User login
Vocal Cord Dysfunction: Unmasking the Asthma Pretender
CE/CME No: CR-1412
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest and evaluation. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
• Discuss the evolution in thinking about the pathogenesis of and treatment for vocal cord dysfunction (VCD).
• Describe the three primary functions of the healthy vocal cords.
• List the conditions or factors that may trigger VCD.
• Explain how to differentiate VCD from asthma.
• Develop a treatment plan for VCD that addresses both patient-specific VCD triggers and management of symptomatic episodes.
FACULTY
Linda S. MacConnell is an Assistant Professor in the Department of Physician Assistant Studies and Randy D. Danielsen is a Professor and Dean at the Arizona School of Health Sciences, AT Still University, Mesa. Ms. MacConnell is also a clinical PA affiliated with Enticare, an otolaryngology practice in Chandler, Arizona. Susan Symington is a clinical PA with the Arizona Asthma & Allergy Institute, with which Dr. Danielsen is also affiliated.
Linda MacConnell and Randy Danielsen have no significant financial relationships to disclose. Susan Symington is a member of the speaker’s bureau for Teva Respiratory and Thermo Fisher Scientific, Inc.
ACCREDITATION STATEMENT
This program has been reviewed and is approved for a maximum of 1.0 hour of American Academy of Physician Assistants (AAPA) Category 1 CME credit by the Physician Assistant Review Panel. [NPs: Both ANCC and the AANP Certification Program recognize AAPA as an approved provider of Category 1 credit.] Approval is valid for one year from the issue date of December 2014.
Article begins on next page >>
The symptoms of vocal cord dysfunction (VCD) can be mistaken for those
of asthma or other respiratory illnesses. As a result, VCD is often misdiagnosed,
leading to unnecessary, ineffective, costly, or even dangerous treatment. Here are
the facts that will enable you to avoid making an erroneous diagnosis, choosing
potentially harmful treatment, and delaying effective treatment.
A 33-year-old oncology nurse, JD, had moved from Seattle to Phoenix about six months earlier for a job opportunity. Shortly after starting her new job, she had developed intermittent dyspnea on exertion, with a cough lasting several minutes at a time, along with a sensation of heaviness over the larynx and a choking sensation. These symptoms were precipitated by gastroesophageal reflux disease (GERD), postnasal drainage, stress, and significant environmental change (ie, Seattle to Phoenix). She noticed that, since moving to Phoenix, she frequently cleared her throat but denied any hoarseness, dysphagia, chest tightness, chest pain, or wheezing. She noted nasal congestion and clear nasal discharge on exposure to inhaled irritants (eg, woodstove smoke) and strong fragrances (eg, perfume or cologne).
On physical examination, the patient was alert, oriented, and in no acute distress. She was coughing intermittently but was able to speak in complete sentences. No stridor or dyspnea was noted, either on exertion (jogging in place) or at rest.
HEENT examination was normal, with no scalp lesions or tenderness; face, symmetric; light reflex, symmetric; conjunctivae, clear; sclera white, without lesions or redness; pupils, equal, reactive to light and accommodation; tympanic membranes and canals, clear with intact landmarks; no nasal deformities; nasal mucosa, mildly erythematous with mild engorgement of the turbinates; no nasal polyps seen; nasal septum midline without perforation; no sinus tenderness on percussion; pharynx, clear without exudate; uvula rises on phonation; and oral mucosa and gingivae, pink without lesions. Neck was supple without masses or thyromegaly, and trachea was midline. Lungs were clear to auscultation with normal respiratory movement and no accessory muscle use, with normal anteroposterior diameter. Heart examination revealed regular rate and rhythm, without murmur, clicks, or gallops.
Examination of the skin was normal, without rashes, hives, swelling, petechiae, or significant ecchymosis. There was no palpable cervical, supraclavicular, or axillary adenopathy.
Results of laboratory studies included a normal complete blood count with differential and a normal IgE level of 46.3 IU/mL. Spirometry testing revealed normal values without obstruction; however, there was a flattening of the inspiratory flow loop, with no reversibility after bronchodilator, which was highly suggestive of vocal cord dysfunction (VCD). Perennial nonallergic rhinitis (formerly called vasomotor rhinitis) was confirmed because the patient experienced fewer symptoms to perfume after nasal corticosteroid use. The patient’s GERD was generally well controlled with esomeprazole but was likely a contributing factor to her vocal cord symptoms.
On laryngoscopy, abnormal vocal cord movement toward the midline during both inspiration and expiration was visualized, confirming the diagnosis of VCD.
BACKGROUND
VCD is a partial upper airway obstruction caused by paradoxical adduction (medial movement) of the vocal cords.1 Although it is primarily associated with inspiration, it sometimes manifests during expiration as well.
The true incidence of VCD is uncertain; different studies have found incidence rates varying from 2% to 27%, with higher rates in patients with asthma.1,2 However, highlighting the risk for misdiagnosis, some 10% of patients evaluated for asthma unresponsive to aggressive treatment were found, in fact, to have VCD alone.2
Similarly, although VCD is generally more common in women than in men, the reported female-to-male ratio has varied from 2:1 to 4:1.1,2,4 Some reports suggest that VCD is seen more frequently in younger women, with average ages at diagnosis of 14.5 in adolescents and 33 in adults.2,3 Others identify a broader age range, with most patients older than 50.4
Historically, VCD has been known by a variety of names and has been observed clinically since 1842. In that year, Dunglison referred to it as hysteric croup, describing a disorder of the laryngeal muscles brought on by “hysteria.”5 Later, Mackenzie was able to visualize adduction of the vocal cords during inspiration in patients with stridor by using a laryngoscope.6 Osler demonstrated his understanding of the condition in 1902, stating, “Spasm of muscles may occur with violent inspiratory effort and great distress, and may even lead to cyanosis. Extraordinary cries may be produced either inspiratory or expiratory.”7
More recently, in 1974, Patterson et al reported finding laryngoscopic evidence of VCD, which they termed Munchausen’s stridor.8 They used this descriptor to report on the case of a young woman with 15 hospital admissions for this condition. At the time, the etiology of the condition was believed to be largely psychologic, and its evaluation was consigned to psychiatrists and other mental health practitioners.
As laryngoscopy became more widely available in the 1970s and 1980s, diagnosis of VCD increased, although the condition remains underrecognized.9 Ibrahim et al suggest that primary care clinicians may not be as aware of VCD as they should be and may not consider laryngoscopy for possible VCD in patients whose asthma is poorly controlled.2
Disagreement persists with regard to the preferred name for the condition. Because numerous disorders involve abnormal vocal cord function, Christopher proposed moving away from the broad term VCD and toward a more descriptive term: paradoxical vocal fold motion (PVFM) disorder.10 Interestingly, use of the two terms seems to be divided along specialty lines: VCD is preferred by allergy, pulmonology, and mental health specialists, while PVFM is favored by otolaryngology specialists and speech-language pathologists.11
Further complicating awareness and recognition of VCD is its longstanding reputation as a psychologic disorder. In fact, the paradigm has shifted away from defining VCD as a purely psychopathologic entity to the identification of numerous functional etiologies for the disorder. This, however, has resulted in many new terms to describe the condition, including nonorganic upper airway obstruction, pseudoasthma, irritable larynx syndrome, factitious asthma, spasmodic croup, functional upper airway obstruction, episodic laryngeal dyskinesia, functional laryngeal obstruction, functional laryngeal stridor, and episodic paroxysmal laryngospasm.1
Regardless of its name, an understanding of VCD is essential for both primary care and specialty clinicians because of its frequent misdiagnosis as asthma, allergies, or severe upper airway obstruction. When it is misdiagnosed as asthma, aggressive asthma treatments—to which VCD does not respond—may be prescribed, including high-dose inhaled and systemic corticosteroids and bronchodilators. Patients may experience multiple emergency department (ED) visits and hospitalizations and, in some cases, may be subjected to tracheostomies and intubation.
Continue for vocal cord physiology and functions >>
VOCAL CORD PHYSIOLOGY AND FUNCTIONS
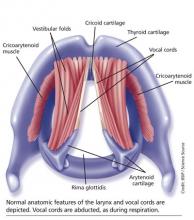
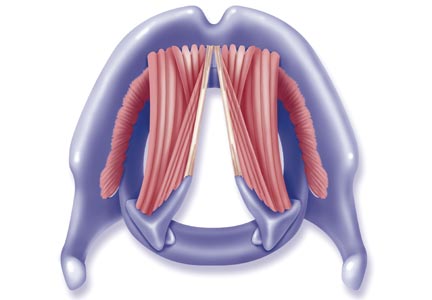
The vocal cords are located within the larynx. Abduction, or opening, of the cords is controlled by the posterior cricoarytenoid muscle; adduction, or closing, occurs via contraction of the lateral cricoarytenoid muscle. These muscles are innervated by the recurrent laryngeal nerve to control the width of the space—the rima glottidis—between the cords. During inspiration, the glottis opens; during expiration, it narrows but remains open.12
The vocal cords are involved in three main functions: protection of the airway, respiration, and phonation (vocal production). These functions are at least partially controlled involuntarily by brain stem reflexes; however, only airway protection—the most important of these functions—is reflexive and involuntary.12 Respiration may be controlled voluntarily, and phonation is primarily voluntary. Closure of the vocal cords is under the control of the laryngeal nerve branches of the vagal nerve.12,13
The vocal cords normally abduct during inspiration to allow air to pass through them into the trachea and the lungs. Sniffing, puffing, snuffling, and panting also cause the vocal cords to abduct. The vocal cords adduct with phonation (talking, singing), coughing, clearing the throat, performing the Valsalva maneuver, and swallowing. During expiration, 10% to 40% adduction is considered normal.14
VOCAL CORD DYSFUNCTION
Pathogenesis and etiology
VCD is a nonspecific term, and a number of factors may be involved in its development.15 Although the precise cause of VCD is unknown, it is believed to result from laryngeal hyperresponsiveness. This exaggerated responsiveness may be prompted by irritant and nonirritant triggers of the sensory receptors in the larynx, trachea, and large airways that mediate cough and glottis closure reflexes.16
VCD may be among a group of airway disorders triggered by occupational exposures, including irritants and psychologic stressors. For example, occupationally triggered VCD was diagnosed in rescue, recovery, and cleanup workers at the World Trade Center disaster site.4
A history of childhood sexual abuse has also been associated by some researchers with the development of VCD. For example, Freedman et al reported that, of 47 patients with VCD, 14 identified such a history and five were suspected of having been sexually abused as children.17
Paradoxical movement of the vocal cords causes them to close when they should open. (Click here for a video on normal and abnormal vocal cord movement.) VCD generally occurs during inspiration, causing obstruction of the incoming air through the larynx. Symptoms of VCD frequently include dyspnea, coughing, wheezing, hoarseness, and tightness or pain in the throat.
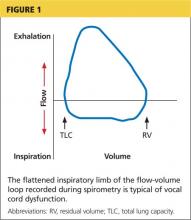
Examination of the flow-volume loops recorded when a patient experiences “wheezing” during spirometry testing reveals a flattened inspiratory loop, indicating a decrease of airflow into the lungs (see Figure 1).13,16 “Wheezing” is actually a misnomer in this situation because the term typically refers to sounds that occur during expiration.
Triggers
Physiologic, psychologic, and neurologic factors may all contribute to VCD.1,15 Conditions that can trigger VCD include
• Asthma
• Postnasal drip
• Recent upper respiratory illness (URI)
• Talking, singing
• Exercise
• Cough
• Voice strain
• Stress, anxiety, tension, elevated emotions
• Common irritants (eg, strong smells)
• Airborne irritants
• Rhinosinusitis
• GERD
• Use of certain medications
Identification of a particular patient’s triggers is key to successful management of VCD.
PATIENT PRESENTATION
Although there is no “typical” patient with VCD, the condition occurs more frequently in women, with the most common age at onset between 20 and 40 years. However, VCD has been seen in very young children and in adults as old as 83, and its diagnosis in the pediatric population is increasing.18
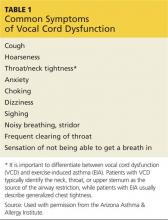
The patient may present with complaints of atypical chest pain, throat tightness, stridor, choking, difficult vocalization, cough and sometimes dysphagia, GERD, or rhinosinusitis (see Table 1). These signs and symptoms may occur without provocation, or patients may relate a history of triggers such as anxiety, irritant exposure, or exercise. In fact, about 14% of VCD is associated with exercise, particularly in young female athletes who experience shortness of breath and even stridor with exercise.19
A characteristic finding on physical examination is inspiratory stridor, along with respiratory distress.20 The stridor is best auscultated not over the anterior chest wall but over the tracheal area of the anterior neck.
Continue for differential diagnosis >>
DIFFERENTIAL DIAGNOSIS
Distinguishing VCD from other disorders can be challenging. Differential diagnosis should include
• Non–vocal cord adduction disorders, such as thyroid goiter, upper airway hemorrhage, caustic ingestion, neoplastic disorders, rheumatoid cricoarytenoid arthritis, pharyngeal abscess, angioedema, pulmonary embolus21
• Anatomic defects (eg, laryngomalacia, subglottic stenosis)
• Tracheal masses (eg, enlarged thyroid gland)
• Vocal cord polyps
• Laryngospasm
• Vocal cord paresis
• Neurologic causes (eg, brain stem compression, severe cortical injury, nuclear or lower motor neuron injury, movement disorders)
• Nonorganic causes (eg, factitious symptoms or malingering; conversion disorder)22
• Reactive airway disease.
Some disorders are easier to distinguish from VCD than others. For example, although laryngospasm may produce similar symptoms, episodes are brief, lasting seconds to minutes; VCD episodes may last hours to days.
Asthma
Even the most astute clinician will be unable to obtain adequate information from the patient history to differentiate VCD from asthma. There is a significant overlap of symptoms—shortness of breath, cough, wheezing—and frequently, the diseases coexist. History is often negative for chest pain, but it is common for patients with VCD, when asked to describe their symptoms, to report chest tightness. The clinician therefore needs to ask the patient to point to where the tightness is felt—in the chest or in the neck over the laryngeal area—to distinguish the source.
Asthma symptoms usually increase over a few hours, days, or weeks but respond to medications that open the airway and reduce inflammation (inhaled β-agonists and corticosteroids). VCD symptoms usually occur or decrease suddenly and do not respond well to traditional asthma treatments.
Other differences between asthma and VCD symptoms include voice changes and time of day when symptoms occur. The person with VCD will experience voice changes, such as hoarseness, as well as prolonged coughing episodes. Patients with asthma may awaken at night because of breathlessness, while most patients with VCD experience symptoms only during the day.
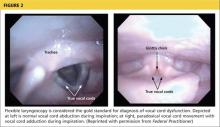
The diagnosis is generally confirmed if VCD is seen on direct laryngoscopic visualization during a symptomatic episode. In terms of adduction, the anterior cords will appear normal, but the posterior portion of the cords will display the classic “glottis chink” (see Figure 2).9
If the diagnosis is in question, videostroboscopy, a technique that provides a magnified slow-motion view of vocal cord vibration, can help identify or exclude pathologic conditions of the vocal cords.23
Convincing the patient of the validity of the diagnosis may be problematic if the patient has been previously diagnosed with and treated for another condition. The diagnosis should be explained and the patient counseled what appropriate care for VCD entails (see discussion under “Patient education and self-care”).
TREATMENT
Acute episode
During an acute VCD episode, offering the patient calm reassurance can be effective in resolving the episode. Simple breathing guidance may also be beneficial; instructing the patient to breathe rapidly and shallowly (ie, pant) can result in immediate resolution of symptoms.24 The patient can be advised to utilize other techniques, such as diaphragmatic breathing, breathing through the nose, breathing through a straw, pursed-lip breathing, and exhaling with a hissing sound.25
Long-term management
Although various strategies are employed in the management of VCD, well-designed studies on which to base treatment decisions have not been performed. Of course, control and management of possible underlying triggers or disorders should be implemented. Because etiology is rarely known, treatment for VCD is generally empiric.
Evidence does exist, however, to suggest that voice therapy, the treatment of choice for muscle tension dysphonia, is also effective for VCD. Speech therapy with specific voice and breathing exercises can enable the patient to manage the condition, thereby reducing ED visits, hospitalizations, and treatment costs.26
Patient education and self-care
Patient education is a critical component of VCD management. The clinician should explain the functions of the larynx to the patient, including the normal functioning of the vocal cords during respiration, speaking, swallowing, coughing, throat clearing, and breath holding. It may also enhance patients’ understanding of VCD to view their diagnostic laryngoscopy or videostroboscopy films.21
The patient should be advised to rest the voice, hydrate, utilize sialagogues (lozenges, gum) to stimulate salivation, reduce exposure to triggers when possible, and decrease stress. She should be encouraged to track VCD triggers by documenting what she is doing, where, and when, at the time of a VCD episode.
Two exercises—“paused breathing” and “belly breathing”—can be used by patients to learn how to relax the vocal cords (see “Patient Handout”). Patients should practice these exercises three times a day so that they can be easily recalled and performed during VCD episodes.
Continue for outcomes >>
OUTCOMES
Little is known about long-term outcomes for patients with VCD. The current literature consists of poorly described and conflicting case reports and results of small trials. Although documentation is lacking, the authors agree that, by educating the patient about the diagnosis, teaching effective VCD management strategies, and referring patients for voice therapy, clinicians can help patients achieve signicant improvement. Further investigation is needed to enhance our knowledge of the causes of VCD and to research additional diagnostic modalities and treatments.2
CASE PATIENT
After diagnosing VCD, the clinician explained the normal functioning of the vocal cords and how certain factors may cause them to close during inspiration. The patient then understood why bronchodilator therapy had failed to relieve her symptoms. She was counseled to continue her inhaled nasal steroid and proton pump inhibitor for her perennial nonallergic rhinitis and GERD, respectively, because these conditions may trigger her VCD, and to take steps to manage her stress. She learned breathing techniques to alleviate acute episodes of VCD and was informed of the option of voice therapy with a speech therapist if needed.
At six-week follow-up, the patient reported that she was complying with her medication regimen, had made an effort to relax more, and had experienced no acute attacks of VCD since her last visit.
CONCLUSION
Patients with symptoms suggestive of VCD require a thorough evaluation, including laryngoscopic examination, to ensure accurate diagnosis and avoid a too-common misdiagnosis. Primary care clinicians should know about VCD and, if not trained in the performance of flexible laryngoscopy, should refer the symptomatic patient to a specialist for appropriate work-up.
1. Hoyte FCL. Vocal cord dysfunction. Immunol Allergy Clin N Am. 2013;33:1-22.
2. Ibrahim WH, Gheriani HA, Almohamed AA, Raza T. Paradoxical vocal cord motion disorder: past, present and future. Postgrad Med J. 2007;83:164-172.
3. Powell DM, Karanfilov BI, Beechler KB, et al. Paradoxical vocal cord dysfunction in juveniles. Arch Otolaryngol Head Neck Surg. 2000;126(1):29-34.
4. Husein OF, Husein TN, Gardner R, et al. Formal psychological testing in patients with paradoxical vocal fold dysfunction. Laryngoscope. 2008; 118(4):740-747.
5. Dunglison RD. The Practice of Medicine. Philadelphia, PA: Lea and Blanchard; 1842:257-258.
6. MacKenzie M. Use of Laryngoscopy in Diseases of the Throat. Philadelphia, PA: Lindsey and Blackeston; 1869:246-250.
7. Osler W. Hysteria. In: The Principles and Practice of Medicine. 4th ed. New York, NY: Appleton; 1902:1111-1112.
8. Patterson R, Schatz M, Horton M. Munchausen’s stridor: non-organic laryngeal obstruction. Clin Allergy. 1974;4:307-310.
9. Christopher KL, Wood RP 2nd, Eckert RC, et al. Vocal cord dysfunction presenting as asthma. N Engl J Med. 1983;308(26):1566-1570.
10. Christopher KL. Understanding vocal cord dysfunction: a step in the right direction with a long road ahead. Chest. 2006;129(4):842-843.
11. Christopher KL, Morris MJ. Vocal cord dysfunction, paradoxic vocal fold motion, or laryngomalacia? Our understanding requires an interdisciplinary approach. Otolaryngol Clin N Am. 2010;43:43-66.
12. Sasaki CT, Weaver EM. Physiology of the larynx. Am J Med. 1997;103:9S-18S.
13. Balkissoon R. Occupational upper airway disease. Clin Chest Med. 2002;23:717-725.
14. Murakami Y, Kirschner JA. Mechanical and physiological properties of reflex laryngeal closure. Ann Otol Rhinol Laryngol. 1972;81(1):59-71.
15. Forrest LA, Husein T, Husein O. Paradoxical vocal cord motion disorder: classification and treatment. Laryngoscope. 2012;122:844-853.
16. Altman KW, Simpson CB, Amin MR, et al. Cough and paradoxical vocal fold motion. Otolaryngol Head Neck Surg. 2002;127(6):501-511.
17. Freedman MR, Rosenberg SJ, Schmaling KB. Childhood sexual abuse in patients with paradoxical vocal cord dysfunction. J Nerv Ment Dis. 1991;179(5):295-298.
18. Buddiga P. Vocal cord dysfunction. Medscape. http://emedicine.medscape.com/article/137782-overview. Accessed November 12, 2014.
19. Chiang T, Marcinow AM, deSilva BW, et al. Exercise-induced paradoxical vocal fold motion disorder: diagnosis and management. Laryngoscope. 2013;123:727-731.
20. Morris MJ, Deal LE, Bean DR, et al. Vocal cord dysfunction in patients with exertional dyspnea. Chest. 1999;116(6):1676-1682.
21. Hicks M, Brugman SM, Katial R. Vocal cord dysfunction/paradoxical vocal fold motion. Prim Care. 2008;35(1):81-103.
22. Maschka DA, Bauman NM, McCray PB, et al. A classification scheme for paradoxical vocal fold motion. Laryngoscope. 1997;107(11):1429-1435.
23. Uloza V, Vegiene A, Pribuisiene R, Saferis V. Quantitative evaluation of video laryngostroboscopy: reliability of the basic parameters. J Voice. 2013;27(3):361-368.
24. Pitchenik AF. Functional laryngeal obstruction relieved by panting. Chest. 1991;100(5):1465-1467.
25. Deckert J, Deckert L. Vocal cord dysfunction. Am Fam Physician. 2010;81(2):156-160.
26. Carding PN, Horsley IA, Docherty GJ. A study of the effectiveness of voice therapy in the treatment of 45 patients with nonorganic dysphonia. J Voice. 1999;13(1):72-104.
CE/CME No: CR-1412
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest and evaluation. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
• Discuss the evolution in thinking about the pathogenesis of and treatment for vocal cord dysfunction (VCD).
• Describe the three primary functions of the healthy vocal cords.
• List the conditions or factors that may trigger VCD.
• Explain how to differentiate VCD from asthma.
• Develop a treatment plan for VCD that addresses both patient-specific VCD triggers and management of symptomatic episodes.
FACULTY
Linda S. MacConnell is an Assistant Professor in the Department of Physician Assistant Studies and Randy D. Danielsen is a Professor and Dean at the Arizona School of Health Sciences, AT Still University, Mesa. Ms. MacConnell is also a clinical PA affiliated with Enticare, an otolaryngology practice in Chandler, Arizona. Susan Symington is a clinical PA with the Arizona Asthma & Allergy Institute, with which Dr. Danielsen is also affiliated.
Linda MacConnell and Randy Danielsen have no significant financial relationships to disclose. Susan Symington is a member of the speaker’s bureau for Teva Respiratory and Thermo Fisher Scientific, Inc.
ACCREDITATION STATEMENT
This program has been reviewed and is approved for a maximum of 1.0 hour of American Academy of Physician Assistants (AAPA) Category 1 CME credit by the Physician Assistant Review Panel. [NPs: Both ANCC and the AANP Certification Program recognize AAPA as an approved provider of Category 1 credit.] Approval is valid for one year from the issue date of December 2014.
Article begins on next page >>
The symptoms of vocal cord dysfunction (VCD) can be mistaken for those
of asthma or other respiratory illnesses. As a result, VCD is often misdiagnosed,
leading to unnecessary, ineffective, costly, or even dangerous treatment. Here are
the facts that will enable you to avoid making an erroneous diagnosis, choosing
potentially harmful treatment, and delaying effective treatment.
A 33-year-old oncology nurse, JD, had moved from Seattle to Phoenix about six months earlier for a job opportunity. Shortly after starting her new job, she had developed intermittent dyspnea on exertion, with a cough lasting several minutes at a time, along with a sensation of heaviness over the larynx and a choking sensation. These symptoms were precipitated by gastroesophageal reflux disease (GERD), postnasal drainage, stress, and significant environmental change (ie, Seattle to Phoenix). She noticed that, since moving to Phoenix, she frequently cleared her throat but denied any hoarseness, dysphagia, chest tightness, chest pain, or wheezing. She noted nasal congestion and clear nasal discharge on exposure to inhaled irritants (eg, woodstove smoke) and strong fragrances (eg, perfume or cologne).
On physical examination, the patient was alert, oriented, and in no acute distress. She was coughing intermittently but was able to speak in complete sentences. No stridor or dyspnea was noted, either on exertion (jogging in place) or at rest.
HEENT examination was normal, with no scalp lesions or tenderness; face, symmetric; light reflex, symmetric; conjunctivae, clear; sclera white, without lesions or redness; pupils, equal, reactive to light and accommodation; tympanic membranes and canals, clear with intact landmarks; no nasal deformities; nasal mucosa, mildly erythematous with mild engorgement of the turbinates; no nasal polyps seen; nasal septum midline without perforation; no sinus tenderness on percussion; pharynx, clear without exudate; uvula rises on phonation; and oral mucosa and gingivae, pink without lesions. Neck was supple without masses or thyromegaly, and trachea was midline. Lungs were clear to auscultation with normal respiratory movement and no accessory muscle use, with normal anteroposterior diameter. Heart examination revealed regular rate and rhythm, without murmur, clicks, or gallops.
Examination of the skin was normal, without rashes, hives, swelling, petechiae, or significant ecchymosis. There was no palpable cervical, supraclavicular, or axillary adenopathy.
Results of laboratory studies included a normal complete blood count with differential and a normal IgE level of 46.3 IU/mL. Spirometry testing revealed normal values without obstruction; however, there was a flattening of the inspiratory flow loop, with no reversibility after bronchodilator, which was highly suggestive of vocal cord dysfunction (VCD). Perennial nonallergic rhinitis (formerly called vasomotor rhinitis) was confirmed because the patient experienced fewer symptoms to perfume after nasal corticosteroid use. The patient’s GERD was generally well controlled with esomeprazole but was likely a contributing factor to her vocal cord symptoms.
On laryngoscopy, abnormal vocal cord movement toward the midline during both inspiration and expiration was visualized, confirming the diagnosis of VCD.
BACKGROUND
VCD is a partial upper airway obstruction caused by paradoxical adduction (medial movement) of the vocal cords.1 Although it is primarily associated with inspiration, it sometimes manifests during expiration as well.
The true incidence of VCD is uncertain; different studies have found incidence rates varying from 2% to 27%, with higher rates in patients with asthma.1,2 However, highlighting the risk for misdiagnosis, some 10% of patients evaluated for asthma unresponsive to aggressive treatment were found, in fact, to have VCD alone.2
Similarly, although VCD is generally more common in women than in men, the reported female-to-male ratio has varied from 2:1 to 4:1.1,2,4 Some reports suggest that VCD is seen more frequently in younger women, with average ages at diagnosis of 14.5 in adolescents and 33 in adults.2,3 Others identify a broader age range, with most patients older than 50.4
Historically, VCD has been known by a variety of names and has been observed clinically since 1842. In that year, Dunglison referred to it as hysteric croup, describing a disorder of the laryngeal muscles brought on by “hysteria.”5 Later, Mackenzie was able to visualize adduction of the vocal cords during inspiration in patients with stridor by using a laryngoscope.6 Osler demonstrated his understanding of the condition in 1902, stating, “Spasm of muscles may occur with violent inspiratory effort and great distress, and may even lead to cyanosis. Extraordinary cries may be produced either inspiratory or expiratory.”7
More recently, in 1974, Patterson et al reported finding laryngoscopic evidence of VCD, which they termed Munchausen’s stridor.8 They used this descriptor to report on the case of a young woman with 15 hospital admissions for this condition. At the time, the etiology of the condition was believed to be largely psychologic, and its evaluation was consigned to psychiatrists and other mental health practitioners.
As laryngoscopy became more widely available in the 1970s and 1980s, diagnosis of VCD increased, although the condition remains underrecognized.9 Ibrahim et al suggest that primary care clinicians may not be as aware of VCD as they should be and may not consider laryngoscopy for possible VCD in patients whose asthma is poorly controlled.2
Disagreement persists with regard to the preferred name for the condition. Because numerous disorders involve abnormal vocal cord function, Christopher proposed moving away from the broad term VCD and toward a more descriptive term: paradoxical vocal fold motion (PVFM) disorder.10 Interestingly, use of the two terms seems to be divided along specialty lines: VCD is preferred by allergy, pulmonology, and mental health specialists, while PVFM is favored by otolaryngology specialists and speech-language pathologists.11
Further complicating awareness and recognition of VCD is its longstanding reputation as a psychologic disorder. In fact, the paradigm has shifted away from defining VCD as a purely psychopathologic entity to the identification of numerous functional etiologies for the disorder. This, however, has resulted in many new terms to describe the condition, including nonorganic upper airway obstruction, pseudoasthma, irritable larynx syndrome, factitious asthma, spasmodic croup, functional upper airway obstruction, episodic laryngeal dyskinesia, functional laryngeal obstruction, functional laryngeal stridor, and episodic paroxysmal laryngospasm.1
Regardless of its name, an understanding of VCD is essential for both primary care and specialty clinicians because of its frequent misdiagnosis as asthma, allergies, or severe upper airway obstruction. When it is misdiagnosed as asthma, aggressive asthma treatments—to which VCD does not respond—may be prescribed, including high-dose inhaled and systemic corticosteroids and bronchodilators. Patients may experience multiple emergency department (ED) visits and hospitalizations and, in some cases, may be subjected to tracheostomies and intubation.
Continue for vocal cord physiology and functions >>
VOCAL CORD PHYSIOLOGY AND FUNCTIONS
The vocal cords are located within the larynx. Abduction, or opening, of the cords is controlled by the posterior cricoarytenoid muscle; adduction, or closing, occurs via contraction of the lateral cricoarytenoid muscle. These muscles are innervated by the recurrent laryngeal nerve to control the width of the space—the rima glottidis—between the cords. During inspiration, the glottis opens; during expiration, it narrows but remains open.12
The vocal cords are involved in three main functions: protection of the airway, respiration, and phonation (vocal production). These functions are at least partially controlled involuntarily by brain stem reflexes; however, only airway protection—the most important of these functions—is reflexive and involuntary.12 Respiration may be controlled voluntarily, and phonation is primarily voluntary. Closure of the vocal cords is under the control of the laryngeal nerve branches of the vagal nerve.12,13
The vocal cords normally abduct during inspiration to allow air to pass through them into the trachea and the lungs. Sniffing, puffing, snuffling, and panting also cause the vocal cords to abduct. The vocal cords adduct with phonation (talking, singing), coughing, clearing the throat, performing the Valsalva maneuver, and swallowing. During expiration, 10% to 40% adduction is considered normal.14
VOCAL CORD DYSFUNCTION
Pathogenesis and etiology
VCD is a nonspecific term, and a number of factors may be involved in its development.15 Although the precise cause of VCD is unknown, it is believed to result from laryngeal hyperresponsiveness. This exaggerated responsiveness may be prompted by irritant and nonirritant triggers of the sensory receptors in the larynx, trachea, and large airways that mediate cough and glottis closure reflexes.16
VCD may be among a group of airway disorders triggered by occupational exposures, including irritants and psychologic stressors. For example, occupationally triggered VCD was diagnosed in rescue, recovery, and cleanup workers at the World Trade Center disaster site.4
A history of childhood sexual abuse has also been associated by some researchers with the development of VCD. For example, Freedman et al reported that, of 47 patients with VCD, 14 identified such a history and five were suspected of having been sexually abused as children.17
Paradoxical movement of the vocal cords causes them to close when they should open. (Click here for a video on normal and abnormal vocal cord movement.) VCD generally occurs during inspiration, causing obstruction of the incoming air through the larynx. Symptoms of VCD frequently include dyspnea, coughing, wheezing, hoarseness, and tightness or pain in the throat.
Examination of the flow-volume loops recorded when a patient experiences “wheezing” during spirometry testing reveals a flattened inspiratory loop, indicating a decrease of airflow into the lungs (see Figure 1).13,16 “Wheezing” is actually a misnomer in this situation because the term typically refers to sounds that occur during expiration.
Triggers
Physiologic, psychologic, and neurologic factors may all contribute to VCD.1,15 Conditions that can trigger VCD include
• Asthma
• Postnasal drip
• Recent upper respiratory illness (URI)
• Talking, singing
• Exercise
• Cough
• Voice strain
• Stress, anxiety, tension, elevated emotions
• Common irritants (eg, strong smells)
• Airborne irritants
• Rhinosinusitis
• GERD
• Use of certain medications
Identification of a particular patient’s triggers is key to successful management of VCD.
PATIENT PRESENTATION
Although there is no “typical” patient with VCD, the condition occurs more frequently in women, with the most common age at onset between 20 and 40 years. However, VCD has been seen in very young children and in adults as old as 83, and its diagnosis in the pediatric population is increasing.18
The patient may present with complaints of atypical chest pain, throat tightness, stridor, choking, difficult vocalization, cough and sometimes dysphagia, GERD, or rhinosinusitis (see Table 1). These signs and symptoms may occur without provocation, or patients may relate a history of triggers such as anxiety, irritant exposure, or exercise. In fact, about 14% of VCD is associated with exercise, particularly in young female athletes who experience shortness of breath and even stridor with exercise.19
A characteristic finding on physical examination is inspiratory stridor, along with respiratory distress.20 The stridor is best auscultated not over the anterior chest wall but over the tracheal area of the anterior neck.
Continue for differential diagnosis >>
DIFFERENTIAL DIAGNOSIS
Distinguishing VCD from other disorders can be challenging. Differential diagnosis should include
• Non–vocal cord adduction disorders, such as thyroid goiter, upper airway hemorrhage, caustic ingestion, neoplastic disorders, rheumatoid cricoarytenoid arthritis, pharyngeal abscess, angioedema, pulmonary embolus21
• Anatomic defects (eg, laryngomalacia, subglottic stenosis)
• Tracheal masses (eg, enlarged thyroid gland)
• Vocal cord polyps
• Laryngospasm
• Vocal cord paresis
• Neurologic causes (eg, brain stem compression, severe cortical injury, nuclear or lower motor neuron injury, movement disorders)
• Nonorganic causes (eg, factitious symptoms or malingering; conversion disorder)22
• Reactive airway disease.
Some disorders are easier to distinguish from VCD than others. For example, although laryngospasm may produce similar symptoms, episodes are brief, lasting seconds to minutes; VCD episodes may last hours to days.
Asthma
Even the most astute clinician will be unable to obtain adequate information from the patient history to differentiate VCD from asthma. There is a significant overlap of symptoms—shortness of breath, cough, wheezing—and frequently, the diseases coexist. History is often negative for chest pain, but it is common for patients with VCD, when asked to describe their symptoms, to report chest tightness. The clinician therefore needs to ask the patient to point to where the tightness is felt—in the chest or in the neck over the laryngeal area—to distinguish the source.
Asthma symptoms usually increase over a few hours, days, or weeks but respond to medications that open the airway and reduce inflammation (inhaled β-agonists and corticosteroids). VCD symptoms usually occur or decrease suddenly and do not respond well to traditional asthma treatments.
Other differences between asthma and VCD symptoms include voice changes and time of day when symptoms occur. The person with VCD will experience voice changes, such as hoarseness, as well as prolonged coughing episodes. Patients with asthma may awaken at night because of breathlessness, while most patients with VCD experience symptoms only during the day.
The diagnosis is generally confirmed if VCD is seen on direct laryngoscopic visualization during a symptomatic episode. In terms of adduction, the anterior cords will appear normal, but the posterior portion of the cords will display the classic “glottis chink” (see Figure 2).9
If the diagnosis is in question, videostroboscopy, a technique that provides a magnified slow-motion view of vocal cord vibration, can help identify or exclude pathologic conditions of the vocal cords.23
Convincing the patient of the validity of the diagnosis may be problematic if the patient has been previously diagnosed with and treated for another condition. The diagnosis should be explained and the patient counseled what appropriate care for VCD entails (see discussion under “Patient education and self-care”).
TREATMENT
Acute episode
During an acute VCD episode, offering the patient calm reassurance can be effective in resolving the episode. Simple breathing guidance may also be beneficial; instructing the patient to breathe rapidly and shallowly (ie, pant) can result in immediate resolution of symptoms.24 The patient can be advised to utilize other techniques, such as diaphragmatic breathing, breathing through the nose, breathing through a straw, pursed-lip breathing, and exhaling with a hissing sound.25
Long-term management
Although various strategies are employed in the management of VCD, well-designed studies on which to base treatment decisions have not been performed. Of course, control and management of possible underlying triggers or disorders should be implemented. Because etiology is rarely known, treatment for VCD is generally empiric.
Evidence does exist, however, to suggest that voice therapy, the treatment of choice for muscle tension dysphonia, is also effective for VCD. Speech therapy with specific voice and breathing exercises can enable the patient to manage the condition, thereby reducing ED visits, hospitalizations, and treatment costs.26
Patient education and self-care
Patient education is a critical component of VCD management. The clinician should explain the functions of the larynx to the patient, including the normal functioning of the vocal cords during respiration, speaking, swallowing, coughing, throat clearing, and breath holding. It may also enhance patients’ understanding of VCD to view their diagnostic laryngoscopy or videostroboscopy films.21
The patient should be advised to rest the voice, hydrate, utilize sialagogues (lozenges, gum) to stimulate salivation, reduce exposure to triggers when possible, and decrease stress. She should be encouraged to track VCD triggers by documenting what she is doing, where, and when, at the time of a VCD episode.
Two exercises—“paused breathing” and “belly breathing”—can be used by patients to learn how to relax the vocal cords (see “Patient Handout”). Patients should practice these exercises three times a day so that they can be easily recalled and performed during VCD episodes.
Continue for outcomes >>
OUTCOMES
Little is known about long-term outcomes for patients with VCD. The current literature consists of poorly described and conflicting case reports and results of small trials. Although documentation is lacking, the authors agree that, by educating the patient about the diagnosis, teaching effective VCD management strategies, and referring patients for voice therapy, clinicians can help patients achieve signicant improvement. Further investigation is needed to enhance our knowledge of the causes of VCD and to research additional diagnostic modalities and treatments.2
CASE PATIENT
After diagnosing VCD, the clinician explained the normal functioning of the vocal cords and how certain factors may cause them to close during inspiration. The patient then understood why bronchodilator therapy had failed to relieve her symptoms. She was counseled to continue her inhaled nasal steroid and proton pump inhibitor for her perennial nonallergic rhinitis and GERD, respectively, because these conditions may trigger her VCD, and to take steps to manage her stress. She learned breathing techniques to alleviate acute episodes of VCD and was informed of the option of voice therapy with a speech therapist if needed.
At six-week follow-up, the patient reported that she was complying with her medication regimen, had made an effort to relax more, and had experienced no acute attacks of VCD since her last visit.
CONCLUSION
Patients with symptoms suggestive of VCD require a thorough evaluation, including laryngoscopic examination, to ensure accurate diagnosis and avoid a too-common misdiagnosis. Primary care clinicians should know about VCD and, if not trained in the performance of flexible laryngoscopy, should refer the symptomatic patient to a specialist for appropriate work-up.
CE/CME No: CR-1412
PROGRAM OVERVIEW
Earn credit by reading this article and successfully completing the posttest and evaluation. Successful completion is defined as a cumulative score of at least 70% correct.
EDUCATIONAL OBJECTIVES
• Discuss the evolution in thinking about the pathogenesis of and treatment for vocal cord dysfunction (VCD).
• Describe the three primary functions of the healthy vocal cords.
• List the conditions or factors that may trigger VCD.
• Explain how to differentiate VCD from asthma.
• Develop a treatment plan for VCD that addresses both patient-specific VCD triggers and management of symptomatic episodes.
FACULTY
Linda S. MacConnell is an Assistant Professor in the Department of Physician Assistant Studies and Randy D. Danielsen is a Professor and Dean at the Arizona School of Health Sciences, AT Still University, Mesa. Ms. MacConnell is also a clinical PA affiliated with Enticare, an otolaryngology practice in Chandler, Arizona. Susan Symington is a clinical PA with the Arizona Asthma & Allergy Institute, with which Dr. Danielsen is also affiliated.
Linda MacConnell and Randy Danielsen have no significant financial relationships to disclose. Susan Symington is a member of the speaker’s bureau for Teva Respiratory and Thermo Fisher Scientific, Inc.
ACCREDITATION STATEMENT
This program has been reviewed and is approved for a maximum of 1.0 hour of American Academy of Physician Assistants (AAPA) Category 1 CME credit by the Physician Assistant Review Panel. [NPs: Both ANCC and the AANP Certification Program recognize AAPA as an approved provider of Category 1 credit.] Approval is valid for one year from the issue date of December 2014.
Article begins on next page >>
The symptoms of vocal cord dysfunction (VCD) can be mistaken for those
of asthma or other respiratory illnesses. As a result, VCD is often misdiagnosed,
leading to unnecessary, ineffective, costly, or even dangerous treatment. Here are
the facts that will enable you to avoid making an erroneous diagnosis, choosing
potentially harmful treatment, and delaying effective treatment.
A 33-year-old oncology nurse, JD, had moved from Seattle to Phoenix about six months earlier for a job opportunity. Shortly after starting her new job, she had developed intermittent dyspnea on exertion, with a cough lasting several minutes at a time, along with a sensation of heaviness over the larynx and a choking sensation. These symptoms were precipitated by gastroesophageal reflux disease (GERD), postnasal drainage, stress, and significant environmental change (ie, Seattle to Phoenix). She noticed that, since moving to Phoenix, she frequently cleared her throat but denied any hoarseness, dysphagia, chest tightness, chest pain, or wheezing. She noted nasal congestion and clear nasal discharge on exposure to inhaled irritants (eg, woodstove smoke) and strong fragrances (eg, perfume or cologne).
On physical examination, the patient was alert, oriented, and in no acute distress. She was coughing intermittently but was able to speak in complete sentences. No stridor or dyspnea was noted, either on exertion (jogging in place) or at rest.
HEENT examination was normal, with no scalp lesions or tenderness; face, symmetric; light reflex, symmetric; conjunctivae, clear; sclera white, without lesions or redness; pupils, equal, reactive to light and accommodation; tympanic membranes and canals, clear with intact landmarks; no nasal deformities; nasal mucosa, mildly erythematous with mild engorgement of the turbinates; no nasal polyps seen; nasal septum midline without perforation; no sinus tenderness on percussion; pharynx, clear without exudate; uvula rises on phonation; and oral mucosa and gingivae, pink without lesions. Neck was supple without masses or thyromegaly, and trachea was midline. Lungs were clear to auscultation with normal respiratory movement and no accessory muscle use, with normal anteroposterior diameter. Heart examination revealed regular rate and rhythm, without murmur, clicks, or gallops.
Examination of the skin was normal, without rashes, hives, swelling, petechiae, or significant ecchymosis. There was no palpable cervical, supraclavicular, or axillary adenopathy.
Results of laboratory studies included a normal complete blood count with differential and a normal IgE level of 46.3 IU/mL. Spirometry testing revealed normal values without obstruction; however, there was a flattening of the inspiratory flow loop, with no reversibility after bronchodilator, which was highly suggestive of vocal cord dysfunction (VCD). Perennial nonallergic rhinitis (formerly called vasomotor rhinitis) was confirmed because the patient experienced fewer symptoms to perfume after nasal corticosteroid use. The patient’s GERD was generally well controlled with esomeprazole but was likely a contributing factor to her vocal cord symptoms.
On laryngoscopy, abnormal vocal cord movement toward the midline during both inspiration and expiration was visualized, confirming the diagnosis of VCD.
BACKGROUND
VCD is a partial upper airway obstruction caused by paradoxical adduction (medial movement) of the vocal cords.1 Although it is primarily associated with inspiration, it sometimes manifests during expiration as well.
The true incidence of VCD is uncertain; different studies have found incidence rates varying from 2% to 27%, with higher rates in patients with asthma.1,2 However, highlighting the risk for misdiagnosis, some 10% of patients evaluated for asthma unresponsive to aggressive treatment were found, in fact, to have VCD alone.2
Similarly, although VCD is generally more common in women than in men, the reported female-to-male ratio has varied from 2:1 to 4:1.1,2,4 Some reports suggest that VCD is seen more frequently in younger women, with average ages at diagnosis of 14.5 in adolescents and 33 in adults.2,3 Others identify a broader age range, with most patients older than 50.4
Historically, VCD has been known by a variety of names and has been observed clinically since 1842. In that year, Dunglison referred to it as hysteric croup, describing a disorder of the laryngeal muscles brought on by “hysteria.”5 Later, Mackenzie was able to visualize adduction of the vocal cords during inspiration in patients with stridor by using a laryngoscope.6 Osler demonstrated his understanding of the condition in 1902, stating, “Spasm of muscles may occur with violent inspiratory effort and great distress, and may even lead to cyanosis. Extraordinary cries may be produced either inspiratory or expiratory.”7
More recently, in 1974, Patterson et al reported finding laryngoscopic evidence of VCD, which they termed Munchausen’s stridor.8 They used this descriptor to report on the case of a young woman with 15 hospital admissions for this condition. At the time, the etiology of the condition was believed to be largely psychologic, and its evaluation was consigned to psychiatrists and other mental health practitioners.
As laryngoscopy became more widely available in the 1970s and 1980s, diagnosis of VCD increased, although the condition remains underrecognized.9 Ibrahim et al suggest that primary care clinicians may not be as aware of VCD as they should be and may not consider laryngoscopy for possible VCD in patients whose asthma is poorly controlled.2
Disagreement persists with regard to the preferred name for the condition. Because numerous disorders involve abnormal vocal cord function, Christopher proposed moving away from the broad term VCD and toward a more descriptive term: paradoxical vocal fold motion (PVFM) disorder.10 Interestingly, use of the two terms seems to be divided along specialty lines: VCD is preferred by allergy, pulmonology, and mental health specialists, while PVFM is favored by otolaryngology specialists and speech-language pathologists.11
Further complicating awareness and recognition of VCD is its longstanding reputation as a psychologic disorder. In fact, the paradigm has shifted away from defining VCD as a purely psychopathologic entity to the identification of numerous functional etiologies for the disorder. This, however, has resulted in many new terms to describe the condition, including nonorganic upper airway obstruction, pseudoasthma, irritable larynx syndrome, factitious asthma, spasmodic croup, functional upper airway obstruction, episodic laryngeal dyskinesia, functional laryngeal obstruction, functional laryngeal stridor, and episodic paroxysmal laryngospasm.1
Regardless of its name, an understanding of VCD is essential for both primary care and specialty clinicians because of its frequent misdiagnosis as asthma, allergies, or severe upper airway obstruction. When it is misdiagnosed as asthma, aggressive asthma treatments—to which VCD does not respond—may be prescribed, including high-dose inhaled and systemic corticosteroids and bronchodilators. Patients may experience multiple emergency department (ED) visits and hospitalizations and, in some cases, may be subjected to tracheostomies and intubation.
Continue for vocal cord physiology and functions >>
VOCAL CORD PHYSIOLOGY AND FUNCTIONS
The vocal cords are located within the larynx. Abduction, or opening, of the cords is controlled by the posterior cricoarytenoid muscle; adduction, or closing, occurs via contraction of the lateral cricoarytenoid muscle. These muscles are innervated by the recurrent laryngeal nerve to control the width of the space—the rima glottidis—between the cords. During inspiration, the glottis opens; during expiration, it narrows but remains open.12
The vocal cords are involved in three main functions: protection of the airway, respiration, and phonation (vocal production). These functions are at least partially controlled involuntarily by brain stem reflexes; however, only airway protection—the most important of these functions—is reflexive and involuntary.12 Respiration may be controlled voluntarily, and phonation is primarily voluntary. Closure of the vocal cords is under the control of the laryngeal nerve branches of the vagal nerve.12,13
The vocal cords normally abduct during inspiration to allow air to pass through them into the trachea and the lungs. Sniffing, puffing, snuffling, and panting also cause the vocal cords to abduct. The vocal cords adduct with phonation (talking, singing), coughing, clearing the throat, performing the Valsalva maneuver, and swallowing. During expiration, 10% to 40% adduction is considered normal.14
VOCAL CORD DYSFUNCTION
Pathogenesis and etiology
VCD is a nonspecific term, and a number of factors may be involved in its development.15 Although the precise cause of VCD is unknown, it is believed to result from laryngeal hyperresponsiveness. This exaggerated responsiveness may be prompted by irritant and nonirritant triggers of the sensory receptors in the larynx, trachea, and large airways that mediate cough and glottis closure reflexes.16
VCD may be among a group of airway disorders triggered by occupational exposures, including irritants and psychologic stressors. For example, occupationally triggered VCD was diagnosed in rescue, recovery, and cleanup workers at the World Trade Center disaster site.4
A history of childhood sexual abuse has also been associated by some researchers with the development of VCD. For example, Freedman et al reported that, of 47 patients with VCD, 14 identified such a history and five were suspected of having been sexually abused as children.17
Paradoxical movement of the vocal cords causes them to close when they should open. (Click here for a video on normal and abnormal vocal cord movement.) VCD generally occurs during inspiration, causing obstruction of the incoming air through the larynx. Symptoms of VCD frequently include dyspnea, coughing, wheezing, hoarseness, and tightness or pain in the throat.
Examination of the flow-volume loops recorded when a patient experiences “wheezing” during spirometry testing reveals a flattened inspiratory loop, indicating a decrease of airflow into the lungs (see Figure 1).13,16 “Wheezing” is actually a misnomer in this situation because the term typically refers to sounds that occur during expiration.
Triggers
Physiologic, psychologic, and neurologic factors may all contribute to VCD.1,15 Conditions that can trigger VCD include
• Asthma
• Postnasal drip
• Recent upper respiratory illness (URI)
• Talking, singing
• Exercise
• Cough
• Voice strain
• Stress, anxiety, tension, elevated emotions
• Common irritants (eg, strong smells)
• Airborne irritants
• Rhinosinusitis
• GERD
• Use of certain medications
Identification of a particular patient’s triggers is key to successful management of VCD.
PATIENT PRESENTATION
Although there is no “typical” patient with VCD, the condition occurs more frequently in women, with the most common age at onset between 20 and 40 years. However, VCD has been seen in very young children and in adults as old as 83, and its diagnosis in the pediatric population is increasing.18
The patient may present with complaints of atypical chest pain, throat tightness, stridor, choking, difficult vocalization, cough and sometimes dysphagia, GERD, or rhinosinusitis (see Table 1). These signs and symptoms may occur without provocation, or patients may relate a history of triggers such as anxiety, irritant exposure, or exercise. In fact, about 14% of VCD is associated with exercise, particularly in young female athletes who experience shortness of breath and even stridor with exercise.19
A characteristic finding on physical examination is inspiratory stridor, along with respiratory distress.20 The stridor is best auscultated not over the anterior chest wall but over the tracheal area of the anterior neck.
Continue for differential diagnosis >>
DIFFERENTIAL DIAGNOSIS
Distinguishing VCD from other disorders can be challenging. Differential diagnosis should include
• Non–vocal cord adduction disorders, such as thyroid goiter, upper airway hemorrhage, caustic ingestion, neoplastic disorders, rheumatoid cricoarytenoid arthritis, pharyngeal abscess, angioedema, pulmonary embolus21
• Anatomic defects (eg, laryngomalacia, subglottic stenosis)
• Tracheal masses (eg, enlarged thyroid gland)
• Vocal cord polyps
• Laryngospasm
• Vocal cord paresis
• Neurologic causes (eg, brain stem compression, severe cortical injury, nuclear or lower motor neuron injury, movement disorders)
• Nonorganic causes (eg, factitious symptoms or malingering; conversion disorder)22
• Reactive airway disease.
Some disorders are easier to distinguish from VCD than others. For example, although laryngospasm may produce similar symptoms, episodes are brief, lasting seconds to minutes; VCD episodes may last hours to days.
Asthma
Even the most astute clinician will be unable to obtain adequate information from the patient history to differentiate VCD from asthma. There is a significant overlap of symptoms—shortness of breath, cough, wheezing—and frequently, the diseases coexist. History is often negative for chest pain, but it is common for patients with VCD, when asked to describe their symptoms, to report chest tightness. The clinician therefore needs to ask the patient to point to where the tightness is felt—in the chest or in the neck over the laryngeal area—to distinguish the source.
Asthma symptoms usually increase over a few hours, days, or weeks but respond to medications that open the airway and reduce inflammation (inhaled β-agonists and corticosteroids). VCD symptoms usually occur or decrease suddenly and do not respond well to traditional asthma treatments.
Other differences between asthma and VCD symptoms include voice changes and time of day when symptoms occur. The person with VCD will experience voice changes, such as hoarseness, as well as prolonged coughing episodes. Patients with asthma may awaken at night because of breathlessness, while most patients with VCD experience symptoms only during the day.
The diagnosis is generally confirmed if VCD is seen on direct laryngoscopic visualization during a symptomatic episode. In terms of adduction, the anterior cords will appear normal, but the posterior portion of the cords will display the classic “glottis chink” (see Figure 2).9
If the diagnosis is in question, videostroboscopy, a technique that provides a magnified slow-motion view of vocal cord vibration, can help identify or exclude pathologic conditions of the vocal cords.23
Convincing the patient of the validity of the diagnosis may be problematic if the patient has been previously diagnosed with and treated for another condition. The diagnosis should be explained and the patient counseled what appropriate care for VCD entails (see discussion under “Patient education and self-care”).
TREATMENT
Acute episode
During an acute VCD episode, offering the patient calm reassurance can be effective in resolving the episode. Simple breathing guidance may also be beneficial; instructing the patient to breathe rapidly and shallowly (ie, pant) can result in immediate resolution of symptoms.24 The patient can be advised to utilize other techniques, such as diaphragmatic breathing, breathing through the nose, breathing through a straw, pursed-lip breathing, and exhaling with a hissing sound.25
Long-term management
Although various strategies are employed in the management of VCD, well-designed studies on which to base treatment decisions have not been performed. Of course, control and management of possible underlying triggers or disorders should be implemented. Because etiology is rarely known, treatment for VCD is generally empiric.
Evidence does exist, however, to suggest that voice therapy, the treatment of choice for muscle tension dysphonia, is also effective for VCD. Speech therapy with specific voice and breathing exercises can enable the patient to manage the condition, thereby reducing ED visits, hospitalizations, and treatment costs.26
Patient education and self-care
Patient education is a critical component of VCD management. The clinician should explain the functions of the larynx to the patient, including the normal functioning of the vocal cords during respiration, speaking, swallowing, coughing, throat clearing, and breath holding. It may also enhance patients’ understanding of VCD to view their diagnostic laryngoscopy or videostroboscopy films.21
The patient should be advised to rest the voice, hydrate, utilize sialagogues (lozenges, gum) to stimulate salivation, reduce exposure to triggers when possible, and decrease stress. She should be encouraged to track VCD triggers by documenting what she is doing, where, and when, at the time of a VCD episode.
Two exercises—“paused breathing” and “belly breathing”—can be used by patients to learn how to relax the vocal cords (see “Patient Handout”). Patients should practice these exercises three times a day so that they can be easily recalled and performed during VCD episodes.
Continue for outcomes >>
OUTCOMES
Little is known about long-term outcomes for patients with VCD. The current literature consists of poorly described and conflicting case reports and results of small trials. Although documentation is lacking, the authors agree that, by educating the patient about the diagnosis, teaching effective VCD management strategies, and referring patients for voice therapy, clinicians can help patients achieve signicant improvement. Further investigation is needed to enhance our knowledge of the causes of VCD and to research additional diagnostic modalities and treatments.2
CASE PATIENT
After diagnosing VCD, the clinician explained the normal functioning of the vocal cords and how certain factors may cause them to close during inspiration. The patient then understood why bronchodilator therapy had failed to relieve her symptoms. She was counseled to continue her inhaled nasal steroid and proton pump inhibitor for her perennial nonallergic rhinitis and GERD, respectively, because these conditions may trigger her VCD, and to take steps to manage her stress. She learned breathing techniques to alleviate acute episodes of VCD and was informed of the option of voice therapy with a speech therapist if needed.
At six-week follow-up, the patient reported that she was complying with her medication regimen, had made an effort to relax more, and had experienced no acute attacks of VCD since her last visit.
CONCLUSION
Patients with symptoms suggestive of VCD require a thorough evaluation, including laryngoscopic examination, to ensure accurate diagnosis and avoid a too-common misdiagnosis. Primary care clinicians should know about VCD and, if not trained in the performance of flexible laryngoscopy, should refer the symptomatic patient to a specialist for appropriate work-up.
1. Hoyte FCL. Vocal cord dysfunction. Immunol Allergy Clin N Am. 2013;33:1-22.
2. Ibrahim WH, Gheriani HA, Almohamed AA, Raza T. Paradoxical vocal cord motion disorder: past, present and future. Postgrad Med J. 2007;83:164-172.
3. Powell DM, Karanfilov BI, Beechler KB, et al. Paradoxical vocal cord dysfunction in juveniles. Arch Otolaryngol Head Neck Surg. 2000;126(1):29-34.
4. Husein OF, Husein TN, Gardner R, et al. Formal psychological testing in patients with paradoxical vocal fold dysfunction. Laryngoscope. 2008; 118(4):740-747.
5. Dunglison RD. The Practice of Medicine. Philadelphia, PA: Lea and Blanchard; 1842:257-258.
6. MacKenzie M. Use of Laryngoscopy in Diseases of the Throat. Philadelphia, PA: Lindsey and Blackeston; 1869:246-250.
7. Osler W. Hysteria. In: The Principles and Practice of Medicine. 4th ed. New York, NY: Appleton; 1902:1111-1112.
8. Patterson R, Schatz M, Horton M. Munchausen’s stridor: non-organic laryngeal obstruction. Clin Allergy. 1974;4:307-310.
9. Christopher KL, Wood RP 2nd, Eckert RC, et al. Vocal cord dysfunction presenting as asthma. N Engl J Med. 1983;308(26):1566-1570.
10. Christopher KL. Understanding vocal cord dysfunction: a step in the right direction with a long road ahead. Chest. 2006;129(4):842-843.
11. Christopher KL, Morris MJ. Vocal cord dysfunction, paradoxic vocal fold motion, or laryngomalacia? Our understanding requires an interdisciplinary approach. Otolaryngol Clin N Am. 2010;43:43-66.
12. Sasaki CT, Weaver EM. Physiology of the larynx. Am J Med. 1997;103:9S-18S.
13. Balkissoon R. Occupational upper airway disease. Clin Chest Med. 2002;23:717-725.
14. Murakami Y, Kirschner JA. Mechanical and physiological properties of reflex laryngeal closure. Ann Otol Rhinol Laryngol. 1972;81(1):59-71.
15. Forrest LA, Husein T, Husein O. Paradoxical vocal cord motion disorder: classification and treatment. Laryngoscope. 2012;122:844-853.
16. Altman KW, Simpson CB, Amin MR, et al. Cough and paradoxical vocal fold motion. Otolaryngol Head Neck Surg. 2002;127(6):501-511.
17. Freedman MR, Rosenberg SJ, Schmaling KB. Childhood sexual abuse in patients with paradoxical vocal cord dysfunction. J Nerv Ment Dis. 1991;179(5):295-298.
18. Buddiga P. Vocal cord dysfunction. Medscape. http://emedicine.medscape.com/article/137782-overview. Accessed November 12, 2014.
19. Chiang T, Marcinow AM, deSilva BW, et al. Exercise-induced paradoxical vocal fold motion disorder: diagnosis and management. Laryngoscope. 2013;123:727-731.
20. Morris MJ, Deal LE, Bean DR, et al. Vocal cord dysfunction in patients with exertional dyspnea. Chest. 1999;116(6):1676-1682.
21. Hicks M, Brugman SM, Katial R. Vocal cord dysfunction/paradoxical vocal fold motion. Prim Care. 2008;35(1):81-103.
22. Maschka DA, Bauman NM, McCray PB, et al. A classification scheme for paradoxical vocal fold motion. Laryngoscope. 1997;107(11):1429-1435.
23. Uloza V, Vegiene A, Pribuisiene R, Saferis V. Quantitative evaluation of video laryngostroboscopy: reliability of the basic parameters. J Voice. 2013;27(3):361-368.
24. Pitchenik AF. Functional laryngeal obstruction relieved by panting. Chest. 1991;100(5):1465-1467.
25. Deckert J, Deckert L. Vocal cord dysfunction. Am Fam Physician. 2010;81(2):156-160.
26. Carding PN, Horsley IA, Docherty GJ. A study of the effectiveness of voice therapy in the treatment of 45 patients with nonorganic dysphonia. J Voice. 1999;13(1):72-104.
1. Hoyte FCL. Vocal cord dysfunction. Immunol Allergy Clin N Am. 2013;33:1-22.
2. Ibrahim WH, Gheriani HA, Almohamed AA, Raza T. Paradoxical vocal cord motion disorder: past, present and future. Postgrad Med J. 2007;83:164-172.
3. Powell DM, Karanfilov BI, Beechler KB, et al. Paradoxical vocal cord dysfunction in juveniles. Arch Otolaryngol Head Neck Surg. 2000;126(1):29-34.
4. Husein OF, Husein TN, Gardner R, et al. Formal psychological testing in patients with paradoxical vocal fold dysfunction. Laryngoscope. 2008; 118(4):740-747.
5. Dunglison RD. The Practice of Medicine. Philadelphia, PA: Lea and Blanchard; 1842:257-258.
6. MacKenzie M. Use of Laryngoscopy in Diseases of the Throat. Philadelphia, PA: Lindsey and Blackeston; 1869:246-250.
7. Osler W. Hysteria. In: The Principles and Practice of Medicine. 4th ed. New York, NY: Appleton; 1902:1111-1112.
8. Patterson R, Schatz M, Horton M. Munchausen’s stridor: non-organic laryngeal obstruction. Clin Allergy. 1974;4:307-310.
9. Christopher KL, Wood RP 2nd, Eckert RC, et al. Vocal cord dysfunction presenting as asthma. N Engl J Med. 1983;308(26):1566-1570.
10. Christopher KL. Understanding vocal cord dysfunction: a step in the right direction with a long road ahead. Chest. 2006;129(4):842-843.
11. Christopher KL, Morris MJ. Vocal cord dysfunction, paradoxic vocal fold motion, or laryngomalacia? Our understanding requires an interdisciplinary approach. Otolaryngol Clin N Am. 2010;43:43-66.
12. Sasaki CT, Weaver EM. Physiology of the larynx. Am J Med. 1997;103:9S-18S.
13. Balkissoon R. Occupational upper airway disease. Clin Chest Med. 2002;23:717-725.
14. Murakami Y, Kirschner JA. Mechanical and physiological properties of reflex laryngeal closure. Ann Otol Rhinol Laryngol. 1972;81(1):59-71.
15. Forrest LA, Husein T, Husein O. Paradoxical vocal cord motion disorder: classification and treatment. Laryngoscope. 2012;122:844-853.
16. Altman KW, Simpson CB, Amin MR, et al. Cough and paradoxical vocal fold motion. Otolaryngol Head Neck Surg. 2002;127(6):501-511.
17. Freedman MR, Rosenberg SJ, Schmaling KB. Childhood sexual abuse in patients with paradoxical vocal cord dysfunction. J Nerv Ment Dis. 1991;179(5):295-298.
18. Buddiga P. Vocal cord dysfunction. Medscape. http://emedicine.medscape.com/article/137782-overview. Accessed November 12, 2014.
19. Chiang T, Marcinow AM, deSilva BW, et al. Exercise-induced paradoxical vocal fold motion disorder: diagnosis and management. Laryngoscope. 2013;123:727-731.
20. Morris MJ, Deal LE, Bean DR, et al. Vocal cord dysfunction in patients with exertional dyspnea. Chest. 1999;116(6):1676-1682.
21. Hicks M, Brugman SM, Katial R. Vocal cord dysfunction/paradoxical vocal fold motion. Prim Care. 2008;35(1):81-103.
22. Maschka DA, Bauman NM, McCray PB, et al. A classification scheme for paradoxical vocal fold motion. Laryngoscope. 1997;107(11):1429-1435.
23. Uloza V, Vegiene A, Pribuisiene R, Saferis V. Quantitative evaluation of video laryngostroboscopy: reliability of the basic parameters. J Voice. 2013;27(3):361-368.
24. Pitchenik AF. Functional laryngeal obstruction relieved by panting. Chest. 1991;100(5):1465-1467.
25. Deckert J, Deckert L. Vocal cord dysfunction. Am Fam Physician. 2010;81(2):156-160.
26. Carding PN, Horsley IA, Docherty GJ. A study of the effectiveness of voice therapy in the treatment of 45 patients with nonorganic dysphonia. J Voice. 1999;13(1):72-104.
‘Shotgun’ skin prick testing for food allergy held flawed
VANCOUVER, B.C. – The “shotgun” style of skin prick testing in children and adolescents with suspected IgE-mediated food allergy shows sensitization, but not necessarily allergy, according to Dr. James Bergman.
A positive skin test measures the presence of a specific IgE antibody, which does not necessarily equate to an allergy. Consequently, children may have multiple positive skin prick tests yet clinically tolerate the tested food, he said. “Sensitization is just the presence of a specific IgE to a food. Allergy is sensitization plus signs or symptoms upon exposure to the food.”
Dr. Bergman, who also holds a faculty position in the department of dermatology and skin science at the University of British Columbia, said the practice of shotgun skin prick testing can lead to unnecessary avoidance of specific foods. One group of researchers conducted oral food challenge tests in 125 children aged 1-9 years with a diagnosis of food allergy based on IgE tests. Nearly all of them (93%) had no reactivity when challenged with the suspect food (J. Peds. 2011; 158[4]:578-83). “Ninety-three percent of the children would have been avoiding their ‘allergic foods’ perhaps indefinitely,” said Dr. Bergman, who was not involved with the study.
“The general rule is, if you’re not having clinical symptoms that suggest an IgE-mediated reaction, then don’t test,” Dr. Bergman, a dermatologist who practices in Vancouver, said at the annual meeting of the Pacific Dermatologic Association.
“I explain to parents that if they want to test for a food in the situation where there is no IgE-mediated reaction, then it can be done, but there is a significant risk of a false positive or ‘fake allergy,’ ” he said. “In this situation the only way of knowing for sure whether it is an allergy is to undertake a formal oral food challenge, which is the (highest) standard for diagnosing food allergy.”
Telltale symptoms of an IgE-mediated food allergy include hives, vomiting, diarrhea, breathing problems, and change in level of consciousness. “These symptoms typically occur within minutes of ingestion, sometimes within 30 minutes and rarely up to 2 hours,” Dr. Bergman said. “If it’s beyond 2 hours, it’s unlikely to be IgE mediated.”
“If someone has a true food allergy, advise them to avoid the culprit food, give them an epinephrine injector, and refer them to an allergist for testing, education, and follow-up,” he advised.
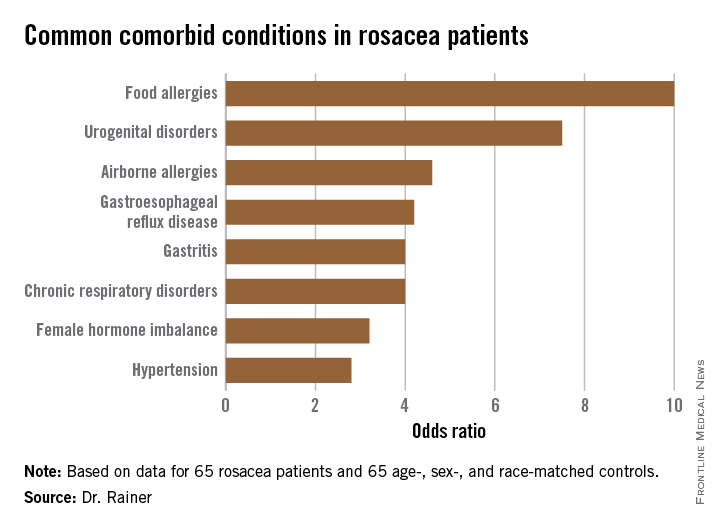
Food allergies affect 6%-8% of pediatric patients, yet 35%- 90% of families self-report food allergies depending on the population studied. Milk, egg, wheat, peanuts, nuts, soy, and seafood account for 90% of food allergens. Most children outgrow allergy to milk, egg, wheat, and soy, while few outgrow allergy to peanuts, nuts, fish, and shellfish.
Most patients and many physicians believe that eczema is caused by food allergies. In fact, only a small minority of patients have food allergies that directly cause eczema. “Eczema could occur secondary to scratching induced by an urticarial food reaction or by a primary irritant reaction, but food directly causing isolated eczema is rare,” Dr. Bergman said. “The belief that food allergies directly cause eczema is completely understandable given that eczema patients do have an increased rate of allergies, the cyclic pattern of eczema, and the parent’s desire to find a cause for the child’s rash. Eczema’s cyclic nature can easily lead to a specific food being implicated due to recall bias. The parent will remember the flares that occurred with exposure to the specific food, while not recalling the times when the food was tolerated or the flares that were not associated with the food.”
If a parent is worried about a food causing eczema and there are no IgE mediated symptoms, then instead of testing he will often recommend that the family keep a formal food symptom diary while they are intermittently ingesting the food of concern. “The vast majority of parents will see no consistent direct correlation with the food and they can feel comfortable with ongoing future ingestion,” he said.
Some clinicians are offering oral immune therapy to patients with IgE-mediated food allergy. Dr. Bergman characterized such practice as “risky” at this point in time. “It’s like the traditional allergy shots you’d get for your pollen allergy, except it’s done orally,” he explained. “Research is being done in this area by introducing small amounts [of the allergen], in an attempt to induce tolerance. The results are encouraging, but the problem is that patients can have bad reactions. We also don’t know how well or for how long it will work. At this point, while promising, the field is not yet ready for prime time.”
He also said there is no current evidence supporting IgG testing, Vega testing, or muscle strength testing in the investigation of suspected IgE-mediated food allergy. “What I tell patients is that if any of these tests identifies something, it probably identifies something that’s mild and very temporary, because in my experience patients with positive IgG tests are usually told to avoid the food for 1-4 months and then to reintroduce that food in a rotation basis. Avoidance of food allergens based on this type of testing is not necessary. However, for patients who still wish to practice short term avoidance of the food then this is fine provided the diet does not compromise nutrition.”
Dr. Bergman reported having no financial disclosures.
On Twitter @dougbrunk
VANCOUVER, B.C. – The “shotgun” style of skin prick testing in children and adolescents with suspected IgE-mediated food allergy shows sensitization, but not necessarily allergy, according to Dr. James Bergman.
A positive skin test measures the presence of a specific IgE antibody, which does not necessarily equate to an allergy. Consequently, children may have multiple positive skin prick tests yet clinically tolerate the tested food, he said. “Sensitization is just the presence of a specific IgE to a food. Allergy is sensitization plus signs or symptoms upon exposure to the food.”
Dr. Bergman, who also holds a faculty position in the department of dermatology and skin science at the University of British Columbia, said the practice of shotgun skin prick testing can lead to unnecessary avoidance of specific foods. One group of researchers conducted oral food challenge tests in 125 children aged 1-9 years with a diagnosis of food allergy based on IgE tests. Nearly all of them (93%) had no reactivity when challenged with the suspect food (J. Peds. 2011; 158[4]:578-83). “Ninety-three percent of the children would have been avoiding their ‘allergic foods’ perhaps indefinitely,” said Dr. Bergman, who was not involved with the study.
“The general rule is, if you’re not having clinical symptoms that suggest an IgE-mediated reaction, then don’t test,” Dr. Bergman, a dermatologist who practices in Vancouver, said at the annual meeting of the Pacific Dermatologic Association.
“I explain to parents that if they want to test for a food in the situation where there is no IgE-mediated reaction, then it can be done, but there is a significant risk of a false positive or ‘fake allergy,’ ” he said. “In this situation the only way of knowing for sure whether it is an allergy is to undertake a formal oral food challenge, which is the (highest) standard for diagnosing food allergy.”
Telltale symptoms of an IgE-mediated food allergy include hives, vomiting, diarrhea, breathing problems, and change in level of consciousness. “These symptoms typically occur within minutes of ingestion, sometimes within 30 minutes and rarely up to 2 hours,” Dr. Bergman said. “If it’s beyond 2 hours, it’s unlikely to be IgE mediated.”
“If someone has a true food allergy, advise them to avoid the culprit food, give them an epinephrine injector, and refer them to an allergist for testing, education, and follow-up,” he advised.
Food allergies affect 6%-8% of pediatric patients, yet 35%- 90% of families self-report food allergies depending on the population studied. Milk, egg, wheat, peanuts, nuts, soy, and seafood account for 90% of food allergens. Most children outgrow allergy to milk, egg, wheat, and soy, while few outgrow allergy to peanuts, nuts, fish, and shellfish.
Most patients and many physicians believe that eczema is caused by food allergies. In fact, only a small minority of patients have food allergies that directly cause eczema. “Eczema could occur secondary to scratching induced by an urticarial food reaction or by a primary irritant reaction, but food directly causing isolated eczema is rare,” Dr. Bergman said. “The belief that food allergies directly cause eczema is completely understandable given that eczema patients do have an increased rate of allergies, the cyclic pattern of eczema, and the parent’s desire to find a cause for the child’s rash. Eczema’s cyclic nature can easily lead to a specific food being implicated due to recall bias. The parent will remember the flares that occurred with exposure to the specific food, while not recalling the times when the food was tolerated or the flares that were not associated with the food.”
If a parent is worried about a food causing eczema and there are no IgE mediated symptoms, then instead of testing he will often recommend that the family keep a formal food symptom diary while they are intermittently ingesting the food of concern. “The vast majority of parents will see no consistent direct correlation with the food and they can feel comfortable with ongoing future ingestion,” he said.
Some clinicians are offering oral immune therapy to patients with IgE-mediated food allergy. Dr. Bergman characterized such practice as “risky” at this point in time. “It’s like the traditional allergy shots you’d get for your pollen allergy, except it’s done orally,” he explained. “Research is being done in this area by introducing small amounts [of the allergen], in an attempt to induce tolerance. The results are encouraging, but the problem is that patients can have bad reactions. We also don’t know how well or for how long it will work. At this point, while promising, the field is not yet ready for prime time.”
He also said there is no current evidence supporting IgG testing, Vega testing, or muscle strength testing in the investigation of suspected IgE-mediated food allergy. “What I tell patients is that if any of these tests identifies something, it probably identifies something that’s mild and very temporary, because in my experience patients with positive IgG tests are usually told to avoid the food for 1-4 months and then to reintroduce that food in a rotation basis. Avoidance of food allergens based on this type of testing is not necessary. However, for patients who still wish to practice short term avoidance of the food then this is fine provided the diet does not compromise nutrition.”
Dr. Bergman reported having no financial disclosures.
On Twitter @dougbrunk
VANCOUVER, B.C. – The “shotgun” style of skin prick testing in children and adolescents with suspected IgE-mediated food allergy shows sensitization, but not necessarily allergy, according to Dr. James Bergman.
A positive skin test measures the presence of a specific IgE antibody, which does not necessarily equate to an allergy. Consequently, children may have multiple positive skin prick tests yet clinically tolerate the tested food, he said. “Sensitization is just the presence of a specific IgE to a food. Allergy is sensitization plus signs or symptoms upon exposure to the food.”
Dr. Bergman, who also holds a faculty position in the department of dermatology and skin science at the University of British Columbia, said the practice of shotgun skin prick testing can lead to unnecessary avoidance of specific foods. One group of researchers conducted oral food challenge tests in 125 children aged 1-9 years with a diagnosis of food allergy based on IgE tests. Nearly all of them (93%) had no reactivity when challenged with the suspect food (J. Peds. 2011; 158[4]:578-83). “Ninety-three percent of the children would have been avoiding their ‘allergic foods’ perhaps indefinitely,” said Dr. Bergman, who was not involved with the study.
“The general rule is, if you’re not having clinical symptoms that suggest an IgE-mediated reaction, then don’t test,” Dr. Bergman, a dermatologist who practices in Vancouver, said at the annual meeting of the Pacific Dermatologic Association.
“I explain to parents that if they want to test for a food in the situation where there is no IgE-mediated reaction, then it can be done, but there is a significant risk of a false positive or ‘fake allergy,’ ” he said. “In this situation the only way of knowing for sure whether it is an allergy is to undertake a formal oral food challenge, which is the (highest) standard for diagnosing food allergy.”
Telltale symptoms of an IgE-mediated food allergy include hives, vomiting, diarrhea, breathing problems, and change in level of consciousness. “These symptoms typically occur within minutes of ingestion, sometimes within 30 minutes and rarely up to 2 hours,” Dr. Bergman said. “If it’s beyond 2 hours, it’s unlikely to be IgE mediated.”
“If someone has a true food allergy, advise them to avoid the culprit food, give them an epinephrine injector, and refer them to an allergist for testing, education, and follow-up,” he advised.
Food allergies affect 6%-8% of pediatric patients, yet 35%- 90% of families self-report food allergies depending on the population studied. Milk, egg, wheat, peanuts, nuts, soy, and seafood account for 90% of food allergens. Most children outgrow allergy to milk, egg, wheat, and soy, while few outgrow allergy to peanuts, nuts, fish, and shellfish.
Most patients and many physicians believe that eczema is caused by food allergies. In fact, only a small minority of patients have food allergies that directly cause eczema. “Eczema could occur secondary to scratching induced by an urticarial food reaction or by a primary irritant reaction, but food directly causing isolated eczema is rare,” Dr. Bergman said. “The belief that food allergies directly cause eczema is completely understandable given that eczema patients do have an increased rate of allergies, the cyclic pattern of eczema, and the parent’s desire to find a cause for the child’s rash. Eczema’s cyclic nature can easily lead to a specific food being implicated due to recall bias. The parent will remember the flares that occurred with exposure to the specific food, while not recalling the times when the food was tolerated or the flares that were not associated with the food.”
If a parent is worried about a food causing eczema and there are no IgE mediated symptoms, then instead of testing he will often recommend that the family keep a formal food symptom diary while they are intermittently ingesting the food of concern. “The vast majority of parents will see no consistent direct correlation with the food and they can feel comfortable with ongoing future ingestion,” he said.
Some clinicians are offering oral immune therapy to patients with IgE-mediated food allergy. Dr. Bergman characterized such practice as “risky” at this point in time. “It’s like the traditional allergy shots you’d get for your pollen allergy, except it’s done orally,” he explained. “Research is being done in this area by introducing small amounts [of the allergen], in an attempt to induce tolerance. The results are encouraging, but the problem is that patients can have bad reactions. We also don’t know how well or for how long it will work. At this point, while promising, the field is not yet ready for prime time.”
He also said there is no current evidence supporting IgG testing, Vega testing, or muscle strength testing in the investigation of suspected IgE-mediated food allergy. “What I tell patients is that if any of these tests identifies something, it probably identifies something that’s mild and very temporary, because in my experience patients with positive IgG tests are usually told to avoid the food for 1-4 months and then to reintroduce that food in a rotation basis. Avoidance of food allergens based on this type of testing is not necessary. However, for patients who still wish to practice short term avoidance of the food then this is fine provided the diet does not compromise nutrition.”
Dr. Bergman reported having no financial disclosures.
On Twitter @dougbrunk
EXPERT ANALYSIS FROM THE PDA ANNUAL MEETING
Topical steroid might improve mucosal integrity in eosinophilic esophagitis
Topical steroid therapy improved some indicators of mucosal integrity in patients with eosinophilic esophagitis, but proton pump inhibitor therapy did not, according to two studies reported in the November issue of Clinical Gastroenterology and Hepatology.
The first study found that topical fluticasone therapy at a dose of 880 mcg twice daily for 2 months helped correct esophageal spongiosis, or dilated intercellular space, in patients with eosinophilic esophagitis (EoE). Spongiosis scores for treated patients were significantly lower than for untreated patients (0.4 vs. 1.3; P = .016), said Dr. David Katzka at the Mayo Clinic in Rochester, Minn. and his associates (Clin. Gastroenterol. Hepatol. 2014 [doi:10.1016/j.cgh.2014.02.039]).
In the study, histologic analyses also showed that improved spongiosis scores in treated patients correlated with increased density of two tight junction proteins, filaggrin (P = .001) and zonula occludens-3 (P = .016), said the investigators. These proteins might help regulate antigenic penetration of the esophageal mucosa and also could permit migration of white blood cells, they said. “Loss of tight junction regulators and dilation of intercellular spaces appear to be involved in the pathophysiology of EoE and could be targets for treatment,” the researchers concluded. But they also noted that their study did not examine the same patients before and after steroid therapy and did not look at desmosomes, intercellular junctions that past research has suggested might be affected in EoE.
For the second study, Dr. Bram van Rhijn and his associates at the Academic Medical Center in the Netherlands compared endoscopies of 16 patients with dysphagia and suspected (unconfirmed) EoE with 11 controls, both at baseline and after 8 weeks of high-dose esomeprazole treatment. Esophageal mucosal integrity was “severely impaired” in patients with confirmed EoE and in those with proton pump inhibitor–responsive eosinophilia (PPRE), the researchers said (Clin. Gasteroenterol. Hepatol. 2014 [doi:10.1016/j.cgh.2014.02.037]).
In both forms of disease, molecules as large as 40,000 daltons were able to pass through the compromised esophageal mucosa, Dr. Bram van Rhijn and his associates reported. “This size is similar to the size of most plant and animal food allergens to which EoE patients are sensitized,” they added. Esophageal permeability might increase the rate of immune exposure to allergens, thereby mediating EoE and PPRE, they said.
On mucosal functional tests, both EoE and PPRE were associated with reduced transepithelial electrical resistance and lower electrical tissue impedance, most notably in patients with EoE (P less than .001 for both, compared with controls), the investigators reported. Proton pump inhibitor treatment partially reversed these changes in patients with PPRE but showed no effect for patients with EoE, they said. This finding suggests that acid reflux might play a role in PPRE, but not in EOE, they concluded.
Dr. Katzka and his associates disclosed no funding sources and reported having no conflicts of interest. Dr. Rhijn and his associates were supported by the Netherlands Organization for Scientific Research. Two of Dr. Rhijn’s coauthors reported financial relationships with AstraZeneca, Endostim, Medical Measurement Systems, Shire, and GlaxoSmithKline.
In the past year, the topic of mucosal integrity in eosinophilic esophagitis has garnered growing attention. Epithelial permeability defects have been described in the pathogenesis of GI disorders, including inflammatory bowel disease and celiac sprue, as well as allergic disorders such as atopic dermatitis. In EoE, both experimental as well as clinical studies have shown an eosinophil-predominant inflammatory response to specific antigens, particularly common food allergens. Increased permeability may predispose genetically susceptible individuals to swallowed allergen penetration through the esophageal epithelium. Beneath the epithelial barrier, antigens have access to antigen presenting cells, including dendritic cells, leading to both allergic sensitization and perpetuation of the TH-2 chronic inflammatory response.

|
| Dr. Ikuo Hirano |
The article by Dr. Katzka and his colleagues supports the concept of epithelial barrier defects in EoE through the demonstration of reduced immunohistochemical expression of filaggrin, zonula occludens-3, and claudin-1, important tight junction proteins. Expression was increased in EoE patients treated with topical steroids. Similarly, the study by Dr. van Rhijn and his associates identified impaired mucosal integrity in EoE by a variety of techniques that included electron microscopic demonstration of dilated intercellular spaces, electrical tissue impedance as an in vivo biomarker, and in vitro transepithelial molecular flux in an Ussing chamber. Furthermore, they found that proton pump inhibitor therapy partially restored mucosal permeability defects to a greater degree in patients with PPI-responsive esophageal eosinophilia, compared with patients with EoE. These two studies substantiate studies from the Cincinnati group that previously identified reduced mRNA expression of filaggrin in esophageal mucosal biopsies as well as reduced expression of the intercellular adhesion molecule, desmoglein 1.
In spite of these novel data, the exact role of altered esophageal epithelial permeability in the pathogenesis of EoE is yet unclear. The reversibility of the defect with medical therapy argues against defective cell junction proteins as an intrinsic abnormality. Furthermore, the location of antigen presentation in EoE may occur through other routes such as the small intestine, nasal epithelium, or skin. In the meantime, these studies provide an important advance in our understanding of EoE and open the door to novel therapeutic approaches.
Dr. Ikuo Hirano, AGAF, is professor of medicine at Northwestern University, Chicago. He reported no conflicts of interest.
In the past year, the topic of mucosal integrity in eosinophilic esophagitis has garnered growing attention. Epithelial permeability defects have been described in the pathogenesis of GI disorders, including inflammatory bowel disease and celiac sprue, as well as allergic disorders such as atopic dermatitis. In EoE, both experimental as well as clinical studies have shown an eosinophil-predominant inflammatory response to specific antigens, particularly common food allergens. Increased permeability may predispose genetically susceptible individuals to swallowed allergen penetration through the esophageal epithelium. Beneath the epithelial barrier, antigens have access to antigen presenting cells, including dendritic cells, leading to both allergic sensitization and perpetuation of the TH-2 chronic inflammatory response.

|
| Dr. Ikuo Hirano |
The article by Dr. Katzka and his colleagues supports the concept of epithelial barrier defects in EoE through the demonstration of reduced immunohistochemical expression of filaggrin, zonula occludens-3, and claudin-1, important tight junction proteins. Expression was increased in EoE patients treated with topical steroids. Similarly, the study by Dr. van Rhijn and his associates identified impaired mucosal integrity in EoE by a variety of techniques that included electron microscopic demonstration of dilated intercellular spaces, electrical tissue impedance as an in vivo biomarker, and in vitro transepithelial molecular flux in an Ussing chamber. Furthermore, they found that proton pump inhibitor therapy partially restored mucosal permeability defects to a greater degree in patients with PPI-responsive esophageal eosinophilia, compared with patients with EoE. These two studies substantiate studies from the Cincinnati group that previously identified reduced mRNA expression of filaggrin in esophageal mucosal biopsies as well as reduced expression of the intercellular adhesion molecule, desmoglein 1.
In spite of these novel data, the exact role of altered esophageal epithelial permeability in the pathogenesis of EoE is yet unclear. The reversibility of the defect with medical therapy argues against defective cell junction proteins as an intrinsic abnormality. Furthermore, the location of antigen presentation in EoE may occur through other routes such as the small intestine, nasal epithelium, or skin. In the meantime, these studies provide an important advance in our understanding of EoE and open the door to novel therapeutic approaches.
Dr. Ikuo Hirano, AGAF, is professor of medicine at Northwestern University, Chicago. He reported no conflicts of interest.
In the past year, the topic of mucosal integrity in eosinophilic esophagitis has garnered growing attention. Epithelial permeability defects have been described in the pathogenesis of GI disorders, including inflammatory bowel disease and celiac sprue, as well as allergic disorders such as atopic dermatitis. In EoE, both experimental as well as clinical studies have shown an eosinophil-predominant inflammatory response to specific antigens, particularly common food allergens. Increased permeability may predispose genetically susceptible individuals to swallowed allergen penetration through the esophageal epithelium. Beneath the epithelial barrier, antigens have access to antigen presenting cells, including dendritic cells, leading to both allergic sensitization and perpetuation of the TH-2 chronic inflammatory response.

|
| Dr. Ikuo Hirano |
The article by Dr. Katzka and his colleagues supports the concept of epithelial barrier defects in EoE through the demonstration of reduced immunohistochemical expression of filaggrin, zonula occludens-3, and claudin-1, important tight junction proteins. Expression was increased in EoE patients treated with topical steroids. Similarly, the study by Dr. van Rhijn and his associates identified impaired mucosal integrity in EoE by a variety of techniques that included electron microscopic demonstration of dilated intercellular spaces, electrical tissue impedance as an in vivo biomarker, and in vitro transepithelial molecular flux in an Ussing chamber. Furthermore, they found that proton pump inhibitor therapy partially restored mucosal permeability defects to a greater degree in patients with PPI-responsive esophageal eosinophilia, compared with patients with EoE. These two studies substantiate studies from the Cincinnati group that previously identified reduced mRNA expression of filaggrin in esophageal mucosal biopsies as well as reduced expression of the intercellular adhesion molecule, desmoglein 1.
In spite of these novel data, the exact role of altered esophageal epithelial permeability in the pathogenesis of EoE is yet unclear. The reversibility of the defect with medical therapy argues against defective cell junction proteins as an intrinsic abnormality. Furthermore, the location of antigen presentation in EoE may occur through other routes such as the small intestine, nasal epithelium, or skin. In the meantime, these studies provide an important advance in our understanding of EoE and open the door to novel therapeutic approaches.
Dr. Ikuo Hirano, AGAF, is professor of medicine at Northwestern University, Chicago. He reported no conflicts of interest.
Topical steroid therapy improved some indicators of mucosal integrity in patients with eosinophilic esophagitis, but proton pump inhibitor therapy did not, according to two studies reported in the November issue of Clinical Gastroenterology and Hepatology.
The first study found that topical fluticasone therapy at a dose of 880 mcg twice daily for 2 months helped correct esophageal spongiosis, or dilated intercellular space, in patients with eosinophilic esophagitis (EoE). Spongiosis scores for treated patients were significantly lower than for untreated patients (0.4 vs. 1.3; P = .016), said Dr. David Katzka at the Mayo Clinic in Rochester, Minn. and his associates (Clin. Gastroenterol. Hepatol. 2014 [doi:10.1016/j.cgh.2014.02.039]).
In the study, histologic analyses also showed that improved spongiosis scores in treated patients correlated with increased density of two tight junction proteins, filaggrin (P = .001) and zonula occludens-3 (P = .016), said the investigators. These proteins might help regulate antigenic penetration of the esophageal mucosa and also could permit migration of white blood cells, they said. “Loss of tight junction regulators and dilation of intercellular spaces appear to be involved in the pathophysiology of EoE and could be targets for treatment,” the researchers concluded. But they also noted that their study did not examine the same patients before and after steroid therapy and did not look at desmosomes, intercellular junctions that past research has suggested might be affected in EoE.
For the second study, Dr. Bram van Rhijn and his associates at the Academic Medical Center in the Netherlands compared endoscopies of 16 patients with dysphagia and suspected (unconfirmed) EoE with 11 controls, both at baseline and after 8 weeks of high-dose esomeprazole treatment. Esophageal mucosal integrity was “severely impaired” in patients with confirmed EoE and in those with proton pump inhibitor–responsive eosinophilia (PPRE), the researchers said (Clin. Gasteroenterol. Hepatol. 2014 [doi:10.1016/j.cgh.2014.02.037]).
In both forms of disease, molecules as large as 40,000 daltons were able to pass through the compromised esophageal mucosa, Dr. Bram van Rhijn and his associates reported. “This size is similar to the size of most plant and animal food allergens to which EoE patients are sensitized,” they added. Esophageal permeability might increase the rate of immune exposure to allergens, thereby mediating EoE and PPRE, they said.
On mucosal functional tests, both EoE and PPRE were associated with reduced transepithelial electrical resistance and lower electrical tissue impedance, most notably in patients with EoE (P less than .001 for both, compared with controls), the investigators reported. Proton pump inhibitor treatment partially reversed these changes in patients with PPRE but showed no effect for patients with EoE, they said. This finding suggests that acid reflux might play a role in PPRE, but not in EOE, they concluded.
Dr. Katzka and his associates disclosed no funding sources and reported having no conflicts of interest. Dr. Rhijn and his associates were supported by the Netherlands Organization for Scientific Research. Two of Dr. Rhijn’s coauthors reported financial relationships with AstraZeneca, Endostim, Medical Measurement Systems, Shire, and GlaxoSmithKline.
Topical steroid therapy improved some indicators of mucosal integrity in patients with eosinophilic esophagitis, but proton pump inhibitor therapy did not, according to two studies reported in the November issue of Clinical Gastroenterology and Hepatology.
The first study found that topical fluticasone therapy at a dose of 880 mcg twice daily for 2 months helped correct esophageal spongiosis, or dilated intercellular space, in patients with eosinophilic esophagitis (EoE). Spongiosis scores for treated patients were significantly lower than for untreated patients (0.4 vs. 1.3; P = .016), said Dr. David Katzka at the Mayo Clinic in Rochester, Minn. and his associates (Clin. Gastroenterol. Hepatol. 2014 [doi:10.1016/j.cgh.2014.02.039]).
In the study, histologic analyses also showed that improved spongiosis scores in treated patients correlated with increased density of two tight junction proteins, filaggrin (P = .001) and zonula occludens-3 (P = .016), said the investigators. These proteins might help regulate antigenic penetration of the esophageal mucosa and also could permit migration of white blood cells, they said. “Loss of tight junction regulators and dilation of intercellular spaces appear to be involved in the pathophysiology of EoE and could be targets for treatment,” the researchers concluded. But they also noted that their study did not examine the same patients before and after steroid therapy and did not look at desmosomes, intercellular junctions that past research has suggested might be affected in EoE.
For the second study, Dr. Bram van Rhijn and his associates at the Academic Medical Center in the Netherlands compared endoscopies of 16 patients with dysphagia and suspected (unconfirmed) EoE with 11 controls, both at baseline and after 8 weeks of high-dose esomeprazole treatment. Esophageal mucosal integrity was “severely impaired” in patients with confirmed EoE and in those with proton pump inhibitor–responsive eosinophilia (PPRE), the researchers said (Clin. Gasteroenterol. Hepatol. 2014 [doi:10.1016/j.cgh.2014.02.037]).
In both forms of disease, molecules as large as 40,000 daltons were able to pass through the compromised esophageal mucosa, Dr. Bram van Rhijn and his associates reported. “This size is similar to the size of most plant and animal food allergens to which EoE patients are sensitized,” they added. Esophageal permeability might increase the rate of immune exposure to allergens, thereby mediating EoE and PPRE, they said.
On mucosal functional tests, both EoE and PPRE were associated with reduced transepithelial electrical resistance and lower electrical tissue impedance, most notably in patients with EoE (P less than .001 for both, compared with controls), the investigators reported. Proton pump inhibitor treatment partially reversed these changes in patients with PPRE but showed no effect for patients with EoE, they said. This finding suggests that acid reflux might play a role in PPRE, but not in EOE, they concluded.
Dr. Katzka and his associates disclosed no funding sources and reported having no conflicts of interest. Dr. Rhijn and his associates were supported by the Netherlands Organization for Scientific Research. Two of Dr. Rhijn’s coauthors reported financial relationships with AstraZeneca, Endostim, Medical Measurement Systems, Shire, and GlaxoSmithKline.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Topical steroids seemed to improve mucosal integrity in patients with eosinophilic esophagitis, but proton pump inhibitor therapy did not.
Major finding: Mean spongiosis score was significantly lower among treated vs. untreated patients (0.4 vs. 1.3; P = .016).
Data source: Immunohistochemistry, histology, endoscopy, and mucosal functional analyses of 57 subjects in two separate studies.
Disclosures: Dr. Katzka and associates disclosed no funding sources and reported having no conflicts of interest. Dr. Rhijn and associates were supported by the Netherlands Organization for Scientific Research. Two of Dr. Rhijn’s coauthors reported financial relationships with AstraZeneca, Endostim, Medical Measurement Systems, Shire, and GlaxoSmithKline.
Miss the ear, and you may miss the diagnosis
A 52-year-old woman presented with pain in both ears associated with redness and swelling. The symptoms appeared 3 weeks earlier. The pain had started on one side, then spread to the other over a period of 2 weeks. She denied fever, chills, rigor, rash, or upper respiratory symptoms. She had experienced similar but unilateral ear pain months before. Her medical history included bilateral knee pain and swelling (treated as osteoarthritis), hypertension, hyperlipidemia, and hypothyroidism. She also reported progressive bilateral hearing loss, for which she now uses hearing aids. She had no history of conjunctivitis or uveitis.
Physical examination showed swelling and erythema of both ears, sparing the earlobes (Figure 1), as well as bilateral knee-joint tenderness and restricted joint movement. The erythrocyte sedimentation rate was elevated at 52 mm/h (reference range 0–20); the complete blood cell count, creatinine, and liver enzyme levels were normal. An autoimmune panel was negative for antinuclear antibody, antineutrophil cytoplasmic antibody, and rheumatoid factor.
A clinical diagnosis of relapsing polychondritis was made based on the McAdam criteria.1 The patient was initially started on steroids and then was maintained on methotrexate. Her symptoms improved dramatically by 3 weeks.
RELAPSING POLYCHONDRITIS
Relapsing polychondritis is a rare, chronic, and potentially multisystem disorder characterized by recurrent episodes of cartilaginous inflammation that often lead to progressive destruction of the cartilage.2,3
Auricular chondritis is the initial presentation in 43% of cases and eventually develops in 89% of patients.2,4 The earlobes are spared, as they are devoid of cartilage, and this feature helps to differentiate the condition from an infection.
If the condition is not treated, recurrent attacks can result in irreversible cartilage damage and drooping of the pinna (ie, “cauliflower ear”). Biopsy is usually avoided, as it may further damage the ear. The diagnostic criteria for relapsing polychondritis formulated by McAdam et al1 accommodate the different presentations in order to limit the need for biopsy. Systemic involvement may include external eye structures, vasculitis affecting the eighth cranial (vestibulocochlear) nerve, noninflammatory large-joint arthritis, and the trachea. There is also an association with myelodysplasia.
- McAdam LP, O’Hanlan MA, Bluestone R, Pearson CM. Relapsing polychondritis: prospective study of 23 patients and a review of the literature. Medicine (Baltimore) 1976; 55:193–215.
- Mathew SD, Battafarano DF, Morris MJ. Relapsing polychondritis in the Department of Defense population and review of the literature. Semin Arthritis Rheum 2012; 42:70–83.
- Letko E, Zafirakis P, Baltatzis S, Voudouri A, Livir-Rallatos C, Foster CS. Relapsing polychondritis: a clinical review. Semin Arthritis Rheum 2002; 31:384–395.
- Kent PD, Michet CJ, Luthra HS. Relapsing polychondritis. Curr Opin Rheumatol 2004; 16:56–61.
A 52-year-old woman presented with pain in both ears associated with redness and swelling. The symptoms appeared 3 weeks earlier. The pain had started on one side, then spread to the other over a period of 2 weeks. She denied fever, chills, rigor, rash, or upper respiratory symptoms. She had experienced similar but unilateral ear pain months before. Her medical history included bilateral knee pain and swelling (treated as osteoarthritis), hypertension, hyperlipidemia, and hypothyroidism. She also reported progressive bilateral hearing loss, for which she now uses hearing aids. She had no history of conjunctivitis or uveitis.
Physical examination showed swelling and erythema of both ears, sparing the earlobes (Figure 1), as well as bilateral knee-joint tenderness and restricted joint movement. The erythrocyte sedimentation rate was elevated at 52 mm/h (reference range 0–20); the complete blood cell count, creatinine, and liver enzyme levels were normal. An autoimmune panel was negative for antinuclear antibody, antineutrophil cytoplasmic antibody, and rheumatoid factor.
A clinical diagnosis of relapsing polychondritis was made based on the McAdam criteria.1 The patient was initially started on steroids and then was maintained on methotrexate. Her symptoms improved dramatically by 3 weeks.
RELAPSING POLYCHONDRITIS
Relapsing polychondritis is a rare, chronic, and potentially multisystem disorder characterized by recurrent episodes of cartilaginous inflammation that often lead to progressive destruction of the cartilage.2,3
Auricular chondritis is the initial presentation in 43% of cases and eventually develops in 89% of patients.2,4 The earlobes are spared, as they are devoid of cartilage, and this feature helps to differentiate the condition from an infection.
If the condition is not treated, recurrent attacks can result in irreversible cartilage damage and drooping of the pinna (ie, “cauliflower ear”). Biopsy is usually avoided, as it may further damage the ear. The diagnostic criteria for relapsing polychondritis formulated by McAdam et al1 accommodate the different presentations in order to limit the need for biopsy. Systemic involvement may include external eye structures, vasculitis affecting the eighth cranial (vestibulocochlear) nerve, noninflammatory large-joint arthritis, and the trachea. There is also an association with myelodysplasia.
A 52-year-old woman presented with pain in both ears associated with redness and swelling. The symptoms appeared 3 weeks earlier. The pain had started on one side, then spread to the other over a period of 2 weeks. She denied fever, chills, rigor, rash, or upper respiratory symptoms. She had experienced similar but unilateral ear pain months before. Her medical history included bilateral knee pain and swelling (treated as osteoarthritis), hypertension, hyperlipidemia, and hypothyroidism. She also reported progressive bilateral hearing loss, for which she now uses hearing aids. She had no history of conjunctivitis or uveitis.
Physical examination showed swelling and erythema of both ears, sparing the earlobes (Figure 1), as well as bilateral knee-joint tenderness and restricted joint movement. The erythrocyte sedimentation rate was elevated at 52 mm/h (reference range 0–20); the complete blood cell count, creatinine, and liver enzyme levels were normal. An autoimmune panel was negative for antinuclear antibody, antineutrophil cytoplasmic antibody, and rheumatoid factor.
A clinical diagnosis of relapsing polychondritis was made based on the McAdam criteria.1 The patient was initially started on steroids and then was maintained on methotrexate. Her symptoms improved dramatically by 3 weeks.
RELAPSING POLYCHONDRITIS
Relapsing polychondritis is a rare, chronic, and potentially multisystem disorder characterized by recurrent episodes of cartilaginous inflammation that often lead to progressive destruction of the cartilage.2,3
Auricular chondritis is the initial presentation in 43% of cases and eventually develops in 89% of patients.2,4 The earlobes are spared, as they are devoid of cartilage, and this feature helps to differentiate the condition from an infection.
If the condition is not treated, recurrent attacks can result in irreversible cartilage damage and drooping of the pinna (ie, “cauliflower ear”). Biopsy is usually avoided, as it may further damage the ear. The diagnostic criteria for relapsing polychondritis formulated by McAdam et al1 accommodate the different presentations in order to limit the need for biopsy. Systemic involvement may include external eye structures, vasculitis affecting the eighth cranial (vestibulocochlear) nerve, noninflammatory large-joint arthritis, and the trachea. There is also an association with myelodysplasia.
- McAdam LP, O’Hanlan MA, Bluestone R, Pearson CM. Relapsing polychondritis: prospective study of 23 patients and a review of the literature. Medicine (Baltimore) 1976; 55:193–215.
- Mathew SD, Battafarano DF, Morris MJ. Relapsing polychondritis in the Department of Defense population and review of the literature. Semin Arthritis Rheum 2012; 42:70–83.
- Letko E, Zafirakis P, Baltatzis S, Voudouri A, Livir-Rallatos C, Foster CS. Relapsing polychondritis: a clinical review. Semin Arthritis Rheum 2002; 31:384–395.
- Kent PD, Michet CJ, Luthra HS. Relapsing polychondritis. Curr Opin Rheumatol 2004; 16:56–61.
- McAdam LP, O’Hanlan MA, Bluestone R, Pearson CM. Relapsing polychondritis: prospective study of 23 patients and a review of the literature. Medicine (Baltimore) 1976; 55:193–215.
- Mathew SD, Battafarano DF, Morris MJ. Relapsing polychondritis in the Department of Defense population and review of the literature. Semin Arthritis Rheum 2012; 42:70–83.
- Letko E, Zafirakis P, Baltatzis S, Voudouri A, Livir-Rallatos C, Foster CS. Relapsing polychondritis: a clinical review. Semin Arthritis Rheum 2002; 31:384–395.
- Kent PD, Michet CJ, Luthra HS. Relapsing polychondritis. Curr Opin Rheumatol 2004; 16:56–61.
The 2014-2015 Influenza Season: What You Need to Know
As clinicians and the CDC prepare for the upcoming influenza season, many of the immunization recommendations remain unchanged from last season. Vaccination continues to be recommended for everyone ages 6 months and older. However, for the first time, a specific vaccine is preferred for children ages 2 through 8 years. Here’s what you need to know about this change, as well as how to handle vaccination in patients who are, or might be, allergic to eggs.
USE LAIV FOR KIDS AGES 2 THROUGH 8 (IF AVAILABLE)
For the first time, the CDC’s Advisory Committee on Immunization Practices (ACIP) has stated a preference for a specific influenza vaccine for a specific age-group. It recommends using the live attenuated influenza vaccine (LAIV), which is a nasal spray, for children ages 2 through 8 years.1
A systematic review found evidence of increased efficacy of LAIV compared to inactivated influenza vaccine (IIV) in this age-group; both types of vaccine have similar rates of adverse reactions.2 This increased effectiveness results in 46 fewer cases of confirmed influenza per 1,000 children vaccinated (number needed to treat, 24). Although the evidence of LAIV’s increased effectiveness was found for children ages 2 to 6 years, ACIP extended this recommendation through age 8 because this is the age through which clinicians need to consider two doses of vaccine for a child previously unvaccinated with the influenza vaccine. Children younger than 2 should receive IIV3 or IIV4.3
ACIP realizes that due to programmatic constraints it would be difficult to vaccinate all children with LAIV this year; the committee emphasizes that this recommendation should be implemented when feasible this year but no later than the 2015-2016 influenza season. IIV is effective in children and should be given if LAIV is not available or is contraindicated. Vaccination should not be delayed in the hopes of receiving a supply of LAIV if IIV is available.1
LAIV should not be used in children younger than 2 or adults older than 49. This vaccine is contraindicated in children and adolescents who are taking chronic aspirin therapy, pregnant women, or persons who are immunosuppressed, have a history of egg allergy, or have taken influenza antiviral medications in the past 48 hours.1 LAIV also is not recommended for children ages 2 through 4 years who have asthma or have had a wheezing episode in the past 12 months.1
There are precautions for the use of LAIV in patients with chronic medical conditions that can place them at high risk for complications from influenza. These include chronic lung, heart, renal, neurologic, liver, blood, or metabolic disorders—particularly, asthma and diabetes.1
WHICH VACCINE FOR PATIENTS WHO ARE ALLERGIC TO EGGS?
Two influenza vaccines are now available that are not prepared in embryonated eggs: recombinant influenza vaccine (RIV3) and cell culture–based inactivated influenza vaccine (ccIIV3). Both are trivalent products that contain antigens from two influenza A viruses and one influenza B virus; they were introduced in time for the 2013-2014 flu season. The RIV3 is considered egg-free but ccIIV3 is not, although the amount of egg protein in the latter is miniscule (estimated at 5 × 10-8 mg/0.5 mL dose).1 Neither product is licensed for use in children younger than 18, and RIV3 is licensed only for those ages 18 through 49.
Patients who experience only hives after egg exposure can receive any of the flu vaccines except LAIV—and only because of a lack of data on this product, not because it has been shown to be less safe than the other vaccines. Patients who are unsure if they have an egg allergy or who only get hives when they eat eggs should be observed for at least 30 minutes1 following injection as a precaution. Those ages 18 through 49 who have a history of severe reactions to eggs should receive RIV3. Patients younger than 18 and older than 49 can receive IIV vaccines approved for their specific age-group.
Any patient who is severely allergic but who cannot receive an egg-free vaccine should be vaccinated by a clinician with experience managing severe allergic conditions. Although severe anaphylactic reactions to influenza vaccine are very rare, clinicians should be equipped and prepared to respond to a severe allergic reaction after providing influenza vaccine to anyone with a history of egg allergy.
Continue for additional tips and resources >>
ADDITIONAL TIPS AND RESOURCES
In addition to the LAIV, RIV3, and ccIIV3 vaccines described here, 10 other vaccines are available: five egg-based IIV3 products in standard-dose form, one IIV3 vaccine for intradermal use, one high-dose IIV3 product for patients ages 65 or older, and three standard-dose IIV4 products. More details on each of these vaccines are available on the CDC website (www.cdc.gov/mmwr/preview/mmwrhtml/rr6207a1.htm?s_cid=rr6207a1_w#Tab1).
Regardless of which type of flu vaccine they receive, children ages 6 months through 8 years should receive two doses, at least four weeks apart, unless they received
• One dose during the 2013-2014 season, or
• Two or more doses of seasonal influenza vaccine since July 2010, or
• Two or more doses of seasonal influenza vaccine before July 2010 and at least one dose of monovalent H1N1 vaccine, or
• At least one dose of seasonal influenza vaccine prior to July 2010 and one or more after.
Vaccine effectiveness. The CDC estimated that vaccine effectiveness during the 2013-2014 flu season was 66%.3 While this degree of effectiveness is important for minimizing morbidity and mortality from influenza each year, it’s important to appreciate the limitations of the vaccine and not rely on it as the only preventive intervention.
Other forms of prevention. We need to advise and practice good respiratory hygiene, frequent hand washing, self-isolation when sick, effective infection control practices at health care facilities, targeted early treatment with antivirals, and targeted pre- and postexposure antiviral chemoprevention. Details on each of these interventions, including recommendations on the use of antiviral medications, can be found on the CDC website (www.cdc.gov/flu).
REFERENCES
1. Grohskopf LA, Olsen SJ, Sokolow LZ, et al; Influenza Division, National Center for Immunization and Respiratory Diseases, CDC. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States 2014-2015 influenza season. MMWR Morb Mortal Wkly Rep. 2014;63: 691-697.
2. Grohskopf L, Olsen S, Sokolow L. Effectiveness of live-attenuated vs inactivated influenza vaccines for healthy children. Presented at: Meeting of the Advisory Committee on Immunization Practices; February 26, 2014; Atlanta, GA. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2014-02/05-Flu-Grohskopf.pdf. Accessed October 19, 2014.
3. Flannery B. Interim estimates of 2013-14 seasonal influenza vaccine effectiveness. Presented at: Meeting of the Advisory Committee on Immunization Practices; February 26, 2014; Atlanta, GA. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2014-02/04-Flu-Flannery.pdf. Accessed October 19, 2014.
As clinicians and the CDC prepare for the upcoming influenza season, many of the immunization recommendations remain unchanged from last season. Vaccination continues to be recommended for everyone ages 6 months and older. However, for the first time, a specific vaccine is preferred for children ages 2 through 8 years. Here’s what you need to know about this change, as well as how to handle vaccination in patients who are, or might be, allergic to eggs.
USE LAIV FOR KIDS AGES 2 THROUGH 8 (IF AVAILABLE)
For the first time, the CDC’s Advisory Committee on Immunization Practices (ACIP) has stated a preference for a specific influenza vaccine for a specific age-group. It recommends using the live attenuated influenza vaccine (LAIV), which is a nasal spray, for children ages 2 through 8 years.1
A systematic review found evidence of increased efficacy of LAIV compared to inactivated influenza vaccine (IIV) in this age-group; both types of vaccine have similar rates of adverse reactions.2 This increased effectiveness results in 46 fewer cases of confirmed influenza per 1,000 children vaccinated (number needed to treat, 24). Although the evidence of LAIV’s increased effectiveness was found for children ages 2 to 6 years, ACIP extended this recommendation through age 8 because this is the age through which clinicians need to consider two doses of vaccine for a child previously unvaccinated with the influenza vaccine. Children younger than 2 should receive IIV3 or IIV4.3
ACIP realizes that due to programmatic constraints it would be difficult to vaccinate all children with LAIV this year; the committee emphasizes that this recommendation should be implemented when feasible this year but no later than the 2015-2016 influenza season. IIV is effective in children and should be given if LAIV is not available or is contraindicated. Vaccination should not be delayed in the hopes of receiving a supply of LAIV if IIV is available.1
LAIV should not be used in children younger than 2 or adults older than 49. This vaccine is contraindicated in children and adolescents who are taking chronic aspirin therapy, pregnant women, or persons who are immunosuppressed, have a history of egg allergy, or have taken influenza antiviral medications in the past 48 hours.1 LAIV also is not recommended for children ages 2 through 4 years who have asthma or have had a wheezing episode in the past 12 months.1
There are precautions for the use of LAIV in patients with chronic medical conditions that can place them at high risk for complications from influenza. These include chronic lung, heart, renal, neurologic, liver, blood, or metabolic disorders—particularly, asthma and diabetes.1
WHICH VACCINE FOR PATIENTS WHO ARE ALLERGIC TO EGGS?
Two influenza vaccines are now available that are not prepared in embryonated eggs: recombinant influenza vaccine (RIV3) and cell culture–based inactivated influenza vaccine (ccIIV3). Both are trivalent products that contain antigens from two influenza A viruses and one influenza B virus; they were introduced in time for the 2013-2014 flu season. The RIV3 is considered egg-free but ccIIV3 is not, although the amount of egg protein in the latter is miniscule (estimated at 5 × 10-8 mg/0.5 mL dose).1 Neither product is licensed for use in children younger than 18, and RIV3 is licensed only for those ages 18 through 49.
Patients who experience only hives after egg exposure can receive any of the flu vaccines except LAIV—and only because of a lack of data on this product, not because it has been shown to be less safe than the other vaccines. Patients who are unsure if they have an egg allergy or who only get hives when they eat eggs should be observed for at least 30 minutes1 following injection as a precaution. Those ages 18 through 49 who have a history of severe reactions to eggs should receive RIV3. Patients younger than 18 and older than 49 can receive IIV vaccines approved for their specific age-group.
Any patient who is severely allergic but who cannot receive an egg-free vaccine should be vaccinated by a clinician with experience managing severe allergic conditions. Although severe anaphylactic reactions to influenza vaccine are very rare, clinicians should be equipped and prepared to respond to a severe allergic reaction after providing influenza vaccine to anyone with a history of egg allergy.
Continue for additional tips and resources >>
ADDITIONAL TIPS AND RESOURCES
In addition to the LAIV, RIV3, and ccIIV3 vaccines described here, 10 other vaccines are available: five egg-based IIV3 products in standard-dose form, one IIV3 vaccine for intradermal use, one high-dose IIV3 product for patients ages 65 or older, and three standard-dose IIV4 products. More details on each of these vaccines are available on the CDC website (www.cdc.gov/mmwr/preview/mmwrhtml/rr6207a1.htm?s_cid=rr6207a1_w#Tab1).
Regardless of which type of flu vaccine they receive, children ages 6 months through 8 years should receive two doses, at least four weeks apart, unless they received
• One dose during the 2013-2014 season, or
• Two or more doses of seasonal influenza vaccine since July 2010, or
• Two or more doses of seasonal influenza vaccine before July 2010 and at least one dose of monovalent H1N1 vaccine, or
• At least one dose of seasonal influenza vaccine prior to July 2010 and one or more after.
Vaccine effectiveness. The CDC estimated that vaccine effectiveness during the 2013-2014 flu season was 66%.3 While this degree of effectiveness is important for minimizing morbidity and mortality from influenza each year, it’s important to appreciate the limitations of the vaccine and not rely on it as the only preventive intervention.
Other forms of prevention. We need to advise and practice good respiratory hygiene, frequent hand washing, self-isolation when sick, effective infection control practices at health care facilities, targeted early treatment with antivirals, and targeted pre- and postexposure antiviral chemoprevention. Details on each of these interventions, including recommendations on the use of antiviral medications, can be found on the CDC website (www.cdc.gov/flu).
REFERENCES
1. Grohskopf LA, Olsen SJ, Sokolow LZ, et al; Influenza Division, National Center for Immunization and Respiratory Diseases, CDC. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States 2014-2015 influenza season. MMWR Morb Mortal Wkly Rep. 2014;63: 691-697.
2. Grohskopf L, Olsen S, Sokolow L. Effectiveness of live-attenuated vs inactivated influenza vaccines for healthy children. Presented at: Meeting of the Advisory Committee on Immunization Practices; February 26, 2014; Atlanta, GA. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2014-02/05-Flu-Grohskopf.pdf. Accessed October 19, 2014.
3. Flannery B. Interim estimates of 2013-14 seasonal influenza vaccine effectiveness. Presented at: Meeting of the Advisory Committee on Immunization Practices; February 26, 2014; Atlanta, GA. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2014-02/04-Flu-Flannery.pdf. Accessed October 19, 2014.
As clinicians and the CDC prepare for the upcoming influenza season, many of the immunization recommendations remain unchanged from last season. Vaccination continues to be recommended for everyone ages 6 months and older. However, for the first time, a specific vaccine is preferred for children ages 2 through 8 years. Here’s what you need to know about this change, as well as how to handle vaccination in patients who are, or might be, allergic to eggs.
USE LAIV FOR KIDS AGES 2 THROUGH 8 (IF AVAILABLE)
For the first time, the CDC’s Advisory Committee on Immunization Practices (ACIP) has stated a preference for a specific influenza vaccine for a specific age-group. It recommends using the live attenuated influenza vaccine (LAIV), which is a nasal spray, for children ages 2 through 8 years.1
A systematic review found evidence of increased efficacy of LAIV compared to inactivated influenza vaccine (IIV) in this age-group; both types of vaccine have similar rates of adverse reactions.2 This increased effectiveness results in 46 fewer cases of confirmed influenza per 1,000 children vaccinated (number needed to treat, 24). Although the evidence of LAIV’s increased effectiveness was found for children ages 2 to 6 years, ACIP extended this recommendation through age 8 because this is the age through which clinicians need to consider two doses of vaccine for a child previously unvaccinated with the influenza vaccine. Children younger than 2 should receive IIV3 or IIV4.3
ACIP realizes that due to programmatic constraints it would be difficult to vaccinate all children with LAIV this year; the committee emphasizes that this recommendation should be implemented when feasible this year but no later than the 2015-2016 influenza season. IIV is effective in children and should be given if LAIV is not available or is contraindicated. Vaccination should not be delayed in the hopes of receiving a supply of LAIV if IIV is available.1
LAIV should not be used in children younger than 2 or adults older than 49. This vaccine is contraindicated in children and adolescents who are taking chronic aspirin therapy, pregnant women, or persons who are immunosuppressed, have a history of egg allergy, or have taken influenza antiviral medications in the past 48 hours.1 LAIV also is not recommended for children ages 2 through 4 years who have asthma or have had a wheezing episode in the past 12 months.1
There are precautions for the use of LAIV in patients with chronic medical conditions that can place them at high risk for complications from influenza. These include chronic lung, heart, renal, neurologic, liver, blood, or metabolic disorders—particularly, asthma and diabetes.1
WHICH VACCINE FOR PATIENTS WHO ARE ALLERGIC TO EGGS?
Two influenza vaccines are now available that are not prepared in embryonated eggs: recombinant influenza vaccine (RIV3) and cell culture–based inactivated influenza vaccine (ccIIV3). Both are trivalent products that contain antigens from two influenza A viruses and one influenza B virus; they were introduced in time for the 2013-2014 flu season. The RIV3 is considered egg-free but ccIIV3 is not, although the amount of egg protein in the latter is miniscule (estimated at 5 × 10-8 mg/0.5 mL dose).1 Neither product is licensed for use in children younger than 18, and RIV3 is licensed only for those ages 18 through 49.
Patients who experience only hives after egg exposure can receive any of the flu vaccines except LAIV—and only because of a lack of data on this product, not because it has been shown to be less safe than the other vaccines. Patients who are unsure if they have an egg allergy or who only get hives when they eat eggs should be observed for at least 30 minutes1 following injection as a precaution. Those ages 18 through 49 who have a history of severe reactions to eggs should receive RIV3. Patients younger than 18 and older than 49 can receive IIV vaccines approved for their specific age-group.
Any patient who is severely allergic but who cannot receive an egg-free vaccine should be vaccinated by a clinician with experience managing severe allergic conditions. Although severe anaphylactic reactions to influenza vaccine are very rare, clinicians should be equipped and prepared to respond to a severe allergic reaction after providing influenza vaccine to anyone with a history of egg allergy.
Continue for additional tips and resources >>
ADDITIONAL TIPS AND RESOURCES
In addition to the LAIV, RIV3, and ccIIV3 vaccines described here, 10 other vaccines are available: five egg-based IIV3 products in standard-dose form, one IIV3 vaccine for intradermal use, one high-dose IIV3 product for patients ages 65 or older, and three standard-dose IIV4 products. More details on each of these vaccines are available on the CDC website (www.cdc.gov/mmwr/preview/mmwrhtml/rr6207a1.htm?s_cid=rr6207a1_w#Tab1).
Regardless of which type of flu vaccine they receive, children ages 6 months through 8 years should receive two doses, at least four weeks apart, unless they received
• One dose during the 2013-2014 season, or
• Two or more doses of seasonal influenza vaccine since July 2010, or
• Two or more doses of seasonal influenza vaccine before July 2010 and at least one dose of monovalent H1N1 vaccine, or
• At least one dose of seasonal influenza vaccine prior to July 2010 and one or more after.
Vaccine effectiveness. The CDC estimated that vaccine effectiveness during the 2013-2014 flu season was 66%.3 While this degree of effectiveness is important for minimizing morbidity and mortality from influenza each year, it’s important to appreciate the limitations of the vaccine and not rely on it as the only preventive intervention.
Other forms of prevention. We need to advise and practice good respiratory hygiene, frequent hand washing, self-isolation when sick, effective infection control practices at health care facilities, targeted early treatment with antivirals, and targeted pre- and postexposure antiviral chemoprevention. Details on each of these interventions, including recommendations on the use of antiviral medications, can be found on the CDC website (www.cdc.gov/flu).
REFERENCES
1. Grohskopf LA, Olsen SJ, Sokolow LZ, et al; Influenza Division, National Center for Immunization and Respiratory Diseases, CDC. Prevention and control of seasonal influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP)—United States 2014-2015 influenza season. MMWR Morb Mortal Wkly Rep. 2014;63: 691-697.
2. Grohskopf L, Olsen S, Sokolow L. Effectiveness of live-attenuated vs inactivated influenza vaccines for healthy children. Presented at: Meeting of the Advisory Committee on Immunization Practices; February 26, 2014; Atlanta, GA. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2014-02/05-Flu-Grohskopf.pdf. Accessed October 19, 2014.
3. Flannery B. Interim estimates of 2013-14 seasonal influenza vaccine effectiveness. Presented at: Meeting of the Advisory Committee on Immunization Practices; February 26, 2014; Atlanta, GA. www.cdc.gov/vaccines/acip/meetings/downloads/slides-2014-02/04-Flu-Flannery.pdf. Accessed October 19, 2014.
Rosacea’s Comorbidities Are More Than Skin Deep
AMSTERDAM – Rosacea is associated with increased risk of a range of chronic systemic diseases, including allergies and urogential disorders, a case-control study showed.
The common denominator among this linked diverse collection of diseases is probably underlying systemic inflammation, Dr. Barbara M. Rainer explained at the annual congress of the European Academy of Dermatology and Venereology. But regardless of the pathophysiologic mechanisms at work, the important thing is that physicians be on the lookout for these comorbid conditions in their patients with rosacea.
Dr. Rainer of Johns Hopkins University in Baltimore presented a case-control study involving 130 subjects: 65 rosacea patients and an equal number of controls matched for age, sex, and race.
The most common comorbidity was food allergies (odds ratio, 10), followed by urogenital disorders (OR, 7.5).
The rosacea patients averaged 50 years of age and had a mean 11.8-year history of their skin disease. Body mass index, smoking status, alcohol intake, and coffee consumption were similar in cases and controls. Two-thirds of subjects were women. Relative risks for comorbid conditions were calculated using logistic regression analysis.
Dr. Rainer reported no relevant financial conflicts.
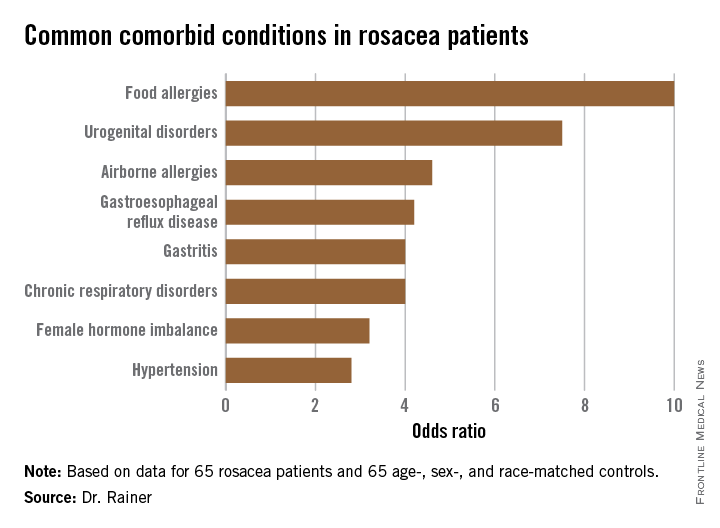
AMSTERDAM – Rosacea is associated with increased risk of a range of chronic systemic diseases, including allergies and urogential disorders, a case-control study showed.
The common denominator among this linked diverse collection of diseases is probably underlying systemic inflammation, Dr. Barbara M. Rainer explained at the annual congress of the European Academy of Dermatology and Venereology. But regardless of the pathophysiologic mechanisms at work, the important thing is that physicians be on the lookout for these comorbid conditions in their patients with rosacea.
Dr. Rainer of Johns Hopkins University in Baltimore presented a case-control study involving 130 subjects: 65 rosacea patients and an equal number of controls matched for age, sex, and race.
The most common comorbidity was food allergies (odds ratio, 10), followed by urogenital disorders (OR, 7.5).
The rosacea patients averaged 50 years of age and had a mean 11.8-year history of their skin disease. Body mass index, smoking status, alcohol intake, and coffee consumption were similar in cases and controls. Two-thirds of subjects were women. Relative risks for comorbid conditions were calculated using logistic regression analysis.
Dr. Rainer reported no relevant financial conflicts.

AMSTERDAM – Rosacea is associated with increased risk of a range of chronic systemic diseases, including allergies and urogential disorders, a case-control study showed.
The common denominator among this linked diverse collection of diseases is probably underlying systemic inflammation, Dr. Barbara M. Rainer explained at the annual congress of the European Academy of Dermatology and Venereology. But regardless of the pathophysiologic mechanisms at work, the important thing is that physicians be on the lookout for these comorbid conditions in their patients with rosacea.
Dr. Rainer of Johns Hopkins University in Baltimore presented a case-control study involving 130 subjects: 65 rosacea patients and an equal number of controls matched for age, sex, and race.
The most common comorbidity was food allergies (odds ratio, 10), followed by urogenital disorders (OR, 7.5).
The rosacea patients averaged 50 years of age and had a mean 11.8-year history of their skin disease. Body mass index, smoking status, alcohol intake, and coffee consumption were similar in cases and controls. Two-thirds of subjects were women. Relative risks for comorbid conditions were calculated using logistic regression analysis.
Dr. Rainer reported no relevant financial conflicts.

AT THE EADV CONGRESS
To dream the maybe possible dream: A breast cancer vaccine
The Journal generally does not publish articles on topics not yet clinically relevant. But since the topic of immunization is so often in the news, and since immunotherapeutic strategies against cancer continue to be tested in clinical trials, we decided to include in this issue an edited transcript of a Medicine Grand Rounds presentation at Cleveland Clinic by Dr. Vincent Tuohy on a novel strategy to develop a vaccine against a particularly virulent form of breast cancer.
The immune system’s response to cancer is complex. Melanoma and renal cell carcinoma seem particularly susceptible to suppression by the native or augmented immune response. But most cancers seem to grow—and many metastasize—seemingly unaffected by our immune system, and sometimes even in the presence of detectable antitumor cell-directed lymphocytes and antibodies. Many attempts at devising human antitumor vaccines and immunotherapies have failed. On the other hand, we have seen the successful development of effective monoclonal antibody therapies (eg, rituximab for B cell lymphoma), immunomodulatory treatments for patients with advanced disease, and vaccines against viruses that cause cancer, ie, human papillomavirus and hepatitis B.
To fully appreciate the nuances of Dr. Tuohy’s proposed strategy, which has not yet been tested in clinical trials, and the complexities of tumor immunology, a very brief primer on the challenges is in order.
CHALLENGES TO DEVELOPING CANCER IMMUNOTHERAPY
Solid tumors can be triggered by multiple mechanisms, alone or in combination, including viruses, spontaneous mutations, overexpression of tumor promoters, and underexpression of tumor suppressors. Once growing, the solid tumor establishes its own rogue growth community, complete with a new infrastructure to supply nutrition and oxygen, potential means for expansion locally and distally, and a system to defend itself from the body’s immune system. The last of these poses specific challenges to successful spontaneous immune surveillance and to immunotherapy designed to kill cancer cells.
Microbial pathogens trigger both a nonspecific (innate) and a specific immune response in the human body. Initially, the immune system is nonspecifically “revved up,” triggered by shared “danger signals” associated with the perceived pathogen and its specific antigens. Then, specialized cells including dendritic cells locally and in proximate lymph nodes are primed to present the de novo antigens in a way that generates a specific and maturing immune response capable of getting rid of the pathogen. Tumors are also pathogenic and in some ways “foreign.” However, they are also similar to normal tissue and interact quite differently with the immune response in ways that enhance their likelihood of growth and survival. Tumor cells often do not send a danger signal to the immune system akin to what is generated by a staphylococcal or mycobacterial invader.
Tumor cells express specific antigens on their surface, such as viral proteins, cancer-associated mutated proteins, and overexpressed differentiated or undifferentiated antigens, including in some cases what Dr. Tuohy discusses as “retired proteins.” But some of these antigens may also be expressed in normal tissues, especially in an environment of resolving inflammation. Some signal the immune system to down-regulate what could otherwise be a vigorous self-destructive response every time there was inflammation.
The dendritic cell activation of the potential antitumor T-cell response and the antitumor T-cell response itself seem to be systematically blunted by many tumors. Reversal of this blunting represents one strategy currently used with very modest clinical success in treating advanced melanoma. Some newly generated tumor-antigen–recognizing T cells may in fact exert suppressor (or regulator) and not cytotoxic activity. Some tumors exhibit systemic immunosuppressive activity; this can be manifested not only by unchecked tumor growth, but also by an increased susceptibility to certain infections.
PITFALLS OF MESSING WITH THE IMMUNE SYSTEM
Messing with the immune system is not without pitfalls. Not all toxicities will be predicted by preclinical animal studies, and human immunity is not a mirror image of rodents’ or even other primates’ immune systems. Augmentation of an antitumor response, in part from the interplay of the complexities noted above, may lead to destruction of normal tissue elsewhere, or even to disruption of tolerance with the expression of autoimmunity. The toxicities will be different than the somewhat predictable toxicities from traditional antiproliferative chemotherapies—witness the striking systemic toxicity from interleukin 2-based therapies.
Whether Dr. Tuohy’s approach to developing a tumor vaccine will ultimately reach our formularies remains to be seen. The work is in an extremely preliminary phase. But the concept of immunotherapy for cancer remains an active area of research that is worth keeping an eye on.
The Journal generally does not publish articles on topics not yet clinically relevant. But since the topic of immunization is so often in the news, and since immunotherapeutic strategies against cancer continue to be tested in clinical trials, we decided to include in this issue an edited transcript of a Medicine Grand Rounds presentation at Cleveland Clinic by Dr. Vincent Tuohy on a novel strategy to develop a vaccine against a particularly virulent form of breast cancer.
The immune system’s response to cancer is complex. Melanoma and renal cell carcinoma seem particularly susceptible to suppression by the native or augmented immune response. But most cancers seem to grow—and many metastasize—seemingly unaffected by our immune system, and sometimes even in the presence of detectable antitumor cell-directed lymphocytes and antibodies. Many attempts at devising human antitumor vaccines and immunotherapies have failed. On the other hand, we have seen the successful development of effective monoclonal antibody therapies (eg, rituximab for B cell lymphoma), immunomodulatory treatments for patients with advanced disease, and vaccines against viruses that cause cancer, ie, human papillomavirus and hepatitis B.
To fully appreciate the nuances of Dr. Tuohy’s proposed strategy, which has not yet been tested in clinical trials, and the complexities of tumor immunology, a very brief primer on the challenges is in order.
CHALLENGES TO DEVELOPING CANCER IMMUNOTHERAPY
Solid tumors can be triggered by multiple mechanisms, alone or in combination, including viruses, spontaneous mutations, overexpression of tumor promoters, and underexpression of tumor suppressors. Once growing, the solid tumor establishes its own rogue growth community, complete with a new infrastructure to supply nutrition and oxygen, potential means for expansion locally and distally, and a system to defend itself from the body’s immune system. The last of these poses specific challenges to successful spontaneous immune surveillance and to immunotherapy designed to kill cancer cells.
Microbial pathogens trigger both a nonspecific (innate) and a specific immune response in the human body. Initially, the immune system is nonspecifically “revved up,” triggered by shared “danger signals” associated with the perceived pathogen and its specific antigens. Then, specialized cells including dendritic cells locally and in proximate lymph nodes are primed to present the de novo antigens in a way that generates a specific and maturing immune response capable of getting rid of the pathogen. Tumors are also pathogenic and in some ways “foreign.” However, they are also similar to normal tissue and interact quite differently with the immune response in ways that enhance their likelihood of growth and survival. Tumor cells often do not send a danger signal to the immune system akin to what is generated by a staphylococcal or mycobacterial invader.
Tumor cells express specific antigens on their surface, such as viral proteins, cancer-associated mutated proteins, and overexpressed differentiated or undifferentiated antigens, including in some cases what Dr. Tuohy discusses as “retired proteins.” But some of these antigens may also be expressed in normal tissues, especially in an environment of resolving inflammation. Some signal the immune system to down-regulate what could otherwise be a vigorous self-destructive response every time there was inflammation.
The dendritic cell activation of the potential antitumor T-cell response and the antitumor T-cell response itself seem to be systematically blunted by many tumors. Reversal of this blunting represents one strategy currently used with very modest clinical success in treating advanced melanoma. Some newly generated tumor-antigen–recognizing T cells may in fact exert suppressor (or regulator) and not cytotoxic activity. Some tumors exhibit systemic immunosuppressive activity; this can be manifested not only by unchecked tumor growth, but also by an increased susceptibility to certain infections.
PITFALLS OF MESSING WITH THE IMMUNE SYSTEM
Messing with the immune system is not without pitfalls. Not all toxicities will be predicted by preclinical animal studies, and human immunity is not a mirror image of rodents’ or even other primates’ immune systems. Augmentation of an antitumor response, in part from the interplay of the complexities noted above, may lead to destruction of normal tissue elsewhere, or even to disruption of tolerance with the expression of autoimmunity. The toxicities will be different than the somewhat predictable toxicities from traditional antiproliferative chemotherapies—witness the striking systemic toxicity from interleukin 2-based therapies.
Whether Dr. Tuohy’s approach to developing a tumor vaccine will ultimately reach our formularies remains to be seen. The work is in an extremely preliminary phase. But the concept of immunotherapy for cancer remains an active area of research that is worth keeping an eye on.
The Journal generally does not publish articles on topics not yet clinically relevant. But since the topic of immunization is so often in the news, and since immunotherapeutic strategies against cancer continue to be tested in clinical trials, we decided to include in this issue an edited transcript of a Medicine Grand Rounds presentation at Cleveland Clinic by Dr. Vincent Tuohy on a novel strategy to develop a vaccine against a particularly virulent form of breast cancer.
The immune system’s response to cancer is complex. Melanoma and renal cell carcinoma seem particularly susceptible to suppression by the native or augmented immune response. But most cancers seem to grow—and many metastasize—seemingly unaffected by our immune system, and sometimes even in the presence of detectable antitumor cell-directed lymphocytes and antibodies. Many attempts at devising human antitumor vaccines and immunotherapies have failed. On the other hand, we have seen the successful development of effective monoclonal antibody therapies (eg, rituximab for B cell lymphoma), immunomodulatory treatments for patients with advanced disease, and vaccines against viruses that cause cancer, ie, human papillomavirus and hepatitis B.
To fully appreciate the nuances of Dr. Tuohy’s proposed strategy, which has not yet been tested in clinical trials, and the complexities of tumor immunology, a very brief primer on the challenges is in order.
CHALLENGES TO DEVELOPING CANCER IMMUNOTHERAPY
Solid tumors can be triggered by multiple mechanisms, alone or in combination, including viruses, spontaneous mutations, overexpression of tumor promoters, and underexpression of tumor suppressors. Once growing, the solid tumor establishes its own rogue growth community, complete with a new infrastructure to supply nutrition and oxygen, potential means for expansion locally and distally, and a system to defend itself from the body’s immune system. The last of these poses specific challenges to successful spontaneous immune surveillance and to immunotherapy designed to kill cancer cells.
Microbial pathogens trigger both a nonspecific (innate) and a specific immune response in the human body. Initially, the immune system is nonspecifically “revved up,” triggered by shared “danger signals” associated with the perceived pathogen and its specific antigens. Then, specialized cells including dendritic cells locally and in proximate lymph nodes are primed to present the de novo antigens in a way that generates a specific and maturing immune response capable of getting rid of the pathogen. Tumors are also pathogenic and in some ways “foreign.” However, they are also similar to normal tissue and interact quite differently with the immune response in ways that enhance their likelihood of growth and survival. Tumor cells often do not send a danger signal to the immune system akin to what is generated by a staphylococcal or mycobacterial invader.
Tumor cells express specific antigens on their surface, such as viral proteins, cancer-associated mutated proteins, and overexpressed differentiated or undifferentiated antigens, including in some cases what Dr. Tuohy discusses as “retired proteins.” But some of these antigens may also be expressed in normal tissues, especially in an environment of resolving inflammation. Some signal the immune system to down-regulate what could otherwise be a vigorous self-destructive response every time there was inflammation.
The dendritic cell activation of the potential antitumor T-cell response and the antitumor T-cell response itself seem to be systematically blunted by many tumors. Reversal of this blunting represents one strategy currently used with very modest clinical success in treating advanced melanoma. Some newly generated tumor-antigen–recognizing T cells may in fact exert suppressor (or regulator) and not cytotoxic activity. Some tumors exhibit systemic immunosuppressive activity; this can be manifested not only by unchecked tumor growth, but also by an increased susceptibility to certain infections.
PITFALLS OF MESSING WITH THE IMMUNE SYSTEM
Messing with the immune system is not without pitfalls. Not all toxicities will be predicted by preclinical animal studies, and human immunity is not a mirror image of rodents’ or even other primates’ immune systems. Augmentation of an antitumor response, in part from the interplay of the complexities noted above, may lead to destruction of normal tissue elsewhere, or even to disruption of tolerance with the expression of autoimmunity. The toxicities will be different than the somewhat predictable toxicities from traditional antiproliferative chemotherapies—witness the striking systemic toxicity from interleukin 2-based therapies.
Whether Dr. Tuohy’s approach to developing a tumor vaccine will ultimately reach our formularies remains to be seen. The work is in an extremely preliminary phase. But the concept of immunotherapy for cancer remains an active area of research that is worth keeping an eye on.
Bench-to-bedside challenges in developing immune protection against breast cancer
The most proven, effective way to control disease is through prophylactic vaccination. The childhood vaccination program is a testament to this disease prevention approach, and in its current form protects us from diseases caused by 16 different pathogens.1
Childhood immunization ends in the teen years with recommended vaccination against multiple strains of human papillomavirus that are associated with several cancers, most notably cervical carcinoma.2 However, even though we have known for over 30 years that the immune system can provide considerable vaccine-induced protection against the development of cancer,3 we have not produced any vaccines that prevent cancers that commonly occur with age, such as breast and prostate cancer, which afflict 1 of 8 women and 1 of 6 men, respectively.4,5
The lack of an adult vaccine program that provides protection against such commonly occurring adult-onset cancers represents a glaring health care deficiency and a challenge for this current generation to protect coming generations.
THE ‘RETIRED’ PROTEIN HYPOTHESIS
Given that most cancers are not associated with any disease-inducing pathogens, at what targets can we aim our immune system to induce safe and effective protection against these commonly occurring adult-onset cancers?
Perhaps an understanding of the natural aging process may provide us with insights regarding possible vaccine targets. As we age, there is a decline in expression of many tissue-specific proteins, often to the point where they may be considered “retired” and no longer found at detectable or immunogenic levels in normal cells. Examples of this natural aging process include the pigment proteins as our hair whitens, certain lactation proteins when breastfeeding ceases, and some ovarian proteins as menopause begins and production of mature egg follicles ceases. If these retired proteins are expressed in invigorated emerging tumors, then preemptive immunity directed against these retired proteins would attack and destroy the emerging tumors and ignore normal tissues, thereby avoiding any complicating collateral autoimmune damage.
Thus, we propose that retired tissue-specific self-proteins may substitute for unavailable pathogens as targets for mediating safe and effective immune protection against adult-onset cancers such as breast cancer.
SAFE AND EFFECTIVE PREVENTION OF BREAST CANCER IN MICE
To test this retired-protein hypothesis for immunoprevention of breast cancer, we selected alpha-lactalbumin as our vaccine target, for two reasons:
- Alpha-lactalbumin is a protein expressed exclusively in lactating breast tissue and is not expressed at immunogenic levels in either normal nonlactating breast tissues or in any of 78 other normal human tissues examined.6–8
- Alpha-lactalbumin is expressed in most human triple-negative breast cancers (TNBC),9,10 the most aggressive and lethal form of breast cancer, and the predominant form that occurs in women with mutations in the breast cancer 1, early-onset gene (BRCA1).11,12
We found that alpha-lactalbumin vaccination consistently inhibited the formation and growth of breast tumors in three different mouse models commonly used in breast cancer research.13 More importantly, the observed immune protection against the development of breast cancer in mice occurred in the absence of any detectable autoimmune inflammatory damage in any normal tissues examined. Thus, we concluded that alpha-lactalbumin vaccination could provide healthy women with safe and effective immune protection against the more malignant forms of breast cancer.
FROM BENCH TO BEDSIDE
How then do we determine whether alpha-lactalbumin vaccination prevents the development of TNBC in otherwise healthy cancer-free women, and whether it prevents recurrence of TNBC in women already diagnosed with TNBC? Our initial approach will involve two phase 1 clinical trials designed to determine the safety of the vaccine as well as the dose and number of vaccinations needed to induce optimum tumor immunity.
The first (phase 1a) trial will involve vaccination of women recently diagnosed with TNBC who have recovered with the current standard of care. These women will be vaccinated in groups receiving various doses of both recombinant human alpha-lactalbumin and an appropriate immune adjuvant that activates the immune system so it responds aggressively to the alpha-lactalbumin and creates the proinflammatory T-cell response needed for effective tumor immunity. This trial will simply provide dosage and safety profiles of the vaccine and will thereby lay the groundwork for subsequent (phase 2 and 3) trials designed to determine whether alpha-lactalbumin vaccination is effective in preventing recurrence of TNBC in women already diagnosed with this disease.
The dosage and number of immunizations shown to provide optimum immunity in the phase 1a trial will be used in a second (phase 1b) trial designed primarily to determine the safety of alpha-lactalbumin vaccination in healthy cancer-free women who have elected to undergo voluntary prophylactic mastectomy to reduce their breast cancer risk. Most of the women who elect to have this surgery have an established family history of breast cancer or a known BRCA1 mutation associated with high breast cancer risk, or both.11,12 Consenting women will be vaccinated against alpha-lactalbumin several months before their mastectomy, and their surgically removed breast tissues will be examined extensively for signs of vaccine-induced autoimmune damage. Thus, this trial will determine the safety of alpha-lactalbumin vaccination in healthy cancer-free women and will lay the groundwork for subsequent phase 2 and 3 trials designed to determine whether alpha-lactalbumin vaccination is effective in preventing TNBC in women at high risk of developing this form of breast cancer.
We estimate that completing our preclinical studies, obtaining permission from the US Food and Drug Administration to test our investigational new drug, and completing both phase 1 clinical trials will require about 5 years. Thereafter, completion of phase 2 and 3 trials designed to prevent both recurrence of TNBC in women already diagnosed with this disease and occurrence of TNBC in otherwise healthy, cancer-free women will likely take at least another 5 years, so that this vaccine will likely not be available to the general public before 2024.
TO SUM UP
Although our immune system is potentially capable of protecting us from some cancers, we currently have no immune protection against cancers we commonly confront as we age. We propose that tissue-specific self proteins that are retired from expression with age in normal tissues but are expressed at immunogenic levels in emerging tumors may substitute for unavailable pathogens as targets for immunoprevention of adult-onset cancers that commonly occur with age. We know that the retired breast-specific protein, alpha-lactalbumin, is overexpressed in TNBC and that vaccination with alpha-lactalbumin provides safe and effective protection from breast cancer in preclinical mouse studies. Clinical trials are planned to ultimately determine whether alpha-lactalbumin vaccination can prevent recurrence of TNBC in women already diagnosed with this disease and prevent the initiation of TNBC in women at high risk of developing this most aggressive and lethal form of breast cancer.
Acknowledgment: This work was supported by a grant from Shield Biotech, Inc., Cleveland, OH. In addition, the author wishes to recognize and express his sincere gratitude for the support and encouragement received from numerous organizations that have been instrumental in making this work possible, including November Philanthropy, Brakes for Breasts, the Breast Health and Healing Foundation, the Toni Turchi Foundation, the Coalition of Women Who Care About Breast Cancer, the Sisters for Prevention, the Previvors and Survivors, the Champions of the Pink Vaccine, the Race at Legacy Village, the National Greek Orthodox Ladies Philopto-chos Society, the Daughters of Penelope Icarus Chapter 321, Can’t Stop Won’t Stop, the Babylon Breast Cancer Coalition, and Walk With A Doc.
- Centers for Disease Control and Prevention. Immunization schedules. www.cdc.gov/vaccines/schedules/. Accessed September 4, 2014.
- Schiller JT, Lowy DR. Understanding and learning from the success of prophylactic human papillomavirus vaccines. Nat Rev Microbiol 2012; 10:681–692.
- Van Pel A, Boon T. Protection against a nonimmunogenic mouse leukemia by an immunogenic variant obtained by mutagenesis. Proc Natl Acad Sci USA 1982; 79:4718–4722.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013; 63:11–30.
- National Cancer Institute. Surveillance, Epidemiology, and End Results (SEER) Program. Previous version: SEER cancer statistics review 1975–2010. http://seer.cancer.gov/csr/1975_2010/. Accessed September 4, 2014.
- Uhlen M, Oksvold P, Fagerberg L, et al. Towards a knowledge-based human protein atlas. Nat Biotechnol 2010; 28:1248–1250.
- Pontén F, Gry M, Fagerberg L, et al. A global view of protein expression in human cells, tissues, and organs. Mol Syst Biol 2009; 5:337.
- The Human Protein Atlas. www.proteinatlas.org. Accessed September 4, 2014.
- Rhodes DR, Yu J, Shanker K, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 2004; 6:1–6.
- ONCOMINEdatabase. www.oncomine.org/resource/login.html. Accessed September 4, 2014.
- Atchley DP, Albarracin CT, Lopez A, et al. Clinical and pathologic characteristics of patients with BRCA-positive and BRCA-negative breast cancer. J Clin Oncol 2008; 26:4282–4288.
- Comen E, Davids M, Kirchhoff T, Hudis C, Offit K, Robson M. Relative contributions of BRCA1 and BRCA2 mutations to “triple-negative” breast cancer in Ashkenazi women. Breast Cancer Res Treat 2011; 129:185–190.
- Jaini R, Kesaraju P, Johnson JM, Altuntas CZ, Jane-Wit D, Tuohy VK. An autoimmune-mediated strategy for prophylactic breast cancer vaccination. Nat Med 2010; 16:799–803.
The most proven, effective way to control disease is through prophylactic vaccination. The childhood vaccination program is a testament to this disease prevention approach, and in its current form protects us from diseases caused by 16 different pathogens.1
Childhood immunization ends in the teen years with recommended vaccination against multiple strains of human papillomavirus that are associated with several cancers, most notably cervical carcinoma.2 However, even though we have known for over 30 years that the immune system can provide considerable vaccine-induced protection against the development of cancer,3 we have not produced any vaccines that prevent cancers that commonly occur with age, such as breast and prostate cancer, which afflict 1 of 8 women and 1 of 6 men, respectively.4,5
The lack of an adult vaccine program that provides protection against such commonly occurring adult-onset cancers represents a glaring health care deficiency and a challenge for this current generation to protect coming generations.
THE ‘RETIRED’ PROTEIN HYPOTHESIS
Given that most cancers are not associated with any disease-inducing pathogens, at what targets can we aim our immune system to induce safe and effective protection against these commonly occurring adult-onset cancers?
Perhaps an understanding of the natural aging process may provide us with insights regarding possible vaccine targets. As we age, there is a decline in expression of many tissue-specific proteins, often to the point where they may be considered “retired” and no longer found at detectable or immunogenic levels in normal cells. Examples of this natural aging process include the pigment proteins as our hair whitens, certain lactation proteins when breastfeeding ceases, and some ovarian proteins as menopause begins and production of mature egg follicles ceases. If these retired proteins are expressed in invigorated emerging tumors, then preemptive immunity directed against these retired proteins would attack and destroy the emerging tumors and ignore normal tissues, thereby avoiding any complicating collateral autoimmune damage.
Thus, we propose that retired tissue-specific self-proteins may substitute for unavailable pathogens as targets for mediating safe and effective immune protection against adult-onset cancers such as breast cancer.
SAFE AND EFFECTIVE PREVENTION OF BREAST CANCER IN MICE
To test this retired-protein hypothesis for immunoprevention of breast cancer, we selected alpha-lactalbumin as our vaccine target, for two reasons:
- Alpha-lactalbumin is a protein expressed exclusively in lactating breast tissue and is not expressed at immunogenic levels in either normal nonlactating breast tissues or in any of 78 other normal human tissues examined.6–8
- Alpha-lactalbumin is expressed in most human triple-negative breast cancers (TNBC),9,10 the most aggressive and lethal form of breast cancer, and the predominant form that occurs in women with mutations in the breast cancer 1, early-onset gene (BRCA1).11,12
We found that alpha-lactalbumin vaccination consistently inhibited the formation and growth of breast tumors in three different mouse models commonly used in breast cancer research.13 More importantly, the observed immune protection against the development of breast cancer in mice occurred in the absence of any detectable autoimmune inflammatory damage in any normal tissues examined. Thus, we concluded that alpha-lactalbumin vaccination could provide healthy women with safe and effective immune protection against the more malignant forms of breast cancer.
FROM BENCH TO BEDSIDE
How then do we determine whether alpha-lactalbumin vaccination prevents the development of TNBC in otherwise healthy cancer-free women, and whether it prevents recurrence of TNBC in women already diagnosed with TNBC? Our initial approach will involve two phase 1 clinical trials designed to determine the safety of the vaccine as well as the dose and number of vaccinations needed to induce optimum tumor immunity.
The first (phase 1a) trial will involve vaccination of women recently diagnosed with TNBC who have recovered with the current standard of care. These women will be vaccinated in groups receiving various doses of both recombinant human alpha-lactalbumin and an appropriate immune adjuvant that activates the immune system so it responds aggressively to the alpha-lactalbumin and creates the proinflammatory T-cell response needed for effective tumor immunity. This trial will simply provide dosage and safety profiles of the vaccine and will thereby lay the groundwork for subsequent (phase 2 and 3) trials designed to determine whether alpha-lactalbumin vaccination is effective in preventing recurrence of TNBC in women already diagnosed with this disease.
The dosage and number of immunizations shown to provide optimum immunity in the phase 1a trial will be used in a second (phase 1b) trial designed primarily to determine the safety of alpha-lactalbumin vaccination in healthy cancer-free women who have elected to undergo voluntary prophylactic mastectomy to reduce their breast cancer risk. Most of the women who elect to have this surgery have an established family history of breast cancer or a known BRCA1 mutation associated with high breast cancer risk, or both.11,12 Consenting women will be vaccinated against alpha-lactalbumin several months before their mastectomy, and their surgically removed breast tissues will be examined extensively for signs of vaccine-induced autoimmune damage. Thus, this trial will determine the safety of alpha-lactalbumin vaccination in healthy cancer-free women and will lay the groundwork for subsequent phase 2 and 3 trials designed to determine whether alpha-lactalbumin vaccination is effective in preventing TNBC in women at high risk of developing this form of breast cancer.
We estimate that completing our preclinical studies, obtaining permission from the US Food and Drug Administration to test our investigational new drug, and completing both phase 1 clinical trials will require about 5 years. Thereafter, completion of phase 2 and 3 trials designed to prevent both recurrence of TNBC in women already diagnosed with this disease and occurrence of TNBC in otherwise healthy, cancer-free women will likely take at least another 5 years, so that this vaccine will likely not be available to the general public before 2024.
TO SUM UP
Although our immune system is potentially capable of protecting us from some cancers, we currently have no immune protection against cancers we commonly confront as we age. We propose that tissue-specific self proteins that are retired from expression with age in normal tissues but are expressed at immunogenic levels in emerging tumors may substitute for unavailable pathogens as targets for immunoprevention of adult-onset cancers that commonly occur with age. We know that the retired breast-specific protein, alpha-lactalbumin, is overexpressed in TNBC and that vaccination with alpha-lactalbumin provides safe and effective protection from breast cancer in preclinical mouse studies. Clinical trials are planned to ultimately determine whether alpha-lactalbumin vaccination can prevent recurrence of TNBC in women already diagnosed with this disease and prevent the initiation of TNBC in women at high risk of developing this most aggressive and lethal form of breast cancer.
Acknowledgment: This work was supported by a grant from Shield Biotech, Inc., Cleveland, OH. In addition, the author wishes to recognize and express his sincere gratitude for the support and encouragement received from numerous organizations that have been instrumental in making this work possible, including November Philanthropy, Brakes for Breasts, the Breast Health and Healing Foundation, the Toni Turchi Foundation, the Coalition of Women Who Care About Breast Cancer, the Sisters for Prevention, the Previvors and Survivors, the Champions of the Pink Vaccine, the Race at Legacy Village, the National Greek Orthodox Ladies Philopto-chos Society, the Daughters of Penelope Icarus Chapter 321, Can’t Stop Won’t Stop, the Babylon Breast Cancer Coalition, and Walk With A Doc.
The most proven, effective way to control disease is through prophylactic vaccination. The childhood vaccination program is a testament to this disease prevention approach, and in its current form protects us from diseases caused by 16 different pathogens.1
Childhood immunization ends in the teen years with recommended vaccination against multiple strains of human papillomavirus that are associated with several cancers, most notably cervical carcinoma.2 However, even though we have known for over 30 years that the immune system can provide considerable vaccine-induced protection against the development of cancer,3 we have not produced any vaccines that prevent cancers that commonly occur with age, such as breast and prostate cancer, which afflict 1 of 8 women and 1 of 6 men, respectively.4,5
The lack of an adult vaccine program that provides protection against such commonly occurring adult-onset cancers represents a glaring health care deficiency and a challenge for this current generation to protect coming generations.
THE ‘RETIRED’ PROTEIN HYPOTHESIS
Given that most cancers are not associated with any disease-inducing pathogens, at what targets can we aim our immune system to induce safe and effective protection against these commonly occurring adult-onset cancers?
Perhaps an understanding of the natural aging process may provide us with insights regarding possible vaccine targets. As we age, there is a decline in expression of many tissue-specific proteins, often to the point where they may be considered “retired” and no longer found at detectable or immunogenic levels in normal cells. Examples of this natural aging process include the pigment proteins as our hair whitens, certain lactation proteins when breastfeeding ceases, and some ovarian proteins as menopause begins and production of mature egg follicles ceases. If these retired proteins are expressed in invigorated emerging tumors, then preemptive immunity directed against these retired proteins would attack and destroy the emerging tumors and ignore normal tissues, thereby avoiding any complicating collateral autoimmune damage.
Thus, we propose that retired tissue-specific self-proteins may substitute for unavailable pathogens as targets for mediating safe and effective immune protection against adult-onset cancers such as breast cancer.
SAFE AND EFFECTIVE PREVENTION OF BREAST CANCER IN MICE
To test this retired-protein hypothesis for immunoprevention of breast cancer, we selected alpha-lactalbumin as our vaccine target, for two reasons:
- Alpha-lactalbumin is a protein expressed exclusively in lactating breast tissue and is not expressed at immunogenic levels in either normal nonlactating breast tissues or in any of 78 other normal human tissues examined.6–8
- Alpha-lactalbumin is expressed in most human triple-negative breast cancers (TNBC),9,10 the most aggressive and lethal form of breast cancer, and the predominant form that occurs in women with mutations in the breast cancer 1, early-onset gene (BRCA1).11,12
We found that alpha-lactalbumin vaccination consistently inhibited the formation and growth of breast tumors in three different mouse models commonly used in breast cancer research.13 More importantly, the observed immune protection against the development of breast cancer in mice occurred in the absence of any detectable autoimmune inflammatory damage in any normal tissues examined. Thus, we concluded that alpha-lactalbumin vaccination could provide healthy women with safe and effective immune protection against the more malignant forms of breast cancer.
FROM BENCH TO BEDSIDE
How then do we determine whether alpha-lactalbumin vaccination prevents the development of TNBC in otherwise healthy cancer-free women, and whether it prevents recurrence of TNBC in women already diagnosed with TNBC? Our initial approach will involve two phase 1 clinical trials designed to determine the safety of the vaccine as well as the dose and number of vaccinations needed to induce optimum tumor immunity.
The first (phase 1a) trial will involve vaccination of women recently diagnosed with TNBC who have recovered with the current standard of care. These women will be vaccinated in groups receiving various doses of both recombinant human alpha-lactalbumin and an appropriate immune adjuvant that activates the immune system so it responds aggressively to the alpha-lactalbumin and creates the proinflammatory T-cell response needed for effective tumor immunity. This trial will simply provide dosage and safety profiles of the vaccine and will thereby lay the groundwork for subsequent (phase 2 and 3) trials designed to determine whether alpha-lactalbumin vaccination is effective in preventing recurrence of TNBC in women already diagnosed with this disease.
The dosage and number of immunizations shown to provide optimum immunity in the phase 1a trial will be used in a second (phase 1b) trial designed primarily to determine the safety of alpha-lactalbumin vaccination in healthy cancer-free women who have elected to undergo voluntary prophylactic mastectomy to reduce their breast cancer risk. Most of the women who elect to have this surgery have an established family history of breast cancer or a known BRCA1 mutation associated with high breast cancer risk, or both.11,12 Consenting women will be vaccinated against alpha-lactalbumin several months before their mastectomy, and their surgically removed breast tissues will be examined extensively for signs of vaccine-induced autoimmune damage. Thus, this trial will determine the safety of alpha-lactalbumin vaccination in healthy cancer-free women and will lay the groundwork for subsequent phase 2 and 3 trials designed to determine whether alpha-lactalbumin vaccination is effective in preventing TNBC in women at high risk of developing this form of breast cancer.
We estimate that completing our preclinical studies, obtaining permission from the US Food and Drug Administration to test our investigational new drug, and completing both phase 1 clinical trials will require about 5 years. Thereafter, completion of phase 2 and 3 trials designed to prevent both recurrence of TNBC in women already diagnosed with this disease and occurrence of TNBC in otherwise healthy, cancer-free women will likely take at least another 5 years, so that this vaccine will likely not be available to the general public before 2024.
TO SUM UP
Although our immune system is potentially capable of protecting us from some cancers, we currently have no immune protection against cancers we commonly confront as we age. We propose that tissue-specific self proteins that are retired from expression with age in normal tissues but are expressed at immunogenic levels in emerging tumors may substitute for unavailable pathogens as targets for immunoprevention of adult-onset cancers that commonly occur with age. We know that the retired breast-specific protein, alpha-lactalbumin, is overexpressed in TNBC and that vaccination with alpha-lactalbumin provides safe and effective protection from breast cancer in preclinical mouse studies. Clinical trials are planned to ultimately determine whether alpha-lactalbumin vaccination can prevent recurrence of TNBC in women already diagnosed with this disease and prevent the initiation of TNBC in women at high risk of developing this most aggressive and lethal form of breast cancer.
Acknowledgment: This work was supported by a grant from Shield Biotech, Inc., Cleveland, OH. In addition, the author wishes to recognize and express his sincere gratitude for the support and encouragement received from numerous organizations that have been instrumental in making this work possible, including November Philanthropy, Brakes for Breasts, the Breast Health and Healing Foundation, the Toni Turchi Foundation, the Coalition of Women Who Care About Breast Cancer, the Sisters for Prevention, the Previvors and Survivors, the Champions of the Pink Vaccine, the Race at Legacy Village, the National Greek Orthodox Ladies Philopto-chos Society, the Daughters of Penelope Icarus Chapter 321, Can’t Stop Won’t Stop, the Babylon Breast Cancer Coalition, and Walk With A Doc.
- Centers for Disease Control and Prevention. Immunization schedules. www.cdc.gov/vaccines/schedules/. Accessed September 4, 2014.
- Schiller JT, Lowy DR. Understanding and learning from the success of prophylactic human papillomavirus vaccines. Nat Rev Microbiol 2012; 10:681–692.
- Van Pel A, Boon T. Protection against a nonimmunogenic mouse leukemia by an immunogenic variant obtained by mutagenesis. Proc Natl Acad Sci USA 1982; 79:4718–4722.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013; 63:11–30.
- National Cancer Institute. Surveillance, Epidemiology, and End Results (SEER) Program. Previous version: SEER cancer statistics review 1975–2010. http://seer.cancer.gov/csr/1975_2010/. Accessed September 4, 2014.
- Uhlen M, Oksvold P, Fagerberg L, et al. Towards a knowledge-based human protein atlas. Nat Biotechnol 2010; 28:1248–1250.
- Pontén F, Gry M, Fagerberg L, et al. A global view of protein expression in human cells, tissues, and organs. Mol Syst Biol 2009; 5:337.
- The Human Protein Atlas. www.proteinatlas.org. Accessed September 4, 2014.
- Rhodes DR, Yu J, Shanker K, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 2004; 6:1–6.
- ONCOMINEdatabase. www.oncomine.org/resource/login.html. Accessed September 4, 2014.
- Atchley DP, Albarracin CT, Lopez A, et al. Clinical and pathologic characteristics of patients with BRCA-positive and BRCA-negative breast cancer. J Clin Oncol 2008; 26:4282–4288.
- Comen E, Davids M, Kirchhoff T, Hudis C, Offit K, Robson M. Relative contributions of BRCA1 and BRCA2 mutations to “triple-negative” breast cancer in Ashkenazi women. Breast Cancer Res Treat 2011; 129:185–190.
- Jaini R, Kesaraju P, Johnson JM, Altuntas CZ, Jane-Wit D, Tuohy VK. An autoimmune-mediated strategy for prophylactic breast cancer vaccination. Nat Med 2010; 16:799–803.
- Centers for Disease Control and Prevention. Immunization schedules. www.cdc.gov/vaccines/schedules/. Accessed September 4, 2014.
- Schiller JT, Lowy DR. Understanding and learning from the success of prophylactic human papillomavirus vaccines. Nat Rev Microbiol 2012; 10:681–692.
- Van Pel A, Boon T. Protection against a nonimmunogenic mouse leukemia by an immunogenic variant obtained by mutagenesis. Proc Natl Acad Sci USA 1982; 79:4718–4722.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013; 63:11–30.
- National Cancer Institute. Surveillance, Epidemiology, and End Results (SEER) Program. Previous version: SEER cancer statistics review 1975–2010. http://seer.cancer.gov/csr/1975_2010/. Accessed September 4, 2014.
- Uhlen M, Oksvold P, Fagerberg L, et al. Towards a knowledge-based human protein atlas. Nat Biotechnol 2010; 28:1248–1250.
- Pontén F, Gry M, Fagerberg L, et al. A global view of protein expression in human cells, tissues, and organs. Mol Syst Biol 2009; 5:337.
- The Human Protein Atlas. www.proteinatlas.org. Accessed September 4, 2014.
- Rhodes DR, Yu J, Shanker K, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 2004; 6:1–6.
- ONCOMINEdatabase. www.oncomine.org/resource/login.html. Accessed September 4, 2014.
- Atchley DP, Albarracin CT, Lopez A, et al. Clinical and pathologic characteristics of patients with BRCA-positive and BRCA-negative breast cancer. J Clin Oncol 2008; 26:4282–4288.
- Comen E, Davids M, Kirchhoff T, Hudis C, Offit K, Robson M. Relative contributions of BRCA1 and BRCA2 mutations to “triple-negative” breast cancer in Ashkenazi women. Breast Cancer Res Treat 2011; 129:185–190.
- Jaini R, Kesaraju P, Johnson JM, Altuntas CZ, Jane-Wit D, Tuohy VK. An autoimmune-mediated strategy for prophylactic breast cancer vaccination. Nat Med 2010; 16:799–803.
KEY POINTS
- “Retired” tissue-specific self proteins may substitute for unavailable pathogens as vaccine targets for mediating immune prevention of adult-onset cancers.
- Vaccination against the retired breast-specific protein alpha-lactalbumin provides safe and effective immune protection against the development of breast tumors in several mouse models.
- Alpha-lactalbumin is overexpressed in most human triple-negative breast cancers (TNBC), the most aggressive and lethal form of human breast cancer.
- Forthcoming are clinical trials designed to prevent the initiation of TNBC in otherwise healthy cancer-free women, as well as trials designed to prevent recurrence of TNBC in women already diagnosed with this disease.
Postop Patient Reports “Wound Infection”
ANSWER
The correct answer is an allergic reaction to a contactant, most likely the triple-antibiotic ointment (choice “d”).
Irritant reactions to tape adhesive (choice “a”) are extremely common. However, the resultant rash would have been confined to the linear areas where the tape touched his skin.
Dissolving sutures, such as those used in this case, can provoke a “suture granuloma”—essentially a foreign body reaction to the suture material (choice “b”). But this would have caused a focal area of swelling and redness, and very possibly a show of pus.
Postop wound infections (choice “c”) are also quite common. However, they would not manifest solely with itching in a papulovesicular rash surrounding the wound. Had infection developed, the redness would have been broad-based, with ill-defined margins, and the patient’s complaint would have been of pain, not itching. No vesicles would have been seen with bacterial infection.
DISCUSSION
This case illustrates the phenomenon of “treatment as problem,” in which the medication the patient applies becomes more problematic than the condition being addressed. Reactions to the neomycin in triple-antibiotic ointment are common but still provoke considerable worry on the part of patients and providers alike, especially when mistaken for “infection.”
This patient, like many, was dubious of the diagnosis, pointing out that he had used this same topical medication on many occasions without incident (though not recently). What he didn’t know is that it takes repeated exposure to a given allergen to develop T-memory cells that eventually begin to react. This same phenomenon is seen with poison ivy; patients will recall the ability, as a child, to practically wallow in poison ivy with impunity, making them doubtful about being allergic to it as an adult.
Neomycin, an aminoglycoside with a fairly wide spectrum of antibacterial activity, was first noted as a contact allergen in 1952. It is such a notorious offender that it was named Allergen of the Year in 2010 by the American Contact Dermatology Society.
For the past 20 years, 7% to 13% of patch tests surveyed were positive for neomycin. For reasons not entirely clear, Americans older than 60 are 150% more likely to experience a reaction to neomycin than are younger patients. (It could simply be that they’ve had more chances for exposure.)
In another interesting twist, the ointment vehicle appears to play a role. A reaction to this preparation is considerably more likely than to the same drug in other forms (eg, powders, solutions, creams). This is true of most medications, such as topical steroids, which are effectively self-occluded by this vehicle.
Persons with impaired barrier function, such as those with atopic dermatitis or whose skin has been prepped for surgery, appear to be at increased risk for these types of contact dermatoses.
Though there are other items in the differential, the configuration of the papulovesicular rash and the sole symptom of itching are essentially pathognomic for contact dermatitis. Besides the use of potent topical steroids for a few days, the real “cure” for this problem is for the patient to switch to “double-antibiotic” creams or ointments that do not include neomycin.
ANSWER
The correct answer is an allergic reaction to a contactant, most likely the triple-antibiotic ointment (choice “d”).
Irritant reactions to tape adhesive (choice “a”) are extremely common. However, the resultant rash would have been confined to the linear areas where the tape touched his skin.
Dissolving sutures, such as those used in this case, can provoke a “suture granuloma”—essentially a foreign body reaction to the suture material (choice “b”). But this would have caused a focal area of swelling and redness, and very possibly a show of pus.
Postop wound infections (choice “c”) are also quite common. However, they would not manifest solely with itching in a papulovesicular rash surrounding the wound. Had infection developed, the redness would have been broad-based, with ill-defined margins, and the patient’s complaint would have been of pain, not itching. No vesicles would have been seen with bacterial infection.
DISCUSSION
This case illustrates the phenomenon of “treatment as problem,” in which the medication the patient applies becomes more problematic than the condition being addressed. Reactions to the neomycin in triple-antibiotic ointment are common but still provoke considerable worry on the part of patients and providers alike, especially when mistaken for “infection.”
This patient, like many, was dubious of the diagnosis, pointing out that he had used this same topical medication on many occasions without incident (though not recently). What he didn’t know is that it takes repeated exposure to a given allergen to develop T-memory cells that eventually begin to react. This same phenomenon is seen with poison ivy; patients will recall the ability, as a child, to practically wallow in poison ivy with impunity, making them doubtful about being allergic to it as an adult.
Neomycin, an aminoglycoside with a fairly wide spectrum of antibacterial activity, was first noted as a contact allergen in 1952. It is such a notorious offender that it was named Allergen of the Year in 2010 by the American Contact Dermatology Society.
For the past 20 years, 7% to 13% of patch tests surveyed were positive for neomycin. For reasons not entirely clear, Americans older than 60 are 150% more likely to experience a reaction to neomycin than are younger patients. (It could simply be that they’ve had more chances for exposure.)
In another interesting twist, the ointment vehicle appears to play a role. A reaction to this preparation is considerably more likely than to the same drug in other forms (eg, powders, solutions, creams). This is true of most medications, such as topical steroids, which are effectively self-occluded by this vehicle.
Persons with impaired barrier function, such as those with atopic dermatitis or whose skin has been prepped for surgery, appear to be at increased risk for these types of contact dermatoses.
Though there are other items in the differential, the configuration of the papulovesicular rash and the sole symptom of itching are essentially pathognomic for contact dermatitis. Besides the use of potent topical steroids for a few days, the real “cure” for this problem is for the patient to switch to “double-antibiotic” creams or ointments that do not include neomycin.
ANSWER
The correct answer is an allergic reaction to a contactant, most likely the triple-antibiotic ointment (choice “d”).
Irritant reactions to tape adhesive (choice “a”) are extremely common. However, the resultant rash would have been confined to the linear areas where the tape touched his skin.
Dissolving sutures, such as those used in this case, can provoke a “suture granuloma”—essentially a foreign body reaction to the suture material (choice “b”). But this would have caused a focal area of swelling and redness, and very possibly a show of pus.
Postop wound infections (choice “c”) are also quite common. However, they would not manifest solely with itching in a papulovesicular rash surrounding the wound. Had infection developed, the redness would have been broad-based, with ill-defined margins, and the patient’s complaint would have been of pain, not itching. No vesicles would have been seen with bacterial infection.
DISCUSSION
This case illustrates the phenomenon of “treatment as problem,” in which the medication the patient applies becomes more problematic than the condition being addressed. Reactions to the neomycin in triple-antibiotic ointment are common but still provoke considerable worry on the part of patients and providers alike, especially when mistaken for “infection.”
This patient, like many, was dubious of the diagnosis, pointing out that he had used this same topical medication on many occasions without incident (though not recently). What he didn’t know is that it takes repeated exposure to a given allergen to develop T-memory cells that eventually begin to react. This same phenomenon is seen with poison ivy; patients will recall the ability, as a child, to practically wallow in poison ivy with impunity, making them doubtful about being allergic to it as an adult.
Neomycin, an aminoglycoside with a fairly wide spectrum of antibacterial activity, was first noted as a contact allergen in 1952. It is such a notorious offender that it was named Allergen of the Year in 2010 by the American Contact Dermatology Society.
For the past 20 years, 7% to 13% of patch tests surveyed were positive for neomycin. For reasons not entirely clear, Americans older than 60 are 150% more likely to experience a reaction to neomycin than are younger patients. (It could simply be that they’ve had more chances for exposure.)
In another interesting twist, the ointment vehicle appears to play a role. A reaction to this preparation is considerably more likely than to the same drug in other forms (eg, powders, solutions, creams). This is true of most medications, such as topical steroids, which are effectively self-occluded by this vehicle.
Persons with impaired barrier function, such as those with atopic dermatitis or whose skin has been prepped for surgery, appear to be at increased risk for these types of contact dermatoses.
Though there are other items in the differential, the configuration of the papulovesicular rash and the sole symptom of itching are essentially pathognomic for contact dermatitis. Besides the use of potent topical steroids for a few days, the real “cure” for this problem is for the patient to switch to “double-antibiotic” creams or ointments that do not include neomycin.
A week ago, a 56-year-old man had a skin cancer surgically removed. Last night, he presented to an urgent care clinic for evaluation of a “wound infection” and received a prescription for double-strength trimethoprim/sulfa tablets (to be taken bid for 10 days). He is now in the dermatology office for follow-up. According to the patient, the problem manifested two days postop. There was no associated pain, only itching. The patient feels fine, with no fever or malaise, and there is no history of immunosuppression. He reports following his postop instructions well, changing his bandage daily and using triple-antibiotic ointment to dress the wound directly. The immediate peri-incisional area is indicated as the source of the problem. Surrounding the incision, which is healing well otherwise, is a sharply defined, bright pink, papulovesicular rash on a slightly edematous base. There is no tenderness on palpation, and no purulent material can be expressed from the wound. The area is only slightly warmer than the surrounding skin.
ACIP Recommends PCV13 for All Adults 65 and Up
All adults who are 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) routinely in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23), according to a new recommendation from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The recommendation appears in the Sept. 19 issue of Morbidity and Mortality Weekly Report.
The ACIP recommendation calls for pneumococcal vaccine-naive adults aged 65 and older to receive one dose of PCV13 vaccine, followed by a dose of PPSV23 6-12 months later (MMWR 2014:63;822-5). Older adults who have previously received only PPSV23 should receive a dose of PCV13 at least 12 months later, wrote Sara Tomczyk of the CDC and her associates.
ACIP has recommended PPSV23 for older adults since 2010. In 2012, the committee made its first recommendation for PCV13, targeting patients 19 years and older who have immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leak, or cochlear implants. The new PCV13 recommendation for all older adults is based on a randomized, placebo-controlled trial of the vaccine in about 85,000 adults aged 65 years and older in the Netherlands who had no prior pneumococcal vaccine exposure. The vaccine showed a moderate level of evidence for efficacy against community-acquired pneumonia in this cohort, ACIP determined. Efficacy against nonbacteremic vaccine-type pneumococcal pneumonia was about 45%, while efficacy against vaccine-type invasive pneumococcal disease was about 75%, the reviewers wrote.
ACIP will reevaluate the recommendations in 2018.
Ms. Tomczyk and her colleagues disclosed no funding sources or conflicts of interest.
All adults who are 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) routinely in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23), according to a new recommendation from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The recommendation appears in the Sept. 19 issue of Morbidity and Mortality Weekly Report.
The ACIP recommendation calls for pneumococcal vaccine-naive adults aged 65 and older to receive one dose of PCV13 vaccine, followed by a dose of PPSV23 6-12 months later (MMWR 2014:63;822-5). Older adults who have previously received only PPSV23 should receive a dose of PCV13 at least 12 months later, wrote Sara Tomczyk of the CDC and her associates.
ACIP has recommended PPSV23 for older adults since 2010. In 2012, the committee made its first recommendation for PCV13, targeting patients 19 years and older who have immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leak, or cochlear implants. The new PCV13 recommendation for all older adults is based on a randomized, placebo-controlled trial of the vaccine in about 85,000 adults aged 65 years and older in the Netherlands who had no prior pneumococcal vaccine exposure. The vaccine showed a moderate level of evidence for efficacy against community-acquired pneumonia in this cohort, ACIP determined. Efficacy against nonbacteremic vaccine-type pneumococcal pneumonia was about 45%, while efficacy against vaccine-type invasive pneumococcal disease was about 75%, the reviewers wrote.
ACIP will reevaluate the recommendations in 2018.
Ms. Tomczyk and her colleagues disclosed no funding sources or conflicts of interest.
All adults who are 65 years or older should receive 13-valent pneumococcal conjugate vaccine (PCV13) routinely in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23), according to a new recommendation from the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices (ACIP). The recommendation appears in the Sept. 19 issue of Morbidity and Mortality Weekly Report.
The ACIP recommendation calls for pneumococcal vaccine-naive adults aged 65 and older to receive one dose of PCV13 vaccine, followed by a dose of PPSV23 6-12 months later (MMWR 2014:63;822-5). Older adults who have previously received only PPSV23 should receive a dose of PCV13 at least 12 months later, wrote Sara Tomczyk of the CDC and her associates.
ACIP has recommended PPSV23 for older adults since 2010. In 2012, the committee made its first recommendation for PCV13, targeting patients 19 years and older who have immunocompromising conditions, functional or anatomic asplenia, cerebrospinal fluid leak, or cochlear implants. The new PCV13 recommendation for all older adults is based on a randomized, placebo-controlled trial of the vaccine in about 85,000 adults aged 65 years and older in the Netherlands who had no prior pneumococcal vaccine exposure. The vaccine showed a moderate level of evidence for efficacy against community-acquired pneumonia in this cohort, ACIP determined. Efficacy against nonbacteremic vaccine-type pneumococcal pneumonia was about 45%, while efficacy against vaccine-type invasive pneumococcal disease was about 75%, the reviewers wrote.
ACIP will reevaluate the recommendations in 2018.
Ms. Tomczyk and her colleagues disclosed no funding sources or conflicts of interest.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT