User login
How to cut the CRT nonresponder rate
SNOWMASS, COLO. – The 12-lead Holter monitor is an invaluable and underutilized tool for turning cardiac resynchronization therapy nonresponders into responders, according to Dr. N.A. Mark Estes III.
“The Achilles heel of CRT right now is the failure to respond that occurs in one-quarter to one-third of patients who receive the device. And I think it’s less and less due to a problem of poor patient selection or technical failure, but instead due to common mistakes made in patients who’ve been properly selected and in whom a CRT device has been placed appropriately. I think there’s a lot of room for improvement here,” said Dr. Estes, director of the cardiac arrhythmia center and professor of medicine at Tufts University in Boston.
One common mistake is to accept at face value the CRT device counter when it shows a high percentage of ventricular pacing that’s supposed to be indicative of adequate biventricular pacing in patients. That’s not reliable in patients with coexisting heart failure and atrial fibrillation. In fact, the device pacing counter can markedly overestimate the degree of effective biventricular pacing in patients with permanent atrial fibrillation undergoing CRT. That’s because it may incorrectly count fusion and pseudofusion beats as paced events, even though ventricular capture and atrioventricular resynchronization fail to occur during such beats. The 12-lead Holter monitor will tell the real story regarding ventricular rate control and the percentage of fully paced beats, Dr. Estes said at the Annual Cardiovascular Conference at Snowmass.
“In patients with atrial fibrillation, you can’t just do an EKG, and you can’t just do an interrogation of the device and see that they have 90% or 95% biventricular pacing, which is the sweet spot you need to be North of to make sure they’re having benefit. You really need to do the 24-hour Holter monitor,” the cardiologist emphasized.
“It happens all the time,” he continued. “It happens in my own institution, where we have people come in with heart failure who are being evaluated for an LVAD [left ventricular assist device] because when their CRT device is interrogated, it shows 95% pacing. Everyone’s getting ready to put in the LVAD, and we put them on a Holter monitor and guess what? They’re 60% paced. And sometimes you can have dramatic – really dramatic – improvements by just slowing the ventricular response.”
Increasingly, however, he and his fellow electrophysiologists are alternatively turning to atrioventricular junction ablation in suitable candidates for the catheter-based procedure in order to improve their clinical response to CRT.
Dr. Estes cited a study by Columbia University cardiologists as being highly influential in shaping the new understanding of how to use 12-lead Holter monitoring to identify patients with atrial fibrillation who aren’t responding to CRT. They studied 19 patients with permanent atrial fibrillation who were on CRT, all of whom were on beta-blockers, amiodarone, and digoxin for rate control. Although device interrogation showed all 19 patients had in excess of 90% biventricular pacing 12 months after CRT implantation, only 9 patients were actually clinical responders to CRT as defined by at least a 1-grade improvement in New York Heart Association functional class.
Upon wearing an ambulatory 12-lead Holter monitor for 24 hours, only the nine patients with clinical improvement in response to CRT actually had effective pacing; 94% of their heart beats over the course of 24 hours were fully paced, with a mere 2% fusion beats and less than 4% pseudofusion beats. The other 10 patients had an average of only 60% fully paced beats, along with 16% fusion and 24% pseudofusion beats.
The CRT clinical responders also showed evidence of reverse remodeling, with significantly greater improvements in left ventricular ejection fraction, end-systolic dimension, end-diastolic diameter, and end-systolic and end-diastolic volume, compared with nonresponders.
The lesson, according to the investigators, is that the percentage of biventricular pacing, as obtained from CRT device interrogation, is an unreliable indicator of effective pacing in patients with permanent atrial fibrillation. The goal of CRT programming in this population is to try to achieve 100% biventricular capture on the 12-lead Holter monitor (J. Am. Coll. Cardiol. 2009;53:1050-5).
A series of landmark randomized trials has established that in suitable candidates for CRT with NYHA class I through ambulatory class IV heart failure with reduced ejection fraction, the device therapy provides on average about a 17% reduction in all-cause mortality and a 29% decrease in hospitalizations for heart failure above and beyond the benefits obtained through guideline-directed medical therapy. However, the improvements seen in patients with NYHA class I symptoms are less impressive than in those with NYHA class II-IV disease. And it’s possible that the randomized controlled trials overestimate the true benefit from CRT.
“This is a group of extremely experienced investigators who know the literature well. They’re typically quite savvy with patient selection, and they’re vested in getting the right patients into these trials. Unfortunately, there’s not a registry of all eligible patients, so we can’t tell exactly how those patients were selected. It would have been helpful,” Dr. Estes said.
It’s clear from the randomized trials that the ideal patient for CRT is a woman under age 65 with nonischemic cardiomyopathy with a left bundle branch block, normal sinus rhythm, and a QRS duration of 150 milliseconds or more. That’s who’s most likely to obtain clinical benefit from the therapy. The flip side of that is that an older male patient with ischemia, atrial fibrillation, a QRS of 120-150 milliseconds, and non–left bundle branch block is less likely to respond.
“We’ve figured this out. I think we’ve gotten pretty good at giving patients an honest estimate of their probability of responding independent of our ability to get a lead into a good spot in the coronary sinus,” Dr. Estes said. “More than 95% of patients can be adequately resynchronized currently with the tools we have, so it’s not the barrier that it once represented. We have many different techniques available.”
Despite intensive efforts by investigators using sophisticated nuclear studies, echocardiography, and MRI, however, nothing to date has been able to predict who’s going to respond to CRT as well as a simple electrical parameter: the QRS duration.
“It remains really the gold standard. It’s an electrical marker for what is a mechanical event,” Dr. Estes said.
He reported serving as a consultant to Medtronic, St. Jude Medical, and Boston Scientific and receiving research support from Boston Scientific and St. Jude Medical.
SNOWMASS, COLO. – The 12-lead Holter monitor is an invaluable and underutilized tool for turning cardiac resynchronization therapy nonresponders into responders, according to Dr. N.A. Mark Estes III.
“The Achilles heel of CRT right now is the failure to respond that occurs in one-quarter to one-third of patients who receive the device. And I think it’s less and less due to a problem of poor patient selection or technical failure, but instead due to common mistakes made in patients who’ve been properly selected and in whom a CRT device has been placed appropriately. I think there’s a lot of room for improvement here,” said Dr. Estes, director of the cardiac arrhythmia center and professor of medicine at Tufts University in Boston.
One common mistake is to accept at face value the CRT device counter when it shows a high percentage of ventricular pacing that’s supposed to be indicative of adequate biventricular pacing in patients. That’s not reliable in patients with coexisting heart failure and atrial fibrillation. In fact, the device pacing counter can markedly overestimate the degree of effective biventricular pacing in patients with permanent atrial fibrillation undergoing CRT. That’s because it may incorrectly count fusion and pseudofusion beats as paced events, even though ventricular capture and atrioventricular resynchronization fail to occur during such beats. The 12-lead Holter monitor will tell the real story regarding ventricular rate control and the percentage of fully paced beats, Dr. Estes said at the Annual Cardiovascular Conference at Snowmass.
“In patients with atrial fibrillation, you can’t just do an EKG, and you can’t just do an interrogation of the device and see that they have 90% or 95% biventricular pacing, which is the sweet spot you need to be North of to make sure they’re having benefit. You really need to do the 24-hour Holter monitor,” the cardiologist emphasized.
“It happens all the time,” he continued. “It happens in my own institution, where we have people come in with heart failure who are being evaluated for an LVAD [left ventricular assist device] because when their CRT device is interrogated, it shows 95% pacing. Everyone’s getting ready to put in the LVAD, and we put them on a Holter monitor and guess what? They’re 60% paced. And sometimes you can have dramatic – really dramatic – improvements by just slowing the ventricular response.”
Increasingly, however, he and his fellow electrophysiologists are alternatively turning to atrioventricular junction ablation in suitable candidates for the catheter-based procedure in order to improve their clinical response to CRT.
Dr. Estes cited a study by Columbia University cardiologists as being highly influential in shaping the new understanding of how to use 12-lead Holter monitoring to identify patients with atrial fibrillation who aren’t responding to CRT. They studied 19 patients with permanent atrial fibrillation who were on CRT, all of whom were on beta-blockers, amiodarone, and digoxin for rate control. Although device interrogation showed all 19 patients had in excess of 90% biventricular pacing 12 months after CRT implantation, only 9 patients were actually clinical responders to CRT as defined by at least a 1-grade improvement in New York Heart Association functional class.
Upon wearing an ambulatory 12-lead Holter monitor for 24 hours, only the nine patients with clinical improvement in response to CRT actually had effective pacing; 94% of their heart beats over the course of 24 hours were fully paced, with a mere 2% fusion beats and less than 4% pseudofusion beats. The other 10 patients had an average of only 60% fully paced beats, along with 16% fusion and 24% pseudofusion beats.
The CRT clinical responders also showed evidence of reverse remodeling, with significantly greater improvements in left ventricular ejection fraction, end-systolic dimension, end-diastolic diameter, and end-systolic and end-diastolic volume, compared with nonresponders.
The lesson, according to the investigators, is that the percentage of biventricular pacing, as obtained from CRT device interrogation, is an unreliable indicator of effective pacing in patients with permanent atrial fibrillation. The goal of CRT programming in this population is to try to achieve 100% biventricular capture on the 12-lead Holter monitor (J. Am. Coll. Cardiol. 2009;53:1050-5).
A series of landmark randomized trials has established that in suitable candidates for CRT with NYHA class I through ambulatory class IV heart failure with reduced ejection fraction, the device therapy provides on average about a 17% reduction in all-cause mortality and a 29% decrease in hospitalizations for heart failure above and beyond the benefits obtained through guideline-directed medical therapy. However, the improvements seen in patients with NYHA class I symptoms are less impressive than in those with NYHA class II-IV disease. And it’s possible that the randomized controlled trials overestimate the true benefit from CRT.
“This is a group of extremely experienced investigators who know the literature well. They’re typically quite savvy with patient selection, and they’re vested in getting the right patients into these trials. Unfortunately, there’s not a registry of all eligible patients, so we can’t tell exactly how those patients were selected. It would have been helpful,” Dr. Estes said.
It’s clear from the randomized trials that the ideal patient for CRT is a woman under age 65 with nonischemic cardiomyopathy with a left bundle branch block, normal sinus rhythm, and a QRS duration of 150 milliseconds or more. That’s who’s most likely to obtain clinical benefit from the therapy. The flip side of that is that an older male patient with ischemia, atrial fibrillation, a QRS of 120-150 milliseconds, and non–left bundle branch block is less likely to respond.
“We’ve figured this out. I think we’ve gotten pretty good at giving patients an honest estimate of their probability of responding independent of our ability to get a lead into a good spot in the coronary sinus,” Dr. Estes said. “More than 95% of patients can be adequately resynchronized currently with the tools we have, so it’s not the barrier that it once represented. We have many different techniques available.”
Despite intensive efforts by investigators using sophisticated nuclear studies, echocardiography, and MRI, however, nothing to date has been able to predict who’s going to respond to CRT as well as a simple electrical parameter: the QRS duration.
“It remains really the gold standard. It’s an electrical marker for what is a mechanical event,” Dr. Estes said.
He reported serving as a consultant to Medtronic, St. Jude Medical, and Boston Scientific and receiving research support from Boston Scientific and St. Jude Medical.
SNOWMASS, COLO. – The 12-lead Holter monitor is an invaluable and underutilized tool for turning cardiac resynchronization therapy nonresponders into responders, according to Dr. N.A. Mark Estes III.
“The Achilles heel of CRT right now is the failure to respond that occurs in one-quarter to one-third of patients who receive the device. And I think it’s less and less due to a problem of poor patient selection or technical failure, but instead due to common mistakes made in patients who’ve been properly selected and in whom a CRT device has been placed appropriately. I think there’s a lot of room for improvement here,” said Dr. Estes, director of the cardiac arrhythmia center and professor of medicine at Tufts University in Boston.
One common mistake is to accept at face value the CRT device counter when it shows a high percentage of ventricular pacing that’s supposed to be indicative of adequate biventricular pacing in patients. That’s not reliable in patients with coexisting heart failure and atrial fibrillation. In fact, the device pacing counter can markedly overestimate the degree of effective biventricular pacing in patients with permanent atrial fibrillation undergoing CRT. That’s because it may incorrectly count fusion and pseudofusion beats as paced events, even though ventricular capture and atrioventricular resynchronization fail to occur during such beats. The 12-lead Holter monitor will tell the real story regarding ventricular rate control and the percentage of fully paced beats, Dr. Estes said at the Annual Cardiovascular Conference at Snowmass.
“In patients with atrial fibrillation, you can’t just do an EKG, and you can’t just do an interrogation of the device and see that they have 90% or 95% biventricular pacing, which is the sweet spot you need to be North of to make sure they’re having benefit. You really need to do the 24-hour Holter monitor,” the cardiologist emphasized.
“It happens all the time,” he continued. “It happens in my own institution, where we have people come in with heart failure who are being evaluated for an LVAD [left ventricular assist device] because when their CRT device is interrogated, it shows 95% pacing. Everyone’s getting ready to put in the LVAD, and we put them on a Holter monitor and guess what? They’re 60% paced. And sometimes you can have dramatic – really dramatic – improvements by just slowing the ventricular response.”
Increasingly, however, he and his fellow electrophysiologists are alternatively turning to atrioventricular junction ablation in suitable candidates for the catheter-based procedure in order to improve their clinical response to CRT.
Dr. Estes cited a study by Columbia University cardiologists as being highly influential in shaping the new understanding of how to use 12-lead Holter monitoring to identify patients with atrial fibrillation who aren’t responding to CRT. They studied 19 patients with permanent atrial fibrillation who were on CRT, all of whom were on beta-blockers, amiodarone, and digoxin for rate control. Although device interrogation showed all 19 patients had in excess of 90% biventricular pacing 12 months after CRT implantation, only 9 patients were actually clinical responders to CRT as defined by at least a 1-grade improvement in New York Heart Association functional class.
Upon wearing an ambulatory 12-lead Holter monitor for 24 hours, only the nine patients with clinical improvement in response to CRT actually had effective pacing; 94% of their heart beats over the course of 24 hours were fully paced, with a mere 2% fusion beats and less than 4% pseudofusion beats. The other 10 patients had an average of only 60% fully paced beats, along with 16% fusion and 24% pseudofusion beats.
The CRT clinical responders also showed evidence of reverse remodeling, with significantly greater improvements in left ventricular ejection fraction, end-systolic dimension, end-diastolic diameter, and end-systolic and end-diastolic volume, compared with nonresponders.
The lesson, according to the investigators, is that the percentage of biventricular pacing, as obtained from CRT device interrogation, is an unreliable indicator of effective pacing in patients with permanent atrial fibrillation. The goal of CRT programming in this population is to try to achieve 100% biventricular capture on the 12-lead Holter monitor (J. Am. Coll. Cardiol. 2009;53:1050-5).
A series of landmark randomized trials has established that in suitable candidates for CRT with NYHA class I through ambulatory class IV heart failure with reduced ejection fraction, the device therapy provides on average about a 17% reduction in all-cause mortality and a 29% decrease in hospitalizations for heart failure above and beyond the benefits obtained through guideline-directed medical therapy. However, the improvements seen in patients with NYHA class I symptoms are less impressive than in those with NYHA class II-IV disease. And it’s possible that the randomized controlled trials overestimate the true benefit from CRT.
“This is a group of extremely experienced investigators who know the literature well. They’re typically quite savvy with patient selection, and they’re vested in getting the right patients into these trials. Unfortunately, there’s not a registry of all eligible patients, so we can’t tell exactly how those patients were selected. It would have been helpful,” Dr. Estes said.
It’s clear from the randomized trials that the ideal patient for CRT is a woman under age 65 with nonischemic cardiomyopathy with a left bundle branch block, normal sinus rhythm, and a QRS duration of 150 milliseconds or more. That’s who’s most likely to obtain clinical benefit from the therapy. The flip side of that is that an older male patient with ischemia, atrial fibrillation, a QRS of 120-150 milliseconds, and non–left bundle branch block is less likely to respond.
“We’ve figured this out. I think we’ve gotten pretty good at giving patients an honest estimate of their probability of responding independent of our ability to get a lead into a good spot in the coronary sinus,” Dr. Estes said. “More than 95% of patients can be adequately resynchronized currently with the tools we have, so it’s not the barrier that it once represented. We have many different techniques available.”
Despite intensive efforts by investigators using sophisticated nuclear studies, echocardiography, and MRI, however, nothing to date has been able to predict who’s going to respond to CRT as well as a simple electrical parameter: the QRS duration.
“It remains really the gold standard. It’s an electrical marker for what is a mechanical event,” Dr. Estes said.
He reported serving as a consultant to Medtronic, St. Jude Medical, and Boston Scientific and receiving research support from Boston Scientific and St. Jude Medical.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
How to tell constrictive pericarditis from mimickers
SNOWMASS, COLO. – Differentiating constrictive pericarditis from restrictive cardiomyopathy in patients with right heart failure with a normal ejection fraction is “one of the most difficult diagnostic challenges in cardiology today,” but reliable results can be achieved using a careful step-by-step approach, Dr. Rick A. Nishimura said at the Annual Cardiovascular Conference at Snowmass.
Under current terminology for heart failure, cardiologists speak of HFrEF, or heart failure with reduced ejection fraction, and HFpEF, or heart failure with preserved ejection fraction. But there is a third group of patients who are often mistakenly thought to have HFpEF: those with severe right heart failure and a normal ejection fraction, classically caused by constrictive pericarditis or restrictive cardiomyopathy.
Unlike patients with HFpEF, these people are not hypertensive and they don’t have pulmonary congestion. Instead, they present predominantly with ascites, peripheral edema, fatigue, and marked elevation in jugular venous pressure, noted Dr. Nishimura, professor of medicine at the Mayo Clinic in Rochester, Minn.
Before elaborating on his own tried-and-true, step-by-step approach, he highlighted several diagnostic procedures he considers less than reliable. One is advanced imaging with CT or MRI looking for the pericardial thickening that is widely viewed as an anatomic hallmark of constrictive pericarditis.
“Remember, 22% of patients with proven constrictive pericarditis actually have a normal pericardium on CT or MRI, because it’s their fibrotic epicardium that’s causing the constrictive pericarditis. And roughly 70% of patients are going to have some thickened pericardium after radiation therapy or coronary artery bypass graft surgery without having constrictive pericarditis. So CT and MRI are helpful, but they’re not going to be diagnostic,” according to the cardiologist.
Similarly, while it’s often been said that constrictive pericarditis can be diagnosed based upon a classic trio of hemodynamic findings obtained through heart catheterization – namely, early rapid filling with a left ventricular end-diastolic pressure equal to the right ventricular end-diastolic pressure, a right ventricular end-diastolic pressure greater than one-third of the right ventricular systolic pressure, and a pulmonary artery pressure below 50 mm Hg – these criteria didn’t reliably separate the last 100 patients who came to the catheterization lab at the Mayo Clinic with either constrictive pericarditis or restrictive cardiomyopathy, he continued.
Dr. Nishimura’s approach to the work-up of patients with unexplained right heart failure and a normal ejection begins with the history and physical examination. The history in a patient with constrictive pericarditis is classically one of radiation therapy years earlier for a malignancy, or prior CABG surgery. And the physical exam has to reveal the presence of high neck veins.
“If you don’t see high neck veins due to elevated jugular venous pressure with rapid x and y descents, the patient doesn’t have constrictive pericarditis, no matter what the echocardiogram shows,” Dr. Nishimura asserted.
If those findings are present, however, then on 2-D echocardiography he’s looking for three things that point to constrictive pericarditis: a brisk septal shudder due to rapid filling in early diastole with every heart beat; an early diastolic posterior motion of the intraventricular septum, known as the septal bounce, that occurs as a consequence of the less compliant ventricular walls; and dilation of the inferior vena cava indicative of increased right atrial pressure.
When all three findings are present on 2-D echo, he turns to Doppler echo for hemodynamic information. If Doppler shows a reduction in transmitral driving pressure from the lungs to the heart during inspiration, as the intrathoracic pressure drops but the left ventricular pressure does not, the work-up is done. That patient has constrictive pericarditis and needs to be referred to surgery for pericardiectomy, which will bring rapid improvement.
In roughly one-quarter of patients with constrictive pericarditis, however, that full constellation of 2-D and Doppler echocardiographic findings isn’t present. It then becomes necessary to move on to cardiac catheterization. The first two things to look for in the cath lab are elevated end-equalization of diastolic pressures and low cardiac output.
“If they’re in the cath lab and they’ve got normal filling pressures and a normal cardiac output, they do not have clinically significant constrictive pericarditis, no matter what the echo shows. So those two things are necessary to see, but of course they’re not diagnostic. So we go further,” Dr. Nishimura said.
A patient with constrictive pericarditis will have enhanced ventricular interaction arising from the restraint imposed by a rigid, diseased pericardium. That’s crucial. This increased ventricular interaction is manifest as an increase in the size of the right ventricle during inspiration while the area of the left ventricle is getting smaller.
In contrast, during inspiration and expiration in a patient with restrictive cardiomyopathy, as the right ventricle gets smaller, so does the left ventricle.
“The ratio of the right ventricle to left ventricle area under the curve during inspiration versus expiration gives a very nice distinction between constrictive pericarditis and restrictive cardiomyopathy. Enhanced ventricular interaction is the most sensitive and specific finding for constrictive pericarditis,” according to Dr. Nishimura.
He added that, in addition to constrictive pericarditis and restrictive cardiomyopathy, there is a third and underappreciated cause of right heart failure with a normal ejection fraction: severe tricuspid regurgitation. This abnormality may not be readily apparent upon echocardiography in a patient with a pacemaker lead or automatic implantable cardioverter-defibrillator lead, which can cause acoustic shadowing that results in underestimation of the severity of tricuspid regurgitation. The clue here is the observation of hepatic vein systolic flow reversal, which can only be caused by severe tricuspid regurgitation.
“Think tricuspid regurgitation in patients who have a pacemaker lead or AICD, and also in older women with longstanding atrial fibrillation who dilate their tricuspid annulus and develop more and more tricuspid regurgitation. Take those patients to the cath lab and do a right ventriculogram, which will show tricuspid regurgitation,” Dr. Nishimura advised.
He reported having no financial conflicts.
SNOWMASS, COLO. – Differentiating constrictive pericarditis from restrictive cardiomyopathy in patients with right heart failure with a normal ejection fraction is “one of the most difficult diagnostic challenges in cardiology today,” but reliable results can be achieved using a careful step-by-step approach, Dr. Rick A. Nishimura said at the Annual Cardiovascular Conference at Snowmass.
Under current terminology for heart failure, cardiologists speak of HFrEF, or heart failure with reduced ejection fraction, and HFpEF, or heart failure with preserved ejection fraction. But there is a third group of patients who are often mistakenly thought to have HFpEF: those with severe right heart failure and a normal ejection fraction, classically caused by constrictive pericarditis or restrictive cardiomyopathy.
Unlike patients with HFpEF, these people are not hypertensive and they don’t have pulmonary congestion. Instead, they present predominantly with ascites, peripheral edema, fatigue, and marked elevation in jugular venous pressure, noted Dr. Nishimura, professor of medicine at the Mayo Clinic in Rochester, Minn.
Before elaborating on his own tried-and-true, step-by-step approach, he highlighted several diagnostic procedures he considers less than reliable. One is advanced imaging with CT or MRI looking for the pericardial thickening that is widely viewed as an anatomic hallmark of constrictive pericarditis.
“Remember, 22% of patients with proven constrictive pericarditis actually have a normal pericardium on CT or MRI, because it’s their fibrotic epicardium that’s causing the constrictive pericarditis. And roughly 70% of patients are going to have some thickened pericardium after radiation therapy or coronary artery bypass graft surgery without having constrictive pericarditis. So CT and MRI are helpful, but they’re not going to be diagnostic,” according to the cardiologist.
Similarly, while it’s often been said that constrictive pericarditis can be diagnosed based upon a classic trio of hemodynamic findings obtained through heart catheterization – namely, early rapid filling with a left ventricular end-diastolic pressure equal to the right ventricular end-diastolic pressure, a right ventricular end-diastolic pressure greater than one-third of the right ventricular systolic pressure, and a pulmonary artery pressure below 50 mm Hg – these criteria didn’t reliably separate the last 100 patients who came to the catheterization lab at the Mayo Clinic with either constrictive pericarditis or restrictive cardiomyopathy, he continued.
Dr. Nishimura’s approach to the work-up of patients with unexplained right heart failure and a normal ejection begins with the history and physical examination. The history in a patient with constrictive pericarditis is classically one of radiation therapy years earlier for a malignancy, or prior CABG surgery. And the physical exam has to reveal the presence of high neck veins.
“If you don’t see high neck veins due to elevated jugular venous pressure with rapid x and y descents, the patient doesn’t have constrictive pericarditis, no matter what the echocardiogram shows,” Dr. Nishimura asserted.
If those findings are present, however, then on 2-D echocardiography he’s looking for three things that point to constrictive pericarditis: a brisk septal shudder due to rapid filling in early diastole with every heart beat; an early diastolic posterior motion of the intraventricular septum, known as the septal bounce, that occurs as a consequence of the less compliant ventricular walls; and dilation of the inferior vena cava indicative of increased right atrial pressure.
When all three findings are present on 2-D echo, he turns to Doppler echo for hemodynamic information. If Doppler shows a reduction in transmitral driving pressure from the lungs to the heart during inspiration, as the intrathoracic pressure drops but the left ventricular pressure does not, the work-up is done. That patient has constrictive pericarditis and needs to be referred to surgery for pericardiectomy, which will bring rapid improvement.
In roughly one-quarter of patients with constrictive pericarditis, however, that full constellation of 2-D and Doppler echocardiographic findings isn’t present. It then becomes necessary to move on to cardiac catheterization. The first two things to look for in the cath lab are elevated end-equalization of diastolic pressures and low cardiac output.
“If they’re in the cath lab and they’ve got normal filling pressures and a normal cardiac output, they do not have clinically significant constrictive pericarditis, no matter what the echo shows. So those two things are necessary to see, but of course they’re not diagnostic. So we go further,” Dr. Nishimura said.
A patient with constrictive pericarditis will have enhanced ventricular interaction arising from the restraint imposed by a rigid, diseased pericardium. That’s crucial. This increased ventricular interaction is manifest as an increase in the size of the right ventricle during inspiration while the area of the left ventricle is getting smaller.
In contrast, during inspiration and expiration in a patient with restrictive cardiomyopathy, as the right ventricle gets smaller, so does the left ventricle.
“The ratio of the right ventricle to left ventricle area under the curve during inspiration versus expiration gives a very nice distinction between constrictive pericarditis and restrictive cardiomyopathy. Enhanced ventricular interaction is the most sensitive and specific finding for constrictive pericarditis,” according to Dr. Nishimura.
He added that, in addition to constrictive pericarditis and restrictive cardiomyopathy, there is a third and underappreciated cause of right heart failure with a normal ejection fraction: severe tricuspid regurgitation. This abnormality may not be readily apparent upon echocardiography in a patient with a pacemaker lead or automatic implantable cardioverter-defibrillator lead, which can cause acoustic shadowing that results in underestimation of the severity of tricuspid regurgitation. The clue here is the observation of hepatic vein systolic flow reversal, which can only be caused by severe tricuspid regurgitation.
“Think tricuspid regurgitation in patients who have a pacemaker lead or AICD, and also in older women with longstanding atrial fibrillation who dilate their tricuspid annulus and develop more and more tricuspid regurgitation. Take those patients to the cath lab and do a right ventriculogram, which will show tricuspid regurgitation,” Dr. Nishimura advised.
He reported having no financial conflicts.
SNOWMASS, COLO. – Differentiating constrictive pericarditis from restrictive cardiomyopathy in patients with right heart failure with a normal ejection fraction is “one of the most difficult diagnostic challenges in cardiology today,” but reliable results can be achieved using a careful step-by-step approach, Dr. Rick A. Nishimura said at the Annual Cardiovascular Conference at Snowmass.
Under current terminology for heart failure, cardiologists speak of HFrEF, or heart failure with reduced ejection fraction, and HFpEF, or heart failure with preserved ejection fraction. But there is a third group of patients who are often mistakenly thought to have HFpEF: those with severe right heart failure and a normal ejection fraction, classically caused by constrictive pericarditis or restrictive cardiomyopathy.
Unlike patients with HFpEF, these people are not hypertensive and they don’t have pulmonary congestion. Instead, they present predominantly with ascites, peripheral edema, fatigue, and marked elevation in jugular venous pressure, noted Dr. Nishimura, professor of medicine at the Mayo Clinic in Rochester, Minn.
Before elaborating on his own tried-and-true, step-by-step approach, he highlighted several diagnostic procedures he considers less than reliable. One is advanced imaging with CT or MRI looking for the pericardial thickening that is widely viewed as an anatomic hallmark of constrictive pericarditis.
“Remember, 22% of patients with proven constrictive pericarditis actually have a normal pericardium on CT or MRI, because it’s their fibrotic epicardium that’s causing the constrictive pericarditis. And roughly 70% of patients are going to have some thickened pericardium after radiation therapy or coronary artery bypass graft surgery without having constrictive pericarditis. So CT and MRI are helpful, but they’re not going to be diagnostic,” according to the cardiologist.
Similarly, while it’s often been said that constrictive pericarditis can be diagnosed based upon a classic trio of hemodynamic findings obtained through heart catheterization – namely, early rapid filling with a left ventricular end-diastolic pressure equal to the right ventricular end-diastolic pressure, a right ventricular end-diastolic pressure greater than one-third of the right ventricular systolic pressure, and a pulmonary artery pressure below 50 mm Hg – these criteria didn’t reliably separate the last 100 patients who came to the catheterization lab at the Mayo Clinic with either constrictive pericarditis or restrictive cardiomyopathy, he continued.
Dr. Nishimura’s approach to the work-up of patients with unexplained right heart failure and a normal ejection begins with the history and physical examination. The history in a patient with constrictive pericarditis is classically one of radiation therapy years earlier for a malignancy, or prior CABG surgery. And the physical exam has to reveal the presence of high neck veins.
“If you don’t see high neck veins due to elevated jugular venous pressure with rapid x and y descents, the patient doesn’t have constrictive pericarditis, no matter what the echocardiogram shows,” Dr. Nishimura asserted.
If those findings are present, however, then on 2-D echocardiography he’s looking for three things that point to constrictive pericarditis: a brisk septal shudder due to rapid filling in early diastole with every heart beat; an early diastolic posterior motion of the intraventricular septum, known as the septal bounce, that occurs as a consequence of the less compliant ventricular walls; and dilation of the inferior vena cava indicative of increased right atrial pressure.
When all three findings are present on 2-D echo, he turns to Doppler echo for hemodynamic information. If Doppler shows a reduction in transmitral driving pressure from the lungs to the heart during inspiration, as the intrathoracic pressure drops but the left ventricular pressure does not, the work-up is done. That patient has constrictive pericarditis and needs to be referred to surgery for pericardiectomy, which will bring rapid improvement.
In roughly one-quarter of patients with constrictive pericarditis, however, that full constellation of 2-D and Doppler echocardiographic findings isn’t present. It then becomes necessary to move on to cardiac catheterization. The first two things to look for in the cath lab are elevated end-equalization of diastolic pressures and low cardiac output.
“If they’re in the cath lab and they’ve got normal filling pressures and a normal cardiac output, they do not have clinically significant constrictive pericarditis, no matter what the echo shows. So those two things are necessary to see, but of course they’re not diagnostic. So we go further,” Dr. Nishimura said.
A patient with constrictive pericarditis will have enhanced ventricular interaction arising from the restraint imposed by a rigid, diseased pericardium. That’s crucial. This increased ventricular interaction is manifest as an increase in the size of the right ventricle during inspiration while the area of the left ventricle is getting smaller.
In contrast, during inspiration and expiration in a patient with restrictive cardiomyopathy, as the right ventricle gets smaller, so does the left ventricle.
“The ratio of the right ventricle to left ventricle area under the curve during inspiration versus expiration gives a very nice distinction between constrictive pericarditis and restrictive cardiomyopathy. Enhanced ventricular interaction is the most sensitive and specific finding for constrictive pericarditis,” according to Dr. Nishimura.
He added that, in addition to constrictive pericarditis and restrictive cardiomyopathy, there is a third and underappreciated cause of right heart failure with a normal ejection fraction: severe tricuspid regurgitation. This abnormality may not be readily apparent upon echocardiography in a patient with a pacemaker lead or automatic implantable cardioverter-defibrillator lead, which can cause acoustic shadowing that results in underestimation of the severity of tricuspid regurgitation. The clue here is the observation of hepatic vein systolic flow reversal, which can only be caused by severe tricuspid regurgitation.
“Think tricuspid regurgitation in patients who have a pacemaker lead or AICD, and also in older women with longstanding atrial fibrillation who dilate their tricuspid annulus and develop more and more tricuspid regurgitation. Take those patients to the cath lab and do a right ventriculogram, which will show tricuspid regurgitation,” Dr. Nishimura advised.
He reported having no financial conflicts.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
ICD benefit persists with increasing patient age
The survival benefit conferred by implantable cardioverter-defibrillators (ICDs) persists with increasing patient age, but does attenuate, according to a report published online Feb.10 in Circulation: Cardiovascular Quality and Outcomes.
This indicates that “age per se should not be a contraindication to ICD placement. Rather, clinical judgment should take into account other factors, including patient preference, periprocedural risk, and comorbidity burden,” said Dr. Paul L. Hess of Duke Clinical Research Institute, Durham N.C., and his associates.
It is uncertain whether ICDs are effective at reducing sudden cardiac death in patients aged 75 years and older, largely because this age group has been underrepresented in clinical trials. Yet more than 40% of new ICDs are placed in older patients, including 10% placed in patients aged 80 years and older. Dr. Hess and his colleagues pooled data from five major clinical trials of ICDs to increase the sample size of older patients, allowing better appraisal of treatment effects in this age group. They retrospectively assessed survival outcomes in 3,530 patients who participated in the MADIT-I, MUSTT, MADIT-II, DEFINITE, and SCD-HeFT studies conducted in the United States, Canada, Italy, Germany, the Netherlands, and Israel.
The median age in this pooled sample was 62 years. There were 390 patients (11%) aged 75 years and older. Median follow-up after ICD placement was 2.6 years.
The primary outcome of interest – all-cause mortality – was 21.3% among ICD recipients, compared with 30.6% in patients who didn’t receive ICDs. “After adjusting for patient demographics, medical comorbidities, and laboratory values, point estimates indicated that the survival benefit of ICD therapy persists across the age spectrum,” the investigators said (Circ. Cardiovasc. Qual. Outcomes 2015 Feb. 10; [doi:10.1161/circoutcomes.114.001306]).
The survival benefit attenuated with increasing patient age. This may be because of an increase in competing causes of death as patients aged. However, the total number of participants aged 75 years and older was “modest” despite the pooling of data, “and this may have affected the corresponding efficacy estimates and the observed attenuation of ICD survival benefit,” Dr. Hess and his associates wrote.
The study also found no evidence that age influences the risk of rehospitalization after ICD placement.
The survival benefit conferred by implantable cardioverter-defibrillators (ICDs) persists with increasing patient age, but does attenuate, according to a report published online Feb.10 in Circulation: Cardiovascular Quality and Outcomes.
This indicates that “age per se should not be a contraindication to ICD placement. Rather, clinical judgment should take into account other factors, including patient preference, periprocedural risk, and comorbidity burden,” said Dr. Paul L. Hess of Duke Clinical Research Institute, Durham N.C., and his associates.
It is uncertain whether ICDs are effective at reducing sudden cardiac death in patients aged 75 years and older, largely because this age group has been underrepresented in clinical trials. Yet more than 40% of new ICDs are placed in older patients, including 10% placed in patients aged 80 years and older. Dr. Hess and his colleagues pooled data from five major clinical trials of ICDs to increase the sample size of older patients, allowing better appraisal of treatment effects in this age group. They retrospectively assessed survival outcomes in 3,530 patients who participated in the MADIT-I, MUSTT, MADIT-II, DEFINITE, and SCD-HeFT studies conducted in the United States, Canada, Italy, Germany, the Netherlands, and Israel.
The median age in this pooled sample was 62 years. There were 390 patients (11%) aged 75 years and older. Median follow-up after ICD placement was 2.6 years.
The primary outcome of interest – all-cause mortality – was 21.3% among ICD recipients, compared with 30.6% in patients who didn’t receive ICDs. “After adjusting for patient demographics, medical comorbidities, and laboratory values, point estimates indicated that the survival benefit of ICD therapy persists across the age spectrum,” the investigators said (Circ. Cardiovasc. Qual. Outcomes 2015 Feb. 10; [doi:10.1161/circoutcomes.114.001306]).
The survival benefit attenuated with increasing patient age. This may be because of an increase in competing causes of death as patients aged. However, the total number of participants aged 75 years and older was “modest” despite the pooling of data, “and this may have affected the corresponding efficacy estimates and the observed attenuation of ICD survival benefit,” Dr. Hess and his associates wrote.
The study also found no evidence that age influences the risk of rehospitalization after ICD placement.
The survival benefit conferred by implantable cardioverter-defibrillators (ICDs) persists with increasing patient age, but does attenuate, according to a report published online Feb.10 in Circulation: Cardiovascular Quality and Outcomes.
This indicates that “age per se should not be a contraindication to ICD placement. Rather, clinical judgment should take into account other factors, including patient preference, periprocedural risk, and comorbidity burden,” said Dr. Paul L. Hess of Duke Clinical Research Institute, Durham N.C., and his associates.
It is uncertain whether ICDs are effective at reducing sudden cardiac death in patients aged 75 years and older, largely because this age group has been underrepresented in clinical trials. Yet more than 40% of new ICDs are placed in older patients, including 10% placed in patients aged 80 years and older. Dr. Hess and his colleagues pooled data from five major clinical trials of ICDs to increase the sample size of older patients, allowing better appraisal of treatment effects in this age group. They retrospectively assessed survival outcomes in 3,530 patients who participated in the MADIT-I, MUSTT, MADIT-II, DEFINITE, and SCD-HeFT studies conducted in the United States, Canada, Italy, Germany, the Netherlands, and Israel.
The median age in this pooled sample was 62 years. There were 390 patients (11%) aged 75 years and older. Median follow-up after ICD placement was 2.6 years.
The primary outcome of interest – all-cause mortality – was 21.3% among ICD recipients, compared with 30.6% in patients who didn’t receive ICDs. “After adjusting for patient demographics, medical comorbidities, and laboratory values, point estimates indicated that the survival benefit of ICD therapy persists across the age spectrum,” the investigators said (Circ. Cardiovasc. Qual. Outcomes 2015 Feb. 10; [doi:10.1161/circoutcomes.114.001306]).
The survival benefit attenuated with increasing patient age. This may be because of an increase in competing causes of death as patients aged. However, the total number of participants aged 75 years and older was “modest” despite the pooling of data, “and this may have affected the corresponding efficacy estimates and the observed attenuation of ICD survival benefit,” Dr. Hess and his associates wrote.
The study also found no evidence that age influences the risk of rehospitalization after ICD placement.
FROM CIRCULATION: CARDIOVASCULAR QUALITY AND OUTCOMES
Key clinical point: The survival benefit of ICDs persists with increasing patient age, but attenuates.
Major finding: All-cause mortality was 21.3% among ICD recipients, compared with 30.6% among patients who didn’t receive ICDs.
Data source: A pooled-data analysis from five major ICD clinical trials in 3,530 patients aged 18-80 years who were followed for a median of 2.6 years after ICD implantation.
Disclosures: This study was primarily funded by the Agency for Healthcare Research and Quality. Dr. Hess and one of his associates reported receiving consulting fees and honoraria from Sanofi-Aventis.
HFpEF: Time for a new approach
SNOWMASS, COLO. – The plethora of comorbidities typically present in patients with heart failure with preserved ejection fraction is increasingly thought to be a key driver of the cardiac structural remodeling and poor clinical outcomes characteristic of this increasingly common condition.
“The new message is that even though HFpHF [heart failure with preserved ejection fraction] is a problem of the heart involving diastolic filling and structural remodeling of the ventricle, it’s also a problem of factors outside the heart. Outcomes are driven not just by the cardiac abnormalities, but by the comorbidities that are so common in this elderly population,” Dr. Akshay S. Desai said at the annual Cardiovascular Conference at Snowmass.
“The evolving theoretical model is one that emphasizes the role these comorbidities play, not just in remodeling of the heart, but also in microvascular inflammation, with its consequences for inflammatory cell migration, transforming growth factor–beta activation, myocardial fibrosis, oxidative stress, endothelial inflammation, and downstream impairment of cyclic guanosine monophosphate signaling,” explained Dr. Desai of Brigham and Women’s Hospital, Boston.
He credited Dr. Walter J. Paulus of the Institute for Cardiovascular Research at VU University Medical Center, Amsterdam, as being the primary developer of the new paradigm, which veers away from the traditional emphasis upon excessive afterload as the primary driver of diastolic dysfunction.
As elaborated in detail by Dr. Paulus, the noncardiac comorbidities that are so highly prevalent in HFpEF – especially obesity, diabetes, chronic obstructive pulmonary disease, hypertension, chronic kidney disease, and anemia – induce a systemic inflammatory state which promotes diastolic left ventricular stiffness, cardiac hypertrophy, and the development of heart failure. Dr. Paulus has buttressed his theoretical framework with endomyocardial biopsy studies that document abnormal myocyte structure and function (J. Am. Coll. Cardiol. 2013;62:263-71).
Dr. Desai said the fresh perspective provided by Dr. Paulus is most welcome because it is easily tested, and also because it points to new pathways for treatment. New therapeutic targets are needed desperately because of the striking lack of progress to date in treatment of HFpEF. No drug has been convincingly shown effective in reducing the morbidity and mortality of HFpEF, although a secondary analysis of the flawed TOPCAT trial did strongly suggest spironolactone may reduce the risks of mortality and heart failure hospitalizations (Circulation 2015;131:34-42).
Clinical trials that are now planned or underway as a consequence of the new HFpEF paradigm are investigating novel treatment strategies targeting low myocardial nitric oxide bioavailability and endothelial dysfunction. Agents under study include statins, interleukin-1 receptor antagonists, oral nitrates aimed at boosting cellular levels of nitric oxide, the oral soluble guanylate cyclase stimulator riociguat (Adempas), and the phosphodiesterase-5 inhibitor sildenafil.
Also, all eyes are on the 4,300 patient, 37-country, phase III, randomized PARAGON HF study which began last summer. PARAGON is evaluating LCZ696, the combined angiotensin receptor neprilysin inhibitor that scored a smashing success in heart failure with reduced ejection fraction in the landmark PARADIGM-HF study (N. Engl. J. Med. 2014;371:993-1004).
The rising prevalence of diabetes, obesity, and other proinflammatory chronic conditions could help explain the increasing proportion of patients with heart failure who have HFpEF.
“Depending on where you draw the cut point for preserved ejection fraction, you could say half or as many as 60% of patients hospitalized for decompensated heart failure do so in the setting of preserved ejection fraction,” Dr. Desai observed.
It’s noteworthy that the trajectory of decline following hospitalization for heart failure is similar in HFpEF and heart failure with reduced ejection fraction.
While awaiting the outcome of clinical trials of novel treatments, and with so little evidence-based therapy available at this point, physicians should redouble their efforts to aggressively manage hypertension and other comorbidities in an effort to prevent HFpEF or slow its progression. The favorable TOPCAT results in the Western Hemisphere are also worthy of consideration, the cardiologist argued.
Dr. Desai reported serving as a consultant to 5AM Ventures, AtCor Medical, Novartis, and St. Jude Medical.
SNOWMASS, COLO. – The plethora of comorbidities typically present in patients with heart failure with preserved ejection fraction is increasingly thought to be a key driver of the cardiac structural remodeling and poor clinical outcomes characteristic of this increasingly common condition.
“The new message is that even though HFpHF [heart failure with preserved ejection fraction] is a problem of the heart involving diastolic filling and structural remodeling of the ventricle, it’s also a problem of factors outside the heart. Outcomes are driven not just by the cardiac abnormalities, but by the comorbidities that are so common in this elderly population,” Dr. Akshay S. Desai said at the annual Cardiovascular Conference at Snowmass.
“The evolving theoretical model is one that emphasizes the role these comorbidities play, not just in remodeling of the heart, but also in microvascular inflammation, with its consequences for inflammatory cell migration, transforming growth factor–beta activation, myocardial fibrosis, oxidative stress, endothelial inflammation, and downstream impairment of cyclic guanosine monophosphate signaling,” explained Dr. Desai of Brigham and Women’s Hospital, Boston.
He credited Dr. Walter J. Paulus of the Institute for Cardiovascular Research at VU University Medical Center, Amsterdam, as being the primary developer of the new paradigm, which veers away from the traditional emphasis upon excessive afterload as the primary driver of diastolic dysfunction.
As elaborated in detail by Dr. Paulus, the noncardiac comorbidities that are so highly prevalent in HFpEF – especially obesity, diabetes, chronic obstructive pulmonary disease, hypertension, chronic kidney disease, and anemia – induce a systemic inflammatory state which promotes diastolic left ventricular stiffness, cardiac hypertrophy, and the development of heart failure. Dr. Paulus has buttressed his theoretical framework with endomyocardial biopsy studies that document abnormal myocyte structure and function (J. Am. Coll. Cardiol. 2013;62:263-71).
Dr. Desai said the fresh perspective provided by Dr. Paulus is most welcome because it is easily tested, and also because it points to new pathways for treatment. New therapeutic targets are needed desperately because of the striking lack of progress to date in treatment of HFpEF. No drug has been convincingly shown effective in reducing the morbidity and mortality of HFpEF, although a secondary analysis of the flawed TOPCAT trial did strongly suggest spironolactone may reduce the risks of mortality and heart failure hospitalizations (Circulation 2015;131:34-42).
Clinical trials that are now planned or underway as a consequence of the new HFpEF paradigm are investigating novel treatment strategies targeting low myocardial nitric oxide bioavailability and endothelial dysfunction. Agents under study include statins, interleukin-1 receptor antagonists, oral nitrates aimed at boosting cellular levels of nitric oxide, the oral soluble guanylate cyclase stimulator riociguat (Adempas), and the phosphodiesterase-5 inhibitor sildenafil.
Also, all eyes are on the 4,300 patient, 37-country, phase III, randomized PARAGON HF study which began last summer. PARAGON is evaluating LCZ696, the combined angiotensin receptor neprilysin inhibitor that scored a smashing success in heart failure with reduced ejection fraction in the landmark PARADIGM-HF study (N. Engl. J. Med. 2014;371:993-1004).
The rising prevalence of diabetes, obesity, and other proinflammatory chronic conditions could help explain the increasing proportion of patients with heart failure who have HFpEF.
“Depending on where you draw the cut point for preserved ejection fraction, you could say half or as many as 60% of patients hospitalized for decompensated heart failure do so in the setting of preserved ejection fraction,” Dr. Desai observed.
It’s noteworthy that the trajectory of decline following hospitalization for heart failure is similar in HFpEF and heart failure with reduced ejection fraction.
While awaiting the outcome of clinical trials of novel treatments, and with so little evidence-based therapy available at this point, physicians should redouble their efforts to aggressively manage hypertension and other comorbidities in an effort to prevent HFpEF or slow its progression. The favorable TOPCAT results in the Western Hemisphere are also worthy of consideration, the cardiologist argued.
Dr. Desai reported serving as a consultant to 5AM Ventures, AtCor Medical, Novartis, and St. Jude Medical.
SNOWMASS, COLO. – The plethora of comorbidities typically present in patients with heart failure with preserved ejection fraction is increasingly thought to be a key driver of the cardiac structural remodeling and poor clinical outcomes characteristic of this increasingly common condition.
“The new message is that even though HFpHF [heart failure with preserved ejection fraction] is a problem of the heart involving diastolic filling and structural remodeling of the ventricle, it’s also a problem of factors outside the heart. Outcomes are driven not just by the cardiac abnormalities, but by the comorbidities that are so common in this elderly population,” Dr. Akshay S. Desai said at the annual Cardiovascular Conference at Snowmass.
“The evolving theoretical model is one that emphasizes the role these comorbidities play, not just in remodeling of the heart, but also in microvascular inflammation, with its consequences for inflammatory cell migration, transforming growth factor–beta activation, myocardial fibrosis, oxidative stress, endothelial inflammation, and downstream impairment of cyclic guanosine monophosphate signaling,” explained Dr. Desai of Brigham and Women’s Hospital, Boston.
He credited Dr. Walter J. Paulus of the Institute for Cardiovascular Research at VU University Medical Center, Amsterdam, as being the primary developer of the new paradigm, which veers away from the traditional emphasis upon excessive afterload as the primary driver of diastolic dysfunction.
As elaborated in detail by Dr. Paulus, the noncardiac comorbidities that are so highly prevalent in HFpEF – especially obesity, diabetes, chronic obstructive pulmonary disease, hypertension, chronic kidney disease, and anemia – induce a systemic inflammatory state which promotes diastolic left ventricular stiffness, cardiac hypertrophy, and the development of heart failure. Dr. Paulus has buttressed his theoretical framework with endomyocardial biopsy studies that document abnormal myocyte structure and function (J. Am. Coll. Cardiol. 2013;62:263-71).
Dr. Desai said the fresh perspective provided by Dr. Paulus is most welcome because it is easily tested, and also because it points to new pathways for treatment. New therapeutic targets are needed desperately because of the striking lack of progress to date in treatment of HFpEF. No drug has been convincingly shown effective in reducing the morbidity and mortality of HFpEF, although a secondary analysis of the flawed TOPCAT trial did strongly suggest spironolactone may reduce the risks of mortality and heart failure hospitalizations (Circulation 2015;131:34-42).
Clinical trials that are now planned or underway as a consequence of the new HFpEF paradigm are investigating novel treatment strategies targeting low myocardial nitric oxide bioavailability and endothelial dysfunction. Agents under study include statins, interleukin-1 receptor antagonists, oral nitrates aimed at boosting cellular levels of nitric oxide, the oral soluble guanylate cyclase stimulator riociguat (Adempas), and the phosphodiesterase-5 inhibitor sildenafil.
Also, all eyes are on the 4,300 patient, 37-country, phase III, randomized PARAGON HF study which began last summer. PARAGON is evaluating LCZ696, the combined angiotensin receptor neprilysin inhibitor that scored a smashing success in heart failure with reduced ejection fraction in the landmark PARADIGM-HF study (N. Engl. J. Med. 2014;371:993-1004).
The rising prevalence of diabetes, obesity, and other proinflammatory chronic conditions could help explain the increasing proportion of patients with heart failure who have HFpEF.
“Depending on where you draw the cut point for preserved ejection fraction, you could say half or as many as 60% of patients hospitalized for decompensated heart failure do so in the setting of preserved ejection fraction,” Dr. Desai observed.
It’s noteworthy that the trajectory of decline following hospitalization for heart failure is similar in HFpEF and heart failure with reduced ejection fraction.
While awaiting the outcome of clinical trials of novel treatments, and with so little evidence-based therapy available at this point, physicians should redouble their efforts to aggressively manage hypertension and other comorbidities in an effort to prevent HFpEF or slow its progression. The favorable TOPCAT results in the Western Hemisphere are also worthy of consideration, the cardiologist argued.
Dr. Desai reported serving as a consultant to 5AM Ventures, AtCor Medical, Novartis, and St. Jude Medical.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
IV fluid use in heart failure patients raises concerns
Intravenous fluids are frequently administered during early hospitalization in patients with acute heart failure who are receiving loop diuretics, and the practice is associated with worse outcomes, according to findings from a retrospective cohort study.
Of 131,430 hospitalizations for heart failure at 346 hospitals from 2009 to 2010, 13,806 (11%) involved administration of at least 500 mL of IV fluids during the first 2 days in patients on diuretics. Normal saline was used most often (80% of cases), followed by half-normal saline (12%), and the median administered volume was 1,000 mL.
Those who received fluids were significantly more likely than were those who did not to have subsequent critical care admission (odds ratio, 1.57), intubation (OR, 1.46), renal replacement therapy (OR, 2.04), and hospital death (OR, 2.02), reported Dr. Behnood Bikdeli of Yale New Haven (Conn.) Hospital, and colleagues. The report was published Feb. 2 in the Journal of the American College of Cardiology.
Wide variation was seen in the proportion of hospitalizations that used fluid treatment (range of 0% to 71%), the researchers noted (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.09.007]).
The use of IV fluids in heart failure patients on diuretics may be inadvertent given that “guidelines generally suggest fluid restriction for patients with heart failure and do not generally recommend intravenous fluid therapy,” the investigators noted, adding that the findings highlight an opportunity for improvement.
The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Ct. Dr. Bikdeli reported having no disclosures.
The finding by Bikdeli et al. that fluid administration is common in patients admitted to the hospital with heart failure points to a path forward with respect to improving acute care, according to Dr. Larry A. Allen.
“Now that this relatively common practice is revealed, it behooves us to better understand exactly why it is happening; this understanding can then guide efforts to extinguish truly inappropriate care,” he wrote (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.11.001]).
Particular attention should be paid to what matters most with respect to heart failure: a thoughtful approach to the control of fluid status, he said, adding that “good care is grounded in physiology, guided by evidence, and tailored to the patient.
“In the end, we deliver an incredible number and diversity of therapies well in the modern U.S. health care system. The pressing question is whether the totality of therapies makes sense in each individual,” he concluded.
Dr. Allen is with the University of Colorado, Aurora, and the Colorado Cardiovascular Outcomes Research Consortium, Denver. He disclosed funding from the National Heart, Lung, and Blood Institute.
The finding by Bikdeli et al. that fluid administration is common in patients admitted to the hospital with heart failure points to a path forward with respect to improving acute care, according to Dr. Larry A. Allen.
“Now that this relatively common practice is revealed, it behooves us to better understand exactly why it is happening; this understanding can then guide efforts to extinguish truly inappropriate care,” he wrote (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.11.001]).
Particular attention should be paid to what matters most with respect to heart failure: a thoughtful approach to the control of fluid status, he said, adding that “good care is grounded in physiology, guided by evidence, and tailored to the patient.
“In the end, we deliver an incredible number and diversity of therapies well in the modern U.S. health care system. The pressing question is whether the totality of therapies makes sense in each individual,” he concluded.
Dr. Allen is with the University of Colorado, Aurora, and the Colorado Cardiovascular Outcomes Research Consortium, Denver. He disclosed funding from the National Heart, Lung, and Blood Institute.
The finding by Bikdeli et al. that fluid administration is common in patients admitted to the hospital with heart failure points to a path forward with respect to improving acute care, according to Dr. Larry A. Allen.
“Now that this relatively common practice is revealed, it behooves us to better understand exactly why it is happening; this understanding can then guide efforts to extinguish truly inappropriate care,” he wrote (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.11.001]).
Particular attention should be paid to what matters most with respect to heart failure: a thoughtful approach to the control of fluid status, he said, adding that “good care is grounded in physiology, guided by evidence, and tailored to the patient.
“In the end, we deliver an incredible number and diversity of therapies well in the modern U.S. health care system. The pressing question is whether the totality of therapies makes sense in each individual,” he concluded.
Dr. Allen is with the University of Colorado, Aurora, and the Colorado Cardiovascular Outcomes Research Consortium, Denver. He disclosed funding from the National Heart, Lung, and Blood Institute.
Intravenous fluids are frequently administered during early hospitalization in patients with acute heart failure who are receiving loop diuretics, and the practice is associated with worse outcomes, according to findings from a retrospective cohort study.
Of 131,430 hospitalizations for heart failure at 346 hospitals from 2009 to 2010, 13,806 (11%) involved administration of at least 500 mL of IV fluids during the first 2 days in patients on diuretics. Normal saline was used most often (80% of cases), followed by half-normal saline (12%), and the median administered volume was 1,000 mL.
Those who received fluids were significantly more likely than were those who did not to have subsequent critical care admission (odds ratio, 1.57), intubation (OR, 1.46), renal replacement therapy (OR, 2.04), and hospital death (OR, 2.02), reported Dr. Behnood Bikdeli of Yale New Haven (Conn.) Hospital, and colleagues. The report was published Feb. 2 in the Journal of the American College of Cardiology.
Wide variation was seen in the proportion of hospitalizations that used fluid treatment (range of 0% to 71%), the researchers noted (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.09.007]).
The use of IV fluids in heart failure patients on diuretics may be inadvertent given that “guidelines generally suggest fluid restriction for patients with heart failure and do not generally recommend intravenous fluid therapy,” the investigators noted, adding that the findings highlight an opportunity for improvement.
The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Ct. Dr. Bikdeli reported having no disclosures.
Intravenous fluids are frequently administered during early hospitalization in patients with acute heart failure who are receiving loop diuretics, and the practice is associated with worse outcomes, according to findings from a retrospective cohort study.
Of 131,430 hospitalizations for heart failure at 346 hospitals from 2009 to 2010, 13,806 (11%) involved administration of at least 500 mL of IV fluids during the first 2 days in patients on diuretics. Normal saline was used most often (80% of cases), followed by half-normal saline (12%), and the median administered volume was 1,000 mL.
Those who received fluids were significantly more likely than were those who did not to have subsequent critical care admission (odds ratio, 1.57), intubation (OR, 1.46), renal replacement therapy (OR, 2.04), and hospital death (OR, 2.02), reported Dr. Behnood Bikdeli of Yale New Haven (Conn.) Hospital, and colleagues. The report was published Feb. 2 in the Journal of the American College of Cardiology.
Wide variation was seen in the proportion of hospitalizations that used fluid treatment (range of 0% to 71%), the researchers noted (J. Am. Coll. Cardiol. HF 2015 Feb. 2 [doi:10.1016/j.jchf.2014.09.007]).
The use of IV fluids in heart failure patients on diuretics may be inadvertent given that “guidelines generally suggest fluid restriction for patients with heart failure and do not generally recommend intravenous fluid therapy,” the investigators noted, adding that the findings highlight an opportunity for improvement.
The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Ct. Dr. Bikdeli reported having no disclosures.
Key clinical point: Patients hospitalized for heart failure may receive IV fluids inappropriately.
Major finding: Eleven percent of heart failure patients on diuretics received IV fluids during the first 2 inpatient days.
Data source: A retrospective cohort study of 131,430 hospitalizations.
Disclosures: The study was funded by two of the National Institutes of Health as well as grants from the Patrick and Catherine Weldon Donaghue Medical Research Foundation in West Hartford, Conn. Dr. Bikdeli reported having no disclosures.
Renal denervation therapy: What’s next
SNOWMASS, COLO. – Reports of the demise of catheter-based renal denervation therapy for resistant hypertension in light of the disappointing SYMPLICITY HTN-3 trial results are greatly exaggerated, according to Dr. Bernard J. Gersh.
“I think what we can say about renal denervation is that the initial enthusiasm has been tempered and the number of unanswered questions is not decreasing. But the concept, I assure you, still maintains its promise,” the cardiologist said at the annual Cardiovascular Conference at Snowmass.
Expectations for SYMPLICITY HTN-3 were sky high in the cardiology and business worlds on the basis of the prior unblinded SYMPLICITY HTN-1 and -2 studies showing renal denervation (RDN) achieved spectacular reductions in systolic blood pressure of 25-30 mm Hg in patients with multidrug-resistant hypertension. As the chair of the SYMPLICITY HTN-3 data safety monitoring board, he knew the outcome prior to release of the results, and he was struck that during that period the talk at cardiology conferences was only of next-generation RDN catheters and expanded indications, such as heart failure and even metabolic syndrome. Absolutely no one seemed to entertain the possibility that the results wouldn’t be positive.
So the general reaction to the SYMPLICITY HTN-3 results was profound, crushing disappointment. The trial, which was the first blinded, controlled study of RDN, showed significant reduction in blood pressure at 6 months in the RDN recipients, but a similar reduction in medically managed controls who underwent a sham RDN procedure, making for a neutral study outcome (N. Engl. J. Med. 2014;370:1393-59).
Yet the physiologic basis remains strong for RDN as a treatment for hypertension, heart failure, and perhaps other cardiovascular disorders in which sympathetic nervous system activation plays a crucial role, according to Dr. Gersh, who chaired the data safety monitoring board for SYMPLICITY HTN-3 and is a professor of medicine at the Mayo Clinic in Rochester, Minn.
He cited a recommended review written by a pioneer in the field, Dr. Murray Esler of Australia, who observed that increased renal sympathetic nervous system activity increases the secretion rate of renin, reduces renal blood flow, and boosts renal tubular sodium reabsorption while reducing urinary sodium excretion, all actions that contribute to hypertension (Exp. Physiol. 2011;96:611-22).
Dr. Gersh ticked off many potential explanations for the neutral outcome in SYMPLICITY HTN-3, including the placebo effect; regression to the mean; operator inexperience; limitations of the current technology; and the likelihood that a fair number of study participants didn’t have true treatment-resistant hypertension, but were merely resistant to taking their medication until they entered a structured, supervised randomized trial setting. The power of a placebo procedure is not to be underestimated, the cardiologist emphasized. He cited a striking illustration from another field of medicine, in which a recent meta-analysis of 137 controlled studies in more than 33,000 patients with knee osteoarthritis showed that intra-articular placebo injections outperformed oral naproxen, celecoxib, and placebo in terms of pain relief (Ann. Intern. Med. 2015;162:46-54).
Regression to the mean definitely occurred in SYMPLICITY HTN-3, as evidenced by the fact that reductions in office systolic blood pressure (SBP) averaged 25.7 and 19.7 mm Hg, respectively, in RDN recipients and controls with a baseline greater than 184 mm Hg, compared with 13.8 and 9.8 mm Hg in those with a lower baseline SBP, of 170-184 mm Hg, Dr. Gersh continued.
Operator inexperience was likely a factor in the study outcomes: Nearly one-third of the operators dad done just one procedure. And while the study protocol called for four to six ablations per side, a recent secondary analysis of SYMPLICITY HTN-3 showed that the greater the number of ablations, the greater the drop in SBP. Patients who received at least seven ablations per side averaged a nearly threefold larger blood pressure reduction at 6 months, compared with those who got five or six per side (Eur. Heart J. 2015;36:219-27).
Some 39% of study participants had a medication change because of side effects during the 6-month trial. This confounds attempts to assess the true impact of RDN. To answer this question, the Food and Drug Administration recently granted Boston Scientific approval to test RDN in a sham-controlled trial in patients with mild to moderate hypertension after a 4-week medication washout period. The 6-month trial is due to start shortly. This is an important study because it will demonstrate whether RDN in its present form actually works.
“Clearly, what we’ve learned in SYMPLICITY HTN-3 is ‘resistant’ hypertension is amenable to expert pharmacologic control. This raises an interesting question: If renal nerve denervation works, could we offer it as an alternative to taking two or three drugs for the next 10 or 20 years? That might be, in certain patient populations, a very attractive option. That’s why we’ve got to show whether this procedure works well,” according to Dr. Gersh.
Now that SYMPLICITY HTN-3 has provided a reality check, the necessary next steps include improving the technology. Current levels of achieved RDN using only proximal ablation are suboptimal. What’s needed are higher-energy, multipolar electrodes that deliver energy both proximally and distally; such equipment is well along in development.
A thornier limitation involves the lack of a method for immediate testing of the completeness of an RDN procedure in a given patient. Unlike in coronary stent placement, where an interventional cardiologist can immediately see angiographically whether the device is properly seated, RDN operators have no way to tell intraprocedurally whether effective RDN has been achieved. Two methods now under investigation are intra-arterial adenosine and urinary biomarkers of nerve degradation.
The pathophysiology of hypertension is variable, so efforts are underway to identify patient subsets in which sympathetic nervous system overactivity is a primary underlying mechanism and RDN should have its greatest impact.
“I think renal denervation is still worthy of investigation, particularly in patients with resistant hypertension and perhaps other disease states characterized by sympathetic overactivity, such as heart failure,” Dr. Gersh concluded. “How do I think it’s going to all turn out? I just don’t know. We’ll see. We need the trials. I don’t think we should close the book on this very exciting technique. I simply don’t know what the trials are going to show.”
SYMPLICITY HTN-3 was funded by Medtronic. Dr. Gersh reported serving as a consultant to Merck and Ortho-McNeil-Janssen and on data safety monitoring boards for trials sponsored by Baxter, Medtronic, and Teva Pharmaceuticals.
SNOWMASS, COLO. – Reports of the demise of catheter-based renal denervation therapy for resistant hypertension in light of the disappointing SYMPLICITY HTN-3 trial results are greatly exaggerated, according to Dr. Bernard J. Gersh.
“I think what we can say about renal denervation is that the initial enthusiasm has been tempered and the number of unanswered questions is not decreasing. But the concept, I assure you, still maintains its promise,” the cardiologist said at the annual Cardiovascular Conference at Snowmass.
Expectations for SYMPLICITY HTN-3 were sky high in the cardiology and business worlds on the basis of the prior unblinded SYMPLICITY HTN-1 and -2 studies showing renal denervation (RDN) achieved spectacular reductions in systolic blood pressure of 25-30 mm Hg in patients with multidrug-resistant hypertension. As the chair of the SYMPLICITY HTN-3 data safety monitoring board, he knew the outcome prior to release of the results, and he was struck that during that period the talk at cardiology conferences was only of next-generation RDN catheters and expanded indications, such as heart failure and even metabolic syndrome. Absolutely no one seemed to entertain the possibility that the results wouldn’t be positive.
So the general reaction to the SYMPLICITY HTN-3 results was profound, crushing disappointment. The trial, which was the first blinded, controlled study of RDN, showed significant reduction in blood pressure at 6 months in the RDN recipients, but a similar reduction in medically managed controls who underwent a sham RDN procedure, making for a neutral study outcome (N. Engl. J. Med. 2014;370:1393-59).
Yet the physiologic basis remains strong for RDN as a treatment for hypertension, heart failure, and perhaps other cardiovascular disorders in which sympathetic nervous system activation plays a crucial role, according to Dr. Gersh, who chaired the data safety monitoring board for SYMPLICITY HTN-3 and is a professor of medicine at the Mayo Clinic in Rochester, Minn.
He cited a recommended review written by a pioneer in the field, Dr. Murray Esler of Australia, who observed that increased renal sympathetic nervous system activity increases the secretion rate of renin, reduces renal blood flow, and boosts renal tubular sodium reabsorption while reducing urinary sodium excretion, all actions that contribute to hypertension (Exp. Physiol. 2011;96:611-22).
Dr. Gersh ticked off many potential explanations for the neutral outcome in SYMPLICITY HTN-3, including the placebo effect; regression to the mean; operator inexperience; limitations of the current technology; and the likelihood that a fair number of study participants didn’t have true treatment-resistant hypertension, but were merely resistant to taking their medication until they entered a structured, supervised randomized trial setting. The power of a placebo procedure is not to be underestimated, the cardiologist emphasized. He cited a striking illustration from another field of medicine, in which a recent meta-analysis of 137 controlled studies in more than 33,000 patients with knee osteoarthritis showed that intra-articular placebo injections outperformed oral naproxen, celecoxib, and placebo in terms of pain relief (Ann. Intern. Med. 2015;162:46-54).
Regression to the mean definitely occurred in SYMPLICITY HTN-3, as evidenced by the fact that reductions in office systolic blood pressure (SBP) averaged 25.7 and 19.7 mm Hg, respectively, in RDN recipients and controls with a baseline greater than 184 mm Hg, compared with 13.8 and 9.8 mm Hg in those with a lower baseline SBP, of 170-184 mm Hg, Dr. Gersh continued.
Operator inexperience was likely a factor in the study outcomes: Nearly one-third of the operators dad done just one procedure. And while the study protocol called for four to six ablations per side, a recent secondary analysis of SYMPLICITY HTN-3 showed that the greater the number of ablations, the greater the drop in SBP. Patients who received at least seven ablations per side averaged a nearly threefold larger blood pressure reduction at 6 months, compared with those who got five or six per side (Eur. Heart J. 2015;36:219-27).
Some 39% of study participants had a medication change because of side effects during the 6-month trial. This confounds attempts to assess the true impact of RDN. To answer this question, the Food and Drug Administration recently granted Boston Scientific approval to test RDN in a sham-controlled trial in patients with mild to moderate hypertension after a 4-week medication washout period. The 6-month trial is due to start shortly. This is an important study because it will demonstrate whether RDN in its present form actually works.
“Clearly, what we’ve learned in SYMPLICITY HTN-3 is ‘resistant’ hypertension is amenable to expert pharmacologic control. This raises an interesting question: If renal nerve denervation works, could we offer it as an alternative to taking two or three drugs for the next 10 or 20 years? That might be, in certain patient populations, a very attractive option. That’s why we’ve got to show whether this procedure works well,” according to Dr. Gersh.
Now that SYMPLICITY HTN-3 has provided a reality check, the necessary next steps include improving the technology. Current levels of achieved RDN using only proximal ablation are suboptimal. What’s needed are higher-energy, multipolar electrodes that deliver energy both proximally and distally; such equipment is well along in development.
A thornier limitation involves the lack of a method for immediate testing of the completeness of an RDN procedure in a given patient. Unlike in coronary stent placement, where an interventional cardiologist can immediately see angiographically whether the device is properly seated, RDN operators have no way to tell intraprocedurally whether effective RDN has been achieved. Two methods now under investigation are intra-arterial adenosine and urinary biomarkers of nerve degradation.
The pathophysiology of hypertension is variable, so efforts are underway to identify patient subsets in which sympathetic nervous system overactivity is a primary underlying mechanism and RDN should have its greatest impact.
“I think renal denervation is still worthy of investigation, particularly in patients with resistant hypertension and perhaps other disease states characterized by sympathetic overactivity, such as heart failure,” Dr. Gersh concluded. “How do I think it’s going to all turn out? I just don’t know. We’ll see. We need the trials. I don’t think we should close the book on this very exciting technique. I simply don’t know what the trials are going to show.”
SYMPLICITY HTN-3 was funded by Medtronic. Dr. Gersh reported serving as a consultant to Merck and Ortho-McNeil-Janssen and on data safety monitoring boards for trials sponsored by Baxter, Medtronic, and Teva Pharmaceuticals.
SNOWMASS, COLO. – Reports of the demise of catheter-based renal denervation therapy for resistant hypertension in light of the disappointing SYMPLICITY HTN-3 trial results are greatly exaggerated, according to Dr. Bernard J. Gersh.
“I think what we can say about renal denervation is that the initial enthusiasm has been tempered and the number of unanswered questions is not decreasing. But the concept, I assure you, still maintains its promise,” the cardiologist said at the annual Cardiovascular Conference at Snowmass.
Expectations for SYMPLICITY HTN-3 were sky high in the cardiology and business worlds on the basis of the prior unblinded SYMPLICITY HTN-1 and -2 studies showing renal denervation (RDN) achieved spectacular reductions in systolic blood pressure of 25-30 mm Hg in patients with multidrug-resistant hypertension. As the chair of the SYMPLICITY HTN-3 data safety monitoring board, he knew the outcome prior to release of the results, and he was struck that during that period the talk at cardiology conferences was only of next-generation RDN catheters and expanded indications, such as heart failure and even metabolic syndrome. Absolutely no one seemed to entertain the possibility that the results wouldn’t be positive.
So the general reaction to the SYMPLICITY HTN-3 results was profound, crushing disappointment. The trial, which was the first blinded, controlled study of RDN, showed significant reduction in blood pressure at 6 months in the RDN recipients, but a similar reduction in medically managed controls who underwent a sham RDN procedure, making for a neutral study outcome (N. Engl. J. Med. 2014;370:1393-59).
Yet the physiologic basis remains strong for RDN as a treatment for hypertension, heart failure, and perhaps other cardiovascular disorders in which sympathetic nervous system activation plays a crucial role, according to Dr. Gersh, who chaired the data safety monitoring board for SYMPLICITY HTN-3 and is a professor of medicine at the Mayo Clinic in Rochester, Minn.
He cited a recommended review written by a pioneer in the field, Dr. Murray Esler of Australia, who observed that increased renal sympathetic nervous system activity increases the secretion rate of renin, reduces renal blood flow, and boosts renal tubular sodium reabsorption while reducing urinary sodium excretion, all actions that contribute to hypertension (Exp. Physiol. 2011;96:611-22).
Dr. Gersh ticked off many potential explanations for the neutral outcome in SYMPLICITY HTN-3, including the placebo effect; regression to the mean; operator inexperience; limitations of the current technology; and the likelihood that a fair number of study participants didn’t have true treatment-resistant hypertension, but were merely resistant to taking their medication until they entered a structured, supervised randomized trial setting. The power of a placebo procedure is not to be underestimated, the cardiologist emphasized. He cited a striking illustration from another field of medicine, in which a recent meta-analysis of 137 controlled studies in more than 33,000 patients with knee osteoarthritis showed that intra-articular placebo injections outperformed oral naproxen, celecoxib, and placebo in terms of pain relief (Ann. Intern. Med. 2015;162:46-54).
Regression to the mean definitely occurred in SYMPLICITY HTN-3, as evidenced by the fact that reductions in office systolic blood pressure (SBP) averaged 25.7 and 19.7 mm Hg, respectively, in RDN recipients and controls with a baseline greater than 184 mm Hg, compared with 13.8 and 9.8 mm Hg in those with a lower baseline SBP, of 170-184 mm Hg, Dr. Gersh continued.
Operator inexperience was likely a factor in the study outcomes: Nearly one-third of the operators dad done just one procedure. And while the study protocol called for four to six ablations per side, a recent secondary analysis of SYMPLICITY HTN-3 showed that the greater the number of ablations, the greater the drop in SBP. Patients who received at least seven ablations per side averaged a nearly threefold larger blood pressure reduction at 6 months, compared with those who got five or six per side (Eur. Heart J. 2015;36:219-27).
Some 39% of study participants had a medication change because of side effects during the 6-month trial. This confounds attempts to assess the true impact of RDN. To answer this question, the Food and Drug Administration recently granted Boston Scientific approval to test RDN in a sham-controlled trial in patients with mild to moderate hypertension after a 4-week medication washout period. The 6-month trial is due to start shortly. This is an important study because it will demonstrate whether RDN in its present form actually works.
“Clearly, what we’ve learned in SYMPLICITY HTN-3 is ‘resistant’ hypertension is amenable to expert pharmacologic control. This raises an interesting question: If renal nerve denervation works, could we offer it as an alternative to taking two or three drugs for the next 10 or 20 years? That might be, in certain patient populations, a very attractive option. That’s why we’ve got to show whether this procedure works well,” according to Dr. Gersh.
Now that SYMPLICITY HTN-3 has provided a reality check, the necessary next steps include improving the technology. Current levels of achieved RDN using only proximal ablation are suboptimal. What’s needed are higher-energy, multipolar electrodes that deliver energy both proximally and distally; such equipment is well along in development.
A thornier limitation involves the lack of a method for immediate testing of the completeness of an RDN procedure in a given patient. Unlike in coronary stent placement, where an interventional cardiologist can immediately see angiographically whether the device is properly seated, RDN operators have no way to tell intraprocedurally whether effective RDN has been achieved. Two methods now under investigation are intra-arterial adenosine and urinary biomarkers of nerve degradation.
The pathophysiology of hypertension is variable, so efforts are underway to identify patient subsets in which sympathetic nervous system overactivity is a primary underlying mechanism and RDN should have its greatest impact.
“I think renal denervation is still worthy of investigation, particularly in patients with resistant hypertension and perhaps other disease states characterized by sympathetic overactivity, such as heart failure,” Dr. Gersh concluded. “How do I think it’s going to all turn out? I just don’t know. We’ll see. We need the trials. I don’t think we should close the book on this very exciting technique. I simply don’t know what the trials are going to show.”
SYMPLICITY HTN-3 was funded by Medtronic. Dr. Gersh reported serving as a consultant to Merck and Ortho-McNeil-Janssen and on data safety monitoring boards for trials sponsored by Baxter, Medtronic, and Teva Pharmaceuticals.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Moderate alcohol consumption can decrease heart failure risk
Although alcohol is known to be a cardiac toxin, consumption of up to seven drinks per week in early to middle age was found to be associated with a decreased risk of future heart failure in a study published by the European Society of Cardiology.
“Although heavy alcohol consumption is associated with impairment in left ventricular function and eventual alcoholic cardiomyopathy with symptomatic HF, moderate alcohol intake could, conversely, lower the risk for HF,” wrote Dr. Alexandra Gonçalves of Brigham and Women’s Hospital, Boston, and her associates. “However, the association between moderate alcohol intake and the risk of heart failure is still controversial, as some studies did not find an association and the cardiovascular mechanisms of potential benefit of alcohol consumption in heart failure are uncertain” (Eur. Heart J. 2014 [doi:10.1093/eurheartj/ehu514]).
From the ongoing, prospective, observational Atherosclerosis Risk in Communities (ARIC) Study, the investigators analyzed 14,629 subjects aged 45-64 years, 55% of whom were female, and 74% of whom were white. Baseline data for all subjects were taken between 1987 and 1989, and no participants reported prevalent heart failure.
Self-reported alcohol consumption was assessed at baseline as the weekly number of drinks, with one drink equaling 14 grams of alcohol, and updated cumulative average alcohol intake was calculated over roughly 9 years. Subjects were placed into one of six groups: former drinkers, abstainers, drinkers of less than 7 drinks per week, drinkers of 7-14 drinks per week, drinkers of 14-21 drinks per week, and those who had 21 or more drinks per week. Using multivariable Cox proportional hazards models, investigators analyzed relationships between alcohol consumption and heart failure and whether those associations were affected in any way by the subjects’ sex.
Results showed that 61% of participants reported consuming no alcohol at all – 19% being former drinkers and 42% abstainers – while 25% of participants said they drank up to 7 drinks per week, 8% between 7 and 14 drinks per week, 3% between 14 and 21 drinks per week, and 3% 21 or more drinks per week. Among former drinkers, there were 376 incident heart failure events in men and 266 in women. Among abstainers, there were 333 for men and 717 for women. Rates per 100 person-years equaled 1.50, 1.12, 1.02, and 0.79, respectively.
For subjects who consumed fewer than seven drinks weekly, there were 281 heart failure incidents among men and 191 among women, for rates per 100 person-years of 0.77 and 0.53, respectively, significantly lower than the rates for abstainers. In the higher drinking categories, no significant differences in heart failure rates, compared with abstainers, were observed.
“We observed that participants who consumed up to 7 drinks/week of alcohol had a lower risk of incident HF compared with abstainers, with a less pronounced association in women than in men,” concluded Dr. Gonçalves and her coinvestigators, adding that they “did not find significant differences between white and black men and women in the risk of HF by alcohol consumption [but] once men and women were stratified by race, the number of cases was relatively small in each category of alcohol intake, limiting our ability to detect small differences by race.”
This study was funded by the National Heart, Lung, and Blood Institute, with support from the Portuguese Foundation for Science and Technology Grant and the Ellison Foundation. The authors declared no relevant financial disclosures.
Although alcohol is known to be a cardiac toxin, consumption of up to seven drinks per week in early to middle age was found to be associated with a decreased risk of future heart failure in a study published by the European Society of Cardiology.
“Although heavy alcohol consumption is associated with impairment in left ventricular function and eventual alcoholic cardiomyopathy with symptomatic HF, moderate alcohol intake could, conversely, lower the risk for HF,” wrote Dr. Alexandra Gonçalves of Brigham and Women’s Hospital, Boston, and her associates. “However, the association between moderate alcohol intake and the risk of heart failure is still controversial, as some studies did not find an association and the cardiovascular mechanisms of potential benefit of alcohol consumption in heart failure are uncertain” (Eur. Heart J. 2014 [doi:10.1093/eurheartj/ehu514]).
From the ongoing, prospective, observational Atherosclerosis Risk in Communities (ARIC) Study, the investigators analyzed 14,629 subjects aged 45-64 years, 55% of whom were female, and 74% of whom were white. Baseline data for all subjects were taken between 1987 and 1989, and no participants reported prevalent heart failure.
Self-reported alcohol consumption was assessed at baseline as the weekly number of drinks, with one drink equaling 14 grams of alcohol, and updated cumulative average alcohol intake was calculated over roughly 9 years. Subjects were placed into one of six groups: former drinkers, abstainers, drinkers of less than 7 drinks per week, drinkers of 7-14 drinks per week, drinkers of 14-21 drinks per week, and those who had 21 or more drinks per week. Using multivariable Cox proportional hazards models, investigators analyzed relationships between alcohol consumption and heart failure and whether those associations were affected in any way by the subjects’ sex.
Results showed that 61% of participants reported consuming no alcohol at all – 19% being former drinkers and 42% abstainers – while 25% of participants said they drank up to 7 drinks per week, 8% between 7 and 14 drinks per week, 3% between 14 and 21 drinks per week, and 3% 21 or more drinks per week. Among former drinkers, there were 376 incident heart failure events in men and 266 in women. Among abstainers, there were 333 for men and 717 for women. Rates per 100 person-years equaled 1.50, 1.12, 1.02, and 0.79, respectively.
For subjects who consumed fewer than seven drinks weekly, there were 281 heart failure incidents among men and 191 among women, for rates per 100 person-years of 0.77 and 0.53, respectively, significantly lower than the rates for abstainers. In the higher drinking categories, no significant differences in heart failure rates, compared with abstainers, were observed.
“We observed that participants who consumed up to 7 drinks/week of alcohol had a lower risk of incident HF compared with abstainers, with a less pronounced association in women than in men,” concluded Dr. Gonçalves and her coinvestigators, adding that they “did not find significant differences between white and black men and women in the risk of HF by alcohol consumption [but] once men and women were stratified by race, the number of cases was relatively small in each category of alcohol intake, limiting our ability to detect small differences by race.”
This study was funded by the National Heart, Lung, and Blood Institute, with support from the Portuguese Foundation for Science and Technology Grant and the Ellison Foundation. The authors declared no relevant financial disclosures.
Although alcohol is known to be a cardiac toxin, consumption of up to seven drinks per week in early to middle age was found to be associated with a decreased risk of future heart failure in a study published by the European Society of Cardiology.
“Although heavy alcohol consumption is associated with impairment in left ventricular function and eventual alcoholic cardiomyopathy with symptomatic HF, moderate alcohol intake could, conversely, lower the risk for HF,” wrote Dr. Alexandra Gonçalves of Brigham and Women’s Hospital, Boston, and her associates. “However, the association between moderate alcohol intake and the risk of heart failure is still controversial, as some studies did not find an association and the cardiovascular mechanisms of potential benefit of alcohol consumption in heart failure are uncertain” (Eur. Heart J. 2014 [doi:10.1093/eurheartj/ehu514]).
From the ongoing, prospective, observational Atherosclerosis Risk in Communities (ARIC) Study, the investigators analyzed 14,629 subjects aged 45-64 years, 55% of whom were female, and 74% of whom were white. Baseline data for all subjects were taken between 1987 and 1989, and no participants reported prevalent heart failure.
Self-reported alcohol consumption was assessed at baseline as the weekly number of drinks, with one drink equaling 14 grams of alcohol, and updated cumulative average alcohol intake was calculated over roughly 9 years. Subjects were placed into one of six groups: former drinkers, abstainers, drinkers of less than 7 drinks per week, drinkers of 7-14 drinks per week, drinkers of 14-21 drinks per week, and those who had 21 or more drinks per week. Using multivariable Cox proportional hazards models, investigators analyzed relationships between alcohol consumption and heart failure and whether those associations were affected in any way by the subjects’ sex.
Results showed that 61% of participants reported consuming no alcohol at all – 19% being former drinkers and 42% abstainers – while 25% of participants said they drank up to 7 drinks per week, 8% between 7 and 14 drinks per week, 3% between 14 and 21 drinks per week, and 3% 21 or more drinks per week. Among former drinkers, there were 376 incident heart failure events in men and 266 in women. Among abstainers, there were 333 for men and 717 for women. Rates per 100 person-years equaled 1.50, 1.12, 1.02, and 0.79, respectively.
For subjects who consumed fewer than seven drinks weekly, there were 281 heart failure incidents among men and 191 among women, for rates per 100 person-years of 0.77 and 0.53, respectively, significantly lower than the rates for abstainers. In the higher drinking categories, no significant differences in heart failure rates, compared with abstainers, were observed.
“We observed that participants who consumed up to 7 drinks/week of alcohol had a lower risk of incident HF compared with abstainers, with a less pronounced association in women than in men,” concluded Dr. Gonçalves and her coinvestigators, adding that they “did not find significant differences between white and black men and women in the risk of HF by alcohol consumption [but] once men and women were stratified by race, the number of cases was relatively small in each category of alcohol intake, limiting our ability to detect small differences by race.”
This study was funded by the National Heart, Lung, and Blood Institute, with support from the Portuguese Foundation for Science and Technology Grant and the Ellison Foundation. The authors declared no relevant financial disclosures.
FROM THE EUROPEAN HEART JOURNAL
Key clinical point: Consumption of up to seven alcoholic beverages per week in early-middle age can lower risk for future heart failure.
Major finding: Among those who consumed up to seven drinks weekly, there were 281 heart failure incidents for men and 191 for women, with rates per 100 person-years of 0.77 and 0.53, respectively (P = .05).
Data source: The biracial, community-based ARIC study, comprising 14,629 adults.
Disclosures: ARIC is funded by the National Heart, Lung, and Blood Institute, with support from the Portuguese Foundation for Science and Technology Grant and the Ellison Foundation. The authors declared no relevant financial disclosures.
First expression of cardiovascular disease differs by gender
CHICAGO – Two out of three 55-year-old men and women will develop some form of cardiovascular disease during their remaining lifetime, but the first manifestation differs considerably by gender, according to a new analysis from The Rotterdam Study.
In men, the first manifestation of cardiovascular disease is most often a coronary heart disease event. Women are more likely to experience cerebrovascular disease or heart failure as their first event, Dr. Maarten J.G. Leening reported at the American Heart Association scientific sessions. Simultaneously with his presentation, the findings were published online in BMJ.
This new finding has important implications for primary prevention, noted Dr. Leening of Erasmus University, Rotterdam.
“Most of the attention of late in primary prevention has been drawn to the 2013 [AHA/ACC] lipid guidelines. But as we can see, for women it’s heart failure and cerebrovascular disease that are the major contributors to overall risk. And we know from the statin trials that there is no or only very limited reduction in heart failure and that the risk reduction for cerebrovascular disease is much smaller than for CHD. So our findings highlight the importance of blood pressure control and lifestyle factors, especially smoking, for primary prevention in women, since those are the best-established risk factors for cerebrovascular disease and heart failure,” he said.
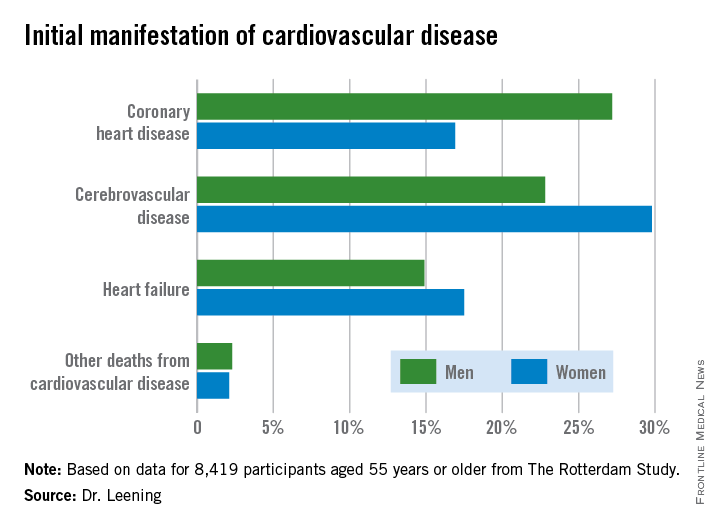
Dr. Leening reported on 8,419 participants of The Rotterdam Study aged 55 or older, all free from cardiovascular disease at baseline. During a median 13.5 years and up to 20 years of follow-up in this ongoing prospective population-based study, 2,888 subjects developed cardiovascular disease. Coronary heart disease accounted for 826 of these first events, cerebrovascular events accounted for another 1,198, heart failure 726, and there were 102 other cardiovascular deaths.
At age 55, the overall lifetime risk of developing cardiovascular disease was 67.1% in men and 66.4% in women, although these rates slightly underestimate the true risks because data on peripheral arterial disease, nonfatal aortic aneurysms, and angina managed exclusively medically weren’t collected.
This is the first study to scrutinize gender differences in the first manifestations of cardiovascular disease, broadly defined. For purposes of primary prevention, the first manifestation of cardiovascular disease is what’s most relevant, not the final fatal one, he noted.
From an absolute risk perspective, CHD as the first manifestation of cardiovascular disease clearly stood out as the major difference between men and women. In a comparison of 1,000 women with an equal number of men, there were 102 fewer cases of CHD in the women. This was offset by 70 more cases of cerebrovascular disease and 26 additional cases of heart failure in the women.
It’s particularly noteworthy that heart failure accounted for one-quarter of the first manifestations of cardiovascular disease in women, given that by definition none of the cases of heart failure recorded in this study were preceded by CHD. This underscores the importance of focusing of risk factors other than dyslipidemia, the physician said.
The Netherlands Organization for Health Research and Development and a variety of Dutch foundations funded the study. Dr. Leening reported having no financial conflicts of interest.
CHICAGO – Two out of three 55-year-old men and women will develop some form of cardiovascular disease during their remaining lifetime, but the first manifestation differs considerably by gender, according to a new analysis from The Rotterdam Study.
In men, the first manifestation of cardiovascular disease is most often a coronary heart disease event. Women are more likely to experience cerebrovascular disease or heart failure as their first event, Dr. Maarten J.G. Leening reported at the American Heart Association scientific sessions. Simultaneously with his presentation, the findings were published online in BMJ.
This new finding has important implications for primary prevention, noted Dr. Leening of Erasmus University, Rotterdam.
“Most of the attention of late in primary prevention has been drawn to the 2013 [AHA/ACC] lipid guidelines. But as we can see, for women it’s heart failure and cerebrovascular disease that are the major contributors to overall risk. And we know from the statin trials that there is no or only very limited reduction in heart failure and that the risk reduction for cerebrovascular disease is much smaller than for CHD. So our findings highlight the importance of blood pressure control and lifestyle factors, especially smoking, for primary prevention in women, since those are the best-established risk factors for cerebrovascular disease and heart failure,” he said.

Dr. Leening reported on 8,419 participants of The Rotterdam Study aged 55 or older, all free from cardiovascular disease at baseline. During a median 13.5 years and up to 20 years of follow-up in this ongoing prospective population-based study, 2,888 subjects developed cardiovascular disease. Coronary heart disease accounted for 826 of these first events, cerebrovascular events accounted for another 1,198, heart failure 726, and there were 102 other cardiovascular deaths.
At age 55, the overall lifetime risk of developing cardiovascular disease was 67.1% in men and 66.4% in women, although these rates slightly underestimate the true risks because data on peripheral arterial disease, nonfatal aortic aneurysms, and angina managed exclusively medically weren’t collected.
This is the first study to scrutinize gender differences in the first manifestations of cardiovascular disease, broadly defined. For purposes of primary prevention, the first manifestation of cardiovascular disease is what’s most relevant, not the final fatal one, he noted.
From an absolute risk perspective, CHD as the first manifestation of cardiovascular disease clearly stood out as the major difference between men and women. In a comparison of 1,000 women with an equal number of men, there were 102 fewer cases of CHD in the women. This was offset by 70 more cases of cerebrovascular disease and 26 additional cases of heart failure in the women.
It’s particularly noteworthy that heart failure accounted for one-quarter of the first manifestations of cardiovascular disease in women, given that by definition none of the cases of heart failure recorded in this study were preceded by CHD. This underscores the importance of focusing of risk factors other than dyslipidemia, the physician said.
The Netherlands Organization for Health Research and Development and a variety of Dutch foundations funded the study. Dr. Leening reported having no financial conflicts of interest.
CHICAGO – Two out of three 55-year-old men and women will develop some form of cardiovascular disease during their remaining lifetime, but the first manifestation differs considerably by gender, according to a new analysis from The Rotterdam Study.
In men, the first manifestation of cardiovascular disease is most often a coronary heart disease event. Women are more likely to experience cerebrovascular disease or heart failure as their first event, Dr. Maarten J.G. Leening reported at the American Heart Association scientific sessions. Simultaneously with his presentation, the findings were published online in BMJ.
This new finding has important implications for primary prevention, noted Dr. Leening of Erasmus University, Rotterdam.
“Most of the attention of late in primary prevention has been drawn to the 2013 [AHA/ACC] lipid guidelines. But as we can see, for women it’s heart failure and cerebrovascular disease that are the major contributors to overall risk. And we know from the statin trials that there is no or only very limited reduction in heart failure and that the risk reduction for cerebrovascular disease is much smaller than for CHD. So our findings highlight the importance of blood pressure control and lifestyle factors, especially smoking, for primary prevention in women, since those are the best-established risk factors for cerebrovascular disease and heart failure,” he said.

Dr. Leening reported on 8,419 participants of The Rotterdam Study aged 55 or older, all free from cardiovascular disease at baseline. During a median 13.5 years and up to 20 years of follow-up in this ongoing prospective population-based study, 2,888 subjects developed cardiovascular disease. Coronary heart disease accounted for 826 of these first events, cerebrovascular events accounted for another 1,198, heart failure 726, and there were 102 other cardiovascular deaths.
At age 55, the overall lifetime risk of developing cardiovascular disease was 67.1% in men and 66.4% in women, although these rates slightly underestimate the true risks because data on peripheral arterial disease, nonfatal aortic aneurysms, and angina managed exclusively medically weren’t collected.
This is the first study to scrutinize gender differences in the first manifestations of cardiovascular disease, broadly defined. For purposes of primary prevention, the first manifestation of cardiovascular disease is what’s most relevant, not the final fatal one, he noted.
From an absolute risk perspective, CHD as the first manifestation of cardiovascular disease clearly stood out as the major difference between men and women. In a comparison of 1,000 women with an equal number of men, there were 102 fewer cases of CHD in the women. This was offset by 70 more cases of cerebrovascular disease and 26 additional cases of heart failure in the women.
It’s particularly noteworthy that heart failure accounted for one-quarter of the first manifestations of cardiovascular disease in women, given that by definition none of the cases of heart failure recorded in this study were preceded by CHD. This underscores the importance of focusing of risk factors other than dyslipidemia, the physician said.
The Netherlands Organization for Health Research and Development and a variety of Dutch foundations funded the study. Dr. Leening reported having no financial conflicts of interest.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: Women are more likely to have cerebrovascular disease or heart failure as their first cardiovascular disease event.
Major finding: Among 1,000 women free of cardiovascular disease at age 55, there were 102 fewer cases of coronary heart disease as the initial manifestation of cardiovascular disease during their remaining lifetime than in an equal number of 55-year-old men.
Data source: An analysis from the ongoing, prospective, population-based Rotterdam Study of 8,419 subjects.
Disclosures: The Netherlands Organization for Health Research and Development and a variety of Dutch foundations funded the study. The presenter reported having no financial conflicts.
Emergency cardiac echocardiography accepted by Europeans
VIENNA – Rapid echocardiographic assessment has become routine for many patients who arrive at an emergency department with suspected acute heart failure, and experts consider these examinations critical for quickly getting patients on the right treatment.
Growing use and the important role for emergency echo exams prompted the European echocardiography community to issue in 2014 both recommendations and a position statement on the practice.
With their actions, European echocardiographers joined their U.S. colleagues who had earlier endorsed rapid, focused echocardiography exams. The European position also highlighted the limitations and pitfalls of emergency echo and the need for proper training.
Use of limited, directed, ultrasound heart examinations on an emergency basis by physicians who are not cardiologists is “an irreversible process, but without appropriate training it may become dangerous,” Dr. Nuno Cardim said at the annual meeting of the European Association of Cardiovascular Imaging (EACVI).
A focused cardiac ultrasound (FoCUS) examination for patients with an emergency cardiac condition such as acute heart failure is not a new concept. In 2010, the American Society of Echocardiography and the American College of Emergency Physicians jointly issued a consensus statement on emergency FoCUS (J. Am. Soc. Echocardiogr. 2010;23:1225-30), and the American Society of Echocardiography followed with additional recommendations in 2013 that also dealt with nonemergency uses for FoCUS (J. Am. Soc. Echocardiogr. 2013;26:567-81).
In its 2014 position statement released last May, the EACVI directly addressed FoCUS for the first time (Eur. Heart J. Cardiovasc. Imaging 2014:15;956-60). The statement acknowledged the important role for a circumscribed, point-of-care ultrasound exam in patients undergoing cardiopulmonary resuscitation and in other critical cardiac conditions, but highlighted that a FoCUS exam does not substitute for a comprehensive echocardiographic exam, and that FoCUS should only be done by properly trained clinicians who appreciate the limits of a FoCUS exam.
The EASVI recommendations, which came out a few months later in collaboration with the Acute Cardiovascular Care Association, said that “echocardiography is now recommended (where appropriately trained practitioners are available) in the management of cardiac arrest. However, FoCUS should always be used and interpreted thoughtfully, since this fundamentally limited approach may lead to missing/misinterpretation of important findings unless the practitioner is aware of its (and their) limitations” (Eur. Heart J. Cardiovasc. Imaging 2014 [doi:10.1093/ehjci/jeu210]).
“Of course all patients with suspected acute heart failure in the emergency department should undergo an echo exam. The question is, who will do it? These are patients who are the most difficult to assess,” said Dr. Susanna Price, a member of the EACVI recommendations panel and a specialist in critical care cardiology at Royal Brompton Hospital in London.
“Without proper training, the person doing FoCUS could make a false positive diagnosis, or might miss something and make a false negative diagnosis,” said Dr. Cardim, professor and director of echocardiography and cardiac imaging at Hospital da Luz in Lisbon, and another member of the EACVI panel.
To avoid this, emergency-medicine physicians and others who often triage patients with acute heart disorders should be trained in echocardiography and especially the FoCUS exam, which aims to quickly evaluate several important abnormalities of cardiac function: pericardial effusion, cardiac tamponade, left and right ventricular size and function, and intravascular volume status. A FoCUS exam also screens for pulmonary embolism. FoCUS assesses each of these in a yes-or-no or present-or-absent way, information critical for guiding emergency management but lacking the quantitative and detailed information available with a comprehensive echocardiography exam.
“FoCUS must never substitute” for the comprehensive exam, which should always also be done, he said. FoCUS “should be used wisely and cautiously because of its limitations.”
The FoCUS exam also has equipment specifications. Ideally, clinicians should use a portable, hand-held ultrasound machine, which is larger than “pocket-sized” ultrasound devices and hence gives much better image quality compared with pocket-sized devices, Dr. Cardim said in an interview.
Dr. Cardim and Dr. Price had no disclosures.
On Twitter @mitchelzoler
VIENNA – Rapid echocardiographic assessment has become routine for many patients who arrive at an emergency department with suspected acute heart failure, and experts consider these examinations critical for quickly getting patients on the right treatment.
Growing use and the important role for emergency echo exams prompted the European echocardiography community to issue in 2014 both recommendations and a position statement on the practice.
With their actions, European echocardiographers joined their U.S. colleagues who had earlier endorsed rapid, focused echocardiography exams. The European position also highlighted the limitations and pitfalls of emergency echo and the need for proper training.
Use of limited, directed, ultrasound heart examinations on an emergency basis by physicians who are not cardiologists is “an irreversible process, but without appropriate training it may become dangerous,” Dr. Nuno Cardim said at the annual meeting of the European Association of Cardiovascular Imaging (EACVI).
A focused cardiac ultrasound (FoCUS) examination for patients with an emergency cardiac condition such as acute heart failure is not a new concept. In 2010, the American Society of Echocardiography and the American College of Emergency Physicians jointly issued a consensus statement on emergency FoCUS (J. Am. Soc. Echocardiogr. 2010;23:1225-30), and the American Society of Echocardiography followed with additional recommendations in 2013 that also dealt with nonemergency uses for FoCUS (J. Am. Soc. Echocardiogr. 2013;26:567-81).
In its 2014 position statement released last May, the EACVI directly addressed FoCUS for the first time (Eur. Heart J. Cardiovasc. Imaging 2014:15;956-60). The statement acknowledged the important role for a circumscribed, point-of-care ultrasound exam in patients undergoing cardiopulmonary resuscitation and in other critical cardiac conditions, but highlighted that a FoCUS exam does not substitute for a comprehensive echocardiographic exam, and that FoCUS should only be done by properly trained clinicians who appreciate the limits of a FoCUS exam.
The EASVI recommendations, which came out a few months later in collaboration with the Acute Cardiovascular Care Association, said that “echocardiography is now recommended (where appropriately trained practitioners are available) in the management of cardiac arrest. However, FoCUS should always be used and interpreted thoughtfully, since this fundamentally limited approach may lead to missing/misinterpretation of important findings unless the practitioner is aware of its (and their) limitations” (Eur. Heart J. Cardiovasc. Imaging 2014 [doi:10.1093/ehjci/jeu210]).
“Of course all patients with suspected acute heart failure in the emergency department should undergo an echo exam. The question is, who will do it? These are patients who are the most difficult to assess,” said Dr. Susanna Price, a member of the EACVI recommendations panel and a specialist in critical care cardiology at Royal Brompton Hospital in London.
“Without proper training, the person doing FoCUS could make a false positive diagnosis, or might miss something and make a false negative diagnosis,” said Dr. Cardim, professor and director of echocardiography and cardiac imaging at Hospital da Luz in Lisbon, and another member of the EACVI panel.
To avoid this, emergency-medicine physicians and others who often triage patients with acute heart disorders should be trained in echocardiography and especially the FoCUS exam, which aims to quickly evaluate several important abnormalities of cardiac function: pericardial effusion, cardiac tamponade, left and right ventricular size and function, and intravascular volume status. A FoCUS exam also screens for pulmonary embolism. FoCUS assesses each of these in a yes-or-no or present-or-absent way, information critical for guiding emergency management but lacking the quantitative and detailed information available with a comprehensive echocardiography exam.
“FoCUS must never substitute” for the comprehensive exam, which should always also be done, he said. FoCUS “should be used wisely and cautiously because of its limitations.”
The FoCUS exam also has equipment specifications. Ideally, clinicians should use a portable, hand-held ultrasound machine, which is larger than “pocket-sized” ultrasound devices and hence gives much better image quality compared with pocket-sized devices, Dr. Cardim said in an interview.
Dr. Cardim and Dr. Price had no disclosures.
On Twitter @mitchelzoler
VIENNA – Rapid echocardiographic assessment has become routine for many patients who arrive at an emergency department with suspected acute heart failure, and experts consider these examinations critical for quickly getting patients on the right treatment.
Growing use and the important role for emergency echo exams prompted the European echocardiography community to issue in 2014 both recommendations and a position statement on the practice.
With their actions, European echocardiographers joined their U.S. colleagues who had earlier endorsed rapid, focused echocardiography exams. The European position also highlighted the limitations and pitfalls of emergency echo and the need for proper training.
Use of limited, directed, ultrasound heart examinations on an emergency basis by physicians who are not cardiologists is “an irreversible process, but without appropriate training it may become dangerous,” Dr. Nuno Cardim said at the annual meeting of the European Association of Cardiovascular Imaging (EACVI).
A focused cardiac ultrasound (FoCUS) examination for patients with an emergency cardiac condition such as acute heart failure is not a new concept. In 2010, the American Society of Echocardiography and the American College of Emergency Physicians jointly issued a consensus statement on emergency FoCUS (J. Am. Soc. Echocardiogr. 2010;23:1225-30), and the American Society of Echocardiography followed with additional recommendations in 2013 that also dealt with nonemergency uses for FoCUS (J. Am. Soc. Echocardiogr. 2013;26:567-81).
In its 2014 position statement released last May, the EACVI directly addressed FoCUS for the first time (Eur. Heart J. Cardiovasc. Imaging 2014:15;956-60). The statement acknowledged the important role for a circumscribed, point-of-care ultrasound exam in patients undergoing cardiopulmonary resuscitation and in other critical cardiac conditions, but highlighted that a FoCUS exam does not substitute for a comprehensive echocardiographic exam, and that FoCUS should only be done by properly trained clinicians who appreciate the limits of a FoCUS exam.
The EASVI recommendations, which came out a few months later in collaboration with the Acute Cardiovascular Care Association, said that “echocardiography is now recommended (where appropriately trained practitioners are available) in the management of cardiac arrest. However, FoCUS should always be used and interpreted thoughtfully, since this fundamentally limited approach may lead to missing/misinterpretation of important findings unless the practitioner is aware of its (and their) limitations” (Eur. Heart J. Cardiovasc. Imaging 2014 [doi:10.1093/ehjci/jeu210]).
“Of course all patients with suspected acute heart failure in the emergency department should undergo an echo exam. The question is, who will do it? These are patients who are the most difficult to assess,” said Dr. Susanna Price, a member of the EACVI recommendations panel and a specialist in critical care cardiology at Royal Brompton Hospital in London.
“Without proper training, the person doing FoCUS could make a false positive diagnosis, or might miss something and make a false negative diagnosis,” said Dr. Cardim, professor and director of echocardiography and cardiac imaging at Hospital da Luz in Lisbon, and another member of the EACVI panel.
To avoid this, emergency-medicine physicians and others who often triage patients with acute heart disorders should be trained in echocardiography and especially the FoCUS exam, which aims to quickly evaluate several important abnormalities of cardiac function: pericardial effusion, cardiac tamponade, left and right ventricular size and function, and intravascular volume status. A FoCUS exam also screens for pulmonary embolism. FoCUS assesses each of these in a yes-or-no or present-or-absent way, information critical for guiding emergency management but lacking the quantitative and detailed information available with a comprehensive echocardiography exam.
“FoCUS must never substitute” for the comprehensive exam, which should always also be done, he said. FoCUS “should be used wisely and cautiously because of its limitations.”
The FoCUS exam also has equipment specifications. Ideally, clinicians should use a portable, hand-held ultrasound machine, which is larger than “pocket-sized” ultrasound devices and hence gives much better image quality compared with pocket-sized devices, Dr. Cardim said in an interview.
Dr. Cardim and Dr. Price had no disclosures.
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM EUROECHO-IMAGING 2014
HF: Glucose at ED presentation may predict mortality
Among adults who present to the emergency department with acute heart failure, blood glucose levels may predict 30-day mortality, regardless of whether or not the patients have preexisting diabetes.
At ED presentation, patients with acute heart failure syndrome “demonstrate a wide spectrum of physiological and metabolic perturbations.” Hyperglycemia occurs in up to 40% of them, irrespective of their diabetes status. “If blood glucose measurement is prognostically useful, it may be of broad potential utility because it is a rapid, readily available, and inexpensive test that can be used in the acute setting to allow rapid risk stratification for a wide range of potential outcomes,” according to Dr. Maneesh Sud of the University of Toronto and Toronto General Hospital and his associates.
To examine whether initial glucose level correlated with later outcomes, the investigators analyzed data from two large population-based cohorts of patients hospitalized for acute HF during a 3-year period. A total of 9,275 of the 16,524 patients (median age, 79 years) did not have preexisting diabetes (56%), and the remaining 44% did (Eur. Heart J. 2015 [doi:10.1093/eurheartj/ehu462]).
Among patients with diabetes, a blood glucose level exceeding 11.1 mmol/L was associated with significantly increased all-cause 30-day mortality, compared with normal glucose levels, with a hazard ratio (HR) of 1.48. Among patients without diabetes, a blood glucose level exceeding 6.1 mmol/L was associated with significantly increased all-cause 30-day mortality, with an HR of 1.26, and that risk rose with increasing glucose levels to 1.50 at the maximum level of 11.1 mmol/L.
The risk for cardiovascular death within 30 days increased as blood glucose levels rose for both groups of patients, as did the risk for cardiovascular hospitalization. In addition, both patients who had preexisting diabetes and those who did not were at significantly increased risk for diabetes-related hospitalization if their blood glucose level exceeded the normal range at presentation to the ED. And, in patients without diabetes who had elevated blood glucose levels, the risk of developing diabetes was significantly increased, in a dose-dependent fashion. These findings suggest that determining blood glucose levels at ED presentation “may serve as a screen to identify high-risk patients who warrant formal testing for diabetes, allowing for prompt referral to prevent further morbidity and mortality,” Dr. Sud and his associates said.
Among adults who present to the emergency department with acute heart failure, blood glucose levels may predict 30-day mortality, regardless of whether or not the patients have preexisting diabetes.
At ED presentation, patients with acute heart failure syndrome “demonstrate a wide spectrum of physiological and metabolic perturbations.” Hyperglycemia occurs in up to 40% of them, irrespective of their diabetes status. “If blood glucose measurement is prognostically useful, it may be of broad potential utility because it is a rapid, readily available, and inexpensive test that can be used in the acute setting to allow rapid risk stratification for a wide range of potential outcomes,” according to Dr. Maneesh Sud of the University of Toronto and Toronto General Hospital and his associates.
To examine whether initial glucose level correlated with later outcomes, the investigators analyzed data from two large population-based cohorts of patients hospitalized for acute HF during a 3-year period. A total of 9,275 of the 16,524 patients (median age, 79 years) did not have preexisting diabetes (56%), and the remaining 44% did (Eur. Heart J. 2015 [doi:10.1093/eurheartj/ehu462]).
Among patients with diabetes, a blood glucose level exceeding 11.1 mmol/L was associated with significantly increased all-cause 30-day mortality, compared with normal glucose levels, with a hazard ratio (HR) of 1.48. Among patients without diabetes, a blood glucose level exceeding 6.1 mmol/L was associated with significantly increased all-cause 30-day mortality, with an HR of 1.26, and that risk rose with increasing glucose levels to 1.50 at the maximum level of 11.1 mmol/L.
The risk for cardiovascular death within 30 days increased as blood glucose levels rose for both groups of patients, as did the risk for cardiovascular hospitalization. In addition, both patients who had preexisting diabetes and those who did not were at significantly increased risk for diabetes-related hospitalization if their blood glucose level exceeded the normal range at presentation to the ED. And, in patients without diabetes who had elevated blood glucose levels, the risk of developing diabetes was significantly increased, in a dose-dependent fashion. These findings suggest that determining blood glucose levels at ED presentation “may serve as a screen to identify high-risk patients who warrant formal testing for diabetes, allowing for prompt referral to prevent further morbidity and mortality,” Dr. Sud and his associates said.
Among adults who present to the emergency department with acute heart failure, blood glucose levels may predict 30-day mortality, regardless of whether or not the patients have preexisting diabetes.
At ED presentation, patients with acute heart failure syndrome “demonstrate a wide spectrum of physiological and metabolic perturbations.” Hyperglycemia occurs in up to 40% of them, irrespective of their diabetes status. “If blood glucose measurement is prognostically useful, it may be of broad potential utility because it is a rapid, readily available, and inexpensive test that can be used in the acute setting to allow rapid risk stratification for a wide range of potential outcomes,” according to Dr. Maneesh Sud of the University of Toronto and Toronto General Hospital and his associates.
To examine whether initial glucose level correlated with later outcomes, the investigators analyzed data from two large population-based cohorts of patients hospitalized for acute HF during a 3-year period. A total of 9,275 of the 16,524 patients (median age, 79 years) did not have preexisting diabetes (56%), and the remaining 44% did (Eur. Heart J. 2015 [doi:10.1093/eurheartj/ehu462]).
Among patients with diabetes, a blood glucose level exceeding 11.1 mmol/L was associated with significantly increased all-cause 30-day mortality, compared with normal glucose levels, with a hazard ratio (HR) of 1.48. Among patients without diabetes, a blood glucose level exceeding 6.1 mmol/L was associated with significantly increased all-cause 30-day mortality, with an HR of 1.26, and that risk rose with increasing glucose levels to 1.50 at the maximum level of 11.1 mmol/L.
The risk for cardiovascular death within 30 days increased as blood glucose levels rose for both groups of patients, as did the risk for cardiovascular hospitalization. In addition, both patients who had preexisting diabetes and those who did not were at significantly increased risk for diabetes-related hospitalization if their blood glucose level exceeded the normal range at presentation to the ED. And, in patients without diabetes who had elevated blood glucose levels, the risk of developing diabetes was significantly increased, in a dose-dependent fashion. These findings suggest that determining blood glucose levels at ED presentation “may serve as a screen to identify high-risk patients who warrant formal testing for diabetes, allowing for prompt referral to prevent further morbidity and mortality,” Dr. Sud and his associates said.
FROM EUROPEAN HEART JOURNAL
Key clinical point: Among patients who present to the ED with acute HF, blood glucose levels predict 30-day mortality and other outcomes.
Major finding: Patients with diabetes who had elevated blood glucose levels showed increased 30-day mortality with a hazard ratio of up to 1.48, while those without diabetes showed increased 30-day mortality with a hazard ratio of up to 1.50.
Data source: A secondary analysis of data from two population-based cohorts comprising 16,524 patients who presented to the ED with acute HF during a 3-year period.
Disclosures: This study was supported by the Canadian Institutes of Health Research and the Heart and Stroke Foundation of Ontario. Dr. Sud and his associates reported having no relevant financial disclosures.