User login
AHRQ report examines interventions for lowering readmissions in heart failure patients
Home-visiting programs, multidisciplinary heart failure clinics, and structured telephone support interventions "should receive the greatest consideration by systems or providers seeking to implement transitional care interventions" for people hospitalized for heart failure, according to a report issued May 27 by the Agency for Healthcare Research and Quality Effective Health Care Program.
But for these interventions to be effective, "you need a very mature infrastructure – communication infrastructure, information technology infrastructure, and systems-level infrastructure," Dr. Hiren Shah, medical director of the cardiac telemetry unit at Northwestern Memorial Hospital, Chicago, said in an interview about the implications of the report. "A lot of people point to cost and the fact that these interventions are expensive. But even if you have the ability to invest in these interventions, do you have the infrastructure that’s necessary to implement them?"
Further, the report lacks details on the cost-effectiveness of these interventions, a vital bit of information for hospitals looking to potentially implement them, Dr. Shah said. "It’s very important that not only do we know what may potentially work as an intervention, but we need to know whether that intervention or those interventions are cost effective. Within the scientific literature, we have very little information on the cost effectiveness of interventions."
The report’s conclusions are based on a systematic review and meta-analysis of the efficacy, comparative effectiveness, and harms of transitional care interventions aimed at reducing hospital readmissions and mortality for adults hospitalized with heart failure. The researchers, led by Dr. Cynthia Feltner of Duke University Medical Center in Durham, N.C., drew from 53 published articles on 47 randomized, controlled trials. Most studies compared a transitional care intervention with usual care, though the report notes that usual care was not consistently or well described.
In general, the trials included patients around age 70 years with moderate to severe heart failure. Most patients were prescribed an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker; the percentage of patients prescribed a beta-blocker varied widely across the study populations. Trial settings included academic medical centers, Veteran Affairs hospitals, and community hospitals.
Few trials reported 30-day readmission rates, as most measured outcomes over 3-6 months. Home-visiting programs and multidisciplinary heart failure clinic interventions reduced all-cause readmissions and mortality; structured telephone support reduced heart failure–specific readmissions and mortality.
Successful programs and interventions included heart failure education that emphasized self-care and recognition of symptoms and weight monitoring; patient education about medications and adherence to regimens as well as evidence-based therapies before and after discharge; face-to-face contact with visiting or clinic personnel within a week following discharge; streamlined mechanisms for contacting care delivery personnel outside of scheduled visits; and mechanisms for postdischarge medication adjustment.
"Separating out individual components from the overall categories [or "bundles"] of interventions that showed efficacy was not possible," the report notes.
None of the investigators have affiliations or financial involvements that constitute a conflict of interest with the material presented in the report.
Home-visiting programs, multidisciplinary heart failure clinics, and structured telephone support interventions "should receive the greatest consideration by systems or providers seeking to implement transitional care interventions" for people hospitalized for heart failure, according to a report issued May 27 by the Agency for Healthcare Research and Quality Effective Health Care Program.
But for these interventions to be effective, "you need a very mature infrastructure – communication infrastructure, information technology infrastructure, and systems-level infrastructure," Dr. Hiren Shah, medical director of the cardiac telemetry unit at Northwestern Memorial Hospital, Chicago, said in an interview about the implications of the report. "A lot of people point to cost and the fact that these interventions are expensive. But even if you have the ability to invest in these interventions, do you have the infrastructure that’s necessary to implement them?"
Further, the report lacks details on the cost-effectiveness of these interventions, a vital bit of information for hospitals looking to potentially implement them, Dr. Shah said. "It’s very important that not only do we know what may potentially work as an intervention, but we need to know whether that intervention or those interventions are cost effective. Within the scientific literature, we have very little information on the cost effectiveness of interventions."
The report’s conclusions are based on a systematic review and meta-analysis of the efficacy, comparative effectiveness, and harms of transitional care interventions aimed at reducing hospital readmissions and mortality for adults hospitalized with heart failure. The researchers, led by Dr. Cynthia Feltner of Duke University Medical Center in Durham, N.C., drew from 53 published articles on 47 randomized, controlled trials. Most studies compared a transitional care intervention with usual care, though the report notes that usual care was not consistently or well described.
In general, the trials included patients around age 70 years with moderate to severe heart failure. Most patients were prescribed an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker; the percentage of patients prescribed a beta-blocker varied widely across the study populations. Trial settings included academic medical centers, Veteran Affairs hospitals, and community hospitals.
Few trials reported 30-day readmission rates, as most measured outcomes over 3-6 months. Home-visiting programs and multidisciplinary heart failure clinic interventions reduced all-cause readmissions and mortality; structured telephone support reduced heart failure–specific readmissions and mortality.
Successful programs and interventions included heart failure education that emphasized self-care and recognition of symptoms and weight monitoring; patient education about medications and adherence to regimens as well as evidence-based therapies before and after discharge; face-to-face contact with visiting or clinic personnel within a week following discharge; streamlined mechanisms for contacting care delivery personnel outside of scheduled visits; and mechanisms for postdischarge medication adjustment.
"Separating out individual components from the overall categories [or "bundles"] of interventions that showed efficacy was not possible," the report notes.
None of the investigators have affiliations or financial involvements that constitute a conflict of interest with the material presented in the report.
Home-visiting programs, multidisciplinary heart failure clinics, and structured telephone support interventions "should receive the greatest consideration by systems or providers seeking to implement transitional care interventions" for people hospitalized for heart failure, according to a report issued May 27 by the Agency for Healthcare Research and Quality Effective Health Care Program.
But for these interventions to be effective, "you need a very mature infrastructure – communication infrastructure, information technology infrastructure, and systems-level infrastructure," Dr. Hiren Shah, medical director of the cardiac telemetry unit at Northwestern Memorial Hospital, Chicago, said in an interview about the implications of the report. "A lot of people point to cost and the fact that these interventions are expensive. But even if you have the ability to invest in these interventions, do you have the infrastructure that’s necessary to implement them?"
Further, the report lacks details on the cost-effectiveness of these interventions, a vital bit of information for hospitals looking to potentially implement them, Dr. Shah said. "It’s very important that not only do we know what may potentially work as an intervention, but we need to know whether that intervention or those interventions are cost effective. Within the scientific literature, we have very little information on the cost effectiveness of interventions."
The report’s conclusions are based on a systematic review and meta-analysis of the efficacy, comparative effectiveness, and harms of transitional care interventions aimed at reducing hospital readmissions and mortality for adults hospitalized with heart failure. The researchers, led by Dr. Cynthia Feltner of Duke University Medical Center in Durham, N.C., drew from 53 published articles on 47 randomized, controlled trials. Most studies compared a transitional care intervention with usual care, though the report notes that usual care was not consistently or well described.
In general, the trials included patients around age 70 years with moderate to severe heart failure. Most patients were prescribed an angiotensin-converting enzyme inhibitor or an angiotensin receptor blocker; the percentage of patients prescribed a beta-blocker varied widely across the study populations. Trial settings included academic medical centers, Veteran Affairs hospitals, and community hospitals.
Few trials reported 30-day readmission rates, as most measured outcomes over 3-6 months. Home-visiting programs and multidisciplinary heart failure clinic interventions reduced all-cause readmissions and mortality; structured telephone support reduced heart failure–specific readmissions and mortality.
Successful programs and interventions included heart failure education that emphasized self-care and recognition of symptoms and weight monitoring; patient education about medications and adherence to regimens as well as evidence-based therapies before and after discharge; face-to-face contact with visiting or clinic personnel within a week following discharge; streamlined mechanisms for contacting care delivery personnel outside of scheduled visits; and mechanisms for postdischarge medication adjustment.
"Separating out individual components from the overall categories [or "bundles"] of interventions that showed efficacy was not possible," the report notes.
None of the investigators have affiliations or financial involvements that constitute a conflict of interest with the material presented in the report.
Fatty heart may bring on diabetes
SAN FRANCISCO – A high volume of pericardial adipose tissue was associated with the prevalence of diabetes, independent of overall obesity, results from a long-term, diverse cohort study showed.
"Obesity is associated with an increased risk for cardiovascular disease and type 2 diabetes, and specific fat deposits may increase the risk more than others," Amy C. Alman, Ph.D., said at the annual scientific sessions of the American Diabetes Association.
The findings come from an analysis of year-25 exam data among 3,079 participants in CARDIA (Coronary Artery Risk Development in Young Adults), a longitudinal cohort study of the development of cardiovascular risk and disease that began in 1985 and was conducted at centers in Alabama, Minnesota, Illinois, and California.
Visceral adiposity "is metabolically active and more strongly associated with cardiovascular risk than subcutaneous adiposity. Ectopic adipose depots are metabolically active fat found in and around tissues and organs throughout the body, including the liver, muscles, and around the heart," explained Dr. Alman of the department of epidemiology and biostatistics at the University of South Florida, Tampa.
Pericardial adipose tissue (PAT) is an ectopic fat depot composed of epicardial adipose tissue deep to the pericardium and surrounding the coronary arteries and paracardial tissue, which is located along the surface of the parietal pericardium. She and her associates tested whether PAT was positively associated with prevalent diabetes at the year-25 exam of the CARDIA study.
Examinations including volume of PAT measures from chest CT scans were performed during 2010-2011, and the researchers used multivariable logistic regression to examine the relation between quartiles of PAT and diabetes. There were four PAT quartiles: less than 33.5 cm3 (quartile 1/referent volume), between 33.5 and less than 48.7 cm3 (quartile 2), between 48.7 and less than 71.7 cm3 (quartile 3), and greater than 71.7 cm3 (quartile 4).
The mean age of the 3,079 study participants was 51 years, 44% were male, and their average body mass index was 31 kg/m2. Of these, 419 had prevalent diabetes. The prevalence of diabetes was highest in the fourth quartile of PAT volume (46% vs. 12% in the first quartile, 18% in the second, and 24% in the third), Dr. Alman reported.
In a logistic regression model of PAT volume on diabetes status by obesity, only PAT volume in the fourth quartile was significantly associated with diabetes status (odds ratio, 2.47), adjusted for field center, gender, age, race, systolic blood pressure, total cholesterol, triglycerides, and treatment with blood pressure– and cholesterol-lowering medications. A similar association was observed among nonobese patients (OR, 3.78).
"Deposition of a higher proportion of metabolically active ectopic fat is associated with diabetes in both obese and nonobese individuals," Dr. Alman concluded.
The analysis was "a very well characterized multicenter cohort study with a diverse racial cohort of middle-aged men and women," she added. Limitations of the study, she said, included its cross-sectional design, the fact that PAT was measured at a single point in time, and that it did not account for other ectopic fat depots such as liver fat. A longitudinal analysis of the participants is planned.
Dr. Alman had no relevant financial conflicts to disclose.
SAN FRANCISCO – A high volume of pericardial adipose tissue was associated with the prevalence of diabetes, independent of overall obesity, results from a long-term, diverse cohort study showed.
"Obesity is associated with an increased risk for cardiovascular disease and type 2 diabetes, and specific fat deposits may increase the risk more than others," Amy C. Alman, Ph.D., said at the annual scientific sessions of the American Diabetes Association.
The findings come from an analysis of year-25 exam data among 3,079 participants in CARDIA (Coronary Artery Risk Development in Young Adults), a longitudinal cohort study of the development of cardiovascular risk and disease that began in 1985 and was conducted at centers in Alabama, Minnesota, Illinois, and California.
Visceral adiposity "is metabolically active and more strongly associated with cardiovascular risk than subcutaneous adiposity. Ectopic adipose depots are metabolically active fat found in and around tissues and organs throughout the body, including the liver, muscles, and around the heart," explained Dr. Alman of the department of epidemiology and biostatistics at the University of South Florida, Tampa.
Pericardial adipose tissue (PAT) is an ectopic fat depot composed of epicardial adipose tissue deep to the pericardium and surrounding the coronary arteries and paracardial tissue, which is located along the surface of the parietal pericardium. She and her associates tested whether PAT was positively associated with prevalent diabetes at the year-25 exam of the CARDIA study.
Examinations including volume of PAT measures from chest CT scans were performed during 2010-2011, and the researchers used multivariable logistic regression to examine the relation between quartiles of PAT and diabetes. There were four PAT quartiles: less than 33.5 cm3 (quartile 1/referent volume), between 33.5 and less than 48.7 cm3 (quartile 2), between 48.7 and less than 71.7 cm3 (quartile 3), and greater than 71.7 cm3 (quartile 4).
The mean age of the 3,079 study participants was 51 years, 44% were male, and their average body mass index was 31 kg/m2. Of these, 419 had prevalent diabetes. The prevalence of diabetes was highest in the fourth quartile of PAT volume (46% vs. 12% in the first quartile, 18% in the second, and 24% in the third), Dr. Alman reported.
In a logistic regression model of PAT volume on diabetes status by obesity, only PAT volume in the fourth quartile was significantly associated with diabetes status (odds ratio, 2.47), adjusted for field center, gender, age, race, systolic blood pressure, total cholesterol, triglycerides, and treatment with blood pressure– and cholesterol-lowering medications. A similar association was observed among nonobese patients (OR, 3.78).
"Deposition of a higher proportion of metabolically active ectopic fat is associated with diabetes in both obese and nonobese individuals," Dr. Alman concluded.
The analysis was "a very well characterized multicenter cohort study with a diverse racial cohort of middle-aged men and women," she added. Limitations of the study, she said, included its cross-sectional design, the fact that PAT was measured at a single point in time, and that it did not account for other ectopic fat depots such as liver fat. A longitudinal analysis of the participants is planned.
Dr. Alman had no relevant financial conflicts to disclose.
SAN FRANCISCO – A high volume of pericardial adipose tissue was associated with the prevalence of diabetes, independent of overall obesity, results from a long-term, diverse cohort study showed.
"Obesity is associated with an increased risk for cardiovascular disease and type 2 diabetes, and specific fat deposits may increase the risk more than others," Amy C. Alman, Ph.D., said at the annual scientific sessions of the American Diabetes Association.
The findings come from an analysis of year-25 exam data among 3,079 participants in CARDIA (Coronary Artery Risk Development in Young Adults), a longitudinal cohort study of the development of cardiovascular risk and disease that began in 1985 and was conducted at centers in Alabama, Minnesota, Illinois, and California.
Visceral adiposity "is metabolically active and more strongly associated with cardiovascular risk than subcutaneous adiposity. Ectopic adipose depots are metabolically active fat found in and around tissues and organs throughout the body, including the liver, muscles, and around the heart," explained Dr. Alman of the department of epidemiology and biostatistics at the University of South Florida, Tampa.
Pericardial adipose tissue (PAT) is an ectopic fat depot composed of epicardial adipose tissue deep to the pericardium and surrounding the coronary arteries and paracardial tissue, which is located along the surface of the parietal pericardium. She and her associates tested whether PAT was positively associated with prevalent diabetes at the year-25 exam of the CARDIA study.
Examinations including volume of PAT measures from chest CT scans were performed during 2010-2011, and the researchers used multivariable logistic regression to examine the relation between quartiles of PAT and diabetes. There were four PAT quartiles: less than 33.5 cm3 (quartile 1/referent volume), between 33.5 and less than 48.7 cm3 (quartile 2), between 48.7 and less than 71.7 cm3 (quartile 3), and greater than 71.7 cm3 (quartile 4).
The mean age of the 3,079 study participants was 51 years, 44% were male, and their average body mass index was 31 kg/m2. Of these, 419 had prevalent diabetes. The prevalence of diabetes was highest in the fourth quartile of PAT volume (46% vs. 12% in the first quartile, 18% in the second, and 24% in the third), Dr. Alman reported.
In a logistic regression model of PAT volume on diabetes status by obesity, only PAT volume in the fourth quartile was significantly associated with diabetes status (odds ratio, 2.47), adjusted for field center, gender, age, race, systolic blood pressure, total cholesterol, triglycerides, and treatment with blood pressure– and cholesterol-lowering medications. A similar association was observed among nonobese patients (OR, 3.78).
"Deposition of a higher proportion of metabolically active ectopic fat is associated with diabetes in both obese and nonobese individuals," Dr. Alman concluded.
The analysis was "a very well characterized multicenter cohort study with a diverse racial cohort of middle-aged men and women," she added. Limitations of the study, she said, included its cross-sectional design, the fact that PAT was measured at a single point in time, and that it did not account for other ectopic fat depots such as liver fat. A longitudinal analysis of the participants is planned.
Dr. Alman had no relevant financial conflicts to disclose.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Key clinical point: The greater the volume of pericardial adipose tissue, the greater the risk of diabetes.
Major finding: In a logistic regression model of PAT volume on diabetes status by obesity, only PAT volume in the fourth quartile was significantly associated with diabetes status (OR, 2.47).
Data source: An analysis of data from 3,079 participants at the year-25 exam of the CARDIA study.
Disclosures: Dr. Alman said that she had no relevant financial conflicts.
STEMI: Prereperfusion IV metoprolol shows long-term benefits
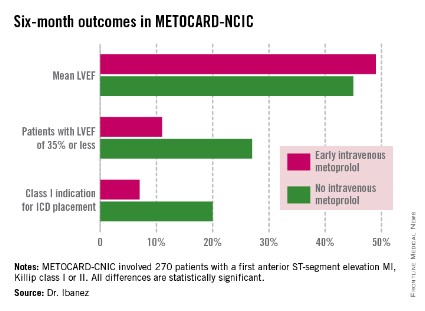
WASHINGTON – Early administration of intravenous metoprolol prior to primary percutaneous coronary intervention in patients with Killip class I or II anterior ST-elevation myocardial infarction reaped impressive long-term benefits in updated results from the Spanish METOCARD-CNIC trial.
At 6 months post infarct, the mean left ventricular ejection fraction (LVEF) was significantly higher in the early IV beta-blocker group than in controls. Also, the prevalence of a severely depressed LVEF of 35% or less was significantly lower, as was the proportion of patients having a class I indication for an implantable cardioverter-defibrillator, Dr. Borja Ibanez reported at the annual meeting of the American College of Cardiology.
METOCARD-CNIC
The primary study endpoint was infarct size as measured by magnetic resonance imaging 7 days post STEMI. As previously reported, infarct size was 20% smaller in the IV metoprolol recipients (Circulation 2013;128:1495-1503). This finding was encouraging, Dr. Ibanez observed, because infarct size is a major determinant of long-term morbidity and mortality. And while primary PCI for STEMI results in very low acute mortality, there is a high residual risk of subsequent heart failure and death. So the search is on for treatments that reduce infarct size. And prior to METOCARD-CNIC there had been no randomized controlled trials of early IV beta-blocker therapy during the primary PCI era.
At ACC 14, Dr. Ibanez presented prespecified secondary endpoints based upon outcomes at 6 months (see chart) and 2 years post STEMI.
At a median follow-up of 2 years, the composite endpoint comprised of death, reinfarction, hospital admission for heart failure, or malignant arrhythmia had occurred in 10.8% of the IV metoprolol group, compared with 18.3% of controls. This translated to an adjusted 45% relative risk reduction which didn’t quite reach statistical significance, but then again the trial wasn’t of sufficient size to be powered to evaluate clinical endpoints.
Of note, the heart failure hospital admission component of the composite endpoint occurred in 2.2% of the IV beta-blocker group, compared with 6.9% of controls, for a 68% relative risk reduction that was statistically significant. The curves began to split at 12 months and continued to diverge through 24 months, according to Dr. Ibanez.
Based upon the encouraging findings of METOCARD-CNIC, planning is underway for a large randomized trial powered to evaluate hard clinical endpoints. It will be called the MOVE ON! trial, the cardiologist added.
METOCARD-CNIC was funded by the Carlos III National Center for Cardiovascular Investigations and the Spanish Ministry of Health and Social Policy. Dr. Ibanez reported having no financial conflicts of interest.
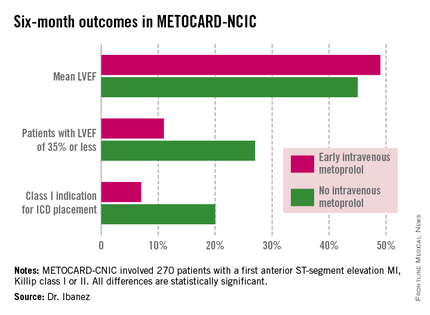
WASHINGTON – Early administration of intravenous metoprolol prior to primary percutaneous coronary intervention in patients with Killip class I or II anterior ST-elevation myocardial infarction reaped impressive long-term benefits in updated results from the Spanish METOCARD-CNIC trial.
At 6 months post infarct, the mean left ventricular ejection fraction (LVEF) was significantly higher in the early IV beta-blocker group than in controls. Also, the prevalence of a severely depressed LVEF of 35% or less was significantly lower, as was the proportion of patients having a class I indication for an implantable cardioverter-defibrillator, Dr. Borja Ibanez reported at the annual meeting of the American College of Cardiology.

METOCARD-CNIC
The primary study endpoint was infarct size as measured by magnetic resonance imaging 7 days post STEMI. As previously reported, infarct size was 20% smaller in the IV metoprolol recipients (Circulation 2013;128:1495-1503). This finding was encouraging, Dr. Ibanez observed, because infarct size is a major determinant of long-term morbidity and mortality. And while primary PCI for STEMI results in very low acute mortality, there is a high residual risk of subsequent heart failure and death. So the search is on for treatments that reduce infarct size. And prior to METOCARD-CNIC there had been no randomized controlled trials of early IV beta-blocker therapy during the primary PCI era.
At ACC 14, Dr. Ibanez presented prespecified secondary endpoints based upon outcomes at 6 months (see chart) and 2 years post STEMI.
At a median follow-up of 2 years, the composite endpoint comprised of death, reinfarction, hospital admission for heart failure, or malignant arrhythmia had occurred in 10.8% of the IV metoprolol group, compared with 18.3% of controls. This translated to an adjusted 45% relative risk reduction which didn’t quite reach statistical significance, but then again the trial wasn’t of sufficient size to be powered to evaluate clinical endpoints.
Of note, the heart failure hospital admission component of the composite endpoint occurred in 2.2% of the IV beta-blocker group, compared with 6.9% of controls, for a 68% relative risk reduction that was statistically significant. The curves began to split at 12 months and continued to diverge through 24 months, according to Dr. Ibanez.
Based upon the encouraging findings of METOCARD-CNIC, planning is underway for a large randomized trial powered to evaluate hard clinical endpoints. It will be called the MOVE ON! trial, the cardiologist added.
METOCARD-CNIC was funded by the Carlos III National Center for Cardiovascular Investigations and the Spanish Ministry of Health and Social Policy. Dr. Ibanez reported having no financial conflicts of interest.
WASHINGTON – Early administration of intravenous metoprolol prior to primary percutaneous coronary intervention in patients with Killip class I or II anterior ST-elevation myocardial infarction reaped impressive long-term benefits in updated results from the Spanish METOCARD-CNIC trial.
At 6 months post infarct, the mean left ventricular ejection fraction (LVEF) was significantly higher in the early IV beta-blocker group than in controls. Also, the prevalence of a severely depressed LVEF of 35% or less was significantly lower, as was the proportion of patients having a class I indication for an implantable cardioverter-defibrillator, Dr. Borja Ibanez reported at the annual meeting of the American College of Cardiology.

METOCARD-CNIC
The primary study endpoint was infarct size as measured by magnetic resonance imaging 7 days post STEMI. As previously reported, infarct size was 20% smaller in the IV metoprolol recipients (Circulation 2013;128:1495-1503). This finding was encouraging, Dr. Ibanez observed, because infarct size is a major determinant of long-term morbidity and mortality. And while primary PCI for STEMI results in very low acute mortality, there is a high residual risk of subsequent heart failure and death. So the search is on for treatments that reduce infarct size. And prior to METOCARD-CNIC there had been no randomized controlled trials of early IV beta-blocker therapy during the primary PCI era.
At ACC 14, Dr. Ibanez presented prespecified secondary endpoints based upon outcomes at 6 months (see chart) and 2 years post STEMI.
At a median follow-up of 2 years, the composite endpoint comprised of death, reinfarction, hospital admission for heart failure, or malignant arrhythmia had occurred in 10.8% of the IV metoprolol group, compared with 18.3% of controls. This translated to an adjusted 45% relative risk reduction which didn’t quite reach statistical significance, but then again the trial wasn’t of sufficient size to be powered to evaluate clinical endpoints.
Of note, the heart failure hospital admission component of the composite endpoint occurred in 2.2% of the IV beta-blocker group, compared with 6.9% of controls, for a 68% relative risk reduction that was statistically significant. The curves began to split at 12 months and continued to diverge through 24 months, according to Dr. Ibanez.
Based upon the encouraging findings of METOCARD-CNIC, planning is underway for a large randomized trial powered to evaluate hard clinical endpoints. It will be called the MOVE ON! trial, the cardiologist added.
METOCARD-CNIC was funded by the Carlos III National Center for Cardiovascular Investigations and the Spanish Ministry of Health and Social Policy. Dr. Ibanez reported having no financial conflicts of interest.
AT ACC 14
Key clinical point: Prereperfusion IV metoprolol may reduce heart failure readmissions.
Major finding: Patients with anterior ST-elevation MI who received intravenous metoprolol prior to primary PCI had a significantly greater left ventricular ejection fraction at 6 months follow-up than those who didn’t. They also were 68% less likely to be hospitalized for heart failure during 2 years of follow-up.
Data source: A six-center prospective trial in which 270 Spanish patients with a first anterior STEMI were randomized to have administration of IV metoprolol or not while being transported for primary PCI.
Disclosures: The METOCARD-CNIC trial was sponsored by the Spanish Ministry of Health and Social Policy and the Carlos III National Center for Cardiovascular Investigations. The presenter reported having no financial conflicts.
Survival higher with ICD in patients with LVEF of 30%-35%
Prophylactic implantable cardioverter-defibrillator use was associated with improved 3-year survival in Medicare beneficiaries with heart failure and a left ventricular ejection fraction of 30%-35% and in those with LVEF less than 30%, compared with no prophylactic ICD use in comparable patients in a retrospective cohort study.
All-cause mortality at 3 years in 408 patients with LVEF of 30% -35% who received a prophylactic ICD was 51.4%, compared with 55% in 408 matched controls without an ICD (hazard ratio, 0.83). The all-cause mortality rates at 3 years in 1,088 patients with LVEF of less than 30% was 45% in those with a prophylactic ICD vs. 57.6% in 1,088 matched controls without an ICD (HR, 0.72), Dr. Sana M. Al-Khatib of Duke University, Durham, N.C., and her colleagues reported in the June 4 issue of JAMA.
Study subjects were Medicare beneficiaries in the National Cardiovascular Data Registry ICD registry during 2006-2007, and similar patients in the Get With the Guidelines-Heart Failure database during 2005-2009 (JAMA 2014;311:2209-15).
Prior studies of prophylactic ICD use have included only a minority of patients with LVEF of 30%-35%; the current findings demonstrate a "clearly significant" benefit in this population and support guideline recommendations calling for prophylactic ICD use in eligible patients with an LVEF of 35% or less, the investigators concluded.
This study was funded by a National Heart, Lung, and Blood Institute grant. Dr. Al-Khatib reported having no disclosures. Detailed disclosures for several of the other study authors are available with the full text of the article at jama.com.
Prophylactic implantable cardioverter-defibrillator use was associated with improved 3-year survival in Medicare beneficiaries with heart failure and a left ventricular ejection fraction of 30%-35% and in those with LVEF less than 30%, compared with no prophylactic ICD use in comparable patients in a retrospective cohort study.
All-cause mortality at 3 years in 408 patients with LVEF of 30% -35% who received a prophylactic ICD was 51.4%, compared with 55% in 408 matched controls without an ICD (hazard ratio, 0.83). The all-cause mortality rates at 3 years in 1,088 patients with LVEF of less than 30% was 45% in those with a prophylactic ICD vs. 57.6% in 1,088 matched controls without an ICD (HR, 0.72), Dr. Sana M. Al-Khatib of Duke University, Durham, N.C., and her colleagues reported in the June 4 issue of JAMA.
Study subjects were Medicare beneficiaries in the National Cardiovascular Data Registry ICD registry during 2006-2007, and similar patients in the Get With the Guidelines-Heart Failure database during 2005-2009 (JAMA 2014;311:2209-15).
Prior studies of prophylactic ICD use have included only a minority of patients with LVEF of 30%-35%; the current findings demonstrate a "clearly significant" benefit in this population and support guideline recommendations calling for prophylactic ICD use in eligible patients with an LVEF of 35% or less, the investigators concluded.
This study was funded by a National Heart, Lung, and Blood Institute grant. Dr. Al-Khatib reported having no disclosures. Detailed disclosures for several of the other study authors are available with the full text of the article at jama.com.
Prophylactic implantable cardioverter-defibrillator use was associated with improved 3-year survival in Medicare beneficiaries with heart failure and a left ventricular ejection fraction of 30%-35% and in those with LVEF less than 30%, compared with no prophylactic ICD use in comparable patients in a retrospective cohort study.
All-cause mortality at 3 years in 408 patients with LVEF of 30% -35% who received a prophylactic ICD was 51.4%, compared with 55% in 408 matched controls without an ICD (hazard ratio, 0.83). The all-cause mortality rates at 3 years in 1,088 patients with LVEF of less than 30% was 45% in those with a prophylactic ICD vs. 57.6% in 1,088 matched controls without an ICD (HR, 0.72), Dr. Sana M. Al-Khatib of Duke University, Durham, N.C., and her colleagues reported in the June 4 issue of JAMA.
Study subjects were Medicare beneficiaries in the National Cardiovascular Data Registry ICD registry during 2006-2007, and similar patients in the Get With the Guidelines-Heart Failure database during 2005-2009 (JAMA 2014;311:2209-15).
Prior studies of prophylactic ICD use have included only a minority of patients with LVEF of 30%-35%; the current findings demonstrate a "clearly significant" benefit in this population and support guideline recommendations calling for prophylactic ICD use in eligible patients with an LVEF of 35% or less, the investigators concluded.
This study was funded by a National Heart, Lung, and Blood Institute grant. Dr. Al-Khatib reported having no disclosures. Detailed disclosures for several of the other study authors are available with the full text of the article at jama.com.
FROM JAMA
Key clinical point: Consider recommending an ICD for heart failure patients with an LVEF of 30%-35%, a population that is not well studied in clinical trials.
Major finding: All-cause mortality was 51.4% with and 55% without a prophylactic ICD (HR, 0.83).
Data source: A retrospective cohort study involving nearly 3,000 heart failure patients with an LVEF of 30%-35%.
Disclosures: This study was funded by a National Heart, Lung, and Blood Institute grant. Dr. Al-Khatib reported having no disclosures. Detailed disclosures for several of the other study authors are available with the full text of the article at jama.com.
Implantable device approved for remotely monitoring PA in heart failure patients
An implantable device that provides measurements of pulmonary arterial pressure in patients with class III heart failure has been approved by the Food and Drug Administration, based on a study that showed the use of the device to remotely monitor patients reduced heart failure hospitalization rates.
The CardioMEMS HF System "is the first permanently implantable wireless system intended to provide PA pressure measurements, including systolic, diastolic, and mean PA pressures," according to the FDA statement announcing the approval on May 28. This information is remotely reviewed by the patient’s physician, who "can make decisions regarding the status of the patient and, if necessary, initiate changes in medical therapy, with the goal of reducing hospitalization due to heart failure," the statement said.
It is specifically approved for patients with New York Heart Association (NYHA) class III heart failure (HF) who have been hospitalized for heart failure in the previous year.
The three components of the system are the battery-free sensor/monitor that is permanently implanted in the pulmonary artery, a transvenous catheter that deploys the sensor in the distal PA, and an electronic system that receives and processes the signals from the sensor/monitor and transfers the PA pressure measurements to a secure database, the statement said. Patients can be monitored from their home or another remote location.
Approval was based on a study of 550 patients with NYHA class III HF and a recent hospitalization for HF, who had the device implanted. Physicians had access to daily PA measurements only for the patients randomized to the treatment group, and adjusted HF medications based on the values provided. At 6 months, the HF hospitalization rate was significantly lower among those in the treatment group. The FDA statement noted that at 6 months, almost 99% of the patients who had the device implanted or in whom implantation was attempted had no complications related to the device or system, and all of the devices that were implanted were operating normally.
However, concerns about the study held up approval of the device for several years, and at a meeting in December 2011, the majority of the FDA’s Circulatory System Devices Panel agreed that the risks of the device outweighed the benefits. The manufacturer, CardioMEMS, provided follow-up data and further analyses of the data that were provided at another meeting of the panel, in October 2013. At that meeting, the majority of the panel agreed that the benefits of the device outweighed its risks for monitoring patients who met the criteria specified in the indication that has been approved, patients with NYHA class III heart failure who have been hospitalized for HF in the previous year.
In the May 28 statement, the FDA said that the company is required to conduct a postmarketing study to evaluate the performance of the device when used outside of a clinical trial. One concern of the panelists who supported approval at the 2013 meeting was that the benefit in terms of HF hospitalizations was not evident in women in the study, which they said could have been due to the low number of women enrolled in the trial, and they recommended that the device be studied in more women.
An implantable device that provides measurements of pulmonary arterial pressure in patients with class III heart failure has been approved by the Food and Drug Administration, based on a study that showed the use of the device to remotely monitor patients reduced heart failure hospitalization rates.
The CardioMEMS HF System "is the first permanently implantable wireless system intended to provide PA pressure measurements, including systolic, diastolic, and mean PA pressures," according to the FDA statement announcing the approval on May 28. This information is remotely reviewed by the patient’s physician, who "can make decisions regarding the status of the patient and, if necessary, initiate changes in medical therapy, with the goal of reducing hospitalization due to heart failure," the statement said.
It is specifically approved for patients with New York Heart Association (NYHA) class III heart failure (HF) who have been hospitalized for heart failure in the previous year.
The three components of the system are the battery-free sensor/monitor that is permanently implanted in the pulmonary artery, a transvenous catheter that deploys the sensor in the distal PA, and an electronic system that receives and processes the signals from the sensor/monitor and transfers the PA pressure measurements to a secure database, the statement said. Patients can be monitored from their home or another remote location.
Approval was based on a study of 550 patients with NYHA class III HF and a recent hospitalization for HF, who had the device implanted. Physicians had access to daily PA measurements only for the patients randomized to the treatment group, and adjusted HF medications based on the values provided. At 6 months, the HF hospitalization rate was significantly lower among those in the treatment group. The FDA statement noted that at 6 months, almost 99% of the patients who had the device implanted or in whom implantation was attempted had no complications related to the device or system, and all of the devices that were implanted were operating normally.
However, concerns about the study held up approval of the device for several years, and at a meeting in December 2011, the majority of the FDA’s Circulatory System Devices Panel agreed that the risks of the device outweighed the benefits. The manufacturer, CardioMEMS, provided follow-up data and further analyses of the data that were provided at another meeting of the panel, in October 2013. At that meeting, the majority of the panel agreed that the benefits of the device outweighed its risks for monitoring patients who met the criteria specified in the indication that has been approved, patients with NYHA class III heart failure who have been hospitalized for HF in the previous year.
In the May 28 statement, the FDA said that the company is required to conduct a postmarketing study to evaluate the performance of the device when used outside of a clinical trial. One concern of the panelists who supported approval at the 2013 meeting was that the benefit in terms of HF hospitalizations was not evident in women in the study, which they said could have been due to the low number of women enrolled in the trial, and they recommended that the device be studied in more women.
An implantable device that provides measurements of pulmonary arterial pressure in patients with class III heart failure has been approved by the Food and Drug Administration, based on a study that showed the use of the device to remotely monitor patients reduced heart failure hospitalization rates.
The CardioMEMS HF System "is the first permanently implantable wireless system intended to provide PA pressure measurements, including systolic, diastolic, and mean PA pressures," according to the FDA statement announcing the approval on May 28. This information is remotely reviewed by the patient’s physician, who "can make decisions regarding the status of the patient and, if necessary, initiate changes in medical therapy, with the goal of reducing hospitalization due to heart failure," the statement said.
It is specifically approved for patients with New York Heart Association (NYHA) class III heart failure (HF) who have been hospitalized for heart failure in the previous year.
The three components of the system are the battery-free sensor/monitor that is permanently implanted in the pulmonary artery, a transvenous catheter that deploys the sensor in the distal PA, and an electronic system that receives and processes the signals from the sensor/monitor and transfers the PA pressure measurements to a secure database, the statement said. Patients can be monitored from their home or another remote location.
Approval was based on a study of 550 patients with NYHA class III HF and a recent hospitalization for HF, who had the device implanted. Physicians had access to daily PA measurements only for the patients randomized to the treatment group, and adjusted HF medications based on the values provided. At 6 months, the HF hospitalization rate was significantly lower among those in the treatment group. The FDA statement noted that at 6 months, almost 99% of the patients who had the device implanted or in whom implantation was attempted had no complications related to the device or system, and all of the devices that were implanted were operating normally.
However, concerns about the study held up approval of the device for several years, and at a meeting in December 2011, the majority of the FDA’s Circulatory System Devices Panel agreed that the risks of the device outweighed the benefits. The manufacturer, CardioMEMS, provided follow-up data and further analyses of the data that were provided at another meeting of the panel, in October 2013. At that meeting, the majority of the panel agreed that the benefits of the device outweighed its risks for monitoring patients who met the criteria specified in the indication that has been approved, patients with NYHA class III heart failure who have been hospitalized for HF in the previous year.
In the May 28 statement, the FDA said that the company is required to conduct a postmarketing study to evaluate the performance of the device when used outside of a clinical trial. One concern of the panelists who supported approval at the 2013 meeting was that the benefit in terms of HF hospitalizations was not evident in women in the study, which they said could have been due to the low number of women enrolled in the trial, and they recommended that the device be studied in more women.
Registry renal denervation skews from resistant hypertension
WASHINGTON – Renal denervation may have struck out as antihypertensive therapy in the sham-controlled SYMPLICITY HTN-3 trial reported in March at the American College of Cardiology’s annual meeting, but an independent report at the same meeting from a worldwide renal denervation registry showed that the vast majority of patients who’ve undergone with renal denervation recently were nothing like the patients enrolled in the failed trial.
In fact, patients who met enrollment criteria for the SYMPLICITY HTN-3 trial constituted a bare 8% of "real world" patients treated with renal denervation, based on results from the first 1,000 patients enrolled in the Global SYMPLICITY Registry, said Dr. Michael Böhm, professor and chairman of cardiology at Saarland University Hospital in Homburg/Saar, Germany.
Out of the 751 patients of the first 1,000 in the registry with data available on their office systolic blood pressure 6 months after treatment, 62 patients (8%) fulfilled the entry criteria of the SYMPLICITY HTN-3 (Renal Denervation in Patients With Uncontrolled Hypertension) trial by having an office systolic pressure of at least 160 mm Hg, a systolic pressure of at least 135 mm Hg by ambulatory blood pressure monitoring, and on treatment at maximally tolerated dosages with at least three different classes of antihypertensive medications (N. Engl. J. Med. 2014;370:1393-1401).
This low level of patients with clinically confirmed, resistant hypertension occurred against a backdrop in which a scant majority of registry patients even had severe hypertension, let alone a medically uncontrolled form. Among the 751 patients with 6-month follow-up blood pressure data, 433 (58%) received renal denervation with an office systolic blood pressure at baseline of at least 160 mm Hg, said Dr. Böhm.
"SYMPLICITY HTN-3 covered only a few percent of patients" who undergo renal denervation, he said. "The goal of the registry is to include a broad population, including patients without hypertension but other conditions associated with overactivity of the sympathetic nervous system" such as arrhythmia or heart failure.
Among the 433 patients with an office systolic blood pressure of at least 160 mm Hg – the minimum level of hypertension to warrant routine treatment by renal denervation according to several current policies – 244 (56%) were at this high pressure despite treatment with at least three classes of antihypertensive drugs. In this more clearly drug-resistant group, office systolic pressure fell by an average 20.2 mm Hg at 6 months after renal denervation. But only 62 patients on at least three types of drugs were also at maximally tolerated dosages of these drugs, another key element in defining drug resistance. Patients on maximally tolerated dosages represented only a quarter of all patients on multiple drugs in the Registry, and just 14% of the 433 patients with severe hypertension.
Among the 62 patients who matched the SYMPLICITY HTN-3 enrollment criteria, the average drop in office, systolic blood pressure at 6 months after treatment was 17.3 mm Hg, not too different from the average drop of 14.1 mm Hg seen in the renal denervation arm of SYMPLICITY HTN-3, Dr. Böhm noted.
The registry results showed that in patients with an office systolic pressure of less than 160 mm Hg, renal denervation was lousy at blood pressure reduction. Among the 222 patients who had pretreatment systolic pressures of 140-159 mm Hg and 6-month follow-up, office systolic blood pressure dropped by an average of 4.6 mm Hg. The procedure was even less effective in the 96 registry patients with an office systolic pressure of less than 140 mm Hg prior to treatment. In this subgroup systolic pressures averaged a 14.2-mm Hg increase 6 months after treatment.
"The only significant interaction between blood pressure lowering and no lowering was blood pressure at baseline," Dr. Böhm said. He gave no details on how many patients in the registry underwent renal denervation to treat hypertension and how many for other reasons.
The Global SYMPLICITY Register involves more than 200 centers in many European countries as well as in Canada, Australia, Korea, and elsewhere. A large number of registry centers are in Germany, and many centers there and elsewhere participated in the first two SYMPLICITY HTN trials. Participating centers entered the registry based on their experience with renal denervation and had to have performed at least 30 procedures prior to entry into the registry. The 6-month data also showed that renal denervation was "very safe," with "very low" rates of adverse events, Dr. Böhm said.
The Global SYMPLICITY Registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
On Twitter @mitchelzoler
WASHINGTON – Renal denervation may have struck out as antihypertensive therapy in the sham-controlled SYMPLICITY HTN-3 trial reported in March at the American College of Cardiology’s annual meeting, but an independent report at the same meeting from a worldwide renal denervation registry showed that the vast majority of patients who’ve undergone with renal denervation recently were nothing like the patients enrolled in the failed trial.
In fact, patients who met enrollment criteria for the SYMPLICITY HTN-3 trial constituted a bare 8% of "real world" patients treated with renal denervation, based on results from the first 1,000 patients enrolled in the Global SYMPLICITY Registry, said Dr. Michael Böhm, professor and chairman of cardiology at Saarland University Hospital in Homburg/Saar, Germany.
Out of the 751 patients of the first 1,000 in the registry with data available on their office systolic blood pressure 6 months after treatment, 62 patients (8%) fulfilled the entry criteria of the SYMPLICITY HTN-3 (Renal Denervation in Patients With Uncontrolled Hypertension) trial by having an office systolic pressure of at least 160 mm Hg, a systolic pressure of at least 135 mm Hg by ambulatory blood pressure monitoring, and on treatment at maximally tolerated dosages with at least three different classes of antihypertensive medications (N. Engl. J. Med. 2014;370:1393-1401).
This low level of patients with clinically confirmed, resistant hypertension occurred against a backdrop in which a scant majority of registry patients even had severe hypertension, let alone a medically uncontrolled form. Among the 751 patients with 6-month follow-up blood pressure data, 433 (58%) received renal denervation with an office systolic blood pressure at baseline of at least 160 mm Hg, said Dr. Böhm.
"SYMPLICITY HTN-3 covered only a few percent of patients" who undergo renal denervation, he said. "The goal of the registry is to include a broad population, including patients without hypertension but other conditions associated with overactivity of the sympathetic nervous system" such as arrhythmia or heart failure.
Among the 433 patients with an office systolic blood pressure of at least 160 mm Hg – the minimum level of hypertension to warrant routine treatment by renal denervation according to several current policies – 244 (56%) were at this high pressure despite treatment with at least three classes of antihypertensive drugs. In this more clearly drug-resistant group, office systolic pressure fell by an average 20.2 mm Hg at 6 months after renal denervation. But only 62 patients on at least three types of drugs were also at maximally tolerated dosages of these drugs, another key element in defining drug resistance. Patients on maximally tolerated dosages represented only a quarter of all patients on multiple drugs in the Registry, and just 14% of the 433 patients with severe hypertension.
Among the 62 patients who matched the SYMPLICITY HTN-3 enrollment criteria, the average drop in office, systolic blood pressure at 6 months after treatment was 17.3 mm Hg, not too different from the average drop of 14.1 mm Hg seen in the renal denervation arm of SYMPLICITY HTN-3, Dr. Böhm noted.
The registry results showed that in patients with an office systolic pressure of less than 160 mm Hg, renal denervation was lousy at blood pressure reduction. Among the 222 patients who had pretreatment systolic pressures of 140-159 mm Hg and 6-month follow-up, office systolic blood pressure dropped by an average of 4.6 mm Hg. The procedure was even less effective in the 96 registry patients with an office systolic pressure of less than 140 mm Hg prior to treatment. In this subgroup systolic pressures averaged a 14.2-mm Hg increase 6 months after treatment.
"The only significant interaction between blood pressure lowering and no lowering was blood pressure at baseline," Dr. Böhm said. He gave no details on how many patients in the registry underwent renal denervation to treat hypertension and how many for other reasons.
The Global SYMPLICITY Register involves more than 200 centers in many European countries as well as in Canada, Australia, Korea, and elsewhere. A large number of registry centers are in Germany, and many centers there and elsewhere participated in the first two SYMPLICITY HTN trials. Participating centers entered the registry based on their experience with renal denervation and had to have performed at least 30 procedures prior to entry into the registry. The 6-month data also showed that renal denervation was "very safe," with "very low" rates of adverse events, Dr. Böhm said.
The Global SYMPLICITY Registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
On Twitter @mitchelzoler
WASHINGTON – Renal denervation may have struck out as antihypertensive therapy in the sham-controlled SYMPLICITY HTN-3 trial reported in March at the American College of Cardiology’s annual meeting, but an independent report at the same meeting from a worldwide renal denervation registry showed that the vast majority of patients who’ve undergone with renal denervation recently were nothing like the patients enrolled in the failed trial.
In fact, patients who met enrollment criteria for the SYMPLICITY HTN-3 trial constituted a bare 8% of "real world" patients treated with renal denervation, based on results from the first 1,000 patients enrolled in the Global SYMPLICITY Registry, said Dr. Michael Böhm, professor and chairman of cardiology at Saarland University Hospital in Homburg/Saar, Germany.
Out of the 751 patients of the first 1,000 in the registry with data available on their office systolic blood pressure 6 months after treatment, 62 patients (8%) fulfilled the entry criteria of the SYMPLICITY HTN-3 (Renal Denervation in Patients With Uncontrolled Hypertension) trial by having an office systolic pressure of at least 160 mm Hg, a systolic pressure of at least 135 mm Hg by ambulatory blood pressure monitoring, and on treatment at maximally tolerated dosages with at least three different classes of antihypertensive medications (N. Engl. J. Med. 2014;370:1393-1401).
This low level of patients with clinically confirmed, resistant hypertension occurred against a backdrop in which a scant majority of registry patients even had severe hypertension, let alone a medically uncontrolled form. Among the 751 patients with 6-month follow-up blood pressure data, 433 (58%) received renal denervation with an office systolic blood pressure at baseline of at least 160 mm Hg, said Dr. Böhm.
"SYMPLICITY HTN-3 covered only a few percent of patients" who undergo renal denervation, he said. "The goal of the registry is to include a broad population, including patients without hypertension but other conditions associated with overactivity of the sympathetic nervous system" such as arrhythmia or heart failure.
Among the 433 patients with an office systolic blood pressure of at least 160 mm Hg – the minimum level of hypertension to warrant routine treatment by renal denervation according to several current policies – 244 (56%) were at this high pressure despite treatment with at least three classes of antihypertensive drugs. In this more clearly drug-resistant group, office systolic pressure fell by an average 20.2 mm Hg at 6 months after renal denervation. But only 62 patients on at least three types of drugs were also at maximally tolerated dosages of these drugs, another key element in defining drug resistance. Patients on maximally tolerated dosages represented only a quarter of all patients on multiple drugs in the Registry, and just 14% of the 433 patients with severe hypertension.
Among the 62 patients who matched the SYMPLICITY HTN-3 enrollment criteria, the average drop in office, systolic blood pressure at 6 months after treatment was 17.3 mm Hg, not too different from the average drop of 14.1 mm Hg seen in the renal denervation arm of SYMPLICITY HTN-3, Dr. Böhm noted.
The registry results showed that in patients with an office systolic pressure of less than 160 mm Hg, renal denervation was lousy at blood pressure reduction. Among the 222 patients who had pretreatment systolic pressures of 140-159 mm Hg and 6-month follow-up, office systolic blood pressure dropped by an average of 4.6 mm Hg. The procedure was even less effective in the 96 registry patients with an office systolic pressure of less than 140 mm Hg prior to treatment. In this subgroup systolic pressures averaged a 14.2-mm Hg increase 6 months after treatment.
"The only significant interaction between blood pressure lowering and no lowering was blood pressure at baseline," Dr. Böhm said. He gave no details on how many patients in the registry underwent renal denervation to treat hypertension and how many for other reasons.
The Global SYMPLICITY Register involves more than 200 centers in many European countries as well as in Canada, Australia, Korea, and elsewhere. A large number of registry centers are in Germany, and many centers there and elsewhere participated in the first two SYMPLICITY HTN trials. Participating centers entered the registry based on their experience with renal denervation and had to have performed at least 30 procedures prior to entry into the registry. The 6-month data also showed that renal denervation was "very safe," with "very low" rates of adverse events, Dr. Böhm said.
The Global SYMPLICITY Registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
On Twitter @mitchelzoler
AT ACC 2014
Key clinical point: Real-world use of renal denervation has not targeted patients with drug-resistant hypertension.
Major finding: Only 8% of patient in the Global SYMPLICITY Registry for renal denervation matched the patients enrolled in the SYMPLICITY HTN-3 trial.
Data source: The Global SYMPLICITY Registry, which enrolled 1,000 patients who underwent renal denervation at more than 200 centers worldwide.
Disclosures: The registry is sponsored by Medtronic, the company that markets Symplicity renal denervation devices. Dr, Böhm said that he has served on an advisory board to, been a speaker for, and received research support from Medtronic as well as from several other drug and device companies.
In diabetes, women far more likely to develop CHD than men
Women with diabetes have a 44% greater chance of developing coronary heart disease than do men with diabetes, a large review and meta-analysis has shown.
In a retrospective review of data from more than 850,000 people that included over 28,000 confirmed coronary heart disease (CHD) events across the globe between 1966 and 2011, women with diabetes had nearly three times the likelihood (relative risk, 2.82) of developing CHD than did women without diabetes. Meanwhile, men with diabetes were only twice as likely (RR, 2.16) to have CHD than men without the disease. After adjustment for sex differences in other CHD factors such as tobacco use, women with diabetes had a 44% increased risk of developing heart disease, compared with men with the disease (RR, 1.44).
Sex differences in diabetes-related risk for CHD remained consistent across subgroups defined by age and region, and were unchanged when factoring nonfatal CHD events.
The study, conducted by researchers from Europe and Australia, appears online in Diabetologia 2014 [doi:10.1007/s00125-014-3260-6]. The findings corroborate a previous meta-analysis that also showed clinically meaningful sex differences lead to a greater CHD risk in women with diabetes than in men with diabetes (BMJ 2006;332:73-8 [doi:10.1136/bmj.38678.389583.7C]. Dr. Rachel Huxley of the University of Queensland School of Population Health in Australia, was an investigator in both studies.
Reasons for the disparity cannot be attributed to pharmacotherapy alone, according to the authors of the current study. Data reviewed in the study showed that women with diabetes were undertreated for cardiovascular disease risk factors prior to 1986, but that despite current increased awareness of the cardiovascular risks posed to women with diabetes, they are still less likely to achieve treatment targets.
“We hypothesize that the excess risk in women is due to a combination of both a greater deterioration in cardiovascular risk factor levels and a chronically elevated cardiovascular risk profile in the prediabetic state, driven by greater levels of adiposity in women, compared with men,” the authors wrote.
The authors cited previous research indicating that men develop diabetes at a lower body mass index (BMI), compared with women. As an example, in the UK General Practice Research Database, the BMI of those diagnosed with diabetes was on average 1.8 kg/m2 higher in women than in men. (Diabetologia 2012;55:1556-7).
“Greater awareness of early symptoms of CHD in women and sex-specific therapeutic risk factor management, irrespective of the presence of diabetes, is optimal for improving clinical outcomes in both women and men,” the authors concluded.
wmcknight@frontlinemedcom.com
On Twitter @whitneymcknight
Women with diabetes have a 44% greater chance of developing coronary heart disease than do men with diabetes, a large review and meta-analysis has shown.
In a retrospective review of data from more than 850,000 people that included over 28,000 confirmed coronary heart disease (CHD) events across the globe between 1966 and 2011, women with diabetes had nearly three times the likelihood (relative risk, 2.82) of developing CHD than did women without diabetes. Meanwhile, men with diabetes were only twice as likely (RR, 2.16) to have CHD than men without the disease. After adjustment for sex differences in other CHD factors such as tobacco use, women with diabetes had a 44% increased risk of developing heart disease, compared with men with the disease (RR, 1.44).
Sex differences in diabetes-related risk for CHD remained consistent across subgroups defined by age and region, and were unchanged when factoring nonfatal CHD events.
The study, conducted by researchers from Europe and Australia, appears online in Diabetologia 2014 [doi:10.1007/s00125-014-3260-6]. The findings corroborate a previous meta-analysis that also showed clinically meaningful sex differences lead to a greater CHD risk in women with diabetes than in men with diabetes (BMJ 2006;332:73-8 [doi:10.1136/bmj.38678.389583.7C]. Dr. Rachel Huxley of the University of Queensland School of Population Health in Australia, was an investigator in both studies.
Reasons for the disparity cannot be attributed to pharmacotherapy alone, according to the authors of the current study. Data reviewed in the study showed that women with diabetes were undertreated for cardiovascular disease risk factors prior to 1986, but that despite current increased awareness of the cardiovascular risks posed to women with diabetes, they are still less likely to achieve treatment targets.
“We hypothesize that the excess risk in women is due to a combination of both a greater deterioration in cardiovascular risk factor levels and a chronically elevated cardiovascular risk profile in the prediabetic state, driven by greater levels of adiposity in women, compared with men,” the authors wrote.
The authors cited previous research indicating that men develop diabetes at a lower body mass index (BMI), compared with women. As an example, in the UK General Practice Research Database, the BMI of those diagnosed with diabetes was on average 1.8 kg/m2 higher in women than in men. (Diabetologia 2012;55:1556-7).
“Greater awareness of early symptoms of CHD in women and sex-specific therapeutic risk factor management, irrespective of the presence of diabetes, is optimal for improving clinical outcomes in both women and men,” the authors concluded.
wmcknight@frontlinemedcom.com
On Twitter @whitneymcknight
Women with diabetes have a 44% greater chance of developing coronary heart disease than do men with diabetes, a large review and meta-analysis has shown.
In a retrospective review of data from more than 850,000 people that included over 28,000 confirmed coronary heart disease (CHD) events across the globe between 1966 and 2011, women with diabetes had nearly three times the likelihood (relative risk, 2.82) of developing CHD than did women without diabetes. Meanwhile, men with diabetes were only twice as likely (RR, 2.16) to have CHD than men without the disease. After adjustment for sex differences in other CHD factors such as tobacco use, women with diabetes had a 44% increased risk of developing heart disease, compared with men with the disease (RR, 1.44).
Sex differences in diabetes-related risk for CHD remained consistent across subgroups defined by age and region, and were unchanged when factoring nonfatal CHD events.
The study, conducted by researchers from Europe and Australia, appears online in Diabetologia 2014 [doi:10.1007/s00125-014-3260-6]. The findings corroborate a previous meta-analysis that also showed clinically meaningful sex differences lead to a greater CHD risk in women with diabetes than in men with diabetes (BMJ 2006;332:73-8 [doi:10.1136/bmj.38678.389583.7C]. Dr. Rachel Huxley of the University of Queensland School of Population Health in Australia, was an investigator in both studies.
Reasons for the disparity cannot be attributed to pharmacotherapy alone, according to the authors of the current study. Data reviewed in the study showed that women with diabetes were undertreated for cardiovascular disease risk factors prior to 1986, but that despite current increased awareness of the cardiovascular risks posed to women with diabetes, they are still less likely to achieve treatment targets.
“We hypothesize that the excess risk in women is due to a combination of both a greater deterioration in cardiovascular risk factor levels and a chronically elevated cardiovascular risk profile in the prediabetic state, driven by greater levels of adiposity in women, compared with men,” the authors wrote.
The authors cited previous research indicating that men develop diabetes at a lower body mass index (BMI), compared with women. As an example, in the UK General Practice Research Database, the BMI of those diagnosed with diabetes was on average 1.8 kg/m2 higher in women than in men. (Diabetologia 2012;55:1556-7).
“Greater awareness of early symptoms of CHD in women and sex-specific therapeutic risk factor management, irrespective of the presence of diabetes, is optimal for improving clinical outcomes in both women and men,” the authors concluded.
wmcknight@frontlinemedcom.com
On Twitter @whitneymcknight
FROM DIABETOLOGIA
Key clinical point: Consider screening asymptomatic women for diabetes and offering risk
factor management.
Major finding: Women with diabetes are 44% more
likely to develop CHD than men with diabetes.
Data source: Systematic review and meta-analysis of
more than 850,000 people and over 28,000 CHD events recorded in 64 studies
worldwide between 1966 and 2011.
Disclosures: The authors of this study declared
they had no relevant disclosures.
Heart failure: Quality of life, diastolic function rose with intensity-interval exercise
WASHINGTON – A high-intensity cardiac rehabilitation program safely improved quality of life, diastolic function, depressive symptoms, and physical fitness in patients with systolic heart failure and reduced ejection fraction in a randomized controlled trial.
In the subgroup of study participants over age 65, however, the benefits were limited and the dropout rate high, Dr. Christina Chrysohoou said at the annual meeting of the American College of Cardiology. "I think patients over age 65 may benefit more from a less-intensive exercise time and rest periods."
Her study included 100 consecutive patients with a left ventricular ejection fraction (LVEF) below 30%; heart failure of ischemic etiology was present in 70%. One-third of subjects had an implantable cardioverter-defibrillator at study entry. All participants underwent pre-enrollment Holter monitoring with electrophysiologic follow-up as warranted. Participants were randomized to a high-intensity exercise program or to a control arm of standard dietary advice and a recommendation to walk for up to 2 miles daily, said Dr. Chrysohoou, a cardiologist at the University of Athens.
The exercise program consisted of a warm-up followed a 30-minute session of alternating 30-second bursts of ergometric exercise at 100% of a patient’s maximum workload followed by 30 seconds of recovery at 40%-60% of maximum workload. Maximum workload was determined from a baseline treadmill exercise test. The 12-week study was completed by 33 of 50 patients in the high-intensity exercise group and 39 of 50 controls.
Quality of life scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean score of 21 at baseline to 7 in the intensive-exercise group, and declined slightly from 19 to 21 in the control group.
At baseline, the mean score on the Zung Depression Scale was 37 out of a possible 80; at 12 weeks, scores improved to 30 in the high-intensity exercise group and increased to 41 in the controls.
Maximal oxygen consumption, or VO2max, improved from 16 to 21 mL/kg per minute in the interval-exercise group while remaining unchanged in controls. Similarly, peak power output increased from 84 to 105 W in the exercise program participants while remaining unchanged in controls. Six-minute walk time improved from 422 m to 476 m in the intensive exercisers, a 13% better result than that seen in controls.
Diastolic function on Doppler imaging significantly improved in the exercise group but not in the controls. The E/A wave ratio, which represents the relationship between early passive left ventricular filling and atrial contraction in late diastole, decreased by 24%. Also, the left ventricular outflow velocity integral increased by 4%.
There were no adverse events in either study arm.
LVEF did not significantly improve in either study arm, but that was not surprising, said Dr. Chrysohoou. "You may have an LVEF of 20% and be able to run a marathon, or an LVEF of 30%-35% and not even be able to walk around the house."
This was an unfunded study, and Dr. Chrysohoou reported having no financial conflicts.
WASHINGTON – A high-intensity cardiac rehabilitation program safely improved quality of life, diastolic function, depressive symptoms, and physical fitness in patients with systolic heart failure and reduced ejection fraction in a randomized controlled trial.
In the subgroup of study participants over age 65, however, the benefits were limited and the dropout rate high, Dr. Christina Chrysohoou said at the annual meeting of the American College of Cardiology. "I think patients over age 65 may benefit more from a less-intensive exercise time and rest periods."
Her study included 100 consecutive patients with a left ventricular ejection fraction (LVEF) below 30%; heart failure of ischemic etiology was present in 70%. One-third of subjects had an implantable cardioverter-defibrillator at study entry. All participants underwent pre-enrollment Holter monitoring with electrophysiologic follow-up as warranted. Participants were randomized to a high-intensity exercise program or to a control arm of standard dietary advice and a recommendation to walk for up to 2 miles daily, said Dr. Chrysohoou, a cardiologist at the University of Athens.
The exercise program consisted of a warm-up followed a 30-minute session of alternating 30-second bursts of ergometric exercise at 100% of a patient’s maximum workload followed by 30 seconds of recovery at 40%-60% of maximum workload. Maximum workload was determined from a baseline treadmill exercise test. The 12-week study was completed by 33 of 50 patients in the high-intensity exercise group and 39 of 50 controls.
Quality of life scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean score of 21 at baseline to 7 in the intensive-exercise group, and declined slightly from 19 to 21 in the control group.
At baseline, the mean score on the Zung Depression Scale was 37 out of a possible 80; at 12 weeks, scores improved to 30 in the high-intensity exercise group and increased to 41 in the controls.
Maximal oxygen consumption, or VO2max, improved from 16 to 21 mL/kg per minute in the interval-exercise group while remaining unchanged in controls. Similarly, peak power output increased from 84 to 105 W in the exercise program participants while remaining unchanged in controls. Six-minute walk time improved from 422 m to 476 m in the intensive exercisers, a 13% better result than that seen in controls.
Diastolic function on Doppler imaging significantly improved in the exercise group but not in the controls. The E/A wave ratio, which represents the relationship between early passive left ventricular filling and atrial contraction in late diastole, decreased by 24%. Also, the left ventricular outflow velocity integral increased by 4%.
There were no adverse events in either study arm.
LVEF did not significantly improve in either study arm, but that was not surprising, said Dr. Chrysohoou. "You may have an LVEF of 20% and be able to run a marathon, or an LVEF of 30%-35% and not even be able to walk around the house."
This was an unfunded study, and Dr. Chrysohoou reported having no financial conflicts.
WASHINGTON – A high-intensity cardiac rehabilitation program safely improved quality of life, diastolic function, depressive symptoms, and physical fitness in patients with systolic heart failure and reduced ejection fraction in a randomized controlled trial.
In the subgroup of study participants over age 65, however, the benefits were limited and the dropout rate high, Dr. Christina Chrysohoou said at the annual meeting of the American College of Cardiology. "I think patients over age 65 may benefit more from a less-intensive exercise time and rest periods."
Her study included 100 consecutive patients with a left ventricular ejection fraction (LVEF) below 30%; heart failure of ischemic etiology was present in 70%. One-third of subjects had an implantable cardioverter-defibrillator at study entry. All participants underwent pre-enrollment Holter monitoring with electrophysiologic follow-up as warranted. Participants were randomized to a high-intensity exercise program or to a control arm of standard dietary advice and a recommendation to walk for up to 2 miles daily, said Dr. Chrysohoou, a cardiologist at the University of Athens.
The exercise program consisted of a warm-up followed a 30-minute session of alternating 30-second bursts of ergometric exercise at 100% of a patient’s maximum workload followed by 30 seconds of recovery at 40%-60% of maximum workload. Maximum workload was determined from a baseline treadmill exercise test. The 12-week study was completed by 33 of 50 patients in the high-intensity exercise group and 39 of 50 controls.
Quality of life scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean score of 21 at baseline to 7 in the intensive-exercise group, and declined slightly from 19 to 21 in the control group.
At baseline, the mean score on the Zung Depression Scale was 37 out of a possible 80; at 12 weeks, scores improved to 30 in the high-intensity exercise group and increased to 41 in the controls.
Maximal oxygen consumption, or VO2max, improved from 16 to 21 mL/kg per minute in the interval-exercise group while remaining unchanged in controls. Similarly, peak power output increased from 84 to 105 W in the exercise program participants while remaining unchanged in controls. Six-minute walk time improved from 422 m to 476 m in the intensive exercisers, a 13% better result than that seen in controls.
Diastolic function on Doppler imaging significantly improved in the exercise group but not in the controls. The E/A wave ratio, which represents the relationship between early passive left ventricular filling and atrial contraction in late diastole, decreased by 24%. Also, the left ventricular outflow velocity integral increased by 4%.
There were no adverse events in either study arm.
LVEF did not significantly improve in either study arm, but that was not surprising, said Dr. Chrysohoou. "You may have an LVEF of 20% and be able to run a marathon, or an LVEF of 30%-35% and not even be able to walk around the house."
This was an unfunded study, and Dr. Chrysohoou reported having no financial conflicts.
AT ACC 14
Key clinical point: Cardiac rehabilitation benefits systolic heart failure patients, especially those under age 65.
Major finding: Quality of life scores on the Minnesota Living with Heart Failure Questionnaire improved from a mean of 21 to 7 over the course of a 12-week structured program of high-intensity interval exercise.
Data source: A 12-week, randomized, prospective study of 100 consecutive patients with heart failure and an LVEF below 30%.
Disclosures: This study was unfunded, and Dr. Chrysohoou reported having no financial conflicts.
FDA rejects serelaxin for acute heart failure
The Food and Drug Administration has decided that more evidence of serelaxin’s efficacy as a treatment for acute heart failure is needed before the drug can be approved in the United States, Novartis Pharmaceuticals announced May 16.
Serelaxin is a recombinant form of human relaxin-2 hormone that is administered in an intravenous infusion over 48 hours. For approval, the company submitted data from the phase III RELAX-AHF study, an international, randomized, placebo-controlled, double-blind study of about 1,100 patients with acute heart failure. At a meeting in March, the FDA’s Cardiovascular and Renal Drugs Advisory Committee agreed there was evidence that serelaxin had a positive effect on worsening heart failure in the study and that the data were promising, but unanimously voted against recommending approval, citing the need for more studies.
"In accordance with the FDA’s advice we will continue to expedite our clinical trial program to build the supporting body of evidence," Tim Wright, global head of development of the company, said in a statement issued by the company. He added that the company was encouraged by the FDA panel’s feedback.
The company is collecting more data on the drug in acute heart failure, which includes the RELAX-AHF-2 study, which will enroll more than 6,300 patients, according to the statement.
The FDA informs companies about nonapproval decisions in complete response letters, which include advice about what would be needed before the drug can be approved. The FDA does not announce these decisions, but companies often announce when these decisions are made.
The Food and Drug Administration has decided that more evidence of serelaxin’s efficacy as a treatment for acute heart failure is needed before the drug can be approved in the United States, Novartis Pharmaceuticals announced May 16.
Serelaxin is a recombinant form of human relaxin-2 hormone that is administered in an intravenous infusion over 48 hours. For approval, the company submitted data from the phase III RELAX-AHF study, an international, randomized, placebo-controlled, double-blind study of about 1,100 patients with acute heart failure. At a meeting in March, the FDA’s Cardiovascular and Renal Drugs Advisory Committee agreed there was evidence that serelaxin had a positive effect on worsening heart failure in the study and that the data were promising, but unanimously voted against recommending approval, citing the need for more studies.
"In accordance with the FDA’s advice we will continue to expedite our clinical trial program to build the supporting body of evidence," Tim Wright, global head of development of the company, said in a statement issued by the company. He added that the company was encouraged by the FDA panel’s feedback.
The company is collecting more data on the drug in acute heart failure, which includes the RELAX-AHF-2 study, which will enroll more than 6,300 patients, according to the statement.
The FDA informs companies about nonapproval decisions in complete response letters, which include advice about what would be needed before the drug can be approved. The FDA does not announce these decisions, but companies often announce when these decisions are made.
The Food and Drug Administration has decided that more evidence of serelaxin’s efficacy as a treatment for acute heart failure is needed before the drug can be approved in the United States, Novartis Pharmaceuticals announced May 16.
Serelaxin is a recombinant form of human relaxin-2 hormone that is administered in an intravenous infusion over 48 hours. For approval, the company submitted data from the phase III RELAX-AHF study, an international, randomized, placebo-controlled, double-blind study of about 1,100 patients with acute heart failure. At a meeting in March, the FDA’s Cardiovascular and Renal Drugs Advisory Committee agreed there was evidence that serelaxin had a positive effect on worsening heart failure in the study and that the data were promising, but unanimously voted against recommending approval, citing the need for more studies.
"In accordance with the FDA’s advice we will continue to expedite our clinical trial program to build the supporting body of evidence," Tim Wright, global head of development of the company, said in a statement issued by the company. He added that the company was encouraged by the FDA panel’s feedback.
The company is collecting more data on the drug in acute heart failure, which includes the RELAX-AHF-2 study, which will enroll more than 6,300 patients, according to the statement.
The FDA informs companies about nonapproval decisions in complete response letters, which include advice about what would be needed before the drug can be approved. The FDA does not announce these decisions, but companies often announce when these decisions are made.
20-study analysis finds no MACE increase with saxagliptin
Treatment with saxagliptin was not associated with an increased risk of major adverse cardiovascular events or heart failure in a pooled analysis of 20 studies of patients with type 2 diabetes.
The analysis also found no association with saxagliptin therapy and individual components of the major adverse cardiovascular events (MACE) endpoint, a composite of cardiovascular death, myocardial infarction, and stroke, according to a study being presented at the meeting. The results were released May 15 at the annual meeting of the American Association of Clinical Endocrinologists in Las Vegas.
Saxagliptin, a dipeptidyl peptidase–4 (DPP-44) inhibitor taken orally, was approved by the Food and Drug Administration in 2009 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes.
The study evaluated adjudicated reports of MACE and investigator reports of heart failure associated with saxagliptin in 20 studies of saxagliptin as monotherapy or as add-on therapy in 9,156 patients with type 2 diabetes. Unlike the SAVOR-TIMI 53 study (Saxagliptin Assessment of Vascular Outcomes Recorded in Patient With Diabetes Mellitus–Thrombolysis in Myocardial Infarction 53) study, published in 2013, which found an increased risk of heart failure hospitalizations in patients with prior CV disease or multiple CV risk factors, the patients with type 2 diabetes enrolled in the 20 studies were at lower risk for CV events.
In the SAVOR-TIMI 53, which evaluated the drug’s cardiovascular safety in more than 16,000 patients with type 2 diabetes and established cardiovascular disease or risk factors for CHD, treatment with saxagliptin was not associated with an increased or reduced risk of ischemic risk for the composite of CV death, MI, or ischemic stroke, the primary endpoint, over a median of 2 years. But it was associated with a significantly greater risk of being hospitalized for heart failure (hazard ratio, 1.27) (N. Engl. J. Med. 2013;369:1317-26). In a January 2014 safety alert, the FDA said that the agency had requested the manufacturer of saxagliptin study “a possible association” between the use of the drug and heart failure, because of the increased risk of heart failure hospitalizations in that study.
In the 20-study analysis, based on 43 MACE reports in patients treated with saxagliptin and 31 MACE reports among controls, the MACE incidence rate (IR) – the number of events per 100 person-years – was 0.85/100 person-years for those on saxagliptin and 1.12/100 person-years for the controls. The IR ratio (the number of reports among saxagliptin-treated patients divided by the number of reports among the controls), was 0.74. The hazard ratio was 0.75, which suggests there is no increased risk of MACE associated with saxagliptin.
In a subset of 11 studies that evaluated saxagliptin as add-on therapy to metformin in 5,171 patients, the incidence rates were similar: 0.79/100 person-years for those on saxagliptin and 0.85/100 person-years for controls, with an IR ratio of 0.93.
In the 20 studies, the components of the MACE endpoint were also not statistically significantly different between those on saxagliptin and controls, and 21 patients on saxagliptin and 18 controls had heart failure, with an IR ratio of 0.55, he said.
Consistent with the SAVOR-TIMI 53 study, which analyzed MACE in patients with type 2 diabetes and cardiovascular disease or multiple cardiovascular risk factors, this 20-trial pooled analysis of patients from the general type 2 diabetes population found that saxagliptin was not associated with an increased risk of MACE or its components, or heart failure.
The pooled results “are somewhat reassuring in that the use of saxagliptin in patients with type 2 diabetes at lower risk for cardiovascular events is not associated with increased MACE risk,” and would have passed the FDA-recommended boundary for unacceptable risk for diabetes drugs (HR, 1.3), Dr. Sanjay Kaul said in an interview. However, he added, “it is disappointing that glycemic control did not translate into a macrovascular benefit, even though a lower risk population was evaluated.”
Moreover, the study “did not yield a sufficient number of heart failure events to reliably adjudicate the risk of heart failure in this patient population,” noted Dr. Kaul, professor of medicine, University of California, Los Angeles, who was not at the meeting. Dr. Kaul was a panelist at the FDA advisory panel meeting that reviewed the cardiovascular safety of rosiglitazone (Avandia) last year.
Three of the five authors are employed by Bristol-Myers Squibb; two are employed by AstraZeneca Dr. Kaul had no relevant disclosures related to saxagliptin; he has disclosures for diabetes agents manufactured by other companies.
Saxagliptin is marketed as Onglyza by AstraZeneca.
emechcatie@frontlinemedcom.com
Treatment with saxagliptin was not associated with an increased risk of major adverse cardiovascular events or heart failure in a pooled analysis of 20 studies of patients with type 2 diabetes.
The analysis also found no association with saxagliptin therapy and individual components of the major adverse cardiovascular events (MACE) endpoint, a composite of cardiovascular death, myocardial infarction, and stroke, according to a study being presented at the meeting. The results were released May 15 at the annual meeting of the American Association of Clinical Endocrinologists in Las Vegas.
Saxagliptin, a dipeptidyl peptidase–4 (DPP-44) inhibitor taken orally, was approved by the Food and Drug Administration in 2009 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes.
The study evaluated adjudicated reports of MACE and investigator reports of heart failure associated with saxagliptin in 20 studies of saxagliptin as monotherapy or as add-on therapy in 9,156 patients with type 2 diabetes. Unlike the SAVOR-TIMI 53 study (Saxagliptin Assessment of Vascular Outcomes Recorded in Patient With Diabetes Mellitus–Thrombolysis in Myocardial Infarction 53) study, published in 2013, which found an increased risk of heart failure hospitalizations in patients with prior CV disease or multiple CV risk factors, the patients with type 2 diabetes enrolled in the 20 studies were at lower risk for CV events.
In the SAVOR-TIMI 53, which evaluated the drug’s cardiovascular safety in more than 16,000 patients with type 2 diabetes and established cardiovascular disease or risk factors for CHD, treatment with saxagliptin was not associated with an increased or reduced risk of ischemic risk for the composite of CV death, MI, or ischemic stroke, the primary endpoint, over a median of 2 years. But it was associated with a significantly greater risk of being hospitalized for heart failure (hazard ratio, 1.27) (N. Engl. J. Med. 2013;369:1317-26). In a January 2014 safety alert, the FDA said that the agency had requested the manufacturer of saxagliptin study “a possible association” between the use of the drug and heart failure, because of the increased risk of heart failure hospitalizations in that study.
In the 20-study analysis, based on 43 MACE reports in patients treated with saxagliptin and 31 MACE reports among controls, the MACE incidence rate (IR) – the number of events per 100 person-years – was 0.85/100 person-years for those on saxagliptin and 1.12/100 person-years for the controls. The IR ratio (the number of reports among saxagliptin-treated patients divided by the number of reports among the controls), was 0.74. The hazard ratio was 0.75, which suggests there is no increased risk of MACE associated with saxagliptin.
In a subset of 11 studies that evaluated saxagliptin as add-on therapy to metformin in 5,171 patients, the incidence rates were similar: 0.79/100 person-years for those on saxagliptin and 0.85/100 person-years for controls, with an IR ratio of 0.93.
In the 20 studies, the components of the MACE endpoint were also not statistically significantly different between those on saxagliptin and controls, and 21 patients on saxagliptin and 18 controls had heart failure, with an IR ratio of 0.55, he said.
Consistent with the SAVOR-TIMI 53 study, which analyzed MACE in patients with type 2 diabetes and cardiovascular disease or multiple cardiovascular risk factors, this 20-trial pooled analysis of patients from the general type 2 diabetes population found that saxagliptin was not associated with an increased risk of MACE or its components, or heart failure.
The pooled results “are somewhat reassuring in that the use of saxagliptin in patients with type 2 diabetes at lower risk for cardiovascular events is not associated with increased MACE risk,” and would have passed the FDA-recommended boundary for unacceptable risk for diabetes drugs (HR, 1.3), Dr. Sanjay Kaul said in an interview. However, he added, “it is disappointing that glycemic control did not translate into a macrovascular benefit, even though a lower risk population was evaluated.”
Moreover, the study “did not yield a sufficient number of heart failure events to reliably adjudicate the risk of heart failure in this patient population,” noted Dr. Kaul, professor of medicine, University of California, Los Angeles, who was not at the meeting. Dr. Kaul was a panelist at the FDA advisory panel meeting that reviewed the cardiovascular safety of rosiglitazone (Avandia) last year.
Three of the five authors are employed by Bristol-Myers Squibb; two are employed by AstraZeneca Dr. Kaul had no relevant disclosures related to saxagliptin; he has disclosures for diabetes agents manufactured by other companies.
Saxagliptin is marketed as Onglyza by AstraZeneca.
emechcatie@frontlinemedcom.com
Treatment with saxagliptin was not associated with an increased risk of major adverse cardiovascular events or heart failure in a pooled analysis of 20 studies of patients with type 2 diabetes.
The analysis also found no association with saxagliptin therapy and individual components of the major adverse cardiovascular events (MACE) endpoint, a composite of cardiovascular death, myocardial infarction, and stroke, according to a study being presented at the meeting. The results were released May 15 at the annual meeting of the American Association of Clinical Endocrinologists in Las Vegas.
Saxagliptin, a dipeptidyl peptidase–4 (DPP-44) inhibitor taken orally, was approved by the Food and Drug Administration in 2009 as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes.
The study evaluated adjudicated reports of MACE and investigator reports of heart failure associated with saxagliptin in 20 studies of saxagliptin as monotherapy or as add-on therapy in 9,156 patients with type 2 diabetes. Unlike the SAVOR-TIMI 53 study (Saxagliptin Assessment of Vascular Outcomes Recorded in Patient With Diabetes Mellitus–Thrombolysis in Myocardial Infarction 53) study, published in 2013, which found an increased risk of heart failure hospitalizations in patients with prior CV disease or multiple CV risk factors, the patients with type 2 diabetes enrolled in the 20 studies were at lower risk for CV events.
In the SAVOR-TIMI 53, which evaluated the drug’s cardiovascular safety in more than 16,000 patients with type 2 diabetes and established cardiovascular disease or risk factors for CHD, treatment with saxagliptin was not associated with an increased or reduced risk of ischemic risk for the composite of CV death, MI, or ischemic stroke, the primary endpoint, over a median of 2 years. But it was associated with a significantly greater risk of being hospitalized for heart failure (hazard ratio, 1.27) (N. Engl. J. Med. 2013;369:1317-26). In a January 2014 safety alert, the FDA said that the agency had requested the manufacturer of saxagliptin study “a possible association” between the use of the drug and heart failure, because of the increased risk of heart failure hospitalizations in that study.
In the 20-study analysis, based on 43 MACE reports in patients treated with saxagliptin and 31 MACE reports among controls, the MACE incidence rate (IR) – the number of events per 100 person-years – was 0.85/100 person-years for those on saxagliptin and 1.12/100 person-years for the controls. The IR ratio (the number of reports among saxagliptin-treated patients divided by the number of reports among the controls), was 0.74. The hazard ratio was 0.75, which suggests there is no increased risk of MACE associated with saxagliptin.
In a subset of 11 studies that evaluated saxagliptin as add-on therapy to metformin in 5,171 patients, the incidence rates were similar: 0.79/100 person-years for those on saxagliptin and 0.85/100 person-years for controls, with an IR ratio of 0.93.
In the 20 studies, the components of the MACE endpoint were also not statistically significantly different between those on saxagliptin and controls, and 21 patients on saxagliptin and 18 controls had heart failure, with an IR ratio of 0.55, he said.
Consistent with the SAVOR-TIMI 53 study, which analyzed MACE in patients with type 2 diabetes and cardiovascular disease or multiple cardiovascular risk factors, this 20-trial pooled analysis of patients from the general type 2 diabetes population found that saxagliptin was not associated with an increased risk of MACE or its components, or heart failure.
The pooled results “are somewhat reassuring in that the use of saxagliptin in patients with type 2 diabetes at lower risk for cardiovascular events is not associated with increased MACE risk,” and would have passed the FDA-recommended boundary for unacceptable risk for diabetes drugs (HR, 1.3), Dr. Sanjay Kaul said in an interview. However, he added, “it is disappointing that glycemic control did not translate into a macrovascular benefit, even though a lower risk population was evaluated.”
Moreover, the study “did not yield a sufficient number of heart failure events to reliably adjudicate the risk of heart failure in this patient population,” noted Dr. Kaul, professor of medicine, University of California, Los Angeles, who was not at the meeting. Dr. Kaul was a panelist at the FDA advisory panel meeting that reviewed the cardiovascular safety of rosiglitazone (Avandia) last year.
Three of the five authors are employed by Bristol-Myers Squibb; two are employed by AstraZeneca Dr. Kaul had no relevant disclosures related to saxagliptin; he has disclosures for diabetes agents manufactured by other companies.
Saxagliptin is marketed as Onglyza by AstraZeneca.
emechcatie@frontlinemedcom.com
FROM AACE 2014
Major finding: The incidence of MACE composite endpoint was similar among patients treated with saxagliptin (0.85/100 person-years) and controls (1.12/100 person-years) in 20 saxagliptin studies of patients with type 2 diabetes at a lower cardiovascular risk.
Data source: A pooled analysis of 20 randomized, controlled trials of saxagliptin as monotherapy or as add-on therapy in about 9,000 patients with type 2 diabetes compared the rates of the MACE endpoint and heart failure in patients on saxagliptin and controls.
Disclosures: Three of the five authors are employed by Bristol-Myers Squibb; two are employed by AstraZeneca.