User login
Longitudinal course of atopic dermatitis often overlooked, expert says
In the opinion of Raj Chovatiya, MD, PhD, the longitudinal course of atopic dermatitis (AD) is an important yet overlooked clinical domain of the disease.
“We know that AD is associated with fluctuating severity, disease flares, long-term persistence, and periods of quiescence, but its longitudinal course is not routinely incorporated into guidelines or clinical trials,” Dr. Chovatiya, assistant professor in the department of dermatology at Northwestern University, Chicago, said during the Revolutionizing Atopic Dermatitis virtual symposium. “Understanding the long-term course may improve our ability to phenotype, prognosticate, and personalize our care.”
The classic view of AD is that it starts in early childhood, follows a waxing and waning course for a few years, and burns out by adulthood. “I think we all know that this is generally false,” he said. “This was largely based on anecdotal clinical experience and large cross-sectional studies, not ones that consider the heterogeneity of AD.”
Results from a large-scale, prospective study of 7,157 children enrolled in the Pediatric Eczema Elective Registry (PEER), suggests that AD commonly persists beyond adulthood. PEER was a phase IV postmarketing safety study of children aged 12-17 with moderate to severe AD who were exposed to topical pimecrolimus and who were surveyed every 6 months (JAMA Dermatol. 2014;150[6]:593-600). The researchers found that more persistent disease was associated with self-reported disease activity, many environmental exposures, White race, history of AD, and an annual household income of less than $50,000. By age 20, 50% reported at least one 6-month symptom- and medication-free period. “The important takeaway was that at every age, greater than 80% reported active AD as defined by symptoms or medication use, meaning that persistence was extremely high – much higher than what was originally thought,” Dr. Chovatiya said. “If you take a look at the literature before this study, many were retrospective analyses, and persistence was estimated to be in the 40%-60% range.”
International prospective studies have provided a more conservative estimate of persistence. For example, the German Multicenter Allergy Study followed 1,314 from birth through age 7 (J Allergy Clin Immunol. 2004;113[5]:925-31). Of these, 22% had AD within the first 2 years of life. Of these, 43% were in remission by age 3, while 38% had intermittent AD, and 19% had symptoms every year of the study. “Studies of other birth cohorts in the world came out suggesting that the rates of AD persistence ranges in the single digits to the teens,” Dr. Chovatiya said.
To reconcile these heterogeneous estimates of AD persistence, researchers conducted a systematic review and meta-analysis of 45 studies that included 110,651 subjects from 15 countries and spanned 434,992 patient-years (J Am Acad Dermatol. 2016;75:681-7.e11). They found that 80% of childhood AD had at least one observed period of disease clearance by 8 years of age. “Most importantly, less than 5% of childhood AD was persistent 20 years after diagnosis,” Dr. Chovatiya said. “However, interestingly, increased persistence was associated with later onset AD, more years of persistence, and more patient/caregiver-assessed disease.” He pointed out inherent limitations to all studies of AD persistence, including nonuniform methods of data collection, differing cohorts, different ways of diagnosing AD, different disease severity scales, and the fact that most don’t assess flares or recurrence beyond the initial period of disease clearance. “This can lead to a potential underestimation of longer-term persistence,” he said.
Childhood AD features unique predictors of persistence that may define AD trajectories. For example, in several existing studies, more persistent disease was associated with higher baseline severity, earlier-onset AD, personal history of atopy, family history of AD, AD genetic risk score (heritability, including common Filaggrin mutations), urban environment, non-White race, Hispanic ethnicity, female sex, lower household income, and overall poorer health status.
Dr. Chovatiya said. “I think that AD classification can take a lesson from asthma. When we think about how our allergy colleagues think about asthma, it is commonly classified as intermittent, mild persistent, moderate persistent, and severe persistent. Those that have intermittent disease get reactive treatment, while those with persistent disease get proactive treatment. Similarly, AD could be classified as mild intermittent, mild persistent, moderate to severe intermittent and moderate to severe persistent.”
He concluded his presentation by recommending that the fluctuating course of AD be better captured in clinical trials. “Current randomized, controlled trials use validated measures of AD signs and symptoms as inclusion criteria and measures of efficacy,” he said. “Static assessments may confound treatment effects, and assessment of prespecified time points are somewhat arbitrary in the context of disease subsets.” He proposes studies that examine aggregate measures of long-term disease control, such as number of itch-free days, weeks with clear skin, and flares experienced. “Long-term control assessment in RCTs should include signs, symptoms, health-related quality of life, and a patient global domain over time to better understand how AD is doing in the long run,” he said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arcutis, Arena, Incyte, Pfizer, Regeneron, and Sanofi Genzyme.
In the opinion of Raj Chovatiya, MD, PhD, the longitudinal course of atopic dermatitis (AD) is an important yet overlooked clinical domain of the disease.
“We know that AD is associated with fluctuating severity, disease flares, long-term persistence, and periods of quiescence, but its longitudinal course is not routinely incorporated into guidelines or clinical trials,” Dr. Chovatiya, assistant professor in the department of dermatology at Northwestern University, Chicago, said during the Revolutionizing Atopic Dermatitis virtual symposium. “Understanding the long-term course may improve our ability to phenotype, prognosticate, and personalize our care.”
The classic view of AD is that it starts in early childhood, follows a waxing and waning course for a few years, and burns out by adulthood. “I think we all know that this is generally false,” he said. “This was largely based on anecdotal clinical experience and large cross-sectional studies, not ones that consider the heterogeneity of AD.”
Results from a large-scale, prospective study of 7,157 children enrolled in the Pediatric Eczema Elective Registry (PEER), suggests that AD commonly persists beyond adulthood. PEER was a phase IV postmarketing safety study of children aged 12-17 with moderate to severe AD who were exposed to topical pimecrolimus and who were surveyed every 6 months (JAMA Dermatol. 2014;150[6]:593-600). The researchers found that more persistent disease was associated with self-reported disease activity, many environmental exposures, White race, history of AD, and an annual household income of less than $50,000. By age 20, 50% reported at least one 6-month symptom- and medication-free period. “The important takeaway was that at every age, greater than 80% reported active AD as defined by symptoms or medication use, meaning that persistence was extremely high – much higher than what was originally thought,” Dr. Chovatiya said. “If you take a look at the literature before this study, many were retrospective analyses, and persistence was estimated to be in the 40%-60% range.”
International prospective studies have provided a more conservative estimate of persistence. For example, the German Multicenter Allergy Study followed 1,314 from birth through age 7 (J Allergy Clin Immunol. 2004;113[5]:925-31). Of these, 22% had AD within the first 2 years of life. Of these, 43% were in remission by age 3, while 38% had intermittent AD, and 19% had symptoms every year of the study. “Studies of other birth cohorts in the world came out suggesting that the rates of AD persistence ranges in the single digits to the teens,” Dr. Chovatiya said.
To reconcile these heterogeneous estimates of AD persistence, researchers conducted a systematic review and meta-analysis of 45 studies that included 110,651 subjects from 15 countries and spanned 434,992 patient-years (J Am Acad Dermatol. 2016;75:681-7.e11). They found that 80% of childhood AD had at least one observed period of disease clearance by 8 years of age. “Most importantly, less than 5% of childhood AD was persistent 20 years after diagnosis,” Dr. Chovatiya said. “However, interestingly, increased persistence was associated with later onset AD, more years of persistence, and more patient/caregiver-assessed disease.” He pointed out inherent limitations to all studies of AD persistence, including nonuniform methods of data collection, differing cohorts, different ways of diagnosing AD, different disease severity scales, and the fact that most don’t assess flares or recurrence beyond the initial period of disease clearance. “This can lead to a potential underestimation of longer-term persistence,” he said.
Childhood AD features unique predictors of persistence that may define AD trajectories. For example, in several existing studies, more persistent disease was associated with higher baseline severity, earlier-onset AD, personal history of atopy, family history of AD, AD genetic risk score (heritability, including common Filaggrin mutations), urban environment, non-White race, Hispanic ethnicity, female sex, lower household income, and overall poorer health status.
Dr. Chovatiya said. “I think that AD classification can take a lesson from asthma. When we think about how our allergy colleagues think about asthma, it is commonly classified as intermittent, mild persistent, moderate persistent, and severe persistent. Those that have intermittent disease get reactive treatment, while those with persistent disease get proactive treatment. Similarly, AD could be classified as mild intermittent, mild persistent, moderate to severe intermittent and moderate to severe persistent.”
He concluded his presentation by recommending that the fluctuating course of AD be better captured in clinical trials. “Current randomized, controlled trials use validated measures of AD signs and symptoms as inclusion criteria and measures of efficacy,” he said. “Static assessments may confound treatment effects, and assessment of prespecified time points are somewhat arbitrary in the context of disease subsets.” He proposes studies that examine aggregate measures of long-term disease control, such as number of itch-free days, weeks with clear skin, and flares experienced. “Long-term control assessment in RCTs should include signs, symptoms, health-related quality of life, and a patient global domain over time to better understand how AD is doing in the long run,” he said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arcutis, Arena, Incyte, Pfizer, Regeneron, and Sanofi Genzyme.
In the opinion of Raj Chovatiya, MD, PhD, the longitudinal course of atopic dermatitis (AD) is an important yet overlooked clinical domain of the disease.
“We know that AD is associated with fluctuating severity, disease flares, long-term persistence, and periods of quiescence, but its longitudinal course is not routinely incorporated into guidelines or clinical trials,” Dr. Chovatiya, assistant professor in the department of dermatology at Northwestern University, Chicago, said during the Revolutionizing Atopic Dermatitis virtual symposium. “Understanding the long-term course may improve our ability to phenotype, prognosticate, and personalize our care.”
The classic view of AD is that it starts in early childhood, follows a waxing and waning course for a few years, and burns out by adulthood. “I think we all know that this is generally false,” he said. “This was largely based on anecdotal clinical experience and large cross-sectional studies, not ones that consider the heterogeneity of AD.”
Results from a large-scale, prospective study of 7,157 children enrolled in the Pediatric Eczema Elective Registry (PEER), suggests that AD commonly persists beyond adulthood. PEER was a phase IV postmarketing safety study of children aged 12-17 with moderate to severe AD who were exposed to topical pimecrolimus and who were surveyed every 6 months (JAMA Dermatol. 2014;150[6]:593-600). The researchers found that more persistent disease was associated with self-reported disease activity, many environmental exposures, White race, history of AD, and an annual household income of less than $50,000. By age 20, 50% reported at least one 6-month symptom- and medication-free period. “The important takeaway was that at every age, greater than 80% reported active AD as defined by symptoms or medication use, meaning that persistence was extremely high – much higher than what was originally thought,” Dr. Chovatiya said. “If you take a look at the literature before this study, many were retrospective analyses, and persistence was estimated to be in the 40%-60% range.”
International prospective studies have provided a more conservative estimate of persistence. For example, the German Multicenter Allergy Study followed 1,314 from birth through age 7 (J Allergy Clin Immunol. 2004;113[5]:925-31). Of these, 22% had AD within the first 2 years of life. Of these, 43% were in remission by age 3, while 38% had intermittent AD, and 19% had symptoms every year of the study. “Studies of other birth cohorts in the world came out suggesting that the rates of AD persistence ranges in the single digits to the teens,” Dr. Chovatiya said.
To reconcile these heterogeneous estimates of AD persistence, researchers conducted a systematic review and meta-analysis of 45 studies that included 110,651 subjects from 15 countries and spanned 434,992 patient-years (J Am Acad Dermatol. 2016;75:681-7.e11). They found that 80% of childhood AD had at least one observed period of disease clearance by 8 years of age. “Most importantly, less than 5% of childhood AD was persistent 20 years after diagnosis,” Dr. Chovatiya said. “However, interestingly, increased persistence was associated with later onset AD, more years of persistence, and more patient/caregiver-assessed disease.” He pointed out inherent limitations to all studies of AD persistence, including nonuniform methods of data collection, differing cohorts, different ways of diagnosing AD, different disease severity scales, and the fact that most don’t assess flares or recurrence beyond the initial period of disease clearance. “This can lead to a potential underestimation of longer-term persistence,” he said.
Childhood AD features unique predictors of persistence that may define AD trajectories. For example, in several existing studies, more persistent disease was associated with higher baseline severity, earlier-onset AD, personal history of atopy, family history of AD, AD genetic risk score (heritability, including common Filaggrin mutations), urban environment, non-White race, Hispanic ethnicity, female sex, lower household income, and overall poorer health status.
Dr. Chovatiya said. “I think that AD classification can take a lesson from asthma. When we think about how our allergy colleagues think about asthma, it is commonly classified as intermittent, mild persistent, moderate persistent, and severe persistent. Those that have intermittent disease get reactive treatment, while those with persistent disease get proactive treatment. Similarly, AD could be classified as mild intermittent, mild persistent, moderate to severe intermittent and moderate to severe persistent.”
He concluded his presentation by recommending that the fluctuating course of AD be better captured in clinical trials. “Current randomized, controlled trials use validated measures of AD signs and symptoms as inclusion criteria and measures of efficacy,” he said. “Static assessments may confound treatment effects, and assessment of prespecified time points are somewhat arbitrary in the context of disease subsets.” He proposes studies that examine aggregate measures of long-term disease control, such as number of itch-free days, weeks with clear skin, and flares experienced. “Long-term control assessment in RCTs should include signs, symptoms, health-related quality of life, and a patient global domain over time to better understand how AD is doing in the long run,” he said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arcutis, Arena, Incyte, Pfizer, Regeneron, and Sanofi Genzyme.
FROM REVOLUTIONIZING AD 2021
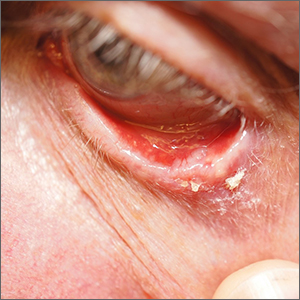
Growth on eyelid
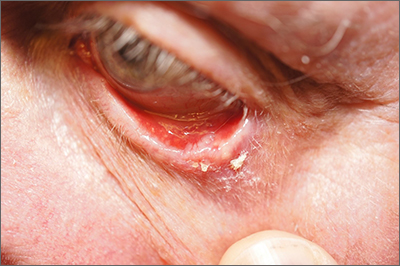
A shave biopsy (performed carefully to avoid caustic hemostatic agents irritating the conjunctiva) confirmed the diagnosis of a micronodular basal cell carcinoma (BCC).
BCC is a common tumor occurring on the eyelids and in the periocular region. Any new growing papule on the eyelids, history of focal bleeding, irritation, or focal loss of eyelashes should cause suspicion for BCC. Patients are often unaware of any symptoms when lesions begin, highlighting the importance of close inspection of the eyelids when skin or eye exams are performed. The differential diagnosis includes benign lesions such as hidrocystomas and nevi, as well as malignancies, including sebaceous carcinoma and squamous cell carcinoma.1
Factors that come into play when exploring eyelid BCC treatment options include tumor removal, eyelid function, and appearance. The potential morbidity associated with tumor spread in the periorbital region highlights the importance of early detection of eyelid cancers. Mohs micrographic surgery (MMS) is a first choice for tumor removal of an eyelid BCC and offers a high cure rate with minimal tissue removal.
Removal of an eyelid BCC may be a multidisciplinary endeavor with MMS achieving a clear margin, and Ophthalmology or Oculoplastics following with repair and closure soon after. Patients who can’t tolerate surgery should consider vismodegib, a targeted chemotherapy, or radiotherapy.
The patient in this case opted for a single staged excision and repair with Oculoplastics and has had no recurrence. He subsequently underwent a revision procedure to improve ectropion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment. Onco Targets Ther. 2017;10:2483-2489. doi: 10.2147/OTT.S130371

A shave biopsy (performed carefully to avoid caustic hemostatic agents irritating the conjunctiva) confirmed the diagnosis of a micronodular basal cell carcinoma (BCC).
BCC is a common tumor occurring on the eyelids and in the periocular region. Any new growing papule on the eyelids, history of focal bleeding, irritation, or focal loss of eyelashes should cause suspicion for BCC. Patients are often unaware of any symptoms when lesions begin, highlighting the importance of close inspection of the eyelids when skin or eye exams are performed. The differential diagnosis includes benign lesions such as hidrocystomas and nevi, as well as malignancies, including sebaceous carcinoma and squamous cell carcinoma.1
Factors that come into play when exploring eyelid BCC treatment options include tumor removal, eyelid function, and appearance. The potential morbidity associated with tumor spread in the periorbital region highlights the importance of early detection of eyelid cancers. Mohs micrographic surgery (MMS) is a first choice for tumor removal of an eyelid BCC and offers a high cure rate with minimal tissue removal.
Removal of an eyelid BCC may be a multidisciplinary endeavor with MMS achieving a clear margin, and Ophthalmology or Oculoplastics following with repair and closure soon after. Patients who can’t tolerate surgery should consider vismodegib, a targeted chemotherapy, or radiotherapy.
The patient in this case opted for a single staged excision and repair with Oculoplastics and has had no recurrence. He subsequently underwent a revision procedure to improve ectropion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

A shave biopsy (performed carefully to avoid caustic hemostatic agents irritating the conjunctiva) confirmed the diagnosis of a micronodular basal cell carcinoma (BCC).
BCC is a common tumor occurring on the eyelids and in the periocular region. Any new growing papule on the eyelids, history of focal bleeding, irritation, or focal loss of eyelashes should cause suspicion for BCC. Patients are often unaware of any symptoms when lesions begin, highlighting the importance of close inspection of the eyelids when skin or eye exams are performed. The differential diagnosis includes benign lesions such as hidrocystomas and nevi, as well as malignancies, including sebaceous carcinoma and squamous cell carcinoma.1
Factors that come into play when exploring eyelid BCC treatment options include tumor removal, eyelid function, and appearance. The potential morbidity associated with tumor spread in the periorbital region highlights the importance of early detection of eyelid cancers. Mohs micrographic surgery (MMS) is a first choice for tumor removal of an eyelid BCC and offers a high cure rate with minimal tissue removal.
Removal of an eyelid BCC may be a multidisciplinary endeavor with MMS achieving a clear margin, and Ophthalmology or Oculoplastics following with repair and closure soon after. Patients who can’t tolerate surgery should consider vismodegib, a targeted chemotherapy, or radiotherapy.
The patient in this case opted for a single staged excision and repair with Oculoplastics and has had no recurrence. He subsequently underwent a revision procedure to improve ectropion.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment. Onco Targets Ther. 2017;10:2483-2489. doi: 10.2147/OTT.S130371
1. Shi Y, Jia R, Fan X. Ocular basal cell carcinoma: a brief literature review of clinical diagnosis and treatment. Onco Targets Ther. 2017;10:2483-2489. doi: 10.2147/OTT.S130371
Itch-dominant atopic dermatitis often flies under the radar
In the clinical experience of Jonathan I. Silverberg, MD, PhD, MPH,
That’s because a disconnect often exists between clinician-reported and patient-reported outcome measures, Dr. Silverberg, director of clinical research in the division of dermatology at George Washington University School of Medicine and Health Sciences, said during the Revolutionizing Atopic Dermatitis virtual symposium. For example, multiple studies showed only weak to moderate correlations between the patient-focused Worst Itch Numeric Rating Scale (NRS) and Average Pruritus NRS compared with clinician-reported outcomes such as the Eczema Area and Severity Index (EASI), the objective SCORAD, body surface area (BSA), and the Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD), with only moderate correlation coefficients ranging from 0.3 to 0.6.
“These findings suggest that clinician-reported outcome measures are poor indicators of the patient experience,” he said. “We need to do a better job capturing patient-reported outcomes to understand how patients are impacted. But there’s something more novel to this because the weak correlations may also suggest that itch and other symptoms follow a different course than the signs of the disease. Just because the lesions flare up doesn’t mean the itch does, and vice versa. Anecdotally, this came up at many patient encounters where the skin looked good, but the patient was miserable with itch.”
To understand how the combination of itch and lesion severity predicts the severity assessment, longitudinal course, burden, and treatment of AD, Dr. Silverberg and colleagues prospectively evaluated 592 adults with AD . They defined four different AD subsets using the verbal rating scale for NRS average itch combined with either the EASI, objective-SCORAD, or vIGA-AD as follows: mild-moderate itch and lesions (MI/ML), mild-moderate itch and severe lesions (MI/SL), severe itch and mild-moderate lesions (SI/ML; the itch dominant subset), and severe itch and lesions (SI/SL). They found that most patients had MI/ML (59.4%-62.3%), followed by SI/ML (21.3%-29.1%), SI/SL (6%-12.9%), and MI/SL (3.8%-6.4%). SI/ML was more common in female and Black patients.
In addition, patients with MI/SL or SI/ML described their AD as being more severe on patient global assessment and had poor quality of life (QOL) scores, while patients with SI/SL were most likely to describe their disease as severe and have poor QOL scores. Patients with SI/ML described their disease as being more severe overall, yet patients with MI/SL or SI/SL were far more likely to be assigned severe PGA scores by clinicians. “The patients who have severe itch and mild lesions consider their disease severe, but the clinician is missing it,” Dr. Silverberg said. “Occasionally they’re picking it up but they’re missing a lot of these severe itch cases when there are milder lesions.”
In other findings, patients who had baseline MI/SL, SI/ML, and SI/SL were associated with similar frequency of AD flares, periods of AD clearance/remission, more itch triggers, and longitudinal courses over time, “which is remarkable,” he said. “It means those that have severe itch, even when they have milder lesions, are going to have unstable, more persistent disease, and have a harder time keeping control of it, and are ultimately going to require systemic therapies.” In fact, most patients with SI/SL (57.8%-66.7%) and MI/SL (53.9%-57.7%) but fewer patients with MI/ML (36.7%-38.4%) and SI/ML (30.8%-32%) initiated systemic, biologic, or phototherapy for their AD during follow-up. “There is a real upshot here clinically, in that patients are just not getting stepped up appropriately to achieve better control of their disease when they have itch-dominant AD,” Dr. Silverberg said.
He described itch-dominant AD as a novel disease phenotype that requires further investigation. “Why is it that some patients are getting such severe itch and milder looking lesions?” he asked. “I don’t think it’s just a matter of poor outcome measures that we have. So, what is it? It’s not entirely clear. Clinically, itch-dominant AD is important as it relates to the issues of diversity and skin of color because in darker skin tones, we cannot easily appreciate erythema. We may totally miss the active lesions. I think that’s a big part of why we see this itch-dominant AD more commonly in Black patients. Therefore, it is so important to ask our patients about their symptoms and to assess the severity of itch. But, even if they have what we think are milder lesions and severe itch, we must recognize they may not be well controlled. They may not be happy. They may have poor quality of life, and they may need to be stepped up appropriately. We need a lot more information to guide the assessment and management of this important subset of patients.”
Dr. Silverberg disclosed that he is a consultant to numerous pharmaceutical companies, receives fees for non-CME/CE services from Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi Genzyme, as well as contracted research fees from Galderma.
In the clinical experience of Jonathan I. Silverberg, MD, PhD, MPH,
That’s because a disconnect often exists between clinician-reported and patient-reported outcome measures, Dr. Silverberg, director of clinical research in the division of dermatology at George Washington University School of Medicine and Health Sciences, said during the Revolutionizing Atopic Dermatitis virtual symposium. For example, multiple studies showed only weak to moderate correlations between the patient-focused Worst Itch Numeric Rating Scale (NRS) and Average Pruritus NRS compared with clinician-reported outcomes such as the Eczema Area and Severity Index (EASI), the objective SCORAD, body surface area (BSA), and the Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD), with only moderate correlation coefficients ranging from 0.3 to 0.6.
“These findings suggest that clinician-reported outcome measures are poor indicators of the patient experience,” he said. “We need to do a better job capturing patient-reported outcomes to understand how patients are impacted. But there’s something more novel to this because the weak correlations may also suggest that itch and other symptoms follow a different course than the signs of the disease. Just because the lesions flare up doesn’t mean the itch does, and vice versa. Anecdotally, this came up at many patient encounters where the skin looked good, but the patient was miserable with itch.”
To understand how the combination of itch and lesion severity predicts the severity assessment, longitudinal course, burden, and treatment of AD, Dr. Silverberg and colleagues prospectively evaluated 592 adults with AD . They defined four different AD subsets using the verbal rating scale for NRS average itch combined with either the EASI, objective-SCORAD, or vIGA-AD as follows: mild-moderate itch and lesions (MI/ML), mild-moderate itch and severe lesions (MI/SL), severe itch and mild-moderate lesions (SI/ML; the itch dominant subset), and severe itch and lesions (SI/SL). They found that most patients had MI/ML (59.4%-62.3%), followed by SI/ML (21.3%-29.1%), SI/SL (6%-12.9%), and MI/SL (3.8%-6.4%). SI/ML was more common in female and Black patients.
In addition, patients with MI/SL or SI/ML described their AD as being more severe on patient global assessment and had poor quality of life (QOL) scores, while patients with SI/SL were most likely to describe their disease as severe and have poor QOL scores. Patients with SI/ML described their disease as being more severe overall, yet patients with MI/SL or SI/SL were far more likely to be assigned severe PGA scores by clinicians. “The patients who have severe itch and mild lesions consider their disease severe, but the clinician is missing it,” Dr. Silverberg said. “Occasionally they’re picking it up but they’re missing a lot of these severe itch cases when there are milder lesions.”
In other findings, patients who had baseline MI/SL, SI/ML, and SI/SL were associated with similar frequency of AD flares, periods of AD clearance/remission, more itch triggers, and longitudinal courses over time, “which is remarkable,” he said. “It means those that have severe itch, even when they have milder lesions, are going to have unstable, more persistent disease, and have a harder time keeping control of it, and are ultimately going to require systemic therapies.” In fact, most patients with SI/SL (57.8%-66.7%) and MI/SL (53.9%-57.7%) but fewer patients with MI/ML (36.7%-38.4%) and SI/ML (30.8%-32%) initiated systemic, biologic, or phototherapy for their AD during follow-up. “There is a real upshot here clinically, in that patients are just not getting stepped up appropriately to achieve better control of their disease when they have itch-dominant AD,” Dr. Silverberg said.
He described itch-dominant AD as a novel disease phenotype that requires further investigation. “Why is it that some patients are getting such severe itch and milder looking lesions?” he asked. “I don’t think it’s just a matter of poor outcome measures that we have. So, what is it? It’s not entirely clear. Clinically, itch-dominant AD is important as it relates to the issues of diversity and skin of color because in darker skin tones, we cannot easily appreciate erythema. We may totally miss the active lesions. I think that’s a big part of why we see this itch-dominant AD more commonly in Black patients. Therefore, it is so important to ask our patients about their symptoms and to assess the severity of itch. But, even if they have what we think are milder lesions and severe itch, we must recognize they may not be well controlled. They may not be happy. They may have poor quality of life, and they may need to be stepped up appropriately. We need a lot more information to guide the assessment and management of this important subset of patients.”
Dr. Silverberg disclosed that he is a consultant to numerous pharmaceutical companies, receives fees for non-CME/CE services from Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi Genzyme, as well as contracted research fees from Galderma.
In the clinical experience of Jonathan I. Silverberg, MD, PhD, MPH,
That’s because a disconnect often exists between clinician-reported and patient-reported outcome measures, Dr. Silverberg, director of clinical research in the division of dermatology at George Washington University School of Medicine and Health Sciences, said during the Revolutionizing Atopic Dermatitis virtual symposium. For example, multiple studies showed only weak to moderate correlations between the patient-focused Worst Itch Numeric Rating Scale (NRS) and Average Pruritus NRS compared with clinician-reported outcomes such as the Eczema Area and Severity Index (EASI), the objective SCORAD, body surface area (BSA), and the Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD), with only moderate correlation coefficients ranging from 0.3 to 0.6.
“These findings suggest that clinician-reported outcome measures are poor indicators of the patient experience,” he said. “We need to do a better job capturing patient-reported outcomes to understand how patients are impacted. But there’s something more novel to this because the weak correlations may also suggest that itch and other symptoms follow a different course than the signs of the disease. Just because the lesions flare up doesn’t mean the itch does, and vice versa. Anecdotally, this came up at many patient encounters where the skin looked good, but the patient was miserable with itch.”
To understand how the combination of itch and lesion severity predicts the severity assessment, longitudinal course, burden, and treatment of AD, Dr. Silverberg and colleagues prospectively evaluated 592 adults with AD . They defined four different AD subsets using the verbal rating scale for NRS average itch combined with either the EASI, objective-SCORAD, or vIGA-AD as follows: mild-moderate itch and lesions (MI/ML), mild-moderate itch and severe lesions (MI/SL), severe itch and mild-moderate lesions (SI/ML; the itch dominant subset), and severe itch and lesions (SI/SL). They found that most patients had MI/ML (59.4%-62.3%), followed by SI/ML (21.3%-29.1%), SI/SL (6%-12.9%), and MI/SL (3.8%-6.4%). SI/ML was more common in female and Black patients.
In addition, patients with MI/SL or SI/ML described their AD as being more severe on patient global assessment and had poor quality of life (QOL) scores, while patients with SI/SL were most likely to describe their disease as severe and have poor QOL scores. Patients with SI/ML described their disease as being more severe overall, yet patients with MI/SL or SI/SL were far more likely to be assigned severe PGA scores by clinicians. “The patients who have severe itch and mild lesions consider their disease severe, but the clinician is missing it,” Dr. Silverberg said. “Occasionally they’re picking it up but they’re missing a lot of these severe itch cases when there are milder lesions.”
In other findings, patients who had baseline MI/SL, SI/ML, and SI/SL were associated with similar frequency of AD flares, periods of AD clearance/remission, more itch triggers, and longitudinal courses over time, “which is remarkable,” he said. “It means those that have severe itch, even when they have milder lesions, are going to have unstable, more persistent disease, and have a harder time keeping control of it, and are ultimately going to require systemic therapies.” In fact, most patients with SI/SL (57.8%-66.7%) and MI/SL (53.9%-57.7%) but fewer patients with MI/ML (36.7%-38.4%) and SI/ML (30.8%-32%) initiated systemic, biologic, or phototherapy for their AD during follow-up. “There is a real upshot here clinically, in that patients are just not getting stepped up appropriately to achieve better control of their disease when they have itch-dominant AD,” Dr. Silverberg said.
He described itch-dominant AD as a novel disease phenotype that requires further investigation. “Why is it that some patients are getting such severe itch and milder looking lesions?” he asked. “I don’t think it’s just a matter of poor outcome measures that we have. So, what is it? It’s not entirely clear. Clinically, itch-dominant AD is important as it relates to the issues of diversity and skin of color because in darker skin tones, we cannot easily appreciate erythema. We may totally miss the active lesions. I think that’s a big part of why we see this itch-dominant AD more commonly in Black patients. Therefore, it is so important to ask our patients about their symptoms and to assess the severity of itch. But, even if they have what we think are milder lesions and severe itch, we must recognize they may not be well controlled. They may not be happy. They may have poor quality of life, and they may need to be stepped up appropriately. We need a lot more information to guide the assessment and management of this important subset of patients.”
Dr. Silverberg disclosed that he is a consultant to numerous pharmaceutical companies, receives fees for non-CME/CE services from Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi Genzyme, as well as contracted research fees from Galderma.
FROM REVOLUTIONIZING AD 2021
Case series show no consensus on treatment for palmoplantar pustulosis, generalized pustular psoriasis
that evaluated the characteristics and course of the disease in patients diagnosed with PPP or GPP.
“These case series confirm the rarity of both generalized pustular psoriasis and palmoplantar pustulosis (PPP) and highlight the persistence of symptoms over time and the lack of effective treatment options available to patients,” Megan H. Noe, MD, MPH, MSCE, first author of both case series and assistant professor of dermatology, Harvard Medical School, and a dermatologist at Brigham and Women’s Hospital, both in Boston, said in an interview. In both studies, she added, “more than 20 different therapies were utilized, demonstrating a lack of consensus regarding effective treatment.”
The two case series were published in JAMA Dermatology.
Palmoplantar pustulosis
In the case series of 197 patients with PPP , data were obtained from a retrospective review at 20 academic dermatology practices in the United States between January 2007 and December 2018. The patients were mostly women (73.6%) who were White (60.9%), with a mean age of 53 years; 38.1% were current smokers, and 27.4% were former smokers, and the mean follow-up time was 22.1 months. About half (48.2%) of patients who presented to their respective centers had skin pain, 19.8% had problems using their hands and feet, 12.7% had arthralgias, and 2% had myalgias. Clinicians who examined these patients found pustules on the palms (80.2%), soles (76.7%), and both palms and soles (59.9%); some nail unit involvement was reported in 10.2%.
Patients were treated with a variety of topical therapies, systemic steroids, systemic anti-infectives, and systemic psoriasis therapies, Dr. Noe and colleagues said. The most common initial treatments included a topical steroid (84.8%), with the vast majority of clinicians using a high-potency topical steroid (153 of 167 patients; 91.6%), or topical therapy only (64.5%).
Other initial treatments used were other types of topical medications in 34 of the patients in the series (17.3%), such as a vitamin D analogue in 27 patients (79.4%); oral systemic treatments such as acitretin in 27 patients (13.7%) or methotrexate in 22 patients (11.2%); narrowband UVB phototherapy in 15 patients (7.7%); systemic steroids in 10 patients (5.1%); or systemic antibiotics in 9 patients (4.6%). Less commonly used were biologic agents like adalimumab, used in 6 patients (3.1%).
The researchers also examined health care utilization in 128 patients and found that 82% had at least one follow-up visit, 31.3% required two to three follow-up visits, and 18.8% had five or more follow-up visits. When adjusted to account for age and sex, there was a decreased risk of requiring five or more healthcare visits per year for women (odds ratio, 0.49; 95% confidence interval, 0.25-0.95)
Generalized pustular psoriasis
Dr. Noe and colleagues also evaluated 95 patients with GPP in a retrospective longitudinal case series of patients treated at 20 academic dermatology practices in the United States between January 2007 and December 2018. As in the PPP group, most patients in the GPP case series were women (70.5%), and over half were White (53.7%); the mean age was 50.3 years old, and the mean follow-up time was 19.8 months. A majority of patients with GPP were never-smokers (52.6%) or former smokers (20%). When patients with GPP initially presented to the study sites, 36.8% were admitted as inpatients, 9.5% presented in the emergency department, and 53.7% presented in an outpatient or ambulatory dermatology setting.
GPP commonly appeared on the trunk and extremities, but was “also reported on the scalp, face, genitals, nail unit, and mucous membranes in a minority of patients,” the researchers said. Overall, 62.1% of patients had skin pain, 26.2% had joint pain, 16.8% reported tachycardia, and 9.5% reported fever. Hypertension, depression, diabetes, chronic kidney disease, and hypothyroidism were common comorbidities of GPP, the researchers noted.
Clinicians reported treating GPP with topical steroids (86.3%) and topical treatments alone (32.3%). Oral systemic treatments such as acitretin (24.2%), cyclosporine (22.1%), and methotrexate (13.7%) were also used, as well as systemic steroids (20%). Other treatments used were narrowband UVB phototherapy (5.3%) and biologic agents like adalimumab (4.2%) and infliximab (4.2%).
For 53 patients with follow-up data of at least 6 months, 19 (35.8%) had been hospitalized because of their symptoms, and 8 patients were hospitalized for further GPP-specific concerns. Patients with GPP had a median 3.2 dermatology visits per year and a maximum of 18 visits. A model that was adjusted for age and sex showed women were at a decreased risk for being admitted to the hospital or emergency department in the follow-up period (odds ratio, 0.19; 95% confidence interval, 0.04-0.83).
PPP and GPP in practice
Sylvia Hsu, MD, professor and chair of the department of dermatology at Temple University, Philadelphia, who was not involved with the research, noted that most dermatologists will see few, if any, cases of PPP and GPP in a year. At her center, she estimated that she sees about one PPP case per week, and one or two cases of GPP a year. In general, she said that her clinical experience matched what was found by the authors of both case series.
For patients with PPP, “I would say the average dermatologist would probably start out with a superpotent topical steroid like clobetasol or halobetasol ointment,” Dr. Hsu said.
If they are not of childbearing age, she added, she would also prescribe acitretin, which she avoids giving to patients of childbearing age because of its teratogenicity. “Acitretin has the reputation that it doesn’t work well or fast for psoriasis. It doesn’t work well or fast for plaque-type psoriasis, but it works well and fast for pustular psoriasis,” she said.
In place of acitretin, Dr. Hsu recommended cyclosporine for a patient of childbearing age as a short-term solution to resolve symptoms before transitioning them to another therapy. “A woman of childbearing age, you put on cyclosporine, you’ve got to transition to something else,” she said. “And so many times you wean them off, the pustular psoriasis comes back because the topical steroid doesn’t work that well.”
One possible option is the interluekin-23 inhibitor guselkumab (approved by the Food and Drug Administration for treating moderate to severe plaque psoriasis and psoriatic arthritis) but cost and effectiveness can be a factor. Although studies have shown efficacy, biologics as treatments for PPP are “hit or miss,” Dr. Hsu said.
Regarding use of systemic therapies, Dr. Hsu cautioned against using them to treat plaque-type psoriasis. “We always learn, don’t use a systemic steroid like prednisone to treat psoriasis because it helps, but it comes back with a vengeance,” she said. “Sometimes when you treat plaque-type psoriasis with prednisone, it could come back with a vengeance, and it can come back as generalized pustular psoriasis.”
For patients with GPP, “you need a quick fix” because of the painful symptoms associated with the disease, Dr. Hsu said. In this case, she recommended cyclosporine and said she would avoid prescribing topical medications. “You’re going to have to give an oral drug because usually when we’re seeing somebody with GPP, they’re either a hospital consult or they just walked in the door,” she said. After prescribing cyclosporine, you would transition to another treatment like a biologic “as quickly as you can” with the knowledge that the biologic “may or may not work.”
New treatment options needed
Commenting on both case series in a related editorial, Edward W. Cowen, MD, MHSc, senior clinician and head of the dermatology consultation service in the dermatology branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md., said that “much of the clinical presentation of pustular disease remains a mystery,” including why tobacco use is a risk factor for developing pustular psoriasis, and why tumor necrosis factor inhibitors “induce pustular disease in a small number of patients” with psoriasis vulgaris.
“Most importantly, we still do not know if localized and generalized pustular psoriasis all truly represent different variants of the same disease process, and if not, which biologic treatment represents the best option for a given clinical variant,” he wrote.
Dr. Cowen noted that the multi-institutional approach to collecting the retrospective data in these case series could be used as a “basic framework to build on for future clinical trials for rare skin diseases such as pustular psoriasis.”
In the interview, Dr. Noe said that she hoped that the “Pustular Psoriasis in the US Research Group” she and her coauthors created for the case series could help with the development of prospective clinical trials. “For pustular psoriasis and other rare diseases in dermatology, multi-institutional collaborations are necessary to conduct prospective research,” she said.
“While not directly studied in our research, I think it is important to consider the negative impact on quality of life, experienced by patients with pustular psoriasis. In our study, many patients experienced exacerbations of their disease over time, and it is important to consider the impact this has on patients,” she said in the interview. “Continued research on pustular psoriasis is necessary to decrease the negative impact of these diseases on the lives of our patients.”
The case series were funded in part by an institutional grant from Boehringer Ingelheim. The authors report relationships with various pharmaceutical and biopharmaceutical companies, technology companies, medical publishing companies, medical journals, and medical societies with connections to the topic area in the form of serving in roles as a chief medical editor, consultant, data safety monitoring board member, deputy editor, principal investigator, research investigator, scientific adviser, or speaker; or having received grants, honoraria, personal fees, or research funding. Dr. Cowen has no disclosures. Dr. Hsu reports serving on a Boehringer Ingelheim advisory board for a product being evaluated as a potential treatment for GPP.
that evaluated the characteristics and course of the disease in patients diagnosed with PPP or GPP.
“These case series confirm the rarity of both generalized pustular psoriasis and palmoplantar pustulosis (PPP) and highlight the persistence of symptoms over time and the lack of effective treatment options available to patients,” Megan H. Noe, MD, MPH, MSCE, first author of both case series and assistant professor of dermatology, Harvard Medical School, and a dermatologist at Brigham and Women’s Hospital, both in Boston, said in an interview. In both studies, she added, “more than 20 different therapies were utilized, demonstrating a lack of consensus regarding effective treatment.”
The two case series were published in JAMA Dermatology.
Palmoplantar pustulosis
In the case series of 197 patients with PPP , data were obtained from a retrospective review at 20 academic dermatology practices in the United States between January 2007 and December 2018. The patients were mostly women (73.6%) who were White (60.9%), with a mean age of 53 years; 38.1% were current smokers, and 27.4% were former smokers, and the mean follow-up time was 22.1 months. About half (48.2%) of patients who presented to their respective centers had skin pain, 19.8% had problems using their hands and feet, 12.7% had arthralgias, and 2% had myalgias. Clinicians who examined these patients found pustules on the palms (80.2%), soles (76.7%), and both palms and soles (59.9%); some nail unit involvement was reported in 10.2%.
Patients were treated with a variety of topical therapies, systemic steroids, systemic anti-infectives, and systemic psoriasis therapies, Dr. Noe and colleagues said. The most common initial treatments included a topical steroid (84.8%), with the vast majority of clinicians using a high-potency topical steroid (153 of 167 patients; 91.6%), or topical therapy only (64.5%).
Other initial treatments used were other types of topical medications in 34 of the patients in the series (17.3%), such as a vitamin D analogue in 27 patients (79.4%); oral systemic treatments such as acitretin in 27 patients (13.7%) or methotrexate in 22 patients (11.2%); narrowband UVB phototherapy in 15 patients (7.7%); systemic steroids in 10 patients (5.1%); or systemic antibiotics in 9 patients (4.6%). Less commonly used were biologic agents like adalimumab, used in 6 patients (3.1%).
The researchers also examined health care utilization in 128 patients and found that 82% had at least one follow-up visit, 31.3% required two to three follow-up visits, and 18.8% had five or more follow-up visits. When adjusted to account for age and sex, there was a decreased risk of requiring five or more healthcare visits per year for women (odds ratio, 0.49; 95% confidence interval, 0.25-0.95)
Generalized pustular psoriasis
Dr. Noe and colleagues also evaluated 95 patients with GPP in a retrospective longitudinal case series of patients treated at 20 academic dermatology practices in the United States between January 2007 and December 2018. As in the PPP group, most patients in the GPP case series were women (70.5%), and over half were White (53.7%); the mean age was 50.3 years old, and the mean follow-up time was 19.8 months. A majority of patients with GPP were never-smokers (52.6%) or former smokers (20%). When patients with GPP initially presented to the study sites, 36.8% were admitted as inpatients, 9.5% presented in the emergency department, and 53.7% presented in an outpatient or ambulatory dermatology setting.
GPP commonly appeared on the trunk and extremities, but was “also reported on the scalp, face, genitals, nail unit, and mucous membranes in a minority of patients,” the researchers said. Overall, 62.1% of patients had skin pain, 26.2% had joint pain, 16.8% reported tachycardia, and 9.5% reported fever. Hypertension, depression, diabetes, chronic kidney disease, and hypothyroidism were common comorbidities of GPP, the researchers noted.
Clinicians reported treating GPP with topical steroids (86.3%) and topical treatments alone (32.3%). Oral systemic treatments such as acitretin (24.2%), cyclosporine (22.1%), and methotrexate (13.7%) were also used, as well as systemic steroids (20%). Other treatments used were narrowband UVB phototherapy (5.3%) and biologic agents like adalimumab (4.2%) and infliximab (4.2%).
For 53 patients with follow-up data of at least 6 months, 19 (35.8%) had been hospitalized because of their symptoms, and 8 patients were hospitalized for further GPP-specific concerns. Patients with GPP had a median 3.2 dermatology visits per year and a maximum of 18 visits. A model that was adjusted for age and sex showed women were at a decreased risk for being admitted to the hospital or emergency department in the follow-up period (odds ratio, 0.19; 95% confidence interval, 0.04-0.83).
PPP and GPP in practice
Sylvia Hsu, MD, professor and chair of the department of dermatology at Temple University, Philadelphia, who was not involved with the research, noted that most dermatologists will see few, if any, cases of PPP and GPP in a year. At her center, she estimated that she sees about one PPP case per week, and one or two cases of GPP a year. In general, she said that her clinical experience matched what was found by the authors of both case series.
For patients with PPP, “I would say the average dermatologist would probably start out with a superpotent topical steroid like clobetasol or halobetasol ointment,” Dr. Hsu said.
If they are not of childbearing age, she added, she would also prescribe acitretin, which she avoids giving to patients of childbearing age because of its teratogenicity. “Acitretin has the reputation that it doesn’t work well or fast for psoriasis. It doesn’t work well or fast for plaque-type psoriasis, but it works well and fast for pustular psoriasis,” she said.
In place of acitretin, Dr. Hsu recommended cyclosporine for a patient of childbearing age as a short-term solution to resolve symptoms before transitioning them to another therapy. “A woman of childbearing age, you put on cyclosporine, you’ve got to transition to something else,” she said. “And so many times you wean them off, the pustular psoriasis comes back because the topical steroid doesn’t work that well.”
One possible option is the interluekin-23 inhibitor guselkumab (approved by the Food and Drug Administration for treating moderate to severe plaque psoriasis and psoriatic arthritis) but cost and effectiveness can be a factor. Although studies have shown efficacy, biologics as treatments for PPP are “hit or miss,” Dr. Hsu said.
Regarding use of systemic therapies, Dr. Hsu cautioned against using them to treat plaque-type psoriasis. “We always learn, don’t use a systemic steroid like prednisone to treat psoriasis because it helps, but it comes back with a vengeance,” she said. “Sometimes when you treat plaque-type psoriasis with prednisone, it could come back with a vengeance, and it can come back as generalized pustular psoriasis.”
For patients with GPP, “you need a quick fix” because of the painful symptoms associated with the disease, Dr. Hsu said. In this case, she recommended cyclosporine and said she would avoid prescribing topical medications. “You’re going to have to give an oral drug because usually when we’re seeing somebody with GPP, they’re either a hospital consult or they just walked in the door,” she said. After prescribing cyclosporine, you would transition to another treatment like a biologic “as quickly as you can” with the knowledge that the biologic “may or may not work.”
New treatment options needed
Commenting on both case series in a related editorial, Edward W. Cowen, MD, MHSc, senior clinician and head of the dermatology consultation service in the dermatology branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md., said that “much of the clinical presentation of pustular disease remains a mystery,” including why tobacco use is a risk factor for developing pustular psoriasis, and why tumor necrosis factor inhibitors “induce pustular disease in a small number of patients” with psoriasis vulgaris.
“Most importantly, we still do not know if localized and generalized pustular psoriasis all truly represent different variants of the same disease process, and if not, which biologic treatment represents the best option for a given clinical variant,” he wrote.
Dr. Cowen noted that the multi-institutional approach to collecting the retrospective data in these case series could be used as a “basic framework to build on for future clinical trials for rare skin diseases such as pustular psoriasis.”
In the interview, Dr. Noe said that she hoped that the “Pustular Psoriasis in the US Research Group” she and her coauthors created for the case series could help with the development of prospective clinical trials. “For pustular psoriasis and other rare diseases in dermatology, multi-institutional collaborations are necessary to conduct prospective research,” she said.
“While not directly studied in our research, I think it is important to consider the negative impact on quality of life, experienced by patients with pustular psoriasis. In our study, many patients experienced exacerbations of their disease over time, and it is important to consider the impact this has on patients,” she said in the interview. “Continued research on pustular psoriasis is necessary to decrease the negative impact of these diseases on the lives of our patients.”
The case series were funded in part by an institutional grant from Boehringer Ingelheim. The authors report relationships with various pharmaceutical and biopharmaceutical companies, technology companies, medical publishing companies, medical journals, and medical societies with connections to the topic area in the form of serving in roles as a chief medical editor, consultant, data safety monitoring board member, deputy editor, principal investigator, research investigator, scientific adviser, or speaker; or having received grants, honoraria, personal fees, or research funding. Dr. Cowen has no disclosures. Dr. Hsu reports serving on a Boehringer Ingelheim advisory board for a product being evaluated as a potential treatment for GPP.
that evaluated the characteristics and course of the disease in patients diagnosed with PPP or GPP.
“These case series confirm the rarity of both generalized pustular psoriasis and palmoplantar pustulosis (PPP) and highlight the persistence of symptoms over time and the lack of effective treatment options available to patients,” Megan H. Noe, MD, MPH, MSCE, first author of both case series and assistant professor of dermatology, Harvard Medical School, and a dermatologist at Brigham and Women’s Hospital, both in Boston, said in an interview. In both studies, she added, “more than 20 different therapies were utilized, demonstrating a lack of consensus regarding effective treatment.”
The two case series were published in JAMA Dermatology.
Palmoplantar pustulosis
In the case series of 197 patients with PPP , data were obtained from a retrospective review at 20 academic dermatology practices in the United States between January 2007 and December 2018. The patients were mostly women (73.6%) who were White (60.9%), with a mean age of 53 years; 38.1% were current smokers, and 27.4% were former smokers, and the mean follow-up time was 22.1 months. About half (48.2%) of patients who presented to their respective centers had skin pain, 19.8% had problems using their hands and feet, 12.7% had arthralgias, and 2% had myalgias. Clinicians who examined these patients found pustules on the palms (80.2%), soles (76.7%), and both palms and soles (59.9%); some nail unit involvement was reported in 10.2%.
Patients were treated with a variety of topical therapies, systemic steroids, systemic anti-infectives, and systemic psoriasis therapies, Dr. Noe and colleagues said. The most common initial treatments included a topical steroid (84.8%), with the vast majority of clinicians using a high-potency topical steroid (153 of 167 patients; 91.6%), or topical therapy only (64.5%).
Other initial treatments used were other types of topical medications in 34 of the patients in the series (17.3%), such as a vitamin D analogue in 27 patients (79.4%); oral systemic treatments such as acitretin in 27 patients (13.7%) or methotrexate in 22 patients (11.2%); narrowband UVB phototherapy in 15 patients (7.7%); systemic steroids in 10 patients (5.1%); or systemic antibiotics in 9 patients (4.6%). Less commonly used were biologic agents like adalimumab, used in 6 patients (3.1%).
The researchers also examined health care utilization in 128 patients and found that 82% had at least one follow-up visit, 31.3% required two to three follow-up visits, and 18.8% had five or more follow-up visits. When adjusted to account for age and sex, there was a decreased risk of requiring five or more healthcare visits per year for women (odds ratio, 0.49; 95% confidence interval, 0.25-0.95)
Generalized pustular psoriasis
Dr. Noe and colleagues also evaluated 95 patients with GPP in a retrospective longitudinal case series of patients treated at 20 academic dermatology practices in the United States between January 2007 and December 2018. As in the PPP group, most patients in the GPP case series were women (70.5%), and over half were White (53.7%); the mean age was 50.3 years old, and the mean follow-up time was 19.8 months. A majority of patients with GPP were never-smokers (52.6%) or former smokers (20%). When patients with GPP initially presented to the study sites, 36.8% were admitted as inpatients, 9.5% presented in the emergency department, and 53.7% presented in an outpatient or ambulatory dermatology setting.
GPP commonly appeared on the trunk and extremities, but was “also reported on the scalp, face, genitals, nail unit, and mucous membranes in a minority of patients,” the researchers said. Overall, 62.1% of patients had skin pain, 26.2% had joint pain, 16.8% reported tachycardia, and 9.5% reported fever. Hypertension, depression, diabetes, chronic kidney disease, and hypothyroidism were common comorbidities of GPP, the researchers noted.
Clinicians reported treating GPP with topical steroids (86.3%) and topical treatments alone (32.3%). Oral systemic treatments such as acitretin (24.2%), cyclosporine (22.1%), and methotrexate (13.7%) were also used, as well as systemic steroids (20%). Other treatments used were narrowband UVB phototherapy (5.3%) and biologic agents like adalimumab (4.2%) and infliximab (4.2%).
For 53 patients with follow-up data of at least 6 months, 19 (35.8%) had been hospitalized because of their symptoms, and 8 patients were hospitalized for further GPP-specific concerns. Patients with GPP had a median 3.2 dermatology visits per year and a maximum of 18 visits. A model that was adjusted for age and sex showed women were at a decreased risk for being admitted to the hospital or emergency department in the follow-up period (odds ratio, 0.19; 95% confidence interval, 0.04-0.83).
PPP and GPP in practice
Sylvia Hsu, MD, professor and chair of the department of dermatology at Temple University, Philadelphia, who was not involved with the research, noted that most dermatologists will see few, if any, cases of PPP and GPP in a year. At her center, she estimated that she sees about one PPP case per week, and one or two cases of GPP a year. In general, she said that her clinical experience matched what was found by the authors of both case series.
For patients with PPP, “I would say the average dermatologist would probably start out with a superpotent topical steroid like clobetasol or halobetasol ointment,” Dr. Hsu said.
If they are not of childbearing age, she added, she would also prescribe acitretin, which she avoids giving to patients of childbearing age because of its teratogenicity. “Acitretin has the reputation that it doesn’t work well or fast for psoriasis. It doesn’t work well or fast for plaque-type psoriasis, but it works well and fast for pustular psoriasis,” she said.
In place of acitretin, Dr. Hsu recommended cyclosporine for a patient of childbearing age as a short-term solution to resolve symptoms before transitioning them to another therapy. “A woman of childbearing age, you put on cyclosporine, you’ve got to transition to something else,” she said. “And so many times you wean them off, the pustular psoriasis comes back because the topical steroid doesn’t work that well.”
One possible option is the interluekin-23 inhibitor guselkumab (approved by the Food and Drug Administration for treating moderate to severe plaque psoriasis and psoriatic arthritis) but cost and effectiveness can be a factor. Although studies have shown efficacy, biologics as treatments for PPP are “hit or miss,” Dr. Hsu said.
Regarding use of systemic therapies, Dr. Hsu cautioned against using them to treat plaque-type psoriasis. “We always learn, don’t use a systemic steroid like prednisone to treat psoriasis because it helps, but it comes back with a vengeance,” she said. “Sometimes when you treat plaque-type psoriasis with prednisone, it could come back with a vengeance, and it can come back as generalized pustular psoriasis.”
For patients with GPP, “you need a quick fix” because of the painful symptoms associated with the disease, Dr. Hsu said. In this case, she recommended cyclosporine and said she would avoid prescribing topical medications. “You’re going to have to give an oral drug because usually when we’re seeing somebody with GPP, they’re either a hospital consult or they just walked in the door,” she said. After prescribing cyclosporine, you would transition to another treatment like a biologic “as quickly as you can” with the knowledge that the biologic “may or may not work.”
New treatment options needed
Commenting on both case series in a related editorial, Edward W. Cowen, MD, MHSc, senior clinician and head of the dermatology consultation service in the dermatology branch of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Bethesda, Md., said that “much of the clinical presentation of pustular disease remains a mystery,” including why tobacco use is a risk factor for developing pustular psoriasis, and why tumor necrosis factor inhibitors “induce pustular disease in a small number of patients” with psoriasis vulgaris.
“Most importantly, we still do not know if localized and generalized pustular psoriasis all truly represent different variants of the same disease process, and if not, which biologic treatment represents the best option for a given clinical variant,” he wrote.
Dr. Cowen noted that the multi-institutional approach to collecting the retrospective data in these case series could be used as a “basic framework to build on for future clinical trials for rare skin diseases such as pustular psoriasis.”
In the interview, Dr. Noe said that she hoped that the “Pustular Psoriasis in the US Research Group” she and her coauthors created for the case series could help with the development of prospective clinical trials. “For pustular psoriasis and other rare diseases in dermatology, multi-institutional collaborations are necessary to conduct prospective research,” she said.
“While not directly studied in our research, I think it is important to consider the negative impact on quality of life, experienced by patients with pustular psoriasis. In our study, many patients experienced exacerbations of their disease over time, and it is important to consider the impact this has on patients,” she said in the interview. “Continued research on pustular psoriasis is necessary to decrease the negative impact of these diseases on the lives of our patients.”
The case series were funded in part by an institutional grant from Boehringer Ingelheim. The authors report relationships with various pharmaceutical and biopharmaceutical companies, technology companies, medical publishing companies, medical journals, and medical societies with connections to the topic area in the form of serving in roles as a chief medical editor, consultant, data safety monitoring board member, deputy editor, principal investigator, research investigator, scientific adviser, or speaker; or having received grants, honoraria, personal fees, or research funding. Dr. Cowen has no disclosures. Dr. Hsu reports serving on a Boehringer Ingelheim advisory board for a product being evaluated as a potential treatment for GPP.
FROM JAMA DERMATOLOGY
Atopic dermatitis can be especially burdensome in the elderly
During the Revolutionizing Atopic Dermatitis virtual symposium, Katrina Abuabara, MD, highlighted the epidemiology and burden of AD among older adults. She began by noting that the disease peaks in infancy and older adulthood. In an analysis that she and her colleagues made of physician-diagnosed AD among more than 8.6 million patients in the United Kingdom between 1994 and 2013, the mean prevalence in a given year was 12.3% among those aged 0-17 years, 5.1% among those age 18-74 years, and 8.7% among those age 75 and older.
“We saw what we expected in early infancy with very high rates of active disease,” said Dr. Abuabara, associate professor of dermatology and epidemiology at the University of California, San Francisco. “We also saw a second peak in older adulthood. This was more surprising to us because the disease hadn’t been as well studied in this population.” Researchers who analyzed data from the Global Burden of Disease Study, which evaluates disease-related morbidity and mortality worldwide, found a somewhat attenuated peak but a similar trend around the world. Its authors ranked AD as 15th among all nonfatal diseases.
In a separate analysis, Dr. Abuabara and colleagues evaluated records of more than 9.1 million primary care patients in the United Kingdom between 1994 and 2013, and who were followed for an average of 6 years. They examined AD activity and found that, based on doctor visits and prescriptions, AD appeared to be active in 48% of those aged 0-17 years, compared with 42% of those aged 18-74 years, and 60% of those aged 75 years and older. “Also, when we looked at the distribution of active disease in older adults, we saw that those who were older had more severe disease,” she said. When they evaluated the prevalence of AD by sociodemographic factors, AD increased with age among older adults (adjusted odd ratio, 1.06), while it decreased by 14% annually among children. In addition, female older adults had about three-fourths the odds of prevalent disease as their male counterparts (aOR, 0.73).
“We also looked at rural and urban differences and found that across ages it was more common in urban as compared to rural populations,” she said. “As for socioeconomic status, it tends to be more common among those of higher socioeconomic status in children and in the older adult group.”
In a study that drew from medical records of 3.85 million primary care patients in the United Kingdom, AD was more common in Asian and Black ethnic groups than in people of White ethnicity. In addition, higher socioeconomic status was associated with a greater incidence of eczema in infants aged younger than 2 years, but the reverse was seen for all other age groups.
To identify subtypes of atopic eczema based on patterns of disease activity through mid-adulthood, Dr. Abuabara and colleagues evaluated members of two population-based birth cohorts: the 1958 National Childhood Development Study and the 1970 British Cohort Study. The patients were classified into one of four patters of disease activity followed to age 50: rare/none, increasing, decreasing, and high. “We found that there was the early-onset decreasing subgroup, which tend to have a lower probability of AD over time,” Dr. Abuabara said. “We also found that there was a small subgroup that had a constant high probability of AD over time. But we were surprised to find a subgroup with increasing probability over time. This was a fairly sizable subgroup.”
In an earlier study, she and her colleagues examined whether there were differences based on whether people had adult-onset or childhood-onset disease in the same two cohorts of U.K. patients. Those with childhood-onset disease had stronger associations with known genetic risk factors and they tended to be of higher socioeconomic status. “They also tended to have more asthma and other allergic comorbidities,” Dr. Abuabara said. “On the other hand, the adult-onset group [after age 23] were more likely to be female, more likely to be smokers, and tended to have lower childhood socioeconomic status.”
According to the best available evidence, she continued, there is good data on higher relative risk of osteoporosis/fractures and dementia specifically among older adults with AD, and good data on associations with cardiometabolic disease and atopic disease among adults overall, as well as data showing that AD does not seem to be associated with cancer overall. In a study conducted by Jonathan I. Silverberg, MD, PhD, MPH, and Mohammed S. Shaheen, JD, the researchers used physician-diagnosed AD to investigate the associations of osteopenia and osteoporosis in two large U.S. databases: the 2006-2012 Nationwide Emergency Department Sample (NEDS) database and 2002-2012 National Inpatient Sample (NIS). Among patients aged 50 years and older, AD was associated with a higher odds of osteoporosis in NEDS (aOR, 1.31) and NIS (aOR, 1.25) and osteopenia in NEDS (aOR, 1.86).
In a separate matched cohort study, Dr. Abuabara and colleagues used U.K. primary care patient data to evaluate the association between AD and fracture and whether fracture risk varies with AD severity. Overall, they observed a 10% increase in fracture risk among people with AD, compared with those without, especially those of the hip, spine, pelvis, and wrist. “We found that there was a dose-response effect,” she said. “Those with more severe eczema had a much higher risk of fractures. When we looked at different age groups, we found a similar increased risk in the oldest adults as in younger adults.”
In a longitudinal cohort study of primary care medical records from more than 1.1 million individuals in the United Kingdom, AD was associated with an increased risk of vascular dementia (hazard ratio, 1.88), Alzheimer’s disease (HR, 1.69, and other/unspecified dementia (HR, 1.48; .269). “We found a nice dose response, where people with more severe AD had higher rates of dementia,” Dr. Abuabara said. Results from a more recent, smaller study of patients in Taiwan also found an increased risk between AD and the risk of dementia, but not a dose-response effect, likely because of a much smaller sample size.
Mounting research suggests that the risk for cardiovascular disease is also elevated in patients with AD. “There is some variability in the literature, but I think it’s important that when we’re talking about atopic dermatitis to think about the heterogeneity of the disease,” Dr. Abuabara said. In a meta-analysis and systematic review of 19 studies on the topic, she and her colleagues found that AD was associated with an increased risk of myocardial infarction (relative risk, 1.12), stroke (RR, 1.10), ischemic stroke (RR, 1.17), angina (RR, 1.18), and heart failure (RR, 1.26). “For all the different [cardiovascular disease] outcomes there was increasing risk with increasing disease severity,” she said.
She reported that UCSF receives research funding from Pfizer and Cosmetique Active International. She also receives consulting fees from Target RWE.
During the Revolutionizing Atopic Dermatitis virtual symposium, Katrina Abuabara, MD, highlighted the epidemiology and burden of AD among older adults. She began by noting that the disease peaks in infancy and older adulthood. In an analysis that she and her colleagues made of physician-diagnosed AD among more than 8.6 million patients in the United Kingdom between 1994 and 2013, the mean prevalence in a given year was 12.3% among those aged 0-17 years, 5.1% among those age 18-74 years, and 8.7% among those age 75 and older.
“We saw what we expected in early infancy with very high rates of active disease,” said Dr. Abuabara, associate professor of dermatology and epidemiology at the University of California, San Francisco. “We also saw a second peak in older adulthood. This was more surprising to us because the disease hadn’t been as well studied in this population.” Researchers who analyzed data from the Global Burden of Disease Study, which evaluates disease-related morbidity and mortality worldwide, found a somewhat attenuated peak but a similar trend around the world. Its authors ranked AD as 15th among all nonfatal diseases.
In a separate analysis, Dr. Abuabara and colleagues evaluated records of more than 9.1 million primary care patients in the United Kingdom between 1994 and 2013, and who were followed for an average of 6 years. They examined AD activity and found that, based on doctor visits and prescriptions, AD appeared to be active in 48% of those aged 0-17 years, compared with 42% of those aged 18-74 years, and 60% of those aged 75 years and older. “Also, when we looked at the distribution of active disease in older adults, we saw that those who were older had more severe disease,” she said. When they evaluated the prevalence of AD by sociodemographic factors, AD increased with age among older adults (adjusted odd ratio, 1.06), while it decreased by 14% annually among children. In addition, female older adults had about three-fourths the odds of prevalent disease as their male counterparts (aOR, 0.73).
“We also looked at rural and urban differences and found that across ages it was more common in urban as compared to rural populations,” she said. “As for socioeconomic status, it tends to be more common among those of higher socioeconomic status in children and in the older adult group.”
In a study that drew from medical records of 3.85 million primary care patients in the United Kingdom, AD was more common in Asian and Black ethnic groups than in people of White ethnicity. In addition, higher socioeconomic status was associated with a greater incidence of eczema in infants aged younger than 2 years, but the reverse was seen for all other age groups.
To identify subtypes of atopic eczema based on patterns of disease activity through mid-adulthood, Dr. Abuabara and colleagues evaluated members of two population-based birth cohorts: the 1958 National Childhood Development Study and the 1970 British Cohort Study. The patients were classified into one of four patters of disease activity followed to age 50: rare/none, increasing, decreasing, and high. “We found that there was the early-onset decreasing subgroup, which tend to have a lower probability of AD over time,” Dr. Abuabara said. “We also found that there was a small subgroup that had a constant high probability of AD over time. But we were surprised to find a subgroup with increasing probability over time. This was a fairly sizable subgroup.”
In an earlier study, she and her colleagues examined whether there were differences based on whether people had adult-onset or childhood-onset disease in the same two cohorts of U.K. patients. Those with childhood-onset disease had stronger associations with known genetic risk factors and they tended to be of higher socioeconomic status. “They also tended to have more asthma and other allergic comorbidities,” Dr. Abuabara said. “On the other hand, the adult-onset group [after age 23] were more likely to be female, more likely to be smokers, and tended to have lower childhood socioeconomic status.”
According to the best available evidence, she continued, there is good data on higher relative risk of osteoporosis/fractures and dementia specifically among older adults with AD, and good data on associations with cardiometabolic disease and atopic disease among adults overall, as well as data showing that AD does not seem to be associated with cancer overall. In a study conducted by Jonathan I. Silverberg, MD, PhD, MPH, and Mohammed S. Shaheen, JD, the researchers used physician-diagnosed AD to investigate the associations of osteopenia and osteoporosis in two large U.S. databases: the 2006-2012 Nationwide Emergency Department Sample (NEDS) database and 2002-2012 National Inpatient Sample (NIS). Among patients aged 50 years and older, AD was associated with a higher odds of osteoporosis in NEDS (aOR, 1.31) and NIS (aOR, 1.25) and osteopenia in NEDS (aOR, 1.86).
In a separate matched cohort study, Dr. Abuabara and colleagues used U.K. primary care patient data to evaluate the association between AD and fracture and whether fracture risk varies with AD severity. Overall, they observed a 10% increase in fracture risk among people with AD, compared with those without, especially those of the hip, spine, pelvis, and wrist. “We found that there was a dose-response effect,” she said. “Those with more severe eczema had a much higher risk of fractures. When we looked at different age groups, we found a similar increased risk in the oldest adults as in younger adults.”
In a longitudinal cohort study of primary care medical records from more than 1.1 million individuals in the United Kingdom, AD was associated with an increased risk of vascular dementia (hazard ratio, 1.88), Alzheimer’s disease (HR, 1.69, and other/unspecified dementia (HR, 1.48; .269). “We found a nice dose response, where people with more severe AD had higher rates of dementia,” Dr. Abuabara said. Results from a more recent, smaller study of patients in Taiwan also found an increased risk between AD and the risk of dementia, but not a dose-response effect, likely because of a much smaller sample size.
Mounting research suggests that the risk for cardiovascular disease is also elevated in patients with AD. “There is some variability in the literature, but I think it’s important that when we’re talking about atopic dermatitis to think about the heterogeneity of the disease,” Dr. Abuabara said. In a meta-analysis and systematic review of 19 studies on the topic, she and her colleagues found that AD was associated with an increased risk of myocardial infarction (relative risk, 1.12), stroke (RR, 1.10), ischemic stroke (RR, 1.17), angina (RR, 1.18), and heart failure (RR, 1.26). “For all the different [cardiovascular disease] outcomes there was increasing risk with increasing disease severity,” she said.
She reported that UCSF receives research funding from Pfizer and Cosmetique Active International. She also receives consulting fees from Target RWE.
During the Revolutionizing Atopic Dermatitis virtual symposium, Katrina Abuabara, MD, highlighted the epidemiology and burden of AD among older adults. She began by noting that the disease peaks in infancy and older adulthood. In an analysis that she and her colleagues made of physician-diagnosed AD among more than 8.6 million patients in the United Kingdom between 1994 and 2013, the mean prevalence in a given year was 12.3% among those aged 0-17 years, 5.1% among those age 18-74 years, and 8.7% among those age 75 and older.
“We saw what we expected in early infancy with very high rates of active disease,” said Dr. Abuabara, associate professor of dermatology and epidemiology at the University of California, San Francisco. “We also saw a second peak in older adulthood. This was more surprising to us because the disease hadn’t been as well studied in this population.” Researchers who analyzed data from the Global Burden of Disease Study, which evaluates disease-related morbidity and mortality worldwide, found a somewhat attenuated peak but a similar trend around the world. Its authors ranked AD as 15th among all nonfatal diseases.
In a separate analysis, Dr. Abuabara and colleagues evaluated records of more than 9.1 million primary care patients in the United Kingdom between 1994 and 2013, and who were followed for an average of 6 years. They examined AD activity and found that, based on doctor visits and prescriptions, AD appeared to be active in 48% of those aged 0-17 years, compared with 42% of those aged 18-74 years, and 60% of those aged 75 years and older. “Also, when we looked at the distribution of active disease in older adults, we saw that those who were older had more severe disease,” she said. When they evaluated the prevalence of AD by sociodemographic factors, AD increased with age among older adults (adjusted odd ratio, 1.06), while it decreased by 14% annually among children. In addition, female older adults had about three-fourths the odds of prevalent disease as their male counterparts (aOR, 0.73).
“We also looked at rural and urban differences and found that across ages it was more common in urban as compared to rural populations,” she said. “As for socioeconomic status, it tends to be more common among those of higher socioeconomic status in children and in the older adult group.”
In a study that drew from medical records of 3.85 million primary care patients in the United Kingdom, AD was more common in Asian and Black ethnic groups than in people of White ethnicity. In addition, higher socioeconomic status was associated with a greater incidence of eczema in infants aged younger than 2 years, but the reverse was seen for all other age groups.
To identify subtypes of atopic eczema based on patterns of disease activity through mid-adulthood, Dr. Abuabara and colleagues evaluated members of two population-based birth cohorts: the 1958 National Childhood Development Study and the 1970 British Cohort Study. The patients were classified into one of four patters of disease activity followed to age 50: rare/none, increasing, decreasing, and high. “We found that there was the early-onset decreasing subgroup, which tend to have a lower probability of AD over time,” Dr. Abuabara said. “We also found that there was a small subgroup that had a constant high probability of AD over time. But we were surprised to find a subgroup with increasing probability over time. This was a fairly sizable subgroup.”
In an earlier study, she and her colleagues examined whether there were differences based on whether people had adult-onset or childhood-onset disease in the same two cohorts of U.K. patients. Those with childhood-onset disease had stronger associations with known genetic risk factors and they tended to be of higher socioeconomic status. “They also tended to have more asthma and other allergic comorbidities,” Dr. Abuabara said. “On the other hand, the adult-onset group [after age 23] were more likely to be female, more likely to be smokers, and tended to have lower childhood socioeconomic status.”
According to the best available evidence, she continued, there is good data on higher relative risk of osteoporosis/fractures and dementia specifically among older adults with AD, and good data on associations with cardiometabolic disease and atopic disease among adults overall, as well as data showing that AD does not seem to be associated with cancer overall. In a study conducted by Jonathan I. Silverberg, MD, PhD, MPH, and Mohammed S. Shaheen, JD, the researchers used physician-diagnosed AD to investigate the associations of osteopenia and osteoporosis in two large U.S. databases: the 2006-2012 Nationwide Emergency Department Sample (NEDS) database and 2002-2012 National Inpatient Sample (NIS). Among patients aged 50 years and older, AD was associated with a higher odds of osteoporosis in NEDS (aOR, 1.31) and NIS (aOR, 1.25) and osteopenia in NEDS (aOR, 1.86).
In a separate matched cohort study, Dr. Abuabara and colleagues used U.K. primary care patient data to evaluate the association between AD and fracture and whether fracture risk varies with AD severity. Overall, they observed a 10% increase in fracture risk among people with AD, compared with those without, especially those of the hip, spine, pelvis, and wrist. “We found that there was a dose-response effect,” she said. “Those with more severe eczema had a much higher risk of fractures. When we looked at different age groups, we found a similar increased risk in the oldest adults as in younger adults.”
In a longitudinal cohort study of primary care medical records from more than 1.1 million individuals in the United Kingdom, AD was associated with an increased risk of vascular dementia (hazard ratio, 1.88), Alzheimer’s disease (HR, 1.69, and other/unspecified dementia (HR, 1.48; .269). “We found a nice dose response, where people with more severe AD had higher rates of dementia,” Dr. Abuabara said. Results from a more recent, smaller study of patients in Taiwan also found an increased risk between AD and the risk of dementia, but not a dose-response effect, likely because of a much smaller sample size.
Mounting research suggests that the risk for cardiovascular disease is also elevated in patients with AD. “There is some variability in the literature, but I think it’s important that when we’re talking about atopic dermatitis to think about the heterogeneity of the disease,” Dr. Abuabara said. In a meta-analysis and systematic review of 19 studies on the topic, she and her colleagues found that AD was associated with an increased risk of myocardial infarction (relative risk, 1.12), stroke (RR, 1.10), ischemic stroke (RR, 1.17), angina (RR, 1.18), and heart failure (RR, 1.26). “For all the different [cardiovascular disease] outcomes there was increasing risk with increasing disease severity,” she said.
She reported that UCSF receives research funding from Pfizer and Cosmetique Active International. She also receives consulting fees from Target RWE.
FROM REVOLUTIONIZING AD 2021
FDA to review PDE4-inhibitor roflumilast for psoriasis
The , according to a statement from the manufacturer.
Roflumilast cream (also known as ARQ-151) is a small molecule inhibitor of PDE4, an enzyme that increases proinflammatory mediators and decreases anti-inflammatory mediators. PDE4 is an established treatment target in dermatology: The FDA approved PDE-4 inhibitor crisaborole (Eucrisa) as a topical treatment for mild to moderate atopic dermatitis in 2016, and an oral PDE-4 inhibitor, orismilast, is being studied for the treatment of plaque psoriasis.
Topical roflumilast, if approved, would be the first topical PDE4 inhibitor for psoriasis in particular, according to the Arcutis Biotherapeutics statement. The cream is designed for use on the entire body, including the face and sensitive intertriginous areas.
The NDA is based on data from a pair of phase 3 randomized, double-blind 8-week studies known as DERMIS 1 and DERMIS 2 (Trials of PDE4 Inhibition with Roflumilast for the Management of Plaque Psoriasis” One and Two) and a long-term phase 2b open-label study.
DERMIS 1 and DERMIS 2 were identical multinational, multicenter studies designed to assess the safety and efficacy of 0.3% roflumilast cream. In the studies, roflumilast met its primary endpoint and patients treated with it demonstrated an Investigator Global Assessment (IGA) success rate of 42.4% compared with 6.1% for the vehicle control (P < .0001), and 37.5% compared with 6.9% for the vehicle control (P < .0001), in the DERMIS 1 and 2 trials, respectively, according to Arcutis.
In the phase 2b study, the treatment effect lasted for 52-64 weeks. Roflumilast was well tolerated across the three studies.
Overall, the most common adverse events reported in the studies were diarrhea (3%), headache (2%), insomnia (1%), nausea (1%), upper respiratory tract infections (1%), and urinary tract infections (1%).
Roflumilast also showed statistically significant improvement compared to a vehicle on secondary endpoints including Intertriginous IGA (I-IGA) Success, Psoriasis Area Severity Index-75 (PASI-75), reductions in itch as measured by the Worst Itch-Numerical Rating Scale (WI-NRS), and patient perceptions of symptoms based on the Psoriasis Symptoms Diary (PSD).
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of July 29, 2022, according to the manufacturer’s statement. An oral formulation of roflumilast was approved by the FDA in 2011, for reducing the risk of exacerbations of chronic obstructive pulmonary disease (COPD) in patients with severe COPD.
The , according to a statement from the manufacturer.
Roflumilast cream (also known as ARQ-151) is a small molecule inhibitor of PDE4, an enzyme that increases proinflammatory mediators and decreases anti-inflammatory mediators. PDE4 is an established treatment target in dermatology: The FDA approved PDE-4 inhibitor crisaborole (Eucrisa) as a topical treatment for mild to moderate atopic dermatitis in 2016, and an oral PDE-4 inhibitor, orismilast, is being studied for the treatment of plaque psoriasis.
Topical roflumilast, if approved, would be the first topical PDE4 inhibitor for psoriasis in particular, according to the Arcutis Biotherapeutics statement. The cream is designed for use on the entire body, including the face and sensitive intertriginous areas.
The NDA is based on data from a pair of phase 3 randomized, double-blind 8-week studies known as DERMIS 1 and DERMIS 2 (Trials of PDE4 Inhibition with Roflumilast for the Management of Plaque Psoriasis” One and Two) and a long-term phase 2b open-label study.
DERMIS 1 and DERMIS 2 were identical multinational, multicenter studies designed to assess the safety and efficacy of 0.3% roflumilast cream. In the studies, roflumilast met its primary endpoint and patients treated with it demonstrated an Investigator Global Assessment (IGA) success rate of 42.4% compared with 6.1% for the vehicle control (P < .0001), and 37.5% compared with 6.9% for the vehicle control (P < .0001), in the DERMIS 1 and 2 trials, respectively, according to Arcutis.
In the phase 2b study, the treatment effect lasted for 52-64 weeks. Roflumilast was well tolerated across the three studies.
Overall, the most common adverse events reported in the studies were diarrhea (3%), headache (2%), insomnia (1%), nausea (1%), upper respiratory tract infections (1%), and urinary tract infections (1%).
Roflumilast also showed statistically significant improvement compared to a vehicle on secondary endpoints including Intertriginous IGA (I-IGA) Success, Psoriasis Area Severity Index-75 (PASI-75), reductions in itch as measured by the Worst Itch-Numerical Rating Scale (WI-NRS), and patient perceptions of symptoms based on the Psoriasis Symptoms Diary (PSD).
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of July 29, 2022, according to the manufacturer’s statement. An oral formulation of roflumilast was approved by the FDA in 2011, for reducing the risk of exacerbations of chronic obstructive pulmonary disease (COPD) in patients with severe COPD.
The , according to a statement from the manufacturer.
Roflumilast cream (also known as ARQ-151) is a small molecule inhibitor of PDE4, an enzyme that increases proinflammatory mediators and decreases anti-inflammatory mediators. PDE4 is an established treatment target in dermatology: The FDA approved PDE-4 inhibitor crisaborole (Eucrisa) as a topical treatment for mild to moderate atopic dermatitis in 2016, and an oral PDE-4 inhibitor, orismilast, is being studied for the treatment of plaque psoriasis.
Topical roflumilast, if approved, would be the first topical PDE4 inhibitor for psoriasis in particular, according to the Arcutis Biotherapeutics statement. The cream is designed for use on the entire body, including the face and sensitive intertriginous areas.
The NDA is based on data from a pair of phase 3 randomized, double-blind 8-week studies known as DERMIS 1 and DERMIS 2 (Trials of PDE4 Inhibition with Roflumilast for the Management of Plaque Psoriasis” One and Two) and a long-term phase 2b open-label study.
DERMIS 1 and DERMIS 2 were identical multinational, multicenter studies designed to assess the safety and efficacy of 0.3% roflumilast cream. In the studies, roflumilast met its primary endpoint and patients treated with it demonstrated an Investigator Global Assessment (IGA) success rate of 42.4% compared with 6.1% for the vehicle control (P < .0001), and 37.5% compared with 6.9% for the vehicle control (P < .0001), in the DERMIS 1 and 2 trials, respectively, according to Arcutis.
In the phase 2b study, the treatment effect lasted for 52-64 weeks. Roflumilast was well tolerated across the three studies.
Overall, the most common adverse events reported in the studies were diarrhea (3%), headache (2%), insomnia (1%), nausea (1%), upper respiratory tract infections (1%), and urinary tract infections (1%).
Roflumilast also showed statistically significant improvement compared to a vehicle on secondary endpoints including Intertriginous IGA (I-IGA) Success, Psoriasis Area Severity Index-75 (PASI-75), reductions in itch as measured by the Worst Itch-Numerical Rating Scale (WI-NRS), and patient perceptions of symptoms based on the Psoriasis Symptoms Diary (PSD).
The FDA has set a Prescription Drug User Fee Act (PDUFA) target action date of July 29, 2022, according to the manufacturer’s statement. An oral formulation of roflumilast was approved by the FDA in 2011, for reducing the risk of exacerbations of chronic obstructive pulmonary disease (COPD) in patients with severe COPD.
FDA gives nod to tralokinumab for adults with moderate to severe AD
whose disease is not well controlled with topical prescription therapies or when those therapies are not advisable.
Administered subcutaneously, tralokinumab is a fully human IgG4 monoclonal antibody that specifically binds to interleukin-13, a key driver of underlying inflammation in AD. The drug, which has been developed by LEO Pharma, comes as a single-dose (150 mg) prefilled syringe with needle guard.
In two pivotal phase 3 trials, ECZTRA 1 and ECZTRA 2, tralokinumab monotherapy was superior to placebo at week 16 for all primary and secondary endpoints. For example, at week 16, for the ECZTRA 1 and 2 monotherapy trials, respectively, 16% and 21% of patients treated with tralokinumab 300 mg every other week achieved clear or almost clear skin (IGA 0/1) versus 7% and 9% with placebo.
In addition, 25% and 33% of patients treated with tralokinumab 300 mg every other week achieved an improvement of 75% or more in the Eczema Area and Severity Index score (EASI-75) versus 13% and 10% with placebo. At 52 weeks, 51% and 60% of patients who responded at week 16 maintained IGA 0/1 response with tralokinumab 300 mg every other week in ECZTRA 1 and 2, respectively.
Finally, 60% and 57% of patients who responded at week 16 maintained EASI-75 response with tralokinumab 300 mg every other week.
In the drug’s third pivotal trial, ECZTRA 3, researchers evaluated the efficacy and safety of tralokinumab 300 mg in combination with topical corticosteroids (TCS) as needed in adults with moderate to severe atopic dermatitis who are candidates for systemic therapy. At week 16, 38% of patients treated with tralokinumab 300 mg every other week plus TCS achieved clear or almost clear skin (IGA 0/1) versus 27% with placebo plus TCS. In addition, 56% of patients treated with tralokinumab 300 mg every other week plus TCS achieved an improvement of 75% or more in the EASI-75 versus 37% with placebo plus TCS. At 32 weeks, 89% and 92% of patients who responded at week 16 maintained response (IGA 0/1 and EASI-75, respectively) with tralokinumab 300 mg every other week.
A link to prescribing information can be found here. Tralokinumab is expected to be available by February 2022.
whose disease is not well controlled with topical prescription therapies or when those therapies are not advisable.
Administered subcutaneously, tralokinumab is a fully human IgG4 monoclonal antibody that specifically binds to interleukin-13, a key driver of underlying inflammation in AD. The drug, which has been developed by LEO Pharma, comes as a single-dose (150 mg) prefilled syringe with needle guard.
In two pivotal phase 3 trials, ECZTRA 1 and ECZTRA 2, tralokinumab monotherapy was superior to placebo at week 16 for all primary and secondary endpoints. For example, at week 16, for the ECZTRA 1 and 2 monotherapy trials, respectively, 16% and 21% of patients treated with tralokinumab 300 mg every other week achieved clear or almost clear skin (IGA 0/1) versus 7% and 9% with placebo.
In addition, 25% and 33% of patients treated with tralokinumab 300 mg every other week achieved an improvement of 75% or more in the Eczema Area and Severity Index score (EASI-75) versus 13% and 10% with placebo. At 52 weeks, 51% and 60% of patients who responded at week 16 maintained IGA 0/1 response with tralokinumab 300 mg every other week in ECZTRA 1 and 2, respectively.
Finally, 60% and 57% of patients who responded at week 16 maintained EASI-75 response with tralokinumab 300 mg every other week.
In the drug’s third pivotal trial, ECZTRA 3, researchers evaluated the efficacy and safety of tralokinumab 300 mg in combination with topical corticosteroids (TCS) as needed in adults with moderate to severe atopic dermatitis who are candidates for systemic therapy. At week 16, 38% of patients treated with tralokinumab 300 mg every other week plus TCS achieved clear or almost clear skin (IGA 0/1) versus 27% with placebo plus TCS. In addition, 56% of patients treated with tralokinumab 300 mg every other week plus TCS achieved an improvement of 75% or more in the EASI-75 versus 37% with placebo plus TCS. At 32 weeks, 89% and 92% of patients who responded at week 16 maintained response (IGA 0/1 and EASI-75, respectively) with tralokinumab 300 mg every other week.
A link to prescribing information can be found here. Tralokinumab is expected to be available by February 2022.
whose disease is not well controlled with topical prescription therapies or when those therapies are not advisable.
Administered subcutaneously, tralokinumab is a fully human IgG4 monoclonal antibody that specifically binds to interleukin-13, a key driver of underlying inflammation in AD. The drug, which has been developed by LEO Pharma, comes as a single-dose (150 mg) prefilled syringe with needle guard.
In two pivotal phase 3 trials, ECZTRA 1 and ECZTRA 2, tralokinumab monotherapy was superior to placebo at week 16 for all primary and secondary endpoints. For example, at week 16, for the ECZTRA 1 and 2 monotherapy trials, respectively, 16% and 21% of patients treated with tralokinumab 300 mg every other week achieved clear or almost clear skin (IGA 0/1) versus 7% and 9% with placebo.
In addition, 25% and 33% of patients treated with tralokinumab 300 mg every other week achieved an improvement of 75% or more in the Eczema Area and Severity Index score (EASI-75) versus 13% and 10% with placebo. At 52 weeks, 51% and 60% of patients who responded at week 16 maintained IGA 0/1 response with tralokinumab 300 mg every other week in ECZTRA 1 and 2, respectively.
Finally, 60% and 57% of patients who responded at week 16 maintained EASI-75 response with tralokinumab 300 mg every other week.
In the drug’s third pivotal trial, ECZTRA 3, researchers evaluated the efficacy and safety of tralokinumab 300 mg in combination with topical corticosteroids (TCS) as needed in adults with moderate to severe atopic dermatitis who are candidates for systemic therapy. At week 16, 38% of patients treated with tralokinumab 300 mg every other week plus TCS achieved clear or almost clear skin (IGA 0/1) versus 27% with placebo plus TCS. In addition, 56% of patients treated with tralokinumab 300 mg every other week plus TCS achieved an improvement of 75% or more in the EASI-75 versus 37% with placebo plus TCS. At 32 weeks, 89% and 92% of patients who responded at week 16 maintained response (IGA 0/1 and EASI-75, respectively) with tralokinumab 300 mg every other week.
A link to prescribing information can be found here. Tralokinumab is expected to be available by February 2022.
High-fiber diet may improve melanoma immunotherapy response, outcomes
a new study shows.
Investigators found that the patients who reported consuming at least 20 g of dietary fiber daily had significantly better progression-free survival (PFS) than those who reported consuming lower amounts of dietary fiber. However, patients who took a probiotic supplement in the past month had slightly shorter PFS, but the results were not statistically significant.
And after adjusting for clinical factors, each 5-g increase in daily dietary fiber intake corresponded to a 30% lower risk of disease progression, according to the analysis, published online Dec. 23, 2021, in Science.
“Our study sheds light on the potential effects of a patient’s diet and supplement use when starting treatment with immune checkpoint blockade,” co–lead study author Jennifer Wargo, MD, professor of genomic medicine and surgical oncology at University of Texas MD Anderson Cancer Center, Houston, said in a press release. “These results provide further support for clinical trials to modulate the microbiome with the goal of improving cancer outcomes using dietary and other strategies.”
Previous research has suggested that the microbiome can influence patients’ response to immunotherapy. One recent analysis, for instance, found that fecal microbiota transplant can improve response to immunotherapy in advanced melanoma. And a small 2019 analysis from Dr. Dr. Wargo and colleagues hinted that a high-fiber diet may enhance patients’ ability to respond to immunotherapy in advanced melanoma, while probiotics appear to dampen that response.
Still, the role diet and probiotic supplements play in treatment response remains poorly understood.
In the current study, Dr. Wargo and colleagues assessed fecal microbiota profiles and dietary habits, including fiber intake and probiotic use, in 158 patients with advanced melanoma who received immune checkpoint blockade inhibitors.
In the cohort, 31% (49 of 158) of late-stage melanoma patients reported taking a commercially available probiotic in the past month. When assessing whether probiotic use influenced patient outcomes, the investigators observed a shorter but not statistically significant difference in PFS in those who took a probiotic (median, 17 months) versus those who did not (23 months).
Higher dietary fiber, however, was associated with significantly improved PFS in a subset of 128 patients. The team divided patients into a higher-fiber intake group (those consuming at least 20 g/day) and a low-fiber group (those consuming less than 20 g).
The 37 patients reporting higher fiber intake demonstrated improved PFS, compared with those in the low-intake group (median PFS not reached vs. 13 months), plus a 30% lower risk of disease progression or death for each additional 5 g consumed each day.
“The observed protective effect of dietary fiber intake in relation to PFS and response remained consistent among the subset of patients treated with anti–PD-1 monotherapy, with the exclusion of patients reporting recent antibiotic use,” the authors noted.
When assessing fiber and probiotic intake together, the researchers found that immunotherapy response rate was higher (82%) in the 22 patients who reported sufficient dietary fiber intake with no probiotic use versus 59% in 101 patients who reported either insufficient fiber intake or probiotic use.
Overall, the research suggests that “consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy,” co–lead author Giorgio Trinchieri, MD, chief of the Laboratory of Integrative Cancer Immunology in the National Cancer Institute’s Center for Cancer Research, Bethesda, Md., said in a press statement. “The data also suggest that it’s probably better for people with cancer receiving immunotherapy not to use commercially available probiotics.”
The investigators also explored whether dietary fiber intake enhanced treatment response in preclinical mouse models of melanoma. In this instance, mice receiving a fiber-rich diet showed delayed tumor growth after anti–PD-1 treatment, compared with mice given a low-fiber diet or probiotics.
According to the authors, “our preclinical models support the hypothesis that dietary fiber and probiotics modulate the microbiome and that antitumor immunity is impaired in mice receiving a low-fiber diet and in those receiving probiotics – with suppression of intratumoral [interferon-gamma] T-cell responses in both cases.”
Dietary fiber may exert beneficial effect by increasing specific types of bacteria in the gut, such as Ruminococcaceae, which “produce high levels of certain short-chain fatty acids that have an antitumor effect,” Dr. Trinchieri explained.
However, “the impact of dietary fiber and probiotics on the gut microbiota is only part of the bigger picture,” Dr. Trinchieri said in a press release. “Many factors can affect the ability of a patient with melanoma to respond to immunotherapy” but, according to this analysis, “the microbiota seems to be one of the dominant factors.”
While Jeffrey S. Weber, MD, PhD, applauded the “innovative and interesting” research, he believes the patient population is too small to confirm that a high-fiber diet does indeed contribute to improved immunotherapy response and PFS in patients with advanced melanoma.
Additional data are needed to clarify these findings. “I will believe it if I could see it replicated in a larger study,” Dr. Weber, professor and deputy director of the Laura and Isaac Perlmutter Cancer Center, New York University, said in an interview.
Dr. Wargo noted that a randomized clinical trial exploring how diets with varying fiber content affect the microbiome and immune response is currently enrolling patients with stage III and IV melanoma receiving immunotherapy.
This study was supported by the Melanoma Moon Shot, among others. Dr. Wargo is a collaborator on a U.S. patent application that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome. Dr. Weber reported relationships with Bristol-Myers Squibb, GlaxoSmithKline, Genentech BioOncology, Merck, Novartis, EMD Serono, Celldex, CytomX, Nektar, Roche, Altor, Daiichi Sankyo, and Eli Lilly, and is named on patents filed for biomarkers for ipilimumab and nivolumab.
A version of this article first appeared on Medscape.com.
a new study shows.
Investigators found that the patients who reported consuming at least 20 g of dietary fiber daily had significantly better progression-free survival (PFS) than those who reported consuming lower amounts of dietary fiber. However, patients who took a probiotic supplement in the past month had slightly shorter PFS, but the results were not statistically significant.
And after adjusting for clinical factors, each 5-g increase in daily dietary fiber intake corresponded to a 30% lower risk of disease progression, according to the analysis, published online Dec. 23, 2021, in Science.
“Our study sheds light on the potential effects of a patient’s diet and supplement use when starting treatment with immune checkpoint blockade,” co–lead study author Jennifer Wargo, MD, professor of genomic medicine and surgical oncology at University of Texas MD Anderson Cancer Center, Houston, said in a press release. “These results provide further support for clinical trials to modulate the microbiome with the goal of improving cancer outcomes using dietary and other strategies.”
Previous research has suggested that the microbiome can influence patients’ response to immunotherapy. One recent analysis, for instance, found that fecal microbiota transplant can improve response to immunotherapy in advanced melanoma. And a small 2019 analysis from Dr. Dr. Wargo and colleagues hinted that a high-fiber diet may enhance patients’ ability to respond to immunotherapy in advanced melanoma, while probiotics appear to dampen that response.
Still, the role diet and probiotic supplements play in treatment response remains poorly understood.
In the current study, Dr. Wargo and colleagues assessed fecal microbiota profiles and dietary habits, including fiber intake and probiotic use, in 158 patients with advanced melanoma who received immune checkpoint blockade inhibitors.
In the cohort, 31% (49 of 158) of late-stage melanoma patients reported taking a commercially available probiotic in the past month. When assessing whether probiotic use influenced patient outcomes, the investigators observed a shorter but not statistically significant difference in PFS in those who took a probiotic (median, 17 months) versus those who did not (23 months).
Higher dietary fiber, however, was associated with significantly improved PFS in a subset of 128 patients. The team divided patients into a higher-fiber intake group (those consuming at least 20 g/day) and a low-fiber group (those consuming less than 20 g).
The 37 patients reporting higher fiber intake demonstrated improved PFS, compared with those in the low-intake group (median PFS not reached vs. 13 months), plus a 30% lower risk of disease progression or death for each additional 5 g consumed each day.
“The observed protective effect of dietary fiber intake in relation to PFS and response remained consistent among the subset of patients treated with anti–PD-1 monotherapy, with the exclusion of patients reporting recent antibiotic use,” the authors noted.
When assessing fiber and probiotic intake together, the researchers found that immunotherapy response rate was higher (82%) in the 22 patients who reported sufficient dietary fiber intake with no probiotic use versus 59% in 101 patients who reported either insufficient fiber intake or probiotic use.
Overall, the research suggests that “consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy,” co–lead author Giorgio Trinchieri, MD, chief of the Laboratory of Integrative Cancer Immunology in the National Cancer Institute’s Center for Cancer Research, Bethesda, Md., said in a press statement. “The data also suggest that it’s probably better for people with cancer receiving immunotherapy not to use commercially available probiotics.”
The investigators also explored whether dietary fiber intake enhanced treatment response in preclinical mouse models of melanoma. In this instance, mice receiving a fiber-rich diet showed delayed tumor growth after anti–PD-1 treatment, compared with mice given a low-fiber diet or probiotics.
According to the authors, “our preclinical models support the hypothesis that dietary fiber and probiotics modulate the microbiome and that antitumor immunity is impaired in mice receiving a low-fiber diet and in those receiving probiotics – with suppression of intratumoral [interferon-gamma] T-cell responses in both cases.”
Dietary fiber may exert beneficial effect by increasing specific types of bacteria in the gut, such as Ruminococcaceae, which “produce high levels of certain short-chain fatty acids that have an antitumor effect,” Dr. Trinchieri explained.
However, “the impact of dietary fiber and probiotics on the gut microbiota is only part of the bigger picture,” Dr. Trinchieri said in a press release. “Many factors can affect the ability of a patient with melanoma to respond to immunotherapy” but, according to this analysis, “the microbiota seems to be one of the dominant factors.”
While Jeffrey S. Weber, MD, PhD, applauded the “innovative and interesting” research, he believes the patient population is too small to confirm that a high-fiber diet does indeed contribute to improved immunotherapy response and PFS in patients with advanced melanoma.
Additional data are needed to clarify these findings. “I will believe it if I could see it replicated in a larger study,” Dr. Weber, professor and deputy director of the Laura and Isaac Perlmutter Cancer Center, New York University, said in an interview.
Dr. Wargo noted that a randomized clinical trial exploring how diets with varying fiber content affect the microbiome and immune response is currently enrolling patients with stage III and IV melanoma receiving immunotherapy.
This study was supported by the Melanoma Moon Shot, among others. Dr. Wargo is a collaborator on a U.S. patent application that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome. Dr. Weber reported relationships with Bristol-Myers Squibb, GlaxoSmithKline, Genentech BioOncology, Merck, Novartis, EMD Serono, Celldex, CytomX, Nektar, Roche, Altor, Daiichi Sankyo, and Eli Lilly, and is named on patents filed for biomarkers for ipilimumab and nivolumab.
A version of this article first appeared on Medscape.com.
a new study shows.
Investigators found that the patients who reported consuming at least 20 g of dietary fiber daily had significantly better progression-free survival (PFS) than those who reported consuming lower amounts of dietary fiber. However, patients who took a probiotic supplement in the past month had slightly shorter PFS, but the results were not statistically significant.
And after adjusting for clinical factors, each 5-g increase in daily dietary fiber intake corresponded to a 30% lower risk of disease progression, according to the analysis, published online Dec. 23, 2021, in Science.
“Our study sheds light on the potential effects of a patient’s diet and supplement use when starting treatment with immune checkpoint blockade,” co–lead study author Jennifer Wargo, MD, professor of genomic medicine and surgical oncology at University of Texas MD Anderson Cancer Center, Houston, said in a press release. “These results provide further support for clinical trials to modulate the microbiome with the goal of improving cancer outcomes using dietary and other strategies.”
Previous research has suggested that the microbiome can influence patients’ response to immunotherapy. One recent analysis, for instance, found that fecal microbiota transplant can improve response to immunotherapy in advanced melanoma. And a small 2019 analysis from Dr. Dr. Wargo and colleagues hinted that a high-fiber diet may enhance patients’ ability to respond to immunotherapy in advanced melanoma, while probiotics appear to dampen that response.
Still, the role diet and probiotic supplements play in treatment response remains poorly understood.
In the current study, Dr. Wargo and colleagues assessed fecal microbiota profiles and dietary habits, including fiber intake and probiotic use, in 158 patients with advanced melanoma who received immune checkpoint blockade inhibitors.
In the cohort, 31% (49 of 158) of late-stage melanoma patients reported taking a commercially available probiotic in the past month. When assessing whether probiotic use influenced patient outcomes, the investigators observed a shorter but not statistically significant difference in PFS in those who took a probiotic (median, 17 months) versus those who did not (23 months).
Higher dietary fiber, however, was associated with significantly improved PFS in a subset of 128 patients. The team divided patients into a higher-fiber intake group (those consuming at least 20 g/day) and a low-fiber group (those consuming less than 20 g).
The 37 patients reporting higher fiber intake demonstrated improved PFS, compared with those in the low-intake group (median PFS not reached vs. 13 months), plus a 30% lower risk of disease progression or death for each additional 5 g consumed each day.
“The observed protective effect of dietary fiber intake in relation to PFS and response remained consistent among the subset of patients treated with anti–PD-1 monotherapy, with the exclusion of patients reporting recent antibiotic use,” the authors noted.
When assessing fiber and probiotic intake together, the researchers found that immunotherapy response rate was higher (82%) in the 22 patients who reported sufficient dietary fiber intake with no probiotic use versus 59% in 101 patients who reported either insufficient fiber intake or probiotic use.
Overall, the research suggests that “consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy,” co–lead author Giorgio Trinchieri, MD, chief of the Laboratory of Integrative Cancer Immunology in the National Cancer Institute’s Center for Cancer Research, Bethesda, Md., said in a press statement. “The data also suggest that it’s probably better for people with cancer receiving immunotherapy not to use commercially available probiotics.”
The investigators also explored whether dietary fiber intake enhanced treatment response in preclinical mouse models of melanoma. In this instance, mice receiving a fiber-rich diet showed delayed tumor growth after anti–PD-1 treatment, compared with mice given a low-fiber diet or probiotics.
According to the authors, “our preclinical models support the hypothesis that dietary fiber and probiotics modulate the microbiome and that antitumor immunity is impaired in mice receiving a low-fiber diet and in those receiving probiotics – with suppression of intratumoral [interferon-gamma] T-cell responses in both cases.”
Dietary fiber may exert beneficial effect by increasing specific types of bacteria in the gut, such as Ruminococcaceae, which “produce high levels of certain short-chain fatty acids that have an antitumor effect,” Dr. Trinchieri explained.
However, “the impact of dietary fiber and probiotics on the gut microbiota is only part of the bigger picture,” Dr. Trinchieri said in a press release. “Many factors can affect the ability of a patient with melanoma to respond to immunotherapy” but, according to this analysis, “the microbiota seems to be one of the dominant factors.”
While Jeffrey S. Weber, MD, PhD, applauded the “innovative and interesting” research, he believes the patient population is too small to confirm that a high-fiber diet does indeed contribute to improved immunotherapy response and PFS in patients with advanced melanoma.
Additional data are needed to clarify these findings. “I will believe it if I could see it replicated in a larger study,” Dr. Weber, professor and deputy director of the Laura and Isaac Perlmutter Cancer Center, New York University, said in an interview.
Dr. Wargo noted that a randomized clinical trial exploring how diets with varying fiber content affect the microbiome and immune response is currently enrolling patients with stage III and IV melanoma receiving immunotherapy.
This study was supported by the Melanoma Moon Shot, among others. Dr. Wargo is a collaborator on a U.S. patent application that covers methods to enhance immune checkpoint blockade responses by modulating the microbiome. Dr. Weber reported relationships with Bristol-Myers Squibb, GlaxoSmithKline, Genentech BioOncology, Merck, Novartis, EMD Serono, Celldex, CytomX, Nektar, Roche, Altor, Daiichi Sankyo, and Eli Lilly, and is named on patents filed for biomarkers for ipilimumab and nivolumab.
A version of this article first appeared on Medscape.com.
FROM NATURE
Spesolimab speeds lesion clearance in generalized pustular psoriasis
GPP is a life-threatening skin condition involving the widespread eruption of sterile pustules, with a clinical course that “can be relapsing with recurrent flares or persistent with intermittent flares,” Hervé Bachelez, MD, of the Université de Paris and coauthors wrote. GPP patients are often hospitalized, and mortality ranges from 2% to 16% from causes that include sepsis and cardiorespiratory failure.
“The role of the interleukin-36 pathway in GPP is supported by the finding of loss-of-function mutations in the interleukin-36 receptor antagonist gene (IL36RN) and associated genes (CARD14, AP1S3, SERPINA3, and MPO) and by the overexpression of interleukin-36 cytokines in GPP skin lesions,” therefore, IL-36 is a potential treatment target to manage flares, they explained.
In the multicenter, double-blind trial, published in the New England Journal of Medicine, the researchers randomized 35 adults with GPP flares to a single 900-mg intravenous dose of spesolimab and 18 to placebo. Patients in both groups could receive an open-label dose of spesolimab after day 8; all patients were followed for 12 weeks.
The primary study endpoint was the Generalized Pustular Psoriasis Physician Global Assessment (GPPGA) pustulation subscore of 0 at 1 week after treatment. The GPPGA ranges from 0 (no visible pustules) to 4 (severe pustules). At baseline, 46% spesolimab patients and 39% placebo patients had a GPPGA pustulation subscore of 3, and 37% and 33%, respectively, had a pustulation subscore of 4.
After 1 week, 54% of the spesolimab patients had no visible pustules, compared with 6% of placebo patients; the difference was statistically significant (P < .001). The main secondary endpoint was a score of 0 or 1 (clear or almost clear skin) on the GPPGA total score after 1 week. Significantly more spesolimab patients had GPPGA total scores of 0 or 1, compared with placebo patients (43% vs. 11%, respectively; P = .02).
Overall, 6 of 35 spesolimab patients (17%) and 6% of those in the placebo groups developed infections during the first week, and 24 of 51 patients (47%) who had received spesolimab at any point during the study developed infections by week 12. Infections included urinary tract infections (three cases), influenza (three), otitis externa (two), folliculitis (two), upper respiratory tract infection (two), and pustule (two).
In the first week, 6% of spesolimab patients and none of the placebo patients reported serious adverse events; at week 12, 12% of patients who had received at least one spesolimab dose reported a serious adverse event. In addition, antidrug antibodies were identified in 23 (46%) of the 50 patients who received at least one dose of spesolimab.
“Symptoms that were observed in two patients who received spesolimab were reported as a drug reaction with eosinophilia and systemic symptoms (DRESS),” the authors noted. One patient had a RegiSCAR (European Registry of Severe Cutaneous Adverse Reactions) score and the other had a score of 3; a score below 2 indicates no DRESS, and a score of 2 or 3 indicates “possible DRESS,” they added.
“Because 15 of the 18 patients who were assigned to the placebo group received open-label spesolimab, the effect of spesolimab as compared with that of placebo could not be determined after week 1,” the researchers noted.
The study findings were limited by several factors including the short randomization period and small study population, the researchers noted. However, the effect sizes for both the primary and secondary endpoints were large, which strengthened the results.
The results support data from previous studies suggesting a role for IL-36 in the pathogenesis of GPP, and support the need for longer and larger studies of the safety and effectiveness of spesolimab for GPP patients, they concluded.
No FDA-approved therapy
“GPP is a very rare but devastating life-threatening disease that presents with the sudden onset of pustules throughout the skin,” Joel Gelfand, MD, professor of dermatology and director of the psoriasis and phototherapy center at the University of Pennsylvania, Philadelphia, said in an interview. “Without rapid treatment, GPP can result in death. Currently there are no [Food and Drug Administration]–approved treatments for this orphan disease.”
Dr. Gelfand said he was surprised by the degree of efficacy and the speed of the patient response to spesolimab, compared with placebo, which he described as “truly remarkable.” Based on the current study results, “spesolimab offers a tremendous step forward for our patients,” he added.
Looking ahead, Dr. Gelfand noted that “longer-term studies with a comparator, such as a biologic that targets IL-17, would be helpful to more fully understand the safety, efficacy, and role that spesolimab will have in real-world patients.”
On Dec. 15, Boehringer Ingelheim announced that the FDA had granted priority review for spesolimab for treating GPP flares.
The study was supported by Boehringer Ingelheim. Lead author Dr. Bachelez had no financial conflicts to disclose. Several authors are employees of Boehringer Ingelheim. Dr. Gelfand is a consultant for the study sponsor Boehringer Ingelheim and has received research grants from Boehringer Ingelheim to his institution to support an investigator-initiated study. He also disclosed serving as a consultant and receiving research grants from other manufacturers of psoriasis products.
GPP is a life-threatening skin condition involving the widespread eruption of sterile pustules, with a clinical course that “can be relapsing with recurrent flares or persistent with intermittent flares,” Hervé Bachelez, MD, of the Université de Paris and coauthors wrote. GPP patients are often hospitalized, and mortality ranges from 2% to 16% from causes that include sepsis and cardiorespiratory failure.
“The role of the interleukin-36 pathway in GPP is supported by the finding of loss-of-function mutations in the interleukin-36 receptor antagonist gene (IL36RN) and associated genes (CARD14, AP1S3, SERPINA3, and MPO) and by the overexpression of interleukin-36 cytokines in GPP skin lesions,” therefore, IL-36 is a potential treatment target to manage flares, they explained.
In the multicenter, double-blind trial, published in the New England Journal of Medicine, the researchers randomized 35 adults with GPP flares to a single 900-mg intravenous dose of spesolimab and 18 to placebo. Patients in both groups could receive an open-label dose of spesolimab after day 8; all patients were followed for 12 weeks.
The primary study endpoint was the Generalized Pustular Psoriasis Physician Global Assessment (GPPGA) pustulation subscore of 0 at 1 week after treatment. The GPPGA ranges from 0 (no visible pustules) to 4 (severe pustules). At baseline, 46% spesolimab patients and 39% placebo patients had a GPPGA pustulation subscore of 3, and 37% and 33%, respectively, had a pustulation subscore of 4.
After 1 week, 54% of the spesolimab patients had no visible pustules, compared with 6% of placebo patients; the difference was statistically significant (P < .001). The main secondary endpoint was a score of 0 or 1 (clear or almost clear skin) on the GPPGA total score after 1 week. Significantly more spesolimab patients had GPPGA total scores of 0 or 1, compared with placebo patients (43% vs. 11%, respectively; P = .02).
Overall, 6 of 35 spesolimab patients (17%) and 6% of those in the placebo groups developed infections during the first week, and 24 of 51 patients (47%) who had received spesolimab at any point during the study developed infections by week 12. Infections included urinary tract infections (three cases), influenza (three), otitis externa (two), folliculitis (two), upper respiratory tract infection (two), and pustule (two).
In the first week, 6% of spesolimab patients and none of the placebo patients reported serious adverse events; at week 12, 12% of patients who had received at least one spesolimab dose reported a serious adverse event. In addition, antidrug antibodies were identified in 23 (46%) of the 50 patients who received at least one dose of spesolimab.
“Symptoms that were observed in two patients who received spesolimab were reported as a drug reaction with eosinophilia and systemic symptoms (DRESS),” the authors noted. One patient had a RegiSCAR (European Registry of Severe Cutaneous Adverse Reactions) score and the other had a score of 3; a score below 2 indicates no DRESS, and a score of 2 or 3 indicates “possible DRESS,” they added.
“Because 15 of the 18 patients who were assigned to the placebo group received open-label spesolimab, the effect of spesolimab as compared with that of placebo could not be determined after week 1,” the researchers noted.
The study findings were limited by several factors including the short randomization period and small study population, the researchers noted. However, the effect sizes for both the primary and secondary endpoints were large, which strengthened the results.
The results support data from previous studies suggesting a role for IL-36 in the pathogenesis of GPP, and support the need for longer and larger studies of the safety and effectiveness of spesolimab for GPP patients, they concluded.
No FDA-approved therapy
“GPP is a very rare but devastating life-threatening disease that presents with the sudden onset of pustules throughout the skin,” Joel Gelfand, MD, professor of dermatology and director of the psoriasis and phototherapy center at the University of Pennsylvania, Philadelphia, said in an interview. “Without rapid treatment, GPP can result in death. Currently there are no [Food and Drug Administration]–approved treatments for this orphan disease.”
Dr. Gelfand said he was surprised by the degree of efficacy and the speed of the patient response to spesolimab, compared with placebo, which he described as “truly remarkable.” Based on the current study results, “spesolimab offers a tremendous step forward for our patients,” he added.
Looking ahead, Dr. Gelfand noted that “longer-term studies with a comparator, such as a biologic that targets IL-17, would be helpful to more fully understand the safety, efficacy, and role that spesolimab will have in real-world patients.”
On Dec. 15, Boehringer Ingelheim announced that the FDA had granted priority review for spesolimab for treating GPP flares.
The study was supported by Boehringer Ingelheim. Lead author Dr. Bachelez had no financial conflicts to disclose. Several authors are employees of Boehringer Ingelheim. Dr. Gelfand is a consultant for the study sponsor Boehringer Ingelheim and has received research grants from Boehringer Ingelheim to his institution to support an investigator-initiated study. He also disclosed serving as a consultant and receiving research grants from other manufacturers of psoriasis products.
GPP is a life-threatening skin condition involving the widespread eruption of sterile pustules, with a clinical course that “can be relapsing with recurrent flares or persistent with intermittent flares,” Hervé Bachelez, MD, of the Université de Paris and coauthors wrote. GPP patients are often hospitalized, and mortality ranges from 2% to 16% from causes that include sepsis and cardiorespiratory failure.
“The role of the interleukin-36 pathway in GPP is supported by the finding of loss-of-function mutations in the interleukin-36 receptor antagonist gene (IL36RN) and associated genes (CARD14, AP1S3, SERPINA3, and MPO) and by the overexpression of interleukin-36 cytokines in GPP skin lesions,” therefore, IL-36 is a potential treatment target to manage flares, they explained.
In the multicenter, double-blind trial, published in the New England Journal of Medicine, the researchers randomized 35 adults with GPP flares to a single 900-mg intravenous dose of spesolimab and 18 to placebo. Patients in both groups could receive an open-label dose of spesolimab after day 8; all patients were followed for 12 weeks.
The primary study endpoint was the Generalized Pustular Psoriasis Physician Global Assessment (GPPGA) pustulation subscore of 0 at 1 week after treatment. The GPPGA ranges from 0 (no visible pustules) to 4 (severe pustules). At baseline, 46% spesolimab patients and 39% placebo patients had a GPPGA pustulation subscore of 3, and 37% and 33%, respectively, had a pustulation subscore of 4.
After 1 week, 54% of the spesolimab patients had no visible pustules, compared with 6% of placebo patients; the difference was statistically significant (P < .001). The main secondary endpoint was a score of 0 or 1 (clear or almost clear skin) on the GPPGA total score after 1 week. Significantly more spesolimab patients had GPPGA total scores of 0 or 1, compared with placebo patients (43% vs. 11%, respectively; P = .02).
Overall, 6 of 35 spesolimab patients (17%) and 6% of those in the placebo groups developed infections during the first week, and 24 of 51 patients (47%) who had received spesolimab at any point during the study developed infections by week 12. Infections included urinary tract infections (three cases), influenza (three), otitis externa (two), folliculitis (two), upper respiratory tract infection (two), and pustule (two).
In the first week, 6% of spesolimab patients and none of the placebo patients reported serious adverse events; at week 12, 12% of patients who had received at least one spesolimab dose reported a serious adverse event. In addition, antidrug antibodies were identified in 23 (46%) of the 50 patients who received at least one dose of spesolimab.
“Symptoms that were observed in two patients who received spesolimab were reported as a drug reaction with eosinophilia and systemic symptoms (DRESS),” the authors noted. One patient had a RegiSCAR (European Registry of Severe Cutaneous Adverse Reactions) score and the other had a score of 3; a score below 2 indicates no DRESS, and a score of 2 or 3 indicates “possible DRESS,” they added.
“Because 15 of the 18 patients who were assigned to the placebo group received open-label spesolimab, the effect of spesolimab as compared with that of placebo could not be determined after week 1,” the researchers noted.
The study findings were limited by several factors including the short randomization period and small study population, the researchers noted. However, the effect sizes for both the primary and secondary endpoints were large, which strengthened the results.
The results support data from previous studies suggesting a role for IL-36 in the pathogenesis of GPP, and support the need for longer and larger studies of the safety and effectiveness of spesolimab for GPP patients, they concluded.
No FDA-approved therapy
“GPP is a very rare but devastating life-threatening disease that presents with the sudden onset of pustules throughout the skin,” Joel Gelfand, MD, professor of dermatology and director of the psoriasis and phototherapy center at the University of Pennsylvania, Philadelphia, said in an interview. “Without rapid treatment, GPP can result in death. Currently there are no [Food and Drug Administration]–approved treatments for this orphan disease.”
Dr. Gelfand said he was surprised by the degree of efficacy and the speed of the patient response to spesolimab, compared with placebo, which he described as “truly remarkable.” Based on the current study results, “spesolimab offers a tremendous step forward for our patients,” he added.
Looking ahead, Dr. Gelfand noted that “longer-term studies with a comparator, such as a biologic that targets IL-17, would be helpful to more fully understand the safety, efficacy, and role that spesolimab will have in real-world patients.”
On Dec. 15, Boehringer Ingelheim announced that the FDA had granted priority review for spesolimab for treating GPP flares.
The study was supported by Boehringer Ingelheim. Lead author Dr. Bachelez had no financial conflicts to disclose. Several authors are employees of Boehringer Ingelheim. Dr. Gelfand is a consultant for the study sponsor Boehringer Ingelheim and has received research grants from Boehringer Ingelheim to his institution to support an investigator-initiated study. He also disclosed serving as a consultant and receiving research grants from other manufacturers of psoriasis products.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
AD burden may be greater for those with head, neck, face, and hand involvement
of patients with AD.
“While we know that head, neck, face, and hands seem to be significantly affected by patients with AD, there is a limited evidence basis regarding the prevalence and health-related quality of life impact of AD in these areas,” presenting author Lawrence F. Eichenfield, MD, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis symposium.
For the study, Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and colleagues evaluated 533 patients from the TARGET-DERM AD cohort, an ongoing, longitudinal, observational study launched in 2019 that captures patients with AD in 44 community or academic sites in the United States.
Adult, adolescent, and pediatric patients with moderate or severe Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) scores at enrollment were included in the analysis. The researchers used the Patient-Oriented Scoring AD (PO-SCORAD) index to gather information on involvement of the head, neck, face, hands, or other areas, and the Patient-Oriented Eczema Measure (POEM) and Dermatology Life Quality Index/Children’s DLQI (CDLQI) to measure health-related quality of life outcomes.
Of the 533 study participants, 453 (85%) had AD affecting the head, neck, face, hands, and other areas, while 80 (15%) had AD located in other body regions not including the head, neck, face, or hands. About 38% of all patients were using systemic treatments; most were using topical treatments.
Comorbid immune system disorders (including allergic and hypersensitivity disorders) were noted in 44.8% of patients, infections in 32.5%, asthma in 26.5%, hypertension in 18.6%, depression in 15.8%, and anxiety in 12.4%, with similar proportions observed in those with or without head, neck, face, and hand involvement.
However, patients with head, face, neck, and hand involvement, when compared with patients without those affected areas, were more likely to have severe vIGA scores (28.5% vs. 16.3%, P = .02) and a higher median total body surface area affected (15% vs. 10%, P ≤ .01). Also, while bivariable analyses did not detect statistical differences in POEM and DLQI/CDLQI by body region involvement, multivariable-adjusted models showed that patients with head, neck, face, and hand involvement were more than twice as likely to report higher DLQI/CDLQI (odds ratio, 2.09) and POEM (OR, 2.51) scores than those without head, face, neck, and hand involvement.
“These findings highlight the importance of detailed assessment of specific areas affected by AD to personalize treatment approaches to the needs of patients,” Dr. Eichenfield concluded.
Raj Chovatiya MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that the findings confirm clinical suspicions about the unique and heightened impact of facial, head/neck, and hand dermatitis. “These data show that a detailed skin examination is necessary for a complete assessment of AD,” he said. “Future studies should focus on characterizing the optimal treatment approaches for each of these special sites.”
“This is important data,” added primary study author Jonathan I. Silverberg, MD, PhD, MPH, director of clinical research in the division of dermatology at George Washington University, Washington. “We need more high-quality studies like this; we need to create long-term longitudinal data to better understand [the impact of AD on] this and other cohorts.”
TARGET-DERM is sponsored by Target RWE. Dr. Eichenfield disclosed that he has served as a consultant to or investigator for numerous pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme. Dr. Silverberg disclosed that he is a consultant to numerous pharmaceutical companies, receives fees for non-CME/CE services from Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi Genzyme, as well as contracted research fees from Galderma.
Commentary by Robert Sidbury, MD, MPH
Patients with atopic dermatitis (AD) in “visible” areas such as the head, neck, and hands experience a higher impact on their quality of life than those who do not have these areas of involvement. This is a self-evident and unsurprising result but also a particularly important one to document for several reasons. First, evidence-based demonstration of quality-of-life impact is critical as we petition carriers to support the use of newer, more expensive medications. Second, from a topical therapy standpoint, we often use different medications on the head, neck, face, and hands relative to other areas. On the head and neck area we often use either weaker topical steroids to avoid side effects or nonsteroids like topical calcineurin or phosphodiesterase inhibitors; conversely, on the hands we use stronger steroids and are less likely to use nonsteroidal agents that are perceived to be less potent. These data emphasize the need to tailor therapy but ascertain whether standard approaches are satisfactory. If patients are not responding, particularly in these sensitive areas, providers should consider the outsized impact AD may be having on quality of life.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/10/22.
of patients with AD.
“While we know that head, neck, face, and hands seem to be significantly affected by patients with AD, there is a limited evidence basis regarding the prevalence and health-related quality of life impact of AD in these areas,” presenting author Lawrence F. Eichenfield, MD, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis symposium.
For the study, Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and colleagues evaluated 533 patients from the TARGET-DERM AD cohort, an ongoing, longitudinal, observational study launched in 2019 that captures patients with AD in 44 community or academic sites in the United States.
Adult, adolescent, and pediatric patients with moderate or severe Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) scores at enrollment were included in the analysis. The researchers used the Patient-Oriented Scoring AD (PO-SCORAD) index to gather information on involvement of the head, neck, face, hands, or other areas, and the Patient-Oriented Eczema Measure (POEM) and Dermatology Life Quality Index/Children’s DLQI (CDLQI) to measure health-related quality of life outcomes.
Of the 533 study participants, 453 (85%) had AD affecting the head, neck, face, hands, and other areas, while 80 (15%) had AD located in other body regions not including the head, neck, face, or hands. About 38% of all patients were using systemic treatments; most were using topical treatments.
Comorbid immune system disorders (including allergic and hypersensitivity disorders) were noted in 44.8% of patients, infections in 32.5%, asthma in 26.5%, hypertension in 18.6%, depression in 15.8%, and anxiety in 12.4%, with similar proportions observed in those with or without head, neck, face, and hand involvement.
However, patients with head, face, neck, and hand involvement, when compared with patients without those affected areas, were more likely to have severe vIGA scores (28.5% vs. 16.3%, P = .02) and a higher median total body surface area affected (15% vs. 10%, P ≤ .01). Also, while bivariable analyses did not detect statistical differences in POEM and DLQI/CDLQI by body region involvement, multivariable-adjusted models showed that patients with head, neck, face, and hand involvement were more than twice as likely to report higher DLQI/CDLQI (odds ratio, 2.09) and POEM (OR, 2.51) scores than those without head, face, neck, and hand involvement.
“These findings highlight the importance of detailed assessment of specific areas affected by AD to personalize treatment approaches to the needs of patients,” Dr. Eichenfield concluded.
Raj Chovatiya MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that the findings confirm clinical suspicions about the unique and heightened impact of facial, head/neck, and hand dermatitis. “These data show that a detailed skin examination is necessary for a complete assessment of AD,” he said. “Future studies should focus on characterizing the optimal treatment approaches for each of these special sites.”
“This is important data,” added primary study author Jonathan I. Silverberg, MD, PhD, MPH, director of clinical research in the division of dermatology at George Washington University, Washington. “We need more high-quality studies like this; we need to create long-term longitudinal data to better understand [the impact of AD on] this and other cohorts.”
TARGET-DERM is sponsored by Target RWE. Dr. Eichenfield disclosed that he has served as a consultant to or investigator for numerous pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme. Dr. Silverberg disclosed that he is a consultant to numerous pharmaceutical companies, receives fees for non-CME/CE services from Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi Genzyme, as well as contracted research fees from Galderma.
Commentary by Robert Sidbury, MD, MPH
Patients with atopic dermatitis (AD) in “visible” areas such as the head, neck, and hands experience a higher impact on their quality of life than those who do not have these areas of involvement. This is a self-evident and unsurprising result but also a particularly important one to document for several reasons. First, evidence-based demonstration of quality-of-life impact is critical as we petition carriers to support the use of newer, more expensive medications. Second, from a topical therapy standpoint, we often use different medications on the head, neck, face, and hands relative to other areas. On the head and neck area we often use either weaker topical steroids to avoid side effects or nonsteroids like topical calcineurin or phosphodiesterase inhibitors; conversely, on the hands we use stronger steroids and are less likely to use nonsteroidal agents that are perceived to be less potent. These data emphasize the need to tailor therapy but ascertain whether standard approaches are satisfactory. If patients are not responding, particularly in these sensitive areas, providers should consider the outsized impact AD may be having on quality of life.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/10/22.
of patients with AD.
“While we know that head, neck, face, and hands seem to be significantly affected by patients with AD, there is a limited evidence basis regarding the prevalence and health-related quality of life impact of AD in these areas,” presenting author Lawrence F. Eichenfield, MD, said during a late-breaking abstract session at the Revolutionizing Atopic Dermatitis symposium.
For the study, Dr. Eichenfield, professor of dermatology and pediatrics at the University of California, San Diego, and colleagues evaluated 533 patients from the TARGET-DERM AD cohort, an ongoing, longitudinal, observational study launched in 2019 that captures patients with AD in 44 community or academic sites in the United States.
Adult, adolescent, and pediatric patients with moderate or severe Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) scores at enrollment were included in the analysis. The researchers used the Patient-Oriented Scoring AD (PO-SCORAD) index to gather information on involvement of the head, neck, face, hands, or other areas, and the Patient-Oriented Eczema Measure (POEM) and Dermatology Life Quality Index/Children’s DLQI (CDLQI) to measure health-related quality of life outcomes.
Of the 533 study participants, 453 (85%) had AD affecting the head, neck, face, hands, and other areas, while 80 (15%) had AD located in other body regions not including the head, neck, face, or hands. About 38% of all patients were using systemic treatments; most were using topical treatments.
Comorbid immune system disorders (including allergic and hypersensitivity disorders) were noted in 44.8% of patients, infections in 32.5%, asthma in 26.5%, hypertension in 18.6%, depression in 15.8%, and anxiety in 12.4%, with similar proportions observed in those with or without head, neck, face, and hand involvement.
However, patients with head, face, neck, and hand involvement, when compared with patients without those affected areas, were more likely to have severe vIGA scores (28.5% vs. 16.3%, P = .02) and a higher median total body surface area affected (15% vs. 10%, P ≤ .01). Also, while bivariable analyses did not detect statistical differences in POEM and DLQI/CDLQI by body region involvement, multivariable-adjusted models showed that patients with head, neck, face, and hand involvement were more than twice as likely to report higher DLQI/CDLQI (odds ratio, 2.09) and POEM (OR, 2.51) scores than those without head, face, neck, and hand involvement.
“These findings highlight the importance of detailed assessment of specific areas affected by AD to personalize treatment approaches to the needs of patients,” Dr. Eichenfield concluded.
Raj Chovatiya MD, PhD, assistant professor of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that the findings confirm clinical suspicions about the unique and heightened impact of facial, head/neck, and hand dermatitis. “These data show that a detailed skin examination is necessary for a complete assessment of AD,” he said. “Future studies should focus on characterizing the optimal treatment approaches for each of these special sites.”
“This is important data,” added primary study author Jonathan I. Silverberg, MD, PhD, MPH, director of clinical research in the division of dermatology at George Washington University, Washington. “We need more high-quality studies like this; we need to create long-term longitudinal data to better understand [the impact of AD on] this and other cohorts.”
TARGET-DERM is sponsored by Target RWE. Dr. Eichenfield disclosed that he has served as a consultant to or investigator for numerous pharmaceutical companies. Dr. Chovatiya disclosed that he is a consultant to, a speaker for, and/or a member of the advisory board for AbbVie, Arena, Arcutis, Incyte, Pfizer, Regeneron, and Sanofi-Genzyme. Dr. Silverberg disclosed that he is a consultant to numerous pharmaceutical companies, receives fees for non-CME/CE services from Eli Lilly, Leo Pharma, Pfizer, Regeneron, and Sanofi Genzyme, as well as contracted research fees from Galderma.
Commentary by Robert Sidbury, MD, MPH
Patients with atopic dermatitis (AD) in “visible” areas such as the head, neck, and hands experience a higher impact on their quality of life than those who do not have these areas of involvement. This is a self-evident and unsurprising result but also a particularly important one to document for several reasons. First, evidence-based demonstration of quality-of-life impact is critical as we petition carriers to support the use of newer, more expensive medications. Second, from a topical therapy standpoint, we often use different medications on the head, neck, face, and hands relative to other areas. On the head and neck area we often use either weaker topical steroids to avoid side effects or nonsteroids like topical calcineurin or phosphodiesterase inhibitors; conversely, on the hands we use stronger steroids and are less likely to use nonsteroidal agents that are perceived to be less potent. These data emphasize the need to tailor therapy but ascertain whether standard approaches are satisfactory. If patients are not responding, particularly in these sensitive areas, providers should consider the outsized impact AD may be having on quality of life.
Dr. Sidbury is chief of dermatology at Seattle Children's Hospital and professor, department of pediatrics, University of Washington, Seattle. He is a site principal investigator for dupilumab trials, for which the hospital has a contract with Regeneron.
A version of this article first appeared on Medscape.com.
This article was updated 6/10/22.
FROM REVOLUTIONIZING AD 2021