User login
Painful ear lesion
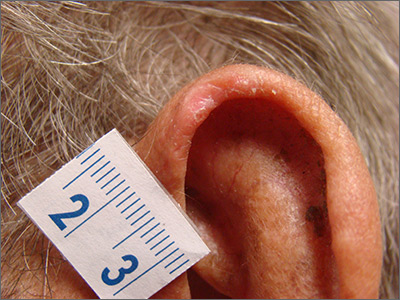
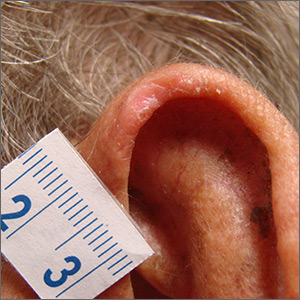
The patient was given a diagnosis of chondrodermatitis nodularis helicis (CNH), an inflammation of the cartilage and overlying skin causing a painful nodule of the helix. These lesions typically have a more prominent nodular component and a central ulceration or firm scale. They are thought to be due to chronic pressure on the ear.
There is a slight male predominance of CNH and onset is usually gradual. Patients often experience pain when sleeping on the affected side. The tenderness usually can be reproduced clinically by pressing on the lesion. The ears also are a high-risk area for actinic keratoses (AK) and nonmelanoma skin cancer (NMSC); if there is doubt about the diagnosis, a biopsy may be warranted to rule out AK or NMSC. In this patient, a shave biopsy was performed.
Various treatment regimens are available for CNH. The least invasive treatment approach is to use a “cut out” foam or a special donut-shaped pillow to protect the area from further pressure. By protecting from pressure and irritation, the lesion resolves in 57% to 92% of cases in clinical studies. Intralesional injection with 0.2 mL of 10 mg/mL triamcinolone acetonide is a simple in-office procedure that frequently helps the pain and may be curative; although, repeat injections may be necessary. Excision of the overlying skin and the affected cartilage is a more aggressive treatment with high success rates. More recently, treatment with topical nitroglycerin patches or photodynamic therapy have been described in small trials.
After confirming by a shave biopsy that the lesion was not cancerous, the patient returned for an elliptical excision of the lesion. A dermal curette was used to remove the rough abnormal inflammation of the underlying cartilage, and the elliptical wound was sutured with a linear closure.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Juul Nielsen L, Holkmann Olsen C, Lock-Andersen J. Therapeutic options of chondrodermatitis nodularis helicis [published online January 27, 2016]. Plast Surg Int. 2016;2016:4340168.

The patient was given a diagnosis of chondrodermatitis nodularis helicis (CNH), an inflammation of the cartilage and overlying skin causing a painful nodule of the helix. These lesions typically have a more prominent nodular component and a central ulceration or firm scale. They are thought to be due to chronic pressure on the ear.
There is a slight male predominance of CNH and onset is usually gradual. Patients often experience pain when sleeping on the affected side. The tenderness usually can be reproduced clinically by pressing on the lesion. The ears also are a high-risk area for actinic keratoses (AK) and nonmelanoma skin cancer (NMSC); if there is doubt about the diagnosis, a biopsy may be warranted to rule out AK or NMSC. In this patient, a shave biopsy was performed.
Various treatment regimens are available for CNH. The least invasive treatment approach is to use a “cut out” foam or a special donut-shaped pillow to protect the area from further pressure. By protecting from pressure and irritation, the lesion resolves in 57% to 92% of cases in clinical studies. Intralesional injection with 0.2 mL of 10 mg/mL triamcinolone acetonide is a simple in-office procedure that frequently helps the pain and may be curative; although, repeat injections may be necessary. Excision of the overlying skin and the affected cartilage is a more aggressive treatment with high success rates. More recently, treatment with topical nitroglycerin patches or photodynamic therapy have been described in small trials.
After confirming by a shave biopsy that the lesion was not cancerous, the patient returned for an elliptical excision of the lesion. A dermal curette was used to remove the rough abnormal inflammation of the underlying cartilage, and the elliptical wound was sutured with a linear closure.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The patient was given a diagnosis of chondrodermatitis nodularis helicis (CNH), an inflammation of the cartilage and overlying skin causing a painful nodule of the helix. These lesions typically have a more prominent nodular component and a central ulceration or firm scale. They are thought to be due to chronic pressure on the ear.
There is a slight male predominance of CNH and onset is usually gradual. Patients often experience pain when sleeping on the affected side. The tenderness usually can be reproduced clinically by pressing on the lesion. The ears also are a high-risk area for actinic keratoses (AK) and nonmelanoma skin cancer (NMSC); if there is doubt about the diagnosis, a biopsy may be warranted to rule out AK or NMSC. In this patient, a shave biopsy was performed.
Various treatment regimens are available for CNH. The least invasive treatment approach is to use a “cut out” foam or a special donut-shaped pillow to protect the area from further pressure. By protecting from pressure and irritation, the lesion resolves in 57% to 92% of cases in clinical studies. Intralesional injection with 0.2 mL of 10 mg/mL triamcinolone acetonide is a simple in-office procedure that frequently helps the pain and may be curative; although, repeat injections may be necessary. Excision of the overlying skin and the affected cartilage is a more aggressive treatment with high success rates. More recently, treatment with topical nitroglycerin patches or photodynamic therapy have been described in small trials.
After confirming by a shave biopsy that the lesion was not cancerous, the patient returned for an elliptical excision of the lesion. A dermal curette was used to remove the rough abnormal inflammation of the underlying cartilage, and the elliptical wound was sutured with a linear closure.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Juul Nielsen L, Holkmann Olsen C, Lock-Andersen J. Therapeutic options of chondrodermatitis nodularis helicis [published online January 27, 2016]. Plast Surg Int. 2016;2016:4340168.
Juul Nielsen L, Holkmann Olsen C, Lock-Andersen J. Therapeutic options of chondrodermatitis nodularis helicis [published online January 27, 2016]. Plast Surg Int. 2016;2016:4340168.
Today’s top news highlights: COVID-19 could worsen gambling problems, food allergies less common than thought
Here are the stories our MDedge editors across specialties think you need to know about today:
Could COVID-19 worsen gambling problems?
Take isolation, add excess available time and anxiety about illness or finances and you get the potential to increase problem gambling behaviors during the COVID-19 pandemic. A call to action, recently published in the Journal of Addiction Medicine, says it’s essential to gather data and supply guidance on this issue. “People are likely to be experiencing stress at levels they haven’t experienced previously,” said coauthor Marc N. Potenza, MD, PhD, of Yale University, New Haven, Conn. While multiple factors can contribute to addictive behaviors, “with respect to the pandemic, one concern is that so-called negative reinforcement motivations – engaging in an addictive behavior to escape from depressed or negative mood states – may be a driving motivation for a significant number of people during this time,” he said. Read more.
Food allergies in children are less frequent than expected
Food allergies appear to be less common than previously reported among 6- to 10-year-olds in Europe, according to a recent study. Prevalance ranged from a low of 1.4% to a high of 3.8%, both of which are “considerably lower” than the 16% rate based on parental reports of symptoms such as rash, itching, or diarrhea, Linus Grabenhenrich, MD, MPH, and colleagues reported in Allergy. The most commonly reported allergies were to peanuts and hazelnuts, with a prevalence of just over 5% for both. Previous research on pediatric food allergy prevalence has largely consisted of single-center studies with heterogeneous designs, the researchers noted. Read more.
The grocery store hug
William G. Wilkoff, MD, grew up in a family that didn’t embrace hugging, but as a small-town pediatrician he warmed up to the concept so much that he would frequently hug a passing acquaintance at the grocery store. That’s something he misses in the current environment and that he doesn’t expect will return. “[N]early every week I encounter one or two people with whom I have a long and sometimes emotionally charged relationship,” Dr. Wilkoff wrote in a column on MDedge. “Nurses with whom I sweated over difficult delivery room resuscitations. Parents for whom their anxiety was getting in the way of their ability to parent. Parents and caregivers of complex multiply disabled children who are now adults. Peers who have lost a spouse or a child. I’m sure you have your own list of people who send off that we-need-to-hug spark.” Read more.
Identifying structural lesions of axial spondyloarthritis
What constitutes a structural lesion of the sacroiliac joints on MRI that’s indicative of axial spondyloarthritis (axSpA) has long been a matter of conjecture, but the Assessment of SpondyloArthritis International Society (ASAS) MRI Working Group has developed new definitions that show a high degree of specificity in identifying such lesions in the disease. “Previous studies have described structural lesions in different ways, precluding meaningful comparisons between studies,” Walter P. Maksymowych, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19. “The ASAS MRI group has generated updated consensus lesion definitions that describe each of the MRI lesions in the sacroiliac joint. These definitions have been validated by seven expert readers from the ASAS MRI group on MRI images from the ASAS classification cohort.” Read more.
Making the world’s skin crawl
Clinicians should be aware of the skin manifestations of COVID-19, especially when triaging patients. In a commentary published on MDedge, Kathleen M. Coerdt and Amor Khachemoune, MD, describe the dermatologic implications of COVID-19, including the clinical manifestations of the disease, risk reduction techniques for patients and providers, personal protective equipment-associated adverse reactions, and the financial impact on dermatologists. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Could COVID-19 worsen gambling problems?
Take isolation, add excess available time and anxiety about illness or finances and you get the potential to increase problem gambling behaviors during the COVID-19 pandemic. A call to action, recently published in the Journal of Addiction Medicine, says it’s essential to gather data and supply guidance on this issue. “People are likely to be experiencing stress at levels they haven’t experienced previously,” said coauthor Marc N. Potenza, MD, PhD, of Yale University, New Haven, Conn. While multiple factors can contribute to addictive behaviors, “with respect to the pandemic, one concern is that so-called negative reinforcement motivations – engaging in an addictive behavior to escape from depressed or negative mood states – may be a driving motivation for a significant number of people during this time,” he said. Read more.
Food allergies in children are less frequent than expected
Food allergies appear to be less common than previously reported among 6- to 10-year-olds in Europe, according to a recent study. Prevalance ranged from a low of 1.4% to a high of 3.8%, both of which are “considerably lower” than the 16% rate based on parental reports of symptoms such as rash, itching, or diarrhea, Linus Grabenhenrich, MD, MPH, and colleagues reported in Allergy. The most commonly reported allergies were to peanuts and hazelnuts, with a prevalence of just over 5% for both. Previous research on pediatric food allergy prevalence has largely consisted of single-center studies with heterogeneous designs, the researchers noted. Read more.
The grocery store hug
William G. Wilkoff, MD, grew up in a family that didn’t embrace hugging, but as a small-town pediatrician he warmed up to the concept so much that he would frequently hug a passing acquaintance at the grocery store. That’s something he misses in the current environment and that he doesn’t expect will return. “[N]early every week I encounter one or two people with whom I have a long and sometimes emotionally charged relationship,” Dr. Wilkoff wrote in a column on MDedge. “Nurses with whom I sweated over difficult delivery room resuscitations. Parents for whom their anxiety was getting in the way of their ability to parent. Parents and caregivers of complex multiply disabled children who are now adults. Peers who have lost a spouse or a child. I’m sure you have your own list of people who send off that we-need-to-hug spark.” Read more.
Identifying structural lesions of axial spondyloarthritis
What constitutes a structural lesion of the sacroiliac joints on MRI that’s indicative of axial spondyloarthritis (axSpA) has long been a matter of conjecture, but the Assessment of SpondyloArthritis International Society (ASAS) MRI Working Group has developed new definitions that show a high degree of specificity in identifying such lesions in the disease. “Previous studies have described structural lesions in different ways, precluding meaningful comparisons between studies,” Walter P. Maksymowych, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19. “The ASAS MRI group has generated updated consensus lesion definitions that describe each of the MRI lesions in the sacroiliac joint. These definitions have been validated by seven expert readers from the ASAS MRI group on MRI images from the ASAS classification cohort.” Read more.
Making the world’s skin crawl
Clinicians should be aware of the skin manifestations of COVID-19, especially when triaging patients. In a commentary published on MDedge, Kathleen M. Coerdt and Amor Khachemoune, MD, describe the dermatologic implications of COVID-19, including the clinical manifestations of the disease, risk reduction techniques for patients and providers, personal protective equipment-associated adverse reactions, and the financial impact on dermatologists. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Could COVID-19 worsen gambling problems?
Take isolation, add excess available time and anxiety about illness or finances and you get the potential to increase problem gambling behaviors during the COVID-19 pandemic. A call to action, recently published in the Journal of Addiction Medicine, says it’s essential to gather data and supply guidance on this issue. “People are likely to be experiencing stress at levels they haven’t experienced previously,” said coauthor Marc N. Potenza, MD, PhD, of Yale University, New Haven, Conn. While multiple factors can contribute to addictive behaviors, “with respect to the pandemic, one concern is that so-called negative reinforcement motivations – engaging in an addictive behavior to escape from depressed or negative mood states – may be a driving motivation for a significant number of people during this time,” he said. Read more.
Food allergies in children are less frequent than expected
Food allergies appear to be less common than previously reported among 6- to 10-year-olds in Europe, according to a recent study. Prevalance ranged from a low of 1.4% to a high of 3.8%, both of which are “considerably lower” than the 16% rate based on parental reports of symptoms such as rash, itching, or diarrhea, Linus Grabenhenrich, MD, MPH, and colleagues reported in Allergy. The most commonly reported allergies were to peanuts and hazelnuts, with a prevalence of just over 5% for both. Previous research on pediatric food allergy prevalence has largely consisted of single-center studies with heterogeneous designs, the researchers noted. Read more.
The grocery store hug
William G. Wilkoff, MD, grew up in a family that didn’t embrace hugging, but as a small-town pediatrician he warmed up to the concept so much that he would frequently hug a passing acquaintance at the grocery store. That’s something he misses in the current environment and that he doesn’t expect will return. “[N]early every week I encounter one or two people with whom I have a long and sometimes emotionally charged relationship,” Dr. Wilkoff wrote in a column on MDedge. “Nurses with whom I sweated over difficult delivery room resuscitations. Parents for whom their anxiety was getting in the way of their ability to parent. Parents and caregivers of complex multiply disabled children who are now adults. Peers who have lost a spouse or a child. I’m sure you have your own list of people who send off that we-need-to-hug spark.” Read more.
Identifying structural lesions of axial spondyloarthritis
What constitutes a structural lesion of the sacroiliac joints on MRI that’s indicative of axial spondyloarthritis (axSpA) has long been a matter of conjecture, but the Assessment of SpondyloArthritis International Society (ASAS) MRI Working Group has developed new definitions that show a high degree of specificity in identifying such lesions in the disease. “Previous studies have described structural lesions in different ways, precluding meaningful comparisons between studies,” Walter P. Maksymowych, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19. “The ASAS MRI group has generated updated consensus lesion definitions that describe each of the MRI lesions in the sacroiliac joint. These definitions have been validated by seven expert readers from the ASAS MRI group on MRI images from the ASAS classification cohort.” Read more.
Making the world’s skin crawl
Clinicians should be aware of the skin manifestations of COVID-19, especially when triaging patients. In a commentary published on MDedge, Kathleen M. Coerdt and Amor Khachemoune, MD, describe the dermatologic implications of COVID-19, including the clinical manifestations of the disease, risk reduction techniques for patients and providers, personal protective equipment-associated adverse reactions, and the financial impact on dermatologists. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Atopic dermatitis in adults, children linked to neuropsychiatric disorders
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
FROM SID 2020
Cancer risk elevated in hidradenitis suppurativa patients
.
HS is associated with severe comorbidities, and previous studies have suggested a link between HS and cancer development, wrote Joon Min Jung, MD, of the University of Ulsan College of Medicine, Seoul, Korea, and colleagues.
“The aberrant immune response and chronic inflammation in HS and genetic and environmental factors associated with the disease may all be factors in the development of cancer,” but large, population-based studies of cancer in HS patients are limited, they noted.
In a study published in JAMA Dermatology, the researchers reviewed data from 22,468 adults with HS and 179,734 matched controls, in the Korean National Health Insurance System, seen by physicians between January 2009 and December 2017. The average age of the participants was 34 years, and 64% were male.
Overall, HS patients had a significantly higher risk of cancer compared with controls, with an adjusted hazard ratio (aHR) of 1.28.
As for specific cancers, HS patients had a significantly higher risk for Hodgkin lymphoma (aHR 5.08), oral cavity and pharyngeal cancer (aHR 3.10), central nervous system cancer (aHR 2.40), nonmelanoma skin cancer (aHR 2.06), prostate cancer (aHR 2.05), and colorectal cancer (aHR 1.45).
The risk of any cancer was not significantly different between women with HS and female controls (after adjustment for comorbidities), but was significantly higher among men with HS compared with male controls, also after adjustment for comorbidities (aHR, 1.37). In addition, HS patients in both younger (less than 40 years) and older (aged 40 years and older) age groups had increased cancer risk compared with age-matched controls. Overall cancer risk and the risk of most cancer types were higher among HS patients with moderate to severe disease than in those with mild disease, with the exception of nonmelanoma skin cancer, prostate cancer, lymphoma, and leukemia.
“Overall cancer risk showed a tendency to increase with worsening HS severity, reinforcing the possibility of an association between HS and cancer development,” the researchers noted. “However, we could not identify tendencies in some specific cancers, such as nonmelanoma skin cancer, CNS cancer, and prostate cancer, because the number of occurrences of those cancers was too small in the group with moderate to severe HS.”
The study findings were limited by several factors including the potential underestimate of HS cases in the population and the inability of the study design to adjust for factors including smoking status, alcohol use, and obesity, the researchers noted. However, the results support an increased cancer risk in HS patients and suggest the need to promote lifestyle modifications to reduce risk, and to increase cancer surveillance in these patients, they said. “For early detection of skin cancer, more aggressive histologic examination and a high level of suspicion are required,” they added.
The study was supported by the National Research Foundation of Korea and the Korea Health Technology R&D Project. The researchers had no financial conflicts to disclose.
SOURCE: Jung JM et al. JAMA Dermatol. 2020 May 27. doi: 10.1001/jamadermatol.2020.1422.
.
HS is associated with severe comorbidities, and previous studies have suggested a link between HS and cancer development, wrote Joon Min Jung, MD, of the University of Ulsan College of Medicine, Seoul, Korea, and colleagues.
“The aberrant immune response and chronic inflammation in HS and genetic and environmental factors associated with the disease may all be factors in the development of cancer,” but large, population-based studies of cancer in HS patients are limited, they noted.
In a study published in JAMA Dermatology, the researchers reviewed data from 22,468 adults with HS and 179,734 matched controls, in the Korean National Health Insurance System, seen by physicians between January 2009 and December 2017. The average age of the participants was 34 years, and 64% were male.
Overall, HS patients had a significantly higher risk of cancer compared with controls, with an adjusted hazard ratio (aHR) of 1.28.
As for specific cancers, HS patients had a significantly higher risk for Hodgkin lymphoma (aHR 5.08), oral cavity and pharyngeal cancer (aHR 3.10), central nervous system cancer (aHR 2.40), nonmelanoma skin cancer (aHR 2.06), prostate cancer (aHR 2.05), and colorectal cancer (aHR 1.45).
The risk of any cancer was not significantly different between women with HS and female controls (after adjustment for comorbidities), but was significantly higher among men with HS compared with male controls, also after adjustment for comorbidities (aHR, 1.37). In addition, HS patients in both younger (less than 40 years) and older (aged 40 years and older) age groups had increased cancer risk compared with age-matched controls. Overall cancer risk and the risk of most cancer types were higher among HS patients with moderate to severe disease than in those with mild disease, with the exception of nonmelanoma skin cancer, prostate cancer, lymphoma, and leukemia.
“Overall cancer risk showed a tendency to increase with worsening HS severity, reinforcing the possibility of an association between HS and cancer development,” the researchers noted. “However, we could not identify tendencies in some specific cancers, such as nonmelanoma skin cancer, CNS cancer, and prostate cancer, because the number of occurrences of those cancers was too small in the group with moderate to severe HS.”
The study findings were limited by several factors including the potential underestimate of HS cases in the population and the inability of the study design to adjust for factors including smoking status, alcohol use, and obesity, the researchers noted. However, the results support an increased cancer risk in HS patients and suggest the need to promote lifestyle modifications to reduce risk, and to increase cancer surveillance in these patients, they said. “For early detection of skin cancer, more aggressive histologic examination and a high level of suspicion are required,” they added.
The study was supported by the National Research Foundation of Korea and the Korea Health Technology R&D Project. The researchers had no financial conflicts to disclose.
SOURCE: Jung JM et al. JAMA Dermatol. 2020 May 27. doi: 10.1001/jamadermatol.2020.1422.
.
HS is associated with severe comorbidities, and previous studies have suggested a link between HS and cancer development, wrote Joon Min Jung, MD, of the University of Ulsan College of Medicine, Seoul, Korea, and colleagues.
“The aberrant immune response and chronic inflammation in HS and genetic and environmental factors associated with the disease may all be factors in the development of cancer,” but large, population-based studies of cancer in HS patients are limited, they noted.
In a study published in JAMA Dermatology, the researchers reviewed data from 22,468 adults with HS and 179,734 matched controls, in the Korean National Health Insurance System, seen by physicians between January 2009 and December 2017. The average age of the participants was 34 years, and 64% were male.
Overall, HS patients had a significantly higher risk of cancer compared with controls, with an adjusted hazard ratio (aHR) of 1.28.
As for specific cancers, HS patients had a significantly higher risk for Hodgkin lymphoma (aHR 5.08), oral cavity and pharyngeal cancer (aHR 3.10), central nervous system cancer (aHR 2.40), nonmelanoma skin cancer (aHR 2.06), prostate cancer (aHR 2.05), and colorectal cancer (aHR 1.45).
The risk of any cancer was not significantly different between women with HS and female controls (after adjustment for comorbidities), but was significantly higher among men with HS compared with male controls, also after adjustment for comorbidities (aHR, 1.37). In addition, HS patients in both younger (less than 40 years) and older (aged 40 years and older) age groups had increased cancer risk compared with age-matched controls. Overall cancer risk and the risk of most cancer types were higher among HS patients with moderate to severe disease than in those with mild disease, with the exception of nonmelanoma skin cancer, prostate cancer, lymphoma, and leukemia.
“Overall cancer risk showed a tendency to increase with worsening HS severity, reinforcing the possibility of an association between HS and cancer development,” the researchers noted. “However, we could not identify tendencies in some specific cancers, such as nonmelanoma skin cancer, CNS cancer, and prostate cancer, because the number of occurrences of those cancers was too small in the group with moderate to severe HS.”
The study findings were limited by several factors including the potential underestimate of HS cases in the population and the inability of the study design to adjust for factors including smoking status, alcohol use, and obesity, the researchers noted. However, the results support an increased cancer risk in HS patients and suggest the need to promote lifestyle modifications to reduce risk, and to increase cancer surveillance in these patients, they said. “For early detection of skin cancer, more aggressive histologic examination and a high level of suspicion are required,” they added.
The study was supported by the National Research Foundation of Korea and the Korea Health Technology R&D Project. The researchers had no financial conflicts to disclose.
SOURCE: Jung JM et al. JAMA Dermatol. 2020 May 27. doi: 10.1001/jamadermatol.2020.1422.
FROM JAMA DERMATOLOGY
MCC response varies based on immunosuppression type, especially CLL
Patients with Merkel cell carcinoma and chronic immunosuppression may fare better or worse on immunotherapy based on the reason for immunosuppression, according to recent research at the annual meeting of the Society for Investigative Dermatology, held virtually.
About 10% of patients with Merkel cell carcinoma (MCC) are immunosuppressed at diagnosis, and these patients tend to have a more aggressive disease course and worse disease-specific survival compared with immunocompetent patients, Lauren Zawacki, a research assistant in the Nghiem Lab at the University of Washington, Seattle, said in her presentation. Although patients are receiving immune checkpoint inhibitors such as anti-PD-1 and anti-PD-L1 as treatments, the efficacy and side effects on immunosuppressed patients have not been well studied because many of these patients are not eligible for clinical trials.
Ms. Zawacki and colleagues analyzed data from a prospective Seattle registry of 1,442 patients with MCC, identifying 179 patients with MCC who had chronic immunosuppression due to chronic lymphocytic leukemia (CLL), solid organ transplants, autoimmune disorders, other hematological malignancies, and HIV and AIDS. Non-Hodgkin lymphoma comprised 7 of 8 patients in the group with other hematological malignancies, and Crohn’s disease made up 5 of 6 patients in the autoimmune disorder group. Of the 179 patients with MCC and immunosuppression, 31 patients were treated with either anti-PD-1 or anti-PD-L1 therapy.
There was an objective response rate of 52%, with 14 patients having a complete response, 2 patients having a partial response, and 15 patients experiencing disease progression. Of the patients with disease progression, 11 died of MCC. The response rate in immunocompromised patients is similar to results seen by her group in immunocompetent patients (Nghiem P et al. N Engl J Med 2016; 374:2542-52), said Ms. Zawacki. “While the overall objective response rate is comparable between immunocompetent and immunosuppressed patients, the response rates vary greatly between the different types of immunosuppression,” she said.
When grouping response rates by immunosuppression type, they found 2 of 11 patients with CLL (18%) and 2 of 6 patients with autoimmune disease (33%) had an objective response, while 2 of 3 patients with HIV/AIDS (66%) and 7 of 7 patients with other hematologic malignancies (100%) had an objective response.
“While the numbers of the cohort are small, there still seems to be a considerable difference in the response rate between the different types of immune suppression, which is critical when we’re treating patients who typically have a more aggressive disease course,” said Ms. Zawacki.
In particular, the finding of no patients with MCC and CLL achieving a complete response interested Ms. Zawacki and her colleagues, since about one-fourth of patients in the Seattle registry have this combination of disease. “Not only did none of the CLL patients have a complete response, but 7 out of the 11 patients with CLL died from MCC,” she explained. When examining further, the researchers found 45% of patients in this group discontinued because of side effects of immunotherapy and had a median time to recurrence of 1.5 months. “This finding suggests that CLL in particular plays a large role in impairing the function of the immune system, leading to not only a more aggressive disease course, but a poorer response to immunotherapy,” she said.
“There is a significant need for improved interventions for patients with CLL and autoimmune disorders,” she added. “Research for immunosuppressed patients is critical given the associated aggressive disease course and their lack of inclusion in clinical trials.”
Ms. Zawacki acknowledged the small number of patients in the study as a limitation, and patients who received follow-up at outside facilities may have received slightly different care, which could impact adverse event reporting or reasons for study discontinuation.
“A multi-institutional study would be beneficial to expand the number of patients in that cohort and to help confirm the trend observed in this study. In addition, future studies should assess the role of combination systemic therapy, such as neutron radiation and immunotherapy together in order to see if the objective response can be approved among immunosuppressed patients,” she said.
This study was supported by funding from the MCC Patient Gift Fund, the National Cancer Institute, and a grant from NIH. Ms. Zawacki reports no relevant conflicts of interest.
SOURCE: Zawacki L. SID 2020, Abstract 497.
Patients with Merkel cell carcinoma and chronic immunosuppression may fare better or worse on immunotherapy based on the reason for immunosuppression, according to recent research at the annual meeting of the Society for Investigative Dermatology, held virtually.
About 10% of patients with Merkel cell carcinoma (MCC) are immunosuppressed at diagnosis, and these patients tend to have a more aggressive disease course and worse disease-specific survival compared with immunocompetent patients, Lauren Zawacki, a research assistant in the Nghiem Lab at the University of Washington, Seattle, said in her presentation. Although patients are receiving immune checkpoint inhibitors such as anti-PD-1 and anti-PD-L1 as treatments, the efficacy and side effects on immunosuppressed patients have not been well studied because many of these patients are not eligible for clinical trials.
Ms. Zawacki and colleagues analyzed data from a prospective Seattle registry of 1,442 patients with MCC, identifying 179 patients with MCC who had chronic immunosuppression due to chronic lymphocytic leukemia (CLL), solid organ transplants, autoimmune disorders, other hematological malignancies, and HIV and AIDS. Non-Hodgkin lymphoma comprised 7 of 8 patients in the group with other hematological malignancies, and Crohn’s disease made up 5 of 6 patients in the autoimmune disorder group. Of the 179 patients with MCC and immunosuppression, 31 patients were treated with either anti-PD-1 or anti-PD-L1 therapy.
There was an objective response rate of 52%, with 14 patients having a complete response, 2 patients having a partial response, and 15 patients experiencing disease progression. Of the patients with disease progression, 11 died of MCC. The response rate in immunocompromised patients is similar to results seen by her group in immunocompetent patients (Nghiem P et al. N Engl J Med 2016; 374:2542-52), said Ms. Zawacki. “While the overall objective response rate is comparable between immunocompetent and immunosuppressed patients, the response rates vary greatly between the different types of immunosuppression,” she said.
When grouping response rates by immunosuppression type, they found 2 of 11 patients with CLL (18%) and 2 of 6 patients with autoimmune disease (33%) had an objective response, while 2 of 3 patients with HIV/AIDS (66%) and 7 of 7 patients with other hematologic malignancies (100%) had an objective response.
“While the numbers of the cohort are small, there still seems to be a considerable difference in the response rate between the different types of immune suppression, which is critical when we’re treating patients who typically have a more aggressive disease course,” said Ms. Zawacki.
In particular, the finding of no patients with MCC and CLL achieving a complete response interested Ms. Zawacki and her colleagues, since about one-fourth of patients in the Seattle registry have this combination of disease. “Not only did none of the CLL patients have a complete response, but 7 out of the 11 patients with CLL died from MCC,” she explained. When examining further, the researchers found 45% of patients in this group discontinued because of side effects of immunotherapy and had a median time to recurrence of 1.5 months. “This finding suggests that CLL in particular plays a large role in impairing the function of the immune system, leading to not only a more aggressive disease course, but a poorer response to immunotherapy,” she said.
“There is a significant need for improved interventions for patients with CLL and autoimmune disorders,” she added. “Research for immunosuppressed patients is critical given the associated aggressive disease course and their lack of inclusion in clinical trials.”
Ms. Zawacki acknowledged the small number of patients in the study as a limitation, and patients who received follow-up at outside facilities may have received slightly different care, which could impact adverse event reporting or reasons for study discontinuation.
“A multi-institutional study would be beneficial to expand the number of patients in that cohort and to help confirm the trend observed in this study. In addition, future studies should assess the role of combination systemic therapy, such as neutron radiation and immunotherapy together in order to see if the objective response can be approved among immunosuppressed patients,” she said.
This study was supported by funding from the MCC Patient Gift Fund, the National Cancer Institute, and a grant from NIH. Ms. Zawacki reports no relevant conflicts of interest.
SOURCE: Zawacki L. SID 2020, Abstract 497.
Patients with Merkel cell carcinoma and chronic immunosuppression may fare better or worse on immunotherapy based on the reason for immunosuppression, according to recent research at the annual meeting of the Society for Investigative Dermatology, held virtually.
About 10% of patients with Merkel cell carcinoma (MCC) are immunosuppressed at diagnosis, and these patients tend to have a more aggressive disease course and worse disease-specific survival compared with immunocompetent patients, Lauren Zawacki, a research assistant in the Nghiem Lab at the University of Washington, Seattle, said in her presentation. Although patients are receiving immune checkpoint inhibitors such as anti-PD-1 and anti-PD-L1 as treatments, the efficacy and side effects on immunosuppressed patients have not been well studied because many of these patients are not eligible for clinical trials.
Ms. Zawacki and colleagues analyzed data from a prospective Seattle registry of 1,442 patients with MCC, identifying 179 patients with MCC who had chronic immunosuppression due to chronic lymphocytic leukemia (CLL), solid organ transplants, autoimmune disorders, other hematological malignancies, and HIV and AIDS. Non-Hodgkin lymphoma comprised 7 of 8 patients in the group with other hematological malignancies, and Crohn’s disease made up 5 of 6 patients in the autoimmune disorder group. Of the 179 patients with MCC and immunosuppression, 31 patients were treated with either anti-PD-1 or anti-PD-L1 therapy.
There was an objective response rate of 52%, with 14 patients having a complete response, 2 patients having a partial response, and 15 patients experiencing disease progression. Of the patients with disease progression, 11 died of MCC. The response rate in immunocompromised patients is similar to results seen by her group in immunocompetent patients (Nghiem P et al. N Engl J Med 2016; 374:2542-52), said Ms. Zawacki. “While the overall objective response rate is comparable between immunocompetent and immunosuppressed patients, the response rates vary greatly between the different types of immunosuppression,” she said.
When grouping response rates by immunosuppression type, they found 2 of 11 patients with CLL (18%) and 2 of 6 patients with autoimmune disease (33%) had an objective response, while 2 of 3 patients with HIV/AIDS (66%) and 7 of 7 patients with other hematologic malignancies (100%) had an objective response.
“While the numbers of the cohort are small, there still seems to be a considerable difference in the response rate between the different types of immune suppression, which is critical when we’re treating patients who typically have a more aggressive disease course,” said Ms. Zawacki.
In particular, the finding of no patients with MCC and CLL achieving a complete response interested Ms. Zawacki and her colleagues, since about one-fourth of patients in the Seattle registry have this combination of disease. “Not only did none of the CLL patients have a complete response, but 7 out of the 11 patients with CLL died from MCC,” she explained. When examining further, the researchers found 45% of patients in this group discontinued because of side effects of immunotherapy and had a median time to recurrence of 1.5 months. “This finding suggests that CLL in particular plays a large role in impairing the function of the immune system, leading to not only a more aggressive disease course, but a poorer response to immunotherapy,” she said.
“There is a significant need for improved interventions for patients with CLL and autoimmune disorders,” she added. “Research for immunosuppressed patients is critical given the associated aggressive disease course and their lack of inclusion in clinical trials.”
Ms. Zawacki acknowledged the small number of patients in the study as a limitation, and patients who received follow-up at outside facilities may have received slightly different care, which could impact adverse event reporting or reasons for study discontinuation.
“A multi-institutional study would be beneficial to expand the number of patients in that cohort and to help confirm the trend observed in this study. In addition, future studies should assess the role of combination systemic therapy, such as neutron radiation and immunotherapy together in order to see if the objective response can be approved among immunosuppressed patients,” she said.
This study was supported by funding from the MCC Patient Gift Fund, the National Cancer Institute, and a grant from NIH. Ms. Zawacki reports no relevant conflicts of interest.
SOURCE: Zawacki L. SID 2020, Abstract 497.
FROM SID 2020
California wildfires caused uptick in clinic visits for atopic dermatitis, itch
During the deadliest wildfire in California’s history in 2018, dermatology clinics 175 miles away at the University of California, San Francisco, experienced an increase in the number of pediatric and adult visits for pruritus and atopic dermatitis associated with air pollution created from the wildfire, according to research presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
Raj Fadadu, a medical student at the University of California, San Francisco, said in his presentation.
Not many studies have examined this potential association, but includes those that have found significant positive associations between exposure to air pollution and pruritus, the development of AD, and exacerbation of AD (J Allergy Clin Immunol. 2014 Nov;134[5]:993-9). Another study found outpatient visits for patients with eczema and dermatitis in Beijing increased as the level of particulate matter, nitrogen dioxide, and sulfur dioxide concentrations increased (Environ Sci Process Impacts. 2019 Jan 23;21[1]:163-73).
Mr. Faduda and colleagues set out to determine whether the number of appointments for and severity of skin disease increased as a result of the 2018 Camp Fire, which started in Paradise, Calif., using measures of air pollution and clinic visits in years where California did not experience a wildfire event as controls. Using the National Oceanic and Atmospheric Administration Hazard Mapping System for fire and smoke, the researchers graphed smoke plume density scores and particulate matter (PM2.5) concentrations in the area. They then calculated the number of UCSF dermatology clinic visits for AD/eczema, and measured severity of skin disease with appointments for itch symptoms, and the number of prescribed medications during that time using ICD-10 codes.
The Camp Fire rapidly spread over a period of 17 days, between Nov. 8 and 25, 2018, during which time, PM2.5 particulate matter concentrations increased 10-fold, while the NOAA smoke plume density score sharply increased. More pediatric and adult patients also seemed to be visiting clinics during this time, compared with several weeks before and several weeks after the fire, prompting a more expanded analysis of this signal, Mr. Fadadu said.
He and his coinvestigators compared data between October 2015 and February 2016 – a period of time where there were no wildfires in California – with data in 2018, when the Camp Fire occurred. They collected data on 3,448 adults and 699 children across 3 years with a total of 5,539 adult appointments for AD, 924 pediatric appointments for AD, 1,319 adult itch appointments, and 294 pediatric itch appointments. Cumulative and exposure lags were used to measure the effect of the wildfire in a Poisson regression analysis.
They found that, during the wildfire, pediatric AD weekly clinic visits were 1.75 times higher (95% confidence interval, 1.21-2.50) and pediatric itch visits were 2.10 times higher (95% CI, 1.44-3.00), compared with weeks where there was no fire. During the wildfire, pediatric AD clinic visits increased by 8% (rate ratio, 1.08; 95% CI, 1.04-1.12) per 10 mcg/m3 increase in PM2.5 concentration.
In adults, clinic visits for AD were 1.28 times higher (95% CI, 1.08-1.51) during the wildfire, compared with nonfire weeks. While there was a positive association between pollution exposure and adult AD, “this effect is less than what we observed” for pediatric AD visits, said Mr. Fadadu. Air pollution was positively associated with the development of itch symptoms in adults and more prescriptions for AD medications, but the results were not statistically significant.
“This may be explained by the fact that 80% of pediatric itch patients carried an AD diagnosis, while in contrast, only half of the adult itch patients also have a diagnosis of AD,” he said.
While there are several possible limitations of the research, including assessment of air pollution exposure, Mr. Fadadu said, “these results can inform how dermatologists counsel patients during future episodes of poor air quality, as well as expand comprehension of the broader health effects of climate change that can significantly impact quality of life.”
This study was funded by the UCSF Summer Explore Fellowship, Marguerite Schoeneman Award, and Joint Medical Program Thesis Grant.
During the deadliest wildfire in California’s history in 2018, dermatology clinics 175 miles away at the University of California, San Francisco, experienced an increase in the number of pediatric and adult visits for pruritus and atopic dermatitis associated with air pollution created from the wildfire, according to research presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
Raj Fadadu, a medical student at the University of California, San Francisco, said in his presentation.
Not many studies have examined this potential association, but includes those that have found significant positive associations between exposure to air pollution and pruritus, the development of AD, and exacerbation of AD (J Allergy Clin Immunol. 2014 Nov;134[5]:993-9). Another study found outpatient visits for patients with eczema and dermatitis in Beijing increased as the level of particulate matter, nitrogen dioxide, and sulfur dioxide concentrations increased (Environ Sci Process Impacts. 2019 Jan 23;21[1]:163-73).
Mr. Faduda and colleagues set out to determine whether the number of appointments for and severity of skin disease increased as a result of the 2018 Camp Fire, which started in Paradise, Calif., using measures of air pollution and clinic visits in years where California did not experience a wildfire event as controls. Using the National Oceanic and Atmospheric Administration Hazard Mapping System for fire and smoke, the researchers graphed smoke plume density scores and particulate matter (PM2.5) concentrations in the area. They then calculated the number of UCSF dermatology clinic visits for AD/eczema, and measured severity of skin disease with appointments for itch symptoms, and the number of prescribed medications during that time using ICD-10 codes.
The Camp Fire rapidly spread over a period of 17 days, between Nov. 8 and 25, 2018, during which time, PM2.5 particulate matter concentrations increased 10-fold, while the NOAA smoke plume density score sharply increased. More pediatric and adult patients also seemed to be visiting clinics during this time, compared with several weeks before and several weeks after the fire, prompting a more expanded analysis of this signal, Mr. Fadadu said.
He and his coinvestigators compared data between October 2015 and February 2016 – a period of time where there were no wildfires in California – with data in 2018, when the Camp Fire occurred. They collected data on 3,448 adults and 699 children across 3 years with a total of 5,539 adult appointments for AD, 924 pediatric appointments for AD, 1,319 adult itch appointments, and 294 pediatric itch appointments. Cumulative and exposure lags were used to measure the effect of the wildfire in a Poisson regression analysis.
They found that, during the wildfire, pediatric AD weekly clinic visits were 1.75 times higher (95% confidence interval, 1.21-2.50) and pediatric itch visits were 2.10 times higher (95% CI, 1.44-3.00), compared with weeks where there was no fire. During the wildfire, pediatric AD clinic visits increased by 8% (rate ratio, 1.08; 95% CI, 1.04-1.12) per 10 mcg/m3 increase in PM2.5 concentration.
In adults, clinic visits for AD were 1.28 times higher (95% CI, 1.08-1.51) during the wildfire, compared with nonfire weeks. While there was a positive association between pollution exposure and adult AD, “this effect is less than what we observed” for pediatric AD visits, said Mr. Fadadu. Air pollution was positively associated with the development of itch symptoms in adults and more prescriptions for AD medications, but the results were not statistically significant.
“This may be explained by the fact that 80% of pediatric itch patients carried an AD diagnosis, while in contrast, only half of the adult itch patients also have a diagnosis of AD,” he said.
While there are several possible limitations of the research, including assessment of air pollution exposure, Mr. Fadadu said, “these results can inform how dermatologists counsel patients during future episodes of poor air quality, as well as expand comprehension of the broader health effects of climate change that can significantly impact quality of life.”
This study was funded by the UCSF Summer Explore Fellowship, Marguerite Schoeneman Award, and Joint Medical Program Thesis Grant.
During the deadliest wildfire in California’s history in 2018, dermatology clinics 175 miles away at the University of California, San Francisco, experienced an increase in the number of pediatric and adult visits for pruritus and atopic dermatitis associated with air pollution created from the wildfire, according to research presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
Raj Fadadu, a medical student at the University of California, San Francisco, said in his presentation.
Not many studies have examined this potential association, but includes those that have found significant positive associations between exposure to air pollution and pruritus, the development of AD, and exacerbation of AD (J Allergy Clin Immunol. 2014 Nov;134[5]:993-9). Another study found outpatient visits for patients with eczema and dermatitis in Beijing increased as the level of particulate matter, nitrogen dioxide, and sulfur dioxide concentrations increased (Environ Sci Process Impacts. 2019 Jan 23;21[1]:163-73).
Mr. Faduda and colleagues set out to determine whether the number of appointments for and severity of skin disease increased as a result of the 2018 Camp Fire, which started in Paradise, Calif., using measures of air pollution and clinic visits in years where California did not experience a wildfire event as controls. Using the National Oceanic and Atmospheric Administration Hazard Mapping System for fire and smoke, the researchers graphed smoke plume density scores and particulate matter (PM2.5) concentrations in the area. They then calculated the number of UCSF dermatology clinic visits for AD/eczema, and measured severity of skin disease with appointments for itch symptoms, and the number of prescribed medications during that time using ICD-10 codes.
The Camp Fire rapidly spread over a period of 17 days, between Nov. 8 and 25, 2018, during which time, PM2.5 particulate matter concentrations increased 10-fold, while the NOAA smoke plume density score sharply increased. More pediatric and adult patients also seemed to be visiting clinics during this time, compared with several weeks before and several weeks after the fire, prompting a more expanded analysis of this signal, Mr. Fadadu said.
He and his coinvestigators compared data between October 2015 and February 2016 – a period of time where there were no wildfires in California – with data in 2018, when the Camp Fire occurred. They collected data on 3,448 adults and 699 children across 3 years with a total of 5,539 adult appointments for AD, 924 pediatric appointments for AD, 1,319 adult itch appointments, and 294 pediatric itch appointments. Cumulative and exposure lags were used to measure the effect of the wildfire in a Poisson regression analysis.
They found that, during the wildfire, pediatric AD weekly clinic visits were 1.75 times higher (95% confidence interval, 1.21-2.50) and pediatric itch visits were 2.10 times higher (95% CI, 1.44-3.00), compared with weeks where there was no fire. During the wildfire, pediatric AD clinic visits increased by 8% (rate ratio, 1.08; 95% CI, 1.04-1.12) per 10 mcg/m3 increase in PM2.5 concentration.
In adults, clinic visits for AD were 1.28 times higher (95% CI, 1.08-1.51) during the wildfire, compared with nonfire weeks. While there was a positive association between pollution exposure and adult AD, “this effect is less than what we observed” for pediatric AD visits, said Mr. Fadadu. Air pollution was positively associated with the development of itch symptoms in adults and more prescriptions for AD medications, but the results were not statistically significant.
“This may be explained by the fact that 80% of pediatric itch patients carried an AD diagnosis, while in contrast, only half of the adult itch patients also have a diagnosis of AD,” he said.
While there are several possible limitations of the research, including assessment of air pollution exposure, Mr. Fadadu said, “these results can inform how dermatologists counsel patients during future episodes of poor air quality, as well as expand comprehension of the broader health effects of climate change that can significantly impact quality of life.”
This study was funded by the UCSF Summer Explore Fellowship, Marguerite Schoeneman Award, and Joint Medical Program Thesis Grant.
FROM SID 2020
Rapid-onset rash
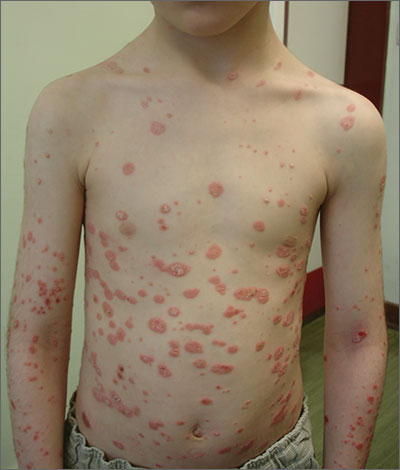
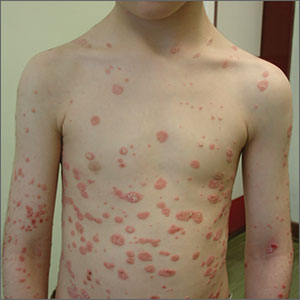
A diagnosis of guttate psoriasis was made based on the physical exam findings and the preceding group A beta-hemolytic streptococcal infection.
This condition affects approximately 2% of all patients with psoriasis; it is characterized by the acute onset of multiple erythematous and scaly papules and small plaques that look like droplets (“gutta”). It tends to affect children and young adults and typically occurs following an acute infection (eg, streptococcal pharyngitis). In this case, a rapid strep test was positive for group A Streptococcus, which supported the diagnosis.
The differential includes skin conditions such as pityriasis rosea, tinea corporis, and contact dermatitis.
The first-line treatment for streptococcal infection is amoxicillin (50 mg/kg/d [maximum: 1000 mg/d] orally for 10 d) or penicillin G benzathine (for children < 60 lb, 6 × 105 units intramuscularly; children ≥ 60 lb, 1.2 × 106 units intramuscularly). For the psoriasis lesions, treatment options include topical steroids, vitamin D derivatives, or combinations of both. In most cases, guttate psoriasis completely resolves. However, one-third of children with guttate psoriasis go on to develop plaque psoriasis later in life.
This patient was treated with penicillin G benzathine (1.2 × 106 units intramuscularly) and a calcipotriol/betamethasone combination gel. However, a less costly treatment is generic betamethasone (or triamcinolone) and/or generic calcipotriol (a vitamin D derivative). The streptococcal infection and skin lesions completely resolved. No adverse events were reported, and no relapse was observed after 3 months.
This case was adapted from: Matos RS, Torres T. Rapid onset rash in child. J Fam Pract. 2018;67:E1-E2.

A diagnosis of guttate psoriasis was made based on the physical exam findings and the preceding group A beta-hemolytic streptococcal infection.
This condition affects approximately 2% of all patients with psoriasis; it is characterized by the acute onset of multiple erythematous and scaly papules and small plaques that look like droplets (“gutta”). It tends to affect children and young adults and typically occurs following an acute infection (eg, streptococcal pharyngitis). In this case, a rapid strep test was positive for group A Streptococcus, which supported the diagnosis.
The differential includes skin conditions such as pityriasis rosea, tinea corporis, and contact dermatitis.
The first-line treatment for streptococcal infection is amoxicillin (50 mg/kg/d [maximum: 1000 mg/d] orally for 10 d) or penicillin G benzathine (for children < 60 lb, 6 × 105 units intramuscularly; children ≥ 60 lb, 1.2 × 106 units intramuscularly). For the psoriasis lesions, treatment options include topical steroids, vitamin D derivatives, or combinations of both. In most cases, guttate psoriasis completely resolves. However, one-third of children with guttate psoriasis go on to develop plaque psoriasis later in life.
This patient was treated with penicillin G benzathine (1.2 × 106 units intramuscularly) and a calcipotriol/betamethasone combination gel. However, a less costly treatment is generic betamethasone (or triamcinolone) and/or generic calcipotriol (a vitamin D derivative). The streptococcal infection and skin lesions completely resolved. No adverse events were reported, and no relapse was observed after 3 months.
This case was adapted from: Matos RS, Torres T. Rapid onset rash in child. J Fam Pract. 2018;67:E1-E2.

A diagnosis of guttate psoriasis was made based on the physical exam findings and the preceding group A beta-hemolytic streptococcal infection.
This condition affects approximately 2% of all patients with psoriasis; it is characterized by the acute onset of multiple erythematous and scaly papules and small plaques that look like droplets (“gutta”). It tends to affect children and young adults and typically occurs following an acute infection (eg, streptococcal pharyngitis). In this case, a rapid strep test was positive for group A Streptococcus, which supported the diagnosis.
The differential includes skin conditions such as pityriasis rosea, tinea corporis, and contact dermatitis.
The first-line treatment for streptococcal infection is amoxicillin (50 mg/kg/d [maximum: 1000 mg/d] orally for 10 d) or penicillin G benzathine (for children < 60 lb, 6 × 105 units intramuscularly; children ≥ 60 lb, 1.2 × 106 units intramuscularly). For the psoriasis lesions, treatment options include topical steroids, vitamin D derivatives, or combinations of both. In most cases, guttate psoriasis completely resolves. However, one-third of children with guttate psoriasis go on to develop plaque psoriasis later in life.
This patient was treated with penicillin G benzathine (1.2 × 106 units intramuscularly) and a calcipotriol/betamethasone combination gel. However, a less costly treatment is generic betamethasone (or triamcinolone) and/or generic calcipotriol (a vitamin D derivative). The streptococcal infection and skin lesions completely resolved. No adverse events were reported, and no relapse was observed after 3 months.
This case was adapted from: Matos RS, Torres T. Rapid onset rash in child. J Fam Pract. 2018;67:E1-E2.
COVID-19 complicates prescribing for children with inflammatory skin disease
designed to offer guidance to specialists and nonspecialists faced with tough choices about risks.
Some 87% reported that they were reducing the frequency of lab monitoring for some medications, while more than half said they had reached out to patients and their families to discuss the implications of continuing or stopping a drug.
Virtually all – 97% – said that the COVID-19 crisis had affected their decision to initiate immunosuppressive medications, with 84% saying the decision depended on a patient’s risk factors for contracting COVID-19 infection, and also the potential consequences of infection while treated, compared with the risks of not optimally treating the skin condition.
To develop a consensus-based guidance for clinicians, published online April 22 in Pediatric Dermatology, Kelly Cordoro, MD, professor of dermatology at the University of California, San Francisco, assembled a task force of pediatric dermatologists at academic institutions (the Pediatric Dermatology COVID-19 Response Task Force). Together with Sean Reynolds, MD, a pediatric dermatology fellow at UCSF and colleagues, they issued a survey to the 37 members of the task force with questions on how the pandemic has affected their prescribing decisions and certain therapies specifically. All the recipients responded.
The dermatologists were asked about conventional systemic and biologic medications. Most felt confident in continuing biologics, with 78% saying they would keep patients with no signs of COVID-19 exposure or infection on tumor necrosis factor (TNF) inhibitors. More than 90% of respondents said they would continue patients on dupilumab, as well as anti–interleukin (IL)–17, anti–IL-12/23, and anti–IL-23 therapies.
Responses varied more on approaches to the nonbiologic treatments. Fewer than half (46%) said they would continue patients without apparent COVID-19 exposure on systemic steroids, with another 46% saying it depended on the clinical context.
For other systemic therapies, respondents were more likely to want to continue their patients with no signs or symptoms of COVID-19 on methotrexate and apremilast (78% and 83%, respectively) than others (mycophenolate mofetil, azathioprine, cyclosporine, and JAK inhibitors), which saw between 50% and 60% support in the survey.
Patients on any immunosuppressive medications with likely exposure to COVID-19 or who test positive for the virus should be temporarily taken off their medications, the majority concurred. Exceptions were for systemic steroids, which must be tapered. And a significant minority of the dermatologists said that they would continue apremilast or dupilumab (24% and 16%, respectively) in the event of a confirmed COVID-19 infection.
In an interview, Dr. Cordoro commented that, even in normal times, most systemic or biological immunosuppressive treatments are used off-label by pediatric dermatologists. “There’s no way this could have been an evidence-based document, as we didn’t have the data to drive this. Many of the medications have been tested in children but not necessarily for dermatologic indications; some are chemotherapy agents or drugs used in rheumatologic diseases.”
The COVID-19 pandemic complicated an already difficult decision-making process, she said.
The researchers cautioned against attempting to make decisions about medications based on data on other infections from clinical trials. “Infection data from standard infections that were identified and watched for in clinical trials really still has no bearing on COVID-19 because it’s such a different virus,” Dr. Cordoro said.
And while some immunosuppressive medications could potentially attenuate a SARS-CoV-2–induced cytokine storm, “we certainly don’t assume this is necessarily going to help.”
The authors advised that physicians anxious about initiating an immunosuppressive treatment should take into consideration whether early intervention could “prevent permanent physical impairment or disfigurement” in diseases such as erythrodermic pustular psoriasis or rapidly progressive linear morphea.
Other diseases, such as atopic dermatitis, “may be acceptably, though not optimally, managed with topical and other home-based therapeutic options” during the pandemic, they wrote.
Dr. Cordoro commented that, given how fast new findings are emerging from the pandemic, the guidance on medications could change. “We will know so much more 3 months from now,” she said. And while there are no formal plans to reissue the survey, “we’re maintaining communication and will have some kind of follow up” with the academic dermatologists.
“If we recognize any signals that are counter to what we say in this work we will immediately let people know,” she said.
The researchers received no outside funding for their study. Of the study’s 24 coauthors, nine disclosed financial relationships with industry.
SOURCE: Add the first auSOURCE: Reynolds et al. Pediatr Dermatol. 2020. doi: 10.1111/pde.14202.
designed to offer guidance to specialists and nonspecialists faced with tough choices about risks.
Some 87% reported that they were reducing the frequency of lab monitoring for some medications, while more than half said they had reached out to patients and their families to discuss the implications of continuing or stopping a drug.
Virtually all – 97% – said that the COVID-19 crisis had affected their decision to initiate immunosuppressive medications, with 84% saying the decision depended on a patient’s risk factors for contracting COVID-19 infection, and also the potential consequences of infection while treated, compared with the risks of not optimally treating the skin condition.
To develop a consensus-based guidance for clinicians, published online April 22 in Pediatric Dermatology, Kelly Cordoro, MD, professor of dermatology at the University of California, San Francisco, assembled a task force of pediatric dermatologists at academic institutions (the Pediatric Dermatology COVID-19 Response Task Force). Together with Sean Reynolds, MD, a pediatric dermatology fellow at UCSF and colleagues, they issued a survey to the 37 members of the task force with questions on how the pandemic has affected their prescribing decisions and certain therapies specifically. All the recipients responded.
The dermatologists were asked about conventional systemic and biologic medications. Most felt confident in continuing biologics, with 78% saying they would keep patients with no signs of COVID-19 exposure or infection on tumor necrosis factor (TNF) inhibitors. More than 90% of respondents said they would continue patients on dupilumab, as well as anti–interleukin (IL)–17, anti–IL-12/23, and anti–IL-23 therapies.
Responses varied more on approaches to the nonbiologic treatments. Fewer than half (46%) said they would continue patients without apparent COVID-19 exposure on systemic steroids, with another 46% saying it depended on the clinical context.
For other systemic therapies, respondents were more likely to want to continue their patients with no signs or symptoms of COVID-19 on methotrexate and apremilast (78% and 83%, respectively) than others (mycophenolate mofetil, azathioprine, cyclosporine, and JAK inhibitors), which saw between 50% and 60% support in the survey.
Patients on any immunosuppressive medications with likely exposure to COVID-19 or who test positive for the virus should be temporarily taken off their medications, the majority concurred. Exceptions were for systemic steroids, which must be tapered. And a significant minority of the dermatologists said that they would continue apremilast or dupilumab (24% and 16%, respectively) in the event of a confirmed COVID-19 infection.
In an interview, Dr. Cordoro commented that, even in normal times, most systemic or biological immunosuppressive treatments are used off-label by pediatric dermatologists. “There’s no way this could have been an evidence-based document, as we didn’t have the data to drive this. Many of the medications have been tested in children but not necessarily for dermatologic indications; some are chemotherapy agents or drugs used in rheumatologic diseases.”
The COVID-19 pandemic complicated an already difficult decision-making process, she said.
The researchers cautioned against attempting to make decisions about medications based on data on other infections from clinical trials. “Infection data from standard infections that were identified and watched for in clinical trials really still has no bearing on COVID-19 because it’s such a different virus,” Dr. Cordoro said.
And while some immunosuppressive medications could potentially attenuate a SARS-CoV-2–induced cytokine storm, “we certainly don’t assume this is necessarily going to help.”
The authors advised that physicians anxious about initiating an immunosuppressive treatment should take into consideration whether early intervention could “prevent permanent physical impairment or disfigurement” in diseases such as erythrodermic pustular psoriasis or rapidly progressive linear morphea.
Other diseases, such as atopic dermatitis, “may be acceptably, though not optimally, managed with topical and other home-based therapeutic options” during the pandemic, they wrote.
Dr. Cordoro commented that, given how fast new findings are emerging from the pandemic, the guidance on medications could change. “We will know so much more 3 months from now,” she said. And while there are no formal plans to reissue the survey, “we’re maintaining communication and will have some kind of follow up” with the academic dermatologists.
“If we recognize any signals that are counter to what we say in this work we will immediately let people know,” she said.
The researchers received no outside funding for their study. Of the study’s 24 coauthors, nine disclosed financial relationships with industry.
SOURCE: Add the first auSOURCE: Reynolds et al. Pediatr Dermatol. 2020. doi: 10.1111/pde.14202.
designed to offer guidance to specialists and nonspecialists faced with tough choices about risks.
Some 87% reported that they were reducing the frequency of lab monitoring for some medications, while more than half said they had reached out to patients and their families to discuss the implications of continuing or stopping a drug.
Virtually all – 97% – said that the COVID-19 crisis had affected their decision to initiate immunosuppressive medications, with 84% saying the decision depended on a patient’s risk factors for contracting COVID-19 infection, and also the potential consequences of infection while treated, compared with the risks of not optimally treating the skin condition.
To develop a consensus-based guidance for clinicians, published online April 22 in Pediatric Dermatology, Kelly Cordoro, MD, professor of dermatology at the University of California, San Francisco, assembled a task force of pediatric dermatologists at academic institutions (the Pediatric Dermatology COVID-19 Response Task Force). Together with Sean Reynolds, MD, a pediatric dermatology fellow at UCSF and colleagues, they issued a survey to the 37 members of the task force with questions on how the pandemic has affected their prescribing decisions and certain therapies specifically. All the recipients responded.
The dermatologists were asked about conventional systemic and biologic medications. Most felt confident in continuing biologics, with 78% saying they would keep patients with no signs of COVID-19 exposure or infection on tumor necrosis factor (TNF) inhibitors. More than 90% of respondents said they would continue patients on dupilumab, as well as anti–interleukin (IL)–17, anti–IL-12/23, and anti–IL-23 therapies.
Responses varied more on approaches to the nonbiologic treatments. Fewer than half (46%) said they would continue patients without apparent COVID-19 exposure on systemic steroids, with another 46% saying it depended on the clinical context.
For other systemic therapies, respondents were more likely to want to continue their patients with no signs or symptoms of COVID-19 on methotrexate and apremilast (78% and 83%, respectively) than others (mycophenolate mofetil, azathioprine, cyclosporine, and JAK inhibitors), which saw between 50% and 60% support in the survey.
Patients on any immunosuppressive medications with likely exposure to COVID-19 or who test positive for the virus should be temporarily taken off their medications, the majority concurred. Exceptions were for systemic steroids, which must be tapered. And a significant minority of the dermatologists said that they would continue apremilast or dupilumab (24% and 16%, respectively) in the event of a confirmed COVID-19 infection.
In an interview, Dr. Cordoro commented that, even in normal times, most systemic or biological immunosuppressive treatments are used off-label by pediatric dermatologists. “There’s no way this could have been an evidence-based document, as we didn’t have the data to drive this. Many of the medications have been tested in children but not necessarily for dermatologic indications; some are chemotherapy agents or drugs used in rheumatologic diseases.”
The COVID-19 pandemic complicated an already difficult decision-making process, she said.
The researchers cautioned against attempting to make decisions about medications based on data on other infections from clinical trials. “Infection data from standard infections that were identified and watched for in clinical trials really still has no bearing on COVID-19 because it’s such a different virus,” Dr. Cordoro said.
And while some immunosuppressive medications could potentially attenuate a SARS-CoV-2–induced cytokine storm, “we certainly don’t assume this is necessarily going to help.”
The authors advised that physicians anxious about initiating an immunosuppressive treatment should take into consideration whether early intervention could “prevent permanent physical impairment or disfigurement” in diseases such as erythrodermic pustular psoriasis or rapidly progressive linear morphea.
Other diseases, such as atopic dermatitis, “may be acceptably, though not optimally, managed with topical and other home-based therapeutic options” during the pandemic, they wrote.
Dr. Cordoro commented that, given how fast new findings are emerging from the pandemic, the guidance on medications could change. “We will know so much more 3 months from now,” she said. And while there are no formal plans to reissue the survey, “we’re maintaining communication and will have some kind of follow up” with the academic dermatologists.
“If we recognize any signals that are counter to what we say in this work we will immediately let people know,” she said.
The researchers received no outside funding for their study. Of the study’s 24 coauthors, nine disclosed financial relationships with industry.
SOURCE: Add the first auSOURCE: Reynolds et al. Pediatr Dermatol. 2020. doi: 10.1111/pde.14202.
FROM PEDIATRIC DERMATOLOGY
Hyperkalemia most common adverse event in women taking spironolactone
, according to new research.
Spironolactone, which is approved to treat heart failure, hypertension, edema, and primary hyperaldosteronism, has antagonistic effects on progesterone and androgen receptors and has been used as an off-label treatment for acne in women. “Numerous guidelines have recommended its off-label use for acne therapy to avoid antibiotic resistance and potential side effects,” wrote Yu Wang of Stony Brook (N.Y.) University and Shari R. Lipner MD, PhD, of Weill Cornell Medicine, New York. Their report is in the International Journal of Women’s Dermatology.
In a retrospective study, the investigators analyzed 7,920 adverse events with spironolactone reported by women of all ages between Jan. 1, 1969, and Dec. 30, 2018, to the Food and Drug Administration’s Adverse Event Reporting System database, for all indications. The most common adverse event was hyperkalemia, reported in 16.1%, followed by kidney injury (15.2%) and drug interactions (9%). Of the 1,272 cases of hyperkalemia reported, 25 occurred in women aged 45 years or younger; 59.3% occurred in women aged 65-85 years.
While spironolactone prescribing information was not available, the investigators compared yearly reports of adverse events with annual public interest in spironolactone using the Google Trends search term spironolactone and annual scholarly mentions of spironolactone in the Altmetric database. There was a strong correlation between the number of cases reported to the FDA and the Google Trends search (Spearman coefficient, 0.94; P less than .001) and to the Altmetric database (Spearman coefficient, 0.64; P less than .01).
Noting that hyperkalemia is “exceptionally uncommon” in women aged 45 years and younger, the investigators concluded that “in the absence of risk factors for hyperkalemia or reduced renal function, potassium laboratory monitoring is unnecessary in younger females taking spironolactone.” Because the incidence increases with age, “interval laboratory monitoring is recommended for females older than 45 years old,” they noted.
Limitations of the study, they noted, include the retrospective design and no available data before 1969. “In addition, since the [FDA Adverse Event Reporting System] data does not differentiate whether spironolactone was prescribed for heart failure, hypertension, edema, primary hyperaldosteronism, or for acne,” the study could not control for these or other confounding comorbidities or associated therapies.
“For future studies, it is important to analyze drug interactions more carefully to determine which other medications may potentiate the risk for hyperkalemia in patients taking spironolactone. It is also important to quantitate overall U.S. prescription data to better understand the relative frequency of these adverse effects reported to the FDA,” they wrote.
The investigators reported that they had no conflicts of interest; the study had no funding.
SOURCE: Wang Y, Lipner SR. Int J Womens Dermatol. 2020 May 18. doi: 10.1016/j.ijwd.2020.05.002.
, according to new research.
Spironolactone, which is approved to treat heart failure, hypertension, edema, and primary hyperaldosteronism, has antagonistic effects on progesterone and androgen receptors and has been used as an off-label treatment for acne in women. “Numerous guidelines have recommended its off-label use for acne therapy to avoid antibiotic resistance and potential side effects,” wrote Yu Wang of Stony Brook (N.Y.) University and Shari R. Lipner MD, PhD, of Weill Cornell Medicine, New York. Their report is in the International Journal of Women’s Dermatology.
In a retrospective study, the investigators analyzed 7,920 adverse events with spironolactone reported by women of all ages between Jan. 1, 1969, and Dec. 30, 2018, to the Food and Drug Administration’s Adverse Event Reporting System database, for all indications. The most common adverse event was hyperkalemia, reported in 16.1%, followed by kidney injury (15.2%) and drug interactions (9%). Of the 1,272 cases of hyperkalemia reported, 25 occurred in women aged 45 years or younger; 59.3% occurred in women aged 65-85 years.
While spironolactone prescribing information was not available, the investigators compared yearly reports of adverse events with annual public interest in spironolactone using the Google Trends search term spironolactone and annual scholarly mentions of spironolactone in the Altmetric database. There was a strong correlation between the number of cases reported to the FDA and the Google Trends search (Spearman coefficient, 0.94; P less than .001) and to the Altmetric database (Spearman coefficient, 0.64; P less than .01).
Noting that hyperkalemia is “exceptionally uncommon” in women aged 45 years and younger, the investigators concluded that “in the absence of risk factors for hyperkalemia or reduced renal function, potassium laboratory monitoring is unnecessary in younger females taking spironolactone.” Because the incidence increases with age, “interval laboratory monitoring is recommended for females older than 45 years old,” they noted.
Limitations of the study, they noted, include the retrospective design and no available data before 1969. “In addition, since the [FDA Adverse Event Reporting System] data does not differentiate whether spironolactone was prescribed for heart failure, hypertension, edema, primary hyperaldosteronism, or for acne,” the study could not control for these or other confounding comorbidities or associated therapies.
“For future studies, it is important to analyze drug interactions more carefully to determine which other medications may potentiate the risk for hyperkalemia in patients taking spironolactone. It is also important to quantitate overall U.S. prescription data to better understand the relative frequency of these adverse effects reported to the FDA,” they wrote.
The investigators reported that they had no conflicts of interest; the study had no funding.
SOURCE: Wang Y, Lipner SR. Int J Womens Dermatol. 2020 May 18. doi: 10.1016/j.ijwd.2020.05.002.
, according to new research.
Spironolactone, which is approved to treat heart failure, hypertension, edema, and primary hyperaldosteronism, has antagonistic effects on progesterone and androgen receptors and has been used as an off-label treatment for acne in women. “Numerous guidelines have recommended its off-label use for acne therapy to avoid antibiotic resistance and potential side effects,” wrote Yu Wang of Stony Brook (N.Y.) University and Shari R. Lipner MD, PhD, of Weill Cornell Medicine, New York. Their report is in the International Journal of Women’s Dermatology.
In a retrospective study, the investigators analyzed 7,920 adverse events with spironolactone reported by women of all ages between Jan. 1, 1969, and Dec. 30, 2018, to the Food and Drug Administration’s Adverse Event Reporting System database, for all indications. The most common adverse event was hyperkalemia, reported in 16.1%, followed by kidney injury (15.2%) and drug interactions (9%). Of the 1,272 cases of hyperkalemia reported, 25 occurred in women aged 45 years or younger; 59.3% occurred in women aged 65-85 years.
While spironolactone prescribing information was not available, the investigators compared yearly reports of adverse events with annual public interest in spironolactone using the Google Trends search term spironolactone and annual scholarly mentions of spironolactone in the Altmetric database. There was a strong correlation between the number of cases reported to the FDA and the Google Trends search (Spearman coefficient, 0.94; P less than .001) and to the Altmetric database (Spearman coefficient, 0.64; P less than .01).
Noting that hyperkalemia is “exceptionally uncommon” in women aged 45 years and younger, the investigators concluded that “in the absence of risk factors for hyperkalemia or reduced renal function, potassium laboratory monitoring is unnecessary in younger females taking spironolactone.” Because the incidence increases with age, “interval laboratory monitoring is recommended for females older than 45 years old,” they noted.
Limitations of the study, they noted, include the retrospective design and no available data before 1969. “In addition, since the [FDA Adverse Event Reporting System] data does not differentiate whether spironolactone was prescribed for heart failure, hypertension, edema, primary hyperaldosteronism, or for acne,” the study could not control for these or other confounding comorbidities or associated therapies.
“For future studies, it is important to analyze drug interactions more carefully to determine which other medications may potentiate the risk for hyperkalemia in patients taking spironolactone. It is also important to quantitate overall U.S. prescription data to better understand the relative frequency of these adverse effects reported to the FDA,” they wrote.
The investigators reported that they had no conflicts of interest; the study had no funding.
SOURCE: Wang Y, Lipner SR. Int J Womens Dermatol. 2020 May 18. doi: 10.1016/j.ijwd.2020.05.002.
FROM THE INTERNATIONAL JOURNAL OF WOMEN’S DERMATOLOGY
Biologic approved for atopic dermatitis in children
The Food and Drug Administration has approved dupilumab for children aged 6-11 years with moderate to severe atopic dermatitis, the manufacturers announced.
The new indication is for children “whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable,” Regeneron and Sanofi said in a press release, which points out that this is the first biologic approved for AD in this age group.
For children aged 6-11, the two available dupilumab (Dupixent) doses in prefilled syringes are given based on weight – 300 mg every 4 weeks for children between 15 to 29 kg and 200 mg every 2 weeks for children 30 to 59 kg – following an initial loading dose.
In phase 3 trials, children with severe AD who received dupilumab and topical corticosteroids improved significantly in overall disease severity, skin clearance, and itch, compared with those getting steroids alone. Eczema Area and Severity Index-75, for example, was reached by 75% of patients on either dupilumab dose, compared with 28% and 26% , respectively, for those receiving steroids alone every 4 and every 2 weeks, the statement said.
Over the 16-week treatment period, overall rates of adverse events were 65% for those getting dupilumab every 4 weeks and 61% for every 2 weeks – compared with steroids alone (72% and 75%, respectively), the statement said.
The fully human monoclonal antibody inhibits signaling of the interleukin-4 and interleukin-13 proteins and is already approved as an add-on maintenance treatment in children aged 12 years and older with moderate to severe asthma (eosinophilic phenotype or oral-corticosteroid dependent) and in adults with inadequately controlled chronic rhinosinusitis with nasal polyposis, according to the prescribing information.
The Food and Drug Administration has approved dupilumab for children aged 6-11 years with moderate to severe atopic dermatitis, the manufacturers announced.
The new indication is for children “whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable,” Regeneron and Sanofi said in a press release, which points out that this is the first biologic approved for AD in this age group.
For children aged 6-11, the two available dupilumab (Dupixent) doses in prefilled syringes are given based on weight – 300 mg every 4 weeks for children between 15 to 29 kg and 200 mg every 2 weeks for children 30 to 59 kg – following an initial loading dose.
In phase 3 trials, children with severe AD who received dupilumab and topical corticosteroids improved significantly in overall disease severity, skin clearance, and itch, compared with those getting steroids alone. Eczema Area and Severity Index-75, for example, was reached by 75% of patients on either dupilumab dose, compared with 28% and 26% , respectively, for those receiving steroids alone every 4 and every 2 weeks, the statement said.
Over the 16-week treatment period, overall rates of adverse events were 65% for those getting dupilumab every 4 weeks and 61% for every 2 weeks – compared with steroids alone (72% and 75%, respectively), the statement said.
The fully human monoclonal antibody inhibits signaling of the interleukin-4 and interleukin-13 proteins and is already approved as an add-on maintenance treatment in children aged 12 years and older with moderate to severe asthma (eosinophilic phenotype or oral-corticosteroid dependent) and in adults with inadequately controlled chronic rhinosinusitis with nasal polyposis, according to the prescribing information.
The Food and Drug Administration has approved dupilumab for children aged 6-11 years with moderate to severe atopic dermatitis, the manufacturers announced.
The new indication is for children “whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable,” Regeneron and Sanofi said in a press release, which points out that this is the first biologic approved for AD in this age group.
For children aged 6-11, the two available dupilumab (Dupixent) doses in prefilled syringes are given based on weight – 300 mg every 4 weeks for children between 15 to 29 kg and 200 mg every 2 weeks for children 30 to 59 kg – following an initial loading dose.
In phase 3 trials, children with severe AD who received dupilumab and topical corticosteroids improved significantly in overall disease severity, skin clearance, and itch, compared with those getting steroids alone. Eczema Area and Severity Index-75, for example, was reached by 75% of patients on either dupilumab dose, compared with 28% and 26% , respectively, for those receiving steroids alone every 4 and every 2 weeks, the statement said.
Over the 16-week treatment period, overall rates of adverse events were 65% for those getting dupilumab every 4 weeks and 61% for every 2 weeks – compared with steroids alone (72% and 75%, respectively), the statement said.
The fully human monoclonal antibody inhibits signaling of the interleukin-4 and interleukin-13 proteins and is already approved as an add-on maintenance treatment in children aged 12 years and older with moderate to severe asthma (eosinophilic phenotype or oral-corticosteroid dependent) and in adults with inadequately controlled chronic rhinosinusitis with nasal polyposis, according to the prescribing information.