User login
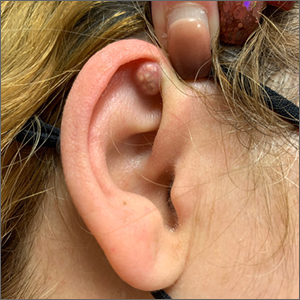
A mass on the ear
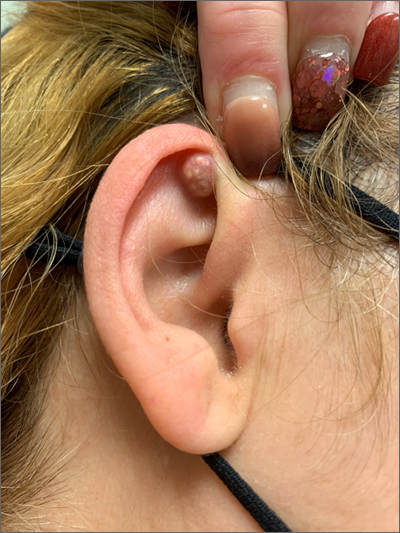
Pathology indicated a proliferation of basaloid cells with matrical differentiation in transition and “shadow” cells, pointing to a diagnosis of pilomatricoma.
Pilomatricoma, also known as pilomatrixoma, is a benign skin tumor associated with hair follicles. The lesions are most often found on the neck or head area but can occur on the arms, legs, or torso. They are usually slow growing, solitary, and painless. The frequency of occurrence is rare, accounting for less than 1% of all benign skin tumors.1
A mutation in the Catenin beta-1 (CTNNB1) gene is the most common cause of isolated pilomatricoma and is a somatic defect, meaning it is acquired, not inherited. The mutation of the CTNNB1 gene causes disruption of normal function and maturation of the hair follicle. This leads to rapid cell growth and uncontrolled division, resulting in the formation of the pilomatricoma.1
A comprehensive review performed in 2018 noted that only 16% of pilomatricomas were accurately diagnosed on clinical exam.1 Clues that point to the diagnosis of pilomatricoma are the irregular, whitish yellow spots just under the skin. In contrast, epidermoid cysts usually have a central pore and a ballotable feel. The expression of calcification and gritty material from the lesion in this case ruled out a diagnosis of an epidermoid cyst. The most common method of treatment is surgical removal.1
This patient was counseled regarding her diagnosis and given the option of a plastic surgery referral to excise the affected tissue in its entirety. She opted to wait and see if the growth would scar down and not return.
Image courtesy of Edward A. Jackson, MD. Text courtesy of Edward A. Jackson, MD, FAAFP, Advent Health Medical Group Family Medicine at East Orlando, FL, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Jones CD, Ho W, Robertson BF, et al. Pilomatrixoma: a comprehensive review of the literature. Am J Dermatopathol. 2018;40:631-641. doi: 10.1097/DAD.0000000000001118

Pathology indicated a proliferation of basaloid cells with matrical differentiation in transition and “shadow” cells, pointing to a diagnosis of pilomatricoma.
Pilomatricoma, also known as pilomatrixoma, is a benign skin tumor associated with hair follicles. The lesions are most often found on the neck or head area but can occur on the arms, legs, or torso. They are usually slow growing, solitary, and painless. The frequency of occurrence is rare, accounting for less than 1% of all benign skin tumors.1
A mutation in the Catenin beta-1 (CTNNB1) gene is the most common cause of isolated pilomatricoma and is a somatic defect, meaning it is acquired, not inherited. The mutation of the CTNNB1 gene causes disruption of normal function and maturation of the hair follicle. This leads to rapid cell growth and uncontrolled division, resulting in the formation of the pilomatricoma.1
A comprehensive review performed in 2018 noted that only 16% of pilomatricomas were accurately diagnosed on clinical exam.1 Clues that point to the diagnosis of pilomatricoma are the irregular, whitish yellow spots just under the skin. In contrast, epidermoid cysts usually have a central pore and a ballotable feel. The expression of calcification and gritty material from the lesion in this case ruled out a diagnosis of an epidermoid cyst. The most common method of treatment is surgical removal.1
This patient was counseled regarding her diagnosis and given the option of a plastic surgery referral to excise the affected tissue in its entirety. She opted to wait and see if the growth would scar down and not return.
Image courtesy of Edward A. Jackson, MD. Text courtesy of Edward A. Jackson, MD, FAAFP, Advent Health Medical Group Family Medicine at East Orlando, FL, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

Pathology indicated a proliferation of basaloid cells with matrical differentiation in transition and “shadow” cells, pointing to a diagnosis of pilomatricoma.
Pilomatricoma, also known as pilomatrixoma, is a benign skin tumor associated with hair follicles. The lesions are most often found on the neck or head area but can occur on the arms, legs, or torso. They are usually slow growing, solitary, and painless. The frequency of occurrence is rare, accounting for less than 1% of all benign skin tumors.1
A mutation in the Catenin beta-1 (CTNNB1) gene is the most common cause of isolated pilomatricoma and is a somatic defect, meaning it is acquired, not inherited. The mutation of the CTNNB1 gene causes disruption of normal function and maturation of the hair follicle. This leads to rapid cell growth and uncontrolled division, resulting in the formation of the pilomatricoma.1
A comprehensive review performed in 2018 noted that only 16% of pilomatricomas were accurately diagnosed on clinical exam.1 Clues that point to the diagnosis of pilomatricoma are the irregular, whitish yellow spots just under the skin. In contrast, epidermoid cysts usually have a central pore and a ballotable feel. The expression of calcification and gritty material from the lesion in this case ruled out a diagnosis of an epidermoid cyst. The most common method of treatment is surgical removal.1
This patient was counseled regarding her diagnosis and given the option of a plastic surgery referral to excise the affected tissue in its entirety. She opted to wait and see if the growth would scar down and not return.
Image courtesy of Edward A. Jackson, MD. Text courtesy of Edward A. Jackson, MD, FAAFP, Advent Health Medical Group Family Medicine at East Orlando, FL, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Jones CD, Ho W, Robertson BF, et al. Pilomatrixoma: a comprehensive review of the literature. Am J Dermatopathol. 2018;40:631-641. doi: 10.1097/DAD.0000000000001118
1. Jones CD, Ho W, Robertson BF, et al. Pilomatrixoma: a comprehensive review of the literature. Am J Dermatopathol. 2018;40:631-641. doi: 10.1097/DAD.0000000000001118
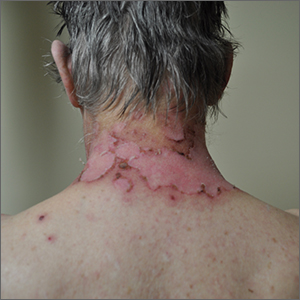
How does atopic dermatitis present in skin of color?
“We see very heterogenous and broad clinical presentations across the diverse patient populations that we see,” Andrew F. Alexis, MD, MPH, said at the Revolutionizing Atopic Dermatitis symposium. “Some of these differences might be related to population variations in skin barrier function, immunologic factors, genetic factors, and environmental factors, which all interplay to produce variations in the clinical presentation and overall impact of AD. Many nongenetic factors also contribute to differences that we see, including some socioeconomic and other factors that feed into health disparities.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, discussed four main clinical features of AD in skin of color.
Erythema is less visible because it is masked by pigment
“There can be some masking of the redness and alteration of that color such that it doesn’t look bright red as it would in the background of lightly pigmented skin,” Dr. Alexis said. “Instead, the [AD lesions] have shades of grayish-red or grayish-brown or reddish-brown. It’s important to recognize this clinical presentation and look carefully and assess the patient – not just visually but with palpation and take into consideration symptomatology so that you don’t fall into the trap of calling an AD lesion postinflammatory hyperpigmentation. It’s also helpful to isolate the islands of normal or nonlesional skin and contrast that with the areas of lesional skin, to get a sense of how active and inflamed the areas are. Palpation really helps to appreciate the elevation of the lesions that are involved.”
Follicular accentuation
Morphological variants common in skin of color include the follicular variant or micropapular variant of AD. “You might just see a collection of papules that are 1-2 mm in size and pruritic and in typical sites of predilection [for] eczema,” he said. Prurigo nodularis–like lesions or prurigo nodularis in association with AD are also seen more frequently in skin of color.
Lichenification
The lichenoid variant of AD is characterized by a violaceous hue and other features that resemble lichen planus and has been reported to be more common in individuals of African descent. A prospective study of about 1,000 patients with AD seen over 2 years at a dermatology clinic in southeastern Nigeria found that 54% of patients had papular lichenoid lesions. In addition, 51% had elevated blood eosinophil counts, especially those with severe disease.
Dr. Alexis added that psoriasiform features have been reported in studies of East Asian populations with AD. These plaques may be more well demarcated and have clinical and histologic features that resemble psoriasis.
Dyspigmentation
One common feature across the spectrum of patients with skin of color “is the risk of longstanding pigmentary sequelae in the form of hyperpigmentation or hypopigmentation,” said Dr. Alexis, who is also vice chair for diversity and inclusion for the department of dermatology at Weill Cornell Medicine. “In very severe longstanding areas with chronic excoriation to the point of breaking of the skin, eroding of the skin, causing permanent damage to the melanocytes, dyspigmentation that resembles vitiligo can be seen. We can also see hypopigmentation as a consequence of topical corticosteroids, particularly those that are class I or class II and are used for prolonged periods of time.”
Dr. Alexis noted that delays in treatment and undertreatment can contribute to a higher risk of pigmentary and other long-term sequelae. “New therapies show promise in improving outcomes in AD patients with skin of color. When it comes to therapeutic responses, there are some post hoc studies that have investigated potential differences in safety and efficacy of the agents that have been recently approved. We clearly need more data to better understand if there are potential racial or ethnic differences.”
Dr. Alexis reported no relevant financial relationships.
Commentary by Lawrence F. Eichenfield, MD
Atopic dermatitis (AD) is highly heterogenous, with tremendous variations in extent, qualities of eczema, symptom complex, and physical presentation. Prior studies have reported disparities of care delivered to racial and ethnic minorities in the United States, as well as higher susceptibility to AD and odds of persistent disease into adulthood from child-onset AD. Recognizing some differences in presentation of AD in patients with skin of color is important as we select our therapeutic interventions, including assessing new treatments being added to our armamentarium. Erythema may be harder to notice in darker skin, but attempting to blanch the skin with pressure can help to assess the color and inflammation. Appreciating lichenoid changes, including papular and “micropapular” AD, and psoriasiform-like thickening in certain patients (reportedly more common in East Asian populations) are important as well. And dyspigmentation is an important aspect of the disease presentation and patient and parental concern, given both hypopigmentaton and hyperpigmentation commonly seen over the course of AD.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
“We see very heterogenous and broad clinical presentations across the diverse patient populations that we see,” Andrew F. Alexis, MD, MPH, said at the Revolutionizing Atopic Dermatitis symposium. “Some of these differences might be related to population variations in skin barrier function, immunologic factors, genetic factors, and environmental factors, which all interplay to produce variations in the clinical presentation and overall impact of AD. Many nongenetic factors also contribute to differences that we see, including some socioeconomic and other factors that feed into health disparities.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, discussed four main clinical features of AD in skin of color.
Erythema is less visible because it is masked by pigment
“There can be some masking of the redness and alteration of that color such that it doesn’t look bright red as it would in the background of lightly pigmented skin,” Dr. Alexis said. “Instead, the [AD lesions] have shades of grayish-red or grayish-brown or reddish-brown. It’s important to recognize this clinical presentation and look carefully and assess the patient – not just visually but with palpation and take into consideration symptomatology so that you don’t fall into the trap of calling an AD lesion postinflammatory hyperpigmentation. It’s also helpful to isolate the islands of normal or nonlesional skin and contrast that with the areas of lesional skin, to get a sense of how active and inflamed the areas are. Palpation really helps to appreciate the elevation of the lesions that are involved.”
Follicular accentuation
Morphological variants common in skin of color include the follicular variant or micropapular variant of AD. “You might just see a collection of papules that are 1-2 mm in size and pruritic and in typical sites of predilection [for] eczema,” he said. Prurigo nodularis–like lesions or prurigo nodularis in association with AD are also seen more frequently in skin of color.
Lichenification
The lichenoid variant of AD is characterized by a violaceous hue and other features that resemble lichen planus and has been reported to be more common in individuals of African descent. A prospective study of about 1,000 patients with AD seen over 2 years at a dermatology clinic in southeastern Nigeria found that 54% of patients had papular lichenoid lesions. In addition, 51% had elevated blood eosinophil counts, especially those with severe disease.
Dr. Alexis added that psoriasiform features have been reported in studies of East Asian populations with AD. These plaques may be more well demarcated and have clinical and histologic features that resemble psoriasis.
Dyspigmentation
One common feature across the spectrum of patients with skin of color “is the risk of longstanding pigmentary sequelae in the form of hyperpigmentation or hypopigmentation,” said Dr. Alexis, who is also vice chair for diversity and inclusion for the department of dermatology at Weill Cornell Medicine. “In very severe longstanding areas with chronic excoriation to the point of breaking of the skin, eroding of the skin, causing permanent damage to the melanocytes, dyspigmentation that resembles vitiligo can be seen. We can also see hypopigmentation as a consequence of topical corticosteroids, particularly those that are class I or class II and are used for prolonged periods of time.”
Dr. Alexis noted that delays in treatment and undertreatment can contribute to a higher risk of pigmentary and other long-term sequelae. “New therapies show promise in improving outcomes in AD patients with skin of color. When it comes to therapeutic responses, there are some post hoc studies that have investigated potential differences in safety and efficacy of the agents that have been recently approved. We clearly need more data to better understand if there are potential racial or ethnic differences.”
Dr. Alexis reported no relevant financial relationships.
Commentary by Lawrence F. Eichenfield, MD
Atopic dermatitis (AD) is highly heterogenous, with tremendous variations in extent, qualities of eczema, symptom complex, and physical presentation. Prior studies have reported disparities of care delivered to racial and ethnic minorities in the United States, as well as higher susceptibility to AD and odds of persistent disease into adulthood from child-onset AD. Recognizing some differences in presentation of AD in patients with skin of color is important as we select our therapeutic interventions, including assessing new treatments being added to our armamentarium. Erythema may be harder to notice in darker skin, but attempting to blanch the skin with pressure can help to assess the color and inflammation. Appreciating lichenoid changes, including papular and “micropapular” AD, and psoriasiform-like thickening in certain patients (reportedly more common in East Asian populations) are important as well. And dyspigmentation is an important aspect of the disease presentation and patient and parental concern, given both hypopigmentaton and hyperpigmentation commonly seen over the course of AD.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
“We see very heterogenous and broad clinical presentations across the diverse patient populations that we see,” Andrew F. Alexis, MD, MPH, said at the Revolutionizing Atopic Dermatitis symposium. “Some of these differences might be related to population variations in skin barrier function, immunologic factors, genetic factors, and environmental factors, which all interplay to produce variations in the clinical presentation and overall impact of AD. Many nongenetic factors also contribute to differences that we see, including some socioeconomic and other factors that feed into health disparities.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, discussed four main clinical features of AD in skin of color.
Erythema is less visible because it is masked by pigment
“There can be some masking of the redness and alteration of that color such that it doesn’t look bright red as it would in the background of lightly pigmented skin,” Dr. Alexis said. “Instead, the [AD lesions] have shades of grayish-red or grayish-brown or reddish-brown. It’s important to recognize this clinical presentation and look carefully and assess the patient – not just visually but with palpation and take into consideration symptomatology so that you don’t fall into the trap of calling an AD lesion postinflammatory hyperpigmentation. It’s also helpful to isolate the islands of normal or nonlesional skin and contrast that with the areas of lesional skin, to get a sense of how active and inflamed the areas are. Palpation really helps to appreciate the elevation of the lesions that are involved.”
Follicular accentuation
Morphological variants common in skin of color include the follicular variant or micropapular variant of AD. “You might just see a collection of papules that are 1-2 mm in size and pruritic and in typical sites of predilection [for] eczema,” he said. Prurigo nodularis–like lesions or prurigo nodularis in association with AD are also seen more frequently in skin of color.
Lichenification
The lichenoid variant of AD is characterized by a violaceous hue and other features that resemble lichen planus and has been reported to be more common in individuals of African descent. A prospective study of about 1,000 patients with AD seen over 2 years at a dermatology clinic in southeastern Nigeria found that 54% of patients had papular lichenoid lesions. In addition, 51% had elevated blood eosinophil counts, especially those with severe disease.
Dr. Alexis added that psoriasiform features have been reported in studies of East Asian populations with AD. These plaques may be more well demarcated and have clinical and histologic features that resemble psoriasis.
Dyspigmentation
One common feature across the spectrum of patients with skin of color “is the risk of longstanding pigmentary sequelae in the form of hyperpigmentation or hypopigmentation,” said Dr. Alexis, who is also vice chair for diversity and inclusion for the department of dermatology at Weill Cornell Medicine. “In very severe longstanding areas with chronic excoriation to the point of breaking of the skin, eroding of the skin, causing permanent damage to the melanocytes, dyspigmentation that resembles vitiligo can be seen. We can also see hypopigmentation as a consequence of topical corticosteroids, particularly those that are class I or class II and are used for prolonged periods of time.”
Dr. Alexis noted that delays in treatment and undertreatment can contribute to a higher risk of pigmentary and other long-term sequelae. “New therapies show promise in improving outcomes in AD patients with skin of color. When it comes to therapeutic responses, there are some post hoc studies that have investigated potential differences in safety and efficacy of the agents that have been recently approved. We clearly need more data to better understand if there are potential racial or ethnic differences.”
Dr. Alexis reported no relevant financial relationships.
Commentary by Lawrence F. Eichenfield, MD
Atopic dermatitis (AD) is highly heterogenous, with tremendous variations in extent, qualities of eczema, symptom complex, and physical presentation. Prior studies have reported disparities of care delivered to racial and ethnic minorities in the United States, as well as higher susceptibility to AD and odds of persistent disease into adulthood from child-onset AD. Recognizing some differences in presentation of AD in patients with skin of color is important as we select our therapeutic interventions, including assessing new treatments being added to our armamentarium. Erythema may be harder to notice in darker skin, but attempting to blanch the skin with pressure can help to assess the color and inflammation. Appreciating lichenoid changes, including papular and “micropapular” AD, and psoriasiform-like thickening in certain patients (reportedly more common in East Asian populations) are important as well. And dyspigmentation is an important aspect of the disease presentation and patient and parental concern, given both hypopigmentaton and hyperpigmentation commonly seen over the course of AD.
Dr. Eichenfield is chief of pediatric and adolescent dermatology at Rady Children's Hospital-San Diego. He is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego. He disclosed that he has served as an investigator and/or consultant to AbbVie, Lilly, Pfizer, Regeneron, Sanofi-Genzyme, and Verrica.
A version of this article first appeared on Medscape.com.
This article was updated 6/18/22.
FROM REVOLUTIONIZING AD 2021
Abrocitinib efficacy dose-dependent, similar across AD age groups
and was comparable in patients aged 51 years and older, results from a post hoc analysis of four trials showed.
Abrocitinib (Cibinqo) is an oral, once-daily, Janus kinase 1 selective inhibitor that has shown good efficacy and safety as monotherapy or combined with topical therapy for treatment of patients with moderate to severe AD. The agent was approved in mid-December in Europe for the treatment of moderate to severe AD in adults who are candidates for systemic therapy and is currently under review by the Food and Drug Administration.
“We know that responses to, and adverse events associated with, systemic therapies may vary among patients of different ages,” Andrew F. Alexis, MD, MPH, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “The efficacy and safety of abrocitinib monotherapy were previously evaluated in adolescent and adult subpopulations from controlled clinical trials in patients with moderate to severe AD. The objective of the current study was to assess the impact of age on short-term responses to abrocitinib treatment in patients with moderate to severe AD.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, and colleagues performed a post hoc analysis across four randomized, double-blind studies that was stratified by age group: 12-17 years, 18-40 years, 41-50 years, and 51 years and older. Efficacy data were assessed separately for patients in the monotherapy pool and in the JADE COMPARE trial. The monotherapy pool included patients from one phase 2b study and two phase 3 studies who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo monotherapy for 12 weeks (JADE-MONO-1 and JADE-MONO-2).
The JADE COMPARE pool included patients who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo, plus medicated topical therapy for 16 weeks. Data from patients in all four trials were pooled for the analysis of treatment-emergent adverse events. Efficacy points analyzed were the Investigator Global Assessment (IGA) score of 0/1 (clear or almost clear), a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75), or Peak Pruritus Numeric Rating Scale score (PP-NRS4) at week 12 for the monotherapy pool and at week 16 for COMPARE.
In the monotherapy pool, the proportions of patients ages 12-17 years, 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response at 12 weeks were 31.3%, 40.2%, 43.8%, and 50.8% (abrocitinib 200 mg); 22%, 23.7%, 22.4%, and 40.8% (abrocitinib 100 mg); and 8.7%, 8%, 3.3%, and 10% (placebo).
In JADE COMPARE, the proportions of patients aged 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response were 50%, 53.2%, and 34.8% (abrocitinib 200 mg); 36.9%, 37.1%, and 26.1% (abrocitinib 100 mg); and 12%, 11.8%, and 16.7% (placebo) at 16 weeks. Similar trends were observed for EASI-75 and PP-NRS4 responses at 12 weeks.
Across all age groups, the most common treatment-emergent adverse events were infections/infestations and gastrointestinal effects; most cases were mild or moderate. Nausea was more frequent in the two younger age groups and was dose related: For abrocitinib 200 mg and abrocitinib 100 mg, respectively, the rates of nausea were 18.8% and 7.8% in patients aged 12-17 years; 17.1% and 6.4% in patients aged 18-40 years; and 7.1% and 3.3% in patients aged 51 and older.
“Efficacy responses in patients 51 years of age and older were comparable to those in other age groups,” concluded Dr. Alexis, vice chair for diversity and inclusion in the department of dermatology at Weill Cornell. “The safety profile was consistent across age ranges and was similar to that reported previously.”
The investigators found that treatment response to abrocitinib “in the absence or presence of medicated topical therapy was fairly consistent across age groups, showed similar dose-dependency, and importantly, did not show reduced efficacy in older adults as measured by lesional severity, extent, and itch at 4 months,” said Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study.
“Furthermore, the safety profile was consistent across all adults, though notably, nausea was more common among younger age groups, highlighting an area of future investigation,” he added. “Overall, these data show that abrocitinib is associated with similar short-term responses across adulthood and underscore the importance of the JAK-STAT pathway in the underlying pathophysiology of AD in different age groups. It will be interesting to see how these data reflect the real-world setting with both short- and long-term outcomes in a heterogeneous patient population.”
In the interview, Dr. Chovatiya said, “the next frontier in personalized therapy for AD involves deeper clinical phenotyping of our patients and a better understanding of how efficacy and safety vary across patient groups.” For example, he noted, “AD in earlier versus later adulthood may be associated with different clinical signs, symptoms, comorbidities, and other measures of patient burden, and thus, may be associated with different treatment responses to systemic therapy.”
Dr. Alexis disclosed that he has served as an adviser to, or has received consulting fees from, Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oréal, BMS, Bausch Health, UCB, Vyne, Arcutis, Janssen, Allergan, Almirall, AbbVie, Sol-Gel, and Amgen.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme.
A version of this article first appeared on Medscape.com.
and was comparable in patients aged 51 years and older, results from a post hoc analysis of four trials showed.
Abrocitinib (Cibinqo) is an oral, once-daily, Janus kinase 1 selective inhibitor that has shown good efficacy and safety as monotherapy or combined with topical therapy for treatment of patients with moderate to severe AD. The agent was approved in mid-December in Europe for the treatment of moderate to severe AD in adults who are candidates for systemic therapy and is currently under review by the Food and Drug Administration.
“We know that responses to, and adverse events associated with, systemic therapies may vary among patients of different ages,” Andrew F. Alexis, MD, MPH, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “The efficacy and safety of abrocitinib monotherapy were previously evaluated in adolescent and adult subpopulations from controlled clinical trials in patients with moderate to severe AD. The objective of the current study was to assess the impact of age on short-term responses to abrocitinib treatment in patients with moderate to severe AD.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, and colleagues performed a post hoc analysis across four randomized, double-blind studies that was stratified by age group: 12-17 years, 18-40 years, 41-50 years, and 51 years and older. Efficacy data were assessed separately for patients in the monotherapy pool and in the JADE COMPARE trial. The monotherapy pool included patients from one phase 2b study and two phase 3 studies who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo monotherapy for 12 weeks (JADE-MONO-1 and JADE-MONO-2).
The JADE COMPARE pool included patients who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo, plus medicated topical therapy for 16 weeks. Data from patients in all four trials were pooled for the analysis of treatment-emergent adverse events. Efficacy points analyzed were the Investigator Global Assessment (IGA) score of 0/1 (clear or almost clear), a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75), or Peak Pruritus Numeric Rating Scale score (PP-NRS4) at week 12 for the monotherapy pool and at week 16 for COMPARE.
In the monotherapy pool, the proportions of patients ages 12-17 years, 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response at 12 weeks were 31.3%, 40.2%, 43.8%, and 50.8% (abrocitinib 200 mg); 22%, 23.7%, 22.4%, and 40.8% (abrocitinib 100 mg); and 8.7%, 8%, 3.3%, and 10% (placebo).
In JADE COMPARE, the proportions of patients aged 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response were 50%, 53.2%, and 34.8% (abrocitinib 200 mg); 36.9%, 37.1%, and 26.1% (abrocitinib 100 mg); and 12%, 11.8%, and 16.7% (placebo) at 16 weeks. Similar trends were observed for EASI-75 and PP-NRS4 responses at 12 weeks.
Across all age groups, the most common treatment-emergent adverse events were infections/infestations and gastrointestinal effects; most cases were mild or moderate. Nausea was more frequent in the two younger age groups and was dose related: For abrocitinib 200 mg and abrocitinib 100 mg, respectively, the rates of nausea were 18.8% and 7.8% in patients aged 12-17 years; 17.1% and 6.4% in patients aged 18-40 years; and 7.1% and 3.3% in patients aged 51 and older.
“Efficacy responses in patients 51 years of age and older were comparable to those in other age groups,” concluded Dr. Alexis, vice chair for diversity and inclusion in the department of dermatology at Weill Cornell. “The safety profile was consistent across age ranges and was similar to that reported previously.”
The investigators found that treatment response to abrocitinib “in the absence or presence of medicated topical therapy was fairly consistent across age groups, showed similar dose-dependency, and importantly, did not show reduced efficacy in older adults as measured by lesional severity, extent, and itch at 4 months,” said Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study.
“Furthermore, the safety profile was consistent across all adults, though notably, nausea was more common among younger age groups, highlighting an area of future investigation,” he added. “Overall, these data show that abrocitinib is associated with similar short-term responses across adulthood and underscore the importance of the JAK-STAT pathway in the underlying pathophysiology of AD in different age groups. It will be interesting to see how these data reflect the real-world setting with both short- and long-term outcomes in a heterogeneous patient population.”
In the interview, Dr. Chovatiya said, “the next frontier in personalized therapy for AD involves deeper clinical phenotyping of our patients and a better understanding of how efficacy and safety vary across patient groups.” For example, he noted, “AD in earlier versus later adulthood may be associated with different clinical signs, symptoms, comorbidities, and other measures of patient burden, and thus, may be associated with different treatment responses to systemic therapy.”
Dr. Alexis disclosed that he has served as an adviser to, or has received consulting fees from, Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oréal, BMS, Bausch Health, UCB, Vyne, Arcutis, Janssen, Allergan, Almirall, AbbVie, Sol-Gel, and Amgen.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme.
A version of this article first appeared on Medscape.com.
and was comparable in patients aged 51 years and older, results from a post hoc analysis of four trials showed.
Abrocitinib (Cibinqo) is an oral, once-daily, Janus kinase 1 selective inhibitor that has shown good efficacy and safety as monotherapy or combined with topical therapy for treatment of patients with moderate to severe AD. The agent was approved in mid-December in Europe for the treatment of moderate to severe AD in adults who are candidates for systemic therapy and is currently under review by the Food and Drug Administration.
“We know that responses to, and adverse events associated with, systemic therapies may vary among patients of different ages,” Andrew F. Alexis, MD, MPH, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “The efficacy and safety of abrocitinib monotherapy were previously evaluated in adolescent and adult subpopulations from controlled clinical trials in patients with moderate to severe AD. The objective of the current study was to assess the impact of age on short-term responses to abrocitinib treatment in patients with moderate to severe AD.”
Dr. Alexis, professor of clinical dermatology at Weill Cornell Medicine, New York, and colleagues performed a post hoc analysis across four randomized, double-blind studies that was stratified by age group: 12-17 years, 18-40 years, 41-50 years, and 51 years and older. Efficacy data were assessed separately for patients in the monotherapy pool and in the JADE COMPARE trial. The monotherapy pool included patients from one phase 2b study and two phase 3 studies who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo monotherapy for 12 weeks (JADE-MONO-1 and JADE-MONO-2).
The JADE COMPARE pool included patients who received abrocitinib 200 mg, abrocitinib 100 mg, or placebo, plus medicated topical therapy for 16 weeks. Data from patients in all four trials were pooled for the analysis of treatment-emergent adverse events. Efficacy points analyzed were the Investigator Global Assessment (IGA) score of 0/1 (clear or almost clear), a 75% reduction from baseline in the Eczema Area and Severity Index (EASI-75), or Peak Pruritus Numeric Rating Scale score (PP-NRS4) at week 12 for the monotherapy pool and at week 16 for COMPARE.
In the monotherapy pool, the proportions of patients ages 12-17 years, 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response at 12 weeks were 31.3%, 40.2%, 43.8%, and 50.8% (abrocitinib 200 mg); 22%, 23.7%, 22.4%, and 40.8% (abrocitinib 100 mg); and 8.7%, 8%, 3.3%, and 10% (placebo).
In JADE COMPARE, the proportions of patients aged 18-40 years, 41-50 years, and 51 years and older who achieved an IGA 0/1 response were 50%, 53.2%, and 34.8% (abrocitinib 200 mg); 36.9%, 37.1%, and 26.1% (abrocitinib 100 mg); and 12%, 11.8%, and 16.7% (placebo) at 16 weeks. Similar trends were observed for EASI-75 and PP-NRS4 responses at 12 weeks.
Across all age groups, the most common treatment-emergent adverse events were infections/infestations and gastrointestinal effects; most cases were mild or moderate. Nausea was more frequent in the two younger age groups and was dose related: For abrocitinib 200 mg and abrocitinib 100 mg, respectively, the rates of nausea were 18.8% and 7.8% in patients aged 12-17 years; 17.1% and 6.4% in patients aged 18-40 years; and 7.1% and 3.3% in patients aged 51 and older.
“Efficacy responses in patients 51 years of age and older were comparable to those in other age groups,” concluded Dr. Alexis, vice chair for diversity and inclusion in the department of dermatology at Weill Cornell. “The safety profile was consistent across age ranges and was similar to that reported previously.”
The investigators found that treatment response to abrocitinib “in the absence or presence of medicated topical therapy was fairly consistent across age groups, showed similar dose-dependency, and importantly, did not show reduced efficacy in older adults as measured by lesional severity, extent, and itch at 4 months,” said Raj Chovatiya, MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study.
“Furthermore, the safety profile was consistent across all adults, though notably, nausea was more common among younger age groups, highlighting an area of future investigation,” he added. “Overall, these data show that abrocitinib is associated with similar short-term responses across adulthood and underscore the importance of the JAK-STAT pathway in the underlying pathophysiology of AD in different age groups. It will be interesting to see how these data reflect the real-world setting with both short- and long-term outcomes in a heterogeneous patient population.”
In the interview, Dr. Chovatiya said, “the next frontier in personalized therapy for AD involves deeper clinical phenotyping of our patients and a better understanding of how efficacy and safety vary across patient groups.” For example, he noted, “AD in earlier versus later adulthood may be associated with different clinical signs, symptoms, comorbidities, and other measures of patient burden, and thus, may be associated with different treatment responses to systemic therapy.”
Dr. Alexis disclosed that he has served as an adviser to, or has received consulting fees from, Leo, Galderma, Pfizer, Sanofi-Regeneron, Dermavant, Beiersdorf, Valeant, L’Oréal, BMS, Bausch Health, UCB, Vyne, Arcutis, Janssen, Allergan, Almirall, AbbVie, Sol-Gel, and Amgen.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme.
A version of this article first appeared on Medscape.com.
FROM REVOLUTIONIZING AD 2021
CRP elevated in adults with AD and sleep disturbance
and mortality, results from a large cohort analysis showed.
“The implications of these findings are vast,” presenting author Varsha Parthasarathy said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “Poor sleep quality is known to be associated with increased inflammatory markers such as IL-6, IL-17, and CRP, so it is interesting to see this reflected in AD patients with versus without sleep disturbance. Additionally, we know that CRP is a driver of inflammation and is strongly associated with cardiovascular complications such as heart attack and stroke. Therefore, CRP may be a useful prognostic marker in AD patients with sleep disturbances.”
To examine the comorbidity burden of sleep disorders in AD patients and associate findings with inflammatory CRP and cardiovascular comorbidities, Mr. Parthasarathy, a medical student and itch fellow in the department of dermatology at the Johns Hopkins University School of Medicine, Baltimore, and colleagues drew from TriNetX, a health care network of approximately 73 million de-identified medical records in 53 organizations. The years of study were 2015 to 2021. The researchers limited the analysis to adults with at least two instances of International Classification of Diseases, Tenth Revision (ICD-10) code L28 for AD, to capture a population with true AD. Controls were adults without AD who presented for general checkup and were matched to AD patients by age, race, and sex.
The study population consisted of 120,480 AD patients and matched controls. Their mean age was 36 years, 61% were female, and 26% were Black. Compared with controls, AD patients had an increased risk of developing general sleep disorders over the 6-year period (relative risk, 1.10), as well as obstructive sleep apnea (RR, 1.13), insomnia (RR, 1.10), hypersomnia (RR, 1.24), sleep-related movement disorders (RR, 1.36), restless legs syndrome (RR, 1.25), sleep deprivation (RR, 1.36), and unspecified sleep disorders (RR, 1.22).
To examine the association of sleep disturbance with the inflammatory biomarker CRP, the researchers measured CRP levels between these patient groups. They found a substantially higher CRP in AD patients compared with controls (21.2 mg/L vs. 7.6 mg/L, respectively; P < .0001). This finding “is suggestive of a higher level of inflammation in these patients,” Mr. Parthasarathy said. Interestingly, he added, they also found a higher CRP level in AD patients with sleep disturbances compared to AD patients without sleep disturbances (23.3 vs. 20.6 mg/L; P = .02), “also pointing to a higher inflammatory burden in AD patients whose sleep was affected.”
Compared to matched AD patients without sleep disorders, AD patients with sleep disorders were more likely to develop obesity (RR, 2.65), hyperlipidemia (RR, 2.18), type 2 diabetes (RR, 2.45), metabolic syndrome (RR, 4.16), atherosclerosis (RR, 2.42), peripheral vascular disease (RR, 2.47), stroke (RR, 2.37), venous thromboembolism (RR, 2.93), and mortality (hazard ratio, 1.24).
“There is a consequence of not treating patients with atopic dermatitis, especially those patients with sleep disturbance,” the study’s primary author, Shawn G. Kwatra, MD, associate professor of dermatology at Johns Hopkins, told this news organization. “Chronic inflammation can lead to the development of comorbidities, so it is important to offer patients early treatment to reduce their overall inflammation.” He said that he was most surprised by the degree of increased inflammation in the blood of AD as compared to healthy controls. “This likely plays a part in the development of several comorbidities,” he said.
Mr. Parthasarathy acknowledged certain limitations of the study, including the inability to infer causal relationships, as uncontrolled factors may be present. “Additionally, sampling of only patients that have had medical encounters limits the generalizability of the findings,” she said. “However, findings in this large cohort study suggest that clinicians should seek to identify sleep disorders in AD patients and screen for cardiac comorbidities secondary to inflammation in this patient population.”
“There is increased data to suggest that adults with AD, particularly those with more severe disease, may be at an increased risk of cardiovascular disease and the results from [this study] further support the concept of AD as systemic disease,” said Zelma C. Chiesa Fuxench, MD, MSCE, assistant professor of dermatology at the University of Pennsylvania, Philadelphia, who was asked to comment on the study. She cited the large population-based, retrospective design and use of two instances of ICD codes for AD to confirm diagnosis as key strengths of the research. “However, it is unclear if for each patient CRP levels were measured at one single timepoint,” Dr. Chiesa Fuxench said. “For future studies, it would be interesting to see if these levels fluctuate with time and if persistently elevated levels are associated with worse cardiovascular outcomes in this population. More data is needed to better understand the relationship better atopic dermatitis disease severity, impact on sleep, and how this relates to increased systemic inflammation and worse cardiovascular outcomes in this population.”
Dr. Kwatra disclosed support by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number K23AR077073-01A1 and previous funding by the Dermatology Foundation and Skin of Color Society. Dr. Kwatra is also an advisory board member/consultant for AbbVie, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, Novartis Pharmaceuticals Corporation, Pfizer, Regeneron Pharmaceuticals, Sanofi, and Kiniksa Pharmaceuticals and has served as an investigator for Galderma, Pfizer, and Sanofi. Dr. Chiesa Fuxench disclosed research grants from several pharmaceutical companies for work related to AD. She has also served as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, AbbVie, Incyte Corporation, and Pfizer.
A version of this article first appeared on Medscape.com.
and mortality, results from a large cohort analysis showed.
“The implications of these findings are vast,” presenting author Varsha Parthasarathy said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “Poor sleep quality is known to be associated with increased inflammatory markers such as IL-6, IL-17, and CRP, so it is interesting to see this reflected in AD patients with versus without sleep disturbance. Additionally, we know that CRP is a driver of inflammation and is strongly associated with cardiovascular complications such as heart attack and stroke. Therefore, CRP may be a useful prognostic marker in AD patients with sleep disturbances.”
To examine the comorbidity burden of sleep disorders in AD patients and associate findings with inflammatory CRP and cardiovascular comorbidities, Mr. Parthasarathy, a medical student and itch fellow in the department of dermatology at the Johns Hopkins University School of Medicine, Baltimore, and colleagues drew from TriNetX, a health care network of approximately 73 million de-identified medical records in 53 organizations. The years of study were 2015 to 2021. The researchers limited the analysis to adults with at least two instances of International Classification of Diseases, Tenth Revision (ICD-10) code L28 for AD, to capture a population with true AD. Controls were adults without AD who presented for general checkup and were matched to AD patients by age, race, and sex.
The study population consisted of 120,480 AD patients and matched controls. Their mean age was 36 years, 61% were female, and 26% were Black. Compared with controls, AD patients had an increased risk of developing general sleep disorders over the 6-year period (relative risk, 1.10), as well as obstructive sleep apnea (RR, 1.13), insomnia (RR, 1.10), hypersomnia (RR, 1.24), sleep-related movement disorders (RR, 1.36), restless legs syndrome (RR, 1.25), sleep deprivation (RR, 1.36), and unspecified sleep disorders (RR, 1.22).
To examine the association of sleep disturbance with the inflammatory biomarker CRP, the researchers measured CRP levels between these patient groups. They found a substantially higher CRP in AD patients compared with controls (21.2 mg/L vs. 7.6 mg/L, respectively; P < .0001). This finding “is suggestive of a higher level of inflammation in these patients,” Mr. Parthasarathy said. Interestingly, he added, they also found a higher CRP level in AD patients with sleep disturbances compared to AD patients without sleep disturbances (23.3 vs. 20.6 mg/L; P = .02), “also pointing to a higher inflammatory burden in AD patients whose sleep was affected.”
Compared to matched AD patients without sleep disorders, AD patients with sleep disorders were more likely to develop obesity (RR, 2.65), hyperlipidemia (RR, 2.18), type 2 diabetes (RR, 2.45), metabolic syndrome (RR, 4.16), atherosclerosis (RR, 2.42), peripheral vascular disease (RR, 2.47), stroke (RR, 2.37), venous thromboembolism (RR, 2.93), and mortality (hazard ratio, 1.24).
“There is a consequence of not treating patients with atopic dermatitis, especially those patients with sleep disturbance,” the study’s primary author, Shawn G. Kwatra, MD, associate professor of dermatology at Johns Hopkins, told this news organization. “Chronic inflammation can lead to the development of comorbidities, so it is important to offer patients early treatment to reduce their overall inflammation.” He said that he was most surprised by the degree of increased inflammation in the blood of AD as compared to healthy controls. “This likely plays a part in the development of several comorbidities,” he said.
Mr. Parthasarathy acknowledged certain limitations of the study, including the inability to infer causal relationships, as uncontrolled factors may be present. “Additionally, sampling of only patients that have had medical encounters limits the generalizability of the findings,” she said. “However, findings in this large cohort study suggest that clinicians should seek to identify sleep disorders in AD patients and screen for cardiac comorbidities secondary to inflammation in this patient population.”
“There is increased data to suggest that adults with AD, particularly those with more severe disease, may be at an increased risk of cardiovascular disease and the results from [this study] further support the concept of AD as systemic disease,” said Zelma C. Chiesa Fuxench, MD, MSCE, assistant professor of dermatology at the University of Pennsylvania, Philadelphia, who was asked to comment on the study. She cited the large population-based, retrospective design and use of two instances of ICD codes for AD to confirm diagnosis as key strengths of the research. “However, it is unclear if for each patient CRP levels were measured at one single timepoint,” Dr. Chiesa Fuxench said. “For future studies, it would be interesting to see if these levels fluctuate with time and if persistently elevated levels are associated with worse cardiovascular outcomes in this population. More data is needed to better understand the relationship better atopic dermatitis disease severity, impact on sleep, and how this relates to increased systemic inflammation and worse cardiovascular outcomes in this population.”
Dr. Kwatra disclosed support by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number K23AR077073-01A1 and previous funding by the Dermatology Foundation and Skin of Color Society. Dr. Kwatra is also an advisory board member/consultant for AbbVie, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, Novartis Pharmaceuticals Corporation, Pfizer, Regeneron Pharmaceuticals, Sanofi, and Kiniksa Pharmaceuticals and has served as an investigator for Galderma, Pfizer, and Sanofi. Dr. Chiesa Fuxench disclosed research grants from several pharmaceutical companies for work related to AD. She has also served as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, AbbVie, Incyte Corporation, and Pfizer.
A version of this article first appeared on Medscape.com.
and mortality, results from a large cohort analysis showed.
“The implications of these findings are vast,” presenting author Varsha Parthasarathy said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis virtual symposium. “Poor sleep quality is known to be associated with increased inflammatory markers such as IL-6, IL-17, and CRP, so it is interesting to see this reflected in AD patients with versus without sleep disturbance. Additionally, we know that CRP is a driver of inflammation and is strongly associated with cardiovascular complications such as heart attack and stroke. Therefore, CRP may be a useful prognostic marker in AD patients with sleep disturbances.”
To examine the comorbidity burden of sleep disorders in AD patients and associate findings with inflammatory CRP and cardiovascular comorbidities, Mr. Parthasarathy, a medical student and itch fellow in the department of dermatology at the Johns Hopkins University School of Medicine, Baltimore, and colleagues drew from TriNetX, a health care network of approximately 73 million de-identified medical records in 53 organizations. The years of study were 2015 to 2021. The researchers limited the analysis to adults with at least two instances of International Classification of Diseases, Tenth Revision (ICD-10) code L28 for AD, to capture a population with true AD. Controls were adults without AD who presented for general checkup and were matched to AD patients by age, race, and sex.
The study population consisted of 120,480 AD patients and matched controls. Their mean age was 36 years, 61% were female, and 26% were Black. Compared with controls, AD patients had an increased risk of developing general sleep disorders over the 6-year period (relative risk, 1.10), as well as obstructive sleep apnea (RR, 1.13), insomnia (RR, 1.10), hypersomnia (RR, 1.24), sleep-related movement disorders (RR, 1.36), restless legs syndrome (RR, 1.25), sleep deprivation (RR, 1.36), and unspecified sleep disorders (RR, 1.22).
To examine the association of sleep disturbance with the inflammatory biomarker CRP, the researchers measured CRP levels between these patient groups. They found a substantially higher CRP in AD patients compared with controls (21.2 mg/L vs. 7.6 mg/L, respectively; P < .0001). This finding “is suggestive of a higher level of inflammation in these patients,” Mr. Parthasarathy said. Interestingly, he added, they also found a higher CRP level in AD patients with sleep disturbances compared to AD patients without sleep disturbances (23.3 vs. 20.6 mg/L; P = .02), “also pointing to a higher inflammatory burden in AD patients whose sleep was affected.”
Compared to matched AD patients without sleep disorders, AD patients with sleep disorders were more likely to develop obesity (RR, 2.65), hyperlipidemia (RR, 2.18), type 2 diabetes (RR, 2.45), metabolic syndrome (RR, 4.16), atherosclerosis (RR, 2.42), peripheral vascular disease (RR, 2.47), stroke (RR, 2.37), venous thromboembolism (RR, 2.93), and mortality (hazard ratio, 1.24).
“There is a consequence of not treating patients with atopic dermatitis, especially those patients with sleep disturbance,” the study’s primary author, Shawn G. Kwatra, MD, associate professor of dermatology at Johns Hopkins, told this news organization. “Chronic inflammation can lead to the development of comorbidities, so it is important to offer patients early treatment to reduce their overall inflammation.” He said that he was most surprised by the degree of increased inflammation in the blood of AD as compared to healthy controls. “This likely plays a part in the development of several comorbidities,” he said.
Mr. Parthasarathy acknowledged certain limitations of the study, including the inability to infer causal relationships, as uncontrolled factors may be present. “Additionally, sampling of only patients that have had medical encounters limits the generalizability of the findings,” she said. “However, findings in this large cohort study suggest that clinicians should seek to identify sleep disorders in AD patients and screen for cardiac comorbidities secondary to inflammation in this patient population.”
“There is increased data to suggest that adults with AD, particularly those with more severe disease, may be at an increased risk of cardiovascular disease and the results from [this study] further support the concept of AD as systemic disease,” said Zelma C. Chiesa Fuxench, MD, MSCE, assistant professor of dermatology at the University of Pennsylvania, Philadelphia, who was asked to comment on the study. She cited the large population-based, retrospective design and use of two instances of ICD codes for AD to confirm diagnosis as key strengths of the research. “However, it is unclear if for each patient CRP levels were measured at one single timepoint,” Dr. Chiesa Fuxench said. “For future studies, it would be interesting to see if these levels fluctuate with time and if persistently elevated levels are associated with worse cardiovascular outcomes in this population. More data is needed to better understand the relationship better atopic dermatitis disease severity, impact on sleep, and how this relates to increased systemic inflammation and worse cardiovascular outcomes in this population.”
Dr. Kwatra disclosed support by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number K23AR077073-01A1 and previous funding by the Dermatology Foundation and Skin of Color Society. Dr. Kwatra is also an advisory board member/consultant for AbbVie, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, Novartis Pharmaceuticals Corporation, Pfizer, Regeneron Pharmaceuticals, Sanofi, and Kiniksa Pharmaceuticals and has served as an investigator for Galderma, Pfizer, and Sanofi. Dr. Chiesa Fuxench disclosed research grants from several pharmaceutical companies for work related to AD. She has also served as a consultant for the Asthma and Allergy Foundation of America, National Eczema Association, AbbVie, Incyte Corporation, and Pfizer.
A version of this article first appeared on Medscape.com.
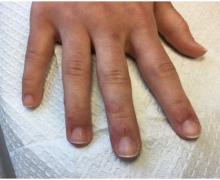
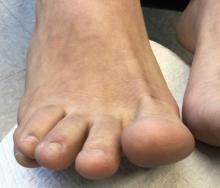
A 22-year-old presented with erythematous papules on her fingers and toes
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
Abrocitinib approved for atopic dermatitis in Europe
who are candidates for systemic therapy, the manufacturer announced.
Approval by the European Commission was based on the results of studies that include four phase 3 clinical trials (JADE MONO-1, JADE-MONO-2, JADE COMPARE, JADE REGIMEN) and an ongoing open-label extension study (JADE EXTEND) in over 2,800 patients, according to the Pfizer press release announcing the approval. The approved doses are 100 and 200 mg a day; a 50-mg dose was approved for patients with moderate and severe renal impairment and “ certain patients receiving treatment with inhibitors of cytochrome P450 (CYP) 2C19,” the release said.
The approval follows a positive opinion by the Committee for Medicinal Products for Human Use of the European Medicines Agency supporting marketing authorization for treating AD, issued in October. It will be marketed as Cibinqo.
Abrocitinib is under review at the Food and Drug Administration. It was approved earlier in 2021 for treating AD in the United Kingdom, Japan, and Korea.
emechcatie@mdedge.com
who are candidates for systemic therapy, the manufacturer announced.
Approval by the European Commission was based on the results of studies that include four phase 3 clinical trials (JADE MONO-1, JADE-MONO-2, JADE COMPARE, JADE REGIMEN) and an ongoing open-label extension study (JADE EXTEND) in over 2,800 patients, according to the Pfizer press release announcing the approval. The approved doses are 100 and 200 mg a day; a 50-mg dose was approved for patients with moderate and severe renal impairment and “ certain patients receiving treatment with inhibitors of cytochrome P450 (CYP) 2C19,” the release said.
The approval follows a positive opinion by the Committee for Medicinal Products for Human Use of the European Medicines Agency supporting marketing authorization for treating AD, issued in October. It will be marketed as Cibinqo.
Abrocitinib is under review at the Food and Drug Administration. It was approved earlier in 2021 for treating AD in the United Kingdom, Japan, and Korea.
emechcatie@mdedge.com
who are candidates for systemic therapy, the manufacturer announced.
Approval by the European Commission was based on the results of studies that include four phase 3 clinical trials (JADE MONO-1, JADE-MONO-2, JADE COMPARE, JADE REGIMEN) and an ongoing open-label extension study (JADE EXTEND) in over 2,800 patients, according to the Pfizer press release announcing the approval. The approved doses are 100 and 200 mg a day; a 50-mg dose was approved for patients with moderate and severe renal impairment and “ certain patients receiving treatment with inhibitors of cytochrome P450 (CYP) 2C19,” the release said.
The approval follows a positive opinion by the Committee for Medicinal Products for Human Use of the European Medicines Agency supporting marketing authorization for treating AD, issued in October. It will be marketed as Cibinqo.
Abrocitinib is under review at the Food and Drug Administration. It was approved earlier in 2021 for treating AD in the United Kingdom, Japan, and Korea.
emechcatie@mdedge.com
What is the diagnosis?
As the lesion was growing, getting more violaceous and indurated, a biopsy was performed. The biopsy showed multiple discrete lobules of dermal capillaries with slight extension into the superficial subcutis. Capillary lobules demonstrate the “cannonball-like” architecture often associated with tufted angioma, and some lobules showed bulging into adjacent thin-walled vessels. Spindled endothelial cells lining slit-like vessels were present in the mid dermis, although this comprises a minority of the lesion. The majority of the subcutis was uninvolved. The findings are overall most consistent with a tufted angioma.
Kaposiform hemangioendothelioma (KHE) has been considered given the presence of occasional slit-like vascular spaces; however, the lesion is predominantly superficial and therefore the lesion is best classified as tufted angioma. GLUT–1 staining was negative.
At the time of biopsy, blood work was ordered, which showed a normal complete blood count with normal number of platelets, slightly elevated D-dimer, and slightly low fibrinogen. Several repeat blood counts and coagulation tests once a week for a few weeks revealed no changes.
The patient was started on aspirin at a dose of 5 mg/kg per day. After a week on the medication the lesion was starting to get smaller and less red.
Tufted angiomas are a rare type of vascular tumor within the spectrum of kaposiform hemangioendotheliomas. Most cases present within the first year of life; some occur at birth. They usually present as papules, plaques, or erythematous, violaceous indurated nodules on the face, neck, trunk, and extremities. The lesions can also be present with hyperhidrosis and hypertrichosis. Clinically, the lesions will have to be differentiated from other vascular tumors such as infantile hemangiomas, congenital hemangiomas, and Kaposi’s sarcoma, as well as subcutaneous fat necrosis of the newborn, cellulitis, and nonaccidental trauma.
Pathogenesis of tufted angiomas is poorly understood. A recent case report found a somatic mutation on GNA14.This protein regulates Ras activity and modulates endothelial cell permeability and migration in response to FGF2 and VEGFA. The p.205L mutation causes activation of GNA14, which upregulates pERK-MAPK pathway, suggesting MAPK inhibition as a potential target for therapy. Clinically, tufted angioma can present in three patterns: uncomplicated tufted angioma (most common type); tufted angioma without thrombocytopenia but with chronic coagulopathy, as it was seen in our patient; and tufted angioma associated with Kasabach-Merritt phenomenon (KMP). KMP is characterized by thrombocytopenia in association with microangiopathic hemolytic anemia, consumptive coagulopathy, and enlarging vascular tumor. Treatment of uncomplicated tufted angioma will depend on symptomatology, size, and location of the lesion. Smaller lesions in noncosmetically sensitive areas can be treated with surgical excision. Cases that are not amenable to excision can be treated with aspirin. There are also reports of response to topical modalities including tacrolimus and timolol. For complicated cases associated with KMP, sirolimus, systemic corticosteroids, ticlopidine, interferon, or vincristine are recommended. Some lesions may demonstrate spontaneous regression.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Cohen S et al. Dermatol Online J. 2019 Sep 15;25(9):13030/qt6pv254mc.
Lim YH et al. Pediatr Dermatol. 2019 Nov;36(6):963-4.
Prasuna A, Rao PN. Indian Dermatol Online J. 2015;6:266-8.
As the lesion was growing, getting more violaceous and indurated, a biopsy was performed. The biopsy showed multiple discrete lobules of dermal capillaries with slight extension into the superficial subcutis. Capillary lobules demonstrate the “cannonball-like” architecture often associated with tufted angioma, and some lobules showed bulging into adjacent thin-walled vessels. Spindled endothelial cells lining slit-like vessels were present in the mid dermis, although this comprises a minority of the lesion. The majority of the subcutis was uninvolved. The findings are overall most consistent with a tufted angioma.
Kaposiform hemangioendothelioma (KHE) has been considered given the presence of occasional slit-like vascular spaces; however, the lesion is predominantly superficial and therefore the lesion is best classified as tufted angioma. GLUT–1 staining was negative.
At the time of biopsy, blood work was ordered, which showed a normal complete blood count with normal number of platelets, slightly elevated D-dimer, and slightly low fibrinogen. Several repeat blood counts and coagulation tests once a week for a few weeks revealed no changes.
The patient was started on aspirin at a dose of 5 mg/kg per day. After a week on the medication the lesion was starting to get smaller and less red.
Tufted angiomas are a rare type of vascular tumor within the spectrum of kaposiform hemangioendotheliomas. Most cases present within the first year of life; some occur at birth. They usually present as papules, plaques, or erythematous, violaceous indurated nodules on the face, neck, trunk, and extremities. The lesions can also be present with hyperhidrosis and hypertrichosis. Clinically, the lesions will have to be differentiated from other vascular tumors such as infantile hemangiomas, congenital hemangiomas, and Kaposi’s sarcoma, as well as subcutaneous fat necrosis of the newborn, cellulitis, and nonaccidental trauma.
Pathogenesis of tufted angiomas is poorly understood. A recent case report found a somatic mutation on GNA14.This protein regulates Ras activity and modulates endothelial cell permeability and migration in response to FGF2 and VEGFA. The p.205L mutation causes activation of GNA14, which upregulates pERK-MAPK pathway, suggesting MAPK inhibition as a potential target for therapy. Clinically, tufted angioma can present in three patterns: uncomplicated tufted angioma (most common type); tufted angioma without thrombocytopenia but with chronic coagulopathy, as it was seen in our patient; and tufted angioma associated with Kasabach-Merritt phenomenon (KMP). KMP is characterized by thrombocytopenia in association with microangiopathic hemolytic anemia, consumptive coagulopathy, and enlarging vascular tumor. Treatment of uncomplicated tufted angioma will depend on symptomatology, size, and location of the lesion. Smaller lesions in noncosmetically sensitive areas can be treated with surgical excision. Cases that are not amenable to excision can be treated with aspirin. There are also reports of response to topical modalities including tacrolimus and timolol. For complicated cases associated with KMP, sirolimus, systemic corticosteroids, ticlopidine, interferon, or vincristine are recommended. Some lesions may demonstrate spontaneous regression.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Cohen S et al. Dermatol Online J. 2019 Sep 15;25(9):13030/qt6pv254mc.
Lim YH et al. Pediatr Dermatol. 2019 Nov;36(6):963-4.
Prasuna A, Rao PN. Indian Dermatol Online J. 2015;6:266-8.
As the lesion was growing, getting more violaceous and indurated, a biopsy was performed. The biopsy showed multiple discrete lobules of dermal capillaries with slight extension into the superficial subcutis. Capillary lobules demonstrate the “cannonball-like” architecture often associated with tufted angioma, and some lobules showed bulging into adjacent thin-walled vessels. Spindled endothelial cells lining slit-like vessels were present in the mid dermis, although this comprises a minority of the lesion. The majority of the subcutis was uninvolved. The findings are overall most consistent with a tufted angioma.
Kaposiform hemangioendothelioma (KHE) has been considered given the presence of occasional slit-like vascular spaces; however, the lesion is predominantly superficial and therefore the lesion is best classified as tufted angioma. GLUT–1 staining was negative.
At the time of biopsy, blood work was ordered, which showed a normal complete blood count with normal number of platelets, slightly elevated D-dimer, and slightly low fibrinogen. Several repeat blood counts and coagulation tests once a week for a few weeks revealed no changes.
The patient was started on aspirin at a dose of 5 mg/kg per day. After a week on the medication the lesion was starting to get smaller and less red.
Tufted angiomas are a rare type of vascular tumor within the spectrum of kaposiform hemangioendotheliomas. Most cases present within the first year of life; some occur at birth. They usually present as papules, plaques, or erythematous, violaceous indurated nodules on the face, neck, trunk, and extremities. The lesions can also be present with hyperhidrosis and hypertrichosis. Clinically, the lesions will have to be differentiated from other vascular tumors such as infantile hemangiomas, congenital hemangiomas, and Kaposi’s sarcoma, as well as subcutaneous fat necrosis of the newborn, cellulitis, and nonaccidental trauma.
Pathogenesis of tufted angiomas is poorly understood. A recent case report found a somatic mutation on GNA14.This protein regulates Ras activity and modulates endothelial cell permeability and migration in response to FGF2 and VEGFA. The p.205L mutation causes activation of GNA14, which upregulates pERK-MAPK pathway, suggesting MAPK inhibition as a potential target for therapy. Clinically, tufted angioma can present in three patterns: uncomplicated tufted angioma (most common type); tufted angioma without thrombocytopenia but with chronic coagulopathy, as it was seen in our patient; and tufted angioma associated with Kasabach-Merritt phenomenon (KMP). KMP is characterized by thrombocytopenia in association with microangiopathic hemolytic anemia, consumptive coagulopathy, and enlarging vascular tumor. Treatment of uncomplicated tufted angioma will depend on symptomatology, size, and location of the lesion. Smaller lesions in noncosmetically sensitive areas can be treated with surgical excision. Cases that are not amenable to excision can be treated with aspirin. There are also reports of response to topical modalities including tacrolimus and timolol. For complicated cases associated with KMP, sirolimus, systemic corticosteroids, ticlopidine, interferon, or vincristine are recommended. Some lesions may demonstrate spontaneous regression.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Cohen S et al. Dermatol Online J. 2019 Sep 15;25(9):13030/qt6pv254mc.
Lim YH et al. Pediatr Dermatol. 2019 Nov;36(6):963-4.
Prasuna A, Rao PN. Indian Dermatol Online J. 2015;6:266-8.
A 35-day-old female was referred to our pediatric dermatology clinic for evaluation of a red lesion on the right arm. The lesion presented at about 4 days of life as a red plaque (image 1 at 8 days of life).
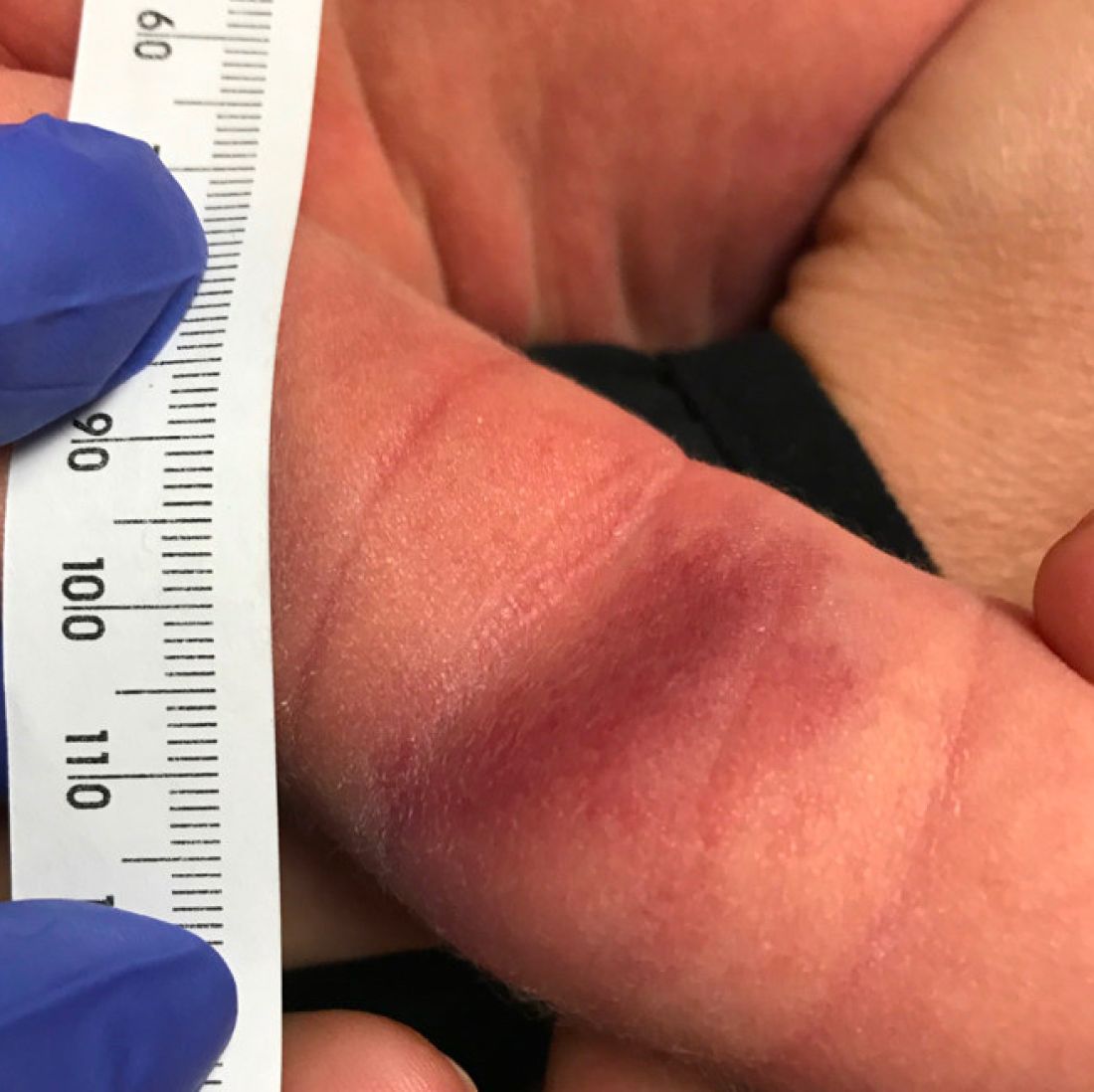
On the following days, the lesion started growing but it didn't seem to be tender or bothersome to the patient (image 2, at 35 days of life).
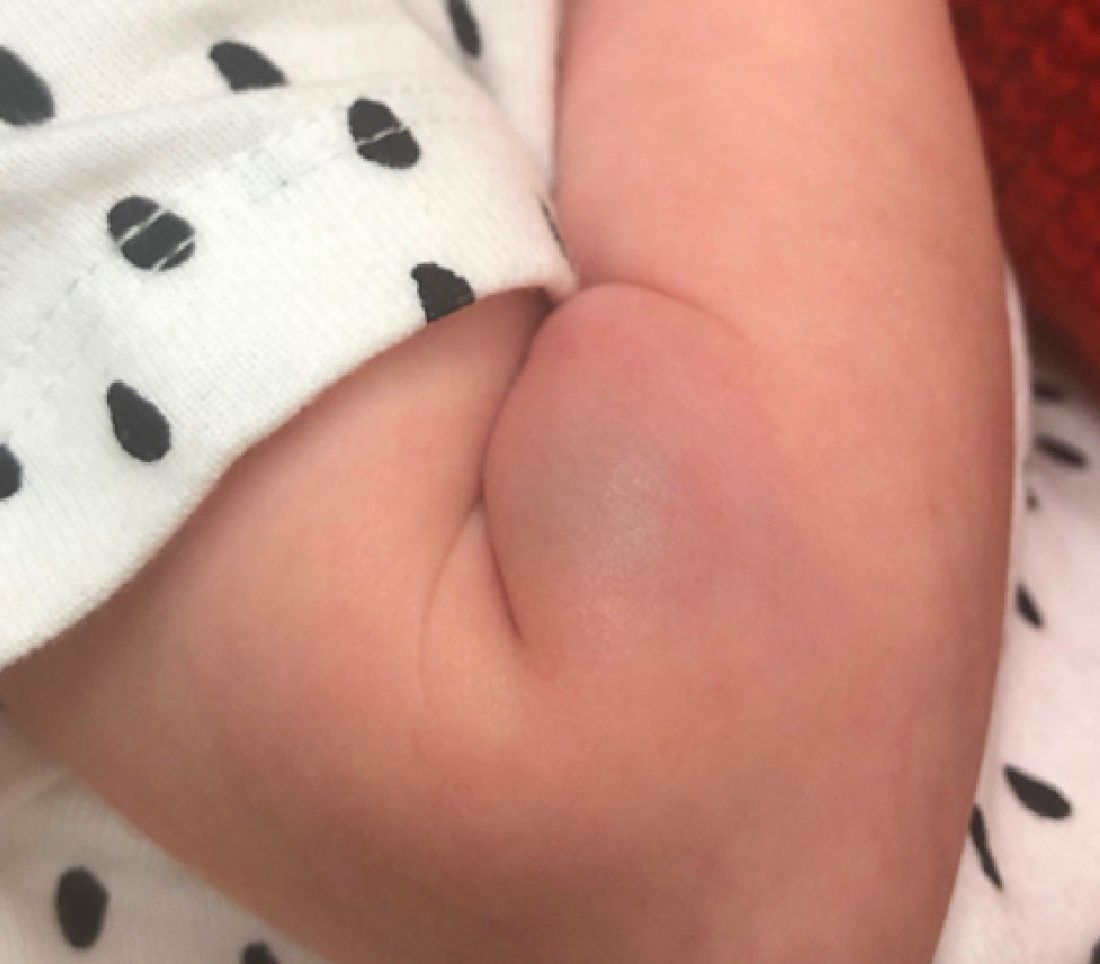
At a 2-week follow up the lesion was getting fuller and more violaceous. There was no history of fever and the lesion didn't appear tender to the touch.
She was born via normal spontaneous vaginal delivery. There were no complications and the mother received prenatal care.
On exam she had a red to violaceous nodule on the right arm (image 3 at 45 days of life).
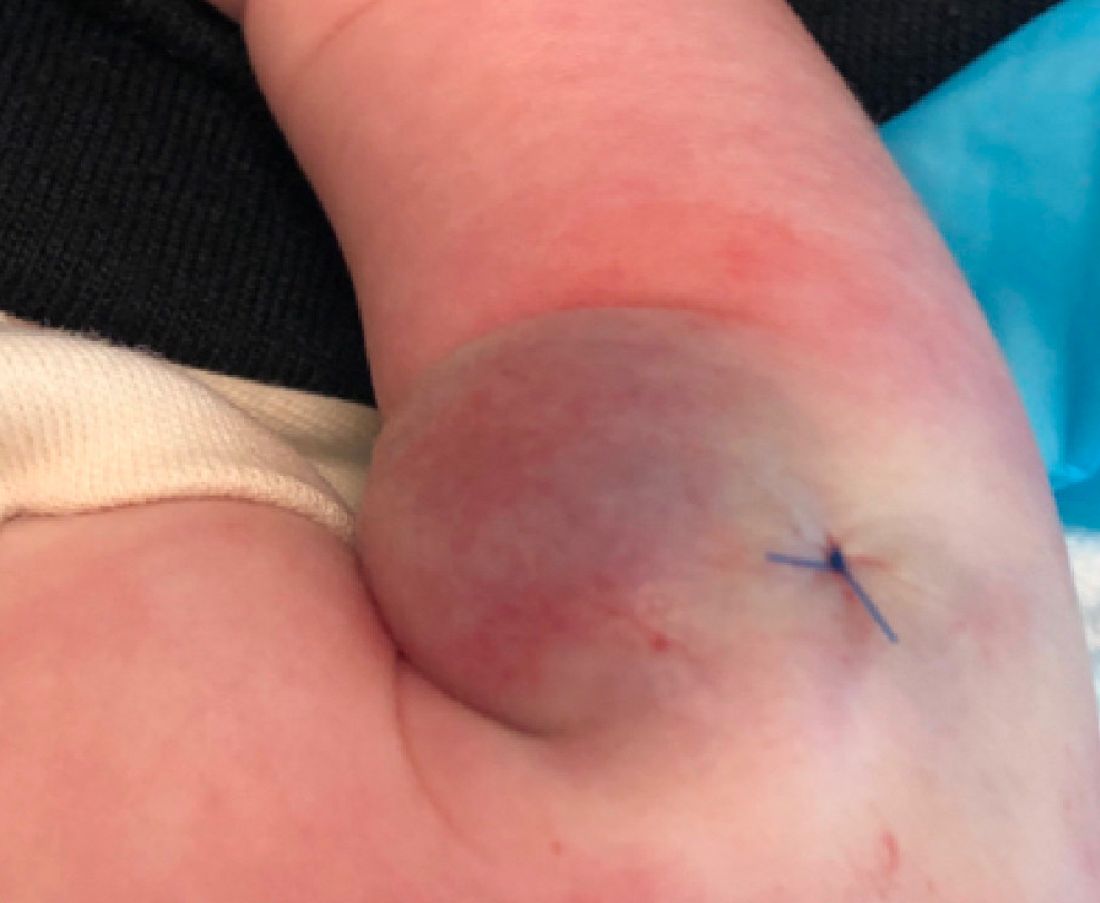
A sun distributed rash

The photo distribution and annular quality of this patient’s rash, combined with his positive autoimmune work-up, led to a diagnosis of subacute cutaneous lupus erythematosus (SCLE), a nonscarring subtype of cutaneous lupus erythematosus.
SCLE is a chronic and relapsing condition that may manifest as either a papulosquamous or annular eruption.1 It most commonly affects areas of sun exposure such as the shoulders, upper back, and extensor surfaces of the arms. This disorder typically affects young or middle-aged women between the ages of 30 and 40 years.
The differential diagnosis of this eruption includes dermatomyositis, polymorphous light eruption, psoriasis, tinea corporis, and other photodermatoses. The etiology of SCLE is multifactorial and may include a genetic susceptibility in combination with environmental triggers that provoke an autoimmune response to sunlight.1 There is strong evidence linking drug-induced SCLE with proton pump inhibitors, anticonvulsants, beta-blockers, terbinafine, and immune modulators.2
As many as 70% of patients with SCLE have positive anti-Ro/SSA autoantibodies, and this is most often associated with Sjogren syndrome.1 Interestingly, SCLE patients often exhibit symptoms that overlap with Sjogren syndrome. Systemic involvement is rare in SCLE, and if present, these symptoms are usually limited to arthritis and myalgia.
Treatment of SCLE includes photo-protective behaviors, topical corticosteroids/calcineurin inhibitors, and systemic therapies such as hydroxychloroquine (first-line), methotrexate, and mycophenolate mofetil (second-line).2
Our patient was started on hydroxychloroquine 200 mg orally bid, with complete resolution of the lesions at his 2 month–follow-up appointment. This case emphasizes the importance of distinguishing SCLE from other subtypes of lupus erythematosus as the prognostic course and treatment varies between these conditions.
Photos courtesy of Kriti Mishra, MD. Text courtesy of Jaimie Lin, BS, Kriti Mishra, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Okon LG, Werth VP. Cutaneous lupus erythematosus: diagnosis and treatment. Best Pract Res Clin Rheumatol. 2013;27:391-404. https://doi.org/10.1016/j.berh.2013.07.008
2. Jatwani S, Hearth Holmes MP. Subacute cutaneous lupus erythematosus. 2021. StatPearls. StatPearls Publishing; 2021.

The photo distribution and annular quality of this patient’s rash, combined with his positive autoimmune work-up, led to a diagnosis of subacute cutaneous lupus erythematosus (SCLE), a nonscarring subtype of cutaneous lupus erythematosus.
SCLE is a chronic and relapsing condition that may manifest as either a papulosquamous or annular eruption.1 It most commonly affects areas of sun exposure such as the shoulders, upper back, and extensor surfaces of the arms. This disorder typically affects young or middle-aged women between the ages of 30 and 40 years.
The differential diagnosis of this eruption includes dermatomyositis, polymorphous light eruption, psoriasis, tinea corporis, and other photodermatoses. The etiology of SCLE is multifactorial and may include a genetic susceptibility in combination with environmental triggers that provoke an autoimmune response to sunlight.1 There is strong evidence linking drug-induced SCLE with proton pump inhibitors, anticonvulsants, beta-blockers, terbinafine, and immune modulators.2
As many as 70% of patients with SCLE have positive anti-Ro/SSA autoantibodies, and this is most often associated with Sjogren syndrome.1 Interestingly, SCLE patients often exhibit symptoms that overlap with Sjogren syndrome. Systemic involvement is rare in SCLE, and if present, these symptoms are usually limited to arthritis and myalgia.
Treatment of SCLE includes photo-protective behaviors, topical corticosteroids/calcineurin inhibitors, and systemic therapies such as hydroxychloroquine (first-line), methotrexate, and mycophenolate mofetil (second-line).2
Our patient was started on hydroxychloroquine 200 mg orally bid, with complete resolution of the lesions at his 2 month–follow-up appointment. This case emphasizes the importance of distinguishing SCLE from other subtypes of lupus erythematosus as the prognostic course and treatment varies between these conditions.
Photos courtesy of Kriti Mishra, MD. Text courtesy of Jaimie Lin, BS, Kriti Mishra, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The photo distribution and annular quality of this patient’s rash, combined with his positive autoimmune work-up, led to a diagnosis of subacute cutaneous lupus erythematosus (SCLE), a nonscarring subtype of cutaneous lupus erythematosus.
SCLE is a chronic and relapsing condition that may manifest as either a papulosquamous or annular eruption.1 It most commonly affects areas of sun exposure such as the shoulders, upper back, and extensor surfaces of the arms. This disorder typically affects young or middle-aged women between the ages of 30 and 40 years.
The differential diagnosis of this eruption includes dermatomyositis, polymorphous light eruption, psoriasis, tinea corporis, and other photodermatoses. The etiology of SCLE is multifactorial and may include a genetic susceptibility in combination with environmental triggers that provoke an autoimmune response to sunlight.1 There is strong evidence linking drug-induced SCLE with proton pump inhibitors, anticonvulsants, beta-blockers, terbinafine, and immune modulators.2
As many as 70% of patients with SCLE have positive anti-Ro/SSA autoantibodies, and this is most often associated with Sjogren syndrome.1 Interestingly, SCLE patients often exhibit symptoms that overlap with Sjogren syndrome. Systemic involvement is rare in SCLE, and if present, these symptoms are usually limited to arthritis and myalgia.
Treatment of SCLE includes photo-protective behaviors, topical corticosteroids/calcineurin inhibitors, and systemic therapies such as hydroxychloroquine (first-line), methotrexate, and mycophenolate mofetil (second-line).2
Our patient was started on hydroxychloroquine 200 mg orally bid, with complete resolution of the lesions at his 2 month–follow-up appointment. This case emphasizes the importance of distinguishing SCLE from other subtypes of lupus erythematosus as the prognostic course and treatment varies between these conditions.
Photos courtesy of Kriti Mishra, MD. Text courtesy of Jaimie Lin, BS, Kriti Mishra, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Okon LG, Werth VP. Cutaneous lupus erythematosus: diagnosis and treatment. Best Pract Res Clin Rheumatol. 2013;27:391-404. https://doi.org/10.1016/j.berh.2013.07.008
2. Jatwani S, Hearth Holmes MP. Subacute cutaneous lupus erythematosus. 2021. StatPearls. StatPearls Publishing; 2021.
1. Okon LG, Werth VP. Cutaneous lupus erythematosus: diagnosis and treatment. Best Pract Res Clin Rheumatol. 2013;27:391-404. https://doi.org/10.1016/j.berh.2013.07.008
2. Jatwani S, Hearth Holmes MP. Subacute cutaneous lupus erythematosus. 2021. StatPearls. StatPearls Publishing; 2021.
FDA expands pembrolizumab approval for advanced melanoma
The over age 12 years. The FDA also extended the approval to those with stage III disease.
The FDA approval on Dec. 3 was based on first interim findings from the randomized, placebo-controlled KEYNOTE-716 trial, which evaluated patients with stage IIB and IIC disease.
Since the anti-PD-1 therapy was approved in metastatic melanoma 7 years ago, “we have built on this foundation in melanoma and have expanded the use of KEYTRUDA into earlier stages of this disease,” said Scot Ebbinghaus, MD, vice president, clinical research, Merck Research Laboratories, in a press release. “With today’s approval, we can now offer health care providers and patients 12 years and older the opportunity to help prevent melanoma recurrence with Keytruda across resected stage IIB, stage IIC, and stage III melanoma.”
In KEYNOTE-716, patients with completely resected stage IIB or IIC melanoma were randomly assigned to receive 200 mg of intravenous pembrolizumab, the pediatric dose 2 mg/kg (up to a maximum of 200 mg) every 3 weeks, or placebo for up to 1 year until disease recurrence or unacceptable toxicity.
After a median follow-up of 14.4 months, investigators reported a statistically significant 35% improvement in recurrence-free survival (RFS) in those treated with pembrolizumab, compared with those who received placebo (hazard ratio, 0.65).
The most common adverse reactions reported in patients receiving pembrolizumab in KEYNOTE-716 were fatigue, diarrhea, pruritus, and arthralgia, each occurring in at least 20% of patients.
“Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of Keytruda,” according to Merck.
A version of this article first appeared on Medscape.com.
The over age 12 years. The FDA also extended the approval to those with stage III disease.
The FDA approval on Dec. 3 was based on first interim findings from the randomized, placebo-controlled KEYNOTE-716 trial, which evaluated patients with stage IIB and IIC disease.
Since the anti-PD-1 therapy was approved in metastatic melanoma 7 years ago, “we have built on this foundation in melanoma and have expanded the use of KEYTRUDA into earlier stages of this disease,” said Scot Ebbinghaus, MD, vice president, clinical research, Merck Research Laboratories, in a press release. “With today’s approval, we can now offer health care providers and patients 12 years and older the opportunity to help prevent melanoma recurrence with Keytruda across resected stage IIB, stage IIC, and stage III melanoma.”
In KEYNOTE-716, patients with completely resected stage IIB or IIC melanoma were randomly assigned to receive 200 mg of intravenous pembrolizumab, the pediatric dose 2 mg/kg (up to a maximum of 200 mg) every 3 weeks, or placebo for up to 1 year until disease recurrence or unacceptable toxicity.
After a median follow-up of 14.4 months, investigators reported a statistically significant 35% improvement in recurrence-free survival (RFS) in those treated with pembrolizumab, compared with those who received placebo (hazard ratio, 0.65).
The most common adverse reactions reported in patients receiving pembrolizumab in KEYNOTE-716 were fatigue, diarrhea, pruritus, and arthralgia, each occurring in at least 20% of patients.
“Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of Keytruda,” according to Merck.
A version of this article first appeared on Medscape.com.
The over age 12 years. The FDA also extended the approval to those with stage III disease.
The FDA approval on Dec. 3 was based on first interim findings from the randomized, placebo-controlled KEYNOTE-716 trial, which evaluated patients with stage IIB and IIC disease.
Since the anti-PD-1 therapy was approved in metastatic melanoma 7 years ago, “we have built on this foundation in melanoma and have expanded the use of KEYTRUDA into earlier stages of this disease,” said Scot Ebbinghaus, MD, vice president, clinical research, Merck Research Laboratories, in a press release. “With today’s approval, we can now offer health care providers and patients 12 years and older the opportunity to help prevent melanoma recurrence with Keytruda across resected stage IIB, stage IIC, and stage III melanoma.”
In KEYNOTE-716, patients with completely resected stage IIB or IIC melanoma were randomly assigned to receive 200 mg of intravenous pembrolizumab, the pediatric dose 2 mg/kg (up to a maximum of 200 mg) every 3 weeks, or placebo for up to 1 year until disease recurrence or unacceptable toxicity.
After a median follow-up of 14.4 months, investigators reported a statistically significant 35% improvement in recurrence-free survival (RFS) in those treated with pembrolizumab, compared with those who received placebo (hazard ratio, 0.65).
The most common adverse reactions reported in patients receiving pembrolizumab in KEYNOTE-716 were fatigue, diarrhea, pruritus, and arthralgia, each occurring in at least 20% of patients.
“Early identification and management of immune-mediated adverse reactions are essential to ensure safe use of Keytruda,” according to Merck.
A version of this article first appeared on Medscape.com.
Pink or red actinic keratoses signal more inflammation
lesions.
Data suggest that up to 65% of squamous cell carcinomas (SCCs) were at some point diagnosed as AKs, wrote Jessica G. Labadie, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues. Early identification of AKs is important to prevent progression to SCCs; previous studies have used histology or morphology, but not color, to characterize AK lesions, they said. In the study published in Dermatologic Surgery, the researchers analyzed images and histopathology slides to characterize AKs by color and to examine associations with inflammation and vasculature. They identified AKs from patients diagnosed between January 2018 and October 2019. The lesions were classified as white (4), brown (9), red (15), or pink (21).
Overall, white AKs had an absence of erythema and were significantly less likely to show inflammation on histopathology, compared with other colors. Brown AKs showed no significant increase in vascularity, but were significantly associated with pigment incontinence, basilar pigment presence, and absence of inflammation.
Notably, dermoscopy of red AKs revealed a distinctive polymorphous vessel pattern in most samples, as well as erythema in all. Similarly, all pink AKs showed erythema, and all showed inflammatory infiltrate on histology, although most did not show increased vascularity.
For all colors of AKs, there was a significant association between the presence of erythema on dermoscopy and the presence of inflammation on histology, while the absence of erythema on dermoscopy corresponded to a significant absence of inflammation on histology, the researchers noted.
“This report adds to the armamentarium of a dermatologist by proposing a novel way to characterize AK lesions,” the researchers wrote.
The study findings were limited by several factors including the inability to confirm which AKs would transform into SCCs based on color, and the inclusion of a study population with advanced AKs that may not generalize to typical AKs, the researchers noted. More research is needed to explore the impact of AK color on SCC development, they emphasized.
However, the results represent a novel way to characterize AK lesions, and triage them in a way that may spare some patients from unneeded biopsies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
lesions.
Data suggest that up to 65% of squamous cell carcinomas (SCCs) were at some point diagnosed as AKs, wrote Jessica G. Labadie, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues. Early identification of AKs is important to prevent progression to SCCs; previous studies have used histology or morphology, but not color, to characterize AK lesions, they said. In the study published in Dermatologic Surgery, the researchers analyzed images and histopathology slides to characterize AKs by color and to examine associations with inflammation and vasculature. They identified AKs from patients diagnosed between January 2018 and October 2019. The lesions were classified as white (4), brown (9), red (15), or pink (21).
Overall, white AKs had an absence of erythema and were significantly less likely to show inflammation on histopathology, compared with other colors. Brown AKs showed no significant increase in vascularity, but were significantly associated with pigment incontinence, basilar pigment presence, and absence of inflammation.
Notably, dermoscopy of red AKs revealed a distinctive polymorphous vessel pattern in most samples, as well as erythema in all. Similarly, all pink AKs showed erythema, and all showed inflammatory infiltrate on histology, although most did not show increased vascularity.
For all colors of AKs, there was a significant association between the presence of erythema on dermoscopy and the presence of inflammation on histology, while the absence of erythema on dermoscopy corresponded to a significant absence of inflammation on histology, the researchers noted.
“This report adds to the armamentarium of a dermatologist by proposing a novel way to characterize AK lesions,” the researchers wrote.
The study findings were limited by several factors including the inability to confirm which AKs would transform into SCCs based on color, and the inclusion of a study population with advanced AKs that may not generalize to typical AKs, the researchers noted. More research is needed to explore the impact of AK color on SCC development, they emphasized.
However, the results represent a novel way to characterize AK lesions, and triage them in a way that may spare some patients from unneeded biopsies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
lesions.
Data suggest that up to 65% of squamous cell carcinomas (SCCs) were at some point diagnosed as AKs, wrote Jessica G. Labadie, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues. Early identification of AKs is important to prevent progression to SCCs; previous studies have used histology or morphology, but not color, to characterize AK lesions, they said. In the study published in Dermatologic Surgery, the researchers analyzed images and histopathology slides to characterize AKs by color and to examine associations with inflammation and vasculature. They identified AKs from patients diagnosed between January 2018 and October 2019. The lesions were classified as white (4), brown (9), red (15), or pink (21).
Overall, white AKs had an absence of erythema and were significantly less likely to show inflammation on histopathology, compared with other colors. Brown AKs showed no significant increase in vascularity, but were significantly associated with pigment incontinence, basilar pigment presence, and absence of inflammation.
Notably, dermoscopy of red AKs revealed a distinctive polymorphous vessel pattern in most samples, as well as erythema in all. Similarly, all pink AKs showed erythema, and all showed inflammatory infiltrate on histology, although most did not show increased vascularity.
For all colors of AKs, there was a significant association between the presence of erythema on dermoscopy and the presence of inflammation on histology, while the absence of erythema on dermoscopy corresponded to a significant absence of inflammation on histology, the researchers noted.
“This report adds to the armamentarium of a dermatologist by proposing a novel way to characterize AK lesions,” the researchers wrote.
The study findings were limited by several factors including the inability to confirm which AKs would transform into SCCs based on color, and the inclusion of a study population with advanced AKs that may not generalize to typical AKs, the researchers noted. More research is needed to explore the impact of AK color on SCC development, they emphasized.
However, the results represent a novel way to characterize AK lesions, and triage them in a way that may spare some patients from unneeded biopsies, they concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM DERMATOLOGIC SURGERY