ includes recommendations for the following 24 cancer types:
Acute myeloid leukemia
Anal cancer
B-cell lymphomas
Bladder cancer
Breast cancer
Chronic lymphocytic leukemia/small lymphoblastic lymphoma
Colon cancer
Hepatobiliary cancers
Kidney cancer
Malignant pleural mesothelioma
Melanoma
Multiple myeloma
Neuroendocrine tumors
Non-small cell lung cancer
Occult primary cancer
Pancreatic adenocarcinoma
Penile cancer
Primary cutaneous B-cell lymphomas
Prostate cancer
Rectal cancer
Small cell lung cancer
Soft tissue sarcoma
T-cell lymphomas
Testicular cancer
NCCN said additional cancer types will be published on a rolling basis over the coming months. ![]()
The National Comprehensive Cancer Network® (NCCN®) recently launched the NCCN Radiation Therapy Compendium™, which provides a single access point for NCCN recommendations pertaining to radiation therapy.
The compendium provides guidance on all radiation therapy modalities recommended within NCCN guidelines, including intensity modulated radiation therapy, intra-operative radiation therapy, stereotactic radiosurgery/stereotactic body radiotherapy/stereotactic ablative radiotherapy, image-guided radiotherapy, low dose-rate brachytherapy/high dose-rate brachytherapy, radioisotope, and particle therapy.
“As a single source for all radiation therapy recommendations within the NCCN guidelines, the compendium benefits patients with cancer by assisting providers and payers in making evidence-based treatment and coverage decisions,” said Robert W. Carlson, MD, chief executive officer of NCCN.
The NCCN Radiation Therapy Compendium™ includes recommendations for the following 24 cancer types:
Acute myeloid leukemia
Anal cancer
B-cell lymphomas
Bladder cancer
Breast cancer
Chronic lymphocytic leukemia/small lymphoblastic lymphoma
Colon cancer
Hepatobiliary cancers
Kidney cancer
Malignant pleural mesothelioma
Melanoma
Multiple myeloma
Neuroendocrine tumors
Non-small cell lung cancer
Occult primary cancer
Pancreatic adenocarcinoma
Penile cancer
Primary cutaneous B-cell lymphomas
Prostate cancer
Rectal cancer
Small cell lung cancer
Soft tissue sarcoma
T-cell lymphomas
Testicular cancer
NCCN said additional cancer types will be published on a rolling basis over the coming months. ![]()