User login
VIDEO: Oral drugs tackle hepatitis C genotypes 2, 3
CHICAGO – The oral drug duo of sofosbuvir and ribavirin produced good cure rates in a study of 419 patients with hepatitis C genotype 2 or 3.
Dr. Bruce R. Bacon, who was not involved in the study, highlighted the regimen’s efficacy even in patients with cirrhosis or who had failed previous therapy. Hear his perspective on treating genotypes 2 and 3 in this interview at the annual Digestive Disease Week.
Dr. Bacon is the James F. King Endowed Chair in Gastroenterology and a professor of medicine at Saint Louis University.
He reported financial associations with Gilead Sciences, which funded the study, and with AbbVie and Janssen Pharmaceuticals.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – The oral drug duo of sofosbuvir and ribavirin produced good cure rates in a study of 419 patients with hepatitis C genotype 2 or 3.
Dr. Bruce R. Bacon, who was not involved in the study, highlighted the regimen’s efficacy even in patients with cirrhosis or who had failed previous therapy. Hear his perspective on treating genotypes 2 and 3 in this interview at the annual Digestive Disease Week.
Dr. Bacon is the James F. King Endowed Chair in Gastroenterology and a professor of medicine at Saint Louis University.
He reported financial associations with Gilead Sciences, which funded the study, and with AbbVie and Janssen Pharmaceuticals.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
CHICAGO – The oral drug duo of sofosbuvir and ribavirin produced good cure rates in a study of 419 patients with hepatitis C genotype 2 or 3.
Dr. Bruce R. Bacon, who was not involved in the study, highlighted the regimen’s efficacy even in patients with cirrhosis or who had failed previous therapy. Hear his perspective on treating genotypes 2 and 3 in this interview at the annual Digestive Disease Week.
Dr. Bacon is the James F. King Endowed Chair in Gastroenterology and a professor of medicine at Saint Louis University.
He reported financial associations with Gilead Sciences, which funded the study, and with AbbVie and Janssen Pharmaceuticals.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT DDW 2014
Interferon-free regimen improves response in HCV
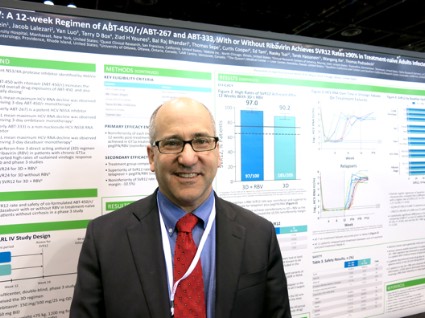
CHICAGO – Treating patients with hepatitis C virus genotype 1a or 1b with 12 weeks of an experimental interferon-free, triple-drug oral regimen produced good sustained virologic response rates with or without the use of ribavirin, but the addition of ribavirin appeared helpful to those with genotype 1a infection, according to results of two separate phase III studies.
Both studies recruited previously untreated patients with no cirrhosis to undergo a regimen of ABT-450 with ritonavir and ombitasvir (ABT-450/r-ombitasvir) plus dasabuvir and either ribavirin or a matching placebo. The PEARL-III study randomized 419 patients with hepatitis C virus genotype 1b, and the PEARL-IV study randomized 305 patients with genotype 1a infection. Patients received a daily dose of 150 mg ABT-450, 100 mg ritonavir, and 25 mg ombitasvir, twice-daily doses of 250 mg dasabuvir, and either placebo or ribavirin dosed according to body weight.
In patients with genotype 1a, 97% who got the regimen plus ribavirin and 90.2% of those who got the regimen plus placebo achieved a sustained virologic response (SVR) 12 weeks after treatment ended (defined as a hepatitis C virus RNA level of less than 25 IU/ml). The difference between those two groups was statistically significant, and the treatment without ribavirin was inferior to the regimen with ribavirin in genotype 1a patients. These response rates were superior, however, to rates in the medical literature for treatment-naive patients who got conventional treatment with telaprevir plus peginterferon-ribavirin, Dr. Peter Ferenci and his associates reported.
Virologic failure was significantly more likely in patients with genotype 1a who got the regimen plus placebo in the current study, compared with those who got ribavirin – 7.8% vs. 2%, respectively, reported Dr. Ferenci of the Medical University of Vienna. Only 2 of the 18 patients with genotype 1a who developed virologic failure received ribavirin.
The findings were published online by the New England Journal of Medicine (N. Engl. J. Med. 2014 May 4 [doi:10.1056/NEJMoa1402338]).
Previous data on treatment with telaprevir plus peginterferon-ribavirin showed sustained virologic response rates of 60%-65%, coinvestigator Dr. David Bernstein said in an interview. With the experimental regimen, "the take-home message is that there are extraordinarily high cure rates for patients with hepatitis C genotype 1," said Dr. Bernstein, chief of hepatology at the Center for Liver Disease, North Shore University Hospital, Manhasset, N.Y. He reported results of the PEARL IV study in a poster presentation at the annual Digestive Disease Week.
Genotype 1b is the most prevalent form of hepatitis C, especially in Europe and East Asia, but genotype 1a is more prevalent in North America.
For patients with genotype 1b infection in the study, 99.5% who received the regimen with ribavirin and 99% who got placebo achieved SVR at 12 weeks.
ABT-450/4, ombitasvir, and dasabuvir are not yet approved for treatment, but approval is expected later this year, Dr. Bernstein said.
Fewer than 1% of patients discontinued treatment due to adverse events. "The second take-home message is that it’s a 12-week course of therapy and the overall side effect profile is minimal," he said. "We will be using these therapies once they become available."
ABT-450 inhibits the hepatitis C virus nonstructural 3/4A protease and is given with ritonavir to increase ABT-450 plasma levels and half-life. Ombitasvir inhibits the hepatitis C virus NS5A replication complex. Dasabuvir is a nonnucleoside NS5B polymerase inhibitor.
AbbVie, which is developing the new drugs, funded the study. Dr. Ferenci reported financial associations with 10 companies and his coinvestigators reported financial associations with dozens of companies.
On Twitter @sherryboschert
With evidence from these trials and other recent studies suggesting that interferon-free regimens will revolutionize treatment of hepatitis C infection, "the future is here," Dr. T. Jake Liang and Dr. Marc G. Ghany wrote in an editorial accompanying the study (N. Engl. J. Med. 2014 May 4 [doi:10.1056/NEJMe1403619]).
"The side effects associated with interferon-based therapy have prevented many patients from undergoing treatment and are a major reason for treatment failure. Perhaps the more important achievement of these interferon-free regimens is the lower rate and severity of side effects associated with treatment," they wrote.
Much remains to be learned, they added. The new interferon-free regimens have been tested mainly in white, middle-aged men without cirrhosis, and not as much in more difficult to treat patients.
Dr. T. Jake Liang and Dr. Marc G. Ghany, both of the National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Md., reported having no financial disclosures.
With evidence from these trials and other recent studies suggesting that interferon-free regimens will revolutionize treatment of hepatitis C infection, "the future is here," Dr. T. Jake Liang and Dr. Marc G. Ghany wrote in an editorial accompanying the study (N. Engl. J. Med. 2014 May 4 [doi:10.1056/NEJMe1403619]).
"The side effects associated with interferon-based therapy have prevented many patients from undergoing treatment and are a major reason for treatment failure. Perhaps the more important achievement of these interferon-free regimens is the lower rate and severity of side effects associated with treatment," they wrote.
Much remains to be learned, they added. The new interferon-free regimens have been tested mainly in white, middle-aged men without cirrhosis, and not as much in more difficult to treat patients.
Dr. T. Jake Liang and Dr. Marc G. Ghany, both of the National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Md., reported having no financial disclosures.
With evidence from these trials and other recent studies suggesting that interferon-free regimens will revolutionize treatment of hepatitis C infection, "the future is here," Dr. T. Jake Liang and Dr. Marc G. Ghany wrote in an editorial accompanying the study (N. Engl. J. Med. 2014 May 4 [doi:10.1056/NEJMe1403619]).
"The side effects associated with interferon-based therapy have prevented many patients from undergoing treatment and are a major reason for treatment failure. Perhaps the more important achievement of these interferon-free regimens is the lower rate and severity of side effects associated with treatment," they wrote.
Much remains to be learned, they added. The new interferon-free regimens have been tested mainly in white, middle-aged men without cirrhosis, and not as much in more difficult to treat patients.
Dr. T. Jake Liang and Dr. Marc G. Ghany, both of the National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Md., reported having no financial disclosures.
CHICAGO – Treating patients with hepatitis C virus genotype 1a or 1b with 12 weeks of an experimental interferon-free, triple-drug oral regimen produced good sustained virologic response rates with or without the use of ribavirin, but the addition of ribavirin appeared helpful to those with genotype 1a infection, according to results of two separate phase III studies.
Both studies recruited previously untreated patients with no cirrhosis to undergo a regimen of ABT-450 with ritonavir and ombitasvir (ABT-450/r-ombitasvir) plus dasabuvir and either ribavirin or a matching placebo. The PEARL-III study randomized 419 patients with hepatitis C virus genotype 1b, and the PEARL-IV study randomized 305 patients with genotype 1a infection. Patients received a daily dose of 150 mg ABT-450, 100 mg ritonavir, and 25 mg ombitasvir, twice-daily doses of 250 mg dasabuvir, and either placebo or ribavirin dosed according to body weight.
In patients with genotype 1a, 97% who got the regimen plus ribavirin and 90.2% of those who got the regimen plus placebo achieved a sustained virologic response (SVR) 12 weeks after treatment ended (defined as a hepatitis C virus RNA level of less than 25 IU/ml). The difference between those two groups was statistically significant, and the treatment without ribavirin was inferior to the regimen with ribavirin in genotype 1a patients. These response rates were superior, however, to rates in the medical literature for treatment-naive patients who got conventional treatment with telaprevir plus peginterferon-ribavirin, Dr. Peter Ferenci and his associates reported.
Virologic failure was significantly more likely in patients with genotype 1a who got the regimen plus placebo in the current study, compared with those who got ribavirin – 7.8% vs. 2%, respectively, reported Dr. Ferenci of the Medical University of Vienna. Only 2 of the 18 patients with genotype 1a who developed virologic failure received ribavirin.
The findings were published online by the New England Journal of Medicine (N. Engl. J. Med. 2014 May 4 [doi:10.1056/NEJMoa1402338]).
Previous data on treatment with telaprevir plus peginterferon-ribavirin showed sustained virologic response rates of 60%-65%, coinvestigator Dr. David Bernstein said in an interview. With the experimental regimen, "the take-home message is that there are extraordinarily high cure rates for patients with hepatitis C genotype 1," said Dr. Bernstein, chief of hepatology at the Center for Liver Disease, North Shore University Hospital, Manhasset, N.Y. He reported results of the PEARL IV study in a poster presentation at the annual Digestive Disease Week.
Genotype 1b is the most prevalent form of hepatitis C, especially in Europe and East Asia, but genotype 1a is more prevalent in North America.
For patients with genotype 1b infection in the study, 99.5% who received the regimen with ribavirin and 99% who got placebo achieved SVR at 12 weeks.
ABT-450/4, ombitasvir, and dasabuvir are not yet approved for treatment, but approval is expected later this year, Dr. Bernstein said.
Fewer than 1% of patients discontinued treatment due to adverse events. "The second take-home message is that it’s a 12-week course of therapy and the overall side effect profile is minimal," he said. "We will be using these therapies once they become available."
ABT-450 inhibits the hepatitis C virus nonstructural 3/4A protease and is given with ritonavir to increase ABT-450 plasma levels and half-life. Ombitasvir inhibits the hepatitis C virus NS5A replication complex. Dasabuvir is a nonnucleoside NS5B polymerase inhibitor.
AbbVie, which is developing the new drugs, funded the study. Dr. Ferenci reported financial associations with 10 companies and his coinvestigators reported financial associations with dozens of companies.
On Twitter @sherryboschert
CHICAGO – Treating patients with hepatitis C virus genotype 1a or 1b with 12 weeks of an experimental interferon-free, triple-drug oral regimen produced good sustained virologic response rates with or without the use of ribavirin, but the addition of ribavirin appeared helpful to those with genotype 1a infection, according to results of two separate phase III studies.
Both studies recruited previously untreated patients with no cirrhosis to undergo a regimen of ABT-450 with ritonavir and ombitasvir (ABT-450/r-ombitasvir) plus dasabuvir and either ribavirin or a matching placebo. The PEARL-III study randomized 419 patients with hepatitis C virus genotype 1b, and the PEARL-IV study randomized 305 patients with genotype 1a infection. Patients received a daily dose of 150 mg ABT-450, 100 mg ritonavir, and 25 mg ombitasvir, twice-daily doses of 250 mg dasabuvir, and either placebo or ribavirin dosed according to body weight.
In patients with genotype 1a, 97% who got the regimen plus ribavirin and 90.2% of those who got the regimen plus placebo achieved a sustained virologic response (SVR) 12 weeks after treatment ended (defined as a hepatitis C virus RNA level of less than 25 IU/ml). The difference between those two groups was statistically significant, and the treatment without ribavirin was inferior to the regimen with ribavirin in genotype 1a patients. These response rates were superior, however, to rates in the medical literature for treatment-naive patients who got conventional treatment with telaprevir plus peginterferon-ribavirin, Dr. Peter Ferenci and his associates reported.
Virologic failure was significantly more likely in patients with genotype 1a who got the regimen plus placebo in the current study, compared with those who got ribavirin – 7.8% vs. 2%, respectively, reported Dr. Ferenci of the Medical University of Vienna. Only 2 of the 18 patients with genotype 1a who developed virologic failure received ribavirin.
The findings were published online by the New England Journal of Medicine (N. Engl. J. Med. 2014 May 4 [doi:10.1056/NEJMoa1402338]).
Previous data on treatment with telaprevir plus peginterferon-ribavirin showed sustained virologic response rates of 60%-65%, coinvestigator Dr. David Bernstein said in an interview. With the experimental regimen, "the take-home message is that there are extraordinarily high cure rates for patients with hepatitis C genotype 1," said Dr. Bernstein, chief of hepatology at the Center for Liver Disease, North Shore University Hospital, Manhasset, N.Y. He reported results of the PEARL IV study in a poster presentation at the annual Digestive Disease Week.
Genotype 1b is the most prevalent form of hepatitis C, especially in Europe and East Asia, but genotype 1a is more prevalent in North America.
For patients with genotype 1b infection in the study, 99.5% who received the regimen with ribavirin and 99% who got placebo achieved SVR at 12 weeks.
ABT-450/4, ombitasvir, and dasabuvir are not yet approved for treatment, but approval is expected later this year, Dr. Bernstein said.
Fewer than 1% of patients discontinued treatment due to adverse events. "The second take-home message is that it’s a 12-week course of therapy and the overall side effect profile is minimal," he said. "We will be using these therapies once they become available."
ABT-450 inhibits the hepatitis C virus nonstructural 3/4A protease and is given with ritonavir to increase ABT-450 plasma levels and half-life. Ombitasvir inhibits the hepatitis C virus NS5A replication complex. Dasabuvir is a nonnucleoside NS5B polymerase inhibitor.
AbbVie, which is developing the new drugs, funded the study. Dr. Ferenci reported financial associations with 10 companies and his coinvestigators reported financial associations with dozens of companies.
On Twitter @sherryboschert
AT DDW 2014
Major finding: For patients with genotype 1a infection, SVR occurred in 97% with ribavirin and 90.2% without ribavirin. For patients with genotype 1b infection, rates were 99.5% and 99%, respectively.
Data source: Two randomized, phase III trials of previously untreated patients with hepatitis C and no cirrhosis who were treated with 12 weeks of ABT-450/r, ombitasvir, and dasabuvir with or without ribavirin.
Disclosures: AbbVie, which is developing the new drugs, funded the study. Dr. Ferenci reported financial associations with 10 companies and his coinvestigators reported financial associations with dozens of companies.
Sofosbuvir-ribavirin treats hepatitis C virus genotypes 2, 3
CHICAGO – Oral sofosbuvir and ribavirin given for 12 weeks produced a sustained virologic response in 93% of 250 patients with hepatitis C virus genotype 2 and in 85% of patients with genotype 3 infection who were treated for 24 weeks.
The 77-center European study defined a sustained virologic response as a hepatitis C virus RNA level of less than 25 IU/mL, Dr. Stefan Zeuzem and his associates reported. More than half of the patients (58%) had undergone prior therapy with interferon-based regimens, with no response in 30%. Cirrhosis was present in 21% of patients.
The study began as a phase III trial randomizing patients to receive 12 weeks of once-daily oral sofosbuvir 400 mg and twice-daily oral ribavirin (administered according to body weight), or matching placebo. The investigators turned the trial into a descriptive study after a previous study reported that patients with genotype 3 infection might benefit from more than 12 weeks of sofosbuvir therapy (N. Engl. J. Med. 2013;368:1867-77).
Dr. Zeuzem and his associates unblinded the treatment arms, dropped placebo treatment, and extended treatment for patients with genotype 3 infection to 24 weeks. The findings were published online in the New England Journal of Medicine (N. Engl. J. Med. 2014 May 4 [doi: 1056/NEJMoa1316145]).
Among patients with genotype 3 infection, a sustained virologic response developed in 68% of those with cirrhosis and 91% without cirrhosis, reported Dr. Zeuzem, who is professor of medicine and chief of the department of medicine I at Johann Wolfgang Goethe University Medical Center, Frankfurt, Germany.
Headache, fatigue, and pruritus were the most common adverse events. Approximately 1% of patients in both the genotype 3 and 4 groups discontinued treatment due to side effects.
Treatment history and the presence of cirrhosis affected response rates in patients with genotype 3 infection. In previously untreated patients, a sustained virologic response was seen in 92% of those with cirrhosis and in 95% without cirrhosis. Response rates were lower in previously treated patients with cirrhosis (62%) and without cirrhosis (87%). The reasons for these differences are unknown.
Four factors were independently associated with a sustained virologic response in patients with genotype 3 infection, a multivariate regression analysis found. A sustained response was four times as likely in those with a baseline hepatitis C virus RNA level of less than 6 log10 IU and three times as likely in women, patients without cirrhosis, or those under age 50, compared with patients without those characteristics. These predictive factors must be validated in further studies, Dr. Zeuzem said.
Three patients with genotype 3 infection had a virologic relapse. The response rate and relapse rate for patients with genotype 3 who were treated for 24 weeks were "substantially" better than rates reported in previous studies using the same regimen for 12 or 16 weeks, the investigators said. Extending the duration of treatment did not significantly increase adverse events or discontinuation of therapy.
The findings suggest that sofosbuvir-ribavirin can be an alternative therapy for patients with hepatitis C virus genotype 2 or 3 who have contraindications to peginterferon-based treatment, Dr. Zeuzem said.
Gilead Sciences, which markets sofosbuvir, funded the study. Dr. Zeuzem reported financial associations with Gilead and multiple other pharmaceutical companies, as did many of his coinvestigators.
On Twitter @sherryboschert
CHICAGO – Oral sofosbuvir and ribavirin given for 12 weeks produced a sustained virologic response in 93% of 250 patients with hepatitis C virus genotype 2 and in 85% of patients with genotype 3 infection who were treated for 24 weeks.
The 77-center European study defined a sustained virologic response as a hepatitis C virus RNA level of less than 25 IU/mL, Dr. Stefan Zeuzem and his associates reported. More than half of the patients (58%) had undergone prior therapy with interferon-based regimens, with no response in 30%. Cirrhosis was present in 21% of patients.
The study began as a phase III trial randomizing patients to receive 12 weeks of once-daily oral sofosbuvir 400 mg and twice-daily oral ribavirin (administered according to body weight), or matching placebo. The investigators turned the trial into a descriptive study after a previous study reported that patients with genotype 3 infection might benefit from more than 12 weeks of sofosbuvir therapy (N. Engl. J. Med. 2013;368:1867-77).
Dr. Zeuzem and his associates unblinded the treatment arms, dropped placebo treatment, and extended treatment for patients with genotype 3 infection to 24 weeks. The findings were published online in the New England Journal of Medicine (N. Engl. J. Med. 2014 May 4 [doi: 1056/NEJMoa1316145]).
Among patients with genotype 3 infection, a sustained virologic response developed in 68% of those with cirrhosis and 91% without cirrhosis, reported Dr. Zeuzem, who is professor of medicine and chief of the department of medicine I at Johann Wolfgang Goethe University Medical Center, Frankfurt, Germany.
Headache, fatigue, and pruritus were the most common adverse events. Approximately 1% of patients in both the genotype 3 and 4 groups discontinued treatment due to side effects.
Treatment history and the presence of cirrhosis affected response rates in patients with genotype 3 infection. In previously untreated patients, a sustained virologic response was seen in 92% of those with cirrhosis and in 95% without cirrhosis. Response rates were lower in previously treated patients with cirrhosis (62%) and without cirrhosis (87%). The reasons for these differences are unknown.
Four factors were independently associated with a sustained virologic response in patients with genotype 3 infection, a multivariate regression analysis found. A sustained response was four times as likely in those with a baseline hepatitis C virus RNA level of less than 6 log10 IU and three times as likely in women, patients without cirrhosis, or those under age 50, compared with patients without those characteristics. These predictive factors must be validated in further studies, Dr. Zeuzem said.
Three patients with genotype 3 infection had a virologic relapse. The response rate and relapse rate for patients with genotype 3 who were treated for 24 weeks were "substantially" better than rates reported in previous studies using the same regimen for 12 or 16 weeks, the investigators said. Extending the duration of treatment did not significantly increase adverse events or discontinuation of therapy.
The findings suggest that sofosbuvir-ribavirin can be an alternative therapy for patients with hepatitis C virus genotype 2 or 3 who have contraindications to peginterferon-based treatment, Dr. Zeuzem said.
Gilead Sciences, which markets sofosbuvir, funded the study. Dr. Zeuzem reported financial associations with Gilead and multiple other pharmaceutical companies, as did many of his coinvestigators.
On Twitter @sherryboschert
CHICAGO – Oral sofosbuvir and ribavirin given for 12 weeks produced a sustained virologic response in 93% of 250 patients with hepatitis C virus genotype 2 and in 85% of patients with genotype 3 infection who were treated for 24 weeks.
The 77-center European study defined a sustained virologic response as a hepatitis C virus RNA level of less than 25 IU/mL, Dr. Stefan Zeuzem and his associates reported. More than half of the patients (58%) had undergone prior therapy with interferon-based regimens, with no response in 30%. Cirrhosis was present in 21% of patients.
The study began as a phase III trial randomizing patients to receive 12 weeks of once-daily oral sofosbuvir 400 mg and twice-daily oral ribavirin (administered according to body weight), or matching placebo. The investigators turned the trial into a descriptive study after a previous study reported that patients with genotype 3 infection might benefit from more than 12 weeks of sofosbuvir therapy (N. Engl. J. Med. 2013;368:1867-77).
Dr. Zeuzem and his associates unblinded the treatment arms, dropped placebo treatment, and extended treatment for patients with genotype 3 infection to 24 weeks. The findings were published online in the New England Journal of Medicine (N. Engl. J. Med. 2014 May 4 [doi: 1056/NEJMoa1316145]).
Among patients with genotype 3 infection, a sustained virologic response developed in 68% of those with cirrhosis and 91% without cirrhosis, reported Dr. Zeuzem, who is professor of medicine and chief of the department of medicine I at Johann Wolfgang Goethe University Medical Center, Frankfurt, Germany.
Headache, fatigue, and pruritus were the most common adverse events. Approximately 1% of patients in both the genotype 3 and 4 groups discontinued treatment due to side effects.
Treatment history and the presence of cirrhosis affected response rates in patients with genotype 3 infection. In previously untreated patients, a sustained virologic response was seen in 92% of those with cirrhosis and in 95% without cirrhosis. Response rates were lower in previously treated patients with cirrhosis (62%) and without cirrhosis (87%). The reasons for these differences are unknown.
Four factors were independently associated with a sustained virologic response in patients with genotype 3 infection, a multivariate regression analysis found. A sustained response was four times as likely in those with a baseline hepatitis C virus RNA level of less than 6 log10 IU and three times as likely in women, patients without cirrhosis, or those under age 50, compared with patients without those characteristics. These predictive factors must be validated in further studies, Dr. Zeuzem said.
Three patients with genotype 3 infection had a virologic relapse. The response rate and relapse rate for patients with genotype 3 who were treated for 24 weeks were "substantially" better than rates reported in previous studies using the same regimen for 12 or 16 weeks, the investigators said. Extending the duration of treatment did not significantly increase adverse events or discontinuation of therapy.
The findings suggest that sofosbuvir-ribavirin can be an alternative therapy for patients with hepatitis C virus genotype 2 or 3 who have contraindications to peginterferon-based treatment, Dr. Zeuzem said.
Gilead Sciences, which markets sofosbuvir, funded the study. Dr. Zeuzem reported financial associations with Gilead and multiple other pharmaceutical companies, as did many of his coinvestigators.
On Twitter @sherryboschert
AT DDW 2014
Major finding: Sustained virologic response occurred in 93% after 12 weeks of treatment for genotype 2 and in 85% after 24 weeks of therapy for genotype 3.
Data source: A descriptive cohort study of 419 patients with hepatitis C genotypes 2 or 3 treated with sofosbuvir and ribavirin.
Disclosures: Gilead Sciences, which markets sofosbuvir, funded the study. Dr. Zeuzem reported financial associations with Gilead and multiple other pharmaceutical companies, as did many of his coinvestigators.
NAFLD mortality higher in normal weight patients
Lean patients with nonalcoholic fatty liver disease had a higher overall mortality than did overweight or obese patients with NAFLD, according to a review of 1,090 biopsy-confirmed patients in the United States, Australia, Thailand, and Europe.
Reviewing blood and other samples taken at the time of liver biopsy, the investigators found that the 125 (11.5%) patients with a body mass index (BMI) below 25 kg/m2 (average 23 kg/m2) had less insulin resistance and less advanced fibrosis than did the 965 (88.5%) who presented with higher BMIs (average 33 kg/m2).
But among the 483 patients biopsied before 2005, 9 of the 32 (28%) lean patients died, compared with 62 of the 451 (14%) who were overweight or obese.
The cumulative survival was significantly shorter in lean patients, as well; only lean NAFLD (hazard ratio, 11.8; 95% confidence interval, 2.8-50.1; P = .001) and age (HR, 1.05; 95% CI, 1.008-1.1; P =.02) remained significant when the findings were adjusted for sex, degree of fibrosis, and other confounding factors.
Cardiovascular disease, malignancy, and liver problems were the main causes of death in both lean and overweight patients; there were no significant differences between the groups, and the reason is unclear. Perhaps lean patients have greater central fat distribution; the team plans to investigate the matter further.
"I thought patients with lean fatty liver would have lower mortality; it was exactly the opposite. The risk factors for fatty liver go beyond a person’s body weight, and signs of liver disease secondary to NAFLD in lean patients should be taken very seriously. We must not assume that patients of relatively healthy weight can’t have fatty liver disease," said senior investigator Dr. Paul Angulo, chief of hepatology at the University of Kentucky Medical Center in Lexington.
"We need to be more aggressive in patients with lean fatty liver in terms of biopsy to see how much liver injury they have, and really make a concerted effort to increase their physical activity and change their diet. They don’t need to lose weight, but they need less fat and carbohydrates and more exercise to increase insulin sensitivity, especially in the muscle mass and liver," he said at a teleconference in advance of the annual Digestive Disease Week.
Lean NAFLD patients were more likely to be men; nonwhite, especially Asian and Hispanic; and to have fewer chronic conditions, such as diabetes, hypertension, and dyslipidemia. They also had lower levels of alanine aminotransferase, fewer fatty liver deposits, and less advanced fibrosis, but more severe lobular inflammation. There was no significant difference between the two groups in age (average, 46 years), hepatocyte ballooning, or incidence of nonalcoholic steatohepatitis.
The senior investigator has no disclosures. One of the other dozen investigators is on the board of Abbott, and another advises Bristol-Myers Squibb, Gilead, Merck, Novartis, and Roche.
I have patients like this in my practice, and I am certainly going to pay attention to them now. In the past, I had assumed that the absence of risk factors was a good thing, but now I’ve learned it may not be. Pending study that demonstrates a benefit for specific interventions, I am going to follow Dr. Angulo’s advice and encourage these patients to exercise more robustly. We need to find out the optimal approach for them.
Dr. Lawrence Friedman is chair of the department of medicine at Newton-Wellesley (Mass.) Hospital. He moderated Dr. Angulo’s presentation but was not involved in the project.
I have patients like this in my practice, and I am certainly going to pay attention to them now. In the past, I had assumed that the absence of risk factors was a good thing, but now I’ve learned it may not be. Pending study that demonstrates a benefit for specific interventions, I am going to follow Dr. Angulo’s advice and encourage these patients to exercise more robustly. We need to find out the optimal approach for them.
Dr. Lawrence Friedman is chair of the department of medicine at Newton-Wellesley (Mass.) Hospital. He moderated Dr. Angulo’s presentation but was not involved in the project.
I have patients like this in my practice, and I am certainly going to pay attention to them now. In the past, I had assumed that the absence of risk factors was a good thing, but now I’ve learned it may not be. Pending study that demonstrates a benefit for specific interventions, I am going to follow Dr. Angulo’s advice and encourage these patients to exercise more robustly. We need to find out the optimal approach for them.
Dr. Lawrence Friedman is chair of the department of medicine at Newton-Wellesley (Mass.) Hospital. He moderated Dr. Angulo’s presentation but was not involved in the project.
Lean patients with nonalcoholic fatty liver disease had a higher overall mortality than did overweight or obese patients with NAFLD, according to a review of 1,090 biopsy-confirmed patients in the United States, Australia, Thailand, and Europe.
Reviewing blood and other samples taken at the time of liver biopsy, the investigators found that the 125 (11.5%) patients with a body mass index (BMI) below 25 kg/m2 (average 23 kg/m2) had less insulin resistance and less advanced fibrosis than did the 965 (88.5%) who presented with higher BMIs (average 33 kg/m2).
But among the 483 patients biopsied before 2005, 9 of the 32 (28%) lean patients died, compared with 62 of the 451 (14%) who were overweight or obese.
The cumulative survival was significantly shorter in lean patients, as well; only lean NAFLD (hazard ratio, 11.8; 95% confidence interval, 2.8-50.1; P = .001) and age (HR, 1.05; 95% CI, 1.008-1.1; P =.02) remained significant when the findings were adjusted for sex, degree of fibrosis, and other confounding factors.
Cardiovascular disease, malignancy, and liver problems were the main causes of death in both lean and overweight patients; there were no significant differences between the groups, and the reason is unclear. Perhaps lean patients have greater central fat distribution; the team plans to investigate the matter further.
"I thought patients with lean fatty liver would have lower mortality; it was exactly the opposite. The risk factors for fatty liver go beyond a person’s body weight, and signs of liver disease secondary to NAFLD in lean patients should be taken very seriously. We must not assume that patients of relatively healthy weight can’t have fatty liver disease," said senior investigator Dr. Paul Angulo, chief of hepatology at the University of Kentucky Medical Center in Lexington.
"We need to be more aggressive in patients with lean fatty liver in terms of biopsy to see how much liver injury they have, and really make a concerted effort to increase their physical activity and change their diet. They don’t need to lose weight, but they need less fat and carbohydrates and more exercise to increase insulin sensitivity, especially in the muscle mass and liver," he said at a teleconference in advance of the annual Digestive Disease Week.
Lean NAFLD patients were more likely to be men; nonwhite, especially Asian and Hispanic; and to have fewer chronic conditions, such as diabetes, hypertension, and dyslipidemia. They also had lower levels of alanine aminotransferase, fewer fatty liver deposits, and less advanced fibrosis, but more severe lobular inflammation. There was no significant difference between the two groups in age (average, 46 years), hepatocyte ballooning, or incidence of nonalcoholic steatohepatitis.
The senior investigator has no disclosures. One of the other dozen investigators is on the board of Abbott, and another advises Bristol-Myers Squibb, Gilead, Merck, Novartis, and Roche.
Lean patients with nonalcoholic fatty liver disease had a higher overall mortality than did overweight or obese patients with NAFLD, according to a review of 1,090 biopsy-confirmed patients in the United States, Australia, Thailand, and Europe.
Reviewing blood and other samples taken at the time of liver biopsy, the investigators found that the 125 (11.5%) patients with a body mass index (BMI) below 25 kg/m2 (average 23 kg/m2) had less insulin resistance and less advanced fibrosis than did the 965 (88.5%) who presented with higher BMIs (average 33 kg/m2).
But among the 483 patients biopsied before 2005, 9 of the 32 (28%) lean patients died, compared with 62 of the 451 (14%) who were overweight or obese.
The cumulative survival was significantly shorter in lean patients, as well; only lean NAFLD (hazard ratio, 11.8; 95% confidence interval, 2.8-50.1; P = .001) and age (HR, 1.05; 95% CI, 1.008-1.1; P =.02) remained significant when the findings were adjusted for sex, degree of fibrosis, and other confounding factors.
Cardiovascular disease, malignancy, and liver problems were the main causes of death in both lean and overweight patients; there were no significant differences between the groups, and the reason is unclear. Perhaps lean patients have greater central fat distribution; the team plans to investigate the matter further.
"I thought patients with lean fatty liver would have lower mortality; it was exactly the opposite. The risk factors for fatty liver go beyond a person’s body weight, and signs of liver disease secondary to NAFLD in lean patients should be taken very seriously. We must not assume that patients of relatively healthy weight can’t have fatty liver disease," said senior investigator Dr. Paul Angulo, chief of hepatology at the University of Kentucky Medical Center in Lexington.
"We need to be more aggressive in patients with lean fatty liver in terms of biopsy to see how much liver injury they have, and really make a concerted effort to increase their physical activity and change their diet. They don’t need to lose weight, but they need less fat and carbohydrates and more exercise to increase insulin sensitivity, especially in the muscle mass and liver," he said at a teleconference in advance of the annual Digestive Disease Week.
Lean NAFLD patients were more likely to be men; nonwhite, especially Asian and Hispanic; and to have fewer chronic conditions, such as diabetes, hypertension, and dyslipidemia. They also had lower levels of alanine aminotransferase, fewer fatty liver deposits, and less advanced fibrosis, but more severe lobular inflammation. There was no significant difference between the two groups in age (average, 46 years), hepatocyte ballooning, or incidence of nonalcoholic steatohepatitis.
The senior investigator has no disclosures. One of the other dozen investigators is on the board of Abbott, and another advises Bristol-Myers Squibb, Gilead, Merck, Novartis, and Roche.
FROM DDW 2014
Major finding: Among 483 patients with liver biopsies before 2005, 9 of the 32 (28%) lean patients died, compared with 62 of the 451 (14%) who were overweight or obese.
Data source: Retrospective, international review of 1,090 NAFLD cases.
Disclosures: The senior investigator has no disclosures. One of the other dozen investigators is on the board of Abbott, and another advises Bristol-Myers Squibb, Gilead, Merck, Novartis, and Roche.
Liver transplant exceptions deserve fresh look
Room air hypoxemia was associated with greater post–liver transplant mortality in hepatopulmonary syndrome patients, according to Dr. David S. Goldberg and his colleagues.
On the other hand, HPS transplant candidates had overall decreased pretransplantation mortality compared with non-HPS patients, suggesting that "current exception policy might overprioritize waitlisted HPS patients," they wrote.
The report appears in the May 1 issue of Gastroenterology (doi:10.1053/j.gastro.2014.01.005).
Dr. Goldberg of the University of Pennsylvania, Philadelphia, looked at all 973 liver transplant candidates from the United Network for Organ Sharing database who had at least one exception application for HPS approved between Feb. 27, 2002, and Dec. 14, 2012.
For comparison, the authors assessed 59,619 non-HPS adult waitlist candidates who registered for their first liver transplantation on or after Feb. 27, 2002.
Overall, there was a 1-year posttransplant survival rate of 91%, a 3-year survival rate of 81%, and a 5-year rate of 76% among the HPS cohort, the authors wrote. Those rates were comparable with the 1-year, 3-year, and 5-year posttransplant rates for non-HPS patients of 89%, 81%, and 74%.
However, looking at pretransplant survival, the authors found that a significantly greater proportion of non-HPS transplant candidates died on the waitlist, compared with HPS patients (20% vs. 9%; P less than .001). That translated to a hazard ratio of 0.82 among HPS patients for dying on the waitlist, compared with non-HPS patients (95% confidence interval, 0.70-0.96).
Next, the authors assessed the relationship between pretransplant room air oxygenation among HPS patients on posttransplant survival rates. They found that patients with pretransplant PaO2 (partial pressure of oxygen in arterial blood) levels of less than 50 mm Hg had a significantly higher posttransplant mortality, compared with patients with PaO2 levels between 50 and 59 mm Hg (HR = 1.56; 95% CI, 1.02-2.38).
Similarly, in a cubic spline model, transplant recipients with a PaO2 of less than 44.0 mm Hg had significantly increased posttransplantation mortality compared with recipients with a PaO2 of 44.1-54.0 mm Hg (HR = 1.58; 95% CI, 1.15-2.18).
"These data must be taken in context, as the 5-year posttransplantation patient survival in HPS patients with the lowest values of PaO2 is still at or above a threshold many would consider acceptable for a transplant recipient," the authors cautioned. "Therefore, the transplant community must decide what degree of hypoxemia makes a patient too high risk," they added.
The authors conceded several limitations. "First, we were unable to employ the strict criteria defining HPS used in prospective multicenter studies," they wrote.
"However, we are confident that most, if not all, of the patients had HPS based on the data documenting hypoxemia and shunting in nearly 90% of patients."
Nevertheless, "excellent posttransplantation outcomes in those with less severe hypoxemia suggest that it might be possible to optimize posttransplantation outcomes for patients with HPS without disadvantaging the broader transplant population," they wrote.
This could be accomplished by a review of current exception algorithms, and "by decreasing the initial number of exception points for HPS patients, while offering additional priority to those whose PaO2 values decline toward higher-risk values," they wrote.
Given the fact that this would increase the overall waitlist time, "an increase rather than decrease in data collected regarding these patients is needed to guide policy," they concluded.
The authors disclosed no conflicts of interest. Dr. Goldberg reported receiving funding from the National Institutes of Health, and the study was partially supported by the Health Resources and Services Administration.
This study is an important reminder of the need for ongoing evidence-based revision of the MELD-based liver allocation system. It has been clear since the adoption of MELD for the purpose of organ allocation that there are patients for whom the laboratory components of the MELD score do not accurately portray their disease-associated risk of death. Thus, there is widespread petitioning for MELD exception points for a number of conditions, including hepatopulmonary syndrome (HPS). While criteria for some such conditions are clearly defined, most prominently including hepatocellular carcinoma, for most other conditions practices vary by region and criteria are not always strictly adhered to.
The initial number of points granted for most conditions, including HPS, is arbitrary rather than based on objective analysis. This study suggests that while posttransplant outcomes are similar between patients with HPS exception points and those without points, pretransplant survival is superior in the HPS group, suggesting that perhaps a lower MELD score should be assigned to these patients.
While exceptions are currently a fundamental part of MELD-based liver allocation, greater standardization of MELD exception point criteria is urgently needed. Additional analysis of wait list survival and survival benefit models will be required to ensure that individual groups of patients are not being unjustly overprioritized.
Dr. Elizabeth C. Verna is assistant professor of medicine, Center for Liver Disease and Transplantation, Division of Digestive and Liver Diseases, Columbia University College of Physicians and Surgeons, New York. She disclosed no conflicts of interest.
This study is an important reminder of the need for ongoing evidence-based revision of the MELD-based liver allocation system. It has been clear since the adoption of MELD for the purpose of organ allocation that there are patients for whom the laboratory components of the MELD score do not accurately portray their disease-associated risk of death. Thus, there is widespread petitioning for MELD exception points for a number of conditions, including hepatopulmonary syndrome (HPS). While criteria for some such conditions are clearly defined, most prominently including hepatocellular carcinoma, for most other conditions practices vary by region and criteria are not always strictly adhered to.
The initial number of points granted for most conditions, including HPS, is arbitrary rather than based on objective analysis. This study suggests that while posttransplant outcomes are similar between patients with HPS exception points and those without points, pretransplant survival is superior in the HPS group, suggesting that perhaps a lower MELD score should be assigned to these patients.
While exceptions are currently a fundamental part of MELD-based liver allocation, greater standardization of MELD exception point criteria is urgently needed. Additional analysis of wait list survival and survival benefit models will be required to ensure that individual groups of patients are not being unjustly overprioritized.
Dr. Elizabeth C. Verna is assistant professor of medicine, Center for Liver Disease and Transplantation, Division of Digestive and Liver Diseases, Columbia University College of Physicians and Surgeons, New York. She disclosed no conflicts of interest.
This study is an important reminder of the need for ongoing evidence-based revision of the MELD-based liver allocation system. It has been clear since the adoption of MELD for the purpose of organ allocation that there are patients for whom the laboratory components of the MELD score do not accurately portray their disease-associated risk of death. Thus, there is widespread petitioning for MELD exception points for a number of conditions, including hepatopulmonary syndrome (HPS). While criteria for some such conditions are clearly defined, most prominently including hepatocellular carcinoma, for most other conditions practices vary by region and criteria are not always strictly adhered to.
The initial number of points granted for most conditions, including HPS, is arbitrary rather than based on objective analysis. This study suggests that while posttransplant outcomes are similar between patients with HPS exception points and those without points, pretransplant survival is superior in the HPS group, suggesting that perhaps a lower MELD score should be assigned to these patients.
While exceptions are currently a fundamental part of MELD-based liver allocation, greater standardization of MELD exception point criteria is urgently needed. Additional analysis of wait list survival and survival benefit models will be required to ensure that individual groups of patients are not being unjustly overprioritized.
Dr. Elizabeth C. Verna is assistant professor of medicine, Center for Liver Disease and Transplantation, Division of Digestive and Liver Diseases, Columbia University College of Physicians and Surgeons, New York. She disclosed no conflicts of interest.
Room air hypoxemia was associated with greater post–liver transplant mortality in hepatopulmonary syndrome patients, according to Dr. David S. Goldberg and his colleagues.
On the other hand, HPS transplant candidates had overall decreased pretransplantation mortality compared with non-HPS patients, suggesting that "current exception policy might overprioritize waitlisted HPS patients," they wrote.
The report appears in the May 1 issue of Gastroenterology (doi:10.1053/j.gastro.2014.01.005).
Dr. Goldberg of the University of Pennsylvania, Philadelphia, looked at all 973 liver transplant candidates from the United Network for Organ Sharing database who had at least one exception application for HPS approved between Feb. 27, 2002, and Dec. 14, 2012.
For comparison, the authors assessed 59,619 non-HPS adult waitlist candidates who registered for their first liver transplantation on or after Feb. 27, 2002.
Overall, there was a 1-year posttransplant survival rate of 91%, a 3-year survival rate of 81%, and a 5-year rate of 76% among the HPS cohort, the authors wrote. Those rates were comparable with the 1-year, 3-year, and 5-year posttransplant rates for non-HPS patients of 89%, 81%, and 74%.
However, looking at pretransplant survival, the authors found that a significantly greater proportion of non-HPS transplant candidates died on the waitlist, compared with HPS patients (20% vs. 9%; P less than .001). That translated to a hazard ratio of 0.82 among HPS patients for dying on the waitlist, compared with non-HPS patients (95% confidence interval, 0.70-0.96).
Next, the authors assessed the relationship between pretransplant room air oxygenation among HPS patients on posttransplant survival rates. They found that patients with pretransplant PaO2 (partial pressure of oxygen in arterial blood) levels of less than 50 mm Hg had a significantly higher posttransplant mortality, compared with patients with PaO2 levels between 50 and 59 mm Hg (HR = 1.56; 95% CI, 1.02-2.38).
Similarly, in a cubic spline model, transplant recipients with a PaO2 of less than 44.0 mm Hg had significantly increased posttransplantation mortality compared with recipients with a PaO2 of 44.1-54.0 mm Hg (HR = 1.58; 95% CI, 1.15-2.18).
"These data must be taken in context, as the 5-year posttransplantation patient survival in HPS patients with the lowest values of PaO2 is still at or above a threshold many would consider acceptable for a transplant recipient," the authors cautioned. "Therefore, the transplant community must decide what degree of hypoxemia makes a patient too high risk," they added.
The authors conceded several limitations. "First, we were unable to employ the strict criteria defining HPS used in prospective multicenter studies," they wrote.
"However, we are confident that most, if not all, of the patients had HPS based on the data documenting hypoxemia and shunting in nearly 90% of patients."
Nevertheless, "excellent posttransplantation outcomes in those with less severe hypoxemia suggest that it might be possible to optimize posttransplantation outcomes for patients with HPS without disadvantaging the broader transplant population," they wrote.
This could be accomplished by a review of current exception algorithms, and "by decreasing the initial number of exception points for HPS patients, while offering additional priority to those whose PaO2 values decline toward higher-risk values," they wrote.
Given the fact that this would increase the overall waitlist time, "an increase rather than decrease in data collected regarding these patients is needed to guide policy," they concluded.
The authors disclosed no conflicts of interest. Dr. Goldberg reported receiving funding from the National Institutes of Health, and the study was partially supported by the Health Resources and Services Administration.
Room air hypoxemia was associated with greater post–liver transplant mortality in hepatopulmonary syndrome patients, according to Dr. David S. Goldberg and his colleagues.
On the other hand, HPS transplant candidates had overall decreased pretransplantation mortality compared with non-HPS patients, suggesting that "current exception policy might overprioritize waitlisted HPS patients," they wrote.
The report appears in the May 1 issue of Gastroenterology (doi:10.1053/j.gastro.2014.01.005).
Dr. Goldberg of the University of Pennsylvania, Philadelphia, looked at all 973 liver transplant candidates from the United Network for Organ Sharing database who had at least one exception application for HPS approved between Feb. 27, 2002, and Dec. 14, 2012.
For comparison, the authors assessed 59,619 non-HPS adult waitlist candidates who registered for their first liver transplantation on or after Feb. 27, 2002.
Overall, there was a 1-year posttransplant survival rate of 91%, a 3-year survival rate of 81%, and a 5-year rate of 76% among the HPS cohort, the authors wrote. Those rates were comparable with the 1-year, 3-year, and 5-year posttransplant rates for non-HPS patients of 89%, 81%, and 74%.
However, looking at pretransplant survival, the authors found that a significantly greater proportion of non-HPS transplant candidates died on the waitlist, compared with HPS patients (20% vs. 9%; P less than .001). That translated to a hazard ratio of 0.82 among HPS patients for dying on the waitlist, compared with non-HPS patients (95% confidence interval, 0.70-0.96).
Next, the authors assessed the relationship between pretransplant room air oxygenation among HPS patients on posttransplant survival rates. They found that patients with pretransplant PaO2 (partial pressure of oxygen in arterial blood) levels of less than 50 mm Hg had a significantly higher posttransplant mortality, compared with patients with PaO2 levels between 50 and 59 mm Hg (HR = 1.56; 95% CI, 1.02-2.38).
Similarly, in a cubic spline model, transplant recipients with a PaO2 of less than 44.0 mm Hg had significantly increased posttransplantation mortality compared with recipients with a PaO2 of 44.1-54.0 mm Hg (HR = 1.58; 95% CI, 1.15-2.18).
"These data must be taken in context, as the 5-year posttransplantation patient survival in HPS patients with the lowest values of PaO2 is still at or above a threshold many would consider acceptable for a transplant recipient," the authors cautioned. "Therefore, the transplant community must decide what degree of hypoxemia makes a patient too high risk," they added.
The authors conceded several limitations. "First, we were unable to employ the strict criteria defining HPS used in prospective multicenter studies," they wrote.
"However, we are confident that most, if not all, of the patients had HPS based on the data documenting hypoxemia and shunting in nearly 90% of patients."
Nevertheless, "excellent posttransplantation outcomes in those with less severe hypoxemia suggest that it might be possible to optimize posttransplantation outcomes for patients with HPS without disadvantaging the broader transplant population," they wrote.
This could be accomplished by a review of current exception algorithms, and "by decreasing the initial number of exception points for HPS patients, while offering additional priority to those whose PaO2 values decline toward higher-risk values," they wrote.
Given the fact that this would increase the overall waitlist time, "an increase rather than decrease in data collected regarding these patients is needed to guide policy," they concluded.
The authors disclosed no conflicts of interest. Dr. Goldberg reported receiving funding from the National Institutes of Health, and the study was partially supported by the Health Resources and Services Administration.
FROM GASTROENTEROLOGY
Major finding: Liver transplant recipients with hepatopulmonary syndrome have success similar to that of other transplant candidates, unless severe hypoxemia is present.
Data source: A retrospective cohort study of data submitted to the United Network for Organ Sharing.
Disclosures: The authors disclosed no conflicts of interest. Dr. Goldberg reported receiving funding from the National Institutes of Health, and the study was partially supported by the Health Resources and Services Administration.
Simeprevir and sofosbuvir combo offers high SVR rates
LONDON – Cure rates of 93%-100% were achieved in patients with chronic hepatitis C virus genotype 1 treated with both simeprevir and sofosbuvir despite the presence of advanced cirrhosis in a phase IIa trial.
Sustained virologic responses with dual therapy for 12 weeks (SVR12) were 93% with and 93% without the additional use of ribavirin. SVR rates following 24 weeks of treatment were 93% for dual therapy with ribavirin and 100% with no ribavirin. The findings cast further doubt on the benefit of using ribavirin with newer antiviral agents.
"SVR12 rates were high, regardless of baseline characteristics," Dr. Eric Lawitz said when presenting these data from the COSMOS study at the International Liver Congress 2014.
Dr. Lawitz, of the Texas Liver Institute at the University of Texas Health Science Center in San Antonio, added that the baseline characteristics included prior treatment history, METAVIR score, and the presence of known resistance factors – the Q80K polymorphism or IL28B genotype.
Simeprevir (Olysio) and sofosbuvir (Solvadi) are once-daily formulations recently approved for the treatment of chronic hepatitis C virus (HCV) genotype 1 in several countries, including Canada and the United States.
The aim of the COSMOS study was to test the efficacy and safety of these two novel antiviral treatments in tandem in patients with chronic HCV genotype 1 infection who had not previously received any anti-HCV therapy or who had been treated with pegylated interferon plus ribavirin but had not adequately responded.
There were two cohorts of patients, Dr. Lawitz explained. The first cohort comprised 80 patients without cirrhosis (METAVIR F0-F2) who had not responded to prior treatment, while the second cohort included 87 patients with advanced cirrhosis (METAVIR F3-F4) who were either null responders or treatment naive. He presented data only on cohort 2.
Patients were randomized to receive 12 or 24 weeks of simeprevir plus sofosbuvir with or without additional ribavirin. The median age of the study population overall was 58 years, 67% was male, and 91% were white. Just less than half of randomized patients had cirrhosis, and 54% were null responders. Dr. Lawitz pointed out that genotype 1a was the most prevalent subtype of HCV, with around 40% having the Q80K polymorphism and 79% having the IL28B (non-CC) genotype.
Three patients randomized to the 24-week study arm discontinued treatment early. Reasons for discontinuation included one death unrelated to treatment, withdrawal of consent by one patient, and one adverse event unrelated to treatment (dual therapy–only arm).
"When we look at all 87 patients, the SVR12 was 82/87, with two [2.3%] patients failing for nonvirologic reasons, and three [3.4%] patients relapsing," Dr. Lawitz reported at the meeting, which was sponsored by the European Association for the Study of the Liver.
SVR12 rates were 98% and 95% in patients with METAVIR F3 and F4, respectively, and 96% and 94% in null responders and never-treated patients. SVR12 in patients with the IL28B genotype were 94%, 98%, and 95%, respectively, for the CC, CT, and TT forms.
Looking more closely at the three patients with viral relapse, no common factors were found that could help to predict the response to treatment.
On the whole, simeprevir plus sofosbuvir with or without ribavirin was generally well tolerated, Dr. Lawitz said. The most frequent adverse events were fatigue, headache, and nausea, occurring in 37.9%, 19.5%, and 17.2% of the overall study population, respectively.
"Numerically, to look at adverse events, they were more frequent in the ribavirin-containing arms compared to the ribavirin-free arms," he observed. And although bilirubin elevations were frequent, they were mostly mild and occurred primarily in the ribavirin-containing arms.
The study was sponsored by Janssen. Dr. Lawitz has received research support and speaker fees, and has acted as an advisory board member for Janssen, Gilead Sciences, and several other companies.
LONDON – Cure rates of 93%-100% were achieved in patients with chronic hepatitis C virus genotype 1 treated with both simeprevir and sofosbuvir despite the presence of advanced cirrhosis in a phase IIa trial.
Sustained virologic responses with dual therapy for 12 weeks (SVR12) were 93% with and 93% without the additional use of ribavirin. SVR rates following 24 weeks of treatment were 93% for dual therapy with ribavirin and 100% with no ribavirin. The findings cast further doubt on the benefit of using ribavirin with newer antiviral agents.
"SVR12 rates were high, regardless of baseline characteristics," Dr. Eric Lawitz said when presenting these data from the COSMOS study at the International Liver Congress 2014.
Dr. Lawitz, of the Texas Liver Institute at the University of Texas Health Science Center in San Antonio, added that the baseline characteristics included prior treatment history, METAVIR score, and the presence of known resistance factors – the Q80K polymorphism or IL28B genotype.
Simeprevir (Olysio) and sofosbuvir (Solvadi) are once-daily formulations recently approved for the treatment of chronic hepatitis C virus (HCV) genotype 1 in several countries, including Canada and the United States.
The aim of the COSMOS study was to test the efficacy and safety of these two novel antiviral treatments in tandem in patients with chronic HCV genotype 1 infection who had not previously received any anti-HCV therapy or who had been treated with pegylated interferon plus ribavirin but had not adequately responded.
There were two cohorts of patients, Dr. Lawitz explained. The first cohort comprised 80 patients without cirrhosis (METAVIR F0-F2) who had not responded to prior treatment, while the second cohort included 87 patients with advanced cirrhosis (METAVIR F3-F4) who were either null responders or treatment naive. He presented data only on cohort 2.
Patients were randomized to receive 12 or 24 weeks of simeprevir plus sofosbuvir with or without additional ribavirin. The median age of the study population overall was 58 years, 67% was male, and 91% were white. Just less than half of randomized patients had cirrhosis, and 54% were null responders. Dr. Lawitz pointed out that genotype 1a was the most prevalent subtype of HCV, with around 40% having the Q80K polymorphism and 79% having the IL28B (non-CC) genotype.
Three patients randomized to the 24-week study arm discontinued treatment early. Reasons for discontinuation included one death unrelated to treatment, withdrawal of consent by one patient, and one adverse event unrelated to treatment (dual therapy–only arm).
"When we look at all 87 patients, the SVR12 was 82/87, with two [2.3%] patients failing for nonvirologic reasons, and three [3.4%] patients relapsing," Dr. Lawitz reported at the meeting, which was sponsored by the European Association for the Study of the Liver.
SVR12 rates were 98% and 95% in patients with METAVIR F3 and F4, respectively, and 96% and 94% in null responders and never-treated patients. SVR12 in patients with the IL28B genotype were 94%, 98%, and 95%, respectively, for the CC, CT, and TT forms.
Looking more closely at the three patients with viral relapse, no common factors were found that could help to predict the response to treatment.
On the whole, simeprevir plus sofosbuvir with or without ribavirin was generally well tolerated, Dr. Lawitz said. The most frequent adverse events were fatigue, headache, and nausea, occurring in 37.9%, 19.5%, and 17.2% of the overall study population, respectively.
"Numerically, to look at adverse events, they were more frequent in the ribavirin-containing arms compared to the ribavirin-free arms," he observed. And although bilirubin elevations were frequent, they were mostly mild and occurred primarily in the ribavirin-containing arms.
The study was sponsored by Janssen. Dr. Lawitz has received research support and speaker fees, and has acted as an advisory board member for Janssen, Gilead Sciences, and several other companies.
LONDON – Cure rates of 93%-100% were achieved in patients with chronic hepatitis C virus genotype 1 treated with both simeprevir and sofosbuvir despite the presence of advanced cirrhosis in a phase IIa trial.
Sustained virologic responses with dual therapy for 12 weeks (SVR12) were 93% with and 93% without the additional use of ribavirin. SVR rates following 24 weeks of treatment were 93% for dual therapy with ribavirin and 100% with no ribavirin. The findings cast further doubt on the benefit of using ribavirin with newer antiviral agents.
"SVR12 rates were high, regardless of baseline characteristics," Dr. Eric Lawitz said when presenting these data from the COSMOS study at the International Liver Congress 2014.
Dr. Lawitz, of the Texas Liver Institute at the University of Texas Health Science Center in San Antonio, added that the baseline characteristics included prior treatment history, METAVIR score, and the presence of known resistance factors – the Q80K polymorphism or IL28B genotype.
Simeprevir (Olysio) and sofosbuvir (Solvadi) are once-daily formulations recently approved for the treatment of chronic hepatitis C virus (HCV) genotype 1 in several countries, including Canada and the United States.
The aim of the COSMOS study was to test the efficacy and safety of these two novel antiviral treatments in tandem in patients with chronic HCV genotype 1 infection who had not previously received any anti-HCV therapy or who had been treated with pegylated interferon plus ribavirin but had not adequately responded.
There were two cohorts of patients, Dr. Lawitz explained. The first cohort comprised 80 patients without cirrhosis (METAVIR F0-F2) who had not responded to prior treatment, while the second cohort included 87 patients with advanced cirrhosis (METAVIR F3-F4) who were either null responders or treatment naive. He presented data only on cohort 2.
Patients were randomized to receive 12 or 24 weeks of simeprevir plus sofosbuvir with or without additional ribavirin. The median age of the study population overall was 58 years, 67% was male, and 91% were white. Just less than half of randomized patients had cirrhosis, and 54% were null responders. Dr. Lawitz pointed out that genotype 1a was the most prevalent subtype of HCV, with around 40% having the Q80K polymorphism and 79% having the IL28B (non-CC) genotype.
Three patients randomized to the 24-week study arm discontinued treatment early. Reasons for discontinuation included one death unrelated to treatment, withdrawal of consent by one patient, and one adverse event unrelated to treatment (dual therapy–only arm).
"When we look at all 87 patients, the SVR12 was 82/87, with two [2.3%] patients failing for nonvirologic reasons, and three [3.4%] patients relapsing," Dr. Lawitz reported at the meeting, which was sponsored by the European Association for the Study of the Liver.
SVR12 rates were 98% and 95% in patients with METAVIR F3 and F4, respectively, and 96% and 94% in null responders and never-treated patients. SVR12 in patients with the IL28B genotype were 94%, 98%, and 95%, respectively, for the CC, CT, and TT forms.
Looking more closely at the three patients with viral relapse, no common factors were found that could help to predict the response to treatment.
On the whole, simeprevir plus sofosbuvir with or without ribavirin was generally well tolerated, Dr. Lawitz said. The most frequent adverse events were fatigue, headache, and nausea, occurring in 37.9%, 19.5%, and 17.2% of the overall study population, respectively.
"Numerically, to look at adverse events, they were more frequent in the ribavirin-containing arms compared to the ribavirin-free arms," he observed. And although bilirubin elevations were frequent, they were mostly mild and occurred primarily in the ribavirin-containing arms.
The study was sponsored by Janssen. Dr. Lawitz has received research support and speaker fees, and has acted as an advisory board member for Janssen, Gilead Sciences, and several other companies.
AT THE INTERNATIONAL LIVER CONGRESS 2014
Major finding: SVR12 rates were 93% and 93% after 12 weeks and 93% and 100% after 24 weeks of dual treatment with or without ribavirin.
Data source: Open-label, multicenter, randomized phase IIa study of simeprevir plus sofosbuvir with or without ribavirin for the treatment of hepatitis C virus genotype 1 in 87 treatment-naive or nonresponding patients with advanced cirrhosis.
Disclosures: The study was sponsored by Janssen. Dr. Lawitz has received research support and speaker fees, and has acted as an advisory board member for Janssen, Gilead Sciences, and several other companies.
Herbal medicines and supplements carry risk of hepatoxicity
LONDON – Herbal medicines and other home remedies or supplements are a significant cause of hepatotoxicity experts warned recently during a symposium at the International Liver Congress, which unintentionally coincided with World Homeopathy Awareness Week.
Although they are not at the very top of the list when it comes to drug-induced liver injury (DILI) – that accolade being reserved for antimicrobial agents used to treat tuberculosis – the use of homeopathy-based approaches are potentially on the increase in the western world and the use of such substances are often not reported to physicians.
"Herbal medicines represent a significant cause of liver injury," Dr. Dominique Larrey (Central University Hospital, Montpellier, France), said at the meeting that was sponsored by the European Association for the Study of the Liver. "Herbs can cause almost the whole spectrum of hepatic and biliary lesions, acute hepatitis being the most frequent one," he added.
The rise of herbal medicine
Dr. Larrey, who works in the liver and transplantation department of Saint Eloi Hospital, also in Montpellier, noted that the use of herbs in traditional medicine was very important in many parts of the world, notably in Asia, Africa, and Central and South America. They are used for both traditional and cultural reasons, he added, are often easy to access and are low cost in comparison to regulated medicines.
Their use is probably on the increase in western countries for a variety of reasons, Dr. Larrey suggested, such as the migration of people from cultures in which the use of traditional medicines is high, to the thinking that "what is natural can only be good" and "herbal medicines are considered completely innocuous in contrast to classical drugs." Furthermore, the lack of satisfactory treatments for some severe diseases – cancer, multiple sclerosis, AIDS, and hepatitis C virus infection to name a few – mean that people often are willing to try out complementary or alternative medicines (CAM).
In the United States, the total sale of herbal remedies in 2010 was an estimated $5.2 billion per year, having increased around 3% a year over the past decade, Dr. Larrey pointed out.
The problem is that patients do not often tell their doctors about their use of CAM. A staggering 90% of patients taking the anticoagulant drug warfarin – which is renowned for having a very narrow therapeutic window and careful monitoring is required – were taking herbal medicines in one study, he said.
Prospective studies on the use of herbal medicines in western countries are scarce but those that have been conducted specifically in patients with liver disease suggest that as many as one-fifth (Hepatology 2008;47:605-12) to one-third (Gastroenterology 2001;120[Suppl 1]:A228) might be taking herbal remedies unbeknownst to their doctor.
The problem of assessment
There are limited data on how frequently herbal medicines cause liver damage, but estimates range from 2% to 16%, Dr. Larrey observed, adding that reported cases could be just the tip of the iceberg.
"Herbal medicine hepatoxicity is clearly underestimated for many reasons," he suggested. First, their intake is hard to analyze. Second, the mechanism of liver damage is often uncertain, and third, it is hard to confirm causality. Indeed, herbal medicines do not have to undergo the rigorous testing or regulation in the same way that prescribed medicines do, and sales via the Internet make them easily available to all.
There is then the uncertainty of what is really in the preparations, if they contain the right plant at all or the wrong part of it, and then whether or not they have been stored correctly, or if they have been contaminated with other liver-damaging agents or microorganisms.
Advice for physicians
DILI from prescription and nonprescription medicines is an important but rare event in the westernized world, Dr. Robert Fontana of the University of Michigan in Ann Arbor said in an interview. However, because it can bring about very bad and unpredictable liver injury, it is of great importance for hepatologists and general family physicians alike.
"In the United States and I think worldwide, the frequency in use of [herbal treatments] is increasing and as we start to see registry data I think we will start to see more and more cases [of hepatoxicity]," Dr. Fontana said.
Dr. Fontana is part of the National Institutes of Health–funded Drug-Induced Liver Injury Network (DILIN), a multicenter, prospective registry looking at the etiologies, risk factors, and outcomes of DILI in the United States (Drug Saf. 2009;32:55-68). Data from the registry show the prevalence of herbal and dietary supplements is around 9% (n = 300) in confirmed DILI cases.
"Patients need to tell their doctors what they are taking," he advised, adding that, as physicians, "we all need to be aware and maybe ask more questions of our patients."
The LiverTox website – produced by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Library of Medicine – is a valuable online and freely available resource for determining if a medication, herbal, or other supplement is known to cause liver problems. This is going to have a new chapter on herbal medicines, Dr. Fontana said, and is worth using in daily practice to help advise patients on the prescription or CAM they might be taking.
Dr. Larrey and Dr. Fontana had no disclosures relevant to their comments.
New Chinese herbal medicine inhibits HCV activity
A compound named SBEL1 after the laboratory in which it was discovered has multiple effects on the hepatitis C virus (HCV) life cycle, according to data from a late-breaking poster presented at the meeting.
Researchers from the Systems Biology of Epithelia Laboratory at the National Taiwan University, Taipei, screened six herbal medicines and found that one of these – SBEL1 – inhibited HCV activity by about 90% in infected cells.
Cheng-Wei Lin and Ming-Jiun Yu pretreated liver cells with the herbal extract and then infected these cells with HCV. Compared with control cells, SBEL1-treated cells contained 23% less viral protein. This suggested that SBEL1 prevented HCV from entering the pretreated cells.
Their findings also suggested that SBEL1 reduced internal-ribosome entry site–mediated translation, a process vital for viral protein production, and might also have interfered with the RNA replication process.
"SBEL1 has demonstrated significant inhibition of HCV at multiple stages of the viral life cycle," Dr. Markus Peck-Radosavljevic, the secretary-general of the European Association for the Study of the Liver, said in a press release issued by the Society.
Dr. Peck-Radosavljevic (University of Vienna, Austria), who was not involved in the research, added that this "is an exciting discovery because it allows us to gain a deeper understanding of the virus and its interactions with other compounds. Ultimately, this adds to our library of knowledge that may bring us closer to improving future treatment options."
LONDON – Herbal medicines and other home remedies or supplements are a significant cause of hepatotoxicity experts warned recently during a symposium at the International Liver Congress, which unintentionally coincided with World Homeopathy Awareness Week.
Although they are not at the very top of the list when it comes to drug-induced liver injury (DILI) – that accolade being reserved for antimicrobial agents used to treat tuberculosis – the use of homeopathy-based approaches are potentially on the increase in the western world and the use of such substances are often not reported to physicians.
"Herbal medicines represent a significant cause of liver injury," Dr. Dominique Larrey (Central University Hospital, Montpellier, France), said at the meeting that was sponsored by the European Association for the Study of the Liver. "Herbs can cause almost the whole spectrum of hepatic and biliary lesions, acute hepatitis being the most frequent one," he added.
The rise of herbal medicine
Dr. Larrey, who works in the liver and transplantation department of Saint Eloi Hospital, also in Montpellier, noted that the use of herbs in traditional medicine was very important in many parts of the world, notably in Asia, Africa, and Central and South America. They are used for both traditional and cultural reasons, he added, are often easy to access and are low cost in comparison to regulated medicines.
Their use is probably on the increase in western countries for a variety of reasons, Dr. Larrey suggested, such as the migration of people from cultures in which the use of traditional medicines is high, to the thinking that "what is natural can only be good" and "herbal medicines are considered completely innocuous in contrast to classical drugs." Furthermore, the lack of satisfactory treatments for some severe diseases – cancer, multiple sclerosis, AIDS, and hepatitis C virus infection to name a few – mean that people often are willing to try out complementary or alternative medicines (CAM).
In the United States, the total sale of herbal remedies in 2010 was an estimated $5.2 billion per year, having increased around 3% a year over the past decade, Dr. Larrey pointed out.
The problem is that patients do not often tell their doctors about their use of CAM. A staggering 90% of patients taking the anticoagulant drug warfarin – which is renowned for having a very narrow therapeutic window and careful monitoring is required – were taking herbal medicines in one study, he said.
Prospective studies on the use of herbal medicines in western countries are scarce but those that have been conducted specifically in patients with liver disease suggest that as many as one-fifth (Hepatology 2008;47:605-12) to one-third (Gastroenterology 2001;120[Suppl 1]:A228) might be taking herbal remedies unbeknownst to their doctor.
The problem of assessment
There are limited data on how frequently herbal medicines cause liver damage, but estimates range from 2% to 16%, Dr. Larrey observed, adding that reported cases could be just the tip of the iceberg.
"Herbal medicine hepatoxicity is clearly underestimated for many reasons," he suggested. First, their intake is hard to analyze. Second, the mechanism of liver damage is often uncertain, and third, it is hard to confirm causality. Indeed, herbal medicines do not have to undergo the rigorous testing or regulation in the same way that prescribed medicines do, and sales via the Internet make them easily available to all.
There is then the uncertainty of what is really in the preparations, if they contain the right plant at all or the wrong part of it, and then whether or not they have been stored correctly, or if they have been contaminated with other liver-damaging agents or microorganisms.
Advice for physicians
DILI from prescription and nonprescription medicines is an important but rare event in the westernized world, Dr. Robert Fontana of the University of Michigan in Ann Arbor said in an interview. However, because it can bring about very bad and unpredictable liver injury, it is of great importance for hepatologists and general family physicians alike.
"In the United States and I think worldwide, the frequency in use of [herbal treatments] is increasing and as we start to see registry data I think we will start to see more and more cases [of hepatoxicity]," Dr. Fontana said.
Dr. Fontana is part of the National Institutes of Health–funded Drug-Induced Liver Injury Network (DILIN), a multicenter, prospective registry looking at the etiologies, risk factors, and outcomes of DILI in the United States (Drug Saf. 2009;32:55-68). Data from the registry show the prevalence of herbal and dietary supplements is around 9% (n = 300) in confirmed DILI cases.
"Patients need to tell their doctors what they are taking," he advised, adding that, as physicians, "we all need to be aware and maybe ask more questions of our patients."
The LiverTox website – produced by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Library of Medicine – is a valuable online and freely available resource for determining if a medication, herbal, or other supplement is known to cause liver problems. This is going to have a new chapter on herbal medicines, Dr. Fontana said, and is worth using in daily practice to help advise patients on the prescription or CAM they might be taking.
Dr. Larrey and Dr. Fontana had no disclosures relevant to their comments.
New Chinese herbal medicine inhibits HCV activity
A compound named SBEL1 after the laboratory in which it was discovered has multiple effects on the hepatitis C virus (HCV) life cycle, according to data from a late-breaking poster presented at the meeting.
Researchers from the Systems Biology of Epithelia Laboratory at the National Taiwan University, Taipei, screened six herbal medicines and found that one of these – SBEL1 – inhibited HCV activity by about 90% in infected cells.
Cheng-Wei Lin and Ming-Jiun Yu pretreated liver cells with the herbal extract and then infected these cells with HCV. Compared with control cells, SBEL1-treated cells contained 23% less viral protein. This suggested that SBEL1 prevented HCV from entering the pretreated cells.
Their findings also suggested that SBEL1 reduced internal-ribosome entry site–mediated translation, a process vital for viral protein production, and might also have interfered with the RNA replication process.
"SBEL1 has demonstrated significant inhibition of HCV at multiple stages of the viral life cycle," Dr. Markus Peck-Radosavljevic, the secretary-general of the European Association for the Study of the Liver, said in a press release issued by the Society.
Dr. Peck-Radosavljevic (University of Vienna, Austria), who was not involved in the research, added that this "is an exciting discovery because it allows us to gain a deeper understanding of the virus and its interactions with other compounds. Ultimately, this adds to our library of knowledge that may bring us closer to improving future treatment options."
LONDON – Herbal medicines and other home remedies or supplements are a significant cause of hepatotoxicity experts warned recently during a symposium at the International Liver Congress, which unintentionally coincided with World Homeopathy Awareness Week.
Although they are not at the very top of the list when it comes to drug-induced liver injury (DILI) – that accolade being reserved for antimicrobial agents used to treat tuberculosis – the use of homeopathy-based approaches are potentially on the increase in the western world and the use of such substances are often not reported to physicians.
"Herbal medicines represent a significant cause of liver injury," Dr. Dominique Larrey (Central University Hospital, Montpellier, France), said at the meeting that was sponsored by the European Association for the Study of the Liver. "Herbs can cause almost the whole spectrum of hepatic and biliary lesions, acute hepatitis being the most frequent one," he added.
The rise of herbal medicine
Dr. Larrey, who works in the liver and transplantation department of Saint Eloi Hospital, also in Montpellier, noted that the use of herbs in traditional medicine was very important in many parts of the world, notably in Asia, Africa, and Central and South America. They are used for both traditional and cultural reasons, he added, are often easy to access and are low cost in comparison to regulated medicines.
Their use is probably on the increase in western countries for a variety of reasons, Dr. Larrey suggested, such as the migration of people from cultures in which the use of traditional medicines is high, to the thinking that "what is natural can only be good" and "herbal medicines are considered completely innocuous in contrast to classical drugs." Furthermore, the lack of satisfactory treatments for some severe diseases – cancer, multiple sclerosis, AIDS, and hepatitis C virus infection to name a few – mean that people often are willing to try out complementary or alternative medicines (CAM).
In the United States, the total sale of herbal remedies in 2010 was an estimated $5.2 billion per year, having increased around 3% a year over the past decade, Dr. Larrey pointed out.
The problem is that patients do not often tell their doctors about their use of CAM. A staggering 90% of patients taking the anticoagulant drug warfarin – which is renowned for having a very narrow therapeutic window and careful monitoring is required – were taking herbal medicines in one study, he said.
Prospective studies on the use of herbal medicines in western countries are scarce but those that have been conducted specifically in patients with liver disease suggest that as many as one-fifth (Hepatology 2008;47:605-12) to one-third (Gastroenterology 2001;120[Suppl 1]:A228) might be taking herbal remedies unbeknownst to their doctor.
The problem of assessment
There are limited data on how frequently herbal medicines cause liver damage, but estimates range from 2% to 16%, Dr. Larrey observed, adding that reported cases could be just the tip of the iceberg.
"Herbal medicine hepatoxicity is clearly underestimated for many reasons," he suggested. First, their intake is hard to analyze. Second, the mechanism of liver damage is often uncertain, and third, it is hard to confirm causality. Indeed, herbal medicines do not have to undergo the rigorous testing or regulation in the same way that prescribed medicines do, and sales via the Internet make them easily available to all.
There is then the uncertainty of what is really in the preparations, if they contain the right plant at all or the wrong part of it, and then whether or not they have been stored correctly, or if they have been contaminated with other liver-damaging agents or microorganisms.
Advice for physicians
DILI from prescription and nonprescription medicines is an important but rare event in the westernized world, Dr. Robert Fontana of the University of Michigan in Ann Arbor said in an interview. However, because it can bring about very bad and unpredictable liver injury, it is of great importance for hepatologists and general family physicians alike.
"In the United States and I think worldwide, the frequency in use of [herbal treatments] is increasing and as we start to see registry data I think we will start to see more and more cases [of hepatoxicity]," Dr. Fontana said.
Dr. Fontana is part of the National Institutes of Health–funded Drug-Induced Liver Injury Network (DILIN), a multicenter, prospective registry looking at the etiologies, risk factors, and outcomes of DILI in the United States (Drug Saf. 2009;32:55-68). Data from the registry show the prevalence of herbal and dietary supplements is around 9% (n = 300) in confirmed DILI cases.
"Patients need to tell their doctors what they are taking," he advised, adding that, as physicians, "we all need to be aware and maybe ask more questions of our patients."
The LiverTox website – produced by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Library of Medicine – is a valuable online and freely available resource for determining if a medication, herbal, or other supplement is known to cause liver problems. This is going to have a new chapter on herbal medicines, Dr. Fontana said, and is worth using in daily practice to help advise patients on the prescription or CAM they might be taking.
Dr. Larrey and Dr. Fontana had no disclosures relevant to their comments.
New Chinese herbal medicine inhibits HCV activity
A compound named SBEL1 after the laboratory in which it was discovered has multiple effects on the hepatitis C virus (HCV) life cycle, according to data from a late-breaking poster presented at the meeting.
Researchers from the Systems Biology of Epithelia Laboratory at the National Taiwan University, Taipei, screened six herbal medicines and found that one of these – SBEL1 – inhibited HCV activity by about 90% in infected cells.
Cheng-Wei Lin and Ming-Jiun Yu pretreated liver cells with the herbal extract and then infected these cells with HCV. Compared with control cells, SBEL1-treated cells contained 23% less viral protein. This suggested that SBEL1 prevented HCV from entering the pretreated cells.
Their findings also suggested that SBEL1 reduced internal-ribosome entry site–mediated translation, a process vital for viral protein production, and might also have interfered with the RNA replication process.
"SBEL1 has demonstrated significant inhibition of HCV at multiple stages of the viral life cycle," Dr. Markus Peck-Radosavljevic, the secretary-general of the European Association for the Study of the Liver, said in a press release issued by the Society.
Dr. Peck-Radosavljevic (University of Vienna, Austria), who was not involved in the research, added that this "is an exciting discovery because it allows us to gain a deeper understanding of the virus and its interactions with other compounds. Ultimately, this adds to our library of knowledge that may bring us closer to improving future treatment options."
AT THE INTERNATIONAL LIVER CONGRESS 2014
All-oral, triple-acting HCV regimen cures more than 90% of cirrhotic cases
LONDON – More than 90% of patients with chronic hepatitis C and compensated cirrhosis treated with a triple-acting, all-oral, direct-acting antiviral (DAA) regimen achieved sustained virologic responses 12 weeks after the end of therapy (SVR12) in a phase III trial.
In the TURQUOISE II study, 380 patients infected with the hepatitis C virus (HVC) genotype 1 were treated with the interferon (IFN)-free combination of ABT-450/ritonavir, ombitasvir, and dasabuvir with or without ribavirin for 12 or 24 weeks. Results showed that SVR12 rates were 91.8% and 95.9%.
Results were striking, compared with a historical control group using telaprevir plus pegylated IFN plus ribavirin, with the threshold for noninferiority and superiority set at 43% and 54%, respectively.
The study is the first to look at the effects of an all-oral, direct-acting antiviral regimen exclusively in patients with HVC genotype 1 infection and cirrhosis, said the lead study investigator Dr. Fred Poordad. The study findings were published online to coincide with his presentation at the International Liver Congress 2014 (N. Engl. J. Med. 2014 Apr. 12 [doi: 10.1056/NEJMoa1402869]).
TURQUOISE II was an international, multicenter, randomized, open-label trial of 380 patients with Child-Pugh class A cirrhosis caused by HCV infection, explained Dr. Poordad of the Texas Liver Institute at the University of Texas Health Science Center, San Antonio. The trial was conducted at 78 sites in Europe and North America and included both treatment-experienced (about 60%) and treatment-naive (about 40%) patients.
Results by HCV subtype showed that the regimen worked equally well in patients with genotype 1a as genotype 1b, with SVR12 rates of 88.6% and 98.5% with 12 weeks’ treatment and 94.2% and 100% with 24 weeks’ treatment, respectively.
SVR rates were also similarly high in patients according to prior treatment response and in those with portal hypertension or reduced hepatic function identified by low baseline platelet counts or low serum albumin.
Dr. Poordad noted that 8.2% and 4.1% of patients did not achieve SVR12 in the 12- and 24-week therapy groups, respectively. Premature discontinuations occurred in 1.9% and 1.7% and were with caused by an adverse event (four vs. one case) or withdrawal of consent (0 vs. two cases).
"Virologic breakthrough was an unusual event in either arm (0.5% and 1.7%, respectively)," the researcher observed. Relapse occurred in 5.9% and 0.6% of patients in the 12- and 24-week arms. "Of the 12 patients who relapsed in the 12-week arm, 7 of them were [genotype]1a null responders, largely driving the difference in relapse between these two arms," Dr. Poordad said.
He noted that 17 patients overall had a virologic failure and of these, 15 of them had at least one resistance-associated variant. The most common variants in GT1a-infected patients were D168V found in NS3 and Q30R found in NS5A. The significance and persistence of these variants is currently under investigation.
In terms of tolerability, fatigue, and shortness of breath were the most common adverse events seen, occurring in a higher percentage of patients treated with 24 (46.5% and 12.2%) vs. 12 (12.2% vs. 5.8%) weeks of treatment (P less than .05 for both comparisons). There were low rates of severe or serious adverse events and low rates of treatment discontinuation (about 2%).
"The efficacy and safety in this large cirrhotic population is similar to noncirrhotics treated with the same regimen," Dr. Poordad noted. He highlighted that the three DAA antiviral regimen – nicknamed 3D – has also been assessed in two pivotal phase III trials and has shown exceptionally high SVR12 rates. In the SAPPHIRE I trial (N. Engl. J. Med. 2014 Apr. 10 [doi:10.1056/NEJMoa1315722]), in treatment-naive patients without cirrhosis, SVR12 after 12 weeks’ therapy with the 3D regimen was 96.2% and in the SAPPHIRE II trial (N. Engl. J. Med. 2014 Apr. 10 [doi: 10.1056/NEJMoa1401561]) in treatment-experienced patients without cirrhosis it was 96.3% in treatment-experienced patients, which included those who had previously not responded to IFN-based therapy. The results of both SAPPHIRE trials were also presented at the meeting, which was sponsored by the European Association for the Study of the Liver.
Dr. Jean-Michel Pawlotsky of Hôpital Henri Mondor, Créteil, France, who chaired the late-breakers session at which the TURQUOISE II data were presented, commented that these data were "outstanding." He asked Dr. Poordad to comment on the treatment duration and when a longer treatment course might be warranted in certain patients.
"I think one has to use their clinical judgment, so if you have a patient who comes in and you cannot determine if they were a previous nonresponder or not then you can certainly consider treating a little bit longer," the investigator responded. "They certainly tolerate treatment just as well."
AbbVie sponsored the study. Dr. Poordad has received research support and acted as a consultant to the company. He also disclosed receiving grants or research support or acting as an adviser or speaker for Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Gilead Sciences, GlaxoSmithKline, in addition to others.
LONDON – More than 90% of patients with chronic hepatitis C and compensated cirrhosis treated with a triple-acting, all-oral, direct-acting antiviral (DAA) regimen achieved sustained virologic responses 12 weeks after the end of therapy (SVR12) in a phase III trial.
In the TURQUOISE II study, 380 patients infected with the hepatitis C virus (HVC) genotype 1 were treated with the interferon (IFN)-free combination of ABT-450/ritonavir, ombitasvir, and dasabuvir with or without ribavirin for 12 or 24 weeks. Results showed that SVR12 rates were 91.8% and 95.9%.
Results were striking, compared with a historical control group using telaprevir plus pegylated IFN plus ribavirin, with the threshold for noninferiority and superiority set at 43% and 54%, respectively.
The study is the first to look at the effects of an all-oral, direct-acting antiviral regimen exclusively in patients with HVC genotype 1 infection and cirrhosis, said the lead study investigator Dr. Fred Poordad. The study findings were published online to coincide with his presentation at the International Liver Congress 2014 (N. Engl. J. Med. 2014 Apr. 12 [doi: 10.1056/NEJMoa1402869]).
TURQUOISE II was an international, multicenter, randomized, open-label trial of 380 patients with Child-Pugh class A cirrhosis caused by HCV infection, explained Dr. Poordad of the Texas Liver Institute at the University of Texas Health Science Center, San Antonio. The trial was conducted at 78 sites in Europe and North America and included both treatment-experienced (about 60%) and treatment-naive (about 40%) patients.
Results by HCV subtype showed that the regimen worked equally well in patients with genotype 1a as genotype 1b, with SVR12 rates of 88.6% and 98.5% with 12 weeks’ treatment and 94.2% and 100% with 24 weeks’ treatment, respectively.
SVR rates were also similarly high in patients according to prior treatment response and in those with portal hypertension or reduced hepatic function identified by low baseline platelet counts or low serum albumin.
Dr. Poordad noted that 8.2% and 4.1% of patients did not achieve SVR12 in the 12- and 24-week therapy groups, respectively. Premature discontinuations occurred in 1.9% and 1.7% and were with caused by an adverse event (four vs. one case) or withdrawal of consent (0 vs. two cases).
"Virologic breakthrough was an unusual event in either arm (0.5% and 1.7%, respectively)," the researcher observed. Relapse occurred in 5.9% and 0.6% of patients in the 12- and 24-week arms. "Of the 12 patients who relapsed in the 12-week arm, 7 of them were [genotype]1a null responders, largely driving the difference in relapse between these two arms," Dr. Poordad said.
He noted that 17 patients overall had a virologic failure and of these, 15 of them had at least one resistance-associated variant. The most common variants in GT1a-infected patients were D168V found in NS3 and Q30R found in NS5A. The significance and persistence of these variants is currently under investigation.
In terms of tolerability, fatigue, and shortness of breath were the most common adverse events seen, occurring in a higher percentage of patients treated with 24 (46.5% and 12.2%) vs. 12 (12.2% vs. 5.8%) weeks of treatment (P less than .05 for both comparisons). There were low rates of severe or serious adverse events and low rates of treatment discontinuation (about 2%).
"The efficacy and safety in this large cirrhotic population is similar to noncirrhotics treated with the same regimen," Dr. Poordad noted. He highlighted that the three DAA antiviral regimen – nicknamed 3D – has also been assessed in two pivotal phase III trials and has shown exceptionally high SVR12 rates. In the SAPPHIRE I trial (N. Engl. J. Med. 2014 Apr. 10 [doi:10.1056/NEJMoa1315722]), in treatment-naive patients without cirrhosis, SVR12 after 12 weeks’ therapy with the 3D regimen was 96.2% and in the SAPPHIRE II trial (N. Engl. J. Med. 2014 Apr. 10 [doi: 10.1056/NEJMoa1401561]) in treatment-experienced patients without cirrhosis it was 96.3% in treatment-experienced patients, which included those who had previously not responded to IFN-based therapy. The results of both SAPPHIRE trials were also presented at the meeting, which was sponsored by the European Association for the Study of the Liver.
Dr. Jean-Michel Pawlotsky of Hôpital Henri Mondor, Créteil, France, who chaired the late-breakers session at which the TURQUOISE II data were presented, commented that these data were "outstanding." He asked Dr. Poordad to comment on the treatment duration and when a longer treatment course might be warranted in certain patients.
"I think one has to use their clinical judgment, so if you have a patient who comes in and you cannot determine if they were a previous nonresponder or not then you can certainly consider treating a little bit longer," the investigator responded. "They certainly tolerate treatment just as well."
AbbVie sponsored the study. Dr. Poordad has received research support and acted as a consultant to the company. He also disclosed receiving grants or research support or acting as an adviser or speaker for Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Gilead Sciences, GlaxoSmithKline, in addition to others.
LONDON – More than 90% of patients with chronic hepatitis C and compensated cirrhosis treated with a triple-acting, all-oral, direct-acting antiviral (DAA) regimen achieved sustained virologic responses 12 weeks after the end of therapy (SVR12) in a phase III trial.
In the TURQUOISE II study, 380 patients infected with the hepatitis C virus (HVC) genotype 1 were treated with the interferon (IFN)-free combination of ABT-450/ritonavir, ombitasvir, and dasabuvir with or without ribavirin for 12 or 24 weeks. Results showed that SVR12 rates were 91.8% and 95.9%.
Results were striking, compared with a historical control group using telaprevir plus pegylated IFN plus ribavirin, with the threshold for noninferiority and superiority set at 43% and 54%, respectively.
The study is the first to look at the effects of an all-oral, direct-acting antiviral regimen exclusively in patients with HVC genotype 1 infection and cirrhosis, said the lead study investigator Dr. Fred Poordad. The study findings were published online to coincide with his presentation at the International Liver Congress 2014 (N. Engl. J. Med. 2014 Apr. 12 [doi: 10.1056/NEJMoa1402869]).
TURQUOISE II was an international, multicenter, randomized, open-label trial of 380 patients with Child-Pugh class A cirrhosis caused by HCV infection, explained Dr. Poordad of the Texas Liver Institute at the University of Texas Health Science Center, San Antonio. The trial was conducted at 78 sites in Europe and North America and included both treatment-experienced (about 60%) and treatment-naive (about 40%) patients.
Results by HCV subtype showed that the regimen worked equally well in patients with genotype 1a as genotype 1b, with SVR12 rates of 88.6% and 98.5% with 12 weeks’ treatment and 94.2% and 100% with 24 weeks’ treatment, respectively.
SVR rates were also similarly high in patients according to prior treatment response and in those with portal hypertension or reduced hepatic function identified by low baseline platelet counts or low serum albumin.
Dr. Poordad noted that 8.2% and 4.1% of patients did not achieve SVR12 in the 12- and 24-week therapy groups, respectively. Premature discontinuations occurred in 1.9% and 1.7% and were with caused by an adverse event (four vs. one case) or withdrawal of consent (0 vs. two cases).
"Virologic breakthrough was an unusual event in either arm (0.5% and 1.7%, respectively)," the researcher observed. Relapse occurred in 5.9% and 0.6% of patients in the 12- and 24-week arms. "Of the 12 patients who relapsed in the 12-week arm, 7 of them were [genotype]1a null responders, largely driving the difference in relapse between these two arms," Dr. Poordad said.
He noted that 17 patients overall had a virologic failure and of these, 15 of them had at least one resistance-associated variant. The most common variants in GT1a-infected patients were D168V found in NS3 and Q30R found in NS5A. The significance and persistence of these variants is currently under investigation.
In terms of tolerability, fatigue, and shortness of breath were the most common adverse events seen, occurring in a higher percentage of patients treated with 24 (46.5% and 12.2%) vs. 12 (12.2% vs. 5.8%) weeks of treatment (P less than .05 for both comparisons). There were low rates of severe or serious adverse events and low rates of treatment discontinuation (about 2%).
"The efficacy and safety in this large cirrhotic population is similar to noncirrhotics treated with the same regimen," Dr. Poordad noted. He highlighted that the three DAA antiviral regimen – nicknamed 3D – has also been assessed in two pivotal phase III trials and has shown exceptionally high SVR12 rates. In the SAPPHIRE I trial (N. Engl. J. Med. 2014 Apr. 10 [doi:10.1056/NEJMoa1315722]), in treatment-naive patients without cirrhosis, SVR12 after 12 weeks’ therapy with the 3D regimen was 96.2% and in the SAPPHIRE II trial (N. Engl. J. Med. 2014 Apr. 10 [doi: 10.1056/NEJMoa1401561]) in treatment-experienced patients without cirrhosis it was 96.3% in treatment-experienced patients, which included those who had previously not responded to IFN-based therapy. The results of both SAPPHIRE trials were also presented at the meeting, which was sponsored by the European Association for the Study of the Liver.
Dr. Jean-Michel Pawlotsky of Hôpital Henri Mondor, Créteil, France, who chaired the late-breakers session at which the TURQUOISE II data were presented, commented that these data were "outstanding." He asked Dr. Poordad to comment on the treatment duration and when a longer treatment course might be warranted in certain patients.
"I think one has to use their clinical judgment, so if you have a patient who comes in and you cannot determine if they were a previous nonresponder or not then you can certainly consider treating a little bit longer," the investigator responded. "They certainly tolerate treatment just as well."
AbbVie sponsored the study. Dr. Poordad has received research support and acted as a consultant to the company. He also disclosed receiving grants or research support or acting as an adviser or speaker for Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Gilead Sciences, GlaxoSmithKline, in addition to others.
AT THE INTERNATIONAL LIVER CONGRESS 2014
Major finding: Sustained virologic responses 12 weeks after treatment was stopped were 91.8% and 96.1%, respectively, with a 12-week and a 24-week regimen.
Data source: TURQUOISE II, a phase III international, multicenter, randomized, open-label trial of 380 patients with Child-Pugh class A cirrhosis caused by hepatitis C viral infection treated with the interferon-free combination of ABD-450/ritnavir, ombitasvir, and dasabuvir with or without ribavirin for 12 or 24 weeks.
Disclosures: AbbVie sponsored the study. Dr. Poordad has received research support and acted as a consultant to the company. He also disclosed receiving grants or research support or acting as an adviser or speaker for Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Gilead Sciences, GlaxoSmithKline, in addition to others.
Antiviral combo pill cures HCV in 8-12 weeks
LONDON – A single daily pill containing two novel antiviral agents produced sustained virologic response rates of more than 90% in as little as 8-12 weeks in three multicenter, open-label phase III studies involving 1,952 patients with chronic hepatitis C virus infection.
The combination pill, containing fixed doses of 400 mg sofosbuvir and 90 mg ledipasvir, was given alone or together with ribavirin but without pegylated interferon (peg-IFN) for 8, 12, or 24 weeks in the ION-1, ION-2, and ION-3 trials. Results of the first two trials showed that a 12-week course of sofosbuvir/ledipasvir was enough to clear the virus in the majority of patients, but results of the latter showed that an 8-week regimen of the daily pill was sufficient.
The findings of all three studies were published online in the New England Journal of Medicine to coincide with their presentation at the International Liver Congress sponsored by the European Association for the Study of the Liver.
"The real advance seen in the ION trials is that the sofosbuvir-ledipasvir combination tablet enables us to treat almost all genotype 1 patients with a short duration of 8-12 weeks of treatment," Dr. Nezam Afdhal* of Beth Israel Deaconess Medical Center, Boston, said in a press statement issued by his institution, which was one of the key centers involved in the studies.
Since patients who were unable to be treated with peg-IFN were included in the studies, these data could potentially expand the population of patients who could now be treated, as well as increase the overall cure rate, Dr. Afdhal added. Patients with compensated cirrhosis also were included in the ION-1 (n = 136) and ION-2 trials (n = 88).
ION-1 (N. Engl. J. Med. 2014 Apr. 12 [doi:10.1056/NEJMoa1402454]) and ION-2 (N. Engl. J. Med. 2014 Apr. 12 [doi:10.1056/NEJMoa1316366]) were designed to investigate the efficacy and safety of the sofosbuvir/ledipasvir fixed-dose combination given with or without ribavirin for 12 and 24 weeks in patients with chronic genotype 1 hepatitis C virus (HCV) infection who were treatment experienced and treatment naive, respectively. Results showed that similar cure rates could be achieved in patients regardless of the duration of the regimen.
Indeed, sustained virologic response at 12 weeks after the end of treatment (SVR12) in ION-1 in treatment-naive patients (n = 865) was 99% with the daily pill alone and 97% if ribavirin also was given. There was little benefit of continuing therapy for up to 24 weeks, with SVR12 of 98% for the daily pill given alone and 99% with additional ribavirin.
SVR12 in previously treated patients in the ION-2 trial (n = 440) was 94% with the daily pill alone and 96% if ribavirin also was given for 12 weeks. There was little benefit of continuing therapy for 24 weeks, with SVR12 of 99% for the daily pill used alone and together with ribavirin.
ION-3 (N. Engl. J. Med. 2014 Apr. 11 [doi:10.1056/NEJMoa1402355]) looked at whether 8 weeks of treatment was as effective as 12 weeks of treatment. The open-label study included 647 patients with HCV genotype 1 infection without cirrhosis who had not been previously treated. SVR at 8 weeks was 94%, versus 95% for the 12-week regimen. The addition of ribavirin did not increase the SVR, which was 93% at 8 weeks, showing that a shorter regimen was just as effective as the 12-week course.
Adverse events were more common in the ribavirin-treated patients than in those who received only the combined sofosbuvir/ledipasvir pill. The most common adverse events seen in the trials – fatigue, headache, insomnia, and nausea – were linked to ribavirin therapy.
"With cure rates well in excess of 90% with as little as 8 weeks of treatment for some patients, these data represent a significant advance in the race to develop a new, all-oral treatment for hepatitis C," said Dr. Markus Peck-Radosavljevic, the Secretary General of EASL. He noted, in a press statement issued by the association, that avoiding the use of both peg-IFN and ribavirin had the potential to bring forward more patients for treatment as it is likely to be much more tolerable.
Gilead Sciences funded the trials. Dr. Afdhal has received research support, honoraria, or other fees in the past 12 months from Gilead Sciences, Abbott, BMS, Boehringer Ingelheim, Echosens, Janssen, GSK, Idenix, Ligan, Merck, Medgenics, Novartis, Quest, Springbank, TRIO Healthcare, and Vertex. Dr. Peck-Radosavljevic made no disclosures.
*Correction 5/8/2014: In a previous version of the story Dr. Afdhal's name was misspelled. This version has been updated.
LONDON – A single daily pill containing two novel antiviral agents produced sustained virologic response rates of more than 90% in as little as 8-12 weeks in three multicenter, open-label phase III studies involving 1,952 patients with chronic hepatitis C virus infection.
The combination pill, containing fixed doses of 400 mg sofosbuvir and 90 mg ledipasvir, was given alone or together with ribavirin but without pegylated interferon (peg-IFN) for 8, 12, or 24 weeks in the ION-1, ION-2, and ION-3 trials. Results of the first two trials showed that a 12-week course of sofosbuvir/ledipasvir was enough to clear the virus in the majority of patients, but results of the latter showed that an 8-week regimen of the daily pill was sufficient.
The findings of all three studies were published online in the New England Journal of Medicine to coincide with their presentation at the International Liver Congress sponsored by the European Association for the Study of the Liver.
"The real advance seen in the ION trials is that the sofosbuvir-ledipasvir combination tablet enables us to treat almost all genotype 1 patients with a short duration of 8-12 weeks of treatment," Dr. Nezam Afdhal* of Beth Israel Deaconess Medical Center, Boston, said in a press statement issued by his institution, which was one of the key centers involved in the studies.
Since patients who were unable to be treated with peg-IFN were included in the studies, these data could potentially expand the population of patients who could now be treated, as well as increase the overall cure rate, Dr. Afdhal added. Patients with compensated cirrhosis also were included in the ION-1 (n = 136) and ION-2 trials (n = 88).
ION-1 (N. Engl. J. Med. 2014 Apr. 12 [doi:10.1056/NEJMoa1402454]) and ION-2 (N. Engl. J. Med. 2014 Apr. 12 [doi:10.1056/NEJMoa1316366]) were designed to investigate the efficacy and safety of the sofosbuvir/ledipasvir fixed-dose combination given with or without ribavirin for 12 and 24 weeks in patients with chronic genotype 1 hepatitis C virus (HCV) infection who were treatment experienced and treatment naive, respectively. Results showed that similar cure rates could be achieved in patients regardless of the duration of the regimen.
Indeed, sustained virologic response at 12 weeks after the end of treatment (SVR12) in ION-1 in treatment-naive patients (n = 865) was 99% with the daily pill alone and 97% if ribavirin also was given. There was little benefit of continuing therapy for up to 24 weeks, with SVR12 of 98% for the daily pill given alone and 99% with additional ribavirin.
SVR12 in previously treated patients in the ION-2 trial (n = 440) was 94% with the daily pill alone and 96% if ribavirin also was given for 12 weeks. There was little benefit of continuing therapy for 24 weeks, with SVR12 of 99% for the daily pill used alone and together with ribavirin.
ION-3 (N. Engl. J. Med. 2014 Apr. 11 [doi:10.1056/NEJMoa1402355]) looked at whether 8 weeks of treatment was as effective as 12 weeks of treatment. The open-label study included 647 patients with HCV genotype 1 infection without cirrhosis who had not been previously treated. SVR at 8 weeks was 94%, versus 95% for the 12-week regimen. The addition of ribavirin did not increase the SVR, which was 93% at 8 weeks, showing that a shorter regimen was just as effective as the 12-week course.
Adverse events were more common in the ribavirin-treated patients than in those who received only the combined sofosbuvir/ledipasvir pill. The most common adverse events seen in the trials – fatigue, headache, insomnia, and nausea – were linked to ribavirin therapy.
"With cure rates well in excess of 90% with as little as 8 weeks of treatment for some patients, these data represent a significant advance in the race to develop a new, all-oral treatment for hepatitis C," said Dr. Markus Peck-Radosavljevic, the Secretary General of EASL. He noted, in a press statement issued by the association, that avoiding the use of both peg-IFN and ribavirin had the potential to bring forward more patients for treatment as it is likely to be much more tolerable.
Gilead Sciences funded the trials. Dr. Afdhal has received research support, honoraria, or other fees in the past 12 months from Gilead Sciences, Abbott, BMS, Boehringer Ingelheim, Echosens, Janssen, GSK, Idenix, Ligan, Merck, Medgenics, Novartis, Quest, Springbank, TRIO Healthcare, and Vertex. Dr. Peck-Radosavljevic made no disclosures.
*Correction 5/8/2014: In a previous version of the story Dr. Afdhal's name was misspelled. This version has been updated.
LONDON – A single daily pill containing two novel antiviral agents produced sustained virologic response rates of more than 90% in as little as 8-12 weeks in three multicenter, open-label phase III studies involving 1,952 patients with chronic hepatitis C virus infection.
The combination pill, containing fixed doses of 400 mg sofosbuvir and 90 mg ledipasvir, was given alone or together with ribavirin but without pegylated interferon (peg-IFN) for 8, 12, or 24 weeks in the ION-1, ION-2, and ION-3 trials. Results of the first two trials showed that a 12-week course of sofosbuvir/ledipasvir was enough to clear the virus in the majority of patients, but results of the latter showed that an 8-week regimen of the daily pill was sufficient.
The findings of all three studies were published online in the New England Journal of Medicine to coincide with their presentation at the International Liver Congress sponsored by the European Association for the Study of the Liver.
"The real advance seen in the ION trials is that the sofosbuvir-ledipasvir combination tablet enables us to treat almost all genotype 1 patients with a short duration of 8-12 weeks of treatment," Dr. Nezam Afdhal* of Beth Israel Deaconess Medical Center, Boston, said in a press statement issued by his institution, which was one of the key centers involved in the studies.
Since patients who were unable to be treated with peg-IFN were included in the studies, these data could potentially expand the population of patients who could now be treated, as well as increase the overall cure rate, Dr. Afdhal added. Patients with compensated cirrhosis also were included in the ION-1 (n = 136) and ION-2 trials (n = 88).
ION-1 (N. Engl. J. Med. 2014 Apr. 12 [doi:10.1056/NEJMoa1402454]) and ION-2 (N. Engl. J. Med. 2014 Apr. 12 [doi:10.1056/NEJMoa1316366]) were designed to investigate the efficacy and safety of the sofosbuvir/ledipasvir fixed-dose combination given with or without ribavirin for 12 and 24 weeks in patients with chronic genotype 1 hepatitis C virus (HCV) infection who were treatment experienced and treatment naive, respectively. Results showed that similar cure rates could be achieved in patients regardless of the duration of the regimen.
Indeed, sustained virologic response at 12 weeks after the end of treatment (SVR12) in ION-1 in treatment-naive patients (n = 865) was 99% with the daily pill alone and 97% if ribavirin also was given. There was little benefit of continuing therapy for up to 24 weeks, with SVR12 of 98% for the daily pill given alone and 99% with additional ribavirin.
SVR12 in previously treated patients in the ION-2 trial (n = 440) was 94% with the daily pill alone and 96% if ribavirin also was given for 12 weeks. There was little benefit of continuing therapy for 24 weeks, with SVR12 of 99% for the daily pill used alone and together with ribavirin.
ION-3 (N. Engl. J. Med. 2014 Apr. 11 [doi:10.1056/NEJMoa1402355]) looked at whether 8 weeks of treatment was as effective as 12 weeks of treatment. The open-label study included 647 patients with HCV genotype 1 infection without cirrhosis who had not been previously treated. SVR at 8 weeks was 94%, versus 95% for the 12-week regimen. The addition of ribavirin did not increase the SVR, which was 93% at 8 weeks, showing that a shorter regimen was just as effective as the 12-week course.
Adverse events were more common in the ribavirin-treated patients than in those who received only the combined sofosbuvir/ledipasvir pill. The most common adverse events seen in the trials – fatigue, headache, insomnia, and nausea – were linked to ribavirin therapy.
"With cure rates well in excess of 90% with as little as 8 weeks of treatment for some patients, these data represent a significant advance in the race to develop a new, all-oral treatment for hepatitis C," said Dr. Markus Peck-Radosavljevic, the Secretary General of EASL. He noted, in a press statement issued by the association, that avoiding the use of both peg-IFN and ribavirin had the potential to bring forward more patients for treatment as it is likely to be much more tolerable.
Gilead Sciences funded the trials. Dr. Afdhal has received research support, honoraria, or other fees in the past 12 months from Gilead Sciences, Abbott, BMS, Boehringer Ingelheim, Echosens, Janssen, GSK, Idenix, Ligan, Merck, Medgenics, Novartis, Quest, Springbank, TRIO Healthcare, and Vertex. Dr. Peck-Radosavljevic made no disclosures.
*Correction 5/8/2014: In a previous version of the story Dr. Afdhal's name was misspelled. This version has been updated.
AT THE INTERNATIONAL LIVER CONGRESS 2014
Key clinical point: A sofosbuvir-ledipasvir combination tablet can be used in almost all genotype 1 patients with a short duration of 8-12 weeks of treatment.
Major finding: Sustained virologic responses at 8 weeks were 94% and 93%, respectively, for the fixed-dose combination pill alone or with ribavirin, and 95% at 12 weeks in the ION-3 trial.
Data source: Three multicenter, open-label, phase III studies involving 1,952 patients with chronic hepatitis C with or without cirrhosis who were randomized to receive 8, 12, or 24 weeks of a fixed-dose combination tablet containing sofosbuvir 400 mg and ledipasvir 90 mg with or without additional ribavirin.
Disclosures: Gilead Sciences funded the trials. Dr. Afdhal has received research support, honoraria, or other fees in the past 12 months from Gilead Sciences, Abbott, BMS, Boehringer Ingelheim, Echosens, Janssen, GSK, Idenix, Ligan, Merck, Medgenics, Novartis, Quest, Springbank, TRIO Healthcare, and Vertex. Dr. Peck-Radosavljevic made no disclosures.
Obeticholic acid shows promise in primary biliary cirrhosis
LONDON – Obeticholic acid significantly reduced key liver enzyme and bilirubin levels in patients with primary biliary cirrhosis in a phase III trial.
Results of POISE [PBC OCA International Study of Efficacy] showed that at the final 1-year assessment, 46% of patients treated with an initial 5-mg daily dose of OCA, and 47% of those treated with a 10-mg daily dose, achieved the primary endpoint of an alkaline phosphatase (ALP) level less than 1.67 times the upper limit of normal (ULN) with bilirubin levels less than the ULN and a 15% or greater reduction ALP. In comparison, just 10% of placebo-treated patients achieved this endpoint, which has been linked to a reduction in the need for liver transplantation and death.
Significant reductions (P less than .001) in levels of other key liver enzymes – gamma glutamyl transferase, alanine aminotransferase, and aspartate aminotransferase – were also seen with active versus placebo treatment.
Study investigator Frederik Nevens said in an interview that OCA represents the first new treatment for PBC in 2 decades and it could even be "aspirin for hepatologists" if data from translational research are borne out in clinical trials.
Indeed, Dr. Nevens, of University Hospital Gasthuisberg in Leuven, Belgium, said that because OCA targets the farnesoid-X receptor, which is a nuclear receptor involved in multiple metabolic and other pathways, it could have widespread clinical applications.
"It has an effect on fibrosis, probably, it has an effect on portal hypertension, and it has an effect on cholestasis," he said. Furthermore, it reduces bacterial translocation from the gut, which has implications for the prevention of liver infections, and there is also the suggestion that it might be of benefit in patients with diabetes, Crohn’s disease, and malignancies.
"Certainly it also has a beneficial effect on NASH [nonalcoholic steatohepatitis], and NASH is one of the most common [liver] problems these days," Dr. Nevens observed. Recently, the phase II FLINT trial conducted with OCA in patients with NASH was halted because of positive interim results.
PBC affects mostly women, at rate of about 1 in 1,000 women over 40 years of age in the United States, and is the fifth most common reason for liver transplantation in the country, Dr. Nevens said. Currently, ursodeoxycholic acid is the only approved treatment, although fewer than 50% of patients respond adequately to the drug.
The POISE data clearly have shown a potential benefit for OCA in patients with PBC, the researcher said at the meeting, sponsored by the European Association for the Study of the Liver. Data from a phase II trial had already shown that OCA with or without the standard treatment (ursodeoxycholic acid) at a dose of 10-50 mg produced significant improvement in cholestasis but the main side effect was pruritus. While itching is a characteristic of PBC itself, a key aim of the phase III trial was to reduce the prevalence of this side effect.
Significantly lowering and titrating the dose reduced the occurrence and impact of pruritus, Dr. Nevens reported during a late-breaking trials session. Nevertheless, the side effect was seen in more than two-thirds of patients given the 10-mg dose and 56% of those given the 5-mg dose, which was titrated up to 10 mg over a 6-month period, compared with 38% of placebo-treated patients. The bile acid sequestrant cholestyramine was permitted as a means to alleviate itching and was used in 26%, 19%, and 11% of patients, respectively.
"Titration from 5 to 10 mg based on clinical response improved tolerance, minimized dropouts due to pruritus, and showed comparable efficacy to 10 mg OCA," Dr. Nevens said. "The long-term safety and efficacy is currently being evaluated in an open-label extension study," he added, noting almost all patients (95%) completing the phase III trial had opted to continue therapy in the extension study.
Further confirmation of OCA’s benefits are needed, but the problem with doing a trial to prove that mortality is reduced as a result of long-term treatment is that it would not be ethical to treat patients with placebo for 10 years, Dr. Nevens pointed out. Reduction in ALP and bilirubin levels, as used in POISE, was a good surrogate marker for outcome, he said. The Global PBC Study Group found that ALP less than 1.67 times the ULN and normal predicted the likelihood of transplant-free survival with a hazard ratio of 3.53.
Intercept Pharmaceuticals sponsored the study. Dr. Nevens was an investigator for the POISE trial but did not receive any grants or speaker fees from the company.
LONDON – Obeticholic acid significantly reduced key liver enzyme and bilirubin levels in patients with primary biliary cirrhosis in a phase III trial.
Results of POISE [PBC OCA International Study of Efficacy] showed that at the final 1-year assessment, 46% of patients treated with an initial 5-mg daily dose of OCA, and 47% of those treated with a 10-mg daily dose, achieved the primary endpoint of an alkaline phosphatase (ALP) level less than 1.67 times the upper limit of normal (ULN) with bilirubin levels less than the ULN and a 15% or greater reduction ALP. In comparison, just 10% of placebo-treated patients achieved this endpoint, which has been linked to a reduction in the need for liver transplantation and death.
Significant reductions (P less than .001) in levels of other key liver enzymes – gamma glutamyl transferase, alanine aminotransferase, and aspartate aminotransferase – were also seen with active versus placebo treatment.
Study investigator Frederik Nevens said in an interview that OCA represents the first new treatment for PBC in 2 decades and it could even be "aspirin for hepatologists" if data from translational research are borne out in clinical trials.
Indeed, Dr. Nevens, of University Hospital Gasthuisberg in Leuven, Belgium, said that because OCA targets the farnesoid-X receptor, which is a nuclear receptor involved in multiple metabolic and other pathways, it could have widespread clinical applications.
"It has an effect on fibrosis, probably, it has an effect on portal hypertension, and it has an effect on cholestasis," he said. Furthermore, it reduces bacterial translocation from the gut, which has implications for the prevention of liver infections, and there is also the suggestion that it might be of benefit in patients with diabetes, Crohn’s disease, and malignancies.
"Certainly it also has a beneficial effect on NASH [nonalcoholic steatohepatitis], and NASH is one of the most common [liver] problems these days," Dr. Nevens observed. Recently, the phase II FLINT trial conducted with OCA in patients with NASH was halted because of positive interim results.
PBC affects mostly women, at rate of about 1 in 1,000 women over 40 years of age in the United States, and is the fifth most common reason for liver transplantation in the country, Dr. Nevens said. Currently, ursodeoxycholic acid is the only approved treatment, although fewer than 50% of patients respond adequately to the drug.
The POISE data clearly have shown a potential benefit for OCA in patients with PBC, the researcher said at the meeting, sponsored by the European Association for the Study of the Liver. Data from a phase II trial had already shown that OCA with or without the standard treatment (ursodeoxycholic acid) at a dose of 10-50 mg produced significant improvement in cholestasis but the main side effect was pruritus. While itching is a characteristic of PBC itself, a key aim of the phase III trial was to reduce the prevalence of this side effect.
Significantly lowering and titrating the dose reduced the occurrence and impact of pruritus, Dr. Nevens reported during a late-breaking trials session. Nevertheless, the side effect was seen in more than two-thirds of patients given the 10-mg dose and 56% of those given the 5-mg dose, which was titrated up to 10 mg over a 6-month period, compared with 38% of placebo-treated patients. The bile acid sequestrant cholestyramine was permitted as a means to alleviate itching and was used in 26%, 19%, and 11% of patients, respectively.
"Titration from 5 to 10 mg based on clinical response improved tolerance, minimized dropouts due to pruritus, and showed comparable efficacy to 10 mg OCA," Dr. Nevens said. "The long-term safety and efficacy is currently being evaluated in an open-label extension study," he added, noting almost all patients (95%) completing the phase III trial had opted to continue therapy in the extension study.
Further confirmation of OCA’s benefits are needed, but the problem with doing a trial to prove that mortality is reduced as a result of long-term treatment is that it would not be ethical to treat patients with placebo for 10 years, Dr. Nevens pointed out. Reduction in ALP and bilirubin levels, as used in POISE, was a good surrogate marker for outcome, he said. The Global PBC Study Group found that ALP less than 1.67 times the ULN and normal predicted the likelihood of transplant-free survival with a hazard ratio of 3.53.
Intercept Pharmaceuticals sponsored the study. Dr. Nevens was an investigator for the POISE trial but did not receive any grants or speaker fees from the company.
LONDON – Obeticholic acid significantly reduced key liver enzyme and bilirubin levels in patients with primary biliary cirrhosis in a phase III trial.
Results of POISE [PBC OCA International Study of Efficacy] showed that at the final 1-year assessment, 46% of patients treated with an initial 5-mg daily dose of OCA, and 47% of those treated with a 10-mg daily dose, achieved the primary endpoint of an alkaline phosphatase (ALP) level less than 1.67 times the upper limit of normal (ULN) with bilirubin levels less than the ULN and a 15% or greater reduction ALP. In comparison, just 10% of placebo-treated patients achieved this endpoint, which has been linked to a reduction in the need for liver transplantation and death.
Significant reductions (P less than .001) in levels of other key liver enzymes – gamma glutamyl transferase, alanine aminotransferase, and aspartate aminotransferase – were also seen with active versus placebo treatment.
Study investigator Frederik Nevens said in an interview that OCA represents the first new treatment for PBC in 2 decades and it could even be "aspirin for hepatologists" if data from translational research are borne out in clinical trials.
Indeed, Dr. Nevens, of University Hospital Gasthuisberg in Leuven, Belgium, said that because OCA targets the farnesoid-X receptor, which is a nuclear receptor involved in multiple metabolic and other pathways, it could have widespread clinical applications.
"It has an effect on fibrosis, probably, it has an effect on portal hypertension, and it has an effect on cholestasis," he said. Furthermore, it reduces bacterial translocation from the gut, which has implications for the prevention of liver infections, and there is also the suggestion that it might be of benefit in patients with diabetes, Crohn’s disease, and malignancies.
"Certainly it also has a beneficial effect on NASH [nonalcoholic steatohepatitis], and NASH is one of the most common [liver] problems these days," Dr. Nevens observed. Recently, the phase II FLINT trial conducted with OCA in patients with NASH was halted because of positive interim results.
PBC affects mostly women, at rate of about 1 in 1,000 women over 40 years of age in the United States, and is the fifth most common reason for liver transplantation in the country, Dr. Nevens said. Currently, ursodeoxycholic acid is the only approved treatment, although fewer than 50% of patients respond adequately to the drug.
The POISE data clearly have shown a potential benefit for OCA in patients with PBC, the researcher said at the meeting, sponsored by the European Association for the Study of the Liver. Data from a phase II trial had already shown that OCA with or without the standard treatment (ursodeoxycholic acid) at a dose of 10-50 mg produced significant improvement in cholestasis but the main side effect was pruritus. While itching is a characteristic of PBC itself, a key aim of the phase III trial was to reduce the prevalence of this side effect.
Significantly lowering and titrating the dose reduced the occurrence and impact of pruritus, Dr. Nevens reported during a late-breaking trials session. Nevertheless, the side effect was seen in more than two-thirds of patients given the 10-mg dose and 56% of those given the 5-mg dose, which was titrated up to 10 mg over a 6-month period, compared with 38% of placebo-treated patients. The bile acid sequestrant cholestyramine was permitted as a means to alleviate itching and was used in 26%, 19%, and 11% of patients, respectively.
"Titration from 5 to 10 mg based on clinical response improved tolerance, minimized dropouts due to pruritus, and showed comparable efficacy to 10 mg OCA," Dr. Nevens said. "The long-term safety and efficacy is currently being evaluated in an open-label extension study," he added, noting almost all patients (95%) completing the phase III trial had opted to continue therapy in the extension study.
Further confirmation of OCA’s benefits are needed, but the problem with doing a trial to prove that mortality is reduced as a result of long-term treatment is that it would not be ethical to treat patients with placebo for 10 years, Dr. Nevens pointed out. Reduction in ALP and bilirubin levels, as used in POISE, was a good surrogate marker for outcome, he said. The Global PBC Study Group found that ALP less than 1.67 times the ULN and normal predicted the likelihood of transplant-free survival with a hazard ratio of 3.53.
Intercept Pharmaceuticals sponsored the study. Dr. Nevens was an investigator for the POISE trial but did not receive any grants or speaker fees from the company.
AT THE INTERNATIONAL LIVER CONGRESS 2014
Major finding: The combined primary endpoint of reduction in ALP and bilirubin was achieved by 46%-47% of obeticholic acid (OCA)- and 10% of placebo-treated patients (P less than .0001).
Data source: Phase III, double blind, placebo-controlled trial of OCA in 216 patients with primary biliary cirrhosis.
Disclosures: Intercept Pharmaceuticals sponsored the study. Dr. Nevens was an investigator for the POISE trial but did not receive any grants or speaker fees from the company.