User login
Lesion Has Been Giving Him an Earful
ANSWER
The correct answer is gouty tophus (choice “b”).
DISCUSSION
Gout is a defect of purine metabolism, usually caused by underexcretion of uric acid. Diet and heredity also play parts in gout’s development. Gouty tophi usually develop after years of hyperuricemia. As uric acid builds up in the bloodstream over time, it can then begin to be deposited into joints—most commonly the first metatarsal-phalangeal—as well as cartilage or even bones.
On further questioning, the patient recalled having been told on several occasions that his serum uric acid was elevated. In retrospect, his arthritis was most likely gouty in nature.
In terms of the differential, BCC (choice “a”) is common on helical rims, but it would not have contained the type of material found in this patient’s lesion. Also, it would not have waxed and waned as this lesion had done.
Epidermal cysts (choice “c”) can certainly come and go in prominence, but they are filled with a cheesy, pasty material—not the dry crystalline substance found in this lesion. Moreover, most epidermal cysts will have a small comedonal punctum over the center of the lesion. Dystrophic calcification (choice “d”) can mimic gouty tophi, but it is usually rough, firm, and fixed. It certainly would not be coming and going as it pleases.
TREATMENT
Surgical excision of the tophus was offered, but the patient was content with knowing the correct diagnosis. His PCP had previously explained therapeutic options—such as medication and dietary changes—that could address the overall problem. The patient elected to pursue treatment with his PCP.
ANSWER
The correct answer is gouty tophus (choice “b”).
DISCUSSION
Gout is a defect of purine metabolism, usually caused by underexcretion of uric acid. Diet and heredity also play parts in gout’s development. Gouty tophi usually develop after years of hyperuricemia. As uric acid builds up in the bloodstream over time, it can then begin to be deposited into joints—most commonly the first metatarsal-phalangeal—as well as cartilage or even bones.
On further questioning, the patient recalled having been told on several occasions that his serum uric acid was elevated. In retrospect, his arthritis was most likely gouty in nature.
In terms of the differential, BCC (choice “a”) is common on helical rims, but it would not have contained the type of material found in this patient’s lesion. Also, it would not have waxed and waned as this lesion had done.
Epidermal cysts (choice “c”) can certainly come and go in prominence, but they are filled with a cheesy, pasty material—not the dry crystalline substance found in this lesion. Moreover, most epidermal cysts will have a small comedonal punctum over the center of the lesion. Dystrophic calcification (choice “d”) can mimic gouty tophi, but it is usually rough, firm, and fixed. It certainly would not be coming and going as it pleases.
TREATMENT
Surgical excision of the tophus was offered, but the patient was content with knowing the correct diagnosis. His PCP had previously explained therapeutic options—such as medication and dietary changes—that could address the overall problem. The patient elected to pursue treatment with his PCP.
ANSWER
The correct answer is gouty tophus (choice “b”).
DISCUSSION
Gout is a defect of purine metabolism, usually caused by underexcretion of uric acid. Diet and heredity also play parts in gout’s development. Gouty tophi usually develop after years of hyperuricemia. As uric acid builds up in the bloodstream over time, it can then begin to be deposited into joints—most commonly the first metatarsal-phalangeal—as well as cartilage or even bones.
On further questioning, the patient recalled having been told on several occasions that his serum uric acid was elevated. In retrospect, his arthritis was most likely gouty in nature.
In terms of the differential, BCC (choice “a”) is common on helical rims, but it would not have contained the type of material found in this patient’s lesion. Also, it would not have waxed and waned as this lesion had done.
Epidermal cysts (choice “c”) can certainly come and go in prominence, but they are filled with a cheesy, pasty material—not the dry crystalline substance found in this lesion. Moreover, most epidermal cysts will have a small comedonal punctum over the center of the lesion. Dystrophic calcification (choice “d”) can mimic gouty tophi, but it is usually rough, firm, and fixed. It certainly would not be coming and going as it pleases.
TREATMENT
Surgical excision of the tophus was offered, but the patient was content with knowing the correct diagnosis. His PCP had previously explained therapeutic options—such as medication and dietary changes—that could address the overall problem. The patient elected to pursue treatment with his PCP.
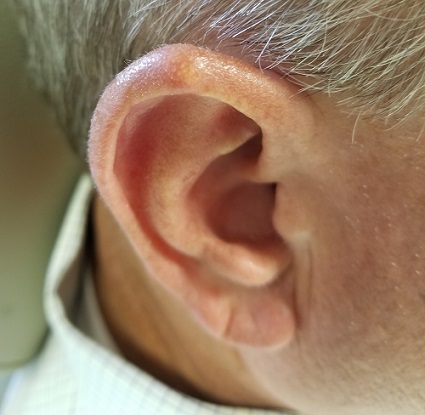
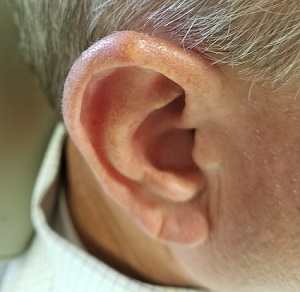
Over the years, the lesion on this 49-year-old man’s right ear has waxed and waned in prominence. Although it never causes pain, its unrelenting existence coupled with a history of basal cell carcinoma (BCC) on his face has caused him to worry. He has had no other lesions and is in otherwise good health, except for occasional bouts of arthritis, for which he takes ibuprofen, with good results.
While his family had suggested that the lesion could be a cyst, his primary care provider (PCP) disagreed and referred him to dermatology.
A 5-mm firm papule is located at the right helical crest. The lesion is skin-colored, with no redness, and nontender on palpation, although it is moderately firm and mobile. No punctum is noted.
The surrounding skin has no signs of sun damage, although the patient is quite fair. As a young man, he had far too much sun exposure, burning easily and tanning only with difficulty.
After consultation with the patient, the decision is made to incise the lesion using a #11 blade to examine its contents. The incision reveals a dry crystalline substance, which is easily cleared with brief curettage. This effectively leads to a flattening of the lesion.
Protean manifestations of COVID-19: “Our ignorance is profound”
Although a cause-and-effect relationship is unknown, people with the virus have presented with or developed heart disease, acute liver injury, ongoing GI issues, skin manifestations, neurologic damage, and other problems, especially among sicker people.
For example, French physicians described an association with encephalopathy, agitation, confusion, and corticospinal tract signs among 58 people hospitalized with acute respiratory distress (N Engl J Med. 2020 Apr 15. doi: 10.1056/NEJMc2008597).
In particular, Yale New Haven (Conn.) Hospital is dealing with unexpected complications up close. Almost half of the beds there are occupied by COVID-19 patients. Over 100 people are in the ICU, and almost 70 intubated. Of the more than 750 COVID admissions so far, only about 350 have been discharged. “Even in a bad flu season, you never see something like this; it’s just unheard of,” said Harlan Krumholz, MD, a Yale cardiologist and professor of medicine helping lead the efforts there.
Kidney injuries prominent
“When they get to the ICU, we are seeing lots of people with acute kidney injuries; lots of people developing endocrine problems; people having blood sugar control issues, coagulation issues, blood clots. We are just waking up to the wide range of ways this virus can affect people. Our ignorance is profound,” Dr. Krumholz said, but physicians “recognize that this thing has the capability of attacking almost every single organ system, and it may or may not present with respiratory symptoms.”
It’s a similar story at Mt. Sinai South Nassau, a hospital in Oceanside, N.Y. “We’ve seen a lot of renal injury in people having complications, a lot of acute dialysis,” but it’s unclear how much is caused by the virus and how much is simply because people are so sick, said Aaron Glatt, MD, infectious disease professor and chair of medicine at the hospital. However, he said things are looking brighter than at Yale.
“We are not seeing the same level of increase in cases that we had previously, and we are starting to see extubations and discharges. We’ve treated a number of patients with plasma therapy, and hopefully that will be of benefit. We’ve seen some response to” the immunosuppressive “tocilizumab [Actemra], and a lot of response to very good respiratory therapy. I think we are starting to flatten the curve,” Dr. Glatt said.
“Look for tricky symptoms”
The growing awareness of COVID’s protean manifestations is evident in Medscape’s Consult forum, an online community where physicians and medical students share information and seek advice; there’s been over 200 COVID-19 cases and questions since January.
Early on, traffic was mostly about typical pulmonary presentations, but lately it’s shifted to nonrespiratory involvement. Physicians want to know if what they are seeing is related to the virus, and if other people are seeing the same things.
There’s a case on Consult of a 37-year-old man with stomach pain, vomiting, and diarrhea, but no respiratory symptoms and a positive COVID test. A chest CT incidental to his abdominal scan revealed significant bilateral lung involvement.
A 69-year-old woman with a history of laparotomy and new onset intestinal subocclusion had only adhesions on a subsequent exploratory laparotomy, and was doing okay otherwise. She suddenly went into respiratory failure with progressive bradycardia and died 3 days later. Aspiration pneumonia, pulmonary embolism, and MI had been ruled out. “The pattern of cardiovascular failure was in favor of myocarditis, but we don’t have any other clue,” the physician said after describing a second similar case.
Another doctor on the forum reported elevated cardiac enzymes without coronary artery obstruction in a positive patient who went into shock, with an ejection fraction of 40% and markedly increased heart wall thickness, but no lung involvement. There are also two cases of idiopathic thrombocytopenia without fever of hypoxia.
An Italian gastroenterologist said: “Look for tricky symptoms.” Expand “patient history, asking about the sudden occurrence of dysgeusia and/or anosmia. These symptoms have become my guiding diagnostic light” in Verona. “Most patients become nauseated, [and] the taste of any food is unbearable. When I find these symptoms by history, the patient is COVID positive 100%.”
‘Make sure that they didn’t die in vain’
There was interest in those and other reports on Consult, and comments from physicians who have theories, but no certain answers about what is, and is not, caused by the virus.
Direct viral attack is likely a part of it, said Stanley Perlman, MD, PhD, a professor of microbiology and immunology at the University of Iowa, Iowa City.
The ACE2 receptor the virus uses to enter cells is common in many organs, plus there were extrapulmonary manifestations with severe acute respiratory syndrome (SARS), another pandemic caused by a zoonotic coronavirus almost 20 years ago. At least with SARS, “many organs were infected when examined at autopsy,” he said.
The body’s inflammatory response is almost certainly also in play. Progressive derangements in inflammatory markers – C-reactive protein, D-dimer, ferritin – correlate with worse prognosis, and “the cytokine storm that occurs in these patients can lead to a degree of encephalopathy, myocarditis, liver impairment, and kidney impairment; multiorgan dysfunction, in other words,” said William Shaffner, MD, a professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
But in some cases, the virus might simply be a bystander to an unrelated disease process; in others, the experimental treatments being used might cause problems. Indeed, cardiology groups recently warned of torsade de pointes – a dangerously abnormal heart rhythm – with hydroxychloroquine and azithromycin.
“We think it’s some combination,” but don’t really know, Dr. Krumholz said. In the meantime, “we are forced to treat patients by instinct and first principles,” and long-term sequelae are unknown. “We don’t want to be in this position for long.”
To that end, he said, “this is the time for us all to hold hands and be together because we need to learn rapidly from each other. Our job is both to care for the people in front of us and make sure that they didn’t die in vain, that the experience they had is funneled into a larger set of data to make sure the next person is better off.”
Although a cause-and-effect relationship is unknown, people with the virus have presented with or developed heart disease, acute liver injury, ongoing GI issues, skin manifestations, neurologic damage, and other problems, especially among sicker people.
For example, French physicians described an association with encephalopathy, agitation, confusion, and corticospinal tract signs among 58 people hospitalized with acute respiratory distress (N Engl J Med. 2020 Apr 15. doi: 10.1056/NEJMc2008597).
In particular, Yale New Haven (Conn.) Hospital is dealing with unexpected complications up close. Almost half of the beds there are occupied by COVID-19 patients. Over 100 people are in the ICU, and almost 70 intubated. Of the more than 750 COVID admissions so far, only about 350 have been discharged. “Even in a bad flu season, you never see something like this; it’s just unheard of,” said Harlan Krumholz, MD, a Yale cardiologist and professor of medicine helping lead the efforts there.
Kidney injuries prominent
“When they get to the ICU, we are seeing lots of people with acute kidney injuries; lots of people developing endocrine problems; people having blood sugar control issues, coagulation issues, blood clots. We are just waking up to the wide range of ways this virus can affect people. Our ignorance is profound,” Dr. Krumholz said, but physicians “recognize that this thing has the capability of attacking almost every single organ system, and it may or may not present with respiratory symptoms.”
It’s a similar story at Mt. Sinai South Nassau, a hospital in Oceanside, N.Y. “We’ve seen a lot of renal injury in people having complications, a lot of acute dialysis,” but it’s unclear how much is caused by the virus and how much is simply because people are so sick, said Aaron Glatt, MD, infectious disease professor and chair of medicine at the hospital. However, he said things are looking brighter than at Yale.
“We are not seeing the same level of increase in cases that we had previously, and we are starting to see extubations and discharges. We’ve treated a number of patients with plasma therapy, and hopefully that will be of benefit. We’ve seen some response to” the immunosuppressive “tocilizumab [Actemra], and a lot of response to very good respiratory therapy. I think we are starting to flatten the curve,” Dr. Glatt said.
“Look for tricky symptoms”
The growing awareness of COVID’s protean manifestations is evident in Medscape’s Consult forum, an online community where physicians and medical students share information and seek advice; there’s been over 200 COVID-19 cases and questions since January.
Early on, traffic was mostly about typical pulmonary presentations, but lately it’s shifted to nonrespiratory involvement. Physicians want to know if what they are seeing is related to the virus, and if other people are seeing the same things.
There’s a case on Consult of a 37-year-old man with stomach pain, vomiting, and diarrhea, but no respiratory symptoms and a positive COVID test. A chest CT incidental to his abdominal scan revealed significant bilateral lung involvement.
A 69-year-old woman with a history of laparotomy and new onset intestinal subocclusion had only adhesions on a subsequent exploratory laparotomy, and was doing okay otherwise. She suddenly went into respiratory failure with progressive bradycardia and died 3 days later. Aspiration pneumonia, pulmonary embolism, and MI had been ruled out. “The pattern of cardiovascular failure was in favor of myocarditis, but we don’t have any other clue,” the physician said after describing a second similar case.
Another doctor on the forum reported elevated cardiac enzymes without coronary artery obstruction in a positive patient who went into shock, with an ejection fraction of 40% and markedly increased heart wall thickness, but no lung involvement. There are also two cases of idiopathic thrombocytopenia without fever of hypoxia.
An Italian gastroenterologist said: “Look for tricky symptoms.” Expand “patient history, asking about the sudden occurrence of dysgeusia and/or anosmia. These symptoms have become my guiding diagnostic light” in Verona. “Most patients become nauseated, [and] the taste of any food is unbearable. When I find these symptoms by history, the patient is COVID positive 100%.”
‘Make sure that they didn’t die in vain’
There was interest in those and other reports on Consult, and comments from physicians who have theories, but no certain answers about what is, and is not, caused by the virus.
Direct viral attack is likely a part of it, said Stanley Perlman, MD, PhD, a professor of microbiology and immunology at the University of Iowa, Iowa City.
The ACE2 receptor the virus uses to enter cells is common in many organs, plus there were extrapulmonary manifestations with severe acute respiratory syndrome (SARS), another pandemic caused by a zoonotic coronavirus almost 20 years ago. At least with SARS, “many organs were infected when examined at autopsy,” he said.
The body’s inflammatory response is almost certainly also in play. Progressive derangements in inflammatory markers – C-reactive protein, D-dimer, ferritin – correlate with worse prognosis, and “the cytokine storm that occurs in these patients can lead to a degree of encephalopathy, myocarditis, liver impairment, and kidney impairment; multiorgan dysfunction, in other words,” said William Shaffner, MD, a professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
But in some cases, the virus might simply be a bystander to an unrelated disease process; in others, the experimental treatments being used might cause problems. Indeed, cardiology groups recently warned of torsade de pointes – a dangerously abnormal heart rhythm – with hydroxychloroquine and azithromycin.
“We think it’s some combination,” but don’t really know, Dr. Krumholz said. In the meantime, “we are forced to treat patients by instinct and first principles,” and long-term sequelae are unknown. “We don’t want to be in this position for long.”
To that end, he said, “this is the time for us all to hold hands and be together because we need to learn rapidly from each other. Our job is both to care for the people in front of us and make sure that they didn’t die in vain, that the experience they had is funneled into a larger set of data to make sure the next person is better off.”
Although a cause-and-effect relationship is unknown, people with the virus have presented with or developed heart disease, acute liver injury, ongoing GI issues, skin manifestations, neurologic damage, and other problems, especially among sicker people.
For example, French physicians described an association with encephalopathy, agitation, confusion, and corticospinal tract signs among 58 people hospitalized with acute respiratory distress (N Engl J Med. 2020 Apr 15. doi: 10.1056/NEJMc2008597).
In particular, Yale New Haven (Conn.) Hospital is dealing with unexpected complications up close. Almost half of the beds there are occupied by COVID-19 patients. Over 100 people are in the ICU, and almost 70 intubated. Of the more than 750 COVID admissions so far, only about 350 have been discharged. “Even in a bad flu season, you never see something like this; it’s just unheard of,” said Harlan Krumholz, MD, a Yale cardiologist and professor of medicine helping lead the efforts there.
Kidney injuries prominent
“When they get to the ICU, we are seeing lots of people with acute kidney injuries; lots of people developing endocrine problems; people having blood sugar control issues, coagulation issues, blood clots. We are just waking up to the wide range of ways this virus can affect people. Our ignorance is profound,” Dr. Krumholz said, but physicians “recognize that this thing has the capability of attacking almost every single organ system, and it may or may not present with respiratory symptoms.”
It’s a similar story at Mt. Sinai South Nassau, a hospital in Oceanside, N.Y. “We’ve seen a lot of renal injury in people having complications, a lot of acute dialysis,” but it’s unclear how much is caused by the virus and how much is simply because people are so sick, said Aaron Glatt, MD, infectious disease professor and chair of medicine at the hospital. However, he said things are looking brighter than at Yale.
“We are not seeing the same level of increase in cases that we had previously, and we are starting to see extubations and discharges. We’ve treated a number of patients with plasma therapy, and hopefully that will be of benefit. We’ve seen some response to” the immunosuppressive “tocilizumab [Actemra], and a lot of response to very good respiratory therapy. I think we are starting to flatten the curve,” Dr. Glatt said.
“Look for tricky symptoms”
The growing awareness of COVID’s protean manifestations is evident in Medscape’s Consult forum, an online community where physicians and medical students share information and seek advice; there’s been over 200 COVID-19 cases and questions since January.
Early on, traffic was mostly about typical pulmonary presentations, but lately it’s shifted to nonrespiratory involvement. Physicians want to know if what they are seeing is related to the virus, and if other people are seeing the same things.
There’s a case on Consult of a 37-year-old man with stomach pain, vomiting, and diarrhea, but no respiratory symptoms and a positive COVID test. A chest CT incidental to his abdominal scan revealed significant bilateral lung involvement.
A 69-year-old woman with a history of laparotomy and new onset intestinal subocclusion had only adhesions on a subsequent exploratory laparotomy, and was doing okay otherwise. She suddenly went into respiratory failure with progressive bradycardia and died 3 days later. Aspiration pneumonia, pulmonary embolism, and MI had been ruled out. “The pattern of cardiovascular failure was in favor of myocarditis, but we don’t have any other clue,” the physician said after describing a second similar case.
Another doctor on the forum reported elevated cardiac enzymes without coronary artery obstruction in a positive patient who went into shock, with an ejection fraction of 40% and markedly increased heart wall thickness, but no lung involvement. There are also two cases of idiopathic thrombocytopenia without fever of hypoxia.
An Italian gastroenterologist said: “Look for tricky symptoms.” Expand “patient history, asking about the sudden occurrence of dysgeusia and/or anosmia. These symptoms have become my guiding diagnostic light” in Verona. “Most patients become nauseated, [and] the taste of any food is unbearable. When I find these symptoms by history, the patient is COVID positive 100%.”
‘Make sure that they didn’t die in vain’
There was interest in those and other reports on Consult, and comments from physicians who have theories, but no certain answers about what is, and is not, caused by the virus.
Direct viral attack is likely a part of it, said Stanley Perlman, MD, PhD, a professor of microbiology and immunology at the University of Iowa, Iowa City.
The ACE2 receptor the virus uses to enter cells is common in many organs, plus there were extrapulmonary manifestations with severe acute respiratory syndrome (SARS), another pandemic caused by a zoonotic coronavirus almost 20 years ago. At least with SARS, “many organs were infected when examined at autopsy,” he said.
The body’s inflammatory response is almost certainly also in play. Progressive derangements in inflammatory markers – C-reactive protein, D-dimer, ferritin – correlate with worse prognosis, and “the cytokine storm that occurs in these patients can lead to a degree of encephalopathy, myocarditis, liver impairment, and kidney impairment; multiorgan dysfunction, in other words,” said William Shaffner, MD, a professor of preventive medicine and infectious diseases at Vanderbilt University Medical Center, Nashville, Tenn.
But in some cases, the virus might simply be a bystander to an unrelated disease process; in others, the experimental treatments being used might cause problems. Indeed, cardiology groups recently warned of torsade de pointes – a dangerously abnormal heart rhythm – with hydroxychloroquine and azithromycin.
“We think it’s some combination,” but don’t really know, Dr. Krumholz said. In the meantime, “we are forced to treat patients by instinct and first principles,” and long-term sequelae are unknown. “We don’t want to be in this position for long.”
To that end, he said, “this is the time for us all to hold hands and be together because we need to learn rapidly from each other. Our job is both to care for the people in front of us and make sure that they didn’t die in vain, that the experience they had is funneled into a larger set of data to make sure the next person is better off.”
Financial incentives affect the adoption of biosimilars
during the same time period in 2015-2019, according to an analysis published in Arthritis and Rheumatology.
The use of the biosimilars also was associated with cost savings at the VAMC, but not at the academic medical center, which illustrates that insufficient financial incentives can delay the adoption of biosimilars and the health care system’s realization of cost savings, according to the authors.
Medicare, which is not allowed to negotiate drug prices, is one of the largest payers for infused therapies. Medicare reimbursement for infused therapies is based on the latter’s average selling price (ASP) during the previous quarter. Institutions may negotiate purchase prices with drug manufacturers and receive Medicare reimbursement. Biosimilars generally have lower ASPs than their corresponding reference therapies, and biosimilar manufacturers may have less room to negotiate prices than reference therapy manufacturers. Consequently, a given institution might have a greater incentive to use reference products than to use biosimilars.
An examination of pharmacy data
The VA negotiates drug prices for all of its medical centers and has mandated that clinicians prefer biosimilars to their corresponding reference therapies, so Joshua F. Baker, MD, of the University of Pennsylvania and the Corporal Michael J. Crescenz VAMC, both in Philadelphia, and his colleagues hypothesized that the adoption of biosimilars had proceeded more quickly at a VAMC than at a nearby academic medical center.
The investigators examined pharmacy data from the University of Pennsylvania Health System (UPHS) electronic medical record and the Corporal Michael J. Crescenz VAMC to compare the frequency of prescribing biosimilars at these sites between Jan. 1, 2015, and May 31, 2019. Dr. Baker and his associates focused specifically on reference infliximab (Remicade) and the reference noninfusion therapies filgrastim (Neupogen) and pegfilgrastim (Neulasta) and on biosimilars of these therapies (infliximab-dyyb [Inflectra], infliximab-abda [Renflexis], filgrastim-sndz [Zarxio], and pegfilgrastim-jmdb [Fulphila]).
Because Medicare was the predominant payer, the researchers estimated reimbursement for reference and biosimilar infliximabs according to the Medicare Part B reimbursement policy. They defined an institution’s incentive to use a given therapy as the difference between the reimbursement and acquisition cost for that therapy. Dr. Baker and colleagues compared the incentives for UPHS with those for the VAMC.
VAMC saved 81% of reference product cost
The researchers identified 15,761 infusions of infliximab at UPHS and 446 at the VAMC during the study period. The proportion of infusions that used the reference product was 99% at UPHS and 62% at the VAMC. ASPs for biosimilar infliximab have been consistently lower than those for the reference product since July 2017. In December 2017, the VAMC switched to the biosimilar infliximab.
Institutional incentives based on Medicare Part B reimbursement and acquisitions costs for reference and biosimilar infliximab have been similar since 2018. In 2019, the institutional incentive favored the reference product by $49-$64 per 100-mg vial. But at the VAMC, the cost per 100-mg vial was $623.48 for the reference product and $115.58 for the biosimilar Renflexis. Purchasing the biosimilar thus yielded a savings of 81%. The current costs for the therapies are $546 and $116, respectively.
In addition, Dr. Baker and colleagues identified 46,683 orders for filgrastim or pegfilgrastim at UPHS. Approximately 90% of the orders were for either of the two reference products despite the ASP of biosimilar filgrastim being approximately 40% lower than that of its reference product. At the VAMC, about 88% of orders were for the reference products. Biosimilars became available in 2016. UPHS began using them at a modest rate, but their adoption was greater at the VAMC, which designated them as preferred products.
Tendering and a nationwide policy mandating use of biosimilars have resulted in financial savings for the VAMC, wrote Dr. Baker and colleagues. “These data suggest that, with current Medicare Part B reimbursement policy, the absence of financial incentives to encourage use of infliximab biosimilars has resulted in slower uptake of biosimilar use at institutions outside of the VA system. The implications of this are a slower reduction in costs to the health care system, since decreases in ASP over time are predicated on negotiations at the institutional level, which have been gradual and stepwise. ...
“Although some of our results may not be applicable to other geographical regions of the U.S., the comparison of two affiliated institutions in geographical proximity and with shared health care providers is a strength,” they continued. “Our findings should be replicated using national VAMC data or data from other health care systems.”
The researchers said that their findings may not apply to noninfused therapies, which are not covered under Medicare Part B, and they did not directly study the impact of pharmacy benefit managers. However, they noted that their data on filgrastim and pegfilgrastim support the hypothesis that pharmacy benefit managers receive “incentives that continue to promote the use of reference products that have higher manufacturer’s list prices, which likely will limit the uptake of both infused and injectable biosimilar therapies over time.” They said that “this finding has important implications for when noninfused biosimilars (e.g. etanercept and adalimumab) are eventually introduced to the U.S. market.”
European governments incentivize use of biosimilars
Government and institutional incentives have increased the adoption of biosimilars in Europe, wrote Guro Lovik Goll, MD, and Tore Kristian Kvien, MD, of the department of rheumatology at Diakonhjemmet Hospital in Oslo, in an accompanying editorial. Norway and Denmark have annual national tender systems in which biosimilars and reference products compete. The price of infliximab biosimilar was 39% lower than the reference product in 2014 and 69% lower in 2015. “Competition has caused dramatically lower prices both for biosimilars and also for the originator drugs competing with them,” wrote the authors.
In 2015, the government of Denmark mandated that patients on infliximab be switched to a biosimilar, and patients in Norway also have been switched to biosimilars. The use of etanercept in Norway increased by 40% from 2016 to 2019, and the use of infliximab has increased by more than threefold since 2015. “In Norway, the consequence of competition, national tenders, and availability of biosimilars have led to better access to therapy for more people in need of biologic drugs, while at the same time showing a total cost reduction of biologics for use in rheumatology, gastroenterology, and dermatology,” wrote the authors.
Health care costs $10,000 per capita in the United States, compared with $5,300 for other wealthy countries in the Organization for Economic Cooperation and Development. Low life expectancy and high infant mortality in the U.S. indicate that high costs are not associated with better outcomes. “As Americans seem to lose out on the cost-cutting potential of biosimilars, this missed opportunity is set to get even more expensive,” the authors concluded.
The U.S. Department of Veterans Affairs, the National Institutes of Health, and the American Diabetes Association contributed funding for the study. Dr. Baker reported receiving consulting fees from Bristol-Myers Squibb and Gilead, and another author reported receiving research support paid to his institution by Pfizer and UCB, as well as receiving consulting fees from nine pharmaceutical companies. Dr. Goll and Dr. Kvien both reported receiving fees for speaking and/or consulting from numerous pharmaceutical companies, including Pfizer.
SOURCES: Baker JF et al. Arthritis Rheumatol. 2020 Apr 6. doi: 10.1002/art.41277.
during the same time period in 2015-2019, according to an analysis published in Arthritis and Rheumatology.
The use of the biosimilars also was associated with cost savings at the VAMC, but not at the academic medical center, which illustrates that insufficient financial incentives can delay the adoption of biosimilars and the health care system’s realization of cost savings, according to the authors.
Medicare, which is not allowed to negotiate drug prices, is one of the largest payers for infused therapies. Medicare reimbursement for infused therapies is based on the latter’s average selling price (ASP) during the previous quarter. Institutions may negotiate purchase prices with drug manufacturers and receive Medicare reimbursement. Biosimilars generally have lower ASPs than their corresponding reference therapies, and biosimilar manufacturers may have less room to negotiate prices than reference therapy manufacturers. Consequently, a given institution might have a greater incentive to use reference products than to use biosimilars.
An examination of pharmacy data
The VA negotiates drug prices for all of its medical centers and has mandated that clinicians prefer biosimilars to their corresponding reference therapies, so Joshua F. Baker, MD, of the University of Pennsylvania and the Corporal Michael J. Crescenz VAMC, both in Philadelphia, and his colleagues hypothesized that the adoption of biosimilars had proceeded more quickly at a VAMC than at a nearby academic medical center.
The investigators examined pharmacy data from the University of Pennsylvania Health System (UPHS) electronic medical record and the Corporal Michael J. Crescenz VAMC to compare the frequency of prescribing biosimilars at these sites between Jan. 1, 2015, and May 31, 2019. Dr. Baker and his associates focused specifically on reference infliximab (Remicade) and the reference noninfusion therapies filgrastim (Neupogen) and pegfilgrastim (Neulasta) and on biosimilars of these therapies (infliximab-dyyb [Inflectra], infliximab-abda [Renflexis], filgrastim-sndz [Zarxio], and pegfilgrastim-jmdb [Fulphila]).
Because Medicare was the predominant payer, the researchers estimated reimbursement for reference and biosimilar infliximabs according to the Medicare Part B reimbursement policy. They defined an institution’s incentive to use a given therapy as the difference between the reimbursement and acquisition cost for that therapy. Dr. Baker and colleagues compared the incentives for UPHS with those for the VAMC.
VAMC saved 81% of reference product cost
The researchers identified 15,761 infusions of infliximab at UPHS and 446 at the VAMC during the study period. The proportion of infusions that used the reference product was 99% at UPHS and 62% at the VAMC. ASPs for biosimilar infliximab have been consistently lower than those for the reference product since July 2017. In December 2017, the VAMC switched to the biosimilar infliximab.
Institutional incentives based on Medicare Part B reimbursement and acquisitions costs for reference and biosimilar infliximab have been similar since 2018. In 2019, the institutional incentive favored the reference product by $49-$64 per 100-mg vial. But at the VAMC, the cost per 100-mg vial was $623.48 for the reference product and $115.58 for the biosimilar Renflexis. Purchasing the biosimilar thus yielded a savings of 81%. The current costs for the therapies are $546 and $116, respectively.
In addition, Dr. Baker and colleagues identified 46,683 orders for filgrastim or pegfilgrastim at UPHS. Approximately 90% of the orders were for either of the two reference products despite the ASP of biosimilar filgrastim being approximately 40% lower than that of its reference product. At the VAMC, about 88% of orders were for the reference products. Biosimilars became available in 2016. UPHS began using them at a modest rate, but their adoption was greater at the VAMC, which designated them as preferred products.
Tendering and a nationwide policy mandating use of biosimilars have resulted in financial savings for the VAMC, wrote Dr. Baker and colleagues. “These data suggest that, with current Medicare Part B reimbursement policy, the absence of financial incentives to encourage use of infliximab biosimilars has resulted in slower uptake of biosimilar use at institutions outside of the VA system. The implications of this are a slower reduction in costs to the health care system, since decreases in ASP over time are predicated on negotiations at the institutional level, which have been gradual and stepwise. ...
“Although some of our results may not be applicable to other geographical regions of the U.S., the comparison of two affiliated institutions in geographical proximity and with shared health care providers is a strength,” they continued. “Our findings should be replicated using national VAMC data or data from other health care systems.”
The researchers said that their findings may not apply to noninfused therapies, which are not covered under Medicare Part B, and they did not directly study the impact of pharmacy benefit managers. However, they noted that their data on filgrastim and pegfilgrastim support the hypothesis that pharmacy benefit managers receive “incentives that continue to promote the use of reference products that have higher manufacturer’s list prices, which likely will limit the uptake of both infused and injectable biosimilar therapies over time.” They said that “this finding has important implications for when noninfused biosimilars (e.g. etanercept and adalimumab) are eventually introduced to the U.S. market.”
European governments incentivize use of biosimilars
Government and institutional incentives have increased the adoption of biosimilars in Europe, wrote Guro Lovik Goll, MD, and Tore Kristian Kvien, MD, of the department of rheumatology at Diakonhjemmet Hospital in Oslo, in an accompanying editorial. Norway and Denmark have annual national tender systems in which biosimilars and reference products compete. The price of infliximab biosimilar was 39% lower than the reference product in 2014 and 69% lower in 2015. “Competition has caused dramatically lower prices both for biosimilars and also for the originator drugs competing with them,” wrote the authors.
In 2015, the government of Denmark mandated that patients on infliximab be switched to a biosimilar, and patients in Norway also have been switched to biosimilars. The use of etanercept in Norway increased by 40% from 2016 to 2019, and the use of infliximab has increased by more than threefold since 2015. “In Norway, the consequence of competition, national tenders, and availability of biosimilars have led to better access to therapy for more people in need of biologic drugs, while at the same time showing a total cost reduction of biologics for use in rheumatology, gastroenterology, and dermatology,” wrote the authors.
Health care costs $10,000 per capita in the United States, compared with $5,300 for other wealthy countries in the Organization for Economic Cooperation and Development. Low life expectancy and high infant mortality in the U.S. indicate that high costs are not associated with better outcomes. “As Americans seem to lose out on the cost-cutting potential of biosimilars, this missed opportunity is set to get even more expensive,” the authors concluded.
The U.S. Department of Veterans Affairs, the National Institutes of Health, and the American Diabetes Association contributed funding for the study. Dr. Baker reported receiving consulting fees from Bristol-Myers Squibb and Gilead, and another author reported receiving research support paid to his institution by Pfizer and UCB, as well as receiving consulting fees from nine pharmaceutical companies. Dr. Goll and Dr. Kvien both reported receiving fees for speaking and/or consulting from numerous pharmaceutical companies, including Pfizer.
SOURCES: Baker JF et al. Arthritis Rheumatol. 2020 Apr 6. doi: 10.1002/art.41277.
during the same time period in 2015-2019, according to an analysis published in Arthritis and Rheumatology.
The use of the biosimilars also was associated with cost savings at the VAMC, but not at the academic medical center, which illustrates that insufficient financial incentives can delay the adoption of biosimilars and the health care system’s realization of cost savings, according to the authors.
Medicare, which is not allowed to negotiate drug prices, is one of the largest payers for infused therapies. Medicare reimbursement for infused therapies is based on the latter’s average selling price (ASP) during the previous quarter. Institutions may negotiate purchase prices with drug manufacturers and receive Medicare reimbursement. Biosimilars generally have lower ASPs than their corresponding reference therapies, and biosimilar manufacturers may have less room to negotiate prices than reference therapy manufacturers. Consequently, a given institution might have a greater incentive to use reference products than to use biosimilars.
An examination of pharmacy data
The VA negotiates drug prices for all of its medical centers and has mandated that clinicians prefer biosimilars to their corresponding reference therapies, so Joshua F. Baker, MD, of the University of Pennsylvania and the Corporal Michael J. Crescenz VAMC, both in Philadelphia, and his colleagues hypothesized that the adoption of biosimilars had proceeded more quickly at a VAMC than at a nearby academic medical center.
The investigators examined pharmacy data from the University of Pennsylvania Health System (UPHS) electronic medical record and the Corporal Michael J. Crescenz VAMC to compare the frequency of prescribing biosimilars at these sites between Jan. 1, 2015, and May 31, 2019. Dr. Baker and his associates focused specifically on reference infliximab (Remicade) and the reference noninfusion therapies filgrastim (Neupogen) and pegfilgrastim (Neulasta) and on biosimilars of these therapies (infliximab-dyyb [Inflectra], infliximab-abda [Renflexis], filgrastim-sndz [Zarxio], and pegfilgrastim-jmdb [Fulphila]).
Because Medicare was the predominant payer, the researchers estimated reimbursement for reference and biosimilar infliximabs according to the Medicare Part B reimbursement policy. They defined an institution’s incentive to use a given therapy as the difference between the reimbursement and acquisition cost for that therapy. Dr. Baker and colleagues compared the incentives for UPHS with those for the VAMC.
VAMC saved 81% of reference product cost
The researchers identified 15,761 infusions of infliximab at UPHS and 446 at the VAMC during the study period. The proportion of infusions that used the reference product was 99% at UPHS and 62% at the VAMC. ASPs for biosimilar infliximab have been consistently lower than those for the reference product since July 2017. In December 2017, the VAMC switched to the biosimilar infliximab.
Institutional incentives based on Medicare Part B reimbursement and acquisitions costs for reference and biosimilar infliximab have been similar since 2018. In 2019, the institutional incentive favored the reference product by $49-$64 per 100-mg vial. But at the VAMC, the cost per 100-mg vial was $623.48 for the reference product and $115.58 for the biosimilar Renflexis. Purchasing the biosimilar thus yielded a savings of 81%. The current costs for the therapies are $546 and $116, respectively.
In addition, Dr. Baker and colleagues identified 46,683 orders for filgrastim or pegfilgrastim at UPHS. Approximately 90% of the orders were for either of the two reference products despite the ASP of biosimilar filgrastim being approximately 40% lower than that of its reference product. At the VAMC, about 88% of orders were for the reference products. Biosimilars became available in 2016. UPHS began using them at a modest rate, but their adoption was greater at the VAMC, which designated them as preferred products.
Tendering and a nationwide policy mandating use of biosimilars have resulted in financial savings for the VAMC, wrote Dr. Baker and colleagues. “These data suggest that, with current Medicare Part B reimbursement policy, the absence of financial incentives to encourage use of infliximab biosimilars has resulted in slower uptake of biosimilar use at institutions outside of the VA system. The implications of this are a slower reduction in costs to the health care system, since decreases in ASP over time are predicated on negotiations at the institutional level, which have been gradual and stepwise. ...
“Although some of our results may not be applicable to other geographical regions of the U.S., the comparison of two affiliated institutions in geographical proximity and with shared health care providers is a strength,” they continued. “Our findings should be replicated using national VAMC data or data from other health care systems.”
The researchers said that their findings may not apply to noninfused therapies, which are not covered under Medicare Part B, and they did not directly study the impact of pharmacy benefit managers. However, they noted that their data on filgrastim and pegfilgrastim support the hypothesis that pharmacy benefit managers receive “incentives that continue to promote the use of reference products that have higher manufacturer’s list prices, which likely will limit the uptake of both infused and injectable biosimilar therapies over time.” They said that “this finding has important implications for when noninfused biosimilars (e.g. etanercept and adalimumab) are eventually introduced to the U.S. market.”
European governments incentivize use of biosimilars
Government and institutional incentives have increased the adoption of biosimilars in Europe, wrote Guro Lovik Goll, MD, and Tore Kristian Kvien, MD, of the department of rheumatology at Diakonhjemmet Hospital in Oslo, in an accompanying editorial. Norway and Denmark have annual national tender systems in which biosimilars and reference products compete. The price of infliximab biosimilar was 39% lower than the reference product in 2014 and 69% lower in 2015. “Competition has caused dramatically lower prices both for biosimilars and also for the originator drugs competing with them,” wrote the authors.
In 2015, the government of Denmark mandated that patients on infliximab be switched to a biosimilar, and patients in Norway also have been switched to biosimilars. The use of etanercept in Norway increased by 40% from 2016 to 2019, and the use of infliximab has increased by more than threefold since 2015. “In Norway, the consequence of competition, national tenders, and availability of biosimilars have led to better access to therapy for more people in need of biologic drugs, while at the same time showing a total cost reduction of biologics for use in rheumatology, gastroenterology, and dermatology,” wrote the authors.
Health care costs $10,000 per capita in the United States, compared with $5,300 for other wealthy countries in the Organization for Economic Cooperation and Development. Low life expectancy and high infant mortality in the U.S. indicate that high costs are not associated with better outcomes. “As Americans seem to lose out on the cost-cutting potential of biosimilars, this missed opportunity is set to get even more expensive,” the authors concluded.
The U.S. Department of Veterans Affairs, the National Institutes of Health, and the American Diabetes Association contributed funding for the study. Dr. Baker reported receiving consulting fees from Bristol-Myers Squibb and Gilead, and another author reported receiving research support paid to his institution by Pfizer and UCB, as well as receiving consulting fees from nine pharmaceutical companies. Dr. Goll and Dr. Kvien both reported receiving fees for speaking and/or consulting from numerous pharmaceutical companies, including Pfizer.
SOURCES: Baker JF et al. Arthritis Rheumatol. 2020 Apr 6. doi: 10.1002/art.41277.
FROM ARTHRITIS & RHEUMATOLOGY
COVID-19 PPE-related skin effects described in survey of Chinese doctors, nurses
Almost 75% of doctors and nurses in and around Wuhan, China, where the outbreak first emerged, reported skin problems during a single week in early February 2020, in a survey of health care workers (HCW) caring for COVID-19 patients at five university and five regional hospitals. Hands, cheeks, and the nasal bridge were the most commonly affected areas, with skin dryness, maceration, papules, and erythema the most common problems, according to research published in the British Journal of Dermatology.
In New York City, masks in particular are “really an issue,” said Ellen Marmur, MD, a dermatologist in private practice and an associate clinical professor at the Mount Sinai School of Medicine, New York.
She’s dealing with patients who have abrasions and skin infections at the tip of the nose, bruising from the metal strap that goes across the bridge of the nose, and skin irritation from the straps. “Rosacea is [also] definitely flaring up, [and] people’s acne is definitely flaring up, not only because of the stress, but because of the sweat and humidity” that builds up under the masks, she said.
“It’s not a life-threatening thing, but it’s definitely something we’ve been helping people with,” she said. This includes her husband, a cardiologist pulling 12-hour shifts in a New York City hospital wearing an N95 mask; when he comes home, the tip of his nose is red and abraded.
Treatment entails first aid skin care: a dab of a gentle ointment like Aquaphor to prevent abrasions while the mask is on and to help them heal after it’s off, and bacitracin if infection is a worry. For acne and rosacea flares, a course of minocycline or topical clindamycin might help, Dr. Marmur said.
Although almost 75% of the doctors and nurses in the Chinese study reported skin problems, the response rate was low, just 376 of the 1,000 surveyed (37.6%). That might have tilted the results to providers who actually ran into problems, wrote the investigators, led by Ping Lin of the department of dermatology and venereology at Peking University First Hospital, Beijing.
Still, 280 (74.5%) reported adverse skin reactions from caring for COVID-19 patients. “Of note, this rate was much higher than the rate of occupational contact dermatitis (31.5%) in HCWs under normal working condition[s], and that of adverse skin reactions (21.4%-35.5%)” during the outbreak of another coronavirus in 2003, severe acute respiratory syndrome, they wrote.
Most providers in the study washed their hands more than 10 times a day, but only about 22% applied hand cream afterwards, they reported.
On multivariate analysis, working in hospitals harder hit by the pandemic (odds ratio, 2.41; P = .001), working on inpatient wards (OR, 2.44; P = .003), wearing full-body personal protective equipment over 6 hours (OR, 4.26; P < .001), and female sex (OR, 1.87; P = .038) increased the risk of adverse skin reactions. The team suggested moisturizers would help to protect against hand dermatitis, and alcohol-based products instead of soaps “as the former show high antimicrobial activity and low risks of skin damage.” Also, “restricting duration of wearing” of protection gear “to no more than 6 hours would help.”
The study investigators reported that they had no conflicts of interest.
SOURCE: Lin P et al. Br J Dermatol. 2020 Apr 7. doi: 10.1111/bjd.19089.
Almost 75% of doctors and nurses in and around Wuhan, China, where the outbreak first emerged, reported skin problems during a single week in early February 2020, in a survey of health care workers (HCW) caring for COVID-19 patients at five university and five regional hospitals. Hands, cheeks, and the nasal bridge were the most commonly affected areas, with skin dryness, maceration, papules, and erythema the most common problems, according to research published in the British Journal of Dermatology.
In New York City, masks in particular are “really an issue,” said Ellen Marmur, MD, a dermatologist in private practice and an associate clinical professor at the Mount Sinai School of Medicine, New York.
She’s dealing with patients who have abrasions and skin infections at the tip of the nose, bruising from the metal strap that goes across the bridge of the nose, and skin irritation from the straps. “Rosacea is [also] definitely flaring up, [and] people’s acne is definitely flaring up, not only because of the stress, but because of the sweat and humidity” that builds up under the masks, she said.
“It’s not a life-threatening thing, but it’s definitely something we’ve been helping people with,” she said. This includes her husband, a cardiologist pulling 12-hour shifts in a New York City hospital wearing an N95 mask; when he comes home, the tip of his nose is red and abraded.
Treatment entails first aid skin care: a dab of a gentle ointment like Aquaphor to prevent abrasions while the mask is on and to help them heal after it’s off, and bacitracin if infection is a worry. For acne and rosacea flares, a course of minocycline or topical clindamycin might help, Dr. Marmur said.
Although almost 75% of the doctors and nurses in the Chinese study reported skin problems, the response rate was low, just 376 of the 1,000 surveyed (37.6%). That might have tilted the results to providers who actually ran into problems, wrote the investigators, led by Ping Lin of the department of dermatology and venereology at Peking University First Hospital, Beijing.
Still, 280 (74.5%) reported adverse skin reactions from caring for COVID-19 patients. “Of note, this rate was much higher than the rate of occupational contact dermatitis (31.5%) in HCWs under normal working condition[s], and that of adverse skin reactions (21.4%-35.5%)” during the outbreak of another coronavirus in 2003, severe acute respiratory syndrome, they wrote.
Most providers in the study washed their hands more than 10 times a day, but only about 22% applied hand cream afterwards, they reported.
On multivariate analysis, working in hospitals harder hit by the pandemic (odds ratio, 2.41; P = .001), working on inpatient wards (OR, 2.44; P = .003), wearing full-body personal protective equipment over 6 hours (OR, 4.26; P < .001), and female sex (OR, 1.87; P = .038) increased the risk of adverse skin reactions. The team suggested moisturizers would help to protect against hand dermatitis, and alcohol-based products instead of soaps “as the former show high antimicrobial activity and low risks of skin damage.” Also, “restricting duration of wearing” of protection gear “to no more than 6 hours would help.”
The study investigators reported that they had no conflicts of interest.
SOURCE: Lin P et al. Br J Dermatol. 2020 Apr 7. doi: 10.1111/bjd.19089.
Almost 75% of doctors and nurses in and around Wuhan, China, where the outbreak first emerged, reported skin problems during a single week in early February 2020, in a survey of health care workers (HCW) caring for COVID-19 patients at five university and five regional hospitals. Hands, cheeks, and the nasal bridge were the most commonly affected areas, with skin dryness, maceration, papules, and erythema the most common problems, according to research published in the British Journal of Dermatology.
In New York City, masks in particular are “really an issue,” said Ellen Marmur, MD, a dermatologist in private practice and an associate clinical professor at the Mount Sinai School of Medicine, New York.
She’s dealing with patients who have abrasions and skin infections at the tip of the nose, bruising from the metal strap that goes across the bridge of the nose, and skin irritation from the straps. “Rosacea is [also] definitely flaring up, [and] people’s acne is definitely flaring up, not only because of the stress, but because of the sweat and humidity” that builds up under the masks, she said.
“It’s not a life-threatening thing, but it’s definitely something we’ve been helping people with,” she said. This includes her husband, a cardiologist pulling 12-hour shifts in a New York City hospital wearing an N95 mask; when he comes home, the tip of his nose is red and abraded.
Treatment entails first aid skin care: a dab of a gentle ointment like Aquaphor to prevent abrasions while the mask is on and to help them heal after it’s off, and bacitracin if infection is a worry. For acne and rosacea flares, a course of minocycline or topical clindamycin might help, Dr. Marmur said.
Although almost 75% of the doctors and nurses in the Chinese study reported skin problems, the response rate was low, just 376 of the 1,000 surveyed (37.6%). That might have tilted the results to providers who actually ran into problems, wrote the investigators, led by Ping Lin of the department of dermatology and venereology at Peking University First Hospital, Beijing.
Still, 280 (74.5%) reported adverse skin reactions from caring for COVID-19 patients. “Of note, this rate was much higher than the rate of occupational contact dermatitis (31.5%) in HCWs under normal working condition[s], and that of adverse skin reactions (21.4%-35.5%)” during the outbreak of another coronavirus in 2003, severe acute respiratory syndrome, they wrote.
Most providers in the study washed their hands more than 10 times a day, but only about 22% applied hand cream afterwards, they reported.
On multivariate analysis, working in hospitals harder hit by the pandemic (odds ratio, 2.41; P = .001), working on inpatient wards (OR, 2.44; P = .003), wearing full-body personal protective equipment over 6 hours (OR, 4.26; P < .001), and female sex (OR, 1.87; P = .038) increased the risk of adverse skin reactions. The team suggested moisturizers would help to protect against hand dermatitis, and alcohol-based products instead of soaps “as the former show high antimicrobial activity and low risks of skin damage.” Also, “restricting duration of wearing” of protection gear “to no more than 6 hours would help.”
The study investigators reported that they had no conflicts of interest.
SOURCE: Lin P et al. Br J Dermatol. 2020 Apr 7. doi: 10.1111/bjd.19089.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
What's your diagnosis?
A punch biopsy of one of the lesions showed a superficial and deep mixed inflammatory cell infiltrate, including neutrophils and eosinophils. There was also vasculitis, karyorrhexis and extravasated red blood cells. The findings are those of leukocytoclastic vasculitis, suggestive of acute hemorrhagic edema of infancy. Direct immunofluorescence was positive for IgM, C3, and fibrinogen, but negative for IgA.
Acute hemorrhagic edema of infancy (AHEI), also known as Finkelstein disease, is form of leukocytoclastic vasculitis that occurs in infants and toddlers aged between4 months and 3 years.
The lesions start as petechiae or edematous, erythematous to violaceous nodules that later coalesce and form “cockade”-like plaques with a central clearing on the face and extremities. Gastrointestinal, renal, and joint involvement are rare.1 AHEI follows a benign course with resolution of the lesions and symptoms within days to weeks. The etiology of this condition is not known but infection triggers have been reported including coronavirus infections, coxsackie virus infections, Escherichia coli urinary tract infections, herpes simplex virus stomatitis, and pneumococcal bacteremia.2,3 Our patient had a prior history of pneumococcal pneumonia and metapneumovirus infection. MMR vaccine also has been reported as a possible trigger, as well as some medications.
Laboratory results are usually normal, but some patients may have elevated inflammatory markers (C-reactive protein and erythrocyte sedimentation rate), as noted in our patient, and leukocytosis, thrombocytosis, and eosinophilia. Microscopic analysis demonstrates leukocytoclastic vasculitis of small vessels with associated karyorrhexis and extravasated red blood cells.
The differential diagnosis includes other vasculitic conditions, primarily Henoch-Schönlein purpura (HSP). Patients with HSP tend to be older in age and the lesions described as palpable purpura commonly affect the lower extremities and buttocks. These patients can present with abdominal pain and arthritis; renal compromise also can occur. Direct immunofluorescence can commonly be positive for IgA, which was negative in our patient.
AHEI and HSP are considered different entities, but both present with leukocytoclastic vasculitis.1 Another condition to consider in patients with fever, rash, and edema is Kawasaki disease, also a form of vasculitis, that affects small- and medium-size muscular vessels with predilection for the coronary arteries. Patients with Kawasaki disease present with fever (usually longer than 5 days), facial and extremity edema (similar to AHEI), skin lesions (which may have multiple presentations, the most common being macular, papular and erythematous, and urticarial eruptions), but also lymphadenopathy and conjunctivitis. These patients appear sicker than children with AHEI. Their laboratory results show leukocytosis, thrombocytosis or thrombocytopenia, elevated inflammatory markers, and sterile pyuria.4
Patients with erythema nodosum present with tender erythematous nodules, which can look like early AHEI lesions. The most common location is the lower extremities, but in children erythema nodosum can occur on the face, trunk, and arms. The lesions can occur secondary to infections such as streptococcus, mycoplasma, tuberculosis, coccidioidomycosis, and sarcoidosis, as well as to malignancy or medications. These patients do not appear sick, are not febrile, and are rarely seen under 2 years of age.5
Acute febrile neutrophilic dermatosis – Sweets’ syndrome – also should be considered in a patient with tender nodules, fever, and leukocytosis. The skin lesions in Sweets’ syndrome, compared with those in AHEI, are painful and can present as papules, nodules, and bullae on the face and extremities. A prior history of an upper respiratory infection is commonly described in children with Sweets’ syndrome. These patients present with fever, which may start days to weeks prior to the lesions starting. Children with Sweets’ syndrome also can have conjunctivitis, myalgias, polyarthritis, and in severe cases septic shock and multiorgan dysfunction. Sweets’ syndrome can be seen in patients with inflammatory bowel disease, systemic lupus erythematosus, chronic multifocal osteomyelitis, and malignancy; it also may be induced by certain medications.6
As mentioned above, the course of AHEI is benign, and the condition resolves within days to weeks. Treatment is supportive.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She had no relevant financial disclosures. Email Dr. Matiz at pdnews@mdedge.com.
References
1. F1000Res. 2019;8:1771.
2. Pediatr Dermatol. 2006 Jul-Aug;23(4):361-4.
3. Pediatr Dermatol. 2015 Nov-Dec;32(6):e309-11.
4. Clin Dermatol. 2017 Nov-Dec;35(6):530-40.
5. Yonsei Med J. 2019 Mar;60(3):312-4.
6. Pediatr Dermatol. 2015 Jul-Aug;32(4):437-46.
A punch biopsy of one of the lesions showed a superficial and deep mixed inflammatory cell infiltrate, including neutrophils and eosinophils. There was also vasculitis, karyorrhexis and extravasated red blood cells. The findings are those of leukocytoclastic vasculitis, suggestive of acute hemorrhagic edema of infancy. Direct immunofluorescence was positive for IgM, C3, and fibrinogen, but negative for IgA.
Acute hemorrhagic edema of infancy (AHEI), also known as Finkelstein disease, is form of leukocytoclastic vasculitis that occurs in infants and toddlers aged between4 months and 3 years.
The lesions start as petechiae or edematous, erythematous to violaceous nodules that later coalesce and form “cockade”-like plaques with a central clearing on the face and extremities. Gastrointestinal, renal, and joint involvement are rare.1 AHEI follows a benign course with resolution of the lesions and symptoms within days to weeks. The etiology of this condition is not known but infection triggers have been reported including coronavirus infections, coxsackie virus infections, Escherichia coli urinary tract infections, herpes simplex virus stomatitis, and pneumococcal bacteremia.2,3 Our patient had a prior history of pneumococcal pneumonia and metapneumovirus infection. MMR vaccine also has been reported as a possible trigger, as well as some medications.
Laboratory results are usually normal, but some patients may have elevated inflammatory markers (C-reactive protein and erythrocyte sedimentation rate), as noted in our patient, and leukocytosis, thrombocytosis, and eosinophilia. Microscopic analysis demonstrates leukocytoclastic vasculitis of small vessels with associated karyorrhexis and extravasated red blood cells.
The differential diagnosis includes other vasculitic conditions, primarily Henoch-Schönlein purpura (HSP). Patients with HSP tend to be older in age and the lesions described as palpable purpura commonly affect the lower extremities and buttocks. These patients can present with abdominal pain and arthritis; renal compromise also can occur. Direct immunofluorescence can commonly be positive for IgA, which was negative in our patient.
AHEI and HSP are considered different entities, but both present with leukocytoclastic vasculitis.1 Another condition to consider in patients with fever, rash, and edema is Kawasaki disease, also a form of vasculitis, that affects small- and medium-size muscular vessels with predilection for the coronary arteries. Patients with Kawasaki disease present with fever (usually longer than 5 days), facial and extremity edema (similar to AHEI), skin lesions (which may have multiple presentations, the most common being macular, papular and erythematous, and urticarial eruptions), but also lymphadenopathy and conjunctivitis. These patients appear sicker than children with AHEI. Their laboratory results show leukocytosis, thrombocytosis or thrombocytopenia, elevated inflammatory markers, and sterile pyuria.4
Patients with erythema nodosum present with tender erythematous nodules, which can look like early AHEI lesions. The most common location is the lower extremities, but in children erythema nodosum can occur on the face, trunk, and arms. The lesions can occur secondary to infections such as streptococcus, mycoplasma, tuberculosis, coccidioidomycosis, and sarcoidosis, as well as to malignancy or medications. These patients do not appear sick, are not febrile, and are rarely seen under 2 years of age.5
Acute febrile neutrophilic dermatosis – Sweets’ syndrome – also should be considered in a patient with tender nodules, fever, and leukocytosis. The skin lesions in Sweets’ syndrome, compared with those in AHEI, are painful and can present as papules, nodules, and bullae on the face and extremities. A prior history of an upper respiratory infection is commonly described in children with Sweets’ syndrome. These patients present with fever, which may start days to weeks prior to the lesions starting. Children with Sweets’ syndrome also can have conjunctivitis, myalgias, polyarthritis, and in severe cases septic shock and multiorgan dysfunction. Sweets’ syndrome can be seen in patients with inflammatory bowel disease, systemic lupus erythematosus, chronic multifocal osteomyelitis, and malignancy; it also may be induced by certain medications.6
As mentioned above, the course of AHEI is benign, and the condition resolves within days to weeks. Treatment is supportive.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She had no relevant financial disclosures. Email Dr. Matiz at pdnews@mdedge.com.
References
1. F1000Res. 2019;8:1771.
2. Pediatr Dermatol. 2006 Jul-Aug;23(4):361-4.
3. Pediatr Dermatol. 2015 Nov-Dec;32(6):e309-11.
4. Clin Dermatol. 2017 Nov-Dec;35(6):530-40.
5. Yonsei Med J. 2019 Mar;60(3):312-4.
6. Pediatr Dermatol. 2015 Jul-Aug;32(4):437-46.
A punch biopsy of one of the lesions showed a superficial and deep mixed inflammatory cell infiltrate, including neutrophils and eosinophils. There was also vasculitis, karyorrhexis and extravasated red blood cells. The findings are those of leukocytoclastic vasculitis, suggestive of acute hemorrhagic edema of infancy. Direct immunofluorescence was positive for IgM, C3, and fibrinogen, but negative for IgA.
Acute hemorrhagic edema of infancy (AHEI), also known as Finkelstein disease, is form of leukocytoclastic vasculitis that occurs in infants and toddlers aged between4 months and 3 years.
The lesions start as petechiae or edematous, erythematous to violaceous nodules that later coalesce and form “cockade”-like plaques with a central clearing on the face and extremities. Gastrointestinal, renal, and joint involvement are rare.1 AHEI follows a benign course with resolution of the lesions and symptoms within days to weeks. The etiology of this condition is not known but infection triggers have been reported including coronavirus infections, coxsackie virus infections, Escherichia coli urinary tract infections, herpes simplex virus stomatitis, and pneumococcal bacteremia.2,3 Our patient had a prior history of pneumococcal pneumonia and metapneumovirus infection. MMR vaccine also has been reported as a possible trigger, as well as some medications.
Laboratory results are usually normal, but some patients may have elevated inflammatory markers (C-reactive protein and erythrocyte sedimentation rate), as noted in our patient, and leukocytosis, thrombocytosis, and eosinophilia. Microscopic analysis demonstrates leukocytoclastic vasculitis of small vessels with associated karyorrhexis and extravasated red blood cells.
The differential diagnosis includes other vasculitic conditions, primarily Henoch-Schönlein purpura (HSP). Patients with HSP tend to be older in age and the lesions described as palpable purpura commonly affect the lower extremities and buttocks. These patients can present with abdominal pain and arthritis; renal compromise also can occur. Direct immunofluorescence can commonly be positive for IgA, which was negative in our patient.
AHEI and HSP are considered different entities, but both present with leukocytoclastic vasculitis.1 Another condition to consider in patients with fever, rash, and edema is Kawasaki disease, also a form of vasculitis, that affects small- and medium-size muscular vessels with predilection for the coronary arteries. Patients with Kawasaki disease present with fever (usually longer than 5 days), facial and extremity edema (similar to AHEI), skin lesions (which may have multiple presentations, the most common being macular, papular and erythematous, and urticarial eruptions), but also lymphadenopathy and conjunctivitis. These patients appear sicker than children with AHEI. Their laboratory results show leukocytosis, thrombocytosis or thrombocytopenia, elevated inflammatory markers, and sterile pyuria.4
Patients with erythema nodosum present with tender erythematous nodules, which can look like early AHEI lesions. The most common location is the lower extremities, but in children erythema nodosum can occur on the face, trunk, and arms. The lesions can occur secondary to infections such as streptococcus, mycoplasma, tuberculosis, coccidioidomycosis, and sarcoidosis, as well as to malignancy or medications. These patients do not appear sick, are not febrile, and are rarely seen under 2 years of age.5
Acute febrile neutrophilic dermatosis – Sweets’ syndrome – also should be considered in a patient with tender nodules, fever, and leukocytosis. The skin lesions in Sweets’ syndrome, compared with those in AHEI, are painful and can present as papules, nodules, and bullae on the face and extremities. A prior history of an upper respiratory infection is commonly described in children with Sweets’ syndrome. These patients present with fever, which may start days to weeks prior to the lesions starting. Children with Sweets’ syndrome also can have conjunctivitis, myalgias, polyarthritis, and in severe cases septic shock and multiorgan dysfunction. Sweets’ syndrome can be seen in patients with inflammatory bowel disease, systemic lupus erythematosus, chronic multifocal osteomyelitis, and malignancy; it also may be induced by certain medications.6
As mentioned above, the course of AHEI is benign, and the condition resolves within days to weeks. Treatment is supportive.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She had no relevant financial disclosures. Email Dr. Matiz at pdnews@mdedge.com.
References
1. F1000Res. 2019;8:1771.
2. Pediatr Dermatol. 2006 Jul-Aug;23(4):361-4.
3. Pediatr Dermatol. 2015 Nov-Dec;32(6):e309-11.
4. Clin Dermatol. 2017 Nov-Dec;35(6):530-40.
5. Yonsei Med J. 2019 Mar;60(3):312-4.
6. Pediatr Dermatol. 2015 Jul-Aug;32(4):437-46.
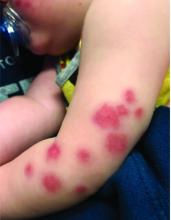
At 3 a.m., you receive a call from the ED for a baby with a new rash on the arms, legs, and face. Some of the lesions appear to be tender. He has a mild fever of 38.4° C (101.1° F) and is not in acute distress. He is drinking, but not eating much.
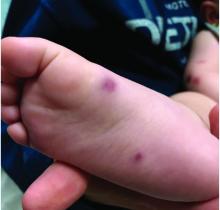
The parents also have noted some swelling on the hands and the feet. He has no upper respiratory or gastrointestinal symptoms. He is not walking yet.
He was admitted to the hospital 3 weeks prior for streptococcal pneumonia and metapneumovirus infection. He was treated with ceftriaxone, supportive respiratory care, and an albuterol inhaler. Influenza and respiratory syncytial virus tests were negative.
On physical exam, the child is tired and sleeping in his mom's arms. He has red and some purpuric papules on the face. On the arms and legs, he has purpuric papules and nodules. There is some edema on the face, hands, and feet. His conjunctiva is normal, and he has no oral lesions. He has no lymphadenopathy or hepatosplenomegaly.
Blood work shows normal complete blood count, coagulation tests, comprehensive metabolic panel, and urinalysis, but he has an elevated C-reactive protein of 114 mg/L and an elevated erythrocyte sedimentation rate of 71 mm/hour.
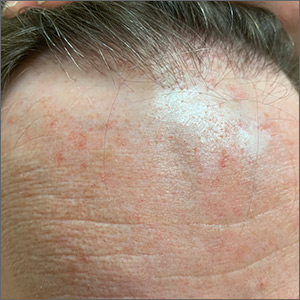
Receding hairline
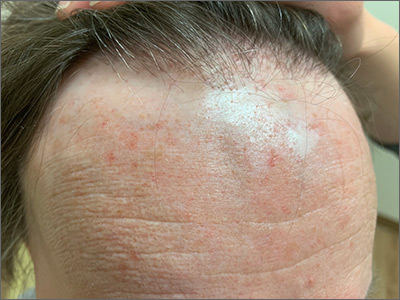
This patient had frontal fibrosing alopecia (FFA), a subtype of lichen planopilaris (LPP), or follicular lichen planus. LPP causes cicatricial (scarring) alopecia where the follicular epithelium is replaced with connective tissue and the hair follicle is permanently lost. LPP is caused by lymphocytic inflammation that initially presents as perifollicular erythema, with scale and keratotic plugs, and later progresses to scarring. If there is uncertainty in the diagnosis, biopsy can be helpful.
The LPP subtype, FFA, usually occurs in postmenopausal women. It follows a distinctive pattern, as in this patient, where the hair is progressively lost along the frontoparietal hair line (and sometimes the eyebrows). A careful physical examination reveals smooth skin where follicles are lost and there is erythema around the base of the hairs due to active inflammation and keratotic plugging. The specific mechanism of FFA is poorly understood, and hormones may play a role, in addition to the inflammatory response.
The goal of treatment is to arrest the progression of additional hair loss (which usually is permanent). Intralesional steroid injections, which also are used for alopecia areata, are the most common therapy. Triamcinolone 2.5 to 5 mg/mL is injected in the affected dermal layer of the scalp. Oral finasteride (a 5-alpha-reductase inhibitor to decrease androgens) 1 mg/d can be helpful, as can oral hydroxychloroquine 200 mg bid.
Once the inflammation has subsided, treatment can be discontinued. Hair transplantation has been used, but often fails due to the inflammatory scarring process. Our patient noted that her disease process had been stable and declined treatment.
Photo courtesy of Daniel Stulberg, MD, FAAFP. Text courtesy of Rory Aufderheide, MD, and Daniel Stulberg, MD, FAAFP. Drs. Stulberg and Aufderheide are from the Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
To D, Beecker J. Frontal fibrosing alopecia: update and review of challenges and successes. J Cutan Med Surg. 2018;22:182-189.

This patient had frontal fibrosing alopecia (FFA), a subtype of lichen planopilaris (LPP), or follicular lichen planus. LPP causes cicatricial (scarring) alopecia where the follicular epithelium is replaced with connective tissue and the hair follicle is permanently lost. LPP is caused by lymphocytic inflammation that initially presents as perifollicular erythema, with scale and keratotic plugs, and later progresses to scarring. If there is uncertainty in the diagnosis, biopsy can be helpful.
The LPP subtype, FFA, usually occurs in postmenopausal women. It follows a distinctive pattern, as in this patient, where the hair is progressively lost along the frontoparietal hair line (and sometimes the eyebrows). A careful physical examination reveals smooth skin where follicles are lost and there is erythema around the base of the hairs due to active inflammation and keratotic plugging. The specific mechanism of FFA is poorly understood, and hormones may play a role, in addition to the inflammatory response.
The goal of treatment is to arrest the progression of additional hair loss (which usually is permanent). Intralesional steroid injections, which also are used for alopecia areata, are the most common therapy. Triamcinolone 2.5 to 5 mg/mL is injected in the affected dermal layer of the scalp. Oral finasteride (a 5-alpha-reductase inhibitor to decrease androgens) 1 mg/d can be helpful, as can oral hydroxychloroquine 200 mg bid.
Once the inflammation has subsided, treatment can be discontinued. Hair transplantation has been used, but often fails due to the inflammatory scarring process. Our patient noted that her disease process had been stable and declined treatment.
Photo courtesy of Daniel Stulberg, MD, FAAFP. Text courtesy of Rory Aufderheide, MD, and Daniel Stulberg, MD, FAAFP. Drs. Stulberg and Aufderheide are from the Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This patient had frontal fibrosing alopecia (FFA), a subtype of lichen planopilaris (LPP), or follicular lichen planus. LPP causes cicatricial (scarring) alopecia where the follicular epithelium is replaced with connective tissue and the hair follicle is permanently lost. LPP is caused by lymphocytic inflammation that initially presents as perifollicular erythema, with scale and keratotic plugs, and later progresses to scarring. If there is uncertainty in the diagnosis, biopsy can be helpful.
The LPP subtype, FFA, usually occurs in postmenopausal women. It follows a distinctive pattern, as in this patient, where the hair is progressively lost along the frontoparietal hair line (and sometimes the eyebrows). A careful physical examination reveals smooth skin where follicles are lost and there is erythema around the base of the hairs due to active inflammation and keratotic plugging. The specific mechanism of FFA is poorly understood, and hormones may play a role, in addition to the inflammatory response.
The goal of treatment is to arrest the progression of additional hair loss (which usually is permanent). Intralesional steroid injections, which also are used for alopecia areata, are the most common therapy. Triamcinolone 2.5 to 5 mg/mL is injected in the affected dermal layer of the scalp. Oral finasteride (a 5-alpha-reductase inhibitor to decrease androgens) 1 mg/d can be helpful, as can oral hydroxychloroquine 200 mg bid.
Once the inflammation has subsided, treatment can be discontinued. Hair transplantation has been used, but often fails due to the inflammatory scarring process. Our patient noted that her disease process had been stable and declined treatment.
Photo courtesy of Daniel Stulberg, MD, FAAFP. Text courtesy of Rory Aufderheide, MD, and Daniel Stulberg, MD, FAAFP. Drs. Stulberg and Aufderheide are from the Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
To D, Beecker J. Frontal fibrosing alopecia: update and review of challenges and successes. J Cutan Med Surg. 2018;22:182-189.
To D, Beecker J. Frontal fibrosing alopecia: update and review of challenges and successes. J Cutan Med Surg. 2018;22:182-189.
Most e-consults not followed by specialist visit
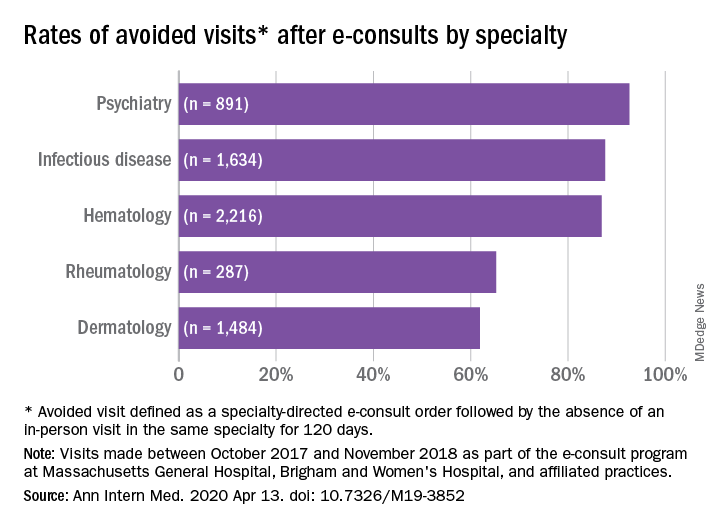
Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.

Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.

Studies have shown that e-consults increase access to specialist care and primary care physician (PCP) education, according to research published in the Annals of Internal Medicine (2020. Apr 14. doi: 10.7326/M19-3852) by Salman Ahmed, MD, and colleagues.
These resources are already being frequently used by physicians, but more often by general internists and hospitalists than by subspecialists, according to a recent survey by the American College of Physicians. That survey found that 42% of its respondents are using e-consults and that subspecialists’ use is less common primarily because of the lack of access to e-consult technology.
What hasn’t been widely researched are the effects of large-scale e-consult programs, said Dr. Ahmed, who is associate physician in the renal division at Brigham and Women’s Hospital, Boston, in an interview.
For frontline providers such as PCPs, e-consults are a way to quickly seek out answers to clinical questions from specialists. In turn, the specialist can help a wider pool of participants, he noted.
The findings of Dr. Ahmed’s study, which included several academic centers and hospitals affiliated with Partners HealthCare System, a nonprofit network in eastern Massachusetts that includes Brigham and Women’s Hospital, used several metrics to analyze the appropriateness and utility of e-consults across a range of specialties. An e-consult was considered useful if it resulted in the avoidance of a visit to a specialist, which was defined as the absence of an in-person visit to the type of specialist consulted electronically for 120 days. An e-consult was considered appropriate if it met the following four criteria.
- It could not be answered by referring to society guidelines or widely available, evidence-based summary sources.
- It did not seek logistic information, such as where to have a specific laboratory test done.
- It did not include a question of high urgency.
- The medical complexity of the clinical situation was not substantial enough to warrant an in-person consultation.
The investigators examined e-consult inquiries to mostly physician health care providers in five specialties – hematology, infectious disease, dermatology, rheumatology, and psychiatry – over a year.
High rates of appropriateness
The search spanned 6,512 eligible e-consults from 1,096 referring providers to 121 specialist consultants. Narrowing their search to 741 records with complete data, the investigators found that 70.2% of these consults met the criteria for appropriateness. In an analysis of four reviewers blinded to each other’s results, raters agreed on the appropriateness of 94% of e-consults.
Across specialties, more than 81% of e-consults were associated with avoided in-person visits.
The reasons for most e-consults were to seek answers to questions about diagnosis, therapeutics, or patient inquiries, or to request further education by PCPs.
“Across all specialties, the most common reasons an e-consult was not considered appropriate were failing the point-of-care resource test and asking a question of inappropriately high complexity,” the authors summarized.
Physicians and PCPs from tertiary care practices made up the majority of referring providers, with turnaround time for consults averaging 24 hours across specialties.
Rates of appropriateness, content, patient demographics, and timeliness of e-consult responses varied among the four specialties. Those with high avoidance of visits rates tended to have high appropriateness rates, indicating that some specialties may be more conducive to e-consults than others, the authors noted. Psychiatry and hematology had the highest proportion of appropriate e-consults (77.9% and 73.3% respectively). Rheumatology had the lowest proportion of appropriate e-consults and one of the lowest rates of avoided in-person visits, and dermatology had the lowest rate of avoided in-person visits, at 61.9%.
The majority (93%) of e-consults sought in psychiatry were therapy related, whereas 88.4% of the e-consult questions in rheumatology related to diagnosis.
“Questions about diagnosis were less likely to be answerable via e-consult, which suggests that to provide diagnoses, consultants may wish to engage with the patient directly,” Dr. Ahmed said in an interview.
Infectious disease specialists seemed to be the fastest responders, with nearly 90% of their consultations having been answered within a day. Dermatology specialists had the distinction of having the youngest e-consult patients (mean age, 38.6 years).
PCPs weigh in on results
Physicians said in interviews that the study data reflects their own positive experiences with e-consults.
“Although I don’t always think [an e-consult] is able to fully prevent the specialist visit, it does allow the specialist to provide recommendations for work-up that can be done prior to the specialist visit,” said Santina Wheat MD, a family physician at Erie Family Health Center in Chicago. This reduces the time in which the consult is placed to when effective treatment can take place.
Patients who may have to wait months or even years to see a specialty doctor, benefit from e-consults, said Dr. Wheat, who is also a member of the editorial advisory board of Family Practice News. “As part of an organization that does e-consults to another hospital with a different electronic medical record, the e-consult increases the likelihood that all of the clinical information reaches the specialists and prevents tests from being repeated.”
Starting an e-consult may also increase the likelihood that the patient quickly sees a specialist at the contracted hospital, she added.
Sarah G. Candler, MD, said in an interview that she also sees e-consults as an essential tool. “When patients present with rare, complex, or atypical pictures, I find it helpful to have specialists weigh in. The e-consult helps me ensure that I work to the top of my abilities as an internist,” said Dr. Candler, who is practice medical director and physician director of academic relations at Iora Primary Care, Northside Clinic, Houston. However, she did not agree with the study’s avoided in-person visits metric for assessing utility.
“In some cases, the end result of an e-consult is a referral for an in-person evaluation, and the role of the e-consult is to ensure that I have done my due diligence as a primary care doctor asking the correct questions, getting the appropriate work-up completed, and referring to the appropriate specialty for next steps, when necessary,” noted Dr. Candler, who also serves on the editorial advisory board of Internal Medicine News.
Financial considerations
The study’s authors suggested taking a closer look at standardizing payment for the use of e-consults and developing appropriateness criteria for them.
Health systems could use such criteria to study what makes an e-consult useful and how to best utilize this tool, Dr. Ahmed said in an interview.
“Compensation models that promote high-quality, effective, and efficient e-consults are needed to reinforce the ability of health systems to optimize the mix of e-consults and in-person visits,” Dr. Ahmed and colleagues suggested.
Because not all patient care requires e-consults, the model makes the most sense in practices that already participate in value-based payment programs. In these types of programs, the cost can be shared according to the variable risk and patient need for the service, Dr. Candler explained.
“I have been fortunate to work in two different systems that function in this way, which means that e-consults have been readily available and encouraged-both to improve patient care and decrease overall cost by decreasing unnecessary testing or specialist referral,” she said.
Dr. Wheat said that the managed care organization affiliated with her practice seems to be saving money with e-consults, as it decreases the need to pay for specialist visits in some instances and for repeated work-ups.
Future studies
The study’s cohort represented just one large health care system with a shared electronic health record. “Single-system descriptive studies, such as that of Ahmed and colleagues, are particularly useful for local evaluation and quality improvement efforts,” Varsha G. Vimalananda, MD, and B. Graeme Fincke, MD, both of the Center for Healthcare Organization and Implementation Research at Bedford (Mass.) Veterans Affairs Hospital, wrote in a related editorial.
“However, we need innovative approaches to evaluation that estimate the effect of e-consults on quality and cost of care across health care systems and over time. Implementation studies can help to identify key contributors to success,” the editorialists wrote.
One of the study authors, reported receiving personal fees from Bayer outside the submitted work. The other authors of the paper and the authors of the editorial reported no conflicts of interest. Dr. Candler said her employer contracts with an e-consult service, but that she is not compensated for use of the service. She is also a coeditor of Annals of Internal Medicine’s blog, “Fresh Look.”
SOURCE: Ahmed S et al. Ann Intern Med. 2020 Apr 14. doi: 10.7326/M19-3852.
FROM ANNALS OF INTERNAL MEDICINE
A case of neutrophilic eccrine hidradenitis attributed to HIV treatment
arising in an affected patient, Jessica Kalen, MD, advised during a virtual meeting held by the George Washington University department of dermatology.
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
In a presentation entitled, “When HAART [highly active antiretroviral therapy] Hurts,” Dr. Kalen, a dermatology resident at the university, presented a case report involving a 65-year-old man who presented with juicy red edematous papules and plaques on his scalp and ears. He was on the three-drug combination of rilpivirine (a non-nucleoside reverse transcriptase inhibitor), and the NRTIs tenofovir, and emtricitabine (Odefsey) for treatment of HIV infection, which was well controlled, with no detectable viral load.
The patient was also on insulin detemir for diabetes; pravastatin, amlodipine, and lisinopril for hypertension; and episodic acyclovir for recurrent herpes simplex outbreaks. However, none of those drugs has been associated with NEH. In contrast, Dr. Kalen found three published case reports describing a link between NRTIs and NEH.
Lesional biopsy of her patient showed the classic features of NEH: a dermal neutrophilic infiltrate surrounding the eccrine secretory coils and ducts, with vacuolar degeneration that spared the acrosyringium.
The most common causes of NEH, a rare dermatologic disorder first described in 1982, are hematologic malignancies and some of the chemotherapeutic agents used in treating them. Particularly prominent are acute myelogenous leukemia and cytarabine, which are often prescribed for that cancer. Carbamazepine, granulocyte-colony stimulating factor, and BRAF inhibitors have also been associated with NEH.
The pathogenesis of NEH is not fully worked out; however, NRTIs are secreted via eccrine structures, and that close contact could potentially promote an environment favoring inflammation and destruction of the eccrine coils. Also, NRTIs inhibit DNA polymerase, as does cytarabine, Dr. Kalen noted.
Her patient’s NEH was treated with triamcinolone. His skin condition resolved completely while he remained on NRTI therapy, with no relapses to date.
Dr. Kalen reported having no financial conflicts regarding her presentation.
arising in an affected patient, Jessica Kalen, MD, advised during a virtual meeting held by the George Washington University department of dermatology.
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
In a presentation entitled, “When HAART [highly active antiretroviral therapy] Hurts,” Dr. Kalen, a dermatology resident at the university, presented a case report involving a 65-year-old man who presented with juicy red edematous papules and plaques on his scalp and ears. He was on the three-drug combination of rilpivirine (a non-nucleoside reverse transcriptase inhibitor), and the NRTIs tenofovir, and emtricitabine (Odefsey) for treatment of HIV infection, which was well controlled, with no detectable viral load.
The patient was also on insulin detemir for diabetes; pravastatin, amlodipine, and lisinopril for hypertension; and episodic acyclovir for recurrent herpes simplex outbreaks. However, none of those drugs has been associated with NEH. In contrast, Dr. Kalen found three published case reports describing a link between NRTIs and NEH.
Lesional biopsy of her patient showed the classic features of NEH: a dermal neutrophilic infiltrate surrounding the eccrine secretory coils and ducts, with vacuolar degeneration that spared the acrosyringium.
The most common causes of NEH, a rare dermatologic disorder first described in 1982, are hematologic malignancies and some of the chemotherapeutic agents used in treating them. Particularly prominent are acute myelogenous leukemia and cytarabine, which are often prescribed for that cancer. Carbamazepine, granulocyte-colony stimulating factor, and BRAF inhibitors have also been associated with NEH.
The pathogenesis of NEH is not fully worked out; however, NRTIs are secreted via eccrine structures, and that close contact could potentially promote an environment favoring inflammation and destruction of the eccrine coils. Also, NRTIs inhibit DNA polymerase, as does cytarabine, Dr. Kalen noted.
Her patient’s NEH was treated with triamcinolone. His skin condition resolved completely while he remained on NRTI therapy, with no relapses to date.
Dr. Kalen reported having no financial conflicts regarding her presentation.
arising in an affected patient, Jessica Kalen, MD, advised during a virtual meeting held by the George Washington University department of dermatology.
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
In a presentation entitled, “When HAART [highly active antiretroviral therapy] Hurts,” Dr. Kalen, a dermatology resident at the university, presented a case report involving a 65-year-old man who presented with juicy red edematous papules and plaques on his scalp and ears. He was on the three-drug combination of rilpivirine (a non-nucleoside reverse transcriptase inhibitor), and the NRTIs tenofovir, and emtricitabine (Odefsey) for treatment of HIV infection, which was well controlled, with no detectable viral load.
The patient was also on insulin detemir for diabetes; pravastatin, amlodipine, and lisinopril for hypertension; and episodic acyclovir for recurrent herpes simplex outbreaks. However, none of those drugs has been associated with NEH. In contrast, Dr. Kalen found three published case reports describing a link between NRTIs and NEH.
Lesional biopsy of her patient showed the classic features of NEH: a dermal neutrophilic infiltrate surrounding the eccrine secretory coils and ducts, with vacuolar degeneration that spared the acrosyringium.
The most common causes of NEH, a rare dermatologic disorder first described in 1982, are hematologic malignancies and some of the chemotherapeutic agents used in treating them. Particularly prominent are acute myelogenous leukemia and cytarabine, which are often prescribed for that cancer. Carbamazepine, granulocyte-colony stimulating factor, and BRAF inhibitors have also been associated with NEH.
The pathogenesis of NEH is not fully worked out; however, NRTIs are secreted via eccrine structures, and that close contact could potentially promote an environment favoring inflammation and destruction of the eccrine coils. Also, NRTIs inhibit DNA polymerase, as does cytarabine, Dr. Kalen noted.
Her patient’s NEH was treated with triamcinolone. His skin condition resolved completely while he remained on NRTI therapy, with no relapses to date.
Dr. Kalen reported having no financial conflicts regarding her presentation.
Expert discusses her approach to using systemic agents in children and adolescents with severe skin disease
In the clinical opinion of Kaiane A. Habeshian, MD, dermatologists shouldn’t think twice about using systemic agents in pediatric patients with severe dermatologic diseases.
“By the time patients come to us pediatric dermatologists, they have been treated by multiple other doctors, and are frustrated,” Dr. Habeshian said during a virtual meeting held by the George Washington University department of dermatology. “Childhood eczema affects not only patients, but the whole family. For instance, if the child is not sleeping due to itch, their parents are probably not sleeping, either. Parental well-being and workplace productivity are affected, and finances are affected.”
Only a limited number of medications are Food and Drug Administration approved in pediatric patients for common dermatologic indications. These include dupilumab for atopic dermatitis (AD), etanercept and ustekinumab for psoriasis, adalimumab for hidradenitis suppurativa, and omalizumab for chronic idiopathic urticaria. “The approvals are mainly for the adolescent age group, except for etanercept, which is approved at the age of 4 years and above,” said Dr. Habeshian of the department of dermatology at Children’s National Hospital, Washington.
. “These agents are approved for other indications in infants and have many years of data to describe their use in these other conditions, although comprehensive randomized, controlled studies in pediatric patients for dermatologic conditions are lacking,” she said. “What’s in clinical trials for pediatric skin disease? There are multiple ongoing clinical studies of biologic agents in pediatric dermatology, mainly for psoriasis and also for dupilumab in younger patients, as well as a JAK [Janus kinase] inhibitor for alopecia areata.”
Dr. Habeshian noted that while some clinicians may have a knee-jerk reaction to go straight to dupilumab, which was approved in March of 2019 for adolescents with moderate to severe AD, that agent is not currently approved for the most sizable pediatric population with this condition – those under 12 years of age. “FDA approval is important in part because it helps establish safety and optimal dosing, which is often different and weight based in children,” she said. “In addition, FDA approval significantly impacts access to these newer, more expensive medications.”
Speaking from her experience treating patients in the DC/Maryland/Virginia area, Medicaid has consistently denied dupilumab coverage in children under age 12, “even in severe eczema that is suboptimally controlled with both methotrexate and cyclosporine, despite multiple levels of appeal, including letters of medical necessity and peer-to-peer evaluation,” she said. “This can vary across the country among states. However, dupilumab has been completely unattainable in those under 12 in our practice.”
When dupilumab is approved, most insurers first require step therapy with off-label agents for at least 3 months, as well as documented failure of topical corticosteroids, calcineurin inhibitors, crisaborole ointment, and phototherapy (if done). “It’s important to document an objective measure of severity at the very first visit with the SCORAD [scoring atopic dermatitis] or IGA [investigator global assessment],” she said. “Often, that is required if there is any hope for coverage. A familiarity with these requirements is often acquired through trial and error, and may change over time. This can lead to many delays in getting patients these treatments.” Additional information to consider documenting include the disease impact on quality of life, sleep, and school attendance, any hospitalizations for AD flares or secondary infections, and comorbid disease such as asthma.
Meanwhile, dupilumab is under priority review for children aged 6-11 years with moderate to severe AD, with a target action date of May 26, 2020. “It’s unclear how recent events [with the COVID-19 pandemic] will impact that, but there is something to look forward to, and give us hope for our patients,” she said.
Typically, Dr. Habeshian starts her pediatric patients with moderate to severe AD on methotrexate, which she characterized as “a time-tested, affordable, and very accessible option. It requires a little bit less monitoring upon initiation than cyclosporine, and it can be used for longer periods of time before weaning is required.”
In cases when disease is severe or intolerable, she often starts methotrexate and cyclosporine together. “I will usually start right at the 0.5 mg/kg per week rather than titrating up, because this maximizes the response and reduces the amount of blood work needed, unless they have an underlying risk factor for GI distress, or obese patients who are at increased risk for LFT [liver function test] elevation,” she noted. “Patients will note some improvement as early as 2 weeks on methotrexate, but I counsel them to expect 4-6 weeks for maximum improvement. We do not do a test dose of methotrexate at our institution. If there is a slight LFT elevation upon checking labs, ensure that the labs were done at least 4-6 days after the dose, because transient LFT dose elevations are common in 3-4 days.”
GI distress is by far the most common clinical side effect of methotrexate. “We do not do much intramuscular injection of methotrexate, so we rely a lot on folic acid, which reduces the risk of GI distress and elevated LFTs without reducing efficacy,” she said. “We recommend daily folic acid for simplicity, or folic acid 6 days per week.”
Dr. Habeshian said that many pediatric patients can swallow the 2.5 mg tablets of methotrexate “because they’re quite small, and most patients don’t have a problem taking the methotrexate when it’s crushed and mixed with food such as apple sauce or pudding. However, it is critical to discuss proper handling to avoid lung toxicity.” This includes placing the pills in a plastic bag prior to crushing, avoiding inhalation, and avoiding handling near pregnant women and pets, she noted. In addition, she said, “in adolescents, we need to consider the teratogenicity of methotrexate, as well as the possibility of alcohol consumption worsening liver complications. If I prescribe methotrexate in patients of childbearing age, I will counsel them extensively regarding the risk of fetal death and birth defects. If needed, I will start combined oral contraceptives. Ultimately, I’m willing to use these medicines safely, with significant counseling.”
When addressing the risk of methotrexate overdose, she reminds parents to store the medication in a safe place, out of the reach of children. “Patients are at the highest risk of overdose complications if they are given the medication multiple days in a row rather than a one-time, single high dose,” she said. “The literature suggests that one-time overdoses of methotrexate – deliberate or accidental – are unlikely to cause acute bone marrow suppression or hepatitis. This is probably because GI absorption of methotrexate reaches a saturation point, and the kidneys passively and actively excrete the medication at quite a rapid pace so that the methotrexate is often undetectable in the blood at 24 hours post ingestion. I do prescribe a limited supply to help prevent accidental overdoses. In part, this is because if the patient is receiving the medication daily, they’ll run out very quickly, and it will come the family’s attention and to your attention that it’s not being administered correctly.”
Another treatment option to consider for cases of moderate to severe AD is cyclosporine, “which works extremely quickly,” Dr. Habeshian said. “It is very good to rapidly control severe disease while methotrexate or other modes of treatment kick in. It’s best used as a bridge, given the risks of renal damage with long-term use. I like to limit its use to 6 months.”
Cyclosporine comes in two formulations: a modified oral formulation and a nonmodified oral formulation. The modified formulation is absorbed much better than the unmodified formulation. “We start at 5 mg/kg divided b.i.d., which is higher than the recommended dosing for dermatologic conditions in adults,” she said. “This is because children may not absorb the medication as well and may have improved renal clearance. Higher doses may be needed to achieve the desirable effect. In contrast to methotrexate, cyclosporine is available in a capsule, so it cannot be crushed.”
The choice of medication for psoriasis is generally guided by insurance step therapy requirements and is limited in the pediatric population (new guidelines on the care of pediatric psoriasis patients can be found at J Am Acad Dermatol 2020; 82[1]:161-201). In Dr. Habeshian’s experience, methotrexate is the go-to for most patients. “It treats concomitant psoriatic arthritis and can be used as monotherapy or combined with biologics,” she said. “Cyclosporine is useful for erythrodermic, pustular, and severe plaque psoriasis as a bridge. Other options include etanercept weekly in patients age 4-17 years and ustekinumab weekly dosing in patients age 12-17 years.”
Acitretin can be a useful adjunct for younger patients who are unable to obtain biologic agents. “It is most useful in widespread guttate and pustular psoriasis, but can be used be used in plaque psoriasis as well,” Dr. Habeshian said. “It is usually dosed as 0.1-1 mg/kg per day. Improvement in plaque disease is generally seen in 2-3 months of therapy, so it has a slow onset, whereas improvement in pustular psoriasis is seen within 3 weeks.” The most common side effects are dry skin and mucous membranes, while an important consideration is the potential for inducing premature bone toxicity. “It is thought that the risk is relatively low if the daily and total doses are kept low,” she said. “There is no consensus for monitoring bone health. Some clinicians will consider radiography periodically.”
Dr. Habeshian concluded her talk by noting that clinicians should give vaccinations/boosters before starting systemic therapy in young children. “The safety and efficacy of live immunization administered to children on biologics is not known,” she said. “Therefore, if live vaccination is needed, it’s generally recommended to postpone initiating biologic treatment.” The MMR and varicella vaccines are given at 12-15 months of life, with a booster at 4-6 years. The varicella vaccine should be given at least 6 weeks before starting immunosuppressive therapy, and the MMR vaccine at least 4 weeks before starting therapy.
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic. Dr. Habeshian reported having no disclosures.
In the clinical opinion of Kaiane A. Habeshian, MD, dermatologists shouldn’t think twice about using systemic agents in pediatric patients with severe dermatologic diseases.
“By the time patients come to us pediatric dermatologists, they have been treated by multiple other doctors, and are frustrated,” Dr. Habeshian said during a virtual meeting held by the George Washington University department of dermatology. “Childhood eczema affects not only patients, but the whole family. For instance, if the child is not sleeping due to itch, their parents are probably not sleeping, either. Parental well-being and workplace productivity are affected, and finances are affected.”
Only a limited number of medications are Food and Drug Administration approved in pediatric patients for common dermatologic indications. These include dupilumab for atopic dermatitis (AD), etanercept and ustekinumab for psoriasis, adalimumab for hidradenitis suppurativa, and omalizumab for chronic idiopathic urticaria. “The approvals are mainly for the adolescent age group, except for etanercept, which is approved at the age of 4 years and above,” said Dr. Habeshian of the department of dermatology at Children’s National Hospital, Washington.
. “These agents are approved for other indications in infants and have many years of data to describe their use in these other conditions, although comprehensive randomized, controlled studies in pediatric patients for dermatologic conditions are lacking,” she said. “What’s in clinical trials for pediatric skin disease? There are multiple ongoing clinical studies of biologic agents in pediatric dermatology, mainly for psoriasis and also for dupilumab in younger patients, as well as a JAK [Janus kinase] inhibitor for alopecia areata.”
Dr. Habeshian noted that while some clinicians may have a knee-jerk reaction to go straight to dupilumab, which was approved in March of 2019 for adolescents with moderate to severe AD, that agent is not currently approved for the most sizable pediatric population with this condition – those under 12 years of age. “FDA approval is important in part because it helps establish safety and optimal dosing, which is often different and weight based in children,” she said. “In addition, FDA approval significantly impacts access to these newer, more expensive medications.”
Speaking from her experience treating patients in the DC/Maryland/Virginia area, Medicaid has consistently denied dupilumab coverage in children under age 12, “even in severe eczema that is suboptimally controlled with both methotrexate and cyclosporine, despite multiple levels of appeal, including letters of medical necessity and peer-to-peer evaluation,” she said. “This can vary across the country among states. However, dupilumab has been completely unattainable in those under 12 in our practice.”
When dupilumab is approved, most insurers first require step therapy with off-label agents for at least 3 months, as well as documented failure of topical corticosteroids, calcineurin inhibitors, crisaborole ointment, and phototherapy (if done). “It’s important to document an objective measure of severity at the very first visit with the SCORAD [scoring atopic dermatitis] or IGA [investigator global assessment],” she said. “Often, that is required if there is any hope for coverage. A familiarity with these requirements is often acquired through trial and error, and may change over time. This can lead to many delays in getting patients these treatments.” Additional information to consider documenting include the disease impact on quality of life, sleep, and school attendance, any hospitalizations for AD flares or secondary infections, and comorbid disease such as asthma.
Meanwhile, dupilumab is under priority review for children aged 6-11 years with moderate to severe AD, with a target action date of May 26, 2020. “It’s unclear how recent events [with the COVID-19 pandemic] will impact that, but there is something to look forward to, and give us hope for our patients,” she said.
Typically, Dr. Habeshian starts her pediatric patients with moderate to severe AD on methotrexate, which she characterized as “a time-tested, affordable, and very accessible option. It requires a little bit less monitoring upon initiation than cyclosporine, and it can be used for longer periods of time before weaning is required.”
In cases when disease is severe or intolerable, she often starts methotrexate and cyclosporine together. “I will usually start right at the 0.5 mg/kg per week rather than titrating up, because this maximizes the response and reduces the amount of blood work needed, unless they have an underlying risk factor for GI distress, or obese patients who are at increased risk for LFT [liver function test] elevation,” she noted. “Patients will note some improvement as early as 2 weeks on methotrexate, but I counsel them to expect 4-6 weeks for maximum improvement. We do not do a test dose of methotrexate at our institution. If there is a slight LFT elevation upon checking labs, ensure that the labs were done at least 4-6 days after the dose, because transient LFT dose elevations are common in 3-4 days.”
GI distress is by far the most common clinical side effect of methotrexate. “We do not do much intramuscular injection of methotrexate, so we rely a lot on folic acid, which reduces the risk of GI distress and elevated LFTs without reducing efficacy,” she said. “We recommend daily folic acid for simplicity, or folic acid 6 days per week.”
Dr. Habeshian said that many pediatric patients can swallow the 2.5 mg tablets of methotrexate “because they’re quite small, and most patients don’t have a problem taking the methotrexate when it’s crushed and mixed with food such as apple sauce or pudding. However, it is critical to discuss proper handling to avoid lung toxicity.” This includes placing the pills in a plastic bag prior to crushing, avoiding inhalation, and avoiding handling near pregnant women and pets, she noted. In addition, she said, “in adolescents, we need to consider the teratogenicity of methotrexate, as well as the possibility of alcohol consumption worsening liver complications. If I prescribe methotrexate in patients of childbearing age, I will counsel them extensively regarding the risk of fetal death and birth defects. If needed, I will start combined oral contraceptives. Ultimately, I’m willing to use these medicines safely, with significant counseling.”
When addressing the risk of methotrexate overdose, she reminds parents to store the medication in a safe place, out of the reach of children. “Patients are at the highest risk of overdose complications if they are given the medication multiple days in a row rather than a one-time, single high dose,” she said. “The literature suggests that one-time overdoses of methotrexate – deliberate or accidental – are unlikely to cause acute bone marrow suppression or hepatitis. This is probably because GI absorption of methotrexate reaches a saturation point, and the kidneys passively and actively excrete the medication at quite a rapid pace so that the methotrexate is often undetectable in the blood at 24 hours post ingestion. I do prescribe a limited supply to help prevent accidental overdoses. In part, this is because if the patient is receiving the medication daily, they’ll run out very quickly, and it will come the family’s attention and to your attention that it’s not being administered correctly.”
Another treatment option to consider for cases of moderate to severe AD is cyclosporine, “which works extremely quickly,” Dr. Habeshian said. “It is very good to rapidly control severe disease while methotrexate or other modes of treatment kick in. It’s best used as a bridge, given the risks of renal damage with long-term use. I like to limit its use to 6 months.”
Cyclosporine comes in two formulations: a modified oral formulation and a nonmodified oral formulation. The modified formulation is absorbed much better than the unmodified formulation. “We start at 5 mg/kg divided b.i.d., which is higher than the recommended dosing for dermatologic conditions in adults,” she said. “This is because children may not absorb the medication as well and may have improved renal clearance. Higher doses may be needed to achieve the desirable effect. In contrast to methotrexate, cyclosporine is available in a capsule, so it cannot be crushed.”
The choice of medication for psoriasis is generally guided by insurance step therapy requirements and is limited in the pediatric population (new guidelines on the care of pediatric psoriasis patients can be found at J Am Acad Dermatol 2020; 82[1]:161-201). In Dr. Habeshian’s experience, methotrexate is the go-to for most patients. “It treats concomitant psoriatic arthritis and can be used as monotherapy or combined with biologics,” she said. “Cyclosporine is useful for erythrodermic, pustular, and severe plaque psoriasis as a bridge. Other options include etanercept weekly in patients age 4-17 years and ustekinumab weekly dosing in patients age 12-17 years.”
Acitretin can be a useful adjunct for younger patients who are unable to obtain biologic agents. “It is most useful in widespread guttate and pustular psoriasis, but can be used be used in plaque psoriasis as well,” Dr. Habeshian said. “It is usually dosed as 0.1-1 mg/kg per day. Improvement in plaque disease is generally seen in 2-3 months of therapy, so it has a slow onset, whereas improvement in pustular psoriasis is seen within 3 weeks.” The most common side effects are dry skin and mucous membranes, while an important consideration is the potential for inducing premature bone toxicity. “It is thought that the risk is relatively low if the daily and total doses are kept low,” she said. “There is no consensus for monitoring bone health. Some clinicians will consider radiography periodically.”
Dr. Habeshian concluded her talk by noting that clinicians should give vaccinations/boosters before starting systemic therapy in young children. “The safety and efficacy of live immunization administered to children on biologics is not known,” she said. “Therefore, if live vaccination is needed, it’s generally recommended to postpone initiating biologic treatment.” The MMR and varicella vaccines are given at 12-15 months of life, with a booster at 4-6 years. The varicella vaccine should be given at least 6 weeks before starting immunosuppressive therapy, and the MMR vaccine at least 4 weeks before starting therapy.
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic. Dr. Habeshian reported having no disclosures.
In the clinical opinion of Kaiane A. Habeshian, MD, dermatologists shouldn’t think twice about using systemic agents in pediatric patients with severe dermatologic diseases.
“By the time patients come to us pediatric dermatologists, they have been treated by multiple other doctors, and are frustrated,” Dr. Habeshian said during a virtual meeting held by the George Washington University department of dermatology. “Childhood eczema affects not only patients, but the whole family. For instance, if the child is not sleeping due to itch, their parents are probably not sleeping, either. Parental well-being and workplace productivity are affected, and finances are affected.”
Only a limited number of medications are Food and Drug Administration approved in pediatric patients for common dermatologic indications. These include dupilumab for atopic dermatitis (AD), etanercept and ustekinumab for psoriasis, adalimumab for hidradenitis suppurativa, and omalizumab for chronic idiopathic urticaria. “The approvals are mainly for the adolescent age group, except for etanercept, which is approved at the age of 4 years and above,” said Dr. Habeshian of the department of dermatology at Children’s National Hospital, Washington.
. “These agents are approved for other indications in infants and have many years of data to describe their use in these other conditions, although comprehensive randomized, controlled studies in pediatric patients for dermatologic conditions are lacking,” she said. “What’s in clinical trials for pediatric skin disease? There are multiple ongoing clinical studies of biologic agents in pediatric dermatology, mainly for psoriasis and also for dupilumab in younger patients, as well as a JAK [Janus kinase] inhibitor for alopecia areata.”
Dr. Habeshian noted that while some clinicians may have a knee-jerk reaction to go straight to dupilumab, which was approved in March of 2019 for adolescents with moderate to severe AD, that agent is not currently approved for the most sizable pediatric population with this condition – those under 12 years of age. “FDA approval is important in part because it helps establish safety and optimal dosing, which is often different and weight based in children,” she said. “In addition, FDA approval significantly impacts access to these newer, more expensive medications.”
Speaking from her experience treating patients in the DC/Maryland/Virginia area, Medicaid has consistently denied dupilumab coverage in children under age 12, “even in severe eczema that is suboptimally controlled with both methotrexate and cyclosporine, despite multiple levels of appeal, including letters of medical necessity and peer-to-peer evaluation,” she said. “This can vary across the country among states. However, dupilumab has been completely unattainable in those under 12 in our practice.”
When dupilumab is approved, most insurers first require step therapy with off-label agents for at least 3 months, as well as documented failure of topical corticosteroids, calcineurin inhibitors, crisaborole ointment, and phototherapy (if done). “It’s important to document an objective measure of severity at the very first visit with the SCORAD [scoring atopic dermatitis] or IGA [investigator global assessment],” she said. “Often, that is required if there is any hope for coverage. A familiarity with these requirements is often acquired through trial and error, and may change over time. This can lead to many delays in getting patients these treatments.” Additional information to consider documenting include the disease impact on quality of life, sleep, and school attendance, any hospitalizations for AD flares or secondary infections, and comorbid disease such as asthma.
Meanwhile, dupilumab is under priority review for children aged 6-11 years with moderate to severe AD, with a target action date of May 26, 2020. “It’s unclear how recent events [with the COVID-19 pandemic] will impact that, but there is something to look forward to, and give us hope for our patients,” she said.
Typically, Dr. Habeshian starts her pediatric patients with moderate to severe AD on methotrexate, which she characterized as “a time-tested, affordable, and very accessible option. It requires a little bit less monitoring upon initiation than cyclosporine, and it can be used for longer periods of time before weaning is required.”
In cases when disease is severe or intolerable, she often starts methotrexate and cyclosporine together. “I will usually start right at the 0.5 mg/kg per week rather than titrating up, because this maximizes the response and reduces the amount of blood work needed, unless they have an underlying risk factor for GI distress, or obese patients who are at increased risk for LFT [liver function test] elevation,” she noted. “Patients will note some improvement as early as 2 weeks on methotrexate, but I counsel them to expect 4-6 weeks for maximum improvement. We do not do a test dose of methotrexate at our institution. If there is a slight LFT elevation upon checking labs, ensure that the labs were done at least 4-6 days after the dose, because transient LFT dose elevations are common in 3-4 days.”
GI distress is by far the most common clinical side effect of methotrexate. “We do not do much intramuscular injection of methotrexate, so we rely a lot on folic acid, which reduces the risk of GI distress and elevated LFTs without reducing efficacy,” she said. “We recommend daily folic acid for simplicity, or folic acid 6 days per week.”
Dr. Habeshian said that many pediatric patients can swallow the 2.5 mg tablets of methotrexate “because they’re quite small, and most patients don’t have a problem taking the methotrexate when it’s crushed and mixed with food such as apple sauce or pudding. However, it is critical to discuss proper handling to avoid lung toxicity.” This includes placing the pills in a plastic bag prior to crushing, avoiding inhalation, and avoiding handling near pregnant women and pets, she noted. In addition, she said, “in adolescents, we need to consider the teratogenicity of methotrexate, as well as the possibility of alcohol consumption worsening liver complications. If I prescribe methotrexate in patients of childbearing age, I will counsel them extensively regarding the risk of fetal death and birth defects. If needed, I will start combined oral contraceptives. Ultimately, I’m willing to use these medicines safely, with significant counseling.”
When addressing the risk of methotrexate overdose, she reminds parents to store the medication in a safe place, out of the reach of children. “Patients are at the highest risk of overdose complications if they are given the medication multiple days in a row rather than a one-time, single high dose,” she said. “The literature suggests that one-time overdoses of methotrexate – deliberate or accidental – are unlikely to cause acute bone marrow suppression or hepatitis. This is probably because GI absorption of methotrexate reaches a saturation point, and the kidneys passively and actively excrete the medication at quite a rapid pace so that the methotrexate is often undetectable in the blood at 24 hours post ingestion. I do prescribe a limited supply to help prevent accidental overdoses. In part, this is because if the patient is receiving the medication daily, they’ll run out very quickly, and it will come the family’s attention and to your attention that it’s not being administered correctly.”
Another treatment option to consider for cases of moderate to severe AD is cyclosporine, “which works extremely quickly,” Dr. Habeshian said. “It is very good to rapidly control severe disease while methotrexate or other modes of treatment kick in. It’s best used as a bridge, given the risks of renal damage with long-term use. I like to limit its use to 6 months.”
Cyclosporine comes in two formulations: a modified oral formulation and a nonmodified oral formulation. The modified formulation is absorbed much better than the unmodified formulation. “We start at 5 mg/kg divided b.i.d., which is higher than the recommended dosing for dermatologic conditions in adults,” she said. “This is because children may not absorb the medication as well and may have improved renal clearance. Higher doses may be needed to achieve the desirable effect. In contrast to methotrexate, cyclosporine is available in a capsule, so it cannot be crushed.”
The choice of medication for psoriasis is generally guided by insurance step therapy requirements and is limited in the pediatric population (new guidelines on the care of pediatric psoriasis patients can be found at J Am Acad Dermatol 2020; 82[1]:161-201). In Dr. Habeshian’s experience, methotrexate is the go-to for most patients. “It treats concomitant psoriatic arthritis and can be used as monotherapy or combined with biologics,” she said. “Cyclosporine is useful for erythrodermic, pustular, and severe plaque psoriasis as a bridge. Other options include etanercept weekly in patients age 4-17 years and ustekinumab weekly dosing in patients age 12-17 years.”
Acitretin can be a useful adjunct for younger patients who are unable to obtain biologic agents. “It is most useful in widespread guttate and pustular psoriasis, but can be used be used in plaque psoriasis as well,” Dr. Habeshian said. “It is usually dosed as 0.1-1 mg/kg per day. Improvement in plaque disease is generally seen in 2-3 months of therapy, so it has a slow onset, whereas improvement in pustular psoriasis is seen within 3 weeks.” The most common side effects are dry skin and mucous membranes, while an important consideration is the potential for inducing premature bone toxicity. “It is thought that the risk is relatively low if the daily and total doses are kept low,” she said. “There is no consensus for monitoring bone health. Some clinicians will consider radiography periodically.”
Dr. Habeshian concluded her talk by noting that clinicians should give vaccinations/boosters before starting systemic therapy in young children. “The safety and efficacy of live immunization administered to children on biologics is not known,” she said. “Therefore, if live vaccination is needed, it’s generally recommended to postpone initiating biologic treatment.” The MMR and varicella vaccines are given at 12-15 months of life, with a booster at 4-6 years. The varicella vaccine should be given at least 6 weeks before starting immunosuppressive therapy, and the MMR vaccine at least 4 weeks before starting therapy.
The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic. Dr. Habeshian reported having no disclosures.
When to suspect calciphylaxis and what to do about it
If the shoe fits a presumptive clinical diagnosis of calciphylaxis, wear it – and don’t assume that ordering imaging studies or histology will make for a better fit or is even necessary.
That was the key message of Karl M. Saardi, MD, during his video presentation at a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
“ said Dr. Saardi, a dermatology resident at Georgetown University in Washington, D.C.
He presented a single-center, retrospective study that underscored the diagnostic challenges posed by calciphylaxis, a condition for which there are no generally accepted clinical, radiographic, or histologic diagnostic criteria.
The rare skin condition is characterized by calcium deposition in small arterioles and capillaries in the skin and subcutaneous tissue. It’s most common in patients with end-stage renal disease who are on dialysis; however, there is also an increasingly recognized nonuremic variant that’s associated with the use of warfarin, chronic steroids, obesity, and possibly with being antiphospholipid antibody positive.
Calciphylaxis is an extremely painful condition – the pain is ischemic in nature – and it’s associated with substantial morbidity as well as a mortality rate that in many series exceeds 50%. Affected individuals typically present with progressive, painful retiform purpura on the legs, belly, buttocks, and other fatty body sites.
Dr. Saardi’s study entailed a retrospective look at the medical records and pathologic reports of 57 patients who underwent skin biopsy for suspected calciphylaxis. Of the 57, 18 had no antecedent imaging studies done during the preceding 3 months; 8 of those 18 (40%), had a confirmatory positive biopsy. A total of 39 patients did have imaging studies, deemed positive for calciphylaxis in 11 cases, which in only 5 of the 11 imaging-positive cases (45%) were subsequently confirmed by positive biopsy.
And finally, of the 28 patients with negative imaging studies, 10 (36%), had a positive biopsy. Those positive biopsy rates, ranging from 36% to 45%, did not differ statistically. Thus, whether an imaging study was positive or negative, or wasn’t even done, made no difference in terms of the ultimate diagnosis.
“You may not need imaging studies, because imaging has often been done before the consultation is requested because people are looking for things like arterial thrombus, cellulitis, [deep vein thrombosis] or something like that,” Dr. Saardi noted. “In our series, the indication was never calciphylaxis, it was always something like pain, infection, swelling, suspected [deep vein thrombosis], things like that.”
The classic signature of calciphylaxis on plain x-ray is net-like calcifications in skin and subcutaneous tissue. In one study, this often-subtle finding was associated with a 830% increased likelihood of a positive biopsy, with a specificity of 90%; however, these x-ray changes were only found in 13 of 29 patients with biopsy-confirmed calciphylaxis.
“It’s really important when you request plain films in these patients to review the images yourself or together with the radiologist because oftentimes the indication for imaging will be very different from what we’re looking for. Radiologists often won’t know to look for this specifically,” Dr. Saardi said.
The classic histopathologic finding is calcification of the small- and medium-sized vessels in the dermis and subcutaneous soft tissue. However, sometimes all that’s present are small intravascular inflammatory thrombi with intimal hyperplasia.
Skin biopsies are not infrequently falsely negative or nondiagnostic. To maximize the utility of the procedure, it’s important to go deep and gather a tissue sample that extends into subcutaneous tissue.
“You need to do a very deep punch or double-punch biopsy,” he said. “Another key is to avoid biopsy if the pretest probability of calciphylaxis is high because a negative biopsy shouldn’t necessarily reassure you or cause you to withhold treatment. And with the concern about pathergy or Koebnerization of the area causing a wound that’s never going to heal, sometimes a biopsy is not needed if the pretest suspicion is high enough.”
Other investigators have shown that the likelihood of an informative biopsy is enhanced by using a calcium stain on the specimen and having an experienced dermatopathologist do the evaluation. Also, the use of a radiographically guided core needle biopsy to ensure that the physician is getting sufficiently deep into subcutaneous fat is now under evaluation.
In addition to plain radiographs, other imaging methods that are sometimes used to evaluate soft-tissue sites for suspected calciphylaxis included CT and ultrasound. Dr. Saardi is particularly intrigued by reports from investigators at Harvard University regarding the utility of nuclear bone scintigraphy; in one study, this form of imaging was positive in 16 of 18 patients with clinically diagnosed calciphylaxis, versus just 1 of 31 controls with end-stage renal disease.
“We’ve started doing this in situations where biopsy is not desirable or feasible at that moment,” he said.
Dr. Saardi reported having no financial conflicts regarding his presentation.
If the shoe fits a presumptive clinical diagnosis of calciphylaxis, wear it – and don’t assume that ordering imaging studies or histology will make for a better fit or is even necessary.
That was the key message of Karl M. Saardi, MD, during his video presentation at a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
“ said Dr. Saardi, a dermatology resident at Georgetown University in Washington, D.C.
He presented a single-center, retrospective study that underscored the diagnostic challenges posed by calciphylaxis, a condition for which there are no generally accepted clinical, radiographic, or histologic diagnostic criteria.
The rare skin condition is characterized by calcium deposition in small arterioles and capillaries in the skin and subcutaneous tissue. It’s most common in patients with end-stage renal disease who are on dialysis; however, there is also an increasingly recognized nonuremic variant that’s associated with the use of warfarin, chronic steroids, obesity, and possibly with being antiphospholipid antibody positive.
Calciphylaxis is an extremely painful condition – the pain is ischemic in nature – and it’s associated with substantial morbidity as well as a mortality rate that in many series exceeds 50%. Affected individuals typically present with progressive, painful retiform purpura on the legs, belly, buttocks, and other fatty body sites.
Dr. Saardi’s study entailed a retrospective look at the medical records and pathologic reports of 57 patients who underwent skin biopsy for suspected calciphylaxis. Of the 57, 18 had no antecedent imaging studies done during the preceding 3 months; 8 of those 18 (40%), had a confirmatory positive biopsy. A total of 39 patients did have imaging studies, deemed positive for calciphylaxis in 11 cases, which in only 5 of the 11 imaging-positive cases (45%) were subsequently confirmed by positive biopsy.
And finally, of the 28 patients with negative imaging studies, 10 (36%), had a positive biopsy. Those positive biopsy rates, ranging from 36% to 45%, did not differ statistically. Thus, whether an imaging study was positive or negative, or wasn’t even done, made no difference in terms of the ultimate diagnosis.
“You may not need imaging studies, because imaging has often been done before the consultation is requested because people are looking for things like arterial thrombus, cellulitis, [deep vein thrombosis] or something like that,” Dr. Saardi noted. “In our series, the indication was never calciphylaxis, it was always something like pain, infection, swelling, suspected [deep vein thrombosis], things like that.”
The classic signature of calciphylaxis on plain x-ray is net-like calcifications in skin and subcutaneous tissue. In one study, this often-subtle finding was associated with a 830% increased likelihood of a positive biopsy, with a specificity of 90%; however, these x-ray changes were only found in 13 of 29 patients with biopsy-confirmed calciphylaxis.
“It’s really important when you request plain films in these patients to review the images yourself or together with the radiologist because oftentimes the indication for imaging will be very different from what we’re looking for. Radiologists often won’t know to look for this specifically,” Dr. Saardi said.
The classic histopathologic finding is calcification of the small- and medium-sized vessels in the dermis and subcutaneous soft tissue. However, sometimes all that’s present are small intravascular inflammatory thrombi with intimal hyperplasia.
Skin biopsies are not infrequently falsely negative or nondiagnostic. To maximize the utility of the procedure, it’s important to go deep and gather a tissue sample that extends into subcutaneous tissue.
“You need to do a very deep punch or double-punch biopsy,” he said. “Another key is to avoid biopsy if the pretest probability of calciphylaxis is high because a negative biopsy shouldn’t necessarily reassure you or cause you to withhold treatment. And with the concern about pathergy or Koebnerization of the area causing a wound that’s never going to heal, sometimes a biopsy is not needed if the pretest suspicion is high enough.”
Other investigators have shown that the likelihood of an informative biopsy is enhanced by using a calcium stain on the specimen and having an experienced dermatopathologist do the evaluation. Also, the use of a radiographically guided core needle biopsy to ensure that the physician is getting sufficiently deep into subcutaneous fat is now under evaluation.
In addition to plain radiographs, other imaging methods that are sometimes used to evaluate soft-tissue sites for suspected calciphylaxis included CT and ultrasound. Dr. Saardi is particularly intrigued by reports from investigators at Harvard University regarding the utility of nuclear bone scintigraphy; in one study, this form of imaging was positive in 16 of 18 patients with clinically diagnosed calciphylaxis, versus just 1 of 31 controls with end-stage renal disease.
“We’ve started doing this in situations where biopsy is not desirable or feasible at that moment,” he said.
Dr. Saardi reported having no financial conflicts regarding his presentation.
If the shoe fits a presumptive clinical diagnosis of calciphylaxis, wear it – and don’t assume that ordering imaging studies or histology will make for a better fit or is even necessary.
That was the key message of Karl M. Saardi, MD, during his video presentation at a virtual meeting held by the George Washington University department of dermatology. The virtual meeting included presentations that had been slated for the annual meeting of the American Academy of Dermatology, which was canceled because of the COVID-19 pandemic.
“ said Dr. Saardi, a dermatology resident at Georgetown University in Washington, D.C.
He presented a single-center, retrospective study that underscored the diagnostic challenges posed by calciphylaxis, a condition for which there are no generally accepted clinical, radiographic, or histologic diagnostic criteria.
The rare skin condition is characterized by calcium deposition in small arterioles and capillaries in the skin and subcutaneous tissue. It’s most common in patients with end-stage renal disease who are on dialysis; however, there is also an increasingly recognized nonuremic variant that’s associated with the use of warfarin, chronic steroids, obesity, and possibly with being antiphospholipid antibody positive.
Calciphylaxis is an extremely painful condition – the pain is ischemic in nature – and it’s associated with substantial morbidity as well as a mortality rate that in many series exceeds 50%. Affected individuals typically present with progressive, painful retiform purpura on the legs, belly, buttocks, and other fatty body sites.
Dr. Saardi’s study entailed a retrospective look at the medical records and pathologic reports of 57 patients who underwent skin biopsy for suspected calciphylaxis. Of the 57, 18 had no antecedent imaging studies done during the preceding 3 months; 8 of those 18 (40%), had a confirmatory positive biopsy. A total of 39 patients did have imaging studies, deemed positive for calciphylaxis in 11 cases, which in only 5 of the 11 imaging-positive cases (45%) were subsequently confirmed by positive biopsy.
And finally, of the 28 patients with negative imaging studies, 10 (36%), had a positive biopsy. Those positive biopsy rates, ranging from 36% to 45%, did not differ statistically. Thus, whether an imaging study was positive or negative, or wasn’t even done, made no difference in terms of the ultimate diagnosis.
“You may not need imaging studies, because imaging has often been done before the consultation is requested because people are looking for things like arterial thrombus, cellulitis, [deep vein thrombosis] or something like that,” Dr. Saardi noted. “In our series, the indication was never calciphylaxis, it was always something like pain, infection, swelling, suspected [deep vein thrombosis], things like that.”
The classic signature of calciphylaxis on plain x-ray is net-like calcifications in skin and subcutaneous tissue. In one study, this often-subtle finding was associated with a 830% increased likelihood of a positive biopsy, with a specificity of 90%; however, these x-ray changes were only found in 13 of 29 patients with biopsy-confirmed calciphylaxis.
“It’s really important when you request plain films in these patients to review the images yourself or together with the radiologist because oftentimes the indication for imaging will be very different from what we’re looking for. Radiologists often won’t know to look for this specifically,” Dr. Saardi said.
The classic histopathologic finding is calcification of the small- and medium-sized vessels in the dermis and subcutaneous soft tissue. However, sometimes all that’s present are small intravascular inflammatory thrombi with intimal hyperplasia.
Skin biopsies are not infrequently falsely negative or nondiagnostic. To maximize the utility of the procedure, it’s important to go deep and gather a tissue sample that extends into subcutaneous tissue.
“You need to do a very deep punch or double-punch biopsy,” he said. “Another key is to avoid biopsy if the pretest probability of calciphylaxis is high because a negative biopsy shouldn’t necessarily reassure you or cause you to withhold treatment. And with the concern about pathergy or Koebnerization of the area causing a wound that’s never going to heal, sometimes a biopsy is not needed if the pretest suspicion is high enough.”
Other investigators have shown that the likelihood of an informative biopsy is enhanced by using a calcium stain on the specimen and having an experienced dermatopathologist do the evaluation. Also, the use of a radiographically guided core needle biopsy to ensure that the physician is getting sufficiently deep into subcutaneous fat is now under evaluation.
In addition to plain radiographs, other imaging methods that are sometimes used to evaluate soft-tissue sites for suspected calciphylaxis included CT and ultrasound. Dr. Saardi is particularly intrigued by reports from investigators at Harvard University regarding the utility of nuclear bone scintigraphy; in one study, this form of imaging was positive in 16 of 18 patients with clinically diagnosed calciphylaxis, versus just 1 of 31 controls with end-stage renal disease.
“We’ve started doing this in situations where biopsy is not desirable or feasible at that moment,” he said.
Dr. Saardi reported having no financial conflicts regarding his presentation.