User login
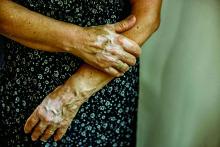
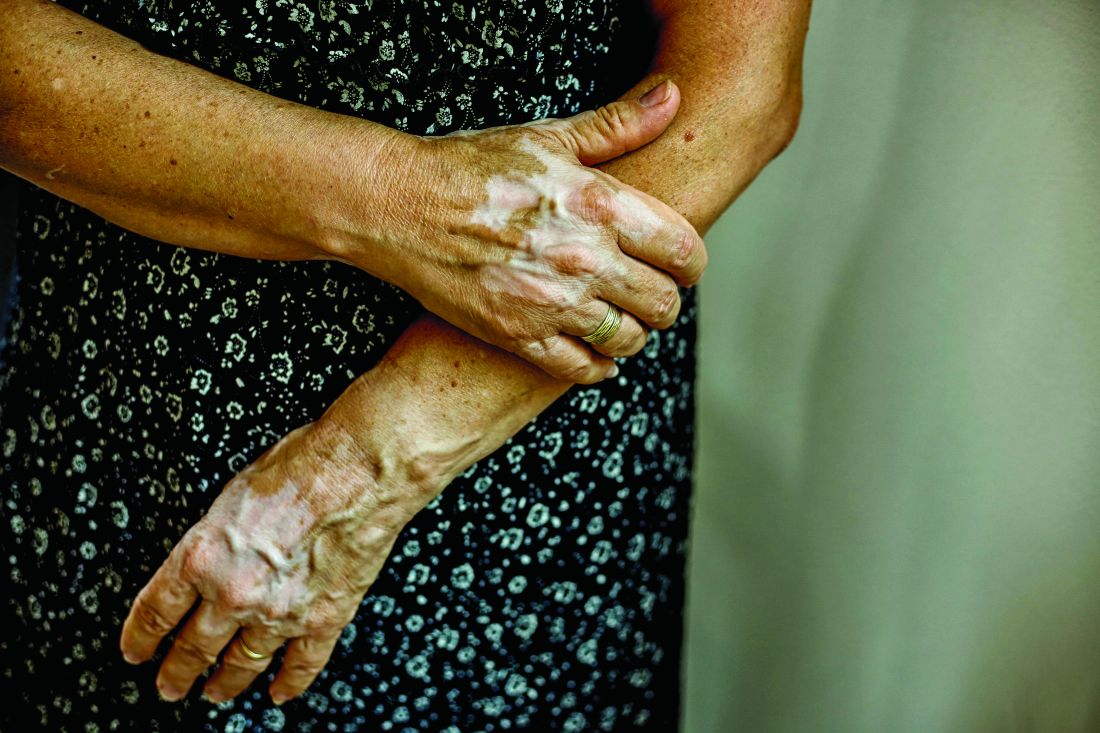
Insurance coverage for vitiligo varies widely in the U.S., analysis finds
, which may disproportionately affect patients of color.
Those are the conclusions from an analysis of vitiligo treatment coverage policies across major health insurers in the United States.
“Vitiligo can be less noticeable in patients with lighter skin types, becoming apparent only when affected patches fail to tan,” first authors Andrew Blundell, MD, MSc, and Moniyka Sachar, MD, wrote in a study published online on July 16 in Pediatric Dermatology. However, they pointed out that, in patients with darker skin types, “vitiligo can be far more evident due to the stark contrast of involved versus uninvolved skin, and as such can lead to a significant impact on quality of life, as well as heightened stigmatization.”
Nevertheless, they noted many health care insurers consider vitiligo as a cosmetic condition, and do not cover treatments, and for the 1%-2% of the general population with vitiligo, “this lack of recognition from health care insurers makes treatments both less accessible and affordable, and only further marginalizes patients with this condition.”
Dr. Blundell, of San Juan Bautista School of Medicine, Caguas, P.R., and Dr. Sachar, of the department of dermatology at Brown University, Providence, R.I., and colleagues surveyed 15 commercial health care insurers, 50 BlueCross BlueShield plans, Medicare, Medicaid, and Veterans Affairs to determine the level of treatment coverage for vitiligo. They looked at office visits, medications (the topical calcineurin inhibitors [TCIs] pimecrolimus, and tacrolimus), excimer laser therapy, and phototherapy (psoralen with UVA [PUVA] and narrow-band UVB [nbUVB]). They collected information from medical policies available online or by direct contact with the plans in 2018.
The researchers reported data from 17 organizations with regional or national coverage policies for vitiligo treatment and two others – BlueCross BlueShield and Medicaid – which had policies that differed by state and plan. Of the 17 organizations, only 12% did not cover TCIs, 56% did not cover nbUVB phototherapy, 53% did not cover PUVA phototherapy, and 41% did not cover laser therapy.
As for BlueCross BlueShield, the health plan did not cover pimecrolimus and tacrolimus in 39% and 35% of states, respectively. At the same time, NbUVB and PUVA therapy were not covered in 20% and 10% of states, respectively, while excimer laser therapy was not covered in 82% of states.
Of accessible Medicaid information from 32 states, 11 did not cover topicals, 5 did not cover nbUVB, 4 did not cover PUVA, and 7 did not cover laser therapy. “The two most commonly cited reasons for denial of coverage were (a) vitiligo is considered a cosmetic condition and (b) certain therapies are not FDA-approved for vitiligo, though they may be approved for other skin conditions,” the study authors wrote.
While the analysis revealed that topical TCI therapy is more widely covered by insurance companies, compared with phototherapy, “multiple studies have shown that a combination of both topical and phototherapy is more effective in treating vitiligo than either alone,” they noted. “Vitiligo treatments can delay the progression of the disease and result in better outcomes when started early, furthering the need for insurance coverage of these treatments. If all proven and accepted vitiligo treatments were covered by their health insurers, patients would have better access, as well as timely and affordable ways by which to limit depigmentation and to repigment affected areas.”
In addition, lack of access to treatments “may increase health disparities among already-marginalized groups, such as children and adults of darker skin phototypes,” they wrote.
Seemal R. Desai, MD, who was asked to comment on the study, said that the findings resonate with him based on his clinical experience as a dermatologist at the University of Texas Southwestern Medical Center in Dallas and in clinical practice. “Vitiligo has a high psychological impact, continues to increase in its prevalence, and has been shown to be an autoimmune, chronic, inflammatory skin disease, yet we’re still having challenges with treatment,” said Dr. Desai, who is also a member of the board of directors for the American Academy of Dermatology and the Global Vitiligo Foundation (GVF).
He said that he is working with the AAD, the GVF, and other stakeholders to improve treatment coverage. For example, in Massachusetts, the Tufts Health Plan had stopped covering treatment for vitiligo. “Through a series of advocacy efforts, that was reversed a couple of years ago,” said Dr. Desai, who is also a past president of the Skin of Color Society. “We also have seen isolated reports of Medicaid and Medicare coverage where local contractors aren’t following national Centers for Medicare and Medicaid Service directive guidance. The challenge becomes, how do you get consistency in treatment coverage, and how do you make sure patients continue to get access to treatment?”
Turning the tide will require “a concerted effort” by dermatologists to engage with the payers, he added. “I’ve had to get on the phone with countless insurance companies on behalf of my patients and make them understand the comorbidities associated with vitiligo, sending them copies of studies that show it’s an autoimmune disease linked to thyroid issues,” Dr. Desai continued. “We talk a lot about the psychological burden and quality of life. There’s still a lot of work to be done in this sphere, but I think we’re making progress.”
With hopes that Janus kinase (JAK) inhibitors and other new products being investigated will soon be approved as a treatment option for vitiligo, Dr. Desai said that now is the time to standardize coverage for patients. “It’s important that we start talking about insurance coverage and denial issues now and get ahead of it, so that when we get those JAK inhibitors available, we don’t fight coverage decisions then.”
The researchers acknowledged certain limitations of the study, including the fact that it was based on insurance coverage from 2017 to 2018 and the lack of easily available state Medicaid policies.
The study coauthors were Colleen K. Gabel, MD, of the University of Massachusetts, Worcester, and Lionel G. Bercovitch, MD, of Brown University. None of the study authors reported financial disclosures.
Dr. Desai disclosed that he has conducted vitiligo research trials and has done consulting work for several pharmaceutical companies.
, which may disproportionately affect patients of color.
Those are the conclusions from an analysis of vitiligo treatment coverage policies across major health insurers in the United States.
“Vitiligo can be less noticeable in patients with lighter skin types, becoming apparent only when affected patches fail to tan,” first authors Andrew Blundell, MD, MSc, and Moniyka Sachar, MD, wrote in a study published online on July 16 in Pediatric Dermatology. However, they pointed out that, in patients with darker skin types, “vitiligo can be far more evident due to the stark contrast of involved versus uninvolved skin, and as such can lead to a significant impact on quality of life, as well as heightened stigmatization.”
Nevertheless, they noted many health care insurers consider vitiligo as a cosmetic condition, and do not cover treatments, and for the 1%-2% of the general population with vitiligo, “this lack of recognition from health care insurers makes treatments both less accessible and affordable, and only further marginalizes patients with this condition.”
Dr. Blundell, of San Juan Bautista School of Medicine, Caguas, P.R., and Dr. Sachar, of the department of dermatology at Brown University, Providence, R.I., and colleagues surveyed 15 commercial health care insurers, 50 BlueCross BlueShield plans, Medicare, Medicaid, and Veterans Affairs to determine the level of treatment coverage for vitiligo. They looked at office visits, medications (the topical calcineurin inhibitors [TCIs] pimecrolimus, and tacrolimus), excimer laser therapy, and phototherapy (psoralen with UVA [PUVA] and narrow-band UVB [nbUVB]). They collected information from medical policies available online or by direct contact with the plans in 2018.
The researchers reported data from 17 organizations with regional or national coverage policies for vitiligo treatment and two others – BlueCross BlueShield and Medicaid – which had policies that differed by state and plan. Of the 17 organizations, only 12% did not cover TCIs, 56% did not cover nbUVB phototherapy, 53% did not cover PUVA phototherapy, and 41% did not cover laser therapy.
As for BlueCross BlueShield, the health plan did not cover pimecrolimus and tacrolimus in 39% and 35% of states, respectively. At the same time, NbUVB and PUVA therapy were not covered in 20% and 10% of states, respectively, while excimer laser therapy was not covered in 82% of states.
Of accessible Medicaid information from 32 states, 11 did not cover topicals, 5 did not cover nbUVB, 4 did not cover PUVA, and 7 did not cover laser therapy. “The two most commonly cited reasons for denial of coverage were (a) vitiligo is considered a cosmetic condition and (b) certain therapies are not FDA-approved for vitiligo, though they may be approved for other skin conditions,” the study authors wrote.
While the analysis revealed that topical TCI therapy is more widely covered by insurance companies, compared with phototherapy, “multiple studies have shown that a combination of both topical and phototherapy is more effective in treating vitiligo than either alone,” they noted. “Vitiligo treatments can delay the progression of the disease and result in better outcomes when started early, furthering the need for insurance coverage of these treatments. If all proven and accepted vitiligo treatments were covered by their health insurers, patients would have better access, as well as timely and affordable ways by which to limit depigmentation and to repigment affected areas.”
In addition, lack of access to treatments “may increase health disparities among already-marginalized groups, such as children and adults of darker skin phototypes,” they wrote.
Seemal R. Desai, MD, who was asked to comment on the study, said that the findings resonate with him based on his clinical experience as a dermatologist at the University of Texas Southwestern Medical Center in Dallas and in clinical practice. “Vitiligo has a high psychological impact, continues to increase in its prevalence, and has been shown to be an autoimmune, chronic, inflammatory skin disease, yet we’re still having challenges with treatment,” said Dr. Desai, who is also a member of the board of directors for the American Academy of Dermatology and the Global Vitiligo Foundation (GVF).
He said that he is working with the AAD, the GVF, and other stakeholders to improve treatment coverage. For example, in Massachusetts, the Tufts Health Plan had stopped covering treatment for vitiligo. “Through a series of advocacy efforts, that was reversed a couple of years ago,” said Dr. Desai, who is also a past president of the Skin of Color Society. “We also have seen isolated reports of Medicaid and Medicare coverage where local contractors aren’t following national Centers for Medicare and Medicaid Service directive guidance. The challenge becomes, how do you get consistency in treatment coverage, and how do you make sure patients continue to get access to treatment?”
Turning the tide will require “a concerted effort” by dermatologists to engage with the payers, he added. “I’ve had to get on the phone with countless insurance companies on behalf of my patients and make them understand the comorbidities associated with vitiligo, sending them copies of studies that show it’s an autoimmune disease linked to thyroid issues,” Dr. Desai continued. “We talk a lot about the psychological burden and quality of life. There’s still a lot of work to be done in this sphere, but I think we’re making progress.”
With hopes that Janus kinase (JAK) inhibitors and other new products being investigated will soon be approved as a treatment option for vitiligo, Dr. Desai said that now is the time to standardize coverage for patients. “It’s important that we start talking about insurance coverage and denial issues now and get ahead of it, so that when we get those JAK inhibitors available, we don’t fight coverage decisions then.”
The researchers acknowledged certain limitations of the study, including the fact that it was based on insurance coverage from 2017 to 2018 and the lack of easily available state Medicaid policies.
The study coauthors were Colleen K. Gabel, MD, of the University of Massachusetts, Worcester, and Lionel G. Bercovitch, MD, of Brown University. None of the study authors reported financial disclosures.
Dr. Desai disclosed that he has conducted vitiligo research trials and has done consulting work for several pharmaceutical companies.
, which may disproportionately affect patients of color.
Those are the conclusions from an analysis of vitiligo treatment coverage policies across major health insurers in the United States.
“Vitiligo can be less noticeable in patients with lighter skin types, becoming apparent only when affected patches fail to tan,” first authors Andrew Blundell, MD, MSc, and Moniyka Sachar, MD, wrote in a study published online on July 16 in Pediatric Dermatology. However, they pointed out that, in patients with darker skin types, “vitiligo can be far more evident due to the stark contrast of involved versus uninvolved skin, and as such can lead to a significant impact on quality of life, as well as heightened stigmatization.”
Nevertheless, they noted many health care insurers consider vitiligo as a cosmetic condition, and do not cover treatments, and for the 1%-2% of the general population with vitiligo, “this lack of recognition from health care insurers makes treatments both less accessible and affordable, and only further marginalizes patients with this condition.”
Dr. Blundell, of San Juan Bautista School of Medicine, Caguas, P.R., and Dr. Sachar, of the department of dermatology at Brown University, Providence, R.I., and colleagues surveyed 15 commercial health care insurers, 50 BlueCross BlueShield plans, Medicare, Medicaid, and Veterans Affairs to determine the level of treatment coverage for vitiligo. They looked at office visits, medications (the topical calcineurin inhibitors [TCIs] pimecrolimus, and tacrolimus), excimer laser therapy, and phototherapy (psoralen with UVA [PUVA] and narrow-band UVB [nbUVB]). They collected information from medical policies available online or by direct contact with the plans in 2018.
The researchers reported data from 17 organizations with regional or national coverage policies for vitiligo treatment and two others – BlueCross BlueShield and Medicaid – which had policies that differed by state and plan. Of the 17 organizations, only 12% did not cover TCIs, 56% did not cover nbUVB phototherapy, 53% did not cover PUVA phototherapy, and 41% did not cover laser therapy.
As for BlueCross BlueShield, the health plan did not cover pimecrolimus and tacrolimus in 39% and 35% of states, respectively. At the same time, NbUVB and PUVA therapy were not covered in 20% and 10% of states, respectively, while excimer laser therapy was not covered in 82% of states.
Of accessible Medicaid information from 32 states, 11 did not cover topicals, 5 did not cover nbUVB, 4 did not cover PUVA, and 7 did not cover laser therapy. “The two most commonly cited reasons for denial of coverage were (a) vitiligo is considered a cosmetic condition and (b) certain therapies are not FDA-approved for vitiligo, though they may be approved for other skin conditions,” the study authors wrote.
While the analysis revealed that topical TCI therapy is more widely covered by insurance companies, compared with phototherapy, “multiple studies have shown that a combination of both topical and phototherapy is more effective in treating vitiligo than either alone,” they noted. “Vitiligo treatments can delay the progression of the disease and result in better outcomes when started early, furthering the need for insurance coverage of these treatments. If all proven and accepted vitiligo treatments were covered by their health insurers, patients would have better access, as well as timely and affordable ways by which to limit depigmentation and to repigment affected areas.”
In addition, lack of access to treatments “may increase health disparities among already-marginalized groups, such as children and adults of darker skin phototypes,” they wrote.
Seemal R. Desai, MD, who was asked to comment on the study, said that the findings resonate with him based on his clinical experience as a dermatologist at the University of Texas Southwestern Medical Center in Dallas and in clinical practice. “Vitiligo has a high psychological impact, continues to increase in its prevalence, and has been shown to be an autoimmune, chronic, inflammatory skin disease, yet we’re still having challenges with treatment,” said Dr. Desai, who is also a member of the board of directors for the American Academy of Dermatology and the Global Vitiligo Foundation (GVF).
He said that he is working with the AAD, the GVF, and other stakeholders to improve treatment coverage. For example, in Massachusetts, the Tufts Health Plan had stopped covering treatment for vitiligo. “Through a series of advocacy efforts, that was reversed a couple of years ago,” said Dr. Desai, who is also a past president of the Skin of Color Society. “We also have seen isolated reports of Medicaid and Medicare coverage where local contractors aren’t following national Centers for Medicare and Medicaid Service directive guidance. The challenge becomes, how do you get consistency in treatment coverage, and how do you make sure patients continue to get access to treatment?”
Turning the tide will require “a concerted effort” by dermatologists to engage with the payers, he added. “I’ve had to get on the phone with countless insurance companies on behalf of my patients and make them understand the comorbidities associated with vitiligo, sending them copies of studies that show it’s an autoimmune disease linked to thyroid issues,” Dr. Desai continued. “We talk a lot about the psychological burden and quality of life. There’s still a lot of work to be done in this sphere, but I think we’re making progress.”
With hopes that Janus kinase (JAK) inhibitors and other new products being investigated will soon be approved as a treatment option for vitiligo, Dr. Desai said that now is the time to standardize coverage for patients. “It’s important that we start talking about insurance coverage and denial issues now and get ahead of it, so that when we get those JAK inhibitors available, we don’t fight coverage decisions then.”
The researchers acknowledged certain limitations of the study, including the fact that it was based on insurance coverage from 2017 to 2018 and the lack of easily available state Medicaid policies.
The study coauthors were Colleen K. Gabel, MD, of the University of Massachusetts, Worcester, and Lionel G. Bercovitch, MD, of Brown University. None of the study authors reported financial disclosures.
Dr. Desai disclosed that he has conducted vitiligo research trials and has done consulting work for several pharmaceutical companies.
FROM PEDIATRIC DERMATOLOGY
Exploring the Utility of Artificial Intelligence During COVID-19 in Dermatology Practice
With the need to adapt to the given challenges associated with COVID-19, artificial intelligence (AI) serves as a potential tool in providing access to medical-based diagnosis in a novel way. Artificial intelligence is defined as intelligence harnessed by machines that have the ability to perform what is called cognitive thinking and to mimic the problem-solving abilities of the human mind. Virtual AI in dermatology entails neural network–based guidance that includes developing algorithms to detect skin pathology through photographs.1 To use AI in dermatology, recognition of visual patterns must be established to give diagnoses. These neural networks have been used to classify skin diseases, including cancer, actinic keratosis, and warts.2
AI for Skin Cancer
The use of AI to classify melanoma and nonmelanoma skin cancer has been studied extensively, including the following 2 research projects.
Convolutional Neural Network
In 2017, Stanford University published a study in which a deep-learning algorithm known as a convolutional neural network was used to classify skin lesions.3 The network was trained using a dataset of 129,450 clinical images of 2032 diseases. Its performance was compared to that of 21 board-certified dermatologists on biopsy-proven clinical images with 2 classifications of cases: (1) keratinocyte carcinoma as opposed to benign seborrheic keratosis and (2) malignant melanoma as opposed to benign nevi—the first representing the most common skin cancers, and the second, the deadliest skin cancers. The study showed that the machine could accurately identify and classify skin cancers compared to the work of board-certified dermatologists. The study did not include demographic information, which limits its external validity.3
Dermoscopic Image Classification
A 2019 study by Brinker and colleagues4 showed the superiority of automated dermoscopic melanoma image classifications compared to the work of board-certified dermatologists. For the study, 804 biopsy-proven images of melanoma and nevi (1:1 ratio) were randomly presented to dermatologists for their evaluation and recommended treatment (yielding 19,296 recommendations). The dermatologists classified the lesions with a sensitivity of 67.2% and specificity of 62.2%; the trained convolutional neural network attained both higher sensitivity (82.3%) and higher specificity (77.9%).4
Smartphone Diagnosis of Melanoma
An application of AI has been to use smartphone apps for the diagnosis of melanoma. The most utilized and novel algorithm-based smartphone app that assesses skin lesions for malignancy characteristics is SkinVision. With a simple download from Apple’s App Store, this technology allows a person to check their skin spots by taking a photograph and receiving algorithmic risk-assessment feedback. This inexpensive software ($51.78 a year) also allows a patient’s physician to assess the photograph and then validate their assessment by comparing it with the algorithmic analysis that the program provides.5
A review of SkinVision conducted by Thissen and colleagues6 found that, in a hypothetical population of 1000 adults of whom 3% actually had melanoma, 4 of those 30 people would not have been flagged as at “high risk” by SkinVision. There also was a high false-positive rate with the app, with more than 200 people flagged as at high risk. The analysis pegged SkinVision as having a sensitivity of 88% and specificity of 79%.6
In summary, systematic review of diagnostic accuracy has shown that, although there is accuracy in AI analyses, it should be used only as a guide for health care advice due to variability in algorithm performance.7
Utility of AI in Telehealth
Artificial intelligence algorithms could be created to ensure telehealth image accuracy, stratify risk, and track patient progress. With teledermatology visits on the rise during the COVID-19 pandemic, AI algorithms could ensure that photographs of appropriate quality are taken. Also, patients could be organized by risk factors with such algorithms, allowing physicians to save time on triage and stratification. Algorithms also could be used to track a telehealth patient’s treatment and progress.8
Furthermore, there is a need for an algorithm that has the ability to detect, quantify, and monitor changes in dermatologic conditions using images that patients have uploaded. This capability will lead to creation of a standardized quantification scale that will allow physicians to virtually track the progression of visible skin pathologies.
Hazards of Racial Bias in AI
Artificial intelligence is limited by racial disparity bias seen in computerized medicine. For years, the majority of dermatology research, especially in skin cancer, has been conducted on fairer-skinned populations. This bias has existed at the expense of darker-skinned patients, whose skin conditions and symptoms present differently,9 and reflects directly in available data sets that can be used to develop AI algorithms. Because these data are inadequate to the task, AI might misdiagnose skin cancer in people of color or miss an existing condition entirely.10 Consequently, the higher rate of skin cancer mortality that is reported in people of color is likely to persist with the rise of AI in dermatology.11 A more representative database of imaged skin lesions needs to be utilized to create a diversely representative and applicable data set for AI algorithms.12
Benefits of Conversational Agents
Another method by which AI could be incorporated into dermatology is through what is known as a conversational agent (CA)—AI software that engages in a dialogue with users by interpreting their voice and replying to them through text, image, or voice.13 Conversational agents facilitate remote patient management, allow clinicians to focus on other functions, and aid in data collection.14 A 2014 study showed that patients were significantly more likely to disclose history and emotions when informed they were interacting with a CA than with a human clinician (P=.007).15 Such benefits could be invaluable in dermatology, where emotions and patient perceptions of skin conditions play into the treatment process.
However, some evidence showed that CAs cannot respond to patients’ statements in all circumstances.16 It also is unclear how well CAs recognize nuanced statements that might signal potential harm. This fits into the greater theme of a major problem with AI: the lack of a reliable response in all circumstances.13
Final Thoughts
The practical implementations of AI in dermatology are still being explored. Given the uncertainty surrounding the COVID-19 pandemic and the future of patient care, AI might serve as an important asset in assisting with the diagnosis and treatment of dermatologic conditions, physician productivity, and patient monitoring.
- Amisha, Malik P, Pathania M, et al. Overview of artificial intelligence in medicine. J Family Med Prim Care. 2019;8:2328-2331. doi:10.4103/jfmpc.jfmpc_440_19
- Han SS, Kim MS, Lim W, et al. Classification of the clinical images for benign and malignant cutaneous tumors using a deep learning algorithm. J Invest Dermatol. 2018;138:1529-1538. doi:10.1016/j.jid.2018.01.028
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
- Regulated medical device for detecting skin cancer. SkinVision website. Accessed July 23, 2021. https://www.skinvision.com/hcp/
- Thissen M, Udrea A, Hacking M, et al. mHealth app for risk assessment of pigmented and nonpigmented skin lesions—a study on sensitivity and specificity in detecting malignancy. Telemed J E Health. 2017;23:948-954. doi:10.1089/tmj.2016.0259
- Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies. BMJ. 2020;368:m127. doi:10.1136/bmj.m127
- Puri P, Comfere N, Pittelkow MR, et al. COVID-19: an opportunity to build dermatology’s digital future. Dermatol Ther. 2020;33:e14149. doi:10.1111/dth.14149
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59,viii. doi:10.1016/j.det.2011.08.002
- Adamson AS, Smith A. Machine learning and health care disparities in dermatology. JAMA Dermatol. 2018;154:1247-1248. doi:10.1001/jamadermatol.2018.2348
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70:748-762. doi:S0190-9622(13)01296-6
- Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatol Dermatolog Surg. 2019;23:57-60. doi:10.4103/jdds.jdds_19_19
- McGreevey JD 3rd, Hanson CW 3rd, Koppel R. Clinical, legal, and ethical aspects of artificial intelligence-assisted conversational agents in health care. JAMA. 2020;324:552-553. doi:10.1001/jama.2020.2724
- Piau A, Crissey R, Brechemier D, et al. A smartphone chatbot application to optimize monitoring of older patients with cancer. Int J Med Inform. 2019;128:18-23. doi:10.1016/j.ijmedinf.2019.05.013
- Lucas GM, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Comput Human Behav. 2014;37:94-100. https://doi.org/10.1016/j.chb.2014.04.043
- Miner AS, Milstein A, Schueller S, et al. Smartphone-based conversational agents and responses to questions about mental health, interpersonal violence, and physical health. JAMA Intern Med. 2016;176:619-625. doi:10.1001/jamainternmed.2016.0400
With the need to adapt to the given challenges associated with COVID-19, artificial intelligence (AI) serves as a potential tool in providing access to medical-based diagnosis in a novel way. Artificial intelligence is defined as intelligence harnessed by machines that have the ability to perform what is called cognitive thinking and to mimic the problem-solving abilities of the human mind. Virtual AI in dermatology entails neural network–based guidance that includes developing algorithms to detect skin pathology through photographs.1 To use AI in dermatology, recognition of visual patterns must be established to give diagnoses. These neural networks have been used to classify skin diseases, including cancer, actinic keratosis, and warts.2
AI for Skin Cancer
The use of AI to classify melanoma and nonmelanoma skin cancer has been studied extensively, including the following 2 research projects.
Convolutional Neural Network
In 2017, Stanford University published a study in which a deep-learning algorithm known as a convolutional neural network was used to classify skin lesions.3 The network was trained using a dataset of 129,450 clinical images of 2032 diseases. Its performance was compared to that of 21 board-certified dermatologists on biopsy-proven clinical images with 2 classifications of cases: (1) keratinocyte carcinoma as opposed to benign seborrheic keratosis and (2) malignant melanoma as opposed to benign nevi—the first representing the most common skin cancers, and the second, the deadliest skin cancers. The study showed that the machine could accurately identify and classify skin cancers compared to the work of board-certified dermatologists. The study did not include demographic information, which limits its external validity.3
Dermoscopic Image Classification
A 2019 study by Brinker and colleagues4 showed the superiority of automated dermoscopic melanoma image classifications compared to the work of board-certified dermatologists. For the study, 804 biopsy-proven images of melanoma and nevi (1:1 ratio) were randomly presented to dermatologists for their evaluation and recommended treatment (yielding 19,296 recommendations). The dermatologists classified the lesions with a sensitivity of 67.2% and specificity of 62.2%; the trained convolutional neural network attained both higher sensitivity (82.3%) and higher specificity (77.9%).4
Smartphone Diagnosis of Melanoma
An application of AI has been to use smartphone apps for the diagnosis of melanoma. The most utilized and novel algorithm-based smartphone app that assesses skin lesions for malignancy characteristics is SkinVision. With a simple download from Apple’s App Store, this technology allows a person to check their skin spots by taking a photograph and receiving algorithmic risk-assessment feedback. This inexpensive software ($51.78 a year) also allows a patient’s physician to assess the photograph and then validate their assessment by comparing it with the algorithmic analysis that the program provides.5
A review of SkinVision conducted by Thissen and colleagues6 found that, in a hypothetical population of 1000 adults of whom 3% actually had melanoma, 4 of those 30 people would not have been flagged as at “high risk” by SkinVision. There also was a high false-positive rate with the app, with more than 200 people flagged as at high risk. The analysis pegged SkinVision as having a sensitivity of 88% and specificity of 79%.6
In summary, systematic review of diagnostic accuracy has shown that, although there is accuracy in AI analyses, it should be used only as a guide for health care advice due to variability in algorithm performance.7
Utility of AI in Telehealth
Artificial intelligence algorithms could be created to ensure telehealth image accuracy, stratify risk, and track patient progress. With teledermatology visits on the rise during the COVID-19 pandemic, AI algorithms could ensure that photographs of appropriate quality are taken. Also, patients could be organized by risk factors with such algorithms, allowing physicians to save time on triage and stratification. Algorithms also could be used to track a telehealth patient’s treatment and progress.8
Furthermore, there is a need for an algorithm that has the ability to detect, quantify, and monitor changes in dermatologic conditions using images that patients have uploaded. This capability will lead to creation of a standardized quantification scale that will allow physicians to virtually track the progression of visible skin pathologies.
Hazards of Racial Bias in AI
Artificial intelligence is limited by racial disparity bias seen in computerized medicine. For years, the majority of dermatology research, especially in skin cancer, has been conducted on fairer-skinned populations. This bias has existed at the expense of darker-skinned patients, whose skin conditions and symptoms present differently,9 and reflects directly in available data sets that can be used to develop AI algorithms. Because these data are inadequate to the task, AI might misdiagnose skin cancer in people of color or miss an existing condition entirely.10 Consequently, the higher rate of skin cancer mortality that is reported in people of color is likely to persist with the rise of AI in dermatology.11 A more representative database of imaged skin lesions needs to be utilized to create a diversely representative and applicable data set for AI algorithms.12
Benefits of Conversational Agents
Another method by which AI could be incorporated into dermatology is through what is known as a conversational agent (CA)—AI software that engages in a dialogue with users by interpreting their voice and replying to them through text, image, or voice.13 Conversational agents facilitate remote patient management, allow clinicians to focus on other functions, and aid in data collection.14 A 2014 study showed that patients were significantly more likely to disclose history and emotions when informed they were interacting with a CA than with a human clinician (P=.007).15 Such benefits could be invaluable in dermatology, where emotions and patient perceptions of skin conditions play into the treatment process.
However, some evidence showed that CAs cannot respond to patients’ statements in all circumstances.16 It also is unclear how well CAs recognize nuanced statements that might signal potential harm. This fits into the greater theme of a major problem with AI: the lack of a reliable response in all circumstances.13
Final Thoughts
The practical implementations of AI in dermatology are still being explored. Given the uncertainty surrounding the COVID-19 pandemic and the future of patient care, AI might serve as an important asset in assisting with the diagnosis and treatment of dermatologic conditions, physician productivity, and patient monitoring.
With the need to adapt to the given challenges associated with COVID-19, artificial intelligence (AI) serves as a potential tool in providing access to medical-based diagnosis in a novel way. Artificial intelligence is defined as intelligence harnessed by machines that have the ability to perform what is called cognitive thinking and to mimic the problem-solving abilities of the human mind. Virtual AI in dermatology entails neural network–based guidance that includes developing algorithms to detect skin pathology through photographs.1 To use AI in dermatology, recognition of visual patterns must be established to give diagnoses. These neural networks have been used to classify skin diseases, including cancer, actinic keratosis, and warts.2
AI for Skin Cancer
The use of AI to classify melanoma and nonmelanoma skin cancer has been studied extensively, including the following 2 research projects.
Convolutional Neural Network
In 2017, Stanford University published a study in which a deep-learning algorithm known as a convolutional neural network was used to classify skin lesions.3 The network was trained using a dataset of 129,450 clinical images of 2032 diseases. Its performance was compared to that of 21 board-certified dermatologists on biopsy-proven clinical images with 2 classifications of cases: (1) keratinocyte carcinoma as opposed to benign seborrheic keratosis and (2) malignant melanoma as opposed to benign nevi—the first representing the most common skin cancers, and the second, the deadliest skin cancers. The study showed that the machine could accurately identify and classify skin cancers compared to the work of board-certified dermatologists. The study did not include demographic information, which limits its external validity.3
Dermoscopic Image Classification
A 2019 study by Brinker and colleagues4 showed the superiority of automated dermoscopic melanoma image classifications compared to the work of board-certified dermatologists. For the study, 804 biopsy-proven images of melanoma and nevi (1:1 ratio) were randomly presented to dermatologists for their evaluation and recommended treatment (yielding 19,296 recommendations). The dermatologists classified the lesions with a sensitivity of 67.2% and specificity of 62.2%; the trained convolutional neural network attained both higher sensitivity (82.3%) and higher specificity (77.9%).4
Smartphone Diagnosis of Melanoma
An application of AI has been to use smartphone apps for the diagnosis of melanoma. The most utilized and novel algorithm-based smartphone app that assesses skin lesions for malignancy characteristics is SkinVision. With a simple download from Apple’s App Store, this technology allows a person to check their skin spots by taking a photograph and receiving algorithmic risk-assessment feedback. This inexpensive software ($51.78 a year) also allows a patient’s physician to assess the photograph and then validate their assessment by comparing it with the algorithmic analysis that the program provides.5
A review of SkinVision conducted by Thissen and colleagues6 found that, in a hypothetical population of 1000 adults of whom 3% actually had melanoma, 4 of those 30 people would not have been flagged as at “high risk” by SkinVision. There also was a high false-positive rate with the app, with more than 200 people flagged as at high risk. The analysis pegged SkinVision as having a sensitivity of 88% and specificity of 79%.6
In summary, systematic review of diagnostic accuracy has shown that, although there is accuracy in AI analyses, it should be used only as a guide for health care advice due to variability in algorithm performance.7
Utility of AI in Telehealth
Artificial intelligence algorithms could be created to ensure telehealth image accuracy, stratify risk, and track patient progress. With teledermatology visits on the rise during the COVID-19 pandemic, AI algorithms could ensure that photographs of appropriate quality are taken. Also, patients could be organized by risk factors with such algorithms, allowing physicians to save time on triage and stratification. Algorithms also could be used to track a telehealth patient’s treatment and progress.8
Furthermore, there is a need for an algorithm that has the ability to detect, quantify, and monitor changes in dermatologic conditions using images that patients have uploaded. This capability will lead to creation of a standardized quantification scale that will allow physicians to virtually track the progression of visible skin pathologies.
Hazards of Racial Bias in AI
Artificial intelligence is limited by racial disparity bias seen in computerized medicine. For years, the majority of dermatology research, especially in skin cancer, has been conducted on fairer-skinned populations. This bias has existed at the expense of darker-skinned patients, whose skin conditions and symptoms present differently,9 and reflects directly in available data sets that can be used to develop AI algorithms. Because these data are inadequate to the task, AI might misdiagnose skin cancer in people of color or miss an existing condition entirely.10 Consequently, the higher rate of skin cancer mortality that is reported in people of color is likely to persist with the rise of AI in dermatology.11 A more representative database of imaged skin lesions needs to be utilized to create a diversely representative and applicable data set for AI algorithms.12
Benefits of Conversational Agents
Another method by which AI could be incorporated into dermatology is through what is known as a conversational agent (CA)—AI software that engages in a dialogue with users by interpreting their voice and replying to them through text, image, or voice.13 Conversational agents facilitate remote patient management, allow clinicians to focus on other functions, and aid in data collection.14 A 2014 study showed that patients were significantly more likely to disclose history and emotions when informed they were interacting with a CA than with a human clinician (P=.007).15 Such benefits could be invaluable in dermatology, where emotions and patient perceptions of skin conditions play into the treatment process.
However, some evidence showed that CAs cannot respond to patients’ statements in all circumstances.16 It also is unclear how well CAs recognize nuanced statements that might signal potential harm. This fits into the greater theme of a major problem with AI: the lack of a reliable response in all circumstances.13
Final Thoughts
The practical implementations of AI in dermatology are still being explored. Given the uncertainty surrounding the COVID-19 pandemic and the future of patient care, AI might serve as an important asset in assisting with the diagnosis and treatment of dermatologic conditions, physician productivity, and patient monitoring.
- Amisha, Malik P, Pathania M, et al. Overview of artificial intelligence in medicine. J Family Med Prim Care. 2019;8:2328-2331. doi:10.4103/jfmpc.jfmpc_440_19
- Han SS, Kim MS, Lim W, et al. Classification of the clinical images for benign and malignant cutaneous tumors using a deep learning algorithm. J Invest Dermatol. 2018;138:1529-1538. doi:10.1016/j.jid.2018.01.028
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
- Regulated medical device for detecting skin cancer. SkinVision website. Accessed July 23, 2021. https://www.skinvision.com/hcp/
- Thissen M, Udrea A, Hacking M, et al. mHealth app for risk assessment of pigmented and nonpigmented skin lesions—a study on sensitivity and specificity in detecting malignancy. Telemed J E Health. 2017;23:948-954. doi:10.1089/tmj.2016.0259
- Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies. BMJ. 2020;368:m127. doi:10.1136/bmj.m127
- Puri P, Comfere N, Pittelkow MR, et al. COVID-19: an opportunity to build dermatology’s digital future. Dermatol Ther. 2020;33:e14149. doi:10.1111/dth.14149
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59,viii. doi:10.1016/j.det.2011.08.002
- Adamson AS, Smith A. Machine learning and health care disparities in dermatology. JAMA Dermatol. 2018;154:1247-1248. doi:10.1001/jamadermatol.2018.2348
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70:748-762. doi:S0190-9622(13)01296-6
- Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatol Dermatolog Surg. 2019;23:57-60. doi:10.4103/jdds.jdds_19_19
- McGreevey JD 3rd, Hanson CW 3rd, Koppel R. Clinical, legal, and ethical aspects of artificial intelligence-assisted conversational agents in health care. JAMA. 2020;324:552-553. doi:10.1001/jama.2020.2724
- Piau A, Crissey R, Brechemier D, et al. A smartphone chatbot application to optimize monitoring of older patients with cancer. Int J Med Inform. 2019;128:18-23. doi:10.1016/j.ijmedinf.2019.05.013
- Lucas GM, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Comput Human Behav. 2014;37:94-100. https://doi.org/10.1016/j.chb.2014.04.043
- Miner AS, Milstein A, Schueller S, et al. Smartphone-based conversational agents and responses to questions about mental health, interpersonal violence, and physical health. JAMA Intern Med. 2016;176:619-625. doi:10.1001/jamainternmed.2016.0400
- Amisha, Malik P, Pathania M, et al. Overview of artificial intelligence in medicine. J Family Med Prim Care. 2019;8:2328-2331. doi:10.4103/jfmpc.jfmpc_440_19
- Han SS, Kim MS, Lim W, et al. Classification of the clinical images for benign and malignant cutaneous tumors using a deep learning algorithm. J Invest Dermatol. 2018;138:1529-1538. doi:10.1016/j.jid.2018.01.028
- Esteva A, Kuprel B, Novoa RA, et al. Dermatologist-level classification of skin cancer with deep neural networks. Nature. 2017;542:115-118. doi:10.1038/nature21056
- Brinker TJ, Hekler A, Enk AH, et al. Deep neural networks are superior to dermatologists in melanoma image classification. Eur J Cancer. 2019;119:11-17. doi:10.1016/j.ejca.2019.05.023
- Regulated medical device for detecting skin cancer. SkinVision website. Accessed July 23, 2021. https://www.skinvision.com/hcp/
- Thissen M, Udrea A, Hacking M, et al. mHealth app for risk assessment of pigmented and nonpigmented skin lesions—a study on sensitivity and specificity in detecting malignancy. Telemed J E Health. 2017;23:948-954. doi:10.1089/tmj.2016.0259
- Freeman K, Dinnes J, Chuchu N, et al. Algorithm based smartphone apps to assess risk of skin cancer in adults: systematic review of diagnostic accuracy studies. BMJ. 2020;368:m127. doi:10.1136/bmj.m127
- Puri P, Comfere N, Pittelkow MR, et al. COVID-19: an opportunity to build dermatology’s digital future. Dermatol Ther. 2020;33:e14149. doi:10.1111/dth.14149
- Buster KJ, Stevens EI, Elmets CA. Dermatologic health disparities. Dermatol Clin. 2012;30:53-59,viii. doi:10.1016/j.det.2011.08.002
- Adamson AS, Smith A. Machine learning and health care disparities in dermatology. JAMA Dermatol. 2018;154:1247-1248. doi:10.1001/jamadermatol.2018.2348
- Agbai ON, Buster K, Sanchez M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol. 2014;70:748-762. doi:S0190-9622(13)01296-6
- Alabdulkareem A. Artificial intelligence and dermatologists: friends or foes? J Dermatol Dermatolog Surg. 2019;23:57-60. doi:10.4103/jdds.jdds_19_19
- McGreevey JD 3rd, Hanson CW 3rd, Koppel R. Clinical, legal, and ethical aspects of artificial intelligence-assisted conversational agents in health care. JAMA. 2020;324:552-553. doi:10.1001/jama.2020.2724
- Piau A, Crissey R, Brechemier D, et al. A smartphone chatbot application to optimize monitoring of older patients with cancer. Int J Med Inform. 2019;128:18-23. doi:10.1016/j.ijmedinf.2019.05.013
- Lucas GM, Gratch J, King A, et al. It’s only a computer: virtual humans increase willingness to disclose. Comput Human Behav. 2014;37:94-100. https://doi.org/10.1016/j.chb.2014.04.043
- Miner AS, Milstein A, Schueller S, et al. Smartphone-based conversational agents and responses to questions about mental health, interpersonal violence, and physical health. JAMA Intern Med. 2016;176:619-625. doi:10.1001/jamainternmed.2016.0400
Practice Points
- Dermatologists should amass pictures of dermatologic conditions in skin of color to contribute to growing awareness and knowledge of presentation of disease in this population.
- Dermatologists should use artificial intelligence as a tool for delivering more efficient and beneficial patient care.
Medical residents need breastfeeding support too
As working mothers with babies in tow when the COVID-19 crisis struck, countless uncertainties threatened our already precarious work-life balance. We suddenly had many questions:
“If my daycare closes, what will I do for childcare?”
“How do I navigate diaper changes, feedings, and naps with my hectic remote work schedule?”
“If I’m constantly interrupted during the day, should I skip sleep to catch up on work and not let my colleagues down?”
As professionals who work closely with medical trainees, we knew our parenting dilemmas were being experienced even more acutely by our frontline worker colleagues.
Medical training is an increasingly common time to start a family. In a recent study, 34% of trainees in Harvard-affiliated residency programs became parents during training, and another 52% planned to do so. Trainees have higher breastfeeding initiation rates but lower continuation rates than the general population. Early nursing cessation among trainees is well documented nationally and is most often attributed to work-related barriers. These barriers range from insufficient time and limited access to facilities to a lack of support and discrimination by supervisors and peers.
This trend does not discriminate by specialty. Even among training programs known to be “family friendly,” the average duration of nursing is just 4.5 months. Residents of color are disproportionately affected by inadequate support. Studies show that Black parents breastfeed at lower rates than White parents. This has been largely attributed to structural racism and implicit bias, such as Black parents receiving less assistance initiating nursing after delivery. Adequate lactation support and inclusivity are also lacking for transgender parents who choose to breastfeed or chestfeed.
The very nature of residency training, which includes shifts that can span more than 24 hours, conflicts with many health-promoting behaviors like sleeping and eating well. However, its interference with lactation is correlated with gender. Women are disproportionately affected by the negative outcomes of unmet lactation goals. These include work-life imbalance, career dissatisfaction, and negative emotions. In a study of pediatric residents, one in four did not achieve their breastfeeding goals. Respondents reported feeling “sad, devastated, defeated, disappointed, guilty, embarrassed, frustrated, angry, like a failure, and inadequate.” Among physician mothers more broadly, discrimination related to pregnancy, parental leave, and nursing is associated with higher self-reported burnout.
Navigating nursing during residency training has more than just emotional and psychological consequences – it also has professional ones. Pursuing personal lactation goals can delay residency program completion and board certification, influence specialty selection, negatively impact research productivity, impede career advancement, and lead to misgivings about career choice.
Trainees and their families are not the only ones harmed by inadequate support in residency programs. Patients and their families are affected, too. Research suggests that physicians’ personal breastfeeding practices affect the advice they give to patients. Those who receive lactation support are more likely to help patients meet their own goals. In the previously mentioned study of pediatric residents, more than 90% of the 400 respondents said their own or their partner’s nursing experience affected their interaction with lactating patients in their clinic or hospital.
Increased lactation support is a straightforward, low-cost, high-impact intervention. It benefits trainee well-being, satisfaction, workflow, and future patient care. The Accreditation Council for Graduate Medical Education mandated in July 2019 that all residency programs provide adequate lactation facilities – including refrigeration capabilities and proximity for safe patient care. However, to our knowledge, rates of compliance with this new policy and citation for noncompliance have yet to be seen. Regardless, facilities alone are not enough. Residency programs should develop and enforce formal lactation policies.
Several institutions have successfully piloted such policies in recent years. One in particular from the University of Michigan’s surgery residency program inspired the development of a lactation policy within the internal medicine residency at our institution. These policies designate appropriate spaces at each clinical rotation site, clarify that residents are encouraged to take pumping breaks as needed – in coordination with clinical teams so as not to compromise patient care – and communicate support from supervisors.
Our program also established an informal peer mentoring program. Residents with experience pumping at work pair up with newer trainees. The policy benefits residents who wish to chestfeed or breastfeed, normalizes lactation, and empowers trainees by diminishing the need to ask for individual accommodations. It also costs the program nothing.
As more women enter medicine and more trainees become parents during residency, the need for support in this area will only continue to grow. The widespread lack of such resources, and the fact that clean and private facilities are only now being mandated, is symbolic. If even this basic need is rarely acknowledged or met, what other resident needs are being neglected?
A version of this article first appeared on Medscape.com.
As working mothers with babies in tow when the COVID-19 crisis struck, countless uncertainties threatened our already precarious work-life balance. We suddenly had many questions:
“If my daycare closes, what will I do for childcare?”
“How do I navigate diaper changes, feedings, and naps with my hectic remote work schedule?”
“If I’m constantly interrupted during the day, should I skip sleep to catch up on work and not let my colleagues down?”
As professionals who work closely with medical trainees, we knew our parenting dilemmas were being experienced even more acutely by our frontline worker colleagues.
Medical training is an increasingly common time to start a family. In a recent study, 34% of trainees in Harvard-affiliated residency programs became parents during training, and another 52% planned to do so. Trainees have higher breastfeeding initiation rates but lower continuation rates than the general population. Early nursing cessation among trainees is well documented nationally and is most often attributed to work-related barriers. These barriers range from insufficient time and limited access to facilities to a lack of support and discrimination by supervisors and peers.
This trend does not discriminate by specialty. Even among training programs known to be “family friendly,” the average duration of nursing is just 4.5 months. Residents of color are disproportionately affected by inadequate support. Studies show that Black parents breastfeed at lower rates than White parents. This has been largely attributed to structural racism and implicit bias, such as Black parents receiving less assistance initiating nursing after delivery. Adequate lactation support and inclusivity are also lacking for transgender parents who choose to breastfeed or chestfeed.
The very nature of residency training, which includes shifts that can span more than 24 hours, conflicts with many health-promoting behaviors like sleeping and eating well. However, its interference with lactation is correlated with gender. Women are disproportionately affected by the negative outcomes of unmet lactation goals. These include work-life imbalance, career dissatisfaction, and negative emotions. In a study of pediatric residents, one in four did not achieve their breastfeeding goals. Respondents reported feeling “sad, devastated, defeated, disappointed, guilty, embarrassed, frustrated, angry, like a failure, and inadequate.” Among physician mothers more broadly, discrimination related to pregnancy, parental leave, and nursing is associated with higher self-reported burnout.
Navigating nursing during residency training has more than just emotional and psychological consequences – it also has professional ones. Pursuing personal lactation goals can delay residency program completion and board certification, influence specialty selection, negatively impact research productivity, impede career advancement, and lead to misgivings about career choice.
Trainees and their families are not the only ones harmed by inadequate support in residency programs. Patients and their families are affected, too. Research suggests that physicians’ personal breastfeeding practices affect the advice they give to patients. Those who receive lactation support are more likely to help patients meet their own goals. In the previously mentioned study of pediatric residents, more than 90% of the 400 respondents said their own or their partner’s nursing experience affected their interaction with lactating patients in their clinic or hospital.
Increased lactation support is a straightforward, low-cost, high-impact intervention. It benefits trainee well-being, satisfaction, workflow, and future patient care. The Accreditation Council for Graduate Medical Education mandated in July 2019 that all residency programs provide adequate lactation facilities – including refrigeration capabilities and proximity for safe patient care. However, to our knowledge, rates of compliance with this new policy and citation for noncompliance have yet to be seen. Regardless, facilities alone are not enough. Residency programs should develop and enforce formal lactation policies.
Several institutions have successfully piloted such policies in recent years. One in particular from the University of Michigan’s surgery residency program inspired the development of a lactation policy within the internal medicine residency at our institution. These policies designate appropriate spaces at each clinical rotation site, clarify that residents are encouraged to take pumping breaks as needed – in coordination with clinical teams so as not to compromise patient care – and communicate support from supervisors.
Our program also established an informal peer mentoring program. Residents with experience pumping at work pair up with newer trainees. The policy benefits residents who wish to chestfeed or breastfeed, normalizes lactation, and empowers trainees by diminishing the need to ask for individual accommodations. It also costs the program nothing.
As more women enter medicine and more trainees become parents during residency, the need for support in this area will only continue to grow. The widespread lack of such resources, and the fact that clean and private facilities are only now being mandated, is symbolic. If even this basic need is rarely acknowledged or met, what other resident needs are being neglected?
A version of this article first appeared on Medscape.com.
As working mothers with babies in tow when the COVID-19 crisis struck, countless uncertainties threatened our already precarious work-life balance. We suddenly had many questions:
“If my daycare closes, what will I do for childcare?”
“How do I navigate diaper changes, feedings, and naps with my hectic remote work schedule?”
“If I’m constantly interrupted during the day, should I skip sleep to catch up on work and not let my colleagues down?”
As professionals who work closely with medical trainees, we knew our parenting dilemmas were being experienced even more acutely by our frontline worker colleagues.
Medical training is an increasingly common time to start a family. In a recent study, 34% of trainees in Harvard-affiliated residency programs became parents during training, and another 52% planned to do so. Trainees have higher breastfeeding initiation rates but lower continuation rates than the general population. Early nursing cessation among trainees is well documented nationally and is most often attributed to work-related barriers. These barriers range from insufficient time and limited access to facilities to a lack of support and discrimination by supervisors and peers.
This trend does not discriminate by specialty. Even among training programs known to be “family friendly,” the average duration of nursing is just 4.5 months. Residents of color are disproportionately affected by inadequate support. Studies show that Black parents breastfeed at lower rates than White parents. This has been largely attributed to structural racism and implicit bias, such as Black parents receiving less assistance initiating nursing after delivery. Adequate lactation support and inclusivity are also lacking for transgender parents who choose to breastfeed or chestfeed.
The very nature of residency training, which includes shifts that can span more than 24 hours, conflicts with many health-promoting behaviors like sleeping and eating well. However, its interference with lactation is correlated with gender. Women are disproportionately affected by the negative outcomes of unmet lactation goals. These include work-life imbalance, career dissatisfaction, and negative emotions. In a study of pediatric residents, one in four did not achieve their breastfeeding goals. Respondents reported feeling “sad, devastated, defeated, disappointed, guilty, embarrassed, frustrated, angry, like a failure, and inadequate.” Among physician mothers more broadly, discrimination related to pregnancy, parental leave, and nursing is associated with higher self-reported burnout.
Navigating nursing during residency training has more than just emotional and psychological consequences – it also has professional ones. Pursuing personal lactation goals can delay residency program completion and board certification, influence specialty selection, negatively impact research productivity, impede career advancement, and lead to misgivings about career choice.
Trainees and their families are not the only ones harmed by inadequate support in residency programs. Patients and their families are affected, too. Research suggests that physicians’ personal breastfeeding practices affect the advice they give to patients. Those who receive lactation support are more likely to help patients meet their own goals. In the previously mentioned study of pediatric residents, more than 90% of the 400 respondents said their own or their partner’s nursing experience affected their interaction with lactating patients in their clinic or hospital.
Increased lactation support is a straightforward, low-cost, high-impact intervention. It benefits trainee well-being, satisfaction, workflow, and future patient care. The Accreditation Council for Graduate Medical Education mandated in July 2019 that all residency programs provide adequate lactation facilities – including refrigeration capabilities and proximity for safe patient care. However, to our knowledge, rates of compliance with this new policy and citation for noncompliance have yet to be seen. Regardless, facilities alone are not enough. Residency programs should develop and enforce formal lactation policies.
Several institutions have successfully piloted such policies in recent years. One in particular from the University of Michigan’s surgery residency program inspired the development of a lactation policy within the internal medicine residency at our institution. These policies designate appropriate spaces at each clinical rotation site, clarify that residents are encouraged to take pumping breaks as needed – in coordination with clinical teams so as not to compromise patient care – and communicate support from supervisors.
Our program also established an informal peer mentoring program. Residents with experience pumping at work pair up with newer trainees. The policy benefits residents who wish to chestfeed or breastfeed, normalizes lactation, and empowers trainees by diminishing the need to ask for individual accommodations. It also costs the program nothing.
As more women enter medicine and more trainees become parents during residency, the need for support in this area will only continue to grow. The widespread lack of such resources, and the fact that clean and private facilities are only now being mandated, is symbolic. If even this basic need is rarely acknowledged or met, what other resident needs are being neglected?
A version of this article first appeared on Medscape.com.
A sizzling hybrid meeting of the Society of Gynecologic Surgeons
The 47th Annual Scientific Meeting of the Society of Gynecologic Surgeons (SGS), like so many things in our modern world, endured many changes and had to stay nimble and evolve to changing times. In the end, however, SGS was able to adapt and succeed, just like a skilled gynecologic surgeon in the operating room, to deliver a fresh new type of meeting.
When we chose the meeting theme, “Working together: How collaboration enables us to better help our patients,” we anticipated a meeting discussing medical colleagues and consultants. In our forever-changed world, we knew we needed to reinterpret this to a broader social context. Our special lectures and panel discussions sought to open attendees’ eyes to disparities in health care for people of color and women.
While we highlighted the realities faced by colleagues in medicine, the topics addressed also were designed to grow awareness about struggles our patients encounter as well. Social disparities are sobering, long-standing, and sometimes require creative collaborations to achieve successful outcomes for all patients. The faculty of one of our postgraduate courses reviews in this special 2-part section to
The meeting also kicked off with a postgraduate course on fibroid management, with workshops on harnessing the power of social media and lessons on leadership from a female Fortune 500 CEO, Lori Ryerkerk, offered as well. As the scientific program launched, we were once again treated to strong science on gynecologic surgery, with only a small dip in abstract submissions, despite the challenges of research during a pandemic. Mark Walters, MD, gave the inaugural lecture in his name on the crucial topic of surgical education and teaching. We also heard a special report from the SGS SOCOVID research group, led by Dr. Rosanne Kho, on gynecologic surgery during the pandemic. We also convened a virtual panel for our hybrid attendees on the benefits to patients of a multidisciplinary approach to gynecologic surgery, presented here by Cecile Ferrando, MD.
As our practices continue to grow and evolve, the introduction of innovative technologies can pose a new challenge, as Miles Murphy, MD, and members of the panel on novel gynecologic office procedures will present in this series next month.
The TeLinde keynote speaker was Janet Dombrowski, who works as a coach for many surgeons in various disciplines across the country. She spoke to the resilience gained through community and collaboration.
While our meeting theme dated to the “before” pandemic era, those who were able to be in attendance in person can attest to the value we can all place now on community and personal interactions. With experience strengthened by science, I hope this meeting summary serves to highlight the many ways in which we can collaborate to improve outcomes for ourselves in medicine and for patients.
The 47th Annual Scientific Meeting of the Society of Gynecologic Surgeons (SGS), like so many things in our modern world, endured many changes and had to stay nimble and evolve to changing times. In the end, however, SGS was able to adapt and succeed, just like a skilled gynecologic surgeon in the operating room, to deliver a fresh new type of meeting.
When we chose the meeting theme, “Working together: How collaboration enables us to better help our patients,” we anticipated a meeting discussing medical colleagues and consultants. In our forever-changed world, we knew we needed to reinterpret this to a broader social context. Our special lectures and panel discussions sought to open attendees’ eyes to disparities in health care for people of color and women.
While we highlighted the realities faced by colleagues in medicine, the topics addressed also were designed to grow awareness about struggles our patients encounter as well. Social disparities are sobering, long-standing, and sometimes require creative collaborations to achieve successful outcomes for all patients. The faculty of one of our postgraduate courses reviews in this special 2-part section to
The meeting also kicked off with a postgraduate course on fibroid management, with workshops on harnessing the power of social media and lessons on leadership from a female Fortune 500 CEO, Lori Ryerkerk, offered as well. As the scientific program launched, we were once again treated to strong science on gynecologic surgery, with only a small dip in abstract submissions, despite the challenges of research during a pandemic. Mark Walters, MD, gave the inaugural lecture in his name on the crucial topic of surgical education and teaching. We also heard a special report from the SGS SOCOVID research group, led by Dr. Rosanne Kho, on gynecologic surgery during the pandemic. We also convened a virtual panel for our hybrid attendees on the benefits to patients of a multidisciplinary approach to gynecologic surgery, presented here by Cecile Ferrando, MD.
As our practices continue to grow and evolve, the introduction of innovative technologies can pose a new challenge, as Miles Murphy, MD, and members of the panel on novel gynecologic office procedures will present in this series next month.
The TeLinde keynote speaker was Janet Dombrowski, who works as a coach for many surgeons in various disciplines across the country. She spoke to the resilience gained through community and collaboration.
While our meeting theme dated to the “before” pandemic era, those who were able to be in attendance in person can attest to the value we can all place now on community and personal interactions. With experience strengthened by science, I hope this meeting summary serves to highlight the many ways in which we can collaborate to improve outcomes for ourselves in medicine and for patients.
The 47th Annual Scientific Meeting of the Society of Gynecologic Surgeons (SGS), like so many things in our modern world, endured many changes and had to stay nimble and evolve to changing times. In the end, however, SGS was able to adapt and succeed, just like a skilled gynecologic surgeon in the operating room, to deliver a fresh new type of meeting.
When we chose the meeting theme, “Working together: How collaboration enables us to better help our patients,” we anticipated a meeting discussing medical colleagues and consultants. In our forever-changed world, we knew we needed to reinterpret this to a broader social context. Our special lectures and panel discussions sought to open attendees’ eyes to disparities in health care for people of color and women.
While we highlighted the realities faced by colleagues in medicine, the topics addressed also were designed to grow awareness about struggles our patients encounter as well. Social disparities are sobering, long-standing, and sometimes require creative collaborations to achieve successful outcomes for all patients. The faculty of one of our postgraduate courses reviews in this special 2-part section to
The meeting also kicked off with a postgraduate course on fibroid management, with workshops on harnessing the power of social media and lessons on leadership from a female Fortune 500 CEO, Lori Ryerkerk, offered as well. As the scientific program launched, we were once again treated to strong science on gynecologic surgery, with only a small dip in abstract submissions, despite the challenges of research during a pandemic. Mark Walters, MD, gave the inaugural lecture in his name on the crucial topic of surgical education and teaching. We also heard a special report from the SGS SOCOVID research group, led by Dr. Rosanne Kho, on gynecologic surgery during the pandemic. We also convened a virtual panel for our hybrid attendees on the benefits to patients of a multidisciplinary approach to gynecologic surgery, presented here by Cecile Ferrando, MD.
As our practices continue to grow and evolve, the introduction of innovative technologies can pose a new challenge, as Miles Murphy, MD, and members of the panel on novel gynecologic office procedures will present in this series next month.
The TeLinde keynote speaker was Janet Dombrowski, who works as a coach for many surgeons in various disciplines across the country. She spoke to the resilience gained through community and collaboration.
While our meeting theme dated to the “before” pandemic era, those who were able to be in attendance in person can attest to the value we can all place now on community and personal interactions. With experience strengthened by science, I hope this meeting summary serves to highlight the many ways in which we can collaborate to improve outcomes for ourselves in medicine and for patients.
Physicians wearing white coats rated more experienced
Physicians wearing white coats were rated as significantly more experienced and professional than peers wearing casual attire. Regardless of their attire, however, female physicians were more likely to be judged as appearing less professional and were more likely to be misidentified as medical technicians, physician assistants, or nurses, found research published in JAMA Network Open.
“A white coat with scrubs attire was most preferred for surgeons (mean preference index, 1.3), whereas a white coat with business attire was preferred for family physicians and dermatologists (mean preference indexes, 1.6 and 1.2, respectively; P < .001),” Helen Xun, MD, Johns Hopkins University, Baltimore, and colleagues wrote. “A male model wearing business inner wear with a white coat, fleece jacket, or softshell jacket was perceived as significantly more professional than a female model wearing the same attire (mean professionalism score: male, 65.8; female, 56.2; mean difference in professionalism score: white coat, 12.06; fleece, 7.89; softshell, 8.82; P < .001). ... A male model wearing hospital scrubs or fashion scrubs alone was also perceived as more professional than a female model in the same attire.”
While casual attire, such as fleece or softshell jackets emblazoned with the names of the institution and wearer, has become more popular attire for physicians in recent years, the researchers noted theirs is the first published research to identify associations between gender, attire, and how people distinguish between various health care roles. The study authors launched their web-based survey from May to June 2020 and asked people aged 18 years and older to rate a series of photographs of deidentified models wearing health care attire. Inner wear choices were business attire versus scrubs with and without outer wear options of a long white coat, gray fleece jacket, or black softshell jackets. Survey respondents ranked the images on a 6-point Likert scale with 1 being the least experienced, professional, and friendly and 6 being the most experienced, professional, and friendly. Survey respondents also viewed individual images of male or female models and were asked to rate their professionalism on a scale of 0-100 – with 100 as the “most professional” as well as to identify their profession as either physician, surgeon, nurse, medical technician, or physician assistant.
The study team included 487 (93.3%) of 522 completed surveys in their analyses. Respondents’ mean age was 36.2 years; 260 (53.4%) were female; 372 (76.4%) were White; 33 (6.8%) were Black or African American. Younger respondents and those living in the Western United States who had more exposure to physician casual attire appeared more accepting of it, the authors wrote.
“I remember attending my white-coat ceremony as a medical student, and the symbolism of it all representing me entering the profession. It felt very emotional and heavy and I felt very proud to be there. I also remember taking a ‘selfie’ in my long white coat as a doctor for the first time before my first shift as a resident. But, I’ve also been wearing that same white coat, and a large badge with a ‘DOCTOR’ label on it, and been mistaken by a patient or parent for something other than the physician,” Alexandra M. Sims, a pediatrician and health equity researcher in Cincinnati, said in an interview. “So, I’d really hope that the take-home here is not simply that we must wear our white coats to be considered more professional. I think we have to unpack and dismantle how we’ve even built this notion of ‘professionalism’ in the first place. Women, people of color, and other marginalized groups were certainly not a part of the defining, but we must be a part of the reimagining of an equitable health care profession in this new era.”
As sartorial trends usher in more casual attire, clinicians should redouble efforts to build rapport and enhance communication with patients, such as clarifying team members’ roles when introducing themselves. Dr. Xun and coauthors noted that addressing gender bias is important for all clinicians – not just women – and point to the need for institutional and organizational support for disciplines where gender bias is “especially prevalent,” like surgery. “This responsibility should not be undertaken only by the individuals that experience the biases, which may result in additional cumulative career disadvantages. The promotion of equality and diversity begins with recognition, characterization, and evidence-supported interventions and is a community operation,” Dr. Xun and colleagues concluded.
“I do not equate attire to professionalism or experience, nor is it connected to my satisfaction with the physician. For myself and my daughter, it is the experience of care that ultimately influences our perceptions regarding the professionalism of the physician,” Hala H. Durrah, MTA, parent to a chronically ill child with special health care needs and a Patient and Family Engagement Consultant, said in an interview. “My respect for a physician will ultimately be determined by how my daughter and I were treated, not just from a clinical perspective, but how we felt during those interactions.”
Physicians wearing white coats were rated as significantly more experienced and professional than peers wearing casual attire. Regardless of their attire, however, female physicians were more likely to be judged as appearing less professional and were more likely to be misidentified as medical technicians, physician assistants, or nurses, found research published in JAMA Network Open.
“A white coat with scrubs attire was most preferred for surgeons (mean preference index, 1.3), whereas a white coat with business attire was preferred for family physicians and dermatologists (mean preference indexes, 1.6 and 1.2, respectively; P < .001),” Helen Xun, MD, Johns Hopkins University, Baltimore, and colleagues wrote. “A male model wearing business inner wear with a white coat, fleece jacket, or softshell jacket was perceived as significantly more professional than a female model wearing the same attire (mean professionalism score: male, 65.8; female, 56.2; mean difference in professionalism score: white coat, 12.06; fleece, 7.89; softshell, 8.82; P < .001). ... A male model wearing hospital scrubs or fashion scrubs alone was also perceived as more professional than a female model in the same attire.”
While casual attire, such as fleece or softshell jackets emblazoned with the names of the institution and wearer, has become more popular attire for physicians in recent years, the researchers noted theirs is the first published research to identify associations between gender, attire, and how people distinguish between various health care roles. The study authors launched their web-based survey from May to June 2020 and asked people aged 18 years and older to rate a series of photographs of deidentified models wearing health care attire. Inner wear choices were business attire versus scrubs with and without outer wear options of a long white coat, gray fleece jacket, or black softshell jackets. Survey respondents ranked the images on a 6-point Likert scale with 1 being the least experienced, professional, and friendly and 6 being the most experienced, professional, and friendly. Survey respondents also viewed individual images of male or female models and were asked to rate their professionalism on a scale of 0-100 – with 100 as the “most professional” as well as to identify their profession as either physician, surgeon, nurse, medical technician, or physician assistant.
The study team included 487 (93.3%) of 522 completed surveys in their analyses. Respondents’ mean age was 36.2 years; 260 (53.4%) were female; 372 (76.4%) were White; 33 (6.8%) were Black or African American. Younger respondents and those living in the Western United States who had more exposure to physician casual attire appeared more accepting of it, the authors wrote.
“I remember attending my white-coat ceremony as a medical student, and the symbolism of it all representing me entering the profession. It felt very emotional and heavy and I felt very proud to be there. I also remember taking a ‘selfie’ in my long white coat as a doctor for the first time before my first shift as a resident. But, I’ve also been wearing that same white coat, and a large badge with a ‘DOCTOR’ label on it, and been mistaken by a patient or parent for something other than the physician,” Alexandra M. Sims, a pediatrician and health equity researcher in Cincinnati, said in an interview. “So, I’d really hope that the take-home here is not simply that we must wear our white coats to be considered more professional. I think we have to unpack and dismantle how we’ve even built this notion of ‘professionalism’ in the first place. Women, people of color, and other marginalized groups were certainly not a part of the defining, but we must be a part of the reimagining of an equitable health care profession in this new era.”
As sartorial trends usher in more casual attire, clinicians should redouble efforts to build rapport and enhance communication with patients, such as clarifying team members’ roles when introducing themselves. Dr. Xun and coauthors noted that addressing gender bias is important for all clinicians – not just women – and point to the need for institutional and organizational support for disciplines where gender bias is “especially prevalent,” like surgery. “This responsibility should not be undertaken only by the individuals that experience the biases, which may result in additional cumulative career disadvantages. The promotion of equality and diversity begins with recognition, characterization, and evidence-supported interventions and is a community operation,” Dr. Xun and colleagues concluded.
“I do not equate attire to professionalism or experience, nor is it connected to my satisfaction with the physician. For myself and my daughter, it is the experience of care that ultimately influences our perceptions regarding the professionalism of the physician,” Hala H. Durrah, MTA, parent to a chronically ill child with special health care needs and a Patient and Family Engagement Consultant, said in an interview. “My respect for a physician will ultimately be determined by how my daughter and I were treated, not just from a clinical perspective, but how we felt during those interactions.”
Physicians wearing white coats were rated as significantly more experienced and professional than peers wearing casual attire. Regardless of their attire, however, female physicians were more likely to be judged as appearing less professional and were more likely to be misidentified as medical technicians, physician assistants, or nurses, found research published in JAMA Network Open.
“A white coat with scrubs attire was most preferred for surgeons (mean preference index, 1.3), whereas a white coat with business attire was preferred for family physicians and dermatologists (mean preference indexes, 1.6 and 1.2, respectively; P < .001),” Helen Xun, MD, Johns Hopkins University, Baltimore, and colleagues wrote. “A male model wearing business inner wear with a white coat, fleece jacket, or softshell jacket was perceived as significantly more professional than a female model wearing the same attire (mean professionalism score: male, 65.8; female, 56.2; mean difference in professionalism score: white coat, 12.06; fleece, 7.89; softshell, 8.82; P < .001). ... A male model wearing hospital scrubs or fashion scrubs alone was also perceived as more professional than a female model in the same attire.”
While casual attire, such as fleece or softshell jackets emblazoned with the names of the institution and wearer, has become more popular attire for physicians in recent years, the researchers noted theirs is the first published research to identify associations between gender, attire, and how people distinguish between various health care roles. The study authors launched their web-based survey from May to June 2020 and asked people aged 18 years and older to rate a series of photographs of deidentified models wearing health care attire. Inner wear choices were business attire versus scrubs with and without outer wear options of a long white coat, gray fleece jacket, or black softshell jackets. Survey respondents ranked the images on a 6-point Likert scale with 1 being the least experienced, professional, and friendly and 6 being the most experienced, professional, and friendly. Survey respondents also viewed individual images of male or female models and were asked to rate their professionalism on a scale of 0-100 – with 100 as the “most professional” as well as to identify their profession as either physician, surgeon, nurse, medical technician, or physician assistant.
The study team included 487 (93.3%) of 522 completed surveys in their analyses. Respondents’ mean age was 36.2 years; 260 (53.4%) were female; 372 (76.4%) were White; 33 (6.8%) were Black or African American. Younger respondents and those living in the Western United States who had more exposure to physician casual attire appeared more accepting of it, the authors wrote.
“I remember attending my white-coat ceremony as a medical student, and the symbolism of it all representing me entering the profession. It felt very emotional and heavy and I felt very proud to be there. I also remember taking a ‘selfie’ in my long white coat as a doctor for the first time before my first shift as a resident. But, I’ve also been wearing that same white coat, and a large badge with a ‘DOCTOR’ label on it, and been mistaken by a patient or parent for something other than the physician,” Alexandra M. Sims, a pediatrician and health equity researcher in Cincinnati, said in an interview. “So, I’d really hope that the take-home here is not simply that we must wear our white coats to be considered more professional. I think we have to unpack and dismantle how we’ve even built this notion of ‘professionalism’ in the first place. Women, people of color, and other marginalized groups were certainly not a part of the defining, but we must be a part of the reimagining of an equitable health care profession in this new era.”
As sartorial trends usher in more casual attire, clinicians should redouble efforts to build rapport and enhance communication with patients, such as clarifying team members’ roles when introducing themselves. Dr. Xun and coauthors noted that addressing gender bias is important for all clinicians – not just women – and point to the need for institutional and organizational support for disciplines where gender bias is “especially prevalent,” like surgery. “This responsibility should not be undertaken only by the individuals that experience the biases, which may result in additional cumulative career disadvantages. The promotion of equality and diversity begins with recognition, characterization, and evidence-supported interventions and is a community operation,” Dr. Xun and colleagues concluded.
“I do not equate attire to professionalism or experience, nor is it connected to my satisfaction with the physician. For myself and my daughter, it is the experience of care that ultimately influences our perceptions regarding the professionalism of the physician,” Hala H. Durrah, MTA, parent to a chronically ill child with special health care needs and a Patient and Family Engagement Consultant, said in an interview. “My respect for a physician will ultimately be determined by how my daughter and I were treated, not just from a clinical perspective, but how we felt during those interactions.”
FROM JAMA NETWORK OPEN
COVID-19 leaves wake of medical debt among U.S. adults
Despite the passage of four major relief bills in 2020 and 2021 and federal efforts to offset pandemic- and job-related coverage loss, many people continued to face financial challenges, especially those with a low income and those who are Black or Latino.
The survey, which included responses from 5,450 adults, revealed that 10% of adults aged 19-64 were uninsured during the first half of 2021, a rate lower than what was recorded in 2020 and 2019 in both federal and private surveys. However, uninsured rates were highest among those with low income, those younger than 50 years old, and Black and Latino adults.
For most adults who lost employee health insurance, the coverage gap was relatively brief, with 54% saying their coverage gap lasted 3-4 months. Only 16% of adults said coverage gaps lasted a year or longer.
“The good news is that this survey is suggesting that the coverage losses during the pandemic may have been offset by federal efforts to help people get and maintain health insurance coverage,” lead author Sara Collins, PhD, Commonwealth Fund vice president for health care coverage, access, and tracking, said in an interview.
“The bad news is that a third of Americans continue to struggle with medical bills and medical debt, even among those who have health insurance coverage,” Dr. Collins added.
Indeed, the survey found that about one-third of insured adults reported a medical bill problem or that they were paying off medical debt, as did approximately half of those who were uninsured. Medical debt caused 35% of respondents to use up most or all of their savings to pay it off.
Meanwhile, 27% of adults said medical bills left them unable to pay for necessities such as food, heat, or rent. What surprised Dr. Collins was that 43% of adults said they received a lower credit rating as a result of their medical debt, and 35% said they had taken on more credit card debt to pay off these bills.
“The fact that it’s bleeding over into people’s financial security in terms of their credit scores, I think is something that really needs to be looked at by policymakers,” Dr. Collins said.
When analyzed by race/ethnicity, the researchers found that 55% of Black adults and 44% of Latino/Hispanic adults reported medical bills and debt problems, compared with 32% of White adults. In addition, 47% of those living below the poverty line also reported problems with medical bills.
According to the survey, 45% of respondents were directly affected by the pandemic in at least one of three ways – testing positive or getting sick from COVID-19, losing income, or losing employer coverage – with Black and Latinx adults and those with lower incomes at greater risk.
George Abraham, MD, president of the American College of Physicians, said the Commonwealth Fund’s findings were not surprising because it has always been known that underrepresented populations struggle for access to care because of socioeconomic factors. He said these populations were more vulnerable in terms of more severe infections and disease burden during the pandemic.
“[This study] validates what primary care physicians have been saying all along in regard to our patients’ access to care and their ability to cover health care costs,” said Dr. Abraham, who was not involved with the study. “This will hopefully be an eye-opener and wake-up call that reiterates that we still do not have equitable access to care and vulnerable populations are disproportionately affected.”
He believes that, although people are insured, many of them may contend with medical debt when they fall ill because they can’t afford the premiums.
“Even though they may have been registered for health coverage, they may not have active coverage at the time of illness simply because they weren’t able to make their last premium payments because they’ve been down, because they lost their job, or whatever else,” Dr. Abraham explained. “On paper, they appear to have health care coverage. But in reality, clearly, that coverage does not match their needs or it’s not affordable.”
For Dr. Abraham, the study emphasizes the need to continue support for health care reform, including pricing it so that insurance is available for those with fewer socioeconomic resources.
Yalda Jabbarpour, MD, medical director of the Robert Graham Center for Policy Studies, Washington, said high-deductible health plans need to be “reined in” because they can lead to greater debt, particularly among vulnerable populations.
“Hopefully this will encourage policymakers to look more closely at the problem of medical debt as a contributing factor to financial instability,” Dr. Jabbarpour said. “Federal relief is important, so is expanding access to comprehensive, affordable health care coverage.”
Dr. Collins said there should also be a way to raise awareness of the health care marketplace and coverage options so that people have an easier time getting insured.
A version of this article first appeared on Medscape.com.
Despite the passage of four major relief bills in 2020 and 2021 and federal efforts to offset pandemic- and job-related coverage loss, many people continued to face financial challenges, especially those with a low income and those who are Black or Latino.
The survey, which included responses from 5,450 adults, revealed that 10% of adults aged 19-64 were uninsured during the first half of 2021, a rate lower than what was recorded in 2020 and 2019 in both federal and private surveys. However, uninsured rates were highest among those with low income, those younger than 50 years old, and Black and Latino adults.
For most adults who lost employee health insurance, the coverage gap was relatively brief, with 54% saying their coverage gap lasted 3-4 months. Only 16% of adults said coverage gaps lasted a year or longer.
“The good news is that this survey is suggesting that the coverage losses during the pandemic may have been offset by federal efforts to help people get and maintain health insurance coverage,” lead author Sara Collins, PhD, Commonwealth Fund vice president for health care coverage, access, and tracking, said in an interview.
“The bad news is that a third of Americans continue to struggle with medical bills and medical debt, even among those who have health insurance coverage,” Dr. Collins added.
Indeed, the survey found that about one-third of insured adults reported a medical bill problem or that they were paying off medical debt, as did approximately half of those who were uninsured. Medical debt caused 35% of respondents to use up most or all of their savings to pay it off.
Meanwhile, 27% of adults said medical bills left them unable to pay for necessities such as food, heat, or rent. What surprised Dr. Collins was that 43% of adults said they received a lower credit rating as a result of their medical debt, and 35% said they had taken on more credit card debt to pay off these bills.
“The fact that it’s bleeding over into people’s financial security in terms of their credit scores, I think is something that really needs to be looked at by policymakers,” Dr. Collins said.
When analyzed by race/ethnicity, the researchers found that 55% of Black adults and 44% of Latino/Hispanic adults reported medical bills and debt problems, compared with 32% of White adults. In addition, 47% of those living below the poverty line also reported problems with medical bills.
According to the survey, 45% of respondents were directly affected by the pandemic in at least one of three ways – testing positive or getting sick from COVID-19, losing income, or losing employer coverage – with Black and Latinx adults and those with lower incomes at greater risk.
George Abraham, MD, president of the American College of Physicians, said the Commonwealth Fund’s findings were not surprising because it has always been known that underrepresented populations struggle for access to care because of socioeconomic factors. He said these populations were more vulnerable in terms of more severe infections and disease burden during the pandemic.
“[This study] validates what primary care physicians have been saying all along in regard to our patients’ access to care and their ability to cover health care costs,” said Dr. Abraham, who was not involved with the study. “This will hopefully be an eye-opener and wake-up call that reiterates that we still do not have equitable access to care and vulnerable populations are disproportionately affected.”
He believes that, although people are insured, many of them may contend with medical debt when they fall ill because they can’t afford the premiums.
“Even though they may have been registered for health coverage, they may not have active coverage at the time of illness simply because they weren’t able to make their last premium payments because they’ve been down, because they lost their job, or whatever else,” Dr. Abraham explained. “On paper, they appear to have health care coverage. But in reality, clearly, that coverage does not match their needs or it’s not affordable.”
For Dr. Abraham, the study emphasizes the need to continue support for health care reform, including pricing it so that insurance is available for those with fewer socioeconomic resources.
Yalda Jabbarpour, MD, medical director of the Robert Graham Center for Policy Studies, Washington, said high-deductible health plans need to be “reined in” because they can lead to greater debt, particularly among vulnerable populations.
“Hopefully this will encourage policymakers to look more closely at the problem of medical debt as a contributing factor to financial instability,” Dr. Jabbarpour said. “Federal relief is important, so is expanding access to comprehensive, affordable health care coverage.”
Dr. Collins said there should also be a way to raise awareness of the health care marketplace and coverage options so that people have an easier time getting insured.
A version of this article first appeared on Medscape.com.
Despite the passage of four major relief bills in 2020 and 2021 and federal efforts to offset pandemic- and job-related coverage loss, many people continued to face financial challenges, especially those with a low income and those who are Black or Latino.
The survey, which included responses from 5,450 adults, revealed that 10% of adults aged 19-64 were uninsured during the first half of 2021, a rate lower than what was recorded in 2020 and 2019 in both federal and private surveys. However, uninsured rates were highest among those with low income, those younger than 50 years old, and Black and Latino adults.
For most adults who lost employee health insurance, the coverage gap was relatively brief, with 54% saying their coverage gap lasted 3-4 months. Only 16% of adults said coverage gaps lasted a year or longer.
“The good news is that this survey is suggesting that the coverage losses during the pandemic may have been offset by federal efforts to help people get and maintain health insurance coverage,” lead author Sara Collins, PhD, Commonwealth Fund vice president for health care coverage, access, and tracking, said in an interview.
“The bad news is that a third of Americans continue to struggle with medical bills and medical debt, even among those who have health insurance coverage,” Dr. Collins added.
Indeed, the survey found that about one-third of insured adults reported a medical bill problem or that they were paying off medical debt, as did approximately half of those who were uninsured. Medical debt caused 35% of respondents to use up most or all of their savings to pay it off.
Meanwhile, 27% of adults said medical bills left them unable to pay for necessities such as food, heat, or rent. What surprised Dr. Collins was that 43% of adults said they received a lower credit rating as a result of their medical debt, and 35% said they had taken on more credit card debt to pay off these bills.
“The fact that it’s bleeding over into people’s financial security in terms of their credit scores, I think is something that really needs to be looked at by policymakers,” Dr. Collins said.
When analyzed by race/ethnicity, the researchers found that 55% of Black adults and 44% of Latino/Hispanic adults reported medical bills and debt problems, compared with 32% of White adults. In addition, 47% of those living below the poverty line also reported problems with medical bills.
According to the survey, 45% of respondents were directly affected by the pandemic in at least one of three ways – testing positive or getting sick from COVID-19, losing income, or losing employer coverage – with Black and Latinx adults and those with lower incomes at greater risk.
George Abraham, MD, president of the American College of Physicians, said the Commonwealth Fund’s findings were not surprising because it has always been known that underrepresented populations struggle for access to care because of socioeconomic factors. He said these populations were more vulnerable in terms of more severe infections and disease burden during the pandemic.
“[This study] validates what primary care physicians have been saying all along in regard to our patients’ access to care and their ability to cover health care costs,” said Dr. Abraham, who was not involved with the study. “This will hopefully be an eye-opener and wake-up call that reiterates that we still do not have equitable access to care and vulnerable populations are disproportionately affected.”
He believes that, although people are insured, many of them may contend with medical debt when they fall ill because they can’t afford the premiums.
“Even though they may have been registered for health coverage, they may not have active coverage at the time of illness simply because they weren’t able to make their last premium payments because they’ve been down, because they lost their job, or whatever else,” Dr. Abraham explained. “On paper, they appear to have health care coverage. But in reality, clearly, that coverage does not match their needs or it’s not affordable.”
For Dr. Abraham, the study emphasizes the need to continue support for health care reform, including pricing it so that insurance is available for those with fewer socioeconomic resources.
Yalda Jabbarpour, MD, medical director of the Robert Graham Center for Policy Studies, Washington, said high-deductible health plans need to be “reined in” because they can lead to greater debt, particularly among vulnerable populations.
“Hopefully this will encourage policymakers to look more closely at the problem of medical debt as a contributing factor to financial instability,” Dr. Jabbarpour said. “Federal relief is important, so is expanding access to comprehensive, affordable health care coverage.”
Dr. Collins said there should also be a way to raise awareness of the health care marketplace and coverage options so that people have an easier time getting insured.
A version of this article first appeared on Medscape.com.
Let’s talk about race
“I feel like my aggression is being racialized.” “Of course I wouldn’t call the cops if I felt like hurting myself. I’m Black.”
Those statements represent the heightened trauma our Black and Brown patients with mental health issues have been experiencing. In the wake of increasingly publicized police brutality against Black and Brown communities, the role race plays in mental health decompensation is evident. At this moment in time, we must continue to improve our understanding of the role race plays in psychiatric disorders. We must also ask ourselves: At times, does psychiatry worsen the traumas of the communities we serve?
Some psychiatrists are afraid to speak about race. They may believe it to be too “political.” But avoiding these necessary conversations perpetuates the trauma of those we treat. It suggests that physicians are ignorant of an issue at the forefront of patients’ mental health. Psychiatry, today, is primarily focused on the biological aspects of disease. We must not forget that psychiatry is biopsychosocial. It is imperative that psychiatrists have conversations about race – and its significance to our patients and their care.
Only 10.4% of psychiatrists in the United States comprise those considered underrepresented in medicine (URM). Yet, those very groups make up 32.6% of the U.S. population and are overrepresented in psychiatric hospitals.1 Many studies have shown that concordant racial backgrounds between patient and physician lead to a more positive patient experience2 and arguably, the subsequent potential for better health outcomes. Our efforts in addressing this disparity often fall short. URM applicants may be hesitant to join an institution where diversity is lacking or where they may be the only minority.3 While there is no simple solution, I propose that psychiatrists promote the importance of mental health to Black and Brown students of all ages by collaborating with schools and community leaders.
It is important to acknowledge that the lack of diversity within psychiatry is reflective of that among all physicians. This in part stems from the barriers to medical education that Black and Brown communities face. Those who start off with more resources or have parents who are physicians are at an advantage when trying to get into medical school. In fact, one in five medical students have a parent who is a physician4 and about three-fourths of students come from families whose income falls among the top two quintiles.5 Impoverished communities, which have a disproportionate share of Black and Brown people, cannot afford to take MCAT preparatory classes or to accept unpaid “resume building” opportunities. Many medical schools continue to place more weight on test scores and research/medical experiences, despite a shift to a more holistic review process. Institutions that have tried a different approach and accepted students from more diverse backgrounds may often overlook the challenges that URM students face while in medical school and fail to provide appropriate support resources.
The result is a failure to retain such students. A study conducted at Stony Brook (N.Y.) University showed that those underrepresented in medicine were six times more likely to get dismissed from medical school, and three times more likely to both withdraw or graduate beyond 4 years, compared with their White counterparts.6 This is a serious issue that needs to change on a structural and systemic level.
Any discussion of race and psychiatry must acknowledge psychiatry’s history of racism against Black and Brown communities to engage in racially informed discussions with our patients. Only then can we play a better role advocating against racism within the field in the future. Dating back to the 18th century, psychiatry has promoted ideologies that promote racism. Benjamin Rush, considered the “father of American Psychiatry,” believed that Black skin was a disease derived from leprosy called “negritude.” In the late 19th century, this twisted ideology continued with the invention of the term “drapetomania,” which was used to describe enslaved people who ran away as having a mental disorder.7 Black prisoners were subjected to experimental treatment with substances such as LSD and bulbocapnine to subdue them.8 This idea that minorities were dangerous and needed to be subdued translated into a higher number of schizophrenia diagnoses, particularly among Black men, as it was used as a tool to vilify them in the 1970s. Although schizophrenia is equally prevalent among Whites and non-Whites, Black people are four times more likely to be diagnosed, compared with their White counterparts, while Hispanics are three times more likely. Studies have shown that Black and Brown men are also more likely to receive higher doses of antipsychotics.9
Given this history, it is not surprising that Black and Brown representation within the field is lacking and that patients may be hesitant to share their feelings about race with us. While we can’t change history, we can take a stance condemning the harmful behavior of the past. The American Psychiatric Association issued an apology earlier this year to Black, Indigenous, and People of Color for its support in structural racism.10 This is a step in the right direction, but we need more than statements or performative actions. We need to amplify the voices of Black and Brown psychiatrists and patients, as well as highlight their current and past contributions to the field. While my educational experiences focused on the work of prominent White scholars, medical curricula should showcase the work of people like Solomon Carter Fuller, MD, a Black psychiatrist who was essential to understanding Alzheimer’s, or Joseph White, PhD, sometimes referred to as the “godfather of Black psychology.”11
At times, I have found myself witness to situations where colleagues make statements that not only do not condemn racism, but in fact encourage it. I have unfortunately heard some use the all-too-familiar rhetoric of reverse racism, such as: “They just assume I am racist because I am a White male” or “They’re being racist against me” or statements like “Don’t you think it is far-fetched to believe she was just sitting on a college campus doing nothing when the police were called?” Rhetoric such as this is problematic to the field of psychiatry and medicine as a whole – and only serves to further invalidate the feelings of our Black and Brown patients. We must increase exposure and education regarding racism to address this, especially the meaning of microaggressions, a concept many fail to understand.
Attention to the subject of racism has increased within medical schools and residency training programs in the wake of George Floyd’s death. However, most programs often make these lectures optional or only have one to two limited sessions. Furthermore, many do not make it mandatory for faculty to attend; they are arguably the most in need of this training given that they set the precedent of how to practice psychiatry. Some institutions have incorporated comprehensive antiracist curriculums into medical training. One model that has been successful is the Social Justice and Health Equity program within Yale University’s psychiatry residency. The curriculum has four tracks:
- Structural competency, which focuses on the mental health impact of extraclinical structures, for example a patient’s neighborhood and associated barriers of access.
- Human experience, which explores the interaction of patients and providers and how biases play a role.
- Advocacy, which teaches residents the written and oral skills to lobby for patient interests on a community and legislative level.
- History of psychiatry, which focuses on understanding psychiatry’s prior role in racism.
In each track, there are group discussions, cases led by faculty, and meetings with community leaders. Through this curriculum, residents learn about power, privilege, and how to interact with and advocate for patients in a way that promotes equity, rather than racial disparity.12,13 This is a model that other psychiatric residency programs can promote, emulate, and benefit from.
Educating ourselves will hopefully lead to a deeper introspection of how we interact with patients and if we are promoting antiracism through our attitude and actions. Reflecting on my own personal practice, I have noted that the interplay of race, mental health, and provider decision-making becomes particularly complex when dealing with situations in which a patient exhibits increased aggression or agitation. As a second-year psychiatric resident immersed in the inpatient world, I have become familiar with patients at higher risk and greater need. The first attempt toward de-escalation involves verbal cues without any other more intrusive measures. If that fails, intramuscular (IM) medications are typically considered. If a patient has a history of aggressive behavior, the threshold to use IM medications can decrease dramatically. This is mainly to protect ourselves and our nursing staff and to prioritize safety. While I understand this rationale, I wonder about the patient’s experience. What constitutes “aggressive” behavior? For patients who have had violence used against them because of their race or who have suffered from police brutality, having police present or threatening IM medications will increasingly trigger them and escalate the situation. The aftermath can deepen the distrust of psychiatry by Black and Brown people.
How do we then handle such situations in a way that both protects our staff from physical harm and protects our patients from racial trauma? While I don’t have a great answer, I think we can benefit from standardizing what we consider aggressive behavior and have specific criteria that patients need to exhibit before administering an IM medication. In addition, discussions with the team, including residents, nurses, and attending physicians, about how to address an emergent situation before it actually happens are essential. Specifically discussing the patient’s history and race and how it may affect the situation is not something to be shied away from. Lastly, in the event that an IM medication is administered and police are present, debriefing with the patient afterward is necessary. The patient may not be willing or able to listen to you or trust you, but taking accountability and acknowledging what happened, justified or not, is a part of the process of rebuilding rapport.
Both in the purview of the individual psychiatrist and the field of psychiatry as a whole, we need to examine our behavior and not be afraid to make changes for the betterment of our patients. We must learn to talk about race with our patients and in the process, advocate for more representation of Black and Brown psychiatrists, understanding the barriers faced by these communities when pursuing the medical field. We must educate ourselves on psychiatry’s history, and equip ourselves with knowledge and tools to promote antiracism and shape psychiatry’s future. We can then apply these very tools to challenging situations we may encounter daily with the ultimate goal of improving the mental health of our patients. This is the only way we will progress and ensure that psychiatry is an equitable, antiracist field. As Ibram X. Kendi, PhD, has written, “The heartbeat of antiracism is self-reflection, recognition, admission, and fundamentally self-critique.”
Dr. Malik is a second-year psychiatry resident at the University of California, San Diego. She has a background in policy and grassroots organizing through her time working at the National Coalition for the Homeless and the Women’s Law Project. Dr. Malik has no disclosures.
References
1. Wyse R et al. Acad Psychiatry. 2020 Oct;44(5):523-30.
2. Cooper LA et al. Ann Intern Med. 2003;139:907-15.
3. Pierre JM et al. Acad Psychiatry. 2017;41:226-32.
4. Hartocollis A. “Getting into med school without hard sciences.” New York Times. 2010 Jul 29.
5. AAMC. An updated look at the economic diversity of U.S. medical students. Analysis in Brief. 2018 Oct;18(5).
6. Rainey ML. How do we retain minority health professions students. In: Smedley BD et al. The right thing to do, the smart thing to do: Enhancing diversity in the health professions: Summary of the Symposium on Diversity in Health Professions in Honor of Herbert W. Nickens, M.D. Institute of Medicine. National Academies Press. 2001.
7. Geller J. “Structural racism in American psychiatry and APA: Part 1.” Psychiatric News. 2020 Jun 23.
8. Mohr CL and Gordon JE. Tulane: The emergence of a modern university, 1945-1980. Louisiana State University Press, Baton Rouge. 2001.
9. Metzl JM. The protest psychosis: How schizophrenia became a Black disease. Beacon Press. 2010.
10. APA’s apology to Black, indigenous and people of color for its support of structural racism in psychiatry. American Psychiatric Association. 2021 Jan 18.
11. Black pioneers in mental health. Mental Health America. 2021.
12. Belli B. For Yale’s emerging psychiatrists, confronting racism is in the curriculum. Yale News. 2020 Jul 30.
13. Jordan A and Jackson D. Social justice and health equity curriculum. Yale School of Medicine. 2019 Sep 24.
“I feel like my aggression is being racialized.” “Of course I wouldn’t call the cops if I felt like hurting myself. I’m Black.”
Those statements represent the heightened trauma our Black and Brown patients with mental health issues have been experiencing. In the wake of increasingly publicized police brutality against Black and Brown communities, the role race plays in mental health decompensation is evident. At this moment in time, we must continue to improve our understanding of the role race plays in psychiatric disorders. We must also ask ourselves: At times, does psychiatry worsen the traumas of the communities we serve?
Some psychiatrists are afraid to speak about race. They may believe it to be too “political.” But avoiding these necessary conversations perpetuates the trauma of those we treat. It suggests that physicians are ignorant of an issue at the forefront of patients’ mental health. Psychiatry, today, is primarily focused on the biological aspects of disease. We must not forget that psychiatry is biopsychosocial. It is imperative that psychiatrists have conversations about race – and its significance to our patients and their care.
Only 10.4% of psychiatrists in the United States comprise those considered underrepresented in medicine (URM). Yet, those very groups make up 32.6% of the U.S. population and are overrepresented in psychiatric hospitals.1 Many studies have shown that concordant racial backgrounds between patient and physician lead to a more positive patient experience2 and arguably, the subsequent potential for better health outcomes. Our efforts in addressing this disparity often fall short. URM applicants may be hesitant to join an institution where diversity is lacking or where they may be the only minority.3 While there is no simple solution, I propose that psychiatrists promote the importance of mental health to Black and Brown students of all ages by collaborating with schools and community leaders.
It is important to acknowledge that the lack of diversity within psychiatry is reflective of that among all physicians. This in part stems from the barriers to medical education that Black and Brown communities face. Those who start off with more resources or have parents who are physicians are at an advantage when trying to get into medical school. In fact, one in five medical students have a parent who is a physician4 and about three-fourths of students come from families whose income falls among the top two quintiles.5 Impoverished communities, which have a disproportionate share of Black and Brown people, cannot afford to take MCAT preparatory classes or to accept unpaid “resume building” opportunities. Many medical schools continue to place more weight on test scores and research/medical experiences, despite a shift to a more holistic review process. Institutions that have tried a different approach and accepted students from more diverse backgrounds may often overlook the challenges that URM students face while in medical school and fail to provide appropriate support resources.
The result is a failure to retain such students. A study conducted at Stony Brook (N.Y.) University showed that those underrepresented in medicine were six times more likely to get dismissed from medical school, and three times more likely to both withdraw or graduate beyond 4 years, compared with their White counterparts.6 This is a serious issue that needs to change on a structural and systemic level.
Any discussion of race and psychiatry must acknowledge psychiatry’s history of racism against Black and Brown communities to engage in racially informed discussions with our patients. Only then can we play a better role advocating against racism within the field in the future. Dating back to the 18th century, psychiatry has promoted ideologies that promote racism. Benjamin Rush, considered the “father of American Psychiatry,” believed that Black skin was a disease derived from leprosy called “negritude.” In the late 19th century, this twisted ideology continued with the invention of the term “drapetomania,” which was used to describe enslaved people who ran away as having a mental disorder.7 Black prisoners were subjected to experimental treatment with substances such as LSD and bulbocapnine to subdue them.8 This idea that minorities were dangerous and needed to be subdued translated into a higher number of schizophrenia diagnoses, particularly among Black men, as it was used as a tool to vilify them in the 1970s. Although schizophrenia is equally prevalent among Whites and non-Whites, Black people are four times more likely to be diagnosed, compared with their White counterparts, while Hispanics are three times more likely. Studies have shown that Black and Brown men are also more likely to receive higher doses of antipsychotics.9
Given this history, it is not surprising that Black and Brown representation within the field is lacking and that patients may be hesitant to share their feelings about race with us. While we can’t change history, we can take a stance condemning the harmful behavior of the past. The American Psychiatric Association issued an apology earlier this year to Black, Indigenous, and People of Color for its support in structural racism.10 This is a step in the right direction, but we need more than statements or performative actions. We need to amplify the voices of Black and Brown psychiatrists and patients, as well as highlight their current and past contributions to the field. While my educational experiences focused on the work of prominent White scholars, medical curricula should showcase the work of people like Solomon Carter Fuller, MD, a Black psychiatrist who was essential to understanding Alzheimer’s, or Joseph White, PhD, sometimes referred to as the “godfather of Black psychology.”11
At times, I have found myself witness to situations where colleagues make statements that not only do not condemn racism, but in fact encourage it. I have unfortunately heard some use the all-too-familiar rhetoric of reverse racism, such as: “They just assume I am racist because I am a White male” or “They’re being racist against me” or statements like “Don’t you think it is far-fetched to believe she was just sitting on a college campus doing nothing when the police were called?” Rhetoric such as this is problematic to the field of psychiatry and medicine as a whole – and only serves to further invalidate the feelings of our Black and Brown patients. We must increase exposure and education regarding racism to address this, especially the meaning of microaggressions, a concept many fail to understand.
Attention to the subject of racism has increased within medical schools and residency training programs in the wake of George Floyd’s death. However, most programs often make these lectures optional or only have one to two limited sessions. Furthermore, many do not make it mandatory for faculty to attend; they are arguably the most in need of this training given that they set the precedent of how to practice psychiatry. Some institutions have incorporated comprehensive antiracist curriculums into medical training. One model that has been successful is the Social Justice and Health Equity program within Yale University’s psychiatry residency. The curriculum has four tracks:
- Structural competency, which focuses on the mental health impact of extraclinical structures, for example a patient’s neighborhood and associated barriers of access.
- Human experience, which explores the interaction of patients and providers and how biases play a role.
- Advocacy, which teaches residents the written and oral skills to lobby for patient interests on a community and legislative level.
- History of psychiatry, which focuses on understanding psychiatry’s prior role in racism.
In each track, there are group discussions, cases led by faculty, and meetings with community leaders. Through this curriculum, residents learn about power, privilege, and how to interact with and advocate for patients in a way that promotes equity, rather than racial disparity.12,13 This is a model that other psychiatric residency programs can promote, emulate, and benefit from.
Educating ourselves will hopefully lead to a deeper introspection of how we interact with patients and if we are promoting antiracism through our attitude and actions. Reflecting on my own personal practice, I have noted that the interplay of race, mental health, and provider decision-making becomes particularly complex when dealing with situations in which a patient exhibits increased aggression or agitation. As a second-year psychiatric resident immersed in the inpatient world, I have become familiar with patients at higher risk and greater need. The first attempt toward de-escalation involves verbal cues without any other more intrusive measures. If that fails, intramuscular (IM) medications are typically considered. If a patient has a history of aggressive behavior, the threshold to use IM medications can decrease dramatically. This is mainly to protect ourselves and our nursing staff and to prioritize safety. While I understand this rationale, I wonder about the patient’s experience. What constitutes “aggressive” behavior? For patients who have had violence used against them because of their race or who have suffered from police brutality, having police present or threatening IM medications will increasingly trigger them and escalate the situation. The aftermath can deepen the distrust of psychiatry by Black and Brown people.
How do we then handle such situations in a way that both protects our staff from physical harm and protects our patients from racial trauma? While I don’t have a great answer, I think we can benefit from standardizing what we consider aggressive behavior and have specific criteria that patients need to exhibit before administering an IM medication. In addition, discussions with the team, including residents, nurses, and attending physicians, about how to address an emergent situation before it actually happens are essential. Specifically discussing the patient’s history and race and how it may affect the situation is not something to be shied away from. Lastly, in the event that an IM medication is administered and police are present, debriefing with the patient afterward is necessary. The patient may not be willing or able to listen to you or trust you, but taking accountability and acknowledging what happened, justified or not, is a part of the process of rebuilding rapport.
Both in the purview of the individual psychiatrist and the field of psychiatry as a whole, we need to examine our behavior and not be afraid to make changes for the betterment of our patients. We must learn to talk about race with our patients and in the process, advocate for more representation of Black and Brown psychiatrists, understanding the barriers faced by these communities when pursuing the medical field. We must educate ourselves on psychiatry’s history, and equip ourselves with knowledge and tools to promote antiracism and shape psychiatry’s future. We can then apply these very tools to challenging situations we may encounter daily with the ultimate goal of improving the mental health of our patients. This is the only way we will progress and ensure that psychiatry is an equitable, antiracist field. As Ibram X. Kendi, PhD, has written, “The heartbeat of antiracism is self-reflection, recognition, admission, and fundamentally self-critique.”
Dr. Malik is a second-year psychiatry resident at the University of California, San Diego. She has a background in policy and grassroots organizing through her time working at the National Coalition for the Homeless and the Women’s Law Project. Dr. Malik has no disclosures.
References
1. Wyse R et al. Acad Psychiatry. 2020 Oct;44(5):523-30.
2. Cooper LA et al. Ann Intern Med. 2003;139:907-15.
3. Pierre JM et al. Acad Psychiatry. 2017;41:226-32.
4. Hartocollis A. “Getting into med school without hard sciences.” New York Times. 2010 Jul 29.
5. AAMC. An updated look at the economic diversity of U.S. medical students. Analysis in Brief. 2018 Oct;18(5).
6. Rainey ML. How do we retain minority health professions students. In: Smedley BD et al. The right thing to do, the smart thing to do: Enhancing diversity in the health professions: Summary of the Symposium on Diversity in Health Professions in Honor of Herbert W. Nickens, M.D. Institute of Medicine. National Academies Press. 2001.
7. Geller J. “Structural racism in American psychiatry and APA: Part 1.” Psychiatric News. 2020 Jun 23.
8. Mohr CL and Gordon JE. Tulane: The emergence of a modern university, 1945-1980. Louisiana State University Press, Baton Rouge. 2001.
9. Metzl JM. The protest psychosis: How schizophrenia became a Black disease. Beacon Press. 2010.
10. APA’s apology to Black, indigenous and people of color for its support of structural racism in psychiatry. American Psychiatric Association. 2021 Jan 18.
11. Black pioneers in mental health. Mental Health America. 2021.
12. Belli B. For Yale’s emerging psychiatrists, confronting racism is in the curriculum. Yale News. 2020 Jul 30.
13. Jordan A and Jackson D. Social justice and health equity curriculum. Yale School of Medicine. 2019 Sep 24.
“I feel like my aggression is being racialized.” “Of course I wouldn’t call the cops if I felt like hurting myself. I’m Black.”
Those statements represent the heightened trauma our Black and Brown patients with mental health issues have been experiencing. In the wake of increasingly publicized police brutality against Black and Brown communities, the role race plays in mental health decompensation is evident. At this moment in time, we must continue to improve our understanding of the role race plays in psychiatric disorders. We must also ask ourselves: At times, does psychiatry worsen the traumas of the communities we serve?
Some psychiatrists are afraid to speak about race. They may believe it to be too “political.” But avoiding these necessary conversations perpetuates the trauma of those we treat. It suggests that physicians are ignorant of an issue at the forefront of patients’ mental health. Psychiatry, today, is primarily focused on the biological aspects of disease. We must not forget that psychiatry is biopsychosocial. It is imperative that psychiatrists have conversations about race – and its significance to our patients and their care.
Only 10.4% of psychiatrists in the United States comprise those considered underrepresented in medicine (URM). Yet, those very groups make up 32.6% of the U.S. population and are overrepresented in psychiatric hospitals.1 Many studies have shown that concordant racial backgrounds between patient and physician lead to a more positive patient experience2 and arguably, the subsequent potential for better health outcomes. Our efforts in addressing this disparity often fall short. URM applicants may be hesitant to join an institution where diversity is lacking or where they may be the only minority.3 While there is no simple solution, I propose that psychiatrists promote the importance of mental health to Black and Brown students of all ages by collaborating with schools and community leaders.
It is important to acknowledge that the lack of diversity within psychiatry is reflective of that among all physicians. This in part stems from the barriers to medical education that Black and Brown communities face. Those who start off with more resources or have parents who are physicians are at an advantage when trying to get into medical school. In fact, one in five medical students have a parent who is a physician4 and about three-fourths of students come from families whose income falls among the top two quintiles.5 Impoverished communities, which have a disproportionate share of Black and Brown people, cannot afford to take MCAT preparatory classes or to accept unpaid “resume building” opportunities. Many medical schools continue to place more weight on test scores and research/medical experiences, despite a shift to a more holistic review process. Institutions that have tried a different approach and accepted students from more diverse backgrounds may often overlook the challenges that URM students face while in medical school and fail to provide appropriate support resources.
The result is a failure to retain such students. A study conducted at Stony Brook (N.Y.) University showed that those underrepresented in medicine were six times more likely to get dismissed from medical school, and three times more likely to both withdraw or graduate beyond 4 years, compared with their White counterparts.6 This is a serious issue that needs to change on a structural and systemic level.
Any discussion of race and psychiatry must acknowledge psychiatry’s history of racism against Black and Brown communities to engage in racially informed discussions with our patients. Only then can we play a better role advocating against racism within the field in the future. Dating back to the 18th century, psychiatry has promoted ideologies that promote racism. Benjamin Rush, considered the “father of American Psychiatry,” believed that Black skin was a disease derived from leprosy called “negritude.” In the late 19th century, this twisted ideology continued with the invention of the term “drapetomania,” which was used to describe enslaved people who ran away as having a mental disorder.7 Black prisoners were subjected to experimental treatment with substances such as LSD and bulbocapnine to subdue them.8 This idea that minorities were dangerous and needed to be subdued translated into a higher number of schizophrenia diagnoses, particularly among Black men, as it was used as a tool to vilify them in the 1970s. Although schizophrenia is equally prevalent among Whites and non-Whites, Black people are four times more likely to be diagnosed, compared with their White counterparts, while Hispanics are three times more likely. Studies have shown that Black and Brown men are also more likely to receive higher doses of antipsychotics.9
Given this history, it is not surprising that Black and Brown representation within the field is lacking and that patients may be hesitant to share their feelings about race with us. While we can’t change history, we can take a stance condemning the harmful behavior of the past. The American Psychiatric Association issued an apology earlier this year to Black, Indigenous, and People of Color for its support in structural racism.10 This is a step in the right direction, but we need more than statements or performative actions. We need to amplify the voices of Black and Brown psychiatrists and patients, as well as highlight their current and past contributions to the field. While my educational experiences focused on the work of prominent White scholars, medical curricula should showcase the work of people like Solomon Carter Fuller, MD, a Black psychiatrist who was essential to understanding Alzheimer’s, or Joseph White, PhD, sometimes referred to as the “godfather of Black psychology.”11
At times, I have found myself witness to situations where colleagues make statements that not only do not condemn racism, but in fact encourage it. I have unfortunately heard some use the all-too-familiar rhetoric of reverse racism, such as: “They just assume I am racist because I am a White male” or “They’re being racist against me” or statements like “Don’t you think it is far-fetched to believe she was just sitting on a college campus doing nothing when the police were called?” Rhetoric such as this is problematic to the field of psychiatry and medicine as a whole – and only serves to further invalidate the feelings of our Black and Brown patients. We must increase exposure and education regarding racism to address this, especially the meaning of microaggressions, a concept many fail to understand.
Attention to the subject of racism has increased within medical schools and residency training programs in the wake of George Floyd’s death. However, most programs often make these lectures optional or only have one to two limited sessions. Furthermore, many do not make it mandatory for faculty to attend; they are arguably the most in need of this training given that they set the precedent of how to practice psychiatry. Some institutions have incorporated comprehensive antiracist curriculums into medical training. One model that has been successful is the Social Justice and Health Equity program within Yale University’s psychiatry residency. The curriculum has four tracks:
- Structural competency, which focuses on the mental health impact of extraclinical structures, for example a patient’s neighborhood and associated barriers of access.
- Human experience, which explores the interaction of patients and providers and how biases play a role.
- Advocacy, which teaches residents the written and oral skills to lobby for patient interests on a community and legislative level.
- History of psychiatry, which focuses on understanding psychiatry’s prior role in racism.
In each track, there are group discussions, cases led by faculty, and meetings with community leaders. Through this curriculum, residents learn about power, privilege, and how to interact with and advocate for patients in a way that promotes equity, rather than racial disparity.12,13 This is a model that other psychiatric residency programs can promote, emulate, and benefit from.
Educating ourselves will hopefully lead to a deeper introspection of how we interact with patients and if we are promoting antiracism through our attitude and actions. Reflecting on my own personal practice, I have noted that the interplay of race, mental health, and provider decision-making becomes particularly complex when dealing with situations in which a patient exhibits increased aggression or agitation. As a second-year psychiatric resident immersed in the inpatient world, I have become familiar with patients at higher risk and greater need. The first attempt toward de-escalation involves verbal cues without any other more intrusive measures. If that fails, intramuscular (IM) medications are typically considered. If a patient has a history of aggressive behavior, the threshold to use IM medications can decrease dramatically. This is mainly to protect ourselves and our nursing staff and to prioritize safety. While I understand this rationale, I wonder about the patient’s experience. What constitutes “aggressive” behavior? For patients who have had violence used against them because of their race or who have suffered from police brutality, having police present or threatening IM medications will increasingly trigger them and escalate the situation. The aftermath can deepen the distrust of psychiatry by Black and Brown people.
How do we then handle such situations in a way that both protects our staff from physical harm and protects our patients from racial trauma? While I don’t have a great answer, I think we can benefit from standardizing what we consider aggressive behavior and have specific criteria that patients need to exhibit before administering an IM medication. In addition, discussions with the team, including residents, nurses, and attending physicians, about how to address an emergent situation before it actually happens are essential. Specifically discussing the patient’s history and race and how it may affect the situation is not something to be shied away from. Lastly, in the event that an IM medication is administered and police are present, debriefing with the patient afterward is necessary. The patient may not be willing or able to listen to you or trust you, but taking accountability and acknowledging what happened, justified or not, is a part of the process of rebuilding rapport.
Both in the purview of the individual psychiatrist and the field of psychiatry as a whole, we need to examine our behavior and not be afraid to make changes for the betterment of our patients. We must learn to talk about race with our patients and in the process, advocate for more representation of Black and Brown psychiatrists, understanding the barriers faced by these communities when pursuing the medical field. We must educate ourselves on psychiatry’s history, and equip ourselves with knowledge and tools to promote antiracism and shape psychiatry’s future. We can then apply these very tools to challenging situations we may encounter daily with the ultimate goal of improving the mental health of our patients. This is the only way we will progress and ensure that psychiatry is an equitable, antiracist field. As Ibram X. Kendi, PhD, has written, “The heartbeat of antiracism is self-reflection, recognition, admission, and fundamentally self-critique.”
Dr. Malik is a second-year psychiatry resident at the University of California, San Diego. She has a background in policy and grassroots organizing through her time working at the National Coalition for the Homeless and the Women’s Law Project. Dr. Malik has no disclosures.
References
1. Wyse R et al. Acad Psychiatry. 2020 Oct;44(5):523-30.
2. Cooper LA et al. Ann Intern Med. 2003;139:907-15.
3. Pierre JM et al. Acad Psychiatry. 2017;41:226-32.
4. Hartocollis A. “Getting into med school without hard sciences.” New York Times. 2010 Jul 29.
5. AAMC. An updated look at the economic diversity of U.S. medical students. Analysis in Brief. 2018 Oct;18(5).
6. Rainey ML. How do we retain minority health professions students. In: Smedley BD et al. The right thing to do, the smart thing to do: Enhancing diversity in the health professions: Summary of the Symposium on Diversity in Health Professions in Honor of Herbert W. Nickens, M.D. Institute of Medicine. National Academies Press. 2001.
7. Geller J. “Structural racism in American psychiatry and APA: Part 1.” Psychiatric News. 2020 Jun 23.
8. Mohr CL and Gordon JE. Tulane: The emergence of a modern university, 1945-1980. Louisiana State University Press, Baton Rouge. 2001.
9. Metzl JM. The protest psychosis: How schizophrenia became a Black disease. Beacon Press. 2010.
10. APA’s apology to Black, indigenous and people of color for its support of structural racism in psychiatry. American Psychiatric Association. 2021 Jan 18.
11. Black pioneers in mental health. Mental Health America. 2021.
12. Belli B. For Yale’s emerging psychiatrists, confronting racism is in the curriculum. Yale News. 2020 Jul 30.
13. Jordan A and Jackson D. Social justice and health equity curriculum. Yale School of Medicine. 2019 Sep 24.
Common outcome measures for AD lack adequate reporting of race, skin tone
, according to results from a systematic review.
“AD is associated with considerable heterogeneity across different races and skin tones,” presenting study author Trisha Kaundinya said at the Revolutionizing Atopic Dermatitis symposium. “Compared with lighter skin tones, darker skin tones more commonly have diffuse xerosis, Dennis-Morgan lines, hyperlinearity of the palms, periorbital dark circles, lichenification, and prurigo nodularis. This heterogeneity can be challenging to assess in clinical trials and in practice.”
The Harmonizing Outcome Measures for Eczema (HOME) group has selected several scales by international consensus. For clinical trials, the group recommends the Patient-Oriented Eczema Measure (POEM), Eczema Area and Severity Index (EASI), and Dermatology Life Quality Index (DLQI). In clinical practice, the HOME group recommends the POEM, Patient-Oriented Scoring Atopic Dermatitis (PO-SCORAD), and the Numeric Rating Scale (NRS)-itch measures. “The psychometric validity and reliability of these outcome measures have undergone robust investigation before, but the validity and reliability of these outcome measures remains uncertain across different races, ethnicities, and skin tones,” Ms. Kaundinya said.
Jonathan Silverberg, MD, PhD, associate professor of dermatology at George Washington University, Washington, in collaboration with Andrew F. Alexis, MD, MPH, vice-chair for diversity and inclusion for the department of dermatology at Weill-Cornell Medicine, New York, and Jacob P. Thyssen, MD, PhD, at the University of Copenhagen, Denmark, sought to examine reporting of race, ethnicity, and skin tone, and to compare results across these groups from studies of psychometric properties for outcome measures in AD. Under the mentorship of Dr. Silverberg, Ms. Kaundinya, a medical student at Northwestern University, Chicago, and her research associates conducted a systematic review that searched PubMed and Embase and identified 165 relevant published studies of 41,146 individuals.
Of the individuals participating in these 165 studies, 73% had an unspecified racial background, 18% were White, 4% were Asian, 2% were Black, 2% were Hispanic, 1% were multiracial/other, and the remainder were American Indian/Alaskan Native. Only 55 of the studies (33%) reported the distribution of race or ethnicity, 5 (3%) reported the distribution of skin tone, and 16 (10%) reported psychometric differences in patients with different races, ethnicities, or skin tones. In addition, only 5 of 113 (4%) studies that did not report race, ethnicity, or skin tone–based differences acknowledged absence of stratification as a limitation.
Of note, significant differential item functioning was found between race subgroups for one or more items of the PO-SCORAD, the Patient-Reported Outcomes Measurement Information System (PROMIS) Itch Questionnaire (PIQ) Short Forms, POEM, DLQI, Hospital Anxiety and Depression Scale (HADS), Itchy Quality of Life (ItchyQOL) scale, 5-dimensions (5D) itch scale, Short Form (SF)-12, and NRS-itch. “Correlations of the POEM with the Investigator’s Global Assessment (IGA) differed the most between skin of color and lighter skin,” Ms. Kaundinya said.
“The POEM did seem to correlate similarly with the DLQI and the EASI in both white and nonwhite participants, which may indicate why this trifecta of instruments is recommended by the HOME group. One study found that substituting the erythema component of the EASI scale with greyness for darker skin, in which erythema is more challenging to assess, did not significantly improve the reliability of EASI. This indicates that further research is needed to investigate how EASI can be modified to perform better in darker skin tones.”
She pointed out that some studies of clinician-reported outcome measures were underpowered to detect meaningful differences between patient subgroups. “There were also insufficient data to perform meta-regression of differences between patient subgroups,” she said. “Overall, future studies are needed to determine whether outcome measures recommended by the HOME and other tools perform equally well across diverse patient populations. This systematic review indicates significant reporting and knowledge gaps for psychometric properties of outcome measures by race, ethnicity, or skin tone in AD.”
Ms. Kaundinya reported having no relevant financial disclosures. Dr. Silverberg, the study’s senior author, is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
, according to results from a systematic review.
“AD is associated with considerable heterogeneity across different races and skin tones,” presenting study author Trisha Kaundinya said at the Revolutionizing Atopic Dermatitis symposium. “Compared with lighter skin tones, darker skin tones more commonly have diffuse xerosis, Dennis-Morgan lines, hyperlinearity of the palms, periorbital dark circles, lichenification, and prurigo nodularis. This heterogeneity can be challenging to assess in clinical trials and in practice.”
The Harmonizing Outcome Measures for Eczema (HOME) group has selected several scales by international consensus. For clinical trials, the group recommends the Patient-Oriented Eczema Measure (POEM), Eczema Area and Severity Index (EASI), and Dermatology Life Quality Index (DLQI). In clinical practice, the HOME group recommends the POEM, Patient-Oriented Scoring Atopic Dermatitis (PO-SCORAD), and the Numeric Rating Scale (NRS)-itch measures. “The psychometric validity and reliability of these outcome measures have undergone robust investigation before, but the validity and reliability of these outcome measures remains uncertain across different races, ethnicities, and skin tones,” Ms. Kaundinya said.
Jonathan Silverberg, MD, PhD, associate professor of dermatology at George Washington University, Washington, in collaboration with Andrew F. Alexis, MD, MPH, vice-chair for diversity and inclusion for the department of dermatology at Weill-Cornell Medicine, New York, and Jacob P. Thyssen, MD, PhD, at the University of Copenhagen, Denmark, sought to examine reporting of race, ethnicity, and skin tone, and to compare results across these groups from studies of psychometric properties for outcome measures in AD. Under the mentorship of Dr. Silverberg, Ms. Kaundinya, a medical student at Northwestern University, Chicago, and her research associates conducted a systematic review that searched PubMed and Embase and identified 165 relevant published studies of 41,146 individuals.
Of the individuals participating in these 165 studies, 73% had an unspecified racial background, 18% were White, 4% were Asian, 2% were Black, 2% were Hispanic, 1% were multiracial/other, and the remainder were American Indian/Alaskan Native. Only 55 of the studies (33%) reported the distribution of race or ethnicity, 5 (3%) reported the distribution of skin tone, and 16 (10%) reported psychometric differences in patients with different races, ethnicities, or skin tones. In addition, only 5 of 113 (4%) studies that did not report race, ethnicity, or skin tone–based differences acknowledged absence of stratification as a limitation.
Of note, significant differential item functioning was found between race subgroups for one or more items of the PO-SCORAD, the Patient-Reported Outcomes Measurement Information System (PROMIS) Itch Questionnaire (PIQ) Short Forms, POEM, DLQI, Hospital Anxiety and Depression Scale (HADS), Itchy Quality of Life (ItchyQOL) scale, 5-dimensions (5D) itch scale, Short Form (SF)-12, and NRS-itch. “Correlations of the POEM with the Investigator’s Global Assessment (IGA) differed the most between skin of color and lighter skin,” Ms. Kaundinya said.
“The POEM did seem to correlate similarly with the DLQI and the EASI in both white and nonwhite participants, which may indicate why this trifecta of instruments is recommended by the HOME group. One study found that substituting the erythema component of the EASI scale with greyness for darker skin, in which erythema is more challenging to assess, did not significantly improve the reliability of EASI. This indicates that further research is needed to investigate how EASI can be modified to perform better in darker skin tones.”
She pointed out that some studies of clinician-reported outcome measures were underpowered to detect meaningful differences between patient subgroups. “There were also insufficient data to perform meta-regression of differences between patient subgroups,” she said. “Overall, future studies are needed to determine whether outcome measures recommended by the HOME and other tools perform equally well across diverse patient populations. This systematic review indicates significant reporting and knowledge gaps for psychometric properties of outcome measures by race, ethnicity, or skin tone in AD.”
Ms. Kaundinya reported having no relevant financial disclosures. Dr. Silverberg, the study’s senior author, is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
, according to results from a systematic review.
“AD is associated with considerable heterogeneity across different races and skin tones,” presenting study author Trisha Kaundinya said at the Revolutionizing Atopic Dermatitis symposium. “Compared with lighter skin tones, darker skin tones more commonly have diffuse xerosis, Dennis-Morgan lines, hyperlinearity of the palms, periorbital dark circles, lichenification, and prurigo nodularis. This heterogeneity can be challenging to assess in clinical trials and in practice.”
The Harmonizing Outcome Measures for Eczema (HOME) group has selected several scales by international consensus. For clinical trials, the group recommends the Patient-Oriented Eczema Measure (POEM), Eczema Area and Severity Index (EASI), and Dermatology Life Quality Index (DLQI). In clinical practice, the HOME group recommends the POEM, Patient-Oriented Scoring Atopic Dermatitis (PO-SCORAD), and the Numeric Rating Scale (NRS)-itch measures. “The psychometric validity and reliability of these outcome measures have undergone robust investigation before, but the validity and reliability of these outcome measures remains uncertain across different races, ethnicities, and skin tones,” Ms. Kaundinya said.
Jonathan Silverberg, MD, PhD, associate professor of dermatology at George Washington University, Washington, in collaboration with Andrew F. Alexis, MD, MPH, vice-chair for diversity and inclusion for the department of dermatology at Weill-Cornell Medicine, New York, and Jacob P. Thyssen, MD, PhD, at the University of Copenhagen, Denmark, sought to examine reporting of race, ethnicity, and skin tone, and to compare results across these groups from studies of psychometric properties for outcome measures in AD. Under the mentorship of Dr. Silverberg, Ms. Kaundinya, a medical student at Northwestern University, Chicago, and her research associates conducted a systematic review that searched PubMed and Embase and identified 165 relevant published studies of 41,146 individuals.
Of the individuals participating in these 165 studies, 73% had an unspecified racial background, 18% were White, 4% were Asian, 2% were Black, 2% were Hispanic, 1% were multiracial/other, and the remainder were American Indian/Alaskan Native. Only 55 of the studies (33%) reported the distribution of race or ethnicity, 5 (3%) reported the distribution of skin tone, and 16 (10%) reported psychometric differences in patients with different races, ethnicities, or skin tones. In addition, only 5 of 113 (4%) studies that did not report race, ethnicity, or skin tone–based differences acknowledged absence of stratification as a limitation.
Of note, significant differential item functioning was found between race subgroups for one or more items of the PO-SCORAD, the Patient-Reported Outcomes Measurement Information System (PROMIS) Itch Questionnaire (PIQ) Short Forms, POEM, DLQI, Hospital Anxiety and Depression Scale (HADS), Itchy Quality of Life (ItchyQOL) scale, 5-dimensions (5D) itch scale, Short Form (SF)-12, and NRS-itch. “Correlations of the POEM with the Investigator’s Global Assessment (IGA) differed the most between skin of color and lighter skin,” Ms. Kaundinya said.
“The POEM did seem to correlate similarly with the DLQI and the EASI in both white and nonwhite participants, which may indicate why this trifecta of instruments is recommended by the HOME group. One study found that substituting the erythema component of the EASI scale with greyness for darker skin, in which erythema is more challenging to assess, did not significantly improve the reliability of EASI. This indicates that further research is needed to investigate how EASI can be modified to perform better in darker skin tones.”
She pointed out that some studies of clinician-reported outcome measures were underpowered to detect meaningful differences between patient subgroups. “There were also insufficient data to perform meta-regression of differences between patient subgroups,” she said. “Overall, future studies are needed to determine whether outcome measures recommended by the HOME and other tools perform equally well across diverse patient populations. This systematic review indicates significant reporting and knowledge gaps for psychometric properties of outcome measures by race, ethnicity, or skin tone in AD.”
Ms. Kaundinya reported having no relevant financial disclosures. Dr. Silverberg, the study’s senior author, is a consultant to and/or an advisory board member for several pharmaceutical companies. He is also a speaker for Regeneron and Sanofi and has received a grant from Galderma.
FROM REVOLUTIONIZING AD 2021
Psoriasis
THE COMPARISON
A Elbow and forearm with erythematous, well-demarcated, pink plaques with mild micaceous scale in a 42-year-old White woman.
B Elbow and forearm with violaceous, well-demarcated plaques with micaceous scale and hyperpigmented patches around the active plaques in a 58-year-old Black man.
Epidemiology
Psoriasis prevalence in the United States has been estimated at 3.7%.1-3 If broken down by race or ethnicity, the prevalence of psoriasis varies: 2.5% to 3.7% in White adults1-4; 1.3% to 2% in Black adults1-4; 1.6% in Hispanics/other adults1-3; 1% in children overall; 0.29% in White children1,5; and 0.06% in Black children.1,5
Key clinical features in people with darker skin tones include:
- plaques that may appear more violaceous in color instead of pink or erythematous
- higher body surface area of involvement4 and thicker, more scaly plaques6
- increased likelihood of postinflammatory hyperpigmentation (PIH).
Worth noting
Although individuals of all skin tones may experience the psychosocial impact of psoriasis, quality-of-life measures have been found to be worse in those with skin of color (SOC) compared to White patients. 1,4 This may be due to the lingering PIH and hypopigmentation that occurs even after inflammatory plaques are treated. Of course, lack of access to care contributes to greater disease burden and more devastating psychological impact.
Health disparity highlight
Psoriasis may be underreported and underdiagnosed in individuals with SOC, as factors contributing to health care disparities may play a role, such as access to health care in general,1,7 and access to clinicians proficient in diagnosing cutaneous diseases in SOC may be delayed.8
Biologic medications are used less often in Black patients than in White patients, despite biologic medications being very efficacious for treatment of psoriasis.1,9,10
1. Kaufman BP, Alexis AF. Psoriasis in skin of color: insights into the epidemiology, clinical presentation, genetics, quality-of-life impact, and treatment of psoriasis in non-white racial/ethnic groups. Am J Clin Dermatol. 2018;19:405-423.
2. Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
3. Helmick CG, Lee-Han H, Hirsch SC, et al. Prevalence of psoriasis among adults in the U.S.: 2003-2006 and 2009-2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47:37-45.
4. Gelfand JM, Stern RS, Nijsten T, et al. The prevalence of psoriasis in African Americans: results from a population-based study. J Am Acad Dermatol. 2005;52:23-26.
5. Wu JJ, Black MH, Smith N, et al. Low prevalence of psoriasis among children and adolescents in a large multiethnic cohort in southern California. J Am Acad Dermatol. 2011;65:957-964.
6. Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
7. Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
8. Mundluru SN, Ramalingam ND, Tran HN. Addressing internal medicine residents’ discomfort with basic dermatology in persons of color in the primary care clinic. Am J Med Qual. 2019;34:513-513.
9. Kerr GS, Qaiyumi S, Richards J, et al. Psoriasis and psoriatic arthritis in African-American patients—the need to measure disease burden. Clin Rheumatol. 2015;34:1753-1759.
10. Takeshita J, Gelfand JM, Li P, et al. Psoriasis in the US Medicare population: prevalence, treatment, and factors associated with biologic use. J Invest Dermatol. 2015;135:2955-2963.
THE COMPARISON
A Elbow and forearm with erythematous, well-demarcated, pink plaques with mild micaceous scale in a 42-year-old White woman.
B Elbow and forearm with violaceous, well-demarcated plaques with micaceous scale and hyperpigmented patches around the active plaques in a 58-year-old Black man.

Epidemiology
Psoriasis prevalence in the United States has been estimated at 3.7%.1-3 If broken down by race or ethnicity, the prevalence of psoriasis varies: 2.5% to 3.7% in White adults1-4; 1.3% to 2% in Black adults1-4; 1.6% in Hispanics/other adults1-3; 1% in children overall; 0.29% in White children1,5; and 0.06% in Black children.1,5
Key clinical features in people with darker skin tones include:
- plaques that may appear more violaceous in color instead of pink or erythematous
- higher body surface area of involvement4 and thicker, more scaly plaques6
- increased likelihood of postinflammatory hyperpigmentation (PIH).
Worth noting
Although individuals of all skin tones may experience the psychosocial impact of psoriasis, quality-of-life measures have been found to be worse in those with skin of color (SOC) compared to White patients. 1,4 This may be due to the lingering PIH and hypopigmentation that occurs even after inflammatory plaques are treated. Of course, lack of access to care contributes to greater disease burden and more devastating psychological impact.
Health disparity highlight
Psoriasis may be underreported and underdiagnosed in individuals with SOC, as factors contributing to health care disparities may play a role, such as access to health care in general,1,7 and access to clinicians proficient in diagnosing cutaneous diseases in SOC may be delayed.8
Biologic medications are used less often in Black patients than in White patients, despite biologic medications being very efficacious for treatment of psoriasis.1,9,10
THE COMPARISON
A Elbow and forearm with erythematous, well-demarcated, pink plaques with mild micaceous scale in a 42-year-old White woman.
B Elbow and forearm with violaceous, well-demarcated plaques with micaceous scale and hyperpigmented patches around the active plaques in a 58-year-old Black man.

Epidemiology
Psoriasis prevalence in the United States has been estimated at 3.7%.1-3 If broken down by race or ethnicity, the prevalence of psoriasis varies: 2.5% to 3.7% in White adults1-4; 1.3% to 2% in Black adults1-4; 1.6% in Hispanics/other adults1-3; 1% in children overall; 0.29% in White children1,5; and 0.06% in Black children.1,5
Key clinical features in people with darker skin tones include:
- plaques that may appear more violaceous in color instead of pink or erythematous
- higher body surface area of involvement4 and thicker, more scaly plaques6
- increased likelihood of postinflammatory hyperpigmentation (PIH).
Worth noting
Although individuals of all skin tones may experience the psychosocial impact of psoriasis, quality-of-life measures have been found to be worse in those with skin of color (SOC) compared to White patients. 1,4 This may be due to the lingering PIH and hypopigmentation that occurs even after inflammatory plaques are treated. Of course, lack of access to care contributes to greater disease burden and more devastating psychological impact.
Health disparity highlight
Psoriasis may be underreported and underdiagnosed in individuals with SOC, as factors contributing to health care disparities may play a role, such as access to health care in general,1,7 and access to clinicians proficient in diagnosing cutaneous diseases in SOC may be delayed.8
Biologic medications are used less often in Black patients than in White patients, despite biologic medications being very efficacious for treatment of psoriasis.1,9,10
1. Kaufman BP, Alexis AF. Psoriasis in skin of color: insights into the epidemiology, clinical presentation, genetics, quality-of-life impact, and treatment of psoriasis in non-white racial/ethnic groups. Am J Clin Dermatol. 2018;19:405-423.
2. Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
3. Helmick CG, Lee-Han H, Hirsch SC, et al. Prevalence of psoriasis among adults in the U.S.: 2003-2006 and 2009-2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47:37-45.
4. Gelfand JM, Stern RS, Nijsten T, et al. The prevalence of psoriasis in African Americans: results from a population-based study. J Am Acad Dermatol. 2005;52:23-26.
5. Wu JJ, Black MH, Smith N, et al. Low prevalence of psoriasis among children and adolescents in a large multiethnic cohort in southern California. J Am Acad Dermatol. 2011;65:957-964.
6. Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
7. Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
8. Mundluru SN, Ramalingam ND, Tran HN. Addressing internal medicine residents’ discomfort with basic dermatology in persons of color in the primary care clinic. Am J Med Qual. 2019;34:513-513.
9. Kerr GS, Qaiyumi S, Richards J, et al. Psoriasis and psoriatic arthritis in African-American patients—the need to measure disease burden. Clin Rheumatol. 2015;34:1753-1759.
10. Takeshita J, Gelfand JM, Li P, et al. Psoriasis in the US Medicare population: prevalence, treatment, and factors associated with biologic use. J Invest Dermatol. 2015;135:2955-2963.
1. Kaufman BP, Alexis AF. Psoriasis in skin of color: insights into the epidemiology, clinical presentation, genetics, quality-of-life impact, and treatment of psoriasis in non-white racial/ethnic groups. Am J Clin Dermatol. 2018;19:405-423.
2. Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
3. Helmick CG, Lee-Han H, Hirsch SC, et al. Prevalence of psoriasis among adults in the U.S.: 2003-2006 and 2009-2010 National Health and Nutrition Examination Surveys. Am J Prev Med. 2014;47:37-45.
4. Gelfand JM, Stern RS, Nijsten T, et al. The prevalence of psoriasis in African Americans: results from a population-based study. J Am Acad Dermatol. 2005;52:23-26.
5. Wu JJ, Black MH, Smith N, et al. Low prevalence of psoriasis among children and adolescents in a large multiethnic cohort in southern California. J Am Acad Dermatol. 2011;65:957-964.
6. Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
7. Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
8. Mundluru SN, Ramalingam ND, Tran HN. Addressing internal medicine residents’ discomfort with basic dermatology in persons of color in the primary care clinic. Am J Med Qual. 2019;34:513-513.
9. Kerr GS, Qaiyumi S, Richards J, et al. Psoriasis and psoriatic arthritis in African-American patients—the need to measure disease burden. Clin Rheumatol. 2015;34:1753-1759.
10. Takeshita J, Gelfand JM, Li P, et al. Psoriasis in the US Medicare population: prevalence, treatment, and factors associated with biologic use. J Invest Dermatol. 2015;135:2955-2963.
Dyspigmentation common in SOC patients with bullous pemphigoid
Patients of skin of color (SOC) with bullous pemphigoid presented significantly more often with dyspigmentation than did White patients in a retrospective observational study of patients diagnosed with BP at New York University Langone Health and Bellevue Hospital, also in New York.
“Dyspigmentation in the skin-of-color patient population is important to recognize not only for an objective evaluation of the disease process, but also from a quality of life perspective ... to ensure there is timely diagnosis and initiation of treatment in the skin-of-color population,” said medical student Payal Shah, BS, of New York University, in presenting the findings at the annual Skin of Color Society symposium.
Ms. Shah and coresearchers identified 94 cases of BP through retrospective view of electronic health records – 59 in White patients and 35 in SOC patients. The physical examination features most commonly found at initial presentation were bullae or vesicles in both White patients (64.4% ) and SOC patients (80%). Erosions or ulcers were also commonly found in both groups (42.4% of White patients and 60% of SOC patients).
Erythema was more commonly found in White patients at initial presentation: 35.6% vs. 14.3% of SOC patients (P = .032). Dyspigmentation, defined as areas of hyper- or hypopigmentation, was more commonly found in SOC patients: 54.3% versus 10.2% in White patients (P < .001). The difference in erythema of inflammatory bullae in BP may stem from the fact that erythema is more difficult to discern in patients with darker skin types, Ms. Shah said.
SOC patients also were significantly younger at the time of initial presentation; their mean age was 63 years, compared with 77 years in the White population (P < .001).
The time to diagnosis, defined as the time from initial symptoms to dermatologic diagnosis, was greater for the SOC population –7.6 months vs. 6.2 months for white patients –though the difference was not statistically significant, they said in the abstract .
Dyspigmentation has been shown to be among the top dermatologic concerns of Black patients and has important quality of life implications. “Early diagnosis to prevent difficult-to-treat dyspigmentation is therefore of utmost importance,” they said in the abstract.
Prior research has demonstrated that non-White populations are at greater risk for hospitalization secondary to BP and have a greater risk of disease mortality, Ms. Shah noted in her presentation.
Patients of skin of color (SOC) with bullous pemphigoid presented significantly more often with dyspigmentation than did White patients in a retrospective observational study of patients diagnosed with BP at New York University Langone Health and Bellevue Hospital, also in New York.
“Dyspigmentation in the skin-of-color patient population is important to recognize not only for an objective evaluation of the disease process, but also from a quality of life perspective ... to ensure there is timely diagnosis and initiation of treatment in the skin-of-color population,” said medical student Payal Shah, BS, of New York University, in presenting the findings at the annual Skin of Color Society symposium.
Ms. Shah and coresearchers identified 94 cases of BP through retrospective view of electronic health records – 59 in White patients and 35 in SOC patients. The physical examination features most commonly found at initial presentation were bullae or vesicles in both White patients (64.4% ) and SOC patients (80%). Erosions or ulcers were also commonly found in both groups (42.4% of White patients and 60% of SOC patients).
Erythema was more commonly found in White patients at initial presentation: 35.6% vs. 14.3% of SOC patients (P = .032). Dyspigmentation, defined as areas of hyper- or hypopigmentation, was more commonly found in SOC patients: 54.3% versus 10.2% in White patients (P < .001). The difference in erythema of inflammatory bullae in BP may stem from the fact that erythema is more difficult to discern in patients with darker skin types, Ms. Shah said.
SOC patients also were significantly younger at the time of initial presentation; their mean age was 63 years, compared with 77 years in the White population (P < .001).
The time to diagnosis, defined as the time from initial symptoms to dermatologic diagnosis, was greater for the SOC population –7.6 months vs. 6.2 months for white patients –though the difference was not statistically significant, they said in the abstract .
Dyspigmentation has been shown to be among the top dermatologic concerns of Black patients and has important quality of life implications. “Early diagnosis to prevent difficult-to-treat dyspigmentation is therefore of utmost importance,” they said in the abstract.
Prior research has demonstrated that non-White populations are at greater risk for hospitalization secondary to BP and have a greater risk of disease mortality, Ms. Shah noted in her presentation.
Patients of skin of color (SOC) with bullous pemphigoid presented significantly more often with dyspigmentation than did White patients in a retrospective observational study of patients diagnosed with BP at New York University Langone Health and Bellevue Hospital, also in New York.
“Dyspigmentation in the skin-of-color patient population is important to recognize not only for an objective evaluation of the disease process, but also from a quality of life perspective ... to ensure there is timely diagnosis and initiation of treatment in the skin-of-color population,” said medical student Payal Shah, BS, of New York University, in presenting the findings at the annual Skin of Color Society symposium.
Ms. Shah and coresearchers identified 94 cases of BP through retrospective view of electronic health records – 59 in White patients and 35 in SOC patients. The physical examination features most commonly found at initial presentation were bullae or vesicles in both White patients (64.4% ) and SOC patients (80%). Erosions or ulcers were also commonly found in both groups (42.4% of White patients and 60% of SOC patients).
Erythema was more commonly found in White patients at initial presentation: 35.6% vs. 14.3% of SOC patients (P = .032). Dyspigmentation, defined as areas of hyper- or hypopigmentation, was more commonly found in SOC patients: 54.3% versus 10.2% in White patients (P < .001). The difference in erythema of inflammatory bullae in BP may stem from the fact that erythema is more difficult to discern in patients with darker skin types, Ms. Shah said.
SOC patients also were significantly younger at the time of initial presentation; their mean age was 63 years, compared with 77 years in the White population (P < .001).
The time to diagnosis, defined as the time from initial symptoms to dermatologic diagnosis, was greater for the SOC population –7.6 months vs. 6.2 months for white patients –though the difference was not statistically significant, they said in the abstract .
Dyspigmentation has been shown to be among the top dermatologic concerns of Black patients and has important quality of life implications. “Early diagnosis to prevent difficult-to-treat dyspigmentation is therefore of utmost importance,” they said in the abstract.
Prior research has demonstrated that non-White populations are at greater risk for hospitalization secondary to BP and have a greater risk of disease mortality, Ms. Shah noted in her presentation.
FROM SOC 2021