User login
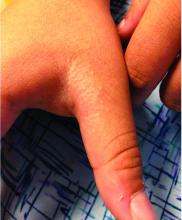
Rash on hands and feet
Lichenoid dermatoses are a heterogeneous group of diseases with varying clinical presentations. The term “lichenoid” refers to the popular lesions of certain skin disorders of which lichen planus (LP) is the prototype. The papules are shiny, flat topped, polygonal, of different sizes, and occur in clusters creating a pattern that resembles lichen growing on a rock. Lichenoid eruptions are quite common in children and can result from many different origins. In most instances the precise mechanism of disease is not known, although it is usually believed to be immunologic in nature. Certain disorders are common in children, whereas others more often affect the adult population.
Lichen striatus, lichen nitidus (LN), and lichen spinulosus are lichenoid lesions that are more common in children than adults.
LN – as seen in the patient described here – is an uncommon benign inflammatory skin disease, primarily of children. Individual lesions are sharply demarcated, pinpoint to pinhead sized, round or polygonal, and strikingly monomorphous in nature. The papules are usually flesh colored, however, the color varies from yellow and brown to violet hues depending on the background color of the patient’s skin. This variation in color is in contrast with LP which is characteristically violaceous. The surfaces of the papules are flat, shiny, and slightly elevated. They may have a fine scale or a hyperkeratotic plug. The lesions tend to occur in groups, primarily on the abdomen, chest, glans penis, and upper extremities. The Koebner phenomenon is observed and is a hallmark for the disorder. LN is generally asymptomatic, unlike LP, which is exceedingly pruritic.
The cause of LN is unknown; however, it has been proposed that LN, in particular generalized LN, may be associated with immune alterations in the patient. The course of LN is slowly progressive with a tendency toward remission. The lesions can remain stationary for years; however, they sometimes disappear spontaneously and completely.
The differential diagnosis of LN beyond the entities discussed above includes frictional lichenoid eruption, lichenoid drug eruption, LP, and keratosis pilaris.
LP is the classic lichenoid eruption. It is rare in children and occurs most frequently in individuals aged 30-60 years. LP usually manifests as an extremely pruritic eruption of flat-topped polygonal and violaceous papules that often have fine linear white scales known as Wickham striae. The distribution is usually bilateral and symmetric with most of the papules and plaques located on the legs, flexor wrists, neck, and genitalia. The lesions may exhibit the Koebner phenomenon, appearing in a linear pattern along the site of a scratch. Generally, in childhood cases there is reported itching, and oral and nail lesions are less common.
Frictional lichenoid eruption occurs in childhood. The lesions consist of lichenoid papules with regular borders 1-2 mm in diameter that generally are asymptomatic, although they may be mildly pruritic. The papules are found in a very characteristic distribution with almost exclusive involvement of the backs of the hands, fingers, elbows, and knees with occasional involvement of the extensor forearms and cheeks. This disorder occurs in predisposed children who have been exposed to significant frictional force during play, and typically resolves spontaneously after removal of the stimulus.
Keratosis pilaris is a rash that usually is found on the outer areas of the upper arms, upper thighs, buttocks, and cheeks. It consists of small bumps that are flesh colored to red. The bumps generally don’t hurt or itch.
The lack of symptoms and spontaneous healing have rendered treatment unnecessary in most cases. LN generally is self-limiting, thus treatment may not be necessary. However, topical treatment with mid- to high-potency corticosteroids has hastened resolution of lesions in some children, as have topical dinitrochlorobenzene and systemic treatment with psoralens, astemizole, etretinate, and psoralen-UVA.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Neither Dr. Eichenfield nor Dr. Bhatti has any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
Pickert A. Cutis. 2012 Sep;90(3):E1-3. https://mdedge-files-live.s3.us-east-2.amazonaws.com/files/s3fs-public/Document/September-2017/0900300E1.pdf Tziotzios C et al. J Am Acad Dermatol. 2018 Nov;79(5):789-804. Tilly JJ et al. J Am Acad Dermatol. 2004 Oct;51(4):606-24.
Lichenoid dermatoses are a heterogeneous group of diseases with varying clinical presentations. The term “lichenoid” refers to the popular lesions of certain skin disorders of which lichen planus (LP) is the prototype. The papules are shiny, flat topped, polygonal, of different sizes, and occur in clusters creating a pattern that resembles lichen growing on a rock. Lichenoid eruptions are quite common in children and can result from many different origins. In most instances the precise mechanism of disease is not known, although it is usually believed to be immunologic in nature. Certain disorders are common in children, whereas others more often affect the adult population.
Lichen striatus, lichen nitidus (LN), and lichen spinulosus are lichenoid lesions that are more common in children than adults.
LN – as seen in the patient described here – is an uncommon benign inflammatory skin disease, primarily of children. Individual lesions are sharply demarcated, pinpoint to pinhead sized, round or polygonal, and strikingly monomorphous in nature. The papules are usually flesh colored, however, the color varies from yellow and brown to violet hues depending on the background color of the patient’s skin. This variation in color is in contrast with LP which is characteristically violaceous. The surfaces of the papules are flat, shiny, and slightly elevated. They may have a fine scale or a hyperkeratotic plug. The lesions tend to occur in groups, primarily on the abdomen, chest, glans penis, and upper extremities. The Koebner phenomenon is observed and is a hallmark for the disorder. LN is generally asymptomatic, unlike LP, which is exceedingly pruritic.
The cause of LN is unknown; however, it has been proposed that LN, in particular generalized LN, may be associated with immune alterations in the patient. The course of LN is slowly progressive with a tendency toward remission. The lesions can remain stationary for years; however, they sometimes disappear spontaneously and completely.
The differential diagnosis of LN beyond the entities discussed above includes frictional lichenoid eruption, lichenoid drug eruption, LP, and keratosis pilaris.
LP is the classic lichenoid eruption. It is rare in children and occurs most frequently in individuals aged 30-60 years. LP usually manifests as an extremely pruritic eruption of flat-topped polygonal and violaceous papules that often have fine linear white scales known as Wickham striae. The distribution is usually bilateral and symmetric with most of the papules and plaques located on the legs, flexor wrists, neck, and genitalia. The lesions may exhibit the Koebner phenomenon, appearing in a linear pattern along the site of a scratch. Generally, in childhood cases there is reported itching, and oral and nail lesions are less common.
Frictional lichenoid eruption occurs in childhood. The lesions consist of lichenoid papules with regular borders 1-2 mm in diameter that generally are asymptomatic, although they may be mildly pruritic. The papules are found in a very characteristic distribution with almost exclusive involvement of the backs of the hands, fingers, elbows, and knees with occasional involvement of the extensor forearms and cheeks. This disorder occurs in predisposed children who have been exposed to significant frictional force during play, and typically resolves spontaneously after removal of the stimulus.
Keratosis pilaris is a rash that usually is found on the outer areas of the upper arms, upper thighs, buttocks, and cheeks. It consists of small bumps that are flesh colored to red. The bumps generally don’t hurt or itch.
The lack of symptoms and spontaneous healing have rendered treatment unnecessary in most cases. LN generally is self-limiting, thus treatment may not be necessary. However, topical treatment with mid- to high-potency corticosteroids has hastened resolution of lesions in some children, as have topical dinitrochlorobenzene and systemic treatment with psoralens, astemizole, etretinate, and psoralen-UVA.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Neither Dr. Eichenfield nor Dr. Bhatti has any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
Pickert A. Cutis. 2012 Sep;90(3):E1-3. https://mdedge-files-live.s3.us-east-2.amazonaws.com/files/s3fs-public/Document/September-2017/0900300E1.pdf Tziotzios C et al. J Am Acad Dermatol. 2018 Nov;79(5):789-804. Tilly JJ et al. J Am Acad Dermatol. 2004 Oct;51(4):606-24.
Lichenoid dermatoses are a heterogeneous group of diseases with varying clinical presentations. The term “lichenoid” refers to the popular lesions of certain skin disorders of which lichen planus (LP) is the prototype. The papules are shiny, flat topped, polygonal, of different sizes, and occur in clusters creating a pattern that resembles lichen growing on a rock. Lichenoid eruptions are quite common in children and can result from many different origins. In most instances the precise mechanism of disease is not known, although it is usually believed to be immunologic in nature. Certain disorders are common in children, whereas others more often affect the adult population.
Lichen striatus, lichen nitidus (LN), and lichen spinulosus are lichenoid lesions that are more common in children than adults.
LN – as seen in the patient described here – is an uncommon benign inflammatory skin disease, primarily of children. Individual lesions are sharply demarcated, pinpoint to pinhead sized, round or polygonal, and strikingly monomorphous in nature. The papules are usually flesh colored, however, the color varies from yellow and brown to violet hues depending on the background color of the patient’s skin. This variation in color is in contrast with LP which is characteristically violaceous. The surfaces of the papules are flat, shiny, and slightly elevated. They may have a fine scale or a hyperkeratotic plug. The lesions tend to occur in groups, primarily on the abdomen, chest, glans penis, and upper extremities. The Koebner phenomenon is observed and is a hallmark for the disorder. LN is generally asymptomatic, unlike LP, which is exceedingly pruritic.
The cause of LN is unknown; however, it has been proposed that LN, in particular generalized LN, may be associated with immune alterations in the patient. The course of LN is slowly progressive with a tendency toward remission. The lesions can remain stationary for years; however, they sometimes disappear spontaneously and completely.
The differential diagnosis of LN beyond the entities discussed above includes frictional lichenoid eruption, lichenoid drug eruption, LP, and keratosis pilaris.
LP is the classic lichenoid eruption. It is rare in children and occurs most frequently in individuals aged 30-60 years. LP usually manifests as an extremely pruritic eruption of flat-topped polygonal and violaceous papules that often have fine linear white scales known as Wickham striae. The distribution is usually bilateral and symmetric with most of the papules and plaques located on the legs, flexor wrists, neck, and genitalia. The lesions may exhibit the Koebner phenomenon, appearing in a linear pattern along the site of a scratch. Generally, in childhood cases there is reported itching, and oral and nail lesions are less common.
Frictional lichenoid eruption occurs in childhood. The lesions consist of lichenoid papules with regular borders 1-2 mm in diameter that generally are asymptomatic, although they may be mildly pruritic. The papules are found in a very characteristic distribution with almost exclusive involvement of the backs of the hands, fingers, elbows, and knees with occasional involvement of the extensor forearms and cheeks. This disorder occurs in predisposed children who have been exposed to significant frictional force during play, and typically resolves spontaneously after removal of the stimulus.
Keratosis pilaris is a rash that usually is found on the outer areas of the upper arms, upper thighs, buttocks, and cheeks. It consists of small bumps that are flesh colored to red. The bumps generally don’t hurt or itch.
The lack of symptoms and spontaneous healing have rendered treatment unnecessary in most cases. LN generally is self-limiting, thus treatment may not be necessary. However, topical treatment with mid- to high-potency corticosteroids has hastened resolution of lesions in some children, as have topical dinitrochlorobenzene and systemic treatment with psoralens, astemizole, etretinate, and psoralen-UVA.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Neither Dr. Eichenfield nor Dr. Bhatti has any relevant financial disclosures. Email them at pdnews@mdedge.com.
References
Pickert A. Cutis. 2012 Sep;90(3):E1-3. https://mdedge-files-live.s3.us-east-2.amazonaws.com/files/s3fs-public/Document/September-2017/0900300E1.pdf Tziotzios C et al. J Am Acad Dermatol. 2018 Nov;79(5):789-804. Tilly JJ et al. J Am Acad Dermatol. 2004 Oct;51(4):606-24.
A 9-year-old healthy Kuwaiti male with no significant past medical history presents with a rash on his hands and feet that has been present for 3 years.
His mother reports that he has been seen by dermatologists in various countries and was last seen by a dermatologist in Kuwait 3 years ago. At that time, he was told that it was dryness and advised to not shower daily. Since then he has been taking showers three times weekly and using Cetaphil once weekly without improvement. He was seen by his pediatrician 6 months ago, diagnosed with xerosis, and was given hydrocortisone 2.5% to use twice daily, again without any improvement.
The rash is not itchy, and he has no oral lesions or nail involvement. Exam revealed lichenoid papules on bilateral dorsal hands and feet, bilateral upper arms, bilateral axilla, lower abdomen, and left upper chest.
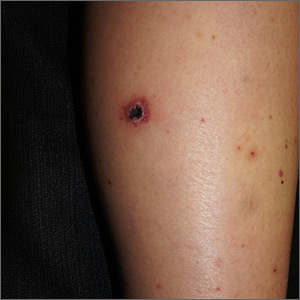
Chin There, Done That—Now What?
ANSWER
The correct answer is to perform a 3-mm punch biopsy (choice “a”).
DISCUSSION
When the history and physical exam fail to point to a clear diagnosis, skin biopsy can answer a very basic question: What is the pathophysiologic process associated with the lesions? The information thus obtained doesn’t always produce a blinking neon sign of a diagnosis, but at a minimum, it can rule out a great number of things, such as cancer or infection.
More often, the types of cells seen, and the patterns in which they are arranged, tell us a great deal. In this case, accumulations of monocytes, macrophages, and activated T-lymphocytes were arranged in circular patterns, but there was no evidence of necrosis (caseation), which can be seen in tubercular granulomas. Stains performed to identify mycobacterial organisms were negative. These findings established the diagnosis of cutaneous sarcoidosis, a granulomatous disease thought to represent a reaction to an unknown antigen (eg, a microorganism) or environmental substance. It is far more common in those with darker skin types.
Although this patient’s disease was confined to the skin, sarcoidosis can affect virtually any other organ in the body—in particular, the lungs, kidneys, liver, nervous system, or even the eyes. For this reason, patients with sarcoidosis are usually referred to pulmonology for a workup intended to rule out systemic involvement and to other specialists as symptoms dictate.
Many cases of sarcoidosis remit on their own, without treatment. When treatment is initiated, the 2 most commonly used medications are systemic glucocorticoids and/or methotrexate. Both drugs have been associated with serious adverse effects and should only be prescribed by experienced providers.
This combination was prescribed for the case patient, who obtained rapid relief. Blood work (complete blood count, comprehensive metabolic panel) was within normal limits prior to and during treatment, and the pulmonologist pronounced her free of lung involvement.
ANSWER
The correct answer is to perform a 3-mm punch biopsy (choice “a”).
DISCUSSION
When the history and physical exam fail to point to a clear diagnosis, skin biopsy can answer a very basic question: What is the pathophysiologic process associated with the lesions? The information thus obtained doesn’t always produce a blinking neon sign of a diagnosis, but at a minimum, it can rule out a great number of things, such as cancer or infection.
More often, the types of cells seen, and the patterns in which they are arranged, tell us a great deal. In this case, accumulations of monocytes, macrophages, and activated T-lymphocytes were arranged in circular patterns, but there was no evidence of necrosis (caseation), which can be seen in tubercular granulomas. Stains performed to identify mycobacterial organisms were negative. These findings established the diagnosis of cutaneous sarcoidosis, a granulomatous disease thought to represent a reaction to an unknown antigen (eg, a microorganism) or environmental substance. It is far more common in those with darker skin types.
Although this patient’s disease was confined to the skin, sarcoidosis can affect virtually any other organ in the body—in particular, the lungs, kidneys, liver, nervous system, or even the eyes. For this reason, patients with sarcoidosis are usually referred to pulmonology for a workup intended to rule out systemic involvement and to other specialists as symptoms dictate.
Many cases of sarcoidosis remit on their own, without treatment. When treatment is initiated, the 2 most commonly used medications are systemic glucocorticoids and/or methotrexate. Both drugs have been associated with serious adverse effects and should only be prescribed by experienced providers.
This combination was prescribed for the case patient, who obtained rapid relief. Blood work (complete blood count, comprehensive metabolic panel) was within normal limits prior to and during treatment, and the pulmonologist pronounced her free of lung involvement.
ANSWER
The correct answer is to perform a 3-mm punch biopsy (choice “a”).
DISCUSSION
When the history and physical exam fail to point to a clear diagnosis, skin biopsy can answer a very basic question: What is the pathophysiologic process associated with the lesions? The information thus obtained doesn’t always produce a blinking neon sign of a diagnosis, but at a minimum, it can rule out a great number of things, such as cancer or infection.
More often, the types of cells seen, and the patterns in which they are arranged, tell us a great deal. In this case, accumulations of monocytes, macrophages, and activated T-lymphocytes were arranged in circular patterns, but there was no evidence of necrosis (caseation), which can be seen in tubercular granulomas. Stains performed to identify mycobacterial organisms were negative. These findings established the diagnosis of cutaneous sarcoidosis, a granulomatous disease thought to represent a reaction to an unknown antigen (eg, a microorganism) or environmental substance. It is far more common in those with darker skin types.
Although this patient’s disease was confined to the skin, sarcoidosis can affect virtually any other organ in the body—in particular, the lungs, kidneys, liver, nervous system, or even the eyes. For this reason, patients with sarcoidosis are usually referred to pulmonology for a workup intended to rule out systemic involvement and to other specialists as symptoms dictate.
Many cases of sarcoidosis remit on their own, without treatment. When treatment is initiated, the 2 most commonly used medications are systemic glucocorticoids and/or methotrexate. Both drugs have been associated with serious adverse effects and should only be prescribed by experienced providers.
This combination was prescribed for the case patient, who obtained rapid relief. Blood work (complete blood count, comprehensive metabolic panel) was within normal limits prior to and during treatment, and the pulmonologist pronounced her free of lung involvement.
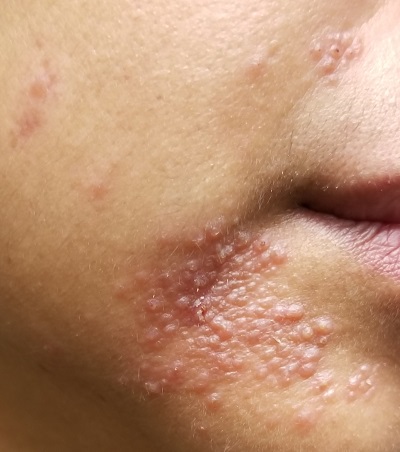
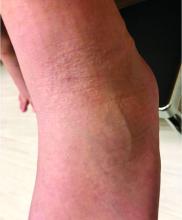
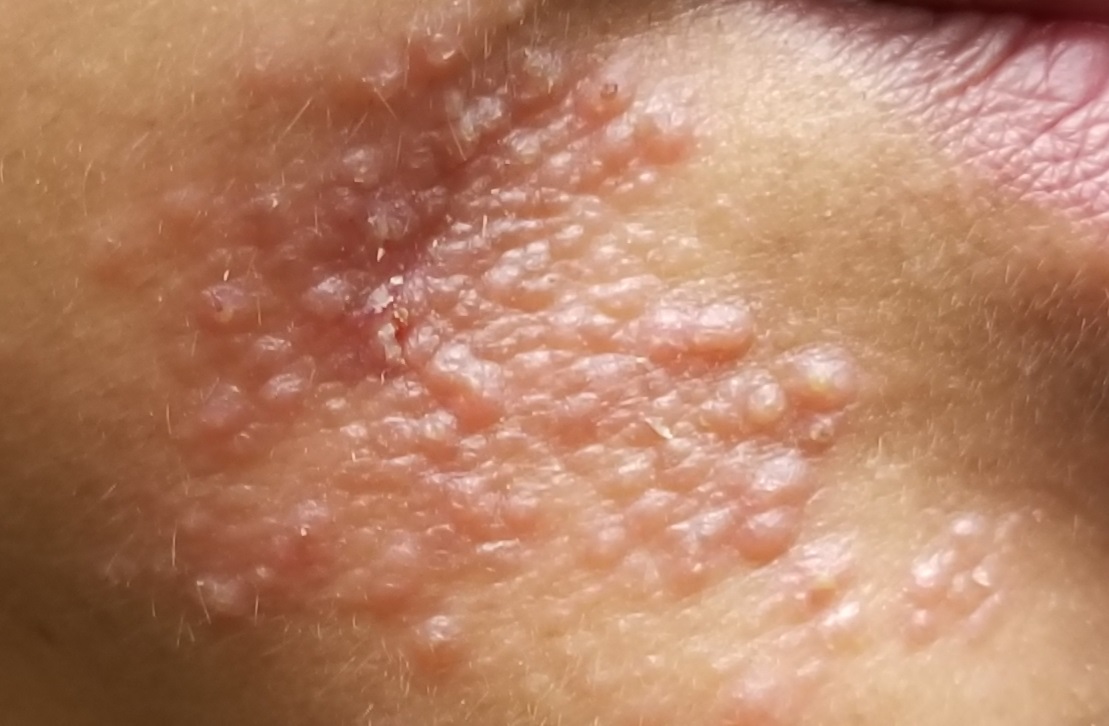
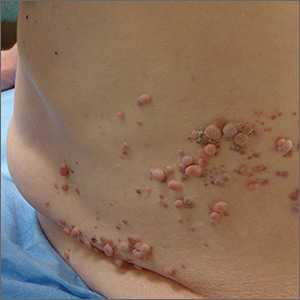
A 51-year-old woman is referred to dermatology for evaluation of a facial rash that has been present, on and off, for years. It usually affects her right chin and perioral area but occasionally manifests with smaller yet similar lesions elsewhere on the face. The lesions are slightly itchy, but the patient’s main concern is their impact on her appearance.
She has consulted several primary care providers over the years, all of whom initially diagnosed and treated for acne—to no avail. This was typically followed by a recommendation to try an OTC topical product, such as an antifungal (tolnaftate and clotrimazole), hydrocortisone 1%, or triple-antibiotic cream—again, without improvement. At no point was a biopsy suggested.
Examination reveals a dense cluster of papules covering a 5×4-cm section of the right chin and perioral area. Although the lesions appear vesicular, they are in fact solid but soft, shiny, and confluent. The papules are about the same color as her type IV skin. There are no comedones or pustules.
No adenopathy is detected in the region. There is no involvement of the adjacent oral mucosal surfaces.
The patient’s skin elsewhere is unremarkable, and in general, she appears to be in good health (certainly in no distress). She claims to be healthy otherwise, with no fever, malaise, joint pain, fatigue, or shortness of breath. No one else in her household is similarly affected.
Frequent lab testing is common, but low-yield, for isotretinoin patients
Abnormalities in lipids, liver enzymes, and blood counts were rare, and
In a review of 1,863 patients receiving isotretinoin, there were no cases of grade 4 abnormalities of lipids, liver enzymes, or complete blood count (CBC). Further, fewer than 1% of patients had grade 2-3 laboratory abnormalities, and no patients had cholesterol or CBC abnormalities of grade 3 or higher.
The retrospective cohort study used an electronic database to identify patients who were prescribed isotretinoin for acne from 2007 to 2017, with inclusion criteria structured to “increase the likelihood of capturing a complete course of isotretinoin therapy,” wrote John Barbieri, MD, and coauthors. The database allowed the investigators to group lab values into baseline testing, and testing by month of therapy for individual deidentified patient records.
Dr. Barbieri, a dermatologist and postdoctoral research fellow at the University of Pennsylvania, Philadelphia, and coinvestigators found that over half of all patients had baseline triglyceride, total cholesterol, AST, ALT, and platelet and white blood cell count levels.
Though the number of patients who had any of these levels checked in a given month of treatment declined over time, as did the total number of patients still on isotretinoin therapy, monthly AST and ALT monitoring occurred in 37.6%-58.5% of patients. Monthly triglyceride monitoring was conducted in between 39.6% and 61.4% of participants, and CBCs were obtained in 26.8%-37.4% of participants.
In terms of the abnormalities that were seen, grade 1 triglyceride elevations of 150-300 mg/dL were present in about 13% of patients at baseline, rising to 39% of participants who were still receiving isotretinoin at month 6. However, grade 2 elevations of up to 500 mg/dL were seen in 1.4% of patients at baseline and 2.4%-5.6% of patients during subsequent months.
Grade 1 liver enzyme abnormalities of less than three times the upper limit of normal values were seen at baseline in under 4% of patients, and in no more than 6.7% of patients through the course of treatment.
Leukopenia of between 3 x 103/mcL and the lower limit of normal occurred in 4.1% of baseline tests and in 6.6%-10.1% of tests in subsequent months. Grade 1 thrombocytopenia (values between 75 x 103/mcL and the lower limit of normal) occurred in 1.9% of baseline tests and no more than 2.9% of tests in the following months.
The results, wrote Dr. Barbieri and coauthors, affirm that most patients fare well on isotretinoin, and frequent laboratory testing is likely to be low-yield. Even using relatively low Medicare reimbursement rates for these tests yielded an estimated $134 in per-patient charges for the studied population. If baseline lipid and liver functions were followed only by repeat testing when peak isotretinoin dose was reached, charges would drop to about $87 per patient. Using the iPLEDGE database figures, this would save $17.4 million in monitoring costs annually, they wrote.
They also calculated that the monitoring regimen they observed puts the cost of detecting one single grade 3 hepatic enzyme elevation at $6,000; one grade 3 triglyceride elevation would cost $7,750.
Of the patients, 49% were female, the median age was 18.2 years, and the median duration of isotretinoin therapy was under 5 months (148 days). Nearly 90% of patients were white and non-Hispanic; 2.5% were black.
The data used for the analysis did not give the investigators access to clinician notes, but they did observe that, even when abnormal test values were seen, isotretinoin prescribing continued. This, they added, pointed toward reassuring clinical scenarios, even in cases of abnormal lab values.
“These findings are consistent with prior studies and suggest that extensive laboratory monitoring observed in this population may be of low value,” concluded Dr. Barbieri and colleagues. “In addition, changes to lipid levels observed in this study typically occurred during the first 2-3 months of therapy before stabilizing, which is consistent with findings in prior studies.”
The investigators noted that, despite mounting evidence of isotretinoin’s safety, there was no trend toward decreased CBC testing over the decade-long period of the study, and there were only “modest” decreases in hepatic enzyme and lipid monitoring. They called for an awareness campaign on the part of professional societies, and consideration for “more specific guideline recommendations” that may ease the testing burden on the adolescent and young adult population receiving isotretinoin.
The study was funded in part by the National Institutes of Health, and Dr. Barbieri receives partial salary support from Pfizer through a grant to the University of Pennsylvania. He has received support for unrelated work from Eli Lilly and Novartis. The other authors reported no conflicts of interest.
SOURCE: Barbieri J et al. J Am Acad Dermatol. 2020 Jan;82(1):72-9.
Abnormalities in lipids, liver enzymes, and blood counts were rare, and
In a review of 1,863 patients receiving isotretinoin, there were no cases of grade 4 abnormalities of lipids, liver enzymes, or complete blood count (CBC). Further, fewer than 1% of patients had grade 2-3 laboratory abnormalities, and no patients had cholesterol or CBC abnormalities of grade 3 or higher.
The retrospective cohort study used an electronic database to identify patients who were prescribed isotretinoin for acne from 2007 to 2017, with inclusion criteria structured to “increase the likelihood of capturing a complete course of isotretinoin therapy,” wrote John Barbieri, MD, and coauthors. The database allowed the investigators to group lab values into baseline testing, and testing by month of therapy for individual deidentified patient records.
Dr. Barbieri, a dermatologist and postdoctoral research fellow at the University of Pennsylvania, Philadelphia, and coinvestigators found that over half of all patients had baseline triglyceride, total cholesterol, AST, ALT, and platelet and white blood cell count levels.
Though the number of patients who had any of these levels checked in a given month of treatment declined over time, as did the total number of patients still on isotretinoin therapy, monthly AST and ALT monitoring occurred in 37.6%-58.5% of patients. Monthly triglyceride monitoring was conducted in between 39.6% and 61.4% of participants, and CBCs were obtained in 26.8%-37.4% of participants.
In terms of the abnormalities that were seen, grade 1 triglyceride elevations of 150-300 mg/dL were present in about 13% of patients at baseline, rising to 39% of participants who were still receiving isotretinoin at month 6. However, grade 2 elevations of up to 500 mg/dL were seen in 1.4% of patients at baseline and 2.4%-5.6% of patients during subsequent months.
Grade 1 liver enzyme abnormalities of less than three times the upper limit of normal values were seen at baseline in under 4% of patients, and in no more than 6.7% of patients through the course of treatment.
Leukopenia of between 3 x 103/mcL and the lower limit of normal occurred in 4.1% of baseline tests and in 6.6%-10.1% of tests in subsequent months. Grade 1 thrombocytopenia (values between 75 x 103/mcL and the lower limit of normal) occurred in 1.9% of baseline tests and no more than 2.9% of tests in the following months.
The results, wrote Dr. Barbieri and coauthors, affirm that most patients fare well on isotretinoin, and frequent laboratory testing is likely to be low-yield. Even using relatively low Medicare reimbursement rates for these tests yielded an estimated $134 in per-patient charges for the studied population. If baseline lipid and liver functions were followed only by repeat testing when peak isotretinoin dose was reached, charges would drop to about $87 per patient. Using the iPLEDGE database figures, this would save $17.4 million in monitoring costs annually, they wrote.
They also calculated that the monitoring regimen they observed puts the cost of detecting one single grade 3 hepatic enzyme elevation at $6,000; one grade 3 triglyceride elevation would cost $7,750.
Of the patients, 49% were female, the median age was 18.2 years, and the median duration of isotretinoin therapy was under 5 months (148 days). Nearly 90% of patients were white and non-Hispanic; 2.5% were black.
The data used for the analysis did not give the investigators access to clinician notes, but they did observe that, even when abnormal test values were seen, isotretinoin prescribing continued. This, they added, pointed toward reassuring clinical scenarios, even in cases of abnormal lab values.
“These findings are consistent with prior studies and suggest that extensive laboratory monitoring observed in this population may be of low value,” concluded Dr. Barbieri and colleagues. “In addition, changes to lipid levels observed in this study typically occurred during the first 2-3 months of therapy before stabilizing, which is consistent with findings in prior studies.”
The investigators noted that, despite mounting evidence of isotretinoin’s safety, there was no trend toward decreased CBC testing over the decade-long period of the study, and there were only “modest” decreases in hepatic enzyme and lipid monitoring. They called for an awareness campaign on the part of professional societies, and consideration for “more specific guideline recommendations” that may ease the testing burden on the adolescent and young adult population receiving isotretinoin.
The study was funded in part by the National Institutes of Health, and Dr. Barbieri receives partial salary support from Pfizer through a grant to the University of Pennsylvania. He has received support for unrelated work from Eli Lilly and Novartis. The other authors reported no conflicts of interest.
SOURCE: Barbieri J et al. J Am Acad Dermatol. 2020 Jan;82(1):72-9.
Abnormalities in lipids, liver enzymes, and blood counts were rare, and
In a review of 1,863 patients receiving isotretinoin, there were no cases of grade 4 abnormalities of lipids, liver enzymes, or complete blood count (CBC). Further, fewer than 1% of patients had grade 2-3 laboratory abnormalities, and no patients had cholesterol or CBC abnormalities of grade 3 or higher.
The retrospective cohort study used an electronic database to identify patients who were prescribed isotretinoin for acne from 2007 to 2017, with inclusion criteria structured to “increase the likelihood of capturing a complete course of isotretinoin therapy,” wrote John Barbieri, MD, and coauthors. The database allowed the investigators to group lab values into baseline testing, and testing by month of therapy for individual deidentified patient records.
Dr. Barbieri, a dermatologist and postdoctoral research fellow at the University of Pennsylvania, Philadelphia, and coinvestigators found that over half of all patients had baseline triglyceride, total cholesterol, AST, ALT, and platelet and white blood cell count levels.
Though the number of patients who had any of these levels checked in a given month of treatment declined over time, as did the total number of patients still on isotretinoin therapy, monthly AST and ALT monitoring occurred in 37.6%-58.5% of patients. Monthly triglyceride monitoring was conducted in between 39.6% and 61.4% of participants, and CBCs were obtained in 26.8%-37.4% of participants.
In terms of the abnormalities that were seen, grade 1 triglyceride elevations of 150-300 mg/dL were present in about 13% of patients at baseline, rising to 39% of participants who were still receiving isotretinoin at month 6. However, grade 2 elevations of up to 500 mg/dL were seen in 1.4% of patients at baseline and 2.4%-5.6% of patients during subsequent months.
Grade 1 liver enzyme abnormalities of less than three times the upper limit of normal values were seen at baseline in under 4% of patients, and in no more than 6.7% of patients through the course of treatment.
Leukopenia of between 3 x 103/mcL and the lower limit of normal occurred in 4.1% of baseline tests and in 6.6%-10.1% of tests in subsequent months. Grade 1 thrombocytopenia (values between 75 x 103/mcL and the lower limit of normal) occurred in 1.9% of baseline tests and no more than 2.9% of tests in the following months.
The results, wrote Dr. Barbieri and coauthors, affirm that most patients fare well on isotretinoin, and frequent laboratory testing is likely to be low-yield. Even using relatively low Medicare reimbursement rates for these tests yielded an estimated $134 in per-patient charges for the studied population. If baseline lipid and liver functions were followed only by repeat testing when peak isotretinoin dose was reached, charges would drop to about $87 per patient. Using the iPLEDGE database figures, this would save $17.4 million in monitoring costs annually, they wrote.
They also calculated that the monitoring regimen they observed puts the cost of detecting one single grade 3 hepatic enzyme elevation at $6,000; one grade 3 triglyceride elevation would cost $7,750.
Of the patients, 49% were female, the median age was 18.2 years, and the median duration of isotretinoin therapy was under 5 months (148 days). Nearly 90% of patients were white and non-Hispanic; 2.5% were black.
The data used for the analysis did not give the investigators access to clinician notes, but they did observe that, even when abnormal test values were seen, isotretinoin prescribing continued. This, they added, pointed toward reassuring clinical scenarios, even in cases of abnormal lab values.
“These findings are consistent with prior studies and suggest that extensive laboratory monitoring observed in this population may be of low value,” concluded Dr. Barbieri and colleagues. “In addition, changes to lipid levels observed in this study typically occurred during the first 2-3 months of therapy before stabilizing, which is consistent with findings in prior studies.”
The investigators noted that, despite mounting evidence of isotretinoin’s safety, there was no trend toward decreased CBC testing over the decade-long period of the study, and there were only “modest” decreases in hepatic enzyme and lipid monitoring. They called for an awareness campaign on the part of professional societies, and consideration for “more specific guideline recommendations” that may ease the testing burden on the adolescent and young adult population receiving isotretinoin.
The study was funded in part by the National Institutes of Health, and Dr. Barbieri receives partial salary support from Pfizer through a grant to the University of Pennsylvania. He has received support for unrelated work from Eli Lilly and Novartis. The other authors reported no conflicts of interest.
SOURCE: Barbieri J et al. J Am Acad Dermatol. 2020 Jan;82(1):72-9.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Rash on legs and abdomen
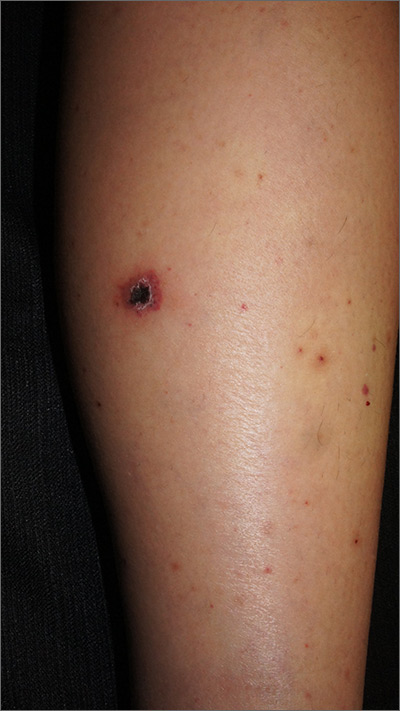
The rash was consistent with nonblanching purpura. Two punch biopsies were performed for hematoxylin and eosin stain and direct immunofluorescence, which were consistent with IgA mediated small vessel vasculitis, or Henoch-Schoenlein purpura.
IgA small vessel vasculitis commonly occurs in children after a transient viral illness or as an allergic reaction to a medication. Nonblanching purpura occurs because small vessels have been cracked open by neutrophils and lymphocytes and leaked blood cells outside of the vascular circulation. Pressure fails to move these cells downstream, and thus, the skin does not blanch.
Joint pain is common, as is a self-resolving IgA mediated nephropathy. Approximately 1% to 3% of children will progress to end stage renal disease. IgA vasculitis also occurs in adults, with a higher portion developing nephropathy. In adults, lesions that present on the abdomen are suspected to correspond with gravity dependency and the total burden of IgA, leading to a higher risk for nephropathy.
The differential diagnosis of purpura is broad, but includes leukocytoclastic vasculitis, antineutrophil cytoplasmic antibodies-associated vasculitis, capillaritis, and disseminated intravascular coagulation.
The patient in this case was given topical triamcinolone 0.1% ointment to treat the rash. She returned 3 weeks later and her blood pressure was 160/105 mm Hg and she had protein and blood in her urine. The FP recommended a renal biopsy, which confirmed severe IgA nephropathy and end stage renal failure. She was given systemic steroids and ultimately received a renal transplant. Her outcome was atypical and unfortunate. Usually IgA vasculitis is benign and self-resolves with rest. Even when nephropathy is present, it typically resolves over weeks to months.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Audemard-Verger A, Terrier B, Dechartres A, et al. French Vasculitis Study Group. Characteristics and management of IgA vasculitis (Henoch-Schönlein) in adults: data from 260 patients included in a French multicenter retrospective survey. Arthritis Rheumatol. 2017;69:1862-1870.

The rash was consistent with nonblanching purpura. Two punch biopsies were performed for hematoxylin and eosin stain and direct immunofluorescence, which were consistent with IgA mediated small vessel vasculitis, or Henoch-Schoenlein purpura.
IgA small vessel vasculitis commonly occurs in children after a transient viral illness or as an allergic reaction to a medication. Nonblanching purpura occurs because small vessels have been cracked open by neutrophils and lymphocytes and leaked blood cells outside of the vascular circulation. Pressure fails to move these cells downstream, and thus, the skin does not blanch.
Joint pain is common, as is a self-resolving IgA mediated nephropathy. Approximately 1% to 3% of children will progress to end stage renal disease. IgA vasculitis also occurs in adults, with a higher portion developing nephropathy. In adults, lesions that present on the abdomen are suspected to correspond with gravity dependency and the total burden of IgA, leading to a higher risk for nephropathy.
The differential diagnosis of purpura is broad, but includes leukocytoclastic vasculitis, antineutrophil cytoplasmic antibodies-associated vasculitis, capillaritis, and disseminated intravascular coagulation.
The patient in this case was given topical triamcinolone 0.1% ointment to treat the rash. She returned 3 weeks later and her blood pressure was 160/105 mm Hg and she had protein and blood in her urine. The FP recommended a renal biopsy, which confirmed severe IgA nephropathy and end stage renal failure. She was given systemic steroids and ultimately received a renal transplant. Her outcome was atypical and unfortunate. Usually IgA vasculitis is benign and self-resolves with rest. Even when nephropathy is present, it typically resolves over weeks to months.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The rash was consistent with nonblanching purpura. Two punch biopsies were performed for hematoxylin and eosin stain and direct immunofluorescence, which were consistent with IgA mediated small vessel vasculitis, or Henoch-Schoenlein purpura.
IgA small vessel vasculitis commonly occurs in children after a transient viral illness or as an allergic reaction to a medication. Nonblanching purpura occurs because small vessels have been cracked open by neutrophils and lymphocytes and leaked blood cells outside of the vascular circulation. Pressure fails to move these cells downstream, and thus, the skin does not blanch.
Joint pain is common, as is a self-resolving IgA mediated nephropathy. Approximately 1% to 3% of children will progress to end stage renal disease. IgA vasculitis also occurs in adults, with a higher portion developing nephropathy. In adults, lesions that present on the abdomen are suspected to correspond with gravity dependency and the total burden of IgA, leading to a higher risk for nephropathy.
The differential diagnosis of purpura is broad, but includes leukocytoclastic vasculitis, antineutrophil cytoplasmic antibodies-associated vasculitis, capillaritis, and disseminated intravascular coagulation.
The patient in this case was given topical triamcinolone 0.1% ointment to treat the rash. She returned 3 weeks later and her blood pressure was 160/105 mm Hg and she had protein and blood in her urine. The FP recommended a renal biopsy, which confirmed severe IgA nephropathy and end stage renal failure. She was given systemic steroids and ultimately received a renal transplant. Her outcome was atypical and unfortunate. Usually IgA vasculitis is benign and self-resolves with rest. Even when nephropathy is present, it typically resolves over weeks to months.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Audemard-Verger A, Terrier B, Dechartres A, et al. French Vasculitis Study Group. Characteristics and management of IgA vasculitis (Henoch-Schönlein) in adults: data from 260 patients included in a French multicenter retrospective survey. Arthritis Rheumatol. 2017;69:1862-1870.
Audemard-Verger A, Terrier B, Dechartres A, et al. French Vasculitis Study Group. Characteristics and management of IgA vasculitis (Henoch-Schönlein) in adults: data from 260 patients included in a French multicenter retrospective survey. Arthritis Rheumatol. 2017;69:1862-1870.
Oral lichen planus prevalence estimates go global
for the general population and 0.98% among clinical patients.
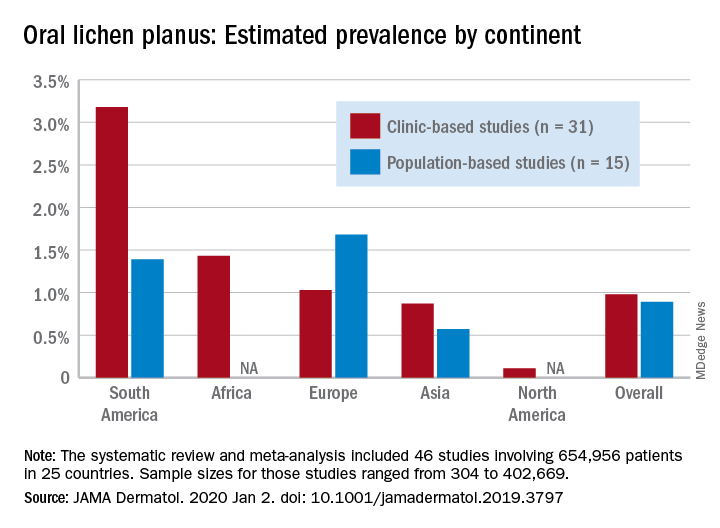
Globally, oral lichen planus (OLP) appears to be more prevalent in women than men (1.55% vs. 1.11% in population-based studies; 1.69% vs. 1.09% in clinic-based), in those aged 40 years and older (1.90% vs. 0.62% in clinic-based studies), and in non-Asian countries (see graph), Changchang Li, MD, and associates reported in JAMA Dermatology.
Of the 25 countries represented among the 46 included studies, Brazil had the highest OLP prevalence at 6.04% and India had the lowest at 0.02%. “Smokers and patients who abuse alcohol have a higher prevalence of OLP. This factor may explain why the highest prevalence … was found in Brazil, where 18.18% of residents report being smokers and 29.09% report consumption of alcoholic beverages,” wrote Dr. Li of the department of dermatology at Zhejiang University of Traditional Chinese Medicine, Wenzhou, China, and associates.
The difference in OLP prevalence by sex may be related to fluctuating female hormone levels, “especially during menstruation or menopause, and that different social roles may lead to the body being in a state of stress,” the investigators suggested.
The age-related difference in OLP could be the result of “longstanding oral habits” or changes to the oral mucosa over time, such as mucosal thinning, decreased elasticity, less saliva secretion, and greater tissue permeability. The higher prevalence among those aged 40 years and older also may be “associated with metabolic changes during aging or with decreased immunity, nutritional deficiencies, medication use, or denture wear,” they wrote.
The review and meta-analysis involved 15 studies (n = 462,993) that included general population data and 31 (n = 191,963) that used information from clinical patients. Sample sizes for those studies ranged from 308 to 402,669.
Statistically significant publication bias was seen among the clinic-based studies but not those that were population based, Dr. Li and associates wrote, adding that “our findings should be considered with caution because of the high heterogeneity of the included studies.”
The study was funded by the First-Class Discipline Construction Foundation of Guangzhou University of Chinese Medicine, the Young Top Talent Project of Scientific and Technological Innovation in Special Support Plan for Training High-level Talents, and the Youth Research and Cultivation Project of Guangzhou University of Chinese Medicine. The investigators did not report any conflicts of interest.
SOURCE: C Li et al. JAMA Dermatol. 2020 Jan 2. doi: 10.1001/jamadermatol.2019.3797.
for the general population and 0.98% among clinical patients.

Globally, oral lichen planus (OLP) appears to be more prevalent in women than men (1.55% vs. 1.11% in population-based studies; 1.69% vs. 1.09% in clinic-based), in those aged 40 years and older (1.90% vs. 0.62% in clinic-based studies), and in non-Asian countries (see graph), Changchang Li, MD, and associates reported in JAMA Dermatology.
Of the 25 countries represented among the 46 included studies, Brazil had the highest OLP prevalence at 6.04% and India had the lowest at 0.02%. “Smokers and patients who abuse alcohol have a higher prevalence of OLP. This factor may explain why the highest prevalence … was found in Brazil, where 18.18% of residents report being smokers and 29.09% report consumption of alcoholic beverages,” wrote Dr. Li of the department of dermatology at Zhejiang University of Traditional Chinese Medicine, Wenzhou, China, and associates.
The difference in OLP prevalence by sex may be related to fluctuating female hormone levels, “especially during menstruation or menopause, and that different social roles may lead to the body being in a state of stress,” the investigators suggested.
The age-related difference in OLP could be the result of “longstanding oral habits” or changes to the oral mucosa over time, such as mucosal thinning, decreased elasticity, less saliva secretion, and greater tissue permeability. The higher prevalence among those aged 40 years and older also may be “associated with metabolic changes during aging or with decreased immunity, nutritional deficiencies, medication use, or denture wear,” they wrote.
The review and meta-analysis involved 15 studies (n = 462,993) that included general population data and 31 (n = 191,963) that used information from clinical patients. Sample sizes for those studies ranged from 308 to 402,669.
Statistically significant publication bias was seen among the clinic-based studies but not those that were population based, Dr. Li and associates wrote, adding that “our findings should be considered with caution because of the high heterogeneity of the included studies.”
The study was funded by the First-Class Discipline Construction Foundation of Guangzhou University of Chinese Medicine, the Young Top Talent Project of Scientific and Technological Innovation in Special Support Plan for Training High-level Talents, and the Youth Research and Cultivation Project of Guangzhou University of Chinese Medicine. The investigators did not report any conflicts of interest.
SOURCE: C Li et al. JAMA Dermatol. 2020 Jan 2. doi: 10.1001/jamadermatol.2019.3797.
for the general population and 0.98% among clinical patients.

Globally, oral lichen planus (OLP) appears to be more prevalent in women than men (1.55% vs. 1.11% in population-based studies; 1.69% vs. 1.09% in clinic-based), in those aged 40 years and older (1.90% vs. 0.62% in clinic-based studies), and in non-Asian countries (see graph), Changchang Li, MD, and associates reported in JAMA Dermatology.
Of the 25 countries represented among the 46 included studies, Brazil had the highest OLP prevalence at 6.04% and India had the lowest at 0.02%. “Smokers and patients who abuse alcohol have a higher prevalence of OLP. This factor may explain why the highest prevalence … was found in Brazil, where 18.18% of residents report being smokers and 29.09% report consumption of alcoholic beverages,” wrote Dr. Li of the department of dermatology at Zhejiang University of Traditional Chinese Medicine, Wenzhou, China, and associates.
The difference in OLP prevalence by sex may be related to fluctuating female hormone levels, “especially during menstruation or menopause, and that different social roles may lead to the body being in a state of stress,” the investigators suggested.
The age-related difference in OLP could be the result of “longstanding oral habits” or changes to the oral mucosa over time, such as mucosal thinning, decreased elasticity, less saliva secretion, and greater tissue permeability. The higher prevalence among those aged 40 years and older also may be “associated with metabolic changes during aging or with decreased immunity, nutritional deficiencies, medication use, or denture wear,” they wrote.
The review and meta-analysis involved 15 studies (n = 462,993) that included general population data and 31 (n = 191,963) that used information from clinical patients. Sample sizes for those studies ranged from 308 to 402,669.
Statistically significant publication bias was seen among the clinic-based studies but not those that were population based, Dr. Li and associates wrote, adding that “our findings should be considered with caution because of the high heterogeneity of the included studies.”
The study was funded by the First-Class Discipline Construction Foundation of Guangzhou University of Chinese Medicine, the Young Top Talent Project of Scientific and Technological Innovation in Special Support Plan for Training High-level Talents, and the Youth Research and Cultivation Project of Guangzhou University of Chinese Medicine. The investigators did not report any conflicts of interest.
SOURCE: C Li et al. JAMA Dermatol. 2020 Jan 2. doi: 10.1001/jamadermatol.2019.3797.
FROM JAMA DERMATOLOGY
Experts in Europe issue guidance on atopic dermatitis in pregnancy
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
Jenny Murase, MD, is with the department of dermatology, University of California, San Francisco, and is the director of medical consultative dermatology at the Palo Alto Foundation Medical Group, Mountain View, Calif. She has served on advisory boards for Dermira, Sanofi, and UCB; performed dermatologic consulting for UpToDate and Ferndale, and given nonbranded lectures for disease state management awareness for Regeneron and UCB.
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
MADRID – European atopic dermatitis experts have issued formal guidance on a seriously neglected topic: treatment of the disease during pregnancy, breastfeeding, and in men planning to father children.
The impetus for the project was clear: “,” Christian Vestergaard, MD, PhD, declared at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
He presented highlights of the task force’s position paper on the topic, for which he served as first author. The group’s recommendations are based on expert opinion, since randomized clinical trial literature in this area is nonexistent because of ethical concerns. But the task force, comprising a who’s who in European dermatology, drew on a wealth of collective clinical experience in this area.
“We have all of Europe involved in doing this position statement. It’s meant as what we think is proper treatment and what we can say about the different drugs,” explained Dr. Vestergaard, a dermatologist at the University of Aarhus (Denmark).
Most nonobstetricians are intimidated by atopic dermatitis (AD) in pregnancy, and are concerned about the potential for treatment-related harm to the fetus. As a consequence, they are reluctant to recommend anything beyond weak class I topical corticosteroids and emollients. That’s clearly insufficient in light of the vast scope of need, he asserted. After all, AD affects 15%-20% of all children and persists or reappears in adulthood in one out of five of them. Half of those adults are women, many of whom will at some point wish to become pregnant. And many men with AD will eventually want to father children.
A key message from the task force is that untreated AD in pregnancy potentially places the mother and fetus at risk of serious complications, including Staphylococcus aureus infection and eczema herpeticum.
“If you take one thing away from our position paper, it’s that you can use class II or III topical corticosteroids in pregnant women as first-line therapy,” Dr. Vestergaard said.
This stance contradicts a longstanding widely held concern that topical steroids in pregnancy might increase the risk of facial cleft in the offspring, a worry that has been convincingly debunked in a Cochrane systematic review of 14 studies including more than 1.6 million pregnancies. The report concluded there was no association between topical corticosteroids of any potency with preterm delivery, birth defects, or low Apgar scores (Cochrane Database Syst Rev. 2015 Oct 26. doi: 10.1002/14651858.CD007346.pub3).
The task force recommends that if class II or III topical corticosteroid use in pregnancy exceeds 200 g/month, it’s worth considering add-on UV therapy, with narrow band UVB-311 nm as the regimen of choice; it can be used liberally. UV therapy with psoralens is not advised because of a theoretical risk of mutagenicity.
Product labeling for the topical calcineurin inhibitors declares that the agents should not be used during pregnancy. However, the European task force position paper takes issue with that and declares that topical tacrolimus (Protopic) can be considered an off-label first-line therapy in pregnant women with an insufficient response to liberal use of emollients. The same holds true for breastfeeding patients with AD. Just as when topical corticosteroids are used in the nipple area, topical tacrolimus should be applied after nursing, and the nipple area should be gently cleaned before nursing.
The rationale behind recommending topical tacrolimus as a first-line treatment is that systemic absorption of the drug is trivial. Plus, observational studies of oral tacrolimus in pregnant women who have received a solid organ transplant have shown no increase in congenital malformations.
The task force recommends against the use of topical pimecrolimus (Elidel) or crisaborole (Eucrisa) in pregnancy or lactation due to lack of clinical experience in these settings, Dr. Vestergaard continued.
The task force position is that chlorhexidine and other topical antiseptics – with the notable exception of triclosan – can be used in pregnancy to prevent recurrent skin infections. Aminoglycosides should be avoided, but topical fusidic acid is a reasonable antibiotic for treatment of small areas of clinically infected atopic dermatitis in pregnancy.
Systemic therapies
If disease control is insufficient with topical therapy, it’s appropriate to engage in shared decision-making with the patient regarding systemic treatment. She needs to understand up front that the worldwide overall background stillbirth rate in the general population is about 3%, and that severe congenital malformations are present in up to 6% of all live births.
“You need to inform them that they can have systemic therapy and give birth to a child with congenital defects which have nothing to do with the medication,” noted Dr. Vestergaard.
That said, the task force recommends cyclosporine as the off-label, first-line systemic therapy in pregnancy and lactation when long-term treatment is required. This guidance is based largely upon reassuring evidence in solid organ transplant recipients.
The recommended second-line therapy is systemic corticosteroids, but it’s a qualified recommendation. Dr. Vestergaard and colleagues find that systemic corticosteroid therapy is only rarely needed in pregnant AD patients, and the task force recommendation is to limit the use to less than 2-3 weeks and no more than 0.5 mg/kg per day of prednisone. Dexamethasone is not recommended.
Azathioprine should not be started in pregnancy, according to the task force, but when no other options are available, it may be continued in women already on the drug, albeit at half of the prepregnancy dose.
Dupilumab (Dupixent) is to be avoided in pregnant women with AD until more clinical experience becomes available.
Treatment of prospective fathers with AD
The European task force recommends that topical therapies can be prescribed in prospective fathers without any special concerns. The same is true for systemic corticosteroids. Methotrexate should be halted 3 months before planned pregnancy, as is the case for mycophenolate mofetil (CellCept). Azathioprine is recommended when other options have failed. Cyclosporine is deemed a reasonable option in the treatment of men with severe AD at the time of conception if other treatments have failed; of note, neither the Food and Drug Administration nor the European regulatory agency have issued contraindications for the use of the drug in men who wish to become fathers.
Mycophenolate mofetil carries a theoretical risk of teratogenicity. The European task force recommends that men should use condoms while on the drug and for at least 90 days afterward.
Unplanned pregnancy in women on systemic therapy
The recommended course of action is to immediately stop systemic therapy, intensify appropriate topical therapy in anticipation of worsening AD, and refer the patient to an obstetrician and a teratology information center for an individualized risk assessment. Methotrexate and mycophenolate mofetil are known teratogens.
The full 16-page task force position paper was published shortly before EADV 2019 (J Eur Acad Dermatol Venereol. 2019 Sep;33[9]:1644-59).
The report was developed without commercial sponsorship. Dr. Vestergaard indicated he has received research grants from and/or serves as a consultant to eight pharmaceutical companies.
REPORTING FROM THE EADV CONGRESS
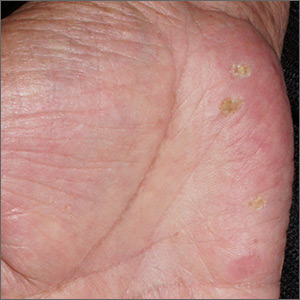
Papules on hands and soles
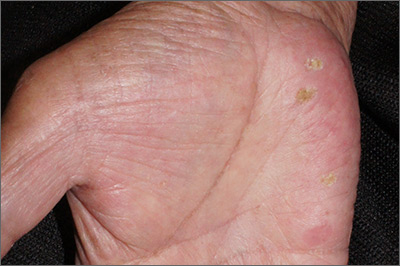
Although these lesions were consistent with actinic keratosis, the location on the palms and the patient’s history of growing up on a ranch in northern Mexico (where he said he was exposed to high arsenic levels in the water and soil), suggested that this was a case of arsenical keratosis. A biopsy excluded frank squamous cell carcinoma; further testing was not needed to make the diagnosis.
While both actinic and arsenical keratoses are precancerous growths with delayed presentations of 20 to 30 years, arsenical keratoses do not typically appear in sun-exposed areas. Rather, these lesions appear on a patient’s palms or soles. They also may present as plate-like hyperpigmentation of the palms and soles or corn-like keratotic papules.
Arsenic occurs naturally in soil and groundwater worldwide and acts as a toxin and carcinogen. It accumulates in the skin, hair, nails, and teeth—making these sites markers of chronic disease. Chronic exposure leads to skin and lung cancer. Cancer of other organs also is possible. Peripheral neuropathy and pancytopenia are hallmarks of chronic exposure.
If arsenical keratosis is suspected, testing for arsenic levels in water or in the work environment is recommended. That said, arsenic exposure is often remote (given the time that has elapsed) and hard to prove.
A blood count and renal and liver function tests should be considered to assess for organ damage. In this case, these tests were normal and the patient was treated with cryotherapy and monitored for recurrence with annual skin exams. Oral retinoids, such as acitretin, have had success in suppressing the frequency and severity of episodes of new lesions in small case reports.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
US Department of Health and Human Services Agency for Toxic Substances and Disease Registry. Arsenic Toxicity. https://www.atsdr.cdc.gov/csem/csem.asp?csem=1&po=0. Updated January 15, 2010. Accessed January 2, 2020.

Although these lesions were consistent with actinic keratosis, the location on the palms and the patient’s history of growing up on a ranch in northern Mexico (where he said he was exposed to high arsenic levels in the water and soil), suggested that this was a case of arsenical keratosis. A biopsy excluded frank squamous cell carcinoma; further testing was not needed to make the diagnosis.
While both actinic and arsenical keratoses are precancerous growths with delayed presentations of 20 to 30 years, arsenical keratoses do not typically appear in sun-exposed areas. Rather, these lesions appear on a patient’s palms or soles. They also may present as plate-like hyperpigmentation of the palms and soles or corn-like keratotic papules.
Arsenic occurs naturally in soil and groundwater worldwide and acts as a toxin and carcinogen. It accumulates in the skin, hair, nails, and teeth—making these sites markers of chronic disease. Chronic exposure leads to skin and lung cancer. Cancer of other organs also is possible. Peripheral neuropathy and pancytopenia are hallmarks of chronic exposure.
If arsenical keratosis is suspected, testing for arsenic levels in water or in the work environment is recommended. That said, arsenic exposure is often remote (given the time that has elapsed) and hard to prove.
A blood count and renal and liver function tests should be considered to assess for organ damage. In this case, these tests were normal and the patient was treated with cryotherapy and monitored for recurrence with annual skin exams. Oral retinoids, such as acitretin, have had success in suppressing the frequency and severity of episodes of new lesions in small case reports.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Although these lesions were consistent with actinic keratosis, the location on the palms and the patient’s history of growing up on a ranch in northern Mexico (where he said he was exposed to high arsenic levels in the water and soil), suggested that this was a case of arsenical keratosis. A biopsy excluded frank squamous cell carcinoma; further testing was not needed to make the diagnosis.
While both actinic and arsenical keratoses are precancerous growths with delayed presentations of 20 to 30 years, arsenical keratoses do not typically appear in sun-exposed areas. Rather, these lesions appear on a patient’s palms or soles. They also may present as plate-like hyperpigmentation of the palms and soles or corn-like keratotic papules.
Arsenic occurs naturally in soil and groundwater worldwide and acts as a toxin and carcinogen. It accumulates in the skin, hair, nails, and teeth—making these sites markers of chronic disease. Chronic exposure leads to skin and lung cancer. Cancer of other organs also is possible. Peripheral neuropathy and pancytopenia are hallmarks of chronic exposure.
If arsenical keratosis is suspected, testing for arsenic levels in water or in the work environment is recommended. That said, arsenic exposure is often remote (given the time that has elapsed) and hard to prove.
A blood count and renal and liver function tests should be considered to assess for organ damage. In this case, these tests were normal and the patient was treated with cryotherapy and monitored for recurrence with annual skin exams. Oral retinoids, such as acitretin, have had success in suppressing the frequency and severity of episodes of new lesions in small case reports.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
US Department of Health and Human Services Agency for Toxic Substances and Disease Registry. Arsenic Toxicity. https://www.atsdr.cdc.gov/csem/csem.asp?csem=1&po=0. Updated January 15, 2010. Accessed January 2, 2020.
US Department of Health and Human Services Agency for Toxic Substances and Disease Registry. Arsenic Toxicity. https://www.atsdr.cdc.gov/csem/csem.asp?csem=1&po=0. Updated January 15, 2010. Accessed January 2, 2020.
Dupilumab-induced head and neck erythema described in atopic dermatitis patients
MADRID – A growing recognition that from their background skin disease emerged as a hot topic of discussion at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“During treatment with dupilumab, we saw something that is really different from the classic eczema that patients experienced prior to dupilumab, with no or minimal scaling, itch, or burning sensation. We do not believe this is a delayed effect of dupilumab on that specific region. We think this is a dupilumab-induced entity that we’re looking at. You should take home, in my opinion, that this is a common side effect that’s underreported in daily practice at this moment, and it’s not reported in clinical trials at all,” said Linde de Wijs, MD, of the department of dermatology, Erasmus University Medical Center in Rotterdam, the Netherlands.
She presented a detailed case series of seven affected patients which included histologic examination of lesional skin biopsies. The biopsies were characterized by a perivascular lymphohistiocytic infiltrate, an increase in ectatic capillaries in the papillary dermis, and a notable dearth of spongiosis, eosinophils, and neutrophils. Four patients had bulbous elongated rete ridges evocative of a psoriasiform dermatitis. The overall histologic picture was suggestive of a drug-induced skin reaction.
A striking finding was that, even though AD patients typically place high importance on achieving total clearing of disease on the head and neck, these seven closely studied patients nonetheless rated their treatment satisfaction as 9 out of a possible 10 points. Dr. de Wijs interpreted this as testimony to dupilumab’s potent efficacy and comparatively acceptable safety profile, especially the apparent side effect’s absence of scaling and itch.
“Remember, these are patients with really severe atopic dermatitis who’ve been treated with a lot of immunosuppressants prior to dupilumab,” she said.
Once the investigators began to suspect the existence of a novel dupilumab-induced skin reaction, they conducted a retrospective chart review of more than 150 patients treated with dupilumab (Dupixent) and determined that roughly 30% had developed this distinctive sharply demarcated patchy erythema on the head and neck characterized by absence of itch. The sequence involved clearance of the AD in response to dupilumab, followed by gradual development of the head and neck erythema 10-39 weeks after the start of treatment.
The erythema proved treatment refractory. Dr. de Wijs and her colleagues tried topical corticosteroids, including potent ones, as well as topical tacrolimus, antifungals, antibiotics, emollients, oral steroids, and antihistamines, to no avail. Patch testing to investigate allergic contact dermatitis as a possible etiology was unremarkable.
She hypothesized that, since dupilumab blocks the key signaling pathways for Th2 T-cell differentiation by targeting the interleukin-4 receptor alpha, it’s possible that the biologic promotes a shift towards activation of the Th17 pathway, which might explain the observed histologic findings.
The fact that this erythema wasn’t reported in the major randomized clinical trials of dupilumab underscores the enormous value of clinical practice registries, she said.
“We are not the only ones observing this phenomenon,” noted Dr. de Wijs, citing recently published reports by other investigators (J Am Acad Dermatol. 2020 Jan;82[1]:230-2; JAMA Dermatol. 2019 Jul 1;155[7]:850-2).
Indeed, her talk was immediately followed by a presentation by Sebastien Barbarot, MD, PhD, who reported on a French national retrospective study of head and neck dermatitis arising in patients on dupilumab that was conducted by the French Atopic Dermatitis Network using the organization’s GREAT database. Among 1,000 adult patients with AD treated with the biologic at 29 French centers, 10 developed a de novo head and neck dermatitis, and 32 others experienced more than 50% worsening of eczema signs on the head and neck from baseline beginning about 2 months after starting on dupilumab.
This 4.2% incidence is probably an underestimate, since dermatologists weren’t aware of the phenomenon and didn’t specifically ask patients about it, observed Dr. Barbarot, a dermatologist at the University of Nantes (France).
Among the key findings: No differences in clinical characteristics were found between the de novo and exacerbation groups, nearly half of affected patients had concomitant conjunctivitis, and seven patients discontinued dupilumab because of an intolerable burning sensation on the head/neck.
“I think this condition is quite different from rosacea,” Dr. Barbarot emphasized.
French dermatologists generally turned to topical corticosteroids or topical tacrolimus to treat the face and neck dermatitis, with mixed results; 22 of the 42 patients showed improvement and 8 worsened.
Marjolein de Bruin-Weller, MD, PhD, a dermatologist at Utrecht (the Netherlands) University and head of the Dutch National Eczema Expertise Center, said she and her colleagues have also encountered this dupilumab-related head and neck erythema and are convinced that a subset of affected patients have Malassezia-induced dermatitis with neutrophils present on lesional biopsies. “It responds very well to treatment. I think it’s very important to try itraconazole because sometimes it works,” she said.
Dr. de Wijs replied that she and her coworkers tried 2 weeks of itraconazole in several patients, with no effect. And none of their seven biopsied patients had an increase in neutrophils.
“It might be a very heterogenous polyform entity that we’re now observing,” she commented. Dr. de Bruin-Weller concurred.
Dr. Barbarot said he’d be interested in a formal study of antifungal therapy in patients with dupilumab-related head and neck dermatitis. Mechanistically, it seems plausible that dupilumab-induced activation of the TH17 pathway might lead to proliferation of Malassezia fungus.
Dr. de Wijs and Dr. Barbarot reported having no financial conflicts regarding their respective studies, which were conducted free of commercial sponsorship.
MADRID – A growing recognition that from their background skin disease emerged as a hot topic of discussion at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“During treatment with dupilumab, we saw something that is really different from the classic eczema that patients experienced prior to dupilumab, with no or minimal scaling, itch, or burning sensation. We do not believe this is a delayed effect of dupilumab on that specific region. We think this is a dupilumab-induced entity that we’re looking at. You should take home, in my opinion, that this is a common side effect that’s underreported in daily practice at this moment, and it’s not reported in clinical trials at all,” said Linde de Wijs, MD, of the department of dermatology, Erasmus University Medical Center in Rotterdam, the Netherlands.
She presented a detailed case series of seven affected patients which included histologic examination of lesional skin biopsies. The biopsies were characterized by a perivascular lymphohistiocytic infiltrate, an increase in ectatic capillaries in the papillary dermis, and a notable dearth of spongiosis, eosinophils, and neutrophils. Four patients had bulbous elongated rete ridges evocative of a psoriasiform dermatitis. The overall histologic picture was suggestive of a drug-induced skin reaction.
A striking finding was that, even though AD patients typically place high importance on achieving total clearing of disease on the head and neck, these seven closely studied patients nonetheless rated their treatment satisfaction as 9 out of a possible 10 points. Dr. de Wijs interpreted this as testimony to dupilumab’s potent efficacy and comparatively acceptable safety profile, especially the apparent side effect’s absence of scaling and itch.
“Remember, these are patients with really severe atopic dermatitis who’ve been treated with a lot of immunosuppressants prior to dupilumab,” she said.
Once the investigators began to suspect the existence of a novel dupilumab-induced skin reaction, they conducted a retrospective chart review of more than 150 patients treated with dupilumab (Dupixent) and determined that roughly 30% had developed this distinctive sharply demarcated patchy erythema on the head and neck characterized by absence of itch. The sequence involved clearance of the AD in response to dupilumab, followed by gradual development of the head and neck erythema 10-39 weeks after the start of treatment.
The erythema proved treatment refractory. Dr. de Wijs and her colleagues tried topical corticosteroids, including potent ones, as well as topical tacrolimus, antifungals, antibiotics, emollients, oral steroids, and antihistamines, to no avail. Patch testing to investigate allergic contact dermatitis as a possible etiology was unremarkable.
She hypothesized that, since dupilumab blocks the key signaling pathways for Th2 T-cell differentiation by targeting the interleukin-4 receptor alpha, it’s possible that the biologic promotes a shift towards activation of the Th17 pathway, which might explain the observed histologic findings.
The fact that this erythema wasn’t reported in the major randomized clinical trials of dupilumab underscores the enormous value of clinical practice registries, she said.
“We are not the only ones observing this phenomenon,” noted Dr. de Wijs, citing recently published reports by other investigators (J Am Acad Dermatol. 2020 Jan;82[1]:230-2; JAMA Dermatol. 2019 Jul 1;155[7]:850-2).
Indeed, her talk was immediately followed by a presentation by Sebastien Barbarot, MD, PhD, who reported on a French national retrospective study of head and neck dermatitis arising in patients on dupilumab that was conducted by the French Atopic Dermatitis Network using the organization’s GREAT database. Among 1,000 adult patients with AD treated with the biologic at 29 French centers, 10 developed a de novo head and neck dermatitis, and 32 others experienced more than 50% worsening of eczema signs on the head and neck from baseline beginning about 2 months after starting on dupilumab.
This 4.2% incidence is probably an underestimate, since dermatologists weren’t aware of the phenomenon and didn’t specifically ask patients about it, observed Dr. Barbarot, a dermatologist at the University of Nantes (France).
Among the key findings: No differences in clinical characteristics were found between the de novo and exacerbation groups, nearly half of affected patients had concomitant conjunctivitis, and seven patients discontinued dupilumab because of an intolerable burning sensation on the head/neck.
“I think this condition is quite different from rosacea,” Dr. Barbarot emphasized.
French dermatologists generally turned to topical corticosteroids or topical tacrolimus to treat the face and neck dermatitis, with mixed results; 22 of the 42 patients showed improvement and 8 worsened.
Marjolein de Bruin-Weller, MD, PhD, a dermatologist at Utrecht (the Netherlands) University and head of the Dutch National Eczema Expertise Center, said she and her colleagues have also encountered this dupilumab-related head and neck erythema and are convinced that a subset of affected patients have Malassezia-induced dermatitis with neutrophils present on lesional biopsies. “It responds very well to treatment. I think it’s very important to try itraconazole because sometimes it works,” she said.
Dr. de Wijs replied that she and her coworkers tried 2 weeks of itraconazole in several patients, with no effect. And none of their seven biopsied patients had an increase in neutrophils.
“It might be a very heterogenous polyform entity that we’re now observing,” she commented. Dr. de Bruin-Weller concurred.
Dr. Barbarot said he’d be interested in a formal study of antifungal therapy in patients with dupilumab-related head and neck dermatitis. Mechanistically, it seems plausible that dupilumab-induced activation of the TH17 pathway might lead to proliferation of Malassezia fungus.
Dr. de Wijs and Dr. Barbarot reported having no financial conflicts regarding their respective studies, which were conducted free of commercial sponsorship.
MADRID – A growing recognition that from their background skin disease emerged as a hot topic of discussion at a meeting of the European Task Force on Atopic Dermatitis held in conjunction with the annual congress of the European Academy of Dermatology and Venereology.
“During treatment with dupilumab, we saw something that is really different from the classic eczema that patients experienced prior to dupilumab, with no or minimal scaling, itch, or burning sensation. We do not believe this is a delayed effect of dupilumab on that specific region. We think this is a dupilumab-induced entity that we’re looking at. You should take home, in my opinion, that this is a common side effect that’s underreported in daily practice at this moment, and it’s not reported in clinical trials at all,” said Linde de Wijs, MD, of the department of dermatology, Erasmus University Medical Center in Rotterdam, the Netherlands.
She presented a detailed case series of seven affected patients which included histologic examination of lesional skin biopsies. The biopsies were characterized by a perivascular lymphohistiocytic infiltrate, an increase in ectatic capillaries in the papillary dermis, and a notable dearth of spongiosis, eosinophils, and neutrophils. Four patients had bulbous elongated rete ridges evocative of a psoriasiform dermatitis. The overall histologic picture was suggestive of a drug-induced skin reaction.
A striking finding was that, even though AD patients typically place high importance on achieving total clearing of disease on the head and neck, these seven closely studied patients nonetheless rated their treatment satisfaction as 9 out of a possible 10 points. Dr. de Wijs interpreted this as testimony to dupilumab’s potent efficacy and comparatively acceptable safety profile, especially the apparent side effect’s absence of scaling and itch.
“Remember, these are patients with really severe atopic dermatitis who’ve been treated with a lot of immunosuppressants prior to dupilumab,” she said.
Once the investigators began to suspect the existence of a novel dupilumab-induced skin reaction, they conducted a retrospective chart review of more than 150 patients treated with dupilumab (Dupixent) and determined that roughly 30% had developed this distinctive sharply demarcated patchy erythema on the head and neck characterized by absence of itch. The sequence involved clearance of the AD in response to dupilumab, followed by gradual development of the head and neck erythema 10-39 weeks after the start of treatment.
The erythema proved treatment refractory. Dr. de Wijs and her colleagues tried topical corticosteroids, including potent ones, as well as topical tacrolimus, antifungals, antibiotics, emollients, oral steroids, and antihistamines, to no avail. Patch testing to investigate allergic contact dermatitis as a possible etiology was unremarkable.
She hypothesized that, since dupilumab blocks the key signaling pathways for Th2 T-cell differentiation by targeting the interleukin-4 receptor alpha, it’s possible that the biologic promotes a shift towards activation of the Th17 pathway, which might explain the observed histologic findings.
The fact that this erythema wasn’t reported in the major randomized clinical trials of dupilumab underscores the enormous value of clinical practice registries, she said.
“We are not the only ones observing this phenomenon,” noted Dr. de Wijs, citing recently published reports by other investigators (J Am Acad Dermatol. 2020 Jan;82[1]:230-2; JAMA Dermatol. 2019 Jul 1;155[7]:850-2).
Indeed, her talk was immediately followed by a presentation by Sebastien Barbarot, MD, PhD, who reported on a French national retrospective study of head and neck dermatitis arising in patients on dupilumab that was conducted by the French Atopic Dermatitis Network using the organization’s GREAT database. Among 1,000 adult patients with AD treated with the biologic at 29 French centers, 10 developed a de novo head and neck dermatitis, and 32 others experienced more than 50% worsening of eczema signs on the head and neck from baseline beginning about 2 months after starting on dupilumab.
This 4.2% incidence is probably an underestimate, since dermatologists weren’t aware of the phenomenon and didn’t specifically ask patients about it, observed Dr. Barbarot, a dermatologist at the University of Nantes (France).
Among the key findings: No differences in clinical characteristics were found between the de novo and exacerbation groups, nearly half of affected patients had concomitant conjunctivitis, and seven patients discontinued dupilumab because of an intolerable burning sensation on the head/neck.
“I think this condition is quite different from rosacea,” Dr. Barbarot emphasized.
French dermatologists generally turned to topical corticosteroids or topical tacrolimus to treat the face and neck dermatitis, with mixed results; 22 of the 42 patients showed improvement and 8 worsened.
Marjolein de Bruin-Weller, MD, PhD, a dermatologist at Utrecht (the Netherlands) University and head of the Dutch National Eczema Expertise Center, said she and her colleagues have also encountered this dupilumab-related head and neck erythema and are convinced that a subset of affected patients have Malassezia-induced dermatitis with neutrophils present on lesional biopsies. “It responds very well to treatment. I think it’s very important to try itraconazole because sometimes it works,” she said.
Dr. de Wijs replied that she and her coworkers tried 2 weeks of itraconazole in several patients, with no effect. And none of their seven biopsied patients had an increase in neutrophils.
“It might be a very heterogenous polyform entity that we’re now observing,” she commented. Dr. de Bruin-Weller concurred.
Dr. Barbarot said he’d be interested in a formal study of antifungal therapy in patients with dupilumab-related head and neck dermatitis. Mechanistically, it seems plausible that dupilumab-induced activation of the TH17 pathway might lead to proliferation of Malassezia fungus.
Dr. de Wijs and Dr. Barbarot reported having no financial conflicts regarding their respective studies, which were conducted free of commercial sponsorship.
REPORTING FROM THE EADV CONGRESS
Efficacy and safety of lowering dupilumab frequency for AD
Patients with moderate to severe atopic dermatitis who responded well to the approved dupilumab regimen of 300 mg every 2 weeks in pivotal phase 3 monotherapy trials were more likely to have a continued response over the longer term if they maintained this regimen rather than switching to longer dosing intervals or discontinuing the medication.
This finding comes from a 36-week, randomized, double-blind, placebo-controlled trial that enrolled 422 – SOLO 1 and SOLO 2.
The new international study – SOLO-CONTINUE – randomized these patients to continue the original regimen (weekly or every 2 weeks), to receive 300 mg of the biologic medication every 4 or 8 weeks, or to receive placebo.
Patients who continued the original regimen had the most consistent maintenance of treatment effect, while patients on longer dosage intervals or placebo had a dose-dependent reduction in response and no safety advantage. The incidence of treatment-emergent antidrug antibody was lowest with dupilumab weekly or every 2 weeks, and slightly higher with less-frequent dosing intervals, reported Margitta Worm, MD, of the Charité Universitätsmedizin Berlin, and coinvestigators.
“Because administration every 4 weeks or every 8 weeks did not provide an additional safety advantage and was numerically outperformed by administration weekly or every 2 weeks, we believe that it is prudent to adhere to the approved every 2 weeks regimen for adults and avoid less frequent treatment regimens (every 4 weeks or every 8 weeks) for long-term maintenance of efficacy,” they wrote in JAMA Dermatology.
Treatment success in the original SOLO trials was defined as having achieved an Investigator’s Global Assessment score of 0-1, or 75% improvement in Eczema Area and Severity Index Scores (EASI-75). As primary endpoints, SOLO-CONTINUE looked at the mean percentage change in EASI score over the course of the trial, and the percentage of patients who maintained EASI-75 at week 36.
Patients in the SOLO-CONTINUE trial who were randomized to receive dupilumab weekly or every 2 weeks had a mean percent change in EASI score of –0.06%. In contrast, patients assigned to the placebo group had a 21.7% decrease, and those taking the medication at 4- and 8-week intervals had mean changes of –3.84% and –6.84%, respectively. Post hoc analyses showed no apparent difference between dupilumab weekly and every 2 weeks in the maintenance of clinical response, the investigators reported.
Among patients with EASI-75 response at baseline, significantly more patients maintained this response at week 36 than patients receiving placebo, and there was again an apparent dose-dependent response. The percentage with EASI-75 at week 36 was 71.6% with the weekly or every-2-weeks regimen, 58.3% with the 4-week regimen, 54.9% with the 8-week regimen, and 30.4% in the placebo group.
Continuing treatment with 300 mg weekly or every 2 weeks resulted in greater maintenance of response across multiple other clinical endpoints and patient-reported outcomes as well (such as pruritus, atopic dermatitis symptoms, sleep, pain or discomfort, quality of life, and symptoms of anxiety and depression).
The more-frequent regimens also conferred no greater risk than less-frequent administration, and there were no new safety signals over the 36-week trial. Treatment-emergent adverse events (the most common were headache, nasopharyngitis, injection-site reactions, and herpes simplex virus infection) occurred in 70.7% of patients in the weekly or every-2-weeks group, 73.6% in the 4-week group, 75% in the 8-week group, and 81.7% in the placebo group.
Unlike earlier studies, the incidence of conjunctivitis was low (less than 6%) across all treatment groups, possibly because patients in the SOLO-CONTINUE trial were all high-level responders who tend to have conjunctivitis less frequently, the authors wrote.
Patients receiving less-frequent doses of dupilumab, particularly every 8 weeks, had greater rates of skin infections, flares, and rescue medication use than patients receiving doses weekly or every 2 weeks, the investigators reported. Treatment-emergent antidrug antibody incidence was slightly higher with less-frequent doses (11.7% and 6% in the 8-week and 4-week groups, respectively, compared with 4.3% and 1.2% in the every-2-weeks and weekly groups), which indicates a “higher incidence of immunogenicity with less-frequent dosage intervals” and is “consistent with other biologics,” they wrote.
Dupilumab is a human monoclonal antibody against the interleukin-4 receptor alpha that inhibits signaling of IL-4 and IL-13. The study was conducted at 185 sites in North America, Europe, Asia, and Japan. Patients had a mean age of 38.2 years; 53.8% were male.
While the trial suggests that the approved regimen of 300 mg every 2 weeks is best for long-term treatment, “therapeutic decisions are often influenced by cost-benefit considerations, in which case practitioners and other stakeholders involved in these decisions should carefully balance potential savings against suboptimal efficacy and long-term risks associated with discontinuous treatment regimens,” the investigators wrote.
The SOLO-CONTINUE trial was funded by Sanofi and Regeneron, the companies that market dupilumab. Dr. Worm reported receiving honoraria for consulting and lecture activity from Regeneron and Sanofi during and outside the conduct of the study, among other disclosures. The other authors had multiple disclosures related to these and multiple other pharmaceutical companies, or were employees of Sanofi or Regeneron.
SOURCE: Worm M et al. JAMA Dermatol. 2019 Dec 26. doi: 10.1001/jamadermatol.2019.3617.
The desire to decrease or stop a therapy such as dupilumab may be motivated by cost, current and potential adverse effects, and individual needs. Because atopic dermatitis is a waxing-and-waning disease with a predilection for cycles of escalation, there is some thought a priori that reduced treatment schedules or discontinued use of a drug may be possible in a state of low disease activity.
The investigators of the SOLO-CONTINUE trial found, however, that continuous treatment with the dosage used in the original SOLO trials (300 mg weekly or every 2 weeks) resulted in a better maintenance of response than a less-frequent dosage and was significantly better than placebo for all endpoints. The less-frequent dosage regimens (every 4 weeks and every 8 weeks), on the other hand, produced some dose-dependent reduction in efficacy.
The development of antidrug antibodies was found in approximately 11% of individuals who received placebo or dupilumab every 8 weeks, 6% of the monthly treatment group, and only 1% in the weekly group, suggesting that less-frequent administration results in higher immunogenicity. However, most of the antidrug antibody levels were low and did not seem to have any clinical effect, making this finding of uncertain relevance to patient care.
The study is valuable because, as more patients are exposed to the drug, more will want or need to reduce the dosage or stop use over time – and although it seems optimal to continue an every-2-weeks treatment regimen, this may not always be feasible. As we integrate new therapies and learn more about atopic dermatitis, it is important that we understand the options and implications around decreasing the dosage of dupilumab. This newly concluded trial is helpful in this regard.
Peter A. Lio, MD, is with the department of dermatology at Northwestern University, Chicago, and the Chicago Integrative Eczema Center. He reported receiving grants and personal fees from Regeneron, Sanofi Genzyme, and other companies, as well as other disclosures. His comments appear in JAMA Dermatology (2019 Dec 26. doi: 10.1001/jamadermatol.2019.3331).
The desire to decrease or stop a therapy such as dupilumab may be motivated by cost, current and potential adverse effects, and individual needs. Because atopic dermatitis is a waxing-and-waning disease with a predilection for cycles of escalation, there is some thought a priori that reduced treatment schedules or discontinued use of a drug may be possible in a state of low disease activity.
The investigators of the SOLO-CONTINUE trial found, however, that continuous treatment with the dosage used in the original SOLO trials (300 mg weekly or every 2 weeks) resulted in a better maintenance of response than a less-frequent dosage and was significantly better than placebo for all endpoints. The less-frequent dosage regimens (every 4 weeks and every 8 weeks), on the other hand, produced some dose-dependent reduction in efficacy.
The development of antidrug antibodies was found in approximately 11% of individuals who received placebo or dupilumab every 8 weeks, 6% of the monthly treatment group, and only 1% in the weekly group, suggesting that less-frequent administration results in higher immunogenicity. However, most of the antidrug antibody levels were low and did not seem to have any clinical effect, making this finding of uncertain relevance to patient care.
The study is valuable because, as more patients are exposed to the drug, more will want or need to reduce the dosage or stop use over time – and although it seems optimal to continue an every-2-weeks treatment regimen, this may not always be feasible. As we integrate new therapies and learn more about atopic dermatitis, it is important that we understand the options and implications around decreasing the dosage of dupilumab. This newly concluded trial is helpful in this regard.
Peter A. Lio, MD, is with the department of dermatology at Northwestern University, Chicago, and the Chicago Integrative Eczema Center. He reported receiving grants and personal fees from Regeneron, Sanofi Genzyme, and other companies, as well as other disclosures. His comments appear in JAMA Dermatology (2019 Dec 26. doi: 10.1001/jamadermatol.2019.3331).
The desire to decrease or stop a therapy such as dupilumab may be motivated by cost, current and potential adverse effects, and individual needs. Because atopic dermatitis is a waxing-and-waning disease with a predilection for cycles of escalation, there is some thought a priori that reduced treatment schedules or discontinued use of a drug may be possible in a state of low disease activity.
The investigators of the SOLO-CONTINUE trial found, however, that continuous treatment with the dosage used in the original SOLO trials (300 mg weekly or every 2 weeks) resulted in a better maintenance of response than a less-frequent dosage and was significantly better than placebo for all endpoints. The less-frequent dosage regimens (every 4 weeks and every 8 weeks), on the other hand, produced some dose-dependent reduction in efficacy.
The development of antidrug antibodies was found in approximately 11% of individuals who received placebo or dupilumab every 8 weeks, 6% of the monthly treatment group, and only 1% in the weekly group, suggesting that less-frequent administration results in higher immunogenicity. However, most of the antidrug antibody levels were low and did not seem to have any clinical effect, making this finding of uncertain relevance to patient care.
The study is valuable because, as more patients are exposed to the drug, more will want or need to reduce the dosage or stop use over time – and although it seems optimal to continue an every-2-weeks treatment regimen, this may not always be feasible. As we integrate new therapies and learn more about atopic dermatitis, it is important that we understand the options and implications around decreasing the dosage of dupilumab. This newly concluded trial is helpful in this regard.
Peter A. Lio, MD, is with the department of dermatology at Northwestern University, Chicago, and the Chicago Integrative Eczema Center. He reported receiving grants and personal fees from Regeneron, Sanofi Genzyme, and other companies, as well as other disclosures. His comments appear in JAMA Dermatology (2019 Dec 26. doi: 10.1001/jamadermatol.2019.3331).
Patients with moderate to severe atopic dermatitis who responded well to the approved dupilumab regimen of 300 mg every 2 weeks in pivotal phase 3 monotherapy trials were more likely to have a continued response over the longer term if they maintained this regimen rather than switching to longer dosing intervals or discontinuing the medication.
This finding comes from a 36-week, randomized, double-blind, placebo-controlled trial that enrolled 422 – SOLO 1 and SOLO 2.
The new international study – SOLO-CONTINUE – randomized these patients to continue the original regimen (weekly or every 2 weeks), to receive 300 mg of the biologic medication every 4 or 8 weeks, or to receive placebo.
Patients who continued the original regimen had the most consistent maintenance of treatment effect, while patients on longer dosage intervals or placebo had a dose-dependent reduction in response and no safety advantage. The incidence of treatment-emergent antidrug antibody was lowest with dupilumab weekly or every 2 weeks, and slightly higher with less-frequent dosing intervals, reported Margitta Worm, MD, of the Charité Universitätsmedizin Berlin, and coinvestigators.
“Because administration every 4 weeks or every 8 weeks did not provide an additional safety advantage and was numerically outperformed by administration weekly or every 2 weeks, we believe that it is prudent to adhere to the approved every 2 weeks regimen for adults and avoid less frequent treatment regimens (every 4 weeks or every 8 weeks) for long-term maintenance of efficacy,” they wrote in JAMA Dermatology.
Treatment success in the original SOLO trials was defined as having achieved an Investigator’s Global Assessment score of 0-1, or 75% improvement in Eczema Area and Severity Index Scores (EASI-75). As primary endpoints, SOLO-CONTINUE looked at the mean percentage change in EASI score over the course of the trial, and the percentage of patients who maintained EASI-75 at week 36.
Patients in the SOLO-CONTINUE trial who were randomized to receive dupilumab weekly or every 2 weeks had a mean percent change in EASI score of –0.06%. In contrast, patients assigned to the placebo group had a 21.7% decrease, and those taking the medication at 4- and 8-week intervals had mean changes of –3.84% and –6.84%, respectively. Post hoc analyses showed no apparent difference between dupilumab weekly and every 2 weeks in the maintenance of clinical response, the investigators reported.
Among patients with EASI-75 response at baseline, significantly more patients maintained this response at week 36 than patients receiving placebo, and there was again an apparent dose-dependent response. The percentage with EASI-75 at week 36 was 71.6% with the weekly or every-2-weeks regimen, 58.3% with the 4-week regimen, 54.9% with the 8-week regimen, and 30.4% in the placebo group.
Continuing treatment with 300 mg weekly or every 2 weeks resulted in greater maintenance of response across multiple other clinical endpoints and patient-reported outcomes as well (such as pruritus, atopic dermatitis symptoms, sleep, pain or discomfort, quality of life, and symptoms of anxiety and depression).
The more-frequent regimens also conferred no greater risk than less-frequent administration, and there were no new safety signals over the 36-week trial. Treatment-emergent adverse events (the most common were headache, nasopharyngitis, injection-site reactions, and herpes simplex virus infection) occurred in 70.7% of patients in the weekly or every-2-weeks group, 73.6% in the 4-week group, 75% in the 8-week group, and 81.7% in the placebo group.
Unlike earlier studies, the incidence of conjunctivitis was low (less than 6%) across all treatment groups, possibly because patients in the SOLO-CONTINUE trial were all high-level responders who tend to have conjunctivitis less frequently, the authors wrote.
Patients receiving less-frequent doses of dupilumab, particularly every 8 weeks, had greater rates of skin infections, flares, and rescue medication use than patients receiving doses weekly or every 2 weeks, the investigators reported. Treatment-emergent antidrug antibody incidence was slightly higher with less-frequent doses (11.7% and 6% in the 8-week and 4-week groups, respectively, compared with 4.3% and 1.2% in the every-2-weeks and weekly groups), which indicates a “higher incidence of immunogenicity with less-frequent dosage intervals” and is “consistent with other biologics,” they wrote.
Dupilumab is a human monoclonal antibody against the interleukin-4 receptor alpha that inhibits signaling of IL-4 and IL-13. The study was conducted at 185 sites in North America, Europe, Asia, and Japan. Patients had a mean age of 38.2 years; 53.8% were male.
While the trial suggests that the approved regimen of 300 mg every 2 weeks is best for long-term treatment, “therapeutic decisions are often influenced by cost-benefit considerations, in which case practitioners and other stakeholders involved in these decisions should carefully balance potential savings against suboptimal efficacy and long-term risks associated with discontinuous treatment regimens,” the investigators wrote.
The SOLO-CONTINUE trial was funded by Sanofi and Regeneron, the companies that market dupilumab. Dr. Worm reported receiving honoraria for consulting and lecture activity from Regeneron and Sanofi during and outside the conduct of the study, among other disclosures. The other authors had multiple disclosures related to these and multiple other pharmaceutical companies, or were employees of Sanofi or Regeneron.
SOURCE: Worm M et al. JAMA Dermatol. 2019 Dec 26. doi: 10.1001/jamadermatol.2019.3617.
Patients with moderate to severe atopic dermatitis who responded well to the approved dupilumab regimen of 300 mg every 2 weeks in pivotal phase 3 monotherapy trials were more likely to have a continued response over the longer term if they maintained this regimen rather than switching to longer dosing intervals or discontinuing the medication.
This finding comes from a 36-week, randomized, double-blind, placebo-controlled trial that enrolled 422 – SOLO 1 and SOLO 2.
The new international study – SOLO-CONTINUE – randomized these patients to continue the original regimen (weekly or every 2 weeks), to receive 300 mg of the biologic medication every 4 or 8 weeks, or to receive placebo.
Patients who continued the original regimen had the most consistent maintenance of treatment effect, while patients on longer dosage intervals or placebo had a dose-dependent reduction in response and no safety advantage. The incidence of treatment-emergent antidrug antibody was lowest with dupilumab weekly or every 2 weeks, and slightly higher with less-frequent dosing intervals, reported Margitta Worm, MD, of the Charité Universitätsmedizin Berlin, and coinvestigators.
“Because administration every 4 weeks or every 8 weeks did not provide an additional safety advantage and was numerically outperformed by administration weekly or every 2 weeks, we believe that it is prudent to adhere to the approved every 2 weeks regimen for adults and avoid less frequent treatment regimens (every 4 weeks or every 8 weeks) for long-term maintenance of efficacy,” they wrote in JAMA Dermatology.
Treatment success in the original SOLO trials was defined as having achieved an Investigator’s Global Assessment score of 0-1, or 75% improvement in Eczema Area and Severity Index Scores (EASI-75). As primary endpoints, SOLO-CONTINUE looked at the mean percentage change in EASI score over the course of the trial, and the percentage of patients who maintained EASI-75 at week 36.
Patients in the SOLO-CONTINUE trial who were randomized to receive dupilumab weekly or every 2 weeks had a mean percent change in EASI score of –0.06%. In contrast, patients assigned to the placebo group had a 21.7% decrease, and those taking the medication at 4- and 8-week intervals had mean changes of –3.84% and –6.84%, respectively. Post hoc analyses showed no apparent difference between dupilumab weekly and every 2 weeks in the maintenance of clinical response, the investigators reported.
Among patients with EASI-75 response at baseline, significantly more patients maintained this response at week 36 than patients receiving placebo, and there was again an apparent dose-dependent response. The percentage with EASI-75 at week 36 was 71.6% with the weekly or every-2-weeks regimen, 58.3% with the 4-week regimen, 54.9% with the 8-week regimen, and 30.4% in the placebo group.
Continuing treatment with 300 mg weekly or every 2 weeks resulted in greater maintenance of response across multiple other clinical endpoints and patient-reported outcomes as well (such as pruritus, atopic dermatitis symptoms, sleep, pain or discomfort, quality of life, and symptoms of anxiety and depression).
The more-frequent regimens also conferred no greater risk than less-frequent administration, and there were no new safety signals over the 36-week trial. Treatment-emergent adverse events (the most common were headache, nasopharyngitis, injection-site reactions, and herpes simplex virus infection) occurred in 70.7% of patients in the weekly or every-2-weeks group, 73.6% in the 4-week group, 75% in the 8-week group, and 81.7% in the placebo group.
Unlike earlier studies, the incidence of conjunctivitis was low (less than 6%) across all treatment groups, possibly because patients in the SOLO-CONTINUE trial were all high-level responders who tend to have conjunctivitis less frequently, the authors wrote.
Patients receiving less-frequent doses of dupilumab, particularly every 8 weeks, had greater rates of skin infections, flares, and rescue medication use than patients receiving doses weekly or every 2 weeks, the investigators reported. Treatment-emergent antidrug antibody incidence was slightly higher with less-frequent doses (11.7% and 6% in the 8-week and 4-week groups, respectively, compared with 4.3% and 1.2% in the every-2-weeks and weekly groups), which indicates a “higher incidence of immunogenicity with less-frequent dosage intervals” and is “consistent with other biologics,” they wrote.
Dupilumab is a human monoclonal antibody against the interleukin-4 receptor alpha that inhibits signaling of IL-4 and IL-13. The study was conducted at 185 sites in North America, Europe, Asia, and Japan. Patients had a mean age of 38.2 years; 53.8% were male.
While the trial suggests that the approved regimen of 300 mg every 2 weeks is best for long-term treatment, “therapeutic decisions are often influenced by cost-benefit considerations, in which case practitioners and other stakeholders involved in these decisions should carefully balance potential savings against suboptimal efficacy and long-term risks associated with discontinuous treatment regimens,” the investigators wrote.
The SOLO-CONTINUE trial was funded by Sanofi and Regeneron, the companies that market dupilumab. Dr. Worm reported receiving honoraria for consulting and lecture activity from Regeneron and Sanofi during and outside the conduct of the study, among other disclosures. The other authors had multiple disclosures related to these and multiple other pharmaceutical companies, or were employees of Sanofi or Regeneron.
SOURCE: Worm M et al. JAMA Dermatol. 2019 Dec 26. doi: 10.1001/jamadermatol.2019.3617.
FROM JAMA DERMATOLOGY
Papules on trunk
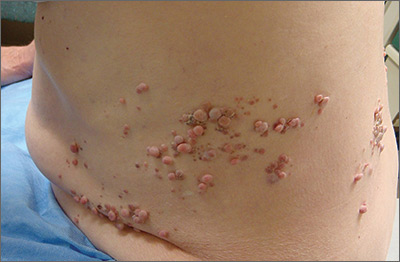
Dermatopathologic evaluation of the tissue sample indicated that the lesion was a neurofibroma, and clinical correlation fine-tuned the diagnosis to segmental neurofibromatosis (NF). The diagnosis of segmental NF is clinical, with biopsy to confirm that the lesions are neurofibromas. Segmental NF is a mosaic form of neurofibromatosis type 1 (NF1) that results from a postzygotic mutation of the NF1 gene. While NF1 is a relatively common neurocutaneous disorder that occurs with a frequency of 1 in 3000, segmental NF is uncommon, with an estimated prevalence of 1 in 40,000.
NF1 often follows an autosomal dominant inheritance pattern, although up to 50% of patients with NF1 arise de novo from spontaneous mutations. NF1 is characterized by multiple café-au-lait macules, axillary freckling, neurofibromas, and Lisch nodules (pigmented iris hamartomas). Systemic findings associated with NF1 include malignant peripheral nerve sheath tumors, optic gliomas, and vasculopathy. While patients with segmental NF may exhibit some of these same findings, the distribution of the neurofibromas is often limited to a single dermatome. Additionally, patients with segmental NF typically do not exhibit extracutaneous lesions, systemic involvement, or a family history of NF. Segmental NF treatment typically focuses on symptomatic management or cosmetic concerns.
This patient did not have any of the systemic complications that occasionally occur with segmental NF, so no medical treatment was required. The patient was advised that cutaneous and subcutaneous neurofibromas do not require removal unless there is pain, bleeding, disfigurement, or signs of malignant transformation. The patient was not interested in removal of the nodules for cosmetic reasons, so he was encouraged to seek follow-up care, as needed.
This case was adapted from: Laurent KJ, Beachkofsky TM, Loyd A, et al. Segmental distribution of nodules on trunk. J Fam Pract. 2017;66: 765-767.

Dermatopathologic evaluation of the tissue sample indicated that the lesion was a neurofibroma, and clinical correlation fine-tuned the diagnosis to segmental neurofibromatosis (NF). The diagnosis of segmental NF is clinical, with biopsy to confirm that the lesions are neurofibromas. Segmental NF is a mosaic form of neurofibromatosis type 1 (NF1) that results from a postzygotic mutation of the NF1 gene. While NF1 is a relatively common neurocutaneous disorder that occurs with a frequency of 1 in 3000, segmental NF is uncommon, with an estimated prevalence of 1 in 40,000.
NF1 often follows an autosomal dominant inheritance pattern, although up to 50% of patients with NF1 arise de novo from spontaneous mutations. NF1 is characterized by multiple café-au-lait macules, axillary freckling, neurofibromas, and Lisch nodules (pigmented iris hamartomas). Systemic findings associated with NF1 include malignant peripheral nerve sheath tumors, optic gliomas, and vasculopathy. While patients with segmental NF may exhibit some of these same findings, the distribution of the neurofibromas is often limited to a single dermatome. Additionally, patients with segmental NF typically do not exhibit extracutaneous lesions, systemic involvement, or a family history of NF. Segmental NF treatment typically focuses on symptomatic management or cosmetic concerns.
This patient did not have any of the systemic complications that occasionally occur with segmental NF, so no medical treatment was required. The patient was advised that cutaneous and subcutaneous neurofibromas do not require removal unless there is pain, bleeding, disfigurement, or signs of malignant transformation. The patient was not interested in removal of the nodules for cosmetic reasons, so he was encouraged to seek follow-up care, as needed.
This case was adapted from: Laurent KJ, Beachkofsky TM, Loyd A, et al. Segmental distribution of nodules on trunk. J Fam Pract. 2017;66: 765-767.

Dermatopathologic evaluation of the tissue sample indicated that the lesion was a neurofibroma, and clinical correlation fine-tuned the diagnosis to segmental neurofibromatosis (NF). The diagnosis of segmental NF is clinical, with biopsy to confirm that the lesions are neurofibromas. Segmental NF is a mosaic form of neurofibromatosis type 1 (NF1) that results from a postzygotic mutation of the NF1 gene. While NF1 is a relatively common neurocutaneous disorder that occurs with a frequency of 1 in 3000, segmental NF is uncommon, with an estimated prevalence of 1 in 40,000.
NF1 often follows an autosomal dominant inheritance pattern, although up to 50% of patients with NF1 arise de novo from spontaneous mutations. NF1 is characterized by multiple café-au-lait macules, axillary freckling, neurofibromas, and Lisch nodules (pigmented iris hamartomas). Systemic findings associated with NF1 include malignant peripheral nerve sheath tumors, optic gliomas, and vasculopathy. While patients with segmental NF may exhibit some of these same findings, the distribution of the neurofibromas is often limited to a single dermatome. Additionally, patients with segmental NF typically do not exhibit extracutaneous lesions, systemic involvement, or a family history of NF. Segmental NF treatment typically focuses on symptomatic management or cosmetic concerns.
This patient did not have any of the systemic complications that occasionally occur with segmental NF, so no medical treatment was required. The patient was advised that cutaneous and subcutaneous neurofibromas do not require removal unless there is pain, bleeding, disfigurement, or signs of malignant transformation. The patient was not interested in removal of the nodules for cosmetic reasons, so he was encouraged to seek follow-up care, as needed.
This case was adapted from: Laurent KJ, Beachkofsky TM, Loyd A, et al. Segmental distribution of nodules on trunk. J Fam Pract. 2017;66: 765-767.