User login
Diastolic heart failure and TOPCAT
The TOPCAT study, reported at the recent American Heart Association meeting in Dallas, examined the murky world of our understanding of heart failure occurring in patients with preserved left ventricular ejection fraction.
That seeming paradox has been the subject of physiologic and therapeutic controversy for some time. The realization that at least half of the patients admitted to the hospital with heart failure have normal or even supernormal left ventricular ejection fraction (HFpEF) has raised the therapeutic importance of this clinical entity. Of even more importance is the fact that patients with HFpEF exhibit morbidity and mortality similar to those heart failure patients with reduced left ventricular ejection fraction (HFrEF).
In an epidemiologic study in Olmsted County, Minnesota (N. Engl. J. Med. 2006;355:251-9), the 1-year mortality was 29% for HFpEF and 32% for HFrEF. Patients with HFpEF were more likely to be female (65.7% vs. 34.6%) and to have hypertension and atrial fibrillation than were those with HFrEF (62.7% vs. 48% and 41.3% vs. 28.5%, respectively).
Although we have significantly impacted mortality in patients who have HFrEF with the use of cardiac resynchronization therapy, implantable cardiac defibrillators, and medical therapy with beta-blockers and renin angiotensin inhibitors, we have failed to modify clinical outcomes in patients with HFpEF.
This has not been for a lack of trying. Several randomized clinical trials have been conducted with all of the drugs currently being used for HFrEF without any definitive results. An important problem in treating this population has been the heterogeneity of patients and multiple comorbidities, including chronic renal and pulmonary disease, acute hypertension, and atrial fibrillation that patients with HFpEF experience with the acute event. In addition, many of these patients are already receiving a multiplicity of concurrent therapies that have been approved for HFrEF for management.
Our understanding of the pathophysiology of HFpEF also remains cloudy. Both left and right ventricular hypertrophy with concomitant decrease in ventricular diastolic relaxation is the common observed echocardiographic abnormality. We have not as yet developed therapy for the treatment of diastolic dysfunction. Aldosterone antagonists, previously shown to be beneficial in patients with HFrEF, have emerged as likely candidates to improve HFpEF. Small clinical studies have shown improvement in diastolic function in elderly patients with hypertension and chronic renal disease. Consequently, the National Heart, Lung, and Blood Institute embarked on the TOPCAT study in 2006.
TOPCAT randomized 3,345 symptomatic heart failure patients who had a heart failure hospitalization in the previous year and with evidence of fluid retention, a left ventricular ejection fraction of more than 45%, controlled systolic blood pressure of less than 140 mm Hg, and elevated brain natriuretic peptide to treatment with placebo or spironolactone at 25 or 50 mg daily. A history of hypertension was present in 91%, coronary artery disease in 57%, atrial fibrillation in 35%, chronic renal disease in 35%, and diabetes in 32%. Patients included in the study had a mean LVEF of 56%; two thirds were in New York Heart Association class II and one third were in NYHA class III.
Over 80% of patients were receiving an ACE inhibitor or angiotensin receptor blocker, beta-blockers, and a diuretic. The mean dose of spironolactone was 25 mg. There was no significant difference in the primary outcome of cardiovascular death, nonfatal hospitalization or resuscitated cardiac arrest in the placebo and treated groups (20.4% and 18.6%, respectively). There was a significant decrease in heart failure hospitalization in the placebo compared to spironolactone patients (14.2% vs. 12.0%; P = .042). Both hyperkalemia greater than 5.5 mmol/L and an increase in serum creatinine were observed in the treated patients.
The striking observation in this trial, as it has been in previous trials, is the disparity between the epidemiologic mortality and the randomized controlled trial experience: 29% annual mortality in Olmsted County, compared with the 10.2% three-year mortality in TOPCAT. It appears that we are studying two separate diseases. And we are. After all the exclusion criteria included in the design of TOPCAT, we are unable to encapsulate the population at risk in this complex heart failure syndrome.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
The TOPCAT study, reported at the recent American Heart Association meeting in Dallas, examined the murky world of our understanding of heart failure occurring in patients with preserved left ventricular ejection fraction.
That seeming paradox has been the subject of physiologic and therapeutic controversy for some time. The realization that at least half of the patients admitted to the hospital with heart failure have normal or even supernormal left ventricular ejection fraction (HFpEF) has raised the therapeutic importance of this clinical entity. Of even more importance is the fact that patients with HFpEF exhibit morbidity and mortality similar to those heart failure patients with reduced left ventricular ejection fraction (HFrEF).
In an epidemiologic study in Olmsted County, Minnesota (N. Engl. J. Med. 2006;355:251-9), the 1-year mortality was 29% for HFpEF and 32% for HFrEF. Patients with HFpEF were more likely to be female (65.7% vs. 34.6%) and to have hypertension and atrial fibrillation than were those with HFrEF (62.7% vs. 48% and 41.3% vs. 28.5%, respectively).
Although we have significantly impacted mortality in patients who have HFrEF with the use of cardiac resynchronization therapy, implantable cardiac defibrillators, and medical therapy with beta-blockers and renin angiotensin inhibitors, we have failed to modify clinical outcomes in patients with HFpEF.
This has not been for a lack of trying. Several randomized clinical trials have been conducted with all of the drugs currently being used for HFrEF without any definitive results. An important problem in treating this population has been the heterogeneity of patients and multiple comorbidities, including chronic renal and pulmonary disease, acute hypertension, and atrial fibrillation that patients with HFpEF experience with the acute event. In addition, many of these patients are already receiving a multiplicity of concurrent therapies that have been approved for HFrEF for management.
Our understanding of the pathophysiology of HFpEF also remains cloudy. Both left and right ventricular hypertrophy with concomitant decrease in ventricular diastolic relaxation is the common observed echocardiographic abnormality. We have not as yet developed therapy for the treatment of diastolic dysfunction. Aldosterone antagonists, previously shown to be beneficial in patients with HFrEF, have emerged as likely candidates to improve HFpEF. Small clinical studies have shown improvement in diastolic function in elderly patients with hypertension and chronic renal disease. Consequently, the National Heart, Lung, and Blood Institute embarked on the TOPCAT study in 2006.
TOPCAT randomized 3,345 symptomatic heart failure patients who had a heart failure hospitalization in the previous year and with evidence of fluid retention, a left ventricular ejection fraction of more than 45%, controlled systolic blood pressure of less than 140 mm Hg, and elevated brain natriuretic peptide to treatment with placebo or spironolactone at 25 or 50 mg daily. A history of hypertension was present in 91%, coronary artery disease in 57%, atrial fibrillation in 35%, chronic renal disease in 35%, and diabetes in 32%. Patients included in the study had a mean LVEF of 56%; two thirds were in New York Heart Association class II and one third were in NYHA class III.
Over 80% of patients were receiving an ACE inhibitor or angiotensin receptor blocker, beta-blockers, and a diuretic. The mean dose of spironolactone was 25 mg. There was no significant difference in the primary outcome of cardiovascular death, nonfatal hospitalization or resuscitated cardiac arrest in the placebo and treated groups (20.4% and 18.6%, respectively). There was a significant decrease in heart failure hospitalization in the placebo compared to spironolactone patients (14.2% vs. 12.0%; P = .042). Both hyperkalemia greater than 5.5 mmol/L and an increase in serum creatinine were observed in the treated patients.
The striking observation in this trial, as it has been in previous trials, is the disparity between the epidemiologic mortality and the randomized controlled trial experience: 29% annual mortality in Olmsted County, compared with the 10.2% three-year mortality in TOPCAT. It appears that we are studying two separate diseases. And we are. After all the exclusion criteria included in the design of TOPCAT, we are unable to encapsulate the population at risk in this complex heart failure syndrome.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
The TOPCAT study, reported at the recent American Heart Association meeting in Dallas, examined the murky world of our understanding of heart failure occurring in patients with preserved left ventricular ejection fraction.
That seeming paradox has been the subject of physiologic and therapeutic controversy for some time. The realization that at least half of the patients admitted to the hospital with heart failure have normal or even supernormal left ventricular ejection fraction (HFpEF) has raised the therapeutic importance of this clinical entity. Of even more importance is the fact that patients with HFpEF exhibit morbidity and mortality similar to those heart failure patients with reduced left ventricular ejection fraction (HFrEF).
In an epidemiologic study in Olmsted County, Minnesota (N. Engl. J. Med. 2006;355:251-9), the 1-year mortality was 29% for HFpEF and 32% for HFrEF. Patients with HFpEF were more likely to be female (65.7% vs. 34.6%) and to have hypertension and atrial fibrillation than were those with HFrEF (62.7% vs. 48% and 41.3% vs. 28.5%, respectively).
Although we have significantly impacted mortality in patients who have HFrEF with the use of cardiac resynchronization therapy, implantable cardiac defibrillators, and medical therapy with beta-blockers and renin angiotensin inhibitors, we have failed to modify clinical outcomes in patients with HFpEF.
This has not been for a lack of trying. Several randomized clinical trials have been conducted with all of the drugs currently being used for HFrEF without any definitive results. An important problem in treating this population has been the heterogeneity of patients and multiple comorbidities, including chronic renal and pulmonary disease, acute hypertension, and atrial fibrillation that patients with HFpEF experience with the acute event. In addition, many of these patients are already receiving a multiplicity of concurrent therapies that have been approved for HFrEF for management.
Our understanding of the pathophysiology of HFpEF also remains cloudy. Both left and right ventricular hypertrophy with concomitant decrease in ventricular diastolic relaxation is the common observed echocardiographic abnormality. We have not as yet developed therapy for the treatment of diastolic dysfunction. Aldosterone antagonists, previously shown to be beneficial in patients with HFrEF, have emerged as likely candidates to improve HFpEF. Small clinical studies have shown improvement in diastolic function in elderly patients with hypertension and chronic renal disease. Consequently, the National Heart, Lung, and Blood Institute embarked on the TOPCAT study in 2006.
TOPCAT randomized 3,345 symptomatic heart failure patients who had a heart failure hospitalization in the previous year and with evidence of fluid retention, a left ventricular ejection fraction of more than 45%, controlled systolic blood pressure of less than 140 mm Hg, and elevated brain natriuretic peptide to treatment with placebo or spironolactone at 25 or 50 mg daily. A history of hypertension was present in 91%, coronary artery disease in 57%, atrial fibrillation in 35%, chronic renal disease in 35%, and diabetes in 32%. Patients included in the study had a mean LVEF of 56%; two thirds were in New York Heart Association class II and one third were in NYHA class III.
Over 80% of patients were receiving an ACE inhibitor or angiotensin receptor blocker, beta-blockers, and a diuretic. The mean dose of spironolactone was 25 mg. There was no significant difference in the primary outcome of cardiovascular death, nonfatal hospitalization or resuscitated cardiac arrest in the placebo and treated groups (20.4% and 18.6%, respectively). There was a significant decrease in heart failure hospitalization in the placebo compared to spironolactone patients (14.2% vs. 12.0%; P = .042). Both hyperkalemia greater than 5.5 mmol/L and an increase in serum creatinine were observed in the treated patients.
The striking observation in this trial, as it has been in previous trials, is the disparity between the epidemiologic mortality and the randomized controlled trial experience: 29% annual mortality in Olmsted County, compared with the 10.2% three-year mortality in TOPCAT. It appears that we are studying two separate diseases. And we are. After all the exclusion criteria included in the design of TOPCAT, we are unable to encapsulate the population at risk in this complex heart failure syndrome.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies.
TOPCAT: Spironolactone cuts hospitalizations for diastolic heart failure
DALLAS – Spironolactone did not hit a home run in the large international "treatment of preserved cardiac function heart failure with an aldosterone antagonist" (TOPCAT) trial, but it did knock out a solid single in the form of significantly reduced hospitalizations for this extremely common, chronic, high morbidity/mortality condition.
It was this positive result for an important prespecified secondary outcome that enabled TOPCAT to avoid becoming roadkill. Technically, TOPCAT was a negative clinical trial in that spironolactone did not significantly outperform placebo on the primary composite outcome of cardiovascular mortality, heart failure hospitalization, or aborted cardiac arrest.
Yet that negative primary outcome was controversial: The aldosterone antagonist actually showed a significant positive result for the composite endpoint in North and South American participants, yet the results were resoundingly negative – and also considerably out of whack with the characteristic arc of progressive heart failure – among the nearly one-half of TOPCAT participants in Russia and the Republic of Georgia.
"What happened in Russia and Georgia we just don’t understand," Dr. Bertram Pitt, TOPCAT steering committee chair, said in an interview, shaking his head. "The event rate with placebo in Eastern Europe was so low it’s not compatible with anything we know about heart failure. The signs and symptoms of HFpEF [heart failure with preserved ejection fraction] are nonspecific; they can be due to obesity, lung disease, and other things. Clearly there are some people getting into the major trials of HFpEF that probably don’t have it."
TOPCAT was a randomized, double-blind clinical trial comprising 3,445 participants with symptomatic HFpEF at 250 sites in the United States and five other countries. They were randomized to spironolactone or placebo and followed prospectively for a mean of 3.3 years. The starting dose of the aldosterone antagonist was 15 mg/day, with a target of 30 mg/day. The drug could be titrated within the range of 15-45 mg/day. Eight months into the trial, the mean daily dose was 25 mg.
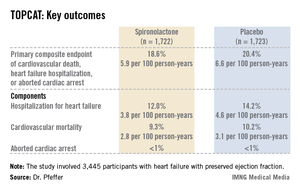
Presenting the TOPCAT results at the American Heart Association scientific sessions, Dr. Marc A. Pfeffer noted that the primary composite endpoint occurred in 20.4% of placebo-treated controls and 18.6% on spironolactone, a statistically nonsignificant difference. In contrast, the 17% reduction in the rate of hospitalization for HFpEF in the spironolactone group relative to controls was significant (P = .04). Moreover, the spironolactone-treated patients had a collective 394 HFpEF hospitalizations, markedly fewer than the 475 in controls. This translated to a hospitalization for heart failure with a preserved left ventricular ejection fraction occurring at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, compared with the 4.6 per 100 person-years in placebo-treated controls.
Hyperkalemia in excess of 5.5 mmol/L occurred in 18.7% of the spironolactone group, twice the rate of controls (9.1%). And the incidence of a creatinine level more than double the upper limit of normal was 49% greater in the spironolactone group. That said, neither of these laboratory abnormalities resulted in any serious adverse consequences because investigators adjusted the dose in response, explained Dr. Pfeffer, professor of medicine at Harvard University, Boston.
He drew special attention to two points: The primary composite event rate in placebo-treated patients in the Americas was 31.8% consistent with what has been seen in other studies of HFpEF – compared to a mere 8.4% in Eastern Europe. And patients who qualified for TOPCAT on the basis of an elevated natriuretic peptide level had a primary endpoint rate of 15.9% with spironolactone, a highly significant 35% reduction compared with the 23.6% in controls, suggesting that an elevated baseline natriuretic peptide level may be a biomarker useful in identifying those HFpEF patients most likely to respond to an aldosterone antagonist.
Dr. Pfeffer said that because the pharmaceutical industry has zero interest in spironolactone and the National Heart, Lung, and Blood Institute (NHLBI), which sponsored TOPCAT, has finite resources, he doubts there will be any further large studies of the drug in HFpEF.
"One is going to have to make decisions based on this trial. I don’t see another trial behind us," the cardiologist said.
And while TOPCAT was flawed, he said it contains a compelling message for clinicians: "I think we have an important finding here. We’re very confident that with this generic medication, which costs pennies per day, we can reduce hospitalizations for heart failure, which are the major burden in patients with HFpEF."
Discussant Dr. Margaret M. Redfield was more cautious. While there was a sound rationale for studying spironolactone in HFpEF based upon its impressive benefits in systolic heart failure as shown in earlier landmark clinical trials, given that the drug didn’t result in a significant reduction in all-cause hospitalizations, she said she wants to see evidence of improved patient-centered outcomes, such as quality of life, before prescribing spironolactone for HFpEF.
"One has to worry that the problems with worsening renal function and hyperkalemia may be much more common in clinical practice than in the highly monitored environment of a clinical trial," added Dr. Redfield, professor of medicine at the Mayo Clinic, Rochester, Minn.
Other heart failure experts not involved in TOPCAT took a more positive view of the study.
Dr. Clyde W. Yancy, AHA spokesperson, said in an interview that HFpEF is probably even more common than systolic heart failure, and it is a disease for which physicians have had no proven-effective treatments.
"You can almost justifiably say, ‘Let’s just look at the Americas, where patients had event rates that are consistent with the disease we think we understand.’ And it looks like there’s a signal there. I think that there’s enough evidence in TOPCAT for the clinical pragmatist to be able to be comfortable that spironolactone is probably beneficial and can be given safely. If you are convinced you can give it safely in your own clinical scenario, I think you should use it," said Dr. Yancy, professor of medicine and chief of cardiology at Northwestern University, Chicago.
Dr. Marco Metra, a cardiologist at the University of Brescia (Italy), said he interprets the TOPCAT results as an indication for prescribing spironolactone, at least in HFpEF patients with high natriuretic peptide levels or multiple heart failure hospitalizations.
Dr. Pitt, emeritus professor of cardiovascular medicine at the University of Michigan, Ann Arbor, emphasized that in prescribing spironolactone, whether for HFpEF or systolic heart failure, it’s obligatory to measure potassium and creatinine levels at baseline, when changing the dose, and at each routine follow-up visit, titrating in response to the results. Failure to do so is akin to prescribing warfarin for a patient with atrial fibrillation and then never measuring the INR (international normalized ratio) he said.
TOPCAT was sponsored by the National, Heart, Lung, and Blood Institute. Dr. Pfeffer and Dr. Pitt serve as consultants to numerous pharmaceutical companies that had no involvement in the trial.
DALLAS – Spironolactone did not hit a home run in the large international "treatment of preserved cardiac function heart failure with an aldosterone antagonist" (TOPCAT) trial, but it did knock out a solid single in the form of significantly reduced hospitalizations for this extremely common, chronic, high morbidity/mortality condition.
It was this positive result for an important prespecified secondary outcome that enabled TOPCAT to avoid becoming roadkill. Technically, TOPCAT was a negative clinical trial in that spironolactone did not significantly outperform placebo on the primary composite outcome of cardiovascular mortality, heart failure hospitalization, or aborted cardiac arrest.

Yet that negative primary outcome was controversial: The aldosterone antagonist actually showed a significant positive result for the composite endpoint in North and South American participants, yet the results were resoundingly negative – and also considerably out of whack with the characteristic arc of progressive heart failure – among the nearly one-half of TOPCAT participants in Russia and the Republic of Georgia.
"What happened in Russia and Georgia we just don’t understand," Dr. Bertram Pitt, TOPCAT steering committee chair, said in an interview, shaking his head. "The event rate with placebo in Eastern Europe was so low it’s not compatible with anything we know about heart failure. The signs and symptoms of HFpEF [heart failure with preserved ejection fraction] are nonspecific; they can be due to obesity, lung disease, and other things. Clearly there are some people getting into the major trials of HFpEF that probably don’t have it."
TOPCAT was a randomized, double-blind clinical trial comprising 3,445 participants with symptomatic HFpEF at 250 sites in the United States and five other countries. They were randomized to spironolactone or placebo and followed prospectively for a mean of 3.3 years. The starting dose of the aldosterone antagonist was 15 mg/day, with a target of 30 mg/day. The drug could be titrated within the range of 15-45 mg/day. Eight months into the trial, the mean daily dose was 25 mg.
Presenting the TOPCAT results at the American Heart Association scientific sessions, Dr. Marc A. Pfeffer noted that the primary composite endpoint occurred in 20.4% of placebo-treated controls and 18.6% on spironolactone, a statistically nonsignificant difference. In contrast, the 17% reduction in the rate of hospitalization for HFpEF in the spironolactone group relative to controls was significant (P = .04). Moreover, the spironolactone-treated patients had a collective 394 HFpEF hospitalizations, markedly fewer than the 475 in controls. This translated to a hospitalization for heart failure with a preserved left ventricular ejection fraction occurring at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, compared with the 4.6 per 100 person-years in placebo-treated controls.
Hyperkalemia in excess of 5.5 mmol/L occurred in 18.7% of the spironolactone group, twice the rate of controls (9.1%). And the incidence of a creatinine level more than double the upper limit of normal was 49% greater in the spironolactone group. That said, neither of these laboratory abnormalities resulted in any serious adverse consequences because investigators adjusted the dose in response, explained Dr. Pfeffer, professor of medicine at Harvard University, Boston.
He drew special attention to two points: The primary composite event rate in placebo-treated patients in the Americas was 31.8% consistent with what has been seen in other studies of HFpEF – compared to a mere 8.4% in Eastern Europe. And patients who qualified for TOPCAT on the basis of an elevated natriuretic peptide level had a primary endpoint rate of 15.9% with spironolactone, a highly significant 35% reduction compared with the 23.6% in controls, suggesting that an elevated baseline natriuretic peptide level may be a biomarker useful in identifying those HFpEF patients most likely to respond to an aldosterone antagonist.
Dr. Pfeffer said that because the pharmaceutical industry has zero interest in spironolactone and the National Heart, Lung, and Blood Institute (NHLBI), which sponsored TOPCAT, has finite resources, he doubts there will be any further large studies of the drug in HFpEF.
"One is going to have to make decisions based on this trial. I don’t see another trial behind us," the cardiologist said.
And while TOPCAT was flawed, he said it contains a compelling message for clinicians: "I think we have an important finding here. We’re very confident that with this generic medication, which costs pennies per day, we can reduce hospitalizations for heart failure, which are the major burden in patients with HFpEF."
Discussant Dr. Margaret M. Redfield was more cautious. While there was a sound rationale for studying spironolactone in HFpEF based upon its impressive benefits in systolic heart failure as shown in earlier landmark clinical trials, given that the drug didn’t result in a significant reduction in all-cause hospitalizations, she said she wants to see evidence of improved patient-centered outcomes, such as quality of life, before prescribing spironolactone for HFpEF.
"One has to worry that the problems with worsening renal function and hyperkalemia may be much more common in clinical practice than in the highly monitored environment of a clinical trial," added Dr. Redfield, professor of medicine at the Mayo Clinic, Rochester, Minn.
Other heart failure experts not involved in TOPCAT took a more positive view of the study.
Dr. Clyde W. Yancy, AHA spokesperson, said in an interview that HFpEF is probably even more common than systolic heart failure, and it is a disease for which physicians have had no proven-effective treatments.
"You can almost justifiably say, ‘Let’s just look at the Americas, where patients had event rates that are consistent with the disease we think we understand.’ And it looks like there’s a signal there. I think that there’s enough evidence in TOPCAT for the clinical pragmatist to be able to be comfortable that spironolactone is probably beneficial and can be given safely. If you are convinced you can give it safely in your own clinical scenario, I think you should use it," said Dr. Yancy, professor of medicine and chief of cardiology at Northwestern University, Chicago.
Dr. Marco Metra, a cardiologist at the University of Brescia (Italy), said he interprets the TOPCAT results as an indication for prescribing spironolactone, at least in HFpEF patients with high natriuretic peptide levels or multiple heart failure hospitalizations.
Dr. Pitt, emeritus professor of cardiovascular medicine at the University of Michigan, Ann Arbor, emphasized that in prescribing spironolactone, whether for HFpEF or systolic heart failure, it’s obligatory to measure potassium and creatinine levels at baseline, when changing the dose, and at each routine follow-up visit, titrating in response to the results. Failure to do so is akin to prescribing warfarin for a patient with atrial fibrillation and then never measuring the INR (international normalized ratio) he said.
TOPCAT was sponsored by the National, Heart, Lung, and Blood Institute. Dr. Pfeffer and Dr. Pitt serve as consultants to numerous pharmaceutical companies that had no involvement in the trial.
DALLAS – Spironolactone did not hit a home run in the large international "treatment of preserved cardiac function heart failure with an aldosterone antagonist" (TOPCAT) trial, but it did knock out a solid single in the form of significantly reduced hospitalizations for this extremely common, chronic, high morbidity/mortality condition.
It was this positive result for an important prespecified secondary outcome that enabled TOPCAT to avoid becoming roadkill. Technically, TOPCAT was a negative clinical trial in that spironolactone did not significantly outperform placebo on the primary composite outcome of cardiovascular mortality, heart failure hospitalization, or aborted cardiac arrest.

Yet that negative primary outcome was controversial: The aldosterone antagonist actually showed a significant positive result for the composite endpoint in North and South American participants, yet the results were resoundingly negative – and also considerably out of whack with the characteristic arc of progressive heart failure – among the nearly one-half of TOPCAT participants in Russia and the Republic of Georgia.
"What happened in Russia and Georgia we just don’t understand," Dr. Bertram Pitt, TOPCAT steering committee chair, said in an interview, shaking his head. "The event rate with placebo in Eastern Europe was so low it’s not compatible with anything we know about heart failure. The signs and symptoms of HFpEF [heart failure with preserved ejection fraction] are nonspecific; they can be due to obesity, lung disease, and other things. Clearly there are some people getting into the major trials of HFpEF that probably don’t have it."
TOPCAT was a randomized, double-blind clinical trial comprising 3,445 participants with symptomatic HFpEF at 250 sites in the United States and five other countries. They were randomized to spironolactone or placebo and followed prospectively for a mean of 3.3 years. The starting dose of the aldosterone antagonist was 15 mg/day, with a target of 30 mg/day. The drug could be titrated within the range of 15-45 mg/day. Eight months into the trial, the mean daily dose was 25 mg.
Presenting the TOPCAT results at the American Heart Association scientific sessions, Dr. Marc A. Pfeffer noted that the primary composite endpoint occurred in 20.4% of placebo-treated controls and 18.6% on spironolactone, a statistically nonsignificant difference. In contrast, the 17% reduction in the rate of hospitalization for HFpEF in the spironolactone group relative to controls was significant (P = .04). Moreover, the spironolactone-treated patients had a collective 394 HFpEF hospitalizations, markedly fewer than the 475 in controls. This translated to a hospitalization for heart failure with a preserved left ventricular ejection fraction occurring at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, compared with the 4.6 per 100 person-years in placebo-treated controls.
Hyperkalemia in excess of 5.5 mmol/L occurred in 18.7% of the spironolactone group, twice the rate of controls (9.1%). And the incidence of a creatinine level more than double the upper limit of normal was 49% greater in the spironolactone group. That said, neither of these laboratory abnormalities resulted in any serious adverse consequences because investigators adjusted the dose in response, explained Dr. Pfeffer, professor of medicine at Harvard University, Boston.
He drew special attention to two points: The primary composite event rate in placebo-treated patients in the Americas was 31.8% consistent with what has been seen in other studies of HFpEF – compared to a mere 8.4% in Eastern Europe. And patients who qualified for TOPCAT on the basis of an elevated natriuretic peptide level had a primary endpoint rate of 15.9% with spironolactone, a highly significant 35% reduction compared with the 23.6% in controls, suggesting that an elevated baseline natriuretic peptide level may be a biomarker useful in identifying those HFpEF patients most likely to respond to an aldosterone antagonist.
Dr. Pfeffer said that because the pharmaceutical industry has zero interest in spironolactone and the National Heart, Lung, and Blood Institute (NHLBI), which sponsored TOPCAT, has finite resources, he doubts there will be any further large studies of the drug in HFpEF.
"One is going to have to make decisions based on this trial. I don’t see another trial behind us," the cardiologist said.
And while TOPCAT was flawed, he said it contains a compelling message for clinicians: "I think we have an important finding here. We’re very confident that with this generic medication, which costs pennies per day, we can reduce hospitalizations for heart failure, which are the major burden in patients with HFpEF."
Discussant Dr. Margaret M. Redfield was more cautious. While there was a sound rationale for studying spironolactone in HFpEF based upon its impressive benefits in systolic heart failure as shown in earlier landmark clinical trials, given that the drug didn’t result in a significant reduction in all-cause hospitalizations, she said she wants to see evidence of improved patient-centered outcomes, such as quality of life, before prescribing spironolactone for HFpEF.
"One has to worry that the problems with worsening renal function and hyperkalemia may be much more common in clinical practice than in the highly monitored environment of a clinical trial," added Dr. Redfield, professor of medicine at the Mayo Clinic, Rochester, Minn.
Other heart failure experts not involved in TOPCAT took a more positive view of the study.
Dr. Clyde W. Yancy, AHA spokesperson, said in an interview that HFpEF is probably even more common than systolic heart failure, and it is a disease for which physicians have had no proven-effective treatments.
"You can almost justifiably say, ‘Let’s just look at the Americas, where patients had event rates that are consistent with the disease we think we understand.’ And it looks like there’s a signal there. I think that there’s enough evidence in TOPCAT for the clinical pragmatist to be able to be comfortable that spironolactone is probably beneficial and can be given safely. If you are convinced you can give it safely in your own clinical scenario, I think you should use it," said Dr. Yancy, professor of medicine and chief of cardiology at Northwestern University, Chicago.
Dr. Marco Metra, a cardiologist at the University of Brescia (Italy), said he interprets the TOPCAT results as an indication for prescribing spironolactone, at least in HFpEF patients with high natriuretic peptide levels or multiple heart failure hospitalizations.
Dr. Pitt, emeritus professor of cardiovascular medicine at the University of Michigan, Ann Arbor, emphasized that in prescribing spironolactone, whether for HFpEF or systolic heart failure, it’s obligatory to measure potassium and creatinine levels at baseline, when changing the dose, and at each routine follow-up visit, titrating in response to the results. Failure to do so is akin to prescribing warfarin for a patient with atrial fibrillation and then never measuring the INR (international normalized ratio) he said.
TOPCAT was sponsored by the National, Heart, Lung, and Blood Institute. Dr. Pfeffer and Dr. Pitt serve as consultants to numerous pharmaceutical companies that had no involvement in the trial.
AT THE AHA SCIENTIFIC SESSIONS
Major finding: Hospitalization for heart failure with a preserved left ventricular ejection fraction occurred at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, a significant 17% risk reduction compared with the 4.6 per 100 person-years in placebo-treated controls.
Data source: TOPCAT, a randomized, double-blind, six-country clinical trial involving 3,445 patients with symptomatic heart failure and an ejection fraction of 45% or more.
Disclosures:. TOPCAT was sponsored by the National, Heart, Lung, and Blood Institute. Dr. Pfeffer and Dr. Pitt serve as consultants to pharmaceutical companies that had no involvement in the trial.
Improved outcomes with CoreValve in extreme-risk patients
SAN FRANCISCO – Transcatheter aortic valve replacement with the self-expanding CoreValve in patients at extreme surgical risk significantly reduced the rate of death or major stroke at 1 year, from 43% to 26%, in a 487-patient pivotal trial.
The CoreValve Extreme Risk study gathered data from a registry of patients with symptomatic severe aortic stenosis who attempted an iliofemoral implantation procedure with the CoreValve at 40 U.S. sites. Results were compared with an "objective performance goal" derived from two sources: a meta-analysis of five contemporary balloon valvuloplasty series that found a 43% mortality and major stroke rate at 1 year, and the 1-year rate from the PARTNER B trial in inoperable patients (Placement of Aortic Transcatheter Valves, Cohort B), which was 50% but had a lower confidence bound of 43%.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The performance-goal comparison was necessary in the current study because randomizing these patients to medical therapy in a control group is no longer an acceptable option in the United States, Dr. Jeffrey J. Popma said at the Transcatheter Cardiovascular Therapeutics annual meeting.
The 1-year all-cause mortality rate was 24%, and the cardiovascular mortality was 18%, reported Dr. Popma, professor of medicine at Harvard Medical School, Boston.
Two percent of patients developed a major stroke within 1 month and 4% did so within 1 year.
Data from a continued access study involving another 830 extreme-risk patients who received the CoreValve through an iliofemoral approach are showing even better results, with a 16% rate of mortality or major stroke at 6 months, he added.
Among secondary endpoints at 1 year in the main study, 7% of patients developed any kind of stroke, 2% had an MI, 2% need reintervention, 41% had bleeding that met Valve Academic Research Consortium criteria, 8% had major vascular complications, and 27% required implantation of a permanent pacemaker, Dr. Popma reported at the meeting, cosponsored by the American College of Cardiology. Ninety percent of patients improved by at least one New York Heart Association functional class and 60% improved by at least two classes at 1 year of follow-up.
Paravalvular regurgitation of any severity was seen in 53% of patients 1 month after implantation and in 33% at 1 year. Moderate paravalvular leakage (PVL) affected 9% at 1 month and 4% at 1 year, and severe PVL affected 1.6% at 1 month and no patients at 1 year, Dr. Popma said. Among the 11% of patients with moderate PVL at 1 month, 80% of those who survived to 1 year had a reduction in leakage over time.
"We believe that’s why we did not find an association in the study between mild or moderate aortic regurgitation with respect to late-term mortality," though mortality risk was substantially higher with severe regurgitation, he said. One-year mortality rates were 86% with severe PVL and 24% with either moderate or mild PVL, compared with 18% in patients with no PVL.
The improvement in PVL rates over time may be due to use of CT angiography to select appropriate valve sizes for patients and continued expansion of the self-expanding frame over time. "That’s a remarkable finding, and it needs to be confirmed," Dr. Popma said.
The study focused on patients whose severe frailty, comorbidity, or disability put them at extreme risk of at least a 50% chance of death or irreversible morbidity within 30 days had they undergone surgical aortic valve replacement. A second U.S. pivotal trial of the CoreValve is focusing on patients at high (but not extreme) risk.
Most of the sites in the study had no experience with CoreValve before this study, Dr. Michael J. Mack noted at a press briefing. "The results are outstanding, but especially putting it in that light," said Dr. Mack, director of cardiovascular disease for the Baylor Scott & White Health System, Dallas, and a member of the steering committee for the PARTNER trial.
Dr. Popma reported financial associations with Medtronic, which sponsored the study and makes CoreValve, and with six other companies.
On Twitter @sherryboschert
This is a brilliantly executed, rigorously performed study. After doing sham-control studies, as we did, in these extreme-risk patients, it’s difficult to do a randomized trial. I think these investigators did they best that they could. Using a performance goal standard was the right thing to do.

|
|
I think that the results are good. It’s always difficult to compare study to study. They’re at different points in time, and we learn things over time. The CoreValve device is a lower-profile device, so there were fewer vascular complications, there was less major bleeding. Those things, in fact, can have an impact on 1-year mortality. The overall 1-year mortality was lower than in PARTNER. In PARTNER by intention to treat the mortality rate was 30.7%, and in the CoreValve study it was in the low 20% range. I think that that’s encouraging.
The stroke rates are encouraging. The pacemaker rates are predictable; it’s a by-product of the specific device. The importance of that is debatable, but it’s not really a concern. It was anticipated.
The paravalvular leakage (PVL) rate is interesting. Many studies with CoreValve suggest that either moderate or severe PVL is associated with an increase in late mortality, if not at 1 year later. It’s not a huge sample size; maybe over time with different definitions of PVL we’d begin to see that.
I am surprised by the 30-day to 1-year data that suggest a reduced PVL rate. I can understand how a self-expanding platform might change in the first 30 days; I can’t quite understand how it would change between 1 month and 12 months in a way that PVL would go down.
Martin B. Leon, M.D., is director of the Center for Interventional Vascular Therapy at Columbia University, New York, and served as principal investigator of the PARTNER trial of the competing Sapien valve.
This is a brilliantly executed, rigorously performed study. After doing sham-control studies, as we did, in these extreme-risk patients, it’s difficult to do a randomized trial. I think these investigators did they best that they could. Using a performance goal standard was the right thing to do.

|
|
I think that the results are good. It’s always difficult to compare study to study. They’re at different points in time, and we learn things over time. The CoreValve device is a lower-profile device, so there were fewer vascular complications, there was less major bleeding. Those things, in fact, can have an impact on 1-year mortality. The overall 1-year mortality was lower than in PARTNER. In PARTNER by intention to treat the mortality rate was 30.7%, and in the CoreValve study it was in the low 20% range. I think that that’s encouraging.
The stroke rates are encouraging. The pacemaker rates are predictable; it’s a by-product of the specific device. The importance of that is debatable, but it’s not really a concern. It was anticipated.
The paravalvular leakage (PVL) rate is interesting. Many studies with CoreValve suggest that either moderate or severe PVL is associated with an increase in late mortality, if not at 1 year later. It’s not a huge sample size; maybe over time with different definitions of PVL we’d begin to see that.
I am surprised by the 30-day to 1-year data that suggest a reduced PVL rate. I can understand how a self-expanding platform might change in the first 30 days; I can’t quite understand how it would change between 1 month and 12 months in a way that PVL would go down.
Martin B. Leon, M.D., is director of the Center for Interventional Vascular Therapy at Columbia University, New York, and served as principal investigator of the PARTNER trial of the competing Sapien valve.
This is a brilliantly executed, rigorously performed study. After doing sham-control studies, as we did, in these extreme-risk patients, it’s difficult to do a randomized trial. I think these investigators did they best that they could. Using a performance goal standard was the right thing to do.

|
|
I think that the results are good. It’s always difficult to compare study to study. They’re at different points in time, and we learn things over time. The CoreValve device is a lower-profile device, so there were fewer vascular complications, there was less major bleeding. Those things, in fact, can have an impact on 1-year mortality. The overall 1-year mortality was lower than in PARTNER. In PARTNER by intention to treat the mortality rate was 30.7%, and in the CoreValve study it was in the low 20% range. I think that that’s encouraging.
The stroke rates are encouraging. The pacemaker rates are predictable; it’s a by-product of the specific device. The importance of that is debatable, but it’s not really a concern. It was anticipated.
The paravalvular leakage (PVL) rate is interesting. Many studies with CoreValve suggest that either moderate or severe PVL is associated with an increase in late mortality, if not at 1 year later. It’s not a huge sample size; maybe over time with different definitions of PVL we’d begin to see that.
I am surprised by the 30-day to 1-year data that suggest a reduced PVL rate. I can understand how a self-expanding platform might change in the first 30 days; I can’t quite understand how it would change between 1 month and 12 months in a way that PVL would go down.
Martin B. Leon, M.D., is director of the Center for Interventional Vascular Therapy at Columbia University, New York, and served as principal investigator of the PARTNER trial of the competing Sapien valve.
SAN FRANCISCO – Transcatheter aortic valve replacement with the self-expanding CoreValve in patients at extreme surgical risk significantly reduced the rate of death or major stroke at 1 year, from 43% to 26%, in a 487-patient pivotal trial.
The CoreValve Extreme Risk study gathered data from a registry of patients with symptomatic severe aortic stenosis who attempted an iliofemoral implantation procedure with the CoreValve at 40 U.S. sites. Results were compared with an "objective performance goal" derived from two sources: a meta-analysis of five contemporary balloon valvuloplasty series that found a 43% mortality and major stroke rate at 1 year, and the 1-year rate from the PARTNER B trial in inoperable patients (Placement of Aortic Transcatheter Valves, Cohort B), which was 50% but had a lower confidence bound of 43%.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The performance-goal comparison was necessary in the current study because randomizing these patients to medical therapy in a control group is no longer an acceptable option in the United States, Dr. Jeffrey J. Popma said at the Transcatheter Cardiovascular Therapeutics annual meeting.
The 1-year all-cause mortality rate was 24%, and the cardiovascular mortality was 18%, reported Dr. Popma, professor of medicine at Harvard Medical School, Boston.
Two percent of patients developed a major stroke within 1 month and 4% did so within 1 year.
Data from a continued access study involving another 830 extreme-risk patients who received the CoreValve through an iliofemoral approach are showing even better results, with a 16% rate of mortality or major stroke at 6 months, he added.
Among secondary endpoints at 1 year in the main study, 7% of patients developed any kind of stroke, 2% had an MI, 2% need reintervention, 41% had bleeding that met Valve Academic Research Consortium criteria, 8% had major vascular complications, and 27% required implantation of a permanent pacemaker, Dr. Popma reported at the meeting, cosponsored by the American College of Cardiology. Ninety percent of patients improved by at least one New York Heart Association functional class and 60% improved by at least two classes at 1 year of follow-up.
Paravalvular regurgitation of any severity was seen in 53% of patients 1 month after implantation and in 33% at 1 year. Moderate paravalvular leakage (PVL) affected 9% at 1 month and 4% at 1 year, and severe PVL affected 1.6% at 1 month and no patients at 1 year, Dr. Popma said. Among the 11% of patients with moderate PVL at 1 month, 80% of those who survived to 1 year had a reduction in leakage over time.
"We believe that’s why we did not find an association in the study between mild or moderate aortic regurgitation with respect to late-term mortality," though mortality risk was substantially higher with severe regurgitation, he said. One-year mortality rates were 86% with severe PVL and 24% with either moderate or mild PVL, compared with 18% in patients with no PVL.
The improvement in PVL rates over time may be due to use of CT angiography to select appropriate valve sizes for patients and continued expansion of the self-expanding frame over time. "That’s a remarkable finding, and it needs to be confirmed," Dr. Popma said.
The study focused on patients whose severe frailty, comorbidity, or disability put them at extreme risk of at least a 50% chance of death or irreversible morbidity within 30 days had they undergone surgical aortic valve replacement. A second U.S. pivotal trial of the CoreValve is focusing on patients at high (but not extreme) risk.
Most of the sites in the study had no experience with CoreValve before this study, Dr. Michael J. Mack noted at a press briefing. "The results are outstanding, but especially putting it in that light," said Dr. Mack, director of cardiovascular disease for the Baylor Scott & White Health System, Dallas, and a member of the steering committee for the PARTNER trial.
Dr. Popma reported financial associations with Medtronic, which sponsored the study and makes CoreValve, and with six other companies.
On Twitter @sherryboschert
SAN FRANCISCO – Transcatheter aortic valve replacement with the self-expanding CoreValve in patients at extreme surgical risk significantly reduced the rate of death or major stroke at 1 year, from 43% to 26%, in a 487-patient pivotal trial.
The CoreValve Extreme Risk study gathered data from a registry of patients with symptomatic severe aortic stenosis who attempted an iliofemoral implantation procedure with the CoreValve at 40 U.S. sites. Results were compared with an "objective performance goal" derived from two sources: a meta-analysis of five contemporary balloon valvuloplasty series that found a 43% mortality and major stroke rate at 1 year, and the 1-year rate from the PARTNER B trial in inoperable patients (Placement of Aortic Transcatheter Valves, Cohort B), which was 50% but had a lower confidence bound of 43%.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The performance-goal comparison was necessary in the current study because randomizing these patients to medical therapy in a control group is no longer an acceptable option in the United States, Dr. Jeffrey J. Popma said at the Transcatheter Cardiovascular Therapeutics annual meeting.
The 1-year all-cause mortality rate was 24%, and the cardiovascular mortality was 18%, reported Dr. Popma, professor of medicine at Harvard Medical School, Boston.
Two percent of patients developed a major stroke within 1 month and 4% did so within 1 year.
Data from a continued access study involving another 830 extreme-risk patients who received the CoreValve through an iliofemoral approach are showing even better results, with a 16% rate of mortality or major stroke at 6 months, he added.
Among secondary endpoints at 1 year in the main study, 7% of patients developed any kind of stroke, 2% had an MI, 2% need reintervention, 41% had bleeding that met Valve Academic Research Consortium criteria, 8% had major vascular complications, and 27% required implantation of a permanent pacemaker, Dr. Popma reported at the meeting, cosponsored by the American College of Cardiology. Ninety percent of patients improved by at least one New York Heart Association functional class and 60% improved by at least two classes at 1 year of follow-up.
Paravalvular regurgitation of any severity was seen in 53% of patients 1 month after implantation and in 33% at 1 year. Moderate paravalvular leakage (PVL) affected 9% at 1 month and 4% at 1 year, and severe PVL affected 1.6% at 1 month and no patients at 1 year, Dr. Popma said. Among the 11% of patients with moderate PVL at 1 month, 80% of those who survived to 1 year had a reduction in leakage over time.
"We believe that’s why we did not find an association in the study between mild or moderate aortic regurgitation with respect to late-term mortality," though mortality risk was substantially higher with severe regurgitation, he said. One-year mortality rates were 86% with severe PVL and 24% with either moderate or mild PVL, compared with 18% in patients with no PVL.
The improvement in PVL rates over time may be due to use of CT angiography to select appropriate valve sizes for patients and continued expansion of the self-expanding frame over time. "That’s a remarkable finding, and it needs to be confirmed," Dr. Popma said.
The study focused on patients whose severe frailty, comorbidity, or disability put them at extreme risk of at least a 50% chance of death or irreversible morbidity within 30 days had they undergone surgical aortic valve replacement. A second U.S. pivotal trial of the CoreValve is focusing on patients at high (but not extreme) risk.
Most of the sites in the study had no experience with CoreValve before this study, Dr. Michael J. Mack noted at a press briefing. "The results are outstanding, but especially putting it in that light," said Dr. Mack, director of cardiovascular disease for the Baylor Scott & White Health System, Dallas, and a member of the steering committee for the PARTNER trial.
Dr. Popma reported financial associations with Medtronic, which sponsored the study and makes CoreValve, and with six other companies.
On Twitter @sherryboschert
AT TCT 2013
Major finding: The rate of all-cause mortality or stroke at 1 year was 26%, compared with an objective performance goal of 43%.
Data source: Study of registry data on 487 patients with symptomatic severe aortic stenosis and attempted CoreValve implantation via iliofemoral access.
Disclosures: Dr. Popma reported financial associations with Medtronic, which sponsored the study and makes CoreValve, and with six other companies.
CV events scuttle bardoxolone for diabetic kidney disease
Bardoxolone methyl may have reduced the risk of end-stage renal disease in patients with type 2 diabetes and stage 4 chronic kidney disease in a phase III clinical trial, but researchers stopped the study early after patients taking the drug had an increased rate of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke.
The BEACON trial had only an estimated 40% statistical power to determine bardoxolone methyl’s true effects, given its termination because of safety concerns, according to data reported at the annual Kidney Week meeting sponsored by the American Society of Nephrology.
The findings were simultaneously reported online Nov. 9 in the New England Journal of Medicine (doi:10.1056/NEJMoa1306033).
Bardoxolone methyl, the most potent known activator of a transcription factor that regulates antioxidant genes, was previously shown to raise the estimated glomerular filtration rate (eGFR) in patients with diabetes-related kidney disease. However, it also increased the incidence of albuminuria and induced unintended weight loss.
To determine whether longer-term treatment with bardoxolone methyl might translate that eGFR benefit into a slower progression to end-stage renal disease (ESRD), Dr. Dick de Zeeuw of the University of Groningen (the Netherlands) and his associates performed the double-blind BEACON trial (Bardoxolone Methyl Evaluation in Patients With Chronic Kidney Disease and Type 2 Diabetes Mellitus: The Occurrence of Renal Events).
BEACON was sponsored by Reata Pharmaceuticals and included patients from the United States, Europe, Australia, Canada, Israel, and Mexico, resulting in a diverse patient population with regard to age, race/ethnicity, and area of residence. Both diabetic retinopathy and neuropathy were common comorbidities, as was cardiovascular disease.
The 2,185 study participants had type 2 diabetes and moderate to severe chronic kidney diseases, with a baseline eGFR of 15 to less than 30 mL/min per 1.73 m2 of body surface area. They were randomly assigned to receive either once-daily bardoxolone methyl 20 mg (1,088 patients) or a matching placebo (1,097 patients), along with conventional background therapies given at the discretion of their treating physicians. Those included inhibitors of the renin-angiotensin-aldosterone system, insulin or other hypoglycemic agents, and appropriate cardiovascular medications.
The median duration of exposure was 7 months for bardoxolone methyl and 8 months for placebo, and the median follow-up for both was 9 months.
Bardoxolone significantly improved eGFR, compared with placebo, and fewer patients who took the drug progressed to ESRD. But BEACON was terminated early because of an excess of CV events among patients receiving bardoxolone methyl. That "truncated" study duration limited the trial’s statistical power.
The primary endpoint was a composite of progression to ESRD or cardiovascular death, and it occurred in 6% of both study groups. However, deaths from CV causes were significantly more frequent in the active treatment group (27 patients) than in the placebo group (19 patients), with a hazard ratio of 1.44, Dr. de Zeeuw reported.
In particular, 96 patients in the bardoxolone group had heart failure (HF) events, compared with only 55 patients in the placebo group. Moreover, significantly more of the HF events in the active treatment group were severe enough to require hospitalization or cause death.
Similarly, significantly more patients taking bardoxolone methyl had a composite outcome of nonfatal myocardial infarction, nonfatal stroke, HF hospitalization, or CV death. And the number of deaths from any cause was greater – though not significantly – with bardoxolone methyl (44 deaths) than with placebo (31 deaths) (HR, 1.47; P = .10).
Compared with placebo, bardoxolone methyl also raised blood pressure and heart rate, increased levels of B-type natriuretic peptide, raised the rate of albuminuria, and caused substantial unintended weight loss. The investigators were unable to determine whether there was a loss of body fat, intracellular (that is, skeletal muscle) water, or extracellular (interstitial) water. There was a concomitant fall in serum albumin and hemoglobin levels, which may reflect hemodilution caused by fluid retention, the investigators found.
The increases in blood pressure and heart rate "constitute a potentially potent combination of factors that are likely to precipitate HF in an at-risk population." and the increase in B-type natriuretic peptide "is consistent with an increase in left ventricular wall stress," Dr. de Zeeuw said.
The investigators attempted to identify patient characteristics that might be associated with the development of HF in the bardoxolone methyl recipients, but were unable to do so.
Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
*This article was updated November 11, 2013.
The adverse events linked to bardoxolone methyl in this study included excess HF and cardiovascular events, as well as increased rates of high blood pressure, high heart rate, albuminuria, GI symptoms, and muscle-related symptoms, said Dr. Jonathan Himmelfarb and Dr. Katherine R. Tuttle.
"The authors speculate that fluid retention, increased afterload, and higher heart rate contributed to heart failure," but it also is possible that bardoxolone methyl may exert direct toxic effects on the heart, Dr. Himmelfarb and Dr. Tuttle said.
In any case, "caution should be exercised whenever any drug for diabetic kidney disease increases, rather than decreases, albuminuria," they noted.
Dr. Himmelfarb and Dr. Tuttle are at the Kidney Research Institute and the division of nephrology at the University of Washington, Seattle. Dr. Tuttle also is at Providence Sacred Heart Medical Center and Children’s Hospital, Spokane. Dr. Himmelfarb reported ties to Abbott Laboratories, and Dr. Tuttle reported ties to Eli Lilly. These remarks were taken from their editorial accompanying Dr. de Zeeuw’s report (N. Engl. J. Med. 2013 Nov. 9 [doi:10.1056/NEJMe1313104]).
The adverse events linked to bardoxolone methyl in this study included excess HF and cardiovascular events, as well as increased rates of high blood pressure, high heart rate, albuminuria, GI symptoms, and muscle-related symptoms, said Dr. Jonathan Himmelfarb and Dr. Katherine R. Tuttle.
"The authors speculate that fluid retention, increased afterload, and higher heart rate contributed to heart failure," but it also is possible that bardoxolone methyl may exert direct toxic effects on the heart, Dr. Himmelfarb and Dr. Tuttle said.
In any case, "caution should be exercised whenever any drug for diabetic kidney disease increases, rather than decreases, albuminuria," they noted.
Dr. Himmelfarb and Dr. Tuttle are at the Kidney Research Institute and the division of nephrology at the University of Washington, Seattle. Dr. Tuttle also is at Providence Sacred Heart Medical Center and Children’s Hospital, Spokane. Dr. Himmelfarb reported ties to Abbott Laboratories, and Dr. Tuttle reported ties to Eli Lilly. These remarks were taken from their editorial accompanying Dr. de Zeeuw’s report (N. Engl. J. Med. 2013 Nov. 9 [doi:10.1056/NEJMe1313104]).
The adverse events linked to bardoxolone methyl in this study included excess HF and cardiovascular events, as well as increased rates of high blood pressure, high heart rate, albuminuria, GI symptoms, and muscle-related symptoms, said Dr. Jonathan Himmelfarb and Dr. Katherine R. Tuttle.
"The authors speculate that fluid retention, increased afterload, and higher heart rate contributed to heart failure," but it also is possible that bardoxolone methyl may exert direct toxic effects on the heart, Dr. Himmelfarb and Dr. Tuttle said.
In any case, "caution should be exercised whenever any drug for diabetic kidney disease increases, rather than decreases, albuminuria," they noted.
Dr. Himmelfarb and Dr. Tuttle are at the Kidney Research Institute and the division of nephrology at the University of Washington, Seattle. Dr. Tuttle also is at Providence Sacred Heart Medical Center and Children’s Hospital, Spokane. Dr. Himmelfarb reported ties to Abbott Laboratories, and Dr. Tuttle reported ties to Eli Lilly. These remarks were taken from their editorial accompanying Dr. de Zeeuw’s report (N. Engl. J. Med. 2013 Nov. 9 [doi:10.1056/NEJMe1313104]).
Bardoxolone methyl may have reduced the risk of end-stage renal disease in patients with type 2 diabetes and stage 4 chronic kidney disease in a phase III clinical trial, but researchers stopped the study early after patients taking the drug had an increased rate of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke.
The BEACON trial had only an estimated 40% statistical power to determine bardoxolone methyl’s true effects, given its termination because of safety concerns, according to data reported at the annual Kidney Week meeting sponsored by the American Society of Nephrology.
The findings were simultaneously reported online Nov. 9 in the New England Journal of Medicine (doi:10.1056/NEJMoa1306033).
Bardoxolone methyl, the most potent known activator of a transcription factor that regulates antioxidant genes, was previously shown to raise the estimated glomerular filtration rate (eGFR) in patients with diabetes-related kidney disease. However, it also increased the incidence of albuminuria and induced unintended weight loss.
To determine whether longer-term treatment with bardoxolone methyl might translate that eGFR benefit into a slower progression to end-stage renal disease (ESRD), Dr. Dick de Zeeuw of the University of Groningen (the Netherlands) and his associates performed the double-blind BEACON trial (Bardoxolone Methyl Evaluation in Patients With Chronic Kidney Disease and Type 2 Diabetes Mellitus: The Occurrence of Renal Events).
BEACON was sponsored by Reata Pharmaceuticals and included patients from the United States, Europe, Australia, Canada, Israel, and Mexico, resulting in a diverse patient population with regard to age, race/ethnicity, and area of residence. Both diabetic retinopathy and neuropathy were common comorbidities, as was cardiovascular disease.
The 2,185 study participants had type 2 diabetes and moderate to severe chronic kidney diseases, with a baseline eGFR of 15 to less than 30 mL/min per 1.73 m2 of body surface area. They were randomly assigned to receive either once-daily bardoxolone methyl 20 mg (1,088 patients) or a matching placebo (1,097 patients), along with conventional background therapies given at the discretion of their treating physicians. Those included inhibitors of the renin-angiotensin-aldosterone system, insulin or other hypoglycemic agents, and appropriate cardiovascular medications.
The median duration of exposure was 7 months for bardoxolone methyl and 8 months for placebo, and the median follow-up for both was 9 months.
Bardoxolone significantly improved eGFR, compared with placebo, and fewer patients who took the drug progressed to ESRD. But BEACON was terminated early because of an excess of CV events among patients receiving bardoxolone methyl. That "truncated" study duration limited the trial’s statistical power.
The primary endpoint was a composite of progression to ESRD or cardiovascular death, and it occurred in 6% of both study groups. However, deaths from CV causes were significantly more frequent in the active treatment group (27 patients) than in the placebo group (19 patients), with a hazard ratio of 1.44, Dr. de Zeeuw reported.
In particular, 96 patients in the bardoxolone group had heart failure (HF) events, compared with only 55 patients in the placebo group. Moreover, significantly more of the HF events in the active treatment group were severe enough to require hospitalization or cause death.
Similarly, significantly more patients taking bardoxolone methyl had a composite outcome of nonfatal myocardial infarction, nonfatal stroke, HF hospitalization, or CV death. And the number of deaths from any cause was greater – though not significantly – with bardoxolone methyl (44 deaths) than with placebo (31 deaths) (HR, 1.47; P = .10).
Compared with placebo, bardoxolone methyl also raised blood pressure and heart rate, increased levels of B-type natriuretic peptide, raised the rate of albuminuria, and caused substantial unintended weight loss. The investigators were unable to determine whether there was a loss of body fat, intracellular (that is, skeletal muscle) water, or extracellular (interstitial) water. There was a concomitant fall in serum albumin and hemoglobin levels, which may reflect hemodilution caused by fluid retention, the investigators found.
The increases in blood pressure and heart rate "constitute a potentially potent combination of factors that are likely to precipitate HF in an at-risk population." and the increase in B-type natriuretic peptide "is consistent with an increase in left ventricular wall stress," Dr. de Zeeuw said.
The investigators attempted to identify patient characteristics that might be associated with the development of HF in the bardoxolone methyl recipients, but were unable to do so.
Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
*This article was updated November 11, 2013.
Bardoxolone methyl may have reduced the risk of end-stage renal disease in patients with type 2 diabetes and stage 4 chronic kidney disease in a phase III clinical trial, but researchers stopped the study early after patients taking the drug had an increased rate of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke.
The BEACON trial had only an estimated 40% statistical power to determine bardoxolone methyl’s true effects, given its termination because of safety concerns, according to data reported at the annual Kidney Week meeting sponsored by the American Society of Nephrology.
The findings were simultaneously reported online Nov. 9 in the New England Journal of Medicine (doi:10.1056/NEJMoa1306033).
Bardoxolone methyl, the most potent known activator of a transcription factor that regulates antioxidant genes, was previously shown to raise the estimated glomerular filtration rate (eGFR) in patients with diabetes-related kidney disease. However, it also increased the incidence of albuminuria and induced unintended weight loss.
To determine whether longer-term treatment with bardoxolone methyl might translate that eGFR benefit into a slower progression to end-stage renal disease (ESRD), Dr. Dick de Zeeuw of the University of Groningen (the Netherlands) and his associates performed the double-blind BEACON trial (Bardoxolone Methyl Evaluation in Patients With Chronic Kidney Disease and Type 2 Diabetes Mellitus: The Occurrence of Renal Events).
BEACON was sponsored by Reata Pharmaceuticals and included patients from the United States, Europe, Australia, Canada, Israel, and Mexico, resulting in a diverse patient population with regard to age, race/ethnicity, and area of residence. Both diabetic retinopathy and neuropathy were common comorbidities, as was cardiovascular disease.
The 2,185 study participants had type 2 diabetes and moderate to severe chronic kidney diseases, with a baseline eGFR of 15 to less than 30 mL/min per 1.73 m2 of body surface area. They were randomly assigned to receive either once-daily bardoxolone methyl 20 mg (1,088 patients) or a matching placebo (1,097 patients), along with conventional background therapies given at the discretion of their treating physicians. Those included inhibitors of the renin-angiotensin-aldosterone system, insulin or other hypoglycemic agents, and appropriate cardiovascular medications.
The median duration of exposure was 7 months for bardoxolone methyl and 8 months for placebo, and the median follow-up for both was 9 months.
Bardoxolone significantly improved eGFR, compared with placebo, and fewer patients who took the drug progressed to ESRD. But BEACON was terminated early because of an excess of CV events among patients receiving bardoxolone methyl. That "truncated" study duration limited the trial’s statistical power.
The primary endpoint was a composite of progression to ESRD or cardiovascular death, and it occurred in 6% of both study groups. However, deaths from CV causes were significantly more frequent in the active treatment group (27 patients) than in the placebo group (19 patients), with a hazard ratio of 1.44, Dr. de Zeeuw reported.
In particular, 96 patients in the bardoxolone group had heart failure (HF) events, compared with only 55 patients in the placebo group. Moreover, significantly more of the HF events in the active treatment group were severe enough to require hospitalization or cause death.
Similarly, significantly more patients taking bardoxolone methyl had a composite outcome of nonfatal myocardial infarction, nonfatal stroke, HF hospitalization, or CV death. And the number of deaths from any cause was greater – though not significantly – with bardoxolone methyl (44 deaths) than with placebo (31 deaths) (HR, 1.47; P = .10).
Compared with placebo, bardoxolone methyl also raised blood pressure and heart rate, increased levels of B-type natriuretic peptide, raised the rate of albuminuria, and caused substantial unintended weight loss. The investigators were unable to determine whether there was a loss of body fat, intracellular (that is, skeletal muscle) water, or extracellular (interstitial) water. There was a concomitant fall in serum albumin and hemoglobin levels, which may reflect hemodilution caused by fluid retention, the investigators found.
The increases in blood pressure and heart rate "constitute a potentially potent combination of factors that are likely to precipitate HF in an at-risk population." and the increase in B-type natriuretic peptide "is consistent with an increase in left ventricular wall stress," Dr. de Zeeuw said.
The investigators attempted to identify patient characteristics that might be associated with the development of HF in the bardoxolone methyl recipients, but were unable to do so.
Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
*This article was updated November 11, 2013.
FROM KIDNEY WEEK
Major Finding: Bardoxolone methyl improved eGFR and may have delayed progression to ESRD, but that result was inconclusive because the trial was terminated early as a result of an excess of cardiovascular deaths, heart failure events, nonfatal myocardial infarction, and nonfatal stroke in patients given the drug.
Data Source: An international phase III double-blind trial involving 2,185 patients with type 2 diabetes and stage 4 chronic kidney disease who were randomly assigned to receive daily oral bardoxolone methyl or placebo and were followed for a median of 9 months.
Disclosures: Reata Pharmaceuticals funded the BEACON trial. Dr. de Zeeuw reported ties to AbbVie, Astellas, Chemocentryx, Johnson & Johnson, and Reata, and his associates reported ties to numerous industry sources.
Heart irradiation is lower with contemporary breast radiotherapy
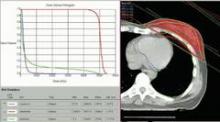
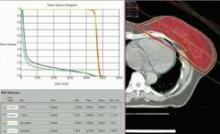
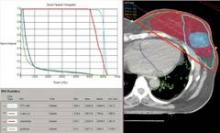
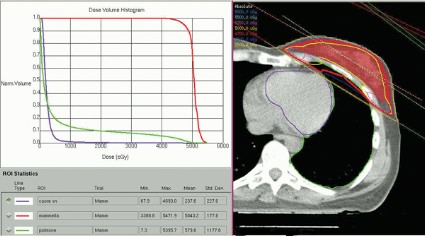
SAN FRANCISCO – The hearts of 100 consecutive patients who underwent adjuvant radiotherapy for left-sided breast cancer in 2011 received an average of 2.9 Gray of radiation, considerably less than the mean cardiac exposure of 4.9-Gy reported in a recent review of 2,168 patients treated from 1958 to 2001 in Sweden and Denmark.
The findings confirm that three-dimensional conformal radiation therapy (3D-CRT) reduces cardiac exposure to radiation, Dr. Federico Lonardi and his associates reported at a breast cancer symposium sponsored by the American Society of Clinical Oncology. But certain areas of the heart still receive high doses when patients have adverse anatomic conditions that are not well suited to 3D-CRT. Because heart structures may differ in radiosensitivity, higher doses to small volumes of the heart, such as the coronary artery, might be associated with more risk, the researchers cautioned.
Most patients received a mean cardiac dose of 2-3 Gy (32%), 21% of patients were exposed to 1.15-1.99 Gy, and 1% got 0.8 Gy in a study of a consecutive series of breast cancer patients treated at Mater Salutis Hospital in Legnago, Italy. Only 17% of patients received a mean cardiac dose of more than 5 Gy, and 13% received 4.16-4.83 Gy.
The cardiac dose ranged from 0.8 to 13.05 Gy in Dr. Lonardi’s study, compared with a range of 0.03 to 27.72 Gy in the recently published Scandinavian study (N. Engl. J. Med. 2013;368:987-998). In the published study, the longitudinal risk for major cardiac events increased in a linear fashion, with a 7% increase for cardiac events with every 1 Gy increase in radiation to the heart.
In the Italian study, the median volumes of heart exposed to higher doses of radiation were "consistently low" with 4% of heart volumes exposed to 5 Gy or more, 3% exposed to 10Gy or more, 2% exposed to 15 Gy or more, and 0.7% exposed to 25 Gy or more Dr. Lonardi reported.
These patients received full-breast 3D-CRT with two to four customized tangential fields after mastectomy (10% of patients) or quadrantectomy (90%). The whole breast (or chest wall) received 50 Gy/25 fractions in 66 patients and 45 Gy/18 fractions in 34 patients. Boost to surgical bed (10 Gy/4-5 fractions) was delivered by photons in 10 patients. Median number of tangential fields was two (range, two to four). Patients were treated while supine on a breast board, without immobilization devices or instructions to hold their breath. They were freely breathing but were asked to minimize respiratory motion during the CT scan used to plan radiation delivery and the treatment itself. No dose constraints were specified for heart structures; a mean heart dose lower than 5 Gy was recommended at the time of treatment.
A preliminary assessment of radiation delivered to the left anterior descending coronary arteries in this series suggests that they received 9-25 Gy, Dr. Lonardi reported.
Based on estimates using previous models, the probability of death from cardiac causes within 15 years after standard fractionated radiotherapy may be less than 1% if less than 10% of the heart is exposed to 25Gy or more, he noted. "In this perspective, our results appear very favorable, though they confirm that the heart may receive high doses to limited volumes despite the use of standard 3D techniques. In such cases, high-conformal, intensity-modulated techniques are helpful" to further reduce the exposure of critical heart structures to radiation.
The symposium was cosponsored by the American Society of Breast Disease, the American Society of Breast Surgeons, the National Consortium of Breast Centers, the Society of Surgical Oncology, and the American Society for Radiation Oncology.
Dr. Lonardi reported having no financial disclosures.
On Twitter @sherryboschert
This was an interesting study. I really give the authors a lot of credit. They basically looked at 100 consecutive cases of patients treated with adjuvant radiotherapy for left-sided breast cancer and brought forward what they were doing without trying to optimize or minimize the presentation or cardiac dose in the patients they were treating.

|
| Dr. Julia White |
Their mean doses were lower than those reported by Darby et al. in a study that looked at 2,168 patients treated during 1958-2001 in both Sweden and Denmark. The researchers reviewed individual radiotherapy charts, and then did 20 consecutive individual CT-based three-dimensional planning scans to model what the type of radiotherapy looked like many years ago (N. Engl. J. Med. 2013;368:987-98).
This was an interesting study. I really give the authors a lot of credit. They basically looked at 100 consecutive cases of patients treated with adjuvant radiotherapy for left-sided breast cancer and brought forward what they were doing without trying to optimize or minimize the presentation or cardiac dose in the patients they were treating.

|
| Dr. Julia White |
Their mean doses were lower than those reported by Darby et al. in a study that looked at 2,168 patients treated during 1958-2001 in both Sweden and Denmark. The researchers reviewed individual radiotherapy charts, and then did 20 consecutive individual CT-based three-dimensional planning scans to model what the type of radiotherapy looked like many years ago (N. Engl. J. Med. 2013;368:987-98).
This was an interesting study. I really give the authors a lot of credit. They basically looked at 100 consecutive cases of patients treated with adjuvant radiotherapy for left-sided breast cancer and brought forward what they were doing without trying to optimize or minimize the presentation or cardiac dose in the patients they were treating.

|
| Dr. Julia White |
Their mean doses were lower than those reported by Darby et al. in a study that looked at 2,168 patients treated during 1958-2001 in both Sweden and Denmark. The researchers reviewed individual radiotherapy charts, and then did 20 consecutive individual CT-based three-dimensional planning scans to model what the type of radiotherapy looked like many years ago (N. Engl. J. Med. 2013;368:987-98).
SAN FRANCISCO – The hearts of 100 consecutive patients who underwent adjuvant radiotherapy for left-sided breast cancer in 2011 received an average of 2.9 Gray of radiation, considerably less than the mean cardiac exposure of 4.9-Gy reported in a recent review of 2,168 patients treated from 1958 to 2001 in Sweden and Denmark.
The findings confirm that three-dimensional conformal radiation therapy (3D-CRT) reduces cardiac exposure to radiation, Dr. Federico Lonardi and his associates reported at a breast cancer symposium sponsored by the American Society of Clinical Oncology. But certain areas of the heart still receive high doses when patients have adverse anatomic conditions that are not well suited to 3D-CRT. Because heart structures may differ in radiosensitivity, higher doses to small volumes of the heart, such as the coronary artery, might be associated with more risk, the researchers cautioned.
Most patients received a mean cardiac dose of 2-3 Gy (32%), 21% of patients were exposed to 1.15-1.99 Gy, and 1% got 0.8 Gy in a study of a consecutive series of breast cancer patients treated at Mater Salutis Hospital in Legnago, Italy. Only 17% of patients received a mean cardiac dose of more than 5 Gy, and 13% received 4.16-4.83 Gy.
The cardiac dose ranged from 0.8 to 13.05 Gy in Dr. Lonardi’s study, compared with a range of 0.03 to 27.72 Gy in the recently published Scandinavian study (N. Engl. J. Med. 2013;368:987-998). In the published study, the longitudinal risk for major cardiac events increased in a linear fashion, with a 7% increase for cardiac events with every 1 Gy increase in radiation to the heart.
In the Italian study, the median volumes of heart exposed to higher doses of radiation were "consistently low" with 4% of heart volumes exposed to 5 Gy or more, 3% exposed to 10Gy or more, 2% exposed to 15 Gy or more, and 0.7% exposed to 25 Gy or more Dr. Lonardi reported.
These patients received full-breast 3D-CRT with two to four customized tangential fields after mastectomy (10% of patients) or quadrantectomy (90%). The whole breast (or chest wall) received 50 Gy/25 fractions in 66 patients and 45 Gy/18 fractions in 34 patients. Boost to surgical bed (10 Gy/4-5 fractions) was delivered by photons in 10 patients. Median number of tangential fields was two (range, two to four). Patients were treated while supine on a breast board, without immobilization devices or instructions to hold their breath. They were freely breathing but were asked to minimize respiratory motion during the CT scan used to plan radiation delivery and the treatment itself. No dose constraints were specified for heart structures; a mean heart dose lower than 5 Gy was recommended at the time of treatment.
A preliminary assessment of radiation delivered to the left anterior descending coronary arteries in this series suggests that they received 9-25 Gy, Dr. Lonardi reported.
Based on estimates using previous models, the probability of death from cardiac causes within 15 years after standard fractionated radiotherapy may be less than 1% if less than 10% of the heart is exposed to 25Gy or more, he noted. "In this perspective, our results appear very favorable, though they confirm that the heart may receive high doses to limited volumes despite the use of standard 3D techniques. In such cases, high-conformal, intensity-modulated techniques are helpful" to further reduce the exposure of critical heart structures to radiation.
The symposium was cosponsored by the American Society of Breast Disease, the American Society of Breast Surgeons, the National Consortium of Breast Centers, the Society of Surgical Oncology, and the American Society for Radiation Oncology.
Dr. Lonardi reported having no financial disclosures.
On Twitter @sherryboschert
SAN FRANCISCO – The hearts of 100 consecutive patients who underwent adjuvant radiotherapy for left-sided breast cancer in 2011 received an average of 2.9 Gray of radiation, considerably less than the mean cardiac exposure of 4.9-Gy reported in a recent review of 2,168 patients treated from 1958 to 2001 in Sweden and Denmark.
The findings confirm that three-dimensional conformal radiation therapy (3D-CRT) reduces cardiac exposure to radiation, Dr. Federico Lonardi and his associates reported at a breast cancer symposium sponsored by the American Society of Clinical Oncology. But certain areas of the heart still receive high doses when patients have adverse anatomic conditions that are not well suited to 3D-CRT. Because heart structures may differ in radiosensitivity, higher doses to small volumes of the heart, such as the coronary artery, might be associated with more risk, the researchers cautioned.
Most patients received a mean cardiac dose of 2-3 Gy (32%), 21% of patients were exposed to 1.15-1.99 Gy, and 1% got 0.8 Gy in a study of a consecutive series of breast cancer patients treated at Mater Salutis Hospital in Legnago, Italy. Only 17% of patients received a mean cardiac dose of more than 5 Gy, and 13% received 4.16-4.83 Gy.
The cardiac dose ranged from 0.8 to 13.05 Gy in Dr. Lonardi’s study, compared with a range of 0.03 to 27.72 Gy in the recently published Scandinavian study (N. Engl. J. Med. 2013;368:987-998). In the published study, the longitudinal risk for major cardiac events increased in a linear fashion, with a 7% increase for cardiac events with every 1 Gy increase in radiation to the heart.
In the Italian study, the median volumes of heart exposed to higher doses of radiation were "consistently low" with 4% of heart volumes exposed to 5 Gy or more, 3% exposed to 10Gy or more, 2% exposed to 15 Gy or more, and 0.7% exposed to 25 Gy or more Dr. Lonardi reported.
These patients received full-breast 3D-CRT with two to four customized tangential fields after mastectomy (10% of patients) or quadrantectomy (90%). The whole breast (or chest wall) received 50 Gy/25 fractions in 66 patients and 45 Gy/18 fractions in 34 patients. Boost to surgical bed (10 Gy/4-5 fractions) was delivered by photons in 10 patients. Median number of tangential fields was two (range, two to four). Patients were treated while supine on a breast board, without immobilization devices or instructions to hold their breath. They were freely breathing but were asked to minimize respiratory motion during the CT scan used to plan radiation delivery and the treatment itself. No dose constraints were specified for heart structures; a mean heart dose lower than 5 Gy was recommended at the time of treatment.
A preliminary assessment of radiation delivered to the left anterior descending coronary arteries in this series suggests that they received 9-25 Gy, Dr. Lonardi reported.
Based on estimates using previous models, the probability of death from cardiac causes within 15 years after standard fractionated radiotherapy may be less than 1% if less than 10% of the heart is exposed to 25Gy or more, he noted. "In this perspective, our results appear very favorable, though they confirm that the heart may receive high doses to limited volumes despite the use of standard 3D techniques. In such cases, high-conformal, intensity-modulated techniques are helpful" to further reduce the exposure of critical heart structures to radiation.
The symposium was cosponsored by the American Society of Breast Disease, the American Society of Breast Surgeons, the National Consortium of Breast Centers, the Society of Surgical Oncology, and the American Society for Radiation Oncology.
Dr. Lonardi reported having no financial disclosures.
On Twitter @sherryboschert
AT THE ASCO BREAST CANCER SYMPOSIUM
Major finding: In an Italian study, the median volumes of heart exposed to higher doses of radiation were "consistently low" with 4% of heart volumes exposed to 5 Gy or more, 3% exposed to 10Gy or more, 2% exposed to 15 Gy or more, and 0.7% exposed to 25 Gy or more.
Data source: Retrospective review of 100 consecutive patients treated with radiotherapy for left-sided breast cancer at one institution in 2011.
Disclosures: Dr. Lonardi reported having no financial disclosures.
Trial explores ranolazine in pulmonary hypertension-linked heart failure
CHICAGO – The antianginal drug ranolazine improved hemodynamics and functional status in patients with pulmonary hypertension associated with heart failure with preserved ejection fraction in a proof-of-concept trial of just 10 patients.
Six months of twice-daily ranolazine (Ranexa) decreased baseline mean pulmonary arterial pressure (PAP) by 41% and mean pulmonary capillary wedge pressure (PCWP) by 40% in this hard-to-treat population.
Half of the patients improved from World Health Organization functional class III to II, and no patient deteriorated.
"This pilot study suggests ranolazine might be a promising treatment for pulmonary hypertension associated with heart failure with preserved EF [ejection fraction]," Dr. Harrison Farber said during a late-breaking abstract session at the annual meeting of the American College of Chest Physicians.
Pulmonary hypertension (PH) associated with heart failure with preserved ejection fraction (HFpEF) is an increasingly common condition, but currently no effective treatment exists for HFpEF alone or PH associated with it. Disappointing results were reported earlier this year for sildenafil (Viagra) in patients with diastolic heart failure in the RELAX trial.
"This pilot study suggests ranolazine might be a promising treatment for pulmonary hypertension associated with heart failure with preserved EF [ejection fraction]..."
Ranolazine is approved for the treatment of chronic angina, and was selected because animal and human data suggest it reduces left ventricular wall stiffness and mechanical dysfunction in conditions associated with diastolic dysfunction, said Dr. Farber, director of the pulmonary hypertension center, Boston University.
To ensure patients in the single-center, open-label, prospective, non–placebo controlled trial truly had HFpEF, entry criteria were fairly strict. They included left ventricular ejection fraction (LVEF) greater than 50% by echocardiogram, mean PAP of at least 25 mm Hg, PCWP between 18 mm Hg and 30 mm Hg, and a pulmonary arterial diastolic pressure-PCWP gradient of 10 mm Hg or less.
The eight women and two men received twice-daily ranolazine 500 mg, titrated to 1,000 mg twice daily, as tolerated. Their mean age was 63 years, mean body mass index 41.6 kg/m2, and 80% had diabetes.
At 6 months, mean PAP dropped from 39 mm Hg to 23 mm Hg, mean PCWP from 22 mm Hg to 13 mm HG, and 6-minute walk distances increased from 286 meters to 319 meters, Dr. Farber said.
"Seven of the subjects have actually continued ranolazine for a year or greater and the 6-minute-walk and functional class improvements have actually been maintained to this point," he said.
No significant changes were seen in cardiac index, pulmonary vascular resistance, echocardiogram parameters, brain natriuretic peptide, N-terminal pro-BNP, or troponin.
There was, however, a significant reduction in LV mass at 6 months on blinded MRI evaluation, and a trend for improved right ventricular ejection fraction and decrease in RV mass, he said.
Session comoderator Dr. Namita Sood with Ohio State University in Columbus congratulated the authors on "a courageous study" in an understudied population and asked whether the drop in PAP and PCWP was a consequence of improved LV and diastolic dysfunction or whether ranolazine may have a direct effect on pulmonary vasculature.
Dr. Farber responded, "To be honest, the answer is I don’t know, but since the two were sort of in concert, my guess would be – although it was a small study and I can’t tell you for sure – it was more of an effect on the left ventricle and a concomitant drop in the pulmonary pressures."
Some audience members questioned whether the investigators targeted the right population since patients did not appear to be sufficiently diuresed at entry. Dr. Farber said the exploratory analysis was done to determine if a signal was present and that tighter controls would be needed if they go forward. Diuretics were not controlled in the 10 patients, although no dramatic changes in doses occurred. No patients were on nitrates.
One patient discontinued treatment because of the worsening of an existing symptomatic bradyarrhythmia. Otherwise, the drug was well tolerated, with the only other side effect being headache that went away in the first week of treatment, he said.
Dr. Farber and his coauthors reported grant support from Gilead Sciences, which makes ranolazine.
This article was updated 11/7/13.
CHICAGO – The antianginal drug ranolazine improved hemodynamics and functional status in patients with pulmonary hypertension associated with heart failure with preserved ejection fraction in a proof-of-concept trial of just 10 patients.
Six months of twice-daily ranolazine (Ranexa) decreased baseline mean pulmonary arterial pressure (PAP) by 41% and mean pulmonary capillary wedge pressure (PCWP) by 40% in this hard-to-treat population.
Half of the patients improved from World Health Organization functional class III to II, and no patient deteriorated.
"This pilot study suggests ranolazine might be a promising treatment for pulmonary hypertension associated with heart failure with preserved EF [ejection fraction]," Dr. Harrison Farber said during a late-breaking abstract session at the annual meeting of the American College of Chest Physicians.
Pulmonary hypertension (PH) associated with heart failure with preserved ejection fraction (HFpEF) is an increasingly common condition, but currently no effective treatment exists for HFpEF alone or PH associated with it. Disappointing results were reported earlier this year for sildenafil (Viagra) in patients with diastolic heart failure in the RELAX trial.
"This pilot study suggests ranolazine might be a promising treatment for pulmonary hypertension associated with heart failure with preserved EF [ejection fraction]..."
Ranolazine is approved for the treatment of chronic angina, and was selected because animal and human data suggest it reduces left ventricular wall stiffness and mechanical dysfunction in conditions associated with diastolic dysfunction, said Dr. Farber, director of the pulmonary hypertension center, Boston University.
To ensure patients in the single-center, open-label, prospective, non–placebo controlled trial truly had HFpEF, entry criteria were fairly strict. They included left ventricular ejection fraction (LVEF) greater than 50% by echocardiogram, mean PAP of at least 25 mm Hg, PCWP between 18 mm Hg and 30 mm Hg, and a pulmonary arterial diastolic pressure-PCWP gradient of 10 mm Hg or less.
The eight women and two men received twice-daily ranolazine 500 mg, titrated to 1,000 mg twice daily, as tolerated. Their mean age was 63 years, mean body mass index 41.6 kg/m2, and 80% had diabetes.
At 6 months, mean PAP dropped from 39 mm Hg to 23 mm Hg, mean PCWP from 22 mm Hg to 13 mm HG, and 6-minute walk distances increased from 286 meters to 319 meters, Dr. Farber said.
"Seven of the subjects have actually continued ranolazine for a year or greater and the 6-minute-walk and functional class improvements have actually been maintained to this point," he said.
No significant changes were seen in cardiac index, pulmonary vascular resistance, echocardiogram parameters, brain natriuretic peptide, N-terminal pro-BNP, or troponin.
There was, however, a significant reduction in LV mass at 6 months on blinded MRI evaluation, and a trend for improved right ventricular ejection fraction and decrease in RV mass, he said.
Session comoderator Dr. Namita Sood with Ohio State University in Columbus congratulated the authors on "a courageous study" in an understudied population and asked whether the drop in PAP and PCWP was a consequence of improved LV and diastolic dysfunction or whether ranolazine may have a direct effect on pulmonary vasculature.
Dr. Farber responded, "To be honest, the answer is I don’t know, but since the two were sort of in concert, my guess would be – although it was a small study and I can’t tell you for sure – it was more of an effect on the left ventricle and a concomitant drop in the pulmonary pressures."
Some audience members questioned whether the investigators targeted the right population since patients did not appear to be sufficiently diuresed at entry. Dr. Farber said the exploratory analysis was done to determine if a signal was present and that tighter controls would be needed if they go forward. Diuretics were not controlled in the 10 patients, although no dramatic changes in doses occurred. No patients were on nitrates.
One patient discontinued treatment because of the worsening of an existing symptomatic bradyarrhythmia. Otherwise, the drug was well tolerated, with the only other side effect being headache that went away in the first week of treatment, he said.
Dr. Farber and his coauthors reported grant support from Gilead Sciences, which makes ranolazine.
This article was updated 11/7/13.
CHICAGO – The antianginal drug ranolazine improved hemodynamics and functional status in patients with pulmonary hypertension associated with heart failure with preserved ejection fraction in a proof-of-concept trial of just 10 patients.
Six months of twice-daily ranolazine (Ranexa) decreased baseline mean pulmonary arterial pressure (PAP) by 41% and mean pulmonary capillary wedge pressure (PCWP) by 40% in this hard-to-treat population.
Half of the patients improved from World Health Organization functional class III to II, and no patient deteriorated.
"This pilot study suggests ranolazine might be a promising treatment for pulmonary hypertension associated with heart failure with preserved EF [ejection fraction]," Dr. Harrison Farber said during a late-breaking abstract session at the annual meeting of the American College of Chest Physicians.
Pulmonary hypertension (PH) associated with heart failure with preserved ejection fraction (HFpEF) is an increasingly common condition, but currently no effective treatment exists for HFpEF alone or PH associated with it. Disappointing results were reported earlier this year for sildenafil (Viagra) in patients with diastolic heart failure in the RELAX trial.
"This pilot study suggests ranolazine might be a promising treatment for pulmonary hypertension associated with heart failure with preserved EF [ejection fraction]..."
Ranolazine is approved for the treatment of chronic angina, and was selected because animal and human data suggest it reduces left ventricular wall stiffness and mechanical dysfunction in conditions associated with diastolic dysfunction, said Dr. Farber, director of the pulmonary hypertension center, Boston University.
To ensure patients in the single-center, open-label, prospective, non–placebo controlled trial truly had HFpEF, entry criteria were fairly strict. They included left ventricular ejection fraction (LVEF) greater than 50% by echocardiogram, mean PAP of at least 25 mm Hg, PCWP between 18 mm Hg and 30 mm Hg, and a pulmonary arterial diastolic pressure-PCWP gradient of 10 mm Hg or less.
The eight women and two men received twice-daily ranolazine 500 mg, titrated to 1,000 mg twice daily, as tolerated. Their mean age was 63 years, mean body mass index 41.6 kg/m2, and 80% had diabetes.
At 6 months, mean PAP dropped from 39 mm Hg to 23 mm Hg, mean PCWP from 22 mm Hg to 13 mm HG, and 6-minute walk distances increased from 286 meters to 319 meters, Dr. Farber said.
"Seven of the subjects have actually continued ranolazine for a year or greater and the 6-minute-walk and functional class improvements have actually been maintained to this point," he said.
No significant changes were seen in cardiac index, pulmonary vascular resistance, echocardiogram parameters, brain natriuretic peptide, N-terminal pro-BNP, or troponin.
There was, however, a significant reduction in LV mass at 6 months on blinded MRI evaluation, and a trend for improved right ventricular ejection fraction and decrease in RV mass, he said.
Session comoderator Dr. Namita Sood with Ohio State University in Columbus congratulated the authors on "a courageous study" in an understudied population and asked whether the drop in PAP and PCWP was a consequence of improved LV and diastolic dysfunction or whether ranolazine may have a direct effect on pulmonary vasculature.
Dr. Farber responded, "To be honest, the answer is I don’t know, but since the two were sort of in concert, my guess would be – although it was a small study and I can’t tell you for sure – it was more of an effect on the left ventricle and a concomitant drop in the pulmonary pressures."
Some audience members questioned whether the investigators targeted the right population since patients did not appear to be sufficiently diuresed at entry. Dr. Farber said the exploratory analysis was done to determine if a signal was present and that tighter controls would be needed if they go forward. Diuretics were not controlled in the 10 patients, although no dramatic changes in doses occurred. No patients were on nitrates.
One patient discontinued treatment because of the worsening of an existing symptomatic bradyarrhythmia. Otherwise, the drug was well tolerated, with the only other side effect being headache that went away in the first week of treatment, he said.
Dr. Farber and his coauthors reported grant support from Gilead Sciences, which makes ranolazine.
This article was updated 11/7/13.
AT CHEST 2013
Major finding: At 6 months, ranolazine decreased baseline mean pulmonary arterial pressure 41% and mean pulmonary wedge pressure by 40%.
Data source: A prospective open-label trial in 10 patients with pulmonary hypertension associated with heart failure with preserved ejection fraction.
Disclosures: Dr. Farber and his coauthors reported grant support from Gilead Sciences, which makes ranolazine.
Esmolol stabilizes heart rate in septic shock
The short-acting, intravenous beta-blocker esmolol has been shown to reduce and stabilize heart rates without adverse effects in patients with severe septic shock, a new phase II study has found.
In an open-label study that randomized 154 patients with septic shock and a heart rate of 95 or higher to standard care or titrated esmolol, the beta-blocker was associated with successful reductions in heart rate to between 80 and 94 beats per minute over a 96-hour period: a median of –28 BPM for the esmolol group compared with –6 for controls (P less than .001).
For their research, Dr. Andrea Morelli of the University of Rome La Sapienza and colleagues recruited from the hospital’s intensive care unit patients with septic shock and a heart rate of 95 BPM or above (JAMA 2013;310:1683-91).
Patients with lower heart rates or with previous beta-blocker use were excluded. Subjects in both groups required norepinephrine to maintain a mean arterial pressure of 65 mm Hg or higher. The primary outcome measure was heart rate stabilization at between 80 and 94 BPM.
The esmolol group, which received a median continuous infusion of 100 mg/hr during the treatment period, also saw improved stroke work index and left ventricular stroke work, which investigators suspected was a result of improved diastolic filling. Esmolol treatment was associated with maintenance of mean arterial pressure and reduced need for norepinephrine. It was not associated with higher hepatic, renal, or myocardial injury compared with controls. Importantly, mortality at 28 days was considerably and significantly lower in the esmolol group than in controls: 49.4% vs. 80.5%. Each group comprised 77 patients.
In an editorial, Dr. Michael R. Pinsky of the department of critical care at the University of Pittsburgh called the findings "consistent with selective blockage of beta-adrenergic hyperactivity causing improved myocardial performance and decreased metabolic demand without compromising peripheral vascular function." Nonetheless, he cautioned clinicians against applying these results to all patients in septic shock (JAMA 2013;310:1677-8). "The reasons for this caution involve the limitations of this study and limitations in the current understanding of how beta-blocker therapy can cause such effects."
Dr. Morelli and colleagues acknowledged several limitations of their study. One was its single-center, open-label design. (As Dr. Pinsky noted in his editorial, a blinded study would be almost impossible to carry out because heart rate titration would be difficult to mask). The results should be replicated in a larger, multicenter trial, the researchers wrote. They noted that they had used "an arbitrary predefined heart rate threshold rather than an individualized approach titrated to specific myocardial characteristics or other biomarkers." Finally, the researchers allowed that the unexpectedly large mortality difference seen in the study could have been the result of confounding.
The study was funded by the University of Rome La Sapienza. Dr. Morelli disclosed honoraria from Baxter, the manufacturer of esmolol. A coauthor, Dr. Mervyn Singer, reported ties with Baxter. Dr. Pinsky did not report any disclosures relevant to his editorial.
The short-acting, intravenous beta-blocker esmolol has been shown to reduce and stabilize heart rates without adverse effects in patients with severe septic shock, a new phase II study has found.
In an open-label study that randomized 154 patients with septic shock and a heart rate of 95 or higher to standard care or titrated esmolol, the beta-blocker was associated with successful reductions in heart rate to between 80 and 94 beats per minute over a 96-hour period: a median of –28 BPM for the esmolol group compared with –6 for controls (P less than .001).
For their research, Dr. Andrea Morelli of the University of Rome La Sapienza and colleagues recruited from the hospital’s intensive care unit patients with septic shock and a heart rate of 95 BPM or above (JAMA 2013;310:1683-91).
Patients with lower heart rates or with previous beta-blocker use were excluded. Subjects in both groups required norepinephrine to maintain a mean arterial pressure of 65 mm Hg or higher. The primary outcome measure was heart rate stabilization at between 80 and 94 BPM.
The esmolol group, which received a median continuous infusion of 100 mg/hr during the treatment period, also saw improved stroke work index and left ventricular stroke work, which investigators suspected was a result of improved diastolic filling. Esmolol treatment was associated with maintenance of mean arterial pressure and reduced need for norepinephrine. It was not associated with higher hepatic, renal, or myocardial injury compared with controls. Importantly, mortality at 28 days was considerably and significantly lower in the esmolol group than in controls: 49.4% vs. 80.5%. Each group comprised 77 patients.
In an editorial, Dr. Michael R. Pinsky of the department of critical care at the University of Pittsburgh called the findings "consistent with selective blockage of beta-adrenergic hyperactivity causing improved myocardial performance and decreased metabolic demand without compromising peripheral vascular function." Nonetheless, he cautioned clinicians against applying these results to all patients in septic shock (JAMA 2013;310:1677-8). "The reasons for this caution involve the limitations of this study and limitations in the current understanding of how beta-blocker therapy can cause such effects."
Dr. Morelli and colleagues acknowledged several limitations of their study. One was its single-center, open-label design. (As Dr. Pinsky noted in his editorial, a blinded study would be almost impossible to carry out because heart rate titration would be difficult to mask). The results should be replicated in a larger, multicenter trial, the researchers wrote. They noted that they had used "an arbitrary predefined heart rate threshold rather than an individualized approach titrated to specific myocardial characteristics or other biomarkers." Finally, the researchers allowed that the unexpectedly large mortality difference seen in the study could have been the result of confounding.
The study was funded by the University of Rome La Sapienza. Dr. Morelli disclosed honoraria from Baxter, the manufacturer of esmolol. A coauthor, Dr. Mervyn Singer, reported ties with Baxter. Dr. Pinsky did not report any disclosures relevant to his editorial.
The short-acting, intravenous beta-blocker esmolol has been shown to reduce and stabilize heart rates without adverse effects in patients with severe septic shock, a new phase II study has found.
In an open-label study that randomized 154 patients with septic shock and a heart rate of 95 or higher to standard care or titrated esmolol, the beta-blocker was associated with successful reductions in heart rate to between 80 and 94 beats per minute over a 96-hour period: a median of –28 BPM for the esmolol group compared with –6 for controls (P less than .001).
For their research, Dr. Andrea Morelli of the University of Rome La Sapienza and colleagues recruited from the hospital’s intensive care unit patients with septic shock and a heart rate of 95 BPM or above (JAMA 2013;310:1683-91).
Patients with lower heart rates or with previous beta-blocker use were excluded. Subjects in both groups required norepinephrine to maintain a mean arterial pressure of 65 mm Hg or higher. The primary outcome measure was heart rate stabilization at between 80 and 94 BPM.
The esmolol group, which received a median continuous infusion of 100 mg/hr during the treatment period, also saw improved stroke work index and left ventricular stroke work, which investigators suspected was a result of improved diastolic filling. Esmolol treatment was associated with maintenance of mean arterial pressure and reduced need for norepinephrine. It was not associated with higher hepatic, renal, or myocardial injury compared with controls. Importantly, mortality at 28 days was considerably and significantly lower in the esmolol group than in controls: 49.4% vs. 80.5%. Each group comprised 77 patients.
In an editorial, Dr. Michael R. Pinsky of the department of critical care at the University of Pittsburgh called the findings "consistent with selective blockage of beta-adrenergic hyperactivity causing improved myocardial performance and decreased metabolic demand without compromising peripheral vascular function." Nonetheless, he cautioned clinicians against applying these results to all patients in septic shock (JAMA 2013;310:1677-8). "The reasons for this caution involve the limitations of this study and limitations in the current understanding of how beta-blocker therapy can cause such effects."
Dr. Morelli and colleagues acknowledged several limitations of their study. One was its single-center, open-label design. (As Dr. Pinsky noted in his editorial, a blinded study would be almost impossible to carry out because heart rate titration would be difficult to mask). The results should be replicated in a larger, multicenter trial, the researchers wrote. They noted that they had used "an arbitrary predefined heart rate threshold rather than an individualized approach titrated to specific myocardial characteristics or other biomarkers." Finally, the researchers allowed that the unexpectedly large mortality difference seen in the study could have been the result of confounding.
The study was funded by the University of Rome La Sapienza. Dr. Morelli disclosed honoraria from Baxter, the manufacturer of esmolol. A coauthor, Dr. Mervyn Singer, reported ties with Baxter. Dr. Pinsky did not report any disclosures relevant to his editorial.
FROM JAMA
Main finding: A short-acting beta-blocker, esmolol, is effective in reducing heart rates among patients with septic shock and tachycardia.
Data source: A single-center, open-label randomized trial (n = 154).
Disclosures: The study was funded by the University of Rome La Sapienza. Dr. Morelli disclosed honoraria from Baxter, the manufacturer of esmolol. A coauthor, Dr. Mervyn Singer, reported ties with Baxter. Dr. Pinsky did not report any disclosures relevant to his editorial.
On heart failure and beta-blocker dosages
Getting the right therapeutic dose of any drug is not always easy. Using antibiotics to treat infection or antihypertensive drugs to lower blood pressure can be measured easily by simple physiologic measurements.
The treatment of heart failure with beta-blockers or ACE inhibitors, however, has been largely defined by clinical trials, which by their nature use one dosage and usually provide the clinician with limited information about the range of the best and most effective dosages. The rigor of choosing the correct dosage in clinical trials is often limited to small, underpowered phase II studies carried out well before the major phase III trials, which are designed to support efficacy and safety, usually at that one dosage. And still, physicians usually pick the lowest dose, following Hippocrates’ dictum to "do no harm." This dilemma has particular importance in picking the best dose of a beta-blocker in heart failure.
A recent presentation at the annual congress of the European Society of Cardiology by Dr. L. Brent Mitchell ("Full-dose beta-blockers still show benefit," October 2013, p. 26) sheds some important light on the benefit of maximum dosing with beta-blockers in heart failure patients treated with cardiac resynchronization therapy (CRT) or implantable cardiac defibrillators (ICDs) in whom bradycardia escape pacing was present.
Although all patients received standard drug therapy, patients receiving less than 50% of the full recommended dose of beta-blocker had a worse outcome in regard to mortality and rehospitalization when compared with patients receiving the full recommended dose, regardless of the beta-blocker used. Roughly one-half of these heart failure ICD/CRT patients were receiving less than half of the recommended dose for heart failure therapy. Older patients and those with more advance heart failure tended to receive the lower dose. In this patient population with pacemaker-controlled low heart rate, the issue of beta-blocker–induced bradycardia is no longer an issue: the higher the better.
In patients with atrial-controlled heart rates with sinus rhythm or atrial fibrillation, however, the induction of bradycardia has been an issue as physicians up-titrate dosages. The effect on morbidity and mortality of varying doses of metoprolol succinate (Toprol) was examined in the MERIT-HF trial (J. Am. Coll. Cardiol. 2002;40:491-8), in which physicians were encouraged to up-titrate to the highest dose. The limitation of up-titration was bradycardia. The high-dose (greater than 100 mg/day) and low-dose (100 mg/day or less) patients received 192 mg and 76 mg/day, respectively. Despite the different maximal doses, the final heart rate achieved with the up-titration was 68 beats/min. Patients receiving the high dose and low dose achieved the same relative benefit of therapy. The low-dose patient group was older and had a higher New York Heart Association functional class.
These observations suggest that there was a significant variability in the patient’s sensitivity to beta-blocker therapy, but the achievement of a low heart rate, regardless of dose, was effective in achieving the best therapeutic benefit. In a small dose-ranging study, patients were randomized to receive 50 or 200 mg/day of Toprol. The patients receiving 200 mg demonstrated an increase in ejection fraction and a decrease in end systolic volume, compared with the 50 mg–dose patients, who failed to evidence any hemodynamic improvement (Circulation 2007;116:49-56).
These observations emphasize the uncertainties of drug dosing in heart failure with our standard therapy. The benefit of high doses of beta-blockers in the ICD/CRT trial in patients whose heart rate was controlled with bradycardia pacing provides important support for the use of high doses in these individuals. In patients whose heart rate was controlled by atrial rhythms in the MERIT-HF trial, heart rate became the major limitation of drug therapy. In these patients, up-titration to maximal heart rate expressed the presence of a variable sensitivity to beta-blockade. The achievement of a slow heart rate, regardless of dose, appeared to achieve a similar benefit on heart failure outcomes.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies, and was the co-principal investigator of the MERIT-HF trial.
Getting the right therapeutic dose of any drug is not always easy. Using antibiotics to treat infection or antihypertensive drugs to lower blood pressure can be measured easily by simple physiologic measurements.
The treatment of heart failure with beta-blockers or ACE inhibitors, however, has been largely defined by clinical trials, which by their nature use one dosage and usually provide the clinician with limited information about the range of the best and most effective dosages. The rigor of choosing the correct dosage in clinical trials is often limited to small, underpowered phase II studies carried out well before the major phase III trials, which are designed to support efficacy and safety, usually at that one dosage. And still, physicians usually pick the lowest dose, following Hippocrates’ dictum to "do no harm." This dilemma has particular importance in picking the best dose of a beta-blocker in heart failure.
A recent presentation at the annual congress of the European Society of Cardiology by Dr. L. Brent Mitchell ("Full-dose beta-blockers still show benefit," October 2013, p. 26) sheds some important light on the benefit of maximum dosing with beta-blockers in heart failure patients treated with cardiac resynchronization therapy (CRT) or implantable cardiac defibrillators (ICDs) in whom bradycardia escape pacing was present.
Although all patients received standard drug therapy, patients receiving less than 50% of the full recommended dose of beta-blocker had a worse outcome in regard to mortality and rehospitalization when compared with patients receiving the full recommended dose, regardless of the beta-blocker used. Roughly one-half of these heart failure ICD/CRT patients were receiving less than half of the recommended dose for heart failure therapy. Older patients and those with more advance heart failure tended to receive the lower dose. In this patient population with pacemaker-controlled low heart rate, the issue of beta-blocker–induced bradycardia is no longer an issue: the higher the better.
In patients with atrial-controlled heart rates with sinus rhythm or atrial fibrillation, however, the induction of bradycardia has been an issue as physicians up-titrate dosages. The effect on morbidity and mortality of varying doses of metoprolol succinate (Toprol) was examined in the MERIT-HF trial (J. Am. Coll. Cardiol. 2002;40:491-8), in which physicians were encouraged to up-titrate to the highest dose. The limitation of up-titration was bradycardia. The high-dose (greater than 100 mg/day) and low-dose (100 mg/day or less) patients received 192 mg and 76 mg/day, respectively. Despite the different maximal doses, the final heart rate achieved with the up-titration was 68 beats/min. Patients receiving the high dose and low dose achieved the same relative benefit of therapy. The low-dose patient group was older and had a higher New York Heart Association functional class.
These observations suggest that there was a significant variability in the patient’s sensitivity to beta-blocker therapy, but the achievement of a low heart rate, regardless of dose, was effective in achieving the best therapeutic benefit. In a small dose-ranging study, patients were randomized to receive 50 or 200 mg/day of Toprol. The patients receiving 200 mg demonstrated an increase in ejection fraction and a decrease in end systolic volume, compared with the 50 mg–dose patients, who failed to evidence any hemodynamic improvement (Circulation 2007;116:49-56).
These observations emphasize the uncertainties of drug dosing in heart failure with our standard therapy. The benefit of high doses of beta-blockers in the ICD/CRT trial in patients whose heart rate was controlled with bradycardia pacing provides important support for the use of high doses in these individuals. In patients whose heart rate was controlled by atrial rhythms in the MERIT-HF trial, heart rate became the major limitation of drug therapy. In these patients, up-titration to maximal heart rate expressed the presence of a variable sensitivity to beta-blockade. The achievement of a slow heart rate, regardless of dose, appeared to achieve a similar benefit on heart failure outcomes.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies, and was the co-principal investigator of the MERIT-HF trial.
Getting the right therapeutic dose of any drug is not always easy. Using antibiotics to treat infection or antihypertensive drugs to lower blood pressure can be measured easily by simple physiologic measurements.
The treatment of heart failure with beta-blockers or ACE inhibitors, however, has been largely defined by clinical trials, which by their nature use one dosage and usually provide the clinician with limited information about the range of the best and most effective dosages. The rigor of choosing the correct dosage in clinical trials is often limited to small, underpowered phase II studies carried out well before the major phase III trials, which are designed to support efficacy and safety, usually at that one dosage. And still, physicians usually pick the lowest dose, following Hippocrates’ dictum to "do no harm." This dilemma has particular importance in picking the best dose of a beta-blocker in heart failure.
A recent presentation at the annual congress of the European Society of Cardiology by Dr. L. Brent Mitchell ("Full-dose beta-blockers still show benefit," October 2013, p. 26) sheds some important light on the benefit of maximum dosing with beta-blockers in heart failure patients treated with cardiac resynchronization therapy (CRT) or implantable cardiac defibrillators (ICDs) in whom bradycardia escape pacing was present.
Although all patients received standard drug therapy, patients receiving less than 50% of the full recommended dose of beta-blocker had a worse outcome in regard to mortality and rehospitalization when compared with patients receiving the full recommended dose, regardless of the beta-blocker used. Roughly one-half of these heart failure ICD/CRT patients were receiving less than half of the recommended dose for heart failure therapy. Older patients and those with more advance heart failure tended to receive the lower dose. In this patient population with pacemaker-controlled low heart rate, the issue of beta-blocker–induced bradycardia is no longer an issue: the higher the better.
In patients with atrial-controlled heart rates with sinus rhythm or atrial fibrillation, however, the induction of bradycardia has been an issue as physicians up-titrate dosages. The effect on morbidity and mortality of varying doses of metoprolol succinate (Toprol) was examined in the MERIT-HF trial (J. Am. Coll. Cardiol. 2002;40:491-8), in which physicians were encouraged to up-titrate to the highest dose. The limitation of up-titration was bradycardia. The high-dose (greater than 100 mg/day) and low-dose (100 mg/day or less) patients received 192 mg and 76 mg/day, respectively. Despite the different maximal doses, the final heart rate achieved with the up-titration was 68 beats/min. Patients receiving the high dose and low dose achieved the same relative benefit of therapy. The low-dose patient group was older and had a higher New York Heart Association functional class.
These observations suggest that there was a significant variability in the patient’s sensitivity to beta-blocker therapy, but the achievement of a low heart rate, regardless of dose, was effective in achieving the best therapeutic benefit. In a small dose-ranging study, patients were randomized to receive 50 or 200 mg/day of Toprol. The patients receiving 200 mg demonstrated an increase in ejection fraction and a decrease in end systolic volume, compared with the 50 mg–dose patients, who failed to evidence any hemodynamic improvement (Circulation 2007;116:49-56).
These observations emphasize the uncertainties of drug dosing in heart failure with our standard therapy. The benefit of high doses of beta-blockers in the ICD/CRT trial in patients whose heart rate was controlled with bradycardia pacing provides important support for the use of high doses in these individuals. In patients whose heart rate was controlled by atrial rhythms in the MERIT-HF trial, heart rate became the major limitation of drug therapy. In these patients, up-titration to maximal heart rate expressed the presence of a variable sensitivity to beta-blockade. The achievement of a slow heart rate, regardless of dose, appeared to achieve a similar benefit on heart failure outcomes.
Dr. Goldstein, medical editor of Cardiology News, is professor of medicine at Wayne State University and division head emeritus of cardiovascular medicine at Henry Ford Hospital, both in Detroit. He is on data safety monitoring committees for the National Institutes of Health and several pharmaceutical companies, and was the co-principal investigator of the MERIT-HF trial.
Life-saving therapies could eliminate wait-list disparities
ORLANDO – More women than men died during the first year of being on the heart transplant waiting list, and the disparity may be influenced by the difference in the use of life-saving therapies, according to a 12-year analysis of a national database.
After adjusting for several variables, researchers found that female gender was associated with a 10% increased risk of being removed from the waiting list because the women died or were deemed too sick during the first year. When the researchers added implantable cardioverter defibrillator use to the analysis, that risk was attenuated to 8%, although still significant, but adding ICDs and left ventricular assist devices (LVADs) eliminated the risk associated with the female gender, said Dr. Alanna Morris of Emory University, Atlanta.
The study, which looked at the Organ Procurement and Transplantation Network (OPTN) database, showed that women on the waiting list were significantly less likely to have an ICD (55% vs. 64%; P less than .001), or a ventricular assist device (24% vs. 30%; P less than .001) than were men, said Dr. Morris, who presented her unpublished abstract at the annual meeting of the Heart Failure Society of America.
The finding is in line with studies that have shown significantly lower rates of ICD implantation among women with end-stage heart failure, compared with men. Research has also shown that women are less likely to be referred for LVAD implantation, even though no survival difference between genders while on LVAD support has been observed, the authors noted.
Meanwhile, the proportion of women on the wait list has increased by more than 4% in the past decade, but studies on women’s survival while on wait lists have shown conflicting results, the researchers added.
They identified nearly 27,000 adult patients (23% were women) in the OPTN database between January 2000 and September 2012, who were listed for their first heart transplant.
There were several statistically significant differences between genders at baseline, aside from ICD and LVAD use. Female heart transplant candidates were younger (52 vs. 56 years), were less likely to have diabetes (21% vs. 27%), were less likely to have a normal glomerular filtration rate (49% vs. 53%), had a lower pulmonary capillary wedge pressure (20 vs. 21 mm Hg), and had fewer median days (67 vs. 84) on a wait list (P less than .001 for all).
The 1-year unadjusted survival rate in women on the waiting list was significantly lower, at 70%, than in men, at 73% (P = .006).
After adjustment for age, race, blood type, and support with extracorporeal membrane oxygenation or intra-aortic balloon pump, female gender was still associated with a higher risk of the primary end point, which was removal from the wait list due to death or being deemed too sick to transplant at 1 year (hazard ratio 1.10; P = .026), the authors reported.
But, after adjustment for ICD and LVAD use, the gender gap was eliminated (HR 1.06; P = .2).
Dr. Morris said that a more aggressive use of these life-saving therapies can eliminate wait-list disparities. The findings also point to the importance of educating community physicians and heart failure patients about the current standards of care, she said.
Changes in the allocation algorithm and improvements in LVAD technology have led to advancements in management of patients on heart transplant waiting lists, the authors said. Between 2001 and 2011, wait list mortality declined from 17 deaths per 100 wait-list years, to 12 deaths. During the same period, wait-list death among patients with an LVAD declined from 102 deaths per 100 wait-list years to 13.
Dr. Morris had no financial relationships to disclose.
On Twitter @NaseemSMiller
ORLANDO – More women than men died during the first year of being on the heart transplant waiting list, and the disparity may be influenced by the difference in the use of life-saving therapies, according to a 12-year analysis of a national database.
After adjusting for several variables, researchers found that female gender was associated with a 10% increased risk of being removed from the waiting list because the women died or were deemed too sick during the first year. When the researchers added implantable cardioverter defibrillator use to the analysis, that risk was attenuated to 8%, although still significant, but adding ICDs and left ventricular assist devices (LVADs) eliminated the risk associated with the female gender, said Dr. Alanna Morris of Emory University, Atlanta.
The study, which looked at the Organ Procurement and Transplantation Network (OPTN) database, showed that women on the waiting list were significantly less likely to have an ICD (55% vs. 64%; P less than .001), or a ventricular assist device (24% vs. 30%; P less than .001) than were men, said Dr. Morris, who presented her unpublished abstract at the annual meeting of the Heart Failure Society of America.
The finding is in line with studies that have shown significantly lower rates of ICD implantation among women with end-stage heart failure, compared with men. Research has also shown that women are less likely to be referred for LVAD implantation, even though no survival difference between genders while on LVAD support has been observed, the authors noted.
Meanwhile, the proportion of women on the wait list has increased by more than 4% in the past decade, but studies on women’s survival while on wait lists have shown conflicting results, the researchers added.
They identified nearly 27,000 adult patients (23% were women) in the OPTN database between January 2000 and September 2012, who were listed for their first heart transplant.
There were several statistically significant differences between genders at baseline, aside from ICD and LVAD use. Female heart transplant candidates were younger (52 vs. 56 years), were less likely to have diabetes (21% vs. 27%), were less likely to have a normal glomerular filtration rate (49% vs. 53%), had a lower pulmonary capillary wedge pressure (20 vs. 21 mm Hg), and had fewer median days (67 vs. 84) on a wait list (P less than .001 for all).
The 1-year unadjusted survival rate in women on the waiting list was significantly lower, at 70%, than in men, at 73% (P = .006).
After adjustment for age, race, blood type, and support with extracorporeal membrane oxygenation or intra-aortic balloon pump, female gender was still associated with a higher risk of the primary end point, which was removal from the wait list due to death or being deemed too sick to transplant at 1 year (hazard ratio 1.10; P = .026), the authors reported.
But, after adjustment for ICD and LVAD use, the gender gap was eliminated (HR 1.06; P = .2).
Dr. Morris said that a more aggressive use of these life-saving therapies can eliminate wait-list disparities. The findings also point to the importance of educating community physicians and heart failure patients about the current standards of care, she said.
Changes in the allocation algorithm and improvements in LVAD technology have led to advancements in management of patients on heart transplant waiting lists, the authors said. Between 2001 and 2011, wait list mortality declined from 17 deaths per 100 wait-list years, to 12 deaths. During the same period, wait-list death among patients with an LVAD declined from 102 deaths per 100 wait-list years to 13.
Dr. Morris had no financial relationships to disclose.
On Twitter @NaseemSMiller
ORLANDO – More women than men died during the first year of being on the heart transplant waiting list, and the disparity may be influenced by the difference in the use of life-saving therapies, according to a 12-year analysis of a national database.
After adjusting for several variables, researchers found that female gender was associated with a 10% increased risk of being removed from the waiting list because the women died or were deemed too sick during the first year. When the researchers added implantable cardioverter defibrillator use to the analysis, that risk was attenuated to 8%, although still significant, but adding ICDs and left ventricular assist devices (LVADs) eliminated the risk associated with the female gender, said Dr. Alanna Morris of Emory University, Atlanta.
The study, which looked at the Organ Procurement and Transplantation Network (OPTN) database, showed that women on the waiting list were significantly less likely to have an ICD (55% vs. 64%; P less than .001), or a ventricular assist device (24% vs. 30%; P less than .001) than were men, said Dr. Morris, who presented her unpublished abstract at the annual meeting of the Heart Failure Society of America.
The finding is in line with studies that have shown significantly lower rates of ICD implantation among women with end-stage heart failure, compared with men. Research has also shown that women are less likely to be referred for LVAD implantation, even though no survival difference between genders while on LVAD support has been observed, the authors noted.
Meanwhile, the proportion of women on the wait list has increased by more than 4% in the past decade, but studies on women’s survival while on wait lists have shown conflicting results, the researchers added.
They identified nearly 27,000 adult patients (23% were women) in the OPTN database between January 2000 and September 2012, who were listed for their first heart transplant.
There were several statistically significant differences between genders at baseline, aside from ICD and LVAD use. Female heart transplant candidates were younger (52 vs. 56 years), were less likely to have diabetes (21% vs. 27%), were less likely to have a normal glomerular filtration rate (49% vs. 53%), had a lower pulmonary capillary wedge pressure (20 vs. 21 mm Hg), and had fewer median days (67 vs. 84) on a wait list (P less than .001 for all).
The 1-year unadjusted survival rate in women on the waiting list was significantly lower, at 70%, than in men, at 73% (P = .006).
After adjustment for age, race, blood type, and support with extracorporeal membrane oxygenation or intra-aortic balloon pump, female gender was still associated with a higher risk of the primary end point, which was removal from the wait list due to death or being deemed too sick to transplant at 1 year (hazard ratio 1.10; P = .026), the authors reported.
But, after adjustment for ICD and LVAD use, the gender gap was eliminated (HR 1.06; P = .2).
Dr. Morris said that a more aggressive use of these life-saving therapies can eliminate wait-list disparities. The findings also point to the importance of educating community physicians and heart failure patients about the current standards of care, she said.
Changes in the allocation algorithm and improvements in LVAD technology have led to advancements in management of patients on heart transplant waiting lists, the authors said. Between 2001 and 2011, wait list mortality declined from 17 deaths per 100 wait-list years, to 12 deaths. During the same period, wait-list death among patients with an LVAD declined from 102 deaths per 100 wait-list years to 13.
Dr. Morris had no financial relationships to disclose.
On Twitter @NaseemSMiller
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Major finding: Female gender was associated with a 10% increased risk of being removed from the waiting list due to death or being too sick during the first year. When ICD was added to the analysis, risk was attenuated to 8%, but adding ICDs and LVADs eliminated the risk associated with the female gender.
Data source: Analysis of Organ Procurement and Transplantation Network database during 2000-2012.
Disclosures: Dr. Morris had no financial relationships to disclose.
Remote monitoring reduced death in advanced heart failure
AMSTERDAM – An implant-based remote monitoring system in patients with advanced heart failure significantly reduced the worsening of their condition, overnight hospitalization, and total mortality, according to a trial conducted by the device-maker.
The IN-TIME study, a multicenter, prospective, randomized, controlled trial, showed that the mortality at 1 year follow-up was 3.4% in the home monitoring group, compared with 8.7% in patients with standard care. This is the first time a home monitoring trial has shown a significant drop in death rate.
"IN-TIME is really a timely study," said Dr. Angelo Auricchio of Lugano, Switzerland, because the study coincides with the most recent ESC guidelines for CRT and pacemakers, which for the very first time contain recommendation for remote monitoring. Dr. Auricchio was one of the guideline’s authors.
Rehospitalization or death resulting from worsening heart failure are often preceded by clinical events or specific trends in clinical parameters. Use of home monitoring can detect some of these events and trends early, "offering the possibility to intervene and prevent hospitalization for worsening heart failure," said Dr. Gerhard Hindricks, the lead investigator of the trial, who presented the results at the annual congress of the European Society of Cardiology.
Findings from other trials, including TRUST and REFORM have hinted at the benefits of remote monitoring, but they didn’t show the significant results seen with IN-TIME.
Patients received Biotronik’s ICD or CRT-D equipped with home monitoring software. The device detects biological signals, such as heart rate, occurrence of arrhythmias, onset of arrhythmia or shocks, and transmits the signals to a small external patient communicator called Cardio Messenger, which in turn sends the signals to a centralized monitoring unit in Heart Center Leipzig, Germany.
"Trained nursing staff took the incoming data, and then activated a chain of action according to prespecified workflows,\" said Dr. Hindricks of University of Leipzig. In some cases, if the physician was not available, the providers would directly contact the patient to advise a follow-up with a general practitioner or appropriate clinic. The physician would also enter the information about the measure taken into the centralized monitoring system.
Researchers randomized 664 patients – 1:1, with no crossover – to either home monitoring (333) or standard of care (331). All patients were receiving optimal medical therapy.
The only significant difference in the use of ACE inhibitors between home monitoring and control groups (92% v. 86%, respectively), said Dr. Hindricks.
Patients were on average 65 years old and the majority was male. On average, 60% had an implanted CRT-D, and 40%, an ICD. Follow-up was 1 year.
The primary endpoint was the modified Packer Score, and 19% of the patients in the home monitoring group suffered from a worsening of their condition, compared with close to 28% in the group who were receiving the current standard of care only (P less than .05).
The secondary endpoint of all-cause mortality, also showed highly significant reduction in the home monitoring group (10 deaths), compared with the control arm (27 deaths) (P = .004). And, while 80% of all deaths were due to cardiovascular causes, there was a significant reduction in the cardiovascular mortality among the home monitoring group (8 deaths vs. 21 deaths in control; P = .012).
The home monitoring transmission rate was 85%, and there were 1,311 home monitoring observations, 66% of which were related to home monitoring issues; for instance, the patient’s device hadn’t transmitted data for 3 consecutive days, triggering an alert. The rest resulted from clinical events, with atrial fibrillation events being the most common (8.5%).
A total of 696 of patient contacts were made as a result of home monitoring alerts. Preliminary analysis of the data showed that 13% were from drug incompliance and 16% triggered additional visit to physician.
Dr. Hindricks said that one of the keys to the success of the program was minimal patient involvement.
Dr. Auricchio said that IN-TIME was a very important study "but we should have a cautionary note about the fact that the technology used is quite proprietary to one company, so the question is if it applies to other providers." He added that the study was done in a very defined patient population, and it’s yet to be seen whether its findings apply to other heart failure patients.
Dr. Hindricks has received honoraria for lectures and has been an adviser/consultant for Biosense, Biotronik, Stereotaxis, and St. Jude Medical. He has also been an adviser/consultant for CyberHeart. Dr. Auricchio has received payments from Biotronik, St. Jude Medical, Medtronic, Abbott, Philips, and several other companies.
On Twitter @NaseemSMiller
AMSTERDAM – An implant-based remote monitoring system in patients with advanced heart failure significantly reduced the worsening of their condition, overnight hospitalization, and total mortality, according to a trial conducted by the device-maker.
The IN-TIME study, a multicenter, prospective, randomized, controlled trial, showed that the mortality at 1 year follow-up was 3.4% in the home monitoring group, compared with 8.7% in patients with standard care. This is the first time a home monitoring trial has shown a significant drop in death rate.
"IN-TIME is really a timely study," said Dr. Angelo Auricchio of Lugano, Switzerland, because the study coincides with the most recent ESC guidelines for CRT and pacemakers, which for the very first time contain recommendation for remote monitoring. Dr. Auricchio was one of the guideline’s authors.
Rehospitalization or death resulting from worsening heart failure are often preceded by clinical events or specific trends in clinical parameters. Use of home monitoring can detect some of these events and trends early, "offering the possibility to intervene and prevent hospitalization for worsening heart failure," said Dr. Gerhard Hindricks, the lead investigator of the trial, who presented the results at the annual congress of the European Society of Cardiology.
Findings from other trials, including TRUST and REFORM have hinted at the benefits of remote monitoring, but they didn’t show the significant results seen with IN-TIME.
Patients received Biotronik’s ICD or CRT-D equipped with home monitoring software. The device detects biological signals, such as heart rate, occurrence of arrhythmias, onset of arrhythmia or shocks, and transmits the signals to a small external patient communicator called Cardio Messenger, which in turn sends the signals to a centralized monitoring unit in Heart Center Leipzig, Germany.
"Trained nursing staff took the incoming data, and then activated a chain of action according to prespecified workflows,\" said Dr. Hindricks of University of Leipzig. In some cases, if the physician was not available, the providers would directly contact the patient to advise a follow-up with a general practitioner or appropriate clinic. The physician would also enter the information about the measure taken into the centralized monitoring system.
Researchers randomized 664 patients – 1:1, with no crossover – to either home monitoring (333) or standard of care (331). All patients were receiving optimal medical therapy.
The only significant difference in the use of ACE inhibitors between home monitoring and control groups (92% v. 86%, respectively), said Dr. Hindricks.
Patients were on average 65 years old and the majority was male. On average, 60% had an implanted CRT-D, and 40%, an ICD. Follow-up was 1 year.
The primary endpoint was the modified Packer Score, and 19% of the patients in the home monitoring group suffered from a worsening of their condition, compared with close to 28% in the group who were receiving the current standard of care only (P less than .05).
The secondary endpoint of all-cause mortality, also showed highly significant reduction in the home monitoring group (10 deaths), compared with the control arm (27 deaths) (P = .004). And, while 80% of all deaths were due to cardiovascular causes, there was a significant reduction in the cardiovascular mortality among the home monitoring group (8 deaths vs. 21 deaths in control; P = .012).
The home monitoring transmission rate was 85%, and there were 1,311 home monitoring observations, 66% of which were related to home monitoring issues; for instance, the patient’s device hadn’t transmitted data for 3 consecutive days, triggering an alert. The rest resulted from clinical events, with atrial fibrillation events being the most common (8.5%).
A total of 696 of patient contacts were made as a result of home monitoring alerts. Preliminary analysis of the data showed that 13% were from drug incompliance and 16% triggered additional visit to physician.
Dr. Hindricks said that one of the keys to the success of the program was minimal patient involvement.
Dr. Auricchio said that IN-TIME was a very important study "but we should have a cautionary note about the fact that the technology used is quite proprietary to one company, so the question is if it applies to other providers." He added that the study was done in a very defined patient population, and it’s yet to be seen whether its findings apply to other heart failure patients.
Dr. Hindricks has received honoraria for lectures and has been an adviser/consultant for Biosense, Biotronik, Stereotaxis, and St. Jude Medical. He has also been an adviser/consultant for CyberHeart. Dr. Auricchio has received payments from Biotronik, St. Jude Medical, Medtronic, Abbott, Philips, and several other companies.
On Twitter @NaseemSMiller
AMSTERDAM – An implant-based remote monitoring system in patients with advanced heart failure significantly reduced the worsening of their condition, overnight hospitalization, and total mortality, according to a trial conducted by the device-maker.
The IN-TIME study, a multicenter, prospective, randomized, controlled trial, showed that the mortality at 1 year follow-up was 3.4% in the home monitoring group, compared with 8.7% in patients with standard care. This is the first time a home monitoring trial has shown a significant drop in death rate.
"IN-TIME is really a timely study," said Dr. Angelo Auricchio of Lugano, Switzerland, because the study coincides with the most recent ESC guidelines for CRT and pacemakers, which for the very first time contain recommendation for remote monitoring. Dr. Auricchio was one of the guideline’s authors.
Rehospitalization or death resulting from worsening heart failure are often preceded by clinical events or specific trends in clinical parameters. Use of home monitoring can detect some of these events and trends early, "offering the possibility to intervene and prevent hospitalization for worsening heart failure," said Dr. Gerhard Hindricks, the lead investigator of the trial, who presented the results at the annual congress of the European Society of Cardiology.
Findings from other trials, including TRUST and REFORM have hinted at the benefits of remote monitoring, but they didn’t show the significant results seen with IN-TIME.
Patients received Biotronik’s ICD or CRT-D equipped with home monitoring software. The device detects biological signals, such as heart rate, occurrence of arrhythmias, onset of arrhythmia or shocks, and transmits the signals to a small external patient communicator called Cardio Messenger, which in turn sends the signals to a centralized monitoring unit in Heart Center Leipzig, Germany.
"Trained nursing staff took the incoming data, and then activated a chain of action according to prespecified workflows,\" said Dr. Hindricks of University of Leipzig. In some cases, if the physician was not available, the providers would directly contact the patient to advise a follow-up with a general practitioner or appropriate clinic. The physician would also enter the information about the measure taken into the centralized monitoring system.
Researchers randomized 664 patients – 1:1, with no crossover – to either home monitoring (333) or standard of care (331). All patients were receiving optimal medical therapy.
The only significant difference in the use of ACE inhibitors between home monitoring and control groups (92% v. 86%, respectively), said Dr. Hindricks.
Patients were on average 65 years old and the majority was male. On average, 60% had an implanted CRT-D, and 40%, an ICD. Follow-up was 1 year.
The primary endpoint was the modified Packer Score, and 19% of the patients in the home monitoring group suffered from a worsening of their condition, compared with close to 28% in the group who were receiving the current standard of care only (P less than .05).
The secondary endpoint of all-cause mortality, also showed highly significant reduction in the home monitoring group (10 deaths), compared with the control arm (27 deaths) (P = .004). And, while 80% of all deaths were due to cardiovascular causes, there was a significant reduction in the cardiovascular mortality among the home monitoring group (8 deaths vs. 21 deaths in control; P = .012).
The home monitoring transmission rate was 85%, and there were 1,311 home monitoring observations, 66% of which were related to home monitoring issues; for instance, the patient’s device hadn’t transmitted data for 3 consecutive days, triggering an alert. The rest resulted from clinical events, with atrial fibrillation events being the most common (8.5%).
A total of 696 of patient contacts were made as a result of home monitoring alerts. Preliminary analysis of the data showed that 13% were from drug incompliance and 16% triggered additional visit to physician.
Dr. Hindricks said that one of the keys to the success of the program was minimal patient involvement.
Dr. Auricchio said that IN-TIME was a very important study "but we should have a cautionary note about the fact that the technology used is quite proprietary to one company, so the question is if it applies to other providers." He added that the study was done in a very defined patient population, and it’s yet to be seen whether its findings apply to other heart failure patients.
Dr. Hindricks has received honoraria for lectures and has been an adviser/consultant for Biosense, Biotronik, Stereotaxis, and St. Jude Medical. He has also been an adviser/consultant for CyberHeart. Dr. Auricchio has received payments from Biotronik, St. Jude Medical, Medtronic, Abbott, Philips, and several other companies.
On Twitter @NaseemSMiller
AT THE ESC CONGRESS 2013
Major finding: At 1-year, the mortality of home monitoring group was 3.4%, compared with 8.7% in patients receiving standard care (P = .004).
Data source: A prospective randomized, controlled trial of nearly 700 patients.
Disclosures: Dr. Hindricks has received honoraria for lectures and has been an adviser/consultant for Biosense, Biotronik, Stereotaxis, and St. Jude Medical. He has also been an adviser/consultant for CyberHeart. Dr. Auricchio has received payments from Biotronik, St. Jude Medical, Medtronic, Abbott, Philips, and several other companies.