User login
Bumps on the thighs

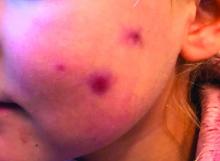
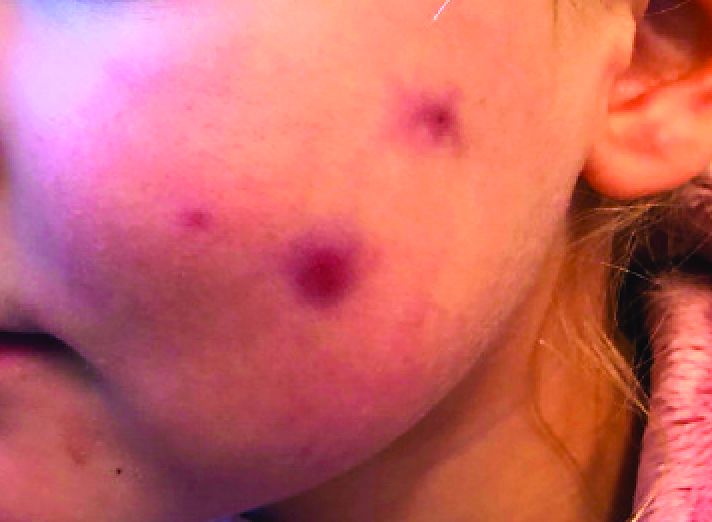
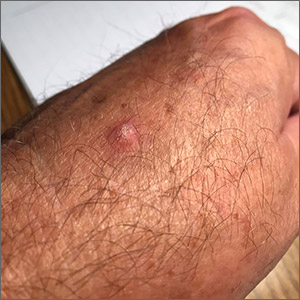
The photograph submitted for the telemedicine visit showed 2 classic umbilicated lesions and 1 dome-shaped papule consistent with molluscum contagiosum. Not all skin conditions can be diagnosed or treated via telehealth, but with a careful history, cooperative patients (and parents in this case), and photos taken on newer cell phones or digital cameras, many conditions can be diagnosed and managed appropriately.
Molluscum contagiosum is caused by the Molluscipox genus poxvirus and Is commonly seen in preschool and school-aged children. It can be passed through direct contact with infected individuals or spread by fomites. (In this case, the child may have picked up the virus by sharing a towel with an infected individual.)
The flesh-colored lesions are umbilicated or popular, and occur in clusters on the trunk, face, and extremities. Typically, the lesions will resolve spontaneously, but it may take several weeks to many months for resolution.
Given this lengthy time for spontaneous resolution, the risk of spreading to family members or other contacts, and the skin’s appearance, many patients choose to treat the lesions. Treatment options include curettage, cryosurgery, and laser. Available topical destructive agents include podophyllotoxin, trichloroacetic acid, benzoyl peroxide, potassium hydroxide, and cantharidin (which is from the blister beetle and often difficult to obtain). There also are naturopathic topical products and immune system modulators, including topical imiquimod. These treatments are commonly used, but are off-label for the treatment of molluscum contagiosum.
The family was counseled that there is debate about the effectiveness of imiquimod for molluscum contagiosum, but that some studies find it to be useful. In this case, the mother chose a prescription for imiquimod cream 5%, to be applied 3 times weekly at bedtime until the lesions resolved. (The cream can be used for up to 16 weeks.) The family was advised that erythema and irritation are expected adverse effects at the application site.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Badavanis G, Pasmatzi E, Monastirli A, et al. Topical imiquimod is an effective and safe drug for molluscum contagiosum in children. Acta Dermatovenerol Croat. 2017;25:164-166.

The photograph submitted for the telemedicine visit showed 2 classic umbilicated lesions and 1 dome-shaped papule consistent with molluscum contagiosum. Not all skin conditions can be diagnosed or treated via telehealth, but with a careful history, cooperative patients (and parents in this case), and photos taken on newer cell phones or digital cameras, many conditions can be diagnosed and managed appropriately.
Molluscum contagiosum is caused by the Molluscipox genus poxvirus and Is commonly seen in preschool and school-aged children. It can be passed through direct contact with infected individuals or spread by fomites. (In this case, the child may have picked up the virus by sharing a towel with an infected individual.)
The flesh-colored lesions are umbilicated or popular, and occur in clusters on the trunk, face, and extremities. Typically, the lesions will resolve spontaneously, but it may take several weeks to many months for resolution.
Given this lengthy time for spontaneous resolution, the risk of spreading to family members or other contacts, and the skin’s appearance, many patients choose to treat the lesions. Treatment options include curettage, cryosurgery, and laser. Available topical destructive agents include podophyllotoxin, trichloroacetic acid, benzoyl peroxide, potassium hydroxide, and cantharidin (which is from the blister beetle and often difficult to obtain). There also are naturopathic topical products and immune system modulators, including topical imiquimod. These treatments are commonly used, but are off-label for the treatment of molluscum contagiosum.
The family was counseled that there is debate about the effectiveness of imiquimod for molluscum contagiosum, but that some studies find it to be useful. In this case, the mother chose a prescription for imiquimod cream 5%, to be applied 3 times weekly at bedtime until the lesions resolved. (The cream can be used for up to 16 weeks.) The family was advised that erythema and irritation are expected adverse effects at the application site.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The photograph submitted for the telemedicine visit showed 2 classic umbilicated lesions and 1 dome-shaped papule consistent with molluscum contagiosum. Not all skin conditions can be diagnosed or treated via telehealth, but with a careful history, cooperative patients (and parents in this case), and photos taken on newer cell phones or digital cameras, many conditions can be diagnosed and managed appropriately.
Molluscum contagiosum is caused by the Molluscipox genus poxvirus and Is commonly seen in preschool and school-aged children. It can be passed through direct contact with infected individuals or spread by fomites. (In this case, the child may have picked up the virus by sharing a towel with an infected individual.)
The flesh-colored lesions are umbilicated or popular, and occur in clusters on the trunk, face, and extremities. Typically, the lesions will resolve spontaneously, but it may take several weeks to many months for resolution.
Given this lengthy time for spontaneous resolution, the risk of spreading to family members or other contacts, and the skin’s appearance, many patients choose to treat the lesions. Treatment options include curettage, cryosurgery, and laser. Available topical destructive agents include podophyllotoxin, trichloroacetic acid, benzoyl peroxide, potassium hydroxide, and cantharidin (which is from the blister beetle and often difficult to obtain). There also are naturopathic topical products and immune system modulators, including topical imiquimod. These treatments are commonly used, but are off-label for the treatment of molluscum contagiosum.
The family was counseled that there is debate about the effectiveness of imiquimod for molluscum contagiosum, but that some studies find it to be useful. In this case, the mother chose a prescription for imiquimod cream 5%, to be applied 3 times weekly at bedtime until the lesions resolved. (The cream can be used for up to 16 weeks.) The family was advised that erythema and irritation are expected adverse effects at the application site.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Badavanis G, Pasmatzi E, Monastirli A, et al. Topical imiquimod is an effective and safe drug for molluscum contagiosum in children. Acta Dermatovenerol Croat. 2017;25:164-166.
Badavanis G, Pasmatzi E, Monastirli A, et al. Topical imiquimod is an effective and safe drug for molluscum contagiosum in children. Acta Dermatovenerol Croat. 2017;25:164-166.
Mapping melasma management
Melasma has such a high recurrence rate that, once the facial hyperpigmentation has been cleared, it’s best that treatment never entirely stops, Amit G. Pandya, MD, said at the virtual annual meeting of the American Academy of Dermatology.
He recommended alternating between a less-intensive maintenance therapy regimen in the winter months and an acute care regimen in the sunnier summer months. But . And that is largely a matter of location.
Location, location, location
Melasma has a distinctive symmetric bilateral distribution: “Melasma likes the central area of the forehead, whereas the lateral areas of the forehead are more involved in lichen planus pigmentosus. Melanoma likes the area above the eyebrow or under the eyebrow. However, it does not go below the superior orbital rim or above the inferior orbital rim,”said Dr. Pandya, a dermatologist at the Palo Alto Medical Foundation in Sunnyvale, Calif., who is also on the faculty at the University of Texas Southwestern Medical Center, Dallas.
Melasma is common on the bridge of the nose, but usually not along the nasolabial fold, where hyperpigmentation is much more likely to be due to seborrheic dermatitis or drug-induced hyperpigmentation. Melasma doesn’t affect the tip of the nose; that’s more likely a sign of sarcoidosis or drug-induced hyperpigmentation. Melasma is common on the zygomatic prominence, while acanthosis nigricans favors the concave area below the zygomatic prominence. And melasma stays above the mandible; pigmentation below the mandible is more suggestive of poikiloderma of Civatte. Lentigines are scattered broadly across sun-exposed areas of the face. They also tend to be less symmetrical than melasma, the dermatologist continued.
Acute treatment
Dr. Pandya’s acute treatment algorithm begins with topical 4% hydroquinone in patients who’ve never been on it before. A response to the drug, which blocks the tyrosine-to-melanin pathway, takes 4-6 weeks, with maximum effect not seen until 3-6 months or longer. Bluish-grey ochronosis is a rare side effect at the 4% concentration but becomes more common at higher concentrations or when the drug is used in combination therapy.
“Hydroquinone is a workhorse, the oldest and most effective depigmenting agent,” he said.
If the patient hasn’t responded positively by 3 months, Dr. Pandya moves on to daily use of the triple-drug combination of fluocinolone acetonide 0.01%/hydroquinone 4%/tretinoin 0.05% known as Tri-Luma, a kinder, gentler descendant of the 45-year-old Kligman-Willis compounded formula comprised of 0.1% dexamethasone, 5% hydroquinone, and 0.1% tretinoin.
If Tri-Luma also proves ineffective, Dr. Pandya turns to oral tranexamic acid. This is off-label therapy for the drug, a plasmin inhibitor, which is approved for the treatment of menorrhagia. But oral tranexamic acid is widely used for treatment of melasma in East Asia, and Dr. Pandya and others have evaluated it in placebo-controlled clinical trials. His conclusion is that oral tranexamic acid appears to be safe and effective for treatment of melasma.
“The drug is not approved for melasma, it’s approved for menorrhagia, so every doctor has to decide how much risk they want to take. The evidence suggests 500 mg per day is a good dose,” he said.
The collective clinical trials experience with oral tranexamic acid for melasma shows a side effect profile consisting of mild GI upset, headache, and myalgia. While increased thromboembolic risk is a theoretic concern, it hasn’t been an issue in the published studies, which typically exclude patients with a history of thromboembolic disease from enrollment. Patient satisfaction with the oral agent is high, according to Dr. Pandya.
In one randomized, open-label, 40-patient study, oral tranexamic acid plus a triple-combination cream featuring fluocinolone 0.01%, hydroquinone 2%, and tretinoin 0.05%, applied once a day, was significantly more effective and faster-acting than the topical therapy alone. At 8 weeks, the dual-therapy group averaged an 88% improvement in the Melasma Activity and Severity Index (MASI) scores, compared with 55% with the topical therapy alone (Indian J Dermatol. Sep-Oct 2015;60[5]:520).
Cysteamine 5% cream, which is available over the counter as Cyspera but is pricey, showed promising efficacy in a 40-patient, randomized, double-blind trial (J Dermatolog Treat. 2018 Mar;29[2]:182-9). Dr. Pandya said he’s looking forward to seeing further studies.
Chemical peels can be used, but multiple treatment sessions using a superficial peeling agent are required, and even then “the efficacy is usually not profound,” according to Dr. Pandya. Together with two colleagues he recently published a comprehensive systematic review of 113 published studies of all treatments for melasma in nearly 7,000 patients (Am J Clin Dermatol. 2020 Apr;21(2):173-225).
Newer lasers with various pulse lengths, fluences, wave lengths, and treatment frequency show “some promise,” but there have also been published reports of hypopigmentation and rebound hyperpigmentation. The optimal laser regimen remains elusive, he said.
Maintenance therapy
Dr. Pandya usually switches from hydroquinone to a different topical tyrosinase inhibitor for maintenance therapy, such as kojic acid, arbutin, or azelaic acid, all available OTC in many formulations. Alternatively, he might drop down to 2% hydroquinone for the winter months. Another option is triple-combination cream applied two or three times per week. A topical formulation of tranexamic acid is available, but studies of this agent in patients with melasma have yielded mixed results.
“I don’t think topical tranexamic acid is going to harm the patient, but I don’t think the efficacy is as good as with oral tranexamic acid,” he said.
Slap that melasma in irons
A comprehensive melasma management plan requires year-round frequent daily application of a broad spectrum sunscreen. And since it’s now evident that visible-wavelength light can worsen melasma through mechanisms similar to UVA and UVB, which are long recognized as the major drivers of the hyperpigmentation disorder, serious consideration should be given to the use of a tinted broad-spectrum sunscreen or makeup containing more than 3% iron oxide, which blocks visible light. In contrast, zinc oxide does not, Dr. Pandya noted.
In one influential study, aminolevulinic acid was applied on the arms of 20 patients; two sunscreens were applied on areas where the ALA was applied, and on one area, no sunscreen was applied. The minimal phototoxic dose of visible blue light was doubled with application of a broad-spectrum sunscreen containing titanium dioxide, zinc oxide, and 0.2% iron oxide, compared with no sunscreen, but increased 21-fold using a sunscreen containing titanium dioxide, zinc oxide, and 3.2% iron oxide (Dermatol Surg. 2008 Nov;34[11]:1469-76).
Moreover, in a double-blind, randomized trial including 61 patients with melasma, all on background 4% hydroquinone, those assigned to a broad-spectrum sunscreen containing iron oxide had a 78% improvement in MASI scores at 8 weeks, compared with a 62% improvement with a broad-spectrum UV-only sunscreen. Both sunscreens had a sun protection factor of at least 50 (Photodermatol Photoimmunol Photomed. 2014 Feb;30[1]:35-42).
Numerous sunscreen and makeup products containing more than 3% iron oxide are available OTC in various tints. It’s a matter of finding a color that matches the patient’s skin.
Concern has been raised that exposure to the visible blue light emitted by computer screens and cell phones could worsen melasma. Dr. Pandya noted that reassurance on that score was recently provided by French investigators. They measured the intensity of visible light at the wavelengths emitted by computer screens and laptops and determined that it was 100- to 1,000-fold less than sunlight in the same spectrum. They also conducted a prospective, randomized, split-face trial in 12 melasma patients. One side of the face was exposed to the visible blue light at the same wavelengths emitted by device screens, but at far greater intensity. Blinded evaluators found no split-face difference in modified MASI scores.
“These results suggest that at a 20-cm distance, a maximized use of a high-intensity computer screen for 8 hours per day during a 5-day period does not worsen melasma lesions. Although it is very unlikely that similar exposure during a longer period would start to affect melasma lesions, such a possibility cannot be ruled out,” according to the investigators (J Am Acad Dermatol. 2019 Dec 27;S0190-9622(19)33324-9. doi: 10.1016/j.jaad.2019.12.047).
Dr. Pandya reported serving as a consultant to Incyte, Pfizer, Viela Bio, and Villaris.
Melasma has such a high recurrence rate that, once the facial hyperpigmentation has been cleared, it’s best that treatment never entirely stops, Amit G. Pandya, MD, said at the virtual annual meeting of the American Academy of Dermatology.
He recommended alternating between a less-intensive maintenance therapy regimen in the winter months and an acute care regimen in the sunnier summer months. But . And that is largely a matter of location.
Location, location, location
Melasma has a distinctive symmetric bilateral distribution: “Melasma likes the central area of the forehead, whereas the lateral areas of the forehead are more involved in lichen planus pigmentosus. Melanoma likes the area above the eyebrow or under the eyebrow. However, it does not go below the superior orbital rim or above the inferior orbital rim,”said Dr. Pandya, a dermatologist at the Palo Alto Medical Foundation in Sunnyvale, Calif., who is also on the faculty at the University of Texas Southwestern Medical Center, Dallas.
Melasma is common on the bridge of the nose, but usually not along the nasolabial fold, where hyperpigmentation is much more likely to be due to seborrheic dermatitis or drug-induced hyperpigmentation. Melasma doesn’t affect the tip of the nose; that’s more likely a sign of sarcoidosis or drug-induced hyperpigmentation. Melasma is common on the zygomatic prominence, while acanthosis nigricans favors the concave area below the zygomatic prominence. And melasma stays above the mandible; pigmentation below the mandible is more suggestive of poikiloderma of Civatte. Lentigines are scattered broadly across sun-exposed areas of the face. They also tend to be less symmetrical than melasma, the dermatologist continued.
Acute treatment
Dr. Pandya’s acute treatment algorithm begins with topical 4% hydroquinone in patients who’ve never been on it before. A response to the drug, which blocks the tyrosine-to-melanin pathway, takes 4-6 weeks, with maximum effect not seen until 3-6 months or longer. Bluish-grey ochronosis is a rare side effect at the 4% concentration but becomes more common at higher concentrations or when the drug is used in combination therapy.
“Hydroquinone is a workhorse, the oldest and most effective depigmenting agent,” he said.
If the patient hasn’t responded positively by 3 months, Dr. Pandya moves on to daily use of the triple-drug combination of fluocinolone acetonide 0.01%/hydroquinone 4%/tretinoin 0.05% known as Tri-Luma, a kinder, gentler descendant of the 45-year-old Kligman-Willis compounded formula comprised of 0.1% dexamethasone, 5% hydroquinone, and 0.1% tretinoin.
If Tri-Luma also proves ineffective, Dr. Pandya turns to oral tranexamic acid. This is off-label therapy for the drug, a plasmin inhibitor, which is approved for the treatment of menorrhagia. But oral tranexamic acid is widely used for treatment of melasma in East Asia, and Dr. Pandya and others have evaluated it in placebo-controlled clinical trials. His conclusion is that oral tranexamic acid appears to be safe and effective for treatment of melasma.
“The drug is not approved for melasma, it’s approved for menorrhagia, so every doctor has to decide how much risk they want to take. The evidence suggests 500 mg per day is a good dose,” he said.
The collective clinical trials experience with oral tranexamic acid for melasma shows a side effect profile consisting of mild GI upset, headache, and myalgia. While increased thromboembolic risk is a theoretic concern, it hasn’t been an issue in the published studies, which typically exclude patients with a history of thromboembolic disease from enrollment. Patient satisfaction with the oral agent is high, according to Dr. Pandya.
In one randomized, open-label, 40-patient study, oral tranexamic acid plus a triple-combination cream featuring fluocinolone 0.01%, hydroquinone 2%, and tretinoin 0.05%, applied once a day, was significantly more effective and faster-acting than the topical therapy alone. At 8 weeks, the dual-therapy group averaged an 88% improvement in the Melasma Activity and Severity Index (MASI) scores, compared with 55% with the topical therapy alone (Indian J Dermatol. Sep-Oct 2015;60[5]:520).
Cysteamine 5% cream, which is available over the counter as Cyspera but is pricey, showed promising efficacy in a 40-patient, randomized, double-blind trial (J Dermatolog Treat. 2018 Mar;29[2]:182-9). Dr. Pandya said he’s looking forward to seeing further studies.
Chemical peels can be used, but multiple treatment sessions using a superficial peeling agent are required, and even then “the efficacy is usually not profound,” according to Dr. Pandya. Together with two colleagues he recently published a comprehensive systematic review of 113 published studies of all treatments for melasma in nearly 7,000 patients (Am J Clin Dermatol. 2020 Apr;21(2):173-225).
Newer lasers with various pulse lengths, fluences, wave lengths, and treatment frequency show “some promise,” but there have also been published reports of hypopigmentation and rebound hyperpigmentation. The optimal laser regimen remains elusive, he said.
Maintenance therapy
Dr. Pandya usually switches from hydroquinone to a different topical tyrosinase inhibitor for maintenance therapy, such as kojic acid, arbutin, or azelaic acid, all available OTC in many formulations. Alternatively, he might drop down to 2% hydroquinone for the winter months. Another option is triple-combination cream applied two or three times per week. A topical formulation of tranexamic acid is available, but studies of this agent in patients with melasma have yielded mixed results.
“I don’t think topical tranexamic acid is going to harm the patient, but I don’t think the efficacy is as good as with oral tranexamic acid,” he said.
Slap that melasma in irons
A comprehensive melasma management plan requires year-round frequent daily application of a broad spectrum sunscreen. And since it’s now evident that visible-wavelength light can worsen melasma through mechanisms similar to UVA and UVB, which are long recognized as the major drivers of the hyperpigmentation disorder, serious consideration should be given to the use of a tinted broad-spectrum sunscreen or makeup containing more than 3% iron oxide, which blocks visible light. In contrast, zinc oxide does not, Dr. Pandya noted.
In one influential study, aminolevulinic acid was applied on the arms of 20 patients; two sunscreens were applied on areas where the ALA was applied, and on one area, no sunscreen was applied. The minimal phototoxic dose of visible blue light was doubled with application of a broad-spectrum sunscreen containing titanium dioxide, zinc oxide, and 0.2% iron oxide, compared with no sunscreen, but increased 21-fold using a sunscreen containing titanium dioxide, zinc oxide, and 3.2% iron oxide (Dermatol Surg. 2008 Nov;34[11]:1469-76).
Moreover, in a double-blind, randomized trial including 61 patients with melasma, all on background 4% hydroquinone, those assigned to a broad-spectrum sunscreen containing iron oxide had a 78% improvement in MASI scores at 8 weeks, compared with a 62% improvement with a broad-spectrum UV-only sunscreen. Both sunscreens had a sun protection factor of at least 50 (Photodermatol Photoimmunol Photomed. 2014 Feb;30[1]:35-42).
Numerous sunscreen and makeup products containing more than 3% iron oxide are available OTC in various tints. It’s a matter of finding a color that matches the patient’s skin.
Concern has been raised that exposure to the visible blue light emitted by computer screens and cell phones could worsen melasma. Dr. Pandya noted that reassurance on that score was recently provided by French investigators. They measured the intensity of visible light at the wavelengths emitted by computer screens and laptops and determined that it was 100- to 1,000-fold less than sunlight in the same spectrum. They also conducted a prospective, randomized, split-face trial in 12 melasma patients. One side of the face was exposed to the visible blue light at the same wavelengths emitted by device screens, but at far greater intensity. Blinded evaluators found no split-face difference in modified MASI scores.
“These results suggest that at a 20-cm distance, a maximized use of a high-intensity computer screen for 8 hours per day during a 5-day period does not worsen melasma lesions. Although it is very unlikely that similar exposure during a longer period would start to affect melasma lesions, such a possibility cannot be ruled out,” according to the investigators (J Am Acad Dermatol. 2019 Dec 27;S0190-9622(19)33324-9. doi: 10.1016/j.jaad.2019.12.047).
Dr. Pandya reported serving as a consultant to Incyte, Pfizer, Viela Bio, and Villaris.
Melasma has such a high recurrence rate that, once the facial hyperpigmentation has been cleared, it’s best that treatment never entirely stops, Amit G. Pandya, MD, said at the virtual annual meeting of the American Academy of Dermatology.
He recommended alternating between a less-intensive maintenance therapy regimen in the winter months and an acute care regimen in the sunnier summer months. But . And that is largely a matter of location.
Location, location, location
Melasma has a distinctive symmetric bilateral distribution: “Melasma likes the central area of the forehead, whereas the lateral areas of the forehead are more involved in lichen planus pigmentosus. Melanoma likes the area above the eyebrow or under the eyebrow. However, it does not go below the superior orbital rim or above the inferior orbital rim,”said Dr. Pandya, a dermatologist at the Palo Alto Medical Foundation in Sunnyvale, Calif., who is also on the faculty at the University of Texas Southwestern Medical Center, Dallas.
Melasma is common on the bridge of the nose, but usually not along the nasolabial fold, where hyperpigmentation is much more likely to be due to seborrheic dermatitis or drug-induced hyperpigmentation. Melasma doesn’t affect the tip of the nose; that’s more likely a sign of sarcoidosis or drug-induced hyperpigmentation. Melasma is common on the zygomatic prominence, while acanthosis nigricans favors the concave area below the zygomatic prominence. And melasma stays above the mandible; pigmentation below the mandible is more suggestive of poikiloderma of Civatte. Lentigines are scattered broadly across sun-exposed areas of the face. They also tend to be less symmetrical than melasma, the dermatologist continued.
Acute treatment
Dr. Pandya’s acute treatment algorithm begins with topical 4% hydroquinone in patients who’ve never been on it before. A response to the drug, which blocks the tyrosine-to-melanin pathway, takes 4-6 weeks, with maximum effect not seen until 3-6 months or longer. Bluish-grey ochronosis is a rare side effect at the 4% concentration but becomes more common at higher concentrations or when the drug is used in combination therapy.
“Hydroquinone is a workhorse, the oldest and most effective depigmenting agent,” he said.
If the patient hasn’t responded positively by 3 months, Dr. Pandya moves on to daily use of the triple-drug combination of fluocinolone acetonide 0.01%/hydroquinone 4%/tretinoin 0.05% known as Tri-Luma, a kinder, gentler descendant of the 45-year-old Kligman-Willis compounded formula comprised of 0.1% dexamethasone, 5% hydroquinone, and 0.1% tretinoin.
If Tri-Luma also proves ineffective, Dr. Pandya turns to oral tranexamic acid. This is off-label therapy for the drug, a plasmin inhibitor, which is approved for the treatment of menorrhagia. But oral tranexamic acid is widely used for treatment of melasma in East Asia, and Dr. Pandya and others have evaluated it in placebo-controlled clinical trials. His conclusion is that oral tranexamic acid appears to be safe and effective for treatment of melasma.
“The drug is not approved for melasma, it’s approved for menorrhagia, so every doctor has to decide how much risk they want to take. The evidence suggests 500 mg per day is a good dose,” he said.
The collective clinical trials experience with oral tranexamic acid for melasma shows a side effect profile consisting of mild GI upset, headache, and myalgia. While increased thromboembolic risk is a theoretic concern, it hasn’t been an issue in the published studies, which typically exclude patients with a history of thromboembolic disease from enrollment. Patient satisfaction with the oral agent is high, according to Dr. Pandya.
In one randomized, open-label, 40-patient study, oral tranexamic acid plus a triple-combination cream featuring fluocinolone 0.01%, hydroquinone 2%, and tretinoin 0.05%, applied once a day, was significantly more effective and faster-acting than the topical therapy alone. At 8 weeks, the dual-therapy group averaged an 88% improvement in the Melasma Activity and Severity Index (MASI) scores, compared with 55% with the topical therapy alone (Indian J Dermatol. Sep-Oct 2015;60[5]:520).
Cysteamine 5% cream, which is available over the counter as Cyspera but is pricey, showed promising efficacy in a 40-patient, randomized, double-blind trial (J Dermatolog Treat. 2018 Mar;29[2]:182-9). Dr. Pandya said he’s looking forward to seeing further studies.
Chemical peels can be used, but multiple treatment sessions using a superficial peeling agent are required, and even then “the efficacy is usually not profound,” according to Dr. Pandya. Together with two colleagues he recently published a comprehensive systematic review of 113 published studies of all treatments for melasma in nearly 7,000 patients (Am J Clin Dermatol. 2020 Apr;21(2):173-225).
Newer lasers with various pulse lengths, fluences, wave lengths, and treatment frequency show “some promise,” but there have also been published reports of hypopigmentation and rebound hyperpigmentation. The optimal laser regimen remains elusive, he said.
Maintenance therapy
Dr. Pandya usually switches from hydroquinone to a different topical tyrosinase inhibitor for maintenance therapy, such as kojic acid, arbutin, or azelaic acid, all available OTC in many formulations. Alternatively, he might drop down to 2% hydroquinone for the winter months. Another option is triple-combination cream applied two or three times per week. A topical formulation of tranexamic acid is available, but studies of this agent in patients with melasma have yielded mixed results.
“I don’t think topical tranexamic acid is going to harm the patient, but I don’t think the efficacy is as good as with oral tranexamic acid,” he said.
Slap that melasma in irons
A comprehensive melasma management plan requires year-round frequent daily application of a broad spectrum sunscreen. And since it’s now evident that visible-wavelength light can worsen melasma through mechanisms similar to UVA and UVB, which are long recognized as the major drivers of the hyperpigmentation disorder, serious consideration should be given to the use of a tinted broad-spectrum sunscreen or makeup containing more than 3% iron oxide, which blocks visible light. In contrast, zinc oxide does not, Dr. Pandya noted.
In one influential study, aminolevulinic acid was applied on the arms of 20 patients; two sunscreens were applied on areas where the ALA was applied, and on one area, no sunscreen was applied. The minimal phototoxic dose of visible blue light was doubled with application of a broad-spectrum sunscreen containing titanium dioxide, zinc oxide, and 0.2% iron oxide, compared with no sunscreen, but increased 21-fold using a sunscreen containing titanium dioxide, zinc oxide, and 3.2% iron oxide (Dermatol Surg. 2008 Nov;34[11]:1469-76).
Moreover, in a double-blind, randomized trial including 61 patients with melasma, all on background 4% hydroquinone, those assigned to a broad-spectrum sunscreen containing iron oxide had a 78% improvement in MASI scores at 8 weeks, compared with a 62% improvement with a broad-spectrum UV-only sunscreen. Both sunscreens had a sun protection factor of at least 50 (Photodermatol Photoimmunol Photomed. 2014 Feb;30[1]:35-42).
Numerous sunscreen and makeup products containing more than 3% iron oxide are available OTC in various tints. It’s a matter of finding a color that matches the patient’s skin.
Concern has been raised that exposure to the visible blue light emitted by computer screens and cell phones could worsen melasma. Dr. Pandya noted that reassurance on that score was recently provided by French investigators. They measured the intensity of visible light at the wavelengths emitted by computer screens and laptops and determined that it was 100- to 1,000-fold less than sunlight in the same spectrum. They also conducted a prospective, randomized, split-face trial in 12 melasma patients. One side of the face was exposed to the visible blue light at the same wavelengths emitted by device screens, but at far greater intensity. Blinded evaluators found no split-face difference in modified MASI scores.
“These results suggest that at a 20-cm distance, a maximized use of a high-intensity computer screen for 8 hours per day during a 5-day period does not worsen melasma lesions. Although it is very unlikely that similar exposure during a longer period would start to affect melasma lesions, such a possibility cannot be ruled out,” according to the investigators (J Am Acad Dermatol. 2019 Dec 27;S0190-9622(19)33324-9. doi: 10.1016/j.jaad.2019.12.047).
Dr. Pandya reported serving as a consultant to Incyte, Pfizer, Viela Bio, and Villaris.
FROM AAD 20
Large study finds no link between TCI use, skin cancer in patients with AD
The results also suggest dose, frequency, and exposure duration to the topical calcineurin inhibitors (TCIs) tacrolimus and pimecrolimus are not associated with an increased risk of keratinocyte carcinomas (KCs), basal cell carcinomas (BCCs), and squamous cell carcinomas (SCCs) in patients with atopic dermatitis (AD), according to Maryam M. Asgari, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues. In 2006, the Food and Drug Administration announced the addition of the boxed warning to the labeling of TCIs regarding a possible risk of cancer associated with use of pimecrolimus (Elidel) and with tacrolimus (Protopic), because of an increased risk of KCs associated with oral calcineurin inhibitors and reports of skin cancer in patients on TCIs.
“Controversy has surrounded the association between TCI exposure and KC risk since the black-box warning was issued by the FDA. A hypothesized mechanism of action for TCIs increasing KC risk includes a direct effect of calcineurin inhibition on DNA repair and apoptosis, which could influence keratinocyte carcinogenesis,” the authors of the study wrote in JAMA Dermatology. But, they added, there have been “conflicting results” in research exploring this association.
In the retrospective cohort study, Dr. Asgari and coauthors evaluated 93,746 adult patients with AD at Kaiser Permanente Northern California, diagnosed between January 2002 and December 2013, comparing skin cancer risk among 7,033 patients exposed to TCIs, 73,674 patients taking topical corticosteroids, and 46,141 patients who had not been exposed to TCIs or topical corticosteroids. Results were adjusted in a multivariate Cox regression analysis for age, gender, race/ethnicity, calendar year, number of dermatology visits per year, history of KCs, immunosuppression, prior systemic AD treatment, autoimmune disease, treatment with ultraviolet therapy, chemotherapy, and radiotherapy.
The researchers also examined how TCI dose, frequency and exposure duration impacted skin cancer risk. Patients were grouped by high-dose (0.1%) and low-dose (0.03%) formulations of tacrolimus; and the 1% formulation of pimecrolimus. Frequency of use was defined as low (once daily or less) or high (twice daily or more), and exposure duration was based on short- (less than 2 years), moderate- (2-4 years), and long-term (4 years or more) use. Patients were at least 40 years old (mean age, 58.5 years), 58.7% were women, 50.5% were White, 20.6% were Asian, 12.2% were Hispanic, and 7.9% were Black. They were followed for a mean of 7.70 years.
Compared with patients who were exposed to topical corticosteroids, there was no association between risk of KCs and exposure to TCIs in patients with AD (adjusted hazard ratio, 1.02; 95% confidence interval, 0.93-1.13). There were also no significant differences in risk of BCCs and TCI exposure (aHR, 1.01; 95% CI, 0.90-1.14) and risk of SCCs and TCI exposure (aHR, 0.94; 95% CI, 0.82-1.08), compared with patients exposed to topical corticosteroids.
Results were similar for risk of KCs (aHR, 1.03; 95% CI, 0.92-1.14), BCCs (aHR, 1.04; 95% CI, 0.91-1.19), and SCCs (aHR, 0.91; 95% CI, 0.78-1.06) when patients exposed to TCIs were compared with those with AD who were unexposed to any medication. In secondary analyses, Dr. Asgari and coauthors found no association with overall risk of KCs, or risk of BCCs or SCCs, and the dose, frequency, or exposure duration to TCIs.
“Our findings appear to support those of smaller postmarketing surveillance studies of TCI and KC risk and may provide some reassurance about the safety profile of this class of topical agents in the treatment of AD,” they concluded.
In an interview, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, said initial concerns surrounding TCIs were based on high doses potentially increasing the risk of malignancy, and off-label use of TCIs for inflammatory skin diseases other than AD.
“However, the FDA’s concerns may not have been justified,” he said. The manufacturers of pimecrolimus and tacrolimus have published results of 10-year observational registries that assess cancer risk, which “found no evidence of any associations between TCIs and malignancy,” noted Dr. Silverberg, who is also director of clinical research and contact dermatitis at George Washington University.
Elizabeth Hughes, MD, a dermatologist in private practice in San Antonio, said in an interview that initial enthusiasm was “huge” for use of TCIs like tacrolimus in patients with AD when they first became available, especially in the pediatric population, for whom clinicians are hesitant to use long-term strong topical steroids. However, parents of children taking the medication soon became concerned about potential side effects.
“The TCIs can be absorbed to a small extent through body surface area, so it was not a big leap to become concerned that infants and small children could absorb enough ... into the bloodstream to give a similar side effect profile as oral tacrolimus,” she said.
The addition of the boxed warning in 2006 was frustrating for dermatologists “because a medication we needed very much for a young population now was ‘labeled’ and parents were scared to use it,” Dr. Hughes explained.
Dr. Silverberg noted that, while the results of the new study are unlikely to change clinical practice, they are reassuring, and provide real-world data and “further confirmation of previous studies showing no associations between AD and malignancy.”
“Since AD and skin cancer are both commonly managed by dermatologists, there is potential for increased surveillance and detection of skin cancers in AD patients. So, the greatest chance of seeing a false-positive signal for malignancy would likely occur with skin cancers,” he pointed out. “Yet, even in the case of skin cancers, there were no demonstrable signals.”
Based on the results, “I think it is definitely reasonable to reconsider” the TCI boxed warning, but there isn’t much precedent for boxed warnings to be removed from labeling, Dr. Silverberg commented. “Unfortunately, the black-box warning may persist despite a lot of reassuring data.”
In a related editorial, Aaron M. Drucker, MD, ScM, and Mina Tadrous, PharmD, PhD, of the University of Toronto, said the boxed warning “had the intent of helping patients and clinicians understand possible risks,” but also carried the “potential for harm” if patients discontinued or did not adhere to treatment. “Safety warnings on topical medications could lead to undertreatment of atopic dermatitis, reduced quality of life and, potentially, increased use of more toxic systemic medications.”
Long-term studies of medications and cancer risk are challenging to perform, having to account for dose-response relationships, confounding by indication, and time bias, among other factors, and this study “recognizes and attempts to address many of these challenges,” Dr. Drucker and Dr. Tadrous wrote.
These results are similar to previous studies that have “consistently reported no or minimal association between TCI use and skin cancer,” they noted, adding that, “if an association exists, it is likely very small, meaning that skin cancer attributable to TCI use is rare. Clinicians can use this evidence to counsel and reassure patients for whom the benefits of ongoing treatment with TCIs may outweigh the harms.”
This study was funded by a grant from Valeant Pharmaceuticals. Dr. Asgari reported receiving grants from Valeant during the study, and from Pfizer not related to the study. The other authors reported no relevant conflicts of interest. Dr. Drucker reported relationships with the Canadian Agency for Drugs and Technology in Health, CME Outfitters, Eczema Society of Canada, Sanofi, Regeneron, and RTI Health Solutions in the form of paid fees, consultancies, honoraria, educational grants, and other compensation paid to him and/or his institution. Dr. Tadrous reported no relevant disclosures. Dr. Silverberg reported receiving honoraria for advisory board, speaker, and consultant services from numerous pharmaceutical manufacturers, and research grants for investigator services from GlaxoSmithKline and Galderma. Dr. Hughes Tichy reported no relevant financial disclosures. Dr. Silverberg is a member of the Dermatology News editorial advisory board.
SOURCE: Asgari MM et al. JAMA Dermatol. 2020 Aug 12. doi: 10.1001/jamadermatol.2020.2240.
The results also suggest dose, frequency, and exposure duration to the topical calcineurin inhibitors (TCIs) tacrolimus and pimecrolimus are not associated with an increased risk of keratinocyte carcinomas (KCs), basal cell carcinomas (BCCs), and squamous cell carcinomas (SCCs) in patients with atopic dermatitis (AD), according to Maryam M. Asgari, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues. In 2006, the Food and Drug Administration announced the addition of the boxed warning to the labeling of TCIs regarding a possible risk of cancer associated with use of pimecrolimus (Elidel) and with tacrolimus (Protopic), because of an increased risk of KCs associated with oral calcineurin inhibitors and reports of skin cancer in patients on TCIs.
“Controversy has surrounded the association between TCI exposure and KC risk since the black-box warning was issued by the FDA. A hypothesized mechanism of action for TCIs increasing KC risk includes a direct effect of calcineurin inhibition on DNA repair and apoptosis, which could influence keratinocyte carcinogenesis,” the authors of the study wrote in JAMA Dermatology. But, they added, there have been “conflicting results” in research exploring this association.
In the retrospective cohort study, Dr. Asgari and coauthors evaluated 93,746 adult patients with AD at Kaiser Permanente Northern California, diagnosed between January 2002 and December 2013, comparing skin cancer risk among 7,033 patients exposed to TCIs, 73,674 patients taking topical corticosteroids, and 46,141 patients who had not been exposed to TCIs or topical corticosteroids. Results were adjusted in a multivariate Cox regression analysis for age, gender, race/ethnicity, calendar year, number of dermatology visits per year, history of KCs, immunosuppression, prior systemic AD treatment, autoimmune disease, treatment with ultraviolet therapy, chemotherapy, and radiotherapy.
The researchers also examined how TCI dose, frequency and exposure duration impacted skin cancer risk. Patients were grouped by high-dose (0.1%) and low-dose (0.03%) formulations of tacrolimus; and the 1% formulation of pimecrolimus. Frequency of use was defined as low (once daily or less) or high (twice daily or more), and exposure duration was based on short- (less than 2 years), moderate- (2-4 years), and long-term (4 years or more) use. Patients were at least 40 years old (mean age, 58.5 years), 58.7% were women, 50.5% were White, 20.6% were Asian, 12.2% were Hispanic, and 7.9% were Black. They were followed for a mean of 7.70 years.
Compared with patients who were exposed to topical corticosteroids, there was no association between risk of KCs and exposure to TCIs in patients with AD (adjusted hazard ratio, 1.02; 95% confidence interval, 0.93-1.13). There were also no significant differences in risk of BCCs and TCI exposure (aHR, 1.01; 95% CI, 0.90-1.14) and risk of SCCs and TCI exposure (aHR, 0.94; 95% CI, 0.82-1.08), compared with patients exposed to topical corticosteroids.
Results were similar for risk of KCs (aHR, 1.03; 95% CI, 0.92-1.14), BCCs (aHR, 1.04; 95% CI, 0.91-1.19), and SCCs (aHR, 0.91; 95% CI, 0.78-1.06) when patients exposed to TCIs were compared with those with AD who were unexposed to any medication. In secondary analyses, Dr. Asgari and coauthors found no association with overall risk of KCs, or risk of BCCs or SCCs, and the dose, frequency, or exposure duration to TCIs.
“Our findings appear to support those of smaller postmarketing surveillance studies of TCI and KC risk and may provide some reassurance about the safety profile of this class of topical agents in the treatment of AD,” they concluded.
In an interview, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, said initial concerns surrounding TCIs were based on high doses potentially increasing the risk of malignancy, and off-label use of TCIs for inflammatory skin diseases other than AD.
“However, the FDA’s concerns may not have been justified,” he said. The manufacturers of pimecrolimus and tacrolimus have published results of 10-year observational registries that assess cancer risk, which “found no evidence of any associations between TCIs and malignancy,” noted Dr. Silverberg, who is also director of clinical research and contact dermatitis at George Washington University.
Elizabeth Hughes, MD, a dermatologist in private practice in San Antonio, said in an interview that initial enthusiasm was “huge” for use of TCIs like tacrolimus in patients with AD when they first became available, especially in the pediatric population, for whom clinicians are hesitant to use long-term strong topical steroids. However, parents of children taking the medication soon became concerned about potential side effects.
“The TCIs can be absorbed to a small extent through body surface area, so it was not a big leap to become concerned that infants and small children could absorb enough ... into the bloodstream to give a similar side effect profile as oral tacrolimus,” she said.
The addition of the boxed warning in 2006 was frustrating for dermatologists “because a medication we needed very much for a young population now was ‘labeled’ and parents were scared to use it,” Dr. Hughes explained.
Dr. Silverberg noted that, while the results of the new study are unlikely to change clinical practice, they are reassuring, and provide real-world data and “further confirmation of previous studies showing no associations between AD and malignancy.”
“Since AD and skin cancer are both commonly managed by dermatologists, there is potential for increased surveillance and detection of skin cancers in AD patients. So, the greatest chance of seeing a false-positive signal for malignancy would likely occur with skin cancers,” he pointed out. “Yet, even in the case of skin cancers, there were no demonstrable signals.”
Based on the results, “I think it is definitely reasonable to reconsider” the TCI boxed warning, but there isn’t much precedent for boxed warnings to be removed from labeling, Dr. Silverberg commented. “Unfortunately, the black-box warning may persist despite a lot of reassuring data.”
In a related editorial, Aaron M. Drucker, MD, ScM, and Mina Tadrous, PharmD, PhD, of the University of Toronto, said the boxed warning “had the intent of helping patients and clinicians understand possible risks,” but also carried the “potential for harm” if patients discontinued or did not adhere to treatment. “Safety warnings on topical medications could lead to undertreatment of atopic dermatitis, reduced quality of life and, potentially, increased use of more toxic systemic medications.”
Long-term studies of medications and cancer risk are challenging to perform, having to account for dose-response relationships, confounding by indication, and time bias, among other factors, and this study “recognizes and attempts to address many of these challenges,” Dr. Drucker and Dr. Tadrous wrote.
These results are similar to previous studies that have “consistently reported no or minimal association between TCI use and skin cancer,” they noted, adding that, “if an association exists, it is likely very small, meaning that skin cancer attributable to TCI use is rare. Clinicians can use this evidence to counsel and reassure patients for whom the benefits of ongoing treatment with TCIs may outweigh the harms.”
This study was funded by a grant from Valeant Pharmaceuticals. Dr. Asgari reported receiving grants from Valeant during the study, and from Pfizer not related to the study. The other authors reported no relevant conflicts of interest. Dr. Drucker reported relationships with the Canadian Agency for Drugs and Technology in Health, CME Outfitters, Eczema Society of Canada, Sanofi, Regeneron, and RTI Health Solutions in the form of paid fees, consultancies, honoraria, educational grants, and other compensation paid to him and/or his institution. Dr. Tadrous reported no relevant disclosures. Dr. Silverberg reported receiving honoraria for advisory board, speaker, and consultant services from numerous pharmaceutical manufacturers, and research grants for investigator services from GlaxoSmithKline and Galderma. Dr. Hughes Tichy reported no relevant financial disclosures. Dr. Silverberg is a member of the Dermatology News editorial advisory board.
SOURCE: Asgari MM et al. JAMA Dermatol. 2020 Aug 12. doi: 10.1001/jamadermatol.2020.2240.
The results also suggest dose, frequency, and exposure duration to the topical calcineurin inhibitors (TCIs) tacrolimus and pimecrolimus are not associated with an increased risk of keratinocyte carcinomas (KCs), basal cell carcinomas (BCCs), and squamous cell carcinomas (SCCs) in patients with atopic dermatitis (AD), according to Maryam M. Asgari, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues. In 2006, the Food and Drug Administration announced the addition of the boxed warning to the labeling of TCIs regarding a possible risk of cancer associated with use of pimecrolimus (Elidel) and with tacrolimus (Protopic), because of an increased risk of KCs associated with oral calcineurin inhibitors and reports of skin cancer in patients on TCIs.
“Controversy has surrounded the association between TCI exposure and KC risk since the black-box warning was issued by the FDA. A hypothesized mechanism of action for TCIs increasing KC risk includes a direct effect of calcineurin inhibition on DNA repair and apoptosis, which could influence keratinocyte carcinogenesis,” the authors of the study wrote in JAMA Dermatology. But, they added, there have been “conflicting results” in research exploring this association.
In the retrospective cohort study, Dr. Asgari and coauthors evaluated 93,746 adult patients with AD at Kaiser Permanente Northern California, diagnosed between January 2002 and December 2013, comparing skin cancer risk among 7,033 patients exposed to TCIs, 73,674 patients taking topical corticosteroids, and 46,141 patients who had not been exposed to TCIs or topical corticosteroids. Results were adjusted in a multivariate Cox regression analysis for age, gender, race/ethnicity, calendar year, number of dermatology visits per year, history of KCs, immunosuppression, prior systemic AD treatment, autoimmune disease, treatment with ultraviolet therapy, chemotherapy, and radiotherapy.
The researchers also examined how TCI dose, frequency and exposure duration impacted skin cancer risk. Patients were grouped by high-dose (0.1%) and low-dose (0.03%) formulations of tacrolimus; and the 1% formulation of pimecrolimus. Frequency of use was defined as low (once daily or less) or high (twice daily or more), and exposure duration was based on short- (less than 2 years), moderate- (2-4 years), and long-term (4 years or more) use. Patients were at least 40 years old (mean age, 58.5 years), 58.7% were women, 50.5% were White, 20.6% were Asian, 12.2% were Hispanic, and 7.9% were Black. They were followed for a mean of 7.70 years.
Compared with patients who were exposed to topical corticosteroids, there was no association between risk of KCs and exposure to TCIs in patients with AD (adjusted hazard ratio, 1.02; 95% confidence interval, 0.93-1.13). There were also no significant differences in risk of BCCs and TCI exposure (aHR, 1.01; 95% CI, 0.90-1.14) and risk of SCCs and TCI exposure (aHR, 0.94; 95% CI, 0.82-1.08), compared with patients exposed to topical corticosteroids.
Results were similar for risk of KCs (aHR, 1.03; 95% CI, 0.92-1.14), BCCs (aHR, 1.04; 95% CI, 0.91-1.19), and SCCs (aHR, 0.91; 95% CI, 0.78-1.06) when patients exposed to TCIs were compared with those with AD who were unexposed to any medication. In secondary analyses, Dr. Asgari and coauthors found no association with overall risk of KCs, or risk of BCCs or SCCs, and the dose, frequency, or exposure duration to TCIs.
“Our findings appear to support those of smaller postmarketing surveillance studies of TCI and KC risk and may provide some reassurance about the safety profile of this class of topical agents in the treatment of AD,” they concluded.
In an interview, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, said initial concerns surrounding TCIs were based on high doses potentially increasing the risk of malignancy, and off-label use of TCIs for inflammatory skin diseases other than AD.
“However, the FDA’s concerns may not have been justified,” he said. The manufacturers of pimecrolimus and tacrolimus have published results of 10-year observational registries that assess cancer risk, which “found no evidence of any associations between TCIs and malignancy,” noted Dr. Silverberg, who is also director of clinical research and contact dermatitis at George Washington University.
Elizabeth Hughes, MD, a dermatologist in private practice in San Antonio, said in an interview that initial enthusiasm was “huge” for use of TCIs like tacrolimus in patients with AD when they first became available, especially in the pediatric population, for whom clinicians are hesitant to use long-term strong topical steroids. However, parents of children taking the medication soon became concerned about potential side effects.
“The TCIs can be absorbed to a small extent through body surface area, so it was not a big leap to become concerned that infants and small children could absorb enough ... into the bloodstream to give a similar side effect profile as oral tacrolimus,” she said.
The addition of the boxed warning in 2006 was frustrating for dermatologists “because a medication we needed very much for a young population now was ‘labeled’ and parents were scared to use it,” Dr. Hughes explained.
Dr. Silverberg noted that, while the results of the new study are unlikely to change clinical practice, they are reassuring, and provide real-world data and “further confirmation of previous studies showing no associations between AD and malignancy.”
“Since AD and skin cancer are both commonly managed by dermatologists, there is potential for increased surveillance and detection of skin cancers in AD patients. So, the greatest chance of seeing a false-positive signal for malignancy would likely occur with skin cancers,” he pointed out. “Yet, even in the case of skin cancers, there were no demonstrable signals.”
Based on the results, “I think it is definitely reasonable to reconsider” the TCI boxed warning, but there isn’t much precedent for boxed warnings to be removed from labeling, Dr. Silverberg commented. “Unfortunately, the black-box warning may persist despite a lot of reassuring data.”
In a related editorial, Aaron M. Drucker, MD, ScM, and Mina Tadrous, PharmD, PhD, of the University of Toronto, said the boxed warning “had the intent of helping patients and clinicians understand possible risks,” but also carried the “potential for harm” if patients discontinued or did not adhere to treatment. “Safety warnings on topical medications could lead to undertreatment of atopic dermatitis, reduced quality of life and, potentially, increased use of more toxic systemic medications.”
Long-term studies of medications and cancer risk are challenging to perform, having to account for dose-response relationships, confounding by indication, and time bias, among other factors, and this study “recognizes and attempts to address many of these challenges,” Dr. Drucker and Dr. Tadrous wrote.
These results are similar to previous studies that have “consistently reported no or minimal association between TCI use and skin cancer,” they noted, adding that, “if an association exists, it is likely very small, meaning that skin cancer attributable to TCI use is rare. Clinicians can use this evidence to counsel and reassure patients for whom the benefits of ongoing treatment with TCIs may outweigh the harms.”
This study was funded by a grant from Valeant Pharmaceuticals. Dr. Asgari reported receiving grants from Valeant during the study, and from Pfizer not related to the study. The other authors reported no relevant conflicts of interest. Dr. Drucker reported relationships with the Canadian Agency for Drugs and Technology in Health, CME Outfitters, Eczema Society of Canada, Sanofi, Regeneron, and RTI Health Solutions in the form of paid fees, consultancies, honoraria, educational grants, and other compensation paid to him and/or his institution. Dr. Tadrous reported no relevant disclosures. Dr. Silverberg reported receiving honoraria for advisory board, speaker, and consultant services from numerous pharmaceutical manufacturers, and research grants for investigator services from GlaxoSmithKline and Galderma. Dr. Hughes Tichy reported no relevant financial disclosures. Dr. Silverberg is a member of the Dermatology News editorial advisory board.
SOURCE: Asgari MM et al. JAMA Dermatol. 2020 Aug 12. doi: 10.1001/jamadermatol.2020.2240.
FROM JAMA DERMATOLOGY
TNF inhibitors linked to inflammatory CNS events
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
, new research suggests
The nested case-control study included more than 200 participants with diseases such as rheumatoid arthritis, psoriasis, and Crohn’s disease. Results showed that exposure to TNF inhibitors was significantly associated with increased risk for demyelinating CNS events, such as multiple sclerosis, and nondemyelinating events, such as meningitis and encephalitis.
Interestingly, disease-specific secondary analyses showed that the strongest association for inflammatory events was in patients with rheumatoid arthritis.
Lead author Amy Kunchok, MD, of Mayo Clinic, Rochester, Minn., noted that “these are highly effective therapies for patients” and that these CNS events are likely uncommon.
“Our study has observed an association, but this does not imply causality. Therefore, we are not cautioning against using these therapies in appropriate patients,” Dr. Kunchok said in an interview.
“Rather, we recommend that clinicians assessing patients with both inflammatory demyelinating and nondemyelinating CNS events consider a detailed evaluation of the medication history, particularly in patients with coexistent autoimmune diseases who may have a current or past history of biological therapies,” she said.
The findings were published in JAMA Neurology.
Poorly understood
TNF inhibitors “are common therapies for certain autoimmune diseases,” the investigators noted.
Previously, a link between exposure to these inhibitors and inflammatory CNS events “has been postulated but is poorly understood,” they wrote.
In the current study, they examined records for 106 patients who were treated at Mayo clinics in Minnesota, Arizona, or Florida from January 2003 through February 2019. All participants had been diagnosed with an autoimmune disease that the Food and Drug Administration has listed as an indication for TNF inhibitor use. This included rheumatoid arthritis (n = 48), ankylosing spondylitis (n = 4), psoriasis and psoriatic arthritis (n = 21), Crohn’s disease (n = 27), and ulcerative colitis (n = 6). Their records also showed diagnostic codes for the inflammatory demyelinating CNS events of relapsing-remitting or primary progressive MS, clinically isolated syndrome, radiologically isolated syndrome, neuromyelitis optica spectrum disorder, and transverse myelitis or for the inflammatory nondemyelinating CNS events of meningitis, meningoencephalitis, encephalitis, neurosarcoidosis, and CNS vasculitis. The investigators also included 106 age-, sex-, and autoimmune disease–matched participants 1:1 to act as the control group.
In the total study population, 64% were women and the median age at disease onset was 52 years. In addition, 60% of the patient group and 40% of the control group were exposed to TNF inhibitors.
Novel finding?
Results showed that TNF inhibitor exposure was significantly linked to increased risk for developing any inflammatory CNS event (adjusted odds ratio, 3.01; 95% CI, 1.55-5.82; P = .001). When the outcomes were stratified by class of inflammatory event, these results were similar. The aOR was 3.09 (95% CI, 1.19-8.04; P = .02) for inflammatory demyelinating CNS events and was 2.97 (95% CI, 1.15-7.65; P = .02) for inflammatory nondemyelinating events.
Dr. Kunchok noted that the association between the inhibitors and nondemyelinating events was “a novel finding from this study.”
In secondary analyses, patients with rheumatoid arthritis and exposure to TNF inhibitors had the strongest association with any inflammatory CNS event (aOR, 4.82; 95% CI, 1.62-14.36; P = .005).
A pooled cohort comprising only the participants with the other autoimmune diseases did not show a significant association between exposure to TNF inhibitors and development of CNS events (P = .09).
“Because of the lack of power, further stratification by individual autoimmune diseases was not analyzed,” the investigators reported.
Although the overall findings showed that exposure to TNF inhibitors was linked to increased risk for inflammatory events, whether this association “represents de novo or exacerbated inflammatory pathways requires further research,” the authors wrote.
Dr. Kunchok added that more research, especially population-based studies, is also needed to examine the incidence of these inflammatory CNS events in patients exposed to TNF-alpha inhibitors.
Adds to the literature
In an accompanying editorial, Jeffrey M. Gelfand, MD, department of neurology at the University of California, San Francisco, and Jinoos Yazdany, MD, Zuckerberg San Francisco General Hospital at UCSF, noted that although the study adds to the literature, the magnitude of the risk found “remains unclear.”
“Randomized clinical trials are not suited to the study of rare adverse events,” Dr. Gelfand and Dr. Yazdany wrote. They agree with Dr. Kunchok that “next steps should include population-based observational studies that control for disease severity.”
Still, the current study provides additional evidence of rare adverse events in patients receiving TNF inhibitors, they noted. So how should prescribers proceed?
“As with all treatments, the risk-benefit ratio for the individual patient’s situation must be weighed and appropriate counseling must be given to facilitate shared decision-making discussions,” wrote the editorialists.
“Given what is known about the risk of harm, avoiding TNF inhibitors is advisable in patients with known MS,” they wrote.
In addition, neurologic consultation can be helpful for clarifying diagnoses and providing advice on monitoring strategies for TNF inhibitor treatment in those with possible MS or other demyelinating conditions, noted the editorialists.
“In patients who develop new concerning neurological symptoms while receiving TNF inhibitor treatment, timely evaluation is indicated, including consideration of neuroinflammatory, infectious, and neurological diagnoses that may be unrelated to treatment,” they added.
“Broader awareness of risks that studies such as this one by Kunchok et al provide can ... encourage timelier recognition of potential TNF inhibitor–associated neuroinflammatory events and may improve outcomes for patients,” Dr. Gelfand and Dr. Yazdany concluded.
The study was funded by a grant from the National Center for Advancing Translational Sciences. Dr. Kunchok reports having received research funding from Biogen outside this study. A full list of disclosures for the other study authors is in the original article. Dr. Gelfand reports having received g rants for a clinical trial from Genentech and consulting fees from Biogen, Alexion, Theranica, Impel Neuropharma, Advanced Clinical, Biohaven, and Satsuma. Dr. Yazdany reports having received grants from Pfizer and consulting fees from AstraZeneca and Eli Lilly outside the submitted work.
A version of this article originally appeared on Medscape.com.
FDA updates hydrochlorothiazide label to include nonmelanoma skin cancer risk
and undergo regular skin cancer screening, according to updates to the medication’s label.
The skin cancer risk is small, however, and patients should continue taking HCTZ, a commonly used diuretic and antihypertensive drug, unless their doctor says otherwise, according to a U.S. Food and Drug Administration announcement about the labeling changes, which the agency approved on Aug. 20.
HCTZ, first approved in 1959, is associated with photosensitivity. Researchers identified a relationship between HCTZ and nonmelanoma skin cancer in postmarketing studies. Investigators have described dose-response patterns for basal cell carcinoma and squamous cell carcinoma (SCC).
An FDA analysis found that the risk mostly was increased for SCC. The drug was associated with approximately one additional case of SCC per 16,000 patients per year. For white patients who received a cumulative dose of 50,000 mg or more, the risk was greater. In this patient population, HCTZ was associated with about one additional case of SCC per 6,700 patients per year, according to the label.
Reliably estimating the frequency of nonmelanoma skin cancer and establishing a causal relationship to drug exposure is not possible with the available postmarketing data, the label notes
“Treatment for nonmelanoma skin cancer is typically local and successful, with very low rates of death,” the FDA said. “Meanwhile, the risks of uncontrolled blood pressure can be severe and include life-threatening heart attacks or stroke. Given this information, patients should continue to use HCTZ and take protective skin care measures to reduce their risk of nonmelanoma skin cancer, unless directed otherwise from their health care provider.”
Patients can reduce sun exposure by using broad-spectrum sunscreens with a sun protection factor value of at least 15, limiting time in the sun, and wearing protective clothing, the agency advised.
and undergo regular skin cancer screening, according to updates to the medication’s label.
The skin cancer risk is small, however, and patients should continue taking HCTZ, a commonly used diuretic and antihypertensive drug, unless their doctor says otherwise, according to a U.S. Food and Drug Administration announcement about the labeling changes, which the agency approved on Aug. 20.
HCTZ, first approved in 1959, is associated with photosensitivity. Researchers identified a relationship between HCTZ and nonmelanoma skin cancer in postmarketing studies. Investigators have described dose-response patterns for basal cell carcinoma and squamous cell carcinoma (SCC).
An FDA analysis found that the risk mostly was increased for SCC. The drug was associated with approximately one additional case of SCC per 16,000 patients per year. For white patients who received a cumulative dose of 50,000 mg or more, the risk was greater. In this patient population, HCTZ was associated with about one additional case of SCC per 6,700 patients per year, according to the label.
Reliably estimating the frequency of nonmelanoma skin cancer and establishing a causal relationship to drug exposure is not possible with the available postmarketing data, the label notes
“Treatment for nonmelanoma skin cancer is typically local and successful, with very low rates of death,” the FDA said. “Meanwhile, the risks of uncontrolled blood pressure can be severe and include life-threatening heart attacks or stroke. Given this information, patients should continue to use HCTZ and take protective skin care measures to reduce their risk of nonmelanoma skin cancer, unless directed otherwise from their health care provider.”
Patients can reduce sun exposure by using broad-spectrum sunscreens with a sun protection factor value of at least 15, limiting time in the sun, and wearing protective clothing, the agency advised.
and undergo regular skin cancer screening, according to updates to the medication’s label.
The skin cancer risk is small, however, and patients should continue taking HCTZ, a commonly used diuretic and antihypertensive drug, unless their doctor says otherwise, according to a U.S. Food and Drug Administration announcement about the labeling changes, which the agency approved on Aug. 20.
HCTZ, first approved in 1959, is associated with photosensitivity. Researchers identified a relationship between HCTZ and nonmelanoma skin cancer in postmarketing studies. Investigators have described dose-response patterns for basal cell carcinoma and squamous cell carcinoma (SCC).
An FDA analysis found that the risk mostly was increased for SCC. The drug was associated with approximately one additional case of SCC per 16,000 patients per year. For white patients who received a cumulative dose of 50,000 mg or more, the risk was greater. In this patient population, HCTZ was associated with about one additional case of SCC per 6,700 patients per year, according to the label.
Reliably estimating the frequency of nonmelanoma skin cancer and establishing a causal relationship to drug exposure is not possible with the available postmarketing data, the label notes
“Treatment for nonmelanoma skin cancer is typically local and successful, with very low rates of death,” the FDA said. “Meanwhile, the risks of uncontrolled blood pressure can be severe and include life-threatening heart attacks or stroke. Given this information, patients should continue to use HCTZ and take protective skin care measures to reduce their risk of nonmelanoma skin cancer, unless directed otherwise from their health care provider.”
Patients can reduce sun exposure by using broad-spectrum sunscreens with a sun protection factor value of at least 15, limiting time in the sun, and wearing protective clothing, the agency advised.
Ulcerated hand lesion
While the eroded scabbed area in a raised lesion with pearly borders raised the suspicion of basal cell carcinoma (BCC), a superficial shave biopsy revealed that this was sclerosing basosquamous carcinoma.
Basosquamous carcinoma is an uncommon form of BCC. It features nests/clusters of basaloid cells arising from the basal layer of the epidermis (a hallmark of BCC), as well as keratinization, which is seen in squamous cell carcinoma (SCC). Basosquamous carcinoma occurs in the same areas as more common types of nonmelanoma skin cancers (NMSCs)—that is, sun-exposed areas. Not surprisingly, then, the highest risk areas for basosquamous carcinoma are the face, hands, arms, scalp (in those without hair), and back. People with occupational or intentional sun exposure through tanning have even higher rates of NMSC than the average population; patients using tanning booths exposing their entire bodies warrant more extensive physical exams.
Basosquamous carcinoma is one of the subtypes of BCC that is associated with a higher risk of local invasion, recurrence, and metastasis. Treatment with Mohs micrographic surgery (MMS) reduces the risk of recurrence to 4% (from 12% to 51%). Even with MMS, basosquamous carcinoma can be particularly challenging. It may require additional excision stages, as well as a larger than anticipated excision size at the time of surgery.
In this case, the patient was referred for MMS due to the high-risk nature of this lesion.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Garcia C, Poletti E, Crowson AN. Basosquamous carcinoma. J Am Acad Dermatol. 2009;60:137-143.

While the eroded scabbed area in a raised lesion with pearly borders raised the suspicion of basal cell carcinoma (BCC), a superficial shave biopsy revealed that this was sclerosing basosquamous carcinoma.
Basosquamous carcinoma is an uncommon form of BCC. It features nests/clusters of basaloid cells arising from the basal layer of the epidermis (a hallmark of BCC), as well as keratinization, which is seen in squamous cell carcinoma (SCC). Basosquamous carcinoma occurs in the same areas as more common types of nonmelanoma skin cancers (NMSCs)—that is, sun-exposed areas. Not surprisingly, then, the highest risk areas for basosquamous carcinoma are the face, hands, arms, scalp (in those without hair), and back. People with occupational or intentional sun exposure through tanning have even higher rates of NMSC than the average population; patients using tanning booths exposing their entire bodies warrant more extensive physical exams.
Basosquamous carcinoma is one of the subtypes of BCC that is associated with a higher risk of local invasion, recurrence, and metastasis. Treatment with Mohs micrographic surgery (MMS) reduces the risk of recurrence to 4% (from 12% to 51%). Even with MMS, basosquamous carcinoma can be particularly challenging. It may require additional excision stages, as well as a larger than anticipated excision size at the time of surgery.
In this case, the patient was referred for MMS due to the high-risk nature of this lesion.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

While the eroded scabbed area in a raised lesion with pearly borders raised the suspicion of basal cell carcinoma (BCC), a superficial shave biopsy revealed that this was sclerosing basosquamous carcinoma.
Basosquamous carcinoma is an uncommon form of BCC. It features nests/clusters of basaloid cells arising from the basal layer of the epidermis (a hallmark of BCC), as well as keratinization, which is seen in squamous cell carcinoma (SCC). Basosquamous carcinoma occurs in the same areas as more common types of nonmelanoma skin cancers (NMSCs)—that is, sun-exposed areas. Not surprisingly, then, the highest risk areas for basosquamous carcinoma are the face, hands, arms, scalp (in those without hair), and back. People with occupational or intentional sun exposure through tanning have even higher rates of NMSC than the average population; patients using tanning booths exposing their entire bodies warrant more extensive physical exams.
Basosquamous carcinoma is one of the subtypes of BCC that is associated with a higher risk of local invasion, recurrence, and metastasis. Treatment with Mohs micrographic surgery (MMS) reduces the risk of recurrence to 4% (from 12% to 51%). Even with MMS, basosquamous carcinoma can be particularly challenging. It may require additional excision stages, as well as a larger than anticipated excision size at the time of surgery.
In this case, the patient was referred for MMS due to the high-risk nature of this lesion.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Garcia C, Poletti E, Crowson AN. Basosquamous carcinoma. J Am Acad Dermatol. 2009;60:137-143.
Garcia C, Poletti E, Crowson AN. Basosquamous carcinoma. J Am Acad Dermatol. 2009;60:137-143.
Compression therapy cuts cellulitis risk in chronic leg edema
The effect was so striking that the randomized controlled trial was stopped early and all patients in the study were given the therapy.
“In a climate of increasing antibiotic resistance, we are delighted to have discovered a nondrug management strategy that has such a dramatic impact on the risk of cellulitis,” senior author Bernie Bissett, PhD, from the Discipline of Physiotherapy, Faculty of Health, the University of Canberra, Australia, said in an interview.
“We hope this leads to a shift in preventative medical strategy for patients with chronic edema and cellulitis around the world,” she said.
Lead author Elizabeth Webb, MPH, from the Physiotherapy Department at Calvary Public Hospital Bruce, in Bruce, Australia, and colleagues report their findings in an article published online August 12 in The New England Journal of Medicine.
Dr. Bisset explained that Webb is a “leading lymphedema physiotherapist” and a PhD candidate at the University of Canberra. She added that this is the first study to show that “compression therapy dramatically reduces the risk of cellulitis for patients with chronic edema.”
Penicillin is often given preventively; some research suggests effectiveness wanes after the antibiotic is stopped.
For the current trial, Ms. Webb and colleagues enrolled 84 adults with chronic edema of the leg and recurrent cellulitis. They randomly assigned patients in a 1:1 ratio to receive leg compression therapy plus education about preventing cellulitis (compression group; n = 41) or education only (control group; n = 43).
Compression therapy consisted of wearing knee-high stockings that applied maximum compression at the ankles. The compression gradually decreased up the legs. In addition, 26 patients were treated with “therapist-applied compression bandaging” for 3 to 5 days before receiving the stockings.
Participants underwent follow-up assessments every 6 months for a maximum of 3 years or until 45 episodes of cellulitis, the primary outcome, occurred. Those in the control group crossed over to the compression group once they experienced cellulitis.
The trial was stopped early for reasons of efficacy. “The statistical analysis plan prespecified that after 23 episodes of cellulitis had occurred, an independent data monitoring committee would review the results of the interim analysis and recommend whether the trial should stop early,” the authors write.
At the time of the monitoring committee’s review, six patients (15%) who wore compression stockings and 17 (40%) in the control group had experienced a cellulitis episode (hazard ratio, 0.23; P = .002; relative risk [post hoc analysis], 0.37; P = .02). On the basis of those findings, the researchers stopped the study, and patients in the control group were started on compression therapy.
“Clinicians should definitely consider referring their patients to a skilled lymphedema therapist who can individually prescribe and fit compression garments,” Dr. Bissett said. “In our study, these were well tolerated and reduced the risk of another episode of cellulitis by a huge 77%,” she added.
Secondary outcomes included hospitalization related to cellulitis and quality-of-life assessments.
Three patients (7%) in the compression group and six (14%) in the control group were admitted to the hospital for cellulitis (hazard ratio, 0.38). There were no differences in quality of life outcomes between the treatment groups.
The authors say compression therapy has the potential to decrease cellulitis risk by reducing edema, boosting immune response and skin integrity, and protecting the skin.
“Patients with a history of leg swelling (chronic edema) and previous episodes of cellulitis are ideal candidates for this compression therapy,” Dr. Bissett said.
“Given the lack of side effects of the therapy in our study and the potential to reduce other skin problems in these patients, compression therapy is an ideal prophylactic strategy,” she said.
The authors note several study limitations, including a lack of blinding. In addition, patients in the study had to have access to lymphedema specialists, who might be unavailable to patients outside the study. This could have influenced adherence and limit generalizability. Difficulty putting on and taking off compression garments often leads patients to be less adherent to compression therapy, but 88% of patients in this study wore them at least 4 days per week.
Dr. Bissett said compression therapy would be useful for primary care physicians to consider for patients with chronic edema.
“Primary care physicians are highly likely to encounter patients with chronic edema in their day-to-day practice. We can now confidently say that referral to lymphedema therapists for compression therapy should be a first line of defense against future episodes of cellulitis in this vulnerable patient group,” she explained.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The effect was so striking that the randomized controlled trial was stopped early and all patients in the study were given the therapy.
“In a climate of increasing antibiotic resistance, we are delighted to have discovered a nondrug management strategy that has such a dramatic impact on the risk of cellulitis,” senior author Bernie Bissett, PhD, from the Discipline of Physiotherapy, Faculty of Health, the University of Canberra, Australia, said in an interview.
“We hope this leads to a shift in preventative medical strategy for patients with chronic edema and cellulitis around the world,” she said.
Lead author Elizabeth Webb, MPH, from the Physiotherapy Department at Calvary Public Hospital Bruce, in Bruce, Australia, and colleagues report their findings in an article published online August 12 in The New England Journal of Medicine.
Dr. Bisset explained that Webb is a “leading lymphedema physiotherapist” and a PhD candidate at the University of Canberra. She added that this is the first study to show that “compression therapy dramatically reduces the risk of cellulitis for patients with chronic edema.”
Penicillin is often given preventively; some research suggests effectiveness wanes after the antibiotic is stopped.
For the current trial, Ms. Webb and colleagues enrolled 84 adults with chronic edema of the leg and recurrent cellulitis. They randomly assigned patients in a 1:1 ratio to receive leg compression therapy plus education about preventing cellulitis (compression group; n = 41) or education only (control group; n = 43).
Compression therapy consisted of wearing knee-high stockings that applied maximum compression at the ankles. The compression gradually decreased up the legs. In addition, 26 patients were treated with “therapist-applied compression bandaging” for 3 to 5 days before receiving the stockings.
Participants underwent follow-up assessments every 6 months for a maximum of 3 years or until 45 episodes of cellulitis, the primary outcome, occurred. Those in the control group crossed over to the compression group once they experienced cellulitis.
The trial was stopped early for reasons of efficacy. “The statistical analysis plan prespecified that after 23 episodes of cellulitis had occurred, an independent data monitoring committee would review the results of the interim analysis and recommend whether the trial should stop early,” the authors write.
At the time of the monitoring committee’s review, six patients (15%) who wore compression stockings and 17 (40%) in the control group had experienced a cellulitis episode (hazard ratio, 0.23; P = .002; relative risk [post hoc analysis], 0.37; P = .02). On the basis of those findings, the researchers stopped the study, and patients in the control group were started on compression therapy.
“Clinicians should definitely consider referring their patients to a skilled lymphedema therapist who can individually prescribe and fit compression garments,” Dr. Bissett said. “In our study, these were well tolerated and reduced the risk of another episode of cellulitis by a huge 77%,” she added.
Secondary outcomes included hospitalization related to cellulitis and quality-of-life assessments.
Three patients (7%) in the compression group and six (14%) in the control group were admitted to the hospital for cellulitis (hazard ratio, 0.38). There were no differences in quality of life outcomes between the treatment groups.
The authors say compression therapy has the potential to decrease cellulitis risk by reducing edema, boosting immune response and skin integrity, and protecting the skin.
“Patients with a history of leg swelling (chronic edema) and previous episodes of cellulitis are ideal candidates for this compression therapy,” Dr. Bissett said.
“Given the lack of side effects of the therapy in our study and the potential to reduce other skin problems in these patients, compression therapy is an ideal prophylactic strategy,” she said.
The authors note several study limitations, including a lack of blinding. In addition, patients in the study had to have access to lymphedema specialists, who might be unavailable to patients outside the study. This could have influenced adherence and limit generalizability. Difficulty putting on and taking off compression garments often leads patients to be less adherent to compression therapy, but 88% of patients in this study wore them at least 4 days per week.
Dr. Bissett said compression therapy would be useful for primary care physicians to consider for patients with chronic edema.
“Primary care physicians are highly likely to encounter patients with chronic edema in their day-to-day practice. We can now confidently say that referral to lymphedema therapists for compression therapy should be a first line of defense against future episodes of cellulitis in this vulnerable patient group,” she explained.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The effect was so striking that the randomized controlled trial was stopped early and all patients in the study were given the therapy.
“In a climate of increasing antibiotic resistance, we are delighted to have discovered a nondrug management strategy that has such a dramatic impact on the risk of cellulitis,” senior author Bernie Bissett, PhD, from the Discipline of Physiotherapy, Faculty of Health, the University of Canberra, Australia, said in an interview.
“We hope this leads to a shift in preventative medical strategy for patients with chronic edema and cellulitis around the world,” she said.
Lead author Elizabeth Webb, MPH, from the Physiotherapy Department at Calvary Public Hospital Bruce, in Bruce, Australia, and colleagues report their findings in an article published online August 12 in The New England Journal of Medicine.
Dr. Bisset explained that Webb is a “leading lymphedema physiotherapist” and a PhD candidate at the University of Canberra. She added that this is the first study to show that “compression therapy dramatically reduces the risk of cellulitis for patients with chronic edema.”
Penicillin is often given preventively; some research suggests effectiveness wanes after the antibiotic is stopped.
For the current trial, Ms. Webb and colleagues enrolled 84 adults with chronic edema of the leg and recurrent cellulitis. They randomly assigned patients in a 1:1 ratio to receive leg compression therapy plus education about preventing cellulitis (compression group; n = 41) or education only (control group; n = 43).
Compression therapy consisted of wearing knee-high stockings that applied maximum compression at the ankles. The compression gradually decreased up the legs. In addition, 26 patients were treated with “therapist-applied compression bandaging” for 3 to 5 days before receiving the stockings.
Participants underwent follow-up assessments every 6 months for a maximum of 3 years or until 45 episodes of cellulitis, the primary outcome, occurred. Those in the control group crossed over to the compression group once they experienced cellulitis.
The trial was stopped early for reasons of efficacy. “The statistical analysis plan prespecified that after 23 episodes of cellulitis had occurred, an independent data monitoring committee would review the results of the interim analysis and recommend whether the trial should stop early,” the authors write.
At the time of the monitoring committee’s review, six patients (15%) who wore compression stockings and 17 (40%) in the control group had experienced a cellulitis episode (hazard ratio, 0.23; P = .002; relative risk [post hoc analysis], 0.37; P = .02). On the basis of those findings, the researchers stopped the study, and patients in the control group were started on compression therapy.
“Clinicians should definitely consider referring their patients to a skilled lymphedema therapist who can individually prescribe and fit compression garments,” Dr. Bissett said. “In our study, these were well tolerated and reduced the risk of another episode of cellulitis by a huge 77%,” she added.
Secondary outcomes included hospitalization related to cellulitis and quality-of-life assessments.
Three patients (7%) in the compression group and six (14%) in the control group were admitted to the hospital for cellulitis (hazard ratio, 0.38). There were no differences in quality of life outcomes between the treatment groups.
The authors say compression therapy has the potential to decrease cellulitis risk by reducing edema, boosting immune response and skin integrity, and protecting the skin.
“Patients with a history of leg swelling (chronic edema) and previous episodes of cellulitis are ideal candidates for this compression therapy,” Dr. Bissett said.
“Given the lack of side effects of the therapy in our study and the potential to reduce other skin problems in these patients, compression therapy is an ideal prophylactic strategy,” she said.
The authors note several study limitations, including a lack of blinding. In addition, patients in the study had to have access to lymphedema specialists, who might be unavailable to patients outside the study. This could have influenced adherence and limit generalizability. Difficulty putting on and taking off compression garments often leads patients to be less adherent to compression therapy, but 88% of patients in this study wore them at least 4 days per week.
Dr. Bissett said compression therapy would be useful for primary care physicians to consider for patients with chronic edema.
“Primary care physicians are highly likely to encounter patients with chronic edema in their day-to-day practice. We can now confidently say that referral to lymphedema therapists for compression therapy should be a first line of defense against future episodes of cellulitis in this vulnerable patient group,” she explained.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A 4-year-old with a lesion on her cheek, which grew and became firmer over two months
The patient was diagnosed with idiopathic facial aseptic granuloma (IFAG) based on the clinical findings, as well as the associated history of chalazia and erythematous papules seen in childhood rosacea.
She was treated with several months of azithromycin, sulfur wash, and metronidazole cream with improvement of some of the smaller lesions but no change on the larger nodules. Later she was treated with oral and topical ivermectin with no improvement. Some of the nodules slowly resolved except for the larger lesion on the right cheek. She was later treated with a 6-week course of clarithromycin with partial improvement of the nodule. The lesion resolved after 2 months of stopping clarithromycin.
IFAG is a rare condition seen in prepubescent children. The etiology of this condition is not well understood and is thought to be on the spectrum of childhood rosacea.1 From several recent reports, IFAG usually is seen in children with associated conditions including chalazia, conjunctivitis, blepharitis, and telangiectasias, which can be seen in patients with rosacea. These associated findings suggest the possibility of IFAG being a form of granulomatous rosacea in children.
This condition presents in childhood between the ages of 8 months and 13 years. Most of the cases occur in toddlers, and girls appear to be more affected than boys. The lesions appear as pink, rubbery, nontender, nonfluctuant nodules on the cheeks, which can be single or multiple. A large prospective study in 30 children demonstrated that more 70% of the lesions cultured were negative for bacteria. Histologic analysis of some of the lesions showed a chronic dermal lymphohistiocytic granulomatous perifollicular infiltrate with numerous foreign body–type giant cells.2
The differential diagnosis of these lesions should include infectious pyodermas such as mycobacterial infections, cutaneous leishmaniasis, and botryomycosis; deep fungal infections such as sporotrichosis, coccidioidomycosis, and cryptococcosis; childhood nodulocystic acne; pilomatrixoma; epidermoid cyst; vascular tumors or malformations; and leukemia cutis.3
The diagnosis is usually clinical but in atypical cases a skin biopsy with tissue cultures should be performed. The decision to biopsy these lesions will need to be done in a one by one basis, as a biopsy may leave scaring on the area affected.
It has been postulated that a color Doppler ultrasound of the lesion may be a helpful ancillary study. Echographic findings show a well demarcated solid-cystic, hypoechoic dermal lesion, the largest axis of which lies parallel to the skin surface. The lesion lacks calcium deposits. Other findings include increased echogenicity of the underlaying hypodermis. The findings may vary depending on the stage of the lesion.4
The course of the condition may last on average months to years. Some lesions resolve spontaneously and others may respond to courses of oral antibiotics such as clarithromycin, azithromycin, or ivermectin. In our patient, several lesions improved with oral antibiotics, but the larger lesions were more persistent and resolved after a year.
The lesions usually resolve without scarring. In those patients with associated rosacea, maintenance topical treatments may be warranted and also may need follow-up with ophthalmology because they tend to commonly have ocular rosacea as well.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at pdnews@mdedge.com.
References
1. Pediatr Dermatol. 2013 Jan-Feb;30(1):109-11.
2. Br J Dermatol. 2007 Apr;156(4):705-8.
3. Pediatr Dermatol. 2018 Jul;35(4):490-3.
4. Actas Dermosifiliogr. 2019 Oct;110(8):637-41.
The patient was diagnosed with idiopathic facial aseptic granuloma (IFAG) based on the clinical findings, as well as the associated history of chalazia and erythematous papules seen in childhood rosacea.
She was treated with several months of azithromycin, sulfur wash, and metronidazole cream with improvement of some of the smaller lesions but no change on the larger nodules. Later she was treated with oral and topical ivermectin with no improvement. Some of the nodules slowly resolved except for the larger lesion on the right cheek. She was later treated with a 6-week course of clarithromycin with partial improvement of the nodule. The lesion resolved after 2 months of stopping clarithromycin.
IFAG is a rare condition seen in prepubescent children. The etiology of this condition is not well understood and is thought to be on the spectrum of childhood rosacea.1 From several recent reports, IFAG usually is seen in children with associated conditions including chalazia, conjunctivitis, blepharitis, and telangiectasias, which can be seen in patients with rosacea. These associated findings suggest the possibility of IFAG being a form of granulomatous rosacea in children.
This condition presents in childhood between the ages of 8 months and 13 years. Most of the cases occur in toddlers, and girls appear to be more affected than boys. The lesions appear as pink, rubbery, nontender, nonfluctuant nodules on the cheeks, which can be single or multiple. A large prospective study in 30 children demonstrated that more 70% of the lesions cultured were negative for bacteria. Histologic analysis of some of the lesions showed a chronic dermal lymphohistiocytic granulomatous perifollicular infiltrate with numerous foreign body–type giant cells.2
The differential diagnosis of these lesions should include infectious pyodermas such as mycobacterial infections, cutaneous leishmaniasis, and botryomycosis; deep fungal infections such as sporotrichosis, coccidioidomycosis, and cryptococcosis; childhood nodulocystic acne; pilomatrixoma; epidermoid cyst; vascular tumors or malformations; and leukemia cutis.3
The diagnosis is usually clinical but in atypical cases a skin biopsy with tissue cultures should be performed. The decision to biopsy these lesions will need to be done in a one by one basis, as a biopsy may leave scaring on the area affected.
It has been postulated that a color Doppler ultrasound of the lesion may be a helpful ancillary study. Echographic findings show a well demarcated solid-cystic, hypoechoic dermal lesion, the largest axis of which lies parallel to the skin surface. The lesion lacks calcium deposits. Other findings include increased echogenicity of the underlaying hypodermis. The findings may vary depending on the stage of the lesion.4
The course of the condition may last on average months to years. Some lesions resolve spontaneously and others may respond to courses of oral antibiotics such as clarithromycin, azithromycin, or ivermectin. In our patient, several lesions improved with oral antibiotics, but the larger lesions were more persistent and resolved after a year.
The lesions usually resolve without scarring. In those patients with associated rosacea, maintenance topical treatments may be warranted and also may need follow-up with ophthalmology because they tend to commonly have ocular rosacea as well.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at pdnews@mdedge.com.
References
1. Pediatr Dermatol. 2013 Jan-Feb;30(1):109-11.
2. Br J Dermatol. 2007 Apr;156(4):705-8.
3. Pediatr Dermatol. 2018 Jul;35(4):490-3.
4. Actas Dermosifiliogr. 2019 Oct;110(8):637-41.
The patient was diagnosed with idiopathic facial aseptic granuloma (IFAG) based on the clinical findings, as well as the associated history of chalazia and erythematous papules seen in childhood rosacea.
She was treated with several months of azithromycin, sulfur wash, and metronidazole cream with improvement of some of the smaller lesions but no change on the larger nodules. Later she was treated with oral and topical ivermectin with no improvement. Some of the nodules slowly resolved except for the larger lesion on the right cheek. She was later treated with a 6-week course of clarithromycin with partial improvement of the nodule. The lesion resolved after 2 months of stopping clarithromycin.
IFAG is a rare condition seen in prepubescent children. The etiology of this condition is not well understood and is thought to be on the spectrum of childhood rosacea.1 From several recent reports, IFAG usually is seen in children with associated conditions including chalazia, conjunctivitis, blepharitis, and telangiectasias, which can be seen in patients with rosacea. These associated findings suggest the possibility of IFAG being a form of granulomatous rosacea in children.
This condition presents in childhood between the ages of 8 months and 13 years. Most of the cases occur in toddlers, and girls appear to be more affected than boys. The lesions appear as pink, rubbery, nontender, nonfluctuant nodules on the cheeks, which can be single or multiple. A large prospective study in 30 children demonstrated that more 70% of the lesions cultured were negative for bacteria. Histologic analysis of some of the lesions showed a chronic dermal lymphohistiocytic granulomatous perifollicular infiltrate with numerous foreign body–type giant cells.2
The differential diagnosis of these lesions should include infectious pyodermas such as mycobacterial infections, cutaneous leishmaniasis, and botryomycosis; deep fungal infections such as sporotrichosis, coccidioidomycosis, and cryptococcosis; childhood nodulocystic acne; pilomatrixoma; epidermoid cyst; vascular tumors or malformations; and leukemia cutis.3
The diagnosis is usually clinical but in atypical cases a skin biopsy with tissue cultures should be performed. The decision to biopsy these lesions will need to be done in a one by one basis, as a biopsy may leave scaring on the area affected.
It has been postulated that a color Doppler ultrasound of the lesion may be a helpful ancillary study. Echographic findings show a well demarcated solid-cystic, hypoechoic dermal lesion, the largest axis of which lies parallel to the skin surface. The lesion lacks calcium deposits. Other findings include increased echogenicity of the underlaying hypodermis. The findings may vary depending on the stage of the lesion.4
The course of the condition may last on average months to years. Some lesions resolve spontaneously and others may respond to courses of oral antibiotics such as clarithromycin, azithromycin, or ivermectin. In our patient, several lesions improved with oral antibiotics, but the larger lesions were more persistent and resolved after a year.
The lesions usually resolve without scarring. In those patients with associated rosacea, maintenance topical treatments may be warranted and also may need follow-up with ophthalmology because they tend to commonly have ocular rosacea as well.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at pdnews@mdedge.com.
References
1. Pediatr Dermatol. 2013 Jan-Feb;30(1):109-11.
2. Br J Dermatol. 2007 Apr;156(4):705-8.
3. Pediatr Dermatol. 2018 Jul;35(4):490-3.
4. Actas Dermosifiliogr. 2019 Oct;110(8):637-41.
A 4-year-old female is brought to our pediatric dermatology clinic for evaluation of a persistent lesion on the cheek.
The mother of the child reports that the lesion started as a small "bug bite" and then started growing and getting firmer for the past 2 months. The girl has developed other smaller red, pimple-like lesions on the cheeks and one of them is starting to increase in size.
She denies any tenderness on the area or any purulent discharge. She has had no fevers, chills, weight loss, nose bleeds, fatigue, or any other symptoms. The mother has not noted any changes on the child's body odor, any rapid growth, or hair on her axillary or pubic area. She was treated with three different courses of oral antibiotics including cephalexin, trimethoprim/sulfamethoxazole, and clindamycin, as well as topical mupirocin, with no improvement.
Her past medical history is significant for several episodes of eyelid cysts that were treated with warm compresses and topical erythromycin ointment. The family history is significant for the father having severe acne as a teenager. She has two cats, she has not traveled, and she has an older sister who has no lesions.
On physical examination she is a lovely 4-year-old female in no acute distress. Her height is on the 70th percentile and weight on the 40th percentile for her age. Her blood pressure is 95/84 with a heart rate of 96. On skin examination she has several pink macules and papules on her bilateral cheeks. On the left cheek there are two pink nodules: One is 1 cm, and the other is 7 mm. The nodules are not tender. There is no warmth, fluctuance, or discharge from the lesions.
She has no cervical lymphadenopathy. She has no axillary or pubic hair. She is Tanner stage I.
A Roundabout Journey to Diagnosis
ANSWER
The correct answer is all the above (choice “e”).
DISCUSSION
The differential for round or annular, scaly lesions is lengthy. In addition to all 4 conditions listed above, it includes eczema, basal cell carcinoma, and irritant or contact dermatitis.
With this patient’s history, the diagnosis of fungal infection was not unreasonable. However, the total lack of response to antifungal treatment—along with a negative KOH prep—made that diagnosis questionable at best. Then there was the lack of lymphadenopathy, which would almost certainly have been present with such longstanding infection. Given her country of origin, cutaneous New World leishmaniasis (caused by a protozoan delivered to the patient by an insect vector) was a possibility.
For this patient, skin biopsy with a 4-mm punch was the only way to establish the correct diagnosis. The defect from the biopsy was closed with 5-0 nylon sutures to minimize scarring.
The pathological findings included interface dermatitis, apoptotic keratinocytes, and a brisk periadnexal lymphocytic infiltrate, which—given the morphological and historical context—were entirely consistent with discoid lupus erythematosus (DLE; otherwise known as subacute cutaneous lupus erythematosus). Subsequent bloodwork failed to show any connection with systemic lupus erythematosus (SLE), which is not surprising because only about 18% of DLE cases evolve into SLE.
DLE is more common in women, especially in those with skin of color. The associated lesions are seldom as impressive as this patient’s, manifesting as papulosquamous patches typically found on the ears, neck, face, arms, and other sun-exposed areas. Indeed, it appears that sun exposure is a major trigger for the disease—a clue that can assist with the diagnosis.
TREATMENT
In addition to proscribing excessive sun exposure, providers should encourage the use of sunscreen. DLE is often treated with topical steroids. More advanced cases, such as this patient’s, may require oral hydroxychloroquine (200 mg bid). Though these treatments are effective in most cases, DLE can leave serious scarring and/or discoloration, especially in those with darker skin.
ANSWER
The correct answer is all the above (choice “e”).
DISCUSSION
The differential for round or annular, scaly lesions is lengthy. In addition to all 4 conditions listed above, it includes eczema, basal cell carcinoma, and irritant or contact dermatitis.
With this patient’s history, the diagnosis of fungal infection was not unreasonable. However, the total lack of response to antifungal treatment—along with a negative KOH prep—made that diagnosis questionable at best. Then there was the lack of lymphadenopathy, which would almost certainly have been present with such longstanding infection. Given her country of origin, cutaneous New World leishmaniasis (caused by a protozoan delivered to the patient by an insect vector) was a possibility.
For this patient, skin biopsy with a 4-mm punch was the only way to establish the correct diagnosis. The defect from the biopsy was closed with 5-0 nylon sutures to minimize scarring.
The pathological findings included interface dermatitis, apoptotic keratinocytes, and a brisk periadnexal lymphocytic infiltrate, which—given the morphological and historical context—were entirely consistent with discoid lupus erythematosus (DLE; otherwise known as subacute cutaneous lupus erythematosus). Subsequent bloodwork failed to show any connection with systemic lupus erythematosus (SLE), which is not surprising because only about 18% of DLE cases evolve into SLE.
DLE is more common in women, especially in those with skin of color. The associated lesions are seldom as impressive as this patient’s, manifesting as papulosquamous patches typically found on the ears, neck, face, arms, and other sun-exposed areas. Indeed, it appears that sun exposure is a major trigger for the disease—a clue that can assist with the diagnosis.
TREATMENT
In addition to proscribing excessive sun exposure, providers should encourage the use of sunscreen. DLE is often treated with topical steroids. More advanced cases, such as this patient’s, may require oral hydroxychloroquine (200 mg bid). Though these treatments are effective in most cases, DLE can leave serious scarring and/or discoloration, especially in those with darker skin.
ANSWER
The correct answer is all the above (choice “e”).
DISCUSSION
The differential for round or annular, scaly lesions is lengthy. In addition to all 4 conditions listed above, it includes eczema, basal cell carcinoma, and irritant or contact dermatitis.
With this patient’s history, the diagnosis of fungal infection was not unreasonable. However, the total lack of response to antifungal treatment—along with a negative KOH prep—made that diagnosis questionable at best. Then there was the lack of lymphadenopathy, which would almost certainly have been present with such longstanding infection. Given her country of origin, cutaneous New World leishmaniasis (caused by a protozoan delivered to the patient by an insect vector) was a possibility.
For this patient, skin biopsy with a 4-mm punch was the only way to establish the correct diagnosis. The defect from the biopsy was closed with 5-0 nylon sutures to minimize scarring.
The pathological findings included interface dermatitis, apoptotic keratinocytes, and a brisk periadnexal lymphocytic infiltrate, which—given the morphological and historical context—were entirely consistent with discoid lupus erythematosus (DLE; otherwise known as subacute cutaneous lupus erythematosus). Subsequent bloodwork failed to show any connection with systemic lupus erythematosus (SLE), which is not surprising because only about 18% of DLE cases evolve into SLE.
DLE is more common in women, especially in those with skin of color. The associated lesions are seldom as impressive as this patient’s, manifesting as papulosquamous patches typically found on the ears, neck, face, arms, and other sun-exposed areas. Indeed, it appears that sun exposure is a major trigger for the disease—a clue that can assist with the diagnosis.
TREATMENT
In addition to proscribing excessive sun exposure, providers should encourage the use of sunscreen. DLE is often treated with topical steroids. More advanced cases, such as this patient’s, may require oral hydroxychloroquine (200 mg bid). Though these treatments are effective in most cases, DLE can leave serious scarring and/or discoloration, especially in those with darker skin.
After journeying for several months from Honduras, this 30-year-old woman visits the clinic for evaluation of a lesion that has been growing on her cheek since before she started traveling. She saw several providers—mostly in NGO clinics—along her journey. The diagnosis they gave was consistently “ringworm.” She was offered various topical creams, none of which produced any results.
Though the lesion is not painful, it causes some itching. The patient is much more concerned about its appearance. Through interpreters, she claims to be in otherwise good health. She has no other lesions, joint pain, fever, or malaise. She reports neither her family nor fellow travelers have such lesions.
Examination reveals an impressive 3-cm, round, papulosquamous plaque on the right side of her face (below the malar area). The lesion is neither tender nor notably warm. There are no palpable lymph nodes in the area. The scaling is mostly on the periphery. A KOH prep of the scaling shows no fungal elements.
Scaly hand papule
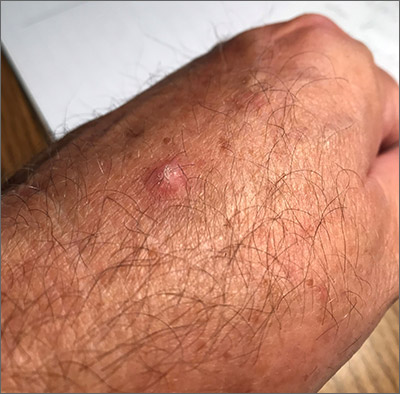
This pink raised nodule underlying a scaly surface was suspicious for squamous cell carcinoma (SCC). Since this was a virtual visit, and the lesion required pathology due to the likelihood of cancer, the patient was brought into the clinic for additional evaluation. A broad-based deep shave biopsy was performed to remove the visible lesion. Pathology showed SCC in situ, with borders uninvolved.
Patients who have had AKs are extremely likely to develop additional AKs. A notable percentage of AKs will, over time, develop into SCC in situ and then invasive SCC if not treated. While cryosurgery of an SK should not result in SCC, it’s most likely that in this case, an AK adjacent to the SK progressed to the SCC in situ.
There are multiple treatments available for SCC in situ. Topical imiquimod has been shown to be somewhat effective in stimulating the immune system, thus leading to resolution of SCC in situ. But there is a significant risk of recurrence. Topical 5-FU can be utilized on a daily or twice daily basis for 2 weeks (or up to several months). The risk of recurrence ranges from 7% to 33%. Electrodesiccation and curettage is often used for SCC in situ, with recurrence rates of 2% to 19%. Cryosurgery for SCC in situ requires an aggressive freeze, with freeze times of up to 30 seconds. Photodynamic therapy also is an option; however, it requires multiple sessions and is more costly than other treatment options.
This patient’s borders were uninvolved on pathology, but it was possible that there was some residual SCC in situ due to the standard “bread loaf slicing” used for routine pathology. To treat possible residual SCC in situ at the wound site and surrounding tissue, the patient was given a prescription for topical 5-FU to apply twice daily for 6 weeks. The patient was instructed to return for follow-up in 6 months, or sooner, if any problems arose.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Shimizu I, Cruz A, Chang KH, et al. Treatment of squamous cell carcinoma in situ: a review. Dermatol Surg. 2011;37:1394-1411.

This pink raised nodule underlying a scaly surface was suspicious for squamous cell carcinoma (SCC). Since this was a virtual visit, and the lesion required pathology due to the likelihood of cancer, the patient was brought into the clinic for additional evaluation. A broad-based deep shave biopsy was performed to remove the visible lesion. Pathology showed SCC in situ, with borders uninvolved.
Patients who have had AKs are extremely likely to develop additional AKs. A notable percentage of AKs will, over time, develop into SCC in situ and then invasive SCC if not treated. While cryosurgery of an SK should not result in SCC, it’s most likely that in this case, an AK adjacent to the SK progressed to the SCC in situ.
There are multiple treatments available for SCC in situ. Topical imiquimod has been shown to be somewhat effective in stimulating the immune system, thus leading to resolution of SCC in situ. But there is a significant risk of recurrence. Topical 5-FU can be utilized on a daily or twice daily basis for 2 weeks (or up to several months). The risk of recurrence ranges from 7% to 33%. Electrodesiccation and curettage is often used for SCC in situ, with recurrence rates of 2% to 19%. Cryosurgery for SCC in situ requires an aggressive freeze, with freeze times of up to 30 seconds. Photodynamic therapy also is an option; however, it requires multiple sessions and is more costly than other treatment options.
This patient’s borders were uninvolved on pathology, but it was possible that there was some residual SCC in situ due to the standard “bread loaf slicing” used for routine pathology. To treat possible residual SCC in situ at the wound site and surrounding tissue, the patient was given a prescription for topical 5-FU to apply twice daily for 6 weeks. The patient was instructed to return for follow-up in 6 months, or sooner, if any problems arose.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This pink raised nodule underlying a scaly surface was suspicious for squamous cell carcinoma (SCC). Since this was a virtual visit, and the lesion required pathology due to the likelihood of cancer, the patient was brought into the clinic for additional evaluation. A broad-based deep shave biopsy was performed to remove the visible lesion. Pathology showed SCC in situ, with borders uninvolved.
Patients who have had AKs are extremely likely to develop additional AKs. A notable percentage of AKs will, over time, develop into SCC in situ and then invasive SCC if not treated. While cryosurgery of an SK should not result in SCC, it’s most likely that in this case, an AK adjacent to the SK progressed to the SCC in situ.
There are multiple treatments available for SCC in situ. Topical imiquimod has been shown to be somewhat effective in stimulating the immune system, thus leading to resolution of SCC in situ. But there is a significant risk of recurrence. Topical 5-FU can be utilized on a daily or twice daily basis for 2 weeks (or up to several months). The risk of recurrence ranges from 7% to 33%. Electrodesiccation and curettage is often used for SCC in situ, with recurrence rates of 2% to 19%. Cryosurgery for SCC in situ requires an aggressive freeze, with freeze times of up to 30 seconds. Photodynamic therapy also is an option; however, it requires multiple sessions and is more costly than other treatment options.
This patient’s borders were uninvolved on pathology, but it was possible that there was some residual SCC in situ due to the standard “bread loaf slicing” used for routine pathology. To treat possible residual SCC in situ at the wound site and surrounding tissue, the patient was given a prescription for topical 5-FU to apply twice daily for 6 weeks. The patient was instructed to return for follow-up in 6 months, or sooner, if any problems arose.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Shimizu I, Cruz A, Chang KH, et al. Treatment of squamous cell carcinoma in situ: a review. Dermatol Surg. 2011;37:1394-1411.
Shimizu I, Cruz A, Chang KH, et al. Treatment of squamous cell carcinoma in situ: a review. Dermatol Surg. 2011;37:1394-1411.