User login
Itchy scalp with scale
An 11-year-old boy sought care at a small village’s health center in Panama for scalp itching and subtle hair loss. He was seen by a family physician (RU) and a team of medical students who were there as part of a humanitarian trip. The patient denied any hair pulling. He had a history of treatment for head lice.
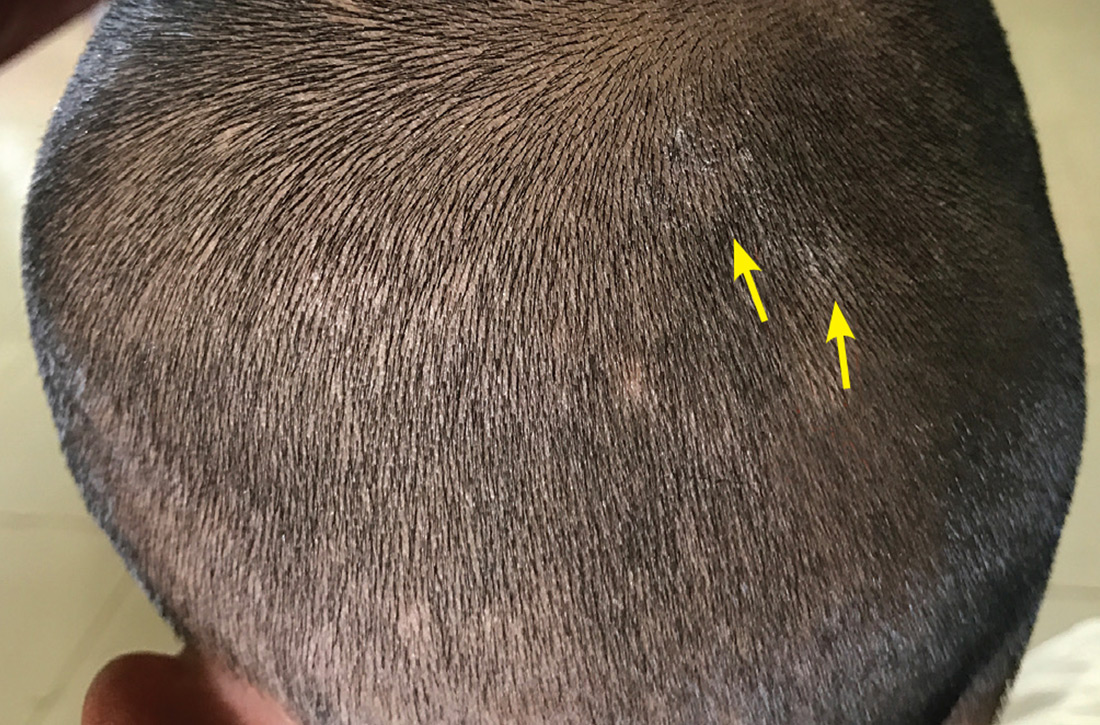
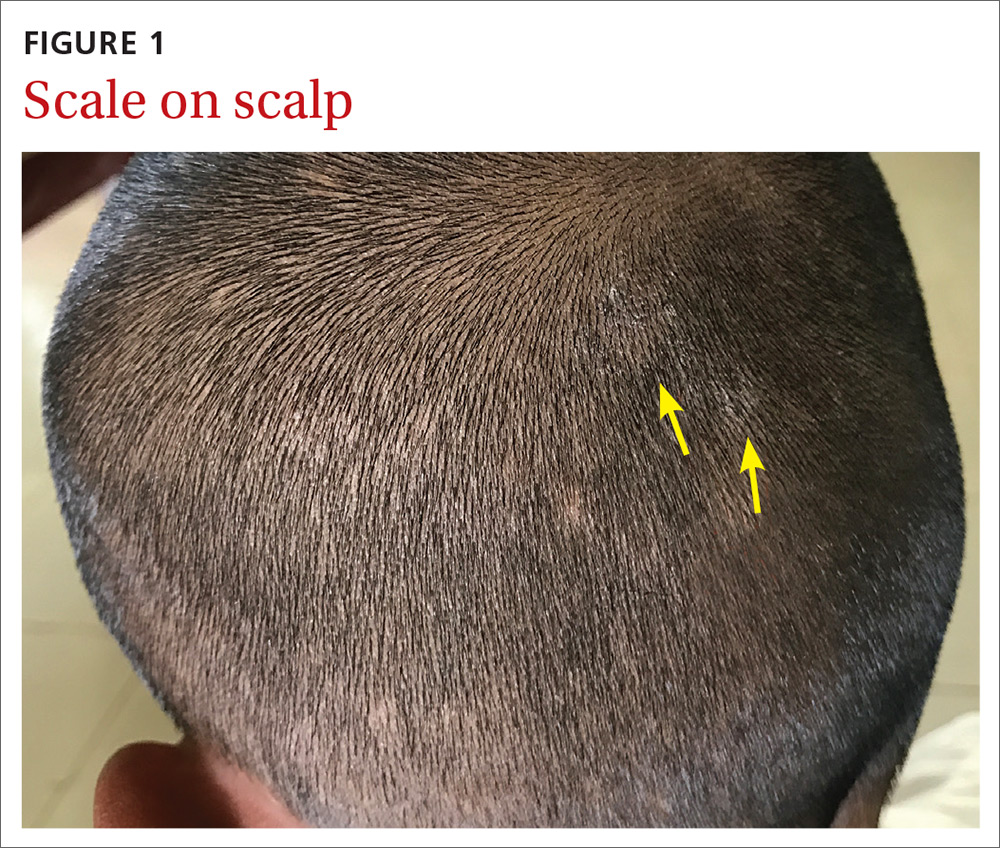
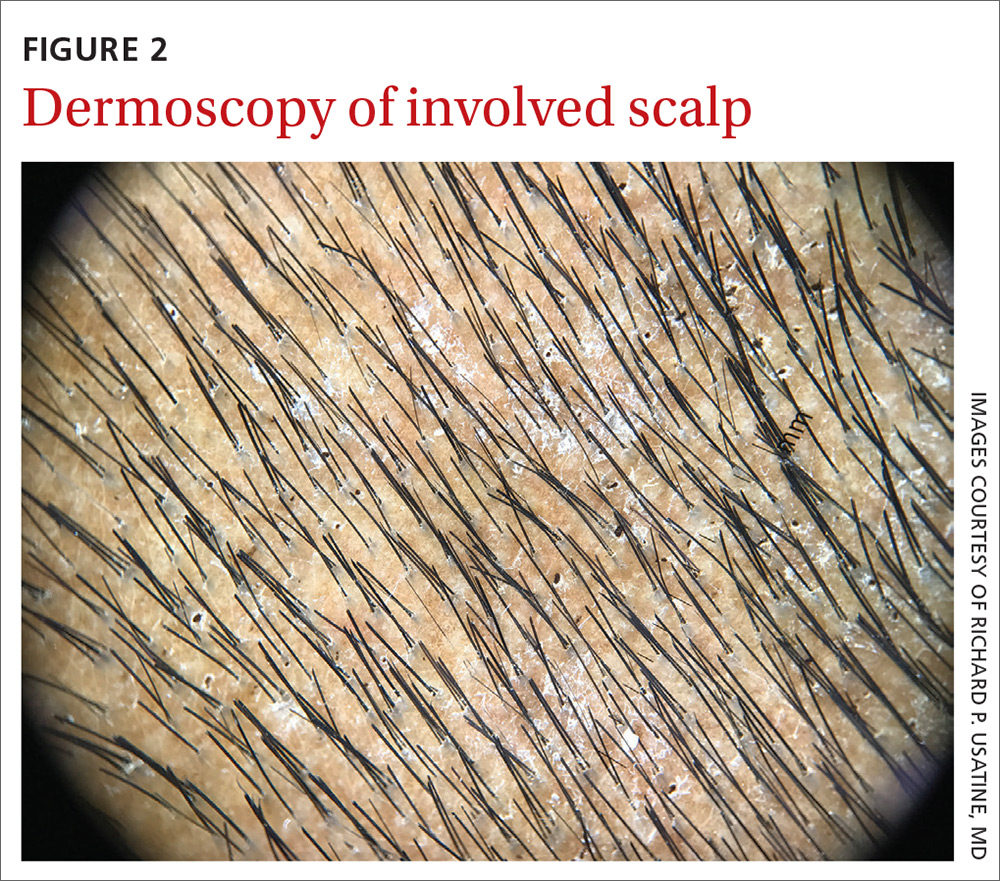
Our physical examination revealed mild alopecia and scaling on the scalp (FIGURE 1), but what we saw through the dermatoscope (FIGURE 2) made the diagnosis clear.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea capitis
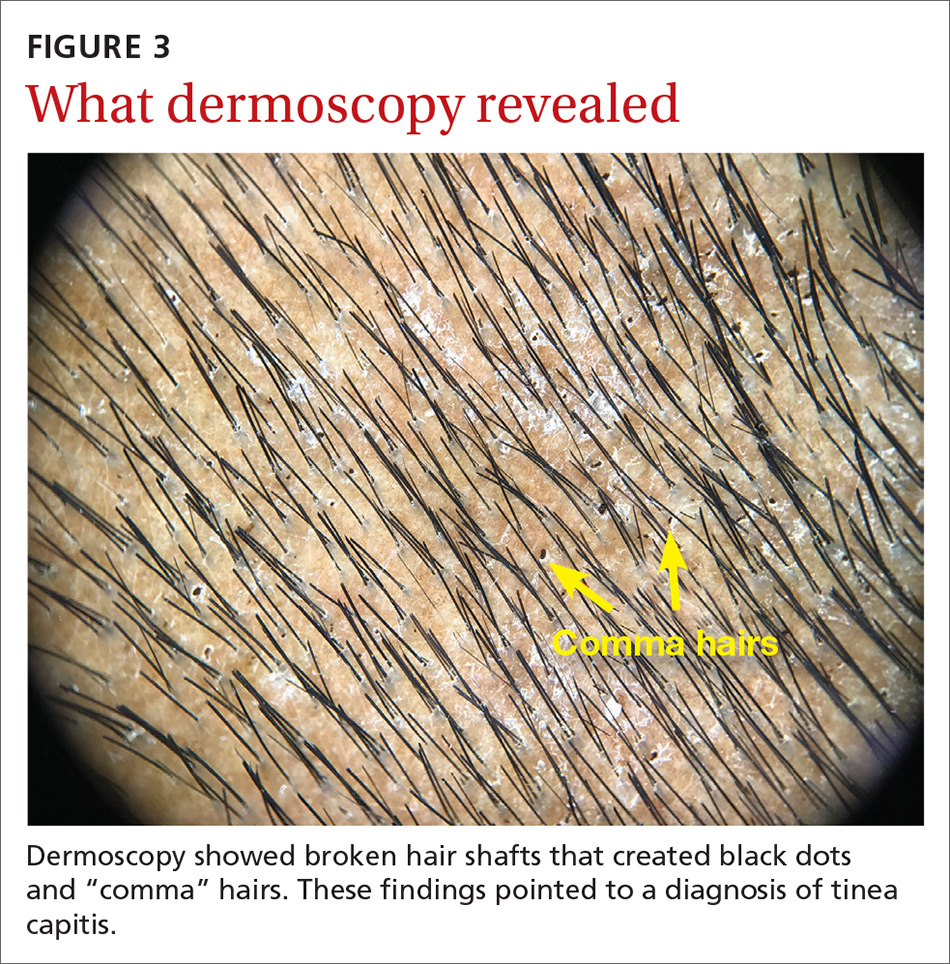
On dermatoscopic examination (10× magnification), there were numerous “black dots” or broken hair shafts within patches of hair loss (FIGURE 3), which is indicative of tinea capitis.1,2 This condition causes hair shafts to break, creating “comma hairs” and black dots. The hairs are uniform in thickness and color and bend distally, like a comma.3
Tinea capitis (commonly called ringworm of the scalp) is a fungal infection caused by Trichophyton and Microsporum dermatophytes. It is the most common pediatric dermatophyte infection in the world; the usual age of onset is 5 to 10 years.2 The incidence of tinea capitis in the United States is not known because cases are no longer registered by public health agencies. That said, a Northern California study that tracked occurrences in children younger than 15 years from 1998 to 2007 found that the incidence was on the decline and lower in girls compared to boys (111.9 vs 146.4, respectively, in 1998; 27.9 vs 39.9, respectively, in 2007).4 Incidence rates were calculated per 10,000 eligible children.4
Tinea capitis can spread by contact with infected individuals and contaminated objects, including combs, towels, toys, and bedding.1 Fungal spores can remain viable on these surfaces for months.
In a study of 69 patients with tinea capitis (23 females, 46 males; mean age, 12 years), the risk factors for spreading infection included participation in sports, contact with an animal, a recent haircut, and use of a swimming pool.5
4 conditions you’ll want to rule out
The following conditions should be considered as part of the differential when a patient presents with an itchy scalp and/or hair loss.
Continue to: Psoriasis of the scalp...
Psoriasis of the scalp is characterized by scaling of the scalp along with crusted plaques. It is often accompanied by similar psoriatic plaques on the elbows, knees, and other areas of the body. Examination of our patient showed no psoriatic plaques.
Seborrhea of the scalp (also known as dandruff) is a very common diagnosis. However, it is unlikely to cause hair loss. It has widespread involvement of the scalp compared to tinea capitis, which is local and patchy. Our patient’s patches of hair loss indicated that seborrhea was unlikely.
Alopecia areata. Individuals develop this condition due to an autoimmune process affecting hair follicles. However, the resulting hair loss does not cause significant scaling, inflammation, scarring, or pain in the affected area. Further, this condition can cause the loss of the entire hair shaft.
Trichotillomania is an impulse control disorder that causes patients to pull out their own hair. There is no scaling of the scalp in this condition.
A dermatoscope can beuseful in making the Dx
Although clinical appearance and patient presentation are adequate to establish the diagnosis of tinea capitis, this case demonstrates the utility of a dermatoscope in making the diagnosis of tinea capitis. Previous studies have shown that dermoscopy allows for rapid identification of the broken hair shafts, which are a key distinction from alopecia areata.3,6
Microscopic inspection. Samples from the scaling of the scalp can be examined with potassium hydroxide (KOH) on a microscope slide. Hyphae, spores, and endo/ectothrix invasion can be seen through the microsope.
Continue to: Laboratory testing is helpful, but not needed.
Laboratory testing is helpful, but not needed. Testing for tinea capitis would require that you obtain a sample from the affected area using a swab, edge of a scalpel blade, or scalp brush.7 Because treatment can require weeks of medication, diagnosis should be confirmed with a KOH or culture when possible.
Newer antifungalsprovide a Tx advantage
Oral antifungal medications are the treatment of choice for tinea capitis. Newer antifungals, such as terbinafine and fluconazole, require a 3- to 6-week course compared to the standard 6- to 8-week course of griseofulvin.1 Also, antifungal shampoos—such as those that contain selenium sulfide—may be used for topical treatment but only as adjuvant therapy.1,2
For our patient, we dispensed a 3-week course of oral fluconazole, 3 to 6 mg/kg, to be given daily by his parents. We also recommended the use of an antidandruff shampoo, if possible. The treatment outcome was not known because our team’s humanitarian global health trip had ended.
1. Usatine R, Smith MA, Mayeaux Jr EJ, Chumley HS. The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019.
2. Handler MZ. Tinea capitis. Medscape. https://emedicine.medscape.com/article/1091351-overview. Updated February 21, 2020. Accessed November 30, 2020.
3. Hernández-Bel P, Malvehy J, Crocker A, et al. Comma hairs: a new dermoscopic marker for tinea capitis [in Spanish]. Actas Dermosifiliogr. 2012;103:836-837.
4. Mirmirani P, Lue-Yen T. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol. 2013;69:916-921.
5. Mikaeili A, Kavaoussi H, Hashemian AH, et al. Clinico-mycological profile of tinea capitis and its comparative response to griseofulvin versus terbinafine. Curr Med Mycol. 2019;5:15-20.
6. Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. Journal of the American Academy of Dermatology. 2008;59(suppl 5):S77-S79.
An 11-year-old boy sought care at a small village’s health center in Panama for scalp itching and subtle hair loss. He was seen by a family physician (RU) and a team of medical students who were there as part of a humanitarian trip. The patient denied any hair pulling. He had a history of treatment for head lice.
Our physical examination revealed mild alopecia and scaling on the scalp (FIGURE 1), but what we saw through the dermatoscope (FIGURE 2) made the diagnosis clear.


WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea capitis
On dermatoscopic examination (10× magnification), there were numerous “black dots” or broken hair shafts within patches of hair loss (FIGURE 3), which is indicative of tinea capitis.1,2 This condition causes hair shafts to break, creating “comma hairs” and black dots. The hairs are uniform in thickness and color and bend distally, like a comma.3
Tinea capitis (commonly called ringworm of the scalp) is a fungal infection caused by Trichophyton and Microsporum dermatophytes. It is the most common pediatric dermatophyte infection in the world; the usual age of onset is 5 to 10 years.2 The incidence of tinea capitis in the United States is not known because cases are no longer registered by public health agencies. That said, a Northern California study that tracked occurrences in children younger than 15 years from 1998 to 2007 found that the incidence was on the decline and lower in girls compared to boys (111.9 vs 146.4, respectively, in 1998; 27.9 vs 39.9, respectively, in 2007).4 Incidence rates were calculated per 10,000 eligible children.4

Tinea capitis can spread by contact with infected individuals and contaminated objects, including combs, towels, toys, and bedding.1 Fungal spores can remain viable on these surfaces for months.
In a study of 69 patients with tinea capitis (23 females, 46 males; mean age, 12 years), the risk factors for spreading infection included participation in sports, contact with an animal, a recent haircut, and use of a swimming pool.5
4 conditions you’ll want to rule out
The following conditions should be considered as part of the differential when a patient presents with an itchy scalp and/or hair loss.
Continue to: Psoriasis of the scalp...
Psoriasis of the scalp is characterized by scaling of the scalp along with crusted plaques. It is often accompanied by similar psoriatic plaques on the elbows, knees, and other areas of the body. Examination of our patient showed no psoriatic plaques.
Seborrhea of the scalp (also known as dandruff) is a very common diagnosis. However, it is unlikely to cause hair loss. It has widespread involvement of the scalp compared to tinea capitis, which is local and patchy. Our patient’s patches of hair loss indicated that seborrhea was unlikely.
Alopecia areata. Individuals develop this condition due to an autoimmune process affecting hair follicles. However, the resulting hair loss does not cause significant scaling, inflammation, scarring, or pain in the affected area. Further, this condition can cause the loss of the entire hair shaft.
Trichotillomania is an impulse control disorder that causes patients to pull out their own hair. There is no scaling of the scalp in this condition.
A dermatoscope can beuseful in making the Dx
Although clinical appearance and patient presentation are adequate to establish the diagnosis of tinea capitis, this case demonstrates the utility of a dermatoscope in making the diagnosis of tinea capitis. Previous studies have shown that dermoscopy allows for rapid identification of the broken hair shafts, which are a key distinction from alopecia areata.3,6
Microscopic inspection. Samples from the scaling of the scalp can be examined with potassium hydroxide (KOH) on a microscope slide. Hyphae, spores, and endo/ectothrix invasion can be seen through the microsope.
Continue to: Laboratory testing is helpful, but not needed.
Laboratory testing is helpful, but not needed. Testing for tinea capitis would require that you obtain a sample from the affected area using a swab, edge of a scalpel blade, or scalp brush.7 Because treatment can require weeks of medication, diagnosis should be confirmed with a KOH or culture when possible.
Newer antifungalsprovide a Tx advantage
Oral antifungal medications are the treatment of choice for tinea capitis. Newer antifungals, such as terbinafine and fluconazole, require a 3- to 6-week course compared to the standard 6- to 8-week course of griseofulvin.1 Also, antifungal shampoos—such as those that contain selenium sulfide—may be used for topical treatment but only as adjuvant therapy.1,2
For our patient, we dispensed a 3-week course of oral fluconazole, 3 to 6 mg/kg, to be given daily by his parents. We also recommended the use of an antidandruff shampoo, if possible. The treatment outcome was not known because our team’s humanitarian global health trip had ended.
An 11-year-old boy sought care at a small village’s health center in Panama for scalp itching and subtle hair loss. He was seen by a family physician (RU) and a team of medical students who were there as part of a humanitarian trip. The patient denied any hair pulling. He had a history of treatment for head lice.
Our physical examination revealed mild alopecia and scaling on the scalp (FIGURE 1), but what we saw through the dermatoscope (FIGURE 2) made the diagnosis clear.


WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea capitis
On dermatoscopic examination (10× magnification), there were numerous “black dots” or broken hair shafts within patches of hair loss (FIGURE 3), which is indicative of tinea capitis.1,2 This condition causes hair shafts to break, creating “comma hairs” and black dots. The hairs are uniform in thickness and color and bend distally, like a comma.3
Tinea capitis (commonly called ringworm of the scalp) is a fungal infection caused by Trichophyton and Microsporum dermatophytes. It is the most common pediatric dermatophyte infection in the world; the usual age of onset is 5 to 10 years.2 The incidence of tinea capitis in the United States is not known because cases are no longer registered by public health agencies. That said, a Northern California study that tracked occurrences in children younger than 15 years from 1998 to 2007 found that the incidence was on the decline and lower in girls compared to boys (111.9 vs 146.4, respectively, in 1998; 27.9 vs 39.9, respectively, in 2007).4 Incidence rates were calculated per 10,000 eligible children.4

Tinea capitis can spread by contact with infected individuals and contaminated objects, including combs, towels, toys, and bedding.1 Fungal spores can remain viable on these surfaces for months.
In a study of 69 patients with tinea capitis (23 females, 46 males; mean age, 12 years), the risk factors for spreading infection included participation in sports, contact with an animal, a recent haircut, and use of a swimming pool.5
4 conditions you’ll want to rule out
The following conditions should be considered as part of the differential when a patient presents with an itchy scalp and/or hair loss.
Continue to: Psoriasis of the scalp...
Psoriasis of the scalp is characterized by scaling of the scalp along with crusted plaques. It is often accompanied by similar psoriatic plaques on the elbows, knees, and other areas of the body. Examination of our patient showed no psoriatic plaques.
Seborrhea of the scalp (also known as dandruff) is a very common diagnosis. However, it is unlikely to cause hair loss. It has widespread involvement of the scalp compared to tinea capitis, which is local and patchy. Our patient’s patches of hair loss indicated that seborrhea was unlikely.
Alopecia areata. Individuals develop this condition due to an autoimmune process affecting hair follicles. However, the resulting hair loss does not cause significant scaling, inflammation, scarring, or pain in the affected area. Further, this condition can cause the loss of the entire hair shaft.
Trichotillomania is an impulse control disorder that causes patients to pull out their own hair. There is no scaling of the scalp in this condition.
A dermatoscope can beuseful in making the Dx
Although clinical appearance and patient presentation are adequate to establish the diagnosis of tinea capitis, this case demonstrates the utility of a dermatoscope in making the diagnosis of tinea capitis. Previous studies have shown that dermoscopy allows for rapid identification of the broken hair shafts, which are a key distinction from alopecia areata.3,6
Microscopic inspection. Samples from the scaling of the scalp can be examined with potassium hydroxide (KOH) on a microscope slide. Hyphae, spores, and endo/ectothrix invasion can be seen through the microsope.
Continue to: Laboratory testing is helpful, but not needed.
Laboratory testing is helpful, but not needed. Testing for tinea capitis would require that you obtain a sample from the affected area using a swab, edge of a scalpel blade, or scalp brush.7 Because treatment can require weeks of medication, diagnosis should be confirmed with a KOH or culture when possible.
Newer antifungalsprovide a Tx advantage
Oral antifungal medications are the treatment of choice for tinea capitis. Newer antifungals, such as terbinafine and fluconazole, require a 3- to 6-week course compared to the standard 6- to 8-week course of griseofulvin.1 Also, antifungal shampoos—such as those that contain selenium sulfide—may be used for topical treatment but only as adjuvant therapy.1,2
For our patient, we dispensed a 3-week course of oral fluconazole, 3 to 6 mg/kg, to be given daily by his parents. We also recommended the use of an antidandruff shampoo, if possible. The treatment outcome was not known because our team’s humanitarian global health trip had ended.
1. Usatine R, Smith MA, Mayeaux Jr EJ, Chumley HS. The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019.
2. Handler MZ. Tinea capitis. Medscape. https://emedicine.medscape.com/article/1091351-overview. Updated February 21, 2020. Accessed November 30, 2020.
3. Hernández-Bel P, Malvehy J, Crocker A, et al. Comma hairs: a new dermoscopic marker for tinea capitis [in Spanish]. Actas Dermosifiliogr. 2012;103:836-837.
4. Mirmirani P, Lue-Yen T. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol. 2013;69:916-921.
5. Mikaeili A, Kavaoussi H, Hashemian AH, et al. Clinico-mycological profile of tinea capitis and its comparative response to griseofulvin versus terbinafine. Curr Med Mycol. 2019;5:15-20.
6. Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. Journal of the American Academy of Dermatology. 2008;59(suppl 5):S77-S79.
1. Usatine R, Smith MA, Mayeaux Jr EJ, Chumley HS. The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY: McGraw-Hill; 2019.
2. Handler MZ. Tinea capitis. Medscape. https://emedicine.medscape.com/article/1091351-overview. Updated February 21, 2020. Accessed November 30, 2020.
3. Hernández-Bel P, Malvehy J, Crocker A, et al. Comma hairs: a new dermoscopic marker for tinea capitis [in Spanish]. Actas Dermosifiliogr. 2012;103:836-837.
4. Mirmirani P, Lue-Yen T. Epidemiologic trends in pediatric tinea capitis: a population-based study from Kaiser Permanente Northern California. J Am Acad Dermatol. 2013;69:916-921.
5. Mikaeili A, Kavaoussi H, Hashemian AH, et al. Clinico-mycological profile of tinea capitis and its comparative response to griseofulvin versus terbinafine. Curr Med Mycol. 2019;5:15-20.
6. Slowinska M, Rudnicka L, Schwartz RA, et al. Comma hairs: a dermatoscopic marker for tinea capitis: a rapid diagnostic method. Journal of the American Academy of Dermatology. 2008;59(suppl 5):S77-S79.
How to identify and treat common bites and stings
Insect, arachnid, and other arthropod bites and stings are common patient complaints in a primary care office. A thorough history and physical exam can often isolate the specific offender and guide management. In this article, we outline how to identify, diagnose, and treat common bites and stings from bees and wasps; centipedes and spiders; fleas; flies and biting midges; mosquitoes; and ticks, and discuss how high-risk patients should be triaged and referred for additional testing and treatment, such as venom immunotherapy (VIT).
Insects and arachnids:Background and epidemiology
Insects are arthropods with 3-part exoskeletons: head, thorax, and abdomen. They have 6 jointed legs, compound eyes, and antennae. There are approximately 91,000 insect species in the United States, the most abundant orders being Coleoptera (beetles), Diptera (flies), and Hymenoptera (includes ants, bees, wasps, and sawflies).1
The reported incidence of insect bites and stings varies widely because most people experience mild symptoms and therefore do not seek medical care. Best statistics are for Hymenoptera stings, which are more likely to cause a severe reaction. In Europe, 56% to 94% of the general population has reported being bitten or stung by one of the Hymenoptera species.2 In many areas of Australia, the incidence of jack jumper ant stings is only 2% to 3%3; in the United States, 55% of people report being stung by nonnative fire ants within 3 weeks of moving into an endemic area.4
Arachnids are some of the earliest terrestrial organisms, of the class Arachnida, which includes scorpions, ticks, spiders, mites, and daddy longlegs (harvestmen).5 Arachnids are wingless and characterized by segmented bodies, jointed appendages, and exoskeletons.6,7 In most, the body is separated into 2 segments (the cephalothorax and abdomen), except for mites, ticks, and daddy longlegs, in which the entire body comprises a single segment.5
Arthropod bites are common in the United States; almost one-half are caused by spiders.7 Brown recluse (Loxosceles spp) and black widow (Latrodectus spp) spider bites are the most concerning: Although usually mild, these bites can be life-threatening but are rarely fatal. In 2013, almost 3500 bites by black widow and brown recluse spiders were reported.8
Risk factors
Risk factors for insect, arachnid, and other arthropod bites and stings are primarily environmental. People who live or work in proximity of biting or stinging insects (eg, gardeners and beekeepers) are more likely to be affected; so are those who work with animals or live next to standing water or grassy or wooded locales.
Continue to: There are also risk factors...
There are also risk factors for a systemic sting reaction:
- A sting reaction < 2 months earlier increases the risk of a subsequent systemic sting reaction by ≥ 50%.9
- Among beekeepers, paradoxically, the risk of a systemic reaction is higher in those stung < 15 times a year than in those stung > 200 times.10
- Patients with an elevated baseline serum level of tryptase (reference range, < 11.4 ng/mL), which is part of the allergenic response, or with biopsy-proven systemic mastocytosis are at increased risk of a systemic sting reaction.11
Presentation: Signs and symptomsvary with severity
Insect bites and stings usually cause transient local inflammation and, occasionally, a toxic reaction. Allergic hypersensitivity can result in a large local reaction or a generalized systemic reaction12:
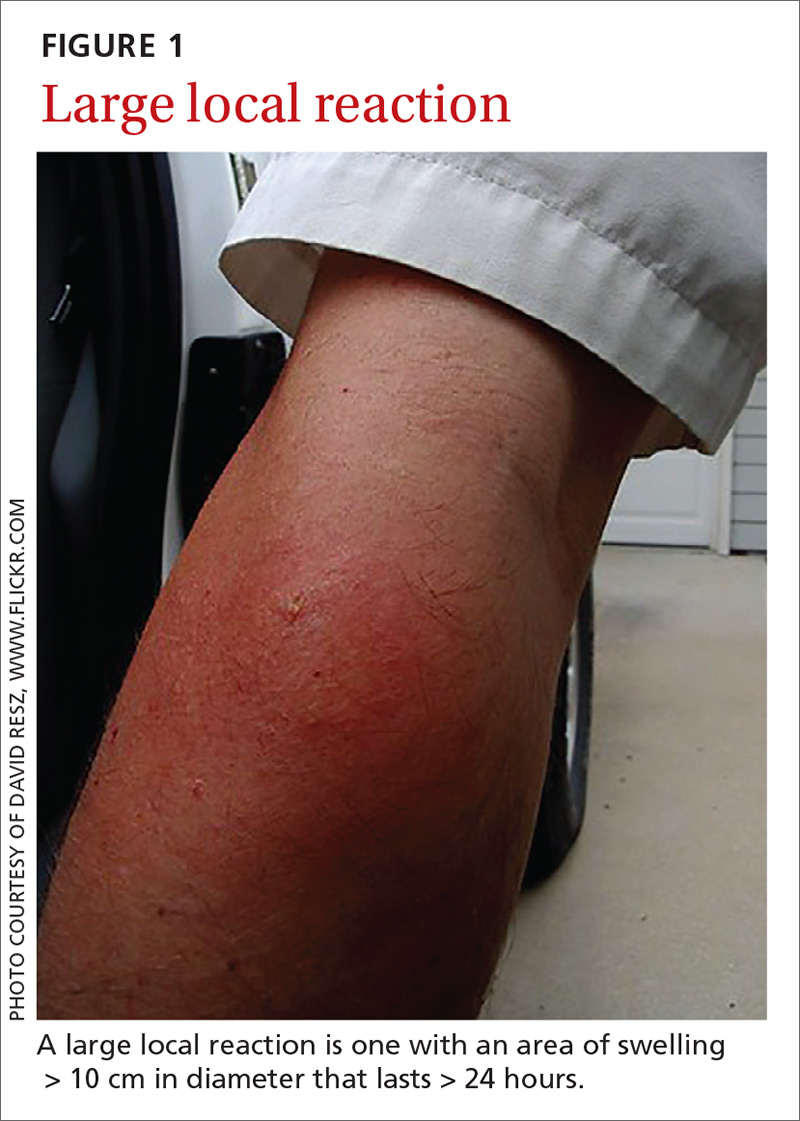
- A small local reaction is transient and mild, develops directly at the site of the sting, and can last several days.13
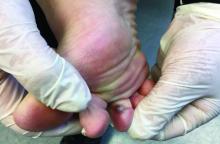
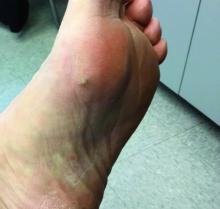
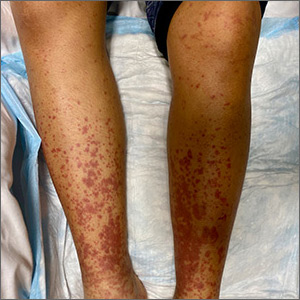
- A large (or significant) local reaction, defined as swelling > 10 cm in diameter (FIGURE 1) and lasting > 24 hours, occurs in 2% to 26% of people who have been bitten or stung.14 This is an immunoglobulin (Ig) E–mediated late-phase reaction that can be accompanied by fatigue and nausea.12,13,15 For a patient with a large local reaction, the risk of a concomitant systemic reaction is 4% to 10%, typically beginning within 30 minutes after envenomation or, possibly, delayed for several hours or marked by a biphasic interval.16
- Characteristics of a systemic reaction are urticaria, angioedema, bronchospasm, large-airway edema, hypotension, and other clinical manifestations of anaphylaxis.17 In the United States, a systemic sting reaction is reported to occur in approximately 3% of bite and sting victims. Mortality among the general population from a systemic bite or sting reaction is 0.16 for every 100,000 people,2 and at least 40 to 100 die every year in the United States from anaphylaxis resulting from an insect bite or sting.18
- The most severe anaphylactic reactions involve the cardiovascular and respiratory systems, commonly including hypotension and symptoms of upper- or lower-airway obstruction. Laryngeal edema and circulatory failure are the most common mechanisms of anaphylactic death.19
Bees and wasps
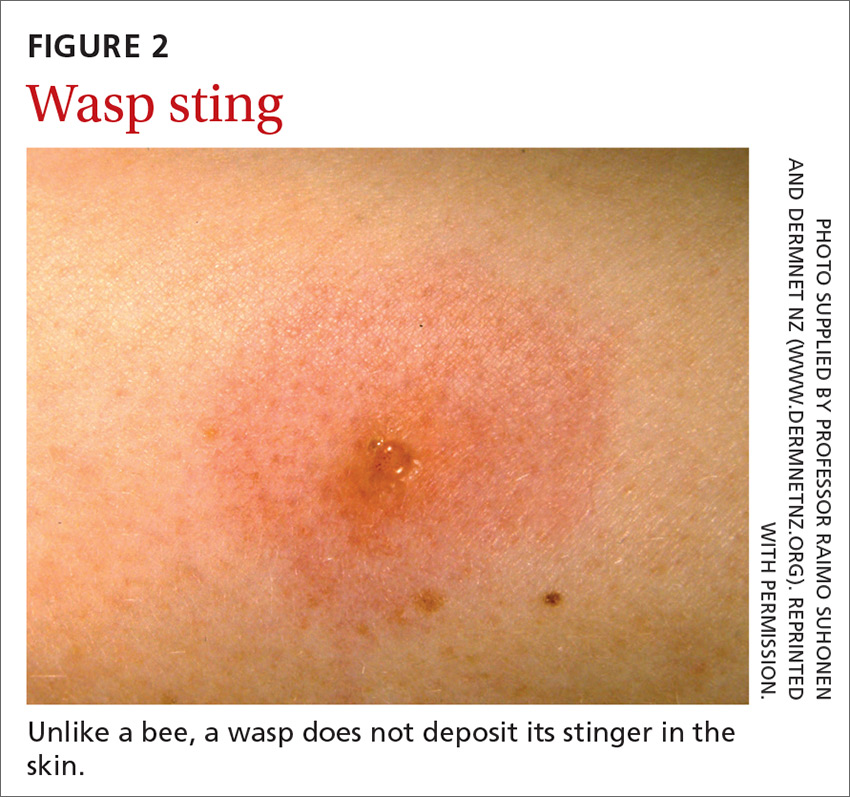
Hymenoptera stinging insects include the family Apidae (honey bee, bumblebee, and sweat bee) and Vespidae (yellow jacket, yellow- and white-faced hornets, and paper wasp). A worker honey bee can sting only once, leaving its barbed stinger in the skin; a wasp, hornet, and yellow jacket can sting multiple times (FIGURE 2).2
Continue to: Bee and wasp sting...
Bee and wasp sting allergies are the most common insect venom allergic reactions. A bee sting is more likely to lead to a severe allergic reaction than a wasp sting. Allergic reactions to hornet and bumblebee stings are less common but can occur in patients already sensitized to wasp and honey bee stings.20,21
Management. Remove honey bee stingers by scraping the skin with a fingernail or credit card. Ideally, the stinger should be removed in the first 30 seconds, before the venom sac empties. Otherwise, intense local inflammation, with possible lymphangitic streaking, can result.22
For guidance on localized symptomatic care of bee and wasp stings and bites and stings from other sources discussed in this article, see “Providing relief and advanced care” on page E6.
Centipedes and spiders
Centipedes are arthropods of the class Chilopoda, subphylum Myriapoda, that are characterized by repeating linear (metameric) segments, each containing 1 pair of legs.23 Centipedes have a pair of poison claws behind the head that are used to paralyze prey—usually, small insects.23,24 The bite of a larger centipede can cause a painful reaction that generally subsides after a few hours but can last several days. Centipede bites are usually nonfatal to humans.23
Spiders belong to the class Arachnida, order Araneae. They have 8 legs with chelicerae (mouthpiece, or “jaws”) that inject venom into prey.25 Most spiders found in the United States cannot bite through human skin.26,27 Common exceptions are black widow and brown recluse spiders, which each produce a distinct toxic venom that can cause significant morbidity in humans through a bite, although bites are rarely fatal.26,27
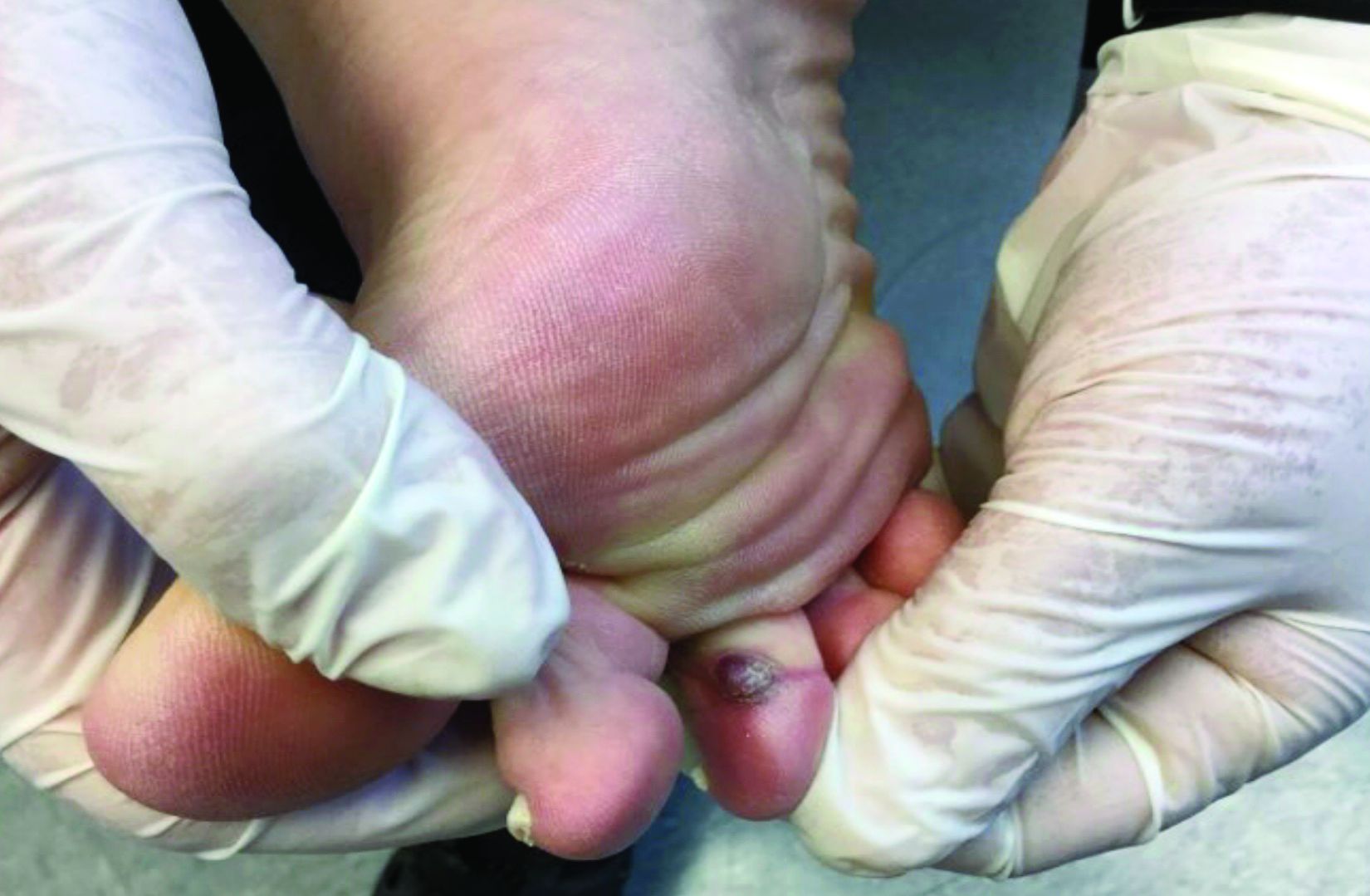
The brown recluse spider is described as having a violin-shaped marking on the abdomen; the body is yellowish, tan, or dark brown. A bite can produce tiny fang marks and cause dull pain at the site of the bite that spreads quickly; myalgia; and pain in the stomach, back, chest, and legs.28,29 The bite takes approximately 7 days to resolve. In a minority of cases, a tender erythematous halo develops, followed by a severe necrotic ulcer, or loxoscelism (FIGURE 3; 40% of cases) or scarring (13%), or both.29,30
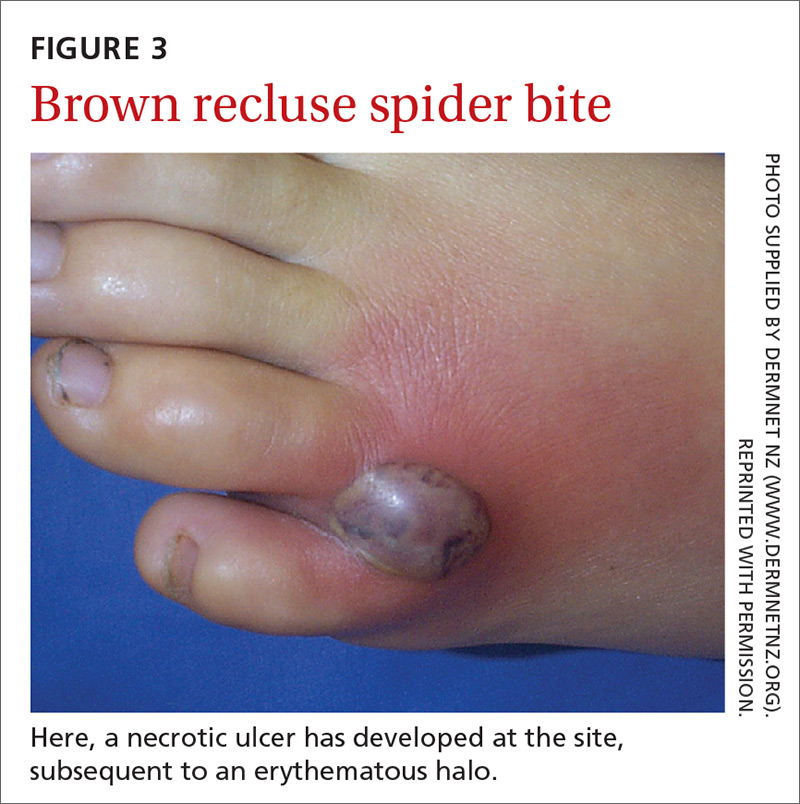
Continue to: In contrast...
In contrast, the body of a black widow spider is black; females exhibit a distinctive red or yellow hourglass marking on their ventral aspect.28,31 The pinprick sensation of a bite leads to symptoms that can include erythema, swelling, pain, stiffness, chills, fever, nausea, and stomach pain.30,32
Management. Again, see “Providing relief and advanced care” on page E6. Consider providing antivenin treatment for moderate or severe bites of brown recluse and black widow spiders.
Fleas
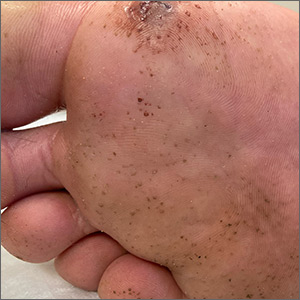
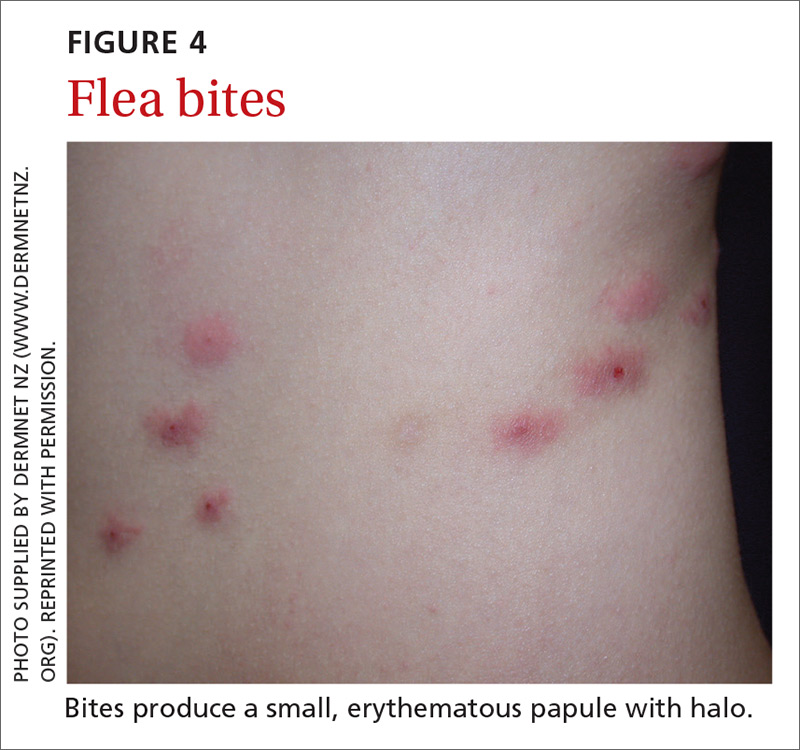
Fleas are members of the order Siphonaptera. They are small (1.5-3.2 mm long), reddish brown, wingless, blood-sucking insects with long legs that allow them to jump far (12 or 13 inches) and high (6 or 7 inches).33 Domesticated cats and dogs are the source of most flea infestations, resulting in an increased risk of exposure for humans.34,35 Flea bites, which generally occur on lower extremities, develop into a small, erythematous papule with a halo (FIGURE 4) and associated mild edema, and cause intense pruritus 30 minutes after the bite.35-37
Fleas are a vector for severe microbial infections, including bartonellosis, bubonic plague, cat-flea typhus, murine typhus, cat-scratch disease, rickettsial disease, and tularemia. Tungiasis is an inflammatory burrowing flea infestation—not a secondary infection for which the flea is a vector.34,35
Preventive management. Repellents, including products that contain DEET (N,N-diethyl-meta-toluamide), picaridin (2-[2-hydroxyethyl]-1-piperidinecarboxylic acid 1-methylpropyl ester), and PMD (p-menthane-3,8-diol, a chemical constituent of Eucalyptus citriodora oil) can be used to prevent flea bites in humans.33,38 Studies show that the scent of other botanic oils, including lavender, cedarwood, and peppermint, can also help prevent infestation by fleas; however, these compounds are not as effective as traditional insect repellents.33,38
Flea control is difficult, requiring a multimodal approach to treating the infested animal and its environment.39 Treatment of the infested domestic animal is the primary method of preventing human bites. Nonpesticidal control involves frequent cleaning of carpeting, furniture, animal bedding, and kennels. Insecticides can be applied throughout the house to combat severe infestation.33,38
Continue to: The Centers for Disease Control and Prevention...
The Centers for Disease Control and Prevention provide a general introduction to getting rid of fleas for pet owners.40 For specific guidance on flea-eradication strategies and specific flea-control products, advise patients to seek the advice of their veterinarian.
Flies and biting midges
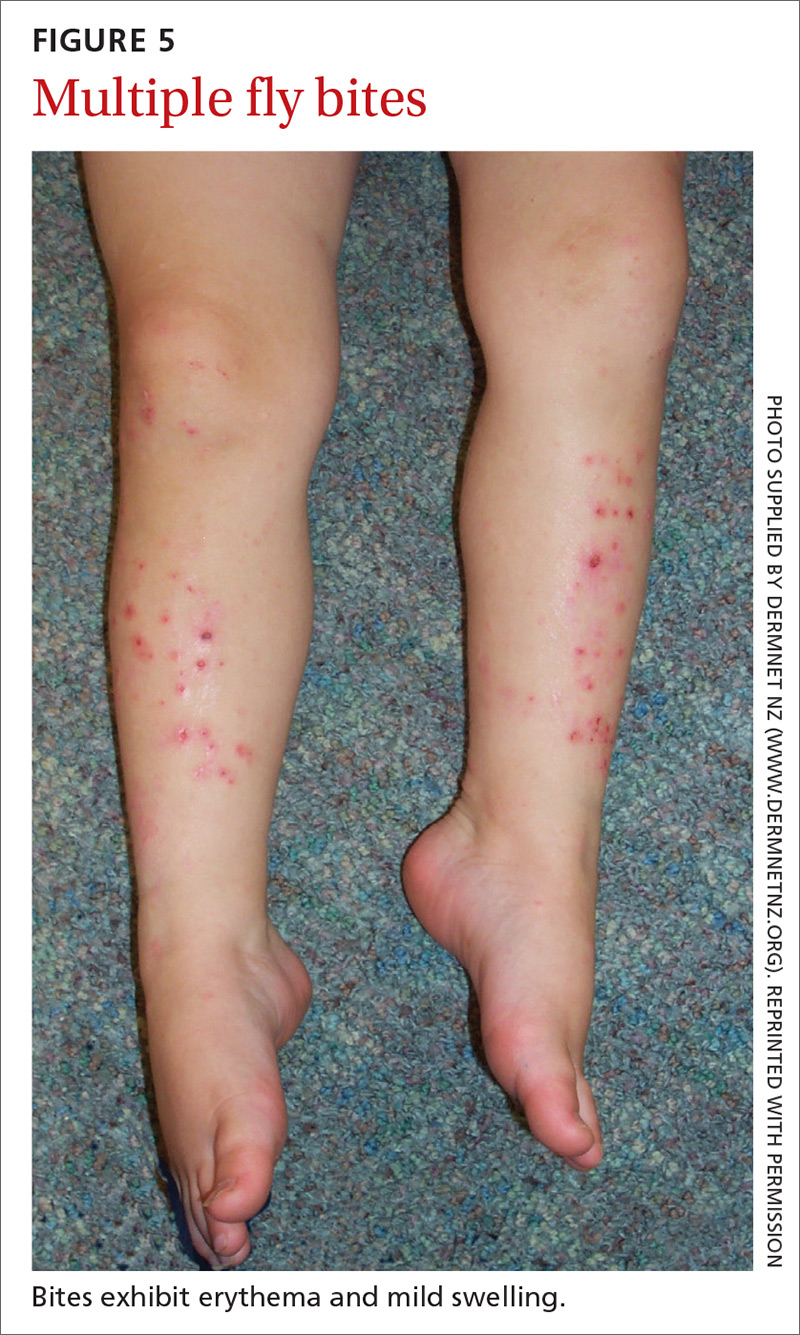
Flies are 2-winged insects belonging to the order Diptera. Several fly species can bite, causing a local inflammatory reaction; these include black flies, deer flies, horse flies, and sand flies. Signs and symptoms of a fly bite include pain, pruritus, erythema, and mild swelling (FIGURE 5).41,42 Flies can transmit several infections, including bartonellosis, enteric bacterial disease (eg, caused by Campylobacter spp), leishmaniasis, loiasis, onchocerciasis, and trypanosomiasis.43
Biting midges, also called “no-see-ums,” biting gnats, moose flies, and “punkies,”44 are tiny (1-3 mm long) blood-sucking flies.45 Bitten patients often report not having seen the midge because it is so small. The bite typically starts as a small, erythematous papule that develops into a dome-shaped blister and can be extraordinarily pruritic and painful.44 The majority of people who have been bitten develop a hypersensitivity reaction, which usually resolves in a few weeks.
Management. Suppressing adult biting midges with an environmental insecticide is typically insufficient because the insecticide must be sprayed daily to eradicate active midges and generally does not affect larval habitat. Insect repellents and biopesticides, such as oil of lemon eucalyptus, can be effective in reducing the risk of bites.44,45
Mosquitoes
Mosquitoes are flying, blood-sucking insects of the order Diptera and family Culicidae. Anopheles, Culex, and Aedes genera are responsible for most bites of humans.
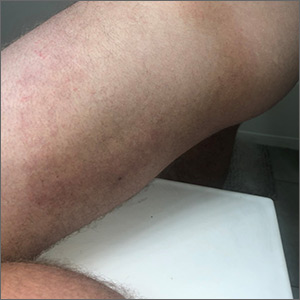
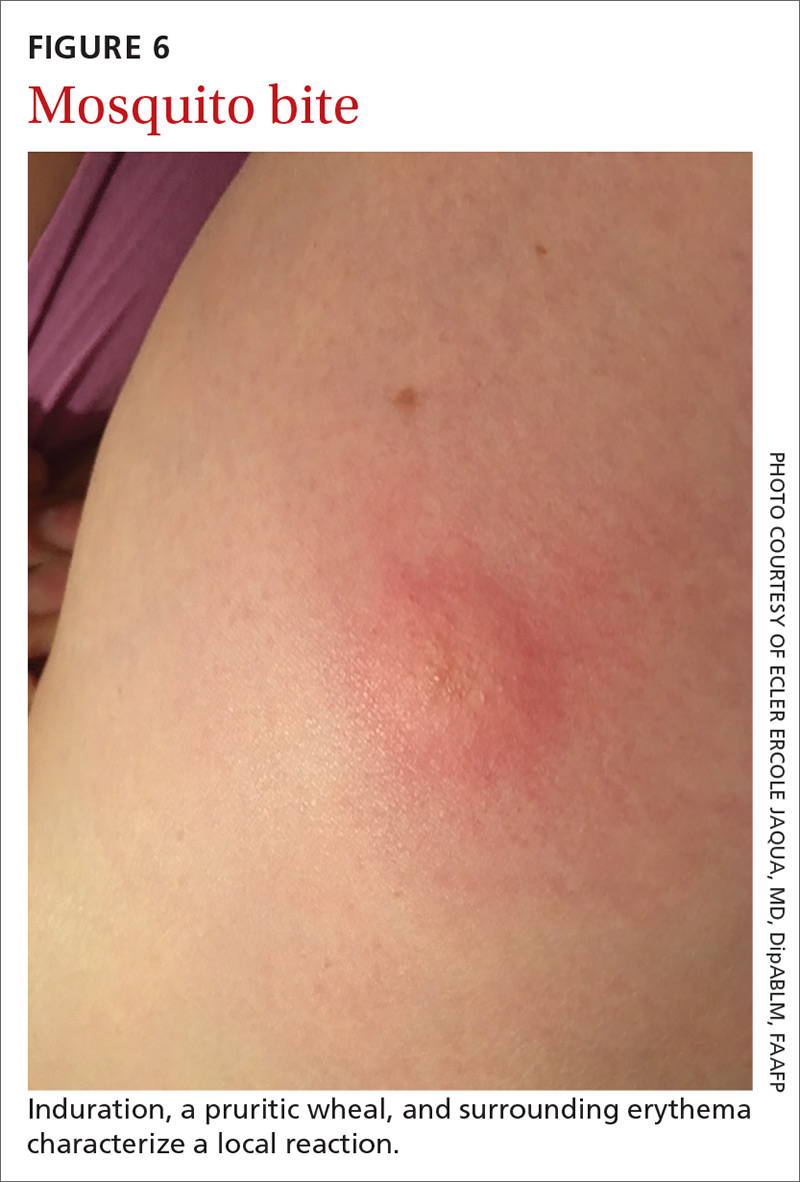
The bite of a mosquito produces an indurated, limited local reaction characterized by a pruritic wheal (3-29 mm in diameter) with surrounding erythema (FIGURE 6) that peaks in approximately 30 minutes, although patients might have a delayed reaction hours later.46 Immunocompromised patients might experience a more significant local inflammatory reaction that is accompanied by low-grade fever, hives, or swollen lymph nodes.46,47
Mosquitoes are a vector for serious infections, including dengue, Japanese encephalitis, malaria, and yellow fever, and disease caused by Chikungunya, West Nile, and Zika viruses.
Continue to: Management
Management. Advise patients to reduce their risk by using insect repellent, sleeping under mosquito netting, and wearing a long-sleeve shirt and long pants when traveling to endemic areas or when a local outbreak occurs.48
Ticks
Ticks belong to the order Parasitiformes and families Ixodidae and Argasidae. Hard ticks are found in brushy fields and tall grasses and can bite and feed on humans for days. Soft ticks are generally found around animal nests.29 Tick bites can cause a local reaction that includes painful, erythematous, inflammatory papular lesions (FIGURE 7).49
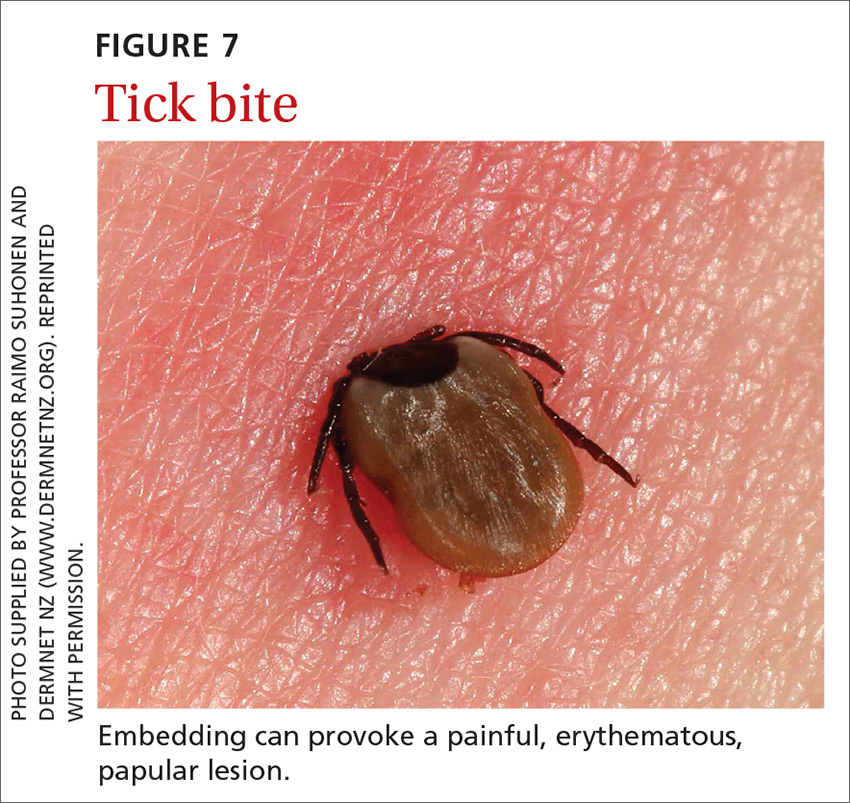
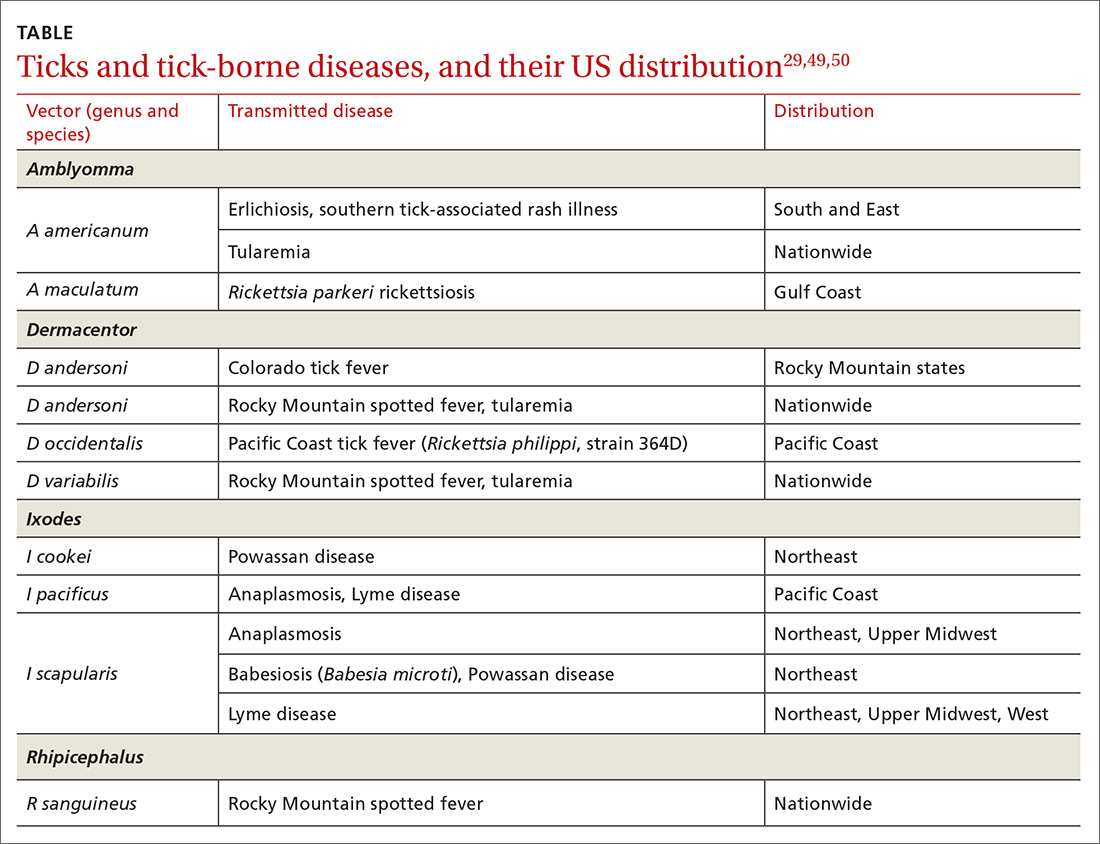
Ticks can transmit several infectious diseases. Depending on the microbial pathogen and the genus and species of tick, it takes 2 to 96 hours for the tick to attach to skin and transmit the pathogen to the human host. The TABLE29,49,50 provides an overview of tick species in the United States, diseases that they can transmit, and the geographic distribution of those diseases.
Management. Ticks should be removed with fine-tipped tweezers. Grasp the body of the tick close to the skin and pull upward while applying steady, even pressure. After removing the tick, clean the bite and the surrounding area with alcohol or with soap and water. Dispose of a live tick by flushing it down the toilet; or, kill it in alcohol and either seal it in a bag with tape or place it in a container.50
Diagnosis and the utilityof special testing
The diagnosis of insect, arachnid, and other arthropod bites and stings depends on the history, including obtaining a record of possible exposure and a travel history; the timing of the bite or sting; and associated signs and symptoms.18,51
Venom skin testing. For Hymenoptera stings, intradermal tests using a venom concentration of 0.001 to 1 μg/mL are positive in 65% to 80% of patients with a history of a systemic insect-sting allergic reaction. A negative venom skin test can occur during the 3-to-6-week refractory period after a sting reaction or many years later, which represents a loss of sensitivity. Positive venom skin tests are used to confirm allergy and identify specific insects to which the patient is allergic.11,12
Continue to: Allergen-specific IgE antibody testing.
Allergen-specific IgE antibody testing. These serum assays—typically, radioallergosorbent testing (RAST)—are less sensitive than venom skin tests. RAST is useful when venom skin testing cannot be performed or when skin testing is negative in a patient who has had a severe allergic reaction to an insect bite or sting. Serum IgE-specific antibody testing is preferred over venom skin testing in patients who are at high risk of anaphylaxis.52,53
Providing reliefand advanced care
Symptomatic treatment of mild bites and stings includes washing the affected area with soap and water and applying a cold compress to reduce swelling.54 For painful lesions, an oral analgesic can be prescribed.
For mild or moderate pruritus, a low- to midpotency topical corticosteroid (eg, hydrocortisone valerate cream 0.2% bid), topical calamine, or pramoxine can be applied,or a nonsedating oral antihistamine, such as loratadine (10 mg/d) or cetirizine (10 mg/d), can be used.14,55 For severe itching, a sedating antihistamine, such as hydroxyzine (10-25 mg every 4 to 6 hours prn), might help relieve symptoms; H1- and H2-receptor antagonists can be used concomitantly.54,55
Significant local symptoms. Large local reactions are treated with a midpotency topical corticosteroid (eg, triamcinolone acetonide cream 0.1% bid) plus an oral antihistamine to relieve pruritus and reduce allergic inflammation. For a more severe reaction, an oral corticosteroid (prednisone 1 mg/kg; maximum dosage, 50 mg/d) can be given for 5 to 7 days.54-56
Management of a necrotic ulcer secondary to a brown recluse spider bite is symptomatic and supportive. The size of these wounds can increase for as long as 10 days after the bite; resolution can require months of wound care, possibly with debridement. Rarely, skin grafting is required.27,28,31
VIT. Some studies show that VIT can improve quality of life in patients with prolonged, frequent, and worsening reactions to insect bites or stings and repeated, unavoidable exposures.55,56 VIT is recommended for patients with systemic hypersensitivity and a positive venom skin test result. It is approximately 95% effective in preventing or reducing severe systemic reactions and reduces the risk of anaphylaxis (see next section) and death.57 The maintenance dosage of VIT is usually 100 μg every 4 to 6 weeks; optimal duration of treatment is 3 to 5 years.58
Continue to: After VIT is complete...
After VIT is complete, counsel patients that a mild systemic reaction is still possible after an insect bite or sting. More prolonged, even lifetime, treatment should be considered for patients who have58,59
- a history of severe, life-threatening allergic reactions to bites and stings
- honey bee sting allergy
- mast-cell disease
- a history of anaphylaxis while receiving VIT.
Absolute contraindications to VIT include a history of serious immune disease, chronic infection, or cancer.58,59
Managing anaphylaxis
This severe allergic reaction can lead to death if untreated. First-line therapy is intramuscular epinephrine, 0.01 mg/kg (maximum single dose, 0.5 mg) given every 5 to 15 minutes.14,60 Epinephrine auto-injectors deliver a fixed dose and are labeled according to weight. Administration of O2 and intravenous fluids is recommended for hemodynamically unstable patients.60,61 Antihistamines and corticosteroids can be used as secondary treatment but should not replace epinephrine.56
After preliminary improvement, patients might decompensate when the epinephrine dose wears off. Furthermore, a biphasic reaction, variously reported in < 5% to as many as 20% of patients,61,62 occurs hours after the initial anaphylactic reaction. Patients should be monitored, therefore, for at least 6 to 8 hours after an anaphylactic reaction, preferably in a facility equipped to treat anaphylaxis.17,56
Before discharge, patients who have had an anaphylactic reaction should be given a prescription for epinephrine and training in the use of an epinephrine auto-injector. Allergen avoidance, along with an emergency plan in the event of a bite or sting, is recommended. Follow-up evaluation with an allergist or immunologist is essential for proper diagnosis and to determine whether the patient is a candidate for VIT.14,17
CORRESPONDENCE
Ecler Ercole Jaqua, MD, DipABLM, FAAFP, 1200 California Street, Suite 240, Redlands, CA 92374; ejaqua@llu.edu.
1. Numbers of insects (species and individuals). Smithsonian BugInfo Web site. www.si.edu/spotlight/buginfo/bugnos. Accessed November 25, 2020.
2. Antonicelli L, Bilò MB, Bonifazi F. Epidemiology of Hymenoptera allergy. Curr Opin Allergy Clin Immunol. 2002;2:341-346.
3. Jack jumper ant allergy. Australasian Society of Clinical Immunology and Allergy (ASCIA) Web site. Updated October 19, 2019. www.allergy.org.au/patients/insect-allergy-bites-and-stings/jack-jumper-ant-allergy. Accessed November 25, 2020.
4. Kemp SF, deShazo RD, Moffit JE, et al. Expanding habitat of the imported fire ant (Solenopsis invicta): a public health concern. J Allergy Clin Immunol. 2000;105:683-691.
5. Goodnight ML. Arachnid. In: Encyclopædia Britannica. 2012. www.britannica.com/animal/arachnid. Accessed November 25, 2020.
6. Despommier DD, Gwadz RW, Hotez PJ. Arachnids. In: Despommier DD, Gwadz RW, Hotez PJ. Parasitic Diseases. 3rd ed. Springer-Verlag; 1995:268-283.
7. Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician. 2007;75:869-873.
8. Mowry JB, Spyker DA, Cantilena LR Jr, McMillan N, Ford M. 2013 Annual report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 31st Annual Report. Clin Toxicol (Phila). 2014;52:1032-1283.
9. Pucci S, Antonicelli L, Bilò MB, et al. Shortness of interval between two stings as risk factor for developing Hymenoptera venom allergy. Allergy.1994;49:894-896.
10. Müller UR. Bee venom allergy in beekeepers and their family members. Curr Opin Allergy Clin Immunol. 2005;5:343-347.
11. Müller UR. Cardiovascular disease and anaphylaxis. Curr Opin Allergy Clin Immunol. 2007;7:337-341.
12. Golden DBK. Stinging insect allergy. Am Fam Physician. 2003;67:2541-2546.
13. Golden DBK, Demain T, Freeman T, et al. Stinging insect hypersensitivity: a practice parameter update 2016. Ann Allergy Asthma Immunol. 2017;118:28-54.
14. Bilò BM, Rueff F, Mosbech H, et al; EAACI Interest Group on Insect Venom Hypersensitivity. Diagnosis of Hymenoptera venom allergy. Allergy. 2005;60:1339-1349.
15. Reisman RE. Insect stings. N Engl J Med. 1994;331:523-527.
16. Pucci S, D’Alò S, De Pasquale T, et al. Risk of anaphylaxis in patients with large local reactions to hymenoptera stings: a retrospective and prospective study. Clin Mol Allergy. 2015;13:21.
17. Golden DBK. Large local reactions to insect stings. J Allergy Clin Immunol Pract. 2015;3:331-334.
18. Clark S, Camargo CA Jr. Emergency treatment and prevention of insect-sting anaphylaxis. Curr Opin Allergy Clin Immunol. 2006;6:279-283.
19. Stinging insect allergy. In: Volcheck GW. Clinical Allergy: Diagnosis and Management. Humana Press; 2009:465-479.
20. Järvinen KM, Celestin J. Anaphylaxis avoidance and management: educating patients and their caregivers. J Asthma Allergy. 2014;7:95-104.
21. Institute for Quality and Efficiency in Health Care (IQWiG). Insect venom allergies: overview. InformedHealth.org. Updated May 7, 2020. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0096282/. Accessed November 25, 2020.
22. Casale TB, Burks AW. Clinical practice. Hymenoptera-sting hypersensitivity. N Engl J Med. 2014;370:1432-1439.
23. Shelley RM. Centipedes and millipedes with emphasis on North American fauna. Kansas School Naturalist. 1999;45:1-16. https://sites.google.com/g.emporia.edu/ksn/ksn-home/vol-45-no-3-centipedes-and-millipedes-with-emphasis-on-n-america-fauna#h.p_JEf3uDlTg0jw. Accessed November 25, 2020.
24. Ogg B. Centipedes and millipedes. Nebraska Extension in Lancaster County Web site. https://lancaster.unl.edu/pest/resources/CentipedeMillipede012.shtml. Accessed November 25, 2020.
25. Cushing PE. Spiders (Arachnida: Araneae). In: Capinera JL, ed. Encyclopedia of Entomology. Springer, Dordrecht; 2008:226.
26. Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician. 2007;75:869-873.
27. The National Institute for Occupational Safety and Health (NIOSH), Centers for Disease Control and Prevention. Venomous spiders. www.cdc.gov/niosh/topics/spiders/. Accessed November 25, 2020.
28. Starr S. What you need to know to prevent a poisonous spider bite. AAP News. 2013;34:42. www.aappublications.org/content/aapnews/34/9/42.5.full.pdf. Accessed November 25, 2020.
29. Spider bites. Mayo Clinic Web site. www.mayoclinic.org/diseases-conditions/spider-bites/symptoms-causes/syc-20352371. Accessed November 25, 2020.
30. Barish RA, Arnold T. Spider bites. In: Merck Manual (Professional Version). Merck Sharp & Dohme Corp.; 2016. www.merckmanuals.com/professional/injuries-poisoning/bites-and-stings/spider-bites. Accessed November 25, 2020.
31. Juckett G. Arthropod bites. Am Fam Physician. 2013;88:841-847.
32. Clark RF, Wethern-Kestner S, Vance MV, et al. Clinical presentation and treatment of black widow spider envenomation: a review of 163 cases. Ann Emerg Med. 1992;21:782-787.
33. Koehler PG, Pereira RM, Diclaro JW II. Fleas. Publication ENY-025. University of Florida IFAS Extension. Revised January 2012. https://edis.ifas.ufl.edu/ig087. Accessed November 25, 2020.
34. Bitam I, Dittmar K, Parola P, et al. Fleas and flea-borne diseases. Int J Infect Dis. 2010;14:e667-e676.
35. Leulmi H, Socolovschi C, Laudisoit A, et al. Detection of Rickettsia felis, Rickettsia typhi, Bartonella species and Yersinia pestis in fleas (Siphonaptera) from Africa. PLoS Negl Trop Dis. 2014;8:e3152.
36. Naimer SA, Cohen AD, Mumcuoglu KY, et al. Household papular urticaria. Isr Med Assoc J. 2002;4(11 suppl):911-913.
37. Golomb MR, Golomb HS. What’s eating you? Cat flea (Ctenocephalides felis). Cutis. 2010;85:10-11.
38. Dryden MW. Flea and tick control in the 21st century: challenges and opportunities. Vet Dermatol. 2009;20:435-440.
39. Dryden MW. Fleas in dogs and cats. Merck Sharp & Dohme Corporation: Merck Manual Veterinary Manual. Updated December 2014. www.merckvetmanual.com/integumentary-system/fleas-and-flea-allergy-dermatitis/fleas-in-dogs-and-cats. Accessed November 25, 2020.
40. Centers for Disease Control and Prevention. Getting rid of fleas. www.cdc.gov/fleas/getting_rid.html. Accessed November 25, 2020.
41. Chattopadhyay P, Goyary D, Dhiman S, et al. Immunomodulating effects and hypersensitivity reactions caused by Northeast Indian black fly salivary gland extract. J Immunotoxicol. 2014;11:126-132.
42. Hrabak TM, Dice JP. Use of immunotherapy in the management of presumed anaphylaxis to the deer fly. Ann Allergy Asthma Immunol. 2003;90:351-354.
43. Royden A, Wedley A, Merga JY, et al. A role for flies (Diptera) in the transmission of Campylobacter to broilers? Epidemiol Infect. 2016;144:3326-3334.
44. Fradin MS, Day JF. Comparative efficacy of insect repellents against mosquito bites. N Engl J Med. 2002;347:13-18.

45. Carpenter S, Groschup MH, Garros C, et al. Culicoides biting midges, arboviruses and public health in Europe. Antiviral Res. 2013;100:102-113.
46. Peng Z, Yang M, Simons FE. Immunologic mechanisms in mosquito allergy: correlation of skin reactions with specific IgE and IgG anti-bodies and lymphocyte proliferation response to mosquito antigens. Ann Allergy Asthma Immunol. 1996;77:238-244.
47. Simons FE, Peng Z. Skeeter syndrome. J Allergy Clin Immunol. 1999;104:705-707.
48. Centers for Disease Control and Prevention. Travelers’ health. Clinician resources. wwwnc.cdc.gov/travel/page/clinician-information-center. Accessed November 25, 2020.
49. Gauci M, Loh RK, Stone BF, et al. Allergic reactions to the Australian paralysis tick, Ixodes holocyclus: diagnostic evaluation by skin test and radioimmunoassay. Clin Exp Allergy. 1989;19:279-283.
50. Centers for Disease Control and Prevention. Ticks. Removing a tick. www.cdc.gov/ticks/removing_a_tick.html. Accessed November 25, 2020.
51. Golden DB, Kagey-Sobotka A, Norman PS, et al. Insect sting allergy with negative venom skin test responses. J Allergy Clin Immunol. 2001;107:897-901.
52. Arzt L, Bokanovic D, Schrautzer C, et al. Immunological differences between insect venom-allergic patients with and without immunotherapy and asymptomatically sensitized subjects. Allergy. 2018;73:1223-1231.
53. Heddle R, Golden DBK. Allergy to insect stings and bites. World Allergy Organization Web site. Updated August 2015. www.worldallergy.org/education-and-programs/education/allergic-disease-resource-center/professionals/allergy-to-insect-stings-and-bites. Accessed November 25, 2020.
54. RuëffF, Przybilla B, Müller U, et al. The sting challenge test in Hymenoptera venom allergy. Position paper of the Subcommittee on Insect Venom Allergy of the European Academy of Allergology and Clinical Immunology. Allergy. 1996;51:216-225.
55. Management of simple insect bites: where’s the evidence? Drug Ther Bull. 2012;50:45-48.
56. Tracy JM. Insect allergy. Mt Sinai J Med. 2011;78:773-783.
57. Golden DBK. Insect sting allergy and venom immunotherapy: a model and a mystery. J Allergy Clin Immunol. 2005;115:439-447.
58. Winther L, Arnved J, Malling H-J, et al. Side-effects of allergen-specific immunotherapy: a prospective multi-centre study. Clin Exp Allergy. 2006;36:254-260.
59. Mellerup MT, Hahn GW, Poulsen LK, et al. Safety of allergen-specific immunotherapy. Relation between dosage regimen, allergen extract, disease and systemic side-effects during induction treatment. Clin Exp Allergy. 2000;30:1423-1429.
60. Anaphylaxis and insect stings and bites. Med Lett Drugs Ther. 2017;59:e79-e82.
61. Sampson HA, Muñoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: summary report—second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. Ann Emerg Med. 2006;47:373-380.
62. Pflipsen MC, Vega Colon KM. Anaphylaxis: recognition and management. Am Fam Physician. 2020;102:355-362. Accessed November 25, 2020.
Insect, arachnid, and other arthropod bites and stings are common patient complaints in a primary care office. A thorough history and physical exam can often isolate the specific offender and guide management. In this article, we outline how to identify, diagnose, and treat common bites and stings from bees and wasps; centipedes and spiders; fleas; flies and biting midges; mosquitoes; and ticks, and discuss how high-risk patients should be triaged and referred for additional testing and treatment, such as venom immunotherapy (VIT).
Insects and arachnids:Background and epidemiology
Insects are arthropods with 3-part exoskeletons: head, thorax, and abdomen. They have 6 jointed legs, compound eyes, and antennae. There are approximately 91,000 insect species in the United States, the most abundant orders being Coleoptera (beetles), Diptera (flies), and Hymenoptera (includes ants, bees, wasps, and sawflies).1
The reported incidence of insect bites and stings varies widely because most people experience mild symptoms and therefore do not seek medical care. Best statistics are for Hymenoptera stings, which are more likely to cause a severe reaction. In Europe, 56% to 94% of the general population has reported being bitten or stung by one of the Hymenoptera species.2 In many areas of Australia, the incidence of jack jumper ant stings is only 2% to 3%3; in the United States, 55% of people report being stung by nonnative fire ants within 3 weeks of moving into an endemic area.4
Arachnids are some of the earliest terrestrial organisms, of the class Arachnida, which includes scorpions, ticks, spiders, mites, and daddy longlegs (harvestmen).5 Arachnids are wingless and characterized by segmented bodies, jointed appendages, and exoskeletons.6,7 In most, the body is separated into 2 segments (the cephalothorax and abdomen), except for mites, ticks, and daddy longlegs, in which the entire body comprises a single segment.5
Arthropod bites are common in the United States; almost one-half are caused by spiders.7 Brown recluse (Loxosceles spp) and black widow (Latrodectus spp) spider bites are the most concerning: Although usually mild, these bites can be life-threatening but are rarely fatal. In 2013, almost 3500 bites by black widow and brown recluse spiders were reported.8
Risk factors
Risk factors for insect, arachnid, and other arthropod bites and stings are primarily environmental. People who live or work in proximity of biting or stinging insects (eg, gardeners and beekeepers) are more likely to be affected; so are those who work with animals or live next to standing water or grassy or wooded locales.
Continue to: There are also risk factors...
There are also risk factors for a systemic sting reaction:
- A sting reaction < 2 months earlier increases the risk of a subsequent systemic sting reaction by ≥ 50%.9
- Among beekeepers, paradoxically, the risk of a systemic reaction is higher in those stung < 15 times a year than in those stung > 200 times.10
- Patients with an elevated baseline serum level of tryptase (reference range, < 11.4 ng/mL), which is part of the allergenic response, or with biopsy-proven systemic mastocytosis are at increased risk of a systemic sting reaction.11
Presentation: Signs and symptomsvary with severity
Insect bites and stings usually cause transient local inflammation and, occasionally, a toxic reaction. Allergic hypersensitivity can result in a large local reaction or a generalized systemic reaction12:
- A small local reaction is transient and mild, develops directly at the site of the sting, and can last several days.13
- A large (or significant) local reaction, defined as swelling > 10 cm in diameter (FIGURE 1) and lasting > 24 hours, occurs in 2% to 26% of people who have been bitten or stung.14 This is an immunoglobulin (Ig) E–mediated late-phase reaction that can be accompanied by fatigue and nausea.12,13,15 For a patient with a large local reaction, the risk of a concomitant systemic reaction is 4% to 10%, typically beginning within 30 minutes after envenomation or, possibly, delayed for several hours or marked by a biphasic interval.16
- Characteristics of a systemic reaction are urticaria, angioedema, bronchospasm, large-airway edema, hypotension, and other clinical manifestations of anaphylaxis.17 In the United States, a systemic sting reaction is reported to occur in approximately 3% of bite and sting victims. Mortality among the general population from a systemic bite or sting reaction is 0.16 for every 100,000 people,2 and at least 40 to 100 die every year in the United States from anaphylaxis resulting from an insect bite or sting.18
- The most severe anaphylactic reactions involve the cardiovascular and respiratory systems, commonly including hypotension and symptoms of upper- or lower-airway obstruction. Laryngeal edema and circulatory failure are the most common mechanisms of anaphylactic death.19

Bees and wasps
Hymenoptera stinging insects include the family Apidae (honey bee, bumblebee, and sweat bee) and Vespidae (yellow jacket, yellow- and white-faced hornets, and paper wasp). A worker honey bee can sting only once, leaving its barbed stinger in the skin; a wasp, hornet, and yellow jacket can sting multiple times (FIGURE 2).2

Continue to: Bee and wasp sting...
Bee and wasp sting allergies are the most common insect venom allergic reactions. A bee sting is more likely to lead to a severe allergic reaction than a wasp sting. Allergic reactions to hornet and bumblebee stings are less common but can occur in patients already sensitized to wasp and honey bee stings.20,21
Management. Remove honey bee stingers by scraping the skin with a fingernail or credit card. Ideally, the stinger should be removed in the first 30 seconds, before the venom sac empties. Otherwise, intense local inflammation, with possible lymphangitic streaking, can result.22
For guidance on localized symptomatic care of bee and wasp stings and bites and stings from other sources discussed in this article, see “Providing relief and advanced care” on page E6.
Centipedes and spiders
Centipedes are arthropods of the class Chilopoda, subphylum Myriapoda, that are characterized by repeating linear (metameric) segments, each containing 1 pair of legs.23 Centipedes have a pair of poison claws behind the head that are used to paralyze prey—usually, small insects.23,24 The bite of a larger centipede can cause a painful reaction that generally subsides after a few hours but can last several days. Centipede bites are usually nonfatal to humans.23
Spiders belong to the class Arachnida, order Araneae. They have 8 legs with chelicerae (mouthpiece, or “jaws”) that inject venom into prey.25 Most spiders found in the United States cannot bite through human skin.26,27 Common exceptions are black widow and brown recluse spiders, which each produce a distinct toxic venom that can cause significant morbidity in humans through a bite, although bites are rarely fatal.26,27
The brown recluse spider is described as having a violin-shaped marking on the abdomen; the body is yellowish, tan, or dark brown. A bite can produce tiny fang marks and cause dull pain at the site of the bite that spreads quickly; myalgia; and pain in the stomach, back, chest, and legs.28,29 The bite takes approximately 7 days to resolve. In a minority of cases, a tender erythematous halo develops, followed by a severe necrotic ulcer, or loxoscelism (FIGURE 3; 40% of cases) or scarring (13%), or both.29,30

Continue to: In contrast...
In contrast, the body of a black widow spider is black; females exhibit a distinctive red or yellow hourglass marking on their ventral aspect.28,31 The pinprick sensation of a bite leads to symptoms that can include erythema, swelling, pain, stiffness, chills, fever, nausea, and stomach pain.30,32
Management. Again, see “Providing relief and advanced care” on page E6. Consider providing antivenin treatment for moderate or severe bites of brown recluse and black widow spiders.
Fleas
Fleas are members of the order Siphonaptera. They are small (1.5-3.2 mm long), reddish brown, wingless, blood-sucking insects with long legs that allow them to jump far (12 or 13 inches) and high (6 or 7 inches).33 Domesticated cats and dogs are the source of most flea infestations, resulting in an increased risk of exposure for humans.34,35 Flea bites, which generally occur on lower extremities, develop into a small, erythematous papule with a halo (FIGURE 4) and associated mild edema, and cause intense pruritus 30 minutes after the bite.35-37

Fleas are a vector for severe microbial infections, including bartonellosis, bubonic plague, cat-flea typhus, murine typhus, cat-scratch disease, rickettsial disease, and tularemia. Tungiasis is an inflammatory burrowing flea infestation—not a secondary infection for which the flea is a vector.34,35
Preventive management. Repellents, including products that contain DEET (N,N-diethyl-meta-toluamide), picaridin (2-[2-hydroxyethyl]-1-piperidinecarboxylic acid 1-methylpropyl ester), and PMD (p-menthane-3,8-diol, a chemical constituent of Eucalyptus citriodora oil) can be used to prevent flea bites in humans.33,38 Studies show that the scent of other botanic oils, including lavender, cedarwood, and peppermint, can also help prevent infestation by fleas; however, these compounds are not as effective as traditional insect repellents.33,38
Flea control is difficult, requiring a multimodal approach to treating the infested animal and its environment.39 Treatment of the infested domestic animal is the primary method of preventing human bites. Nonpesticidal control involves frequent cleaning of carpeting, furniture, animal bedding, and kennels. Insecticides can be applied throughout the house to combat severe infestation.33,38
Continue to: The Centers for Disease Control and Prevention...
The Centers for Disease Control and Prevention provide a general introduction to getting rid of fleas for pet owners.40 For specific guidance on flea-eradication strategies and specific flea-control products, advise patients to seek the advice of their veterinarian.
Flies and biting midges
Flies are 2-winged insects belonging to the order Diptera. Several fly species can bite, causing a local inflammatory reaction; these include black flies, deer flies, horse flies, and sand flies. Signs and symptoms of a fly bite include pain, pruritus, erythema, and mild swelling (FIGURE 5).41,42 Flies can transmit several infections, including bartonellosis, enteric bacterial disease (eg, caused by Campylobacter spp), leishmaniasis, loiasis, onchocerciasis, and trypanosomiasis.43

Biting midges, also called “no-see-ums,” biting gnats, moose flies, and “punkies,”44 are tiny (1-3 mm long) blood-sucking flies.45 Bitten patients often report not having seen the midge because it is so small. The bite typically starts as a small, erythematous papule that develops into a dome-shaped blister and can be extraordinarily pruritic and painful.44 The majority of people who have been bitten develop a hypersensitivity reaction, which usually resolves in a few weeks.
Management. Suppressing adult biting midges with an environmental insecticide is typically insufficient because the insecticide must be sprayed daily to eradicate active midges and generally does not affect larval habitat. Insect repellents and biopesticides, such as oil of lemon eucalyptus, can be effective in reducing the risk of bites.44,45
Mosquitoes
Mosquitoes are flying, blood-sucking insects of the order Diptera and family Culicidae. Anopheles, Culex, and Aedes genera are responsible for most bites of humans.
The bite of a mosquito produces an indurated, limited local reaction characterized by a pruritic wheal (3-29 mm in diameter) with surrounding erythema (FIGURE 6) that peaks in approximately 30 minutes, although patients might have a delayed reaction hours later.46 Immunocompromised patients might experience a more significant local inflammatory reaction that is accompanied by low-grade fever, hives, or swollen lymph nodes.46,47

Mosquitoes are a vector for serious infections, including dengue, Japanese encephalitis, malaria, and yellow fever, and disease caused by Chikungunya, West Nile, and Zika viruses.
Continue to: Management
Management. Advise patients to reduce their risk by using insect repellent, sleeping under mosquito netting, and wearing a long-sleeve shirt and long pants when traveling to endemic areas or when a local outbreak occurs.48
Ticks
Ticks belong to the order Parasitiformes and families Ixodidae and Argasidae. Hard ticks are found in brushy fields and tall grasses and can bite and feed on humans for days. Soft ticks are generally found around animal nests.29 Tick bites can cause a local reaction that includes painful, erythematous, inflammatory papular lesions (FIGURE 7).49

Ticks can transmit several infectious diseases. Depending on the microbial pathogen and the genus and species of tick, it takes 2 to 96 hours for the tick to attach to skin and transmit the pathogen to the human host. The TABLE29,49,50 provides an overview of tick species in the United States, diseases that they can transmit, and the geographic distribution of those diseases.

Management. Ticks should be removed with fine-tipped tweezers. Grasp the body of the tick close to the skin and pull upward while applying steady, even pressure. After removing the tick, clean the bite and the surrounding area with alcohol or with soap and water. Dispose of a live tick by flushing it down the toilet; or, kill it in alcohol and either seal it in a bag with tape or place it in a container.50
Diagnosis and the utilityof special testing
The diagnosis of insect, arachnid, and other arthropod bites and stings depends on the history, including obtaining a record of possible exposure and a travel history; the timing of the bite or sting; and associated signs and symptoms.18,51
Venom skin testing. For Hymenoptera stings, intradermal tests using a venom concentration of 0.001 to 1 μg/mL are positive in 65% to 80% of patients with a history of a systemic insect-sting allergic reaction. A negative venom skin test can occur during the 3-to-6-week refractory period after a sting reaction or many years later, which represents a loss of sensitivity. Positive venom skin tests are used to confirm allergy and identify specific insects to which the patient is allergic.11,12
Continue to: Allergen-specific IgE antibody testing.
Allergen-specific IgE antibody testing. These serum assays—typically, radioallergosorbent testing (RAST)—are less sensitive than venom skin tests. RAST is useful when venom skin testing cannot be performed or when skin testing is negative in a patient who has had a severe allergic reaction to an insect bite or sting. Serum IgE-specific antibody testing is preferred over venom skin testing in patients who are at high risk of anaphylaxis.52,53
Providing reliefand advanced care
Symptomatic treatment of mild bites and stings includes washing the affected area with soap and water and applying a cold compress to reduce swelling.54 For painful lesions, an oral analgesic can be prescribed.
For mild or moderate pruritus, a low- to midpotency topical corticosteroid (eg, hydrocortisone valerate cream 0.2% bid), topical calamine, or pramoxine can be applied,or a nonsedating oral antihistamine, such as loratadine (10 mg/d) or cetirizine (10 mg/d), can be used.14,55 For severe itching, a sedating antihistamine, such as hydroxyzine (10-25 mg every 4 to 6 hours prn), might help relieve symptoms; H1- and H2-receptor antagonists can be used concomitantly.54,55
Significant local symptoms. Large local reactions are treated with a midpotency topical corticosteroid (eg, triamcinolone acetonide cream 0.1% bid) plus an oral antihistamine to relieve pruritus and reduce allergic inflammation. For a more severe reaction, an oral corticosteroid (prednisone 1 mg/kg; maximum dosage, 50 mg/d) can be given for 5 to 7 days.54-56
Management of a necrotic ulcer secondary to a brown recluse spider bite is symptomatic and supportive. The size of these wounds can increase for as long as 10 days after the bite; resolution can require months of wound care, possibly with debridement. Rarely, skin grafting is required.27,28,31
VIT. Some studies show that VIT can improve quality of life in patients with prolonged, frequent, and worsening reactions to insect bites or stings and repeated, unavoidable exposures.55,56 VIT is recommended for patients with systemic hypersensitivity and a positive venom skin test result. It is approximately 95% effective in preventing or reducing severe systemic reactions and reduces the risk of anaphylaxis (see next section) and death.57 The maintenance dosage of VIT is usually 100 μg every 4 to 6 weeks; optimal duration of treatment is 3 to 5 years.58
Continue to: After VIT is complete...
After VIT is complete, counsel patients that a mild systemic reaction is still possible after an insect bite or sting. More prolonged, even lifetime, treatment should be considered for patients who have58,59
- a history of severe, life-threatening allergic reactions to bites and stings
- honey bee sting allergy
- mast-cell disease
- a history of anaphylaxis while receiving VIT.
Absolute contraindications to VIT include a history of serious immune disease, chronic infection, or cancer.58,59
Managing anaphylaxis
This severe allergic reaction can lead to death if untreated. First-line therapy is intramuscular epinephrine, 0.01 mg/kg (maximum single dose, 0.5 mg) given every 5 to 15 minutes.14,60 Epinephrine auto-injectors deliver a fixed dose and are labeled according to weight. Administration of O2 and intravenous fluids is recommended for hemodynamically unstable patients.60,61 Antihistamines and corticosteroids can be used as secondary treatment but should not replace epinephrine.56
After preliminary improvement, patients might decompensate when the epinephrine dose wears off. Furthermore, a biphasic reaction, variously reported in < 5% to as many as 20% of patients,61,62 occurs hours after the initial anaphylactic reaction. Patients should be monitored, therefore, for at least 6 to 8 hours after an anaphylactic reaction, preferably in a facility equipped to treat anaphylaxis.17,56
Before discharge, patients who have had an anaphylactic reaction should be given a prescription for epinephrine and training in the use of an epinephrine auto-injector. Allergen avoidance, along with an emergency plan in the event of a bite or sting, is recommended. Follow-up evaluation with an allergist or immunologist is essential for proper diagnosis and to determine whether the patient is a candidate for VIT.14,17
CORRESPONDENCE
Ecler Ercole Jaqua, MD, DipABLM, FAAFP, 1200 California Street, Suite 240, Redlands, CA 92374; ejaqua@llu.edu.
Insect, arachnid, and other arthropod bites and stings are common patient complaints in a primary care office. A thorough history and physical exam can often isolate the specific offender and guide management. In this article, we outline how to identify, diagnose, and treat common bites and stings from bees and wasps; centipedes and spiders; fleas; flies and biting midges; mosquitoes; and ticks, and discuss how high-risk patients should be triaged and referred for additional testing and treatment, such as venom immunotherapy (VIT).
Insects and arachnids:Background and epidemiology
Insects are arthropods with 3-part exoskeletons: head, thorax, and abdomen. They have 6 jointed legs, compound eyes, and antennae. There are approximately 91,000 insect species in the United States, the most abundant orders being Coleoptera (beetles), Diptera (flies), and Hymenoptera (includes ants, bees, wasps, and sawflies).1
The reported incidence of insect bites and stings varies widely because most people experience mild symptoms and therefore do not seek medical care. Best statistics are for Hymenoptera stings, which are more likely to cause a severe reaction. In Europe, 56% to 94% of the general population has reported being bitten or stung by one of the Hymenoptera species.2 In many areas of Australia, the incidence of jack jumper ant stings is only 2% to 3%3; in the United States, 55% of people report being stung by nonnative fire ants within 3 weeks of moving into an endemic area.4
Arachnids are some of the earliest terrestrial organisms, of the class Arachnida, which includes scorpions, ticks, spiders, mites, and daddy longlegs (harvestmen).5 Arachnids are wingless and characterized by segmented bodies, jointed appendages, and exoskeletons.6,7 In most, the body is separated into 2 segments (the cephalothorax and abdomen), except for mites, ticks, and daddy longlegs, in which the entire body comprises a single segment.5
Arthropod bites are common in the United States; almost one-half are caused by spiders.7 Brown recluse (Loxosceles spp) and black widow (Latrodectus spp) spider bites are the most concerning: Although usually mild, these bites can be life-threatening but are rarely fatal. In 2013, almost 3500 bites by black widow and brown recluse spiders were reported.8
Risk factors
Risk factors for insect, arachnid, and other arthropod bites and stings are primarily environmental. People who live or work in proximity of biting or stinging insects (eg, gardeners and beekeepers) are more likely to be affected; so are those who work with animals or live next to standing water or grassy or wooded locales.
Continue to: There are also risk factors...
There are also risk factors for a systemic sting reaction:
- A sting reaction < 2 months earlier increases the risk of a subsequent systemic sting reaction by ≥ 50%.9
- Among beekeepers, paradoxically, the risk of a systemic reaction is higher in those stung < 15 times a year than in those stung > 200 times.10
- Patients with an elevated baseline serum level of tryptase (reference range, < 11.4 ng/mL), which is part of the allergenic response, or with biopsy-proven systemic mastocytosis are at increased risk of a systemic sting reaction.11
Presentation: Signs and symptomsvary with severity
Insect bites and stings usually cause transient local inflammation and, occasionally, a toxic reaction. Allergic hypersensitivity can result in a large local reaction or a generalized systemic reaction12:
- A small local reaction is transient and mild, develops directly at the site of the sting, and can last several days.13
- A large (or significant) local reaction, defined as swelling > 10 cm in diameter (FIGURE 1) and lasting > 24 hours, occurs in 2% to 26% of people who have been bitten or stung.14 This is an immunoglobulin (Ig) E–mediated late-phase reaction that can be accompanied by fatigue and nausea.12,13,15 For a patient with a large local reaction, the risk of a concomitant systemic reaction is 4% to 10%, typically beginning within 30 minutes after envenomation or, possibly, delayed for several hours or marked by a biphasic interval.16
- Characteristics of a systemic reaction are urticaria, angioedema, bronchospasm, large-airway edema, hypotension, and other clinical manifestations of anaphylaxis.17 In the United States, a systemic sting reaction is reported to occur in approximately 3% of bite and sting victims. Mortality among the general population from a systemic bite or sting reaction is 0.16 for every 100,000 people,2 and at least 40 to 100 die every year in the United States from anaphylaxis resulting from an insect bite or sting.18
- The most severe anaphylactic reactions involve the cardiovascular and respiratory systems, commonly including hypotension and symptoms of upper- or lower-airway obstruction. Laryngeal edema and circulatory failure are the most common mechanisms of anaphylactic death.19

Bees and wasps
Hymenoptera stinging insects include the family Apidae (honey bee, bumblebee, and sweat bee) and Vespidae (yellow jacket, yellow- and white-faced hornets, and paper wasp). A worker honey bee can sting only once, leaving its barbed stinger in the skin; a wasp, hornet, and yellow jacket can sting multiple times (FIGURE 2).2

Continue to: Bee and wasp sting...
Bee and wasp sting allergies are the most common insect venom allergic reactions. A bee sting is more likely to lead to a severe allergic reaction than a wasp sting. Allergic reactions to hornet and bumblebee stings are less common but can occur in patients already sensitized to wasp and honey bee stings.20,21
Management. Remove honey bee stingers by scraping the skin with a fingernail or credit card. Ideally, the stinger should be removed in the first 30 seconds, before the venom sac empties. Otherwise, intense local inflammation, with possible lymphangitic streaking, can result.22
For guidance on localized symptomatic care of bee and wasp stings and bites and stings from other sources discussed in this article, see “Providing relief and advanced care” on page E6.
Centipedes and spiders
Centipedes are arthropods of the class Chilopoda, subphylum Myriapoda, that are characterized by repeating linear (metameric) segments, each containing 1 pair of legs.23 Centipedes have a pair of poison claws behind the head that are used to paralyze prey—usually, small insects.23,24 The bite of a larger centipede can cause a painful reaction that generally subsides after a few hours but can last several days. Centipede bites are usually nonfatal to humans.23
Spiders belong to the class Arachnida, order Araneae. They have 8 legs with chelicerae (mouthpiece, or “jaws”) that inject venom into prey.25 Most spiders found in the United States cannot bite through human skin.26,27 Common exceptions are black widow and brown recluse spiders, which each produce a distinct toxic venom that can cause significant morbidity in humans through a bite, although bites are rarely fatal.26,27
The brown recluse spider is described as having a violin-shaped marking on the abdomen; the body is yellowish, tan, or dark brown. A bite can produce tiny fang marks and cause dull pain at the site of the bite that spreads quickly; myalgia; and pain in the stomach, back, chest, and legs.28,29 The bite takes approximately 7 days to resolve. In a minority of cases, a tender erythematous halo develops, followed by a severe necrotic ulcer, or loxoscelism (FIGURE 3; 40% of cases) or scarring (13%), or both.29,30

Continue to: In contrast...
In contrast, the body of a black widow spider is black; females exhibit a distinctive red or yellow hourglass marking on their ventral aspect.28,31 The pinprick sensation of a bite leads to symptoms that can include erythema, swelling, pain, stiffness, chills, fever, nausea, and stomach pain.30,32
Management. Again, see “Providing relief and advanced care” on page E6. Consider providing antivenin treatment for moderate or severe bites of brown recluse and black widow spiders.
Fleas
Fleas are members of the order Siphonaptera. They are small (1.5-3.2 mm long), reddish brown, wingless, blood-sucking insects with long legs that allow them to jump far (12 or 13 inches) and high (6 or 7 inches).33 Domesticated cats and dogs are the source of most flea infestations, resulting in an increased risk of exposure for humans.34,35 Flea bites, which generally occur on lower extremities, develop into a small, erythematous papule with a halo (FIGURE 4) and associated mild edema, and cause intense pruritus 30 minutes after the bite.35-37

Fleas are a vector for severe microbial infections, including bartonellosis, bubonic plague, cat-flea typhus, murine typhus, cat-scratch disease, rickettsial disease, and tularemia. Tungiasis is an inflammatory burrowing flea infestation—not a secondary infection for which the flea is a vector.34,35
Preventive management. Repellents, including products that contain DEET (N,N-diethyl-meta-toluamide), picaridin (2-[2-hydroxyethyl]-1-piperidinecarboxylic acid 1-methylpropyl ester), and PMD (p-menthane-3,8-diol, a chemical constituent of Eucalyptus citriodora oil) can be used to prevent flea bites in humans.33,38 Studies show that the scent of other botanic oils, including lavender, cedarwood, and peppermint, can also help prevent infestation by fleas; however, these compounds are not as effective as traditional insect repellents.33,38
Flea control is difficult, requiring a multimodal approach to treating the infested animal and its environment.39 Treatment of the infested domestic animal is the primary method of preventing human bites. Nonpesticidal control involves frequent cleaning of carpeting, furniture, animal bedding, and kennels. Insecticides can be applied throughout the house to combat severe infestation.33,38
Continue to: The Centers for Disease Control and Prevention...
The Centers for Disease Control and Prevention provide a general introduction to getting rid of fleas for pet owners.40 For specific guidance on flea-eradication strategies and specific flea-control products, advise patients to seek the advice of their veterinarian.
Flies and biting midges
Flies are 2-winged insects belonging to the order Diptera. Several fly species can bite, causing a local inflammatory reaction; these include black flies, deer flies, horse flies, and sand flies. Signs and symptoms of a fly bite include pain, pruritus, erythema, and mild swelling (FIGURE 5).41,42 Flies can transmit several infections, including bartonellosis, enteric bacterial disease (eg, caused by Campylobacter spp), leishmaniasis, loiasis, onchocerciasis, and trypanosomiasis.43

Biting midges, also called “no-see-ums,” biting gnats, moose flies, and “punkies,”44 are tiny (1-3 mm long) blood-sucking flies.45 Bitten patients often report not having seen the midge because it is so small. The bite typically starts as a small, erythematous papule that develops into a dome-shaped blister and can be extraordinarily pruritic and painful.44 The majority of people who have been bitten develop a hypersensitivity reaction, which usually resolves in a few weeks.
Management. Suppressing adult biting midges with an environmental insecticide is typically insufficient because the insecticide must be sprayed daily to eradicate active midges and generally does not affect larval habitat. Insect repellents and biopesticides, such as oil of lemon eucalyptus, can be effective in reducing the risk of bites.44,45
Mosquitoes
Mosquitoes are flying, blood-sucking insects of the order Diptera and family Culicidae. Anopheles, Culex, and Aedes genera are responsible for most bites of humans.
The bite of a mosquito produces an indurated, limited local reaction characterized by a pruritic wheal (3-29 mm in diameter) with surrounding erythema (FIGURE 6) that peaks in approximately 30 minutes, although patients might have a delayed reaction hours later.46 Immunocompromised patients might experience a more significant local inflammatory reaction that is accompanied by low-grade fever, hives, or swollen lymph nodes.46,47

Mosquitoes are a vector for serious infections, including dengue, Japanese encephalitis, malaria, and yellow fever, and disease caused by Chikungunya, West Nile, and Zika viruses.
Continue to: Management
Management. Advise patients to reduce their risk by using insect repellent, sleeping under mosquito netting, and wearing a long-sleeve shirt and long pants when traveling to endemic areas or when a local outbreak occurs.48
Ticks
Ticks belong to the order Parasitiformes and families Ixodidae and Argasidae. Hard ticks are found in brushy fields and tall grasses and can bite and feed on humans for days. Soft ticks are generally found around animal nests.29 Tick bites can cause a local reaction that includes painful, erythematous, inflammatory papular lesions (FIGURE 7).49

Ticks can transmit several infectious diseases. Depending on the microbial pathogen and the genus and species of tick, it takes 2 to 96 hours for the tick to attach to skin and transmit the pathogen to the human host. The TABLE29,49,50 provides an overview of tick species in the United States, diseases that they can transmit, and the geographic distribution of those diseases.

Management. Ticks should be removed with fine-tipped tweezers. Grasp the body of the tick close to the skin and pull upward while applying steady, even pressure. After removing the tick, clean the bite and the surrounding area with alcohol or with soap and water. Dispose of a live tick by flushing it down the toilet; or, kill it in alcohol and either seal it in a bag with tape or place it in a container.50
Diagnosis and the utilityof special testing
The diagnosis of insect, arachnid, and other arthropod bites and stings depends on the history, including obtaining a record of possible exposure and a travel history; the timing of the bite or sting; and associated signs and symptoms.18,51
Venom skin testing. For Hymenoptera stings, intradermal tests using a venom concentration of 0.001 to 1 μg/mL are positive in 65% to 80% of patients with a history of a systemic insect-sting allergic reaction. A negative venom skin test can occur during the 3-to-6-week refractory period after a sting reaction or many years later, which represents a loss of sensitivity. Positive venom skin tests are used to confirm allergy and identify specific insects to which the patient is allergic.11,12
Continue to: Allergen-specific IgE antibody testing.
Allergen-specific IgE antibody testing. These serum assays—typically, radioallergosorbent testing (RAST)—are less sensitive than venom skin tests. RAST is useful when venom skin testing cannot be performed or when skin testing is negative in a patient who has had a severe allergic reaction to an insect bite or sting. Serum IgE-specific antibody testing is preferred over venom skin testing in patients who are at high risk of anaphylaxis.52,53
Providing reliefand advanced care
Symptomatic treatment of mild bites and stings includes washing the affected area with soap and water and applying a cold compress to reduce swelling.54 For painful lesions, an oral analgesic can be prescribed.
For mild or moderate pruritus, a low- to midpotency topical corticosteroid (eg, hydrocortisone valerate cream 0.2% bid), topical calamine, or pramoxine can be applied,or a nonsedating oral antihistamine, such as loratadine (10 mg/d) or cetirizine (10 mg/d), can be used.14,55 For severe itching, a sedating antihistamine, such as hydroxyzine (10-25 mg every 4 to 6 hours prn), might help relieve symptoms; H1- and H2-receptor antagonists can be used concomitantly.54,55
Significant local symptoms. Large local reactions are treated with a midpotency topical corticosteroid (eg, triamcinolone acetonide cream 0.1% bid) plus an oral antihistamine to relieve pruritus and reduce allergic inflammation. For a more severe reaction, an oral corticosteroid (prednisone 1 mg/kg; maximum dosage, 50 mg/d) can be given for 5 to 7 days.54-56
Management of a necrotic ulcer secondary to a brown recluse spider bite is symptomatic and supportive. The size of these wounds can increase for as long as 10 days after the bite; resolution can require months of wound care, possibly with debridement. Rarely, skin grafting is required.27,28,31
VIT. Some studies show that VIT can improve quality of life in patients with prolonged, frequent, and worsening reactions to insect bites or stings and repeated, unavoidable exposures.55,56 VIT is recommended for patients with systemic hypersensitivity and a positive venom skin test result. It is approximately 95% effective in preventing or reducing severe systemic reactions and reduces the risk of anaphylaxis (see next section) and death.57 The maintenance dosage of VIT is usually 100 μg every 4 to 6 weeks; optimal duration of treatment is 3 to 5 years.58
Continue to: After VIT is complete...
After VIT is complete, counsel patients that a mild systemic reaction is still possible after an insect bite or sting. More prolonged, even lifetime, treatment should be considered for patients who have58,59
- a history of severe, life-threatening allergic reactions to bites and stings
- honey bee sting allergy
- mast-cell disease
- a history of anaphylaxis while receiving VIT.
Absolute contraindications to VIT include a history of serious immune disease, chronic infection, or cancer.58,59
Managing anaphylaxis
This severe allergic reaction can lead to death if untreated. First-line therapy is intramuscular epinephrine, 0.01 mg/kg (maximum single dose, 0.5 mg) given every 5 to 15 minutes.14,60 Epinephrine auto-injectors deliver a fixed dose and are labeled according to weight. Administration of O2 and intravenous fluids is recommended for hemodynamically unstable patients.60,61 Antihistamines and corticosteroids can be used as secondary treatment but should not replace epinephrine.56
After preliminary improvement, patients might decompensate when the epinephrine dose wears off. Furthermore, a biphasic reaction, variously reported in < 5% to as many as 20% of patients,61,62 occurs hours after the initial anaphylactic reaction. Patients should be monitored, therefore, for at least 6 to 8 hours after an anaphylactic reaction, preferably in a facility equipped to treat anaphylaxis.17,56
Before discharge, patients who have had an anaphylactic reaction should be given a prescription for epinephrine and training in the use of an epinephrine auto-injector. Allergen avoidance, along with an emergency plan in the event of a bite or sting, is recommended. Follow-up evaluation with an allergist or immunologist is essential for proper diagnosis and to determine whether the patient is a candidate for VIT.14,17
CORRESPONDENCE
Ecler Ercole Jaqua, MD, DipABLM, FAAFP, 1200 California Street, Suite 240, Redlands, CA 92374; ejaqua@llu.edu.
1. Numbers of insects (species and individuals). Smithsonian BugInfo Web site. www.si.edu/spotlight/buginfo/bugnos. Accessed November 25, 2020.
2. Antonicelli L, Bilò MB, Bonifazi F. Epidemiology of Hymenoptera allergy. Curr Opin Allergy Clin Immunol. 2002;2:341-346.
3. Jack jumper ant allergy. Australasian Society of Clinical Immunology and Allergy (ASCIA) Web site. Updated October 19, 2019. www.allergy.org.au/patients/insect-allergy-bites-and-stings/jack-jumper-ant-allergy. Accessed November 25, 2020.
4. Kemp SF, deShazo RD, Moffit JE, et al. Expanding habitat of the imported fire ant (Solenopsis invicta): a public health concern. J Allergy Clin Immunol. 2000;105:683-691.
5. Goodnight ML. Arachnid. In: Encyclopædia Britannica. 2012. www.britannica.com/animal/arachnid. Accessed November 25, 2020.
6. Despommier DD, Gwadz RW, Hotez PJ. Arachnids. In: Despommier DD, Gwadz RW, Hotez PJ. Parasitic Diseases. 3rd ed. Springer-Verlag; 1995:268-283.
7. Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician. 2007;75:869-873.
8. Mowry JB, Spyker DA, Cantilena LR Jr, McMillan N, Ford M. 2013 Annual report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 31st Annual Report. Clin Toxicol (Phila). 2014;52:1032-1283.
9. Pucci S, Antonicelli L, Bilò MB, et al. Shortness of interval between two stings as risk factor for developing Hymenoptera venom allergy. Allergy.1994;49:894-896.
10. Müller UR. Bee venom allergy in beekeepers and their family members. Curr Opin Allergy Clin Immunol. 2005;5:343-347.
11. Müller UR. Cardiovascular disease and anaphylaxis. Curr Opin Allergy Clin Immunol. 2007;7:337-341.
12. Golden DBK. Stinging insect allergy. Am Fam Physician. 2003;67:2541-2546.
13. Golden DBK, Demain T, Freeman T, et al. Stinging insect hypersensitivity: a practice parameter update 2016. Ann Allergy Asthma Immunol. 2017;118:28-54.
14. Bilò BM, Rueff F, Mosbech H, et al; EAACI Interest Group on Insect Venom Hypersensitivity. Diagnosis of Hymenoptera venom allergy. Allergy. 2005;60:1339-1349.
15. Reisman RE. Insect stings. N Engl J Med. 1994;331:523-527.
16. Pucci S, D’Alò S, De Pasquale T, et al. Risk of anaphylaxis in patients with large local reactions to hymenoptera stings: a retrospective and prospective study. Clin Mol Allergy. 2015;13:21.
17. Golden DBK. Large local reactions to insect stings. J Allergy Clin Immunol Pract. 2015;3:331-334.
18. Clark S, Camargo CA Jr. Emergency treatment and prevention of insect-sting anaphylaxis. Curr Opin Allergy Clin Immunol. 2006;6:279-283.
19. Stinging insect allergy. In: Volcheck GW. Clinical Allergy: Diagnosis and Management. Humana Press; 2009:465-479.
20. Järvinen KM, Celestin J. Anaphylaxis avoidance and management: educating patients and their caregivers. J Asthma Allergy. 2014;7:95-104.
21. Institute for Quality and Efficiency in Health Care (IQWiG). Insect venom allergies: overview. InformedHealth.org. Updated May 7, 2020. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0096282/. Accessed November 25, 2020.
22. Casale TB, Burks AW. Clinical practice. Hymenoptera-sting hypersensitivity. N Engl J Med. 2014;370:1432-1439.
23. Shelley RM. Centipedes and millipedes with emphasis on North American fauna. Kansas School Naturalist. 1999;45:1-16. https://sites.google.com/g.emporia.edu/ksn/ksn-home/vol-45-no-3-centipedes-and-millipedes-with-emphasis-on-n-america-fauna#h.p_JEf3uDlTg0jw. Accessed November 25, 2020.
24. Ogg B. Centipedes and millipedes. Nebraska Extension in Lancaster County Web site. https://lancaster.unl.edu/pest/resources/CentipedeMillipede012.shtml. Accessed November 25, 2020.
25. Cushing PE. Spiders (Arachnida: Araneae). In: Capinera JL, ed. Encyclopedia of Entomology. Springer, Dordrecht; 2008:226.
26. Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician. 2007;75:869-873.
27. The National Institute for Occupational Safety and Health (NIOSH), Centers for Disease Control and Prevention. Venomous spiders. www.cdc.gov/niosh/topics/spiders/. Accessed November 25, 2020.
28. Starr S. What you need to know to prevent a poisonous spider bite. AAP News. 2013;34:42. www.aappublications.org/content/aapnews/34/9/42.5.full.pdf. Accessed November 25, 2020.
29. Spider bites. Mayo Clinic Web site. www.mayoclinic.org/diseases-conditions/spider-bites/symptoms-causes/syc-20352371. Accessed November 25, 2020.
30. Barish RA, Arnold T. Spider bites. In: Merck Manual (Professional Version). Merck Sharp & Dohme Corp.; 2016. www.merckmanuals.com/professional/injuries-poisoning/bites-and-stings/spider-bites. Accessed November 25, 2020.
31. Juckett G. Arthropod bites. Am Fam Physician. 2013;88:841-847.
32. Clark RF, Wethern-Kestner S, Vance MV, et al. Clinical presentation and treatment of black widow spider envenomation: a review of 163 cases. Ann Emerg Med. 1992;21:782-787.
33. Koehler PG, Pereira RM, Diclaro JW II. Fleas. Publication ENY-025. University of Florida IFAS Extension. Revised January 2012. https://edis.ifas.ufl.edu/ig087. Accessed November 25, 2020.
34. Bitam I, Dittmar K, Parola P, et al. Fleas and flea-borne diseases. Int J Infect Dis. 2010;14:e667-e676.
35. Leulmi H, Socolovschi C, Laudisoit A, et al. Detection of Rickettsia felis, Rickettsia typhi, Bartonella species and Yersinia pestis in fleas (Siphonaptera) from Africa. PLoS Negl Trop Dis. 2014;8:e3152.
36. Naimer SA, Cohen AD, Mumcuoglu KY, et al. Household papular urticaria. Isr Med Assoc J. 2002;4(11 suppl):911-913.
37. Golomb MR, Golomb HS. What’s eating you? Cat flea (Ctenocephalides felis). Cutis. 2010;85:10-11.
38. Dryden MW. Flea and tick control in the 21st century: challenges and opportunities. Vet Dermatol. 2009;20:435-440.
39. Dryden MW. Fleas in dogs and cats. Merck Sharp & Dohme Corporation: Merck Manual Veterinary Manual. Updated December 2014. www.merckvetmanual.com/integumentary-system/fleas-and-flea-allergy-dermatitis/fleas-in-dogs-and-cats. Accessed November 25, 2020.
40. Centers for Disease Control and Prevention. Getting rid of fleas. www.cdc.gov/fleas/getting_rid.html. Accessed November 25, 2020.
41. Chattopadhyay P, Goyary D, Dhiman S, et al. Immunomodulating effects and hypersensitivity reactions caused by Northeast Indian black fly salivary gland extract. J Immunotoxicol. 2014;11:126-132.
42. Hrabak TM, Dice JP. Use of immunotherapy in the management of presumed anaphylaxis to the deer fly. Ann Allergy Asthma Immunol. 2003;90:351-354.
43. Royden A, Wedley A, Merga JY, et al. A role for flies (Diptera) in the transmission of Campylobacter to broilers? Epidemiol Infect. 2016;144:3326-3334.
44. Fradin MS, Day JF. Comparative efficacy of insect repellents against mosquito bites. N Engl J Med. 2002;347:13-18.

45. Carpenter S, Groschup MH, Garros C, et al. Culicoides biting midges, arboviruses and public health in Europe. Antiviral Res. 2013;100:102-113.
46. Peng Z, Yang M, Simons FE. Immunologic mechanisms in mosquito allergy: correlation of skin reactions with specific IgE and IgG anti-bodies and lymphocyte proliferation response to mosquito antigens. Ann Allergy Asthma Immunol. 1996;77:238-244.
47. Simons FE, Peng Z. Skeeter syndrome. J Allergy Clin Immunol. 1999;104:705-707.
48. Centers for Disease Control and Prevention. Travelers’ health. Clinician resources. wwwnc.cdc.gov/travel/page/clinician-information-center. Accessed November 25, 2020.
49. Gauci M, Loh RK, Stone BF, et al. Allergic reactions to the Australian paralysis tick, Ixodes holocyclus: diagnostic evaluation by skin test and radioimmunoassay. Clin Exp Allergy. 1989;19:279-283.
50. Centers for Disease Control and Prevention. Ticks. Removing a tick. www.cdc.gov/ticks/removing_a_tick.html. Accessed November 25, 2020.
51. Golden DB, Kagey-Sobotka A, Norman PS, et al. Insect sting allergy with negative venom skin test responses. J Allergy Clin Immunol. 2001;107:897-901.
52. Arzt L, Bokanovic D, Schrautzer C, et al. Immunological differences between insect venom-allergic patients with and without immunotherapy and asymptomatically sensitized subjects. Allergy. 2018;73:1223-1231.
53. Heddle R, Golden DBK. Allergy to insect stings and bites. World Allergy Organization Web site. Updated August 2015. www.worldallergy.org/education-and-programs/education/allergic-disease-resource-center/professionals/allergy-to-insect-stings-and-bites. Accessed November 25, 2020.
54. RuëffF, Przybilla B, Müller U, et al. The sting challenge test in Hymenoptera venom allergy. Position paper of the Subcommittee on Insect Venom Allergy of the European Academy of Allergology and Clinical Immunology. Allergy. 1996;51:216-225.
55. Management of simple insect bites: where’s the evidence? Drug Ther Bull. 2012;50:45-48.
56. Tracy JM. Insect allergy. Mt Sinai J Med. 2011;78:773-783.
57. Golden DBK. Insect sting allergy and venom immunotherapy: a model and a mystery. J Allergy Clin Immunol. 2005;115:439-447.
58. Winther L, Arnved J, Malling H-J, et al. Side-effects of allergen-specific immunotherapy: a prospective multi-centre study. Clin Exp Allergy. 2006;36:254-260.
59. Mellerup MT, Hahn GW, Poulsen LK, et al. Safety of allergen-specific immunotherapy. Relation between dosage regimen, allergen extract, disease and systemic side-effects during induction treatment. Clin Exp Allergy. 2000;30:1423-1429.
60. Anaphylaxis and insect stings and bites. Med Lett Drugs Ther. 2017;59:e79-e82.
61. Sampson HA, Muñoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: summary report—second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. Ann Emerg Med. 2006;47:373-380.
62. Pflipsen MC, Vega Colon KM. Anaphylaxis: recognition and management. Am Fam Physician. 2020;102:355-362. Accessed November 25, 2020.
1. Numbers of insects (species and individuals). Smithsonian BugInfo Web site. www.si.edu/spotlight/buginfo/bugnos. Accessed November 25, 2020.
2. Antonicelli L, Bilò MB, Bonifazi F. Epidemiology of Hymenoptera allergy. Curr Opin Allergy Clin Immunol. 2002;2:341-346.
3. Jack jumper ant allergy. Australasian Society of Clinical Immunology and Allergy (ASCIA) Web site. Updated October 19, 2019. www.allergy.org.au/patients/insect-allergy-bites-and-stings/jack-jumper-ant-allergy. Accessed November 25, 2020.
4. Kemp SF, deShazo RD, Moffit JE, et al. Expanding habitat of the imported fire ant (Solenopsis invicta): a public health concern. J Allergy Clin Immunol. 2000;105:683-691.
5. Goodnight ML. Arachnid. In: Encyclopædia Britannica. 2012. www.britannica.com/animal/arachnid. Accessed November 25, 2020.
6. Despommier DD, Gwadz RW, Hotez PJ. Arachnids. In: Despommier DD, Gwadz RW, Hotez PJ. Parasitic Diseases. 3rd ed. Springer-Verlag; 1995:268-283.
7. Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician. 2007;75:869-873.
8. Mowry JB, Spyker DA, Cantilena LR Jr, McMillan N, Ford M. 2013 Annual report of the American Association of Poison Control Centers’ National Poison Data System (NPDS): 31st Annual Report. Clin Toxicol (Phila). 2014;52:1032-1283.
9. Pucci S, Antonicelli L, Bilò MB, et al. Shortness of interval between two stings as risk factor for developing Hymenoptera venom allergy. Allergy.1994;49:894-896.
10. Müller UR. Bee venom allergy in beekeepers and their family members. Curr Opin Allergy Clin Immunol. 2005;5:343-347.
11. Müller UR. Cardiovascular disease and anaphylaxis. Curr Opin Allergy Clin Immunol. 2007;7:337-341.
12. Golden DBK. Stinging insect allergy. Am Fam Physician. 2003;67:2541-2546.
13. Golden DBK, Demain T, Freeman T, et al. Stinging insect hypersensitivity: a practice parameter update 2016. Ann Allergy Asthma Immunol. 2017;118:28-54.
14. Bilò BM, Rueff F, Mosbech H, et al; EAACI Interest Group on Insect Venom Hypersensitivity. Diagnosis of Hymenoptera venom allergy. Allergy. 2005;60:1339-1349.
15. Reisman RE. Insect stings. N Engl J Med. 1994;331:523-527.
16. Pucci S, D’Alò S, De Pasquale T, et al. Risk of anaphylaxis in patients with large local reactions to hymenoptera stings: a retrospective and prospective study. Clin Mol Allergy. 2015;13:21.
17. Golden DBK. Large local reactions to insect stings. J Allergy Clin Immunol Pract. 2015;3:331-334.
18. Clark S, Camargo CA Jr. Emergency treatment and prevention of insect-sting anaphylaxis. Curr Opin Allergy Clin Immunol. 2006;6:279-283.
19. Stinging insect allergy. In: Volcheck GW. Clinical Allergy: Diagnosis and Management. Humana Press; 2009:465-479.
20. Järvinen KM, Celestin J. Anaphylaxis avoidance and management: educating patients and their caregivers. J Asthma Allergy. 2014;7:95-104.
21. Institute for Quality and Efficiency in Health Care (IQWiG). Insect venom allergies: overview. InformedHealth.org. Updated May 7, 2020. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0096282/. Accessed November 25, 2020.
22. Casale TB, Burks AW. Clinical practice. Hymenoptera-sting hypersensitivity. N Engl J Med. 2014;370:1432-1439.
23. Shelley RM. Centipedes and millipedes with emphasis on North American fauna. Kansas School Naturalist. 1999;45:1-16. https://sites.google.com/g.emporia.edu/ksn/ksn-home/vol-45-no-3-centipedes-and-millipedes-with-emphasis-on-n-america-fauna#h.p_JEf3uDlTg0jw. Accessed November 25, 2020.
24. Ogg B. Centipedes and millipedes. Nebraska Extension in Lancaster County Web site. https://lancaster.unl.edu/pest/resources/CentipedeMillipede012.shtml. Accessed November 25, 2020.
25. Cushing PE. Spiders (Arachnida: Araneae). In: Capinera JL, ed. Encyclopedia of Entomology. Springer, Dordrecht; 2008:226.
26. Diaz JH, Leblanc KE. Common spider bites. Am Fam Physician. 2007;75:869-873.
27. The National Institute for Occupational Safety and Health (NIOSH), Centers for Disease Control and Prevention. Venomous spiders. www.cdc.gov/niosh/topics/spiders/. Accessed November 25, 2020.
28. Starr S. What you need to know to prevent a poisonous spider bite. AAP News. 2013;34:42. www.aappublications.org/content/aapnews/34/9/42.5.full.pdf. Accessed November 25, 2020.
29. Spider bites. Mayo Clinic Web site. www.mayoclinic.org/diseases-conditions/spider-bites/symptoms-causes/syc-20352371. Accessed November 25, 2020.
30. Barish RA, Arnold T. Spider bites. In: Merck Manual (Professional Version). Merck Sharp & Dohme Corp.; 2016. www.merckmanuals.com/professional/injuries-poisoning/bites-and-stings/spider-bites. Accessed November 25, 2020.
31. Juckett G. Arthropod bites. Am Fam Physician. 2013;88:841-847.
32. Clark RF, Wethern-Kestner S, Vance MV, et al. Clinical presentation and treatment of black widow spider envenomation: a review of 163 cases. Ann Emerg Med. 1992;21:782-787.
33. Koehler PG, Pereira RM, Diclaro JW II. Fleas. Publication ENY-025. University of Florida IFAS Extension. Revised January 2012. https://edis.ifas.ufl.edu/ig087. Accessed November 25, 2020.
34. Bitam I, Dittmar K, Parola P, et al. Fleas and flea-borne diseases. Int J Infect Dis. 2010;14:e667-e676.
35. Leulmi H, Socolovschi C, Laudisoit A, et al. Detection of Rickettsia felis, Rickettsia typhi, Bartonella species and Yersinia pestis in fleas (Siphonaptera) from Africa. PLoS Negl Trop Dis. 2014;8:e3152.
36. Naimer SA, Cohen AD, Mumcuoglu KY, et al. Household papular urticaria. Isr Med Assoc J. 2002;4(11 suppl):911-913.
37. Golomb MR, Golomb HS. What’s eating you? Cat flea (Ctenocephalides felis). Cutis. 2010;85:10-11.
38. Dryden MW. Flea and tick control in the 21st century: challenges and opportunities. Vet Dermatol. 2009;20:435-440.
39. Dryden MW. Fleas in dogs and cats. Merck Sharp & Dohme Corporation: Merck Manual Veterinary Manual. Updated December 2014. www.merckvetmanual.com/integumentary-system/fleas-and-flea-allergy-dermatitis/fleas-in-dogs-and-cats. Accessed November 25, 2020.
40. Centers for Disease Control and Prevention. Getting rid of fleas. www.cdc.gov/fleas/getting_rid.html. Accessed November 25, 2020.
41. Chattopadhyay P, Goyary D, Dhiman S, et al. Immunomodulating effects and hypersensitivity reactions caused by Northeast Indian black fly salivary gland extract. J Immunotoxicol. 2014;11:126-132.
42. Hrabak TM, Dice JP. Use of immunotherapy in the management of presumed anaphylaxis to the deer fly. Ann Allergy Asthma Immunol. 2003;90:351-354.
43. Royden A, Wedley A, Merga JY, et al. A role for flies (Diptera) in the transmission of Campylobacter to broilers? Epidemiol Infect. 2016;144:3326-3334.
44. Fradin MS, Day JF. Comparative efficacy of insect repellents against mosquito bites. N Engl J Med. 2002;347:13-18.

45. Carpenter S, Groschup MH, Garros C, et al. Culicoides biting midges, arboviruses and public health in Europe. Antiviral Res. 2013;100:102-113.
46. Peng Z, Yang M, Simons FE. Immunologic mechanisms in mosquito allergy: correlation of skin reactions with specific IgE and IgG anti-bodies and lymphocyte proliferation response to mosquito antigens. Ann Allergy Asthma Immunol. 1996;77:238-244.
47. Simons FE, Peng Z. Skeeter syndrome. J Allergy Clin Immunol. 1999;104:705-707.
48. Centers for Disease Control and Prevention. Travelers’ health. Clinician resources. wwwnc.cdc.gov/travel/page/clinician-information-center. Accessed November 25, 2020.
49. Gauci M, Loh RK, Stone BF, et al. Allergic reactions to the Australian paralysis tick, Ixodes holocyclus: diagnostic evaluation by skin test and radioimmunoassay. Clin Exp Allergy. 1989;19:279-283.
50. Centers for Disease Control and Prevention. Ticks. Removing a tick. www.cdc.gov/ticks/removing_a_tick.html. Accessed November 25, 2020.
51. Golden DB, Kagey-Sobotka A, Norman PS, et al. Insect sting allergy with negative venom skin test responses. J Allergy Clin Immunol. 2001;107:897-901.
52. Arzt L, Bokanovic D, Schrautzer C, et al. Immunological differences between insect venom-allergic patients with and without immunotherapy and asymptomatically sensitized subjects. Allergy. 2018;73:1223-1231.
53. Heddle R, Golden DBK. Allergy to insect stings and bites. World Allergy Organization Web site. Updated August 2015. www.worldallergy.org/education-and-programs/education/allergic-disease-resource-center/professionals/allergy-to-insect-stings-and-bites. Accessed November 25, 2020.
54. RuëffF, Przybilla B, Müller U, et al. The sting challenge test in Hymenoptera venom allergy. Position paper of the Subcommittee on Insect Venom Allergy of the European Academy of Allergology and Clinical Immunology. Allergy. 1996;51:216-225.
55. Management of simple insect bites: where’s the evidence? Drug Ther Bull. 2012;50:45-48.
56. Tracy JM. Insect allergy. Mt Sinai J Med. 2011;78:773-783.
57. Golden DBK. Insect sting allergy and venom immunotherapy: a model and a mystery. J Allergy Clin Immunol. 2005;115:439-447.
58. Winther L, Arnved J, Malling H-J, et al. Side-effects of allergen-specific immunotherapy: a prospective multi-centre study. Clin Exp Allergy. 2006;36:254-260.
59. Mellerup MT, Hahn GW, Poulsen LK, et al. Safety of allergen-specific immunotherapy. Relation between dosage regimen, allergen extract, disease and systemic side-effects during induction treatment. Clin Exp Allergy. 2000;30:1423-1429.
60. Anaphylaxis and insect stings and bites. Med Lett Drugs Ther. 2017;59:e79-e82.
61. Sampson HA, Muñoz-Furlong A, Campbell RL, et al. Second symposium on the definition and management of anaphylaxis: summary report—second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. Ann Emerg Med. 2006;47:373-380.
62. Pflipsen MC, Vega Colon KM. Anaphylaxis: recognition and management. Am Fam Physician. 2020;102:355-362. Accessed November 25, 2020.
PRACTICE RECOMMENDATIONS
❯ Recommend that patients use an insect repellent, such as an over-the-counter formulation that contains DEET, picaridin, or PMD (a chemical constituent of Eucalyptus citriodora oil) to prevent flea bites. C
❯ Prescribe nonsedating oral antihistamines as first-line symptomatic treatment of mild-to-moderate pruritus secondary to an insect bite. C
❯ When indicated, refer patients for venom immunotherapy, which is approximately 95% effective in preventing or reducing severe systemic reactions and reduces the risk of anaphylaxis and death. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Antihistamine prescribing for AD varies by specialty
(AD), according to an analysis of a national database.
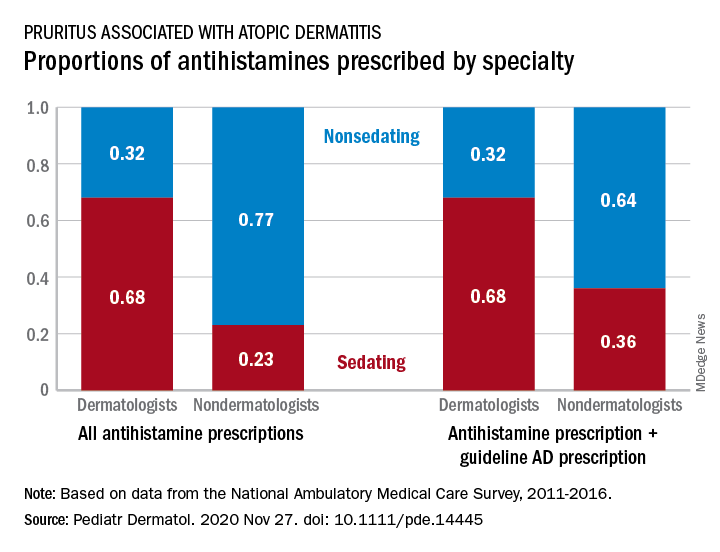
Dermatologists were more likely to prescribe sedating than nonsedating antihistamines (0.68 vs. 0.32) for patients with AD, but the reverse applied to nondermatologists, whose antihistamine distribution was 0.23 sedating and 0.77 nonsedating, based on 2011-2016 data from the National Ambulatory Medical Care Survey.
The numbers were similar for new antihistamine prescriptions, with sedating/nonsedating proportions of 0.60/0.40 for dermatologists and 0.24/0.76 for nondermatologists. Addition of guideline-recommended drugs such as topical corticosteroids and calcineurin inhibitors to the AD equation did not change the result, as dermatologists again showed a preference for sedating antihistamines, compared with nondermatologists, the investigators said.
The data also showed that Black patients with AD were more likely than were White patients to receive prescriptions for first-generation antihistamines and for therapies recommended by the AAD guidelines, and that patients under 21 years received more sedating antihistamines than did patients over age 21, they reported.
The age disparity “may be due to patient preference, as sedation effects may be less desirable to adult patients,” the investigators noted.
SOURCE: Garg S et al. Pediatr Dermatol. 2020 Nov 27. doi: 10.1111/pde.14445.
(AD), according to an analysis of a national database.

Dermatologists were more likely to prescribe sedating than nonsedating antihistamines (0.68 vs. 0.32) for patients with AD, but the reverse applied to nondermatologists, whose antihistamine distribution was 0.23 sedating and 0.77 nonsedating, based on 2011-2016 data from the National Ambulatory Medical Care Survey.
The numbers were similar for new antihistamine prescriptions, with sedating/nonsedating proportions of 0.60/0.40 for dermatologists and 0.24/0.76 for nondermatologists. Addition of guideline-recommended drugs such as topical corticosteroids and calcineurin inhibitors to the AD equation did not change the result, as dermatologists again showed a preference for sedating antihistamines, compared with nondermatologists, the investigators said.
The data also showed that Black patients with AD were more likely than were White patients to receive prescriptions for first-generation antihistamines and for therapies recommended by the AAD guidelines, and that patients under 21 years received more sedating antihistamines than did patients over age 21, they reported.
The age disparity “may be due to patient preference, as sedation effects may be less desirable to adult patients,” the investigators noted.
SOURCE: Garg S et al. Pediatr Dermatol. 2020 Nov 27. doi: 10.1111/pde.14445.
(AD), according to an analysis of a national database.

Dermatologists were more likely to prescribe sedating than nonsedating antihistamines (0.68 vs. 0.32) for patients with AD, but the reverse applied to nondermatologists, whose antihistamine distribution was 0.23 sedating and 0.77 nonsedating, based on 2011-2016 data from the National Ambulatory Medical Care Survey.
The numbers were similar for new antihistamine prescriptions, with sedating/nonsedating proportions of 0.60/0.40 for dermatologists and 0.24/0.76 for nondermatologists. Addition of guideline-recommended drugs such as topical corticosteroids and calcineurin inhibitors to the AD equation did not change the result, as dermatologists again showed a preference for sedating antihistamines, compared with nondermatologists, the investigators said.
The data also showed that Black patients with AD were more likely than were White patients to receive prescriptions for first-generation antihistamines and for therapies recommended by the AAD guidelines, and that patients under 21 years received more sedating antihistamines than did patients over age 21, they reported.
The age disparity “may be due to patient preference, as sedation effects may be less desirable to adult patients,” the investigators noted.
SOURCE: Garg S et al. Pediatr Dermatol. 2020 Nov 27. doi: 10.1111/pde.14445.
FROM PEDIATRIC DERMATOLOGY
A 70-year-old presented with a 3-week history of asymptomatic violaceous papules on his feet
and named the condition multiple benign pigmented hemorrhagic sarcoma. The disease emerged again at the onset of the AIDS epidemic among homosexual men. There are five variants: HIV/AIDS–related KS, classic KS, African cutaneous KS, African lymphadenopathic KS, and immunosuppression-associated KS (from immunosuppressive therapy or malignancies such as lymphoma).
KS is caused by human herpes virus type 8 (HHV-8). Patients with KS have an increased risk of developing other malignancies such as lymphomas, leukemia, and myeloma. This patient exhibited classic KS.
The various forms of KS may appear different clinically. The lesions may appear as erythematous macules, small violaceous papules, large plaques, or ulcerated nodules. In classic KS, violaceous to bluish-black macules evolve to papules or plaques. Lesions are generally asymptomatic. The most common locations are the toes and soles, although other areas may be affected. Any mucocutaneous surface can be involved. The most common areas of internal involvement are the gastrointestinal system and lymphatics.
Histology reveals angular vessels lined by atypical cells. An associated inflammatory infiltrate containing plasma cells may be present in the upper dermis and perivascular areas. Nodules and plaques reveal a spindle cell neoplasm pattern. Lesions will stain positive for HHV-8.
In patients with HIV/AIDS–related KS, highly active antiretroviral therapy is the most important and beneficial treatment. Since the introduction of HAART, the incidence of KS has greatly decreased. However, there are a proportion of HIV/AIDS–associated Kaposi’s sarcoma patients with well-controlled HIV and undetectable viral loads who require further treatment.
Lesions may spontaneously resolve on their own. Other treatment methods include: cryotherapy, topical alitretinoin (9-cis-retinoic acid), intralesional interferon-alpha or vinblastine, superficial radiotherapy, liposomal doxorubicin, daunorubicin or paclitaxel. Small lesions that are asymptomatic may be monitored.
This patient had no internal involvement and responded well to cryotherapy.
This case and photo were provided by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
and named the condition multiple benign pigmented hemorrhagic sarcoma. The disease emerged again at the onset of the AIDS epidemic among homosexual men. There are five variants: HIV/AIDS–related KS, classic KS, African cutaneous KS, African lymphadenopathic KS, and immunosuppression-associated KS (from immunosuppressive therapy or malignancies such as lymphoma).
KS is caused by human herpes virus type 8 (HHV-8). Patients with KS have an increased risk of developing other malignancies such as lymphomas, leukemia, and myeloma. This patient exhibited classic KS.
The various forms of KS may appear different clinically. The lesions may appear as erythematous macules, small violaceous papules, large plaques, or ulcerated nodules. In classic KS, violaceous to bluish-black macules evolve to papules or plaques. Lesions are generally asymptomatic. The most common locations are the toes and soles, although other areas may be affected. Any mucocutaneous surface can be involved. The most common areas of internal involvement are the gastrointestinal system and lymphatics.
Histology reveals angular vessels lined by atypical cells. An associated inflammatory infiltrate containing plasma cells may be present in the upper dermis and perivascular areas. Nodules and plaques reveal a spindle cell neoplasm pattern. Lesions will stain positive for HHV-8.
In patients with HIV/AIDS–related KS, highly active antiretroviral therapy is the most important and beneficial treatment. Since the introduction of HAART, the incidence of KS has greatly decreased. However, there are a proportion of HIV/AIDS–associated Kaposi’s sarcoma patients with well-controlled HIV and undetectable viral loads who require further treatment.
Lesions may spontaneously resolve on their own. Other treatment methods include: cryotherapy, topical alitretinoin (9-cis-retinoic acid), intralesional interferon-alpha or vinblastine, superficial radiotherapy, liposomal doxorubicin, daunorubicin or paclitaxel. Small lesions that are asymptomatic may be monitored.
This patient had no internal involvement and responded well to cryotherapy.
This case and photo were provided by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
and named the condition multiple benign pigmented hemorrhagic sarcoma. The disease emerged again at the onset of the AIDS epidemic among homosexual men. There are five variants: HIV/AIDS–related KS, classic KS, African cutaneous KS, African lymphadenopathic KS, and immunosuppression-associated KS (from immunosuppressive therapy or malignancies such as lymphoma).
KS is caused by human herpes virus type 8 (HHV-8). Patients with KS have an increased risk of developing other malignancies such as lymphomas, leukemia, and myeloma. This patient exhibited classic KS.
The various forms of KS may appear different clinically. The lesions may appear as erythematous macules, small violaceous papules, large plaques, or ulcerated nodules. In classic KS, violaceous to bluish-black macules evolve to papules or plaques. Lesions are generally asymptomatic. The most common locations are the toes and soles, although other areas may be affected. Any mucocutaneous surface can be involved. The most common areas of internal involvement are the gastrointestinal system and lymphatics.
Histology reveals angular vessels lined by atypical cells. An associated inflammatory infiltrate containing plasma cells may be present in the upper dermis and perivascular areas. Nodules and plaques reveal a spindle cell neoplasm pattern. Lesions will stain positive for HHV-8.
In patients with HIV/AIDS–related KS, highly active antiretroviral therapy is the most important and beneficial treatment. Since the introduction of HAART, the incidence of KS has greatly decreased. However, there are a proportion of HIV/AIDS–associated Kaposi’s sarcoma patients with well-controlled HIV and undetectable viral loads who require further treatment.
Lesions may spontaneously resolve on their own. Other treatment methods include: cryotherapy, topical alitretinoin (9-cis-retinoic acid), intralesional interferon-alpha or vinblastine, superficial radiotherapy, liposomal doxorubicin, daunorubicin or paclitaxel. Small lesions that are asymptomatic may be monitored.
This patient had no internal involvement and responded well to cryotherapy.
This case and photo were provided by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
Study results support screening rosacea patients for cardiometabolic disease
according to the results of a meta-analysis of more than 50,000 patients.
To date, “mounting comorbidities of rosacea have been identified, suggesting that rosacea is not simply a skin disease but has links to multiple systemic illnesses,” wrote Qi Chen, MD, of Central South University, Changsha, China, and colleagues. The association with rosacea and cardiometabolic disease has been controversial, they added.
In a study published in the Journal of the American Academy of Dermatology, they identified 13 studies including 50,442 rosacea patients and 1,525,864 controls. Approximately 71% of the rosacea patients were women.
Overall, patients with rosacea showed a statistically significant association for hypertension (risk ratio, 1.20; 95% confidence interval, 1.08-1.34; P = .001) and dyslipidemia (RR, 1.32; 95% CI, 1.10-1.58; P = .002). Specifically, rosacea patients averaged higher standard mean differences of systolic and diastolic blood pressure, total cholesterol, HDL cholesterol and LDL cholesterol, and triglycerides, compared with controls.
Rosacea was not significantly associated with an increased risk for ischemic heart disease, stroke, or diabetes, although the rosacea patients showed significantly increased risk of higher fasting blood glucose, compared with controls.
Findings don’t show causality
The study findings were limited by several factors, including the observational nature of some of the studies and the inability to perform subgroup analyses based on subtype and disease severity, the researchers noted. In addition, most of the rosacea patients were outpatients. “Further investigations are warranted to identify the relationship between rosacea and [cardiometabolic disease] in general populations to further validate the significance of our findings.”
However, the results support the value of screening for cardiometabolic disease in rosacea patients to facilitate diagnosis and treatment of disease at an early stage, they concluded.
“Rosacea has been linked statistically to many comorbidities including depression, anxiety, hypertension, and diabetes mellitus,” Julie Harper, MD, of the Dermatology and Skin Care Center of Birmingham (Alabama), said in an interview.
“This study looked more specifically at cardiometabolic disease and found a statistically significant correlation between rosacea and hypertension, higher total cholesterol, higher triglycerides and higher fasting blood glucose,” she said. However, “while there is an association present in this meta-analysis, we cannot assume a cause-and-effect relationship.”
Although the analysis does not prove causality, the key message for clinicians is that cardiometabolic disease is quite common in rosacea patients, and risk factors should be identified and treated early, said Dr. Harper. “Our patients with and without rosacea will benefit from age-appropriate screening, physical examination, and laboratory evaluation with a primary care physician. For rosacea patients in particular, we can advise them that early research suggests that individuals with rosacea might have an increased risk of hypertension and/or high cholesterol and triglycerides. It never hurts to make an appointment with primary care and to be checked.”
“We need more confirmatory studies that minimize the influence of confounding,” Dr. Harper added. Rosacea also has also been linked to obesity, which is another risk factor for cardiometabolic disease.
The study was supported by multiple grants from the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose. Dr. Harper had no relevant financial conflicts to disclose.
SOURCE: Chen Q et al. J Am Acad Dermatol. 2020 Nov;83(5):1331-40.
according to the results of a meta-analysis of more than 50,000 patients.
To date, “mounting comorbidities of rosacea have been identified, suggesting that rosacea is not simply a skin disease but has links to multiple systemic illnesses,” wrote Qi Chen, MD, of Central South University, Changsha, China, and colleagues. The association with rosacea and cardiometabolic disease has been controversial, they added.
In a study published in the Journal of the American Academy of Dermatology, they identified 13 studies including 50,442 rosacea patients and 1,525,864 controls. Approximately 71% of the rosacea patients were women.
Overall, patients with rosacea showed a statistically significant association for hypertension (risk ratio, 1.20; 95% confidence interval, 1.08-1.34; P = .001) and dyslipidemia (RR, 1.32; 95% CI, 1.10-1.58; P = .002). Specifically, rosacea patients averaged higher standard mean differences of systolic and diastolic blood pressure, total cholesterol, HDL cholesterol and LDL cholesterol, and triglycerides, compared with controls.
Rosacea was not significantly associated with an increased risk for ischemic heart disease, stroke, or diabetes, although the rosacea patients showed significantly increased risk of higher fasting blood glucose, compared with controls.
Findings don’t show causality
The study findings were limited by several factors, including the observational nature of some of the studies and the inability to perform subgroup analyses based on subtype and disease severity, the researchers noted. In addition, most of the rosacea patients were outpatients. “Further investigations are warranted to identify the relationship between rosacea and [cardiometabolic disease] in general populations to further validate the significance of our findings.”
However, the results support the value of screening for cardiometabolic disease in rosacea patients to facilitate diagnosis and treatment of disease at an early stage, they concluded.
“Rosacea has been linked statistically to many comorbidities including depression, anxiety, hypertension, and diabetes mellitus,” Julie Harper, MD, of the Dermatology and Skin Care Center of Birmingham (Alabama), said in an interview.
“This study looked more specifically at cardiometabolic disease and found a statistically significant correlation between rosacea and hypertension, higher total cholesterol, higher triglycerides and higher fasting blood glucose,” she said. However, “while there is an association present in this meta-analysis, we cannot assume a cause-and-effect relationship.”
Although the analysis does not prove causality, the key message for clinicians is that cardiometabolic disease is quite common in rosacea patients, and risk factors should be identified and treated early, said Dr. Harper. “Our patients with and without rosacea will benefit from age-appropriate screening, physical examination, and laboratory evaluation with a primary care physician. For rosacea patients in particular, we can advise them that early research suggests that individuals with rosacea might have an increased risk of hypertension and/or high cholesterol and triglycerides. It never hurts to make an appointment with primary care and to be checked.”
“We need more confirmatory studies that minimize the influence of confounding,” Dr. Harper added. Rosacea also has also been linked to obesity, which is another risk factor for cardiometabolic disease.
The study was supported by multiple grants from the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose. Dr. Harper had no relevant financial conflicts to disclose.
SOURCE: Chen Q et al. J Am Acad Dermatol. 2020 Nov;83(5):1331-40.
according to the results of a meta-analysis of more than 50,000 patients.
To date, “mounting comorbidities of rosacea have been identified, suggesting that rosacea is not simply a skin disease but has links to multiple systemic illnesses,” wrote Qi Chen, MD, of Central South University, Changsha, China, and colleagues. The association with rosacea and cardiometabolic disease has been controversial, they added.
In a study published in the Journal of the American Academy of Dermatology, they identified 13 studies including 50,442 rosacea patients and 1,525,864 controls. Approximately 71% of the rosacea patients were women.
Overall, patients with rosacea showed a statistically significant association for hypertension (risk ratio, 1.20; 95% confidence interval, 1.08-1.34; P = .001) and dyslipidemia (RR, 1.32; 95% CI, 1.10-1.58; P = .002). Specifically, rosacea patients averaged higher standard mean differences of systolic and diastolic blood pressure, total cholesterol, HDL cholesterol and LDL cholesterol, and triglycerides, compared with controls.
Rosacea was not significantly associated with an increased risk for ischemic heart disease, stroke, or diabetes, although the rosacea patients showed significantly increased risk of higher fasting blood glucose, compared with controls.
Findings don’t show causality
The study findings were limited by several factors, including the observational nature of some of the studies and the inability to perform subgroup analyses based on subtype and disease severity, the researchers noted. In addition, most of the rosacea patients were outpatients. “Further investigations are warranted to identify the relationship between rosacea and [cardiometabolic disease] in general populations to further validate the significance of our findings.”
However, the results support the value of screening for cardiometabolic disease in rosacea patients to facilitate diagnosis and treatment of disease at an early stage, they concluded.
“Rosacea has been linked statistically to many comorbidities including depression, anxiety, hypertension, and diabetes mellitus,” Julie Harper, MD, of the Dermatology and Skin Care Center of Birmingham (Alabama), said in an interview.
“This study looked more specifically at cardiometabolic disease and found a statistically significant correlation between rosacea and hypertension, higher total cholesterol, higher triglycerides and higher fasting blood glucose,” she said. However, “while there is an association present in this meta-analysis, we cannot assume a cause-and-effect relationship.”
Although the analysis does not prove causality, the key message for clinicians is that cardiometabolic disease is quite common in rosacea patients, and risk factors should be identified and treated early, said Dr. Harper. “Our patients with and without rosacea will benefit from age-appropriate screening, physical examination, and laboratory evaluation with a primary care physician. For rosacea patients in particular, we can advise them that early research suggests that individuals with rosacea might have an increased risk of hypertension and/or high cholesterol and triglycerides. It never hurts to make an appointment with primary care and to be checked.”
“We need more confirmatory studies that minimize the influence of confounding,” Dr. Harper added. Rosacea also has also been linked to obesity, which is another risk factor for cardiometabolic disease.
The study was supported by multiple grants from the National Natural Science Foundation of China. The researchers had no financial conflicts to disclose. Dr. Harper had no relevant financial conflicts to disclose.
SOURCE: Chen Q et al. J Am Acad Dermatol. 2020 Nov;83(5):1331-40.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Moving from subtypes to phenotypes is simplifying management of rosacea
When a new phenotype approach to the diagnosis of rosacea was proposed 2 years ago, this simpler and more accurate method was accompanied by several corollary advantages, including a more rational approach to treatment and better methods of measuring treatment efficacy, according to an expert speaking at the annual Coastal Dermatology Symposium, held virtually.
“By looking at rosacea in a more simple way – but a more accurate way – we are able to track what happens [to key features] over time,” explained Jerry Tan, MD, of the University of Western Ontario, London.
With the previous method of subtyping, many rosacea patients failed to fit neatly into any of the four categories, producing confusion and diverting attention from troublesome symptoms.
“Rosacea patients often present with a range of features that span multiple subtypes or progress between them,” Dr. Tan explained. The risk is not just a delay in diagnosis but a failure to focus on symptoms patients find most bothersome.
The previous diagnostic criteria for rosacea, published in 2002, identified primary and secondary symptoms within its four subtypes. The new diagnostic criteria, endorsed by the National Rosacea Society and published in 2018, rely on phenotypes defined by diagnostic, major, and minor symptoms. Rather than the four previous subtypes, which were erythematotelangiectatic, papulopustular, phymatous, and ocular, the phenotypes facilitate diagnosis in patients with mixed features.
By replacing “the old thought process of subtyping” with a newer focus on phenotypes, the updated criteria were “aimed toward accuracy, simplicity and practicality,” Dr. Tan said.
Moreover, without squeezing patients into subgroups where they do not neatly fit, the new criteria draw attention to the specific symptoms that bring patients to the clinician.
The phenotype approach to treatment strategies was reflected in a systematic review of treatments based on phenotypes that was published in 2019, not long after the new classification system became available. In this review, coauthored by Dr. Tan, the GRADE certainty-of-evidence approach was employed to identify effective therapies, matching specific symptoms with specific therapies such as low-dose isotretinoin for papules or omega-3 fatty acids for dry eyes.
Based on a patient-centric approach that emphasizes control of key symptoms, Dr. Tan also described a method of documenting the severity of major and minor symptoms at each visit. With this method, called a rosacea patient tracker, patients and physicians can determine whether therapies are effective against the signs and symptoms of disease that they find most burdensome, according to Dr. Tan, who was the first author of an article he cited as a reference to this phenotype-based methodology.
Overall, the phenotype approach to rosacea “rationalizes treatment,” he said.
Specifically, the heterogeneity of symptoms in rosacea is mirrored in the heterogeneity of underlying pathophysiology. According to Dr. Tan, the upregulation of cytokines for inflammation, of angiogenic pathways for vascular symptoms, and of matrix metalloproteinases for tissue remodeling are all implicated in rosacea but drive different types of symptoms. While appropriate skin care and efforts to identify and minimize symptom triggers is appropriate for all patients, phenotypes provide a guide to the most appropriate therapies.
He said he hopes that the focus on phenotypes will draw attention to differences in these pathophysiological mechanisms. According to Dr. Tan, evaluating rosacea from the perspective of phenotypes has represented an important paradigm shift that extends beyond diagnosis.
“The move to the phenotype approach is hopefully simpler, more accurate, and more relevant,” Dr. Tan said.
This same approach has been advocated by others, including Esther J. van Zurren, MD, professor of dermatology at Leiden University Medical Centre in the Netherlands, the lead author of the 2018 systematic review article discussed by Dr. Tan. In this review article on the phenotype approach, specific strategies were recommended for specific symptoms on the basis of grading by an international group of experts that included Dr. Tan, a coauthor.
“These strategies should be directed toward achieving improvements in general well-being by targeting those aspects most bothersome to the patient,” the article advises. Like Dr. Tan, she considers this phenotype-based approach to diagnosis and treatment to be a meaningful clinical advance over the guidelines published in 2002.
“Management strategies for people with rosacea should include phenotype-based treatments,” she agreed, adding that specific choices should be made on the basis of these phenotypes “instead of the previous subtype classification.”
The meeting was jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are owned by the same parent company.
When a new phenotype approach to the diagnosis of rosacea was proposed 2 years ago, this simpler and more accurate method was accompanied by several corollary advantages, including a more rational approach to treatment and better methods of measuring treatment efficacy, according to an expert speaking at the annual Coastal Dermatology Symposium, held virtually.
“By looking at rosacea in a more simple way – but a more accurate way – we are able to track what happens [to key features] over time,” explained Jerry Tan, MD, of the University of Western Ontario, London.
With the previous method of subtyping, many rosacea patients failed to fit neatly into any of the four categories, producing confusion and diverting attention from troublesome symptoms.
“Rosacea patients often present with a range of features that span multiple subtypes or progress between them,” Dr. Tan explained. The risk is not just a delay in diagnosis but a failure to focus on symptoms patients find most bothersome.
The previous diagnostic criteria for rosacea, published in 2002, identified primary and secondary symptoms within its four subtypes. The new diagnostic criteria, endorsed by the National Rosacea Society and published in 2018, rely on phenotypes defined by diagnostic, major, and minor symptoms. Rather than the four previous subtypes, which were erythematotelangiectatic, papulopustular, phymatous, and ocular, the phenotypes facilitate diagnosis in patients with mixed features.
By replacing “the old thought process of subtyping” with a newer focus on phenotypes, the updated criteria were “aimed toward accuracy, simplicity and practicality,” Dr. Tan said.
Moreover, without squeezing patients into subgroups where they do not neatly fit, the new criteria draw attention to the specific symptoms that bring patients to the clinician.
The phenotype approach to treatment strategies was reflected in a systematic review of treatments based on phenotypes that was published in 2019, not long after the new classification system became available. In this review, coauthored by Dr. Tan, the GRADE certainty-of-evidence approach was employed to identify effective therapies, matching specific symptoms with specific therapies such as low-dose isotretinoin for papules or omega-3 fatty acids for dry eyes.
Based on a patient-centric approach that emphasizes control of key symptoms, Dr. Tan also described a method of documenting the severity of major and minor symptoms at each visit. With this method, called a rosacea patient tracker, patients and physicians can determine whether therapies are effective against the signs and symptoms of disease that they find most burdensome, according to Dr. Tan, who was the first author of an article he cited as a reference to this phenotype-based methodology.
Overall, the phenotype approach to rosacea “rationalizes treatment,” he said.
Specifically, the heterogeneity of symptoms in rosacea is mirrored in the heterogeneity of underlying pathophysiology. According to Dr. Tan, the upregulation of cytokines for inflammation, of angiogenic pathways for vascular symptoms, and of matrix metalloproteinases for tissue remodeling are all implicated in rosacea but drive different types of symptoms. While appropriate skin care and efforts to identify and minimize symptom triggers is appropriate for all patients, phenotypes provide a guide to the most appropriate therapies.
He said he hopes that the focus on phenotypes will draw attention to differences in these pathophysiological mechanisms. According to Dr. Tan, evaluating rosacea from the perspective of phenotypes has represented an important paradigm shift that extends beyond diagnosis.
“The move to the phenotype approach is hopefully simpler, more accurate, and more relevant,” Dr. Tan said.
This same approach has been advocated by others, including Esther J. van Zurren, MD, professor of dermatology at Leiden University Medical Centre in the Netherlands, the lead author of the 2018 systematic review article discussed by Dr. Tan. In this review article on the phenotype approach, specific strategies were recommended for specific symptoms on the basis of grading by an international group of experts that included Dr. Tan, a coauthor.
“These strategies should be directed toward achieving improvements in general well-being by targeting those aspects most bothersome to the patient,” the article advises. Like Dr. Tan, she considers this phenotype-based approach to diagnosis and treatment to be a meaningful clinical advance over the guidelines published in 2002.
“Management strategies for people with rosacea should include phenotype-based treatments,” she agreed, adding that specific choices should be made on the basis of these phenotypes “instead of the previous subtype classification.”
The meeting was jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are owned by the same parent company.
When a new phenotype approach to the diagnosis of rosacea was proposed 2 years ago, this simpler and more accurate method was accompanied by several corollary advantages, including a more rational approach to treatment and better methods of measuring treatment efficacy, according to an expert speaking at the annual Coastal Dermatology Symposium, held virtually.
“By looking at rosacea in a more simple way – but a more accurate way – we are able to track what happens [to key features] over time,” explained Jerry Tan, MD, of the University of Western Ontario, London.
With the previous method of subtyping, many rosacea patients failed to fit neatly into any of the four categories, producing confusion and diverting attention from troublesome symptoms.
“Rosacea patients often present with a range of features that span multiple subtypes or progress between them,” Dr. Tan explained. The risk is not just a delay in diagnosis but a failure to focus on symptoms patients find most bothersome.
The previous diagnostic criteria for rosacea, published in 2002, identified primary and secondary symptoms within its four subtypes. The new diagnostic criteria, endorsed by the National Rosacea Society and published in 2018, rely on phenotypes defined by diagnostic, major, and minor symptoms. Rather than the four previous subtypes, which were erythematotelangiectatic, papulopustular, phymatous, and ocular, the phenotypes facilitate diagnosis in patients with mixed features.
By replacing “the old thought process of subtyping” with a newer focus on phenotypes, the updated criteria were “aimed toward accuracy, simplicity and practicality,” Dr. Tan said.
Moreover, without squeezing patients into subgroups where they do not neatly fit, the new criteria draw attention to the specific symptoms that bring patients to the clinician.
The phenotype approach to treatment strategies was reflected in a systematic review of treatments based on phenotypes that was published in 2019, not long after the new classification system became available. In this review, coauthored by Dr. Tan, the GRADE certainty-of-evidence approach was employed to identify effective therapies, matching specific symptoms with specific therapies such as low-dose isotretinoin for papules or omega-3 fatty acids for dry eyes.
Based on a patient-centric approach that emphasizes control of key symptoms, Dr. Tan also described a method of documenting the severity of major and minor symptoms at each visit. With this method, called a rosacea patient tracker, patients and physicians can determine whether therapies are effective against the signs and symptoms of disease that they find most burdensome, according to Dr. Tan, who was the first author of an article he cited as a reference to this phenotype-based methodology.
Overall, the phenotype approach to rosacea “rationalizes treatment,” he said.
Specifically, the heterogeneity of symptoms in rosacea is mirrored in the heterogeneity of underlying pathophysiology. According to Dr. Tan, the upregulation of cytokines for inflammation, of angiogenic pathways for vascular symptoms, and of matrix metalloproteinases for tissue remodeling are all implicated in rosacea but drive different types of symptoms. While appropriate skin care and efforts to identify and minimize symptom triggers is appropriate for all patients, phenotypes provide a guide to the most appropriate therapies.
He said he hopes that the focus on phenotypes will draw attention to differences in these pathophysiological mechanisms. According to Dr. Tan, evaluating rosacea from the perspective of phenotypes has represented an important paradigm shift that extends beyond diagnosis.
“The move to the phenotype approach is hopefully simpler, more accurate, and more relevant,” Dr. Tan said.
This same approach has been advocated by others, including Esther J. van Zurren, MD, professor of dermatology at Leiden University Medical Centre in the Netherlands, the lead author of the 2018 systematic review article discussed by Dr. Tan. In this review article on the phenotype approach, specific strategies were recommended for specific symptoms on the basis of grading by an international group of experts that included Dr. Tan, a coauthor.
“These strategies should be directed toward achieving improvements in general well-being by targeting those aspects most bothersome to the patient,” the article advises. Like Dr. Tan, she considers this phenotype-based approach to diagnosis and treatment to be a meaningful clinical advance over the guidelines published in 2002.
“Management strategies for people with rosacea should include phenotype-based treatments,” she agreed, adding that specific choices should be made on the basis of these phenotypes “instead of the previous subtype classification.”
The meeting was jointly presented by the University of Louisville and Global Academy for Medical Education. This publication and Global Academy for Medical Education are owned by the same parent company.
FROM COASTAL DERM
Phase 1 study: Beta-blocker may improve melanoma treatment response
Response rates were high without dose-limiting toxicities in a small phase 1 study that evaluated the addition of propranolol to pembrolizumab in treatment-naive patients with metastatic melanoma.
“To our knowledge, this effort is the,” wrote the two co-first authors, Shipra Gandhi, MD, and Manu Pandey, MBBS, from the Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and coauthors.
The need for combinations built on anti-PD1 checkpoint inhibitor therapy strategies in metastatic melanoma that safely improve outcomes is underscored by the high (59%) grade 3 or 4 treatment-related adverse event (TRAE) rates when an anti-CTLA4 agent (ipilimumab) was added to an anti-PD-1 agent (nivolumab), they noted. In contrast, a TRAE rate of only 17% has been reported with pembrolizumab monotherapy.
The phase 1b study was stimulated by preclinical, retrospective observations of improved overall survival (OS) in cancer patients treated with beta-blockers. These were preceded by murine melanoma studies showing decreased tumor growth and metastasis with the nonselective beta-blocker propranolol. “Propranolol exerts an antitumor effect,” the authors stated, “by favorably modulating the tumor microenvironment (TME) by decreasing myeloid-derived suppressor cells and increasing CD8+ T-cell and natural killer cells in the TME.” Other research in a melanoma model in chronically-stressed mice has demonstrated synergy between an anti-PD1 antibody and propranolol.
“We know that stress can have a significant negative effect on health, but the extent to which stress may impact the outcome of cancer therapy is not well understood at all,” Dr. Ghandi said in a statement provided by Roswell Park. “We set out to better understand this relationship and to explore its implications for cancer treatment.”
The investigators recruited nine White adults (median age 65 years) with treatment-naive, histologically confirmed unresectable stage III or IV melanoma and Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 to the open-label, single arm, nonrandomized, single-center, dose-finding study. Patients received standard of care intravenous pembrolizumab 200 mg every 3 weeks and, in three groups, propranolol doses of 10 mg, 20 mg, or 30 mg twice a day until 2 years on study or disease progression or the development of dose-limiting toxicities (DLTs). Assessing the safety and efficacy (overall response rate [ORR] within 6 months of starting therapy) of pembrolizumab with the increasing doses of propranolol and selecting the recommended phase 2 dose were the study’s primary objectives.
Objective responses (complete or partial responses) were reported in seven of the nine patients, with partial tumor responses in two patients in the propranolol 10-mg group, two partial responses in the 20-mg group, and three partial responses in the 30-mg group.
While all patients experienced TRAEs, only one was above grade 2. The most commonly reported TRAEs were fatigue, rash and vitiligo, reported in four of the nine patients. Two patients in the 20-mg twice-a-day group discontinued therapy because of TRAEs (hemophagocytic lymphohistiocytosis and labyrinthitis). No DLTs were observed at any of the three dose levels, and no deaths occurred on study treatment.
The authors said that propranolol 30 mg twice a day was chosen as the recommended phase 2 dose, because in combination with pembrolizumab, there were no DLTs, and preliminary antitumor efficacy was observed in all three patients. Also, in all three patients, the investigators observed a trend toward higher CD8+T-cell percentage, higher ratios of CD8+T-cell/ Treg and CD8+T-cell/ polymorphonuclear myeloid-derived suppressor cells. They underscored, however, that the small size and significant heterogeneity in biomarkers made a statistically sound and meaningful interpretation of biomarkers for deciding the phase 2 dose difficult.
“In repurposing propranolol,” Dr. Pandey said in the Roswell statement, “we’ve gained important insights on how to manage stress in people with cancer – who can face dangerously elevated levels of mental and physical stress related to their diagnosis and treatment.”
In an interview, one of the two senior authors, Elizabeth Repasky, PhD, professor of oncology and immunology at Roswell Park, said, “it’s exciting that an extremely inexpensive drug like propranolol that could be used in every country around the world could have an impact on cancer by blocking stress, especially chronic stress.” Her murine research showing that adding propranolol to immunotherapy or radiotherapy or chemotherapy improved tumor growth control provided rationale for the current study.
“The breakthrough in this study is that it reveals the immune system as the best target to look at, and shows that what stress reduction is doing is improving a patient’s immune response to his or her own tumor,” Dr. Repasky said. “The mind/body connection is so important, but we have not had a handle on how to study it,” she added.
Further research funded by Herd of Hope grants at Roswell will look at tumor effects of propranolol and nonpharmacological reducers of chronic stress such as exercise, meditation, yoga, and Tai Chi, with first studies in breast cancer.
The study was funded by Roswell Park, private, and NIH grants. The authors had no disclosures.
SOURCE: Gandhi S et al. Clin Cancer Res. 2020 Oct 30. doi: 10.1158/1078-0432.CCR-20-2381
Response rates were high without dose-limiting toxicities in a small phase 1 study that evaluated the addition of propranolol to pembrolizumab in treatment-naive patients with metastatic melanoma.
“To our knowledge, this effort is the,” wrote the two co-first authors, Shipra Gandhi, MD, and Manu Pandey, MBBS, from the Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and coauthors.
The need for combinations built on anti-PD1 checkpoint inhibitor therapy strategies in metastatic melanoma that safely improve outcomes is underscored by the high (59%) grade 3 or 4 treatment-related adverse event (TRAE) rates when an anti-CTLA4 agent (ipilimumab) was added to an anti-PD-1 agent (nivolumab), they noted. In contrast, a TRAE rate of only 17% has been reported with pembrolizumab monotherapy.
The phase 1b study was stimulated by preclinical, retrospective observations of improved overall survival (OS) in cancer patients treated with beta-blockers. These were preceded by murine melanoma studies showing decreased tumor growth and metastasis with the nonselective beta-blocker propranolol. “Propranolol exerts an antitumor effect,” the authors stated, “by favorably modulating the tumor microenvironment (TME) by decreasing myeloid-derived suppressor cells and increasing CD8+ T-cell and natural killer cells in the TME.” Other research in a melanoma model in chronically-stressed mice has demonstrated synergy between an anti-PD1 antibody and propranolol.
“We know that stress can have a significant negative effect on health, but the extent to which stress may impact the outcome of cancer therapy is not well understood at all,” Dr. Ghandi said in a statement provided by Roswell Park. “We set out to better understand this relationship and to explore its implications for cancer treatment.”
The investigators recruited nine White adults (median age 65 years) with treatment-naive, histologically confirmed unresectable stage III or IV melanoma and Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 to the open-label, single arm, nonrandomized, single-center, dose-finding study. Patients received standard of care intravenous pembrolizumab 200 mg every 3 weeks and, in three groups, propranolol doses of 10 mg, 20 mg, or 30 mg twice a day until 2 years on study or disease progression or the development of dose-limiting toxicities (DLTs). Assessing the safety and efficacy (overall response rate [ORR] within 6 months of starting therapy) of pembrolizumab with the increasing doses of propranolol and selecting the recommended phase 2 dose were the study’s primary objectives.
Objective responses (complete or partial responses) were reported in seven of the nine patients, with partial tumor responses in two patients in the propranolol 10-mg group, two partial responses in the 20-mg group, and three partial responses in the 30-mg group.
While all patients experienced TRAEs, only one was above grade 2. The most commonly reported TRAEs were fatigue, rash and vitiligo, reported in four of the nine patients. Two patients in the 20-mg twice-a-day group discontinued therapy because of TRAEs (hemophagocytic lymphohistiocytosis and labyrinthitis). No DLTs were observed at any of the three dose levels, and no deaths occurred on study treatment.
The authors said that propranolol 30 mg twice a day was chosen as the recommended phase 2 dose, because in combination with pembrolizumab, there were no DLTs, and preliminary antitumor efficacy was observed in all three patients. Also, in all three patients, the investigators observed a trend toward higher CD8+T-cell percentage, higher ratios of CD8+T-cell/ Treg and CD8+T-cell/ polymorphonuclear myeloid-derived suppressor cells. They underscored, however, that the small size and significant heterogeneity in biomarkers made a statistically sound and meaningful interpretation of biomarkers for deciding the phase 2 dose difficult.
“In repurposing propranolol,” Dr. Pandey said in the Roswell statement, “we’ve gained important insights on how to manage stress in people with cancer – who can face dangerously elevated levels of mental and physical stress related to their diagnosis and treatment.”
In an interview, one of the two senior authors, Elizabeth Repasky, PhD, professor of oncology and immunology at Roswell Park, said, “it’s exciting that an extremely inexpensive drug like propranolol that could be used in every country around the world could have an impact on cancer by blocking stress, especially chronic stress.” Her murine research showing that adding propranolol to immunotherapy or radiotherapy or chemotherapy improved tumor growth control provided rationale for the current study.
“The breakthrough in this study is that it reveals the immune system as the best target to look at, and shows that what stress reduction is doing is improving a patient’s immune response to his or her own tumor,” Dr. Repasky said. “The mind/body connection is so important, but we have not had a handle on how to study it,” she added.
Further research funded by Herd of Hope grants at Roswell will look at tumor effects of propranolol and nonpharmacological reducers of chronic stress such as exercise, meditation, yoga, and Tai Chi, with first studies in breast cancer.
The study was funded by Roswell Park, private, and NIH grants. The authors had no disclosures.
SOURCE: Gandhi S et al. Clin Cancer Res. 2020 Oct 30. doi: 10.1158/1078-0432.CCR-20-2381
Response rates were high without dose-limiting toxicities in a small phase 1 study that evaluated the addition of propranolol to pembrolizumab in treatment-naive patients with metastatic melanoma.
“To our knowledge, this effort is the,” wrote the two co-first authors, Shipra Gandhi, MD, and Manu Pandey, MBBS, from the Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., and coauthors.
The need for combinations built on anti-PD1 checkpoint inhibitor therapy strategies in metastatic melanoma that safely improve outcomes is underscored by the high (59%) grade 3 or 4 treatment-related adverse event (TRAE) rates when an anti-CTLA4 agent (ipilimumab) was added to an anti-PD-1 agent (nivolumab), they noted. In contrast, a TRAE rate of only 17% has been reported with pembrolizumab monotherapy.
The phase 1b study was stimulated by preclinical, retrospective observations of improved overall survival (OS) in cancer patients treated with beta-blockers. These were preceded by murine melanoma studies showing decreased tumor growth and metastasis with the nonselective beta-blocker propranolol. “Propranolol exerts an antitumor effect,” the authors stated, “by favorably modulating the tumor microenvironment (TME) by decreasing myeloid-derived suppressor cells and increasing CD8+ T-cell and natural killer cells in the TME.” Other research in a melanoma model in chronically-stressed mice has demonstrated synergy between an anti-PD1 antibody and propranolol.
“We know that stress can have a significant negative effect on health, but the extent to which stress may impact the outcome of cancer therapy is not well understood at all,” Dr. Ghandi said in a statement provided by Roswell Park. “We set out to better understand this relationship and to explore its implications for cancer treatment.”
The investigators recruited nine White adults (median age 65 years) with treatment-naive, histologically confirmed unresectable stage III or IV melanoma and Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 to the open-label, single arm, nonrandomized, single-center, dose-finding study. Patients received standard of care intravenous pembrolizumab 200 mg every 3 weeks and, in three groups, propranolol doses of 10 mg, 20 mg, or 30 mg twice a day until 2 years on study or disease progression or the development of dose-limiting toxicities (DLTs). Assessing the safety and efficacy (overall response rate [ORR] within 6 months of starting therapy) of pembrolizumab with the increasing doses of propranolol and selecting the recommended phase 2 dose were the study’s primary objectives.
Objective responses (complete or partial responses) were reported in seven of the nine patients, with partial tumor responses in two patients in the propranolol 10-mg group, two partial responses in the 20-mg group, and three partial responses in the 30-mg group.
While all patients experienced TRAEs, only one was above grade 2. The most commonly reported TRAEs were fatigue, rash and vitiligo, reported in four of the nine patients. Two patients in the 20-mg twice-a-day group discontinued therapy because of TRAEs (hemophagocytic lymphohistiocytosis and labyrinthitis). No DLTs were observed at any of the three dose levels, and no deaths occurred on study treatment.
The authors said that propranolol 30 mg twice a day was chosen as the recommended phase 2 dose, because in combination with pembrolizumab, there were no DLTs, and preliminary antitumor efficacy was observed in all three patients. Also, in all three patients, the investigators observed a trend toward higher CD8+T-cell percentage, higher ratios of CD8+T-cell/ Treg and CD8+T-cell/ polymorphonuclear myeloid-derived suppressor cells. They underscored, however, that the small size and significant heterogeneity in biomarkers made a statistically sound and meaningful interpretation of biomarkers for deciding the phase 2 dose difficult.
“In repurposing propranolol,” Dr. Pandey said in the Roswell statement, “we’ve gained important insights on how to manage stress in people with cancer – who can face dangerously elevated levels of mental and physical stress related to their diagnosis and treatment.”
In an interview, one of the two senior authors, Elizabeth Repasky, PhD, professor of oncology and immunology at Roswell Park, said, “it’s exciting that an extremely inexpensive drug like propranolol that could be used in every country around the world could have an impact on cancer by blocking stress, especially chronic stress.” Her murine research showing that adding propranolol to immunotherapy or radiotherapy or chemotherapy improved tumor growth control provided rationale for the current study.
“The breakthrough in this study is that it reveals the immune system as the best target to look at, and shows that what stress reduction is doing is improving a patient’s immune response to his or her own tumor,” Dr. Repasky said. “The mind/body connection is so important, but we have not had a handle on how to study it,” she added.
Further research funded by Herd of Hope grants at Roswell will look at tumor effects of propranolol and nonpharmacological reducers of chronic stress such as exercise, meditation, yoga, and Tai Chi, with first studies in breast cancer.
The study was funded by Roswell Park, private, and NIH grants. The authors had no disclosures.
SOURCE: Gandhi S et al. Clin Cancer Res. 2020 Oct 30. doi: 10.1158/1078-0432.CCR-20-2381
FROM CLINICAL CANCER RESEARCH
Painful, lower extremity rash
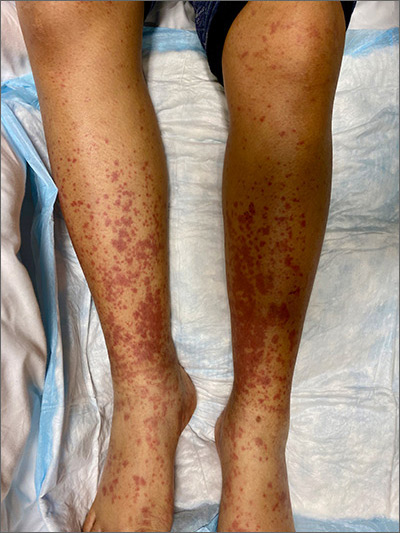
This woman’s palpable purpura with edema in her lower extremities was consistent with cutaneous leukocytoclastic vasculitis (LCV).
LCV is characterized by the circulation of immune complexes that promote activation of complement, leading to endothelial injury and palpable purpura. Pain, arthralgia, cutaneous ulceration, and constitutional symptoms may be observed. About 50% of LCV cases are idiopathic. Identified causes include infection (including syphilis infection), drugs, malignancy, and connective tissue disease.
Systemic involvement must be ruled out in any patient with cutaneous LCV. The work-up is based on the individual patient assessment and may include a complete blood count with differential, complete metabolic panel, inflammatory markers, urinalysis, hepatitis panel, anti-nuclear antibody, rheumatoid factor, anti-neutrophil cytoplasmic antibodies, cryoglobulins, serum protein electrophoresis, and serum complement. A cutaneous punch biopsy for both hematoxylin and eosin (H&E) and direct immunofluorescence (DIF) confirms the diagnosis of LCV.
For uncomplicated LCV cases without systemic involvement, treatment is generally supportive. Any identified underlying cause should be addressed. Analgesics may be considered for pain. Systemic therapy is indicated for patients with cutaneous ulceration, systemic vasculitis, or recurrent cases; this therapy may include colchicine, dapsone, corticosteroids, mycophenolate mofetil, and methotrexate.
In this patient’s case, a punch biopsy of the left lower extremity showed findings consistent with cutaneous LCV. She denied a history of intravenous drug use or initiation of new medications. Labs were notable for an elevated erythrocyte sedimentation rate, c-reactive protein, and elevated creatinine.
Incidentally, she was found to be 32-weeks pregnant, went into pre-term labor while admitted, and delivered her baby without complication.
She had a reactive treponemal antibody, with rapid plasma reagin titer of 1:128, which confirmed a diagnosis of syphilis. She was treated with 1 dose of intra-muscular penicillin G while an inpatient. Her arthralgias improved during her hospitalization without initiation of steroids or other immunomodulatory therapy. Outpatient follow-up was expected to consist of completion of 3 total doses of IM penicillin, as well as renal studies, given her elevated creatinine.
Photo courtesy of Cyrelle R. Fermin, MD, and text courtesy of Cyrelle R. Fermin, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Goeser MR, Laniosz V, Wetter DA. A practical approach to the diagnosis, evaluation, and management of cutaneous small-vessel vasculitis. Am J Clin Dermatol. 2014;15:299-306.

This woman’s palpable purpura with edema in her lower extremities was consistent with cutaneous leukocytoclastic vasculitis (LCV).
LCV is characterized by the circulation of immune complexes that promote activation of complement, leading to endothelial injury and palpable purpura. Pain, arthralgia, cutaneous ulceration, and constitutional symptoms may be observed. About 50% of LCV cases are idiopathic. Identified causes include infection (including syphilis infection), drugs, malignancy, and connective tissue disease.
Systemic involvement must be ruled out in any patient with cutaneous LCV. The work-up is based on the individual patient assessment and may include a complete blood count with differential, complete metabolic panel, inflammatory markers, urinalysis, hepatitis panel, anti-nuclear antibody, rheumatoid factor, anti-neutrophil cytoplasmic antibodies, cryoglobulins, serum protein electrophoresis, and serum complement. A cutaneous punch biopsy for both hematoxylin and eosin (H&E) and direct immunofluorescence (DIF) confirms the diagnosis of LCV.
For uncomplicated LCV cases without systemic involvement, treatment is generally supportive. Any identified underlying cause should be addressed. Analgesics may be considered for pain. Systemic therapy is indicated for patients with cutaneous ulceration, systemic vasculitis, or recurrent cases; this therapy may include colchicine, dapsone, corticosteroids, mycophenolate mofetil, and methotrexate.
In this patient’s case, a punch biopsy of the left lower extremity showed findings consistent with cutaneous LCV. She denied a history of intravenous drug use or initiation of new medications. Labs were notable for an elevated erythrocyte sedimentation rate, c-reactive protein, and elevated creatinine.
Incidentally, she was found to be 32-weeks pregnant, went into pre-term labor while admitted, and delivered her baby without complication.
She had a reactive treponemal antibody, with rapid plasma reagin titer of 1:128, which confirmed a diagnosis of syphilis. She was treated with 1 dose of intra-muscular penicillin G while an inpatient. Her arthralgias improved during her hospitalization without initiation of steroids or other immunomodulatory therapy. Outpatient follow-up was expected to consist of completion of 3 total doses of IM penicillin, as well as renal studies, given her elevated creatinine.
Photo courtesy of Cyrelle R. Fermin, MD, and text courtesy of Cyrelle R. Fermin, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This woman’s palpable purpura with edema in her lower extremities was consistent with cutaneous leukocytoclastic vasculitis (LCV).
LCV is characterized by the circulation of immune complexes that promote activation of complement, leading to endothelial injury and palpable purpura. Pain, arthralgia, cutaneous ulceration, and constitutional symptoms may be observed. About 50% of LCV cases are idiopathic. Identified causes include infection (including syphilis infection), drugs, malignancy, and connective tissue disease.
Systemic involvement must be ruled out in any patient with cutaneous LCV. The work-up is based on the individual patient assessment and may include a complete blood count with differential, complete metabolic panel, inflammatory markers, urinalysis, hepatitis panel, anti-nuclear antibody, rheumatoid factor, anti-neutrophil cytoplasmic antibodies, cryoglobulins, serum protein electrophoresis, and serum complement. A cutaneous punch biopsy for both hematoxylin and eosin (H&E) and direct immunofluorescence (DIF) confirms the diagnosis of LCV.
For uncomplicated LCV cases without systemic involvement, treatment is generally supportive. Any identified underlying cause should be addressed. Analgesics may be considered for pain. Systemic therapy is indicated for patients with cutaneous ulceration, systemic vasculitis, or recurrent cases; this therapy may include colchicine, dapsone, corticosteroids, mycophenolate mofetil, and methotrexate.
In this patient’s case, a punch biopsy of the left lower extremity showed findings consistent with cutaneous LCV. She denied a history of intravenous drug use or initiation of new medications. Labs were notable for an elevated erythrocyte sedimentation rate, c-reactive protein, and elevated creatinine.
Incidentally, she was found to be 32-weeks pregnant, went into pre-term labor while admitted, and delivered her baby without complication.
She had a reactive treponemal antibody, with rapid plasma reagin titer of 1:128, which confirmed a diagnosis of syphilis. She was treated with 1 dose of intra-muscular penicillin G while an inpatient. Her arthralgias improved during her hospitalization without initiation of steroids or other immunomodulatory therapy. Outpatient follow-up was expected to consist of completion of 3 total doses of IM penicillin, as well as renal studies, given her elevated creatinine.
Photo courtesy of Cyrelle R. Fermin, MD, and text courtesy of Cyrelle R. Fermin, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Goeser MR, Laniosz V, Wetter DA. A practical approach to the diagnosis, evaluation, and management of cutaneous small-vessel vasculitis. Am J Clin Dermatol. 2014;15:299-306.
Goeser MR, Laniosz V, Wetter DA. A practical approach to the diagnosis, evaluation, and management of cutaneous small-vessel vasculitis. Am J Clin Dermatol. 2014;15:299-306.
“Polka-dotted” feet
This man has pitted keratolysis (PK), characterized by multiple small pits on the soles of the feet. PK is often associated with hyperhidrosis and significant odor. The lesions usually have a punched-out appearance and are flesh-colored. The dark color of these lesions was due to the patient’s footwear.
PK is caused by bacterial overgrowth in the stratum corneum. Corynebacterium is the most common bacterial culprit, but Kytococcus, Actinomyces, and Dermatophilus have also been implicated. The bacterial infection is thought to be secondary to hyperhidrosis or as a result of hygiene, footwear, or other conditions that retain moisture and promote maceration of the soles of the feet. Therefore, treatment includes a 2-pronged approach: Resolve the bacterial infection and reduce excess moisture. Effective antibacterials include topical clindamycin, erythromycin, fusidic acid, and benzoyl peroxide. Oral antibiotics are not often required.
Hyperhidrosis can be treated with prescription strength 20% aluminum chloride antiperspirant applied to the feet in a tapering schedule, first daily and then 2 or 3 times weekly. Aluminum chloride is frequently not covered by insurance companies, but over-the-counter (OTC) 12% formulations (Certain DRI) usually suffice. Additionally, changing socks and using moisture-wicking shoes or socks are helpful measures to keep feet dry.
One study treated PK with topical erythromycin 3% gel twice daily, without the use of aluminum chloride antiperspirants, and found that the hyperhidrosis greatly improved. The authors theorized that the gram-positive bacterial infection upregulated eccrine sweat glands causing hyperhidrosis as a secondary, rather than the primary, cause of PK.
This patient was prescribed topical erythromycin gel twice daily for the soles of his feet. For his hyperhidrosis, he was advised to purchase OTC aluminum chloride antiperspirants to apply to his feet daily for the first week and to then decrease to 2 or 3 times per week. He was counseled to take an extra pair of socks for changing midway through his workday and to return for reevaluation if his skin did not improve.
Image courtesy of Sarah Friedberg, MD, and text courtesy of Daniel Stulberg, MD, FAAFP, and Sarah Friedberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Pranteda G, Carlesimo M, Pranteda G, et al. Pitted keratolysis, erythromycin, and hyperhidrosis. Dermatol Ther. 2014;27:101-104.

This man has pitted keratolysis (PK), characterized by multiple small pits on the soles of the feet. PK is often associated with hyperhidrosis and significant odor. The lesions usually have a punched-out appearance and are flesh-colored. The dark color of these lesions was due to the patient’s footwear.
PK is caused by bacterial overgrowth in the stratum corneum. Corynebacterium is the most common bacterial culprit, but Kytococcus, Actinomyces, and Dermatophilus have also been implicated. The bacterial infection is thought to be secondary to hyperhidrosis or as a result of hygiene, footwear, or other conditions that retain moisture and promote maceration of the soles of the feet. Therefore, treatment includes a 2-pronged approach: Resolve the bacterial infection and reduce excess moisture. Effective antibacterials include topical clindamycin, erythromycin, fusidic acid, and benzoyl peroxide. Oral antibiotics are not often required.
Hyperhidrosis can be treated with prescription strength 20% aluminum chloride antiperspirant applied to the feet in a tapering schedule, first daily and then 2 or 3 times weekly. Aluminum chloride is frequently not covered by insurance companies, but over-the-counter (OTC) 12% formulations (Certain DRI) usually suffice. Additionally, changing socks and using moisture-wicking shoes or socks are helpful measures to keep feet dry.
One study treated PK with topical erythromycin 3% gel twice daily, without the use of aluminum chloride antiperspirants, and found that the hyperhidrosis greatly improved. The authors theorized that the gram-positive bacterial infection upregulated eccrine sweat glands causing hyperhidrosis as a secondary, rather than the primary, cause of PK.
This patient was prescribed topical erythromycin gel twice daily for the soles of his feet. For his hyperhidrosis, he was advised to purchase OTC aluminum chloride antiperspirants to apply to his feet daily for the first week and to then decrease to 2 or 3 times per week. He was counseled to take an extra pair of socks for changing midway through his workday and to return for reevaluation if his skin did not improve.
Image courtesy of Sarah Friedberg, MD, and text courtesy of Daniel Stulberg, MD, FAAFP, and Sarah Friedberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This man has pitted keratolysis (PK), characterized by multiple small pits on the soles of the feet. PK is often associated with hyperhidrosis and significant odor. The lesions usually have a punched-out appearance and are flesh-colored. The dark color of these lesions was due to the patient’s footwear.
PK is caused by bacterial overgrowth in the stratum corneum. Corynebacterium is the most common bacterial culprit, but Kytococcus, Actinomyces, and Dermatophilus have also been implicated. The bacterial infection is thought to be secondary to hyperhidrosis or as a result of hygiene, footwear, or other conditions that retain moisture and promote maceration of the soles of the feet. Therefore, treatment includes a 2-pronged approach: Resolve the bacterial infection and reduce excess moisture. Effective antibacterials include topical clindamycin, erythromycin, fusidic acid, and benzoyl peroxide. Oral antibiotics are not often required.
Hyperhidrosis can be treated with prescription strength 20% aluminum chloride antiperspirant applied to the feet in a tapering schedule, first daily and then 2 or 3 times weekly. Aluminum chloride is frequently not covered by insurance companies, but over-the-counter (OTC) 12% formulations (Certain DRI) usually suffice. Additionally, changing socks and using moisture-wicking shoes or socks are helpful measures to keep feet dry.
One study treated PK with topical erythromycin 3% gel twice daily, without the use of aluminum chloride antiperspirants, and found that the hyperhidrosis greatly improved. The authors theorized that the gram-positive bacterial infection upregulated eccrine sweat glands causing hyperhidrosis as a secondary, rather than the primary, cause of PK.
This patient was prescribed topical erythromycin gel twice daily for the soles of his feet. For his hyperhidrosis, he was advised to purchase OTC aluminum chloride antiperspirants to apply to his feet daily for the first week and to then decrease to 2 or 3 times per week. He was counseled to take an extra pair of socks for changing midway through his workday and to return for reevaluation if his skin did not improve.
Image courtesy of Sarah Friedberg, MD, and text courtesy of Daniel Stulberg, MD, FAAFP, and Sarah Friedberg, MD, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Pranteda G, Carlesimo M, Pranteda G, et al. Pitted keratolysis, erythromycin, and hyperhidrosis. Dermatol Ther. 2014;27:101-104.
Pranteda G, Carlesimo M, Pranteda G, et al. Pitted keratolysis, erythromycin, and hyperhidrosis. Dermatol Ther. 2014;27:101-104.
Large circular thigh rash
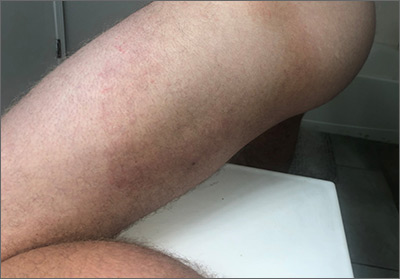
This patient had the deep form of erythema annulare centrifugum (EAC). As the name implies, it manifests as an expanding, red circular pattern that often clears in the middle. There is usually a ring of scale that trails behind the advancing border. However, in the deep form, it may be more subtle than the pronounced scale of the superficial form. Pruritus is a very common symptom associated with this condition.
EAC is a hypersensitivity reaction, which can be in response to several stimuli including underlying malignancy, medications, fungal and dermatophyte infections, inflammatory conditions, and pregnancy. A careful history and physical exam can be helpful in determining if a work-up for malignancy is warranted.
Since many medications including nonsteroidal anti-inflammatory drugs (NSAIDs), antidepressants, and biologicals can cause this condition, a history of which medications were started within the previous several months may be helpful.
When EAC is due to an underlying malignancy, it is called paraneoplastic erythema annulare centrifugum. It can be secondary to solid tumors or lymphoproliferative disorders.
More than 50% percent of the cases are idiopathic, and no underlying condition is identified. The skin findings may last for weeks—and even years.
If an underlying cause is found, treatment is directed at that condition, and the skin findings usually improve with resolution of the instigating condition. If no specific cause is found, the itching can be managed with systemic antihistamines or topical steroids. Some case studies have reported success with the use of systemic antibiotics, including erythromycin. Improvement with antibiotics may be due to treatment of an occult underlying bacterial process or owing to the anti-inflammatory effects of many antibiotics.
Since the patient in this case had onychomycosis of his toenails, and fungal and dermatophyte infections are a common trigger, he was placed on a 12-week course of oral terbinafine 250 mg/d. The plan was to biopsy the rash if it didn’t resolve. At 3 weeks, the rash had resolved, and the patient was asymptomatic.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
McDaniel B, Cook C. Erythema annulare centrifugum. In: Abai B, Abu-Ghosh A, Acharya AB, et al, eds. StatPearls. Treasure Island, FL; 2020. https://www.ncbi.nlm.nih.gov/books/NBK482494/. Accessed December 2, 2020.

This patient had the deep form of erythema annulare centrifugum (EAC). As the name implies, it manifests as an expanding, red circular pattern that often clears in the middle. There is usually a ring of scale that trails behind the advancing border. However, in the deep form, it may be more subtle than the pronounced scale of the superficial form. Pruritus is a very common symptom associated with this condition.
EAC is a hypersensitivity reaction, which can be in response to several stimuli including underlying malignancy, medications, fungal and dermatophyte infections, inflammatory conditions, and pregnancy. A careful history and physical exam can be helpful in determining if a work-up for malignancy is warranted.
Since many medications including nonsteroidal anti-inflammatory drugs (NSAIDs), antidepressants, and biologicals can cause this condition, a history of which medications were started within the previous several months may be helpful.
When EAC is due to an underlying malignancy, it is called paraneoplastic erythema annulare centrifugum. It can be secondary to solid tumors or lymphoproliferative disorders.
More than 50% percent of the cases are idiopathic, and no underlying condition is identified. The skin findings may last for weeks—and even years.
If an underlying cause is found, treatment is directed at that condition, and the skin findings usually improve with resolution of the instigating condition. If no specific cause is found, the itching can be managed with systemic antihistamines or topical steroids. Some case studies have reported success with the use of systemic antibiotics, including erythromycin. Improvement with antibiotics may be due to treatment of an occult underlying bacterial process or owing to the anti-inflammatory effects of many antibiotics.
Since the patient in this case had onychomycosis of his toenails, and fungal and dermatophyte infections are a common trigger, he was placed on a 12-week course of oral terbinafine 250 mg/d. The plan was to biopsy the rash if it didn’t resolve. At 3 weeks, the rash had resolved, and the patient was asymptomatic.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

This patient had the deep form of erythema annulare centrifugum (EAC). As the name implies, it manifests as an expanding, red circular pattern that often clears in the middle. There is usually a ring of scale that trails behind the advancing border. However, in the deep form, it may be more subtle than the pronounced scale of the superficial form. Pruritus is a very common symptom associated with this condition.
EAC is a hypersensitivity reaction, which can be in response to several stimuli including underlying malignancy, medications, fungal and dermatophyte infections, inflammatory conditions, and pregnancy. A careful history and physical exam can be helpful in determining if a work-up for malignancy is warranted.
Since many medications including nonsteroidal anti-inflammatory drugs (NSAIDs), antidepressants, and biologicals can cause this condition, a history of which medications were started within the previous several months may be helpful.
When EAC is due to an underlying malignancy, it is called paraneoplastic erythema annulare centrifugum. It can be secondary to solid tumors or lymphoproliferative disorders.
More than 50% percent of the cases are idiopathic, and no underlying condition is identified. The skin findings may last for weeks—and even years.
If an underlying cause is found, treatment is directed at that condition, and the skin findings usually improve with resolution of the instigating condition. If no specific cause is found, the itching can be managed with systemic antihistamines or topical steroids. Some case studies have reported success with the use of systemic antibiotics, including erythromycin. Improvement with antibiotics may be due to treatment of an occult underlying bacterial process or owing to the anti-inflammatory effects of many antibiotics.
Since the patient in this case had onychomycosis of his toenails, and fungal and dermatophyte infections are a common trigger, he was placed on a 12-week course of oral terbinafine 250 mg/d. The plan was to biopsy the rash if it didn’t resolve. At 3 weeks, the rash had resolved, and the patient was asymptomatic.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
McDaniel B, Cook C. Erythema annulare centrifugum. In: Abai B, Abu-Ghosh A, Acharya AB, et al, eds. StatPearls. Treasure Island, FL; 2020. https://www.ncbi.nlm.nih.gov/books/NBK482494/. Accessed December 2, 2020.
McDaniel B, Cook C. Erythema annulare centrifugum. In: Abai B, Abu-Ghosh A, Acharya AB, et al, eds. StatPearls. Treasure Island, FL; 2020. https://www.ncbi.nlm.nih.gov/books/NBK482494/. Accessed December 2, 2020.