User login
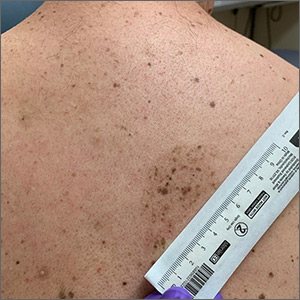
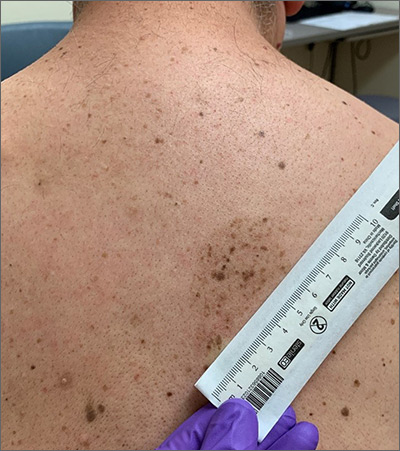
Less pain, same gain with tirbanibulin for actinic keratosis
“with transient local reactions,” according to the results of two identically designed trials.
However, the results, assessed at day 57 and out to 1 year of follow-up, were associated with recurrence of lesions at 1 year, noted lead author Andrew Blauvelt, MD, president of the Oregon Medical Research Center, Portland, and colleagues.
“The incidence of recurrence with conventional treatment has ranged from 20% to 96%,” they noted. “Among patients who had complete clearance at day 57 in the current trials, the estimated incidence of recurrence of previously cleared lesions was 47% at 1 year.” At 1 year, they added, “the estimated incidence of any lesions (new or recurrent) within the application area was 73%” and the estimate of sustained complete clearance was 27%.
A total of 700 adults completed the two multicenter, double-blind, parallel-group, vehicle-controlled trials, conducted concurrently between September 2017 and April 2019 at 62 U.S. sites. The results were published in the New England Journal of Medicine.
To be eligible, patients, mostly White men, had to have four to eight clinically typical, visible, and discrete AK lesions on the face or scalp within a contiguous area measuring 25 cm2. They were randomly assigned to treatment with either tirbanibulin 1% ointment or vehicle ointment (containing monoglycerides, diglycerides, and propylene glycol), which they applied once daily to the entire contiguous area for 5 days.
Pooled data across the two trials showed that the primary outcome, complete clearance of all lesions at day 57, occurred in 49% of the tirbanibulin groups versus 9% of the vehicle groups, and partial clearance (the secondary outcome) occurred in 72% versus 18% respectively. For both outcomes, and in both trials, all results were statistically significant.
Of the 174 patients who received tirbanibulin and had complete clearance, 124 had one or more lesions develop within the application area during follow-up, the authors reported. Of these, 58% had recurrences, while 42% had new lesions.
While individual AK lesions are typically treated with cryosurgery, the study authors noted that treatment of multiple lesions involves topical agents, such as fluorouracil, diclofenac, imiquimod, or ingenol mebutate, and photodynamic therapy, some of which have to be administered over periods of weeks or months and “may be associated with local reactions of pain, irritation, erosions, ulcerations, and irreversible skin changes of pigmentation and scarring,” which may reduce adherence.
In contrast, the current studies showed the most common local reactions to tirbanibulin were erythema in 91% of patients and flaking or scaling in 82%, with transient adverse events including application-site pain in 10% and pruritus in 9%.
“Unlike with most topical treatments for actinic keratosis ... severe local reactions, including vesiculation or pustulation and erosion or ulceration, were infrequent with tirbanibulin ointment,” the authors noted. “This could be due to the relatively short, 5-day course of once-daily treatment.”
They concluded that “larger and longer trials are necessary to determine the effects and risks” of treatment with tirbanibulin for treating AK.
Tirbanibulin, a synthetic inhibitor of tubulin polymerization and Src kinase signaling, was approved by the Food and Drug Administration in December 2020, for the topical treatment of AK of the face or scalp.
Asked to comment on the findings, Neal Bhatia, MD, a dermatologist and researcher at Therapeutics Dermatology, San Diego, who was not involved with the study, said that “a treatment with a 5-day course and excellent tolerability will make dermatologists rethink the old practice of ‘freeze and go.’ ”
In an interview, he added, “tirbanibulin comes to the U.S. market for treating AKs at a great time, as ingenol mebutate has been withdrawn and the others are not widely supported. The mechanism of promoting apoptosis and inducing cell cycle arrest directly correlates to the local skin reaction profile of less crusting, vesiculation, and overall signs of skin necrosis as compared to [5-fluorouracil] and ingenol mebutate, which work via that pathway. As a result, there is a direct impact on the hyperproliferation of atypical keratinocytes that will treat visible and subclinical disease.”
“The ointment vehicle is also novel as previous therapies have been in either creams or gels,” he said.
The two trials were funded by tirbanibulin manufacturer Athenex. Dr. Blauvelt reported receiving consulting fees from Athenex and other pharmaceutical companies, including Almirall, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant Sciences. Other author disclosures included serving as a consultant to Athenex and other companies. Several authors are Athenex employees. Dr. Bhatia disclosed that he is an adviser and consultant for Almirall and has been an investigator for multiple other AK treatments.
“with transient local reactions,” according to the results of two identically designed trials.
However, the results, assessed at day 57 and out to 1 year of follow-up, were associated with recurrence of lesions at 1 year, noted lead author Andrew Blauvelt, MD, president of the Oregon Medical Research Center, Portland, and colleagues.
“The incidence of recurrence with conventional treatment has ranged from 20% to 96%,” they noted. “Among patients who had complete clearance at day 57 in the current trials, the estimated incidence of recurrence of previously cleared lesions was 47% at 1 year.” At 1 year, they added, “the estimated incidence of any lesions (new or recurrent) within the application area was 73%” and the estimate of sustained complete clearance was 27%.
A total of 700 adults completed the two multicenter, double-blind, parallel-group, vehicle-controlled trials, conducted concurrently between September 2017 and April 2019 at 62 U.S. sites. The results were published in the New England Journal of Medicine.
To be eligible, patients, mostly White men, had to have four to eight clinically typical, visible, and discrete AK lesions on the face or scalp within a contiguous area measuring 25 cm2. They were randomly assigned to treatment with either tirbanibulin 1% ointment or vehicle ointment (containing monoglycerides, diglycerides, and propylene glycol), which they applied once daily to the entire contiguous area for 5 days.
Pooled data across the two trials showed that the primary outcome, complete clearance of all lesions at day 57, occurred in 49% of the tirbanibulin groups versus 9% of the vehicle groups, and partial clearance (the secondary outcome) occurred in 72% versus 18% respectively. For both outcomes, and in both trials, all results were statistically significant.
Of the 174 patients who received tirbanibulin and had complete clearance, 124 had one or more lesions develop within the application area during follow-up, the authors reported. Of these, 58% had recurrences, while 42% had new lesions.
While individual AK lesions are typically treated with cryosurgery, the study authors noted that treatment of multiple lesions involves topical agents, such as fluorouracil, diclofenac, imiquimod, or ingenol mebutate, and photodynamic therapy, some of which have to be administered over periods of weeks or months and “may be associated with local reactions of pain, irritation, erosions, ulcerations, and irreversible skin changes of pigmentation and scarring,” which may reduce adherence.
In contrast, the current studies showed the most common local reactions to tirbanibulin were erythema in 91% of patients and flaking or scaling in 82%, with transient adverse events including application-site pain in 10% and pruritus in 9%.
“Unlike with most topical treatments for actinic keratosis ... severe local reactions, including vesiculation or pustulation and erosion or ulceration, were infrequent with tirbanibulin ointment,” the authors noted. “This could be due to the relatively short, 5-day course of once-daily treatment.”
They concluded that “larger and longer trials are necessary to determine the effects and risks” of treatment with tirbanibulin for treating AK.
Tirbanibulin, a synthetic inhibitor of tubulin polymerization and Src kinase signaling, was approved by the Food and Drug Administration in December 2020, for the topical treatment of AK of the face or scalp.
Asked to comment on the findings, Neal Bhatia, MD, a dermatologist and researcher at Therapeutics Dermatology, San Diego, who was not involved with the study, said that “a treatment with a 5-day course and excellent tolerability will make dermatologists rethink the old practice of ‘freeze and go.’ ”
In an interview, he added, “tirbanibulin comes to the U.S. market for treating AKs at a great time, as ingenol mebutate has been withdrawn and the others are not widely supported. The mechanism of promoting apoptosis and inducing cell cycle arrest directly correlates to the local skin reaction profile of less crusting, vesiculation, and overall signs of skin necrosis as compared to [5-fluorouracil] and ingenol mebutate, which work via that pathway. As a result, there is a direct impact on the hyperproliferation of atypical keratinocytes that will treat visible and subclinical disease.”
“The ointment vehicle is also novel as previous therapies have been in either creams or gels,” he said.
The two trials were funded by tirbanibulin manufacturer Athenex. Dr. Blauvelt reported receiving consulting fees from Athenex and other pharmaceutical companies, including Almirall, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant Sciences. Other author disclosures included serving as a consultant to Athenex and other companies. Several authors are Athenex employees. Dr. Bhatia disclosed that he is an adviser and consultant for Almirall and has been an investigator for multiple other AK treatments.
“with transient local reactions,” according to the results of two identically designed trials.
However, the results, assessed at day 57 and out to 1 year of follow-up, were associated with recurrence of lesions at 1 year, noted lead author Andrew Blauvelt, MD, president of the Oregon Medical Research Center, Portland, and colleagues.
“The incidence of recurrence with conventional treatment has ranged from 20% to 96%,” they noted. “Among patients who had complete clearance at day 57 in the current trials, the estimated incidence of recurrence of previously cleared lesions was 47% at 1 year.” At 1 year, they added, “the estimated incidence of any lesions (new or recurrent) within the application area was 73%” and the estimate of sustained complete clearance was 27%.
A total of 700 adults completed the two multicenter, double-blind, parallel-group, vehicle-controlled trials, conducted concurrently between September 2017 and April 2019 at 62 U.S. sites. The results were published in the New England Journal of Medicine.
To be eligible, patients, mostly White men, had to have four to eight clinically typical, visible, and discrete AK lesions on the face or scalp within a contiguous area measuring 25 cm2. They were randomly assigned to treatment with either tirbanibulin 1% ointment or vehicle ointment (containing monoglycerides, diglycerides, and propylene glycol), which they applied once daily to the entire contiguous area for 5 days.
Pooled data across the two trials showed that the primary outcome, complete clearance of all lesions at day 57, occurred in 49% of the tirbanibulin groups versus 9% of the vehicle groups, and partial clearance (the secondary outcome) occurred in 72% versus 18% respectively. For both outcomes, and in both trials, all results were statistically significant.
Of the 174 patients who received tirbanibulin and had complete clearance, 124 had one or more lesions develop within the application area during follow-up, the authors reported. Of these, 58% had recurrences, while 42% had new lesions.
While individual AK lesions are typically treated with cryosurgery, the study authors noted that treatment of multiple lesions involves topical agents, such as fluorouracil, diclofenac, imiquimod, or ingenol mebutate, and photodynamic therapy, some of which have to be administered over periods of weeks or months and “may be associated with local reactions of pain, irritation, erosions, ulcerations, and irreversible skin changes of pigmentation and scarring,” which may reduce adherence.
In contrast, the current studies showed the most common local reactions to tirbanibulin were erythema in 91% of patients and flaking or scaling in 82%, with transient adverse events including application-site pain in 10% and pruritus in 9%.
“Unlike with most topical treatments for actinic keratosis ... severe local reactions, including vesiculation or pustulation and erosion or ulceration, were infrequent with tirbanibulin ointment,” the authors noted. “This could be due to the relatively short, 5-day course of once-daily treatment.”
They concluded that “larger and longer trials are necessary to determine the effects and risks” of treatment with tirbanibulin for treating AK.
Tirbanibulin, a synthetic inhibitor of tubulin polymerization and Src kinase signaling, was approved by the Food and Drug Administration in December 2020, for the topical treatment of AK of the face or scalp.
Asked to comment on the findings, Neal Bhatia, MD, a dermatologist and researcher at Therapeutics Dermatology, San Diego, who was not involved with the study, said that “a treatment with a 5-day course and excellent tolerability will make dermatologists rethink the old practice of ‘freeze and go.’ ”
In an interview, he added, “tirbanibulin comes to the U.S. market for treating AKs at a great time, as ingenol mebutate has been withdrawn and the others are not widely supported. The mechanism of promoting apoptosis and inducing cell cycle arrest directly correlates to the local skin reaction profile of less crusting, vesiculation, and overall signs of skin necrosis as compared to [5-fluorouracil] and ingenol mebutate, which work via that pathway. As a result, there is a direct impact on the hyperproliferation of atypical keratinocytes that will treat visible and subclinical disease.”
“The ointment vehicle is also novel as previous therapies have been in either creams or gels,” he said.
The two trials were funded by tirbanibulin manufacturer Athenex. Dr. Blauvelt reported receiving consulting fees from Athenex and other pharmaceutical companies, including Almirall, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant Sciences. Other author disclosures included serving as a consultant to Athenex and other companies. Several authors are Athenex employees. Dr. Bhatia disclosed that he is an adviser and consultant for Almirall and has been an investigator for multiple other AK treatments.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
ASDSA warns of rogue insulin pen use for DIY fillers
.
In the safety warning, issued on Feb. 18, the ASDSA reported that ASDSA members, all board-certified dermatologists, have seen evidence online of young people using so-called “hyaluron pens” to inject hyaluronic acid filler in the epidermal and upper dermal skin.
The pens being used and promoted in social media for do-it-yourself filler injections are medical devices originally developed for insulin injections. “The use of air pressure technology causes these pens to deliver the hyaluronic acid to insert nanoscale molecules of the filler through the skin,” according to the ASDSA statement. Marketing materials state that the pens can be used to create volume and shape in the lips, and to improve the appearance of nasolabial lines, marionette lines, brow lines known as “elevens,” and forehead wrinkles. Claims that the hyaluronic acid only reaches the papillary layer of the dermis, and is therefore safe, do not alleviate the risk of injury in inexperienced hands, the ASDSA statement points out.
“We are concerned about California children falling prey to products that are not appropriate and safe for them to use,” Elan Newland, MD, member of the ASDSA and the California Society for Dermatology and Dermatological Surgery (CalDerm), said in the statement. “The power of social media is very strong, especially for impressionable teenagers. CalDerm supports alerting consumers and regulators of the dangers of these pens,” he said.
“TikTok is proving to be an extremely powerful platform to communicate, entertain, and even educate, which is why many physicians are getting involved and finding success there. Unfortunately, just like the World Wide Web, there is misinformation there and even dangerous lies,” Sandra Lee, MD, who practices in Upland, Calif. (and is also known as “Dr. Pimple Popper”), said in the statement.
“It’s very concerning to see young people posting a How To on injecting their own lips with hyaluronic acid serum using an ‘airgun’ pen, which acts much like a BB gun to push with force the product under the skin,” she added. “So many things can go wrong.”
The ASDSA has contacted the Food and Drug Administration to report these safety concerns. “In addition, the ASDSA is alerting state medical and estheticians’ boards regarding these patient safety concerns and alerting consumers directly about the risks through social media and other education materials,” according to the statement.
.
In the safety warning, issued on Feb. 18, the ASDSA reported that ASDSA members, all board-certified dermatologists, have seen evidence online of young people using so-called “hyaluron pens” to inject hyaluronic acid filler in the epidermal and upper dermal skin.
The pens being used and promoted in social media for do-it-yourself filler injections are medical devices originally developed for insulin injections. “The use of air pressure technology causes these pens to deliver the hyaluronic acid to insert nanoscale molecules of the filler through the skin,” according to the ASDSA statement. Marketing materials state that the pens can be used to create volume and shape in the lips, and to improve the appearance of nasolabial lines, marionette lines, brow lines known as “elevens,” and forehead wrinkles. Claims that the hyaluronic acid only reaches the papillary layer of the dermis, and is therefore safe, do not alleviate the risk of injury in inexperienced hands, the ASDSA statement points out.
“We are concerned about California children falling prey to products that are not appropriate and safe for them to use,” Elan Newland, MD, member of the ASDSA and the California Society for Dermatology and Dermatological Surgery (CalDerm), said in the statement. “The power of social media is very strong, especially for impressionable teenagers. CalDerm supports alerting consumers and regulators of the dangers of these pens,” he said.
“TikTok is proving to be an extremely powerful platform to communicate, entertain, and even educate, which is why many physicians are getting involved and finding success there. Unfortunately, just like the World Wide Web, there is misinformation there and even dangerous lies,” Sandra Lee, MD, who practices in Upland, Calif. (and is also known as “Dr. Pimple Popper”), said in the statement.
“It’s very concerning to see young people posting a How To on injecting their own lips with hyaluronic acid serum using an ‘airgun’ pen, which acts much like a BB gun to push with force the product under the skin,” she added. “So many things can go wrong.”
The ASDSA has contacted the Food and Drug Administration to report these safety concerns. “In addition, the ASDSA is alerting state medical and estheticians’ boards regarding these patient safety concerns and alerting consumers directly about the risks through social media and other education materials,” according to the statement.
.
In the safety warning, issued on Feb. 18, the ASDSA reported that ASDSA members, all board-certified dermatologists, have seen evidence online of young people using so-called “hyaluron pens” to inject hyaluronic acid filler in the epidermal and upper dermal skin.
The pens being used and promoted in social media for do-it-yourself filler injections are medical devices originally developed for insulin injections. “The use of air pressure technology causes these pens to deliver the hyaluronic acid to insert nanoscale molecules of the filler through the skin,” according to the ASDSA statement. Marketing materials state that the pens can be used to create volume and shape in the lips, and to improve the appearance of nasolabial lines, marionette lines, brow lines known as “elevens,” and forehead wrinkles. Claims that the hyaluronic acid only reaches the papillary layer of the dermis, and is therefore safe, do not alleviate the risk of injury in inexperienced hands, the ASDSA statement points out.
“We are concerned about California children falling prey to products that are not appropriate and safe for them to use,” Elan Newland, MD, member of the ASDSA and the California Society for Dermatology and Dermatological Surgery (CalDerm), said in the statement. “The power of social media is very strong, especially for impressionable teenagers. CalDerm supports alerting consumers and regulators of the dangers of these pens,” he said.
“TikTok is proving to be an extremely powerful platform to communicate, entertain, and even educate, which is why many physicians are getting involved and finding success there. Unfortunately, just like the World Wide Web, there is misinformation there and even dangerous lies,” Sandra Lee, MD, who practices in Upland, Calif. (and is also known as “Dr. Pimple Popper”), said in the statement.
“It’s very concerning to see young people posting a How To on injecting their own lips with hyaluronic acid serum using an ‘airgun’ pen, which acts much like a BB gun to push with force the product under the skin,” she added. “So many things can go wrong.”
The ASDSA has contacted the Food and Drug Administration to report these safety concerns. “In addition, the ASDSA is alerting state medical and estheticians’ boards regarding these patient safety concerns and alerting consumers directly about the risks through social media and other education materials,” according to the statement.
Make the Diagnosis - March 2021
Because of the lack of improvement with topical corticosteroids, a skin biopsy was performed from a lesion on the lower back which showed an epidermis with compact hyperkeratosis and a thickened granular layer. Within the dermis, there was a lichenoid infiltrate of lymphocytes with a prominent interface change and rare dyskeratotic keratinocytes consistent with lichen planus.
Lichen planus is an inflammatory condition of the skin seen mainly in the adult population and is rare in children. This condition affects 0.5%-1% of the population, with maybe a higher prevalence in woman with no racial predilection in the adult or pediatric population. Most patients diagnosed are described to be over 40 years of age, but in children, the mean age for presentation is reported between the ages of 7 and 11.8 years.1 Interestingly, most of the published larger studies of lichen planus in children originate from India. In a U.K. study, about 80% of the cases reported were from children of Indian descent, as is our patient; so it is possible that lichen planus may be more prevalent in India.1 In a study based in the United States, cases were more prevalent in African American children.2
The exact cause of this condition is not known but studies have suggested that activated T cells, particularly CD8+, attack and cause apoptosis of the basal keratinocytes.3 There appears to be an up-regulation of Th1 cytokines such as interferon‐gamma, tumor necrosis factor–alpha, interleukin‐1 alpha, IL‐6, and IL‐8, as well as other apoptosis-related molecules.3
Lichen planus has been associated with other systemic conditions especially liver disease (chronic active hepatitis C and primary biliary cirrhosis). Children and adults may also have coexistence of other autoimmune diseases such as autoimmune polyendocrinopathy, myasthenia gravis, autoimmune thyroid disease, vitiligo, and thymoma. Some reports have also found a higher prevalence of atopic dermatitis in children with lichen planus.4
The lesions are typically described as the four “Ps” for pruritic, polygonal, purpuric flat-topped papules, and plaques. The papules of lichen planus have characteristically dry fine white streaks known as Wickham’s striae. The lesions can occur anywhere on the body, but they tend to occur more commonly on the flexures of the forearms, the wrists, ankles, shins, knees, and the torso. The face is rarely affected. In some patients oral, scalp (lichen planopilaris), nails, and rarely conjunctival, genital, and esophageal involvement can occur.2
In histopathology, the lesions are characterized by a wedge-shaped hypergranulosis, marked hyperkeratosis, and irregular sawtooth-like acanthosis of rete ridges on the epidermis. The dermal-epidermal junction typically shows an interstitial dermatitis. Civatte bodies may also be seen. On direct immunofluorescence, IgM-staining of the cytoid bodies in the dermal papilla or peribasilar areas are suggestive of lichen planus.1
The differential diagnosis of lichen planus includes severe lichenified atopic dermatitis, drug-induced lichen planus, graft-versus-host disease, psoriasis, pityriasis rosea, subacute cutaneous lupus, discoid lupus, secondary syphilis, and lichen simplex chronicus. Interestingly, our patient presented with lesions that were not pruritic and more generalized. Compared with eczema, were flexures are commonly affected, our patient’s lesions were localized to the ankles, wrists, extensor knees, and elbows, and no pruritus was reported. Lichenification of skin lesions occurs as a response to chronic scratching as it occurs in atopic dermatitis and lichen simplex chronicus, was considered in our patient, but the lack of pruritus and the more acute presentation made it unlikely.
Lichen planus is considered a self-limiting disease, so treatment is focused on the control of pruritus and to accelerate resolution. The first-line therapy for classic cutaneous lichen planus is the use of potent or superpotent topical corticosteroids for localized disease on the body and extremities and mild to mid-potency for intertriginous areas and the face. Clinical response should be assessed after 2-3 weeks of treatment. For patients with more generalized or recalcitrant disease like our patient, other treatment modalities like phototherapy (narrow-band UVB), a 4- to 6-week course of oral glucocorticoids, or acitretin may be considered. Our patient recently started narrow-band UVB. Other medications that have been reported beneficial for more severe cases include methotrexate, cyclosporine, griseofulvin, hydroxychloroquine, metronidazole, dapsone, and mycophenolate. Recent studies in the adult population have shown apremilast, a phosphodiesterase inhibitor, to be a promising medication for patients with cutaneous lichen planus, though this medication has not been approved yet for use in the pediatric population.5
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Payette MJ et al. Clin Dermatol. 2015 Nov-Dec;33(6):631-43.
2. Walton KE et al. Pediatr Dermatol. 2010;27:34-8.
3. Lehman JS et al. Int J Dermatol. 2009 Jul;48(7):682-94.
4. Laughter D et al. J Am Acad Dermatol. 2000;43:649-55.
5. Paul J et al. J Am Acad Dermatol. 2013 Feb;68(2):255-61.
Because of the lack of improvement with topical corticosteroids, a skin biopsy was performed from a lesion on the lower back which showed an epidermis with compact hyperkeratosis and a thickened granular layer. Within the dermis, there was a lichenoid infiltrate of lymphocytes with a prominent interface change and rare dyskeratotic keratinocytes consistent with lichen planus.
Lichen planus is an inflammatory condition of the skin seen mainly in the adult population and is rare in children. This condition affects 0.5%-1% of the population, with maybe a higher prevalence in woman with no racial predilection in the adult or pediatric population. Most patients diagnosed are described to be over 40 years of age, but in children, the mean age for presentation is reported between the ages of 7 and 11.8 years.1 Interestingly, most of the published larger studies of lichen planus in children originate from India. In a U.K. study, about 80% of the cases reported were from children of Indian descent, as is our patient; so it is possible that lichen planus may be more prevalent in India.1 In a study based in the United States, cases were more prevalent in African American children.2
The exact cause of this condition is not known but studies have suggested that activated T cells, particularly CD8+, attack and cause apoptosis of the basal keratinocytes.3 There appears to be an up-regulation of Th1 cytokines such as interferon‐gamma, tumor necrosis factor–alpha, interleukin‐1 alpha, IL‐6, and IL‐8, as well as other apoptosis-related molecules.3
Lichen planus has been associated with other systemic conditions especially liver disease (chronic active hepatitis C and primary biliary cirrhosis). Children and adults may also have coexistence of other autoimmune diseases such as autoimmune polyendocrinopathy, myasthenia gravis, autoimmune thyroid disease, vitiligo, and thymoma. Some reports have also found a higher prevalence of atopic dermatitis in children with lichen planus.4
The lesions are typically described as the four “Ps” for pruritic, polygonal, purpuric flat-topped papules, and plaques. The papules of lichen planus have characteristically dry fine white streaks known as Wickham’s striae. The lesions can occur anywhere on the body, but they tend to occur more commonly on the flexures of the forearms, the wrists, ankles, shins, knees, and the torso. The face is rarely affected. In some patients oral, scalp (lichen planopilaris), nails, and rarely conjunctival, genital, and esophageal involvement can occur.2
In histopathology, the lesions are characterized by a wedge-shaped hypergranulosis, marked hyperkeratosis, and irregular sawtooth-like acanthosis of rete ridges on the epidermis. The dermal-epidermal junction typically shows an interstitial dermatitis. Civatte bodies may also be seen. On direct immunofluorescence, IgM-staining of the cytoid bodies in the dermal papilla or peribasilar areas are suggestive of lichen planus.1
The differential diagnosis of lichen planus includes severe lichenified atopic dermatitis, drug-induced lichen planus, graft-versus-host disease, psoriasis, pityriasis rosea, subacute cutaneous lupus, discoid lupus, secondary syphilis, and lichen simplex chronicus. Interestingly, our patient presented with lesions that were not pruritic and more generalized. Compared with eczema, were flexures are commonly affected, our patient’s lesions were localized to the ankles, wrists, extensor knees, and elbows, and no pruritus was reported. Lichenification of skin lesions occurs as a response to chronic scratching as it occurs in atopic dermatitis and lichen simplex chronicus, was considered in our patient, but the lack of pruritus and the more acute presentation made it unlikely.
Lichen planus is considered a self-limiting disease, so treatment is focused on the control of pruritus and to accelerate resolution. The first-line therapy for classic cutaneous lichen planus is the use of potent or superpotent topical corticosteroids for localized disease on the body and extremities and mild to mid-potency for intertriginous areas and the face. Clinical response should be assessed after 2-3 weeks of treatment. For patients with more generalized or recalcitrant disease like our patient, other treatment modalities like phototherapy (narrow-band UVB), a 4- to 6-week course of oral glucocorticoids, or acitretin may be considered. Our patient recently started narrow-band UVB. Other medications that have been reported beneficial for more severe cases include methotrexate, cyclosporine, griseofulvin, hydroxychloroquine, metronidazole, dapsone, and mycophenolate. Recent studies in the adult population have shown apremilast, a phosphodiesterase inhibitor, to be a promising medication for patients with cutaneous lichen planus, though this medication has not been approved yet for use in the pediatric population.5
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Payette MJ et al. Clin Dermatol. 2015 Nov-Dec;33(6):631-43.
2. Walton KE et al. Pediatr Dermatol. 2010;27:34-8.
3. Lehman JS et al. Int J Dermatol. 2009 Jul;48(7):682-94.
4. Laughter D et al. J Am Acad Dermatol. 2000;43:649-55.
5. Paul J et al. J Am Acad Dermatol. 2013 Feb;68(2):255-61.
Because of the lack of improvement with topical corticosteroids, a skin biopsy was performed from a lesion on the lower back which showed an epidermis with compact hyperkeratosis and a thickened granular layer. Within the dermis, there was a lichenoid infiltrate of lymphocytes with a prominent interface change and rare dyskeratotic keratinocytes consistent with lichen planus.
Lichen planus is an inflammatory condition of the skin seen mainly in the adult population and is rare in children. This condition affects 0.5%-1% of the population, with maybe a higher prevalence in woman with no racial predilection in the adult or pediatric population. Most patients diagnosed are described to be over 40 years of age, but in children, the mean age for presentation is reported between the ages of 7 and 11.8 years.1 Interestingly, most of the published larger studies of lichen planus in children originate from India. In a U.K. study, about 80% of the cases reported were from children of Indian descent, as is our patient; so it is possible that lichen planus may be more prevalent in India.1 In a study based in the United States, cases were more prevalent in African American children.2
The exact cause of this condition is not known but studies have suggested that activated T cells, particularly CD8+, attack and cause apoptosis of the basal keratinocytes.3 There appears to be an up-regulation of Th1 cytokines such as interferon‐gamma, tumor necrosis factor–alpha, interleukin‐1 alpha, IL‐6, and IL‐8, as well as other apoptosis-related molecules.3
Lichen planus has been associated with other systemic conditions especially liver disease (chronic active hepatitis C and primary biliary cirrhosis). Children and adults may also have coexistence of other autoimmune diseases such as autoimmune polyendocrinopathy, myasthenia gravis, autoimmune thyroid disease, vitiligo, and thymoma. Some reports have also found a higher prevalence of atopic dermatitis in children with lichen planus.4
The lesions are typically described as the four “Ps” for pruritic, polygonal, purpuric flat-topped papules, and plaques. The papules of lichen planus have characteristically dry fine white streaks known as Wickham’s striae. The lesions can occur anywhere on the body, but they tend to occur more commonly on the flexures of the forearms, the wrists, ankles, shins, knees, and the torso. The face is rarely affected. In some patients oral, scalp (lichen planopilaris), nails, and rarely conjunctival, genital, and esophageal involvement can occur.2
In histopathology, the lesions are characterized by a wedge-shaped hypergranulosis, marked hyperkeratosis, and irregular sawtooth-like acanthosis of rete ridges on the epidermis. The dermal-epidermal junction typically shows an interstitial dermatitis. Civatte bodies may also be seen. On direct immunofluorescence, IgM-staining of the cytoid bodies in the dermal papilla or peribasilar areas are suggestive of lichen planus.1
The differential diagnosis of lichen planus includes severe lichenified atopic dermatitis, drug-induced lichen planus, graft-versus-host disease, psoriasis, pityriasis rosea, subacute cutaneous lupus, discoid lupus, secondary syphilis, and lichen simplex chronicus. Interestingly, our patient presented with lesions that were not pruritic and more generalized. Compared with eczema, were flexures are commonly affected, our patient’s lesions were localized to the ankles, wrists, extensor knees, and elbows, and no pruritus was reported. Lichenification of skin lesions occurs as a response to chronic scratching as it occurs in atopic dermatitis and lichen simplex chronicus, was considered in our patient, but the lack of pruritus and the more acute presentation made it unlikely.
Lichen planus is considered a self-limiting disease, so treatment is focused on the control of pruritus and to accelerate resolution. The first-line therapy for classic cutaneous lichen planus is the use of potent or superpotent topical corticosteroids for localized disease on the body and extremities and mild to mid-potency for intertriginous areas and the face. Clinical response should be assessed after 2-3 weeks of treatment. For patients with more generalized or recalcitrant disease like our patient, other treatment modalities like phototherapy (narrow-band UVB), a 4- to 6-week course of oral glucocorticoids, or acitretin may be considered. Our patient recently started narrow-band UVB. Other medications that have been reported beneficial for more severe cases include methotrexate, cyclosporine, griseofulvin, hydroxychloroquine, metronidazole, dapsone, and mycophenolate. Recent studies in the adult population have shown apremilast, a phosphodiesterase inhibitor, to be a promising medication for patients with cutaneous lichen planus, though this medication has not been approved yet for use in the pediatric population.5
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Payette MJ et al. Clin Dermatol. 2015 Nov-Dec;33(6):631-43.
2. Walton KE et al. Pediatr Dermatol. 2010;27:34-8.
3. Lehman JS et al. Int J Dermatol. 2009 Jul;48(7):682-94.
4. Laughter D et al. J Am Acad Dermatol. 2000;43:649-55.
5. Paul J et al. J Am Acad Dermatol. 2013 Feb;68(2):255-61.
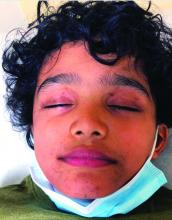
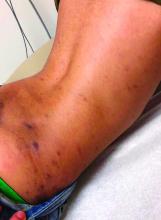
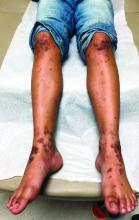
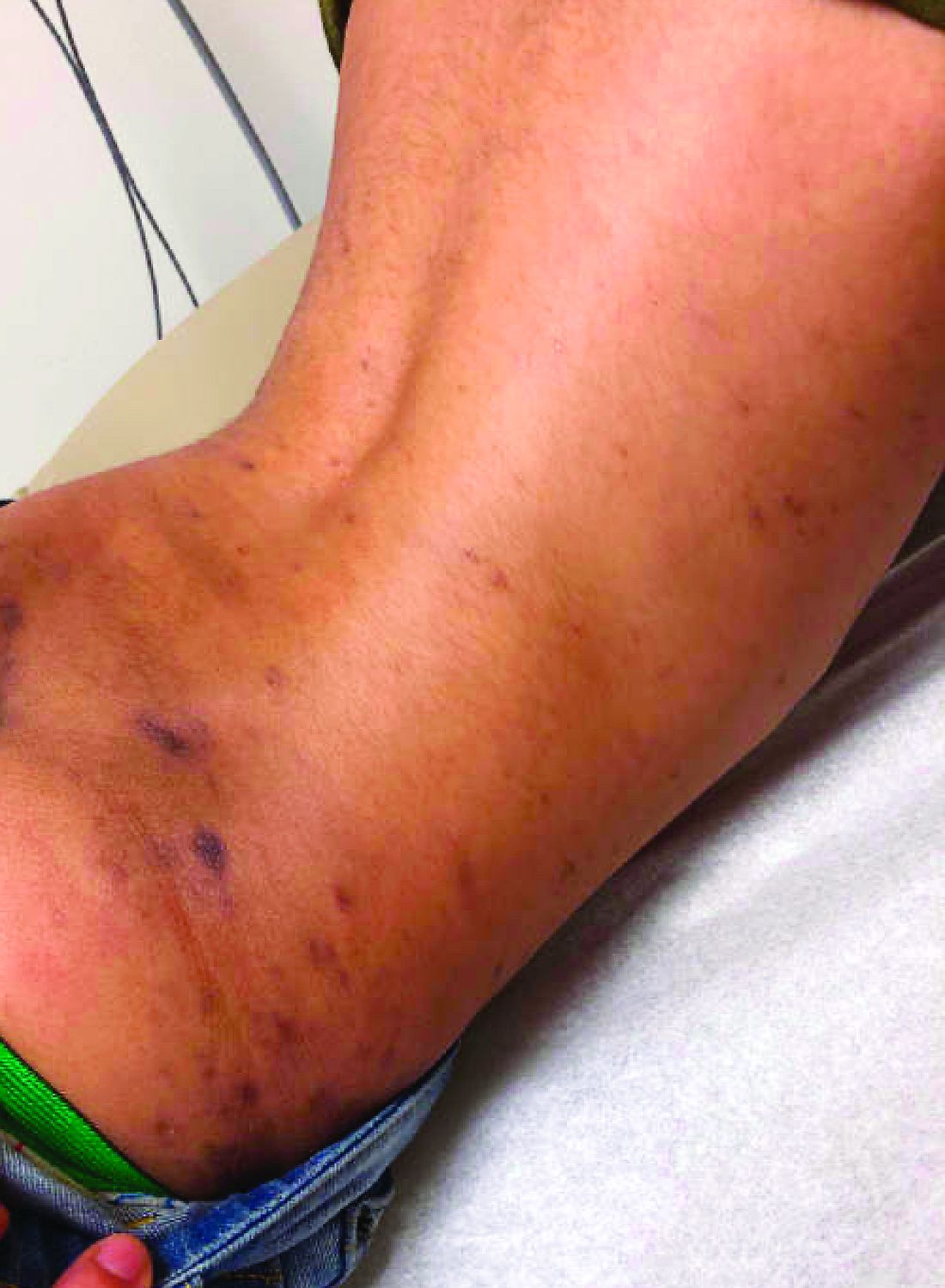
There was no prior personal or family history of atopic dermatitis or psoriasis. He has no other medical conditions and is not taking any medications.
He denied any joint pain, sun sensitivity, mouth sores, or other symptoms. After the initial consultation he was treated with fluocinonide 0.05% ointment for 2 weeks with slight improvement on the lesions.
On physical exam he presented with hyperpigmented and violaceous lichenified papules and plaques on the extremities and the torso. (photos 1 and 2). He also had hyperpigmented violaceous macules on the eyelids and around the mouth (photos 1 and 2).
Cluster of hyperpigmented spots
A large hyperpigmented patch with overlying darker macules and papules is characteristic of a speckled lentiginous nevus (SLN), also called a nevus spilus.
SLN is a cafe-au-lait˗like nevus that initially appears with a hyperpigmented background, usually at or around birth. Later, a speckled or polka-dot pattern of dark macules and papules appears over time. SLN is believed to be a form of congenital melanocytic nevus. There are believed to be 2 subtypes of SLN: nevus spilus maculosus and nevus spilus papulosus.
The maculosus subtype is characterized by flat and evenly distributed macules, that look like polka-dots. Histopathology reveals elongated interpapillary ridges containing increased numbers of melanocytes that form nests at the dermo-epidermal junction.
The papulosus subtype (which this patient had) is differentiated by superimposed speckles and papules whose size and distribution vary; this subype looks similar to a starry sky. Histopathology of the papulosus subtype shows melanocytic nevi of either the dermal or compound type—hence the papular appearance.
Given that SLN is a congenital melanocytic nevus, there is a small risk of transformation to malignant melanoma. The papulosus subtype is believed to have a more dynamic course with more lesions appearing over time. The maculosus subtype is considered to have a slightly higher risk of transformation into malignant melanoma compared to the papulosus subtype.
It is important to recognize that SLN is a distinct clinical entity, rather than a large irregular nevus. Mistaking it for a large suspicious nevus would require multiple biopsies of the most suspicious areas or excision of the entire lesion. Treatment for SLN includes serial surveillance with biopsy or excision of any suspicious areas that arise. In this case, the patient did not have any areas warranting biopsy, so the plan was to have him followed with annual clinical surveillance.
Photo and text courtesy of Erik Unruh, MD, MPH, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Happl, R. Speckled lentiginous naevus: which of the two disorders do you mean? Clin Exp Dermatol. 2009;34:133-135. doi: 10.1111/j.1365-2230.2008.02966.x.

A large hyperpigmented patch with overlying darker macules and papules is characteristic of a speckled lentiginous nevus (SLN), also called a nevus spilus.
SLN is a cafe-au-lait˗like nevus that initially appears with a hyperpigmented background, usually at or around birth. Later, a speckled or polka-dot pattern of dark macules and papules appears over time. SLN is believed to be a form of congenital melanocytic nevus. There are believed to be 2 subtypes of SLN: nevus spilus maculosus and nevus spilus papulosus.
The maculosus subtype is characterized by flat and evenly distributed macules, that look like polka-dots. Histopathology reveals elongated interpapillary ridges containing increased numbers of melanocytes that form nests at the dermo-epidermal junction.
The papulosus subtype (which this patient had) is differentiated by superimposed speckles and papules whose size and distribution vary; this subype looks similar to a starry sky. Histopathology of the papulosus subtype shows melanocytic nevi of either the dermal or compound type—hence the papular appearance.
Given that SLN is a congenital melanocytic nevus, there is a small risk of transformation to malignant melanoma. The papulosus subtype is believed to have a more dynamic course with more lesions appearing over time. The maculosus subtype is considered to have a slightly higher risk of transformation into malignant melanoma compared to the papulosus subtype.
It is important to recognize that SLN is a distinct clinical entity, rather than a large irregular nevus. Mistaking it for a large suspicious nevus would require multiple biopsies of the most suspicious areas or excision of the entire lesion. Treatment for SLN includes serial surveillance with biopsy or excision of any suspicious areas that arise. In this case, the patient did not have any areas warranting biopsy, so the plan was to have him followed with annual clinical surveillance.
Photo and text courtesy of Erik Unruh, MD, MPH, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

A large hyperpigmented patch with overlying darker macules and papules is characteristic of a speckled lentiginous nevus (SLN), also called a nevus spilus.
SLN is a cafe-au-lait˗like nevus that initially appears with a hyperpigmented background, usually at or around birth. Later, a speckled or polka-dot pattern of dark macules and papules appears over time. SLN is believed to be a form of congenital melanocytic nevus. There are believed to be 2 subtypes of SLN: nevus spilus maculosus and nevus spilus papulosus.
The maculosus subtype is characterized by flat and evenly distributed macules, that look like polka-dots. Histopathology reveals elongated interpapillary ridges containing increased numbers of melanocytes that form nests at the dermo-epidermal junction.
The papulosus subtype (which this patient had) is differentiated by superimposed speckles and papules whose size and distribution vary; this subype looks similar to a starry sky. Histopathology of the papulosus subtype shows melanocytic nevi of either the dermal or compound type—hence the papular appearance.
Given that SLN is a congenital melanocytic nevus, there is a small risk of transformation to malignant melanoma. The papulosus subtype is believed to have a more dynamic course with more lesions appearing over time. The maculosus subtype is considered to have a slightly higher risk of transformation into malignant melanoma compared to the papulosus subtype.
It is important to recognize that SLN is a distinct clinical entity, rather than a large irregular nevus. Mistaking it for a large suspicious nevus would require multiple biopsies of the most suspicious areas or excision of the entire lesion. Treatment for SLN includes serial surveillance with biopsy or excision of any suspicious areas that arise. In this case, the patient did not have any areas warranting biopsy, so the plan was to have him followed with annual clinical surveillance.
Photo and text courtesy of Erik Unruh, MD, MPH, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Happl, R. Speckled lentiginous naevus: which of the two disorders do you mean? Clin Exp Dermatol. 2009;34:133-135. doi: 10.1111/j.1365-2230.2008.02966.x.
Happl, R. Speckled lentiginous naevus: which of the two disorders do you mean? Clin Exp Dermatol. 2009;34:133-135. doi: 10.1111/j.1365-2230.2008.02966.x.
Consider home subcutaneous immune globulin for refractory dermatomyositis
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
Home-based subcutaneous immune globulin therapy is a promising alternative to intravenous immune globulin therapy for patients with refractory dermatomyositis or polymyositis, Anna Postolova, MD, MPH, declared at the 2021 Rheumatology Winter Clinical Symposium.
“This is really exciting. I think in the years to come we may see a change to having our patients be able to do immune globulin therapy at home,” said Dr. Postolova, a rheumatologist and allergist/immunologist at Stanford (Calif.) Health Care.
“The technology is there. I think our patients might feel more comfortable getting immune globulin at home,” she said. “I would love to switch more patients from IVIg to SCIg [subcutaneous immune globulin] in my practice.”
A few caveats: SCIg remains off label for treatment of dermatomyositis (DM) or polymyositis (PM). Its approved indication is as replacement therapy in patients with primary or secondary immunodeficiency diseases. IVIg is approved for this indication, but is also approved for DM/PM refractory to high-dose corticosteroids and immunosuppressants. Yet SCIg is clearly effective for these autoimmune inflammatory diseases, albeit to date the supporting evidence comes chiefly from observational studies and anecdotal experience.
“I don’t know if insurers will cover it, but they should because it’s obviously a lot cheaper to do it at home,” she noted.
SCIg advantages
SCIg offers compelling advantages over IVIg in addition to its substantially lower cost. These include far fewer systemic side effects, shorter infusion time, greater bioavailability, and better quality of life. Patients self-administer SCIg at home, avoiding the inconvenience of IVIg therapy, which entails travel time for once-monthly hospitalization or long hours spent in an infusion center, she explained.
French investigators recently documented a previously unappreciated further advantage of home-based SCIg. They convened a focus group of patients with DM or PM experienced with both IVIg and home SCIg and determined that participants uniformly preferred home SCIg. The patients cited a new and welcome feeling of autonomy and control.
“All patients with experience of IVIg and SCIg expressed a clear preference for SCIg, which was described to be easy, less disruptive for daily life, well tolerated, and less time-consuming. Preference was mainly related to a restoration of autonomy. Home-based self-administration reinforced the feeling of independence,” according to the investigators.
Available products
Six preparations of SCIg are commercially available. Most are in 10% concentration, as are all IVIg products. However, a 20% formulation of SCIg known as Hizentra allows for a smaller infusion volume and quicker completion of a treatment session. And one SCIg product, HyQvia, uses recombinant human hyaluronidase-facilitated 10% immune globulin, allowing home infusion of large volumes of sustained-release immune globulin on a once-monthly basis.
The relatively recent introduction of home SCIg for treatment of autoimmune inflammatory diseases, including DM, PM, and chronic inflammatory demyelinating polyneuropathy, has been pioneered mainly by European investigators. The treatment is often given by programmable mechanical pump once weekly. Italian investigators have reported efficacy in DM using 0.2 g/kg per week, which is about half the monthly total dose of IVIg employed. The infusion rate is 10-40 mL/hour, with a volume of around 35 mL per injection site.
Alternatively, SCIg can be delivered by rapid push infusions of smaller volumes with a syringe two or three times per week; that’s the regimen that was used at 2 g/kg over the course of a month by patients in the French focus group study, who didn’t mind the more frequent dosing.
“As they have had severe long-lasting symptoms, SCIg was perceived as a curative rather than a preventive therapy,” according to the French investigators.
More than 40% of patients experience adverse reactions to IVIg. These often involve headaches, nausea, back or abdominal pain, arthralgias, and/or difficulty breathing. Thromboembolic events and acute renal failure occur occasionally. For this reason, many physicians give a prophylactic dose of corticosteroids an hour before a patient’s first dose of IVIg. These systemic side effects are so rare with SCIg that Dr. Postolova has never pretreated with steroids, even though the main reason she resorts to the home therapy is a patient’s track record of poor tolerance of IVIg. The lower abdomen and thigh are the most commonly used subcutaneous infusion sites. Mild local infusion site reactions are fairly common.
Formulating IVIg and SCIg is a complex process that entails plasma procurement and pooling, fractionation, and purification. It takes 10,000-60,000 plasma donations to make one lot of IVIg. Donations are accepted only from repeated donors. Samples are held for 6 months and tested for infectious agents. However, efforts are underway to develop bioengineered recombinant immune globulin products that don’t require donated plasma. These products are being designed to capture and enhance the most important mechanisms of benefit of plasma-derived immunoglobulins using Fc fragments that target key receptors, rather than relying on full-length immune globulin. The goal is enhanced efficacy at much lower doses than with IVIg or SCIg.
Dr. Postolova reported having no financial conflicts regarding her presentation.
FROM RWCS 2021
Checkpoint inhibitors’ ‘big picture’ safety shown with preexisting autoimmune diseases
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
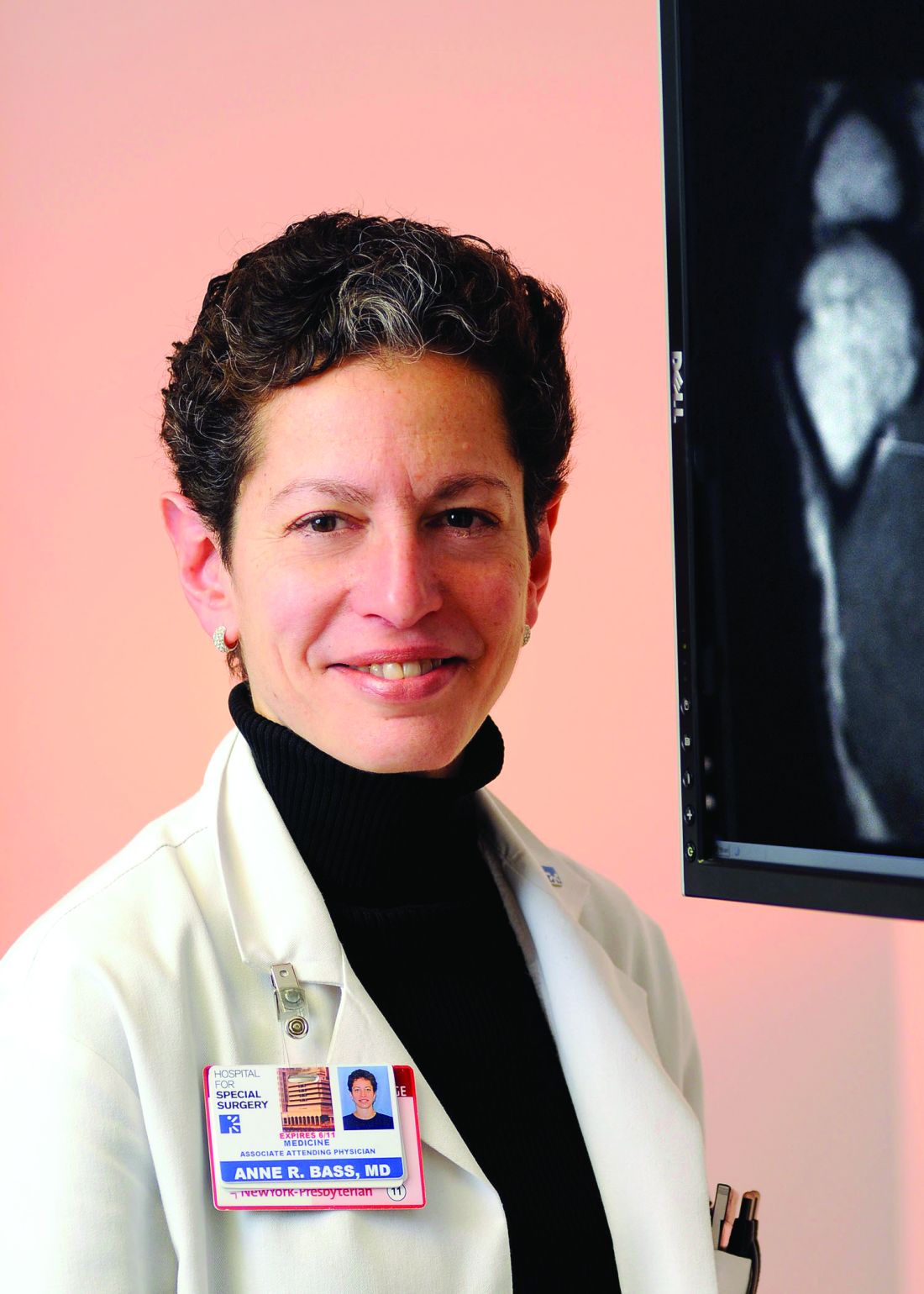
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
Patients with advanced melanoma and preexisting autoimmune diseases (AIDs) who were treated with immune checkpoint inhibitors (ICIs) responded well and did not suffer more grade 3 or higher immune-related adverse events than patients without an AID, a new study finds, although some concerns were raised regarding patients with inflammatory bowel disease (IBD).
“To our knowledge, this is the first study to bridge this knowledge gap by presenting ‘real-world’ data on the safety and efficacy of ICI on a national scale,” wrote Monique K. van der Kooij, MD, of Leiden (the Netherlands) University Medical Center and coauthors. The study was published online in Annals of Internal Medicine.
To investigate ICI use and response among this specific subset of melanoma patients, the researchers launched a nationwide cohort study set in the Netherlands. Data were gathered via the Dutch Melanoma Treatment Registry (DMTR), in which 4,367 patients with advanced melanoma were enrolled between July 2013 and July 2018.
Within that cohort, 415 (9.5%) had preexisting AIDs. Nearly 55% had rheumatologic AIDs (n = 227) – which included RA, systemic lupus erythematosus, scleroderma, sarcoidosis, and vasculitis – with the next most frequent being endocrine AID (n = 143) and IBD (n = 55). Patients with AID were older than patients without (67 vs. 63 years) and were more likely to be female (53% vs. 41%).
The ICIs used in the study included anti-CTLA4 (ipilimumab), anti–programmed death 1 (PD-1) (nivolumab or pembrolizumab), or a combination of nivolumab and ipilimumab. Of the patients with AID, 55% (n = 228) were treated with ICI, compared with 58% of patients without AID. A total of 87 AID patients were treated with anti-CTLA4, 187 received anti-PD-1, and 34 received the combination. The combination was not readily available in the Netherlands until 2017, the authors stated, acknowledging that it may be wise to revisit its effects in the coming years.
Incidence of immune-related adverse events
The incidence of immune-related adverse events (irAEs) grade 3 and above for patients with and without AID who were given anti-CTLA4 was 30%. The incidence rate of irAEs was also similar for patients with (17%; 95% confidence interval, 12%-23%) and without (13%; 95% CI, 12%-15%) AID on anti-PD-1. Patients with AIDs who took anti-PD-1 therapy discontinued it more often because of toxicity than did the patients without AIDs.
The combination group had irAE incidence rates of 44% (95% CI, 27%-62%) for patients with AID, compared with 48% (95% CI, 43%-53%) for patients without AIDs. Overall, no patients with AIDs on ICIs died of toxicity, compared with three deaths among patients without AID on anti-CTLA4, five deaths among patients on anti-PD-1, and one patient on the combination.
Patients with IBD had a notably higher risk of anti-PD-1–induced colitis (19%; 95% CI, 7%-37%), compared with patients with other AIDs (3%; 95% CI, 0%-6%) and patients without AIDs (2%; 95% CI, 2%-3%). IBD patients were also more likely than all other groups on ICIs to stop treatment because of toxicity, leading the researchers to note that “close monitoring in patients with IBD is advised.”
Overall survival after diagnosis was similar in patients with AIDs (median, 13 months; 95% CI, 10-16 months) and without (median, 14 months; 95% CI, 13-15 months), as was the objective response rate to anti-CTLA4 treatment (10% vs. 16%), anti-PD-1 treatment (40% vs. 44%), and combination therapy (39% vs. 43%).
Study largely bypasses the effects of checkpoint inhibitors on RA patients
“For detail, you can’t look to this study,” Anne R. Bass, MD, of the division of rheumatology at the Hospital for Special Surgery in New York, said in an interview. “But for a big-picture look at ‘how safe are checkpoint inhibitors,’ I think it’s an important one.”
Dr. Bass noted that the investigators lumped certain elements together and bypassed others, including their focus on grade 3 or higher adverse events. That was a decision the authors themselves recognized as a potential limitation of their research.
“Understandably, they were worried about life-threatening adverse events, and that’s fine,” she said. But for patients with arthritis who flare, their events are usually grade 2 or even grade 1 and therefore not captured or analyzed in the study. “This does not really address the risk of flare in an RA patient.”
She also questioned their grouping of AIDs, with a bevy of rheumatic diseases categorized as one cluster and the “other” group being particularly broad in its inclusion of “all AIDs not listed” – though only eight patients were placed into that group.
That said, the researchers relied on an oncology database, not one aimed at AID or adverse events. “The numbers are so much bigger than any other study in this area that’s been done,” she said. “It’s both a strength and a weakness of this kind of database.”
Indeed, the authors considered their use of nationwide, population-based data from the DMTR a benefit, calling it “a strength of our approach.”
The DMTR was funded by a grant from the Netherlands Organization for Health Research and Development and sponsored by Bristol-Myers Squibb, Novartis, Roche Nederland, Merck Sharp & Dohme, and Pierre Fabre via the Dutch Institute for Clinical Auditing.
FROM ANNALS OF INTERNAL MEDICINE
Cumulative exposure to high-potency topical steroid doses drives osteoporosis fractures
In support of previously published case reports, in a dose-response relationship.
In a stepwise manner, the hazard ratios for major osteoporotic fracture (MOF) were found to start climbing incrementally for those with a cumulative topical steroid dose equivalent of more than 500 g of mometasone furoate when compared with exposure of 200-499 g, according to the team of investigators from the University of Copenhagen.
“Use of these drugs is very common, and we found an estimated population-attributable risk of as much as 4.3%,” the investigators reported in the study, published in JAMA Dermatology.
The retrospective cohort study drew data from the Danish National Patient Registry, which covers 99% of the country’s population. It was linked to the Danish National Prescription Registry, which captures data on pharmacy-dispensed medications. Data collected from the beginning of 2003 to the end of 2017 were evaluated.
Exposures to potent or very potent topical corticosteroids were converted into a single standard with potency equivalent to 1 mg/g of mometasone furoate. Four strata of exposure were compared to a reference exposure of 200-499 g. These were 500-999 g, 1,000-1,999 g, 2,000-9,999 g, and 10,000 g or greater.
For the first strata, the small increased risk for MOF did not reach significance (HR, 1.01; 95% confidence interval, 0.99-1.03), but each of the others did. These climbed from a 5% greater risk (HR 1.05 95% CI 1.02-1.08) for a cumulative exposure of 1,000 to 1,999 g, to a 10% greater risk (HR, 1.10; 95% CI, 1.07-1.13) for a cumulative exposure of 2,000-9,999 g, and finally to a 27% greater risk (HR, 1.27; 95% CI, 1.19-1.35) for a cumulative exposure of 10,000 g or higher.
The study included more than 700,000 individuals exposed to topical mometasone at a potency equivalent of 200 g or more over the study period. The reference group (200-499 g) was the largest (317,907 individuals). The first strata (500-999 g) included 186,359 patients; the second (1,000-1,999 g), 111,203 patients; the third (2,000-9,999 g), 94,334 patients; and the fifth (10,000 g or more), 13,448 patients.
“A 3% increase in the relative risk of osteoporosis and MOF was observed per doubling of the TCS dose,” according to the investigators.
Patients exposed to doses of high-potency topical steroids that put them at risk of MOF is limited but substantial, according to the senior author, Alexander Egeberg, MD, PhD, of the department of dermatology and allergy at Herlev and Gentofte Hospital, Copenhagen.
“It is true that the risk is modest for the average user of topical steroids,” Dr. Egeberg said in an interview. However, despite the fact that topical steroids are intended for short-term use, “2% of all our users had been exposed to the equivalent of 10,000 g of mometasone, which mean 100 tubes of 100 g.”
If the other two strata at significantly increased risk of MOF (greater than 1,000 g) are included, an additional 28% of all users are facing the potential for clinically significant osteoporosis, according to the Danish data.
The adverse effect of steroids on bone metabolism has been established previously, and several studies have linked systemic corticosteroid exposure, including inhaled corticosteroids, with increased risk of osteoporotic fracture. For example, one study showed that patients with chronic obstructive pulmonary disease on daily inhaled doses of the equivalent of fluticasone at or above 1,000 mcg for more than 4 years had about a 10% increased risk of MOF relative to those not exposed.
The data associate topical steroids with increased risk of osteoporotic fracture, but Dr. Egeberg said osteoporosis is not the only reason to use topical steroids prudently.
“It is important to keep in mind that osteoporosis and fractures are at the extreme end of the side-effect profile and that other side effects, such as striae formation, skin thinning, and dysregulated diabetes, can occur with much lower quantities of topical steroids,” Dr. Egeberg said
For avoiding this risk, “there are no specific cutoffs” recommended for topical steroids in current guidelines, but dermatologists should be aware that many of the indications for topical steroids, such as psoriasis and atopic dermatitis, involve skin with an impaired barrier function, exposing patients to an increased likelihood of absorption, according to Dr. Egeberg.
“A general rule of thumb that we use is that, if a patient with persistent disease activity requires a new prescription of the equivalent of 100 g mometasone every 1-2 months, it might be worth considering if there is a suitable alternative,” Dr. Egeberg said.
In an accompanying editorial, Rebecca D. Jackson, MD, of the division of endocrinology, diabetes, and metabolism in the department of internal medicine at Ohio State University, Columbus, agreed that no guidelines specific to avoiding the risks of topical corticosteroids are currently available, but she advised clinicians to be considering these risks nonetheless. In general, she suggested that topical steroids, like oral steroids, should be used at “the lowest dose for the shortest duration necessary to manage the underlying medical condition.”
The correlation between topical corticosteroids and increased risk of osteoporotic fracture, although not established previously in a large study, is not surprising, according to Victoria Werth, MD, chief of dermatology at the Philadelphia Veterans Affairs Hospital and professor of dermatology at the University of Pennsylvania, also in Philadelphia.
“Systemic absorption of potent topical steroids has previously been demonstrated with a rapid decrease in serum cortisol levels,” Dr. Werth said in an interview. She indicated that concern about the risk of osteoporosis imposed by use of potent steroids over large body surface areas is appropriate.
To minimize this risk, “it is reasonable to use the lowest dose of steroid possible and to try to substitute other medications when possible,” she said.
Dr. Egeberg reported financial relationships with Abbvie, Almirall, Bristol-Myers Squibb, Dermavant Sciences, Galderma, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Samsung, Bioepis, and UCB. Five authors had disclosures related to some of those pharmaceutical companies and/or others. Dr. Jackson had no disclosures.
In support of previously published case reports, in a dose-response relationship.
In a stepwise manner, the hazard ratios for major osteoporotic fracture (MOF) were found to start climbing incrementally for those with a cumulative topical steroid dose equivalent of more than 500 g of mometasone furoate when compared with exposure of 200-499 g, according to the team of investigators from the University of Copenhagen.
“Use of these drugs is very common, and we found an estimated population-attributable risk of as much as 4.3%,” the investigators reported in the study, published in JAMA Dermatology.
The retrospective cohort study drew data from the Danish National Patient Registry, which covers 99% of the country’s population. It was linked to the Danish National Prescription Registry, which captures data on pharmacy-dispensed medications. Data collected from the beginning of 2003 to the end of 2017 were evaluated.
Exposures to potent or very potent topical corticosteroids were converted into a single standard with potency equivalent to 1 mg/g of mometasone furoate. Four strata of exposure were compared to a reference exposure of 200-499 g. These were 500-999 g, 1,000-1,999 g, 2,000-9,999 g, and 10,000 g or greater.
For the first strata, the small increased risk for MOF did not reach significance (HR, 1.01; 95% confidence interval, 0.99-1.03), but each of the others did. These climbed from a 5% greater risk (HR 1.05 95% CI 1.02-1.08) for a cumulative exposure of 1,000 to 1,999 g, to a 10% greater risk (HR, 1.10; 95% CI, 1.07-1.13) for a cumulative exposure of 2,000-9,999 g, and finally to a 27% greater risk (HR, 1.27; 95% CI, 1.19-1.35) for a cumulative exposure of 10,000 g or higher.
The study included more than 700,000 individuals exposed to topical mometasone at a potency equivalent of 200 g or more over the study period. The reference group (200-499 g) was the largest (317,907 individuals). The first strata (500-999 g) included 186,359 patients; the second (1,000-1,999 g), 111,203 patients; the third (2,000-9,999 g), 94,334 patients; and the fifth (10,000 g or more), 13,448 patients.
“A 3% increase in the relative risk of osteoporosis and MOF was observed per doubling of the TCS dose,” according to the investigators.
Patients exposed to doses of high-potency topical steroids that put them at risk of MOF is limited but substantial, according to the senior author, Alexander Egeberg, MD, PhD, of the department of dermatology and allergy at Herlev and Gentofte Hospital, Copenhagen.
“It is true that the risk is modest for the average user of topical steroids,” Dr. Egeberg said in an interview. However, despite the fact that topical steroids are intended for short-term use, “2% of all our users had been exposed to the equivalent of 10,000 g of mometasone, which mean 100 tubes of 100 g.”
If the other two strata at significantly increased risk of MOF (greater than 1,000 g) are included, an additional 28% of all users are facing the potential for clinically significant osteoporosis, according to the Danish data.
The adverse effect of steroids on bone metabolism has been established previously, and several studies have linked systemic corticosteroid exposure, including inhaled corticosteroids, with increased risk of osteoporotic fracture. For example, one study showed that patients with chronic obstructive pulmonary disease on daily inhaled doses of the equivalent of fluticasone at or above 1,000 mcg for more than 4 years had about a 10% increased risk of MOF relative to those not exposed.
The data associate topical steroids with increased risk of osteoporotic fracture, but Dr. Egeberg said osteoporosis is not the only reason to use topical steroids prudently.
“It is important to keep in mind that osteoporosis and fractures are at the extreme end of the side-effect profile and that other side effects, such as striae formation, skin thinning, and dysregulated diabetes, can occur with much lower quantities of topical steroids,” Dr. Egeberg said
For avoiding this risk, “there are no specific cutoffs” recommended for topical steroids in current guidelines, but dermatologists should be aware that many of the indications for topical steroids, such as psoriasis and atopic dermatitis, involve skin with an impaired barrier function, exposing patients to an increased likelihood of absorption, according to Dr. Egeberg.
“A general rule of thumb that we use is that, if a patient with persistent disease activity requires a new prescription of the equivalent of 100 g mometasone every 1-2 months, it might be worth considering if there is a suitable alternative,” Dr. Egeberg said.
In an accompanying editorial, Rebecca D. Jackson, MD, of the division of endocrinology, diabetes, and metabolism in the department of internal medicine at Ohio State University, Columbus, agreed that no guidelines specific to avoiding the risks of topical corticosteroids are currently available, but she advised clinicians to be considering these risks nonetheless. In general, she suggested that topical steroids, like oral steroids, should be used at “the lowest dose for the shortest duration necessary to manage the underlying medical condition.”
The correlation between topical corticosteroids and increased risk of osteoporotic fracture, although not established previously in a large study, is not surprising, according to Victoria Werth, MD, chief of dermatology at the Philadelphia Veterans Affairs Hospital and professor of dermatology at the University of Pennsylvania, also in Philadelphia.
“Systemic absorption of potent topical steroids has previously been demonstrated with a rapid decrease in serum cortisol levels,” Dr. Werth said in an interview. She indicated that concern about the risk of osteoporosis imposed by use of potent steroids over large body surface areas is appropriate.
To minimize this risk, “it is reasonable to use the lowest dose of steroid possible and to try to substitute other medications when possible,” she said.
Dr. Egeberg reported financial relationships with Abbvie, Almirall, Bristol-Myers Squibb, Dermavant Sciences, Galderma, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Samsung, Bioepis, and UCB. Five authors had disclosures related to some of those pharmaceutical companies and/or others. Dr. Jackson had no disclosures.
In support of previously published case reports, in a dose-response relationship.
In a stepwise manner, the hazard ratios for major osteoporotic fracture (MOF) were found to start climbing incrementally for those with a cumulative topical steroid dose equivalent of more than 500 g of mometasone furoate when compared with exposure of 200-499 g, according to the team of investigators from the University of Copenhagen.
“Use of these drugs is very common, and we found an estimated population-attributable risk of as much as 4.3%,” the investigators reported in the study, published in JAMA Dermatology.
The retrospective cohort study drew data from the Danish National Patient Registry, which covers 99% of the country’s population. It was linked to the Danish National Prescription Registry, which captures data on pharmacy-dispensed medications. Data collected from the beginning of 2003 to the end of 2017 were evaluated.
Exposures to potent or very potent topical corticosteroids were converted into a single standard with potency equivalent to 1 mg/g of mometasone furoate. Four strata of exposure were compared to a reference exposure of 200-499 g. These were 500-999 g, 1,000-1,999 g, 2,000-9,999 g, and 10,000 g or greater.
For the first strata, the small increased risk for MOF did not reach significance (HR, 1.01; 95% confidence interval, 0.99-1.03), but each of the others did. These climbed from a 5% greater risk (HR 1.05 95% CI 1.02-1.08) for a cumulative exposure of 1,000 to 1,999 g, to a 10% greater risk (HR, 1.10; 95% CI, 1.07-1.13) for a cumulative exposure of 2,000-9,999 g, and finally to a 27% greater risk (HR, 1.27; 95% CI, 1.19-1.35) for a cumulative exposure of 10,000 g or higher.
The study included more than 700,000 individuals exposed to topical mometasone at a potency equivalent of 200 g or more over the study period. The reference group (200-499 g) was the largest (317,907 individuals). The first strata (500-999 g) included 186,359 patients; the second (1,000-1,999 g), 111,203 patients; the third (2,000-9,999 g), 94,334 patients; and the fifth (10,000 g or more), 13,448 patients.
“A 3% increase in the relative risk of osteoporosis and MOF was observed per doubling of the TCS dose,” according to the investigators.
Patients exposed to doses of high-potency topical steroids that put them at risk of MOF is limited but substantial, according to the senior author, Alexander Egeberg, MD, PhD, of the department of dermatology and allergy at Herlev and Gentofte Hospital, Copenhagen.
“It is true that the risk is modest for the average user of topical steroids,” Dr. Egeberg said in an interview. However, despite the fact that topical steroids are intended for short-term use, “2% of all our users had been exposed to the equivalent of 10,000 g of mometasone, which mean 100 tubes of 100 g.”
If the other two strata at significantly increased risk of MOF (greater than 1,000 g) are included, an additional 28% of all users are facing the potential for clinically significant osteoporosis, according to the Danish data.
The adverse effect of steroids on bone metabolism has been established previously, and several studies have linked systemic corticosteroid exposure, including inhaled corticosteroids, with increased risk of osteoporotic fracture. For example, one study showed that patients with chronic obstructive pulmonary disease on daily inhaled doses of the equivalent of fluticasone at or above 1,000 mcg for more than 4 years had about a 10% increased risk of MOF relative to those not exposed.
The data associate topical steroids with increased risk of osteoporotic fracture, but Dr. Egeberg said osteoporosis is not the only reason to use topical steroids prudently.
“It is important to keep in mind that osteoporosis and fractures are at the extreme end of the side-effect profile and that other side effects, such as striae formation, skin thinning, and dysregulated diabetes, can occur with much lower quantities of topical steroids,” Dr. Egeberg said
For avoiding this risk, “there are no specific cutoffs” recommended for topical steroids in current guidelines, but dermatologists should be aware that many of the indications for topical steroids, such as psoriasis and atopic dermatitis, involve skin with an impaired barrier function, exposing patients to an increased likelihood of absorption, according to Dr. Egeberg.
“A general rule of thumb that we use is that, if a patient with persistent disease activity requires a new prescription of the equivalent of 100 g mometasone every 1-2 months, it might be worth considering if there is a suitable alternative,” Dr. Egeberg said.
In an accompanying editorial, Rebecca D. Jackson, MD, of the division of endocrinology, diabetes, and metabolism in the department of internal medicine at Ohio State University, Columbus, agreed that no guidelines specific to avoiding the risks of topical corticosteroids are currently available, but she advised clinicians to be considering these risks nonetheless. In general, she suggested that topical steroids, like oral steroids, should be used at “the lowest dose for the shortest duration necessary to manage the underlying medical condition.”
The correlation between topical corticosteroids and increased risk of osteoporotic fracture, although not established previously in a large study, is not surprising, according to Victoria Werth, MD, chief of dermatology at the Philadelphia Veterans Affairs Hospital and professor of dermatology at the University of Pennsylvania, also in Philadelphia.
“Systemic absorption of potent topical steroids has previously been demonstrated with a rapid decrease in serum cortisol levels,” Dr. Werth said in an interview. She indicated that concern about the risk of osteoporosis imposed by use of potent steroids over large body surface areas is appropriate.
To minimize this risk, “it is reasonable to use the lowest dose of steroid possible and to try to substitute other medications when possible,” she said.
Dr. Egeberg reported financial relationships with Abbvie, Almirall, Bristol-Myers Squibb, Dermavant Sciences, Galderma, Janssen Pharmaceuticals, Eli Lilly, Novartis, Pfizer, Samsung, Bioepis, and UCB. Five authors had disclosures related to some of those pharmaceutical companies and/or others. Dr. Jackson had no disclosures.
FROM JAMA DERMATOLOGY
Psoriasis registry study finds normal pregnancy outcomes
according to one of the largest studies to examine the issue to date.
However, “pregnancy-specific registries that include a larger number of pregnant women with psoriasis ... are needed to more fully characterize the association between psoriasis and treatment and birth outcomes,” acknowledged first author Alexa B. Kimball, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues.
The cohort study, published in JAMA Dermatology, used data from the Psoriasis Longitudinal Assessment and Registry (PSOLAR), which “is not a pregnancy specific registry, and medical history is captured only at baseline,” they noted.
Their findings showed pregnancy outcomes such as spontaneous abortion, neonatal problems, and congenital anomalies among women with moderate to severe psoriasis were similar to rates in the general U.S. population, and are “consistent with previously reported data,” they reported. “And pregnancy outcomes for women exposed to biologics were similar to those for women with exposure to nonbiologics.”
The study “provides further reassurance that the biologics appear safe at least related to pregnancy outcomes,” commented Jenny Murase, MD, associate professor of dermatology at the University of California, San Francisco, who was not involved in the study. In an interview, she noted that the study “did not examine any potential immunosuppression of the fetus in the first 6 months of life,” which she described as “the heart of the concern, more than whether or not the psoriasis or the biologic affects the pregnancy itself.”
The study used data from the PSOLAR registry collected from June 20, 2007, to Aug.23, 2019, which included 2,224 women of childbearing age (18-45 years) who were collectively followed up for 12,929 patient-years. Among these women, 220 had 298 pregnancies, with 244 live births (81.9%).
“Birth outcomes among all 244 births included 231 healthy newborns (94.7%), 10 infants with a neonatal problem (4.1%), 1 stillbirth (0.4%), and 2 congenital anomalies (0.8%),” the authors reported.
There were also 41 spontaneous abortions (13.8%), and 13 elective terminations (4.4%). “No elective terminations were known to derive from a congenital anomaly or other medical issue,” they added.
Among the documented pregnancies, 252 occurred in women with exposure to biologic therapy either before or during pregnancy, including 168 (56.4%) during the prenatal period, while 46 pregnancies occurred in women with no exposure to biologic therapy.
Dr. Murase, director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif., said that a more detailed comparison of the different psoriasis treatments, as well as the offspring outcomes during the first 6 months of life, might offer some further important insight,.
Infants born after exposure to infliximab “and potentially other anti–tumor necrosis factor–alpha agents during the third trimester may be unable to develop an appropriate immune response to live vaccines,” she and her coauthors cautioned in a letter published in 2011, which referred to a case of an infant with disseminated bacillus Calmette-Guérin infection, whose mother had received infliximab for Crohn’s disease throughout pregnancy.
Dr. Murase pointed out that, in the registry study, exposures to certolizumab, which is pegylated and does not cross the placental barrier, were not separated from other cases. It is important to consider “the cross over late in the second trimester and especially third trimester as the infant is getting the ‘antibody boost’ from the mother as it gets ready to set foot in this world and needs the maternal antibodies to prepare its immune system. If the IgG biologics cross third trimester and immunosuppress the infant ... then I think a medication that does not cross the placental barrier is important to consider.”
The study was sponsored by Janssen Scientific Affairs. Dr. Kimball’s disclosures included serving as a consultant and investigator for companies that included AbbVie, Bristol-Myers Squibb, and Janssen; several other authors also had disclosures related to multiple pharmaceutical companies. Dr. Murase’s disclosures included serving as a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron.
according to one of the largest studies to examine the issue to date.
However, “pregnancy-specific registries that include a larger number of pregnant women with psoriasis ... are needed to more fully characterize the association between psoriasis and treatment and birth outcomes,” acknowledged first author Alexa B. Kimball, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues.
The cohort study, published in JAMA Dermatology, used data from the Psoriasis Longitudinal Assessment and Registry (PSOLAR), which “is not a pregnancy specific registry, and medical history is captured only at baseline,” they noted.
Their findings showed pregnancy outcomes such as spontaneous abortion, neonatal problems, and congenital anomalies among women with moderate to severe psoriasis were similar to rates in the general U.S. population, and are “consistent with previously reported data,” they reported. “And pregnancy outcomes for women exposed to biologics were similar to those for women with exposure to nonbiologics.”
The study “provides further reassurance that the biologics appear safe at least related to pregnancy outcomes,” commented Jenny Murase, MD, associate professor of dermatology at the University of California, San Francisco, who was not involved in the study. In an interview, she noted that the study “did not examine any potential immunosuppression of the fetus in the first 6 months of life,” which she described as “the heart of the concern, more than whether or not the psoriasis or the biologic affects the pregnancy itself.”
The study used data from the PSOLAR registry collected from June 20, 2007, to Aug.23, 2019, which included 2,224 women of childbearing age (18-45 years) who were collectively followed up for 12,929 patient-years. Among these women, 220 had 298 pregnancies, with 244 live births (81.9%).
“Birth outcomes among all 244 births included 231 healthy newborns (94.7%), 10 infants with a neonatal problem (4.1%), 1 stillbirth (0.4%), and 2 congenital anomalies (0.8%),” the authors reported.
There were also 41 spontaneous abortions (13.8%), and 13 elective terminations (4.4%). “No elective terminations were known to derive from a congenital anomaly or other medical issue,” they added.
Among the documented pregnancies, 252 occurred in women with exposure to biologic therapy either before or during pregnancy, including 168 (56.4%) during the prenatal period, while 46 pregnancies occurred in women with no exposure to biologic therapy.
Dr. Murase, director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif., said that a more detailed comparison of the different psoriasis treatments, as well as the offspring outcomes during the first 6 months of life, might offer some further important insight,.
Infants born after exposure to infliximab “and potentially other anti–tumor necrosis factor–alpha agents during the third trimester may be unable to develop an appropriate immune response to live vaccines,” she and her coauthors cautioned in a letter published in 2011, which referred to a case of an infant with disseminated bacillus Calmette-Guérin infection, whose mother had received infliximab for Crohn’s disease throughout pregnancy.
Dr. Murase pointed out that, in the registry study, exposures to certolizumab, which is pegylated and does not cross the placental barrier, were not separated from other cases. It is important to consider “the cross over late in the second trimester and especially third trimester as the infant is getting the ‘antibody boost’ from the mother as it gets ready to set foot in this world and needs the maternal antibodies to prepare its immune system. If the IgG biologics cross third trimester and immunosuppress the infant ... then I think a medication that does not cross the placental barrier is important to consider.”
The study was sponsored by Janssen Scientific Affairs. Dr. Kimball’s disclosures included serving as a consultant and investigator for companies that included AbbVie, Bristol-Myers Squibb, and Janssen; several other authors also had disclosures related to multiple pharmaceutical companies. Dr. Murase’s disclosures included serving as a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron.
according to one of the largest studies to examine the issue to date.
However, “pregnancy-specific registries that include a larger number of pregnant women with psoriasis ... are needed to more fully characterize the association between psoriasis and treatment and birth outcomes,” acknowledged first author Alexa B. Kimball, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues.
The cohort study, published in JAMA Dermatology, used data from the Psoriasis Longitudinal Assessment and Registry (PSOLAR), which “is not a pregnancy specific registry, and medical history is captured only at baseline,” they noted.
Their findings showed pregnancy outcomes such as spontaneous abortion, neonatal problems, and congenital anomalies among women with moderate to severe psoriasis were similar to rates in the general U.S. population, and are “consistent with previously reported data,” they reported. “And pregnancy outcomes for women exposed to biologics were similar to those for women with exposure to nonbiologics.”
The study “provides further reassurance that the biologics appear safe at least related to pregnancy outcomes,” commented Jenny Murase, MD, associate professor of dermatology at the University of California, San Francisco, who was not involved in the study. In an interview, she noted that the study “did not examine any potential immunosuppression of the fetus in the first 6 months of life,” which she described as “the heart of the concern, more than whether or not the psoriasis or the biologic affects the pregnancy itself.”
The study used data from the PSOLAR registry collected from June 20, 2007, to Aug.23, 2019, which included 2,224 women of childbearing age (18-45 years) who were collectively followed up for 12,929 patient-years. Among these women, 220 had 298 pregnancies, with 244 live births (81.9%).
“Birth outcomes among all 244 births included 231 healthy newborns (94.7%), 10 infants with a neonatal problem (4.1%), 1 stillbirth (0.4%), and 2 congenital anomalies (0.8%),” the authors reported.
There were also 41 spontaneous abortions (13.8%), and 13 elective terminations (4.4%). “No elective terminations were known to derive from a congenital anomaly or other medical issue,” they added.
Among the documented pregnancies, 252 occurred in women with exposure to biologic therapy either before or during pregnancy, including 168 (56.4%) during the prenatal period, while 46 pregnancies occurred in women with no exposure to biologic therapy.
Dr. Murase, director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif., said that a more detailed comparison of the different psoriasis treatments, as well as the offspring outcomes during the first 6 months of life, might offer some further important insight,.
Infants born after exposure to infliximab “and potentially other anti–tumor necrosis factor–alpha agents during the third trimester may be unable to develop an appropriate immune response to live vaccines,” she and her coauthors cautioned in a letter published in 2011, which referred to a case of an infant with disseminated bacillus Calmette-Guérin infection, whose mother had received infliximab for Crohn’s disease throughout pregnancy.
Dr. Murase pointed out that, in the registry study, exposures to certolizumab, which is pegylated and does not cross the placental barrier, were not separated from other cases. It is important to consider “the cross over late in the second trimester and especially third trimester as the infant is getting the ‘antibody boost’ from the mother as it gets ready to set foot in this world and needs the maternal antibodies to prepare its immune system. If the IgG biologics cross third trimester and immunosuppress the infant ... then I think a medication that does not cross the placental barrier is important to consider.”
The study was sponsored by Janssen Scientific Affairs. Dr. Kimball’s disclosures included serving as a consultant and investigator for companies that included AbbVie, Bristol-Myers Squibb, and Janssen; several other authors also had disclosures related to multiple pharmaceutical companies. Dr. Murase’s disclosures included serving as a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron.
FROM JAMA DERMATOLOGY
Expert calls for paradigm shift in lab monitoring of some dermatology drugs
From time to time, Joslyn Kirby, MD, asks other physicians about their experience with certain medications used in dermatology, especially when something new hits the market.
“Sometimes I get an answer like, ‘The last time I used that medicine, my patient needed a liver transplant,’ ” Dr. Kirby, associate professor of dermatology, Penn State University, Hershey, said during the Orlando Dermatology Aesthetic and Clinical Conference. “It’s typically a story of something rare, uncommon, and awful. The challenge with an anecdote is that for all its power, it has a lower level of evidence. But it sticks with us and influences us more than a better level of evidence because it’s a situation and a story that we might relate to.”
Dr. Kirby said that when she thinks about managing side effects from drugs used in dermatology, it usually relates to something common and low-risk such as sore, dry skin with isotretinoin use. In contrast, if there is an uncommon but serious side effect, then mitigation rather than management is key. “I want to mitigate the risk – meaning warn my patient about it or be careful about how I select my patients when it is a serious side effect that happens infrequently,” she said. “The worst combination is a frequent and severe side effect. That is something we should avoid, for sure.”
Isotretinoin
But another aspect of prescribing a new drug for patients can be less clear-cut, Dr. Kirby continued, such as the rationale for routine lab monitoring. She began by discussing one of her male patients with moderate to severe acne. After he failed oral antibiotics and topical retinoids, she recommended isotretinoin, which carries a risk of hypertriglyceridemia-associated pancreatitis. “Early in my career, I was getting a lot of monthly labs in patients on this drug that were totally normal and not influencing my practice,” Dr. Kirby recalled. “We’ve seen studies coming out on isotretinoin lab monitoring, showing us that we can keep our patients safe and that we really don’t need to be checking labs as often, because lab changes are infrequent.”
In one of those studies, researchers evaluated 1,863 patients treated with isotretinoin for acne between Jan. 1, 2008, and June 30, 2017 (J Am Acad Dermatol. 2020 Jan;82[1]:72-9).Over time, fewer than 1% of patients screened developed grade 3 or greater triglyceride testing abnormalities, while fewer than 0.5% developed liver function testing (LFT) abnormalities. Authors of a separate systematic review concluded that for patients on isotretinoin therapy without elevated baseline triglycerides, or risk thereof, monitoring triglycerides is of little value (Br J Dermatol. 2017 Oct;177[4]:960-6). Of the 25 patients in the analysis who developed pancreatitis on isotretinoin, only 3 had elevated triglycerides at baseline.
“I was taught that I need to check triglycerides frequently due to the risk of pancreatitis developing with isotretinoin use,” Dr. Kirby said. “Lipid changes on therapy are expected, but they tend to peak early, meaning the first 3 months of treatment when we’re ramping up from a starting dose to a maintenance dose. It’s rare for somebody to be a late bloomer, meaning that they have totally normal labs in the first 3 months and then suddenly develop an abnormality. People are either going to demonstrate an abnormality early or not have one at all.”
When Dr. Kirby starts patients on isotretinoin, she orders baseline LFTs and a lipid panel and repeats them 60 days later. “If everything is fine or only mildly high, we don’t do more testing, only a review of systems,” she said. “This is valuable to our patients because fear of needles and fainting peak during adolescence.”
Spironolactone
The clinical use of regularly monitoring potassium levels in young women taking spironolactone for acne has also been questioned. The drug has been linked to an increased risk for hyperkalemia, but the prevalence is unclear. “I got a lot of normal potassium levels in these patients [when] I was in training and I really questioned, ‘Why am I doing this? What is the rationale?’ ” Dr. Kirby said.
In a study that informed her own practice, researchers reviewed the rate of hyperkalemia in 974 healthy young women taking spironolactone for acne or for an endocrine disorder with associated acne between Dec. 1, 2000, and March 31, 2014 (JAMA Dermatol. 2015 Sep;151[9]:941-4). Of the total of 1,802 serum potassium measurements taken during treatment, 13 (0.72%) were mildly elevated levels and none of the patients had a potassium level above 5.5 mEq/L. Retesting within 1 to 3 weeks in 6 of 13 patients with elevated levels found that potassium levels were normal. “The recommendation for spironolactone in healthy women is not to check the potassium level,” Dr. Kirby said, adding that she does counsel patients about the risk of breast tenderness (which can occur 5% to 40% of the time) and spotting (which can occur in 10% to 20% of patients). Gynecomastia can occur in 10% to 30% of men, which is one of the reasons she does not use spironolactone in male patients.
TB testing and biologics
Whether or not to test for TB in patients with psoriasis taking biologic therapies represents another conundrum, she continued. Patients taking biologics are at risk of reactivation of latent TB infection, but in her experience, package inserts contain language like “perform TB testing at baseline, then periodically,” or “use at baseline, then with active TB symptoms,” and “after treatment is discontinued.”
“What the inserts didn’t recommend was to perform TB testing every year, which is what my routine had been,” Dr. Kirby said. “In the United States, thankfully we don’t have a lot of TB.” In a study that informed her own practice, researchers at a single academic medical center retrospectively reviewed the TB seroconversion rate among 316 patients treated with second-generation biologics (J Am Acad Dermatol. 2020 Oct 1;S0190-9622[20]32676-1. doi: 10.1016/j.jaad.2020.09.075). It found that only six patients (2%) converted and had a positive TB test later during treatment with the biologic. “Of these six people, all had grown up outside the U.S., had traveled outside of the U.S., or were in a group living situation,” said Dr. Kirby, who was not affiliated with the study.
“This informs our rationale for how we can do this testing. If insurance requires it every year, fine. But if they don’t, I ask patients about travel, about their living situation, and how they’re feeling. If everything’s going great, I don’t order TB testing. I do favor the interferon-gamma release assays because they’re a lot more effective than PPDs [purified protein derivative skin tests]. Also, PPDs are difficult for patients who have a low rate of returning to have that test read.”
Terbinafine for onychomycosis
Dr. Kirby also discussed the rationale for ordering regular LFTs in patients taking terbinafine for onychomycosis. “There is a risk of drug-induced liver injury from taking terbinafine, but it’s rare,” she said. “Can we be thoughtful about which patients we expose?”
Evidence suggests that patients with hyperkeratosis greater than 2 mm, with nail matrix involvement, with 50% or more of the nail involved, or having concomitant peripheral vascular disease and diabetes are recalcitrant to treatment with terbinafine
(J Am Acad Dermatol. 2019 Apr;80[4]:853-67). “If we can frame this risk, then we can frame it for our patients,” she said. “We’re more likely to cause liver injury with an antibiotic. When it comes to an oral antifungal, itraconazole is more likely than terbinafine to cause liver injury. The rate of liver injury with terbinafine is only about 2 out of 100,000. It’s five times more likely with itraconazole and 21 times more likely with Augmentin.”
She recommends obtaining a baseline LFT in patients starting terbinafine therapy “to make sure their liver is normal from the start.” In addition, she advised, “let them know that there is a TB seroconversion risk of about 1 in 50,000 people, and that if it happens there would be symptomatic changes. They would maybe notice pruritus and have a darkening in their urine, and they’d have some flu-like symptoms, which would mean stop the drug and get some care.”
Dr. Kirby emphasized that a patient’s propensity for developing drug-induced liver injury from terbinafine use is not predictable from LFT monitoring. “What you’re more likely to find is an asymptomatic LFT rise in about 1% of people,” she said.
She disclosed that she has received honoraria from AbbVie, ChemoCentryx, Incyte, Janssen, Novartis, and UCB Pharma.
From time to time, Joslyn Kirby, MD, asks other physicians about their experience with certain medications used in dermatology, especially when something new hits the market.
“Sometimes I get an answer like, ‘The last time I used that medicine, my patient needed a liver transplant,’ ” Dr. Kirby, associate professor of dermatology, Penn State University, Hershey, said during the Orlando Dermatology Aesthetic and Clinical Conference. “It’s typically a story of something rare, uncommon, and awful. The challenge with an anecdote is that for all its power, it has a lower level of evidence. But it sticks with us and influences us more than a better level of evidence because it’s a situation and a story that we might relate to.”
Dr. Kirby said that when she thinks about managing side effects from drugs used in dermatology, it usually relates to something common and low-risk such as sore, dry skin with isotretinoin use. In contrast, if there is an uncommon but serious side effect, then mitigation rather than management is key. “I want to mitigate the risk – meaning warn my patient about it or be careful about how I select my patients when it is a serious side effect that happens infrequently,” she said. “The worst combination is a frequent and severe side effect. That is something we should avoid, for sure.”
Isotretinoin
But another aspect of prescribing a new drug for patients can be less clear-cut, Dr. Kirby continued, such as the rationale for routine lab monitoring. She began by discussing one of her male patients with moderate to severe acne. After he failed oral antibiotics and topical retinoids, she recommended isotretinoin, which carries a risk of hypertriglyceridemia-associated pancreatitis. “Early in my career, I was getting a lot of monthly labs in patients on this drug that were totally normal and not influencing my practice,” Dr. Kirby recalled. “We’ve seen studies coming out on isotretinoin lab monitoring, showing us that we can keep our patients safe and that we really don’t need to be checking labs as often, because lab changes are infrequent.”
In one of those studies, researchers evaluated 1,863 patients treated with isotretinoin for acne between Jan. 1, 2008, and June 30, 2017 (J Am Acad Dermatol. 2020 Jan;82[1]:72-9).Over time, fewer than 1% of patients screened developed grade 3 or greater triglyceride testing abnormalities, while fewer than 0.5% developed liver function testing (LFT) abnormalities. Authors of a separate systematic review concluded that for patients on isotretinoin therapy without elevated baseline triglycerides, or risk thereof, monitoring triglycerides is of little value (Br J Dermatol. 2017 Oct;177[4]:960-6). Of the 25 patients in the analysis who developed pancreatitis on isotretinoin, only 3 had elevated triglycerides at baseline.
“I was taught that I need to check triglycerides frequently due to the risk of pancreatitis developing with isotretinoin use,” Dr. Kirby said. “Lipid changes on therapy are expected, but they tend to peak early, meaning the first 3 months of treatment when we’re ramping up from a starting dose to a maintenance dose. It’s rare for somebody to be a late bloomer, meaning that they have totally normal labs in the first 3 months and then suddenly develop an abnormality. People are either going to demonstrate an abnormality early or not have one at all.”
When Dr. Kirby starts patients on isotretinoin, she orders baseline LFTs and a lipid panel and repeats them 60 days later. “If everything is fine or only mildly high, we don’t do more testing, only a review of systems,” she said. “This is valuable to our patients because fear of needles and fainting peak during adolescence.”
Spironolactone
The clinical use of regularly monitoring potassium levels in young women taking spironolactone for acne has also been questioned. The drug has been linked to an increased risk for hyperkalemia, but the prevalence is unclear. “I got a lot of normal potassium levels in these patients [when] I was in training and I really questioned, ‘Why am I doing this? What is the rationale?’ ” Dr. Kirby said.
In a study that informed her own practice, researchers reviewed the rate of hyperkalemia in 974 healthy young women taking spironolactone for acne or for an endocrine disorder with associated acne between Dec. 1, 2000, and March 31, 2014 (JAMA Dermatol. 2015 Sep;151[9]:941-4). Of the total of 1,802 serum potassium measurements taken during treatment, 13 (0.72%) were mildly elevated levels and none of the patients had a potassium level above 5.5 mEq/L. Retesting within 1 to 3 weeks in 6 of 13 patients with elevated levels found that potassium levels were normal. “The recommendation for spironolactone in healthy women is not to check the potassium level,” Dr. Kirby said, adding that she does counsel patients about the risk of breast tenderness (which can occur 5% to 40% of the time) and spotting (which can occur in 10% to 20% of patients). Gynecomastia can occur in 10% to 30% of men, which is one of the reasons she does not use spironolactone in male patients.
TB testing and biologics
Whether or not to test for TB in patients with psoriasis taking biologic therapies represents another conundrum, she continued. Patients taking biologics are at risk of reactivation of latent TB infection, but in her experience, package inserts contain language like “perform TB testing at baseline, then periodically,” or “use at baseline, then with active TB symptoms,” and “after treatment is discontinued.”
“What the inserts didn’t recommend was to perform TB testing every year, which is what my routine had been,” Dr. Kirby said. “In the United States, thankfully we don’t have a lot of TB.” In a study that informed her own practice, researchers at a single academic medical center retrospectively reviewed the TB seroconversion rate among 316 patients treated with second-generation biologics (J Am Acad Dermatol. 2020 Oct 1;S0190-9622[20]32676-1. doi: 10.1016/j.jaad.2020.09.075). It found that only six patients (2%) converted and had a positive TB test later during treatment with the biologic. “Of these six people, all had grown up outside the U.S., had traveled outside of the U.S., or were in a group living situation,” said Dr. Kirby, who was not affiliated with the study.
“This informs our rationale for how we can do this testing. If insurance requires it every year, fine. But if they don’t, I ask patients about travel, about their living situation, and how they’re feeling. If everything’s going great, I don’t order TB testing. I do favor the interferon-gamma release assays because they’re a lot more effective than PPDs [purified protein derivative skin tests]. Also, PPDs are difficult for patients who have a low rate of returning to have that test read.”
Terbinafine for onychomycosis
Dr. Kirby also discussed the rationale for ordering regular LFTs in patients taking terbinafine for onychomycosis. “There is a risk of drug-induced liver injury from taking terbinafine, but it’s rare,” she said. “Can we be thoughtful about which patients we expose?”
Evidence suggests that patients with hyperkeratosis greater than 2 mm, with nail matrix involvement, with 50% or more of the nail involved, or having concomitant peripheral vascular disease and diabetes are recalcitrant to treatment with terbinafine
(J Am Acad Dermatol. 2019 Apr;80[4]:853-67). “If we can frame this risk, then we can frame it for our patients,” she said. “We’re more likely to cause liver injury with an antibiotic. When it comes to an oral antifungal, itraconazole is more likely than terbinafine to cause liver injury. The rate of liver injury with terbinafine is only about 2 out of 100,000. It’s five times more likely with itraconazole and 21 times more likely with Augmentin.”
She recommends obtaining a baseline LFT in patients starting terbinafine therapy “to make sure their liver is normal from the start.” In addition, she advised, “let them know that there is a TB seroconversion risk of about 1 in 50,000 people, and that if it happens there would be symptomatic changes. They would maybe notice pruritus and have a darkening in their urine, and they’d have some flu-like symptoms, which would mean stop the drug and get some care.”
Dr. Kirby emphasized that a patient’s propensity for developing drug-induced liver injury from terbinafine use is not predictable from LFT monitoring. “What you’re more likely to find is an asymptomatic LFT rise in about 1% of people,” she said.
She disclosed that she has received honoraria from AbbVie, ChemoCentryx, Incyte, Janssen, Novartis, and UCB Pharma.
From time to time, Joslyn Kirby, MD, asks other physicians about their experience with certain medications used in dermatology, especially when something new hits the market.
“Sometimes I get an answer like, ‘The last time I used that medicine, my patient needed a liver transplant,’ ” Dr. Kirby, associate professor of dermatology, Penn State University, Hershey, said during the Orlando Dermatology Aesthetic and Clinical Conference. “It’s typically a story of something rare, uncommon, and awful. The challenge with an anecdote is that for all its power, it has a lower level of evidence. But it sticks with us and influences us more than a better level of evidence because it’s a situation and a story that we might relate to.”
Dr. Kirby said that when she thinks about managing side effects from drugs used in dermatology, it usually relates to something common and low-risk such as sore, dry skin with isotretinoin use. In contrast, if there is an uncommon but serious side effect, then mitigation rather than management is key. “I want to mitigate the risk – meaning warn my patient about it or be careful about how I select my patients when it is a serious side effect that happens infrequently,” she said. “The worst combination is a frequent and severe side effect. That is something we should avoid, for sure.”
Isotretinoin
But another aspect of prescribing a new drug for patients can be less clear-cut, Dr. Kirby continued, such as the rationale for routine lab monitoring. She began by discussing one of her male patients with moderate to severe acne. After he failed oral antibiotics and topical retinoids, she recommended isotretinoin, which carries a risk of hypertriglyceridemia-associated pancreatitis. “Early in my career, I was getting a lot of monthly labs in patients on this drug that were totally normal and not influencing my practice,” Dr. Kirby recalled. “We’ve seen studies coming out on isotretinoin lab monitoring, showing us that we can keep our patients safe and that we really don’t need to be checking labs as often, because lab changes are infrequent.”
In one of those studies, researchers evaluated 1,863 patients treated with isotretinoin for acne between Jan. 1, 2008, and June 30, 2017 (J Am Acad Dermatol. 2020 Jan;82[1]:72-9).Over time, fewer than 1% of patients screened developed grade 3 or greater triglyceride testing abnormalities, while fewer than 0.5% developed liver function testing (LFT) abnormalities. Authors of a separate systematic review concluded that for patients on isotretinoin therapy without elevated baseline triglycerides, or risk thereof, monitoring triglycerides is of little value (Br J Dermatol. 2017 Oct;177[4]:960-6). Of the 25 patients in the analysis who developed pancreatitis on isotretinoin, only 3 had elevated triglycerides at baseline.
“I was taught that I need to check triglycerides frequently due to the risk of pancreatitis developing with isotretinoin use,” Dr. Kirby said. “Lipid changes on therapy are expected, but they tend to peak early, meaning the first 3 months of treatment when we’re ramping up from a starting dose to a maintenance dose. It’s rare for somebody to be a late bloomer, meaning that they have totally normal labs in the first 3 months and then suddenly develop an abnormality. People are either going to demonstrate an abnormality early or not have one at all.”
When Dr. Kirby starts patients on isotretinoin, she orders baseline LFTs and a lipid panel and repeats them 60 days later. “If everything is fine or only mildly high, we don’t do more testing, only a review of systems,” she said. “This is valuable to our patients because fear of needles and fainting peak during adolescence.”
Spironolactone
The clinical use of regularly monitoring potassium levels in young women taking spironolactone for acne has also been questioned. The drug has been linked to an increased risk for hyperkalemia, but the prevalence is unclear. “I got a lot of normal potassium levels in these patients [when] I was in training and I really questioned, ‘Why am I doing this? What is the rationale?’ ” Dr. Kirby said.
In a study that informed her own practice, researchers reviewed the rate of hyperkalemia in 974 healthy young women taking spironolactone for acne or for an endocrine disorder with associated acne between Dec. 1, 2000, and March 31, 2014 (JAMA Dermatol. 2015 Sep;151[9]:941-4). Of the total of 1,802 serum potassium measurements taken during treatment, 13 (0.72%) were mildly elevated levels and none of the patients had a potassium level above 5.5 mEq/L. Retesting within 1 to 3 weeks in 6 of 13 patients with elevated levels found that potassium levels were normal. “The recommendation for spironolactone in healthy women is not to check the potassium level,” Dr. Kirby said, adding that she does counsel patients about the risk of breast tenderness (which can occur 5% to 40% of the time) and spotting (which can occur in 10% to 20% of patients). Gynecomastia can occur in 10% to 30% of men, which is one of the reasons she does not use spironolactone in male patients.
TB testing and biologics
Whether or not to test for TB in patients with psoriasis taking biologic therapies represents another conundrum, she continued. Patients taking biologics are at risk of reactivation of latent TB infection, but in her experience, package inserts contain language like “perform TB testing at baseline, then periodically,” or “use at baseline, then with active TB symptoms,” and “after treatment is discontinued.”
“What the inserts didn’t recommend was to perform TB testing every year, which is what my routine had been,” Dr. Kirby said. “In the United States, thankfully we don’t have a lot of TB.” In a study that informed her own practice, researchers at a single academic medical center retrospectively reviewed the TB seroconversion rate among 316 patients treated with second-generation biologics (J Am Acad Dermatol. 2020 Oct 1;S0190-9622[20]32676-1. doi: 10.1016/j.jaad.2020.09.075). It found that only six patients (2%) converted and had a positive TB test later during treatment with the biologic. “Of these six people, all had grown up outside the U.S., had traveled outside of the U.S., or were in a group living situation,” said Dr. Kirby, who was not affiliated with the study.
“This informs our rationale for how we can do this testing. If insurance requires it every year, fine. But if they don’t, I ask patients about travel, about their living situation, and how they’re feeling. If everything’s going great, I don’t order TB testing. I do favor the interferon-gamma release assays because they’re a lot more effective than PPDs [purified protein derivative skin tests]. Also, PPDs are difficult for patients who have a low rate of returning to have that test read.”
Terbinafine for onychomycosis
Dr. Kirby also discussed the rationale for ordering regular LFTs in patients taking terbinafine for onychomycosis. “There is a risk of drug-induced liver injury from taking terbinafine, but it’s rare,” she said. “Can we be thoughtful about which patients we expose?”
Evidence suggests that patients with hyperkeratosis greater than 2 mm, with nail matrix involvement, with 50% or more of the nail involved, or having concomitant peripheral vascular disease and diabetes are recalcitrant to treatment with terbinafine
(J Am Acad Dermatol. 2019 Apr;80[4]:853-67). “If we can frame this risk, then we can frame it for our patients,” she said. “We’re more likely to cause liver injury with an antibiotic. When it comes to an oral antifungal, itraconazole is more likely than terbinafine to cause liver injury. The rate of liver injury with terbinafine is only about 2 out of 100,000. It’s five times more likely with itraconazole and 21 times more likely with Augmentin.”
She recommends obtaining a baseline LFT in patients starting terbinafine therapy “to make sure their liver is normal from the start.” In addition, she advised, “let them know that there is a TB seroconversion risk of about 1 in 50,000 people, and that if it happens there would be symptomatic changes. They would maybe notice pruritus and have a darkening in their urine, and they’d have some flu-like symptoms, which would mean stop the drug and get some care.”
Dr. Kirby emphasized that a patient’s propensity for developing drug-induced liver injury from terbinafine use is not predictable from LFT monitoring. “What you’re more likely to find is an asymptomatic LFT rise in about 1% of people,” she said.
She disclosed that she has received honoraria from AbbVie, ChemoCentryx, Incyte, Janssen, Novartis, and UCB Pharma.
FROM ODAC 2021
Child with yellow nodule
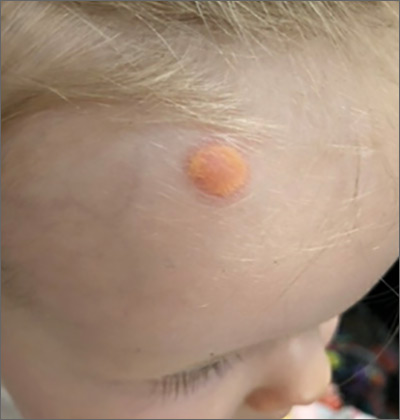
The characteristic orange-yellow color is the tip-off to the diagnosis of juvenile xanthogranuloma (JXG). It manifests as asymptomatic solitary or scattered papules or nodules, congenitally, or most commonly during the first year of life.
JXG is an unusual non-Langerhans cell histiocytosis that more commonly affects males. The etiology of JXG is unclear; it is presumed to be due to physical or infectious stimuli that produce a granulomatous histiocytic reaction. JXG typically manifests on the head, neck, upper extremities, and trunk. The appearance of JXG may be similar to that of Langerhans cell histiocytosis. If necessary, the diagnosis of JXG can be confirmed with a skin biopsy, which will reveal Touton-type giant cells and foamy histiocytes.
JXG is a benign and self-limiting disorder and spontaneously regresses within a few years. In rare cases, it can be systemic. If there are multiple lesions, relevant history, or physical exam features suggesting space-occupying lesions, imaging should be performed to rule out lesions in internal organs or structures. Treatment is indicated when there is systemic or symptomatic ocular involvement and may include surgical excision, radiotherapy, and/or systemic chemotherapy. In this case, the patient’s JXG management involved routine monitoring in anticipation of spontaneous resolution.
Image courtesy of John Durkin, MD, FAAD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kerry Song, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Collie JS, Harper CD, Fillman EP. Juvenile Xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2020 Jan. Accessed January 29, 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/#_NBK526103_pubdet

The characteristic orange-yellow color is the tip-off to the diagnosis of juvenile xanthogranuloma (JXG). It manifests as asymptomatic solitary or scattered papules or nodules, congenitally, or most commonly during the first year of life.
JXG is an unusual non-Langerhans cell histiocytosis that more commonly affects males. The etiology of JXG is unclear; it is presumed to be due to physical or infectious stimuli that produce a granulomatous histiocytic reaction. JXG typically manifests on the head, neck, upper extremities, and trunk. The appearance of JXG may be similar to that of Langerhans cell histiocytosis. If necessary, the diagnosis of JXG can be confirmed with a skin biopsy, which will reveal Touton-type giant cells and foamy histiocytes.
JXG is a benign and self-limiting disorder and spontaneously regresses within a few years. In rare cases, it can be systemic. If there are multiple lesions, relevant history, or physical exam features suggesting space-occupying lesions, imaging should be performed to rule out lesions in internal organs or structures. Treatment is indicated when there is systemic or symptomatic ocular involvement and may include surgical excision, radiotherapy, and/or systemic chemotherapy. In this case, the patient’s JXG management involved routine monitoring in anticipation of spontaneous resolution.
Image courtesy of John Durkin, MD, FAAD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kerry Song, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The characteristic orange-yellow color is the tip-off to the diagnosis of juvenile xanthogranuloma (JXG). It manifests as asymptomatic solitary or scattered papules or nodules, congenitally, or most commonly during the first year of life.
JXG is an unusual non-Langerhans cell histiocytosis that more commonly affects males. The etiology of JXG is unclear; it is presumed to be due to physical or infectious stimuli that produce a granulomatous histiocytic reaction. JXG typically manifests on the head, neck, upper extremities, and trunk. The appearance of JXG may be similar to that of Langerhans cell histiocytosis. If necessary, the diagnosis of JXG can be confirmed with a skin biopsy, which will reveal Touton-type giant cells and foamy histiocytes.
JXG is a benign and self-limiting disorder and spontaneously regresses within a few years. In rare cases, it can be systemic. If there are multiple lesions, relevant history, or physical exam features suggesting space-occupying lesions, imaging should be performed to rule out lesions in internal organs or structures. Treatment is indicated when there is systemic or symptomatic ocular involvement and may include surgical excision, radiotherapy, and/or systemic chemotherapy. In this case, the patient’s JXG management involved routine monitoring in anticipation of spontaneous resolution.
Image courtesy of John Durkin, MD, FAAD, Department of Dermatology, University of New Mexico School of Medicine, Albuquerque. Text courtesy of Kerry Song, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Collie JS, Harper CD, Fillman EP. Juvenile Xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2020 Jan. Accessed January 29, 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/#_NBK526103_pubdet
Collie JS, Harper CD, Fillman EP. Juvenile Xanthogranuloma. In: StatPearls [Internet]. StatPearls Publishing; 2020 Jan. Accessed January 29, 2021. https://www.ncbi.nlm.nih.gov/books/NBK526103/#_NBK526103_pubdet