User login
Biologics for pediatric psoriasis don’t increase infection risk
AUSTIN, TEX. – Among children with psoriasis, there appears to be no strong evidence that biologic immunomodulating drugs increase the 6-month risk of serious infections, compared with systemic nonbiologics or phototherapy, according to results from the largest population-based study of its kind to date.
However, children with psoriasis face a 64% increased risk of infection, compared with risk-matched pediatric patients without the disease.
“We know that pediatric psoriasis affects up to 1.3% of children, and we know that is associated with multiple potential comorbidities and that it has a significant impact on quality of life for children affected,” lead study author Maria Schneeweiss, MD, said at the annual meeting of the Society for Pediatric Dermatology. “Increasingly, we see that biologic and nonbiologic systemic agents are used to treat moderate to severe pediatric psoriasis. While we have a lot of experience in adult psoriasis and a lot of comparative safety studies of these drugs in adult psoriasis, there are very few population-based studies on the safety of these systemic agents for treating pediatric psoriasis.”
In an effort to evaluate the 6-month risk of serious bacterial and opportunistic infections in children with psoriasis treated with systemic immunomodulatory medications, Dr. Schneeweiss and Joseph F. Merola, MD, of the department of dermatology at Brigham and Women’s Hospital, Boston, and Jennifer Huang, MD, of Boston Children’s Hospital drew from longitudinal, patient-level U.S. claims data in the MarketScan database between 2003 and 2017. They limited the analysis to patients younger than age 18; those who had a recorded diagnosis of psoriasis; and those who were treated with biologics, nonbiologic immunomodulatory agents, or phototherapy. The researchers used hospital discharge diagnoses to compute the risk of serious bacterial and opportunistic infections, and propensity score matching to determine relative risks.
A total of 54,355 children with psoriasis were identified in the database. Before propensity score matching, 635 patients initiated biologic therapy, 919 initiated nonbiologic systemic agents, and 2,537 initiated phototherapy. Their mean age was 12-14 years and slightly more than half were female. In nonbiologic initiators, the 6-month risk of serious infections was 4.75 per 1,000 patients, while in biologic initiators it was 5.44 per 1,000 patients, resulting in a propensity score–matched ratio of 0.60. There was no statistically significant increased risk when the use of nonbiologics was compared with the use of phototherapy.
Independent of treatment, the risk of infection among psoriasis patients was 1.1 per 1,000 patients, which was 60% higher than matched pediatric patients without psoriasis (risk ratio, 1.64).
“When treating pediatric patients with psoriasis, clinicians should remain mindful that the presence of psoriasis itself may increase the risk of infection in children and adolescents, independent of treatment, but that biologic immunomodulatory agents do not further increase that risk,” Dr. Schneeweiss said in an interview. “Our findings suggest that, while there may be an increased risk of certain infections based on the presence of psoriasis alone, all appropriate treatment options should be discussed with patients in shared decision making with their physician. Patients should understand the risks, benefits, and alternatives to any treatment option but not necessarily be restricted as such and have access to newer, targeted and highly effective therapy as appropriate to each individual case.”
She added that, based on the National Psoriasis Foundation guidance of treat-to-target strategies, “our pediatric patients should be offered the same level of disease control as all psoriasis patients.”
She acknowledged certain limitations of the analysis, including the inability to stratify by disease severity and to determine specific doses of medication used.
The study was funded by the Brigham and Women’s Hospital departments of dermatology and medicine. Dr. Schneeweiss and Dr. Huang reported having no financial disclosures. Dr. Merola reported that he has served as a consultant and/or investigator for numerous pharmaceutical companies.
AUSTIN, TEX. – Among children with psoriasis, there appears to be no strong evidence that biologic immunomodulating drugs increase the 6-month risk of serious infections, compared with systemic nonbiologics or phototherapy, according to results from the largest population-based study of its kind to date.
However, children with psoriasis face a 64% increased risk of infection, compared with risk-matched pediatric patients without the disease.
“We know that pediatric psoriasis affects up to 1.3% of children, and we know that is associated with multiple potential comorbidities and that it has a significant impact on quality of life for children affected,” lead study author Maria Schneeweiss, MD, said at the annual meeting of the Society for Pediatric Dermatology. “Increasingly, we see that biologic and nonbiologic systemic agents are used to treat moderate to severe pediatric psoriasis. While we have a lot of experience in adult psoriasis and a lot of comparative safety studies of these drugs in adult psoriasis, there are very few population-based studies on the safety of these systemic agents for treating pediatric psoriasis.”
In an effort to evaluate the 6-month risk of serious bacterial and opportunistic infections in children with psoriasis treated with systemic immunomodulatory medications, Dr. Schneeweiss and Joseph F. Merola, MD, of the department of dermatology at Brigham and Women’s Hospital, Boston, and Jennifer Huang, MD, of Boston Children’s Hospital drew from longitudinal, patient-level U.S. claims data in the MarketScan database between 2003 and 2017. They limited the analysis to patients younger than age 18; those who had a recorded diagnosis of psoriasis; and those who were treated with biologics, nonbiologic immunomodulatory agents, or phototherapy. The researchers used hospital discharge diagnoses to compute the risk of serious bacterial and opportunistic infections, and propensity score matching to determine relative risks.
A total of 54,355 children with psoriasis were identified in the database. Before propensity score matching, 635 patients initiated biologic therapy, 919 initiated nonbiologic systemic agents, and 2,537 initiated phototherapy. Their mean age was 12-14 years and slightly more than half were female. In nonbiologic initiators, the 6-month risk of serious infections was 4.75 per 1,000 patients, while in biologic initiators it was 5.44 per 1,000 patients, resulting in a propensity score–matched ratio of 0.60. There was no statistically significant increased risk when the use of nonbiologics was compared with the use of phototherapy.
Independent of treatment, the risk of infection among psoriasis patients was 1.1 per 1,000 patients, which was 60% higher than matched pediatric patients without psoriasis (risk ratio, 1.64).
“When treating pediatric patients with psoriasis, clinicians should remain mindful that the presence of psoriasis itself may increase the risk of infection in children and adolescents, independent of treatment, but that biologic immunomodulatory agents do not further increase that risk,” Dr. Schneeweiss said in an interview. “Our findings suggest that, while there may be an increased risk of certain infections based on the presence of psoriasis alone, all appropriate treatment options should be discussed with patients in shared decision making with their physician. Patients should understand the risks, benefits, and alternatives to any treatment option but not necessarily be restricted as such and have access to newer, targeted and highly effective therapy as appropriate to each individual case.”
She added that, based on the National Psoriasis Foundation guidance of treat-to-target strategies, “our pediatric patients should be offered the same level of disease control as all psoriasis patients.”
She acknowledged certain limitations of the analysis, including the inability to stratify by disease severity and to determine specific doses of medication used.
The study was funded by the Brigham and Women’s Hospital departments of dermatology and medicine. Dr. Schneeweiss and Dr. Huang reported having no financial disclosures. Dr. Merola reported that he has served as a consultant and/or investigator for numerous pharmaceutical companies.
AUSTIN, TEX. – Among children with psoriasis, there appears to be no strong evidence that biologic immunomodulating drugs increase the 6-month risk of serious infections, compared with systemic nonbiologics or phototherapy, according to results from the largest population-based study of its kind to date.
However, children with psoriasis face a 64% increased risk of infection, compared with risk-matched pediatric patients without the disease.
“We know that pediatric psoriasis affects up to 1.3% of children, and we know that is associated with multiple potential comorbidities and that it has a significant impact on quality of life for children affected,” lead study author Maria Schneeweiss, MD, said at the annual meeting of the Society for Pediatric Dermatology. “Increasingly, we see that biologic and nonbiologic systemic agents are used to treat moderate to severe pediatric psoriasis. While we have a lot of experience in adult psoriasis and a lot of comparative safety studies of these drugs in adult psoriasis, there are very few population-based studies on the safety of these systemic agents for treating pediatric psoriasis.”
In an effort to evaluate the 6-month risk of serious bacterial and opportunistic infections in children with psoriasis treated with systemic immunomodulatory medications, Dr. Schneeweiss and Joseph F. Merola, MD, of the department of dermatology at Brigham and Women’s Hospital, Boston, and Jennifer Huang, MD, of Boston Children’s Hospital drew from longitudinal, patient-level U.S. claims data in the MarketScan database between 2003 and 2017. They limited the analysis to patients younger than age 18; those who had a recorded diagnosis of psoriasis; and those who were treated with biologics, nonbiologic immunomodulatory agents, or phototherapy. The researchers used hospital discharge diagnoses to compute the risk of serious bacterial and opportunistic infections, and propensity score matching to determine relative risks.
A total of 54,355 children with psoriasis were identified in the database. Before propensity score matching, 635 patients initiated biologic therapy, 919 initiated nonbiologic systemic agents, and 2,537 initiated phototherapy. Their mean age was 12-14 years and slightly more than half were female. In nonbiologic initiators, the 6-month risk of serious infections was 4.75 per 1,000 patients, while in biologic initiators it was 5.44 per 1,000 patients, resulting in a propensity score–matched ratio of 0.60. There was no statistically significant increased risk when the use of nonbiologics was compared with the use of phototherapy.
Independent of treatment, the risk of infection among psoriasis patients was 1.1 per 1,000 patients, which was 60% higher than matched pediatric patients without psoriasis (risk ratio, 1.64).
“When treating pediatric patients with psoriasis, clinicians should remain mindful that the presence of psoriasis itself may increase the risk of infection in children and adolescents, independent of treatment, but that biologic immunomodulatory agents do not further increase that risk,” Dr. Schneeweiss said in an interview. “Our findings suggest that, while there may be an increased risk of certain infections based on the presence of psoriasis alone, all appropriate treatment options should be discussed with patients in shared decision making with their physician. Patients should understand the risks, benefits, and alternatives to any treatment option but not necessarily be restricted as such and have access to newer, targeted and highly effective therapy as appropriate to each individual case.”
She added that, based on the National Psoriasis Foundation guidance of treat-to-target strategies, “our pediatric patients should be offered the same level of disease control as all psoriasis patients.”
She acknowledged certain limitations of the analysis, including the inability to stratify by disease severity and to determine specific doses of medication used.
The study was funded by the Brigham and Women’s Hospital departments of dermatology and medicine. Dr. Schneeweiss and Dr. Huang reported having no financial disclosures. Dr. Merola reported that he has served as a consultant and/or investigator for numerous pharmaceutical companies.
REPORTING FROM SPD 2019
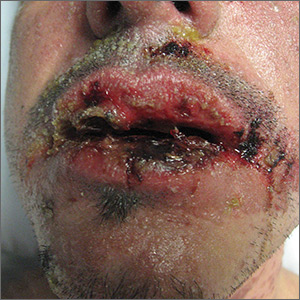
Rash with fever and shortness of breath
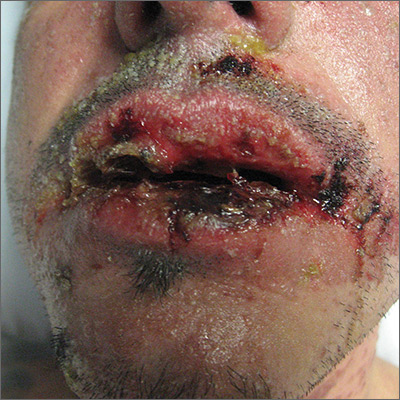
The skin biopsy confirmed toxic epidermal necrolysis (TEN), possibly secondary to one of the antibiotics he’d been given upon hospital admission. The health care team suspected that the patient had Stevens-Johns Syndrome when he sought care and that it had evolved into TEN. Both diseases are life-threatening and require intensive in-hospital care.
TEN is part of a spectrum of disorders that includes erythema multiforme (< 10% of the body surface is involved); SJS/TEN (10%-30% involvement with erythematous or pruritic macules, widespread blisters on the trunk and face, and erosions of ≥ 1 mucous membranes); and TEN (> 30% involvement).
Drugs most commonly known to cause SJS and TEN include sulfonamide antibiotics, allopurinol, nonsteroidal anti-inflammatory agents, amine antiepileptic drugs (phenytoin and carbamazepine), and lamotrigine. Fifty percent of SJS/TEN cases have no identifiable cause. Not all SJS is secondary to drug exposure, but it is the job of the clinician to investigate this cause and stop any suspicious medications.
In this case, trimethoprim/sulfamethoxazole was discontinued immediately and the patient was transferred to a burn unit and given intravenous gamma-globulin 1 g/kg for 3 days. Fortunately, the patient survived with intensive supportive care in the burn unit. The exfoliation of > 30% of the skin is like a large secondary burn, and a burn unit is the optimal location for lifesaving measures.
Photo courtesy of Robert T. Gilson, MD, and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Milana C, Smith M. Erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1161-1168.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the Color Atlas of Family Medicine app by clicking on this link: usatinemedia.com

The skin biopsy confirmed toxic epidermal necrolysis (TEN), possibly secondary to one of the antibiotics he’d been given upon hospital admission. The health care team suspected that the patient had Stevens-Johns Syndrome when he sought care and that it had evolved into TEN. Both diseases are life-threatening and require intensive in-hospital care.
TEN is part of a spectrum of disorders that includes erythema multiforme (< 10% of the body surface is involved); SJS/TEN (10%-30% involvement with erythematous or pruritic macules, widespread blisters on the trunk and face, and erosions of ≥ 1 mucous membranes); and TEN (> 30% involvement).
Drugs most commonly known to cause SJS and TEN include sulfonamide antibiotics, allopurinol, nonsteroidal anti-inflammatory agents, amine antiepileptic drugs (phenytoin and carbamazepine), and lamotrigine. Fifty percent of SJS/TEN cases have no identifiable cause. Not all SJS is secondary to drug exposure, but it is the job of the clinician to investigate this cause and stop any suspicious medications.
In this case, trimethoprim/sulfamethoxazole was discontinued immediately and the patient was transferred to a burn unit and given intravenous gamma-globulin 1 g/kg for 3 days. Fortunately, the patient survived with intensive supportive care in the burn unit. The exfoliation of > 30% of the skin is like a large secondary burn, and a burn unit is the optimal location for lifesaving measures.
Photo courtesy of Robert T. Gilson, MD, and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Milana C, Smith M. Erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1161-1168.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the Color Atlas of Family Medicine app by clicking on this link: usatinemedia.com

The skin biopsy confirmed toxic epidermal necrolysis (TEN), possibly secondary to one of the antibiotics he’d been given upon hospital admission. The health care team suspected that the patient had Stevens-Johns Syndrome when he sought care and that it had evolved into TEN. Both diseases are life-threatening and require intensive in-hospital care.
TEN is part of a spectrum of disorders that includes erythema multiforme (< 10% of the body surface is involved); SJS/TEN (10%-30% involvement with erythematous or pruritic macules, widespread blisters on the trunk and face, and erosions of ≥ 1 mucous membranes); and TEN (> 30% involvement).
Drugs most commonly known to cause SJS and TEN include sulfonamide antibiotics, allopurinol, nonsteroidal anti-inflammatory agents, amine antiepileptic drugs (phenytoin and carbamazepine), and lamotrigine. Fifty percent of SJS/TEN cases have no identifiable cause. Not all SJS is secondary to drug exposure, but it is the job of the clinician to investigate this cause and stop any suspicious medications.
In this case, trimethoprim/sulfamethoxazole was discontinued immediately and the patient was transferred to a burn unit and given intravenous gamma-globulin 1 g/kg for 3 days. Fortunately, the patient survived with intensive supportive care in the burn unit. The exfoliation of > 30% of the skin is like a large secondary burn, and a burn unit is the optimal location for lifesaving measures.
Photo courtesy of Robert T. Gilson, MD, and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Milana C, Smith M. Erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis In: Usatine R, Smith M, Mayeaux EJ, et al. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1161-1168.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the Color Atlas of Family Medicine app by clicking on this link: usatinemedia.com
Hadlima approved as fourth adalimumab biosimilar in U.S.
The Food and Drug Administration has approved the Humira biosimilar Hadlima (adalimumab-bwwd), making it the fourth adalimumab biosimilar approved in the United States, the agency announced.
Hadlima is approved for seven of the reference product’s indications, which include rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, and ulcerative colitis.
The product will launch in the United States on June 30, 2023. Other FDA-approved adalimumab biosimilars – Amjevita (adalimunab-atto), Cyltezo (adalimumab-adbm), Hyrimoz (adalimumab-adaz) – similarly will not reach the U.S. market until 2023.
Hadlima is developed by Samsung Bioepis and commercialized by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co.
*This article was updated on July 24, 2019.
The Food and Drug Administration has approved the Humira biosimilar Hadlima (adalimumab-bwwd), making it the fourth adalimumab biosimilar approved in the United States, the agency announced.
Hadlima is approved for seven of the reference product’s indications, which include rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, and ulcerative colitis.
The product will launch in the United States on June 30, 2023. Other FDA-approved adalimumab biosimilars – Amjevita (adalimunab-atto), Cyltezo (adalimumab-adbm), Hyrimoz (adalimumab-adaz) – similarly will not reach the U.S. market until 2023.
Hadlima is developed by Samsung Bioepis and commercialized by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co.
*This article was updated on July 24, 2019.
The Food and Drug Administration has approved the Humira biosimilar Hadlima (adalimumab-bwwd), making it the fourth adalimumab biosimilar approved in the United States, the agency announced.
Hadlima is approved for seven of the reference product’s indications, which include rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, plaque psoriasis, psoriatic arthritis, ankylosing spondylitis, adult Crohn’s disease, and ulcerative colitis.
The product will launch in the United States on June 30, 2023. Other FDA-approved adalimumab biosimilars – Amjevita (adalimunab-atto), Cyltezo (adalimumab-adbm), Hyrimoz (adalimumab-adaz) – similarly will not reach the U.S. market until 2023.
Hadlima is developed by Samsung Bioepis and commercialized by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co.
*This article was updated on July 24, 2019.
FDA approves Otezla for treatment of Behçet’s-associated oral ulcers
The Food and Drug Administration has expanded the indication for apremilast (Otezla) to include the treatment of oral ulcers associated with Behçet’s disease in adults, according to an announcement from the manufacturer, Celgene.
FDA approval was based on results of the randomized, placebo-controlled, double-blind, phase 3 RELIEF trial, in which 207 patients with Behçet’s disease with active ulcers underwent treatment for 12 weeks with 30 mg apremilast or placebo. When measured on a visual analog scale, the reduction in pain from oral ulcers after 12 weeks in patients receiving apremilast was 42.7 points, compared with 18.7 points in the placebo group. Just over 50% of apremilast patients achieved complete response by week 12, compared with 22.3% in the placebo group.
The most common adverse events associated with apremilast during RELIEF were diarrhea, nausea, headache, and upper respiratory infection. This was consistent with apremilast’s known safety profile.
Apremilast is also indicated for treatment of patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, and for patients with active psoriatic arthritis.
“Oral ulcers are a recurring and debilitating manifestation that affects nearly everyone living with Behçet’s disease and have an important negative impact on the quality of life for these patients. In the clinical trial, Otezla demonstrated improvements in measures of oral ulcers at week 12. Otezla has the potential to be a needed treatment option for U.S. patients and their physicians, who previously had limited options available,” Yusuf Yazici, MD, clinical associate professor in the department of medicine at New York University, said in the announcement.
The Food and Drug Administration has expanded the indication for apremilast (Otezla) to include the treatment of oral ulcers associated with Behçet’s disease in adults, according to an announcement from the manufacturer, Celgene.
FDA approval was based on results of the randomized, placebo-controlled, double-blind, phase 3 RELIEF trial, in which 207 patients with Behçet’s disease with active ulcers underwent treatment for 12 weeks with 30 mg apremilast or placebo. When measured on a visual analog scale, the reduction in pain from oral ulcers after 12 weeks in patients receiving apremilast was 42.7 points, compared with 18.7 points in the placebo group. Just over 50% of apremilast patients achieved complete response by week 12, compared with 22.3% in the placebo group.
The most common adverse events associated with apremilast during RELIEF were diarrhea, nausea, headache, and upper respiratory infection. This was consistent with apremilast’s known safety profile.
Apremilast is also indicated for treatment of patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, and for patients with active psoriatic arthritis.
“Oral ulcers are a recurring and debilitating manifestation that affects nearly everyone living with Behçet’s disease and have an important negative impact on the quality of life for these patients. In the clinical trial, Otezla demonstrated improvements in measures of oral ulcers at week 12. Otezla has the potential to be a needed treatment option for U.S. patients and their physicians, who previously had limited options available,” Yusuf Yazici, MD, clinical associate professor in the department of medicine at New York University, said in the announcement.
The Food and Drug Administration has expanded the indication for apremilast (Otezla) to include the treatment of oral ulcers associated with Behçet’s disease in adults, according to an announcement from the manufacturer, Celgene.
FDA approval was based on results of the randomized, placebo-controlled, double-blind, phase 3 RELIEF trial, in which 207 patients with Behçet’s disease with active ulcers underwent treatment for 12 weeks with 30 mg apremilast or placebo. When measured on a visual analog scale, the reduction in pain from oral ulcers after 12 weeks in patients receiving apremilast was 42.7 points, compared with 18.7 points in the placebo group. Just over 50% of apremilast patients achieved complete response by week 12, compared with 22.3% in the placebo group.
The most common adverse events associated with apremilast during RELIEF were diarrhea, nausea, headache, and upper respiratory infection. This was consistent with apremilast’s known safety profile.
Apremilast is also indicated for treatment of patients with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, and for patients with active psoriatic arthritis.
“Oral ulcers are a recurring and debilitating manifestation that affects nearly everyone living with Behçet’s disease and have an important negative impact on the quality of life for these patients. In the clinical trial, Otezla demonstrated improvements in measures of oral ulcers at week 12. Otezla has the potential to be a needed treatment option for U.S. patients and their physicians, who previously had limited options available,” Yusuf Yazici, MD, clinical associate professor in the department of medicine at New York University, said in the announcement.
Expert shares contact dermatitis trends
AUSTIN – Not long ago, Rajani Katta, MD, received a text message from a friend who expressed concern about a rash that developed in the underarm of her teenage daughter.
The culprit turned out to be the lavender essential oil contained in an “all natural” deodorant that her daughter had recently switched to – a storyline that Dr. Katta encounters with increasing frequency in her role as clinical professor of dermatology at the University of Texas Health Science Center at Houston.
Dr. Katta said at the annual meeting of the Society for Pediatric Dermatology. “When you talk about a natural allergy, it is more likely to occur if your skin barrier is compromised, so I think that’s why we’re seeing it, especially in young girls in the underarm area. If you shave the underarm, you impair that skin barrier and you’re more likely to develop a reaction to something you’re using over it.”
Her list of recommended deodorants includes Almay Roll-On Antiperspirant & Deodorant, Crystal Body Deodorant Stick, Crystal Roll-On Body Deodorant, Vanicream Deodorant for Sensitive Skin (aluminum-free), Vanicream Antiperspirant/Deodorant, and CertainDri Clinical Strength Roll-On. They are fragrance-free and lack propylene glycol, which is a common allergen.
Increasingly, essential oils are being added to lip balms and toothpastes, said Dr. Katta, who is also author of the 2018 book “Glow: The Dermatologist’s Guide to a Whole Foods Younger Skin Diet.” She recalled one patient who presented with chronic chapped lips. “It doesn’t matter how many lip glosses I try; it just keeps getting worse,” the patient told her. The likely culprit turned out to be ingredients contained in flavored lip balm from EOS. Reports of blistering and cracking of the lips from use of the products prompted a class action lawsuit and a notice to consumers from the Food and Drug Administration.
Another patient presented with cracked lips after switching to an “all natural” toothpaste that was labeled “gluten free.”
“It looked great,” Dr. Katta recalled. “Unfortunately it was not flavoring free. She reacted to multiple essential oils, including tea tree oil, contained in the toothpaste. This is being added to a number of toothpastes, and I think we’re going to see more of these types of reactions.”
Other toothpastes contain balsam of Peru, “which is consistently one of the top allergens in patch test clinics,” she said. “One of the components of balsam of Peru is a cinnamon compound, which can be an issue.”
Dr. Katta advises her patients to use Vaseline petroleum jelly as a lip balm and recommends Tom’s of Maine Silly Strawberry Flavor (this flavor only) toothpaste for children.
A few years ago, a teenager presented to Dr. Katta with intense bullae on the dorsum of the foot after wearing shoes without socks. “She was wearing white canvas Keds, which looked very innocuous,” she said. Patch testing revealed that the teen reacted to four different rubber accelerators. “When we contacted the company, they [acknowledged] using rubber cement to make the canvas Keds,” Dr. Katta said. “Rubber cement is an adhesive and it does contain rubber accelerators. Later, I saw two cases of children who had walked around all day at the amusement park wearing their Sperry Topsiders without any socks. We couldn’t get any information from that manufacturer, but I suspect that they also use a rubber-based glue to make those shoes.” She characterized shoes as “a real setup for a foot allergy because you have friction, sweat that’s pulling allergen out of an object, and sweat is carrying it over, especially to the dorsum of the foot.”
Dr. Katta has also noticed an uptick in the number of young patients who develop allergic reactions to dyes used to make workout clothing. “If you ever see rashes that do not involve the axillary vault but do have peraxillary accentuation, think textile allergy,” she said. “We’re seeing a lot of reactions to disperse blue clothing dyes. When you think about textile allergy from the dyes, it tends to be the blue and black clothing. It’s more likely in the setting of synthetic fabrics because they leach out dyes more easily, and it’s more likely in the setting of sweat because sweat helps pull allergen out. I’m seeing it a lot from sports uniforms and tight black leggings and tight sports bras that people are wearing. I’m also seeing some from bathing suits and swim shirts.”
Exposure to products containing the preservative methylisothiazolinone (MI) is also on the rise. It ranks as the second most frequent allergen for which the North American Contact Dermatitis Group is seeing positive results on patch testing, with rates of 13.4%. MI can be found in many skin care products and “probably about half of school glues, fabric glues, and craft glues,” Dr. Katta said. “Stick versus liquid doesn’t make a difference.” Children and teens often use craft glues, laundry detergents, and other products to create “slime” as a way to learn about viscosity, polymers, and chemical reactions. “Sometimes these children have sensitive skin, or they’re using it with prolonged contact, so they may be sensitizing themselves to the MI,” she said.
She concluded her remarks by noting that an increasing number of young patients are developing reactions to wearable medical devices such as insulin pumps and glucose monitors. “With this, the first thing to think about is frictional irritant dermatitis,” she said. “You can put Scanpor medical paper tape on people’s back for 48 hours straight to patch test them. Some people are incredibly reactive to the friction of just that tape. You also have to think about trapped allergen. One of my patients reacted to colophony, fragrance mix, and propylene glycol, all of which were contained in his skin care products. Some people are getting advice from other patients to use Mastisol liquid adhesive to help their glucose monitors stick better. Mastisol has a high rate of cross-reactivity with balsam of Peru, so it’s a fragrance allergen. That’s the first thing you want to ask patients about: what products they’re using.”
One of her patients thought she was reacting to adhesive tape on her skin, but in fact she was reacting to two different acrylates: ethylene glycol dimethacrylate (EGDMA) and hydroxyethyl methacrylate (HEMA). “I know about these allergens because I see reactions from butterfly needles in dialysis patients,” Dr. Katta explained. “What happens is, these acrylates are glues or plastics used somewhere else on the device, and they can migrate through barriers.”
In one published case, a 9-year-old boy developed a reaction to ethyl cyanoacrylate contained in a glucose sensor adhesive (Dermatitis. 2017; 28[4]:289-91). It never touched the boy’s skin directly but was presumed to migrate through that tape. “The bottom line is that acrylates may induce contact dermatitis even through perceived barriers,” she said. “Their use anywhere in medical devices may prove problematic.”
Dr. Katta reported that she is a member of the advisory board for Vichy Laboratories.
AUSTIN – Not long ago, Rajani Katta, MD, received a text message from a friend who expressed concern about a rash that developed in the underarm of her teenage daughter.
The culprit turned out to be the lavender essential oil contained in an “all natural” deodorant that her daughter had recently switched to – a storyline that Dr. Katta encounters with increasing frequency in her role as clinical professor of dermatology at the University of Texas Health Science Center at Houston.
Dr. Katta said at the annual meeting of the Society for Pediatric Dermatology. “When you talk about a natural allergy, it is more likely to occur if your skin barrier is compromised, so I think that’s why we’re seeing it, especially in young girls in the underarm area. If you shave the underarm, you impair that skin barrier and you’re more likely to develop a reaction to something you’re using over it.”
Her list of recommended deodorants includes Almay Roll-On Antiperspirant & Deodorant, Crystal Body Deodorant Stick, Crystal Roll-On Body Deodorant, Vanicream Deodorant for Sensitive Skin (aluminum-free), Vanicream Antiperspirant/Deodorant, and CertainDri Clinical Strength Roll-On. They are fragrance-free and lack propylene glycol, which is a common allergen.
Increasingly, essential oils are being added to lip balms and toothpastes, said Dr. Katta, who is also author of the 2018 book “Glow: The Dermatologist’s Guide to a Whole Foods Younger Skin Diet.” She recalled one patient who presented with chronic chapped lips. “It doesn’t matter how many lip glosses I try; it just keeps getting worse,” the patient told her. The likely culprit turned out to be ingredients contained in flavored lip balm from EOS. Reports of blistering and cracking of the lips from use of the products prompted a class action lawsuit and a notice to consumers from the Food and Drug Administration.
Another patient presented with cracked lips after switching to an “all natural” toothpaste that was labeled “gluten free.”
“It looked great,” Dr. Katta recalled. “Unfortunately it was not flavoring free. She reacted to multiple essential oils, including tea tree oil, contained in the toothpaste. This is being added to a number of toothpastes, and I think we’re going to see more of these types of reactions.”
Other toothpastes contain balsam of Peru, “which is consistently one of the top allergens in patch test clinics,” she said. “One of the components of balsam of Peru is a cinnamon compound, which can be an issue.”
Dr. Katta advises her patients to use Vaseline petroleum jelly as a lip balm and recommends Tom’s of Maine Silly Strawberry Flavor (this flavor only) toothpaste for children.
A few years ago, a teenager presented to Dr. Katta with intense bullae on the dorsum of the foot after wearing shoes without socks. “She was wearing white canvas Keds, which looked very innocuous,” she said. Patch testing revealed that the teen reacted to four different rubber accelerators. “When we contacted the company, they [acknowledged] using rubber cement to make the canvas Keds,” Dr. Katta said. “Rubber cement is an adhesive and it does contain rubber accelerators. Later, I saw two cases of children who had walked around all day at the amusement park wearing their Sperry Topsiders without any socks. We couldn’t get any information from that manufacturer, but I suspect that they also use a rubber-based glue to make those shoes.” She characterized shoes as “a real setup for a foot allergy because you have friction, sweat that’s pulling allergen out of an object, and sweat is carrying it over, especially to the dorsum of the foot.”
Dr. Katta has also noticed an uptick in the number of young patients who develop allergic reactions to dyes used to make workout clothing. “If you ever see rashes that do not involve the axillary vault but do have peraxillary accentuation, think textile allergy,” she said. “We’re seeing a lot of reactions to disperse blue clothing dyes. When you think about textile allergy from the dyes, it tends to be the blue and black clothing. It’s more likely in the setting of synthetic fabrics because they leach out dyes more easily, and it’s more likely in the setting of sweat because sweat helps pull allergen out. I’m seeing it a lot from sports uniforms and tight black leggings and tight sports bras that people are wearing. I’m also seeing some from bathing suits and swim shirts.”
Exposure to products containing the preservative methylisothiazolinone (MI) is also on the rise. It ranks as the second most frequent allergen for which the North American Contact Dermatitis Group is seeing positive results on patch testing, with rates of 13.4%. MI can be found in many skin care products and “probably about half of school glues, fabric glues, and craft glues,” Dr. Katta said. “Stick versus liquid doesn’t make a difference.” Children and teens often use craft glues, laundry detergents, and other products to create “slime” as a way to learn about viscosity, polymers, and chemical reactions. “Sometimes these children have sensitive skin, or they’re using it with prolonged contact, so they may be sensitizing themselves to the MI,” she said.
She concluded her remarks by noting that an increasing number of young patients are developing reactions to wearable medical devices such as insulin pumps and glucose monitors. “With this, the first thing to think about is frictional irritant dermatitis,” she said. “You can put Scanpor medical paper tape on people’s back for 48 hours straight to patch test them. Some people are incredibly reactive to the friction of just that tape. You also have to think about trapped allergen. One of my patients reacted to colophony, fragrance mix, and propylene glycol, all of which were contained in his skin care products. Some people are getting advice from other patients to use Mastisol liquid adhesive to help their glucose monitors stick better. Mastisol has a high rate of cross-reactivity with balsam of Peru, so it’s a fragrance allergen. That’s the first thing you want to ask patients about: what products they’re using.”
One of her patients thought she was reacting to adhesive tape on her skin, but in fact she was reacting to two different acrylates: ethylene glycol dimethacrylate (EGDMA) and hydroxyethyl methacrylate (HEMA). “I know about these allergens because I see reactions from butterfly needles in dialysis patients,” Dr. Katta explained. “What happens is, these acrylates are glues or plastics used somewhere else on the device, and they can migrate through barriers.”
In one published case, a 9-year-old boy developed a reaction to ethyl cyanoacrylate contained in a glucose sensor adhesive (Dermatitis. 2017; 28[4]:289-91). It never touched the boy’s skin directly but was presumed to migrate through that tape. “The bottom line is that acrylates may induce contact dermatitis even through perceived barriers,” she said. “Their use anywhere in medical devices may prove problematic.”
Dr. Katta reported that she is a member of the advisory board for Vichy Laboratories.
AUSTIN – Not long ago, Rajani Katta, MD, received a text message from a friend who expressed concern about a rash that developed in the underarm of her teenage daughter.
The culprit turned out to be the lavender essential oil contained in an “all natural” deodorant that her daughter had recently switched to – a storyline that Dr. Katta encounters with increasing frequency in her role as clinical professor of dermatology at the University of Texas Health Science Center at Houston.
Dr. Katta said at the annual meeting of the Society for Pediatric Dermatology. “When you talk about a natural allergy, it is more likely to occur if your skin barrier is compromised, so I think that’s why we’re seeing it, especially in young girls in the underarm area. If you shave the underarm, you impair that skin barrier and you’re more likely to develop a reaction to something you’re using over it.”
Her list of recommended deodorants includes Almay Roll-On Antiperspirant & Deodorant, Crystal Body Deodorant Stick, Crystal Roll-On Body Deodorant, Vanicream Deodorant for Sensitive Skin (aluminum-free), Vanicream Antiperspirant/Deodorant, and CertainDri Clinical Strength Roll-On. They are fragrance-free and lack propylene glycol, which is a common allergen.
Increasingly, essential oils are being added to lip balms and toothpastes, said Dr. Katta, who is also author of the 2018 book “Glow: The Dermatologist’s Guide to a Whole Foods Younger Skin Diet.” She recalled one patient who presented with chronic chapped lips. “It doesn’t matter how many lip glosses I try; it just keeps getting worse,” the patient told her. The likely culprit turned out to be ingredients contained in flavored lip balm from EOS. Reports of blistering and cracking of the lips from use of the products prompted a class action lawsuit and a notice to consumers from the Food and Drug Administration.
Another patient presented with cracked lips after switching to an “all natural” toothpaste that was labeled “gluten free.”
“It looked great,” Dr. Katta recalled. “Unfortunately it was not flavoring free. She reacted to multiple essential oils, including tea tree oil, contained in the toothpaste. This is being added to a number of toothpastes, and I think we’re going to see more of these types of reactions.”
Other toothpastes contain balsam of Peru, “which is consistently one of the top allergens in patch test clinics,” she said. “One of the components of balsam of Peru is a cinnamon compound, which can be an issue.”
Dr. Katta advises her patients to use Vaseline petroleum jelly as a lip balm and recommends Tom’s of Maine Silly Strawberry Flavor (this flavor only) toothpaste for children.
A few years ago, a teenager presented to Dr. Katta with intense bullae on the dorsum of the foot after wearing shoes without socks. “She was wearing white canvas Keds, which looked very innocuous,” she said. Patch testing revealed that the teen reacted to four different rubber accelerators. “When we contacted the company, they [acknowledged] using rubber cement to make the canvas Keds,” Dr. Katta said. “Rubber cement is an adhesive and it does contain rubber accelerators. Later, I saw two cases of children who had walked around all day at the amusement park wearing their Sperry Topsiders without any socks. We couldn’t get any information from that manufacturer, but I suspect that they also use a rubber-based glue to make those shoes.” She characterized shoes as “a real setup for a foot allergy because you have friction, sweat that’s pulling allergen out of an object, and sweat is carrying it over, especially to the dorsum of the foot.”
Dr. Katta has also noticed an uptick in the number of young patients who develop allergic reactions to dyes used to make workout clothing. “If you ever see rashes that do not involve the axillary vault but do have peraxillary accentuation, think textile allergy,” she said. “We’re seeing a lot of reactions to disperse blue clothing dyes. When you think about textile allergy from the dyes, it tends to be the blue and black clothing. It’s more likely in the setting of synthetic fabrics because they leach out dyes more easily, and it’s more likely in the setting of sweat because sweat helps pull allergen out. I’m seeing it a lot from sports uniforms and tight black leggings and tight sports bras that people are wearing. I’m also seeing some from bathing suits and swim shirts.”
Exposure to products containing the preservative methylisothiazolinone (MI) is also on the rise. It ranks as the second most frequent allergen for which the North American Contact Dermatitis Group is seeing positive results on patch testing, with rates of 13.4%. MI can be found in many skin care products and “probably about half of school glues, fabric glues, and craft glues,” Dr. Katta said. “Stick versus liquid doesn’t make a difference.” Children and teens often use craft glues, laundry detergents, and other products to create “slime” as a way to learn about viscosity, polymers, and chemical reactions. “Sometimes these children have sensitive skin, or they’re using it with prolonged contact, so they may be sensitizing themselves to the MI,” she said.
She concluded her remarks by noting that an increasing number of young patients are developing reactions to wearable medical devices such as insulin pumps and glucose monitors. “With this, the first thing to think about is frictional irritant dermatitis,” she said. “You can put Scanpor medical paper tape on people’s back for 48 hours straight to patch test them. Some people are incredibly reactive to the friction of just that tape. You also have to think about trapped allergen. One of my patients reacted to colophony, fragrance mix, and propylene glycol, all of which were contained in his skin care products. Some people are getting advice from other patients to use Mastisol liquid adhesive to help their glucose monitors stick better. Mastisol has a high rate of cross-reactivity with balsam of Peru, so it’s a fragrance allergen. That’s the first thing you want to ask patients about: what products they’re using.”
One of her patients thought she was reacting to adhesive tape on her skin, but in fact she was reacting to two different acrylates: ethylene glycol dimethacrylate (EGDMA) and hydroxyethyl methacrylate (HEMA). “I know about these allergens because I see reactions from butterfly needles in dialysis patients,” Dr. Katta explained. “What happens is, these acrylates are glues or plastics used somewhere else on the device, and they can migrate through barriers.”
In one published case, a 9-year-old boy developed a reaction to ethyl cyanoacrylate contained in a glucose sensor adhesive (Dermatitis. 2017; 28[4]:289-91). It never touched the boy’s skin directly but was presumed to migrate through that tape. “The bottom line is that acrylates may induce contact dermatitis even through perceived barriers,” she said. “Their use anywhere in medical devices may prove problematic.”
Dr. Katta reported that she is a member of the advisory board for Vichy Laboratories.
EXPERT ANALYSIS FROM SPD 2019
Cases of pediatric invasive melanoma have declined since 2002, study finds
AUSTIN – The compared with females. The risk of death is also significantly increased in black patients, other nonwhite patients, and in cases where surgery was not performed.
Those are key findings from a study that set out to investigate the incidence of pediatric melanoma over the last 2 decades and factors influencing survival. At the annual meeting of the Society for Pediatric Dermatology, one of the study authors, Spandana Maddukuri, said that pediatric melanoma is the most common skin cancer in the pediatric population, accounting for 1-3% of all pediatric malignancies and 1%-4% of all cases of melanoma (Pediatr Surg. 2013;48[11]:2207-13).
“Nonmodifiable risk factors are similar to those in adult melanoma and include fair skin, light hair and eye color, increased number of congenital nevi, and family history of melanoma,” said Ms. Maddukuri, a third-year student at New Jersey Medical School, Newark. “Environmental risk factors are similar to those in adult melanoma and include exposure to UV radiation. About 60% of children do not meet standard ABCDE [asymmetrical, border, color, diameter, evolving] diagnosis criteria, which often leads to delayed diagnosis.”
Some of the characteristics that are more commonly found in pediatric lesions include amelanosis, bleeding, uniform color, and variable diameter (J Am Acad Dermatol. 2013; 68[6]:913-25).
Ms. Maddukuri and colleagues queried the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database for cases of malignant melanoma that were diagnosed in individuals aged younger than 20 years between 2002 and 2015. After excluding all cases of adult melanoma and all cases of in situ melanoma, they included 1,620 patients in the final analysis and divided them into five age groups: less than 1 year, 1-4 years, 5-9 years, 10-14 years, and 15-19 years. They calculated the overall incidence rate per 100,000 population of pediatric melanoma based on data from the 2000 U.S. Census. Age-, sex-, and race-specific incidence rates were also calculated. Kaplan-Meier and Cox regression analyses to investigate disease-specific survival and risk factors.
With each successive age group, the investigators observed that incidence rate was significantly higher than that of the previous age group (P less than .005). “However, the most striking increase in incidence occurs between the age group of 10-14 and 15-19,” she said. “Sex also influenced incidence rates. Males had an incidence rate of 0.396 per 100,000 population while females had an incidence rate of 0.579 per 100,000 population.”
Race also influenced incidence rates. White patients had the highest incidence rate of 0.605 per 100,000 population, while blacks had the lowest incident rate at 0.042 per 100,000 population. American Indian and Alaska Native patients had incidence rates of 0.046 per 100,000 population, while Asians and Pacific Islanders had an incidence rate of 0.127 per 100,000 population.
The researchers found that increased survival was associated with white race, female sex, treatment with surgical intervention, and age older than 5 years. No differences in survival were observed regarding the primary anatomic location or extent of disease. The hazard ratio of death from invasive melanoma was significantly increased in males (HR, 2.34), black patients (HR, 3.96), other nonwhite patients (HR, 3.64), and in cases where surgery was not performed (HR, 6.04).
“It is surprising that, although incidence is significantly higher in white patients and females, compared to black patients and males, respectively, the risk of dying from melanoma is much higher in black patients and males,” Ms. Maddukuri said in an interview at the meeting. “Overall, the dermatologic community is on the right track in screening and diagnosing pediatric melanoma, as seen by the decreased incidence over the last 2 decades. However, increased awareness regarding pediatric melanoma is still encouraged. I believe we were able to identify certain populations that need more attention in terms of screening, diagnosis, and treatment, which are patients less than 5 years old, black and other nonwhite patients, and males.”
She acknowledged certain shortcomings of the study, including a limited clinical history of the patient population because of the nature of the database. She also said that further studies are required to investigate the contributing factors to decreasing incidence and to evaluate the relationship of the favorable prognostic factors to increased survival. The researchers are currently working on correlating incidence rates with UV exposure and geographical location.
They reported having no financial disclosures.
AUSTIN – The compared with females. The risk of death is also significantly increased in black patients, other nonwhite patients, and in cases where surgery was not performed.
Those are key findings from a study that set out to investigate the incidence of pediatric melanoma over the last 2 decades and factors influencing survival. At the annual meeting of the Society for Pediatric Dermatology, one of the study authors, Spandana Maddukuri, said that pediatric melanoma is the most common skin cancer in the pediatric population, accounting for 1-3% of all pediatric malignancies and 1%-4% of all cases of melanoma (Pediatr Surg. 2013;48[11]:2207-13).
“Nonmodifiable risk factors are similar to those in adult melanoma and include fair skin, light hair and eye color, increased number of congenital nevi, and family history of melanoma,” said Ms. Maddukuri, a third-year student at New Jersey Medical School, Newark. “Environmental risk factors are similar to those in adult melanoma and include exposure to UV radiation. About 60% of children do not meet standard ABCDE [asymmetrical, border, color, diameter, evolving] diagnosis criteria, which often leads to delayed diagnosis.”
Some of the characteristics that are more commonly found in pediatric lesions include amelanosis, bleeding, uniform color, and variable diameter (J Am Acad Dermatol. 2013; 68[6]:913-25).
Ms. Maddukuri and colleagues queried the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database for cases of malignant melanoma that were diagnosed in individuals aged younger than 20 years between 2002 and 2015. After excluding all cases of adult melanoma and all cases of in situ melanoma, they included 1,620 patients in the final analysis and divided them into five age groups: less than 1 year, 1-4 years, 5-9 years, 10-14 years, and 15-19 years. They calculated the overall incidence rate per 100,000 population of pediatric melanoma based on data from the 2000 U.S. Census. Age-, sex-, and race-specific incidence rates were also calculated. Kaplan-Meier and Cox regression analyses to investigate disease-specific survival and risk factors.
With each successive age group, the investigators observed that incidence rate was significantly higher than that of the previous age group (P less than .005). “However, the most striking increase in incidence occurs between the age group of 10-14 and 15-19,” she said. “Sex also influenced incidence rates. Males had an incidence rate of 0.396 per 100,000 population while females had an incidence rate of 0.579 per 100,000 population.”
Race also influenced incidence rates. White patients had the highest incidence rate of 0.605 per 100,000 population, while blacks had the lowest incident rate at 0.042 per 100,000 population. American Indian and Alaska Native patients had incidence rates of 0.046 per 100,000 population, while Asians and Pacific Islanders had an incidence rate of 0.127 per 100,000 population.
The researchers found that increased survival was associated with white race, female sex, treatment with surgical intervention, and age older than 5 years. No differences in survival were observed regarding the primary anatomic location or extent of disease. The hazard ratio of death from invasive melanoma was significantly increased in males (HR, 2.34), black patients (HR, 3.96), other nonwhite patients (HR, 3.64), and in cases where surgery was not performed (HR, 6.04).
“It is surprising that, although incidence is significantly higher in white patients and females, compared to black patients and males, respectively, the risk of dying from melanoma is much higher in black patients and males,” Ms. Maddukuri said in an interview at the meeting. “Overall, the dermatologic community is on the right track in screening and diagnosing pediatric melanoma, as seen by the decreased incidence over the last 2 decades. However, increased awareness regarding pediatric melanoma is still encouraged. I believe we were able to identify certain populations that need more attention in terms of screening, diagnosis, and treatment, which are patients less than 5 years old, black and other nonwhite patients, and males.”
She acknowledged certain shortcomings of the study, including a limited clinical history of the patient population because of the nature of the database. She also said that further studies are required to investigate the contributing factors to decreasing incidence and to evaluate the relationship of the favorable prognostic factors to increased survival. The researchers are currently working on correlating incidence rates with UV exposure and geographical location.
They reported having no financial disclosures.
AUSTIN – The compared with females. The risk of death is also significantly increased in black patients, other nonwhite patients, and in cases where surgery was not performed.
Those are key findings from a study that set out to investigate the incidence of pediatric melanoma over the last 2 decades and factors influencing survival. At the annual meeting of the Society for Pediatric Dermatology, one of the study authors, Spandana Maddukuri, said that pediatric melanoma is the most common skin cancer in the pediatric population, accounting for 1-3% of all pediatric malignancies and 1%-4% of all cases of melanoma (Pediatr Surg. 2013;48[11]:2207-13).
“Nonmodifiable risk factors are similar to those in adult melanoma and include fair skin, light hair and eye color, increased number of congenital nevi, and family history of melanoma,” said Ms. Maddukuri, a third-year student at New Jersey Medical School, Newark. “Environmental risk factors are similar to those in adult melanoma and include exposure to UV radiation. About 60% of children do not meet standard ABCDE [asymmetrical, border, color, diameter, evolving] diagnosis criteria, which often leads to delayed diagnosis.”
Some of the characteristics that are more commonly found in pediatric lesions include amelanosis, bleeding, uniform color, and variable diameter (J Am Acad Dermatol. 2013; 68[6]:913-25).
Ms. Maddukuri and colleagues queried the National Cancer Institute Surveillance, Epidemiology, and End Results (SEER) database for cases of malignant melanoma that were diagnosed in individuals aged younger than 20 years between 2002 and 2015. After excluding all cases of adult melanoma and all cases of in situ melanoma, they included 1,620 patients in the final analysis and divided them into five age groups: less than 1 year, 1-4 years, 5-9 years, 10-14 years, and 15-19 years. They calculated the overall incidence rate per 100,000 population of pediatric melanoma based on data from the 2000 U.S. Census. Age-, sex-, and race-specific incidence rates were also calculated. Kaplan-Meier and Cox regression analyses to investigate disease-specific survival and risk factors.
With each successive age group, the investigators observed that incidence rate was significantly higher than that of the previous age group (P less than .005). “However, the most striking increase in incidence occurs between the age group of 10-14 and 15-19,” she said. “Sex also influenced incidence rates. Males had an incidence rate of 0.396 per 100,000 population while females had an incidence rate of 0.579 per 100,000 population.”
Race also influenced incidence rates. White patients had the highest incidence rate of 0.605 per 100,000 population, while blacks had the lowest incident rate at 0.042 per 100,000 population. American Indian and Alaska Native patients had incidence rates of 0.046 per 100,000 population, while Asians and Pacific Islanders had an incidence rate of 0.127 per 100,000 population.
The researchers found that increased survival was associated with white race, female sex, treatment with surgical intervention, and age older than 5 years. No differences in survival were observed regarding the primary anatomic location or extent of disease. The hazard ratio of death from invasive melanoma was significantly increased in males (HR, 2.34), black patients (HR, 3.96), other nonwhite patients (HR, 3.64), and in cases where surgery was not performed (HR, 6.04).
“It is surprising that, although incidence is significantly higher in white patients and females, compared to black patients and males, respectively, the risk of dying from melanoma is much higher in black patients and males,” Ms. Maddukuri said in an interview at the meeting. “Overall, the dermatologic community is on the right track in screening and diagnosing pediatric melanoma, as seen by the decreased incidence over the last 2 decades. However, increased awareness regarding pediatric melanoma is still encouraged. I believe we were able to identify certain populations that need more attention in terms of screening, diagnosis, and treatment, which are patients less than 5 years old, black and other nonwhite patients, and males.”
She acknowledged certain shortcomings of the study, including a limited clinical history of the patient population because of the nature of the database. She also said that further studies are required to investigate the contributing factors to decreasing incidence and to evaluate the relationship of the favorable prognostic factors to increased survival. The researchers are currently working on correlating incidence rates with UV exposure and geographical location.
They reported having no financial disclosures.
REPORTING FROM SPD 2019
Online ped-derm searches: What are folks looking for?
AUSTIN – After searching online for information about a suspected pediatric dermatologic condition, one in five parents and/or pediatric patients make dermatology appointments sooner than they normally would, results from a novel survey showed.
In an interview at the annual meeting of the Society for Pediatric Dermatology, study author Jamie P. Schlarbaum noted that about one-third of Americans use the Internet to research their condition or symptoms prior to visiting a physician, mostly through Google. “While nearly 50% of parents look up health care information online for their children, rashes were the most common search in pediatrics in 2011,” said Mr. Schlarbaum, who is a fourth-year medical student at the University of Minnesota, Minneapolis. “However, no studies have examined the characteristics and implications of these searches; our study is the first in pediatric dermatology and also adds a new dimension to concern in an online era: How these searches influence health care behaviors.”
During February 2018–February 2019, Kristen Hook, MD, a pediatric dermatologist in Minneapolis and the study’s principal investigator, and Mr. Schlarbaum administered a survey to 220 parents/guardians and pediatric patients who had appointments in pediatric dermatology at a University of Minnesota clinic. The survey consisted of questions about demographics, search tools, search terms, and health care decisions based on this information.
Of the 220 respondents, more than half (59%) did not use an online search engine/tool prior to their appointment. Compared with parents who did not use an online search tool, those who did were slightly younger (34 vs. 36 years, respectively), more likely to be college educated (68% vs. 48%), and less likely to have the patient in question be their first child (37% vs. 52%).
Google ranked as the most common search engine used by the survey respondents (92%), followed distantly by WebMD (18%). About 15% of respondents became more concerned about the pediatric skin condition after searching online, and 20% made appointments sooner because of the information they gleaned from their searches. “Online dermatology clearly has an influence on care today,” Mr. Schlarbaum said. “As we become an even more technologically advanced and dependent society, we anticipate that both of these numbers will grow.”
The researchers also found that (33%), moles (15%), and infections (11%). “The big takeaway [from this study] is to ask your parents and teenagers if they’ve looked up information online,” Mr. Schlarbaum said. “Whether it’s photos of the ‘worst cases’ or concerning differentials that might pop up, it’s worth it to take a few seconds to ask what they’re worried about and why.”
He acknowledged certain limitations of the study, including its small sample size and single-center design. The researchers reported having no financial disclosures.
AUSTIN – After searching online for information about a suspected pediatric dermatologic condition, one in five parents and/or pediatric patients make dermatology appointments sooner than they normally would, results from a novel survey showed.
In an interview at the annual meeting of the Society for Pediatric Dermatology, study author Jamie P. Schlarbaum noted that about one-third of Americans use the Internet to research their condition or symptoms prior to visiting a physician, mostly through Google. “While nearly 50% of parents look up health care information online for their children, rashes were the most common search in pediatrics in 2011,” said Mr. Schlarbaum, who is a fourth-year medical student at the University of Minnesota, Minneapolis. “However, no studies have examined the characteristics and implications of these searches; our study is the first in pediatric dermatology and also adds a new dimension to concern in an online era: How these searches influence health care behaviors.”
During February 2018–February 2019, Kristen Hook, MD, a pediatric dermatologist in Minneapolis and the study’s principal investigator, and Mr. Schlarbaum administered a survey to 220 parents/guardians and pediatric patients who had appointments in pediatric dermatology at a University of Minnesota clinic. The survey consisted of questions about demographics, search tools, search terms, and health care decisions based on this information.
Of the 220 respondents, more than half (59%) did not use an online search engine/tool prior to their appointment. Compared with parents who did not use an online search tool, those who did were slightly younger (34 vs. 36 years, respectively), more likely to be college educated (68% vs. 48%), and less likely to have the patient in question be their first child (37% vs. 52%).
Google ranked as the most common search engine used by the survey respondents (92%), followed distantly by WebMD (18%). About 15% of respondents became more concerned about the pediatric skin condition after searching online, and 20% made appointments sooner because of the information they gleaned from their searches. “Online dermatology clearly has an influence on care today,” Mr. Schlarbaum said. “As we become an even more technologically advanced and dependent society, we anticipate that both of these numbers will grow.”
The researchers also found that (33%), moles (15%), and infections (11%). “The big takeaway [from this study] is to ask your parents and teenagers if they’ve looked up information online,” Mr. Schlarbaum said. “Whether it’s photos of the ‘worst cases’ or concerning differentials that might pop up, it’s worth it to take a few seconds to ask what they’re worried about and why.”
He acknowledged certain limitations of the study, including its small sample size and single-center design. The researchers reported having no financial disclosures.
AUSTIN – After searching online for information about a suspected pediatric dermatologic condition, one in five parents and/or pediatric patients make dermatology appointments sooner than they normally would, results from a novel survey showed.
In an interview at the annual meeting of the Society for Pediatric Dermatology, study author Jamie P. Schlarbaum noted that about one-third of Americans use the Internet to research their condition or symptoms prior to visiting a physician, mostly through Google. “While nearly 50% of parents look up health care information online for their children, rashes were the most common search in pediatrics in 2011,” said Mr. Schlarbaum, who is a fourth-year medical student at the University of Minnesota, Minneapolis. “However, no studies have examined the characteristics and implications of these searches; our study is the first in pediatric dermatology and also adds a new dimension to concern in an online era: How these searches influence health care behaviors.”
During February 2018–February 2019, Kristen Hook, MD, a pediatric dermatologist in Minneapolis and the study’s principal investigator, and Mr. Schlarbaum administered a survey to 220 parents/guardians and pediatric patients who had appointments in pediatric dermatology at a University of Minnesota clinic. The survey consisted of questions about demographics, search tools, search terms, and health care decisions based on this information.
Of the 220 respondents, more than half (59%) did not use an online search engine/tool prior to their appointment. Compared with parents who did not use an online search tool, those who did were slightly younger (34 vs. 36 years, respectively), more likely to be college educated (68% vs. 48%), and less likely to have the patient in question be their first child (37% vs. 52%).
Google ranked as the most common search engine used by the survey respondents (92%), followed distantly by WebMD (18%). About 15% of respondents became more concerned about the pediatric skin condition after searching online, and 20% made appointments sooner because of the information they gleaned from their searches. “Online dermatology clearly has an influence on care today,” Mr. Schlarbaum said. “As we become an even more technologically advanced and dependent society, we anticipate that both of these numbers will grow.”
The researchers also found that (33%), moles (15%), and infections (11%). “The big takeaway [from this study] is to ask your parents and teenagers if they’ve looked up information online,” Mr. Schlarbaum said. “Whether it’s photos of the ‘worst cases’ or concerning differentials that might pop up, it’s worth it to take a few seconds to ask what they’re worried about and why.”
He acknowledged certain limitations of the study, including its small sample size and single-center design. The researchers reported having no financial disclosures.
REPORTING FROM SPD 2019
Collagen powder deemed noninferior to primary closure for punch-biopsy healing
Collagen powder may be noninferior to primary closure for healing punch biopsy–induced wounds and possibly leads to improved early cosmetic outcomes and accelerated wound maturation, according to Azam Qureshi of the University of Maryland, Baltimore, and associates.
In a small pilot study published in Journal of Drugs in Dermatology, eight volunteers (mean age, 37 years) received a 4-mm punch biopsy on each thigh. One wound was managed with primary closure, the other with daily application of collagen powder. The wounds were biopsied at 4 weeks for histopathologic analysis, and the study subjects rated pain, itch, and treatment preferences at 1, 2, 4, 6, and 12 weeks.
The size of wounds treated with collagen was reduced by 28.95% at 1 week, 55.76% at 2 weeks, and 95.94% at 4 weeks; six of the eight collagen-treated wounds were completely healed at 4 weeks. Wound size was reduced by 75.71% 1 week after the second biopsy, much faster than the initial healing. In addition to collagen, one patient required hyfrecation for hemostasis, which did not affect results; three of the eight subjects rated the collagen treatment as “annoying,” but no one rated it as “difficult,” and patients generally regarded collagen treatment as more time consuming.
The histopathologic analysis showed epidermal reepithelialization in collagen-treated wounds and wounds managed with primary closure, with more organized granulation tissue in the collagen-treated wounds. Similar pain and itch ratings were reported between wound types, and both patients and blinded dermatologists observing the study preferred the appearance of collagen-treated wounds.
“Future research elucidating the optimal duration of collagen therapy is needed, as less than 4 weeks may be sufficient. Shortened treatment courses would decrease the cost and effort required by patients. Future studies should also investigate the efficacy of collagen powder in healing larger wounds and in comparison to healing by secondary intention,” the investigators wrote.
CPN Biosciences funded the study. No authors had relevant financial disclosures.
SOURCE: Qureshi A et al. J Drug Dermatol. 2019;18(7):667-73
Collagen powder may be noninferior to primary closure for healing punch biopsy–induced wounds and possibly leads to improved early cosmetic outcomes and accelerated wound maturation, according to Azam Qureshi of the University of Maryland, Baltimore, and associates.
In a small pilot study published in Journal of Drugs in Dermatology, eight volunteers (mean age, 37 years) received a 4-mm punch biopsy on each thigh. One wound was managed with primary closure, the other with daily application of collagen powder. The wounds were biopsied at 4 weeks for histopathologic analysis, and the study subjects rated pain, itch, and treatment preferences at 1, 2, 4, 6, and 12 weeks.
The size of wounds treated with collagen was reduced by 28.95% at 1 week, 55.76% at 2 weeks, and 95.94% at 4 weeks; six of the eight collagen-treated wounds were completely healed at 4 weeks. Wound size was reduced by 75.71% 1 week after the second biopsy, much faster than the initial healing. In addition to collagen, one patient required hyfrecation for hemostasis, which did not affect results; three of the eight subjects rated the collagen treatment as “annoying,” but no one rated it as “difficult,” and patients generally regarded collagen treatment as more time consuming.
The histopathologic analysis showed epidermal reepithelialization in collagen-treated wounds and wounds managed with primary closure, with more organized granulation tissue in the collagen-treated wounds. Similar pain and itch ratings were reported between wound types, and both patients and blinded dermatologists observing the study preferred the appearance of collagen-treated wounds.
“Future research elucidating the optimal duration of collagen therapy is needed, as less than 4 weeks may be sufficient. Shortened treatment courses would decrease the cost and effort required by patients. Future studies should also investigate the efficacy of collagen powder in healing larger wounds and in comparison to healing by secondary intention,” the investigators wrote.
CPN Biosciences funded the study. No authors had relevant financial disclosures.
SOURCE: Qureshi A et al. J Drug Dermatol. 2019;18(7):667-73
Collagen powder may be noninferior to primary closure for healing punch biopsy–induced wounds and possibly leads to improved early cosmetic outcomes and accelerated wound maturation, according to Azam Qureshi of the University of Maryland, Baltimore, and associates.
In a small pilot study published in Journal of Drugs in Dermatology, eight volunteers (mean age, 37 years) received a 4-mm punch biopsy on each thigh. One wound was managed with primary closure, the other with daily application of collagen powder. The wounds were biopsied at 4 weeks for histopathologic analysis, and the study subjects rated pain, itch, and treatment preferences at 1, 2, 4, 6, and 12 weeks.
The size of wounds treated with collagen was reduced by 28.95% at 1 week, 55.76% at 2 weeks, and 95.94% at 4 weeks; six of the eight collagen-treated wounds were completely healed at 4 weeks. Wound size was reduced by 75.71% 1 week after the second biopsy, much faster than the initial healing. In addition to collagen, one patient required hyfrecation for hemostasis, which did not affect results; three of the eight subjects rated the collagen treatment as “annoying,” but no one rated it as “difficult,” and patients generally regarded collagen treatment as more time consuming.
The histopathologic analysis showed epidermal reepithelialization in collagen-treated wounds and wounds managed with primary closure, with more organized granulation tissue in the collagen-treated wounds. Similar pain and itch ratings were reported between wound types, and both patients and blinded dermatologists observing the study preferred the appearance of collagen-treated wounds.
“Future research elucidating the optimal duration of collagen therapy is needed, as less than 4 weeks may be sufficient. Shortened treatment courses would decrease the cost and effort required by patients. Future studies should also investigate the efficacy of collagen powder in healing larger wounds and in comparison to healing by secondary intention,” the investigators wrote.
CPN Biosciences funded the study. No authors had relevant financial disclosures.
SOURCE: Qureshi A et al. J Drug Dermatol. 2019;18(7):667-73
FROM JOURNAL OF DRUGS IN DERMATOLOGY
A 3-year-old is brought to the clinic for evaluation of a localized, scaling inflamed lesion on the left leg
Nummular dermatitis, or nummular eczema, is an inflammatory skin condition that is considered to be a distinctive form of idiopathic eczema, while the term also is used to describe lesional morphology associated with other conditions.
The term nummular derives from the Latin word for “coin,” as lesions are commonly annular plaques. Lesions of nummular dermatitis can be single or multiple. The typical distribution involves the extremities and, although less common, it can affect the trunk as well.
Nummular dermatitis may be associated with atopic dermatitis, or it can be an isolated condition.1 While the pathogenesis is uncertain, instigating factors include xerotic skin, insect bites, or scratches or scrapes.1Staphylococcus infection or colonization, contact allergies to metals such as nickel and less commonly mercury, sensitivity to formaldehyde or medicines such as neomycin, and sensitization to an environmental aeroallergen (such as Candida albicans, dust mites) are considered risk factors.2
The diagnosis of nummular dermatitis is clinical. Laboratory testing and/or biopsy generally are not necessary, although a bacterial culture can be considered in patients with exudative and/or crusted lesions to rule out impetigo as a primary process of secondary infection. In some cases, patch testing for allergic contact dermatitis may be useful.
The differential diagnosis of nummular dermatitis includes tinea corporis (ringworm), atopic dermatitis, allergic contact dermatitis, impetigo, and psoriasis. Tinea corporis usually presents as annular lesions with a distinct peripheral scaling, rather than the diffuse induration of nummular dermatitis. Potassium hydroxide preparation or fungal culture can identify tinea species. Nummular dermatitis may be seen in patients with atopic dermatitis, who should have typical history, morphology, and course consistent with standard diagnostic criteria. Allergic contact dermatitis can present with regional, localized eczematous plaques in areas exposed to contact allergens. Patterns of lesions in areas of contact and worsening with repeat exposures can be clues to this diagnosis. Impetigo can present with honey-colored crusted lesions and/or superficial erosions, or purulent pyoderma. Lesions can be single or multiple and generally appear less inflammatory than nummular dermatitis. Psoriasis lesions may be annular, are more common on extensor surfaces, and usually have more prominent overlying pinkish, silvery white or micaceous scale.
Management of nummular dermatitis requires strong anti-inflammatory medications, usually mid-potency or higher topical corticosteroids, along with moisturizers and limiting exposure to skin irritants. “Wet wraps,” with application of topical corticosteroids to wet skin with occlusive wet dressings can enhance response. Transition from higher strength topical corticosteroids to lower strength agents used intermittently can help achieve remission or cure. Management practices include less frequent bathing with lukewarm water, using hypoallergenic cleansers and detergents, and applying moisturizers frequently. If plaques do recur, they tend to do so in the same location and in some patients resolution may result in hyper or hypopigmentation. Refractory disease may be managed with intralesional steroid injections, or systemic medications such as methotrexate.3
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither Dr. Tracy nor Dr. Eichenfield have any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
1. Pediatr Dermatol. 2012 Sep-Oct;29(5):580-3.
2. American Academy of Dermatology. Nummular Dermatitis Overview
3. Pediatr Dermatol. 2018 Sep;35(5):611-5.
Nummular dermatitis, or nummular eczema, is an inflammatory skin condition that is considered to be a distinctive form of idiopathic eczema, while the term also is used to describe lesional morphology associated with other conditions.
The term nummular derives from the Latin word for “coin,” as lesions are commonly annular plaques. Lesions of nummular dermatitis can be single or multiple. The typical distribution involves the extremities and, although less common, it can affect the trunk as well.
Nummular dermatitis may be associated with atopic dermatitis, or it can be an isolated condition.1 While the pathogenesis is uncertain, instigating factors include xerotic skin, insect bites, or scratches or scrapes.1Staphylococcus infection or colonization, contact allergies to metals such as nickel and less commonly mercury, sensitivity to formaldehyde or medicines such as neomycin, and sensitization to an environmental aeroallergen (such as Candida albicans, dust mites) are considered risk factors.2
The diagnosis of nummular dermatitis is clinical. Laboratory testing and/or biopsy generally are not necessary, although a bacterial culture can be considered in patients with exudative and/or crusted lesions to rule out impetigo as a primary process of secondary infection. In some cases, patch testing for allergic contact dermatitis may be useful.
The differential diagnosis of nummular dermatitis includes tinea corporis (ringworm), atopic dermatitis, allergic contact dermatitis, impetigo, and psoriasis. Tinea corporis usually presents as annular lesions with a distinct peripheral scaling, rather than the diffuse induration of nummular dermatitis. Potassium hydroxide preparation or fungal culture can identify tinea species. Nummular dermatitis may be seen in patients with atopic dermatitis, who should have typical history, morphology, and course consistent with standard diagnostic criteria. Allergic contact dermatitis can present with regional, localized eczematous plaques in areas exposed to contact allergens. Patterns of lesions in areas of contact and worsening with repeat exposures can be clues to this diagnosis. Impetigo can present with honey-colored crusted lesions and/or superficial erosions, or purulent pyoderma. Lesions can be single or multiple and generally appear less inflammatory than nummular dermatitis. Psoriasis lesions may be annular, are more common on extensor surfaces, and usually have more prominent overlying pinkish, silvery white or micaceous scale.
Management of nummular dermatitis requires strong anti-inflammatory medications, usually mid-potency or higher topical corticosteroids, along with moisturizers and limiting exposure to skin irritants. “Wet wraps,” with application of topical corticosteroids to wet skin with occlusive wet dressings can enhance response. Transition from higher strength topical corticosteroids to lower strength agents used intermittently can help achieve remission or cure. Management practices include less frequent bathing with lukewarm water, using hypoallergenic cleansers and detergents, and applying moisturizers frequently. If plaques do recur, they tend to do so in the same location and in some patients resolution may result in hyper or hypopigmentation. Refractory disease may be managed with intralesional steroid injections, or systemic medications such as methotrexate.3
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither Dr. Tracy nor Dr. Eichenfield have any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
1. Pediatr Dermatol. 2012 Sep-Oct;29(5):580-3.
2. American Academy of Dermatology. Nummular Dermatitis Overview
3. Pediatr Dermatol. 2018 Sep;35(5):611-5.
Nummular dermatitis, or nummular eczema, is an inflammatory skin condition that is considered to be a distinctive form of idiopathic eczema, while the term also is used to describe lesional morphology associated with other conditions.
The term nummular derives from the Latin word for “coin,” as lesions are commonly annular plaques. Lesions of nummular dermatitis can be single or multiple. The typical distribution involves the extremities and, although less common, it can affect the trunk as well.
Nummular dermatitis may be associated with atopic dermatitis, or it can be an isolated condition.1 While the pathogenesis is uncertain, instigating factors include xerotic skin, insect bites, or scratches or scrapes.1Staphylococcus infection or colonization, contact allergies to metals such as nickel and less commonly mercury, sensitivity to formaldehyde or medicines such as neomycin, and sensitization to an environmental aeroallergen (such as Candida albicans, dust mites) are considered risk factors.2
The diagnosis of nummular dermatitis is clinical. Laboratory testing and/or biopsy generally are not necessary, although a bacterial culture can be considered in patients with exudative and/or crusted lesions to rule out impetigo as a primary process of secondary infection. In some cases, patch testing for allergic contact dermatitis may be useful.
The differential diagnosis of nummular dermatitis includes tinea corporis (ringworm), atopic dermatitis, allergic contact dermatitis, impetigo, and psoriasis. Tinea corporis usually presents as annular lesions with a distinct peripheral scaling, rather than the diffuse induration of nummular dermatitis. Potassium hydroxide preparation or fungal culture can identify tinea species. Nummular dermatitis may be seen in patients with atopic dermatitis, who should have typical history, morphology, and course consistent with standard diagnostic criteria. Allergic contact dermatitis can present with regional, localized eczematous plaques in areas exposed to contact allergens. Patterns of lesions in areas of contact and worsening with repeat exposures can be clues to this diagnosis. Impetigo can present with honey-colored crusted lesions and/or superficial erosions, or purulent pyoderma. Lesions can be single or multiple and generally appear less inflammatory than nummular dermatitis. Psoriasis lesions may be annular, are more common on extensor surfaces, and usually have more prominent overlying pinkish, silvery white or micaceous scale.
Management of nummular dermatitis requires strong anti-inflammatory medications, usually mid-potency or higher topical corticosteroids, along with moisturizers and limiting exposure to skin irritants. “Wet wraps,” with application of topical corticosteroids to wet skin with occlusive wet dressings can enhance response. Transition from higher strength topical corticosteroids to lower strength agents used intermittently can help achieve remission or cure. Management practices include less frequent bathing with lukewarm water, using hypoallergenic cleansers and detergents, and applying moisturizers frequently. If plaques do recur, they tend to do so in the same location and in some patients resolution may result in hyper or hypopigmentation. Refractory disease may be managed with intralesional steroid injections, or systemic medications such as methotrexate.3
Dr. Tracy is a research fellow in pediatric dermatology at Rady Children’s Hospital–San Diego and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Neither Dr. Tracy nor Dr. Eichenfield have any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
1. Pediatr Dermatol. 2012 Sep-Oct;29(5):580-3.
2. American Academy of Dermatology. Nummular Dermatitis Overview
3. Pediatr Dermatol. 2018 Sep;35(5):611-5.
On physical exam, he is noted to have a localized eczematous plaque with erythema and edema. Also, he is noted to have diffuse, fine xerosis of the bilateral lower extremities. His skin is otherwise nonremarkable.
iPledge: Fetal exposure to isotretinoin continues
but pregnancy, abortions, and fetal defects associated with isotretinoin exposure are still occurring in women of reproductive age, according to a retrospective study published in JAMA Dermatology.
In 2006, the Food and Drug Administration implemented the iPledge program, with requirements that include women of childbearing age having a negative pregnancy test and evidence of using two forms of contraception monthly to use isotretinoin, a teratogen. “Although the number of pregnancy-related adverse events for patients taking isotretinoin has decreased since 2006, pregnancies, abortions, and fetal defects associated with isotretinoin exposure continue to be a problem,” Elizabeth Tkachenko, BS, from the University of Massachusetts Medical School, Worcester, and coauthors concluded. “Further research is required to determine the most efficacious system to reduce complications for patients and administrative requirements for physicians while at the same time maintaining access to this important drug.” (iPledge followed other Risk Evaluation and Mitigation Strategy systems for isotretinoin.)
She and her colleagues performed a retrospective evaluation of pregnancy-related adverse events related to isotretinoin that had occurred between January 1997 and December 2017 using the FDA Adverse Event Reporting System (FAERS), which receives reports from prescribers, consumers, and pharmaceutical manufacturers. While there could be many different classification terms for each individual, any number of adverse events reported by an individual was counted as one pregnancy. Ms. Tkachenko and colleagues classified abortions, pregnancies during contraception use, and pregnancy-related defects into separate subgroups for analysis.
From 1997 to 2017, there were 6,740 pregnancies among women (mean age, 24.6 years) during treatment with isotretinoin reported to FAERS, with 7 reports in 1997, and a peak of 768 pregnancies in 2006. Almost 70% (4,647) of the pregnancies were reported after iPledge was introduced. Between 2011 and 2017, there were 218-310 pregnancy reports each year.
Of the total number of pregnancy reports during the study period, 1,896 were abortions (28.1% of the total); 10.9% of the total number of pregnancy reports were spontaneous abortions (733). The number of abortions peaked in 2008, with 291 reports, of which 85% were therapeutic abortions. Also peaking in 2008 was the number of reports of pregnancies while taking a contraceptive (64). After 2008, pregnancies and abortions dropped.
Fetal defects peaked in 2000, with 34 cases reported, and dropped to four or fewer reports annually after 2008.
“Our findings demonstrate that reports of pregnancy among women taking isotretinoin are concentrated among those aged 20 to 29 years, peaked in 2006, and have been consistent since 2011,” the authors wrote.
Limitations of the study, they noted, include limitations of FAERS data and possible reporting fatigue among doctors and patients. The total number of isotretinoin courses prescribed to this patient population is also unknown, which affected their ability to determine the true rate of pregnancy-related adverse events, they noted.
The other authors for this study were from Harvard Medical School and the departments of dermatology at Brigham and Women’s Hospital, both in Boston, as well as the University of Pennsylvania, Philadelphia. One author reported support from an award by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health and salary support from a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the trustees of the University of Pennsylvania. The other authors reported no relevant conflicts of interest.
SOURCE: Tkachenko E et al. JAMA Dermatol. 2019. doi: 10.1001/jamadermatol.2019.1388.
The rate of fetal exposure to isotretinoin has generally decreased since the implementation of the iPledge program, but rates have plateaued since 2011, and it is unclear why the exposure rate does not continue to decrease, Arielle R. Nagler, MD, wrote in a related editorial.
As noted by Tkachenko et al., it is not possible to infer that iPledge resulted in declines in fetal exposure, abortions, and pregnancy-related complications. Use of long-acting reversible contraception, education about contraception use, and reporting fatigue could be factors in the decline, Dr. Nagler noted. “The inability to clearly demonstrate causality, combined with the unexplained delay and plateau in the number of fetal exposures to isotretinoin after the implementation of iPledge, makes it difficult to draw firm conclusions about the role of iPledge in this reported trend,” she said.
The decrease in fetal exposure could also potentially be explained by effects of iPledge on the availability of isotretinoin for women of childbearing age. Indeed, studies have shown a significant decrease in isotretinoin prescriptions in this patient population after iPledge was implemented.
Despite lack of data, there is still too much fetal exposure to isotretinoin, wrote Dr. Nagler, which calls into question the efficacy of the iPledge program. “We can all agree that 1 fetal exposure to isotretinoin should be too many, but without taking isotretinoin off the market, we will never achieve zero fetal exposures to isotretinoin. Still, we can – and should – expect more from a REMS [Risk Evaluation and Mitigation Strategy] program,” Dr. Nagler concluded.
Dr. Nagler is with the department of dermatology at New York University. She reported no relevant conflicts of interest.
The rate of fetal exposure to isotretinoin has generally decreased since the implementation of the iPledge program, but rates have plateaued since 2011, and it is unclear why the exposure rate does not continue to decrease, Arielle R. Nagler, MD, wrote in a related editorial.
As noted by Tkachenko et al., it is not possible to infer that iPledge resulted in declines in fetal exposure, abortions, and pregnancy-related complications. Use of long-acting reversible contraception, education about contraception use, and reporting fatigue could be factors in the decline, Dr. Nagler noted. “The inability to clearly demonstrate causality, combined with the unexplained delay and plateau in the number of fetal exposures to isotretinoin after the implementation of iPledge, makes it difficult to draw firm conclusions about the role of iPledge in this reported trend,” she said.
The decrease in fetal exposure could also potentially be explained by effects of iPledge on the availability of isotretinoin for women of childbearing age. Indeed, studies have shown a significant decrease in isotretinoin prescriptions in this patient population after iPledge was implemented.
Despite lack of data, there is still too much fetal exposure to isotretinoin, wrote Dr. Nagler, which calls into question the efficacy of the iPledge program. “We can all agree that 1 fetal exposure to isotretinoin should be too many, but without taking isotretinoin off the market, we will never achieve zero fetal exposures to isotretinoin. Still, we can – and should – expect more from a REMS [Risk Evaluation and Mitigation Strategy] program,” Dr. Nagler concluded.
Dr. Nagler is with the department of dermatology at New York University. She reported no relevant conflicts of interest.
The rate of fetal exposure to isotretinoin has generally decreased since the implementation of the iPledge program, but rates have plateaued since 2011, and it is unclear why the exposure rate does not continue to decrease, Arielle R. Nagler, MD, wrote in a related editorial.
As noted by Tkachenko et al., it is not possible to infer that iPledge resulted in declines in fetal exposure, abortions, and pregnancy-related complications. Use of long-acting reversible contraception, education about contraception use, and reporting fatigue could be factors in the decline, Dr. Nagler noted. “The inability to clearly demonstrate causality, combined with the unexplained delay and plateau in the number of fetal exposures to isotretinoin after the implementation of iPledge, makes it difficult to draw firm conclusions about the role of iPledge in this reported trend,” she said.
The decrease in fetal exposure could also potentially be explained by effects of iPledge on the availability of isotretinoin for women of childbearing age. Indeed, studies have shown a significant decrease in isotretinoin prescriptions in this patient population after iPledge was implemented.
Despite lack of data, there is still too much fetal exposure to isotretinoin, wrote Dr. Nagler, which calls into question the efficacy of the iPledge program. “We can all agree that 1 fetal exposure to isotretinoin should be too many, but without taking isotretinoin off the market, we will never achieve zero fetal exposures to isotretinoin. Still, we can – and should – expect more from a REMS [Risk Evaluation and Mitigation Strategy] program,” Dr. Nagler concluded.
Dr. Nagler is with the department of dermatology at New York University. She reported no relevant conflicts of interest.
but pregnancy, abortions, and fetal defects associated with isotretinoin exposure are still occurring in women of reproductive age, according to a retrospective study published in JAMA Dermatology.
In 2006, the Food and Drug Administration implemented the iPledge program, with requirements that include women of childbearing age having a negative pregnancy test and evidence of using two forms of contraception monthly to use isotretinoin, a teratogen. “Although the number of pregnancy-related adverse events for patients taking isotretinoin has decreased since 2006, pregnancies, abortions, and fetal defects associated with isotretinoin exposure continue to be a problem,” Elizabeth Tkachenko, BS, from the University of Massachusetts Medical School, Worcester, and coauthors concluded. “Further research is required to determine the most efficacious system to reduce complications for patients and administrative requirements for physicians while at the same time maintaining access to this important drug.” (iPledge followed other Risk Evaluation and Mitigation Strategy systems for isotretinoin.)
She and her colleagues performed a retrospective evaluation of pregnancy-related adverse events related to isotretinoin that had occurred between January 1997 and December 2017 using the FDA Adverse Event Reporting System (FAERS), which receives reports from prescribers, consumers, and pharmaceutical manufacturers. While there could be many different classification terms for each individual, any number of adverse events reported by an individual was counted as one pregnancy. Ms. Tkachenko and colleagues classified abortions, pregnancies during contraception use, and pregnancy-related defects into separate subgroups for analysis.
From 1997 to 2017, there were 6,740 pregnancies among women (mean age, 24.6 years) during treatment with isotretinoin reported to FAERS, with 7 reports in 1997, and a peak of 768 pregnancies in 2006. Almost 70% (4,647) of the pregnancies were reported after iPledge was introduced. Between 2011 and 2017, there were 218-310 pregnancy reports each year.
Of the total number of pregnancy reports during the study period, 1,896 were abortions (28.1% of the total); 10.9% of the total number of pregnancy reports were spontaneous abortions (733). The number of abortions peaked in 2008, with 291 reports, of which 85% were therapeutic abortions. Also peaking in 2008 was the number of reports of pregnancies while taking a contraceptive (64). After 2008, pregnancies and abortions dropped.
Fetal defects peaked in 2000, with 34 cases reported, and dropped to four or fewer reports annually after 2008.
“Our findings demonstrate that reports of pregnancy among women taking isotretinoin are concentrated among those aged 20 to 29 years, peaked in 2006, and have been consistent since 2011,” the authors wrote.
Limitations of the study, they noted, include limitations of FAERS data and possible reporting fatigue among doctors and patients. The total number of isotretinoin courses prescribed to this patient population is also unknown, which affected their ability to determine the true rate of pregnancy-related adverse events, they noted.
The other authors for this study were from Harvard Medical School and the departments of dermatology at Brigham and Women’s Hospital, both in Boston, as well as the University of Pennsylvania, Philadelphia. One author reported support from an award by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health and salary support from a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the trustees of the University of Pennsylvania. The other authors reported no relevant conflicts of interest.
SOURCE: Tkachenko E et al. JAMA Dermatol. 2019. doi: 10.1001/jamadermatol.2019.1388.
but pregnancy, abortions, and fetal defects associated with isotretinoin exposure are still occurring in women of reproductive age, according to a retrospective study published in JAMA Dermatology.
In 2006, the Food and Drug Administration implemented the iPledge program, with requirements that include women of childbearing age having a negative pregnancy test and evidence of using two forms of contraception monthly to use isotretinoin, a teratogen. “Although the number of pregnancy-related adverse events for patients taking isotretinoin has decreased since 2006, pregnancies, abortions, and fetal defects associated with isotretinoin exposure continue to be a problem,” Elizabeth Tkachenko, BS, from the University of Massachusetts Medical School, Worcester, and coauthors concluded. “Further research is required to determine the most efficacious system to reduce complications for patients and administrative requirements for physicians while at the same time maintaining access to this important drug.” (iPledge followed other Risk Evaluation and Mitigation Strategy systems for isotretinoin.)
She and her colleagues performed a retrospective evaluation of pregnancy-related adverse events related to isotretinoin that had occurred between January 1997 and December 2017 using the FDA Adverse Event Reporting System (FAERS), which receives reports from prescribers, consumers, and pharmaceutical manufacturers. While there could be many different classification terms for each individual, any number of adverse events reported by an individual was counted as one pregnancy. Ms. Tkachenko and colleagues classified abortions, pregnancies during contraception use, and pregnancy-related defects into separate subgroups for analysis.
From 1997 to 2017, there were 6,740 pregnancies among women (mean age, 24.6 years) during treatment with isotretinoin reported to FAERS, with 7 reports in 1997, and a peak of 768 pregnancies in 2006. Almost 70% (4,647) of the pregnancies were reported after iPledge was introduced. Between 2011 and 2017, there were 218-310 pregnancy reports each year.
Of the total number of pregnancy reports during the study period, 1,896 were abortions (28.1% of the total); 10.9% of the total number of pregnancy reports were spontaneous abortions (733). The number of abortions peaked in 2008, with 291 reports, of which 85% were therapeutic abortions. Also peaking in 2008 was the number of reports of pregnancies while taking a contraceptive (64). After 2008, pregnancies and abortions dropped.
Fetal defects peaked in 2000, with 34 cases reported, and dropped to four or fewer reports annually after 2008.
“Our findings demonstrate that reports of pregnancy among women taking isotretinoin are concentrated among those aged 20 to 29 years, peaked in 2006, and have been consistent since 2011,” the authors wrote.
Limitations of the study, they noted, include limitations of FAERS data and possible reporting fatigue among doctors and patients. The total number of isotretinoin courses prescribed to this patient population is also unknown, which affected their ability to determine the true rate of pregnancy-related adverse events, they noted.
The other authors for this study were from Harvard Medical School and the departments of dermatology at Brigham and Women’s Hospital, both in Boston, as well as the University of Pennsylvania, Philadelphia. One author reported support from an award by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health and salary support from a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the trustees of the University of Pennsylvania. The other authors reported no relevant conflicts of interest.
SOURCE: Tkachenko E et al. JAMA Dermatol. 2019. doi: 10.1001/jamadermatol.2019.1388.
FROM JAMA DERMATOLOGY