User login
Aetna customers to receive discount on Gilead’s hepatitis C treatment
Gilead Sciences Inc. and Aetna Inc. have announced a partnership wherein the drug company will offer Aetna’s approximately 20 million health plan members a discounted rate on its hepatitis C drugs, Sovaldi (sofosbuvir) and Harvoni (ledipasvir and sofosbuvir).
The amount of the discount has not yet been announced, although it should be significantly less than the current rate: Sovaldi costs $84,000 for a 12-week course of treatment, and Harvoni costs $94,500 for 12 weeks.
For more information, go to www.reuters.com.
Gilead Sciences Inc. and Aetna Inc. have announced a partnership wherein the drug company will offer Aetna’s approximately 20 million health plan members a discounted rate on its hepatitis C drugs, Sovaldi (sofosbuvir) and Harvoni (ledipasvir and sofosbuvir).
The amount of the discount has not yet been announced, although it should be significantly less than the current rate: Sovaldi costs $84,000 for a 12-week course of treatment, and Harvoni costs $94,500 for 12 weeks.
For more information, go to www.reuters.com.
Gilead Sciences Inc. and Aetna Inc. have announced a partnership wherein the drug company will offer Aetna’s approximately 20 million health plan members a discounted rate on its hepatitis C drugs, Sovaldi (sofosbuvir) and Harvoni (ledipasvir and sofosbuvir).
The amount of the discount has not yet been announced, although it should be significantly less than the current rate: Sovaldi costs $84,000 for a 12-week course of treatment, and Harvoni costs $94,500 for 12 weeks.
For more information, go to www.reuters.com.
Transsplenic TIPS procedure catching on for portal vein thrombosis
CHICAGO – Portal vein recanalization using a transsplenic approach can be utilized to improve transplant candidacy in patients with cirrhosis and chronic portal vein thrombosis, according to Dr. Bartley Thornburg.
“It’s a safe and effective procedure that allows for end-to-end anastomoses at transplant in patients who otherwise would not be able to have them or would require thrombectomy without transplant, and we know that end-to-end anastomoses are associated with decreased morbidity and mortality,” said Dr. Thornburg of Northwestern University Medical Center in Chicago.
Historically, and at his institution, portal vein thrombosis (PVT) is a relative contraindication to liver transplant. The American Association for the Study of Liver Diseases also recommends that transjugular intrahepatic portosystemic shunt (TIPS) placement be avoided in patients with a Model for End-State Liver Disease (MELD) score >18.
In 2013, colleague Dr. Riad Salem demonstrated the efficacy of portal vein recanalization during TIPS using a transhepatic approach in 44 patients, with only one technical failure and three cases of rethrombosis. Among six patients with a baseline MELD score >18, four went on to successful liver transplant, one is awaiting transplant, and one died as a result of bleeding 45 days post procedure despite an improvement in MELD score.
The last three patients in the series underwent TIPS with a transsplenic approach, and not only were the results equally good, but the approach was technically easier, Dr. Thornburg said at a symposium on vascular surgery sponsored by Northwestern University.
Since then, this increasingly common alternative approach has been assessed in another 11 consecutive patients with cirrhosis, portal hypertension, and chronic PVT. All patients had been denied listing for transplant because of their PVT, and four had a baseline MELD score >18.
At the end of the procedure, thrombus persisted in 45% (5 of 11 patients). On a 1-month follow-up venogram, however, three of the five patients had complete resolution of the thrombus without any added anticoagulation, one had persistent partial thrombus that was smaller than at stent placement, and one went on to transplant, Dr. Thornburg said.
All six of the patients with portal vein (PV) patency post procedure have retained patency after a median follow-up of 6.4 months.
“What we’ve learned from doing these cases is that complete elimination of the portal vein thrombus at the time of TIPS placement isn’t necessary,” he said. “It would be easy to get carried away and do suction or AngioJet [mechanical thrombectomy], but what we’ve found is that because of how much flow there is in the portal vein once it’s recanalized and the TIPS is in place, basically establishing a flow allowed for clot clearance by 1 month in all patients, except one.”
The procedure starts like a typical TIPS, with access achieved by advancing a 21-guage needle into the peripheral splenic vein or hilum under ultrasound guidance. A 5-French sheath is then placed through the parenchyma to the origin of splenic vein or the clot and an intrahepatic venogram performed to confirm occlusion, Dr. Thornburg said.
A 5-French Kumpe catheter and glide wire are used to recanalize the thrombosed portal vein, with a 10-mm gooseneck snare placed through the Kumpe in the peripheral portal vein as a target for the TIPS needle.
“Then, we basically get through-and-through access from the IJ [internal jugular] through this splenic access, and that gives us the workability to get our sheath across the portal vein and place our TIPS,” he said.
The remainder of the procedure is similar to that of the transhepatic approach. Angioplasty of the thrombosed PV is performed with an 8-by-40-mm balloon, followed by deployment of a Viatorr stent graft. The stent and PV are dilated with a 10-by-40-mm balloon and the splenic tract embolized with a couple of 4-by-14-cm Nester coils.
Based on their experience, short TIPS are always placed to maximize the amount of portal vein that is available at transplant for the end-to-end anastomoses, Dr. Thornburg said.
All patients who went on to transplant have received end-to-end anastomoses on what transplant surgeons have described as “totally normal” walled portal veins, including one patient who underwent transplant just 1 week post TIPS, he added.
There have been no major bleeding events with the transsplenic approach and only two adverse events: one case of transient encephalopathy and one low-grade fever.
Dr. Thornburg and Dr. Salem reported having no relevant financial disclosures.
CHICAGO – Portal vein recanalization using a transsplenic approach can be utilized to improve transplant candidacy in patients with cirrhosis and chronic portal vein thrombosis, according to Dr. Bartley Thornburg.
“It’s a safe and effective procedure that allows for end-to-end anastomoses at transplant in patients who otherwise would not be able to have them or would require thrombectomy without transplant, and we know that end-to-end anastomoses are associated with decreased morbidity and mortality,” said Dr. Thornburg of Northwestern University Medical Center in Chicago.
Historically, and at his institution, portal vein thrombosis (PVT) is a relative contraindication to liver transplant. The American Association for the Study of Liver Diseases also recommends that transjugular intrahepatic portosystemic shunt (TIPS) placement be avoided in patients with a Model for End-State Liver Disease (MELD) score >18.
In 2013, colleague Dr. Riad Salem demonstrated the efficacy of portal vein recanalization during TIPS using a transhepatic approach in 44 patients, with only one technical failure and three cases of rethrombosis. Among six patients with a baseline MELD score >18, four went on to successful liver transplant, one is awaiting transplant, and one died as a result of bleeding 45 days post procedure despite an improvement in MELD score.
The last three patients in the series underwent TIPS with a transsplenic approach, and not only were the results equally good, but the approach was technically easier, Dr. Thornburg said at a symposium on vascular surgery sponsored by Northwestern University.
Since then, this increasingly common alternative approach has been assessed in another 11 consecutive patients with cirrhosis, portal hypertension, and chronic PVT. All patients had been denied listing for transplant because of their PVT, and four had a baseline MELD score >18.
At the end of the procedure, thrombus persisted in 45% (5 of 11 patients). On a 1-month follow-up venogram, however, three of the five patients had complete resolution of the thrombus without any added anticoagulation, one had persistent partial thrombus that was smaller than at stent placement, and one went on to transplant, Dr. Thornburg said.
All six of the patients with portal vein (PV) patency post procedure have retained patency after a median follow-up of 6.4 months.
“What we’ve learned from doing these cases is that complete elimination of the portal vein thrombus at the time of TIPS placement isn’t necessary,” he said. “It would be easy to get carried away and do suction or AngioJet [mechanical thrombectomy], but what we’ve found is that because of how much flow there is in the portal vein once it’s recanalized and the TIPS is in place, basically establishing a flow allowed for clot clearance by 1 month in all patients, except one.”
The procedure starts like a typical TIPS, with access achieved by advancing a 21-guage needle into the peripheral splenic vein or hilum under ultrasound guidance. A 5-French sheath is then placed through the parenchyma to the origin of splenic vein or the clot and an intrahepatic venogram performed to confirm occlusion, Dr. Thornburg said.
A 5-French Kumpe catheter and glide wire are used to recanalize the thrombosed portal vein, with a 10-mm gooseneck snare placed through the Kumpe in the peripheral portal vein as a target for the TIPS needle.
“Then, we basically get through-and-through access from the IJ [internal jugular] through this splenic access, and that gives us the workability to get our sheath across the portal vein and place our TIPS,” he said.
The remainder of the procedure is similar to that of the transhepatic approach. Angioplasty of the thrombosed PV is performed with an 8-by-40-mm balloon, followed by deployment of a Viatorr stent graft. The stent and PV are dilated with a 10-by-40-mm balloon and the splenic tract embolized with a couple of 4-by-14-cm Nester coils.
Based on their experience, short TIPS are always placed to maximize the amount of portal vein that is available at transplant for the end-to-end anastomoses, Dr. Thornburg said.
All patients who went on to transplant have received end-to-end anastomoses on what transplant surgeons have described as “totally normal” walled portal veins, including one patient who underwent transplant just 1 week post TIPS, he added.
There have been no major bleeding events with the transsplenic approach and only two adverse events: one case of transient encephalopathy and one low-grade fever.
Dr. Thornburg and Dr. Salem reported having no relevant financial disclosures.
CHICAGO – Portal vein recanalization using a transsplenic approach can be utilized to improve transplant candidacy in patients with cirrhosis and chronic portal vein thrombosis, according to Dr. Bartley Thornburg.
“It’s a safe and effective procedure that allows for end-to-end anastomoses at transplant in patients who otherwise would not be able to have them or would require thrombectomy without transplant, and we know that end-to-end anastomoses are associated with decreased morbidity and mortality,” said Dr. Thornburg of Northwestern University Medical Center in Chicago.
Historically, and at his institution, portal vein thrombosis (PVT) is a relative contraindication to liver transplant. The American Association for the Study of Liver Diseases also recommends that transjugular intrahepatic portosystemic shunt (TIPS) placement be avoided in patients with a Model for End-State Liver Disease (MELD) score >18.
In 2013, colleague Dr. Riad Salem demonstrated the efficacy of portal vein recanalization during TIPS using a transhepatic approach in 44 patients, with only one technical failure and three cases of rethrombosis. Among six patients with a baseline MELD score >18, four went on to successful liver transplant, one is awaiting transplant, and one died as a result of bleeding 45 days post procedure despite an improvement in MELD score.
The last three patients in the series underwent TIPS with a transsplenic approach, and not only were the results equally good, but the approach was technically easier, Dr. Thornburg said at a symposium on vascular surgery sponsored by Northwestern University.
Since then, this increasingly common alternative approach has been assessed in another 11 consecutive patients with cirrhosis, portal hypertension, and chronic PVT. All patients had been denied listing for transplant because of their PVT, and four had a baseline MELD score >18.
At the end of the procedure, thrombus persisted in 45% (5 of 11 patients). On a 1-month follow-up venogram, however, three of the five patients had complete resolution of the thrombus without any added anticoagulation, one had persistent partial thrombus that was smaller than at stent placement, and one went on to transplant, Dr. Thornburg said.
All six of the patients with portal vein (PV) patency post procedure have retained patency after a median follow-up of 6.4 months.
“What we’ve learned from doing these cases is that complete elimination of the portal vein thrombus at the time of TIPS placement isn’t necessary,” he said. “It would be easy to get carried away and do suction or AngioJet [mechanical thrombectomy], but what we’ve found is that because of how much flow there is in the portal vein once it’s recanalized and the TIPS is in place, basically establishing a flow allowed for clot clearance by 1 month in all patients, except one.”
The procedure starts like a typical TIPS, with access achieved by advancing a 21-guage needle into the peripheral splenic vein or hilum under ultrasound guidance. A 5-French sheath is then placed through the parenchyma to the origin of splenic vein or the clot and an intrahepatic venogram performed to confirm occlusion, Dr. Thornburg said.
A 5-French Kumpe catheter and glide wire are used to recanalize the thrombosed portal vein, with a 10-mm gooseneck snare placed through the Kumpe in the peripheral portal vein as a target for the TIPS needle.
“Then, we basically get through-and-through access from the IJ [internal jugular] through this splenic access, and that gives us the workability to get our sheath across the portal vein and place our TIPS,” he said.
The remainder of the procedure is similar to that of the transhepatic approach. Angioplasty of the thrombosed PV is performed with an 8-by-40-mm balloon, followed by deployment of a Viatorr stent graft. The stent and PV are dilated with a 10-by-40-mm balloon and the splenic tract embolized with a couple of 4-by-14-cm Nester coils.
Based on their experience, short TIPS are always placed to maximize the amount of portal vein that is available at transplant for the end-to-end anastomoses, Dr. Thornburg said.
All patients who went on to transplant have received end-to-end anastomoses on what transplant surgeons have described as “totally normal” walled portal veins, including one patient who underwent transplant just 1 week post TIPS, he added.
There have been no major bleeding events with the transsplenic approach and only two adverse events: one case of transient encephalopathy and one low-grade fever.
Dr. Thornburg and Dr. Salem reported having no relevant financial disclosures.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Analysis: Push for expanded hepatitis C screening appears premature
The recent advent of new treatments for hepatitis C prompted organizations including the Centers for Disease Control and Prevention, the U.S. Preventive Services Task Force, and the World Health Organization to recommend expanded hepatitis C screening, but such screening may be premature, according to a subject analysis.
Too much uncertainty exists regarding the validity of surrogate markers for treatment efficacy that were used in trials, and evidence regarding clinical outcomes and screening strategies is lacking, according to Dr. Ronald L. Koretz of the University of California, Los Angeles, and his colleagues, who evaluated the current understanding of the incidence and natural course of hepatitis C infection, treatment efficacy, and potential harms of treatment for their analysis.
The best available data suggest that 80%-85% of patients with chronic hepatitis C will die of nonhepatic causes; thus screening could lead to unnecessary treatment. This is important, as safety data for newer drugs are limited, and the existing data suggest a small but concerning rate of serious adverse events associated with the use of some treatments and treatment regimens; the risk-benefit profile of treatment cannot be adequately evaluated because of the lack of data regarding treatment benefits, the investigators reported online Jan. 13 in the British Medical Journal ([doi:10.10036/bmj.g7809]).
Clinical trials to determine the outcomes of treatment in screen-detected patients, as well as the long-term hazards of treatment, are needed, they said, noting that currently available trials included small numbers of patients and/or only short-term follow-up. Until data from such trials are available, physicians should not be pressured to enforce recommended screening strategies “out of enthusiasm for new treatments that have not yet been shown to cause long-term clinical improvement,” they concluded.
Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other financial conflicts.
The authors question the merits of hepatitis C (HCV) screening despite the endorsement of the Centers for Disease Control and Prevention, U.S. Preventive Services Task Force, and World Health Organization. They suggest HCV does not lead to sufficient mortality, treatment may cause harm, sustained virologic response (SVR) does not represent a ‘cure’ and question if treating HCV truly reduces long term morbidity/mortality. While it is true the majority of patients with HCV will not develop cirrhosis or death attributable to their liver disease, the authors downplayed the morbidity and mortality related to HCV and the significant cost to the health care system as well (Ann. Intern. Med. 2012;156:271; Hepatology 2013;57:2164]. The authors also downplayed the importance of sustained viral response (SVR), suggesting that it does not represent a cure, yet neglect to mention several studies demonstrating late recurrence in only 1%-3% of patients who have achieved SVR. Furthermore, the authors describe the “harms of treatment,” yet they largely reference therapies that are no longer used and make minimal comment on the clear safety and efficacy of the current interferon-free regimens. Additionally, they report that while clearing HCV may reduce the risk of decompensated cirrhosis and hepatocellular carcinoma, it does not completely eliminate these risks and therefore treatment may not be of value. This brings us back to the motivation of the CDC, USPSTF, and WHO to recommend screening for HCV – identifying asymptomatic infection to allow for administration of safe, effective antiviral therapies before the development of cirrhosis and all of its complications.
Dr. Sean Koppe is director of hepatology, University of Illinois Hospital & Health Sciences System. He has no conflicts of interest.
The authors question the merits of hepatitis C (HCV) screening despite the endorsement of the Centers for Disease Control and Prevention, U.S. Preventive Services Task Force, and World Health Organization. They suggest HCV does not lead to sufficient mortality, treatment may cause harm, sustained virologic response (SVR) does not represent a ‘cure’ and question if treating HCV truly reduces long term morbidity/mortality. While it is true the majority of patients with HCV will not develop cirrhosis or death attributable to their liver disease, the authors downplayed the morbidity and mortality related to HCV and the significant cost to the health care system as well (Ann. Intern. Med. 2012;156:271; Hepatology 2013;57:2164]. The authors also downplayed the importance of sustained viral response (SVR), suggesting that it does not represent a cure, yet neglect to mention several studies demonstrating late recurrence in only 1%-3% of patients who have achieved SVR. Furthermore, the authors describe the “harms of treatment,” yet they largely reference therapies that are no longer used and make minimal comment on the clear safety and efficacy of the current interferon-free regimens. Additionally, they report that while clearing HCV may reduce the risk of decompensated cirrhosis and hepatocellular carcinoma, it does not completely eliminate these risks and therefore treatment may not be of value. This brings us back to the motivation of the CDC, USPSTF, and WHO to recommend screening for HCV – identifying asymptomatic infection to allow for administration of safe, effective antiviral therapies before the development of cirrhosis and all of its complications.
Dr. Sean Koppe is director of hepatology, University of Illinois Hospital & Health Sciences System. He has no conflicts of interest.
The authors question the merits of hepatitis C (HCV) screening despite the endorsement of the Centers for Disease Control and Prevention, U.S. Preventive Services Task Force, and World Health Organization. They suggest HCV does not lead to sufficient mortality, treatment may cause harm, sustained virologic response (SVR) does not represent a ‘cure’ and question if treating HCV truly reduces long term morbidity/mortality. While it is true the majority of patients with HCV will not develop cirrhosis or death attributable to their liver disease, the authors downplayed the morbidity and mortality related to HCV and the significant cost to the health care system as well (Ann. Intern. Med. 2012;156:271; Hepatology 2013;57:2164]. The authors also downplayed the importance of sustained viral response (SVR), suggesting that it does not represent a cure, yet neglect to mention several studies demonstrating late recurrence in only 1%-3% of patients who have achieved SVR. Furthermore, the authors describe the “harms of treatment,” yet they largely reference therapies that are no longer used and make minimal comment on the clear safety and efficacy of the current interferon-free regimens. Additionally, they report that while clearing HCV may reduce the risk of decompensated cirrhosis and hepatocellular carcinoma, it does not completely eliminate these risks and therefore treatment may not be of value. This brings us back to the motivation of the CDC, USPSTF, and WHO to recommend screening for HCV – identifying asymptomatic infection to allow for administration of safe, effective antiviral therapies before the development of cirrhosis and all of its complications.
Dr. Sean Koppe is director of hepatology, University of Illinois Hospital & Health Sciences System. He has no conflicts of interest.
The recent advent of new treatments for hepatitis C prompted organizations including the Centers for Disease Control and Prevention, the U.S. Preventive Services Task Force, and the World Health Organization to recommend expanded hepatitis C screening, but such screening may be premature, according to a subject analysis.
Too much uncertainty exists regarding the validity of surrogate markers for treatment efficacy that were used in trials, and evidence regarding clinical outcomes and screening strategies is lacking, according to Dr. Ronald L. Koretz of the University of California, Los Angeles, and his colleagues, who evaluated the current understanding of the incidence and natural course of hepatitis C infection, treatment efficacy, and potential harms of treatment for their analysis.
The best available data suggest that 80%-85% of patients with chronic hepatitis C will die of nonhepatic causes; thus screening could lead to unnecessary treatment. This is important, as safety data for newer drugs are limited, and the existing data suggest a small but concerning rate of serious adverse events associated with the use of some treatments and treatment regimens; the risk-benefit profile of treatment cannot be adequately evaluated because of the lack of data regarding treatment benefits, the investigators reported online Jan. 13 in the British Medical Journal ([doi:10.10036/bmj.g7809]).
Clinical trials to determine the outcomes of treatment in screen-detected patients, as well as the long-term hazards of treatment, are needed, they said, noting that currently available trials included small numbers of patients and/or only short-term follow-up. Until data from such trials are available, physicians should not be pressured to enforce recommended screening strategies “out of enthusiasm for new treatments that have not yet been shown to cause long-term clinical improvement,” they concluded.
Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other financial conflicts.
The recent advent of new treatments for hepatitis C prompted organizations including the Centers for Disease Control and Prevention, the U.S. Preventive Services Task Force, and the World Health Organization to recommend expanded hepatitis C screening, but such screening may be premature, according to a subject analysis.
Too much uncertainty exists regarding the validity of surrogate markers for treatment efficacy that were used in trials, and evidence regarding clinical outcomes and screening strategies is lacking, according to Dr. Ronald L. Koretz of the University of California, Los Angeles, and his colleagues, who evaluated the current understanding of the incidence and natural course of hepatitis C infection, treatment efficacy, and potential harms of treatment for their analysis.
The best available data suggest that 80%-85% of patients with chronic hepatitis C will die of nonhepatic causes; thus screening could lead to unnecessary treatment. This is important, as safety data for newer drugs are limited, and the existing data suggest a small but concerning rate of serious adverse events associated with the use of some treatments and treatment regimens; the risk-benefit profile of treatment cannot be adequately evaluated because of the lack of data regarding treatment benefits, the investigators reported online Jan. 13 in the British Medical Journal ([doi:10.10036/bmj.g7809]).
Clinical trials to determine the outcomes of treatment in screen-detected patients, as well as the long-term hazards of treatment, are needed, they said, noting that currently available trials included small numbers of patients and/or only short-term follow-up. Until data from such trials are available, physicians should not be pressured to enforce recommended screening strategies “out of enthusiasm for new treatments that have not yet been shown to cause long-term clinical improvement,” they concluded.
Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other financial conflicts.
Key clinical point: Evidence to support expanded hepatitis C screening is lacking.
Major finding: An estimated 80%-85% of patients with chronic hepatitis C will die from nonhepatic causes.
Data source: An analysis of existing evidence.
Disclosures: Dr. Koretz is a member of the editorial board of the Cochrane Hepato-Biliary Group. The authors reported having no other relevant financial disclosures.
Best HCV value? Screen all baby boomers, treat all infections
BOSTON – Screening all adults born between 1945 and 1965 for hepatitis C and then treating all infected patients with oral drug regimens is the most cost effective strategy for society, because better outcomes more than offset the higher costs of wider screening and newer drugs, Dr. Zobair Younossi said.
A computer simulation analysis that compared four strategies for screening and treatment (plus the option of no screening or treatment) found that birth cohort screening and treatment of all hepatitis C virus–positive patients would save more than 4 million life-years at an incremental cost of $36,585 per quality-adjusted life-year (QALY), he reported at the annual meeting of the American Association for the Study of Liver Diseases.
That strategy produced an incremental cost-effectiveness ratio well below the widely accepted threshold of $50,000 per QALY used to define cost effectiveness in health care, said Dr. Younossi, professor of medicine at Virginia Commonwealth University’s Inova Campus and chair of Liver Disease Services for Inova, Falls Church, Va.
In first-generation treatment with direct-acting antivirals – namely, triple therapy with protease inhibitors – the mean cost to achieve a sustained virologic response was $172,889, he said, “and we did not get a lot of pushback from the payers at that point.”
Risk-based screening for HCV has been recommended, and the Centers for Disease Control and Prevention recently recommended birth cohort screening of people born between 1945 and 1965, the so-called baby boomers, Dr. Younossi said.
Still, studies suggest that less than 10% of patients with chronic HCV have been treated successfully, because of the failure of risk-based screening to identify all infected patients and the low efficacy and high rate of side effects from regimens based on interferon and ribavirin, he said.
Higher costs, lower risks
Dr. Younossi’s study used computer simulations to analyze the economic impact of various screening strategies followed by treatment with oral anti-HCV regimens.
The four strategies involved either risk-based screening or birth cohort screening, followed by treatment that either gave all HCV-infected patients oral regimens or that based treatment on staging, giving oral medications only to patients with significant fibrosis. The investigators also considered a fifth strategy: no screening and no treatment.
Birth cohort screening would lead to 1,162,323 patients being diagnosed with previously unknown chronic HCV, they estimated.
To estimate treatment costs, the investigators began with risk-based screening probabilities from a previous study: They assumed that 98% of patients would be medically eligible for treatment and have no contraindications to all-oral treatment regimens, and 98% of those treated would achieve a sustained virologic response for 12 weeks (SVR12), based on results of published trials.
The cost of oral direct-acting antiviral therapy was based on the cost of sofosbuvir – around $1,000 per day for 12 weeks, Dr. Younossi said. Costs for testing, staging, monitoring, and other data were taken from previous treatment models. The investigators calculated QALY from patients’ health utility reports in clinical trials.
Costs averaged approximately $88,000 per patient with birth cohort screening and treating all positive patients, $72,000 per patient with birth cohort screening and treatment based on staging, $60,000-$61,000 per patient for the risk-based screening strategies, and $53,000 per patient with no screening and no treatment.
The probabilities of cirrhosis, hepatocellular carcinoma, or liver transplant were much lower with either birth cohort screening strategy, however, compared with the risk-based strategies or no screening. The probability of cirrhosis was approximately 1% with birth cohort screening and treating all positive patients, 7% with birth cohort screening and treatment based on staging, 55%-60% with the risk-based strategies, and 67% with no screening.
The risk of hepatocellular carcinoma was 4% with birth cohort screening and treating all positive patients, 5% with birth cohort screening and treatment based on staging, 15% with either risk-based screening strategy, and 20% with no screening. The risk of liver transplant was 0.6% with either birth cohort screening strategy, 3% with either risk-based screening strategy, and nearly 4% with no screening.
A total of 4% of patients would be expected to die of liver-related causes after birth cohort screening and treating all positive patients, compared with 7% liver-related mortality after birth cohort screening and staging-based treatment, 25% liver-related mortality in either of the risk-based screening groups, and 34% liver-related mortality with no screening.
HCV costlier than other conditions?
When considering governmental health policy and budgetary issues, Dr. Younossi explained, the cost of curing HCV is not very different from lifetime treatment costs for type 2 diabetes, rheumatoid arthritis, or breast cancer with metastases – and it’s significantly lower than lifetime treatment costs for HIV or relapsing-remitting multiple sclerosis.
As advocates for patients, physicians need to lobby officials so that “as they are dividing the pie, hepatology and HCV are represented,” he said. “These regimens are cost effective.”
The study was an underestimation of savings from birth cohort screening and treating all HCV positives, Dr. Younossi noted, because it did not incorporate an estimated $3 billion per year in savings from work productivity in the United States by curing HCV.
A physician in the audience countered Dr. Younossi’s call for more enlightened health policy supporting funding for HCV treatment, noting that drug companies also need to be pressured to lower the price of the newer drugs.
“The biggest bridge we have to cross is the cost of the drugs,” he said. “This is like denying somebody treatment for tuberculosis by making the cost of the drug too high.”
“I agree,” Dr. Younossi said. “We have to focus on our colleagues in the industry. But we also have to focus on our colleagues in Congress and the policymakers to make sure that, even if we get help from the industry side, we also get help from the policymakers to provide us” with the funding for access to HCV screening and treatment.
Dr. Younossi reported having no financial disclosures. One of his coinvestigators reported financial associations with Gilead and Bristol-Myers Squibb.
On Twitter @sherryboschert
AGA RESOURCE
View the technical review, guideline and clinical decision support tool at www.gastro.org/guideline. Join the guideline authors for a webinar about the guidelines on Jan. 29, 2015, at noon ET. Register at http://ow.ly/FWcsx.
Chronic HCV infection is a common worldwide problem afflicting approximately 170 million people. HCV has significant morbidity and mortality, and it is currently the leading indication for liver transplantation in the Western world. Yet, the natural history is long, and most patients are asymptomatic and will not develop advanced liver disease.
The recent year has been marked by the stunning approval in the U.S. of all-oral, interferon-free treatments for chronic HCV, including genotypes 1, 2, and 3. Approved regimens are characterized by excellent tolerability and cure (sustained virologic response, SVR) rates for most patient subgroups above 90%. For genotype 1, the most common in the U.S., SVR rates in excess of 95% were observed in clinical trials.
Since the regimens are so well tolerated and effective, the number of patients seeking therapy has increased dramatically. Because the medical regimens are expensive, there have been draconian restrictions by many third-party payers for provision of antiviral therapy for HCV patients. This has led to analyses on cost-effectiveness of contemporary therapy.
The article describing a study presented at AASLD 2014 by Younossi et al. is an important contribution to this discussion. Younossi assessed a computer simulation analysis comparing four strategies for screening and treatment of HCV plus an option of no screening or treatment. He found that birth cohort screening and treatment of all HCV patients would save more than 4 million life-years at an incremental cost of $36,585 per quality-adjusted life-year (QALY); well below the accepted threshold used to define cost-effectiveness in health care. The assumptions regarding cost of the regimens, the rates of cirrhosis, hepatocellular carcinoma, liver transplantation and liver-related death were appropriate. The conclusion that all baby boomers should be screened and treated is not surprising.
The savings did not include an estimated $3 billion dollars annually in savings from work productivity. The analysis also did not account for the costs associated with many extrahepatic manifestations of HCV including diabetes mellitus, lymphoma, cryoglublinemia, chronic renal disease, and others that are not associated with the stage of liver disease and have significant morbidity and mortality. All-cause mortality, not only liver-related mortality, has been shown to decline in patients treated successfully compared with unsuccessful therapeutic attempts.
Never before has there been a serious medical illness for which a well-tolerated and highly effective therapy is available that has been subject to denial of therapy based upon cost by third-party payers. This is an unfortunate precedent.
All patients do not require therapy on an urgent basis. However, the decision to initiate therapy should be between a patient and his or her health care provider. Although it would be best if the cost of the medications could be reduced, even at the current costs, Younossi’s study provides clear data to support identification and treatment of HCV patients sooner rather than later.
Dr. Steven L. Flamm is chief of transplantation hepatology and professor of medicine and surgery at Northwestern University Feinberg School of Medicine, Chicago. He is a speaker for AbbVie, Janssen, and Gilead; and consults for AbbVie, Gilead, Janssen, BMS, and Merck.
Chronic HCV infection is a common worldwide problem afflicting approximately 170 million people. HCV has significant morbidity and mortality, and it is currently the leading indication for liver transplantation in the Western world. Yet, the natural history is long, and most patients are asymptomatic and will not develop advanced liver disease.
The recent year has been marked by the stunning approval in the U.S. of all-oral, interferon-free treatments for chronic HCV, including genotypes 1, 2, and 3. Approved regimens are characterized by excellent tolerability and cure (sustained virologic response, SVR) rates for most patient subgroups above 90%. For genotype 1, the most common in the U.S., SVR rates in excess of 95% were observed in clinical trials.
Since the regimens are so well tolerated and effective, the number of patients seeking therapy has increased dramatically. Because the medical regimens are expensive, there have been draconian restrictions by many third-party payers for provision of antiviral therapy for HCV patients. This has led to analyses on cost-effectiveness of contemporary therapy.
The article describing a study presented at AASLD 2014 by Younossi et al. is an important contribution to this discussion. Younossi assessed a computer simulation analysis comparing four strategies for screening and treatment of HCV plus an option of no screening or treatment. He found that birth cohort screening and treatment of all HCV patients would save more than 4 million life-years at an incremental cost of $36,585 per quality-adjusted life-year (QALY); well below the accepted threshold used to define cost-effectiveness in health care. The assumptions regarding cost of the regimens, the rates of cirrhosis, hepatocellular carcinoma, liver transplantation and liver-related death were appropriate. The conclusion that all baby boomers should be screened and treated is not surprising.
The savings did not include an estimated $3 billion dollars annually in savings from work productivity. The analysis also did not account for the costs associated with many extrahepatic manifestations of HCV including diabetes mellitus, lymphoma, cryoglublinemia, chronic renal disease, and others that are not associated with the stage of liver disease and have significant morbidity and mortality. All-cause mortality, not only liver-related mortality, has been shown to decline in patients treated successfully compared with unsuccessful therapeutic attempts.
Never before has there been a serious medical illness for which a well-tolerated and highly effective therapy is available that has been subject to denial of therapy based upon cost by third-party payers. This is an unfortunate precedent.
All patients do not require therapy on an urgent basis. However, the decision to initiate therapy should be between a patient and his or her health care provider. Although it would be best if the cost of the medications could be reduced, even at the current costs, Younossi’s study provides clear data to support identification and treatment of HCV patients sooner rather than later.
Dr. Steven L. Flamm is chief of transplantation hepatology and professor of medicine and surgery at Northwestern University Feinberg School of Medicine, Chicago. He is a speaker for AbbVie, Janssen, and Gilead; and consults for AbbVie, Gilead, Janssen, BMS, and Merck.
Chronic HCV infection is a common worldwide problem afflicting approximately 170 million people. HCV has significant morbidity and mortality, and it is currently the leading indication for liver transplantation in the Western world. Yet, the natural history is long, and most patients are asymptomatic and will not develop advanced liver disease.
The recent year has been marked by the stunning approval in the U.S. of all-oral, interferon-free treatments for chronic HCV, including genotypes 1, 2, and 3. Approved regimens are characterized by excellent tolerability and cure (sustained virologic response, SVR) rates for most patient subgroups above 90%. For genotype 1, the most common in the U.S., SVR rates in excess of 95% were observed in clinical trials.
Since the regimens are so well tolerated and effective, the number of patients seeking therapy has increased dramatically. Because the medical regimens are expensive, there have been draconian restrictions by many third-party payers for provision of antiviral therapy for HCV patients. This has led to analyses on cost-effectiveness of contemporary therapy.
The article describing a study presented at AASLD 2014 by Younossi et al. is an important contribution to this discussion. Younossi assessed a computer simulation analysis comparing four strategies for screening and treatment of HCV plus an option of no screening or treatment. He found that birth cohort screening and treatment of all HCV patients would save more than 4 million life-years at an incremental cost of $36,585 per quality-adjusted life-year (QALY); well below the accepted threshold used to define cost-effectiveness in health care. The assumptions regarding cost of the regimens, the rates of cirrhosis, hepatocellular carcinoma, liver transplantation and liver-related death were appropriate. The conclusion that all baby boomers should be screened and treated is not surprising.
The savings did not include an estimated $3 billion dollars annually in savings from work productivity. The analysis also did not account for the costs associated with many extrahepatic manifestations of HCV including diabetes mellitus, lymphoma, cryoglublinemia, chronic renal disease, and others that are not associated with the stage of liver disease and have significant morbidity and mortality. All-cause mortality, not only liver-related mortality, has been shown to decline in patients treated successfully compared with unsuccessful therapeutic attempts.
Never before has there been a serious medical illness for which a well-tolerated and highly effective therapy is available that has been subject to denial of therapy based upon cost by third-party payers. This is an unfortunate precedent.
All patients do not require therapy on an urgent basis. However, the decision to initiate therapy should be between a patient and his or her health care provider. Although it would be best if the cost of the medications could be reduced, even at the current costs, Younossi’s study provides clear data to support identification and treatment of HCV patients sooner rather than later.
Dr. Steven L. Flamm is chief of transplantation hepatology and professor of medicine and surgery at Northwestern University Feinberg School of Medicine, Chicago. He is a speaker for AbbVie, Janssen, and Gilead; and consults for AbbVie, Gilead, Janssen, BMS, and Merck.
BOSTON – Screening all adults born between 1945 and 1965 for hepatitis C and then treating all infected patients with oral drug regimens is the most cost effective strategy for society, because better outcomes more than offset the higher costs of wider screening and newer drugs, Dr. Zobair Younossi said.
A computer simulation analysis that compared four strategies for screening and treatment (plus the option of no screening or treatment) found that birth cohort screening and treatment of all hepatitis C virus–positive patients would save more than 4 million life-years at an incremental cost of $36,585 per quality-adjusted life-year (QALY), he reported at the annual meeting of the American Association for the Study of Liver Diseases.
That strategy produced an incremental cost-effectiveness ratio well below the widely accepted threshold of $50,000 per QALY used to define cost effectiveness in health care, said Dr. Younossi, professor of medicine at Virginia Commonwealth University’s Inova Campus and chair of Liver Disease Services for Inova, Falls Church, Va.
In first-generation treatment with direct-acting antivirals – namely, triple therapy with protease inhibitors – the mean cost to achieve a sustained virologic response was $172,889, he said, “and we did not get a lot of pushback from the payers at that point.”
Risk-based screening for HCV has been recommended, and the Centers for Disease Control and Prevention recently recommended birth cohort screening of people born between 1945 and 1965, the so-called baby boomers, Dr. Younossi said.
Still, studies suggest that less than 10% of patients with chronic HCV have been treated successfully, because of the failure of risk-based screening to identify all infected patients and the low efficacy and high rate of side effects from regimens based on interferon and ribavirin, he said.
Higher costs, lower risks
Dr. Younossi’s study used computer simulations to analyze the economic impact of various screening strategies followed by treatment with oral anti-HCV regimens.
The four strategies involved either risk-based screening or birth cohort screening, followed by treatment that either gave all HCV-infected patients oral regimens or that based treatment on staging, giving oral medications only to patients with significant fibrosis. The investigators also considered a fifth strategy: no screening and no treatment.
Birth cohort screening would lead to 1,162,323 patients being diagnosed with previously unknown chronic HCV, they estimated.
To estimate treatment costs, the investigators began with risk-based screening probabilities from a previous study: They assumed that 98% of patients would be medically eligible for treatment and have no contraindications to all-oral treatment regimens, and 98% of those treated would achieve a sustained virologic response for 12 weeks (SVR12), based on results of published trials.
The cost of oral direct-acting antiviral therapy was based on the cost of sofosbuvir – around $1,000 per day for 12 weeks, Dr. Younossi said. Costs for testing, staging, monitoring, and other data were taken from previous treatment models. The investigators calculated QALY from patients’ health utility reports in clinical trials.
Costs averaged approximately $88,000 per patient with birth cohort screening and treating all positive patients, $72,000 per patient with birth cohort screening and treatment based on staging, $60,000-$61,000 per patient for the risk-based screening strategies, and $53,000 per patient with no screening and no treatment.
The probabilities of cirrhosis, hepatocellular carcinoma, or liver transplant were much lower with either birth cohort screening strategy, however, compared with the risk-based strategies or no screening. The probability of cirrhosis was approximately 1% with birth cohort screening and treating all positive patients, 7% with birth cohort screening and treatment based on staging, 55%-60% with the risk-based strategies, and 67% with no screening.
The risk of hepatocellular carcinoma was 4% with birth cohort screening and treating all positive patients, 5% with birth cohort screening and treatment based on staging, 15% with either risk-based screening strategy, and 20% with no screening. The risk of liver transplant was 0.6% with either birth cohort screening strategy, 3% with either risk-based screening strategy, and nearly 4% with no screening.
A total of 4% of patients would be expected to die of liver-related causes after birth cohort screening and treating all positive patients, compared with 7% liver-related mortality after birth cohort screening and staging-based treatment, 25% liver-related mortality in either of the risk-based screening groups, and 34% liver-related mortality with no screening.
HCV costlier than other conditions?
When considering governmental health policy and budgetary issues, Dr. Younossi explained, the cost of curing HCV is not very different from lifetime treatment costs for type 2 diabetes, rheumatoid arthritis, or breast cancer with metastases – and it’s significantly lower than lifetime treatment costs for HIV or relapsing-remitting multiple sclerosis.
As advocates for patients, physicians need to lobby officials so that “as they are dividing the pie, hepatology and HCV are represented,” he said. “These regimens are cost effective.”
The study was an underestimation of savings from birth cohort screening and treating all HCV positives, Dr. Younossi noted, because it did not incorporate an estimated $3 billion per year in savings from work productivity in the United States by curing HCV.
A physician in the audience countered Dr. Younossi’s call for more enlightened health policy supporting funding for HCV treatment, noting that drug companies also need to be pressured to lower the price of the newer drugs.
“The biggest bridge we have to cross is the cost of the drugs,” he said. “This is like denying somebody treatment for tuberculosis by making the cost of the drug too high.”
“I agree,” Dr. Younossi said. “We have to focus on our colleagues in the industry. But we also have to focus on our colleagues in Congress and the policymakers to make sure that, even if we get help from the industry side, we also get help from the policymakers to provide us” with the funding for access to HCV screening and treatment.
Dr. Younossi reported having no financial disclosures. One of his coinvestigators reported financial associations with Gilead and Bristol-Myers Squibb.
On Twitter @sherryboschert
AGA RESOURCE
View the technical review, guideline and clinical decision support tool at www.gastro.org/guideline. Join the guideline authors for a webinar about the guidelines on Jan. 29, 2015, at noon ET. Register at http://ow.ly/FWcsx.
BOSTON – Screening all adults born between 1945 and 1965 for hepatitis C and then treating all infected patients with oral drug regimens is the most cost effective strategy for society, because better outcomes more than offset the higher costs of wider screening and newer drugs, Dr. Zobair Younossi said.
A computer simulation analysis that compared four strategies for screening and treatment (plus the option of no screening or treatment) found that birth cohort screening and treatment of all hepatitis C virus–positive patients would save more than 4 million life-years at an incremental cost of $36,585 per quality-adjusted life-year (QALY), he reported at the annual meeting of the American Association for the Study of Liver Diseases.
That strategy produced an incremental cost-effectiveness ratio well below the widely accepted threshold of $50,000 per QALY used to define cost effectiveness in health care, said Dr. Younossi, professor of medicine at Virginia Commonwealth University’s Inova Campus and chair of Liver Disease Services for Inova, Falls Church, Va.
In first-generation treatment with direct-acting antivirals – namely, triple therapy with protease inhibitors – the mean cost to achieve a sustained virologic response was $172,889, he said, “and we did not get a lot of pushback from the payers at that point.”
Risk-based screening for HCV has been recommended, and the Centers for Disease Control and Prevention recently recommended birth cohort screening of people born between 1945 and 1965, the so-called baby boomers, Dr. Younossi said.
Still, studies suggest that less than 10% of patients with chronic HCV have been treated successfully, because of the failure of risk-based screening to identify all infected patients and the low efficacy and high rate of side effects from regimens based on interferon and ribavirin, he said.
Higher costs, lower risks
Dr. Younossi’s study used computer simulations to analyze the economic impact of various screening strategies followed by treatment with oral anti-HCV regimens.
The four strategies involved either risk-based screening or birth cohort screening, followed by treatment that either gave all HCV-infected patients oral regimens or that based treatment on staging, giving oral medications only to patients with significant fibrosis. The investigators also considered a fifth strategy: no screening and no treatment.
Birth cohort screening would lead to 1,162,323 patients being diagnosed with previously unknown chronic HCV, they estimated.
To estimate treatment costs, the investigators began with risk-based screening probabilities from a previous study: They assumed that 98% of patients would be medically eligible for treatment and have no contraindications to all-oral treatment regimens, and 98% of those treated would achieve a sustained virologic response for 12 weeks (SVR12), based on results of published trials.
The cost of oral direct-acting antiviral therapy was based on the cost of sofosbuvir – around $1,000 per day for 12 weeks, Dr. Younossi said. Costs for testing, staging, monitoring, and other data were taken from previous treatment models. The investigators calculated QALY from patients’ health utility reports in clinical trials.
Costs averaged approximately $88,000 per patient with birth cohort screening and treating all positive patients, $72,000 per patient with birth cohort screening and treatment based on staging, $60,000-$61,000 per patient for the risk-based screening strategies, and $53,000 per patient with no screening and no treatment.
The probabilities of cirrhosis, hepatocellular carcinoma, or liver transplant were much lower with either birth cohort screening strategy, however, compared with the risk-based strategies or no screening. The probability of cirrhosis was approximately 1% with birth cohort screening and treating all positive patients, 7% with birth cohort screening and treatment based on staging, 55%-60% with the risk-based strategies, and 67% with no screening.
The risk of hepatocellular carcinoma was 4% with birth cohort screening and treating all positive patients, 5% with birth cohort screening and treatment based on staging, 15% with either risk-based screening strategy, and 20% with no screening. The risk of liver transplant was 0.6% with either birth cohort screening strategy, 3% with either risk-based screening strategy, and nearly 4% with no screening.
A total of 4% of patients would be expected to die of liver-related causes after birth cohort screening and treating all positive patients, compared with 7% liver-related mortality after birth cohort screening and staging-based treatment, 25% liver-related mortality in either of the risk-based screening groups, and 34% liver-related mortality with no screening.
HCV costlier than other conditions?
When considering governmental health policy and budgetary issues, Dr. Younossi explained, the cost of curing HCV is not very different from lifetime treatment costs for type 2 diabetes, rheumatoid arthritis, or breast cancer with metastases – and it’s significantly lower than lifetime treatment costs for HIV or relapsing-remitting multiple sclerosis.
As advocates for patients, physicians need to lobby officials so that “as they are dividing the pie, hepatology and HCV are represented,” he said. “These regimens are cost effective.”
The study was an underestimation of savings from birth cohort screening and treating all HCV positives, Dr. Younossi noted, because it did not incorporate an estimated $3 billion per year in savings from work productivity in the United States by curing HCV.
A physician in the audience countered Dr. Younossi’s call for more enlightened health policy supporting funding for HCV treatment, noting that drug companies also need to be pressured to lower the price of the newer drugs.
“The biggest bridge we have to cross is the cost of the drugs,” he said. “This is like denying somebody treatment for tuberculosis by making the cost of the drug too high.”
“I agree,” Dr. Younossi said. “We have to focus on our colleagues in the industry. But we also have to focus on our colleagues in Congress and the policymakers to make sure that, even if we get help from the industry side, we also get help from the policymakers to provide us” with the funding for access to HCV screening and treatment.
Dr. Younossi reported having no financial disclosures. One of his coinvestigators reported financial associations with Gilead and Bristol-Myers Squibb.
On Twitter @sherryboschert
AGA RESOURCE
View the technical review, guideline and clinical decision support tool at www.gastro.org/guideline. Join the guideline authors for a webinar about the guidelines on Jan. 29, 2015, at noon ET. Register at http://ow.ly/FWcsx.
AT THE LIVER MEETING 2014
Key clinical point: Screening adults born in 1945-1965 and treating all who have HCV with oral anti-HCV regimens is most cost effective.
Major finding: The strategy’s incremental cost of $36,585 is below the $50,000 per QALY threshold for cost effectiveness.
Data source: A computer simulation analysis that compared four strategies for screening and treatment, with treatment costs based on an estimated $1,000/day for sofosbuvir.
Disclosures: Dr. Younossi reported having no financial disclosures. One of his coinvestigators reported financial associations with Gilead, which markets sofosbuvir, and Bristol-Myers Squibb.
Sofosbuvir and ribavirin critical to preventing posttransplantation HCV recurrence
Sofosbuvir and ribavirin treatments should be administered to patients with hepatitis C virus who undergo liver transplantations in order to significantly decrease the risks of posttransplant HCV recurrence, according to two new studies published in the January issue of Gastroenterology (10.1053/j.gastro.2014.09.023 and 10.1053/j.gastro.2014.10.001).
“In clinical trials, administration of sofosbuvir with ribavirin was associated with rapid decreases of HCV RNA to undetectable levels in patients with HCV genotype 1, 2, 3, 4, and 6 infections,” wrote lead author Dr. Michael P. Curry of the Beth Israel Deaconess Medical Center in Boston, and his coauthors on the first of these two studies. “In more than 3,000 patients treated to date, sofosbuvir has been shown to be safe, viral breakthrough during treatment has been rare (and associated with nonadherence), and few drug interactions have been observed.”
In a phase II, open-label study, Dr. Curry and his coinvestigators enrolled 61 patients with HCV of any genotype, and cirrhosis with a Child-Turcotte-Pugh score no greater than 7, who were all wait-listed to receive liver transplantations. Subjects received up to 48 weeks of treatment with 400 mg of sofosbuvir, and a separate dose of ribavirin prior to liver transplantation, while 43 patients received transplantations alone. The primary outcome sought by investigators was HCV-RNA levels less than 25 IU/mL at 12 weeks after transplantation among patients that had this level prior to the operation.
The investigators found that 43 subjects had the desired HCV-RNA levels; of that population, 49% had a posttransplantation virologic response, with the most frequent side effects reported by subjects being fatigue (38%), headache (23%), and anemia (21%). Of the 43 applicable subjects, 30 (70% of the population) had a posttransplantation virologic response at 12 weeks, 10 (23%) had recurrent infection, and 3 (7%) died.
“This study provides proof of concept that virologic suppression without interferon significantly can reduce the rate of recurrent HCV after liver transplantation,” the study says, adding that the results “compare favorably with those observed in other trials of pretransplantation antiviral therapy.”
In the second study, the authors ascertained that combination therapy consisting of sofosbuvir and ribavirin for 24 weeks is effective at preventing hepatitis C virus recurrence in patients who undergo liver transplantations.
“Recurrent HCV infection is the most common cause of mortality and graft loss following transplantation, and up to 30% of patients with recurrent infection develop cirrhosis within 5 years,” wrote the study’s authors, led by Dr. Michael Charlton of the Mayo Clinic in Rochester, Minn.
Using a prospective, multicenter, open-label pilot study, investigators enrolled and treated 40 patients with a 24-week regimen of 400 mg sofosbuvir and ribavirin starting at 400 mg, which was subsequently adjusted per patient based on individual creatinine clearance and hemoglobin levels. Subjects were 78% male and 85% white, with 83% having HCV genotype 1, 40% having cirrhosis, and 88% having been previously treated with interferon. The primary outcome investigators looked for was “sustained virologic response 12 weeks after treatment (SVR12).”
Data showed that SVR12 was achieved by 28 of the 40 subjects that received treatment, or 70%. The most commonly reported adverse effects were fatigue (30%), diarrhea (28%), headache (25%), and anemia (20%). No patients exhibited detectable viral resistance during or after treatment, and although two patients terminated their treatment because of adverse events, investigators reported no deaths, graft losses, or episodes of rejection.
“In contrast,” Dr. Charlton and his coauthors noted, “interferon-based treatments have been associated with posttreatment immunological dysfunction (particularly plasma cell hepatitis) and even hepatic decompensation in LT [liver transplant] recipients.”
The authors of the first study disclosed that Dr. Curry has received grants from and been affiliated with Gilead, which was a sponsor of the study. The authors of the second study reported no relevant financial disclosures.
Sofosbuvir and ribavirin treatments should be administered to patients with hepatitis C virus who undergo liver transplantations in order to significantly decrease the risks of posttransplant HCV recurrence, according to two new studies published in the January issue of Gastroenterology (10.1053/j.gastro.2014.09.023 and 10.1053/j.gastro.2014.10.001).
“In clinical trials, administration of sofosbuvir with ribavirin was associated with rapid decreases of HCV RNA to undetectable levels in patients with HCV genotype 1, 2, 3, 4, and 6 infections,” wrote lead author Dr. Michael P. Curry of the Beth Israel Deaconess Medical Center in Boston, and his coauthors on the first of these two studies. “In more than 3,000 patients treated to date, sofosbuvir has been shown to be safe, viral breakthrough during treatment has been rare (and associated with nonadherence), and few drug interactions have been observed.”
In a phase II, open-label study, Dr. Curry and his coinvestigators enrolled 61 patients with HCV of any genotype, and cirrhosis with a Child-Turcotte-Pugh score no greater than 7, who were all wait-listed to receive liver transplantations. Subjects received up to 48 weeks of treatment with 400 mg of sofosbuvir, and a separate dose of ribavirin prior to liver transplantation, while 43 patients received transplantations alone. The primary outcome sought by investigators was HCV-RNA levels less than 25 IU/mL at 12 weeks after transplantation among patients that had this level prior to the operation.
The investigators found that 43 subjects had the desired HCV-RNA levels; of that population, 49% had a posttransplantation virologic response, with the most frequent side effects reported by subjects being fatigue (38%), headache (23%), and anemia (21%). Of the 43 applicable subjects, 30 (70% of the population) had a posttransplantation virologic response at 12 weeks, 10 (23%) had recurrent infection, and 3 (7%) died.
“This study provides proof of concept that virologic suppression without interferon significantly can reduce the rate of recurrent HCV after liver transplantation,” the study says, adding that the results “compare favorably with those observed in other trials of pretransplantation antiviral therapy.”
In the second study, the authors ascertained that combination therapy consisting of sofosbuvir and ribavirin for 24 weeks is effective at preventing hepatitis C virus recurrence in patients who undergo liver transplantations.
“Recurrent HCV infection is the most common cause of mortality and graft loss following transplantation, and up to 30% of patients with recurrent infection develop cirrhosis within 5 years,” wrote the study’s authors, led by Dr. Michael Charlton of the Mayo Clinic in Rochester, Minn.
Using a prospective, multicenter, open-label pilot study, investigators enrolled and treated 40 patients with a 24-week regimen of 400 mg sofosbuvir and ribavirin starting at 400 mg, which was subsequently adjusted per patient based on individual creatinine clearance and hemoglobin levels. Subjects were 78% male and 85% white, with 83% having HCV genotype 1, 40% having cirrhosis, and 88% having been previously treated with interferon. The primary outcome investigators looked for was “sustained virologic response 12 weeks after treatment (SVR12).”
Data showed that SVR12 was achieved by 28 of the 40 subjects that received treatment, or 70%. The most commonly reported adverse effects were fatigue (30%), diarrhea (28%), headache (25%), and anemia (20%). No patients exhibited detectable viral resistance during or after treatment, and although two patients terminated their treatment because of adverse events, investigators reported no deaths, graft losses, or episodes of rejection.
“In contrast,” Dr. Charlton and his coauthors noted, “interferon-based treatments have been associated with posttreatment immunological dysfunction (particularly plasma cell hepatitis) and even hepatic decompensation in LT [liver transplant] recipients.”
The authors of the first study disclosed that Dr. Curry has received grants from and been affiliated with Gilead, which was a sponsor of the study. The authors of the second study reported no relevant financial disclosures.
Sofosbuvir and ribavirin treatments should be administered to patients with hepatitis C virus who undergo liver transplantations in order to significantly decrease the risks of posttransplant HCV recurrence, according to two new studies published in the January issue of Gastroenterology (10.1053/j.gastro.2014.09.023 and 10.1053/j.gastro.2014.10.001).
“In clinical trials, administration of sofosbuvir with ribavirin was associated with rapid decreases of HCV RNA to undetectable levels in patients with HCV genotype 1, 2, 3, 4, and 6 infections,” wrote lead author Dr. Michael P. Curry of the Beth Israel Deaconess Medical Center in Boston, and his coauthors on the first of these two studies. “In more than 3,000 patients treated to date, sofosbuvir has been shown to be safe, viral breakthrough during treatment has been rare (and associated with nonadherence), and few drug interactions have been observed.”
In a phase II, open-label study, Dr. Curry and his coinvestigators enrolled 61 patients with HCV of any genotype, and cirrhosis with a Child-Turcotte-Pugh score no greater than 7, who were all wait-listed to receive liver transplantations. Subjects received up to 48 weeks of treatment with 400 mg of sofosbuvir, and a separate dose of ribavirin prior to liver transplantation, while 43 patients received transplantations alone. The primary outcome sought by investigators was HCV-RNA levels less than 25 IU/mL at 12 weeks after transplantation among patients that had this level prior to the operation.
The investigators found that 43 subjects had the desired HCV-RNA levels; of that population, 49% had a posttransplantation virologic response, with the most frequent side effects reported by subjects being fatigue (38%), headache (23%), and anemia (21%). Of the 43 applicable subjects, 30 (70% of the population) had a posttransplantation virologic response at 12 weeks, 10 (23%) had recurrent infection, and 3 (7%) died.
“This study provides proof of concept that virologic suppression without interferon significantly can reduce the rate of recurrent HCV after liver transplantation,” the study says, adding that the results “compare favorably with those observed in other trials of pretransplantation antiviral therapy.”
In the second study, the authors ascertained that combination therapy consisting of sofosbuvir and ribavirin for 24 weeks is effective at preventing hepatitis C virus recurrence in patients who undergo liver transplantations.
“Recurrent HCV infection is the most common cause of mortality and graft loss following transplantation, and up to 30% of patients with recurrent infection develop cirrhosis within 5 years,” wrote the study’s authors, led by Dr. Michael Charlton of the Mayo Clinic in Rochester, Minn.
Using a prospective, multicenter, open-label pilot study, investigators enrolled and treated 40 patients with a 24-week regimen of 400 mg sofosbuvir and ribavirin starting at 400 mg, which was subsequently adjusted per patient based on individual creatinine clearance and hemoglobin levels. Subjects were 78% male and 85% white, with 83% having HCV genotype 1, 40% having cirrhosis, and 88% having been previously treated with interferon. The primary outcome investigators looked for was “sustained virologic response 12 weeks after treatment (SVR12).”
Data showed that SVR12 was achieved by 28 of the 40 subjects that received treatment, or 70%. The most commonly reported adverse effects were fatigue (30%), diarrhea (28%), headache (25%), and anemia (20%). No patients exhibited detectable viral resistance during or after treatment, and although two patients terminated their treatment because of adverse events, investigators reported no deaths, graft losses, or episodes of rejection.
“In contrast,” Dr. Charlton and his coauthors noted, “interferon-based treatments have been associated with posttreatment immunological dysfunction (particularly plasma cell hepatitis) and even hepatic decompensation in LT [liver transplant] recipients.”
The authors of the first study disclosed that Dr. Curry has received grants from and been affiliated with Gilead, which was a sponsor of the study. The authors of the second study reported no relevant financial disclosures.
FROM GASTROENTEROLOGY
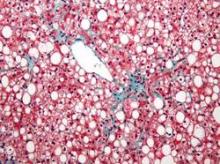
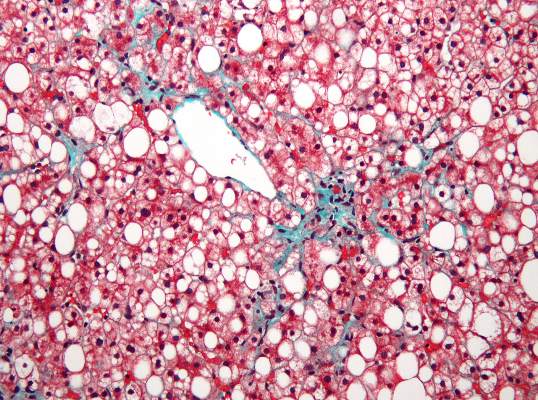
Diet, exercise reduced portal hypertension in obese cirrhotic patients
BOSTON – A 16-week diet and exercise program reduced body weight and portal pressure in a prospective pilot study of 50 obese patients with cirrhosis and portal hypertension.
Twenty-six patients achieved a clinically relevant weight loss of at least 5%, compared with baseline weight (52% of the cohort). The hepatic venous pressure gradient (HVPG) decreased by at least 10% in 21 patients (42%), compared with baseline, Dr. Annalisa Berzigotti and her associates reported at the annual meeting of the American Association for the Study of Liver Diseases.
Obesity has been shown to increase the risk of clinical decompensation in patients with compensated cirrhosis and portal hypertension, possibly by increasing portal hypertension, said Dr. Berzigotti of the Networked Research Center for Hepatic and Digestive Diseases (CIBERehd), Barcelona.
“An intensive 16-week program of tailored diet and moderate exercise can be safely recommended to obtain weight loss,” she said.
Dr. Berzigotti and her associates at two Spanish centers put patients through a 16-week program of a normoproteic, hypocaloric diet supervised by nutritionists. It followed a personalized decrease of 500-1,000 kcal/day, with 20%-25% of the diet consisting of proteins. The exercise intervention consisted of 60 minutes per week of supervised, moderately-intense physical activity in small groups plus personalized advice and use of a daily physical activity log.
Patients had a baseline HVPG of at least 6 mm Hg with or without esophageal varices and regardless of whether they were receiving nonselective beta-blocker medications.
The investigators recruited 60 patients, 50 of whom completed the study and were included in the analysis.
The lifestyle intervention decreased the mean body weight by 5 kg and the median body weight by 5%, which was associated with significant decreases in waist circumference and percentage body fat. Eight patients achieved at least a 10% reduction in body weight (16%), she reported.
The mean HVPG decreased significantly from 13.9 mm Hg at baseline to 12.3 mm Hg after treatment, with an average 11% reduction. The HVPG decreased by at least 20% in 12 patients (24%).
Four patients who lost at least 5% of their body weight reduced their HVPG to below 10 mm Hg. Patients who lost at least 10% of body weight reduced their HVPG to a greater degree than did patients who lost less than 10% of body weight, with 24% and 8% reductions in HVPG, respectively.
Changes in body weight and HVPG were more subtle in patients with diabetes than in those without diabetes. Results did not differ significantly across other subgroups based on cirrhosis etiology, clinically significant portal hypertension and esophageal varices, treatment with nonselective beta-blockers, history of variceal bleeding, or medical center.
No patients clinically decompensated during the study. Patients’ Child-Pugh scores and Model for End-Stage Liver Disease scores did not change.
Patients who lost weight kept it off for 6 months, with average weights of 86 kg at 16 weeks and 85 kg at a 6-month follow-up.
Patients had a mean age of 56 years and 62% were male. The etiologies of cirrhosis were viral in 36% of patients, alcoholic in 38%, and nonalcoholic steatohepatitis in 26%. Patients had an average body mass index of 33 kg/m2, and 72% had an HVPG of at least 10 mm Hg at baseline. Thirty percent of patients had a previous variceal hemorrhage but currently were compensated. Sixty-two percent of patients had a history of esophageal varices and 60% were on nonselective beta-blockers.
Dr. Berzigotti reported having no financial disclosures. One of her associates reported financial associations with Falk, Gilead, Norgine, Ono Pharma USA, Intercept Pharmaceuticals, Exalenz Bioscience, Almirall, and Conatus Pharmaceuticals.
On Twitter @sherryboschert
BOSTON – A 16-week diet and exercise program reduced body weight and portal pressure in a prospective pilot study of 50 obese patients with cirrhosis and portal hypertension.
Twenty-six patients achieved a clinically relevant weight loss of at least 5%, compared with baseline weight (52% of the cohort). The hepatic venous pressure gradient (HVPG) decreased by at least 10% in 21 patients (42%), compared with baseline, Dr. Annalisa Berzigotti and her associates reported at the annual meeting of the American Association for the Study of Liver Diseases.
Obesity has been shown to increase the risk of clinical decompensation in patients with compensated cirrhosis and portal hypertension, possibly by increasing portal hypertension, said Dr. Berzigotti of the Networked Research Center for Hepatic and Digestive Diseases (CIBERehd), Barcelona.
“An intensive 16-week program of tailored diet and moderate exercise can be safely recommended to obtain weight loss,” she said.
Dr. Berzigotti and her associates at two Spanish centers put patients through a 16-week program of a normoproteic, hypocaloric diet supervised by nutritionists. It followed a personalized decrease of 500-1,000 kcal/day, with 20%-25% of the diet consisting of proteins. The exercise intervention consisted of 60 minutes per week of supervised, moderately-intense physical activity in small groups plus personalized advice and use of a daily physical activity log.
Patients had a baseline HVPG of at least 6 mm Hg with or without esophageal varices and regardless of whether they were receiving nonselective beta-blocker medications.
The investigators recruited 60 patients, 50 of whom completed the study and were included in the analysis.
The lifestyle intervention decreased the mean body weight by 5 kg and the median body weight by 5%, which was associated with significant decreases in waist circumference and percentage body fat. Eight patients achieved at least a 10% reduction in body weight (16%), she reported.
The mean HVPG decreased significantly from 13.9 mm Hg at baseline to 12.3 mm Hg after treatment, with an average 11% reduction. The HVPG decreased by at least 20% in 12 patients (24%).
Four patients who lost at least 5% of their body weight reduced their HVPG to below 10 mm Hg. Patients who lost at least 10% of body weight reduced their HVPG to a greater degree than did patients who lost less than 10% of body weight, with 24% and 8% reductions in HVPG, respectively.
Changes in body weight and HVPG were more subtle in patients with diabetes than in those without diabetes. Results did not differ significantly across other subgroups based on cirrhosis etiology, clinically significant portal hypertension and esophageal varices, treatment with nonselective beta-blockers, history of variceal bleeding, or medical center.
No patients clinically decompensated during the study. Patients’ Child-Pugh scores and Model for End-Stage Liver Disease scores did not change.
Patients who lost weight kept it off for 6 months, with average weights of 86 kg at 16 weeks and 85 kg at a 6-month follow-up.
Patients had a mean age of 56 years and 62% were male. The etiologies of cirrhosis were viral in 36% of patients, alcoholic in 38%, and nonalcoholic steatohepatitis in 26%. Patients had an average body mass index of 33 kg/m2, and 72% had an HVPG of at least 10 mm Hg at baseline. Thirty percent of patients had a previous variceal hemorrhage but currently were compensated. Sixty-two percent of patients had a history of esophageal varices and 60% were on nonselective beta-blockers.
Dr. Berzigotti reported having no financial disclosures. One of her associates reported financial associations with Falk, Gilead, Norgine, Ono Pharma USA, Intercept Pharmaceuticals, Exalenz Bioscience, Almirall, and Conatus Pharmaceuticals.
On Twitter @sherryboschert
BOSTON – A 16-week diet and exercise program reduced body weight and portal pressure in a prospective pilot study of 50 obese patients with cirrhosis and portal hypertension.
Twenty-six patients achieved a clinically relevant weight loss of at least 5%, compared with baseline weight (52% of the cohort). The hepatic venous pressure gradient (HVPG) decreased by at least 10% in 21 patients (42%), compared with baseline, Dr. Annalisa Berzigotti and her associates reported at the annual meeting of the American Association for the Study of Liver Diseases.
Obesity has been shown to increase the risk of clinical decompensation in patients with compensated cirrhosis and portal hypertension, possibly by increasing portal hypertension, said Dr. Berzigotti of the Networked Research Center for Hepatic and Digestive Diseases (CIBERehd), Barcelona.
“An intensive 16-week program of tailored diet and moderate exercise can be safely recommended to obtain weight loss,” she said.
Dr. Berzigotti and her associates at two Spanish centers put patients through a 16-week program of a normoproteic, hypocaloric diet supervised by nutritionists. It followed a personalized decrease of 500-1,000 kcal/day, with 20%-25% of the diet consisting of proteins. The exercise intervention consisted of 60 minutes per week of supervised, moderately-intense physical activity in small groups plus personalized advice and use of a daily physical activity log.
Patients had a baseline HVPG of at least 6 mm Hg with or without esophageal varices and regardless of whether they were receiving nonselective beta-blocker medications.
The investigators recruited 60 patients, 50 of whom completed the study and were included in the analysis.
The lifestyle intervention decreased the mean body weight by 5 kg and the median body weight by 5%, which was associated with significant decreases in waist circumference and percentage body fat. Eight patients achieved at least a 10% reduction in body weight (16%), she reported.
The mean HVPG decreased significantly from 13.9 mm Hg at baseline to 12.3 mm Hg after treatment, with an average 11% reduction. The HVPG decreased by at least 20% in 12 patients (24%).
Four patients who lost at least 5% of their body weight reduced their HVPG to below 10 mm Hg. Patients who lost at least 10% of body weight reduced their HVPG to a greater degree than did patients who lost less than 10% of body weight, with 24% and 8% reductions in HVPG, respectively.
Changes in body weight and HVPG were more subtle in patients with diabetes than in those without diabetes. Results did not differ significantly across other subgroups based on cirrhosis etiology, clinically significant portal hypertension and esophageal varices, treatment with nonselective beta-blockers, history of variceal bleeding, or medical center.
No patients clinically decompensated during the study. Patients’ Child-Pugh scores and Model for End-Stage Liver Disease scores did not change.
Patients who lost weight kept it off for 6 months, with average weights of 86 kg at 16 weeks and 85 kg at a 6-month follow-up.
Patients had a mean age of 56 years and 62% were male. The etiologies of cirrhosis were viral in 36% of patients, alcoholic in 38%, and nonalcoholic steatohepatitis in 26%. Patients had an average body mass index of 33 kg/m2, and 72% had an HVPG of at least 10 mm Hg at baseline. Thirty percent of patients had a previous variceal hemorrhage but currently were compensated. Sixty-two percent of patients had a history of esophageal varices and 60% were on nonselective beta-blockers.
Dr. Berzigotti reported having no financial disclosures. One of her associates reported financial associations with Falk, Gilead, Norgine, Ono Pharma USA, Intercept Pharmaceuticals, Exalenz Bioscience, Almirall, and Conatus Pharmaceuticals.
On Twitter @sherryboschert
AT THE LIVER MEETING 2014
Key clinical point: A 16-week lifestyle intervention safely reduced body weight and portal pressure in obese patients with cirrhosis.
Major finding: Clinically relevant weight loss occurred in 26 patients (52%) and the HVPG decreased by at least 10% in 21 patients (42%), compared with baseline.
Data source: A prospective, multicenter pilot study of 50 obese patients with cirrhosis and portal hypertension.
Disclosures:Dr. Berzigotti reported having no financial disclosures. One of her associates reported ties with Falk, Gilead, Norgine, Ono Pharma USA, Intercept Pharmaceuticals, Exalenz Bioscience, Almirall, and Conatus Pharmaceuticals.
Vasopressor terlipressin helped patients with cirrhosis, shock
BOSTON – Terlipressin proved noninferior to noradrenaline as a vasopressor for patients with cirrhosis and septic shock in an open-label study that randomized 84 patients.
Terlipressin also seemed to provide advantages over noradrenaline by contributing to greater hemodynamic stability, improved urine output, reduced variceal bleeding, a lower risk of spontaneous bacterial peritonitis, and reduced mortality in the first 48 hours but not at 28 days, Dr. Ashok K. Choudhury and his associates reported.
“Terlipressin is noninferior to noradrenaline as a vasopressor,” he said at the annual meeting of the American Association for the Study of Liver Diseases.
The investigators randomized consecutive, matched adults with cirrhosis and septic shock not responding to fluid resuscitation for 2 hours. They aimed to achieve a mean arterial pressure (MAP) > 65 mm Hg at 6 hours by treating 42 patients with a continuous infusion of terlipressin 1.3-5.2 mcg/min, stepped up every 15 minutes, and 42 patients with noradrenaline 7.5-6 mcg/min, stepped up every 15 minutes. Both groups received standard medical care of intravenous fluids, antibiotics, and ICU care.
The target MAP was reached within 6 hours in 28 patients on terlipressin (67%) and 23 patients on noradrenaline (55%), a difference that was not statistically significant, said Dr. Choudhury of the Institute of Liver and Biliary Sciences, New Delhi. He won a research award from the Association.
Patients in the two groups were matched by age, sex, etiology and severity of cirrhosis, and scores on the Model for End-Stage Liver Disease and the Sequential Organ Failure Assessment.
Spontaneous bacterial peritonitis was the main cause of sepsis at admission, followed by pneumonia, but pneumonia was the main cause of “second hit” pneumonia, he said.
Terlipressin therapy was associated with a lower failure rate at 6 hours, a greater likelihood of MAP maintenance with cessation of vasopressor requirements at 48 hours, and improved urine output at 24 hours, compared with noradrenaline treatment. At 48 hours, 19 of 40 patients on terlipressin (48%) and 11 of 30 patients on noradrenaline (36%) had an MAP > 65 mm Hg.
No patients on terlipressin developed a variceal bleed, compared with seven patients in the noradrenaline group (10%). Overall rates of adverse events did not differ significantly between groups, but were higher in the terlipressin group than in the noradrenaline group.
Better rates of lactate clearance, central venous oxygen saturation, and carbon dioxide gradient in venous-arterial blood gases in the terlipressin group were not statistically different from rates in the noradrenaline group.
Significantly more patients on terlipressin survived the first 48 hours than patients on noradrenaline, but by day 28 of follow-up survival rates did not differ significantly between groups.
On the whole, septic shock in these patients with cirrhosis was associated with a high mortality rate (80%). Twelve percent of patients were responsive to fluids in 2 hours.
Dr. Choudhury reported having no financial disclosures.
On Twitter @sherryboschert
BOSTON – Terlipressin proved noninferior to noradrenaline as a vasopressor for patients with cirrhosis and septic shock in an open-label study that randomized 84 patients.
Terlipressin also seemed to provide advantages over noradrenaline by contributing to greater hemodynamic stability, improved urine output, reduced variceal bleeding, a lower risk of spontaneous bacterial peritonitis, and reduced mortality in the first 48 hours but not at 28 days, Dr. Ashok K. Choudhury and his associates reported.
“Terlipressin is noninferior to noradrenaline as a vasopressor,” he said at the annual meeting of the American Association for the Study of Liver Diseases.
The investigators randomized consecutive, matched adults with cirrhosis and septic shock not responding to fluid resuscitation for 2 hours. They aimed to achieve a mean arterial pressure (MAP) > 65 mm Hg at 6 hours by treating 42 patients with a continuous infusion of terlipressin 1.3-5.2 mcg/min, stepped up every 15 minutes, and 42 patients with noradrenaline 7.5-6 mcg/min, stepped up every 15 minutes. Both groups received standard medical care of intravenous fluids, antibiotics, and ICU care.
The target MAP was reached within 6 hours in 28 patients on terlipressin (67%) and 23 patients on noradrenaline (55%), a difference that was not statistically significant, said Dr. Choudhury of the Institute of Liver and Biliary Sciences, New Delhi. He won a research award from the Association.
Patients in the two groups were matched by age, sex, etiology and severity of cirrhosis, and scores on the Model for End-Stage Liver Disease and the Sequential Organ Failure Assessment.
Spontaneous bacterial peritonitis was the main cause of sepsis at admission, followed by pneumonia, but pneumonia was the main cause of “second hit” pneumonia, he said.
Terlipressin therapy was associated with a lower failure rate at 6 hours, a greater likelihood of MAP maintenance with cessation of vasopressor requirements at 48 hours, and improved urine output at 24 hours, compared with noradrenaline treatment. At 48 hours, 19 of 40 patients on terlipressin (48%) and 11 of 30 patients on noradrenaline (36%) had an MAP > 65 mm Hg.
No patients on terlipressin developed a variceal bleed, compared with seven patients in the noradrenaline group (10%). Overall rates of adverse events did not differ significantly between groups, but were higher in the terlipressin group than in the noradrenaline group.
Better rates of lactate clearance, central venous oxygen saturation, and carbon dioxide gradient in venous-arterial blood gases in the terlipressin group were not statistically different from rates in the noradrenaline group.
Significantly more patients on terlipressin survived the first 48 hours than patients on noradrenaline, but by day 28 of follow-up survival rates did not differ significantly between groups.
On the whole, septic shock in these patients with cirrhosis was associated with a high mortality rate (80%). Twelve percent of patients were responsive to fluids in 2 hours.
Dr. Choudhury reported having no financial disclosures.
On Twitter @sherryboschert
BOSTON – Terlipressin proved noninferior to noradrenaline as a vasopressor for patients with cirrhosis and septic shock in an open-label study that randomized 84 patients.
Terlipressin also seemed to provide advantages over noradrenaline by contributing to greater hemodynamic stability, improved urine output, reduced variceal bleeding, a lower risk of spontaneous bacterial peritonitis, and reduced mortality in the first 48 hours but not at 28 days, Dr. Ashok K. Choudhury and his associates reported.
“Terlipressin is noninferior to noradrenaline as a vasopressor,” he said at the annual meeting of the American Association for the Study of Liver Diseases.
The investigators randomized consecutive, matched adults with cirrhosis and septic shock not responding to fluid resuscitation for 2 hours. They aimed to achieve a mean arterial pressure (MAP) > 65 mm Hg at 6 hours by treating 42 patients with a continuous infusion of terlipressin 1.3-5.2 mcg/min, stepped up every 15 minutes, and 42 patients with noradrenaline 7.5-6 mcg/min, stepped up every 15 minutes. Both groups received standard medical care of intravenous fluids, antibiotics, and ICU care.
The target MAP was reached within 6 hours in 28 patients on terlipressin (67%) and 23 patients on noradrenaline (55%), a difference that was not statistically significant, said Dr. Choudhury of the Institute of Liver and Biliary Sciences, New Delhi. He won a research award from the Association.
Patients in the two groups were matched by age, sex, etiology and severity of cirrhosis, and scores on the Model for End-Stage Liver Disease and the Sequential Organ Failure Assessment.
Spontaneous bacterial peritonitis was the main cause of sepsis at admission, followed by pneumonia, but pneumonia was the main cause of “second hit” pneumonia, he said.
Terlipressin therapy was associated with a lower failure rate at 6 hours, a greater likelihood of MAP maintenance with cessation of vasopressor requirements at 48 hours, and improved urine output at 24 hours, compared with noradrenaline treatment. At 48 hours, 19 of 40 patients on terlipressin (48%) and 11 of 30 patients on noradrenaline (36%) had an MAP > 65 mm Hg.
No patients on terlipressin developed a variceal bleed, compared with seven patients in the noradrenaline group (10%). Overall rates of adverse events did not differ significantly between groups, but were higher in the terlipressin group than in the noradrenaline group.
Better rates of lactate clearance, central venous oxygen saturation, and carbon dioxide gradient in venous-arterial blood gases in the terlipressin group were not statistically different from rates in the noradrenaline group.
Significantly more patients on terlipressin survived the first 48 hours than patients on noradrenaline, but by day 28 of follow-up survival rates did not differ significantly between groups.
On the whole, septic shock in these patients with cirrhosis was associated with a high mortality rate (80%). Twelve percent of patients were responsive to fluids in 2 hours.
Dr. Choudhury reported having no financial disclosures.
On Twitter @sherryboschert
AT THE LIVER MEETING 2014
Key clinical point: Terlipressin worked as well as noradrenaline as a vasopressor.
Major finding: Treatment achieved a MAP of >65 mm Hg by 6 hours in 28 patients on terlipressin (67%) and in 23 patients on noradrenaline (55%).
Data source: A prospective, randomized, open-label study of 84 patients with cirrhosis and septic shock.
Disclosures: Dr. Choudhury reported having no financial disclosures.
Preventing, treating HBV reactivation during immunosuppressive therapy
Patients who are to undergo immunosuppressive therapy but are at high risk for reactivation of hepatitis B infection should receive antiviral prophylaxis, rather than being monitored for reactivation and treated only if it develops, according to a new guideline published online in Gastroenterology.
The American Gastroenterological Association conducted a rigorous review of the available evidence and compiled seven recommendations to guide clinicians and researchers in preventing HBV reactivation before patients initiate immunosuppressive therapy and in treating reactivated HBV if it arises during immunosuppressive therapy. “Despite the large number of published studies, in most cases our recommendations are weak because either (1) the quality of the available data and/or the baseline risk of HBV reactivation is low or uncertain, and/or (2) the balance of risks and benefits for a particular strategy does not overwhelmingly support its use,” said Dr. K. Rajender Reddy, lead writer of the guideline and professor of gastroenterology and hepatology at the University of Pennsylvania, Philadelphia, and his associates.
In contrast, the data supporting the recommendation to provide prophylaxis for high-risk patients are moderately robust, so that recommendation is strong, they noted.
Patients’ level of risk is based on their HBV serologic status and the type of immunosuppression they require. For example, patients are considered at high risk for HBV reactivation if they are hepatitis B surface antigen (HBsAg) positive/anti–hepatitis B core (HBc) positive or are HBsAg negative/anti-HBc positive and are to be treated with B-cell–depleting agents such as rituximab or ofatumumab. Also at high risk are HBsAg-positive/anti-HBc–positive patients to be treated with anthracycline derivatives such as doxorubicin or epirubicin, and HBsAg-positive/anti-HBc–positive patients to be treated with moderate- or high-dose corticosteroids for 4 weeks or longer.
In these high-risk patients, antiviral prophylaxis is definitely warranted, and it should extend for at least 6 months after the immunosuppressive therapy is completed, according to the guideline. In contrast, antiviral prophylaxis is “suggested” for moderate-risk patients, but those who place a higher value on avoiding antivirals and a lower value on avoiding HBV reactivation “may reasonably select no prophylaxis over antiviral prophylaxis,” Dr. Reddy and his associates said.
In contrast, routine antiviral prophylaxis is not recommended for patients undergoing immunosuppressive therapy who are at low risk for HBV reactivation.
Other recommendations in the new guideline concern which antiviral agents are preferred in different situations. The AGA strongly recommends antivirals that have a high barrier to resistance, in preference to lamivudine, in most patients. But it acknowledges that the evidence comparing various antivirals is weak, and that the drugs vary considerably in price. “Patients who put a higher value on cost and a lower value on avoiding the potentially small risk of resistance development may reasonably select the least expensive antiviral hepatitis B medication over more expensive antiviral drugs with a higher barrier to resistance,” Dr. Reddy and his associates said (Gastroenterol. 2014 [doi:10.1053/j.gastro.2014.10.039]).
The evidence regarding patient monitoring for HBV reactivation instead of prophylaxis is so sparse that the AGA makes no recommendation for or against this strategy at present. Most studies of the issue are of poor quality, use different definitions of HBV reactivation, have inconsistent reporting of patient outcomes, and disagree about the best methods and frequency of HBV DNA monitoring. Moreover, frequent monitoring requires “considerable” personnel resources, and the methods used in clinical studies may not even be adaptable to real world practice, the investigators said.
Patients who are to undergo immunosuppressive therapy but are at high risk for reactivation of hepatitis B infection should receive antiviral prophylaxis, rather than being monitored for reactivation and treated only if it develops, according to a new guideline published online in Gastroenterology.
The American Gastroenterological Association conducted a rigorous review of the available evidence and compiled seven recommendations to guide clinicians and researchers in preventing HBV reactivation before patients initiate immunosuppressive therapy and in treating reactivated HBV if it arises during immunosuppressive therapy. “Despite the large number of published studies, in most cases our recommendations are weak because either (1) the quality of the available data and/or the baseline risk of HBV reactivation is low or uncertain, and/or (2) the balance of risks and benefits for a particular strategy does not overwhelmingly support its use,” said Dr. K. Rajender Reddy, lead writer of the guideline and professor of gastroenterology and hepatology at the University of Pennsylvania, Philadelphia, and his associates.
In contrast, the data supporting the recommendation to provide prophylaxis for high-risk patients are moderately robust, so that recommendation is strong, they noted.
Patients’ level of risk is based on their HBV serologic status and the type of immunosuppression they require. For example, patients are considered at high risk for HBV reactivation if they are hepatitis B surface antigen (HBsAg) positive/anti–hepatitis B core (HBc) positive or are HBsAg negative/anti-HBc positive and are to be treated with B-cell–depleting agents such as rituximab or ofatumumab. Also at high risk are HBsAg-positive/anti-HBc–positive patients to be treated with anthracycline derivatives such as doxorubicin or epirubicin, and HBsAg-positive/anti-HBc–positive patients to be treated with moderate- or high-dose corticosteroids for 4 weeks or longer.
In these high-risk patients, antiviral prophylaxis is definitely warranted, and it should extend for at least 6 months after the immunosuppressive therapy is completed, according to the guideline. In contrast, antiviral prophylaxis is “suggested” for moderate-risk patients, but those who place a higher value on avoiding antivirals and a lower value on avoiding HBV reactivation “may reasonably select no prophylaxis over antiviral prophylaxis,” Dr. Reddy and his associates said.
In contrast, routine antiviral prophylaxis is not recommended for patients undergoing immunosuppressive therapy who are at low risk for HBV reactivation.
Other recommendations in the new guideline concern which antiviral agents are preferred in different situations. The AGA strongly recommends antivirals that have a high barrier to resistance, in preference to lamivudine, in most patients. But it acknowledges that the evidence comparing various antivirals is weak, and that the drugs vary considerably in price. “Patients who put a higher value on cost and a lower value on avoiding the potentially small risk of resistance development may reasonably select the least expensive antiviral hepatitis B medication over more expensive antiviral drugs with a higher barrier to resistance,” Dr. Reddy and his associates said (Gastroenterol. 2014 [doi:10.1053/j.gastro.2014.10.039]).
The evidence regarding patient monitoring for HBV reactivation instead of prophylaxis is so sparse that the AGA makes no recommendation for or against this strategy at present. Most studies of the issue are of poor quality, use different definitions of HBV reactivation, have inconsistent reporting of patient outcomes, and disagree about the best methods and frequency of HBV DNA monitoring. Moreover, frequent monitoring requires “considerable” personnel resources, and the methods used in clinical studies may not even be adaptable to real world practice, the investigators said.
Patients who are to undergo immunosuppressive therapy but are at high risk for reactivation of hepatitis B infection should receive antiviral prophylaxis, rather than being monitored for reactivation and treated only if it develops, according to a new guideline published online in Gastroenterology.
The American Gastroenterological Association conducted a rigorous review of the available evidence and compiled seven recommendations to guide clinicians and researchers in preventing HBV reactivation before patients initiate immunosuppressive therapy and in treating reactivated HBV if it arises during immunosuppressive therapy. “Despite the large number of published studies, in most cases our recommendations are weak because either (1) the quality of the available data and/or the baseline risk of HBV reactivation is low or uncertain, and/or (2) the balance of risks and benefits for a particular strategy does not overwhelmingly support its use,” said Dr. K. Rajender Reddy, lead writer of the guideline and professor of gastroenterology and hepatology at the University of Pennsylvania, Philadelphia, and his associates.
In contrast, the data supporting the recommendation to provide prophylaxis for high-risk patients are moderately robust, so that recommendation is strong, they noted.
Patients’ level of risk is based on their HBV serologic status and the type of immunosuppression they require. For example, patients are considered at high risk for HBV reactivation if they are hepatitis B surface antigen (HBsAg) positive/anti–hepatitis B core (HBc) positive or are HBsAg negative/anti-HBc positive and are to be treated with B-cell–depleting agents such as rituximab or ofatumumab. Also at high risk are HBsAg-positive/anti-HBc–positive patients to be treated with anthracycline derivatives such as doxorubicin or epirubicin, and HBsAg-positive/anti-HBc–positive patients to be treated with moderate- or high-dose corticosteroids for 4 weeks or longer.
In these high-risk patients, antiviral prophylaxis is definitely warranted, and it should extend for at least 6 months after the immunosuppressive therapy is completed, according to the guideline. In contrast, antiviral prophylaxis is “suggested” for moderate-risk patients, but those who place a higher value on avoiding antivirals and a lower value on avoiding HBV reactivation “may reasonably select no prophylaxis over antiviral prophylaxis,” Dr. Reddy and his associates said.
In contrast, routine antiviral prophylaxis is not recommended for patients undergoing immunosuppressive therapy who are at low risk for HBV reactivation.
Other recommendations in the new guideline concern which antiviral agents are preferred in different situations. The AGA strongly recommends antivirals that have a high barrier to resistance, in preference to lamivudine, in most patients. But it acknowledges that the evidence comparing various antivirals is weak, and that the drugs vary considerably in price. “Patients who put a higher value on cost and a lower value on avoiding the potentially small risk of resistance development may reasonably select the least expensive antiviral hepatitis B medication over more expensive antiviral drugs with a higher barrier to resistance,” Dr. Reddy and his associates said (Gastroenterol. 2014 [doi:10.1053/j.gastro.2014.10.039]).
The evidence regarding patient monitoring for HBV reactivation instead of prophylaxis is so sparse that the AGA makes no recommendation for or against this strategy at present. Most studies of the issue are of poor quality, use different definitions of HBV reactivation, have inconsistent reporting of patient outcomes, and disagree about the best methods and frequency of HBV DNA monitoring. Moreover, frequent monitoring requires “considerable” personnel resources, and the methods used in clinical studies may not even be adaptable to real world practice, the investigators said.
FROM GASTROENTEROLOGY
Key clinical point: The American Gastroenterological Association published a guideline for preventing the reactivation of HBV in patients who need immunosuppressive therapy and for treating reactivated HBV when it develops in patients undergoing immunosuppression.
Major finding: Robust evidence supports the recommendation that antiviral prophylaxis is warranted in patients at high risk for HBV reactivation, “suggested” for patients at moderate risk, and not recommended for patients at low risk.
Data source: A rigorous review and summary of the available evidence regarding prevention and treatment of HBV reactivation, and a compilation of recommendations for clinicians and researchers.
Disclosures: Dr. Reddy and his associates’ disclosure statements are available at the American Gastroenterological Association, Bethesda, Md. .
Aramchol shows promise for NAFLD; resveratrol disappoints
Administration of a novel fatty acid–bile acid conjugate safely and significantly reduced liver fat content in patients with nonalcoholic fatty liver disease in a phase II trial.
Administration of resveratrol for 8 weeks, however, failed to improve any features of the disease in a separate placebo-controlled study.
In 60 patients aged 18-75 years in a randomized, double-blind, placebo-controlled study of the fatty acid–bile acid conjugate aramchol (Trima Israel Pharmaceutical Products Ltd. Maabarot), 3 months of treatment was associated with a decrease in liver fat content by a mean of 12.57% as measured by magnetic resonance spectroscopy, compared with an increase in liver fat by a mean of 6.39% in patients who received placebo, Dr. Rifaat Safadi of Hadassah University Medical Center, Jerusalem and colleagues reported in the DEcember issue of Clinical Gastroenterology and Hepatology.
The difference between the treatment and placebo groups was significant, even after age, sex, and body mass index were adjusted for.
Liver fat content also decreased in patients who received treatment with a 100-mg dose of aramchol, but the difference compared with placebo was not statistically significant, indicating a dose-response relationship in treated patients, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.1016/j.cgh.2014.04.038]).
A trend was seen for improvements over time in endothelial function and levels of alanine aminotransferase and adiponectin.
Patients in the study were from 10 centers in Israel, and all had biopsy-confirmed NAFLD (54 patients) or nonalcoholic steatohepatitis (NASH, 6 patients). They were randomized to receive 100 or 300 mg of aramchol or placebo once daily for 3 months.
No serious or drug-related adverse events occurred in the treated patients, the investigators said.
The findings suggest that aramchol is a candidate for the treatment of fatty liver–related diseases, which are “an increasingly relevant public health issue because of their association with the worldwide epidemics of diabetes and obesity,” and for which treatments are lacking, they said, concluding that longer trials in patients with NASH and metabolic complications are warranted to evaluate metabolic and histologic benefits of treatment.
Findings from the resveratrol study were less encouraging.
Eight weeks of treatment at 3,000 mg daily in overweight or obese men with NAFLD who were recruited from clinics in Brisbane from 2011 through 2012 was no better than placebo for improving insulin resistance, hepatic steatosis, or abdominal fat distribution, Dr. Veronique S. Chachay of the University of Queensland, Brisbane, Australia and her colleagues also reported in the December issue of Clinical Gastroenterology and Hepatology.
No changes were observed in plasma lipids or antioxidant activity, and levels of alanine and aspartate aminotransferases increased significantly (from 45 to 63 U/L and from 35 to 45 U/L, respectively) until week 6 in the 10 treated patients, compared with the 10 who received placebo (40 to 48 U/L and 36 to 38 U/L, respectively). Resveratrol did not significantly alter transcription of NQ01, PTP1B, IL6, or H01, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.106/j.cgh.2014.02.024]).
NAFLD is associated with abdominal obesity, insulin resistance, and inflammation, and while weight loss via calorie restriction is known to reduce features of the disease, there is no pharmacologic therapy available to achieve this end.
The investigators sought to determine if resveratrol – a polyphenol that has been shown to prevent high-energy diet-induced steatosis and insulin resistance in animals – might be beneficial in patients with NAFLD.
“The present study demonstrates that the preventive role of resveratrol observed in diet-induced preclinical models of NAFLD does not translate into a therapeutic role in clinically established NAFLD,” they said, adding that clinical dose-finding studies are “paramount to elucidate the dose-response relationship” and that “the purported calorie-restriction mimicking of resveratrol may require investigation in combination with dietary prescription, standard care, and lifestyle modifications to target adequately the complexity of dysregulation in obesity-related chronic disease.”
The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Administration of a novel fatty acid–bile acid conjugate safely and significantly reduced liver fat content in patients with nonalcoholic fatty liver disease in a phase II trial.
Administration of resveratrol for 8 weeks, however, failed to improve any features of the disease in a separate placebo-controlled study.
In 60 patients aged 18-75 years in a randomized, double-blind, placebo-controlled study of the fatty acid–bile acid conjugate aramchol (Trima Israel Pharmaceutical Products Ltd. Maabarot), 3 months of treatment was associated with a decrease in liver fat content by a mean of 12.57% as measured by magnetic resonance spectroscopy, compared with an increase in liver fat by a mean of 6.39% in patients who received placebo, Dr. Rifaat Safadi of Hadassah University Medical Center, Jerusalem and colleagues reported in the DEcember issue of Clinical Gastroenterology and Hepatology.
The difference between the treatment and placebo groups was significant, even after age, sex, and body mass index were adjusted for.
Liver fat content also decreased in patients who received treatment with a 100-mg dose of aramchol, but the difference compared with placebo was not statistically significant, indicating a dose-response relationship in treated patients, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.1016/j.cgh.2014.04.038]).
A trend was seen for improvements over time in endothelial function and levels of alanine aminotransferase and adiponectin.
Patients in the study were from 10 centers in Israel, and all had biopsy-confirmed NAFLD (54 patients) or nonalcoholic steatohepatitis (NASH, 6 patients). They were randomized to receive 100 or 300 mg of aramchol or placebo once daily for 3 months.
No serious or drug-related adverse events occurred in the treated patients, the investigators said.
The findings suggest that aramchol is a candidate for the treatment of fatty liver–related diseases, which are “an increasingly relevant public health issue because of their association with the worldwide epidemics of diabetes and obesity,” and for which treatments are lacking, they said, concluding that longer trials in patients with NASH and metabolic complications are warranted to evaluate metabolic and histologic benefits of treatment.
Findings from the resveratrol study were less encouraging.
Eight weeks of treatment at 3,000 mg daily in overweight or obese men with NAFLD who were recruited from clinics in Brisbane from 2011 through 2012 was no better than placebo for improving insulin resistance, hepatic steatosis, or abdominal fat distribution, Dr. Veronique S. Chachay of the University of Queensland, Brisbane, Australia and her colleagues also reported in the December issue of Clinical Gastroenterology and Hepatology.
No changes were observed in plasma lipids or antioxidant activity, and levels of alanine and aspartate aminotransferases increased significantly (from 45 to 63 U/L and from 35 to 45 U/L, respectively) until week 6 in the 10 treated patients, compared with the 10 who received placebo (40 to 48 U/L and 36 to 38 U/L, respectively). Resveratrol did not significantly alter transcription of NQ01, PTP1B, IL6, or H01, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.106/j.cgh.2014.02.024]).
NAFLD is associated with abdominal obesity, insulin resistance, and inflammation, and while weight loss via calorie restriction is known to reduce features of the disease, there is no pharmacologic therapy available to achieve this end.
The investigators sought to determine if resveratrol – a polyphenol that has been shown to prevent high-energy diet-induced steatosis and insulin resistance in animals – might be beneficial in patients with NAFLD.
“The present study demonstrates that the preventive role of resveratrol observed in diet-induced preclinical models of NAFLD does not translate into a therapeutic role in clinically established NAFLD,” they said, adding that clinical dose-finding studies are “paramount to elucidate the dose-response relationship” and that “the purported calorie-restriction mimicking of resveratrol may require investigation in combination with dietary prescription, standard care, and lifestyle modifications to target adequately the complexity of dysregulation in obesity-related chronic disease.”
The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Administration of a novel fatty acid–bile acid conjugate safely and significantly reduced liver fat content in patients with nonalcoholic fatty liver disease in a phase II trial.
Administration of resveratrol for 8 weeks, however, failed to improve any features of the disease in a separate placebo-controlled study.
In 60 patients aged 18-75 years in a randomized, double-blind, placebo-controlled study of the fatty acid–bile acid conjugate aramchol (Trima Israel Pharmaceutical Products Ltd. Maabarot), 3 months of treatment was associated with a decrease in liver fat content by a mean of 12.57% as measured by magnetic resonance spectroscopy, compared with an increase in liver fat by a mean of 6.39% in patients who received placebo, Dr. Rifaat Safadi of Hadassah University Medical Center, Jerusalem and colleagues reported in the DEcember issue of Clinical Gastroenterology and Hepatology.
The difference between the treatment and placebo groups was significant, even after age, sex, and body mass index were adjusted for.
Liver fat content also decreased in patients who received treatment with a 100-mg dose of aramchol, but the difference compared with placebo was not statistically significant, indicating a dose-response relationship in treated patients, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.1016/j.cgh.2014.04.038]).
A trend was seen for improvements over time in endothelial function and levels of alanine aminotransferase and adiponectin.
Patients in the study were from 10 centers in Israel, and all had biopsy-confirmed NAFLD (54 patients) or nonalcoholic steatohepatitis (NASH, 6 patients). They were randomized to receive 100 or 300 mg of aramchol or placebo once daily for 3 months.
No serious or drug-related adverse events occurred in the treated patients, the investigators said.
The findings suggest that aramchol is a candidate for the treatment of fatty liver–related diseases, which are “an increasingly relevant public health issue because of their association with the worldwide epidemics of diabetes and obesity,” and for which treatments are lacking, they said, concluding that longer trials in patients with NASH and metabolic complications are warranted to evaluate metabolic and histologic benefits of treatment.
Findings from the resveratrol study were less encouraging.
Eight weeks of treatment at 3,000 mg daily in overweight or obese men with NAFLD who were recruited from clinics in Brisbane from 2011 through 2012 was no better than placebo for improving insulin resistance, hepatic steatosis, or abdominal fat distribution, Dr. Veronique S. Chachay of the University of Queensland, Brisbane, Australia and her colleagues also reported in the December issue of Clinical Gastroenterology and Hepatology.
No changes were observed in plasma lipids or antioxidant activity, and levels of alanine and aspartate aminotransferases increased significantly (from 45 to 63 U/L and from 35 to 45 U/L, respectively) until week 6 in the 10 treated patients, compared with the 10 who received placebo (40 to 48 U/L and 36 to 38 U/L, respectively). Resveratrol did not significantly alter transcription of NQ01, PTP1B, IL6, or H01, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.106/j.cgh.2014.02.024]).
NAFLD is associated with abdominal obesity, insulin resistance, and inflammation, and while weight loss via calorie restriction is known to reduce features of the disease, there is no pharmacologic therapy available to achieve this end.
The investigators sought to determine if resveratrol – a polyphenol that has been shown to prevent high-energy diet-induced steatosis and insulin resistance in animals – might be beneficial in patients with NAFLD.
“The present study demonstrates that the preventive role of resveratrol observed in diet-induced preclinical models of NAFLD does not translate into a therapeutic role in clinically established NAFLD,” they said, adding that clinical dose-finding studies are “paramount to elucidate the dose-response relationship” and that “the purported calorie-restriction mimicking of resveratrol may require investigation in combination with dietary prescription, standard care, and lifestyle modifications to target adequately the complexity of dysregulation in obesity-related chronic disease.”
The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Key clinical point: Aramchol is a candidate treatment for fatty liver–related diseases; resveratrol requires dose-finding and other studies.
Major finding: The change in liver fat content was a mean of -12.57% with aramchol vs. +6.39% with placebo. Resveratol had no therapeutic benefit in NAFLD.
Data source: A phase II study of 60 patients, and a randomized, controlled study of 20 patients.
Disclosures: The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
VIDEO: Pediatric NAFLD worsens as kids age
BOSTON – Pediatric nonalcoholic fatty liver disease progresses as children age to a more adult pattern of disease, a paired-biopsy study shows.
“As they grow older, they are facing liver transplant and potentially hepatocellular carcinoma, just as the adults do,” Dr. Elizabeth M. Brunt said during an interview at the annual meeting of the American Association for the Study of Liver Diseases.
Among 102 children studied, the zone 1 (borderline 1b) diagnostic pattern decreased from 27.5% to 9.8%, while the more “adult” NAFLD zone 3 (borderline 1a) pattern and definite steatohepatitis patterns both increased from 14.7% and 28.4% to 18.6% and 29.4%.
Moreover, cirrhosis was seen in nearly 3% of children at first biopsy, but by the second biopsy, nearly 20% of children had advanced fibrosis or cirrhosis, she said.
The findings are troubling because the United States is in the midst of an obesity epidemic, and obesity is associated with high rates of fatty liver disease, said Dr. Brunt of Washington University, St. Louis.
The National Institutes of Health supported the study. Dr. Brunt reported consulting for Synageva, serving as an independent contractor for Rottapharm and Kadmon, and speaking and teaching for the Geneva Foundation.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BOSTON – Pediatric nonalcoholic fatty liver disease progresses as children age to a more adult pattern of disease, a paired-biopsy study shows.
“As they grow older, they are facing liver transplant and potentially hepatocellular carcinoma, just as the adults do,” Dr. Elizabeth M. Brunt said during an interview at the annual meeting of the American Association for the Study of Liver Diseases.
Among 102 children studied, the zone 1 (borderline 1b) diagnostic pattern decreased from 27.5% to 9.8%, while the more “adult” NAFLD zone 3 (borderline 1a) pattern and definite steatohepatitis patterns both increased from 14.7% and 28.4% to 18.6% and 29.4%.
Moreover, cirrhosis was seen in nearly 3% of children at first biopsy, but by the second biopsy, nearly 20% of children had advanced fibrosis or cirrhosis, she said.
The findings are troubling because the United States is in the midst of an obesity epidemic, and obesity is associated with high rates of fatty liver disease, said Dr. Brunt of Washington University, St. Louis.
The National Institutes of Health supported the study. Dr. Brunt reported consulting for Synageva, serving as an independent contractor for Rottapharm and Kadmon, and speaking and teaching for the Geneva Foundation.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BOSTON – Pediatric nonalcoholic fatty liver disease progresses as children age to a more adult pattern of disease, a paired-biopsy study shows.
“As they grow older, they are facing liver transplant and potentially hepatocellular carcinoma, just as the adults do,” Dr. Elizabeth M. Brunt said during an interview at the annual meeting of the American Association for the Study of Liver Diseases.
Among 102 children studied, the zone 1 (borderline 1b) diagnostic pattern decreased from 27.5% to 9.8%, while the more “adult” NAFLD zone 3 (borderline 1a) pattern and definite steatohepatitis patterns both increased from 14.7% and 28.4% to 18.6% and 29.4%.
Moreover, cirrhosis was seen in nearly 3% of children at first biopsy, but by the second biopsy, nearly 20% of children had advanced fibrosis or cirrhosis, she said.
The findings are troubling because the United States is in the midst of an obesity epidemic, and obesity is associated with high rates of fatty liver disease, said Dr. Brunt of Washington University, St. Louis.
The National Institutes of Health supported the study. Dr. Brunt reported consulting for Synageva, serving as an independent contractor for Rottapharm and Kadmon, and speaking and teaching for the Geneva Foundation.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
FROM THE LIVER MEETING 2014