User login
When Life’s an Itch
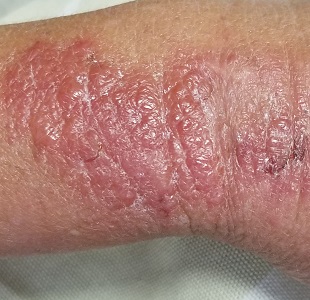
A 34-year-old woman self-refers to dermatology for evaluation of a very itchy rash that manifested 2 weeks ago on her right arm. She immediately went to an urgent care clinic, where she was diagnosed with shingles and prescribed valacyclovir. This diagnosis was upsetting to the patient, as she was advised to avoid contact with her newborn niece for at least 2 weeks.
Despite the prescribed medication, however, the rash began to pop up in other areas, including her left arm, chest, and face. Through all of this, the patient felt fine: no fever, myalgia, or malaise.
Her husband suggested she seek an appointment with dermatology, which was expedited by a phone call from her primary care provider.
EXAMINATION
The patient is afebrile and in no acute distress. She is, however, quite upset with the widespread collections of vesicles on mildly erythematous bases, many in a linear configuration. In several areas, there is ecchymosis secondary to scratching.
What’s the diagnosis?
DISCUSSION
Poison ivy, or Rhus dermatitis, is one of the most common dermatologic problems seen in medicine—and yet, its various presentations can, as this case illustrates, be quite confusing. Even when it is recognized, treatment is far from satisfactory (but more on that later). Furthermore, there is a lot of misinformation about everything from the appearance of the offending plant to the condition’s “contagious” nature.
From a broader perspective, poison ivy is becoming more prevalent and its effects more pronounced as cities expand into formerly open country. The Rhus plant family (Toxicodendron radicans and others) thrives on our increasing levels of CO2, effectively making the “poisonous” resin in the stems, leaves, and berries more potent.
With repeated exposure, the vast majority of the population will develop an allergy to this resin, known as urushiol, which can persist even on long-dead plants, vines, and leaves. (It does take repeated exposure to develop the requisite T-cell population, which is why many children are immune to it.) The urushiol does not serve as a protective substance for the plant; rather, it helps the plant retain water. In fact, many animals feed on the plant with impunity.
Virtually all members of the poison ivy family display “leaves of three” emerging from a single stem, with each triplet alternating first on one side of the branch and then on the other. Several varieties of the plant flourish over vast areas of the world, but in the United States, east of the Rockies, Toxicodendron radicans is the dominant member of the family. It can grow as a low vine, a shrub, or a climbing vine, each with a distinct appearance aside from the leaves, which are almond-shaped, smooth, and usually shiny with smooth surfaces. Most mature leaves will have a single notch, sometimes called a “thumb,” on otherwise smooth, nonserrated edges. In the summer, tiny white and yellow berries begin to grow.
The climbing vines of older plants can reach heights of 10 meters or more. These vines can reach a thickness of 3 inches and often appear “furry,” with tiny rootlets covering their surfaces. Plants this large can produce leaves 12 to 14 inches long.
Clinically, the appearance of linear pink to red pruritic vesicular streaks typify this contact dermatitis, which can immediately follow exposure or take days to appear. Said exposure can be direct or via pets, tools, or aerosols (eg, from neighbors mowing their lawns). Besides avoidance of the great outdoors, washing thoroughly immediately after exposure makes sense (but many are unaware that they’ve been exposed until it’s too late).
Poison ivy is not contagious, though the general public firmly believes otherwise. Left untreated, it clears within 2 weeks (except in unusual cases). For those who cannot bear to wait, treatment is problematic, to say the least. OTC products, such as calamine lotion, do nothing for the itching but may help with blistering. Topical or systemic steroids reduce itching somewhat. Antihistamines are useless, since this condition does not involve histamine release.
TAKE-HOME LEARNING POINTS
- Poison ivy (Rhus dermatitis) is quite common and becoming more so due to encroaching civilization and increasing CO2 levels.
- “Leaves of three, let it be” is still good advice, because the poison ivy plant Toxicodendron radicans manifests with three almond-shaped, shiny, green leaves grouped in threes.
- Urushiol is the name of the oily resin found in the plant’s stem, leaves, and berries, and is the trigger resulting in contact dermatitis.
- Poison ivy is not contagious and typically clears in 2 weeks.
A 34-year-old woman self-refers to dermatology for evaluation of a very itchy rash that manifested 2 weeks ago on her right arm. She immediately went to an urgent care clinic, where she was diagnosed with shingles and prescribed valacyclovir. This diagnosis was upsetting to the patient, as she was advised to avoid contact with her newborn niece for at least 2 weeks.
Despite the prescribed medication, however, the rash began to pop up in other areas, including her left arm, chest, and face. Through all of this, the patient felt fine: no fever, myalgia, or malaise.
Her husband suggested she seek an appointment with dermatology, which was expedited by a phone call from her primary care provider.

EXAMINATION
The patient is afebrile and in no acute distress. She is, however, quite upset with the widespread collections of vesicles on mildly erythematous bases, many in a linear configuration. In several areas, there is ecchymosis secondary to scratching.
What’s the diagnosis?
DISCUSSION
Poison ivy, or Rhus dermatitis, is one of the most common dermatologic problems seen in medicine—and yet, its various presentations can, as this case illustrates, be quite confusing. Even when it is recognized, treatment is far from satisfactory (but more on that later). Furthermore, there is a lot of misinformation about everything from the appearance of the offending plant to the condition’s “contagious” nature.
From a broader perspective, poison ivy is becoming more prevalent and its effects more pronounced as cities expand into formerly open country. The Rhus plant family (Toxicodendron radicans and others) thrives on our increasing levels of CO2, effectively making the “poisonous” resin in the stems, leaves, and berries more potent.
With repeated exposure, the vast majority of the population will develop an allergy to this resin, known as urushiol, which can persist even on long-dead plants, vines, and leaves. (It does take repeated exposure to develop the requisite T-cell population, which is why many children are immune to it.) The urushiol does not serve as a protective substance for the plant; rather, it helps the plant retain water. In fact, many animals feed on the plant with impunity.
Virtually all members of the poison ivy family display “leaves of three” emerging from a single stem, with each triplet alternating first on one side of the branch and then on the other. Several varieties of the plant flourish over vast areas of the world, but in the United States, east of the Rockies, Toxicodendron radicans is the dominant member of the family. It can grow as a low vine, a shrub, or a climbing vine, each with a distinct appearance aside from the leaves, which are almond-shaped, smooth, and usually shiny with smooth surfaces. Most mature leaves will have a single notch, sometimes called a “thumb,” on otherwise smooth, nonserrated edges. In the summer, tiny white and yellow berries begin to grow.
The climbing vines of older plants can reach heights of 10 meters or more. These vines can reach a thickness of 3 inches and often appear “furry,” with tiny rootlets covering their surfaces. Plants this large can produce leaves 12 to 14 inches long.
Clinically, the appearance of linear pink to red pruritic vesicular streaks typify this contact dermatitis, which can immediately follow exposure or take days to appear. Said exposure can be direct or via pets, tools, or aerosols (eg, from neighbors mowing their lawns). Besides avoidance of the great outdoors, washing thoroughly immediately after exposure makes sense (but many are unaware that they’ve been exposed until it’s too late).
Poison ivy is not contagious, though the general public firmly believes otherwise. Left untreated, it clears within 2 weeks (except in unusual cases). For those who cannot bear to wait, treatment is problematic, to say the least. OTC products, such as calamine lotion, do nothing for the itching but may help with blistering. Topical or systemic steroids reduce itching somewhat. Antihistamines are useless, since this condition does not involve histamine release.
TAKE-HOME LEARNING POINTS
- Poison ivy (Rhus dermatitis) is quite common and becoming more so due to encroaching civilization and increasing CO2 levels.
- “Leaves of three, let it be” is still good advice, because the poison ivy plant Toxicodendron radicans manifests with three almond-shaped, shiny, green leaves grouped in threes.
- Urushiol is the name of the oily resin found in the plant’s stem, leaves, and berries, and is the trigger resulting in contact dermatitis.
- Poison ivy is not contagious and typically clears in 2 weeks.
A 34-year-old woman self-refers to dermatology for evaluation of a very itchy rash that manifested 2 weeks ago on her right arm. She immediately went to an urgent care clinic, where she was diagnosed with shingles and prescribed valacyclovir. This diagnosis was upsetting to the patient, as she was advised to avoid contact with her newborn niece for at least 2 weeks.
Despite the prescribed medication, however, the rash began to pop up in other areas, including her left arm, chest, and face. Through all of this, the patient felt fine: no fever, myalgia, or malaise.
Her husband suggested she seek an appointment with dermatology, which was expedited by a phone call from her primary care provider.

EXAMINATION
The patient is afebrile and in no acute distress. She is, however, quite upset with the widespread collections of vesicles on mildly erythematous bases, many in a linear configuration. In several areas, there is ecchymosis secondary to scratching.
What’s the diagnosis?
DISCUSSION
Poison ivy, or Rhus dermatitis, is one of the most common dermatologic problems seen in medicine—and yet, its various presentations can, as this case illustrates, be quite confusing. Even when it is recognized, treatment is far from satisfactory (but more on that later). Furthermore, there is a lot of misinformation about everything from the appearance of the offending plant to the condition’s “contagious” nature.
From a broader perspective, poison ivy is becoming more prevalent and its effects more pronounced as cities expand into formerly open country. The Rhus plant family (Toxicodendron radicans and others) thrives on our increasing levels of CO2, effectively making the “poisonous” resin in the stems, leaves, and berries more potent.
With repeated exposure, the vast majority of the population will develop an allergy to this resin, known as urushiol, which can persist even on long-dead plants, vines, and leaves. (It does take repeated exposure to develop the requisite T-cell population, which is why many children are immune to it.) The urushiol does not serve as a protective substance for the plant; rather, it helps the plant retain water. In fact, many animals feed on the plant with impunity.
Virtually all members of the poison ivy family display “leaves of three” emerging from a single stem, with each triplet alternating first on one side of the branch and then on the other. Several varieties of the plant flourish over vast areas of the world, but in the United States, east of the Rockies, Toxicodendron radicans is the dominant member of the family. It can grow as a low vine, a shrub, or a climbing vine, each with a distinct appearance aside from the leaves, which are almond-shaped, smooth, and usually shiny with smooth surfaces. Most mature leaves will have a single notch, sometimes called a “thumb,” on otherwise smooth, nonserrated edges. In the summer, tiny white and yellow berries begin to grow.
The climbing vines of older plants can reach heights of 10 meters or more. These vines can reach a thickness of 3 inches and often appear “furry,” with tiny rootlets covering their surfaces. Plants this large can produce leaves 12 to 14 inches long.
Clinically, the appearance of linear pink to red pruritic vesicular streaks typify this contact dermatitis, which can immediately follow exposure or take days to appear. Said exposure can be direct or via pets, tools, or aerosols (eg, from neighbors mowing their lawns). Besides avoidance of the great outdoors, washing thoroughly immediately after exposure makes sense (but many are unaware that they’ve been exposed until it’s too late).
Poison ivy is not contagious, though the general public firmly believes otherwise. Left untreated, it clears within 2 weeks (except in unusual cases). For those who cannot bear to wait, treatment is problematic, to say the least. OTC products, such as calamine lotion, do nothing for the itching but may help with blistering. Topical or systemic steroids reduce itching somewhat. Antihistamines are useless, since this condition does not involve histamine release.
TAKE-HOME LEARNING POINTS
- Poison ivy (Rhus dermatitis) is quite common and becoming more so due to encroaching civilization and increasing CO2 levels.
- “Leaves of three, let it be” is still good advice, because the poison ivy plant Toxicodendron radicans manifests with three almond-shaped, shiny, green leaves grouped in threes.
- Urushiol is the name of the oily resin found in the plant’s stem, leaves, and berries, and is the trigger resulting in contact dermatitis.
- Poison ivy is not contagious and typically clears in 2 weeks.
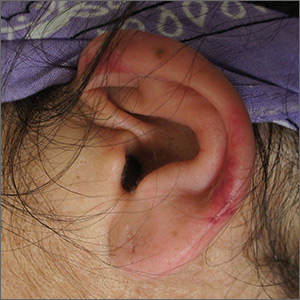
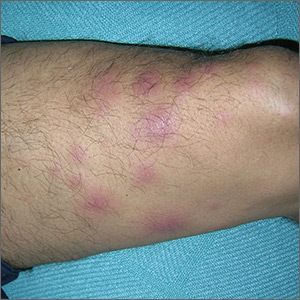
Left ear pain
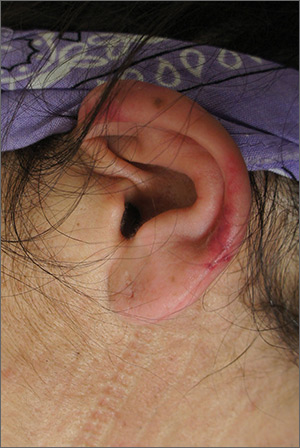
The FP suspected cutaneous vasculitis of the ear caused by levamisole-adulterated cocaine.
Levamisole is an antihelminthic drug approved for veterinary purposes. In the past, the drug had been used as an immune modulator in autoimmune disorders, but no longer is considered safe for human use, as it can cause agranulocytosis. Sellers around the world often lace cocaine with levamisole because it boosts the profits and potentiates the psychoactive effects of the cocaine. Cutaneous vasculitis secondary to levamisole-adulterated cocaine has been reported many times in the literature.
Levamisole-associated vasculitis presents with ear purpura, retiform (like a net) purpura of the trunk or extremities, and neutropenia. Patients will test positive for perinuclear antineutrophil cytoplasmic antibody (pANCA). This cutaneous vasculitis also may present on the nose or face. There are reports of cocaine/levamisole-associated autoimmune syndrome involving agranulocytosis and cutaneous vasculitis.
The patient tested positive for pANCA, as was expected. The FP told her to discontinue her cocaine use, as she ran the risk of worse manifestations. She refused any treatment for her drug use and stated she could stop it on her own. The FP referred the patient to Dermatology, but the vasculitis was barely visible by the time she was seen. Convincing the patient not to use cocaine again remained the only treatment.
Photo courtesy of Jon Karnes, MD, and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R, Martin N, et al. Vasculitis. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1169-1173.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected cutaneous vasculitis of the ear caused by levamisole-adulterated cocaine.
Levamisole is an antihelminthic drug approved for veterinary purposes. In the past, the drug had been used as an immune modulator in autoimmune disorders, but no longer is considered safe for human use, as it can cause agranulocytosis. Sellers around the world often lace cocaine with levamisole because it boosts the profits and potentiates the psychoactive effects of the cocaine. Cutaneous vasculitis secondary to levamisole-adulterated cocaine has been reported many times in the literature.
Levamisole-associated vasculitis presents with ear purpura, retiform (like a net) purpura of the trunk or extremities, and neutropenia. Patients will test positive for perinuclear antineutrophil cytoplasmic antibody (pANCA). This cutaneous vasculitis also may present on the nose or face. There are reports of cocaine/levamisole-associated autoimmune syndrome involving agranulocytosis and cutaneous vasculitis.
The patient tested positive for pANCA, as was expected. The FP told her to discontinue her cocaine use, as she ran the risk of worse manifestations. She refused any treatment for her drug use and stated she could stop it on her own. The FP referred the patient to Dermatology, but the vasculitis was barely visible by the time she was seen. Convincing the patient not to use cocaine again remained the only treatment.
Photo courtesy of Jon Karnes, MD, and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R, Martin N, et al. Vasculitis. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1169-1173.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected cutaneous vasculitis of the ear caused by levamisole-adulterated cocaine.
Levamisole is an antihelminthic drug approved for veterinary purposes. In the past, the drug had been used as an immune modulator in autoimmune disorders, but no longer is considered safe for human use, as it can cause agranulocytosis. Sellers around the world often lace cocaine with levamisole because it boosts the profits and potentiates the psychoactive effects of the cocaine. Cutaneous vasculitis secondary to levamisole-adulterated cocaine has been reported many times in the literature.
Levamisole-associated vasculitis presents with ear purpura, retiform (like a net) purpura of the trunk or extremities, and neutropenia. Patients will test positive for perinuclear antineutrophil cytoplasmic antibody (pANCA). This cutaneous vasculitis also may present on the nose or face. There are reports of cocaine/levamisole-associated autoimmune syndrome involving agranulocytosis and cutaneous vasculitis.
The patient tested positive for pANCA, as was expected. The FP told her to discontinue her cocaine use, as she ran the risk of worse manifestations. She refused any treatment for her drug use and stated she could stop it on her own. The FP referred the patient to Dermatology, but the vasculitis was barely visible by the time she was seen. Convincing the patient not to use cocaine again remained the only treatment.
Photo courtesy of Jon Karnes, MD, and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux EJ, Usatine R, Martin N, et al. Vasculitis. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1169-1173.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/
Expert spotlights telltale clinical signs of xeroderma pigmentosum
AUSTIN, TEX. – If a child presents with acute (XP), a rare autosomal recessive disorder.
Other telltale symptoms of XP include the presence of skin cancer at an early age and a large number of skin cancers.
At the annual meeting of the Society for Pediatric Dermatology, John J. DiGiovanna, MD, described XP as a disorder of genomic instability, which has no cure. It’s caused by a mutation in genes XPA through XPG and the XP variant (XPV) gene. “The genome controls our genes, and UV rays damage DNA,” said Dr. DiGiovanna, who is a senior research physician at the National Cancer Institute’s Laboratory of Cancer and Biology and Genetics, Bethesda, Md. “This damage from UV radiation is similar to damage from chemical agents that form DNA adducts, such as cigarette smoke and certain chemotherapy agents such as cisplatinum.”
XP patients present with or without acute burning after minimal sun exposure, while children with both subtypes develop “freckling” by the time they reach 2 years of age. Dr. DiGiovanna pointed out that lentigo maligna lesions associated with XP resemble freckles at first glance, yet they vary in size, intensity, and border. Meanwhile, freckles in healthy patients are similar in size, are light tan in color, and have a regular border.
“The burning with minimal sun exposure that occurs during childhood leads to pigmentary changes, atrophy, xerosis, and telangiectasias,” he said. A follow-up analysis of 106 XP patients admitted to the National Institutes of Health between 1971 and 2009 found that patients were diagnosed with their first nonmelanoma skin cancer at a median age of 9 years, compared with age 67 among those in the general population (J Med Genet 2011 Mar;48[3]:168-76). “This is a 58-year decrease in age at risk, which is a 10,000-fold increase in skin cancer,” said Dr. DiGiovanna, who was one of the study authors.
Melanoma also occurs at an earlier age among XP patients – a median age of 22 years, compared with a median of 55 years in the general population. “In the general population, melanoma occurs at a younger age than nonmelanoma skin cancer, while in the XP population, melanoma occurs at an older age,” he said. “This is giving us a good biologic lesson that the melanoma induction mechanism must be different from nonmelanoma skin cancer.”
He recalled one XP patient who was followed by NIH researchers for 4 decades. She worked in a doctor’s office and drove a car, but developed progressive neurologic degeneration and died at the age of 40. “This was not due to unrepaired UV damage, but there are other agents which damage other neurons,” Dr. DiGiovanna explained. “Over time, what you get is a decrease in brain volume, an increase in the brain ventricles, and a loss of brain tissue. At postmortem examination, her brain was of infantile size, compared with that of an equivalent 40-year-old. This is a disease of neuronal loss, and it’s progressive. Only about 20%-25% of XP patients experience neural degeneration.”
Management of XP involves strict sun avoidance, including use of a portable UV meter and many layers of UV protection, including application of sunscreen, wearing protective clothing, sunglasses, hats, and face shields, and the use of UV-blocking window film, LED lights, and a vitamin D diet or oral supplementation. Affected individuals also require frequent skin monitoring by the patients and their family members, frequent dermatologic exams by clinicians, biopsy of suspicious lesions, removal of any skin cancers found, field treatments with agents such as 5-fluorouracil and imiquimod, and chemoprevention with oral retinoids for patients who are actively developing large numbers of new lesions (N Engl J Med. 1988 Jun23;318[25]:1633-7).
“Probably the most important thing you can do is refer them to patient support groups,” Dr. DiGiovanna said. “They are present in many countries and can help them manage the day-to-day issues of their condition.” Support groups based in North America include the XP Family Support Group, XP Society, and XP Grupo Luz De Esperanza.
Dr. DiGiovanna reported having no financial disclosures.
AUSTIN, TEX. – If a child presents with acute (XP), a rare autosomal recessive disorder.
Other telltale symptoms of XP include the presence of skin cancer at an early age and a large number of skin cancers.
At the annual meeting of the Society for Pediatric Dermatology, John J. DiGiovanna, MD, described XP as a disorder of genomic instability, which has no cure. It’s caused by a mutation in genes XPA through XPG and the XP variant (XPV) gene. “The genome controls our genes, and UV rays damage DNA,” said Dr. DiGiovanna, who is a senior research physician at the National Cancer Institute’s Laboratory of Cancer and Biology and Genetics, Bethesda, Md. “This damage from UV radiation is similar to damage from chemical agents that form DNA adducts, such as cigarette smoke and certain chemotherapy agents such as cisplatinum.”
XP patients present with or without acute burning after minimal sun exposure, while children with both subtypes develop “freckling” by the time they reach 2 years of age. Dr. DiGiovanna pointed out that lentigo maligna lesions associated with XP resemble freckles at first glance, yet they vary in size, intensity, and border. Meanwhile, freckles in healthy patients are similar in size, are light tan in color, and have a regular border.
“The burning with minimal sun exposure that occurs during childhood leads to pigmentary changes, atrophy, xerosis, and telangiectasias,” he said. A follow-up analysis of 106 XP patients admitted to the National Institutes of Health between 1971 and 2009 found that patients were diagnosed with their first nonmelanoma skin cancer at a median age of 9 years, compared with age 67 among those in the general population (J Med Genet 2011 Mar;48[3]:168-76). “This is a 58-year decrease in age at risk, which is a 10,000-fold increase in skin cancer,” said Dr. DiGiovanna, who was one of the study authors.
Melanoma also occurs at an earlier age among XP patients – a median age of 22 years, compared with a median of 55 years in the general population. “In the general population, melanoma occurs at a younger age than nonmelanoma skin cancer, while in the XP population, melanoma occurs at an older age,” he said. “This is giving us a good biologic lesson that the melanoma induction mechanism must be different from nonmelanoma skin cancer.”
He recalled one XP patient who was followed by NIH researchers for 4 decades. She worked in a doctor’s office and drove a car, but developed progressive neurologic degeneration and died at the age of 40. “This was not due to unrepaired UV damage, but there are other agents which damage other neurons,” Dr. DiGiovanna explained. “Over time, what you get is a decrease in brain volume, an increase in the brain ventricles, and a loss of brain tissue. At postmortem examination, her brain was of infantile size, compared with that of an equivalent 40-year-old. This is a disease of neuronal loss, and it’s progressive. Only about 20%-25% of XP patients experience neural degeneration.”
Management of XP involves strict sun avoidance, including use of a portable UV meter and many layers of UV protection, including application of sunscreen, wearing protective clothing, sunglasses, hats, and face shields, and the use of UV-blocking window film, LED lights, and a vitamin D diet or oral supplementation. Affected individuals also require frequent skin monitoring by the patients and their family members, frequent dermatologic exams by clinicians, biopsy of suspicious lesions, removal of any skin cancers found, field treatments with agents such as 5-fluorouracil and imiquimod, and chemoprevention with oral retinoids for patients who are actively developing large numbers of new lesions (N Engl J Med. 1988 Jun23;318[25]:1633-7).
“Probably the most important thing you can do is refer them to patient support groups,” Dr. DiGiovanna said. “They are present in many countries and can help them manage the day-to-day issues of their condition.” Support groups based in North America include the XP Family Support Group, XP Society, and XP Grupo Luz De Esperanza.
Dr. DiGiovanna reported having no financial disclosures.
AUSTIN, TEX. – If a child presents with acute (XP), a rare autosomal recessive disorder.
Other telltale symptoms of XP include the presence of skin cancer at an early age and a large number of skin cancers.
At the annual meeting of the Society for Pediatric Dermatology, John J. DiGiovanna, MD, described XP as a disorder of genomic instability, which has no cure. It’s caused by a mutation in genes XPA through XPG and the XP variant (XPV) gene. “The genome controls our genes, and UV rays damage DNA,” said Dr. DiGiovanna, who is a senior research physician at the National Cancer Institute’s Laboratory of Cancer and Biology and Genetics, Bethesda, Md. “This damage from UV radiation is similar to damage from chemical agents that form DNA adducts, such as cigarette smoke and certain chemotherapy agents such as cisplatinum.”
XP patients present with or without acute burning after minimal sun exposure, while children with both subtypes develop “freckling” by the time they reach 2 years of age. Dr. DiGiovanna pointed out that lentigo maligna lesions associated with XP resemble freckles at first glance, yet they vary in size, intensity, and border. Meanwhile, freckles in healthy patients are similar in size, are light tan in color, and have a regular border.
“The burning with minimal sun exposure that occurs during childhood leads to pigmentary changes, atrophy, xerosis, and telangiectasias,” he said. A follow-up analysis of 106 XP patients admitted to the National Institutes of Health between 1971 and 2009 found that patients were diagnosed with their first nonmelanoma skin cancer at a median age of 9 years, compared with age 67 among those in the general population (J Med Genet 2011 Mar;48[3]:168-76). “This is a 58-year decrease in age at risk, which is a 10,000-fold increase in skin cancer,” said Dr. DiGiovanna, who was one of the study authors.
Melanoma also occurs at an earlier age among XP patients – a median age of 22 years, compared with a median of 55 years in the general population. “In the general population, melanoma occurs at a younger age than nonmelanoma skin cancer, while in the XP population, melanoma occurs at an older age,” he said. “This is giving us a good biologic lesson that the melanoma induction mechanism must be different from nonmelanoma skin cancer.”
He recalled one XP patient who was followed by NIH researchers for 4 decades. She worked in a doctor’s office and drove a car, but developed progressive neurologic degeneration and died at the age of 40. “This was not due to unrepaired UV damage, but there are other agents which damage other neurons,” Dr. DiGiovanna explained. “Over time, what you get is a decrease in brain volume, an increase in the brain ventricles, and a loss of brain tissue. At postmortem examination, her brain was of infantile size, compared with that of an equivalent 40-year-old. This is a disease of neuronal loss, and it’s progressive. Only about 20%-25% of XP patients experience neural degeneration.”
Management of XP involves strict sun avoidance, including use of a portable UV meter and many layers of UV protection, including application of sunscreen, wearing protective clothing, sunglasses, hats, and face shields, and the use of UV-blocking window film, LED lights, and a vitamin D diet or oral supplementation. Affected individuals also require frequent skin monitoring by the patients and their family members, frequent dermatologic exams by clinicians, biopsy of suspicious lesions, removal of any skin cancers found, field treatments with agents such as 5-fluorouracil and imiquimod, and chemoprevention with oral retinoids for patients who are actively developing large numbers of new lesions (N Engl J Med. 1988 Jun23;318[25]:1633-7).
“Probably the most important thing you can do is refer them to patient support groups,” Dr. DiGiovanna said. “They are present in many countries and can help them manage the day-to-day issues of their condition.” Support groups based in North America include the XP Family Support Group, XP Society, and XP Grupo Luz De Esperanza.
Dr. DiGiovanna reported having no financial disclosures.
REPORTING FROM SPD 2019
Pediatric HSCT recipients still risking sunburn
Young people who have received allogeneic hematopoietic stem cell transplants (HSCTs) are more likely to wear hats, sunscreen and other sun protection, but still intentionally tan and experience sunburn at the same rate as their peers, new research suggests.
In a survey‐based, cross‐sectional cohort study, researchers compared sun-protection behaviors and sun exposure in 85 children aged 21 years and younger who had undergone HSCT and 85 age-, sex-, and skin type–matched controls. The findings were published in Pediatric Dermatology.
HSCT recipients have a higher risk of long-term complications such as skin cancer, for which sun exposure is a major modifiable environmental risk factor.
“Therefore, consistent sun avoidance and protection as well as regular dermatologic evaluations are important for HSCT recipients,” wrote Edward B. Li, PhD, from Harvard Medical School, Boston, and coauthors.
The survey found no significant difference between the transplant and control group in the amount of intentional sun exposure, such as the amount of time spent outside on weekdays and weekends during the peak sun intensity hours of 10 a.m. and 4 p.m. More than one in five transplant recipients (21.2%) reported spending at least 3 hours a day outside between 10 a.m. and 4 p.m. on weekdays, as did 36.5% of transplant recipients on weekends.
There were also no significant differences between the two groups in terms of time spent tanning, either in the sun or in a tanning bed. Additionally, a similar number of transplant recipients and controls experienced one or more red or painful sunburns in the past year (25.9% vs. 27.1%).
However, transplant patients did practice better sun protection behaviors than did the control group, with 60% reporting that they always wore sunscreen, compared with 29.4% of controls. The transplant recipients were also significantly more likely to wear sunglasses and a hat and to stay in the shade or use an umbrella.
“While these data may reflect that HSCT patients are not practicing adequate sun avoidance, it may also suggest that these long‐term survivors are able to enjoy being outdoors as much as their peers and have a similar desire to have a tanned appearance,” the researchers wrote. “While a healthy and active lifestyle should be encouraged for all children, our results emphasize the need for pediatric HSCT survivors to be educated on their increased risk for UV‐related skin cancers, counseled on avoidance of intentional tanning, and advised on the importance of sun protection behaviors in an effort to improve long-term outcomes.”
The researchers noted that transplant recipients were significantly more likely to have had a full body skin exam from a health care professional than were individuals in the control group (61.2% vs. 4.7%) and were more likely to have done a self-check or been checked by a partner in the previous year.
The study was supported by the Society for Pediatric Dermatology, the Dermatology Foundation, the National Institutes of Health, and the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation. One author declared a financial interest in a company developing a dermatological product. No other conflicts of interest were declared.
SOURCE: Li EB et al. Pediatr Dermatol. 2019 Aug 13. doi: 10.1111/pde.13984.
Young people who have received allogeneic hematopoietic stem cell transplants (HSCTs) are more likely to wear hats, sunscreen and other sun protection, but still intentionally tan and experience sunburn at the same rate as their peers, new research suggests.
In a survey‐based, cross‐sectional cohort study, researchers compared sun-protection behaviors and sun exposure in 85 children aged 21 years and younger who had undergone HSCT and 85 age-, sex-, and skin type–matched controls. The findings were published in Pediatric Dermatology.
HSCT recipients have a higher risk of long-term complications such as skin cancer, for which sun exposure is a major modifiable environmental risk factor.
“Therefore, consistent sun avoidance and protection as well as regular dermatologic evaluations are important for HSCT recipients,” wrote Edward B. Li, PhD, from Harvard Medical School, Boston, and coauthors.
The survey found no significant difference between the transplant and control group in the amount of intentional sun exposure, such as the amount of time spent outside on weekdays and weekends during the peak sun intensity hours of 10 a.m. and 4 p.m. More than one in five transplant recipients (21.2%) reported spending at least 3 hours a day outside between 10 a.m. and 4 p.m. on weekdays, as did 36.5% of transplant recipients on weekends.
There were also no significant differences between the two groups in terms of time spent tanning, either in the sun or in a tanning bed. Additionally, a similar number of transplant recipients and controls experienced one or more red or painful sunburns in the past year (25.9% vs. 27.1%).
However, transplant patients did practice better sun protection behaviors than did the control group, with 60% reporting that they always wore sunscreen, compared with 29.4% of controls. The transplant recipients were also significantly more likely to wear sunglasses and a hat and to stay in the shade or use an umbrella.
“While these data may reflect that HSCT patients are not practicing adequate sun avoidance, it may also suggest that these long‐term survivors are able to enjoy being outdoors as much as their peers and have a similar desire to have a tanned appearance,” the researchers wrote. “While a healthy and active lifestyle should be encouraged for all children, our results emphasize the need for pediatric HSCT survivors to be educated on their increased risk for UV‐related skin cancers, counseled on avoidance of intentional tanning, and advised on the importance of sun protection behaviors in an effort to improve long-term outcomes.”
The researchers noted that transplant recipients were significantly more likely to have had a full body skin exam from a health care professional than were individuals in the control group (61.2% vs. 4.7%) and were more likely to have done a self-check or been checked by a partner in the previous year.
The study was supported by the Society for Pediatric Dermatology, the Dermatology Foundation, the National Institutes of Health, and the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation. One author declared a financial interest in a company developing a dermatological product. No other conflicts of interest were declared.
SOURCE: Li EB et al. Pediatr Dermatol. 2019 Aug 13. doi: 10.1111/pde.13984.
Young people who have received allogeneic hematopoietic stem cell transplants (HSCTs) are more likely to wear hats, sunscreen and other sun protection, but still intentionally tan and experience sunburn at the same rate as their peers, new research suggests.
In a survey‐based, cross‐sectional cohort study, researchers compared sun-protection behaviors and sun exposure in 85 children aged 21 years and younger who had undergone HSCT and 85 age-, sex-, and skin type–matched controls. The findings were published in Pediatric Dermatology.
HSCT recipients have a higher risk of long-term complications such as skin cancer, for which sun exposure is a major modifiable environmental risk factor.
“Therefore, consistent sun avoidance and protection as well as regular dermatologic evaluations are important for HSCT recipients,” wrote Edward B. Li, PhD, from Harvard Medical School, Boston, and coauthors.
The survey found no significant difference between the transplant and control group in the amount of intentional sun exposure, such as the amount of time spent outside on weekdays and weekends during the peak sun intensity hours of 10 a.m. and 4 p.m. More than one in five transplant recipients (21.2%) reported spending at least 3 hours a day outside between 10 a.m. and 4 p.m. on weekdays, as did 36.5% of transplant recipients on weekends.
There were also no significant differences between the two groups in terms of time spent tanning, either in the sun or in a tanning bed. Additionally, a similar number of transplant recipients and controls experienced one or more red or painful sunburns in the past year (25.9% vs. 27.1%).
However, transplant patients did practice better sun protection behaviors than did the control group, with 60% reporting that they always wore sunscreen, compared with 29.4% of controls. The transplant recipients were also significantly more likely to wear sunglasses and a hat and to stay in the shade or use an umbrella.
“While these data may reflect that HSCT patients are not practicing adequate sun avoidance, it may also suggest that these long‐term survivors are able to enjoy being outdoors as much as their peers and have a similar desire to have a tanned appearance,” the researchers wrote. “While a healthy and active lifestyle should be encouraged for all children, our results emphasize the need for pediatric HSCT survivors to be educated on their increased risk for UV‐related skin cancers, counseled on avoidance of intentional tanning, and advised on the importance of sun protection behaviors in an effort to improve long-term outcomes.”
The researchers noted that transplant recipients were significantly more likely to have had a full body skin exam from a health care professional than were individuals in the control group (61.2% vs. 4.7%) and were more likely to have done a self-check or been checked by a partner in the previous year.
The study was supported by the Society for Pediatric Dermatology, the Dermatology Foundation, the National Institutes of Health, and the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation. One author declared a financial interest in a company developing a dermatological product. No other conflicts of interest were declared.
SOURCE: Li EB et al. Pediatr Dermatol. 2019 Aug 13. doi: 10.1111/pde.13984.
FROM PEDIATRIC DERMATOLOGY
New evidence supports immune system involvement in hidradenitis suppurativa
Immune dysregulation is a substantial feature of hidradenitis suppurativa in African American patients, according to data from 16 adults.
The etiology of hidradenitis suppurativa (HS) remains unknown, but neutrophils are prominent in affected areas of skin, wrote Angel S. Byrd, MD, PhD, of Johns Hopkins University, Baltimore, and colleagues.
“Among the several functions of neutrophils, these cells have the ability to form neutrophil extracellular traps (NETs) after exposure to certain microbes or sterile stimuli,” the researchers noted. These NETs are weblike structures that have been shown to activate several types of immune responses and promote inflammation.
In a study published in Science Translational Medicine, the researchers analyzed biospecimens from African American HS patients. They determined that NET structures were present in the lesional skin of HS patients, but not in control skin. Additionally, serum from HS patients did not properly degrade healthy control NETs in an in vitro examination.
A significant correlation (P less than .0001) occurred between the amount of NETs released in hidradenitis suppurativa lesions and hidradenitis suppurativa severity.
“Together, these results indicate that NET formation is enhanced in HS, both systemically and in lesional skin, in association with disease severity and that NET degradation mechanisms may be impaired in this disease,” the researchers wrote.
The researchers also found that total immunoglobulin G was significantly increased in the sera of HS patients, compared with that of healthy volunteers (P = .0011), and that enzymes involved in skin citrullination were elevated in the skin of HS patients, compared with controls’ skin.
Previous studies have shown that NETs can promote type I interferon (IFN) signatures in the blood of patients with autoimmune and chronic inflammatory conditions, and in this study, several type I IFN–regulated genes had increased expression in HS skin, including IFI44L, MX1, CXCL10, RSAD2, and IFI27.
The study findings were limited by the small sample size and homogenous population, the researchers noted. Future research should include “the role that neutrophil/type I IFN dysregulation plays in the clinical manifestations of the disease, and how these abnormalities in immune responses regulate other innate and adaptive immune cell types in HS,” which could affect the development of new treatments, they wrote.
The study was supported in part by the National Institutes of Health, the Johns Hopkins School of Medicine Physician Scientist Training Program, and the Danby Hidradenitis Suppurativa Foundation. Dr. Byrd disclosed participating in the Johns Hopkins Ethnic Skin Fellowship, which was supported in part by Valeant. Dr. Byrd also disclosed serving as a former investigator for Eli Lilly.
SOURCE: Byrd AS et al. Sci Transl Med. 2019 Sep 4. doi: 10.1126/scitranslmed.aav5908.
Immune dysregulation is a substantial feature of hidradenitis suppurativa in African American patients, according to data from 16 adults.
The etiology of hidradenitis suppurativa (HS) remains unknown, but neutrophils are prominent in affected areas of skin, wrote Angel S. Byrd, MD, PhD, of Johns Hopkins University, Baltimore, and colleagues.
“Among the several functions of neutrophils, these cells have the ability to form neutrophil extracellular traps (NETs) after exposure to certain microbes or sterile stimuli,” the researchers noted. These NETs are weblike structures that have been shown to activate several types of immune responses and promote inflammation.
In a study published in Science Translational Medicine, the researchers analyzed biospecimens from African American HS patients. They determined that NET structures were present in the lesional skin of HS patients, but not in control skin. Additionally, serum from HS patients did not properly degrade healthy control NETs in an in vitro examination.
A significant correlation (P less than .0001) occurred between the amount of NETs released in hidradenitis suppurativa lesions and hidradenitis suppurativa severity.
“Together, these results indicate that NET formation is enhanced in HS, both systemically and in lesional skin, in association with disease severity and that NET degradation mechanisms may be impaired in this disease,” the researchers wrote.
The researchers also found that total immunoglobulin G was significantly increased in the sera of HS patients, compared with that of healthy volunteers (P = .0011), and that enzymes involved in skin citrullination were elevated in the skin of HS patients, compared with controls’ skin.
Previous studies have shown that NETs can promote type I interferon (IFN) signatures in the blood of patients with autoimmune and chronic inflammatory conditions, and in this study, several type I IFN–regulated genes had increased expression in HS skin, including IFI44L, MX1, CXCL10, RSAD2, and IFI27.
The study findings were limited by the small sample size and homogenous population, the researchers noted. Future research should include “the role that neutrophil/type I IFN dysregulation plays in the clinical manifestations of the disease, and how these abnormalities in immune responses regulate other innate and adaptive immune cell types in HS,” which could affect the development of new treatments, they wrote.
The study was supported in part by the National Institutes of Health, the Johns Hopkins School of Medicine Physician Scientist Training Program, and the Danby Hidradenitis Suppurativa Foundation. Dr. Byrd disclosed participating in the Johns Hopkins Ethnic Skin Fellowship, which was supported in part by Valeant. Dr. Byrd also disclosed serving as a former investigator for Eli Lilly.
SOURCE: Byrd AS et al. Sci Transl Med. 2019 Sep 4. doi: 10.1126/scitranslmed.aav5908.
Immune dysregulation is a substantial feature of hidradenitis suppurativa in African American patients, according to data from 16 adults.
The etiology of hidradenitis suppurativa (HS) remains unknown, but neutrophils are prominent in affected areas of skin, wrote Angel S. Byrd, MD, PhD, of Johns Hopkins University, Baltimore, and colleagues.
“Among the several functions of neutrophils, these cells have the ability to form neutrophil extracellular traps (NETs) after exposure to certain microbes or sterile stimuli,” the researchers noted. These NETs are weblike structures that have been shown to activate several types of immune responses and promote inflammation.
In a study published in Science Translational Medicine, the researchers analyzed biospecimens from African American HS patients. They determined that NET structures were present in the lesional skin of HS patients, but not in control skin. Additionally, serum from HS patients did not properly degrade healthy control NETs in an in vitro examination.
A significant correlation (P less than .0001) occurred between the amount of NETs released in hidradenitis suppurativa lesions and hidradenitis suppurativa severity.
“Together, these results indicate that NET formation is enhanced in HS, both systemically and in lesional skin, in association with disease severity and that NET degradation mechanisms may be impaired in this disease,” the researchers wrote.
The researchers also found that total immunoglobulin G was significantly increased in the sera of HS patients, compared with that of healthy volunteers (P = .0011), and that enzymes involved in skin citrullination were elevated in the skin of HS patients, compared with controls’ skin.
Previous studies have shown that NETs can promote type I interferon (IFN) signatures in the blood of patients with autoimmune and chronic inflammatory conditions, and in this study, several type I IFN–regulated genes had increased expression in HS skin, including IFI44L, MX1, CXCL10, RSAD2, and IFI27.
The study findings were limited by the small sample size and homogenous population, the researchers noted. Future research should include “the role that neutrophil/type I IFN dysregulation plays in the clinical manifestations of the disease, and how these abnormalities in immune responses regulate other innate and adaptive immune cell types in HS,” which could affect the development of new treatments, they wrote.
The study was supported in part by the National Institutes of Health, the Johns Hopkins School of Medicine Physician Scientist Training Program, and the Danby Hidradenitis Suppurativa Foundation. Dr. Byrd disclosed participating in the Johns Hopkins Ethnic Skin Fellowship, which was supported in part by Valeant. Dr. Byrd also disclosed serving as a former investigator for Eli Lilly.
SOURCE: Byrd AS et al. Sci Transl Med. 2019 Sep 4. doi: 10.1126/scitranslmed.aav5908.
FROM SCIENCE TRANSLATIONAL MEDICINE
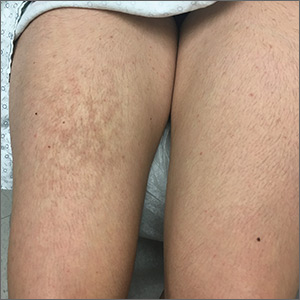
Rash on the thigh
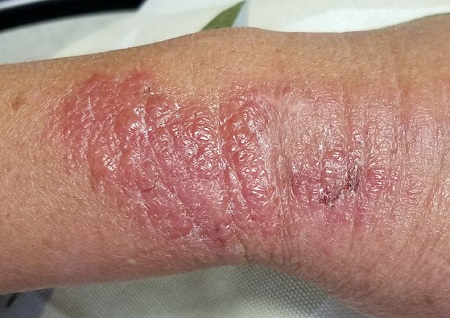
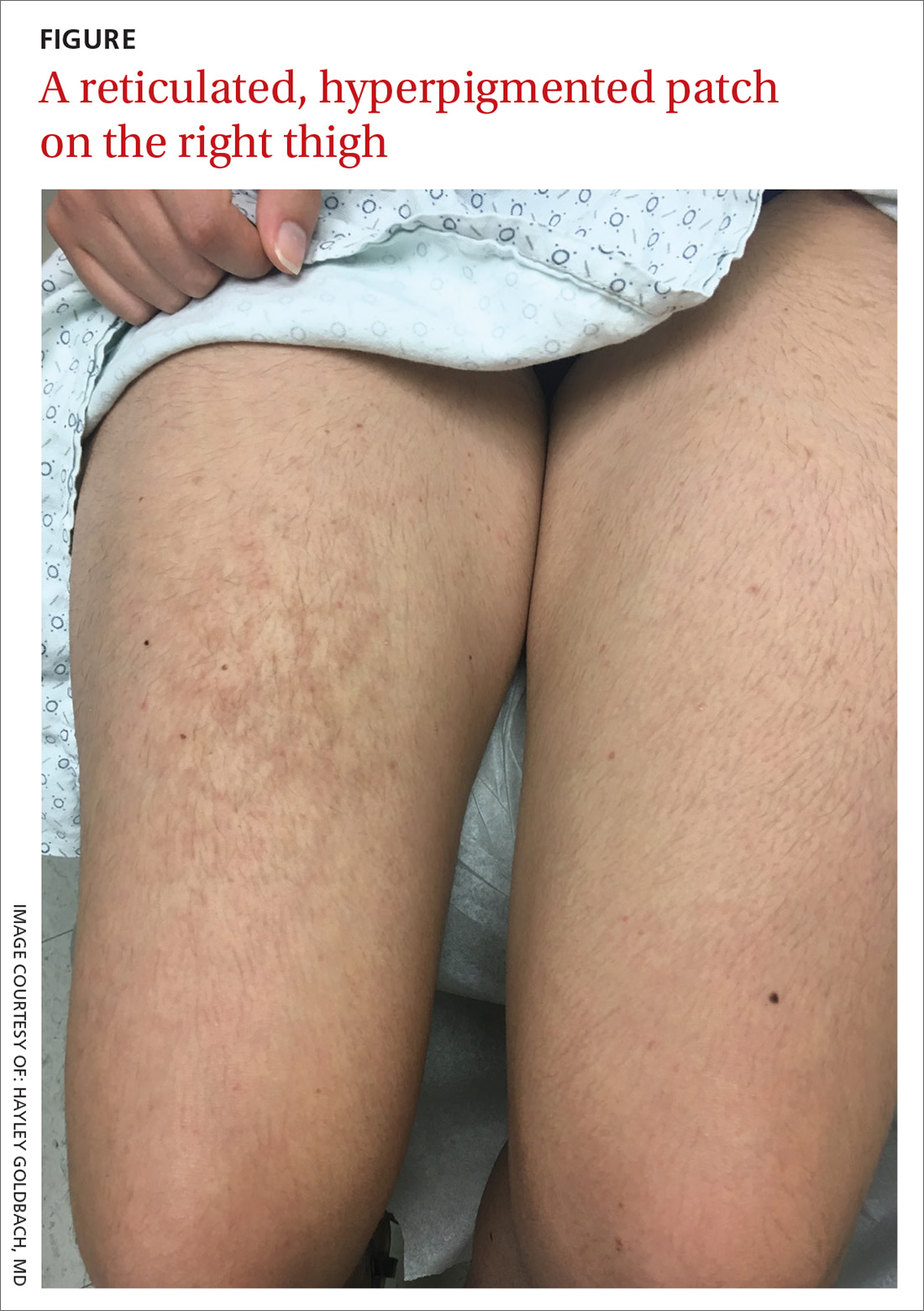
A 21-year-old woman presented with a rash on her right thigh of 3 to 4 months’ duration. She reported that the patch was asymptomatic. She was not taking any medications and otherwise was in good health. A review of systems was negative. The patient was a student who used her laptop frequently. On physical examination, a 10×5-cm reticulated, hyperpigmented patch was seen on her right thigh (FIGURE).
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Erythema ab igne
Erythema ab igne (EAI) is a common dermatosis caused by repeated exposure to infrared radiation, most commonly in the form of low-grade heat (43–47°C).1 Common heat sources include heating pads, heaters, fire, and battery-charged devices. The distribution of the rash is dependent on the location of the heat source and appears as a hyperpigmented, reticulated rash. The pathophysiology is not well understood, but likely involves changes in dermal elastic fibers as well as the dermal venous plexus.2 Though rare, chronic cases of EAI have been associated with cutaneous dysplasia.3
Diagnosis of EAI is made by a combination of medical history and clinical features. Laboratory tests are not required. Additionally, clinicians should inquire about possible heat sources. In this case, we asked the patient whether she rested anything on her thighs, and she acknowledged that this was where she typically placed her laptop computer.
Differential includes other reticulated conditions
The differential diagnosis of a reticulated patch includes other entities likely sharing vascular pathology. The age, sex, and medical history of the patient offer additional diagnostic clues.
Livedo reticularis presents with reticulated erythema. It is unrelated to heat exposure, but may be associated with cold exposure. It can be physiologic or can be associated with vasculitis or another obstruction of blood flow.
Erythema infectiosum is a parvovirus B19 infection that usually presents in young children. It often results in a lacy reticulated exanthem on the face that resembles a slapped cheek in children. Adolescent and adult contacts often present with a more petechial rash in an acral to periflexural distribution.4
Continue to: Polyarteritis nodosa
Polyarteritis nodosa is a rare necrotizing vasculitis of small and medium arteries with an incidence of 4 to 16 cases per million.4 It usually is painful and can present with nodules, ulcers, or bullae and may be associated with livedo-like reticulated pigmentation.
Livedoid vasculitis is a hyalinization of blood vessels leading to the obstruction of vessels due to a hypercoagulable state. It can be acquired or congenital and usually manifests in middle-aged women.4
Management is straight-forward: Remove the heat source
EAI typically is asymptomatic, although there are reports of mild pruritus or a burning sensation. Management includes withdrawal of the heat source and patient education. Our patient’s rash went away when she stopped resting her laptop computer on her lap.
CORRESPONDENCE
Lorraine C. Young, MD, 200 UCLA, Medical Plaza Driveway, Suites 450 & 465, Los Angeles, CA 90095; lcyoung@mednet.ucla.edu
1. Miller K, Hunt R, Chu J, et al. Erythema ab igne. Dermatol Online J. 2011;17:28.
2. Salgado F, Handler MZ, Schwartz RA. Erythema ab igne: new technology rebounding upon its users? Int J Dermatol. 2018;57:393-396.
3. Sigmon JR, Cantrell J, Teague D, et al. Poorly differentiated carcinoma arising in the setting of erythema ab igne. Am J Dermatopathol. 2013;35:676-678.
4. Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier Saunders; 2017.
A 21-year-old woman presented with a rash on her right thigh of 3 to 4 months’ duration. She reported that the patch was asymptomatic. She was not taking any medications and otherwise was in good health. A review of systems was negative. The patient was a student who used her laptop frequently. On physical examination, a 10×5-cm reticulated, hyperpigmented patch was seen on her right thigh (FIGURE).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Erythema ab igne
Erythema ab igne (EAI) is a common dermatosis caused by repeated exposure to infrared radiation, most commonly in the form of low-grade heat (43–47°C).1 Common heat sources include heating pads, heaters, fire, and battery-charged devices. The distribution of the rash is dependent on the location of the heat source and appears as a hyperpigmented, reticulated rash. The pathophysiology is not well understood, but likely involves changes in dermal elastic fibers as well as the dermal venous plexus.2 Though rare, chronic cases of EAI have been associated with cutaneous dysplasia.3
Diagnosis of EAI is made by a combination of medical history and clinical features. Laboratory tests are not required. Additionally, clinicians should inquire about possible heat sources. In this case, we asked the patient whether she rested anything on her thighs, and she acknowledged that this was where she typically placed her laptop computer.
Differential includes other reticulated conditions
The differential diagnosis of a reticulated patch includes other entities likely sharing vascular pathology. The age, sex, and medical history of the patient offer additional diagnostic clues.
Livedo reticularis presents with reticulated erythema. It is unrelated to heat exposure, but may be associated with cold exposure. It can be physiologic or can be associated with vasculitis or another obstruction of blood flow.
Erythema infectiosum is a parvovirus B19 infection that usually presents in young children. It often results in a lacy reticulated exanthem on the face that resembles a slapped cheek in children. Adolescent and adult contacts often present with a more petechial rash in an acral to periflexural distribution.4
Continue to: Polyarteritis nodosa
Polyarteritis nodosa is a rare necrotizing vasculitis of small and medium arteries with an incidence of 4 to 16 cases per million.4 It usually is painful and can present with nodules, ulcers, or bullae and may be associated with livedo-like reticulated pigmentation.
Livedoid vasculitis is a hyalinization of blood vessels leading to the obstruction of vessels due to a hypercoagulable state. It can be acquired or congenital and usually manifests in middle-aged women.4
Management is straight-forward: Remove the heat source
EAI typically is asymptomatic, although there are reports of mild pruritus or a burning sensation. Management includes withdrawal of the heat source and patient education. Our patient’s rash went away when she stopped resting her laptop computer on her lap.
CORRESPONDENCE
Lorraine C. Young, MD, 200 UCLA, Medical Plaza Driveway, Suites 450 & 465, Los Angeles, CA 90095; lcyoung@mednet.ucla.edu
A 21-year-old woman presented with a rash on her right thigh of 3 to 4 months’ duration. She reported that the patch was asymptomatic. She was not taking any medications and otherwise was in good health. A review of systems was negative. The patient was a student who used her laptop frequently. On physical examination, a 10×5-cm reticulated, hyperpigmented patch was seen on her right thigh (FIGURE).

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Erythema ab igne
Erythema ab igne (EAI) is a common dermatosis caused by repeated exposure to infrared radiation, most commonly in the form of low-grade heat (43–47°C).1 Common heat sources include heating pads, heaters, fire, and battery-charged devices. The distribution of the rash is dependent on the location of the heat source and appears as a hyperpigmented, reticulated rash. The pathophysiology is not well understood, but likely involves changes in dermal elastic fibers as well as the dermal venous plexus.2 Though rare, chronic cases of EAI have been associated with cutaneous dysplasia.3
Diagnosis of EAI is made by a combination of medical history and clinical features. Laboratory tests are not required. Additionally, clinicians should inquire about possible heat sources. In this case, we asked the patient whether she rested anything on her thighs, and she acknowledged that this was where she typically placed her laptop computer.
Differential includes other reticulated conditions
The differential diagnosis of a reticulated patch includes other entities likely sharing vascular pathology. The age, sex, and medical history of the patient offer additional diagnostic clues.
Livedo reticularis presents with reticulated erythema. It is unrelated to heat exposure, but may be associated with cold exposure. It can be physiologic or can be associated with vasculitis or another obstruction of blood flow.
Erythema infectiosum is a parvovirus B19 infection that usually presents in young children. It often results in a lacy reticulated exanthem on the face that resembles a slapped cheek in children. Adolescent and adult contacts often present with a more petechial rash in an acral to periflexural distribution.4
Continue to: Polyarteritis nodosa
Polyarteritis nodosa is a rare necrotizing vasculitis of small and medium arteries with an incidence of 4 to 16 cases per million.4 It usually is painful and can present with nodules, ulcers, or bullae and may be associated with livedo-like reticulated pigmentation.
Livedoid vasculitis is a hyalinization of blood vessels leading to the obstruction of vessels due to a hypercoagulable state. It can be acquired or congenital and usually manifests in middle-aged women.4
Management is straight-forward: Remove the heat source
EAI typically is asymptomatic, although there are reports of mild pruritus or a burning sensation. Management includes withdrawal of the heat source and patient education. Our patient’s rash went away when she stopped resting her laptop computer on her lap.
CORRESPONDENCE
Lorraine C. Young, MD, 200 UCLA, Medical Plaza Driveway, Suites 450 & 465, Los Angeles, CA 90095; lcyoung@mednet.ucla.edu
1. Miller K, Hunt R, Chu J, et al. Erythema ab igne. Dermatol Online J. 2011;17:28.
2. Salgado F, Handler MZ, Schwartz RA. Erythema ab igne: new technology rebounding upon its users? Int J Dermatol. 2018;57:393-396.
3. Sigmon JR, Cantrell J, Teague D, et al. Poorly differentiated carcinoma arising in the setting of erythema ab igne. Am J Dermatopathol. 2013;35:676-678.
4. Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier Saunders; 2017.
1. Miller K, Hunt R, Chu J, et al. Erythema ab igne. Dermatol Online J. 2011;17:28.
2. Salgado F, Handler MZ, Schwartz RA. Erythema ab igne: new technology rebounding upon its users? Int J Dermatol. 2018;57:393-396.
3. Sigmon JR, Cantrell J, Teague D, et al. Poorly differentiated carcinoma arising in the setting of erythema ab igne. Am J Dermatopathol. 2013;35:676-678.
4. Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier Saunders; 2017.
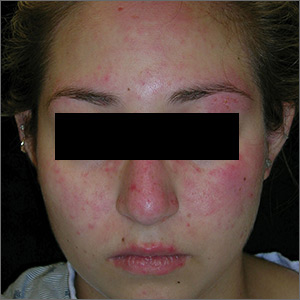
Rash on face, chest, upper arms, and thighs
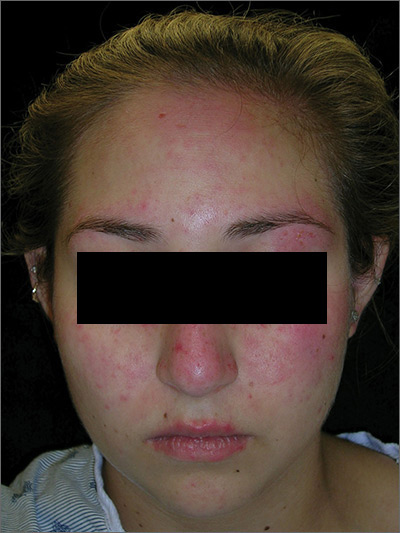
The FP suspected acute systemic lupus erythematosus (SLE) with an acute cutaneous component. Laboratory testing showed a very high level of antinuclear antibodies. The patient was referred to Rheumatology and Dermatology. The dermatologist was available for a phone consult and suggested starting the patient on prednisone 60 mg/d as the Medrol Dosepak that the patient previously received had insufficient prednisolone for this severe flare of acute cutaneous lupus.
Although the classic description of acute SLE involves a butterfly rash, the rash of acute cutaneous lupus can include other areas of the face and body. As was seen in this case, the nasolabial fold tends to be spared and there are often skin erosions and crusting.
Based on the patient’s lab tests and symptoms, the dermatologist determined that the patient met the criteria for SLE. The patient was started on hydroxychloroquine 400 mg/d. The plan was to taper the patient’s prednisone slowly. By the following week, her skin and fatigue were much improved.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Pye A, Mayeaux EJ, Mishra V, et al. Lupus. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1183-1193.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected acute systemic lupus erythematosus (SLE) with an acute cutaneous component. Laboratory testing showed a very high level of antinuclear antibodies. The patient was referred to Rheumatology and Dermatology. The dermatologist was available for a phone consult and suggested starting the patient on prednisone 60 mg/d as the Medrol Dosepak that the patient previously received had insufficient prednisolone for this severe flare of acute cutaneous lupus.
Although the classic description of acute SLE involves a butterfly rash, the rash of acute cutaneous lupus can include other areas of the face and body. As was seen in this case, the nasolabial fold tends to be spared and there are often skin erosions and crusting.
Based on the patient’s lab tests and symptoms, the dermatologist determined that the patient met the criteria for SLE. The patient was started on hydroxychloroquine 400 mg/d. The plan was to taper the patient’s prednisone slowly. By the following week, her skin and fatigue were much improved.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Pye A, Mayeaux EJ, Mishra V, et al. Lupus. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1183-1193.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/

The FP suspected acute systemic lupus erythematosus (SLE) with an acute cutaneous component. Laboratory testing showed a very high level of antinuclear antibodies. The patient was referred to Rheumatology and Dermatology. The dermatologist was available for a phone consult and suggested starting the patient on prednisone 60 mg/d as the Medrol Dosepak that the patient previously received had insufficient prednisolone for this severe flare of acute cutaneous lupus.
Although the classic description of acute SLE involves a butterfly rash, the rash of acute cutaneous lupus can include other areas of the face and body. As was seen in this case, the nasolabial fold tends to be spared and there are often skin erosions and crusting.
Based on the patient’s lab tests and symptoms, the dermatologist determined that the patient met the criteria for SLE. The patient was started on hydroxychloroquine 400 mg/d. The plan was to taper the patient’s prednisone slowly. By the following week, her skin and fatigue were much improved.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Pye A, Mayeaux EJ, Mishra V, et al. Lupus. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1183-1193.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the 3rd edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/
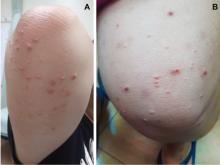
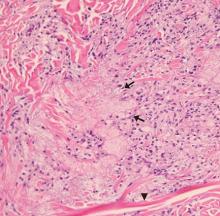
Diabetic dyslipidemia with eruptive xanthoma
A workup for secondary causes of hypertriglyceridemia was negative for hypothyroidism and nephrotic syndrome. She was currently taking no medications. She had no family history of dyslipidemia, and she denied alcohol consumption.
Based on the patient’s presentation, history, and the results of laboratory testing and skin biopsy, the diagnosis was eruptive xanthoma.
A RESULT OF ELEVATED TRIGLYCERIDES
Eruptive xanthoma is associated with elevation of chylomicrons and triglycerides.1 Hyperlipidemia that causes eruptive xanthoma may be familial (ie, due to a primary genetic defect) or secondary to another disease, or both.
Types of primary hypertriglyceridemia include elevated chylomicrons (Frederickson classification type I), elevated very-low-density lipoprotein (VLDL) (Frederickson type IV), and elevation of both chylomicrons and VLDL (Frederickson type V).2,3 Hypertriglyceridemia may also be secondary to obesity, diabetes mellitus, hypothyroidism, nephrotic syndrome, liver cirrhosis, excess ethanol ingestion, and medicines such as retinoids and estrogens.2,3
Lesions of eruptive xanthoma are yellowish papules 2 to 5 mm in diameter surrounded by an erythematous border. They are formed by clusters of foamy cells caused by phagocytosis of macrophages as a consequence of increased accumulations of intracellular lipids. The most common sites are the buttocks, extensor surfaces of the arms, and the back.4
Eruptive xanthoma occurs with markedly elevated triglyceride levels (ie, > 1,000 mg/dL),5 with an estimated prevalence of 18 cases per 100,000 people (< 0.02%).6 Diagnosis is usually established through the clinical history, physical examination, and prompt laboratory confirmation of hypertriglyceridemia. Skin biopsy is rarely if ever needed.
RECOGNIZE AND TREAT PROMPTLY TO AVOID FURTHER COMPLICATIONS
Severe hypertriglyceridemia poses an increased risk of acute pancreatitis. Early recognition and medical treatment in our patient prevented serious complications.
Treatment of eruptive xanthoma includes identifying the underlying cause of hypertriglyceridemia and commencing lifestyle modifications that include weight reduction, aerobic exercise, a strict low-fat diet with avoidance of simple carbohydrates and alcohol,7 and drug therapy.
The patient’s treatment plan
Although HMG-CoA reductase inhibitors (statins) have a modest triglyceride-lowering effect and are useful to modify cardiovascular risk, fibric acid derivatives (eg, gemfibrozil, fenofibrate) are the first-line therapy.8 Omega-3 fatty acids, statins, or niacin may be added if necessary.8
Our patient’s uncontrolled glycemia caused marked hypertriglyceridemia, perhaps from a decrease in lipoprotein lipase activity in adipose tissue and muscle. Lifestyle modifications, glucose-lowering agents (metformin, glimepiride), and fenofibrate were prescribed. She was also advised to seek medical attention if she developed upper-abdominal pain, which could be a symptom of pancreatitis.
- Flynn PD, Burns T, Breathnach S, Cox N, Griffiths C. Xanthomas and abnormalities of lipid metabolism and storage. In: Rook’s Textbook of Dermatology. 8th ed. Oxford: Blackwell Science; 2010.
- Breckenridge WC, Alaupovic P, Cox DW, Little JA. Apolipoprotein and lipoprotein concentrations in familial apolipoprotein C-II deficiency. Atherosclerosis 1982; 44(2):223–235. pmid:7138621
- Santamarina-Fojo S. The familial chylomicronemia syndrome. Endocrinol Metab Clin North Am 1998; 27(3):551–567. pmid:9785052
- Melmed S, Polonsky KS, Larsen PR, Kronenberg H. Williams Textbook of Endocrinology. 13th ed. Philadelphia: Elsevier; 2016.
- Zak A, Zeman M, Slaby A, Vecka M. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2014; 158(2):181–188. doi:10.5507/bp.2014.016
- Leaf DA. Chylomicronemia and the chylomicronemia syndrome: a practical approach to management. Am J Med 2008; 121(1):10–12. doi:10.1016/j.amjmed.2007.10.004
- Hegele RA, Ginsberg HN, Chapman MJ, et al; European Atherosclerosis Society Consensus Panel. The polygenic nature of hypertriglyceridaemia: implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol 2014; 2(8):655–666. doi:10.1016/S2213-8587(13)70191-8
- Berglund L, Brunzell JD, Goldberg AC, et al; Endocrine Society. Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2012; 97(9):2969–2989. doi:10.1210/jc.2011-3213
A workup for secondary causes of hypertriglyceridemia was negative for hypothyroidism and nephrotic syndrome. She was currently taking no medications. She had no family history of dyslipidemia, and she denied alcohol consumption.
Based on the patient’s presentation, history, and the results of laboratory testing and skin biopsy, the diagnosis was eruptive xanthoma.
A RESULT OF ELEVATED TRIGLYCERIDES
Eruptive xanthoma is associated with elevation of chylomicrons and triglycerides.1 Hyperlipidemia that causes eruptive xanthoma may be familial (ie, due to a primary genetic defect) or secondary to another disease, or both.
Types of primary hypertriglyceridemia include elevated chylomicrons (Frederickson classification type I), elevated very-low-density lipoprotein (VLDL) (Frederickson type IV), and elevation of both chylomicrons and VLDL (Frederickson type V).2,3 Hypertriglyceridemia may also be secondary to obesity, diabetes mellitus, hypothyroidism, nephrotic syndrome, liver cirrhosis, excess ethanol ingestion, and medicines such as retinoids and estrogens.2,3
Lesions of eruptive xanthoma are yellowish papules 2 to 5 mm in diameter surrounded by an erythematous border. They are formed by clusters of foamy cells caused by phagocytosis of macrophages as a consequence of increased accumulations of intracellular lipids. The most common sites are the buttocks, extensor surfaces of the arms, and the back.4
Eruptive xanthoma occurs with markedly elevated triglyceride levels (ie, > 1,000 mg/dL),5 with an estimated prevalence of 18 cases per 100,000 people (< 0.02%).6 Diagnosis is usually established through the clinical history, physical examination, and prompt laboratory confirmation of hypertriglyceridemia. Skin biopsy is rarely if ever needed.
RECOGNIZE AND TREAT PROMPTLY TO AVOID FURTHER COMPLICATIONS
Severe hypertriglyceridemia poses an increased risk of acute pancreatitis. Early recognition and medical treatment in our patient prevented serious complications.
Treatment of eruptive xanthoma includes identifying the underlying cause of hypertriglyceridemia and commencing lifestyle modifications that include weight reduction, aerobic exercise, a strict low-fat diet with avoidance of simple carbohydrates and alcohol,7 and drug therapy.
The patient’s treatment plan
Although HMG-CoA reductase inhibitors (statins) have a modest triglyceride-lowering effect and are useful to modify cardiovascular risk, fibric acid derivatives (eg, gemfibrozil, fenofibrate) are the first-line therapy.8 Omega-3 fatty acids, statins, or niacin may be added if necessary.8
Our patient’s uncontrolled glycemia caused marked hypertriglyceridemia, perhaps from a decrease in lipoprotein lipase activity in adipose tissue and muscle. Lifestyle modifications, glucose-lowering agents (metformin, glimepiride), and fenofibrate were prescribed. She was also advised to seek medical attention if she developed upper-abdominal pain, which could be a symptom of pancreatitis.
A workup for secondary causes of hypertriglyceridemia was negative for hypothyroidism and nephrotic syndrome. She was currently taking no medications. She had no family history of dyslipidemia, and she denied alcohol consumption.
Based on the patient’s presentation, history, and the results of laboratory testing and skin biopsy, the diagnosis was eruptive xanthoma.
A RESULT OF ELEVATED TRIGLYCERIDES
Eruptive xanthoma is associated with elevation of chylomicrons and triglycerides.1 Hyperlipidemia that causes eruptive xanthoma may be familial (ie, due to a primary genetic defect) or secondary to another disease, or both.
Types of primary hypertriglyceridemia include elevated chylomicrons (Frederickson classification type I), elevated very-low-density lipoprotein (VLDL) (Frederickson type IV), and elevation of both chylomicrons and VLDL (Frederickson type V).2,3 Hypertriglyceridemia may also be secondary to obesity, diabetes mellitus, hypothyroidism, nephrotic syndrome, liver cirrhosis, excess ethanol ingestion, and medicines such as retinoids and estrogens.2,3
Lesions of eruptive xanthoma are yellowish papules 2 to 5 mm in diameter surrounded by an erythematous border. They are formed by clusters of foamy cells caused by phagocytosis of macrophages as a consequence of increased accumulations of intracellular lipids. The most common sites are the buttocks, extensor surfaces of the arms, and the back.4
Eruptive xanthoma occurs with markedly elevated triglyceride levels (ie, > 1,000 mg/dL),5 with an estimated prevalence of 18 cases per 100,000 people (< 0.02%).6 Diagnosis is usually established through the clinical history, physical examination, and prompt laboratory confirmation of hypertriglyceridemia. Skin biopsy is rarely if ever needed.
RECOGNIZE AND TREAT PROMPTLY TO AVOID FURTHER COMPLICATIONS
Severe hypertriglyceridemia poses an increased risk of acute pancreatitis. Early recognition and medical treatment in our patient prevented serious complications.
Treatment of eruptive xanthoma includes identifying the underlying cause of hypertriglyceridemia and commencing lifestyle modifications that include weight reduction, aerobic exercise, a strict low-fat diet with avoidance of simple carbohydrates and alcohol,7 and drug therapy.
The patient’s treatment plan
Although HMG-CoA reductase inhibitors (statins) have a modest triglyceride-lowering effect and are useful to modify cardiovascular risk, fibric acid derivatives (eg, gemfibrozil, fenofibrate) are the first-line therapy.8 Omega-3 fatty acids, statins, or niacin may be added if necessary.8
Our patient’s uncontrolled glycemia caused marked hypertriglyceridemia, perhaps from a decrease in lipoprotein lipase activity in adipose tissue and muscle. Lifestyle modifications, glucose-lowering agents (metformin, glimepiride), and fenofibrate were prescribed. She was also advised to seek medical attention if she developed upper-abdominal pain, which could be a symptom of pancreatitis.
- Flynn PD, Burns T, Breathnach S, Cox N, Griffiths C. Xanthomas and abnormalities of lipid metabolism and storage. In: Rook’s Textbook of Dermatology. 8th ed. Oxford: Blackwell Science; 2010.
- Breckenridge WC, Alaupovic P, Cox DW, Little JA. Apolipoprotein and lipoprotein concentrations in familial apolipoprotein C-II deficiency. Atherosclerosis 1982; 44(2):223–235. pmid:7138621
- Santamarina-Fojo S. The familial chylomicronemia syndrome. Endocrinol Metab Clin North Am 1998; 27(3):551–567. pmid:9785052
- Melmed S, Polonsky KS, Larsen PR, Kronenberg H. Williams Textbook of Endocrinology. 13th ed. Philadelphia: Elsevier; 2016.
- Zak A, Zeman M, Slaby A, Vecka M. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2014; 158(2):181–188. doi:10.5507/bp.2014.016
- Leaf DA. Chylomicronemia and the chylomicronemia syndrome: a practical approach to management. Am J Med 2008; 121(1):10–12. doi:10.1016/j.amjmed.2007.10.004
- Hegele RA, Ginsberg HN, Chapman MJ, et al; European Atherosclerosis Society Consensus Panel. The polygenic nature of hypertriglyceridaemia: implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol 2014; 2(8):655–666. doi:10.1016/S2213-8587(13)70191-8
- Berglund L, Brunzell JD, Goldberg AC, et al; Endocrine Society. Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2012; 97(9):2969–2989. doi:10.1210/jc.2011-3213
- Flynn PD, Burns T, Breathnach S, Cox N, Griffiths C. Xanthomas and abnormalities of lipid metabolism and storage. In: Rook’s Textbook of Dermatology. 8th ed. Oxford: Blackwell Science; 2010.
- Breckenridge WC, Alaupovic P, Cox DW, Little JA. Apolipoprotein and lipoprotein concentrations in familial apolipoprotein C-II deficiency. Atherosclerosis 1982; 44(2):223–235. pmid:7138621
- Santamarina-Fojo S. The familial chylomicronemia syndrome. Endocrinol Metab Clin North Am 1998; 27(3):551–567. pmid:9785052
- Melmed S, Polonsky KS, Larsen PR, Kronenberg H. Williams Textbook of Endocrinology. 13th ed. Philadelphia: Elsevier; 2016.
- Zak A, Zeman M, Slaby A, Vecka M. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2014; 158(2):181–188. doi:10.5507/bp.2014.016
- Leaf DA. Chylomicronemia and the chylomicronemia syndrome: a practical approach to management. Am J Med 2008; 121(1):10–12. doi:10.1016/j.amjmed.2007.10.004
- Hegele RA, Ginsberg HN, Chapman MJ, et al; European Atherosclerosis Society Consensus Panel. The polygenic nature of hypertriglyceridaemia: implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol 2014; 2(8):655–666. doi:10.1016/S2213-8587(13)70191-8
- Berglund L, Brunzell JD, Goldberg AC, et al; Endocrine Society. Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2012; 97(9):2969–2989. doi:10.1210/jc.2011-3213
Cephalosporins remain empiric therapy for skin infections in pediatric AD
“Clindamycin, tetracyclines, or TMP‐SMX can be considered in patients suspected to have, or with a history of, MRSA [methicillin‐resistant S. aureus] infection,” wrote Cristopher C. Briscoe, MD, of the Washington University School of Medicine in St. Louis, Missouri, and his coauthors. The study was published in Pediatric Dermatology.
To determine the optimal empiric antibiotic for pediatric AD patients with skin infections, the researchers analyzed skin cultures from 106 patients seen at Saint Louis Children’s Hospital (SLCH). The results were also compared to cultures from pediatric patients who presented at the SLCH emergency department (ED) with S. aureus skin abscesses.
Of the 170 cultures that grew S. aureus, 130 (77.8%) grew MSSA, and 37 (22.2%) grew MRSA. Three of the cultures grew both. The prevalence of MRSA in the cohort differed from the prevalence in the ED patients (44%). The prevalence of either infection did not differ significantly by age, sex or race, though the average number of cultures in African American patients topped the average for Caucasian patients (1.8 vs. 1.2, P less than .003).
All patients with MSSA – in both the cohort and the ED – proved 100% susceptible to cefazolin. Cohort patients with MSSA saw lower susceptibility to doxycycline compared to the ED patients (89.4% vs. 97%), as did MRSA cohort patients to trimethoprim‐sulfamethoxazole (92% vs. 98%).
“When a patient with AD walks into your office and looks like they have an infection of their eczema, your go-to antibiotic is going to be one that targets MSSA,” said coauthor Carrie Coughlin, MD, of the Washington University School of Medicine in an interview. “You’ll still do a culture to prove or disprove that assumption, but it gives you a guide to help make that patient better in the short term while you work things up.”
“Also, remember that MSSA is not ‘better’ to have than MRSA,” she added. “You can now see some of the virulence factors from MRSA strains in MSSA strains, so treating both of them is important.”
The authors acknowledged their study’s limitations, including the limited generalizability of a single-center design and a lack of information as to the body sites from which the cultures were obtained. They were also unable to reliably determine prior antibiotic exposure, noting that “future work could examine whether prior exposure differed significantly in the MRSA and MSSA groups.”
The study was funded by grants from the Agency for Healthcare Research and Quality. The authors reported no conflicts of interest.
SOURCE: Briscoe CC et al. Pediatr Dermatol. 2019 May 24. doi: 10.1111/pde.13867.
“Clindamycin, tetracyclines, or TMP‐SMX can be considered in patients suspected to have, or with a history of, MRSA [methicillin‐resistant S. aureus] infection,” wrote Cristopher C. Briscoe, MD, of the Washington University School of Medicine in St. Louis, Missouri, and his coauthors. The study was published in Pediatric Dermatology.
To determine the optimal empiric antibiotic for pediatric AD patients with skin infections, the researchers analyzed skin cultures from 106 patients seen at Saint Louis Children’s Hospital (SLCH). The results were also compared to cultures from pediatric patients who presented at the SLCH emergency department (ED) with S. aureus skin abscesses.
Of the 170 cultures that grew S. aureus, 130 (77.8%) grew MSSA, and 37 (22.2%) grew MRSA. Three of the cultures grew both. The prevalence of MRSA in the cohort differed from the prevalence in the ED patients (44%). The prevalence of either infection did not differ significantly by age, sex or race, though the average number of cultures in African American patients topped the average for Caucasian patients (1.8 vs. 1.2, P less than .003).
All patients with MSSA – in both the cohort and the ED – proved 100% susceptible to cefazolin. Cohort patients with MSSA saw lower susceptibility to doxycycline compared to the ED patients (89.4% vs. 97%), as did MRSA cohort patients to trimethoprim‐sulfamethoxazole (92% vs. 98%).
“When a patient with AD walks into your office and looks like they have an infection of their eczema, your go-to antibiotic is going to be one that targets MSSA,” said coauthor Carrie Coughlin, MD, of the Washington University School of Medicine in an interview. “You’ll still do a culture to prove or disprove that assumption, but it gives you a guide to help make that patient better in the short term while you work things up.”
“Also, remember that MSSA is not ‘better’ to have than MRSA,” she added. “You can now see some of the virulence factors from MRSA strains in MSSA strains, so treating both of them is important.”
The authors acknowledged their study’s limitations, including the limited generalizability of a single-center design and a lack of information as to the body sites from which the cultures were obtained. They were also unable to reliably determine prior antibiotic exposure, noting that “future work could examine whether prior exposure differed significantly in the MRSA and MSSA groups.”
The study was funded by grants from the Agency for Healthcare Research and Quality. The authors reported no conflicts of interest.
SOURCE: Briscoe CC et al. Pediatr Dermatol. 2019 May 24. doi: 10.1111/pde.13867.
“Clindamycin, tetracyclines, or TMP‐SMX can be considered in patients suspected to have, or with a history of, MRSA [methicillin‐resistant S. aureus] infection,” wrote Cristopher C. Briscoe, MD, of the Washington University School of Medicine in St. Louis, Missouri, and his coauthors. The study was published in Pediatric Dermatology.
To determine the optimal empiric antibiotic for pediatric AD patients with skin infections, the researchers analyzed skin cultures from 106 patients seen at Saint Louis Children’s Hospital (SLCH). The results were also compared to cultures from pediatric patients who presented at the SLCH emergency department (ED) with S. aureus skin abscesses.
Of the 170 cultures that grew S. aureus, 130 (77.8%) grew MSSA, and 37 (22.2%) grew MRSA. Three of the cultures grew both. The prevalence of MRSA in the cohort differed from the prevalence in the ED patients (44%). The prevalence of either infection did not differ significantly by age, sex or race, though the average number of cultures in African American patients topped the average for Caucasian patients (1.8 vs. 1.2, P less than .003).
All patients with MSSA – in both the cohort and the ED – proved 100% susceptible to cefazolin. Cohort patients with MSSA saw lower susceptibility to doxycycline compared to the ED patients (89.4% vs. 97%), as did MRSA cohort patients to trimethoprim‐sulfamethoxazole (92% vs. 98%).
“When a patient with AD walks into your office and looks like they have an infection of their eczema, your go-to antibiotic is going to be one that targets MSSA,” said coauthor Carrie Coughlin, MD, of the Washington University School of Medicine in an interview. “You’ll still do a culture to prove or disprove that assumption, but it gives you a guide to help make that patient better in the short term while you work things up.”
“Also, remember that MSSA is not ‘better’ to have than MRSA,” she added. “You can now see some of the virulence factors from MRSA strains in MSSA strains, so treating both of them is important.”
The authors acknowledged their study’s limitations, including the limited generalizability of a single-center design and a lack of information as to the body sites from which the cultures were obtained. They were also unable to reliably determine prior antibiotic exposure, noting that “future work could examine whether prior exposure differed significantly in the MRSA and MSSA groups.”
The study was funded by grants from the Agency for Healthcare Research and Quality. The authors reported no conflicts of interest.
SOURCE: Briscoe CC et al. Pediatr Dermatol. 2019 May 24. doi: 10.1111/pde.13867.
FROM PEDIATRIC DERMATOLOGY
Tender nodules after leprosy Tx
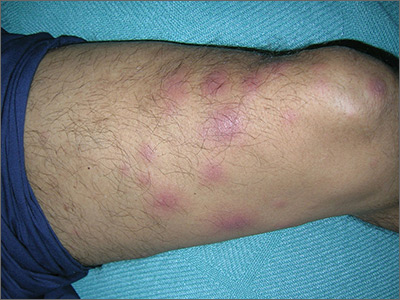
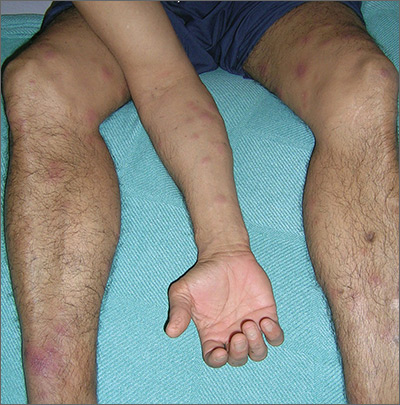
The treating physicians believed that the patient had developed erythema nodosum leprosum (ENL), based on the tender nodules that are characteristic of this treatment reaction. The CDC was again consulted and representatives there agreed.
ENL is a known as a Type 2 reaction to the treatment for leprosy. Management involves the use of oral prednisone, which may be needed for a prolonged time. The antibiotics used to treat leprosy should not be stopped, as ENL is not an allergic reaction. It is due to the destruction of bacilli and the immune response to the release of bacterial antigens.
The patient was transferred to a leprosy hospital in Baton Rouge, Louisiana for further management. The prednisone was not sufficient, and he required further treatment with thalidomide. After 2 years of treatment, he appeared to be free of leprosy and no longer had the ENL.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux, EJ, Diaz L, Paulis R. Erythema nodosum. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1169-1173.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the third edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/


The treating physicians believed that the patient had developed erythema nodosum leprosum (ENL), based on the tender nodules that are characteristic of this treatment reaction. The CDC was again consulted and representatives there agreed.
ENL is a known as a Type 2 reaction to the treatment for leprosy. Management involves the use of oral prednisone, which may be needed for a prolonged time. The antibiotics used to treat leprosy should not be stopped, as ENL is not an allergic reaction. It is due to the destruction of bacilli and the immune response to the release of bacterial antigens.
The patient was transferred to a leprosy hospital in Baton Rouge, Louisiana for further management. The prednisone was not sufficient, and he required further treatment with thalidomide. After 2 years of treatment, he appeared to be free of leprosy and no longer had the ENL.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux, EJ, Diaz L, Paulis R. Erythema nodosum. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1169-1173.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the third edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/


The treating physicians believed that the patient had developed erythema nodosum leprosum (ENL), based on the tender nodules that are characteristic of this treatment reaction. The CDC was again consulted and representatives there agreed.
ENL is a known as a Type 2 reaction to the treatment for leprosy. Management involves the use of oral prednisone, which may be needed for a prolonged time. The antibiotics used to treat leprosy should not be stopped, as ENL is not an allergic reaction. It is due to the destruction of bacilli and the immune response to the release of bacterial antigens.
The patient was transferred to a leprosy hospital in Baton Rouge, Louisiana for further management. The prednisone was not sufficient, and he required further treatment with thalidomide. After 2 years of treatment, he appeared to be free of leprosy and no longer had the ENL.
Photos and text for Photo Rounds Friday courtesy of Richard P. Usatine, MD. This case was adapted from: Mayeaux, EJ, Diaz L, Paulis R. Erythema nodosum. In: Usatine R, Smith M, Mayeaux EJ, et al, eds. Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019:1169-1173.
To learn more about the newest 3rd edition of the Color Atlas and Synopsis of Family Medicine, see: https://www.amazon.com/Color-Atlas-Synopsis-Family-Medicine/dp/1259862046/
You can get the third edition of the Color Atlas and Synopsis of Family Medicine as an app by clicking on this link: https://usatinemedia.com/app/color-atlas-of-family-medicine/