User login
EC grants drug orphan designation for CTCL
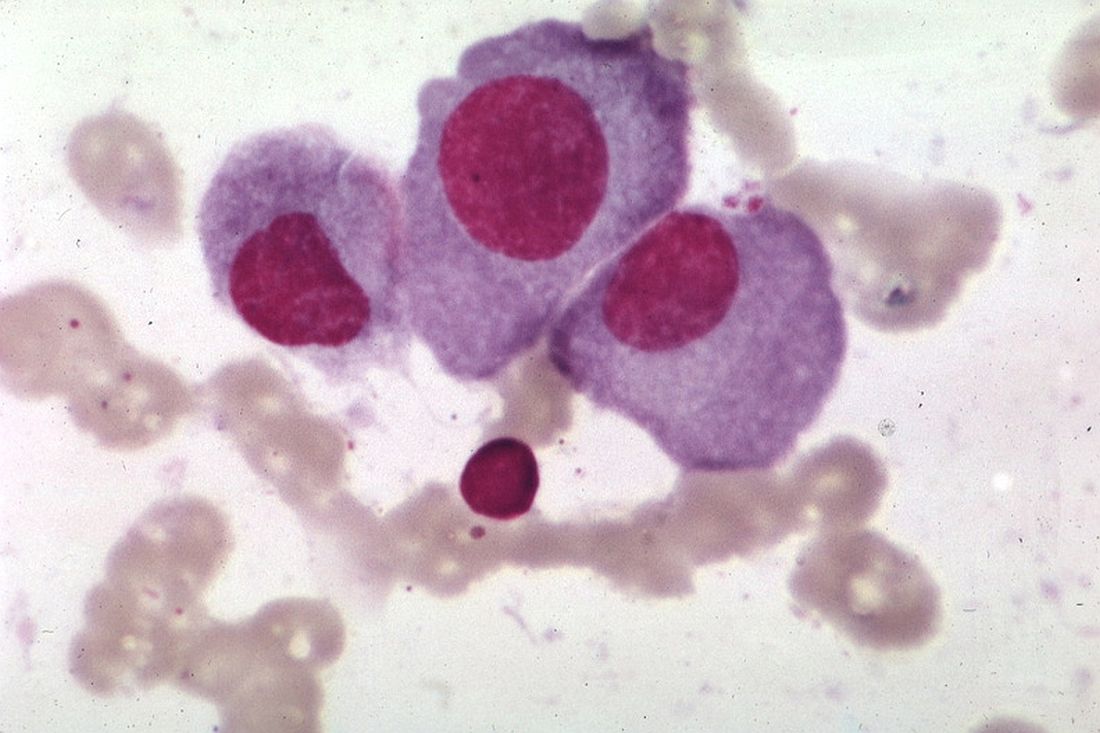
The European Commission (EC) has granted orphan designation to MRG-106 for the treatment of cutaneous T-cell lymphoma (CTCL).
MRG-106 is a locked nucleic acid-modified oligonucleotide inhibitor of miR-155-5p.
miRagen Therapeutics, Inc., the company developing MRG-106, is currently testing the drug in a phase 1 trial of CTCL patients.
Early results from this trial were presented at the 2016 ASH Annual Meeting.
Researchers presented results in 6 patients with stage I-III mycosis fungoides.
The patients received 4 or 5 intratumoral injections of MRG-106 (at 75 mg) over 2 weeks. Four patients received saline injections in a second lesion on the same schedule.
There were 3 adverse events related to MRG-106—pain during injection, burning sensation during injection, and tingling at the injection site.
Adverse events considered possibly related to MRG-106 were pruritus, erythema, skin inflammation, sore on hand, nausea, decrease in white blood cells, neutropenia, and prolonged partial thromboplastin time.
One patient was taken off the trial due to rapid disease progression. The other 5 patients completed the dosing period.
All 5 patients had a reduction in the baseline Composite Assessment of Index Lesion Severity score in MRG-106-treated and saline-treated lesions.
The average maximal reduction was 55% (range, 33% to 77%) in MRG-106-treated lesions and 39% (range, 13% to 75%) in saline-treated lesions.
About orphan designation
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
The European Medicines Agency adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the EC for a final decision. The EC typically makes a decision within 30 days of that submission. ![]()
The European Commission (EC) has granted orphan designation to MRG-106 for the treatment of cutaneous T-cell lymphoma (CTCL).
MRG-106 is a locked nucleic acid-modified oligonucleotide inhibitor of miR-155-5p.
miRagen Therapeutics, Inc., the company developing MRG-106, is currently testing the drug in a phase 1 trial of CTCL patients.
Early results from this trial were presented at the 2016 ASH Annual Meeting.
Researchers presented results in 6 patients with stage I-III mycosis fungoides.
The patients received 4 or 5 intratumoral injections of MRG-106 (at 75 mg) over 2 weeks. Four patients received saline injections in a second lesion on the same schedule.
There were 3 adverse events related to MRG-106—pain during injection, burning sensation during injection, and tingling at the injection site.
Adverse events considered possibly related to MRG-106 were pruritus, erythema, skin inflammation, sore on hand, nausea, decrease in white blood cells, neutropenia, and prolonged partial thromboplastin time.
One patient was taken off the trial due to rapid disease progression. The other 5 patients completed the dosing period.
All 5 patients had a reduction in the baseline Composite Assessment of Index Lesion Severity score in MRG-106-treated and saline-treated lesions.
The average maximal reduction was 55% (range, 33% to 77%) in MRG-106-treated lesions and 39% (range, 13% to 75%) in saline-treated lesions.
About orphan designation
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
The European Medicines Agency adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the EC for a final decision. The EC typically makes a decision within 30 days of that submission. ![]()
The European Commission (EC) has granted orphan designation to MRG-106 for the treatment of cutaneous T-cell lymphoma (CTCL).
MRG-106 is a locked nucleic acid-modified oligonucleotide inhibitor of miR-155-5p.
miRagen Therapeutics, Inc., the company developing MRG-106, is currently testing the drug in a phase 1 trial of CTCL patients.
Early results from this trial were presented at the 2016 ASH Annual Meeting.
Researchers presented results in 6 patients with stage I-III mycosis fungoides.
The patients received 4 or 5 intratumoral injections of MRG-106 (at 75 mg) over 2 weeks. Four patients received saline injections in a second lesion on the same schedule.
There were 3 adverse events related to MRG-106—pain during injection, burning sensation during injection, and tingling at the injection site.
Adverse events considered possibly related to MRG-106 were pruritus, erythema, skin inflammation, sore on hand, nausea, decrease in white blood cells, neutropenia, and prolonged partial thromboplastin time.
One patient was taken off the trial due to rapid disease progression. The other 5 patients completed the dosing period.
All 5 patients had a reduction in the baseline Composite Assessment of Index Lesion Severity score in MRG-106-treated and saline-treated lesions.
The average maximal reduction was 55% (range, 33% to 77%) in MRG-106-treated lesions and 39% (range, 13% to 75%) in saline-treated lesions.
About orphan designation
Orphan designation provides regulatory and financial incentives for companies to develop and market therapies that treat life-threatening or chronically debilitating conditions affecting no more than 5 in 10,000 people in the European Union, and where no satisfactory treatment is available.
Orphan designation provides a 10-year period of marketing exclusivity if the drug receives regulatory approval.
The designation also provides incentives for companies seeking protocol assistance from the European Medicines Agency during the product development phase and direct access to the centralized authorization procedure.
The European Medicines Agency adopts an opinion on the granting of orphan drug designation, and that opinion is submitted to the EC for a final decision. The EC typically makes a decision within 30 days of that submission. ![]()
Why fewer blood cancer patients receive hospice care
New research provides an explanation for the fact that US patients with hematologic malignancies are less likely to enroll in hospice care than patients with solid tumor malignancies.
Results of a national survey suggest that concerns about the adequacy of hospice may prevent hematologic oncologists from referring their patients.
Researchers say this finding, published in Cancer, points to potential means of improving end-of-life care for patients with hematologic malignancies.
Oreofe Odejide, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues carried out this study.
The team conducted a survey of a national sample of hematologic oncologists listed in the publicly available clinical directory of the American Society of Hematology.
More than 57% of physicians who were contacted provided responses, for a total of 349 respondents.
The survey included questions about views regarding the helpfulness and adequacy of home hospice services for patients with hematologic malignancies, as well as factors that would impact oncologists’ likelihood of referring patients to hospice.
More than 68% of hematologic oncologists strongly agreed that hospice care is “helpful” for patients with hematologic malignancies.
However, 46% of the oncologists felt that home hospice is “inadequate” for the needs of patients with hematologic malignancies, when compared to inpatient hospice.
Still, most of the respondents who believed home hospice is inadequate said they would be more likely to refer patients if platelet and red blood cell transfusions were readily available.
“Our findings are important as they shed light on factors that are potential barriers to hospice referrals,” Dr Odejide said. “These findings can be employed to develop targeted interventions to address hospice underuse for patients with blood cancers.” ![]()
New research provides an explanation for the fact that US patients with hematologic malignancies are less likely to enroll in hospice care than patients with solid tumor malignancies.
Results of a national survey suggest that concerns about the adequacy of hospice may prevent hematologic oncologists from referring their patients.
Researchers say this finding, published in Cancer, points to potential means of improving end-of-life care for patients with hematologic malignancies.
Oreofe Odejide, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues carried out this study.
The team conducted a survey of a national sample of hematologic oncologists listed in the publicly available clinical directory of the American Society of Hematology.
More than 57% of physicians who were contacted provided responses, for a total of 349 respondents.
The survey included questions about views regarding the helpfulness and adequacy of home hospice services for patients with hematologic malignancies, as well as factors that would impact oncologists’ likelihood of referring patients to hospice.
More than 68% of hematologic oncologists strongly agreed that hospice care is “helpful” for patients with hematologic malignancies.
However, 46% of the oncologists felt that home hospice is “inadequate” for the needs of patients with hematologic malignancies, when compared to inpatient hospice.
Still, most of the respondents who believed home hospice is inadequate said they would be more likely to refer patients if platelet and red blood cell transfusions were readily available.
“Our findings are important as they shed light on factors that are potential barriers to hospice referrals,” Dr Odejide said. “These findings can be employed to develop targeted interventions to address hospice underuse for patients with blood cancers.” ![]()
New research provides an explanation for the fact that US patients with hematologic malignancies are less likely to enroll in hospice care than patients with solid tumor malignancies.
Results of a national survey suggest that concerns about the adequacy of hospice may prevent hematologic oncologists from referring their patients.
Researchers say this finding, published in Cancer, points to potential means of improving end-of-life care for patients with hematologic malignancies.
Oreofe Odejide, MD, of the Dana-Farber Cancer Institute in Boston, Massachusetts, and her colleagues carried out this study.
The team conducted a survey of a national sample of hematologic oncologists listed in the publicly available clinical directory of the American Society of Hematology.
More than 57% of physicians who were contacted provided responses, for a total of 349 respondents.
The survey included questions about views regarding the helpfulness and adequacy of home hospice services for patients with hematologic malignancies, as well as factors that would impact oncologists’ likelihood of referring patients to hospice.
More than 68% of hematologic oncologists strongly agreed that hospice care is “helpful” for patients with hematologic malignancies.
However, 46% of the oncologists felt that home hospice is “inadequate” for the needs of patients with hematologic malignancies, when compared to inpatient hospice.
Still, most of the respondents who believed home hospice is inadequate said they would be more likely to refer patients if platelet and red blood cell transfusions were readily available.
“Our findings are important as they shed light on factors that are potential barriers to hospice referrals,” Dr Odejide said. “These findings can be employed to develop targeted interventions to address hospice underuse for patients with blood cancers.” ![]()
Global study reveals healthcare inequity, preventable deaths
A global study has revealed inequity of access to and quality of healthcare among and within countries and suggests people are dying from causes with well-known treatments.
“What we have found about healthcare access and quality is disturbing,” said Christopher Murray, MD, DPhil, of the University of Washington in Seattle.
“Having a strong economy does not guarantee good healthcare. Having great medical technology doesn’t either. We know this because people are not getting the care that should be expected for diseases with established treatments.”
Dr Murray and his colleagues reported these findings in The Lancet.
For this study, the researchers assessed access to and quality of healthcare services in 195 countries from 1990 to 2015.
The group used the Healthcare Access and Quality Index, a summary measure based on 32 causes* that, in the presence of high-quality healthcare, should not result in death. Leukemia and Hodgkin lymphoma are among these causes.
Countries were assigned scores for each of the causes, based on estimates from the annual Global Burden of Diseases, Injuries, and Risk Factors study (GBD), a systematic, scientific effort to quantify the magnitude of health loss from all major diseases, injuries, and risk factors by age, sex, and population.
In addition, data were extracted from the most recent GBD update and evaluated using a Socio-demographic Index based on rates of education, fertility, and income.
Results
The 195 countries were assigned scores on a scale of 1 to 100 for healthcare access and quality. They received scores for the 32 causes as well as overall scores.
In 2015, the top-ranked nation was Andorra, with an overall score of 95. Its lowest treatment score was 70, for Hodgkin lymphoma.
The lowest-ranked nation was Central African Republic, with a score of 29. Its highest treatment score was 65, for diphtheria.
Nations in much of sub-Saharan Africa, as well as in south Asia and several countries in Latin America and the Caribbean, also had low rankings.
However, many countries in these regions, including China (score: 74) and Ethiopia (score: 44), have seen sizeable gains since 1990.
‘Developed’ nations falling short
The US had an overall score of 81 (in 2015), tied with Estonia and Montenegro. As with many other nations, the US scored 100 in treating common vaccine-preventable diseases, such as diphtheria, tetanus, and measles.
However, the US had 9 treatment categories in which it scored in the 60s: lower respiratory infections (60), neonatal disorders (69), non-melanoma skin cancer (68), Hodgkin lymphoma (67), ischemic heart disease (62), hypertensive heart disease (64), diabetes (67), chronic kidney disease (62), and the adverse effects of medical treatment itself (68).
“America’s ranking is an embarrassment, especially considering the US spends more than $9000 per person on healthcare annually, more than any other country,” Dr Murray said.
“Anyone with a stake in the current healthcare debate, including elected officials at the federal, state, and local levels, should take a look at where the US is falling short.”
Other nations with strong economies and advanced medical technology are falling short in some areas as well.
For example, Norway and Australia each scored 90 overall, among the highest in the world. However, Norway scored 65 in its treatment for testicular cancer, and Australia scored 52 for treating non-melanoma skin cancer.
“In the majority of cases, both of these cancers can be treated effectively,” Dr Murray said. “Shouldn’t it cause serious concern that people are dying of these causes in countries that have the resources to address them?” ![]()
*The 32 causes are:
- Adverse effects of medical treatment
- Appendicitis
- Breast cancer
- Cerebrovascular disease (stroke)
- Cervical cancer
- Chronic kidney disease
- Chronic respiratory diseases
- Colon and rectum cancer
- Congenital anomalies
- Diabetes mellitus
- Diarrhea-related diseases
- Diphtheria
- Epilepsy
- Gallbladder and biliary diseases
- Hodgkin lymphoma
- Hypertensive heart disease
- Inguinal, femoral, and abdominal hernia
- Ischemic heart disease
- Leukemia
- Lower respiratory infections
- Maternal disorders
- Measles
- Neonatal disorders
- Non-melanoma skin cancer
- Peptic ulcer disease
- Rheumatic heart disease
- Testicular cancer
- Tetanus
- Tuberculosis
- Upper respiratory infections
- Uterine cancer
- Whooping cough.
A global study has revealed inequity of access to and quality of healthcare among and within countries and suggests people are dying from causes with well-known treatments.
“What we have found about healthcare access and quality is disturbing,” said Christopher Murray, MD, DPhil, of the University of Washington in Seattle.
“Having a strong economy does not guarantee good healthcare. Having great medical technology doesn’t either. We know this because people are not getting the care that should be expected for diseases with established treatments.”
Dr Murray and his colleagues reported these findings in The Lancet.
For this study, the researchers assessed access to and quality of healthcare services in 195 countries from 1990 to 2015.
The group used the Healthcare Access and Quality Index, a summary measure based on 32 causes* that, in the presence of high-quality healthcare, should not result in death. Leukemia and Hodgkin lymphoma are among these causes.
Countries were assigned scores for each of the causes, based on estimates from the annual Global Burden of Diseases, Injuries, and Risk Factors study (GBD), a systematic, scientific effort to quantify the magnitude of health loss from all major diseases, injuries, and risk factors by age, sex, and population.
In addition, data were extracted from the most recent GBD update and evaluated using a Socio-demographic Index based on rates of education, fertility, and income.
Results
The 195 countries were assigned scores on a scale of 1 to 100 for healthcare access and quality. They received scores for the 32 causes as well as overall scores.
In 2015, the top-ranked nation was Andorra, with an overall score of 95. Its lowest treatment score was 70, for Hodgkin lymphoma.
The lowest-ranked nation was Central African Republic, with a score of 29. Its highest treatment score was 65, for diphtheria.
Nations in much of sub-Saharan Africa, as well as in south Asia and several countries in Latin America and the Caribbean, also had low rankings.
However, many countries in these regions, including China (score: 74) and Ethiopia (score: 44), have seen sizeable gains since 1990.
‘Developed’ nations falling short
The US had an overall score of 81 (in 2015), tied with Estonia and Montenegro. As with many other nations, the US scored 100 in treating common vaccine-preventable diseases, such as diphtheria, tetanus, and measles.
However, the US had 9 treatment categories in which it scored in the 60s: lower respiratory infections (60), neonatal disorders (69), non-melanoma skin cancer (68), Hodgkin lymphoma (67), ischemic heart disease (62), hypertensive heart disease (64), diabetes (67), chronic kidney disease (62), and the adverse effects of medical treatment itself (68).
“America’s ranking is an embarrassment, especially considering the US spends more than $9000 per person on healthcare annually, more than any other country,” Dr Murray said.
“Anyone with a stake in the current healthcare debate, including elected officials at the federal, state, and local levels, should take a look at where the US is falling short.”
Other nations with strong economies and advanced medical technology are falling short in some areas as well.
For example, Norway and Australia each scored 90 overall, among the highest in the world. However, Norway scored 65 in its treatment for testicular cancer, and Australia scored 52 for treating non-melanoma skin cancer.
“In the majority of cases, both of these cancers can be treated effectively,” Dr Murray said. “Shouldn’t it cause serious concern that people are dying of these causes in countries that have the resources to address them?” ![]()
*The 32 causes are:
- Adverse effects of medical treatment
- Appendicitis
- Breast cancer
- Cerebrovascular disease (stroke)
- Cervical cancer
- Chronic kidney disease
- Chronic respiratory diseases
- Colon and rectum cancer
- Congenital anomalies
- Diabetes mellitus
- Diarrhea-related diseases
- Diphtheria
- Epilepsy
- Gallbladder and biliary diseases
- Hodgkin lymphoma
- Hypertensive heart disease
- Inguinal, femoral, and abdominal hernia
- Ischemic heart disease
- Leukemia
- Lower respiratory infections
- Maternal disorders
- Measles
- Neonatal disorders
- Non-melanoma skin cancer
- Peptic ulcer disease
- Rheumatic heart disease
- Testicular cancer
- Tetanus
- Tuberculosis
- Upper respiratory infections
- Uterine cancer
- Whooping cough.
A global study has revealed inequity of access to and quality of healthcare among and within countries and suggests people are dying from causes with well-known treatments.
“What we have found about healthcare access and quality is disturbing,” said Christopher Murray, MD, DPhil, of the University of Washington in Seattle.
“Having a strong economy does not guarantee good healthcare. Having great medical technology doesn’t either. We know this because people are not getting the care that should be expected for diseases with established treatments.”
Dr Murray and his colleagues reported these findings in The Lancet.
For this study, the researchers assessed access to and quality of healthcare services in 195 countries from 1990 to 2015.
The group used the Healthcare Access and Quality Index, a summary measure based on 32 causes* that, in the presence of high-quality healthcare, should not result in death. Leukemia and Hodgkin lymphoma are among these causes.
Countries were assigned scores for each of the causes, based on estimates from the annual Global Burden of Diseases, Injuries, and Risk Factors study (GBD), a systematic, scientific effort to quantify the magnitude of health loss from all major diseases, injuries, and risk factors by age, sex, and population.
In addition, data were extracted from the most recent GBD update and evaluated using a Socio-demographic Index based on rates of education, fertility, and income.
Results
The 195 countries were assigned scores on a scale of 1 to 100 for healthcare access and quality. They received scores for the 32 causes as well as overall scores.
In 2015, the top-ranked nation was Andorra, with an overall score of 95. Its lowest treatment score was 70, for Hodgkin lymphoma.
The lowest-ranked nation was Central African Republic, with a score of 29. Its highest treatment score was 65, for diphtheria.
Nations in much of sub-Saharan Africa, as well as in south Asia and several countries in Latin America and the Caribbean, also had low rankings.
However, many countries in these regions, including China (score: 74) and Ethiopia (score: 44), have seen sizeable gains since 1990.
‘Developed’ nations falling short
The US had an overall score of 81 (in 2015), tied with Estonia and Montenegro. As with many other nations, the US scored 100 in treating common vaccine-preventable diseases, such as diphtheria, tetanus, and measles.
However, the US had 9 treatment categories in which it scored in the 60s: lower respiratory infections (60), neonatal disorders (69), non-melanoma skin cancer (68), Hodgkin lymphoma (67), ischemic heart disease (62), hypertensive heart disease (64), diabetes (67), chronic kidney disease (62), and the adverse effects of medical treatment itself (68).
“America’s ranking is an embarrassment, especially considering the US spends more than $9000 per person on healthcare annually, more than any other country,” Dr Murray said.
“Anyone with a stake in the current healthcare debate, including elected officials at the federal, state, and local levels, should take a look at where the US is falling short.”
Other nations with strong economies and advanced medical technology are falling short in some areas as well.
For example, Norway and Australia each scored 90 overall, among the highest in the world. However, Norway scored 65 in its treatment for testicular cancer, and Australia scored 52 for treating non-melanoma skin cancer.
“In the majority of cases, both of these cancers can be treated effectively,” Dr Murray said. “Shouldn’t it cause serious concern that people are dying of these causes in countries that have the resources to address them?” ![]()
*The 32 causes are:
- Adverse effects of medical treatment
- Appendicitis
- Breast cancer
- Cerebrovascular disease (stroke)
- Cervical cancer
- Chronic kidney disease
- Chronic respiratory diseases
- Colon and rectum cancer
- Congenital anomalies
- Diabetes mellitus
- Diarrhea-related diseases
- Diphtheria
- Epilepsy
- Gallbladder and biliary diseases
- Hodgkin lymphoma
- Hypertensive heart disease
- Inguinal, femoral, and abdominal hernia
- Ischemic heart disease
- Leukemia
- Lower respiratory infections
- Maternal disorders
- Measles
- Neonatal disorders
- Non-melanoma skin cancer
- Peptic ulcer disease
- Rheumatic heart disease
- Testicular cancer
- Tetanus
- Tuberculosis
- Upper respiratory infections
- Uterine cancer
- Whooping cough.
ALC/AMC prognostic in mantle cell lymphoma
The peripheral blood absolute lymphocyte-to-monocyte ratio (ALC/AMC) was prognostic for overall survival in mantle cell lymphoma patients who have undergone induction therapy, based on a retrospective review study of 96 patients by Andre Goy, MD, of John Theurer Cancer Center, Hackensack (NJ) University, and his colleagues.
Overall survival was better when ALC/AMC was 2 or greater following induction therapy, the researchers wrote in an abstract published in conjunction with the annual meeting of the American Society of Clinical Oncology.
The finding indicates that novel maintenance programs, including targeting the microenvironment or immune response, might be appropriate when patients with mantle cell lymphoma have low ALC/AMC.
The researchers examined data for 96 consecutive mantle cell lymphoma patients. The ALC/AMC was determined from peripheral blood counts obtained approximately 30 days following completion of initial therapy or immediately prior to stem cell mobilization in patients who had first line stem cell transplants.
The ALC/AMC was less than 2 in 67 patients and was 2 or greater in 29 patients. The two patient cohorts were similar in median age, ethnicities, stage distributions, elevated beta-2-microglobulin, elevated lactate dehydrogenate, and Mantle Cell Lymphoma International Prognostic Index scores.
ALC/AMC was less than 2 in 10 of 13 transplanted patients and in 57 of 83 patients who did not undergo transplants. At a median follow-up of 43 months, the median overall survival has not been reached in either cohort.
The 5-year survival rate was 90% among patients with an ALC/AMC of 2 or greater and 68% in those with an ALC/AMC less than 2 (log-rank P less than .05).
Similar ALC/AMC 5-year survival trends were noted when subsetting to the 25 patients with high risk Mantle Cell Lymphoma International Prognostic Index scores (72% vs. 45%; P = .07).
Dr. Goy disclosed honoraria from Acerta Pharma, Celgene, Pharmacyclics, and Takeda; a consulting or advisory role with Acerta Pharma, Celgene, Infinity Pharmaceuticals, Pharmacyclics, and Takeda; and speakers’ bureaus participation for Pharmacyclics and Takeda.
Prognostic value of the absolute lymphocyte-to-monocyte (ALC/AMC) ratio on overall survival among patients with mantle cell lymphoma. Published in conjunction with the 2017 ASCO Annual Meeting. Abstract No: e19030.
mdales@frontlinemedcom.com
On Twitter @maryjodales
The peripheral blood absolute lymphocyte-to-monocyte ratio (ALC/AMC) was prognostic for overall survival in mantle cell lymphoma patients who have undergone induction therapy, based on a retrospective review study of 96 patients by Andre Goy, MD, of John Theurer Cancer Center, Hackensack (NJ) University, and his colleagues.
Overall survival was better when ALC/AMC was 2 or greater following induction therapy, the researchers wrote in an abstract published in conjunction with the annual meeting of the American Society of Clinical Oncology.
The finding indicates that novel maintenance programs, including targeting the microenvironment or immune response, might be appropriate when patients with mantle cell lymphoma have low ALC/AMC.
The researchers examined data for 96 consecutive mantle cell lymphoma patients. The ALC/AMC was determined from peripheral blood counts obtained approximately 30 days following completion of initial therapy or immediately prior to stem cell mobilization in patients who had first line stem cell transplants.
The ALC/AMC was less than 2 in 67 patients and was 2 or greater in 29 patients. The two patient cohorts were similar in median age, ethnicities, stage distributions, elevated beta-2-microglobulin, elevated lactate dehydrogenate, and Mantle Cell Lymphoma International Prognostic Index scores.
ALC/AMC was less than 2 in 10 of 13 transplanted patients and in 57 of 83 patients who did not undergo transplants. At a median follow-up of 43 months, the median overall survival has not been reached in either cohort.
The 5-year survival rate was 90% among patients with an ALC/AMC of 2 or greater and 68% in those with an ALC/AMC less than 2 (log-rank P less than .05).
Similar ALC/AMC 5-year survival trends were noted when subsetting to the 25 patients with high risk Mantle Cell Lymphoma International Prognostic Index scores (72% vs. 45%; P = .07).
Dr. Goy disclosed honoraria from Acerta Pharma, Celgene, Pharmacyclics, and Takeda; a consulting or advisory role with Acerta Pharma, Celgene, Infinity Pharmaceuticals, Pharmacyclics, and Takeda; and speakers’ bureaus participation for Pharmacyclics and Takeda.
Prognostic value of the absolute lymphocyte-to-monocyte (ALC/AMC) ratio on overall survival among patients with mantle cell lymphoma. Published in conjunction with the 2017 ASCO Annual Meeting. Abstract No: e19030.
mdales@frontlinemedcom.com
On Twitter @maryjodales
The peripheral blood absolute lymphocyte-to-monocyte ratio (ALC/AMC) was prognostic for overall survival in mantle cell lymphoma patients who have undergone induction therapy, based on a retrospective review study of 96 patients by Andre Goy, MD, of John Theurer Cancer Center, Hackensack (NJ) University, and his colleagues.
Overall survival was better when ALC/AMC was 2 or greater following induction therapy, the researchers wrote in an abstract published in conjunction with the annual meeting of the American Society of Clinical Oncology.
The finding indicates that novel maintenance programs, including targeting the microenvironment or immune response, might be appropriate when patients with mantle cell lymphoma have low ALC/AMC.
The researchers examined data for 96 consecutive mantle cell lymphoma patients. The ALC/AMC was determined from peripheral blood counts obtained approximately 30 days following completion of initial therapy or immediately prior to stem cell mobilization in patients who had first line stem cell transplants.
The ALC/AMC was less than 2 in 67 patients and was 2 or greater in 29 patients. The two patient cohorts were similar in median age, ethnicities, stage distributions, elevated beta-2-microglobulin, elevated lactate dehydrogenate, and Mantle Cell Lymphoma International Prognostic Index scores.
ALC/AMC was less than 2 in 10 of 13 transplanted patients and in 57 of 83 patients who did not undergo transplants. At a median follow-up of 43 months, the median overall survival has not been reached in either cohort.
The 5-year survival rate was 90% among patients with an ALC/AMC of 2 or greater and 68% in those with an ALC/AMC less than 2 (log-rank P less than .05).
Similar ALC/AMC 5-year survival trends were noted when subsetting to the 25 patients with high risk Mantle Cell Lymphoma International Prognostic Index scores (72% vs. 45%; P = .07).
Dr. Goy disclosed honoraria from Acerta Pharma, Celgene, Pharmacyclics, and Takeda; a consulting or advisory role with Acerta Pharma, Celgene, Infinity Pharmaceuticals, Pharmacyclics, and Takeda; and speakers’ bureaus participation for Pharmacyclics and Takeda.
Prognostic value of the absolute lymphocyte-to-monocyte (ALC/AMC) ratio on overall survival among patients with mantle cell lymphoma. Published in conjunction with the 2017 ASCO Annual Meeting. Abstract No: e19030.
mdales@frontlinemedcom.com
On Twitter @maryjodales
IN CONJUNCTION WITH ASCO 2017
Key clinical point:
Major finding: The 5-year survival rate was 90% among patients with an ALC/AMC of 2 or greater and 68% in those with an ALC/AMC less than 2 (log-rank P less than .05).
Data source: A retrospective review study of 96 patients.
Disclosures: Dr. Goy disclosed honoraria from Acerta Pharma, Celgene, Pharmacyclics, and Takeda; a consulting or advisory role with Acerta Pharma, Celgene, Infinity Pharmaceuticals, Pharmacyclics, and Takeda; and speakers’ bureaus participation for Pharmacyclics and Takeda.
Citation: Prognostic value of the absolute lymphocyte-to-monocyte (ALC/AMC) ratio on overall survival among patients with mantle cell lymphoma. Published in conjunction with the 2017 ASCO Annual Meeting. Abstract No: e19030.
Smoldering myeloma progressed more rapidly in patients with elevated BMIs
An elevated body mass index appears to be a risk factor for progression of smoldering multiple myeloma, according to Wilson I. Gonsalves, MD, and his colleagues at Mayo Clinic, Rochester, Minn.
The findings, based on median follow up data of 106 months from 306 patients diagnosed with smoldering multiple myeloma from 2000-2010 at the Mayo Clinic, need to be confirmed in larger studies. Nevertheless, the results imply that patient weight is a potentially modifiable risk factor for progression from smoldering disease to multiple myeloma, Dr. Gonsalves and his colleagues wrote.
At initial evaluation, 28% of patients had myeloma defining events, such as a serum free light chain ratio greater than 100 or over 60% clonal bone marrow plasma cells. Myeloma defining events were present in 17% of patients with normal BMIs and 33% of patients with elevated BMIs, a statistically significant difference (P = .011).
When the analysis was limited to the 187 patients without myeloma-defining events at initial evaluation, the 2-year rate of progression to symptomatic multiple myeloma was 15% in those with a normal BMI and 33% in those with an elevated BMI (P = .013).
In a multivariable model, only elevated BMI (P = .004) and increasing clonal bone marrow plasma cells (P = .001) were statistically significant in predicting 2-year progression to multiple myeloma.
At last follow-up, 66% of patients had progressed to symptomatic multiple myeloma.
Dr. Gonsalves had no relationships to disclose.
The impact of body mass index on the risk of early progression of smoldering multiple myeloma to symptomatic myeloma. 2017 ASCO annual meeting. Abstract No: 8032.
mdales@frontlinemedcom.com
On Twitter @maryjodales
An elevated body mass index appears to be a risk factor for progression of smoldering multiple myeloma, according to Wilson I. Gonsalves, MD, and his colleagues at Mayo Clinic, Rochester, Minn.
The findings, based on median follow up data of 106 months from 306 patients diagnosed with smoldering multiple myeloma from 2000-2010 at the Mayo Clinic, need to be confirmed in larger studies. Nevertheless, the results imply that patient weight is a potentially modifiable risk factor for progression from smoldering disease to multiple myeloma, Dr. Gonsalves and his colleagues wrote.
At initial evaluation, 28% of patients had myeloma defining events, such as a serum free light chain ratio greater than 100 or over 60% clonal bone marrow plasma cells. Myeloma defining events were present in 17% of patients with normal BMIs and 33% of patients with elevated BMIs, a statistically significant difference (P = .011).
When the analysis was limited to the 187 patients without myeloma-defining events at initial evaluation, the 2-year rate of progression to symptomatic multiple myeloma was 15% in those with a normal BMI and 33% in those with an elevated BMI (P = .013).
In a multivariable model, only elevated BMI (P = .004) and increasing clonal bone marrow plasma cells (P = .001) were statistically significant in predicting 2-year progression to multiple myeloma.
At last follow-up, 66% of patients had progressed to symptomatic multiple myeloma.
Dr. Gonsalves had no relationships to disclose.
The impact of body mass index on the risk of early progression of smoldering multiple myeloma to symptomatic myeloma. 2017 ASCO annual meeting. Abstract No: 8032.
mdales@frontlinemedcom.com
On Twitter @maryjodales
An elevated body mass index appears to be a risk factor for progression of smoldering multiple myeloma, according to Wilson I. Gonsalves, MD, and his colleagues at Mayo Clinic, Rochester, Minn.
The findings, based on median follow up data of 106 months from 306 patients diagnosed with smoldering multiple myeloma from 2000-2010 at the Mayo Clinic, need to be confirmed in larger studies. Nevertheless, the results imply that patient weight is a potentially modifiable risk factor for progression from smoldering disease to multiple myeloma, Dr. Gonsalves and his colleagues wrote.
At initial evaluation, 28% of patients had myeloma defining events, such as a serum free light chain ratio greater than 100 or over 60% clonal bone marrow plasma cells. Myeloma defining events were present in 17% of patients with normal BMIs and 33% of patients with elevated BMIs, a statistically significant difference (P = .011).
When the analysis was limited to the 187 patients without myeloma-defining events at initial evaluation, the 2-year rate of progression to symptomatic multiple myeloma was 15% in those with a normal BMI and 33% in those with an elevated BMI (P = .013).
In a multivariable model, only elevated BMI (P = .004) and increasing clonal bone marrow plasma cells (P = .001) were statistically significant in predicting 2-year progression to multiple myeloma.
At last follow-up, 66% of patients had progressed to symptomatic multiple myeloma.
Dr. Gonsalves had no relationships to disclose.
The impact of body mass index on the risk of early progression of smoldering multiple myeloma to symptomatic myeloma. 2017 ASCO annual meeting. Abstract No: 8032.
mdales@frontlinemedcom.com
On Twitter @maryjodales
FROM ASCO 2017
Key clinical point:
Major finding: The 2-year rate of progression from smoldering disease to symptomatic multiple myeloma was 16% in patients with a normal BMI and 42% in patients with an elevated BMI (P less than 0.0001).
Data source: Median follow up data of 106 months from 306 patients diagnosed with smoldering multiple myeloma during 2000-2010 at the Mayo Clinic.
Disclosures: Dr. Gonsalves had no relationships to disclose.
Citation: The impact of body mass index on the risk of early progression of smoldering multiple myeloma to symptomatic myeloma. 2017 ASCO annual meeting. Abstract No: 8032.
Myeloma patients who get solid tumor cancers do as well as other cancer patients
With improved treatment, patients with multiple myeloma are surviving long enough to develop other cancers, Jorge J. Castillo, MD, and Adam J. Olszewski, MD, reported in a poster to be presented at the annual meeting of the American Society of Clinical Oncology.
The good news is that myeloma patients, when diagnosed with a subsequent solid tumor, are just as likely to respond to treatment and do just as well as patients without myeloma, according to Dr. Castillo of Dana-Farber Cancer Institute, Boston, and Dr. Olszewski of Alpert Medical School of Brown University, Providence, R.I.
They based their conclusion on Surveillance, Epidemiology, and End Results (SEER) data for patients diagnosed with six common cancers from 2004-2013.
“Among them, we identified [nearly 1,300] myeloma survivors, and we matched each to 50 randomly sampled controls with the same cancer by age, sex, race, and year of diagnosis. We then compared [cancer specific survival], cumulative incidence function (CIF) for death from the non-myeloma index cancer, and whether patients had surgery for non-metastastic, stage-matched tumors only,” the researchers wrote in their abstract.
They did analyses for breast, lung, prostate, colorectal, melanoma, and bladder cancers. The median time from diagnosis of myeloma to diagnosis of the second ranged from 35 months (bladder [133 myeloma patients] and lung [286 myeloma patients] cancers) to 50 months (melanoma [140 myeloma patients]). The median time after myeloma diagnosis was 40 months for those patients who developed breast, prostate, or colorectal cancers.
In the comparisons, myeloma survivors were significantly older (P less than .001) than patients initially diagnosed with the same respective cancers. In the case-control analysis, breast (P = .002) and lung cancers (P = .003) were more often diagnosed at an early stage among myeloma survivors.
The hazard ratio (HR) for cancer-specific survival for 189 myeloma patients diagnosed with breast cancer as compared to other breast cancer patients, for example, was 0.99, 95% confidence interval (CI) 0.61-1.61. The HR for the cumulative incidence function of cancer death was 0.82, 95% CI 0.50-1.35.
Myeloma patients were no less likely than were case-control subjects to have surgery for their cancers, with the exception of the 330 myeloma patients who developed prostate cancer (odds ratio, 0.59, 95% CI, 0.44-0.81).
Cancer-specific survival significantly differed (P less than .05) only for lung cancer, and was better among the 286 myeloma patients with lung cancer even when stratified by stage (HR 0.64, 95% CI 0.54-0.75). For cumulative incidence function of cancer death for lung cancer, the hazard ratio was 0.52 (95% CI 0.44-0.61). Better outcomes in lung cancer are not fully explained by earlier detection, suggesting a biological difference, the researchers reported.
Cumulative incidence function of cancer death was significantly lower for myeloma patients with lung and colorectal cancers.
Dr. Castillo disclosed honoraria from Celgene and Janssen; a consulting or advisory role with Biogen, Otsuka, and Pharmacyclics; and institutional research funding from Abbvie, Gilead Sciences, Millennium, and Pharmacyclics. Dr. Olszewski disclosed institutional research funding from Genentech, Incyte, and TG Therapeutics.
Citation: Outcomes of secondary cancers among myeloma survivors. 2017 ASCO annual meeting. Abstract No. 8043.
mdales@frontlinemedcom.com
On Twitter @maryjodales
With improved treatment, patients with multiple myeloma are surviving long enough to develop other cancers, Jorge J. Castillo, MD, and Adam J. Olszewski, MD, reported in a poster to be presented at the annual meeting of the American Society of Clinical Oncology.
The good news is that myeloma patients, when diagnosed with a subsequent solid tumor, are just as likely to respond to treatment and do just as well as patients without myeloma, according to Dr. Castillo of Dana-Farber Cancer Institute, Boston, and Dr. Olszewski of Alpert Medical School of Brown University, Providence, R.I.
They based their conclusion on Surveillance, Epidemiology, and End Results (SEER) data for patients diagnosed with six common cancers from 2004-2013.
“Among them, we identified [nearly 1,300] myeloma survivors, and we matched each to 50 randomly sampled controls with the same cancer by age, sex, race, and year of diagnosis. We then compared [cancer specific survival], cumulative incidence function (CIF) for death from the non-myeloma index cancer, and whether patients had surgery for non-metastastic, stage-matched tumors only,” the researchers wrote in their abstract.
They did analyses for breast, lung, prostate, colorectal, melanoma, and bladder cancers. The median time from diagnosis of myeloma to diagnosis of the second ranged from 35 months (bladder [133 myeloma patients] and lung [286 myeloma patients] cancers) to 50 months (melanoma [140 myeloma patients]). The median time after myeloma diagnosis was 40 months for those patients who developed breast, prostate, or colorectal cancers.
In the comparisons, myeloma survivors were significantly older (P less than .001) than patients initially diagnosed with the same respective cancers. In the case-control analysis, breast (P = .002) and lung cancers (P = .003) were more often diagnosed at an early stage among myeloma survivors.
The hazard ratio (HR) for cancer-specific survival for 189 myeloma patients diagnosed with breast cancer as compared to other breast cancer patients, for example, was 0.99, 95% confidence interval (CI) 0.61-1.61. The HR for the cumulative incidence function of cancer death was 0.82, 95% CI 0.50-1.35.
Myeloma patients were no less likely than were case-control subjects to have surgery for their cancers, with the exception of the 330 myeloma patients who developed prostate cancer (odds ratio, 0.59, 95% CI, 0.44-0.81).
Cancer-specific survival significantly differed (P less than .05) only for lung cancer, and was better among the 286 myeloma patients with lung cancer even when stratified by stage (HR 0.64, 95% CI 0.54-0.75). For cumulative incidence function of cancer death for lung cancer, the hazard ratio was 0.52 (95% CI 0.44-0.61). Better outcomes in lung cancer are not fully explained by earlier detection, suggesting a biological difference, the researchers reported.
Cumulative incidence function of cancer death was significantly lower for myeloma patients with lung and colorectal cancers.
Dr. Castillo disclosed honoraria from Celgene and Janssen; a consulting or advisory role with Biogen, Otsuka, and Pharmacyclics; and institutional research funding from Abbvie, Gilead Sciences, Millennium, and Pharmacyclics. Dr. Olszewski disclosed institutional research funding from Genentech, Incyte, and TG Therapeutics.
Citation: Outcomes of secondary cancers among myeloma survivors. 2017 ASCO annual meeting. Abstract No. 8043.
mdales@frontlinemedcom.com
On Twitter @maryjodales
With improved treatment, patients with multiple myeloma are surviving long enough to develop other cancers, Jorge J. Castillo, MD, and Adam J. Olszewski, MD, reported in a poster to be presented at the annual meeting of the American Society of Clinical Oncology.
The good news is that myeloma patients, when diagnosed with a subsequent solid tumor, are just as likely to respond to treatment and do just as well as patients without myeloma, according to Dr. Castillo of Dana-Farber Cancer Institute, Boston, and Dr. Olszewski of Alpert Medical School of Brown University, Providence, R.I.
They based their conclusion on Surveillance, Epidemiology, and End Results (SEER) data for patients diagnosed with six common cancers from 2004-2013.
“Among them, we identified [nearly 1,300] myeloma survivors, and we matched each to 50 randomly sampled controls with the same cancer by age, sex, race, and year of diagnosis. We then compared [cancer specific survival], cumulative incidence function (CIF) for death from the non-myeloma index cancer, and whether patients had surgery for non-metastastic, stage-matched tumors only,” the researchers wrote in their abstract.
They did analyses for breast, lung, prostate, colorectal, melanoma, and bladder cancers. The median time from diagnosis of myeloma to diagnosis of the second ranged from 35 months (bladder [133 myeloma patients] and lung [286 myeloma patients] cancers) to 50 months (melanoma [140 myeloma patients]). The median time after myeloma diagnosis was 40 months for those patients who developed breast, prostate, or colorectal cancers.
In the comparisons, myeloma survivors were significantly older (P less than .001) than patients initially diagnosed with the same respective cancers. In the case-control analysis, breast (P = .002) and lung cancers (P = .003) were more often diagnosed at an early stage among myeloma survivors.
The hazard ratio (HR) for cancer-specific survival for 189 myeloma patients diagnosed with breast cancer as compared to other breast cancer patients, for example, was 0.99, 95% confidence interval (CI) 0.61-1.61. The HR for the cumulative incidence function of cancer death was 0.82, 95% CI 0.50-1.35.
Myeloma patients were no less likely than were case-control subjects to have surgery for their cancers, with the exception of the 330 myeloma patients who developed prostate cancer (odds ratio, 0.59, 95% CI, 0.44-0.81).
Cancer-specific survival significantly differed (P less than .05) only for lung cancer, and was better among the 286 myeloma patients with lung cancer even when stratified by stage (HR 0.64, 95% CI 0.54-0.75). For cumulative incidence function of cancer death for lung cancer, the hazard ratio was 0.52 (95% CI 0.44-0.61). Better outcomes in lung cancer are not fully explained by earlier detection, suggesting a biological difference, the researchers reported.
Cumulative incidence function of cancer death was significantly lower for myeloma patients with lung and colorectal cancers.
Dr. Castillo disclosed honoraria from Celgene and Janssen; a consulting or advisory role with Biogen, Otsuka, and Pharmacyclics; and institutional research funding from Abbvie, Gilead Sciences, Millennium, and Pharmacyclics. Dr. Olszewski disclosed institutional research funding from Genentech, Incyte, and TG Therapeutics.
Citation: Outcomes of secondary cancers among myeloma survivors. 2017 ASCO annual meeting. Abstract No. 8043.
mdales@frontlinemedcom.com
On Twitter @maryjodales
FROM 2017 ASCO ANNUAL MEETING
Key clinical point:
Major finding: Cancer-specific survival significantly differed (P less than .05) only for lung cancer, and was better among the 286 myeloma patients with lung cancer even when stratified by stage (HR, 0.64, 95% CI 0.54-0.75).
Data source: Surveillance, Epidemiology, and End Results (SEER) data for patients diagnosed with six common cancers from 2004-2013.
Disclosures: Dr. Castillo disclosed honoraria from Celgene and Janssen; a consulting or advisory role with Biogen, Otsuka, and Pharmacyclics; and institutional research funding from Abbvie, Gilead Sciences, Millennium, and Pharmacyclics. Dr. Olszewski disclosed institutional research funding from Genentech, Incyte, and TG Therapeutics.
Citation: Outcomes of secondary cancers among myeloma survivors. 2017 ASCO annual meeting. Abstract No. 8043.
Lenalidomide maintenance boosted depth of response in myeloma patients
Lenalidomide maintenance therapy further improved depth of response in newly diagnosed, transplant-eligible patients with multiple myeloma in the EMN02/HO95 trial, based on the abstract of a poster to be presented at the annual meeting of the American Society of Clinical Oncology.
The study results also show that using multiparameter flow cytometry to monitor minimal residual disease (MRD) was predictive of patient outcome. A high-risk cytogenetic profile – defined as having del17, translocation (4;14), or translocation (14;16) – was the most important prognostic factor in MRD-positive patients, according to Stefania Oliva, MD, of the University of Torino [Italy] and her colleagues.
At 3 years, progression-free survival was 50% in MRD-positive patients and 77% in MRD-negative patients (hazard ratio, 2.87; P less than .001). High-risk cytogenetics was the most important risk factor (HR, 9.87; interaction-P = .001). Further, 48% of patients who had MRD before maintenance therapy and had a second evaluation for minimal residual disease after at least 1 year of lenalidomide therapy became MRD negative.
The trial (NCT01208766) participants were no older than age 65 years and received received bortezomib-cyclophosphamide-dexamethasone (VCD) induction therapy, then bortezomib-melphalan-prednisone (VMP) or high-dose melphalan intensification therapy followed by stem cell transplant, and subsequently bortezomib-lenalidomide-dexamethasone (VRD) consolidation therapy or no consolidation therapy, followed by lenalidomide maintenance therapy.
Of 316 patients who were evaluable before maintenance therapy, 18% had International Staging System stage III disease (beta-2 microglobulin of 5.5 mg/L or greater) and 22% had a high-risk cytogenetic profile.
For intensification therapy, 63% had received high-dose melphalan and 37% got VMP; thereafter 51% had received VRD. Nearly two-thirds of the 76% of patients who were MRD negative got high-dose melphalan, with a median follow-up of 30 months from MRD enrollment.
Patients who had at least a very good partial response underwent minimal residual disease evaluation before starting maintenance therapy and then every 6-12 months during maintenance therapy. Multiparameter flow cytometry was performed on bone marrow according to Euroflow-based methods (eight colors, two tubes) with a sensitivity of 10-5, and quality checks were performed to compare sensitivity and to show correlation between protocols.
Dr. Oliva disclosed receiving honoraria from Celgene and Takeda.
Citation: Minimal residual disease (MRD) monitoring by multiparameter flow cytometry (MFC) in newly diagnosed transplant eligible multiple myeloma (MM) patients: Results from the EMN02/HO95 phase 3 trial. 2017 ASCO annual meeting. Abstract No: 8011
mdales@frontlinemedcom.com
On Twitter @maryjodales
Lenalidomide maintenance therapy further improved depth of response in newly diagnosed, transplant-eligible patients with multiple myeloma in the EMN02/HO95 trial, based on the abstract of a poster to be presented at the annual meeting of the American Society of Clinical Oncology.
The study results also show that using multiparameter flow cytometry to monitor minimal residual disease (MRD) was predictive of patient outcome. A high-risk cytogenetic profile – defined as having del17, translocation (4;14), or translocation (14;16) – was the most important prognostic factor in MRD-positive patients, according to Stefania Oliva, MD, of the University of Torino [Italy] and her colleagues.
At 3 years, progression-free survival was 50% in MRD-positive patients and 77% in MRD-negative patients (hazard ratio, 2.87; P less than .001). High-risk cytogenetics was the most important risk factor (HR, 9.87; interaction-P = .001). Further, 48% of patients who had MRD before maintenance therapy and had a second evaluation for minimal residual disease after at least 1 year of lenalidomide therapy became MRD negative.
The trial (NCT01208766) participants were no older than age 65 years and received received bortezomib-cyclophosphamide-dexamethasone (VCD) induction therapy, then bortezomib-melphalan-prednisone (VMP) or high-dose melphalan intensification therapy followed by stem cell transplant, and subsequently bortezomib-lenalidomide-dexamethasone (VRD) consolidation therapy or no consolidation therapy, followed by lenalidomide maintenance therapy.
Of 316 patients who were evaluable before maintenance therapy, 18% had International Staging System stage III disease (beta-2 microglobulin of 5.5 mg/L or greater) and 22% had a high-risk cytogenetic profile.
For intensification therapy, 63% had received high-dose melphalan and 37% got VMP; thereafter 51% had received VRD. Nearly two-thirds of the 76% of patients who were MRD negative got high-dose melphalan, with a median follow-up of 30 months from MRD enrollment.
Patients who had at least a very good partial response underwent minimal residual disease evaluation before starting maintenance therapy and then every 6-12 months during maintenance therapy. Multiparameter flow cytometry was performed on bone marrow according to Euroflow-based methods (eight colors, two tubes) with a sensitivity of 10-5, and quality checks were performed to compare sensitivity and to show correlation between protocols.
Dr. Oliva disclosed receiving honoraria from Celgene and Takeda.
Citation: Minimal residual disease (MRD) monitoring by multiparameter flow cytometry (MFC) in newly diagnosed transplant eligible multiple myeloma (MM) patients: Results from the EMN02/HO95 phase 3 trial. 2017 ASCO annual meeting. Abstract No: 8011
mdales@frontlinemedcom.com
On Twitter @maryjodales
Lenalidomide maintenance therapy further improved depth of response in newly diagnosed, transplant-eligible patients with multiple myeloma in the EMN02/HO95 trial, based on the abstract of a poster to be presented at the annual meeting of the American Society of Clinical Oncology.
The study results also show that using multiparameter flow cytometry to monitor minimal residual disease (MRD) was predictive of patient outcome. A high-risk cytogenetic profile – defined as having del17, translocation (4;14), or translocation (14;16) – was the most important prognostic factor in MRD-positive patients, according to Stefania Oliva, MD, of the University of Torino [Italy] and her colleagues.
At 3 years, progression-free survival was 50% in MRD-positive patients and 77% in MRD-negative patients (hazard ratio, 2.87; P less than .001). High-risk cytogenetics was the most important risk factor (HR, 9.87; interaction-P = .001). Further, 48% of patients who had MRD before maintenance therapy and had a second evaluation for minimal residual disease after at least 1 year of lenalidomide therapy became MRD negative.
The trial (NCT01208766) participants were no older than age 65 years and received received bortezomib-cyclophosphamide-dexamethasone (VCD) induction therapy, then bortezomib-melphalan-prednisone (VMP) or high-dose melphalan intensification therapy followed by stem cell transplant, and subsequently bortezomib-lenalidomide-dexamethasone (VRD) consolidation therapy or no consolidation therapy, followed by lenalidomide maintenance therapy.
Of 316 patients who were evaluable before maintenance therapy, 18% had International Staging System stage III disease (beta-2 microglobulin of 5.5 mg/L or greater) and 22% had a high-risk cytogenetic profile.
For intensification therapy, 63% had received high-dose melphalan and 37% got VMP; thereafter 51% had received VRD. Nearly two-thirds of the 76% of patients who were MRD negative got high-dose melphalan, with a median follow-up of 30 months from MRD enrollment.
Patients who had at least a very good partial response underwent minimal residual disease evaluation before starting maintenance therapy and then every 6-12 months during maintenance therapy. Multiparameter flow cytometry was performed on bone marrow according to Euroflow-based methods (eight colors, two tubes) with a sensitivity of 10-5, and quality checks were performed to compare sensitivity and to show correlation between protocols.
Dr. Oliva disclosed receiving honoraria from Celgene and Takeda.
Citation: Minimal residual disease (MRD) monitoring by multiparameter flow cytometry (MFC) in newly diagnosed transplant eligible multiple myeloma (MM) patients: Results from the EMN02/HO95 phase 3 trial. 2017 ASCO annual meeting. Abstract No: 8011
mdales@frontlinemedcom.com
On Twitter @maryjodales
FROM 2017 ASCO ANNUAL MEETING
Key clinical point:
Major finding: 48% of patients who had minimal residual disease before maintenance therapy and had a second evaluation for MRD after at least 1 year of lenalidomide therapy became MRD negative.
Data source: A 3-year study of 316 patients who were evaluable before maintenance therapy in the EMN02/HO95 trial.
Disclosures: Dr. Oliva disclosed receiving honoraria from Celgene and Takeda.
Citation: Minimal residual disease (MRD) monitoring by multiparameter flow cytometry (MFC) in newly diagnosed transplant eligible multiple myeloma (MM) patients: Results from the EMN02/HO95 phase 3 trial. 2017 ASCO annual meeting. Abstract No: 8011
FDA grants priority review to NDA for copanlisib
The US Food and Drug Administration (FDA) has granted priority review to the new drug application (NDA) for copanlisib, an intravenous PI3K inhibitor.
The NDA is for copanlisib as a treatment for patients with relapsed or refractory follicular lymphoma (FL) who have received at least 2 prior therapies.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The application for copanlisib is supported by data from the CHRONOS-1 trial. This phase 2 trial enrolled 141 patients with relapsed/refractory, indolent non-Hodgkin lymphoma. Most of these patients had FL (n=104).
In all patients, copanlisib produced an objective response rate of 59.2%, with a complete response rate of 12%. The median duration of response exceeded 98 weeks.
In the FL subset, copanlisib produced an overall response rate of 58.7%, with a complete response rate of 14.4%. The median duration of response exceeded 52 weeks.
In the entire cohort, there were 3 deaths considered related to copanlisib.
The most common treatment-related adverse events were transient hyperglycemia (all grades: 49%/grade 3-4: 40%) and hypertension (all grades: 29%/grade 3: 23%).
“Patients with relapsed or refractory follicular lymphoma have a poor prognosis, and new treatment options which are well tolerated and effective are needed to prolong progression-free survival and improve quality of life for these patients,” said Martin Dreyling, MD, a professor at the University of Munich Hospital (Grosshadern) in Germany and lead investigator of the CHRONOS-1 study.
“Based on the CHRONOS-1 results, where copanlisib showed durable efficacy with a manageable and distinct safety profile, the compound may have the potential to address this unmet medical need.”
Data from CHRONOS-1 were presented at the AACR Annual Meeting 2017.
Data from the FL subset of the trial are scheduled to be presented at the 2017 ASCO Annual Meeting in June.
Copanlisib is being developed by Bayer. The compound has fast track and orphan drug designations from the FDA.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The FDA’s fast track program is designed to facilitate the development and expedite the review of products intended to treat or prevent serious or life-threatening conditions and address unmet medical need.
Through the fast track program, a product may be eligible for priority review. In addition, the company developing the product may be allowed to submit sections of the NDA or biologic license application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings and written communications with the FDA. ![]()
The US Food and Drug Administration (FDA) has granted priority review to the new drug application (NDA) for copanlisib, an intravenous PI3K inhibitor.
The NDA is for copanlisib as a treatment for patients with relapsed or refractory follicular lymphoma (FL) who have received at least 2 prior therapies.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The application for copanlisib is supported by data from the CHRONOS-1 trial. This phase 2 trial enrolled 141 patients with relapsed/refractory, indolent non-Hodgkin lymphoma. Most of these patients had FL (n=104).
In all patients, copanlisib produced an objective response rate of 59.2%, with a complete response rate of 12%. The median duration of response exceeded 98 weeks.
In the FL subset, copanlisib produced an overall response rate of 58.7%, with a complete response rate of 14.4%. The median duration of response exceeded 52 weeks.
In the entire cohort, there were 3 deaths considered related to copanlisib.
The most common treatment-related adverse events were transient hyperglycemia (all grades: 49%/grade 3-4: 40%) and hypertension (all grades: 29%/grade 3: 23%).
“Patients with relapsed or refractory follicular lymphoma have a poor prognosis, and new treatment options which are well tolerated and effective are needed to prolong progression-free survival and improve quality of life for these patients,” said Martin Dreyling, MD, a professor at the University of Munich Hospital (Grosshadern) in Germany and lead investigator of the CHRONOS-1 study.
“Based on the CHRONOS-1 results, where copanlisib showed durable efficacy with a manageable and distinct safety profile, the compound may have the potential to address this unmet medical need.”
Data from CHRONOS-1 were presented at the AACR Annual Meeting 2017.
Data from the FL subset of the trial are scheduled to be presented at the 2017 ASCO Annual Meeting in June.
Copanlisib is being developed by Bayer. The compound has fast track and orphan drug designations from the FDA.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The FDA’s fast track program is designed to facilitate the development and expedite the review of products intended to treat or prevent serious or life-threatening conditions and address unmet medical need.
Through the fast track program, a product may be eligible for priority review. In addition, the company developing the product may be allowed to submit sections of the NDA or biologic license application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings and written communications with the FDA. ![]()
The US Food and Drug Administration (FDA) has granted priority review to the new drug application (NDA) for copanlisib, an intravenous PI3K inhibitor.
The NDA is for copanlisib as a treatment for patients with relapsed or refractory follicular lymphoma (FL) who have received at least 2 prior therapies.
The FDA grants priority review to applications for products that may provide significant improvements in the treatment, diagnosis, or prevention of serious conditions.
The agency’s goal is to take action on a priority review application within 6 months of receiving it, rather than the standard 10 months.
The application for copanlisib is supported by data from the CHRONOS-1 trial. This phase 2 trial enrolled 141 patients with relapsed/refractory, indolent non-Hodgkin lymphoma. Most of these patients had FL (n=104).
In all patients, copanlisib produced an objective response rate of 59.2%, with a complete response rate of 12%. The median duration of response exceeded 98 weeks.
In the FL subset, copanlisib produced an overall response rate of 58.7%, with a complete response rate of 14.4%. The median duration of response exceeded 52 weeks.
In the entire cohort, there were 3 deaths considered related to copanlisib.
The most common treatment-related adverse events were transient hyperglycemia (all grades: 49%/grade 3-4: 40%) and hypertension (all grades: 29%/grade 3: 23%).
“Patients with relapsed or refractory follicular lymphoma have a poor prognosis, and new treatment options which are well tolerated and effective are needed to prolong progression-free survival and improve quality of life for these patients,” said Martin Dreyling, MD, a professor at the University of Munich Hospital (Grosshadern) in Germany and lead investigator of the CHRONOS-1 study.
“Based on the CHRONOS-1 results, where copanlisib showed durable efficacy with a manageable and distinct safety profile, the compound may have the potential to address this unmet medical need.”
Data from CHRONOS-1 were presented at the AACR Annual Meeting 2017.
Data from the FL subset of the trial are scheduled to be presented at the 2017 ASCO Annual Meeting in June.
Copanlisib is being developed by Bayer. The compound has fast track and orphan drug designations from the FDA.
The FDA grants orphan designation to products intended to treat, diagnose, or prevent diseases/disorders that affect fewer than 200,000 people in the US.
The designation provides incentives for sponsors to develop products for rare diseases. This may include tax credits toward the cost of clinical trials, prescription drug user fee waivers, and 7 years of market exclusivity if the product is approved.
The FDA’s fast track program is designed to facilitate the development and expedite the review of products intended to treat or prevent serious or life-threatening conditions and address unmet medical need.
Through the fast track program, a product may be eligible for priority review. In addition, the company developing the product may be allowed to submit sections of the NDA or biologic license application on a rolling basis as data become available.
Fast track designation also provides the company with opportunities for more frequent meetings and written communications with the FDA. ![]()
Compound could treat lymphoma, myeloma
A nucleoside analog has shown potential for treating primary effusion lymphoma (PEL) and multiple myeloma (MM), according to researchers.
The compound, 6-ethylthioinosine (6-ETI), killed both PEL and MM cells in vitro.
6-ETI also reduced tumor burden and prolonged survival in mouse models of MM and PEL.
Ethel Cesarman, MD, PhD, of Weill Cornell Medicine in New York, New York, and her colleagues conducted this research and disclosed their results in the Journal of Clinical Investigation.
The researchers identified 6-ETI via high-throughput screening, and they initially tested the compound in PEL cell lines. 6-ETI induced necrosis, apoptosis, and autophagy in these cells.
In a xenograft model of PEL, mice treated with 6-ETI experienced “striking and immediate regression of the implanted xenograft within 3 days of treatment,” according to the researchers.
In addition, mice that received 6-ETI had significantly longer progression-free and overall survival than control mice (P<0.0001 for both), and the researchers said there were no obvious toxicities from the treatment.
Looking into the mechanism of 6-ETI, the researchers found that adenosine kinase (ADK) is required to phosphorylate and activate the compound, and PEL cells have high levels of ADK.
Because PEL cells closely resemble plasma cells, the researchers theorized that MM might produce high levels of ADK as well. Experiments in MM cells proved this theory correct.
So the researchers tested 6-ETI in MM cell lines and samples from MM patients. The compound induced apoptosis and autophagy, and it activated a DNA damage response in MM cells.
In a mouse model of MM, 6-ETI treatment significantly reduced tumor burden but did not result in weight loss. And treated mice had significantly longer overall survival than control mice (P<0.005).
Dr Cesarman and her colleagues are now trying to better understand how 6-ETI works and determine what other cancers expressing high levels of ADK might respond to the drug.
“This compound could provide a much-needed approach to treat people with some forms of plasma malignancies as well as other cancers that express ADK,” Dr Cesarman said. ![]()
A nucleoside analog has shown potential for treating primary effusion lymphoma (PEL) and multiple myeloma (MM), according to researchers.
The compound, 6-ethylthioinosine (6-ETI), killed both PEL and MM cells in vitro.
6-ETI also reduced tumor burden and prolonged survival in mouse models of MM and PEL.
Ethel Cesarman, MD, PhD, of Weill Cornell Medicine in New York, New York, and her colleagues conducted this research and disclosed their results in the Journal of Clinical Investigation.
The researchers identified 6-ETI via high-throughput screening, and they initially tested the compound in PEL cell lines. 6-ETI induced necrosis, apoptosis, and autophagy in these cells.
In a xenograft model of PEL, mice treated with 6-ETI experienced “striking and immediate regression of the implanted xenograft within 3 days of treatment,” according to the researchers.
In addition, mice that received 6-ETI had significantly longer progression-free and overall survival than control mice (P<0.0001 for both), and the researchers said there were no obvious toxicities from the treatment.
Looking into the mechanism of 6-ETI, the researchers found that adenosine kinase (ADK) is required to phosphorylate and activate the compound, and PEL cells have high levels of ADK.
Because PEL cells closely resemble plasma cells, the researchers theorized that MM might produce high levels of ADK as well. Experiments in MM cells proved this theory correct.
So the researchers tested 6-ETI in MM cell lines and samples from MM patients. The compound induced apoptosis and autophagy, and it activated a DNA damage response in MM cells.
In a mouse model of MM, 6-ETI treatment significantly reduced tumor burden but did not result in weight loss. And treated mice had significantly longer overall survival than control mice (P<0.005).
Dr Cesarman and her colleagues are now trying to better understand how 6-ETI works and determine what other cancers expressing high levels of ADK might respond to the drug.
“This compound could provide a much-needed approach to treat people with some forms of plasma malignancies as well as other cancers that express ADK,” Dr Cesarman said. ![]()
A nucleoside analog has shown potential for treating primary effusion lymphoma (PEL) and multiple myeloma (MM), according to researchers.
The compound, 6-ethylthioinosine (6-ETI), killed both PEL and MM cells in vitro.
6-ETI also reduced tumor burden and prolonged survival in mouse models of MM and PEL.
Ethel Cesarman, MD, PhD, of Weill Cornell Medicine in New York, New York, and her colleagues conducted this research and disclosed their results in the Journal of Clinical Investigation.
The researchers identified 6-ETI via high-throughput screening, and they initially tested the compound in PEL cell lines. 6-ETI induced necrosis, apoptosis, and autophagy in these cells.
In a xenograft model of PEL, mice treated with 6-ETI experienced “striking and immediate regression of the implanted xenograft within 3 days of treatment,” according to the researchers.
In addition, mice that received 6-ETI had significantly longer progression-free and overall survival than control mice (P<0.0001 for both), and the researchers said there were no obvious toxicities from the treatment.
Looking into the mechanism of 6-ETI, the researchers found that adenosine kinase (ADK) is required to phosphorylate and activate the compound, and PEL cells have high levels of ADK.
Because PEL cells closely resemble plasma cells, the researchers theorized that MM might produce high levels of ADK as well. Experiments in MM cells proved this theory correct.
So the researchers tested 6-ETI in MM cell lines and samples from MM patients. The compound induced apoptosis and autophagy, and it activated a DNA damage response in MM cells.
In a mouse model of MM, 6-ETI treatment significantly reduced tumor burden but did not result in weight loss. And treated mice had significantly longer overall survival than control mice (P<0.005).
Dr Cesarman and her colleagues are now trying to better understand how 6-ETI works and determine what other cancers expressing high levels of ADK might respond to the drug.
“This compound could provide a much-needed approach to treat people with some forms of plasma malignancies as well as other cancers that express ADK,” Dr Cesarman said. ![]()
Intensive chemo upfront means DHL patients can skip HSCT
A new study suggests that patients with double-hit lymphoma (DHL) in first remission only benefit from an autologous hematopoietic stem cell transplant (auto-HSCT) if they received standard frontline chemotherapy.
Researchers looked at long-term outcomes for DHL patients who achieved remission and, overall, found that auto-HSCT did not significantly prolong remission or survival.
However, patients who received standard chemotherapy as frontline treatment did appear to benefit from auto-HSCT, as these patients had worse outcomes than patients who received intensive frontline chemotherapy.
This finding led the researchers to recommend that DHL patients receive intensive chemotherapy upfront and forgo subsequent auto-HSCT.
Daniel J. Landsburg, MD, of the University of Pennsylvania in Philadelphia, and his colleagues made these recommendations in the Journal of Clinical Oncology.
“A major dilemma for oncologists who treat [DHL] was whether or not to recommend the potentially harmful therapy of auto-[H]SCT to patients with this disease as a strategy to help keep them in remission,” Dr Landsburg said.
To gain some insight into the issue, Dr Landsburg and his colleagues looked at data on 159 patients from 19 academic medical centers across the US.
Patients were diagnosed with DHL between 2006 and 2015, and all achieved remission following frontline chemotherapy.
Thirty-five patients received standard frontline therapy—R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone).
The remaining patients received intensive frontline chemotherapy:
- 81 received DA-EPOCH-R (etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab)
- 32 received R-hyperCVAD (rituximab, cyclophosphamide, vincristine, doxorubicin, dexamethasone, methotrexate, and cytarabine)
- 11 received R-CODOX-M/IVAC (rituximab, cyclophosphamide, doxorubicin, vincristine, methotrexate/ifosfamide, etoposide, high-dose cytarabine).
Sixty-two patients underwent auto-HSCT, and 97 patients did not. There were no significant differences between these 2 patient groups at baseline.
“Our result is not explained by differences in patients’ overall health or disease features,” Dr Landsburg said. “The transplant and non-transplant arms of this study were very well-matched.”
Relapse and survival
For the entire patient cohort, the 3-year relapse-free survival (RFS) rate was 80%, and the 3-year overall survival (OS) rate was 87%.
There was no significant difference in RFS or OS between patients who underwent auto-HSCT and those who did not.
The RFS rate was 89% in patients who underwent auto-HSCT and 75% in patients who did not (P=0.12). The OS rate was 91% and 85%, respectively (P=0.74).
“Once these patients achieve remission, the data show they are likely to stay in remission,” Dr Landsburg said.
“In the absence of a large, randomized, controlled trial, which would be very challenging to carry out in this case, this is the best evidence we have, and it shows there’s no clear benefit to these patients undergoing auto-[H]SCT.”
Impact of frontline therapy
Patients who received R-CHOP upfront had worse RFS and OS than those who received intensive chemotherapy, although the OS difference was not significant.
RFS rates were 56% in patients who received R-CHOP, 88% in those who received DA-EPOCH-R, 87% in those who received R-hyperCVAD, and 91% in those who received R-CODOX-M/IVAC (P=0.003).
OS rates were 77% in patients who received R-CHOP, 87% in those who received DA-EPOCH-R, 90% in those who received R-hyperCVAD, and 100% in those who received R-CODOX-M/IVAC, respectively (P=0.36).
When the 3 intensive regimens were combined, the RFS rate was 88% (vs 56% for R-CHOP, P=0.002), and the OS rate was 90% (vs 77% for R-CHOP, P=0.13).
Frontline therapy and HSCT
Patients who received R-CHOP upfront benefited from auto-HSCT, but patients who received intensive chemotherapy did not.
The RFS was 51% for patients who received R-CHOP and did not undergo auto-HSCT, and it was 75% for patients who received R-CHOP followed by auto-HSCT.
The OS was 75% for patients who received R-CHOP and did not undergo auto-HSCT, and it was 83% for patients who received R-CHOP followed by auto-HSCT.
The RFS was 86% for patients who received intensive chemotherapy and did not undergo auto-HSCT, and it was 91% for patients who received intensive chemotherapy followed by auto-HSCT.
The OS was 89% for patients who received intensive chemotherapy and did not undergo auto-HSCT, and it was 92% for patients who received intensive chemotherapy followed by auto-HSCT.
An intergroup comparison showed a significant difference in RFS (P=0.003), which was driven by a significantly lower rate of RFS for patients who received R-CHOP without auto-HSCT, compared with patients who received intensive chemotherapy without auto-HSCT (P=0.003) or intensive chemotherapy with auto-HSCT (P=0.001).
“[I]f patients do go into remission with R-CHOP, it appears to be less durable, so, in these cases, going forward with auto-[H]SCT may still make sense,” Dr Landsburg said.
On the other hand, there was no significant difference between the groups with regard to OS (P=0.50).
Dr Landsburg said the next step for this research will be to study features of patients who don’t go into remission in order to understand why their disease is resistant to therapy and if that can be overcome with different treatment strategies. He also said it’s important to try to find more effective therapies for DHL patients who relapse. ![]()
A new study suggests that patients with double-hit lymphoma (DHL) in first remission only benefit from an autologous hematopoietic stem cell transplant (auto-HSCT) if they received standard frontline chemotherapy.
Researchers looked at long-term outcomes for DHL patients who achieved remission and, overall, found that auto-HSCT did not significantly prolong remission or survival.
However, patients who received standard chemotherapy as frontline treatment did appear to benefit from auto-HSCT, as these patients had worse outcomes than patients who received intensive frontline chemotherapy.
This finding led the researchers to recommend that DHL patients receive intensive chemotherapy upfront and forgo subsequent auto-HSCT.
Daniel J. Landsburg, MD, of the University of Pennsylvania in Philadelphia, and his colleagues made these recommendations in the Journal of Clinical Oncology.
“A major dilemma for oncologists who treat [DHL] was whether or not to recommend the potentially harmful therapy of auto-[H]SCT to patients with this disease as a strategy to help keep them in remission,” Dr Landsburg said.
To gain some insight into the issue, Dr Landsburg and his colleagues looked at data on 159 patients from 19 academic medical centers across the US.
Patients were diagnosed with DHL between 2006 and 2015, and all achieved remission following frontline chemotherapy.
Thirty-five patients received standard frontline therapy—R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone).
The remaining patients received intensive frontline chemotherapy:
- 81 received DA-EPOCH-R (etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab)
- 32 received R-hyperCVAD (rituximab, cyclophosphamide, vincristine, doxorubicin, dexamethasone, methotrexate, and cytarabine)
- 11 received R-CODOX-M/IVAC (rituximab, cyclophosphamide, doxorubicin, vincristine, methotrexate/ifosfamide, etoposide, high-dose cytarabine).
Sixty-two patients underwent auto-HSCT, and 97 patients did not. There were no significant differences between these 2 patient groups at baseline.
“Our result is not explained by differences in patients’ overall health or disease features,” Dr Landsburg said. “The transplant and non-transplant arms of this study were very well-matched.”
Relapse and survival
For the entire patient cohort, the 3-year relapse-free survival (RFS) rate was 80%, and the 3-year overall survival (OS) rate was 87%.
There was no significant difference in RFS or OS between patients who underwent auto-HSCT and those who did not.
The RFS rate was 89% in patients who underwent auto-HSCT and 75% in patients who did not (P=0.12). The OS rate was 91% and 85%, respectively (P=0.74).
“Once these patients achieve remission, the data show they are likely to stay in remission,” Dr Landsburg said.
“In the absence of a large, randomized, controlled trial, which would be very challenging to carry out in this case, this is the best evidence we have, and it shows there’s no clear benefit to these patients undergoing auto-[H]SCT.”
Impact of frontline therapy
Patients who received R-CHOP upfront had worse RFS and OS than those who received intensive chemotherapy, although the OS difference was not significant.
RFS rates were 56% in patients who received R-CHOP, 88% in those who received DA-EPOCH-R, 87% in those who received R-hyperCVAD, and 91% in those who received R-CODOX-M/IVAC (P=0.003).
OS rates were 77% in patients who received R-CHOP, 87% in those who received DA-EPOCH-R, 90% in those who received R-hyperCVAD, and 100% in those who received R-CODOX-M/IVAC, respectively (P=0.36).
When the 3 intensive regimens were combined, the RFS rate was 88% (vs 56% for R-CHOP, P=0.002), and the OS rate was 90% (vs 77% for R-CHOP, P=0.13).
Frontline therapy and HSCT
Patients who received R-CHOP upfront benefited from auto-HSCT, but patients who received intensive chemotherapy did not.
The RFS was 51% for patients who received R-CHOP and did not undergo auto-HSCT, and it was 75% for patients who received R-CHOP followed by auto-HSCT.
The OS was 75% for patients who received R-CHOP and did not undergo auto-HSCT, and it was 83% for patients who received R-CHOP followed by auto-HSCT.
The RFS was 86% for patients who received intensive chemotherapy and did not undergo auto-HSCT, and it was 91% for patients who received intensive chemotherapy followed by auto-HSCT.
The OS was 89% for patients who received intensive chemotherapy and did not undergo auto-HSCT, and it was 92% for patients who received intensive chemotherapy followed by auto-HSCT.
An intergroup comparison showed a significant difference in RFS (P=0.003), which was driven by a significantly lower rate of RFS for patients who received R-CHOP without auto-HSCT, compared with patients who received intensive chemotherapy without auto-HSCT (P=0.003) or intensive chemotherapy with auto-HSCT (P=0.001).
“[I]f patients do go into remission with R-CHOP, it appears to be less durable, so, in these cases, going forward with auto-[H]SCT may still make sense,” Dr Landsburg said.
On the other hand, there was no significant difference between the groups with regard to OS (P=0.50).
Dr Landsburg said the next step for this research will be to study features of patients who don’t go into remission in order to understand why their disease is resistant to therapy and if that can be overcome with different treatment strategies. He also said it’s important to try to find more effective therapies for DHL patients who relapse. ![]()
A new study suggests that patients with double-hit lymphoma (DHL) in first remission only benefit from an autologous hematopoietic stem cell transplant (auto-HSCT) if they received standard frontline chemotherapy.
Researchers looked at long-term outcomes for DHL patients who achieved remission and, overall, found that auto-HSCT did not significantly prolong remission or survival.
However, patients who received standard chemotherapy as frontline treatment did appear to benefit from auto-HSCT, as these patients had worse outcomes than patients who received intensive frontline chemotherapy.
This finding led the researchers to recommend that DHL patients receive intensive chemotherapy upfront and forgo subsequent auto-HSCT.
Daniel J. Landsburg, MD, of the University of Pennsylvania in Philadelphia, and his colleagues made these recommendations in the Journal of Clinical Oncology.
“A major dilemma for oncologists who treat [DHL] was whether or not to recommend the potentially harmful therapy of auto-[H]SCT to patients with this disease as a strategy to help keep them in remission,” Dr Landsburg said.
To gain some insight into the issue, Dr Landsburg and his colleagues looked at data on 159 patients from 19 academic medical centers across the US.
Patients were diagnosed with DHL between 2006 and 2015, and all achieved remission following frontline chemotherapy.
Thirty-five patients received standard frontline therapy—R-CHOP (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone).
The remaining patients received intensive frontline chemotherapy:
- 81 received DA-EPOCH-R (etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab)
- 32 received R-hyperCVAD (rituximab, cyclophosphamide, vincristine, doxorubicin, dexamethasone, methotrexate, and cytarabine)
- 11 received R-CODOX-M/IVAC (rituximab, cyclophosphamide, doxorubicin, vincristine, methotrexate/ifosfamide, etoposide, high-dose cytarabine).
Sixty-two patients underwent auto-HSCT, and 97 patients did not. There were no significant differences between these 2 patient groups at baseline.
“Our result is not explained by differences in patients’ overall health or disease features,” Dr Landsburg said. “The transplant and non-transplant arms of this study were very well-matched.”
Relapse and survival
For the entire patient cohort, the 3-year relapse-free survival (RFS) rate was 80%, and the 3-year overall survival (OS) rate was 87%.
There was no significant difference in RFS or OS between patients who underwent auto-HSCT and those who did not.
The RFS rate was 89% in patients who underwent auto-HSCT and 75% in patients who did not (P=0.12). The OS rate was 91% and 85%, respectively (P=0.74).
“Once these patients achieve remission, the data show they are likely to stay in remission,” Dr Landsburg said.
“In the absence of a large, randomized, controlled trial, which would be very challenging to carry out in this case, this is the best evidence we have, and it shows there’s no clear benefit to these patients undergoing auto-[H]SCT.”
Impact of frontline therapy
Patients who received R-CHOP upfront had worse RFS and OS than those who received intensive chemotherapy, although the OS difference was not significant.
RFS rates were 56% in patients who received R-CHOP, 88% in those who received DA-EPOCH-R, 87% in those who received R-hyperCVAD, and 91% in those who received R-CODOX-M/IVAC (P=0.003).
OS rates were 77% in patients who received R-CHOP, 87% in those who received DA-EPOCH-R, 90% in those who received R-hyperCVAD, and 100% in those who received R-CODOX-M/IVAC, respectively (P=0.36).
When the 3 intensive regimens were combined, the RFS rate was 88% (vs 56% for R-CHOP, P=0.002), and the OS rate was 90% (vs 77% for R-CHOP, P=0.13).
Frontline therapy and HSCT
Patients who received R-CHOP upfront benefited from auto-HSCT, but patients who received intensive chemotherapy did not.
The RFS was 51% for patients who received R-CHOP and did not undergo auto-HSCT, and it was 75% for patients who received R-CHOP followed by auto-HSCT.
The OS was 75% for patients who received R-CHOP and did not undergo auto-HSCT, and it was 83% for patients who received R-CHOP followed by auto-HSCT.
The RFS was 86% for patients who received intensive chemotherapy and did not undergo auto-HSCT, and it was 91% for patients who received intensive chemotherapy followed by auto-HSCT.
The OS was 89% for patients who received intensive chemotherapy and did not undergo auto-HSCT, and it was 92% for patients who received intensive chemotherapy followed by auto-HSCT.
An intergroup comparison showed a significant difference in RFS (P=0.003), which was driven by a significantly lower rate of RFS for patients who received R-CHOP without auto-HSCT, compared with patients who received intensive chemotherapy without auto-HSCT (P=0.003) or intensive chemotherapy with auto-HSCT (P=0.001).
“[I]f patients do go into remission with R-CHOP, it appears to be less durable, so, in these cases, going forward with auto-[H]SCT may still make sense,” Dr Landsburg said.
On the other hand, there was no significant difference between the groups with regard to OS (P=0.50).
Dr Landsburg said the next step for this research will be to study features of patients who don’t go into remission in order to understand why their disease is resistant to therapy and if that can be overcome with different treatment strategies. He also said it’s important to try to find more effective therapies for DHL patients who relapse. ![]()