User login
Increased hypothyroidism risk seen in young men with HS
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
Anna Figueiredo, MD, declared at the virtual annual meeting of the American Academy of Dermatology.
The surprise about this finding from a large retrospective case-control study stems from the fact that the elevated risk for hypothyroidism didn’t also extend to younger women with hidradenitis suppurativa (HS) nor to patients older than 40 years of either gender, explained Dr. Figueiredo of the department of dermatology at Northwestern University, Chicago.
She presented a retrospective case-control study based on information extracted from a medical records database of more than 8 million Midwestern adults. Among nearly 141,000 dermatology patients with follow-up in the database for at least 1 year, there were 405 HS patients aged 18-40 years and 327 aged 41-89.
In an age-matched comparison with the dermatology patients without HS, the younger HS cohort was at a significant 1.52-fold increased risk for comorbid hypothyroidism. Upon further stratification by sex, only the younger men with HS were at increased risk. Those patients were at 3.95-fold greater risk for having a diagnosis of hypothyroidism than were age-matched younger male dermatology patients.
Both younger and older HS patients were at numerically increased risk for being diagnosed with hyperthyroidism; however, this difference didn’t approach statistical significance because there were so few cases: a total of just eight in the HS population across the full age spectrum.
Hidradenitis suppurativa is a chronic inflammatory disease with an estimated prevalence of up to 4% in the United States. Growing evidence suggests it is an immune-mediated disorder because the tumor necrosis factor inhibitor adalimumab (Humira) has been approved for treatment of HS.
Thyroid disease is also often autoimmune-mediated, but its relationship with HS hasn’t been extensively examined. A recent meta-analysis of five case-control studies concluded that HS was associated with a 1.36-fold increased risk of thyroid disease; however, the Nepalese investigators didn’t distinguish between hypo- and hyperthyroidism (J Am Acad Dermatol. 2020 Feb;82[2]:491-3).
Dr. Figueiredo reported having no financial conflicts regarding her study, which was without commercial support.
FROM AAD 20
More phase 3 data reported for abrocitinib for atopic dermatitis
Melinda Gooderham, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The positive results of this 391-patient, international, randomized, double-blind, placebo-controlled clinical trial mirror those previously reported in the identically designed JADE-MONO-1 pivotal phase 3 trial, noted Dr. Gooderham, medical director of the SKiN Centre for Dermatology and a dermatologist at Queen’s University in Kingston, Ont.
Participants in JADE-MONO-2 were randomized 2:2:1 to abrocitinib at 200 mg once daily, 100 mg once daily, or placebo for 12 weeks. The coprimary endpoint of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with an improvement of at least two grades at week 12 was achieved in 38.1% and 28.4% of patients on 200 and 100 mg of the JAK-1 inhibitor, respectively, compared with 9.1% of placebo-treated controls. The other coprimary endpoint – significant improvement in disease extent as defined by at least a 75% reduction from baseline in Eczema Area and Severity Index (EASI-75 response) at 12 weeks – was reached in 61% of patients on abrocitinib at 200 mg/day, 44.5% on 100 mg/day, and 10.4% of controls.
A key secondary endpoint was improvement in itch based on at least a 4-point improvement at week 12 on the Peak Pruritus Numerical Rating Scale from a mean baseline score of 7. This outcome was reached by 55.3% of patients on abrocitinib at 200 mg, 45.2% on 100 mg, and 11.5% on placebo. Of note, the reduction in itch was impressively fast, with significant separation from placebo occurring within the first 24 hours of the study, after just a single dose of abrocitinib. By week 2, roughly one-third of patients on high-dose and one-quarter of those on low-dose abrocitinib had already reached the itch endpoint, the dermatologist continued.
The improvement in pruritus scores in abrocitinib-treated patients was accompanied by significantly greater gains on validated measures of quality of life, another secondary endpoint. The EASI-90 response rate, yet another key secondary outcome, was 37.7% with abrocitinib at 200 mg, 23.9% with 100 mg, and 3.9% with placebo.
The safety profile of abrocitinib was essentially the same as for placebo with the exception of a 3.2% incidence of thrombocytopenia in patients on abrocitinib at 200 mg/day; no cases occurred in controls or patients on abrocitinib at 100 mg/day. Although venous thromboembolism has arisen as a potential concern in clinical trials of oral JAK inhibitors for rheumatoid arthritis, there were no cases in JADE-MONO-2. A long-term safety extension study in JADE-MONO participants is underway.
In an interview, Dr. Gooderham said that, based on the phase 2 study data that’s available for upadacitinib, another JAK-1-selective oral agent, abrocitinib and upadacitinib appear to be in the same ballpark with respect to efficacy as defined by IGA response, EASI improvement, and itch relief.
“The JAK-1 selectivity does seem to offer some advantage in levels of response over more broad JAK inhibition, such as with baritinib,” she added.
Asked how she foresees abrocitinib fitting into clinical practice, should it win regulatory approval for treatment of atopic dermatitis, Dr. Gooderham said it might be considered on a par with the injectable interleukin-4 and -13 inhibitor dupilumab (Dupixent) as next-line therapy after failure on topical therapy or as an option in patients who haven’t responded to or could not tolerate dupilumab. Abrocitinib will be an attractive option for patients who prefer oral therapy and will be an especially appealing medication in patients with a strong itch component to their atopic dermatitis, she added.
The results of JADE COMPARE, a phase 3, head-to-head randomized comparison of abrocitinib and dupilumab, are expected to be presented later this year at the virtual annual congress of the European Academy of Dermatology and Venereology. Pfizer has announced the key results, reporting that the JAK inhibitor at 200 mg/day achieved significantly greater improvements than dupilumab in the coprimary IGA and EASI-75 endpoints at 12 weeks.
JADE-MONO-2 was sponsored by Pfizer. Dr. Gooderham reported receiving research grants from that company and close to two dozen others.
The JADE-MONO-2 results have been published online (JAMA Dermatol. 2020 Jun 3;e201406. doi: 10.1001/jamadermatol.2020.1406).
Melinda Gooderham, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The positive results of this 391-patient, international, randomized, double-blind, placebo-controlled clinical trial mirror those previously reported in the identically designed JADE-MONO-1 pivotal phase 3 trial, noted Dr. Gooderham, medical director of the SKiN Centre for Dermatology and a dermatologist at Queen’s University in Kingston, Ont.
Participants in JADE-MONO-2 were randomized 2:2:1 to abrocitinib at 200 mg once daily, 100 mg once daily, or placebo for 12 weeks. The coprimary endpoint of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with an improvement of at least two grades at week 12 was achieved in 38.1% and 28.4% of patients on 200 and 100 mg of the JAK-1 inhibitor, respectively, compared with 9.1% of placebo-treated controls. The other coprimary endpoint – significant improvement in disease extent as defined by at least a 75% reduction from baseline in Eczema Area and Severity Index (EASI-75 response) at 12 weeks – was reached in 61% of patients on abrocitinib at 200 mg/day, 44.5% on 100 mg/day, and 10.4% of controls.
A key secondary endpoint was improvement in itch based on at least a 4-point improvement at week 12 on the Peak Pruritus Numerical Rating Scale from a mean baseline score of 7. This outcome was reached by 55.3% of patients on abrocitinib at 200 mg, 45.2% on 100 mg, and 11.5% on placebo. Of note, the reduction in itch was impressively fast, with significant separation from placebo occurring within the first 24 hours of the study, after just a single dose of abrocitinib. By week 2, roughly one-third of patients on high-dose and one-quarter of those on low-dose abrocitinib had already reached the itch endpoint, the dermatologist continued.
The improvement in pruritus scores in abrocitinib-treated patients was accompanied by significantly greater gains on validated measures of quality of life, another secondary endpoint. The EASI-90 response rate, yet another key secondary outcome, was 37.7% with abrocitinib at 200 mg, 23.9% with 100 mg, and 3.9% with placebo.
The safety profile of abrocitinib was essentially the same as for placebo with the exception of a 3.2% incidence of thrombocytopenia in patients on abrocitinib at 200 mg/day; no cases occurred in controls or patients on abrocitinib at 100 mg/day. Although venous thromboembolism has arisen as a potential concern in clinical trials of oral JAK inhibitors for rheumatoid arthritis, there were no cases in JADE-MONO-2. A long-term safety extension study in JADE-MONO participants is underway.
In an interview, Dr. Gooderham said that, based on the phase 2 study data that’s available for upadacitinib, another JAK-1-selective oral agent, abrocitinib and upadacitinib appear to be in the same ballpark with respect to efficacy as defined by IGA response, EASI improvement, and itch relief.
“The JAK-1 selectivity does seem to offer some advantage in levels of response over more broad JAK inhibition, such as with baritinib,” she added.
Asked how she foresees abrocitinib fitting into clinical practice, should it win regulatory approval for treatment of atopic dermatitis, Dr. Gooderham said it might be considered on a par with the injectable interleukin-4 and -13 inhibitor dupilumab (Dupixent) as next-line therapy after failure on topical therapy or as an option in patients who haven’t responded to or could not tolerate dupilumab. Abrocitinib will be an attractive option for patients who prefer oral therapy and will be an especially appealing medication in patients with a strong itch component to their atopic dermatitis, she added.
The results of JADE COMPARE, a phase 3, head-to-head randomized comparison of abrocitinib and dupilumab, are expected to be presented later this year at the virtual annual congress of the European Academy of Dermatology and Venereology. Pfizer has announced the key results, reporting that the JAK inhibitor at 200 mg/day achieved significantly greater improvements than dupilumab in the coprimary IGA and EASI-75 endpoints at 12 weeks.
JADE-MONO-2 was sponsored by Pfizer. Dr. Gooderham reported receiving research grants from that company and close to two dozen others.
The JADE-MONO-2 results have been published online (JAMA Dermatol. 2020 Jun 3;e201406. doi: 10.1001/jamadermatol.2020.1406).
Melinda Gooderham, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The positive results of this 391-patient, international, randomized, double-blind, placebo-controlled clinical trial mirror those previously reported in the identically designed JADE-MONO-1 pivotal phase 3 trial, noted Dr. Gooderham, medical director of the SKiN Centre for Dermatology and a dermatologist at Queen’s University in Kingston, Ont.
Participants in JADE-MONO-2 were randomized 2:2:1 to abrocitinib at 200 mg once daily, 100 mg once daily, or placebo for 12 weeks. The coprimary endpoint of skin clearance as reflected in an Investigator’s Global Assessment (IGA) score of 0 or 1 (clear or almost clear) with an improvement of at least two grades at week 12 was achieved in 38.1% and 28.4% of patients on 200 and 100 mg of the JAK-1 inhibitor, respectively, compared with 9.1% of placebo-treated controls. The other coprimary endpoint – significant improvement in disease extent as defined by at least a 75% reduction from baseline in Eczema Area and Severity Index (EASI-75 response) at 12 weeks – was reached in 61% of patients on abrocitinib at 200 mg/day, 44.5% on 100 mg/day, and 10.4% of controls.
A key secondary endpoint was improvement in itch based on at least a 4-point improvement at week 12 on the Peak Pruritus Numerical Rating Scale from a mean baseline score of 7. This outcome was reached by 55.3% of patients on abrocitinib at 200 mg, 45.2% on 100 mg, and 11.5% on placebo. Of note, the reduction in itch was impressively fast, with significant separation from placebo occurring within the first 24 hours of the study, after just a single dose of abrocitinib. By week 2, roughly one-third of patients on high-dose and one-quarter of those on low-dose abrocitinib had already reached the itch endpoint, the dermatologist continued.
The improvement in pruritus scores in abrocitinib-treated patients was accompanied by significantly greater gains on validated measures of quality of life, another secondary endpoint. The EASI-90 response rate, yet another key secondary outcome, was 37.7% with abrocitinib at 200 mg, 23.9% with 100 mg, and 3.9% with placebo.
The safety profile of abrocitinib was essentially the same as for placebo with the exception of a 3.2% incidence of thrombocytopenia in patients on abrocitinib at 200 mg/day; no cases occurred in controls or patients on abrocitinib at 100 mg/day. Although venous thromboembolism has arisen as a potential concern in clinical trials of oral JAK inhibitors for rheumatoid arthritis, there were no cases in JADE-MONO-2. A long-term safety extension study in JADE-MONO participants is underway.
In an interview, Dr. Gooderham said that, based on the phase 2 study data that’s available for upadacitinib, another JAK-1-selective oral agent, abrocitinib and upadacitinib appear to be in the same ballpark with respect to efficacy as defined by IGA response, EASI improvement, and itch relief.
“The JAK-1 selectivity does seem to offer some advantage in levels of response over more broad JAK inhibition, such as with baritinib,” she added.
Asked how she foresees abrocitinib fitting into clinical practice, should it win regulatory approval for treatment of atopic dermatitis, Dr. Gooderham said it might be considered on a par with the injectable interleukin-4 and -13 inhibitor dupilumab (Dupixent) as next-line therapy after failure on topical therapy or as an option in patients who haven’t responded to or could not tolerate dupilumab. Abrocitinib will be an attractive option for patients who prefer oral therapy and will be an especially appealing medication in patients with a strong itch component to their atopic dermatitis, she added.
The results of JADE COMPARE, a phase 3, head-to-head randomized comparison of abrocitinib and dupilumab, are expected to be presented later this year at the virtual annual congress of the European Academy of Dermatology and Venereology. Pfizer has announced the key results, reporting that the JAK inhibitor at 200 mg/day achieved significantly greater improvements than dupilumab in the coprimary IGA and EASI-75 endpoints at 12 weeks.
JADE-MONO-2 was sponsored by Pfizer. Dr. Gooderham reported receiving research grants from that company and close to two dozen others.
The JADE-MONO-2 results have been published online (JAMA Dermatol. 2020 Jun 3;e201406. doi: 10.1001/jamadermatol.2020.1406).
FROM AAD 20
ID dermatology: Advancements, but new challenges, over 50 years
When Stephen Tyring, MD, PhD, an infectious disease dermatologist, started his career in the early 1980s, he said “we were diagnosing Kaposi’s sarcoma right and left. We would see a new case every day or two.”
It was the early days of the HIV/AIDS epidemic, and dermatologists were at the forefront because HIV/AIDS often presented with skin manifestations. Dr. Tyring, clinical professor in the departments of dermatology, microbiology & molecular genetics and internal medicine at the University of Texas Health Science Center, Houston, and his colleagues referred Kaposi’s patients for chemotherapy and radiation, but the outlook was often grim, especially if lesions developed in the lungs.
Dermatologist don’t see much Kaposi’s anymore because of highly effective treatments for HIV.
Members of the original editorial advisory board saw it coming. In a feature in which board members provided their prediction for the 1970s that appeared in the first issue, New York dermatologist Norman Orentreich, MD, counted the “probable introduction of virucidal agents” as one of the “significant advances or changes that I foresee in the next 10 years.” J. Lamar Callaway, MD, professor of dermatology at Duke University, Durham, N.C., predicted that “the next 10 years should develop effective anti-viral agents for warts, herpes simplex, and herpes zoster.”
To celebrate the 50th anniversary of Dermatology News, we are looking back at how the field has changed since that first issue. The focus this month is infectious disease. There’s a lot to be grateful for but there are also challenges like antibiotic resistance that weren’t on the radar screens of Dr. Orentreich, Dr. Callaway, and their peers in 1970.
All in all, “the only thing I wish we did the old way is sit at the bedside and talk to patients more. We rely so much on technology now that we sometimes lose the art of medicine, which is comforting to the patient,” said Theodore Rosen, MD, an ID dermatologist and professor of dermatology at Baylor College of Medicine, Houston, who’s been in practice for 42 years.
“A lot of advancements against herpes viruses”
One of the biggest wins for ID dermatology over the last 5 decades has been the management of herpes, both herpes simplex virus 1 and 2, as well as herpes zoster virus. It started with the approval of acyclovir in 1981. Before then, “we had no direct therapy for genital herpes, herpes zoster, or disseminated herpes in immunosuppressed or cancer patients,” Dr. Rosen said.
“I can remember doing an interview with Good Morning America when I gave the first IV dose of acyclovir in the city of Houston for really bad disseminated herpes” in an HIV patient, he said, and it worked.
Two derivatives, valacyclovir and famciclovir, became available in the mid-1990s, so today “we have three drugs and some others at the periphery that are all highly effective not only” against herpes, but also for preventing outbreaks; valacyclovir can even prevent asymptomatic shedding, therefore possibly preventing new infections. “That’s a concept we didn’t even have 40 years ago,” Dr. Rosen said.
Cidofovir has also made a difference. The IV formulation was approved for AIDS-associated cytomegalovirus retinitis in 1996 but discontinued a few years later amid concerns of severe renal toxicity. It’s found a new home in dermatology since then, explained ID dermatologist Carrie Kovarik, MD, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
Dermatologists see acyclovir-resistant herpes “heaped up on the genitals in HIV patients,” and there weren’t many options in the past. A few years ago, “we [tried] injecting cidofovir directly into the skin lesions, and it’s been remarkably successful. It is a good way to treat these lesions” if dermatologists can get it compounded, she said.
Shingles vaccines, first the live attenuated zoster vaccine (Zostavax) approved by the Food and Drug Administration in 2006 and the more effective recombinant zoster vaccine (Shingrix) approved in 2017, have also had a significant impact.
Dr. Rosen remembers what it was like when he first started practicing over 40 years ago. Not uncommonly, “we saw horrible cases of shingles,” including one in his uncle, who was left with permanent hand pain long after the rash subsided.
Today, “I see much less shingles, and when I do see it, it’s in a much-attenuated form. [Shingrix], even if it doesn’t prevent the disease, often prevents postherpetic neuralgia,” he said.
Also, with pediatric vaccinations against chicken pox, “we’re probably going to see a whole new generation without shingles, which is huge. We’ve made a lot of advancements against herpes viruses,” Dr. Kovarik said.
“We finally found something that helps”
“We’ve [also] come a really long way with genital wart treatment,” Dr. Kovarik said.
It started with approval of topical imiquimod in 1997. “Before that, we were just killing one wart here and one wart there” but they would often come back and pop up in other areas. Injectable interferon was an option at the time, but people didn’t like all the needles.
With imiquimod, “we finally [had] a way to target HPV [human papillomavirus] and not just scrape” or freeze one wart at a time, and “we were able to generate an inflammatory response in the whole area to clear the virus.” Working with HIV patients, “I see sheets and sheets of confluent warts throughout the whole genital area; to try to freeze that is impossible. Now I have a way to get rid of [genital] warts and keep them away even if you have a big cluster,” she said.
“Sometimes, we’ll do both liquid nitrogen and imiquimod. That’s a good way to tackle people who have a high burden of warts,” Dr. Kovarik noted. Other effective treatments have come out as well, including an ointment formulation of sinecatechins, extracted from green tea, “but you have to put it on several times a day, and insurance companies don’t cover it often,” she said.
Intralesional cidofovir is also proving to be boon for potentially malignant refractory warts in HIV and transplant patients. “It’s an incredible treatment. We can inject that antiviral into warts and get rid of them. We finally found something that helps” these people, Dr. Kovarik said.
The HPV vaccine Gardasil is making a difference, as well. In addition to cervical dysplasia and anogenital cancers, it protects against two condyloma strains. Dr. Rosen said he’s seeing fewer cases of genital warts now than when he started practicing, likely because of the vaccine.
“Organisms that weren’t pathogens are now pathogens”
Antibiotic resistance probably tops the list for what’s changed in a bad way in ID dermatology since 1970. Dr. Rosen remembers at the start of his career that “we never worried about antibiotic resistance. We’d put people on antibiotics for acne, rosacea, and we’d keep them on them for 3 years, 6 years”; resistance wasn’t on the radar screen and was not mentioned once in the first issue of Dermatology News, which was packed with articles and ran 24 pages.
The situation is different now. Driven by decades of overuse in agriculture and the medical system, antibiotic resistance is a concern throughout medicine, and unfortunately, “we have not come nearly as far as fast with antibiotics,” at least the ones dermatologists use, “as we have with antivirals,” Dr. Tyring said.
For instance, methicillin-resistant Staphylococcus aureus (MRSA), first described in the United States in 1968, is “no longer the exception to the rule, but the rule” itself, he said, with carbuncles, furuncles, and abscesses not infrequently growing out MRSA. There are also new drug-resistant forms of old problems like gonorrhea and tuberculosis, among other developments, and impetigo has shifted since 1970 from mostly a Streptococcus infection easily treated with penicillin to often a Staphylococcus disease that’s resistant to it. There’s also been a steady march of new pathogens, including the latest one, SARS-CoV-2, the virus that causes COVID-19, which has been recognized as having a variety of skin manifestations.
“No matter how smart we think we are, nature has a way of putting us back in our place,” Dr. Rosen said.
The bright spot is that “we’ve become very adept at identifying and characterizing” microbes “based on techniques we didn’t even have when I started practicing,” such as polymerase chain reaction. “It has taken a lot of guess work out of treating infectious diseases,” he said.
The widespread use of immunosuppressives such as cyclophosphamide, mycophenolate, azathioprine, rituximab, and other agents used in conjunction with solid organ transplantation, has also been a challenge. “We are seeing infections with really odd organisms. Just recently, I had a patient with fusarium in the skin; it’s a fungus that lives in the dirt. I saw a patient with a species of algae” that normally lives in stagnant water, he commented. “We used to get [things like that] back on reports, and we’d throw them away. You can’t do that anymore. Organisms that weren’t pathogens in the past are now pathogens,” particularly in immunosuppressed people, Dr. Rosen said.
Venereologists no more
There’s been another big change in the field. “Back in the not too distant past, dermatologists in the U.S. were referred to as ‘dermatologist-venereologists.’ ” It goes back to the time when syphilis wasn’t diagnosed and treated early, so patients often presented with secondary skin complications and went to dermatologists for help. As a result, “dermatologists became the most experienced at treating it,” Dr. Tyring said.
That’s faded from practice. Part of the reason is that as late as 2000, syphilis seemed to be on the way out; the Centers for Disease and Control and Prevention even raised the possibility of elimination. Dermatologists turned their attention to other areas.
It might have been short-sighted, Dr. Rosen said. Syphilis has made a strong comeback, and drug-resistant gonorrhea has also emerged globally and in at least a few states. No other medical field has stepped in to take up the slack. “Ob.gyns. are busy delivering babies, ID [physicians are] concerned about HIV, and urologists are worried about kidney stones and cancer.” Other than herpes and genital warts, “we have not done well” with management of sexually transmitted diseases, he said.
“I could sense” his frustration
The first issue of Dermatology News carried an article and photospread about scabies that could run today, except that topical permethrin and oral ivermectin have largely replaced benzyl benzoate and sulfur ointments for treatment in the United States. In the article, Scottish dermatologist J. O’D. Alexander, MD, called scabies “the scourge of mankind” and blamed it’s prevalence on “an offhand attitude to the disease which makes control very difficult.”
“I could sense this man’s frustration that people were not recognizing scabies,” Dr. Kovarik said, and it’s no closer to being eradicated than it was in 1970. “It’s still around, and we see it in our clinics. It’s a horrible disease in kids we see in dermatology not infrequently,” and treatment has only advanced a bit.
The article highlights what hasn’t changed much in ID dermatology over the years. Common warts are another one. “With all the evolution in medicine, we don’t have any better treatments approved for common warts than we ever had.” Injecting cidofovir “works great,” but access is a problem, Dr. Tyring said.
Onychomycosis has also proven a tough nut to crack. Readers back in 1970 counted the introduction of the antifungal, griseofulvin, as a major advancement in the 1960s; it’s still a go-to for tinea capitis, but it didn’t work very well for toenail fungus. Terbinafine (Lamisil), approved in 1993, and subsequent developments have helped, but the field still awaits more effective options; a few potential new agents are in the pipeline.
Although there have been major advancements for serious systemic fungal infections, “we’ve mainly seen small steps forward” in ID dermatology, Dr. Tyring said.
Dr. Tyring, Dr. Kovarik, and Dr. Rosen said they had no relevant disclosures.
When Stephen Tyring, MD, PhD, an infectious disease dermatologist, started his career in the early 1980s, he said “we were diagnosing Kaposi’s sarcoma right and left. We would see a new case every day or two.”
It was the early days of the HIV/AIDS epidemic, and dermatologists were at the forefront because HIV/AIDS often presented with skin manifestations. Dr. Tyring, clinical professor in the departments of dermatology, microbiology & molecular genetics and internal medicine at the University of Texas Health Science Center, Houston, and his colleagues referred Kaposi’s patients for chemotherapy and radiation, but the outlook was often grim, especially if lesions developed in the lungs.
Dermatologist don’t see much Kaposi’s anymore because of highly effective treatments for HIV.
Members of the original editorial advisory board saw it coming. In a feature in which board members provided their prediction for the 1970s that appeared in the first issue, New York dermatologist Norman Orentreich, MD, counted the “probable introduction of virucidal agents” as one of the “significant advances or changes that I foresee in the next 10 years.” J. Lamar Callaway, MD, professor of dermatology at Duke University, Durham, N.C., predicted that “the next 10 years should develop effective anti-viral agents for warts, herpes simplex, and herpes zoster.”
To celebrate the 50th anniversary of Dermatology News, we are looking back at how the field has changed since that first issue. The focus this month is infectious disease. There’s a lot to be grateful for but there are also challenges like antibiotic resistance that weren’t on the radar screens of Dr. Orentreich, Dr. Callaway, and their peers in 1970.
All in all, “the only thing I wish we did the old way is sit at the bedside and talk to patients more. We rely so much on technology now that we sometimes lose the art of medicine, which is comforting to the patient,” said Theodore Rosen, MD, an ID dermatologist and professor of dermatology at Baylor College of Medicine, Houston, who’s been in practice for 42 years.
“A lot of advancements against herpes viruses”
One of the biggest wins for ID dermatology over the last 5 decades has been the management of herpes, both herpes simplex virus 1 and 2, as well as herpes zoster virus. It started with the approval of acyclovir in 1981. Before then, “we had no direct therapy for genital herpes, herpes zoster, or disseminated herpes in immunosuppressed or cancer patients,” Dr. Rosen said.
“I can remember doing an interview with Good Morning America when I gave the first IV dose of acyclovir in the city of Houston for really bad disseminated herpes” in an HIV patient, he said, and it worked.
Two derivatives, valacyclovir and famciclovir, became available in the mid-1990s, so today “we have three drugs and some others at the periphery that are all highly effective not only” against herpes, but also for preventing outbreaks; valacyclovir can even prevent asymptomatic shedding, therefore possibly preventing new infections. “That’s a concept we didn’t even have 40 years ago,” Dr. Rosen said.
Cidofovir has also made a difference. The IV formulation was approved for AIDS-associated cytomegalovirus retinitis in 1996 but discontinued a few years later amid concerns of severe renal toxicity. It’s found a new home in dermatology since then, explained ID dermatologist Carrie Kovarik, MD, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
Dermatologists see acyclovir-resistant herpes “heaped up on the genitals in HIV patients,” and there weren’t many options in the past. A few years ago, “we [tried] injecting cidofovir directly into the skin lesions, and it’s been remarkably successful. It is a good way to treat these lesions” if dermatologists can get it compounded, she said.
Shingles vaccines, first the live attenuated zoster vaccine (Zostavax) approved by the Food and Drug Administration in 2006 and the more effective recombinant zoster vaccine (Shingrix) approved in 2017, have also had a significant impact.
Dr. Rosen remembers what it was like when he first started practicing over 40 years ago. Not uncommonly, “we saw horrible cases of shingles,” including one in his uncle, who was left with permanent hand pain long after the rash subsided.
Today, “I see much less shingles, and when I do see it, it’s in a much-attenuated form. [Shingrix], even if it doesn’t prevent the disease, often prevents postherpetic neuralgia,” he said.
Also, with pediatric vaccinations against chicken pox, “we’re probably going to see a whole new generation without shingles, which is huge. We’ve made a lot of advancements against herpes viruses,” Dr. Kovarik said.
“We finally found something that helps”
“We’ve [also] come a really long way with genital wart treatment,” Dr. Kovarik said.
It started with approval of topical imiquimod in 1997. “Before that, we were just killing one wart here and one wart there” but they would often come back and pop up in other areas. Injectable interferon was an option at the time, but people didn’t like all the needles.
With imiquimod, “we finally [had] a way to target HPV [human papillomavirus] and not just scrape” or freeze one wart at a time, and “we were able to generate an inflammatory response in the whole area to clear the virus.” Working with HIV patients, “I see sheets and sheets of confluent warts throughout the whole genital area; to try to freeze that is impossible. Now I have a way to get rid of [genital] warts and keep them away even if you have a big cluster,” she said.
“Sometimes, we’ll do both liquid nitrogen and imiquimod. That’s a good way to tackle people who have a high burden of warts,” Dr. Kovarik noted. Other effective treatments have come out as well, including an ointment formulation of sinecatechins, extracted from green tea, “but you have to put it on several times a day, and insurance companies don’t cover it often,” she said.
Intralesional cidofovir is also proving to be boon for potentially malignant refractory warts in HIV and transplant patients. “It’s an incredible treatment. We can inject that antiviral into warts and get rid of them. We finally found something that helps” these people, Dr. Kovarik said.
The HPV vaccine Gardasil is making a difference, as well. In addition to cervical dysplasia and anogenital cancers, it protects against two condyloma strains. Dr. Rosen said he’s seeing fewer cases of genital warts now than when he started practicing, likely because of the vaccine.
“Organisms that weren’t pathogens are now pathogens”
Antibiotic resistance probably tops the list for what’s changed in a bad way in ID dermatology since 1970. Dr. Rosen remembers at the start of his career that “we never worried about antibiotic resistance. We’d put people on antibiotics for acne, rosacea, and we’d keep them on them for 3 years, 6 years”; resistance wasn’t on the radar screen and was not mentioned once in the first issue of Dermatology News, which was packed with articles and ran 24 pages.
The situation is different now. Driven by decades of overuse in agriculture and the medical system, antibiotic resistance is a concern throughout medicine, and unfortunately, “we have not come nearly as far as fast with antibiotics,” at least the ones dermatologists use, “as we have with antivirals,” Dr. Tyring said.
For instance, methicillin-resistant Staphylococcus aureus (MRSA), first described in the United States in 1968, is “no longer the exception to the rule, but the rule” itself, he said, with carbuncles, furuncles, and abscesses not infrequently growing out MRSA. There are also new drug-resistant forms of old problems like gonorrhea and tuberculosis, among other developments, and impetigo has shifted since 1970 from mostly a Streptococcus infection easily treated with penicillin to often a Staphylococcus disease that’s resistant to it. There’s also been a steady march of new pathogens, including the latest one, SARS-CoV-2, the virus that causes COVID-19, which has been recognized as having a variety of skin manifestations.
“No matter how smart we think we are, nature has a way of putting us back in our place,” Dr. Rosen said.
The bright spot is that “we’ve become very adept at identifying and characterizing” microbes “based on techniques we didn’t even have when I started practicing,” such as polymerase chain reaction. “It has taken a lot of guess work out of treating infectious diseases,” he said.
The widespread use of immunosuppressives such as cyclophosphamide, mycophenolate, azathioprine, rituximab, and other agents used in conjunction with solid organ transplantation, has also been a challenge. “We are seeing infections with really odd organisms. Just recently, I had a patient with fusarium in the skin; it’s a fungus that lives in the dirt. I saw a patient with a species of algae” that normally lives in stagnant water, he commented. “We used to get [things like that] back on reports, and we’d throw them away. You can’t do that anymore. Organisms that weren’t pathogens in the past are now pathogens,” particularly in immunosuppressed people, Dr. Rosen said.
Venereologists no more
There’s been another big change in the field. “Back in the not too distant past, dermatologists in the U.S. were referred to as ‘dermatologist-venereologists.’ ” It goes back to the time when syphilis wasn’t diagnosed and treated early, so patients often presented with secondary skin complications and went to dermatologists for help. As a result, “dermatologists became the most experienced at treating it,” Dr. Tyring said.
That’s faded from practice. Part of the reason is that as late as 2000, syphilis seemed to be on the way out; the Centers for Disease and Control and Prevention even raised the possibility of elimination. Dermatologists turned their attention to other areas.
It might have been short-sighted, Dr. Rosen said. Syphilis has made a strong comeback, and drug-resistant gonorrhea has also emerged globally and in at least a few states. No other medical field has stepped in to take up the slack. “Ob.gyns. are busy delivering babies, ID [physicians are] concerned about HIV, and urologists are worried about kidney stones and cancer.” Other than herpes and genital warts, “we have not done well” with management of sexually transmitted diseases, he said.
“I could sense” his frustration
The first issue of Dermatology News carried an article and photospread about scabies that could run today, except that topical permethrin and oral ivermectin have largely replaced benzyl benzoate and sulfur ointments for treatment in the United States. In the article, Scottish dermatologist J. O’D. Alexander, MD, called scabies “the scourge of mankind” and blamed it’s prevalence on “an offhand attitude to the disease which makes control very difficult.”
“I could sense this man’s frustration that people were not recognizing scabies,” Dr. Kovarik said, and it’s no closer to being eradicated than it was in 1970. “It’s still around, and we see it in our clinics. It’s a horrible disease in kids we see in dermatology not infrequently,” and treatment has only advanced a bit.
The article highlights what hasn’t changed much in ID dermatology over the years. Common warts are another one. “With all the evolution in medicine, we don’t have any better treatments approved for common warts than we ever had.” Injecting cidofovir “works great,” but access is a problem, Dr. Tyring said.
Onychomycosis has also proven a tough nut to crack. Readers back in 1970 counted the introduction of the antifungal, griseofulvin, as a major advancement in the 1960s; it’s still a go-to for tinea capitis, but it didn’t work very well for toenail fungus. Terbinafine (Lamisil), approved in 1993, and subsequent developments have helped, but the field still awaits more effective options; a few potential new agents are in the pipeline.
Although there have been major advancements for serious systemic fungal infections, “we’ve mainly seen small steps forward” in ID dermatology, Dr. Tyring said.
Dr. Tyring, Dr. Kovarik, and Dr. Rosen said they had no relevant disclosures.
When Stephen Tyring, MD, PhD, an infectious disease dermatologist, started his career in the early 1980s, he said “we were diagnosing Kaposi’s sarcoma right and left. We would see a new case every day or two.”
It was the early days of the HIV/AIDS epidemic, and dermatologists were at the forefront because HIV/AIDS often presented with skin manifestations. Dr. Tyring, clinical professor in the departments of dermatology, microbiology & molecular genetics and internal medicine at the University of Texas Health Science Center, Houston, and his colleagues referred Kaposi’s patients for chemotherapy and radiation, but the outlook was often grim, especially if lesions developed in the lungs.
Dermatologist don’t see much Kaposi’s anymore because of highly effective treatments for HIV.
Members of the original editorial advisory board saw it coming. In a feature in which board members provided their prediction for the 1970s that appeared in the first issue, New York dermatologist Norman Orentreich, MD, counted the “probable introduction of virucidal agents” as one of the “significant advances or changes that I foresee in the next 10 years.” J. Lamar Callaway, MD, professor of dermatology at Duke University, Durham, N.C., predicted that “the next 10 years should develop effective anti-viral agents for warts, herpes simplex, and herpes zoster.”
To celebrate the 50th anniversary of Dermatology News, we are looking back at how the field has changed since that first issue. The focus this month is infectious disease. There’s a lot to be grateful for but there are also challenges like antibiotic resistance that weren’t on the radar screens of Dr. Orentreich, Dr. Callaway, and their peers in 1970.
All in all, “the only thing I wish we did the old way is sit at the bedside and talk to patients more. We rely so much on technology now that we sometimes lose the art of medicine, which is comforting to the patient,” said Theodore Rosen, MD, an ID dermatologist and professor of dermatology at Baylor College of Medicine, Houston, who’s been in practice for 42 years.
“A lot of advancements against herpes viruses”
One of the biggest wins for ID dermatology over the last 5 decades has been the management of herpes, both herpes simplex virus 1 and 2, as well as herpes zoster virus. It started with the approval of acyclovir in 1981. Before then, “we had no direct therapy for genital herpes, herpes zoster, or disseminated herpes in immunosuppressed or cancer patients,” Dr. Rosen said.
“I can remember doing an interview with Good Morning America when I gave the first IV dose of acyclovir in the city of Houston for really bad disseminated herpes” in an HIV patient, he said, and it worked.
Two derivatives, valacyclovir and famciclovir, became available in the mid-1990s, so today “we have three drugs and some others at the periphery that are all highly effective not only” against herpes, but also for preventing outbreaks; valacyclovir can even prevent asymptomatic shedding, therefore possibly preventing new infections. “That’s a concept we didn’t even have 40 years ago,” Dr. Rosen said.
Cidofovir has also made a difference. The IV formulation was approved for AIDS-associated cytomegalovirus retinitis in 1996 but discontinued a few years later amid concerns of severe renal toxicity. It’s found a new home in dermatology since then, explained ID dermatologist Carrie Kovarik, MD, associate professor of dermatology at the University of Pennsylvania, Philadelphia.
Dermatologists see acyclovir-resistant herpes “heaped up on the genitals in HIV patients,” and there weren’t many options in the past. A few years ago, “we [tried] injecting cidofovir directly into the skin lesions, and it’s been remarkably successful. It is a good way to treat these lesions” if dermatologists can get it compounded, she said.
Shingles vaccines, first the live attenuated zoster vaccine (Zostavax) approved by the Food and Drug Administration in 2006 and the more effective recombinant zoster vaccine (Shingrix) approved in 2017, have also had a significant impact.
Dr. Rosen remembers what it was like when he first started practicing over 40 years ago. Not uncommonly, “we saw horrible cases of shingles,” including one in his uncle, who was left with permanent hand pain long after the rash subsided.
Today, “I see much less shingles, and when I do see it, it’s in a much-attenuated form. [Shingrix], even if it doesn’t prevent the disease, often prevents postherpetic neuralgia,” he said.
Also, with pediatric vaccinations against chicken pox, “we’re probably going to see a whole new generation without shingles, which is huge. We’ve made a lot of advancements against herpes viruses,” Dr. Kovarik said.
“We finally found something that helps”
“We’ve [also] come a really long way with genital wart treatment,” Dr. Kovarik said.
It started with approval of topical imiquimod in 1997. “Before that, we were just killing one wart here and one wart there” but they would often come back and pop up in other areas. Injectable interferon was an option at the time, but people didn’t like all the needles.
With imiquimod, “we finally [had] a way to target HPV [human papillomavirus] and not just scrape” or freeze one wart at a time, and “we were able to generate an inflammatory response in the whole area to clear the virus.” Working with HIV patients, “I see sheets and sheets of confluent warts throughout the whole genital area; to try to freeze that is impossible. Now I have a way to get rid of [genital] warts and keep them away even if you have a big cluster,” she said.
“Sometimes, we’ll do both liquid nitrogen and imiquimod. That’s a good way to tackle people who have a high burden of warts,” Dr. Kovarik noted. Other effective treatments have come out as well, including an ointment formulation of sinecatechins, extracted from green tea, “but you have to put it on several times a day, and insurance companies don’t cover it often,” she said.
Intralesional cidofovir is also proving to be boon for potentially malignant refractory warts in HIV and transplant patients. “It’s an incredible treatment. We can inject that antiviral into warts and get rid of them. We finally found something that helps” these people, Dr. Kovarik said.
The HPV vaccine Gardasil is making a difference, as well. In addition to cervical dysplasia and anogenital cancers, it protects against two condyloma strains. Dr. Rosen said he’s seeing fewer cases of genital warts now than when he started practicing, likely because of the vaccine.
“Organisms that weren’t pathogens are now pathogens”
Antibiotic resistance probably tops the list for what’s changed in a bad way in ID dermatology since 1970. Dr. Rosen remembers at the start of his career that “we never worried about antibiotic resistance. We’d put people on antibiotics for acne, rosacea, and we’d keep them on them for 3 years, 6 years”; resistance wasn’t on the radar screen and was not mentioned once in the first issue of Dermatology News, which was packed with articles and ran 24 pages.
The situation is different now. Driven by decades of overuse in agriculture and the medical system, antibiotic resistance is a concern throughout medicine, and unfortunately, “we have not come nearly as far as fast with antibiotics,” at least the ones dermatologists use, “as we have with antivirals,” Dr. Tyring said.
For instance, methicillin-resistant Staphylococcus aureus (MRSA), first described in the United States in 1968, is “no longer the exception to the rule, but the rule” itself, he said, with carbuncles, furuncles, and abscesses not infrequently growing out MRSA. There are also new drug-resistant forms of old problems like gonorrhea and tuberculosis, among other developments, and impetigo has shifted since 1970 from mostly a Streptococcus infection easily treated with penicillin to often a Staphylococcus disease that’s resistant to it. There’s also been a steady march of new pathogens, including the latest one, SARS-CoV-2, the virus that causes COVID-19, which has been recognized as having a variety of skin manifestations.
“No matter how smart we think we are, nature has a way of putting us back in our place,” Dr. Rosen said.
The bright spot is that “we’ve become very adept at identifying and characterizing” microbes “based on techniques we didn’t even have when I started practicing,” such as polymerase chain reaction. “It has taken a lot of guess work out of treating infectious diseases,” he said.
The widespread use of immunosuppressives such as cyclophosphamide, mycophenolate, azathioprine, rituximab, and other agents used in conjunction with solid organ transplantation, has also been a challenge. “We are seeing infections with really odd organisms. Just recently, I had a patient with fusarium in the skin; it’s a fungus that lives in the dirt. I saw a patient with a species of algae” that normally lives in stagnant water, he commented. “We used to get [things like that] back on reports, and we’d throw them away. You can’t do that anymore. Organisms that weren’t pathogens in the past are now pathogens,” particularly in immunosuppressed people, Dr. Rosen said.
Venereologists no more
There’s been another big change in the field. “Back in the not too distant past, dermatologists in the U.S. were referred to as ‘dermatologist-venereologists.’ ” It goes back to the time when syphilis wasn’t diagnosed and treated early, so patients often presented with secondary skin complications and went to dermatologists for help. As a result, “dermatologists became the most experienced at treating it,” Dr. Tyring said.
That’s faded from practice. Part of the reason is that as late as 2000, syphilis seemed to be on the way out; the Centers for Disease and Control and Prevention even raised the possibility of elimination. Dermatologists turned their attention to other areas.
It might have been short-sighted, Dr. Rosen said. Syphilis has made a strong comeback, and drug-resistant gonorrhea has also emerged globally and in at least a few states. No other medical field has stepped in to take up the slack. “Ob.gyns. are busy delivering babies, ID [physicians are] concerned about HIV, and urologists are worried about kidney stones and cancer.” Other than herpes and genital warts, “we have not done well” with management of sexually transmitted diseases, he said.
“I could sense” his frustration
The first issue of Dermatology News carried an article and photospread about scabies that could run today, except that topical permethrin and oral ivermectin have largely replaced benzyl benzoate and sulfur ointments for treatment in the United States. In the article, Scottish dermatologist J. O’D. Alexander, MD, called scabies “the scourge of mankind” and blamed it’s prevalence on “an offhand attitude to the disease which makes control very difficult.”
“I could sense this man’s frustration that people were not recognizing scabies,” Dr. Kovarik said, and it’s no closer to being eradicated than it was in 1970. “It’s still around, and we see it in our clinics. It’s a horrible disease in kids we see in dermatology not infrequently,” and treatment has only advanced a bit.
The article highlights what hasn’t changed much in ID dermatology over the years. Common warts are another one. “With all the evolution in medicine, we don’t have any better treatments approved for common warts than we ever had.” Injecting cidofovir “works great,” but access is a problem, Dr. Tyring said.
Onychomycosis has also proven a tough nut to crack. Readers back in 1970 counted the introduction of the antifungal, griseofulvin, as a major advancement in the 1960s; it’s still a go-to for tinea capitis, but it didn’t work very well for toenail fungus. Terbinafine (Lamisil), approved in 1993, and subsequent developments have helped, but the field still awaits more effective options; a few potential new agents are in the pipeline.
Although there have been major advancements for serious systemic fungal infections, “we’ve mainly seen small steps forward” in ID dermatology, Dr. Tyring said.
Dr. Tyring, Dr. Kovarik, and Dr. Rosen said they had no relevant disclosures.
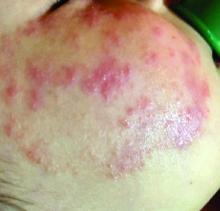
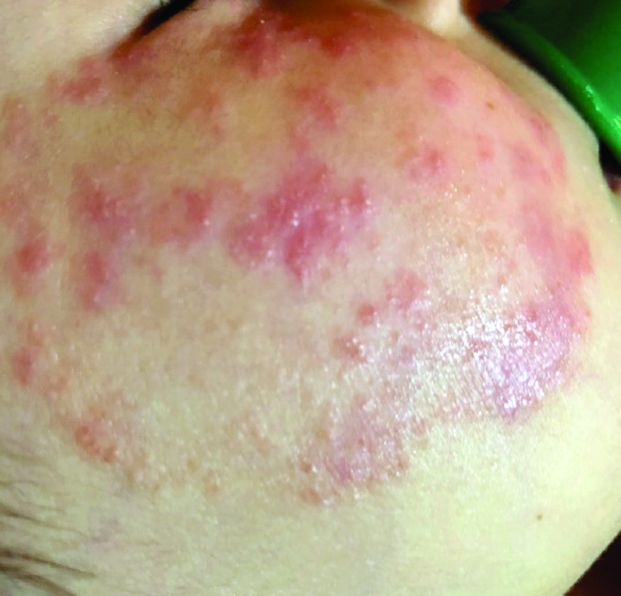
What presents as deep erythematous papules, pustules, and may form an annular or circular plaque?
Scrapings of the child’s rash were analyzed with potassium hydroxide (KOH) under microscopy which revealed multiple septate hyphae.
She was diagnosed with Majocchi’s granuloma. The fungal culture was positive for Trichophyton rubrum.
Majocchi’s granuloma (MG) is cutaneous mycosis in which the fungal infection goes deeper into the hair follicle causing granulomatous folliculitis and perifolliculitis.1 It was first described by Domenico Majocchi in 1883, and he named the condition “granuloma tricofitico.”2
It is commonly caused by T. rubrum but also can be caused by T. mentagrophytes, T. tonsurans, T. verrucosum, Microsporum canis, and Epidermophyton floccosum.2,3 Patients at risk for developing this infection include those previously treated with topical corticosteroids, immunosuppressed patients, patients with areas under occlusion, and those with areas traumatized by shaving. This infection is most commonly seen in the lower extremities, but can happen anywhere in the body. The lesions present as deep erythematous papules, pustules, and may form an annular or circular plaque as seen on our patient.
A KOH test of skin scrapings and hair extractions often can reveal fungal hyphae. Identification of the pathogen can be performed with culture or polymerase chain reaction of skin samples. If the diagnosis is uncertain or the KOH is negative, a skin biopsy can be performed. Histopathologic examination reveals perifollicular granulomas with associated dermal abscesses. Giant cells may be observed. MG is associated with chronic inflammation with lymphocytes, macrophages, epithelioid cells, and scattered multinucleated giant cells.2,3
The differential diagnosis for these lesions in children includes other granulomatous conditions such as granulomatous rosacea, sarcoidosis, and granuloma faciale, as well as bacterial or atypical mycobacterial infections, cutaneous leishmaniasis, and eosinophilic pustular folliculitis.
Treatment of MG requires systemic treatment with griseofulvin, itraconazole, or terbinafine for at least 4-8 weeks or until all the lesions have resolved. Our patient was treated with 6 weeks of high-dose griseofulvin with resolution of her lesions.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at pdnews@mdedge.com.
References
1. Dermatol Online J. 2018 Dec 15;24(12):13030/qt89k4t6wj.
2. Med Mycol. 2012 Jul;50(5):449-57.
3. Clin Microbiol Rev. 2011 Apr;24(2):247-80.
Scrapings of the child’s rash were analyzed with potassium hydroxide (KOH) under microscopy which revealed multiple septate hyphae.
She was diagnosed with Majocchi’s granuloma. The fungal culture was positive for Trichophyton rubrum.
Majocchi’s granuloma (MG) is cutaneous mycosis in which the fungal infection goes deeper into the hair follicle causing granulomatous folliculitis and perifolliculitis.1 It was first described by Domenico Majocchi in 1883, and he named the condition “granuloma tricofitico.”2
It is commonly caused by T. rubrum but also can be caused by T. mentagrophytes, T. tonsurans, T. verrucosum, Microsporum canis, and Epidermophyton floccosum.2,3 Patients at risk for developing this infection include those previously treated with topical corticosteroids, immunosuppressed patients, patients with areas under occlusion, and those with areas traumatized by shaving. This infection is most commonly seen in the lower extremities, but can happen anywhere in the body. The lesions present as deep erythematous papules, pustules, and may form an annular or circular plaque as seen on our patient.
A KOH test of skin scrapings and hair extractions often can reveal fungal hyphae. Identification of the pathogen can be performed with culture or polymerase chain reaction of skin samples. If the diagnosis is uncertain or the KOH is negative, a skin biopsy can be performed. Histopathologic examination reveals perifollicular granulomas with associated dermal abscesses. Giant cells may be observed. MG is associated with chronic inflammation with lymphocytes, macrophages, epithelioid cells, and scattered multinucleated giant cells.2,3
The differential diagnosis for these lesions in children includes other granulomatous conditions such as granulomatous rosacea, sarcoidosis, and granuloma faciale, as well as bacterial or atypical mycobacterial infections, cutaneous leishmaniasis, and eosinophilic pustular folliculitis.
Treatment of MG requires systemic treatment with griseofulvin, itraconazole, or terbinafine for at least 4-8 weeks or until all the lesions have resolved. Our patient was treated with 6 weeks of high-dose griseofulvin with resolution of her lesions.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at pdnews@mdedge.com.
References
1. Dermatol Online J. 2018 Dec 15;24(12):13030/qt89k4t6wj.
2. Med Mycol. 2012 Jul;50(5):449-57.
3. Clin Microbiol Rev. 2011 Apr;24(2):247-80.
Scrapings of the child’s rash were analyzed with potassium hydroxide (KOH) under microscopy which revealed multiple septate hyphae.
She was diagnosed with Majocchi’s granuloma. The fungal culture was positive for Trichophyton rubrum.
Majocchi’s granuloma (MG) is cutaneous mycosis in which the fungal infection goes deeper into the hair follicle causing granulomatous folliculitis and perifolliculitis.1 It was first described by Domenico Majocchi in 1883, and he named the condition “granuloma tricofitico.”2
It is commonly caused by T. rubrum but also can be caused by T. mentagrophytes, T. tonsurans, T. verrucosum, Microsporum canis, and Epidermophyton floccosum.2,3 Patients at risk for developing this infection include those previously treated with topical corticosteroids, immunosuppressed patients, patients with areas under occlusion, and those with areas traumatized by shaving. This infection is most commonly seen in the lower extremities, but can happen anywhere in the body. The lesions present as deep erythematous papules, pustules, and may form an annular or circular plaque as seen on our patient.
A KOH test of skin scrapings and hair extractions often can reveal fungal hyphae. Identification of the pathogen can be performed with culture or polymerase chain reaction of skin samples. If the diagnosis is uncertain or the KOH is negative, a skin biopsy can be performed. Histopathologic examination reveals perifollicular granulomas with associated dermal abscesses. Giant cells may be observed. MG is associated with chronic inflammation with lymphocytes, macrophages, epithelioid cells, and scattered multinucleated giant cells.2,3
The differential diagnosis for these lesions in children includes other granulomatous conditions such as granulomatous rosacea, sarcoidosis, and granuloma faciale, as well as bacterial or atypical mycobacterial infections, cutaneous leishmaniasis, and eosinophilic pustular folliculitis.
Treatment of MG requires systemic treatment with griseofulvin, itraconazole, or terbinafine for at least 4-8 weeks or until all the lesions have resolved. Our patient was treated with 6 weeks of high-dose griseofulvin with resolution of her lesions.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at pdnews@mdedge.com.
References
1. Dermatol Online J. 2018 Dec 15;24(12):13030/qt89k4t6wj.
2. Med Mycol. 2012 Jul;50(5):449-57.
3. Clin Microbiol Rev. 2011 Apr;24(2):247-80.
A 3-year-old girl with a known history of eczema presented to our dermatology clinic for evaluation of a persistent rash for about a year on the right cheek.
The mother reported she was treating the lesions with hydrocortisone cream 2.5% as instructed previously for her eczema. Initially the rash got partially better but then started getting worse again. The area was itchy.
The child was later seen in the emergency department, where she was recommended to treat the area with a combination cream of terbinafine 1% and betamethasone dipropionate 0.05%. The mother applied this cream as instructed for 3 weeks with some improvement of the lesions on the cheek.
A few weeks later, pimples started coming back. The mother tried the medication again but this time it was not helpful and the rash continued to expand.
The mother reported having a rash on her hand months back, which she successfully treated with the combination cream provided at the emergency department. They have no pets at home.
The child has a little sister who also has mild eczema.
She goes to day care and dances ballet.
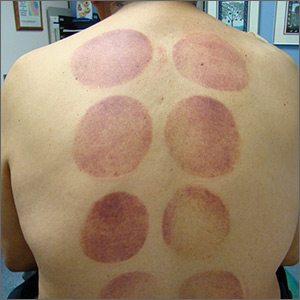
Ecchymotic patches
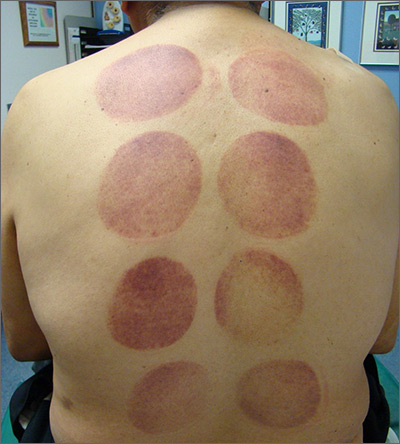
This patient’s circular ecchymotic patches were due to cupping. One of the clues that this was iatrogenic was the regular and repeated pattern on the skin.
Cupping is a centuries old treatment for pain relief (among other things) that involves applying glass globes or other hollow materials to the skin to create a vacuum. Traditionally, this vacuum is created by heating the air inside the vessel and then holding the vessel in place as the air cools. Practitioners may also use more modern instruments to induce the vacuum that are similar to those used to assist in vaginal deliveries. The mechanical devices leave these circular ecchymotic marks. The ecchymosis fades over time, and this procedure has been shown to significantly reduce myofascial neck and back pain in small trials.
It is important to recognize geometric patterns that are iatrogenic or due to abuse when evaluating skin findings. If skin findings do not follow dermatomal distributions, typical exanthem, or other classic patterns or presentations, there is the possibility that the pattern may be the result of neglect or abuse. On inspection, consider whether an odd pattern may have been caused from a belt buckle, striking instrument, furniture, medical equipment, or a hand strike.
This patient’s findings were consistent with his history of visiting a physical therapist for cupping. No treatment was required; the patient’s back pain from his car accident was improving, and the cupping marks were not troubling him.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Wang YT, Qi Y, Tang FY, et al. The effect of cupping therapy for low back pain: a meta-analysis based on existing randomized controlled trials. J Back Musculoskelet Rehabil. 2017;30:1187-1195.

This patient’s circular ecchymotic patches were due to cupping. One of the clues that this was iatrogenic was the regular and repeated pattern on the skin.
Cupping is a centuries old treatment for pain relief (among other things) that involves applying glass globes or other hollow materials to the skin to create a vacuum. Traditionally, this vacuum is created by heating the air inside the vessel and then holding the vessel in place as the air cools. Practitioners may also use more modern instruments to induce the vacuum that are similar to those used to assist in vaginal deliveries. The mechanical devices leave these circular ecchymotic marks. The ecchymosis fades over time, and this procedure has been shown to significantly reduce myofascial neck and back pain in small trials.
It is important to recognize geometric patterns that are iatrogenic or due to abuse when evaluating skin findings. If skin findings do not follow dermatomal distributions, typical exanthem, or other classic patterns or presentations, there is the possibility that the pattern may be the result of neglect or abuse. On inspection, consider whether an odd pattern may have been caused from a belt buckle, striking instrument, furniture, medical equipment, or a hand strike.
This patient’s findings were consistent with his history of visiting a physical therapist for cupping. No treatment was required; the patient’s back pain from his car accident was improving, and the cupping marks were not troubling him.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This patient’s circular ecchymotic patches were due to cupping. One of the clues that this was iatrogenic was the regular and repeated pattern on the skin.
Cupping is a centuries old treatment for pain relief (among other things) that involves applying glass globes or other hollow materials to the skin to create a vacuum. Traditionally, this vacuum is created by heating the air inside the vessel and then holding the vessel in place as the air cools. Practitioners may also use more modern instruments to induce the vacuum that are similar to those used to assist in vaginal deliveries. The mechanical devices leave these circular ecchymotic marks. The ecchymosis fades over time, and this procedure has been shown to significantly reduce myofascial neck and back pain in small trials.
It is important to recognize geometric patterns that are iatrogenic or due to abuse when evaluating skin findings. If skin findings do not follow dermatomal distributions, typical exanthem, or other classic patterns or presentations, there is the possibility that the pattern may be the result of neglect or abuse. On inspection, consider whether an odd pattern may have been caused from a belt buckle, striking instrument, furniture, medical equipment, or a hand strike.
This patient’s findings were consistent with his history of visiting a physical therapist for cupping. No treatment was required; the patient’s back pain from his car accident was improving, and the cupping marks were not troubling him.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Wang YT, Qi Y, Tang FY, et al. The effect of cupping therapy for low back pain: a meta-analysis based on existing randomized controlled trials. J Back Musculoskelet Rehabil. 2017;30:1187-1195.
Wang YT, Qi Y, Tang FY, et al. The effect of cupping therapy for low back pain: a meta-analysis based on existing randomized controlled trials. J Back Musculoskelet Rehabil. 2017;30:1187-1195.
Daily Recap 6/17
Here are the stories our MDedge editors across specialties think you need to know about today:
Comorbidities increase COVID-19 deaths by factor of 12
COVID-19 patients with an underlying condition are 6 times as likely to be hospitalized and 12 times as likely to die, compared with those who have no such condition, according to the CDC.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females.
The pandemic “continues to affect all populations and result in severe outcomes including death,” noted the CDC, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.” Read more.
Preventive services coalition recommends routine anxiety screening for women
Women and girls aged 13 years and older with no current diagnosis of anxiety should be screened routinely for anxiety, according to a new recommendation from the Women’s Preventive Services Initiative.
The lifetime prevalence of anxiety disorders in women in the United States is 40%, approximately twice that of men, and anxiety can be a manifestation of underlying issues including posttraumatic stress, sexual harassment, and assault.
“The WPSI based its rationale for anxiety screening on several considerations,” the researchers noted. “Anxiety disorders are the most prevalent mental health disorders in women, and the problems created by untreated anxiety can impair function in all areas of a woman’s life.” Read more.
High-fat, high-sugar diet may promote adult acne
A diet higher in fat, sugar, and milk was associated with having acne in a cross-sectional study of approximately 24,000 adults in France.
Although acne patients may believe that eating certain foods exacerbates acne, data on the effects of nutrition on acne, including associations between acne and a high-glycemic diet, are limited and have produced conflicting results, noted investigators.
“The results of our study appear to support the hypothesis that the Western diet (rich in animal products and fatty and sugary foods) is associated with the presence of acne in adulthood,” the researchers concluded.
Population study supports migraine-dementia link
Preliminary results from a population-based cohort study support previous reports that migraine is a midlife risk factor for dementia later in life, but further determined that migraine with aura and frequent hospital contacts significantly increased dementia risk after age 60 years, according to results from a Danish registry presented at the virtual annual meeting of the American Headache Society.
The preliminary findings revealed that the median age at diagnosis was 49 years and about 70% of the migraine population were women. “There was a 50% higher dementia rate in individuals who had any migraine diagnosis,” Dr. Islamoska said.
“To the best of our knowledge, no previous national register–based studies have investigated the risk of dementia among individuals who suffer from migraine with aura,” Dr. Sabrina Islamoska said.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Comorbidities increase COVID-19 deaths by factor of 12
COVID-19 patients with an underlying condition are 6 times as likely to be hospitalized and 12 times as likely to die, compared with those who have no such condition, according to the CDC.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females.
The pandemic “continues to affect all populations and result in severe outcomes including death,” noted the CDC, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.” Read more.
Preventive services coalition recommends routine anxiety screening for women
Women and girls aged 13 years and older with no current diagnosis of anxiety should be screened routinely for anxiety, according to a new recommendation from the Women’s Preventive Services Initiative.
The lifetime prevalence of anxiety disorders in women in the United States is 40%, approximately twice that of men, and anxiety can be a manifestation of underlying issues including posttraumatic stress, sexual harassment, and assault.
“The WPSI based its rationale for anxiety screening on several considerations,” the researchers noted. “Anxiety disorders are the most prevalent mental health disorders in women, and the problems created by untreated anxiety can impair function in all areas of a woman’s life.” Read more.
High-fat, high-sugar diet may promote adult acne
A diet higher in fat, sugar, and milk was associated with having acne in a cross-sectional study of approximately 24,000 adults in France.
Although acne patients may believe that eating certain foods exacerbates acne, data on the effects of nutrition on acne, including associations between acne and a high-glycemic diet, are limited and have produced conflicting results, noted investigators.
“The results of our study appear to support the hypothesis that the Western diet (rich in animal products and fatty and sugary foods) is associated with the presence of acne in adulthood,” the researchers concluded.
Population study supports migraine-dementia link
Preliminary results from a population-based cohort study support previous reports that migraine is a midlife risk factor for dementia later in life, but further determined that migraine with aura and frequent hospital contacts significantly increased dementia risk after age 60 years, according to results from a Danish registry presented at the virtual annual meeting of the American Headache Society.
The preliminary findings revealed that the median age at diagnosis was 49 years and about 70% of the migraine population were women. “There was a 50% higher dementia rate in individuals who had any migraine diagnosis,” Dr. Islamoska said.
“To the best of our knowledge, no previous national register–based studies have investigated the risk of dementia among individuals who suffer from migraine with aura,” Dr. Sabrina Islamoska said.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Comorbidities increase COVID-19 deaths by factor of 12
COVID-19 patients with an underlying condition are 6 times as likely to be hospitalized and 12 times as likely to die, compared with those who have no such condition, according to the CDC.
The most frequently reported underlying conditions were cardiovascular disease (32%), diabetes (30%), chronic lung disease (18%), and renal disease (7.6%), and there were no significant differences between males and females.
The pandemic “continues to affect all populations and result in severe outcomes including death,” noted the CDC, emphasizing “the continued need for community mitigation strategies, especially for vulnerable populations, to slow COVID-19 transmission.” Read more.
Preventive services coalition recommends routine anxiety screening for women
Women and girls aged 13 years and older with no current diagnosis of anxiety should be screened routinely for anxiety, according to a new recommendation from the Women’s Preventive Services Initiative.
The lifetime prevalence of anxiety disorders in women in the United States is 40%, approximately twice that of men, and anxiety can be a manifestation of underlying issues including posttraumatic stress, sexual harassment, and assault.
“The WPSI based its rationale for anxiety screening on several considerations,” the researchers noted. “Anxiety disorders are the most prevalent mental health disorders in women, and the problems created by untreated anxiety can impair function in all areas of a woman’s life.” Read more.
High-fat, high-sugar diet may promote adult acne
A diet higher in fat, sugar, and milk was associated with having acne in a cross-sectional study of approximately 24,000 adults in France.
Although acne patients may believe that eating certain foods exacerbates acne, data on the effects of nutrition on acne, including associations between acne and a high-glycemic diet, are limited and have produced conflicting results, noted investigators.
“The results of our study appear to support the hypothesis that the Western diet (rich in animal products and fatty and sugary foods) is associated with the presence of acne in adulthood,” the researchers concluded.
Population study supports migraine-dementia link
Preliminary results from a population-based cohort study support previous reports that migraine is a midlife risk factor for dementia later in life, but further determined that migraine with aura and frequent hospital contacts significantly increased dementia risk after age 60 years, according to results from a Danish registry presented at the virtual annual meeting of the American Headache Society.
The preliminary findings revealed that the median age at diagnosis was 49 years and about 70% of the migraine population were women. “There was a 50% higher dementia rate in individuals who had any migraine diagnosis,” Dr. Islamoska said.
“To the best of our knowledge, no previous national register–based studies have investigated the risk of dementia among individuals who suffer from migraine with aura,” Dr. Sabrina Islamoska said.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Vulvar melanoma is increasing in older women
Maia K. Erickson reported in a poster at the virtual annual meeting of the American Academy of Dermatology.
These are often aggressive malignancies. The 5-year survival following diagnosis of vulvar melanoma in women aged 60 years or older was 39.7%, compared with 61.9% in younger women, according to Ms. Erickson, a visiting research fellow in the department of dermatology at Northwestern University, Chicago.
She presented a population-based study of epidemiologic trends in vulvar melanoma based upon analysis of the National Cancer Institute’s Surveillance, Epidemiology and End Results database. Vulvar melanoma was rare during the study years 2000-2016, with an overall incidence rate of 0.1 cases per 100,000 women. That worked out to 746 analyzable cases. Of note, the incidence rate ratio was 680% higher in older women (age 60 and older).
One reason for the markedly worse 5-year survival in older women was that the predominant histologic subtype of vulvar melanoma in that population was nodular melanoma, accounting for 48% of the cases where a histologic subtype was specified. In contrast, the less-aggressive superficial spreading melanoma subtype prevailed in patients aged under 60 years, accounting for 63% of cases.
About 93% of vulvar melanomas occurred in whites; 63% were local and 8.7% were metastatic.
Ms. Erickson noted that the vulva is the most common site for gynecologic tract melanomas, accounting for 70% of them. And while the female genitalia make up only 1%-2% of body surface area, that’s the anatomic site of up to 7% of all melanomas in women.
She reported having no financial conflicts regarding her study.
Maia K. Erickson reported in a poster at the virtual annual meeting of the American Academy of Dermatology.
These are often aggressive malignancies. The 5-year survival following diagnosis of vulvar melanoma in women aged 60 years or older was 39.7%, compared with 61.9% in younger women, according to Ms. Erickson, a visiting research fellow in the department of dermatology at Northwestern University, Chicago.
She presented a population-based study of epidemiologic trends in vulvar melanoma based upon analysis of the National Cancer Institute’s Surveillance, Epidemiology and End Results database. Vulvar melanoma was rare during the study years 2000-2016, with an overall incidence rate of 0.1 cases per 100,000 women. That worked out to 746 analyzable cases. Of note, the incidence rate ratio was 680% higher in older women (age 60 and older).
One reason for the markedly worse 5-year survival in older women was that the predominant histologic subtype of vulvar melanoma in that population was nodular melanoma, accounting for 48% of the cases where a histologic subtype was specified. In contrast, the less-aggressive superficial spreading melanoma subtype prevailed in patients aged under 60 years, accounting for 63% of cases.
About 93% of vulvar melanomas occurred in whites; 63% were local and 8.7% were metastatic.
Ms. Erickson noted that the vulva is the most common site for gynecologic tract melanomas, accounting for 70% of them. And while the female genitalia make up only 1%-2% of body surface area, that’s the anatomic site of up to 7% of all melanomas in women.
She reported having no financial conflicts regarding her study.
Maia K. Erickson reported in a poster at the virtual annual meeting of the American Academy of Dermatology.
These are often aggressive malignancies. The 5-year survival following diagnosis of vulvar melanoma in women aged 60 years or older was 39.7%, compared with 61.9% in younger women, according to Ms. Erickson, a visiting research fellow in the department of dermatology at Northwestern University, Chicago.
She presented a population-based study of epidemiologic trends in vulvar melanoma based upon analysis of the National Cancer Institute’s Surveillance, Epidemiology and End Results database. Vulvar melanoma was rare during the study years 2000-2016, with an overall incidence rate of 0.1 cases per 100,000 women. That worked out to 746 analyzable cases. Of note, the incidence rate ratio was 680% higher in older women (age 60 and older).
One reason for the markedly worse 5-year survival in older women was that the predominant histologic subtype of vulvar melanoma in that population was nodular melanoma, accounting for 48% of the cases where a histologic subtype was specified. In contrast, the less-aggressive superficial spreading melanoma subtype prevailed in patients aged under 60 years, accounting for 63% of cases.
About 93% of vulvar melanomas occurred in whites; 63% were local and 8.7% were metastatic.
Ms. Erickson noted that the vulva is the most common site for gynecologic tract melanomas, accounting for 70% of them. And while the female genitalia make up only 1%-2% of body surface area, that’s the anatomic site of up to 7% of all melanomas in women.
She reported having no financial conflicts regarding her study.
FROM AAD 2020
Study compares pulse vs. continuous therapy for dermatophyte toenail onychomycosis
There appear to be results from a systematic review and network meta-analysis showed.
“Previous meta-analyses of pulse and continuous therapies have generated ambiguous results,” study authors led by Aditya K. Gupta, MD, PhD, wrote in a poster abstract presented at the virtual annual meeting of the American Academy of Dermatology. “There are few head-to-head clinical studies and no meta-analyses comparing regimens of terbinafine to regimens of itraconazole.”
In what is believed to be the first study of its kind, Dr. Gupta, professor of dermatology at the University of Toronto, and colleagues used network meta-analysis to compare pulse and continuous systemic therapies for toenail onychomycosis. They used PubMed to search for randomized, controlled trials of oral antifungal treatments for the condition in patients aged 18 years and older that included data on mycologic cure, complete cure, adverse events, and dropout rates. Treatment effects were based on intention-to-treat cure rates, and the researchers excluded studies of ketoconazole and griseofulvin since they are no longer indicated for the condition.
For their network meta-analysis, Dr. Gupta and colleagues evaluated 22 studies from 20 publications that included 4,205 randomized patients. Data on complete cure were excluded because of a lack of studies. When the researchers compared all treatments to placebo, the likelihood of mycologic cure did not differ significantly between continuous and pulse regimens for terbinafine and itraconazole. Compared with placebo, the most successful treatments were continuous terbinafine 250 mg daily for 24 weeks (risk ratio of achieving mycologic cure, 11.0) and continuous terbinafine 250 mg daily for 16 weeks (RR, 8.90). The researchers also observed no significant differences in the likelihood of adverse events between any continuous and pulse regimens of terbinafine, itraconazole, and fluconazole.
“Although continuous terbinafine 250 mg for 24 weeks was significantly more likely to produce mycologic cure than continuous itraconazole 200 mg for 12 weeks and weekly fluconazole (150-450 mg), it is not significantly different from the other included treatments,” Dr. Gupta and colleagues wrote in the abstract. “Considering the fungal life cycle, pulse therapy should theoretically be as effective as, or more effective than, continuous therapies: the sudden high concentration of an antifungal drug eliminates hyphae, sparing already-present spores. During the ‘off’ portion, these spores may germinate and be eliminated during the next pulse. Continuous therapy spares the spores, allowing them to germinate once treatment ends.”
They went on to note that, in clinical practice, “neither continuous nor pulse therapy is necessarily better. It is possible that the drug concentration in the nail is maintained during the ‘off’ period of pulse therapy. In both therapies, it may be that residual spores that have not been eliminated by the end of therapy are left to germinate, possibly contributing to the recalcitrant nature of onychomycosis.”
The study was awarded fourth place in the AAD’s 2020 poster awards. Dr. Gupta disclosed that he is a clinical trials investigator for Moberg Pharma and Bausch Health Canada and a speaker for Bausch Health Canada.
SOURCE: Gupta A et al. AAD 20, Abstract 16014.
There appear to be results from a systematic review and network meta-analysis showed.
“Previous meta-analyses of pulse and continuous therapies have generated ambiguous results,” study authors led by Aditya K. Gupta, MD, PhD, wrote in a poster abstract presented at the virtual annual meeting of the American Academy of Dermatology. “There are few head-to-head clinical studies and no meta-analyses comparing regimens of terbinafine to regimens of itraconazole.”
In what is believed to be the first study of its kind, Dr. Gupta, professor of dermatology at the University of Toronto, and colleagues used network meta-analysis to compare pulse and continuous systemic therapies for toenail onychomycosis. They used PubMed to search for randomized, controlled trials of oral antifungal treatments for the condition in patients aged 18 years and older that included data on mycologic cure, complete cure, adverse events, and dropout rates. Treatment effects were based on intention-to-treat cure rates, and the researchers excluded studies of ketoconazole and griseofulvin since they are no longer indicated for the condition.
For their network meta-analysis, Dr. Gupta and colleagues evaluated 22 studies from 20 publications that included 4,205 randomized patients. Data on complete cure were excluded because of a lack of studies. When the researchers compared all treatments to placebo, the likelihood of mycologic cure did not differ significantly between continuous and pulse regimens for terbinafine and itraconazole. Compared with placebo, the most successful treatments were continuous terbinafine 250 mg daily for 24 weeks (risk ratio of achieving mycologic cure, 11.0) and continuous terbinafine 250 mg daily for 16 weeks (RR, 8.90). The researchers also observed no significant differences in the likelihood of adverse events between any continuous and pulse regimens of terbinafine, itraconazole, and fluconazole.
“Although continuous terbinafine 250 mg for 24 weeks was significantly more likely to produce mycologic cure than continuous itraconazole 200 mg for 12 weeks and weekly fluconazole (150-450 mg), it is not significantly different from the other included treatments,” Dr. Gupta and colleagues wrote in the abstract. “Considering the fungal life cycle, pulse therapy should theoretically be as effective as, or more effective than, continuous therapies: the sudden high concentration of an antifungal drug eliminates hyphae, sparing already-present spores. During the ‘off’ portion, these spores may germinate and be eliminated during the next pulse. Continuous therapy spares the spores, allowing them to germinate once treatment ends.”
They went on to note that, in clinical practice, “neither continuous nor pulse therapy is necessarily better. It is possible that the drug concentration in the nail is maintained during the ‘off’ period of pulse therapy. In both therapies, it may be that residual spores that have not been eliminated by the end of therapy are left to germinate, possibly contributing to the recalcitrant nature of onychomycosis.”
The study was awarded fourth place in the AAD’s 2020 poster awards. Dr. Gupta disclosed that he is a clinical trials investigator for Moberg Pharma and Bausch Health Canada and a speaker for Bausch Health Canada.
SOURCE: Gupta A et al. AAD 20, Abstract 16014.
There appear to be results from a systematic review and network meta-analysis showed.
“Previous meta-analyses of pulse and continuous therapies have generated ambiguous results,” study authors led by Aditya K. Gupta, MD, PhD, wrote in a poster abstract presented at the virtual annual meeting of the American Academy of Dermatology. “There are few head-to-head clinical studies and no meta-analyses comparing regimens of terbinafine to regimens of itraconazole.”
In what is believed to be the first study of its kind, Dr. Gupta, professor of dermatology at the University of Toronto, and colleagues used network meta-analysis to compare pulse and continuous systemic therapies for toenail onychomycosis. They used PubMed to search for randomized, controlled trials of oral antifungal treatments for the condition in patients aged 18 years and older that included data on mycologic cure, complete cure, adverse events, and dropout rates. Treatment effects were based on intention-to-treat cure rates, and the researchers excluded studies of ketoconazole and griseofulvin since they are no longer indicated for the condition.
For their network meta-analysis, Dr. Gupta and colleagues evaluated 22 studies from 20 publications that included 4,205 randomized patients. Data on complete cure were excluded because of a lack of studies. When the researchers compared all treatments to placebo, the likelihood of mycologic cure did not differ significantly between continuous and pulse regimens for terbinafine and itraconazole. Compared with placebo, the most successful treatments were continuous terbinafine 250 mg daily for 24 weeks (risk ratio of achieving mycologic cure, 11.0) and continuous terbinafine 250 mg daily for 16 weeks (RR, 8.90). The researchers also observed no significant differences in the likelihood of adverse events between any continuous and pulse regimens of terbinafine, itraconazole, and fluconazole.
“Although continuous terbinafine 250 mg for 24 weeks was significantly more likely to produce mycologic cure than continuous itraconazole 200 mg for 12 weeks and weekly fluconazole (150-450 mg), it is not significantly different from the other included treatments,” Dr. Gupta and colleagues wrote in the abstract. “Considering the fungal life cycle, pulse therapy should theoretically be as effective as, or more effective than, continuous therapies: the sudden high concentration of an antifungal drug eliminates hyphae, sparing already-present spores. During the ‘off’ portion, these spores may germinate and be eliminated during the next pulse. Continuous therapy spares the spores, allowing them to germinate once treatment ends.”
They went on to note that, in clinical practice, “neither continuous nor pulse therapy is necessarily better. It is possible that the drug concentration in the nail is maintained during the ‘off’ period of pulse therapy. In both therapies, it may be that residual spores that have not been eliminated by the end of therapy are left to germinate, possibly contributing to the recalcitrant nature of onychomycosis.”
The study was awarded fourth place in the AAD’s 2020 poster awards. Dr. Gupta disclosed that he is a clinical trials investigator for Moberg Pharma and Bausch Health Canada and a speaker for Bausch Health Canada.
SOURCE: Gupta A et al. AAD 20, Abstract 16014.
FROM AAD 20
High-fat, high-sugar diet may promote adult acne
A diet higher in fat, sugar, and milk was associated with having acne in a cross-sectional study of approximately 24,000 adults in France.
Acne in adults has been associated with social, emotional, and psychological consequences similar to those found with chronic diseases such as asthma, arthritis, epilepsy, and diabetes, wrote Laetitia Penso, MSc, of the University of Paris in Bobigny, France, and colleagues.
Although acne patients may believe that eating certain foods exacerbates acne, data on the effects of nutrition on acne, including associations between acne and a high-glycemic diet, are limited and have produced conflicting results, they noted.
In their study, published in JAMA Dermatology, the researchers identified 24,452 adults who participated in the NutriNet-Santé study, an ongoing, web-based study in France. Approximately 75% of the participants were women, the average age was 57 years, and 46% reported past or current acne.
Participants responded to an 11-item questionnaire between November 2008 and July 2019. Questions were related to the occurrence and diagnosis of acne, as well as medical history. Based on their acne status, participants were identified as falling into the categories of never acne, past acne, or current acne, and their dietary intake was assessed at baseline and every 6 months using three nonconsecutive 24-hour dietary records for 2 weekdays and 1 weekend day.
In an analysis, after adjustment for confounders, current acne was significantly associated with consumption of fatty and sugary foods (per portion, adjusted odds ratio, 1.54; P = .01), as well as with consumption of sugary drinks (per glass, aOR, 1.18; P = .04) and milk (per glass, aOR, 1.12; P = .04). In addition, carbohydrate intake and saturated fatty acid intake were significantly associated with current acne (aOR, 1.43; P = .02; and aOR, 3.90; P = .048, respectively).
Three dietary patterns accounted for 42% of the total variability, the researchers said. A healthy pattern of higher fruit, vegetable, and fish intake accounted for 18%, a fatty and sugary pattern of higher fat and sugar intake (including chocolate) accounted for 13%, and an animal product and cereal pattern of higher intake of meat, milk, and refined cereals accounted for 11%, they explained.
“The results of our study appear to support the hypothesis that the Western diet (rich in animal products and fatty and sugary foods) is associated with the presence of acne in adulthood,” the researchers concluded. Possible explanations for the findings include the effects of a high glycemic-load diet on circulating IGF-1 and insulin, which ultimately increases both oxidative stress and inflammation that promotes the development of acne, they noted.
The study findings were limited by several factors including the use of relatively homogenous younger and female patient population and the reliance on self-reported acne, as well as the observational design, which did not allow for identification of direct, causal associations between diet and acne, the researchers noted. Larger studies are needed to examine the relationship between diet and adult acne to inform prevention and treatment, they wrote.
“Much of the previous literature on the role of diet in acne has focused on the association of milk consumption and high glycemic-load diet with acne,” John S. Barbieri, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, wrote in an accompanying editorial.
Dr. Barbieri acknowledged the inability to make causal associations given the study design and noted that dietary interventions should be implemented with caution because of the potential for other effects such as reduced calcium or vitamin D.
“Nevertheless, given the potential overall health benefits of a healthy or low glycemic-load diet, and 2 small trials supporting its effectiveness in acne, a low glycemic-load diet is a reasonable recommendation for patients looking for dietary modifications that may improve their acne,” he said.
Dr. Barbieri said that he was encouraged to see that the study findings reflected previous research identifying an association between acne and high-glycemic load foods, as well as milk consumption, but he emphasized that more research is needed before general recommendations about diet and acne can be made.
“Trials are needed to evaluate whether dietary interventions can improve or prevent acne and how the effect size of such interventions compares with other standard treatment modalities,” he emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Barbieri disclosed support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health and from a Pfizer Fellowship grant to the Trustees of the University of Pennsylvania.
SOURCE: Penso L et al. JAMA Dermatol. 2020 June 10. doi: 10.1001/jamadermatol.2020.1602.
A diet higher in fat, sugar, and milk was associated with having acne in a cross-sectional study of approximately 24,000 adults in France.
Acne in adults has been associated with social, emotional, and psychological consequences similar to those found with chronic diseases such as asthma, arthritis, epilepsy, and diabetes, wrote Laetitia Penso, MSc, of the University of Paris in Bobigny, France, and colleagues.
Although acne patients may believe that eating certain foods exacerbates acne, data on the effects of nutrition on acne, including associations between acne and a high-glycemic diet, are limited and have produced conflicting results, they noted.
In their study, published in JAMA Dermatology, the researchers identified 24,452 adults who participated in the NutriNet-Santé study, an ongoing, web-based study in France. Approximately 75% of the participants were women, the average age was 57 years, and 46% reported past or current acne.
Participants responded to an 11-item questionnaire between November 2008 and July 2019. Questions were related to the occurrence and diagnosis of acne, as well as medical history. Based on their acne status, participants were identified as falling into the categories of never acne, past acne, or current acne, and their dietary intake was assessed at baseline and every 6 months using three nonconsecutive 24-hour dietary records for 2 weekdays and 1 weekend day.
In an analysis, after adjustment for confounders, current acne was significantly associated with consumption of fatty and sugary foods (per portion, adjusted odds ratio, 1.54; P = .01), as well as with consumption of sugary drinks (per glass, aOR, 1.18; P = .04) and milk (per glass, aOR, 1.12; P = .04). In addition, carbohydrate intake and saturated fatty acid intake were significantly associated with current acne (aOR, 1.43; P = .02; and aOR, 3.90; P = .048, respectively).
Three dietary patterns accounted for 42% of the total variability, the researchers said. A healthy pattern of higher fruit, vegetable, and fish intake accounted for 18%, a fatty and sugary pattern of higher fat and sugar intake (including chocolate) accounted for 13%, and an animal product and cereal pattern of higher intake of meat, milk, and refined cereals accounted for 11%, they explained.
“The results of our study appear to support the hypothesis that the Western diet (rich in animal products and fatty and sugary foods) is associated with the presence of acne in adulthood,” the researchers concluded. Possible explanations for the findings include the effects of a high glycemic-load diet on circulating IGF-1 and insulin, which ultimately increases both oxidative stress and inflammation that promotes the development of acne, they noted.
The study findings were limited by several factors including the use of relatively homogenous younger and female patient population and the reliance on self-reported acne, as well as the observational design, which did not allow for identification of direct, causal associations between diet and acne, the researchers noted. Larger studies are needed to examine the relationship between diet and adult acne to inform prevention and treatment, they wrote.
“Much of the previous literature on the role of diet in acne has focused on the association of milk consumption and high glycemic-load diet with acne,” John S. Barbieri, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, wrote in an accompanying editorial.
Dr. Barbieri acknowledged the inability to make causal associations given the study design and noted that dietary interventions should be implemented with caution because of the potential for other effects such as reduced calcium or vitamin D.
“Nevertheless, given the potential overall health benefits of a healthy or low glycemic-load diet, and 2 small trials supporting its effectiveness in acne, a low glycemic-load diet is a reasonable recommendation for patients looking for dietary modifications that may improve their acne,” he said.
Dr. Barbieri said that he was encouraged to see that the study findings reflected previous research identifying an association between acne and high-glycemic load foods, as well as milk consumption, but he emphasized that more research is needed before general recommendations about diet and acne can be made.
“Trials are needed to evaluate whether dietary interventions can improve or prevent acne and how the effect size of such interventions compares with other standard treatment modalities,” he emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Barbieri disclosed support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health and from a Pfizer Fellowship grant to the Trustees of the University of Pennsylvania.
SOURCE: Penso L et al. JAMA Dermatol. 2020 June 10. doi: 10.1001/jamadermatol.2020.1602.
A diet higher in fat, sugar, and milk was associated with having acne in a cross-sectional study of approximately 24,000 adults in France.
Acne in adults has been associated with social, emotional, and psychological consequences similar to those found with chronic diseases such as asthma, arthritis, epilepsy, and diabetes, wrote Laetitia Penso, MSc, of the University of Paris in Bobigny, France, and colleagues.
Although acne patients may believe that eating certain foods exacerbates acne, data on the effects of nutrition on acne, including associations between acne and a high-glycemic diet, are limited and have produced conflicting results, they noted.
In their study, published in JAMA Dermatology, the researchers identified 24,452 adults who participated in the NutriNet-Santé study, an ongoing, web-based study in France. Approximately 75% of the participants were women, the average age was 57 years, and 46% reported past or current acne.
Participants responded to an 11-item questionnaire between November 2008 and July 2019. Questions were related to the occurrence and diagnosis of acne, as well as medical history. Based on their acne status, participants were identified as falling into the categories of never acne, past acne, or current acne, and their dietary intake was assessed at baseline and every 6 months using three nonconsecutive 24-hour dietary records for 2 weekdays and 1 weekend day.
In an analysis, after adjustment for confounders, current acne was significantly associated with consumption of fatty and sugary foods (per portion, adjusted odds ratio, 1.54; P = .01), as well as with consumption of sugary drinks (per glass, aOR, 1.18; P = .04) and milk (per glass, aOR, 1.12; P = .04). In addition, carbohydrate intake and saturated fatty acid intake were significantly associated with current acne (aOR, 1.43; P = .02; and aOR, 3.90; P = .048, respectively).
Three dietary patterns accounted for 42% of the total variability, the researchers said. A healthy pattern of higher fruit, vegetable, and fish intake accounted for 18%, a fatty and sugary pattern of higher fat and sugar intake (including chocolate) accounted for 13%, and an animal product and cereal pattern of higher intake of meat, milk, and refined cereals accounted for 11%, they explained.
“The results of our study appear to support the hypothesis that the Western diet (rich in animal products and fatty and sugary foods) is associated with the presence of acne in adulthood,” the researchers concluded. Possible explanations for the findings include the effects of a high glycemic-load diet on circulating IGF-1 and insulin, which ultimately increases both oxidative stress and inflammation that promotes the development of acne, they noted.
The study findings were limited by several factors including the use of relatively homogenous younger and female patient population and the reliance on self-reported acne, as well as the observational design, which did not allow for identification of direct, causal associations between diet and acne, the researchers noted. Larger studies are needed to examine the relationship between diet and adult acne to inform prevention and treatment, they wrote.
“Much of the previous literature on the role of diet in acne has focused on the association of milk consumption and high glycemic-load diet with acne,” John S. Barbieri, MD, of the department of dermatology at the University of Pennsylvania, Philadelphia, wrote in an accompanying editorial.
Dr. Barbieri acknowledged the inability to make causal associations given the study design and noted that dietary interventions should be implemented with caution because of the potential for other effects such as reduced calcium or vitamin D.
“Nevertheless, given the potential overall health benefits of a healthy or low glycemic-load diet, and 2 small trials supporting its effectiveness in acne, a low glycemic-load diet is a reasonable recommendation for patients looking for dietary modifications that may improve their acne,” he said.
Dr. Barbieri said that he was encouraged to see that the study findings reflected previous research identifying an association between acne and high-glycemic load foods, as well as milk consumption, but he emphasized that more research is needed before general recommendations about diet and acne can be made.
“Trials are needed to evaluate whether dietary interventions can improve or prevent acne and how the effect size of such interventions compares with other standard treatment modalities,” he emphasized.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Barbieri disclosed support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health and from a Pfizer Fellowship grant to the Trustees of the University of Pennsylvania.
SOURCE: Penso L et al. JAMA Dermatol. 2020 June 10. doi: 10.1001/jamadermatol.2020.1602.
FROM JAMA DERMATOLOGY
Tralokinumab found effective in phase 3 atopic dermatitis studies
presented at the virtual annual meeting of the American Academy of Dermatology.
Tralokinumab is a fully human monoclonal antibody which binds specifically to interleukin-13 and thereby prevents downstream IL-13 signaling. In contrast, dupilumab (Dupixent), at present the only approved biologic agent for AD, blocks both the IL-13 and IL-4 pathways.
Two of the pivotal phase 3 trials presented at AAD 2020 – ECZTRA 1 and ECZTRA 2 – were identically designed, randomized, double-blind, placebo-controlled, 52-week, multinational monotherapy studies including a collective 1,596 adults with moderate to severe AD. In contrast, ECZTRA 3 was a 380-patient, double-blind, randomized, 32-week study of tralokinumab in combination with a topical corticosteroid versus placebo injections plus a topical corticosteroid.
“I would say the take-home point of these trials is they are proof of principle that blocking just IL-13 can be an effective approach. The studies help us understand that IL-13 is an important driver cytokine for the disease,” Eric Simpson, MD, lead clinical investigator for ECZTRA 2, said in an interview.
In all three phase 3 trials, the primary endpoint was achievement of a clinical response as defined by an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) plus at least a 75% improvement in the Eczema Area and Severity Index score (EASI-75) at week 16. In ECZTRA 1 and 2, this was accomplished in 16% and 22% of patients on 300 mg of tralokinumab administered subcutaneously every 2 weeks, compared with 7% and 11% of placebo-treated controls.
Patients with a clinical response at week 16 were then rerandomized to tralokinumab either every other week or every 4 weeks or to placebo for an additional 36 weeks. At 52 weeks, 51% and 59% of patients in ECZTRA 1 and 2, respectively, who had a clinical response at week 16 maintained an IGA 0/1 response while on tralokinumab every 2 weeks, as did 39% and 45% of those switched to treatment every 4 weeks. Similarly, 60% and 56% of clinical responders at week 16 maintained an EASI-75 response at week 52 with tralokinumab every 2 weeks, as did 49% and 51% of those rerandomized to treatment every 4 weeks.
The safety profile of tralokinumab in the two monotherapy trials was comparable with placebo.
In the ECZTRA studies, tralokinumab achieved significant improvement at week 16 in secondary endpoints including itch, health-related quality of life, and severity and extent of skin lesions.
How does tralokinumab, with its narrower focus targeting a single cytokine, stack up against dupilumab, the dual IL-13/IL-4 inhibitor that’s transformed the treatment of patients with moderate or severe AD?
Dr. Simpson, who was also principal investigator in a pivotal phase 3 trial for dupilumab, emphasized that no firm conclusions can be drawn because there have been no head-to-head comparative trials and the tralokinumab and dupilumab trials had different patient populations, geographic locations, and washout periods. With those caveats, however, he commented that, “just on the surface, numerically, for the monotherapy studies, dupilumab hit some higher targets than tralokinumab in terms of the percentage of patients clear or almost clear.”
In terms of safety, it appears that the risk of conjunctivitis may be lower with tralokinumab than dupilumab, with rates of 7% and 3% through 52 weeks in ECZTRA 1 and 2, respectively, versus 2% with placebo, although again this is “a caveated conclusion,” said Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
Tralokinumab combination therapy in ECZTRA 3
At 16 weeks, 39% of patients treated with tralokinumab plus topical corticosteroids had an IGA of 0/1 and 56% had an EASI-75 response, compared with 26% and 36% of patients on topical corticosteroids plus biweekly placebo injections. More than 90% of patients with a good clinical response at week 16 maintained that response at week 32 while on tralokinumab biweekly plus topical steroids. Among good responders at week 16 who were rerandomized to 300 mg of tralokinumab every 4 weeks plus topical steroids, 78% still had an IGA of 0/1 at week 32, and 91% had an EASI-75, reported Jonathan I. Silverberg, MD, PhD, director of clinical research and contact dermatitis at George Washington University, Washington.
A randomized, placebo-controlled combination therapy study such as this provides information that’s especially useful in clinical practice, Dr. Simpson observed.
“When I’m talking to patients about any biologics or oral therapies, I usually quote the figures from the combination therapy studies because the vast majority of our patients are using topical therapy in addition to systemics,” he said in the interview.
Asked how he envisions tralokinumab’s role in clinical practice, should the drug receive regulatory approval, Dr. Simpson said that he welcomes the prospect of having an additional treatment option to discuss with patients. Tralokinumab could be considered either as first-line therapy in patients who are failing on topical therapy or for patients who don’t respond adequately to or experience limiting side effects on dupilumab.
“There isn’t any established, published treatment algorithm in atopic dermatitis, probably for good reason, since we don’t have data to tell us you should start here and then move there. Those are long, difficult studies to perform,” Dr. Simpson said.
LEO Pharma has announced that it has applied for marketing approval for tralokinumab to the European Medicines Agency and plans to do so with the Food and Drug Administration by year’s end.
Dr. Simpson reported receiving research grants from and serving as a consultant to LEO Pharma, sponsor of the ECZTRA trials. He has similar financial relationships with close to a dozen other pharmaceutical companies.
presented at the virtual annual meeting of the American Academy of Dermatology.
Tralokinumab is a fully human monoclonal antibody which binds specifically to interleukin-13 and thereby prevents downstream IL-13 signaling. In contrast, dupilumab (Dupixent), at present the only approved biologic agent for AD, blocks both the IL-13 and IL-4 pathways.
Two of the pivotal phase 3 trials presented at AAD 2020 – ECZTRA 1 and ECZTRA 2 – were identically designed, randomized, double-blind, placebo-controlled, 52-week, multinational monotherapy studies including a collective 1,596 adults with moderate to severe AD. In contrast, ECZTRA 3 was a 380-patient, double-blind, randomized, 32-week study of tralokinumab in combination with a topical corticosteroid versus placebo injections plus a topical corticosteroid.
“I would say the take-home point of these trials is they are proof of principle that blocking just IL-13 can be an effective approach. The studies help us understand that IL-13 is an important driver cytokine for the disease,” Eric Simpson, MD, lead clinical investigator for ECZTRA 2, said in an interview.
In all three phase 3 trials, the primary endpoint was achievement of a clinical response as defined by an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) plus at least a 75% improvement in the Eczema Area and Severity Index score (EASI-75) at week 16. In ECZTRA 1 and 2, this was accomplished in 16% and 22% of patients on 300 mg of tralokinumab administered subcutaneously every 2 weeks, compared with 7% and 11% of placebo-treated controls.
Patients with a clinical response at week 16 were then rerandomized to tralokinumab either every other week or every 4 weeks or to placebo for an additional 36 weeks. At 52 weeks, 51% and 59% of patients in ECZTRA 1 and 2, respectively, who had a clinical response at week 16 maintained an IGA 0/1 response while on tralokinumab every 2 weeks, as did 39% and 45% of those switched to treatment every 4 weeks. Similarly, 60% and 56% of clinical responders at week 16 maintained an EASI-75 response at week 52 with tralokinumab every 2 weeks, as did 49% and 51% of those rerandomized to treatment every 4 weeks.
The safety profile of tralokinumab in the two monotherapy trials was comparable with placebo.
In the ECZTRA studies, tralokinumab achieved significant improvement at week 16 in secondary endpoints including itch, health-related quality of life, and severity and extent of skin lesions.
How does tralokinumab, with its narrower focus targeting a single cytokine, stack up against dupilumab, the dual IL-13/IL-4 inhibitor that’s transformed the treatment of patients with moderate or severe AD?
Dr. Simpson, who was also principal investigator in a pivotal phase 3 trial for dupilumab, emphasized that no firm conclusions can be drawn because there have been no head-to-head comparative trials and the tralokinumab and dupilumab trials had different patient populations, geographic locations, and washout periods. With those caveats, however, he commented that, “just on the surface, numerically, for the monotherapy studies, dupilumab hit some higher targets than tralokinumab in terms of the percentage of patients clear or almost clear.”
In terms of safety, it appears that the risk of conjunctivitis may be lower with tralokinumab than dupilumab, with rates of 7% and 3% through 52 weeks in ECZTRA 1 and 2, respectively, versus 2% with placebo, although again this is “a caveated conclusion,” said Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
Tralokinumab combination therapy in ECZTRA 3
At 16 weeks, 39% of patients treated with tralokinumab plus topical corticosteroids had an IGA of 0/1 and 56% had an EASI-75 response, compared with 26% and 36% of patients on topical corticosteroids plus biweekly placebo injections. More than 90% of patients with a good clinical response at week 16 maintained that response at week 32 while on tralokinumab biweekly plus topical steroids. Among good responders at week 16 who were rerandomized to 300 mg of tralokinumab every 4 weeks plus topical steroids, 78% still had an IGA of 0/1 at week 32, and 91% had an EASI-75, reported Jonathan I. Silverberg, MD, PhD, director of clinical research and contact dermatitis at George Washington University, Washington.
A randomized, placebo-controlled combination therapy study such as this provides information that’s especially useful in clinical practice, Dr. Simpson observed.
“When I’m talking to patients about any biologics or oral therapies, I usually quote the figures from the combination therapy studies because the vast majority of our patients are using topical therapy in addition to systemics,” he said in the interview.
Asked how he envisions tralokinumab’s role in clinical practice, should the drug receive regulatory approval, Dr. Simpson said that he welcomes the prospect of having an additional treatment option to discuss with patients. Tralokinumab could be considered either as first-line therapy in patients who are failing on topical therapy or for patients who don’t respond adequately to or experience limiting side effects on dupilumab.
“There isn’t any established, published treatment algorithm in atopic dermatitis, probably for good reason, since we don’t have data to tell us you should start here and then move there. Those are long, difficult studies to perform,” Dr. Simpson said.
LEO Pharma has announced that it has applied for marketing approval for tralokinumab to the European Medicines Agency and plans to do so with the Food and Drug Administration by year’s end.
Dr. Simpson reported receiving research grants from and serving as a consultant to LEO Pharma, sponsor of the ECZTRA trials. He has similar financial relationships with close to a dozen other pharmaceutical companies.
presented at the virtual annual meeting of the American Academy of Dermatology.
Tralokinumab is a fully human monoclonal antibody which binds specifically to interleukin-13 and thereby prevents downstream IL-13 signaling. In contrast, dupilumab (Dupixent), at present the only approved biologic agent for AD, blocks both the IL-13 and IL-4 pathways.
Two of the pivotal phase 3 trials presented at AAD 2020 – ECZTRA 1 and ECZTRA 2 – were identically designed, randomized, double-blind, placebo-controlled, 52-week, multinational monotherapy studies including a collective 1,596 adults with moderate to severe AD. In contrast, ECZTRA 3 was a 380-patient, double-blind, randomized, 32-week study of tralokinumab in combination with a topical corticosteroid versus placebo injections plus a topical corticosteroid.
“I would say the take-home point of these trials is they are proof of principle that blocking just IL-13 can be an effective approach. The studies help us understand that IL-13 is an important driver cytokine for the disease,” Eric Simpson, MD, lead clinical investigator for ECZTRA 2, said in an interview.
In all three phase 3 trials, the primary endpoint was achievement of a clinical response as defined by an Investigator Global Assessment score of clear or almost clear skin (IGA 0/1) plus at least a 75% improvement in the Eczema Area and Severity Index score (EASI-75) at week 16. In ECZTRA 1 and 2, this was accomplished in 16% and 22% of patients on 300 mg of tralokinumab administered subcutaneously every 2 weeks, compared with 7% and 11% of placebo-treated controls.
Patients with a clinical response at week 16 were then rerandomized to tralokinumab either every other week or every 4 weeks or to placebo for an additional 36 weeks. At 52 weeks, 51% and 59% of patients in ECZTRA 1 and 2, respectively, who had a clinical response at week 16 maintained an IGA 0/1 response while on tralokinumab every 2 weeks, as did 39% and 45% of those switched to treatment every 4 weeks. Similarly, 60% and 56% of clinical responders at week 16 maintained an EASI-75 response at week 52 with tralokinumab every 2 weeks, as did 49% and 51% of those rerandomized to treatment every 4 weeks.
The safety profile of tralokinumab in the two monotherapy trials was comparable with placebo.
In the ECZTRA studies, tralokinumab achieved significant improvement at week 16 in secondary endpoints including itch, health-related quality of life, and severity and extent of skin lesions.
How does tralokinumab, with its narrower focus targeting a single cytokine, stack up against dupilumab, the dual IL-13/IL-4 inhibitor that’s transformed the treatment of patients with moderate or severe AD?
Dr. Simpson, who was also principal investigator in a pivotal phase 3 trial for dupilumab, emphasized that no firm conclusions can be drawn because there have been no head-to-head comparative trials and the tralokinumab and dupilumab trials had different patient populations, geographic locations, and washout periods. With those caveats, however, he commented that, “just on the surface, numerically, for the monotherapy studies, dupilumab hit some higher targets than tralokinumab in terms of the percentage of patients clear or almost clear.”
In terms of safety, it appears that the risk of conjunctivitis may be lower with tralokinumab than dupilumab, with rates of 7% and 3% through 52 weeks in ECZTRA 1 and 2, respectively, versus 2% with placebo, although again this is “a caveated conclusion,” said Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
Tralokinumab combination therapy in ECZTRA 3
At 16 weeks, 39% of patients treated with tralokinumab plus topical corticosteroids had an IGA of 0/1 and 56% had an EASI-75 response, compared with 26% and 36% of patients on topical corticosteroids plus biweekly placebo injections. More than 90% of patients with a good clinical response at week 16 maintained that response at week 32 while on tralokinumab biweekly plus topical steroids. Among good responders at week 16 who were rerandomized to 300 mg of tralokinumab every 4 weeks plus topical steroids, 78% still had an IGA of 0/1 at week 32, and 91% had an EASI-75, reported Jonathan I. Silverberg, MD, PhD, director of clinical research and contact dermatitis at George Washington University, Washington.
A randomized, placebo-controlled combination therapy study such as this provides information that’s especially useful in clinical practice, Dr. Simpson observed.
“When I’m talking to patients about any biologics or oral therapies, I usually quote the figures from the combination therapy studies because the vast majority of our patients are using topical therapy in addition to systemics,” he said in the interview.
Asked how he envisions tralokinumab’s role in clinical practice, should the drug receive regulatory approval, Dr. Simpson said that he welcomes the prospect of having an additional treatment option to discuss with patients. Tralokinumab could be considered either as first-line therapy in patients who are failing on topical therapy or for patients who don’t respond adequately to or experience limiting side effects on dupilumab.
“There isn’t any established, published treatment algorithm in atopic dermatitis, probably for good reason, since we don’t have data to tell us you should start here and then move there. Those are long, difficult studies to perform,” Dr. Simpson said.
LEO Pharma has announced that it has applied for marketing approval for tralokinumab to the European Medicines Agency and plans to do so with the Food and Drug Administration by year’s end.
Dr. Simpson reported receiving research grants from and serving as a consultant to LEO Pharma, sponsor of the ECZTRA trials. He has similar financial relationships with close to a dozen other pharmaceutical companies.
FROM AAD 2020