User login
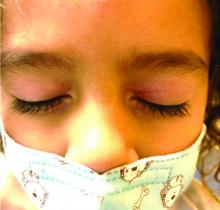
Four-year-old boy presents with itchy rash on face, extremities
Contact dermatitis is an eczematous, pruritic eruption caused by direct contact with a substance and an irritant or allergic reaction. While it may not be contagious or life-threatening, contact dermatitis may be tremendously uncomfortable and impactful. Contact dermatitis may occur from exposure to chemicals in soaps, shampoos, cosmetics, metals, plants and topical products, and medications. The hallmark of contact dermatitis is localized eczematous reactions on the portion of the body that has been directly exposed to the reaction-causing substance. – often with oozing and crusting.
Irritant contact dermatitis is the most common type, which occurs when a substance damages the skin’s outer protective layer and does not require prior exposure or sensitization. Allergic contact dermatitis (ACD) can develop after exposure and sensitization, with an external allergen triggering an acute inflammatory response.1 Common causes of ACD include nickel, cobalt, gold, chromium, poison ivy/oak/sumac, cosmetics/personal care products that contain formaldehyde, fragrances, topical medications (anesthetics, antibiotics, corticosteroids), baby wipes, sunscreens, latex materials, protective equipment, soap/cleansers, resins, and acrylics. Among children, nickel sulfate, ammonium persulfate, gold sodium thiosulfate, thimerosal, and toluene-2,5-diamine are the most common sensitizers. Rarely, ACD can be triggered by something that enters the body through foods, flavorings, medicine, or medical or dental procedures (systemic contact dermatitis).
An Id reaction, or autoeczematization, is a generalized acute cutaneous reaction to a variety of stimuli, including infectious and inflammatory skin conditions such as contact dermatitis, stasis dermatitis, or other eczematous dermatitis.3 Id reactions usually are preceded by a preexisting dermatitis. Lesions are, by definition, at a site distant from the primary infection or dermatitis. They often are distributed symmetrically. Papular or papular-vesicular lesions of the extremities and or trunk are common in children.
Our patient had evidence of a localized periocular contact dermatitis reaction that preceded the symmetric papular, eczematous eruption consistent with an id reaction. Our patient was prescribed hydrocortisone 2.5 % ointment for the eyes and triamcinolone 0.1% ointment for the rash on the body, which resulted in significant improvement.
Rosacea is a chronic and relapsing inflammatory skin disorder that primarily involves the central face. Common clinical features include facial erythema, telangiectasias, and inflammatory papules or pustules. Ocular involvement may occur in the presence or absence of cutaneous manifestations. Patients may report the presence of ocular foreign body sensation, burning, photophobia, blurred vision, redness, and tearing. Ocular disease is usually bilateral and is not proportional to the severity of the skin disease.4 Common skin findings are blepharitis, lid margin telangiectasia, tear abnormalities, meibomian gland inflammation, frequent chalazion, bilateral hordeolum, conjunctivitis, and, rarely, corneal ulcers and vascularization. Our patient initially did have bilateral hordeolum in what may seem to be ocular rosacea. However, given the use of a recent topical antibiotic with subsequent eczematous rash of the eyelids and then resulting distant rash on the body 1week later made the rash likely allergic contact dermatitis with id reaction.
Seborrheic dermatitis is a chronic, relapsing, and usually mild form of dermatitis that occurs in infants and in adults. The severity may vary from minimal, asymptomatic scaliness of the scalp (dandruff) to more widespread involvement. It is usually characterized by well-demarcated, erythematous plaques with greasy-looking, yellowish scales distributed on areas rich in sebaceous glands, such as the scalp, the external ear, the center of the face, the upper part of the trunk, and the intertriginous areas.
Psoriasis typically affects the outside of the elbows, knees, or scalp, although it can appear on any location. It tends to go through cycles, flaring for a few weeks or months, then subsiding for a while or going into remission. Ocular involvement is a well known manifestation of psoriasis.5 Psoriatic lesions of the eyelid are rare, even in the erythrodermic variant of the disease. Occasionally, pustular psoriasis may involve the eyelids, with typical psoriatic lesions visible on the skin and lid margin. The reason for the relative sparing of the eyelid skin in patients with psoriasis is unknown. Other manifestations include meibomian gland dysfunction, decreased tear film break-up time, a nonspecific conjunctivitis, and corneal disease secondary to lid disease such as trichiasis.
Gianotti-Crosti syndrome (GCS), also known as papular acrodermatitis, papular acrodermatitis of childhood, and infantile papular acrodermatitis, is a self-limited skin disorder that most often occurs in young children. Viral infections are common GCS precipitating factors . GCS typically manifests as a symmetric, papular eruption, often with larger (3- to 10-mm) flat topped papulovesicles. Classic sites of involvement include the cheeks, buttocks, and extensor surfaces of the forearms and legs. GCS may be pruritic or asymptomatic, and papules typically resolve spontaneously within 2 months. Occasionally, GCS persists for longer periods. The eyelid lesions and localized pattern, with the absence of larger symmetric papules of the buttocks and legs, was not consistent with papular acrodermatitis of childhood.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. They had no conflicts of interest to disclose. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol 2016 Jun; 74(6):1043-54.
2. Pediatr Dermatol 2016 Jul; 33(4):399-404.
3. Evans M & Bronson D. (2019) Id Reaction (Autoeczematization). Retrieved from emedicine.medscape.com/article/1049760-overview.
4. Curr Opin Ophthalmol. 2004 Dec;15(6):499-502.
5. Clin Dermatol. Mar-Apr 2016;34(2):146-50.
Contact dermatitis is an eczematous, pruritic eruption caused by direct contact with a substance and an irritant or allergic reaction. While it may not be contagious or life-threatening, contact dermatitis may be tremendously uncomfortable and impactful. Contact dermatitis may occur from exposure to chemicals in soaps, shampoos, cosmetics, metals, plants and topical products, and medications. The hallmark of contact dermatitis is localized eczematous reactions on the portion of the body that has been directly exposed to the reaction-causing substance. – often with oozing and crusting.
Irritant contact dermatitis is the most common type, which occurs when a substance damages the skin’s outer protective layer and does not require prior exposure or sensitization. Allergic contact dermatitis (ACD) can develop after exposure and sensitization, with an external allergen triggering an acute inflammatory response.1 Common causes of ACD include nickel, cobalt, gold, chromium, poison ivy/oak/sumac, cosmetics/personal care products that contain formaldehyde, fragrances, topical medications (anesthetics, antibiotics, corticosteroids), baby wipes, sunscreens, latex materials, protective equipment, soap/cleansers, resins, and acrylics. Among children, nickel sulfate, ammonium persulfate, gold sodium thiosulfate, thimerosal, and toluene-2,5-diamine are the most common sensitizers. Rarely, ACD can be triggered by something that enters the body through foods, flavorings, medicine, or medical or dental procedures (systemic contact dermatitis).
An Id reaction, or autoeczematization, is a generalized acute cutaneous reaction to a variety of stimuli, including infectious and inflammatory skin conditions such as contact dermatitis, stasis dermatitis, or other eczematous dermatitis.3 Id reactions usually are preceded by a preexisting dermatitis. Lesions are, by definition, at a site distant from the primary infection or dermatitis. They often are distributed symmetrically. Papular or papular-vesicular lesions of the extremities and or trunk are common in children.
Our patient had evidence of a localized periocular contact dermatitis reaction that preceded the symmetric papular, eczematous eruption consistent with an id reaction. Our patient was prescribed hydrocortisone 2.5 % ointment for the eyes and triamcinolone 0.1% ointment for the rash on the body, which resulted in significant improvement.
Rosacea is a chronic and relapsing inflammatory skin disorder that primarily involves the central face. Common clinical features include facial erythema, telangiectasias, and inflammatory papules or pustules. Ocular involvement may occur in the presence or absence of cutaneous manifestations. Patients may report the presence of ocular foreign body sensation, burning, photophobia, blurred vision, redness, and tearing. Ocular disease is usually bilateral and is not proportional to the severity of the skin disease.4 Common skin findings are blepharitis, lid margin telangiectasia, tear abnormalities, meibomian gland inflammation, frequent chalazion, bilateral hordeolum, conjunctivitis, and, rarely, corneal ulcers and vascularization. Our patient initially did have bilateral hordeolum in what may seem to be ocular rosacea. However, given the use of a recent topical antibiotic with subsequent eczematous rash of the eyelids and then resulting distant rash on the body 1week later made the rash likely allergic contact dermatitis with id reaction.
Seborrheic dermatitis is a chronic, relapsing, and usually mild form of dermatitis that occurs in infants and in adults. The severity may vary from minimal, asymptomatic scaliness of the scalp (dandruff) to more widespread involvement. It is usually characterized by well-demarcated, erythematous plaques with greasy-looking, yellowish scales distributed on areas rich in sebaceous glands, such as the scalp, the external ear, the center of the face, the upper part of the trunk, and the intertriginous areas.
Psoriasis typically affects the outside of the elbows, knees, or scalp, although it can appear on any location. It tends to go through cycles, flaring for a few weeks or months, then subsiding for a while or going into remission. Ocular involvement is a well known manifestation of psoriasis.5 Psoriatic lesions of the eyelid are rare, even in the erythrodermic variant of the disease. Occasionally, pustular psoriasis may involve the eyelids, with typical psoriatic lesions visible on the skin and lid margin. The reason for the relative sparing of the eyelid skin in patients with psoriasis is unknown. Other manifestations include meibomian gland dysfunction, decreased tear film break-up time, a nonspecific conjunctivitis, and corneal disease secondary to lid disease such as trichiasis.
Gianotti-Crosti syndrome (GCS), also known as papular acrodermatitis, papular acrodermatitis of childhood, and infantile papular acrodermatitis, is a self-limited skin disorder that most often occurs in young children. Viral infections are common GCS precipitating factors . GCS typically manifests as a symmetric, papular eruption, often with larger (3- to 10-mm) flat topped papulovesicles. Classic sites of involvement include the cheeks, buttocks, and extensor surfaces of the forearms and legs. GCS may be pruritic or asymptomatic, and papules typically resolve spontaneously within 2 months. Occasionally, GCS persists for longer periods. The eyelid lesions and localized pattern, with the absence of larger symmetric papules of the buttocks and legs, was not consistent with papular acrodermatitis of childhood.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. They had no conflicts of interest to disclose. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol 2016 Jun; 74(6):1043-54.
2. Pediatr Dermatol 2016 Jul; 33(4):399-404.
3. Evans M & Bronson D. (2019) Id Reaction (Autoeczematization). Retrieved from emedicine.medscape.com/article/1049760-overview.
4. Curr Opin Ophthalmol. 2004 Dec;15(6):499-502.
5. Clin Dermatol. Mar-Apr 2016;34(2):146-50.
Contact dermatitis is an eczematous, pruritic eruption caused by direct contact with a substance and an irritant or allergic reaction. While it may not be contagious or life-threatening, contact dermatitis may be tremendously uncomfortable and impactful. Contact dermatitis may occur from exposure to chemicals in soaps, shampoos, cosmetics, metals, plants and topical products, and medications. The hallmark of contact dermatitis is localized eczematous reactions on the portion of the body that has been directly exposed to the reaction-causing substance. – often with oozing and crusting.
Irritant contact dermatitis is the most common type, which occurs when a substance damages the skin’s outer protective layer and does not require prior exposure or sensitization. Allergic contact dermatitis (ACD) can develop after exposure and sensitization, with an external allergen triggering an acute inflammatory response.1 Common causes of ACD include nickel, cobalt, gold, chromium, poison ivy/oak/sumac, cosmetics/personal care products that contain formaldehyde, fragrances, topical medications (anesthetics, antibiotics, corticosteroids), baby wipes, sunscreens, latex materials, protective equipment, soap/cleansers, resins, and acrylics. Among children, nickel sulfate, ammonium persulfate, gold sodium thiosulfate, thimerosal, and toluene-2,5-diamine are the most common sensitizers. Rarely, ACD can be triggered by something that enters the body through foods, flavorings, medicine, or medical or dental procedures (systemic contact dermatitis).
An Id reaction, or autoeczematization, is a generalized acute cutaneous reaction to a variety of stimuli, including infectious and inflammatory skin conditions such as contact dermatitis, stasis dermatitis, or other eczematous dermatitis.3 Id reactions usually are preceded by a preexisting dermatitis. Lesions are, by definition, at a site distant from the primary infection or dermatitis. They often are distributed symmetrically. Papular or papular-vesicular lesions of the extremities and or trunk are common in children.
Our patient had evidence of a localized periocular contact dermatitis reaction that preceded the symmetric papular, eczematous eruption consistent with an id reaction. Our patient was prescribed hydrocortisone 2.5 % ointment for the eyes and triamcinolone 0.1% ointment for the rash on the body, which resulted in significant improvement.
Rosacea is a chronic and relapsing inflammatory skin disorder that primarily involves the central face. Common clinical features include facial erythema, telangiectasias, and inflammatory papules or pustules. Ocular involvement may occur in the presence or absence of cutaneous manifestations. Patients may report the presence of ocular foreign body sensation, burning, photophobia, blurred vision, redness, and tearing. Ocular disease is usually bilateral and is not proportional to the severity of the skin disease.4 Common skin findings are blepharitis, lid margin telangiectasia, tear abnormalities, meibomian gland inflammation, frequent chalazion, bilateral hordeolum, conjunctivitis, and, rarely, corneal ulcers and vascularization. Our patient initially did have bilateral hordeolum in what may seem to be ocular rosacea. However, given the use of a recent topical antibiotic with subsequent eczematous rash of the eyelids and then resulting distant rash on the body 1week later made the rash likely allergic contact dermatitis with id reaction.
Seborrheic dermatitis is a chronic, relapsing, and usually mild form of dermatitis that occurs in infants and in adults. The severity may vary from minimal, asymptomatic scaliness of the scalp (dandruff) to more widespread involvement. It is usually characterized by well-demarcated, erythematous plaques with greasy-looking, yellowish scales distributed on areas rich in sebaceous glands, such as the scalp, the external ear, the center of the face, the upper part of the trunk, and the intertriginous areas.
Psoriasis typically affects the outside of the elbows, knees, or scalp, although it can appear on any location. It tends to go through cycles, flaring for a few weeks or months, then subsiding for a while or going into remission. Ocular involvement is a well known manifestation of psoriasis.5 Psoriatic lesions of the eyelid are rare, even in the erythrodermic variant of the disease. Occasionally, pustular psoriasis may involve the eyelids, with typical psoriatic lesions visible on the skin and lid margin. The reason for the relative sparing of the eyelid skin in patients with psoriasis is unknown. Other manifestations include meibomian gland dysfunction, decreased tear film break-up time, a nonspecific conjunctivitis, and corneal disease secondary to lid disease such as trichiasis.
Gianotti-Crosti syndrome (GCS), also known as papular acrodermatitis, papular acrodermatitis of childhood, and infantile papular acrodermatitis, is a self-limited skin disorder that most often occurs in young children. Viral infections are common GCS precipitating factors . GCS typically manifests as a symmetric, papular eruption, often with larger (3- to 10-mm) flat topped papulovesicles. Classic sites of involvement include the cheeks, buttocks, and extensor surfaces of the forearms and legs. GCS may be pruritic or asymptomatic, and papules typically resolve spontaneously within 2 months. Occasionally, GCS persists for longer periods. The eyelid lesions and localized pattern, with the absence of larger symmetric papules of the buttocks and legs, was not consistent with papular acrodermatitis of childhood.
Dr. Bhatti is a research fellow in pediatric dermatology at Rady Children’s Hospital and the University of California, San Diego. Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children’s Hospital–San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. They had no conflicts of interest to disclose. Email them at pdnews@mdedge.com.
References
1. J Am Acad Dermatol 2016 Jun; 74(6):1043-54.
2. Pediatr Dermatol 2016 Jul; 33(4):399-404.
3. Evans M & Bronson D. (2019) Id Reaction (Autoeczematization). Retrieved from emedicine.medscape.com/article/1049760-overview.
4. Curr Opin Ophthalmol. 2004 Dec;15(6):499-502.
5. Clin Dermatol. Mar-Apr 2016;34(2):146-50.
A 4-year-old healthy male with no significant prior medical history presents for evaluation of "itchy bumps" on the face and extremities of 2 weeks' duration.
The child was well until around 2 and a half weeks ago when he presented for evaluation of two lesions on the lower eyelids, diagnosed as hordeolum (a stye). He was prescribed ofloxacin ophthalmic solution.
One week later he developed bilateral itchy red eyes with red, thickened areas on the upper lids, followed several days later by pruritic papules on the ears, wrists, elbows, knees, and ankles. His mother used Vaseline for the eyelids for 1 week with no improvement. Physical exam at the dermatologist's office showed mild erythema, induration, and lichenification of the upper eyelids, and bilateral periocular eczematous patches with overlying scale. Subtle papules were evident on the elbows and feet.
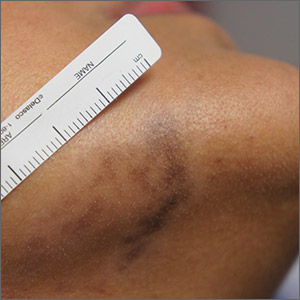
Jaw pigmentation
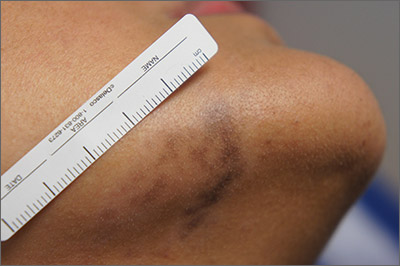
Exam and biopsy led to a diagnosis of erythema dyschromicum perstans. Notably, the punch biopsy was performed as superficially as possible to avoid injuring the marginal mandibular branch of the facial nerve. Histology showed focal vacuolar interface changes, pigmentary incontinence, and prominent dermal melanophages—consistent with erythema dyschromicum perstans.
Erythema dyschromicum perstans is an uncommon dermal macular hyperpigmentation that can affect patients of all ages. It is more often seen in patients with Fitzpatrick skin types III and IV. Some patients have a preceding inflammatory phase with associated erythema.
It is believed that erythema dyschromicum perstans may be a form of pigmented lichen planus with similar histologic changes. Genetic predisposition and medications, such as penicillamine and omeprazole, have been suggested as predisposing risk factors, although these risk factors are not always present. Similarly, cases of erythema dyschromicum perstans associated with human immunodeficiency virus and hepatitis C virus have been reported, but a causal link has not been established. Commonly affected sites include the face and neck, although patches on the trunk and extremities occur, as well.
The differential diagnosis includes lichen planus, discoid lupus, drug-induced hyperpigmentation, Hansen disease, and fixed drug eruption.
Erythema dyschromicum perstans is resistant to most therapies, yet it may clear spontaneously over the years. Therapies that have been successful in case reports include Q-switched ruby laser, topical steroids, UV therapy, isotretinoin, and clofazimine. This patient used desonide cream 0.05% bid for 3 weeks without improvement. She then used camouflage concealer for a year, at which point the area had faded almost completely.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Gutierrez D, Krueger LD, Tan A, et al. Proton pump inhibitor-induced erythema dyschromicum perstans-like pigmentation. JAAD Case Rep. 2019;5:701-703.

Exam and biopsy led to a diagnosis of erythema dyschromicum perstans. Notably, the punch biopsy was performed as superficially as possible to avoid injuring the marginal mandibular branch of the facial nerve. Histology showed focal vacuolar interface changes, pigmentary incontinence, and prominent dermal melanophages—consistent with erythema dyschromicum perstans.
Erythema dyschromicum perstans is an uncommon dermal macular hyperpigmentation that can affect patients of all ages. It is more often seen in patients with Fitzpatrick skin types III and IV. Some patients have a preceding inflammatory phase with associated erythema.
It is believed that erythema dyschromicum perstans may be a form of pigmented lichen planus with similar histologic changes. Genetic predisposition and medications, such as penicillamine and omeprazole, have been suggested as predisposing risk factors, although these risk factors are not always present. Similarly, cases of erythema dyschromicum perstans associated with human immunodeficiency virus and hepatitis C virus have been reported, but a causal link has not been established. Commonly affected sites include the face and neck, although patches on the trunk and extremities occur, as well.
The differential diagnosis includes lichen planus, discoid lupus, drug-induced hyperpigmentation, Hansen disease, and fixed drug eruption.
Erythema dyschromicum perstans is resistant to most therapies, yet it may clear spontaneously over the years. Therapies that have been successful in case reports include Q-switched ruby laser, topical steroids, UV therapy, isotretinoin, and clofazimine. This patient used desonide cream 0.05% bid for 3 weeks without improvement. She then used camouflage concealer for a year, at which point the area had faded almost completely.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Exam and biopsy led to a diagnosis of erythema dyschromicum perstans. Notably, the punch biopsy was performed as superficially as possible to avoid injuring the marginal mandibular branch of the facial nerve. Histology showed focal vacuolar interface changes, pigmentary incontinence, and prominent dermal melanophages—consistent with erythema dyschromicum perstans.
Erythema dyschromicum perstans is an uncommon dermal macular hyperpigmentation that can affect patients of all ages. It is more often seen in patients with Fitzpatrick skin types III and IV. Some patients have a preceding inflammatory phase with associated erythema.
It is believed that erythema dyschromicum perstans may be a form of pigmented lichen planus with similar histologic changes. Genetic predisposition and medications, such as penicillamine and omeprazole, have been suggested as predisposing risk factors, although these risk factors are not always present. Similarly, cases of erythema dyschromicum perstans associated with human immunodeficiency virus and hepatitis C virus have been reported, but a causal link has not been established. Commonly affected sites include the face and neck, although patches on the trunk and extremities occur, as well.
The differential diagnosis includes lichen planus, discoid lupus, drug-induced hyperpigmentation, Hansen disease, and fixed drug eruption.
Erythema dyschromicum perstans is resistant to most therapies, yet it may clear spontaneously over the years. Therapies that have been successful in case reports include Q-switched ruby laser, topical steroids, UV therapy, isotretinoin, and clofazimine. This patient used desonide cream 0.05% bid for 3 weeks without improvement. She then used camouflage concealer for a year, at which point the area had faded almost completely.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Gutierrez D, Krueger LD, Tan A, et al. Proton pump inhibitor-induced erythema dyschromicum perstans-like pigmentation. JAAD Case Rep. 2019;5:701-703.
Gutierrez D, Krueger LD, Tan A, et al. Proton pump inhibitor-induced erythema dyschromicum perstans-like pigmentation. JAAD Case Rep. 2019;5:701-703.
Be wary of ‘for eczema’ claims on labels of popular moisturizers
Be wary of “for eczema” advertising claims contained on the labels of popular skin moisturizers.
Results from a study presented during the virtual annual meeting of the Society for Pediatric Dermatology found that .
“Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products,” co-first study author Catherine L. Ludwig, said in an interview. “Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.”
For the current study, Ms. Ludwig, a medical student at the University of Illinois at Chicago and co-first author Alyssa M. Thompson, a medical student at the University of Arizona, Tucson, and their colleagues compiled a list of the top 30 moisturizers “for eczema” sold by Amazon, Target, and Walmart. For each moisturizer they recorded common ingredients and marketing claims related to benefits for atopic dermatitis, including eczema relief, sensitive/gentle skin, hypoallergenic, anti-itch, anti-inflammatory, clinically proven, oatmeal, dermatologist recommended/approved, organic, fragrance-free, for baby, or National Eczema Association approved. To establish allergenic potential, the researchers used MATLAB to compare ingredient lists to compounds listed as common allergens in the American Contact Dermatitis Society’s Contact Allergen Management Program database (ACDS CAMP). Next, they used the Mann-Whitney U test to evaluate differences in allergen count between products with and without specific marketing claims.
Ms. Ludwig and her associates found that 28 of 30 products analyzed (93%) contained at least one allergen, with an overall average allergen count of 3.60. The three most prevalent allergens were cetyl alcohol (70%), phenoxyethanol (50%), and aloe (33%). “Anti-inflammatory” moisturizers had the greatest average number of allergens (4.00), followed by “anti-itch” (3.71) and “oatmeal” (3.71). Only products claiming to be “hypoallergenic” had significantly lower allergenic ingredient count (an average of 2.45) than those without the claim (P = .011).
“It was validating to see that eczema moisturizer products marketed as ‘hypoallergenic’ truly do have fewer allergenic ingredients than moisturizers without the claim,” Ms. Ludwig said. “However, it was surprising to see that even products marketed to eczema patients, who have a higher prevalence of allergic contact dermatitis, contain an average of 3.6 common allergens. As dermatology providers, we can relay to patients and parents that relying solely on ‘for eczema’ claims is not advisable. Clinicians should acquaint themselves with the top allergens (cetyl alcohol, phenoxyethanol, and aloe) and keep these ingredients, as well as affordability and patient preferences, in mind when making product recommendations.”
The study’s senior author, Vivian Y. Shi, MD, is a stock shareholder of Learn Health and has served as an advisory board member and/or investigator, and/or received research funding from AbbVie, Burt’s Bees, GpSkin, LEO Pharma, Eli Lilly, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Skin Actives Scientific, and SUN Pharma, and the Foundation for Atopic Dermatitis, Global Parents for Eczema Research, and the National Eczema Association. The other study authors reported having no financial disclosures.
Be wary of “for eczema” advertising claims contained on the labels of popular skin moisturizers.
Results from a study presented during the virtual annual meeting of the Society for Pediatric Dermatology found that .
“Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products,” co-first study author Catherine L. Ludwig, said in an interview. “Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.”
For the current study, Ms. Ludwig, a medical student at the University of Illinois at Chicago and co-first author Alyssa M. Thompson, a medical student at the University of Arizona, Tucson, and their colleagues compiled a list of the top 30 moisturizers “for eczema” sold by Amazon, Target, and Walmart. For each moisturizer they recorded common ingredients and marketing claims related to benefits for atopic dermatitis, including eczema relief, sensitive/gentle skin, hypoallergenic, anti-itch, anti-inflammatory, clinically proven, oatmeal, dermatologist recommended/approved, organic, fragrance-free, for baby, or National Eczema Association approved. To establish allergenic potential, the researchers used MATLAB to compare ingredient lists to compounds listed as common allergens in the American Contact Dermatitis Society’s Contact Allergen Management Program database (ACDS CAMP). Next, they used the Mann-Whitney U test to evaluate differences in allergen count between products with and without specific marketing claims.
Ms. Ludwig and her associates found that 28 of 30 products analyzed (93%) contained at least one allergen, with an overall average allergen count of 3.60. The three most prevalent allergens were cetyl alcohol (70%), phenoxyethanol (50%), and aloe (33%). “Anti-inflammatory” moisturizers had the greatest average number of allergens (4.00), followed by “anti-itch” (3.71) and “oatmeal” (3.71). Only products claiming to be “hypoallergenic” had significantly lower allergenic ingredient count (an average of 2.45) than those without the claim (P = .011).
“It was validating to see that eczema moisturizer products marketed as ‘hypoallergenic’ truly do have fewer allergenic ingredients than moisturizers without the claim,” Ms. Ludwig said. “However, it was surprising to see that even products marketed to eczema patients, who have a higher prevalence of allergic contact dermatitis, contain an average of 3.6 common allergens. As dermatology providers, we can relay to patients and parents that relying solely on ‘for eczema’ claims is not advisable. Clinicians should acquaint themselves with the top allergens (cetyl alcohol, phenoxyethanol, and aloe) and keep these ingredients, as well as affordability and patient preferences, in mind when making product recommendations.”
The study’s senior author, Vivian Y. Shi, MD, is a stock shareholder of Learn Health and has served as an advisory board member and/or investigator, and/or received research funding from AbbVie, Burt’s Bees, GpSkin, LEO Pharma, Eli Lilly, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Skin Actives Scientific, and SUN Pharma, and the Foundation for Atopic Dermatitis, Global Parents for Eczema Research, and the National Eczema Association. The other study authors reported having no financial disclosures.
Be wary of “for eczema” advertising claims contained on the labels of popular skin moisturizers.
Results from a study presented during the virtual annual meeting of the Society for Pediatric Dermatology found that .
“Prescription medications are important for managing eczema flares, but a lot of the work in treating eczema is preventative, done by consistently moisturizing the skin at home with drug store products,” co-first study author Catherine L. Ludwig, said in an interview. “Allergic contact dermatitis occurs more commonly in people with eczema. A previous study was done in characterizing the allergenic potential of drug store moisturizers and found that 88% of moisturizers contain at least one common allergen. Many moisturizers are marketed specifically to eczema, but the allergen content of these products are unknown.”
For the current study, Ms. Ludwig, a medical student at the University of Illinois at Chicago and co-first author Alyssa M. Thompson, a medical student at the University of Arizona, Tucson, and their colleagues compiled a list of the top 30 moisturizers “for eczema” sold by Amazon, Target, and Walmart. For each moisturizer they recorded common ingredients and marketing claims related to benefits for atopic dermatitis, including eczema relief, sensitive/gentle skin, hypoallergenic, anti-itch, anti-inflammatory, clinically proven, oatmeal, dermatologist recommended/approved, organic, fragrance-free, for baby, or National Eczema Association approved. To establish allergenic potential, the researchers used MATLAB to compare ingredient lists to compounds listed as common allergens in the American Contact Dermatitis Society’s Contact Allergen Management Program database (ACDS CAMP). Next, they used the Mann-Whitney U test to evaluate differences in allergen count between products with and without specific marketing claims.
Ms. Ludwig and her associates found that 28 of 30 products analyzed (93%) contained at least one allergen, with an overall average allergen count of 3.60. The three most prevalent allergens were cetyl alcohol (70%), phenoxyethanol (50%), and aloe (33%). “Anti-inflammatory” moisturizers had the greatest average number of allergens (4.00), followed by “anti-itch” (3.71) and “oatmeal” (3.71). Only products claiming to be “hypoallergenic” had significantly lower allergenic ingredient count (an average of 2.45) than those without the claim (P = .011).
“It was validating to see that eczema moisturizer products marketed as ‘hypoallergenic’ truly do have fewer allergenic ingredients than moisturizers without the claim,” Ms. Ludwig said. “However, it was surprising to see that even products marketed to eczema patients, who have a higher prevalence of allergic contact dermatitis, contain an average of 3.6 common allergens. As dermatology providers, we can relay to patients and parents that relying solely on ‘for eczema’ claims is not advisable. Clinicians should acquaint themselves with the top allergens (cetyl alcohol, phenoxyethanol, and aloe) and keep these ingredients, as well as affordability and patient preferences, in mind when making product recommendations.”
The study’s senior author, Vivian Y. Shi, MD, is a stock shareholder of Learn Health and has served as an advisory board member and/or investigator, and/or received research funding from AbbVie, Burt’s Bees, GpSkin, LEO Pharma, Eli Lilly, Menlo Therapeutics, Novartis, Pfizer, Regeneron, Sanofi Genzyme, Skin Actives Scientific, and SUN Pharma, and the Foundation for Atopic Dermatitis, Global Parents for Eczema Research, and the National Eczema Association. The other study authors reported having no financial disclosures.
FROM SPD 2020
Topical PDE-4 inhibitor for psoriasis effective in phase 2b trial
Once-daily – including challenging lesions in tough-to-treat intertriginous areas – in a phase 2b, randomized, double-blind, vehicle-controlled clinical trial, Mark G. Lebwohl, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The clinical improvement occurred rapidly. And topical roflumilast’s side effect profile was essentially the same as in vehicle-treated controls, which suggests a potential major advantage for the novel drug in future clinical practice. After all, topical treatment is the mainstay of psoriasis therapy, but the current topical agents – high-potency corticosteroids, vitamin D derivatives, and retinoids – have long-term tolerability, efficacy, or side effect issues, especially in treating sensitive skin areas, including the face and intertriginous areas.
“Roflumilast cream could really be a game changer,” predicted Dr. Lebwohl, professor and chair of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
Phosphodiesterase-4 (PDE-4) activity is elevated in psoriatic skin. Indeed, inhibition of PDE-4 via oral apremilast (Otezla) is an established strategy for improving psoriasis through down-regulation of inflammatory cytokines including tumor necrosis factor–alpha, interleukins-17 and -23, and interferon-gamma. Notably, however, roflumilast is orders of magnitude more potent than any other PDE-4 inhibitor. An oral version marketed as Daliresp has been available for treatment of chronic obstructive pulmonary disease for nearly a decade.
The 12-week, multicenter, phase 2b study included 331 patients with chronic plaque psoriasis who were randomized to once-daily 0.3% roflumilast cream, 0.15% roflumilast cream, or vehicle. Three-quarters of participants had a baseline Investigator Global Assessment (IGA) score of 3, indicative of moderate disease.
The primary endpoint was achievement of an IGA score of 0 or 1 (clear or almost clear) at week 6. The observed improvement was dose related, although both doses of roflumilast were significantly more effective than vehicle. However, peak improvement occurred at week 8, not week 6, with subsequent plateauing of response through week 12. A week 8 IGA of 0 or 1 plus at least a 2-grade improvement from baseline occurred in 32% of the high-dose roflumilast group, 25% of those on the 0.15% formulation, and 10% of controls.
“The effect in improvement was very rapid, with a statistically significant improvement compared to vehicle for both concentrations as early as week 2,” Dr. Lebwohl said.
A key secondary endpoint focused on treatment response in intertriginous areas, since “those are the areas where we really don’t want to use steroids because of major irritation problems,” the dermatologist explained. At week 12, treatment success as defined by an intertriginous IGA score of 0 or 1 plus at least a 2-point improvement from baseline was seen in 86% of the 0.3% roflumilast cream group, 50% on low-dose therapy, and 29% of controls.
About 65% of subjects on high-dose roflumilast cream reported at least a 4-point reduction in the Worst Itch–Numerical Rating Scale by week 8, as did 58% of those on the low-dose version and 42% of controls. Another secondary endpoint – patient-reported burden of disease as captured in a Psoriasis Symptoms Diary – showed a significant divergence between both doses of roflumilast and vehicle as early as week 2.
“Adverse events were negligible,” Dr. Lebwohl said. “In fact, there was only one discontinuation in the 0.3% arm, compared to none with 0.15% and two with vehicle.”
The phase 3 program is now recruiting participants.
The phase 2b study was funded by Arcutis Biotherapeutics. Dr. Lebwohl reported receiving research funding from and serving as a consultant to that company and numerous others.
Once-daily – including challenging lesions in tough-to-treat intertriginous areas – in a phase 2b, randomized, double-blind, vehicle-controlled clinical trial, Mark G. Lebwohl, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The clinical improvement occurred rapidly. And topical roflumilast’s side effect profile was essentially the same as in vehicle-treated controls, which suggests a potential major advantage for the novel drug in future clinical practice. After all, topical treatment is the mainstay of psoriasis therapy, but the current topical agents – high-potency corticosteroids, vitamin D derivatives, and retinoids – have long-term tolerability, efficacy, or side effect issues, especially in treating sensitive skin areas, including the face and intertriginous areas.
“Roflumilast cream could really be a game changer,” predicted Dr. Lebwohl, professor and chair of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
Phosphodiesterase-4 (PDE-4) activity is elevated in psoriatic skin. Indeed, inhibition of PDE-4 via oral apremilast (Otezla) is an established strategy for improving psoriasis through down-regulation of inflammatory cytokines including tumor necrosis factor–alpha, interleukins-17 and -23, and interferon-gamma. Notably, however, roflumilast is orders of magnitude more potent than any other PDE-4 inhibitor. An oral version marketed as Daliresp has been available for treatment of chronic obstructive pulmonary disease for nearly a decade.
The 12-week, multicenter, phase 2b study included 331 patients with chronic plaque psoriasis who were randomized to once-daily 0.3% roflumilast cream, 0.15% roflumilast cream, or vehicle. Three-quarters of participants had a baseline Investigator Global Assessment (IGA) score of 3, indicative of moderate disease.
The primary endpoint was achievement of an IGA score of 0 or 1 (clear or almost clear) at week 6. The observed improvement was dose related, although both doses of roflumilast were significantly more effective than vehicle. However, peak improvement occurred at week 8, not week 6, with subsequent plateauing of response through week 12. A week 8 IGA of 0 or 1 plus at least a 2-grade improvement from baseline occurred in 32% of the high-dose roflumilast group, 25% of those on the 0.15% formulation, and 10% of controls.
“The effect in improvement was very rapid, with a statistically significant improvement compared to vehicle for both concentrations as early as week 2,” Dr. Lebwohl said.
A key secondary endpoint focused on treatment response in intertriginous areas, since “those are the areas where we really don’t want to use steroids because of major irritation problems,” the dermatologist explained. At week 12, treatment success as defined by an intertriginous IGA score of 0 or 1 plus at least a 2-point improvement from baseline was seen in 86% of the 0.3% roflumilast cream group, 50% on low-dose therapy, and 29% of controls.
About 65% of subjects on high-dose roflumilast cream reported at least a 4-point reduction in the Worst Itch–Numerical Rating Scale by week 8, as did 58% of those on the low-dose version and 42% of controls. Another secondary endpoint – patient-reported burden of disease as captured in a Psoriasis Symptoms Diary – showed a significant divergence between both doses of roflumilast and vehicle as early as week 2.
“Adverse events were negligible,” Dr. Lebwohl said. “In fact, there was only one discontinuation in the 0.3% arm, compared to none with 0.15% and two with vehicle.”
The phase 3 program is now recruiting participants.
The phase 2b study was funded by Arcutis Biotherapeutics. Dr. Lebwohl reported receiving research funding from and serving as a consultant to that company and numerous others.
Once-daily – including challenging lesions in tough-to-treat intertriginous areas – in a phase 2b, randomized, double-blind, vehicle-controlled clinical trial, Mark G. Lebwohl, MD, reported at the virtual annual meeting of the American Academy of Dermatology.
The clinical improvement occurred rapidly. And topical roflumilast’s side effect profile was essentially the same as in vehicle-treated controls, which suggests a potential major advantage for the novel drug in future clinical practice. After all, topical treatment is the mainstay of psoriasis therapy, but the current topical agents – high-potency corticosteroids, vitamin D derivatives, and retinoids – have long-term tolerability, efficacy, or side effect issues, especially in treating sensitive skin areas, including the face and intertriginous areas.
“Roflumilast cream could really be a game changer,” predicted Dr. Lebwohl, professor and chair of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York.
Phosphodiesterase-4 (PDE-4) activity is elevated in psoriatic skin. Indeed, inhibition of PDE-4 via oral apremilast (Otezla) is an established strategy for improving psoriasis through down-regulation of inflammatory cytokines including tumor necrosis factor–alpha, interleukins-17 and -23, and interferon-gamma. Notably, however, roflumilast is orders of magnitude more potent than any other PDE-4 inhibitor. An oral version marketed as Daliresp has been available for treatment of chronic obstructive pulmonary disease for nearly a decade.
The 12-week, multicenter, phase 2b study included 331 patients with chronic plaque psoriasis who were randomized to once-daily 0.3% roflumilast cream, 0.15% roflumilast cream, or vehicle. Three-quarters of participants had a baseline Investigator Global Assessment (IGA) score of 3, indicative of moderate disease.
The primary endpoint was achievement of an IGA score of 0 or 1 (clear or almost clear) at week 6. The observed improvement was dose related, although both doses of roflumilast were significantly more effective than vehicle. However, peak improvement occurred at week 8, not week 6, with subsequent plateauing of response through week 12. A week 8 IGA of 0 or 1 plus at least a 2-grade improvement from baseline occurred in 32% of the high-dose roflumilast group, 25% of those on the 0.15% formulation, and 10% of controls.
“The effect in improvement was very rapid, with a statistically significant improvement compared to vehicle for both concentrations as early as week 2,” Dr. Lebwohl said.
A key secondary endpoint focused on treatment response in intertriginous areas, since “those are the areas where we really don’t want to use steroids because of major irritation problems,” the dermatologist explained. At week 12, treatment success as defined by an intertriginous IGA score of 0 or 1 plus at least a 2-point improvement from baseline was seen in 86% of the 0.3% roflumilast cream group, 50% on low-dose therapy, and 29% of controls.
About 65% of subjects on high-dose roflumilast cream reported at least a 4-point reduction in the Worst Itch–Numerical Rating Scale by week 8, as did 58% of those on the low-dose version and 42% of controls. Another secondary endpoint – patient-reported burden of disease as captured in a Psoriasis Symptoms Diary – showed a significant divergence between both doses of roflumilast and vehicle as early as week 2.
“Adverse events were negligible,” Dr. Lebwohl said. “In fact, there was only one discontinuation in the 0.3% arm, compared to none with 0.15% and two with vehicle.”
The phase 3 program is now recruiting participants.
The phase 2b study was funded by Arcutis Biotherapeutics. Dr. Lebwohl reported receiving research funding from and serving as a consultant to that company and numerous others.
FROM AAD 20
Subcutaneous nemolizumab eases itching for atopic dermatitis
of 215 patients in Japan.
Controlling the pruritus associated with atopic dermatitis (AD) can have a significant impact on patients’ quality of life, wrote Kenji Kabashima, MD, PhD, of the department of dermatology at Kyoto University, and coauthors. Frequent scratching can cause not only mechanical skin damage, but also may enhance inflammatory reactions and contribute to sleep problems.
In earlier phase studies, nemolizumab, a humanized monoclonal antibody against interleukin-31 receptor A, showed efficacy in reducing pruritus in patients with AD, but has not been well studied in patients who are also using topical agents, they wrote.
In the study published in the New England Journal of Medicine, the researchers randomized 143 patients with AD and moderate to severe pruritus to 60 mg of subcutaneous nemolizumab and 72 patients to a placebo every 4 weeks for 16 weeks. All patients were aged 13 years and older with a confirmed AD diagnosis and a history of inadequate response to or inability to use treatments, including topical glucocorticoids and oral antihistamines. Their average age was 40 years, approximately two-thirds were male, and the average disease duration was approximately 30 years. Topical treatments included a medium potency glucocorticoid in 97% of patients in both groups, and a topical calcineurin inhibitor in 41% of those on nemolizumab, and 40% of those on placebo; almost 90% of the patients in both groups were on oral antihistamines.
At 16 weeks, scores on the visual analog scale for pruritus (the primary outcome) significantly improved from baseline in the nemolizumab group, compared with the placebo group (a mean change of –42.8% and –21.4%, respectively, P < .001).
In addition, more patients in the nemolizumab group, compared with the placebo group (40% vs. 22%) achieved a score of 4 or less on the Dermatology Life Quality Index, with lower scores reflecting less impact of disease on daily life. In addition, more patients in the nemolizumab group, compared with the placebo group (55% vs. 21%) achieved a score of 7 or less on the Insomnia Severity Index.
During the study, 71% of the patients in each group reported adverse events, most were mild or moderate. The most common adverse event was worsening AD, reported by 24% of the nemolizumab patients and 21% of the placebo patients. Reactions related to the injection occurred in 8% of nemolizumab patients and 3% of placebo patients. Cytokine abnormalities, which included an increased level of thymus and activation regulated chemokine, were reported in 10 (7%) of the patients on nemolizumab, none of which occurred in those on placebo. “Most were not accompanied by a worsening of signs of or the extent of atopic dermatitis,” the authors wrote.
Severe adverse events were reported in three patients (2%) in the nemolizumab group, which were Meniere’s disease, acute pancreatitis, and AD in one patient each. No severe adverse events were reported in the placebo group. In addition, three patients in the nemolizumab group experienced four treatment-related adverse events that led them to discontinue treatment: AD, Meniere’s disease, alopecia, and peripheral edema.
The study findings were limited by several factors including the relatively short treatment period, inclusion only of Japanese patients, inclusion of patients aged as young as 13 years, and the inability to draw conclusions from the secondary endpoints such as quality of life and sleep issues, the researchers noted.
However, the results suggest that “nemolizumab plus topical agents may ameliorate both pruritus and signs of eczema and may lessen the severity of atopic dermatitis by disrupting the itch-scratch cycle,” they added.
“Novel therapies [for AD] are needed, as there are still patients who need better disease control despite current therapies, and AD is a heterogeneous disease that may need different treatment approaches,” Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, said in an interview.
Dr. Simpson, who was not an investigator in this study, said that he was somewhat surprised that the itch reduction was lower in the current study, compared with previous studies by the same group. Also surprising was the increase in cytokine abnormalities in the nemolizumab group, which “needs further study.”
Overall, the data “provide support that blockade of the IL-31 receptor improves itch in AD and appears to have some effect on inflammation,” Dr. Simpson said.
One challenge to the clinical use of nemolizumab will be identifying “where this type of drug fits into the treatment paradigm,” and determining whether specific patients whose disease is driven more by this neuroimmune pathway could benefit more than with the traditional IL-4 or IL-13 blockade, he said.
The study was supported by Maruho. Dr. Kabashima disclosed consulting fees from Maruho and two coauthors were Maruho employees. Dr. Simpson had no financial conflicts relevant to this study, but he reported receiving research grants and other financial relationships with manufacturers of AD therapies.
SOURCE: Kabashima K et al. N Engl J Med. 2020 Jul 9. doi: 10.1056/NEJMoa1917006.
of 215 patients in Japan.
Controlling the pruritus associated with atopic dermatitis (AD) can have a significant impact on patients’ quality of life, wrote Kenji Kabashima, MD, PhD, of the department of dermatology at Kyoto University, and coauthors. Frequent scratching can cause not only mechanical skin damage, but also may enhance inflammatory reactions and contribute to sleep problems.
In earlier phase studies, nemolizumab, a humanized monoclonal antibody against interleukin-31 receptor A, showed efficacy in reducing pruritus in patients with AD, but has not been well studied in patients who are also using topical agents, they wrote.
In the study published in the New England Journal of Medicine, the researchers randomized 143 patients with AD and moderate to severe pruritus to 60 mg of subcutaneous nemolizumab and 72 patients to a placebo every 4 weeks for 16 weeks. All patients were aged 13 years and older with a confirmed AD diagnosis and a history of inadequate response to or inability to use treatments, including topical glucocorticoids and oral antihistamines. Their average age was 40 years, approximately two-thirds were male, and the average disease duration was approximately 30 years. Topical treatments included a medium potency glucocorticoid in 97% of patients in both groups, and a topical calcineurin inhibitor in 41% of those on nemolizumab, and 40% of those on placebo; almost 90% of the patients in both groups were on oral antihistamines.
At 16 weeks, scores on the visual analog scale for pruritus (the primary outcome) significantly improved from baseline in the nemolizumab group, compared with the placebo group (a mean change of –42.8% and –21.4%, respectively, P < .001).
In addition, more patients in the nemolizumab group, compared with the placebo group (40% vs. 22%) achieved a score of 4 or less on the Dermatology Life Quality Index, with lower scores reflecting less impact of disease on daily life. In addition, more patients in the nemolizumab group, compared with the placebo group (55% vs. 21%) achieved a score of 7 or less on the Insomnia Severity Index.
During the study, 71% of the patients in each group reported adverse events, most were mild or moderate. The most common adverse event was worsening AD, reported by 24% of the nemolizumab patients and 21% of the placebo patients. Reactions related to the injection occurred in 8% of nemolizumab patients and 3% of placebo patients. Cytokine abnormalities, which included an increased level of thymus and activation regulated chemokine, were reported in 10 (7%) of the patients on nemolizumab, none of which occurred in those on placebo. “Most were not accompanied by a worsening of signs of or the extent of atopic dermatitis,” the authors wrote.
Severe adverse events were reported in three patients (2%) in the nemolizumab group, which were Meniere’s disease, acute pancreatitis, and AD in one patient each. No severe adverse events were reported in the placebo group. In addition, three patients in the nemolizumab group experienced four treatment-related adverse events that led them to discontinue treatment: AD, Meniere’s disease, alopecia, and peripheral edema.
The study findings were limited by several factors including the relatively short treatment period, inclusion only of Japanese patients, inclusion of patients aged as young as 13 years, and the inability to draw conclusions from the secondary endpoints such as quality of life and sleep issues, the researchers noted.
However, the results suggest that “nemolizumab plus topical agents may ameliorate both pruritus and signs of eczema and may lessen the severity of atopic dermatitis by disrupting the itch-scratch cycle,” they added.
“Novel therapies [for AD] are needed, as there are still patients who need better disease control despite current therapies, and AD is a heterogeneous disease that may need different treatment approaches,” Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, said in an interview.
Dr. Simpson, who was not an investigator in this study, said that he was somewhat surprised that the itch reduction was lower in the current study, compared with previous studies by the same group. Also surprising was the increase in cytokine abnormalities in the nemolizumab group, which “needs further study.”
Overall, the data “provide support that blockade of the IL-31 receptor improves itch in AD and appears to have some effect on inflammation,” Dr. Simpson said.
One challenge to the clinical use of nemolizumab will be identifying “where this type of drug fits into the treatment paradigm,” and determining whether specific patients whose disease is driven more by this neuroimmune pathway could benefit more than with the traditional IL-4 or IL-13 blockade, he said.
The study was supported by Maruho. Dr. Kabashima disclosed consulting fees from Maruho and two coauthors were Maruho employees. Dr. Simpson had no financial conflicts relevant to this study, but he reported receiving research grants and other financial relationships with manufacturers of AD therapies.
SOURCE: Kabashima K et al. N Engl J Med. 2020 Jul 9. doi: 10.1056/NEJMoa1917006.
of 215 patients in Japan.
Controlling the pruritus associated with atopic dermatitis (AD) can have a significant impact on patients’ quality of life, wrote Kenji Kabashima, MD, PhD, of the department of dermatology at Kyoto University, and coauthors. Frequent scratching can cause not only mechanical skin damage, but also may enhance inflammatory reactions and contribute to sleep problems.
In earlier phase studies, nemolizumab, a humanized monoclonal antibody against interleukin-31 receptor A, showed efficacy in reducing pruritus in patients with AD, but has not been well studied in patients who are also using topical agents, they wrote.
In the study published in the New England Journal of Medicine, the researchers randomized 143 patients with AD and moderate to severe pruritus to 60 mg of subcutaneous nemolizumab and 72 patients to a placebo every 4 weeks for 16 weeks. All patients were aged 13 years and older with a confirmed AD diagnosis and a history of inadequate response to or inability to use treatments, including topical glucocorticoids and oral antihistamines. Their average age was 40 years, approximately two-thirds were male, and the average disease duration was approximately 30 years. Topical treatments included a medium potency glucocorticoid in 97% of patients in both groups, and a topical calcineurin inhibitor in 41% of those on nemolizumab, and 40% of those on placebo; almost 90% of the patients in both groups were on oral antihistamines.
At 16 weeks, scores on the visual analog scale for pruritus (the primary outcome) significantly improved from baseline in the nemolizumab group, compared with the placebo group (a mean change of –42.8% and –21.4%, respectively, P < .001).
In addition, more patients in the nemolizumab group, compared with the placebo group (40% vs. 22%) achieved a score of 4 or less on the Dermatology Life Quality Index, with lower scores reflecting less impact of disease on daily life. In addition, more patients in the nemolizumab group, compared with the placebo group (55% vs. 21%) achieved a score of 7 or less on the Insomnia Severity Index.
During the study, 71% of the patients in each group reported adverse events, most were mild or moderate. The most common adverse event was worsening AD, reported by 24% of the nemolizumab patients and 21% of the placebo patients. Reactions related to the injection occurred in 8% of nemolizumab patients and 3% of placebo patients. Cytokine abnormalities, which included an increased level of thymus and activation regulated chemokine, were reported in 10 (7%) of the patients on nemolizumab, none of which occurred in those on placebo. “Most were not accompanied by a worsening of signs of or the extent of atopic dermatitis,” the authors wrote.
Severe adverse events were reported in three patients (2%) in the nemolizumab group, which were Meniere’s disease, acute pancreatitis, and AD in one patient each. No severe adverse events were reported in the placebo group. In addition, three patients in the nemolizumab group experienced four treatment-related adverse events that led them to discontinue treatment: AD, Meniere’s disease, alopecia, and peripheral edema.
The study findings were limited by several factors including the relatively short treatment period, inclusion only of Japanese patients, inclusion of patients aged as young as 13 years, and the inability to draw conclusions from the secondary endpoints such as quality of life and sleep issues, the researchers noted.
However, the results suggest that “nemolizumab plus topical agents may ameliorate both pruritus and signs of eczema and may lessen the severity of atopic dermatitis by disrupting the itch-scratch cycle,” they added.
“Novel therapies [for AD] are needed, as there are still patients who need better disease control despite current therapies, and AD is a heterogeneous disease that may need different treatment approaches,” Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland, said in an interview.
Dr. Simpson, who was not an investigator in this study, said that he was somewhat surprised that the itch reduction was lower in the current study, compared with previous studies by the same group. Also surprising was the increase in cytokine abnormalities in the nemolizumab group, which “needs further study.”
Overall, the data “provide support that blockade of the IL-31 receptor improves itch in AD and appears to have some effect on inflammation,” Dr. Simpson said.
One challenge to the clinical use of nemolizumab will be identifying “where this type of drug fits into the treatment paradigm,” and determining whether specific patients whose disease is driven more by this neuroimmune pathway could benefit more than with the traditional IL-4 or IL-13 blockade, he said.
The study was supported by Maruho. Dr. Kabashima disclosed consulting fees from Maruho and two coauthors were Maruho employees. Dr. Simpson had no financial conflicts relevant to this study, but he reported receiving research grants and other financial relationships with manufacturers of AD therapies.
SOURCE: Kabashima K et al. N Engl J Med. 2020 Jul 9. doi: 10.1056/NEJMoa1917006.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Diagnosing molluscum contagiosum can be tricky
The way James R. Treat, MD, sees it, if there ever were a truism in the field of dermatology, it’s that everyone hates molluscum contagiosum.
“It tortures all of us,” Dr. Treat, a pediatric dermatologist at Children’s Hospital of Philadelphia, said during the virtual Pediatric Dermatology 2020: Best Practices and Innovations Conference. “It’s very distressing to parents, but often more distressing to parents than to kids.”
A viral disorder of the skin and mucous membranes characterized by discrete single or multiple, flesh-colored papules, “When you look at inflamed molluscum it can be very difficult to recognize because it looks like a more complicated infection,” said Dr. Treat, who is also associate professor of clinical pediatrics and dermatology, at the University of Pennsylvania, Philadelphia.
An epidemiologic review of 302 MC cases found that 80% of patients were aged younger than 8 years, 63% had more than 15 lesions, and 24% had concomitant atopic dermatitis (J Am Acad Dermatol. 2006; 2006;54[1]:47-54). “Children with atopic dermatitis often have their molluscum last longer,” he said. “The average time course for molluscum is 18 months, but it can certainly be longer than that. So if you say, ‘it’s probably going to go away in a few months,’ that’s probably not going to happen.”
The telltale MC lesion is glossy and contains a white core in the center that can be revealed by shining an otoscope sideways on the lesion. “Umbilication doesn’t always occur, but if the center part looks white, that will help with diagnosis,” Dr. Treat said. “If they’re inflamed and they’re red and you’re worried that there’s a bacterial infection, do a culture, pop the lesion open, and get some of the pus out. If you’re concerned, start them on antibiotics. It’s always worse to miss an infection than to overtreat molluscum. But once you’ve done it a few times and you realize that the cultures are coming back negative, then you’ll probably have your threshold a little higher.”
The most useful clinical sign of MC is the so-called “BOTE” (beginning of the end) sign, which is characterized by erythema and swelling of MC skin lesions. “When the parents come to us in pediatric dermatology, often it’s because their kids have had molluscum for a while,” he said. “It spreads and becomes inflamed and the parents ask, ‘Is it infected?’ The answer is, yes, it’s an infection, but it’s not infected with what you think it is [which is Staphylococcus or Streptococcus], it’s the virus being recognized by the body. When the virus is recognized by the body, it creates a huge inflammatory reaction. That’s usually the time at which the body has had enough of the virus, and it eradicates the rest of it. It means the inflammatory response is finding the molluscum and it’s going to take care of it.”
MC brings its own eczematous response, which can complicate efforts to confirm the diagnosis. Dr. Treat spoke of a young patient he recently saw who had an eczematous reaction on the inner parts of the arms and the upper flank – with no such clinical history. “It kind of came out of the blue,” he said. “You think about contact allergies and other types of dermatitis, but molluscum brings its own eczema. Often what the parents recognize is the eczematous eruption and not the little dots of molluscum. So if you see someone with a new eruption in typical molluscum areas – the flank and your thighs and the back of the legs – and they’ve never had eczema in the past, or they’ve only had mild eczema, think about eczema as a response to molluscum.”
MC can also result in a Gianotti-Crosti syndrome-like reactions (Arch Dermatol. 2012;148[11]:1257-64). “These are angry, inflamed red papules on the knees and on the elbows and on the buttocks and on the cheeks,” Dr. Treat said. “It typically spares the trunk, and they look like molluscum.”
He went on to note that MC can present as cysts, and that MC in the gluteal cleft is a mimicker of condyloma. MC can also cause conjunctivitis, which is increased in HIV patients and in those with atopic dermatitis. “These are patients who should probably see an ophthalmologist” to make no damage has occurred, Dr. Treat said.
He closed his remarks by noting that rarely, MC can be the presenting sign of an immunodeficiency. “The immune system dysregulation that shows up this way is called a DOCK8 mutation, which have eczema and widespread viral disease including warts and molluscum,” Dr. Treat said.
He reported having no financial disclosures.
The way James R. Treat, MD, sees it, if there ever were a truism in the field of dermatology, it’s that everyone hates molluscum contagiosum.
“It tortures all of us,” Dr. Treat, a pediatric dermatologist at Children’s Hospital of Philadelphia, said during the virtual Pediatric Dermatology 2020: Best Practices and Innovations Conference. “It’s very distressing to parents, but often more distressing to parents than to kids.”
A viral disorder of the skin and mucous membranes characterized by discrete single or multiple, flesh-colored papules, “When you look at inflamed molluscum it can be very difficult to recognize because it looks like a more complicated infection,” said Dr. Treat, who is also associate professor of clinical pediatrics and dermatology, at the University of Pennsylvania, Philadelphia.
An epidemiologic review of 302 MC cases found that 80% of patients were aged younger than 8 years, 63% had more than 15 lesions, and 24% had concomitant atopic dermatitis (J Am Acad Dermatol. 2006; 2006;54[1]:47-54). “Children with atopic dermatitis often have their molluscum last longer,” he said. “The average time course for molluscum is 18 months, but it can certainly be longer than that. So if you say, ‘it’s probably going to go away in a few months,’ that’s probably not going to happen.”
The telltale MC lesion is glossy and contains a white core in the center that can be revealed by shining an otoscope sideways on the lesion. “Umbilication doesn’t always occur, but if the center part looks white, that will help with diagnosis,” Dr. Treat said. “If they’re inflamed and they’re red and you’re worried that there’s a bacterial infection, do a culture, pop the lesion open, and get some of the pus out. If you’re concerned, start them on antibiotics. It’s always worse to miss an infection than to overtreat molluscum. But once you’ve done it a few times and you realize that the cultures are coming back negative, then you’ll probably have your threshold a little higher.”
The most useful clinical sign of MC is the so-called “BOTE” (beginning of the end) sign, which is characterized by erythema and swelling of MC skin lesions. “When the parents come to us in pediatric dermatology, often it’s because their kids have had molluscum for a while,” he said. “It spreads and becomes inflamed and the parents ask, ‘Is it infected?’ The answer is, yes, it’s an infection, but it’s not infected with what you think it is [which is Staphylococcus or Streptococcus], it’s the virus being recognized by the body. When the virus is recognized by the body, it creates a huge inflammatory reaction. That’s usually the time at which the body has had enough of the virus, and it eradicates the rest of it. It means the inflammatory response is finding the molluscum and it’s going to take care of it.”
MC brings its own eczematous response, which can complicate efforts to confirm the diagnosis. Dr. Treat spoke of a young patient he recently saw who had an eczematous reaction on the inner parts of the arms and the upper flank – with no such clinical history. “It kind of came out of the blue,” he said. “You think about contact allergies and other types of dermatitis, but molluscum brings its own eczema. Often what the parents recognize is the eczematous eruption and not the little dots of molluscum. So if you see someone with a new eruption in typical molluscum areas – the flank and your thighs and the back of the legs – and they’ve never had eczema in the past, or they’ve only had mild eczema, think about eczema as a response to molluscum.”
MC can also result in a Gianotti-Crosti syndrome-like reactions (Arch Dermatol. 2012;148[11]:1257-64). “These are angry, inflamed red papules on the knees and on the elbows and on the buttocks and on the cheeks,” Dr. Treat said. “It typically spares the trunk, and they look like molluscum.”
He went on to note that MC can present as cysts, and that MC in the gluteal cleft is a mimicker of condyloma. MC can also cause conjunctivitis, which is increased in HIV patients and in those with atopic dermatitis. “These are patients who should probably see an ophthalmologist” to make no damage has occurred, Dr. Treat said.
He closed his remarks by noting that rarely, MC can be the presenting sign of an immunodeficiency. “The immune system dysregulation that shows up this way is called a DOCK8 mutation, which have eczema and widespread viral disease including warts and molluscum,” Dr. Treat said.
He reported having no financial disclosures.
The way James R. Treat, MD, sees it, if there ever were a truism in the field of dermatology, it’s that everyone hates molluscum contagiosum.
“It tortures all of us,” Dr. Treat, a pediatric dermatologist at Children’s Hospital of Philadelphia, said during the virtual Pediatric Dermatology 2020: Best Practices and Innovations Conference. “It’s very distressing to parents, but often more distressing to parents than to kids.”
A viral disorder of the skin and mucous membranes characterized by discrete single or multiple, flesh-colored papules, “When you look at inflamed molluscum it can be very difficult to recognize because it looks like a more complicated infection,” said Dr. Treat, who is also associate professor of clinical pediatrics and dermatology, at the University of Pennsylvania, Philadelphia.
An epidemiologic review of 302 MC cases found that 80% of patients were aged younger than 8 years, 63% had more than 15 lesions, and 24% had concomitant atopic dermatitis (J Am Acad Dermatol. 2006; 2006;54[1]:47-54). “Children with atopic dermatitis often have their molluscum last longer,” he said. “The average time course for molluscum is 18 months, but it can certainly be longer than that. So if you say, ‘it’s probably going to go away in a few months,’ that’s probably not going to happen.”
The telltale MC lesion is glossy and contains a white core in the center that can be revealed by shining an otoscope sideways on the lesion. “Umbilication doesn’t always occur, but if the center part looks white, that will help with diagnosis,” Dr. Treat said. “If they’re inflamed and they’re red and you’re worried that there’s a bacterial infection, do a culture, pop the lesion open, and get some of the pus out. If you’re concerned, start them on antibiotics. It’s always worse to miss an infection than to overtreat molluscum. But once you’ve done it a few times and you realize that the cultures are coming back negative, then you’ll probably have your threshold a little higher.”
The most useful clinical sign of MC is the so-called “BOTE” (beginning of the end) sign, which is characterized by erythema and swelling of MC skin lesions. “When the parents come to us in pediatric dermatology, often it’s because their kids have had molluscum for a while,” he said. “It spreads and becomes inflamed and the parents ask, ‘Is it infected?’ The answer is, yes, it’s an infection, but it’s not infected with what you think it is [which is Staphylococcus or Streptococcus], it’s the virus being recognized by the body. When the virus is recognized by the body, it creates a huge inflammatory reaction. That’s usually the time at which the body has had enough of the virus, and it eradicates the rest of it. It means the inflammatory response is finding the molluscum and it’s going to take care of it.”
MC brings its own eczematous response, which can complicate efforts to confirm the diagnosis. Dr. Treat spoke of a young patient he recently saw who had an eczematous reaction on the inner parts of the arms and the upper flank – with no such clinical history. “It kind of came out of the blue,” he said. “You think about contact allergies and other types of dermatitis, but molluscum brings its own eczema. Often what the parents recognize is the eczematous eruption and not the little dots of molluscum. So if you see someone with a new eruption in typical molluscum areas – the flank and your thighs and the back of the legs – and they’ve never had eczema in the past, or they’ve only had mild eczema, think about eczema as a response to molluscum.”
MC can also result in a Gianotti-Crosti syndrome-like reactions (Arch Dermatol. 2012;148[11]:1257-64). “These are angry, inflamed red papules on the knees and on the elbows and on the buttocks and on the cheeks,” Dr. Treat said. “It typically spares the trunk, and they look like molluscum.”
He went on to note that MC can present as cysts, and that MC in the gluteal cleft is a mimicker of condyloma. MC can also cause conjunctivitis, which is increased in HIV patients and in those with atopic dermatitis. “These are patients who should probably see an ophthalmologist” to make no damage has occurred, Dr. Treat said.
He closed his remarks by noting that rarely, MC can be the presenting sign of an immunodeficiency. “The immune system dysregulation that shows up this way is called a DOCK8 mutation, which have eczema and widespread viral disease including warts and molluscum,” Dr. Treat said.
He reported having no financial disclosures.
FROM PEDIATRIC DERMATOLOGY 2020
Oral difelikefalin quells severe chronic kidney disease–associated itch
, in a first-of-its-kind randomized clinical trial, Gil Yosipovitch, MD, said at the virtual annual meeting of the American Academy of Dermatology.
“Difelikefalin at 1.0 mg was associated with clinically meaningful improvements in pruritus. The improvement in itch was significant by week 2. And nearly 40% of patients achieved a complete response, which was more than two-and-one-half times more than with placebo,” noted Dr. Yosipovitch, professor of dermatology and director of the Miami Itch Center at the University of Miami.
Pruritus associated with chronic kidney disease (CKD) is a common, underrecognized, and distressing condition that causes markedly impaired quality of life. It occurs in patients across all stages of CKD, not just in those on hemodialysis, as is widely but mistakenly believed. And at present there is no approved drug in any country for treatment of CKD-associated itch.
Difelikefalin, a novel selective agonist of peripheral kappa opioid receptors, is designed to have very limited CNS penetration. The drug, which is renally excreted, doesn’t bind to mu or delta opioid receptors. Its antipruritic effect arises from activation of kappa opioid receptors on peripheral sensory neurons and immune cells, the dermatologist explained.
Dr. Yosipovitch presented the results of a phase 2, randomized, double-blind, placebo-controlled, 12-week trial in which 240 patients with severe chronic pruritus and stage 3-5 CKD were assigned to once-daily oral difelikefalin at 0.25 mg, 0.5 mg, or 1.0 mg, or placebo. More than 80% of participants were not on dialysis. Indeed, this was the first-ever clinical trial targeting itch in patients across such a broad spectrum of CKD stages.
The primary study endpoint was change from baseline to week 12 in the weekly mean score on the 24-hour Worst Itching Intensity Numerical Rating Scale. The average baseline score was 7, considered severe pruritus on the 0-10 scale. Patients randomized to difelikefalin at 1.0 mg/day had a mean 4.4-point decrease, a significantly greater improvement than the 3.3-point reduction in placebo-treated controls.
“More than a 4-point decrease is considered a very meaningful itch reduction,” Dr. Yosipovitch noted.
The mean reductions in itch score in patients on 0.25 mg and 0.5 mg/day of difelikefalin were 4.0 and 3.8 points, respectively, which fell short of statistical significance versus placebo.
A key prespecified secondary endpoint was the proportion of subjects with at least a 3-point improvement in itch score over 12 weeks. This was achieved in 72% of patients on the top dose of difelikefalin, compared with 58% of controls, a significant difference. A 4-point or larger decrease in itch score occurred in 65% of patients on 1.0 mg/day of the kappa opioid recent agonist, versus 50% of controls, also a significant difference.
A complete response, defined as an itch score of 0 or 1 at least 80% of the time, was significantly more common in all three active treatment groups than in controls, with rates of 33%, 31.6%, and 38.6% at difelikefalin 0.25, 0.5, and 1.0 mg, compared with 4.4% among those on placebo.
Falls occurred in 1.5% of patients on difelikefalin. “The therapy does seem to increase the risk of dizziness, falls, fatigue, and GI complaints,” according to the investigator.
Still, most of these adverse events were mild or moderate in severity. Only about 1% of participants discontinued treatment for such reasons.
Earlier this year, a positive phase 3 trial of an intravenous formulation of difelikefalin for pruritus was reported in CKD patients on hemodialysis (N Engl J Med. 2020 Jan 16;382[3]:222-32).
In an interview, Dr. Yosipovitch said that this new phase 2 oral dose-finding study wasn’t powered to detect differences in treatment efficacy between the dialysis and nondialysis groups. However, the proportion of patients with at least a 3-point improvement in itch at week 12 was similar in the two groups.
“The oral formulation would of course be more convenient and would be preferred for patients not undergoing hemodialysis,” he said. “I would expect that the IV formulation would be the preferred route of administration for a patient undergoing hemodialysis. An IV formulation would be very convenient for such patients because it’s administered at the dialysis clinic at the end of the hemodialysis session.”
The oral difelikefalin phase 3 program is scheduled to start later in 2020.
CKD-associated itch poses a therapeutic challenge because it has so many contributory factors. These include CKD-induced peripheral neuropathy, functional and structural neuropathic changes in the brain, cutaneous mast cell activation, an imbalance between mu opioid receptor overexpression and kappa opioid receptor downregulation, secondary parathyroidism, and systemic accumulation of aluminum, beta 2 microglobulin, and other dialysis-related substances, the dermatologist observed.
Dr. Yosipovitch reported receiving research grants from a half-dozen pharmaceutical companies. He also serves as a consultant to numerous companies, including Cara Therapeutics, which sponsored the phase 2 trial.
, in a first-of-its-kind randomized clinical trial, Gil Yosipovitch, MD, said at the virtual annual meeting of the American Academy of Dermatology.
“Difelikefalin at 1.0 mg was associated with clinically meaningful improvements in pruritus. The improvement in itch was significant by week 2. And nearly 40% of patients achieved a complete response, which was more than two-and-one-half times more than with placebo,” noted Dr. Yosipovitch, professor of dermatology and director of the Miami Itch Center at the University of Miami.
Pruritus associated with chronic kidney disease (CKD) is a common, underrecognized, and distressing condition that causes markedly impaired quality of life. It occurs in patients across all stages of CKD, not just in those on hemodialysis, as is widely but mistakenly believed. And at present there is no approved drug in any country for treatment of CKD-associated itch.
Difelikefalin, a novel selective agonist of peripheral kappa opioid receptors, is designed to have very limited CNS penetration. The drug, which is renally excreted, doesn’t bind to mu or delta opioid receptors. Its antipruritic effect arises from activation of kappa opioid receptors on peripheral sensory neurons and immune cells, the dermatologist explained.
Dr. Yosipovitch presented the results of a phase 2, randomized, double-blind, placebo-controlled, 12-week trial in which 240 patients with severe chronic pruritus and stage 3-5 CKD were assigned to once-daily oral difelikefalin at 0.25 mg, 0.5 mg, or 1.0 mg, or placebo. More than 80% of participants were not on dialysis. Indeed, this was the first-ever clinical trial targeting itch in patients across such a broad spectrum of CKD stages.
The primary study endpoint was change from baseline to week 12 in the weekly mean score on the 24-hour Worst Itching Intensity Numerical Rating Scale. The average baseline score was 7, considered severe pruritus on the 0-10 scale. Patients randomized to difelikefalin at 1.0 mg/day had a mean 4.4-point decrease, a significantly greater improvement than the 3.3-point reduction in placebo-treated controls.
“More than a 4-point decrease is considered a very meaningful itch reduction,” Dr. Yosipovitch noted.
The mean reductions in itch score in patients on 0.25 mg and 0.5 mg/day of difelikefalin were 4.0 and 3.8 points, respectively, which fell short of statistical significance versus placebo.
A key prespecified secondary endpoint was the proportion of subjects with at least a 3-point improvement in itch score over 12 weeks. This was achieved in 72% of patients on the top dose of difelikefalin, compared with 58% of controls, a significant difference. A 4-point or larger decrease in itch score occurred in 65% of patients on 1.0 mg/day of the kappa opioid recent agonist, versus 50% of controls, also a significant difference.
A complete response, defined as an itch score of 0 or 1 at least 80% of the time, was significantly more common in all three active treatment groups than in controls, with rates of 33%, 31.6%, and 38.6% at difelikefalin 0.25, 0.5, and 1.0 mg, compared with 4.4% among those on placebo.
Falls occurred in 1.5% of patients on difelikefalin. “The therapy does seem to increase the risk of dizziness, falls, fatigue, and GI complaints,” according to the investigator.
Still, most of these adverse events were mild or moderate in severity. Only about 1% of participants discontinued treatment for such reasons.
Earlier this year, a positive phase 3 trial of an intravenous formulation of difelikefalin for pruritus was reported in CKD patients on hemodialysis (N Engl J Med. 2020 Jan 16;382[3]:222-32).
In an interview, Dr. Yosipovitch said that this new phase 2 oral dose-finding study wasn’t powered to detect differences in treatment efficacy between the dialysis and nondialysis groups. However, the proportion of patients with at least a 3-point improvement in itch at week 12 was similar in the two groups.
“The oral formulation would of course be more convenient and would be preferred for patients not undergoing hemodialysis,” he said. “I would expect that the IV formulation would be the preferred route of administration for a patient undergoing hemodialysis. An IV formulation would be very convenient for such patients because it’s administered at the dialysis clinic at the end of the hemodialysis session.”
The oral difelikefalin phase 3 program is scheduled to start later in 2020.
CKD-associated itch poses a therapeutic challenge because it has so many contributory factors. These include CKD-induced peripheral neuropathy, functional and structural neuropathic changes in the brain, cutaneous mast cell activation, an imbalance between mu opioid receptor overexpression and kappa opioid receptor downregulation, secondary parathyroidism, and systemic accumulation of aluminum, beta 2 microglobulin, and other dialysis-related substances, the dermatologist observed.
Dr. Yosipovitch reported receiving research grants from a half-dozen pharmaceutical companies. He also serves as a consultant to numerous companies, including Cara Therapeutics, which sponsored the phase 2 trial.
, in a first-of-its-kind randomized clinical trial, Gil Yosipovitch, MD, said at the virtual annual meeting of the American Academy of Dermatology.
“Difelikefalin at 1.0 mg was associated with clinically meaningful improvements in pruritus. The improvement in itch was significant by week 2. And nearly 40% of patients achieved a complete response, which was more than two-and-one-half times more than with placebo,” noted Dr. Yosipovitch, professor of dermatology and director of the Miami Itch Center at the University of Miami.
Pruritus associated with chronic kidney disease (CKD) is a common, underrecognized, and distressing condition that causes markedly impaired quality of life. It occurs in patients across all stages of CKD, not just in those on hemodialysis, as is widely but mistakenly believed. And at present there is no approved drug in any country for treatment of CKD-associated itch.
Difelikefalin, a novel selective agonist of peripheral kappa opioid receptors, is designed to have very limited CNS penetration. The drug, which is renally excreted, doesn’t bind to mu or delta opioid receptors. Its antipruritic effect arises from activation of kappa opioid receptors on peripheral sensory neurons and immune cells, the dermatologist explained.
Dr. Yosipovitch presented the results of a phase 2, randomized, double-blind, placebo-controlled, 12-week trial in which 240 patients with severe chronic pruritus and stage 3-5 CKD were assigned to once-daily oral difelikefalin at 0.25 mg, 0.5 mg, or 1.0 mg, or placebo. More than 80% of participants were not on dialysis. Indeed, this was the first-ever clinical trial targeting itch in patients across such a broad spectrum of CKD stages.
The primary study endpoint was change from baseline to week 12 in the weekly mean score on the 24-hour Worst Itching Intensity Numerical Rating Scale. The average baseline score was 7, considered severe pruritus on the 0-10 scale. Patients randomized to difelikefalin at 1.0 mg/day had a mean 4.4-point decrease, a significantly greater improvement than the 3.3-point reduction in placebo-treated controls.
“More than a 4-point decrease is considered a very meaningful itch reduction,” Dr. Yosipovitch noted.
The mean reductions in itch score in patients on 0.25 mg and 0.5 mg/day of difelikefalin were 4.0 and 3.8 points, respectively, which fell short of statistical significance versus placebo.
A key prespecified secondary endpoint was the proportion of subjects with at least a 3-point improvement in itch score over 12 weeks. This was achieved in 72% of patients on the top dose of difelikefalin, compared with 58% of controls, a significant difference. A 4-point or larger decrease in itch score occurred in 65% of patients on 1.0 mg/day of the kappa opioid recent agonist, versus 50% of controls, also a significant difference.
A complete response, defined as an itch score of 0 or 1 at least 80% of the time, was significantly more common in all three active treatment groups than in controls, with rates of 33%, 31.6%, and 38.6% at difelikefalin 0.25, 0.5, and 1.0 mg, compared with 4.4% among those on placebo.
Falls occurred in 1.5% of patients on difelikefalin. “The therapy does seem to increase the risk of dizziness, falls, fatigue, and GI complaints,” according to the investigator.
Still, most of these adverse events were mild or moderate in severity. Only about 1% of participants discontinued treatment for such reasons.
Earlier this year, a positive phase 3 trial of an intravenous formulation of difelikefalin for pruritus was reported in CKD patients on hemodialysis (N Engl J Med. 2020 Jan 16;382[3]:222-32).
In an interview, Dr. Yosipovitch said that this new phase 2 oral dose-finding study wasn’t powered to detect differences in treatment efficacy between the dialysis and nondialysis groups. However, the proportion of patients with at least a 3-point improvement in itch at week 12 was similar in the two groups.
“The oral formulation would of course be more convenient and would be preferred for patients not undergoing hemodialysis,” he said. “I would expect that the IV formulation would be the preferred route of administration for a patient undergoing hemodialysis. An IV formulation would be very convenient for such patients because it’s administered at the dialysis clinic at the end of the hemodialysis session.”
The oral difelikefalin phase 3 program is scheduled to start later in 2020.
CKD-associated itch poses a therapeutic challenge because it has so many contributory factors. These include CKD-induced peripheral neuropathy, functional and structural neuropathic changes in the brain, cutaneous mast cell activation, an imbalance between mu opioid receptor overexpression and kappa opioid receptor downregulation, secondary parathyroidism, and systemic accumulation of aluminum, beta 2 microglobulin, and other dialysis-related substances, the dermatologist observed.
Dr. Yosipovitch reported receiving research grants from a half-dozen pharmaceutical companies. He also serves as a consultant to numerous companies, including Cara Therapeutics, which sponsored the phase 2 trial.
FROM AAD 2020
Even a few days of steroids may be risky, new study suggests
Extended use of corticosteroids for chronic inflammatory conditions puts patients at risk for serious adverse events (AEs), including cardiovascular disease, osteoporosis, cataracts, and diabetes. Now, a growing body of evidence suggests that even short bursts of these drugs are associated with serious risks.
Most recently, a population-based study of more than 2.6 million people found that taking corticosteroids for 14 days or less was associated with a substantially greater risk for gastrointestinal (GI) bleeding, sepsis, and heart failure, particularly within the first 30 days after therapy.
In the study, Tsung-Chieh Yao, MD, PhD, a professor in the division of allergy, asthma, and rheumatology in the department of pediatrics at Chang Gung Memorial Hospital in Taoyuan, Taiwan, and colleagues used a self-controlled case series to analyze data from Taiwan’s National Health Insurance Research Database of medical claims. They compared patients’ conditions in the period from 5 to 90 days before treatment to conditions from the periods from 5 to 30 days and from 31 to 90 days after therapy.
With a median duration of 3 days of treatment, the incidence rate ratios (IRRs) were 1.80 (95% confidence interval, 1.75-1.84) for GI bleeding, 1.99 (95% CI, 1.70-2.32) for sepsis, and 2.37 (95% CI, 2.13-2.63) for heart failure.
Given the findings, physicians should weigh the benefits against the risks of rare but potentially serious consequences of these anti-inflammatory drugs, according to the authors.
“After initiating patients on oral steroid bursts, physicians should be on the lookout for these severe adverse events, particularly within the first month after initiation of steroid therapy,” Dr. Yao said in an interview.
The findings were published online July 6 in Annals of Internal Medicine.
Of the 15,859,129 adult Asians in the Taiwanese database, the study included 2,623,327 adults aged 20-64 years who received single steroid bursts (14 days or less) between Jan. 1, 2013, and Dec. 31, 2015.
Almost 60% of the indications were for skin disorders, such as eczema and urticaria, and for respiratory tract infections, such as sinusitis and acute pharyngitis. Among specialties, dermatology, otolaryngology, family practice, internal medicine, and pediatrics accounted for 88% of prescriptions.
“Our findings are important for physicians and guideline developers because short-term use of oral corticosteroids is common and the real-world safety of this approach remains unclear,” the authors wrote. They acknowledged that the database did not provide information on such potential confounders as disease severity and lifestyle factors, nor did it include children and vulnerable individuals, which may limit the generalizability of the results.
The findings echo those of a 2017 cohort study conducted by researchers at the University of Michigan in Ann Arbor. That study, by Akbar K. Waljee, MD, assistant professor of gastroenterology, University of Michigan, Ann Arbor, and colleagues, included data on more than 1.5 million privately insured U.S. adults. The researchers included somewhat longer steroid bursts of up to 30 days’ duration and found that use of the drugs was associated with a greater than fivefold increased risk for sepsis, a more than threefold increased risk for venous thromboembolism, and a nearly twofold increased risk for fracture within 30 days of starting treatment.
Furthermore, the elevated risk persisted at prednisone-equivalent doses of less than 20 mg/d (IRR, 4.02 for sepsis, 3.61 for venous thromboembolism, and 1.83 for fracture; all P < .001).
The U.S. study also found that during the 3-year period from 2012 to 2014, more than 20% of patients were prescribed short-term oral corticosteroids.
“Both studies indicate that these short-term regimens are more common in the real world than was previously thought and are not risk free,” Dr. Yao said.
Recognition that corticosteroids are associated with adverse events has been building for decades, according to the authors of an editorial that accompanies the new study.
“However, we commonly use short corticosteroid ‘bursts’ for minor ailments despite a lack of evidence for meaningful benefit. We are now learning that bursts as short as 3 days may increase risk for serious AEs, even in young and healthy people,” wrote editorialists Beth I. Wallace, MD, of the Center for Clinical Management Research at the VA Ann Arbor Healthcare System and the Institute for Healthcare Policy and Innovation at Michigan Medicine, Ann Arbor, and Dr. Waljee, who led the 2017 study.
Dr. Wallace and Dr. Waljee drew parallels between corticosteroid bursts and other short-term regimens, such as of antibiotics and opiates, in which prescriber preference and sometimes patient pressure play a role. “All of these treatments have well-defined indications but can cause net harm when used. We can thus conceive of a corticosteroid stewardship model of targeted interventions that aims to reduce inappropriate prescribing,” they wrote.
In an interview, Dr. Wallace, a rheumatologist who prescribes oral steroids fairly frequently, noted that the Taiwan study is the first to investigate steroid bursts. “Up till now, these very short courses have flown under the radar. Clinicians very commonly prescribe short courses to help relieve symptoms of self-limited conditions like bronchitis, and we assume that because the exposure duration is short, the risks are low, especially for patients who are otherwise healthy.”
She warned that the data in the current study indicate that these short bursts – even at the lower end of the 1- to 2-week courses American physicians prescribe most often – carry small but real increases in risk for serious AEs. “And these increases were seen in young, healthy people, not just in people with preexisting conditions,” she said. “So, we might need to start thinking harder about how we are prescribing even these very short courses of steroids and try to use steroids only when their meaningful benefits really outweigh the risk.”
She noted that a patient with a chronic inflammatory condition such as rheumatoid arthritis may benefit substantially from short-term steroids to treat a disease flare. In that specific case, the benefits of short-term steroids may outweigh the risks, Dr. Wallace said.
But not everyone thinks a new strategy is needed. For Whitney A. High, MD, associate professor of dermatology and pathology at the University of Colorado at Denver, Aurora, the overprescribing of short-term corticosteroids is not a problem, and dermatologists are already exercising caution.
“I only prescribe these drugs short term to, at a guess, about 1 in 40 patients and only when a patient is miserable and quality of life is being seriously affected,” he said in an interview. “And that’s something that can’t be measured in a database study like the one from Taiwan but only in a risk-benefit analysis,” he said.
Furthermore, dermatologists have other drugs and technologies in their armamentarium, including topical steroids with occlusion or with wet wraps, phototherapy, phosphodiesterase inhibitors, calcipotriene, methotrexate and other immunosuppressive agents, and biologics. “In fact, many of these agents are specifically referred to as steroid-sparing,” Dr. High said.
Nor does he experience much pressure from patients to prescribe these drugs. “While occasionally I may encounter a patient who places pressure on me for oral steroids, it’s probably not nearly as frequently as providers in other fields are pressured to prescribe antibiotics or narcotics,” he said.
According to the Taiwanese researchers, the next step is to conduct more studies, including clinical trials, to determine optimal use of corticosteroids by monitoring adverse events. In the meantime, for practitioners such as Dr. Wallace and Dr. High, there is ample evidence from several recent studies of the harms of short-term corticosteroids, whereas the benefits for patients with self-limiting conditions remain uncertain. “This and other studies like it quite appropriately remind providers to avoid oral steroids when they’re not necessary and to seek alternatives where possible,” Dr. High said.
The study was supported by the National Health Research Institutes of Taiwan, the Ministry of Science and Technology of Taiwan, the Chang Gung Medical Foundation, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NIH). Dr. Yao has disclosed no relevant financial relationships. Dr. Wu has received grants from GlaxoSmithKline outside the submitted work. The editorialists and Dr. High have disclosed no relevant financial relationships. Dr. Wallace received an NIH grant during the writing of the editorial.
A version of this article originally appeared on Medscape.com.
Extended use of corticosteroids for chronic inflammatory conditions puts patients at risk for serious adverse events (AEs), including cardiovascular disease, osteoporosis, cataracts, and diabetes. Now, a growing body of evidence suggests that even short bursts of these drugs are associated with serious risks.
Most recently, a population-based study of more than 2.6 million people found that taking corticosteroids for 14 days or less was associated with a substantially greater risk for gastrointestinal (GI) bleeding, sepsis, and heart failure, particularly within the first 30 days after therapy.
In the study, Tsung-Chieh Yao, MD, PhD, a professor in the division of allergy, asthma, and rheumatology in the department of pediatrics at Chang Gung Memorial Hospital in Taoyuan, Taiwan, and colleagues used a self-controlled case series to analyze data from Taiwan’s National Health Insurance Research Database of medical claims. They compared patients’ conditions in the period from 5 to 90 days before treatment to conditions from the periods from 5 to 30 days and from 31 to 90 days after therapy.
With a median duration of 3 days of treatment, the incidence rate ratios (IRRs) were 1.80 (95% confidence interval, 1.75-1.84) for GI bleeding, 1.99 (95% CI, 1.70-2.32) for sepsis, and 2.37 (95% CI, 2.13-2.63) for heart failure.
Given the findings, physicians should weigh the benefits against the risks of rare but potentially serious consequences of these anti-inflammatory drugs, according to the authors.
“After initiating patients on oral steroid bursts, physicians should be on the lookout for these severe adverse events, particularly within the first month after initiation of steroid therapy,” Dr. Yao said in an interview.
The findings were published online July 6 in Annals of Internal Medicine.
Of the 15,859,129 adult Asians in the Taiwanese database, the study included 2,623,327 adults aged 20-64 years who received single steroid bursts (14 days or less) between Jan. 1, 2013, and Dec. 31, 2015.
Almost 60% of the indications were for skin disorders, such as eczema and urticaria, and for respiratory tract infections, such as sinusitis and acute pharyngitis. Among specialties, dermatology, otolaryngology, family practice, internal medicine, and pediatrics accounted for 88% of prescriptions.
“Our findings are important for physicians and guideline developers because short-term use of oral corticosteroids is common and the real-world safety of this approach remains unclear,” the authors wrote. They acknowledged that the database did not provide information on such potential confounders as disease severity and lifestyle factors, nor did it include children and vulnerable individuals, which may limit the generalizability of the results.
The findings echo those of a 2017 cohort study conducted by researchers at the University of Michigan in Ann Arbor. That study, by Akbar K. Waljee, MD, assistant professor of gastroenterology, University of Michigan, Ann Arbor, and colleagues, included data on more than 1.5 million privately insured U.S. adults. The researchers included somewhat longer steroid bursts of up to 30 days’ duration and found that use of the drugs was associated with a greater than fivefold increased risk for sepsis, a more than threefold increased risk for venous thromboembolism, and a nearly twofold increased risk for fracture within 30 days of starting treatment.
Furthermore, the elevated risk persisted at prednisone-equivalent doses of less than 20 mg/d (IRR, 4.02 for sepsis, 3.61 for venous thromboembolism, and 1.83 for fracture; all P < .001).
The U.S. study also found that during the 3-year period from 2012 to 2014, more than 20% of patients were prescribed short-term oral corticosteroids.
“Both studies indicate that these short-term regimens are more common in the real world than was previously thought and are not risk free,” Dr. Yao said.
Recognition that corticosteroids are associated with adverse events has been building for decades, according to the authors of an editorial that accompanies the new study.
“However, we commonly use short corticosteroid ‘bursts’ for minor ailments despite a lack of evidence for meaningful benefit. We are now learning that bursts as short as 3 days may increase risk for serious AEs, even in young and healthy people,” wrote editorialists Beth I. Wallace, MD, of the Center for Clinical Management Research at the VA Ann Arbor Healthcare System and the Institute for Healthcare Policy and Innovation at Michigan Medicine, Ann Arbor, and Dr. Waljee, who led the 2017 study.
Dr. Wallace and Dr. Waljee drew parallels between corticosteroid bursts and other short-term regimens, such as of antibiotics and opiates, in which prescriber preference and sometimes patient pressure play a role. “All of these treatments have well-defined indications but can cause net harm when used. We can thus conceive of a corticosteroid stewardship model of targeted interventions that aims to reduce inappropriate prescribing,” they wrote.
In an interview, Dr. Wallace, a rheumatologist who prescribes oral steroids fairly frequently, noted that the Taiwan study is the first to investigate steroid bursts. “Up till now, these very short courses have flown under the radar. Clinicians very commonly prescribe short courses to help relieve symptoms of self-limited conditions like bronchitis, and we assume that because the exposure duration is short, the risks are low, especially for patients who are otherwise healthy.”
She warned that the data in the current study indicate that these short bursts – even at the lower end of the 1- to 2-week courses American physicians prescribe most often – carry small but real increases in risk for serious AEs. “And these increases were seen in young, healthy people, not just in people with preexisting conditions,” she said. “So, we might need to start thinking harder about how we are prescribing even these very short courses of steroids and try to use steroids only when their meaningful benefits really outweigh the risk.”
She noted that a patient with a chronic inflammatory condition such as rheumatoid arthritis may benefit substantially from short-term steroids to treat a disease flare. In that specific case, the benefits of short-term steroids may outweigh the risks, Dr. Wallace said.
But not everyone thinks a new strategy is needed. For Whitney A. High, MD, associate professor of dermatology and pathology at the University of Colorado at Denver, Aurora, the overprescribing of short-term corticosteroids is not a problem, and dermatologists are already exercising caution.
“I only prescribe these drugs short term to, at a guess, about 1 in 40 patients and only when a patient is miserable and quality of life is being seriously affected,” he said in an interview. “And that’s something that can’t be measured in a database study like the one from Taiwan but only in a risk-benefit analysis,” he said.
Furthermore, dermatologists have other drugs and technologies in their armamentarium, including topical steroids with occlusion or with wet wraps, phototherapy, phosphodiesterase inhibitors, calcipotriene, methotrexate and other immunosuppressive agents, and biologics. “In fact, many of these agents are specifically referred to as steroid-sparing,” Dr. High said.
Nor does he experience much pressure from patients to prescribe these drugs. “While occasionally I may encounter a patient who places pressure on me for oral steroids, it’s probably not nearly as frequently as providers in other fields are pressured to prescribe antibiotics or narcotics,” he said.
According to the Taiwanese researchers, the next step is to conduct more studies, including clinical trials, to determine optimal use of corticosteroids by monitoring adverse events. In the meantime, for practitioners such as Dr. Wallace and Dr. High, there is ample evidence from several recent studies of the harms of short-term corticosteroids, whereas the benefits for patients with self-limiting conditions remain uncertain. “This and other studies like it quite appropriately remind providers to avoid oral steroids when they’re not necessary and to seek alternatives where possible,” Dr. High said.
The study was supported by the National Health Research Institutes of Taiwan, the Ministry of Science and Technology of Taiwan, the Chang Gung Medical Foundation, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NIH). Dr. Yao has disclosed no relevant financial relationships. Dr. Wu has received grants from GlaxoSmithKline outside the submitted work. The editorialists and Dr. High have disclosed no relevant financial relationships. Dr. Wallace received an NIH grant during the writing of the editorial.
A version of this article originally appeared on Medscape.com.
Extended use of corticosteroids for chronic inflammatory conditions puts patients at risk for serious adverse events (AEs), including cardiovascular disease, osteoporosis, cataracts, and diabetes. Now, a growing body of evidence suggests that even short bursts of these drugs are associated with serious risks.
Most recently, a population-based study of more than 2.6 million people found that taking corticosteroids for 14 days or less was associated with a substantially greater risk for gastrointestinal (GI) bleeding, sepsis, and heart failure, particularly within the first 30 days after therapy.
In the study, Tsung-Chieh Yao, MD, PhD, a professor in the division of allergy, asthma, and rheumatology in the department of pediatrics at Chang Gung Memorial Hospital in Taoyuan, Taiwan, and colleagues used a self-controlled case series to analyze data from Taiwan’s National Health Insurance Research Database of medical claims. They compared patients’ conditions in the period from 5 to 90 days before treatment to conditions from the periods from 5 to 30 days and from 31 to 90 days after therapy.
With a median duration of 3 days of treatment, the incidence rate ratios (IRRs) were 1.80 (95% confidence interval, 1.75-1.84) for GI bleeding, 1.99 (95% CI, 1.70-2.32) for sepsis, and 2.37 (95% CI, 2.13-2.63) for heart failure.
Given the findings, physicians should weigh the benefits against the risks of rare but potentially serious consequences of these anti-inflammatory drugs, according to the authors.
“After initiating patients on oral steroid bursts, physicians should be on the lookout for these severe adverse events, particularly within the first month after initiation of steroid therapy,” Dr. Yao said in an interview.
The findings were published online July 6 in Annals of Internal Medicine.
Of the 15,859,129 adult Asians in the Taiwanese database, the study included 2,623,327 adults aged 20-64 years who received single steroid bursts (14 days or less) between Jan. 1, 2013, and Dec. 31, 2015.
Almost 60% of the indications were for skin disorders, such as eczema and urticaria, and for respiratory tract infections, such as sinusitis and acute pharyngitis. Among specialties, dermatology, otolaryngology, family practice, internal medicine, and pediatrics accounted for 88% of prescriptions.
“Our findings are important for physicians and guideline developers because short-term use of oral corticosteroids is common and the real-world safety of this approach remains unclear,” the authors wrote. They acknowledged that the database did not provide information on such potential confounders as disease severity and lifestyle factors, nor did it include children and vulnerable individuals, which may limit the generalizability of the results.
The findings echo those of a 2017 cohort study conducted by researchers at the University of Michigan in Ann Arbor. That study, by Akbar K. Waljee, MD, assistant professor of gastroenterology, University of Michigan, Ann Arbor, and colleagues, included data on more than 1.5 million privately insured U.S. adults. The researchers included somewhat longer steroid bursts of up to 30 days’ duration and found that use of the drugs was associated with a greater than fivefold increased risk for sepsis, a more than threefold increased risk for venous thromboembolism, and a nearly twofold increased risk for fracture within 30 days of starting treatment.
Furthermore, the elevated risk persisted at prednisone-equivalent doses of less than 20 mg/d (IRR, 4.02 for sepsis, 3.61 for venous thromboembolism, and 1.83 for fracture; all P < .001).
The U.S. study also found that during the 3-year period from 2012 to 2014, more than 20% of patients were prescribed short-term oral corticosteroids.
“Both studies indicate that these short-term regimens are more common in the real world than was previously thought and are not risk free,” Dr. Yao said.
Recognition that corticosteroids are associated with adverse events has been building for decades, according to the authors of an editorial that accompanies the new study.
“However, we commonly use short corticosteroid ‘bursts’ for minor ailments despite a lack of evidence for meaningful benefit. We are now learning that bursts as short as 3 days may increase risk for serious AEs, even in young and healthy people,” wrote editorialists Beth I. Wallace, MD, of the Center for Clinical Management Research at the VA Ann Arbor Healthcare System and the Institute for Healthcare Policy and Innovation at Michigan Medicine, Ann Arbor, and Dr. Waljee, who led the 2017 study.
Dr. Wallace and Dr. Waljee drew parallels between corticosteroid bursts and other short-term regimens, such as of antibiotics and opiates, in which prescriber preference and sometimes patient pressure play a role. “All of these treatments have well-defined indications but can cause net harm when used. We can thus conceive of a corticosteroid stewardship model of targeted interventions that aims to reduce inappropriate prescribing,” they wrote.
In an interview, Dr. Wallace, a rheumatologist who prescribes oral steroids fairly frequently, noted that the Taiwan study is the first to investigate steroid bursts. “Up till now, these very short courses have flown under the radar. Clinicians very commonly prescribe short courses to help relieve symptoms of self-limited conditions like bronchitis, and we assume that because the exposure duration is short, the risks are low, especially for patients who are otherwise healthy.”
She warned that the data in the current study indicate that these short bursts – even at the lower end of the 1- to 2-week courses American physicians prescribe most often – carry small but real increases in risk for serious AEs. “And these increases were seen in young, healthy people, not just in people with preexisting conditions,” she said. “So, we might need to start thinking harder about how we are prescribing even these very short courses of steroids and try to use steroids only when their meaningful benefits really outweigh the risk.”
She noted that a patient with a chronic inflammatory condition such as rheumatoid arthritis may benefit substantially from short-term steroids to treat a disease flare. In that specific case, the benefits of short-term steroids may outweigh the risks, Dr. Wallace said.
But not everyone thinks a new strategy is needed. For Whitney A. High, MD, associate professor of dermatology and pathology at the University of Colorado at Denver, Aurora, the overprescribing of short-term corticosteroids is not a problem, and dermatologists are already exercising caution.
“I only prescribe these drugs short term to, at a guess, about 1 in 40 patients and only when a patient is miserable and quality of life is being seriously affected,” he said in an interview. “And that’s something that can’t be measured in a database study like the one from Taiwan but only in a risk-benefit analysis,” he said.
Furthermore, dermatologists have other drugs and technologies in their armamentarium, including topical steroids with occlusion or with wet wraps, phototherapy, phosphodiesterase inhibitors, calcipotriene, methotrexate and other immunosuppressive agents, and biologics. “In fact, many of these agents are specifically referred to as steroid-sparing,” Dr. High said.
Nor does he experience much pressure from patients to prescribe these drugs. “While occasionally I may encounter a patient who places pressure on me for oral steroids, it’s probably not nearly as frequently as providers in other fields are pressured to prescribe antibiotics or narcotics,” he said.
According to the Taiwanese researchers, the next step is to conduct more studies, including clinical trials, to determine optimal use of corticosteroids by monitoring adverse events. In the meantime, for practitioners such as Dr. Wallace and Dr. High, there is ample evidence from several recent studies of the harms of short-term corticosteroids, whereas the benefits for patients with self-limiting conditions remain uncertain. “This and other studies like it quite appropriately remind providers to avoid oral steroids when they’re not necessary and to seek alternatives where possible,” Dr. High said.
The study was supported by the National Health Research Institutes of Taiwan, the Ministry of Science and Technology of Taiwan, the Chang Gung Medical Foundation, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health (NIH). Dr. Yao has disclosed no relevant financial relationships. Dr. Wu has received grants from GlaxoSmithKline outside the submitted work. The editorialists and Dr. High have disclosed no relevant financial relationships. Dr. Wallace received an NIH grant during the writing of the editorial.
A version of this article originally appeared on Medscape.com.
Nail discoloration
The clinical findings and medical history were consistent with drug-induced hyperpigmentation and minocycline was the likely culprit. Other medications commonly implicated in drug-induced hyperpigmentation include heavy metals (eg, gold, iron, silver), anticonvulsants, hydroxychloroquine, and amiodarone.
Common sites for minocycline pigment deposition—besides the fingernails—include the gingiva, dorsal hands, shins, and old scars. The diagnosis of drug-induced hyperpigmentation is clinical and does not require a biopsy.
While the use of antibiotics for chronic disease can lead to antimicrobial resistance (and should be avoided when possible), certain cases of rosacea may require chronic therapy, and tetracyclines most commonly are used. When minocycline is chosen as a chronic therapy, part of the treatment surveillance should include monitoring for drug-induced hyperpigmentation.
When drug-induced hyperpigmentation does occur, treatment involves discontinuing the offending agent. It can take years for pigment to develop and years for it to resolve—if it completely resolves at all.
In this case, the physician was concerned about the pigmentation worsening or spreading to other sites, so he discontinued the minocycline and prescribed a topical agent for the patient’s rosacea. Ultimately, the patient required occasional use of doxycycline for flares.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Nisar MS, Iyer K, Brodell RT, et al. Minocycline-induced hyperpigmentation: comparison of 3 Q-switched lasers to reverse its effects. Clin Cosmet Investig Dermatol. 2013;6:159-162.

The clinical findings and medical history were consistent with drug-induced hyperpigmentation and minocycline was the likely culprit. Other medications commonly implicated in drug-induced hyperpigmentation include heavy metals (eg, gold, iron, silver), anticonvulsants, hydroxychloroquine, and amiodarone.
Common sites for minocycline pigment deposition—besides the fingernails—include the gingiva, dorsal hands, shins, and old scars. The diagnosis of drug-induced hyperpigmentation is clinical and does not require a biopsy.
While the use of antibiotics for chronic disease can lead to antimicrobial resistance (and should be avoided when possible), certain cases of rosacea may require chronic therapy, and tetracyclines most commonly are used. When minocycline is chosen as a chronic therapy, part of the treatment surveillance should include monitoring for drug-induced hyperpigmentation.
When drug-induced hyperpigmentation does occur, treatment involves discontinuing the offending agent. It can take years for pigment to develop and years for it to resolve—if it completely resolves at all.
In this case, the physician was concerned about the pigmentation worsening or spreading to other sites, so he discontinued the minocycline and prescribed a topical agent for the patient’s rosacea. Ultimately, the patient required occasional use of doxycycline for flares.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

The clinical findings and medical history were consistent with drug-induced hyperpigmentation and minocycline was the likely culprit. Other medications commonly implicated in drug-induced hyperpigmentation include heavy metals (eg, gold, iron, silver), anticonvulsants, hydroxychloroquine, and amiodarone.
Common sites for minocycline pigment deposition—besides the fingernails—include the gingiva, dorsal hands, shins, and old scars. The diagnosis of drug-induced hyperpigmentation is clinical and does not require a biopsy.
While the use of antibiotics for chronic disease can lead to antimicrobial resistance (and should be avoided when possible), certain cases of rosacea may require chronic therapy, and tetracyclines most commonly are used. When minocycline is chosen as a chronic therapy, part of the treatment surveillance should include monitoring for drug-induced hyperpigmentation.
When drug-induced hyperpigmentation does occur, treatment involves discontinuing the offending agent. It can take years for pigment to develop and years for it to resolve—if it completely resolves at all.
In this case, the physician was concerned about the pigmentation worsening or spreading to other sites, so he discontinued the minocycline and prescribed a topical agent for the patient’s rosacea. Ultimately, the patient required occasional use of doxycycline for flares.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
Nisar MS, Iyer K, Brodell RT, et al. Minocycline-induced hyperpigmentation: comparison of 3 Q-switched lasers to reverse its effects. Clin Cosmet Investig Dermatol. 2013;6:159-162.
Nisar MS, Iyer K, Brodell RT, et al. Minocycline-induced hyperpigmentation: comparison of 3 Q-switched lasers to reverse its effects. Clin Cosmet Investig Dermatol. 2013;6:159-162.
Sorting out the many mimickers of psoriasis
“It has an earlier age of onset, usually in infancy, and can occur with the atopic triad that presents with asthma and seasonal allergies as well,” Israel David “Izzy” Andrews, MD, said at the virtual Pediatric Dermatology 2020: Best Practices and Innovations Conference. “There is typically a very strong family history, as this is an autosomal dominant condition, and it’s far more common than psoriasis. The annual incidence is estimated to be 10%-15% of pediatric patients. It has classic areas of involvement depending on the age of the patient, and lesions are intensely pruritic at all times. There is induration and crust, but it’s important to distinguish crust from scale. Whereas crust is dried exudate, and scale is usually secondary to a hyperproliferation of the skin. Initially, treatments (especially topical) are similar and may also delay the formalized diagnosis of either of the two.”
Another psoriasis mimicker, pityriasis rosea, is thought to be secondary to human herpes virus 6 or 7 infection, said Dr. Andrews, of the department of dermatology at Phoenix Children’s Hospital. It typically appears in the teens and tweens and usually presents as a large herald patch or plaque on the trunk. As the herald patch resolves, smaller lesions will develop on the trunk following skin folds. “It’s rarely symptomatic and it’s very short lived, and clears within 6-12 weeks,” Dr. Andrews noted. “It can present with an inverse pattern involving the face, neck, and groin, but sparing the trunk. This variant, termed inverse pityriasis rosea, can be confused with inverse psoriasis, which has a similar distribution. However, the inverse pattern of pityriasis rosea will still resolve in a similar time frame to its more classic variant.”
Pityriasis lichenoides can also be mistaken for psoriasis. The acute form can present with erythematous, scaly papules and plaques, but lesions are often found in different phases of resolution or healing. “This benign lymphoproliferative skin disorder can be very difficult to distinguish from psoriasis and may require a biopsy to rule in or out,” Dr. Andrews said. “It can last months to years and there are few treatments that are effective. It is typically nonresponsive to topical steroids and other treatments that would be more effective for psoriasis, helping to distinguish the two. It is thought to exist in the spectrum with other lymphoproliferative diseases including cutaneous T-cell lymphoma [CTCL]. However, there are only a few cases in the literature that support a transformation from pityriasis lichenoides to CTCL.”
Seborrheic dermatitis is more common than atopic dermatitis and psoriasis, but it can be mistaken for psoriasis. It is caused by an inflammatory response secondary to overgrowth of Malassezia yeast and has a bimodal age distribution. “Seborrheic dermatitis affects babies, teens, and tweens, and can persist into adulthood,” he said. “Infants with cradle cap usually resolve with moisturization, gentle brushing, and occasional antifungal shampoos.” Petaloid seborrheic dermatitis can predominately involve the face with psoriatic-appearing induration, plaques, and varying degrees of scales. “In skin of color, this can be confused with discoid lupus, sarcoidosis, and psoriasis, occasionally requiring a biopsy to distinguish,” said Dr. Andrews, who is also an assistant professor of pediatrics at the Mayo Clinic College of Medicine and Science in Scottsdale, Ariz.
Another psoriasis mimicker, pityriasis amiantacea, is thought to be a more severe form of seborrheic dermatitis. It presents with concretions of scale around hair follicles that are highly adherent and are sometimes called sebopsoriasis. “It may be associated with cutaneous findings of psoriasis elsewhere, but may also be found with secondarily infected atopic dermatitis and tinea capitis; however, in my clinical experience, it is most often found in isolation,” he said. “There may be a seasonal association with exacerbation in warm temperatures, and treatment often consists of humectants like salicylic acid for loosening scale, topical steroids for inflammation, and gentle combing out of scale.”
Infections can also mimic psoriasis. For example, tinea infections are often misdiagnosed as eczema or psoriasis and treated with topical steroids. “This can lead to tinea incognito, making it harder to diagnose either condition without attention to detail,” Dr. Andrews said. “On the body, look for expanding lesions with more raised peripheral edges, and central flattening, giving a classic annular appearance. It’s also important to inquire about family history and contacts including pets, contact sports/mat sports (think yoga, gymnastics, martial arts), or other contacts with similar rashes.” Work-up typically includes a fungal culture and starting empiric oral antifungal medications. “It is important to be able to distinguish scalp psoriasis from tinea capitis to prevent the more inflammatory form of tinea capitis, kerion (a deeper more symptomatic, painful and purulent dermatitis), which can lead to permanent scarring alopecia,” he said.
Bacterial infections can also mimic psoriasis, specifically nonbullous impetigo and ecthyma, the more ulcerative form of impetigo. The most frequent associations are group A Streptococcus, methicillin-susceptible Staphylococcus aureus and methicillin-resistant S. aureus.
Dr. Andrews closed his presentation by noting that tumor necrosis factor–alpha inhibitor–induced psoriasiform drug eruptions can occur in psoriasis-naive patients or unmask a predilection for psoriasis in patients with Crohn’s disease, juvenile idiopathic arthritis, or other autoinflammatory or autoimmune conditions. “They may improve with continued treatment and resolve with switching treatments,” he said. “Early biopsy in psoriasiform drug eruptions can appear like atopic dermatitis on pathology. When suspecting psoriasis in a pediatric patient, it is important to consider the history and physical exam as well as family history and associated comorbidities. While a biopsy may aide in the work-up, diagnosis can be made clinically.”
Dr. Andrews reported having no financial disclosures.
“It has an earlier age of onset, usually in infancy, and can occur with the atopic triad that presents with asthma and seasonal allergies as well,” Israel David “Izzy” Andrews, MD, said at the virtual Pediatric Dermatology 2020: Best Practices and Innovations Conference. “There is typically a very strong family history, as this is an autosomal dominant condition, and it’s far more common than psoriasis. The annual incidence is estimated to be 10%-15% of pediatric patients. It has classic areas of involvement depending on the age of the patient, and lesions are intensely pruritic at all times. There is induration and crust, but it’s important to distinguish crust from scale. Whereas crust is dried exudate, and scale is usually secondary to a hyperproliferation of the skin. Initially, treatments (especially topical) are similar and may also delay the formalized diagnosis of either of the two.”
Another psoriasis mimicker, pityriasis rosea, is thought to be secondary to human herpes virus 6 or 7 infection, said Dr. Andrews, of the department of dermatology at Phoenix Children’s Hospital. It typically appears in the teens and tweens and usually presents as a large herald patch or plaque on the trunk. As the herald patch resolves, smaller lesions will develop on the trunk following skin folds. “It’s rarely symptomatic and it’s very short lived, and clears within 6-12 weeks,” Dr. Andrews noted. “It can present with an inverse pattern involving the face, neck, and groin, but sparing the trunk. This variant, termed inverse pityriasis rosea, can be confused with inverse psoriasis, which has a similar distribution. However, the inverse pattern of pityriasis rosea will still resolve in a similar time frame to its more classic variant.”
Pityriasis lichenoides can also be mistaken for psoriasis. The acute form can present with erythematous, scaly papules and plaques, but lesions are often found in different phases of resolution or healing. “This benign lymphoproliferative skin disorder can be very difficult to distinguish from psoriasis and may require a biopsy to rule in or out,” Dr. Andrews said. “It can last months to years and there are few treatments that are effective. It is typically nonresponsive to topical steroids and other treatments that would be more effective for psoriasis, helping to distinguish the two. It is thought to exist in the spectrum with other lymphoproliferative diseases including cutaneous T-cell lymphoma [CTCL]. However, there are only a few cases in the literature that support a transformation from pityriasis lichenoides to CTCL.”
Seborrheic dermatitis is more common than atopic dermatitis and psoriasis, but it can be mistaken for psoriasis. It is caused by an inflammatory response secondary to overgrowth of Malassezia yeast and has a bimodal age distribution. “Seborrheic dermatitis affects babies, teens, and tweens, and can persist into adulthood,” he said. “Infants with cradle cap usually resolve with moisturization, gentle brushing, and occasional antifungal shampoos.” Petaloid seborrheic dermatitis can predominately involve the face with psoriatic-appearing induration, plaques, and varying degrees of scales. “In skin of color, this can be confused with discoid lupus, sarcoidosis, and psoriasis, occasionally requiring a biopsy to distinguish,” said Dr. Andrews, who is also an assistant professor of pediatrics at the Mayo Clinic College of Medicine and Science in Scottsdale, Ariz.
Another psoriasis mimicker, pityriasis amiantacea, is thought to be a more severe form of seborrheic dermatitis. It presents with concretions of scale around hair follicles that are highly adherent and are sometimes called sebopsoriasis. “It may be associated with cutaneous findings of psoriasis elsewhere, but may also be found with secondarily infected atopic dermatitis and tinea capitis; however, in my clinical experience, it is most often found in isolation,” he said. “There may be a seasonal association with exacerbation in warm temperatures, and treatment often consists of humectants like salicylic acid for loosening scale, topical steroids for inflammation, and gentle combing out of scale.”
Infections can also mimic psoriasis. For example, tinea infections are often misdiagnosed as eczema or psoriasis and treated with topical steroids. “This can lead to tinea incognito, making it harder to diagnose either condition without attention to detail,” Dr. Andrews said. “On the body, look for expanding lesions with more raised peripheral edges, and central flattening, giving a classic annular appearance. It’s also important to inquire about family history and contacts including pets, contact sports/mat sports (think yoga, gymnastics, martial arts), or other contacts with similar rashes.” Work-up typically includes a fungal culture and starting empiric oral antifungal medications. “It is important to be able to distinguish scalp psoriasis from tinea capitis to prevent the more inflammatory form of tinea capitis, kerion (a deeper more symptomatic, painful and purulent dermatitis), which can lead to permanent scarring alopecia,” he said.
Bacterial infections can also mimic psoriasis, specifically nonbullous impetigo and ecthyma, the more ulcerative form of impetigo. The most frequent associations are group A Streptococcus, methicillin-susceptible Staphylococcus aureus and methicillin-resistant S. aureus.
Dr. Andrews closed his presentation by noting that tumor necrosis factor–alpha inhibitor–induced psoriasiform drug eruptions can occur in psoriasis-naive patients or unmask a predilection for psoriasis in patients with Crohn’s disease, juvenile idiopathic arthritis, or other autoinflammatory or autoimmune conditions. “They may improve with continued treatment and resolve with switching treatments,” he said. “Early biopsy in psoriasiform drug eruptions can appear like atopic dermatitis on pathology. When suspecting psoriasis in a pediatric patient, it is important to consider the history and physical exam as well as family history and associated comorbidities. While a biopsy may aide in the work-up, diagnosis can be made clinically.”
Dr. Andrews reported having no financial disclosures.
“It has an earlier age of onset, usually in infancy, and can occur with the atopic triad that presents with asthma and seasonal allergies as well,” Israel David “Izzy” Andrews, MD, said at the virtual Pediatric Dermatology 2020: Best Practices and Innovations Conference. “There is typically a very strong family history, as this is an autosomal dominant condition, and it’s far more common than psoriasis. The annual incidence is estimated to be 10%-15% of pediatric patients. It has classic areas of involvement depending on the age of the patient, and lesions are intensely pruritic at all times. There is induration and crust, but it’s important to distinguish crust from scale. Whereas crust is dried exudate, and scale is usually secondary to a hyperproliferation of the skin. Initially, treatments (especially topical) are similar and may also delay the formalized diagnosis of either of the two.”
Another psoriasis mimicker, pityriasis rosea, is thought to be secondary to human herpes virus 6 or 7 infection, said Dr. Andrews, of the department of dermatology at Phoenix Children’s Hospital. It typically appears in the teens and tweens and usually presents as a large herald patch or plaque on the trunk. As the herald patch resolves, smaller lesions will develop on the trunk following skin folds. “It’s rarely symptomatic and it’s very short lived, and clears within 6-12 weeks,” Dr. Andrews noted. “It can present with an inverse pattern involving the face, neck, and groin, but sparing the trunk. This variant, termed inverse pityriasis rosea, can be confused with inverse psoriasis, which has a similar distribution. However, the inverse pattern of pityriasis rosea will still resolve in a similar time frame to its more classic variant.”
Pityriasis lichenoides can also be mistaken for psoriasis. The acute form can present with erythematous, scaly papules and plaques, but lesions are often found in different phases of resolution or healing. “This benign lymphoproliferative skin disorder can be very difficult to distinguish from psoriasis and may require a biopsy to rule in or out,” Dr. Andrews said. “It can last months to years and there are few treatments that are effective. It is typically nonresponsive to topical steroids and other treatments that would be more effective for psoriasis, helping to distinguish the two. It is thought to exist in the spectrum with other lymphoproliferative diseases including cutaneous T-cell lymphoma [CTCL]. However, there are only a few cases in the literature that support a transformation from pityriasis lichenoides to CTCL.”
Seborrheic dermatitis is more common than atopic dermatitis and psoriasis, but it can be mistaken for psoriasis. It is caused by an inflammatory response secondary to overgrowth of Malassezia yeast and has a bimodal age distribution. “Seborrheic dermatitis affects babies, teens, and tweens, and can persist into adulthood,” he said. “Infants with cradle cap usually resolve with moisturization, gentle brushing, and occasional antifungal shampoos.” Petaloid seborrheic dermatitis can predominately involve the face with psoriatic-appearing induration, plaques, and varying degrees of scales. “In skin of color, this can be confused with discoid lupus, sarcoidosis, and psoriasis, occasionally requiring a biopsy to distinguish,” said Dr. Andrews, who is also an assistant professor of pediatrics at the Mayo Clinic College of Medicine and Science in Scottsdale, Ariz.
Another psoriasis mimicker, pityriasis amiantacea, is thought to be a more severe form of seborrheic dermatitis. It presents with concretions of scale around hair follicles that are highly adherent and are sometimes called sebopsoriasis. “It may be associated with cutaneous findings of psoriasis elsewhere, but may also be found with secondarily infected atopic dermatitis and tinea capitis; however, in my clinical experience, it is most often found in isolation,” he said. “There may be a seasonal association with exacerbation in warm temperatures, and treatment often consists of humectants like salicylic acid for loosening scale, topical steroids for inflammation, and gentle combing out of scale.”
Infections can also mimic psoriasis. For example, tinea infections are often misdiagnosed as eczema or psoriasis and treated with topical steroids. “This can lead to tinea incognito, making it harder to diagnose either condition without attention to detail,” Dr. Andrews said. “On the body, look for expanding lesions with more raised peripheral edges, and central flattening, giving a classic annular appearance. It’s also important to inquire about family history and contacts including pets, contact sports/mat sports (think yoga, gymnastics, martial arts), or other contacts with similar rashes.” Work-up typically includes a fungal culture and starting empiric oral antifungal medications. “It is important to be able to distinguish scalp psoriasis from tinea capitis to prevent the more inflammatory form of tinea capitis, kerion (a deeper more symptomatic, painful and purulent dermatitis), which can lead to permanent scarring alopecia,” he said.
Bacterial infections can also mimic psoriasis, specifically nonbullous impetigo and ecthyma, the more ulcerative form of impetigo. The most frequent associations are group A Streptococcus, methicillin-susceptible Staphylococcus aureus and methicillin-resistant S. aureus.
Dr. Andrews closed his presentation by noting that tumor necrosis factor–alpha inhibitor–induced psoriasiform drug eruptions can occur in psoriasis-naive patients or unmask a predilection for psoriasis in patients with Crohn’s disease, juvenile idiopathic arthritis, or other autoinflammatory or autoimmune conditions. “They may improve with continued treatment and resolve with switching treatments,” he said. “Early biopsy in psoriasiform drug eruptions can appear like atopic dermatitis on pathology. When suspecting psoriasis in a pediatric patient, it is important to consider the history and physical exam as well as family history and associated comorbidities. While a biopsy may aide in the work-up, diagnosis can be made clinically.”
Dr. Andrews reported having no financial disclosures.
FROM PEDIATRIC DERMATOLOGY 2020